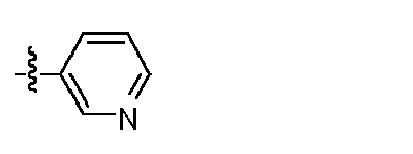EP2923734A1 - Purine analogues and their use as immunosuppressive agents - Google Patents
Purine analogues and their use as immunosuppressive agents Download PDFInfo
- Publication number
- EP2923734A1 EP2923734A1 EP15162142.2A EP15162142A EP2923734A1 EP 2923734 A1 EP2923734 A1 EP 2923734A1 EP 15162142 A EP15162142 A EP 15162142A EP 2923734 A1 EP2923734 A1 EP 2923734A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- amino
- piperazin
- alkyl
- fluorophenyl
- heterocyclic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 239000003018 immunosuppressive agent Substances 0.000 title claims description 20
- 125000000561 purinyl group Chemical class N1=C(N=C2N=CNC2=C1)* 0.000 title description 67
- 229940045686 antimetabolites antineoplastic purine analogs Drugs 0.000 title description 6
- 229940125721 immunosuppressive agent Drugs 0.000 title description 3
- 150000001875 compounds Chemical class 0.000 claims abstract description 141
- 239000003814 drug Substances 0.000 claims abstract description 58
- 208000026278 immune system disease Diseases 0.000 claims abstract description 43
- 210000000056 organ Anatomy 0.000 claims abstract description 37
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 34
- 208000023275 Autoimmune disease Diseases 0.000 claims abstract description 25
- 150000003839 salts Chemical class 0.000 claims abstract description 23
- 239000012453 solvate Substances 0.000 claims abstract description 10
- -1 alkyloximino Chemical class 0.000 claims description 641
- 125000000217 alkyl group Chemical group 0.000 claims description 182
- 125000003282 alkyl amino group Chemical group 0.000 claims description 104
- 125000003118 aryl group Chemical group 0.000 claims description 93
- 150000001408 amides Chemical class 0.000 claims description 81
- 125000001769 aryl amino group Chemical group 0.000 claims description 74
- 229910052736 halogen Inorganic materials 0.000 claims description 68
- 150000002367 halogens Chemical class 0.000 claims description 68
- 125000003396 thiol group Chemical group [H]S* 0.000 claims description 60
- 125000006310 cycloalkyl amino group Chemical group 0.000 claims description 57
- 150000004820 halides Chemical class 0.000 claims description 56
- 150000003254 radicals Chemical class 0.000 claims description 55
- 125000003545 alkoxy group Chemical group 0.000 claims description 54
- 150000008064 anhydrides Chemical class 0.000 claims description 52
- 150000007970 thio esters Chemical class 0.000 claims description 52
- 125000000623 heterocyclic group Chemical group 0.000 claims description 51
- 150000002148 esters Chemical class 0.000 claims description 49
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 49
- 125000001424 substituent group Chemical group 0.000 claims description 49
- 125000005843 halogen group Chemical group 0.000 claims description 45
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 42
- 125000006323 alkenyl amino group Chemical group 0.000 claims description 37
- 125000003342 alkenyl group Chemical group 0.000 claims description 37
- 125000006319 alkynyl amino group Chemical group 0.000 claims description 37
- 125000001691 aryl alkyl amino group Chemical group 0.000 claims description 37
- 125000005191 hydroxyalkylamino group Chemical group 0.000 claims description 36
- 125000000304 alkynyl group Chemical group 0.000 claims description 35
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 34
- 125000004432 carbon atom Chemical group C* 0.000 claims description 34
- 125000000000 cycloalkoxy group Chemical group 0.000 claims description 32
- 229940079593 drug Drugs 0.000 claims description 32
- 125000002252 acyl group Chemical group 0.000 claims description 30
- 125000004659 aryl alkyl thio group Chemical group 0.000 claims description 30
- 125000002102 aryl alkyloxo group Chemical group 0.000 claims description 30
- 125000004104 aryloxy group Chemical group 0.000 claims description 30
- 125000002485 formyl group Chemical class [H]C(*)=O 0.000 claims description 30
- 125000006376 (C3-C10) cycloalkyl group Chemical group 0.000 claims description 28
- 241000124008 Mammalia Species 0.000 claims description 28
- 125000005276 alkyl hydrazino group Chemical class 0.000 claims description 28
- 125000004414 alkyl thio group Chemical class 0.000 claims description 28
- 125000003917 carbamoyl group Chemical class [H]N([H])C(*)=O 0.000 claims description 28
- 125000004442 acylamino group Chemical group 0.000 claims description 27
- 125000005000 thioaryl group Chemical group 0.000 claims description 27
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 26
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims description 25
- 150000003566 thiocarboxylic acids Chemical class 0.000 claims description 24
- 125000000033 alkoxyamino group Chemical class 0.000 claims description 23
- HJMZMZRCABDKKV-UHFFFAOYSA-N carbonocyanidic acid Chemical compound OC(=O)C#N HJMZMZRCABDKKV-UHFFFAOYSA-N 0.000 claims description 22
- 125000001072 heteroaryl group Chemical group 0.000 claims description 22
- 125000000717 hydrazino group Chemical class [H]N([*])N([H])[H] 0.000 claims description 21
- 229910052739 hydrogen Inorganic materials 0.000 claims description 19
- 230000002265 prevention Effects 0.000 claims description 19
- 125000005160 aryl oxy alkyl group Chemical group 0.000 claims description 18
- DHKHKXVYLBGOIT-UHFFFAOYSA-N acetaldehyde Diethyl Acetal Natural products CCOC(C)OCC DHKHKXVYLBGOIT-UHFFFAOYSA-N 0.000 claims description 17
- 125000004947 alkyl aryl amino group Chemical group 0.000 claims description 17
- 230000006870 function Effects 0.000 claims description 17
- 125000005646 oximino group Chemical group 0.000 claims description 17
- 125000003441 thioacyl group Chemical group 0.000 claims description 17
- 150000003556 thioamides Chemical class 0.000 claims description 17
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical group N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 claims description 16
- 125000006852 aliphatic spacer Chemical group 0.000 claims description 16
- 235000001014 amino acid Nutrition 0.000 claims description 16
- 150000001413 amino acids Chemical class 0.000 claims description 16
- 125000004429 atom Chemical group 0.000 claims description 16
- 239000001257 hydrogen Substances 0.000 claims description 16
- 229920006395 saturated elastomer Polymers 0.000 claims description 16
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 16
- 150000003555 thioacetals Chemical class 0.000 claims description 16
- 125000001931 aliphatic group Chemical group 0.000 claims description 15
- 125000005018 aryl alkenyl group Chemical group 0.000 claims description 15
- 150000001733 carboxylic acid esters Chemical class 0.000 claims description 15
- 230000001861 immunosuppressant effect Effects 0.000 claims description 15
- AWIJRPNMLHPLNC-UHFFFAOYSA-N methanethioic s-acid Chemical compound SC=O AWIJRPNMLHPLNC-UHFFFAOYSA-N 0.000 claims description 15
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 15
- 125000005420 sulfonamido group Chemical class S(=O)(=O)(N*)* 0.000 claims description 15
- 238000002054 transplantation Methods 0.000 claims description 15
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 14
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 14
- 229960003444 immunosuppressant agent Drugs 0.000 claims description 14
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 claims description 14
- 125000005842 heteroatom Chemical group 0.000 claims description 13
- 229910052757 nitrogen Inorganic materials 0.000 claims description 13
- 125000002813 thiocarbonyl group Chemical group *C(*)=S 0.000 claims description 13
- 150000001732 carboxylic acid derivatives Chemical class 0.000 claims description 12
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 12
- 239000002955 immunomodulating agent Substances 0.000 claims description 11
- 229940121354 immunomodulator Drugs 0.000 claims description 11
- 230000002584 immunomodulator Effects 0.000 claims description 11
- 229910052760 oxygen Inorganic materials 0.000 claims description 11
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 10
- 125000002877 alkyl aryl group Chemical group 0.000 claims description 10
- 150000005840 aryl radicals Chemical class 0.000 claims description 10
- 125000001316 cycloalkyl alkyl group Chemical group 0.000 claims description 10
- 125000002777 acetyl group Chemical class [H]C([H])([H])C(*)=O 0.000 claims description 9
- 125000004183 alkoxy alkyl group Chemical group 0.000 claims description 9
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 9
- 150000007942 carboxylates Chemical group 0.000 claims description 9
- 125000004985 dialkyl amino alkyl group Chemical group 0.000 claims description 9
- AEOCXXJPGCBFJA-UHFFFAOYSA-N ethionamide Chemical group CCC1=CC(C(N)=S)=CC=N1 AEOCXXJPGCBFJA-UHFFFAOYSA-N 0.000 claims description 9
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 9
- 239000001301 oxygen Chemical group 0.000 claims description 9
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 9
- 229910052717 sulfur Chemical group 0.000 claims description 9
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical group [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 claims description 8
- 229910052799 carbon Inorganic materials 0.000 claims description 8
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 8
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical group NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 claims description 7
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 7
- 125000004423 acyloxy group Chemical group 0.000 claims description 7
- 239000002246 antineoplastic agent Substances 0.000 claims description 7
- 229940041181 antineoplastic drug Drugs 0.000 claims description 7
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 7
- 150000003568 thioethers Chemical class 0.000 claims description 7
- 125000005026 carboxyaryl group Chemical group 0.000 claims description 6
- 125000001841 imino group Chemical group [H]N=* 0.000 claims description 6
- UAJFCVURUYZZSL-UHFFFAOYSA-N (carbamoylamino)carbamothioylcarbamic acid Chemical compound N(C(=O)N)NC(NC(=O)O)=S UAJFCVURUYZZSL-UHFFFAOYSA-N 0.000 claims description 5
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 claims description 5
- 239000011593 sulfur Chemical group 0.000 claims description 5
- 125000003341 7 membered heterocyclic group Chemical group 0.000 claims description 4
- 125000005257 alkyl acyl group Chemical group 0.000 claims description 4
- 125000005251 aryl acyl group Chemical group 0.000 claims description 4
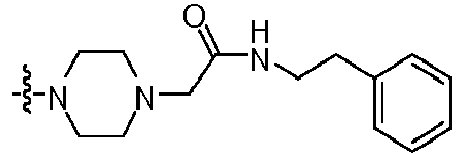
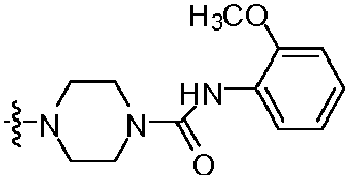
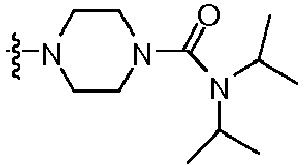
- IOUKLJNBQDYRIB-UHFFFAOYSA-N 1-[4-(2-amino-8-benzylsulfanyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(SCC=3C=CC=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 IOUKLJNBQDYRIB-UHFFFAOYSA-N 0.000 claims description 3
- GVDORJZDLXJLLC-UHFFFAOYSA-N 1-[4-(2-amino-8-cyclopentylsulfanyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(SC3CCCC3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 GVDORJZDLXJLLC-UHFFFAOYSA-N 0.000 claims description 3
- YLBSGBSYQLDSSL-UHFFFAOYSA-N 1-[4-(2-amino-8-cyclopropyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3CC3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 YLBSGBSYQLDSSL-UHFFFAOYSA-N 0.000 claims description 3
- ZGTJRVRNJHZDLH-UHFFFAOYSA-N 1-[4-(2-amino-8-methyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2NC(C)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZGTJRVRNJHZDLH-UHFFFAOYSA-N 0.000 claims description 3
- ZXDSFURVENMPMT-UHFFFAOYSA-N 1-[4-(2-amino-8-methylsulfanyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2NC(SC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZXDSFURVENMPMT-UHFFFAOYSA-N 0.000 claims description 3
- HYVRVCOMRAOJDK-UHFFFAOYSA-N 1-[4-(2-amino-8-propyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2NC(CCC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 HYVRVCOMRAOJDK-UHFFFAOYSA-N 0.000 claims description 3
- LESAQKWQUCDXPW-UHFFFAOYSA-N 1-[4-(2-amino-8-propylsulfanyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2NC(SCCC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 LESAQKWQUCDXPW-UHFFFAOYSA-N 0.000 claims description 3
- URNVXIZJMDWEHN-UHFFFAOYSA-N 1-[4-(2-amino-8-tert-butyl-7h-purin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2NC(C(C)(C)C)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 URNVXIZJMDWEHN-UHFFFAOYSA-N 0.000 claims description 3
- ZDGQRAWDQIUTKI-UHFFFAOYSA-N 1-[4-(2-amino-9-methyl-8-methylsulfanylpurin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2N(C)C(SC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZDGQRAWDQIUTKI-UHFFFAOYSA-N 0.000 claims description 3
- WAVVXNVGFPXSCD-UHFFFAOYSA-N 1-[4-[2-amino-8-(2-phenylethylsulfanyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(SCCC=3C=CC=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 WAVVXNVGFPXSCD-UHFFFAOYSA-N 0.000 claims description 3
- WDRHYMDLFWFQFE-UHFFFAOYSA-N 1-[4-[2-amino-8-(3,4-dimethoxyphenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C1=C(OC)C(OC)=CC=C1C1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=C(N)N=C2N1 WDRHYMDLFWFQFE-UHFFFAOYSA-N 0.000 claims description 3
- FDWOKRZSLGPTBG-UHFFFAOYSA-N 1-[4-[2-amino-8-(3-chlorophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=C(Cl)C=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 FDWOKRZSLGPTBG-UHFFFAOYSA-N 0.000 claims description 3
- IOEBTBZVJQTXQV-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-bromophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(Br)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 IOEBTBZVJQTXQV-UHFFFAOYSA-N 0.000 claims description 3
- QUQMEEYGORZRTH-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-chlorophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(Cl)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 QUQMEEYGORZRTH-UHFFFAOYSA-N 0.000 claims description 3
- DNZVKNGGVATVPL-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]-1,4-diazepan-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCCN1C(=O)COC1=CC=C(Cl)C=C1 DNZVKNGGVATVPL-UHFFFAOYSA-N 0.000 claims description 3
- GKLIAQTVQNVXGM-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 GKLIAQTVQNVXGM-UHFFFAOYSA-N 0.000 claims description 3
- VQWCESHIRHWTOD-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-phenoxyethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC=C1 VQWCESHIRHWTOD-UHFFFAOYSA-N 0.000 claims description 3
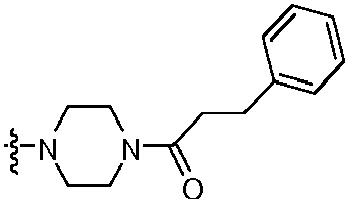
- ANIZKKQNDMMBMZ-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-3-phenylpropan-1-one Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCC1=CC=CC=C1 ANIZKKQNDMMBMZ-UHFFFAOYSA-N 0.000 claims description 3
- SJEYVDWEQVJKAL-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]ethanone Chemical compound C1CN(C(=O)C)CCN1C1=NC(N)=NC2=C1N=C(C=1C=CC(F)=CC=1)N2 SJEYVDWEQVJKAL-UHFFFAOYSA-N 0.000 claims description 3
- KSASEXVJPIPSDF-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-9-methylpurin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2N(C)C(C=3C=CC(F)=CC=3)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 KSASEXVJPIPSDF-UHFFFAOYSA-N 0.000 claims description 3
- ANFGFYABVFGTFG-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-methylphenyl)-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C1=CC(C)=CC=C1C1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=C(N)N=C2N1 ANFGFYABVFGTFG-UHFFFAOYSA-N 0.000 claims description 3
- MPYXGDVWYORQLA-UHFFFAOYSA-N 1-[4-[2-amino-8-[4-(trifluoromethoxy)phenyl]-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(OC(F)(F)F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 MPYXGDVWYORQLA-UHFFFAOYSA-N 0.000 claims description 3
- OZJWOWGFJNFARS-UHFFFAOYSA-N 1-[4-[2-amino-8-[4-(trifluoromethyl)phenyl]-7h-purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(=CC=3)C(F)(F)F)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 OZJWOWGFJNFARS-UHFFFAOYSA-N 0.000 claims description 3
- LHXCVBNRLGZMEN-UHFFFAOYSA-N 1-[4-[2-amino-9-benzyl-8-(4-fluorophenyl)purin-6-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)N(CC=3C=CC=CC=3)C2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 LHXCVBNRLGZMEN-UHFFFAOYSA-N 0.000 claims description 3
- ZLHOYOKEKGKMSG-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-1-morpholin-4-ylethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC(=O)N1CCOCC1 ZLHOYOKEKGKMSG-UHFFFAOYSA-N 0.000 claims description 3
- ZNZJDQVICNNAMH-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-1-pyrrolidin-1-ylethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC(=O)N1CCCC1 ZNZJDQVICNNAMH-UHFFFAOYSA-N 0.000 claims description 3
- ZVNNWLPUFCUROH-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-n-(1,3-thiazol-2-yl)acetamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC(=O)NC1=NC=CS1 ZVNNWLPUFCUROH-UHFFFAOYSA-N 0.000 claims description 3
- JSQVSRNGBHAUTR-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-n-methyl-n-phenylacetamide Chemical compound C=1C=CC=CC=1N(C)C(=O)CN(CC1)CCN1C(C=1N=2)=NC(N)=NC=1NC=2C1=CC=C(F)C=C1 JSQVSRNGBHAUTR-UHFFFAOYSA-N 0.000 claims description 3
- ZHUCXZUDGSFHRR-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-n-pyridin-2-ylacetamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC(=O)NC1=CC=CC=N1 ZHUCXZUDGSFHRR-UHFFFAOYSA-N 0.000 claims description 3
- KYDYULPQMOZORL-UHFFFAOYSA-N 2-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-n-pyridin-3-ylacetamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC(=O)NC1=CC=CN=C1 KYDYULPQMOZORL-UHFFFAOYSA-N 0.000 claims description 3
- XWNDUWVHUAYRRB-UHFFFAOYSA-N 4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]-n-(3-methylphenyl)-1,4-diazepane-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CCC2)C=2C=3N=C(NC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 XWNDUWVHUAYRRB-UHFFFAOYSA-N 0.000 claims description 3
- KQMPBNYANZUHJC-UHFFFAOYSA-N 6-(1,4-diazepan-1-yl)-8-(4-fluorophenyl)-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N1CCCNCC1 KQMPBNYANZUHJC-UHFFFAOYSA-N 0.000 claims description 3
- WFCDDGCDORMVSX-UHFFFAOYSA-N 6-(4-benzylsulfonylpiperazin-1-yl)-8-(4-fluorophenyl)-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1S(=O)(=O)CC1=CC=CC=C1 WFCDDGCDORMVSX-UHFFFAOYSA-N 0.000 claims description 3
- KYOYWKQEYDVFDF-UHFFFAOYSA-N 6-[4-(4-chlorophenyl)piperazin-1-yl]-8-(4-fluorophenyl)-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C1=CC=C(Cl)C=C1 KYOYWKQEYDVFDF-UHFFFAOYSA-N 0.000 claims description 3
- LLYNBBMSVVHPDH-UHFFFAOYSA-N 8-(4-fluorophenyl)-6-(4-pyridin-4-ylpiperazin-1-yl)-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C1=CC=NC=C1 LLYNBBMSVVHPDH-UHFFFAOYSA-N 0.000 claims description 3
- WZUPOVBJDVBHBF-UHFFFAOYSA-N 8-(4-fluorophenyl)-6-[4-(1,3-thiazol-2-yl)piperazin-1-yl]-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C1=NC=CS1 WZUPOVBJDVBHBF-UHFFFAOYSA-N 0.000 claims description 3
- UIUXHYDISLPXPU-UHFFFAOYSA-N 8-(4-fluorophenyl)-6-[4-(4-fluorophenyl)piperazin-1-yl]-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C1=CC=C(F)C=C1 UIUXHYDISLPXPU-UHFFFAOYSA-N 0.000 claims description 3
- CMRRBCRDZQRYSA-UHFFFAOYSA-N 8-(4-fluorophenyl)-6-[4-[(4-fluorophenyl)methyl]piperazin-1-yl]-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1CC1=CC=C(F)C=C1 CMRRBCRDZQRYSA-UHFFFAOYSA-N 0.000 claims description 3
- IAHMVZVRRIVXRK-UHFFFAOYSA-N 8-(4-fluorophenyl)-6-piperazin-1-yl-7h-purin-2-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N1CCNCC1 IAHMVZVRRIVXRK-UHFFFAOYSA-N 0.000 claims description 3
- KPQKKQDXKWGXKY-UHFFFAOYSA-N [4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-(3-methoxyphenyl)methanone Chemical compound COC1=CC=CC(C(=O)N2CCN(CC2)C=2C=3N=C(NC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 KPQKKQDXKWGXKY-UHFFFAOYSA-N 0.000 claims description 3
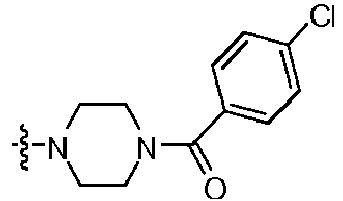
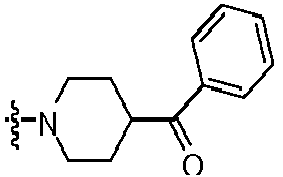
- FCRYKXXNLBDWDD-UHFFFAOYSA-N [4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-(4-chlorophenyl)methanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)C1=CC=C(Cl)C=C1 FCRYKXXNLBDWDD-UHFFFAOYSA-N 0.000 claims description 3
- SEUIDDVFWVCCCJ-UHFFFAOYSA-N [4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-naphthalen-1-ylmethanone Chemical compound N1C2=NC(N)=NC(N3CCN(CC3)C(=O)C=3C4=CC=CC=C4C=CC=3)=C2N=C1C1=CC=C(F)C=C1 SEUIDDVFWVCCCJ-UHFFFAOYSA-N 0.000 claims description 3
- 125000005115 alkyl carbamoyl group Chemical group 0.000 claims description 3
- 125000004390 alkyl sulfonyl group Chemical group 0.000 claims description 3
- 125000003435 aroyl group Chemical group 0.000 claims description 3
- 125000005116 aryl carbamoyl group Chemical group 0.000 claims description 3
- 125000005161 aryl oxy carbonyl group Chemical group 0.000 claims description 3
- ABKYBCYESRQSQH-UHFFFAOYSA-N 1-[4-(2-amino-8-sulfanylidene-7,9-dihydropurin-6-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12NC(=S)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ABKYBCYESRQSQH-UHFFFAOYSA-N 0.000 claims description 2
- YSEHAQZPUFGSCA-UHFFFAOYSA-N 1-[4-[2-amino-8-(4-fluorophenyl)-7h-purin-6-yl]piperazin-1-yl]-2-thiophen-2-ylethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)NC2=NC(N)=NC=1N(CC1)CCN1C(=O)CC1=CC=CS1 YSEHAQZPUFGSCA-UHFFFAOYSA-N 0.000 claims description 2
- 125000000446 sulfanediyl group Chemical group *S* 0.000 claims 8
- 238000000034 method Methods 0.000 abstract description 61
- 238000002360 preparation method Methods 0.000 abstract description 36
- 206010052779 Transplant rejections Diseases 0.000 abstract description 16
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 abstract description 12
- 239000004480 active ingredient Substances 0.000 abstract description 8
- 208000035475 disorder Diseases 0.000 abstract description 5
- 230000001575 pathological effect Effects 0.000 abstract description 5
- 230000015572 biosynthetic process Effects 0.000 description 86
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 81
- 238000003786 synthesis reaction Methods 0.000 description 81
- CDGFEINVQHEUQV-UHFFFAOYSA-N [1,3]thiazolo[5,4-d]pyrimidine Chemical compound N1=CN=C2SC=NC2=C1 CDGFEINVQHEUQV-UHFFFAOYSA-N 0.000 description 77
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 72
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 60
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 60
- 238000006243 chemical reaction Methods 0.000 description 56
- BRIOKNPDCPJCOD-UHFFFAOYSA-N [1,3]oxazolo[5,4-d]pyrimidine Chemical compound N1=CN=C2OC=NC2=C1 BRIOKNPDCPJCOD-UHFFFAOYSA-N 0.000 description 55
- DDWBRNXDKNIQDY-UHFFFAOYSA-N thieno[2,3-d]pyrimidine Chemical compound N1=CN=C2SC=CC2=C1 DDWBRNXDKNIQDY-UHFFFAOYSA-N 0.000 description 55
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 54
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 52
- 238000005160 1H NMR spectroscopy Methods 0.000 description 50
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 44
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 35
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 34
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 34
- 125000004149 thio group Chemical group *S* 0.000 description 34
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 30
- 210000004027 cell Anatomy 0.000 description 30
- 239000007787 solid Substances 0.000 description 29
- 229940083251 peripheral vasodilators purine derivative Drugs 0.000 description 28
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 27
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 26
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 26
- 239000000243 solution Substances 0.000 description 26
- 229910001868 water Inorganic materials 0.000 description 25
- 241001465754 Metazoa Species 0.000 description 24
- 239000002904 solvent Substances 0.000 description 24
- 239000003153 chemical reaction reagent Substances 0.000 description 23
- 239000000203 mixture Substances 0.000 description 23
- 125000004193 piperazinyl group Chemical group 0.000 description 23
- 239000011541 reaction mixture Substances 0.000 description 23
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 23
- 238000003818 flash chromatography Methods 0.000 description 22
- 239000000377 silicon dioxide Substances 0.000 description 22
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 21
- 239000002253 acid Substances 0.000 description 21
- 210000001744 T-lymphocyte Anatomy 0.000 description 20
- 230000004913 activation Effects 0.000 description 20
- 230000000694 effects Effects 0.000 description 20
- 239000012038 nucleophile Substances 0.000 description 19
- 239000002585 base Substances 0.000 description 18
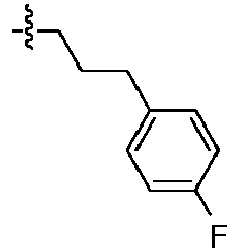
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 17
- 238000003556 assay Methods 0.000 description 17
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 17
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 16
- 239000003795 chemical substances by application Substances 0.000 description 16
- 230000008878 coupling Effects 0.000 description 16
- 238000010168 coupling process Methods 0.000 description 16
- 238000005859 coupling reaction Methods 0.000 description 16
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical group C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 description 15
- 238000007280 thionation reaction Methods 0.000 description 15
- 230000001506 immunosuppresive effect Effects 0.000 description 14
- 210000004698 lymphocyte Anatomy 0.000 description 14
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 14
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 14
- 125000003944 tolyl group Chemical group 0.000 description 14
- 150000001241 acetals Chemical class 0.000 description 13
- 239000000460 chlorine Substances 0.000 description 13
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical group OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 12
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 12
- 235000019441 ethanol Nutrition 0.000 description 12
- 230000002829 reductive effect Effects 0.000 description 12
- 239000004215 Carbon black (E152) Substances 0.000 description 11
- 239000007832 Na2SO4 Substances 0.000 description 11
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 11
- 235000019439 ethyl acetate Nutrition 0.000 description 11
- 229930195733 hydrocarbon Natural products 0.000 description 11
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 11
- 125000004194 piperazin-1-yl group Chemical group [H]N1C([H])([H])C([H])([H])N(*)C([H])([H])C1([H])[H] 0.000 description 11
- 229910052938 sodium sulfate Inorganic materials 0.000 description 11
- 230000002195 synergetic effect Effects 0.000 description 11
- 125000004055 thiomethyl group Chemical group [H]SC([H])([H])* 0.000 description 11
- FPIRBHDGWMWJEP-UHFFFAOYSA-N 1-hydroxy-7-azabenzotriazole Chemical compound C1=CN=C2N(O)N=NC2=C1 FPIRBHDGWMWJEP-UHFFFAOYSA-N 0.000 description 10
- GQHTUMJGOHRCHB-UHFFFAOYSA-N 2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepine Chemical compound C1CCCCN2CCCN=C21 GQHTUMJGOHRCHB-UHFFFAOYSA-N 0.000 description 10
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 10
- 239000000654 additive Substances 0.000 description 10
- RROBIDXNTUAHFW-UHFFFAOYSA-N benzotriazol-1-yloxy-tris(dimethylamino)phosphanium Chemical compound C1=CC=C2N(O[P+](N(C)C)(N(C)C)N(C)C)N=NC2=C1 RROBIDXNTUAHFW-UHFFFAOYSA-N 0.000 description 10
- 238000007339 nucleophilic aromatic substitution reaction Methods 0.000 description 10
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 10
- CYQAYERJWZKYML-UHFFFAOYSA-N phosphorus pentasulfide Chemical group S1P(S2)(=S)SP3(=S)SP1(=S)SP2(=S)S3 CYQAYERJWZKYML-UHFFFAOYSA-N 0.000 description 10
- 230000008569 process Effects 0.000 description 10
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 9
- 150000003951 lactams Chemical group 0.000 description 9
- 125000002757 morpholinyl group Chemical group 0.000 description 9
- 229940002612 prodrug Drugs 0.000 description 9
- 239000000651 prodrug Substances 0.000 description 9
- 239000011734 sodium Substances 0.000 description 9
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 8
- 108010036949 Cyclosporine Proteins 0.000 description 8
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 8
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 8
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 8
- 230000010933 acylation Effects 0.000 description 8
- 238000005917 acylation reaction Methods 0.000 description 8
- 125000003277 amino group Chemical group 0.000 description 8
- 230000002519 immonomodulatory effect Effects 0.000 description 8
- 238000004519 manufacturing process Methods 0.000 description 8
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 8
- 238000007363 ring formation reaction Methods 0.000 description 8
- 229940083542 sodium Drugs 0.000 description 8
- 229910052708 sodium Inorganic materials 0.000 description 8
- 238000012360 testing method Methods 0.000 description 8
- NQELVURVHKIMJG-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 NQELVURVHKIMJG-UHFFFAOYSA-N 0.000 description 7
- 102000004190 Enzymes Human genes 0.000 description 7
- 108090000790 Enzymes Proteins 0.000 description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 7
- 206010062016 Immunosuppression Diseases 0.000 description 7
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 7
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Natural products NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 7
- 230000002378 acidificating effect Effects 0.000 description 7
- 150000001350 alkyl halides Chemical class 0.000 description 7
- 238000001816 cooling Methods 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 230000026030 halogenation Effects 0.000 description 7
- 238000005658 halogenation reaction Methods 0.000 description 7
- 238000001727 in vivo Methods 0.000 description 7
- 239000003112 inhibitor Substances 0.000 description 7
- CFHGBZLNZZVTAY-UHFFFAOYSA-N lawesson's reagent Chemical compound C1=CC(OC)=CC=C1P1(=S)SP(=S)(C=2C=CC(OC)=CC=2)S1 CFHGBZLNZZVTAY-UHFFFAOYSA-N 0.000 description 7
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Natural products C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 7
- 125000002816 methylsulfanyl group Chemical group [H]C([H])([H])S[*] 0.000 description 7
- 238000007254 oxidation reaction Methods 0.000 description 7
- 238000010651 palladium-catalyzed cross coupling reaction Methods 0.000 description 7
- 239000000047 product Substances 0.000 description 7
- 239000007858 starting material Substances 0.000 description 7
- 239000004094 surface-active agent Substances 0.000 description 7
- 239000008096 xylene Substances 0.000 description 7
- BDNKZNFMNDZQMI-UHFFFAOYSA-N 1,3-diisopropylcarbodiimide Chemical compound CC(C)N=C=NC(C)C BDNKZNFMNDZQMI-UHFFFAOYSA-N 0.000 description 6
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 6
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 6
- 125000004189 3,4-dichlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(Cl)C([H])=C1* 0.000 description 6
- 125000004207 3-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(OC([H])([H])[H])=C1[H] 0.000 description 6
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- 239000000427 antigen Substances 0.000 description 6
- 150000001543 aryl boronic acids Chemical class 0.000 description 6
- 230000001363 autoimmune Effects 0.000 description 6
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Chemical compound N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 6
- 238000009835 boiling Methods 0.000 description 6
- 239000012267 brine Substances 0.000 description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 6
- 229910052794 bromium Inorganic materials 0.000 description 6
- 150000001721 carbon Chemical group 0.000 description 6
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 6
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 6
- 229930182912 cyclosporin Natural products 0.000 description 6
- 125000004986 diarylamino group Chemical group 0.000 description 6
- 239000003937 drug carrier Substances 0.000 description 6
- 238000000338 in vitro Methods 0.000 description 6
- 239000000543 intermediate Substances 0.000 description 6
- 210000002540 macrophage Anatomy 0.000 description 6
- 230000001404 mediated effect Effects 0.000 description 6
- 125000000714 pyrimidinyl group Chemical group 0.000 description 6
- 239000000741 silica gel Substances 0.000 description 6
- 229910002027 silica gel Inorganic materials 0.000 description 6
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 6
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea Chemical compound NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 6
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 5
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 5
- 125000004176 4-fluorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1F)C([H])([H])* 0.000 description 5
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 5
- 241000282412 Homo Species 0.000 description 5
- 108010002350 Interleukin-2 Proteins 0.000 description 5
- 102000000588 Interleukin-2 Human genes 0.000 description 5
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 5
- 150000001299 aldehydes Chemical class 0.000 description 5
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 5
- 108091007433 antigens Proteins 0.000 description 5
- 102000036639 antigens Human genes 0.000 description 5
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 5
- 229910052801 chlorine Inorganic materials 0.000 description 5
- 150000001805 chlorine compounds Chemical class 0.000 description 5
- 229960001265 ciclosporin Drugs 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 5
- 239000004615 ingredient Substances 0.000 description 5
- 210000004185 liver Anatomy 0.000 description 5
- DGGJQLCAYQCPDD-UHFFFAOYSA-N methyl 2-aminothiophene-3-carboxylate Chemical compound COC(=O)C=1C=CSC=1N DGGJQLCAYQCPDD-UHFFFAOYSA-N 0.000 description 5
- 125000002950 monocyclic group Chemical group 0.000 description 5
- 125000000018 nitroso group Chemical group N(=O)* 0.000 description 5
- 239000002736 nonionic surfactant Substances 0.000 description 5
- 231100000252 nontoxic Toxicity 0.000 description 5
- 230000003000 nontoxic effect Effects 0.000 description 5
- 125000004043 oxo group Chemical group O=* 0.000 description 5
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 5
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 5
- 125000003386 piperidinyl group Chemical group 0.000 description 5
- 125000003367 polycyclic group Chemical group 0.000 description 5
- 239000002244 precipitate Substances 0.000 description 5
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 5
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 5
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 5
- 229960002930 sirolimus Drugs 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 231100000419 toxicity Toxicity 0.000 description 5
- 230000001988 toxicity Effects 0.000 description 5
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 4
- 101000914514 Homo sapiens T-cell-specific surface glycoprotein CD28 Proteins 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical group [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical group CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 4
- 206010039897 Sedation Diseases 0.000 description 4
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical group O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 4
- 102100027213 T-cell-specific surface glycoprotein CD28 Human genes 0.000 description 4
- 150000007513 acids Chemical class 0.000 description 4
- 238000005804 alkylation reaction Methods 0.000 description 4
- 230000001028 anti-proliverative effect Effects 0.000 description 4
- 238000004587 chromatography analysis Methods 0.000 description 4
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 4
- 210000004443 dendritic cell Anatomy 0.000 description 4
- 238000001212 derivatisation Methods 0.000 description 4
- 150000002170 ethers Chemical class 0.000 description 4
- 229940093499 ethyl acetate Drugs 0.000 description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 4
- 150000004665 fatty acids Chemical class 0.000 description 4
- 238000010438 heat treatment Methods 0.000 description 4
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 4
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 4
- 208000027866 inflammatory disease Diseases 0.000 description 4
- 230000003993 interaction Effects 0.000 description 4
- 239000012044 organic layer Substances 0.000 description 4
- 230000003647 oxidation Effects 0.000 description 4
- 239000002798 polar solvent Substances 0.000 description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 description 4
- 229930195734 saturated hydrocarbon Natural products 0.000 description 4
- 230000036280 sedation Effects 0.000 description 4
- 150000003457 sulfones Chemical class 0.000 description 4
- 150000003462 sulfoxides Chemical class 0.000 description 4
- HJUGFYREWKUQJT-UHFFFAOYSA-N tetrabromomethane Chemical compound BrC(Br)(Br)Br HJUGFYREWKUQJT-UHFFFAOYSA-N 0.000 description 4
- 125000000335 thiazolyl group Chemical group 0.000 description 4
- WYWHKKSPHMUBEB-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 4
- 239000003039 volatile agent Substances 0.000 description 4
- 238000002689 xenotransplantation Methods 0.000 description 4
- BUTTVUBNXIIXRH-UHFFFAOYSA-N 1-(cyclopropylmethyl)pyrazole-4-carbaldehyde Chemical compound C1=C(C=O)C=NN1CC1CC1 BUTTVUBNXIIXRH-UHFFFAOYSA-N 0.000 description 3
- XXNYVMZIPHCWCM-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Br)C=C1 XXNYVMZIPHCWCM-UHFFFAOYSA-N 0.000 description 3
- KURFWIPHTYUQTR-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 KURFWIPHTYUQTR-UHFFFAOYSA-N 0.000 description 3
- AFPOCDSWTQIMCH-UHFFFAOYSA-N 1-[4-[5-amino-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 AFPOCDSWTQIMCH-UHFFFAOYSA-N 0.000 description 3
- JJAZUDGUCBHPLM-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenoxy)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCOC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 JJAZUDGUCBHPLM-UHFFFAOYSA-N 0.000 description 3
- YAXXQUZQCLSGFL-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 YAXXQUZQCLSGFL-UHFFFAOYSA-N 0.000 description 3
- DBFCIRAYUIGTEY-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 DBFCIRAYUIGTEY-UHFFFAOYSA-N 0.000 description 3
- HWSJQFCTYLBBOF-UHFFFAOYSA-N 2,5-diamino-4-hydroxy-1h-pyrimidin-6-one Chemical compound NC1=NC(O)=C(N)C(O)=N1 HWSJQFCTYLBBOF-UHFFFAOYSA-N 0.000 description 3
- DGHLAFBJSWAJIZ-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-(4-fluorophenyl)-5-methyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(C)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 DGHLAFBJSWAJIZ-UHFFFAOYSA-N 0.000 description 3
- TYCJFZIWSCJVJW-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-piperazin-1-ylethanone Chemical compound C1=CC(Cl)=CC=C1OCC(=O)N1CCNCC1 TYCJFZIWSCJVJW-UHFFFAOYSA-N 0.000 description 3
- RFUYQLOLZSUJLA-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-(2-methoxyethoxy)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(OCCOC)=NC(N)=NC=2SC=1C1=CC=C(F)C=C1 RFUYQLOLZSUJLA-UHFFFAOYSA-N 0.000 description 3
- VMNXWHGGIWMLBQ-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-(4-pentylpiperazin-1-yl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C1CN(CCCCC)CCN1C1=NC(N)=NC2=C1N=C(C=1C=CC(F)=CC=1)S2 VMNXWHGGIWMLBQ-UHFFFAOYSA-N 0.000 description 3
- KEJIHXKRBKXUJJ-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-[4-(1,3-thiazol-2-yl)piperazin-1-yl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C1=NC=CS1 KEJIHXKRBKXUJJ-UHFFFAOYSA-N 0.000 description 3
- RSTBWUBZEGOJOB-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-[4-(3-methylphenyl)piperazin-1-yl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound CC1=CC=CC(N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 RSTBWUBZEGOJOB-UHFFFAOYSA-N 0.000 description 3
- FJIYBSRUHOAVDM-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C1=CC=C(F)C=C1 FJIYBSRUHOAVDM-UHFFFAOYSA-N 0.000 description 3
- FJBRVQYRRWGYHD-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-methylsulfonyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(S(=O)(=O)C)=NC(N)=NC=2SC=1C1=CC=C(F)C=C1 FJBRVQYRRWGYHD-UHFFFAOYSA-N 0.000 description 3
- QJFJCCRSKJPSMC-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-morpholin-4-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCOCC1 QJFJCCRSKJPSMC-UHFFFAOYSA-N 0.000 description 3
- RATQQHHPXFJWAE-UHFFFAOYSA-N 2-[(4-fluorophenyl)methyl]-7-morpholin-4-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCOCC1 RATQQHHPXFJWAE-UHFFFAOYSA-N 0.000 description 3
- PKALSDULVHJCMK-UHFFFAOYSA-N 2-[(4-fluorophenyl)methyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 PKALSDULVHJCMK-UHFFFAOYSA-N 0.000 description 3
- IWARXXBAUZGEAF-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-methylsulfonyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(S(=O)(=O)C)=NC(N)=NC=2SC=1CCC1=CC=C(F)C=C1 IWARXXBAUZGEAF-UHFFFAOYSA-N 0.000 description 3
- AYUCYQYSRFCJNO-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 AYUCYQYSRFCJNO-UHFFFAOYSA-N 0.000 description 3
- NSBAUMIEYZOCFH-UHFFFAOYSA-N 2-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-1-morpholin-4-ylethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1CC(=O)N1CCOCC1 NSBAUMIEYZOCFH-UHFFFAOYSA-N 0.000 description 3
- IJYHFOIORVYBLB-UHFFFAOYSA-N 4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3C=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)=C1 IJYHFOIORVYBLB-UHFFFAOYSA-N 0.000 description 3
- FPESSAJSCCWLRY-UHFFFAOYSA-N 4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylic acid Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)O)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 FPESSAJSCCWLRY-UHFFFAOYSA-N 0.000 description 3
- VOZLTVHYIVPTSW-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 VOZLTVHYIVPTSW-UHFFFAOYSA-N 0.000 description 3
- JGYOQHAVAOOBHB-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(4-methylphenyl)piperazine-1-carboxamide Chemical compound C1=CC(C)=CC=C1NC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)CC1 JGYOQHAVAOOBHB-UHFFFAOYSA-N 0.000 description 3
- ATOHKUMFBXFQRR-UHFFFAOYSA-N 4-[5-amino-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(CC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)=C1 ATOHKUMFBXFQRR-UHFFFAOYSA-N 0.000 description 3
- MYPZDIIFCMVUOZ-UHFFFAOYSA-N 5-amino-2-(4-fluorophenyl)-6h-[1,3]thiazolo[5,4-d]pyrimidine-7-thione Chemical compound S1C2=NC(N)=NC(S)=C2N=C1C1=CC=C(F)C=C1 MYPZDIIFCMVUOZ-UHFFFAOYSA-N 0.000 description 3
- SHDNTOJIAPVOQC-UHFFFAOYSA-N 7-(4-benzylpiperazin-1-yl)-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1CC1=CC=CC=C1 SHDNTOJIAPVOQC-UHFFFAOYSA-N 0.000 description 3
- YHSAVZQAAIOMFG-UHFFFAOYSA-N 7-[4-(benzenesulfonyl)piperazin-1-yl]-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1S(=O)(=O)C1=CC=CC=C1 YHSAVZQAAIOMFG-UHFFFAOYSA-N 0.000 description 3
- KPDJDLDLQWMEEF-UHFFFAOYSA-N 7-ethoxy-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(OCC)=NC(N)=NC=2SC=1C1=CC=C(F)C=C1 KPDJDLDLQWMEEF-UHFFFAOYSA-N 0.000 description 3
- SGSJTIHRAWXBGQ-UHFFFAOYSA-N 7-ethoxy-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(OCC)=NC(N)=NC=2SC=1CC1=CC=C(F)C=C1 SGSJTIHRAWXBGQ-UHFFFAOYSA-N 0.000 description 3
- 238000006443 Buchwald-Hartwig cross coupling reaction Methods 0.000 description 3
- 101150013553 CD40 gene Proteins 0.000 description 3
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 3
- 229930105110 Cyclosporin A Natural products 0.000 description 3
- 102000004127 Cytokines Human genes 0.000 description 3
- 108090000695 Cytokines Proteins 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- IVDFJHOHABJVEH-UHFFFAOYSA-N HOCMe2CMe2OH Natural products CC(C)(O)C(C)(C)O IVDFJHOHABJVEH-UHFFFAOYSA-N 0.000 description 3
- 238000007341 Heck reaction Methods 0.000 description 3
- 108010074328 Interferon-gamma Proteins 0.000 description 3
- 108010065805 Interleukin-12 Proteins 0.000 description 3
- 238000005577 Kumada cross-coupling reaction Methods 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 241000699670 Mus sp. Species 0.000 description 3
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 3
- 238000006411 Negishi coupling reaction Methods 0.000 description 3
- 206010028980 Neoplasm Diseases 0.000 description 3
- 229910019142 PO4 Inorganic materials 0.000 description 3
- 229910019213 POCl3 Inorganic materials 0.000 description 3
- 208000002193 Pain Diseases 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- 201000004681 Psoriasis Diseases 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 241000283984 Rodentia Species 0.000 description 3
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 3
- 238000006069 Suzuki reaction reaction Methods 0.000 description 3
- 230000006044 T cell activation Effects 0.000 description 3
- 102100040245 Tumor necrosis factor receptor superfamily member 5 Human genes 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 230000001780 adrenocortical effect Effects 0.000 description 3
- 150000001336 alkenes Chemical group 0.000 description 3
- 150000001345 alkine derivatives Chemical group 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- 150000003863 ammonium salts Chemical class 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 229940045688 antineoplastic antimetabolites pyrimidine analogues Drugs 0.000 description 3
- 150000004982 aromatic amines Chemical class 0.000 description 3
- 150000001502 aryl halides Chemical class 0.000 description 3
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 3
- 229960002170 azathioprine Drugs 0.000 description 3
- 125000003785 benzimidazolyl group Chemical class N1=C(NC2=C1C=CC=C2)* 0.000 description 3
- WBNIMWDRBIBUEK-UHFFFAOYSA-N benzyl 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazine-1-carboxylate Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)OCC1=CC=CC=C1 WBNIMWDRBIBUEK-UHFFFAOYSA-N 0.000 description 3
- 230000004071 biological effect Effects 0.000 description 3
- 230000037396 body weight Effects 0.000 description 3
- 201000011510 cancer Diseases 0.000 description 3
- 150000001735 carboxylic acids Chemical class 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 206010012601 diabetes mellitus Diseases 0.000 description 3
- LQDJGPTUSMDFBA-UHFFFAOYSA-N diethyl 2-[(4-fluorobenzoyl)amino]propanedioate Chemical compound CCOC(=O)C(C(=O)OCC)NC(=O)C1=CC=C(F)C=C1 LQDJGPTUSMDFBA-UHFFFAOYSA-N 0.000 description 3
- GLFVNTDRBTZJIY-UHFFFAOYSA-N diethyl 2-aminopropanedioate;hydron;chloride Chemical compound Cl.CCOC(=O)C(N)C(=O)OCC GLFVNTDRBTZJIY-UHFFFAOYSA-N 0.000 description 3
- MHDFJESNGMDHQD-UHFFFAOYSA-N dimethyl 2-aminopropanedioate Chemical compound COC(=O)C(N)C(=O)OC MHDFJESNGMDHQD-UHFFFAOYSA-N 0.000 description 3
- 229940052303 ethers for general anesthesia Drugs 0.000 description 3
- 239000003349 gelling agent Substances 0.000 description 3
- 150000004795 grignard reagents Chemical class 0.000 description 3
- 210000002216 heart Anatomy 0.000 description 3
- 150000002430 hydrocarbons Chemical group 0.000 description 3
- 230000028993 immune response Effects 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 3
- 238000007799 mixed lymphocyte reaction assay Methods 0.000 description 3
- 201000006417 multiple sclerosis Diseases 0.000 description 3
- MZRVEZGGRBJDDB-UHFFFAOYSA-N n-Butyllithium Substances [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 3
- 230000036407 pain Effects 0.000 description 3
- 235000021317 phosphate Nutrition 0.000 description 3
- 150000003904 phospholipids Chemical class 0.000 description 3
- 229920001223 polyethylene glycol Polymers 0.000 description 3
- 230000035755 proliferation Effects 0.000 description 3
- 150000003335 secondary amines Chemical class 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 150000003431 steroids Chemical class 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 238000011287 therapeutic dose Methods 0.000 description 3
- 150000003571 thiolactams Chemical group 0.000 description 3
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 3
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 3
- 238000013389 whole blood assay Methods 0.000 description 3
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 2
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 2
- 125000006717 (C3-C10) cycloalkenyl group Chemical group 0.000 description 2
- RWVALRMOWBIJHB-SFHVURJKSA-N 1-[(3s)-3-[[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]amino]pyrrolidin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C([C@@H](C1)NC=2N=C(N=C3SC(CCC=4C=CC(F)=CC=4)=NC3=2)N)CN1C(=O)COC1=CC=C(Cl)C=C1 RWVALRMOWBIJHB-SFHVURJKSA-N 0.000 description 2
- GONNHMAPYIHDPT-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-1,4-diazepan-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCCN1C(=O)COC1=CC=C(Cl)C=C1 GONNHMAPYIHDPT-UHFFFAOYSA-N 0.000 description 2
- JNQVMCZJSFSOMS-UHFFFAOYSA-N 1-[4-(5-amino-2-cyclohexyl-[1,3]oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3CCCCC3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 JNQVMCZJSFSOMS-UHFFFAOYSA-N 0.000 description 2
- ONRXBNJZNNNJKH-UHFFFAOYSA-N 1-[4-(5-amino-2-cyclohexyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3CCCCC3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ONRXBNJZNNNJKH-UHFFFAOYSA-N 0.000 description 2
- DOEBSTUCINVUTO-UHFFFAOYSA-N 1-[4-(5-amino-2-cyclopropyl-[1,3]oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3CC3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 DOEBSTUCINVUTO-UHFFFAOYSA-N 0.000 description 2
- MDJGNAZNHPVYES-UHFFFAOYSA-N 1-[4-(5-amino-2-cyclopropyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3CC3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 MDJGNAZNHPVYES-UHFFFAOYSA-N 0.000 description 2
- AGYGNJCHCJDIFU-UHFFFAOYSA-N 1-[4-(5-amino-2-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2SC(SC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 AGYGNJCHCJDIFU-UHFFFAOYSA-N 0.000 description 2
- OHEFJYWOYMPUHR-UHFFFAOYSA-N 1-[4-(5-amino-2-pentyl-[1,3]oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2OC(CCCCC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 OHEFJYWOYMPUHR-UHFFFAOYSA-N 0.000 description 2
- PBAJDSDQGTWEPG-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 PBAJDSDQGTWEPG-UHFFFAOYSA-N 0.000 description 2
- IFCLDWSGEICRTE-UHFFFAOYSA-N 1-[4-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 IFCLDWSGEICRTE-UHFFFAOYSA-N 0.000 description 2
- RWHWHFCCKXUYNL-UHFFFAOYSA-N 1-[4-[2-butyl-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(CCCC)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 RWHWHFCCKXUYNL-UHFFFAOYSA-N 0.000 description 2
- YXOFGHDHCNKHOQ-UHFFFAOYSA-N 1-[4-[5-amino-2-(1-phenylcyclopropyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3(CC3)C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 YXOFGHDHCNKHOQ-UHFFFAOYSA-N 0.000 description 2
- AQFAQDWQTGOIMH-UHFFFAOYSA-N 1-[4-[5-amino-2-(2-phenylethyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 AQFAQDWQTGOIMH-UHFFFAOYSA-N 0.000 description 2
- BGTAIFYUYWWVFH-UHFFFAOYSA-N 1-[4-[5-amino-2-(2-phenylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 BGTAIFYUYWWVFH-UHFFFAOYSA-N 0.000 description 2
- ZIBGHZGPFOOSMD-UHFFFAOYSA-N 1-[4-[5-amino-2-(2-thiophen-2-ylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3SC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZIBGHZGPFOOSMD-UHFFFAOYSA-N 0.000 description 2
- IGRDTEUVLUVFRF-UHFFFAOYSA-N 1-[4-[5-amino-2-(2-thiophen-2-ylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenyl)ethanone Chemical compound C=12N=C(CCC=3SC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CC1=CC=C(Cl)C=C1 IGRDTEUVLUVFRF-UHFFFAOYSA-N 0.000 description 2
- ZIBXOJHPXLDWBG-UHFFFAOYSA-N 1-[4-[5-amino-2-(3,4-dichlorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=C(Cl)C(Cl)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZIBXOJHPXLDWBG-UHFFFAOYSA-N 0.000 description 2
- XDOSZWBFJKIRSQ-UHFFFAOYSA-N 1-[4-[5-amino-2-(3-methoxyphenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound COC1=CC=CC(C=2SC3=NC(N)=NC(=C3N=2)N2CCN(CC2)C(=O)COC=2C=CC(Cl)=CC=2)=C1 XDOSZWBFJKIRSQ-UHFFFAOYSA-N 0.000 description 2
- JGRCFYSJXXAVJW-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-chlorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(Cl)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 JGRCFYSJXXAVJW-UHFFFAOYSA-N 0.000 description 2
- FHZVQMXIOZIBQO-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-chlorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(Cl)=CC=2)CC1 FHZVQMXIOZIBQO-UHFFFAOYSA-N 0.000 description 2
- CESDYLQQGLEKAW-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluoroanilino)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(NC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 CESDYLQQGLEKAW-UHFFFAOYSA-N 0.000 description 2
- MQRPBANEXOSDGN-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-nitrophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC([N+]([O-])=O)=C1 MQRPBANEXOSDGN-UHFFFAOYSA-N 0.000 description 2
- XMDQAICFEBPNJK-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 XMDQAICFEBPNJK-UHFFFAOYSA-N 0.000 description 2
- LKGWCVVCRQXACR-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1CN(C(=O)C)CCN1C1=NC(N)=NC2=C1N=C(C=1C=CC(F)=CC=1)O2 LKGWCVVCRQXACR-UHFFFAOYSA-N 0.000 description 2
- BXKFJPOPOHKAFN-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-nitrophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC([N+]([O-])=O)=C1 BXKFJPOPOHKAFN-UHFFFAOYSA-N 0.000 description 2
- GGNKZXNKCNEDBD-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-phenoxyethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC=C1 GGNKZXNKCNEDBD-UHFFFAOYSA-N 0.000 description 2
- CRCCYIGTBSZZRH-UHFFFAOYSA-N 1-[4-[5-amino-2-(methoxymethyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2OC(COC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 CRCCYIGTBSZZRH-UHFFFAOYSA-N 0.000 description 2
- CUULOWVQNUHTQE-UHFFFAOYSA-N 1-[4-[5-amino-2-[(4-chlorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CC=3C=CC(Cl)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 CUULOWVQNUHTQE-UHFFFAOYSA-N 0.000 description 2
- ROQZGBPPTPAGNF-UHFFFAOYSA-N 1-[4-[5-amino-2-[1-(4-chlorophenyl)cyclopropyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C3(CC3)C=3C=CC(Cl)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ROQZGBPPTPAGNF-UHFFFAOYSA-N 0.000 description 2
- AJCILFMEZBIBSQ-UHFFFAOYSA-N 1-[4-[5-amino-2-[1-(4-chlorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=1C=C(Cl)C=CC=1C(C)C(SC1=NC(N)=N2)=NC1=C2N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 AJCILFMEZBIBSQ-UHFFFAOYSA-N 0.000 description 2
- YJWDQXWACDUIPM-UHFFFAOYSA-N 1-[4-[5-amino-2-[1-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=1C=C(F)C=CC=1C(C)C(SC1=NC(N)=N2)=NC1=C2N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 YJWDQXWACDUIPM-UHFFFAOYSA-N 0.000 description 2
- SCMNQHAJDPTKGO-UHFFFAOYSA-N 1-[4-[5-amino-2-[1-(4-fluorophenyl)propyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=1C=C(F)C=CC=1C(CC)C(SC1=NC(N)=N2)=NC1=C2N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 SCMNQHAJDPTKGO-UHFFFAOYSA-N 0.000 description 2
- WKNLBNROOFYBDI-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 WKNLBNROOFYBDI-UHFFFAOYSA-N 0.000 description 2
- PAOZUHUVVUJRDT-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenyl)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)CC1=CC=C(Cl)C=C1 PAOZUHUVVUJRDT-UHFFFAOYSA-N 0.000 description 2
- LAPVDJYQYXEPJA-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-1,4-diazepan-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCCN1C(=O)COC1=CC=C(Cl)C=C1 LAPVDJYQYXEPJA-UHFFFAOYSA-N 0.000 description 2
- TWTYIRXSDNFQPI-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(2,4-dichlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1Cl TWTYIRXSDNFQPI-UHFFFAOYSA-N 0.000 description 2
- LNVYSVHERQQLJV-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-methylphenoxy)ethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCN(CC2)C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)=C1 LNVYSVHERQQLJV-UHFFFAOYSA-N 0.000 description 2
- BZASDOUCFUIFGM-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-nitrophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC([N+]([O-])=O)=C1 BZASDOUCFUIFGM-UHFFFAOYSA-N 0.000 description 2
- AGOUPLUOZKXDIW-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Br)C=C1 AGOUPLUOZKXDIW-UHFFFAOYSA-N 0.000 description 2
- KQADOWXCIFMNDP-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenyl)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CC1=CC=C(Cl)C=C1 KQADOWXCIFMNDP-UHFFFAOYSA-N 0.000 description 2
- IHVXEJVBRMYPGX-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-fluorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(F)C=C1 IHVXEJVBRMYPGX-UHFFFAOYSA-N 0.000 description 2
- HGKRGWQHCHIQFA-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-phenoxyethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC=C1 HGKRGWQHCHIQFA-UHFFFAOYSA-N 0.000 description 2
- GLXJSZVRBSONQF-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-3-(4-fluorophenoxy)propan-1-one Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCOC1=CC=C(F)C=C1 GLXJSZVRBSONQF-UHFFFAOYSA-N 0.000 description 2
- IYYQYHPOEBKOQE-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-3-phenylpropan-1-one Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCC1=CC=CC=C1 IYYQYHPOEBKOQE-UHFFFAOYSA-N 0.000 description 2
- UGAWSUBVALMCTC-UHFFFAOYSA-N 1-[4-[5-amino-2-[cyclopentyl-(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C(C3CCCC3)C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 UGAWSUBVALMCTC-UHFFFAOYSA-N 0.000 description 2
- LISKJKUMLVQGKE-UHFFFAOYSA-N 1-morpholin-4-yl-2-piperazin-1-ylethanone Chemical compound C1COCCN1C(=O)CN1CCNCC1 LISKJKUMLVQGKE-UHFFFAOYSA-N 0.000 description 2
- 125000004214 1-pyrrolidinyl group Chemical group [H]C1([H])N(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- ZJFSQVPWMJRWFD-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-(4-fluorophenyl)-5-methyl-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)OC2=NC(C)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZJFSQVPWMJRWFD-UHFFFAOYSA-N 0.000 description 2
- ISJYYLKISXNLLZ-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-(4-fluorophenyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1C1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=CN=C2O1 ISJYYLKISXNLLZ-UHFFFAOYSA-N 0.000 description 2
- QWEIUROUCNABOD-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-[(4-fluorophenyl)methyl]-5-methyl-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)OC2=NC(C)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 QWEIUROUCNABOD-UHFFFAOYSA-N 0.000 description 2
- DNNJZQXVILQBDM-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-[(4-fluorophenyl)methyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1CC1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=CN=C2O1 DNNJZQXVILQBDM-UHFFFAOYSA-N 0.000 description 2
- FRSCHISFKLZDNE-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]ethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 FRSCHISFKLZDNE-UHFFFAOYSA-N 0.000 description 2
- ZIZHOHVSCJKUQF-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1C1=CC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=C(C=3C=CC=CC=3)N=C2S1 ZIZHOHVSCJKUQF-UHFFFAOYSA-N 0.000 description 2
- RDIHTEMYAICGMD-UHFFFAOYSA-N 2-(4-fluorophenyl)-5-methyl-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound N=1C=2C(SC)=NC(C)=NC=2SC=1C1=CC=C(F)C=C1 RDIHTEMYAICGMD-UHFFFAOYSA-N 0.000 description 2
- NHKKDPHXAJCPNZ-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-[4-(2-methoxyethyl)piperazin-1-yl]-[1,3]oxazolo[5,4-d]pyrimidin-5-amine Chemical compound C1CN(CCOC)CCN1C1=NC(N)=NC2=C1N=C(C=1C=CC(F)=CC=1)O2 NHKKDPHXAJCPNZ-UHFFFAOYSA-N 0.000 description 2
- NOJHUDGKTBYFRU-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-[4-(2-methylpropyl)piperazin-1-yl]-[1,3]oxazolo[5,4-d]pyrimidin-5-amine Chemical compound C1CN(CC(C)C)CCN1C1=NC(N)=NC2=C1N=C(C=1C=CC(F)=CC=1)O2 NOJHUDGKTBYFRU-UHFFFAOYSA-N 0.000 description 2
- FNYNAXQIHPQOSG-UHFFFAOYSA-N 2-[(4-fluorophenyl)methyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1CC1=CC=C(F)C=C1 FNYNAXQIHPQOSG-UHFFFAOYSA-N 0.000 description 2
- MKVSYRRPGYYUJD-UHFFFAOYSA-N 2-[2-(4-fluorophenoxy)ethyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1CCOC1=CC=C(F)C=C1 MKVSYRRPGYYUJD-UHFFFAOYSA-N 0.000 description 2
- HUSPMFNPRHBKFE-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-[4-(1,3-thiazol-2-yl)piperazin-1-yl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C1=NC=CS1 HUSPMFNPRHBKFE-UHFFFAOYSA-N 0.000 description 2
- QATFSUVESUNVFL-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-[4-(2-phenoxyethyl)piperazin-1-yl]-[1,3]oxazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1CCOC1=CC=CC=C1 QATFSUVESUNVFL-UHFFFAOYSA-N 0.000 description 2
- ORLAOQKPDJUERV-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-[4-(2-phenoxyethyl)piperazin-1-yl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1CCOC1=CC=CC=C1 ORLAOQKPDJUERV-UHFFFAOYSA-N 0.000 description 2
- XXOMNMWCPWSPLS-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1CCC1=CC=C(F)C=C1 XXOMNMWCPWSPLS-UHFFFAOYSA-N 0.000 description 2
- ZFNILYIEESEHPH-UHFFFAOYSA-N 2-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-n-methyl-n-phenylacetamide Chemical compound C=1C=CC=CC=1N(C)C(=O)CN(CC1)CCN1C(C=1C=2)=NC(N)=NC=1SC=2C1=CC=CC=C1 ZFNILYIEESEHPH-UHFFFAOYSA-N 0.000 description 2
- DJNIIAQCDNXCCX-UHFFFAOYSA-N 2-[4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl]acetamide Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(CC(=O)N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 DJNIIAQCDNXCCX-UHFFFAOYSA-N 0.000 description 2
- YRJZOTQVRYZBLK-UHFFFAOYSA-N 2-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-n-methyl-n-phenylacetamide Chemical compound C=1C=CC=CC=1N(C)C(=O)CN(CC1)CCN1C(C=1N=2)=NC(N)=NC=1OC=2CCC1=CC=C(F)C=C1 YRJZOTQVRYZBLK-UHFFFAOYSA-N 0.000 description 2
- PVMRKUCFNMEUQK-UHFFFAOYSA-N 2-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-n-(2-phenylethyl)acetamide Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1CC(=O)NCCC1=CC=CC=C1 PVMRKUCFNMEUQK-UHFFFAOYSA-N 0.000 description 2
- JIUBKRFMEIWBJQ-UHFFFAOYSA-N 2-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-n-methyl-n-phenylacetamide Chemical compound C=1C=CC=CC=1N(C)C(=O)CN(CC1)CCN1C(C=1N=2)=NC(N)=NC=1SC=2CCC1=CC=C(F)C=C1 JIUBKRFMEIWBJQ-UHFFFAOYSA-N 0.000 description 2
- YPCGDGWLGZUUST-UHFFFAOYSA-N 2-butyl-n-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(CCCC)=NC=1NC1=CC=C(F)C(Cl)=C1 YPCGDGWLGZUUST-UHFFFAOYSA-N 0.000 description 2
- IPFLHLWPVQVKAL-UHFFFAOYSA-N 2-cyclopropyl-7-methoxy-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(OC)=NC(N)=NC=2SC=1C1CC1 IPFLHLWPVQVKAL-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- DUFGYCAXVIUXIP-UHFFFAOYSA-N 4,6-dihydroxypyrimidine Chemical class OC1=CC(O)=NC=N1 DUFGYCAXVIUXIP-UHFFFAOYSA-N 0.000 description 2
- UMSFRPADYZSSFV-UHFFFAOYSA-N 4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-n-(4-chlorophenyl)piperazine-1-carboxamide Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(Cl)C=C1 UMSFRPADYZSSFV-UHFFFAOYSA-N 0.000 description 2
- SNVAVVDOPMDHJA-UHFFFAOYSA-N 4-(4-benzylsulfonylpiperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1S(=O)(=O)CC1=CC=CC=C1 SNVAVVDOPMDHJA-UHFFFAOYSA-N 0.000 description 2
- IUNBALWMGYXAJA-UHFFFAOYSA-N 4-(6-phenylthieno[2,3-d]pyrimidin-2-yl)-1,4-diazepan-2-amine Chemical compound C1CCNC(N)CN1C1=NC=C(C=C(S2)C=3C=CC=CC=3)C2=N1 IUNBALWMGYXAJA-UHFFFAOYSA-N 0.000 description 2
- VXTYGWVEFJNJFT-UHFFFAOYSA-N 4-(6-phenylthieno[2,3-d]pyrimidin-2-yl)piperazin-2-amine Chemical compound C1CNC(N)CN1C1=NC=C(C=C(S2)C=3C=CC=CC=3)C2=N1 VXTYGWVEFJNJFT-UHFFFAOYSA-N 0.000 description 2
- YVSXYJNOKKPQGH-UHFFFAOYSA-N 4-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3C=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 YVSXYJNOKKPQGH-UHFFFAOYSA-N 0.000 description 2
- HOFXMOCSYGWGPY-UHFFFAOYSA-N 4-[4-(2-phenoxyethyl)piperazin-1-yl]-6-phenylthieno[2,3-d]pyrimidin-2-amine Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1CCOC1=CC=CC=C1 HOFXMOCSYGWGPY-UHFFFAOYSA-N 0.000 description 2
- HBPMNASYNVTFOC-UHFFFAOYSA-N 4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)-n-(2-methoxyethyl)thieno[2,3-d]pyrimidine-2-carboxamide Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)NCCOC)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 HBPMNASYNVTFOC-UHFFFAOYSA-N 0.000 description 2
- PZWOOCJOKIKIDT-UHFFFAOYSA-N 4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxamide Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 PZWOOCJOKIKIDT-UHFFFAOYSA-N 0.000 description 2
- JKUUKGFWIJBOIZ-UHFFFAOYSA-N 4-[5-amino-2-(2-thiophen-2-ylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(CCC=4SC=CC=4)SC=3N=C(N)N=2)=C1 JKUUKGFWIJBOIZ-UHFFFAOYSA-N 0.000 description 2
- FZYKUEGJXZLSQA-UHFFFAOYSA-N 4-[5-amino-2-[1-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound C=1C=C(F)C=CC=1C(C)C(SC1=NC(N)=N2)=NC1=C2N(CC1)CCN1C(=O)NC1=CC=CC(C)=C1 FZYKUEGJXZLSQA-UHFFFAOYSA-N 0.000 description 2
- PWRBPXKVDUZDKI-UHFFFAOYSA-N 4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(CCC=4C=CC(F)=CC=4)OC=3N=C(N)N=2)=C1 PWRBPXKVDUZDKI-UHFFFAOYSA-N 0.000 description 2
- LYMJTSXMIUWZSW-UHFFFAOYSA-N 4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)=C1 LYMJTSXMIUWZSW-UHFFFAOYSA-N 0.000 description 2
- LTYNLPNHEYQSGJ-UHFFFAOYSA-N 4-[6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl]-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3C=C(SC=3N=C(N=2)C=2C=CC=CC=2)C=2C=CC(F)=CC=2)=C1 LTYNLPNHEYQSGJ-UHFFFAOYSA-N 0.000 description 2
- HZBZCSWVUXVFSC-UHFFFAOYSA-N 4-[6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl]piperazin-2-amine Chemical compound C1CNC(N)CN1C1=NC=C(C=C(S2)C=3C=CC(F)=CC=3)C2=N1 HZBZCSWVUXVFSC-UHFFFAOYSA-N 0.000 description 2
- WUBBRNOQWQTFEX-UHFFFAOYSA-M 4-aminosalicylate(1-) Chemical compound NC1=CC=C(C([O-])=O)C(O)=C1 WUBBRNOQWQTFEX-UHFFFAOYSA-M 0.000 description 2
- QRJBCQJWZPXKOS-UHFFFAOYSA-N 4-ethoxy-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine Chemical compound C=1C=2C(OCC)=NC(N)=NC=2SC=1C1=CC=C(F)C=C1 QRJBCQJWZPXKOS-UHFFFAOYSA-N 0.000 description 2
- PILFPZMUDJMYMY-UHFFFAOYSA-N 4-thieno[2,3-d]pyrimidin-2-ylpiperazin-2-amine Chemical compound C1CNC(N)CN1C1=NC=C(C=CS2)C2=N1 PILFPZMUDJMYMY-UHFFFAOYSA-N 0.000 description 2
- MTBGYVHTKSFEPG-UHFFFAOYSA-N 6-(4-fluorophenyl)-4-[4-[(3-methylphenyl)carbamoyl]piperazin-1-yl]thieno[2,3-d]pyrimidine-2-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3C=C(SC=3N=C(N=2)C(N)=O)C=2C=CC(F)=CC=2)=C1 MTBGYVHTKSFEPG-UHFFFAOYSA-N 0.000 description 2
- WGXOXPSFNAVBMN-UHFFFAOYSA-N 6-(4-fluorophenyl)-4-morpholin-4-ylthieno[2,3-d]pyrimidin-2-amine Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCOCC1 WGXOXPSFNAVBMN-UHFFFAOYSA-N 0.000 description 2
- WEWKDLATYYFPLJ-UHFFFAOYSA-N 7-(4-benzylsulfonylpiperazin-1-yl)-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1S(=O)(=O)CC1=CC=CC=C1 WEWKDLATYYFPLJ-UHFFFAOYSA-N 0.000 description 2
- HESVXKZDSUGWQM-UHFFFAOYSA-N 7-benzylsulfanyl-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1SCC1=CC=CC=C1 HESVXKZDSUGWQM-UHFFFAOYSA-N 0.000 description 2
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 2
- 208000008439 Biliary Liver Cirrhosis Diseases 0.000 description 2
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 2
- 102000004631 Calcineurin Human genes 0.000 description 2
- 108010042955 Calcineurin Proteins 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- 241000282472 Canis lupus familiaris Species 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 208000011231 Crohn disease Diseases 0.000 description 2
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- HKVAMNSJSFKALM-GKUWKFKPSA-N Everolimus Chemical compound C1C[C@@H](OCCO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 HKVAMNSJSFKALM-GKUWKFKPSA-N 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 description 2
- 206010019842 Hepatomegaly Diseases 0.000 description 2
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 2
- 206010020772 Hypertension Diseases 0.000 description 2
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 2
- 102000008070 Interferon-gamma Human genes 0.000 description 2
- 108010002352 Interleukin-1 Proteins 0.000 description 2
- 102000000589 Interleukin-1 Human genes 0.000 description 2
- 108090000172 Interleukin-15 Proteins 0.000 description 2
- 108090001005 Interleukin-6 Proteins 0.000 description 2
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 2
- 101150058160 Lyn gene Proteins 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- 108091000080 Phosphotransferase Proteins 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 2
- ZTHYODDOHIVTJV-UHFFFAOYSA-N Propyl gallate Chemical compound CCCOC(=O)C1=CC(O)=C(O)C(O)=C1 ZTHYODDOHIVTJV-UHFFFAOYSA-N 0.000 description 2
- LCTONWCANYUPML-UHFFFAOYSA-M Pyruvate Chemical compound CC(=O)C([O-])=O LCTONWCANYUPML-UHFFFAOYSA-M 0.000 description 2
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 2
- 102000001332 SRC Human genes 0.000 description 2
- 108060006706 SRC Proteins 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 229920002125 Sokalan® Polymers 0.000 description 2
- 239000012317 TBTU Substances 0.000 description 2
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 2
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 102000008236 Toll-Like Receptor 7 Human genes 0.000 description 2
- 108010060825 Toll-Like Receptor 7 Proteins 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 2
- 206010046851 Uveitis Diseases 0.000 description 2
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 2
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 2
- WCBKNACDYVCBLV-UHFFFAOYSA-N [1-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperidin-4-yl]-phenylmethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCC1C(=O)C1=CC=CC=C1 WCBKNACDYVCBLV-UHFFFAOYSA-N 0.000 description 2
- IOTQXHBOLWBDNO-UHFFFAOYSA-N [1-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperidin-4-yl]-phenylmethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCC1C(=O)C1=CC=CC=C1 IOTQXHBOLWBDNO-UHFFFAOYSA-N 0.000 description 2
- CMLPPNNEVNLWJY-UHFFFAOYSA-N [4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-1,4-diazepan-1-yl]-(4-chlorophenyl)methanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCCN1C(=O)C1=CC=C(Cl)C=C1 CMLPPNNEVNLWJY-UHFFFAOYSA-N 0.000 description 2
- XYOPNYPNRJVRKR-UHFFFAOYSA-N [4-[5-amino-2-(2-thiophen-2-ylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-(4-chlorophenyl)methanone Chemical compound C=12N=C(CCC=3SC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)C1=CC=C(Cl)C=C1 XYOPNYPNRJVRKR-UHFFFAOYSA-N 0.000 description 2
- RTFCJMYSXJQTOP-UHFFFAOYSA-N [4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]oxazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-(4-chlorophenyl)methanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)OC2=NC(N)=NC=1N(CC1)CCN1C(=O)C1=CC=C(Cl)C=C1 RTFCJMYSXJQTOP-UHFFFAOYSA-N 0.000 description 2
- KCVVYWILFYFBKX-UHFFFAOYSA-N [4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-(4-chlorophenyl)methanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)C1=CC=C(Cl)C=C1 KCVVYWILFYFBKX-UHFFFAOYSA-N 0.000 description 2
- 239000012190 activator Substances 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 150000001298 alcohols Chemical class 0.000 description 2
- 230000029936 alkylation Effects 0.000 description 2
- 230000036592 analgesia Effects 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 239000003945 anionic surfactant Substances 0.000 description 2
- 206010003246 arthritis Diseases 0.000 description 2
- 150000001500 aryl chlorides Chemical class 0.000 description 2
- 208000006673 asthma Diseases 0.000 description 2
- 239000005441 aurora Substances 0.000 description 2
- 230000005784 autoimmunity Effects 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 229940050390 benzoate Drugs 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 2
- XPLLVMOMMKUTJI-UHFFFAOYSA-N benzyl 3-[[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]amino]piperidine-1-carboxylate Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1NC(C1)CCCN1C(=O)OCC1=CC=CC=C1 XPLLVMOMMKUTJI-UHFFFAOYSA-N 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 210000001185 bone marrow Anatomy 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 159000000007 calcium salts Chemical class 0.000 description 2
- 150000004657 carbamic acid derivatives Chemical class 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 238000001460 carbon-13 nuclear magnetic resonance spectrum Methods 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 239000004359 castor oil Substances 0.000 description 2
- 235000019438 castor oil Nutrition 0.000 description 2
- 239000003093 cationic surfactant Substances 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 239000012320 chlorinating reagent Substances 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- AOGYCOYQMAVAFD-UHFFFAOYSA-N chlorocarbonic acid Chemical class OC(Cl)=O AOGYCOYQMAVAFD-UHFFFAOYSA-N 0.000 description 2
- 239000008139 complexing agent Substances 0.000 description 2
- 239000007819 coupling partner Substances 0.000 description 2
- 230000006378 damage Effects 0.000 description 2
- 229960003957 dexamethasone Drugs 0.000 description 2
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 2
- 230000004069 differentiation Effects 0.000 description 2
- BGRWYRAHAFMIBJ-UHFFFAOYSA-N diisopropylcarbodiimide Natural products CC(C)NC(=O)NC(C)C BGRWYRAHAFMIBJ-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 238000001647 drug administration Methods 0.000 description 2
- 238000000132 electrospray ionisation Methods 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000001804 emulsifying effect Effects 0.000 description 2
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 2
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 2
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 2
- NJMDQJMSRKJURW-UHFFFAOYSA-N ethyl 2-[4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl]acetate Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(CC(=O)OCC)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 NJMDQJMSRKJURW-UHFFFAOYSA-N 0.000 description 2
- MKJQYFVTEPGXIE-UHFFFAOYSA-N ethyl 2-aminothiophene-3-carboxylate Chemical compound CCOC(=O)C=1C=CSC=1N MKJQYFVTEPGXIE-UHFFFAOYSA-N 0.000 description 2
- LFKCSLOWTJCEHR-UHFFFAOYSA-N ethyl 6-(4-fluorophenyl)-4-[4-[(3-methylphenyl)carbamoyl]piperazin-1-yl]thieno[2,3-d]pyrimidine-2-carboxylate Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)OCC)=NC=1N(CC1)CCN1C(=O)NC1=CC=CC(C)=C1 LFKCSLOWTJCEHR-UHFFFAOYSA-N 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 150000002191 fatty alcohols Chemical class 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 125000004005 formimidoyl group Chemical group [H]\N=C(/[H])* 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical compound [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 2
- FFUAGWLWBBFQJT-UHFFFAOYSA-N hexamethyldisilazane Chemical compound C[Si](C)(C)N[Si](C)(C)C FFUAGWLWBBFQJT-UHFFFAOYSA-N 0.000 description 2
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 2
- 125000002883 imidazolyl group Chemical group 0.000 description 2
- 230000005847 immunogenicity Effects 0.000 description 2
- 229940124589 immunosuppressive drug Drugs 0.000 description 2
- PCKPVGOLPKLUHR-UHFFFAOYSA-N indoxyl Chemical group C1=CC=C2C(O)=CNC2=C1 PCKPVGOLPKLUHR-UHFFFAOYSA-N 0.000 description 2
- 230000002401 inhibitory effect Effects 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 229960003130 interferon gamma Drugs 0.000 description 2
- 239000007926 intracavernous injection Substances 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 239000012948 isocyanate Substances 0.000 description 2
- 150000002513 isocyanates Chemical class 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 208000032839 leukemia Diseases 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000000395 magnesium oxide Substances 0.000 description 2
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 2
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 2
- 229960000485 methotrexate Drugs 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 239000003094 microcapsule Substances 0.000 description 2
- 239000004005 microsphere Substances 0.000 description 2
- 150000002763 monocarboxylic acids Chemical class 0.000 description 2
- 206010028417 myasthenia gravis Diseases 0.000 description 2
- ZZRBFZOYSRTIAN-UHFFFAOYSA-N n-(2-amino-4-hydroxy-6-oxo-1h-pyrimidin-5-yl)-3-(4-fluorophenoxy)propanamide Chemical compound OC1=NC(N)=NC(O)=C1NC(=O)CCOC1=CC=C(F)C=C1 ZZRBFZOYSRTIAN-UHFFFAOYSA-N 0.000 description 2
- PBGBBWKKKNOZIF-UHFFFAOYSA-N n-(2-amino-4-hydroxy-6-oxo-1h-pyrimidin-5-yl)-4-fluorobenzamide Chemical compound OC1=NC(N)=NC(O)=C1NC(=O)C1=CC=C(F)C=C1 PBGBBWKKKNOZIF-UHFFFAOYSA-N 0.000 description 2
- JNRLWYFGYIOHLO-UHFFFAOYSA-N n-(3-chloro-4-fluorophenyl)-2-(4-fluorophenyl)-[1,3]oxazolo[5,4-d]pyrimidin-7-amine Chemical compound C1=CC(F)=CC=C1C(OC1=NC=N2)=NC1=C2NC1=CC=C(F)C(Cl)=C1 JNRLWYFGYIOHLO-UHFFFAOYSA-N 0.000 description 2
- MVHOOTKXNWJNGT-UHFFFAOYSA-N n-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-amine Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C)=NC=1NC1=CC=C(F)C(Cl)=C1 MVHOOTKXNWJNGT-UHFFFAOYSA-N 0.000 description 2
- XVGDSVSDVPYBSF-GOSISDBHSA-N n-[(3r)-1-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]pyrrolidin-3-yl]-2-(4-chlorophenoxy)acetamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(CCC=3C=CC(F)=CC=3)=NC2=1)N)C(=O)COC1=CC=C(Cl)C=C1 XVGDSVSDVPYBSF-GOSISDBHSA-N 0.000 description 2
- JFJXOYMTJHHYAU-GOSISDBHSA-N n-[(3r)-1-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]pyrrolidin-3-yl]-4-chlorobenzamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(CCC=3C=CC(F)=CC=3)=NC2=1)N)C(=O)C1=CC=C(Cl)C=C1 JFJXOYMTJHHYAU-GOSISDBHSA-N 0.000 description 2
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 150000002825 nitriles Chemical class 0.000 description 2
- 230000000269 nucleophilic effect Effects 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 2
- 125000004430 oxygen atom Chemical group O* 0.000 description 2
- 125000000913 palmityl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 210000000496 pancreas Anatomy 0.000 description 2
- 238000005897 peptide coupling reaction Methods 0.000 description 2
- 125000001484 phenothiazinyl group Chemical group C1(=CC=CC=2SC3=CC=CC=C3NC12)* 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 2
- 239000010452 phosphate Substances 0.000 description 2
- XYFCBTPGUUZFHI-UHFFFAOYSA-O phosphonium Chemical compound [PH4+] XYFCBTPGUUZFHI-UHFFFAOYSA-O 0.000 description 2
- 102000020233 phosphotransferase Human genes 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000002243 precursor Substances 0.000 description 2
- 229960005205 prednisolone Drugs 0.000 description 2
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 description 2
- 230000002062 proliferating effect Effects 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 125000006239 protecting group Chemical group 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 2
- UYLWKSJTHLRFBX-UHFFFAOYSA-N purin-6-one Chemical class O=C1N=CN=C2N=CN=C12 UYLWKSJTHLRFBX-UHFFFAOYSA-N 0.000 description 2
- 229940083082 pyrimidine derivative acting on arteriolar smooth muscle Drugs 0.000 description 2
- 150000003230 pyrimidines Chemical class 0.000 description 2
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 206010039073 rheumatoid arthritis Diseases 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-M salicylate Chemical compound OC1=CC=CC=C1C([O-])=O YGSDEFSMJLZEOE-UHFFFAOYSA-M 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000000344 soap Substances 0.000 description 2
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 2
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 2
- 239000012265 solid product Substances 0.000 description 2
- 235000010199 sorbic acid Nutrition 0.000 description 2
- 239000004334 sorbic acid Substances 0.000 description 2
- 229940075582 sorbic acid Drugs 0.000 description 2
- 230000002269 spontaneous effect Effects 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- 229940124530 sulfonamide Drugs 0.000 description 2
- 150000003456 sulfonamides Chemical class 0.000 description 2
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 2
- BDHFUVZGWQCTTF-UHFFFAOYSA-N sulfonic acid Chemical class OS(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-N 0.000 description 2
- 125000004434 sulfur atom Chemical group 0.000 description 2
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 2
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical class ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 2
- 239000011885 synergistic combination Substances 0.000 description 2
- 229960001967 tacrolimus Drugs 0.000 description 2
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 description 2
- WNJOQYYEHQVKSI-HNNXBMFYSA-N tert-butyl (3s)-3-[[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]amino]pyrrolidine-1-carboxylate Chemical compound C1N(C(=O)OC(C)(C)C)CC[C@@H]1NC1=NC(N)=NC2=C1N=C(CCC=1C=CC(F)=CC=1)S2 WNJOQYYEHQVKSI-HNNXBMFYSA-N 0.000 description 2
- ZFXYFBGIUFBOJW-UHFFFAOYSA-N theophylline Chemical compound O=C1N(C)C(=O)N(C)C2=C1NC=N2 ZFXYFBGIUFBOJW-UHFFFAOYSA-N 0.000 description 2
- 150000008634 thiazolopyrimidines Chemical class 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- 125000001425 triazolyl group Chemical group 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 208000035408 type 1 diabetes mellitus 1 Diseases 0.000 description 2
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 2
- RJNRORZRFGUAKL-ADMBVFOFSA-N (1r)-1-[(3ar,5r,6s,6ar)-6-[3-(dimethylamino)propoxy]-2,2-dimethyl-3a,5,6,6a-tetrahydrofuro[2,3-d][1,3]dioxol-5-yl]ethane-1,2-diol;hydrochloride Chemical compound Cl.O1C(C)(C)O[C@@H]2[C@@H](OCCCN(C)C)[C@@H]([C@H](O)CO)O[C@@H]21 RJNRORZRFGUAKL-ADMBVFOFSA-N 0.000 description 1
- FKHUGQZRBPETJR-RXSRXONKSA-N (2r)-2-[[(4r)-4-[[(2s)-2-[[(2r)-2-[(3r,4r,5s,6r)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxypropanoyl]amino]propanoyl]amino]-5-amino-5-oxopentanoyl]amino]-6-(octadecanoylamino)hexanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(=O)NCCCC[C@H](C(O)=O)NC(=O)CC[C@H](C(N)=O)NC(=O)[C@H](C)NC(=O)[C@@H](C)O[C@H]1[C@H](O)[C@@H](CO)OC(O)[C@@H]1NC(C)=O FKHUGQZRBPETJR-RXSRXONKSA-N 0.000 description 1
- VUAFHZCUKUDDBC-SCSAIBSYSA-N (2s)-2-[(2-methyl-2-sulfanylpropanoyl)amino]-3-sulfanylpropanoic acid Chemical compound CC(C)(S)C(=O)N[C@H](CS)C(O)=O VUAFHZCUKUDDBC-SCSAIBSYSA-N 0.000 description 1
- XOYXESIZZFUVRD-UVSAJTFZSA-N (2s,3s,4r,5s,6s)-6-[(2r,3r,4r,5s,6r)-6-[(2r,3s,4r,5s,6r)-5-acetamido-6-[(2r,3r,4r,5s,6r)-4-acetyloxy-6-[(2r,3r,4r,5s,6r)-4-acetyloxy-6-[(2r,3r,4r,5s,6s)-4-acetyloxy-5-hydroxy-2-(hydroxymethyl)-6-methoxyoxan-3-yl]oxy-5-hydroxy-2-(hydroxymethyl)oxan-3-yl]ox Chemical compound CC(=O)O[C@@H]1[C@H](O)[C@@H](OC)O[C@H](CO)[C@H]1O[C@@H]1[C@@H](O)[C@@H](OC(C)=O)[C@H](O[C@@H]2[C@H]([C@@H](OC(C)=O)[C@H](O[C@@H]3[C@H]([C@@H](O)[C@H](O[C@@H]4[C@H]([C@@H](OC(C)=O)[C@H](O[C@@H]5[C@H]([C@@H](OC(C)=O)[C@H](O[C@@H]6[C@H]([C@@H](OC(C)=O)[C@H](O[C@@H]7[C@H]([C@@H](OC(C)=O)[C@H](OC)[C@@H](CO)O7)O)[C@@H](CO)O6)O)[C@H](O5)C(O)=O)O)[C@@H](CO)O4)O)[C@@H](CO)O3)NC(C)=O)[C@@H](CO)O2)O)[C@@H](CO)O1 XOYXESIZZFUVRD-UVSAJTFZSA-N 0.000 description 1
- ASWBNKHCZGQVJV-UHFFFAOYSA-N (3-hexadecanoyloxy-2-hydroxypropyl) 2-(trimethylazaniumyl)ethyl phosphate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(O)COP([O-])(=O)OCC[N+](C)(C)C ASWBNKHCZGQVJV-UHFFFAOYSA-N 0.000 description 1
- JFLCLRXZVNJXLY-UHFFFAOYSA-N (4-methylphenyl) 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazine-1-carboxylate Chemical compound C1=CC(C)=CC=C1OC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CC1 JFLCLRXZVNJXLY-UHFFFAOYSA-N 0.000 description 1
- UUTKICFRNVKFRG-WDSKDSINSA-N (4R)-3-[oxo-[(2S)-5-oxo-2-pyrrolidinyl]methyl]-4-thiazolidinecarboxylic acid Chemical compound OC(=O)[C@@H]1CSCN1C(=O)[C@H]1NC(=O)CC1 UUTKICFRNVKFRG-WDSKDSINSA-N 0.000 description 1
- 125000004400 (C1-C12) alkyl group Chemical group 0.000 description 1
- 125000006586 (C3-C10) cycloalkylene group Chemical group 0.000 description 1
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- NSMXQKNUPPXBRG-SECBINFHSA-N (R)-lisofylline Chemical compound O=C1N(CCCC[C@H](O)C)C(=O)N(C)C2=C1N(C)C=N2 NSMXQKNUPPXBRG-SECBINFHSA-N 0.000 description 1
- WHTVZRBIWZFKQO-AWEZNQCLSA-N (S)-chloroquine Chemical compound ClC1=CC=C2C(N[C@@H](C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-AWEZNQCLSA-N 0.000 description 1
- TXCWKZUXQFCWDW-UHFFFAOYSA-N (hydroxyamino) cyanoformate Chemical class ONOC(=O)C#N TXCWKZUXQFCWDW-UHFFFAOYSA-N 0.000 description 1
- NDQXKKFRNOPRDW-UHFFFAOYSA-N 1,1,1-triethoxyethane Chemical compound CCOC(C)(OCC)OCC NDQXKKFRNOPRDW-UHFFFAOYSA-N 0.000 description 1
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 1
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 description 1
- SRGYIOWEOTVTRE-UHFFFAOYSA-N 1,3-oxazinane-2,6-dione Chemical compound O=C1CCNC(=O)O1 SRGYIOWEOTVTRE-UHFFFAOYSA-N 0.000 description 1
- JIWHIRLNKIUYSM-UHFFFAOYSA-N 1-(3-methylphenyl)piperazine Chemical compound CC1=CC=CC(N2CCNCC2)=C1 JIWHIRLNKIUYSM-UHFFFAOYSA-N 0.000 description 1
- NCXUNZWLEYGQAH-UHFFFAOYSA-N 1-(dimethylamino)propan-2-ol Chemical compound CC(O)CN(C)C NCXUNZWLEYGQAH-UHFFFAOYSA-N 0.000 description 1
- BUXJIFWIEIVJHU-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(3-methylphenoxy)ethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCN(CC2)C=2C=3C=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)=C1 BUXJIFWIEIVJHU-UHFFFAOYSA-N 0.000 description 1
- JPGLGKQUTDLUOC-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Br)C=C1 JPGLGKQUTDLUOC-UHFFFAOYSA-N 0.000 description 1
- XXJOHQDWDMUUGE-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-chlorophenoxy)-2-methylpropan-1-one Chemical compound C1CN(C=2C=3C=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)CCN1C(=O)C(C)(C)OC1=CC=C(Cl)C=C1 XXJOHQDWDMUUGE-UHFFFAOYSA-N 0.000 description 1
- IATXSWLYBQMJTH-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 IATXSWLYBQMJTH-UHFFFAOYSA-N 0.000 description 1
- RALMKQRKLNLTLS-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-fluorophenoxy)ethanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(F)C=C1 RALMKQRKLNLTLS-UHFFFAOYSA-N 0.000 description 1
- NCOVWHUVJSCTNR-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3C=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)CC1 NCOVWHUVJSCTNR-UHFFFAOYSA-N 0.000 description 1
- OGINVPGYZHANFG-UHFFFAOYSA-N 1-[4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-phenoxyethanone Chemical compound C=12C=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC=C1 OGINVPGYZHANFG-UHFFFAOYSA-N 0.000 description 1
- KDSZIYBABLTQOY-UHFFFAOYSA-N 1-[4-(2-aminothieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12C=CSC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 KDSZIYBABLTQOY-UHFFFAOYSA-N 0.000 description 1
- FFYIDMIDTRSYCM-UHFFFAOYSA-N 1-[4-(5-amino-2-pentyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound N1=C(N)N=C2SC(CCCCC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 FFYIDMIDTRSYCM-UHFFFAOYSA-N 0.000 description 1
- NIILYBAXFOQFJE-UHFFFAOYSA-N 1-[4-(5-amino-2-pentyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound N1=C(N)N=C2SC(CCCCC)=NC2=C1N(CC1)CCN1C(=O)COC1=CC=C(OC)C=C1 NIILYBAXFOQFJE-UHFFFAOYSA-N 0.000 description 1
- UGFOVUJJPRYQAK-UHFFFAOYSA-N 1-[4-(5-amino-2-phenyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 UGFOVUJJPRYQAK-UHFFFAOYSA-N 0.000 description 1
- KSWWWUGSAGJHDW-UHFFFAOYSA-N 1-[4-(5-amino-2-phenyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)CC1 KSWWWUGSAGJHDW-UHFFFAOYSA-N 0.000 description 1
- CNLMRCWINTWWLY-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-2-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3N=CC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 CNLMRCWINTWWLY-UHFFFAOYSA-N 0.000 description 1
- YDRDFSKLJIKUTM-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(3-methylphenoxy)ethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)=C1 YDRDFSKLJIKUTM-UHFFFAOYSA-N 0.000 description 1
- FEACADSWWIGHLO-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)-2-methylpropan-1-one Chemical compound C1CN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CCN1C(=O)C(C)(C)OC1=CC=C(Cl)C=C1 FEACADSWWIGHLO-UHFFFAOYSA-N 0.000 description 1
- LQDJVNXWAUMXGY-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-fluorophenoxy)ethanone Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(F)C=C1 LQDJVNXWAUMXGY-UHFFFAOYSA-N 0.000 description 1
- CXCCIUUTRKWCEH-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CC1 CXCCIUUTRKWCEH-UHFFFAOYSA-N 0.000 description 1
- YQEORQRIVXMANJ-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-[4-(trifluoromethoxy)phenoxy]ethanone Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(OC(F)(F)F)C=C1 YQEORQRIVXMANJ-UHFFFAOYSA-N 0.000 description 1
- MHNBQNCHUDWNAY-UHFFFAOYSA-N 1-[4-(5-amino-2-pyridin-4-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CN=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 MHNBQNCHUDWNAY-UHFFFAOYSA-N 0.000 description 1
- BOIQTHTYOPRRMI-UHFFFAOYSA-N 1-[4-[2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=CN=2)C=2C=CC(F)=CC=2)CC1 BOIQTHTYOPRRMI-UHFFFAOYSA-N 0.000 description 1
- AVFMULBAPJSXEE-UHFFFAOYSA-N 1-[4-[2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=CN=2)CC1 AVFMULBAPJSXEE-UHFFFAOYSA-N 0.000 description 1
- IFXBZCOVKOVHIM-UHFFFAOYSA-N 1-[4-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-3-phenylpropan-1-one Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCC1=CC=CC=C1 IFXBZCOVKOVHIM-UHFFFAOYSA-N 0.000 description 1
- KRYFVVOWHBKCIW-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(2,4-dichlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1Cl KRYFVVOWHBKCIW-UHFFFAOYSA-N 0.000 description 1
- YQVHACJRDOVTDZ-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-chlorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC(Cl)=C1 YQVHACJRDOVTDZ-UHFFFAOYSA-N 0.000 description 1
- HSDPBPVPFVKIKS-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-methylphenoxy)ethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)=C1 HSDPBPVPFVKIKS-UHFFFAOYSA-N 0.000 description 1
- HZNMPBDKNXTQDG-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chloro-2-methylphenoxy)ethanone Chemical compound CC1=CC(Cl)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)CC1 HZNMPBDKNXTQDG-UHFFFAOYSA-N 0.000 description 1
- ZFJMLJXZMDIEJI-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)-2-methylpropan-1-one Chemical compound C1CN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)CCN1C(=O)C(C)(C)OC1=CC=C(Cl)C=C1 ZFJMLJXZMDIEJI-UHFFFAOYSA-N 0.000 description 1
- IVSKUPHKMKWFGI-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-fluorophenoxy)ethanone Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(F)C=C1 IVSKUPHKMKWFGI-UHFFFAOYSA-N 0.000 description 1
- WLDHICAHBAKWEB-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)CC1 WLDHICAHBAKWEB-UHFFFAOYSA-N 0.000 description 1
- UUEJVCMVMAWPRX-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-3-(4-fluorophenyl)propan-1-one Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCC1=CC=C(F)C=C1 UUEJVCMVMAWPRX-UHFFFAOYSA-N 0.000 description 1
- LHGQBVDWIADCEH-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-methylphenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C1=CC(C)=CC=C1C1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=C(N)N=C2S1 LHGQBVDWIADCEH-UHFFFAOYSA-N 0.000 description 1
- GKGKGSPCILMFDY-UHFFFAOYSA-N 1-[4-[5-amino-2-(4-methylphenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(C)=CC=2)CC1 GKGKGSPCILMFDY-UHFFFAOYSA-N 0.000 description 1
- VFPNLHBPLKGLIW-UHFFFAOYSA-N 1-[4-[5-amino-2-(furan-2-yl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-methylphenoxy)ethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2OC=CC=2)=C1 VFPNLHBPLKGLIW-UHFFFAOYSA-N 0.000 description 1
- CSYZHVMTSLUGND-UHFFFAOYSA-N 1-[4-[5-amino-2-(furan-2-yl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-fluorophenoxy)ethanone Chemical compound C=12N=C(C=3OC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(F)C=C1 CSYZHVMTSLUGND-UHFFFAOYSA-N 0.000 description 1
- OKFXMLCTWHZGIU-UHFFFAOYSA-N 1-[4-[5-amino-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Br)C=C1 OKFXMLCTWHZGIU-UHFFFAOYSA-N 0.000 description 1
- BDGATOOPYPZCLY-UHFFFAOYSA-N 1-[4-[5-amino-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 BDGATOOPYPZCLY-UHFFFAOYSA-N 0.000 description 1
- KPWUVBHJGDYOGE-UHFFFAOYSA-N 1-[4-[5-amino-2-[1-(4-fluorophenyl)-2-phenylethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(C(CC=3C=CC=CC=3)C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 KPWUVBHJGDYOGE-UHFFFAOYSA-N 0.000 description 1
- LPLQRAXZUOONCH-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(3,4-dimethoxyphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C1=C(OC)C(OC)=CC=C1CCC1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Br)=CC=3)N=C(N)N=C2S1 LPLQRAXZUOONCH-UHFFFAOYSA-N 0.000 description 1
- XNKXOXZSNZKEOM-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(3,4-dimethoxyphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=C(OC)C(OC)=CC=4)SC=3N=C(N)N=2)CC1 XNKXOXZSNZKEOM-UHFFFAOYSA-N 0.000 description 1
- FPVUEEFJDLFBQK-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(3-methoxyphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound COC1=CC=CC(CCC=2SC3=NC(N)=NC(=C3N=2)N2CCN(CC2)C(=O)COC=2C=CC(Br)=CC=2)=C1 FPVUEEFJDLFBQK-UHFFFAOYSA-N 0.000 description 1
- FVWGNQFBISULMT-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(3-methoxyphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound COC1=CC=CC(CCC=2SC3=NC(N)=NC(=C3N=2)N2CCN(CC2)C(=O)COC=2C=CC(Cl)=CC=2)=C1 FVWGNQFBISULMT-UHFFFAOYSA-N 0.000 description 1
- MXCVAZWMSRFAPM-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(3-methoxyphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=C(OC)C=CC=4)SC=3N=C(N)N=2)CC1 MXCVAZWMSRFAPM-UHFFFAOYSA-N 0.000 description 1
- GBYZEOPIDMJAJT-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-bromophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(Br)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 GBYZEOPIDMJAJT-UHFFFAOYSA-N 0.000 description 1
- CGMGZFAXLOIMHO-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-bromophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(Br)=CC=4)SC=3N=C(N)N=2)CC1 CGMGZFAXLOIMHO-UHFFFAOYSA-N 0.000 description 1
- LRSJEAFJBZAIKH-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(3-chlorophenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=CC(Cl)=C1 LRSJEAFJBZAIKH-UHFFFAOYSA-N 0.000 description 1
- UBODTBYRYKMWOV-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-hydroxyphenoxy)ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(O)C=C1 UBODTBYRYKMWOV-UHFFFAOYSA-N 0.000 description 1
- HISDHUMFQUKFPM-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-[4-(trifluoromethoxy)phenoxy]ethanone Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(OC(F)(F)F)C=C1 HISDHUMFQUKFPM-UHFFFAOYSA-N 0.000 description 1
- JXSHRKAJNUZSPG-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-3-(4-bromophenyl)propan-1-one Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)CCC1=CC=C(Br)C=C1 JXSHRKAJNUZSPG-UHFFFAOYSA-N 0.000 description 1
- WFWXCWYFLZYHOJ-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-methylphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-bromophenoxy)ethanone Chemical compound C1=CC(C)=CC=C1CCC1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Br)=CC=3)N=C(N)N=C2S1 WFWXCWYFLZYHOJ-UHFFFAOYSA-N 0.000 description 1
- FIUURXKVYLZCIK-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-methylphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C1=CC(C)=CC=C1CCC1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=C(N)N=C2S1 FIUURXKVYLZCIK-UHFFFAOYSA-N 0.000 description 1
- OTZCMVAMJKQGLU-UHFFFAOYSA-N 1-[4-[5-amino-2-[2-(4-methylphenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(C)=CC=4)SC=3N=C(N)N=2)CC1 OTZCMVAMJKQGLU-UHFFFAOYSA-N 0.000 description 1
- ZMWVFMSNLOCTRT-UHFFFAOYSA-N 1-[4-[5-amino-2-[3-(4-fluorophenyl)propyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 ZMWVFMSNLOCTRT-UHFFFAOYSA-N 0.000 description 1
- ZZWNCBAMZREDMI-UHFFFAOYSA-N 1-[4-[5-amino-2-[3-(4-fluorophenyl)propyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 ZZWNCBAMZREDMI-UHFFFAOYSA-N 0.000 description 1
- NOCYVOZNFYXYHJ-UHFFFAOYSA-N 1-[4-[5-amino-2-[4-(4-fluorophenyl)butyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-chlorophenoxy)ethanone Chemical compound C=12N=C(CCCCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 NOCYVOZNFYXYHJ-UHFFFAOYSA-N 0.000 description 1
- OOQIYNAFAJTXPY-UHFFFAOYSA-N 1-[4-[5-amino-2-[4-(4-fluorophenyl)butyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-(4-methoxyphenoxy)ethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCCCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 OOQIYNAFAJTXPY-UHFFFAOYSA-N 0.000 description 1
- XHQBIYCRFVVHFD-UHFFFAOYSA-N 1-benzothiophen-3-ol Chemical group C1=CC=C2C(O)=CSC2=C1 XHQBIYCRFVVHFD-UHFFFAOYSA-N 0.000 description 1
- IQXXEPZFOOTTBA-UHFFFAOYSA-N 1-benzylpiperazine Chemical compound C=1C=CC=CC=1CN1CCNCC1 IQXXEPZFOOTTBA-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- CCZMQYGSXWZFKI-UHFFFAOYSA-N 1-chloro-4-dichlorophosphoryloxybenzene Chemical compound ClC1=CC=C(OP(Cl)(Cl)=O)C=C1 CCZMQYGSXWZFKI-UHFFFAOYSA-N 0.000 description 1
- NFDXQGNDWIPXQL-UHFFFAOYSA-N 1-cyclooctyldiazocane Chemical group C1CCCCCCC1N1NCCCCCC1 NFDXQGNDWIPXQL-UHFFFAOYSA-N 0.000 description 1
- LDMOEFOXLIZJOW-UHFFFAOYSA-N 1-dodecanesulfonic acid Chemical compound CCCCCCCCCCCCS(O)(=O)=O LDMOEFOXLIZJOW-UHFFFAOYSA-N 0.000 description 1
- WLXGQMVCYPUOLM-UHFFFAOYSA-N 1-hydroxyethanesulfonic acid Chemical compound CC(O)S(O)(=O)=O WLXGQMVCYPUOLM-UHFFFAOYSA-N 0.000 description 1
- CPPGZWWUPFWALU-UHFFFAOYSA-N 1-isocyanato-3-methylbenzene Chemical compound CC1=CC=CC(N=C=O)=C1 CPPGZWWUPFWALU-UHFFFAOYSA-N 0.000 description 1
- 125000001088 1-naphthoyl group Chemical group C1(=CC=CC2=CC=CC=C12)C(=O)* 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- MJWWNBHUIIRNDZ-UHFFFAOYSA-N 1-pentylpiperazine Chemical compound CCCCCN1CCNCC1 MJWWNBHUIIRNDZ-UHFFFAOYSA-N 0.000 description 1
- URGUCKDVRFXOOU-UHFFFAOYSA-N 1-piperazin-1-yl-2-[4-(trifluoromethoxy)phenoxy]ethanone Chemical compound C1=CC(OC(F)(F)F)=CC=C1OCC(=O)N1CCNCC1 URGUCKDVRFXOOU-UHFFFAOYSA-N 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- ZESRJSPZRDMNHY-YFWFAHHUSA-N 11-deoxycorticosterone Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 ZESRJSPZRDMNHY-YFWFAHHUSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- SVUOLADPCWQTTE-UHFFFAOYSA-N 1h-1,2-benzodiazepine Chemical compound N1N=CC=CC2=CC=CC=C12 SVUOLADPCWQTTE-UHFFFAOYSA-N 0.000 description 1
- WVXRAFOPTSTNLL-NKWVEPMBSA-N 2',3'-dideoxyadenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@H]1CC[C@@H](CO)O1 WVXRAFOPTSTNLL-NKWVEPMBSA-N 0.000 description 1
- OHZAHWOAMVVGEL-UHFFFAOYSA-N 2,2'-bithiophene Chemical group C1=CSC(C=2SC=CC=2)=C1 OHZAHWOAMVVGEL-UHFFFAOYSA-N 0.000 description 1
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 1
- 125000005808 2,4,6-trimethoxyphenyl group Chemical group [H][#6]-1=[#6](-[#8]C([H])([H])[H])-[#6](-*)=[#6](-[#8]C([H])([H])[H])-[#6]([H])=[#6]-1-[#8]C([H])([H])[H] 0.000 description 1
- CHHHXKFHOYLYRE-UHFFFAOYSA-M 2,4-Hexadienoic acid, potassium salt (1:1), (2E,4E)- Chemical compound [K+].CC=CC=CC([O-])=O CHHHXKFHOYLYRE-UHFFFAOYSA-M 0.000 description 1
- IIVYOQGIZSPVFZ-UHFFFAOYSA-N 2,5-diamino-4-hydroxy-1h-pyrimidin-6-one;hydrochloride Chemical compound Cl.NC1=NC(O)=C(N)C(=O)N1 IIVYOQGIZSPVFZ-UHFFFAOYSA-N 0.000 description 1
- UBAQQGNYBQOJRN-UHFFFAOYSA-N 2,5-diamino-6h-[1,3]thiazolo[5,4-d]pyrimidin-7-one Chemical compound S1C(N)=NC2=C1N=C(N)NC2=O UBAQQGNYBQOJRN-UHFFFAOYSA-N 0.000 description 1
- SOUUDGAWOJKDRN-UHFFFAOYSA-N 2,6-diamino-1h-pyrimidine-4-thione Chemical compound NC1=CC(=S)N=C(N)N1 SOUUDGAWOJKDRN-UHFFFAOYSA-N 0.000 description 1
- WYDKPTZGVLTYPG-UHFFFAOYSA-N 2,8-diamino-3,7-dihydropurin-6-one Chemical compound N1C(N)=NC(=O)C2=C1N=C(N)N2 WYDKPTZGVLTYPG-UHFFFAOYSA-N 0.000 description 1
- YEGXJZKXGWYYGG-UHFFFAOYSA-N 2-(2,4-dichlorophenoxy)-1-piperazin-1-ylethanone Chemical compound ClC1=CC(Cl)=CC=C1OCC(=O)N1CCNCC1 YEGXJZKXGWYYGG-UHFFFAOYSA-N 0.000 description 1
- LMCJAXCSJHSFRC-UHFFFAOYSA-N 2-(3-chlorophenoxy)-1-piperazin-1-ylethanone Chemical compound ClC1=CC=CC(OCC(=O)N2CCNCC2)=C1 LMCJAXCSJHSFRC-UHFFFAOYSA-N 0.000 description 1
- UGEBOCSGBZAQTQ-UHFFFAOYSA-N 2-(3-methoxyphenyl)-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound COC1=CC=CC(C=2SC3=NC(N)=NC(SC)=C3N=2)=C1 UGEBOCSGBZAQTQ-UHFFFAOYSA-N 0.000 description 1
- VDFSNWBNLMGIEC-UHFFFAOYSA-N 2-(3-methylphenoxy)-1-piperazin-1-ylethanone Chemical compound CC1=CC=CC(OCC(=O)N2CCNCC2)=C1 VDFSNWBNLMGIEC-UHFFFAOYSA-N 0.000 description 1
- VSTIACPBLPUUQL-UHFFFAOYSA-N 2-(4-acetylphenoxy)-1-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(C(=O)C)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 VSTIACPBLPUUQL-UHFFFAOYSA-N 0.000 description 1
- PSAWQBIAETZLGD-UHFFFAOYSA-N 2-(4-acetylphenoxy)-1-piperazin-1-ylethanone Chemical compound C1=CC(C(=O)C)=CC=C1OCC(=O)N1CCNCC1 PSAWQBIAETZLGD-UHFFFAOYSA-N 0.000 description 1
- DTXZJPBYINJULD-UHFFFAOYSA-N 2-(4-bromophenoxy)-1-piperazin-1-ylethanone Chemical compound C1=CC(Br)=CC=C1OCC(=O)N1CCNCC1 DTXZJPBYINJULD-UHFFFAOYSA-N 0.000 description 1
- AYLVKZNYQVSDLY-UHFFFAOYSA-N 2-(4-chloro-2-methylphenoxy)-1-piperazin-1-ylethanone Chemical compound CC1=CC(Cl)=CC=C1OCC(=O)N1CCNCC1 AYLVKZNYQVSDLY-UHFFFAOYSA-N 0.000 description 1
- RIOIXQWQYCVOHU-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1C1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=CN=C2S1 RIOIXQWQYCVOHU-UHFFFAOYSA-N 0.000 description 1
- PBUWPDLSZWMUBF-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1CCC1=NC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=CN=C2S1 PBUWPDLSZWMUBF-UHFFFAOYSA-N 0.000 description 1
- HESMLHIIUNOASP-UHFFFAOYSA-N 2-(4-chlorophenoxy)-1-[4-[6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]ethanone Chemical compound C1=CC(F)=CC=C1C1=CC2=C(N3CCN(CC3)C(=O)COC=3C=CC(Cl)=CC=3)N=CN=C2S1 HESMLHIIUNOASP-UHFFFAOYSA-N 0.000 description 1
- XEISABBVEFAHAO-UHFFFAOYSA-N 2-(4-chlorophenoxy)-2-methyl-1-piperazin-1-ylpropan-1-one Chemical compound C1CNCCN1C(=O)C(C)(C)OC1=CC=C(Cl)C=C1 XEISABBVEFAHAO-UHFFFAOYSA-N 0.000 description 1
- VRBVHQUSAOKVDH-UHFFFAOYSA-N 2-(4-chlorophenoxy)acetyl chloride Chemical compound ClC(=O)COC1=CC=C(Cl)C=C1 VRBVHQUSAOKVDH-UHFFFAOYSA-N 0.000 description 1
- IXFAWIBSYOAYSU-UHFFFAOYSA-N 2-(4-chlorophenyl)-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C1=CC=C(Cl)C=C1 IXFAWIBSYOAYSU-UHFFFAOYSA-N 0.000 description 1
- SSBDHFUDPFLFSS-UHFFFAOYSA-N 2-(4-chlorophenyl)-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CC(Cl)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 SSBDHFUDPFLFSS-UHFFFAOYSA-N 0.000 description 1
- ZRMQGVACZIFSLZ-UHFFFAOYSA-N 2-(4-fluorophenyl)-4h-[1,3]thiazolo[5,4-d]pyrimidine-5,7-dithione Chemical compound C1=CC(F)=CC=C1C1=NC2=C(S)N=C(S)N=C2S1 ZRMQGVACZIFSLZ-UHFFFAOYSA-N 0.000 description 1
- LAVOFWAAXWYLJR-UHFFFAOYSA-N 2-(4-fluorophenyl)-5,7-bis(methylsulfanyl)-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound S1C2=NC(SC)=NC(SC)=C2N=C1C1=CC=C(F)C=C1 LAVOFWAAXWYLJR-UHFFFAOYSA-N 0.000 description 1
- SVXVEIBAXPIVFB-UHFFFAOYSA-N 2-(4-fluorophenyl)-5-methyl-6h-[1,3]thiazolo[5,4-d]pyrimidine-7-thione Chemical compound S1C2=NC(C)=NC(S)=C2N=C1C1=CC=C(F)C=C1 SVXVEIBAXPIVFB-UHFFFAOYSA-N 0.000 description 1
- PRTRKRZZJKJTKM-UHFFFAOYSA-N 2-(4-fluorophenyl)-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound C1=CC(F)=CC=C1C1=NC2=C(N3CCNCC3)N=CN=C2S1 PRTRKRZZJKJTKM-UHFFFAOYSA-N 0.000 description 1
- MGKPFALCNDRSQD-UHFFFAOYSA-N 2-(4-fluorophenyl)acetic acid Chemical compound OC(=O)CC1=CC=C(F)C=C1 MGKPFALCNDRSQD-UHFFFAOYSA-N 0.000 description 1
- IXSCGBODJGIJNN-UHFFFAOYSA-N 2-(4-fluorophenyl)propanoic acid Chemical compound OC(=O)C(C)C1=CC=C(F)C=C1 IXSCGBODJGIJNN-UHFFFAOYSA-N 0.000 description 1
- UFAYWPNRXCCSJH-UHFFFAOYSA-N 2-(4-hydroxyphenoxy)-1-piperazin-1-ylethanone Chemical compound C1=CC(O)=CC=C1OCC(=O)N1CCNCC1 UFAYWPNRXCCSJH-UHFFFAOYSA-N 0.000 description 1
- GPRWAVJHNOECBQ-UHFFFAOYSA-N 2-(4-methoxyphenoxy)-1-piperazin-1-ylethanone Chemical compound C1=CC(OC)=CC=C1OCC(=O)N1CCNCC1 GPRWAVJHNOECBQ-UHFFFAOYSA-N 0.000 description 1
- BHFSBJHPPFJCOS-UHFFFAOYSA-N 2-(4-methoxyphenoxy)acetic acid Chemical compound COC1=CC=C(OCC(O)=O)C=C1 BHFSBJHPPFJCOS-UHFFFAOYSA-N 0.000 description 1
- DOGDGOGCRKKMIA-UHFFFAOYSA-N 2-(4-methylphenyl)-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C1=CC(C)=CC=C1C1=NC2=C(N3CCNCC3)N=C(N)N=C2S1 DOGDGOGCRKKMIA-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- QJJVAUMJKWWKTD-UHFFFAOYSA-N 2-[(3,4-dichloro-5-methyl-1H-pyrrole-2-carbonyl)amino]-4-phenylmethoxy-1,3-benzothiazole-6-carboxylic acid Chemical compound C(C1=CC=CC=C1)OC1=CC(=CC2=C1N=C(S2)NC(=O)C=1NC(=C(C=1Cl)Cl)C)C(=O)O QJJVAUMJKWWKTD-UHFFFAOYSA-N 0.000 description 1
- IPUMKAQQCSJGFB-UHFFFAOYSA-N 2-[(4-chlorophenyl)methyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1CC1=CC=C(Cl)C=C1 IPUMKAQQCSJGFB-UHFFFAOYSA-N 0.000 description 1
- UCFIHBXKQJVCIC-UHFFFAOYSA-N 2-[(4-fluorophenyl)methyl]-7-methylsulfonyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(S(=O)(=O)C)=NC(N)=NC=2SC=1CC1=CC=C(F)C=C1 UCFIHBXKQJVCIC-UHFFFAOYSA-N 0.000 description 1
- DKIAOICEHLLJNU-UHFFFAOYSA-N 2-[1-(4-fluorophenyl)-2-phenylethyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C(C=1C=CC(F)=CC=1)CC1=CC=CC=C1 DKIAOICEHLLJNU-UHFFFAOYSA-N 0.000 description 1
- HKQSTYGBFSURJP-UHFFFAOYSA-N 2-[1-(4-fluorophenyl)ethyl]-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C(C)C1=CC=C(F)C=C1 HKQSTYGBFSURJP-UHFFFAOYSA-N 0.000 description 1
- JDSQBDGCMUXRBM-UHFFFAOYSA-N 2-[2-(2-butoxypropoxy)propoxy]propan-1-ol Chemical group CCCCOC(C)COC(C)COC(C)CO JDSQBDGCMUXRBM-UHFFFAOYSA-N 0.000 description 1
- LQRWOIAZEUKNOQ-UHFFFAOYSA-N 2-[2-(3,4-dimethoxyphenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C1=C(OC)C(OC)=CC=C1CCC1=NC2=C(N3CCNCC3)N=C(N)N=C2S1 LQRWOIAZEUKNOQ-UHFFFAOYSA-N 0.000 description 1
- POHCZCHJNMZJBE-UHFFFAOYSA-N 2-[2-(3-methoxyphenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound COC1=CC=CC(CCC=2SC3=NC(N)=NC(=C3N=2)N2CCNCC2)=C1 POHCZCHJNMZJBE-UHFFFAOYSA-N 0.000 description 1
- TWWRDCDKFACSOM-UHFFFAOYSA-N 2-[2-(4-bromophenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3C=CC(Br)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 TWWRDCDKFACSOM-UHFFFAOYSA-N 0.000 description 1
- RVYUPHXLYVTPBM-UHFFFAOYSA-N 2-[2-(4-fluorophenoxy)ethyl]-7-methylsulfonyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(S(=O)(=O)C)=NC(N)=NC=2SC=1CCOC1=CC=C(F)C=C1 RVYUPHXLYVTPBM-UHFFFAOYSA-N 0.000 description 1
- OXLMODKFMCOXNV-UHFFFAOYSA-N 2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound C1=CC(F)=CC=C1CCC1=NC2=C(N3CCNCC3)N=CN=C2S1 OXLMODKFMCOXNV-UHFFFAOYSA-N 0.000 description 1
- ZORBHCDIZNKKRN-UHFFFAOYSA-N 2-[2-(4-methylphenyl)ethyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C1=CC(C)=CC=C1CCC1=NC2=C(N3CCNCC3)N=C(N)N=C2S1 ZORBHCDIZNKKRN-UHFFFAOYSA-N 0.000 description 1
- AWFGQOQKQHOGAT-UHFFFAOYSA-L 2-[3,5-bis(3-heptyl-4-methyl-1,3-thiazol-3-ium-2-yl)penta-2,4-dienylidene]-3-heptyl-4-methyl-1,3-thiazole;dichloride Chemical compound [Cl-].[Cl-].CCCCCCCN1C(C)=CS\C1=C\C=C(/C1=[N+](C(C)=CS1)CCCCCCC)\C=C\C1=[N+](CCCCCCC)C(C)=CS1 AWFGQOQKQHOGAT-UHFFFAOYSA-L 0.000 description 1
- SMPGSDDXVCKAJW-UHFFFAOYSA-N 2-[3-(4-fluorophenyl)propyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 SMPGSDDXVCKAJW-UHFFFAOYSA-N 0.000 description 1
- IDFZTNSSSIAQRV-UHFFFAOYSA-N 2-[4-(4-fluorophenyl)butyl]-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCCCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N1CCNCC1 IDFZTNSSSIAQRV-UHFFFAOYSA-N 0.000 description 1
- IULOBWFWYDMECP-UHFFFAOYSA-N 2-[4-[2-[(4-chlorophenyl)sulfonylamino]ethyl]phenyl]acetic acid Chemical compound C1=CC(CC(=O)O)=CC=C1CCNS(=O)(=O)C1=CC=C(Cl)C=C1 IULOBWFWYDMECP-UHFFFAOYSA-N 0.000 description 1
- SWHMMZXZNMEPBP-UHFFFAOYSA-N 2-[5-(2-phenylethylamino)furan-2-yl]-[1,3]oxazolo[5,4-d]pyrimidin-7-amine Chemical class N=1C=2C(N)=NC=NC=2OC=1C(O1)=CC=C1NCCC1=CC=CC=C1 SWHMMZXZNMEPBP-UHFFFAOYSA-N 0.000 description 1
- CFWRDBDJAOHXSH-SECBINFHSA-N 2-azaniumylethyl [(2r)-2,3-diacetyloxypropyl] phosphate Chemical compound CC(=O)OC[C@@H](OC(C)=O)COP(O)(=O)OCCN CFWRDBDJAOHXSH-SECBINFHSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- DYWAPFDKPAHSED-UHFFFAOYSA-N 2-cycloheptyloxepane Chemical group C1CCCCCC1C1OCCCCC1 DYWAPFDKPAHSED-UHFFFAOYSA-N 0.000 description 1
- XXNDHVZLNBSADF-UHFFFAOYSA-N 2-cyclohexyl-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C1CCCCC1 XXNDHVZLNBSADF-UHFFFAOYSA-N 0.000 description 1
- KBGTUVBEKLMLLL-UHFFFAOYSA-N 2-cyclopropyl-7-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C1CC1 KBGTUVBEKLMLLL-UHFFFAOYSA-N 0.000 description 1
- WBIQQQGBSDOWNP-UHFFFAOYSA-N 2-dodecylbenzenesulfonic acid Chemical class CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O WBIQQQGBSDOWNP-UHFFFAOYSA-N 0.000 description 1
- 125000004847 2-fluorobenzyl group Chemical group [H]C1=C([H])C(F)=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- 125000002941 2-furyl group Chemical group O1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- ASUDFOJKTJLAIK-UHFFFAOYSA-N 2-methoxyethanamine Chemical compound COCCN ASUDFOJKTJLAIK-UHFFFAOYSA-N 0.000 description 1
- 125000004204 2-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C(OC([H])([H])[H])C([H])=C1[H] 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- 125000001216 2-naphthoyl group Chemical group C1=C(C=CC2=CC=CC=C12)C(=O)* 0.000 description 1
- 125000006024 2-pentenyl group Chemical group 0.000 description 1
- RDPMCUVVUNXNNU-UHFFFAOYSA-N 2-pentyl-7-piperazin-1-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N1=C(N)N=C2SC(CCCCC)=NC2=C1N1CCNCC1 RDPMCUVVUNXNNU-UHFFFAOYSA-N 0.000 description 1
- PKUPAJQAJXVUEK-UHFFFAOYSA-N 2-phenoxyacetyl chloride Chemical compound ClC(=O)COC1=CC=CC=C1 PKUPAJQAJXVUEK-UHFFFAOYSA-N 0.000 description 1
- WQFWIVTXNKRNJZ-UHFFFAOYSA-N 2-piperazin-1-yl-1,3-thiazole Chemical compound C1CNCCN1C1=NC=CS1 WQFWIVTXNKRNJZ-UHFFFAOYSA-N 0.000 description 1
- 125000001494 2-propynyl group Chemical group [H]C#CC([H])([H])* 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- 125000006512 3,4-dichlorobenzyl group Chemical group [H]C1=C(Cl)C(Cl)=C([H])C(=C1[H])C([H])([H])* 0.000 description 1
- 125000003762 3,4-dimethoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C(OC([H])([H])[H])C([H])=C1* 0.000 description 1
- DVYUZHWQRJJQII-UHFFFAOYSA-N 3-(4-bromophenyl)-1-piperazin-1-ylpropan-1-one Chemical compound C1=CC(Br)=CC=C1CCC(=O)N1CCNCC1 DVYUZHWQRJJQII-UHFFFAOYSA-N 0.000 description 1
- QCEDXODAYRWQRH-UHFFFAOYSA-N 3-(4-fluorophenoxy)propanoic acid Chemical compound OC(=O)CCOC1=CC=C(F)C=C1 QCEDXODAYRWQRH-UHFFFAOYSA-N 0.000 description 1
- CHEFSLQUMQCXFW-UHFFFAOYSA-N 3-(4-fluorophenyl)-1-piperazin-1-ylpropan-1-one Chemical compound C1=CC(F)=CC=C1CCC(=O)N1CCNCC1 CHEFSLQUMQCXFW-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-M 3-carboxy-2,3-dihydroxypropanoate Chemical compound OC(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-M 0.000 description 1
- ALKYHXVLJMQRLQ-UHFFFAOYSA-M 3-carboxynaphthalen-2-olate Chemical compound C1=CC=C2C=C(C([O-])=O)C(O)=CC2=C1 ALKYHXVLJMQRLQ-UHFFFAOYSA-M 0.000 description 1
- 125000004080 3-carboxypropanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C(O[H])=O 0.000 description 1
- 125000004179 3-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(Cl)=C1[H] 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- FAXDZWQIWUSWJH-UHFFFAOYSA-N 3-methoxypropan-1-amine Chemical compound COCCCN FAXDZWQIWUSWJH-UHFFFAOYSA-N 0.000 description 1
- 125000006180 3-methyl benzyl group Chemical group [H]C1=C([H])C(=C([H])C(=C1[H])C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- PPAULTVPKLVLII-UHFFFAOYSA-N 4,5-diaminopyrimidine Chemical compound NC1=CN=CN=C1N PPAULTVPKLVLII-UHFFFAOYSA-N 0.000 description 1
- SKKBREFZQQGZAH-UHFFFAOYSA-N 4-(5-amino-2-phenyl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC=CC=2)=C1 SKKBREFZQQGZAH-UHFFFAOYSA-N 0.000 description 1
- HRMLTBQZQRFJTR-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(3-methylphenyl)piperazine-1-carboxamide Chemical compound CC1=CC=CC(NC(=O)N2CCN(CC2)C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)=C1 HRMLTBQZQRFJTR-UHFFFAOYSA-N 0.000 description 1
- BGMMWAVADSFHFV-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(4-chlorophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(Cl)C=C1 BGMMWAVADSFHFV-UHFFFAOYSA-N 0.000 description 1
- BGYPAUDZVQJLJK-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(4-cyanophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(C#N)C=C1 BGYPAUDZVQJLJK-UHFFFAOYSA-N 0.000 description 1
- HLMUNKFFUBFQCX-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(4-methylphenyl)piperazine-1-carbothioamide Chemical compound C1=CC(C)=CC=C1NC(=S)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CC1 HLMUNKFFUBFQCX-UHFFFAOYSA-N 0.000 description 1
- UWTRKIJAGTTXNM-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-(4-methylphenyl)piperazine-1-carboxamide Chemical compound C1=CC(C)=CC=C1NC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CC1 UWTRKIJAGTTXNM-UHFFFAOYSA-N 0.000 description 1
- CSUNQMPVPSYRGH-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-[(4-methoxyphenyl)methyl]piperazine-1-carboxamide Chemical compound C1=CC(OC)=CC=C1CNC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=NC=CC=2)CC1 CSUNQMPVPSYRGH-UHFFFAOYSA-N 0.000 description 1
- MWMQTBSTAVJWAE-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-[4-(trifluoromethyl)phenyl]piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(C(F)(F)F)C=C1 MWMQTBSTAVJWAE-UHFFFAOYSA-N 0.000 description 1
- QBGFMDIGQMQAAY-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-cyclohexylpiperazine-1-carboxamide Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1CCCCC1 QBGFMDIGQMQAAY-UHFFFAOYSA-N 0.000 description 1
- YMAZHUVQDZKBHV-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-hexylpiperazine-1-carboxamide Chemical compound C1CN(C(=O)NCCCCCC)CCN1C1=NC(N)=NC2=C1N=C(C=1C=NC=CC=1)S2 YMAZHUVQDZKBHV-UHFFFAOYSA-N 0.000 description 1
- PPMYXFAWZFQQAR-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-methyl-n-(4-methylphenyl)piperazine-1-carboxamide Chemical compound C=1C=C(C)C=CC=1N(C)C(=O)N(CC1)CCN1C(C=1N=2)=NC(N)=NC=1SC=2C1=CC=CN=C1 PPMYXFAWZFQQAR-UHFFFAOYSA-N 0.000 description 1
- RHWUBFTZEIZGSI-UHFFFAOYSA-N 4-(5-amino-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-7-yl)-n-phenylpiperazine-1-carboxamide Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=CC=C1 RHWUBFTZEIZGSI-UHFFFAOYSA-N 0.000 description 1
- VMRKHSUWSDADFU-LLVKDONJSA-N 4-[(3r)-3-aminopyrrolidin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine Chemical compound C1[C@H](N)CCN1C1=NC(N)=NC2=C1C=C(C=1C=CC(F)=CC=1)S2 VMRKHSUWSDADFU-LLVKDONJSA-N 0.000 description 1
- ZJPUFGIYLGKUMA-UHFFFAOYSA-N 4-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]-n,n-di(propan-2-yl)piperazine-1-carboxamide Chemical compound C1CN(C(=O)N(C(C)C)C(C)C)CCN1C1=NC(N)=NC2=C1C=C(C=1C=CC(F)=CC=1)S2 ZJPUFGIYLGKUMA-UHFFFAOYSA-N 0.000 description 1
- XBUNEQMOGILVTC-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(2,4-difluorophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(F)C=C1F XBUNEQMOGILVTC-UHFFFAOYSA-N 0.000 description 1
- GRKDEJGSDGFCOC-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(2-methoxyphenyl)piperazine-1-carboxamide Chemical compound COC1=CC=CC=C1NC(=O)N1CCN(C=2C=3N=C(SC=3N=C(N)N=2)C=2C=CC(F)=CC=2)CC1 GRKDEJGSDGFCOC-UHFFFAOYSA-N 0.000 description 1
- XTSUOPOZBGPSGW-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(4-bromophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(Br)C=C1 XTSUOPOZBGPSGW-UHFFFAOYSA-N 0.000 description 1
- DNZSAWLKQKHUFZ-UHFFFAOYSA-N 4-[5-amino-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(4-cyanophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(C#N)C=C1 DNZSAWLKQKHUFZ-UHFFFAOYSA-N 0.000 description 1
- MFYPDRPWLKSPKA-UHFFFAOYSA-N 4-[5-amino-2-[(4-fluorophenyl)methyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(4-cyanophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(CC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(C#N)C=C1 MFYPDRPWLKSPKA-UHFFFAOYSA-N 0.000 description 1
- NRZNYMJPPMPOML-UHFFFAOYSA-N 4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]-n-(4-cyanophenyl)piperazine-1-carboxamide Chemical compound C=12N=C(CCC=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)NC1=CC=C(C#N)C=C1 NRZNYMJPPMPOML-UHFFFAOYSA-N 0.000 description 1
- YLDCUKJMEKGGFI-QCSRICIXSA-N 4-acetamidobenzoic acid;9-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3h-purin-6-one;1-(dimethylamino)propan-2-ol Chemical compound CC(O)CN(C)C.CC(O)CN(C)C.CC(O)CN(C)C.CC(=O)NC1=CC=C(C(O)=O)C=C1.CC(=O)NC1=CC=C(C(O)=O)C=C1.CC(=O)NC1=CC=C(C(O)=O)C=C1.O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(NC=NC2=O)=C2N=C1 YLDCUKJMEKGGFI-QCSRICIXSA-N 0.000 description 1
- 125000006283 4-chlorobenzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1Cl)C([H])([H])* 0.000 description 1
- LHGMCUVJFRBVBH-UHFFFAOYSA-N 4-chloropyrimidin-5-amine Chemical class NC1=CN=CN=C1Cl LHGMCUVJFRBVBH-UHFFFAOYSA-N 0.000 description 1
- NZCRUBBNZGVREM-UHFFFAOYSA-N 4-chlorothieno[2,3-d]pyrimidine Chemical class ClC1=NC=NC2=C1C=CS2 NZCRUBBNZGVREM-UHFFFAOYSA-N 0.000 description 1
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 1
- WNWVKZTYMQWFHE-UHFFFAOYSA-N 4-ethylmorpholine Chemical compound [CH2]CN1CCOCC1 WNWVKZTYMQWFHE-UHFFFAOYSA-N 0.000 description 1
- MTFGZYIESVROQD-UHFFFAOYSA-N 4-fluoro-n-(4-hydroxy-2-methyl-6-oxo-1h-pyrimidin-5-yl)benzamide Chemical compound OC1=NC(C)=NC(O)=C1NC(=O)C1=CC=C(F)C=C1 MTFGZYIESVROQD-UHFFFAOYSA-N 0.000 description 1
- XOENXAVLBORGFQ-UHFFFAOYSA-N 4-fluoro-n-(6-hydroxy-4-oxo-2-sulfanylidene-1h-pyrimidin-5-yl)benzamide Chemical compound OC1=NC(S)=NC(O)=C1NC(=O)C1=CC=C(F)C=C1 XOENXAVLBORGFQ-UHFFFAOYSA-N 0.000 description 1
- CZKLEJHVLCMVQR-UHFFFAOYSA-N 4-fluorobenzoyl chloride Chemical compound FC1=CC=C(C(Cl)=O)C=C1 CZKLEJHVLCMVQR-UHFFFAOYSA-N 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- 125000006181 4-methyl benzyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])C([H])([H])* 0.000 description 1
- UQRONKZLYKUEMO-UHFFFAOYSA-N 4-methyl-1-(2,4,6-trimethylphenyl)pent-4-en-2-one Chemical group CC(=C)CC(=O)Cc1c(C)cc(C)cc1C UQRONKZLYKUEMO-UHFFFAOYSA-N 0.000 description 1
- IGFHQQFPSIBGKE-UHFFFAOYSA-N 4-nonylphenol Chemical compound CCCCCCCCCC1=CC=C(O)C=C1 IGFHQQFPSIBGKE-UHFFFAOYSA-N 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- MGYGFNQQGAQEON-UHFFFAOYSA-N 4-tolyl isocyanate Chemical compound CC1=CC=C(N=C=O)C=C1 MGYGFNQQGAQEON-UHFFFAOYSA-N 0.000 description 1
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical compound C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 1
- XBGQGAPUUJJOTA-KWLUMGGGSA-N 4b52439y33 Chemical compound O.C([C@@H]1C2)C3=CC=CC=C3C[C@@]1(C(=O)CO)[C@]1(C)[C@@H]2[C@H](CCC=2[C@@]3(C=CC(=O)C=2)C)[C@]3(F)[C@@H](O)C1 XBGQGAPUUJJOTA-KWLUMGGGSA-N 0.000 description 1
- CEGRCQZYSINSRI-UHFFFAOYSA-N 5,7-bis(benzylsulfanyl)-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound C1=CC(F)=CC=C1C(SC1=NC(SCC=2C=CC=CC=2)=N2)=NC1=C2SCC1=CC=CC=C1 CEGRCQZYSINSRI-UHFFFAOYSA-N 0.000 description 1
- YPQWPFBLOQNPBE-UHFFFAOYSA-N 5,7-bis(butylsulfanyl)-2-(4-fluorophenyl)-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound S1C2=NC(SCCCC)=NC(SCCCC)=C2N=C1C1=CC=C(F)C=C1 YPQWPFBLOQNPBE-UHFFFAOYSA-N 0.000 description 1
- HCXJFMDOHDNDCC-UHFFFAOYSA-N 5-$l^{1}-oxidanyl-3,4-dihydropyrrol-2-one Chemical group O=C1CCC(=O)[N]1 HCXJFMDOHDNDCC-UHFFFAOYSA-N 0.000 description 1
- PIKTYDBZFQILGB-UHFFFAOYSA-N 5-amino-2-[(4-fluorophenyl)methyl]-6h-[1,3]thiazolo[5,4-d]pyrimidine-7-thione Chemical compound S1C2=NC(N)=NC(S)=C2N=C1CC1=CC=C(F)C=C1 PIKTYDBZFQILGB-UHFFFAOYSA-N 0.000 description 1
- CNFKHRLFOBAPKJ-UHFFFAOYSA-N 5-amino-2-[2-(4-fluorophenoxy)ethyl]-6h-[1,3]thiazolo[5,4-d]pyrimidine-7-thione Chemical compound S1C2=NC(N)=NC(S)=C2N=C1CCOC1=CC=C(F)C=C1 CNFKHRLFOBAPKJ-UHFFFAOYSA-N 0.000 description 1
- QCLAELJXWLOJAW-UHFFFAOYSA-N 5-amino-2-[2-(4-fluorophenyl)ethyl]-6h-[1,3]thiazolo[5,4-d]pyrimidine-7-thione Chemical compound S1C2=NC(N)=NC(S)=C2N=C1CCC1=CC=C(F)C=C1 QCLAELJXWLOJAW-UHFFFAOYSA-N 0.000 description 1
- YWKOMSVMLIYEKS-UHFFFAOYSA-N 5-bromothieno[2,3-d]pyrimidine Chemical compound C1=NC=C2C(Br)=CSC2=N1 YWKOMSVMLIYEKS-UHFFFAOYSA-N 0.000 description 1
- IZSHZLKNFQAAKX-UHFFFAOYSA-N 5-cyclopenta-2,4-dien-1-ylcyclopenta-1,3-diene Chemical group C1=CC=CC1C1C=CC=C1 IZSHZLKNFQAAKX-UHFFFAOYSA-N 0.000 description 1
- YZFUAIWTNMWSEP-UHFFFAOYSA-N 5-nitrosopyrimidine Chemical class O=NC1=CN=CN=C1 YZFUAIWTNMWSEP-UHFFFAOYSA-N 0.000 description 1
- UBOIMZIXNXGQOH-RTWVSBIPSA-N 58497-00-0 Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)COC(=O)CC)[C@@]2(C)C[C@@H]1O UBOIMZIXNXGQOH-RTWVSBIPSA-N 0.000 description 1
- TZBDXWBBMOEVPI-XBQQDWOSSA-N 58524-83-7 Chemical compound O=C([C@]12[C@@]3(C)C[C@H](O)[C@]4(F)[C@@]5(C)C=CC(=O)C=C5[C@@H](F)C[C@H]4[C@@H]3C[C@H]1OC(O2)(C)C)COC(=O)C1CC1 TZBDXWBBMOEVPI-XBQQDWOSSA-N 0.000 description 1
- YMMWIKRJZHFROV-UHFFFAOYSA-N 6-(1h-1,2,4-triazol-5-yl)-7h-purine Chemical class N1C=NC(C=2C=3NC=NC=3N=CN=2)=N1 YMMWIKRJZHFROV-UHFFFAOYSA-N 0.000 description 1
- ZKBQDFAWXLTYKS-UHFFFAOYSA-N 6-Chloro-1H-purine Chemical class ClC1=NC=NC2=C1NC=N2 ZKBQDFAWXLTYKS-UHFFFAOYSA-N 0.000 description 1
- YFYYRKDBDBILSD-UHFFFAOYSA-N 6-amino-2-sulfanylidene-1h-pyrimidin-4-one Chemical compound NC1=CC(=O)NC(=S)N1 YFYYRKDBDBILSD-UHFFFAOYSA-N 0.000 description 1
- CCQQNRDRMMVUQI-UHFFFAOYSA-N 6-bromothieno[2,3-d]pyrimidine Chemical compound N1=CN=C2SC(Br)=CC2=C1 CCQQNRDRMMVUQI-UHFFFAOYSA-N 0.000 description 1
- QJIUMVUZDYPQRT-UHFFFAOYSA-N 6-chloro-2,4-pyrimidinediamine Chemical compound NC1=CC(Cl)=NC(N)=N1 QJIUMVUZDYPQRT-UHFFFAOYSA-N 0.000 description 1
- IUIYEHXOIMMQJY-NGXOUOCZSA-N 60135-22-0 Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)C(OC)OC)[C@@]2(C)C[C@@H]1O IUIYEHXOIMMQJY-NGXOUOCZSA-N 0.000 description 1
- JUKPTTSTHGFSGD-UHFFFAOYSA-N 7-chloro-2-(4-fluorophenyl)-5-methyl-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound S1C2=NC(C)=NC(Cl)=C2N=C1C1=CC=C(F)C=C1 JUKPTTSTHGFSGD-UHFFFAOYSA-N 0.000 description 1
- FAYGVDHTAUGZLD-UHFFFAOYSA-N 7-ethoxy-2-(4-fluorophenyl)-5-methylsulfanyl-[1,3]thiazolo[5,4-d]pyrimidine Chemical compound N=1C=2C(OCC)=NC(SC)=NC=2SC=1C1=CC=C(F)C=C1 FAYGVDHTAUGZLD-UHFFFAOYSA-N 0.000 description 1
- PDTXWPHNZWMTOW-UHFFFAOYSA-N 7-methylsulfanyl-2-(2-phenylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1CCC1=CC=CC=C1 PDTXWPHNZWMTOW-UHFFFAOYSA-N 0.000 description 1
- JKHNRZQMCROQLW-UHFFFAOYSA-N 7-methylsulfanyl-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound N=1C=2C(SC)=NC(N)=NC=2SC=1C1=CC=CN=C1 JKHNRZQMCROQLW-UHFFFAOYSA-N 0.000 description 1
- VCFYKQVLQBUZSX-UHFFFAOYSA-N 7-piperazin-1-yl-2-(2-thiophen-2-ylethyl)-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(CCC=3SC=CC=3)SC2=NC(N)=NC=1N1CCNCC1 VCFYKQVLQBUZSX-UHFFFAOYSA-N 0.000 description 1
- FAPHFXKXUWZLLJ-UHFFFAOYSA-N 7-piperazin-1-yl-2-pyridin-2-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3N=CC=CC=3)SC2=NC(N)=NC=1N1CCNCC1 FAPHFXKXUWZLLJ-UHFFFAOYSA-N 0.000 description 1
- HHCFYILHMVJKAW-UHFFFAOYSA-N 7-piperazin-1-yl-2-pyridin-3-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=NC=CC=3)SC2=NC(N)=NC=1N1CCNCC1 HHCFYILHMVJKAW-UHFFFAOYSA-N 0.000 description 1
- GISKMZJKXVZYRY-UHFFFAOYSA-N 7-piperazin-1-yl-2-pyridin-4-yl-[1,3]thiazolo[5,4-d]pyrimidin-5-amine Chemical compound C=12N=C(C=3C=CN=CC=3)SC2=NC(N)=NC=1N1CCNCC1 GISKMZJKXVZYRY-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- RTAPDZBZLSXHQQ-UHFFFAOYSA-N 8-methyl-3,7-dihydropurine-2,6-dione Chemical class N1C(=O)NC(=O)C2=C1N=C(C)N2 RTAPDZBZLSXHQQ-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- LRFVTYWOQMYALW-UHFFFAOYSA-N 9H-xanthine Chemical class O=C1NC(=O)NC2=C1NC=N2 LRFVTYWOQMYALW-UHFFFAOYSA-N 0.000 description 1
- 208000021959 Abnormal metabolism Diseases 0.000 description 1
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Chemical compound CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 1
- OQLZINXFSUDMHM-UHFFFAOYSA-N Acetamidine Chemical compound CC(N)=N OQLZINXFSUDMHM-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 208000026872 Addison Disease Diseases 0.000 description 1
- 102000009346 Adenosine receptors Human genes 0.000 description 1
- 108050000203 Adenosine receptors Proteins 0.000 description 1
- 229910002012 Aerosil® Inorganic materials 0.000 description 1
- 238000010953 Ames test Methods 0.000 description 1
- 231100000039 Ames test Toxicity 0.000 description 1
- 208000032467 Aplastic anaemia Diseases 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 206010003658 Atrial Fibrillation Diseases 0.000 description 1
- 206010003827 Autoimmune hepatitis Diseases 0.000 description 1
- VGGGPCQERPFHOB-MCIONIFRSA-N Bestatin Chemical compound CC(C)C[C@H](C(O)=O)NC(=O)[C@@H](O)[C@H](N)CC1=CC=CC=C1 VGGGPCQERPFHOB-MCIONIFRSA-N 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 206010004659 Biliary cirrhosis Diseases 0.000 description 1
- 208000033222 Biliary cirrhosis primary Diseases 0.000 description 1
- 206010065553 Bone marrow failure Diseases 0.000 description 1
- COAVSPBYGMEKAR-UHFFFAOYSA-N C(C)(C)N(C(=O)N1C(N=CC2=C1SC(=C2)C1=CC=C(C=C1)F)N1CCNCC1)C(C)C Chemical compound C(C)(C)N(C(=O)N1C(N=CC2=C1SC(=C2)C1=CC=C(C=C1)F)N1CCNCC1)C(C)C COAVSPBYGMEKAR-UHFFFAOYSA-N 0.000 description 1
- BJOUYVVAIVTHMQ-UHFFFAOYSA-N C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)O)=NC=1NC1=CC=C(F)C(Cl)=C1 Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)O)=NC=1NC1=CC=C(F)C(Cl)=C1 BJOUYVVAIVTHMQ-UHFFFAOYSA-N 0.000 description 1
- 101710143185 Calcium-dependent protein kinase 1 Proteins 0.000 description 1
- 102000000584 Calmodulin Human genes 0.000 description 1
- 108010041952 Calmodulin Proteins 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 206010062746 Carditis Diseases 0.000 description 1
- 102000011727 Caspases Human genes 0.000 description 1
- 108010076667 Caspases Proteins 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 102000000844 Cell Surface Receptors Human genes 0.000 description 1
- 108010001857 Cell Surface Receptors Proteins 0.000 description 1
- 108010012236 Chemokines Proteins 0.000 description 1
- 102000019034 Chemokines Human genes 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 206010009900 Colitis ulcerative Diseases 0.000 description 1
- 206010011224 Cough Diseases 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- 108010036941 Cyclosporins Proteins 0.000 description 1
- 206010011831 Cytomegalovirus infection Diseases 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 238000009007 Diagnostic Kit Methods 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 208000000059 Dyspnea Diseases 0.000 description 1
- 206010013975 Dyspnoeas Diseases 0.000 description 1
- 102100025137 Early activation antigen CD69 Human genes 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 208000017701 Endocrine disease Diseases 0.000 description 1
- 108010008165 Etanercept Proteins 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical class C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 238000006937 Gewald synthesis reaction Methods 0.000 description 1
- 206010018364 Glomerulonephritis Diseases 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 208000024869 Goodpasture syndrome Diseases 0.000 description 1
- 208000003807 Graves Disease Diseases 0.000 description 1
- 208000015023 Graves' disease Diseases 0.000 description 1
- 102000009465 Growth Factor Receptors Human genes 0.000 description 1
- 108010009202 Growth Factor Receptors Proteins 0.000 description 1
- 229920002907 Guar gum Polymers 0.000 description 1
- 102000006354 HLA-DR Antigens Human genes 0.000 description 1
- 108010058597 HLA-DR Antigens Proteins 0.000 description 1
- 206010066476 Haematological malignancy Diseases 0.000 description 1
- 208000002250 Hematologic Neoplasms Diseases 0.000 description 1
- 206010019851 Hepatotoxicity Diseases 0.000 description 1
- 208000029433 Herpesviridae infectious disease Diseases 0.000 description 1
- 206010020112 Hirsutism Diseases 0.000 description 1
- 101000934374 Homo sapiens Early activation antigen CD69 Proteins 0.000 description 1
- 101000628949 Homo sapiens Mitogen-activated protein kinase 10 Proteins 0.000 description 1
- 101000950695 Homo sapiens Mitogen-activated protein kinase 8 Proteins 0.000 description 1
- 101000777277 Homo sapiens Serine/threonine-protein kinase Chk2 Proteins 0.000 description 1
- 101000914484 Homo sapiens T-lymphocyte activation antigen CD80 Proteins 0.000 description 1
- 101000835093 Homo sapiens Transferrin receptor protein 1 Proteins 0.000 description 1
- 101000679851 Homo sapiens Tumor necrosis factor receptor superfamily member 4 Proteins 0.000 description 1
- 101000934996 Homo sapiens Tyrosine-protein kinase JAK3 Proteins 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 108060006678 I-kappa-B kinase Proteins 0.000 description 1
- 206010021245 Idiopathic thrombocytopenic purpura Diseases 0.000 description 1
- 102100021854 Inhibitor of nuclear factor kappa-B kinase subunit beta Human genes 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 102000003996 Interferon-beta Human genes 0.000 description 1
- 108090000467 Interferon-beta Proteins 0.000 description 1
- 102000051628 Interleukin-1 receptor antagonist Human genes 0.000 description 1
- 108700021006 Interleukin-1 receptor antagonist Proteins 0.000 description 1
- 102000010789 Interleukin-2 Receptors Human genes 0.000 description 1
- 108010038453 Interleukin-2 Receptors Proteins 0.000 description 1
- 108090000978 Interleukin-4 Proteins 0.000 description 1
- 108090001007 Interleukin-8 Proteins 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 1
- 108010024121 Janus Kinases Proteins 0.000 description 1
- 102000015617 Janus Kinases Human genes 0.000 description 1
- 239000005909 Kieselgur Substances 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- QAQJMLQRFWZOBN-LAUBAEHRSA-N L-ascorbyl-6-palmitate Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](O)[C@H]1OC(=O)C(O)=C1O QAQJMLQRFWZOBN-LAUBAEHRSA-N 0.000 description 1
- 239000011786 L-ascorbyl-6-palmitate Substances 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- 229920001491 Lentinan Polymers 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 206010067125 Liver injury Diseases 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- 102000043131 MHC class II family Human genes 0.000 description 1
- 108091054438 MHC class II family Proteins 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 108700018351 Major Histocompatibility Complex Proteins 0.000 description 1
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical group O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- FQISKWAFAHGMGT-SGJOWKDISA-M Methylprednisolone sodium succinate Chemical compound [Na+].C([C@@]12C)=CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2[C@@H](O)C[C@]2(C)[C@@](O)(C(=O)COC(=O)CCC([O-])=O)CC[C@H]21 FQISKWAFAHGMGT-SGJOWKDISA-M 0.000 description 1
- 102100026931 Mitogen-activated protein kinase 10 Human genes 0.000 description 1
- 102100037808 Mitogen-activated protein kinase 8 Human genes 0.000 description 1
- HZQDCMWJEBCWBR-UUOKFMHZSA-N Mizoribine Chemical compound OC1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 HZQDCMWJEBCWBR-UUOKFMHZSA-N 0.000 description 1
- 208000009525 Myocarditis Diseases 0.000 description 1
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- 150000001204 N-oxides Chemical group 0.000 description 1
- 102000003945 NF-kappa B Human genes 0.000 description 1
- 108010057466 NF-kappa B Proteins 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 206010029155 Nephropathy toxic Diseases 0.000 description 1
- 206010029350 Neurotoxicity Diseases 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 208000021384 Obsessive-Compulsive disease Diseases 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical group C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- 108091007960 PI3Ks Proteins 0.000 description 1
- 102000038030 PI3Ks Human genes 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 241000721454 Pemphigus Species 0.000 description 1
- BYPFEZZEUUWMEJ-UHFFFAOYSA-N Pentoxifylline Chemical compound O=C1N(CCCCC(=O)C)C(=O)N(C)C2=C1N(C)C=N2 BYPFEZZEUUWMEJ-UHFFFAOYSA-N 0.000 description 1
- 208000031845 Pernicious anaemia Diseases 0.000 description 1
- 108090000430 Phosphatidylinositol 3-kinases Proteins 0.000 description 1
- 229940123932 Phosphodiesterase 4 inhibitor Drugs 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical group [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 206010035664 Pneumonia Diseases 0.000 description 1
- 206010035742 Pneumonitis Diseases 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920001214 Polysorbate 60 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- 102000004257 Potassium Channel Human genes 0.000 description 1
- 208000012654 Primary biliary cholangitis Diseases 0.000 description 1
- 101710101148 Probable 6-oxopurine nucleoside phosphorylase Proteins 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical compound CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- 102000007327 Protamines Human genes 0.000 description 1
- 108010007568 Protamines Proteins 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- 201000001263 Psoriatic Arthritis Diseases 0.000 description 1
- 208000036824 Psoriatic arthropathy Diseases 0.000 description 1
- 208000028017 Psychotic disease Diseases 0.000 description 1
- 102000030764 Purine-nucleoside phosphorylase Human genes 0.000 description 1
- 239000007868 Raney catalyst Substances 0.000 description 1
- NPXOKRUENSOPAO-UHFFFAOYSA-N Raney nickel Chemical compound [Al].[Ni] NPXOKRUENSOPAO-UHFFFAOYSA-N 0.000 description 1
- 229910000564 Raney nickel Inorganic materials 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 229910006124 SOCl2 Inorganic materials 0.000 description 1
- 239000002262 Schiff base Substances 0.000 description 1
- 150000004753 Schiff bases Chemical class 0.000 description 1
- 206010039710 Scleroderma Diseases 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical group [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- 102100031075 Serine/threonine-protein kinase Chk2 Human genes 0.000 description 1
- 208000021386 Sjogren Syndrome Diseases 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- DWAQJAXMDSEUJJ-UHFFFAOYSA-M Sodium bisulfite Chemical compound [Na+].OS([O-])=O DWAQJAXMDSEUJJ-UHFFFAOYSA-M 0.000 description 1
- IOEJYZSZYUROLN-UHFFFAOYSA-M Sodium diethyldithiocarbamate Chemical compound [Na+].CCN(CC)C([S-])=S IOEJYZSZYUROLN-UHFFFAOYSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 239000004141 Sodium laurylsulphate Substances 0.000 description 1
- 238000003477 Sonogashira cross-coupling reaction Methods 0.000 description 1
- 239000004147 Sorbitan trioleate Substances 0.000 description 1
- PRXRUNOAOLTIEF-ADSICKODSA-N Sorbitan trioleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@@H](OC(=O)CCCCCCC\C=C/CCCCCCCC)[C@H]1OC[C@H](O)[C@H]1OC(=O)CCCCCCC\C=C/CCCCCCCC PRXRUNOAOLTIEF-ADSICKODSA-N 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 239000005864 Sulphur Substances 0.000 description 1
- 208000018359 Systemic autoimmune disease Diseases 0.000 description 1
- 230000006052 T cell proliferation Effects 0.000 description 1
- 108091008874 T cell receptors Proteins 0.000 description 1
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 1
- 102100027222 T-lymphocyte activation antigen CD80 Human genes 0.000 description 1
- 102000003566 TRPV1 Human genes 0.000 description 1
- 206010043275 Teratogenicity Diseases 0.000 description 1
- 208000031981 Thrombocytopenic Idiopathic Purpura Diseases 0.000 description 1
- 101800001703 Thymopentin Proteins 0.000 description 1
- 102400000160 Thymopentin Human genes 0.000 description 1
- DXEXNWDGDYUITL-FXSSSKFRSA-N Tipredane Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1CC[C@@](SC)(SCC)[C@@]1(C)C[C@@H]2O DXEXNWDGDYUITL-FXSSSKFRSA-N 0.000 description 1
- 206010044221 Toxic encephalopathy Diseases 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 102100026144 Transferrin receptor protein 1 Human genes 0.000 description 1
- 102100029613 Transient receptor potential cation channel subfamily V member 1 Human genes 0.000 description 1
- 108050004388 Transient receptor potential cation channel subfamily V member 1 Proteins 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 101150016206 Trpv1 gene Proteins 0.000 description 1
- 102100022153 Tumor necrosis factor receptor superfamily member 4 Human genes 0.000 description 1
- 108091005906 Type I transmembrane proteins Proteins 0.000 description 1
- 108091005956 Type II transmembrane proteins Proteins 0.000 description 1
- 102100025387 Tyrosine-protein kinase JAK3 Human genes 0.000 description 1
- 201000006704 Ulcerative Colitis Diseases 0.000 description 1
- 206010047115 Vasculitis Diseases 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- TUBCXUULQSAOLY-UHFFFAOYSA-N [1,3]oxazolo[5,4-d]pyrimidine;[1,3]thiazolo[5,4-d]pyrimidine Chemical compound N1=CN=C2OC=NC2=C1.N1=CN=C2SC=NC2=C1 TUBCXUULQSAOLY-UHFFFAOYSA-N 0.000 description 1
- CVWNWFURXOCMRH-UHFFFAOYSA-N [2-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-cyclohexylmethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1C1CNCCN1C(=O)C1CCCCC1 CVWNWFURXOCMRH-UHFFFAOYSA-N 0.000 description 1
- SSWNCVHPWOKMEB-UHFFFAOYSA-N [2-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-pyridin-4-ylmethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1C1CNCCN1C(=O)C1=CC=NC=C1 SSWNCVHPWOKMEB-UHFFFAOYSA-N 0.000 description 1
- ZISRWUVYOZPEFC-UHFFFAOYSA-N [4-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]piperazin-1-yl]-cyclohexylmethanone Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(N)=NC=1N(CC1)CCN1C(=O)C1CCCCC1 ZISRWUVYOZPEFC-UHFFFAOYSA-N 0.000 description 1
- CLZISMQKJZCZDN-UHFFFAOYSA-N [benzotriazol-1-yloxy(dimethylamino)methylidene]-dimethylazanium Chemical compound C1=CC=C2N(OC(N(C)C)=[N+](C)C)N=NC2=C1 CLZISMQKJZCZDN-UHFFFAOYSA-N 0.000 description 1
- MCGSCOLBFJQGHM-SCZZXKLOSA-N abacavir Chemical compound C=12N=CN([C@H]3C=C[C@@H](CO)C3)C2=NC(N)=NC=1NC1CC1 MCGSCOLBFJQGHM-SCZZXKLOSA-N 0.000 description 1
- 229960004748 abacavir Drugs 0.000 description 1
- 239000006096 absorbing agent Substances 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- 239000001785 acacia senegal l. willd gum Substances 0.000 description 1
- 229960005327 acemannan Drugs 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 229960004150 aciclovir Drugs 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 125000003647 acryloyl group Chemical group O=C([*])C([H])=C([H])[H] 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 239000003121 adenosine kinase inhibitor Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000004703 alkoxides Chemical class 0.000 description 1
- 125000004171 alkoxy aryl group Chemical group 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000000961 alloantigen Effects 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 229950010999 amiprilose Drugs 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 230000003042 antagnostic effect Effects 0.000 description 1
- 230000008485 antagonism Effects 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 239000002259 anti human immunodeficiency virus agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 229940124411 anti-hiv antiviral agent Drugs 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 230000030741 antigen processing and presentation Effects 0.000 description 1
- 210000000612 antigen-presenting cell Anatomy 0.000 description 1
- 230000000890 antigenic effect Effects 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 239000003443 antiviral agent Substances 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 239000012062 aqueous buffer Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical group 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- 125000004391 aryl sulfonyl group Chemical group 0.000 description 1
- 125000005110 aryl thio group Chemical group 0.000 description 1
- 235000010385 ascorbyl palmitate Nutrition 0.000 description 1
- 239000000305 astragalus gummifer gum Substances 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- 201000003710 autoimmune thrombocytopenic purpura Diseases 0.000 description 1
- 125000005602 azabenzimidazolyl group Chemical group 0.000 description 1
- 125000005334 azaindolyl group Chemical group N1N=C(C2=CC=CC=C12)* 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 125000004045 azirinyl group Chemical group 0.000 description 1
- 239000000987 azo dye Substances 0.000 description 1
- 210000001142 back Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 235000012216 bentonite Nutrition 0.000 description 1
- JUHORIMYRDESRB-UHFFFAOYSA-N benzathine Chemical compound C=1C=CC=CC=1CNCCNCC1=CC=CC=C1 JUHORIMYRDESRB-UHFFFAOYSA-N 0.000 description 1
- 229940077388 benzenesulfonate Drugs 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 1
- 125000004604 benzisothiazolyl group Chemical group S1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 229940049706 benzodiazepine Drugs 0.000 description 1
- 125000004602 benzodiazinyl group Chemical group N1=NC(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000000928 benzodioxinyl group Chemical group O1C(=COC2=C1C=CC=C2)* 0.000 description 1
- 125000002047 benzodioxolyl group Chemical group O1OC(C2=C1C=CC=C2)* 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 1
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004600 benzothiopyranyl group Chemical group S1C(C=CC2=C1C=CC=C2)* 0.000 description 1
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 1
- 125000004622 benzoxazinyl group Chemical group O1NC(=CC2=C1C=CC=C2)* 0.000 description 1
- PASDCCFISLVPSO-UHFFFAOYSA-N benzoyl chloride Chemical compound ClC(=O)C1=CC=CC=C1 PASDCCFISLVPSO-UHFFFAOYSA-N 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- HSDAJNMJOMSNEV-UHFFFAOYSA-N benzyl chloroformate Chemical compound ClC(=O)OCC1=CC=CC=C1 HSDAJNMJOMSNEV-UHFFFAOYSA-N 0.000 description 1
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 1
- 238000012925 biological evaluation Methods 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000002798 bone marrow cell Anatomy 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- PHEZJEYUWHETKO-UHFFFAOYSA-N brequinar Chemical compound N1=C2C=CC(F)=CC2=C(C(O)=O)C(C)=C1C(C=C1)=CC=C1C1=CC=CC=C1F PHEZJEYUWHETKO-UHFFFAOYSA-N 0.000 description 1
- 229950010231 brequinar Drugs 0.000 description 1
- RDHPKYGYEGBMSE-UHFFFAOYSA-N bromoethane Chemical compound CCBr RDHPKYGYEGBMSE-UHFFFAOYSA-N 0.000 description 1
- 229960004272 bucillamine Drugs 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000004067 bulking agent Substances 0.000 description 1
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 1
- 125000004063 butyryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- FATUQANACHZLRT-KMRXSBRUSA-L calcium glucoheptonate Chemical compound [Ca+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O FATUQANACHZLRT-KMRXSBRUSA-L 0.000 description 1
- MIOPJNTWMNEORI-UHFFFAOYSA-N camphorsulfonic acid Chemical compound C1CC2(CS(O)(=O)=O)C(=O)CC1C2(C)C MIOPJNTWMNEORI-UHFFFAOYSA-N 0.000 description 1
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 1
- 125000004623 carbolinyl group Chemical group 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-N carbonic acid monoamide Natural products NC(O)=O KXDHJXZQYSOELW-UHFFFAOYSA-N 0.000 description 1
- 150000003857 carboxamides Chemical class 0.000 description 1
- 239000012876 carrier material Substances 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 230000032823 cell division Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000008614 cellular interaction Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 229940082500 cetostearyl alcohol Drugs 0.000 description 1
- 239000013626 chemical specie Substances 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- VDANGULDQQJODZ-UHFFFAOYSA-N chloroprocaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1Cl VDANGULDQQJODZ-UHFFFAOYSA-N 0.000 description 1
- 229960002023 chloroprocaine Drugs 0.000 description 1
- 229960003677 chloroquine Drugs 0.000 description 1
- WHTVZRBIWZFKQO-UHFFFAOYSA-N chloroquine Natural products ClC1=CC=C2C(NC(C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-UHFFFAOYSA-N 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 125000003016 chromanyl group Chemical group O1C(CCC2=CC=CC=C12)* 0.000 description 1
- 125000004230 chromenyl group Chemical group O1C(C=CC2=CC=CC=C12)* 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000025302 chronic primary adrenal insufficiency Diseases 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 229950002649 ciprocinonide Drugs 0.000 description 1
- 229940001468 citrate Drugs 0.000 description 1
- 238000003501 co-culture Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 239000007859 condensation product Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- OPQARKPSCNTWTJ-UHFFFAOYSA-L copper(ii) acetate Chemical compound [Cu+2].CC([O-])=O.CC([O-])=O OPQARKPSCNTWTJ-UHFFFAOYSA-L 0.000 description 1
- 210000004087 cornea Anatomy 0.000 description 1
- 239000002537 cosmetic Substances 0.000 description 1
- 230000000139 costimulatory effect Effects 0.000 description 1
- 125000000332 coumarinyl group Chemical group O1C(=O)C(=CC2=CC=CC=C12)* 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000001047 cyclobutenyl group Chemical group C1(=CCC1)* 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 229940097362 cyclodextrins Drugs 0.000 description 1
- 125000002188 cycloheptatrienyl group Chemical group C1(=CC=CC=CC1)* 0.000 description 1
- 125000001162 cycloheptenyl group Chemical group C1(=CCCCCC1)* 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000003678 cyclohexadienyl group Chemical group C1(=CC=CCC1)* 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- NKLCHDQGUHMCGL-UHFFFAOYSA-N cyclohexylidenemethanone Chemical group O=C=C1CCCCC1 NKLCHDQGUHMCGL-UHFFFAOYSA-N 0.000 description 1
- 125000004210 cyclohexylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- HCAJEUSONLESMK-UHFFFAOYSA-N cyclohexylsulfamic acid Chemical class OS(=O)(=O)NC1CCCCC1 HCAJEUSONLESMK-UHFFFAOYSA-N 0.000 description 1
- 125000000522 cyclooctenyl group Chemical group C1(=CCCCCCC1)* 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 1
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000004851 cyclopentylmethyl group Chemical group C1(CCCC1)C* 0.000 description 1
- 229960004397 cyclophosphamide Drugs 0.000 description 1
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 1
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 1
- 229950008150 daltroban Drugs 0.000 description 1
- ZESRJSPZRDMNHY-UHFFFAOYSA-N de-oxy corticosterone Natural products O=C1CCC2(C)C3CCC(C)(C(CC4)C(=O)CO)C4C3CCC2=C1 ZESRJSPZRDMNHY-UHFFFAOYSA-N 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 230000002950 deficient Effects 0.000 description 1
- 238000000280 densification Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 229960003654 desoxycortone Drugs 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 229940039227 diagnostic agent Drugs 0.000 description 1
- 239000000032 diagnostic agent Substances 0.000 description 1
- 125000002576 diazepinyl group Chemical group N1N=C(C=CC=C1)* 0.000 description 1
- 125000005509 dibenzothiophenyl group Chemical group 0.000 description 1
- 125000004915 dibutylamino group Chemical group C(CCC)N(CCCC)* 0.000 description 1
- 150000001991 dicarboxylic acids Chemical class 0.000 description 1
- 125000003963 dichloro group Chemical group Cl* 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- ZBCBWPMODOFKDW-UHFFFAOYSA-N diethanolamine Chemical compound OCCNCCO ZBCBWPMODOFKDW-UHFFFAOYSA-N 0.000 description 1
- 229940043237 diethanolamine Drugs 0.000 description 1
- UQIFUBUFYITEAI-UHFFFAOYSA-N diethyl 2-[[2-(4-fluorophenyl)acetyl]amino]propanedioate Chemical compound CCOC(=O)C(C(=O)OCC)NC(=O)CC1=CC=C(F)C=C1 UQIFUBUFYITEAI-UHFFFAOYSA-N 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical group OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 125000005043 dihydropyranyl group Chemical group O1C(CCC=C1)* 0.000 description 1
- 125000004655 dihydropyridinyl group Chemical group N1(CC=CC=C1)* 0.000 description 1
- 125000005057 dihydrothienyl group Chemical group S1C(CC=C1)* 0.000 description 1
- 229950009815 dimepranol Drugs 0.000 description 1
- UIZVAEFPROTLLV-UHFFFAOYSA-N dimethyl 2-[(4-fluorobenzoyl)amino]propanedioate Chemical compound COC(=O)C(C(=O)OC)NC(=O)C1=CC=C(F)C=C1 UIZVAEFPROTLLV-UHFFFAOYSA-N 0.000 description 1
- CYPOXSVGVPUXKE-UHFFFAOYSA-N dimethyl 2-[3-(4-fluorophenoxy)propanoylamino]propanedioate Chemical compound COC(=O)C(C(=O)OC)NC(=O)CCOC1=CC=C(F)C=C1 CYPOXSVGVPUXKE-UHFFFAOYSA-N 0.000 description 1
- QGPLXCMTXOIWHO-UHFFFAOYSA-N dimethyl 2-[3-(4-fluorophenyl)propanoylamino]propanedioate Chemical compound COC(=O)C(C(=O)OC)NC(=O)CCC1=CC=C(F)C=C1 QGPLXCMTXOIWHO-UHFFFAOYSA-N 0.000 description 1
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 1
- 125000000597 dioxinyl group Chemical group 0.000 description 1
- 125000005879 dioxolanyl group Chemical group 0.000 description 1
- 239000001177 diphosphate Substances 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 1
- 235000011180 diphosphates Nutrition 0.000 description 1
- ZGSPNIOCEDOHGS-UHFFFAOYSA-L disodium [3-[2,3-di(octadeca-9,12-dienoyloxy)propoxy-oxidophosphoryl]oxy-2-hydroxypropyl] 2,3-di(octadeca-9,12-dienoyloxy)propyl phosphate Chemical compound [Na+].[Na+].CCCCCC=CCC=CCCCCCCCC(=O)OCC(OC(=O)CCCCCCCC=CCC=CCCCCC)COP([O-])(=O)OCC(O)COP([O-])(=O)OCC(OC(=O)CCCCCCCC=CCC=CCCCCC)COC(=O)CCCCCCCC=CCC=CCCCCC ZGSPNIOCEDOHGS-UHFFFAOYSA-L 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 125000005883 dithianyl group Chemical group 0.000 description 1
- 229950004822 ditiocarb sodium Drugs 0.000 description 1
- 239000000890 drug combination Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 229940009662 edetate Drugs 0.000 description 1
- 229960001484 edetic acid Drugs 0.000 description 1
- 238000002565 electrocardiography Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 229960000403 etanercept Drugs 0.000 description 1
- AFAXGSQYZLGZPG-UHFFFAOYSA-N ethanedisulfonic acid Chemical compound OS(=O)(=O)CCS(O)(=O)=O AFAXGSQYZLGZPG-UHFFFAOYSA-N 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- WCQOBLXWLRDEQA-UHFFFAOYSA-N ethanimidamide;hydrochloride Chemical compound Cl.CC(N)=N WCQOBLXWLRDEQA-UHFFFAOYSA-N 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000003754 ethoxycarbonyl group Chemical group C(=O)(OCC)* 0.000 description 1
- ZIUSEGSNTOUIPT-UHFFFAOYSA-N ethyl 2-cyanoacetate Chemical compound CCOC(=O)CC#N ZIUSEGSNTOUIPT-UHFFFAOYSA-N 0.000 description 1
- NMRAEHALICNNNO-UHFFFAOYSA-N ethyl 4-(3-chloro-4-fluoroanilino)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)OCC)=NC=1NC1=CC=C(F)C(Cl)=C1 NMRAEHALICNNNO-UHFFFAOYSA-N 0.000 description 1
- UWQHZCTYYIVMQR-UHFFFAOYSA-N ethyl 4-[4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl]-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate Chemical compound C=12C=C(C=3C=CC(F)=CC=3)SC2=NC(C(=O)OCC)=NC=1N(CC1)CCN1C(=O)COC1=CC=C(Cl)C=C1 UWQHZCTYYIVMQR-UHFFFAOYSA-N 0.000 description 1
- 125000004494 ethyl ester group Chemical group 0.000 description 1
- 229940093476 ethylene glycol Drugs 0.000 description 1
- 229940012017 ethylenediamine Drugs 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 230000003631 expected effect Effects 0.000 description 1
- 210000001508 eye Anatomy 0.000 description 1
- 210000000887 face Anatomy 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 229960002011 fludrocortisone Drugs 0.000 description 1
- AAXVEMMRQDVLJB-BULBTXNYSA-N fludrocortisone Chemical compound O=C1CC[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 AAXVEMMRQDVLJB-BULBTXNYSA-N 0.000 description 1
- 229950002998 flumoxonide Drugs 0.000 description 1
- 125000003914 fluoranthenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC=C4C1=C23)* 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 229940050411 fumarate Drugs 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 229960002963 ganciclovir Drugs 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 229960004198 guanidine Drugs 0.000 description 1
- 229960000789 guanidine hydrochloride Drugs 0.000 description 1
- PJJJBBJSCAKJQF-UHFFFAOYSA-N guanidinium chloride Chemical compound [Cl-].NC(N)=[NH2+] PJJJBBJSCAKJQF-UHFFFAOYSA-N 0.000 description 1
- 239000000665 guar gum Substances 0.000 description 1
- 235000010417 guar gum Nutrition 0.000 description 1
- 229960002154 guar gum Drugs 0.000 description 1
- IDINUJSAMVOPCM-UHFFFAOYSA-N gusperimus Chemical compound NCCCNCCCCNC(=O)C(O)NC(=O)CCCCCCN=C(N)N IDINUJSAMVOPCM-UHFFFAOYSA-N 0.000 description 1
- 229960002706 gusperimus Drugs 0.000 description 1
- 210000004209 hair Anatomy 0.000 description 1
- 210000002837 heart atrium Anatomy 0.000 description 1
- 210000003709 heart valve Anatomy 0.000 description 1
- 239000003481 heat shock protein 90 inhibitor Substances 0.000 description 1
- 210000002443 helper t lymphocyte Anatomy 0.000 description 1
- 208000007475 hemolytic anemia Diseases 0.000 description 1
- 230000002949 hemolytic effect Effects 0.000 description 1
- 231100000234 hepatic damage Toxicity 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 231100000304 hepatotoxicity Toxicity 0.000 description 1
- 230000007686 hepatotoxicity Effects 0.000 description 1
- UBHWBODXJBSFLH-UHFFFAOYSA-N hexadecan-1-ol;octadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO.CCCCCCCCCCCCCCCCCCO UBHWBODXJBSFLH-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- XGIHQYAWBCFNPY-AZOCGYLKSA-N hydrabamine Chemical compound C([C@@H]12)CC3=CC(C(C)C)=CC=C3[C@@]2(C)CCC[C@@]1(C)CNCCNC[C@@]1(C)[C@@H]2CCC3=CC(C(C)C)=CC=C3[C@@]2(C)CCC1 XGIHQYAWBCFNPY-AZOCGYLKSA-N 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 229960000890 hydrocortisone Drugs 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 150000004679 hydroxides Chemical class 0.000 description 1
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 description 1
- XXSMGPRMXLTPCZ-UHFFFAOYSA-N hydroxychloroquine Chemical compound ClC1=CC=C2C(NC(C)CCCN(CCO)CC)=CC=NC2=C1 XXSMGPRMXLTPCZ-UHFFFAOYSA-N 0.000 description 1
- 229960004171 hydroxychloroquine Drugs 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 230000003463 hyperproliferative effect Effects 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 125000002636 imidazolinyl group Chemical group 0.000 description 1
- DOUYETYNHWVLEO-UHFFFAOYSA-N imiquimod Chemical compound C1=CC=CC2=C3N(CC(C)C)C=NC3=C(N)N=C21 DOUYETYNHWVLEO-UHFFFAOYSA-N 0.000 description 1
- 229960002751 imiquimod Drugs 0.000 description 1
- 125000003427 indacenyl group Chemical group 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000003406 indolizinyl group Chemical group C=1(C=CN2C=CC=CC12)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000000411 inducer Substances 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 208000000509 infertility Diseases 0.000 description 1
- 230000036512 infertility Effects 0.000 description 1
- 231100000535 infertility Toxicity 0.000 description 1
- 230000008595 infiltration Effects 0.000 description 1
- 238000001764 infiltration Methods 0.000 description 1
- 208000021646 inflammation of heart layer Diseases 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 229960000598 infliximab Drugs 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 210000005007 innate immune system Anatomy 0.000 description 1
- 150000007529 inorganic bases Chemical class 0.000 description 1
- 239000001023 inorganic pigment Substances 0.000 description 1
- 229910001867 inorganic solvent Inorganic materials 0.000 description 1
- 239000003049 inorganic solvent Substances 0.000 description 1
- 229960000476 inosine pranobex Drugs 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 230000008611 intercellular interaction Effects 0.000 description 1
- 229960001388 interferon-beta Drugs 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 238000010253 intravenous injection Methods 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 238000005040 ion trap Methods 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 125000001977 isobenzofuranyl group Chemical group C=1(OC=C2C=CC=CC12)* 0.000 description 1
- 125000005929 isobutyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])OC(*)=O 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 125000005928 isopropyloxycarbonyl group Chemical group [H]C([H])([H])C([H])(OC(*)=O)C([H])([H])[H] 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 150000002540 isothiocyanates Chemical class 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 229940054136 kineret Drugs 0.000 description 1
- 230000002045 lasting effect Effects 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 229960000681 leflunomide Drugs 0.000 description 1
- VHOGYURTWQBHIL-UHFFFAOYSA-N leflunomide Chemical compound O1N=CC(C(=O)NC=2C=CC(=CC=2)C(F)(F)F)=C1C VHOGYURTWQBHIL-UHFFFAOYSA-N 0.000 description 1
- 229940115286 lentinan Drugs 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 230000008818 liver damage Effects 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 231100000053 low toxicity Toxicity 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 206010025135 lupus erythematosus Diseases 0.000 description 1
- 125000004401 m-toluyl group Chemical group [H]C1=C([H])C(=C([H])C(=C1[H])C([H])([H])[H])C(*)=O 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 159000000003 magnesium salts Chemical class 0.000 description 1
- 201000004792 malaria Diseases 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-M mandelate Chemical compound [O-]C(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-M 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 230000006371 metabolic abnormality Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- CYEBJEDOHLIWNP-UHFFFAOYSA-N methanethioamide Chemical compound NC=S CYEBJEDOHLIWNP-UHFFFAOYSA-N 0.000 description 1
- WSFSSNUMVMOOMR-BJUDXGSMSA-N methanone Chemical compound O=[11CH2] WSFSSNUMVMOOMR-BJUDXGSMSA-N 0.000 description 1
- 125000001160 methoxycarbonyl group Chemical group [H]C([H])([H])OC(*)=O 0.000 description 1
- PLAHRPLOQQVVQU-UHFFFAOYSA-N methyl 4-[2-[4-[5-amino-2-[2-(4-fluorophenyl)ethyl]-[1,3]thiazolo[5,4-d]pyrimidin-7-yl]piperazin-1-yl]-2-oxoethoxy]benzoate Chemical compound C1=CC(C(=O)OC)=CC=C1OCC(=O)N1CCN(C=2C=3N=C(CCC=4C=CC(F)=CC=4)SC=3N=C(N)N=2)CC1 PLAHRPLOQQVVQU-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 150000004702 methyl esters Chemical group 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- JZMJDSHXVKJFKW-UHFFFAOYSA-M methyl sulfate(1-) Chemical compound COS([O-])(=O)=O JZMJDSHXVKJFKW-UHFFFAOYSA-M 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 229960002900 methylcellulose Drugs 0.000 description 1
- 229960002216 methylparaben Drugs 0.000 description 1
- 229960004584 methylprednisolone Drugs 0.000 description 1
- 125000006216 methylsulfinyl group Chemical group [H]C([H])([H])S(*)=O 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 1
- 239000004530 micro-emulsion Substances 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 244000309715 mini pig Species 0.000 description 1
- 230000011278 mitosis Effects 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 229950000844 mizoribine Drugs 0.000 description 1
- 229950007856 mofetil Drugs 0.000 description 1
- 150000002762 monocarboxylic acid derivatives Chemical class 0.000 description 1
- 210000005087 mononuclear cell Anatomy 0.000 description 1
- 229910052901 montmorillonite Inorganic materials 0.000 description 1
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 231100000150 mutagenicity / genotoxicity testing Toxicity 0.000 description 1
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 1
- 229960000951 mycophenolic acid Drugs 0.000 description 1
- 125000001421 myristyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- HLFMAANNMCCSMF-UHFFFAOYSA-N n-(2,4-difluorophenyl)piperazine-1-carboxamide Chemical compound FC1=CC(F)=CC=C1NC(=O)N1CCNCC1 HLFMAANNMCCSMF-UHFFFAOYSA-N 0.000 description 1
- SOJGXALKMGXYQZ-UHFFFAOYSA-N n-(2-amino-4-hydroxy-6-oxo-1h-pyrimidin-5-yl)-2-(4-fluorophenyl)acetamide Chemical compound OC1=NC(N)=NC(O)=C1NC(=O)CC1=CC=C(F)C=C1 SOJGXALKMGXYQZ-UHFFFAOYSA-N 0.000 description 1
- XOUGSZPIIGXJCW-UHFFFAOYSA-N n-(2-amino-4-hydroxy-6-oxo-1h-pyrimidin-5-yl)-3-(4-fluorophenyl)propanamide Chemical compound OC1=NC(N)=NC(O)=C1NC(=O)CCC1=CC=C(F)C=C1 XOUGSZPIIGXJCW-UHFFFAOYSA-N 0.000 description 1
- KNHZEKUNXNECCN-UHFFFAOYSA-N n-(2-methoxyphenyl)piperazine-1-carboxamide Chemical compound COC1=CC=CC=C1NC(=O)N1CCNCC1 KNHZEKUNXNECCN-UHFFFAOYSA-N 0.000 description 1
- VNZQVXFQDXQRSA-UHFFFAOYSA-N n-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine Chemical compound C1=CC(F)=CC=C1C(SC1=NC=N2)=CC1=C2NC1=CC=C(F)C(Cl)=C1 VNZQVXFQDXQRSA-UHFFFAOYSA-N 0.000 description 1
- HRFZUFHAHQHMAY-UHFFFAOYSA-N n-(4-bromophenyl)piperazine-1-carboxamide Chemical compound C1=CC(Br)=CC=C1NC(=O)N1CCNCC1 HRFZUFHAHQHMAY-UHFFFAOYSA-N 0.000 description 1
- MQLRGWUBLRQSOU-QGZVFWFLSA-N n-[(3r)-1-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)pyrrolidin-3-yl]-2-(4-chlorophenoxy)acetamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(=CC2=1)C=1C=CC=CC=1)N)C(=O)COC1=CC=C(Cl)C=C1 MQLRGWUBLRQSOU-QGZVFWFLSA-N 0.000 description 1
- UMZNPYAAVCVJAB-QGZVFWFLSA-N n-[(3r)-1-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)pyrrolidin-3-yl]-4-chlorobenzamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(=CC2=1)C=1C=CC=CC=1)N)C(=O)C1=CC=C(Cl)C=C1 UMZNPYAAVCVJAB-QGZVFWFLSA-N 0.000 description 1
- RTUCLZBCPNWBHS-QGZVFWFLSA-N n-[(3r)-1-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]pyrrolidin-3-yl]-2-(4-chlorophenoxy)acetamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(=CC2=1)C=1C=CC(F)=CC=1)N)C(=O)COC1=CC=C(Cl)C=C1 RTUCLZBCPNWBHS-QGZVFWFLSA-N 0.000 description 1
- NPJZXUOYPBOCIZ-QGZVFWFLSA-N n-[(3r)-1-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]pyrrolidin-3-yl]-4-chlorobenzamide Chemical compound N([C@@H]1CCN(C1)C=1N=C(N=C2SC(=CC2=1)C=1C=CC(F)=CC=1)N)C(=O)C1=CC=C(Cl)C=C1 NPJZXUOYPBOCIZ-QGZVFWFLSA-N 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229950005486 naflocort Drugs 0.000 description 1
- 239000002088 nanocapsule Substances 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical compound C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- UFWIBTONFRDIAS-UHFFFAOYSA-N naphthalene-acid Natural products C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 229920001206 natural gum Polymers 0.000 description 1
- 230000007694 nephrotoxicity Effects 0.000 description 1
- 231100000417 nephrotoxicity Toxicity 0.000 description 1
- 230000000926 neurological effect Effects 0.000 description 1
- 230000007135 neurotoxicity Effects 0.000 description 1
- 231100000228 neurotoxicity Toxicity 0.000 description 1
- 239000012299 nitrogen atmosphere Substances 0.000 description 1
- 150000002832 nitroso derivatives Chemical class 0.000 description 1
- 125000002868 norbornyl group Chemical group C12(CCC(CC1)C2)* 0.000 description 1
- 239000002777 nucleoside Substances 0.000 description 1
- 125000003835 nucleoside group Chemical group 0.000 description 1
- 125000005441 o-toluyl group Chemical group [H]C1=C([H])C(C(*)=O)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Chemical class CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- UYDLBVPAAFVANX-UHFFFAOYSA-N octylphenoxy polyethoxyethanol Chemical compound CC(C)(C)CC(C)(C)C1=CC=C(OCCOCCOCCOCCO)C=C1 UYDLBVPAAFVANX-UHFFFAOYSA-N 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 150000002888 oleic acid derivatives Chemical class 0.000 description 1
- 125000001117 oleyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])/C([H])=C([H])\C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000010915 one-step procedure Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000007530 organic bases Chemical class 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 239000012860 organic pigment Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 150000002905 orthoesters Chemical class 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000000160 oxazolidinyl group Chemical group 0.000 description 1
- 125000005968 oxazolinyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000003566 oxetanyl group Chemical group 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 238000007243 oxidative cyclization reaction Methods 0.000 description 1
- 125000000466 oxiranyl group Chemical group 0.000 description 1
- TWNQGVIAIRXVLR-UHFFFAOYSA-N oxo(oxoalumanyloxy)alumane Chemical compound O=[Al]O[Al]=O TWNQGVIAIRXVLR-UHFFFAOYSA-N 0.000 description 1
- 125000000352 p-cymenyl group Chemical group C1(=C(C=C(C=C1)C)*)C(C)C 0.000 description 1
- 125000005440 p-toluyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C(*)=O)C([H])([H])[H] 0.000 description 1
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- 229940014662 pantothenate Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 235000019371 penicillin G benzathine Nutrition 0.000 description 1
- 210000003899 penis Anatomy 0.000 description 1
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 1
- 125000005327 perimidinyl group Chemical group N1C(=NC2=CC=CC3=CC=CC1=C23)* 0.000 description 1
- 229940124531 pharmaceutical excipient Drugs 0.000 description 1
- 125000004625 phenanthrolinyl group Chemical group N1=C(C=CC2=CC=C3C=CC=NC3=C12)* 0.000 description 1
- 125000001791 phenazinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3N=C12)* 0.000 description 1
- 229960003742 phenol Drugs 0.000 description 1
- 125000005954 phenoxathiinyl group Chemical group 0.000 description 1
- 125000001644 phenoxazinyl group Chemical group C1(=CC=CC=2OC3=CC=CC=C3NC12)* 0.000 description 1
- 125000006678 phenoxycarbonyl group Chemical group 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 1
- 125000004344 phenylpropyl group Chemical group 0.000 description 1
- 125000003170 phenylsulfonyl group Chemical group C1(=CC=CC=C1)S(=O)(=O)* 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 239000002587 phosphodiesterase IV inhibitor Substances 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical group [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 150000003014 phosphoric acid esters Chemical class 0.000 description 1
- 229910052698 phosphorus Chemical group 0.000 description 1
- 239000011574 phosphorus Chemical group 0.000 description 1
- UHZYTMXLRWXGPK-UHFFFAOYSA-N phosphorus pentachloride Chemical compound ClP(Cl)(Cl)(Cl)Cl UHZYTMXLRWXGPK-UHFFFAOYSA-N 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- 125000002265 phtalazinyl group Chemical group 0.000 description 1
- 125000001388 picenyl group Chemical group C1(=CC=CC2=CC=C3C4=CC=C5C=CC=CC5=C4C=CC3=C21)* 0.000 description 1
- 229960001163 pidotimod Drugs 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 239000003880 polar aprotic solvent Substances 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920003229 poly(methyl methacrylate) Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000151 polyglycol Polymers 0.000 description 1
- 239000010695 polyglycol Substances 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 239000004926 polymethyl methacrylate Substances 0.000 description 1
- 208000005987 polymyositis Diseases 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 108020001213 potassium channel Proteins 0.000 description 1
- 159000000001 potassium salts Chemical class 0.000 description 1
- 235000010241 potassium sorbate Nutrition 0.000 description 1
- 239000004302 potassium sorbate Substances 0.000 description 1
- 229940069338 potassium sorbate Drugs 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 125000001844 prenyl group Chemical group [H]C([*])([H])C([H])=C(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 229950000504 procinonide Drugs 0.000 description 1
- XYWJNTOURDMTPI-UHFFFAOYSA-N procodazole Chemical compound C1=CC=C2NC(CCC(=O)O)=NC2=C1 XYWJNTOURDMTPI-UHFFFAOYSA-N 0.000 description 1
- 229950000989 procodazole Drugs 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- XEABSBMNTNXEJM-UHFFFAOYSA-N propagermanium Chemical compound OC(=O)CC[Ge](=O)O[Ge](=O)CCC(O)=O XEABSBMNTNXEJM-UHFFFAOYSA-N 0.000 description 1
- 229950002828 propagermanium Drugs 0.000 description 1
- ULWHHBHJGPPBCO-UHFFFAOYSA-N propane-1,1-diol Chemical compound CCC(O)O ULWHHBHJGPPBCO-UHFFFAOYSA-N 0.000 description 1
- 125000001501 propionyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000010388 propyl gallate Nutrition 0.000 description 1
- 239000000473 propyl gallate Substances 0.000 description 1
- 229940075579 propyl gallate Drugs 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 229960004063 propylene glycol Drugs 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 125000004742 propyloxycarbonyl group Chemical group 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 229950008679 protamine sulfate Drugs 0.000 description 1
- 239000011253 protective coating Substances 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 125000001042 pteridinyl group Chemical group N1=C(N=CC2=NC=CN=C12)* 0.000 description 1
- 239000002212 purine nucleoside Substances 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000002755 pyrazolinyl group Chemical group 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- CMMSUVYMWKBPQK-UHFFFAOYSA-N pyrido[3,2-d]pyrimidine-2,4-diamine Chemical class N1=CC=CC2=NC(N)=NC(N)=C21 CMMSUVYMWKBPQK-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000005344 pyridylmethyl group Chemical group [H]C1=C([H])C([H])=C([H])C(=N1)C([H])([H])* 0.000 description 1
- FVLAYJRLBLHIPV-UHFFFAOYSA-N pyrimidin-5-amine Chemical compound NC1=CN=CN=C1 FVLAYJRLBLHIPV-UHFFFAOYSA-N 0.000 description 1
- 125000001422 pyrrolinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 125000004621 quinuclidinyl group Chemical group N12C(CC(CC1)CC2)* 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 238000006476 reductive cyclization reaction Methods 0.000 description 1
- 230000004043 responsiveness Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000006798 ring closing metathesis reaction Methods 0.000 description 1
- 239000003419 rna directed dna polymerase inhibitor Substances 0.000 description 1
- 229950003733 romurtide Drugs 0.000 description 1
- 108700033545 romurtide Proteins 0.000 description 1
- LNGNTCFORRWFSF-DVTGEIKXSA-N s-methyl (8s,9r,10s,11s,13s,14s,16s,17r)-9-fluoro-11,17-dihydroxy-10,13,16-trimethyl-3-oxo-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthrene-17-carbothioate Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)SC)(O)[C@@]1(C)C[C@@H]2O LNGNTCFORRWFSF-DVTGEIKXSA-N 0.000 description 1
- 229960001860 salicylate Drugs 0.000 description 1
- 150000004671 saturated fatty acids Chemical class 0.000 description 1
- 235000003441 saturated fatty acids Nutrition 0.000 description 1
- HOZOZZFCZRXYEK-HNHWXVNLSA-M scopolamine butylbromide Chemical compound [Br-].C1([C@@H](CO)C(=O)OC2C[C@@H]3[N+]([C@H](C2)[C@@H]2[C@H]3O2)(C)CCCC)=CC=CC=C1 HOZOZZFCZRXYEK-HNHWXVNLSA-M 0.000 description 1
- 210000005212 secondary lymphoid organ Anatomy 0.000 description 1
- 230000001624 sedative effect Effects 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Chemical group 0.000 description 1
- JPJALAQPGMAKDF-UHFFFAOYSA-N selenium dioxide Chemical group O=[Se]=O JPJALAQPGMAKDF-UHFFFAOYSA-N 0.000 description 1
- 239000003352 sequestering agent Substances 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 210000003491 skin Anatomy 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 229960003885 sodium benzoate Drugs 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 1
- JVBXVOWTABLYPX-UHFFFAOYSA-L sodium dithionite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])=O JVBXVOWTABLYPX-UHFFFAOYSA-L 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- 235000010267 sodium hydrogen sulphite Nutrition 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 229940001584 sodium metabisulfite Drugs 0.000 description 1
- 235000010262 sodium metabisulphite Nutrition 0.000 description 1
- 235000010288 sodium nitrite Nutrition 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 229940079101 sodium sulfide Drugs 0.000 description 1
- 229910052979 sodium sulfide Inorganic materials 0.000 description 1
- ZGHLCBJZQLNUAZ-UHFFFAOYSA-N sodium sulfide nonahydrate Chemical compound O.O.O.O.O.O.O.O.O.[Na+].[Na+].[S-2] ZGHLCBJZQLNUAZ-UHFFFAOYSA-N 0.000 description 1
- JJICLMJFIKGAAU-UHFFFAOYSA-M sodium;2-amino-9-(1,3-dihydroxypropan-2-yloxymethyl)purin-6-olate Chemical compound [Na+].NC1=NC([O-])=C2N=CN(COC(CO)CO)C2=N1 JJICLMJFIKGAAU-UHFFFAOYSA-M 0.000 description 1
- RMLUKZWYIKEASN-UHFFFAOYSA-M sodium;2-amino-9-(2-hydroxyethoxymethyl)purin-6-olate Chemical compound [Na+].O=C1[N-]C(N)=NC2=C1N=CN2COCCO RMLUKZWYIKEASN-UHFFFAOYSA-M 0.000 description 1
- WBQTXTBONIWRGK-UHFFFAOYSA-N sodium;propan-2-olate Chemical compound [Na+].CC(C)[O-] WBQTXTBONIWRGK-UHFFFAOYSA-N 0.000 description 1
- 235000019337 sorbitan trioleate Nutrition 0.000 description 1
- 229960000391 sorbitan trioleate Drugs 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000001694 spray drying Methods 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 238000011272 standard treatment Methods 0.000 description 1
- 239000008117 stearic acid Chemical class 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 125000005504 styryl group Chemical group 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 125000003375 sulfoxide group Chemical group 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 230000020382 suppression by virus of host antigen processing and presentation of peptide antigen via MHC class I Effects 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 229920001059 synthetic polymer Polymers 0.000 description 1
- 229940065721 systemic for obstructive airway disease xanthines Drugs 0.000 description 1
- 229940037128 systemic glucocorticoids Drugs 0.000 description 1
- 201000000596 systemic lupus erythematosus Diseases 0.000 description 1
- 239000003760 tallow Substances 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- 231100000211 teratogenicity Toxicity 0.000 description 1
- UTNUDOFZCWSZMS-YFHOEESVSA-N teriflunomide Chemical compound C\C(O)=C(/C#N)C(=O)NC1=CC=C(C(F)(F)F)C=C1 UTNUDOFZCWSZMS-YFHOEESVSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- RNYWAUFNEDZFSY-CQSZACIVSA-N tert-butyl n-[(3r)-1-[2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl]pyrrolidin-3-yl]carbamate Chemical compound C1[C@H](NC(=O)OC(C)(C)C)CCN1C1=NC(N)=NC2=C1C=C(C=1C=CC(F)=CC=1)S2 RNYWAUFNEDZFSY-CQSZACIVSA-N 0.000 description 1
- 150000005621 tetraalkylammonium salts Chemical class 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical compound C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- OULAJFUGPPVRBK-UHFFFAOYSA-N tetratriacontyl alcohol Natural products CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO OULAJFUGPPVRBK-UHFFFAOYSA-N 0.000 description 1
- 125000005247 tetrazinyl group Chemical group N1=NN=NC(=C1)* 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 229960000278 theophylline Drugs 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 125000004525 thiadiazinyl group Chemical group S1NN=C(C=C1)* 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000004627 thianthrenyl group Chemical group C1(=CC=CC=2SC3=CC=CC=C3SC12)* 0.000 description 1
- 125000006407 thiazinanyl group Chemical group 0.000 description 1
- 125000001984 thiazolidinyl group Chemical group 0.000 description 1
- 125000002769 thiazolinyl group Chemical group 0.000 description 1
- 230000008719 thickening Effects 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000002053 thietanyl group Chemical group 0.000 description 1
- 125000001730 thiiranyl group Chemical group 0.000 description 1
- 125000004862 thiobutyl group Chemical group 0.000 description 1
- 125000000101 thioether group Chemical group 0.000 description 1
- 125000004014 thioethyl group Chemical group [H]SC([H])([H])C([H])([H])* 0.000 description 1
- 125000004035 thiopropyl group Chemical group [H]SC([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000005503 thioxanyl group Chemical group 0.000 description 1
- 229940027755 thymomodulin Drugs 0.000 description 1
- PSWFFKRAVBDQEG-YGQNSOCVSA-N thymopentin Chemical compound NC(N)=NCCC[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=C(O)C=C1 PSWFFKRAVBDQEG-YGQNSOCVSA-N 0.000 description 1
- 229960004517 thymopentin Drugs 0.000 description 1
- 206010043778 thyroiditis Diseases 0.000 description 1
- 229950008689 timobesone Drugs 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- 229950001669 tipredane Drugs 0.000 description 1
- KJAMZCVTJDTESW-UHFFFAOYSA-N tiracizine Chemical compound C1CC2=CC=CC=C2N(C(=O)CN(C)C)C2=CC(NC(=O)OCC)=CC=C21 KJAMZCVTJDTESW-UHFFFAOYSA-N 0.000 description 1
- 230000019432 tissue death Effects 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 125000005147 toluenesulfonyl group Chemical group C=1(C(=CC=CC1)S(=O)(=O)*)C 0.000 description 1
- 125000005425 toluyl group Chemical group 0.000 description 1
- 231100000027 toxicology Toxicity 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 238000013271 transdermal drug delivery Methods 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 238000000844 transformation Methods 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 229960005294 triamcinolone Drugs 0.000 description 1
- GFNANZIMVAIWHM-OBYCQNJPSA-N triamcinolone Chemical compound O=C1C=C[C@]2(C)[C@@]3(F)[C@@H](O)C[C@](C)([C@@]([C@H](O)C4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 GFNANZIMVAIWHM-OBYCQNJPSA-N 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 150000003628 tricarboxylic acids Chemical class 0.000 description 1
- GKASDNZWUGIAMG-UHFFFAOYSA-N triethyl orthoformate Chemical compound CCOC(OCC)OCC GKASDNZWUGIAMG-UHFFFAOYSA-N 0.000 description 1
- 125000004205 trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 230000007306 turnover Effects 0.000 description 1
- 229950009811 ubenimex Drugs 0.000 description 1
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 1
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 1
- 230000003827 upregulation Effects 0.000 description 1
- 125000003774 valeryl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- MWOOGOJBHIARFG-UHFFFAOYSA-N vanillin Chemical compound COC1=CC(C=O)=CC=C1O MWOOGOJBHIARFG-UHFFFAOYSA-N 0.000 description 1
- FGQOOHJZONJGDT-UHFFFAOYSA-N vanillin Natural products COC1=CC(O)=CC(C=O)=C1 FGQOOHJZONJGDT-UHFFFAOYSA-N 0.000 description 1
- 235000012141 vanillin Nutrition 0.000 description 1
- 230000002792 vascular Effects 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 230000009385 viral infection Effects 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 229920001285 xanthan gum Polymers 0.000 description 1
- 125000001834 xanthenyl group Chemical group C1=CC=CC=2OC3=CC=CC=C3C(C12)* 0.000 description 1
- 239000003064 xanthine oxidase inhibitor Substances 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/26—Heterocyclic compounds containing purine ring systems with an oxygen, sulphur, or nitrogen atom directly attached in position 2 or 6, but not in both
- C07D473/32—Nitrogen atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/517—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with carbocyclic ring systems, e.g. quinazoline, perimidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D473/00—Heterocyclic compounds containing purine ring systems
- C07D473/02—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6
- C07D473/18—Heterocyclic compounds containing purine ring systems with oxygen, sulphur, or nitrogen atoms directly attached in positions 2 and 6 one oxygen and one nitrogen atom, e.g. guanine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D513/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00
- C07D513/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for in groups C07D463/00, C07D477/00 or C07D499/00 - C07D507/00 in which the condensed system contains two hetero rings
- C07D513/04—Ortho-condensed systems
Definitions
- the present invention relates to a class of novel thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives and a method for their preparation, as well as to pharmaceutical compositions comprising one or more of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives and one or more pharmaceutically acceptable excipients.
- the present invention further relates to the use of said novel thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives as biologically active ingredients, more specifically as medicaments for the treatment of disorders and pathologic conditions such as, but not limited to, immune and auto-immune disorders, organ and cells transplant rejections.
- Thiazolo[5,4-d]pyrimidines and oxazolo[5,4-d]pyrimidines can be considered as structural analogues of purines, in which the imidazole moiety is replaced by a 1,3-thiazole or 1,3-oxazole ring system.
- purine chemistry is extensively described in literature, the number of medicinal chemistry papers that describe the synthesis and biological evaluation of oxazolopyrimidines and thiazolopyrimidines is limited. Apparently, the oxazolopyrimidine and thiazolopyrimidine scaffold is not very frequently used in drug discovery programs.
- Thiazolo[5,4-d]pyrimidines were covered by several patent applications as activators of caspases and inducers of apoptosis ( WO2008/057402 ), anti-angiogenic agents ( WO2004/01314 ), growth factor receptor inhibitors ( EP1731523 ), heat shock protein 90 inhibitors ( WO2008/059368 ) and xanthine oxidase inhibitors ( WO2007/004688 ).
- WO2008/152390 discloses thiazolo[5,4-d]pyrimidines and their use as inhibitors of phosphatidylinositol-3 kinase.
- WO 2008/005303 discloses vanilloid receptor 1 (TRPV1) modulating thiazolo[5,4-d]pyrimidine analogues and their use for the treatment of diseases, such as pain, arthritis, itch, cough, asthma, or inflammatory bowel disease.
- TRPV1 vanilloid receptor 1
- 2-Aryloxazolo[5,4-d]pyrimidines have been described as adenosine kinase inhibitors ( M. Bauser; et al. Bioorg. Med. Chem. Lett. 2004, 14, 1997-2000 ). 7-Amino-5-phenylethylamino-2-furyl-oxazolo[5,4-d]pyrimidines act as brain A 2A adenosine receptor (A 2A AR) antagonists ( M. H. Holschbach, et al. Eur. J. Med. Chem. 2006, 41, 7-15 ).
- WO 2007/102679 discloses thienopyrimidines with at position 4 a pyrrole-2,5-dione substituent which strongly inhibits IKB kinase- ⁇ (IKK- ⁇ ) involved in the activation of a transcriptional factor, NF- ⁇ B, which is associated with inducing various immune and inflammatory diseases, whereby a composition comprising the compound is a useful therapeutic agent against inflammatory diseases, in particular, arthritis and cancer.
- IKK- ⁇ IKB kinase- ⁇
- WO 2007/084815 discloses 2-carboxamide substituted thieno(2,3-d)pyrimidines inhibitors of one or more of the EGFR, HER-2, c-Src, Lyn, c-Abl, Aurora-A or VEGF kinase proteins and the like possessing anti-tumor cell proliferation activity, and as such are useful in treating or ameliorating a EGFR, HER-2, c-Src, Lyn, c-Abl, Aurora-A or VEGF kinase receptor mediated, angiogenesis-mediated or hyperproliferative disorder.
- WO 2006/071988 discloses certain 4,5-disubstituted thienopyrimidine derivatives which are useful for the inhibition of PDE10 enzymes, and thus are useful for treating psychiatric or neurological syndromes, such as psychoses, obsessive-compulsive disorder and/or Parkinson's disease.
- WO 2004/111057 discloses compounds which are particularly useful for inhibiting potassium channels Kvl. 5), which are known targets for the treatment of cardiac arrhythmia in the atria such as atrial fibrillation.
- none of these documents teaches or suggests thieno(2,3-d)pyrimidine derivatives having the substitution pattern disclosed by the present invention.
- Anti-cancer drugs with a purine based skeleton are known. Examples include theophylline (drug for the treatment of asthma) and azathioprine (drug for the treatment of transplant rejection).
- Anti-cancer drugs with a purine scaffold include 6-mercaptoguanine and thioguanine.
- Purines are also an important constituent of antiviral nucleosides such as acyclovir (used for the treatment of herpes virus infections) and ganciclovir (medication used for treatment of cytomegalovirus infections).
- Abacavir and dideoxyadenosine (ddA) are both purine nucleosides acting as reverse transcriptase inhibitors and both compounds are licensed as anti-HIV agents.
- WO 2009/005687 discloses purine derivatives and their use as modulators of Toll-like receptor 7. Compounds and pharmaceutical compositions which selectively activate toll-like receptor 7 are useful for treating viral infections in patients.
- WO 2008/135232 relates to substituted purines and purine derivatives as inhibitors of Aurora A, Aurora B, Aurora C, CHK2, JNK1 ⁇ 1, JNK3 and abl kinase. These compounds possess antiproliferative properties and are useful in the treatment of proliferative disorders such as cancer, leukemia, psoriasis and the like.
- WO 2008/094737 discloses purine derivatives as inhibitors of calcium dependent protein kinase 1 (PfCDPKI). These purines are useful for treating malaria.
- WO 2008/090181 relates to a new series of purine derivatives as inhibitors of Janus kinases.
- JAK3 kinase inhibitors have been recognized as a new class of effective immunosuppressive agents useful for transplant rejection prevention and in the prevention or treatment of immune, autoimmune, inflammatory and proliferative diseases such as psoriasis, psoriatic arthritis, rheumatoid arthritis, multiple sclerosis, inflammatory bowel diseases, systemic lupus erythematosus, type I diabetes and complications from diabetes, allergic reactions and leukemia.
- WO 2008/060301 also discloses 7-substituted purine derivatives as immunosuppressive drugs useful for treatment of an autoimmune disease, an inflammatory disease, a mast cell mediated disease, hematological malignancy and organ transplant rejection.
- purine derivatives having the substitution pattern disclosed by the present invention.
- immunosuppressive drugs include antiproliferative agents, such as methotrexate (a 2,4-diaminopyrido(3,2-d)pyrimidine derivative disclosed by U.S. Patent No. 2,512,572 ), azathioprine, and cyclophosphamide. Since these drugs affect mitosis and cell division, they have severe toxic effects on normal cells with high turn-over rate such as bone marrow cells and the gastrointestinal tract lining. Accordingly, marrow depression and liver damage are common side effects of these antiproliferative drugs.
- Anti-inflammatory compounds used to induce immunosuppression include adrenocortical steroids such as dexamethasone and prednisolone.
- adrenocortical steroids such as dexamethasone and prednisolone.
- dexamethasone and prednisolone.
- the common side effects observed with the use of these compounds are frequent infections, abnormal metabolism, hypertension, and diabetes.
- cyclosporine Other immunosuppressive compounds currently used to inhibit lymphocyte activation and subsequent proliferation include cyclosporine, tacrolimus and rapamycin. Cyclosporine and its relatives are among the most commonly used immunosuppressant drugs. Cyclosporine is typically used for preventing or treating organ rejection in kidney, liver, heart, pancreas, bone marrow, and heart-lung transplants, as well as for the treatment of autoimmune and inflammatory diseases such as Crohn's disease, aplastic anemia, multiple-sclerosis, myasthenia gravis, uveitis, biliary cirrhosis, etc. However, cyclosporines suffer from a small therapeutic dose window and severe toxic effects including nephrotoxicity, hepatotoxicity, hypertension, hirsutism, cancer, and neurotoxicity.
- monoclonal antibodies with immunosuppressant properties have been used to prevent and/or treat graft rejection.
- organ transplantation is considered a standard treatment and, in many cases, the only alternative to death.
- the immune response to foreign cell surface antigens on the graft encoded by the major histo-compatibility complex (hereinafter referred as MHC) and present on all cells, generally precludes successful transplantation of tissues and organs unless the transplant tissues come from a compatible donor and the normal immune response is suppressed.
- MHC major histo-compatibility complex
- the host response to an organ allograft involves a complex series of cellular interactions among T and B lymphocytes as well as macrophages or dendritic cells that recognize and are activated by foreign antigen.
- Co-stimulatory factors primarily cytokines, and specific cell-cell interactions, provided by activated accessory cells such as macrophages or dendritic cells are essential for T-cell proliferation.
- These macrophages and dendritic cells either directly adhere to T-cells through specific adhesion proteins or secrete cytokines that stimulate T-cells, such as IL-12 and IL-15.
- Accessory cell-derived co-stimulatory signals stimulate activation of interleukin-2 (IL- 2) gene transcription and expression of high affinity IL-2 receptors in T-cells.
- IL- 2 interleukin-2
- IL-2 is secreted by T lymphocytes upon antigen stimulation and is required for normal immune responsiveness. IL-2 stimulates lymphoid cells to proliferate and differentiate by binding to IL-2 specific cell surface receptors (IL-2R). IL-2 also initiates helper T-cell activation of cytotoxic T-cells and stimulates secretion of interferon- ⁇ which in turn activates cytodestructive properties of macrophages. Furthermore, IFN- ⁇ and IL-4 are also important activators of MHC class II expression in the transplanted organ, thereby further expanding the rejection cascade by enhancing the immunogenicity of the grafted organ.
- IL-2R IL-2 specific cell surface receptors
- IFN- ⁇ and IL-4 are also important activators of MHC class II expression in the transplanted organ, thereby further expanding the rejection cascade by enhancing the immunogenicity of the grafted organ.
- T-cells are primed in the T-cell zone of secondary lymphoid organs, primarily by dendritic cells.
- the initial interaction requires cell to cell contact between antigen-loaded MHC molecules on antigen-presenting cells (hereinafter referred as APC) and the T-cell receptor/CD3 complex on T-cells.
- APC antigen-presenting cells
- T-cell receptor/CD3 complex on T-cells.
- Engagement of the TCR/CD3 complex induces CD154 expression predominantly on CD4 T-cells that in turn activate the APC through CD40 engagement, leading to improved antigen presentation. This is caused partly by upregulation of CD80 and CD86 expression on the APC, both of which are ligands for the important CD28 co-stimulatory molecule on T-cells.
- CD40 engagement leads to prolonged surface expression of MHC-antigen complexes, expression of ligands for 4-1 BB and OX-40 (potent co-stimulatory molecules expressed on activated T-cells). Furthermore, CD40 engagement leads to secretion of various cytokines (e.g., IL-12, IL-15, TNF- ⁇ , IL-1, IL-6, and IL-8) and chemokines, all of which have important effects on both APC and T-cell activation and maturation. Similar mechanisms are involved in the development of auto-immune disease, such as type I diabetes.
- insulin- dependent diabetes mellitus results from a spontaneous T-cell dependent autoimmune destruction of insulin-producing pancreatic beta, cells that intensifies with age. The process is preceded by infiltration of the islets with mononuclear cells (insulitis), primarily composed of T lymphocytes. A delicate balance between auto-aggressive T-cells and suppressor-type immune phenomena determines whether expression of auto-immunity is limited to insulitis or not. Therapeutic strategies that target T-cells have been successful in preventing further progress of the autoimmune disease.
- the present invention is based on the unexpected finding that certain combinations of substituents at different positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine ring system, said combinations not being suggested by the prior art, are able to meet one or more of the medical needs recited herein above and to show unexpected biological properties, in particular have significant immunosuppressive activity.
- the present invention concerns a compound of I, II, III or IV : wherein
- the present invention also concerns a compound having formula I, II, III or IV, for use as a medicine.
- the present invention also concerns a compound having formula I, II, III or IV, for use as a medicine for the prevention or treatment of immune disorders in an animal, preferably in a mammal.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said mammal is a human being.
- the present invention also concerns a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound having formula I, II, III or IV, and one or more pharmaceutically acceptable exipients.
- Said composition may further comprise one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- the present invention also concerns a method of prevention or treatment of an immune disorder in an animal, comprising the administration of a therapeutically effective amount of a compound having formula I, II, III or IV, optionally in combination with one or more pharmaceutically acceptable excipients.
- said acylation is performed with a carboxylic acid (R 2 COOH) or an acid chloride (R 2 C(O)Cl).
- step (a) further comprises the addition of a coupling reagent.
- said coupling reagent is N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI).
- step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt).
- step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent.
- said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene.
- step (c) is performed in alkaline conditions.
- said oxidating agent is m -chloro-peroxybenzoic acid or hydrogen peroxide.
- step (e) a piperazine is introduced at position 7.
- the thiazolo(5,4-d)pyrimidine derivatives of formula I are R 2 -substituted 5-amino-7- N -piperazino thiazolo(5,4-d)pyrimidine derivatives.
- R 3 and R 2 have any of the values as described herein and R 1 is selected from the group comprising aryl, amino, acetamido, N -protected amino, (mono- or di) C 1 - 7 alkylamino, (mono- or di) arylamino, (mono- or di) C 3 - 10 cycloalkylamino, (mono- or di) hydroxy C 1 - 7 alkylamino, (mono- or di) C 1-4 alkyl-arylamino; preferably, said R 1 is selected from the group comprising amino, N- protected amino, (mono- or di) C 1 - 7 alkylamino, (mono- or di) arylamino, (mono- or di) C 3 - 10 cycloalkylamino, (mono- or di) C 1-4 alkyl-arylamino; preferably, said R 1 is selected from the group comprising amino, N- protected amino, (mono- or di)
- the present invention concern a compound of formula I, II, III or IV, a pharmaceutical acceptable addition salt thereof and/or a stereoisomer thereof and/or a solvate thereof, wherein R 1 , R 4 , and R 11 are each independently selected from the group consisting of amino, acetamido, N- protected amino, (mono- or di) C 1 - 7 alkylamino, (mono- or di) arylamino, (mono- or di) C 3 - 10 cycloalkylamino, (mono- or di) hydroxy C 1 - 7 alkylamino, (mono- or di) C 1-4 alkyl-arylamino.
- One embodiment of the present invention concerns a compound according to the invention wherein X is S. Another embodiment of the present invention concerns a compound according to the invention wherein X is O.
- R 1 and R 2 have any of the values as described herein and R 3 is selected from the group consisting of monoarylamino; diarylamino; (mono- or di-) arylC 1-4 alkylamino; morpholinyl; C 1-7 alkoxy, aralkylthio, piperidinyl, pyrrolidinyl, homopiperazinyl and piperazinyl, wherein said piperidinyl, pyrrolidinyl, homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R 20 , wherein R 20 has the same meaning as defined herein.
- R 4 and R 5 have any of the values as described herein and R 6 is selected from the group consisting of (mono- or di-) C 1-12 alkylamino, monoarylamino, diarylamino, (mono- or di-) C 3-10 cycloalkylamino, (mono- or di-) arylC 1-4 alkylamino, morpholinyl, C 1-7 alkoxy, homopiperazinyl and piperazinyl, wherein said homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R 21 , wherein R 21 has the same meaning as that defined in herein.
- R 11 , R 7 , R 8 and R 9 have any of the values as described herein and n is 0 and R 10 is selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C 3-10 cycloalkyl-alkyl, C 3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl
- R 1 and R 3 have any of the values as described herein
- R 2 is selected from the group consisting of heteroaryl and aryl groups; C 1-7 alkyl; C 3-10 cycloalkyl; halo C 1-7 alkyl; carboxy C 1-7 alkyl; carboxyaryl; C 1-7 alkoxy; C 3-10 cycloalkoxy; aryloxy; arylalkyloxy; heterocyclic-substituted alkyloxy; thio C 1-7 alkyl; thio C 3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; acylamino; thio-acylamino; alkoxyamino; carbamoyl; thiocarbamoyl; aromatic or heterocyclic substituents substituted with an alipha
- R 4 and R 6 have any of the values as described herein and R 5 is an aryl group, wherein said aryl group is optionally substituted with one or more substituents selected from the group consisting of halogen, C 1-7 alkyl, halo C 1-7 alkyl, nitro, hydroxyl, sulfhydryl, C 1-7 alkoxy, thio C 1-7 alkyl, cyano.
- R 11 , R 7 , R 12 and R 9 have any of the values as described herein, and R 8 is selected from the group consisting of heteroaryl and aryl groups; C 1-7 alkyl; halo C 1-7 alkyl; C 1-7 alkoxy; C 3-10 cycloalkoxy; aryloxy; arylalkyloxy; thio C 1-7 alkyl; thio C 3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the purine ring and the aromatic or heterocyclic substituent; wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C 1-7 alkyl, halo C 1-7 alkyl, nitro
- R 3 , R 6 are each independently homopiperazinyl or piperazinyl, wherein said homopiperazinyl or piperazinyl is each respectively optionally N-substituted with a substituent R 20 , R 21 , wherein R 20 and R 21 have the same meaning as that defined herein.
- One embodiment of the present invention concerns a compound according to the invention wherein R 1 , R 4 and R 11 are each independently amino.
- R 1 and R 3 have any of the values as described herein and R 2 is selected from the group consisting of phenyl; pyridin-3-yl; pyridin-2-yl; pyridin-4-yl; 4-fluorophenethyl; 4-fluorophenyl; 4-bromophenethyl; pentyl; tolyl; (4-fluorophenyl)butyl; (4-fluorophenyl)propyl; 4-chlorophenyl; 4-methylphenethyl; 3,4-dimethoxyphenethyl; 3-methoxyphenethyl; furan-2-yl; 2-phenylethyl; cyclohexyl; methoxylmethyl; cyclopropyl; 2-thiophen-2-ylethyl; cyclopentyl-(4-fluorophenyl)methyl; 1-(4-fluorophenyl)propy
- One embodiment of the present invention concerns a compound according to the invention wherein R 4 and R 6 have any of the values as described herein and R 5 is phenyl or 4-fluorophenyl.
- R 1 and R 2 have any of the values as described herein and R 3 is selected from the group consisting of p-tolyl piperazinyl-1-carboxylate; N-methyl-N-p-tolylpiperazinyl-1-carboxamide; N-p-tolylpiperazinyl-1-carbothioamide; -N-hexylpiperazinyl-1-carboxamide; 4-(N-4-fluorophenylcarboxamide)piperazin-1-yl; N-cyclohexylpiperazinyl-1-carboxamide; N-phenylpiperazinyl-1-carboxamide; N-(4-(trifluoromethyl)phenyl)piperazinyl-1-carboxamide, piperazin-1-yl-2-(4-chlorophenoxy)ethanone; piperazin-1-yl-2-(4-methoxyphenoxy)ethanone; benzylsulfonyl-1-carboxylate; N-methyl-
- R 2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl; R 1 and is amino; and R 3 is selected from the group consisting of methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-
- R 4 and R 6 have any of the values as described herein and R 5 is phenyl or 4-fluorophenyl.
- R 4 and R 5 have any of the values as described herein and R 6 is 4(m-tolylcarbamoyl)piperazin-1-yl or 4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl.
- R 7 , R 8 , R 9 , and R 11 have any of the values as described herein and R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl.
- R 5 is phenyl or 4-fluorophenyl
- R 4 is amino
- R 6 is (4(m-tolylcarbamoyl)piperazin-1-yl or (4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl).
- One embodiment of the present invention concerns a compound according to the invention wherein , R 7 , R 12 , R 9 , and R 11 have any of the values as described herein and R 8 is 4-fluorophenyl or methylthio.
- One embodiment of the present invention concerns a compound according to the invention wherein R 12 , R 8 , and R 11 have any of the values as described herein R 9 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R 8 , R 12 , and R 11 have any of the values as described herein R 7 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R 11 is amino, R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or (4-phenoxyacetyl)piperazin-1-yl, R 8 is 4-fluorophenyl or methylthio, and R 9 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R 11 is amino, R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl, R 8 is 4-fluorophenyl or methylthio, and R 7 is hydrogen or methyl.
- the present invention encompasses a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative having the general formula I :wherein - X is S or O;
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R 2 and R 3 have any of the values as described herein and R 1 is selected from the group consisting of amino, acetamido, N- protected amino, (mono- or di) C 1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C 3 - 10 cycloalkylamino, (mono- or di) hydroxy C 1 - 7 alkylamino, (mono- or di) C 1-4 alkyl-arylamino.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R 2 and R 1 have any of the values as described herein and R 3 is homopiperazinyl or piperazinyl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R 1 and R 3 have any of the values as described herein and R 2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R 2 and R 3 have any of the values as described herein and R 1 is selected from the group consisting of amino, methyl, and hydrogen.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R 2 and R 1 have any of the values as described herein and R 3 is selected from the group consisting of methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-yl, 4-(4-chlorophenoxyacetyl)piperazin-1-yl, 4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl, 4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl, and 4-(2-phenoxyacetyl)-piperazin-1-yl.
- R 2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl; R 5 is selected from the group consisting of amino, methyl, and hydrogen;
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein X is S.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein X is O.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 2-(4-fluorophenyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorobenzyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenethyl)-7-methylthio-thiazolo[5,4-d]pyrimidin-5-amine; 2-(2-(4-fluorophenoxy)ethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-5-methyl-7-methylthio-thiazolo[5,4-d]pyrimidine; 2-(4-fluorophenyl
- the present invention also concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- the present invention also concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal.
- the present invention also concerns the use of the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or a cells transplantation.
- said animal is a mammal, preferably said mammal is a human being.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- the present invention also concerns a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients.
- said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- the present invention also concerns a method of prevention or treatment of an immune disorder in an animal, comprising the administration of a therapeutically effective amount of a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative or formula I, optionally in combination with one or more pharmaceutically acceptable excipients.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said animal is a mammal, preferably said mammal is a human being.
- said acylation is performed with a carboxylic acid (R 2 COOH) or an acid chloride (R 2 C(O)Cl).
- step (a) further comprises the addition of a coupling reagent.
- said coupling reagent is N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI).
- step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt).
- step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent.
- said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene.
- step (c) is performed in alkaline conditions.
- said oxidating agent is m -chloro-peroxybenzoic acid or hydrogen peroxide.
- step (e) a piperazine is introduced at position 7.
- the thiazolo(5,4-d)pyrimidine derivatives of formula I are R 2 -substituted 5-amino-7- N -piperazino thiazolo(5,4-d)pyrimidine derivatives.
- the present invention encompasses a thieno(2,3-d)pyrimidine derivative having the general formula II :wherein
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 5 and R 6 have any of the values as described herein, and wherein R 4 is selected from the group consisting of amino, acetamido, N -protected amino, (mono- or di) C 1 - 7 alkylamino, (mono- or di) arylamino, (mono- or di) C 3 - 10 cycloalkylamino, (mono- or di) hydroxy C 1 - 7 alkylamino, (mono-or di) C 1-4 alkyl-arylamino.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 4 and R 5 have any of the values as described herein and wherein R 6 is homopiperazinyl or piperazinyl.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 4 and R 6 have any of the values as described herein and wherein R 5 is phenyl or 4-fluorophenyl.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 5 and R 6 have any of the values as described herein and wherein R 4 is butyl, methyl, or amino.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 4 and R 5 have any of the values as described herein and wherein R 6 is 4( m -tolylcarbamoyl)piperazin-1-yl or 4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R 5 is phenyl or 4-fluorophenyl; R 4 is butyl, methyl, or amino; and R 6 is (4( m -tolylcarbamoyl)piperazin-1-yl or (4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl).
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 1-(4-(2-butyl-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 2-butyl- N -(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine; 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone; 1-(4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-
- the present invention also concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- the present invention also concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal.
- the present invention also concerns the use of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or a cells transplantation.
- said animal is a mammal, preferably said mammal is a human being.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- the present invention also concerns a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients.
- said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- the present invention also concerns a method of prevention or treatment of an immune disorder in a mammal, comprising the administration of a therapeutically effective amount of a thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof , optionally in combination with one or more pharmaceutically acceptable excipients.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said animal is a mammal, preferably said mammal is a human being.
- the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; activation of carbonyl group by halogenation; a nucleophilic aromatic substitution reaction; introduction of a halogen at position 5; and introduction of R 5 by a palladium-catalyzed cross-coupling reaction.
- the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; treatment with a thionation reagent; an alkylation reaction; conversion of the thiomethyl group to sulfon by oxidation; introduction of the piperazine; introduction of a halogen at position 5; and introduction of R 5 by a palladium-catalyzed cross-coupling reaction.
- the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; treatment with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base, and piperazine leading to the formation of 2-amino-4- N- piperazinyl-thieno[2,3-d]pyrimidine; introduction of a halogen at position 5; and introduction of R 5 by a palladium-catalyzed cross-coupling reaction.
- a ring closure reaction on methyl 2-aminothiophene-3-carboxylate treatment with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base, and pipe
- said ring closure reaction is effected by treatment with chloroformamidine hydrochloride.
- said introduction of a halogen at position 5 is effected by treatment with n-BuLi and carbon tetrahalide as halogen source, preferably said halogen is bromide.
- said introduction of a halogen at position 5 is effected by reaction with N -halosuccinimide (NhS) in CCl 4 .
- said halogen is bromide and said N -halosuccinimide (NhS) is N- bromosuccinimide (NBS).
- said palladium-catalyzed cross-coupling reaction is a Suzuki coupling which include reactions with aryl boronic acids or aryl boronic acid pinacol esters; a Heck reaction which include reactions with terminal alkenes; a Sonogashira reaction which includes reactions with terminal alkynes and a Buchwald-Hartwig reaction which include reactions with arylamines; a Negishi reaction which includes the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides; or a Kumada coupling which includes coupling of Grignard reagents with aryl halides.
- said activation of carbonyl group by halogenation is performed in acidic conditions.
- said activation of carbonyl group by halogenation is performed by using phosphorus oxychloride or thionylchloride.
- said nucleophilic aromatic substitution reaction introduces a piperazine moiety at position 4.
- said treatment with a thionation reagent is performed by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene.
- said alkylation reaction is performed by treatment with an alkylhalide, such as methyliodide or benzylbromide; under alkaline conditions such as NaOH or triethylamine conditions; in a polar solvent, such as water, DMF or DMSO.
- said oxidation is performed by treatment with m -chloro-peroxybenzoic acid or hydrogen peroxide.
- said introduction of the piperazine is performed by a nucleophilic aromatic substitution reaction.
- said base is triethylamine, diisopropylethylamine, or DBU.
- the thieno(2,3-d)pyrimidine derivatives are R 5 -substituted 2-amino-4- N -piperazino-thieno[2,3-d]pyrimidine derivatives.
- the present invention encompasses a purine derivative having the general formula III or IV: wherein substituent R 12 is represented by the general formula V : and wherein schematically represents a saturated or partly unsaturated heterocyclic ring with at least two nitrogen atoms in the said heterocyclic ring and with a total of 5 to 7 atoms in the said heterocyclic ring, and optionally with one or more other heteroatoms in the said heterocyclic ring or attached to one or more carbon atoms of said heterocyclic ring (for instance in the form of a carbonyl or thiocarbonyl group), wherein one of said at least two nitrogen atoms in the heterocyclic ring is attached to a carbon atom 6 of the purine ring;
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 7 , R 8 , R 9 , R 12 have any of the values as described herein and wherein R 11 is amino.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 7 , R 8 , R 9 , R 11 have any of the values as described herein and wherein R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 7 , R 11 , R 9 , R 12 have any of the values as described herein and wherein R 8 is 4-fluorophenyl or methylthio.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 7 , R 8 , R 11 , R 12 have any of the values as described herein and wherein R 9 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 11 , R 8 , R 9 , R 12 have any of the values as described herein and wherein R 7 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 11 is amino, R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or (4-phenoxyacetyl)piperazin-1-yl, R 8 is 4-fluorophenyl or methylthio, and R 9 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R 11 is amino, R 12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl, R 8 is 4-fluorophenyl or methylthio, and R 7 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(3,4-dimethoxyphenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-bromophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-chlorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)
- the present invention also concerns purine derivatives of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- the present invention also concerns purine derivatives of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said animal is a mammal.
- said mammal is a human being.
- the present invention also concerns the use of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said animal is a mammal.
- said mammal is a human being.
- the present invention also concerns a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients.
- said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- the present invention also concerns a method of prevention or treatment of an immune disorder in a mammal, comprising the administration of a therapeutically effective amount of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, optionally in combination with one or more pharmaceutically acceptable excipients.
- said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- said mammal is a human being.
- the present invention also encompasses processes for the preparation of compounds of Formula (I), (II), (III), (IV) and subgroups thereof.
- reactive functional groups for example hydroxy, amino, or carboxy groups, where these are desired in the final product, to avoid their unwanted participation in the reactions.
- Conventional protecting groups can be used in accordance with standard practice, for example, see T. W. Greene and P. G. M. Wuts in "Protective Groups in Organic Chemistry", John Wiley and Sons, 1999 .
- the compounds of Formula (I) (II), (III), (IV) and the subgroups thereof can be prepared by a succession of steps as described herein. They are generally prepared from starting materials which are either commercially available or prepared by standard means obvious to those skilled in the art.
- the compounds of the present invention can be also prepared using standard synthetic processes commonly used by those skilled in the art of organic chemistry. The general preparation of some typical examples is shown below:
- 2,5,7-tri-substituted thiazolo[5,4-d]pyrimidine derivatives of formula Ia can be prepared as shown hereunder in Scheme 1.
- step (a) The synthesis starts from commercially available diethyl aminomalonate hydrochloride or dimethyl aminomalonate hydrochloride (not shown in Scheme 1). Conversion into the corresponding amides in step (a) can achieved by acylation with commercially available acid chlorides, bearing the general formula R 2 C(O)Cl (such as for example, but not limited to acetyl chloride, benzoyl chloride, phenoxyacetyl chloride).
- R 2 C(O)Cl such as for example, but not limited to acetyl chloride, benzoyl chloride, phenoxyacetyl chloride.
- the amino group can also be coupled with commercially available carboxylic acids, using standard peptide coupling procedures, using a carboxylic acid, bearing the general formula R 2 C(O)OH and a coupling reagent such as dicyclohexylcarbodiimide (DCC), diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI).
- DCC dicyclohexylcarbodiimide
- DIC diisopropylcarbodiimide
- EDCI 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
- Additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt) can be added to increase the reaction rate.
- the dialkyl acylaminomalonate formed in step (a) can be treated with guanidine, acetamidine or thiourea in an ethanolic sodium ethoxide solution yielding 2-R 1 -substituted pyrimidines in step (b).
- the carboxylic acid (or acid chloride) and the coupling partner By the judicious choices of the carboxylic acid (or acid chloride) and the coupling partner, different substituents can be introduced at positions 2 and 5 of the thiazolo[5,4-d]pyrimidine scaffold.
- step (c) Conversion of the lactam functionalities of 5-acylaminopyrimidine-4,6-diol into thiolactam groups and a concomitant ring closure reaction affording the thiazolo[5,4-d]pyrimidine scaffold can be achieved in step (c), by heating the compound with thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagent in high-boiling solvents such as pyridine, toluene or xylene.
- thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagent in high-boiling solvents such as pyridine, toluene or xylene.
- the 7-thio-compounds are treated with an alkylhalide (preferably iodomethane as shown in Scheme 1) in the presence of a base (such as for example triethylamine or NaOH) in an appropriate polar solvent (such as for example DMSO, DMF or water) to afford the corresponding alkylsulfanyl analogues.
- a base such as for example triethylamine or NaOH
- an appropriate polar solvent such as for example DMSO, DMF or water
- the thiomethyl group can be exchanged for a nucleophile bearing the general formula R 7 H.
- Nucleophiles can be primary or secondary amines (such as for example, but not limited to, isopropylamine, morpholine and piperazine) or alcoxides.
- step (f) In case of less reactive nucleophiles, it might be necessary to increase the reactivity of thioether group by oxidation to the corresponding sulfones with m-chloroperoxybenzoic acid (m CPBA) in dichloromethane (step (f)).
- m CPBA m-chloroperoxybenzoic acid
- step (f) The reaction of sulfone derivatives with a range of primary and secondary amines affords the 2-R 2 ,5-R 1 ,7-R 3 trisubstituted thiazolo[5,4-d]pyrimidines.
- the second nitrogen can be further derivatised by reaction with chloroformates, isocyanates, acid chlorides (or carboxylic acids) and sulfonyl chlorides yielding carbamates, urea, amides and sulfonamides, respectively.
- a one step procedure in order to introduce an amine at position 7 starting from the 7-thio-thiazolo[5,4-d]pyrimidine derivative is possible using 1,1,1,3,3,3-hexamethyldisilazane (HMDS) in a high boiling solvents such as toluene, xylene or pyridine (step (g)).
- HMDS 1,1,1,3,3,3-hexamethyldisilazane
- step (g) 2,5,7-tri-substituted thiazolo[5,4-d]pyrimidine derivatives of formula Ia can also be prepared as shown hereunder in Scheme 2.
- Scheme 2 depicts an alternative scheme for the synthesis of 2,5,7-trisubstituted thiazolo[5,4-d]pyrimidines, starting from a commercially available 2-R 1 -substituted-5-amino-4,6-dihydroxypyrimidine analogue, from which the amino group can be converted to an amide analogue in step (a) using the methods described before, yielding a 2-R 1 -substituted-5-acylamino-4,6-dihydroxypyrimidine derivative.
- this dihydroxy pyrimidine analogue can also be converted to its dichloro analogue in step (b) using chlorinating agents (such as for example thionyl chloride or phosphorus oxychloride).
- step (c) a nucleophilic aromatic substitution with one equivalent of an appropriate nucleophile, bearing the general formula R 3 H, yields the 2-R 1 ,4-R 3 ,5-amino-6-chloro-pyrimidine analogue.
- Introduction of a sulfhydryl group by reaction with sodium sulfide yields the 2-R 1 ,4-R 3 -5-amino-6-sulfhydryl-pyrimidine analogue in step (d).
- 2,5,7-trisubstituted oxazolo[5,4-d]pyrimidines of formula Ib can be prepared as shown in Scheme 3.
- the synthesis starts from intermediates that have been generated using synthetic schemes described in Schemes 1 and 2.
- a chlorinating agent such as for example POCl 3 , SOCl 2 , PCl 5
- steps (a) and step (b) Treatment of the 4,6-dihydroxy-pyrimidine analogue in steps (a) and step (b) with a chlorinating agent (such as for example POCl 3 , SOCl 2 , PCl 5 ) yields the 7-hydroxy- (step a) or 7-chloro derivative (step b), depending on the reaction time.
- the chlorine can be exchanged for a wide variety of oxygen, nitrogen or sulphur containing nucleophiles, bearing the general formula R 3 H in step (c).
- step (a) Acylation of one of the amino group yielding the amide derivative is achieved in step (a) by reaction with an appropriate acid chloride (bearing the general formula R 2 C(O)Cl) or with an appropriate acid (bearing the general formula R 2 COOH) and a coupling reagent, such as for example N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI).
- DCC N,N'-dicyclohexylcarbodiimide
- DIC N,N'-diisopropylcarbodiimide
- EDCI 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
- Additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy
- the acylaminopyrimidine analogue is heated with thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagents in high-boiling solvents such as pyridine, toluene or xylene.
- thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagents in high-boiling solvents such as pyridine, toluene or xylene.
- the thio group is then converted to its thiomethyl derivative in step (c) by treatment with iodomethane under alkaline conditions.
- 2,4,6-trisubstituted thieno[2,3-d]pyrimidine of formula II can be prepared as shown hereunder in Scheme 5.
- Scheme 5 schematically depicts the synthesis of 2,4,6-trisubstituted thieno[2,3-d]pyrimidine of formula II starting from the commercially available methyl 2-aminothiophene-3-carboxylate or ethyl 2-aminothiophene-3-carboxylate (structure not shown).
- a ring closure reaction can be effected in step (a) with formamide (yielding a hydrogen at position 2) or with chloroformamidine hydrochloride (furnishing an amino group at position 2).
- step (c) the corresponding carboxamide is obtained.
- Reaction of 2-amino-3-carboxamido-thiophene with a wide range of orthoesters e.g. triethylorthoformate and triethylorthoacetate
- 2-amino-3-carboxamido-thiophene can also be reacted in step (d) with a wide range of commercially available acid chlorides in order to construct the thieno[2,3-d]pyrimidine scaffold.
- the 4-chloro-thieno[2,3-d]pyrimidine analogue can be further derivatised either by a nucleophilic aromatic substitution reaction (S N Ar) or by palladium-catalyzed cross-coupling reactions in step (f).
- S N Ar nucleophilic aromatic substitution reaction
- the lactam group can be converted into a thiolactam group in step (g), by reaction with a thionation reagent such as for example by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene.
- the thio group is then alkylated in step (h) by treatment with an alkylhalide (such as for example methyliodide, benzylbromide) under alkaline conditions (e.g. NaOH, triethylamine) in a polar solvent (such as water, DMF or DMSO).
- an alkylhalide such as for example methyliodide, benzylbromide
- alkaline conditions e.g. NaOH, triethylamine
- a polar solvent such as water, DMF or DMSO
- step (k) Treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and a nitrogen or oxygen nucleophile leads to the formation of the corresponding 4-substituted thieno[2,3-d]pyrimidine in step (k).
- a halogen is introduced in step (I).
- the regioselective introduction of a bromine at position 6 can be performed with n-BuLi and carbon tetrabromide as bromine source at low temperature (-78°C).
- reaction with N -bromosuccinimide (NBS) in CCl 4 is also feasible.
- NBS N -bromosuccinimide
- the 6-bromo-thieno[2,3-d]pyrimidine is an ideal starting material for further derivatisation (step (m)) at position 6 by palladium-catalyzed cross-coupling reactions, such as for example Suzuki couplings (reactions with aryl boronic acids or aryl boronic acid pinacol esters), Heck reactions (reactions with terminal alkenes), Sonogashira (reaction with terminal alkynes) and Buchwald-Hartwig reactions (reaction with arylamines), Negishi reaction (the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides), Kumada coupling (coupling of Grignard reagents with aryl halides).
- step (a) The synthesis of 2-amino-4- N -piperazino-6-substituted thieno[2,3-d]pyrimidine of formula IIa starts from commercially available methyl 2-aminothiophene-3-carboxylate.
- step (a) a ring closure reaction is effected by treatment with chloroformamidine hydrochloride.
- the oxo group of the lactam functionality can be converted into a good leaving group such as halogens yielding an aryl chloride.
- This activation of carbonyl group by halogenation in step (b) is usually performed under harsh and acidic conditions using phosphorus oxychloride or thionylchloride.
- An additional step of protection of the amino group is needed before halogenation.
- step (c) Introduction of the piperazine moiety at position 4 of the scaffold happens in step (c) by a nucleophilic aromatic substitution reaction.
- the lactam group can be converted into a thiolactam group in step (d), by reaction with a thionation reagent such as for example by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene.
- the thio group is then alkylated in step (e) by treatment with an alkylhalide (such as for example methyliodide, benzylbromide) under alkaline conditions (e.g. NaOH, triethylamine) in a polar solvent (such as water, DMF or DMSO).
- an alkylhalide such as for example methyliodide, benzylbromide
- alkaline conditions e.g. NaOH, triethylamine
- a polar solvent such as water, DMF
- the thiomethyl group can be oxidized to its corresponding sulfon by treatment with m -chloro-peroxybenzoic acid or hydrogen peroxide in step (f).
- step (g) the piperazine is then introduced.
- treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and piperazine as nucleophile leads to the formation of the corresponding 2-amino-4- N -piperazinyl-thieno[2,3-d]pyrimidine analogue in step (h).
- a halogen at position 5 is introduced in step (i).
- the regioselective introduction of a bromine at position 5 can be performed with n-BuLi and carbon tetrabromide as bromine source at low temperature (-78°C).
- reaction with N -bromosuccinimide (NBS) in CCl 4 is also feasible.
- the 5-bromo-thieno[2,3-d]pyrimidine is an ideal starting material for further derivatisation at position 5 by palladium-catalyzed cross-coupling reactions (in step (j)), such as for example Suzuki couplings (reactions with aryl boronic acids or aryl boronic acid pinacol esters), Heck reactions (reactions with terminal alkenes), Sonogashira (reaction with terminal alkynes) and Buchwald-Hartwig reactions (reaction with arylamines), Negishi reaction (the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides), Kumada coupling (coupling of Grignard reagents with aryl halides).
- Suzuki couplings reactions with aryl boronic acids or aryl boronic acid pinacol esters
- Heck reactions reactions with terminal alkenes
- Sonogashira reaction with terminal alkynes
- Buchwald-Hartwig reactions reaction with
- a first step (a) commercially available 2,6-di-amino-4-chloro-pyrimidine is treated with a nucleophile such as for example piperazine (as shown in Scheme 8).
- a nucleophile such as for example piperazine
- this reaction is not limited to piperazine, but can be expanded to other types of nitrogen-containing nucleophiles such as, but not limited to, morpholine, pyrrolidine, methoxyethylamine and piperidine.
- oxygen-containing nucleophiles such as sodium ethoxide and sodium isopropoxide can be introduced.
- the piperazine moiety at position 4 of the pyrimidine ring is further derivatised.
- step (c) a nitroso substituent is introduced at position 5 of the pyrimidine scaffold, by reaction with sodium nitrite in water under acidic conditions (by use of acetic acid or hydrochloric acid).
- step (d) the exocyclic amino group at position 6 is acylated in THF in the presence of a base such as for example potassium carbonate or triethylamine.
- a base such as for example potassium carbonate or triethylamine.
- the reductive cyclization in order to construct the purine scaffold in step (e) is achieved by treatment of the acylamino intermediate with triphenylphosphine in toluene or xylene as solvent.
- the nitroso group can be reduced to the corresponding amino group in step (f). This can be done with, for example, sodium dithionite in water, or catalytically (using hydrogen gas and a catalyst such as Raney Nickel).
- the 5,6-di-amino-pyrimidine intermediate can then be ring closed in order to construct the purine scaffold.
- carboxylic acids (bearing the general formula R 8 COOH), in which first a 5-acylaminopyrimidne is formed, followed by an acid-catalyzed ring closure.
- an aldehyde (bearing the general formula R 8 CHO) can be used as coupling partner, in which first a Schiff base is formed between the 5-amino-moiety and the aldehyde, followed by oxidative cyclization in the presence of ferric chloride or copper acetate.
- 2,6,8-trisubstituted purine analogues of formula IIIa can be prepared as shown in Scheme 9, using 2,6-diamino-4-mercaptopyrimidine as starting material.
- the thiol group is selectively alkylated (for example methylated, as shown in Scheme 9), by treatment with an appropriate alkylhalide under alkaline conditions (using for example potassium carbonate or sodium hydroxide as a base).
- the nitroso group is introduced at position 5 of the pyrimidine scaffold, using similar reaction circumstances as explained in Scheme 8. This 5-nitroso pyrimidine derivative can then be used to construct the purine scaffold, as already mentioned in Scheme 8 (either directly from the nitroso intermediate or via the amino intermediate), affording a 2-amino-6-thiomethyl-8-substituted purine analogue in step (c).
- the thiomethyl group can directly by displaced by a suitable nucleophile (such as piperazine, as shown in Scheme 9) in step (d).
- a suitable nucleophile such as piperazine, as shown in Scheme 9
- the thiomethyl group can be oxidized in step (e) using an oxidizing agent, such as m-chloro-peroxybenzoic acid, affording the corresponding sulfone derivative, which can be displaced by an appropriate nucleophile in step (f) (such as, but not limited to, piperazine, as shown in Scheme 9).
- the piperazine moiety can be further derivatised in step (g), as explained in step (b) from Scheme 8.
- 2,6,8-trisubstituted purine analogues of formula IIIb can be prepared as shown in Scheme 10, using 2-mercapto-4-hydroxy-6-amino-pyrimidine as starting material.
- step (a) the thiol group is selectively alkylated (for example methylated, as shown in Scheme 10), by treatment with an appropriate alkylhalide under alkaline conditions (using for example potassium carbonate or sodium hydroxide as a base).
- step (b) the nitroso group is introduced at position 5 of the pyrimidine scaffold, using similar reaction circumstances as explained in Scheme 8. This nitroso intermediate can then be used to construct the purine scaffold, as already mentioned in Scheme 6 (either directly from the nitroso derivative or via the 5,6-di-amino analogue), affording a 2-thiomethyl-6-hydroxy-8-substituted purine analogue in step (c).
- Activation of the tautomeric hydroxyl group at position 6 of the purine scaffold for the subsequent nucleophilic displacement reaction occurs in step (d) by preparing the corresponding 6-(1,2,4-triazolyl)-purine derivative or 6-chloro-purine derivative.
- the 6-triazolyl derivative can be obtained by treating the 6-oxo-purine derivative with POCl 3 or 4-chlorophenyl phosphorodichloridate and 1,2,4-triazole in an appropriate solvent such as, but not limited to, pyridine or acetonitrile.
- the 4-chloro derivative can be obtained by treating the 6-oxo-purine derivative with thionyl chloride or POCl 3 .
- the chlorine atom or thiazolyl group is designated as L in Scheme 10.
- Nucleophilic displacement of the triazolyl group or chlorine atom occurs in step (e) by reaction with an appropriate nucleophile, such as, but not limited to, piperazine, as shown in Scheme 10.
- an appropriate nucleophile such as, but not limited to, piperazine
- step (i) it might be necessary to oxidize the thiomethyl group to its corresponding sulfoxide using hydrogen peroxide or m -chloro-peroxybenzoic acid in step (i).
- the sulfoxide group can be exchanged by a suitable nucleophile affording the desired 2,6,8-trisubstituted purine analogue.
- a purine analogue (synthesized according to one of the synthetic pathways explained in Schemes 8-10), is alkylated with an appropriate alkylhalide bearing the general formula RX (such as for example iodomethane, ethylbromide, benzylbromide), and a base (such as for example sodium hydride or potassium carbonate) in a polar aprotic solvent (such as for example DMF).
- RX such as for example iodomethane, ethylbromide, benzylbromide
- a base such as for example sodium hydride or potassium carbonate
- a polar aprotic solvent such as for example DMF
- the present invention also relates to the thiazolo(5,4-d)pyrimidine, oxazolo(5,4-d)pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, II, III or IV, being selected from the group consisting of:
- the invention relates to thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, II, III, or IV, as well as pharmaceutical compositions comprising such thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives as active principle, represented by the above mentioned structural formula I, II, III, or V and being in the form of a pharmaceutically acceptable salt.
- addition salts include any therapeutically active nontoxic addition salt which compounds represented by structural formula I, II, III, or IV are able to form with a salt-forming agent.
- Such addition salts may conveniently be obtained by treating the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of the invention with an appropriate salt-forming acid or base.
- thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives having basic properties may be converted into the corresponding therapeutically active, non-toxic acid addition salt form by treating the free base form with a suitable amount of an appropriate acid following conventional procedures.
- appropriate salt-forming acids include, for instance, inorganic acids resulting in forming salts such as but not limited to hydrohalides (e.g.
- hydrochloride and hydrobromide sulfate, nitrate, phosphate, diphosphate, carbonate, bicarbonate, and the like; and organic monocarboxylic or dicarboxylic acids resulting in forming salts such as, for example, acetate, propanoate, hydroxyacetate, 2-hydroxypropanoate, 2-oxopropanoate, lactate, pyruvate, oxalate, malonate, succinate, maleate, fumarate, malate, tartrate, citrate, methanesulfonate, ethanesulfonate, benzoate, 2-hydroxybenzoate, 4-amino-2-hydroxybenzoate, benzenesulfonate, p-toluenesulfonate, salicylate, p-aminosalicylate, palmoate, bitartrate, camphorsulfonate, edetate, 1 ,2-ethanedisulfonate, fumarate, glucohe
- Thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention including the ones represented by the structural formula I, II, III, or IV, having acidic properties may be converted in a similar manner into the corresponding therapeutically active, non-toxic base addition salt form.
- salt-forming bases include, for instance, inorganic bases like metallic hydroxides such as but not limited to those of alkali and alkaline- earth metals like calcium, lithium, magnesium, potassium and sodium, or zinc, resulting in the corresponding metal salt; organic bases such as but not limited to ammonia, alkylamines, benzathine, hydrabamine, arginine, lysine, N 1 N 1 - dibenzylethylenediamine, chloroprocaine, choline, diethanolamine, ethylene-diamine, N-methylglucamine, procaine and the like.
- inorganic bases like metallic hydroxides such as but not limited to those of alkali and alkaline- earth metals like calcium, lithium, magnesium, potassium and sodium, or zinc, resulting in the corresponding metal salt
- organic bases such as but not limited to ammonia, alkylamines, benzathine, hydrabamine, arginine, lysine, N 1 N 1 - dibenzyl
- Reaction conditions for treating the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, with an appropriate salt-forming acid or base are similar to standard conditions involving the same acid or base but different organic compounds with basic or acidic properties, respectively.
- the pharmaceutically acceptable salt will be designed, i.e.
- the salt-forming acid or base will be selected so as to impart greater water-solubility, lower toxicity, greater stability and/or slower dissolution rate to the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative of this invention.
- Another aspect of the present invention relates to the process for the preparation of the thiazolo(5,4-d)pyrimidine derivatives of this invention, and comprises the steps of: (a) acylation of 2,5-diamino-4,6-dihydroxypyrimidine; (b) treatment with a thionation reagent; (c) treatment with iodomethane; (d) oxidation reaction by adding an oxidating agent; and (e) a nucleophilic aromatic substitution reaction.
- step (a) said acylation is performed with a carboxylic acid (R 2 COOH) or an acid chloride (R 2 C(O)Cl) and/or step (a) further comprises the addition of a coupling reagent such as N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI) and optionally step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt).
- a coupling reagent such as N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcar
- step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent and said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene.
- step (c) is performed in alkaline conditions.
- said oxidating agent is m-chloro-peroxybenzoic acid or hydrogen peroxide.
- step (e) a piperazine is introduced at position 7.
- the present invention relates to the process for the preparation of the thiazolo(5,4-d)pyrimidine derivatives of this invention wherein said thiazolo(5,4-d)pyrimidine derivatives are R 2 -substituted 5-amino-7- N -piperazino thiazolo(5,4-d)pyrimidine derivatives, and wherein R 2 has any of the values as described herein.
- Another aspect of the present invention relates to the thiazolo[5,4-d]pyrimidine oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of the invention, including the thiazolo[5,4-d]pyrimidine, oxazolo(5,4-d)pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, for use as a medicine and to the use of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives as a medicine, more in particular to the use of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives to treat or prevent an immune disorder in an animal, even more in
- Another aspect of the present invention relates to the pharmaceutical composition of the invention for use as a medicine and to the use of said pharmaceutical composition as a medicine, more in particular to the use of said pharmaceutical composition to treat or prevent an immune disorder in an animal, even more in particularly to treat or prevent autoimmune disorders and particular organ and cells transplant rejections in an animal, more specifically a mammal such as a human being.
- the present invention further provides the use of thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, or a pharmaceutically acceptable salt or a solvate thereof, as a biologically active ingredient, i.e. active principle, especially as a medicine or a diagnostic agent or for the manufacture of a medicament or a diagnostic kit.
- said medicament may be for the prevention or treatment of a immune disorders, in particular organ and cells transplant rejections, and autoimmune disorders.
- the present invention further provides the use of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, or a pharmaceutically acceptable salt or a solvate thereof, as a biologically active ingredient, i.e. active principle, especially as a medicine or for the manufacture of a medicament for treating an immune disorder or for preventing a transplant rejection.
- any of the uses mentioned with respect to the present invention may be restricted to a nonmedical use (e.g. in a cosmetic composition), a non-therapeutic use, a non-diagnostic use, a non-human use (e.g. in a veterinary composition), or exclusively an in-vitro use, or a use with cells remote from an animal.
- the invention further relates to a pharmaceutical composition
- a pharmaceutical composition comprising: (a) one or more thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, and (b) one or more pharmaceutically acceptable carriers.
- this invention provides combinations, preferably synergistic combinations, of one or more thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivatives of this invention with one or more biologically active drugs being preferably selected from the group consisting of immunosuppressant and/or immunomodulator drugs.
- one or more biologically active drugs being preferably selected from the group consisting of immunosuppressant and/or immunomodulator drugs.
- the evaluation of a synergistic effect in a drug combination may be made by analyzing the quantification of the interactions between individual drugs, using the median effect principle described by Chou et al. in Adv. Enzyme Reg. (1984) 22:27 .
- this principle states that interactions (synergism, additivity, antagonism) between two drugs can be quantified using the combination index (hereinafter referred as Cl) defined by the following equation: wherein ED x is the dose of the first or respectively second drug used alone (1 a, 2a), or in combination with the second or respectively first drug (1c, 2c), which is needed to produce a given effect.
- this principle may be applied to a number of desirable effects such as, but not limited to, an activity against transplant rejection, an activity against immunosuppression or immunomodulation.
- the present invention relates to a pharmaceutical composition or combined preparation having synergistic effects against immuno-suppression or immunomodulation and containing: (a) one or more immunosuppressant and/or immunomodulator drugs, and (b) at least one thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of the invention, including the ones represented by the structural formula I, II, III, or IV and (c) optionally one or more pharmaceutical excipients or pharmaceutically acceptable carriers, for simultaneous, separate or sequential use in the treatment or prevention of autoimmune disorders and/or in transplant-rejections.
- Suitable immunosuppressant drugs for inclusion in the synergistic compositions or combined preparations of this invention belong to a well known therapeutic class. They are preferably selected from the group consisting of cyclosporine A, substituted xanthines (e.g.
- methylxanthines such as pentoxyfylline), daltroban, sirolimus, tacrolimus, rapamycin (and derivatives thereof such as defined below), leflunomide (or its main active metabolite A771726, or analogs thereof called malononitrilamides), mycophenolic acid and salts thereof (including the sodium salt marketed under the trade name Mofetil ® ), adrenocortical steroids, azathioprine, brequinar, gusperimus, 6-mercaptopurine, mizoribine, chloroquine, hydroxy-chloroquine and monoclonal antibodies with immunosuppressive properties (e.g.
- Adrenocortical steroids within the meaning of this invention mainly include glucocorticoids such as but not limited to ciprocinonide, desoxycorticosterone, fludrocortisone, flumoxonide, hydrocortisone, naflocort, procinonide, timobesone, tipredane, dexamethasone, methylprednisolone, methotrexate, prednisone, prednisolone, triamcinolone and pharmaceutically acceptable salts thereof.
- glucocorticoids such as but not limited to ciprocinonide, desoxycorticosterone, fludrocortisone, flumoxonide, hydrocortisone, naflocort, procinonide, timobesone, tipredane, dexamethasone, methylprednisolone, methotrexate, prednisone, prednisolone, triamcinolone and pharmaceutical
- Rapamycin derivatives as referred herein include O- alkylated derivatives, particularly 9-deoxorapamycins, 26-dihydrorapamycins, 40-O- substituted rapamycins and 28,40-0,0-disubstituted rapamycins (as disclosed in U.S. Patent No. 5,665,772 ) such as 40-O-(2-hydroxy) ethyl rapamycin - also known as SDZ-RAD -, pegylated rapamycin (as disclosed in U.S. Patent No. 5,780,462 ), ethers of 7-desmethylrapamycin (as disclosed in U.S. Patent No. 6,440,991 ) and polyethylene glycol esters of SDZ-RAD (as disclosed in U.S. Patent No. 6,331,547 ).
- Suitable immunomodulator drugs for inclusion into the synergistic immunomodulating pharmaceutical compositions or combined preparations of this invention are preferably selected from the group consisting of acemannan, amiprilose, bucillamine, dimepranol, ditiocarb sodium, imiquimod, Inosine Pranobex, interferon- ⁇ , interferon- ⁇ , lentinan, levamisole, lisophylline, pidotimod, romurtide, platonin, procodazole, propagermanium, thymomodulin, thymopentin and ubenimex.
- Synergistic activity of the pharmaceutical compositions or combined preparations of this invention against immunosuppression or immuno-modulation may be readily determined by means of one or more lymphocyte activation tests. Usually activation is measured via lymphocyte proliferation. Inhibition of proliferation thus always means immunosuppression under the experimental conditions applied.
- MLR mixed lymphocyte reaction
- T-cell activation which proceeds via the Ca 2 +/calmodulin/calcineurin system and can be inhibited e.g. by cyclosporine A (hereinafter referred as CyA); and c) a CD28 assay wherein specific activation of the T-lymphocyte proceeds via an exogenously added antibody against a CD28 molecule which is also located on the lymphocyte membrane and delivers strong co-stimulatory signals. This activation is Ca 2 +-independent and thus cannot be inhibited by CyA.
- CyA cyclosporine A
- Determination of the immunosuppressing or immunomodulating activity of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives of this invention, as well as synergistic combinations comprising them, is preferably based on the determination of one or more, preferably at least three lymphocyte activation in vitro tests, more preferably including at least one of the MLR test, CD3 assay and CD28 assay referred above.
- the lymphocyte activation in vitro tests used include at least two assays for two different clusters of differentiation preferably belonging to the same general type of such clusters and more preferably belonging to type I transmembrane proteins.
- the determination of the immuno-suppressing or immunomodulating activity may be performed on the basis of other lymphocyte activation in vitro tests, for instance by performing a TNF- ⁇ assay or an IL-1 assay or an IL-6 assay or an IL-10 assay or an IL-12 assay or an assay for a cluster of differentiation belonging to a further general type of such clusters and more preferably belonging to type II transmembrane proteins such as, but not limited to, CD69, CD71 or CD134.
- the synergistic effect may be evaluated by the median effect analysis method described herein before.
- Such tests may for instance, according to standard practice in the art, involve the use of equipment, such as flow cytometer, being able to separate and sort a number of cell subcategories at the end of the analysis, before these purified batches can be analyzed further.
- Synergistic activity of the pharmaceutical compositions of this invention in the prevention or treatment of transplant rejection may be readily determined by means of one or more leukocyte activation tests performed in a Whole Blood Assay (hereinafter referred as WBA) described for instance by Lin et al. in Transplantation (1997) 63:1734-1738 .
- WBA used herein is a lymphoproliferation assay performed in vitro using lymphocytes present in the whole blood, taken from animals that were previously given the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivative of this invention, and optionally the other immunosuppressant drug, in vivo.
- this assay reflects the in vivo effect of substances as assessed by an in vitro read-out assay.
- the synergistic effect may be evaluated by the median effect analysis method described herein before.
- Various organ transplantation models in animals are also available in vivo, which are strongly influenced by different immunogenicities, depending on the donor and recipient species used and depending on the nature of the transplanted organ. The survival time of transplanted organs can thus be used to measure the suppression of the immune response.
- the pharmaceutical composition or combined preparation with synergistic activity against immunosuppression or immunomodulation according to this invention may contain the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of this invention, including the ones represented by the structural formulae I, II, III, or IV over a broad content range depending on the contemplated use and the expected effect of the preparation.
- the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative content in the combined preparation is within the range of 0.1 to 99.9 % by weight, preferably from 1 to 99 % by weight, more preferably from about 5 to 95 % by weight.
- Auto-immune disorders to be prevented or treated by the pharmaceutical compositions or combined preparations of this invention include both:
- Transplant rejections to be prevented or treated by the pharmaceutical compositions or combined preparations of this invention include the rejection of transplanted or grafted organs or cells (both allografts and xenografts), such as but not limited to host versus graft reaction disease.
- organ as used herein means all organs or parts of organs in mammals, in particular humans, such as but not limited to kidney, lung, bone marrow, hair, cornea, eye (vitreous), heart, heart valve, liver, pancreas, blood vessel, skin, muscle, bone, intestine or stomach.
- rejection means all reactions of the recipient body or the transplanted organ which in the end lead to cell or tissue death in the transplanted organ or adversely affect the functional ability and viability of the transplanted organ or the recipient. In particular, this means acute and chronic rejection reactions. Also included in this invention is preventing or treating the rejection of cell transplants and xenotransplantation.
- the major hurdle for xenotransplantation is that even before the T lymphocytes, responsible for the rejection of allografts, are activated, the innate immune system, especially T-independent B lymphocytes and macrophages are activated. This provokes two types of severe and early acute rejection called hyperacute rejection and vascular rejection, respectively.
- the present invention addresses the problem that conventional immunosuppressant drugs like cyclosporine A are ineffective in xenotransplantation.
- the ability of the compounds of this invention to suppress T-independent xeno-antibody production as well as macrophage activation may be evaluated in the ability to prevent xenograft rejection in athymic, T-deficient mice receiving xenogenic hamster-heart grafts.
- pharmaceutically acceptable carrier or excipient as used herein in relation to pharmaceutical compositions and combined preparations means any material or substance with which the active principle, i.e. the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivative of this invention, including the ones represented by the structural formula I, II, III, or IV, and optionally the immunosuppressant or immunomodulator may be formulated in order to facilitate its application or dissemination to the locus to be treated, for instance by dissolving, dispersing or diffusing said composition, and/or to facilitate its storage, transport or handling without impairing its effectiveness.
- the pharmaceutically acceptable carrier may be a solid or a liquid or a gas which has been compressed to form a liquid, i.e. the compositions of this invention can suitably be used as concentrates, emulsions, solutions, granulates, dusts, sprays, aerosols, pellets or powders.
- suitable pharmaceutical carriers for use in said pharmaceutical compositions and their formulation are well known to those skilled in the art.
- Suitable pharmaceutical carriers include additives such as wetting agents, dispersing agents, stickers, adhesives, emulsifying or surface-active agents, thickening agents, complexing agents, gelling agents, solvents, coatings, antibacterial and antifungal agents (for example phenol, sorbic acid, chlorobutanol), isotonic agents (such as sugars or sodium chloride) and the like, provided the same are consistent with pharmaceutical practice, i.e. carriers and additives which do not create permanent damage to mammals.
- additives such as wetting agents, dispersing agents, stickers, adhesives, emulsifying or surface-active agents, thickening agents, complexing agents, gelling agents, solvents, coatings, antibacterial and antifungal agents (for example phenol, sorbic acid, chlorobutanol), isotonic agents (such as sugars or sodium chloride) and the like, provided the same are consistent with pharmaceutical practice, i.e. carriers and additives which do not create permanent damage to mammals.
- compositions of the present invention may be prepared in any known manner, for instance by homogeneously mixing, dissolving, spray-drying, coating and/or grinding the active ingredients, in a one-step or a multi-steps procedure, with the selected carrier material and, where appropriate, the other additives such as surface-active agents, may also be prepared by micronisation, for instance in view to obtain them in the form of microspheres usually having a diameter of about 1 to 10 ⁇ m, namely for the manufacture of microcapsules for controlled or sustained release of the biologically active ingredient(s).
- Suitable surface-active agents to be used in the pharmaceutical compositions of the present invention are non-ionic, cationic and/or anionic surfactants having good emulsifying, dispersing and/or wetting properties.
- Suitable anionic surfactants include both water-soluble soaps and water-soluble synthetic surface-active agents.
- Suitable soaps are alkaline or alkaline-earth metal salts, unsubstituted or substituted ammonium salts of higher fatty acids (C 10 -C 22 ), e.g. the sodium or potassium salts of oleic or stearic acid, or of natural fatty acid mixtures obtainable form coconut oil or tallow oil.
- Synthetic surfactants include sodium or calcium salts of polyacrylic acids; fatty sulphonates and sulphates; sulphonated benzimidazole derivatives and alkylarylsulphonates.
- Fatty sulphonates or sulphates are usually in the form of alkaline or alkaline-earth metal salts, unsubstituted ammonium salts or ammonium salts substituted with an alkyl or acyl radical having from 8 to 22 carbon atoms, e.g.
- Suitable sulphonated benzimidazole derivatives preferably contain 8 to 22 carbon atoms.
- alkylarylsulphonates are the sodium, calcium or alcanolamine salts of dodecylbenzene sulphonic acid or dibutyl-naphtalenesulphonic acid or a naphthalene-sulphonic acid/formaldehyde condensation product.
- corresponding phosphates e.g. salts of phosphoric acid ester and an adduct of p-nonylphenol with ethylene and/or propylene oxide, or phospholipids.
- Suitable phospholipids for this purpose are the natural (originating from animal or plant cells) or synthetic phospholipids of the cephalin or lecithin type such as e.g.
- phosphatidylethanolamine phosphatidylserine, phosphatidylglycerine, lysolecithin, cardiolipin, dioctanyl-phosphatidylcholine, dipalmitoylphoshatidylcholine and their mixtures.
- Suitable non-ionic surfactants include polyethoxylated and polypropoxylated derivatives of alkylphenols, fatty alcohols, fatty acids, aliphatic amines or amides containing at least 12 carbon atoms in the molecule, alkylarenesulphonates and dialkylsulphosuccinates, such as polyglycol ether derivatives of aliphatic and cycloaliphatic alcohols, saturated and unsaturated fatty acids and alkylphenols, said derivatives preferably containing 3 to 10 glycol ether groups and 8 to 20 carbon atoms in the (aliphatic) hydrocarbon moiety and 6 to 18 carbon atoms in the alkyl moiety of the alkylphenol.
- non-ionic surfactants are water-soluble adducts of polyethylene oxide with poylypropylene glycol, ethylenediamino-polypropylene glycol containing 1 to 10 carbon atoms in the alkyl chain, which adducts contain 20 to 250 ethyleneglycol ether groups and / or 10 to 100 propyleneglycol ether groups.
- Such compounds usually contain from 1 to 5 ethyleneglycol units per propyleneglycol unit.
- non-ionic surfactants are nonylphenol-polyethoxyethanol, castor oil polyglycolic ethers, polypropylene/polyethylene oxide adducts, tributylphenoxypolyethoxyethanol, polyethyleneglycol and octylphenoxypolyethoxyethanol.
- Fatty acid esters of polyethylene sorbitan such as polyoxyethylene sorbitan trioleate
- glycerol glycerol
- sorbitan sucrose and pentaerythritol are also suitable non-ionic surfactants.
- Suitable cationic surfactants include quaternary ammonium salts, preferably halides, having four hydrocarbon radicals optionally substituted with halo, phenyl, substituted phenyl or hydroxy; for instance quaternary ammonium salts containing as N-substituent at least one C 8 -C 22 alkyl radical (e.g. cetyl, lauryl, palmityl, myristyl, oleyl and the like) and, as further substituents, unsubstituted or halogenated lower alkyl, benzyl and/or hydroxy-C 1-4 alkyl radicals.
- quaternary ammonium salts preferably halides, having four hydrocarbon radicals optionally substituted with halo, phenyl, substituted phenyl or hydroxy
- quaternary ammonium salts containing as N-substituent at least one C 8 -C 22 alkyl radical (e.g. cetyl, lau
- Suitable such agents are in particular highly dispersed silicic acid, such as the product commercially available under the trade name Aerosil; bentonites; tetraalkyl ammonium salts of montmorillonites (e.g., products commercially available under the trade name Bentone), wherein each of the alkyl groups may contain from 1 to 20 carbon atoms; cetostearyl alcohol and modified castor oil products (e.g. the product commercially available under the trade name Antisettle).
- Gelling agents which may be included into the pharmaceutical compositions and combined preparations of the present invention include, but are not limited to, cellulose derivatives such as carboxymethylcellulose, cellulose acetate and the like; natural gums such as arabic gum, xanthum gum, tragacanth gum, guar gum and the like; gelatin; silicon dioxide; synthetic polymers such as carbomers, and mixtures thereof.
- Gelatin and modified celluloses represent a preferred class of gelling agents.
- additives such as magnesium oxide; azo dyes; organic and inorganic pigments such as titanium dioxide; UV-absorbers; stabilisers; odor masking agents; viscosity enhancers; antioxidants such as, for example, ascorbyl palmitate, sodium bisulfite, sodium metabisulfite and the like, and mixtures thereof; preservatives such as, for example, potassium sorbate, sodium benzoate, sorbic acid, propyl gallate, benzylalcohol, methyl paraben, propyl paraben and the like; sequestering agents such as ethylene-diamine tetraacetic acid; flavoring agents such as natural vanillin; buffers such as citric acid and acetic acid; extenders or bulking agents such as silicates, diatomaceous earth, magnesium oxide or aluminum oxide; densification agents such as magnesium salts; and mixtures thereof.
- additives such as magnesium oxide; azo dyes; organic and inorganic pigments such as titanium dioxide; UV-absorb
- Control release compositions may thus be achieved by selecting appropriate polymer carriers such as for example polyesters, polyamino-acids, polyvinyl-pyrrolidone, ethylene-vinyl acetate copolymers, methylcellulose, carboxy-methylcellulose, protamine sulfate and the like.
- the rate of drug release and duration of action may also be controlled by incorporating the active ingredient into particles, e.g. microcapsules, of a polymeric substance such as hydrogels, polylactic acid, hydroxymethyl-cellulose, polymethyl methacrylate and the other above-described polymers.
- Such methods include colloid drug delivery systems including, but not limited to liposomes, microspheres, microemulsions, nanoparticles, nanocapsules and so on.
- the pharmaceutical composition or combined preparation of the invention may also require protective coatings.
- compositions suitable for injectable use include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation thereof.
- Typical carriers for this purpose therefore include biocompatible aqueous buffers, ethanol, glycerol, propylene glycol, polyethylene glycol, complexing agents such as cyclodextrins and the like, and mixtures thereof.
- the selected active agent may be administered by way of intracavernosal injection, or may be administered topically, in an ointment, gel or the like, or transdermal, including transscrotally, using a conventional transdermal drug delivery system.
- Intracavernosal injection can be carried out by use of a syringe or any other suitable device.
- An example of a hypodermic syringe useful herein is described in U.S. Patent No. 4,127,118 , injection being made on the dorsum of the penis by placement of the needle to the side of each dorsal vein and inserting it deep into the corpora.
- the said combined preparation may be in the form of a medical kit or package containing the two ingredients in separate but adjacent form.
- each ingredient may therefore be formulated in a way suitable for an administration route different from that of the other ingredient, e.g. one of them may be in the form of an oral or parenteral formulation whereas the other is in the form of an ampoule for intravenous injection or an aerosol.
- the present invention further relates to a method for preventing or treating a disease selected from the group consisting of immune and auto-immune disorders, transplant rejections, in a patient, preferably a mammal, more preferably a human being.
- the method of this invention consists of administering to the patient in need thereof an effective amount of a thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of this invention, including the ones represented by the structural formula I, II, III, or IV, optionally together with an effective amount of another immunosuppressant or immunomodulator or antineoplastic drug or antiviral agent or phosphodiesterase-4 inhibitor, or a pharmaceutical composition comprising the same, such as disclosed above in extensive details.
- the effective amount is usually in the range of about 0.01 mg to 20 mg, preferably about 0.1 mg to 5 mg, per day per kg bodyweight for humans. Depending upon the pathologic condition to be treated and the patient's condition, the said effective amount may be divided into several sub-units per day or may be administered at more than one day intervals.
- the patient to be treated may be any warm-blooded animal, preferably a mammal, more preferably a human being, suffering from said pathologic condition.
- the preferred compounds of the present invention are non-sedating.
- a dose of such compounds that is twice the minimum dose sufficient to provide analgesia in an animal model for determining pain relief causes only transient (i. e. lasting for no more than half the time that pain relief lasts) or preferably no statistically significant sedation in an animal model assay of sedation (using the method described by Fitzgerald et al. in Toxicology (1988) 49:433-9 ).
- a dose that is five times the minimum dose sufficient to provide analgesia does not produce statistically significant sedation.
- a compound provided herein does not produce sedation at intravenous doses of less than 10 mg/kg per day or at oral doses of less than 30 mg/kg per day.
- compounds provided herein may be evaluated for toxicity (a preferred compound is non-toxic when an immunomodulating amount or a cell anti-proliferative amount is administered to a subject) and/or side effects (a preferred compound produces side effects comparable to placebo when a therapeutically effective amount of the compound is administered to a subject). Toxicity and side effects may be assessed using any standard method.
- the term "non-toxic" as used herein shall be understood as referring to any substance that, in keeping with established criteria, is susceptible to approval by the United States Federal Drug Administration for administration to mammals, preferably humans.
- Toxicity may be also evaluated using assays including bacterial reverse mutation assays, such as an Ames test, as well as standard teratogenicity and tumorogenicity assays.
- administration of compounds provided herein within the therapeutic dose ranges disclosed hereinabove does not result in prolongation of heart QT intervals (e.g. as determined by electrocardiography in guinea pigs, minipigs or dogs).
- such doses also do not cause liver enlargement resulting in an increase of liver to body weight ratio of more than 50 % over matched controls in laboratory rodents (e. g. mice or rats).
- Such doses also preferably do not cause liver enlargement resulting in an increase of liver to body weight ratio of more than 10 % over matched untreated controls in dogs or other non-rodent mammals.
- the preferred compounds of the present invention also do not promote substantial release of liver enzymes from hepatocytes in vivo, i.e. the therapeutic doses do not elevate serum levels of such enzymes by more than 50% over matched untreated controls in vivo in laboratory rodents.
- Another embodiment of this invention includes the various precursor or "prodrug" forms of the compounds of the present invention.
- the compounds of the present invention may be desirable to formulate the compounds of the present invention in the form of a chemical species which itself is not significantly biologically active, but which when delivered to the body of a human being or higher mammal will undergo a chemical reaction catalyzed by the normal function of the body, inter alia, enzymes present in the stomach or in blood serum, said chemical reaction having the effect of releasing a compound as defined herein.
- the term "prodrug” or "pro-drug” thus relates to these species which are converted in vivo into the active pharmaceutical ingredient.
- the pro-drugs of the present invention can have any form suitable to the formulator, for example, esters are non-limiting common pro-drug forms.
- the pro-drug may necessarily exist in a form wherein a covalent bond is cleaved by the action of an enzyme present at the target locus.
- a C-C covalent bond may be selectively cleaved by one or more enzymes at said target locus and, therefore, a pro-drug in a form other than an easily hydrolysable precursor, inter alia an ester, an amide, and the like, may be used.
- the term "therapeutically suitable pro-drug” is defined herein as a compound modified in such a way as to be transformed in vivo to the therapeutically active form, whether by way of a single or by multiple biological transformations, when in contact with the tissues of humans or mammals to which the prodrug has been administered, and without undue toxicity, irritation, or allergic response, and achieving the intended therapeutic outcome.
- the present invention will be further described with reference to certain more specific embodiments and examples, but the present invention is not limited thereto. The following examples are given by way of illustration only.
- C 1-7 alkyl means straight and branched chain saturated acyclic hydrocarbon monovalent radicals having from 1 to 7 carbon atoms such as, for example, methyl, ethyl, propyl, n-butyl, 1-methylethyi (isopropyl), 2-methylpropyl (isobutyl), 1 ,1-dimethylethyl (ter-butyl), 2-methylbutyl, n-pentyl, dimethylpropyl, n- hexyl, 2-methylpentyl, 3-methylpentyl, n-heptyl and the like.
- C 1 - 12 alkyl refers to such radicals having from 1 to 12 carbon atoms, i.e. up to and including dodecyl.
- acyl broadly refers to a substituent derived from an acid such as an organic monocarboxylic acid, a carbonic acid, a carbamic acid (resulting into a carbamoyl substituent) or the thioacid or imidic acid (resulting into a carbamidoyl substituent) corresponding to said acids
- sulfonyl refers to a substituent derived from an organic sulfonic acid, wherein said acids comprise an aliphatic, aromatic or heterocyclic group in the molecule.
- said acyl group refers to a carbonyl (oxo) group adjacent to a C 1-7 alkyl, a C 3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, all of them being such as herein defined.
- Suitable examples of acyl groups are to be found below.
- sulfonyl group refers to a sulfonyl group adjacent to a C 1-7 alkyl, a C 3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, all of them being such as herein defined.
- Acyl and sulfonyl groups originating from aliphatic or cycloaliphatic monocarboxylic acids or sulfonic acids are designated herein as aliphatic or cycloaliphatic acyl and sulfonyl groups and include, but are not limited to, the following:
- Acyl and sulfonyl groups may also originate from aromatic monocarboxylic acids and include, but are not limited to, the following:
- Acyl groups may also originate from an heterocyclic monocarboxylic acids and include, but are not limited to, the following:
- C 1-7 alkylene means the divalent hydrocarbon radical corresponding to the above defined C 1-7 alkyl, such as methylene, bis(methylene), tris(methylene), tetramethylene, hexamethylene and the like.
- C 3-10 cycloalkyl means a mono- or polycyclic saturated hydrocarbon monovalent radical having from 3 to 10 carbon atoms, such as for instance cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and the like, or a C 7-10 polycyclic saturated hydrocarbon monovalent radical having from 7 to 10 carbon atoms such as, for instance, norbornyl, fenchyl, trimethyltricycloheptyl or adamantyl.
- C 3-10 cycloalkyl-alkyl refers to an aliphatic saturated hydrocarbon monovalent radical (preferably a C 1-7 alkyl such as defined above) to which a C 3-10 cycloalkyl (such as defined above) is already linked such as, but not limited to, cyclohexylmethyl, cyclopentylmethyl and the like.
- C 3-10 cycloalkylene means the divalent hydrocarbon radical corresponding to the above defined C 3-10 cycloalkyl.
- aryl designate any mono- or polycyclic aromatic monovalent hydrocarbon radical having from 6 up to 30 carbon atoms such as but not limited to phenyl, naphthyl, anthracenyl, phenantracyl, fluoranthenyl, chrysenyl, pyrenyl, biphenylyl, terphenyl, picenyl, indenyl, biphenyl, indacenyl, benzocyclobutenyl, benzocyclooctenyl and the like, including fused benzo-C 4- ⁇ cycloalkyl radicals (the latter being as defined above) such as, for instance, indanyl, tetrahydronaphthyl, fluorenyl and the like, all of the said radicals being optionally substituted with one or more substituents independently selected from the group consisting of halogen, amino, trifluoromethyl
- a substituting radical such as the combination of substituents in certain positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine ring together with the carbon atoms in the same positions of said ring
- the term "homocyclic” means a mono- or polycyclic, saturated or mono-unsaturated or polyunsaturated hydrocarbon radical having from 4 up to 15 carbon atoms but including no heteroatom in the said ring; for instance said combination of substituents may form a C 2-6 alkylene radical, such as tetramethylene, which cyclizes with the carbon atoms in certain positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine ring.
- heterocyclic means a mono- or polycyclic, saturated or mono-unsaturated or polyunsaturated monovalent hydrocarbon radical having from 2 up to 15 carbon atoms and including one or more heteroatoms in one or more heterocyclic rings, each of said rings having from 3 to 10 atoms (and optionally further including one or more heteroatoms attached to one or more carbon atoms of said ring, for instance in the form of a carbonyl or thiocarbonyl or selenocarbonyl group, and/or to one or more heteroatoms of said ring, for instance in the form of a sulfone,
- each carbon atom of said heterocyclic ring may furthermore be independently substituted with a substituent selected from the group consisting of halogen, nitro, C 1-7 alkyl (optionally containing one or more functions or radicals selected from the group consisting of carbonyl (oxo), alcohol (hydroxyl), ether (alkoxy), acetal, amino, imino
- halogen or halo means any atom selected from the group consisting of fluorine, chlorine, bromine and iodine.
- halo C 1-7 alkyl means a C 1-7 alkyl radical (such as above defined) in which one or more hydrogen atoms are independently replaced by one or more halogens (preferably fluorine, chlorine or bromine), such as but not limited to difluoromethyl, trifluoromethyl, trifluoroethyl, octafluoropentyl, dodecafluoroheptyl, dichloromethyl and the like.
- halogens preferably fluorine, chlorine or bromine
- C 2-7 alkenyl designate a straight and branched acyclic hydrocarbon monovalent radical having one or more ethylenic unsaturations and having from 2 to 7 carbon atoms such as, for example, vinyl, 1-propenyl, 2-propenyl (allyl), 1-butenyl, 2-butenyl, 2-pentenyl, 3-pentenyl, 3-methyl-2-butenyl, 3-hexenyl, 2-hexenyl, 2-heptenyl, 1,3-butadienyl, pentadienyl, hexadienyl, heptadienyl, heptatrienyl and the like, including all possible isomers thereof.
- C 3-10 cycloalkenyl means a monocyclic mono- or polyunsaturated hydrocarbon monovalent radical having from 3 to 8 carbon atoms, such as for instance cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohexadienyl, cycloheptenyl, cycloheptadienyl, cycloheptatrienyl, cyclooctenyl, cyclooctadienyl and the like, or a C 7-10 polycyclic mono- or polyunsaturated hydrocarbon monovalent radical having from 7 to 10 carbon atoms such as dicyclopentadienyl, fenchenyl (including all isomers thereof, such as ⁇ -pinolenyl), bicyclo[2.2.1]hept-2-eny
- C 2-7 alkynyl defines straight and branched chain hydrocarbon radicals containing one or more triple bonds and optionally at least one double bond and having from 2 to 7 carbon atoms such as, for example, acetylenyl, 1-propynyl, 2- propynyl, 1-butynyl, 2-butynyl, 2-pentynyl, 1-pentynyl, 3-methyl-2-butynyl, 3-hexynyl, 2-hexynyl, 1-penten-4-ynyl, 3-penten-1-ynyl, 1,3-hexadien-1-ynyl and the like.
- arylalkyl refers to an aliphatic saturated or ethylenically unsaturated hydrocarbon monovalent radical (preferably a C 1-7 alkyl or C 2-7 alkenyl radical such as defined above) onto which an aryl or heterocyclic radical (such as defined above) is already bonded via a carbon atom, and wherein the said aliphatic radical and/or the said aryl or heterocyclic radical may be optionally substituted with one or more substituents independently selected from the group consisting of halogen, amino, hydroxyl, sulfhydryl, C 1-7 alkyl, C 1-7 alkoxy, trifluoromethyl and nitro, such as but not limited to benzyl, 4-chlorobenzyl, 4-fluorobenzyl, 2-fluorobenzyl, 3,4-dich
- alkylaryl and alkyl-substituted heterocyclic refer to an aryl or, respectively, heterocyclic radical (such as defined above) onto which are bonded one or more aliphatic saturated or unsaturated hydrocarbon monovalent radicals, preferably one or more C 1-7 alkyl, C 2-7 alkenyl or C 3-10 cycloalkyl radicals as defined above such as, but not limited to, o-toluyl, m-toluyl, p-toluyl, 2,3-xylyl, 2,4-xylyl, 3,4- xylyl, o-cumenyl, m-cumenyl, p-cumenyl, o-cymenyl, m-cymenyl, p-cymenyl, mesityl, ter-butylphenyl, lutidinyl (i.e
- alkoxyaryl refers to an aryl radical (such as defined above) onto which is (are) bonded one or more C 1-7 alkoxy radicals as defined above, preferably one or more methoxy radicals, such as, but not limited to, 2-methoxyphenyl, 3-methoxyphenyl, 4-methoxyphenyl, 3,4-dimethoxyphenyl, 2,4,6-trimethoxyphenyl, methoxynaphtyl and the like.
- alkylamino As used herein with respect to a substituting radical, and unless otherwise stated, the terms “ alkylamino ", “ cycloalkylamino “, '.' alkenylamino ", “ cyclo- alkenylamino " , “ arylamino “, “ arylalkylamino “, “ heterocyclic-substituted alkylamino ", “ heterocyclic-substituted arylamino ", “ heterocyclic amino ", " hydroxy- alkylamino ", “ mercaptoalkylamino " and “ alkynylamino " mean that respectively one (thus monosubstituted amino) or even two (thus disubstituted amino) C 1-7 alkyl, C 3-10 cycloalkyl, C 2-7 alkenyl, C 3-10 cycloalkenyl, aryl, arylalkyl, heterocyclic-substituted alkyl, heterocycl
- an alkyl radical and an alkenyl radical or to two different radicals within the same subset of radicals, e.g. methylethylamino; among di-substituted amino radicals, symmetrically-substituted amino radicals are more easily accessible and thus usually preferred from a standpoint of ease of preparation.
- the terms " (thio)carboxylic acid-ester ", “ (thio)carboxylic acid thioester “ and “ (thio)carboxylic acid amide” refer to radicals wherein the carboxyl or thiocarboxyl group is bonded to the hydrocarbonyl residue of an alcohol, a thiol, a polyol, a phenol, a thiophenol, a primary or secondary amine, a polyamine, an amino-alcohol or ammonia, the said hydrocarbonyl residue being selected from the group consisting of alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, arylalkyl, alkylaryl, alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, arylamino, arylalkylamino
- amino-acid refers to a radical derived from a molecule having the chemical formula H 2 N-CHR-COOH 1 wherein R is the side group of atoms characterizing the amino-acid type; said molecule may be one of the 20 naturally- occurring amino-acids or any similar non naturally-occurring amino-acid.
- stereoisomer refers to all possible different isomeric as well as conformational forms which the compounds of formula I, II, III, or IV may possess, in particular all possible stereochemical ⁇ and conformationally isomeric forms, all diastereomers, enantiomers and/or conformers of the basic molecular structure. Some compounds of the present invention may exist in different tautomeric forms, all of the latter being included within the scope of the present invention.
- enantiomer means each individual optically active form of a compound of the invention, having an optical purity or enantiomeric excess (as determined by methods standard in the art) of at least 80% (i.e. at least 90% of one enantiomer and at most 10% of the other enantiomer), preferably at least 90% and more preferably at least 98%.
- solvate includes any combination which may be formed by a thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivative of this invention with a suitable inorganic solvent (e.g. hydrates) or organic solvent, such as but not limited to alcohols, ketones, esters, ethers, nitriles and the like.
- suitable inorganic solvent e.g. hydrates
- organic solvent such as but not limited to alcohols, ketones, esters, ethers, nitriles and the like.
- Mass spectra are obtained with a Finnigan LCQ advantage Max (ion trap) mass spectrophotometer from Thermo Finnigan, San Jose, CA, USA. Exact mass measurements are performed on a quadrupole time-of-flight mass spectrometer (Q-tof-2, Micromass, Manchester, UK) equipped with a standard electrospray-ionization (ESI) interface. Samples were infused in i -PrOH/H 2 O (1:1) at 3 ⁇ l/min. Melting points are determined on a Barnstead IA 9200 apparatus and are uncorrected. Precoated aluminum sheets (Fluka Silica gel/TLC-cards, 254 nm) were used for TLC. Column chromatography was performed on ICN silica gel 63-200, 60 ⁇ .
- This compound was synthesized from dimethyl aminomalonate according to the procedure for the preparation of example 2 using 4-fluorophenylpropionic acid.
- the crude residue was purified by flash chromatography on silica (CH 2 Cl 2 /MeOH 50:1) to yield the title compound as a white solid (53 %).
- This compound was synthesized from dimethyl aminomalonate according to the procedure for the preparation of example 2 using 3-(4-fluorophenoxy)propionic acid.
- the crude residue was purified by flash chromatography on silica (CH 2 Cl 2 /MeOH 50:1) to yield the title compound as a white solid (80 %).
- This compound was prepared from example 3 in a yield of 76%, according to the procedure for the synthesis of example 5.
- This compound was prepared from example 4 in a yield of 34%, according to the procedure for the synthesis of example 5.
- Acetamidine hydrochloride (1.05 g, 11.1 mmol) and dimethyl 2-(4-fluorobenzamido)malonate (1.0 g, 3.71 mmol) were added to a solution of sodium (0.26 g, 11.1 mmol) in ethanol (37 ml). The reaction mixture was refluxed for 3 hours. Then, after cooling, the solid product was collected and washed with ethanol. The product was dissolved in a minimal volume of water and acidified to pH 4-5 with 5M HCl. The precipitate was collected, washed with water and dried to give the title compound as a white solid (0.77 g, 79 %).
- Triethylamine (0.85 ml, 6.12 mmol) and iodomethane (0.31 ml, 4.90 mmol) were added.
- the reaction mixture was stirred for 12 hours at room temperature under a nitrogen atmosphere.
- the mixture was poured into water and extracted with EtOAc.
- the combined organic layers were dried over Na 2 SO 4 and the solvents were removed under reduced pressure.
- the crude residue was purified by chromatography on silica gel (CH 2 Cl 2 /MeOH 100:1), yielding the title compound as a light yellow solid (0.45 g, 15 % over 2 steps).
- This compound was synthesized according to the procedure for example 32, using 1-(thiazol-2-yl)piperazine.
- the crude residue was purified by flash chromatography on silica (CH 2 Cl 2 /MeOH 50:1), yielding the pure title compound as a white solid (43 %).
- This compound was prepared according to the procedure for the synthesis of example 42, using m-tolylisocyanate in a yield of 57%.
- Example 48 Synthesis of 1-(4-(5-amino-2-(2-(4-fluorophenoxy)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- Example 50 Synthesis of 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- Example 51 Synthesis of 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone
- This compound was prepared from example 27 using 4-fluorophenoxyacetic acid in a yield of 56%, according to the procedure for the synthesis of example 50.
- This compound was prepared from example 27 using 4-bromophenoxyacetic acid in a yield of 47%, according to the procedure for the synthesis of example 50.
- This compound was prepared from example 27 using 3-methylphenoxyacetic acid in a yield of 44%, according to the procedure for the synthesis of example 50.
- Example 54 Synthesis of 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(2,4-dichlorophenoxy)ethanone
- This compound was prepared from example 27 using 2,4-dichlorophenoxyacetic acid in a yield of 43%, according to the procedure for the synthesis of example 50.
- Example 55 Synthesis of 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-3-(4-fluorophenoxy)propan-1-one
- This compound was prepared from example 27 using 3-(4-fluorophenoxy)propionic acid in a yield of 43%, according to the procedure for the synthesis of example 50.
- This compound was synthesized in 91% yield, using cyclopropanecarbonyl chloride.
- This compound was synthesized from 2-methoxyacetyl chloride in 97 % yield.
- This compound was synthesized from hexanoyl chloride in 95 % yield.
- This compound was synthesized from diethyl 2-(cyclopropanecarboxamido)malonate in 91 % yield.
- This compound was synthesized from diethyl 2-(2-methoxyacetamido)malonate in 65 % yield.
- This compound was synthesized from diethyl 2-hexanamidomalonate (2.73 g, 10 mmol) in 83% yield.
- This compound was synthesized from cyclohexane carbonyl chloride in 87 % yield.
- This compound was synthesized from nicotinoyl chloride hydrochloride in 81 % yield.
- This compound was synthesized from 3-phenylpropanoyl chloride in 96 % yield.
- This compound was synthesized from 4-chlorophenylacetyl chloride yielding the title compound in 95 % yield.
- This compound was synthesized from 3,4-dichlorobenzoyl chloride yielding the title compound in 89 % yield.
- This compound was synthesized from 3-methoxybenzoyl chloride, yielding the title compound in 94 % yield.
- This compound was synthesized from ethyl chloroformate yielding the title compound in 89 % yield.
- This compound was synthesized using 1-phenylcyclopropanecarboxylic acid, yielding the title compound in 87% yield.
- This compound was synthesized from 1-(4-chlorophenyl)cyclopropanecarboxylic acid yielding the title compound in 84 % yield.
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(acylamino)pyrimidine in 67 % yield.
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3-phenylpropanamido)pyrimidine (550 mg, 2.0 mmol), yielding the title compound (530 mg, 92%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3-nicotinamido)pyrimidine (494 mg, 2.0 mmol), yielding the title compound (260 mg, 50%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-cyclohexanecarboxamidopyrimidine (504 mg, 2.0 mmol), yielding the title compound (400 mg, 75%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(4-chlorophenylacetamido)pyrimidine (589 mg, 2.0 mmol), yielding the title compound (500 mg, 81%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5(4-chlorobenzamido)pyrimidine (560 mg, 2.0 mmol), yielding the title compound (450 mg, 76%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5(4-methoxybenzamido)pyrimidine (1.10 g, 4.0 mmol), yielding the title compound (1.0 g, 86%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3,4-dichlorobenzamido)pyrimidine (1.26 g, 4.0 mmol), yielding the title compound (1.1 g, 83%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(1-phenylcyclopropanecarboxamido)pyrimidine (1.15 g, 4.0 mmol), yielding the title compound (1.1 g, 92%).
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(1-phenylcyclopropanecarboxamido)pyrimidine (1.28 g, 4.0 mmol), yielding the title compound (1.1 g, 82%).
- This compound was synthesized from 5-amino-2-cyclopropyl-thiazolo[5,4-d]pyrimidine-7-thiol in 92 % yield.
- This compound was synthesized from 5-amino-2-(3-pyridinyl)thiazolo[5,4-d]pyrimidine-7-thiol (261 mg, 1.0 mmol), yielding the pure title compound (150 mg, 54%).
- This compound was synthesized from 5-amino-2-(4-chlorobenzyl)thiazolo[5,4-d]pyrimidine-7-thiol (308 mg, 1.0 mmol), yielding the pure title compound (100 mg, 31%).
- This compound was synthesized from 5-amino-2-(3-methoxyphenyl)thiazolo[5,4-d]pyrimidine-7-thiol (290 mg, 1.0 mmol), yielding the pure title compound (250 mg, 82 %).
- This compound was synthesized from 5-amino-2-(3,4-chlorophenyl)thiazolo[5,4-d]pyrimidine-7-thiol in 51 % yield.
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)thiazolo[5,4-d]pyrimidine-7-thiol (600 mg, 2.0 mmol), yielding the pure title compound (450 mg, 64%).
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)thiazolo[5, 4-d]pyrimidine-7-thiol (1.34 g, 4.0 mmol), yielding the pure title compound (1.1 g, 71%).
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine (35 mg, 0.1 mmol), yielding the pure title compound (49 mg, 92%).
- This compound was synthesized from 5-amino-7-( N -piperazino)-2-methylthio-thiazolo[5,4-d]pyrimidine (113 mg, 0.36 mmol), yielding the pure title compound (120 mg, 75%).
- This compound was synthesized from 5-amino-2-(3,4-dichlorophenyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine (190 mg, 0.5 mmol), yielding the pure title compound (150 mg, 55%).
- This compound was synthesized from 5-amino-2-(1-(4-chlorophenyl)cyclopropyl)-7-( N- piperazino)thiazolo[5,4-d]pyrimidine (39 mg, 0.1 mmol), yielding the pure title compound (50 mg, 89%).
- This compound was made from 5-amino-7-methylthio-2-(2-phenylethyl)thiazolo[5, 4-d]pyrimidine in 71% yield.
- This compound was made from 5-amino-2-cyclopropyl-7- N -piperazino-thiazolo[5, 4-d]pyrimidine (15 mg, 0.05 mmol), yielding the pure title compound (20 mg, 83%).
- This compound was made from 5-amino-2-cyclohexyl-7-methylthiothiazolo[5,4-d]pyrimidine (56 mg, 0.2 mmol), yielding the pure title compound (60 mg, 62%).
- Example 121 Synthesis of 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine and 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(N-oxopyridine-3-yl)thiazolo[5,4-d]pyrimidine
- Example 121a 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine (50 mg, 41%)
- Example 121b 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(N-oxopyridine-3-yl)thiazolo[5, 4-d]pyrimidine (20 mg, 17%).
- This compound was made from 5-amino-2-(4-chlorophenylmethyl)-7-methylthiothiazolo[5,4-d]pyrimidine (100 mg, 0.31 mmol), yielding the pure title compound (140 mg, 85%).
- This compound was made from 5-amino-2-(4-chlorophenyl)-7-methylthiothiazolo[5,4-d]pyrimidine (154 mg, 0.5 mmol), yielding the pure title compound (190 mg, 74%).
- This compound was made from 5-amino-2-(3-methoxyphenyl)-7-methylthiothiazolo[5,4-d]pyrimidine (152 mg, 0.5 mmol), yielding the pure title compound (120 mg, 47%).
- Example 140 Synthesis of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-thiazol-2-yl-piperazine-1-yl)-thiazolo[5,4-d]pyrimidine
- Example 150 Synthesis of 5-amino-2-[1-(4-fluorophenyl)propyl]-7-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- This compound was prepared from example 2 in a yield of 24%, according to the procedure for the synthesis of example 158.
- This compound was prepared from example 159 in a yield of 73%, according to the procedure for the synthesis of example 160.
- This compound was synthesized from 5-amino-2-cyclopropyl-7- N -piperazino-oxazolo[5, 4-d]pyrimidine in 93 % yield.
- This compound was synthesized from 5-amino-2-methoxymethyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 60 % yield.
- This compound was synthesized from 5-amino-2-cyclohexyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 73 % yield.
- This compound was synthesized from 5-amino-2-pentyl-7- N -piperazino-oxazolo[5,4-d]pyrimidine in 71 % yield.
- This compound was synthesized from 5-amino-2-pentyl-7- N -piperazino-oxazolo[5,4-d]pyrimidine in 85 % yield.
- Example 196 Synthesis of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4- m- tolylcarbamoylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Transplantation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
- The present invention relates to a class of novel thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives and a method for their preparation, as well as to pharmaceutical compositions comprising one or more of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives and one or more pharmaceutically acceptable excipients. The present invention further relates to the use of said novel thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives as biologically active ingredients, more specifically as medicaments for the treatment of disorders and pathologic conditions such as, but not limited to, immune and auto-immune disorders, organ and cells transplant rejections.
- Thiazolo[5,4-d]pyrimidines and oxazolo[5,4-d]pyrimidines can be considered as structural analogues of purines, in which the imidazole moiety is replaced by a 1,3-thiazole or 1,3-oxazole ring system. Although purine chemistry is extensively described in literature, the number of medicinal chemistry papers that describe the synthesis and biological evaluation of oxazolopyrimidines and thiazolopyrimidines is limited. Apparently, the oxazolopyrimidine and thiazolopyrimidine scaffold is not very frequently used in drug discovery programs. However, biological activities of certain thiazolo[5,4-d]pyrimidines and oxazolo[5,4-d]pyrimidines have been reported. 2,5-Diaminothiazolo[5,4-d]pyrimidin-7(6H)-one, a thio-isostere of 8-amino-guanine, was found to be a weak inhibitor of purine nucleoside phosphorylase (J. C. Sircar et al. J. Med. Chem. 1986, 29, 1804-1806). Thiazolo[5,4-d]pyrimidines were covered by several patent applications as activators of caspases and inducers of apoptosis (
WO2008/057402 ), anti-angiogenic agents (WO2004/01314 EP1731523 ), heat shock protein 90 inhibitors (WO2008/059368 ) and xanthine oxidase inhibitors (WO2007/004688 ).WO2008/152390 discloses thiazolo[5,4-d]pyrimidines and their use as inhibitors of phosphatidylinositol-3 kinase.WO 2008/005303 discloses vanilloid receptor 1 (TRPV1) modulating thiazolo[5,4-d]pyrimidine analogues and their use for the treatment of diseases, such as pain, arthritis, itch, cough, asthma, or inflammatory bowel disease. - 2-Aryloxazolo[5,4-d]pyrimidines have been described as adenosine kinase inhibitors (M. Bauser; et al. Bioorg. Med. Chem. Lett. 2004, 14, 1997-2000). 7-Amino-5-phenylethylamino-2-furyl-oxazolo[5,4-d]pyrimidines act as brain A2A adenosine receptor (A2AAR) antagonists (M. H. Holschbach, et al. Eur. J. Med. Chem. 2006, 41, 7-15). 7-(Substituted-cyclopentyl)aminooxazolo[5,4-d]pyrimidines have been reported to possess tumor growth inhibitory activity (
WO/2008/019124 ). However none of these documents teaches or suggests thiazolo[5,4-d]pyrimidine or oxazolo[5,4-d]pyrimidine derivatives having the substitution pattern disclosed by the present invention. - A huge number of thieno[2,3-d]pyrimidines is already known in the art.
WO 2007/102679 discloses thienopyrimidines with at position 4 a pyrrole-2,5-dione substituent which strongly inhibits IKB kinase-β (IKK-β) involved in the activation of a transcriptional factor, NF-κB, which is associated with inducing various immune and inflammatory diseases, whereby a composition comprising the compound is a useful therapeutic agent against inflammatory diseases, in particular, arthritis and cancer.WO 2007/084815 discloses 2-carboxamide substituted thieno(2,3-d)pyrimidines inhibitors of one or more of the EGFR, HER-2, c-Src, Lyn, c-Abl, Aurora-A or VEGF kinase proteins and the like possessing anti-tumor cell proliferation activity, and as such are useful in treating or ameliorating a EGFR, HER-2, c-Src, Lyn, c-Abl, Aurora-A or VEGF kinase receptor mediated, angiogenesis-mediated or hyperproliferative disorder. -
WO 2006/071988 discloses certain 4,5-disubstituted thienopyrimidine derivatives which are useful for the inhibition of PDE10 enzymes, and thus are useful for treating psychiatric or neurological syndromes, such as psychoses, obsessive-compulsive disorder and/or Parkinson's disease.WO 2004/111057 discloses compounds which are particularly useful for inhibiting potassium channels Kvl. 5), which are known targets for the treatment of cardiac arrhythmia in the atria such as atrial fibrillation. However, none of these documents teaches or suggests thieno(2,3-d)pyrimidine derivatives having the substitution pattern disclosed by the present invention. - Marketed drugs with a purine based skeleton are known. Examples include theophylline (drug for the treatment of asthma) and azathioprine (drug for the treatment of transplant rejection). Anti-cancer drugs with a purine scaffold include 6-mercaptoguanine and thioguanine. Purines are also an important constituent of antiviral nucleosides such as acyclovir (used for the treatment of herpes virus infections) and ganciclovir (medication used for treatment of cytomegalovirus infections). Abacavir and dideoxyadenosine (ddA) are both purine nucleosides acting as reverse transcriptase inhibitors and both compounds are licensed as anti-HIV agents.
- Purines display a broad range of biological activities and as a result a huge number of purine analogues is already known in the art.
WO 2009/005687 discloses purine derivatives and their use as modulators of Toll-like receptor 7. Compounds and pharmaceutical compositions which selectively activate toll-like receptor 7 are useful for treating viral infections in patients.WO 2008/135232 relates to substituted purines and purine derivatives as inhibitors of Aurora A, Aurora B, Aurora C, CHK2, JNK1 α1, JNK3 and abl kinase. These compounds possess antiproliferative properties and are useful in the treatment of proliferative disorders such as cancer, leukemia, psoriasis and the like.WO 2008/094737 discloses purine derivatives as inhibitors of calcium dependent protein kinase 1 (PfCDPKI). These purines are useful for treating malaria.WO 2008/090181 relates to a new series of purine derivatives as inhibitors of Janus kinases. JAK3 kinase inhibitors have been recognized as a new class of effective immunosuppressive agents useful for transplant rejection prevention and in the prevention or treatment of immune, autoimmune, inflammatory and proliferative diseases such as psoriasis, psoriatic arthritis, rheumatoid arthritis, multiple sclerosis, inflammatory bowel diseases, systemic lupus erythematosus, type I diabetes and complications from diabetes, allergic reactions and leukemia.WO 2008/060301 also discloses 7-substituted purine derivatives as immunosuppressive drugs useful for treatment of an autoimmune disease, an inflammatory disease, a mast cell mediated disease, hematological malignancy and organ transplant rejection. However none of these documents teaches or suggests purine derivatives having the substitution pattern disclosed by the present invention. - However there is a continuous need in the art for specific and highly therapeutically active compounds, such as, but not limited to, drugs for treating immune and autoimmune disorders, organ and cells transplant rejections. In particular, there is a need in the art to provide immunosuppressive compounds, which are active in a minor dose in order to replace existing drugs having significant side effects and to decrease treatment costs.
- Currently used immunosuppressive drugs include antiproliferative agents, such as methotrexate (a 2,4-diaminopyrido(3,2-d)pyrimidine derivative disclosed by
U.S. Patent No. 2,512,572 ), azathioprine, and cyclophosphamide. Since these drugs affect mitosis and cell division, they have severe toxic effects on normal cells with high turn-over rate such as bone marrow cells and the gastrointestinal tract lining. Accordingly, marrow depression and liver damage are common side effects of these antiproliferative drugs. - Anti-inflammatory compounds used to induce immunosuppression include adrenocortical steroids such as dexamethasone and prednisolone. The common side effects observed with the use of these compounds are frequent infections, abnormal metabolism, hypertension, and diabetes.
- Other immunosuppressive compounds currently used to inhibit lymphocyte activation and subsequent proliferation include cyclosporine, tacrolimus and rapamycin. Cyclosporine and its relatives are among the most commonly used immunosuppressant drugs. Cyclosporine is typically used for preventing or treating organ rejection in kidney, liver, heart, pancreas, bone marrow, and heart-lung transplants, as well as for the treatment of autoimmune and inflammatory diseases such as Crohn's disease, aplastic anemia, multiple-sclerosis, myasthenia gravis, uveitis, biliary cirrhosis, etc. However, cyclosporines suffer from a small therapeutic dose window and severe toxic effects including nephrotoxicity, hepatotoxicity, hypertension, hirsutism, cancer, and neurotoxicity.
- Additionally, monoclonal antibodies with immunosuppressant properties, such as OKT3, have been used to prevent and/or treat graft rejection. Introduction of such monoclonal antibodies into a patient, as with many biological materials, induces several side-effects, such as dyspnea. Within the context of many life-threatening diseases, organ transplantation is considered a standard treatment and, in many cases, the only alternative to death. The immune response to foreign cell surface antigens on the graft, encoded by the major histo-compatibility complex (hereinafter referred as MHC) and present on all cells, generally precludes successful transplantation of tissues and organs unless the transplant tissues come from a compatible donor and the normal immune response is suppressed. Other than identical twins, the best compatibility and thus, long term rates of engraftment, are achieved using MHC identical sibling donors or MHC identical unrelated cadaver donors. However, such ideal matches are difficult to achieve. Further, with the increasing need of donor organs an increasing shortage of transplanted organs currently exists. Accordingly, xenotransplantation has emerged as an area of intensive study, but faces many hurdles with regard to rejection within the recipient organism.
- The host response to an organ allograft involves a complex series of cellular interactions among T and B lymphocytes as well as macrophages or dendritic cells that recognize and are activated by foreign antigen. Co-stimulatory factors, primarily cytokines, and specific cell-cell interactions, provided by activated accessory cells such as macrophages or dendritic cells are essential for T-cell proliferation. These macrophages and dendritic cells either directly adhere to T-cells through specific adhesion proteins or secrete cytokines that stimulate T-cells, such as IL-12 and IL-15. Accessory cell-derived co-stimulatory signals stimulate activation of interleukin-2 (IL- 2) gene transcription and expression of high affinity IL-2 receptors in T-cells. IL-2 is secreted by T lymphocytes upon antigen stimulation and is required for normal immune responsiveness. IL-2 stimulates lymphoid cells to proliferate and differentiate by binding to IL-2 specific cell surface receptors (IL-2R). IL-2 also initiates helper T-cell activation of cytotoxic T-cells and stimulates secretion of interferon-γ which in turn activates cytodestructive properties of macrophages. Furthermore, IFN-γ and IL-4 are also important activators of MHC class II expression in the transplanted organ, thereby further expanding the rejection cascade by enhancing the immunogenicity of the grafted organ. The current model of a T-cell mediated response suggests that T-cells are primed in the T-cell zone of secondary lymphoid organs, primarily by dendritic cells. The initial interaction requires cell to cell contact between antigen-loaded MHC molecules on antigen-presenting cells (hereinafter referred as APC) and the T-cell receptor/CD3 complex on T-cells. Engagement of the TCR/CD3 complex induces CD154 expression predominantly on CD4 T-cells that in turn activate the APC through CD40 engagement, leading to improved antigen presentation. This is caused partly by upregulation of CD80 and CD86 expression on the APC, both of which are ligands for the important CD28 co-stimulatory molecule on T-cells. However, engagement of CD40 also leads to prolonged surface expression of MHC-antigen complexes, expression of ligands for 4-1 BB and OX-40 (potent co-stimulatory molecules expressed on activated T-cells). Furthermore, CD40 engagement leads to secretion of various cytokines (e.g., IL-12, IL-15, TNF-α, IL-1, IL-6, and IL-8) and chemokines, all of which have important effects on both APC and T-cell activation and maturation. Similar mechanisms are involved in the development of auto-immune disease, such as type I diabetes. In humans and non-obese diabetic mice, insulin- dependent diabetes mellitus results from a spontaneous T-cell dependent autoimmune destruction of insulin-producing pancreatic beta, cells that intensifies with age. The process is preceded by infiltration of the islets with mononuclear cells (insulitis), primarily composed of T lymphocytes. A delicate balance between auto-aggressive T-cells and suppressor-type immune phenomena determines whether expression of auto-immunity is limited to insulitis or not. Therapeutic strategies that target T-cells have been successful in preventing further progress of the autoimmune disease. These include neonatal thymectomy, administration of cyclosporine, and infusion of anti-pan T-cell, anti-CD4, or anti-CD25 (IL-2R) monoclonal antibodies. The aim of all rejection prevention and auto-immunity reversal strategies is to suppress the patient's immune reactivity to the antigenic tissue or agent, with a minimum of morbidity and mortality. Accordingly, a number of drugs are currently being used or investigated for their immunosuppressive properties. As discussed above, the most commonly used immunosuppressant is cyclosporine, which however has numerous side effects. Accordingly, in view of the relatively few choices for agents effective at immunosuppression with low toxicity profiles and manageable side effects, there exists a need in the art for identification of alternative immunosuppressive agents and for agents acting as complement to calcineurin inhibition.
- The present invention is based on the unexpected finding that certain combinations of substituents at different positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine ring system, said combinations not being suggested by the prior art, are able to meet one or more of the medical needs recited herein above and to show unexpected biological properties, in particular have significant immunosuppressive activity.
-
- X is S or O;
- R1 is selected from the group consisting of halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7 alkyloxy;
- R3 is selected from the group consisting of (mono- or di-) C1-12 alkylamino; monoarylamino; diarylamino; (mono- or di-) C3-10 cycloalkylamino; (mono- or di-) hydroxy C1-7 alkylamino; (mono- or di-) C1-4 alkylarylamino; (mono- or di-) arylC1-4 alkylamino; morpholinyl; mercapto C1-7 alkyl; C1-7 alkoxy, aralkylthio, piperidinyl, pyrrolidinyl, homopiperazinyl and piperazinyl, wherein said piperidinyl, pyrrolidinyl, homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R20 selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl, wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino; and
- R2 is selected from the group consisting of heteroaryl and aryl groups; halogen; C1-7 alkyl; C2-7 alkenyl; C2-7 alkynyl; halo C1-7 alkyl; C3-10 cycloalkyl; carboxy C1-7 alkyl; carboxyaryl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; oxyheterocyclic; heterocyclic-substituted alkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; thio-acylamino; alkoxyamino; thioalkyl-amino; acetal; thio-acetal; carboxylic acid; carboxylic acid esters, thiocarboxylic acid; thiocarboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; hydroxyl; sulfhydryl; nitro; cyano; carbamoyl; thiocarbamoyl; ureido; thioureido; amino; alkylamino; cycloalkylamino; alkenylamino; cyclo-alkenylamino; alkynylamino; arylamino; arylalkylamino; hydroxyalkylamino; mercaptoalkyl-amino; heterocyclic amino; heterocyclic substituted arylamino; heterocyclic-substituted alkyl-amino; oximino; alkyloximino; hydrazino; alkylhydrazino; phenylhydrazino; esters, thioesters, halides, anhydrides, amides and thioamides thereof; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine ring and the aromatic or heterocyclic substituent, wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino; wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chains of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, thiol, ether, thio-ether, acetal, thio-acetal, amino, imino, oximino, alkyloximino, aminoacid, cyano, acylamino, thioacylamino, carbamoyl, thiocarbamoyl, ureido, thio-ureido, carboxylic acid ester or halide or anhydride or amide, thiocarboxylic acid or ester or thioester or halide or anhydride or amide, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl, hydroxylamino, mercaptoamino, alkyl-amino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, hetero-cyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl, sulfinyl and sulfonamido;
- R4 is selected from the group consisting of halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7alkyloxy;
- R6 is selected from the group consisting of (mono- or di-) C1-12 alkylamino, monoarylamino, diarylamino, (mono- or di-) C3-10 cycloalkylamino, (mono- or di-) hydroxy C1-7 alkylamino, (mono- or di-) C1-4 alkylarylamino, (mono- or di-) arylC1-4 alkylamino, morpholinyl, mercapto C1-7 alkyl, C1-7 alkoxy, homopiperazinyl and piperazinyl, wherein said homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R21 selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl; wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino;
- R5 is selected from the group consisting of heteroaryl and aryl groups, wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino; wherein R12 is represented by the general formula V :
- each substituent R13 of the heterocyclic ring is a group independently selected from the group consisting of halogen, nitro, C1-7 alkyl (optionally containing one or more functions or radicals selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, sulfhydryl, C1-7 alkoxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, acetal, thioacetal, imino, oximino, alkyloximino, amino-acid, cyano, (thio)carboxylic acid, (thio)carboxylic acid ester or amide, nitro, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenyl-amino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercapto-alkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl and sulfonamido), C3-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl, alkylaryl, alkylacyl, arylacyl, hydroxyl, sulfhydryl, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thioheterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, hydroxylamino, cyano, (thio)carboxylic acid or esters or thioesters or amides or thioamides thereof;
- n is an integer from 0 to 6; for example n is an integer selected from 0, 1, 3, 4 or 5, preferably, n is 0, 1, 2, or 3, more preferably, n is 0 or 1, yet more preferably n is 0;
- R10 is selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl; wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino; and
- R11 is selected from the group consisting of halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, C1-7 alkyl, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7 alkyloxy;
- R8 is selected from the group consisting of heteroaryl and aryl groups; halogen; C1-7 alkyl; C2-7 alkenyl; C2-7 alkynyl; halo C1-7 alkyl; carboxy C1-7 alkyl; carboxyaryl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; oxyheterocyclic; heterocyclic-substituted alkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; acylamino; thio-acylamino; alkoxyamino; thioalkyl-amino; acetal; thio-acetal; carboxylic acid; carboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; thiocarboxylic acid; thiocarboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; hydroxyl; sulfhydryl; nitro; cyano; carbamoyl; thiocarbamoyl; ureido; thioureido; amino; alkylamino; cycloalkylamino; alkenylamino; cyclo-alkenylamino; alkynylamino; arylamino; arylalkylamino; hydroxyalkylamino; mercaptoalkyl-amino; heterocyclic amino; heterocyclic substituted arylamino; heterocyclic-substituted alkyl-amino; oximino; alkyloximino; hydrazino; alkylhydrazino; phenylhydrazino; esters, thioesters, halides, anhydrides, amides and thioamides thereof; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the purine ring and the aromatic or heterocyclic substituent; wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino; and wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chain of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, thiol, ether, thio-ether, acetal, thio-acetal, amino, imino, oximino, alkyloximino, aminoacid, cyano, acylamino, thioacylamino, carbamoyl, thiocarbamoyl, ureido, thio-ureido, carboxylic acid ester or halide or anhydride or amide, thiocarboxylic acid or ester or thioester or halide or anhydride or amide, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl, hydroxylamino, mercaptoamino, alkyl-amino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, hetero-cyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl, sulfinyl and sulfonamido;
- R7 and R9 are selected from the group consisting of hydrogen, C3-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl, alkylaryl, acyl sulfonyl and C1-7 alkyl , wherein said C1-7 alkyl is optionally containing one or more functions or radicals selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, sulfhydryl, C1-7 alkoxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, acetal, thioacetal, imino, oximino, alkyloximino, amino-acid, cyano, (thio)carboxylic acid, (thio)carboxylic acid ester or amide, nitro, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenyl-amino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercapto-alkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl and sulfonamido),
wherein acyl group refers to a carbonyl group adjacent to a C1-7 alkyl, a C3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, or is selected from the group comprising alkanoyl, cycloalkanoyl, cycloalkyl-alkanoyl, alkenoyl, alkylthioalkanoyl, alkanesulfonyl, alkoxycarbonyl, alkylcarbamoyl, alkylcarbamidoyl, alkoxalyl, aroyl, aralkanoyl, aralkenoyl, aryloxyalkanoyl, arylthioalkanoyl, arylaminoalkanoyl, aryloxycarbonyl, aralkoxycarbonyl, arylcarbamoyl, arylglyoxyloyl, arylthiocarbamoyl, arylcarbamidoyl, heterocyclic-carbonyl, heterocyclic-alkanoyl, wherein said heterocyclic group is an aromatic or non-aromatic 5- to 7-membered heterocyclic ring with one or more heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur in said ring;
and/or a pharmaceutical acceptable addition salt thereof and/or a stereoisomer thereof and/or a solvate thereof. - The present invention also concerns a compound having formula I, II, III or IV, for use as a medicine.
- The present invention also concerns a compound having formula I, II, III or IV, for use as a medicine for the prevention or treatment of immune disorders in an animal, preferably in a mammal. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said mammal is a human being.
- The present invention also concerns a pharmaceutical composition comprising a therapeutically effective amount of a compound having formula I, II, III or IV, and one or more pharmaceutically acceptable exipients. Said composition may further comprise one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- The present invention also concerns a method of prevention or treatment of an immune disorder in an animal, comprising the administration of a therapeutically effective amount of a compound having formula I, II, III or IV, optionally in combination with one or more pharmaceutically acceptable excipients.
- The present invention also concerns a process for preparation of the thiazolo(5,4-d)pyrimidine derivatives of formula I, wherein X=S, and comprising the steps of: (a) acylation of 2,5-diamino-4,6-dihydroxypyrimidine; (b) treatment with a thionation reagent; (c) treatment with iodomethane; (d) oxidation reaction by adding an oxidating agent; and (e) a nucleophilic aromatic substitution reaction. In an embodiment, in step (a) said acylation is performed with a carboxylic acid (R2COOH) or an acid chloride (R2C(O)Cl). In an embodiment, step (a) further comprises the addition of a coupling reagent. In an embodiment, said coupling reagent is N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI). In another embodiment, step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt). In an embodiment, in step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent. In an embodiment, in step (b) said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene. In an embodiment, step (c) is performed in alkaline conditions. In an embodiment, in step (d) said oxidating agent is m-chloro-peroxybenzoic acid or hydrogen peroxide. In an embodiment, in step (e) a piperazine is introduced at position 7. In an embodiment, the thiazolo(5,4-d)pyrimidine derivatives of formula I are R2-substituted 5-amino-7-N-piperazino thiazolo(5,4-d)pyrimidine derivatives.
- The present invention will now be further described. In the following passages, different aspects of the invention are defined in more detail. Each aspect so defined may be combined with any other aspect or aspects unless clearly indicated to the contrary. In particular, any feature indicated as being preferred or advantageous may be combined with any other feature or features indicated as being preferred or advantageous.
- One embodiment of the present invention concerns a compound according to the invention wherein , R3 and R2 have any of the values as described herein and R1 is selected from the group comprising aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino; preferably, said R1 is selected from the group comprising amino, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) C1-4 alkyl-arylamino; preferably, said R1 is selected from the group comprising amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino; preferably, said R1 is selected from the group comprising amino, and (mono- or di) C1-7 alkylamino; more preferably R1 is amino.
- In an embodiment, the present invention concern a compound of formula I, II, III or IV, a pharmaceutical acceptable addition salt thereof and/or a stereoisomer thereof and/or a solvate thereof, wherein R1, R4, and R11 are each independently selected from the group consisting of amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino.
- One embodiment of the present invention concerns a compound according to the invention wherein X is S. Another embodiment of the present invention concerns a compound according to the invention wherein X is O.
- One embodiment of the present invention concerns a compound according to the invention wherein R1 and R2 have any of the values as described herein and R3 is selected from the group consisting of monoarylamino; diarylamino; (mono- or di-) arylC1-4 alkylamino; morpholinyl; C1-7 alkoxy, aralkylthio, piperidinyl, pyrrolidinyl, homopiperazinyl and piperazinyl, wherein said piperidinyl, pyrrolidinyl, homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R20, wherein R20 has the same meaning as defined herein. One embodiment of the present invention concerns a compound according to the invention wherein R4 and R5 have any of the values as described herein and R6 is selected from the group consisting of (mono- or di-) C1-12 alkylamino, monoarylamino, diarylamino, (mono- or di-) C3-10 cycloalkylamino, (mono- or di-) arylC1-4 alkylamino, morpholinyl, C1-7 alkoxy, homopiperazinyl and piperazinyl, wherein said homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R21, wherein R21 has the same meaning as that defined in herein.
- One embodiment of the present invention concerns a compound according to the invention wherein R11, R7, R8 and R9 have any of the values as described herein and n is 0 and R10 is selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-carboxylic ester-alkyl, aryloxyalkyl, arylalkyl and aryl; wherein the aryl moiety of each of said aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, formyl, carbamoyl, thiocarbamoyl, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, cyano, alkylamino, cycloalkylamino.
- One embodiment of the present invention concerns a compound according to the invention wherein R1 and R3 have any of the values as described herein R2 is selected from the group consisting of heteroaryl and aryl groups; C1-7 alkyl; C3-10 cycloalkyl; halo C1-7 alkyl; carboxy C1-7 alkyl; carboxyaryl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; heterocyclic-substituted alkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; acylamino; thio-acylamino; alkoxyamino; carbamoyl; thiocarbamoyl; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine ring and the aromatic or heterocyclic substituent, wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, carbamoyl, thiocarbamoyl, sulfonamido, hydroxylamino, alkoxy-amino, acylamino, thioacylamino, cyano, wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chains of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, thiol, ether, thio-ether, amino, cyano, acylamino, nitro, thio C1-7 alkyl. One embodiment of the present invention concerns a compound according to the invention wherein R4 and R6 have any of the values as described herein and R5 is an aryl group, wherein said aryl group is optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, C1-7 alkoxy, thio C1-7 alkyl, cyano.
- One embodiment of the present invention concerns a compound according to the invention wherein R11, R7, R12 and R9 have any of the values as described herein, and R8 is selected from the group consisting of heteroaryl and aryl groups; C1-7 alkyl; halo C1-7 alkyl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the purine ring and the aromatic or heterocyclic substituent; wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, thio C1-7 alkyl, and wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chain of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, hydroxyl, thiol, cyano, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R3, R6 are each independently homopiperazinyl or piperazinyl, wherein said homopiperazinyl or piperazinyl is each respectively optionally N-substituted with a substituent R20, R21, wherein R20 and R21 have the same meaning as that defined herein.
- One embodiment of the present invention concerns a compound according to the invention wherein R1, R4 and R11 are each independently amino.
- One embodiment of the present invention concerns a compound according to the invention wherein R1 and R3 have any of the values as described herein and R2 is selected from the group consisting of phenyl; pyridin-3-yl; pyridin-2-yl; pyridin-4-yl; 4-fluorophenethyl; 4-fluorophenyl; 4-bromophenethyl; pentyl; tolyl; (4-fluorophenyl)butyl; (4-fluorophenyl)propyl; 4-chlorophenyl; 4-methylphenethyl; 3,4-dimethoxyphenethyl; 3-methoxyphenethyl; furan-2-yl; 2-phenylethyl; cyclohexyl; methoxylmethyl; cyclopropyl; 2-thiophen-2-ylethyl; cyclopentyl-(4-fluorophenyl)methyl; 1-(4-fluorophenyl)propyl; 4-fluorophenylamino; methylsulfinyl; 1-(4-chlorophenyl)ethyl; 3-methoxyphenyl; 4-chlorophenyl; 4-chlorophenylmethyl; N-oxopyridine-3-yl; 1-(4-chlorophenyl)cyclopropyl; 3,4-dichlorophenyl; methylthio; 1-phenylcyclopropyl; 1-(4-fluorophenyl)ethyl; 1-(4-fluorophenyl)-2-phenylethyl; 2-(4-fluorophenoxy)ethyl; morpholino; preferably 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R4 and R6 have any of the values as described herein and R5 is phenyl or 4-fluorophenyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R1 and R2 have any of the values as described herein and R3 is selected from the group consisting of p-tolyl piperazinyl-1-carboxylate; N-methyl-N-p-tolylpiperazinyl-1-carboxamide; N-p-tolylpiperazinyl-1-carbothioamide; -N-hexylpiperazinyl-1-carboxamide; 4-(N-4-fluorophenylcarboxamide)piperazin-1-yl; N-cyclohexylpiperazinyl-1-carboxamide; N-phenylpiperazinyl-1-carboxamide; N-(4-(trifluoromethyl)phenyl)piperazinyl-1-carboxamide, piperazin-1-yl-2-(4-chlorophenoxy)ethanone; piperazin-1-yl-2-(4-methoxyphenoxy)ethanone; benzylsulfonylpiperazin-1-yl; N-(4-cyanophenyl)piperazinyl-1-carboxamide; N-(4-methoxybenzyl)piperazinyl-1-carboxamide; N-(4-chlorophenyl)piperazinyl-1-carboxamide; N-m-tolylpiperazinyl-1-carboxamide; piperazin-1-yl-2-(m-tolyloxy)ethanone; piperazin-1-yl-2-(4-chlorophenoxy)-2-methylpropan-1-one; piperazin-1-yl-2-(4-trifluoromethoxyphenoxy)ethanone;piperazin-1-yl-2-(4-fluorophenoxy)ethanone; piperazin-1-yl-2-(4-bromophenoxy)ethanone; piperazin-1-yl-3-(4-fluorophenyl)propan-1-one; piperazin-1-yl-2-(3-chlorophenoxy)ethanone; 4-acetylphenoxy-piperazin-1-yl-ethanone; piperazin-1-yl-2-oxoethoxy)benzoate; piperazin-1-yl-2-(4-hydroxyphenoxy)ethanone; piperazin-1-yl-3-(4-bromophenyl)propan-1-one; N-(2-methoxyphenyl)piperazinyl-carboxamide; N-(4-bromophenyl)piperazinyl-carboxamide; N-(2,4-difluorophenyl)piperazinyl-carboxamide; piperazin-1-yl-2-(4-chloro-2-methylphenoxy)ethanone; piperazin-1-yl-2-(2,4-dichlorophenoxy)ethanone; (methylphenyl-carbamoyl)methyl]piperazin-1-yl; phenoxyethyl)piperazin-1-yl; (4-chlorophenyl)acetyl]-piperazin-1-yl; (4-chlorophenyl)acetyl]-piperazin-1-yl; [2-(3-nitrophenoxy)acetyl]-piperazin-1-yl; 4-(2-methoxyethyl)-piperazin-1-yl; 4-acetylpiperazin-1-yl; 4-isobutylpiperazin-1-yl; 3-chloro-4-fluorophenyl-amino; 4-(2-phenoxyethyl)piperazin-1-yl; 4-benzoylpiperidine-1-yl; 4-chlorophenoxyacetyl)pyrrolidin-3-(S)-ylamino; 1-tert-butoxycarbonylpyrrolidin-3-(S)-ylamino; 1-benzyloxycarbonylpiperidin-3-ylamino; 3-(R)-(4-chlorobenzoylamino)-pyrrolidin-1-yl; 3-(R)-[2-(4-chlorophenoxy)-acetylamino]pyrrolidin-1-yl; 3-(R)-tert-butoxycarbonylamino; 4-(phenethylcarbamoyl-methyl)piperazin-1-yl; 4-thiazol-2-yl-piperazine-1-yl; 4-[(methylphenylcarbamoyl)-methyl]piperazin-1-yl; 4-chlorophenoxy)acetyl]homopiperazin-1-yl; 4-phenylmethanesulfonylpiperazin-1-yl; 4-(3-phenylpropionyl)piperazin-1-yl; 4-[2-phenoxyacetyl]piperazin-1-yl; 4-[2-(4-chlorophenyl)acetyl]piperazin-1-yl; 4-[2-(3-nitrophenoxy)acetyl]piperazin-1-yl; 4-(phenylsulfonyl)piperazin-1-yl; pyrimidin-7-yl-piperazinyl-1-carboxylate; 4-benzylpiperazin-1-yl; piperazin-1-yl-1-morpholinoethanone; 4-pentylpiperazin-1-yl; 4-(thiazol-2-yl)piperazin-1-yl; 4-m-tolylpiperazin-1-yl; 3-methoxypropylamino; ethoxy; 2-methoxyethoxy; benzylthio; benzyl amino; preferably methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-yl, 4-(4-chlorophenoxyacetyl)piperazin-1-yl, 4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl, 4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl, and 4-(2-phenoxyacetyl)-piperazin-1-yl.
- One embodiment of the present invention concerns a compound according to the invention wherein R2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl; R1 and is amino; and R3 is selected from the group consisting of methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-yl, 4-(4-chlorophenoxyacetyl)piperazin-1-yl, 4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl, 4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl, and 4-(2-phenoxyacetyl)-piperazin-1-yl.
- One embodiment of the present invention concerns a compound according to the invention wherein , R4 and R6 have any of the values as described herein and R5 is phenyl or 4-fluorophenyl.
- One embodiment of the present invention concerns a compound according to the invention wherein , R4 and R5 have any of the values as described herein and R6 is 4(m-tolylcarbamoyl)piperazin-1-yl or 4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl.
- One embodiment of the present invention concerns a compound according to the invention wherein R7, R8, R9, and R11 have any of the values as described herein and R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl.
- One embodiment of the present invention concerns a compound according to the invention wherein , R5 is phenyl or 4-fluorophenyl; R4 is amino; and R6 is (4(m-tolylcarbamoyl)piperazin-1-yl or (4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl).
- One embodiment of the present invention concerns a compound according to the invention wherein , R7, R12, R9, and R11 have any of the values as described herein and R8 is 4-fluorophenyl or methylthio.
- One embodiment of the present invention concerns a compound according to the invention wherein R12, R8, and R11 have any of the values as described herein R9 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R8, R12 , and R11 have any of the values as described herein R7 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R11 is amino, R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or (4-phenoxyacetyl)piperazin-1-yl, R8 is 4-fluorophenyl or methylthio, and R9 is hydrogen or methyl.
- One embodiment of the present invention concerns a compound according to the invention wherein R11 is amino, R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl, R8 is 4-fluorophenyl or methylthio, and R7 is hydrogen or methyl.
- In an embodiment, the present invention encompasses a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative having the general formula I :wherein - X is S or O;
- R1 is selected from the group consisting of hydrogen, halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, C1-7 alkyl, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7 alkyloxy;
- R3 is selected from the group consisting of (mono- or di-) C1-12 alkylamino; monoarylamino; diarylamino; (mono- or di-) C3-10 cycloalkylamino; (mono- or di-) hydroxy C1-7 alkylamino; (mono- or di-) C1-4 alkylarylamino; (mono- or di-) arylC1-4 alkylamino; morpholinyl; mercapto C1-7 alkyl; C1-7 alkoxy, homopiperazinyl and piperazinyl,
wherein said homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R20 selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl,
wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino; and - R2 is selected from the group consisting of heteroaryl and aryl groups; hydrogen; halogen; C1-7 alkyl; C2-7 alkenyl; C2-7 alkynyl; halo C1-7 alkyl; carboxy C1-7 alkyl; carboxyaryl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; oxyheterocyclic; heterocyclic-substituted alkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; acylamino; thio-acylamino; alkoxyamino; thioalkyl-amino; acetal; thio-acetal; carboxylic acid; carboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; thiocarboxylic acid; thiocarboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; hydroxyl; sulfhydryl; nitro; cyano; carbamoyl; thiocarbamoyl; ureido; thioureido; amino; alkylamino; cycloalkylamino; alkenylamino; cyclo-alkenylamino; alkynylamino; arylamino; arylalkylamino; hydroxyalkylamino; mercaptoalkyl-amino; heterocyclic amino; heterocyclic substituted arylamino; heterocyclic-substituted alkyl-amino; oximino; alkyloximino; hydrazino; alkylhydrazino; phenylhydrazino; esters, thioesters, halides, anhydrides, amides and thioamides thereof ; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine ring and the aromatic or heterocyclic substituent,
- wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino; and
- wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chains of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, thiol, ether, thio-ether, acetal, thio-acetal, amino, imino, oximino, alkyloximino, aminoacid, cyano, acylamino, thioacylamino, carbamoyl, thiocarbamoyl, ureido, thio-ureido, carboxylic acid ester or halide or anhydride or amide, thiocarboxylic acid or ester or thioester or halide or anhydride or amide, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl, hydroxylamino, mercaptoamino, alkyl-amino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, hetero-cyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl, sulfinyl and sulfonamido; and/or
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R2 and R3 have any of the values as described herein and R1 is selected from the group consisting of amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R2 and R1 have any of the values as described herein and R3 is homopiperazinyl or piperazinyl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R1 and R3 have any of the values as described herein and R2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R2 and R3 have any of the values as described herein and R1 is selected from the group consisting of amino, methyl, and hydrogen.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R2 and R1 have any of the values as described herein and R3 is selected from the group consisting of methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-yl, 4-(4-chlorophenoxyacetyl)piperazin-1-yl, 4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl, 4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl, and 4-(2-phenoxyacetyl)-piperazin-1-yl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein R2 is selected from the group consisting of 4-fluorophenyl, 4-fluorobenzyl, 4-fluorophenethyl, 2-(4-fluorophenoxy)ethyl, 1-(4-fluorophenyl)ethyl, 1-(4-fluorophenyl)-2-phenylethyl, 3-pyridyl, 3-methoxyphenyl, cyclopropyl, cyclohexyl, 3,4-dichlorophenyl, 1-phenylcyclopropyl, 1-(4-chlorophenyl)cyclopropyl, 2-thiophen-2-ylethyl, pentyl, and 2-phenylethyl; R5 is selected from the group consisting of amino, methyl, and hydrogen; and R7 is selected from the group consisting of methylthio, piperazin-1-yl, ethoxy, morpholino, 4-m-tolylcarbamoyl-piperazin-1-yl, 4-(4-chlorophenoxyacetyl)piperazin-1-yl, 4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl, 4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl, and 4-(2-phenoxyacetyl)-piperazin-1-yl.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein X is S. One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, wherein X is O.
- One embodiment of the present invention concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 2-(4-fluorophenyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorobenzyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenethyl)-7-methylthio-thiazolo[5,4-d]pyrimidin-5-amine; 2-(2-(4-fluorophenoxy)ethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-5-methyl-7-methylthio-thiazolo[5,4-d]pyrimidine; 2-(4-fluorophenyl)-7-(piperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorobenzyl)-7-(piperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine; 7-(benzylthio)-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-7-(2-methoxyethoxy)-thiazolo[5,4-d]pyrimidin-5-amine; 7-ethoxy-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-5-amine; 7-ethoxy-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-N-7-(3-methoxypropyl)thiazolo[5,4-d]pyrimidine-5,7-diamine; 2-(4-fluorophenyl)-7-morpholino-thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorobenzyl)-7-morpholino-thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-7-(4-m-tolylpiperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-7-(4-(thiazol-2-yl)piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-fluorophenyl)-7-(4-pentylpiperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine; 2-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-1-morpholinoethanone; 7-(4-benzylpiperazin-1-yl)-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-5-amine; benzyl-4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazine-1-carboxylate; 2-(4-fluorophenyl)-7-(4-(phenylsulfonyl)piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine; 4-(5-amino-2-(4-fluorophenyl)-thiazolo[5,4-d]pyrimidin-7-yl)-N-p-tolylpiperazine-1-carboxamide; 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide; 4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide; 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone;2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)-5-methylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone; 1-(4-(5-amino-2-(2-(4-fluorophenoxy)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)-thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(2,4-dichlorophenoxy)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-3-(4-fluorophenoxy)propan-1-one; 2-(1-(4-fluorophenyl)ethyl)-7-methylthio-thiazolo[5,4-d]pyrimidin-5-amine; 2-(1-(4-fluorophenyl)-2-phenylethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine; 1-(4-(5-amino-2-(1-(4-fluorophenyl)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(5-amino-2-(1-(4-fluorophenyl)-2-phenylethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 4-(5-amino-2-(1-(4-fluorophenyl)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide; 2-(4-fluorophenyl)-5,7-bis(methylthio)thiazolo[5,4-d]pyrimidine; 5,7-bis(butylthio)-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidine; 5,7-bis(benzylthio)-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidine; 7-ethoxy-2-(4-fluorophenyl)-5-(methylthio)thiazolo[5,4-d]pyrimidine; 5-amino-2-cyclopropyl-7-methylthio-thiazolo[5,4-d]pyrimidine; 5-amino-2-(2-phenylethyl)-7-methylthiothiazolo[5,4-d]pyrimidine; 5-amino-2-cyclohexyl-7-methylthiothiazolo[5,4-d]pyrimidine; 5-amino-2-(3-pyridinyl)-7-methylthiothiazolo[5,4-d]pyrimidine; 5-amino-2-(4-chlorobenzyl)-7-methylthiothiazolo[5,4-d]pyrimidine; 5-amino-2-(3-methoxyphenyl)-7-methylthiothiazolo[5,4-d]pyrimidine; 5-amino-2-(4-chlorophenyl)-7-(methylthio)thiazolo[5,4-d]pyrimidine; 5-amino-2-cyclopropyl-7-methoxythiazolo[5,4-d]pyrimidine; 5-amino-2-cyclopropyl-7-N-piperazino-thiazolo[5, 4-d]pyrimidine; 5-amino-2-(3,4-dichlorophenyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine; 5-amino-2-(1-phenylcyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine; 5-amino-2-(1-(4-chlorophenyl)cyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine; 5-amino-7-N-piperazino-2-methylthio-thiazolo[5, 4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-phenylcyclopropyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methylthio-thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(3,4-dichlorophenyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-(4-chlorophenyl)cyclopropyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(2-phenylethyl)thiazolo[5, 4-d]pyrimidine; 5-amino-2-cyclopropyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-cyclohexylthiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(N-oxopyridine-3-yl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-chlorophenylmethyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-chlorophenyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(3-methoxyphenyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-(4-chlorophenyl)ethyl)thiazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-fluorophenylamino)-thiazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-(2-phenoxyacetyl)-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(3-nitrophenoxy)acetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenyl)acetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-m-tolylcarbamoylpiperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-phenoxyacetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4-(4-chlorobenzoyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4-(3-phenylpropionyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4- phenylmethanesulfonylpiperazin-1-yl]-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenoxy)acetyl]homopiperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[(methylphenylcarbamoyl)-methyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-thiazol-2-yl-piperazine-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(phenethylcarbamoyl-methyl)piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-((3-(R)-tert-butoxycarbonylamino)pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(3-(R)-[2-(4-chlorophenoxy)-acetylamino]pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(3-(R)-(4-chlorobenzoylamino)-pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-benzyloxycarbonylpiperidin-3-ylamino)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-tert-butoxycarbonylpyrrolidin-3-(S)-ylamino)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-(4-chlorophenoxyacetyl)pyrrolidin-3-(S)-ylamino)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-benzoylpiperidine-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(2-phenoxyethyl)piperazin-1-yl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-[1-(4-fluorophenyl)propyl]-7-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine; 5-amino-2-[cyclopentyl-(4-fluorophenyl)methyl]-7-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine; 5-amine-7-piperazin-1-yl-2-(2-thiophen-2-yl-ethyl)-thiazolo[5,4-d]pyrimidine; 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-[2-(4-chloro-phenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine; 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-[2-(4-chloro-phenyl)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine; 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-(4-chloro-benzoyl)piperazin-1-yl)thiazolo[5,4-d]pyrimidine; 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-m- tolylcarbamoylpiperazin-1-yl)thiazolo[5,4-d]pyrimidine; 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone; 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)-5-methyl-oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone; 1-(4-(5-amino-2-(4-fluorophenyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorobenzyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone; 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorobenzyl)-5-methyloxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone; 1-(4-(5-amino-2-(4-fluorophenethyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; N-(3-chloro-4-fluorophenyl)-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidin-7-amine; N-7-(3-chloro-4-fluorophenyl)-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidine-5,7-diamine; 5-amino-2-cyclopropyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine; 5-amino-2-methoxymethyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine; 5-amino-2-cyclohexyl-7-N-piperazino- oxazolo[5,4-d]pyrimidine; 5-amino-2-pentyl-7-N-piperazino- oxazolo[5,4-d]pyrimidine; 5-amino-2-(2-phenylethyl)-7-N-piperazino- oxazolo[5, 4-d]pyrimidine; 5-amino-2-cyclopropyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-oxazolo[5,4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methoxylmethyloxazolo[5, 4-d]pyrimidine; 5-amino-2-cyclohexyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]oxazolo[5, 4-d]pyrimidine; 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]- 2-pentyloxazolo[5,4-d]pyrimidine; 5-amino-2-(2-phenylethyl)-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]oxazolo[5, 4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-isobutylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-acetylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-[4-(2-methoxyethyl)-piperazin-1-yl]-oxazolo[5,4-d]pyrimidine; 5-amino-2-(4-fluorophenyl)-7-(4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenyl)acetyl]-piperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[4-chlorobenzoyl]piperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-m-tolylcarbamoylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine; 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(2-phenoxyethyl)piperazin-1-yl)-oxazolo[5,4-d]pyrimidine; and 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[(methylphenyl-carbamoyl)methyl]piperazin-1-yl)-oxazolo[5,4-d]pyrimidine.
- The present invention also concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- The present invention also concerns thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal.
- The present invention also concerns the use of the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or a cells transplantation. In an embodiment, said animal is a mammal, preferably said mammal is a human being. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- The present invention also concerns a pharmaceutical composition comprising a therapeutically effective amount of a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine of formula I, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients. In an embodiment, said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs. The present invention also concerns a method of prevention or treatment of an immune disorder in an animal, comprising the administration of a therapeutically effective amount of a thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative or formula I, optionally in combination with one or more pharmaceutically acceptable excipients. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said animal is a mammal, preferably said mammal is a human being.
- The present invention also concerns a process for preparation of the thiazolo(5,4-d)pyrimidine derivatives of formula I, wherein X=S, and comprising the steps of: (a) acylation of 2,5-diamino-4,6-dihydroxypyrimidine; (b) treatment with a thionation reagent; (c) treatment with iodomethane; (d) oxidation reaction by adding an oxidating agent; and (e) a nucleophilic aromatic substitution reaction. In an embodiment, in step (a) said acylation is performed with a carboxylic acid (R2COOH) or an acid chloride (R2C(O)Cl). In an embodiment, step (a) further comprises the addition of a coupling reagent. In an embodiment, said coupling reagent is N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI). In another embodiment, step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt). In an embodiment, in step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent. In an embodiment, in step (b) said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene. In an embodiment, step (c) is performed in alkaline conditions. In an embodiment, in step (d) said oxidating agent is m-chloro-peroxybenzoic acid or hydrogen peroxide. In an embodiment, in step (e) a piperazine is introduced at position 7. In an embodiment, the thiazolo(5,4-d)pyrimidine derivatives of formula I are R2-substituted 5-amino-7-N-piperazino thiazolo(5,4-d)pyrimidine derivatives.
- In an embodiment, the present invention encompasses a thieno(2,3-d)pyrimidine derivative having the general formula II :wherein
- R4 is selected from the group consisting of halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, C1-7 alkyl, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7 alkyloxy;
- R6 is selected from the group consisting of (mono- or di-) C1-12 alkylamino, monoarylamino, diarylamino, (mono- or di-) C3-10 cycloalkylamino, (mono- or di-) hydroxy C1-7 alkylamino, (mono- or di-) C1-4 alkylarylamino, (mono- or di-) arylC1-4 alkylamino, morpholinyl, mercapto C1-7 alkyl, C1-7 alkoxy, homopiperazinyl and piperazinyl,
wherein said homopiperazinyl or piperazinyl is optionally N-substituted with a substituent R21 selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo d1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl,
wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thioheterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino; and - R5 is selected from the group consisting of heteroaryl and aryl groups, wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, or a pharmaceutical acceptable addition salt thereof, or a stereoisomer thereof, or a solvate thereof.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R5 and R6 have any of the values as described herein, and wherein R4 is selected from the group consisting of amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono-or di) C1-4 alkyl-arylamino.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R4 and R5 have any of the values as described herein and wherein R6 is homopiperazinyl or piperazinyl. One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R4 and R6 have any of the values as described herein and wherein R5 is phenyl or 4-fluorophenyl.
- 5One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R5 and R6 have any of the values as described herein and wherein R4 is butyl, methyl, or amino.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R4 and R5 have any of the values as described herein and wherein R6 is 4(m-tolylcarbamoyl)piperazin-1-yl or 4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl.
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, wherein R5 is phenyl or 4-fluorophenyl; R4 is butyl, methyl, or amino; and R6 is (4(m-tolylcarbamoyl)piperazin-1-yl or (4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl).
- One embodiment of the present invention concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 1-(4-(2-butyl-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 2-butyl-N-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine; 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone; 1-(4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 2-amino-4-N-benzylamino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-N-piperazinyl-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-m-tolylcarbamoylpiperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-m-tolylcarbamoylpiperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-p-chlorophenylcarbamoylpiperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-4-[2-phenoxy)acetyl]piperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-N-homopiperazinyl-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-[2-(4-chlorophenoxy)acetyl]homopiperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-[4-chlorobenzoyl]homopiperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-[(methylphenyl-carbamoyl)methyl]piperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(4-(2-phenoxyethyl)piperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(3-(R)-tert-butoxycarbonylamino)pyrrolidin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(3-(R)-amino)pyrrolidin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-(3-(R)-[2-(4-chlorophenoxy)-acetylamino]pyrrolidin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4--(3-(R)-(4-chlorobenzoylamino)-pyrrolidin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine; 2-amino-4-N-piperazinyl--6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-[N-(hydrocinnamoyl)-piperazin-1-yl]-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-[4-phenylmethanesulfonylpiperazin-1-yl]-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-[N-(cyclohexanoyl)-piperazinyl]-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-[N-(isonicotinoyl)-piperazinyl]-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-[N-(diisopropylcarbamoyl)-piperazinyl-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-(4-benzoylpiperidine-1-yl)-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine; 2-amino-4-N-piperazino-thieno[2,3-d]pyrimidine; 2-amino-4-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)-thieno[2,3-d]pyrimidin-4-(3H)-one; 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone; 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate; ethyl 2-(4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl)acetate; 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxamide; 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)-N-(2-methoxyethyl)thieno[2,3-d]pyrimidine-2-carboxamide; 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylic acid; 2-(4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl)acetamide; 4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)-N-m-tolylpiperazine-1-carboxamide; 4-(6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl)-N-m-tolylpiperazine-1-carboxamide; ethyl 6-(4-fluorophenyl)-4-(4-(m-tolylcarbamoyl)piperazin-1-yl)thieno[2,3-d]pyrimidine-2-carboxylate; 6-(4-fluorophenyl)-4-(4-(m-tolylcarbamoyl)piperazin-1-yl)thieno[2,3-d]pyrimidine-2-carboxamide; 4-ethoxy-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine; 6-(4-fluorophenyl)-4-morpholinothieno[2,3-d]pyrimidin-2-amine; N-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-amine; and 4-(3-chloro-4-fluorophenylamino)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate.
- The present invention also concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- The present invention also concerns thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal.
- The present invention also concerns the use of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or a cells transplantation. In an embodiment, said animal is a mammal, preferably said mammal is a human being. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- The present invention also concerns a pharmaceutical composition comprising a therapeutically effective amount of a thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients. In an embodiment, said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- The present invention also concerns a method of prevention or treatment of an immune disorder in a mammal, comprising the administration of a therapeutically effective amount of a thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof , optionally in combination with one or more pharmaceutically acceptable excipients. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said animal is a mammal, preferably said mammal is a human being.
- In an embodiment, the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; activation of carbonyl group by halogenation; a nucleophilic aromatic substitution reaction; introduction of a halogen at position 5; and introduction of R5 by a palladium-catalyzed cross-coupling reaction.
- In another embodiment, the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; treatment with a thionation reagent; an alkylation reaction; conversion of the thiomethyl group to sulfon by oxidation; introduction of the piperazine; introduction of a halogen at position 5; and introduction of R5 by a palladium-catalyzed cross-coupling reaction.
- In another embodiment, the present invention also concerns a process for preparation of the thieno(2,3-d)pyrimidine derivative of formula II, any subgroup thereof, or stereoisomeric forms thereof, comprising the steps of: a ring closure reaction on methyl 2-aminothiophene-3-carboxylate; treatment with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base, and piperazine leading to the formation of 2-amino-4-N-piperazinyl-thieno[2,3-d]pyrimidine; introduction of a halogen at position 5; and introduction of R5 by a palladium-catalyzed cross-coupling reaction.
- In other specific embodiments, said ring closure reaction is effected by treatment with chloroformamidine hydrochloride. In other specific embodiments, said introduction of a halogen at position 5 is effected by treatment with n-BuLi and carbon tetrahalide as halogen source, preferably said halogen is bromide. In other specific embodiments, said introduction of a halogen at position 5 is effected by reaction with N-halosuccinimide (NhS) in CCl4. In other specific embodiments, said halogen is bromide and said N-halosuccinimide (NhS) is N-bromosuccinimide (NBS). Preferably, said palladium-catalyzed cross-coupling reaction is a Suzuki coupling which include reactions with aryl boronic acids or aryl boronic acid pinacol esters; a Heck reaction which include reactions with terminal alkenes; a Sonogashira reaction which includes reactions with terminal alkynes and a Buchwald-Hartwig reaction which include reactions with arylamines; a Negishi reaction which includes the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides; or a Kumada coupling which includes coupling of Grignard reagents with aryl halides. In an embodiment, said activation of carbonyl group by halogenation is performed in acidic conditions. Preferably, said activation of carbonyl group by halogenation is performed by using phosphorus oxychloride or thionylchloride. In other specific embodiments, said nucleophilic aromatic substitution reaction introduces a piperazine moiety at position 4. In other specific embodiments, said treatment with a thionation reagent is performed by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene. In other specific embodiments, said alkylation reaction is performed by treatment with an alkylhalide, such as methyliodide or benzylbromide; under alkaline conditions such as NaOH or triethylamine conditions; in a polar solvent, such as water, DMF or DMSO. In other specific embodiments, said oxidation is performed by treatment with m-chloro-peroxybenzoic acid or hydrogen peroxide. In other specific embodiments, said introduction of the piperazine is performed by a nucleophilic aromatic substitution reaction. In other specific embodiments, said base is triethylamine, diisopropylethylamine, or DBU. In other specific embodiments, the thieno(2,3-d)pyrimidine derivatives are R5-substituted 2-amino-4-N-piperazino-thieno[2,3-d]pyrimidine derivatives.
- In an embodiment, the present invention encompasses a purine derivative having the general formula III or IV: wherein substituent R12 is represented by the general formula V :
- each substituent R13 of the heterocyclic ring is a group independently selected from the group consisting of halogen, nitro, d1-7 alkyl (optionally containing one or more functions or radicals selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, sulfhydryl, C1-7 alkoxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, acetal, thioacetal, imino, oximino, alkyloximino, amino-acid, cyano, (thio)carboxylic acid, (thio)carboxylic acid ester or amide, nitro, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenyl-amino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercapto-alkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl and sulfonamido), C3-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl, alkylaryl, alkylacyl, arylacyl, hydroxyl, sulfhydryl, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thioheterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, hydroxylamino, cyano, (thio)carboxylic acid or esters or thioesters or amides or thioamides thereof;
- n is an integer from 0 to 6;
- R10 is selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkylalkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylatosubstituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-cyanoalkyl, ω-carboxylic ester-alkyl, halo C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, arylalkenyl, aryloxyalkyl, arylalkyl and aryl,
wherein the aryl moiety of each of said arylalkenyl, aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo- alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino, hydrazino, alkylhydrazino and phenylhydrazino; and - R11 is selected from the group consisting of halogen, cyano, carboxylic acid, acyl, thioacyl, alkoxycarbonyl, acyloxy, carbonate, carbamate, C1-7 alkyl, aryl, amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino, mercapto C1-7 alkyl, C1-7 alkyloxy;
- R8 is selected from the group consisting of heteroaryl and aryl groups; halogen; C1-7 alkyl; C2-7 alkenyl; C2-7 alkynyl; halo C1-7 alkyl; carboxy C1-7 alkyl; carboxyaryl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; oxyheterocyclic; heterocyclic-substituted alkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; hydroxylamino; acylamino; thio-acylamino; alkoxyamino; thioalkyl-amino; acetal; thio-acetal; carboxylic acid; carboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; thiocarboxylic acid; thiocarboxylic acid esters, thioesters, halides, anhydrides, amides and thioamides; hydroxyl; sulfhydryl; nitro; cyano; carbamoyl; thiocarbamoyl; ureido; thioureido; amino; alkylamino; cycloalkylamino; alkenylamino; cyclo-alkenylamino; alkynylamino; arylamino; arylalkylamino; hydroxyalkylamino; mercaptoalkylamino; heterocyclic amino; heterocyclic substituted arylamino; heterocyclic-substituted alkyl-amino; oximino; alkyloximino; hydrazino; alkylhydrazino; phenylhydrazino; esters, thioesters, halides, anhydrides, amides and thioamides thereof ; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the purine ring and the aromatic or heterocyclic substituent,
- wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, C2-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thio-heterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, carbamoyl, thiocarbamoyl, ureido, thioureido, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, acylamino, thioacylamino, cyano, carboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, thiocarboxylic acid or esters or thioesters or halides or anhydrides or amides thereof, alkylamino, cycloalkylamino, alkenylamino, cyclo-alkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic amino; and
- wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chain of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, thiol, ether, thio-ether, acetal, thio-acetal, amino, imino, oximino, alkyloximino, aminoacid, cyano, acylamino, thioacylamino, carbamoyl, thiocarbamoyl, ureido, thio-ureido, carboxylic acid ester or halide or anhydride or amide, thiocarboxylic acid or ester or thioester or halide or anhydride or amide, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl, hydroxylamino, mercaptoamino, alkyl-amino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl-amino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, hetero-cyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl, sulfinyl and sulfonamido;
- R7 and R9 are selected from the group consisting of hydrogen, d1-7 alkyl (optionally containing one or more functions or radicals selected from the group consisting of halogen, carbonyl, thiocarbonyl, hydroxyl, sulfhydryl, C1-7 alkoxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, acetal, thioacetal, imino, oximino, alkyloximino, amino-acid, cyano, (thio)carboxylic acid, (thio)carboxylic acid ester or amide, nitro, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenyl-amino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercapto-alkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl and sulfonamido), C3-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl, alkylaryl, acyl and sulfonyl;
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R7, R8, R9, R12 have any of the values as described herein and wherein R11 is amino.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R7, R8, R9, R11 have any of the values as described herein and wherein R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R7, R11, R9, R12 have any of the values as described herein and wherein R8 is 4-fluorophenyl or methylthio.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R7, R8, R11, R12 have any of the values as described herein and wherein R9 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R11, R8, R9, R12 have any of the values as described herein and wherein R7 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R11 is amino, R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or (4-phenoxyacetyl)piperazin-1-yl, R8 is 4-fluorophenyl or methylthio, and R9 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, wherein R11 is amino, R12 is 4-(4-chlorophenoxy)acetylpiperazin-1-yl or 4-(phenoxyacetyl)piperazin-1-yl, R8 is 4-fluorophenyl or methylthio, and R7 is hydrogen or methyl.
- One embodiment of the present invention concerns purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, being selected from the group consisting of: 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(3,4-dimethoxyphenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-bromophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-chlorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(3-chlorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-trifluoromethylphenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-trifluoromethoxyphenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-methylphenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-propyl-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(cyclopropyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(t-butyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-methyl-9H-purine; 2-amino-6-[4-(phenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(3-methoxy-benzoyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(2-thiophene-acetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-chloro-benzoyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[(4-α-toluenesulfonyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(1-naphthoyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-acetylpiperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(thiazol-2-yl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-(pyrrolidin-1-yl)ethanone; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-morpholino-ethanone; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-3-yl)acetamide; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-methyl-N-phenylacetamide; 2-amino-6-[4-(4-chlorophenyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-fluorophenyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-2-yl)acetamide; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(thiazol-2-yl)acetamide; 2-amino-6-[4-(4-fluorobenzyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-pyridinyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-(homopiperazin-1-yl)-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)homopiperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(N-3-tolylcarbamoyl)-homopiperazin-1-yl]-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-methylthio-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-propylthio-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-benzylthio-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(2-phenylethylthio)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-9-methyl-8-methylthio-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(cyclopentylthio)-9H-purine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9-methylpurine; 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9-benzylpurine; 2-amino-6-(piperazin-1-yl)-8-(4-fluorophenyl)-9H-purine; 2-amino-6-[4-(hydrocinnamoyl)-piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine.
- The present invention also concerns purine derivatives of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine.
- The present invention also concerns purine derivatives of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for use as a medicine for the prevention or treatment of immune disorders in an animal. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said animal is a mammal. In an embodiment, said mammal is a human being.
- The present invention also concerns the use of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, for the manufacture of a medicament for the prevention or treatment of an immune disorder in an animal. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said animal is a mammal. In an embodiment, said mammal is a human being.
- The present invention also concerns a pharmaceutical composition comprising a therapeutically effective amount of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, and one or more pharmaceutically acceptable exipients. In an embodiment, said pharmaceutical composition further comprises one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
- The present invention also concerns a method of prevention or treatment of an immune disorder in a mammal, comprising the administration of a therapeutically effective amount of a purine derivative of formula III or IV, any subgroup thereof, or stereoisomeric forms thereof, optionally in combination with one or more pharmaceutically acceptable excipients. In an embodiment, said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation. In an embodiment, said mammal is a human being.
- The present invention also encompasses processes for the preparation of compounds of Formula (I), (II), (III), (IV) and subgroups thereof. In the reactions described, it can be necessary to protect reactive functional groups, for example hydroxy, amino, or carboxy groups, where these are desired in the final product, to avoid their unwanted participation in the reactions. Conventional protecting groups can be used in accordance with standard practice, for example, see T. W. Greene and P. G. M. Wuts in "Protective Groups in Organic Chemistry", John Wiley and Sons, 1999.
- The compounds of Formula (I) (II), (III), (IV) and the subgroups thereof can be prepared by a succession of steps as described herein. They are generally prepared from starting materials which are either commercially available or prepared by standard means obvious to those skilled in the art. The compounds of the present invention can be also prepared using standard synthetic processes commonly used by those skilled in the art of organic chemistry. The general preparation of some typical examples is shown below:
-
- The synthesis starts from commercially available diethyl aminomalonate hydrochloride or dimethyl aminomalonate hydrochloride (not shown in Scheme 1). Conversion into the corresponding amides in step (a) can achieved by acylation with commercially available acid chlorides, bearing the general formula R2C(O)Cl (such as for example, but not limited to acetyl chloride, benzoyl chloride, phenoxyacetyl chloride). The amino group can also be coupled with commercially available carboxylic acids, using standard peptide coupling procedures, using a carboxylic acid, bearing the general formula R2C(O)OH and a coupling reagent such as dicyclohexylcarbodiimide (DCC), diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI). Additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt) can be added to increase the reaction rate. In order to construct the pyrimidine scaffold, the dialkyl acylaminomalonate formed in step (a) can be treated with guanidine, acetamidine or thiourea in an ethanolic sodium ethoxide solution yielding 2-R1-substituted pyrimidines in step (b). By the judicious choices of the carboxylic acid (or acid chloride) and the coupling partner, different substituents can be introduced at positions 2 and 5 of the thiazolo[5,4-d]pyrimidine scaffold. Conversion of the lactam functionalities of 5-acylaminopyrimidine-4,6-diol into thiolactam groups and a concomitant ring closure reaction affording the thiazolo[5,4-d]pyrimidine scaffold can be achieved in step (c), by heating the compound with thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagent in high-boiling solvents such as pyridine, toluene or xylene. In order to introduce a high degree of molecular diversity at position 7, alkylation of the thio group to the corresponding thioethers is effected in step (d). Therefore, the 7-thio-compounds are treated with an alkylhalide (preferably iodomethane as shown in Scheme 1) in the presence of a base (such as for example triethylamine or NaOH) in an appropriate polar solvent (such as for example DMSO, DMF or water) to afford the corresponding alkylsulfanyl analogues. In step (e), the thiomethyl group can be exchanged for a nucleophile bearing the general formula R7H. Nucleophiles can be primary or secondary amines (such as for example, but not limited to, isopropylamine, morpholine and piperazine) or alcoxides. In case of less reactive nucleophiles, it might be necessary to increase the reactivity of thioether group by oxidation to the corresponding sulfones with m-chloroperoxybenzoic acid (mCPBA) in dichloromethane (step (f)). The reaction of sulfone derivatives with a range of primary and secondary amines affords the 2-R2,5-R1,7-R3 trisubstituted thiazolo[5,4-d]pyrimidines. In case a piperazine substituent is introduced at position 7 of the scaffold (R3 = N-piperazinyl; structure not shown in Scheme 1), the second nitrogen can be further derivatised by reaction with chloroformates, isocyanates, acid chlorides (or carboxylic acids) and sulfonyl chlorides yielding carbamates, urea, amides and sulfonamides, respectively.
- A one step procedure in order to introduce an amine at position 7 starting from the 7-thio-thiazolo[5,4-d]pyrimidine derivative is possible using 1,1,1,3,3,3-hexamethyldisilazane (HMDS) in a high boiling solvents such as toluene, xylene or pyridine (step (g)). 2,5,7-tri-substituted thiazolo[5,4-d]pyrimidine derivatives of formula Ia can also be prepared as shown hereunder in Scheme 2.
- Scheme 2 depicts an alternative scheme for the synthesis of 2,5,7-trisubstituted thiazolo[5,4-d]pyrimidines, starting from a commercially available 2-R1-substituted-5-amino-4,6-dihydroxypyrimidine analogue, from which the amino group can be converted to an amide analogue in step (a) using the methods described before, yielding a 2-R1-substituted-5-acylamino-4,6-dihydroxypyrimidine derivative. Alternatively, this dihydroxy pyrimidine analogue can also be converted to its dichloro analogue in step (b) using chlorinating agents (such as for example thionyl chloride or phosphorus oxychloride). In step (c), a nucleophilic aromatic substitution with one equivalent of an appropriate nucleophile, bearing the general formula R3H, yields the 2-R1,4-R3,5-amino-6-chloro-pyrimidine analogue. Introduction of a sulfhydryl group by reaction with sodium sulfide yields the 2-R1,4-R3-5-amino-6-sulfhydryl-pyrimidine analogue in step (d). A ring closure reaction with commercially available acid chlorides or aldehydes, bearing the general formula R2C(O)Cl or R2CHO, yields then the 2-R2-5-R1-7-R3 trisubstituted thiazolo[5,4-d]pyrimidines in step (e).
-
- The synthesis starts from intermediates that have been generated using synthetic schemes described in Schemes 1 and 2. Treatment of the 4,6-dihydroxy-pyrimidine analogue in steps (a) and step (b) with a chlorinating agent (such as for example POCl3, SOCl2, PCl5) yields the 7-hydroxy- (step a) or 7-chloro derivative (step b), depending on the reaction time. The chlorine can be exchanged for a wide variety of oxygen, nitrogen or sulphur containing nucleophiles, bearing the general formula R3H in step (c). Treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and a nitrogen or oxygen nucleophile also leads to the formation of the corresponding 7-substituted oxazolo[5,4-d]pyrimidine analogues in step (d).
-
- The synthesis of a 2-R2-substituted 5-amino-7-N-piperazino thiazolo(5,4-d)pyrimidine of formula Ic can starts from commercially available 2,5-diamino-4,6-dihydroxypyrimidine hydrochloride. Acylation of one of the amino group yielding the amide derivative is achieved in step (a) by reaction with an appropriate acid chloride (bearing the general formula R2C(O)Cl) or with an appropriate acid (bearing the general formula R2COOH) and a coupling reagent, such as for example N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI). Additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt) can be added. In order to construct the thiazole moiety in step (b), the acylaminopyrimidine analogue is heated with thionation reagents such as for example phosphorus pentasulfide or Lawesson's reagents in high-boiling solvents such as pyridine, toluene or xylene. The thio group is then converted to its thiomethyl derivative in step (c) by treatment with iodomethane under alkaline conditions.
- Further oxidation of the thiomethyl group with an oxidating agent (for example m-chloro-peroxybenzoic acid or hydrogen peroxide) affords the sulfoxide or sulfon derivative. In the final step (e), piperazine is introduced at position 7 of the scaffold by a nucleophilic aromatic substitution.
-
- Scheme 5 schematically depicts the synthesis of 2,4,6-trisubstituted thieno[2,3-d]pyrimidine of formula II starting from the commercially available methyl 2-aminothiophene-3-carboxylate or ethyl 2-aminothiophene-3-carboxylate (structure not shown). In order to construct the thieno[2,3-d]pyrimidine scaffold, a ring closure reaction can be effected in step (a) with formamide (yielding a hydrogen at position 2) or with chloroformamidine hydrochloride (furnishing an amino group at position 2). In order to build up alkyl and aryl groups at position 2 of the scaffold, reactions with commercially available nitriles (such as for example, but not limited to, acetonitrile and benzonitrile) under acidic conditions gives access to 2-substituted thieno[2,3-d]pyrimidine analogues. Examples of substituents that can be introduced that way are methyl (by reaction with acetonitrile) and phenyl (by reaction with benzonitrile). Alternatively, the methyl- or ethylester can be hydrolyzed under basic aqueous conditions (e.g. using sodium hydroxide or lithium hydroxide) in step (b) yielding the free carboxylic acid. By using standard peptide coupling procedures in step (c) the corresponding carboxamide is obtained. This might also be achieved by treatment of the carboxylic acid with thionyl chloride or oxalylchloride affording the acid chloride which can be easily converted to an amide by treatment with ammonia. Reaction of 2-amino-3-carboxamido-thiophene with a wide range of orthoesters (e.g. triethylorthoformate and triethylorthoacetate) yields a 2-substituted-thieno[2,3-d]pyrimidin-4(3H)one analogue in step (d). Alternatively, 2-amino-3-carboxamido-thiophene can also be reacted in step (d) with a wide range of commercially available acid chlorides in order to construct the thieno[2,3-d]pyrimidine scaffold. Once the thieno[2,3-d]pyrimidine scaffold is formed in step (a) or step (d), different substituents at various positions can be introduced. In order to make variations at position 4, the oxo group of the lactam functionality can be converted into a good leaving group such as a halogen yielding an aryl chloride. This activation of carbonyl group by halogenation in step (e) is usually performed under harsh and acidic conditions using phosphorus oxychloride or thionylchloride. It might be that additional steps of protection of labile functional groups are needed before halogenation. The 4-chloro-thieno[2,3-d]pyrimidine analogue can be further derivatised either by a nucleophilic aromatic substitution reaction (SNAr) or by palladium-catalyzed cross-coupling reactions in step (f). As an alternative method the lactam group can be converted into a thiolactam group in step (g), by reaction with a thionation reagent such as for example by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene. The thio group is then alkylated in step (h) by treatment with an alkylhalide (such as for example methyliodide, benzylbromide) under alkaline conditions (e.g. NaOH, triethylamine) in a polar solvent (such as water, DMF or DMSO). The thiomethyl group can then be displaced by a suitable nucleophile bearing the general formula R6H (an amine or an alkoxide) yielding a 4-substituted thieno[2,3-d]pyrimidine analogue in step (i). In case of less reactive nucleophiles (such as for example aniline), it might be that the thiomethyl group first needs to be oxidized to its corresponding sulfoxide or sulfon by treatment with m-chloro-peroxybenzoic acid or hydrogen peroxide in step (j). Recently, phosphonium-mediated SNAr reactions for the derivatisation of heterocyclic amides have been reported (Z. K. Wan et al. J. Org. Chem. 2007, 72, 10194-10210 ; Z. K. Wan et al. Org. Lett. 2006, 2425-2428). Treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and a nitrogen or oxygen nucleophile leads to the formation of the corresponding 4-substituted thieno[2,3-d]pyrimidine in step (k). In order to introduce structural variety at position 6 of the scaffold, a halogen is introduced in step (I). The regioselective introduction of a bromine at position 6 can be performed with n-BuLi and carbon tetrabromide as bromine source at low temperature (-78°C). Alternatively, reaction with N-bromosuccinimide (NBS) in CCl4 is also feasible. The 6-bromo-thieno[2,3-d]pyrimidine is an ideal starting material for further derivatisation (step (m)) at position 6 by palladium-catalyzed cross-coupling reactions, such as for example Suzuki couplings (reactions with aryl boronic acids or aryl boronic acid pinacol esters), Heck reactions (reactions with terminal alkenes), Sonogashira (reaction with terminal alkynes) and Buchwald-Hartwig reactions (reaction with arylamines), Negishi reaction (the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides), Kumada coupling (coupling of Grignard reagents with aryl halides).
- In Scheme 6, an alternative synthesis of 2,4,6-trisubstituted thieno[2,3-d]pyrimidine analogues is depicted, in which a 5-substituted ethyl 2-aminothiophene-3-carboxylate is used as a starting material. This compound can be easily generated using the multicomponent Gewald reaction in step (a), in which an aldehyde (with the general formula R5CH2CHO) is condensed with ethyl cyanoacetate in the presence of a base (e.g. triethylamine, morpholine) and elemental sulfur. Depending on the nature of the substituent R5 in the starting aldehyde, a broad structural variety can be introduced at position 6 of the thieno[2,3-d]pyrimidine scaffold. The compound obtained in this way is very similar to the starting material in Scheme 5 and therefore similar reaction sequences can be followed, as explained already in Scheme 5.
-
- The synthesis of 2-amino-4-N-piperazino-6-substituted thieno[2,3-d]pyrimidine of formula IIa starts from commercially available methyl 2-aminothiophene-3-carboxylate. In step (a), a ring closure reaction is effected by treatment with chloroformamidine hydrochloride. The oxo group of the lactam functionality can be converted into a good leaving group such as halogens yielding an aryl chloride. This activation of carbonyl group by halogenation in step (b) is usually performed under harsh and acidic conditions using phosphorus oxychloride or thionylchloride. An additional step of protection of the amino group is needed before halogenation. Introduction of the piperazine moiety at position 4 of the scaffold happens in step (c) by a nucleophilic aromatic substitution reaction. As an alternative method the lactam group can be converted into a thiolactam group in step (d), by reaction with a thionation reagent such as for example by heating with phosphorus pentasulfide or Lawesson's reagent in pyridine or toluene. The thio group is then alkylated in step (e) by treatment with an alkylhalide (such as for example methyliodide, benzylbromide) under alkaline conditions (e.g. NaOH, triethylamine) in a polar solvent (such as water, DMF or DMSO). The thiomethyl group can be oxidized to its corresponding sulfon by treatment with m-chloro-peroxybenzoic acid or hydrogen peroxide in step (f). In step (g), the piperazine is then introduced. Alternatively, treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and piperazine as nucleophile leads to the formation of the corresponding 2-amino-4-N-piperazinyl-thieno[2,3-d]pyrimidine analogue in step (h).
- A halogen at position 5 is introduced in step (i). The regioselective introduction of a bromine at position 5 can be performed with n-BuLi and carbon tetrabromide as bromine source at low temperature (-78°C). Alternatively, reaction with N-bromosuccinimide (NBS) in CCl4 is also feasible. The 5-bromo-thieno[2,3-d]pyrimidine is an ideal starting material for further derivatisation at position 5 by palladium-catalyzed cross-coupling reactions (in step (j)), such as for example Suzuki couplings (reactions with aryl boronic acids or aryl boronic acid pinacol esters), Heck reactions (reactions with terminal alkenes), Sonogashira (reaction with terminal alkynes) and Buchwald-Hartwig reactions (reaction with arylamines), Negishi reaction (the nickel- or palladium-catalyzed coupling of organozinc compounds with various halides), Kumada coupling (coupling of Grignard reagents with aryl halides).
-
- In a first step (a), commercially available 2,6-di-amino-4-chloro-pyrimidine is treated with a nucleophile such as for example piperazine (as shown in Scheme 8). However, this reaction is not limited to piperazine, but can be expanded to other types of nitrogen-containing nucleophiles such as, but not limited to, morpholine, pyrrolidine, methoxyethylamine and piperidine. Also oxygen-containing nucleophiles, such as sodium ethoxide and sodium isopropoxide can be introduced. In the second step (b), the piperazine moiety at position 4 of the pyrimidine ring is further derivatised. This can be achieved by coupling with appropriate acid chlorides (affording amide derivatives), by coupling with sulfonylchlorides (yielding sulfonamides), by reaction with isocyanates (yielding urea), by reaction with isothiocyanates (furnishing thiourea) and by reaction with chloroformates (yielding carbamates). In step (c), a nitroso substituent is introduced at position 5 of the pyrimidine scaffold, by reaction with sodium nitrite in water under acidic conditions (by use of acetic acid or hydrochloric acid). In step (d), the exocyclic amino group at position 6 is acylated in THF in the presence of a base such as for example potassium carbonate or triethylamine. The reductive cyclization in order to construct the purine scaffold in step (e) is achieved by treatment of the acylamino intermediate with triphenylphosphine in toluene or xylene as solvent. Alternatively, the nitroso group can be reduced to the corresponding amino group in step (f). This can be done with, for example, sodium dithionite in water, or catalytically (using hydrogen gas and a catalyst such as Raney Nickel). In step (g), the 5,6-di-amino-pyrimidine intermediate can then be ring closed in order to construct the purine scaffold. This can be done by reaction with carboxylic acids (bearing the general formula R8COOH), in which first a 5-acylaminopyrimidne is formed, followed by an acid-catalyzed ring closure. Alternatively, an aldehyde (bearing the general formula R8CHO) can be used as coupling partner, in which first a Schiff base is formed between the 5-amino-moiety and the aldehyde, followed by oxidative cyclization in the presence of ferric chloride or copper acetate.
-
- In the first step (a), the thiol group is selectively alkylated (for example methylated, as shown in Scheme 9), by treatment with an appropriate alkylhalide under alkaline conditions (using for example potassium carbonate or sodium hydroxide as a base). In step (b), the nitroso group is introduced at position 5 of the pyrimidine scaffold, using similar reaction circumstances as explained in Scheme 8. This 5-nitroso pyrimidine derivative can then be used to construct the purine scaffold, as already mentioned in Scheme 8 (either directly from the nitroso intermediate or via the amino intermediate), affording a 2-amino-6-thiomethyl-8-substituted purine analogue in step (c). The thiomethyl group can directly by displaced by a suitable nucleophile (such as piperazine, as shown in Scheme 9) in step (d). Alternatively, the thiomethyl group can be oxidized in step (e) using an oxidizing agent, such as m-chloro-peroxybenzoic acid, affording the corresponding sulfone derivative, which can be displaced by an appropriate nucleophile in step (f) (such as, but not limited to, piperazine, as shown in Scheme 9). The piperazine moiety can be further derivatised in step (g), as explained in step (b) from Scheme 8.
-
- This method allows to introduce structural variety at position 2 of the purine scaffold. In the first step (a), the thiol group is selectively alkylated (for example methylated, as shown in Scheme 10), by treatment with an appropriate alkylhalide under alkaline conditions (using for example potassium carbonate or sodium hydroxide as a base). In step (b), the nitroso group is introduced at position 5 of the pyrimidine scaffold, using similar reaction circumstances as explained in Scheme 8. This nitroso intermediate can then be used to construct the purine scaffold, as already mentioned in Scheme 6 (either directly from the nitroso derivative or via the 5,6-di-amino analogue), affording a 2-thiomethyl-6-hydroxy-8-substituted purine analogue in step (c). Activation of the tautomeric hydroxyl group at position 6 of the purine scaffold for the subsequent nucleophilic displacement reaction occurs in step (d) by preparing the corresponding 6-(1,2,4-triazolyl)-purine derivative or 6-chloro-purine derivative. The 6-triazolyl derivative can be obtained by treating the 6-oxo-purine derivative with POCl3 or 4-chlorophenyl phosphorodichloridate and 1,2,4-triazole in an appropriate solvent such as, but not limited to, pyridine or acetonitrile. The 4-chloro derivative can be obtained by treating the 6-oxo-purine derivative with thionyl chloride or POCl3. The chlorine atom or thiazolyl group is designated as L in Scheme 10. Nucleophilic displacement of the triazolyl group or chlorine atom occurs in step (e) by reaction with an appropriate nucleophile, such as, but not limited to, piperazine, as shown in Scheme 10. Recently, phosphonium-mediated SNAr reactions for the derivatisation of heterocyclic amides have been reported (Z. K. Wan et al. J. Org. Chem. 2007, 72, 10194-10210 ; Z. K. Wan et al. Org. Lett. 2006, 2425-2428). Treatment of the lactam with benzotriazol-1-yloxytris(dimethylamino)phosphonium hexafluorophosphate (BOP), a base (such as for example triethylamine, diisopropylethylamine, DBU) and a nucleophile (such as, but not limited to, piperazine) leads to the formation of the corresponding 6-N-piperazino-substituted purine analogue in step (f). The piperazine moiety can be further derivatised in step (g) as explained earlier in step (b) of Scheme 8. The thiomethyl group can directly be displaced by suitable nitrogen-, oxygen- or sulfur- containing nucleophiles in step (h). Alternatively, it might be necessary to oxidize the thiomethyl group to its corresponding sulfoxide using hydrogen peroxide or m-chloro-peroxybenzoic acid in step (i). In the last step (j), the sulfoxide group can be exchanged by a suitable nucleophile affording the desired 2,6,8-trisubstituted purine analogue.
-
- In step (a), a purine analogue (synthesized according to one of the synthetic pathways explained in Schemes 8-10), is alkylated with an appropriate alkylhalide bearing the general formula RX (such as for example iodomethane, ethylbromide, benzylbromide), and a base (such as for example sodium hydride or potassium carbonate) in a polar aprotic solvent (such as for example DMF). This reaction can lead to two regio-isomers, depending on the site of alkylation. Both compounds can be separated through techniques known to the person skilled in the art, such as flash chromatography and HPLC.
- In a particular embodiment, the present invention also relates to the thiazolo(5,4-d)pyrimidine, oxazolo(5,4-d)pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, II, III or IV, being selected from the group consisting of:
- 2-(4-fluorophenyl)-7-(piperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorobenzyl)-7-(piperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenyl)-7-(2-methoxyethoxy)-thiazolo[5,4-d]pyrimidin-5-amine
- 7-ethoxy-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-5-amine
- 7-ethoxy-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenyl)-N-7-(3-methoxypropyl)thiazolo[5,4-d]pyrimidine-5,7-diamine
- 2-(4-fluorophenyl)-7-morpholino-thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorobenzyl)-7-morpholino-thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenyl)-7-(4-m-tolylpiperazin-1-yl)-thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenyl)-7-(4-(thiazol-2-yl)piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-fluorophenyl)-7-(4-pentylpiperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-1-morpholinoethanone
- 7-(4-benzylpiperazin-1-yl)-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-5-amine
- benzyl-4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazine-1-carboxylate
- 2-(4-fluorophenyl)-7-(4-(phenylsulfonyl)piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 4-(5-amino-2-(4-fluorophenyl)-thiazolo[5,4-d]pyrimidin-7-yl)-N-p-tolylpiperazine-1-carboxamide
- 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide
- 4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)-5-methylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 1-(4-(5-amino-2-(2-(4-fluorophenoxy)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)-thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-l-yl)-2-(4-fluorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(2,4-dichlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-3-(4-fluorophenoxy)propan-1-one
- 1-(4-(5-amino-2-(1-(4-fluorophenyl)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(1-(4-fluorophenyl)-2-phenylethyl)thiazolo[5,4-d]pyrimidin-7-yI)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 4-(5-amino-2-(1-(4-fluorophenyl)ethyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide
- 5-amino-2-cyclopropyl-7-methoxythiazolo[5,4-d]pyrimidine
- 5-amino-2-cyclopropyl-7-N-piperazino-thiazolo[5, 4-d]pyrimidine
- 5-amino-2-(3,4-dichlorophenyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine
- 5-amino-2-(1-phenylcyclopropyl)-7-(N-piperazino)thiazolo[5, 4-d]pyrimidine
- 5-amino-2-(1-(4-chlorophenyl)cyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine
- 5-amino-7-N-piperazino-2-methylthio-thiazolo[5, 4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-phenylcyclopropyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methylthio-thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(3,4-dichlorophenyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-(4-chlorophenyl)cyclopropyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(2-phenylethyl)thiazolo[5,4-d]pyrimidine
- 5-amino-2-cyclopropyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-cyclohexylthiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(N-oxopyridine-3-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-chlorophenylmethyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-chlorophenyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(3-methoxyphenyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(1-(4-chlorophenyl)ethyl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-fluorophenylamino)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-[2-(4-bromophenoxy)acetyl]-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-(2-phenoxyacetyl)-piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(3-nitrophenoxy)acetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenyl)acetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-m-tolylcarbamoylpiperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-phenoxyacetyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4-(4-chlorobenzoyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4-(3-phenylpropionyl)piperazin-1-yl]-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-[4-phenylmethanesulfonylpiperazin-1-yl]-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenoxy)acetyl]homopiperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[(methylphenylcarbamoyl)-methyl]piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-thiazol-2-yl-piperazine-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(phenethylcarbamoyl-methyl)piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-((3-(R)-tert-butoxycarbonylamino)pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(3-(R)-[2-(4-chlorophenoxy)-acetylamino]pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(3-(R)-(4-chlorobenzoylamino)-pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-benzyloxycarbonylpiperidin-3-ylamino)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-tert-butoxycarbonylpyrrolidin-3-(S)-ylamino)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-(4-chlorophenoxyacetyl)pyrrolidin-3-(S)-ylamino)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-benzoylpiperidine-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(2-phenoxyethyl)piperazin-1-yl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-[1-(4-fluorophenyl)propyl]-7-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-2-[cyclopentyl-(4-fluorophenyl)methyl]-7-(4-[2-(4-chlorophenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-7-piperazin-1-yl-2-(2-thiophen-2-yl-ethyl)-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-[2-(4-chloro-phenoxy)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-[2-(4-chloro-phenyl)acetyl]piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-(4-chloro-benzoyl)piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 5-amino-2-(2-thiophen-2-ylethyl)-7-(4-m- tolylcarbamoylpiperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorophenyl)-5-methyl-oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorobenzyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 2-(4-chlorophenoxy)-1-(4-(2-(4-fluorobenzyl)-5-methyloxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)oxazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- N-(3-chloro-4-fluorophenyl)-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidin-7-amine
- N-7-(3-chloro-4-fluorophenyl)-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidine-5,7-diamine
- 5-amino-2-cyclopropyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- 5-amino-2-methoxymethyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- 5-amino-2-cyclohexyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- 5-amino-2-pentyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- 5-amino-2-(2-phenylethyl)-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- 5-amino-2-cyclopropyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-oxazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methoxymethyloxazolo[5, 4-d]pyrimidine
- 5-amino-2-cyclohexyl-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]oxazolo[5,4-d]pyrimidine
- 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]- 2-pentyloxazolo[5,4-d]pyrimidine
- 5-amino-2-(2-phenylethyl)-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]oxazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-isobutylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-acetylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-[4-(2-methoxyethyl)-piperazin-1-yl]-oxazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluorophenyl)-7-(4-[2-(3-nitrophenoxy)acetyl]-piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[2-(4-chlorophenyl)acetyl]-piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[4-chlorobenzoyl]piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-m-tolylcarbamoylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(2-phenoxyethyl)piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[(methylphenyl-carbamoyl)methyl]piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
- 5-amino-2-phenyl-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(2-furyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine
- 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine
- 1-(4-(5-amino-2-phenylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(furan-2-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone
- 1-(4-(5-amino-2-(furan-2-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(2,4-dichlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chloro-2-methylphenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(3-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-phenylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-cyanophenyl)piperazine-1-carboxamide
- 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(2,4-difluorophenyl)piperazine-1-carboxamide
- 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-bromophenyl)piperazine-1-carboxamide
- 4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(2-methoxyphenyl)piperazine-1-carboxamide
- 4-(5-amino-2-phenylthiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide
- 5-amino-7-(N-piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5, 4-d]pyrimidine
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-p-tolylpiperazine-1-carboxamide
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-3-(4-bromophenyl)propan-1-one
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-hydroxyphenoxy)ethanone
- methyl 4-(2-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-oxoethoxy)benzoate
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-(trifluoromethoxy)phenoxy)ethanone
- 2-(4-acetylphenoxy)-1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)ethanone
- 1-(4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(3-chlorophenoxy)ethanone
- 4-(5-amino-2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-cyanophenyl)piperazine-1-carboxamide
- 1-(4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 4-(5-amino-2-(4-fluorobenzyl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-cyanophenyl)piperazine-1-carboxamide
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-3-(4-fluorophenyl)propan-1-one
- 1-(4-(5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)-2-methylpropan-1-one
- 2-(3-methoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(3,4-dimethoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-methylphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 1-(4-(5-amino-2-(3-methoxyphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(3-methoxyphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(5-amino-2-(3-methoxyphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(3,4-dimethoxyphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(5-amino-2-(3,4-dimethoxyphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-methylphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(5-amino-2-(4-methylphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-methylphenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 7-(piperazin-1-yl)-2-(pyridin-2-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 7-(piperazin-1-yl)-2-(pyridin-4-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-chlorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 1-(4-(5-amino-2-(4-chlorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-trifluoromethoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)-2-methylpropan-1-one
- 1-(4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-m-tolylpiperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-chlorophenyl)piperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-methoxybenzyl)piperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-cyanophenyl)piperazine-1-carboxamide
- 1-(4-(5-amino-2-(4-chlorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-4-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(pyridin-2-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine
- 2-(3-(4-fluorophenyl)propyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-(4-fluorophenyl)butyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-(4-bromophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 2-pentyl-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
- 7-(piperazin-1-yl)-2-p-tolylthiazolo[5,4-d]pyrimidin-5-amine
- 1-(4-(5-amino-2-(3-(4-fluorophenyl)propyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(3-(4-fluorophenyl)propyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-(4-fluorophenyl)butyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-(4-fluorophenyl)butyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-p-tolylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-p-tolylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-pentylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-pentylthiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(5-amino-2-(4-bromophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(5-amino-2-(4-bromophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(2-(4-fluorophenethyl)thiazolo[5,4-d]pyrimidin-7-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone
- 1-(4-(2-butyl-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- N-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine
- 2-butyl-N-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-amine
- 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone
- 1-(4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-amino-4-N-benzylamino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine
- 2-amino-4-N-piperazinyl-6-phenyl-thieno[2,3-d]pyrimidine
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-N-m-tolylpiperazine-1-carboxamide
- 4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-N-(4-chlorophenyl)piperazine-1-carboxamide
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-phenoxyethanone
- 2-amino-4-N-homopiperazinyl-6-phenyl-thieno[2,3-d]pyrimidine
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-1,4-diazepan-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)-1,4-diazepan-1-yl)(4-chlorophenyl)methanone
- 2-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-N-methyl-N-phenylacetamide
- 4-(4-(2-phenoxyethyl)piperazin-1-yl)-6-phenylthieno[2,3-d]pyrimidin-2-amine
- (R)-tert-butyl 1-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)pyrrolidin-3-ylcarbamate
- (R)-4-(3-aminopyrrolidin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine
- (R)-N-(1-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)pyrrolidin-3-yl)-2-(4-chlorophenoxy)acetamide (R)-N-(1-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)pyrrolidin-3-yl)-4-chlorobenzamide
- 2-amino-4-N-piperazinyl-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine
- 1-(4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-3-phenylpropan-1-one
- 4-(4-(benzylsulfonyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine
- (4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)(cyclohexyl)methanone
- 4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)(pyridin-3-yl)methanone
- 4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)-N,N-diisopropylpiperazine-1-carboxamide
- (1-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)piperidin-4-yl)(phenyl)methanone
- 2-amino-4-N-piperazino-thieno[2,3-d]pyrimidine
- 1-(4-(2-aminothieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-(4-chlorophenoxy)-1-(4-(6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)ethanone
- ethyl 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate
- ethyl 2-(4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl)acetate
- 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxamide
- 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)-N-(2-methoxyethyl)thieno[2,3-d]pyrimidine-2-carboxamide
- 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylic acid
- 2-(4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-yl)acetamide
- 4-(2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-yl)-N-m-tolylpiperazine-1-carboxamide
- 4-(6-(4-fluorophenyl)-2-phenylthieno[2,3-d]pyrimidin-4-yl)-N-m-tolylpiperazine-1-carboxamide
- ethyl 6-(4-fluorophenyl)-4-(4-(m-tolylcarbamoyl)piperazin-1-yl)thieno[2,3-d]pyrimidine-2-carboxylate
- 6-(4-fluorophenyl)-4-(4-(m-tolylcarbamoyl)piperazin-1-yl)thieno[2,3-d]pyrimidine-2-carboxamide
- 4-ethoxy-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-2-amine
- 6-(4-fluorophenyl)-4-morpholinothieno[2,3-d]pyrimidin-2-amine
- N-(3-chloro-4-fluorophenyl)-6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4-amine
- ethyl 4-(3-chloro-4-fluorophenylamino)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-methoxyphenoxy)ethanone
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-fluorophenoxy)ethanone
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(m-tolyloxy)ethanone
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-bromophenoxy)ethanone
- 1-(4-(2-amino-6-phenylthieno[2,3-d]pyrimidin-4-yl)piperazin-1-yl)-2-(4-chlorophenoxy)-2-methylpropan-1-one
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(3,4-dimethoxyphenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-bromophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-chlorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(3-chlorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-(trifluoromethyl)phenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-(trifluoromethoxy)phenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-p-tolyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-propyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-cyclopropyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-tert-butyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-methyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-phenoxyethanone
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(3-methoxyphenyl)methanone
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(thiophen-2-yl)ethanone
- (4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(4-chlorophenyl)methanone
- 6-(4-(benzylsulfonyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine
- (4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(naphthalen-1-yl)methanone
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)ethanone
- 8-(4-fluorophenyl)-6-(4-(thiazol-2-yl)piperazin-1-yl)-9H-purin-2-amine
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-(pyrrolidin-1-yl)ethanone
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-morpholinoethanone
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-3-yl)acetamide
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-methyl-N-phenylacetamide
- 6-(4-(4-chlorophenyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine
- 8-(4-fluorophenyl)-6-(4-(4-fluorophenyl)piperazin-1-yl)-9H-purin-2-amine
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-2-yl)acetamide
- 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(thiazol-2-yl)acetamide
- 6-(4-(4-fluorobenzyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine
- 8-(4-fluorophenyl)-6-(4-(pyridin-4-yl)piperazin-1-yl)-9H-purin-2-amine
- 6-(1,4-diazepan-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)-1,4-diazepan-1-yl)-2-(4-chlorophenoxy)ethanone
- 4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)-N-m-tolyl-1,4-diazepane-1-carboxamide
- 1-(4-(2-amino-8-thioxo-8,9-dihydro-7H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(methylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(propylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(benzylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(phenethylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-9-methyl-8-(methylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(cyclopentylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-8-(4-fluorophenyl)-9-methyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 1-(4-(2-amino-9-benzyl-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone
- 2-amino-6-(piperazin-1-yl)-8-(4-fluorophenyl)-9H-purine
- 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-3-phenylpropan-1-one
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-(4-(trifluoromethyl)phenyl)piperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-phenylpiperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-cyclohexylpiperazine-1-carboxamide
- 5-amino-7-[4-(N-4-fluorophenylcarboxamide)piperazin-1-yl]-2-(pyridine-3-yl)thiazolo[5, 4-d]pyrimidine
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-hexylpiperazine-1-carboxamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-p-tolylpiperazine-1-carbothioamide
- 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-methyl-N-p-tolylpiperazine-1-carboxamide
- p-tolyl 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)piperazine-1-carboxylate.
- In another particular embodiment, the invention relates to thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, II, III, or IV, as well as pharmaceutical compositions comprising such thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives as active principle, represented by the above mentioned structural formula I, II, III, or V and being in the form of a pharmaceutically acceptable salt. The latter include any therapeutically active nontoxic addition salt which compounds represented by structural formula I, II, III, or IV are able to form with a salt-forming agent. Such addition salts may conveniently be obtained by treating the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of the invention with an appropriate salt-forming acid or base. For instance, thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives having basic properties may be converted into the corresponding therapeutically active, non-toxic acid addition salt form by treating the free base form with a suitable amount of an appropriate acid following conventional procedures. Examples of such appropriate salt-forming acids include, for instance, inorganic acids resulting in forming salts such as but not limited to hydrohalides (e.g. hydrochloride and hydrobromide), sulfate, nitrate, phosphate, diphosphate, carbonate, bicarbonate, and the like; and organic monocarboxylic or dicarboxylic acids resulting in forming salts such as, for example, acetate, propanoate, hydroxyacetate, 2-hydroxypropanoate, 2-oxopropanoate, lactate, pyruvate, oxalate, malonate, succinate, maleate, fumarate, malate, tartrate, citrate, methanesulfonate, ethanesulfonate, benzoate, 2-hydroxybenzoate, 4-amino-2-hydroxybenzoate, benzenesulfonate, p-toluenesulfonate, salicylate, p-aminosalicylate, palmoate, bitartrate, camphorsulfonate, edetate, 1 ,2-ethanedisulfonate, fumarate, glucoheptonate, gluconate, glutamate, hexylresorcinate, hydroxynaphthoate, hydroxyethanesulfonate, mandelate, methylsulfate, pantothenate, stearate, as well as salts derived from ethanedioic, propanediol, butanedioic, (Z)-2-butenedioic, (E)2- butenedioic, 2-hydroxybutanedioic, 2,3-dihydroxybutane-dioic, 2-hydroxy-1,2,3- propanetricarboxylic and cyclohexanesulfamic acids and the like. Thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, having acidic properties may be converted in a similar manner into the corresponding therapeutically active, non-toxic base addition salt form. Examples of appropriate salt-forming bases include, for instance, inorganic bases like metallic hydroxides such as but not limited to those of alkali and alkaline- earth metals like calcium, lithium, magnesium, potassium and sodium, or zinc, resulting in the corresponding metal salt; organic bases such as but not limited to ammonia, alkylamines, benzathine, hydrabamine, arginine, lysine, N1N1- dibenzylethylenediamine, chloroprocaine, choline, diethanolamine, ethylene-diamine, N-methylglucamine, procaine and the like.
- Reaction conditions for treating the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, with an appropriate salt-forming acid or base are similar to standard conditions involving the same acid or base but different organic compounds with basic or acidic properties, respectively. Preferably, in view of its use in a pharmaceutical composition or in the manufacture of a medicament for treating specific diseases, the pharmaceutically acceptable salt will be designed, i.e. the salt-forming acid or base will be selected so as to impart greater water-solubility, lower toxicity, greater stability and/or slower dissolution rate to the thiazolo(5,4-d)pyrimidine or oxazolo(5,4-d)pyrimidine derivative of this invention.
- Another aspect of the present invention relates to the process for the preparation of the thiazolo(5,4-d)pyrimidine derivatives of this invention, and comprises the steps of: (a) acylation of 2,5-diamino-4,6-dihydroxypyrimidine; (b) treatment with a thionation reagent; (c) treatment with iodomethane; (d) oxidation reaction by adding an oxidating agent; and (e) a nucleophilic aromatic substitution reaction.
- In a specific embodiment, in step (a) said acylation is performed with a carboxylic acid (R2COOH) or an acid chloride (R2C(O)Cl) and/or step (a) further comprises the addition of a coupling reagent such as N,N'-dicyclohexylcarbodiimide (DCC) or N,N'-diisopropylcarbodiimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI) and optionally step (a) further comprises the addition of additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt). In a specific embodiment, in step (b) said thionation reagent is phosphorus pentasulfide or a Lawesson's reagent and said treatment with a thionation reagent is performed in high-boiling solvents such as pyridine, toluene or xylene. In a specific embodiment, step (c) is performed in alkaline conditions. In a specific embodiment, in step (d) said oxidating agent is m-chloro-peroxybenzoic acid or hydrogen peroxide. In a specific embodiment, in step (e) a piperazine is introduced at position 7. In another specific embodiment, the present invention relates to the process for the preparation of the thiazolo(5,4-d)pyrimidine derivatives of this invention wherein said thiazolo(5,4-d)pyrimidine derivatives are R2-substituted 5-amino-7-N-piperazino thiazolo(5,4-d)pyrimidine derivatives, and wherein R2 has any of the values as described herein.
- Another aspect of the present invention relates to the thiazolo[5,4-d]pyrimidine oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of the invention, including the thiazolo[5,4-d]pyrimidine, oxazolo(5,4-d)pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of formula I, for use as a medicine and to the use of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives as a medicine, more in particular to the use of said thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives to treat or prevent an immune disorder in an animal, even more in particularly to treat or prevent autoimmune disorders and particular organ and cells transplant rejections in an animal, more specifically a mammal such as a human being.
- Another aspect of the present invention relates to the pharmaceutical composition of the invention for use as a medicine and to the use of said pharmaceutical composition as a medicine, more in particular to the use of said pharmaceutical composition to treat or prevent an immune disorder in an animal, even more in particularly to treat or prevent autoimmune disorders and particular organ and cells transplant rejections in an animal, more specifically a mammal such as a human being.
- The present invention further provides the use of thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, or a pharmaceutically acceptable salt or a solvate thereof, as a biologically active ingredient, i.e. active principle, especially as a medicine or a diagnostic agent or for the manufacture of a medicament or a diagnostic kit. In a particular embodiment, said medicament may be for the prevention or treatment of a immune disorders, in particular organ and cells transplant rejections, and autoimmune disorders.
- The present invention further provides the use of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, or a pharmaceutically acceptable salt or a solvate thereof, as a biologically active ingredient, i.e. active principle, especially as a medicine or for the manufacture of a medicament for treating an immune disorder or for preventing a transplant rejection.
- The pathologic conditions and disorders concerned by the said use, and the corresponding methods of prevention or treatment, are detailed herein below. Any of the uses mentioned with respect to the present invention may be restricted to a nonmedical use (e.g. in a cosmetic composition), a non-therapeutic use, a non-diagnostic use, a non-human use (e.g. in a veterinary composition), or exclusively an in-vitro use, or a use with cells remote from an animal. The invention further relates to a pharmaceutical composition comprising: (a) one or more thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivatives of this invention, including the ones represented by the structural formula I, II, III, or IV, and (b) one or more pharmaceutically acceptable carriers.
- In another embodiment, this invention provides combinations, preferably synergistic combinations, of one or more thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivatives of this invention with one or more biologically active drugs being preferably selected from the group consisting of immunosuppressant and/or immunomodulator drugs. As is conventional in the art, the evaluation of a synergistic effect in a drug combination may be made by analyzing the quantification of the interactions between individual drugs, using the median effect principle described by Chou et al. in Adv. Enzyme Reg. (1984) 22:27. Briefly, this principle states that interactions (synergism, additivity, antagonism) between two drugs can be quantified using the combination index (hereinafter referred as Cl) defined by the following equation: wherein EDx is the dose of the first or respectively second drug used alone (1 a, 2a), or in combination with the second or respectively first drug (1c, 2c), which is needed to produce a given effect. The said first and second drug have synergistic or additive or antagonistic effects depending upon Cl < 1 , Cl = 1 , or Cl > 1 , respectively. As will be explained in more detail herein below, this principle may be applied to a number of desirable effects such as, but not limited to, an activity against transplant rejection, an activity against immunosuppression or immunomodulation. For instance the present invention relates to a pharmaceutical composition or combined preparation having synergistic effects against immuno-suppression or immunomodulation and containing: (a) one or more immunosuppressant and/or immunomodulator drugs, and (b) at least one thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of the invention, including the ones represented by the structural formula I, II, III, or IV and (c) optionally one or more pharmaceutical excipients or pharmaceutically acceptable carriers, for simultaneous, separate or sequential use in the treatment or prevention of autoimmune disorders and/or in transplant-rejections.
- Suitable immunosuppressant drugs for inclusion in the synergistic compositions or combined preparations of this invention belong to a well known therapeutic class. They are preferably selected from the group consisting of cyclosporine A, substituted xanthines (e.g. methylxanthines such as pentoxyfylline), daltroban, sirolimus, tacrolimus, rapamycin (and derivatives thereof such as defined below), leflunomide (or its main active metabolite A771726, or analogs thereof called malononitrilamides), mycophenolic acid and salts thereof (including the sodium salt marketed under the trade name Mofetil®), adrenocortical steroids, azathioprine, brequinar, gusperimus, 6-mercaptopurine, mizoribine, chloroquine, hydroxy-chloroquine and monoclonal antibodies with immunosuppressive properties (e.g. etanercept, infliximab or kineret). Adrenocortical steroids within the meaning of this invention mainly include glucocorticoids such as but not limited to ciprocinonide, desoxycorticosterone, fludrocortisone, flumoxonide, hydrocortisone, naflocort, procinonide, timobesone, tipredane, dexamethasone, methylprednisolone, methotrexate, prednisone, prednisolone, triamcinolone and pharmaceutically acceptable salts thereof. Rapamycin derivatives as referred herein include O- alkylated derivatives, particularly 9-deoxorapamycins, 26-dihydrorapamycins, 40-O- substituted rapamycins and 28,40-0,0-disubstituted rapamycins (as disclosed in
U.S. Patent No. 5,665,772 ) such as 40-O-(2-hydroxy) ethyl rapamycin - also known as SDZ-RAD -, pegylated rapamycin (as disclosed inU.S. Patent No. 5,780,462 ), ethers of 7-desmethylrapamycin (as disclosed inU.S. Patent No. 6,440,991 ) and polyethylene glycol esters of SDZ-RAD (as disclosed inU.S. Patent No. 6,331,547 ). - Suitable immunomodulator drugs for inclusion into the synergistic immunomodulating pharmaceutical compositions or combined preparations of this invention are preferably selected from the group consisting of acemannan, amiprilose, bucillamine, dimepranol, ditiocarb sodium, imiquimod, Inosine Pranobex, interferon-β, interferon-γ, lentinan, levamisole, lisophylline, pidotimod, romurtide, platonin, procodazole, propagermanium, thymomodulin, thymopentin and ubenimex.
- Synergistic activity of the pharmaceutical compositions or combined preparations of this invention against immunosuppression or immuno-modulation may be readily determined by means of one or more lymphocyte activation tests. Usually activation is measured via lymphocyte proliferation. Inhibition of proliferation thus always means immunosuppression under the experimental conditions applied. There exist different stimuli for lymphocyte activation, in particular: a) co-culture of lymphocytes of different species (mixed lymphocyte reaction, hereinafter referred as MLR) in a so-called mixed lymphocyte culture test: lymphocytes expressing different minor and major antigens of the HLA-DR type (= alloantigens) activate each other non-specifically; b) a CD3 assay wherein there is an activation of the T-lymphocytes via an exogenously added antibody (OKT3). This antibody reacts against a CD3 molecule located on the lymphocyte membrane which has a costimulatory function. Interaction between OKT3 and CD3 results in T-cell activation which proceeds via the Ca2+/calmodulin/calcineurin system and can be inhibited e.g. by cyclosporine A (hereinafter referred as CyA); and c) a CD28 assay wherein specific activation of the T-lymphocyte proceeds via an exogenously added antibody against a CD28 molecule which is also located on the lymphocyte membrane and delivers strong co-stimulatory signals. This activation is Ca2+-independent and thus cannot be inhibited by CyA. Determination of the immunosuppressing or immunomodulating activity of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivatives of this invention, as well as synergistic combinations comprising them, is preferably based on the determination of one or more, preferably at least three lymphocyte activation in vitro tests, more preferably including at least one of the MLR test, CD3 assay and CD28 assay referred above. Preferably the lymphocyte activation in vitro tests used include at least two assays for two different clusters of differentiation preferably belonging to the same general type of such clusters and more preferably belonging to type I transmembrane proteins. Optionally the determination of the immuno-suppressing or immunomodulating activity may be performed on the basis of other lymphocyte activation in vitro tests, for instance by performing a TNF-α assay or an IL-1 assay or an IL-6 assay or an IL-10 assay or an IL-12 assay or an assay for a cluster of differentiation belonging to a further general type of such clusters and more preferably belonging to type II transmembrane proteins such as, but not limited to, CD69, CD71 or CD134.
- The synergistic effect may be evaluated by the median effect analysis method described herein before. Such tests may for instance, according to standard practice in the art, involve the use of equipment, such as flow cytometer, being able to separate and sort a number of cell subcategories at the end of the analysis, before these purified batches can be analyzed further.
- Synergistic activity of the pharmaceutical compositions of this invention in the prevention or treatment of transplant rejection may be readily determined by means of one or more leukocyte activation tests performed in a Whole Blood Assay (hereinafter referred as WBA) described for instance by Lin et al. in Transplantation (1997) 63:1734-1738. WBA used herein is a lymphoproliferation assay performed in vitro using lymphocytes present in the whole blood, taken from animals that were previously given the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivative of this invention, and optionally the other immunosuppressant drug, in vivo. Hence this assay reflects the in vivo effect of substances as assessed by an in vitro read-out assay. The synergistic effect may be evaluated by the median effect analysis method described herein before. Various organ transplantation models in animals are also available in vivo, which are strongly influenced by different immunogenicities, depending on the donor and recipient species used and depending on the nature of the transplanted organ. The survival time of transplanted organs can thus be used to measure the suppression of the immune response.
- The pharmaceutical composition or combined preparation with synergistic activity against immunosuppression or immunomodulation according to this invention may contain the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of this invention, including the ones represented by the structural formulae I, II, III, or IV over a broad content range depending on the contemplated use and the expected effect of the preparation. Typically, the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative content in the combined preparation is within the range of 0.1 to 99.9 % by weight, preferably from 1 to 99 % by weight, more preferably from about 5 to 95 % by weight.
- Auto-immune disorders to be prevented or treated by the pharmaceutical compositions or combined preparations of this invention include both:
- systemic auto-immune diseases such as, but not limited to, lupus erythematosus, psoriasis, vasculitis, polymyositis, scleroderma, multiple sclerosis, ankylosing spondilytis, rheumatoid arthritis and Sjogren syndrome; auto-immune endocrine disorders such as thyroiditis; and
- organ-specific auto-immune diseases such as, but not limited to, Addison disease, hemolytic or pernicious anemia, Goodpasture syndrome, Graves disease, idiopathic thrombocytopenic purpura, insulin-dependent diabetes mellitus, juvenile diabetes, uveitis, Crohn's disease, ulcerative colitis, pemphigus, atopic dermatitis, autoimmune hepatitis, primary biliary cirrhosis, autoimmune pneumonitis, autoimmune carditis, myasthenia gravis, glomerulonephritis and spontaneous infertility.
- Transplant rejections to be prevented or treated by the pharmaceutical compositions or combined preparations of this invention include the rejection of transplanted or grafted organs or cells (both allografts and xenografts), such as but not limited to host versus graft reaction disease. The term "organ" as used herein means all organs or parts of organs in mammals, in particular humans, such as but not limited to kidney, lung, bone marrow, hair, cornea, eye (vitreous), heart, heart valve, liver, pancreas, blood vessel, skin, muscle, bone, intestine or stomach. The term "rejection" as used herein means all reactions of the recipient body or the transplanted organ which in the end lead to cell or tissue death in the transplanted organ or adversely affect the functional ability and viability of the transplanted organ or the recipient. In particular, this means acute and chronic rejection reactions. Also included in this invention is preventing or treating the rejection of cell transplants and xenotransplantation. The major hurdle for xenotransplantation is that even before the T lymphocytes, responsible for the rejection of allografts, are activated, the innate immune system, especially T-independent B lymphocytes and macrophages are activated. This provokes two types of severe and early acute rejection called hyperacute rejection and vascular rejection, respectively. The present invention addresses the problem that conventional immunosuppressant drugs like cyclosporine A are ineffective in xenotransplantation. The ability of the compounds of this invention to suppress T-independent xeno-antibody production as well as macrophage activation may be evaluated in the ability to prevent xenograft rejection in athymic, T-deficient mice receiving xenogenic hamster-heart grafts.
- The term "pharmaceutically acceptable carrier or excipient" as used herein in relation to pharmaceutical compositions and combined preparations means any material or substance with which the active principle, i.e. the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivative of this invention, including the ones represented by the structural formula I, II, III, or IV, and optionally the immunosuppressant or immunomodulator may be formulated in order to facilitate its application or dissemination to the locus to be treated, for instance by dissolving, dispersing or diffusing said composition, and/or to facilitate its storage, transport or handling without impairing its effectiveness. The pharmaceutically acceptable carrier may be a solid or a liquid or a gas which has been compressed to form a liquid, i.e. the compositions of this invention can suitably be used as concentrates, emulsions, solutions, granulates, dusts, sprays, aerosols, pellets or powders. Suitable pharmaceutical carriers for use in said pharmaceutical compositions and their formulation are well known to those skilled in the art. There is no particular restriction to their selection within the present invention although, due to the usually low or very low water-solubility of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivatives of this invention, special attention will be paid to the selection of suitable carrier combinations that can assist in properly formulating them in view of the expected time release profile. Suitable pharmaceutical carriers include additives such as wetting agents, dispersing agents, stickers, adhesives, emulsifying or surface-active agents, thickening agents, complexing agents, gelling agents, solvents, coatings, antibacterial and antifungal agents (for example phenol, sorbic acid, chlorobutanol), isotonic agents (such as sugars or sodium chloride) and the like, provided the same are consistent with pharmaceutical practice, i.e. carriers and additives which do not create permanent damage to mammals.
- The pharmaceutical compositions of the present invention may be prepared in any known manner, for instance by homogeneously mixing, dissolving, spray-drying, coating and/or grinding the active ingredients, in a one-step or a multi-steps procedure, with the selected carrier material and, where appropriate, the other additives such as surface-active agents, may also be prepared by micronisation, for instance in view to obtain them in the form of microspheres usually having a diameter of about 1 to 10 µm, namely for the manufacture of microcapsules for controlled or sustained release of the biologically active ingredient(s). Suitable surface-active agents to be used in the pharmaceutical compositions of the present invention are non-ionic, cationic and/or anionic surfactants having good emulsifying, dispersing and/or wetting properties. Suitable anionic surfactants include both water-soluble soaps and water-soluble synthetic surface-active agents. Suitable soaps are alkaline or alkaline-earth metal salts, unsubstituted or substituted ammonium salts of higher fatty acids (C10-C22), e.g. the sodium or potassium salts of oleic or stearic acid, or of natural fatty acid mixtures obtainable form coconut oil or tallow oil. Synthetic surfactants include sodium or calcium salts of polyacrylic acids; fatty sulphonates and sulphates; sulphonated benzimidazole derivatives and alkylarylsulphonates. Fatty sulphonates or sulphates are usually in the form of alkaline or alkaline-earth metal salts, unsubstituted ammonium salts or ammonium salts substituted with an alkyl or acyl radical having from 8 to 22 carbon atoms, e.g. the sodium or calcium salt of lignosulphonic acid or dodecylsulphonic acid or a mixture of fatty alcohol sulphates obtained from natural fatty acids, alkaline or alkaline-earth metal salts of sulphuric or sulphonic acid esters (such as sodium lauryl sulphate) and sulphonic acids of fatty alcohol/ethylene oxide adducts. Suitable sulphonated benzimidazole derivatives preferably contain 8 to 22 carbon atoms. Examples of alkylarylsulphonates are the sodium, calcium or alcanolamine salts of dodecylbenzene sulphonic acid or dibutyl-naphtalenesulphonic acid or a naphthalene-sulphonic acid/formaldehyde condensation product. Also suitable are the corresponding phosphates, e.g. salts of phosphoric acid ester and an adduct of p-nonylphenol with ethylene and/or propylene oxide, or phospholipids. Suitable phospholipids for this purpose are the natural (originating from animal or plant cells) or synthetic phospholipids of the cephalin or lecithin type such as e.g. phosphatidylethanolamine, phosphatidylserine, phosphatidylglycerine, lysolecithin, cardiolipin, dioctanyl-phosphatidylcholine, dipalmitoylphoshatidylcholine and their mixtures. Suitable non-ionic surfactants include polyethoxylated and polypropoxylated derivatives of alkylphenols, fatty alcohols, fatty acids, aliphatic amines or amides containing at least 12 carbon atoms in the molecule, alkylarenesulphonates and dialkylsulphosuccinates, such as polyglycol ether derivatives of aliphatic and cycloaliphatic alcohols, saturated and unsaturated fatty acids and alkylphenols, said derivatives preferably containing 3 to 10 glycol ether groups and 8 to 20 carbon atoms in the (aliphatic) hydrocarbon moiety and 6 to 18 carbon atoms in the alkyl moiety of the alkylphenol. Further suitable non-ionic surfactants are water-soluble adducts of polyethylene oxide with poylypropylene glycol, ethylenediamino-polypropylene glycol containing 1 to 10 carbon atoms in the alkyl chain, which adducts contain 20 to 250 ethyleneglycol ether groups and / or 10 to 100 propyleneglycol ether groups. Such compounds usually contain from 1 to 5 ethyleneglycol units per propyleneglycol unit. Representative examples of non-ionic surfactants are nonylphenol-polyethoxyethanol, castor oil polyglycolic ethers, polypropylene/polyethylene oxide adducts, tributylphenoxypolyethoxyethanol, polyethyleneglycol and octylphenoxypolyethoxyethanol. Fatty acid esters of polyethylene sorbitan (such as polyoxyethylene sorbitan trioleate), glycerol, sorbitan, sucrose and pentaerythritol are also suitable non-ionic surfactants. Suitable cationic surfactants include quaternary ammonium salts, preferably halides, having four hydrocarbon radicals optionally substituted with halo, phenyl, substituted phenyl or hydroxy; for instance quaternary ammonium salts containing as N-substituent at least one C8-C22 alkyl radical (e.g. cetyl, lauryl, palmityl, myristyl, oleyl and the like) and, as further substituents, unsubstituted or halogenated lower alkyl, benzyl and/or hydroxy-C1-4 alkyl radicals. A more detailed description of surface-active agents suitable for this purpose may be found for instance in " McCutcheon's Detergents and Emulsifiers Annual " (MC Publishing Crop., Ridgewood, New Jersey, 1981), " Tensid-Taschenbuch ", 2nd ed. (Hanser Verlag, Vienna, 1981) and " Encyclopaedia of Surfactants " (Chemical Publishing Co., New York, 1981). Structure-forming, thickening or gel-forming agents may be included into the pharmaceutical compositions and combined preparations of the invention. Suitable such agents are in particular highly dispersed silicic acid, such as the product commercially available under the trade name Aerosil; bentonites; tetraalkyl ammonium salts of montmorillonites (e.g., products commercially available under the trade name Bentone), wherein each of the alkyl groups may contain from 1 to 20 carbon atoms; cetostearyl alcohol and modified castor oil products (e.g. the product commercially available under the trade name Antisettle).
- Gelling agents which may be included into the pharmaceutical compositions and combined preparations of the present invention include, but are not limited to, cellulose derivatives such as carboxymethylcellulose, cellulose acetate and the like; natural gums such as arabic gum, xanthum gum, tragacanth gum, guar gum and the like; gelatin; silicon dioxide; synthetic polymers such as carbomers, and mixtures thereof. Gelatin and modified celluloses represent a preferred class of gelling agents.
- Other optional excipients which may be included in the pharmaceutical compositions and combined preparations of the present invention include additives such as magnesium oxide; azo dyes; organic and inorganic pigments such as titanium dioxide; UV-absorbers; stabilisers; odor masking agents; viscosity enhancers; antioxidants such as, for example, ascorbyl palmitate, sodium bisulfite, sodium metabisulfite and the like, and mixtures thereof; preservatives such as, for example, potassium sorbate, sodium benzoate, sorbic acid, propyl gallate, benzylalcohol, methyl paraben, propyl paraben and the like; sequestering agents such as ethylene-diamine tetraacetic acid; flavoring agents such as natural vanillin; buffers such as citric acid and acetic acid; extenders or bulking agents such as silicates, diatomaceous earth, magnesium oxide or aluminum oxide; densification agents such as magnesium salts; and mixtures thereof. Additional ingredients may be included in order to control the duration of action of the biologically-active ingredient in the compositions and combined preparations of the invention. Control release compositions may thus be achieved by selecting appropriate polymer carriers such as for example polyesters, polyamino-acids, polyvinyl-pyrrolidone, ethylene-vinyl acetate copolymers, methylcellulose, carboxy-methylcellulose, protamine sulfate and the like. The rate of drug release and duration of action may also be controlled by incorporating the active ingredient into particles, e.g. microcapsules, of a polymeric substance such as hydrogels, polylactic acid, hydroxymethyl-cellulose, polymethyl methacrylate and the other above-described polymers. Such methods include colloid drug delivery systems including, but not limited to liposomes, microspheres, microemulsions, nanoparticles, nanocapsules and so on. Depending on the route of administration, the pharmaceutical composition or combined preparation of the invention may also require protective coatings.
- Pharmaceutical forms suitable for injectable use include sterile aqueous solutions or dispersions and sterile powders for the extemporaneous preparation thereof. Typical carriers for this purpose therefore include biocompatible aqueous buffers, ethanol, glycerol, propylene glycol, polyethylene glycol, complexing agents such as cyclodextrins and the like, and mixtures thereof.
- Other modes of local drug administration can also be used. For example, the selected active agent may be administered by way of intracavernosal injection, or may be administered topically, in an ointment, gel or the like, or transdermal, including transscrotally, using a conventional transdermal drug delivery system. Intracavernosal injection can be carried out by use of a syringe or any other suitable device. An example of a hypodermic syringe useful herein is described in
U.S. Patent No. 4,127,118 , injection being made on the dorsum of the penis by placement of the needle to the side of each dorsal vein and inserting it deep into the corpora. - Since, in the case of combined preparations including the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and purine derivative of this invention and an immunosuppressant or immunomodulator both ingredients do not necessarily bring out their synergistic therapeutic effect directly at the same time in the patient to be treated, the said combined preparation may be in the form of a medical kit or package containing the two ingredients in separate but adjacent form. In the latter context, each ingredient may therefore be formulated in a way suitable for an administration route different from that of the other ingredient, e.g. one of them may be in the form of an oral or parenteral formulation whereas the other is in the form of an ampoule for intravenous injection or an aerosol.
- The present invention further relates to a method for preventing or treating a disease selected from the group consisting of immune and auto-immune disorders, transplant rejections, in a patient, preferably a mammal, more preferably a human being. The method of this invention consists of administering to the patient in need thereof an effective amount of a thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine and/or purine derivative of this invention, including the ones represented by the structural formula I, II, III, or IV, optionally together with an effective amount of another immunosuppressant or immunomodulator or antineoplastic drug or antiviral agent or phosphodiesterase-4 inhibitor, or a pharmaceutical composition comprising the same, such as disclosed above in extensive details. The effective amount is usually in the range of about 0.01 mg to 20 mg, preferably about 0.1 mg to 5 mg, per day per kg bodyweight for humans. Depending upon the pathologic condition to be treated and the patient's condition, the said effective amount may be divided into several sub-units per day or may be administered at more than one day intervals. The patient to be treated may be any warm-blooded animal, preferably a mammal, more preferably a human being, suffering from said pathologic condition.
- The preferred compounds of the present invention are non-sedating. In other words, a dose of such compounds that is twice the minimum dose sufficient to provide analgesia in an animal model for determining pain relief causes only transient (i. e. lasting for no more than half the time that pain relief lasts) or preferably no statistically significant sedation in an animal model assay of sedation (using the method described by Fitzgerald et al. in Toxicology (1988) 49:433-9). Preferably, a dose that is five times the minimum dose sufficient to provide analgesia does not produce statistically significant sedation. More preferably, a compound provided herein does not produce sedation at intravenous doses of less than 10 mg/kg per day or at oral doses of less than 30 mg/kg per day. If desired, compounds provided herein may be evaluated for toxicity (a preferred compound is non-toxic when an immunomodulating amount or a cell anti-proliferative amount is administered to a subject) and/or side effects (a preferred compound produces side effects comparable to placebo when a therapeutically effective amount of the compound is administered to a subject). Toxicity and side effects may be assessed using any standard method. In general, the term "non-toxic" as used herein shall be understood as referring to any substance that, in keeping with established criteria, is susceptible to approval by the United States Federal Drug Administration for administration to mammals, preferably humans. Toxicity may be also evaluated using assays including bacterial reverse mutation assays, such as an Ames test, as well as standard teratogenicity and tumorogenicity assays. Preferably, administration of compounds provided herein within the therapeutic dose ranges disclosed hereinabove does not result in prolongation of heart QT intervals (e.g. as determined by electrocardiography in guinea pigs, minipigs or dogs). When administered daily, such doses also do not cause liver enlargement resulting in an increase of liver to body weight ratio of more than 50 % over matched controls in laboratory rodents (e. g. mice or rats). Such doses also preferably do not cause liver enlargement resulting in an increase of liver to body weight ratio of more than 10 % over matched untreated controls in dogs or other non-rodent mammals. The preferred compounds of the present invention also do not promote substantial release of liver enzymes from hepatocytes in vivo, i.e. the therapeutic doses do not elevate serum levels of such enzymes by more than 50% over matched untreated controls in vivo in laboratory rodents. Another embodiment of this invention includes the various precursor or "prodrug" forms of the compounds of the present invention. It may be desirable to formulate the compounds of the present invention in the form of a chemical species which itself is not significantly biologically active, but which when delivered to the body of a human being or higher mammal will undergo a chemical reaction catalyzed by the normal function of the body, inter alia, enzymes present in the stomach or in blood serum, said chemical reaction having the effect of releasing a compound as defined herein. The term "prodrug" or "pro-drug" thus relates to these species which are converted in vivo into the active pharmaceutical ingredient.
- The pro-drugs of the present invention can have any form suitable to the formulator, for example, esters are non-limiting common pro-drug forms. In the present case, however, the pro-drug may necessarily exist in a form wherein a covalent bond is cleaved by the action of an enzyme present at the target locus. For example, a C-C covalent bond may be selectively cleaved by one or more enzymes at said target locus and, therefore, a pro-drug in a form other than an easily hydrolysable precursor, inter alia an ester, an amide, and the like, may be used.
- For the purposes of the present invention the term "therapeutically suitable pro-drug" is defined herein as a compound modified in such a way as to be transformed in vivo to the therapeutically active form, whether by way of a single or by multiple biological transformations, when in contact with the tissues of humans or mammals to which the prodrug has been administered, and without undue toxicity, irritation, or allergic response, and achieving the intended therapeutic outcome. The present invention will be further described with reference to certain more specific embodiments and examples, but the present invention is not limited thereto. The following examples are given by way of illustration only.
- When describing the compounds of the invention, the terms used are to be construed in accordance with the following definitions, unless a context dictates otherwise.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C1-7 alkyl " means straight and branched chain saturated acyclic hydrocarbon monovalent radicals having from 1 to 7 carbon atoms such as, for example, methyl, ethyl, propyl, n-butyl, 1-methylethyi (isopropyl), 2-methylpropyl (isobutyl), 1 ,1-dimethylethyl (ter-butyl), 2-methylbutyl, n-pentyl, dimethylpropyl, n- hexyl, 2-methylpentyl, 3-methylpentyl, n-heptyl and the like. By analogy, the term " C1-12 alkyl " refers to such radicals having from 1 to 12 carbon atoms, i.e. up to and including dodecyl.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " acyl " broadly refers to a substituent derived from an acid such as an organic monocarboxylic acid, a carbonic acid, a carbamic acid (resulting into a carbamoyl substituent) or the thioacid or imidic acid (resulting into a carbamidoyl substituent) corresponding to said acids, and the term " sulfonyl " refers to a substituent derived from an organic sulfonic acid, wherein said acids comprise an aliphatic, aromatic or heterocyclic group in the molecule. In a more specific embodiment of the invention said acyl group, within the scope of the above definition, refers to a carbonyl (oxo) group adjacent to a C1-7 alkyl, a C3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, all of them being such as herein defined. Suitable examples of acyl groups are to be found below. In a more specific embodiment of the invention said " sulfonyl " group, within the scope of the above definition, refers to a sulfonyl group adjacent to a C1-7 alkyl, a C3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, all of them being such as herein defined.
- Acyl and sulfonyl groups originating from aliphatic or cycloaliphatic monocarboxylic acids or sulfonic acids are designated herein as aliphatic or cycloaliphatic acyl and sulfonyl groups and include, but are not limited to, the following:
- alkanoyl (for example formyl, acetyl, propionyl, butyryl, isobutyryl, valeryl, isovaleryl, pivaloyl and the like);
- cycloalkanoyl (for example cyclobutanecarbonyl, cyclopentanecarbonyl, cyclohexanecarbonyl, 1-adamantanecarbonyl and the like);
- cycloalkyl-alkanoyl (for example cyclohexylacetyl, cyclopentylacetyl and the like);
- alkenoyl (for example acryloyl, methacryloyl, crotonoyl and the like);
- alkylthioalkanoyl (for example methylthioacetyl, ethylthioacetyl and the like);
- alkanesulfonyl (for example mesyl, ethanesulfonyl, propanesulfonyl and the like);
- alkoxycarbonyl (for example methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl and the like);
- alkylcarbamoyl (for example methylcarbamoyl and the like);
- (N-alkyl)-thiocarbamoyl (for example (N-methyl)-thiocarbamoyl and the like);
- alkylcarbamidoyl (for example methylcarbamidoyl and the like); and
- alkoxalyl (for example methoxalyl, ethoxalyl, propoxalyl and the like);
- Acyl and sulfonyl groups may also originate from aromatic monocarboxylic acids and include, but are not limited to, the following:
- aroyl (for example benzoyl, toluoyl, xyloyl, 1-naphthoyl, 2-naphthoyl and the like);
- aralkanoyl (for example phenylacetyl and the like);
- aralkenoyl (for example cinnamoyl and the like);
- aryloxyalkanoyl (for example phenoxyacetyl and the like);
- arylthioalkanoyl (for example phenylthioacetyl and the like);
- arylaminoalkanoyl (for example N-phenylglycyl, and the like);
- arylsulfonyl (for example benzenesulfonyl, toluenesulfonyl, naphthalene sulfonyl and the like);
- aryloxycarbonyl (for example phenoxycarbonyl, naphthyloxycarbonyl and the like);
- aralkoxycarbonyl (for example benzyloxycarbonyl and the like);
- arylcarbamoyl (for example phenylcarbamoyl, naphthylcarbamoyl and the like);
- arylglyoxyloyl (for example phenylglyoxyloyl and the like).
- arylthiocarbamoyl (for example phenylthiocarbamoyl and the like); and
- arylcarbamidoyl (for example phenylcarbamidoyl and the like).
- Acyl groups may also originate from an heterocyclic monocarboxylic acids and include, but are not limited to, the following:
- heterocyclic-carbonyl, in which said heterocyclic group is as defined herein, preferably an aromatic or non-aromatic 5- to 7-membered heterocyclic ring with one or more heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur in said ring (for example thiophenoyl, furoyl, pyrrolecarbonyl, nicotinoyl and the like); and
- heterocyclic-alkanoyl in which said heterocyclic group is as defined herein, preferably an aromatic or non-aromatic 5- to 7-membered heterocyclic ring with one or more heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur in said ring (for example thiopheneneacetyl, furylacetyl, imidazolylpropionyl, tetrazolylacetyl, 2-(2-amino-4-thiazolyl)-2-methoxyiminoacetyl and the like).
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " thioacyl " refers to an acyl group as defined herein-above but wherein a sulfur atom replaces the oxygen atom of the carbonyl (oxo) moiety.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C1-7 alkylene " means the divalent hydrocarbon radical corresponding to the above defined C1-7 alkyl, such as methylene, bis(methylene), tris(methylene), tetramethylene, hexamethylene and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C3-10 cycloalkyl " means a mono- or polycyclic saturated hydrocarbon monovalent radical having from 3 to 10 carbon atoms, such as for instance cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and the like, or a C7-10 polycyclic saturated hydrocarbon monovalent radical having from 7 to 10 carbon atoms such as, for instance, norbornyl, fenchyl, trimethyltricycloheptyl or adamantyl.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C3-10 cycloalkyl-alkyl " refers to an aliphatic saturated hydrocarbon monovalent radical (preferably a C1-7 alkyl such as defined above) to which a C3-10 cycloalkyl (such as defined above) is already linked such as, but not limited to, cyclohexylmethyl, cyclopentylmethyl and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C3-10 cycloalkylene" means the divalent hydrocarbon radical corresponding to the above defined C3-10 cycloalkyl.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " aryl " designate any mono- or polycyclic aromatic monovalent hydrocarbon radical having from 6 up to 30 carbon atoms such as but not limited to phenyl, naphthyl, anthracenyl, phenantracyl, fluoranthenyl, chrysenyl, pyrenyl, biphenylyl, terphenyl, picenyl, indenyl, biphenyl, indacenyl, benzocyclobutenyl, benzocyclooctenyl and the like, including fused benzo-C4-β cycloalkyl radicals (the latter being as defined above) such as, for instance, indanyl, tetrahydronaphthyl, fluorenyl and the like, all of the said radicals being optionally substituted with one or more substituents independently selected from the group consisting of halogen, amino, trifluoromethyl, hydroxyl, sulfhydryl and nitro, such as for instance 4-fluorophenyl, 4-chlorophenyl, 3,4-dichlorophenyl, 4-cyanophenyl, 2,6-dichlorophenyl, 2-fluorophenyl, 3-chlorophenyl, 3,5-dichlorophenyl and the like.
- As used herein, e.g. with respect to a substituting radical such as the combination of substituents in certain positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine ring together with the carbon atoms in the same positions of said ring, and unless otherwise stated, the term "homocyclic" means a mono- or polycyclic, saturated or mono-unsaturated or polyunsaturated hydrocarbon radical having from 4 up to 15 carbon atoms but including no heteroatom in the said ring; for instance said combination of substituents may form a C2-6 alkylene radical, such as tetramethylene, which cyclizes with the carbon atoms in certain positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine ring.
- As used herein with respect to a substituting radical (including the combination of substituents in certain positions of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine ring together with the carbon atoms in the same positions of said ring), and unless otherwise stated, the term " heterocyclic " means a mono- or polycyclic, saturated or mono-unsaturated or polyunsaturated monovalent hydrocarbon radical having from 2 up to 15 carbon atoms and including one or more heteroatoms in one or more heterocyclic rings, each of said rings having from 3 to 10 atoms (and optionally further including one or more heteroatoms attached to one or more carbon atoms of said ring, for instance in the form of a carbonyl or thiocarbonyl or selenocarbonyl group, and/or to one or more heteroatoms of said ring, for instance in the form of a sulfone, sulfoxide, N-oxide, phosphate, phosphonate or selenium oxide group), each of said heteroatoms being independently selected from the group consisting of nitrogen, oxygen, sulfur, selenium and phosphorus, also including radicals wherein a heterocyclic ring is fused to one or more aromatic hydrocarbon rings for instance in the form of benzo-fused, dibenzo-fused and naphtho-fused heterocyclic radicals; within this definition are included heterocyclic radicals such as, but not limited to, diazepinyl, oxadiazinyl, thiadiazinyl, dithiazinyl, triazolonyl, diazepinonyl, triazepinyl, triazepinonyl, tetrazepinonyl, benzoquinolinyl, benzothiazinyl, benzothiazinonyl, benzoxa-thiinyl, benzodioxinyl, benzodithiinyl, benzoxazepinyl, benzothiazepinyl, benzodiazepine, benzodioxepinyl, benzodithiepinyl, beπzoxazocinyl, benzo- thiazocinyl, benzodiazocinyl, benzoxathiocinyl, benzodioxocinyl, benzotrioxepinyl, benzoxathiazepinyl, benzoxadiazepinyl, benzothia-diazepinyl, benzotriazepinyl, benzoxathiepinyl, benzotriazinonyl, benzoxazolinonyl, azetidinonyl, azaspiroundecyl, dithiaspirodecyl, selenazinyl, selenazolyl, selenophenyl, hypoxanthinyl, azahypo- xanthinyl, bipyrazinyl, bipyridinyl, oxazolidinyl, diselenopyrimidinyl, benzodioxocinyl, benzopyrenyl, benzopyranonyl, benzophenazinyl, benzoquinolizinyl, dibenzo- carbazolyl, dibenzoacridinyl, dibenzophenazinyl, dibenzothiepinyl, dibenzoxepinyl, dibenzopyranonyl, dibenzoquinoxalinyl, dibenzothiazepinyl, dibenzisoquinolinyl, tetraazaadamantyl, thiatetraazaadamantyl, oxauracil, oxazinyl, dibenzothiophenyl, dibenzofuranyl, oxazolinyl, oxazolonyl, azaindolyl, azolonyl, thiazolinyl, thiazolonyl, thiazolidinyl, thiazanyl, pyrimidonyl, thiopyrimidonyl, thiamorpholinyl, azlactonyl, naphtindazolyl, naphtindolyl, naphtothiazolyl, naphtothioxolyl, naphtoxindolyl, naphto- triazolyl, naphtopyranyl, oxabicycloheptyl, azabenzimidazolyl, azacycloheptyl, azacyclooctyl, azacyclononyl, azabicyclononyl, tetrahydrofuryl, tetrahydropyraπyl, tetrahydropyronyl, tetrahydroquinoleinyl, tetrahydrothienyl and dioxide thereof, dihydrothienyl dioxide, dioxindolyl, dioxinyl, dioxenyl, dioxazinyl, thioxanyl, thioxolyl, thiourazolyl, thiotriazolyl, thiopyranyl, thiopyronyl, coumarinyl, quinoleinyl, oxyquinoleinyl, quinuclidinyl, xanthinyl, dihydropyranyl, benzodihydrofuryl, benzothiopyronyl, benzothiopyranyl, benzoxazinyl, benzoxazolyl, benzodioxolyl, benzodioxanyl, benzothiadiazolyl, benzotriazinyl, benzothiazolyl, benzoxazolyl, phenothioxinyl, phenothiazolyl, phenothienyl (benzothiofuranyl), phenopyronyl, phenoxazolyl, pyridinyl, dihydropyridinyl, tetrahydropyridinyl, piperidinyl, morpholinyl, thiomorpholinyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl, tetrazinyl, triazolyl, benzotriazolyl, tetrazolyl, imidazolyl, pyrazolyl, thiazolyl, thiadiazolyl, isothiazolyl, oxazolyl, oxadiazolyl, pyrrolyl, furyl, dihydrofutyl, furoyl, hydantoinyl, dioxolanyl, dioxolyl, dithianyl, dithienyl, dithiinyl, thienyl, indolyl, indazolyl, benzofutyl, quinolyl, quinazolinyl, quinoxalinyl, carbazolyl, phenoxazinyl, phenothiazinyl, xanthenyl, purinyl, benzothienyl, naphtothienyl, thianthrenyl, pyranyl, pyronyl, benzopyronyl, isobenzofuranyl, chromenyl, phenoxathiinyl, indolizinyl, quinolizinyl, isoquinolyl, phthalaziπyl, naphthiridinyl, cinnolinyl, pteridinyl, carbolinyl, acridinyl, perimidinyl, phenanthrolinyl, phenazinyl, phenothiazinyl, imidazolinyl, imidazolidinyl, benzimidazolyl, pyrazolinyl, pyrazolidinyl, pyrrolinyl, pyrrolidinyl, piperazinyl, uridinyl, thymidinyl, cytidinyl, azirinyl, aziridinyl, diazirinyl, diaziridinyl, oxiranyl, oxaziridinyl, dioxiranyl, thiiranyl, azetyl, dihydroazetyl, azetidinyl, oxetyl, oxetanyl, oxetanonyl, homopiperazinyl, homopiperidinyl, thietyl, thietanyl, diazabicyclooctyl, diazetyl, diaziridinonyl, diaziridinethionyl, chromanyl, chromanonyl, thiochromanyl, thiochromanonyl, thiochromenyl, benzofuranyl, benzisothiazolyl, benzocarbazolyl, benzochromonyl, benzisoalloxazinyl, benzocoumarinyl, thiocoumarinyl, pheno- metoxazinyl, phenoparoxazinyl, phentriazinyl, thiodiazinyl, thiodiazolyl, indoxyl, thioindoxyl, benzodiazinyl (e.g. phtalazinyl), phtalidyl, phtalimidinyl, phtalazonyl, alloxazinyl, dibenzopyronyl (i.e. xanthonyl), xanthionyl, isatyl, isopyrazolyl, isopyrazolonyl, urazolyl, urazinyl, uretinyl, uretidinyl, succinyl, succinimido, benzylsultimyl, benzylsultamyl and the like, including all possible isomeric forms thereof, wherein each carbon atom of said heterocyclic ring may furthermore be independently substituted with a substituent selected from the group consisting of halogen, nitro, C1-7 alkyl (optionally containing one or more functions or radicals selected from the group consisting of carbonyl (oxo), alcohol (hydroxyl), ether (alkoxy), acetal, amino, imino, oximino, alkyloximino, amino-acid, cyano, carboxylic acid ester or amide, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkyl- amino, hydroxylalkylamino, mercaptoalkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfonyl, sulfonamido and halogen), C3-7 alkenyl, C2-7 alkynyl, halo C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl, alkylaryl, alkylacyl, arylacyl, hydroxyl, amino, C1-7 alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, alkynylamino, arylamino, arylalkylamino, hydroxyalkylamino, mercaptoalkylamino, heterocyclic- substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydrazino, alkylhydrazino, phenylhydrazino, sulfhydryl, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, oxyheterocyclic, heterocyclic-substituted alkyloxy, thio C1-7 alkyl, thio C3-10 cycloalkyl, thioaryl, thioheterocyclic, arylalkylthio, heterocyclic-substituted alkylthio, formyl, hydroxylamino, cyano, carboxylic acid or esters or thioesters or amides thereof, tricarboxylic acid or esters or thioesters or amides thereof; depending upon the number of unsaturations in the 3 to 10 atoms ring, heterocyclic radicals may be sub-divided into heteroaromatic (or " heteroaryl ") radicals and non- aromatic heterocyclic radicals; when a heteroatom of said non-aromatic heterocyclic radical is nitrogen, the latter may be substituted with a substituent selected from the group consisting of C1-7 alkyl, C3-10 cycloalkyl, aryl, arylalkyl and alkylaryl. As used herein with respect to a substituting radical, and unless otherwise stated, the terms " C1-7 alkoxy ", " C3-10 cycloalkoxy ", " aryloxy ", " arylalkyloxy ", " oxyheterocyclic ", " thio C1-7 alkyl ", " thio C3-10 cycloalkyl ", " arylthio ", " arylalkylthio " and " thioheterocyclic" refer to substituents wherein a carbon atom of a C1-7 alkyl, respectively a C3-10 cycloalkyl, aryl, arylalkyl or heterocyclic radical (each of them such as defined herein), is attached to an oxygen atom or a divalent sulfur atom through a single bond such as, but not limited to, methoxy, ethoxy, propoxy, butoxy, pentoxy, isopropoxy, sec-butoxy, tert-butoxy, isopentoxy, cyclopropyloxy, cyclobutyloxy, cyclopentyloxy, thiomethyl, thioethyl, thiopropyl, thiobutyl, thiopentyl, thiocyclopropyl, thiocyclobutyl, thiocyclopentyl, thiophenyl, phenyloxy, benzyloxy, mercaptobenzyl, cresoxy, and the like.
- As used herein with respect to a substituting atom, and unless otherwise stated, the term "halogen" or "halo" means any atom selected from the group consisting of fluorine, chlorine, bromine and iodine.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " halo C1-7 alkyl " means a C1-7 alkyl radical (such as above defined) in which one or more hydrogen atoms are independently replaced by one or more halogens (preferably fluorine, chlorine or bromine), such as but not limited to difluoromethyl, trifluoromethyl, trifluoroethyl, octafluoropentyl, dodecafluoroheptyl, dichloromethyl and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the terms " C2-7 alkenyl " designate a straight and branched acyclic hydrocarbon monovalent radical having one or more ethylenic unsaturations and having from 2 to 7 carbon atoms such as, for example, vinyl, 1-propenyl, 2-propenyl (allyl), 1-butenyl, 2-butenyl, 2-pentenyl, 3-pentenyl, 3-methyl-2-butenyl, 3-hexenyl, 2-hexenyl, 2-heptenyl, 1,3-butadienyl, pentadienyl, hexadienyl, heptadienyl, heptatrienyl and the like, including all possible isomers thereof.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C3-10 cycloalkenyl " means a monocyclic mono- or polyunsaturated hydrocarbon monovalent radical having from 3 to 8 carbon atoms, such as for instance cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohexadienyl, cycloheptenyl, cycloheptadienyl, cycloheptatrienyl, cyclooctenyl, cyclooctadienyl and the like, or a C7-10 polycyclic mono- or polyunsaturated hydrocarbon monovalent radical having from 7 to 10 carbon atoms such as dicyclopentadienyl, fenchenyl (including all isomers thereof, such as α-pinolenyl), bicyclo[2.2.1]hept-2-enyl, bicyclo[2.2.1]hepta-2,5-dienyl, cyclo-fenchenyl and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " C2-7 alkynyl " defines straight and branched chain hydrocarbon radicals containing one or more triple bonds and optionally at least one double bond and having from 2 to 7 carbon atoms such as, for example, acetylenyl, 1-propynyl, 2- propynyl, 1-butynyl, 2-butynyl, 2-pentynyl, 1-pentynyl, 3-methyl-2-butynyl, 3-hexynyl, 2-hexynyl, 1-penten-4-ynyl, 3-penten-1-ynyl, 1,3-hexadien-1-ynyl and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the terms " arylalkyl ", " arylalkenyl " and " heterocyclic-substituted alkyl " refer to an aliphatic saturated or ethylenically unsaturated hydrocarbon monovalent radical (preferably a C1-7 alkyl or C2-7 alkenyl radical such as defined above) onto which an aryl or heterocyclic radical (such as defined above) is already bonded via a carbon atom, and wherein the said aliphatic radical and/or the said aryl or heterocyclic radical may be optionally substituted with one or more substituents independently selected from the group consisting of halogen, amino, hydroxyl, sulfhydryl, C1-7 alkyl, C1-7 alkoxy, trifluoromethyl and nitro, such as but not limited to benzyl, 4-chlorobenzyl, 4-fluorobenzyl, 2-fluorobenzyl, 3,4-dichlorobenzyl, 2,6- dichlorobenzyl, 3-methylbenzyl, 4-methylbenzyl, 4-ter-butylbenzyl, phenylpropyl, 1- naphthylmethyl, phenylethyl, 1-amino-2-phenylethyl, 1-amino-2-[4-hydroxy- phenyl]ethyl, 1-amino-2-[indol-2-yl]ethyl, styryl, pyridylmethyl (including all isomers thereof), pyridylethyl, 2-(2-pyridyl)isopropyl, oxazolylbutyl, 2-thienylmethyl, pyrrolylethyl, morpholinylethyl, imidazol-1-yl-ethyl, benzodioxolylmethyl and 2- furylmethyl.
- As used herein with respect to a substituting radical, and unless otherwise stated, the terms " alkylaryl " and " alkyl-substituted heterocyclic " refer to an aryl or, respectively, heterocyclic radical (such as defined above) onto which are bonded one or more aliphatic saturated or unsaturated hydrocarbon monovalent radicals, preferably one or more C1-7 alkyl, C2-7 alkenyl or C3-10 cycloalkyl radicals as defined above such as, but not limited to, o-toluyl, m-toluyl, p-toluyl, 2,3-xylyl, 2,4-xylyl, 3,4- xylyl, o-cumenyl, m-cumenyl, p-cumenyl, o-cymenyl, m-cymenyl, p-cymenyl, mesityl, ter-butylphenyl, lutidinyl (i.e. dimethylpyridyl), 2-methylaziridinyl, methyl- benzimidazolyl, methylbenzofuranyl, methylbenzothiazolyl, methylbenzotriazolyl, methylbenzoxazolyl and methylbenzselenazolyl.
- As used herein with respect to a substituting radical, and unless otherwise stated, the term " alkoxyaryl " refers to an aryl radical (such as defined above) onto which is (are) bonded one or more C1-7 alkoxy radicals as defined above, preferably one or more methoxy radicals, such as, but not limited to, 2-methoxyphenyl, 3-methoxyphenyl, 4-methoxyphenyl, 3,4-dimethoxyphenyl, 2,4,6-trimethoxyphenyl, methoxynaphtyl and the like.
- As used herein with respect to a substituting radical, and unless otherwise stated, the terms " alkylamino ", " cycloalkylamino ", '.' alkenylamino ", " cyclo- alkenylamino " , " arylamino ", " arylalkylamino ", " heterocyclic-substituted alkylamino ", " heterocyclic-substituted arylamino ", " heterocyclic amino ", " hydroxy- alkylamino ", " mercaptoalkylamino " and " alkynylamino " mean that respectively one (thus monosubstituted amino) or even two (thus disubstituted amino) C1-7 alkyl, C3-10 cycloalkyl, C2-7 alkenyl, C3-10 cycloalkenyl, aryl, arylalkyl, heterocyclic-substituted alkyl, heterocyclic-substituted aryl, heterocyclic (provided in this case the nitrogen atom is attached to a carbon atom of the heterocyclic ring), mono- or polyhydroxy C1-7 alkyl, mono- or polymercapto C1-7 alkyl, or C2-7 alkynyl radical(s) (each of them as defined herein, respectively, and including the presence of optional substituents independently selected from the group consisting of halogen, amino, hydroxyl, sulfhydryl, C1-7 alkyl, C1-7 alkoxy, trifluoromethyl and nitro) is/are attached to a nitrogen atom through a single bond such as, but not limited to, anilino, 2- bromoanilino, 4-bromoanilino, 2-chloroanilino, 3-chloroanilino, 4-chloroanilino, 3- chloro-4-methoxyanilino, 5-chloro-2-methoxyanilino, 2,3-dimethylanilino, 2,4-dimethylanilino, 2,5-dimethylanilino, 2,6-dimethylanilino, 3,4-dimethylanilino, 2- fluoroanilino, 3-fluoroanilino, 4-fluoroanilino, 3-fluoro-2-methoxyanilino, 3-fluoro-4-methoxyanilino, 2-fluoro-4-methylanilino, 2-fluoro-5-methylanilino, 3-fluoro-2- methylanilino, 3-fluoro-4-methylanilino, 4-fluoro-2-methylanilino, 5-fluoro-2- methylanilino, 2-iodoanilino, 3-iodoanilino, 4-iodoanilino, 2-methoxy-5-methylanilino, 4-methoxy-2-methylanilino, 5-methoxy-2-methylanilino, 2-ethoxyanilino, 3-ethoxy- anilino, 4-ethoxyanilino, benzylamino, 2-methoxybenzylamino, 3-methoxybenzylamino, 4-methoxybenzylamino, 2-fluorobenzylamino, 3-fluorobenzylamino, 4-fluoro- benzylamino, 2-chlorobenzylamino, 3-chlorobenzylamino, 4-chlorobenzylamino, 2-aminobenzylamino, diphenylmethylamino, α-naphthylamino, methylamino, dimethylamino, ethylamino, diethylamino, isopropylamino, propenylamino, n- butylamino, ter-butylamino, dibutylamino, 1,2-diaminopropyl, 1,3-diaminopropyl, 1,4-diaminobutyl, 1,5-diaminopentyl, 1,6-diaminohexyl, morpholinomethylamino, 4-morpholinoanilino, hydroxymethylamino, β-hydroxyethylamino and ethynylamino; this definition also includes mixed disubstituted amino radicals wherein the nitrogen atom is attached to two such radicals belonging to two different sub-sets of radicals, e.g. an alkyl radical and an alkenyl radical, or to two different radicals within the same subset of radicals, e.g. methylethylamino; among di-substituted amino radicals, symmetrically-substituted amino radicals are more easily accessible and thus usually preferred from a standpoint of ease of preparation.
- As used herein with respect to a substituting radical, and unless otherwise stated, the terms " (thio)carboxylic acid-ester ", " (thio)carboxylic acid thioester " and " (thio)carboxylic acid amide" refer to radicals wherein the carboxyl or thiocarboxyl group is bonded to the hydrocarbonyl residue of an alcohol, a thiol, a polyol, a phenol, a thiophenol, a primary or secondary amine, a polyamine, an amino-alcohol or ammonia, the said hydrocarbonyl residue being selected from the group consisting of alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, arylalkyl, alkylaryl, alkylamino, cycloalkylamino, alkenylamino, cycloalkenylamino, arylamino, arylalkylamino, heterocyclic-substituted alkylamino, heterocyclic amino, heterocyclic-substituted arylamino, hydroxyalkylamino, mercaptoalkylamino or alkynylamino (such as above defined, respectively). As used herein with respect to a substituting radical, and unless otherwise stated, the term " amino-acid " refers to a radical derived from a molecule having the chemical formula H2N-CHR-COOH1 wherein R is the side group of atoms characterizing the amino-acid type; said molecule may be one of the 20 naturally- occurring amino-acids or any similar non naturally-occurring amino-acid.
- As used herein and unless otherwise stated, the term " stereoisomer " refers to all possible different isomeric as well as conformational forms which the compounds of formula I, II, III, or IV may possess, in particular all possible stereochemical^ and conformationally isomeric forms, all diastereomers, enantiomers and/or conformers of the basic molecular structure. Some compounds of the present invention may exist in different tautomeric forms, all of the latter being included within the scope of the present invention.
- As used herein and unless otherwise stated, the term " enantiomer " means each individual optically active form of a compound of the invention, having an optical purity or enantiomeric excess (as determined by methods standard in the art) of at least 80% (i.e. at least 90% of one enantiomer and at most 10% of the other enantiomer), preferably at least 90% and more preferably at least 98%.
- As used herein and unless otherwise stated, the term " solvate " includes any combination which may be formed by a thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivative of this invention with a suitable inorganic solvent (e.g. hydrates) or organic solvent, such as but not limited to alcohols, ketones, esters, ethers, nitriles and the like.
- The following examples illustrate the present invention.
- For all reactions, analytical grade solvents were used. All moisture-sensitive reactions were carried out in oven-dried glass-ware (135 °C). 1H and 13C NMR spectra were recorded with a Bruker Advance 300 (1H NMR: 300 MHz, 13C NMR: 75 MHz), using tetramethylsilane as internal standard for 1H NMR spectra and DMSO-d6 (39.5 ppm) or CDCl3 (77.2 ppm) for 13C NMR spectra. Abbreviations used are: s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet, br s = broad signal. Coupling constants are expressed in Hertz. Mass spectra are obtained with a Finnigan LCQ advantage Max (ion trap) mass spectrophotometer from Thermo Finnigan, San Jose, CA, USA. Exact mass measurements are performed on a quadrupole time-of-flight mass spectrometer (Q-tof-2, Micromass, Manchester, UK) equipped with a standard electrospray-ionization (ESI) interface. Samples were infused in i-PrOH/H2O (1:1) at 3 µl/min. Melting points are determined on a Barnstead IA 9200 apparatus and are uncorrected. Precoated aluminum sheets (Fluka Silica gel/TLC-cards, 254 nm) were used for TLC. Column chromatography was performed on ICN silica gel 63-200, 60 Å.
- To a solution of diethyl aminomalonate hydrochloride (5.0 g, 23.6 mmol) in pyridine (7.64 ml, 94.5 mmol) and dimethylformamide (60 ml) was added p-fluorobenzoyl chloride (4.19 ml, 35.4 mmol). The reaction mixture was stirred at room temperature for 15 hours. After removing the solvents, the residue was redissolved in dichloromethane, washed with water, brine and dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 30:1) to yield the title compound as a white solid (4.9 g, 70 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.37 (d, J= 7.5 Hz, 1 H, NH), 7.96-8.01 (m, 2H, PhH), 7.33 (t, J= 8.8 Hz, 2H, PhH), 5.30 (d, J= 7.5 Hz, 1 H, CH), 4.12-4.28 (m, 4H, CH2), 1.22 (t, J= 7.1 Hz, 6H, CH3) ppm.
- HRMS: calcd for C14H17FNO5 298.1091, found 298.1092.
- To a solution of 4-fluorophenylacetic acid (4.5 g, 29.3 mmol) and 1-hydroxybenzotriazole (4.35 g, 32.2 mmol) in dichloromethane (140 ml) was added dicyclohexylcarbodiimide (6.65 g, 32.2 mmol). The reaction mixture was stirred at room temperature for 2 hours. The resulting solution was cooled to 0°C, and then a solution of diethyl aminomalonate hydrochloride (6.2 g, 29.3 mmol) in pyridine (4.5 ml, 29.3 mmol) and DMF (10 ml) was added. The temperature was allowed to rise to ambient temperature. After 1 hour, the reaction mixture was concentrated under reduced pressure, and the residue was partitioned between ethyl acetate and a 5% NaHCO3 solution. The organic layer was washed with water, brine and dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (8.5 g, 93 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26-7.29 (m, 2H, PhH), 7.05 (t, J= 8.7 Hz, 2H, PhH), 6.44 (d, J= 6.9 Hz, 1H, NH), 5.12 (d, J= 6.9 Hz, 1H, CH), 4.16-4.32 (m, 4H, CH2), 3.61 (s, 2H, CH 2 Ph), 1.27 (t, J= 7.1 Hz, 6H, CH3) ppm.
- HRMS: calcd for C15H19FNO5 312.1247, found 312.1247.
- This compound was synthesized from dimethyl aminomalonate according to the procedure for the preparation of example 2 using 4-fluorophenylpropionic acid. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (53 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.13-7.18 (m, 2H, PhH), 6.96 (t, J= 8.7 Hz, 2H, PhH), 6.50 (d, J= 6.7 Hz, 1H, NH), 5.19 (d, J= 6.7 Hz, 1H, CH), 3.80 (s, 6H, CH3), 2.95 (t, J= 7.4 Hz, 2H, CH2), 2.58 (t, J= 7.4 Hz, 2H, CH2) ppm.
- HRMS: calcd for C14H17FNO5 298.10908, found 298.10827.
- This compound was synthesized from dimethyl aminomalonate according to the procedure for the preparation of example 2 using 3-(4-fluorophenoxy)propionic acid. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (80 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.01 (br s, 1H, NH), 6.96 (t, J= 9.1 Hz, 2H, PhH), 6.86-6.91 (m, 2H, PhH), 5.22 (d, J= 6.8 Hz, 1 H, CH), 4.23 (t, J= 5.9 Hz, 2H, OCH2), 3.82 (s, 6H, CH3), 2.76 (t, J= 5.9 Hz, 2H, CH2) ppm.
- HRMS: calcd for C14H17FNO6 314.10399, found 314.10315.
- Guanidine hydrochloride (1.35 g, 14.1 mmol) and diethyl 2-(4-fluorobenzamido)malonate (example 1, 3.0 g, 10.1 mmol) were added to a solution of sodium (0.46 g, 20.2 mmol) in ethanol (50 ml). The reaction mixture was refluxed for 3 hours. The reaction was cooled down to room temperature. The solid product was filtered off and washed with ethanol. The product was dissolved in a minimal volume of water and acidified to pH 4-5 with 5M HCl. The precipitate was collected, washed with water and dried to give the title compound as a white solid (1.41 g, 53 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.62 (s, 2H, OH), 8.81 (s, 1H, NH), 7.96-8.01 (m, 2H, PhH), 7.28 (t, J= 8.8 Hz, 2H, PhH), 6.63 (s, 2H, NH2) ppm.
- HRMS: calcd for C11H10FN4O3 265.07369, found 265.07148.
- This compound was synthesized from example 2 (yield of 77%) using a similar procedure as for the preparation of example 5.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.61 (s, 2H, OH), 8.57 (s, 1H, NH), 7.32-7.36 (m, 2H, PhH), 7.10 (t, J= 8.8 Hz, 2H, PhH), 6.57 (s, 2H, NH2), 3.50 (s, 2H, CH2) ppm.
- HRMS: calcd for C12H12FN4O3 279.08934, found 279.08846.
- This compound was prepared from example 3 in a yield of 76%, according to the procedure for the synthesis of example 5.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.67 (s, 2H, OH), 8.41 (s, 1H, NH), 7-26-7.31 (m, 2H, PhH), 7.07 (t, J= 8.8 Hz, 2H, PhH), 6.61 (s, 2H, NH2), 2.82 (t, J= 7.2 Hz, 2H, CH2), 2.50 (t, J= 7.2 Hz, 2H, CH2) ppm.
- HRMS: calcd for C13H14FN4O3 293.10499, found 293.10424
- This compound was prepared from example 4 in a yield of 34%, according to the procedure for the synthesis of example 5.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.65 (s, 2H, OH), 8.51 (s, 1H, NH), 7.10 (t, J= 8.7 Hz, 2H, PhH), 6.90-6.97 (m, 2H, PhH), 6.55 (s, 2H, NH2), 4.15 (t, J= 6.2 Hz, 2H, OCH2), 2.66 (t, J= 6.2 Hz, 2H, CH2) ppm.
- MS 307 [M-H]
- Acetamidine hydrochloride (1.05 g, 11.1 mmol) and dimethyl 2-(4-fluorobenzamido)malonate (1.0 g, 3.71 mmol) were added to a solution of sodium (0.26 g, 11.1 mmol) in ethanol (37 ml). The reaction mixture was refluxed for 3 hours. Then, after cooling, the solid product was collected and washed with ethanol. The product was dissolved in a minimal volume of water and acidified to pH 4-5 with 5M HCl. The precipitate was collected, washed with water and dried to give the title compound as a white solid (0.77 g, 79 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.0 (s, 2H, OH), 9.09 (s, 1H, NH), 8.00 (br s, 2H, PhH), 7.30 (t, J= 8.0 Hz, 2H, PhH), 2.28 (s, 3H, CH3) ppm.
- MS: 261.8 [M-H]
- Thiourea (0.72 g, 9.4 mmol) and diethyl 2-(4-fluorobenzamido)malonate (example 1, 2.0 g, 6.7 mmol) were added to a solution of sodium (0.16 g, 6.7 mmol) in ethanol (50 ml). The reaction mixture was refluxed for 3 hours and then cooled down to room temperature. The precipitate was filtered off and washed with ethanol. The product was dissolved in a minimal volume of water and acidified to pH 4-5 with 5M HCl. The precipitate was collected, washed with water and dried, furnishing the title compound as a white solid (0.78 g, 41 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.47 (s, 1H, SH), 12.32 (s, 2H, OH), 9.15 (s, 1H, NH), 8.00 (br s, 2H, PhH), 7.31 (t, J= 6.8 Hz, 2H, PhH) ppm.
- HRMS: calcd for C11H9FN3O3S 282.03487, found 282.03402.
- A solution of N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-4-fluorobenzamide (example 5, 1.3 g, 4.92 mmol) and phosphorus pentasulfide (2.19 g, 9.84 mmol) in dry pyridine (25 ml) was refluxed for 6 hours. The solvents were evaporated in vacuo. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 30:1), yielding the title compound as a yellow solid (1.28 g, 93 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.51 (s, 1 H, SH), 7.96-8.01 (m, 2H, PhH), 7.38 (t, J= 8.8 Hz, 2H, PhH), 7.10 (s, 2H, NH2) ppm.
- HRMS: calcd for C11H8FN4S2 279.0174, found 279.0165.
- This compound was prepared from example 6 in a yield of 76%, according to the procedure for the synthesis of example 11.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.40 (s, 1 H, SH), 7.37-7.42 (m, 2H, PhH), 7.19 (t, J= 8.9 Hz, 2H, PhH), 6.98 (s, 2H, NH2), 4.29 (s, 2H, CH2) ppm.
- HRMS: calcd for C12H10FN4S2 293.03309, found 293.03260.
- This compound was prepared from example 7 in a yield of 76%, according to the procedure for the synthesis of example 11.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.39 (s, 1 H, SH), 7.31-7.36 (m, 2H, PhH), 7.11 (t, J= 8.8 Hz, 2H, PhH), 6.96 (s, 2H, NH2), 3.23 (t, J= 7.4, 2H, CH2), 3.03 (t, J= 7.4, 2H, CH2) ppm. HRMS: calcd for C13H12FN4S2 307.04874, found 307.04792.
- This compound was prepared from example 9 in a yield of 73%, according to the procedure for the synthesis of example 11.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 14.09 (s, 1H, SH), 8.07-8.15 (m, 2H, PhH), 7.42 (t, J= 8.8 Hz, 2H, PhH), 2.50 (s, 3H, CH3) ppm.
- MS 278.1 [M+H]+
- This compound was prepared from example 10 in a yield of 57%, according to the procedure for the synthesis of example 11.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 13.62 (s, 1 H, SH), 8.00-8.05 (m, 2H, PhH), 7.39 (t, J= 8.7 Hz, 2H, PhH) ppm.
- HRMS: calcd for C11H7FN3S3 295.97861, found 295.97777.
- To a solution of 5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidine-7-thiol (1.2 g, 4.31 mmol) and triethylamine (1.50 ml, 10.8 mmol) in DMSO (25 ml) was added iodomethane (0.54 ml, 8.62 mmol). The reaction mixture was stirred for 12 h under N2 at 25°C. The mixture was poured into water and extracted with EtOAc. The organic extracts were dried over Na2SO4 and the solvents were removed under reduced pressure. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 80:1), yielding the title compound as a light yellow solid (0.76 g, 60 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.98-8.03 (m, 2H, PhH), 7.39 (t, J= 8.8 Hz, 2H, PhH), 7.09 (s, 2H, NH2), 2.59 (s, 3H, CH3) ppm.
- HRMS: calcd for C12H10FN4S2 293.0331, found 293.0328.
- This compound was prepared from example 12 in a yield of 87%, according to the procedure for the synthesis of example 16.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.27-7.32 (m, 2H, PhH), 7.03 (t, J= 8.6 Hz, 2H, PhH), 5.02 (s, 2H, NH2), 4.31 (s, 2H, CH2), 2.61 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H12FN4S2 307.0487, found 307.0482.
- This compound was prepared from example 13 in a yield of 61%, according to the procedure for the synthesis of example 16.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.16-7.21 (m, 2H, PhH), 6.97 (t, J= 8.7 Hz, 2H, PhH), 5.30 (s, 2H, NH2), 3.30 (t, J= 8.3, 2H, CH2), 3.11 (t, J= 8.3, 2H, CH2) ppm.
- HRMS: calcd for C14H14FN4S2 321.06439, found 321.06362.
- A solution of N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-3-(4-fluorophenoxy)propanamide (2.5 g, 8.11 mmol) and phosphorus pentasulfide (3.60 g, 16.2 mmol) in dry pyridine (40 ml) was refluxed for 6 hours. After cooling down to room temperature, the precipitate was filtered off, washed with ethylacetate and dried. The crude 5-amino-2-(2-(4-fluorophenoxy)ethyl)-thiazolo[5,4-d]pyrimidine-7-thiol (2.0 g, 6.20 mmol) was redissolved in DMSO (30 ml). Triethylamine (0.85 ml, 6.12 mmol) and iodomethane (0.31 ml, 4.90 mmol) were added. The reaction mixture was stirred for 12 hours at room temperature under a nitrogen atmosphere. The mixture was poured into water and extracted with EtOAc. The combined organic layers were dried over Na2SO4 and the solvents were removed under reduced pressure. The crude residue was purified by chromatography on silica gel (CH2Cl2/MeOH 100:1), yielding the title compound as a light yellow solid (0.45 g, 15 % over 2 steps).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 6.96 (t, J= 9.1 Hz, 2H, PhH), 6.34-6.88 (m, 2H, PhH), 5.19 (s, 2H, NH2), 4.31 (t, J= 6.2, 2H, CH2), 3.48 (t, J= 6.2, 2H, CH2) ppm.
- HRMS: calcd for C14H14FN4OS2 337.0593, found 337.05827.
- This compound was prepared from example 14 in a yield of 95%, according to the procedure for the synthesis of example 16.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.07-8.11 (m, 2H, PhH), 7.19 (t, J= 8.7 Hz, 2H, PhH), 2.78 (s, 3H, CH3), 2.71 (s, 3H, CH3) ppm.
- To a solution of 2-(4-fluorophenyl)-7-methylthio)-thiazolo[5,4-d]pyrimidin-5-amine (0.30 g, 1.03 mmol) in dichloromethane (5 ml) was added mCPBA (70 %, 0.44 g, 2.57 mmol) at 0°C. The reaction mixture was stirred for 3 hours, whereby the reaction temperature was gradually increased from 0°C to room temperature. The reaction mixture was diluted with CHCl3 and was washed with a saturated NaHCO3 solution, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1), affording the title compound as a white solid (0.31 g, 93 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.11-8.16 (m, 2H, PhH), 7.12 (s, 2H, NH2), 7.44 (t, J= 8.8 Hz, 2H, PhH), 3.58 (s, 3H, CH3) ppm.
- HRMS: calcd for C12H10FN4O2S2 325.02292, found 325.02221.
- This compound was prepared from example 17 in a yield of 71%, according to the procedure for the synthesis of example 21.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.58 (s, 2H, NH2), 7.43-7.48 (m, 2H, PhH), 7.22 (t, J= 6.8 Hz, 2H, PhH), 4.47 (s, 2H, CH2), 3.51 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H12FN4O2S2 339.03857, found 339.03804.
- This compound was prepared from example 18 in a yield of 35%, according to the procedure for the synthesis of example 21.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.17-7.22 (m, 2H, PhH), 6.98 (t, J= 8.6 Hz, 2H, PhH), 5.43 (s, 2H, NH2), 3.43 (s, 3H, CH3), 3.41 (t, J= 7.9, 2H, CH2), 3.17 (t, J= 7.9, 2H, CH2) ppm. HRMS: calcd for C14H14FN4O2S2 353.05422, found 353.05363.
- This compound was prepared from example 19 in a yield of 28%, according to the procedure for the synthesis of example 21.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.57 (s, 2H, NH2), 7.13 (t, J= 8.9 Hz, 2H, PhH), 6.98-7.03 (m, 2H, PhH), 4.39 (t, J= 5.9, 2H, CH2), 3.55 (t, J= 5.9, 2H, CH2) 3.49 (s, 3H, CH3) ppm. HRMS: calcd for C14H14FN4O3S2 369.04913, found 369.04842.
- To a solution of 2-(4-fluorophenyl)-7-methylsulfonyl-thiazolo[5,4-d]pyrimidin-5-amine (0.40 g, 1.23 mmol) and triethylamine (0.26 ml, 1.85 mmol) in dioxane (6 ml) was added piperazine (0.16 g, 1.85 mmol). The reaction mixture was heated at 60 °C for 5 hours. After cooling, the volatiles were removed under reduced pressure. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 15:1), furnishing the title compound as a light yellow solid (0.27 g, 67 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.09 (t, J= 9.2 Hz, 1H, NH), 7.98-8.03 (m, 2H, PhH), 7.37 (t, J= 8.8 Hz, 2H, PhH), 6.58 (s, 2H, NH2), 4.45 (br s, 4H, N(CH2)2), 3.26 (br s, 4H, NH(CH 2 ) 2 ) ppm.
- HRMS: calcd for C15H16FN6S 331.11412, found 331.11290.
- This compound was prepared from example 22 in a yield of 50%, according to the procedure for the synthesis of example 25.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.36-7.41 (m, 2H, PhH), 7.17 (t, J= 8.9 Hz, 2H, PhH), 6.26 (s, 2H, NH2), 4.27 (s, 2H, CH2), 4.14 (br s, 4H, N(CH2)2), 2.85 (t, J= 4.7 Hz, 4H, NH(CH 2 ) 2 ) ppm.
- HRMS: calcd for C16H18FN6S 345.12977, found 345.12883.
- This compound was prepared from example 23 in a yield of 76%, according to the procedure for the synthesis of example 25.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.26-7.31 (m, 2H, PhH), 7.08 (t, J= 8.9 Hz, 2H, PhH), 6.19 (s, 2H, NH2) 3.95 (br s, 4H, N(CH2)2), 3.23 (t, J= 7.4, 2H, CH2), 3.02 (t, J= 7.4, 2H, CH2), 2.74 (br s, 4H, HN(CH 2 ) 2 ) ppm.
- HRMS: calcd for C17H20FN6S 359.14542, found 359.14456.
- To a solution of 5-amino-2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidine-7-thiol (50 mg, 0.18 mmol) and triethylamine (63 µl, 0.45 mmol) in DMSO (1 ml) was added benzyl bromide (43 µl, 0.36 mmol). The reaction mixture was stirred under N2 at 25°C for 3 hours. The mixture was poured onto water and extracted with EtOAc. The organic extracts were dried over Na2SO4 and the solvents were removed under reduced pressure. The residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a light yellow solid (40 mg, 60 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.95-7.98 (m, 2H, PhH), 7.44 (d, J= 6.6 Hz, 2H, PhH), 7.28-7.35 (m, 3H, PhH), 7.15 (t, J= 8.6 Hz, 2H, PhH), 5.09 (s, 2H, NH2), 4.53 (s, 2H, CH2) ppm.
- HRMS: calcd for C18H14FN4S2 369.06439, found 369.06332.
- To a solution of Na (2.0 mg, 0.07 mmol) in 2-methoxyethanol (1 ml) was added 2-(4-fluorophenyl)-7-methylthio-thiazolo[5,4-d]pyrimidin-5-amine (40 mg, 0.14 mmol). The reaction mixture was heated at 80 °C for 20 hours. After cooling, the mixture was neutralized with 1 N HCl and the solvent was removed under reduced pressure. The crude residue was extracted by ethyl acetate, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (35 mg, 80 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.98-8.02 (m, 2H, PhH), 7.14 (t, J= 8.5 Hz, 2H, PhH), 5.02 (s, 2H, NH2), 4.70 (t, J= 5.0 Hz, 2H, OCH2), 3.85 (t, J= 5.0 Hz, 2H, CH 2 OCH3), 3.46 (s, 3H, CH3) ppm.
- HRMS: calcd for C14H14FN4O2S 321.0821, found 321.0812.
- This compound was synthesized according to the procedure for the synthesis of example 29 using ethanol. The crude residue was purified by chromatography on silica gel (ethyl acetate/heptane 1:10) to yield the title compound as a white solid (67 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.97-8.02 (m, 2H, PhH), 7.14 (t, J= 8.6 Hz, 2H, PhH), 5.04 (s, 2H, NH2), 4.61 (q, J= 7.1 Hz, 2H, CH2), 1.51 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C13H12FN4OS 291.0716, found 291.0717.
- This compound was synthesized from example 17 in a yield of 36%, according to the procedure for the synthesis of example 29.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26-7.31 (m, 2H, PhH), 7.02 (t, J= 9.0 Hz, 2H, PhH), 4.95 (s, 2H, NH2), 4.58 (q, J= 7.1 Hz, 2H, CH2), 4.32 (s, 2H, CH2), 1.50 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C14H14FN4OS 305.08723, found 305.08635.
- To a solution of 2-(4-fluorophenyl)-7-methylsulfonyl-thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.15 mmol) and triethylamine (32 µl, 0.23 mmol) in dioxane (1 ml) was added 3-methoxypropylamine (21 µl, 0.20 mmol). The reaction mixture was heated at 60 °C for 5 hours. After cooling, the volatiles were removed under reduced pressure. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 70:1), yielding the title compound as a white solid (32 mg, 62 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.89-7.94 (m, 2H, PhH), 7.13 (t, J= 8.7 Hz, 2H, PhH), 6.37 (br s, 1 H, NH), 4.84 (s, 2H, NH2), 3.68 (q, J= 6.3, 2H, NHCH 2 ), 3.56 (t, J= 5.9 Hz, 2H, OCH2), 3.41 (s, 3H, OCH3), 1.97 (quint, J= 6.3, 2H, CH2) ppm.
- HRMS: calcd for C15H17FN5OS 334.1138, found 334.1121.
- This compound was synthesized from example 21 using morpholine, according to the procedure for the synthesis of example 32. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1) to yield the title compound as a light yellow solid (77 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.86-7.90 (m, 2H, PhH), 7.14 (t, J= 8.6 Hz, 2H, PhH), 4.77 (s, 2H, NH2), 4.35 (br s, 4H, O(CH2)2), 3.85 (t, J= 3.9 Hz, 4H, N(CH2)2) ppm.
- HRMS: calcd for C15H15FN5OS 332.0981, found 332.0975.
- This compound was synthesized from example 22 in a yield of 67%, according to the procedure for the synthesis of example 32.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.24-7.28 (m, 2H, PhH), 7.02 (t, J= 8.7 Hz, 2H, PhH), 4.69 (s, 2H, NH2), 4.27 (br s, 4H, O(CH2)2), 4.21 (s, 2H, CH2), 3.80 (t, J= 5.0 Hz, 4H, N(CH2)2) ppm.
- HRMS: calcd for C16H17FN5OS 346.1138, found 346.1125.
- This compound was synthesized according to the procedure for the preparation of example 32, using 1-m-tolylpiperazine. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 40:1) to yield the title compound as a white solid (60 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.89-7.93 (m, 2H, PhH), 7.12-7.22 (m, 4H, PhH, tolyl H), 6.72-6.82 (m, 3H, tolyl H), 4.77 (s, 2H, NH2), 4.51 (br s, 4H, N(CH2)2), 3.33 (t, J= 5.0 Hz, 4H, N(CH2)2), 2.34 (s, 3H, CH3) ppm.
- HRMS: calcd for C22H22FN6S 421.16107, found 421.15992.
- This compound was synthesized according to the procedure for example 32, using 1-(thiazol-2-yl)piperazine. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1), yielding the pure title compound as a white solid (43 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.87-7.92 (m, 2H, PhH), 7.24 (d, J= 3.6 Hz, 1H, thiazolyl H), 7.15 (t, J= 8.6 Hz, 2H, PhH), 6.63 (d, J= 3.6 Hz, 1H, thiazolyl H), 4.81 (s, 2H, NH2), 4.50 (br s, 4H, N(CH2)2), 3.66 (t, J= 5.3 Hz, 4H, N(CH2)2) ppm.
- HRMS: calcd for C18H17FN7S2 414.09709, found 414.09592.
- This compound was synthesized using a similar procedure as for the preparation of example 32, using 1-pentylpiperazine. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (40 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.86-7.91 (m, 2H, PhH), 7.14 (t, J= 8.6 Hz, 2H, PhH), 4.77 (s, 2H, NH2), 4.38 (br s, 4H, N(CH2)2), 2.61 (br s, 4H, pentylN(CH 2 )2), 2.41 (t, J= 7.8 Hz, 2H, NCH 2 CH2CH2CH2CH3), 2.00 (br s, 2H, NCH2CH 2 CH2CH2CH3), 1.56 (quint, J= 6.7 Hz, 2H, NCH2CH2CH 2 CH2CH3), 1.28-1.34 (m, 2H, NCH2CH2CH2CH 2 CH3), 0.91 (t, J= 6.5 Hz, 3H, NCH2CH2CH2CH2CH 3 ) ppm.
- HRMS: calcd for C20H26FN6S 401.19237, found 401.19102.
- This compound was synthesized according to the procedure for the synthesis of example 32, using 1-morpholino-2-(piperazin-1-yl)ethanone. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 60:1), affording the title compound as a light yellow solid (48 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.85-7.89 (m, 2H, PhH), 7.13 (t, J= 8.4 Hz, 2H, PhH), 4.75 (s, 2H, NH2), 4.37 (br s, 4H, N(CH2)2), 3.71 (br s, 4H, morpholinyl H), 3.66 (br s, 4H, morpholinyl H), 3.25 (S, 2H,CH2), 2.67 (t, J= 4.9 Hz, 4H, CH2N(CH 2 )2) ppm.
- HRMS: calcd for C21H25FN7O2S 458.17745, found 458.17603.
- This compound was synthesized according to the procedure for the preparation of example 32, using 1-benzylpiperazine. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 60:1) to yield the title compound as a white solid (81 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.84-7.88 (m, 2H, PhH), 7.26-7.37 (m, 5H, PhH), 7.12 (t, J= 8.4 Hz, 2H, PhH), 4.73 (s, 2H, NH2), 4.36 (br s, 4H, N(CH2)2), 3.57 (s, 2H, CH2), 2.59 (t, J= 4.7 Hz, 4H, CH2N(CH 2 )2) ppm.
- HRMS: calcd for C22H22FN6S 421.16107, found 421.15986.
- To a solution of 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.15 mmol) and pyridine (18 µl, 0.23 mmol) in DMF (1 ml) was added benzyl chloroformate (24 µl, 0.17 mmol). The reaction mixture was stirred for 3 hours at room temperature. The reaction mixture was quenched with water, extracted with EtOAc, brine and dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (54 mg, 77 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.85-7.89 (m, 2H, PhH), 7.40-7.35 (m, 5H, PhH), 7.13 (t, J= 8.6 Hz, 2H, PhH), 5.19 (s, 2H, CH2), 4.82 (s, 2H, NH2), 4.34 (br s, 4H, N(CH2)2), 3.66 (br s, 4H, CON(CH2)2) ppm.
- HRMS: calcd for C23H22FN6O2S 465.15090, found 465.15005.
- To a solution of 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.15 mmol) and pyridine (18 µl, 0.23 mmol) in DMF (1 ml) was added benzenesulfonyl chloride (21 µl, 0.17 mmol). The reaction mixture was stirred for 3 hours at room temperature. The reaction was quenched with water and extracted with EtOAc. The combined organic layers were dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 80:1) to yield the title compound as a white solid (32 mg, 45 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.94-7.98 (m, 2H, PhH), 7.64-7.79 (m, 5H, PhH), 7.35 (t, J= 8.6 Hz, 2H, PhH), 6.48 (s, 2H, NH2), 4.35 (br s, 4H, N(CH2)2), 3.06 (br s, 4H, S-N(CH2)2) ppm.
- HRMS: calcd for C21H20FN6O2S2 471.10732, found 471.10694.
- To a solution of 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.15 mmol) in DMF (1 ml) was added p-tolyl isocyanate (21 µl, 0.17 mmol) in DMF (0.3 ml). The reaction mixture was stirred for 2 hours at room temperature. The reaction was quenched with water and extracted with EtOAc. The combined organic layers were dried over Na2SO4. After removing the solvents in vacuo, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (31 mg, 44 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.86-7.91 (m, 2H, PhH), 7.26 (d, J=1.6 Hz, 2H, tolyl H), 7.10-7.23 (m, 4H, PhH, tolyl H), 6.34 (s, 1H, NH), 4.81 (s, 2H, NH2), 4.42 (br s, 4H, N(CH2)2), 3.28 (t, J= 5.3 Hz, 4H, CON(CH 2 )2), 2.31 (s, 3H, CH3) ppm.
- HRMS: calcd for C23H23FN7OS 464.1669, found 464.1671.
- This compound was prepared according to the procedure for the synthesis of example 42, using m-tolylisocyanate in a yield of 57%.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.86-7.91 (m, 2H, PhH), 7.25 (s, 1H, tolyl H), 7.12-7.22 (m, 4H, PhH, tolyl H), 6.88 (d, J= 7.2 Hz, 1H, tolyl H), 6.41 (s, 1H, NH), 4.84 (s, 2H, NH2), 4.42 (br s, 4H, N(CH2)2), 3.68 (t, J= 5.3 Hz, 4H, CON(CH 2 )2), 2.33 (s, 3H, CH3) ppm. HRMS: calcd for C23H23FN7OS 464.1669, found 464.1673.
- This compound was prepared from example 26 in a yield of 72%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.06-7.29 (m, 5H, PhH, tolyl H), 7.04 (d, J= 4.7 Hz, 2H, PhH), 6.89 (d, J= 6.2 Hz, 1 H, tolyl H), 6.30 (s, 1 H, NH), 4.71 (s, 2H, NH2), 4.34 (br s, 4H, N(CH2)2), 4.23 (s, 2H, CH2), 3.61-3.65 (m, 4H, CON(CH 2 )2), 2.34 (s, 3H, CH3) ppm.
- HRMS: calcd for C24H25FN7OS 478.1825, found 478.1823.
- To a solution of 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine (40 mg, 0.12 mmol) and pyridine (15 µl, 0.18 mmol) in DMF (1 ml) was added 4-chlorophenoxyacetyl chloride (27 mg, 0.13 mmol). The reaction mixture was stirred for 2 hours at room temperature. The reaction mixture was quenched with water, extracted with EtOAc, brine and was dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (32 mg, 53 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.86-7.91 (m, 2H, PhH), 7.26 (d, J= 9.0 Hz, 2H, tolyl H), 7.15 (t, J= 8.4 Hz, 2H, PhH), 6.93 (d, J= 9.0 Hz, 2H, tolyl H), 4.81 (s, 2H, NH2), 4.75 (s, 2H, CH2), 4.32 (br s, 4H, N(CH2)2), 3.75 (quint, J= 5.0 Hz, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C23H21ClFN6O2S 499.1119, found 499.1130.
- This compound was prepared from example 26 in a yield of 31%, according to the procedure for the synthesis of example 45.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.23-7.28 (m, 4H, PhH), 7.02 (t, J= 8.7 Hz, 2H, PhH), 6.92 (d, J= 9.1 Hz, 2H, PhH), 4.74 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.23-4.28 (br s, 4H, N(CH2)2), 4.22 (s, 2H, CH2), 3.67-3.75 (m, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C24H23ClFN6O2S 513.12758, found 513.12732.
- To a solution of 7-chloro-2-(4-fluorophenyl)-5-methylthiazolo[5,4-d]pyrimidine (30 mg, 0.11 mmol) and triethylamine (22 µl, 0.16 mmol) in dioxane (1 ml) was added 2-(4-chlorophenoxy)-1-(piperazin-1-yl)ethanone (38 mg, 0.16 mmol). The reaction mixture was heated at 70°C for 3 hours. After cooling, the volatiles were removed under reduced pressure. The crude residue was purified by chromatography on silica gel (CH2Cl2/MeOH 70:1) to yield the title compound as a white solid (40 mg, 75 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.93-7.97 (m, 2H, PhH), 7.26 (d, J= 9.1 Hz, 2H, tolyl H), 7.17 (t, J= 8.7 Hz, 2H, PhH), 6.93 (d, J= 9.1 Hz, 2H, tolyl H), 4.76 (s, 2H, CH2), 4.42 (br s, 4H, N(CH2)2), 3.79 (br s, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C24H22ClFN5O2S 498.11668, found 498.11541.
- This compound was prepared from example 24 in a yield of 72%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.25 (d, J= 5.3 Hz, 2H, PhH), 6.83-7.00 (m, 6H, PhH), 4.72 (s, 2H, CH2), 4.31 (t, J= 6.2 Hz, 2H, CH2), 4.23 (br s, 4H, NCH2), 3.71 (br s, 2H, CONCH 2 ), 3.65 (br s, 2H, CONCH 2 ), 3.40 (t, J= 6.2, 2H, CH2) ppm.
- HRMS: calcd for C25H25ClFN6O3S 543.13814, found 543.13690.
- To a solution of 2-(4-fluorophenethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.14 mmol) and triethylamine (30 µl, 0.21 mmol) in dioxane (1 ml) was added 2-(4-chlorophenoxy)-1-(piperazin-1-yl)ethanone (51 mg, 0.21 mmol). The reaction mixture was heated at 70°C for 3 hours. After cooling, the volatiles were removed under reduced pressure. The crude residue was purified by chromatography on silica gel (CH2Cl2/MeOH 70:1) to yield the title compound as a light yellow solid (64 mg, 85 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26 (d, J= 8.9 Hz, 2H, PhH), 7.13-7.18 (m, 2H, PhH), 6.68 (d, J= 8.9, 2H, PhH), 6.93 (t, J= 9.1 Hz, 2H, PhH), 4.73 (br s, 4H, NH2, OCH2), 4.23 (br s, 4H, N(CH2)2), 3.71 (br s, 2H, CONCH 2 ), 3.70 (br s, 2H, CONCH 2 ), 3.23 (t, J= 7.0, 2H, CH2), 3.07 (t, J= 7.0, 2H, CH2) ppm.
- HRMS: calcd for C25H25ClFN6O2S 527.14323, found 527.14215.
- To a solution of 2-(4-fluorophenethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine (40 mg, 0.11 mmol) and 4-methoxyphenoxyacetic acid (30 mg, 0.17 mmol) in DMF (2 ml) was added TBTU (N,N,N',N'-tetramethyl-O-(benzotriazol-1-yl)uronium tetrafluoroborate, 54 mg, 0.17 mmol), followed by DIPEA (29 µl, 0.17 mmol. The reaction mixture was stirred at room temperature for 3 hours. The mixture was diluted with water and extracted with dichloromethane. The combined organic layers were washed with brine and dried over Na2SO4. After removing the solvents under reduced pressure, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 40:1), yielding the title compound as a white solid (40 mg, 69 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.27-7.32 (m, 2H, PhH), 7.09 (t, J= 8.9 Hz, 2H, PhH), 6.84-6.91 (m, 4H, PhH), 6.31 (br s, 2H, NH2), 4.79 (s, 2H, CH2), 4.22 (br s, 2H, NCH2), 4.10 (br s, 2H, NCH2), 3.69 (s, 3H, CH3), 3.56 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.4, 2H, CH2), 3.04 (t, J= 7.4, 2H, CH2) ppm.
- HRMS: calcd for C26H28FN6O3S 523.19276, found 523.19218.
- This compound was prepared from example 27 using 4-fluorophenoxyacetic acid in a yield of 56%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.27-7.32 (m, 2H, PhH), 7.12 (t, J= 8.9 Hz, 2H, PhH), 7.09 (t, J= 8.6 Hz, 2H, PhH), 6.94-6.98 (m, 2H, PhH), 6.31 (br s, 2H, NH2), 4.87 (s, 2H, CH2), 4.23 (brs, 2H, NCH2), 4.10 (br s, 2H, NCH2), 3.56 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.3, 2H, CH2), 3.05 (t, J= 7.3, 2H, CH2) ppm.
- HRMS: calcd for C25H25F2N6O2S 511.17278, found 511.17170.
- This compound was prepared from example 27 using 4-bromophenoxyacetic acid in a yield of 47%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.44 (d, J= 8.9 Hz, 2H, PhH), 7.27-7.32 (m, 2H, PhH), 7.09 (t, J= 8.9 Hz, 2H, PhH), 6.92 (d, J= 8.9 Hz, 2H, PhH), 6.31 (br s, 2H, NH2), 4.91 (s, 2H, CH2), 4.24 (br s, 2H, NCH2), 4.10 (br s, 2H, NCH2), 3.55 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.4, 2H, CH2), 3.04 (t, J= 7.4, 2H, CH2) ppm.
- This compound was prepared from example 27 using 3-methylphenoxyacetic acid in a yield of 44%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ 7.28-7.32 (m, 2H, PhH), 7.16 (t, J= 7.7 Hz, 1H, PhH), 7.09 (t, J= 8.9 Hz, 2H, PhH), 6.73-6.78 (m, 3H, PhH), 6.31 (br s, 2H, NH2), 4.84 (s, 2H, CH2), 4.24 (br s, 2H, NCH2), 4.10 (br s, 2H, NCH2), 3.57 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.4, 2H, CH2), 3.05 (t, J= 7.4, 2H, CH2), 2.27 (s, 3H, CH3) ppm.
- HRMS: calcd for C26H28FN6O2S 507.19785, found 507.19725.
- This compound was prepared from example 27 using 2,4-dichlorophenoxyacetic acid in a yield of 43%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.58 (d, J 2.6 Hz, 1H, PhH), 7.28-7.37 (m, 3H, PhH), 7.07-7.12 (m, 3H, PhH), 6.32 (br s, 2H, NH2), 5.07 (s, 2H, CH2), 4.25 (br s, 2H, NCH2), 4.11 (br s, 2H, NCH2), 3.56 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.5, 2H, CH2), 3.04 (t, J= 7.5, 2H, CH2) ppm.
- HRMS: calcd for C25H24Cl2FN6O2S 561.10425, found 561.10335.
- This compound was prepared from example 27 using 3-(4-fluorophenoxy)propionic acid in a yield of 43%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.27-7.33 (m, 2H, PhH), 7.06-7.14 (m, 4H, PhH), 6.92-6.97 (m, 2H, PhH), 6.30 (br s, 2H, NH2), 4.19 (t, J= 5.8 Hz, 2H, OCH2), 4.17 (br s, 2H, NCH2), 4.09 (br s, 2H, NCH2), 3.59 (br s, 4H, CON(CH 2 )2), 3.25 (t, J= 7.4, 2H, CH2), 3.05 (t, J= 7.4, 2H, CH2), 2.86 (t, J= 5.8, 2H, CH2) ppm.
- To a solution of 2-(4-fluorophenyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine (0.15 g, 0.51 mmol) in dichloromethane (3 ml) was added mCPBA (70 %, 0.32 g, 1.29 mmol) at 0°C. The temperature was gradually raised from 0°C to room temperature and the reaction was stirred for another 3 hours. The reaction mixture was diluted with CHCl3 and washed with a saturated NaHCO3 solution, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by chromatography on silica gel (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (0.10 g, 75 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.80 (s, 1 H, NH), 8.03-8.08 (m, 2H, PhH), 7.40 (t, J= 8.8 Hz, 2H, PhH), 2.41 (s, 3H, CH3) ppm.
- HRMS: calcd for C12H9FN3OS 262.04504, found 262.04433.
- To a solution of 2-(4-fluorophenyl)-5-methyl-thiazolo[5,4-d]pyrimidin-7(6H)-one (0.40 g, 1.23 mmol) in POCl3 was added diisopropylethylamine (0.13 ml, 0.77 mmol). The reaction mixture was stirred under N2 at 90°C for 3.5 hours. After cooling down to room temperature, the reaction mixture was poured into ice-water and the aqueous phase was extracted with diethyl ether. The combined organic layers were washed with a saturated NaHCO3 solution and brine, dried over Na2SO4 and concentrated under reduced pressure. The crude residue was purified by chromatography on silica gel (hexane/EtOAc 10:1) to yield the title compound as a white solid (42.8 mg, 40 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.09-8.14 (m, 2H, PhH), 7.22 (t, J= 8.5 Hz, 2H, PhH), 2.83 (s, 3H, CH3) ppm.
- HRMS: calcd for C12H8ClFN3S 280.01115, found 280.01063.
- To a solution of tert-butyl piperazine-1-carboxylate (0.30 g, 0.16 mmol) and triethylamine (0.34 ml, 2.42 mmol) in dichloromethane (8 ml) was added p-chlorophenoxyacetyl chloride (0.36 mg, 1.77 mmol). The reaction mixture was stirred at room temperature overnight. The mixture was diluted with dichloromethane and washed with water, brine and dried over Na2SO4. After removing the solvents, the crude residue was purified by chromatography on silica gel (CH2Cl2/MeOH 80:1) to yield the title compound as a white solid (0.57g, 100 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.25 (d, J= 8.9 Hz, 2H, PhH), 6.88 (d, J= 8.9 Hz, 2H, PhH), 4.68 (s, 2H, CH2), 3.57 (br s, 4H, CON(CH 2 )2), 3.41 (br s, 4H, CON(CH 2 )2), 1.46 (s, 3H, CH3) ppm.
- HRMS: calcd for C17H24ClN2O4 355.14246, found 355.14167.
- To a solution of ethyl piperazine-1-carboxylate (1.0 g, 6.32 mmol) in dichloromethane (30 ml) was added m-tolylisocyanate (0.90 mg, 6.95 mmol). The reaction mixture was stirred at room temperature for 2 hours. The mixture was diluted with dichloromethane and washed with water, brine and dried over Na2SO4. After removing the solvents, the residue was purified by chromatography on silica gel (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (1.6 g, 87 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.18 (s, 1H, PhH), 7.10-7.15 (m, 2H, PhH), 6.83 (d, J= 6.4 Hz, 1H, PhH), 4.14 (q, J= 7.1 Hz, 2H, OCH 2 CH3), 3.42 (br s, 8H, N(CH 2 )2), 2.27 (s, 3H, CH3), 1.26 (t, J= 7.1 Hz, 3H, OCH2CH 3 ) ppm.
- MS 289.7 [M-H]
- A suspension of tert-butyl 4-(2-(4-chlorophenoxy)acetyl)piperazine-1-carboxylate (example 58, 0.58g, 0.16 mmol) in dichloromethane (8 ml) was treated dropwise at room temperature with TFA until the solid completely dissolved. The reaction mixture was stirred under nitrogen at room temperature overnight. The volatiles were evaporated to dryness, diluted with water and the solid was collected by filtration. The solid was washed with water and dried to yield the title compound as a white solid (0.30 g, 72 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.02 (s, 1 H, NH), 7.32 (d, J= 8.8 Hz, 2H, PhH), 6.96 (d, J= 8.8 Hz, 2H, PhH), 4.90 (s, 2H, CH2), 3.65 (br s, 4H, CON(CH 2 )), 3.18 (br s, 2H, NCH2), 3.10 (br s, 2H, NCH2) ppm.
- HRMS: calcd for C12H16ClN2O2 255.09003, found 255.08913.
- A suspension of ethyl 4-(m-tolylcarbamoyl)piperazine-1-carboxylate (example 59, 1.5 g, 5.15 mmol) in dichloromethane (25 ml) was treated dropwise at room temperature with iodotrimethylsilane (1.6 ml, 11.3 mmol). The reaction mixture was stirred under nitrogen at room temperature overnight. The volatiles were evaporated to dryness, diluted with methanol and the solid was filtered off. The solid was washed with methanol and dichloromethane and dried under vacuum to yield the title compound as a yellow solid (1.1 g, 100 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.62 (s, 1H, NH), 7.29 (s, 1H, PhH), 7.27 (d, J= 7.3 Hz, 1H, PhH), 7.13 (t, J= 7.3 Hz, 1H, PhH), 6.79 (d, J= 7.3 Hz, 1H PhH), 3.65 (br s, 4H, CON(CH 2 )), 3.17 (br s, 4H, N(CH2)2), 2.26 (s, 3H, CH3) ppm.
- MS 227.8 [M-H]
- To a solution of 2-(4-fluorobenzyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine (0.4 g, 1.31 mmol) and 2N NaOH (0.65 ml, 1.31 mmol) in DMSO (7 ml) was added iodomethane (81 µl, 1.31 mmol). The reaction mixture was stirred at room temperature for 2 hours. The mixture was poured into water and extracted with ethyl acetate, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by chromatography on silica gel (hexane/EtOAc 5:1) yielding the title compound as a white solid (0.29 g, 69 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.31 (br s, 2H, PhH), 7.02 (br s, 2H, PhH), 5.16 (s, 2H, NH2), 4.47 (br s, 1 H, CH), 2.58 (s, 3H, CH3), 1.76 (d, J= 6.6Hz, 3H, CHCH 3 ) ppm.
- HRMS: calcd for C14H14FN4S2 321.06439, found 321.06377.
- This compound was synthesized according to the procedure for the preparation of example 62, using benzyl bromide. The crude residue was purified by flash chromatography on silica gel (hexane/EtOAc 5:1) to yield the title compound as a white solid (67 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.13-7.25(m, 5H, PhH), 7.07 (d, J= 6.8 Hz, 2H, PhH), 6.96 (t, J= 8.5 Hz, 2H, PhH), 5.05 (s, 2H, NH2), 4.52 (t, J= 7.8 Hz, 1H, CH), 3.74 (dd, J= 13.8, 7.8 Hz, 1 H, benzylH), 3.30 (dd, J= 13.8, 7.8 Hz, 1 H, benzylH), 2.59 (s, 3H, CH3) ppm. HRMS: calcd for C20H18FN4S2 397.09569, found 397.09495.
- This compound was synthesized from example 62 according to the procedure for the preparation of example 21. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (77 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.35-7.30 (m, 2H, PhH), 7.05 (t, J= 8.6 Hz, 2H, PhH), 5.49 (s, 2H, NH2), 4.54 (q, J= 7.1, 1H, CH), 3.49 (s, 3H, CH3), 1.83 (d, J= 7.1 Hz, 3H, CHCH 3 ) ppm.
- HRMS: calcd for C14H14FN4O2S2 353.05422, found 353.05356.
- This compound was synthesized from example 63 according to the procedure for the preparation of example 21. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (85 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.59 (s, 2H, NH2), 7.44-7.49 (m, 2H, PhH), 7.11-7.24 (m, 7H, PhH), 4.96 (t, J= 7.8 Hz, 1 H, CH), 3.67 (dd, J= 13.8, 7.8 Hz, 1 H, benzylH), 3.53 (s, 3H, CH3), 3.35 (dd, J= 13.8, 7.8 Hz, 1 H, benzylH) ppm.
- HRMS: calcd for C20H18FN4O2S2 429.08552, found 429.08481.
- To a solution of 2-(1-(4-fluorophenyl)ethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.14 mmol) and triethylamine (30 µl, 0.21 mmol) in dioxane (1 ml) was added 2-(4-chlorophenoxy)-1-(piperazin-1-yl)ethanone (51 mg, 0.21 mmol). The reaction mixture was heated at 70°C for 3 hours. After cooling down to room temperature, the volatiles were removed under reduced pressure. The crude residue was purified by chromatography on silica gel (CH2Cl2/MeOH 70:1) furnishing the title compound as a white solid (50 mg, 67 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.29-7.25 (m, 4H, PhH), 7.02 (t, J= 8.1 Hz, 2H, PhH), 6.92 (d, J= 8.9 Hz, 2H, PhH), 4.74 (s, 4H, OCH2, NH2), 4.33 (q, J= 7.3 Hz, 1H, CH), 4.27 (br s, 4H, N(CH2)2), 3.71 (br s, 4H, CON(CH 2 )2), 1.73 (d, J= 7.3 Hz, 3H, CHCH 3 ) ppm.
- HRMS: calcd for C25H25ClFN6O2 527.14323, found 527.14230.
- This compound was synthesized from example 65 according to the procedure for the preparation of example 66. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 70:1) to yield the title compound as a white solid (60 mg, 83 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.17-7.28 (m, 7H, PhH), 6.91-7.05 (m, 6H, PhH), 4.74 (s, 2H, OCH2), 4.70 (s, 2H, NH2), 4.48 (t, J= 7.7 Hz, 1H, CH), 4.24 (br s, 4H, N(CH2)2), 3.72 (br s, 2H, CONCH 2 ), 3.66 (br s, 2H, CONCH 2 ), 3.62 (dd, J= 13.8, 7.7 Hz, 1H, benzylH), 3.26 (dd, J= 13.8, 7.8 Hz, 1 H, benzylH) ppm.
- HRMS: calcd for C31H29ClFN6O2S 603.17453, found 603.17371.
- This compound was synthesized according to the procedure for the preparation of example 66, using N-m-tolylpiperazine-1-carboxamide. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 50:1) furnishing the title compound as a white solid (76 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.50 (s, 1H, NH), 7.38-7.43 (m, 2H, PhH), 7.32 (s, 1H, PhH), 7.28 (d, J= 8.5 Hz, 1H, PhH), 7.18 (t, J= 8.9 Hz, 2H, PhH), 7.12 (t, J= 7.8 Hz, 1H, PhH), 6.76 (d, J= 7.4 Hz, 1 H, PhH), 6.33 (s, 2H, NH2), 4.51 (q, J= 7.1 Hz, 1 H, CH), 4.22 (br s, 4H, N(CH2)2), 3.57 (br s, 4H, CON(CH2)2), 2.25 (s, 3H, CH3), 1.67 (d, J= 7.1 Hz, 3H, CHCH 3 ) ppm.
- To a solution of 2-(4-fluorophenyl)thiazolo[5,4-d]pyrimidine-5,7-dithiol (0.20 g, 0.68 mmol) and triethylamine (0.33 ml, 2.37 mmol) in DMSO (3 ml) was added iodomethane (0.13 ml, 2.03 mmol). The reaction mixture was stirred for 12 hours under N2 at 25°C. The mixture was poured into water and extracted with EtOAc. The organic extracts were dried over Na2SO4 and the solvent was removed under reduced pressure. The crude residue was purified by flash chromatography on silica (EtOAc/Hex 1:40), yielding the title compound as a light yellow solid (0.19 g, 87 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.04-8.09 (m, 2H, PhH), 7.18 (t, J= 8.6 Hz, 2H, PhH), 2.70 (s, 3H, CH3), 2.66 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H11FN3S3 324.00991, found 324.00908.
- This compound was synthesized according to a procedure for the preparation of example 69, using n-butyl bromide. The crude residue was purified by flash chromatography on silica (EtOAc/Hex 1:100) to yield the title compound as a white solid (53 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.03-8.08 (m, 2H, PhH), 7.18 (t, J= 8.6 Hz, 2H, PhH), 3.34 (t, J =7.3 Hz, 2H, SCH2), 3.24 (t, J =7.3 Hz, 2H, SCH2), 1.73-1.82 (m, 4H, SCH2CH2CH2CH3), 1.48-1.55 (m, 4H, SCH2CH2CH 2 CH3), 0.98 (t, J= 7.4 Hz, 3H, CH3), 0.97 (t, J= 7.4 Hz, 3H, CH3) ppm.
- HRMS: calcd for C19H23FN3S3 408.10381, found 408.10259.
- This compound was synthesized according to a procedure for the preparation of example 69, using benzyl bromide. The crude residue was purified by flash chromatography on silica (EtOAc/Hex 1:100) to yield the title compound as a white solid (97 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.01-8.04 (m, 2H, PhH), 7.41-7.45 (m, 4H, PhH), 7.28-7.32 (m, 6H, PhH), 7.17 (t, J= 8.5 Hz, 2H, PhH), 4.54 (s, 2H, CH2), 4.49 (s, 2H, CH2) ppm. HRMS: calcd for C25H19FN3S3 476.0725, found 476.3023.
- To a solution of Na (2.0 mg, 0.08 mmol) in ethanol (1 ml) was added 2-(4-fluorophenyl)-5,7-bis(methylthio)thiazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol). The reaction mixture was heated at 80°C for 3 hours. After cooling, the mixture was neutralized with 1N HCl and the solvent was removed under reduced pressure. The crude residue was redissolved in ethyl acetate, extracted with brine and dried over Na2SO4. After removing the solvents in vacuo, the residue was purified by flash chromatography on silica (EtOAc/Hex 1:50), yielding the title compound as a white solid (30 mg, 60 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.02-8.07 (m, 2H, PhH), 7.17 (t, J= 8.6 Hz, 2H, PhH), 4.71 (q, J= 7.1 Hz, 2H, OCH2), 2.63 (s, 3H, SCH3) 1.54 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C14H13FN3OS2 322.04841, found 322.04753.
- This compound was synthesized according to a procedure for the preparation of example 21. The crude residue was purified by flash chromatography on silica (EtOAc/Hex 1:30) to yield the title compound as a white solid (61 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.11-8.16 (m, 2H, PhH), 7.23 (t, J= 8.6 Hz, 2H, PhH), 4.84 (q, J= 7.1 Hz, 2H, CH2), 1.61 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C14H13FN3O3S2 354.03824, found 354.03748.
- To a solution of diethyl 2-aminomalonate hydrochloride (4.23 g, 20 mmol) in pyridine (50 ml) was added an acid chloride (20 mmol). The resulting mixture was stirred at room temperature for 1 hour. The solvents were evaporated in vacuo. The residue was collected, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure:
- This compound was synthesized in 91% yield, using cyclopropanecarbonyl chloride.
- MS m/z (%): 244 ([M+H]+, 100)
- This compound was synthesized from 2-methoxyacetyl chloride in 97 % yield.
- MS m/z (%): 248 ([M+H]+, 100)
- This compound was synthesized from hexanoyl chloride in 95 % yield.
- MS m/z (%): 274 ([M+H]+, 100)
- Sodium (0.58 g, 25 mmol) was added to absolute ethanol (50 ml). After sodium was completely dissolved, diethyl 2-(acylamino)malonate (2.43 g, 10 mmol) and guanidine hydrochloride (1.20 g, 12.5 mmol) were added. The resulting mixture was heated under reflux for 1.5 h. The reaction mixture was cooled down to room temperature. The precipitate was collected by filtration, and washed with ethanol. The precipitate was then dissolved in a small amount of water and neutralized to pH = 3-4 with acetic acid. The precipitate was collected, washed with water and dried over P2O5, yielding the title compounds
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from diethyl 2-(cyclopropanecarboxamido)malonate in 91 % yield.
- MS m/z (%): 209 ([M-H]-, 100)
- This compound was synthesized from diethyl 2-(2-methoxyacetamido)malonate in 65 % yield.
- MS m/z (%): 213 ([M-H]-, 100)
- This compound was synthesized from diethyl 2-hexanamidomalonate (2.73 g, 10 mmol) in 83% yield.
- MS m/z (%): 239 ([M-H]-, 100)
- To an ice-cooled solution of 2,5-diaminopyrimidine-4,6-diol (1.78 g, 10 mmol) in 1 N NaOH (25 ml), was added slowly an appropriate acid chloride (10 mmol). The resulting mixture was stirred and warmed to room temperature over 1 hour. The reaction mixture was neutralized with HCl to pH 5. The precipitate was filtered off, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this general procedure:
- This compound was synthesized from cyclohexane carbonyl chloride in 87 % yield.
- MS m/z (%): 253 ([M+H]+, 100)
- This compound was synthesized from nicotinoyl chloride hydrochloride in 81 % yield.
- MS m/z (%): 248 ([M+H]+, 100)
- This compound was synthesized from 3-phenylpropanoyl chloride in 96 % yield.
- MS m/z (%): 275 ([M+H]+, 100)
- This compound was synthesized from 4-chlorophenylacetyl chloride yielding the title compound in 95 % yield.
- MS m/z (%): 295 ([M+H]+, 100)
- This compound was synthesized from 3,4-dichlorobenzoyl chloride yielding the title compound in 89 % yield.
- MS m/z (%): 315 ([M+H]+, 100)
- This compound was synthesized from 3-methoxybenzoyl chloride, yielding the title compound in 94 % yield.
- MS m/z (%): 277 ([M+H]+, 100)
- This compound was synthesized from ethyl chloroformate yielding the title compound in 89 % yield.
- A suspension of 1-phenylcyclopropanecarboxylic acid (973 mg, 6.0 mmol) in SOCl2 (5 ml) was heated under reflux for 1 h. After concentration under reduced pressure, the residue was redissolved in dioxane (5 ml) and added to a stirring solution of 2,5-diamino-4,6-dihydroxypyrimidine (893 mg, 5.0 mmol) in 1 N NaOH (20 ml) at 0°C. The mixture was stirred and warmed to room temperature in 1 h. After neutralization with 1 N hydrochloride to pH = 5, the precipitate was filtered off, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure:
- This compound was synthesized using 1-phenylcyclopropanecarboxylic acid, yielding the title compound in 87% yield.
- MS m/z (%): 285 ([M-H]-, 100)
- This compound was synthesized from 1-(4-chlorophenyl)cyclopropanecarboxylic acid yielding the title compound in 84 % yield.
- MS m/z (%): 321 ([M+H]+, 100)
- A suspension of the appropriate 2-amino-4,6-dihydroxy-5-(acylamino)pyrimidine analogue (630 mg, 3 mmol) and P2S5 (1.33 g, 6 mmol) in pyridine (15 ml) was heated under reflux for 6 hours. After concentration under reduced pressure, the residue was resuspended in 20 ml water. Sodium carbonate (1.27 g, 9 mmol) was added and the mixture was stirred at room temperature for 1 h. The precipitate was collected by filtration and washed with water, yielding the crude title compound which was used without further purification (450 mg, 67%). The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(acylamino)pyrimidine in 67 % yield.
- MS m/z (%): 223 ([M-H]-, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3-phenylpropanamido)pyrimidine (550 mg, 2.0 mmol), yielding the title compound (530 mg, 92%).
- MS m/z (%): 289 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3-nicotinamido)pyrimidine (494 mg, 2.0 mmol), yielding the title compound (260 mg, 50%).
- MS m/z (%): 262 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-cyclohexanecarboxamidopyrimidine (504 mg, 2.0 mmol), yielding the title compound (400 mg, 75%).
- MS m/z (%): 267 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(4-chlorophenylacetamido)pyrimidine (589 mg, 2.0 mmol), yielding the title compound (500 mg, 81%).
- MS m/z (%): 309 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5(4-chlorobenzamido)pyrimidine (560 mg, 2.0 mmol), yielding the title compound (450 mg, 76%).
- MS m/z (%): 295 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5(4-methoxybenzamido)pyrimidine (1.10 g, 4.0 mmol), yielding the title compound (1.0 g, 86%).
- MS m/z (%): 291 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(3,4-dichlorobenzamido)pyrimidine (1.26 g, 4.0 mmol), yielding the title compound (1.1 g, 83%).
- MS m/z (%): 329 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(1-phenylcyclopropanecarboxamido)pyrimidine (1.15 g, 4.0 mmol), yielding the title compound (1.1 g, 92%).
- MS m/z (%): 301 ([M+H]+, 100)
- This compound was synthesized from 2-amino-4,6-dihydroxy-5-(1-phenylcyclopropanecarboxamido)pyrimidine (1.28 g, 4.0 mmol), yielding the title compound (1.1 g, 82%).
- MS m/z (%): 335 ([M+H]+, 100)
- To a suspension of an appropriate 5-amino-2-substituted-thiazolo[5,4-d]pyrimidine-7-thiol (340 mg, 1.5 mmol) in 1N NaOH (10 ml), was added Mel (112 µl, 1.8 mmol). The resulting mixture was stirred at room temperature for 30 minutes. The reaction mixture was extracted with dichloromethane and washed with water and brine. The organic layer was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/50), yielding the pure title compounds.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 5-amino-2-cyclopropyl-thiazolo[5,4-d]pyrimidine-7-thiol in 92 % yield.
- MS m/z (%): 239 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(2-phenylethyl)thiazolo[5,4-d]pyrimidine-7-thiol (288 mg, 1.0 mmol), yielding the pure title compound (190 mg, 63%).
- MS m/z (%): 303 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-cyclohexylthiazolo[5,4-d]pyrimidine-7-thiol (133 mg, 0.5 mmol), yielding the pure title compound (80 mg, 57%).
- MS m/z (%): 281 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(3-pyridinyl)thiazolo[5,4-d]pyrimidine-7-thiol (261 mg, 1.0 mmol), yielding the pure title compound (150 mg, 54%).
- MS m/z (%): 276 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(4-chlorobenzyl)thiazolo[5,4-d]pyrimidine-7-thiol (308 mg, 1.0 mmol), yielding the pure title compound (100 mg, 31%).
- MS m/z (%): 323 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(3-methoxyphenyl)thiazolo[5,4-d]pyrimidine-7-thiol (290 mg, 1.0 mmol), yielding the pure title compound (250 mg, 82 %).
- MS m/z (%): 305 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(4-chlorophenyl)thiazolo[5,4-d]pyrimidine-7-thiol (443 mg, 1.5 mmol), yielding the pure title compound (380 mg, 82%).
- MS m/z (%): 309 ([M+H]+, 100)
- To an ice-cooled suspension of 5-amino-2-cyclopropyl-7-methylthiothiazolo[5,4-d]pyrimidine (120 mg, 0.5 mmol) in dichloromethane (5 ml), was added mCPBA (1250 mg, 1.0 mmol). The resulting mixture was stirred at 0°C for 1 hour and then warmed to room temperature. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of acetone and dichloromethane (in a ratio of 1/30), yielding the pure title compound (81 mg, 60%).
- MS m/z (%): 271 ([M+H]+, 100)
- A mixture of 5-amino-2-cyclopropyl-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidine (54 mg, 0.2 mmol) and K2CO3 (69 mg, 0.5 mmol) in dioxane (10 ml) and methanol (5 ml) was stirred at room temperature overnight. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/60), yielding the pure title compound (40 mg, 90%).
- MS m/z (%): 223 ([M+H]+, 100)
- To a solution of 5-amino-2-cyclopropyl-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidine (54 mg, 0.2 mmol) in dioxane (5 ml) was added piperazine (86 mg, 1.0 mmol). The resulting mixture was stirred at room temperature for 12 h. The solvents were evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/5), yielding the pure title compound (50 mg, 91%).
- MS m/z (%): 277 ([M+H]+, 100)
- A mixture of a 5-amino-2-substituted thiazolo[5,4-d]pyrimidine-7-thiol analogue (4.0 mmol) and piperazine (1.72 g, 20 mmol) in 1,1,1,3,3,3-hexamethyldisilazane (HMDS, 5 ml) and pyridine (20 ml) was heated under reflux for 12 hours. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/5), yielding the pure title compounds.
- The following compounds were made according to this procedure:
- This compound was synthesized from 5-amino-2-(3,4-chlorophenyl)thiazolo[5,4-d]pyrimidine-7-thiol in 51 % yield.
- MS m/z (%): 381 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)thiazolo[5,4-d]pyrimidine-7-thiol (600 mg, 2.0 mmol), yielding the pure title compound (450 mg, 64%).
- MS m/z (%): 353 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)thiazolo[5, 4-d]pyrimidine-7-thiol (1.34 g, 4.0 mmol), yielding the pure title compound (1.1 g, 71%).
- MS m/z (%): 387 ([M+H]+, 100)
- A suspension of ethyl 2-amino-4,6-dihydroxypyrimidin-5-ylcarbamate (0.43 g, 2.0 mmol) and P2S5 (1.33 g, 6 mmol) in pyridine (20 ml) was heated under reflux for 6 hours. After concentration under reduced pressure, the residue was suspended in water (20 ml). Sodium carbonate (1.27 g, 9 mmol) was added and the mixture was stirred at room temperature for 1 h. The reaction mixture was neutralized to pH = 5-6, the precipitate was filtered off and washed with water. The crude product was dissolved in 1 N NaOH (20 ml) and Mel (120 µl, 2.0 mmol) was added. The resulting mixture was stirred at room temperature for 30 minutes. The reaction mixture was extracted with dichloromethane and washed with water and brine. The organic layer was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/50), yielding the pure title compound (210 mg, 49%).
- MS m/z (%): 215 ([M+H]+, 100)
- To a suspension of 5-amino-7-oxo-2-thiomethyl-thiazolo[5,4-d]pyrimidine (107 mg, 0.5 mmol) in DMF (5 ml) were added piperazine (215 mg, 2.5 mmol), benzotriazol-1-yloxytris(dimethylamino)-phosphonium hexafluorophosphate (BOP, 290 mg, 0.66 mmol) and 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU, 120 µl, 0.79 mmol), respectively. The resulting reaction mixture was stirred at room temperature for 6 h. The reaction mixture was diluted with dichloromethane and washed with water and brine. The organic phase was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/8), yielding the pure title compound (130 mg, 82%).
- MS m/z (%): 283([M+H]+, 100)
- To a solution of a 5-amino-2-substituted-7-N-piperazino-thiazolo[5,4-d]pyrimidine analogue (260 mg, 1.0 mmol) in dioxane (10 ml), was added DIPEA (330 µl, 2.0 mmol) and 4-chlorophenoxyacetyl chloride (246 mg, 1.2 mmol) respectively. The resulting reaction mixture was stirred at room temperature for 30 minutes. The mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/40), yielding the pure title compounds.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 5-amino-2-(1-phenylcyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine (35 mg, 0.1 mmol), yielding the pure title compound (49 mg, 92%).
- MS m/z (%): 521 ([M+H]+, 100)
- This compound was synthesized from 5-amino-7-(N-piperazino)-2-methylthio-thiazolo[5,4-d]pyrimidine (113 mg, 0.36 mmol), yielding the pure title compound (120 mg, 75%).
- MS m/z (%): 451 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-(3,4-dichlorophenyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine (190 mg, 0.5 mmol), yielding the pure title compound (150 mg, 55%).
- 1H NMR (300 MHz, DMSO-d6, 25 °C): δ = 8.13 (s, 1H, ArH), 7.91 (d, J=8.6 Hz, 1H, ArH), 7.74 (d, J=8.6 Hz, 1 H, ArH), 7.32 (d, J= 9.0 Hz, 2H, ArH), 6.97 (d, J= 9.0 Hz, 2H, ArH), 4.93 (s, 2H, NH2), 4.90 (s, 2H, CH2), 4.42 (br s, 2H, NCH2), 4.22 (br s, 2H, NCH2), 3.64 (br s, 4H, NCH2) ppm.
- MS m/z (%): 549 ([M+H]+, 98)
- This compound was synthesized from 5-amino-2-(1-(4-chlorophenyl)cyclopropyl)-7-(N-piperazino)thiazolo[5,4-d]pyrimidine (39 mg, 0.1 mmol), yielding the pure title compound (50 mg, 89%).
- MS m/z (%): 555 ([M+H]+, 100)
- To a solution of a 5-amino-7-methylthio-2-substituted-thiazolo[5,4-d]pyrimidine analogue (76 mg, 0.25 mmol) in dichloromethane (10 ml) was added mCPBA (250 mg, 1.0 mmol). The solution was stirred at room temperature for 2 h. Then, a solution of piperazine (215 mg, 2.5 mmol) in dioxane (10 ml) was added. The resulting mixture was stirred at room temperature for another 2 h. The reaction mixture was diluted with dichloromethane and washed with water and brine. The organic phase was evaporated in vacuo and the residue was dissolved in dioxane (5 ml). Then, a solution of 4-chlorophenoxyacetyl chloride in dioxane (2 ml) was added. The mixture was stirred at room temperature for 1 h. The solvents were evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/50), yielding the pure title compound.
- The following compounds were made according to this procedure :
- This compound was made from 5-amino-7-methylthio-2-(2-phenylethyl)thiazolo[5, 4-d]pyrimidine in 71% yield.
- MS m/z (%): 509 ([M+H]+, 100)
- This compound was made from 5-amino-2-cyclopropyl-7-N-piperazino-thiazolo[5, 4-d]pyrimidine (15 mg, 0.05 mmol), yielding the pure title compound (20 mg, 83%).
- MS m/z (%): 445 ([M+H]+, 100)
- This compound was made from 5-amino-2-cyclohexyl-7-methylthiothiazolo[5,4-d]pyrimidine (56 mg, 0.2 mmol), yielding the pure title compound (60 mg, 62%).
- MS m/z (%): 487 ([M+H]+, 100)
- These compounds were prepared from 5-amino-7-(methylthio)-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine (69 mg, 0.25 mmol), yielding examples 121 a and 121 b.
- MS m/z (%): 482 ([M+H]+, 100)
and - MS m/z (%): 498 ([M+H]+, 100)
- This compound was made from 5-amino-2-(4-chlorophenylmethyl)-7-methylthiothiazolo[5,4-d]pyrimidine (100 mg, 0.31 mmol), yielding the pure title compound (140 mg, 85%).
- MS m/z (%): 529 ([M+H]+, 100)
- This compound was made from 5-amino-2-(4-chlorophenyl)-7-methylthiothiazolo[5,4-d]pyrimidine (154 mg, 0.5 mmol), yielding the pure title compound (190 mg, 74%).
- 1H NMR (300 MHz, DMSO-d6, 25 °C): δ = 7.94(d, J= 9.0 Hz, 2H, ArH), 7.58 (d, J=9.0 Hz, 2H, ArH), 7.34 (d, J= 9.0 Hz, 2H, ArH), 6.99 (d, J= 9.0 Hz, 2H, ArH), 6.55 (s, 2H, NH2), 4.93 (s, 2H, CH2), 4.35 (br s, 2H, NCH2), 4.22 (br s, 2H, NCH2), 3.64 (br s, 4H, NCH2) ppm.
- MS m/z (%): 515 ([M+H]+, 100)
- This compound was made from 5-amino-2-(3-methoxyphenyl)-7-methylthiothiazolo[5,4-d]pyrimidine (152 mg, 0.5 mmol), yielding the pure title compound (120 mg, 47%).
- 1H NMR (300 MHz, DMSO-d6, 25 °C): δ = 7.51-7.41 (m, 3H, ArH), 7.32 (d, J=9.0 Hz, 2H, ArH), 7.09 (m, 1 H, ArH), 6.97 (d, J= 9.0 Hz, 2H, ArH), 6.52 (s, 2H, NH2), 4.93 (s, 2H, CH2), 4.36 (br s, 2H, NCH2), 4.22 (br s, 2H, NCH2), 3.84 (s, 3H, OCH3), 3.64 (br s, 4H, NCH2) ppm.
- MS m/z (%): 511 ([M+H]+, 100)
- To a suspension of 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-(4-chlorophenylmethyl)thiazolo[5,4-d]pyrimidine (53 mg, 0.1 mmol) in DMF (2 ml) was added 1 N NaOH (150 µl, 0.15 mmol) and Mel (7 µl, 0.11 mmol). The resulting mixture was stirred at room temperature for 1 hour. The reaction mixture was diluted with dichloromethane and washed with water and brine. The organic layer was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/80), yielding the pure title compound (40 mg, 74%).
- MS m/z (%): 543 ([M+H]+, 100)
- To an ice cooled suspension of 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methylthio-thiazolo[5,4-d]pyrimidine (90 mg, 0.2 mmol) in dichloromethane (5 ml) was added mCPBA (125 mg, 0.5 mmol). The resulting mixture was stirred at 0°C for 2 h. The solvents were evaporated in vacuo and the crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/80), yielding the pure title compound (65 mg, 70%).
- MS m/z (%): 467 ([M+H]+, 100)
- A mixture of 5-amino-7-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-2-methylsulfinyl-thiazolo[5,4-d]pyrimidine (47 mg, 0.1 mmol) and 4-fluoroaniline (95 µl, 1 mmol) in dioxane (5 ml) was heated under reflux for 12 hours. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/80), yielding the pure title compound (45 mg, 88%).
- MS m/z (%): 514 ([M+H]+, 100)
- To a solution of 5-amino-2-(4-fluorophenyl)-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol) and a 2-aryloxyacetic acid derivative (0.23 mmol) in DMF (2 ml) was added TBTU (0.23 mmol) followed by diisopropylethylamine (0.23 mmol, 37 µL). The reaction was stirred at room temperature for 24 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were synthesized according to this procedure :
- This compound was obtained from 2-(4-bromophenoxy)acetic acid (43 mg);
- MS m/z (%): 543 ([M+H]+, 100).
- This compound was obtained from 2-(3-nitrophenoxy)acetic acid (49 mg).
- MS m/z (%): 510 ([M+H]+, 100).
- To a solution of 5-amino-2-(4-fluorophenyl)-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol) in DMF (2 ml) was added diisopropylethylamine (0.33 mmol, 55 µL) followed by phenoxyacetyl chloride (0.17 mmol). The reaction was stirred at room temperature for 4 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 0.5% CH3OH in CH2Cl2), yielding the pure title compound (30 mg) which was characterized by its mass spectrum: MS m/z (%) : 465 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) and 3-nitrophenoxyacetic acid (0.23 mmol) in DMF (2 ml) was added
- TBTU (0.21 mmol) followed by diisopropylethylamine (0.21 mmol, 35 µL). The reaction was stirred at room temperature for 24 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 0.5% CH3OH in CH2Cl2), yielding the pure title compound (41 mg) which was characterized by its mass spectrum: MS m/z (%): 538 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) and 4-chlorophenylacetic acid (0.21 mmol) in DMF (2 ml) was added TBTU (0.21 mmol) followed by diisopropylethylamine (0.21 mmol, 35 µL). The reaction was stirred at room temperature for 24 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (44 mg) which was characterized by its mass spectrum: MS m/z (%): 511 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in dichloromethane (4 ml) was added 3-methylphenylisocyanate (0.15 mmol). The reaction was stirred at room temperature for 2 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (28 mg) which was characterized by its mass spectrum: MS m/z (%) : 492 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in dichloromethane (4 ml) was added diisopropylethylamine (0.33 mmol, 55 µL) followed by phenoxyacetyl chloride (0.17 mmol). The reaction was stirred at room temperature for 4 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (34 mg) which was characterized by its mass spectrum: MS m/z (%): 493 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in dichloromethane (4 ml) was added diisopropylethylamine (0.33 mmol, 55 µL) and 4-chlorobenzoyl chloride (0.15 mmol). The reaction was stirred at room temperature for 16 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (31 mg) which was characterized by its mass spectrum: MS m/z (%): 497 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (80 mg, 0.22 mmol) in dichloromethane (4 ml) was added diisopropylethylamine (0.49 mmol, 81 µL) and 3-phenylpropionyl chloride (0.25 mmol). The reaction was stirred at room temperature for 16 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (43 mg) which was characterized by its mass spectrum: MS m/z (%): 491 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-thiazolo[5,4-d]pyrimidine (80 mg, 0.22 mmol) in 1,4-dioxane (4 ml) was added diisopropylethylamine (0.49 mmol, 81 µL) and phenylmethanesulfonyl chloride (0.25 mmol). The reaction was stirred at 90°C for 16 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (13 mg) which was characterized by its mass spectrum: MS m/z (%) : 513 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in dichloromethane (4 ml) was added homopiperazine (1.4 mmol). The reaction mixture was stirred at room temperature for 2 hours. The mixture was extracted, the organic phase was dried over MgSO4 after which the solvent was removed in vacuo. The residue was dissolved in dichloromethane whereupon diisopropylethylamine (0.28 mmol, 47 µL) followed by 4-chlorophenoxyacetyl chloride (0.14 mmol) were added. The reaction was stirred at room temperature for 24 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (38 mg) which was characterized by its mass spectrum: MS m/z (%): 541 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (100 mg, 0.28 mmol) in dichloromethane (5 ml) was added diisopropylethylamine (0.59 mmol, 98 µL) and a piperazine derivative (0.31 mmol). The reaction mixture was stirred at room temperature for 18 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were made according to this procedure :
- This compound was obtained from N-methyl-N-phenyl-2-piperazin-1-yl-acetamide (62 mg). MS m/z (%) : 506 ([M+H]+, 100).
- This compound was obtained from 4-thiazol-2-yl-piperazine (65 mg).
- MS m/z (%) : 442 ([M+H]+, 100).
- This compound was obtained from 4-(phenethylcarbamoyl-methyl)piperazine (84 mg).
- MS m/z (%) : 520 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (150 mg, 0.42 mmol) in dichloromethane (10 ml) was added diisopropylethylamine (0.94 mmol, 155 µL) and 3-(R)-tert-(butoxycarbonylamino)pyrrolidine (0.47 mmol). The reaction mixture was stirred at room temperature for 24 hours. The reaction mixture was extracted with a saturated sodium bicarbonate solution and the organic phase was collected and dried over magnesium sulfate. The solvent was removed in vacuo and the resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (168 mg) which was characterized by its mass spectrum: MS m/z (%) : 459 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-((3-(R)-tert-butoxycarbonylamino)pyrrolidin-1-yl)-thiazolo[5,4-d]pyrimidine (70 mg, 0.15 mmol) in dichloromethane (3 ml) was added trifluoroacetic acid (3 ml). The reaction mixture was stirred at room temperature for 2 hours after which the solvents were removed in vacuo. The residue was dissolved in dichloromethane (3 ml) and diisopropylethylamine (1.5 mmol, 252 µL) and an acyl chloride (0.17 mmol) were added. The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1.5% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were made according to this procedure :
- This compound was obtained from 4-chlorophenoxyacetyl chloride (43 mg);
- MS m/z (%) : 527 ([M+H]+, 100).
- This compound was obtained from 4-chlorobenzoyl chloride (44 mg).
- MS m/z (%) : 497 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in 1,4-dioxane (5 ml) was added potassium carbonate (0.42 mmol) and benzyl 3-aminopiperidine-1-carboxylate hydrogen chloride (0.15 mmol). The reaction mixture was stirred at room temperature for 24 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (59 mg) which was characterized by its mass spectrum: MS m/z (%): 507 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (150 mg, 0.42 mmol) in dichloromethane (10 ml) was added diisopropylethylamine (3.0 mmol, 492 µL) and tert-butyl 3-(S)-aminopyrrolidine-1-carboxylate (2.1 mmol). The reaction mixture was refluxed for 40 hours whereupon the mixture was extracted with a saturated sodium bicarbonate solution. The organic phase was dried over magnesium sulfate and the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (81 mg) which was characterized by its mass spectrum: MS m/z (%) : 459 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(1-tert-butoxycarbonyl-pyrrolidin-3-(S)-ylamino)-thiazolo[5,4-d]pyrimidine (70 mg, 0.15 mmol) in dichloromethane (1 ml) was added trifluoroacetic acid (1 ml). The reaction mixture was stirred at room temperature for 2 hours after which the solvents were removed in vacuo. The residue was dissolved in dichloromethane (3 ml) and diisopropylethylamine (1.5 mmol, 252 µL) and 4-chlorophenoxyacetyl chloride (0.17 mmol) were added. The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1.5% CH3OH in CH2Cl2), yielding the pure title compound (37 mg) which was characterized by its mass spectrum: MS m/z (%): 527 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-methanesulfonyl-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in acetonitrile (5 ml) was added diisopropylethylamine (0.45 mmol, 74 µL) and 4-benzoylpiperidine hydrogen chloride (0.15 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound (59 mg) which was characterized by its mass spectrum: MS m/z (%): 462 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-6H-thiazolo[5,4-d]pyrimidin-7-one (133 mg, 0.46 mmol) in DMF (5 ml) was added DBU (0.69 mmol), BOP (0.59 mmol) and 1-(2-phenoxyethyl)piperazine (1.37 mmol). The reaction mixture was stirred at room temperature for 3 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (56 mg) which was characterized by its mass spectrum: MS m/z (%) : 479 ([M+H]+, 100).
- To a solution of 5-amino-2-(4-fluorobenzyl)-7-methylsulfanylthiazolo[5,4-d]pyrimidine (100 mg, 0.33 mmol) in DMSO (1.5 ml) was added sodium hydroxide (2N, 171 µL) and ethyl iodide (0.34 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the mixture was extracted with ethyl acetate and brine. The organic phase was dried over magnesium sulfate and concentrated by evaporation in vacuo. The resulting residue was dissolved in dichloromethane (3 ml), cooled to 0°C and m-chloroperoxybenzoic acid (0.81 mmol) was added. The reaction mixture was stirred at room temperature for 7 hours. The mixture was first extracted with a saturated sodium bicarbonate solution, then with brine. The combined organic phases were dried over magnesium sulfate. After evaporating the solvents in vacuo, the residue was dissolved again in dichloromethane (3 ml) and piperazine (3.3 mmol) was added. The reaction mixture was stirred at room temperature for 16 hours whereupon the mixture was extracted with brine. The organic phase was dried over magnesium sulfate and evaporated in vacuo. The crude residue was reacted at room temperature for 16 hours with 4-chlorophenoxyacetyl chloride (0.35 mmol) in the presence of diisopropylethylamine (0.72 mmol, 119µL). After removing the solvent in vacuo, the crude mixture was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1.5% CH3OH in CH2Cl2), yielding the pure title compound (30 mg) which was characterized by its mass spectrum: MS m/z (%): 541 ([M+H]+, 100).
- To a solution of 5-amino-2-(4-fluorobenzyl)-7-methylsulfanylthiazolo[5,4-d]pyrimidine (100 mg, 0.33 mmol) in DMSO (1.5 ml) was added sodium hydroxide (2N, 171 µL) and cyclopentyl iodide (0.34 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the mixture was extracted with ethyl acetate and brine. The organic phase was dried over magnesium sulfate and concentrated by evaporation in vacuo. The resulting residue was dissolved in dichloromethane (3 ml), cooled to 0°C and m-chloroperoxybenzoic acid (0.81 mmol) was added. The reaction mixture was stirred at room temperature for 7 hours. The mixture was first extracted with a saturated sodium bicarbonate solution, then with brine. The combined organic phases were dried over magnesium sulfate. After evaporating the solvents in vacuo, the residue was redissolved in dichloromethane (3 ml) and piperazine (3.3 mmol) was added. The reaction mixture was stirred at room temperature for 16 hours whereupon the mixture was extracted with brine. The organic phase was dried over magnesium sulfate and evaporated in vacuo. The crude residue was reacted at room temperature for 16 hours with 4-chlorophenoxyacetyl chloride (0.35 mmol) in the presence of diisopropylethylamine (0.72 mmol, 119 µL). After removing the solvent in vacuo, the crude mixture was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1.5% CH3OH in CH2Cl2), yielding the pure title compound (47 mg) which was characterized by its mass spectrum: MS m/z (%) : 581 ([M+H]+, 100).
- To a solution of 2,5-diaminopyrimidine-4,6-diol hydrogen chloride (1.04 g, 5.8 mmol) in sodium hydroxide (17.8 mmol) solution (25 ml of H2O) was added 3-thiophen-2-ylpropionyl chloride (6.4 mmol). The latter was prepared by refluxing 3-thiophen-2-ylpropionic acid (6.4 mmol) in thionyl chloride (500 µL) for 1 hour whereupon the excess of thionyl chloride was removed by evaporation in vacuo. The reaction mixture was stirred at room temperature for 18 hours. The pH of the suspension was adjusted to approximately 6 and the solids were filtered off. The title compound (1.25 g) was characterized by its mass spectrum: MS m/z (%) : 515 ([M+H]+, 100).
- To a suspension of N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-3-thiophen-2-yl-propionamide (example 153, 1 g, 3.6 mmol) in o-xylene (25 ml) was added phosphorus pentasulfide (5.3 mmol, P4S10). The reaction mixture was refluxed until all starting material was consumed (TLC monitoring) whereupon the mixture was cooled down to room temperature. The reaction was quenched by adding potassium carbonate (32 mmol) and the mixture was stirred at room temperature for one additional hour. The precipitate was filtered off and the solids were extensively washed with water and subsequently dried. Next, the solids were dissolved in pyridine (20 ml) and piperazine (17.8 mmol), ammonium sulfate (36 mg), p-toluenesulfonic acid (36 mg) and 1,1,1,3,3,3-hexamethyldisilazane (3.6 ml) were added. The reaction mixture was refluxed for 24 hours after which the solvent was evaporated in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 5% CH3OH in CH2Cl2), yielding the pure title compound (656 mg) which was characterized by its mass spectrum: MS m/z (%) : 347 ([M+H]+, 100).
- To a solution of 5-amine-7-piperazin-1-yl-2-(2-thiophen-2-yl-ethyl)-thiazolo[5,4-d]pyrimidine (example 154, 50 mg, 0.14 mmol) in dichloromethane (3 ml) was added diisopropylethylamine (35 mmol) and an acyl chloride (0.17 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were synthesized according to this procedure :
- This compound was obtained from 4-chlorophenoxyacetyl chloride (31 mg).
- MS m/z (%) : 515 ([M+H]+, 100).
- This compound was obtained from 4-chlorophenylacetic chloride (23 mg).
- MS m/z (%): 499 ([M+H]+, 100).
- This compound was obtained from 4-chlorobenzoyl chloride (29 mg).
- MS m/z (%): 485 ([M+H]+, 100).
- To a solution of 5-amine-7-piperazin-1-yl-2-(2-thiophen-2-yl-ethyl)-thiazolo[5,4-d]pyrimidine (50 mg, 0.14 mmol) in dichloromethane (3 ml) was added 3-methylphenylisocyanate (0.17 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 0.5% CH3OH in CH2Cl2), yielding the pure title compound (33 mg) which was characterized by its mass spectrum: MS m/z (%) : 480 ([M+H]+, 100).
- Formamidine acetate (0.46 g, 4.46 mmol) and dimethyl 2-(4-fluorobenzamido)malonate (1.0 g, 3.71 mmol) were added to a solution of sodium (0.17 g, 7.43 mmol) in ethanol (37 ml). The reaction mixture was refluxed for 3 hours. After cooling down, the precipitate was filtered off and washed with ethanol. The product was dissolved in a minimal volume of water and acidified to pH 4-5 with 5M HCl. The precipitate was collected, washed with water and dried to yield the title compound as a white solid (0.60 g, 64 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.03 (s, 2H, OH), 9.25 (s, 1H, CH), 8.01-8.05 (m, 3H, PhH, NH), 7.32 (t, J= 8.3 Hz, 2H, PhH) ppm.
- MS: 247.8 [M-H]
- This compound was prepared from example 2 in a yield of 24%, according to the procedure for the synthesis of example 158.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.00 (s, 2H, OH), 9.09 (s, 1 H, NH), 7.97 (s, 1 H, CH), 7.33-7.37 (m, 2H, PhH), 7.12 (t, J= 8.6 Hz, 2H, PhH), 3.59 (s, 2H, CH2) ppm.
- HRMS: calcd for C12H11FN3O3264.07844, found 264.07769.
- This compound was prepared from example 2 in a yield of 29%, according to the procedure for the synthesis of example 9.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 11.97 (s, 2H, OH), 8.89 (s, 1H, NH), 7.32-7.37 (m, 2H, PhH), 7.12 (t, J= 8.6 Hz, 2H, PhH), 3.56 (s, 2H, CH2), 2.24 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H13FN3O3278.09409, found 278.09331.
- To a solution of N-(4,6-dihydroxypyrimidin-5-yl)-4-fluorobenzamide (0.30 g, 1.20 mmol) in POCl3 (6 ml) was added diisopropylethylamine (0.42 ml, 2.41 mmol). The reaction mixture was stirred under N2 at 90°C for 3.5 hours. The reaction mixture was allowed to cool down to room temperature and the volatiles were evaporated to dryness. The residue was diluted with water and the aqueous phase was extracted with diethyl ether. The combined organic layers were washed with a saturated NaHCO3 solution and brine, dried over Na2SO4, and concentrated under reduced pressure. The residue was purified by chromatography on silica gel (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (0.13 g, 30 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.64 (s, 1H, CH), 8.23-8.27 (m, 2H, PhH), 7.25 (t, J= 8.2 Hz, 2H, PhH) ppm.
- This compound was prepared from example 9 in a yield of 40%, according to the procedure for the synthesis of example 161.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.28-8.33 (m, 2H, PhH), 7.26 (t, J= 8.6 Hz, 2H, PhH), 2.84 (s, 3H, CH3) ppm.
- HRMS: calcd for C12H8ClFN3O 264.03399, found 264.03318.
- This compound was prepared from example 5 in a yield of 28%, according to the procedure for the synthesis of example 161.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.13-8.18 (m, 2H, PhH), 7.48 (s, 2H, NH2), 7.45 (t, J= 8.9 Hz, 2H, PhH) ppm.
- HRMS: calcd for C11H7ClFN4O 265.02924, found 265.02851.
- This compound was prepared from example 159 in a yield of 73%, according to the procedure for the synthesis of example 160.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.77 (s, 1H, CH), 7.36-7.41 (m, 2H, PhH), 7.06 (t, J= 8.6 Hz, 2H, PhH), 4.32 (s, 2H, CH2) ppm.
- HRMS: calcd for C12H8ClFN3O 264.03399, found 264.03349.
- This compound was prepared from example 159 in a yield of 40%, according to the procedure for the synthesis of example 161.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.35-7.40 (m, 2H, PhH), 7.08 (t, J= 8.6 Hz, 2H, PhH), 4.14 (s, 2H, CH2), 2.68 (s, 3H, CH3) ppm.
- This compound was prepared from example 7 in a yield of 34%, according to the procedure for the synthesis of example 161.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.28-7.33 (m, 2H, PhH), 7.10 (t, J= 8.8 Hz, 2H, PhH), 6.43 (s, 2H, NH2), 3.16 (br s, 4H, CH 2 CH 2 ) ppm.
- This compound was prepared from example 161 in a yield of 60%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.34 (s, 1H, CH), 7.90-7.94 (m, 2H, PhH), 7.18-7.24 (m, 4H, PhH), 6.83 (d, J= 6.8 Hz, 2H, PhH), 4.66 (s, 2H, CH2), 3.77 (br s, 2H, NCH2), 3.68 (br s, 2H, NCH2), 3.65 (s, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C23H21ClFN6O3 483.13477, found 483.13334.
- This compound was prepared from example 162 in a yield of 93%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.13-8.18 (m, 2H, PhH), 7.27 (d, J= 8.9 Hz, 2H, PhH), 7.20 (t, J= 8.5 Hz, 2H, PhH), 6.93 (d, J= 8.9 Hz, 2H, PhH), 4.76 (s, 2H, CH2), 4.27 (br s, 4H, N(CH2)2), 3.78 (br s, 4H, CON(CH 2 )2), 2.59 (s, 3H, CH3) ppm.
- HRMS: calcd for C24H22ClFN5O3 482.13952, found 482.13800.
- This compound was prepared from example 163 in a yield of 55%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.03-8.08 (m, 2H, PhH), 7.40 (t, J= 8.8 Hz, 2H, PhH), 7.33 (d, J= 8.8 Hz, 2H, PhH), 6.98 (d, J= 8.8 Hz, 2H, PhH), 6.54 (s, 2H, NH2), 4.93 (s, 2H, CH2), 4.19 (br s, 2H, NCH2), 4.09 (br s, 2H, N(CH2)2), 3.62 (br s, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C23H21ClFN6O3 483.13477, found 483.13367.
- This compound was prepared from example 164 in a yield of 58%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.33 (s, 1H, CH), 7.30-7.35 (m, 2H, PhH), 7.26 (d, J= 6.8 Hz, 2H, PhH), 7.03 (t, J= 8.7 Hz, 2H, PhH), 6.91 (d, J= 6.8 Hz, 2H, PhH), 4.74 (s, 2H, OCH2), 4.17 (s, 2H, CH2), 4.17 (br s, 4H, N(CH2)2), 3.75 (br s, 4H, CON(CH 2 )2) ppm.
- HRMS: calcd for C24H22ClFN5O3 482.13952, found 482.13796.
- This compound was prepared from example 165 in a yield of 68%, according to the procedure for the synthesis of example 47..
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.25 (d, J= 8.9 Hz, 2H, PhH), 7.10-7.15 (m, 2H, PhH), 6.99 (t, J= 8.5 Hz, 2H, PhH), 6.86 (d, J= 8.9 Hz, 2H, PhH), 4.67 (s, 2H, OCH2), 3.66 (br s, 4H, N(CH2)2), 3.55 (s, 2H, CH2), 3.53 (br s, 4H, CON(CH 2 )2), 2.61 (s, 3H, CH3) ppm.
- This compound was prepared from example 166 in a yield of 34%, according to the procedure for the synthesis of example 47.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26 (d, J= 8.9 Hz, 2H, PhH), 7.14-7.19 (m, 2H, PhH), 6.96 (t, J= 8.7 Hz, 2H, PhH), 6.91 (d, J= 8.9 Hz, 2H, PhH), 4.78 (s, 2H, NH2), 4.73 (s, 2H, CH2), 4.11 (br s, 4H, N(CH2)2), 3.68 (br s, 4H, CON(CH 2 )2), 3.07 (br s, 4H, CH 2 CH 2 ) ppm.
- To a solution of 7-chloro-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidine (85 mg, 0.34 mmol) in 1,2-dichloroethane/t-BuOH (1:1, 2 ml) was added 3-chloro-4-fluoroaniline (50 mg, 0.34 mmol). The mixture was heated at 90°C for 1 day. After cooling down to room temperature, the solvent was removed under reduced pressure. The crude residue was purified by silica gel chromatography (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (0.1 g, 82 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.52 (s, 1H, NH), 8.55 (s, 1H, CH), 8.23-8.27 (m, 2H, PhH), 7.84-7.87 (m, 1 H, PhH), 7.50 (t, J= 8.8 Hz, 2H, PhH), 7.43 (t, J= 9.1 Hz, 1 H, PhH) ppm.
- HRMS: calcd for C17H10ClF2N4O 359.05112, found 359.05016.
- This compound was prepared from example 163 in a yield of 32%, according to the procedure for the synthesis of example 173.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.00 (s, 1H, NH), 8.22-8.26 (m, 1H, PhH), 8.08-8.12 (m, 2H, PhH), 7.95-7.99 (m, 1 H, PhH), 7.44 (t, J= 8.8 Hz, 2H, PhH), 7.35 (t, J= 9.1 Hz, 1 H, PhH), 6.81 (s, 2H, NH2) ppm.
- HRMS: calcd for C17H11ClF2N5O 374.06202, found 374.06101.
- To a suspension of a 2-amino-4,6-dihydroxy-5-substituted pyrimidine analogue (3.0 mmol) in toluene (20 ml) were added DIPEA (1.19 ml, 9 mmol) and POCl3 (830 µl, 9 mmol) respectively. The resulting mixture was heated at 95°C for 2 h. The solvents were evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of acetone and dichloromethane (in a ratio of 1/50), yielding the pure title compounds.
- The following compounds were synthesized according to this procedure :
- This compound was obtained from 2-amino-4,6-dihydroxy-5-(cyclopropanamido)pyrimidine (630 mg, 3.0 mmol) yielding the pure title compound (550 mg, 87%).
- MS m/z (%): 211 ([M+H]+, 100)
- From 2-amino-4, 6-dihydroxy-5-methoxyacetamidopyrimidine (642 mg, 3.0 mmol), yielding the pure title compound (430 mg, 67%).
- MS m/z (%): 215 ([M+H]+, 100)
- From 2-amino-4, 6-dihydroxy-5-cyclohexanecarboxamidopyrimidine (760 mg, 3.0 mmol), yielding the pure title compound (390 mg, 51%).
- MS m/z (%): 253 ([M+H]+, 100)
- From 2-amino-4, 6-dihydroxy-5-hexanamidopyrimidine (720mg, 3.0 mmol), yielding the pure title compound (480 mg, 67%).
- MS m/z (%): 241 ([M+H]+, 100)
- Example 179 : Synthesis of 5-amino-7-chloro-2-(2-phenylethyl)oxazolo[5,4-d]pyrimidine
- From 2-amino-4,6-dihydroxy-5-phenylpropanamidopyrimidine (686 mg, 2.5 mmol), yielding the pure title compound (320 mg, 46%).
- MS m/z (%): 276 ([M+H]+, 100)
- To a solution of the 5-amino-7-chloro-2-substituted oxazolo[5,4-d]pyrimidine (420 mg, 2.0 mmol) in dioxane (10 ml), was added piperazine (860 mg, 10 mmol). The resulting mixture was stirred at room temperature for 12 h. The mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/6), yielding the pure title compounds.
- The following compounds were synthesized according to this general procedure:
- Example 180 : Synthesis of 5-amino-2-cyclopropyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine
- This compound was obtained in 92 % yield from 5-amino-7-chloro-2-cyclopropyl-oxazolo[5,4-d]pyrimidine.
- MS m/z (%): 261 ([M+H]+, 100)
- This compound was obtained in 57 % yield from 5-amino-7-chloro-2-methoxymethyloxazolo[5, 4-d]pyrimidine.
- MS m/z (%): 265 ([M+H]+, 100)
- This compound was obtained in 97 % yield from 5-amino-7-chloro-2-cyclohexyloxazolo[5, 4-d]pyrimidine.
- MS m/z (%): 303 ([M+H]+, 100)
- This compound was obtained in 86 % yield from 5-amino-7-chloro-2-pentyloxazolo[5, 4-d]pyrimidine.
- MS m/z (%): 291 ([M+H]+, 100)
- This compound was obtained in 99 % yield from 5-amino-7-chloro-2-(2-phenylethyl)oxazolo[5,4-d]pyrimidine.
- MS m/z (%): 325 ([M+H]+, 100)
- To a solution of a 5-amino-2-substituted-7-N-piperazino-oxazolo[5,4-d]pyrimidine analogue (1.0 mmol) in dioxane (10 ml), was added DIPEA (330 µl, 2.0 mmol) and 4-chlorophenoxyacetyl chloride (246 mg, 1.2 mmol), respectively. The resulting mixture was stirred at room temperature for 30 minutes. The solvents were evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/40), yielding the pure title compounds.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 5-amino-2-cyclopropyl-7-N-piperazino-oxazolo[5, 4-d]pyrimidine in 93 % yield.
- MS m/z (%): 429 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-methoxymethyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 60 % yield.
- MS m/z (%): 433 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-cyclohexyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 73 % yield.
- MS m/z (%): 471 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-pentyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 71 % yield.
- MS m/z (%): 459 ([M+H]+, 100)
- This compound was synthesized from 5-amino-2-pentyl-7-N-piperazino-oxazolo[5,4-d]pyrimidine in 85 % yield.
- MS m/z (%): 493 ([M+H]+, 100)
- To a solution of 5-amino-7-chloro-2-(4-fluorophenyl)-oxazolo[5,4-d]pyrimidine X (50 mg, 0.19 mmol) in dioxane (5 ml) was added diisopropylethylamine (0.28 mmol, 47 µL) and a piperazine derivative (0.28 mmol). The reaction was stirred at 70°C for 6 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below:
- The following compounds were synthesized according to this procedure :
- Example 190 : Synthesis of 5-amino-2-(4-fluorophenyl)-7-(4-isobutylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
This compound was obtained from 1-isobutyl-piperazine (32 mg);
MS m/z (%): 371 ([M+H]+, 100). - Example 191 : Synthesis of 5-amino-2-(4-fluorophenyl)-7-(4-acetylpiperazin-1-yl)-oxazolo[5,4-d]pyrimidine
This compound was obtained from 1-acetyl piperazine (27 mg).
MS m/z (%): 357 ([M+H]+, 100). - Example 192 : Synthesis of 5-amino-2-(4-fluorophenyl)-7-[4-(2-methoxyethyl)-piperazin-1-yl]-oxazolo[5,4-d]pyrimidine
This compound was obtained from 1-(2-methoxyethyl) piperazine (30 mg).
MS m/z (%): 373 ([M+H]+, 100). - To a solution of 5-amino-2-(4-fluorophenyl)-7-piperazin-1-yl-oxazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol) and 2-(3-nitrophenoxy)acetic acid (0.22 mmol) in DMF (2 ml) was added TBTU (0.22 mmol) followed by diisopropylethylamine (0.22 mmol, 36 µL). The reaction was stirred at room temperature for 24 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compound which was characterized by its mass spectrum: MS m/z (%) : 522 ([M+H]+, 100).
- To a solution of 5-amino-2-(4-fluorophenyl)-7-piperazin-1-yl-oxazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol) in dichloromethane (4 ml) was added diisopropylethylamine (0.32 mmol, 53 µL) followed by an acyl chloride (0.16 mmol). The reaction was stirred at room temperature for 16 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 1% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were synthesized according to this procedure :
- This compound was obtained from 4-chlorophenylacetyl chloride (57 mg);
- MS m/z (%) : 495 ([M+H]+, 100).
- This compound was obtained from 4-chlorobenzoyl chloride (62 mg).
- MS m/z (%) : 481 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-piperazin-1-yl-oxazolo[5,4-d]pyrimidine (50 mg, 0.15 mmol) in dichloromethane (4 ml) was added 3-methylphenylisocyanate (0.16 mmol). The reaction was stirred at room temperature for 2 hours after which the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (30 mg) which was characterized by its mass spectrum: MS m/z (%) : 476 ([M+H]+, 100).
- To a solution of 5-amino-2-[2-(4-fluorophenyl)ethyl]-6H-oxazolo[5,4-d]pyrimidin-7-one (50 mg, 0.18 mmol) in DMF (2 ml) was added DBU (0.27 mmol), BOP (0.23 mmol) and a piperazine derivative (0.23 mmol). The reaction mixture was stirred at room temperature for 16 hours whereupon the solvent was removed in vacuo. The resulting residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compounds which were characterized by their mass spectra as indicated below.
- The following compounds were made according to this general procedure :
- Example 197 : Synthesis of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-(2-phenoxyethyl)piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
This compound was obtained from 1-(2-phenoxyethyl)-piperazine (57 mg).
MS m/z (%) : 463 ([M+H]+, 100). - Example 198 : Synthesis of 5-amino-2-[2-(4-fluorophenyl)ethyl]-7-(4-[(methylphenyl-carbamoyl)methyl]piperazin-1-yl)-oxazolo[5,4-d]pyrimidine
This compound was obtained from N-methyl-N-phenyl-2-piperazin-1-yl-acetamide (27 mg). MS m/z (%) : 371 ([M+H]+, 100). - To a solution of 2,5-diamino-4,6-dihydroxypyrimidine (1 g, 5.6 mmol) in water (15 ml) was added sodium hydroxide (17 mmol, 672 mg) and an appropriate acid chloride (6.72 mmol) at 0 °C. The reaction was then stirred at room temperature for 3 hours. The reaction mixture was acidified till pH = 5. A light pink precipitate was formed, which was filtered off yielding the pure title compounds, in yields varying from 85-95%.
- The following compounds were made according to this procedure:
- This compound was synthesized according to the general procedure using benzoyl chloride. 1H NMR (300 MHz, DMSO): δ = 10.82 (br s, 2H, 2 x OH), 8.73 (s, 1 H, arom H), 7.9 (m, 2H, arom H), 7.45 (m, 3H, arom H), 6.97 (br s, 2H, NH2) ppm.
- This compound was synthesized according to the general procedure using 2-furoyl chloride 1H NMR (300 MHz, DMSO): δ = 10.67 (br s, 2H, 2 x OH), 8.56 (s, 1 H, arom H), 7.81 (s, 1 H, arom H), 7.16 (s, 1 H, arom H), 6.65 (br s, 2H, NH2) ppm.
- This compound was synthesized according to the general procedure using 4-fluorobenzoyl chloride
- A suspension of the appropriate 2-amino-4,6-dihydroxy-5-(acylamino)pyrimidine analogue (4.24 mmol) and P2S5 (8.47 mmol, 3.77 g) in pyridine (20 ml) was heated under reflux for 12 hours. After concentration under reduced pressure, the residue was resuspended in water (15 ml). Potassium carbonate (1.76 g, 13 mmol) was added and the mixture was stirred at room temperature for 1 h. The precipitate was collected by filtration and washed with water, yielding the crude title compound which was used without further purification.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2-amino-5-benzamido-4,6-dihydroxypyrimidine
- This compound was synthesized from 2-amino-5-(2-furancarboxamido)-4,6-dihydroxypyrimidine
- This compound was synthesized from 2-amino-5-(4-fluorobenzamido)-4,6-dihydroxypyrimidine
- To a solution of a 5-amino-2-substituted-thiazolo[5,4-d]pyrimidine-7-thiol analogue (3.37 mmol) in pyridine (20 ml) was added 1,1,1,3,3,3-hexamethyldisilazane (HMDS, 6.1 ml) and piperazine (33.7 mmol, 2.9 g). The reaction mixture was refluxed overnight. After cooling down to room temperature, the solvents were evaporated in vacuo. The residue was adsorbed on silica and purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a gradient gradually raising from 8% to 9% methanol in dichloromethane), yielding the title compounds as yellow powders, in yields ranging from 40-50%.
- The following compounds were made according to this procedure :
- This compound was synthesized from 5-amino-2-phenyl-thiazolo[5,4-d]pyrimidine-7-thiol.
- This compound was synthesized from 5-amino-2-(2-furyl)-thiazolo[5,4-d]pyrimidine-7-thiol.
- This compound was synthesized from 5-amino-2-(4-fluoro-phenyl)-thiazolo[5,4-d]pyrimidine-7-thiol.
- To a solution of a 5-amino-7-N-piperazinyl-2-substituted-thiazolo[5,4-d]pyrimidine analogue (0.65 mmol) in DMF (10 ml) was added diisopropylamine (1.3 mmol, 215 µl), O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU, 0.78 mmol, 251 mg) and an appropriate carboxylic acid (0.78 mmol). The reaction was stirred at room temperature for 2 hours. An extraction was carried out (water/dichloromethane) and the solvents were evaporated in vacuo. The residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 1% to 1.5% methanol in dichloromethane), yielding pure final compounds in yields varying from 70 to 80%.
- The following compounds were made according to this procedure :
- This compound was obtained from 5-amino-2-phenyl-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 4-methoxy-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.91 (m, 2H, arom H), 7.49 (m, 3H, arom H), 6.87 (q, 4H, arom H), 6.51 (br s, 2H, NH2), 4.81 (s, 2H, CH2), 4.36 (br s, 2H, piperazine H), 4.21 (br s, 2H, piperazine H), 3.68 (s, 3H, OCH3), 3.62 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(2-furyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 4-fluoro- phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.91 (s, 1H, arom H), 7.10-7.14 (m, 3H, arom H), 6.96-6.98 (m, 2H, arom H), 6.72 (q, 1 H, arom H), 6.52 (br s, 2H, NH2), 4.89 (s, 2H, CH2), 4.33 (br s, 2H, piperazine H), 4.19 (br s, 2H, piperazine H), 3.62 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(2-furyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 3-methyl-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.89 (s, 1H, arom H), 7.13-7.16 (m, 2H, arom H), 6.72-6.78 (m, 4H, arom H), 6.49 (br s, 2H, NH2), 4.85 (s, 2H, CH2), 4.32 (br s, 2H, piperazine H), 4.18 (br s, 2H, piperazine H), 3.63 (br s, 4H, piperazine H), 2.27 (s, 3H, CH3) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 3-methyl-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.96-8.00 (m, 2H, arom H), 7.36 (t, 2H, arom H), 7.16 (t, 1 H, arom H), 6.76-6.79 (m, 3H, arom H), 6.51 (br s, 2H, NH2), 4.86 (s, 2H, CH2), 4.35 (br s, 2H, piperazine H), 4.22 (br s, 2H, piperazine H), 3.65 (br s, 4H, piperazine H), 2.28 (s, 3H, CH3) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 2,4-dichloro-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.95-8.00 (m, 2H, arom H), 7.58 (d, 2H, arom H), 7.36 (m, 3H, arom H), 7.01 (d, 1H, arom H), 6.52 (br s, 2H, NH2), 5.09 (s, 2H, CH2), 4.36 (br s, 2H, piperazine H), 4.22 (br s, 2H, piperazine H), 3.64 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 4-chloro-o-tolyl-oxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.95-8.00 (m, 2H, arom H), 7.33-7.39 (t, 2H, arom H), 7.16-7.23 (m, 2H, arom H), 6.90 (d, 1 H, arom H), 6.51 (br s, 2H, NH2), 4.94 (s, 2H, CH2), 4.35 (br s, 2H, piperazine H), 4.22 (br s, 2H, piperazine H), 3.64 (br s, 4H, piperazine H), 2.20 (s, 3H, CH3) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 3-chloro-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.97 (m, 2H, arom H), 7.36 (m, 3H, arom H), 6.93-6.99 (m, 3H, arom H), 6.51 (br s, 2H, arom H), 4.96 (s, 2H, CH2), 4.36 (br s, 2H, piperazine H), 4.23 (br s, 2H, piperazine H), 3.64 (br s, 4H, piperazine H) ppm.
- To a solution of 5-amino-2-phenyl-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine (90 mg, 0.28 mmol) in dioxane (5 ml) was added triethylamine (0.86 mmol, 120 µl) and 4-chlorophenoxyacetyl chloride (0.35 mmol, 71 mg). The reaction was stirred at room temperature for 2 hours. The solvents were evaporated and the residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 1% to 2% methanol in dichloromethane), yielding the pure title compound.
- 1H NMR (300 MHz, DMSO): δ = 7.92 (m, 2H, arom H), 7.51 (m, 3H, arom H), 7.32-7.35 (m, 2H, arom H), 6.97-7.00 (m, 2H, arom H), 6.51 (br s, 2H, arom H), 4.93 (s, 2H, CH2), 4.38 (br s, 2H, piperazine H), 4.22 (br s, 2H, piperazine H), 3.64 (br s, 4H, piperazine H) ppm.
- To a solution of a 5-amino-7-N-piperazinyl-2-substituted-thiazolo[5,4-d]pyrimidine analogue (0.61 mmol) in DMF (10 ml) was added diisopropylamine (1.2 mmol, 200 µl) and an appropriate isocyanate (0.91 mmol). The reaction was stirred at room temperature for 2 hours. The solvents were evaporated in vacuo. The residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 1% to 1.5% methanol in dichloromethane), yielding pure final compounds in yields varying from 70 to 80%.
- The following compounds were made according to this procedure :
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 4-cyanophenylisocyanate.
- 1H NMR (300 MHz, DMSO): δ = 9.08 (s, 1H, NH), 7.95-7.99 (m, 2H, arom H), 7.70 (s, 4H, arom H), 7.37 (t, 2H, arom H), 6.51 (br s, 2H, NH2), 4.31 (br s, 4H, piperazine H), 3.66 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 2,4-difluoro-phenylisocyanate.
- 1H NMR (300 MHz, DMSO): δ = 8.40 (s, 1H, NH), 7.97 (m, 2H, arom H), 7.36 (m, 5H, arom H), 7.04 (m, 1 H, arom H), 6.50 (br s, 2H, NH2), 4.30 (br s, 4H, piperazine H), 3.62 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 4-bromo-phenylisocyanate.
- 1H NMR (300 MHz, DMSO): δ = 8.72 (s, 1H, NH), 7.95-7.98 (m, 2H, arom H), 7.43-7.48 (m, 6H, arom H), 6.49 (br s, 2H, NH2), 4.30 (br s, 4H, piperazine H), 3.63 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 5-amino-2-(4-fluoro-phenyl)-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 2-methoxy-phenylisocyanate.
- This compound was obtained from 5-amino-2-phenyl-7-N-piperazinyl-thiazolo[5,4-d]pyrimidine and 3-methyl-phenylisocyanate.
- 1H NMR (300 MHz, DMSO): δ = 8.52 (s, 1H, NH), 7.91 (dd, 2H, arom H), 7.50 (m, 3H, arom H), 7.32 (m, 2H, arom H), 7.12 (t, 1 H, arom H), 6.76 (d, 1 H, arom H), 6.52 (br s, 2H, NH2), 4.30 (br s, 4H, piperazine H), 3.62 (br s, 4H, piperazine H), 2.26 (s, 3H, CH3) ppm.
- A mixture of 5-amino-7-thiol-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine (1.31 g, 5 mmol), piperazine (2.15 g, 25 mmol) and 1,1,1,3,3,3-hexamethyldisilazane (HMDS, 5 ml) in pyridine (40 ml) was heated in a microwave oven (CEM discover, 150°C, 150 W) for 30 minutes. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/3), yielding the pure title compound as a yellowish solid (0.95 g, 60%).
- MS m/z (%): 314 ([M+H]+, 100)
- To a suspension of 5-amino-7-(piperazin-1-yl)-2-(pyridine-3-yl)thiazolo[5,4-d]pyrimidine (150 mg, 0.48 mmol) in dioxane (10 ml) was added 4-tolyl isocyanate (63 µl, 0.5 mmol). The resulting reaction mixture was stirred at room temperature for 30 minutes. The solvents were evaporated in vacuo and the residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/25), yielding the pure title compound as a yellowish solid (170 mg, 79 %).
- MS m/z (%): 447 ([M+H]+, 100)
- This compound was prepared from example 27 using 3-(4-bromophenyl)propionic acid in a yield of 71%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.42 (d, 2H, PhH), 7.10-7.24 (m, 4H, PhH), 6.96 (t, 2H, PhH), 4.69 (s, 2H, NH2), 4.17 (br s, 4H, N(CH2)2), 3.71 (br s, 2H, NCH2), 3.47 (br s, 2H, NCH2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2), 2.97 (t, 2H, CH2), 2.65 (t, 2H, CH2) ppm.
- This compound was prepared from example 27 using 4-hydroxyphenoxyacetic acid in a yield of 12%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.13-7.18 (m, 2H, PhH), 6.96 (t, 2H, PhH), 6.75-6.87 (m, 4H, PhH), 4.71 (s, 2H, NH2), 4.69 (s, 2H, OCH2), 4.25 (br s, 2H, NCH2), 4.21 (br s, 2H, NCH2), 3.70 (br s, 4H, N(CH2)2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm.
- This compound was prepared from example 27 using 4-methoxycarbonylphenoxyacetic acid in a yield of 41%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.01 (d, 2H, PhH), 7.13-7.18 (m, 2H, PhH), 7.01 (d, 2H, PhH), 6.96 (t, 2H, PhH), 4.81 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.25 (br s, 2H, NCH2), 4.20 (br s, 2H, NCH2), 3.88 (s, 3H, CH3), 3.73 (br s, 2H, NCH2), 3.65 (br s, 2H, NCH2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm
- This compound was prepared from example 27 using 4-trifluoromethoxyphenoxyacetic acid in a yield of 54%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.13-7.18 (m, 4H, PhH), 6.93-6.99 (m, 4H, PhH), 4.75 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.26 (br s, 2H, NCH2), 4.21 (br s, 2H, NCH2), 3.72 (br s, 2H, NCH2), 3.65 (br s, 2H, NCH2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm
- This compound was prepared from example 27 using 4-acetylphenoxyacetic acid in a yield of 67%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.97 (d, 2H PhH), 7.13-7.18 (m, 4H, PhH), 6.93-7.04 (m, 4H, PhH), 4.82 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.25 (br s, 2H, NCH2), 4.20 (br s, 2H, NCH2), 3.72 (br s, 2H, NCH2), 3.65 (br s, 2H, NCH2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2), 2.56 (s, 3H, CH3) ppm
- This compound was prepared from example 27 using 3-chlorophenoxyacetic acid in a yield of 51%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.00-7.23 (m, 3H, PhH), 6.93-6.99 (m, 4H, PhH), 6.87 (d, 1 H, PhH), 4.73 (s, 2H, OCH2), 4.72 (s, 2H, NH2), 4.26 (br s, 2H, NCH2), 4.22 (br s, 2H, NCH2), 3.71 (t, 2H, NCH2), 3.64 (t, 2H, NCH2), 3.23 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm
- This compound was prepared from example 27 using 4-cyanophenyl isocyanate in a yield of 64%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.06 (s, 1H, NH), 7.69 (s, 4H, PhH), 7.28-7.33 (m, 2H, PhH), 7.10 (t, 2H, PhH), 6.30 (s, 2H, NH2), 4.20 (br s, 4H, N(CH2)2), 3.59 (br s, 4H, N(CH2)2) 3.26 (t, 2H, CH2), 3.05 (t, 2H, CH2) ppm.
- This compound was prepared from example 26 using 4-methoxyphenoxyacetic acid in a yield of 59%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.23-7.28 (m, 2H, PhH), 7.02 (t, 2H, PhH), 8.82-6.93 (m, 4H, PhH), 4.78 (s, 2H, NH2), 4.70 (s, 2H, OCH2), 4.28 (br s, 4H, N(CH2)2), 4.21 (s, 2H, CH2), 3.77 (s, 3H, CH3), 3.73 (br s, 4H, N(CH2)2) ppm
- This compound was prepared from example 26 using 4-bromophenoxyacetic acid in a yield of 80%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.40 (d, 2H, PhH), 7.23-7.27 (m, 2H, PhH), 7.02 (t, 2H, PhH), 6.87 (d, 2H, PhH), 4.78 (s, 2H, NH2), 4.73 (s, 2H, OCH2), 4.28 (br s, 4H, N(CH2)2), 4.21 (s, 2H, CH2), 3.67-3.72 (m, 4H, N(CH2)2) ppm
- This compound was prepared from example 26 using 4-cyanophenyl isocyanate in a yield of 58%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.06 (s, 1H, NH), 7.69 (s, 4H, PhH), 7.37-7.42 (m, 2H, PhH), 7.18 (t, 2H, PhH), 6.33 (s, 2H, NH2), 4.29 (s, 2H, CH2), 4.22 (br s, 4H, N(CH2)2), 3.60 (br s, 4H, N(CH2)2) ppm.
- This compound was prepared from example 25 using 4-fluorophenoxyacetic acid in a yield of 58%, according to the procedure for the synthesis of example 50.
- 1H NMR (500 MHz, DMSO, 25 °C): δ = 7.95-7.98 (m, 2H PhH), 7.35 (t, 2H, PhH), 7.11 (t, 2H, PhH), 6.95-6.98 (m, 2H PhH), 6.51 (s, 2H, NH2), 4.88 (s, 2H, CH2), 4,35 (br s, 2H NCH2), 4.21 (br s, 2H, NCH2), 3.63 (br s, 4H N(CH2)2) ppm
- This compound was prepared from example 25 using 4-methoxyphenoxyacetic acid in a yield of 95%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.95-8.00 (m, 2H, PhH), 7.35 (t, 2H, PhH), 6.84-6.92 (m, 4H, PhH), 6.51 (s, 2H, NH2), 4.81 (s, 2H, OCH2), 4.33 (br s, 2H, NCH2), 4.21 (br s, 2H, NCH2), 3.69 (s, 3H, CH3), 3.64 (br s, 4H, N(CH2)2) ppm.
- This compound was prepared from example 25 using 4-bromophenoxyacetic acid in a yield of 47%, according to the procedure for the synthesis of example 50.
- 1H NMR (500 MHz, DMSO, 25 °C): δ = 7.95-7.98 (m, 2H PhH), 7.47 (d, 2H, PhH), 7.35 (t, 2H, PhH), 6.92 (d, 2H, PhH), 6.50 (s, 2H, NH2), 4.91 (s, 2H, CH2), 4,37 (br s, 2H NCH2), 4.21 (br s, 2H, NCH2), 3.63 (br s, 4H N(CH2)2) ppm.
- This compound was prepared from example 25 using 3-(4-fluoropheny)propionic acid in a yield of 62%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.85-7.89 (m, 2H, PhH), 7.11-7.22 (m, 4H, PhH), 6.98 (t, 2H, PhH), 4.84 (s, 2H, NH2), 4.29 (br s, 4H, N(CH2)2), 3.78 (t, 2H, NCH2), 3.55 (t, 2H, NCH2), 3.00 (t, 2H, CH2), 2.67 (t, 2H, CH2) ppm.
- This compound was prepared from example 25 using 2-(4-chlorophenoxy)-2-methylpropanoic acid in a yield of 58%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.94-7.98 (m, 2H, PhH), 7.32-7.38 (m, 4H, PhH), 6.87 (d, 2H, PhH), 6.46 (s, 2H, NH2), 4.14 (br s, 2H, NCH2), 3.93 (br s, 4H, N(CH2)2), 3.68 (br s, 2H, NCH2), 1.56 (s, 6H, CH3, CH3) ppm.
- A suspension of a carboxylic acid (6.7 mmol) in SOCl2 (5 ml) was heated under reflux for 1 h. After concentration under reduced pressure, the residue was redissolved in dioxane (5 ml) and added to a stirring solution of 2,5-diamino-4,6-dihydroxypyrimidine hydrochloride (1.0 g, 5.6 mmol) in 1 N NaOH (20 ml) at 0°C. The mixture was stirred and warmed to room temperature over 1 hour. After neutralization with 1 N hydrochloric acid to pH = 5, the precipitate was filtered off, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure:
- This compound was synthesized using 3-(3-methoxyphenyl)propionic acid, yielding the title compound in 87% yield.
- This compound was synthesized using 3-(3,4-dimethoxyphenyl)propionic acid, yielding the title compound in 46% yield.
- This compound was synthesized using 3-(4-tolyl)propionic acid, yielding the title compound in 67% yield.
- A suspension of a 2-amino-4,6-dihydroxy-5-N-acylamino-pyrimidine analogue (3.3 mmol) and P2S5 (1.68 g, 7.6 mmol) in pyridine (15 ml) was heated under reflux for 6 hours. After concentration under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 30:1), yielding the title compounds as a yellow solid.
- The following compounds were synthesized according to this procedure :
- Example 241: Synthesis of 5-amino-2-(3-methoxyphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol
This compound was synthesized from N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-3-(3-methoxyphenyl)propanamide, yielding the title compound in 82% yield. - Example 242: Synthesis of 5-amino-2-(3,4-dimethoxyphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol
This compound was synthesized from N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-3-(3,4-dimethoxyphenyl)propanamide, yielding the title compound in 48% yield.
MS m/z (%):349 ([M+H]+, 100) - Example 243: Synthesis of 5-amino-2-(4-methylphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol
This compound was synthesized from N-(2-amino-4,6-dihydroxypyrimidin-5-yl)-3-p-tolylpropanamide, yielding the title compound in 76% yield. - To a solution of 5-amino-2-substituted-thiazolo[5,4-d]pyrimidine-7-thiol (2.4 mmol) and triethylamine (0.83 ml, 5.97 mmol) in DMSO (10 ml) was added iodomethane (0.29 ml, 4.77 mmol). The reaction mixture was stirred for 12 h under N2 at 25°C. The mixture was poured into water and extracted with EtOAc. The organic extracts were dried over Na2SO4 and the solvents were removed under reduced pressure. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 80:1), yielding the title compound as a light yellow solid.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 5-amino-2-(3-methoxyphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol, yielding the title compound in 73% yield.
- This compound was synthesized from 5-amino-2-(3,4-dimethoxyphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol, yielding the title compound in 96% yield.
- This compound was synthesized from 5-amino-2-(4-methylphenethyl)thiazolo[5,4-d]pyrimidine-7-thiol, yielding the title compound in 55% yield.
- To a solution of a 2-substituted-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine analogue (0.90 mmol) in dichloromethane (5 ml) was added mCPBA (70 %, 0.39 g, 2.26 mmol) at 0°C. The reaction mixture was stirred for 3 hours, whereby the reaction temperature was gradually increased from 0°C to room temperature. The reaction mixture was diluted with CHCl3 and was washed with a saturated NaHCO3 solution, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1), affording the title compound.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2-(3-methoxyphenethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 85% yield.
- This compound was synthesized from 2-(3,4-dimethoxyphenethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 69% yield.
- This compound was synthesized from 2-(4-methylphenethyl)-7-(methylthio)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 51% yield.
- To a solution of a 2-substituted-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine analogue (0.55 mmol) and triethylamine (0.12 ml, 0.82 mmol) in dioxane (4 ml) was added piperazine (71 mg, 0.82 mmol). The reaction mixture was heated at 60 °C for 5 hours. After cooling, the volatiles were removed under reduced pressure. The crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 15:1), furnishing the title compound.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2-(3-methoxyphenethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 98% yield.
- This compound was synthesized from 2-(3,4-dimethoxyphenethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 89% yield.
- This compound was synthesized from 2-(4-methylphenethyl)-7-(methylsulfonyl)thiazolo[5,4-d]pyrimidin-5-amine, yielding the title compound in 79% yield.
- This compound was prepared from 2-(3-methoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 66%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.22 (t, 1H, PhH), 6.81-6.93 (m, 4H, PhH), 6.73-6.81 (m, 3H, PhH), 4.76 (s, 2H, NH2), 4.69 (s, 2H, OCH2), 4.25 (br s, 2H, NCH2), 4.19 (br s, 2H, NCH2), 3.77 (s, 3H, CH3), 3.76 (s, 3H, CH3), 3.69 (br s, 4H, N(CH2)2), 3.25 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(3-methoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-bromophenoxyacetic acid in a yield of 65%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.39 (d, 2H PhH), 7.20 (t, 1H, PhH), 6.86 (d, 2H, PhH), 6.73-6.81 (m, 3H, PhH), 4.77 (s, 2H, NH2), 4.72 (s, 2H, OCH2), 4.25 (br s, 2H, NCH2), 4.19 (br s, 2H, NCH2), 3.77 (s, 3H, CH3), 3.70 (br s, 2H, NCH2), 3.64 (br s, 2H, NCH2), 3.25 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(3-methoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 48%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26 (d, 2H, PhH), 7.20 (t, 1H, PhH), 6.92 (d, 2H PhH), 6.73-6.82 (m, 3H, PhH), 4.73 (s, 2H, OCH2), 4.71 (2, 2H, NH2), 4.26 (br s, 2H, NCH2), 4.20 (br s, 2H, NCH2), 3.78 (s, 3H, CH3), 3.70 (br s, 2H, NCH2), 3.67 (br s, 2H, NCH2), 3.26 (t, 2H, CH2), 3.07 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(3,4-dimethoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-bromophenoxyacetic acid in a yield of 74%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.39 (d, 2H, PhH), 6.87 (d, 2H, PhH), 6.72-6.78 (m, 3H, PhH), 4.73 (s, 2H, OCH2), 4.69 (s, 2H, NH2), 4.24 (br s, 4H, N(CH2)2), 3.85 (s, 6H, CH3, CH3), 3.71 (br s, 2H, NCH2), 3.65 (br s, 2H, NCH2), 3.24 (t, 2H, CH2), 3.04 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(3,4-dimethoxyphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 71%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 6.73-6.93 (m, 7H, PhH), 4.76 (s, 2H, NH2), 4.70 (s, 2H, OCH2), 4.26 (br s, 2H, NCH2), 4.22 (br s, 2H, NCH2), 3.85 (s, 3H, CH3), 3.84 (s, 3H, CH3), 3.76 (s, 3H, CH3), 3.71 (br s, 4H, N(CH2)2), 3.23 (t, 2H, CH2), 3.04 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(4-methylphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-bromophenoxyacetic acid in a yield of 47%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.39 (d, 2H, PhH), 7.10 (s, 4H, PhH), 6.87 (d, 2H, PhH), 4.73 (s, 2H, OCH2), 4.70 (s, 2H, NH2), 4.27 (br s, 2H, NCH2), 4.20 (br s, 2H, NCH2), 3.64-3.72 (m, 4H, N(CH2)2), 3.24 (t, 2H, CH2), 3.06 (t, 2H, CH2), 2.32 (s, 3H, CH3) ppm.
- This compound was prepared from 2-(4-methylphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 68%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.10 (s, 4H, PhH), 6.82-6.93 (m, 4H, PhH), 4.70 (s, 4H, NH2, OCH2), 4.27 (br s, 2H, NCH2), 4.21 (br s, 2H, NCH2), 3.76 (s, 3H, OCH3), 3.70 (br s, 4H, N(CH2)2), 3.23 (t, 2H, CH2), 3.05 (t, 2H, CH2), 2.31 (s, 3H, CH3) ppm
- This compound was prepared from 2-(4-methylphenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 51%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.26 (d, 2H, PhH), 7.10 (s, 4H, PhH), 6.92 (d, 2H, PhH), 4.73 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.27 (br s, 2H, NCH2), 4.20 (br s, 2H, NCH2), 3.66-3.72 (m, 4H, N(CH2)2), 3.23 (t, 2H, CH2), 3.05 (t, 2H, CH2), 2.31 (s, 3H, CH3) ppm.
- A suspension of 2,5-diamino-4,6-dihydroxypyrimidine hydrochloride (5.0 g, 28 mmol)and tetraethylammonium chloride (27.8 g, 0.167 mmol) in phosphorus oxychloride (80 ml) was heated at 105°C for 20 hours. After cooling down to room temperature, the excess phosphorus oxychloride was distilled off under vacuum. The reaction mixture was poured into ice water and the pH was adjusted to 4, and the mixture was stirred for 1 hour at 50°C. The pH was adjusted to 7 and the product was extracted with ethyl acetate, washed with a saturated NaHCO3 solution, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1), affording the title compound (3.21 g, 64 %).
- To a solution of a 4,6-dichloropyrimidine analogue (11.2 mmol) and DIPEA (2.9 ml, 16.8 mmol) in dioxane (40 ml) was added tert-butyl piperazine-1-carboxylate (3.12 g, 16.8 mmol). The reaction mixture was heated at 100°C for overnight., After cooling, the volatile was removed under reduced pressure. The crude residue was diluted with CHCl3 and was washed with a saturated NaHCO3 solution, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was purified by flash chromatography on silica (CH2Cl2/MeOH 50:1), affording the title compound.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2,5-diamino-4,6-dichloropyrimidine, yielding the title compound in 94% yield.
- This compound was synthesized from 5-amino-4,6-dichloropyrimidine, yielding the title compound in 91% yield.
- To a solution of a tert-butyl 4-(2,5-diamino-6-chloropyrimidin-4-yl)piperazine-1-carboxylate analogue (6.1 mmol) in DMSO (15 ml) was added sodium sulfide nonahydrate (2.9 g, 12.1 mmol). The reaction mixture was heated at 50°C overnight., After cooling down to room temperature, water (15 ml) was added and the solution was evaporated under reduced pressure. The crude residue was diluted with water (20 ml) and neutralized with HCl. The precipitate was collected by filtration, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure :
- Example 264 : Synthesis of tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate
- Example 265 : Synthesis of tert-butyl 4-(5-amino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate
- This compound was synthesized from tert-butyl 4-(5-amino-6-chloropyrimidin-4-yl)piperazine-1-carboxylate, yielding the title compound in 80% yield.
- To a solution of a tert-butyl 4-(6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate analogue (6.1 mmol) in DMSO (10 ml) was added an appropriate aldehyde (2.36 mmol). The reaction mixture was heated at 150°C for 1 hour. After cooling down to room temperature, the mixture was diluted with ethyl acetate and washed with water, brine and dried over Na2SO4. After removing the solvents under reduced pressure, the residue was diluted with dichloromethane (5 ml) and treated with TFA (1.6 ml, 21.4 mmol). The reaction mixture was stirred at room temperature overnight. The volatiles were evaporated under reduced pressure and the residue was diluted with water and neutralized with 1 N NaOH. The precipitate was collected by filtration, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure :
- Example 266 : Synthesis of 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized using nicotinaldehyde, yielding the title compound in 56% yield. - Example 267 : Synthesis of 7-(piperazin-1-yl)-2-(pyridin-2-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized using picolinaldehyde, yielding the title compound in 44% yield. - Example 268 : Synthesis of 7-(piperazin-1-yl)-2-(pyridin-4-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized using isonicotinaldehyde, yielding the title compound in 46% yield. - Example 269 : Synthesis of 2-(4-chlorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized using 4-chlorobenzaldehyde, yielding the title compound in 60% yield. - This compound was prepared from 2-(4-chlorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 63%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.82 (d, 2H, PhH), 7.43 (d, 2H, PhH), 6.83-6.94 (m, 4H, PhH), 4.80 (s, 2H, NH2), 4.72 (s, 2H, CH2), 4.37 (br s, 2H, NCH2), 4.32 (br s, 2H, NCH2), 3.77 (s, 7H, CH3, N(CH2)2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 52%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.14 (s, 1H, 2-pyridyl-H), 8.66 (d, J = 4.7 Hz, 1H, 4-pyridyl-H), 8.30 (d, J = 5.5 Hz, 1 H, 6-pyridyl-H), 7.53-7.57 (m, 1 H, 5-pyridyl-H), 6.84-6.93 (m, 4H, PhH), 6.58 (s, 2H, NH2), 4.81 (s, 2H, OCH2), 4.34 (br s, 2H, CH2), 4.23 (br s, 2H, CH2), 3.70 (s, 3H, CH3), 3.66 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-fluorophenoxyacetic acid in a yield of 67%, according to the procedure for the synthesis of example 50
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.14 (d, J = 2.0 Hz, 1 H, 2-pyridyl-H), 8.66 (d, J = 3.6 Hz, 1H, 4-pyridyl-H), 8.29 (d, J = 8.0 Hz, 1H, 6-pyridyl-H), 7.53-7.57 (m, 1H, 5-pyridyl-H), 7.12 (t, J = 8.9 Hz, 2H, PhH), 6.95-6.99 (m, 2H, PhH), 6.58 (s, 2H, NH2), 4.89 (s, 2H, OCH2), 4.37 (br s, 2H, CH2), 4.24 (br s, 2H, CH2), 3.66 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-trifluoromethoxyphenoxyacetic acid in a yield of 45%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.14 (d, J = 1.7 Hz, 1 H, 2-pyridyl-H), 8.66 (dd, J = 4.7 Hz, J = 1.5 Hz, 1 H, 4-pyridyl-H), 8.28 (dd, J = 8.2 Hz, j = 2.0 Hz, 1 H, 6-pyridyl-H), 7.53-7.57 (m, 1H, 5-pyridyl-H), 7.30 (d, J = 8.6 Hz, 2H, PhH), 7.05 (d, J = 8.6 Hz, 2H, PhH), 6.58 (s, 2H, NH2), 4.96 (s, 2H, OCH2), 4.38 (br s, 2H, CH2), 4.25 (br s, 2H, CH2), 3.65 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 2-(4-chlorophenoxy)-2-methylpropanoic acid in a yield of 45%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.12 (d, J = 2.3 Hz, 1 H, 2-pyridyl-H), 8.66 (d, J = 4.7 Hz, 1 H, 4-pyridyl-H), 8.27 (d, J = 8.0 Hz, 1 H, 6-pyridyl-H), 7.52-7.56 (m, 1 H, 5-pyridyl-H), 7.34 (d, J = 8.8 Hz, 2H, PhH), 6.86 (d, J = 8.8 Hz, 2H, PhH), 6.53 (s, 2H, NH2), 3.94 (br s, 4H, CH2), 3.69 (br s, 4H, CH2), 1.59 (s, 6H, CH3) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 3-tolyloxyacetic acid in a yield of 45%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.13 (s, 1H, pyridyl-H), 8.66 (d, 1H, pyridyl-H), 8.28 (d, 1H, pyridyl-H), 7.52-7.56 (m, 1H, pyridyl-H), 7.16 (t, 1H, PhH), 6.73-6.78 (m, 3H, PhH), 6.56 (s, 2H, NH2), 4.85 (s, 2H, CH2), 4.36 (br s, 2H, NCH2), 4.23 (br s, 2H, NCH2), 3.65 (br s, 4H, N(CH2)2), 2.27 (s, 3H, CH3) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 3-tolyl isocyanate in a yield of 42%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.13 (d, J = 1.7 Hz, 1H, 2-pyridyl-H), 8.66-8.68 (m, 1H, 4-pyridyl-H), 8.52 (s, 1H, NH), 8.28 (d, J = 5.9 Hz, 1H, 6-pyridyl-H), 7.53-7.57 (m, 1H, 5-pyridyl-H), 7.32 (s, 1 H, 2-tolyl-H), 7.29 (d, J = 8.5 Hz, 1 H, 6-tolyl-H), 7.12 (t, J = 7.6 Hz, 1 H,
- 5-tolyl-H), 6.77 (d, J = 7.0 Hz, 1 H, 4-tolyl-H), 6.57 (s, 2H, NH2), 4.30 (br s, 4H, CH2), 3.62 ( br s, 4H, CH2), 2.26 (s, 3H, CH3) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenyl isocyanate in a yield of 38%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = = 9.13 (d, J = 2.3 Hz, 1H, 2-pyridyl-H), 8.73 (s, 1H, NH), 8.66 (d, J = 4.7 Hz, 1 H, 4-pyridyl-H), 8.28 (d, J = 8.0 Hz, 1 H, 6-pyridyl-H), 7.54-7.57 (m, 1H, 5-pyridyl-H), 7.53 (d, J = 8.9 Hz, 2H, PhH), 7.30 (d, J = 8.9 Hz, 2H, PhH), 6.60 (s, 2H, NH2), 4.32 (br s, 4H, CH2), 3.64 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxybenzyl isocyanate in a yield of 40%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.11 (s, 1H, 2-pyridyl-H), 8.66 (d, J = 4.6 Hz, 1H, 4-pyridyl-H), 8.27 (d, J = 8.1 Hz, 1 H, 6-pyridyl-H), 7.53-7.57 (m, 1 H, 5-pyridyl-H), 7.21 (d, J = 8.8 Hz, 2H, PhH), 7.08 (s, 1 H, NH), 6.87 (d, J = 8.8 Hz, 2H, PhH), 6.54 (s, 2H, NH2), 4.20 (br s, 6H, CH2, NHCH 2 ), 3.72 (s, 3H, CH3), 3.50 (br s, 4H, CH2) ppm
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-cyanophenyl isocyanate in a yield of 53%, according to the procedure for the synthesis of example 42.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.14 (s, 1 H, NH), 9.09 (s, 1 H, 2-pyridyl-H), 8.67 (d, J = 4.9 Hz, 1 H, 4-pyridyl-H), 8.29 (d, J = 6.3 Hz, 1 H, 6-pyridyl-H), 7.69 (s, 4H, PhH), 7.54-7.57 (m, 1 H, 5-pyridyl-H), 6.57 (s, 2H, NH2), 4.32 (br s, 4H, CH2), 3.67 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine using phenylmethanesulfonyl chloride in a yield of 40%, according to the procedure for the synthesis of example 41.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 9.14 (s, 1H, 2-pyridyl-H), 8.67 (d, J = 4.4 Hz, 1H, 4-pyridyl-H), 8.29 (d, J = 7.3 Hz, 1H, 6-pyridyl-H), 7.53-7.57 (m, 1H, 5-pyridyl-H), 7.36-7.41 (m, 5H, PhH), 6.59 (s, 2H, NH2), 4.45 (s, 2H, SCH2), 4.31 (br s, 4H, CH2), 3.35 (br s, 4H, CH2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-4-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 43%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.71 (d, 2H, pyridyl-H), 7.87 (d, 2H, pyridyl-H), 7.34 (d, 2H, PhH), 6.99 (d, 2H, PhH), 6.66 (s, 2H, NH2), 4.93 (s, 2H, CH2),4.37 (br s, 2H, NCH2), 4.25 (br s, 2H, NCH2), 3.65 (br s, 4H, N(CH2)2) ppm.
- This compound was prepared from 7-(piperazin-1-yl)-2-(pyridin-2-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyactic acid in a yield of 57%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.63 (d, 1 H, pyridyl-H), 8.16 (d, 1 H, pyridyl-H), 7.96 (t, 1H, pyridyl-H), 7.48 (t, 1H, pyridyl-H), 7.33 (d, 2H, PhH), 6.99 (d, 2H, PhH), 6.54 (s, 2H, NH2), 4.93 (s, 2H, CH2),4.36 (br s, 2H, NCH2), 4.25 (br s, 2H, NCH2), 3.65 (br s, 4H, N(CH2)2) ppm.
- A suspension of an appropriate carboxylic acid (0.42 mmol) in SOCl2 (1 ml) was heated under reflux for 1 hour. After concentration under reduced pressure, the residue was redissolved in dioxane (1 ml) and added to a stirring solution of tert-butyl 4-(5-amino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate (0.1 g, 0.32 mmol) and DIPEA (0.22 ml, 1.28 mmol) in DMF (3 ml). The reaction mixture was stirred for 2 hours. The mixture was extracted with ethyl acetate, washed with water, brine and dried over sodium sulfate. After removing solvent under reduced pressure, the crude mixture was diluted with dioxane (5 ml) and 3M HCl in dioxane (1 ml) was added and the mixture was heated at 60°C for 5 hours. After cooling, the mixture was concentrated under reduced pressure and the residue was diluted with water and neutralized with 1 N NaOH. The precipitate was collected by filtration, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure :
- Example 283 : Synthesis of 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine
This compound was synthesized from tert-butyl 4-(5-amino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using 3-(4-fluorophenyl)propionic acid, yielding the title compound in 80% yield. - Example 284 : Synthesis of 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine
This compound was synthesized from tert-butyl 4-(5-amino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using 4-fluorobenzoic acid, yielding the title compound in 74% yield. - Example 285 : Synthesis of 2-(3-(4-fluorophenyl)propyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized from tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using 4-(4-fluorophenyl)butanoic acid, yielding the title compound in 48% yield. - Example 286 : Synthesis of 2-(4-(4-fluorophenyl)butyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized from tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using 4-(4-fluorophenyl)butanoic acid, yielding the title compound in 58% yield. - Example 287 : Synthesis of 2-(4-bromophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine
This compound was synthesized from tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using 3-(4-bromophenyl)propionic acid, yielding the title compound in 70% yield. - To a solution of tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate (0.1 g, 0.31 mmol) and pyridine (37 µl, 0.46 mmol) in DMF (3 ml) was added an appropriate acid chloride (0.34 mmol). The reaction mixture was stirred for 2 hours. The mixture was extracted with ethyl acetate, washed with water, brine and dried over sodium sulfate. After removing solvent under reduced pressure, the crude mixture was diluted with dioxane (5 ml) and 3M HCl in dioxane (1 ml) was added and the mixture was heated at 60°C for 5 hours. After cooling, the mixture was concentrated under reduced pressure and the residue was diluted with water and neutralized with 1 N NaOH. The precipitate was collected by filtration, washed with water and dried over P2O5, yielding the title compound.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using hexanoyl chloride, yielding the title compound in 67% yield.
- This compound was synthesized from tert-butyl 4-(2,5-diamino-6-mercaptopyrimidin-4-yl)piperazine-1-carboxylate using p-toluoyl chloride, yielding the title compound in 80% yield.
- This compound was prepared from 2-(3-(4-fluorophenyl)propyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 33%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.24 (d, 2H, PhH), 7.10-7.15 (m, 2H, PhH), 6.96 (t, 2H, PhH), 6.90 (d, 2H, PhH), 4.71 (s, 4H, NH2, OCH2), 4.27 (br s, 2H, NCH2), 4.23 (br s, 2H, NCH2), 3.65-3.73 (m, 4H, N(CH2)2), 2.92 (t, 2H, CH2), 2.68 (t, 2H, CH2), 2.07 (quint, 2H, CH2) ppm.
- This compound was prepared from 2-(3-(4-fluorophenyl)propyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 30%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.7.11-7.16 (m, 2H, PhH), 6.94 (t, 2H, PhH), 6.82-6.93 (m, 4H, PhH), 4.71 (s, 2H, NH2), 4.70 (s, 2H, OCH2), 4.29 (br s, 2H, NCH2), 4.24 (br s, 2H, NCH2), 3.76 (s, 3H, CH3), 3.71 (br s, 4H, N(CH2)2), 2.94 (t, 2H, CH2), 2.70 (t, 2H, CH2), 2.08 (quint, 2H, CH2) ppm
- This compound was prepared from 2-(4-(4-fluorophenyl)butyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 23%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.28 (d, 2H, PhH), 7.12-7.16 (m, 2H PhH), 7.00 (t, 2H, PhH), 6.94 (d, 2H PhH), 4.75 (s, 4H, NH2, OCH2), 4.30 (br s, 2H, NCH2), 4.26 (br s, 2H, NCH2), 3.75 (t, 2H, NCH2), 3.69 (t, 2H, NCH2), 2.98 (t, 2H, CH2), 2.66 (t, 2H, CH2), 1.78-1.87 (m, 2H, CH2), 1.70-1.74 (m, 2H CH2) ppm
- This compound was prepared from 2-(4-(4-fluorophenyl)butyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 35%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.08-7.13 (m, 2H, PhH), 6.95 (t, 2H, PhH), 6.82-6.93 (m, 4H, PhH), 4.69 (s, 4H, NH2, OCH2), 4.27 (br s, 2H, NCH2), 4.23 (br s, 2H, NCH2), 3.76 (s, 3H, CH3), 3.71 (br s, 4H, N(CH2)2), 2.95 (t, 2H, CH2), 2.63 (t, 2H, CH2), 1.75-1.84 (m, 2H, CH2), 1.60-1.72 (m, 2H CH2) ppm
- This compound was prepared from 7-(piperazin-1-yl)-2-p-tolylthiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 41%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.78 (d, 2H, PhH), 7.25 (d, 2H, PhH), 6.83-6.94 (m, 4H, PhH), 4.76 (s, 2H, NH2), 4.72 (s, 2H, CH2), 3.77 (br s, 7H, OCH3, N(CH2)2), 42.41 (s, 3H, CH3) ppm
- This compound was prepared from 7-(piperazin-1-yl)-2-p-tolylthiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 32%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.78 (d, 2H, PhH), 7.24-7.27 (m, 4H, PhH), 6.93 (d, 2H, PhH), 4.77 (s, 2H, NH2), 4.75 (s, 2H, CH2), 4.38 (br s, 2H, NCH2), 4.32 (br s, 2H, NCH2), 3.71-3.79 (m, 4H, N(CH2)2), 2.41 (s, 3H, CH3) ppm
- This compound was prepared from 2-pentyl-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 34%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 6.89-6.90 (m, 4H, PhH), 6.30 (s, 2H, NH2), 4.79 (s, 2H, CH2), 4.26 (br s, 2H, NCH2), 4.13 (br s, 2H, NCH2), 3.69 (s, 3H, OCH3), 3.58 (br s, 4H, N(CH2)2), 2.91 (t, 2H, CH2), 1.71 (qiuint, 2H, CH2), 1.31-1.36 (m, 4H, CH2, CH2), 0.87 (t, 3H, CH3) ppm.
- This compound was prepared from 2-pentyl-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 30%, according to the procedure for the synthesis of example 50.
- 1H NMR (500 MHz, CDCl3, 25 °C): δ = 7.25 (d, 2H, PhH), 6.91 (d, 2H, PhH), 4.73 (s, 2H, OCH2), 4.69 (s, 2H, NH2), 4.29 (br s, 2H, NCH2), 4.24 (br s, 2H, NCH2), 3.73 (br s, 2H, NCH2), 3.68 (br s, 2H, NCH2), 2.92 (t, 2H, CH2), 1.78 (quint, 2H, CH2), 1.37 (m, 4H, CH2, CH2), 0.91 (t, 3H, CH3) ppm
- This compound was prepared from 2-(4-bromophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-methoxyphenoxyacetic acid in a yield of 40%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.40 (d, 2H, PhH), 7.07 (d, 2H, PhH), 6.82-6.93 (m, 4H, PhH), 4.71 (s, 2H, NH2), 4.70 (s, 2H, OCH2), 4.23 (br s, 2H, NCH2), 4.19 (br s, 2H, NCH2), 3.77 (s, 3H, CH3), 3.67-3.75 (m, 4H, N(CH2)2), 3.23 (t, 2H, CH2), 3.05 (t, 2H, CH2) ppm
- This compound was prepared from 2-(4-bromophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidin-5-amine using 4-chlorophenoxyacetic acid in a yield of 29%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.40 (d, 2H, PhH), 7.26 (d, 2H, PhH), 7.07 (d, 2H, PhH), 6.92 (d, 2H, PhH), 4.73 (s, 2H, OCH2), 4.71 (s, 2H, NH2), 4.20 (br s, 4H, N(CH2)2), 3.71 (t, 2H, NCH2), 3.65(t, 2H, NCH2), 3.23 (t, 2H, CH2), 3.05 (t, 2H, CH2) ppm
- This compound was prepared from 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine using 4-methoxyphenoxyacetic acid in a yield of 51%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.44 (s, 1H, CH), 7.95-8.00 (m, 2H, PhH), 7.19 (t, 2H, PhH), 6.83-6.95 (m, 4H, PhH), 4.73 (s, 2H, CH2), 4.42 (br s, 4H, N(CH2)2), 3.81 (br s, 4H, N(CH2)2), 3.77 (s, 3H, CH3) ppm.
- This compound was prepared from 2-(4-fluorophenyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine using 4-chlorophenoxyacetic acid in a yield of 48%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.45 (s, 1 H, CH), 7.95-8.00 (m, 2H, PhH), 7.27 (d, 2H, PhH), 7.19 (d, 2H, PhH), 6.93 (d, 2H, PhH), 4.76 (s, 2H, CH2), 4.43 (br s, 4H, N(CH2)2), 3.81 (br s, 4H, N(CH2)2) ppm.
- This compound was prepared from 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine using 4-methoxyphenoxyacetic acid in a yield of 39%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.40 (s, 1H, CH), 7.14-7.19 (m, 2H, PhH), 6.97 (t, 2H, PhH), 6.82-6.91 (m, 4H, PhH), 4.71(s, 2H, NH2), 4.31 (br s, 4H, N(CH2)2), 3.76 (s, 3H, CH3), 3.74(br s, 4H, N(CH2)2), 3.34 (t, 2H, CH2), 3.12 (t, 2H, CH2) ppm.
- This compound was prepared from 2-(4-fluorophenethyl)-7-(piperazin-1-yl)thiazolo[5,4-d]pyrimidine using 4-chlorophenoxyacetic acid in a yield of 48%, according to the procedure for the synthesis of example 50.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.41 (s, 1 H, CH), 7.26 (d, 2H, PhH), 7.14-7.19 (m, 2H, PhH), 6.97 (t, 2H, PhH), 6.91 (d, 2H, PhH), 4.74 (s, 2H, NH2), 4.31 (br s, 4H, N(CH2)2), 3.75 (br s, 2H, NCH2), 3.69 (br s, 2H, NCH2), 3.35 (t, 2H, CH2), 3.12 (t, 2H, CH2) ppm.
- A solution of methyl 2-aminothiophene-3-carboxylate (3.0 g, 19.1 mmol) in formamide (95 ml) was heated at 190°C for 4 hours. The cooled mixture was poured into water. The precipitate was filtered off, washed with water and dried. The crude product was purified by silica gel chromatography (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (1.93 g, 66%).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.49 (s, 1 H, NH), 8.13 (s, 1 H, CH), 7.58 (d, J = 5.8 Hz, 1 H, CH), 7.39 (d, J = 5.8 Hz, 1 H, CH) ppm.
- HRMS: calcd for C6H5N2OS 153.01226, found 153.01155.
- To a solution of ethyl 2-aminothiophene-3-carboxylate (0.1 g, 0.64 mmol) and acetonitrile (50 µl, 0.95 mmol) in dioxane (3 ml) was added 4M HCl in dioxane (3 ml). The mixture was stirred at room temperature overnight. The solvents were removed under reduced pressure. The residue was diluted with water and made alcaline with a saturated aqueous sodium bicarbonate solution. The precipitate was filtered off, washed with water and dried. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (42 mg, 40 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 12.92 (s, 1 H, NH), 7.46 (d, J = 5.8 Hz, 1 H, CH), 7.21 (d, J = 5.8 Hz, 1 H, CH), 2.61 (s, 3H, CH3) ppm.
- A mixture of ethyl 2-aminothiophene-3-carboxylate (0.5 g, 3.18 mmol), chloroformamidine hydrochloride (0.91 g, 7.95 mmol) and dimethylsulfone (1.50 g, 15.9 mmol) was heated at 120-130°C for 30 minutes. After cooling down to room temperature, water (10 ml) was added and ammonium hydroxide was used to neutralize the suspension. The solid was filtered off, washed with water and dried. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 10:1) to yield the title compound as a white solid (0.24 g, 45 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.89 (s, 1H, NH), 7.09 (d, J = 5.8 Hz, 1H, CH), 6.97 (d, J = 5.8 Hz, 1 H, CH), 6.52 (s, 2H, NH2) ppm.
- HRMS: calcd for C6H6N3OS 168.02316, found 168.02239.
- DMF (1.53 ml, 19.7 mmol) in dichloromethane (50 ml) was cooled to 0°C and oxalyl chloride (2.5 ml, 29.6 mmol) was added slowly forming a white gel. Thieno[2,3-d]pyrimidin-4(3H)-one (1.5 g, 9.86 mmol) was added and the reaction mixture was refluxed for 3 hours. The mixture was cooled down to room temperature and poured into water. The mixture was extracted with dichloromethane, dried over sodium sulfate and concentrated in vacuo. The crude residue was purified by silica gel chromatography (EtOAc/hexane 15:1) to yield the title compound as a white solid (1.61 g, 96%).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.88 (s, 1H, CH), 7.64 (d, J = 6.0 Hz, 1H, CH), 7.47 (d, J = 5.8 Hz, 1 H, CH) ppm.
- HRMS: calcd for C6H4ClN2S 170.97837, found 170.97804.
- n-BuLi (1.6 M in hexane, 1.9 ml, 2.5 mmol) in THF (8 ml) was cooled to -78°C. 4-Chlorothieno[2,3-d]pyrimidine (0.34 g, 2.0 mmol) was dissolved in THF (2 ml) and slowly added to the reaction mixture over 5 minutes. After 20 min, CBr4 (0.73 g, 2.2 mmol) in THF (3 ml) was slowly added to the reaction mixture. The temperature was maintained at -78°C for 20 minutes and then warmed to room temperature for 2 hours. The mixture was poured into water and extracted with chloroform, dried over sodium sulfate, and concentrated in vacuo. The crude residue was purified by silica gel chromatography (EtOAc/hexane 40:1) to yield two pure compounds a white solid (example 203: 0.13 g, 25% and example 204: 0.16 g, 26 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.82 (s, 1 H, H-2), 7.49 (s, 1 H, H-5) ppm.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.4 (s, 1H, H-5), 2.99 (t, J= 7.5 Hz, 2H, CH 2CH2CH2CH3), 1.83 (quint, J= 7.6 Hz, 2H, CH2CH 2CH2CH3), 1.42 (sixtet, J= 7.4 Hz, 2H, CH2CH2CH 2CH3), 0.96 (t, J= 7.4 Hz, 2H, CH2CH2CH2CH 3) ppm.
- A solution of 6-bromo-4-chlorothieno[2,3-d]pyrimidine (0.12 g, 0.48 mmol), 4-fluorophenylboronic acid (67 mg, 0.48 mmol), K2CO3 (0.266 g, 1.92 mmol) and Pd(PPh3)4 in dioxane/H2O (3:1, 3 ml) was refluxed under N2 for 2 hours. After cooling to room temperature, 1 N HCl was added slowly to neutralize the mixture to pH=7-8. The mixture was extracted with CH2Cl2, washed with water and brine and dried over Na2SO4. After removing the solvents under reduced pressure, the crude residue was purified by silica gel chromatography (hexane/EtOAc 30:1) to yield the title compound as a pale yellow solid (60 mg, 47 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.82 (s, 1H, CH-2), 7.70-7.74 (m, 2H, PhH), 7.52 (s, 1 H, CH-5), 7.19 (t, J= 8.4 Hz, 2H, PhH) ppm.
- HRMS: calcd for C12H7ClFN2S 265.00025, found 264.99949.
- This compound was prepared from example 309 in a yield of 57%, according to the procedure for the synthesis of example 310.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.57-7.62 (m, 2H, PhH), 7.36 (s, 1H, H-5), 7.08 (t, J= 8.4 Hz, 2H, PhH), 2.94 (t, J= 7.6 Hz, 2H, CH 2CH2CH2CH3), 1.78 (quint, J= 7.6 Hz, 2H, CH2CH 2CH2CH3) , 1.36 (sixtet, J= 7.5 Hz, 2H, CH2CH2CH 2CH3) , 0.89 (t, J= 7.4 Hz, 2H, CH2CH2CH2CH 3) ppm.
- HRMS: calcd for C16H15ClFN2S 321.06285, found 321.06206.
- To a solution of 4-chloro-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine (20 mg, 0.08 mmol) and triethylamine (42 µl, 0.3 mmol) in dioxane (1 ml) was added 2-(4-chlorophenoxy)-1-(piperazin-1-yl)ethanone (25 mg, 0.1 mmol). The mixture was heated at 60°C for 2 hours. After cooling down to room temperature, the solvents were removed under reduced pressure. The crude residue was purified by silica gel chromatography (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (27 mg, 75 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.51 (s, 1H, CH-2), 7.60-7.65 (m, 2H, PhH), 7.36 (s, 1H, CH-5), 7.26 (d, J= 8.8 Hz, PhH), 7.14 (t, J= 8.5 Hz, 2H, PhH), 6.91 (d, J= 8.8 Hz, 2H, PhH), 4.75 (s, 2H, CH2), 3.95 (br s, 4H, NCH2), 3.82 (br s, 4H, NCH2) ppm.
- HRMS: calcd for C24H21ClFN4O2S 483.10578, found 483.10438.
- This compound was prepared from example 311 in a yield of 57%, according to the procedure for the synthesis of example 312.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.58-7.63 (m, 2H, PhH), 7.32 (s, 1H, CH-5), 7.26 (d, J= 9.0 Hz, PhH), 7.13 (t, J= 8.6 Hz, 2H, PhH), 6.91 (d, J= 9.0 Hz, 2H, PhH), 4.74 (s, 2H, CH2), 3.95 (br s, 4H, N(CH2)2), 3.81 (br s, 4H, CON(CH2)2), 2.84 (t, J= 7.6 Hz, 2H, CH 2CH2CH2CH3), 1.80 (quint, J= 7.6 Hz, 2H, CH2CH 2CH2CH3) , 1.42 (sixtet, J= 7.6 Hz, 2H, CH2CH2CH 2CH3) , 0.96 (t, J= 7.6 Hz, 2H, CH2CH2CH2CH 3) ppm.
- HRMS: calcd for C28H29ClFN4O2S 539.16838, found 539.16680.
- To a solution of 4-chloro-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine (40 mg, 0.15 mmol) in 1,2-dichloroethane/t-BuOH (1:1, 1 ml) was added 3-chloro-4-fluoroaniline (22 mg, 0.15 mmol). The mixture was heated at 90°C for 2 days. After cooling down to room temperature, the solvents were removed under reduced pressure. The crude residue was purified by silica gel chromatography (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (28 mg, 50 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.61 (s, 1 H, H-2), 7.89 (dd, J= 6.5 Hz, J= 2.6 Hz, 1 H, PhH), 7.62-7.67 (m, 2H, PhH), 7.48-7.53 (m, 1 H, PhH), 7.28 (s, 1 H, H-5), 7.13-7.26 (m, 3H, PhH), 6.87 (s, 1 H, NH) ppm.
- HRMS: calcd for C18H11ClF2N3S 374.03303, found 374.03216.
- This compound was prepared from example 311 in a yield of 47%, according to the procedure for the synthesis of example 314.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.03 (dd, J= 6.30 Hz, J= 2.3 Hz, 1 H, PhH), 7.59-7.64 (m, 2H, PhH), 7.48-7.53 (m, 1H, PhH), 7.23 (s, 1H, H-5), 7.10-7.19 (m, 3H, PhH, PhH), 6.89 (s, 1H, NH), 2.93 (t, J= 7.5 Hz, 2H, CH 2CH2CH2CH3), 1.87 (quint, J= 7.6 Hz, 2H, CH2CH 2CH2CH3), 1.45 (sixtet, J= 7.5 Hz, 2H, CH2CH2CH 2CH3) , 0.98 (t, J= 7.3 Hz, 2H, CH2CH2CH2CH 3) ppm.
- To a stirred suspension of pyridinium chlorochromate (6.9 g, 21.4 mmol) in CH2Cl2 (100 ml) was added a solution of 2-(4-fluorophenyl)ethanol (3.0 g, 21.4 mmol) in CH2Cl2 (10 ml). The resulting suspension was stirred for 2 hours at room temperature and was then diluted with ether. The resulting suspension was filtered through a pad of Celite and washed with ether. The solvents were removed under reduced pressure to yield the crude title compound as a green oil (2.6 g, 86 %), which was used as such for further reaction.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 9.75 (s, 1H, CH), 7.19-7.22 (m, 2H, PhH), 7.06 (t, J= 8.5 Hz, PhH), 3.68 (s, 2H, CH2) ppm.
- Triethylamine (0.98 ml, 7.01 mmol) was added to a stirred suspension of ethyl cyanoacetate (2.79 ml, 13.7 mmol) and sulfur (0.44 g, 13.7 mmol) in DMF (70 ml). A solution of 2-(4-fluorophenyl)acetaldehyde (example 316, 1.9 g, 13.7 mmol) in DMF (5 ml) was added dropwise over a period of 50 minutes, while the temperature was maintained at 50°C. The solution was cooled down to room temperature and stirred overnight. The reaction was poured into water and the aqueous phase was extracted with diethylether. The organic layer was separated and washed with water, brine and dried over Na2SO4. The solvents were evaporated and the crude residue was purified by flash chromatography on silica gel (EtOAc/Hexane 1:15) to yield the title compound as a white solid (1.3 g, 38 %).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.33-7.39 (m, 2H, PhH), 7.14 (s, 1H, CH), 6.99 (t, J= 8.6 Hz, PhH), 6.06 (s, 2H, NH2), 4.29 (q, J= 7.1, 2H, CH2), 1.36 (t, J= 7.1, 3H, CH3) ppm. HRMS: calcd for C13H13FNO2S 266.06510, found 266.06425.
- This compound was synthesized using the procedure as described for example 317, using phenylacetaldehyde.
- To a solution of ethyl 2-amino-5-(4-fluorophenyl)thiophene-3-carboxylate (0.3 g, 1.13 mmol) and acetonitrile (0.56 ml, 11.3 mmol) in dioxane (4 ml) was added 4M HCl in dioxane (4 ml). The mixture was stirred at room temperature overnight. The solvents were removed under reduced pressure. The residue was diluted with water and made alcaline with a saturated aqueous sodium bicarbonate solution. The precipitate was filtered off, washed with water and dried. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 60:1) to yield the title compound as a white solid (0.29 g, 81 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.47 (s, 1H, NH), 7.76-7.81 (m, 2H, PhH), 7.71 (s, 1 H, CH), 7.28 (t, J= 8.7 Hz, PhH), 2.38 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H10FN2OS 261.04979, found 261.04889.
- A mixture of ethyl 2-amino-5-(4-fluorophenyl)thiophene-3-carboxylate (0.3 g, 1.13 mmol), chloroformamidine hydrochloride (0.33 g, 2.83 mmol) and dimethylsulfone (0.53 g, 5.65 mmol) was heated at 120-130°C for 30 minutes. After cooling down to room temperature, water (10 ml) was added and ammonium hydroxide was used to neutralize the suspension. The solid was filtered off, washed with water and dried. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 10:1) to yield the title compound as a white solid (0.28 g, 95 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 11.05 (s, 1H, NH), 7.66-7.71 (m, 2H, PhH), 7.51 (s, 1 H, CH), 7.23 (t, J= 8.8 Hz, PhH), 6.73 (s, 2H, NH2) ppm.
- HRMS: calcd for C12H9FN3OS 262.04504, found 262.04413.
- This compound was synthesized from example 318 according to the procedure mentioned for the synthesis of example 320.
- MS m/z (%) : 244 ([M+H]+, 100)
- To a solution of ethyl 2-amino-5-(4-fluorophenyl)thiophene-3-carboxylate (0.2 g, 0.75 mmol) and benzonitrile (0.23 g, 2.26 mmol) in dioxane (4 ml) was added 4M HCl in dioxane (4 ml). The mixture was stirred at room temperature overnight. The precipitate was filtered off, washed with diethyl ether and dried. The solid was redissolved in DMF and the mixture was heated at 100°C for 3 hours. The solvents were removed under reduced pressure. The residue was diluted with water and the solid was filtered off, washed with water and dried. The crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 60:1) to yield the title compound as a white solid (0.20 g, 82 %).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.78 (s, 1 H, NH), 8.16 (d, J= 7.4 Hz, 2H, PhH), 7.81-7.85 (m, 3H, PhH), 7.57 (s, 1 H, CH), 7.55 (t, J= 7.4 Hz, 2H, PhH), 7.30 (t, J= 8.5 Hz, PhH) ppm.
- HRMS: calcd for C18H12FN2OS 323.06544, found 323.06461.
- This compound was prepared from example 317 in a yield of 96%, according to the procedure for the synthesis of example 322, using ethyl cyanoformate.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.97 (s, 1H, NH), 7.91 (s, 1H, CH), 7.86-7.89 (m, 2H, PhH), 7.32 (t, J= 8.8 Hz, PhH), 4.38 (q, J= 7.1 Hz, 2H, CH2), 1.36 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C15H12FN2O3S 319.05527, found 319.05433.
- This compound was prepared from example 317 in a yield of 93%, according to the procedure for the synthesis of example 322, using ethyl cyanoacetate.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 12.63 (s, 1H, NH), 7.79-7.84 (m, 2H, PhH), 7.77 (s, 1H, H-5), 7.30 (t, J= 8.7 Hz, PhH), 4.15 (q, J= 7.1 Hz, 2H, OCH2), 3.79 (s, 2H, CH2), 1.21 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C16H14FN2O3S 333.07092, found 333.07010.
- To a solution of 6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4(3H)-one (40 mg, 0.15 mmol) and BOP (88 mg, 0.20 mmol) in CH3CN (1 ml) was added DBU (34 µl, 0.23 mmol). After stirring for 10 minutes at room temperature, 2-(4-chlorophenoxy)-1-(piperazin-1-yl)ethanone (59 mg, 0.23 mmol) was added. The reaction was stirred at room temperature overnight and then heated at 60°C for 4 hours. The solvents were removed under reduced pressure, and the crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (66 mg, 86%).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.56-7.61 (m, 2H, PhH), 7.31 (s, 1H, CH), 7.25 (d, J= 8.9 Hz, 2H, PhH), 7.11 (t, J= 8.5 Hz, PhH), 6.90 (d, J= 8.9 Hz, 2H, PhH), 4.74 (s, 2H, CH2), 3.92 (br s, 4H, NCH2), 3.80 (br s, 4H, NCH2), 2.60 (s, 3H, CH3) ppm.
- HRMS: calcd for C25H23ClFN4O2S 497.12143, found 497.11983.
- This compound was prepared from example 320 in a yield of 55%, according to the procedure for the synthesis of example 325.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.52-7.57 (m, 2H, PhH), 7.27 (s, 1H, CH), 7.22 (d, J= 8.9 Hz, 2H, PhH) 7.10 (t, J= 8.7 Hz, PhH), 6.91 (d, J= 8.9 Hz, 2H, PhH), 4.78 (s, 2H, NH2), 4.74 (s, 2H, CH2), 3.86 (br s, 4H, NCH2), 3.78 (br s, 4H, NCH2) ppm.
- HRMS: calcd for C24H22ClFN5O2S 498.11668, found 498.11511.
- To a solution of 2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4-(3H)-one (100 mg, 0.39 mmol) in acetonitrile (20 ml) was added 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU, 0.58 mmol, 86 µl), benzotriazol-1-yloxytris(dimethylamino)-phosphonium hexafluorophosphate (BOP, 0.77 mmol, 84 µl) and benzylamine (0.77 mmol, 84 µl). The reaction was stirred at room temperature overnight. The reaction mixture was diluted with water and dichloromethane. The organic layer was washed with water. The combined organic layers were evaporated in vacuo and the residue was redissolved in ethylacetate. This solution was extracted with brine (3 x). The combined organic layers were again evaporated and the crude residue was purified by flash chromatography on silica using a mixture of methanol and dichloromethane (in a ratio gradually ranging from 1:99 to 3:97) as mobile phase, yielding the title compound (86 mg, 63 %).
- MS m/z (%) : 351 ([M+H]+, 100)
- This compound was synthesized from example 321 according to the procedure of example 327 in 71 % yield, using piperazine.
- MS m/z (%) : 312 ([M+H]+, 100)
- To a solution of 2-amino-4-N-piperazino-6-phenyl-thieno[2,3-d]pyrimidine (90 mg, 0.29 mmol) in dioxane (10 ml) was added diisopropylethylamine (0.58 mmol, 96 µl) and 4-chlorophenoxyacetyl chloride (0.35 mmol, 71 mg). The reaction was stirred at room temperature for 2 hours. The reaction mixture was diluted with dichloromethane and extracted with water and brine. The organic phase was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (115 mg, 83 %).
- MS m/z (%): 480 ([M+H]+, 100)
- To a solution of 2-amino-4-N-piperazino-6-phenyl-thieno[2,3-d]pyrimidine (70 mg, 0.23 mmol) in dioxane (10 ml) was added m-tolylisocyanate (0.27 mmol, 35 µl). The reaction was stirred at room temperature for 2 hours. The reaction mixture was diluted with dichloromethane and extracted with water and brine. The organic phase was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually ranging from 100% CH2Cl2 to 2% CH3OH in CH2Cl2), yielding the pure title compound (59 mg, 58 %).
- MS m/z (%) : 445 ([M+H]+, 100)
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.56-7.61 (m, 2H, arom H), 7.15-7.45 (m, 6H, arom H), 6.88 (s, 1 H, arom H), 6.33 (s, 1 H, arom H), 4.79 (2 H, br s, NH2), 4.01 (t, 4H, piperazine CH2), 3.74 (br s, 4H, piperazine CH2), 2.34 (br s, 3H, CH3) ppm.
- This compound was synthesized in 76 % yield from example 328 according to the procedure for the synthesis of example 330, using 4-chloro-phenylisocyanate.
- MS m/z (%) : 465 ([M+H]+, 100)
- To a solution of 2-amino-4-(N-piperazin-1-yl)-6-phenyl-thieno[2,3-d]pyrimidine (48 mg, 0.15 mmol) and pyridine (15 µl, 0.18 mmol) in DMF (1 ml) was added phenoxyacetyl chloride (0.17 mmol). The reaction mixture was stirred for 2 hours at room temperature. The reaction mixture was quenched with water, extracted with EtOAc, brine and was dried over Na2SO4. After removing the solvents, the crude residue was purified by flash chromatography on silica (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (39 mg, 58 %).
- MS m/z (%) : 446 ([M+H]+, 100)
- This compound was synthesized from example 321 according to the procedure of example 328, using homopiperazine, in 53 % yield.
- MS m/z (%) : 326 ([M+H]+, 100)
- This compound was synthesized in 69 % yield from example 333, according to the procedure described for the synthesis of example 332, using 4-chloro-phenoxyacetyl chloride.
- MS m/z (%) : 495 ([M+H]+, 100)
- This compound was synthesized from example 333 according to the procedure for the synthesis of example 332, using 4-chlorobenzoylchloride.
- MS m/z (%) : 464 ([M+H]+, 100)
- This compound was synthesized according to the procedure of example 327, using 2-(piperazin-1-yl)-acetic acid N-methyl-N-phenyl-amide in 52 % yield.
- MS m/z (%): 459 ([M+H]+, 100)
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.56 (d, 2H, arom H), 7.20-7.50 (m, 9H, arom H), 4.75 (br s, 2H, NH2), 6.33 (s, 1 H, arom H), 4.79 (2 H, br s, NH2), 4.01 (t, 4H, piperazine CH2), 3.74 (br s, 4H, piperazine CH2), 2.99 (2 H, s, CH2), 2.34 (br s, 3H, CH3) ppm
- This compound was synthesized according to the procedure of example 327, using 1-(2-phenoxyethyl)-piperazine in 49 % yield.
- MS m/z (%) : 432 ([M+H]+, 100)
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.57 (d, 2H, arom H), 7.25-7.35 (m, 5H, arom H), 6.94 (m, 4H, arom H), 4.79 (br s, 2H, NH2), 4.17 (2 H, t, CH2), 3.90 (t, 4H, piperazine CH2), 2.89 (t, 2H, CH2), 2.74 (4H, t, piperazine CH2) ppm.
- This compound was synthesized according to the procedure of example 327, in 61 % yield, using (R)-3-N-Boc-aminopyrrolidine.
- MS m/z (%) : 430 ([M+H]+, 100)
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.51 (m, 2H, arom H), 7.35 (s, 1H, arom H), 7.07 (t, 2H, arom H), 4.78 (br s, 2H, NH2), 4.38 (br s, 1 H, NH), 4.06-3.70 (m, 5H), 2.27 (m, 1 H, CH2), 2.04 (m, 1 H, CH2) ppm.
- To a solution of the compound of example 338 (66 mg, 0.15 mmol) in dichloromethane (6 ml) was added trifluoroacetic acid (3 ml). The solution was stirred at room temperature for 30 minutes. The solvents were evaporated in vacuo and co-evaporated with toluene. The residue was directly used for further reaction without any purification.
- MS m/z (%) : 330 ([M+H]+, 100)
- This compound was synthesized in 78 % yield from example 339 according to the procedure of example 332, using 4-chlorophenoxyacetyl chloride.
- MS m/z (%) : 498 ([M+H]+, 100)
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.42 (d, 1H, NH), 7.67 (d, 2H, arom H), 7.31 (t, 2H, arom H), 6.98(d, 2H, arom H), 6.21 (br s, 2H, NH2), 4.51 (s, 1H, CH2), 3.87-3.67 (m, 5H), 2.17 (m, 1H, CH2), 1.99 (m, 1H, CH2) ppm.
- This compound was synthesized from example 339 according to the procedure of example 332, using 4-chlorobenzoylchloride in 65% yield.
- MS m/z (%): 468 ([M+H]+, 100)
- This compound was synthesized according to the procedure of example 327, using piperazine in 54 % yield.
- MS m/z (%) : 330 ([M+H]+, 100)
- This compound was synthesized in 55 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine according to the procedure for the synthesis of example 329, using hydrocinnamoylchloride.
- MS m/z (%) : 462 ([M+H]+, 100)
- This compound was synthesized in 51 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine according to the procedure for the synthesis of example 329, using α-toluenesulfonyl chloride.
- MS m/z (%) : 484 ([M+H]+, 100)
- This compound was synthesized in 66 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine according to the procedure for the synthesis of example 329, using cyclohexanecarboxylic acid chloride.
- MS m/z (%) : 440 ([M+H]+, 100)
- This compound was synthesized in 49 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine using nicotinoyl chloride according to the procedure for the synthesis of example 329.
- MS m/z (%) : 435 ([M+H]+, 100)
- This compound was synthesized in 61 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine using diisopropylcarbamoyl chloride according to the procedure for the synthesis of example 329, using diisopropylcarbamoyl chloride.
- MS m/z (%) : 457 ([M+H]+, 100)
- This compound was synthesized in 44 % yield from 2-amino-4-N-piperazino-6-(4-fluorophenyl)-thieno[2,3-d]pyrimidine using 4-benzoylpiperidine hydrochloride according to the procedure for the synthesis of example 327, using 4-benzoylpiperidine hydrochloride. MS m/z (%) : 433 ([M+H]+, 100)
- 1H NMR (300 MHz, CHCl3, 25 °C): δ = 7.98 (d, 2H, arom H), 7.53 (m, 5H, arom H), 7.08 (t, 2H, arom H), 4.79 (br s, 2H, NH2), 4.55 (d, 2H, NCH2), 3.61 (m, 1 H, CH), 3.37 (t, 2H, NCH2), 2.01 (m, 4H, CH2) ppm.
- A suspension of ethyl 2-amino-thiophene-3-carboxylate (0.4 g, 2.34 mmol), chloroformamidine hydrochloride (0.67 g, 5.84 mmol) and dimethylsulfone (0.53 g, 11.7 mmol) was heated at 130°C for 30 minutes. After cooling down to room temperature, water (10 ml) was added and ammonium hydroxide was used to neutralize the suspension till pH = 8. The solid was filtered off, washed with water and dried, yielding the crude title compound (71 %, 277 mg). The crude residue was used as such for further reaction.
- MS m/z (%): 168 ([M+H]+, 100)
- To a solution of 2-amino-thieno[2,3-d]pyrimidin-4-(3H)-one (250 mg, 1.5 mmol) in acetonitrile (20 ml) was added 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU, 2.24 mmol, 335 µl), benzotriazol-1-yloxytris(dimethylamino)-phosphonium hexafluorophosphate (BOP, 862 mg) and piperazine (3 mmol, 258 mg). The reaction was stirred at room temperature overnight and then for 4 hours at 70°C. The solvents were evaporated in vacuo and the crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol, dichloromethane and a 33% aq. NH3 solution (in a ratio gradually ranging from 3: 96.5:0.5 to 5:94.5:0.5) yielding the title compound (48 %, 169 mg).
- MS m/z (%) : 236 ([M+H]+, 100)
- To a solution of 2-amino-4-N-piperazino-thieno[2,3-d]pyrimidine (100 mg, 0.43 mmol) in dioxane (15 ml) was added diisopropylethylamine (DIPEA, 0.85 mmol, 141 µl) and 4-chlorophenoxyacetylchloride (0.43 mmol, 88 mg). The reaction was stirred overnight at room temperature. The solvents were evaporated in vacuo. The residue was redissolved in ethylacetate and extracted with brine (3 x). The combined organic layers were evaporated in vacuo and the crude residue was purified by silica gel flash chromatography, the mobile phase consisting of a mixture of methanol and dichloromethane (in a ratio gradually ranging from 1:99 to 2:98), yielding the pure title compound (121 mg, 70 %).
- MS m/z (%) : 404 ([M+H]+, 100)
- 1H NMR (300 MHz, CHCl3, 25 °C): δ = 7.25 (d, 2H, arom H), 7.13 (d, 1H, arom H), 6.91 (d, 1H, arom H), 6.89 (d, 1H, arom H), 4.74 (br s, 2H, NH2), 4.73 (s, 2H, CH2), 3.83 (br s, 4H, CH2), 3.77 (br s, 1H, CH2) ppm.
- This compound was prepared from example 322 in a yield of 77%, according to the procedure for the synthesis of example 325.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.41-8.45 (m, 2H, PhH), 7.58-7.63 (m, 2H, PhH), 7.45-7.47 (m, 3H, PhH), 7.33 (s, 1H, CH), 7.26 (d, J= 8.9 Hz, 2H, PhH), 7.13 (t, J= 8.5 Hz, PhH), 6.92 (d, J= 8.9 Hz, 2H, PhH), 4.74 (s, 2H, CH2), 4.01 (br s, 4H, NCH2), 3.85 (br s, 4H, NCH2) ppm.
- HRMS: calcd for C30H25ClFN4O2S 559.13708, found 559.13554.
- This compound was prepared from example 323 in a yield of 89%, according to the procedure for the synthesis of example 325.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.62-7.67 (m, 2H, PhH), 7.42 (s, 1H, CH), 7.26 (d, J= 8.9 Hz, 2H, PhH), 7.16 (t, J= 8.5 Hz, PhH), 6.91 (d, J= 8.9 Hz, 2H, PhH), 4.75 (s, 2H, CH2), 4.51 (q, J= 7.1 Hz, 2H, CH2), 4.09 (br s, 2H, NCH2), 4.03 (br s, 2H, NCH2), 3.84 (br s, 4H, NCH2), 1.47 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C27H25ClFN4O4S 555.12691, found 555.12507.
- This compound was prepared from example 324 in a yield of 58%, according to the procedure for the synthesis of example 325.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.56-7.60 (m, 2H, PhH), 7.32 (s, 1H, CH), 7.24 (d, J= 8.8 Hz, 2H, PhH), 7.10 (t, J= 8.5 Hz, PhH), 6.89 (d, J= 8.8 Hz, 2H, PhH), 4.72 (s, 2H, OCH2), 4.21 (q, J= 7.1 Hz, 2H, CH2), 3.92 (br s, 4H, NCH2), 3.88 (s, 2H, CH2), 3.78 (br s, 4H, NCH2), 1.28 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C28H27ClFN4O4S 569.1426, found 569.1426.
- A suspension of ethyl 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate (40 mg, 0.07 mmol) in 7N NH3 in MeOH (1 ml) was stirred at room temperature for 3 hours. The solvents were removed under reduced pressure, and the crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 40:1) to yield the title compound as a white solid (25 mg, 66%).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.14 (s, 1H, NH), 8.00 (s, 1H, CH), 7.90-7.95 (m, 2H, PhH), 7.35 (t, J= 8.8 Hz, 2H, PhH), 7.33 (d, J= 9.0 Hz, 2H, PhH), 6.99 (d, J= 9.0 Hz, 2H, PhH), 4.94 (s, 2H, CH2), 4.15 (br s, 2H, NCH2), 4.08 (br s, 2H, NCH2), 3.73 (br s, 4H, NCH2) ppm.
- HRMS: calcd for C25H22ClFN5O3S 526.11159 found 526.11031.
- To a suspension of ethyl 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate (30 mg, 0.05 mmol) in MeOH (7 ml) was added 2-methoxyethylamine (0.7 ml). The mixture was stirred at room temperature for 3 hours. The solvents were removed under reduced pressure and the crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (29 mg, 92%).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.20 (s, 1 H, NH), 7.59-7.64 (m, 2H, PhH), 7.40 (s, 1 H, CH), 7.25 (d, J= 8.8 Hz, 2H, PhH), 7.14 (t, J= 7.15 Hz, 2H, PhH), 6.91 (d, J= 8.8 Hz, 2H, PhH), 4.75 (s, 2H, CH2), 4.05 (br s, 2H, NCH2), 3.99 (br s, 2H, NCH2), 3.69 (br s, 4H, NCH2),3.69 (t, J= 5.1 Hz, 2H, CH2), 3.60 (t, J= 5.1 Hz, 2H, CH2), 3.41 (s, 3H, CH3) ppm.
- HRMS: calcd for C28H28ClFN5O4S 584.15346, found 584.15178.
- A solution of ethyl 4-(4-(2-(4-chlorophenoxy)acetyl)piperazin-1-yl)-6-(4-fluorophenyl)thieno[2,3-d]pyrimidine-2-carboxylate (0.12 g, 0.22 mmol) in methanol/2M NaOH/CH2Cl2 (5:5:1, 5 ml) was stirred at room temperature for 3 hours after which it was neutralized with a 2N HCl solution in dioxane. The solvents were removed under reduced pressure, and the crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 8:1) to yield the title compound as a pale yellow solid (50 mg, 44%).
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.02 (s, 1H, CH), 7.92-7.97 (m, 2H, PhH), 7.32-7.39 (m, 4H, PhH), 6.98 (d, J= 8.7 Hz, 2H, PhH), 4.93 (s, 2H, CH2), 4.11 (br s, 2H, NCH2), 4.04 (br s, 2H, NCH2), 3.68 (br s, 4H, NCH2) ppm.
- HRMS: calcd for C25H21ClFN4O4S 527.09561, found 527.09421.
- This compound was prepared from example 354 in a yield of 34%, according to the procedure for the synthesis of example 355.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 7.92 (s, 1H, H-5), 7.86-7.91 (m, 2H, PhH), 7.44 (s, 1H, NH), 7.30-7.36 (m, 4H, PhH, PhH), 6.99 (s, 1H, NH), 6.98 (d, J= 9.1 Hz, 2H, PhH), 4.92 (s, 2H, OCH2), 4.03 (br s, 2H, NCH2), 3.98 (br s, 2H, NCH2), 3.70 (br s, 4H, CON(CH2)2), 3.57 (s, 2H, CH2) ppm.
- HRMS: calcd for C26H24ClFN5O3S 540.12724 found 540.12544.
- This compound was prepared from example 320 in a yield of 46%, according to the procedure for the synthesis of example 325, using N-m-tolylpiperazine-1-carboxamide.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 8.50 (s, 1 H, NH), 7.73-7.78 (m, 2H, PhH), 7.71 (s, 1 H, PhH), 7.23-7.32 (m, 4H, PhH), 7.12 (t, J= 7.3Hz, PhH), 6.77 (d, J= 7.3 Hz, 1H, PhH), 6.36 (s, 2H, NH2), 3.88 (br s, 4H, NCH2), 3.64 (br s, 4H, NCH2), 2.26 (s, 3H, CH3) ppm.
- HRMS: calcd for C24H24FN6OS 463.1716, found 463.1702.
- This compound was prepared from example 322 in a yield of 69%, according to the procedure for the synthesis of example 325, using N-m-tolylpiperazine-1-carboxamide.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.54 (s, 1 H, NH), 8.41-8.44 (m, 2H, 5-PhH), 7.99 (s, 1 H, CH), 7.89-7.94 (m, 2H, 2-PhH), 7.50-7.52 (m, 3H, PhH), 7.28-7.38 (m, 4H, PhH), 7.13 (t, J= 7.7 Hz, 1 H, tolyl-H), 6.78 (d, J= 7.4 Hz, 1 H, tolyl-H), 4.13 (br s, 4H, NCH2), 3.75 (br s, 4H, NCH2), 2.27 (s, 3H, CH3) ppm.
- HRMS: calcd for C30H27FN5OS 524.1920, found 524.1921.
- This compound was prepared from example 323 in a yield of 61%, according to the procedure for the synthesis of example 325, using N-m-tolylpiperazine-1-carboxamide.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.60-7.65 (m, 2H, PhH), 7.46 (s, 1H, CH), 7.11-7.17 (m, 4H, PhH), 6.87 (br s, 1 H, PhH), 6.66 (s, 1 H, PhH), 4.50 (q, J= 7.1 Hz, 2H, CH2), 4.15 (br s, 4H, NCH2), 3.79 (br s, 4H, NCH2), 1.46 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C27H27FN5O3S 520.18186, found 520.17993.
- This compound was prepared from example 361 in a yield of 66%, according to the procedure for the synthesis of example 355.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 8.55 (s, 1H, NH), 8.15 (s, 1H, CONH), 8.02 (s, 1H, CH), 7.91-7.95 (m, 2H, PhH), 7.68 (s, 1H, CONH), 7.27-7.38 (m, 4H, PhH), 7.13 (t, J= 7.7 Hz, 1H, PhH), 6.77 (d, J= 7.7 Hz, 1H, PhH), 4.11 (br s, 4H, NCH2), 3.71 (br s, 4H, NCH2), 2.26 (s, 3H, CH3) ppm.
- HRMS: calcd for C25H24FN6O2S 491.16655, found 491.16526.
- To a solution of 2-amino-6-(4-fluorophenyl)thieno[2,3-d]pyrimidin-4(3H)-one (50 mg, 0.19 mmol) and BOP (110 mg, 0.25 mmol) in EtOH (1 ml) was added DBU (43 µl, 0.29 mmol). The resulting mixture was heated at 60°C for 4 hours. After cooling down to room temperature, sodium ethoxide (26 mg, 0.38 mmol) was added. The mixture was again heated at 60°C overnight. The solvents were removed under reduced pressure and the crude residue was purified by flash chromatography on silica gel (CH2Cl2/MeOH 60:1) yielding the title compound as a white solid (30 mg, 54 %).
- mp 142°C
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.54-7.59 (m, 2H, PhH), 7.29 (s, 1H, PhH), 7.07 (t, J= 8.6 Hz, PhH), 5.03 (s, 2H, NH2), 4.49 (q, J= 7.1 Hz, 2H, CH2), 1.44 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C14H13FN3OS 290.0763, found 290.0740.
- This compound was prepared from example 320 using morpholine in a yield of 58%, according to the procedure for the synthesis of example 327.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.52-7.75 (m, 2H, PhH), 7.24 (s, 1H, CH), 7.09 (t, J= 8.6 Hz, PhH), 4.82 (s, 2H, NH2), 3.84 (br s, 8H, NCH2, OCH2) ppm.
- HRMS: calcd for C16H16FN4OS 331.1029, found 331.1003.
- A solution of 6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidin-4(3H)-one (0.2 g, 0.77 mmol) and diisopropylethylamine (0.26 ml, 1.54 mmol) in POCl3 (4 ml) was stirred under N2 at 90°C for 3.5 hours. The reaction mixture was allowed to cool down to room temperature and poured into an ice-bath. The aqueous phase was extracted with diethyl ether. The combined organic layers were washed with a half-saturated NaHCO3 solution and brine, dried over Na2SO4, and concentrated under reduced pressure. The crude residue was purified by silica gel chromatography (CH2Cl2/MeOH 50:1) to yield the title compound as a white solid (14 mg, 6%).
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.66-7.71 (m, 2H, PhH), 7.45 (s, 1H, CH), 7.16 (t, J= 8.5 Hz, PhH), 2.81 (s, 3H, CH3) ppm.
- HRMS: calcd for C13H9ClFN2S 279.01590, found 279.01510.
- This compound was prepared from example 323 in a yield of 99%, according to the procedure for the synthesis of example 365.
- 1H NMR (300 MHz, CDCl3, 25 °C): δ = 7.71-7.76 (m, 2H, PhH), 7.57 (s, 1H, CH), 7.19 (t, J= 8.5 Hz, 2H, PhH), 4.58 (q, J= 7.1 Hz, 2H, CH2), 1.49 (t, J= 7.1 Hz, 3H, CH3) ppm.
- HRMS: calcd for C15H11ClFN2O2S 337.02138, found 337.02038.
- To a solution of 4-chloro-6-(4-fluorophenyl)-2-methylthieno[2,3-d]pyrimidine (15 mg, 0.05 mmol) in 1,2-dichloroethane/t-BuOH (1:1, 2 ml) was added 3-chloro-4-fluoroaniline (16 mg, 0.11 mmol). The mixture was heated at 90°C for 2 days. After cooling down to room temperature, the solvents were removed under reduced pressure. The crude residue was purified by silica gel chromatography (CH2Cl2/MeOH 100:1) to yield the title compound as a white solid (11 mg, 53 %).
- HRMS: calcd for C19H13ClF2N3S 388.0487, found 388.0471.
- This compound was prepared from example 366 in a yield of 38%, according to the procedure for the synthesis of example 367.
- 1H NMR (300 MHz, DMSO, 25 °C): δ = 10.04 (s, 1H, NH), 8.65-8.67 (m, 1H, PhH), 8.27 (s, 1H, PhH), 7.79-7.84 (m, 3H, PhH), 7.39-7.51 (m, 3H, PhH), 4.37 (q, J= 7.0 Hz, 2H, CH2), 1.39 (t, J= 7.0 Hz, 3H, CH3) ppm.
- HRMS: calcd for C21H15ClF2N3O2S 446.05416, found 446.05311.
- To a solution of 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine (0.65 mmol) in DMF (10 ml) was added diisopropylethylamine (1.3 mmol, 215 µl), O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU, 0.78 mmol, 250 mg) and an appropriate carboxylic acid (0.78 mmol). The reaction was stirred at room temperature overnight. The reaction was diluted with dichloromethane and extracted with water. The combined organic layers were evaporated in vacuo and the resulting residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 1% to 2% methanol), yielding the pure title compounds in yields varying from 62% to 75%.
- The following compounds were made according to this procedure
- This compound was obtained from 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine and 4-methoxy-phenoxyacetic acid.
- 1H NMR (300 MHz, CDCl3): δ = 7.59 (d, 2H, arom H), 7.26-7.40 (m, 4H, arom H), 6.86-6.90 (m, 4H, arom H), 4.81 (br s, 2H, NH2), 4.71 (s, 2H, CH2), 3.79 (br s, 4H, piperazine H), 3.77 (br s, 7H, piperazine H and OCH3) ppm.
- This compound was obtained from 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine and 4-fluoro-phenoxyacetic acid.
- 1H NMR (300 MHz, DMSO): δ = 7.57 (m, 2H, arom H), 7.26-7.43 (m, 4H, arom H), 6.93-6.99 (m, 4H, arom H), 5.13 (br s, 2H, NH2), 4.72 (s, 2H, CH2), 3.91 (br s, 4H, piperazine H), 3.81 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine and 3-methyl-phenoxyacetic acid.
- This compound was obtained from 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine and 4-bromo-phenoxyacetic acid.
- 1H NMR (300 MHz, CDCl3): δ = 7.57 (m, 2H, arom H), 7.26-7.41 (m, 4H, arom H), 6.86 (m, 4H, arom H), 4.73 (br s, 2H, NH2), 4.64 (s, 2H, CH2), 3.92 (br s, 4H, piperazine H), 3.80 (br s, 4H, piperazine H) ppm.
- This compound was obtained from 2-amino-4-(N-piperazinyl)-6-phenyl-thieno[2,3-d]pyrimidine and 2-(4-chlorophenoxy)-2-methylpropionic acid.
- 1H NMR (300 MHz, CDCl3): δ = 7.57 (d, 2H, arom H), 7.39 (t, 2H, arom H), 7.19-7.32 (m, 4H, arom H), 6.79 (d, 2H, arom H), 4.78 (br s, 2H, NH2), 4.01 (br s, 2H, piperazine H), 3.80 (br s, 4H, piperazine H), 3.56 (br s, 2H, piperazine H), 1.67 (s, 6 H, 2 x CH3) ppm.
- A mixture of 4-chloro-2,6-diaminopyrimidine (4.34 g, 30 mmol), piperazine (2.58 g, 30 mmol) and NaOH (1.2 g, 30 mmol) in water (50 ml) was heated under reflux for 3 hours. After cooling to room temperature, the precipitate was filtered off. The filtrate was concentrated under reduced pressure to yield the crude title product which was used in the following step without further purification.
- MS m/z (%): 195 ([M+H]+, 100)
- To a solution of 2,6-diamino-4-(N-piperazin-1-yl)pyrimidine (crude residue, +/- 30 mmol) and potassium carbonate (8.28 g, 60 mmol) in dioxane/methanol (1:1 ; 100 ml), was added a solution of the appropriate acid chloride (36 mmol) in dioxane (20 ml). The resulting reaction mixture was stirred at room temperature for 1 hour. After concentration under reduced pressure, the residue was resuspended in water (50 ml). The solid was filtered off and washed with water. The precipitate was dried over P2O5, yielding the title compound in yields between 70 - 90 % over 2 steps.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized using 4-chlorophenoxyacetyl chloride.
- MS m/z (%): 363 ([M+H]+, 100)
- This compound was synthesized using phenoxyacetyl chloride.
- MS m/z (%): 329 ([M+H]+, 100)
- This compound was synthesized using 3-methoxybenzoyl chloride.
- MS m/z (%): 329 ([M+H]+, 100)
- This compound was synthesized using 2-thiophene-acetyl chloride.
- MS m/z (%): 319 ([M+H]+, 100)
- This compound was synthesized using 4-chloro-benzoyl chloride.
- MS m/z (%):333 ([M+H]+, 100)
- This compound was synthesized using α-toluenesulfonylchloride.
- MS m/z (%):349 ([M+H]+, 100)
- This compound was synthesized using 1-naphthoylchloride.
- MS m/z (%):349 ([M+H]+, 100)
- To a suspension of 2,6-diamino-4-[4-acyl-piperazin-1-yl]pyrimidine (10 mmol) and sodium nitrite (0.86 g, 12.5 mmol) in a mixture of water/dioxane/methanol (4/2/1 ; 70 ml), was added acetic acid (1.5 g, 25 mmol). The resulting reaction mixture was stirred at room temperature for 2 hours. A pink precipitate was formed indicating the formation of the nitroso intermediate. To this violet suspension was added a 30% ammonia solution in water (75 mmol) and sodium dithionite (25 mmol). The reaction mixture was stirred at 50°C till the violet color completely disappeared (about 2 hours). After concentration under reduced pressure, the residue was suspended in water (50 ml). The solid was filtered off, washed with water and dried over P2O5, yielding the title compound as white solids in yields varying from 60 to 90 %. The following compounds were made according to this procedure :
- MS m/z (%): 378 ([M+H]+, 100)
- MS m/z (%): 344 ([M+H]+, 100)
- MS m/z (%): 344 ([M+H]+, 100)
- MS m/z (%): 334 ([M+H]+, 100)
- MS m/z (%): 348 ([M+H]+, 100)
- MS m/z (%): 364 ([M+H]+, 100)
- MS m/z (%): 378 ([M+H]+, 100)
- A suspension of a 2,5,6-triamino-4-[4-acyl-piperazin-1-yl]pyrimidine analogue (0.5 mmol), an appropriate aldehyde (0.5 mmol) and a drop of acetic acid in methanol (10 ml) was stirred at room temperature for 1 hour, after which the Schiff base intermediate was formed. Then, FeCl3 (0.75 mmol) was added. The mixture was stirred at room temperature overnight. The reaction mixture was evaporated in vacuo and purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1:25), yielding the pure title compound.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde, yielding the pure title compound in 83% yield.
- MS m/z (%): 482 ([M+H]+, 100)
- This compound was synthesized from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and 3,4-dimethoxybenzaldehyde yielding the pure title compound in 57 % yield.
- MS m/z (%): 524 ([M+H]+, 100)
- This compound was synthesized from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and 4-bromobenzaldehyde in 74 % yield.
- MS m/z (%): 543 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and 4-chlorobenzaldehyde in 60 % yield.
- MS m/z (%): 498 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and 3-chlorobenzaldehyde in 67 % yield.
- MS m/z (%): 498 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and p-trifluoromethylbenzaldehyde in 63 % yield.
- MS m/z (%): 532 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and p-trifluoromethoxybenzaldehyde in 61 % yield.
- MS m/z (%): 548 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and p-methylbenzaldehyde in 56 % yield.
- MS m/z (%): 478 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and butyraldehyde in 47 % yield.
- MS m/z (%): 430 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and cyclopropanecarboxaldehyde, in 78 % yield.
- MS m/z (%): 428 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and trimethylacetaldehyde in 45 % yield.
- MS m/z (%): 444 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]pyrimidine and acetaldehyde in 30 % yield.
- MS m/z (%): 402 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[(4-phenoxyacetyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 37 % yield.
- MS m/z (%): 448 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[(3-methoxy-benzoyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 41 % yield.
- MS m/z (%): 448 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[(2-thiophene-acetyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 28 % yield.
- MS m/z (%): 438 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[(4-chloro-benzoyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 31 % yield.
- MS m/z (%): 452 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[4-α-toluenesulfonyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 21 % yield
- MS m/z (%): 468 ([M+H]+, 100)
- This compound was obtained from 2,5,6-triamino-4-[(1-naphthoyl)piperazin-1-yl]pyrimidine and 4-fluorobenzaldehyde in 41 % yield.
- MS m/z (%): 468 ([M+H]+, 100)
- This compound was synthesized from commercially available 2,5,6-triamino-4-hydroxypyrimidine (40 mmol) and 4-fluorobenzaldehyde (40 mmol), yielding the title compound in 60 % yield.
- MS m/z (%): 246 ([M+H]+, 100)
- To a solution of 2-amino-6-hydroxy-8-(4-fluorophenyl)-9H-purine (0.4 mmol), a nitrogen nucleophile (0.6 mmol) and benzotriazol-1-yloxytris(dimethylamino)-phosphonium hexafluorophosphate (BOP, 0.65 mmol) in DMF (5 ml), was added 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU, 0.5 mmol). The resulting reaction mixture was stirred at room temperature until all starting material disappeared on TLC (4 - 8 hours). The solvents were evaporated in vacuo. The crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1:30), yielding the pure title compounds.
- The following compounds were synthesized according to this procedure :
- This compound was obtained from 1-acetylpiperazine in 63 % yield.
- MS m/z (%): 356 ([M+H]+, 100)
- This compound was obtained from 1-(thiazol-2-yl)piperazine in 63 % yield.
- MS m/z (%): 396 ([M+H]+, 100)
- This compound was synthesized from 1-(piperazin-1-yl)-2-(pyrrolidin-1-yl)ethanone in 45 % yield.
- MS m/z (%): 425 ([M+H]+, 100)
- This compound was synthesized from 1-morpholino-2-(piperazin-1-yl)ethanone in 45 % yield.
- MS m/z (%): 441 ([M+H]+, 100)
- This compound was obtained from 2-(piperazin-1-yl)-N-(pyridin-3-yl)acetamide in 16 % yield.
- MS m/z (%): 448 ([M+H]+, 100)
- This compound was obtained from 2-(piperazin-1-yl)-N-methyl-N-phenylacetamide in 43 % yield.
- MS m/z (%): 461 ([M+H]+, 100)
- This compound was obtained from 1-(4-chlorophenyl)piperazine in 58 % yield.
- MS m/z (%): 424 ([M+H]+, 100)
- This compound was obtained from 4-fluorophenylpiperazine in 55 % yield.
- MS m/z (%): 408 ([M+H]+, 100)
- This compound was obtained from 2-(piperazin-1-yl)-N-(pyridin-2-yl)acetamide in 37 % yield.
- MS m/z (%): 448 ([M+H]+, 100)
- This compound was obtained from 2-(piperazin-1-yl)-N-(thiazol-2-yl)acetamide in 37 % yield.
- MS m/z (%): 454 ([M+H]+, 100)
- This compound was obtained from 4-fluorobenzylpiperazine yielding the pure title compound in 36 % yield.
- MS m/z (%): 421 ([M+H]+, 100)
- This compound was obtained from 1-(pyridin-4-yl)piperazine yielding the title compound in 70 % yield.
- MS m/z (%): 391 ([M+H]+, 100)
- This compound was obtained from homopiperazine, yielding the title compound in 92 % yield.
- MS m/z (%): 328 ([M+H]+, 100)
- To a solution of 2-amino-6-(homopiperazin-1-yl)-8-(4-fluorophenyl)-9H-purine (0.2 mmol) in dioxane/methanol (1:1 ; 5 ml) was added potassium carbonate (55 mg, 0.4 mmol) and a solution of 4-chlorophenoxyacetyl chloride (60 mg, 0.24 mmol) in dioxane (1 ml). The resulting mixture was stirred at room temperature for 1 hour. After concentration under reduced pressure, the residue was purified by flash chromatography on silica (methanol/dichloromethane 1:40), yielding the pure title compound (60 mg, 60 %).
- MS m/z (%): 496 ([M+H]+, 100)
- To a solution of 2-amino-6-(N-homopiperazin-1-yl)-8-(4-fluorophenyl)-9H-purine (0.2 mmol) in dioxane/methanol (1:1; 5 ml) was added m-tolyl isocyanate (0.3 mmol). The resulting mixture was stirred at room temperature for 2 hours. After concentration under reduced pressure, the residue was purified by flash chromatography on silica (methanol/dichloromethane 1:40), yielding the pure title compound (60 mg, 65%).
- MS m/z (%): 461 ([M+H]+, 100)
- A mixture of 2,5,6-triamino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-pyrimidine (1.3 g, 3.4 mmol), sodium bicarbonate (1.68 g, 20 mmol) and carbon disulfide (2.5 ml, 40 mmol) in ethanol/water (1:2 ; 30 ml) was heated under reflux for 8 hours. After cooling down to room temperature, the pH of the mixture was adjusted to 5-6. The precipitate was filtered off, washed with water, and dried over P2O5, yielding the title compound (1.0 g, 70 %).
- MS m/z (%): 420 ([M+H]+, 100)
- To a solution of 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-thiol-purine (0.24 mmol) and potassium carbonate (0.5 mmol) in DMF (5 ml), was added an appropriate alkyl halide (0.24 mmol). The resulting mixture was stirred at room temperature for 30 minutes. After concentration under reduced pressure, the residue was purified by flash chromatography on silica (methanol/dichloromethane 1:40), yielding the pure title compounds as a white solid.
- The following compounds were synthesized according to this procedure :
- This compound was synthesized using methyl iodide in 67 % yield.
- MS m/z (%): 434 ([M+H]+, 100)
- This compound was synthesized using propyl iodide yielding the title compound in 65 % yield.
- MS m/z (%): 462 ([M+H]+, 100)
- This compound was synthesized using benzyl bromide yielding the title compound in 63 % yield.
- MS m/z (%): 510 ([M+H]+, 100)
- This compound was synthesized using phenethylbromide yielding the title compound in 67 % yield.
- MS m/z (%): 524 ([M+H]+, 100)
- This compound was synthesized from 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-methylthio-9H-purine using methyl iodide, yielding the title compound in 97 % yield.
- MS m/z (%): 448 ([M+H]+, 100)
- This compound was prepared from 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-thiol-9H-purine and cyclopentyl iodide yielding the title compound in 72 % yield.
- MS m/z (%): 488 ([M+H]+, 100)
- To a solution of 2-amino-6-[4-(4-chlorophenoxyacetyl)piperazin-1-yl]-8-(4-fluorophenyl)-9H-purine (48 mg, 0.1 mmol) in DMF (2 ml) was added potassium carbonate (28 mg, 0.2 mmol) and methyl iodide (6 µl, 0.1 mmol). The reaction mixture was stirred at room temperature for 30 minutes. The reaction mixture was evaporated in vacuo and the crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1:50), yielding the pure title compound (40 mg, 80%).
- MS m/z (%): 496 ([M+H]+, 100)
- This compound was synthesized according to the procedure mentioned for example 430, using benzylbromide instead of iodomethane, yielding the pure title compound in 68 % yield.
- MS m/z (%): 591 ([M+H]+, 100)
- To a solution of 2,6-diamino-4-mercaptopyrimidine sulfate salt (2 g, 10 mmol) in water (20 ml) was added potassium hydroxide (21 mmol, 1.17 g) and iodomethane (16 mmol, 977 µL). The reaction mixture was stirred for 1,5 hours at room temperature, after which a yellow precipitate was formed. The precipitate was filtered off, washed with water and dried. The product was used for further reactions without any further purification.
- MS m/z (%): 157 ([M+H]+, 100)
- To a suspension of 2,6-diamino-4-thiomethyl-pyrimidine (1.21 g, 7.75 mmol) in water (17 ml) was added acetic acid (15 mmol, 888 µl) and sodium nitrite (9.3 mmol, 641 mg). The reaction was stirred at room temperature for 1 hour, after which a pink precipitate was formed. The reaction mixture was put in the fridge for several hours and the precipitate was filtered off, yielding the title compound.
- To a solution of 2,6-diamino-5-nitroso-4-thiomethyl-pyrimidine (140 mg, 0.75 mmol) in THF (10 ml) was added triethylamine (0.98 mmol, 136 µl) and 4-fluorobenzoylchloride (0.83 mmol, 98 µl). The reaction was stirred at room temperature for 3 hours. The solvents were evaporated and the crude residue was used for further reaction, without any purification.
- The crude residue, as obtained in Example 61, was redissolved in o-xylene (10 ml) and triphenylphosphine (1.5 mmol, 393 mg) was added. The reaction mixture was heated at 140 °C overnight. The solvents were evaporated in vacuo and the crude residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 0.5 % to 2 % methanol), yielding the pure title compound (60 mg)
- MS m/z (%): 276 ([M+H]+, 100)
- To a solution of 2-amino-6-thiomethyl-8-(4-fluorophenyl)-9H-purine (60 mg, 0.22 mmol) in dichloromethane (10 ml) was added m-chloroperoxybenzoic acid (mCPBA, 0.66 mmol, 113 mg) at 0°C. The reaction temperature was gradually increased to room temperature over 3 hours. The solvents were evaporated in vacuo yielding a mixture of the corresponding sulfoxide and sulfon derivative, which were used as such in the following reaction.
- To a solution of the crude residue (as obtained in Example 63) in dioxane (10 ml) was added piperazine (1.1 mmol, 94 mg). The reaction was heated at 100 °C overnight. The solvents were evaporated in vacuo and the crude residue was further purified by flash chromatography on silica, with methanol and dichloromethane as the mobile phase (in a ratio gradually ranging from 5 % to 6 % methanol always with 0.5 % aqueous ammonia solution), yielding the pure title compound (30 mg).
- MS m/z (%): 314 ([M+H]+, 100)
- To a solution of 2-amino-6-(N-piperazin-1-yl)-8-(4-fluorophenyl)-9H-purine (30 mg, 0.0958 mmol) in dioxane (10 ml) was added diisopropylethylamine (0.24 mmol, 40 µl) and hydrocinnamoyl chloride (0.12 mmol, 17 µl). The reaction was stirred overnight at room temperature. The solvents were evaporated in vacuo and the crude residue was purified by silica gel flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio gradually raising from 1 % to 3 % methanol) yielding the pure title compound (70% , 28 mg).
- MS m/z (%): 446 ([M+H]+, 100)
- To a suspension of 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine (50 mg, 0.16 mmol) in dioxane (10 ml) was added 4-trifluorotolyl isocyanate (0.18 mmol). The resulting reaction mixture was stirred at room temperature for 30 minutes. The solvents were evaporated in vacuo and the crude residue was purified by flash chromatography, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1:25), yielding the pure title compound as yellowish solid (50 mg, 63 %).
- MS m/z (%): 501 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.13 (d, J = 2.2 Hz, 1 H, Ar-H), 8.98 (s, 1 H, ArNH-), 8.66 (dd, J = 4.7, 1.5 Hz, 1 H, Ar-H), 8.27 (dt, J = 8.0, 1.5 Hz, 1 H, Ar-H), 7.72 (d, J = 8.6 Hz, 2H, Ar-H), 7.61 (d, J = 8.6 Hz, 2H, Ar-H), 7.55 (dd, J = 8.0, 4.7 Hz, 1 H, Ar-H), 6.56 (s, 2H, NH2), 4.32 (br s, 4H, NCH2), 3.67(br s, 4H, NCH2) ppm.
- This compound was synthesized according to the procedure of example 439, using phenyl isocyanate, yielding the title compound in 67% yield.
- MS m/z (%): 433 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.12 (d, J = 1.8 Hz, 1H, Ar-H), 8.66 (dd, J = 4.8, 1.6 Hz, 1H, Ar-H), 8.58 (s, 1H, ArNH-), 8.27 (dt, J = 7.9, 1.5 Hz, 1H, Ar-H), 7.54 (dd, J = 7.9Hz, 4.0 Hz, 1 H, Ar-H), 7.49 (d, J = 8.0 Hz, 2H, Ar-H), 7.25 (t, J = 7.7 Hz, 2H, Ar-H), 6.95 (t, J = 7.2 Hz, 1 H, Ar-H), 6.55 (s, 2H, NH2), 4.31 (br s, 4H, NCH2), 3.64 (br s, 4H, NCH2) ppm.
- This compound was synthesized according to the procedure of example 439, using cyclohexyl isocyanate, yielding the title compound in 63 % yield.
- MS m/z (%): 439 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.09 (d, J = 1.2 Hz, 1 H, Ar-H), 8.65 (d, J = 4.7 Hz, 1 H, Ar-H), 8.25 (d, J = 8.0 Hz, 1 H, Ar-H), 7.53 (dd, J = 8.0 Hz, 4.7 Hz, 1 H, Ar-H), 6.52 (s, 2H, NH2), 6.21 (d, J = 7.6 Hz, -CONH), 4.21 (br s, 4H, NCH2), 3.46 (br s, 5H, NCH- & NCH2), 1.67 (m, 4H, CH2), 1.20 (m, 6H, CH2) ppm.
- This compound was synthesized according to the procedure of example 439, using 4-fluorophenyl isocyanate, yielding the title compound in 72 % yield.
- MS m/z (%): 451 ([M+H]+, 100)
- 1H NMR (300 MHz, DMSO-d6, 25 °C): δ = 9.12(d, J=2.2 Hz, 1 H, Ar-H), 8.66(d, J=4.8 Hz, 1 H, Ar-H), 8.63(s, 1 H, ArNH-), 8.27(d, J=8.0 Hz, 1 H, Ar-H), 7.54(dd, J=8.0 Hz, 4.8 Hz, 1 H, Ar-H), 7.51 (dd, J=8.9, 5.4 Hz, 2H, Ar-H), 7.09(t, J=8.8 Hz, 2H, Ar-H), 6.56(s, 2H, NH2), 4.31 (br. s, 4H, NCH2), 3.64(br.s, 4H, NCH2)ppm.
- This compound was synthesized according to the procedure of example 439, using hexyl isocyanate, yielding the title compound in 72 % yield.
- MS m/z (%): 441 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.09 (dd, J = 2.2, 0.6 Hz, 1H, Ar-H), 8.65 (dd, J = 4.8, 1.6 Hz, 1 H, Ar-H), 8.24 (dt, J = 8.0, 2.2 Hz, 1 H, Ar-H), 7.54 (ddd, J=8.0, 4.8, 0.6 Hz, 1 H, Ar-H), 6.51 (s, 3H, NH2 & -CONH-), 4.22 (br s, 4H, NCH2), 3.46 (br t, J = 5.0 Hz, 4H, NCH2), 3.03 (q, J = 6.9 Hz, 2H, -NHCH2-), 1.41 (m, 2H, CH2), 1.25 (m, 6H, CH2), 0.86 (t, J = 7.0 Hz, CH3) ppm.
- This compound was synthesized according to the procedure of example 439, using 4-tolyl isothiocyanate, yielding the title compound in 71 % yield.
- MS m/z (%): 463 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.29 (s, 1H, CONH-), 9.12 (dd, J = 2.2, 0.6 Hz, 1 H, Ar-H), 8.65 (dd, J = 4.8, 1.6 Hz, 1 H, Ar-H), 8.27 (dt, J = 8.0, 1.6 Hz, 1 H, Ar-H), 7.53 (ddd, J = 8.0 Hz, 4.8, 0.6 Hz, 1H, Ar-H), 7.20 (d, J = 8.2 Hz, 2H, Ar-H), 7.11 (d, J = 8.2 Hz, 2H, Ar-H), 6.56 (s, 2H, NH2), 4.35 (br s, 4H, NCH2), 4.10 (br s, 4H, NCH2), 2.28 (s, 3H, ArCH3) ppm.
- To a suspension of 4-(5-amino-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-7-yl)-N-p-tolylpiperazine-1-carboxamide (90 mg, 0.2 mmol) in DMF (5 ml) was added NaH (60%, 12 mg, 0.31 mmol). The resulting mixture was stirred at room temperature for 10 minutes. Then, methyl iodide (0.3 mmol) was added to the mixture. After stirring at room temperature for 30 minutes, the solvents were evaporated in vacuo and the crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/30), yielding the pure title compound as yellowish solid (40 mg, 43%).
- MS m/z (%): 461 ([M+H]+, 100) 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.07 (dd, J = 2.2, 0.6 Hz, 1H, Ar-H), 8.65 (dd, J = 4.8, 1.6 Hz, 1 H, Ar-H), 8.23 (ddd, J = 8.0, 2.2, 1.6 Hz, 1 H, Ar-H), 7.53 (ddd, J = 8.0, 4.8, 0.6 Hz, 1H, Ar-H), 7.17 (d, J=8.2 Hz, 2H, Ar-H), 7.06 (d, J = 8.2 Hz, 2H, Ar-H), 6.49 (s, 2H, NH2), 4.08 (br s, 4H, NCH2), 3.28 (br.s, 4H, NCH2), 3.10 (s, 3H, CONCH3), 2.26 (s, 3H, ArCH3) ppm.
- To a suspension of 7-(piperazin-1-yl)-2-(pyridin-3-yl)thiazolo[5,4-d]pyrimidin-5-amine (63 mg, 0.2 mmol) in dioxane (10 ml) was added diisopropylethylamine (33 µl, 0.2 mmol) and p-tolyl chloroformate (0.2 mmol). The resulting mixture was stirred at room temperature for 30 minutes. The mixture was evaporated in vacuo and the crude residue was purified by flash chromatography on silica, the mobile phase being a mixture of methanol and dichloromethane (in a ratio of 1/30), yielding the pure title compound as yellowish solid (60 mg, 67%).
- MS m/z (%): 448 ([M+H]+, 100)
- 1H NMR (500 MHz, DMSO-d6, 25 °C): δ = 9.14 (dd, J = 2.2, 0.6 Hz, 1H, Ar-H), 8.65 (dd, J = 4.8, 1.6 Hz, 1 H, Ar-H), 8.30 (ddd, J = 8.0, 2.2, 1.6 Hz, 1 H, Ar-H), 7.53 (ddd, J = 8.0, 4.8, 0.6 Hz, 1 H, Ar-H), 7.19 (d, J = 8.2 Hz, 2H, Ar-H), 7.04 (d, J = 8.2 Hz, 2H, Ar-H), 6.57 (s, 2H, NH2), 4.35 (br s, 4H, NCH2), 3.77-3.62 (br s, 4H, NCH2), 2.30 (s, 3H, ArCH3) ppm.
-
Example X R2 R1 R3 28 S 4-fluorophenyl NH2 29 S 4-fluorophenyl NH2 30 S 4-fluorophenyl NH2 31 S 4-fluorobenzyl NH2 32 S 4-fluorophenyl NH2 33 S 4-fluorophenyl NH2 34 S 4-fluorobenzyl NH2 35 S 4-fluorophenyl NH2 36 S 4-fluorophenyl NH2 37 S 4-fluorophenyl NH2 38 S 4-fluorophenyl NH2 39 S 4-fluorophenyl NH2 40 S 4-fluorophenyl NH2 41 S 4-fluorophenyl NH2 42 S 4-fluorophenyl NH2 43 S 4-fluorophenyl NH2 44 S 4-fluorobenzyl NH2 45 S 4-fluorophenyl NH2 46 S 4-fluorobenzyl NH2 47 S 4-fluorophenyl CH3 48 S NH2 49 S NH2 50 S NH2 51 S NH2 52 S NH2 53 S NH2 54 S NH2 55 S NH2 66 S NH2 67 S NH2 68 S NH2 114 S NH2 115 S SHC3 NH2 116 S NH2 117 S NH2 118 S NH2 119 S NH2 120 S NH2 121a S NH2 121b S NH2 122 S NH2 123 S NH2 124 S NH2 125 S NH2 126 S NH2 127 S NH2 128 S 4-fluorophenyl NH2 129 S 4-fluorophenyl NH2 130 S 4-fluorophenyl NH2 131 S NH2 132 S NH2 133 S NH2 134 S NH2 135 S NH2 136 S NH2 137 S NH2 138 S NH2 139 S NH2 140 S NH2 141 S NH2 142 S NH2 143 S NH2 144 S NH2 145 S NH2 146 S NH2 147 S NH2 148 S NH2 149 S NH2 150 S NH2 151 S NH2 154 S NH2 155 S NH2 156 S NH2 157 S NH2 167 O 4-fluorophenyl H 168 O 4-fluorophenyl CH3 169 O 4-fluorophenyl NH2 170 O 4-fluorobenzyl H 171 O 4-fluorobenzyl CH3 172 O NH2 173 O 4-fluorophenyl H 174 O 4-fluorophenyl NH2 185 O NH2 186 O NH2 187 O NH2 188 O NH2 189 O NH2 190 O 4-fluorophenyl NH2 191 O 4-fluorophenyl NH2 192 O 4-fluorophenyl NH2 193 O NH2 194 O NH2 195 O NH2 196 O NH2 197 O NH2 198 O NH2 208 S phenyl NH2 209 S NH2 210 S NH2 211 S 4-fluorophenyl NH2 212 S 4-fluorophenyl NH2 213 S 4-fluorophenyl NH2 214 S 4-fluorophenyl NH2 215 S phenyl NH2 216 S 4-fluorophenyl NH2 217 S 4-fluorophenyl NH2 218 S 4-fluorophenyl NH2 219 S 4-fluorophenyl NH2 220 S phenyl NH2 222 S NH2 223 S NH2 224 S NH2 225 S NH2 226 S NH2 227 S NH2 228 S NH2 229 S NH2 230 S 4-fluorobenzyl NH2 231 S 4-fluorobenzyl NH2 232 S 4-fluorobenzyl NH2 233 S 4-fluorophenyl NH2 234 S 4-fluorophenyl NH2 235 S 4-fluorophenyl NH2 236 S 4-fluorophenyl NH2 237 S 4-fluorophenyl NH2 253 S NH2 254 S NH2 255 S NH2 256 S NH2 257 S NH2 258 S NH2 259 S NH2 260 S NH2 270 S 4-chlorophenyl NH2 271 S NH2 272 S NH2 273 S NH2 274 S NH2 275 S NH2 276 S NH2 277 S NH2 278 S NH2 279 S NH2 280 S NH2 281 S NH2 282 S NH2 290 S NH2 291 S NH2 292 S NH2 293 S NH2 294 S NH2 295 S NH2 296 S NH2 297 S NH2 298 S NH2 299 S NH2 300 S 4-fluorophenyl H 301 S 4-fluorophenyl H 302 S H 303 S H 439 S NH2 440 S NH2 441 S NH2 442 S NH2 443 S NH2 444 S NH2 445 S NH2 446 S NH2 - Thieno[2,3-d]pyrimidines
Example R5 R4 R6 312 4-fluorophenyl H 313 4-fluorophenyl n-butyl 314 4-fluorophenyl H 315 4-fluorophenyl n-butyl 325 4-fluorophenyl CH3 326 4-fluorophenyl NH2 327 4-fluorophenyl NH2 329 phenyl NH2 330 phenyl NH2 331 phenyl NH2 332 phenyl NH2 334 phenyl NH2 335 phenyl NH2 336 phenyl NH2 337 phenyl NH2 338 4-fluorophenyl NH2 339 4-fluorophenyl NH2 340 4-fluorophenyl NH2 341 4-fluorophenyl NH2 343 4-fluorophenyl NH2 344 4-fluorophenyl NH2 345 4-fluorophenyl NH2 346 4-fluorophenyl NH2 347 4-fluorophenyl NH2 348 4-fluorophenyl NH2 351 H NH2 352 4-fluorophenyl phenyl 353 4-fluorophenyl 354 4-fluorophenyl 355 4-fluorophenyl 356 4-fluorophenyl 357 4-fluorophenyl COOH 358 4-fluorophenyl 359 4-fluorophenyl NH2 360 4-fluorophenyl phenyl 361 4-fluorophenyl COOEt 362 4-fluorophenyl CONH2 363 4-fluorophenyl NH2 ethoxy 364 4-fluorophenyl NH2 367 4-fluorophenyl CH3 368 4-fluorophenyl COOEt 369 phenyl NH2 370 phenyl NH2 371 phenyl NH2 372 phenyl NH2 373 phenyl NH2 -
Example R9 R11 R6 R12 389 H NH2 4-fluorophenyl 390 H NH2 3,4-dimethoxyphenyl 391 H NH2 4-bromophenyl 392 H NH2 4-chlorophenyl 393 H NH2 3-chlorophenyl 394 H NH2 4-trifluoromethylphenyl 395 H NH2 4-trifluoromethoxyphenyl 396 H NH2 4-methylphenyl 397 H NH2 propyl 398 H NH2 cyclopropyl 399 H NH2 tert-butyl 400 H NH2 methyl 401 H NH2 4-fluorophenyl 402 H NH2 4-fluorophenyl 403 H NH2 4-fluorophenyl 404 H NH2 4-fluorophenyl 405 H NH2 4-fluorophenyl 406 H NH2 4-fluorophenyl 408 H NH2 4-fluorophenyl 409 H NH2 4-fluorophenyl 410 H NH2 4-fluorophenyl 411 H NH2 4-fluorophenyl 412 H NH2 4-fluorophenyl 413 H NH2 4-fluorophenyl 414 H NH2 4-fluorophenyl 415 H NH2 4-fluorophenyl 416 H NH2 4-fluorophenyl 417 H NH2 4-fluorophenyl 418 H NH2 4-fluorophenyl 419 H NH2 4-fluorophenyl 421 H NH2 4-fluorophenyl 422 H NH2 4-fluorophenyl 423 H NH2 SH 424 H NH2 SCH3 425 H NH2 SC3H7 426 H NH2 SCH2C6H5 427 H NH2 SCH2CH2C6H5 428 CH3 NH2 SCH3 429 H NH2 430 CH3 NH2 4-fluorophenyl 431 benzyl NH2 4-fluorophenyl 438 H NH2 4-fluorophenyl - Human peripheral blood mononuclear cells (PBMCs) were isolated from buffy coats, obtained from healthy blood donors by Ficoll (Lymphoprep, Axis-Shield PoC AS, Oslo, Norway) density-gradient centrifugation. The cells at the Ficoll-plasma interface were washed three times and used as "Responder" cells. RPMI 1788 (ATCC, N° CCL-156) cells were treated with mitomycin C (Kyowa, Nycomed, Brussel, Belgium) and used as "Stimulator" cells. Responder cells (0.12x106), Stimulator cells (0.045x106) and compounds (in different concentrations) were cocultured for 6 days in RPMI 1640 medium (BioWhittaker, Lonza, Belgium) supplemented with 10% fetal calf serum, 100U/ml Geneticin (Gibco, LifeTechnologies, UK). Cells were cultured in triplicate in flat-bottomed 96-well microtiter tissue culture plates (TTP, Switzerland). After 5 days, cells were pulsed with 1 µCi of methyl-3H thymidine (MP Biomedicals, USA), harvested 18h later on glass filter paper and counted. Proliferation values were expressed as counts per minute (cpm), and converted to % inhibition with respect to a blank MLR test (identical but without added compound). The IC50 was determined from a graph with at least four points, each derived from the mean of 2 experiments. The IC50 value represents the lowest concentration of the thiazolo[5,4-d]pyrimidine, oxazolo[5,4-d]pyrimidine, thieno[2,3-d]pyrimidine or purine derivative (expressed in µM) that resulted in a 50% inhibition of the MLR. The results are shown in Table 1 for a number of compounds, using the following symbols :
Table 1 : MLR data of selected examples Example IC50 (µM) Example IC50 (µM) Example IC50 (µM) 28 >10 191 >10 312 6.35 29 >10 192 >10 313 1.33 30 >10 193 0.7 325 3.39 31 9.7 194 9.04 326 0.7 32 >10 195 >10 327 6.8 33 >10 196 3.97 329 0.33 34 >10 197 1.95 330 0.93 35 >10 198 >10 331 6.7 36 >10 208 0.042 332 0.78 37 >10 209 0.055 334 8.83 38 4.8 210 0.072 335 >10 39 >10 211 0.052 337 9.64 40 4.9 212 0.375 341 7.23 42 0.7 213 0.219 343 6.99 43 0.3 214 0.121 344 >10 44 0.4 215 0.116 345 >10 45 0.3 216 0.019 346 >10 46 0.7 217 0.345 347 8.2 48 3.54 218 0.177 348 8.27 49 4.4 219 0.323 351 1.23 52 >10 220 0.17 352 >10 66 4.38 223 >10 353 >10 101 0.4 224 1.1 355 >10 114 1.56 225 >10 357 >10 115 >10 226 7.12 359 2.8 116 5.29 227 0.61 369 0.072 117 2.51 228 1.72 370 0.066 118 0.86 229 2.66 371 0.21 119 0.6 230 0.27 372 0.32 120 0.83 231 0.55 373 6.92 121a 0.05 232 0.86 389 0.81 121b 0.83 233 0.074 390 0.53 122 >10 234 0.037 391 0.26 123 0.26 235 0.46 392 0.54 124 0.12 236 0.89 393 0.66 126 2.65 237 >10 394 0.63 127 6.77 253 0.71 395 >10 128 0.38 254 1.22 396 0.47 129 0.29 255 0.71 397 0.94 130 0.35 256 0.74 398 0.43 131 3.25 257 1.14 399 6.31 132 0.44 258 0.93 400 0.45 133 >10 259 0.96 401 1.09 134 4.92 260 0.82 402 >10 135 >10 270 0.15 403 7.08 136 >10 271 0.012 404 >10 137 6.68 272 0.012 405 >10 138 2.1 273 0.045 406 6.4 139 3.29 274 >10 408 >10 140 8.82 275 0.003 409 7.47 141 1.41 276 0.001 410 >10 143 >10 277 0.024 411 >10 144 5.13 278 0.08 412 8.61 145 4.35 279 0.1 413 >10 147 4.52 280 >10 414 3.87 148 2.61 281 0.041 415 4.16 149 5.76 282 0.229 416 5.76 150 5.39 290 0.96 417 >10 151 >10 291 1.11 418 9.86 154 0.79 292 0.92 419 >10 155 >10 293 0.93 421 3.71 156 >10 294 0.75 422 1.18 157 0.63 295 0.35 424 0.61 169 3 296 7.59 425 1.02 172 0.8 297 0.69 426 3.28 185 1.76 298 1.19 427 >10 186 1.83 299 3.11 428 0.3 187 >10 300 5.53 429 0.51 188 4.43 301 0.67 430 0.36 189 5.53 303 5.59 431 >10 438 2.58 - The in vivo efficacy of the compound of example 121a was studied in a mouse model of cardiac allograft transplantation. Drug vehicle (n = 6) or compound 121a (n = 4) was given by oral gavage daily, beginning one day prior to transplantation, until day 30 post transplantation. Cyclosporine A, the major immunosuppressive drug used in organ transplantation, was used as a reference and was also administered by daily gavage (n = 4). Animals treated with vehicle alone rejected their allograft within 6-9 days post transplantation. CsA at the given dose achieved all 4 grafts survival as long as the treatment continuing. However, rejection occurred to all 4 grafts within 2 weeks after withdrawal of the treatment. Oral administration of compound 121a (at a dose of 40 mg/kg) resulted in continuous graft survival in 3 out of 4 grafts. The grafts continued beating after withdrawal of the treatment (up to 60 days), indicating the induction of certain type of graft tolerance. The data are shown in Table 2. These data indicate that compound 121a can suppress a robust in vivo allogeneic response.
Table 2 Compd Dosea (mg/kg/d) Surviving days p valueb Vehicle 6,7,8,8,9,9 CsA 40 39,40,42,43 <0.001 121a 40 17, >60, >60, >60 <0.03 a by daily gavage.
b student t test: vs. vehicle. - Inbreed C57BL/6 H-2b and Balb/c H-2d female mice, 8-10 weeks old, 20-25g, were used as donor and recipient, respectively. Heterotopic heart transplantation was performed by implanting the donor heart on the neck of recipients using conventional microsurgery techniques. Graft beating was checked daily by inspection and palpation. Cessation of beating indicated graft rejection, which was confirmed by histological examination. Housing and all experimental animal procedures were approved by the Institutional Animal Care and Research Advisory Committee of the KU Leuven.
- Animals were randomly divided into 3 groups: (i) Vehicle group: vehicle ((30% 2-hydroxypropyl-β-cyclodextrin) only by daily gavage, n = 6 ; (ii) Reference drug group: CsA 40 mg per kg by daily gavage, n = 4 ; (iii) compound 121a group: 40 mg per kg by daily gavage, n = 4. Treatment started one day prior to transplantation (day -1) until day 30 post transplantation.
- The in vivo efficacy of the compound of example 222 was studied in a mouse model of cardiac allograft transplantation as described in Example 448. Animal treated with vehicle rejected the graft within 6-8 days post-transplantation. CsA at 40 mg/kg per day prevented rejection as long as treatment continued in 3 of the 4 grafts. However, all grafts were rejected within 20 days after withdrawal of the treatment on day 30 post-transplantation. 222 at a dose of 20 mg/kg per day slightly prolonged graft survival up to 13 days. 222 at 40 mg/kg per day resulted continuous graft survival of 4/6 grafts. The grafts (3/4 cases) continuously functioned after stopping the treatment on day30, suggesting a kind of immune tolerance. The results are shown in Table 3. These results supported that the compound of example 222 effectively suppressed allograft rejection in a dose dependent manner.
Table 3 Compound Dose# (mg/kg/d) Surviving days p value§ Vehicle 6, 7, 7, 8 CsA 40 10, 40, 41, 50 (n=3) <0,01 Example 222 20 6, 7, 8, 9, 11, 13 >0,05 Example 222 40 9, 11, 50, >60 (n=3) <0,01 # by daily gavage.
§ student t test: vs. vehicle. - Materials and method are identical to the ones mentioned in Example 448.
Claims (12)
- A compound having the general formula III or IV:
wherein acyl group refers to a carbonyl group adjacent to a C1-7 alkyl, a C3-10 cycloalkyl, an aryl, an arylalkyl or a heterocyclic group, or is selected from the group comprising alkanoyl, cycloalkanoyl, cycloalkyl-alkanoyl, alkenoyl, alkylthioalkanoyl, alkanesulfonyl, alkoxycarbonyl, alkylcarbamoyl, alkylcarbamidoyl, alkoxalyl, aroyl, aralkanoyl, aralkenoyl, aryloxyalkanoyl, arylthioalkanoyl, arylaminoalkanoyl, aryloxycarbonyl, aralkoxycarbonyl, arylcarbamoyl, arylglyoxyloyl, arylthiocarbamoyl, arylcarbamidoyl, heterocyclic-carbonyl, heterocyclic-alkanoyl, wherein said heterocyclic group is an aromatic or non-aromatic 5- to 7-membered heterocyclic ring with one or more heteroatoms selected from the group consisting of nitrogen, oxygen and sulfur in said ring;
and/or a pharmaceutical acceptable addition salt thereof and/or a stereoisomer thereof and/or a solvate thereof. - The compound according to claim 1, wherein R11 is selected from the group consisting of amino, acetamido, N-protected amino, (mono- or di) C1-7 alkylamino, (mono- or di) arylamino, (mono- or di) C3-10 cycloalkylamino, (mono- or di) hydroxy C1-7 alkylamino, (mono- or di) C1-4 alkyl-arylamino.
- The compound according to claim 1 or 2, wherein n is 0 and R10 is selected from the group consisting of formyl, acyl, thioacyl, amide, thioamide, sulfonyl, sulfinyl, carboxylate, thiocarboxylate, amino-substituted acyl, alkoxyalkyl, C3-10 cycloalkyl-alkyl, C3-10 cycloalkyl, dialkylaminoalkyl, heterocyclic-substituted alkyl, acyl-substituted alkyl, thioacyl-substituted alkyl, amido-substituted alkyl, thioamido-substituted alkyl, carboxylato-substituted alkyl, thiocarboxylato-substituted alkyl, (amino-substituted acyl)alkyl, heterocyclic, carboxylic acid ester, ω-carboxylic ester-alkyl, aryloxyalkyl, arylalkyl and aryl; wherein the aryl moiety of each of said aryloxyalkyl, arylalkyl and aryl radicals is optionally substituted with one or more substituents independently selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, aryloxy, arylalkyloxy, formyl, carbamoyl, thiocarbamoyl, sulfonamido, hydroxylamino, alkoxy-amino, mercaptoamino, thioalkylamino, cyano, alkylamino, cycloalkylamino.
- The compound according to any of claims 1, 2 or 3, wherein R8 is selected from the group consisting of heteroaryl and aryl groups; C1-7 alkyl; halo C1-7 alkyl; C1-7 alkoxy; C3-10 cycloalkoxy; aryloxy; arylalkyloxy; thio C1-7 alkyl; thio C3-10 cycloalkyl; thioaryl; thioheterocyclic; arylalkylthio; heterocyclic-substituted alkylthio; aromatic or heterocyclic substituents substituted with an aliphatic spacer between the purine ring and the aromatic or heterocyclic substituent; wherein said heteroaryl or aryl groups are optionally substituted with one or more substituents selected from the group consisting of halogen, C1-7 alkyl, halo C1-7 alkyl, nitro, hydroxyl, sulfhydryl, amino, C1-7 alkoxy, C3-10 cycloalkoxy, thio C1-7 alkyl, and wherein said aliphatic spacer is a branched or straight, saturated or unsaturated aliphatic chain of 1 to 7 carbon atoms optionally containing one or more functions, atoms or radicals independently selected from the group consisting of halogen, carbonyl, hydroxyl, thiol, cyano, nitro, thio C1-7 alkyl, thio C3-10 cycloalkyl.
- The compound according to claims 1 or 2, wherein R11 is amino.
- A compound selected from the group consisting of: 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(3,4-dimethoxyphenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-bromophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-chlorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(3-chlorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-(trifluoromethyl)phenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-(trifluoromethoxy)phenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-p-tolyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-propyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-cyclopropyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-tert-butyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-methyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-phenoxyethanone; 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(3-methoxyphenyl)methanone; 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(thiophen-2-yl)ethanone; (4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(4-chlorophenyl)methanone; 6-(4-(benzylsulfonyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine; (4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)(naphthalen-1-yl)methanone; 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)ethanone; 8-(4-fluorophenyl)-6-(4-(thiazol-2-yl)piperazin-1-yl)-9H-purin-2-amine; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-(pyrrolidin-1-yl)ethanone; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-1-morpholinoethanone; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-3-yl)acetamide; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-methyl-N-phenylacetamide; 6-(4-(4-chlorophenyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine; 8-(4-fluorophenyl)-6-(4-(4-fluorophenyl)piperazin-1-yl)-9H-purin-2-amine; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(pyridin-2-yl)acetamide; 2-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-N-(thiazol-2-yl)acetamide; 6-(4-(4-fluorobenzyl)piperazin-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine; 8-(4-fluorophenyl)-6-(4-(pyridin-4-yl)piperazin-1-yl)-9H-purin-2-amine; 6-(1,4-diazepan-1-yl)-8-(4-fluorophenyl)-9H-purin-2-amine; 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)-1,4-diazepan-1-yl)-2-(4-chlorophenoxy)ethanone; 4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)-N-m-tolyl-1,4-diazepane-1-carboxamide; 1-(4-(2-amino-8-thioxo-8,9-dihydro-7H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(methylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(propylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(benzylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(phenethylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-9-methyl-8-(methylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(cyclopentylthio)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-8-(4-fluorophenyl)-9-methyl-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 1-(4-(2-amino-9-benzyl-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-2-(4-chlorophenoxy)ethanone; 2-amino-6-(piperazin-1-yl)-8-(4-fluorophenyl)-9H-purine; and 1-(4-(2-amino-8-(4-fluorophenyl)-9H-purin-6-yl)piperazin-1-yl)-3-phenylpropan-1-one.
- A compound according to any of claims 1 to 6 for use as a medicine.
- A compound according to any of claims 1 to 6 for use as a medicine for the prevention or treatment of immune disorders in a mammal.
- A compound for use according to claim 8, wherein said immune disorder is an autoimmune disorder or an immune disorder as a result from an organ or cells transplantation.
- A compound for use according to claim 8 or 9, wherein said mammal is a human being.
- A pharmaceutical composition comprising a therapeutically effective amount of a compound according to any of claims 1 to 6 and one or more pharmaceutically acceptable excipients.
- The pharmaceutical composition according to claim 11, further comprising one or more biologically active drugs being selected from the group consisting of immunosuppressant and/or immunomodulator drugs, and antineoplastic drugs.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0904353A GB0904353D0 (en) | 2009-03-13 | 2009-03-13 | Novel thiazolopyrimidines |
| GB0904373A GB0904373D0 (en) | 2009-03-13 | 2009-03-13 | Novel thienopyrimidines |
| GB0911022A GB0911022D0 (en) | 2009-06-26 | 2009-06-26 | Novel purines |
| EP20100711871 EP2405973B1 (en) | 2009-03-13 | 2010-03-15 | Thiazolopyrimidine modulators as immunosuppressive agents |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20100711871 Division EP2405973B1 (en) | 2009-03-13 | 2010-03-15 | Thiazolopyrimidine modulators as immunosuppressive agents |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2923734A1 true EP2923734A1 (en) | 2015-09-30 |
| EP2923734B1 EP2923734B1 (en) | 2018-01-10 |
Family
ID=42181129
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20100711871 Active EP2405973B1 (en) | 2009-03-13 | 2010-03-15 | Thiazolopyrimidine modulators as immunosuppressive agents |
| EP15162142.2A Not-in-force EP2923734B1 (en) | 2009-03-13 | 2010-03-15 | Purine analogues and their use as immunosuppressive agents |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20100711871 Active EP2405973B1 (en) | 2009-03-13 | 2010-03-15 | Thiazolopyrimidine modulators as immunosuppressive agents |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US8901132B2 (en) |
| EP (2) | EP2405973B1 (en) |
| JP (2) | JP5647998B2 (en) |
| CA (2) | CA2754890C (en) |
| ES (2) | ES2665277T3 (en) |
| WO (1) | WO2010103130A2 (en) |
Families Citing this family (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DK2035369T3 (en) | 2006-07-05 | 2014-10-06 | Fibrotech Therapeutics Pty Ltd | THERAPEUTIC |
| ES2665277T3 (en) * | 2009-03-13 | 2018-04-25 | Katholieke Universiteit Leuven K.U. Leuven R&D | Purine analogues and their use as immunosuppressive agents |
| EP2491030B1 (en) | 2009-10-22 | 2015-07-15 | Fibrotech Therapeutics PTY LTD | Fused ring analogues of anti-fibrotic agents |
| GB201012889D0 (en) | 2010-08-02 | 2010-09-15 | Univ Leuven Kath | Antiviral activity of novel bicyclic heterocycles |
| GB201015411D0 (en) * | 2010-09-15 | 2010-10-27 | Univ Leuven Kath | Anti-cancer activity of novel bicyclic heterocycles |
| GB201114212D0 (en) | 2011-08-18 | 2011-10-05 | Ucb Pharma Sa | Therapeutic agents |
| GB201115665D0 (en) | 2011-09-09 | 2011-10-26 | Univ Leuven Kath | Autoimmune and inflammatory disorder therapy |
| US20130102601A1 (en) | 2011-10-21 | 2013-04-25 | F. Hoffmann-La Roche Ltd | Pyrimidin-4-one derivatives and their use in the treatment, amelioration or prevention of a viral disease |
| GB201119401D0 (en) | 2011-11-10 | 2011-12-21 | Ucb Pharma Sa | Therapeutic agents |
| GB201217704D0 (en) | 2012-10-03 | 2012-11-14 | Ucb Pharma Sa | Therapeutic agents |
| SG11201504291SA (en) * | 2012-12-20 | 2015-07-30 | Ucb Biopharma Sprl | Therapeutically active pyrazolo-pyrimidine derivatives |
| CN103044460B (en) * | 2013-01-30 | 2015-02-25 | 石家庄学院 | 3,5,7-triphenyl-5H-thiazolo[3,2-a]pyrimidin derivatives and application thereof |
| MA38455B1 (en) * | 2013-04-05 | 2018-05-31 | Polichem Sa | Use of pidotimodes to treat inflammatory bowel disease |
| BR112016006319A2 (en) | 2013-09-27 | 2017-08-01 | Nimbus Iris Inc | irak inhibitors and uses thereof |
| TWI648281B (en) | 2013-10-17 | 2019-01-21 | 日商安斯泰來製藥股份有限公司 | Sulfur-containing bicyclic compound |
| CN104892589A (en) * | 2014-03-07 | 2015-09-09 | 中国科学院上海药物研究所 | Heterocyclic compound, preparation method therefor and use thereof |
| GB201410817D0 (en) | 2014-06-17 | 2014-07-30 | Ucb Biopharma Sprl And Katholieke Universiteit Leuven K U Leuven R & D | Therapeutic agents |
| US9822127B2 (en) * | 2014-07-23 | 2017-11-21 | The Board Of Trustees Of The Leland Stanford Junior University | GAK modulators as antivirals |
| UY36390A (en) | 2014-11-05 | 2016-06-01 | Flexus Biosciences Inc | MODULATING COMPOUNDS OF INDOLAMINE ENZYME 2,3-DIOXYGENASE (IDO), ITS SYNTHESIS METHODS AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| MX2017005462A (en) | 2014-11-05 | 2017-07-28 | Flexus Biosciences Inc | Immunoregulatory agents. |
| GB201517263D0 (en) | 2015-09-30 | 2015-11-11 | Ucb Biopharma Sprl And Katholieke Universiteit Leuven | Therapeutic agents |
| GB201521767D0 (en) | 2015-12-10 | 2016-01-27 | Ucb Biopharma Sprl And Katholieke Universiteit Leuven K U Leuven R&D | Therapeutic agents |
| CA3052036A1 (en) | 2017-02-03 | 2018-08-09 | Certa Therapeutics Pty Ltd. | Anti-fibrotic compounds |
| SG11202012616RA (en) | 2018-08-21 | 2021-02-25 | Kyorin Pharmaceutical Co Ltd | Bicyclic heteroaromatic ring derivative |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0300726A1 (en) * | 1987-07-20 | 1989-01-25 | Merck & Co. Inc. | Piperazinyl derivates of purines and isosteres thereof as hypoglycemic agents |
| US5057517A (en) * | 1987-07-20 | 1991-10-15 | Merck & Co., Inc. | Piperazinyl derivatives of purines and isosteres thereof as hypoglycemic agents |
| WO2004035740A2 (en) * | 2002-10-15 | 2004-04-29 | Synta Pharmaceuticals Corp. | Aromatic bicyclic heterocyles to modulate 1l - 12 production |
| WO2009027732A1 (en) * | 2007-08-24 | 2009-03-05 | Astrazeneca Ab | 5-6-bicyclic heteroaromatic compounds with antibacterial activity |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2512572A (en) | 1950-06-20 | Substituted pteridines and method | ||
| BE791242A (en) * | 1971-11-11 | 1973-05-10 | Thomae Gmbh Dr K | NEWS THIAZOLO (5,4-D) PYRIMIDINES |
| US4127118B1 (en) | 1977-03-16 | 1995-12-19 | Alvaro Latorre | Method of effecting and enhancing an erection |
| ZA857647B (en) * | 1984-10-12 | 1986-05-28 | Warner Lambert Co | Purine derivatives |
| GB9221220D0 (en) | 1992-10-09 | 1992-11-25 | Sandoz Ag | Organic componds |
| US5780462A (en) | 1995-12-27 | 1998-07-14 | American Home Products Corporation | Water soluble rapamycin esters |
| JP2001097979A (en) * | 1999-07-28 | 2001-04-10 | Takeda Chem Ind Ltd | Condensed heterocyclic compound, and method of production and application thereof |
| US6331547B1 (en) | 1999-08-18 | 2001-12-18 | American Home Products Corporation | Water soluble SDZ RAD esters |
| DE60101372T2 (en) | 2000-02-11 | 2004-10-14 | Astrazeneca Ab | PYRIMIDINE COMPOUNDS AND THEIR USE AS MODULATORS OF CHEMOKIN RECEPTOR ACTIVITY |
| US6440991B1 (en) | 2000-10-02 | 2002-08-27 | Wyeth | Ethers of 7-desmethlrapamycin |
| JP2004028376A (en) | 2002-06-21 | 2004-01-29 | Hino Motors Ltd | EGR cooler |
| JP4279784B2 (en) * | 2002-12-04 | 2009-06-17 | エーザイ・アール・アンド・ディー・マネジメント株式会社 | 1,3-dihydro-imidazole fused ring compound |
| ES2217956B1 (en) * | 2003-01-23 | 2006-04-01 | Almirall Prodesfarma, S.A. | NEW DERIVATIVES OF 4-AMINOTIENE (2,3-D) PIRIMIDIN-6-CARBONITRILE. |
| PL1641803T6 (en) | 2003-06-11 | 2011-04-29 | Xention Ltd | Thienopyrimidine derivatives as potassium channel inhibitors |
| DE10348023A1 (en) * | 2003-10-15 | 2005-05-19 | Imtm Gmbh | New alanyl aminopeptidase inhibitors for the functional manipulation of different cells and for the treatment of immunological, inflammatory, neuronal and other diseases |
| US20080269238A1 (en) | 2004-04-01 | 2008-10-30 | Takeda Pharmaceutical Company Limited | Thiazolopyrimidine Derivative |
| GB0407723D0 (en) * | 2004-04-05 | 2004-05-12 | Novartis Ag | Organic compounds |
| US7576080B2 (en) | 2004-12-23 | 2009-08-18 | Memory Pharmaceuticals Corporation | Certain thienopyrimidine derivatives as phosphodiesterase 10 inhibitors |
| FR2880626B1 (en) * | 2005-01-13 | 2008-04-18 | Aventis Pharma Sa | DERIVATIVES OF PURINE, COMPOSITIONS CONTAINING SAME AND USE THEREOF |
| CA2595882A1 (en) * | 2005-01-26 | 2006-08-03 | Pharmacia & Upjohn Company Llc | Thieno [2,3-d] pyrimidine compounds as inhibitors of adp-mediated platelets aggregation |
| US20090149452A1 (en) | 2005-07-01 | 2009-06-11 | Nippon Chemiphar Co., Ltd. | Xanthine oxidase inhibitor |
| GB0526246D0 (en) * | 2005-12-22 | 2006-02-01 | Novartis Ag | Organic compounds |
| WO2007084815A2 (en) | 2006-01-19 | 2007-07-26 | Janssen Pharmaceutica, N.V. | Substituted thienopyrimidine kinase inhibitors |
| KR100846988B1 (en) | 2006-03-06 | 2008-07-16 | 제일약품주식회사 | Novel thienopyrimidine derivatives or pharmaceutically acceptable salts thereof, process for the preparation thereof and pharmaceutical composition comprising the same |
| CN101511840A (en) * | 2006-04-26 | 2009-08-19 | 吉宁特有限公司 | Phosphoinositide 3-kinase inhibitor compounds and methods of use thereof |
| JP5026511B2 (en) | 2006-05-18 | 2012-09-12 | エフ.ホフマン−ラ ロシュ アーゲー | Thiazolo-pyramidine / pyridine urea derivatives as adenosine A2B receptor antagonists |
| WO2008005303A2 (en) * | 2006-06-30 | 2008-01-10 | Janssen Pharmaceutica N.V. | Thiazolopyrimidine modulators of trpv1 |
| ZA200900670B (en) | 2006-08-08 | 2010-04-28 | Millennium Pharm Inc | Heteroaryl compounds useful as inhititors of E1 activating enzymes |
| WO2008057402A2 (en) | 2006-11-02 | 2008-05-15 | Cytovia, Inc. | N-aryl-isoxazolopyrimidin-4-amines and related compounds as activators of caspases and inducers of apoptosis and the use thereof |
| JP2010510211A (en) | 2006-11-16 | 2010-04-02 | ファーマコペイア・リミテッド・ライアビリティ・カンパニー | 7-substituted purine derivatives for immunosuppression |
| WO2008059368A2 (en) * | 2006-11-17 | 2008-05-22 | Pfizer Products Inc. | Fused 2-amino pyrimidine compounds and their use for the treatment of cancer |
| ES2571028T3 (en) * | 2006-12-07 | 2016-05-23 | Genentech Inc | Phosphoinositide 3-kinase inhibitor compounds and methods of use |
| JP2010526027A (en) | 2007-01-23 | 2010-07-29 | パラウ・フアルマ・ソシエダツド・アノニマ | Purine derivatives |
| CN101622001A (en) | 2007-01-26 | 2010-01-06 | Irm责任有限公司 | Purine compounds and compositions as kinase inhibitors for the treatment of malaria parasite-associated diseases |
| WO2008135232A1 (en) | 2007-05-02 | 2008-11-13 | Riccardo Cortese | Use and compositions of purine derivatives for the treatment of proliferative disorders |
| US7893060B2 (en) * | 2007-06-12 | 2011-02-22 | F. Hoffmann-La Roche Ag | Thiazolopyrimidines and their use as inhibitors of phosphatidylinositol-3 kinase |
| CA2691444C (en) | 2007-06-29 | 2016-06-14 | Gilead Sciences, Inc. | Purine derivatives and their use as modulators of toll-like receptor 7 |
| WO2009013545A2 (en) * | 2007-07-26 | 2009-01-29 | Astrazeneca Ab | Chemical compounds |
| WO2009042607A1 (en) * | 2007-09-24 | 2009-04-02 | Genentech, Inc. | Thiazolopyrimidine p13k inhibitor compounds and methods of use |
| UA104010C2 (en) * | 2008-12-18 | 2013-12-25 | Эли Лилли Энд Компани | Purine compounds |
| ES2665277T3 (en) * | 2009-03-13 | 2018-04-25 | Katholieke Universiteit Leuven K.U. Leuven R&D | Purine analogues and their use as immunosuppressive agents |
| AR080711A1 (en) * | 2010-03-31 | 2012-05-02 | Lilly Co Eli | PIPERAZIN-PURINA COMPOSITE PHARMACEUTICAL COMPOSITION THAT INCLUDES IT AND ITS USE TO PREPARE A USEFUL MEDICINAL PRODUCT FOR THE TREATMENT OR PREVENTION OF PAIN |
-
2010
- 2010-03-15 ES ES15162142.2T patent/ES2665277T3/en active Active
- 2010-03-15 CA CA2754890A patent/CA2754890C/en not_active Expired - Fee Related
- 2010-03-15 WO PCT/EP2010/053318 patent/WO2010103130A2/en not_active Ceased
- 2010-03-15 US US13/255,429 patent/US8901132B2/en not_active Expired - Fee Related
- 2010-03-15 JP JP2011553476A patent/JP5647998B2/en not_active Expired - Fee Related
- 2010-03-15 EP EP20100711871 patent/EP2405973B1/en active Active
- 2010-03-15 CA CA2987743A patent/CA2987743A1/en not_active Abandoned
- 2010-03-15 ES ES10711871.3T patent/ES2543216T3/en active Active
- 2010-03-15 EP EP15162142.2A patent/EP2923734B1/en not_active Not-in-force
-
2014
- 2014-10-20 US US14/518,480 patent/US9221822B2/en not_active Expired - Fee Related
- 2014-11-10 JP JP2014228403A patent/JP6021880B2/en not_active Expired - Fee Related
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0300726A1 (en) * | 1987-07-20 | 1989-01-25 | Merck & Co. Inc. | Piperazinyl derivates of purines and isosteres thereof as hypoglycemic agents |
| US5057517A (en) * | 1987-07-20 | 1991-10-15 | Merck & Co., Inc. | Piperazinyl derivatives of purines and isosteres thereof as hypoglycemic agents |
| WO2004035740A2 (en) * | 2002-10-15 | 2004-04-29 | Synta Pharmaceuticals Corp. | Aromatic bicyclic heterocyles to modulate 1l - 12 production |
| WO2009027732A1 (en) * | 2007-08-24 | 2009-03-05 | Astrazeneca Ab | 5-6-bicyclic heteroaromatic compounds with antibacterial activity |
Non-Patent Citations (1)
| Title |
|---|
| BRILL W K-D ET AL: "The bromination of purines with a charge transfer complex between bromine and lutidine", TETRAHEDRON LETTERS, PERGAMON, GB, vol. 42, no. 36, 3 September 2001 (2001-09-03), pages 6279 - 6282, XP004302928, ISSN: 0040-4039, DOI: 10.1016/S0040-4039(01)01246-1 * |
Also Published As
| Publication number | Publication date |
|---|---|
| US8901132B2 (en) | 2014-12-02 |
| JP2015028088A (en) | 2015-02-12 |
| CA2754890C (en) | 2018-01-16 |
| WO2010103130A3 (en) | 2010-12-23 |
| JP5647998B2 (en) | 2015-01-07 |
| US9221822B2 (en) | 2015-12-29 |
| EP2405973A2 (en) | 2012-01-18 |
| EP2405973B1 (en) | 2015-04-22 |
| ES2543216T3 (en) | 2015-08-17 |
| US20150038494A1 (en) | 2015-02-05 |
| US20120046278A1 (en) | 2012-02-23 |
| ES2665277T3 (en) | 2018-04-25 |
| CA2754890A1 (en) | 2010-09-16 |
| WO2010103130A2 (en) | 2010-09-16 |
| JP6021880B2 (en) | 2016-11-09 |
| CA2987743A1 (en) | 2010-09-16 |
| JP2012520269A (en) | 2012-09-06 |
| EP2923734B1 (en) | 2018-01-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9221822B2 (en) | Thiazolopyrimidine modulators as immunosuppressive agents | |
| EP2616077B1 (en) | Anti-cancer activity of novel bicyclic heterocycles | |
| US9730944B2 (en) | Antiviral activity of novel bicyclic heterocycles | |
| US8232278B2 (en) | Pyrido(3,2-D)pyrimidines and pharmaceutical compositions useful for treating hepatitis C | |
| US20090036430A1 (en) | Pyrido(3,2-d)pyrimidines and pharmaceutical compositions useful for medical treatment | |
| US20080004285A1 (en) | Pyrido(3,2-d)pyrimidines and pharmaceutical compositions useful for medical treatment | |
| EP1479682A1 (en) | Immunosuppressive effects of pteridine derivatives | |
| US20090324543A1 (en) | PYRIDO(3,2-d)PYRIMIDINES USEFUL FOR TREATING VIRAL INFECTIONS | |
| EP3492468B1 (en) | Heterocyclic compound as jak inhibitor, and salts and therapeutic use thereof | |
| EP4338737A1 (en) | Parp7 inhibitor and use thereof | |
| JP2008526704A (en) | Pyrido (3,2-d) pyrimidine and pharmaceutical composition useful for medical treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 2405973 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: AL BA ME RS |
|
| 17P | Request for examination filed |
Effective date: 20160330 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| R17P | Request for examination filed (corrected) |
Effective date: 20160330 |
|
| 17Q | First examination report despatched |
Effective date: 20161216 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20170807 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 2405973 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP Ref country code: AT Ref legal event code: REF Ref document number: 961751 Country of ref document: AT Kind code of ref document: T Effective date: 20180115 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602010048076 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2665277 Country of ref document: ES Kind code of ref document: T3 Effective date: 20180425 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20180110 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 961751 Country of ref document: AT Kind code of ref document: T Effective date: 20180110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180410 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180411 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180510 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180410 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602010048076 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20180331 |
|
| 26N | No opposition filed |
Effective date: 20181011 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180315 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180315 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180331 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180315 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20180110 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180110 Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20100315 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20210210 Year of fee payment: 12 Ref country code: IT Payment date: 20210211 Year of fee payment: 12 Ref country code: CH Payment date: 20210317 Year of fee payment: 12 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20210308 Year of fee payment: 12 Ref country code: DE Payment date: 20210302 Year of fee payment: 12 Ref country code: TR Payment date: 20210312 Year of fee payment: 12 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20210405 Year of fee payment: 12 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602010048076 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20220315 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220331 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220315 Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220331 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20221001 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220331 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20230529 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220315 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220316 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220315 |