JP2012201762A - Aqueous printing composition for lamination - Google Patents
Aqueous printing composition for lamination Download PDFInfo
- Publication number
- JP2012201762A JP2012201762A JP2011066707A JP2011066707A JP2012201762A JP 2012201762 A JP2012201762 A JP 2012201762A JP 2011066707 A JP2011066707 A JP 2011066707A JP 2011066707 A JP2011066707 A JP 2011066707A JP 2012201762 A JP2012201762 A JP 2012201762A
- Authority
- JP
- Japan
- Prior art keywords
- water
- ink composition
- lamination
- based ink
- laminating
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 43
- 238000007639 printing Methods 0.000 title claims abstract description 33
- 238000003475 lamination Methods 0.000 title claims abstract description 14
- 239000004743 Polypropylene Substances 0.000 claims abstract description 63
- -1 silicon oxide compound Chemical class 0.000 claims abstract description 56
- 229920001155 polypropylene Polymers 0.000 claims abstract description 49
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 48
- 229920005989 resin Polymers 0.000 claims abstract description 37
- 239000011347 resin Substances 0.000 claims abstract description 37
- 239000006185 dispersion Substances 0.000 claims abstract description 16
- 239000002985 plastic film Substances 0.000 claims abstract description 16
- 229920006255 plastic film Polymers 0.000 claims abstract description 14
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims abstract description 8
- OGIDPMRJRNCKJF-UHFFFAOYSA-N titanium oxide Inorganic materials [Ti]=O OGIDPMRJRNCKJF-UHFFFAOYSA-N 0.000 claims abstract description 8
- 238000010030 laminating Methods 0.000 claims description 30
- 150000003961 organosilicon compounds Chemical class 0.000 claims description 13
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 claims description 9
- 125000000524 functional group Chemical group 0.000 claims description 8
- 239000000758 substrate Substances 0.000 claims description 8
- 229920002803 thermoplastic polyurethane Polymers 0.000 claims description 8
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 claims description 7
- 239000011976 maleic acid Substances 0.000 claims description 7
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 claims description 7
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 claims description 6
- 125000003277 amino group Chemical group 0.000 claims description 5
- 125000003118 aryl group Chemical group 0.000 claims description 5
- 125000003700 epoxy group Chemical group 0.000 claims description 5
- IQPQWNKOIGAROB-UHFFFAOYSA-N isocyanate group Chemical group [N-]=C=O IQPQWNKOIGAROB-UHFFFAOYSA-N 0.000 claims description 5
- 125000003396 thiol group Chemical group [H]S* 0.000 claims description 5
- 150000003673 urethanes Chemical class 0.000 claims description 5
- 239000000565 sealant Substances 0.000 claims description 3
- 150000003377 silicon compounds Chemical class 0.000 claims description 3
- 239000000049 pigment Substances 0.000 abstract description 38
- 239000002253 acid Substances 0.000 abstract description 6
- 239000003795 chemical substances by application Substances 0.000 abstract description 4
- 239000000463 material Substances 0.000 abstract description 3
- 238000009823 thermal lamination Methods 0.000 abstract description 3
- 229910052814 silicon oxide Inorganic materials 0.000 abstract 1
- 239000000976 ink Substances 0.000 description 49
- 238000000034 method Methods 0.000 description 18
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- 239000007787 solid Substances 0.000 description 9
- 239000005022 packaging material Substances 0.000 description 7
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 6
- 239000012860 organic pigment Substances 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 5
- 239000003973 paint Substances 0.000 description 5
- 239000004925 Acrylic resin Substances 0.000 description 4
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N Iron oxide Chemical compound [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 238000011156 evaluation Methods 0.000 description 4
- 238000001125 extrusion Methods 0.000 description 4
- 229920000098 polyolefin Polymers 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- 238000010998 test method Methods 0.000 description 4
- UUEWCQRISZBELL-UHFFFAOYSA-N 3-trimethoxysilylpropane-1-thiol Chemical compound CO[Si](OC)(OC)CCCS UUEWCQRISZBELL-UHFFFAOYSA-N 0.000 description 3
- 229920000178 Acrylic resin Polymers 0.000 description 3
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 235000013305 food Nutrition 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 3
- 238000012545 processing Methods 0.000 description 3
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 235000011114 ammonium hydroxide Nutrition 0.000 description 2
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- QDOXWKRWXJOMAK-UHFFFAOYSA-N dichromium trioxide Chemical compound O=[Cr]O[Cr]=O QDOXWKRWXJOMAK-UHFFFAOYSA-N 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 238000007646 gravure printing Methods 0.000 description 2
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 2
- 239000001023 inorganic pigment Substances 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 239000003960 organic solvent Substances 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 238000012805 post-processing Methods 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 2
- NDKWCCLKSWNDBG-UHFFFAOYSA-N zinc;dioxido(dioxo)chromium Chemical compound [Zn+2].[O-][Cr]([O-])(=O)=O NDKWCCLKSWNDBG-UHFFFAOYSA-N 0.000 description 2
- LTQBNYCMVZQRSD-UHFFFAOYSA-N (4-ethenylphenyl)-trimethoxysilane Chemical compound CO[Si](OC)(OC)C1=CC=C(C=C)C=C1 LTQBNYCMVZQRSD-UHFFFAOYSA-N 0.000 description 1
- UKXDHEBARGMWMO-ARJAWSKDSA-N (z)-4-(2-methylpropoxy)-4-oxobut-2-enoic acid Chemical compound CC(C)COC(=O)\C=C/C(O)=O UKXDHEBARGMWMO-ARJAWSKDSA-N 0.000 description 1
- JVYDLYGCSIHCMR-UHFFFAOYSA-N 2,2-bis(hydroxymethyl)butanoic acid Chemical compound CCC(CO)(CO)C(O)=O JVYDLYGCSIHCMR-UHFFFAOYSA-N 0.000 description 1
- JAHNSTQSQJOJLO-UHFFFAOYSA-N 2-(3-fluorophenyl)-1h-imidazole Chemical compound FC1=CC=CC(C=2NC=CN=2)=C1 JAHNSTQSQJOJLO-UHFFFAOYSA-N 0.000 description 1
- RNLHGQLZWXBQNY-UHFFFAOYSA-N 3-(aminomethyl)-3,5,5-trimethylcyclohexan-1-amine Chemical compound CC1(C)CC(N)CC(C)(CN)C1 RNLHGQLZWXBQNY-UHFFFAOYSA-N 0.000 description 1
- OXYZDRAJMHGSMW-UHFFFAOYSA-N 3-chloropropyl(trimethoxy)silane Chemical compound CO[Si](OC)(OC)CCCCl OXYZDRAJMHGSMW-UHFFFAOYSA-N 0.000 description 1
- SJECZPVISLOESU-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine Chemical compound CO[Si](OC)(OC)CCCN SJECZPVISLOESU-UHFFFAOYSA-N 0.000 description 1
- 239000004342 Benzoyl peroxide Substances 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- 208000031872 Body Remains Diseases 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229920000298 Cellophane Polymers 0.000 description 1
- IEPRKVQEAMIZSS-UHFFFAOYSA-N Di-Et ester-Fumaric acid Natural products CCOC(=O)C=CC(=O)OCC IEPRKVQEAMIZSS-UHFFFAOYSA-N 0.000 description 1
- IEPRKVQEAMIZSS-WAYWQWQTSA-N Diethyl maleate Chemical compound CCOC(=O)\C=C/C(=O)OCC IEPRKVQEAMIZSS-WAYWQWQTSA-N 0.000 description 1
- XLYMOEINVGRTEX-ARJAWSKDSA-N Ethyl hydrogen fumarate Chemical compound CCOC(=O)\C=C/C(O)=O XLYMOEINVGRTEX-ARJAWSKDSA-N 0.000 description 1
- JOYRKODLDBILNP-UHFFFAOYSA-N Ethyl urethane Chemical compound CCOC(N)=O JOYRKODLDBILNP-UHFFFAOYSA-N 0.000 description 1
- 235000000177 Indigofera tinctoria Nutrition 0.000 description 1
- 239000005058 Isophorone diisocyanate Substances 0.000 description 1
- SJEYSFABYSGQBG-UHFFFAOYSA-M Patent blue Chemical compound [Na+].C1=CC(N(CC)CC)=CC=C1C(C=1C(=CC(=CC=1)S([O-])(=O)=O)S([O-])(=O)=O)=C1C=CC(=[N+](CC)CC)C=C1 SJEYSFABYSGQBG-UHFFFAOYSA-M 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 229920002396 Polyurea Polymers 0.000 description 1
- NRCMAYZCPIVABH-UHFFFAOYSA-N Quinacridone Chemical compound N1C2=CC=CC=C2C(=O)C2=C1C=C1C(=O)C3=CC=CC=C3NC1=C2 NRCMAYZCPIVABH-UHFFFAOYSA-N 0.000 description 1
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 1
- 239000006087 Silane Coupling Agent Substances 0.000 description 1
- 241001122767 Theaceae Species 0.000 description 1
- GSEJCLTVZPLZKY-UHFFFAOYSA-N Triethanolamine Chemical compound OCCN(CCO)CCO GSEJCLTVZPLZKY-UHFFFAOYSA-N 0.000 description 1
- WGLPBDUCMAPZCE-UHFFFAOYSA-N Trioxochromium Chemical compound O=[Cr](=O)=O WGLPBDUCMAPZCE-UHFFFAOYSA-N 0.000 description 1
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 1
- FXEDRSGUZBCDMO-PHEQNACWSA-N [(e)-3-phenylprop-2-enoyl] (e)-3-phenylprop-2-enoate Chemical compound C=1C=CC=CC=1/C=C/C(=O)OC(=O)\C=C\C1=CC=CC=C1 FXEDRSGUZBCDMO-PHEQNACWSA-N 0.000 description 1
- YIMQCDZDWXUDCA-UHFFFAOYSA-N [4-(hydroxymethyl)cyclohexyl]methanol Chemical compound OCC1CCC(CO)CC1 YIMQCDZDWXUDCA-UHFFFAOYSA-N 0.000 description 1
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical compound CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 description 1
- 239000000980 acid dye Substances 0.000 description 1
- DGOBMKYRQHEFGQ-UHFFFAOYSA-L acid green 5 Chemical compound [Na+].[Na+].C=1C=C(C(=C2C=CC(C=C2)=[N+](CC)CC=2C=C(C=CC=2)S([O-])(=O)=O)C=2C=CC(=CC=2)S([O-])(=O)=O)C=CC=1N(CC)CC1=CC=CC(S([O-])(=O)=O)=C1 DGOBMKYRQHEFGQ-UHFFFAOYSA-L 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 238000007259 addition reaction Methods 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 125000002009 alkene group Chemical group 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 125000005907 alkyl ester group Chemical group 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 125000003046 allene group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- PYKYMHQGRFAEBM-UHFFFAOYSA-N anthraquinone Natural products CCC(=O)c1c(O)c2C(=O)C3C(C=CC=C3O)C(=O)c2cc1CC(=O)OC PYKYMHQGRFAEBM-UHFFFAOYSA-N 0.000 description 1
- 150000004056 anthraquinones Chemical class 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000010426 asphalt Substances 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- IRERQBUNZFJFGC-UHFFFAOYSA-L azure blue Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[S-]S[S-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] IRERQBUNZFJFGC-UHFFFAOYSA-L 0.000 description 1
- 150000007514 bases Chemical class 0.000 description 1
- 239000000981 basic dye Substances 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 239000001055 blue pigment Substances 0.000 description 1
- 239000001058 brown pigment Substances 0.000 description 1
- UTOVMEACOLCUCK-PLNGDYQASA-N butyl maleate Chemical compound CCCCOC(=O)\C=C/C(O)=O UTOVMEACOLCUCK-PLNGDYQASA-N 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 239000004106 carminic acid Substances 0.000 description 1
- 235000012730 carminic acid Nutrition 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000005660 chlorination reaction Methods 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229910000423 chromium oxide Inorganic materials 0.000 description 1
- FXEDRSGUZBCDMO-UHFFFAOYSA-N cinnamic acid anhydride Natural products C=1C=CC=CC=1C=CC(=O)OC(=O)C=CC1=CC=CC=C1 FXEDRSGUZBCDMO-UHFFFAOYSA-N 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 229910000152 cobalt phosphate Inorganic materials 0.000 description 1
- 229940080423 cochineal Drugs 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000005034 decoration Methods 0.000 description 1
- 230000032798 delamination Effects 0.000 description 1
- JBSLOWBPDRZSMB-FPLPWBNLSA-N dibutyl (z)-but-2-enedioate Chemical compound CCCCOC(=O)\C=C/C(=O)OCCCC JBSLOWBPDRZSMB-FPLPWBNLSA-N 0.000 description 1
- 239000012975 dibutyltin dilaurate Substances 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- LDCRTTXIJACKKU-ARJAWSKDSA-N dimethyl maleate Chemical compound COC(=O)\C=C/C(=O)OC LDCRTTXIJACKKU-ARJAWSKDSA-N 0.000 description 1
- PPSZHCXTGRHULJ-UHFFFAOYSA-N dioxazine Chemical compound O1ON=CC=C1 PPSZHCXTGRHULJ-UHFFFAOYSA-N 0.000 description 1
- 238000009820 dry lamination Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- QHZOMAXECYYXGP-UHFFFAOYSA-N ethene;prop-2-enoic acid Chemical compound C=C.OC(=O)C=C QHZOMAXECYYXGP-UHFFFAOYSA-N 0.000 description 1
- 229920006226 ethylene-acrylic acid Polymers 0.000 description 1
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- XLYMOEINVGRTEX-UHFFFAOYSA-N fumaric acid monoethyl ester Natural products CCOC(=O)C=CC(O)=O XLYMOEINVGRTEX-UHFFFAOYSA-N 0.000 description 1
- NKHAVTQWNUWKEO-UHFFFAOYSA-N fumaric acid monomethyl ester Natural products COC(=O)C=CC(O)=O NKHAVTQWNUWKEO-UHFFFAOYSA-N 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000001056 green pigment Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- 235000019239 indanthrene blue RS Nutrition 0.000 description 1
- UHOKSCJSTAHBSO-UHFFFAOYSA-N indanthrone blue Chemical compound C1=CC=C2C(=O)C3=CC=C4NC5=C6C(=O)C7=CC=CC=C7C(=O)C6=CC=C5NC4=C3C(=O)C2=C1 UHOKSCJSTAHBSO-UHFFFAOYSA-N 0.000 description 1
- 229940097275 indigo Drugs 0.000 description 1
- COHYTHOBJLSHDF-UHFFFAOYSA-N indigo powder Natural products N1C2=CC=CC=C2C(=O)C1=C1C(=O)C2=CC=CC=C2N1 COHYTHOBJLSHDF-UHFFFAOYSA-N 0.000 description 1
- PXZQEOJJUGGUIB-UHFFFAOYSA-N isoindolin-1-one Chemical compound C1=CC=C2C(=O)NCC2=C1 PXZQEOJJUGGUIB-UHFFFAOYSA-N 0.000 description 1
- NIMLQBUJDJZYEJ-UHFFFAOYSA-N isophorone diisocyanate Chemical compound CC1(C)CC(N=C=O)CC(C)(CN=C=O)C1 NIMLQBUJDJZYEJ-UHFFFAOYSA-N 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- NKHAVTQWNUWKEO-IHWYPQMZSA-N methyl hydrogen fumarate Chemical compound COC(=O)\C=C/C(O)=O NKHAVTQWNUWKEO-IHWYPQMZSA-N 0.000 description 1
- LVHBHZANLOWSRM-UHFFFAOYSA-N methylenebutanedioic acid Natural products OC(=O)CC(=C)C(O)=O LVHBHZANLOWSRM-UHFFFAOYSA-N 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 238000002715 modification method Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- KMBPCQSCMCEPMU-UHFFFAOYSA-N n'-(3-aminopropyl)-n'-methylpropane-1,3-diamine Chemical compound NCCCN(C)CCCN KMBPCQSCMCEPMU-UHFFFAOYSA-N 0.000 description 1
- OBJNZHVOCNPSCS-UHFFFAOYSA-N naphtho[2,3-f]quinazoline Chemical compound C1=NC=C2C3=CC4=CC=CC=C4C=C3C=CC2=N1 OBJNZHVOCNPSCS-UHFFFAOYSA-N 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 125000000018 nitroso group Chemical group N(=O)* 0.000 description 1
- 238000010979 pH adjustment Methods 0.000 description 1
- 238000012536 packaging technology Methods 0.000 description 1
- DGBWPZSGHAXYGK-UHFFFAOYSA-N perinone Chemical compound C12=NC3=CC=CC=C3N2C(=O)C2=CC=C3C4=C2C1=CC=C4C(=O)N1C2=CC=CC=C2N=C13 DGBWPZSGHAXYGK-UHFFFAOYSA-N 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 230000000379 polymerizing effect Effects 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920005749 polyurethane resin Polymers 0.000 description 1
- 229920003226 polyurethane urea Polymers 0.000 description 1
- LLBIOIRWAYBCKK-UHFFFAOYSA-N pyranthrene-8,16-dione Chemical compound C12=CC=CC=C2C(=O)C2=CC=C3C=C4C5=CC=CC=C5C(=O)C5=C4C4=C3C2=C1C=C4C=C5 LLBIOIRWAYBCKK-UHFFFAOYSA-N 0.000 description 1
- FYNROBRQIVCIQF-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole-5,6-dione Chemical compound C1=CN=C2C(=O)C(=O)N=C21 FYNROBRQIVCIQF-UHFFFAOYSA-N 0.000 description 1
- 239000013557 residual solvent Substances 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 238000003307 slaughter Methods 0.000 description 1
- 238000013112 stability test Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 230000002123 temporal effect Effects 0.000 description 1
- FRGPKMWIYVTFIQ-UHFFFAOYSA-N triethoxy(3-isocyanatopropyl)silane Chemical compound CCO[Si](OCC)(OCC)CCCN=C=O FRGPKMWIYVTFIQ-UHFFFAOYSA-N 0.000 description 1
- CPUDPFPXCZDNGI-UHFFFAOYSA-N triethoxy(methyl)silane Chemical compound CCO[Si](C)(OCC)OCC CPUDPFPXCZDNGI-UHFFFAOYSA-N 0.000 description 1
- JCVQKRGIASEUKR-UHFFFAOYSA-N triethoxy(phenyl)silane Chemical compound CCO[Si](OCC)(OCC)C1=CC=CC=C1 JCVQKRGIASEUKR-UHFFFAOYSA-N 0.000 description 1
- 239000000984 vat dye Substances 0.000 description 1
- 239000001052 yellow pigment Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
- 235000014692 zinc oxide Nutrition 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Landscapes
- Inks, Pencil-Leads, Or Crayons (AREA)
Abstract
Description
本発明は、ラミネート用水性インキ組成物およびそれを用いた積層物に関し、より詳しくは、各種プラスチックフィルムに適用可能で、特にポリオレフィン系フィルムに対する接着性、溶融ポリプロピレンのダイレクトラミネート適性、サーマルラミネート適性に優れたラミネート用水性インキ組成物およびそれを用いた積層物に関する。 The present invention relates to a water-based ink composition for laminating and a laminate using the same. More specifically, the present invention can be applied to various plastic films, in particular, adhesion to polyolefin films, suitability for direct lamination of molten polypropylene, and suitability for thermal lamination. The present invention relates to an excellent water-based ink composition for laminating and a laminate using the same.
近年、被包装物の多様化、包装技術の高度化に伴ない、包装材料として各種のプラスチックフィルムが開発され、被包装物に適したものが適宜選択して使用されるようになってきている。またプラスチックフィルムを包装材料に使用するにあたってはプラスチックフィルムの装飾または表面保護のために印刷が施されるが、かかる印刷のための印刷インキには、これら種々のプラスチックフィルムに対する高度な性能、品質が要求されるようになってきている。とりわけ印刷インキに関しては包装容器の美粧化、高級化のために各種複合フィルムに対する幅広い接着性、さらにはラミネート加工適性、ボイル適性、レトルト適性等の各種後加工適性が必要になってきている。 In recent years, with the diversification of packaging materials and the advancement of packaging technology, various plastic films have been developed as packaging materials, and those suitable for packaging materials have been appropriately selected and used. . In addition, when plastic films are used for packaging materials, printing is performed for the decoration or surface protection of the plastic film. The printing ink for such printing has high performance and quality for these various plastic films. It is becoming required. In particular, printing inks are required to have various adhesive properties for various composite films and various post-processing aptitudes such as laminating suitability, boil suitability, and retort suitability in order to beautify and upgrade packaging containers.
また、包装材料の高性能化のため、印刷後にドライラミネート加工、エクストルージョンラミネート加工を施すことがある。この場合ドライラミネート加工にはウレタン系等の接着剤を使用し、エクストルージョンラミネート加工の場合はアンカーコート剤を使用する。さらに中間にアルミニウム箔を介在させることもあり、ボイル、レトルト加工が可能な包装材料とすることもある。またレトルト加工までの強度は要求されないが透明基材をベースとした包装材料分野においてはコロナ放電処理延伸ポリプロピレン(OPP)を基材フィルムとし、アンカーコート剤は用いずに、直接、溶融ポリプロピレンで被覆するラミネート加工方法(PPダイレクトラミネート)、溶融ポリマーを用いず、熱圧着により基材フィルムとシーラントフィルムとを直接ラミネートする加工方法(PPサーマルラミネート)も行われている。 Moreover, in order to improve the performance of packaging materials, dry lamination processing and extrusion lamination processing may be performed after printing. In this case, an adhesive such as urethane is used for the dry laminating process, and an anchor coat agent is used for the extrusion laminating process. Furthermore, an aluminum foil may be interposed in the middle, and the packaging material may be boiled or retort processed. Although strength is not required until retorting, in the packaging material field based on transparent substrates, corona discharge-treated expanded polypropylene (OPP) is used as the base film, and it is directly coated with molten polypropylene without using an anchor coat agent. A laminating method (PP direct laminating) and a laminating method (PP thermal laminating) in which a base film and a sealant film are directly laminated by thermocompression bonding without using a molten polymer are also performed.
かかる印刷後に加工を行うためには、その前段階で用いる印刷インキに対して、種々の基材フィルムに対する接着性、印刷適性はもとより、それぞれの後加工に対する適性を具備していることが要求される。PPダイレクトラミネート、PPサーマルラミネートが施されるポリオレフィンに対しては、塩素化ポリプロピレン等の高塩素化度の塩素化ポリオレフィンをバインダーとした溶剤型印刷インキがもっぱら使用されている。しかしながら、これら溶剤型インキではインキ中および希釈溶剤中に多量の有機溶剤を含有しているため、印刷作業環境中に溶剤蒸気が存在するようになり、作業者に対して健康上好ましくない影響を与えているばかりでなく、火災の危険性もあり好ましくない。また溶剤型インキを使用した場合、人体に有害な有機溶剤がラミネート物中に残留するため、食品包装に用いた場合には食品に残留溶剤が移行し易く、食品衛生上好ましくない。 In order to perform processing after such printing, it is required that the printing ink used in the previous stage has not only adhesiveness to various substrate films and printability but also suitability for each post-processing. The For polyolefins to which PP direct lamination and PP thermal lamination are applied, solvent-based printing inks using a chlorinated polyolefin having a high chlorination degree such as chlorinated polypropylene as a binder are exclusively used. However, since these solvent-based inks contain a large amount of organic solvent in the ink and the diluting solvent, solvent vapor is present in the printing work environment, which has an adverse effect on the health. Not only is it giving, but there is also a risk of fire, which is undesirable. In addition, when solvent-based ink is used, an organic solvent harmful to the human body remains in the laminate, and when used in food packaging, the residual solvent tends to migrate to food, which is not preferable for food hygiene.
特開平4−366180号公報には、1,000〜10,000の分子量を有し、かつ酸価が20〜100、軟化点が100〜200℃の酸化ポリプロピレンを、インキ固型分に対し1.0重量%〜10.0重量%含む水性インキを使用することで、コロナ放電処理延伸ポリプロピレンを基材フィルムとした印刷面にアンカーコート剤なしでポリプロピレンを溶融押出しラミネート加工する方法が開示されている。
また、特開平08−269381号公報には印刷インキのバインダー樹脂成分として、水溶性および/または水分散性樹脂および/またはその中間的なハイドロゾル型樹脂を用い、かつインキ中の樹脂固型分に対する塩素化ポリプロピレンの含有量が2.0〜100.0重量%であることを特徴とするポリプロピレン用水性印刷インキが開示されている。
JP-A-4-366180 discloses polypropylene oxide having a molecular weight of 1,000 to 10,000, an acid value of 20 to 100 and a softening point of 100 to 200 ° C. Disclosed is a method of melt-extrusion polypropylene without an anchor coating agent on a printing surface using a corona discharge-treated stretched polypropylene as a base film by using a water-based ink containing 0.0 wt% to 10.0 wt%. Yes.
Japanese Patent Application Laid-Open No. 08-269381 uses a water-soluble and / or water-dispersible resin and / or an intermediate hydrosol-type resin as a binder resin component of printing ink, and is used for a resin solid component in the ink. An aqueous printing ink for polypropylene characterized in that the content of chlorinated polypropylene is 2.0 to 100.0% by weight is disclosed.
さらに、特開平09−31386号公報には水性アクリル樹脂またはエチレンアクリル酸系樹脂が固形分で50〜95重量%、分子量2,000〜200、000のマレイン酸変性ポリプロピレンが5〜50重量%、その他の樹脂が0〜30重量%であることを特徴とする水性印刷インキ組成物が開示されている。 Furthermore, JP 09-31386 A discloses an aqueous acrylic resin or ethylene acrylic acid resin in a solid content of 50 to 95% by weight, a maleic acid-modified polypropylene having a molecular weight of 2,000 to 200,000, 5 to 50% by weight, An aqueous printing ink composition characterized in that the other resin is 0 to 30% by weight is disclosed.
しかしながら何れの発明も、基材密着性を塩素化ポリプロピレンやマレイン酸変性ポリプロピレンにのみ頼っているため、十分なラミネート強度を得ることが出来なかった。
この様な理由から、水性であって、かつ、幅広い種類のプラスチックフィルムに対する接着性、PPダイレクトラミネート適性、サーマルラミネート適性、印刷適性の良好な水性インキ組成物は未だ得られていないのが現状である。
However, in any of the inventions, since the substrate adhesion depends only on chlorinated polypropylene or maleic acid-modified polypropylene, sufficient laminate strength cannot be obtained.
For these reasons, water-based ink compositions that are water-based and have good adhesion to a wide variety of plastic films, PP direct laminate suitability, thermal laminate suitability, and printability have not yet been obtained. is there.
本発明の目的は上記問題を解決することであり、水性印刷インキでありながら幅広い種類のプラスチックフィルムに良好な接着性、ラミネート強度を示し、さらにPPダイレクトラミネート加工適性、サーマルラミネート加工適性の優れたラミネート用水性インキ組成物、およびそれを用いた積層物を提供することにある。 The object of the present invention is to solve the above-mentioned problems, and although it is a water-based printing ink, it exhibits good adhesion and laminating strength to a wide variety of plastic films, and also has excellent PP direct laminating suitability and thermal laminating suitability. An object of the present invention is to provide a water-based ink composition for laminating, and a laminate using the same.
本発明者等は、鋭意検討を行った結果、酸化チタン顔料、酸変性ポリプロピレン樹脂およびその他水性樹脂を含むラミネート用水性インキ組成物に、基材密着性向上剤として働き得る有機ケイ素酸化合物を加えることで、ラミネート強度を大幅に向上させことが可能であることを見出し、本発明を完成させるに至った。 As a result of intensive studies, the present inventors have added an organosilicon acid compound that can serve as a substrate adhesion improver to a water-based ink composition for a laminate containing a titanium oxide pigment, an acid-modified polypropylene resin, and other aqueous resins. As a result, it was found that the laminate strength can be greatly improved, and the present invention has been completed.
すなわち、本発明は、酸変性ポリプロピレン樹脂水分散体(A)、有機ケイ素化合物(B)、酸化チタン(C)および水性樹脂(D)を含有するラミネート用水性インキ組成物であって、
酸変性ポリプロピレン樹脂水分散体(A)が、数平均分子量10000〜200,000のポリプロピレンを、マレイン酸および/または無水マレイン酸で、変性したものであることを特徴とするラミネート用水性インキ組成物に関するものである。
That is, the present invention is a water-based ink composition for laminating comprising an acid-modified polypropylene resin aqueous dispersion (A), an organosilicon compound (B), titanium oxide (C) and an aqueous resin (D),
The water-based ink composition for laminating, wherein the acid-modified polypropylene resin aqueous dispersion (A) is obtained by modifying polypropylene having a number average molecular weight of 10,000 to 200,000 with maleic acid and / or maleic anhydride. It is about.
さらに、本発明は、
有機ケイ素化合物(B)が、
加水分解性の官能基
ならびに
イソシアネート基、アミノ基、エポキシ基、アリール基およびメルカプト基から選 ばれる少なくとも1種の有機官能基
を有することを特徴とする上記のラミネート用水性インキ組成物に関するものである。
Furthermore, the present invention provides
The organosilicon compound (B)
The present invention relates to a water-based ink composition for a laminate as described above, which has a hydrolyzable functional group and at least one organic functional group selected from an isocyanate group, an amino group, an epoxy group, an aryl group and a mercapto group. .
また、本発明は、ラミネート用水性インキ組成物が、有機ケイ素化合物(B)を、ラミネート用水性インキ組成物全量に対して、0.02〜1重量%含有することを特徴とする上記のラミネート用水性インキ組成物に関するものである。 Further, the present invention provides the laminate, wherein the water-based ink composition for laminating contains the organic silicon compound (B) in an amount of 0.02 to 1% by weight based on the total amount of the water-based ink composition for laminating. The present invention relates to a water-based ink composition.
さらに、本発明は、水性樹脂(D)が、アクリル変性ウレタン樹脂であることを特徴とする上記のラミネート用水性インキ組成物に関するものである。 Furthermore, the present invention relates to the above-mentioned water-based ink composition for laminate, wherein the water-based resin (D) is an acrylic-modified urethane resin.
また、本発明は、上記のラミネート用水性インキ組成物を、プラスチックフィルム基材上に、印刷してなることを特徴とする印刷物に関するものである。 The present invention also relates to a printed matter obtained by printing the above water-based ink composition for laminating on a plastic film substrate.
さらに、本発明は、上記の印刷物に、シーラントフィルムまたはプラスチックシートを積層することを特徴とする積層物に関するものである。 Furthermore, this invention relates to the laminated body characterized by laminating | stacking a sealant film or a plastic sheet on said printed matter.
本発明により、水性印刷インキでありながら幅広い種類のプラスチックフィルムに良好な接着性、ラミネート強度を示し、さらにPPダイレクトラミネート加工適性、サーマルラミネート加工適性の優れたラミネート用水性インキ組成物、およびそれを用いた積層物を提供することができる。 According to the present invention, a water-based ink composition for laminating that exhibits good adhesion and laminating strength to a wide variety of plastic films while being water-based printing ink, and has excellent PP direct laminating suitability and thermal laminating suitability, and The laminate used can be provided.
次に、本発明の水性バインダーに用いられる酸変性ポリプロピレン樹脂水分散体(A)について説明する。酸変性ポリプロピレン樹脂水分散体(A)とは、マレイン酸等の共役二重結合を有する二塩基酸で変性した変性ポリプロピレン系樹脂を、水中に分散せしめて得られるエマルジョンである。ポリプロピレンの数平均分子量としては、水性インキ組成物のプラスチックフィルムに対する接着性やダイレクトラミネート適性、耐ブロッキング性の低下を防ぐ観点から重量平均分子量は10000以上であることが必要であり、さらに好ましくは20000以上であることが望ましい。さらに、インキ組成物の経時安定性の観点からは200000以下であることが必要であり、さらに、好ましくは、100000以下であることが望ましい。 Next, the acid-modified polypropylene resin aqueous dispersion (A) used for the aqueous binder of the present invention will be described. The acid-modified polypropylene resin aqueous dispersion (A) is an emulsion obtained by dispersing a modified polypropylene resin modified with a dibasic acid having a conjugated double bond such as maleic acid in water. As the number average molecular weight of polypropylene, the weight average molecular weight is required to be 10,000 or more from the viewpoint of preventing the adhesiveness of the water-based ink composition to the plastic film, suitability for direct lamination, and blocking resistance, and more preferably 20000. The above is desirable. Furthermore, from the viewpoint of the temporal stability of the ink composition, it is necessary that it is 200,000 or less, more preferably 100,000 or less.
共役二重結合を有する二塩基酸としては、例えばマレイン酸、無水マレイン酸、イタコン酸、無水ケイヒ酸等が挙げられる。さらにこれらの二塩基酸のアルキルエステル等も使用することができる。具体的には、マレイン酸モノメチル、マレイン酸モノエチル、マレイン酸モノブチル、マレイン酸イソブチル等のマレイン酸ハーフエステル類、マレイン酸ジメチル、マレイン酸ジエチル、マレイン酸ジブチル等のマレイン酸ジエステル等が挙げられる。本発明においては、ポリプロピレン変性時の安定性の点でマレイン酸および/または無水マレイン酸で変性することが好ましい。さらにコストの点では無水マレイン酸で変性することが好ましい。 Examples of the dibasic acid having a conjugated double bond include maleic acid, maleic anhydride, itaconic acid, and cinnamic anhydride. Furthermore, alkyl esters of these dibasic acids can also be used. Specific examples include maleic acid half esters such as monomethyl maleate, monoethyl maleate, monobutyl maleate and isobutyl maleate, and maleic acid diesters such as dimethyl maleate, diethyl maleate and dibutyl maleate. In the present invention, it is preferable to modify with maleic acid and / or maleic anhydride from the viewpoint of stability when modified with polypropylene. Further, in terms of cost, it is preferable to modify with maleic anhydride.
変性方法については、従来公知の方法を用いることができる。例えば、高分子量のポリプロピレンを熱分解して得られる、二重結合を有するポリプロピレンに無水マレイン酸系モノマーをラジカル重合する方法、特開昭54−15098号公報に開示される、触媒を用いてポリプロピレンのα−オレフィン鎖を異性化させて無水マレイン酸を付加反応する方法等が挙げられる。 As the modification method, a conventionally known method can be used. For example, a method of radically polymerizing a maleic anhydride monomer to a polypropylene having a double bond obtained by thermally decomposing a high molecular weight polypropylene, and a polypropylene using a catalyst disclosed in JP-A No. 54-15098 And a method in which maleic anhydride is subjected to an addition reaction by isomerizing the α-olefin chain.
さらに、変性したポリプロピレンを、塩基性化合物、例えばアンモニア、モノエチルアミン、トリエチルアミン、トリエタノールアミン、等の有機アミン、水酸化ナトリウム、水酸化カリウム等の無機アルカリ類等で中和し、ポリオキシエチレンアルキルエーテル、ポリオキシエチレンアルキルフェニルエーテル等の乳化剤の存在下、水中に分散せしめて、酸変性ポリプロピレン樹脂水分散体(A)を得ることができる。 Further, the modified polypropylene is neutralized with a basic compound such as organic amines such as ammonia, monoethylamine, triethylamine, and triethanolamine, and inorganic alkalis such as sodium hydroxide and potassium hydroxide. An acid-modified polypropylene resin aqueous dispersion (A) can be obtained by dispersing in water in the presence of an emulsifier such as ether or polyoxyethylene alkylphenyl ether.
本発明の水性インキ組成物において、水性インキ組成物全固形分における酸変性ポリプロピレン樹脂水分散体(A)の固形分の量は、ポリオレフィンフィルムとインキ組成物との接着性を良好にする観点から5重量%以上、印刷時の流動性や再溶解性の低下を抑制し、印刷効果を良好にする観点から25重量%以下の範囲であることが好ましい。 In the water-based ink composition of the present invention, the solid content of the acid-modified polypropylene resin aqueous dispersion (A) in the total solid content of the water-based ink composition is from the viewpoint of improving the adhesion between the polyolefin film and the ink composition. It is preferably in the range of 5% by weight or more and 25% by weight or less from the viewpoint of suppressing a decrease in fluidity and re-solubility during printing and improving printing effects.
本発明における有機ケイ素化合物(B)とは、一般にシランカップリング剤として知られる化合物であり、YSi Xの化学式で示される。ここでXはアルコキシ基を示し、加水分解性を有する部位である。またYは、アルキル基、アルケン基、アレン基、アリール基、アミノ基、イソシアネート基、エポキシ基、クロル基、メルカプト基等から選択される1種の官能基を有する部位である。 The organosilicon compound (B) in the present invention is a compound generally known as a silane coupling agent, and is represented by a chemical formula of YSi X. Here, X represents an alkoxy group and is a site having hydrolyzability. Y is a site having one functional group selected from an alkyl group, an alkene group, an allene group, an aryl group, an amino group, an isocyanate group, an epoxy group, a chloro group, a mercapto group, and the like.
Yとしては基材密着性の観点から極性が高い官能基がより好適である。したがってさらに好ましくは、アリール基、アミノ基、イソシアネート基、エポキシ基およびメルカプト基から選択される1種の官能基を有することが望ましい。 Y is more preferably a functional group having high polarity from the viewpoint of substrate adhesion. Therefore, it is more preferable to have one functional group selected from an aryl group, an amino group, an isocyanate group, an epoxy group, and a mercapto group.
有機ケイ素化合物(B)はPPダイレクトラミネート強度に好適に作用するが、基材となるフィルムへの密着性を良好にする観点から、より好ましくは0.02重量%以上、インキ組成物の安定性の観点から1重量%以下であることが望ましい。 The organosilicon compound (B) suitably acts on the PP direct laminate strength, but from the viewpoint of improving the adhesion to the base film, more preferably 0.02% by weight or more, the stability of the ink composition In view of the above, the content is desirably 1% by weight or less.
顔料としては、酸化チタン(C)が含まれることが必須である。なお必要に応じて通常印刷インキまたは塗料に用いられる、有機顔料および無機顔料を併用することができる。 As the pigment, it is essential that titanium oxide (C) is contained. If necessary, organic pigments and inorganic pigments usually used in printing inks or paints can be used in combination.
併用される有機顔料としては、天然有機顔料および合成有機顔料があり、天然有機顔料の具体的な例としては、コチニール・レーキ、マダー・レーキなどが挙げられる。また、合成有機顔料としては、ニトロソ顔料、ニトロ顔料、顔料色素型アゾ顔料、溶性アゾ顔料、不溶性アゾ顔料、フタロシアニン顔料、キナクリドン顔料、イソインドリノン顔料、ペリレン顔料、ペリノン顔料、ジオキサジン顔料、アントラキノン顔料、ジアンスラキノニル顔料、アンスラピリミジン顔料、アンサンスロン顔料、インダンスロン顔料、フラバンスロン顔料、ピランスロン顔料、ジケトピロロピロール顔料などのほか、エオシンなどのキサンタン・レーキ顔料、塩基性染料から作るレーキ顔料(ファナル・カラーなど)、塩基型の酸性染料から作るレーキ顔料(アシッド・グリーン・レーキなど)、バット染料からの顔料(インジゴ、アルゴン・イエローなど)などが挙げられる。 Organic pigments used in combination include natural organic pigments and synthetic organic pigments, and specific examples of natural organic pigments include cochineal lake, mudder lake, and the like. Synthetic organic pigments include nitroso pigments, nitro pigments, pigment dye-type azo pigments, soluble azo pigments, insoluble azo pigments, phthalocyanine pigments, quinacridone pigments, isoindolinone pigments, perylene pigments, perinone pigments, dioxazine pigments and anthraquinone pigments. , Dianthraquinonyl pigments, anthrapyrimidine pigments, ansanthrone pigments, indanthrone pigments, flavanthrone pigments, pyranthrone pigments, diketopyrrolopyrrole pigments, rakes made from xanthane lake pigments such as eosin, basic dyes Examples thereof include pigments (Fanal Color, etc.), lake pigments made from basic acid dyes (Acid Green Lake, etc.), and vat dyes (Indigo, Argon / Yellow, etc.).
無機顔料としては、亜鉛華、リトポン、鉛白などの白色顔料、炭酸カルシウム、硫酸バリウム、珪酸カルシウムなどの透明性白色顔料、カーボンブラック、動物性黒、黒鉛などの黒色顔料、ベンガラ、鉛丹などの色顔料、アンバー、酸化鉄粉、バンダイク茶などの茶色顔料、黄鉛、ジンククロメート、黄酸化鉄などの黄色顔料、クロム緑、酸化クロム、ビリジアンなどの緑色顔料、群青、紺青などの青色顔料、マルス紫、淡口コバルト紫などの紫色顔料、アルミニウム粉、銅粉、ブロンズ粉などの金属粉顔料等が挙げられる。本発明では、これらの顔料を単独または2種類以上を酸化チタン顔料と併用して使用できる。 Inorganic pigments include white pigments such as zinc white, lithopone, and lead white, transparent white pigments such as calcium carbonate, barium sulfate, and calcium silicate, black pigments such as carbon black, animal black, and graphite, bengara, and red lead. Color pigments, brown pigments such as amber, iron oxide powder and bandai tea, yellow pigments such as yellow lead, zinc chromate and yellow iron oxide, green pigments such as chrome green, chromium oxide and viridian, blue pigments such as ultramarine and bitumen Violet pigments such as Mars violet and pale mouth cobalt violet, and metal powder pigments such as aluminum powder, copper powder and bronze powder. In the present invention, these pigments can be used alone or in combination of two or more with a titanium oxide pigment.
水性樹脂(D)としては、一般的にインキ、塗料などに使用される種々の水性樹脂を用いることができるが、インキ化適性に優れるポリウレタン樹脂、ポリウレア樹脂、ポリウレタンウレア樹脂、アクリル変性ウレタン樹脂またはアクリル樹脂を用いることが好ましい。これらの樹脂は、単独で用いても良いし、2種以上の樹脂を混合して用いても良い。なおより好ましくは、基材密着性にも優れるアクリル変性ウレタン樹脂が用いられていることが好ましい。アクリル変性ウレタン樹脂は単独で用いられても良いし、他の樹脂と混合して用いられても良い。 As the water-based resin (D), various water-based resins generally used for inks, paints and the like can be used, but polyurethane resins, polyurea resins, polyurethane-urea resins, acrylic-modified urethane resins, which are excellent in inkability, or It is preferable to use an acrylic resin. These resins may be used alone or as a mixture of two or more resins. More preferably, it is preferable to use an acrylic-modified urethane resin that is also excellent in substrate adhesion. The acrylic-modified urethane resin may be used alone or in combination with other resins.
次に、これら各種材料を使用して、ラミネート用水性インキ組成物を製造する方法としては、酸変性ポリプロピレン樹脂水分散体(A)、酸化チタン(C)および水性樹脂(D)を混合してアイガーミル、ペイントシェーカー、ペイントコンディショナー、スキャンデックス、サンドミル、ボールミル、コロイドミルなどの公知の分散機や、ジスパー、ホモミキサーなどの攪拌機などを用いて分散し、さらに所定の材料の残りを添加、混合する方法が一般的である。 Next, as a method of producing a water-based ink composition for lamination using these various materials, an acid-modified polypropylene resin aqueous dispersion (A), titanium oxide (C) and an aqueous resin (D) are mixed. Disperse using a known disperser such as Eiger mill, paint shaker, paint conditioner, scandex, sand mill, ball mill, colloid mill, etc., stirrer such as disper, homomixer, etc. The method is common.
水性インキ組成物に有機ケイ素化合物(B)を含有させる方法としては、添加剤を水性印刷インキに含有させる一般的な方法でよく、特に限定されるものではない。例えば、(1)予め顔料に乾式または湿式法にて有機ケイ素化合物(B)を処理する、(2)インキ化時に顔料等と有機ケイ素化合物(B)一緒に練肉する、(3) インキを練肉後に有機ケイ素化合物(B)を添加する、等の方法がある。なお、より好ましくは錬肉前に有機ケイ素化合物(B)を顔料と作用し得る(1)または(2)を用いることが望ましい。 The method for containing the organic silicon compound (B) in the water-based ink composition may be a general method for adding the additive to the water-based printing ink, and is not particularly limited. For example, (1) The organosilicon compound (B) is previously treated on the pigment by a dry or wet method, (2) The pigment and the organosilicon compound (B) are kneaded together at the time of inking, (3) Ink There are methods such as adding the organosilicon compound (B) after the meat grinding. More preferably, it is desirable to use (1) or (2), which can act on the organosilicon compound (B) with the pigment before slaughtering.
次いで、水性インキ組成物は、グラビアまたはフレキソ印刷方式等でプラスチックフィルムに印刷され、本発明の印刷物が得られる。
さらに、印刷面に直接溶融ポリプロピレンを圧着して積層するダイレクトラミネート法、溶融ポリマーを用いず、基材フィルムとプラスチックフィルムを熱圧着により直接ラミネートするPPサーマルラミネート法により本発明の積層物が得られる。
Next, the water-based ink composition is printed on a plastic film by a gravure or flexographic printing method to obtain the printed material of the present invention.
Furthermore, the laminate of the present invention can be obtained by a direct laminating method in which a melted polypropylene is directly pressed and laminated on a printing surface, or a PP thermal laminating method in which a base film and a plastic film are directly laminated by thermocompression without using a molten polymer. .
以下、本発明を実施例により説明する。本発明において、特に断らない限り、「部」は、「重量部」であり、「%」は、「重量%」である。 Hereinafter, the present invention will be described with reference to examples. In the present invention, “part” is “part by weight” and “%” is “% by weight” unless otherwise specified.
〔合成例1〕
(酸変性ポリプロピレン樹脂水分散体(A))
還流管、温度計、乳化機を備えた容器に、高分子量のポリプロピレンを熱分解して得られた重量平均分子量(以下Mw)10000のポリプロピレンを無水マレイン酸のトルエン溶液を仕込み、窒素置換下110℃に昇温後、ベンゾイルパーオキサイドを添加して4時間反応させた後、未反応の無水マレイン酸を除去して、無水マレイン酸変性ポリプロピレンのトルエン溶液を得た。さらに、固形分に換算して100部となる無水マレイン酸変性ポリプロピレンのトルエン溶液に対して、中和量の1.2当量のアンモニアを含むアンモニア水250部、ポリオキシエチレンノニルフェニルエーテル20部を加え、均一に乳化し、減圧蒸留、pH調整および水の添加を繰り返してトルエンを留去し、固形分40%、原料のポリプロピレンのMwが10000である無水マレイン酸変性ポリプロピレン樹脂水分散体A1を得た。
[Synthesis Example 1]
(Acid-modified polypropylene resin aqueous dispersion (A))
In a container equipped with a reflux tube, a thermometer, and an emulsifier, a polypropylene solution having a weight average molecular weight (hereinafter referred to as Mw) of 10,000 obtained by thermally decomposing high molecular weight polypropylene was charged with a toluene solution of maleic anhydride, and the atmosphere was replaced with nitrogen. After raising the temperature to 0 ° C., benzoyl peroxide was added and reacted for 4 hours, and then unreacted maleic anhydride was removed to obtain a toluene solution of maleic anhydride-modified polypropylene. Furthermore, with respect to a toluene solution of maleic anhydride-modified polypropylene which is 100 parts in terms of solid content, 250 parts of ammonia water containing 1.2 equivalents of ammonia in neutralization amount and 20 parts of polyoxyethylene nonylphenyl ether were added. In addition, the mixture was uniformly emulsified, distilled under reduced pressure, pH adjustment and addition of water to distill off toluene, to obtain a maleic anhydride-modified polypropylene resin aqueous dispersion A1 having a solid content of 40% and a raw material Mw of 10,000. Obtained.
〔合成例2〜6〕
重量平均分子量がそれぞれ7000、20000、50000、100000、200000および300000であるポリプロピレンを用いて、合成例1と同様の方法で、原料のポリプロピレンのMwが7000、20000、50000、100000、200000および300000である無水マレイン酸変性ポリプロピレン樹脂水分散体A2〜A7を得た。
[Synthesis Examples 2 to 6]
Using polypropylene having weight average molecular weights of 7000, 20000, 50000, 100000, 200000 and 300000, respectively, the raw material polypropylene Mw is 7000, 20000, 50000, 100000, 200000 and 300000 in the same manner as in Synthesis Example 1. Certain maleic anhydride-modified polypropylene resin aqueous dispersions A2 to A7 were obtained.
〔合成例7〕
(ウレタン樹脂)
還流冷却管、滴下漏斗、ガス導入管、撹拌装置、温度計を備えた4ツ口のフラスコに重量平均分子量約2,000(水酸基価55.8)のポリテトラメチレングリコール111.0部、重量平均分子量約2,000(水酸基価56.3)のポリエチレングルコール10.7部、1,4シクロヘキサンジメタノール4.3部、ジメチロールブタン酸23.4部、ジブチル錫ジラウレート0.1部、メチルエチルケトン200部を仕込み、乾燥窒素で置換し80℃まで昇温した。攪拌下、イソホロンジイソシアネート80.4部を20分で滴下し3時間反応させた。反応物を40℃に冷却し、N,N-ビス(アミノプロピル)メチルアミン12.6部、イソホロンジアミン7.7部、メチルエチルケトン100部からなる混合物を30分で滴下し、さらに同じ温度で1時間反応させ鎖延長を行った。次に水880部、アンモニア水9.6部を添加し、温度を上げ溶剤430部を脱溶剤し、固形分が25%になるよう調整し、水溶性のウレタン樹脂が得られた(重量平均分子量38,700)。
[Synthesis Example 7]
(Urethane resin)
111.0 parts of polytetramethylene glycol having a weight average molecular weight of about 2,000 (hydroxyl value 55.8) in a four-necked flask equipped with a reflux condenser, a dropping funnel, a gas inlet tube, a stirrer, and a thermometer, and weight 10.7 parts of polyethylene glycol having an average molecular weight of about 2,000 (hydroxyl value 56.3), 4.3 parts of 1,4 cyclohexanedimethanol, 23.4 parts of dimethylolbutanoic acid, 0.1 part of dibutyltin dilaurate, 200 parts of methyl ethyl ketone was charged, replaced with dry nitrogen, and heated to 80 ° C. Under stirring, 80.4 parts of isophorone diisocyanate was added dropwise over 20 minutes and reacted for 3 hours. The reaction product was cooled to 40 ° C., and a mixture consisting of 12.6 parts of N, N-bis (aminopropyl) methylamine, 7.7 parts of isophoronediamine, and 100 parts of methyl ethyl ketone was added dropwise over 30 minutes. The reaction was allowed to proceed for chain extension. Next, 880 parts of water and 9.6 parts of aqueous ammonia were added, the temperature was raised, 430 parts of the solvent was removed, and the solid content was adjusted to 25% to obtain a water-soluble urethane resin (weight average) Molecular weight 38,700).
アクリル樹脂については、BASF社製、JONCRYL57J、60J等が好適に使用することが出来る。なお本発明においてはJONCRYL60J(固形分34%)を使用した。 As the acrylic resin, BASF Co., Ltd., JONCRYL57J, 60J and the like can be suitably used. In the present invention, JONCRYL60J (solid content 34%) was used.
アクリル変性ウレタン樹脂については、大成ファインケミカル株式会社製、WEM200U(固形分38%)を使用した。 For the acrylic modified urethane resin, WEM200U (solid content 38%) manufactured by Taisei Fine Chemical Co., Ltd. was used.
有機ケイ素化合物については、信越化学株式会社製、3-メルカプトプロピルトリメトキシシラン(KBM-803)、メチルトリメトキシシラン(KB-13)、メチルトリエトキシシラン(KBE-13)、デシルトリメトキシラン(KBM-3013)、3-アミノプロピルトリメトキシシラン(KBM-903)、3-グリシドキシプロピルトリメトキシシラン(KBM-403) 、3-イソシアネートプロピルトリエトキシシラン(KBE-9007)、フェニルトリエトキシシラン(KBE-103)、p-スチリルトリメトキシシラン(KBM-1403) 、3-クロロプロピルトリメトキシシランを使用した。 For organosilicon compounds, Shin-Etsu Chemical Co., Ltd., 3-mercaptopropyltrimethoxysilane (KBM-803), methyltrimethoxysilane (KB-13), methyltriethoxysilane (KBE-13), decyltrimethoxylane ( KBM-3013), 3-aminopropyltrimethoxysilane (KBM-903), 3-glycidoxypropyltrimethoxysilane (KBM-403), 3-isocyanatopropyltriethoxysilane (KBE-9007), phenyltriethoxysilane (KBE-103), p-styryltrimethoxysilane (KBM-1403), and 3-chloropropyltrimethoxysilane were used.
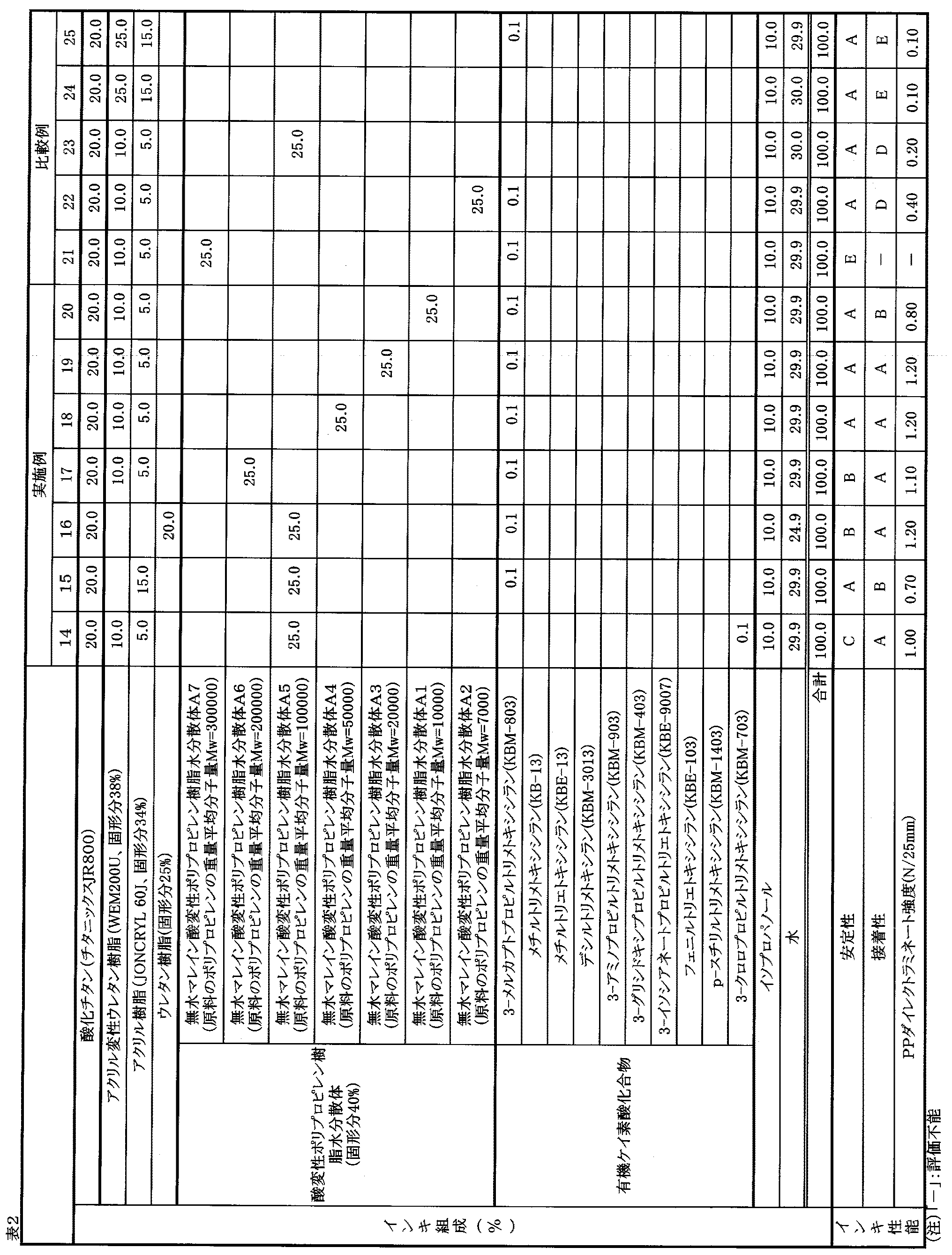
〔実施例1〕
酸化チタン顔料(テイカ株式会社社製、チタニックスJR800)を20部、WEM200Uを10部、JONCRYL60Jを5部、Mw100,000の無水マレイン酸変性ポリプロピレン樹脂分散体を25部、3−メルカプトプロピルトリメトキシシラン(KBM-803)2部、イソプロパノール10部および水28部の混合物をペイントコンディショナーで混練し、実施例1の水性印刷インキ組成物を得た。
[Example 1]
20 parts titanium oxide pigment (Titanics Corporation, Titanics JR800), 10 parts WEM200U, 5 parts JONCRYL60J, 25 parts maleic anhydride-modified polypropylene resin dispersion with Mw 100,000, 3-mercaptopropyltrimethoxy A mixture of 2 parts of silane (KBM-803), 10 parts of isopropanol and 28 parts of water was kneaded with a paint conditioner to obtain an aqueous printing ink composition of Example 1.
表1、2に示した組成で、実施例1に示す方法により実施例2〜20および比較例21〜25を得た。 Examples 2 to 20 and Comparative Examples 21 to 25 were obtained by the method shown in Example 1 with the compositions shown in Tables 1 and 2.
<試験方法および評価>
実施例1〜20び比較例21〜25の水性印刷インキ組成物の安定性、接着性、PPダイレクトラミネート強度を以下の試験方法で行い、その評価結果を表1、2に記載した。
<Test method and evaluation>
The stability, adhesiveness, and PP direct laminate strength of the aqueous printing ink compositions of Examples 1 to 20 and Comparative Examples 21 to 25 were measured by the following test methods, and the evaluation results are shown in Tables 1 and 2.
それぞれの水性印刷インキをザーンカップNo4(25℃)にて粘度を測定し仕上り粘度とした。 The viscosity of each water-based printing ink was measured with Zahn Cup No. 4 (25 ° C.) to obtain a finished viscosity.
<安定性試験方法>
得られた水性印刷インキを40℃にて3ヶ月恒温保管した後に、水性印刷インキをザーンカップNo4(25℃)にて経時粘度を測定し、仕上り(初期)粘度との差を評価した。
(評価基準)
A:仕上り粘度と経時粘度の差が2秒未満のもの
B:仕上り粘度と経時粘度の差が2秒以上4秒未満のもの
C:仕上り粘度と経時粘度の差が4秒以上6秒未満のもの
D:仕上り粘度と経時粘度の差が6秒以上8秒未満のもの
E:仕上り粘度と経時粘度の差が8秒以上のもの
実用レベルはC以上であり、好ましくはB以上であることが望ましい。
<Stability test method>
The obtained water-based printing ink was stored at 40 ° C. for 3 months, and then the viscosity of the water-based printing ink was measured with Zahn Cup No. 4 (25 ° C.) to evaluate the difference from the finished (initial) viscosity.
(Evaluation criteria)
A: The difference between the finished viscosity and the viscosity over time is less than 2 seconds B: The difference between the finished viscosity and the viscosity over time is 2 seconds or more and less than 4 seconds C: The difference between the finished viscosity and the viscosity over time is 4 seconds or more and less than 6 seconds D: The difference between the finished viscosity and the viscosity with time is 6 seconds or more and less than 8 seconds E: The difference between the finished viscosity and the viscosity with time is 8 seconds or more The practical level is C or more, preferably B or more desirable.
<接着性試験方法>
各水性印刷インキをグラビア印刷機で、コロナ放電処理延伸ポリプロピレンフィルム(三井化学東セロ株式会社製CP−SC 30μ)に印刷後、印刷面にセロハンテープを貼り付け、これを急速に剥したときの、印刷皮膜がフィルムから剥離する度合いから接着性を評価した。
(評価基準)
A:印刷皮膜がフィルムから全く剥離しないもの
B:印刷皮膜の面積比率として、10%未満がフィルムから剥離するもの
C:印刷皮膜の面積比率として10%以上20%未満がフィルムから剥離するもの
D:印刷皮膜の面積比率として20%以上50%未満がフィルムから剥離するもの
E:印刷皮膜の面積比率として50%以上がフィルムから剥離するもの
実用レベルはB以上であり、好ましくはAであることが望ましい。
<Adhesion test method>
When each water-based printing ink is printed on a corona discharge-treated stretched polypropylene film (CP-SC 30μ manufactured by Mitsui Chemicals, Inc.) with a gravure printing machine, a cellophane tape is applied to the printed surface, and this is rapidly removed. The adhesiveness was evaluated from the degree to which the printed film peeled from the film.
(Evaluation criteria)
A: The printed film does not peel from the film at all B: The printed film has an area ratio of less than 10% peeled off from the film C: The printed film has an area ratio of 10% or more but less than 20% peeled off from the film D : 20% or more and less than 50% of the printed film is peeled from the film E: 50% or more of the printed film is peeled from the film Practical level is B or more, preferably A Is desirable.
<PPダイレクトラミネート強度試験方法>
各水性印刷インキをグラビア印刷機でコロナ放電処理延伸ポリプロピレンフィルム(東洋紡績株式会社製、パイレンP−2161 20μ)に印刷後、ポリプロピレン「三菱ポリプロFL−25R」(三菱油化株式会社製)にてアンカーコート剤なしでエクストルージョンラミネート加工を行い、得られたラミネート物を15mm幅に切断し、インテスコ株式会社製剥離試験機を用いて、T型剥離強度(N/25mm)を測定した。実用レベルは0.5(N/25mm)以上であり、0.5(N/25mm)未満だと、デラミネーションのトラブルが発生しやすくなる。さらに好ましくは、0.8(N/25mm)以上であることが望ましい。
<PP direct laminate strength test method>
After printing each water-based printing ink on a corona discharge-treated stretched polypropylene film (Toyobo Co., Ltd., Pyrene P-2161 20μ) with a gravure printing machine, with polypropylene “Mitsubishi Polypro FL-25R” (Mitsubishi Yuka Co., Ltd.) Extrusion lamination was performed without an anchor coat agent, and the resulting laminate was cut into a width of 15 mm, and T-type peel strength (N / 25 mm) was measured using a peel tester manufactured by Intesco Corporation. The practical level is 0.5 (N / 25mm) or more, and if it is less than 0.5 (N / 25mm), delamination troubles are likely to occur. More preferably, it is 0.8 (N / 25 mm) or more.
実施例と比較例23より、有機ケイ素化合物(B)を含むことにより、PPダイレクトラミネート強度が向上することが明らかである。特に有機ケイ素化合物(B)の加水分解性の官能基がイソシアネート基、アミノ基、エポキシ基、アリール基およびメルカプト基の場合には際立って向上している。 From Examples and Comparative Example 23, it is clear that the PP direct laminate strength is improved by including the organosilicon compound (B). In particular, when the hydrolyzable functional group of the organosilicon compound (B) is an isocyanate group, amino group, epoxy group, aryl group or mercapto group, it is remarkably improved.
一方、比較例より、酸変性ポリプロピレン樹脂水分散体(A)を含まない場合には接着性やPPラミネート強度が際立って低い。さらに酸変性ポリプロピレン樹脂水分散体(A)を含む場合であっても、ポリプロピレンのMwが10000以下の場合には、PPダイレクトラミネート強度が低く、Mwが200,000を超える場合にはインキの状態が悪化する傾向が確認される。 On the other hand, from the comparative example, when the acid-modified polypropylene resin aqueous dispersion (A) is not included, the adhesiveness and PP laminate strength are remarkably low. Furthermore, even when the acid-modified polypropylene resin aqueous dispersion (A) is included, when the Mw of the polypropylene is 10,000 or less, the PP direct laminate strength is low, and when the Mw exceeds 200,000, the ink state The tendency to get worse is confirmed.
Claims (6)
酸変性ポリプロピレン樹脂水分散体(A)が、重量平均分子量10000〜200,000のポリプロピレンを、マレイン酸および/または無水マレイン酸で、変性したものであることを特徴とするラミネート用水性インキ組成物。 A water-based ink composition for laminating comprising an acid-modified polypropylene resin aqueous dispersion (A), an organosilicon compound (B), titanium oxide (C), and an aqueous resin (D),
The water-based ink composition for laminating, wherein the acid-modified polypropylene resin aqueous dispersion (A) is obtained by modifying polypropylene having a weight average molecular weight of 10,000 to 200,000 with maleic acid and / or maleic anhydride. .
加水分解性の官能基
ならびに
イソシアネート基、アミノ基、エポキシ基、アリール基およびメルカプト基から選 ばれる少なくとも1種の有機官能基
を有することを特徴とする請求項1記載のラミネート用水性インキ組成物。 The organosilicon compound (B)
The water-based ink composition for laminate according to claim 1, which has a hydrolyzable functional group and at least one organic functional group selected from an isocyanate group, an amino group, an epoxy group, an aryl group and a mercapto group.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011066707A JP5678758B2 (en) | 2011-03-24 | 2011-03-24 | Water-based printing composition for laminating |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011066707A JP5678758B2 (en) | 2011-03-24 | 2011-03-24 | Water-based printing composition for laminating |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2012201762A true JP2012201762A (en) | 2012-10-22 |
| JP5678758B2 JP5678758B2 (en) | 2015-03-04 |
Family
ID=47183102
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2011066707A Active JP5678758B2 (en) | 2011-03-24 | 2011-03-24 | Water-based printing composition for laminating |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5678758B2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015163688A (en) * | 2014-01-31 | 2015-09-10 | ユニチカ株式会社 | Anchor coat agent for polypropylene resin extrusion lamination, packaging material and production method thereof |
| WO2021251352A1 (en) * | 2020-06-08 | 2021-12-16 | 関西ペイント株式会社 | Aqueous coating composition |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07138518A (en) * | 1993-11-16 | 1995-05-30 | Dainippon Ink & Chem Inc | Production of pigmented coating composition |
| JPH08120205A (en) * | 1994-10-19 | 1996-05-14 | Sakata Corp | Water-base printing ink composition for laminate and laminating method using same |
| JP2002226758A (en) * | 2001-02-01 | 2002-08-14 | Toyo Ink Mfg Co Ltd | Aqueous binder, water-base ink composition, and laminate using the same |
| JP2003313483A (en) * | 2002-02-19 | 2003-11-06 | Unitika Ltd | Aqueous printing ink and film obtained using the same, laminated film and manufacturing method of laminated film |
| JP2005255879A (en) * | 2004-03-12 | 2005-09-22 | Dainippon Ink & Chem Inc | Aqueous gravure ink for polypropylene tray and polypropylene tray |
| JP2005314530A (en) * | 2004-04-28 | 2005-11-10 | Dainippon Ink & Chem Inc | Alkali-removed type protective coating for plastic and plastic molded article in which protective film is formed |
| JP2006282773A (en) * | 2005-03-31 | 2006-10-19 | Toyo Ink Mfg Co Ltd | Printing ink composition containing silane coupling agent, coated plastic sheet using the same and laminated material |
| JP2009126964A (en) * | 2007-11-26 | 2009-06-11 | Ricoh Co Ltd | Ink for inkjet recording, ink set for inkjet recording, ink-media set for inkjet recording, ink cartridge, inkjet recording method and inkjet recording device |
-
2011
- 2011-03-24 JP JP2011066707A patent/JP5678758B2/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH07138518A (en) * | 1993-11-16 | 1995-05-30 | Dainippon Ink & Chem Inc | Production of pigmented coating composition |
| JPH08120205A (en) * | 1994-10-19 | 1996-05-14 | Sakata Corp | Water-base printing ink composition for laminate and laminating method using same |
| JP2002226758A (en) * | 2001-02-01 | 2002-08-14 | Toyo Ink Mfg Co Ltd | Aqueous binder, water-base ink composition, and laminate using the same |
| JP2003313483A (en) * | 2002-02-19 | 2003-11-06 | Unitika Ltd | Aqueous printing ink and film obtained using the same, laminated film and manufacturing method of laminated film |
| JP2005255879A (en) * | 2004-03-12 | 2005-09-22 | Dainippon Ink & Chem Inc | Aqueous gravure ink for polypropylene tray and polypropylene tray |
| JP2005314530A (en) * | 2004-04-28 | 2005-11-10 | Dainippon Ink & Chem Inc | Alkali-removed type protective coating for plastic and plastic molded article in which protective film is formed |
| JP2006282773A (en) * | 2005-03-31 | 2006-10-19 | Toyo Ink Mfg Co Ltd | Printing ink composition containing silane coupling agent, coated plastic sheet using the same and laminated material |
| JP2009126964A (en) * | 2007-11-26 | 2009-06-11 | Ricoh Co Ltd | Ink for inkjet recording, ink set for inkjet recording, ink-media set for inkjet recording, ink cartridge, inkjet recording method and inkjet recording device |
Non-Patent Citations (2)
| Title |
|---|
| JPN6014031172; 接着・粘着の事典 第5刷, 19970910, P71-74, 株式会社朝倉書店 * |
| JPN6014031174; ゴム・プラスチック配合薬品 改訂第二版, 19931030, P459-464, ラバーダイジェスト社 * |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2015163688A (en) * | 2014-01-31 | 2015-09-10 | ユニチカ株式会社 | Anchor coat agent for polypropylene resin extrusion lamination, packaging material and production method thereof |
| WO2021251352A1 (en) * | 2020-06-08 | 2021-12-16 | 関西ペイント株式会社 | Aqueous coating composition |
| CN115916913A (en) * | 2020-06-08 | 2023-04-04 | 关西涂料株式会社 | Aqueous coating composition |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5678758B2 (en) | 2015-03-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101717596B (en) | Water-based ink for package printing of high-adhesion film and manufacturing method thereof | |
| JP5926630B2 (en) | Liquid ink | |
| JP6090521B1 (en) | Gravure ink for solvent-type lamination, printed matter, and laminate | |
| CN103694872B (en) | A kind of aqueous Stripable paint compositions | |
| CN103045068B (en) | A kind of aluminium foil use coating-forming agent and preparation method thereof | |
| JP2017165025A (en) | Laminate and method for producing the same | |
| JP2014108534A (en) | Printing ink laminate | |
| WO2018021033A1 (en) | Solvent-based gravure printing ink for lamination, printed material and laminate | |
| JP4629767B2 (en) | Water-based flexographic ink for cloth and non-woven fabric | |
| JP5678758B2 (en) | Water-based printing composition for laminating | |
| JP2021046550A (en) | Aqueous adhesive resin composition for olefin base material | |
| JP2018080312A (en) | Aqueous composite resin dispersion and method for producing the same, aqueous ink composition, and printed matter | |
| CN108384317A (en) | A kind of high temperature resistant type gravure water-based ink and preparation method thereof | |
| JP6206553B1 (en) | Non-aqueous printing ink composition | |
| JP2018080311A (en) | Aqueous composite resin and method for producing the same, aqueous ink composition, and printed matter | |
| JPH10204367A (en) | Water-base printing ink composition for lamination, and method for laminating matter printed therewith | |
| JP7156796B2 (en) | Liquid printing ink composition for surface printing and printed matter | |
| JP5761395B2 (en) | Gravure printing ink | |
| JP6255123B1 (en) | Liquid ink composition | |
| JP2024041313A (en) | Gravure printing ink composition, laminate and laminated laminate | |
| JP4742231B2 (en) | Printing ink composition containing silane coupling agent, plastic sheet coating using the same, and laminate laminate thereof | |
| JP4899466B2 (en) | Printing ink composition containing titanium organic compound, plastic sheet covering and laminate laminate using the composition | |
| WO2005005505A1 (en) | A polyurethane resin for color inks | |
| JP2020147745A (en) | Gravure printing ink composition for styrene film for heat lamination | |
| JP7118372B2 (en) | Liquid printing ink composition for surface printing and printed matter |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20131028 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20140716 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20140729 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20140829 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20141209 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20141222 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5678758 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313531 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |