JP2010047646A - Photocurable organopolysiloxane composition - Google Patents
Photocurable organopolysiloxane composition Download PDFInfo
- Publication number
- JP2010047646A JP2010047646A JP2008211097A JP2008211097A JP2010047646A JP 2010047646 A JP2010047646 A JP 2010047646A JP 2008211097 A JP2008211097 A JP 2008211097A JP 2008211097 A JP2008211097 A JP 2008211097A JP 2010047646 A JP2010047646 A JP 2010047646A
- Authority
- JP
- Japan
- Prior art keywords
- units
- sio
- organopolysiloxane
- composition
- organopolysiloxane composition
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 71
- 229920001296 polysiloxane Polymers 0.000 title claims abstract description 51
- 125000003342 alkenyl group Chemical group 0.000 claims abstract description 25
- 230000003287 optical effect Effects 0.000 claims abstract description 13
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims abstract description 10
- WNCWUCSNEKCAAI-UHFFFAOYSA-N carbanide;2-methylcyclopenta-1,3-diene;platinum(4+) Chemical compound [CH3-].[CH3-].[CH3-].[Pt+4].CC1=[C-]CC=C1 WNCWUCSNEKCAAI-UHFFFAOYSA-N 0.000 claims abstract description 10
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims abstract description 8
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical group [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 25
- 229910004283 SiO 4 Inorganic materials 0.000 claims description 11
- 229910052710 silicon Inorganic materials 0.000 claims description 7
- 239000000463 material Substances 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 5
- 239000003566 sealing material Substances 0.000 claims description 5
- 238000004383 yellowing Methods 0.000 abstract description 5
- 239000000047 product Substances 0.000 description 33
- 239000003054 catalyst Substances 0.000 description 19
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 12
- 230000000052 comparative effect Effects 0.000 description 12
- -1 polysiloxane Polymers 0.000 description 12
- 229910052697 platinum Inorganic materials 0.000 description 11
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 9
- 229920005601 base polymer Polymers 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 8
- 238000010438 heat treatment Methods 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 7
- 239000008096 xylene Substances 0.000 description 7
- 238000001816 cooling Methods 0.000 description 6
- 238000002834 transmittance Methods 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 239000003431 cross linking reagent Substances 0.000 description 4
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 4
- 229910052753 mercury Inorganic materials 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 238000007789 sealing Methods 0.000 description 4
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 238000005266 casting Methods 0.000 description 3
- 230000003197 catalytic effect Effects 0.000 description 3
- 238000009833 condensation Methods 0.000 description 3
- 230000005494 condensation Effects 0.000 description 3
- 230000006837 decompression Effects 0.000 description 3
- 125000001183 hydrocarbyl group Chemical group 0.000 description 3
- 238000006460 hydrolysis reaction Methods 0.000 description 3
- 238000006459 hydrosilylation reaction Methods 0.000 description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 230000000704 physical effect Effects 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- BOTDANWDWHJENH-UHFFFAOYSA-N Tetraethyl orthosilicate Chemical compound CCO[Si](OCC)(OCC)OCC BOTDANWDWHJENH-UHFFFAOYSA-N 0.000 description 2
- 238000007259 addition reaction Methods 0.000 description 2
- 239000003513 alkali Substances 0.000 description 2
- 239000006227 byproduct Substances 0.000 description 2
- IJOOHPMOJXWVHK-UHFFFAOYSA-N chlorotrimethylsilane Chemical compound C[Si](C)(C)Cl IJOOHPMOJXWVHK-UHFFFAOYSA-N 0.000 description 2
- 238000013329 compounding Methods 0.000 description 2
- 238000004132 cross linking Methods 0.000 description 2
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- FFUAGWLWBBFQJT-UHFFFAOYSA-N hexamethyldisilazane Chemical compound C[Si](C)(C)N[Si](C)(C)C FFUAGWLWBBFQJT-UHFFFAOYSA-N 0.000 description 2
- 230000001771 impaired effect Effects 0.000 description 2
- 230000001678 irradiating effect Effects 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000002994 raw material Substances 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- QYLFHLNFIHBCPR-UHFFFAOYSA-N 1-ethynylcyclohexan-1-ol Chemical group C#CC1(O)CCCCC1 QYLFHLNFIHBCPR-UHFFFAOYSA-N 0.000 description 1
- SZIFAVKTNFCBPC-UHFFFAOYSA-N 2-chloroethanol Chemical class OCCCl SZIFAVKTNFCBPC-UHFFFAOYSA-N 0.000 description 1
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000004975 3-butenyl group Chemical group C(CC=C)* 0.000 description 1
- 125000006043 5-hexenyl group Chemical group 0.000 description 1
- QTPRHHWVQXPQOP-UHFFFAOYSA-N C1(C=CC=C1)[Pt] Chemical compound C1(C=CC=C1)[Pt] QTPRHHWVQXPQOP-UHFFFAOYSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 239000004651 Radiation Curable Silicone Substances 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- WYUIWUCVZCRTRH-UHFFFAOYSA-N [[[ethenyl(dimethyl)silyl]amino]-dimethylsilyl]ethene Chemical compound C=C[Si](C)(C)N[Si](C)(C)C=C WYUIWUCVZCRTRH-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 125000000217 alkyl group Chemical group 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000001785 cerium compounds Chemical class 0.000 description 1
- FXNONNRUNQPNLF-UHFFFAOYSA-N cerium;2-ethylhexanoic acid Chemical compound [Ce].CCCCC(CC)C(O)=O.CCCCC(CC)C(O)=O.CCCCC(CC)C(O)=O FXNONNRUNQPNLF-UHFFFAOYSA-N 0.000 description 1
- QABCGOSYZHCPGN-UHFFFAOYSA-N chloro(dimethyl)silicon Chemical compound C[Si](C)Cl QABCGOSYZHCPGN-UHFFFAOYSA-N 0.000 description 1
- XSDCTSITJJJDPY-UHFFFAOYSA-N chloro-ethenyl-dimethylsilane Chemical compound C[Si](C)(Cl)C=C XSDCTSITJJJDPY-UHFFFAOYSA-N 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 125000000068 chlorophenyl group Chemical group 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000010292 electrical insulation Methods 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000001125 extrusion Methods 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000005286 illumination Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 238000004898 kneading Methods 0.000 description 1
- VSQYNPJPULBZKU-UHFFFAOYSA-N mercury xenon Chemical compound [Xe].[Hg] VSQYNPJPULBZKU-UHFFFAOYSA-N 0.000 description 1
- 229910001507 metal halide Inorganic materials 0.000 description 1
- 150000005309 metal halides Chemical class 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 238000006386 neutralization reaction Methods 0.000 description 1
- HMMGMWAXVFQUOA-UHFFFAOYSA-N octamethylcyclotetrasiloxane Chemical compound C[Si]1(C)O[Si](C)(C)O[Si](C)(C)O[Si](C)(C)O1 HMMGMWAXVFQUOA-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 238000006068 polycondensation reaction Methods 0.000 description 1
- 239000004848 polyfunctional curative Substances 0.000 description 1
- 229920001843 polymethylhydrosiloxane Polymers 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000007151 ring opening polymerisation reaction Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 229920002050 silicone resin Polymers 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 238000006884 silylation reaction Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 238000001721 transfer moulding Methods 0.000 description 1
- UHUUYVZLXJHWDV-UHFFFAOYSA-N trimethyl(methylsilyloxy)silane Chemical compound C[SiH2]O[Si](C)(C)C UHUUYVZLXJHWDV-UHFFFAOYSA-N 0.000 description 1
- 239000005051 trimethylchlorosilane Substances 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000009281 ultraviolet germicidal irradiation Methods 0.000 description 1
- 229920002554 vinyl polymer Chemical group 0.000 description 1
- 229910052724 xenon Inorganic materials 0.000 description 1
- FHNFHKCVQCLJFQ-UHFFFAOYSA-N xenon atom Chemical compound [Xe] FHNFHKCVQCLJFQ-UHFFFAOYSA-N 0.000 description 1
Landscapes
- Sealing Material Composition (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Led Device Packages (AREA)
Abstract
Description
本発明は、光硬化性オルガノポリシロキサン組成物に関し、特に、硬化して、発光ダイオード(以下、LEDという)の封止材、レンズなどの光学的用途に適した硬化物を与えるオルガノポリシロキサン組成物に関する。また、本発明は、このようなオルガノポリシロキサン組成物を硬化させて得られる硬化物、特に光学用及びLED用に好適な硬化物に関する。 The present invention relates to a photocurable organopolysiloxane composition, and in particular, an organopolysiloxane composition that is cured to give a cured product suitable for optical applications such as a sealing material for a light emitting diode (hereinafter referred to as LED) and a lens. Related to things. The present invention also relates to a cured product obtained by curing such an organopolysiloxane composition, particularly a cured product suitable for optical use and LED use.
シリコーン樹脂及びシリコーンゴムは、その中間の物性を示すポリマーを含め、耐熱性、耐寒性、電気絶縁性などに加えて、透明なものが得られるため、各種の光学用途に用いられている。特に、LEDの封止、保護、レンズなどの用途に、硬化して、高い硬さを有し、透明な硬化物を与えるオルガノポリシロキサン組成物が有用である。 Silicone resins and silicone rubbers are used in various optical applications because they are transparent in addition to heat resistance, cold resistance, electrical insulation, and the like, including polymers having intermediate physical properties. In particular, an organopolysiloxane composition that is cured and has a high hardness and gives a transparent cured product is useful for applications such as LED sealing, protection, and lenses.
特許文献1には、白金触媒を含有する、オルガノポリシロキサン組成物が記載されている。この組成物は加熱により硬化物を与えるが、短時間において高い硬度を達成させることは困難である。また、組成物を調製した直後から白金触媒が活性を有するために、ポットライフを確保するには、硬化遅延剤を添加しなければならず、望まれた使用場所及び使用時期において組成物が適切な硬化性を発揮させるためには、製造ラインの制御が必要となるという問題が存在する。 Patent Document 1 describes an organopolysiloxane composition containing a platinum catalyst. This composition gives a cured product by heating, but it is difficult to achieve high hardness in a short time. In addition, since the platinum catalyst has activity immediately after the preparation of the composition, a curing retarder must be added in order to ensure pot life, and the composition is suitable at the desired use place and use time. In order to exhibit a good curability, there is a problem that it is necessary to control the production line.
特許文献2には、シクロペンタジエニル(Cp)基に、芳香族置換基を有するシクロペンタジエニル白金化合物を用いた光照射硬化性シリコーン組成物が記載されている。しかしながら、未硬化のシリコーン配合物の保存安定性や硬化特性が充分ではなかった。 Patent Document 2 describes a light irradiation curable silicone composition using a cyclopentadienyl platinum compound having an aromatic substituent in a cyclopentadienyl (Cp) group. However, the storage stability and curing characteristics of uncured silicone formulations were not sufficient.
特許文献3には、ヒドロシリル化触媒として光活性型白金錯体を用いた、照射硬化性シリコーン組成物が記載されている。しかしながら、その組成物から得られる硬化物は柔軟なゲルである。
本発明の目的は、ポットライフと硬化特性のコントロールが自由に決定でき、かつ黄変が抑制され、LED用及び光学レンズ用に適した硬化物を与える、光硬化性オルガノポリシロキサン組成物を提供することである。本発明のもう一つの目的は、上記の特性を有し、LEDの封止材及び光学レンズに適した硬化物を提供することである。 An object of the present invention is to provide a photo-curable organopolysiloxane composition that can freely control pot life and curing characteristics, suppress yellowing, and provide a cured product suitable for LEDs and optical lenses. It is to be. Another object of the present invention is to provide a cured product having the above-described properties and suitable for an LED encapsulant and an optical lens.
本発明者は、上記の課題を解決するために研究を重ねた結果、硬化触媒として、光活性触媒能を有する(メチルシクロペンタジエニル)トリメチル白金を用い、ベースポリマーとして、分岐状オルガノポリシロキサンと、場合によって、直鎖状オルガノポリシロキサンとからなる、アルケニル基含有オルガノポリシロキサン、架橋剤として、ポリアルキルハイドロジェンシロキサンを用いることにより、その課題を達成しうることを見出して、本発明を完成させるに至った。 As a result of repeated studies to solve the above problems, the present inventor has used (methylcyclopentadienyl) trimethylplatinum having photoactive catalytic ability as a curing catalyst, and branched organopolysiloxane as a base polymer. And, in some cases, an alkenyl group-containing organopolysiloxane comprising a linear organopolysiloxane, and the use of a polyalkylhydrogensiloxane as a cross-linking agent has found that the problem can be achieved. It came to complete.
すなわち、本発明は、
(A)(A1)SiO4/2単位及びR3SiO1/2単位、並びに場合によってはさらにR2SiO単位及び/又はRSiO3/2単位(式中、Rは、それぞれ独立して、1価の非置換又は置換の炭化水素基を表す)からなり、1分子当たり、少なくとも3個のRがアルケニル基である、分岐状オルガノポリシロキサンと、
場合によって、(A2)ケイ素原子に、R(Rは、上記のとおりである)が結合しており、1分子当たり、少なくとも2個のRがアルケニル基である、直鎖状オルガノポリシロキサンと、
からなる、アルケニル基含有オルガノポリシロキサン;
(B)SiO4/2単位及びR3(CH3)2SiO1/2単位(式中、R3は、それぞれ独立して、水素原子又はメチル基を表す)からなり、1分子当たり、少なくとも3個のR3が水素原子である、ポリアルキルハイドロジェンシロキサン;並びに
(C)(メチルシクロペンタジエニル)トリメチル白金
を含む光硬化性オルガノポリシロキサン組成物に関する。
That is, the present invention
(A) (A1) SiO 4/2 units and R 3 SiO 1/2 units and optionally further R 2 SiO units and / or RSiO 3/2 units (wherein R is independently 1 A branched organopolysiloxane in which at least three R are alkenyl groups per molecule;
In some cases, (A2) a linear organopolysiloxane in which R (R is as described above) is bonded to a silicon atom, and at least two R are alkenyl groups per molecule;
An alkenyl group-containing organopolysiloxane comprising:
(B) SiO 4/2 units and R 3 (CH 3) 2 SiO 1/2 units (wherein, R 3 each independently represents a hydrogen atom or a methyl group) consists, per molecule, at least The present invention relates to a photocurable organopolysiloxane composition comprising a polyalkylhydrogensiloxane in which three R 3 are hydrogen atoms; and (C) (methylcyclopentadienyl) trimethylplatinum.
また、本発明は、上記の光硬化性ポリシロキサン組成物を硬化させて得られる、LEDの封止材及び光学レンズに適した透明な硬化物に関する。 Moreover, this invention relates to the transparent hardened | cured material suitable for the sealing material of LED, and an optical lens obtained by hardening said photocurable polysiloxane composition.
本発明の光硬化性オルガノポリシロキサン組成物は、硬化触媒である(メチルシクロペンタジエニル)トリメチル白金をUV光の照射により活性化しなければ硬化反応が進行しないため、ポットライフ(貯蔵安定性)に優れている。さらに、本発明の硬化触媒は光により活性化されることから、熱への曝露によって生じる硬化物の黄変が限定的であり、耐熱性に優れたLEDの封止材及び光学レンズに適した硬化物を提供することができる。 The photocurable organopolysiloxane composition of the present invention has a pot life (storage stability) because the curing reaction does not proceed unless (methylcyclopentadienyl) trimethylplatinum, which is a curing catalyst, is activated by irradiation with UV light. Is excellent. Furthermore, since the curing catalyst of the present invention is activated by light, yellowing of the cured product caused by exposure to heat is limited, and it is suitable for LED sealing materials and optical lenses having excellent heat resistance. A cured product can be provided.
本発明の光硬化性オルガノポリシロキサン組成物は、(A)ベースポリマー、(B)架橋剤及び(C)硬化触媒としての(メチルシクロペンタジエニル)トリメチル白金を含み、UV光の照射をすると硬化触媒である(メチルシクロペンタジエニル)トリメチル白金が活性化し、(A)のアルケニル基と(B)のケイ素−水素結合との間の付加反応(ヒドロシリル化反応)を促進して、硬化物を与える。 The photocurable organopolysiloxane composition of the present invention comprises (A) a base polymer, (B) a crosslinking agent, and (C) (methylcyclopentadienyl) trimethylplatinum as a curing catalyst, and is irradiated with UV light. The curing catalyst (methylcyclopentadienyl) trimethylplatinum is activated and promotes an addition reaction (hydrosilylation reaction) between the alkenyl group of (A) and the silicon-hydrogen bond of (B) to obtain a cured product. give.
本発明の組成物においては、(A)として、(A1)分岐状オルガノポリシロキサンと、場合によって、(A2)直鎖状オルガノポリシロキサンを用いる。 In the composition of the present invention, as (A), (A1) a branched organopolysiloxane and optionally (A2) a linear organopolysiloxane are used.
(A)として、(A1)を用いることにより、硬化した組成物に優れた機械的強度を付与することができる。(A1)は、SiO4/2単位及びR3SiO1/2単位、並びに場合によってはさらにR2SiO及び/又はRSiO3/2単位(式中、Rは、それぞれ独立して、非置換又は置換の1価の炭化水素基である)からなり、特に硬化反応において架橋点となるように、1分子当たり、少なくとも3個のRがアルケニル基である、分岐状オルガノポリシロキサンである。硬化した組成物に、優れた機械的強度を与えるには、R3SiO1/2単位とSiO4/2単位の比率は、モル比として、1:0.8〜1:3の範囲の、常温で固体ないし粘稠な半固体の樹脂状のものが好ましい。 By using (A1) as (A), excellent mechanical strength can be imparted to the cured composition. (A1) is SiO 4/2 units and R 3 SiO 1/2 units, and optionally further R 2 SiO and / or RSiO 3/2 units (wherein R is each independently unsubstituted or A branched organopolysiloxane in which at least three R are alkenyl groups per molecule so as to be a crosslinking point in the curing reaction. In order to give the cured composition excellent mechanical strength, the ratio of R 3 SiO 1/2 units to SiO 4/2 units is in the range of 1: 0.8 to 1: 3 as a molar ratio, A resinous solid or viscous semisolid at room temperature is preferred.
Rがアルケニル基の場合、ビニル、アリル、3−ブテニル、5−ヘキセニルなどが例示され、合成が容易で、また硬化前の組成物の流動性や、硬化後の組成物の耐熱性を損ねないという点から、ビニル基が最も好ましい。 When R is an alkenyl group, vinyl, allyl, 3-butenyl, 5-hexenyl and the like are exemplified, and synthesis is easy, and fluidity of the composition before curing and heat resistance of the composition after curing are not impaired. From this point, a vinyl group is most preferable.
アルケニル基は、R3SiO1/2単位のRとして存在してもよく、存在する場合には、R2SiO単位又はRSiO3/2単位のRとして存在してもよいが、室温で速い硬化を得るためには、R3SiO1/2単位に存在することが好ましい。 Alkenyl groups may exist as R in R 3 SiO 1/2 units, and if present, may exist as R in R 2 SiO units or RSiO 3/2 units, but fast cure at room temperature In order to obtain, it is preferable to exist in R 3 SiO 1/2 unit.
アルケニル基以外のRとしては、脂肪族不飽和炭素−炭素結合を含まない、非置換又は置換の1価の炭化水素基が挙げられ、メチル、エチル、プロピル、ブチル、ペンチル、ヘキシル、オクチル、デシル、ドデシルのようなアルキル基;シクロヘキシルのようなシクロアルキル基;フェニルのようなアリール基;2−フェニルエチル、2−フェニルプロピルのようなアラルキル基;クロロメチル、クロロフェニル、2−シアノエチル、3,3,3−トリフルオロプロピルのようなハロゲン又はシアノによって置換された1価の炭化水素基が例示される。耐熱性を考慮すると、メチル基が好ましい。 Examples of R other than an alkenyl group include unsubstituted or substituted monovalent hydrocarbon groups that do not contain an aliphatic unsaturated carbon-carbon bond, and include methyl, ethyl, propyl, butyl, pentyl, hexyl, octyl, and decyl. , Alkyl groups such as dodecyl; cycloalkyl groups such as cyclohexyl; aryl groups such as phenyl; aralkyl groups such as 2-phenylethyl and 2-phenylpropyl; chloromethyl, chlorophenyl, 2-cyanoethyl, 3, 3 And monovalent hydrocarbon groups substituted by halogen or cyano such as 3-trifluoropropyl. In view of heat resistance, a methyl group is preferable.
本発明において、(A1)は、通常の製造方法による共加水分解と重縮合の後に、水酸化ナトリウムや水酸化カリウムのようなアルカリ性物質の水溶液を用いて処理した後、常法により中和するか、水分を除去した後、ヘキサメチルジシラザン、1,3−ジビニル−1,1,3,3−テトラメチルジシラザンで処理して、原料等に由来するケイ素原子に結合した水酸基又はアルコキシ基を実質的に含まないようにすることができる。 In the present invention, (A1) is neutralized by a conventional method after treatment with an aqueous solution of an alkaline substance such as sodium hydroxide or potassium hydroxide after co-hydrolysis and polycondensation by a usual production method. Or after removing water, a hydroxyl group or an alkoxy group which is treated with hexamethyldisilazane or 1,3-divinyl-1,1,3,3-tetramethyldisilazane and bonded to a silicon atom derived from a raw material or the like Can be substantially excluded.
本発明においては、(A1)とともに、(A2)を本発明の組成物のベースポリマーとして用いることが好ましい。(A2)は、ケイ素原子に、それぞれ独立して、R(Rは、上記のとおりである)が結合しており、1分子当たり、少なくとも2個のRがアルケニル基である、直鎖状オルガノポリシロキサンである。(A2)は、実質的に直鎖状であればよく、若干の分岐が存在してもよい。 In the present invention, it is preferable to use (A2) together with (A1) as the base polymer of the composition of the present invention. (A2) is a linear organo group in which R (R is as described above) is independently bonded to a silicon atom, and at least two R are alkenyl groups per molecule. Polysiloxane. (A2) may be substantially linear and may have some branching.
(A2)は、末端基として、ジメチルビニルシロキシ基、トリメチルシリル基のようなR3SiO1/2基を導入して、ケイ素原子に結合した水酸基又はアルコキシを実質的に含まないようにすることができ、また、(A1)と同様にアルカリ処理、シリル化処理をすることにより、これらを実質的に含まないようにすることができる。 In (A2), an R 3 SiO 1/2 group such as a dimethylvinylsiloxy group or a trimethylsilyl group is introduced as a terminal group so as not to substantially contain a hydroxyl group or alkoxy bonded to a silicon atom. Moreover, these can be made substantially free by carrying out alkali treatment and silylation treatment in the same manner as (A1).
(A2)についてのRの例示及び好ましい基は、(A1)についてと同様である。なお、アルケニル基であるRは、(A2)の分子鎖の末端又は途中のいずれに存在してもよく、その両方に存在してもよいが、硬化後の組成物に優れた機械的性質を与えるためには、少なくともその両末端に存在していることが好ましい。 Illustrative and preferred groups for R for (A2) are the same as for (A1). In addition, R which is an alkenyl group may be present at either the end or in the middle of the molecular chain of (A2), and may be present at both of them, but has excellent mechanical properties in the composition after curing. In order to give, it is preferable that it exists at the both ends.
(A2)の粘度は、未硬化状態の組成物が、良好な流動性と、優れた作業性を有し、硬化後の組成物が、優れた機械的強度及び適度の硬さを有するために、23℃における粘度が0.01〜1000Pa・sであることが好ましく、より好ましくは0.1〜100Pa・sである。なお、粘度は、JIS K6249 7.1項回転粘度による方法に準拠して測定した値である。 The viscosity of (A2) is because the uncured composition has good fluidity and excellent workability, and the cured composition has excellent mechanical strength and appropriate hardness. The viscosity at 23 ° C. is preferably 0.01 to 1000 Pa · s, more preferably 0.1 to 100 Pa · s. In addition, a viscosity is the value measured based on the method by JISK6249 7.1 term rotational viscosity.
(A2)は、代表的には一般式(II):
(R1)a(R2)bSiO(4−a−b)/2(II)
(式中、
R1は、アルケニル基を表し;
R2は、脂肪族不飽和炭素−炭素結合を含まない、非置換又は置換の1価の炭化水素基を表し;
aは、1又は2であり、好ましくは1であり;
bは0〜2の整数であり、ただし、a+bは2又は3である)で示されるアルケニル基含有シロキサン単位を、分子中に少なくとも2個有する、直鎖状オルガノポリシロキサンである。
(A2) is typically represented by the general formula (II):
(R 1 ) a (R 2 ) b SiO (4-ab) / 2 (II)
(Where
R 1 represents an alkenyl group;
R 2 represents an unsubstituted or substituted monovalent hydrocarbon group that does not contain an aliphatic unsaturated carbon-carbon bond;
a is 1 or 2, preferably 1;
b is an integer of 0 to 2, provided that a + b is 2 or 3. A linear organopolysiloxane having at least two alkenyl group-containing siloxane units represented in the molecule.
R1の例示及び好ましい基は、前述の(A1)のアルケニル基であるRについてと同様であり、R2の例示及び好ましい基は、前述の(A)のアルケニル基以外であるRについてと同様である。 Examples and preferred groups for R 1 are the same as those for R which is the alkenyl group of (A1) described above, and examples and preferred groups for R 2 are the same as those for R which is other than the alkenyl group of (A) described above. It is.
(A)中の(A1)と(A2)の割合は、好ましくは、(A)100重量%中、(A1)が50〜100重量%、(A2)が50〜0重量%である。このような範囲とすることにより、(B)と相溶して均一な組成物を形成し、かつ硬化物に優れた機械的性質、特にLED用及びレンズ用の硬化物として必要な硬さと、低い線膨張率を付与することができる。より好ましくは(A1)が60〜100重量%であり、(A2)が0〜40重量%である。 The ratio of (A1) to (A2) in (A) is preferably 50 to 100% by weight of (A1) and 50 to 0% by weight of (A2) in 100% by weight of (A). By setting it as such a range, it is compatible with (B) to form a uniform composition, and has excellent mechanical properties for the cured product, in particular, the hardness required as a cured product for LEDs and lenses, A low coefficient of linear expansion can be imparted. More preferably, (A1) is 60 to 100% by weight and (A2) is 0 to 40% by weight.
(B)は、分子中にSi−H結合を有するポリオルガノハイドロジェンシロキサンであって、該Si−H結合と(A)ベースポリマーのアルケニル基との間のヒドロシリル化反応によって硬化物を与える架橋剤として寄与する。(A)と相溶して均質な組成物を形成し、かつ硬化物に高い架橋密度を与え、それによって硬化物に高い硬度を付与することから、SiO4/2単位とR3(CH3)2SiO1/2単位(式中、R3は、前述のとおりである)からなる分岐状ポリメチルハイドロジェンシロキサンが選択され、取扱いやすい適切な粘度を有しつつ、上記の目的を達成できることから、R3(CH3)2SiO1/2単位とSiO4/2単位の比率は、SiO2単位1モルに対してR3(CH3)2SiO1/2単位1.5〜2.2モルが好ましく、1.8〜2.1モルがさらに好ましい。分子中に、原料等に由来するケイ素原子に結合した水酸基又はアルコキシ基が実質的に含まれないようにするためには、好ましくは1.9〜2.2モル、より好ましくは2.0〜2.1モルとすることができる。典型的には、[R3(CH3)2SiO1/2]8[SiO4/2]4又は[R3(CH3)2SiO1/2]10[SiO4/2]5のように、4〜5個のQ単位が環状シロキサン骨格を形成し、各Q単位に2個のMH単位及び/又はM単位(ただし、分子中に少なくとも3個はMH単位)が結合しているものが、特に好ましい。 (B) is a polyorganohydrogensiloxane having a Si—H bond in the molecule, and a crosslink that gives a cured product by a hydrosilylation reaction between the Si—H bond and the alkenyl group of the (A) base polymer. Contributes as an agent. Since compatibilized with (A) to form a homogeneous composition, and give a high crosslink density in the cured product, thereby imparting a high hardness in the cured product, SiO 4/2 units and R 3 (CH 3 ) A branched polymethylhydrogensiloxane composed of 2 SiO 1/2 units (wherein R 3 is as described above) is selected, and the above-mentioned object can be achieved while having an appropriate viscosity that is easy to handle. from, R 3 (CH 3) 2 ratio of SiO 1/2 units and SiO 4/2 units, is, R 3 (CH 3) relative to SiO 2 units 1 mol 2 SiO 1/2 units 1.5-2. 2 moles are preferred, and 1.8 to 2.1 moles are more preferred. In order to prevent the hydroxyl group or alkoxy group bonded to the silicon atom derived from the raw material or the like from being substantially contained in the molecule, preferably 1.9 to 2.2 mol, more preferably 2.0 to The amount can be 2.1 mol. Typically, [R 3 (CH 3 ) 2 SiO 1/2 ] 8 [SiO 4/2 ] 4 or [R 3 (CH 3 ) 2 SiO 1/2 ] 10 [SiO 4/2 ] 5 4 to 5 Q units form a cyclic siloxane skeleton, and each Q unit has 2 MH units and / or M units (provided that at least 3 MH units in the molecule are bonded). It is particularly preferable that
(B)中のR3に含まれる、ケイ素原子に直接結合した水素原子の数は、(B)全体として、平均1分子当たり3個以上である。平均3個未満では、必要な硬さの硬化物を得るために充分な架橋密度が得られない。R3の残余は、合成が容易で、耐熱性をはじめとするシロキサンの特徴をバランスよく付与することから、メチル基である。 The number of hydrogen atoms directly bonded to silicon atoms contained in R 3 in (B) is 3 or more per molecule on average as a whole (B). If the average number is less than 3, a sufficient crosslinking density cannot be obtained to obtain a cured product having a required hardness. The remainder of R 3 is a methyl group because it is easy to synthesize and imparts the characteristics of siloxane including heat resistance in a well-balanced manner.
(B)の量は、(A)に存在するアルケニル基1個に対するケイ素原子に結合した水素原子の数の比(H/Vi)は、0.5〜2.0になる量、好ましくは0.7〜1.8になる量である。この範囲であれば、硬化物中に残存するSi−H結合の数が抑制され、充分な物理的性質を有する硬化物が得られる。 The amount of (B) is such that the ratio (H / Vi) of the number of hydrogen atoms bonded to silicon atoms to one alkenyl group present in (A) is 0.5 to 2.0, preferably 0. .7 to 1.8. Within this range, the number of Si-H bonds remaining in the cured product is suppressed, and a cured product having sufficient physical properties can be obtained.
本発明で用いられる(C)の(メチルシクロペンタジエニル)トリメチル白金は、UV光の照射により活性化され、(A)のアルケニル基と(B)のヒドロシリル基との間の付加反応を促進する光活性を有する硬化触媒である。 (C) (methylcyclopentadienyl) trimethylplatinum used in the present invention is activated by irradiation with UV light and promotes the addition reaction between the alkenyl group of (A) and the hydrosilyl group of (B). It is a curing catalyst having photoactivity.
(C)の配合量は、(A)成分および(B)成分の合計量に対する白金金属原子換算で、通常0.1〜1000ppm、例えば0.1〜100ppmである。この範囲であると組成物の優れた硬化速度が得られる。優れた耐熱性を得るためには、好ましくは0.5〜50ppmであり、より好ましくは1〜8ppmである。 The blending amount of (C) is usually 0.1 to 1000 ppm, for example 0.1 to 100 ppm, in terms of platinum metal atoms relative to the total amount of the components (A) and (B). Within this range, an excellent curing rate of the composition can be obtained. In order to obtain excellent heat resistance, the content is preferably 0.5 to 50 ppm, more preferably 1 to 8 ppm.
さらに、本発明の組成物には、硬化物の硬さや透明性などの本発明の目的を損なわないかぎり、必要に応じて、オクタン酸セリウムに代表されるセリウム化合物のような無色の耐熱性向上剤など、各種の添加剤を配合してもよい。また用途によっては、本発明の組成物を、トルエン、キシレンのような有機溶媒に溶解ないし分散させて用いてもよい。 Furthermore, in the composition of the present invention, as long as the purpose of the present invention such as hardness and transparency of the cured product is not impaired, a colorless heat resistance improvement such as a cerium compound typified by cerium octoate is necessary. You may mix | blend various additives, such as an agent. Depending on the application, the composition of the present invention may be dissolved or dispersed in an organic solvent such as toluene or xylene.
本発明の組成物は、(A)〜(C)、及びさらに必要に応じて配合される他の成分を、万能混練機、ニーダーなどの混合手段によって均一に混練して調製することができる。なお、(A1)のうち固体又は粘度の極度に高いものは、取扱いを容易にするために、共加水分解をトルエン、キシレンなどの有機溶媒の存在下に行い、以後の工程を有機溶媒溶液として進め、場合により(A2)と混合した後、減圧加熱により溶媒を留去して、(A)を形成させてもよい。また、(B)成分と(C)成分を別途の容器に保存し、たとえば(A)成分の一部および(C)成分を含む主剤部と、(A)成分の残部および(B)成分を含む硬化剤部を、それぞれ別の容器に保存しておき、使用直前に混合し、減圧で脱泡して使用に供してもよい。 The composition of the present invention can be prepared by uniformly kneading (A) to (C), and other components blended as necessary, by a mixing means such as a universal kneader or a kneader. In addition, in (A1), those having extremely high solids or viscosities are subjected to cohydrolysis in the presence of an organic solvent such as toluene or xylene for easy handling, and the subsequent steps are performed as an organic solvent solution. After proceeding and optionally mixing with (A2), the solvent may be distilled off by heating under reduced pressure to form (A). Further, the component (B) and the component (C) are stored in separate containers. For example, a part of the component (A) and the main agent part containing the component (C), the remainder of the component (A) and the component (B) The hardener parts to be contained may be stored in separate containers, mixed immediately before use, and defoamed under reduced pressure for use.
本発明のオルガノポリシロキサン組成物を、使用すべき部位に注入、滴下、流延、注型、容器からの押出しなどの方法により、又はトランスファー成形や射出成形による一体成形によって、LEDのような対象物と組み合わせる。次いで、UV光を照射してオルガノポリシロキサン組成物を硬化させることにより、硬化物を得ることができる。なお、UV光の照射後において、硬化を促進させるために加熱してもよい。例えば、100〜200℃で10〜60分間加熱することができる。 The organopolysiloxane composition of the present invention is applied to a site such as an LED by a method such as injection, dripping, casting, casting, extrusion from a container, or by integral molding by transfer molding or injection molding. Combine with things. Next, the cured product can be obtained by irradiating UV light to cure the organopolysiloxane composition. In addition, you may heat in order to accelerate | stimulate hardening after irradiation of UV light. For example, it can heat at 100-200 degreeC for 10 to 60 minutes.
本発明の硬化触媒を用いることにより、ポットライフが優れ、硬化の開始時期をUV光の照射のみによって自由に設定することができる。組成物を調製した直後から触媒活性を有する硬化触媒を用いたオルガノポリシロキサン組成物では、大きな対象物に対して封止作業を行う際に、作業中に硬化反応が進行してしまい、均一な強度及び封止を達成することが困難であるが、本発明においては、対象物と組み合わせた後にUV光を照射して硬化を開始させることができるため、そのような問題を回避することができる。 By using the curing catalyst of the present invention, the pot life is excellent, and the curing start time can be freely set only by UV light irradiation. In an organopolysiloxane composition using a curing catalyst having a catalytic activity immediately after the composition is prepared, when a sealing operation is performed on a large object, a curing reaction proceeds during the operation, resulting in a uniform Although it is difficult to achieve strength and sealing, in the present invention, curing can be started by irradiating UV light after being combined with an object, so that such a problem can be avoided. .
本発明において、ポットライフとは、オルガノポリシロキサン組成物が使用可能な液体状態を保つことができる作業性の指標であり、23℃において、すべての構成成分を配合してから、その粘度が配合直後の2倍になるまでの時間をいう。 In the present invention, the pot life is an index of workability that can maintain a liquid state in which the organopolysiloxane composition can be used, and after blending all the components at 23 ° C., the viscosity is blended. This is the time until the next double.
本発明において、(C)硬化触媒の(メチルシクロペンタジエニル)トリメチル白金を活性化させるために、UV光を照射する。照射する光の波長は、200nm〜400nm、好ましくは250nm〜400nmとすることができ、照射エネルギーは、積算光量として100mJ/cm2〜100,000mJ/cm2とすることができる。本発明で用いられる光源としては、UV光を発生する装置であればよく、例えば超高圧水銀灯、キセノンランプ、水銀キセノンランプ、高圧水銀灯、メタルハライドランプ、中圧水銀灯、低圧水銀灯などを挙げることができる。これら複数の光源を組み合わせてもよい。 In the present invention, UV light is irradiated to activate (C) curing catalyst (methylcyclopentadienyl) trimethylplatinum. Wavelength of the irradiated light is 200 nm to 400 nm, and preferably, to 250 nm to 400 nm, radiation energy can be a 100mJ / cm 2 ~100,000mJ / cm 2 as the accumulated light quantity. The light source used in the present invention may be a device that generates UV light, and examples thereof include an ultrahigh pressure mercury lamp, a xenon lamp, a mercury xenon lamp, a high pressure mercury lamp, a metal halide lamp, a medium pressure mercury lamp, and a low pressure mercury lamp. . You may combine these several light sources.
本発明の組成物の硬化物は、JIS K6253のタイプAデュロメータによる硬さが、通常、80以上であり、好ましくは88以上とすることができ、JIS K6253のタイプDデュロメータによる硬さが、通常、30以上であり、好ましくは40以上とすることができる。このため、本発明の組成物の硬化物は、機械的強度があり、表面に傷がつきにくく、またゴミなどが付着しにくい。 The cured product of the composition of the present invention has a hardness according to a JIS K6253 type A durometer of usually 80 or more, preferably 88 or more, and a hardness according to a JIS K6253 type D durometer is usually 30 or more, preferably 40 or more. For this reason, the hardened | cured material of the composition of this invention has mechanical strength, a surface is hard to be damaged, and dust etc. are hard to adhere.
本発明の組成物の硬化物は、青色から紫外領域にかけての短波長領域での透過性に優れ、厚さ10mmの硬化物に対して、波長400nmにおける光透過率が、通常、84%以上であり、より好ましくは86%以上である。さらに、硬化物においては、熱への暴露による黄変が抑制されており、透過率が低下しにくい。 The cured product of the composition of the present invention has excellent transmittance in a short wavelength region from blue to ultraviolet region, and the light transmittance at a wavelength of 400 nm is usually 84% or more with respect to a cured product having a thickness of 10 mm. Yes, more preferably 86% or more. Furthermore, in the cured product, yellowing due to exposure to heat is suppressed, and the transmittance is unlikely to decrease.
また、本発明の組成物の硬化物は、線膨張係数が、通常、100〜300×10−6/K、好ましくは200〜280×10−6/Kである。このように膨張係数を上記範囲とすることで、レンズなどを光学機械などの部品に固定した後の熱履歴や熱衝撃による内部ひずみが緩和され、レンズなどの亀裂を防ぐことができる。さらに、レンズとして光学設計する際のレンズの変形を抑えることができる。 The cured product of the composition of the present invention has a linear expansion coefficient of usually 100 to 300 × 10 −6 / K, preferably 200 to 280 × 10 −6 / K. Thus, by setting the expansion coefficient within the above range, the thermal history after fixing the lens or the like to a component such as an optical machine or the internal strain due to the thermal shock is alleviated, and the lens or the like can be prevented from cracking. Furthermore, it is possible to suppress deformation of the lens when the lens is optically designed.
例えば、本発明の組成物は、LED用の封止に使用することができ、例えば、LEDを搭載した基板に、LEDを封止するように、かつ気泡が残らないように、注型などの方法で送り込み、UV光を照射して、場合により加熱して硬化させて、LEDを内包したレンズの成形品を作製することができる。また、あらかじめ各種の方法で成形したレンズをLED部品にセットし、LEDをそこに嵌め込むか、接着剤で固定することもできる。このように、本発明の組成物の硬化物はLED封止材として好ましく、また、光学レンズにも好適である。 For example, the composition of the present invention can be used for sealing an LED. For example, in order to seal an LED on a substrate on which the LED is mounted and no bubbles remain, casting or the like can be performed. It can be sent by the method, irradiated with UV light, and optionally heated and cured to produce a molded article of a lens including an LED. Alternatively, a lens molded in advance by various methods can be set on an LED component, and the LED can be fitted therein or fixed with an adhesive. Thus, the hardened | cured material of the composition of this invention is preferable as a LED sealing material, and is suitable also for an optical lens.
以下、実施例及び比較例によって、本発明をさらに詳細に説明する。これらの例において、部は重量部、組成の%は重量%を示し、粘度は23℃における粘度を示す。本発明は、これらの実施例によって限定されるものではない。 Hereinafter, the present invention will be described in more detail with reference to Examples and Comparative Examples. In these examples, parts are parts by weight,% of the composition is% by weight, and the viscosity is the viscosity at 23 ° C. The present invention is not limited by these examples.
以下、シロキサン単位を、次のような記号で示す。
M 単位: (CH3)3SiO1/2−
MH単位: (CH3)2HSiO1/2−
Mv単位: (CH3)2(CH2=CH)SiO1/2−
D 単位: −(CH3)2SiO−
DH単位: −(CH3)HSiO−
Q 単位: SiO4/2(4官能性)
Hereinafter, siloxane units are indicated by the following symbols.
M Unit: (CH 3 ) 3 SiO 1/2 −
MH unit: (CH 3 ) 2 HSiO 1/2 −
Mv unit: (CH 3 ) 2 (CH 2 ═CH) SiO 1/2 −
D Unit: - (CH 3) 2 SiO-
D H units: - (CH 3) HSiO-
Q Unit: SiO 4/2 (4-functional)
実施例及び比較例に用いる(A)オルガノポリシロキサンは、以下のとおりである。
(A1):M単位、Mv単位及びQ単位からなり、モル単位比がM5MvQ8で示される分岐状の固体ポリメチルビニルシロキサンの60%キシレン溶液;
(A2):両末端がMv単位で封鎖され、中間単位がすべてD単位であり、23℃における粘度が0.2Pa・sである直鎖状ポリメチルビニルシロキサン;
(A) Organopolysiloxane used in Examples and Comparative Examples is as follows.
(A1): A 60% xylene solution of branched solid polymethylvinylsiloxane consisting of M units, Mv units and Q units and having a molar unit ratio of M 5 M v Q 8 ;
(A2): both terminals blocked with M v units, intermediate units are all in D units, linear polymethylvinylsiloxane having a viscosity at 23 ° C. is at 0.2 Pa · s;
上記のオルガノポリシロキサン(A1)は、以下のように調製したものである。キシレン210部、トリメチルクロロシラン271.3部、ジメチルビニルクロロシラン60.3部及びテトラエトキシシラン832部を、攪拌機、滴下装置、加熱・冷却装置及び減圧装置を備えた反応容器に仕込み、均一に溶解させた。ここに過剰の水を滴下し、副生した塩酸の溶解熱を冷却により除去しつつ、80℃で共加水分解と縮合を行った。得られた有機層を、洗浄水が中性を示すまで水で洗浄し、脱水した後、KOHを200ppmになるように添加し、140℃において系外に水を除きながら脱水縮合を行った。その後、リン酸で中和し、生成した塩をろ過し、不揮発分が60%になるようにキシレンで希釈し調整した。 The organopolysiloxane (A1) is prepared as follows. Charge 210 parts of xylene, 271.3 parts of trimethylchlorosilane, 60.3 parts of dimethylvinylchlorosilane, and 832 parts of tetraethoxysilane to a reaction vessel equipped with a stirrer, a dropping device, a heating / cooling device, and a decompression device, and dissolve them uniformly. It was. Excess water was added dropwise thereto, and co-hydrolysis and condensation were performed at 80 ° C. while removing the heat of dissolution of by-produced hydrochloric acid by cooling. The obtained organic layer was washed with water until the washing water showed neutrality, dehydrated, KOH was added to 200 ppm, and dehydration condensation was performed at 140 ° C. while removing water outside the system. Then, it neutralized with phosphoric acid, the produced | generated salt was filtered, and it diluted and adjusted with xylene so that a non volatile matter might be 60%.
また、上記の直鎖状ポリメチルビニルシロキサン(A2)は、以下のように調製したものである。オクタメチルシクロテトラシロキサン845.6部を攪拌機、滴下装置、加熱・冷却装置及び減圧装置を備えた反応容器に仕込み、N2を流しながら、140℃において攪拌し脱水した。その後、1,7−ジビニル−1,1,3,3,5,5,7,7−オクタメチルシロキサン14.2部及びKOH8部を添加し、140℃で8時間、開環重合を行った。その後、100℃まで冷却し、エチレンクロロヒドリン100gを添加し、100℃2時間で中和を行った。その後、低分子量のポリマーを160℃で4時間、1.3KPa{10mmHg}以下でストリッピングを行い、冷却後ろ過を行い、目的物を得た。 Moreover, said linear polymethylvinylsiloxane (A2) was prepared as follows. 845.6 parts of octamethylcyclotetrasiloxane was charged into a reaction vessel equipped with a stirrer, a dropping device, a heating / cooling device, and a decompression device, and stirred and dehydrated at 140 ° C. while flowing N 2 . Thereafter, 14.2 parts of 1,7-divinyl-1,1,3,3,5,5,7,7-octamethylsiloxane and 8 parts of KOH were added, and ring-opening polymerization was carried out at 140 ° C. for 8 hours. . Then, it cooled to 100 degreeC, 100 g of ethylene chlorohydrins were added, and neutralization was performed at 100 degreeC for 2 hours. Thereafter, the low molecular weight polymer was stripped at 160 ° C. for 4 hours at 1.3 KPa {10 mmHg} or less, filtered after cooling to obtain the desired product.
実施例及び比較例に用いる架橋剤(B)は、以下のように調製したものである。トルエン520部、テトラエトキシシラン879部及びジメチルクロロシラン832部を仕込み、均一に溶解させた。これを、撹拌機、滴下装置、加熱・冷却装置及び減圧装置を備えた反応容器に撹拌中の過剰の水に滴下して、副生した塩酸の溶解熱を冷却により除去しつつ、室温で共加水分解と縮合を行った。得られた有機相を、洗浄水が中性を示すまで水で洗浄し、脱水した後、トルエンと副生したテトラメチルジシロキサンを100℃/667Pa{5mmHg}で留去して、液状のポリメチルハイドロジェンシロキサンを調製した。GPCによる平均分子量は800(理論値:776)であり、これとアルカリ滴定によるSi−H結合の分析結果から、得られたシロキサンが近似的にMH 8Q4で示されるポリメチルハイドロジェンシロキサンであることを確認した。 The crosslinking agent (B) used in the examples and comparative examples is prepared as follows. 520 parts of toluene, 879 parts of tetraethoxysilane and 832 parts of dimethylchlorosilane were charged and dissolved uniformly. This is dropped into excess water under stirring in a reaction vessel equipped with a stirrer, a dripping device, a heating / cooling device, and a decompression device, and the heat of dissolution of the by-product hydrochloric acid is removed by cooling, while being shared at room temperature. Hydrolysis and condensation were performed. The obtained organic phase is washed with water until the washing water shows neutrality, dehydrated, and then toluene and by-product tetramethyldisiloxane are distilled off at 100 ° C./667 Pa {5 mmHg} to obtain a liquid polycrystal. Methyl hydrogen siloxane was prepared. The average molecular weight by GPC is 800 (theoretical value: 776). From this and the result of analysis of Si—H bonds by alkali titration, the resulting siloxane is approximately polymethylhydrogensiloxane represented by MH 8 Q 4 It was confirmed that.
実施例に用いる(C)硬化触媒である(メチルシクロペンタジエニル)トリメチル白金(Me3Pt−Cp−Me錯体)(C1)は、Strem Chemicals, Inc.社製(ドイツ)により市販されている。また、比較例に用いる硬化触媒(Pt−Dv 4錯体)(C2)は、塩化白金酸をDv 4で示される環状シロキサンと加熱して調製され、白金含有量が2重量%である白金−ビニルシロキサン錯体である。 (C) Curing catalyst (methylcyclopentadienyl) trimethylplatinum (Me 3 Pt-Cp-Me complex) (C1) used in the examples was obtained from Strem Chemicals, Inc. It is marketed by the company (Germany). Further, the curing catalyst used in Comparative Example (Pt-D v 4 complex) (C2) is prepared by heating a cyclic siloxane represented chloroplatinic acid with D v 4, the platinum content is 2 weight percent platinum -Vinylsiloxane complex.
比較例に用いる硬化遅延剤は、1−エチニル−1−シクロヘキサノールである。 The curing retarder used in the comparative example is 1-ethynyl-1-cyclohexanol.
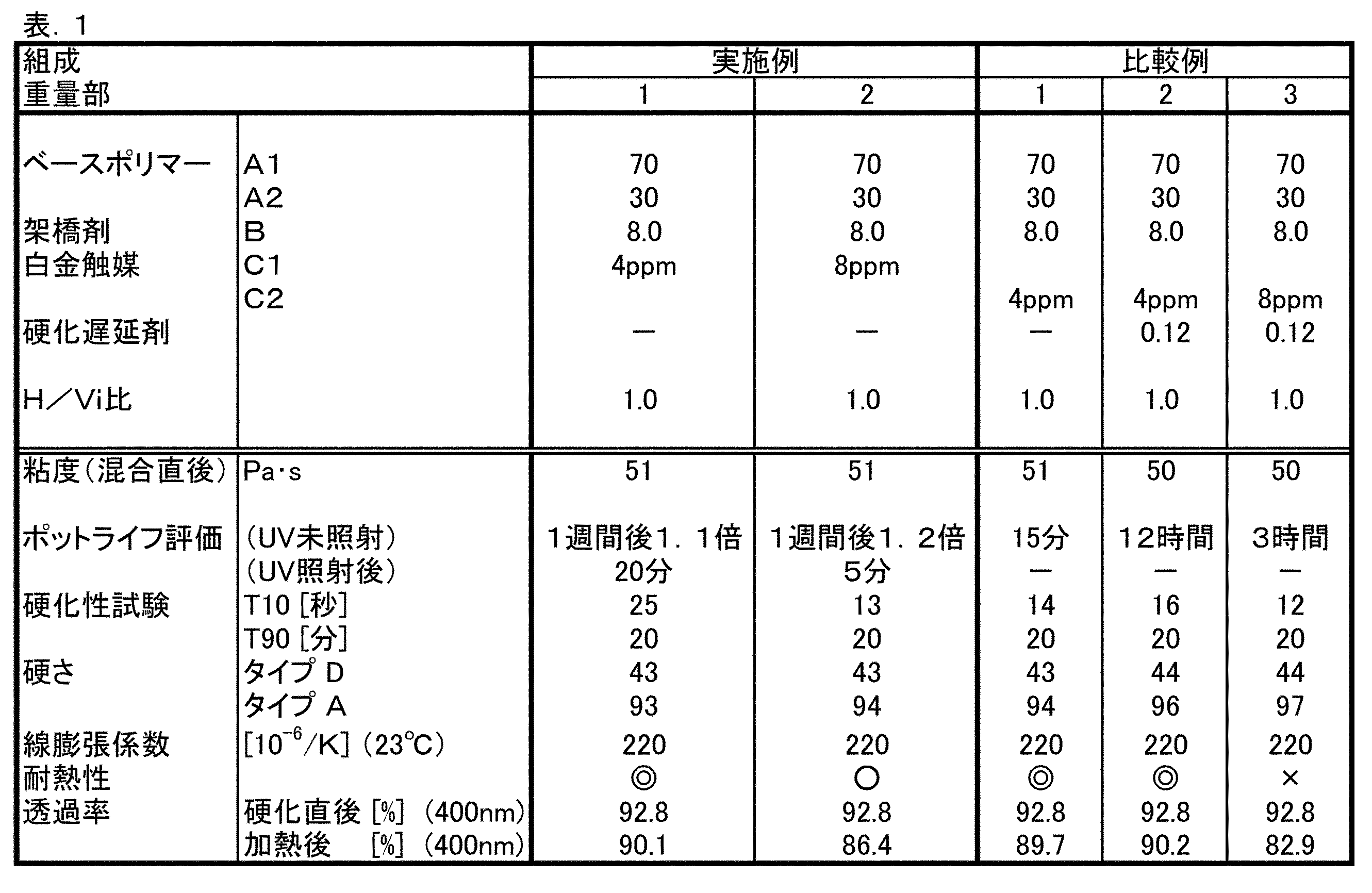
実施例1及び2、比較例1〜3
A1がキシレン溶液なので、減圧加熱装置及び撹拌機を備えた容器にA1とA2を仕込み、均一になるまで撹拌・混合した後、A1に含まれるキシレンを140℃/667Pa{5mmHg}で留去して、液状のベースポリマー混合物を調製した。
Examples 1 and 2, Comparative Examples 1-3
Since A1 is a xylene solution, A1 and A2 are charged in a container equipped with a vacuum heating device and a stirrer, and stirred and mixed until uniform, and then xylene contained in A1 is distilled off at 140 ° C / 667Pa {5mmHg}. A liquid base polymer mixture was prepared.
万能混練機を用いて、表1に示す配合比で、ベースポリマーの一部、触媒からなる主剤部、並びにベースポリマーの残部、架橋剤を含む硬化剤部を、それぞれ調製した。なお、主剤部と硬化剤部がほぼ重量比で1:1になるように、ベースポリマーの配分を行った。主剤部と硬化剤部を混合し、続いて、脱泡して、厚さ2mmのシート状に注型し、実施例1及び2では、次いで、4KWコンベア式UV照射装置(ウシオ電機社製)を使用して、波長250〜400nmで、積算光量1000mJ/cm2のUV光を照射した。続いて、150℃のオーブン中で1時間加熱して硬化させた。無色透明で、若干の伸びを有する樹脂状の硬化物が得られた。 Using a universal kneader, a part of the base polymer, the main part of the catalyst, the remainder of the base polymer, and the curing agent part containing the crosslinking agent were prepared at the compounding ratios shown in Table 1, respectively. The base polymer was distributed so that the weight ratio of the main agent part and the curing agent part was approximately 1: 1. The main agent part and the curing agent part were mixed, then defoamed and cast into a sheet having a thickness of 2 mm. In Examples 1 and 2, a 4 KW conveyor type UV irradiation device (manufactured by USHIO INC.) use, in the wavelength 250 to 400 nm, and irradiated with UV light of the accumulated amount 1000 mJ / cm 2. Subsequently, it was cured by heating in an oven at 150 ° C. for 1 hour. A resinous cured product which was colorless and transparent and had some elongation was obtained.
組成物とその硬化物との評価を、次のようにして行った。
(1)ポットライフ評価:得られた組成物の粘度を測定し、23℃におけるポリシロキサン組成物の粘度が、組成物の調製直後の1.5倍になる時間を測定した。なお、実施例においては、UV光が未照射である場合とUV光を照射した後の場合について測定した。
(2)硬化性試験:未硬化の組成物について、(株)エーアンドディー社製、キュラストメータWR型により、温度150℃測定時間30分において、検出される最大トルクに対して10%になる時間(T10)(秒)及び90%になる時間(T90)(分)を測定した。なお、実施例1および2では、UV光の照射直後に測定を行った。
(3)硬さ:シートを23℃で24時間放置した後、シートを3枚重ねて6mmの厚さとした試料を用いてJIS K6253により、タイプA及びDデュロメータによって硬さを測定した。
(4)線膨張係数:SII社製、熱機械的分析装置TMA/SS6100型によって測定した。
(5)耐熱性:硬化直後の試料および硬化物を180℃24時間放置した後の試料(10mm×10mm×40mm)を、23℃において、日立製作所製、スペクトロフォトメータU−3410型を使用し、光路長10mmで波長400nmにおける透過率を測定した。透過率が、88%以上を◎、84%以上〜88%未満を○、84%未満を×とした。
The composition and its cured product were evaluated as follows.
(1) Pot life evaluation: The viscosity of the obtained composition was measured, and the time during which the viscosity of the polysiloxane composition at 23 ° C. was 1.5 times immediately after the preparation of the composition was measured. In the examples, the measurement was performed for the case where the UV light was not irradiated and the case where the UV light was irradiated.
(2) Curability test: About uncured composition, 10% with respect to the maximum torque detected at a temperature of 150 ° C. for 30 minutes using a curast meter WR type manufactured by A & D Co., Ltd. The time to become (T10) (second) and the time to become 90% (T90) (minute) were measured. In Examples 1 and 2, measurement was performed immediately after UV light irradiation.
(3) Hardness: After the sheet was left at 23 ° C. for 24 hours, the hardness was measured with a type A and D durometer according to JIS K6253 using a sample in which three sheets were stacked to a thickness of 6 mm.
(4) Linear expansion coefficient: Measured with a thermomechanical analyzer TMA / SS6100 manufactured by SII.
(5) Heat resistance: a sample immediately after curing and a sample (10 mm × 10 mm × 40 mm) after leaving the cured product at 180 ° C. for 24 hours at 23 ° C. using a spectrophotometer U-3410 type manufactured by Hitachi, Ltd. The transmittance at an optical path length of 10 mm and a wavelength of 400 nm was measured. A transmittance of 88% or more was evaluated as ◎, 84% or more but less than 88% as ○, and less than 84% as ×.
各実施例及び比較例の配合比、並びに組成物の硬化性と硬化物の物性は、表1に示すとおりであった。なお、白金触媒の含有量(ppm)は、A成分及びB成分の合計量に対する白金金属原子換算による。 Table 1 shows the compounding ratio of each example and comparative example, and the curability of the composition and the physical properties of the cured product. In addition, content (ppm) of a platinum catalyst is based on platinum metal atom conversion with respect to the total amount of A component and B component.
比較例の組成物は、UV光が未照射であっても触媒活性を有する白金触媒を用いたものである。比較例1は、硬化遅延剤を含まない系であり、オルガノポリシロキサン組成物を調製後、15分で粘度が1.5倍になった。
比較例2は、比較例1の組成において、さらに硬化遅延剤を用いた系であり、オルガノポリシロキサン組成物を調製してから12時間で粘度が1.5倍になった。
比較例3は、オルガノポリシロキサン組成物に対して白金含有量が8ppmになるように白金触媒を添加した系であり、硬化反応が十分促進されるため、オルガノポリシロキサン組成物を調製してから3時間で粘度が1.5倍になった。さらに硬化直後の硬化物は白金触媒の含有量が低い組成物と同様の光透過率であったが、耐熱性試験において黄変が観察され、耐熱性が劣っていた。
The composition of the comparative example uses a platinum catalyst having catalytic activity even when UV light is not irradiated. Comparative Example 1 was a system that did not contain a curing retardant, and the viscosity increased 1.5 times in 15 minutes after the preparation of the organopolysiloxane composition.
Comparative Example 2 was a system using a curing retarder in the composition of Comparative Example 1, and the viscosity increased 1.5 times in 12 hours after the preparation of the organopolysiloxane composition.
Comparative Example 3 is a system in which a platinum catalyst is added so that the platinum content is 8 ppm with respect to the organopolysiloxane composition. Since the curing reaction is sufficiently accelerated, the organopolysiloxane composition is prepared. The viscosity increased 1.5 times in 3 hours. Further, the cured product immediately after curing had the same light transmittance as that of the composition having a low platinum catalyst content, but yellowing was observed in the heat resistance test, and the heat resistance was poor.
実施例1および2では、オルガノポリシロキサン組成物を調製してから1週間が経過した後であっても、粘度の増加がわずかであり、ポットライフに優れていることがわかった。また、UV光の照射後は僅かな時間で粘度が1.5倍になっており、硬化特性が優れていた。さらに、実施例2では、白金含有量が8ppmであるにもかかわらず、耐熱性の低下が抑えられていた。 In Examples 1 and 2, it was found that even after one week had passed since the organopolysiloxane composition was prepared, the increase in viscosity was slight and the pot life was excellent. In addition, the viscosity increased 1.5 times in a short time after irradiation with UV light, and the curing characteristics were excellent. Furthermore, in Example 2, although the platinum content was 8 ppm, a decrease in heat resistance was suppressed.
本発明の光硬化性ポリシロキサン組成物は、UV光を照射することで硬化反応を開始させることができるため、組成物のポットライフを容易にかつ自由に決めることができる。さらに、UV光の照射後は、オルガノポリシロキサン組成物が容易に硬化し、耐熱性に優れた硬化物を与えることができる。本発明は、光学用途、特に耐熱性を要求されるLED等の照明関連や自動車用途に有用である。 Since the photocurable polysiloxane composition of the present invention can initiate a curing reaction by irradiation with UV light, the pot life of the composition can be determined easily and freely. Furthermore, after irradiation with UV light, the organopolysiloxane composition is easily cured, and a cured product having excellent heat resistance can be obtained. The present invention is useful for optical applications, particularly for illumination related to LEDs and the like that require heat resistance, and for automotive applications.
Claims (8)
場合によって、(A2)ケイ素原子に、R(Rは、上記のとおりである)が結合しており、1分子当たり、少なくとも2個のRがアルケニル基である、直鎖状オルガノポリシロキサンと、
からなる、アルケニル基含有オルガノポリシロキサン;
(B)SiO4/2単位及びR3(CH3)2SiO1/2単位(式中、R3は、それぞれ独立して、水素原子又はメチル基を表す)からなり、1分子当たり、少なくとも3個のR3が水素原子である、ポリアルキルハイドロジェンシロキサン;並びに
(C)(メチルシクロペンタジエニル)トリメチル白金
を含む光硬化性オルガノポリシロキサン組成物。 (A) (A1) SiO 4/2 units and R 3 SiO 1/2 units and optionally further R 2 SiO units and / or RSiO 3/2 units (wherein R is independently 1 A branched organopolysiloxane in which at least three R are alkenyl groups per molecule;
In some cases, (A2) a linear organopolysiloxane in which R (R is as described above) is bonded to a silicon atom, and at least two R are alkenyl groups per molecule;
An alkenyl group-containing organopolysiloxane comprising:
(B) SiO 4/2 units and R 3 (CH 3 ) 2 SiO 1/2 units (wherein R 3 independently represents a hydrogen atom or a methyl group), A polyalkylhydrogensiloxane, wherein three R 3 are hydrogen atoms; and (C) a photocurable organopolysiloxane composition comprising (methylcyclopentadienyl) trimethylplatinum.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008211097A JP5342830B2 (en) | 2008-08-19 | 2008-08-19 | Photocurable organopolysiloxane composition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008211097A JP5342830B2 (en) | 2008-08-19 | 2008-08-19 | Photocurable organopolysiloxane composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010047646A true JP2010047646A (en) | 2010-03-04 |
| JP5342830B2 JP5342830B2 (en) | 2013-11-13 |
Family
ID=42064996
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2008211097A Active JP5342830B2 (en) | 2008-08-19 | 2008-08-19 | Photocurable organopolysiloxane composition |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP5342830B2 (en) |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010120979A (en) * | 2008-11-17 | 2010-06-03 | Taika:Kk | Thermally conductive silicone gel cured product |
| JP2010144130A (en) * | 2008-12-22 | 2010-07-01 | Taika:Kk | Curable organopolysiloxane composition |
| CN101935455A (en) * | 2010-07-28 | 2011-01-05 | 杭州师范大学 | Organosilicon material for packaging LED and preparation method thereof |
| EP2463334A1 (en) | 2010-12-07 | 2012-06-13 | Shin-Etsu Chemical Co., Ltd. | Method for curing a silicone resin composition |
| JP5002075B2 (en) * | 2010-10-14 | 2012-08-15 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Curable polyorganosiloxane composition |
| EP2570463A1 (en) * | 2011-09-16 | 2013-03-20 | Shin-Etsu Chemical Co., Ltd. | Method of producing cured thin film using photocurable silicone resin composition |
| WO2013084425A1 (en) * | 2011-12-09 | 2013-06-13 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Composition for silicone rubber foams, method for producing silicone rubber foam, and silicone rubber foam |
| EP2617791A1 (en) | 2012-01-18 | 2013-07-24 | Shin-Etsu Chemical Co., Ltd. | UV-curable adhesive silicone composition, UV-curable adhesive silicone composition sheet, optical semiconductor apparatus and method for manufacturing the same |
| EP2628763A1 (en) | 2012-02-20 | 2013-08-21 | Evonik Goldschmidt GmbH | Branched polysiloxanes and their use |
| CN103320087A (en) * | 2013-06-03 | 2013-09-25 | 曹坚林 | Liquid optical transparent organic silicon material and preparation method of same |
| JP2013540872A (en) * | 2010-10-29 | 2013-11-07 | ワッカー ケミー アクチエンゲゼルシャフト | Photocrosslinkable highly transparent silicone blend |
| US20140103546A1 (en) * | 2012-10-16 | 2014-04-17 | Brewer Science Inc. | Silicone polymers with high refractive indices and extended pot life |
| US8809480B2 (en) | 2012-01-16 | 2014-08-19 | Shin-Etsu Chemical Co., Ltd. | Silicone resin composition, luminous substance-containing wavelength-converting film, and cured product thereof |
| KR20140124724A (en) | 2013-04-17 | 2014-10-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Photocurable fluoropolyether gel composition, method for curing the same, gel cured product thereof and gel products using cured product thereof |
| WO2015056483A1 (en) * | 2013-10-18 | 2015-04-23 | 信越化学工業株式会社 | Ultraviolet-ray-curable organopolysiloxane composition, and method for producing printing material |
| WO2016098884A1 (en) * | 2014-12-18 | 2016-06-23 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Polyorganosiloxane composition for molding, optical member, lens or cover for light source, and molding method |
| KR20180124906A (en) * | 2016-03-14 | 2018-11-21 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Heat curing type thermoconductive silicone grease composition and method for producing the cured product |
| JP2018538423A (en) * | 2015-11-06 | 2018-12-27 | ワッカー ケミー アクチエンゲゼルシャフトWacker Chemie AG | Method of laminating base material and product manufactured thereby |
| WO2019004601A1 (en) * | 2017-06-28 | 2019-01-03 | 비즈텍코리아 주식회사 | Photocurable polysiloxane composition for 3d printing, and dental mold comprising same |
| WO2019088067A1 (en) * | 2017-10-31 | 2019-05-09 | ダウ・東レ株式会社 | Method for producing organopolysiloxane cured product, organopolysiloxane cured product, layered product, and optical part |
| WO2019088066A1 (en) * | 2017-10-31 | 2019-05-09 | ダウ・東レ株式会社 | Organopolysiloxane composition, and half-cured product and cured product produced from same |
| CN109825245A (en) * | 2018-12-30 | 2019-05-31 | 苏州桐力光电股份有限公司 | A kind of full fitting liquid crystal module sealing technology |
| CN109825244A (en) * | 2018-12-30 | 2019-05-31 | 苏州桐力光电股份有限公司 | One kind can ultraviolet lighting and the double cured clear silicone gels of heating |
| KR20190074225A (en) | 2017-12-19 | 2019-06-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Ultraviolet-curable resin composition, adhesive and cured product |
| WO2020090797A1 (en) * | 2018-10-30 | 2020-05-07 | ダウ・東レ株式会社 | Curable reactive silicone composition, cured product thereof and uses of composition and cured product |
| JP2020529486A (en) * | 2017-08-04 | 2020-10-08 | ワッカー ケミー アクチエンゲゼルシャフトWacker Chemie AG | Stabilization of precious metal catalysts |
| WO2024000245A1 (en) * | 2022-06-29 | 2024-01-04 | Dow Toray Co., Ltd. | Uv curable organopolysiloxane composition and application thereof |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6565723B2 (en) | 2015-02-04 | 2019-08-28 | 信越化学工業株式会社 | Photocurable fluoropolyether rubber composition, cured product thereof, and curing method thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005042050A (en) * | 2003-07-24 | 2005-02-17 | Dow Corning Asia Ltd | Curable silicone composition and method for forming pattern by using the same |
| JP2006335857A (en) * | 2005-06-01 | 2006-12-14 | Ge Toshiba Silicones Co Ltd | Polyorganosiloxane composition which gives transparent cured product |
| WO2007136956A1 (en) * | 2006-05-17 | 2007-11-29 | 3M Innovative Properties Company | Method of making light emitting device with silicon-containing composition |
| JP2008508382A (en) * | 2004-07-28 | 2008-03-21 | ジーイー・バイエル・シリコーンズ・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング・ウント・コンパニー・コマンジツトゲゼルシヤフト | Use of a photoactivated curable silicone composition for the manufacture of thick molded products or thick coatings |
| WO2008047892A1 (en) * | 2006-10-19 | 2008-04-24 | Momentive Performance Materials Japan Llc | Curable polyorganosiloxane composition |
-
2008
- 2008-08-19 JP JP2008211097A patent/JP5342830B2/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005042050A (en) * | 2003-07-24 | 2005-02-17 | Dow Corning Asia Ltd | Curable silicone composition and method for forming pattern by using the same |
| JP2008508382A (en) * | 2004-07-28 | 2008-03-21 | ジーイー・バイエル・シリコーンズ・ゲゼルシヤフト・ミツト・ベシユレンクテル・ハフツング・ウント・コンパニー・コマンジツトゲゼルシヤフト | Use of a photoactivated curable silicone composition for the manufacture of thick molded products or thick coatings |
| JP2006335857A (en) * | 2005-06-01 | 2006-12-14 | Ge Toshiba Silicones Co Ltd | Polyorganosiloxane composition which gives transparent cured product |
| WO2007136956A1 (en) * | 2006-05-17 | 2007-11-29 | 3M Innovative Properties Company | Method of making light emitting device with silicon-containing composition |
| WO2008047892A1 (en) * | 2006-10-19 | 2008-04-24 | Momentive Performance Materials Japan Llc | Curable polyorganosiloxane composition |
Cited By (64)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010120979A (en) * | 2008-11-17 | 2010-06-03 | Taika:Kk | Thermally conductive silicone gel cured product |
| JP2010144130A (en) * | 2008-12-22 | 2010-07-01 | Taika:Kk | Curable organopolysiloxane composition |
| CN101935455A (en) * | 2010-07-28 | 2011-01-05 | 杭州师范大学 | Organosilicon material for packaging LED and preparation method thereof |
| EP2628770A4 (en) * | 2010-10-14 | 2013-11-20 | Momentive Performance Mat Jp | Curable polyorganosiloxane composition |
| JP5002075B2 (en) * | 2010-10-14 | 2012-08-15 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Curable polyorganosiloxane composition |
| US9303164B2 (en) | 2010-10-14 | 2016-04-05 | Momentive Performance Materials Japan Llc | Method of preparing a curable polyorganosiloxane composition |
| US8940851B2 (en) | 2010-10-29 | 2015-01-27 | Wacker Chemie Ag | Highly transparent silicone mixtures that can be cross-linked by light |
| JP2013540872A (en) * | 2010-10-29 | 2013-11-07 | ワッカー ケミー アクチエンゲゼルシャフト | Photocrosslinkable highly transparent silicone blend |
| KR101873592B1 (en) * | 2010-12-07 | 2018-07-02 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Curing method for silicone resin composition |
| JP2012121960A (en) * | 2010-12-07 | 2012-06-28 | Shin-Etsu Chemical Co Ltd | Method for curing silicone resin composition |
| EP2463334A1 (en) | 2010-12-07 | 2012-06-13 | Shin-Etsu Chemical Co., Ltd. | Method for curing a silicone resin composition |
| JP2013063391A (en) * | 2011-09-16 | 2013-04-11 | Shin-Etsu Chemical Co Ltd | Method of forming cured thin film using photocurable silicone resin composition |
| EP2570463A1 (en) * | 2011-09-16 | 2013-03-20 | Shin-Etsu Chemical Co., Ltd. | Method of producing cured thin film using photocurable silicone resin composition |
| WO2013084425A1 (en) * | 2011-12-09 | 2013-06-13 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Composition for silicone rubber foams, method for producing silicone rubber foam, and silicone rubber foam |
| KR20140109794A (en) | 2011-12-09 | 2014-09-16 | 모멘티브 파포만스 마테리아루즈 쟈판 고도가이샤 | Composition for silicone rubber foams, method for producing silicone rubber foam, and silicone rubber foam |
| US9074097B2 (en) | 2011-12-09 | 2015-07-07 | Momentive Performance Materials Japan Llc | Composition for silicone rubber foam, manufacturing method of silicone rubber foam, and silicone rubber foam |
| US9340655B2 (en) | 2011-12-09 | 2016-05-17 | Momentive Performance Materials Japan Llc | Composition for silicone rubber foam, manufacturing method of silicone rubber foam, and silicone rubber foam |
| US8865784B2 (en) | 2011-12-09 | 2014-10-21 | Momentive Performance Materials Japan Llc | Composition for silicone rubber foam, manufacturing method of silicone rubber foam, and silicone rubber foam |
| JP5281206B1 (en) * | 2011-12-09 | 2013-09-04 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Silicone rubber foam composition, method for producing silicone rubber foam, and silicone rubber foam |
| US8809480B2 (en) | 2012-01-16 | 2014-08-19 | Shin-Etsu Chemical Co., Ltd. | Silicone resin composition, luminous substance-containing wavelength-converting film, and cured product thereof |
| JP2013147546A (en) * | 2012-01-18 | 2013-08-01 | Shin-Etsu Chemical Co Ltd | Uv-curable adhesive silicone composition, uv-curable adhesive silicone composition sheet, optical semiconductor apparatus, and method for manufacturing the same |
| US9268227B2 (en) | 2012-01-18 | 2016-02-23 | Shin-Etsu Chemical Co., Ltd. | UV-curable adhesive silicone composition, UV-curable adhesive silicone composition sheet, optical semiconductor apparatus and method for manufacturing the same |
| KR20130084997A (en) | 2012-01-18 | 2013-07-26 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Uv curable adhesive silicone composition, uv curable adhesive silicone composition sheet, optical semiconductor device and method for producing the same |
| EP2617791A1 (en) | 2012-01-18 | 2013-07-24 | Shin-Etsu Chemical Co., Ltd. | UV-curable adhesive silicone composition, UV-curable adhesive silicone composition sheet, optical semiconductor apparatus and method for manufacturing the same |
| EP2628763A1 (en) | 2012-02-20 | 2013-08-21 | Evonik Goldschmidt GmbH | Branched polysiloxanes and their use |
| DE102012202521A1 (en) | 2012-02-20 | 2013-08-22 | Evonik Goldschmidt Gmbh | Branched polysiloxanes and their use |
| US8946369B2 (en) | 2012-02-20 | 2015-02-03 | Evonik Degussa Gmbh | Branched polysiloxanes and use of these |
| US20140103546A1 (en) * | 2012-10-16 | 2014-04-17 | Brewer Science Inc. | Silicone polymers with high refractive indices and extended pot life |
| US9117757B2 (en) * | 2012-10-16 | 2015-08-25 | Brewer Science Inc. | Silicone polymers with high refractive indices and extended pot life |
| KR20140124724A (en) | 2013-04-17 | 2014-10-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Photocurable fluoropolyether gel composition, method for curing the same, gel cured product thereof and gel products using cured product thereof |
| CN103320087A (en) * | 2013-06-03 | 2013-09-25 | 曹坚林 | Liquid optical transparent organic silicon material and preparation method of same |
| WO2015056483A1 (en) * | 2013-10-18 | 2015-04-23 | 信越化学工業株式会社 | Ultraviolet-ray-curable organopolysiloxane composition, and method for producing printing material |
| US9714344B2 (en) | 2013-10-18 | 2017-07-25 | Shin-Etsu Chemical Co., Ltd. | Ultraviolet-ray-curable organopolysiloxane composition, and method for producing printing material |
| WO2016098883A1 (en) * | 2014-12-18 | 2016-06-23 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, lens or cover for light source, and molding method |
| KR102548854B1 (en) | 2014-12-18 | 2023-06-27 | 모멘티브 파포만스 마테리아루즈 쟈판 고도가이샤 | Polyorganosiloxane composition for molding, optical member, lens or cover for light source, and molding method |
| JP5956698B1 (en) * | 2014-12-18 | 2016-07-27 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Polyorganosiloxane composition for molding, optical member, lens or cover for light source, and molding method |
| CN107001803A (en) * | 2014-12-18 | 2017-08-01 | 迈图高新材料日本合同公司 | Flame retardance poly organosiloxane composition, anti-flammability solidfied material, optics part, light source lens or lid and forming method |
| KR20170098818A (en) * | 2014-12-18 | 2017-08-30 | 모멘티브 파포만스 마테리아루즈 쟈판 고도가이샤 | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, lens or cover for light source, and molding method |
| KR20170098819A (en) * | 2014-12-18 | 2017-08-30 | 모멘티브 파포만스 마테리아루즈 쟈판 고도가이샤 | Polyorganosiloxane composition for molding, optical member, lens or cover for light source, and molding method |
| WO2016098884A1 (en) * | 2014-12-18 | 2016-06-23 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Polyorganosiloxane composition for molding, optical member, lens or cover for light source, and molding method |
| CN107001803B (en) * | 2014-12-18 | 2021-02-26 | 迈图高新材料日本合同公司 | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, lens or cover for light source, and molding method |
| US10752774B2 (en) | 2014-12-18 | 2020-08-25 | Momentive Performance Materials Japan Llc | Polyorganosiloxane composition for molding, optical member, light source lens or cover, and molding method |
| JP5956697B1 (en) * | 2014-12-18 | 2016-07-27 | モメンティブ・パフォーマンス・マテリアルズ・ジャパン合同会社 | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, light source lens or cover, and molding method |
| KR102550073B1 (en) | 2014-12-18 | 2023-06-29 | 모멘티브 파포만스 마테리아루즈 쟈판 고도가이샤 | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, lens or cover for light source, and molding method |
| US10301473B2 (en) | 2014-12-18 | 2019-05-28 | Momentive Performance Materials Japan Llc | Flame-retardant polyorganosiloxane composition, flame-retardant cured product, optical member, light source lens or cover, and molding method |
| JP2018538423A (en) * | 2015-11-06 | 2018-12-27 | ワッカー ケミー アクチエンゲゼルシャフトWacker Chemie AG | Method of laminating base material and product manufactured thereby |
| KR102699279B1 (en) * | 2016-03-14 | 2024-08-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Additional one-time heat-curable thermally conductive silicone grease composition and method for producing a cured product thereof |
| KR20180124906A (en) * | 2016-03-14 | 2018-11-21 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Heat curing type thermoconductive silicone grease composition and method for producing the cured product |
| WO2019004601A1 (en) * | 2017-06-28 | 2019-01-03 | 비즈텍코리아 주식회사 | Photocurable polysiloxane composition for 3d printing, and dental mold comprising same |
| JP2020529486A (en) * | 2017-08-04 | 2020-10-08 | ワッカー ケミー アクチエンゲゼルシャフトWacker Chemie AG | Stabilization of precious metal catalysts |
| WO2019088066A1 (en) * | 2017-10-31 | 2019-05-09 | ダウ・東レ株式会社 | Organopolysiloxane composition, and half-cured product and cured product produced from same |
| WO2019088067A1 (en) * | 2017-10-31 | 2019-05-09 | ダウ・東レ株式会社 | Method for producing organopolysiloxane cured product, organopolysiloxane cured product, layered product, and optical part |
| US11555120B2 (en) | 2017-10-31 | 2023-01-17 | Dow Toray Co., Ltd. | Organopolysiloxane composition, and half-cured product and cured product produced from same |
| JPWO2019088067A1 (en) * | 2017-10-31 | 2020-11-26 | ダウ・東レ株式会社 | Methods for Producing Hardened Organopolysiloxane, Hardened Organopolysiloxane, Laminates, and Optics |
| JPWO2019088066A1 (en) * | 2017-10-31 | 2020-12-17 | ダウ・東レ株式会社 | Organopolysiloxane composition, and semi-cured and cured products to be produced from this. |
| US11591440B2 (en) | 2017-10-31 | 2023-02-28 | Dow Toray Co., Ltd. | Method for producing organopolysiloxane cured product, organopolysiloxane cured product, layered product, and optical part |
| KR20190074225A (en) | 2017-12-19 | 2019-06-27 | 신에쓰 가가꾸 고교 가부시끼가이샤 | Ultraviolet-curable resin composition, adhesive and cured product |
| JPWO2020090797A1 (en) * | 2018-10-30 | 2021-09-30 | ダウ・東レ株式会社 | Curing-reactive silicone compositions, cured products thereof, and their uses |
| WO2020090797A1 (en) * | 2018-10-30 | 2020-05-07 | ダウ・東レ株式会社 | Curable reactive silicone composition, cured product thereof and uses of composition and cured product |
| JP7419251B2 (en) | 2018-10-30 | 2024-01-22 | ダウ・東レ株式会社 | Curing reactive silicone compositions, cured products thereof, and their uses |
| CN109825245B (en) * | 2018-12-30 | 2022-04-01 | 苏州桐力光电股份有限公司 | Full-lamination liquid crystal module sealing process |
| CN109825244A (en) * | 2018-12-30 | 2019-05-31 | 苏州桐力光电股份有限公司 | One kind can ultraviolet lighting and the double cured clear silicone gels of heating |
| CN109825245A (en) * | 2018-12-30 | 2019-05-31 | 苏州桐力光电股份有限公司 | A kind of full fitting liquid crystal module sealing technology |
| WO2024000245A1 (en) * | 2022-06-29 | 2024-01-04 | Dow Toray Co., Ltd. | Uv curable organopolysiloxane composition and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5342830B2 (en) | 2013-11-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5342830B2 (en) | Photocurable organopolysiloxane composition | |
| US8389650B2 (en) | Curable polyorganosiloxane composition | |
| JP5247979B2 (en) | Polyorganosiloxane composition giving a transparent cured product | |
| JP5972511B2 (en) | Curable organopolysiloxane composition and cured product thereof | |
| TWI544665B (en) | Silicon oxide compositions for semiconductor encapsulation | |
| JP4071639B2 (en) | Silicone resin composition for light emitting diode element | |
| KR20040047716A (en) | Silicone Resin Composition for Light Emitting Diode Device | |
| JP2011012264A (en) | Method for producing silicone coating and silicone molded article from photo crosslinkable silicone mixture | |
| JP2007039483A (en) | Curable polyorganosiloxane composition | |
| JP2005162859A (en) | Addition reaction curing type organopolysiloxane resin composition and optical member | |
| JP2009185226A (en) | Heat curable silicone composition and molded product using the same for optical member | |
| WO2019003995A1 (en) | Curable silicone composition for die bonding use | |
| TW201829628A (en) | Reactive hot-melt silicone-filled cartridge and method of producing reactive hot-melt silicone | |
| JP2019218495A (en) | Ultraviolet-curable silicone rubber composition and cured product | |
| JPWO2016136245A1 (en) | Organopolysiloxane, process for producing the same, and curable silicone composition | |
| CN113025055B (en) | Curable white silicone composition, reflective material for optical semiconductor device, and optical semiconductor device | |
| US8674037B2 (en) | Silicon-containing polymer, method of manufacturing thereof, and curable polymer composition | |
| JP2010109034A (en) | Silicone resin composition for light-emitting diode element, and light-emitting diode element | |
| WO2015194159A1 (en) | Organopolysiloxane and method for producing same | |
| JP6319168B2 (en) | Method for producing condensation reaction product, method for producing room temperature curable organopolysiloxane composition containing the condensation reaction product | |
| JP2017200962A (en) | Adhesiveness imparting agent and curable resin composition | |
| JP5913537B2 (en) | Method for producing curable organopolysiloxane composition | |
| JP2014227544A (en) | Resin composition for sealing optical semiconductor element, and optical semiconductor device using the same | |
| JP7337470B1 (en) | UV curable silicone composition | |
| JP2006335845A (en) | Polyorganosiloxane composition which gives cured product having high refractive index |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20110627 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20121010 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20121016 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20121213 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20130730 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20130812 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5342830 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |