JP2004363077A - Nonaqueous electrolytic solution and nonaqueous electrolytic solution secondary battery using it - Google Patents
Nonaqueous electrolytic solution and nonaqueous electrolytic solution secondary battery using it Download PDFInfo
- Publication number
- JP2004363077A JP2004363077A JP2003361115A JP2003361115A JP2004363077A JP 2004363077 A JP2004363077 A JP 2004363077A JP 2003361115 A JP2003361115 A JP 2003361115A JP 2003361115 A JP2003361115 A JP 2003361115A JP 2004363077 A JP2004363077 A JP 2004363077A
- Authority
- JP
- Japan
- Prior art keywords
- group
- secondary battery
- weight
- ethyl
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000008151 electrolyte solution Substances 0.000 title claims abstract description 72
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 24
- 125000005843 halogen group Chemical group 0.000 claims abstract description 21
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 21
- 150000001875 compounds Chemical class 0.000 claims abstract description 14
- 239000011356 non-aqueous organic solvent Substances 0.000 claims abstract description 14
- 125000006165 cyclic alkyl group Chemical group 0.000 claims abstract description 5
- 239000000243 solution Substances 0.000 claims abstract 2
- -1 cyclic ester Chemical class 0.000 claims description 94
- 229910052744 lithium Inorganic materials 0.000 claims description 44
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 claims description 35
- 239000011255 nonaqueous electrolyte Substances 0.000 claims description 30
- 125000004432 carbon atom Chemical group C* 0.000 claims description 20
- 150000005676 cyclic carbonates Chemical class 0.000 claims description 12
- 150000005678 chain carbonates Chemical class 0.000 claims description 11
- 239000003792 electrolyte Substances 0.000 claims description 10
- 229910052723 transition metal Inorganic materials 0.000 claims description 7
- 239000003575 carbonaceous material Substances 0.000 claims description 6
- 239000002905 metal composite material Substances 0.000 claims description 4
- 238000002441 X-ray diffraction Methods 0.000 claims description 3
- 239000012046 mixed solvent Substances 0.000 claims description 3
- 238000003860 storage Methods 0.000 abstract description 27
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 abstract description 8
- 230000006866 deterioration Effects 0.000 abstract description 8
- 229910052799 carbon Inorganic materials 0.000 abstract description 5
- 238000000034 method Methods 0.000 description 11
- 239000011149 active material Substances 0.000 description 10
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 10
- VAYTZRYEBVHVLE-UHFFFAOYSA-N 1,3-dioxol-2-one Chemical compound O=C1OC=CO1 VAYTZRYEBVHVLE-UHFFFAOYSA-N 0.000 description 9
- GQAXWUVTNDQEQR-UHFFFAOYSA-N 1-diethylphosphoryloxyethane Chemical compound CCOP(=O)(CC)CC GQAXWUVTNDQEQR-UHFFFAOYSA-N 0.000 description 9
- 239000000203 mixture Substances 0.000 description 9
- 239000002904 solvent Substances 0.000 description 9
- 238000000354 decomposition reaction Methods 0.000 description 8
- 229910003002 lithium salt Inorganic materials 0.000 description 8
- 159000000002 lithium salts Chemical class 0.000 description 8
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 7
- 239000002131 composite material Substances 0.000 description 7
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 7
- 239000000463 material Substances 0.000 description 7
- YEJRWHAVMIAJKC-UHFFFAOYSA-N 4-Butyrolactone Chemical compound O=C1CCCO1 YEJRWHAVMIAJKC-UHFFFAOYSA-N 0.000 description 6
- 239000000956 alloy Substances 0.000 description 6
- 239000003795 chemical substances by application Substances 0.000 description 6
- 230000000052 comparative effect Effects 0.000 description 6
- 229910052751 metal Inorganic materials 0.000 description 6
- 239000002184 metal Substances 0.000 description 6
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 6
- 239000003960 organic solvent Substances 0.000 description 6
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 5
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 5
- 229910045601 alloy Inorganic materials 0.000 description 5
- 239000004020 conductor Substances 0.000 description 5
- HHNHBFLGXIUXCM-GFCCVEGCSA-N cyclohexylbenzene Chemical compound [CH]1CCCC[C@@H]1C1=CC=CC=C1 HHNHBFLGXIUXCM-GFCCVEGCSA-N 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 239000010410 layer Substances 0.000 description 5
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 5
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 4
- 229910013870 LiPF 6 Inorganic materials 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- 239000011230 binding agent Substances 0.000 description 4
- 229910052802 copper Inorganic materials 0.000 description 4
- 239000010949 copper Substances 0.000 description 4
- 229910052759 nickel Inorganic materials 0.000 description 4
- 238000005498 polishing Methods 0.000 description 4
- 229920002981 polyvinylidene fluoride Polymers 0.000 description 4
- 239000007774 positive electrode material Substances 0.000 description 4
- KKENJBBLHSMHHZ-UHFFFAOYSA-N Cc1ccc(O[PH2]=O)cc1 Chemical compound Cc1ccc(O[PH2]=O)cc1 KKENJBBLHSMHHZ-UHFFFAOYSA-N 0.000 description 3
- OTAZHTQUJLHLMT-UHFFFAOYSA-N Cc1ccccc1O[PH2]=O Chemical compound Cc1ccccc1O[PH2]=O OTAZHTQUJLHLMT-UHFFFAOYSA-N 0.000 description 3
- 239000004698 Polyethylene Substances 0.000 description 3
- WTYQQTPORXDQFL-UHFFFAOYSA-N [PH2](OC1=CC(=CC=C1)C)=O Chemical compound [PH2](OC1=CC(=CC=C1)C)=O WTYQQTPORXDQFL-UHFFFAOYSA-N 0.000 description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 3
- 239000011248 coating agent Substances 0.000 description 3
- 238000000576 coating method Methods 0.000 description 3
- IEJIGPNLZYLLBP-UHFFFAOYSA-N dimethyl carbonate Chemical compound COC(=O)OC IEJIGPNLZYLLBP-UHFFFAOYSA-N 0.000 description 3
- 150000002148 esters Chemical class 0.000 description 3
- 150000002170 ethers Chemical class 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- GTTFJYUWPUKXJH-UHFFFAOYSA-N n-(4-aminophenyl)benzamide Chemical compound C1=CC(N)=CC=C1NC(=O)C1=CC=CC=C1 GTTFJYUWPUKXJH-UHFFFAOYSA-N 0.000 description 3
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 3
- 239000007773 negative electrode material Substances 0.000 description 3
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 125000004437 phosphorous atom Chemical group 0.000 description 3
- 229920000573 polyethylene Polymers 0.000 description 3
- 239000011148 porous material Substances 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 3
- 239000002002 slurry Substances 0.000 description 3
- DHKHKXVYLBGOIT-UHFFFAOYSA-N 1,1-Diethoxyethane Chemical compound CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 description 2
- ZFPGARUNNKGOBB-UHFFFAOYSA-N 1-Ethyl-2-pyrrolidinone Chemical compound CCN1CCCC1=O ZFPGARUNNKGOBB-UHFFFAOYSA-N 0.000 description 2
- ZDQGCACOETVASG-UHFFFAOYSA-N 1-[methoxy(methyl)phosphoryl]butane Chemical compound CCCCP(C)(=O)OC ZDQGCACOETVASG-UHFFFAOYSA-N 0.000 description 2
- GXYBSEVPLCHEBL-UHFFFAOYSA-N 1-dibutylphosphoryloxybutane Chemical compound CCCCOP(=O)(CCCC)CCCC GXYBSEVPLCHEBL-UHFFFAOYSA-N 0.000 description 2
- 229920000049 Carbon (fiber) Polymers 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- 239000001856 Ethyl cellulose Substances 0.000 description 2
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 2
- 229910000733 Li alloy Inorganic materials 0.000 description 2
- 229910013063 LiBF 4 Inorganic materials 0.000 description 2
- 229910012851 LiCoO 2 Inorganic materials 0.000 description 2
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 2
- 239000002033 PVDF binder Substances 0.000 description 2
- 239000005062 Polybutadiene Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- 239000004917 carbon fiber Substances 0.000 description 2
- CKFRRHLHAJZIIN-UHFFFAOYSA-N cobalt lithium Chemical compound [Li].[Co] CKFRRHLHAJZIIN-UHFFFAOYSA-N 0.000 description 2
- 150000004292 cyclic ethers Chemical class 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- TXCDCPKCNAJMEE-UHFFFAOYSA-N dibenzofuran Chemical compound C1=CC=C2C3=CC=CC=C3OC2=C1 TXCDCPKCNAJMEE-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 229920001249 ethyl cellulose Polymers 0.000 description 2
- 235000019325 ethyl cellulose Nutrition 0.000 description 2
- JBTWLSYIZRCDFO-UHFFFAOYSA-N ethyl methyl carbonate Chemical compound CCOC(=O)OC JBTWLSYIZRCDFO-UHFFFAOYSA-N 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011888 foil Substances 0.000 description 2
- GAEKPEKOJKCEMS-UHFFFAOYSA-N gamma-valerolactone Chemical compound CC1CCC(=O)O1 GAEKPEKOJKCEMS-UHFFFAOYSA-N 0.000 description 2
- 229910002804 graphite Inorganic materials 0.000 description 2
- 239000010439 graphite Substances 0.000 description 2
- 239000003112 inhibitor Substances 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000001989 lithium alloy Substances 0.000 description 2
- 229910001416 lithium ion Inorganic materials 0.000 description 2
- RSNHXDVSISOZOB-UHFFFAOYSA-N lithium nickel Chemical compound [Li].[Ni] RSNHXDVSISOZOB-UHFFFAOYSA-N 0.000 description 2
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 2
- 229910052748 manganese Inorganic materials 0.000 description 2
- 239000011572 manganese Substances 0.000 description 2
- 229910044991 metal oxide Inorganic materials 0.000 description 2
- 150000004706 metal oxides Chemical class 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 2
- 229920002857 polybutadiene Polymers 0.000 description 2
- 229920000098 polyolefin Polymers 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 2
- 239000004810 polytetrafluoroethylene Substances 0.000 description 2
- 238000005096 rolling process Methods 0.000 description 2
- 238000007788 roughening Methods 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000007784 solid electrolyte Substances 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 239000002562 thickening agent Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910000314 transition metal oxide Inorganic materials 0.000 description 2
- 150000003624 transition metals Chemical class 0.000 description 2
- ZZXUZKXVROWEIF-UHFFFAOYSA-N 1,2-butylene carbonate Chemical compound CCC1COC(=O)O1 ZZXUZKXVROWEIF-UHFFFAOYSA-N 0.000 description 1
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- VXNZUUAINFGPBY-UHFFFAOYSA-N 1-Butene Chemical group CCC=C VXNZUUAINFGPBY-UHFFFAOYSA-N 0.000 description 1
- RVUOXIOFERPQKI-UHFFFAOYSA-N 1-[butoxy(ethyl)phosphoryl]-2-methylbenzene Chemical compound CCCCOP(=O)(CC)C1=CC=CC=C1C RVUOXIOFERPQKI-UHFFFAOYSA-N 0.000 description 1
- SJHSWOQWNMMYQS-UHFFFAOYSA-N 1-[butoxy(methyl)phosphoryl]-2-methylbenzene Chemical compound CCCCOP(C)(=O)C1=CC=CC=C1C SJHSWOQWNMMYQS-UHFFFAOYSA-N 0.000 description 1
- IUBOLKFNKFMXRW-UHFFFAOYSA-N 1-[butoxy(methyl)phosphoryl]-3-methylbenzene Chemical compound CCCCOP(C)(=O)C1=CC=CC(C)=C1 IUBOLKFNKFMXRW-UHFFFAOYSA-N 0.000 description 1
- PSRZEGPJLIHHDG-UHFFFAOYSA-N 1-[butoxy(methyl)phosphoryl]-4-methylbenzene Chemical compound CCCCOP(C)(=O)C1=CC=C(C)C=C1 PSRZEGPJLIHHDG-UHFFFAOYSA-N 0.000 description 1
- RMTNXDZSBANZQR-UHFFFAOYSA-N 1-[butyl(ethoxy)phosphoryl]-2-methylbenzene Chemical compound CCCCP(=O)(OCC)C1=CC=CC=C1C RMTNXDZSBANZQR-UHFFFAOYSA-N 0.000 description 1
- RINNVHICTDEKBQ-UHFFFAOYSA-N 1-[butyl(ethoxy)phosphoryl]-3-methylbenzene Chemical compound CCCCP(=O)(OCC)C1=CC=CC(C)=C1 RINNVHICTDEKBQ-UHFFFAOYSA-N 0.000 description 1
- RUXZZVVTYSXYQL-UHFFFAOYSA-N 1-[butyl(ethoxy)phosphoryl]-4-methylbenzene Chemical compound CCCCP(=O)(OCC)C1=CC=C(C)C=C1 RUXZZVVTYSXYQL-UHFFFAOYSA-N 0.000 description 1
- ODVIGXMRBJCIPC-UHFFFAOYSA-N 1-[butyl(ethoxy)phosphoryl]butane Chemical compound CCCCP(=O)(OCC)CCCC ODVIGXMRBJCIPC-UHFFFAOYSA-N 0.000 description 1
- LGRUKLULQRFQOO-UHFFFAOYSA-N 1-[butyl(ethyl)phosphoryl]oxybutane Chemical compound CCCCOP(=O)(CC)CCCC LGRUKLULQRFQOO-UHFFFAOYSA-N 0.000 description 1
- XZMHWSPAEYUCGW-UHFFFAOYSA-N 1-[butyl(methoxy)phosphoryl]-3-methylbenzene Chemical compound CCCCP(=O)(OC)C1=CC=CC(C)=C1 XZMHWSPAEYUCGW-UHFFFAOYSA-N 0.000 description 1
- NZJPBCATHTXZGV-UHFFFAOYSA-N 1-[butyl(methoxy)phosphoryl]-4-methylbenzene Chemical compound CCCCP(=O)(OC)C1=CC=C(C)C=C1 NZJPBCATHTXZGV-UHFFFAOYSA-N 0.000 description 1
- VKIVIBIXAUGGQD-UHFFFAOYSA-N 1-[butyl(methyl)phosphoryl]oxy-4-methylbenzene Chemical compound CCCCP(C)(=O)OC1=CC=C(C)C=C1 VKIVIBIXAUGGQD-UHFFFAOYSA-N 0.000 description 1
- KFAKYMRJZDBKBS-UHFFFAOYSA-N 1-[butyl(propoxy)phosphoryl]-2-methylbenzene Chemical compound CCCCP(=O)(OCCC)C1=CC=CC=C1C KFAKYMRJZDBKBS-UHFFFAOYSA-N 0.000 description 1
- KLHHMDKSFKJQIW-UHFFFAOYSA-N 1-[butyl(propoxy)phosphoryl]-3-methylbenzene Chemical compound CCCCP(=O)(OCCC)C1=CC=CC(C)=C1 KLHHMDKSFKJQIW-UHFFFAOYSA-N 0.000 description 1
- OHRFAAFZUZBCFJ-UHFFFAOYSA-N 1-[butyl(propoxy)phosphoryl]-4-methylbenzene Chemical compound CCCCP(=O)(OCCC)C1=CC=C(C)C=C1 OHRFAAFZUZBCFJ-UHFFFAOYSA-N 0.000 description 1
- UEVNGWWHQPOYKR-UHFFFAOYSA-N 1-[ethoxy(1,1,2,2,2-pentafluoroethyl)phosphoryl]-1,1,2,2,2-pentafluoroethane Chemical compound CCOP(=O)(C(F)(F)C(F)(F)F)C(F)(F)C(F)(F)F UEVNGWWHQPOYKR-UHFFFAOYSA-N 0.000 description 1
- NMISTYFFOZZWSK-UHFFFAOYSA-N 1-[ethoxy(ethyl)phosphoryl]-2-methylbenzene Chemical compound CCOP(=O)(CC)C1=CC=CC=C1C NMISTYFFOZZWSK-UHFFFAOYSA-N 0.000 description 1
- IERUGLRSAWFSIL-UHFFFAOYSA-N 1-[ethoxy(ethyl)phosphoryl]-4-methylbenzene Chemical compound CCOP(=O)(CC)C1=CC=C(C)C=C1 IERUGLRSAWFSIL-UHFFFAOYSA-N 0.000 description 1
- BRHNRWZXZNHORR-UHFFFAOYSA-N 1-[ethoxy(methyl)phosphoryl]-4-methylbenzene Chemical compound CCOP(C)(=O)C1=CC=C(C)C=C1 BRHNRWZXZNHORR-UHFFFAOYSA-N 0.000 description 1
- SHZDZMARTASDIB-UHFFFAOYSA-N 1-[ethoxy(propyl)phosphoryl]-2-methylbenzene Chemical compound CCCP(=O)(OCC)C1=CC=CC=C1C SHZDZMARTASDIB-UHFFFAOYSA-N 0.000 description 1
- JMRGUAHWXVNUKK-UHFFFAOYSA-N 1-[ethoxy(propyl)phosphoryl]-3-methylbenzene Chemical compound CCCP(=O)(OCC)C1=CC=CC(C)=C1 JMRGUAHWXVNUKK-UHFFFAOYSA-N 0.000 description 1
- BYWHPYCAGISUIO-UHFFFAOYSA-N 1-[ethoxy(propyl)phosphoryl]-4-methylbenzene Chemical compound CCCP(=O)(OCC)C1=CC=C(C)C=C1 BYWHPYCAGISUIO-UHFFFAOYSA-N 0.000 description 1
- ZKTRSTAMNOXXKB-UHFFFAOYSA-N 1-[ethoxy(propyl)phosphoryl]butane Chemical compound CCCCP(=O)(CCC)OCC ZKTRSTAMNOXXKB-UHFFFAOYSA-N 0.000 description 1
- JBSAFMFBKXGWLF-UHFFFAOYSA-N 1-[ethoxy(propyl)phosphoryl]propane Chemical compound CCCP(=O)(CCC)OCC JBSAFMFBKXGWLF-UHFFFAOYSA-N 0.000 description 1
- NJWMEQMQTLLSJI-UHFFFAOYSA-N 1-[ethyl(methoxy)phosphoryl]-3-methylbenzene Chemical compound CCP(=O)(OC)C1=CC=CC(C)=C1 NJWMEQMQTLLSJI-UHFFFAOYSA-N 0.000 description 1
- FAKIKBRVPHOVNL-UHFFFAOYSA-N 1-[ethyl(methoxy)phosphoryl]-4-methylbenzene Chemical compound CCP(=O)(OC)C1=CC=C(C)C=C1 FAKIKBRVPHOVNL-UHFFFAOYSA-N 0.000 description 1
- WCOICKPRQHKXDC-UHFFFAOYSA-N 1-[ethyl(methoxy)phosphoryl]ethane Chemical compound CCP(=O)(CC)OC WCOICKPRQHKXDC-UHFFFAOYSA-N 0.000 description 1
- FOZBFBCNBDUALX-UHFFFAOYSA-N 1-[ethyl(methyl)phosphoryl]oxypropane Chemical compound CCCOP(C)(=O)CC FOZBFBCNBDUALX-UHFFFAOYSA-N 0.000 description 1
- YLAIMFPNABZNFR-UHFFFAOYSA-N 1-[ethyl(propoxy)phosphoryl]-4-methylbenzene Chemical compound CCCOP(=O)(CC)C1=CC=C(C)C=C1 YLAIMFPNABZNFR-UHFFFAOYSA-N 0.000 description 1
- LXLMKGCCGGLTCE-UHFFFAOYSA-N 1-[ethyl(propyl)phosphoryl]oxy-4-methylbenzene Chemical compound CCCP(=O)(CC)OC1=CC=C(C)C=C1 LXLMKGCCGGLTCE-UHFFFAOYSA-N 0.000 description 1
- VDVKRNWRLVTLBU-UHFFFAOYSA-N 1-[ethyl(propyl)phosphoryl]oxybutane Chemical compound CCCCOP(=O)(CC)CCC VDVKRNWRLVTLBU-UHFFFAOYSA-N 0.000 description 1
- WLPLOBZCNWXZSW-UHFFFAOYSA-N 1-[methoxy(methyl)phosphoryl]-3-methylbenzene Chemical compound COP(C)(=O)C1=CC=CC(C)=C1 WLPLOBZCNWXZSW-UHFFFAOYSA-N 0.000 description 1
- PFCIVFOLWWVGEJ-UHFFFAOYSA-N 1-[methoxy(methyl)phosphoryl]-4-methylbenzene Chemical compound COP(C)(=O)C1=CC=C(C)C=C1 PFCIVFOLWWVGEJ-UHFFFAOYSA-N 0.000 description 1
- VZUYQRUVYFGLAJ-UHFFFAOYSA-N 1-[methoxy(propyl)phosphoryl]-3-methylbenzene Chemical compound CCCP(=O)(OC)C1=CC=CC(C)=C1 VZUYQRUVYFGLAJ-UHFFFAOYSA-N 0.000 description 1
- PDMILKUHBLCNHG-UHFFFAOYSA-N 1-[methoxy(propyl)phosphoryl]-4-methylbenzene Chemical compound CCCP(=O)(OC)C1=CC=C(C)C=C1 PDMILKUHBLCNHG-UHFFFAOYSA-N 0.000 description 1
- LVKKKPSMZPWJSH-UHFFFAOYSA-N 1-[methyl(propoxy)phosphoryl]butane Chemical compound CCCCP(C)(=O)OCCC LVKKKPSMZPWJSH-UHFFFAOYSA-N 0.000 description 1
- VFCKAHVFAABONR-UHFFFAOYSA-N 1-[methyl(propyl)phosphoryl]oxypropane Chemical compound CCCOP(C)(=O)CCC VFCKAHVFAABONR-UHFFFAOYSA-N 0.000 description 1
- FADVPDGMBXTDDD-UHFFFAOYSA-N 1-dimethylphosphoryloxypropane Chemical compound CCCOP(C)(C)=O FADVPDGMBXTDDD-UHFFFAOYSA-N 0.000 description 1
- MGJKGKKAUKXOPR-UHFFFAOYSA-N 1-dipropylphosphoryloxybutane Chemical compound CCCCOP(=O)(CCC)CCC MGJKGKKAUKXOPR-UHFFFAOYSA-N 0.000 description 1
- VOSWHBYTDCCDFU-UHFFFAOYSA-N 1-methyl-2-[methyl(propoxy)phosphoryl]benzene Chemical compound CCCOP(C)(=O)C1=CC=CC=C1C VOSWHBYTDCCDFU-UHFFFAOYSA-N 0.000 description 1
- VLBZVVBNQWWQHY-UHFFFAOYSA-N 1-methyl-2-[propoxy(propyl)phosphoryl]benzene Chemical compound CCCOP(=O)(CCC)C1=CC=CC=C1C VLBZVVBNQWWQHY-UHFFFAOYSA-N 0.000 description 1
- KBYOYCSTWSELEK-UHFFFAOYSA-N 1-methyl-3-[methyl(propoxy)phosphoryl]benzene Chemical compound CCCOP(C)(=O)C1=CC=CC(C)=C1 KBYOYCSTWSELEK-UHFFFAOYSA-N 0.000 description 1
- WHCNJUWIFYMRQP-UHFFFAOYSA-N 1-methyl-3-[propoxy(propyl)phosphoryl]benzene Chemical compound CCCOP(=O)(CCC)C1=CC=CC(C)=C1 WHCNJUWIFYMRQP-UHFFFAOYSA-N 0.000 description 1
- NRRPZBZXLBRRDW-UHFFFAOYSA-N 1-methyl-4-[methyl(propyl)phosphoryl]oxybenzene Chemical compound CCCP(C)(=O)OC1=CC=C(C)C=C1 NRRPZBZXLBRRDW-UHFFFAOYSA-N 0.000 description 1
- QVUQFZSDTHLBHH-UHFFFAOYSA-N 1-methyl-4-[propoxy(propyl)phosphoryl]benzene Chemical compound CCCOP(=O)(CCC)C1=CC=C(C)C=C1 QVUQFZSDTHLBHH-UHFFFAOYSA-N 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000004215 2,4-difluorophenyl group Chemical group [H]C1=C([H])C(*)=C(F)C([H])=C1F 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- JWUJQDFVADABEY-UHFFFAOYSA-N 2-methyltetrahydrofuran Chemical compound CC1CCCO1 JWUJQDFVADABEY-UHFFFAOYSA-N 0.000 description 1
- 125000004211 3,5-difluorophenyl group Chemical group [H]C1=C(F)C([H])=C(*)C([H])=C1F 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- BJWMSGRKJIOCNR-UHFFFAOYSA-N 4-ethenyl-1,3-dioxolan-2-one Chemical compound C=CC1COC(=O)O1 BJWMSGRKJIOCNR-UHFFFAOYSA-N 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 229910000851 Alloy steel Inorganic materials 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- ZMDXDJDSBKKJGO-UHFFFAOYSA-N CCCP(C1=CC=C(C=C1)C)(O)(O)O Chemical compound CCCP(C1=CC=C(C=C1)C)(O)(O)O ZMDXDJDSBKKJGO-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 239000004354 Hydroxyethyl cellulose Substances 0.000 description 1
- 229920000663 Hydroxyethyl cellulose Polymers 0.000 description 1
- 229910015015 LiAsF 6 Inorganic materials 0.000 description 1
- 229910013375 LiC Inorganic materials 0.000 description 1
- 229910015645 LiMn Inorganic materials 0.000 description 1
- 229910014689 LiMnO Inorganic materials 0.000 description 1
- 229910013528 LiN(SO2 CF3)2 Inorganic materials 0.000 description 1
- 229910013385 LiN(SO2C2F5)2 Inorganic materials 0.000 description 1
- 229910013290 LiNiO 2 Inorganic materials 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- RJUFJBKOKNCXHH-UHFFFAOYSA-N Methyl propionate Chemical compound CCC(=O)OC RJUFJBKOKNCXHH-UHFFFAOYSA-N 0.000 description 1
- 239000000020 Nitrocellulose Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 229910004298 SiO 2 Inorganic materials 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- DHXVGJBLRPWPCS-UHFFFAOYSA-N Tetrahydropyran Chemical compound C1CCOCC1 DHXVGJBLRPWPCS-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- PFYQFCKUASLJLL-UHFFFAOYSA-N [Co].[Ni].[Li] Chemical compound [Co].[Ni].[Li] PFYQFCKUASLJLL-UHFFFAOYSA-N 0.000 description 1
- KLARSDUHONHPRF-UHFFFAOYSA-N [Li].[Mn] Chemical compound [Li].[Mn] KLARSDUHONHPRF-UHFFFAOYSA-N 0.000 description 1
- WANYMFQTJWGDFB-UHFFFAOYSA-N [butoxy(butyl)phosphoryl]benzene Chemical compound CCCCOP(=O)(CCCC)C1=CC=CC=C1 WANYMFQTJWGDFB-UHFFFAOYSA-N 0.000 description 1
- ADQFXAGCAHPEHF-UHFFFAOYSA-N [butoxy(ethyl)phosphoryl]benzene Chemical compound CCCCOP(=O)(CC)C1=CC=CC=C1 ADQFXAGCAHPEHF-UHFFFAOYSA-N 0.000 description 1
- DZYAJVAXSAJHDU-UHFFFAOYSA-N [butoxy(phenyl)phosphoryl]benzene Chemical compound C=1C=CC=CC=1P(=O)(OCCCC)C1=CC=CC=C1 DZYAJVAXSAJHDU-UHFFFAOYSA-N 0.000 description 1
- ZMAKVZAHNZETQQ-UHFFFAOYSA-N [butoxy(propyl)phosphoryl]benzene Chemical compound CCCCOP(=O)(CCC)C1=CC=CC=C1 ZMAKVZAHNZETQQ-UHFFFAOYSA-N 0.000 description 1
- DQIPZODSGIASGM-UHFFFAOYSA-N [butyl(ethoxy)phosphoryl]benzene Chemical compound CCCCP(=O)(OCC)C1=CC=CC=C1 DQIPZODSGIASGM-UHFFFAOYSA-N 0.000 description 1
- IVYBZVTUYBUPLE-UHFFFAOYSA-N [butyl(propoxy)phosphoryl]benzene Chemical compound CCCCP(=O)(OCCC)C1=CC=CC=C1 IVYBZVTUYBUPLE-UHFFFAOYSA-N 0.000 description 1
- BHYBJNSQCWMPAJ-UHFFFAOYSA-N [ethoxy(ethyl)phosphoryl]benzene Chemical compound CCOP(=O)(CC)C1=CC=CC=C1 BHYBJNSQCWMPAJ-UHFFFAOYSA-N 0.000 description 1
- QRJASDLTCXIYRK-UHFFFAOYSA-N [ethoxy(phenyl)phosphoryl]benzene Chemical compound C=1C=CC=CC=1P(=O)(OCC)C1=CC=CC=C1 QRJASDLTCXIYRK-UHFFFAOYSA-N 0.000 description 1
- ULMPOWHVOLFVFQ-UHFFFAOYSA-N [ethyl(propoxy)phosphoryl]benzene Chemical compound CCCOP(=O)(CC)C1=CC=CC=C1 ULMPOWHVOLFVFQ-UHFFFAOYSA-N 0.000 description 1
- BHUMZHDFNOXAMC-UHFFFAOYSA-N [methoxy(phenyl)phosphoryl]benzene Chemical compound C=1C=CC=CC=1P(=O)(OC)C1=CC=CC=C1 BHUMZHDFNOXAMC-UHFFFAOYSA-N 0.000 description 1
- WHXLOUWSAUQRIS-UHFFFAOYSA-N [methyl(propoxy)phosphoryl]benzene Chemical compound CCCOP(C)(=O)C1=CC=CC=C1 WHXLOUWSAUQRIS-UHFFFAOYSA-N 0.000 description 1
- KPOPYLQJRSZHIK-UHFFFAOYSA-N [phenyl(propoxy)phosphoryl]benzene Chemical compound C=1C=CC=CC=1P(=O)(OCCC)C1=CC=CC=C1 KPOPYLQJRSZHIK-UHFFFAOYSA-N 0.000 description 1
- DWZSHEVXEREINB-UHFFFAOYSA-N [propoxy(propyl)phosphoryl]benzene Chemical compound CCCOP(=O)(CCC)C1=CC=CC=C1 DWZSHEVXEREINB-UHFFFAOYSA-N 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N acetic acid trimethyl ester Natural products COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- 239000006230 acetylene black Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 229910003481 amorphous carbon Inorganic materials 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 229910021383 artificial graphite Inorganic materials 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 239000012752 auxiliary agent Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 238000005422 blasting Methods 0.000 description 1
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- BNMJSBUIDQYHIN-UHFFFAOYSA-L butyl phosphate Chemical compound CCCCOP([O-])([O-])=O BNMJSBUIDQYHIN-UHFFFAOYSA-L 0.000 description 1
- CPRXHCAKYVTPFP-UHFFFAOYSA-N butyl(ethyl)phosphane Chemical compound CCCCPCC CPRXHCAKYVTPFP-UHFFFAOYSA-N 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 239000006229 carbon black Substances 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 229920003064 carboxyethyl cellulose Polymers 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 238000000748 compression moulding Methods 0.000 description 1
- 239000011889 copper foil Substances 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- UOUBSNKDIDEUCV-UHFFFAOYSA-N dimethylphosphoryloxybenzene Chemical compound CP(C)(=O)OC1=CC=CC=C1 UOUBSNKDIDEUCV-UHFFFAOYSA-N 0.000 description 1
- HAXBLJDZJKJLHZ-UHFFFAOYSA-N dimethylphosphoryloxymethane Chemical compound COP(C)(C)=O HAXBLJDZJKJLHZ-UHFFFAOYSA-N 0.000 description 1
- 238000005868 electrolysis reaction Methods 0.000 description 1
- 229910001651 emery Inorganic materials 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 150000004673 fluoride salts Chemical class 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 125000003784 fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 239000006232 furnace black Substances 0.000 description 1
- 239000003349 gelling agent Substances 0.000 description 1
- 230000020169 heat generation Effects 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 235000019447 hydroxyethyl cellulose Nutrition 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910001506 inorganic fluoride Inorganic materials 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 229920003049 isoprene rubber Polymers 0.000 description 1
- 229910000625 lithium cobalt oxide Inorganic materials 0.000 description 1
- BFZPBUKRYWOWDV-UHFFFAOYSA-N lithium;oxido(oxo)cobalt Chemical compound [Li+].[O-][Co]=O BFZPBUKRYWOWDV-UHFFFAOYSA-N 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 229940017219 methyl propionate Drugs 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229910021382 natural graphite Inorganic materials 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229920001220 nitrocellulos Polymers 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000001254 oxidized starch Substances 0.000 description 1
- 235000013808 oxidized starch Nutrition 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 125000004817 pentamethylene group Chemical group [H]C([H])([*:2])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[*:1] 0.000 description 1
- 239000005011 phenolic resin Substances 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- ACVYVLVWPXVTIT-UHFFFAOYSA-M phosphinate Chemical compound [O-][PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-M 0.000 description 1
- 150000003008 phosphonic acid esters Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000011295 pitch Substances 0.000 description 1
- 229920001483 poly(ethyl methacrylate) polymer Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920002689 polyvinyl acetate Polymers 0.000 description 1
- 239000011118 polyvinyl acetate Substances 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000004321 preservation Methods 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000012779 reinforcing material Substances 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- RIUWBIIVUYSTCN-UHFFFAOYSA-N trilithium borate Chemical compound [Li+].[Li+].[Li+].[O-]B([O-])[O-] RIUWBIIVUYSTCN-UHFFFAOYSA-N 0.000 description 1
- 229910052720 vanadium Inorganic materials 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Abstract
Description
本発明は、二次電池用非水系電解液及びそれを用いる非水系電解液二次電池に関する。詳しくは、高温連続充電時および高温保存を行っても劣化が少ない、信頼性の高い非水系電解液二次電池及びそれを提供するための二次電池用非水系電解液に関する。 The present invention relates to a non-aqueous electrolyte for a secondary battery and a non-aqueous electrolyte secondary battery using the same. More specifically, the present invention relates to a highly reliable non-aqueous electrolyte secondary battery that undergoes little deterioration during high-temperature continuous charging and high-temperature storage, and a non-aqueous electrolyte for a secondary battery for providing the same.
近年、電気製品の軽量化、小型化にともない、高いエネルギー密度を持つリチウム二次電池が注目されている。
リチウム二次電池用の電解液は、リチウム塩などの溶質と、有機溶媒とからなる。有機溶媒は、高い誘電率を有すること、酸化電位が高いこと、及び電池中で安定であることが要求される。これらの要求を一つの溶媒で達成するのは困難なので、リチウム二次電池の電解液の有機溶媒としては、例えば炭酸エチレン、炭酸プロピレン等の環状炭酸エステル類またはγ−ブチロラクトン等の環状カルボン酸エステル類などの高誘電率溶媒と、炭酸ジエチル、炭酸ジメチル等の鎖状炭酸エステル類またはジメトキシエタン等のエーテル類などの低粘度溶媒を組み合わせて使用している。
また、初期容量、サイクル特性、高温保存特性、連続充電特性などを改良するために、種々の化合物を電解液に含有させることが提案されている。
2. Description of the Related Art In recent years, lithium secondary batteries having a high energy density have been attracting attention as electric appliances become lighter and smaller.
An electrolyte for a lithium secondary battery includes a solute such as a lithium salt and an organic solvent. The organic solvent is required to have a high dielectric constant, a high oxidation potential, and stability in a battery. Since it is difficult to achieve these requirements with a single solvent, examples of the organic solvent for the electrolyte of the lithium secondary battery include cyclic carbonates such as ethylene carbonate and propylene carbonate and cyclic carboxylate esters such as γ-butyrolactone. And a low-viscosity solvent such as a chain carbonate such as diethyl carbonate or dimethyl carbonate or an ether such as dimethoxyethane.
Further, in order to improve the initial capacity, cycle characteristics, high-temperature storage characteristics, continuous charging characteristics, and the like, it has been proposed to include various compounds in the electrolyte.
例えば、連続充電特性を向上させる方法として、特許文献1には、電解液中にリン酸エステルを含有させることが記載されている。 For example, as a method for improving continuous charging characteristics, Patent Document 1 describes that a phosphate ester is contained in an electrolytic solution.
また、特許文献2及び特許文献3には、特定のホスホン酸エステル、ホスフィン酸エステルを有機溶媒中に5〜100重量%含有させることにより、電池の性能に悪影響を及ぼすことなく電解液に難燃性を持たせることが記載されている。そして、実施例によれば、炭酸エステル類あるいは鎖状エーテルとこれらのリン酸化合物とを重量比で2:1又は1:1で混合した有機溶媒に、LiPF6を溶解してなる電解液を用いた二次電池は、10
0サイクル目の容量維持率が数%〜十数%の低下に止まることが示されているが、電池の高温特性に関しては記載がない。
Although it is shown that the capacity maintenance ratio at the 0th cycle is reduced only to several percent to several tens of percent, there is no description about the high temperature characteristics of the battery.
リチウム二次電池がノートパソコンや携帯電話などの携帯機器に適用されることが急速に拡大するのに伴い、高性能化への要求は高まっている。特に高温連続充電特性、高温保存特性などの高温時の電池特性の改良である。
例えば、ノートパソコンは、ほとんどの場合、ACアダプターを介して電源に接続した状態で使用されており、使用中も、パソコン中の二次電池は絶えず充電されている。このような連続充電状態では、本体の発熱の影響もあって、電解液の分解が起こり、電池性能が著しく低下するという問題がある。また、電解液の分解は、多くの場合にガスの発生を伴うが、ガスの発生量が多い場合には、電池の変形や破裂が起こり、電池自体が使用不能になるという問題もある。
With the rapid expansion of the application of lithium secondary batteries to portable devices such as notebook computers and mobile phones, demands for higher performance are increasing. In particular, it is an improvement in battery characteristics at high temperatures such as high-temperature continuous charging characteristics and high-temperature storage characteristics.
For example, a notebook computer is almost always used while connected to a power supply via an AC adapter, and the secondary battery in the personal computer is constantly charged even during use. In such a continuous charging state, there is a problem that the electrolytic solution is decomposed due to the influence of heat generation of the main body, and the battery performance is significantly reduced. Decomposition of the electrolytic solution often involves the generation of gas, but when the amount of generated gas is large, there is a problem that the battery is deformed or ruptured, and the battery itself becomes unusable.
また、これらの携帯機器は、日中の自動車内など高温下に放置されることがある。この場合にも、二次電池は高温にさらされることになり、電解液の分解による電池特性の低下や、ガスの発生による電池缶の変形・破裂が起こるという問題がある。
従って、本発明は、高温連続充電時、高温保存時における分解が抑えられた電解液、およびこれを用いた高温特性に優れた二次電池の提供を目的とする。
In addition, these portable devices may be left at high temperatures such as in a car during the day. Also in this case, the secondary battery is exposed to a high temperature, and there is a problem that the battery characteristics deteriorate due to the decomposition of the electrolytic solution, and the battery can is deformed or ruptured due to generation of gas.
Accordingly, an object of the present invention is to provide an electrolytic solution in which decomposition during high-temperature continuous charging and high-temperature storage is suppressed, and a secondary battery using the electrolytic solution and having excellent high-temperature characteristics.
本発明者らは上記課題を解決すべく鋭意検討した結果、一般式(1)で表されるホスフィン酸エステルを非水系電解液中に特定の濃度で含有させることにより、高温連続充電特性や、高温保存特性が著しく改善されることを見出し、本発明を完成した。 The present inventors have conducted intensive studies to solve the above-mentioned problems, and as a result, by including the phosphinic ester represented by the general formula (1) at a specific concentration in the non-aqueous electrolytic solution, the high-temperature continuous charging characteristics, The present inventors have found that the high-temperature storage characteristics are significantly improved, and have completed the present invention.
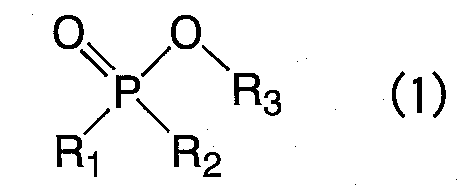
即ち、本発明の要旨は、溶質、下記一般式(1)で表される化合物及びこれらを溶解する非水系有機溶媒を含有する非水系電解液であって、下記一般式(1)で表される化合物の含有量が、非水系電解液の全重量に対して、0.01重量%以上、4.5重量%以下であることを特徴とする二次電池用非水系電解液に存する。 That is, the gist of the present invention is a non-aqueous electrolytic solution containing a solute, a compound represented by the following general formula (1) and a non-aqueous organic solvent for dissolving them, and represented by the following general formula (1). Wherein the content of the compound is 0.01% by weight or more and 4.5% by weight or less with respect to the total weight of the non-aqueous electrolyte.
(式中R1〜R3は、各々独立して、(i)ハロゲン原子で置換されていても良い炭素数1〜8の鎖状もしくは環状アルキル基、(ii)ハロゲン原子で置換されていても良いフェニル基、(iii)炭素数1〜4のアルキル基で置換されていても良いフェニル基および(iv)ハロゲン原子及び炭素数1〜4のアルキル基で置換されていても良いフェニル基からなる群から選ばれるいずれかを表す。なお、R1とR2又はR2とR3がいずれもアルキル基である場合には、互いに結合して環構造を形成していても良い。)
また、本発明の別の要旨は、リチウムを吸蔵・放出可能な負極及び正極、上記二次電池用非水電解液からなることを特徴とする非水系電解液二次電池に存する。
(Wherein R 1 to R 3 each independently represent (i) a chain or cyclic alkyl group having 1 to 8 carbon atoms which may be substituted with a halogen atom, (ii) (Iii) a phenyl group optionally substituted with an alkyl group having 1 to 4 carbon atoms and (iv) a phenyl group optionally substituted with a halogen atom and an alkyl group having 1 to 4 carbon atoms. And when R 1 and R 2 or R 2 and R 3 are both alkyl groups, they may be bonded to each other to form a ring structure.)
Another aspect of the present invention resides in a nonaqueous electrolyte secondary battery including a negative electrode and a positive electrode capable of inserting and extracting lithium, and the above-described nonaqueous electrolyte for a secondary battery.
本発明によれば、高温保存時、高温連続充電時の電池性能劣化が抑制された電解液および二次電池を提供できる。 ADVANTAGE OF THE INVENTION According to this invention, the electrolyte solution and the secondary battery which suppressed the battery performance degradation at the time of high temperature preservation and high temperature continuous charge can be provided.
以下、本発明を詳細に説明する。
本発明に係る二次電池用非水系電解液の主成分は、常用の二次電池用非水系電解液と同じく、溶質およびこれを溶解する非水系有機溶媒である。
溶質としてはリチウム塩を用いる。リチウム塩としては、この用途に用い得ることができるものであれば特に制限はないが、例えば、以下のものが挙げられる。
1)無機リチウム塩:LiAsF6、LiPF6、LiBF4等の無機フッ化物塩、LiC
lO4、LiBrO4、LiIO4等の過ハロゲン酸塩。
2)有機リチウム塩:LiB(C6H5)4等の有機ホウ酸リチウム塩、LiCH3SO3等
のアルカンスルホン酸塩、LiN(SO2CF3)2、LiN(SO2C2F5)2等のパーフ
ルオロアルカンスルホン酸イミド塩、LiCF3SO3等のパーフルオロアルカンスルホン酸塩。
Hereinafter, the present invention will be described in detail.
The main components of the non-aqueous electrolytic solution for a secondary battery according to the present invention are a solute and a non-aqueous organic solvent that dissolves the solute, similarly to a conventional non-aqueous electrolytic solution for a secondary battery.
As a solute, a lithium salt is used. The lithium salt is not particularly limited as long as it can be used for this purpose, and examples thereof include the following.
1) Inorganic lithium salts: inorganic fluoride salts such as LiAsF 6 , LiPF 6 and LiBF 4 , LiC
lO 4, perhalogenate such LiBrO 4, LiIO 4.
2) Organic lithium salt: organic lithium borate such as LiB (C 6 H 5 ) 4 , alkane sulfonate such as LiCH 3 SO 3 , LiN (SO 2 CF 3 ) 2 , LiN (SO 2 C 2 F 5) 2 ) Perfluoroalkanesulfonic acid imide salts such as 2 and perfluoroalkanesulfonic acid salts such as LiCF 3 SO 3 .
なかでも好ましいのは、LiBF4及びLiPF6である。リチウム塩は、単独で用いても、2種以上を混合して用いてもよい。
非水系電解液中のリチウム塩の濃度は、通常0.5モル/リットル以上、好ましくは0
.75モル/リットル以上であり、通常2.5モル/リットル以下、好ましく1.5モル/リットル以下である。リチウム塩の濃度が高すぎても低すぎても電導度の低下が起き、電池特性が低下する恐れがある。
Among them, LiBF 4 and LiPF 6 are preferable. Lithium salts may be used alone or as a mixture of two or more.
The concentration of the lithium salt in the non-aqueous electrolyte is usually 0.5 mol / L or more, preferably 0 mol / L or more.
. It is at least 75 mol / l, usually at most 2.5 mol / l, preferably at most 1.5 mol / l. If the concentration of the lithium salt is too high or too low, the conductivity will decrease, and the battery characteristics may be degraded.
非水系有機溶媒としても、従来から非水系電解液の溶媒として提案されているものの中から適宜選択して用いることができる。例えば、環状カーボネート(環状炭酸エステル)類、鎖状カーボネート(鎖状炭酸エステル)類、環状エステル(環状カルボン酸エステル)類、鎖状エステル(鎖状カルボン酸エステル)類、環状エーテル類及び鎖状エーテル類等が挙げられる。 As the non-aqueous organic solvent, any solvent that has been conventionally proposed as a solvent for a non-aqueous electrolytic solution can be appropriately selected and used. For example, cyclic carbonate (cyclic carbonate), chain carbonate (chain carbonate), cyclic ester (cyclic carboxylate), chain ester (chain carboxylate), cyclic ether and chain Ethers and the like.
電解液の非水系有機溶媒としては、鎖状カーボネートと環状エステルからなる群から選ばれるものと環状カーボネートの混合溶媒が好ましい。
電解液の非水系有機溶媒に環状カーボネートが含まれる場合、好ましい比率は、5体積%から55体積%、さらに好ましくは15体積%から50体積%である。
電解液の非水系有機溶媒に鎖状カーボネートが含まれる場合、好ましい比率は、2体積%から85体積%、さらに好ましくは5体積%から85体積%である。
また、電解液の非水系有機溶媒に環状エステルが含まれる場合、好ましい比率は、40体積%から100体積%、さらに好ましくは50体積%から98体積%である。
As the nonaqueous organic solvent for the electrolytic solution, a mixed solvent of a solvent selected from the group consisting of a chain carbonate and a cyclic ester and a cyclic carbonate is preferable.
When the non-aqueous organic solvent of the electrolytic solution contains a cyclic carbonate, a preferable ratio is 5% by volume to 55% by volume, more preferably 15% by volume to 50% by volume.
When the non-aqueous organic solvent of the electrolytic solution contains a chain carbonate, a preferable ratio is 2% to 85% by volume, more preferably 5% to 85% by volume.
When the cyclic ester is contained in the non-aqueous organic solvent of the electrolytic solution, a preferable ratio is 40% by volume to 100% by volume, more preferably 50% by volume to 98% by volume.
好ましい有機溶媒の組合せとその体積比率としては、以下のものが挙げられる。
1.環状カーボネート+鎖状カーボネート(15〜40:60〜85)
2.環状カーボネート+環状エステル (20〜50:50〜80)
3.環状カーボネート+環状エステル+鎖状カーボネート (20〜50:50〜80:2〜20)
4.環状エステル+鎖状カーボネート (70〜98:2〜30)
5.環状エステル (単一溶媒)
Preferred combinations of organic solvents and their volume ratios include the following.
1. Cyclic carbonate + chain carbonate (15-40: 60-85)
2. Cyclic carbonate + Cyclic ester (20-50: 50-80)
3. Cyclic carbonate + Cyclic ester + Chain carbonate (20-50: 50-80: 2-20)
4. Cyclic ester + chain carbonate (70-98: 2-30)
5. Cyclic ester (single solvent)
環状カーボネート類としては、エチレンカーボネート、プロピレンカーボネート、ブチレンカーボネート等が挙げられる。鎖状カーボネート類としては、ジメチルカーボネート、ジエチルカーボネート、エチルメチルカーボネート等が挙げられる。環状エーテル類としては、テトラヒドロフラン、2−メチルテトラヒドロフラン、テトラヒドロピラン等が挙げられる。鎖状エーテル類としては、ジメトキシエタン、ジエトキシエタン等が挙げられる。環状エステル類としては、γ-ブチロラクトン、γ−バレロラクトン等が挙げられ
る。鎖状エステル類としては、酢酸メチル、プロピオン酸メチル等が挙げられる。
Examples of the cyclic carbonate include ethylene carbonate, propylene carbonate, butylene carbonate, and the like. Examples of the chain carbonates include dimethyl carbonate, diethyl carbonate, ethyl methyl carbonate and the like. Examples of the cyclic ethers include tetrahydrofuran, 2-methyltetrahydrofuran, tetrahydropyran and the like. Examples of chain ethers include dimethoxyethane, diethoxyethane, and the like. Examples of the cyclic esters include γ-butyrolactone, γ-valerolactone, and the like. Examples of the chain esters include methyl acetate and methyl propionate.
これらの非水系有機溶媒は、単独で用いても、2種以上を混合して用いてもよいが、通常、適切な物性が発現するように2種以上が混合して使用される。例えば、環状カーボネート類、鎖状カーボネート類、環状エステル類から選択した2種以上を混合した溶媒が挙げられる。特に好ましいのは、エチレンカーボネート、プロピレンカーボネート、ジメチルカーボネート、ジエチルカーボネート、エチルメチルカーボネート、γ−ブチロラクトン等から2種以上を混合したものである。 These non-aqueous organic solvents may be used alone or as a mixture of two or more, but usually, two or more are used in a mixture so as to exhibit appropriate physical properties. For example, a solvent in which two or more kinds selected from cyclic carbonates, chain carbonates, and cyclic esters are mixed is exemplified. Particularly preferred is a mixture of two or more of ethylene carbonate, propylene carbonate, dimethyl carbonate, diethyl carbonate, ethyl methyl carbonate, γ-butyrolactone, and the like.
本発明に係る非水系電解液は、上記の溶質と非水系有機溶媒を主成分とするが、これに更に下記一般式(1)で表される化合物を含有する。 The non-aqueous electrolyte according to the present invention contains the above-mentioned solute and non-aqueous organic solvent as main components, and further contains a compound represented by the following general formula (1).
式中、R1〜R3は、各々独立して、(i)ハロゲン原子で置換されていても良い炭素数1〜8の鎖状もしくは環状アルキル基、(ii)ハロゲン原子で置換されていても良いフェニル基、(iii)炭素数1〜4のアルキル基で置換されていても良いフェニル基および(iv)ハロゲン原子及び炭素数1〜4のアルキル基で置換されていても良いフェニル基からなる群から選ばれるいずれかを表す。なかでも好ましいのは、(i)ハロゲン原子で置換されていても良い炭素数1〜8の鎖状アルキル基、(ii)ハロゲン原子で置換されていても良いフェニル基、(iii)炭素数1〜4のアルキル基で置換されていても良いフェニル基および(iv)ハロゲン原子及び炭素数1〜4のアルキル基で置換されていても良いフェニル基から選ばれるいずれかである。
R1〜R3が表す置換基のいくつかを例示する。
ハロゲン原子で置換されていても良い鎖状アルキル基としては、メチル基、エチル基、n−プロピル基、イソプロピル基、n−ブチル基、イソブチル基、sec-ブチル基、tert-
ブチル基、n−ペンチル基、2-メチルブチル基、3-メチルブチル基、4-メチルブチル
基、2,2−ジメチルプロピル基、2,3-ジメチルプロピル基、3,3-ジメチルプロピル基、n−ヘキシル基、n−ヘプチル基、n−オクチル基、2-エチルヘキシル基、トリ
フルオロメチル基、2,2,2-トリフルオロエチル基、ペンタフルオロエチル基等が挙
げられる。なかでも好ましいのは、メチル基、エチル基、n−プロピル基、イソプロピル基、n−ブチル基、イソブチル基、sec-ブチル基、tert-ブチル基、トリフルオロメチル
基、2,2,2-トリフルオロエチル基、ペンタフルオロエチル基等のハロゲン原子で置
換されていても良い炭素数1〜4の鎖状アルキル基である。炭素数が1〜3であると更に好ましい。
In the formula, R 1 to R 3 each independently represent (i) a chain or cyclic alkyl group having 1 to 8 carbon atoms which may be substituted with a halogen atom, and (ii) a substituent substituted with a halogen atom. (Iii) a phenyl group optionally substituted with an alkyl group having 1 to 4 carbon atoms and (iv) a phenyl group optionally substituted with a halogen atom and an alkyl group having 1 to 4 carbon atoms. Represents one selected from the group consisting of: Among them, preferred are (i) a chain alkyl group having 1 to 8 carbon atoms which may be substituted with a halogen atom, (ii) a phenyl group which may be substituted with a halogen atom, and (iii) 1 carbon atom. And (iv) a phenyl group optionally substituted with a halogen atom and an alkyl group having 1 to 4 carbon atoms.
Some of the substituents represented by R 1 to R 3 are exemplified.
Examples of the chain alkyl group which may be substituted with a halogen atom include a methyl group, an ethyl group, an n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, a sec-butyl group and a tert-
Butyl group, n-pentyl group, 2-methylbutyl group, 3-methylbutyl group, 4-methylbutyl group, 2,2-dimethylpropyl group, 2,3-dimethylpropyl group, 3,3-dimethylpropyl group, n-hexyl Groups, n-heptyl group, n-octyl group, 2-ethylhexyl group, trifluoromethyl group, 2,2,2-trifluoroethyl group, pentafluoroethyl group and the like. Among them, preferred are a methyl group, an ethyl group, a n-propyl group, an isopropyl group, an n-butyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, a trifluoromethyl group, a 2,2,2-trimethyl group. A chain alkyl group having 1 to 4 carbon atoms which may be substituted with a halogen atom such as a fluoroethyl group and a pentafluoroethyl group. It is more preferable that the number of carbon atoms is 1 to 3.
ハロゲン原子で置換されていても良い環状アルキル基としては、シクロペンチル基、シクロヘキシル基、2−フルオロシクロヘキシル基、3−フルオロシクロヘキシル基、4−フルオロシクロヘキシル基等の炭素数4〜6、好ましくは炭素数5〜6のものが挙げられる。
ハロゲン原子で置換されていても良いフェニル基、炭素数1〜4のアルキル基で置換されていても良いフェニル基、ハロゲン原子及び炭素数1〜4のアルキル基で置換されていても良いフェニル基としては、フェニル基、2-フルオロフェニル基、3-フルオロフェニル基、4-フルオロフェニル基、2−トリル基、3−トリル基、4−トリル基、2,3−
ジフルオロフェニル基、2,4−ジフルオロフェニル基、2,5−ジフルオロフェニル基、2,6−ジフルオロフェニル基、3,4−ジフルオロフェニル基、3,5−ジフルオロフェニル基、4,5−ジフルオロフェニル基、2−フルオロ−3−トリル基、2−フルオロ−4−トリル基、2−フルオロ−5−トリル基、2−フルオロ−6−トリル基、3−フルオロ−2−トリル基、3−フルオロ−4−トリル基、3−フルオロ−5−トリル基、3−フルオロ−6−トリル基、4−フルオロ−2−トリル基、4−フルオロ−3−トリル基等が挙げられる。置換するアルキル基の炭素数が1〜2であると更に好ましい。なかでも好ましいのはフェニル基、2−トリル基、3−トリル基、4−トリル基である。
なお、アルキル基やフェニル基に置換するハロゲン原子としては、上述のようにフッ素原子が好ましいが、塩素原子、臭素原子、ヨウ素原子などであってもよい。
Examples of the cyclic alkyl group which may be substituted with a halogen atom include a cyclopentyl group, a cyclohexyl group, a 2-fluorocyclohexyl group, a 3-fluorocyclohexyl group, a 4-fluorocyclohexyl group and the like having 4 to 6 carbon atoms, preferably 5-6.
A phenyl group optionally substituted with a halogen atom, a phenyl group optionally substituted with an alkyl group having 1 to 4 carbon atoms, a phenyl group optionally substituted with a halogen atom and an alkyl group having 1 to 4 carbon atoms A phenyl group, a 2-fluorophenyl group, a 3-fluorophenyl group, a 4-fluorophenyl group, a 2-tolyl group, a 3-tolyl group, a 4-tolyl group, a 2,3-
Difluorophenyl group, 2,4-difluorophenyl group, 2,5-difluorophenyl group, 2,6-difluorophenyl group, 3,4-difluorophenyl group, 3,5-difluorophenyl group, 4,5-difluorophenyl Group, 2-fluoro-3-tolyl group, 2-fluoro-4-tolyl group, 2-fluoro-5-tolyl group, 2-fluoro-6-tolyl group, 3-fluoro-2-tolyl group, 3-fluoro -4-tolyl group, 3-fluoro-5-tolyl group, 3-fluoro-6-tolyl group, 4-fluoro-2-tolyl group, 4-fluoro-3-tolyl group and the like. More preferably, the alkyl group to be substituted has 1 to 2 carbon atoms. Of these, a phenyl group, a 2-tolyl group, a 3-tolyl group and a 4-tolyl group are preferred.
The halogen atom to be substituted for the alkyl group or the phenyl group is preferably a fluorine atom as described above, but may be a chlorine atom, a bromine atom, an iodine atom or the like.
R1とR2又はR2とR3がいずれもアルキル基の場合、これらは互いに結合して環構造を
形成していても良い。その具体例としては、R1とR2とが連結して、P原子を含む5〜6員環を形成している場合、すなわちP原子がn−ブチレン基の1位及び4位、又はn−ペンチレン基の1位及び5位と結合して環を形成している場合、及び、R2とR3とが連結して、P原子及びO原子を含む5〜6員環を形成する場合等が挙げられる。
When R 1 and R 2 or R 2 and R 3 are both alkyl groups, they may be bonded to each other to form a ring structure. As a specific example, when R 1 and R 2 are linked to form a 5- to 6-membered ring containing a P atom, that is, the P atom is in the 1- and 4-positions of the n-butylene group, or n A case where a ring is formed by bonding to the 1- and 5-positions of the pentylene group, and a case where R 2 and R 3 are linked to form a 5- to 6-membered ring containing a P atom and an O atom And the like.
一般式(1)で表される化合物の具体例としては、以下のものが挙げられる。
ジアルキルホスフィン酸メチル類:ジメチルホスフィン酸メチル、エチルメチルホスフィン酸メチル、メチル−n−プロピルホスフィン酸メチル、n−ブチルメチルホスフィン酸メチル、ジエチルホスフィン酸メチル、エチル−n−プロピルホスフィン酸メチル、n−ブチルエチルホスフィン酸メチル、ジ−n−プロピルホスフィン酸メチル、n−ブチル−n−プロピルホスフィン酸メチル、ジ−n−ブチルホスフィン酸メチル、ビス−(トリフルオロメチル)ホスフィン酸メチル、ビス−(トリフルオロメチル)ホスフィン酸トリフルオロメチル、ビス−(2,2,2−トリフルオロエチル)ホスフィン酸メチル、ビス−(2,2,2−トリフルオロエチル)ホスフィン酸トリフルオロメチル、ビス−(ペンタフルオロエチル)ホスフィン酸メチル、ビス−(ペンタフルオロエチル)ホスフィン酸トリフルオロメチル等が挙げられる。
Specific examples of the compound represented by the general formula (1) include the following.
Methyl dialkyl phosphinates: methyl dimethyl phosphinate, methyl ethyl methyl phosphinate, methyl methyl n-propyl phosphinate, methyl n-butyl methyl phosphinate, methyl diethyl phosphinate, methyl ethyl n-propyl phosphinate, n- Methyl butylethyl phosphinate, methyl di-n-propyl phosphinate, methyl n-butyl-n-propyl phosphinate, methyl di-n-butyl phosphinate, methyl bis- (trifluoromethyl) phosphinate, bis- (tri Fluoromethyl) trifluoromethyl, bis- (2,2,2-trifluoroethyl) phosphinic acid methyl, bis- (2,2,2-trifluoroethyl) phosphinic acid trifluoromethyl, bis- (pentafluoro Ethyl) phosphine Methyl, bis - (pentafluoroethyl) phosphinate trifluoromethyl, and the like.
ジアルキルホスフィン酸エチル類:ジメチルホスフィン酸エチル、エチルメチルホスフィン酸エチル、メチル−n−プロピルホスフィン酸エチル、n−ブチルメチルホスフィン酸エチル、ジエチルホスフィン酸エチル、エチル−n−プロピルホスフィン酸エチル、n−ブチルエチルホスフィン酸エチル、ジ−n−プロピルホスフィン酸エチル、n−ブチル−n−プロピルホスフィン酸エチル、ジ−n−ブチルホスフィン酸エチル、ビス−(トリフルオロメチル)ホスフィン酸エチル、ビス−(トリフルオロメチル)ホスフィン酸−2,2,2−トリフルオロエチル、ビス−(トリフルオロメチル)ホスフィン酸ペンタフルオロエチル、ビス−(2,2,2−トリフルオロエチル)ホスフィン酸エチル、ビス−(2,2,2−トリフルオロエチル)ホスフィン酸−2,2,2−トリフルオロエチル、ビス−(2,2,2−トリフルオロエチル)ホスフィン酸ペンタフルオロエチル、ビス−(ペンタフルオロエチル)ホスフィン酸エチル、ビス−(ペンタフルオロエチル)ホスフィン酸−2,2,2−トリフルオロエチル、ビス−(ペンタフルオロエチル)ホスフィン酸ペンタフルオロエチルが挙げられる。 Ethyl dialkyl phosphinates: ethyl dimethyl phosphinate, ethyl ethyl methyl phosphinate, ethyl methyl-n-propyl phosphinate, ethyl n-butyl methyl phosphinate, ethyl diethyl phosphinate, ethyl ethyl n-propyl phosphinate, n- Ethyl butylethyl phosphinate, ethyl di-n-propylphosphinate, ethyl n-butyl-n-propylphosphinate, ethyl di-n-butylphosphinate, ethyl bis- (trifluoromethyl) phosphinate, bis- (tri Fluoromethyl) phosphinic acid-2,2,2-trifluoroethyl, bis- (trifluoromethyl) phosphinic acid pentafluoroethyl, bis- (2,2,2-trifluoroethyl) phosphinic acid ethyl, bis- (2 , 2,2-trifluoroethyl ) -2,2,2-trifluoroethyl phosphinate, pentafluoroethyl bis- (2,2,2-trifluoroethyl) phosphinate, ethyl bis- (pentafluoroethyl) phosphinate, bis- (pentafluoroethyl) ) -2,2,2-trifluoroethyl phosphinate and pentafluoroethyl bis- (pentafluoroethyl) phosphinate.
ジアルキルホスフィン酸プロピル類:ジメチルホスフィン酸−n−プロピル、エチルメチルホスフィン酸−n−プロピル、メチル−n−プロピルホスフィン酸−n−プロピル、n−ブチルメチルホスフィン酸−n−プロピル、ジエチルホスフィン酸−n−プロピル、エチル−n−プロピルホスフィン酸−n−プロピル、n−ブチルエチルホスフィン酸−n−プロピル、ジ−n−プロピルホスフィン酸−n−プロピル、n−ブチル−n−プロピルホスフィン酸−n−プロピル、ジ−n−ブチルホスフィン酸−n−プロピル等が挙げられる。 Propyl dialkylphosphinates: n-propyl dimethylphosphinate, n-propyl ethylmethylphosphinate, n-propyl methyl-n-propylphosphinate, n-propyl n-butylmethylphosphinate, diethylphosphinate- n-propyl, ethyl-n-propyl-phosphinic acid-n-propyl, n-butylethylphosphinic acid-n-propyl, di-n-propylphosphinic acid-n-propyl, n-butyl-n-propylphosphinic acid-n -Propyl, -n-propyl di-n-butylphosphinate and the like.
ジアルキルホスフィン酸ブチル類:ジメチルホスフィン酸−n−ブチル、エチルメチルホスフィン酸−n−ブチル、メチル−n−プロピルホスフィン酸−n−ブチル、n−ブチルメチルホスフィン酸−n−ブチル、ジエチルホスフィン酸−n−ブチル、エチル−n−プロピルホスフィン酸−n−ブチル、n−ブチルエチルホスフィン酸−n−ブチル、ジ−n−プロピルホスフィン酸−n−ブチル、n−ブチル−n−プロピルホスフィン酸−n−ブチル、ジ−n−ブチルホスフィン酸−n−ブチル等が挙げられる。
ジアリールホスフィン酸アルキル類:ジフェニルホスフィン酸メチル、ジフェニルホスフィン酸エチル、ジフェニルホスフィン酸−n−プロピル、ジフェニルホスフィン酸−n−ブチル、ビス(2−トリル)ホスフィン酸メチル、ビス(2−トリル)ホスフィン酸エチル、ビス(2−トリル)ホスフィン酸−n−プロピル、ビス(2−トリル)ホスフィン酸−n−ブチル、ビス(3−トリル)ホスフィン酸メチル、ビス(3−トリル)ホスフィ
ン酸エチル、ビス(3−トリル)ホスフィン酸−n−プロピル、ビス(3−トリル)ホスフィン酸−n−ブチル、ビス(4−トリル)ホスフィン酸メチル、ビス(4−トリル)ホスフィン酸エチル、ビス(4−トリル)ホスフィン酸−n−プロピル、ビス(4−トリル)ホスフィン酸−n−ブチル等が挙げられる。
アルキルアリールホスフィン酸アルキル類:メチルフェニルホスフィン酸メチル、エチルフェニルホスフィン酸メチル、n−プロピルフェニルホスフィン酸メチル、n−ブチルフェニルホスフィン酸メチル、メチルフェニルホスフィン酸エチル、エチルフェニルホスフィン酸エチル、n−プロピルフェニルホスフィン酸エチル、n−ブチルフェニルホスフィン酸エチル、メチルフェニルホスフィン酸n−プロピル、エチルフェニルホスフィン酸n−プロピル、n−プロピルフェニルホスフィン酸n−プロピル、n−ブチルフェニルホスフィン酸n−プロピル、メチルフェニルホスフィン酸n−ブチル、エチルフェニルホスフィン酸n−ブチル、n−プロピルフェニルホスフィン酸n−ブチル、n−ブチルフェニルホスフィン酸n−ブチル、メチル−2−トリルホスフィン酸メチル、エチル−2−トリルホスフィン酸メチル、n−プロピル−2−トリルホスフィン酸メチル、n−ブチル−2−トリルホスフィン酸メチル、メチル−2−トリルホスフィン酸エチル、エチル−2−トリルホスフィン酸エチル、n−プロピル−2−トリルホスフィン酸エチル、n−ブチル−2−トリルホスフィン酸エチル、メチル−2−トリルホスフィン酸n−プロピル、エチル−2−トリルホスフィン酸n−プロピル、n−プロピル−2−トリルホスフィン酸n−プロピル、n−ブチル−2−トリルホスフィン酸n−プロピル、メチル−2−トリルホスフィン酸n−ブチル、エチル−2−トリルホスフィン酸n−ブチル、n−プロピル−2−トリルホスフィン酸n−ブチル、n−ブチル−2−トリルホスフィン酸n−ブチル、メチル−3−トリルホスフィン酸メチル、エチル−3−トリルホスフィン酸メチル、n−プロピル−3−トリルホスフィン酸メチル、n−ブチル−3−トリルホスフィン酸メチル、メチル−3−トリルホスフィン酸エチル、エチル−3−トリルホスフィン酸エチル、n−プロピル−3−トリルホスフィン酸エチル、n−ブチル−3−トリルホスフィン酸エチル、メチル−3−トリルホスフィン酸n−プロピル、エチル−3−トリルホスフィン酸n−プロピル、n−プロピル−3−トリルホスフィン酸n−プロピル、n−ブチル−3−トリルホスフィン酸n−プロピル、メチル−3−トリルホスフィン酸n−ブチル、エチル−3−トリルホスフィン酸n−ブチル、n−プロピル−3−トリルホスフィン酸n−ブチル、n−ブチル−3−トリルホスフィン酸n−ブチル、メチル−4−トリルホスフィン酸メチル、エチル−4−トリルホスフィン酸メチル、n−プロピル−4−トリルホスフィン酸メチル、n−ブチル−4−トリルホスフィン酸メチル、メチル−4−トリルホスフィン酸エチル、エチル−4−トリルホスフィン酸エチル、n−プロピル−4−トリルホスフィン酸エチル、n−ブチル−4−トリルホスフィン酸エチル、メチル−4−トリルホスフィン酸n−プロピル、エチル−4−トリルホスフィン酸n−プロピル、n−プロピル−4−トリルホスフィン酸n−プロピル、n−ブチル−4−トリルホスフィン酸n−プロピル、メチル−4−トリルホスフィン酸n−ブチル、エチル−4−トリルホスフィン酸n−ブチル、n−プロピル−4−トリルホスフィン酸n−ブチル、n−ブチル−4−トリルホスフィン酸n−ブチル等が挙げられる。
ジアルキルホスフィン酸アリール類:ジメチルホスフィン酸フェニル、エチルメチルホスフィン酸フェニル、ジエチルホスフィン酸フェニル、メチル−n−プロピルホスフィン酸フェニル、メチル−n−ブチルホスフィン酸フェニル、エチル−n−プロピルホスフィン酸フェニル、エチル−n−ブチルホスフィン酸フェニル、ジ−n−プロピルホスフィン酸フェニル、n−ブチル−n−プロピルホスフィン酸フェニル、ジ−n−ブチルホスフィン酸フェニル、ジメチルホスフィン酸−2−トリル、エチルメチルホスフィン酸−2−トリル、ジエチルホスフィン酸−2−トリル、メチル−n−プロピルホスフィン酸−2−トリル、メチル−n−ブチルホスフィン酸−2−トリル、エチル−n−プロピルホスフィン酸−2−トリル、エチル−n−ブチルホスフィン酸−2−トリル、ジ−n−プロピルホスフィン酸−2−トリル、n−ブチル−n−プロピルホスフィン酸−2−トリル、ジ−n−ブチルホスフィン酸−2−トリル、ジメチルホスフィン酸−3−トリル、エチルメチルホ
スフィン酸−3−トリル、ジエチルホスフィン酸−3−トリル、メチル−n−プロピルホスフィン酸−3−トリル、メチル−n−ブチルホスフィン酸−3−トリル、エチル−n−プロピルホスフィン酸−3−トリル、エチル−n−ブチルホスフィン酸−3−トリル、ジ−n−プロピルホスフィン酸−3−トリル、n−ブチル−n−プロピルホスフィン酸−3−トリル、ジ−n−ブチルホスフィン酸−3−トリル、ジメチルホスフィン酸−4−トリル、エチルメチルホスフィン酸−4−トリル、ジエチルホスフィン酸−4−トリル、メチル−n−プロピルホスフィン酸−4−トリル、メチル−n−ブチルホスフィン酸−4−トリル、エチル−n−プロピルホスフィン酸−4−トリル、エチル−n−ブチルホスフィン酸−4−トリル、ジ−n−プロピルホスフィン酸−4−トリル、n−ブチル−n−プロピルホスフィン酸−4−トリル、ジ−n−ブチルホスフィン酸−4−トリル等が挙げられる。
一般式(1)で表される化合物の分子量は、通常500以下、好ましくは400以下、より好ましくは350以下である。分子量が大きすぎると電解液に対する溶解性が悪くなり、本発明の効果を十分に発現できない恐れがある。一般式(1)で表される化合物は、単独で用いても、2種以上を混合して用いてもよい。また、本発明を満たす限りにおいて、一般式(1)以外のホスフィン酸エステル化合物と混合して用いても良い。
Dialkylphosphinic acid butyls: dimethylphosphinic acid-n-butyl, ethylmethylphosphinic acid-n-butyl, methyl-n-propylphosphinic acid-n-butyl, n-butylmethylphosphinic acid-n-butyl, diethylphosphinic acid- n-butyl, n-butyl ethyl-n-propylphosphinate, n-butyl n-butylethylphosphinate, n-butyl di-n-propylphosphinate, n-butyl-n-propylphosphinate-n -Butyl, -n-butyl di-n-butylphosphinate and the like.
Alkyl diarylphosphinates: methyl diphenylphosphinate, ethyl diphenylphosphinate, n-propyl diphenylphosphinate, n-butyl diphenylphosphinate, methyl bis (2-tolyl) phosphinate, bis (2-tolyl) phosphinic acid Ethyl, n-propyl bis (2-tolyl) phosphinate, n-butyl bis (2-tolyl) phosphinate, methyl bis (3-tolyl) phosphinate, ethyl bis (3-tolyl) phosphinate, bis ( N-propyl 3-tolyl) phosphinate, n-butyl bis (3-tolyl) phosphinate, methyl bis (4-tolyl) phosphinate, ethyl bis (4-tolyl) phosphinate, bis (4-tolyl) N-propyl phosphinate, n-butyl bis (4-tolyl) phosphinate, etc. And the like.
Alkyl alkyl aryl phosphinates: methyl methyl phenyl phosphinate, methyl ethyl phenyl phosphinate, methyl n-propyl phenyl phosphinate, methyl n-butyl phenyl phosphinate, ethyl methyl phenyl phosphinate, ethyl ethyl phenyl phosphinate, n-propyl Ethyl phenylphosphinate, ethyl n-butylphenylphosphinate, n-propyl methylphenylphosphinate, n-propyl ethylphenylphosphinate, n-propyl n-propylphenylphosphinate, n-propyl n-butylphenylphosphinate, methyl N-butyl phenylphosphinate, n-butyl ethylphenylphosphinate, n-butyl n-propylphenylphosphinate, n-butyl n-butylphenylphosphinate, Methyl 2-tolyl phosphinate, methyl ethyl-2-tolyl phosphinate, methyl n-propyl-2-tolyl phosphinate, methyl n-butyl-2-tolyl phosphinate, ethyl methyl-2-tolyl phosphinate, ethyl Ethyl-2-tolylphosphinate, ethyl n-propyl-2-tolylphosphinate, ethyl n-butyl-2-tolylphosphinate, n-propyl methyl-2-tolylphosphinate, n-ethyl-2-tolylphosphinate Propyl, n-propyl n-propyl-2-tolylphosphinate, n-propyl n-butyl-2-tolylphosphinate, n-butyl methyl-2-tolylphosphinate, n-butyl ethyl-2-tolylphosphinate, n-butyl-2-tolylphosphinic acid n-butyl, n-butyl-2-tolylphos N-butyl phosphate, methyl methyl-3-tolylphosphinate, methyl ethyl-3-tolylphosphinate, methyl n-propyl-3-tolylphosphinate, methyl n-butyl-3-tolylphosphinate, methyl-3 -Ethyl tolylphosphinate, ethyl-3-ethyltolylphosphinate, ethyl n-propyl-3-tolylphosphinate, ethyl n-butyl-3-tolylphosphinate, n-propyl methyl-3-tolylphosphinate, ethyl- N-propyl 3-tolylphosphinate, n-propyl n-propyl-3-tolylphosphinate, n-propyl n-butyl-3-tolylphosphinate, n-butyl methyl-3-tolylphosphinate, ethyl-3- N-butyl tolyl phosphinate, n-butyl n-propyl-3-tolyl phosphinate, n-butyl N-butyl tyl-3-tolylphosphinate, methyl methyl-4-tolylphosphinate, methyl ethyl-4-tolylphosphinate, methyl n-propyl-4-tolylphosphinate, methyl n-butyl-4-tolylphosphinate , Ethyl methyl-4-tolylphosphinate, ethyl ethyl-4-tolylphosphinate, ethyl n-propyl-4-tolylphosphinate, ethyl n-butyl-4-tolylphosphinate, n-methyl-4-tolylphosphinate Propyl, n-propyl ethyl-4-tolylphosphinate, n-propyl n-propyl-4-tolylphosphinate, n-propyl n-butyl-4-tolylphosphinate, n-butyl methyl-4-tolylphosphinate, N-butyl ethyl-4-tolylphosphinate, n-propyl-4-tolylphosphite Acid n- butyl, n- butyl-4-tolyl phosphinic acid n- butyl, and the like.
Aryl dialkyl phosphinates: phenyl dimethyl phosphinate, phenyl ethyl methyl phosphinate, phenyl diethyl phosphinate, phenyl methyl-n-propyl phosphinate, phenyl methyl-n-butyl phosphinate, phenyl ethyl-n-propyl phosphinate, ethyl -Phenyl n-butyl phosphinate, phenyl di-n-propyl phosphinate, phenyl n-butyl-n-propyl phosphinate, phenyl di-n-butyl phosphinate, dimethyl phosphinate-2-tolyl, ethyl methyl phosphinate- 2-tolyl, diethyl-2-phosphoric acid-2-tolyl, methyl-n-propylphosphinic acid-2-tolyl, methyl-n-butylphosphinic acid-2-tolyl, ethyl-n-propylphosphinic acid-2-tolyl, ethyl- n- Tylphosphinic acid-2-tolyl, di-n-propylphosphinic acid-2-tolyl, n-butyl-n-propylphosphinic acid-2-tolyl, di-n-butylphosphinic acid-2-tolyl, dimethylphosphinic acid- 3-tolyl, ethyl methylphosphinic acid-3-tolyl, diethylphosphinic acid-3-tolyl, methyl-n-propylphosphinic acid-3-tolyl, methyl-n-butylphosphinic acid-3-tolyl, ethyl-n-propyl Phosphinic acid-3-tolyl, ethyl-n-butylphosphinic acid-3-tolyl, di-n-propylphosphinic acid-3-tolyl, n-butyl-n-propylphosphinic acid-3-tolyl, di-n-butyl Phosphinic acid-3-tolyl, dimethylphosphinic acid-4-tolyl, ethylmethylphosphinic acid-4-tolyl, diethylphosphinic acid 4-tolyl diacid, 4-tolyl methyl-n-propylphosphinate, 4-tolyl methyl-n-butylphosphinate, 4-tolyl ethyl-n-propylphosphinate, ethyl-n-butylphosphine Acid-4-tolyl, di-n-propylphosphinic acid-4-tolyl, n-butyl-n-propylphosphinic acid-4-tolyl, di-n-butylphosphinic acid-4-tolyl and the like.
The molecular weight of the compound represented by the general formula (1) is usually 500 or less, preferably 400 or less, more preferably 350 or less. If the molecular weight is too large, the solubility in the electrolytic solution becomes poor, and the effect of the present invention may not be sufficiently exhibited. The compounds represented by the general formula (1) may be used alone or in combination of two or more. Further, as long as the present invention is satisfied, a mixture with a phosphinic acid ester compound other than the general formula (1) may be used.
非水系電解液に占める一般式(1)で表される化合物の含有量は、非水系電解液の全重量に対して、通常0.01重量%以上、好ましくは0.05重量%以上、より好ましくは0.1重量%以上、通常4.5重量%以下、好ましくは3重量%以下、より好ましくは2.5重量%以下である。一般式(1)で表される化合物の濃度が低すぎると十分な効果が得られず、また高すぎるとレート特性などの電池特性が低下する。
本発明に係る非水系電解液は、必要に応じて、常用の他の助剤、例えば、過充電防止剤、電池の活物質表面に被膜(SEI)を形成させるための被膜形成剤等を含有していてもよい。過充電防止剤としては、ビフェニル及びその誘導体、シクロヘキシルベンゼン及びその誘導体、ジベンゾフラン及びその誘導体、ターフェニル及びその誘導体、ジフェニルエーテル及びその誘導体等が挙げられる。被膜形成剤としては、ビニレンカーボネート、ビニルエチレンカーボネート等が挙げられる。
非水系電解液に占める助剤の各々の濃度としては、非水系電解液の全重量に対して、通常0.1重量%以上、好ましくは0.5重量%以上、さらに好ましくは1重量%以上、通常10重量%以下、好ましくは8重量%以下、さらに好ましくは6重量%以下である。また、複数の助剤を併用する場合の濃度も同様である。
The content of the compound represented by the general formula (1) in the nonaqueous electrolyte is usually 0.01% by weight or more, preferably 0.05% by weight or more, based on the total weight of the nonaqueous electrolyte. It is preferably at least 0.1% by weight, usually at most 4.5% by weight, preferably at most 3% by weight, more preferably at most 2.5% by weight. If the concentration of the compound represented by the general formula (1) is too low, a sufficient effect cannot be obtained. If the concentration is too high, battery characteristics such as rate characteristics deteriorate.
The non-aqueous electrolyte according to the present invention contains, if necessary, other conventional auxiliaries, for example, an overcharge inhibitor, a film forming agent for forming a film (SEI) on the surface of the active material of the battery, and the like. It may be. Examples of the overcharge inhibitor include biphenyl and its derivatives, cyclohexylbenzene and its derivatives, dibenzofuran and its derivatives, terphenyl and its derivatives, diphenyl ether and its derivatives, and the like. Examples of the film forming agent include vinylene carbonate and vinyl ethylene carbonate.
The concentration of each auxiliary in the non-aqueous electrolyte is usually 0.1% by weight or more, preferably 0.5% by weight or more, more preferably 1% by weight or more, based on the total weight of the non-aqueous electrolyte. Usually, it is 10% by weight or less, preferably 8% by weight or less, more preferably 6% by weight or less. The same applies to the case where a plurality of auxiliaries are used in combination.
本発明に係る二次電池用非水系電解液は、前述の非水系有機溶媒に、溶質、一般式(1)で表される化合物、及び必要に応じて他の助剤を溶解することにより調製することができる。非水系電解液の調製に際しては、非水系電解液の各原料は、予め脱水しておくのが好ましい。通常は、50ppm以下、好ましくは30ppm以下まで脱水する。非水系電解液に水が存在すると、水の電気分解、水と溶質との反応による、溶質の加水分解などが起こる可能性がある。脱水の手段は特に制限はないが、溶媒などの液体の場合はモレキュラーシーブ等で水を吸着除去すればよい。また、溶質などの固体の場合は分解が起きる温度以下で乾燥すればよい。 The non-aqueous electrolyte for a secondary battery according to the present invention is prepared by dissolving a solute, a compound represented by the general formula (1), and, if necessary, other auxiliaries in the above-mentioned non-aqueous organic solvent. can do. In preparing the non-aqueous electrolyte, each raw material of the non-aqueous electrolyte is preferably dehydrated in advance. Usually, the water is dehydrated to 50 ppm or less, preferably 30 ppm or less. If water is present in the non-aqueous electrolyte, electrolysis of water, hydrolysis of the solute due to the reaction between the water and the solute, and the like may occur. The means for dehydration is not particularly limited. In the case of a liquid such as a solvent, water may be removed by adsorption using a molecular sieve or the like. In the case of a solid such as a solute, drying may be performed at a temperature lower than a temperature at which decomposition occurs.
本発明に係る二次電池用非水系電解液は、リチウム二次電池用の電解液として用いるのに好適である。以下、この電解液を用いた本発明に係るリチウム二次電池について説明する。
本発明に係るリチウム二次電池は、電解液以外は従来公知のリチウム二次電池と同様であり、通常、正極と負極とが本発明の非水系電解液を含んでいるセパレーターを介してケースに収納されている。従って、本発明に係る二次電池の形状は特に限定されるものではなく、シート電極及びセパレータをスパイラル状にしたシリンダータイプ、ペレット電極及びセパレータを組み合わせたインサイドアウト構造のシリンダータイプ、ペレット電極
及びセパレータを積層したコインタイプのいずれであってもよい。
The non-aqueous electrolyte for a secondary battery according to the present invention is suitable for use as an electrolyte for a lithium secondary battery. Hereinafter, the lithium secondary battery according to the present invention using the electrolytic solution will be described.
The lithium secondary battery according to the present invention is the same as a conventionally known lithium secondary battery except for the electrolytic solution.In general, the positive electrode and the negative electrode are connected to the case via a separator containing the nonaqueous electrolytic solution of the present invention. It is stored. Accordingly, the shape of the secondary battery according to the present invention is not particularly limited, and a cylinder type in which a sheet electrode and a separator are formed in a spiral shape, a cylinder type having an inside-out structure in which a pellet electrode and a separator are combined, a pellet electrode and a separator. May be any of coin types in which are stacked.
正極活物質としては、遷移金属の酸化物、遷移金属とリチウムとの複合酸化物、遷移金属の硫化物、金属酸化物等の無機化合物、リチウム金属、リチウム合金が挙げられる。具体的には、MnO、V2O5、V6O13、TiO2等の遷移金属酸化物、基本組成がLiCoO2であるリチウムコバルト複合酸化物、LiNiO2であるリチウムニッケル複合酸化物、LiMn2O4またはLiMnO2であるリチウムマンガン複合酸化物等のリチウム遷移
金属複合酸化物、TiS、FeS等の遷移金属硫化物、SnO2、SiO2等の金属酸化物が挙げられる。中でもリチウム遷移金属複合酸化物、特にリチウムコバルト複合酸化物、リチウムニッケル複合酸化物、リチウムコバルトニッケル複合酸化物は、高容量と高サイクル特性とを両立させ得るので好適に用いられる。また、リチウム遷移金属複合酸化物は、コバルト、ニッケルまたはマンガンの一部をAl、Ti、V、Cr、Mn、Fe、Co、Li、Ni、Cu、Zn、Mg、Ga、Zr等の他の金属で置き換えることにより、その構造を安定化させることができるので好ましい。正極活物質は、単独で用いても、2種以上を混合して用いてもよい。
Examples of the positive electrode active material include transition metal oxides, composite oxides of transition metals and lithium, sulfides of transition metals, inorganic compounds such as metal oxides, lithium metals, and lithium alloys. Specifically, MnO, V 2 O 5, V 6 O 13, TiO transition metal oxides such as 2, lithium-cobalt composite oxide basic composition is LiCoO 2, lithium-nickel composite oxide is LiNiO 2, LiMn Examples thereof include lithium transition metal composite oxides such as lithium manganese composite oxides that are 2 O 4 and LiMnO 2 , transition metal sulfides such as TiS and FeS, and metal oxides such as SnO 2 and SiO 2 . Among them, a lithium transition metal composite oxide, particularly a lithium cobalt composite oxide, a lithium nickel composite oxide, and a lithium cobalt nickel composite oxide are preferably used because they can achieve both high capacity and high cycle characteristics. In addition, the lithium transition metal composite oxide has a part of cobalt, nickel, or manganese which is made of Al, Ti, V, Cr, Mn, Fe, Co, Li, Ni, Cu, Zn, Mg, Ga, or Zr. Replacement with a metal is preferable because its structure can be stabilized. The positive electrode active materials may be used alone or as a mixture of two or more.
負極活物質としては、リチウムを吸蔵及び放出し得る物質であればよく、リチウム金属、リチウム合金などを用いることができるが、サイクル特性及び安全性が良好な点で、炭素質材料が好ましい。炭素質材料としては、天然ないし人造の黒鉛、ピッチの炭化物、フェノール樹脂やセルロース等の炭化物、ピッチ系炭素繊維、PAN系炭素繊維、メソフェーズ小球体などの黒鉛化したもの、更にはファーネスブラック、アセチレンブラックやその黒鉛化物などが挙げられる。また、これらの炭素質材料をピッチ等の有機物で被覆した後、焼成し、表面にこれらの炭素質材料に比べて非晶質の炭素を形成したものも好適に用いることができる。 As the negative electrode active material, any material capable of inserting and extracting lithium may be used, and lithium metal, lithium alloy, and the like can be used. However, a carbonaceous material is preferable in terms of good cycle characteristics and safety. Examples of the carbonaceous material include natural or artificial graphite, carbide of pitch, carbide such as phenol resin and cellulose, pitch-based carbon fiber, PAN-based carbon fiber, graphitized material such as mesophase spherules, furnace black, and acetylene. Black and its graphitized products are exemplified. Further, a material obtained by coating these carbonaceous materials with an organic substance such as pitch and then baking to form amorphous carbon on the surface as compared with these carbonaceous materials can also be suitably used.
これらの炭素質材料は、学振法によるX線回折で求めた格子面(002面)のd値(層間距離)が0.335〜0.340nmであるものが好ましく、0.335〜0.337nmであるものがより好ましい。灰分は1重量%以下であるのが好ましく、0.5重量%以下であるのがより好ましく、0.1重量%以下であるのが特に好ましい。また、学振法によるX線回折で求めた結晶子サイズ(Lc)は30nm以上であるのが好ましく、50nm以上であるのがより好ましく、100nm以上であるのが特に好ましい。 These carbonaceous materials preferably have a lattice plane (002 plane) d value (interlayer distance) of 0.335 to 0.340 nm, preferably 0.335 to 0.30 nm, determined by X-ray diffraction by the Gakushin method. Those having 337 nm are more preferable. The ash content is preferably at most 1% by weight, more preferably at most 0.5% by weight, particularly preferably at most 0.1% by weight. The crystallite size (Lc) determined by X-ray diffraction according to the Gakushin method is preferably 30 nm or more, more preferably 50 nm or more, and particularly preferably 100 nm or more.
活物質を結着する結着剤としては、ポリフッ化ビニリデン、ポリテトラフルオロエチレン等のフッ素系樹脂、スチレン・ブタジエンゴム、イソプレンゴム、ブダジエンゴム、ポリ酢酸ビニル、ポリエチルメタクリレート、ポリエチレン、ニトロセルロース等を挙げることができる。
結着剤の使用量は、活物質100重量部に対して通常0.1重量部以上、好ましくは1重量部以上であり、通常30重量部以下、好ましくは20重量部以下である。結着剤の量が少なすぎると電極の強度が低下する傾向にあり、逆に多すぎるとイオン伝導度が低下する傾向にある。
Examples of the binder for binding the active material include fluorine resins such as polyvinylidene fluoride and polytetrafluoroethylene, styrene / butadiene rubber, isoprene rubber, butadiene rubber, polyvinyl acetate, polyethyl methacrylate, polyethylene, and nitrocellulose. Can be mentioned.
The amount of the binder used is usually at least 0.1 part by weight, preferably at least 1 part by weight, and usually at most 30 parts by weight, preferably at most 20 parts by weight, per 100 parts by weight of the active material. If the amount of the binder is too small, the strength of the electrode tends to decrease, and if it is too large, the ion conductivity tends to decrease.
電極中には、電気伝導度や機械的強度を向上させるために、導電性材料、補強材などの各種の機能を発現する助剤、粉体、充填材、増粘剤を含有させてもよい。導電性材料としては、上記活物質に適量混合して導電性を付与できるものであれば特に制限はないが、通常、銅、ニッケル等の各種金属の繊維や箔、グラファイト、カーボンブラック等の炭素質材料が挙げられる。特に正極には、導電材を含有させるのが好ましい。増粘剤としては、カルボキシエチルセルロース、エチルセルロース、ヒドロキシエチルセルロース、エチルセルロース、ポリビニルアルコール、酸化スターチ、リン酸化スターチ、カゼイン等が挙げられる。 In the electrode, in order to improve electrical conductivity and mechanical strength, conductive materials, auxiliary agents that express various functions such as reinforcing materials, powders, fillers, and thickeners may be contained. . The conductive material is not particularly limited as long as it can impart conductivity by being mixed in an appropriate amount with the above active material, and usually, fibers and foils of various metals such as copper and nickel, graphite and carbon such as carbon black are used. Quality material. In particular, the positive electrode preferably contains a conductive material. Examples of the thickener include carboxyethyl cellulose, ethyl cellulose, hydroxyethyl cellulose, ethyl cellulose, polyvinyl alcohol, oxidized starch, phosphorylated starch, and casein.
電極は、活物質に結着剤及び導電材その他を配合したものをそのままロール成形することによりシート電極としたり、圧縮成形によりペレット電極とすることもできるが、通常は活物質に結着剤及び導電材その他を配合したものを溶剤でスラリー化し、これを、集電体に塗布、乾燥することによって形成する。
塗布により形成される活物質層の乾燥厚さは、通常1μm以上、好ましくは10μm以上、さらに好ましくは20μm以上、最も好ましくは40μm以上であり、通常200μm以下、好ましくは150μm以下、さらに好ましくは100μm以下である。薄すぎると均一塗布が困難になるだけでなく、電池の容量が小さくなる。一方、厚すぎるとレート特性が低下する。
The electrode may be formed into a sheet electrode by directly rolling the mixture of the active material with a binder and a conductive material and the like, or may be formed into a pellet electrode by compression molding. It is formed by slurrying a mixture of a conductive material and the like with a solvent, applying the slurry to a current collector, and drying.
The dry thickness of the active material layer formed by coating is usually 1 μm or more, preferably 10 μm or more, more preferably 20 μm or more, and most preferably 40 μm or more, and usually 200 μm or less, preferably 150 μm or less, and more preferably 100 μm or less. It is as follows. If it is too thin, not only uniform coating becomes difficult, but also the capacity of the battery becomes small. On the other hand, if the thickness is too large, the rate characteristics deteriorate.
集電体としては、通常は金属や合金が用いられる。具体的には、負極集電体としては、銅およびその合金、ニッケルおよびその合金、ステンレス等が挙げられ、なかでも銅およびその合金が好ましい。正極集電体としては、アルミニウム、チタン、タンタルおよびこれらの合金等が挙げられ、なかでもアルミニウムおよびその合金が好ましい。表面に形成される活物質層との結着効果を向上させるため、これら集電体の表面は予め粗面化処理しておくのが好ましい。表面の粗面化方法としては、ブラスト処理、粗面ロールによる圧延、研磨剤粒子を固着した研磨布紙、砥石、エメリバフ、鋼線などを備えたワイヤーブラシなどで集電体表面を研磨する機械的研磨法、電解研磨法、化学研磨法等が挙げられる。 As the current collector, a metal or an alloy is usually used. Specifically, examples of the negative electrode current collector include copper and its alloys, nickel and its alloys, and stainless steel. Among them, copper and its alloys are preferable. Examples of the positive electrode current collector include aluminum, titanium, tantalum, and alloys thereof, and among them, aluminum and alloys thereof are preferable. In order to improve the binding effect with the active material layer formed on the surface, it is preferable that the surface of these current collectors is previously subjected to a roughening treatment. Examples of surface roughening methods include blasting, rolling with a rough roll, and a machine for polishing the current collector surface with a wire cloth equipped with abrasive cloth paper, whetstones, emery buffs, steel wires, etc. to which abrasive particles are fixed. Polishing method, electrolytic polishing method, chemical polishing method and the like.
また、集電体の重量を低減させて電池の重量当たりのエネルギー密度を向上させるために、エキスパンドメタルやパンチングメタルのような穴あきタイプの集電体を使用することもできる。このタイプの集電体は、その開口率を変更することで、重量も自在に変更可能である。また、このタイプの集電体の両面に活物質層を形成させた場合、この穴を通してのリベット効果により活物質層の剥離がさらに起こりにくくなる。しかし、開口率があまりに高くなった場合には、活物質層と集電体との接触面積が小さくなるため、かえって接着強度は低くなることがある。 Further, in order to reduce the weight of the current collector and improve the energy density per unit weight of the battery, a perforated current collector such as an expanded metal or a punched metal may be used. The weight of this type of current collector can be freely changed by changing its aperture ratio. When an active material layer is formed on both sides of this type of current collector, the active material layer is less likely to peel off due to a rivet effect through this hole. However, when the aperture ratio is too high, the contact area between the active material layer and the current collector is reduced, and the bonding strength may be reduced instead.
集電体の厚さは、通常1μm以上、好ましくは5μm以上であり、通常100μm以下、好ましくは50μm以下である。厚すぎると、電池全体の容量が低下しすぎることになり、逆に薄すぎると取り扱いが困難になることがある。
非水系電解液は、これを高分子などのゲル化剤でゲル化して半固体状にして用いてもよい。半固体状電解質における上記非水系電解液の占める比率は、半固体状電解質の総量に対して、通常30重量%以上、好ましくは50重量%以上、さらに好ましくは75重量%以上であり、通常99.95重量%以下、好ましくは99重量%以下、さらに好ましくは98重量%以下である。電解液の比率が大きすぎると、電解液の保持が困難となり液漏れが生じやすくなり、逆に少なすぎると充放電効率や容量の点で不十分となることがある。
The thickness of the current collector is usually 1 μm or more, preferably 5 μm or more, and usually 100 μm or less, preferably 50 μm or less. If it is too thick, the capacity of the entire battery will be too low, and if it is too thin, handling may be difficult.
The non-aqueous electrolyte may be used in a semi-solid state by gelling it with a gelling agent such as a polymer. The proportion of the non-aqueous electrolyte in the semi-solid electrolyte is usually at least 30% by weight, preferably at least 50% by weight, more preferably at least 75% by weight, and usually at least 99% by weight, based on the total amount of the semi-solid electrolyte. 0.95% by weight or less, preferably 99% by weight or less, more preferably 98% by weight or less. If the ratio of the electrolytic solution is too large, it is difficult to hold the electrolytic solution and the liquid is likely to leak. Conversely, if the ratio is too small, the charge / discharge efficiency and capacity may be insufficient.
正極と負極の間には、短絡を防止するために、セパレーターを介在させる。この場合、電解液は、セパレーターに含浸させて用いる。セパレーターの材料や形状については、特に限定されないが、電解液に対して安定な材料で形成された保液性に優れた多孔性シート又は不織布等を用いるのが好ましい。セパレータの材料としては、ポリエチレン、ポリプロピレン等のポリオレフィン、ポリテトラフルオロエチレン、ポリエーテルスルホン等を用いることができるが、好ましくはポリオレフィンである。 A separator is interposed between the positive electrode and the negative electrode to prevent a short circuit. In this case, the electrolytic solution is used by impregnating the separator. The material and shape of the separator are not particularly limited, but it is preferable to use a porous sheet or a nonwoven fabric formed of a material stable to the electrolytic solution and having excellent liquid retaining properties. As a material of the separator, polyolefin such as polyethylene and polypropylene, polytetrafluoroethylene, polyether sulfone, and the like can be used, and polyolefin is preferable.
セパレーターの厚さは、通常1μm以上、好ましくは5μm以上、さらに好ましくは10μm以上であり、通常50μm以下、好ましくは40μm以下、さらに好ましくは30μm以下である。セパレーターが薄すぎると、絶縁性や機械的強度が悪化することがあり、厚すぎるとレート特性等の電池性能が悪化するばかりでなく、電池全体としてのエネルギー密度が低下する。 The thickness of the separator is usually at least 1 μm, preferably at least 5 μm, more preferably at least 10 μm, usually at most 50 μm, preferably at most 40 μm, more preferably at most 30 μm. If the separator is too thin, the insulating properties and mechanical strength may be deteriorated. If the separator is too thick, not only the battery performance such as rate characteristics will deteriorate, but also the energy density of the whole battery will decrease.
セパレーターの空孔率は、通常20%以上、好ましくは35%以上、さらに好ましくは45%以上であり、通常90%以下、好ましくは85%以下、さらに好ましくは75%以下である。空孔率が小さすぎると膜抵抗が大きくなり、レート特性が悪化する傾向にある。また、大きすぎるとセパレーターの機械的強度が低下し、絶縁性が低下する傾向にある。 The porosity of the separator is usually at least 20%, preferably at least 35%, more preferably at least 45%, usually at most 90%, preferably at most 85%, more preferably at most 75%. If the porosity is too small, the film resistance tends to increase, and the rate characteristics tend to deteriorate. On the other hand, if it is too large, the mechanical strength of the separator tends to decrease, and the insulating property tends to decrease.
セパレーターの平均孔径は、通常0.5μm以下、好ましくは0.2μm以下であり、通常0.05μm以上である。平均孔径が大きすぎると短絡が生じやすくなり、小さすぎると膜抵抗が大きくなりレート特性が悪化することがある。
The average pore size of the separator is usually 0.5 μm or less, preferably 0.2 μm or less, and usually 0.05 μm or more. If the average pore size is too large, a short circuit is likely to occur. If the average pore size is too small, the film resistance increases and the rate characteristics may deteriorate.
以下、実施例を挙げて本発明をさらに具体的に説明するが、本発明はその要旨を越えない限り以下の実施例に限定されるものではない。
(正極の製造)
コバルト酸リチウム(LiCoO2)90重量部、アセチレンブラック5重量部及びポ
リフッ化ビニリデン(以下「PVdF」ということがある)5重量部を混合し、N−エチルピロリドンを加えてスラリー状にした。これを厚さ20μmのアルミニウム箔の片面に塗布、乾燥し、さらにプレス機で圧延した。これからポンチで直径12mmの円板に打ち抜き、正極とした。
Hereinafter, the present invention will be described more specifically with reference to examples. However, the present invention is not limited to the following examples unless departing from the gist of the present invention.
(Manufacture of positive electrode)
90 parts by weight of lithium cobalt oxide (LiCoO 2 ), 5 parts by weight of acetylene black and 5 parts by weight of polyvinylidene fluoride (hereinafter sometimes referred to as “PVdF”) were mixed, and N-ethylpyrrolidone was added to form a slurry. This was applied to one side of a 20 μm-thick aluminum foil, dried, and further rolled by a press. From this, a 12 mm diameter disk was punched out with a punch to obtain a positive electrode.
(負極の製造)
黒鉛(面間隔0.336nm)95重量部とPVdF5重量部とを混合し、N−エチルピロリドンを加えてスラリー状にした。これを厚さ20μmの銅箔の片面に塗布、乾燥し、さらにプレス機で圧延した。これから直径12mmの円板を打ち抜き、負極とした。
(リチウム二次電池の製造)
アルゴン雰囲気のドライボックス内で、CR2032型コインセルを使用して、リチウム二次電池を作成した。即ち、コイン型セル(正極缶)に正極を置き、その上に厚さ25μmの多孔性ポリエチレンフィルム(セパレーター)を置き、ポリプロピレン製ガスケットで押さえた。ガスケット上に負極を置き、更に厚み調整用のスペーサーを置いた。電解液を加え電池内に十分しみこませた後、コイン型セル(負極缶)を載せ、封口して電池とした。
(Manufacture of negative electrode)
95 parts by weight of graphite (plane spacing 0.336 nm) and 5 parts by weight of PVdF were mixed, and N-ethylpyrrolidone was added to form a slurry. This was applied to one side of a copper foil having a thickness of 20 μm, dried, and further rolled by a press. From this, a disk having a diameter of 12 mm was punched to obtain a negative electrode.
(Manufacture of lithium secondary batteries)
A lithium secondary battery was prepared in a dry box in an argon atmosphere using a CR2032 type coin cell. That is, the positive electrode was placed in a coin-type cell (positive electrode can), a porous polyethylene film (separator) having a thickness of 25 μm was placed thereon, and pressed with a polypropylene gasket. The negative electrode was placed on the gasket, and a spacer for adjusting the thickness was further placed. After adding an electrolytic solution and sufficiently impregnating the inside of the battery, a coin-type cell (negative electrode can) was placed thereon, and the battery was sealed and closed.
なお、以下の実施例および比較例において、電池の容量は、充電上限4.2V、放電下限3.0Vで約4.0mAhになるように設計した。
正極活物質重量W(c)と負極の活物質重量W(a)の比率は、負極と正極との容量比Rqが1.1≦Rq≦1.2となるように、その重量を決定した。なお、容量比Rqは次式で求めた。
In the following Examples and Comparative Examples, the capacity of the battery was designed to be about 4.0 mAh at a charge upper limit of 4.2 V and a discharge lower limit of 3.0 V.
The ratio of the positive electrode active material weight W (c) to the negative electrode active material weight W (a) was determined such that the capacity ratio Rq between the negative electrode and the positive electrode was 1.1 ≦ Rq ≦ 1.2. . The capacity ratio Rq was determined by the following equation.
ここで、Q(c)(mAh/g)は電池の初期充電条件に対応する条件下での正極活物質の重量当たりの電気容量、Q(a)(mAh/g)はリチウム金属が析出することなしにリチウムを最大限に吸蔵しうる負極活物質の重量当たりの電気容量である。 Here, Q (c) (mAh / g) is the electric capacity per weight of the positive electrode active material under conditions corresponding to the initial charging condition of the battery, and Q (a) (mAh / g) is lithium metal deposited. It is the electric capacity per weight of the negative electrode active material which can occlude lithium to the maximum without any problem.
なお、Q(c)及びQ(a)は、正極または負極を作用極に、対極にリチウム金属を用い、上記の電池を組み立てるのに用いたのと同じ電解液を使用し、作用極と対極の間にセ
パレータを介した試験セルを作成して、可能な限り低い電流密度で初期充電条件(正極の上限電位あるいは負極の下限電位)まで、正極が充電(正極からのリチウムイオンの放出)できる容量をQ(c)、負極が放電(負極へのリチウムイオンの吸蔵)できる容量をQ(a)として求めた。
Note that Q (c) and Q (a) were used for the working electrode and the counter electrode using the same electrolyte solution used for assembling the battery, using the positive electrode or the negative electrode as the working electrode and lithium metal as the counter electrode. The positive electrode can be charged (release of lithium ions from the positive electrode) at the lowest possible current density up to the initial charging conditions (upper potential of the positive electrode or lower potential of the negative electrode) by creating a test cell with a separator between them. The capacity was determined as Q (c), and the capacity at which the negative electrode could discharge (occlude lithium ions in the negative electrode) was determined as Q (a).
(電池の評価)
(1)高温保存試験
得られたリチウム二次電池を室温下、1C(4.0mA)、4.2V上限の定電流定電圧法により充電し、電流値が0.05mAになった時点で充電を終了した。次いで、0.2Cで3.0Vまで放電した。
ここで、1Cとは1時間で満充電できる電流値を表し、本実施例及び比較例で用いる二次電池においては、1C=4.0mAである。従って、0.2Cは0.8mAとなる。
次いで、室温下、1C、4.2V上限の定電流定電圧法により充電し、電流値が0.05mAとなった時点で充電を終了した。この充電された電池を60℃で7日間保持したのち、室温まで冷却して放電容量を測定した。高温保存後の放電容量は数値が大きいほど高温保存における劣化が小さく、熱安定性が高いことを表す。
(Evaluation of battery)
(1) High-temperature storage test The obtained lithium secondary battery was charged at room temperature by 1C (4.0 mA) and a constant current constant voltage method with an upper limit of 4.2 V, and was charged when the current value reached 0.05 mA. Finished. Then, the battery was discharged at 3.0 C to 3.0 V.
Here, 1C represents a current value that can be fully charged in one hour, and 1C = 4.0 mA in the secondary batteries used in the present example and the comparative example. Therefore, 0.2C becomes 0.8mA.
Next, the battery was charged at room temperature by a constant current and constant voltage method having an upper limit of 1 C and 4.2 V, and the charging was terminated when the current value reached 0.05 mA. After maintaining the charged battery at 60 ° C. for 7 days, the battery was cooled to room temperature and the discharge capacity was measured. The larger the value of the discharge capacity after high-temperature storage, the smaller the deterioration in high-temperature storage and the higher the thermal stability.
(2)高温連続充電試験
得られたリチウム二次電池を、1C、4.2V上限の定電流定電圧法により充電し、電流値が0.05mAとなった時点で充電を終了した。次いで、0.2Cの定電流で3.0Vまで放電した。さらに、室温下、1C、4.2V上限の定電流定電圧法により充電し、電流値が0.05mAとなった時点で充電を終了した。この充電した電池に、60℃で7日間、4.2Vの定電圧充電(高温連続充電)を行い、充電容量を測定した。また、充電終了後、室温まで冷却して放電容量を測定した。
(2) High-Temperature Continuous Charging Test The obtained lithium secondary battery was charged by a constant current / constant voltage method with an upper limit of 1 C and 4.2 V, and the charging was terminated when the current value reached 0.05 mA. Next, the battery was discharged to 3.0 V at a constant current of 0.2 C. Further, the battery was charged at room temperature by a constant current and constant voltage method having an upper limit of 1 C and 4.2 V, and the charging was terminated when the current value reached 0.05 mA. The charged battery was charged at a constant voltage of 4.2 V (continuous high-temperature charging) at 60 ° C. for 7 days, and the charging capacity was measured. After completion of charging, the battery was cooled to room temperature, and the discharge capacity was measured.
高温連続充電後の充電容量は、電解液の分解により低下した電圧を補うために充電される電流の量であり、この数値が小さい方が電解液の分解が抑制されていることを表す。また、高温連続充電後の放電容量は、数値が大きい方が、高温連続充電中の劣化が小さく、熱安定性が大きいことを表す。 The charge capacity after high-temperature continuous charging is the amount of current charged to compensate for the voltage reduced by the decomposition of the electrolyte, and a smaller value indicates that the decomposition of the electrolyte is suppressed. The larger the value of the discharge capacity after continuous high-temperature charging, the smaller the deterioration during continuous high-temperature charging and the higher the thermal stability.
(実施例1)
エチレンカーボネート(EC)とジエチルカーボネート(DEC)の体積比3:7の混合溶媒に、1モル/リットルの濃度となるように六フッ化リン酸リチウム(LiPF6)
を溶解させてベース電解液とし、これにジエチルホスフィン酸エチルを1重量%となるように加えて電解液とした。
得られた電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2、3に示す。
(Example 1)
Lithium hexafluorophosphate (LiPF 6 ) in a mixed solvent of ethylene carbonate (EC) and diethyl carbonate (DEC) having a volume ratio of 3: 7 so as to have a concentration of 1 mol / liter.
Was dissolved to obtain a base electrolyte solution, and ethyl diethylphosphinate was added to the base electrolyte solution to a concentration of 1% by weight to obtain an electrolyte solution.
Using the obtained electrolytic solution, a lithium secondary battery was manufactured, and a high-temperature storage test and a high-temperature continuous charging test were performed. The results are shown in Tables 1, 2, and 3.
(実施例2)
ベース電解液にジエチルホスフィン酸エチルを1重量%、ビニレンカーボネートを2重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(実施例3)
ベース電解液にジエチルホスフィン酸エチルを1重量%、ビニレンカーボネートを2重量%、シクロヘキシルベンゼンを2重量%となるように添加した電解液を用いてリチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(実施例4)
ベース電解液にジ−n−ブチルホスフィン酸−n−ブチルを1重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行
った。結果を表−1、2に示す。
(実施例5)
ベース電解液にジ−n−ブチルホスフィン酸−n−ブチルを1重量%、ビニレンカーボネートを2重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(Example 2)
A lithium secondary battery was prepared using an electrolyte solution in which ethyl diethylphosphinate was added to a base electrolyte solution at 1% by weight and vinylene carbonate at 2% by weight, and a high-temperature storage test and a high-temperature continuous charge test were performed. . The results are shown in Tables 1 and 2.
(Example 3)
A lithium secondary battery was prepared using an electrolyte solution containing 1% by weight of ethyl diethylphosphinate, 2% by weight of vinylene carbonate, and 2% by weight of cyclohexylbenzene in a base electrolyte solution. A continuous charging test was performed. The results are shown in Tables 1 and 2.
(Example 4)
A lithium secondary battery was prepared using an electrolytic solution obtained by adding 1% by weight of n-butyl di-n-butylphosphinate to a base electrolytic solution, and a high-temperature storage test and a high-temperature continuous charge test were performed. . The results are shown in Tables 1 and 2.
(Example 5)
A lithium secondary battery was prepared using an electrolyte solution in which 1% by weight of n-butyl di-n-butylphosphinate and 2% by weight of vinylene carbonate were added to a base electrolyte solution. A high temperature continuous charge test was performed. The results are shown in Tables 1 and 2.
(実施例6)
ベース電解液にn−ブチルメチルホスフィン酸メチルを1重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(実施例7)
ベース電解液にn−ブチルメチルホスフィン酸メチルを1重量%、ビニレンカーボネートを2重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(Example 6)
A lithium secondary battery was manufactured using an electrolyte solution in which methyl n-butylmethylphosphinate was added to a base electrolyte solution at 1% by weight, and a high-temperature storage test and a high-temperature continuous charge test were performed. The results are shown in Tables 1 and 2.
(Example 7)
A lithium secondary battery was prepared using an electrolyte solution in which 1% by weight of methyl n-butylmethylphosphinate and 2% by weight of vinylene carbonate were added to a base electrolyte solution, and a high-temperature storage test and a high-temperature continuous charge test were performed. Was done. The results are shown in Tables 1 and 2.
(実施例8)
ベース電解液にメチルフェニルホスフィン酸メチルを1重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(実施例9)
ベース電解液にメチルフェニルホスフィン酸メチルを1重量%、ビニレンカーボネートを2重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(Example 8)
A lithium secondary battery was manufactured using an electrolyte solution in which methyl methylphenylphosphinate was added to the base electrolyte solution so as to be 1% by weight, and a high-temperature storage test and a high-temperature continuous charge test were performed. The results are shown in Tables 1 and 2.
(Example 9)
A lithium secondary battery was prepared using an electrolyte solution in which 1% by weight of methyl methylphenylphosphinate and 2% by weight of vinylene carbonate were added to a base electrolyte solution, and a high-temperature storage test and a high-temperature continuous charge test were performed. Was. The results are shown in Tables 1 and 2.
(実施例10)
ベース電解液にジエチルホスフィン酸エチルを0.1重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験を行った。結果を表−3に示す。(実施例11)
ベース電解液にジエチルホスフィン酸エチルを0.25重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験を行った。結果を表−3に示す。
(実施例12)
ベース電解液にジエチルホスフィン酸エチルを0.5重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験を行った。結果を表−3に示す。(実施例13)
ベース電解液にジエチルホスフィン酸エチルを4重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験を行った。結果を表−3に示す。
(Example 10)
A lithium secondary battery was manufactured using an electrolyte solution in which ethyl diethylphosphinate was added to a base electrolyte solution in an amount of 0.1% by weight, and a high-temperature storage test was performed. Table 3 shows the results. (Example 11)
A lithium secondary battery was prepared using an electrolytic solution obtained by adding ethyl diethylphosphinate to the base electrolytic solution to a concentration of 0.25% by weight, and a high-temperature storage test was performed. Table 3 shows the results.
(Example 12)
A lithium secondary battery was manufactured using an electrolyte solution in which ethyl diethylphosphinate was added to the base electrolyte solution in an amount of 0.5% by weight, and a high-temperature storage test was performed. Table 3 shows the results. (Example 13)
A lithium secondary battery was manufactured using an electrolyte solution in which ethyl diethylphosphinate was added to a base electrolyte solution in an amount of 4% by weight, and a high-temperature storage test was performed. Table 3 shows the results.
(比較例1)
ベース電解液そのものを用いリチウム二次電池を作製し、高温保存試験と高温充電試験を行った。結果を表−1、2、3に示す。
(比較例2)
ベース電解液にビニレンカーボネートを2重量%となるように添加した電解液を用いてリチウム二次電池を作製し、高温保存試験と高温充電試験を行った。結果を表−1、2に示す。
(比較例3)
ベース電解液にビニレンカーボネートを2重量%、シクロヘキシルベンゼンを2重量%となるように添加した電解液を用いてリチウム二次電池を作製し、高温保存試験と高温連続充電試験を行った。結果を表−1、2に示す。
(比較例4)
ベース電解液にジエチルホスフィン酸エチルを5重量%となるように添加した電解液を用いて、リチウム二次電池を作製し、高温保存試験を行った。結果を表−3に示す。
(Comparative Example 1)
A lithium secondary battery was fabricated using the base electrolyte itself, and a high-temperature storage test and a high-temperature charge test were performed. The results are shown in Tables 1, 2, and 3.
(Comparative Example 2)
A lithium secondary battery was manufactured using an electrolyte solution in which vinylene carbonate was added to a base electrolyte solution to a concentration of 2% by weight, and a high-temperature storage test and a high-temperature charge test were performed. The results are shown in Tables 1 and 2.
(Comparative Example 3)
A lithium secondary battery was manufactured using an electrolyte solution in which vinylene carbonate was added to a base electrolyte solution at 2% by weight and cyclohexylbenzene at 2% by weight, and a high-temperature storage test and a high-temperature continuous charge test were performed. The results are shown in Tables 1 and 2.
(Comparative Example 4)
A lithium secondary battery was prepared using an electrolytic solution obtained by adding ethyl diethylphosphinate to 5 wt% to a base electrolytic solution, and a high-temperature storage test was performed. Table 3 shows the results.
表−1より、電解液中に少量のホスフィン酸エステルを含有させることにより、高温保存時の電池の劣化を抑制することができることがわかる。また、この劣化を抑制する効果は、公知の被膜形成剤(ビニレンカーボネート)や、過充電防止剤(シクロヘキシルベンゼン)を併用した場合でも発現することがわかる。 From Table 1, it can be seen that by adding a small amount of phosphinic acid ester to the electrolytic solution, deterioration of the battery during high-temperature storage can be suppressed. Further, it can be seen that the effect of suppressing this deterioration is exhibited even when a known film forming agent (vinylene carbonate) or an overcharge preventing agent (cyclohexylbenzene) is used in combination.
表−2より、電解液中にホスフィン酸エステルを少量含有させることにより、高温連続充電時の電解液の分解を抑制し、電池の劣化を防止することができることがわかる。また、公知の被膜形成剤や過充電防止剤(シクロヘキシルベンゼン)を併用した場合でも発現することがわかる。 From Table 2, it can be seen that by adding a small amount of phosphinic acid ester to the electrolytic solution, decomposition of the electrolytic solution during continuous high-temperature charging can be suppressed, and battery deterioration can be prevented. Further, it can be seen that the expression occurs even when a known film forming agent or an overcharge preventing agent (cyclohexylbenzene) is used in combination.
表−3より、電解液中のホスフィン酸エステルの濃度が0.01重量%以上、4.5重量%以下であれば、高温保存時の電池の劣化を抑制することができることがわかる。 Table 3 shows that when the concentration of the phosphinic acid ester in the electrolytic solution is 0.01% by weight or more and 4.5% by weight or less, deterioration of the battery during high-temperature storage can be suppressed.
本発明の二次電池用非水電解液は、高温連続充電時、高温保存時における分解が抑えられ、リチウム二次電池用電解液として、ノートパソコンや携帯電話などの携帯機器の二次電池用途に用いることができるので、その工業的価値は極めて大きい。 The non-aqueous electrolyte for a secondary battery according to the present invention is capable of suppressing decomposition during high-temperature continuous charging and high-temperature storage, and is used as an electrolyte for a lithium secondary battery in secondary batteries of portable devices such as notebook computers and mobile phones. Therefore, its industrial value is extremely large.
Claims (6)
The nonaqueous electrolyte secondary solution according to claim 4, wherein the negative electrode is mainly composed of a carbon material having a lattice plane (002 plane) d value of 0.335 to 0.340 nm in X-ray diffraction. battery.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2003361115A JP4407233B2 (en) | 2002-10-22 | 2003-10-21 | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the same |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002306901 | 2002-10-22 | ||
| JP2003136322 | 2003-05-14 | ||
| JP2003361115A JP4407233B2 (en) | 2002-10-22 | 2003-10-21 | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2004363077A true JP2004363077A (en) | 2004-12-24 |
| JP4407233B2 JP4407233B2 (en) | 2010-02-03 |
Family
ID=34068867
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2003361115A Expired - Lifetime JP4407233B2 (en) | 2002-10-22 | 2003-10-21 | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the same |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4407233B2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005099023A1 (en) * | 2004-04-07 | 2005-10-20 | Matsushita Electric Industrial Co., Ltd. | Nonaqueous electrolyte secondary battery |
| WO2005104289A1 (en) * | 2004-04-19 | 2005-11-03 | Bridgestone Corporation | Nonaqueous electrolyte solution for battery and nonaqueous electrolyte battery using same |
| WO2008123038A1 (en) | 2007-03-19 | 2008-10-16 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte solution and nonaqueous electrolyte battery |
| WO2008126800A1 (en) | 2007-04-05 | 2008-10-23 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for rechargeable battery, and rechargeable battery with nonaqueous electrolyte |
| JP2009266663A (en) * | 2008-04-25 | 2009-11-12 | Mitsui Chemicals Inc | Nonaqueous electrolyte and lithium secondary battery using the same |
| WO2011012343A1 (en) * | 2009-07-25 | 2011-02-03 | Evonik Degussa Gmbh | Coating method for producing electrodes for electrical energy stores |
-
2003
- 2003-10-21 JP JP2003361115A patent/JP4407233B2/en not_active Expired - Lifetime
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7468225B2 (en) | 2004-04-07 | 2008-12-23 | Panasonic Corporation | Non-aqueous electrolyte secondary battery |
| WO2005099023A1 (en) * | 2004-04-07 | 2005-10-20 | Matsushita Electric Industrial Co., Ltd. | Nonaqueous electrolyte secondary battery |
| US7947399B2 (en) | 2004-04-19 | 2011-05-24 | Bridgestone Corporation | Non-aqueous electrolyte for battery and non-aqueous electrolyte battery comprising the same |
| WO2005104289A1 (en) * | 2004-04-19 | 2005-11-03 | Bridgestone Corporation | Nonaqueous electrolyte solution for battery and nonaqueous electrolyte battery using same |
| WO2008123038A1 (en) | 2007-03-19 | 2008-10-16 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte solution and nonaqueous electrolyte battery |
| US8962192B2 (en) | 2007-03-19 | 2015-02-24 | Mitsubishi Chemical Corporation | Nonaqueous electrolytic solution and nonaqueous electrolyte battery |
| US8685562B2 (en) | 2007-03-19 | 2014-04-01 | Mitsubishi Chemical Corporation | Nonaqueous electrolytic solution and nonaqueous electrolyte battery |
| US8574757B2 (en) | 2007-03-19 | 2013-11-05 | Mitsubishi Chemical Corporation | Nonaqueous electrolytic solution and nonaqueous electrolyte battery |
| EP2237358A1 (en) | 2007-03-19 | 2010-10-06 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte battery |
| EP2378602A3 (en) * | 2007-04-05 | 2011-12-21 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for recherable battery, and recharchable battery with nonaqueous electrolyte |
| EP2940779A1 (en) | 2007-04-05 | 2015-11-04 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| EP2378602A2 (en) | 2007-04-05 | 2011-10-19 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for recherable battery, and recharchable battery with nonaqueous electrolyte |
| US11616253B2 (en) | 2007-04-05 | 2023-03-28 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| EP2418723A1 (en) | 2007-04-05 | 2012-02-15 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| EP2597717A1 (en) | 2007-04-05 | 2013-05-29 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| EP2144321A1 (en) * | 2007-04-05 | 2010-01-13 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for rechargeable battery, and rechargeable battery with nonaqueous electrolyte |
| US11367899B2 (en) | 2007-04-05 | 2022-06-21 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| WO2008126800A1 (en) | 2007-04-05 | 2008-10-23 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for rechargeable battery, and rechargeable battery with nonaqueous electrolyte |
| US9093716B2 (en) | 2007-04-05 | 2015-07-28 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| EP2144321A4 (en) * | 2007-04-05 | 2011-08-10 | Mitsubishi Chem Corp | Nonaqueous electrolyte for rechargeable battery, and rechargeable battery with nonaqueous electrolyte |
| US9281541B2 (en) | 2007-04-05 | 2016-03-08 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| US9853326B2 (en) | 2007-04-05 | 2017-12-26 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| US10468720B2 (en) | 2007-04-05 | 2019-11-05 | Mitsubishi Chemical Corporation | Nonaqueous electrolyte for secondary battery and nonaqueous-electrolyte secondary battery employing the same |
| JP2009266663A (en) * | 2008-04-25 | 2009-11-12 | Mitsui Chemicals Inc | Nonaqueous electrolyte and lithium secondary battery using the same |
| WO2011012343A1 (en) * | 2009-07-25 | 2011-02-03 | Evonik Degussa Gmbh | Coating method for producing electrodes for electrical energy stores |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4407233B2 (en) | 2010-02-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7169511B2 (en) | Nonaqueous electrolyte solution and nonaqueous electrolyte solution secondary battery employing the same | |
| JP4407205B2 (en) | Nonaqueous electrolyte for lithium secondary battery and lithium secondary battery using the same | |
| CN110176630B (en) | Electrolyte solution and electrochemical device using the same | |
| JP5699465B2 (en) | Non-aqueous electrolyte and lithium secondary battery using the same | |
| JP2019102459A (en) | Electrolyte solution for nonaqueous electrolyte solution battery, and nonaqueous electrolyte battery using the same | |
| WO2006137224A1 (en) | Nonaqueous electrolyte, and rechargeable battery with nonaqueous electrolyte | |
| WO2021017709A1 (en) | Electrolyte, and electrochemical device and electronic device comprising same | |
| JP2005251456A (en) | Nonaqueous electrolytic solution for lithium secondary battery, and lithium secondary battery using the same | |
| JP4483253B2 (en) | Positive electrode material for lithium secondary battery, positive electrode for lithium secondary battery, and lithium secondary battery | |
| JP4797403B2 (en) | Non-aqueous electrolyte secondary battery and electrolyte for non-aqueous electrolyte secondary battery | |
| JP2007019012A (en) | Nonaqueous electrolyte solution, nonaqueous electrolyte secondary battery | |
| JP7268796B2 (en) | lithium ion secondary battery | |
| JP7295232B2 (en) | electrochemical devices and electronic devices containing the same | |
| JP6755182B2 (en) | Lithium ion secondary battery | |
| CN109390629B (en) | Electrolyte and battery | |
| JP4586388B2 (en) | Nonaqueous electrolyte, lithium ion secondary battery, and fluorine-containing ester compound | |
| JP2004111359A (en) | Nonaqueous electrolytic solution secondary battery and nonaqueous electrolytic solution | |
| JP4407233B2 (en) | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the same | |
| JP2001126761A (en) | Nonaqueous electrolyte secondary battery | |
| JP5110057B2 (en) | Lithium secondary battery | |
| WO2023123464A1 (en) | Electrolyte solution, electrochemical device containing same, and electronic device | |
| JP2008041412A (en) | Non-aqueous electrolytic solution and non-aqueous electrolyte secondary battery using the same | |
| JP4407237B2 (en) | Non-aqueous electrolyte and non-aqueous electrolyte secondary battery using the same | |
| JP4581501B2 (en) | Non-aqueous electrolyte for secondary battery and non-aqueous electrolyte secondary battery using the same | |
| JP2009283473A5 (en) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20060808 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20090220 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20090224 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20090424 |
|
| RD05 | Notification of revocation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7425 Effective date: 20090424 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20091020 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20091102 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4407233 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20121120 Year of fee payment: 3 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20131120 Year of fee payment: 4 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313111 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313121 |
|
| R371 | Transfer withdrawn |
Free format text: JAPANESE INTERMEDIATE CODE: R371 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313121 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| EXPY | Cancellation because of completion of term |