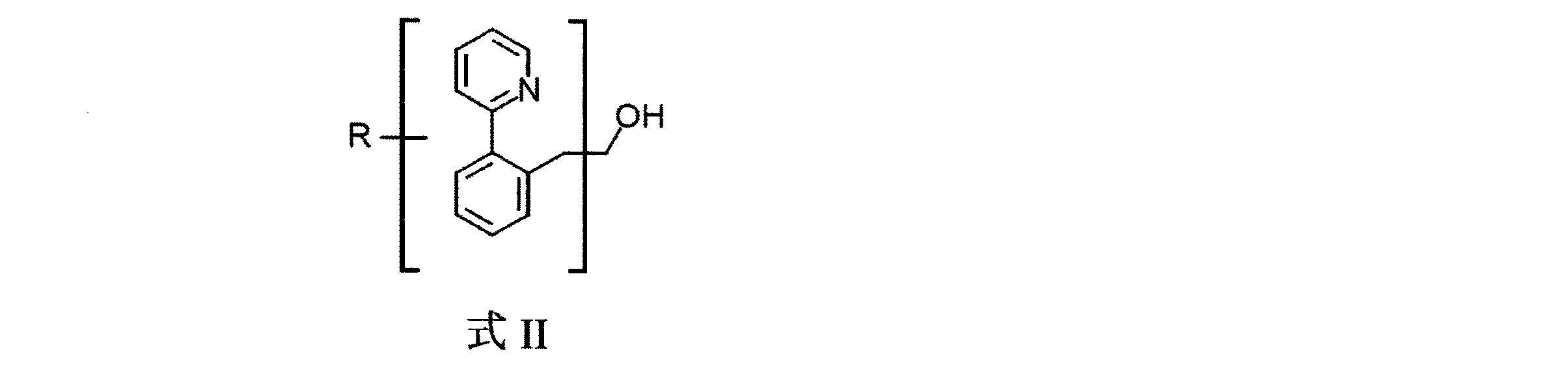JP2010044065A - 有機リン化合物の電気的検出および/または定量 - Google Patents
有機リン化合物の電気的検出および/または定量 Download PDFInfo
- Publication number
- JP2010044065A JP2010044065A JP2009171470A JP2009171470A JP2010044065A JP 2010044065 A JP2010044065 A JP 2010044065A JP 2009171470 A JP2009171470 A JP 2009171470A JP 2009171470 A JP2009171470 A JP 2009171470A JP 2010044065 A JP2010044065 A JP 2010044065A
- Authority
- JP
- Japan
- Prior art keywords
- semiconductor material
- group
- detection
- grafted
- acceptor molecule
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 150000002903 organophosphorus compounds Chemical class 0.000 title claims abstract description 58
- 238000001514 detection method Methods 0.000 title claims abstract description 35
- 239000004065 semiconductor Substances 0.000 claims abstract description 50
- 239000000463 material Substances 0.000 claims abstract description 49
- 230000008859 change Effects 0.000 claims abstract description 13
- 238000000034 method Methods 0.000 claims abstract description 12
- 150000003138 primary alcohols Chemical class 0.000 claims abstract description 12
- 150000003512 tertiary amines Chemical class 0.000 claims abstract description 9
- 125000000524 functional group Chemical group 0.000 claims description 22
- 238000011002 quantification Methods 0.000 claims description 17
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 14
- 229910052710 silicon Inorganic materials 0.000 claims description 14
- 239000010703 silicon Substances 0.000 claims description 14
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 10
- 229910052717 sulfur Inorganic materials 0.000 claims description 10
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 9
- 150000001875 compounds Chemical class 0.000 claims description 9
- 239000011593 sulfur Substances 0.000 claims description 9
- 150000001412 amines Chemical class 0.000 claims description 8
- 239000002041 carbon nanotube Substances 0.000 claims description 7
- 229910021393 carbon nanotube Inorganic materials 0.000 claims description 7
- 238000012360 testing method Methods 0.000 claims description 7
- 239000002253 acid Substances 0.000 claims description 6
- 150000001336 alkenes Chemical group 0.000 claims description 6
- 150000001345 alkine derivatives Chemical class 0.000 claims description 6
- 150000003254 radicals Chemical class 0.000 claims description 6
- 239000012954 diazonium Substances 0.000 claims description 5
- 150000001989 diazonium salts Chemical class 0.000 claims description 5
- 239000002070 nanowire Substances 0.000 claims description 5
- 239000002243 precursor Substances 0.000 claims description 5
- WUPHOULIZUERAE-UHFFFAOYSA-N 3-(oxolan-2-yl)propanoic acid Chemical compound OC(=O)CCC1CCCO1 WUPHOULIZUERAE-UHFFFAOYSA-N 0.000 claims description 4
- 229910000530 Gallium indium arsenide Inorganic materials 0.000 claims description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 claims description 4
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 claims description 4
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 claims description 4
- KXNLCSXBJCPWGL-UHFFFAOYSA-N [Ga].[As].[In] Chemical compound [Ga].[As].[In] KXNLCSXBJCPWGL-UHFFFAOYSA-N 0.000 claims description 4
- 125000003118 aryl group Chemical group 0.000 claims description 4
- 229910052980 cadmium sulfide Inorganic materials 0.000 claims description 4
- 229910052799 carbon Inorganic materials 0.000 claims description 4
- 229910052732 germanium Inorganic materials 0.000 claims description 4
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 claims description 4
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 claims description 4
- 229910052737 gold Inorganic materials 0.000 claims description 4
- 239000010931 gold Substances 0.000 claims description 4
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 4
- 239000005083 Zinc sulfide Substances 0.000 claims description 3
- 238000003402 intramolecular cyclocondensation reaction Methods 0.000 claims description 3
- 239000002071 nanotube Substances 0.000 claims description 3
- 235000011007 phosphoric acid Nutrition 0.000 claims description 3
- AYNNSCRYTDRFCP-UHFFFAOYSA-N triazene Chemical compound NN=N AYNNSCRYTDRFCP-UHFFFAOYSA-N 0.000 claims description 3
- 229910052984 zinc sulfide Inorganic materials 0.000 claims description 3
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 claims description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 2
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 claims description 2
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 2
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 2
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 claims description 2
- 125000003277 amino group Chemical group 0.000 claims description 2
- 229910052793 cadmium Inorganic materials 0.000 claims description 2
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 claims description 2
- AQCDIIAORKRFCD-UHFFFAOYSA-N cadmium selenide Chemical compound [Cd]=[Se] AQCDIIAORKRFCD-UHFFFAOYSA-N 0.000 claims description 2
- 150000001735 carboxylic acids Chemical group 0.000 claims description 2
- 229910052802 copper Inorganic materials 0.000 claims description 2
- 239000010949 copper Substances 0.000 claims description 2
- 230000005669 field effect Effects 0.000 claims description 2
- 238000007306 functionalization reaction Methods 0.000 claims description 2
- 229910052733 gallium Inorganic materials 0.000 claims description 2
- 229910052738 indium Inorganic materials 0.000 claims description 2
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 claims description 2
- 239000007788 liquid Substances 0.000 claims description 2
- 229910052751 metal Inorganic materials 0.000 claims description 2
- 239000002184 metal Substances 0.000 claims description 2
- 229910052759 nickel Inorganic materials 0.000 claims description 2
- 229910052763 palladium Inorganic materials 0.000 claims description 2
- 150000003016 phosphoric acids Chemical class 0.000 claims description 2
- 229910052697 platinum Inorganic materials 0.000 claims description 2
- 229910052709 silver Inorganic materials 0.000 claims description 2
- 239000004332 silver Substances 0.000 claims description 2
- 229910052719 titanium Inorganic materials 0.000 claims description 2
- 239000010936 titanium Substances 0.000 claims description 2
- 229910052725 zinc Inorganic materials 0.000 claims description 2
- 239000011701 zinc Substances 0.000 claims description 2
- 239000011787 zinc oxide Substances 0.000 claims description 2
- DRDVZXDWVBGGMH-UHFFFAOYSA-N zinc;sulfide Chemical compound [S-2].[Zn+2] DRDVZXDWVBGGMH-UHFFFAOYSA-N 0.000 claims description 2
- 150000004654 triazenes Chemical class 0.000 claims 1
- 239000002904 solvent Substances 0.000 abstract description 3
- 238000007363 ring formation reaction Methods 0.000 description 11
- 239000000243 solution Substances 0.000 description 10
- -1 phosphate ester Chemical class 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 7
- MUCZHBLJLSDCSD-UHFFFAOYSA-N diisopropyl fluorophosphate Chemical compound CC(C)OP(F)(=O)OC(C)C MUCZHBLJLSDCSD-UHFFFAOYSA-N 0.000 description 6
- 229960005051 fluostigmine Drugs 0.000 description 6
- 150000003573 thiols Chemical class 0.000 description 6
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 239000007789 gas Substances 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 229910019142 PO4 Inorganic materials 0.000 description 4
- 239000010452 phosphate Substances 0.000 description 4
- BBEAQIROQSPTKN-UHFFFAOYSA-N pyrene Chemical compound C1=CC=C2C=CC3=CC=CC4=CC=C1C2=C43 BBEAQIROQSPTKN-UHFFFAOYSA-N 0.000 description 4
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 4
- 102000003914 Cholinesterases Human genes 0.000 description 3
- 108090000322 Cholinesterases Proteins 0.000 description 3
- 102000004190 Enzymes Human genes 0.000 description 3
- 108090000790 Enzymes Proteins 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- DYAHQFWOVKZOOW-UHFFFAOYSA-N Sarin Chemical compound CC(C)OP(C)(F)=O DYAHQFWOVKZOOW-UHFFFAOYSA-N 0.000 description 3
- 229940048961 cholinesterase Drugs 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 229940088598 enzyme Drugs 0.000 description 3
- 239000002917 insecticide Substances 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 238000010534 nucleophilic substitution reaction Methods 0.000 description 3
- 239000000575 pesticide Substances 0.000 description 3
- 102100033639 Acetylcholinesterase Human genes 0.000 description 2
- 108010022752 Acetylcholinesterase Proteins 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 2
- OIPILFWXSMYKGL-UHFFFAOYSA-N acetylcholine Chemical compound CC(=O)OCC[N+](C)(C)C OIPILFWXSMYKGL-UHFFFAOYSA-N 0.000 description 2
- 229960004373 acetylcholine Drugs 0.000 description 2
- 229940022698 acetylcholinesterase Drugs 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 125000003158 alcohol group Chemical group 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O ammonium group Chemical group [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 2
- 239000004020 conductor Substances 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 238000010511 deprotection reaction Methods 0.000 description 2
- LGTLXDJOAJDFLR-UHFFFAOYSA-N diethyl chlorophosphate Chemical compound CCOP(Cl)(=O)OCC LGTLXDJOAJDFLR-UHFFFAOYSA-N 0.000 description 2
- VONWDASPFIQPDY-UHFFFAOYSA-N dimethyl methylphosphonate Chemical compound COP(C)(=O)OC VONWDASPFIQPDY-UHFFFAOYSA-N 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- GVEPBJHOBDJJJI-UHFFFAOYSA-N fluoranthrene Natural products C1=CC(C2=CC=CC=C22)=C3C2=CC=CC3=C1 GVEPBJHOBDJJJI-UHFFFAOYSA-N 0.000 description 2
- 235000013305 food Nutrition 0.000 description 2
- 230000001537 neural effect Effects 0.000 description 2
- 231100000189 neurotoxic Toxicity 0.000 description 2
- 230000002887 neurotoxic effect Effects 0.000 description 2
- 150000002894 organic compounds Chemical class 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 229920000301 poly(3-hexylthiophene-2,5-diyl) polymer Polymers 0.000 description 2
- 150000004032 porphyrins Chemical class 0.000 description 2
- 125000001453 quaternary ammonium group Chemical group 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 230000027756 respiratory electron transport chain Effects 0.000 description 2
- 239000002094 self assembled monolayer Substances 0.000 description 2
- 239000013545 self-assembled monolayer Substances 0.000 description 2
- 125000006850 spacer group Chemical group 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- ONDSBJMLAHVLMI-UHFFFAOYSA-N trimethylsilyldiazomethane Chemical compound C[Si](C)(C)[CH-][N+]#N ONDSBJMLAHVLMI-UHFFFAOYSA-N 0.000 description 2
- KCGZGJOBKAXVSU-UHFFFAOYSA-N (4-iodophenyl)methanamine Chemical compound NCC1=CC=C(I)C=C1 KCGZGJOBKAXVSU-UHFFFAOYSA-N 0.000 description 1
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 1
- 229920003026 Acene Polymers 0.000 description 1
- 241000252506 Characiformes Species 0.000 description 1
- 239000005944 Chlorpyrifos Substances 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- RWSOTUBLDIXVET-UHFFFAOYSA-N Dihydrogen sulfide Chemical class S RWSOTUBLDIXVET-UHFFFAOYSA-N 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 229910010082 LiAlH Inorganic materials 0.000 description 1
- 239000005949 Malathion Substances 0.000 description 1
- 206010033799 Paralysis Diseases 0.000 description 1
- YGYAWVDWMABLBF-UHFFFAOYSA-N Phosgene Chemical compound ClC(Cl)=O YGYAWVDWMABLBF-UHFFFAOYSA-N 0.000 description 1
- 239000005921 Phosmet Substances 0.000 description 1
- 229920000265 Polyparaphenylene Polymers 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- GRXKLBBBQUKJJZ-UHFFFAOYSA-N Soman Chemical compound CC(C)(C)C(C)OP(C)(F)=O GRXKLBBBQUKJJZ-UHFFFAOYSA-N 0.000 description 1
- 238000003477 Sonogashira cross-coupling reaction Methods 0.000 description 1
- PJVJTCIRVMBVIA-JTQLQIEISA-N [dimethylamino(ethoxy)phosphoryl]formonitrile Chemical compound CCO[P@@](=O)(C#N)N(C)C PJVJTCIRVMBVIA-JTQLQIEISA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 125000000218 acetic acid group Chemical group C(C)(=O)* 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000008064 anhydrides Chemical class 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 150000004982 aromatic amines Chemical class 0.000 description 1
- 125000006615 aromatic heterocyclic group Chemical group 0.000 description 1
- 125000000732 arylene group Chemical group 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 150000001540 azides Chemical class 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 125000003636 chemical group Chemical group 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- SBPBAQFWLVIOKP-UHFFFAOYSA-N chlorpyrifos Chemical compound CCOP(=S)(OCC)OC1=NC(Cl)=C(Cl)C=C1Cl SBPBAQFWLVIOKP-UHFFFAOYSA-N 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 229920001940 conductive polymer Polymers 0.000 description 1
- 239000007822 coupling agent Substances 0.000 description 1
- 230000000593 degrading effect Effects 0.000 description 1
- FHIVAFMUCKRCQO-UHFFFAOYSA-N diazinon Chemical compound CCOP(=S)(OCC)OC1=CC(C)=NC(C(C)C)=N1 FHIVAFMUCKRCQO-UHFFFAOYSA-N 0.000 description 1
- 150000008049 diazo compounds Chemical class 0.000 description 1
- OEBRKCOSUFCWJD-UHFFFAOYSA-N dichlorvos Chemical compound COP(=O)(OC)OC=C(Cl)Cl OEBRKCOSUFCWJD-UHFFFAOYSA-N 0.000 description 1
- 229950001327 dichlorvos Drugs 0.000 description 1
- JXSJBGJIGXNWCI-UHFFFAOYSA-N diethyl 2-[(dimethoxyphosphorothioyl)thio]succinate Chemical compound CCOC(=O)CC(SP(=S)(OC)OC)C(=O)OCC JXSJBGJIGXNWCI-UHFFFAOYSA-N 0.000 description 1
- 150000002019 disulfides Chemical class 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000005284 excitation Effects 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 239000004009 herbicide Substances 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 231100000171 higher toxicity Toxicity 0.000 description 1
- 150000004678 hydrides Chemical class 0.000 description 1
- 238000006459 hydrosilylation reaction Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 150000003949 imides Chemical class 0.000 description 1
- 230000036540 impulse transmission Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 239000012212 insulator Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- IQZZFVDIZRWADY-UHFFFAOYSA-N isocoumarin Chemical compound C1=CC=C2C(=O)OC=CC2=C1 IQZZFVDIZRWADY-UHFFFAOYSA-N 0.000 description 1
- 239000012948 isocyanate Substances 0.000 description 1
- 150000002513 isocyanates Chemical class 0.000 description 1
- 238000011031 large-scale manufacturing process Methods 0.000 description 1
- 150000002642 lithium compounds Chemical class 0.000 description 1
- 229960000453 malathion Drugs 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- AUHZEENZYGFFBQ-UHFFFAOYSA-N mesitylene Substances CC1=CC(C)=CC(C)=C1 AUHZEENZYGFFBQ-UHFFFAOYSA-N 0.000 description 1
- 125000001827 mesitylenyl group Chemical group [H]C1=C(C(*)=C(C([H])=C1C([H])([H])[H])C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 210000005036 nerve Anatomy 0.000 description 1
- 210000002569 neuron Anatomy 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 230000009965 odorless effect Effects 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 150000002901 organomagnesium compounds Chemical class 0.000 description 1
- 150000002902 organometallic compounds Chemical class 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- LCCNCVORNKJIRZ-UHFFFAOYSA-N parathion Chemical compound CCOP(=S)(OCC)OC1=CC=C([N+]([O-])=O)C=C1 LCCNCVORNKJIRZ-UHFFFAOYSA-N 0.000 description 1
- RLBIQVVOMOPOHC-UHFFFAOYSA-N parathion-methyl Chemical compound COP(=S)(OC)OC1=CC=C([N+]([O-])=O)C=C1 RLBIQVVOMOPOHC-UHFFFAOYSA-N 0.000 description 1
- SLIUAWYAILUBJU-UHFFFAOYSA-N pentacene Chemical compound C1=CC=CC2=CC3=CC4=CC5=CC=CC=C5C=C4C=C3C=C21 SLIUAWYAILUBJU-UHFFFAOYSA-N 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- CSHWQDPOILHKBI-UHFFFAOYSA-N peryrene Natural products C1=CC(C2=CC=CC=3C2=C2C=CC=3)=C3C2=CC=CC3=C1 CSHWQDPOILHKBI-UHFFFAOYSA-N 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- LMNZTLDVJIUSHT-UHFFFAOYSA-N phosmet Chemical compound C1=CC=C2C(=O)N(CSP(=S)(OC)OC)C(=O)C2=C1 LMNZTLDVJIUSHT-UHFFFAOYSA-N 0.000 description 1
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 description 1
- 125000004437 phosphorous atom Chemical group 0.000 description 1
- 150000003018 phosphorus compounds Chemical class 0.000 description 1
- 229920000553 poly(phenylenevinylene) Polymers 0.000 description 1
- 229920000548 poly(silane) polymer Polymers 0.000 description 1
- 125000005575 polycyclic aromatic hydrocarbon group Chemical group 0.000 description 1
- 125000003367 polycyclic group Chemical group 0.000 description 1
- 229920002098 polyfluorene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920000128 polypyrrole Polymers 0.000 description 1
- 230000005180 public health Effects 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000023882 response to organophosphorus Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 229910000077 silane Inorganic materials 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 230000006641 stabilisation Effects 0.000 description 1
- 238000011105 stabilization Methods 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N sulfuric acid Substances OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 230000000946 synaptic effect Effects 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- DUYAAUVXQSMXQP-UHFFFAOYSA-M thioacetate Chemical compound CC([S-])=O DUYAAUVXQSMXQP-UHFFFAOYSA-M 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000018405 transmission of nerve impulse Effects 0.000 description 1
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 150000003752 zinc compounds Chemical class 0.000 description 1
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/26—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating electrochemical variables; by using electrolysis or electrophoresis
- G01N27/403—Cells and electrode assemblies
- G01N27/414—Ion-sensitive or chemical field-effect transistors, i.e. ISFETS or CHEMFETS
- G01N27/4146—Ion-sensitive or chemical field-effect transistors, i.e. ISFETS or CHEMFETS involving nanosized elements, e.g. nanotubes, nanowires
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N27/00—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means
- G01N27/02—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating impedance
- G01N27/04—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating impedance by investigating resistance
- G01N27/12—Investigating or analysing materials by the use of electric, electrochemical, or magnetic means by investigating impedance by investigating resistance of a solid body in dependence upon absorption of a fluid; of a solid body in dependence upon reaction with a fluid, for detecting components in the fluid
- G01N27/125—Composition of the body, e.g. the composition of its sensitive layer
- G01N27/127—Composition of the body, e.g. the composition of its sensitive layer comprising nanoparticles
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Analytical Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Electrochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nanotechnology (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Physics & Mathematics (AREA)
- Immunology (AREA)
- Pathology (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Molecular Biology (AREA)
- Investigating Or Analyzing Materials By The Use Of Electric Means (AREA)
- Investigating Or Analyzing Materials By The Use Of Fluid Adsorption Or Reactions (AREA)
Abstract
【解決手段】半導体材料で隔てられたソース電極とドレイン電極とを備える電気デバイスにおいて、基Rと、3級アミンに空間的に近接している1級アルコールとを含み、前記1級アルコールが、有機リン化合物の存在下で前記3級アミンと反応することができる少なくとも1つの受容体分子が、基Rによって、電極(複数)の一方の上または半導体材料の上にグラフトされていることを特徴とする電気デバイスと、前記2つの電極間の正電荷の変化を検出するためのデバイスとを備える、有機リン化合物を検出および/または定量するためのデバイス。
【選択図】なし
Description
− 半導体材料で隔てられたソース電極とドレイン電極とを備える電気デバイスにおいて、基Rと、3級アミンに空間的に近接している1級アルコールとを含み、前記1級アルコールは有機リン化合物の存在下で前記3級アミンと反応することができる、少なくとも1つの受容体分子が、基Rによって、前記電極の一方の上または前記半導体材料の上にグラフトされている、電気デバイスと、
− 前記2つの電極間の正電荷の変化を検出するためのデバイスと
を備える、有機リン化合物を検出および/または定量するためのデバイスに関する。
受容体分子は、下記一般式IIの分子でもよい。
− ジアゾニウム塩、トリアゼン、いずれかのフリーラジカル前駆体、またはカーボンナノチューブの炭素原子と共有結合を形成できるいずれかの分子;
− 芳香族基(例えばピレン、アントラセン、ポルフィリンなど)またはナノチューブの非共有結合(超分子)官能化を可能にするアミン群の誘導体;
− 必要に応じて、エステルまたはアミドの形成のための(例えば、カップリング剤での)化学的活性化後の、カーボンナノチューブの表面に存在するカルボン酸との反応のためのアミンまたはアルコール。
a)液体状または気体状の試験サンプルを、前述した通りの本発明の検出デバイスの存在下に置くステップ;
b)前記デバイスの抵抗、コンダクタンス若しくは相互コンダクタンスの差異を検出および/または測定することによって、有機リン化合物によって誘導された受容体分子の分子内環化の反応により発生した正電荷を証明するステップ
を含む方法に関する。
1.1.試験デバイスの製造
半導体材料が、厚さ77nmの酸化シリコンの層上にエッチングされた、幅280nm、長さ4μm、厚さ16nmのエッチングシリコンナノワイヤーである、SOI上に電気デバイスを製作した。
官能基Rが4−エチニルベンジルである受容体をKemp三酸を用いて合成した。第1のステップで、塩化チオニル中の還流によってKemp三酸を無水物(生成物1)に変換する。残存塩化チオニルの蒸発後、ピリジン中で還流させながら生成物1を4−ヨードベンジルアミンと反応させ、次にトリメチルシリルジアゾメタンで処理する。園頭反応は、トリメチルシリル基で保護されたアルキンの導入を可能にする。分子3のLiAlH4による処理は、アミンおよびアルコール基を与えるように、それぞれイミドおよびエステル基を還元することができる。予想外に、シリル化基の脱保護が観察される。DFPによる処理で得られたKemp誘導体4の環化をNMR管内で行った。環化は完了する。
熱的ヒドロシリル化によって、試験デバイス上に受容体をグラフトする。濃硫酸と30%の過酸化水素水の3対1混合物で構成されるピラニア溶液でデバイスを洗浄し、次に1質量%のHF溶液で処理する。この活性化デバイスを、メシチレン中の受容体4の0.5mM溶液内で還流させる。
実施例1で得られたデバイスを用いて有機リン化合物の存在を検出した。このため、ジイソプロピルフルオロホスフェート蒸気(10ppm)を含むチャンバー内に1分間、本発明のデバイスを置いた。
Claims (18)
- 1)半導体材料で隔てられたソース電極とドレイン電極とを備える電気デバイスにおいて、基Rと、3級アミンに空間的に近接している1級アルコールとを含み、前記1級アルコールが有機リン化合物の存在下で前記3級アミンと反応することができる、少なくとも1つの受容体分子が、前記基Rによって、前記電極の一方の上または前記半導体材料の上にグラフトされていることを特徴とする電気デバイスと、
2)前記2つの電極間の正電荷の変化を検出するためのデバイスと
を備える、有機リン化合物を検出および/または定量するためのデバイス。 - 前記半導体材料が、炭素、シリコン、ゲルマニウム、亜鉛、ガリウム、インジウム、カドミウムまたは有機半導体材料に基づく材料から選択されることを特徴とする、請求項1に記載の検出および/または定量デバイス。
- 前記半導体材料が、シリコンナノワイヤーおよび/またはカーボンナノチューブで構成されることを特徴とする、請求項1または2に記載の検出および/または定量デバイス。
- 前記半導体材料がシリコンナノワイヤーであることを特徴とする、請求項1から3のいずれか一項に記載の検出および/または定量デバイス。
- 前記電極の材料が、金、銀、パラジウム、白金、チタン、ドープシリコン、銅またはニッケルから選択される金属であることを特徴とする、請求項1から4のいずれか一項に記載の検出および/または定量デバイス。
- 前記電気デバイスがレジスタ型であることを特徴とする、請求項1から7のいずれか一項に記載の検出および/または定量デバイス。
- 前記電気デバイスが電界効果トランジスタ型であることを特徴とする、請求項1から7のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料がシリコンであり、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、アルキン、アルケン、ジアゾニウム塩、トリアゼンまたはフリーラジカル前駆体から選択されることを特徴とする、請求項1から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料がカーボンナノチューブで構成され、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、ジアゾニウム塩、トリアゼン、フリーラジカル前駆体、芳香族基、前記ナノチューブの非共有結合官能化を可能にするアミン群の誘導体、アミンまたはアルコールから選択されることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料がゲルマニウム系であり、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、アルキンまたはアルケンであることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料がインジウムガリウムヒ素(InGaAs)で構成され、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、イオウ含有化合物であることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料がセレン化カドミウム(CdSe)または硫化カドミウム(CdS)系であり、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、アミンおよびイオウ含有化合物から選択されることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料が酸化亜鉛系であり、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、カルボン酸およびリン酸から選択されることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記半導体材料が硫化亜鉛系であり、前記受容体分子が前記半導体材料の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、イオウ含有化合物であることを特徴とする、請求項1から3および5から9のいずれか一項に記載の検出および/または定量デバイス。
- 前記電極が金製であり、前記受容体分子が前記電極の一方の上にグラフトされていること、および前記グラフト化に供される、前記受容体分子の前記基Rのグラフト化官能基が、イオウ含有有機誘導体であることを特徴とする、請求項1から9のいずれか一項に記載の検出および/または定量デバイス。
- 有機リン化合物を検出および/または定量するための方法において、以下のステップ:
− 液体状または気体状の試験サンプルを、請求項1から17のいずれか一項に記載の検出および/または定量デバイスの存在下に置くステップと、
− 前記デバイスの抵抗、コンダクタンスまたは相互コンダクタンスの差異を測定することによって、前記有機リン化合物によって誘導された前記受容体分子の分子内環化の反応により発生した正電荷を証明するステップと
を含むことを特徴とする方法。
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR0804319A FR2934685B1 (fr) | 2008-07-29 | 2008-07-29 | Detection et/ou quantification electrique de composes organophosphores |
| FR0804319 | 2008-07-29 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2010044065A true JP2010044065A (ja) | 2010-02-25 |
| JP5295024B2 JP5295024B2 (ja) | 2013-09-18 |
Family
ID=40386500
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009171470A Expired - Fee Related JP5295024B2 (ja) | 2008-07-29 | 2009-07-22 | 有機リン化合物の電気的検出および/または定量 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8552748B2 (ja) |
| EP (1) | EP2154525B1 (ja) |
| JP (1) | JP5295024B2 (ja) |
| AT (1) | ATE513205T1 (ja) |
| ES (1) | ES2365690T3 (ja) |
| FR (1) | FR2934685B1 (ja) |
| IL (1) | IL199823A0 (ja) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010217159A (ja) * | 2009-03-17 | 2010-09-30 | Jiaotong Univ | ラベルフリーセンサー |
| JP2012032396A (ja) * | 2010-07-19 | 2012-02-16 | Commissariat A L'energie Atomique & Aux Energies Alternatives | ジュール効果の熱活性化による選択的官能基化 |
| US10677770B2 (en) | 2016-09-20 | 2020-06-09 | Kabushiki Kaisha Toshiba | Molecular detection apparatus, molecular detection method, and molecular detector |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2950436B1 (fr) * | 2009-09-18 | 2013-09-20 | Commissariat Energie Atomique | Appareil et procede de detection et/ou de quantification de composes d'interet presents sous forme gazeuse ou en solution dans un solvant |
| FR2962549B1 (fr) | 2010-07-08 | 2012-08-24 | Commissariat Energie Atomique | Dispositif de detection et/ou quantification electrique par empreinte moleculaire de composes organophosphores |
| FR2971849A1 (fr) * | 2011-02-17 | 2012-08-24 | Commissariat Energie Atomique | Procede et dispositif de detection et/ou de dosage de composes organiques dans l'air |
| CN102507714A (zh) * | 2011-11-07 | 2012-06-20 | 华中师范大学 | 有机磷农药生物标志物的检测方法 |
| CN102636537A (zh) * | 2012-04-19 | 2012-08-15 | 湖南大学 | 一种无酶甲基对硫磷检测传感器及其制备和使用方法 |
| FR2996308B1 (fr) * | 2012-10-02 | 2015-05-29 | Commissariat Energie Atomique | Dispositif de detection et/ou de quantification du chlorure de thionyle, son procede de fabrication et procede de detection et/ou de quantification du chlorure de thionyle |
| FR3003352B1 (fr) | 2013-03-13 | 2015-04-17 | Commissariat Energie Atomique | Procede et dispositif de detection colorimetrique ou de dosage de traces de composes organiques toxiques dans l'air |
| CN106645356A (zh) * | 2016-11-30 | 2017-05-10 | 浙江大学 | 一种通用的有机磷农药无酶抑制电化学检测方法 |
| CN113311047B (zh) * | 2021-05-06 | 2022-06-21 | 复旦大学 | 一种纳米金颗粒修饰的伪mos生化分子传感器及其制备方法 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63122692A (ja) * | 1986-10-30 | 1988-05-26 | コミツサレ・ア・レナジイ・アトミツク | 水溶液中の有機リン化合物の回収方法 |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5571401A (en) | 1995-03-27 | 1996-11-05 | California Institute Of Technology | Sensor arrays for detecting analytes in fluids |
| US5846753A (en) * | 1995-05-30 | 1998-12-08 | The United States Of America As Represented By The Secretary Of The Army | Chemiluminescence-based method for rapid and sensitive in-situ detection of organophosphorus compounds and metal ions |
| AU2002215335A1 (en) * | 2000-10-14 | 2002-04-29 | Triton Systems, Inc. | Sensors comprising a semi-conductive polymer |
| DE60127469T2 (de) * | 2001-12-19 | 2007-12-06 | Hitachi High-Technologies Corp. | Potentiometrischer dna-mikroarray, verfahren zu dessen herstellung und verfahren zur nukleinsäureanalyse |
| FR2846338B1 (fr) | 2002-10-28 | 2006-12-29 | Univ Claude Bernard Lyon | Biocapteur electrochimique multi-enzymatique a algues unicellulaires, procede et utilisation mettant en oeuvre un tel biocapteur |
| US7776269B2 (en) * | 2005-03-15 | 2010-08-17 | The United States Of America As Represented By The Secretary Of The Navy | Capacitive based sensing of molecular adsorbates on the surface of single wall nanotubes |
-
2008
- 2008-07-29 FR FR0804319A patent/FR2934685B1/fr not_active Expired - Fee Related
-
2009
- 2009-07-09 ES ES09290555T patent/ES2365690T3/es active Active
- 2009-07-09 EP EP09290555A patent/EP2154525B1/fr active Active
- 2009-07-09 AT AT09290555T patent/ATE513205T1/de not_active IP Right Cessation
- 2009-07-13 IL IL199823A patent/IL199823A0/en not_active IP Right Cessation
- 2009-07-13 US US12/501,659 patent/US8552748B2/en not_active Expired - Fee Related
- 2009-07-22 JP JP2009171470A patent/JP5295024B2/ja not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS63122692A (ja) * | 1986-10-30 | 1988-05-26 | コミツサレ・ア・レナジイ・アトミツク | 水溶液中の有機リン化合物の回収方法 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010217159A (ja) * | 2009-03-17 | 2010-09-30 | Jiaotong Univ | ラベルフリーセンサー |
| JP2012032396A (ja) * | 2010-07-19 | 2012-02-16 | Commissariat A L'energie Atomique & Aux Energies Alternatives | ジュール効果の熱活性化による選択的官能基化 |
| US10677770B2 (en) | 2016-09-20 | 2020-06-09 | Kabushiki Kaisha Toshiba | Molecular detection apparatus, molecular detection method, and molecular detector |
Also Published As
| Publication number | Publication date |
|---|---|
| IL199823A0 (en) | 2010-05-17 |
| ES2365690T3 (es) | 2011-10-10 |
| EP2154525A1 (fr) | 2010-02-17 |
| FR2934685A1 (fr) | 2010-02-05 |
| JP5295024B2 (ja) | 2013-09-18 |
| ATE513205T1 (de) | 2011-07-15 |
| EP2154525B1 (fr) | 2011-06-15 |
| FR2934685B1 (fr) | 2010-09-03 |
| US20100033198A1 (en) | 2010-02-11 |
| US8552748B2 (en) | 2013-10-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5295024B2 (ja) | 有機リン化合物の電気的検出および/または定量 | |
| Tan et al. | An electrochemically reduced graphene oxide chemiresistive sensor for sensitive detection of Hg2+ ion in water samples | |
| Chen et al. | Hg (II) ion detection using thermally reduced graphene oxide decorated with functionalized gold nanoparticles | |
| Palaparthy et al. | Graphene oxide based soil moisture microsensor for in situ agriculture applications | |
| Hao et al. | Ratiometric fluorescent detection of Cu2+ with carbon dots chelated Eu-based metal-organic frameworks | |
| Bolat et al. | Sensitive electrochemical detection of fenitrothion pesticide based on self-assembled peptide-nanotubes modified disposable pencil graphite electrode | |
| Mubeen et al. | Sensitive detection of H2S using gold nanoparticle decorated single-walled carbon nanotubes | |
| US8951473B2 (en) | Devices and methods for determination of species including chemical warfare agents | |
| Sharma et al. | A novel disposable electrochemical immunosensor for phenyl urea herbicide diuron | |
| Chen et al. | A nanoelectronic nose: a hybrid nanowire/carbon nanotube sensor array with integrated micromachined hotplates for sensitive gas discrimination | |
| US20120184041A1 (en) | Apparatus and Method for Detecting and/or Quantifying Compounds of Interest Present in Gaseous Form or Dissolved In A Solvent | |
| Chaudhary et al. | Graphene oxide based electrochemical immunosensor for rapid detection of groundnut bud necrosis orthotospovirus in agricultural crops | |
| Huixiang et al. | A non-enzymatic electro-chemical sensor for organophosphorus nerve agents mimics and pesticides detection | |
| JP5416616B2 (ja) | 水銀誘導体の電気的検出および定量化 | |
| US20130244334A1 (en) | Device for the detection and/or electrical quantification of organophosphorus compounds by means of molecular imprinting | |
| Clavaguera et al. | Sub‐ppm Detection of Nerve Agents Using Chemically Functionalized Silicon Nanoribbon Field‐Effect Transistors | |
| Clavaguera et al. | Development of an autonomous detector for sensing of nerve agents based on functionalized silicon nanowire field-effect transistors | |
| García-Briones et al. | Novel supported nanostructured sensors for chemical warfare agents (CWAs) detection | |
| Simonato et al. | New chemically functionalized nanomaterials for electrical nerve agents sensors | |
| Tang | Development of Nanofibrous Membrane-Based Colorimetric Sensors for Personal Protections Against Fumigant Exposures | |
| KR101757199B1 (ko) | 유기인산염을 선택적으로 흡착하는 칼릭스[4]아렌 유도체 및 이를 이용한 화학가스센서 | |
| Caillier et al. | Innovative Gas Sensors for Sub-ppm Detection of Organophosphorus Nerve Agents by Chemically Functionalized Nanomaterial Based Electrical Devices | |
| Velayudham | Conjugated polymers and their composite material for sensing applications | |
| CN112432977A (zh) | 一种有机场效应晶体管气体传感器及其制备方法 | |
| Nicolini et al. | Nanostructured organic matrices and intelligent sensors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20120628 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20121108 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130129 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130425 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20130521 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20130611 |
|
| R150 | Certificate of patent or registration of utility model |
Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| LAPS | Cancellation because of no payment of annual fees |