FR2501201A2 - Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine - Google Patents
Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine Download PDFInfo
- Publication number
- FR2501201A2 FR2501201A2 FR8104478A FR8104478A FR2501201A2 FR 2501201 A2 FR2501201 A2 FR 2501201A2 FR 8104478 A FR8104478 A FR 8104478A FR 8104478 A FR8104478 A FR 8104478A FR 2501201 A2 FR2501201 A2 FR 2501201A2
- Authority
- FR
- France
- Prior art keywords
- radical
- sep
- butenyl
- benzophenone
- methyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
- A61K31/05—Phenols
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Le présent certificat d'addition a pour objet des dérivés benzylidéniques, leur préparation et leur application en thérapeutique.This certificate of addition relates to benzylidenic derivatives, their preparation and their therapeutic application.
Dans son brevet, auquel est rattaché le présent certificat d'addition, la Demanderesse a décrit des composés répondant à la formule dans laquelle
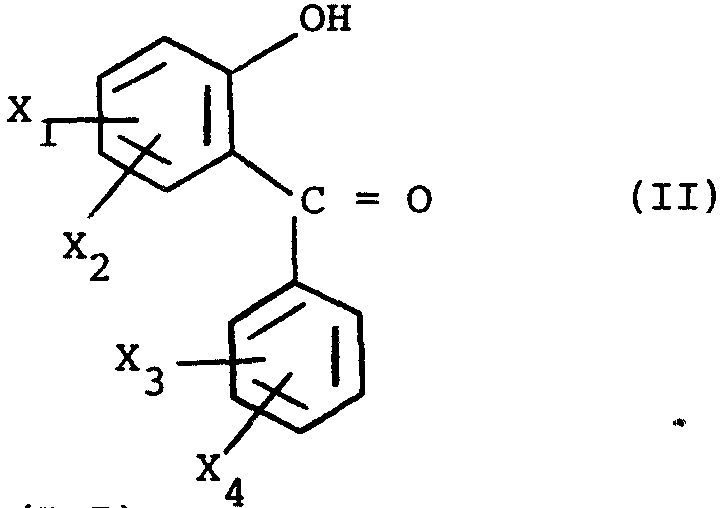
X1, X2, X3 et X4 reorésentent, indépendamment l'un de l'autre, un atome d'hydrogène, un atome d'halogène, un radical alkyle droit ou ramifié de 1 à 4 atomes de carbone, le radical CF3, le radical NO2, le radical phényle, le radical méthoxy ou le radical amino, deux des substituants X1, X2, X3 et X4 étant différents d'un atome d'hydrogène,
R représente un radical alkyle droit ou ramifié de 1 à 16 atomes ou un radical alcényle de 2 à 8 atomes de carbone, en particulier les composés comportant un radical R = alkyle.In her patent, to which this certificate of addition is attached, the Applicant has described compounds corresponding to the formula in which
X1, X2, X3 and X4 represent, independently of each other, a hydrogen atom, a halogen atom, a straight or branched alkyl radical of 1 to 4 carbon atoms, the radical CF 3, the radical NO2, the phenyl radical, the methoxy radical or the amino radical, two of the substituents X1, X2, X3 and X4 being different from a hydrogen atom,
R represents a straight or branched alkyl radical of 1 to 16 atoms or an alkenyl radical of 2 to 8 carbon atoms, in particular compounds comprising an R = alkyl radical.
Dans le présent certificat d'addition seront plus spécifique ment décrits les composés (I) pour lesquels R est un radical alcényle de 4 atomes de carbone.In this certificate of addition will be more specifically described the compounds (I) for which R is an alkenyl radical of 4 carbon atoms.
Parmi ces composés sont préférés ceux pour lesquels les radicaux X1, X2, X3 et X4 sont, indépendamment l'un de l'autre, un atome d'hydrogène ou d'halogène ou le radical méthyle et plus particulièrement ceux pour lesquels X2 = H, X1 est en position 4 et X3 et/ou X4 sont en oosition 2' et/ou 4'. Among these compounds are preferred those for which the radicals X1, X2, X3 and X4 are, independently of one another, a hydrogen or halogen atom or the methyl radical and more particularly those for which X2 = H X1 is in position 4 and X3 and / or X4 are in position 2 'and / or 4'.
Selon l'invention , on peut préparer les composés (I) selon le schéma réactionnel suivant
Schéma réactionnel
According to the invention, the compounds (I) can be prepared according to the following reaction scheme
Reaction scheme
Les composés (II) sont décrits par la Demanderesse dans ses précédents brevets.The compounds (II) are described by the Applicant in his previous patents.
Les composés (III) sont utilisées sous forme de base ou de chlorhydrate et sont décrits dans la littérature. Compounds (III) are used in base form or hydrochloride and are described in the literature.
La réaction est effectuée dans un solvant alcoolique, tel que le méthanol ou l'éthanol, à une température allant de 10 C à la température d'ébullition du solvant, en présence d'un métal alcalin ou d'un alcoolate de métal alcalin.The reaction is carried out in an alcoholic solvent, such as methanol or ethanol, at a temperature ranging from 10 ° C. to the boiling point of the solvent, in the presence of an alkali metal or an alkali metal alcoholate.
L'exemple suivant illustre l'invention. Les analyses et les spectres IR et RMN ont confirmé la structure des composés. The following example illustrates the invention. Analyzes and IR and NMR spectra confirmed the structure of the compounds.
EXEMPLE [(BUTENE -3 YL-IMINO)(CHLORO-2 PHENYL METHYL]-2
CHLORO-4 PHENOL.EXAMPLE [(BUTENE -3 YL-IMINO) (CHLORO-2-PHENYL METHYL) -2
CHLORO-4 PHENOL.
On introduit dans un ballon de 500 ml, 3,5gde (chloro-5 hydroxy-2 phényl) (chloro-2 phényl) méthanone en solution dans 50 mi de méthanol, et on ajoute 12 g. de butène-3yl-amine.3.5 g (5-chloro-2-hydroxyphenyl) (2-chlorophenyl) methanone dissolved in 50 ml of methanol are introduced into a 500 ml flask and 12 g are added. butene-3-yl amine.
On agite jusqu'à dispatirion complète de la cêtone. Ensuite-on évapore à siccité, on reprend le résidu au chloroforme. On lave la phase chloroformique à l'eau (plusieurs fois), on laisse décanter et on sèche sur MgSO4 ; on filtre sur fritté, puis évapore à sec le filtrat.It agitates until complete dispatirion of the ketone. Then it is evaporated to dryness, the residue is taken up in chloroform. The chloroform phase is washed with water (several times), decanted and dried over MgSO4; it is sintered and the filtrate is evaporated to dryness.
On fait cristalliser le produit dans de l'éther de pétrole, on entraine le précipité sur fritté, on l'essore, et on le sèche au dessicateur.The product is crystallized from petroleum ether, the precipitate is sintered, drained, and dried in a desiccator.
Le composé obtenu fond à 67-680C.The compound obtained melts at 67-680C.
Dans le tableau suivant sont représentés les composés préparés à titre d'exemples.In the following table are represented the compounds prepared as examples.
TABLEAU
BOARD
<tb> Composé <SEP> X1 <SEP> X2 <SEP> X3 <SEP> X4 <SEP> R <SEP> F(OC)
<tb> <SEP> ou <SEP> nD
<tb> <SEP> 1 <SEP> C1-5 <SEP> H <SEP> C1-4 <SEP> H <SEP> CH2-CH2-CH"CH2 <SEP> 67-68
<tb> <SEP> 2 <SEP> C1-5 <SEP> H <SEP> C1-4 <SEP> H <SEP> CH2-CH=CH-CH3 <SEP> 19
<tb> <SEP> nD <SEP> =1,6140
<tb> <SEP> 3 <SEP> Cl-S <SEP> H <SEP> Cl-4 <SEP> H <SEP> CH-F=CH2 <SEP> 47-48
<tb> <SEP> CH3
<tb>
Les composés de l'invention ont été soumis a des essais phar- macologiques montrant leur activité sur le système nerveux central.<tb> Compound <SEP> X1 <SEP> X2 <SEP> X3 <SEP> X4 <SEP> R <SEP> F (OC)
<tb><SEP> or <SEP> nD
<tb><SEP> 1 <SEP> C1-5 <SEP> H <SEP> C1-4 <SEP> H <SEP> CH2-CH2-CH "CH2 <SEP> 67-68
<tb><SEP> 2 <SEP> C1-5 <SEP> H <SEP> C1-4 <SEP> H <SEP> CH2-CH = CH-CH3 <SEP> 19
<tb><SEP> nD <SEP> = 1.6140
<tb><SEP> 3 <SEP> Cl-S <SEP> H <SEP> Cl-4 <SEP> H <SEP> CH-F = CH2 <SEP> 47-48
<tb><SEP> CH3
<Tb>
The compounds of the invention have been subjected to pharmacological tests showing their activity on the central nervous system.
La toxicité aiguë a été déterminée chez la souris par voie intrapéritonéale. La DL 50 (dose létale 50 induisant la mort chez 50% des animaux est supérieure a 3000 mg/kg.Acute toxicity was determined in the mouse intraperitoneally. The LD 50 (lethal dose 50 inducing death in 50% of the animals is greater than 3000 mg / kg.
L'activité des composés a été montrée par liantagonisme vis à vis de la mortalité induite par la bicuculline chez la souris.The activity of the compounds was shown by antagonagonism with respect to bicuculline-induced mortality in mice.
La bicuculline est un bloqueur relativement sélectif des récepteurs GABA-ergiques post-synaptiques et des effets convulsivants et létaux sont antagonisés par les composés élevant le taux de GABA cérébral ou possédant une activité GABA-mimutique.Bicuculline is a relatively selective blocker of post-synaptic GABA-ergic receptors and convulsive and lethal effects are antagonized by compounds elevating the levels of cerebral GABA or possessing GABA-mimetic activity.
On a évalué la dose active 50% (DA 50), dose protégeant 50% des animaux contre l'effet de la bicuculline, des substances étudiées.The active dose 50% (DA 50), a dose protecting 50% of animals against the effect of bicuculline, was evaluated for the substances studied.
La DA 50 des composés de l'invention varie de 20 à 80 mg/kg par voie intrapéritonéale. The DA 50 of the compounds of the invention ranges from 20 to 80 mg / kg intraperitoneally.
Les comnosés de l'invention sont actifs comme anticonvulsivants. Ils sont utilisables en thérapeutique humaine et vétérinaire pour le traitement de diverses maladies du système nerveux central, par exemple pour le traitement des psychoses et de certaines maladies neurologiques comme l'épilep- sie.The comnosés of the invention are active as anticonvulsants. They are useful in human and veterinary therapy for the treatment of various diseases of the central nervous system, for example for the treatment of psychoses and certain neurological diseases such as epilepsy.
L'invention comprend, par conséquent, toutes compositions pharmaceutiques renfermant les comoosés (I) comme orincipes actifs, en association avec tous excipients appropriés à leur administration, en particulier par voie orale (comprimés, dragées, gélules, capsules, cachets, solution ou suspensions buvables) ou parentérale. The invention therefore includes any pharmaceutical compositions containing comoose (I) as active ingredients, in combination with any excipients suitable for their administration, in particular orally (tablets, dragees, capsules, capsules, cachets, solutions or suspensions drinkable) or parenteral.
La posologie quotidienne peut aller de 100 à 1500 mg. The daily dosage can range from 100 to 1500 mg.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR8104478A FR2501201A2 (en) | 1981-03-06 | 1981-03-06 | Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR8104478A FR2501201A2 (en) | 1981-03-06 | 1981-03-06 | Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| FR2501201A2 true FR2501201A2 (en) | 1982-09-10 |
| FR2501201B2 FR2501201B2 (en) | 1984-02-24 |
Family
ID=9255931
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| FR8104478A Granted FR2501201A2 (en) | 1981-03-06 | 1981-03-06 | Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine |
Country Status (1)
| Country | Link |
|---|---|
| FR (1) | FR2501201A2 (en) |
-
1981
- 1981-03-06 FR FR8104478A patent/FR2501201A2/en active Granted
Also Published As
| Publication number | Publication date |
|---|---|
| FR2501201B2 (en) | 1984-02-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0015038B1 (en) | Pharmaceutical composition containing a diphenylhydantoin derivative, derivatives used and their preparation | |
| CH631162A5 (en) | Pyrrolidine derivatives, method of preparation and therapeutic use. | |
| EP0007824B1 (en) | Benzofurane derivatives, process for their preparation and therapeutic composition containing them. | |
| FR2637596A1 (en) | METHYL-4 ((ARYL-4-PIPERAZINYL-1) -2-ETHYL) -5 THIAZOLE AND ITS DERIVATIVES, PROCESS FOR PREPARING THEM AND MEDICAMENTS CONTAINING THE SAME | |
| EP0426562B1 (en) | 1-(4-Aminophenyl)-2-piperidinopropanone derivatives, process for their preparation and their therapeutic use | |
| CA1091233A (en) | Process for the preparation of indole substitutes | |
| FR2501201A2 (en) | Anticonvulsant butenyl benzophenone ketimine(s) - prepd. from 2-hydroxy benzophenone and butenyl amine | |
| LU85105A1 (en) | SULFUR BENZYLIDENIC DERIVATIVES, THEIR PREPARATION AND THEIR THERAPEUTIC APPLICATION | |
| EP0183577B1 (en) | Thiadiazole derivatives active on the central nervous system, process for their preparation and pharmaceutical compositions containing them | |
| FR2570695A1 (en) | Diphenylazomethines containing a branched or cyclic chain, their preparation and their application in therapy | |
| CH645096A5 (en) | BENZYLIDENIC DERIVATIVES, THEIR PREPARATION AND PHARMACEUTICAL COMPOSITION CONTAINING THEM. | |
| CH497433A (en) | (A) Benzimidazole derivs. of general formula (I): R = H, halogen, trifluoromethyl, alkyl (C1-C4) or alkoxy (C1-C4); R1 and R2 are same or different haloge | |
| EP0347305B1 (en) | [(Aryl-4-piperazinyl-1)-2-ethoxy]-3 p cymene, the ortho-, meta-, para-monosubstituted or disubstituted phenyl ring derivatives, process for their preparation and medicaments containing the same as the active principle | |
| CH620211A5 (en) | ||
| CA1263391A (en) | N-carboxyalcoyl-2 oxo-3 diaryl 5-6 triazines useful in therapeutics | |
| EP0038731A2 (en) | Thiazole derivatives, their preparation and use in therapeutics | |
| FR2544309A1 (en) | HYDROXYLATED DIPHENYLAZOMETHINES, THEIR PREPARATION AND THEIR THERAPEUTIC APPLICATION | |
| EP0092450A1 (en) | Tryptamine derivatives active on the cardiovascular system, and process for their preparation | |
| EP0123605A1 (en) | N-Cyclopropylmethyl-2-oxo-3-diparamethoxyphenyl-5,6-triazines, process for its preparation and their use as pharmaceutical preparations | |
| CH659649A5 (en) | SUBSTITUTED DIPHENYLAZOMETHINES, THEIR PREPARATION AND MEDICAMENTS CONTAINING THEM. | |
| FR2692894A1 (en) | Aryl-1- (o-alkoxy-phenyl-4-piperazinyl-1) -2,3-or 4-alkanols, process for their preparation and their use in the preparation of medicaments. | |
| LU81146A1 (en) | BENZYLIDENIC ESTERS AND THEIR THERAPEUTIC APPLICATION | |
| EP0226475A1 (en) | Diphenoxyethyl amine derivatives, process for their preparation and pharmaceutical compositions containing them | |
| FR2484410A2 (en) | Alpha-phenyl benzylidenyl aminoacid derivs. - which are anticonvulsants and centrally active, prepd. from di:phenyl methanone and aminoacid | |
| JPH06100509A (en) | Cyclopentenone compound and cerebral function improver containing the same as active ingredient |