EP4214297B1 - Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines - Google Patents
Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines Download PDFInfo
- Publication number
- EP4214297B1 EP4214297B1 EP21773883.0A EP21773883A EP4214297B1 EP 4214297 B1 EP4214297 B1 EP 4214297B1 EP 21773883 A EP21773883 A EP 21773883A EP 4214297 B1 EP4214297 B1 EP 4214297B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- amine
- ethyl
- hydrocarbyl
- fuel
- carbon atoms
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- -1 Aryloxy alkylamines Chemical class 0.000 title claims description 62
- 239000003502 gasoline Substances 0.000 title claims description 22
- 239000002816 fuel additive Substances 0.000 title claims description 20
- 238000002347 injection Methods 0.000 title claims description 11
- 239000007924 injection Substances 0.000 title claims description 11
- 239000000203 mixture Substances 0.000 claims description 78
- 239000000446 fuel Substances 0.000 claims description 53
- 150000001412 amines Chemical class 0.000 claims description 33
- 125000004432 carbon atom Chemical group C* 0.000 claims description 31
- 239000000654 additive Substances 0.000 claims description 27
- 239000003599 detergent Substances 0.000 claims description 21
- 238000000034 method Methods 0.000 claims description 21
- 125000001183 hydrocarbyl group Chemical group 0.000 claims description 20
- 125000000217 alkyl group Chemical group 0.000 claims description 19
- 239000001257 hydrogen Substances 0.000 claims description 17
- 229910052739 hydrogen Inorganic materials 0.000 claims description 17
- 230000000996 additive effect Effects 0.000 claims description 16
- KZNICNPSHKQLFF-UHFFFAOYSA-N succinimide Chemical class O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 claims description 14
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 12
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 11
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 11
- 239000012141 concentrate Substances 0.000 claims description 10
- 238000009835 boiling Methods 0.000 claims description 9
- 238000006683 Mannich reaction Methods 0.000 claims description 7
- 239000007795 chemical reaction product Substances 0.000 claims description 7
- 125000003342 alkenyl group Chemical group 0.000 claims description 5
- 239000006079 antiknock agent Substances 0.000 claims description 3
- 239000003963 antioxidant agent Substances 0.000 claims description 3
- 239000002270 dispersing agent Substances 0.000 claims description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 3
- 239000006078 metal deactivator Substances 0.000 claims description 3
- 239000003960 organic solvent Substances 0.000 claims description 3
- 125000000816 ethylene group Chemical group [H]C([H])([*:1])C([H])([H])[*:2] 0.000 claims 3
- 229960002317 succinimide Drugs 0.000 claims 3
- 238000012360 testing method Methods 0.000 description 24
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 18
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 16
- 229930195733 hydrocarbon Natural products 0.000 description 14
- ZSIAUFGUXNUGDI-UHFFFAOYSA-N hexan-1-ol Chemical compound CCCCCCO ZSIAUFGUXNUGDI-UHFFFAOYSA-N 0.000 description 12
- 239000013618 particulate matter Substances 0.000 description 11
- 229910052757 nitrogen Inorganic materials 0.000 description 10
- 239000003921 oil Substances 0.000 description 10
- 238000010992 reflux Methods 0.000 description 10
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 9
- 229920000768 polyamine Polymers 0.000 description 9
- 239000004215 Carbon black (E152) Substances 0.000 description 8
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 8
- 150000002430 hydrocarbons Chemical class 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 239000012043 crude product Substances 0.000 description 7
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- 125000003118 aryl group Chemical group 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- IZXIZTKNFFYFOF-UHFFFAOYSA-N 2-Oxazolidone Chemical compound O=C1NCCO1 IZXIZTKNFFYFOF-UHFFFAOYSA-N 0.000 description 5
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 5
- 239000003849 aromatic solvent Substances 0.000 description 5
- 238000002485 combustion reaction Methods 0.000 description 5
- 239000012530 fluid Substances 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 238000003756 stirring Methods 0.000 description 5
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 4
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 4
- 125000001931 aliphatic group Chemical group 0.000 description 4
- 125000005037 alkyl phenyl group Chemical group 0.000 description 4
- 239000012267 brine Substances 0.000 description 4
- 150000001875 compounds Chemical class 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 4
- 239000012299 nitrogen atmosphere Substances 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000011541 reaction mixture Substances 0.000 description 4
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 3
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 3
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 150000002431 hydrogen Chemical class 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- IEMXKEMFCFILAA-UHFFFAOYSA-N 2-docosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCCCC1=CC=CC=C1O IEMXKEMFCFILAA-UHFFFAOYSA-N 0.000 description 2
- QEMHBAGGYKJNSS-UHFFFAOYSA-N 2-icosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCC1=CC=CC=C1O QEMHBAGGYKJNSS-UHFFFAOYSA-N 0.000 description 2
- WLMABZIWETWROH-UHFFFAOYSA-N 2-tetracosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCC1=CC=CC=C1O WLMABZIWETWROH-UHFFFAOYSA-N 0.000 description 2
- XCSZUNWWLUTCFZ-UHFFFAOYSA-N 4-docosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCCCC1=CC=C(O)C=C1 XCSZUNWWLUTCFZ-UHFFFAOYSA-N 0.000 description 2
- HMZATDUMBFNSAM-UHFFFAOYSA-N 4-icosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCC1=CC=C(O)C=C1 HMZATDUMBFNSAM-UHFFFAOYSA-N 0.000 description 2
- IGFHQQFPSIBGKE-UHFFFAOYSA-N 4-nonylphenol Chemical compound CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 2
- GDGDNECAKIDBAP-UHFFFAOYSA-N 4-tetracosylphenol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCC1=CC=C(O)C=C1 GDGDNECAKIDBAP-UHFFFAOYSA-N 0.000 description 2
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- OIFBSDVPJOWBCH-UHFFFAOYSA-N Diethyl carbonate Chemical compound CCOC(=O)OCC OIFBSDVPJOWBCH-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 2
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical group CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 150000001336 alkenes Chemical class 0.000 description 2
- IVHKZGYFKJRXBD-UHFFFAOYSA-N amino carbamate Chemical compound NOC(N)=O IVHKZGYFKJRXBD-UHFFFAOYSA-N 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 239000002283 diesel fuel Substances 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000012074 organic phase Substances 0.000 description 2
- 125000005702 oxyalkylene group Chemical group 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 239000000376 reactant Substances 0.000 description 2
- 238000001308 synthesis method Methods 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- VILCJCGEZXAXTO-UHFFFAOYSA-N 2,2,2-tetramine Chemical compound NCCNCCNCCN VILCJCGEZXAXTO-UHFFFAOYSA-N 0.000 description 1
- FALRKNHUBBKYCC-UHFFFAOYSA-N 2-(chloromethyl)pyridine-3-carbonitrile Chemical class ClCC1=NC=CC=C1C#N FALRKNHUBBKYCC-UHFFFAOYSA-N 0.000 description 1
- QSKPIOLLBIHNAC-UHFFFAOYSA-N 2-chloro-acetaldehyde Chemical compound ClCC=O QSKPIOLLBIHNAC-UHFFFAOYSA-N 0.000 description 1
- KJWMCPYEODZESQ-UHFFFAOYSA-N 4-Dodecylphenol Chemical compound CCCCCCCCCCCCC1=CC=C(O)C=C1 KJWMCPYEODZESQ-UHFFFAOYSA-N 0.000 description 1
- ALYNCZNDIQEVRV-UHFFFAOYSA-N 4-aminobenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1 ALYNCZNDIQEVRV-UHFFFAOYSA-N 0.000 description 1
- WVYWICLMDOOCFB-UHFFFAOYSA-N 4-methyl-2-pentanol Chemical compound CC(C)CC(C)O WVYWICLMDOOCFB-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical class ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 239000004952 Polyamide Substances 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004721 Polyphenylene oxide Substances 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N Propene Chemical compound CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001414 amino alcohols Chemical class 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- CREXVNNSNOKDHW-UHFFFAOYSA-N azaniumylideneazanide Chemical group N[N] CREXVNNSNOKDHW-UHFFFAOYSA-N 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 150000002462 imidazolines Chemical class 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- WSFSSNUMVMOOMR-NJFSPNSNSA-N methanone Chemical compound O=[14CH2] WSFSSNUMVMOOMR-NJFSPNSNSA-N 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N octane Chemical compound CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 229920002866 paraformaldehyde Polymers 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 229920013639 polyalphaolefin Polymers 0.000 description 1
- 229920002647 polyamide Polymers 0.000 description 1
- 229920000570 polyether Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000004071 soot Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 229940014800 succinic anhydride Drugs 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 229960001124 trientine Drugs 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/2222—(cyclo)aliphatic amines; polyamines (no macromolecular substituent 30C); quaternair ammonium compounds; carbamates
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/02—Use of additives to fuels or fires for particular purposes for reducing smoke development
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/04—Use of additives to fuels or fires for particular purposes for minimising corrosion or incrustation
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/223—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond having at least one amino group bound to an aromatic carbon atom
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/222—Organic compounds containing nitrogen containing at least one carbon-to-nitrogen single bond
- C10L1/224—Amides; Imides carboxylic acid amides, imides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/23—Organic compounds containing nitrogen containing at least one nitrogen-to-oxygen bond, e.g. nitro-compounds, nitrates, nitrites
- C10L1/231—Organic compounds containing nitrogen containing at least one nitrogen-to-oxygen bond, e.g. nitro-compounds, nitrates, nitrites nitro compounds; nitrates; nitrites
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/234—Macromolecular compounds
- C10L1/238—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds
- C10L1/2383—Polyamines or polyimines, or derivatives thereof (poly)amines and imines; derivatives thereof (substituted by a macromolecular group containing 30C)
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/22—Organic compounds containing nitrogen
- C10L1/234—Macromolecular compounds
- C10L1/238—Macromolecular compounds obtained otherwise than by reactions involving only carbon-to-carbon unsaturated bonds
- C10L1/2383—Polyamines or polyimines, or derivatives thereof (poly)amines and imines; derivatives thereof (substituted by a macromolecular group containing 30C)
- C10L1/2387—Polyoxyalkyleneamines (poly)oxyalkylene amines and derivatives thereof (substituted by a macromolecular group containing 30C)
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L2200/00—Components of fuel compositions
- C10L2200/04—Organic compounds
- C10L2200/0407—Specifically defined hydrocarbon fractions as obtained from, e.g. a distillation column
- C10L2200/0415—Light distillates, e.g. LPG, naphtha
- C10L2200/0423—Gasoline
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L2230/00—Function and purpose of a components of a fuel or the composition as a whole
- C10L2230/08—Inhibitors
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G, C10K; LIQUEFIED PETROLEUM GAS; ADDING MATERIALS TO FUELS OR FIRES TO REDUCE SMOKE OR UNDESIRABLE DEPOSITS OR TO FACILITATE SOOT REMOVAL; FIRELIGHTERS
- C10L2270/00—Specifically adapted fuels
- C10L2270/02—Specifically adapted fuels for internal combustion engines
- C10L2270/023—Specifically adapted fuels for internal combustion engines for gasoline engines
Definitions
- This disclosure relates to fuel additives and fuel compositions containing the same. More specifically, this disclosure describes compositions and methods for controlling deposit formation in direct injection spark ignition gasoline engines.
- PFI port fuel injection
- DISI direct injection spark ignition
- DIG direct injection gasoline
- GDI gasoline direct injection
- injector fouling Traditional fuel additives developed for port fuel injection (PFI) gasoline engines are generally not optimized for controlling the formation of deposits in injectors of direct injection spark ignition (DISI) engines, sometimes referred to as direct injection gasoline (DIG) or gasoline direct injection (GDI) engines. This is largely since unlike PFI engines, DISI engines deliver fuel directly into the combustion chamber. When fuel is directly injected, it is immediately exposed to high temperatures and pressures. In this environment, combustion products can accumulate on the external and/or internal surfaces of the injectors and nozzle (known as injector fouling).
- a method of reducing injector fouling in a direct injection spark ignition gasoline engine comprising providing a gasoline composition comprising: aryloxy alkylamine additive having the following structure wherein the aryloxy alkylamine additive is present in about 10 to about 750 ppm by weight based on total weight of the fuel composition; wherein X is a hydrocarbyl group having 1 or 2 carbon atoms; and wherein R 1 and R 2 are independently hydrogen, alkyl or alkenyl group having up to 36 carbon atoms.
- the present invention describes compositions and methods for deposit control in direct injection engines. More specifically, the present invention provides detergent additive compositions that can be utilized as components of fuel compositions and methods of using the compositions thereof.
- Gasoline fuel refers to a composition containing at least predominantly C 4 -C 12 hydrocarbons.
- gasoline or gasoline boiling range components is further defined to refer to a composition containing at least predominantly C 4 -C 12 hydrocarbons and further having a boiling range of from about 37.8°C (100°F) to about 204°C (400°F).
- gasoline is defined to refer to a composition containing at least predominantly C 4 -C 12 hydrocarbons, having a boiling range of from about 37.8°C (100°F) to about 204°C (400°F), and further defined to meet ASTM D4814.
- the hydrocarbon-based fuel is present in a major amount by weight % of the total fuel composition.
- the hydrocarbon-based fuel is present in about 50 wt% or greater, 55 wt% or greater, 60 wt% or greater, 65 wt% or greater, 70 wt% or greater, 75 wt% or greater, 80 wt% or greater, 85 wt% or greater, 90 wt% or greater, 95 wt% or greater or between any range from about 50 wt% to up to below 100 wt%.
- the gasoline employed in the present invention may be clean burning gasoline (CBG).
- CBG refers to gasoline formulations that contain reduced levels of sulfur, aromatics and olefins. The exact formulation may vary depending on local regulatory definitions.
- the carrier fluids may be employed in amounts ranging from 35 to 5000 ppm by weight of the hydrocarbon fuel (e.g., 50 to 3000 ppm of the fuel). When employed in a fuel concentrate, carrier fluids may be present in amounts ranging from 20 to 60 wt % (e.g., 30 to 50 wt %).
- the aryloxy alkylamine fuel additive of the present invention reduces injector fouling in a direct injection spark ignition gasoline engine.
- the additive is a nitrogen-containing detergent having the following formula: wherein R 1 and R 2 are independently hydrogen, or a hydrocarbyl group having up to 36 carbon atoms. R 2 can be positioned at the ortho- or meta- position with respect to the oxygen atom.
- X is a hydrocarbyl group having 1 or 2 carbon atoms. X may be saturated or unsaturated. In some embodiments, it may be preferable for one of R 1 or R 2 to be a hydrocarbyl group and the other to be a hydrogen.
- the fuel additives of the present invention may be synthesized by any known compatible method. Description of two known synthesis methods are described herein.
- Method B For illustrative purposes, the following example of Method B is provided.
- Suitable secondary fuel additives may be classified as aliphatic hydrocarbyl-substituted amines, hydrocarbyl-substituted poly(oxyalkylene)amines, hydrocarbyl-substituted succinimides, Mannich reaction products, polyalkylphenoxyaminoalkanes, nitro and amino aromatic esters of polyalkylphenoxyalkanols, and nitrogen-containing carburetor/injector detergents.
- Each class of secondary fuel additive will be described in more detail herein.
- hydrocarbyl-substituted poly(oxyalkylene)amines employed in the present invention may include hydrocarbyl poly(oxyalkylene)amines (monoamines or polyamines) wherein the hydrocarbyl group contains from about 1 to about 30 carbon atoms.
- the number of oxyalkylene units can range from about 5 to about 100.
- the amine moiety is derived from ammonia, primary alkyl or secondary dialkyl monoamine, or polyamine having a terminal amino nitrogen atom.
- the oxyalkylene moiety may be oxypropylene or oxybutylene or a mixture thereof.
- Hydrocarbyl-substituted poly(oxyalkylene)amines are described in U.S. Pat. No. 6,217,624 , and U.S. Pat. No. 5,112,364 .
- Specific examples of hydrocarbyl-substituted poly(oxyalkylene)monoamine include alkylphenyl poly(oxyalkylene)monoamine wherein the poly(oxyalkylene) moiety contains oxypropylene units or oxybutylene units or mixtures of oxypropylene and oxybutylene units.
- the alkyl group on the alkylphenyl moiety is a straight or branched-chain alkyl of about 1 to about 24 carbon atoms.
- a preferred alkylphenyl moiety is tetrapropenylphenyl where the alkyl group is a branched-chain alkyl of 12 carbon atoms derived from a propylene tetramer.
- the hydrocarbyl-substituted succinimides employed in the present invention include polyalkyl and polyalkenyl succinimides wherein the polyalkyl or polyalkenyl group has an average molecular weight of about 500 to 5,000, and preferably about 700 to 3,000.
- the hydrocarbyl-substituted succinimides are typically prepared by reacting a hydrocarbyl-substituted succinic anhydride with an amine or polyamine having at least one reactive hydrogen bonded to an amine nitrogen atom.
- Preferred hydrocarbyl-substituted succinimides include polyisobutenyl and polyisobutanyl succinimides, and derivatives thereof.
- each of the foregoing additives when used, is used at a functionally effective amount to impart the desired properties to the fuel composition.
- concentration of each of these additives, when used may range, unless otherwise specified, from about 0.001 to about 20 wt. %, such as about 0.01 to about 10 wt. %.
- the mixture was allowed to cool to room temperature, diluted with hexanes (200 mL) and the organic phase was washed with water (200 mL), brine (4 x 100 mL), dried over anhydrous MgSO 4 , and filtered through a pad of Celite filter aid. The filtrate was concentrated under reduced pressure and then under high vacuum to give a dark colored oil as crude product (150.2 g).
- the crude product was purified by column chromatography using a mixture of ethyl acetate/methanol gradient to give a pale yellow oil (105.8 g).
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Combustion & Propulsion (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Solid Fuels And Fuel-Associated Substances (AREA)
- Liquid Carbonaceous Fuels (AREA)
- Fuel-Injection Apparatus (AREA)
Description
- This disclosure relates to fuel additives and fuel compositions containing the same. More specifically, this disclosure describes compositions and methods for controlling deposit formation in direct injection spark ignition gasoline engines.
- Traditional fuel additives developed for port fuel injection (PFI) gasoline engines are generally not optimized for controlling the formation of deposits in injectors of direct injection spark ignition (DISI) engines, sometimes referred to as direct injection gasoline (DIG) or gasoline direct injection (GDI) engines. This is largely since unlike PFI engines, DISI engines deliver fuel directly into the combustion chamber. When fuel is directly injected, it is immediately exposed to high temperatures and pressures. In this environment, combustion products can accumulate on the external and/or internal surfaces of the injectors and nozzle (known as injector fouling).
- The formation of deposits, both around the injector nozzle and inside the combustion chamber, can have significant negative impact on one or more of fuel flow rate, injection duration, and/or spray pattern. This, in turn, can lead to increased emission, increased particulate matter (PM) formation, reduced fuel economy, loss of power/performance, increased wear, and/or reduced equipment life.
- In one aspect, there is provided a fuel composition comprising gasoline; and aryloxy alkylamine additive having the following structure:
- In another aspect, there is provided a concentrate composition comprising about 10 to 90 wt % of an organic solvent boiling in a range of from 65°C to 205°C and about 10 to 70 wt % of a fuel additive comprising: an aryloxy alkylamine given by formula
- In yet another aspect, there is provided a method of reducing injector fouling in a direct injection spark ignition gasoline engine comprising providing a gasoline composition comprising: aryloxy alkylamine additive having the following structure
-
-
FIG. 1 shows a graph described in the Examples. - FIGS. 2A-2C show photographs described in the Examples.
- The present invention describes compositions and methods for deposit control in direct injection engines. More specifically, the present invention provides detergent additive compositions that can be utilized as components of fuel compositions and methods of using the compositions thereof.
- The fuel composition of the present invention comprises (i) a hydrocarbon-based fuel; and (ii) an aryloxy alkylamine fuel additive. In some embodiments, the fuel composition may comprise a secondary fuel additive.
- The hydrocarbon-based fuel includes gasoline and diesel.
- Gasoline fuel refers to a composition containing at least predominantly C4-C12 hydrocarbons. In one embodiment, gasoline or gasoline boiling range components is further defined to refer to a composition containing at least predominantly C4-C12 hydrocarbons and further having a boiling range of from about 37.8°C (100°F) to about 204°C (400°F). In an alternative embodiment, gasoline is defined to refer to a composition containing at least predominantly C4-C12 hydrocarbons, having a boiling range of from about 37.8°C (100°F) to about 204°C (400°F), and further defined to meet ASTM D4814.
- Diesel fuel refers to middle distillate fuels containing at least predominantly C10-C25 hydrocarbons. In one embodiment, diesel is further defined to refer to a composition containing at least predominantly C10-C25 hydrocarbons, and further having a boiling range of from about 165.6°C (330°F) to about 371.1°C (700°F). In an alternative embodiment, diesel is as defined above to refer to a composition containing at least predominantly C10-C25 hydrocarbons, having a boiling range of from about 165.6°C (330°F) to about 371.1°C (700°F), and further defined to meet ASTM D975.
- The hydrocarbon-based fuel is present in a major amount by weight % of the total fuel composition. In some embodiments, the hydrocarbon-based fuel is present in about 50 wt% or greater, 55 wt% or greater, 60 wt% or greater, 65 wt% or greater, 70 wt% or greater, 75 wt% or greater, 80 wt% or greater, 85 wt% or greater, 90 wt% or greater, 95 wt% or greater or between any range from about 50 wt% to up to below 100 wt%.
- According to some embodiments, the gasoline employed in the present invention may be clean burning gasoline (CBG). CBG refers to gasoline formulations that contain reduced levels of sulfur, aromatics and olefins. The exact formulation may vary depending on local regulatory definitions.
- A fuel-soluble, non-volatile carrier fluid or oil may also be used with compounds of this disclosure. The carrier fluid is a chemically inert hydrocarbon-soluble liquid vehicle which substantially increases the non-volatile residue (NVR), or solvent-free liquid fraction of the fuel additive composition while not overwhelmingly contributing to octane requirement increase. The carrier fluid may be a natural or synthetic oil, such as mineral oil, refined petroleum oils, synthetic polyalkanes and alkenes, including hydrogenated and unhydrogenated polyalphaolefins, synthetic polyoxyalkylene-derived oils, such as those described in
U.S. Patent Nos. 3,756,793 ;4,191,537 ; and5,004,478 ; and inEuropean Patent Appl. Pub. Nos. 356,726 382,159 - The carrier fluids may be employed in amounts ranging from 35 to 5000 ppm by weight of the hydrocarbon fuel (e.g., 50 to 3000 ppm of the fuel). When employed in a fuel concentrate, carrier fluids may be present in amounts ranging from 20 to 60 wt % (e.g., 30 to 50 wt %).
- The aryloxy alkylamine fuel additive of the present invention reduces injector fouling in a direct injection spark ignition gasoline engine. The additive is a nitrogen-containing detergent having the following formula:
- In some embodiments, the hydrocarbyl group is an alkyl group or an alkenyl group. Alkyl groups refer to saturated hydrocarbyl groups, which can be linear, branched, cyclic, or a combination of cyclic, linear and/or branched. Alkenyl groups refer to unsaturated hydrocarbyl groups, which can be linear, branched, cyclic, or a combination of cyclic, linear and/or branched.
- Suitable examples of aryloxy alkylamines include, but are not limited to, 2-(phenoxy)ethyl-1-amine, 2-(4-butylphenoxy)ethyl-1-amine, 2-(4-octylphenoxy)ethyl-1-amine, 2-(4-nonylphenoxy)ethyl-1-amine, 2-(4-dodecylphenoxy)ethyl-1-amine, 2-(4-octadecylphenoxy)ethyl-1-amine, 2-(4-eicosanylphenoxy)ethyl-1-ami ne, 2-(4-docosanylphenoxy)ethyl-1-amine, 2-(4-tetracosanylphenoxy)ethyl-1-amine.
- The aryloxy alkylamines may be obtained commercially or by any known compatible synthesis method with. For example, aryloxy alkylamines may be obtained by reacting salt of alkylphenol with a chloroacetaldehyde. The resulting product is then reacted with an amino alcohol and then hydrogenated in the presence of a nickel catalyst to generate the aryloxy alkylamine. A more detailed description of aryloxy alkylamine synthesis can be found in
U.S. Patent No. 3,954,872 . - In general, the fuel additives of the present invention may be synthesized by any known compatible method. Description of two known synthesis methods are described herein.
- In the first method (Method A), an alkylphenol is initially reacted with a base (e.g., potassium hydroxide) to form an alkylphenoxide said, which will further react with 2-oxazolidinone in an aromatic solvent under reflux conditions to provide the corresponding aminoethylated product.
-
- In the second method (Method B), an alkylphenol is also initially reacted with a base (e.g., potassium hydroxide) to form an alkylphenoxide, which will further react with 2-oxazolidinone generated in situ from the reaction between ethanolamine and diethyl carbonate in an aromatic solvent under reflux conditions to provide the corresponding aminoethylated product.
-
- For illustrative purposes, the following example of Method B is provided.
- A 1000 mL two-neck round bottom flask was charged with a mixture of 4-eicosanylphenol, 4-docosanylphenol, 4-tetracosanylphenol, 2-eicosanylphenol, 2-docosanylphenol, and 2-tetracosanylphenol with an averaged molecular weight of 402.71 g/mol (120 g, 0.298 mol, 1.00 equiv), KOH (2.96 g, 0.0447 mol, 0.150 equiv, 85% active), 150 mL of Aromatic 100 solvent equipped with mechanical stirrer, a Dean-Stark trap and a reflux condenser, and the reaction mixture was refluxed under vigorous stirring and a purging nitrogen atmosphere for 1 hour to remove water. The reaction mixture was cooled to about 120 °C, and ethanolamine (21.87 g, 0.358 mol, 1.20 equiv) and diethyl carbonate (42.3 g, 0.358 mol, 1.20 equiv) were added in that order. The reaction mixture was then warmed to 120 °C under gentle N2 flow until the theoretical amount of ethanol was evolved from the reaction, which was then warmed to 175 °C with vigorous stirring under a gentle flow of nitrogen for 19 hours. The crude reaction mixture was diluted with 250 mL of ethyl acetate and was washed with 3 x 200 mL of water and 200 mL of brine. The organic layer was dried with MgSO4, filtered, and concentrated to give crude product as an amber color oil (128.0 g). It was analyzed by NMR spectroscopy and HPLC.
- The fuel composition of the present invention includes one or more secondary fuel additives. The secondary fuel additive is a nitrogen-containing detergent that provides enhanced detergency when paired with the primary fuel additive of the present invention.
- Suitable secondary fuel additives may be classified as aliphatic hydrocarbyl-substituted amines, hydrocarbyl-substituted poly(oxyalkylene)amines, hydrocarbyl-substituted succinimides, Mannich reaction products, polyalkylphenoxyaminoalkanes, nitro and amino aromatic esters of polyalkylphenoxyalkanols, and nitrogen-containing carburetor/injector detergents. Each class of secondary fuel additive will be described in more detail herein.
- In particular, the aliphatic hydrocarbyl-substituted amines employed in the present invention may be straight or branched chain hydrocarbyl-substituted amines having at least one basic nitrogen and wherein the hydrocarbyl group has a number average molecular weight of about 700 to 3,000. Specific examples of aliphatic hydrocarbyl-substituted amines include polyisobutenyl amines and polyisobutyl amines. These amines can be derived as monoamines or polyamines. Preparation of aliphatic amines are generally known and described in detail in
U.S. Pat. Nos. 3,438,757 ;3,565,804 ;3,574,576 ;3,848,056 ;3,960,515 ;4,832,702 ; and6,203,584 . - In particular, the hydrocarbyl-substituted poly(oxyalkylene)amines employed in the present invention (also referred to as "polyether amines") may include hydrocarbyl poly(oxyalkylene)amines (monoamines or polyamines) wherein the hydrocarbyl group contains from about 1 to about 30 carbon atoms. The number of oxyalkylene units can range from about 5 to about 100. The amine moiety is derived from ammonia, primary alkyl or secondary dialkyl monoamine, or polyamine having a terminal amino nitrogen atom. The oxyalkylene moiety may be oxypropylene or oxybutylene or a mixture thereof. Hydrocarbyl-substituted poly(oxyalkylene)amines are described in
U.S. Pat. No. 6,217,624 , andU.S. Pat. No. 5,112,364 . Specific examples of hydrocarbyl-substituted poly(oxyalkylene)monoamine include alkylphenyl poly(oxyalkylene)monoamine wherein the poly(oxyalkylene) moiety contains oxypropylene units or oxybutylene units or mixtures of oxypropylene and oxybutylene units. The alkyl group on the alkylphenyl moiety is a straight or branched-chain alkyl of about 1 to about 24 carbon atoms. A preferred alkylphenyl moiety is tetrapropenylphenyl where the alkyl group is a branched-chain alkyl of 12 carbon atoms derived from a propylene tetramer. - More particularly, additional hydrocarbyl-substituted poly(oxyalkylene)amines include hydrocarbyl-substituted poly(oxyalkylene)aminocarbamates disclosed in
U.S. Pat. Nos. 4,288,612 ;4,236,020 ;4,160,648 ;4,191,537 ;4,270,930 ;4,233,168 ;4,197,409 ;4,243,798 and4,881,945 . These hydrocarbyl poly(oxyalkylene)aminocarbamates contain at least one basic nitrogen atom and have an average molecular weight of about 500 to 10,000, preferably about 500 to 5,000, and more preferably about 1,000 to 3,000. A preferred aminocarbamate is alkylphenyl poly(oxybutylene)aminocarbamate wherein the amine moiety is derived from ethylene diamine or diethylene triamine. - In particular, the hydrocarbyl-substituted succinimides employed in the present invention include polyalkyl and polyalkenyl succinimides wherein the polyalkyl or polyalkenyl group has an average molecular weight of about 500 to 5,000, and preferably about 700 to 3,000. The hydrocarbyl-substituted succinimides are typically prepared by reacting a hydrocarbyl-substituted succinic anhydride with an amine or polyamine having at least one reactive hydrogen bonded to an amine nitrogen atom. Preferred hydrocarbyl-substituted succinimides include polyisobutenyl and polyisobutanyl succinimides, and derivatives thereof. Hydrocarbyl-substituted succinimides are described in
U.S. Pat. Nos. 5,393,309 ;5,588,973 ;5,620,486 ;5,916,825 ;5,954,843 ;5,993,497 ; and6,114,542 , andBritish Patent No. 1,486,144 - In particular, the Mannich reaction products employed in the present invention include products typically obtained from Mannich condensation of a high molecular weight alkyl-substituted hydroxyaromatic compound, an amine containing at least one reactive hydrogen, and an aldehyde. The high molecular weight alkyl-substituted hydroxyaromatic compounds are preferably polyalkylphenols, such as polypropylphenol and polybutylphenol, especially polyisobutylphenol, wherein the polyalkyl group has an average molecular weight of about 600 to 3,000. The amine reactant is typically a polyamine, such as alkylene polyamines, especially ethylene or polyethylene polyamines, for example, ethylene diamine, diethylene triamine, triethylene tetramine, and the like. The aldehyde reactant is generally an aliphatic aldehyde, such as formaldehyde, including paraformaldehyde and formalin, and acetaldehyde. A preferred Mannich reaction product is obtained by condensing a polyisobutylphenol with formaldehyde and diethylene triamine, wherein the polyisobutyl group has an average molecular weight of about 1,000. The Mannich reaction products suitable for use in the present invention are described, for example, in
U.S. Pat. Nos. 4,231,759 and5,697,988 . - A still further class of detergent additive suitable for use in the present invention are polyalkylphenoxyaminoalkanes. Preferred polyalkylphenoxyaminoalkanes include those having the following formula:
U.S. Pat. No. 5,669,939 . - Certain detergent mixtures may be particularly useful as secondary additives in accordance with the present invention.
- In some embodiments, mixtures of polyalkylphenoxyaminoalkanes and poly(oxyalkylene)amines may be employed. These mixtures are described in detail in
U.S. Pat. No. 5,851,242 . - In some embodiments, mixtures of nitro and amino aromatic esters of polyalkylphenoxyalkanols may be employed. Preferred nitro and amino aromatic esters of polyalkylphenoxyalkanols include those having the formula:
U.S. Pat. No. 5,618,320 . - Mixtures of nitro and amino aromatic esters of polyalkylphenoxyalkanols and hydrocarbyl-substituted poly(oxyalkylene)amines may also be employed in the present invention. These mixtures are described in detail in
U.S. Pat. No. 5,749,929 . Preferred hydrocarbyl-substituted poly(oxyalkylene)amines which may be employed as detergent additives in the present invention include those having the following formula:U.S. Pat. No. 6,217,624 . The hydrocarbyl-substituted poly(oxyalkylene)amines of Formula IV are preferably utilized either by themselves or in combination with other detergent additives, particularly with the polyalkylphenoxyaminoalkanes or the nitro and amino aromatic esters of polyalkylphenoxyalkanols. More preferably, the detergent additives employed in the present invention will be combinations of the hydrocarbyl-substituted poly(oxyalkylene)amines with the nitro and amino aromatic esters of polyalkylphenoxyalkanols. A particularly preferred hydrocarbyl-substituted poly(oxyalkylene)amine detergent additive is dodecylphenoxy poly(oxybutylene)amine and a particularly preferred combination of detergent additives is the combination of dodecylphenoxy poly(oxybutylene)amine and 4-polyisobutylphenoxyethyl para-aminobenzoate. - Another class of detergent additive suitable for use in the present invention include nitrogen-containing carburetor/injector detergents. The carburetor/injector detergent additives are typically low molecular weight compounds having a number average molecular weight of about 100 to about 600 and possessing at least one polar moiety and at least one non-polar moiety. The non-polar moiety is typically a linear or branched-chain alkyl or alkenyl group having about 6 to about 40 carbon atoms. The polar moiety is typically nitrogen-containing. Typical nitrogen-containing polar moieties include amines (for example, as described in
U.S. Pat. No. 5,139,534 andPCT International Publication No. WO 90/10051 U.S. Pat. No. 3,849,083 andPCT International Publication No. WO 90/10051 U.S. Pat. Nos. 2,622,018 ;4,729,769 ; and5,139,534 ; andEuropean Patent Publication No. 149,486 U.S. Pat. No. 4,518,782 ), amine oxides (for example, as described inU.S. Pat. Nos. 4,810,263 and4,836,829 ), hydroxyamines (for example, as described inU.S. Pat. No. 4,409,000 ), and succinimides (for example, as described inU.S. Pat. No. 4,292,046 ). - Each secondary fuel additive can be present in about 50 ppm to about 2500 ppm (such as 100 to 2000, 200 to 1500, 300 to 1000 and the like) by weight of the fuel composition. More preferably, the secondary fuel additive is present in about 50 ppm to about 1000 ppm by weight of the fuel composition.
- The fuel composition may comprise other generally known fuel additives. Suitable examples include, but are not limited to, antioxidants, metal deactivators, demulsifiers, oxygenates, antiknock agents, dispersants and other detergents. In diesel fuel, other well-known additives can be employed such as pour point depressants, flow improvers, and the like.
- Each of the foregoing additives, when used, is used at a functionally effective amount to impart the desired properties to the fuel composition. Generally, the concentration of each of these additives, when used, may range, unless otherwise specified, from about 0.001 to about 20 wt. %, such as about 0.01 to about 10 wt. %.
- The compounds of the present disclosure may be formulated as a concentrate using an inert stable oleophilic (i.e., soluble in hydrocarbon fuel) organic solvent boiling in a range of 65°C to 205°C. An aliphatic or an aromatic hydrocarbon solvent may be used, such as benzene, toluene, xylene, or higher-boiling aromatics or aromatic thinners. Aliphatic alcohols containing 2 to 8 carbon atoms, such as ethanol, isopropanol, methyl isobutyl carbinol, n-butanol and the like, in combination with the hydrocarbon solvents are also suitable for use with the present additives. In the concentrate, the amount of the additive ranges from 10 to 70 wt % (e.g., 20 to 40 wt %).
- The following examples are intended to be non-limiting.
- Inventive Example 1 is 2-(4-dodecylphenoxy)ethyl-1-amine shown below (Formula V).
- 4-Dodecylphenol (200.0 g, 0.76 mol) was dissolved in 1000 mL of naphtha (Aromatic 100) solvent in a 2L 3-neck round bottom flask. To this mixture was added potassium hydroxide (4.3 g) followed by hexanol (78 g, 0.76 mol) and the resulting mixture was stirred at room temperature under a nitrogen atmosphere for 30 minutes by using a mechanical stirrer. After that the mixture was heated to reflux (about 165-170 °C) and stirred under nitrogen for an additional 2 hours. During this time period, distillate liquid (e.g., water/hexanol and aromatic solvent) was collected in a Dean-Stark trap apparatus. After stirring for 2 hours, the mixture was cooled to 120 °C and 2-oxazolidinone (66.0 g, 0.76 mol) was added. The mixture was heated at reflux and stirred under nitrogen for 18 hours (overnight). The mixture was allowed to cool to room temperature, diluted with hexanes (100 mL) and the organic phase was washed with water (200 mL), brine (4 x 100 mL), dried over anhydrous MgSO4, and filtered through a pad of Celite filter aid. The filtrate was concentrated under reduced pressure and then under high vacuum to give an amber colored oil as crude product (240 g).
- Inventive Example 2 is a mixture of 2-(4-eicosanylphenoxy)ethyl-1-ami ne, 2-(4-docosanylphenoxy)ethyl-1-amine, and 2-(4-tetracosanylphenoxy)ethyl-1-amine shown below (Formula VI). It was obtained by Method A as described below.
- A mixture of 4-eicosanylphenol, 4-docosanylphenol, 4-tetracosanylphenol, 2-eicosanylphenol, 2-docosanylphenol, and 2-tetracosanylphenol with an averaged molecular weight of 402.71 g/mol (151.71 g, 0.377 mol) was dissolved in Aromatic 100 solvent (700 mL) in a 2L 3-neck round bottom flask. To this mixture was added potassium hydroxide (2.1 g) followed by hexanol (38.5 g, 0.377 mol) and the resulting mixture was stirred at room temperature under a nitrogen atmosphere for 30 minutes by using a mechanical stirrer. After that the mixture was heated to reflux (about 165-170 °C) and stirred under nitrogen for an additional 2 hours. During this time period, distillate liquid (e.g., water/hexanol and aromatic solvent) was collected in a Dean-Stark trap apparatus. After stirring for 2 hours, the mixture was cooled to 120 °C and 2-oxazolidinone (32.77 g, 0.377 mol) was added. The mixture was heated at reflux and stirred under nitrogen for 18 hours (overnight). The mixture was allowed to cool to room temperature, diluted with hexanes (200 mL) and the organic phase was washed with water (200 mL), brine (4 x 100 mL), dried over anhydrous MgSO4, and filtered through a pad of Celite filter aid. The filtrate was concentrated under reduced pressure and then under high vacuum to give a dark colored oil as crude product (150.2 g). The crude product was purified by column chromatography using a mixture of ethyl acetate/methanol gradient to give a pale yellow oil (105.8 g).
-
- 4-Nonylphenol (200.0 g, 0.908 mol) was dissolved in Aromatic 100 solvent (500 mL) in a 2L 3-neck round bottom flask. To this mixture was added potassium hydroxide (2.1 g) followed by hexanol (92.74 g, 0.908 mol) and the resulting mixture was stirred at room temperature under a nitrogen atmosphere for 30 minutes by using a mechanical stirrer. After that the mixture was heated to reflux (about 165-170 °C) and stirred under nitrogen for an additional 2 hours. During this time period, distillate liquid (e.g., water/hexanol and aromatic solvent) was collected in a Dean-Stark trap apparatus. After stirring for 2 hours, the mixture was cooled to 120 °C and 2-oxazolidinone (102.75 g, 1.180 mol) was added. The mixture was heated at reflux and stirred under nitrogen for 18 hours (overnight). The mixture was allowed to cool to 70 °C and ethylenediamine (10.91 g, 0.182 mol) was added and the mixture was heated at reflux for 4 hours. The mixture was cooled to room temperature and Magnesol® (100 g) was added and stirred for 30 minutes. The mixture was filtered and concentrated under high vacuum to yield a light yellow oil as crude product (243.67 g). This crude product was further washed with water (3 x 500 mL), brine (500 mL), dried over anhydrous MgSO4, and filtered through a pad of Celite filter aid. The filtrate was concentrated under reduced pressure and then under high vacuum. The crude product was purified by column chromatography using a mixture of ethyl acetate/methanol gradient to give a pale yellow oil.
- The Inventive Examples were blended in gasoline and tested for its ability to mitigate DISI direct injector fouling in a test vehicle. A 2017 VW Jetta SE vehicle equipped with a 1.4L 16-valve turbocharged DISI engine was used as the test vehicle.
-
FIG. 1 shows vehicle speed conditions observed during the specified vehicle drive cycle. The vehicle drive cycle is based on 10 hills extracted from the transient phase of the Environmental Protection Agency (EPA) Urban Dynamometer Drive Schedule (UDDS) with additional idle periods added. Total drive cycle is 20 minutes in duration and the overall test duration is 2,000-miles. - Additive testing is conducted in a "keep clean" configuration which starts with a clean injector and combustion chamber. This test configuration evaluates the ability of a given deposit control additive to keep the injector and combustion chamber clean over the duration of the test.
- Injector "keep clean" tests were performed on four fuel samples: (i) unadditized base fuel, (ii) 200 ppmw of inventive example 1 added to sample (i), (iii) 200 ppmw of inventive example 2 added to sample (i), and (iv) 200 ppmw of inventive example 3 added to sample (i).
- A set of four clean injectors were used at the start of each vehicle test. At the end of test, photographs of the four injectors were taken of the deposits formed.
FIG. 2 show photographs of injectors before and after the engine test. - Flow restriction of the injectors were also measured. Table 1 shows average injector flow restriction (%) measured at end of engine test compared to the base fuel reference (i.e., unadditized gasoline).
- As shown, Inventive Example 1 provided much lower flow restriction (0.39% on average) when compared to the base fuel reference (2.81% on average). Inventive Examples 2 and 3 also provided lower flow restriction (1.47% and 2.15% on average, respectively)
- Injector fuel restriction measures the decrease in fuel flow from the injector, representing the presence of deposits in the injector orifices. Injector restriction can force the engine controller to make additional control adjustments to maintain proper engine fuel delivery, and the presence of deposits in the injector orifices can impact fuel mixing, leading to decreased engine performance and increased particulate emissions.
Table 1 Base Fuel Reference Base Fuel + Inventive Example 1 (200 ppmw) Base Fuel + Inventive Example 2 (200 ppmw) Base Fuel + Inventive Example 3 (200 ppmw) Average Restriction [%] 2.81 0.39 1.47 2.15 Decrease in % restriction from base fuel - -2.42 -1.34 -0.66 - A second test was also conducted using an engine on a dynamometer stand. A 2017 Honda DISI 1.5L 16-valve turbocharged engine was the test vehicle engine used. The engine drive cycle is 720 s in duration with engine speeds ranging from idle to 4000 RPM, and load varying up to 160 Nm. The overall test duration is 50 hours (test duration for baseline fuel with 200 ppmw of Example 2 was 25 hours).
FIG. 3 illustrates engine speed and load test conditions. - A set of four clean injectors were used at the start of each engine test. At the end of test, photographs (
FIG. 4 ) of the four injectors were taken of the deposits formed. - PM measurements were made in the engine test stand using an AVL Micro Soot Sensor (MSS). The MSS provides a continuous, fast-response measurement of solid particulate mass and correlates well with the traditional, gravimetric method of PM measurement.
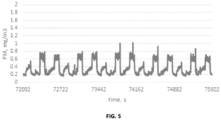
- In the PM emissions trace shown in
FIG. 5 (a 3600 second segment of the larger 50 hr. test), one can observe how quickly PM emissions rise and fall as engine conditions change. In order to provide a useful metric for these data, one can look toward official measurement methodologies used in regulatory vehicle emissions certification (e.g. U.S. Federal Test Procedure, or FTP). In these cases, regulatory agencies will simply report the sum total quantity of emissions from the vehicle tailpipe over an entire drive cycle. Applying a similar strategy to the PM dataset, we integrate PM emissions over the course of one test drive cycle. This integration is then repeated for each drive cycle and results in a PM emissions trendline over the entirety of the test duration. -
FIGS. 6A-6C illustrate the PM emissions traces from a baseline fuel (FIG. 6A ), the baseline fuel with 200ppmw of Inventive Example 1 (FIG. 6B ), and the baseline fuel with 200ppmw of Inventive Example 2 (FIG. 6C ). The addition of Inventive Example 1 or Inventive Example 2 at 200 ppmw maintains PM emissions at the same level as at start of test with a clean injector across the full duration of the test. - For the sake of brevity, only certain ranges are explicitly disclosed herein. However, ranges from any lower limit may be combined with any upper limit to recite a range not explicitly recited, as well as, ranges from any lower limit may be combined with any other lower limit to recite a range not explicitly recited, in the same way, ranges from any upper limit may be combined with any other upper limit to recite a range not explicitly recited. Additionally, within a range includes every point or individual value between its end points even though not explicitly recited. Thus, every point or individual value may serve as its own lower or upper limit combined with any other point or individual value or any other lower or upper limit, to recite a range not explicitly recited.
- Likewise, the term "comprising" is considered synonymous with the term "including." Likewise whenever a composition, an element or a group of elements is preceded with the transitional phrase "comprising," it is understood that we also contemplate the same composition or group of elements with transitional phrases "consisting essentially of," "consisting of," "selected from the group of consisting of," or "is" preceding the recitation of the composition, element, or elements and vice versa.
- The terms "a" and "the" as used herein are understood to encompass the plural as well as the singular.
- It is understood that when combinations, subsets, groups, etc. of elements are disclosed (e.g., combinations of components in a composition, or combinations of steps in a method), that while specific reference of each of the various individual and collective combinations and permutations of these elements may not be explicitly disclosed, each is specifically contemplated and described herein.
- The embodiments described hereinabove are further intended to explain best modes known of practicing it and to enable others skilled in the art to utilize the disclosure in such, or other, embodiments and with the various modifications required by the particular applications or uses. Accordingly, the description is not intended to limit it to the form disclosed herein.
Claims (15)
- A fuel composition comprising:gasoline; andaryloxy alkylamine additive having the following structure:wherein the aryloxy alkylamine additive is present in about 10 to about 750 ppm by weight based on total weight of the fuel composition;wherein X is a hydrocarbyl group having 1 or 2 carbon atoms; andwherein R1 and R2 are independently hydrogen or hydrocarbyl group having up to 36 carbon atoms.
- The fuel composition of claim 1, wherein X is ethylene group.
- The fuel composition of claim 1, further comprising:
a nitrogen-containing detergent, optionally wherein the nitrogen-containing detergent is an aliphatic hydrocarbyl amine, hydrocarbyl-substituted poly(oxyalkylene)amine, hydrocarbyl-substituted succinimide, Mannich reaction product, nitro and amino aromatic ester of polyalkylphenoxyalkanol, or polyalkylphenoxyaminoalkane. - The fuel composition of claim 1, further comprising antioxidants, metal deactivators, demulsifiers, oxygenates, antiknock agents, dispersants, pour point depressants, or flow improvers.
- The fuel composition of claim 1, wherein the aryloxy alkylamine is 2-(4-dodecylphenoxy)ethyl-1-amine, 2-(phenoxy)ethyl-1-amine, 2-(4-butylphenoxy)ethyl-1-amine, 2-(4-octylphenoxy)ethyl-1-amine, 2-(4-nonylphenoxy)ethyl-1-amine, 2-(4-octadecylphenoxy)ethyl-1-amine, 2-(4-eicosanylphenoxy)ethyl-1 amine, 2-(4-docosanylphenoxy)ethyl-1-amine, or 2-(4-tetracosanylphenoxy)ethyl-1-amine.
- A concentrate composition comprising:about 10 to 90 wt % of an organic solvent boiling in a range of from 65°C to 205°C and;
- The concentrate composition of claim 6, wherein X is ethylene group.
- The concentrate composition of claim 6, further comprising:
a nitrogen-containing detergent, optionally wherein the nitrogen-containing detergent is an aliphatic hydrocarbyl amine, hydrocarbyl-substituted poly(oxyalkylene)amine, hydrocarbyl-substituted succinimide, Mannich reaction product, nitro and amino aromatic ester of polyalkylphenoxyalkanol, or polyalkylphenoxyaminoalkane. - The concentrate composition of claim 6, wherein at least one of R1 and R2 is hydrogen.
- The concentrate composition of claim 6, wherein the aryloxy alkylamine is 2-(4-dodecylphenoxy)ethyl-1-amine, 2-(phenoxy)ethyl-1-amine, 2-(4-butylphenoxy)ethyl-1-amine, 2-(4-octylphenoxy)ethyl-1-amine, 2-(4-nonylphenoxy)ethyl-1-amine, 2-(4-octadecylphenoxy)ethyl-1-amine, 2-(4-eicosanylphenoxy)ethyl-1-ami ne, 2-(4-docosanylphenoxy)ethyl-1-amine, or 2-(4-tetracosanylphenoxy)ethyl-1-amine.
- A method of reducing injector fouling in a direct injection spark ignition gasoline engine comprising:
providing a gasoline composition comprising:wherein the aryloxy alkylamine additive is present in about 10 to about 750 ppm by weight based on total weight of the fuel composition;wherein X is a hydrocarbyl group having 1 or 2 carbon atoms; andwherein R1 and R2 are independently hydrogen, alkyl or alkenyl group having up to 36 carbon atoms. - The method of claim 11, wherein X is ethylene group.
- The method of claim 11, further comprising:
a nitrogen-containing detergent, optionally wherein the nitrogen-containing detergent is an aliphatic hydrocarbyl amine, hydrocarbyl-substituted poly(oxyalkylene)amine, hydrocarbyl-substituted succinimide, Mannich reaction product, nitro and amino aromatic ester of polyalkylphenoxyalkanol, or polyalkylphenoxyaminoalkane. - The method of claim 13, further comprising antioxidants, metal deactivators, demulsifiers, oxygenates, antiknock agents, dispersants, pour point depressants, or flow improvers.
- The method of claim 11, wherein the aryloxy alkylamine is 2-(4-dodecylphenoxy)ethyl-1-amine, 2-(phenoxy)ethyl-1-amine, 2-(4-butylphenoxy)ethyl-1-amine, 2-(4-octylphenoxy)ethyl-1-amine, 2-(4-nonylphenoxy)ethyl-1-amine, 2-(4-octadecylphenoxy)ethyl-1-amine, 2-(4-eicosanylphenoxy)ethyl-1-ami ne, 2-(4-docosanylphenoxy)ethyl-1-amine, or 2-(4-tetracosanylphenoxy)ethyl-1-amine.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202063079609P | 2020-09-17 | 2020-09-17 | |
| PCT/IB2021/058387 WO2022058894A1 (en) | 2020-09-17 | 2021-09-15 | Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP4214297A1 EP4214297A1 (en) | 2023-07-26 |
| EP4214297B1 true EP4214297B1 (en) | 2024-07-10 |
Family
ID=77897677
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP21773883.0A Active EP4214297B1 (en) | 2020-09-17 | 2021-09-15 | Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US20240026239A1 (en) |
| EP (1) | EP4214297B1 (en) |
| JP (1) | JP2023541471A (en) |
| KR (1) | KR20230068407A (en) |
| CN (1) | CN116234891A (en) |
| AU (1) | AU2021342779A1 (en) |
| CA (1) | CA3195345A1 (en) |
| CO (1) | CO2023004450A2 (en) |
| MX (1) | MX2023003074A (en) |
| WO (1) | WO2022058894A1 (en) |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5669939A (en) * | 1996-05-14 | 1997-09-23 | Chevron Chemical Company | Polyalkylphenoxyaminoalkanes and fuel compositions containing the same |
Family Cites Families (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US566939A (en) * | 1896-09-01 | eoney | ||
| US2622018A (en) | 1949-10-19 | 1952-12-16 | Socony Vacuum Oil Co Inc | Motor fuel |
| US3574576A (en) | 1965-08-23 | 1971-04-13 | Chevron Res | Distillate fuel compositions having a hydrocarbon substituted alkylene polyamine |
| DE1643240A1 (en) | 1966-09-16 | 1971-06-24 | Boehringer Sohn Ingelheim | Process for the preparation of new racemic or optically active 1-phenoxy-2-aminoalkanes |
| GB1346765A (en) | 1970-06-16 | 1974-02-13 | Shell Int Research | Fuel compositions |
| US3849083A (en) | 1972-04-14 | 1974-11-19 | Ethyl Corp | Gasoline additive |
| US3848056A (en) | 1972-09-05 | 1974-11-12 | Continental Oil Co | Molten alkaline alkanoic mixtures for absorption of sulfur oxides |
| US4231759A (en) | 1973-03-12 | 1980-11-04 | Standard Oil Company (Indiana) | Liquid hydrocarbon fuels containing high molecular weight Mannich bases |
| US3960515A (en) | 1973-10-11 | 1976-06-01 | Chevron Research Company | Hydrocarbyl amine additives for distillate fuels |
| GB1486144A (en) | 1974-03-13 | 1977-09-21 | Cities Service Oil Co | Gasoline additive |
| US4236020A (en) | 1976-06-21 | 1980-11-25 | Chevron Research Company | Carbamate deposit control additives |
| US4191537A (en) | 1976-06-21 | 1980-03-04 | Chevron Research Company | Fuel compositions of poly(oxyalkylene) aminocarbamate |
| US4160648A (en) | 1976-06-21 | 1979-07-10 | Chevron Research Company | Fuel compositions containing deposit control additives |
| US4288612A (en) | 1976-06-21 | 1981-09-08 | Chevron Research Company | Deposit control additives |
| US4233168A (en) | 1978-06-19 | 1980-11-11 | Chevron Research Company | Lubricant compositions containing dispersant additives |
| US4197409A (en) | 1978-08-08 | 1980-04-08 | Chevron Research Company | Poly(oxyalkylene)aminocarbomates of alkylene polyamine |
| US4243798A (en) | 1979-08-09 | 1981-01-06 | Chevron Research Company | Process for the production of a polymeric carbamate |
| US4292046A (en) | 1979-08-10 | 1981-09-29 | Mobil Oil Corporation | Detergent compositions |
| US4270930A (en) | 1979-12-21 | 1981-06-02 | Chevron Research Company | Clean combustion chamber fuel composition |
| US4518782A (en) | 1981-08-10 | 1985-05-21 | Texaco Inc. | Fuel compositions containing N-alkyl glycyl imidazoline |
| US4409000A (en) | 1981-12-14 | 1983-10-11 | The Lubrizol Corporation | Combinations of hydroxy amines and carboxylic dispersants as fuel additives |
| EP0149486A3 (en) | 1984-01-17 | 1986-10-08 | Atlantic Richfield Company | Detergent composition and gasoline composition containing same |
| US4836829A (en) | 1986-03-14 | 1989-06-06 | Exxon Research And Engineering Company | Fuel composition and process for multi-port fuel injection systems (PNE-509) |
| DE3611230A1 (en) | 1986-04-04 | 1987-10-08 | Basf Ag | POLYBUTYL AND POLYISOBUTYLAMINE, METHOD FOR THE PRODUCTION THEREOF AND THE FUEL AND LUBRICANT COMPOSITIONS CONTAINING THE SAME |
| US4810263A (en) | 1986-04-11 | 1989-03-07 | Exxon Research And Engineering Company | Fuel composition |
| US4729769A (en) | 1986-05-08 | 1988-03-08 | Texaco Inc. | Gasoline compositions containing reaction products of fatty acid esters and amines as carburetor detergents |
| US4881945A (en) | 1987-10-23 | 1989-11-21 | Chevron Research Company | Fuel compositions containing very long chain alkylphenyl poly(oxyalkylene) aminocarbonates |
| DE3826608A1 (en) | 1988-08-05 | 1990-02-08 | Basf Ag | FUELS CONTAINING POLYETHERAMINE OR POLYETHERAMINE DERIVATIVES FOR OTTO ENGINES |
| DE3826797A1 (en) | 1988-08-06 | 1990-02-08 | Basf Ag | FUEL COMPOSITIONS CONTAINING POLYCARBONIC ACIDIC LOW-CHAIN ALCOHOLS |
| DE3838918A1 (en) | 1988-11-17 | 1990-05-23 | Basf Ag | FUELS FOR COMBUSTION ENGINES |
| EP0382159A1 (en) | 1989-02-06 | 1990-08-16 | E.I. Du Pont De Nemours And Company | Defouling of fuel systems |
| WO1990010051A1 (en) | 1989-02-21 | 1990-09-07 | Union Oil Company Of California | Fuel composition for control of intake valve deposits |
| GB9007431D0 (en) | 1990-04-03 | 1990-05-30 | Shell Int Research | Diesel fuel additives |
| JP3402606B2 (en) | 1991-09-13 | 2003-05-06 | シェブロン リサーチ アンド テクノロジー カンパニー | Fuel additive composition containing polyisobutenyl succinimide |
| US5697988A (en) | 1991-11-18 | 1997-12-16 | Ethyl Corporation | Fuel compositions |
| GB9208034D0 (en) | 1992-04-10 | 1992-05-27 | Bp Chem Int Ltd | Fuel composition |
| US5620486A (en) | 1994-12-30 | 1997-04-15 | Chevron Chemical Company | Fuel compositions containing aryl succinimides |
| US5618320A (en) | 1996-05-14 | 1997-04-08 | Chevron Chemical Company | Aromatic esters of polyalkylphenoxyalkanols and fuel compositions containing the same |
| US6203584B1 (en) | 1998-03-31 | 2001-03-20 | Chevron Chemical Company Llc | Fuel composition containing an amine compound and an ester |
| US6114542A (en) | 1998-08-28 | 2000-09-05 | Chevron Chemical Company Llc | Ethers of polyalkyl or polyalkenyl N-hydroxyalkyl succinimides and fuel compositions containing the same |
| US5916825A (en) | 1998-08-28 | 1999-06-29 | Chevron Chemical Company Llc | Polyisobutanyl succinimides and fuel compositions containing the same |
| US5954843A (en) | 1998-08-28 | 1999-09-21 | Chevron Chemical Company Llc | Aminocarbamates of polyalkyl or polyalkenyl N-hydroxyalkyl succinimides and fuel compositions containing the same |
| US5993497A (en) | 1998-08-28 | 1999-11-30 | Chevron Chemical Company Llc | Esters of polyalkyl or polyalkenyl N-hydroxyalkyl succinimides and fuel compositions containing the same |
| US6217624B1 (en) | 1999-02-18 | 2001-04-17 | Chevron Chemical Company Llc | Fuel compositions containing hydrocarbyl-substituted polyoxyalkylene amines |
| US20030150154A1 (en) * | 2001-12-21 | 2003-08-14 | Cherpeck Richard E. | Polyalkylphenoxyaminoalkanes and fuel compositions containing the same |
| US9035097B2 (en) * | 2012-09-12 | 2015-05-19 | Chevron Oronite Company Llc | Aminoethylation process having improved yield of aryloxyalkylene amine compounds and reduced urea by-products |
-
2021
- 2021-09-15 CA CA3195345A patent/CA3195345A1/en active Pending
- 2021-09-15 KR KR1020237011754A patent/KR20230068407A/en active Search and Examination
- 2021-09-15 MX MX2023003074A patent/MX2023003074A/en unknown
- 2021-09-15 WO PCT/IB2021/058387 patent/WO2022058894A1/en active Application Filing
- 2021-09-15 US US18/025,303 patent/US20240026239A1/en active Pending
- 2021-09-15 EP EP21773883.0A patent/EP4214297B1/en active Active
- 2021-09-15 CN CN202180066654.2A patent/CN116234891A/en active Pending
- 2021-09-15 AU AU2021342779A patent/AU2021342779A1/en active Pending
- 2021-09-15 JP JP2023517692A patent/JP2023541471A/en active Pending
-
2023
- 2023-04-11 CO CONC2023/0004450A patent/CO2023004450A2/en unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5669939A (en) * | 1996-05-14 | 1997-09-23 | Chevron Chemical Company | Polyalkylphenoxyaminoalkanes and fuel compositions containing the same |
Also Published As
| Publication number | Publication date |
|---|---|
| CO2023004450A2 (en) | 2023-04-27 |
| JP2023541471A (en) | 2023-10-02 |
| CA3195345A1 (en) | 2022-03-24 |
| AU2021342779A1 (en) | 2023-04-27 |
| WO2022058894A1 (en) | 2022-03-24 |
| MX2023003074A (en) | 2023-05-19 |
| CN116234891A (en) | 2023-06-06 |
| KR20230068407A (en) | 2023-05-17 |
| US20240026239A1 (en) | 2024-01-25 |
| EP4214297A1 (en) | 2023-07-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0948587B1 (en) | Fuel compositions | |
| JP4646345B2 (en) | Fuel oil additive and fuel oil composition containing the additive | |
| AU689585B2 (en) | Fuel additive compositions containing an aliphatic amine, a polyolefin and a poly(oxyalkylene) monool | |
| US5752990A (en) | Composition and method for reducing combustion chamber deposits, intake valve deposits or both in spark ignition internal combustion engines | |
| EP3887488B1 (en) | Quaternary fatty amidoamine compound for use as an additive for fuel | |
| US6312481B1 (en) | Fuel compositions | |
| CA2541797C (en) | A fuel composition containing an alkylene oxide-adducted hydrocarbyl amide having reduced amine by-products | |
| KR20080055665A (en) | Improvements in fuel oil compositions | |
| EP4214297B1 (en) | Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines | |
| US5855630A (en) | Fuel compositions | |
| AU2021342779A9 (en) | Aryloxy alkylamines as fuel additives for reducing injector fouling in direct injection spark ignition gasoline engines | |
| CA2440548C (en) | A method of reducing particulate emissions in internal combustion engines | |
| US20220145199A1 (en) | Fuel additives for mitigating injector nozzle fouling and reducing particulate emissions | |
| US6261327B1 (en) | Additive concentrates for rapidly reducing octane requirement | |
| EP0944694B1 (en) | Fuel compositions containing esteramines | |
| JP4330828B2 (en) | Fuel oil additive and fuel oil composition | |
| JP2024533830A (en) | Fuel Additive for Reducing Deposits and Particulate Emissions - Patent application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: UNKNOWN |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20230315 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20240209 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602021015579 Country of ref document: DE |
|
| RAP4 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: CHEVRON U.S.A. INC. Owner name: CHEVRON ORONITE COMPANY LLC |