EP3801543B1 - Als hiv-integrase-hemmer nützliche tetracyclische heterocyclenverbindungen - Google Patents
Als hiv-integrase-hemmer nützliche tetracyclische heterocyclenverbindungen Download PDFInfo
- Publication number
- EP3801543B1 EP3801543B1 EP19814615.1A EP19814615A EP3801543B1 EP 3801543 B1 EP3801543 B1 EP 3801543B1 EP 19814615 A EP19814615 A EP 19814615A EP 3801543 B1 EP3801543 B1 EP 3801543B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- compound
- compound int
- isomer
- mmol
- mhz
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/22—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains four or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/18—Antivirals for RNA viruses for HIV
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/16—Peri-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/22—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains four or more hetero rings
Definitions
- the present invention relates to Tricyclic Heterocycle Compounds, compositions comprising at least one Tricyclic Heterocycle Compound, and methods of using the Tricyclic Heterocycle Compounds for treating or preventing HIV infection in a subject.
- a retrovirus designated human immunodeficiency virus is the strains known as HIV type-1 (HIV-1) virus and type-2 (HIV-2) virus, is the etiological agent of the complex disease that includes progressive destruction of the immune system (acquired immune deficiency syndrome; AIDS) and degeneration of the central and peripheral nervous system.
- HIV human immunodeficiency virus
- a common feature of retrovirus replication is the insertion by virally-encoded integrase of +proviral DNA into the host cell genome, a required step in HIV replication in human T-lymphoid and monocytoid cells.
- Integration is believed to be mediated by integrase in three steps: assembly of a stable nucleoprotein complex with viral DNA sequences; cleavage of two nucleotides from the 3' termini of the linear proviral DNA, and covalent joining of the recessed 3' OH termini of the proviral DNA at a staggered cut made at the host target site.
- the fourth step in the process, repair synthesis of the resultant gap may be accomplished by cellular enzymes.
- Nucleotide sequencing of HIV shows the presence of a pol gene in one open reading frame [ Ratner, L. et al., Nature, 313, 277(1985 )].
- Amino acid sequence homology provides evidence that the pol sequence encodes reverse transcriptase, integrase and an HIV protease [Toh, H. et al., EMBO J. 4, 1267 (1985 ); Power, M.D. et al., Science, 231, 1567 (1986 ); Pearl, L.H. et al., Nature, 329, 351 (1987 )]. All three enzymes have been shown to be essential for the replication of HIV.
- antiviral compounds which act as inhibitors of HIV replication are effective agents in the treatment of AIDS and similar diseases, including reverse transcriptase inhibitors such as azidothymidine (AZT) and efavirenz and protease inhibitors such as indinavir and nelfinavir.
- the compounds of this invention are inhibitors of HIV integrase and inhibitors of HIV replication.
- WO 2014/183532 and WO 2018/102634 describe other HIV integrase inhibitors.
- references to methods of treatment in the subsequent paragraphs of this description are to be interpreted as references to the compounds, pharmaceutical compositions and medicaments of the present invention for use in a method for treatment of the human (or animal) body by therapy (or for diagnosis).
- the present invention provides Compounds of Formula (I):
- the Compounds of Formula (I) (also referred to herein as the "Tricyclic Heterocycle Compounds") and pharmaceutically acceptable salts thereof may be useful, for example, for inhibiting HIV viral replication or replicon activity, or for treating or preventing HIV infection in a subject. Without being bound by any specific theory, it is believed that the Tricyclic Heterocycle Compounds inhibit HIV viral replication by inhibiting HIV Integrase.
- the present invention provides methods for treating or preventing HIV infection in a subject, comprising administering to the subject an effective amount of at least one Tricyclic Heterocycle Compound.
- the present invention includes Tricyclic Heterocycle Compounds, compositions comprising at least one Tricyclic Heterocycle Compound, and methods of using the Tricyclic Heterocycle Compounds for use in treating or preventing HIV infection in a subject.
- a "subject" is a human or non-human mammal.
- a subject is a human.
- a subject is a primate.
- a subject is a monkey.
- a subject is a chimpanzee.
- a subject is a rhesus monkey.
- an effective amount refers to an amount of Tricyclic Heterocycle Compound and/or an additional therapeutic agent, or a composition thereof that is effective in inhibiting HIV replication and in producing the desired therapeutic, ameliorative, inhibitory or preventative effect when administered to a subject suffering from HIV infection or AIDS.
- an effective amount can refer to each individual agent or to the combination as a whole, wherein the amounts of all agents administered are together effective, but wherein the component agent of the combination may not be present individually in an effective amount.
- treating includes inhibiting the severity of HIV infection or AIDS, i.e., arresting or reducing the development of the HIV infection or AIDS or its clinical symptoms; or relieving the HIV infection or AIDS, i.e., causing regression of the severity of HIV infection or AIDS or its clinical symptoms.
- preventing or “prohylaxis,” as used herein with respect to an HIV viral infection or AIDS, refers to reducing the likelihood or severity of HIV infection or AIDS.
- alkyl refers to an aliphatic hydrocarbon group having one of its hydrogen atoms replaced with a bond.
- An alkyl group may be straight or branched and contain from about 1 to about 20 carbon atoms. In one embodiment, an alkyl group contains from about 1 to about 12 carbon atoms. In different embodiments, an alkyl group contains from 1 to 6 carbon atoms (C 1 -C 6 alkyl) or from about 1 to about 4 carbon atoms (C 1 -C 4 alkyl).
- alkyl groups include methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, neopentyl, isopentyl, n-hexyl, isohexyl and neohexyl.
- an alkyl group is linear.
- an alkyl group is branched. Unless otherwise indicated, an alkyl group is unsubstituted.
- halo means -F, -Cl, -Br or -I.
- haloalkyl refers to an alkyl group as defined above, wherein one or more of the alkyl group's hydrogen atoms has been replaced with a halogen. In one embodiment, a haloalkyl group has from 1 to 6 carbon atoms. In another embodiment, a haloalkyl group is substituted with from 1 to 3 F atoms. Non-limiting examples of haloalkyl groups include -CH 2 F, -CHF 2 , -CF 3 , -CH 2 Cl and -CCl 3 .
- C 1 -C 6 haloalkyl refers to a haloalkyl group having from 1 to 6 carbon atoms.
- substituted means that one or more hydrogens on the designated atom is replaced with a selection from the indicated group, provided that the designated atom's normal valency under the existing circumstances is not exceeded, and that the substitution results in a stable compound. Combinations of substituents and/or variables are permissible only if such combinations result in stable compounds.
- stable compound' or “stable structure” is meant a compound that is sufficiently robust to survive isolation to a useful degree of purity from a reaction mixture, and formulation into an efficacious therapeutic agent.
- substantially purified form refers to the physical state of a compound after the compound is isolated from a synthetic process (e.g., from a reaction mixture), a natural source, or a combination thereof.
- substantially purified form also refers to the physical state of a compound after the compound is obtained from a purification process or processes described herein or well-known to the skilled artisan (e.g., chromatography, recrystallization and the like), in sufficient purity to be characterizable by standard analytical techniques described herein or well-known to the skilled artisan.
- protecting groups When a functional group in a compound is termed "protected", this means that the group is in modified form to preclude undesired side reactions at the protected site when the compound is subjected to a reaction. Suitable protecting groups will be recognized by those with ordinary skill in the art as well as by reference to standard textbooks such as, for example, T. W. Greene et al, Protective Groups in Organic Synthesis (1991), Wiley, New York .
- composition is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results from combination of the specified ingredients in the specified amounts.
- prodrugs and solvates of the compounds of the invention are also contemplated herein but prodrugs of the compounds of the invention are not part of the invention and therefore said prodrugs are not comprised by the claims.
- a discussion of prodrugs is provided in T. Higuchi and V. Stella, Pro-drugs as Novel Delivery Systems (1987) 14 of the A.C.S. Symposium Series , and in Bioreversible Carriers in Drug Design, (1987) Edward B. Roche, ed., American Pharmaceutical Association and Pergamon Press .
- the term "prodrug” means a compound (e.g., a drug precursor) that is transformed in vivo to provide a Tricyclic Heterocycle Compound or a pharmaceutically acceptable salt of the compound.
- a prodrug can comprise an ester formed by the replacement of the hydrogen atom of the acid group with a group such as, for example, (Ci-Cs)alkyl, (C 2 -C 12 )alkanoyloxymethyl, 1-(alkanoyloxy)ethyl having from 4 to 9 carbon atoms, 1-methyl-1-(alkanoyloxy)-ethyl having from 5 to 10 carbon atoms, alkoxycarbonyloxymethyl having from 3 to 6 carbon atoms, 1-(alkoxycarbonyloxy)ethyl having from 4 to 7 carbon atoms, 1-methyl-1-(alkoxycarbonyloxy)ethyl having from 5 to 8 carbon
- a prodrug can be formed by the replacement of one or more of the hydrogen atoms of the alcohol groups with a group such as, for example, (C 1 -C 6 )alkanoyloxymethyl, 1-((Ci-C 6 )alkanoyloxy)ethyl, 1-methyl-1-((C 1 -C 6 )alkanoyloxy)ethyl, (C 1 -C 6 )alkoxycarbonyloxymethyl, N-(C 1 -C 6 )alkoxycarbonylaminomethyl, succinoyl, (C 1 -C 6 )alkanoyl, ⁇ -amino(C 1 -C 4 )alkyl, ⁇ -amino(C 1 -C 4 )alkylene-aryl, arylacyl and ⁇ -aminoacyl, or ⁇ -aminoacyl- ⁇ -aminoacyl,
- a prodrug can be formed by the replacement of a hydrogen atom in the amine group with a group such as, for example, R-carbonyl-, RO-carbonyl-, NRR'-carbonyl- wherein R and R' are each independently (C 1 -C 10 )alkyl, (C 3 -C 7 ) cycloalkyl, benzyl, a natural ⁇ -aminoacyl, -C(OH)C(O)OY 1 wherein Y 1 is H, (C 1 -C 6 )alkyl or benzyl, -C(OY 2 )Y 3 wherein Y 2 is (C 1 -C 4 ) alkyl and Y 3 is (C 1 -C 6 )alkyl; carboxy (C 1 -C 6 )alkyl; amino(C 1 -C 4 )alkyl or mono-N- or di-N,N-
- R and R' are each independently (C 1 -C 10 )al
- esters of the present compounds are not part of the invention.
- Said esters include the following groups: (1) carboxylic acid esters obtained by esterification of the hydroxy group of a hydroxyl compound, in which the non-carbonyl moiety of the carboxylic acid portion of the ester grouping is selected from straight or branched chain alkyl ( e . g ., methyl, ethyl, n-propyl, isopropyl, t-butyl, sec-butyl or n-butyl), alkoxyalkyl ( e . g ., methoxymethyl), aralkyl ( e .
- benzyl aryloxyalkyl (for example, phenoxymethyl), aryl ( e . g ., phenyl optionally substituted with, for example, halogen, C 1-4 alkyl, -O-(C 1-4 alkyl) or amino);
- sulfonate esters such as alkyl- or aralkylsulfonyl (for example, methanesulfonyl); (3) amino acid esters, including those corresponding to both natural and non-natural amino acids ( e . g ., L-valyl or L-isoleucyl); (4) phosphonate esters and (5) mono-, di- or triphosphate esters.
- the phosphate esters may be further esterified by, for example, a C 1-20 alcohol or reactive derivative thereof, or by a 2,3-di (C 6-24 )acyl glycerol.
- One or more compounds of the invention may exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like, and it is intended that the invention embrace both solvated and unsolvated forms.
- “Solvate” means a physical association of a compound of this invention with one or more solvent molecules. This physical association involves varying degrees of ionic and covalent bonding, including hydrogen bonding. In certain instances the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated in the crystal lattice of the crystalline solid. "Solvate” encompasses both solution-phase and isolatable solvates. Non-limiting examples of solvates include ethanolates, methanolates, and the like. A “hydrate” is a solvate wherein the solvent molecule is water.
- One or more compounds of the invention may optionally be converted to a solvate.
- Preparation of solvates is generally known.
- M. Caira et al, J. Pharmaceutical Sci., 93(3) , 601-611 (2004) describe the preparation of the solvates of the antifungal fluconazole in ethyl acetate as well as from water.
- Similar preparations of solvates, hemisolvates, hydrates and the like are described by E. C. van Tonder et al, AAPS PharmSciTech., 5(1), article 12 (2004 ); and A. L. Bingham et al, Chem. Commun., 603-604 (2001 ).
- a typical, non-limiting, process involves dissolving the inventive compound in desired amounts of the desired solvent (organic or water or mixtures thereof) at a higher than room temperature, and cooling the solution at a rate sufficient to form crystals which are then isolated by standard methods.
- Analytical techniques such as, for example IR spectroscopy, show the presence of the solvent (or water) in the crystals as a solvate (or hydrate).
- Tricyclic Heterocycle Compounds can form salts which are also within the scope of this invention.
- Reference to a Tricyclic Heterocycle Compound herein is understood to include reference to salts thereof, unless otherwise indicated.
- the term "salt(s)", as employed herein, denotes acidic salts formed with inorganic and/or organic acids, as well as basic salts formed with inorganic and/or organic bases.
- Tricyclic Heterocycle Compound contains both a basic moiety, such as, but not limited to a pyridine or imidazole, and an acidic moiety, such as, but not limited to a carboxylic acid
- zwitterions inner salts
- the salt is a pharmaceutically acceptable (i.e., non-toxic, physiologically acceptable) salt.
- the salt is other than a pharmaceutically acceptable salt.
- Salts of the Compounds of Formula (I) may be formed, for example, by reacting a Tricyclic Heterocycle Compound with an amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization.
- Exemplary acid addition salts include acetates, ascorbates, benzoates, benzenesulfonates, bisulfates, borates, butyrates, citrates, camphorates, camphorsulfonates, fumarates, hydrochlorides, hydrobromides, hydroiodides, lactates, maleates, methanesulfonates, naphthalenesulfonates, nitrates, oxalates, phosphates, propionates, salicylates, succinates, sulfates, tartarates, thiocyanates, toluenesulfonates (also known as tosylates) and the like.
- Exemplary basic salts include ammonium salts, alkali metal salts such as sodium, lithium, and potassium salts, alkaline earth metal salts such as calcium and magnesium salts, salts with organic bases (for example, organic amines) such as dicyclohexylamine, t-butyl amine, choline, and salts with amino acids such as arginine, lysine and the like.
- Basic nitrogen-containing groups may be quartemized with agents such as lower alkyl halides ( e . g ., methyl, ethyl, and butyl chlorides, bromides and iodides), dialkyl sulfates ( e .
- halides e . g ., decyl, lauryl, and stearyl chlorides, bromides and iodides
- arylalkyl halides e . g ., benzyl and phenethyl bromides
- Diastereomeric mixtures can be separated into their individual diastereomers on the basis of their physical chemical differences by methods well-known to those skilled in the art, such as, for example, by chromatography and/or fractional crystallization.
- Enantiomers can be separated by converting the enantiomeric mixture into a diastereomeric mixture by reaction with an appropriate optically active compound (e . g ., chiral auxiliary such as a chiral alcohol or Mosher's acid chloride), separating the diastereomers and converting ( e . g ., hydrolyzing) the individual diastereomers to the corresponding pure enantiomers.
- an appropriate optically active compound e . g ., chiral auxiliary such as a chiral alcohol or Mosher's acid chloride
- Sterochemically pure compounds may also be prepared by using chiral starting materials or by employing salt resolution techniques. Also, some of the Tricyclic Heterocycle Compounds may be atropisomers ( e . g ., substituted biaryls) and are considered as part of this invention. Enantiomers can also be directly separated using chiral chromatographic techniques.
- Tricyclic Heterocycle Compounds may exist in different tautomeric forms, and all such forms are embraced within the scope of the invention.
- all keto-enol and imine-enamine forms of the compounds are included in the invention.
- Individual stereoisomers of the compounds of the invention may, for example, be substantially free of other isomers, or may be admixed, for example, as racemates or with all other, or other selected, stereoisomers.
- the chiral centers of the present invention can have the S or R configuration as defined by the IUPAC 1974 Recommendations.
- the use of the terms "salt”, “solvate”, “ester”, “prodrug” and the like, is intended to apply equally to the salt, solvate, ester and prodrug of enantiomers, stereoisomers, rotamers, tautomers, racemates or prodrugs of the inventive compounds.
- the atoms may exhibit their natural isotopic abundances, or one or more of the atoms may be artificially enriched in a particular isotope having the same atomic number, but an atomic mass or mass number different from the atomic mass or mass number predominantly found in nature.
- the present invention is meant to include all suitable isotopic variations of the compounds of generic Formula I.
- different isotopic forms of hydrogen (H) include protium ( 1 H) and deuterium ( 2 H).
- Protium is the predominant hydrogen isotope found in nature. Enriching for deuterium may provide certain therapeutic advantages, such as increasing in vivo half-life or reducing dosage requirements, or may provide a compound useful as a standard for characterization of biological samples.
- Isotopically-enriched Compounds of Formula (I) can be prepared without undue experimentation by conventional techniques well known to those skilled in the art or by processes analogous to those described in the Schemes and Examples herein using appropriate isotopically-enriched reagents and/or intermediates.
- a Compound of Formula (I) has one or more of its hydrogen atoms replaced with deuterium.
- the Tricyclic Heterocycle Compounds may be useful in human and veterinary medicine for treating or preventing HIV infection in a subject.
- the Tricyclic Heterocycle Compounds can be inhibitors of HIV viral replication.
- the Tricyclic Heterocycle Compounds are inhibitors of HIV-1. Accordingly, the Tricyclic Heterocycle Compounds may be useful for treating HIV infections and AIDS.
- the Tricyclic Heterocycle Compounds can be administered to a subject in need of treatment or prevention of HIV infection.
- the disclosure provides methods for treating HIV infection in a subject comprising administering to the subject an effective amount of at least one Tricyclic Heterocycle Compound or a pharmaceutically acceptable salt thereof.
- the present disclosure provides methods for treating AIDS in a subject comprising administering to the subject an effective amount of at least one Tricyclic Heterocycle Compound or a pharmaceutically acceptable salt thereof.
- the present invention provides Tricyclic Heterocycle Compounds of Formula (I):
- the present invention also provides Tricyclic Heterocycle Compounds of Formula (I): and pharmaceutically acceptable salts thereof, wherein:
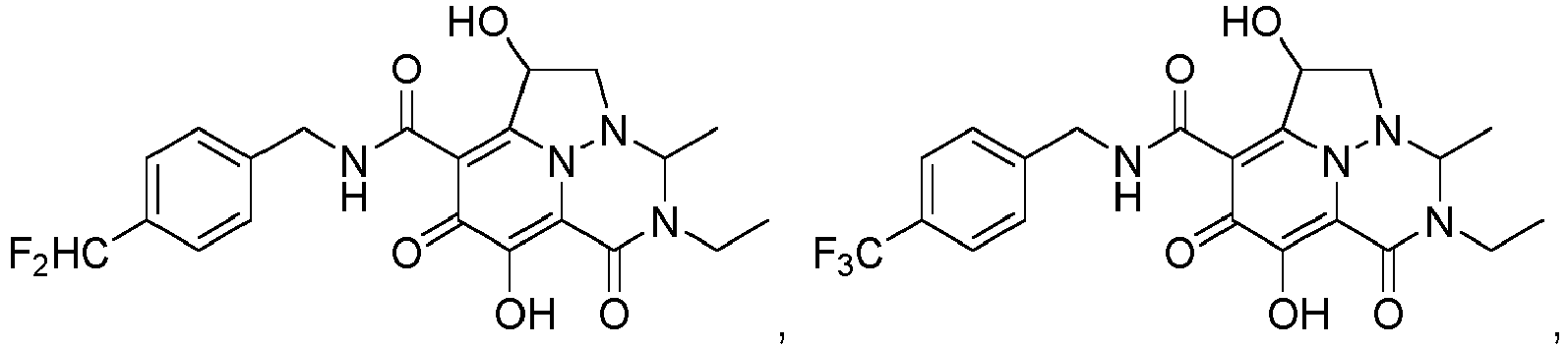
- R 1 is halo. In a class of the embodiment, R 1 is fluoro. In a class of the embodiment, R 1 is chloro. In another embodiment of the invention, R 1 is CHF 2 . In another embodiment of the invention, R 1 is CF 3 .
- R 2 is hydrogen
- R 3 is hydrogen
- R 4 is hydrogen or methyl. In a class of the invention, R 4 is hydrogen. In another class of the invention, R 4 is methyl.
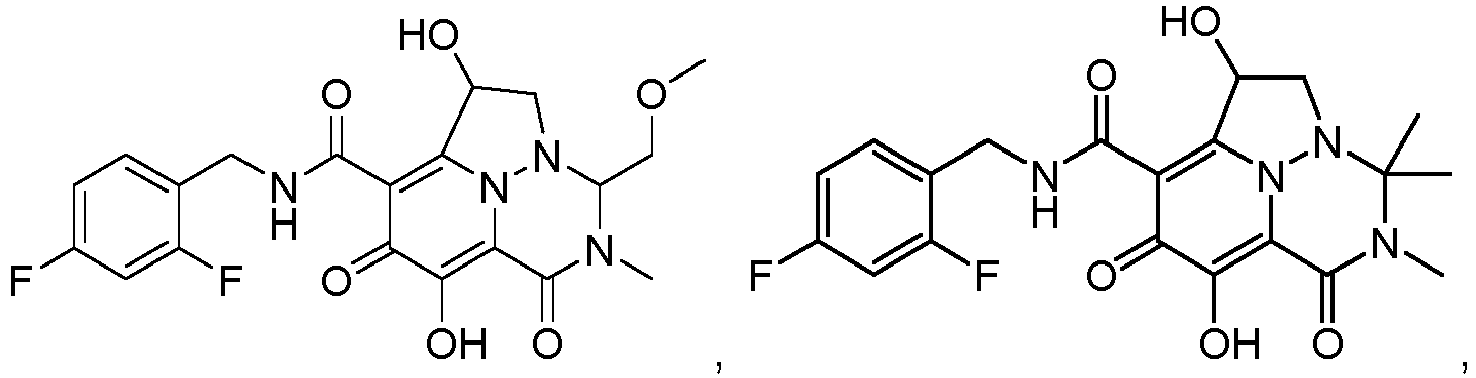
- R 5 is hydrogen, methyl, ethyl, CH 2 OCH 3 or phenyl. In a class of the invention, R 5 is hydrogen. In another class of the invention, R 5 is methyl. In another class of the invention, R 5 is ethyl. In another class of the invention, R 5 is CH 2 OCH 3 . In another class of the invention, R 5 is phenyl.
- R 4 and R 5 can be taken together with the carbon atom to which they are attached to form a 5- or 6-membered heterocyclyl group.
- R 4 and R 5 can be taken together with the carbon atom to which they are attached to form a 5-membered heterocyclyl group.
- R 4 and R 5 can be taken together with the carbon atom to which they are attached to form a tetrahydrofuranyl group.
- R 6 is methyl, ethyl or CH 2 CH 2 OCH 3 . In a class of the invention, R 6 is methyl. In another class of the invention, R 6 is ethyl. In another class of the invention, R 6 is CH 2 CH 2 OCH 3 .
- R 5 and R 6 can be taken together with the atoms between them to form a 6-membered heterocyclyl group. In a class of the invention, R 5 and R 6 can be taken together with the atoms between them to form a morpholinyl group.
- n is one. In another embodiment of the invention, n is two. In another embodiment of the invention, n is three.
- the Compounds of Formula (I) are in substantially purified form.
- the present invention also includes a compound of the present invention for use (i) in, (ii) as a medicament for, or (iii) in the preparation of a medicament for: (a) medicine; (b) inhibiting HIV replication or (c) treating HIV infection and/or reducing the likelihood or severity of symptoms of HIV infection.
- the compounds of the present invention can optionally be employed in combination with one or more second therapeutic agents selected from HIV antiviral agents, anti-infective agents, and immunomodulators.
- Additional embodiments of the disclosure include the pharmaceutical compositions, combinations and methods set forth in (a)-(gg) above and the uses set forth in the preceding paragraph, wherein the compound of the present invention employed therein is a compound of one of the embodiments, aspects, classes, sub-classes, or features of the compounds described above. In all of these embodiments, the compound may optionally be used in the form of a pharmaceutically acceptable salt or hydrate as appropriate.
- compositions and methods provided as (a) through (gg) above are understood to include all embodiments of the compounds, including such embodiments as result from combinations of embodiments.
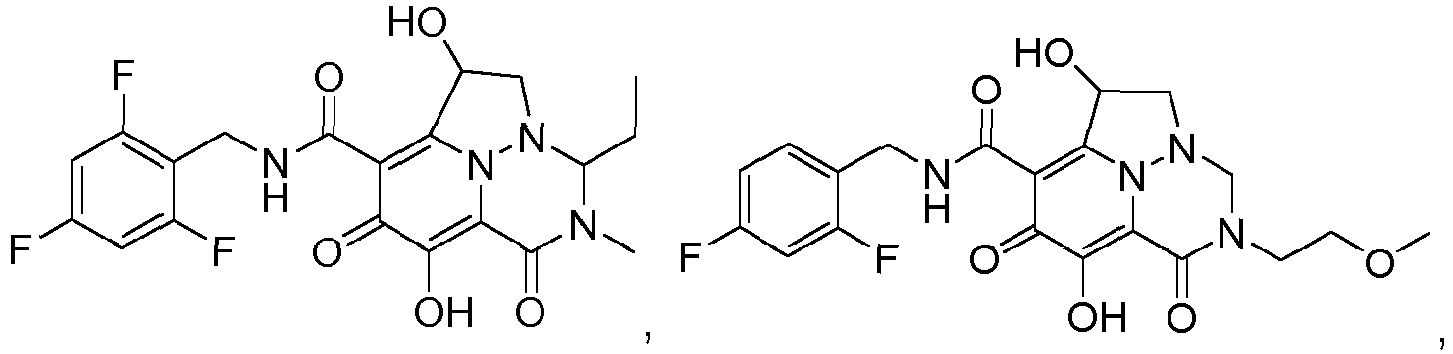
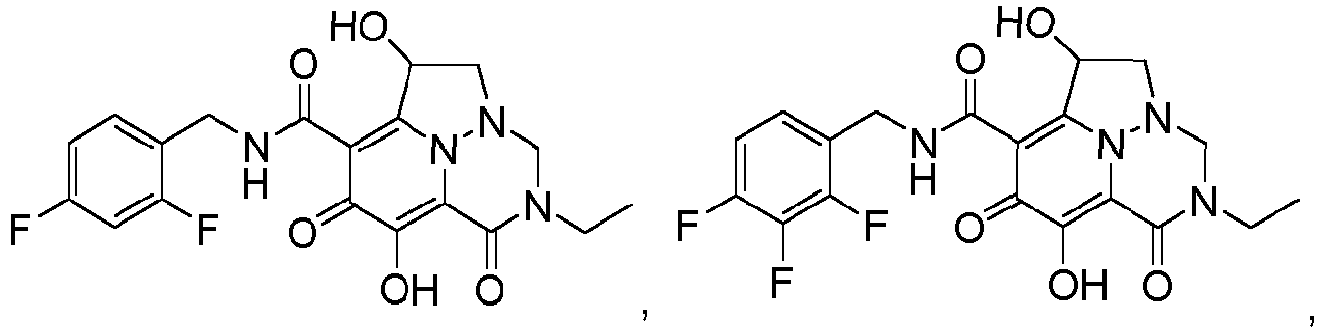
- Non-limiting examples of the Compounds of Formula (I) include compounds 1-122 as set forth in the Examples below, and pharmaceutically acceptable salts thereof.
- the Compounds of Formula (I) may be prepared from known or readily prepared starting materials, following methods known to one skilled in the art of organic synthesis. Methods useful for making the Compounds of Formula (I) are set forth in the Examples below and generalized in the Schemes below. Alternative synthetic pathways and analogous structures will be apparent to those skilled in the art of organic synthesis.
- reactions sensitive to moisture or air were performed under nitrogen or argon using anhydrous solvents and reagents.
- the progress of reactions was determined by either analytical thin layer chromatography (TLC) usually performed with E. Merck pre-coated TLC plates, silica gel 60F-254, layer thickness 0.25 mm or liquid chromatography-mass spectrometry (LC/MS).
- TLC analytical thin layer chromatography
- LC/MS liquid chromatography-mass spectrometry
- the analytical LC-MS system used consisted of a Waters ZQ TM platform with electrospray ionization in positive ion detection mode with an Agilent 1100 series HPLC with autosampler.
- the column was commonly a Waters Xterra MS C18, 3.0 ⁇ 50 mm, 5 ⁇ m or a Waters Acquity UPLC ® BEH C18 1.0 ⁇ 50 mm, 1.7,um.
- the flow rate was 1 mL/min, and the injection volume was 10 ⁇ L.
- UV detection was in the range 210-400 nm.
- the mobile phase consisted of solvent A (water plus 0.05% TFA) and solvent B (MeCN plus 0.05% TFA) with a gradient of 100% solvent A for 0.7 min changing to 100% solvent B over 3.75 min, maintained for 1.1 min, then reverting to 100% solvent A over 0.2 min.
- the column was commonly a Waters Acquity UPLC ® BEH C18 1.0 ⁇ 50 mm, 1.7 ⁇ m. The flow rate was 0.3 mL/min, and the injection volume was 0.5 ⁇ L. UV detection was 215 or 254 nm.
- Preparative HPLC purifications were usually performed using either a mass spectrometry directed system or a non-mass guided system. Usually they were performed on a Waters Chromatography Workstation configured with LC-MS System consisting of: Waters ZQ TM single quad MS system with Electrospray Ionization, Waters 2525 Gradient Pump, Waters 2767 Injecto /Collector, Waters 996 PDA Detector, the MS Conditions of: 150-750 amu, Positive Electrospray, Collection Triggered by MS, and a Waters SUNFIRE ® C-18 5 micron, 30 mm (id) ⁇ 100 mm column. The mobile phases consisted of mixtures of acetonitrile (10-100%) in water containing 0.1% TFA.
- Flow rates were maintained at 50 mL/min, the injection volume was 1800 ⁇ L, and the UV detection range was 210-400 nm.
- An alternate preparative HPLC system used was a Gilson Workstation consisting of: Gilson GX-281 Injector/Collector, Gilson UV/VIS-155 Detector, Gilson 322, 333, and 334 Pumps, and a Phenomenex Gemini-NX C-18 5 micron, 50 mm (id) ⁇ 250 mm column, a Waters XBridge TM C-18 5 micron OBD TM , 30 mm (id) ⁇ 250 mm column, or a Waters SUNFIRE TM C-18 OBD TM 10 micron, 30 mm (id) ⁇ 150 mm column.
- the mobile phases consisted of mixtures of acetonitrile (0-90%) in water containing 0.1% or 0.05% TFA. Flow rates were maintained at 50 mL/min for the Waters Xbridge TM column, 90 mL / min for the Phenomenex Gemini column, and 30 mL/min for the Waters SUNFIRE TM column. The injection volume ranged from 1000-8000 ,uL, and the UV detection range was 210-400 nm. Mobile phase gradients were optimized for the individual compounds. Reactions performed using microwave irradiation were normally carried out using an Emrys Optimizer manufactured by Personal Chemistry, or an Initiator manufactured by Biotage.
- Tetramethylsilane (TMS) was used as internal reference in CDCl 3 solutions, and residual CH 3 OH peak or TMS was used as internal reference in CD 3 OD solutions. Coupling constants (1) were reported in hertz (Hz). Chiral analytical chromatography was most commonly performed on one of CHIRALPAK ® AS, CHIRALPAK ® AD, CHIRALCEL ® OD, CHIRALCEL ® IA, or CHIRALCEL ® OJ columns (250 ⁇ 4.6 mm) (Daicel Chemical Industries, Ltd.) with noted percentage of ethanol in hexane (%EtOH/Hex), isopropanol in heptane (%IPA/Hep), ethanol in carbon dioxide (% EtOH/CO 2 ), or isopropanol in carbon dioxide (%IPA/CO 2 ) as isocratic solvent systems.
- CHIRALPAK AS Chiral preparative chromatography was conducted on one of CHIRALPAK AS, of CHIRALPAK AD, CHIRALCEL ® OD, CHIRALCEL ® IA, CHIRALCEL ® OJ columns (20 ⁇ 250 mm) (Daicel Chemical Industries, Ltd.) with desired isocratic solvent systems identified on chiral analytical chromatography or by supercritical fluid (SFC) conditions.
- SFC supercritical fluid
- the filtrate was purified by reverse phase HPLC (Waters Sunfire C18 OBD, 30 ⁇ 150 mm ⁇ 10 ⁇ m column), eluting with 10-90% (CH 3 CN + 0.05% TFA) / (water + 0.05% TFA).
- the product containing fractions were combined and concentrated under vacuum.
- the enantiomers of the product were further resolved by chiral preparative SFC (AS-H, 21 ⁇ 250 mm column; 50 g / min; 35% EtOH / CO 2 ; 210 nm) to afford compound int-2k-1 (1 st eluting component) and compound int-2k-2 (2 nd eluting component).
- LC/MS (m/z): 439.2 (M+H) + .
- the filtrate was purified by reverse phase HPLC (Waters Sunfire C18 OBD, 10 ⁇ m, 30 ⁇ 150 mm column), eluting with 20-100% (CH 3 CN + 0.05% TFA) / (water + 0.05% TFA). The product containing fractions were combined and concentrated under vacuum.
- Tetrakis(triphenylphosphine)palladium(0) (68 mg, 0.059 mmol), N,N-diisopropylethylamine (206 ⁇ l, 1.185 mmol) and 2,4,6-trifluorobenzylamine (95 mg, 0.592 mmol) were added to a stirred solution of compound int-16b (120 mg, 0.296 mmol) in DMSO (3 mL). The reaction mixture was degassed (3 ⁇ ) and placed under a carbon monoxide balloon.
- reaction mixture was stirred at 90°C for 1 hour before being cooled to room temperature, filtered (0.45 ⁇ m syringe filter), diluted with MeOH, and purified by reverse phase HPLC ( RediSep Rf C18, 100 g column) eluting with 10-100% ACN+ 0.05% TFA / water + 0.05% TFA.
- Tetrakis(triphenylphosphine)palladium(0) (30 mg, 0.026 mmol), N,N- diisopropylethylamine (91 ⁇ l, 0.522 mmol) and 2,4-difluorobenzylamine (37 mg, 0.261 mmol) were added to a stirred solution of compound int-23b (61 mg, 0.131 mmol) in DMSO (3 mL).
- the reaction mixture was purged with nitrogen (3 ⁇ ). Carbon monoxide balloon was attached and carbon monoxide gas was bubbled through a long needle to the mixture for 10 min.
- the reaction mixture was then stirred at 90°C for 1 hour before being cooled to room temperature.
- This material was further purified by chiral preparative SFC (DAICEL CHIRALPAK OJ-H, 21 ⁇ 250 mm column; 50 g / min; 15% EtOH / CO 2 ; 210 nm) to afford stereoisomer A of compound int-24c (1 st eluting component), isomer B of compound int-24c (2 nd eluting component), and a mixture of stereoisomer C and D of compound int-24c (3 rd eluting component).
- chiral preparative SFC DICEL CHIRALPAK OJ-H, 21 ⁇ 250 mm column; 50 g / min; 15% EtOH / CO 2 ; 210 nm
- the mixture of isomer C and D of compound int-24c was further purified by preparative chiral SFC (OJ-H, 21 ⁇ 250 mm column; 50 g / min; 40% EtOH / CO 2 ; 210 nm) to afford sufficient pure isomer C of compound int-24c (1 st eluting component) and isomer D of compound int-24c (2 nd eluting component).
- compounds 111-114 were prepared using essentially the same method described in Step F and Step G in example 27 with the exception of substituting 2,4,6-trifluorobenzylamine with 2,4-difluorobenzylamine, and purifying by chiral preparative SFC (ChiralPak OD-H, 21 ⁇ 250 mm column, 60 mL / min, 100 bar, 50% MeOH / CO 2 ) to afford Isomers A, Isomer B, Isomer C and Isomer D in Step F.
- chiral preparative SFC ChiralPak OD-H, 21 ⁇ 250 mm column, 60 mL / min, 100 bar, 50% MeOH / CO 2
- MT4-gag-GFP clone D3 (hereafter designated MT4-GFP), which are MT-4 cells modified to harbor a GFP reporter gene, the expression of which is dependent on the HIV-1 expressed proteins tat and rev.
- Productive infection of an MT4-GFP cell with HIV-1 results in GFP expression approximately 24 h post-infection.
- MT4-GFP cells were maintained at 37°C/5% CO 2 /90% relative humidity in RPMI 1640 supplemented with 10% fetal bovine serum, 100 U/ml penicillin/streptomycin, and 400 ⁇ g/ml G418 to maintain the reporter gene.
- MT4-GFP cells were placed in the same medium lacking G418 and infected overnight with HIV-1 (H9/IIIB strain) virus at an approximate multiplicity of infection of 0.01 in the same incubation conditions. Cells were then washed and re-suspended in either RPMI 1640 at 2 ⁇ 10 5 cells/mL (0% NHS condition) or 100% normal human serum (NHS) at 2 ⁇ 10 5 cells/mL (100% NHS condition).
- Compound plates were prepared by dispensing compounds dissolved in DMSO into wells of 384 well poly-D-lysine-coated plates (0.2 ⁇ l/well) using an ECHO acoustic dispenser. Each compound was tested in a 10-point serial 3-fold dilution (typical final concentrations: 1050 nM-0.05 nM for 0% NHS condition or 42 ⁇ M-2.13 nM for 100% NHS condition). Controls included no inhibitor (DMSO only) and a combination of three antiviral agents (efavirenz, indinavir, an in-house integrase strand transfer inhibitor at final concentrations of 4 ⁇ M each). Cells were added (50 ⁇ L/well) to compound plates and the infected cells were maintained at 37°C/5% CO 2 /90% relative humidity.
- Infected cells were quantified at two time points, ⁇ 48h and ⁇ 72h post-infection, by counting the number of green cells in each well using an Acumen eX3 scanner. The increase in the number of green cells over -24h period gives the reproductive ratio, R0, which is typically 5-15 and has been shown experimentally to be in logarithmic phase (data not shown). Inhibition of R0 is calculated for each well, and IC 50s determined by non-linear 4-parameter curve fitting. Assay IC 50 results are shown in the table below. Compound No. WILD TYPE CELL ASSAY Viking IP (0% NHS) (nM) Compound No. WILD TYPE CELL ASSAY Viking IP (0% NHS) (nM) Compound No.
- WILD TYPE CELL ASSAY Viking IP (0% NHS) (nM) 1 1.7 42 1.9 83 2.5 2 1.6 43 3.4 84 2.1 3 3.2 44 3.1 85 2.6 4 3.5 45 2.5 86 1.3 5 3.5 46 3.7 87 4.2 6 3.0 47 2.8 88 2.9 7 1.6 48 2.3 89 1.9 8 1.9 49 2.0 90 1.9 9 1.4 50 4.2 91 3.3 10 1.9 51 2.1 92 1.7 11 2.8 52 8.1 93 9.3 12 1.7 53 1.1 94 14 13 1.7 54 4.7 95 7.5 14 1.4 55 1.6 96 26 15 2.6 56 2.9 97 ND 16 1.9 57 2.4 98 ND 17 1.5 58 4.0 99 ND 18 1.6 59 2.5 100 ND 19 2.7 60 2.8 101 2.5 20 1.7 61 1.9 102 3.5 21 1.4 62 102 103 ND 22 2.3 63 1.6 104 3.9 23 2.3 64 5.9 105 ND 24 1.5 65 2.0 106 ND 25 2.0 66
- the Tricyclic Heterocycle Compounds may be useful in the inhibition of HIV, the inhibition of HIV integrase, the treatment of HIV infection and/or reduction of the likelihood or severity of symptoms of HIV infection and the inhibition of HIV viral replication and/or HIV viral production in a cell-based system.
- the Tricyclic Heterocycle Compounds may be useful in treating infection by HIV after suspected past exposure to HIV by such means as blood transfusion, exchange of body fluids, bites, accidental needle stick, or exposure to subject blood during surgery or other medical procedures.
- the disclosure provides methods for treating HIV infection in a subject, the methods comprising administering to the subject an effective amount of at least one Tricyclic Heterocycle Compound or a pharmaceutically acceptable salt.
- the amount administered is effective to treat or prevent infection by HIV in the subject.
- the amount administered is effective to inhibit HIV viral replication and/or viral production in the subject.
- the HIV infection has progressed to AIDS.
- the Tricyclic Heterocycle Compounds are also useful in the preparation and execution of screening assays for antiviral compounds.
- the Tricyclic Heterocycle Compounds may be useful for identifying resistant HIV cell lines harboring mutations, which are excellent screening tools for more powerful antiviral compounds.
- the Tricyclic Heterocycle Compounds may be useful in establishing or determining the binding site of other antivirals to the HIV Integrase.
- compositions and combinations of the present invention may be useful for treating a subject suffering from infection related to any HIV genotype.
- the present methods for treating or preventing HIV infection can further comprise the administration of one or more additional therapeutic agents which are not Tricyclic Heterocycle Compounds.
- the additional therapeutic agent is an antiviral agent.
- the additional therapeutic agent is an immunomodulatory agent, such as an immunosuppressive agent.
- the present disclosure provides methods for treating a viral infection in a subject, the method comprising administering to the subject: (i) at least one Tricyclic Heterocycle Compound (which may include two or more different Tricyclic Heterocycle Compounds), or a pharmaceutically acceptable salt or prodrug thereof, and (ii) at least one additional therapeutic agent that is other than a Tricyclic Heterocycle Compound, wherein the amounts administered are together effective to treat or prevent a viral infection.
- therapeutic agents in the combination may be administered in any order such as, for example, sequentially, concurrently, together, simultaneously and the like.
- the amounts of the various actives in such combination therapy may be different amounts (different dosage amounts) or same amounts (same dosage amounts).
- a Tricyclic Heterocycle Compound and an additional therapeutic agent may be present in fixed amounts (dosage amounts) in a single dosage unit ( e.g. , a capsule, a tablet and the like).
- At least one Tricyclic Heterocycle Compound is administered during a time when the additional therapeutic agent(s) exert their prophylactic or therapeutic effect, or vice versa.
- At least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) are administered in doses commonly employed when such agents are used as monotherapy for treating a viral infection.
- At least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) are administered in doses lower than the doses commonly employed when such agents are used as monotherapy for treating a viral infection.
- At least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) act synergistically and are administered in doses lower than the doses commonly employed when such agents are used as monotherapy for treating a viral infection.
- At least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) are present in the same composition.
- this composition is suitable for oral administration.
- this composition is suitable for intravenous administration.
- this composition is suitable for subcutaneous administration.
- this composition is suitable for parenteral administration.
- Viral infections and virus-related disorders that may be treated or prevented using the combination therapy methods of the present invention include, but are not limited to, those listed above.
- the viral infection is HIV infection.
- the viral infection is AIDS.
- the at least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) can act additively or synergistically.
- a synergistic combination may allow the use of lower dosages of one or more agents and/or less frequent administration of one or more agents of a combination therapy.
- a lower dosage or less frequent administration of one or more agents may lower toxicity of therapy without reducing the efficacy of therapy.
- the administration of at least one Tricyclic Heterocycle Compound and the additional therapeutic agent(s) may inhibit the resistance of a viral infection to these agents.
- an anti-HIV agent is any agent which is directly or indirectly effective in the inhibition of HIV reverse transcriptase or another enzyme required for HIV replication or infection, the treatment or prophylaxis of HIV infection, and/or the treatment, prophylaxis or delay in the onset or progression of AIDS. It is understood that an anti-HIV agent is effective in treating, preventing, or delaying the onset or progression of HIV infection or AIDS and/or diseases or conditions arising therefrom or associated therewith.
- the compounds of this invention may be effectively administered, whether at periods of pre-exposure and/or post-exposure, in combination with effective amounts of one or more anti-HIV agents selected from HIV antiviral agents, immunomodulators, antiinfectives, or vaccines useful for treating HIV infection or AIDS.
- one or more anti-HIV agents selected from HIV antiviral agents, immunomodulators, antiinfectives, or vaccines useful for treating HIV infection or AIDS.
- Suitable HIV antivirals for use in combination with the compounds of the present invention include, for example, those listed in Table A as follows: Table A Name Trade Name abacavir, ABC Ziagen ® abacavir +lamivudine Epzicom ® abacavir + lamivudine + zidovudine Trizivir ® amprenavir Agenerase ® atazanavir Reyataz ® AZT, zidovudine, azidothymidine Retrovir ® darunavir Prezista ® ddC, zalcitabine, dideoxycytidine Hivid ® ddI, didanosine, dideoxyinosine Videx ® ddI (enteric coated) Videx EC ® delavirdine, DLV Rescriptor ® dolutegravir Tivicay ® doravirine efavirenz, EFV Sustiva ® , Stocrin ® efavirenz + e
- one or more anti-HIV drugs are selected from, lamivudine, abacavir, ritonavir, darunavir, atazanavir, emtricitabine, tenofovir, rilpivirine, doravirine, EFdA and lopinavir.
- the compound of formula (I) is used in combination with lamivudine.
- the compound of formula (I) is used in combination atazanavir.
- the compound of formula (I) is used in combination with darunavir.
- the compound of formula (I) is used in combination with rilpivirine.
- the compound of formula (I) is used in combination with lamivudine and abacavir.
- the compound of formula (I) is used in combination with EFdA.
- the compound of formula (I) is used in combination with emtricitabine and tenofovir.
- the compound of formula (I) is used in combination doravirine.
- the compound of formula (I) is used in combination with ritonavir and lopinavir.
- the compound of formula (I) is used in combination with abacavir and lamivudine.
- the compound of formula (I) is used in combination with lopinavir and ritonavir.
- the present invention provides pharmaceutical compositions comprising (i) a compound of formula (I) or a pharmaceutically acceptable salt thereof; (ii) a pharmaceutically acceptable carrier; and (iii) one or more additional anti-HIV agents selected from lamivudine, abacavir, ritonavir and lopinavir, or a pharmaceutically acceptable salt wherein the amounts present of components (i) and (iii) are together effective for the treatment or prophylaxis of infection by HIV or for the treatment, prophylaxis, or delay in the onset or progression of AIDS in the subject in need thereof.
- the present disclosure provides a method for the treatment or prophylaxis of infection by HIV or for the treatment, prophylaxis, or delay in the onset or progression of AIDS in a subject in need thereof, which comprises administering to the subject (i) a compound of formula (I) or a pharmaceutically acceptable salt and (ii) one or more additional anti-HIV agents selected from lamivudine, abacavir, ritonavir and lopinavir, or a pharmaceutically acceptable salt wherein the amounts administered of components (i) and (ii) are together effective for the treatment or prophylaxis of infection by HIV or for the treatment, prophylaxis, or delay in the onset or progression of AIDS in the subject in need thereof.
- HIV antiviral agents and other agents will typically be employed in these combinations in their conventional dosage ranges and regimens as reported in the art, including, for example, the dosages described in the Physicians' Desk Reference, Thomson PDR, Thomson PDR, 57th edition (2003 ), the 58 th edition (2004), the 59 th edition (2005), and the like.
- the dosage ranges for a compound of the invention in these combinations are the same as those set forth above.
- the doses and dosage regimen of the other agents used in the combination therapies of the present invention for the treatment or prevention of HIV infection may be determined by the attending clinician, taking into consideration the approved doses and dosage regimen in the package insert; the age, sex and general health of the subject; and the type and severity of the viral infection or related disease or disorder.
- the Tricyclic Heterocycle Compound(s) and the other agent(s) may be administered simultaneously ( i.e., in the same composition or in separate compositions one right after the other) or sequentially.
- kits comprising the separate dosage forms is therefore advantageous.
- the Tricyclic Heterocycle Compounds When administered to a subject, the Tricyclic Heterocycle Compounds may be administered as a component of a composition that comprises a pharmaceutically acceptable carrier or vehicle.
- the present invention provides pharmaceutical compositions comprising an effective amount of at least one Tricyclic Heterocycle Compound and a pharmaceutically acceptable carrier.
- the active ingredients will typically be administered in admixture with suitable carrier materials suitably selected with respect to the intended form of administration, i.e., oral tablets, capsules (either solid-filled, semi-solid filled or liquid filled), powders for constitution, oral gels, elixirs, dispersible granules, syrups, suspensions, and the like, and consistent with conventional pharmaceutical practices.
- the active drug component may be combined with any oral non-toxic pharmaceutically acceptable inert carrier, such as lactose, starch, sucrose, cellulose, magnesium stearate, dicalcium phosphate, calcium sulfate, talc, mannitol, ethyl alcohol (liquid forms) and the like.
- Solid form preparations include powders, tablets, dispersible granules, capsules, cachets and suppositories. Powders and tablets may be comprised of from about 0.5 to about 95 percent inventive composition. Tablets, powders, cachets and capsules may be used as solid dosage forms suitable for oral administration.
- suitable binders include starch, gelatin, natural sugars, corn sweeteners, natural and synthetic gums such as acacia, sodium alginate, carboxymethylcellulose, polyethylene glycol and waxes.
- lubricants there may be mentioned for use in these dosage forms, boric acid, sodium benzoate, sodium acetate, sodium chloride, and the like.
- Disintegrants include starch, methylcellulose, guar gum, and the like. Sweetening and flavoring agents and preservatives may also be included where appropriate.
- Liquid form preparations include solutions, suspensions and emulsions and may include water or water-propylene glycol solutions for parenteral injection.
- Liquid form preparations may also include solutions for intranasal administration.
- solid form preparations which are intended to be converted, shortly before use, to liquid form preparations for either oral or parenteral administration.
- liquid forms include solutions, suspensions and emulsions.
- a low melting wax such as a mixture of fatty acid glycerides or cocoa butter is first melted, and the active ingredient is dispersed homogeneously therein as by stirring. The molten homogeneous mixture is then poured into convenient sized molds, allowed to cool and thereby solidify.
- compositions of the present invention may be formulated in sustained release form to provide the rate controlled release of any one or more of the components or active ingredients to optimize therapeutic effects, i.e., antiviral activity and the like.
- Suitable dosage forms for sustained release include layered tablets containing layers of varying disintegration rates or controlled release polymeric matrices impregnated with the active components and shaped in tablet form or capsules containing such impregnated or encapsulated porous polymeric matrices.
- the one or more Tricyclic Heterocycle Compounds are administered orally.
- the one or more Tricyclic Heterocycle Compounds are administered intravenously.
- a pharmaceutical preparation comprising at least one Tricyclic Heterocycle Compound is in unit dosage form.
- the preparation is subdivided into unit doses containing effective amounts of the active components.
- compositions may be prepared according to conventional mixing, granulating or coating methods, respectively, and the present compositions can contain, in one embodiment, from about 0.1% to about 99% of the Tricyclic Heterocycle Compound(s) by weight or volume. In various embodiments, the present compositions can contain, in one embodiment, from about 1% to about 70% or from about 5% to about 60% of the Tricyclic Heterocycle Compound(s) by weight or volume.
- the compounds of Formula I may be administered orally in a dosage range of 0.001 to 1000 mg/kg of mammal (e.g., human) body weight per day in a single dose or in divided doses.
- mammal e.g., human
- One dosage range is 0.01 to 500 mg/kg body weight per day orally in a single dose or in divided doses.
- Another dosage range is 0.1 to 100 mg/kg body weight per day orally in single or divided doses.
- the compositions may be provided in the form of tablets or capsules containing 1.0 to 500 milligrams of the active ingredient, particularly 1, 5, 10, 15, 20, 25, 50, 75, 100, 150, 200, 250, 300, 400, and 500 milligrams of the active ingredient for the symptomatic adjustment of the dosage to the subject to be treated.
- the specific dose level and frequency of dosage for any particular subject may be varied and will depend upon a variety of factors including the activity of the specific compound employed, the metabolic stability and length of action of that compound, the age, body weight, general health, sex, diet, mode and time of administration, rate of excretion, drug combination, the severity of the particular condition, and the host undergoing therapy.
- the total daily dosage may be divided and administered in portions during the day if desired. In one embodiment, the daily dosage is administered in one portion. In another embodiment, the total daily dosage is administered in two divided doses over a 24 hour period. In another embodiment, the total daily dosage is administered in three divided doses over a 24 hour period. In still another embodiment, the total daily dosage is administered in four divided doses over a 24 hour period.
- the unit dosages of the Tricyclic Heterocycle Compounds may be administered at varying frequencies. In one embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once daily. In another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered twice weekly. In another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once weekly. In still another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once biweekly. In another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once monthly. In yet another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once bimonthly.
- a unit dosage of a Tricyclic Heterocycle Compound may be administered once every 3 months. In a further embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once every 6 months. In another embodiment, a unit dosage of a Tricyclic Heterocycle Compound may be administered once yearly.
- compositions of the invention can further comprise one or more additional therapeutic agents, selected from those listed above herein.
- the present invention provides a kit comprising a therapeutically effective amount of at least one Tricyclic Heterocycle Compound, or a pharmaceutically acceptable salt or prodrug of said compound and a pharmaceutically acceptable carrier, vehicle or diluent.
- the present invention provides a kit comprising an amount of at least one Tricyclic Heterocycle Compound, or a pharmaceutically acceptable salt or prodrug of said compound and an amount of at least one additional therapeutic agent listed above, wherein the amounts of the two or more active ingredients result in a desired therapeutic effect.
- the one or more Tricyclic Heterocycle Compounds and the one or more additional therapeutic agents are provided in the same container.
- the one or more Tricyclic Heterocycle Compounds and the one or more additional therapeutic agents are provided in separate containers.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Virology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- AIDS & HIV (AREA)
- Tropical Medicine & Parasitology (AREA)
- Molecular Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Claims (15)
- Eine Verbindung der Formel:oder ein pharmazeutisch annehmbares Salz davon,wobei:jedes Vorkommen von R1 unabhängig Halogen oder C1-3-Alkyl ist, wobei die Alkylgruppen gegebenenfalls substituiert sind mit ein bis drei Halogenen,R2 Wasserstoff, Methyl oder Ethyl ist,R3 Wasserstoff, Methyl oder Ethyl ist,R4 Wasserstoff, Methyl oder Ethyl ist,R5 Wasserstoff, C1-3-Alkyl, (C1-3-Alkyl)OR7 oder Phenyl ist,R6 Wasserstoff, C1-6-Alkyl oder (C1-6-Alkyl)OR7 ist,oder R5 und R6 mit den dazwischenliegenden Atomen zusammengenommen sein können unter Bildung einer 5- bis 7-gliedrigen Heterocyclylgruppe,R7 Wasserstoff oder C1-3-Alkyl, das gegebenenfalls mit ein bis drei Halogenen substituiert ist, ist,n eine ganze Zahl zwischen eins und drei ist.
- Die Verbindung nach Anspruch 1, wobei jedes R1 Halogen ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach Anspruch 1 oder 2, wobei R2 Wasserstoff ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 3, wobei R3 Wasserstoff ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 4, wobei R4 Wasserstoff oder Methyl ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 5, wobei R4 Wasserstoff ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 6, wobei R5 Wasserstoff, Methyl, Ethyl, CH2OCH3 oder Phenyl ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 7, wobei R6 Methyl, Ethyl oder CH2CH2OCH3 ist, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 10, wobei R5 und R6 mit dem Kohlenstoffatom, an das sie gebunden sind, zusammengenommen sein können unter Bildung einer 6-gliedrigen Heterocyclylgruppe, oder ein pharmazeutisch annehmbares Salz davon.
- Die Verbindung nach einem der Ansprüche 1 bis 11, wobei R5 und R6 mit den dazwischenliegenden Atomen zusammengenommen sein können unter Bildung einer Morpholinylgruppe, oder ein pharmazeutisch annehmbares Salz davon.
- Eine pharmazeutische Zusammensetzung, umfassend eine wirksame Menge einer Verbindung gemäß einem der Ansprüche 1 bis 11 oder eines pharmazeutisch annehmbaren Salzes davon und einen pharmazeutisch annehmbaren Träger.
- Die pharmazeutische Zusammensetzung nach Anspruch 12, ferner umfassend ein oder mehrere zusätzliche therapeutische Mittel, ausgewählt aus Raltegravir, Lamivudin, Abacavir, Ritonavir, Dolutegravir, Arunavir, Atazanavir, Emtricitabin, Tenofovir, Elvitegravir, Rilpivirin, EFdA, Doravirin und Lopinavir.
- Eine Verbindung gemäß einem der Ansprüche 1 bis 11 oder ein pharmazeutisch annehmbares Salz davon zur Verwendung bei der Inhibierung von HIV-Integrase, zur Behandlung oder Prophylaxe einer HIV-Infektion, oder zur Behandlung, Prophylaxe oder Verzögerung des Ausbruchs oder Fortschreitens von AIDS bei einem Subjekt, das dies benötigt.
- Eine Verbindung gemäß einem der Ansprüche 1 bis 11 oder ein pharmazeutisch annehmbares Salz davon zur Verwendung in der Therapie.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201862680794P | 2018-06-05 | 2018-06-05 | |
| PCT/US2019/034783 WO2019236396A1 (en) | 2018-06-05 | 2019-05-31 | Tricyclic heterocycle compounds useful as hiv integrase inhibitors |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP3801543A1 EP3801543A1 (de) | 2021-04-14 |
| EP3801543A4 EP3801543A4 (de) | 2022-03-02 |
| EP3801543B1 true EP3801543B1 (de) | 2024-09-11 |
Family
ID=68770614
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19814615.1A Active EP3801543B1 (de) | 2018-06-05 | 2019-05-31 | Als hiv-integrase-hemmer nützliche tetracyclische heterocyclenverbindungen |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US11884683B2 (de) |
| EP (1) | EP3801543B1 (de) |
| ES (1) | ES2989103T3 (de) |
| WO (1) | WO2019236396A1 (de) |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4257137A3 (de) | 2018-05-31 | 2023-11-01 | Shionogi & Co., Ltd | Polycyclische carbamoylpyridonderivate zur behandlung von hiv |
| AU2019277547B2 (en) | 2018-05-31 | 2024-10-10 | Shionogi & Co., Ltd. | Polycyclic pyridone derivative |
| WO2020197991A1 (en) | 2019-03-22 | 2020-10-01 | Gilead Sciences, Inc. | Bridged tricyclic carbamoylpyridone compounds and their pharmaceutical use |
| US12577241B2 (en) | 2019-11-28 | 2026-03-17 | Shionogi & Co., Ltd. | Polycyclic pyridopyrazine derivative |
| IL295677B1 (en) | 2020-02-24 | 2026-04-01 | Gilead Sciences Inc | Tetracyclic compounds for the treatment of HIV infection |
| CN112062763B (zh) * | 2020-09-10 | 2021-11-05 | 浙江大学 | 羟基吡啶酮并[1,2-b][1,2,5]三氮卓平衍生物及制备和应用 |
| CA3192145A1 (en) | 2020-09-30 | 2022-04-07 | Gilead Sciences, Inc. | Bridged tricyclic carbamoylpyridone compounds and uses thereof |
| FI4196479T3 (fi) * | 2021-01-19 | 2024-01-17 | Gilead Sciences Inc | Substituoituja pyridotriatsiiniyhdisteitä ja niiden käyttöjä |
| TWI856796B (zh) | 2022-04-06 | 2024-09-21 | 美商基利科學股份有限公司 | 橋聯三環胺甲醯基吡啶酮化合物及其用途 |
Family Cites Families (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2931730B1 (de) * | 2012-12-17 | 2019-08-07 | Merck Sharp & Dohme Corp. | 4-pyridinonetriazinderivate als hiv-integrase-hemmer |
| AU2014267951A1 (en) | 2013-05-17 | 2015-11-19 | Merck Sharp & Dohme Corp. | Fused Tricyclic Heterocyclic compounds as HIV integrase inhibitors |
| WO2016090545A1 (en) | 2014-12-09 | 2016-06-16 | Merck Sharp & Dohme Corp. | Spirocyclic heterocycle compounds useful as hiv integrate inhibitors |
| EP3229804B1 (de) | 2014-12-09 | 2020-05-06 | Merck Sharp & Dohme Corp. | Als hiv-integrase-hemmer nützliche spirocyclische heterocyclische verbindungen |
| WO2016187788A1 (en) * | 2015-05-25 | 2016-12-01 | Merck Sharp & Dohme Corp. | Fused tricyclic heterocyclic compounds useful for treating hiv infection |
| US10548910B2 (en) | 2015-11-17 | 2020-02-04 | Merck Sharp & Dohme Corp. | Amido-substituted pyridotriazine derivatives useful as HIV integrase inhibitors |
| WO2017113288A1 (en) | 2015-12-31 | 2017-07-06 | Merck Sharp & Dohme Corp. | Fused tricyclic heterocyclic compounds as hiv integrase inhibitors |
| JOP20190130A1 (ar) | 2016-12-02 | 2019-06-02 | Merck Sharp & Dohme | مركبات حلقية غير متجانسة رباعية الحلقات مفيدة كمثبطات إنزيم مدمج لفيروس نقص المناعة البشرية (hiv) |
| US10934312B2 (en) | 2016-12-02 | 2021-03-02 | Merck Sharp & Dohme Corp. | Tricyclic heterocycle compounds useful as HIV integrase inhibitors |
| EP3752144B1 (de) | 2018-02-15 | 2022-12-28 | Merck Sharp & Dohme LLC | Als hiv-integrase-hemmer nützliche tricyclische heterocyclenverbindungen |
| CN112088005A (zh) | 2018-04-27 | 2020-12-15 | 默沙东公司 | 可用作hiv整合酶抑制剂的三环杂环化合物 |
-
2019
- 2019-05-31 EP EP19814615.1A patent/EP3801543B1/de active Active
- 2019-05-31 ES ES19814615T patent/ES2989103T3/es active Active
- 2019-05-31 US US17/057,875 patent/US11884683B2/en active Active
- 2019-05-31 WO PCT/US2019/034783 patent/WO2019236396A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| EP3801543A4 (de) | 2022-03-02 |
| WO2019236396A1 (en) | 2019-12-12 |
| ES2989103T3 (es) | 2024-11-25 |
| US11884683B2 (en) | 2024-01-30 |
| EP3801543A1 (de) | 2021-04-14 |
| US20210309671A1 (en) | 2021-10-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3801543B1 (de) | Als hiv-integrase-hemmer nützliche tetracyclische heterocyclenverbindungen | |
| EP3083641B1 (de) | Spirocyclische heterocyclische verbindungen als hiv-integrase-hemmer | |
| EP3752144B1 (de) | Als hiv-integrase-hemmer nützliche tricyclische heterocyclenverbindungen | |
| EP3008044B1 (de) | Kondensierte tricyclische heterocyclische verbindungen als hiv-integrase-hemmer | |
| US9861620B2 (en) | Substituted quinolizine derivatives useful as HIV integrase inhibitors | |
| EP3330272B1 (de) | Kondensierte tricyclische heterocyclische verbindungen als hiv-integrase-hemmer | |
| US10934312B2 (en) | Tricyclic heterocycle compounds useful as HIV integrase inhibitors | |
| US10233193B2 (en) | Fused tricyclic heterocyclic compounds useful for treating HIV infection | |
| EP3548493B1 (de) | Als hiv-integrase-hemmer nützliche tetracyclische heterocyclenverbindungen | |
| EP3397639A1 (de) | Kondensierte tricyclische heterocyclische verbindungen als hiv-integrase-hemmer | |
| CA3101180A1 (en) | Tricyclic heterocycle compounds useful as hiv integrase inhibitors | |
| US20210115044A1 (en) | Tricyclic heterocycle compounds useful as hiv integrase inhibitors | |
| HK40007258A (en) | Tetracyclic heterocycle compounds useful as hiv integrase inhibitors | |
| HK40007258B (en) | Tetracyclic heterocycle compounds useful as hiv integrase inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| TPAC | Observations filed by third parties |
Free format text: ORIGINAL CODE: EPIDOSNTIPA |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20210111 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| A4 | Supplementary search report drawn up and despatched |
Effective date: 20220202 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: A61K 45/06 20060101ALI20220127BHEP Ipc: C07D 491/22 20060101ALI20220127BHEP Ipc: C07D 471/16 20060101ALI20220127BHEP Ipc: A61P 31/12 20060101ALI20220127BHEP Ipc: A61K 31/53 20060101AFI20220127BHEP |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: MERCK SHARP & DOHME LLC |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20231218 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20240419 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602019058806 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: FP |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2989103 Country of ref document: ES Kind code of ref document: T3 Effective date: 20241125 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241211 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241212 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241211 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241211 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241211 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20241212 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1722093 Country of ref document: AT Kind code of ref document: T Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250111 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20250113 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20250421 Year of fee payment: 7 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602019058806 Country of ref document: DE |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20250409 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20250609 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20250512 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20250409 Year of fee payment: 7 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20250601 Year of fee payment: 7 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20250416 Year of fee payment: 7 |
|
| 26N | No opposition filed |
Effective date: 20250612 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20250531 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20250531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20240911 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20260320 Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20250531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20250531 |