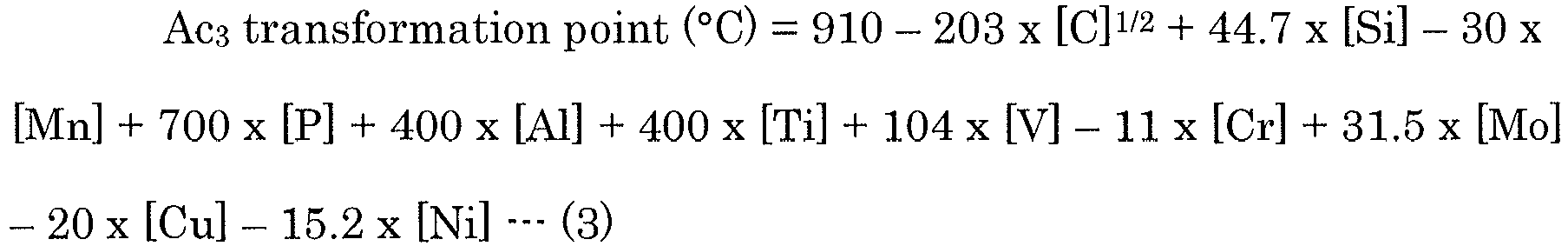EP2719787B1 - Hot press molded article, method for producing same, and thin steel sheet for hot press molding - Google Patents
Hot press molded article, method for producing same, and thin steel sheet for hot press molding Download PDFInfo
- Publication number
- EP2719787B1 EP2719787B1 EP12796568.9A EP12796568A EP2719787B1 EP 2719787 B1 EP2719787 B1 EP 2719787B1 EP 12796568 A EP12796568 A EP 12796568A EP 2719787 B1 EP2719787 B1 EP 2719787B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- steel sheet
- forming
- hot press
- temperature
- formed product
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Revoked
Links
- 229910000831 Steel Inorganic materials 0.000 title claims description 135
- 239000010959 steel Substances 0.000 title claims description 135
- 238000004519 manufacturing process Methods 0.000 title description 9
- 238000000465 moulding Methods 0.000 title 1
- 238000001816 cooling Methods 0.000 claims description 70
- 238000000034 method Methods 0.000 claims description 57
- 229910000859 α-Fe Inorganic materials 0.000 claims description 57
- 230000009466 transformation Effects 0.000 claims description 52
- 229910001566 austenite Inorganic materials 0.000 claims description 49
- 238000010438 heat treatment Methods 0.000 claims description 41
- 230000000717 retained effect Effects 0.000 claims description 41
- 229910000734 martensite Inorganic materials 0.000 claims description 22
- 230000009467 reduction Effects 0.000 claims description 19
- 239000010960 cold rolled steel Substances 0.000 claims description 18
- 229910001563 bainite Inorganic materials 0.000 claims description 12
- 230000008569 process Effects 0.000 claims description 12
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims description 11
- 229910052729 chemical element Inorganic materials 0.000 claims description 8
- 239000000203 mixture Substances 0.000 claims description 8
- 229910052750 molybdenum Inorganic materials 0.000 claims description 7
- 229910052804 chromium Inorganic materials 0.000 claims description 6
- 229910052802 copper Inorganic materials 0.000 claims description 6
- 229910052759 nickel Inorganic materials 0.000 claims description 6
- 229910052742 iron Inorganic materials 0.000 claims description 5
- 239000012535 impurity Substances 0.000 claims description 3
- 229910052719 titanium Inorganic materials 0.000 claims description 3
- 238000012360 testing method Methods 0.000 description 18
- 230000000694 effects Effects 0.000 description 15
- 230000014759 maintenance of location Effects 0.000 description 11
- 230000015572 biosynthetic process Effects 0.000 description 10
- 229910001567 cementite Inorganic materials 0.000 description 7
- KSOKAHYVTMZFBJ-UHFFFAOYSA-N iron;methane Chemical compound C.[Fe].[Fe].[Fe] KSOKAHYVTMZFBJ-UHFFFAOYSA-N 0.000 description 7
- 230000001965 increasing effect Effects 0.000 description 6
- 238000007747 plating Methods 0.000 description 6
- 239000000463 material Substances 0.000 description 5
- 238000010791 quenching Methods 0.000 description 5
- 229910052782 aluminium Inorganic materials 0.000 description 4
- 238000005097 cold rolling Methods 0.000 description 4
- 230000006872 improvement Effects 0.000 description 4
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 239000006104 solid solution Substances 0.000 description 4
- 238000011282 treatment Methods 0.000 description 4
- 230000009471 action Effects 0.000 description 3
- 239000002826 coolant Substances 0.000 description 3
- 230000006866 deterioration Effects 0.000 description 3
- 230000002542 deteriorative effect Effects 0.000 description 3
- 229910052748 manganese Inorganic materials 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 230000000171 quenching effect Effects 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 229910052717 sulfur Inorganic materials 0.000 description 3
- 238000002441 X-ray diffraction Methods 0.000 description 2
- 229910045601 alloy Inorganic materials 0.000 description 2
- 239000000956 alloy Substances 0.000 description 2
- 229910052796 boron Inorganic materials 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 230000002708 enhancing effect Effects 0.000 description 2
- 238000005530 etching Methods 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 238000005098 hot rolling Methods 0.000 description 2
- 229910052757 nitrogen Inorganic materials 0.000 description 2
- 229910001562 pearlite Inorganic materials 0.000 description 2
- 238000003825 pressing Methods 0.000 description 2
- 238000001953 recrystallisation Methods 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 238000005728 strengthening Methods 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000009864 tensile test Methods 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 238000003466 welding Methods 0.000 description 2
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 1
- 229910001335 Galvanized steel Inorganic materials 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000032683 aging Effects 0.000 description 1
- 238000005275 alloying Methods 0.000 description 1
- 239000004411 aluminium Substances 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 238000000137 annealing Methods 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 238000004364 calculation method Methods 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 229910052593 corundum Inorganic materials 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000009713 electroplating Methods 0.000 description 1
- 238000005265 energy consumption Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000000446 fuel Substances 0.000 description 1
- 239000008397 galvanized steel Substances 0.000 description 1
- 238000000227 grinding Methods 0.000 description 1
- 229910052735 hafnium Inorganic materials 0.000 description 1
- 238000007731 hot pressing Methods 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 238000005272 metallurgy Methods 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000009740 moulding (composite fabrication) Methods 0.000 description 1
- 229910052758 niobium Inorganic materials 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 238000005498 polishing Methods 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000004088 simulation Methods 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000005482 strain hardening Methods 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- 230000000930 thermomechanical effect Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 238000009617 vacuum fusion Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 229910001845 yogo sapphire Inorganic materials 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D—WORKING OR PROCESSING OF SHEET METAL OR METAL TUBES, RODS OR PROFILES WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D22/00—Shaping without cutting, by stamping, spinning, or deep-drawing
- B21D22/02—Stamping using rigid devices or tools
- B21D22/022—Stamping using rigid devices or tools by heating the blank or stamping associated with heat treatment
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D—WORKING OR PROCESSING OF SHEET METAL OR METAL TUBES, RODS OR PROFILES WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D22/00—Shaping without cutting, by stamping, spinning, or deep-drawing
- B21D22/20—Deep-drawing
- B21D22/22—Deep-drawing with devices for holding the edge of the blanks
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B21—MECHANICAL METAL-WORKING WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D—WORKING OR PROCESSING OF SHEET METAL OR METAL TUBES, RODS OR PROFILES WITHOUT ESSENTIALLY REMOVING MATERIAL; PUNCHING METAL
- B21D37/00—Tools as parts of machines covered by this subclass
- B21D37/16—Heating or cooling
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D1/00—General methods or devices for heat treatment, e.g. annealing, hardening, quenching or tempering
- C21D1/62—Quenching devices
- C21D1/673—Quenching devices for die quenching
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D9/00—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor
- C21D9/46—Heat treatment, e.g. annealing, hardening, quenching or tempering, adapted for particular articles; Furnaces therefor for sheet metals
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/001—Ferrous alloys, e.g. steel alloys containing N
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/002—Ferrous alloys, e.g. steel alloys containing In, Mg, or other elements not provided for in one single group C22C38/001 - C22C38/60
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/02—Ferrous alloys, e.g. steel alloys containing silicon
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/04—Ferrous alloys, e.g. steel alloys containing manganese
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/06—Ferrous alloys, e.g. steel alloys containing aluminium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/08—Ferrous alloys, e.g. steel alloys containing nickel
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/12—Ferrous alloys, e.g. steel alloys containing tungsten, tantalum, molybdenum, vanadium, or niobium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/14—Ferrous alloys, e.g. steel alloys containing titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/16—Ferrous alloys, e.g. steel alloys containing copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/20—Ferrous alloys, e.g. steel alloys containing chromium with copper
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/22—Ferrous alloys, e.g. steel alloys containing chromium with molybdenum or tungsten
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/28—Ferrous alloys, e.g. steel alloys containing chromium with titanium or zirconium
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C38/00—Ferrous alloys, e.g. steel alloys
- C22C38/18—Ferrous alloys, e.g. steel alloys containing chromium
- C22C38/32—Ferrous alloys, e.g. steel alloys containing chromium with boron
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/002—Bainite
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/005—Ferrite
-
- C—CHEMISTRY; METALLURGY
- C21—METALLURGY OF IRON
- C21D—MODIFYING THE PHYSICAL STRUCTURE OF FERROUS METALS; GENERAL DEVICES FOR HEAT TREATMENT OF FERROUS OR NON-FERROUS METALS OR ALLOYS; MAKING METAL MALLEABLE, e.g. BY DECARBURISATION OR TEMPERING
- C21D2211/00—Microstructure comprising significant phases
- C21D2211/008—Martensite
Definitions
- the present invention relates to a hot press-formed product required to have high strength, such as used for structural members of automobile parts, a process for producing the same, and a thin steel sheet for hot press forming.
- the present invention relates to a hot press-formed product that can be provided with a prescribed shape and at the same time heat treated to have prescribed strength when a preheated steel sheet (blank) is formed into the prescribed shape, a process for producing such a hot press-formed product, and a thin steel sheet for hot press forming.
- a hot press-forming method for production of parts, in which method a steel sheet is heated to a prescribed temperature (e.g., a temperature for change in austenite phase) to lower its strength (i.e., make it easily formable) and then formed with a press tool at a temperature (e.g., room temperature) lower than that of the thin steel sheet, whereby the steel sheet is provided with a shape and at the same time heat treated by rapid cooling (quenching), which makes use of a temperature difference between both, to secure its strength after forming.
- a prescribed temperature e.g., a temperature for change in austenite phase
- a press tool at a temperature (e.g., room temperature) lower than that of the thin steel sheet
- a steel sheet is formed in a state of low strength, and therefore, the steel sheet has decreased springback (favorable shape fixability).
- Such a hot press-forming method has been called with various names, in addition to a hot press method, such as a hot forming method, a hot stamping method, a hot stamp method, and a die quench method.
- Fig. 1 is a schematic explanatory view showing the structure of a press tool for carrying out hot press forming as described above (hereinafter represented sometimes by "hot stamp").
- reference numerals 1, 2, 3, and 4 represent a punch, a die, a blank holder, and a steel sheet (blank), respectively
- abbreviations BHF, rp, rd, and CL represent a blank holding force, a punch shoulder radius, a die shoulder radius, and a clearance between the punch and the die, respectively.
- punch 1 and die 2 have passage 1a and passage 2a, respectively, formed in the inside thereof, through which passages a cooling medium (e.g., water) can be allowed to pass, and the press tool is made to have a structure so that these members can be cooled by allowing the cooling medium to pass through these passages.
- a cooling medium e.g., water
- the forming is started in a state where steel sheet (blank) 4 is softened by heating to a temperature within two-phase region, which is from Ac 1 transformation point to Ac 3 transformation point, or a temperature within single-phase region, which is not lower than Ac 3 transformation point. More specifically, steel sheet 4 is pushed into a cavity of die 2 (between the parts indicated by reference numerals 2 and 2 in Fig. 1 ) by punch 1 with steel sheet 4 in high-temperature state being sandwiched between die 2 and blank holder 3, thereby forming steel sheet 4 into a shape corresponding to the outer shape of punch 1 while reducing the outer diameter of steel sheet 4.
- hot stamp e.g., hot deep drawing
- steel sheets for hot stamp which have widely been used at present, there are known steel sheets based on 22MnB5 steel. These steel sheets have tensile strengths of 1500 MPa and elongations of about 6% to 8%, and have been applied to impact-resistant members (members neither deformed nor fractured as much as possible at the time of impact). In addition, some developments have also proceeded for C content increase and further highly strengthening (in 1500 to 1800 MPa class) based on 22MnB5 steel.
- both functions as an impact-resistant portion and an energy-absorbing portion may sometimes be provided in parts such as B pillars or rear side members.
- To produce such members there has mainly been used so far, for example, a method in which ultra-high tensile strength steel sheets having high strength of 980 MPa class and high tensile strength steel sheets having elongation of 440 MPa class are laser welded (to prepare a tailor welded blank, abbreviated as TWB) and then cold press formed.
- TWB tailor welded blank
- Non-patent Document 1 has proposed a method of laser welding 22MnB5 steel for hot stamp and a material that does not have high strength even if quenched with a press tool (to prepare a tailor welded blank, abbreviated as TWB), followed by hot stamp, in which method different strengths are provided so that tensile strength at a high strength side (i.e., impact-resistant portion side) becomes 1500 MPa (and elongation becomes 6% to 8%) and tensile strength at a low strength side (i.e., energy-absorbing portion side) becomes 440 MPa (and elongation becomes 12%).
- some techniques have also been proposed, such as disclosed in Non-patent Documents 2 to 4.
- Non-patent Documents 1 and 2 provide a tensile strength of not higher than 600 MPa and an elongation of about 12% to 18% at an energy-absorbing portion side, in which techniques, however, laser welding (to prepare a tailor welded blank, abbreviated as TWB) is needed previously, thereby increasing the number of steps and resulting in high cost. In addition, it results in the heating of energy-absorbing portions, which need not to be hardened originally. Therefore, these techniques are not preferred from the viewpoint of energy consumption.
- Non-patent Document 3 is based on 22MnB5 steel, in which boron addition, however, adversely affects the robustness of strength after quenching against heating to a temperature within two-phase region, making difficult the control of strength at an energy-absorbing portion side, and further making it possible to obtain only an elongation as low as 15%.
- Non-patent Document 4 is based on 22MnB5 steel, and therefore, this technique is not economic in that control is made in such a manner that 22MnB5, which originally has excellent hardenability, is not hardened (control of press tool cooling).
- Non-patent Document 1 Klaus Lamprecht, Gunter Deinzer, Anton Stich, Jurgen Lechler, Thomas Stohr, Marion Merklein, "Thermo-Mechanical Properties of Tailor Welded Blanks in Hot Sheet Metal Forming Processes", Proc. IDDRG2010, 2010 .
- Non-patent Document 2 Usibor1500P(22MnB5) /1500MPa-8%-Ductibor500/550-700MPa-17% [searched on April 27, 2013] Internet ⁇ http://www.arcelormittal.com/tailoredblanks/pre/seifware.p1>
- Non-patent Document 3 22MnB5/above AC3/1500MPa-8%-below AC3/Hv190-Ferrite/Cementite Rudiger Erhardt and Johannes Boke, "Industrial application of hot forming process simulation", Proc, of 1st Int. Conf. on Hot Sheet Metal Forming of High-Performance steel, ed. By Steinhoff, K., Oldenburg, M, Steinhoff, and Prakash, B., pp83-88, 2008 .
- Non-patent Document 4 Begona Casas, David Latre, Noemi Rodriguez, and Isaac Valls, "Tailor made tool materials for the present and upcoming tooling solutions in hot sheet metal forming", Proc, of 1st Int. Conf. on Hot Sheet Metal Forming of High-Performance steel, ed. By Steinhoff, K., Oldenburg, M, Steinhoff, and Prakash, B., pp23-35, 2008 .
- JP 2007 016 296 discloses a steel plate for hot press forming into automobile parts.
- the present invention has been made in view of the above-described circumstances, and its object is to provide a hot press-formed product in which balance between strength and elongation can be controlled in a proper range and high ductility can be achieved, a process useful for producing such a hot press-formed product, and a thin steel sheet for hot press forming.
- the hot press-formed product of the present invention which can achieve the above object, is a hot press-formed product, characterized by comprising a thin steel sheet formed by a hot press-forming method, and having a metallic structure that contains ferrite at 30% to 80% by area, bainitic ferrite at lower than 30% by area not including 0% by area, martensite at 30% by area or lower not including 0% by area, and retained austenite at 3% to 20% by area.
- the chemical element composition thereof is not particularly limited, typical examples of which may include the following chemical element composition: C at 0.1% to 0.3% where "%" means “% by mass", and the same applies to the below with respect to the chemical element composition; Si at 0.5% to 3%; Mn at 0.5% to 2%; P at 0.05% or lower not including 0% S at 0.05% or lower not including 0% Al at 0.01% to 0.1%; and N at 0.001% to 0.01%, and the remainder consisting of iron and unavoidable impurities.
- the hot press-formed product of the present invention it is also useful to allow additional elements to be contained, when needed; for example, (a) B at 0.01% or lower not including 0% and Ti at 0.1% or lower not including 0% ; (b) one or more selected from the group consisting of Cu, Ni, Cr, and Mo at 1% or lower not including 0% in total; and (c) V and/or Nb at 0.1% or lower not including 0% in total.
- the hot press-formed product may have further improved characteristics.
- the following steps may be used, i.e., heating a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at a reduction of 30% or higher, to a temperature not lower than Ac 1 transformation point and not higher than AC 1 transformation point x0.3+Ac 3 transformation point x 0.7 and then starting the forming of the hot-rolled steel sheet or the cold-rolled steel sheet with a press tool to produce the hot press-formed product, during which forming an average cooling rate of 20°C/sec or higher is kept in the press tool, and which forming is finished at a temperature not higher than bainite transformation starting temperature Bs - 100° .
- the forming finishing temperature may preferably be controlled in a temperature range of not higher than bainite transformation starting temperature Bs - 100°C and not lower than martensite transformation starting temperature Ms point, in which temperature range the steel sheet may preferably be retained for 10 seconds or longer, followed by the forming.
- the following method may be adopted as the other method.
- the thin steel sheet When a thin steel sheet is press formed with a press tool, the thin steel sheet may be heated to a temperature not lower than Ac 3 transformation point and not higher than 1000°C, and then cooled to a temperature not higher than 700°C and not lower than 500°C at an average cooling rate of 10°C/sec or lower, and then the forming of the thin steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than bainite transformation starting temperature Bs - 100°C.
- the forming finishing temperature may preferably be controlled in a temperature range of not higher than (bainite transformation starting temperature Bs - 100° and not lower than martensite transformation starting temperature Ms point, in which temperature range the steel sheet may preferably be retained for 10 seconds or longer, followed by the forming.
- the present invention further includes a thin steel sheet for hot press forming, which is intended for producing a hot press-formed product as described above, and this thin steel sheet is characterized by being a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at reduction of 30% or higher.
- the present invention makes it possible that: retained austenite can be allowed to exist at a proper fraction to adjust the metallic structure of a hot press-formed product by properly controlling the conditions of a hot press-forming method; a hot press-formed product having more enhanced ductility (retained ductility) inherent to the formed product as compared with the case where conventional 22MnB5 steel is used; and strength and elongation can be controlled by a combination of heat treatment conditions and pre-forming steel sheet structure (initial structure).
- the control of heating temperature within two-phase region makes it possible to provide different strengths and elongations freely.
- Fig. 1 is a schematic explanatory view showing the structure of a press tool for carrying out hot press forming.
- the present inventors have studied from various angles to realize a hot press-formed product having prescribed strength and further exhibiting excellent ductility (elongation) after forming when a steel sheet is heated to a prescribed temperature and then hot press formed to produce the formed product.
- the present inventors have found that a hot press-formed product having excellent balance between strength and ductility can be achieved when the type of a steel sheet, heating temperature, and forming conditions are properly controlled so that its structure is controlled to contain retained austenite at 3% to 20% by area in the hot press forming of a steel sheet with a press tool, thereby completing the present invention.
- High ductility of a hot press-formed product can be achieved by making its structure composed mainly of fine and high-ductility ferrite.
- the area fraction of ferrite should be controlled to 30% by area or higher. However, when this area fraction is higher than 80% by area, prescribed strength becomes not secured.
- the fraction of ferrite may preferably be not lower than 40% by area as the preferred lower limit (more preferably not lower than 45% by area) and not higher than 70% by area as the preferred upper limit (more preferably not higher than 65% by area).
- the fraction of bainitic ferrite should be controlled to lower than 30% by area as the upper limit.
- the fraction of bainitic ferrite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 10% by area) and not higher than 25% by area as the preferred upper limit (more preferably not higher than 20% by area).
- the fraction of martensite should be controlled to not higher than 30% by area as the upper limit.
- the fraction of martensite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 10% by area) and not higher than 25% by area as the preferred upper limit (more preferably not higher than 20% by area).
- Retained austenite is transformed into martensite during plastic deformation, thereby having the effect of increasing work hardening rate (transformation-inducing plasticity) to improve the ductility of a formed product.
- the fraction of retained austenite should be controlled to 3% by area or higher. When the fraction of retained austenite is higher, ductility becomes more excellent.
- retained austenite that can be secured is limited, of which upper limit becomes about 20% by area.
- the fraction of retained austenite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 7% by area) and not higher than 15% by area as the preferred upper limit (more preferably not higher than 10% by area).
- a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at a reduction of 30% or higher may be used, and when the hot-rolled steel sheet or the cold-rolled steel sheet is press formed with a pres tool, the hot-rolled steel sheet or the cold-rolled steel sheet may be heated to a temperature not lower than Ac 1 transformation point and not higher than (Ac 1 transformation point x 0.3 + Ac 3 transformation point x 0.7), and then the forming of the hot-rolled steel sheet or the cold-rolled steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than (bainite transformation starting temperature Bs - 100°C).
- the reasons for defining the respective requirements in this process are as follows:
- the type of a steel sheet should properly be selected.
- the hot-rolled steel sheet to be used may preferably have a metallic structure that contains ferrite at 50% by area or higher.
- the fraction of ferrite may preferably be not lower than 60% by area as the preferred lower limit (more preferably not lower than 70% by area).
- the fraction of ferrite in the hot-rolled steel sheet may preferably be not higher than 95% by area, more preferably not higher than 90% by area.
- a cold-rolled steel sheet it becomes an important requirement that recrystallization occurs during heating to form dislocation-free ferrite, and therefore, rolling (cold rolling) should be carried out at a prescribed reduction or higher so that recrystallization occurs.
- a cold-rolled steel sheet it may have any structure. From this viewpoint, when a cold-rolled steel sheet is used, it is preferable to use a cold-rolled steel sheet at a reduction of 30% or higher. The reduction may preferably be 40% or higher, more preferably 50% or higher.
- the heating temperature should be controlled in a prescribed range.
- the proper control of the heating temperature makes it possible to cause transformation into retained austenite or martensite in the subsequent cooling step to provide the final hot press-formed product with a desired structure.
- the heating temperature of the steel sheet is lower than Ac 1 transformation point, a sufficient fraction of austenite cannot be obtained during heating, and therefore, a prescribed fraction of retained austenite cannot be secured in the final structure (the structure of a formed product).
- the average cooling rate during forming and the forming finishing temperature should properly be controlled.
- the average cooling rate during forming should be controlled to 20°C/sec or higher, and the forming finishing temperature should be controlled to a temperature not higher than (bainite transformation starting temperature Bs point - 100°C, sometimes abbreviated as "Bs - 100°C").
- the average cooling rate during forming may preferably be 30°C/sec or higher (more preferably 40°C/sec or higher).
- the forming may be finished, while cooling to room temperature at an average cooling temperature as described above.
- the cooling is stopped after the cooling to a temperature not higher than Bs - 100°C, and then the forming may be finished.
- the control of the average cooling rate during forming can be achieved by a means of, for example, (a) controlling the temperature of a press tool (using a cooling medium shown in Fig. 1 above) or (b) controlling the thermal conductivity of a press tool (the same applies to the cooling in the method described below).
- the thin steel sheet when a steel sheet is press formed with a press tool, the thin steel sheet may be heated to a temperature not lower than Acs transformation point and not higher than 1000°C, and then the thin steel sheet is cooled to a temperature not higher than 700°C and not lower than 500°C at an average cooling temperature of 10°C/sec or lower, and then the forming of the thin steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than (bainite transformation starting temperature Bs - 100°C).
- the reasons for defining the respective requirements in this process are as follows (the same as described above applies to the cooling finishing temperature):
- the heating temperature should be controlled in a prescribed range.
- the proper control of the heating temperature makes it possible to cause transformation into a structure composed mainly of ferrite while securing a prescribed fraction of retained austenite in the subsequent cooling step to provide the final hot press-formed product with a desired structure.
- the heating temperature of the thin steel sheet is lower than Ac 3 transformation point, a sufficient fraction of austenite cannot be obtained during heating, and therefore, a prescribed fraction of retained austenite cannot be secured in the final structure (the structure of a formed product).
- the heating temperature of the thin steel sheet is higher than 1000°C, the grain size of austenite becomes increased during heating, and therefore, ferrite cannot be formed in the subsequent cooling.
- This cooling step is an important step for forming ferrite during cooling.
- the average cooling rate in this cooling step becomes higher than 10°C/sec, a prescribed fraction of ferrite cannot be secured.
- the average cooling rate may preferably be 7°C/sec or lower, more preferably 5°C/sec or lower.
- the cooling stopping temperature in this cooling step should be controlled to not higher than 700°C and not lower than 500°C.
- the cooling stopping temperature may preferably be not higher than 680°C as the preferred upper limit (more preferably not higher than 660°C) and not lower than 520°C as the preferred lower limit (more preferably not lower than 550°C).
- the forming finishing temperature should be controlled to not higher than (Bs - 100°C), but may preferably be controlled in a temperature range of not lower than martensite transformation starting temperature Ms (a temperature in this range may sometimes be referred to as the "cooling temperature changing temperature), in which temperature range retention may preferably be carried out for 10 seconds or longer.
- the bainite transformation can proceed from super-cooled austenite to form a structure composed mainly of ferrite by retention in the above temperature range for 10 seconds or longer.
- the retention time may preferably be 50 seconds or longer (more preferably 100 seconds or longer). When the retention time becomes too long, austenite starts to decompose, so that the fraction of retained austenite cannot become secured. Therefore, the retention time may preferably be 1000 seconds or shorter (more preferably 800 seconds or shorter).
- Retention as described above may be any of isothermal retention, monotonic cooling, and re-heating step, so long as it is in the above temperature range.
- retention as described above may be added at the stage when forming is finished.
- a retention step may be added within the above temperature range during the finish of forming. After forming is finished in such a manner, the steel sheet may be left as it is for cooling or cooled at a proper cooling rate to room temperature (25°C).
- the process for producing the hot press-formed product of the present invention can be applied, not only to the case where a hot press-formed product having a simple shape as shown in Fig. 1 above is produced (i.e., direct method), but also to the case where a formed product having a relatively complicated shape is produced, even if any of the methods described above is adopted.
- direct method a method of cold press forming in a step prior to hot press forming
- This method includes previously forming a difficult-to-form portion into an approximate shape by cold processing and then hot press forming the other portions.
- a method is used to produce, for example, a formed product having three projections (profile peaks) by forming, two projections are formed by cold press forming and the third projection is then formed by hot press forming.
- the present invention is intended for a hot press-formed product made of a high-strength steel sheet, the steel grade of which is acceptable, if it has an ordinary chemical element composition as a high-strength steel sheet, in which, however, C, Si, Mn, P, S, Al, and N contents may preferably be controlled in their respective proper ranges.
- C, Si, Mn, P, S, Al, and N contents may preferably be controlled in their respective proper ranges.
- C is an important element for securing retained austenite.
- concentration of austenite during heating at a temperature within two-phase region or at a temperature within single-phase region which is not lower than Ac 3 transformation point, allows the formation of retained austenite after quenching. It further contributes to an increase of martensite fraction.
- C content is lower than 0.1%, a prescribed fraction of retained austenite cannot be secured, making it impossible to obtain excellent ductility.
- C content becomes higher than 0.3%, it results in that strength becomes too high.
- C content may more preferably be not lower than 0.15% as the more preferred lower limit (still more preferably not lower than 0.20%) and not higher than 0.27% as the more preferred upper limit (still more preferably not higher than 0.25%).
- Si suppresses austenite after heating at a temperature within two-phase region or at a temperature within single-phase region, which is not lower than Ac 3 transformation point, from being formed into cementite, and exhibits the action of increasing the fraction of retained austenite. It further exhibits the action of enhancing strength by solid solution enhancement without deteriorating ductility too much.
- Si content is lower than 0.5%, retained austenite cannot be secured at a prescribed fraction, making it impossible to obtain excellent ductility.
- Si content becomes higher than 3%, the degree of solid solution enhancement becomes too high, resulting in the drastic deterioration of ductility.
- Si content may more preferably be not lower than 1.15% as the more preferred lower limit (still more preferably not lower than 1.20%) and not higher than 2.7% as the more preferred upper limit (still more preferably not higher than 2.5%).
- Mn is an element to stabilize austenite, and it contributes to an increase of retained austenite. To make such an effect exhibited, Mn may preferably be contained at 0.5% or higher. However, when Mn content becomes excessive, the formation of ferrite is prevented, thereby making it impossible to secure a prescribed fraction of ferrite, and therefore, Mn content may preferably be 2% or lower. In addition, a considerable improvement of austenite strength increases a hot rolling load, thereby making it difficult to produce steel sheets, and therefore, even from the viewpoint of productivity, it is not preferable that Mn is contained at higher than 2%. Mn content may more preferably be not lower than 0.7% as the more preferred lower limit (still more preferably not lower than 0.9%) and not higher than 1.8% as the more preferred higher limit (still more preferably not higher than 1.6%).
- P is an element unavoidably contained in steel and deteriorates ductility. Therefore, P content may preferably be reduced as low as possible. However, extreme reduction causes an increase of steel production cost, and reduction to 0% is difficult in the actual production. Therefore, P content may more preferably be controlled to 0.05% or lower (not including 0%). P content may more preferably be not higher than 0.045% as the more preferred upper limit (still more preferably not higher than 0.040%).
- S is also an element unavoidably contained in steel and deteriorates ductility, similarly to P. Therefore, S content may preferably be reduced as low as possible. However, extreme reduction causes an increase of steel production cost, and reduction to 0% is difficult in the actual production. Therefore, S content may preferably be controlled to 0.05% or lower (not including 0%). S content may more preferably be not higher than 0.045% as the more preferred upper limit (still more preferably not higher than 0.040%).
- Al is useful as a deoxidizing element and further useful for fixation of dissolved N in steel as AlN to improve ductility.
- Al content may preferably be controlled to 0.01% or higher. However, when Al content becomes higher than 0.1%, it results in the excessive formation of Al 2 O 3 to deteriorate ductility.
- A1 content may more preferably be not lower than 0.013% as the more preferred lower limit (still more preferably not lower than 0.015%) and not higher than 0.08% as the more preferred upper limit (still more preferably not higher than 0.06%).
- N is an element unavoidably incorporated in steel, and a reduction of N content may be preferred, which has, however, a limitation in actual process. Therefore, the lower limit of N content was set to 0.001%.
- the upper limit of N content was set to 0.01%. N content may more preferably be not higher than 0.008% as the more preferred upper limit (still more preferably not higher than 0.006%).
- the basic chemical components in the press-formed product of the present invention are as described above, and the remainder consists essentially of iron.
- the wording "consists essentially of iron” means that the press-formed product of the present invention can contain, in addition to iron, minor components (e.g., besides Mg, Ca, Sr, and Ba, REM such as La, and carbide-forming elements such as Zr, Hf, Ta, W, and Mo) in such a level that these minor components do not inhibit the characteristics of the steel sheet of the present invention, and can further contain unavoidable impurities (e.g., O, H) other than P, S, and N.
- minor components e.g., besides Mg, Ca, Sr, and Ba, REM such as La
- carbide-forming elements such as Zr, Hf, Ta, W, and Mo
- press-formed product of the present invention may contain additional elements, when needed; for example, (a) B at 0.01% or lower (not including 0%) and Ti at 0.1% or lower (not including 0%); (b) one or more selected from the group consisting of Cu, Ni, Cr, and Mo at 1% or lower (not including 0%) in total; and (c) V and/or Nb at 0.1% or lower (not including 0%) in total.
- the press-formed product may have further improved characteristics depending on the kinds of elements contained. When these elements are contained, their preferred ranges and grounds for limitation of their ranges are as follows:
- B is an element to prevent the formation of cementite during cooling after heating, thereby contributing to the securement of retained austenite.
- B may preferably be contained at 0.0001% or higher, but even if B is contained beyond 0.01%, the effect is saturated.
- B content may more preferably be not lower than 0.0002% as the more preferred lower limit (still more preferably not lower than 0.0005%) and not higher than 0.008% as the more preferred upper limit (still more preferably not higher than 0.005%).
- Ti fixes N and maintains B in solid solution state, thereby exhibiting the effect of improving hardenability.
- Ti may preferably be contained at least 4 times higher than N content.
- Ti content may more preferably be not lower than 0.05% as the more preferred lower limit (still more preferably not lower than 0.06%) and not higher than 0.09% as the more preferred higher limit (still more preferably not higher than 0.08%).
- these elements may preferably be contained at 0.01% or higher in total. Taking only characteristics into consideration, their content may be preferable when it is higher, but may preferably be controlled to 1% or lower in total because of a cost increase by alloy element addition. In addition, these elements have the action of considerably enhancing the strength of austenite, thereby increasing a hot rolling load so that the production of steel sheets becomes difficult. Therefore, even from the viewpoint of productivity, their content may preferably be controlled to 1% or lower. These elements' content may more preferably be not lower than 0.05% as the more preferred lower limit (still more preferably not lower than 0.06%) in total and not higher than 0.9% as the more preferred upper limit (still more preferably not higher than 0.8%) in total.
- V and Nb have the effect of forming fine carbide and make structure fine by pinning effect.
- these elements may preferably be contained at 0.001% or higher in total.
- these elements' content may preferably be controlled to 0.1% or lower in total.
- These elements' content may more preferably be not lower than 0.005% as the more preferred lower limit (still more preferably not lower than 0.008%) in total and not higher than 0.08% as the more preferred upper limit (still more preferably not higher than 0.06%) in total.
- the thin steel sheet for hot press forming of the present invention may be either a non-plated steel sheet or a plated steel sheet.
- the type of plating may be either ordinary galvanization or aluminium coating.
- the method of plating may be either hot-dip plating or electroplating. After the plating, alloying heat treatment may be carried out, or additional plating may be carried out as multilayer plating.
- the characteristics of formed products can be controlled by properly adjusting press forming conditions (heating temperature and cooling rate), and in addition, hot press-formed products having high ductility (retained ductility) can be obtained, so that they can be applied even to parts (e.g., energy-absorbing members), to which conventional hot press-formed products have hardly been applied; therefore, the present invention is extremely useful for extending the application range of hot press-formed products.
- the formed products, which can be obtained in the present invention have further enhanced residual ductility as compared with formed products, of which structure was adjusted by ordinary annealing after cold press forming.
- Table 1 further shows the calculated values of (Ac 1 transformation point x 0.3 + Ac 3 transformation point x 0.7) (these calculated values may hereinafter be referred to as "A values").
- Ac 1 transformation point °C 723 + 29.1 ⁇ Si ⁇ 10.7 ⁇ Mn + 16.9 ⁇ Cr ⁇ 16.9 ⁇ Ni
- the steel sheets thus obtained were heated under the respective conditions shown in Table 2 below, and then subjected to forming and cooling treatment using a high speed heat treatment testing system for steel sheets (CAS series, available from ULVAC-RIKO, Inc.), which can control an average cooling rate.
- the steel sheets to be subjected to cooling treatment had a size of 190 mm x 70 mm (and a sheet thickness of 1.4 mm).
- Test Nos. 1 to 14, 17 to 19, and 21 to 25 were the cases where hot-rolled steel sheets were used as steel sheets for forming.
- Test Nos. 15, 16, and 20 were the cases where cold-rolled steel sheets were used steel sheets for forming.
- the term "cooling 1" shown in Table 2 indicates cooling from a heating temperature to a temperature of 700°C to 500°C.
- cooling 2 indicates cooling from then to a temperature range of [(Bs - 100°C) to Ms point] (In Test Nos. 19 to 23, forming was started at this stage).
- the steel sheet was subjected to hot-dip galvanization to obtain a hot-dip galvanized steel sheet (Test No. 25).
- JIS No. 5 specimens were used for tensile tests to measure tensile strength (TS) and elongation (EL). At that time, strain rate in the tensile tests was set to 10 mm/sec. In the present invention, the specimens were evaluated as "passing" when fulfilling any of the conditions that: (a) tensile strength (TS) is from 780 to 979 MPa and elongation (EL) is 25% or higher; and (b) tensile strength (TS) is from 980 to 1179 MPa and elongation (EL) is 15% or higher.
- Test Nos. 1 to 10, 13, 15, 19 to 21, and 25 are Examples fulfilling the requirements defined in the present invention, thereby indicating that parts having satisfactory balance between strength and ductility were obtained.
- Test Nos. 11 to 12, 14, 16 to 18, and 22 to 24 are Comparative Examples not fulfilling any of the requirements defined in the present invention, thereby deteriorating any of the characteristics. More specifically, Test No. 11 was the case where steel having insufficient C content (steel grade K shown in Table 1) was used, so that retained austenite was not secured, thereby obtaining only low elongation (EL). Test No. 12 was the case where steel having insufficient Si content (steel grade L shown in Table 1), so that retained austenite was not secured, thereby obtaining only low elongation (EL).
- Test No. 14 was intended for conventional 2MnB5 equivalent steel (steel grade N shown in Table 1), so that retained austenite was not secured, thereby obtaining only low elongation (EL), although high strength was obtained.
- Test No. 16 was the case where cold-rolled steel sheet having low reduction was used, so that the formed product had a structure containing ferrite at 25% by area, thereby lowering elongation (EL).
- Test No. 17 was the case where the heating temperature was lower than Ac 1 transformation point, so that the formed product had a structure containing ferrite at 81% by area (the remainder was martensite and cementite) and retained austenite was not secured, thereby lowering elongation (EL) and tensile strength.
- Test No. 18 was the case where the heating temperature was higher than A value, so that ferrite and bainitic ferrite were not secured by excessive formation of martensite, thereby lowering elongation (EL).
- Test No. 22 was the case where the average cooling rate in cooling 1 was high, so that ferrite was not secured by the formation of bainitic ferrite, thereby lowering elongation (EL).
- Test No. 23 was the case where the average cooling rate in cooling 1 was low and the cooling rate changing temperature was low, so that the formed product had a structure containing ferrite at 83% by area (the remainder was bainitic ferrite) and retained austenite was not secured, thereby lowering elongation (EL).
- Test No. 24 was the case where the forming finishing temperature was high, so that pearlite was formed in the structure of the formed product and retained austenite was not secured, thereby lowing elongation (EL).
- the present invention makes it possible to provide a hot press-formed product, including a steel sheet formed by a hot press-forming method, and having a metallic structure that contains ferrite at 30% to 80% by area, bainitic ferrite at lower than 30% by area (not including 0%), martensite at 30% by area or lower (not including 0%), and retained austenite at 3% to 20% by area, whereby balance between strength and elongation can be controlled in a proper range and high ductility can be achieved.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Mechanical Engineering (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Shaping Metal By Deep-Drawing, Or The Like (AREA)
- Heat Treatment Of Sheet Steel (AREA)
- Heat Treatment Of Articles (AREA)
Description
- The present invention relates to a hot press-formed product required to have high strength, such as used for structural members of automobile parts, a process for producing the same, and a thin steel sheet for hot press forming. In particular, the present invention relates to a hot press-formed product that can be provided with a prescribed shape and at the same time heat treated to have prescribed strength when a preheated steel sheet (blank) is formed into the prescribed shape, a process for producing such a hot press-formed product, and a thin steel sheet for hot press forming.
- As one of the measures for fuel economy improvement of automobiles beginning from global environmental problems, automobile body lightening has proceeded, and steel sheets to be used for automobiles need to be strengthened as highly as possible. However, highly strengthening of steel sheets for automobile lightening lowers elongation EL or r value (Lankford value), resulting in the deterioration of press formability or shape fixability.
- To solve such a problem, a hot press-forming method has been adopted for production of parts, in which method a steel sheet is heated to a prescribed temperature (e.g., a temperature for change in austenite phase) to lower its strength (i.e., make it easily formable) and then formed with a press tool at a temperature (e.g., room temperature) lower than that of the thin steel sheet, whereby the steel sheet is provided with a shape and at the same time heat treated by rapid cooling (quenching), which makes use of a temperature difference between both, to secure its strength after forming.
- According to such a hot pressing method, a steel sheet is formed in a state of low strength, and therefore, the steel sheet has decreased springback (favorable shape fixability). In addition, the use of a material having excellent hardenability, to which alloy elements such as Mn and B have been added, thereby obtaining a strength of 1500 MPa class in terms of tensile strength by rapid cooling. Such a hot press-forming method has been called with various names, in addition to a hot press method, such as a hot forming method, a hot stamping method, a hot stamp method, and a die quench method.
-
Fig. 1 is a schematic explanatory view showing the structure of a press tool for carrying out hot press forming as described above (hereinafter represented sometimes by "hot stamp"). In this figure, reference numerals 1, 2, 3, and 4 represent a punch, a die, a blank holder, and a steel sheet (blank), respectively, and abbreviations BHF, rp, rd, and CL represent a blank holding force, a punch shoulder radius, a die shoulder radius, and a clearance between the punch and the die, respectively. In these parts, punch 1 and die 2 havepassage 1a andpassage 2a, respectively, formed in the inside thereof, through which passages a cooling medium (e.g., water) can be allowed to pass, and the press tool is made to have a structure so that these members can be cooled by allowing the cooling medium to pass through these passages. - When a steel sheet is subjected to hot stamp (e.g., hot deep drawing) with such a press tool, the forming is started in a state where steel sheet (blank) 4 is softened by heating to a temperature within two-phase region, which is from Ac1 transformation point to Ac3 transformation point, or a temperature within single-phase region, which is not lower than Ac3 transformation point. More specifically, steel sheet 4 is pushed into a cavity of die 2 (between the parts indicated by reference numerals 2 and 2 in
Fig. 1 ) by punch 1 with steel sheet 4 in high-temperature state being sandwiched between die 2 and blank holder 3, thereby forming steel sheet 4 into a shape corresponding to the outer shape of punch 1 while reducing the outer diameter of steel sheet 4. In addition, heat is removed from steel sheet 4 to the press tool (punch 1 and die 2) by cooling punch 1 and die 2 in parallel with the forming, and the hardening of the material is carried out by further retaining and cooling steel sheet 4 at the lower dead point in the forming (the point of time when the punch head is positioned at the deepest level: the state shown inFig. 1 ). Formed products with high dimension accuracy and strength of 1500 MPa class can be obtained by carrying out such a forming method. Furthermore, such a forming method results in that the volume of a pressing machine can be made smaller because a forming load can be reduced as compared with the case where parts of the same strength class are formed by cold pressing. - As steel sheets for hot stamp, which have widely been used at present, there are known steel sheets based on 22MnB5 steel. These steel sheets have tensile strengths of 1500 MPa and elongations of about 6% to 8%, and have been applied to impact-resistant members (members neither deformed nor fractured as much as possible at the time of impact). In addition, some developments have also proceeded for C content increase and further highly strengthening (in 1500 to 1800 MPa class) based on 22MnB5 steel.
- However, there is almost no application of steel grades other than 22MnB5 steel. One can find a present situation where little consideration is made on steel grades or methods for controlling the strength and elongation of parts (e.g., strength lowering to 980MPa class and elongation enhancement to 20%) to extend their application range to other than impact-resistant members.
- In middle or higher class automobiles, taking into consideration compatibility (function of, when a small class automobile comes to collide, making safe of the other side) at the time of side or back impact, both functions as an impact-resistant portion and an energy-absorbing portion may sometimes be provided in parts such as B pillars or rear side members. To produce such members, there has mainly been used so far, for example, a method in which ultra-high tensile strength steel sheets having high strength of 980 MPa class and high tensile strength steel sheets having elongation of 440 MPa class are laser welded (to prepare a tailor welded blank, abbreviated as TWB) and then cold press formed. However, in recent years, the development of a technique has proceeded, in which parts are each provided with different strengths by hot stamp.
- For example, Non-patent Document 1 has proposed a method of laser welding 22MnB5 steel for hot stamp and a material that does not have high strength even if quenched with a press tool (to prepare a tailor welded blank, abbreviated as TWB), followed by hot stamp, in which method different strengths are provided so that tensile strength at a high strength side (i.e., impact-resistant portion side) becomes 1500 MPa (and elongation becomes 6% to 8%) and tensile strength at a low strength side (i.e., energy-absorbing portion side) becomes 440 MPa (and elongation becomes 12%). In addition, as the technique of providing parts each with different strengths, some techniques have also been proposed, such as disclosed in Non-patent Documents 2 to 4.
- The techniques disclosed in Non-patent Documents 1 and 2 provide a tensile strength of not higher than 600 MPa and an elongation of about 12% to 18% at an energy-absorbing portion side, in which techniques, however, laser welding (to prepare a tailor welded blank, abbreviated as TWB) is needed previously, thereby increasing the number of steps and resulting in high cost. In addition, it results in the heating of energy-absorbing portions, which need not to be hardened originally. Therefore, these techniques are not preferred from the viewpoint of energy consumption.
- The technique disclosed in Non-patent Document 3 is based on 22MnB5 steel, in which boron addition, however, adversely affects the robustness of strength after quenching against heating to a temperature within two-phase region, making difficult the control of strength at an energy-absorbing portion side, and further making it possible to obtain only an elongation as low as 15%.
- The technique disclosed in Non-patent Document 4 is based on 22MnB5 steel, and therefore, this technique is not economic in that control is made in such a manner that 22MnB5, which originally has excellent hardenability, is not hardened (control of press tool cooling).
- Non-patent Document 1: Klaus Lamprecht, Gunter Deinzer, Anton Stich, Jurgen Lechler, Thomas Stohr, Marion Merklein, "Thermo-Mechanical Properties of Tailor Welded Blanks in Hot Sheet Metal Forming Processes", Proc. IDDRG2010, 2010.
- Non-patent Document 2: Usibor1500P(22MnB5) /1500MPa-8%-Ductibor500/550-700MPa-17% [searched on April 27, 2013] Internet <http://www.arcelormittal.com/tailoredblanks/pre/seifware.p1>
- Non-patent Document 3: 22MnB5/above AC3/1500MPa-8%-below AC3/Hv190-Ferrite/Cementite Rudiger Erhardt and Johannes Boke, "Industrial application of hot forming process simulation", Proc, of 1st Int. Conf. on Hot Sheet Metal Forming of High-Performance steel, ed. By Steinhoff, K., Oldenburg, M, Steinhoff, and Prakash, B., pp83-88, 2008.
- Non-patent Document 4: Begona Casas, David Latre, Noemi Rodriguez, and Isaac Valls, "Tailor made tool materials for the present and upcoming tooling solutions in hot sheet metal forming", Proc, of 1st Int. Conf. on Hot Sheet Metal Forming of High-Performance steel, ed. By Steinhoff, K., Oldenburg, M, Steinhoff, and Prakash, B., pp23-35, 2008.
JP 2007 016 296 - The present invention has been made in view of the above-described circumstances, and its object is to provide a hot press-formed product in which balance between strength and elongation can be controlled in a proper range and high ductility can be achieved, a process useful for producing such a hot press-formed product, and a thin steel sheet for hot press forming.
- The hot press-formed product of the present invention, which can achieve the above object, is a hot press-formed product, characterized by comprising a thin steel sheet formed by a hot press-forming method, and having a metallic structure that contains ferrite at 30% to 80% by area, bainitic ferrite at lower than 30% by area not including 0% by area, martensite at 30% by area or lower not including 0% by area, and retained austenite at 3% to 20% by area.
- In the hot press-formed product of the present invention, the chemical element composition thereof is not particularly limited, typical examples of which may include the following chemical element composition: C at 0.1% to 0.3% where "%" means "% by mass", and the same applies to the below with respect to the chemical element composition; Si at 0.5% to 3%; Mn at 0.5% to 2%; P at 0.05% or lower not including 0% S at 0.05% or lower not including 0% Al at 0.01% to 0.1%; and N at 0.001% to 0.01%, and the remainder consisting of iron and unavoidable impurities.
- In the hot press-formed product of the present invention, it is also useful to allow additional elements to be contained, when needed; for example, (a) B at 0.01% or lower not including 0% and Ti at 0.1% or lower not including 0% ; (b) one or more selected from the group consisting of Cu, Ni, Cr, and Mo at 1% or lower not including 0% in total; and (c) V and/or Nb at 0.1% or lower not including 0% in total. Depending on the kind of element to be contained, the hot press-formed product may have further improved characteristics.
- When the hot press-formed product of the present invention is produced, the following steps may be used, i.e., heating a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at a reduction of 30% or higher, to a temperature not lower than Ac1 transformation point and not higher than AC1 transformation point x0.3+Ac3 transformation point x 0.7 and then starting the forming of the hot-rolled steel sheet or the cold-rolled steel sheet with a press tool to produce the hot press-formed product, during which forming an average cooling rate of 20°C/sec or higher is kept in the press tool, and which forming is finished at a temperature not higher than bainite transformation starting temperature Bs - 100° . The forming finishing temperature may preferably be controlled in a temperature range of not higher than bainite transformation starting temperature Bs - 100°C and not lower than martensite transformation starting temperature Ms point, in which temperature range the steel sheet may preferably be retained for 10 seconds or longer, followed by the forming.
- Alternatively, the following method may be adopted as the other method. When a thin steel sheet is press formed with a press tool, the thin steel sheet may be heated to a temperature not lower than Ac3 transformation point and not higher than 1000°C, and then cooled to a temperature not higher than 700°C and not lower than 500°C at an average cooling rate of 10°C/sec or lower, and then the forming of the thin steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than bainite transformation starting temperature Bs - 100°C. Also in this method, the forming finishing temperature may preferably be controlled in a temperature range of not higher than (bainite transformation starting temperature Bs - 100° and not lower than martensite transformation starting temperature Ms point, in which temperature range the steel sheet may preferably be retained for 10 seconds or longer, followed by the forming.
- The present invention further includes a thin steel sheet for hot press forming, which is intended for producing a hot press-formed product as described above, and this thin steel sheet is characterized by being a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at reduction of 30% or higher.
- The present invention makes it possible that: retained austenite can be allowed to exist at a proper fraction to adjust the metallic structure of a hot press-formed product by properly controlling the conditions of a hot press-forming method; a hot press-formed product having more enhanced ductility (retained ductility) inherent to the formed product as compared with the case where conventional 22MnB5 steel is used; and strength and elongation can be controlled by a combination of heat treatment conditions and pre-forming steel sheet structure (initial structure). In addition, the control of heating temperature within two-phase region makes it possible to provide different strengths and elongations freely.
-
Fig. 1 is a schematic explanatory view showing the structure of a press tool for carrying out hot press forming. - The present inventors have studied from various angles to realize a hot press-formed product having prescribed strength and further exhibiting excellent ductility (elongation) after forming when a steel sheet is heated to a prescribed temperature and then hot press formed to produce the formed product.
- As a result, the present inventors have found that a hot press-formed product having excellent balance between strength and ductility can be achieved when the type of a steel sheet, heating temperature, and forming conditions are properly controlled so that its structure is controlled to contain retained austenite at 3% to 20% by area in the hot press forming of a steel sheet with a press tool, thereby completing the present invention.
- The reasons for setting the ranges of the respective structures (basic structures) in the hot press-formed product of the present invention are as follows:
- High ductility of a hot press-formed product can be achieved by making its structure composed mainly of fine and high-ductility ferrite.
- From this viewpoint, the area fraction of ferrite should be controlled to 30% by area or higher. However, when this area fraction is higher than 80% by area, prescribed strength becomes not secured. The fraction of ferrite may preferably be not lower than 40% by area as the preferred lower limit (more preferably not lower than 45% by area) and not higher than 70% by area as the preferred upper limit (more preferably not higher than 65% by area).
- Bainitic ferrite is effective for strength improvement, but it causes a slight lowering of ductility. Therefore, the fraction of bainitic ferrite should be controlled to lower than 30% by area as the upper limit. The fraction of bainitic ferrite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 10% by area) and not higher than 25% by area as the preferred upper limit (more preferably not higher than 20% by area).
- Martensite is effective for strength improvement, but it causes a considerable lowering of ductility. Therefore, the fraction of martensite should be controlled to not higher than 30% by area as the upper limit. The fraction of martensite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 10% by area) and not higher than 25% by area as the preferred upper limit (more preferably not higher than 20% by area).
- Retained austenite is transformed into martensite during plastic deformation, thereby having the effect of increasing work hardening rate (transformation-inducing plasticity) to improve the ductility of a formed product. To make such an effect exhibited, the fraction of retained austenite should be controlled to 3% by area or higher. When the fraction of retained austenite is higher, ductility becomes more excellent. In a composition to be used for automobile steel sheets, retained austenite that can be secured is limited, of which upper limit becomes about 20% by area. The fraction of retained austenite may preferably be not lower than 5% by area as the preferred lower limit (more preferably not lower than 7% by area) and not higher than 15% by area as the preferred upper limit (more preferably not higher than 10% by area).
- When the hot press-formed product of the present invention is produced, a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at a reduction of 30% or higher, may be used, and when the hot-rolled steel sheet or the cold-rolled steel sheet is press formed with a pres tool, the hot-rolled steel sheet or the cold-rolled steel sheet may be heated to a temperature not lower than Ac1 transformation point and not higher than (Ac1 transformation point x 0.3 + Ac3 transformation point x 0.7), and then the forming of the hot-rolled steel sheet or the cold-rolled steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than (bainite transformation starting temperature Bs - 100°C). The reasons for defining the respective requirements in this process are as follows:
- To obtain ferrite structure, which has high contributions to ductility, during heating to a temperature within two-phase region, the type of a steel sheet (steel sheet for forming) should properly be selected. When a hot-rolled steel sheet is used as the steel sheet for forming, it is important to achieve that the fraction of ferrite is high and ferrite is retained during heating to a temperature within two-phase region. From this viewpoint, the hot-rolled steel sheet to be used may preferably have a metallic structure that contains ferrite at 50% by area or higher. The fraction of ferrite may preferably be not lower than 60% by area as the preferred lower limit (more preferably not lower than 70% by area). When the fraction of ferrite in the hot-rolled steel sheet becomes too high, the fraction of ferrite in the formed product becomes too high. Therefore, the fraction of ferrite in the hot-rolled steel sheet may preferably be not higher than 95% by area, more preferably not higher than 90% by area.
- On the other hand, a cold-rolled steel sheet is used, it becomes an important requirement that recrystallization occurs during heating to form dislocation-free ferrite, and therefore, rolling (cold rolling) should be carried out at a prescribed reduction or higher so that recrystallization occurs. In the case of a cold-rolled steel sheet, it may have any structure. From this viewpoint, when a cold-rolled steel sheet is used, it is preferable to use a cold-rolled steel sheet at a reduction of 30% or higher. The reduction may preferably be 40% or higher, more preferably 50% or higher. The "reduction" as used herein is a value determined by formula (1) below.
- To cause the partial transformation, while retaining, of ferrite, which is contained in the steel sheet, into austenite, the heating temperature should be controlled in a prescribed range. The proper control of the heating temperature makes it possible to cause transformation into retained austenite or martensite in the subsequent cooling step to provide the final hot press-formed product with a desired structure. When the heating temperature of the steel sheet is lower than Ac1 transformation point, a sufficient fraction of austenite cannot be obtained during heating, and therefore, a prescribed fraction of retained austenite cannot be secured in the final structure (the structure of a formed product). When the heating temperature of the thin steel sheet is higher than (Ac1 transformation point x 0.3 + Ac3 transformation point x 0.7), the fraction of transformed austenite is increased too highly during heating, and therefore, a prescribed fraction of ferrite cannot be secured in the final structure (the structure of a formed product).
- To change the austenite, which was formed in the above heating step, into a prescribed fraction of retained austenite, while preventing the formation of cementite, the average cooling rate during forming and the forming finishing temperature should properly be controlled. From this viewpoint, the average cooling rate during forming should be controlled to 20°C/sec or higher, and the forming finishing temperature should be controlled to a temperature not higher than (bainite transformation starting temperature Bs point - 100°C, sometimes abbreviated as "Bs - 100°C"). The average cooling rate during forming may preferably be 30°C/sec or higher (more preferably 40°C/sec or higher). With respect to the forming finishing temperature, the forming may be finished, while cooling to room temperature at an average cooling temperature as described above. Alternatively, the cooling is stopped after the cooling to a temperature not higher than Bs - 100°C, and then the forming may be finished. The control of the average cooling rate during forming can be achieved by a means of, for example, (a) controlling the temperature of a press tool (using a cooling medium shown in
Fig. 1 above) or (b) controlling the thermal conductivity of a press tool (the same applies to the cooling in the method described below). - As another method for producing the press-formed product of the present invention, when a steel sheet is press formed with a press tool, the thin steel sheet may be heated to a temperature not lower than Acs transformation point and not higher than 1000°C, and then the thin steel sheet is cooled to a temperature not higher than 700°C and not lower than 500°C at an average cooling temperature of 10°C/sec or lower, and then the forming of the thin steel sheet may be started, during which forming an average cooling rate of 20°C/sec or higher may be kept in the press tool, and which forming may be finished at a temperature not higher than (bainite transformation starting temperature Bs - 100°C). The reasons for defining the respective requirements in this process are as follows (the same as described above applies to the cooling finishing temperature):
- To properly adjust the structure of a hot press-formed product, the heating temperature should be controlled in a prescribed range. The proper control of the heating temperature makes it possible to cause transformation into a structure composed mainly of ferrite while securing a prescribed fraction of retained austenite in the subsequent cooling step to provide the final hot press-formed product with a desired structure. When the heating temperature of the thin steel sheet is lower than Ac3 transformation point, a sufficient fraction of austenite cannot be obtained during heating, and therefore, a prescribed fraction of retained austenite cannot be secured in the final structure (the structure of a formed product). When the heating temperature of the thin steel sheet is higher than 1000°C, the grain size of austenite becomes increased during heating, and therefore, ferrite cannot be formed in the subsequent cooling.
- This cooling step is an important step for forming ferrite during cooling. When the average cooling rate in this cooling step becomes higher than 10°C/sec, a prescribed fraction of ferrite cannot be secured. The average cooling rate may preferably be 7°C/sec or lower, more preferably 5°C/sec or lower. The cooling stopping temperature in this cooling step (this temperature may sometimes be referred to as the "cooling rate changing temperature") should be controlled to not higher than 700°C and not lower than 500°C. When the cooling stopping temperature becomes higher than 700°C, a sufficient fraction of ferrite cannot be secured. When the cooling stopping temperature becomes lower than 500°C, the fraction of ferrite becomes too high, and therefore, prescribed strength cannot be secured. The cooling stopping temperature may preferably be not higher than 680°C as the preferred upper limit (more preferably not higher than 660°C) and not lower than 520°C as the preferred lower limit (more preferably not lower than 550°C).
- In any of these methods, the forming finishing temperature should be controlled to not higher than (Bs - 100°C), but may preferably be controlled in a temperature range of not lower than martensite transformation starting temperature Ms (a temperature in this range may sometimes be referred to as the "cooling temperature changing temperature), in which temperature range retention may preferably be carried out for 10 seconds or longer. The bainite transformation can proceed from super-cooled austenite to form a structure composed mainly of ferrite by retention in the above temperature range for 10 seconds or longer. The retention time may preferably be 50 seconds or longer (more preferably 100 seconds or longer). When the retention time becomes too long, austenite starts to decompose, so that the fraction of retained austenite cannot become secured. Therefore, the retention time may preferably be 1000 seconds or shorter (more preferably 800 seconds or shorter).
- Retention as described above may be any of isothermal retention, monotonic cooling, and re-heating step, so long as it is in the above temperature range. With regard to a relationship between such retention and forming, retention as described above may be added at the stage when forming is finished. Alternatively, a retention step may be added within the above temperature range during the finish of forming. After forming is finished in such a manner, the steel sheet may be left as it is for cooling or cooled at a proper cooling rate to room temperature (25°C).
- The process for producing the hot press-formed product of the present invention can be applied, not only to the case where a hot press-formed product having a simple shape as shown in
Fig. 1 above is produced (i.e., direct method), but also to the case where a formed product having a relatively complicated shape is produced, even if any of the methods described above is adopted. However, in the case of a complicated product shape, it may be difficult to provide a product with the final shape by a single press forming step. In such a case, there can be used a method of cold press forming in a step prior to hot press forming (this method has been referred to as "indirect method"). This method includes previously forming a difficult-to-form portion into an approximate shape by cold processing and then hot press forming the other portions. When such a method is used to produce, for example, a formed product having three projections (profile peaks) by forming, two projections are formed by cold press forming and the third projection is then formed by hot press forming. - The present invention is intended for a hot press-formed product made of a high-strength steel sheet, the steel grade of which is acceptable, if it has an ordinary chemical element composition as a high-strength steel sheet, in which, however, C, Si, Mn, P, S, Al, and N contents may preferably be controlled in their respective proper ranges. From this viewpoint, the preferred ranges of these chemical elements and the grounds for limiting their ranges are as follows:
- C is an important element for securing retained austenite. The concentration of austenite during heating at a temperature within two-phase region or at a temperature within single-phase region, which is not lower than Ac3 transformation point, allows the formation of retained austenite after quenching. It further contributes to an increase of martensite fraction. When C content is lower than 0.1%, a prescribed fraction of retained austenite cannot be secured, making it impossible to obtain excellent ductility. When C content becomes higher than 0.3%, it results in that strength becomes too high. C content may more preferably be not lower than 0.15% as the more preferred lower limit (still more preferably not lower than 0.20%) and not higher than 0.27% as the more preferred upper limit (still more preferably not higher than 0.25%).
- Si suppresses austenite after heating at a temperature within two-phase region or at a temperature within single-phase region, which is not lower than Ac3 transformation point, from being formed into cementite, and exhibits the action of increasing the fraction of retained austenite. It further exhibits the action of enhancing strength by solid solution enhancement without deteriorating ductility too much. When Si content is lower than 0.5%, retained austenite cannot be secured at a prescribed fraction, making it impossible to obtain excellent ductility. When Si content becomes higher than 3%, the degree of solid solution enhancement becomes too high, resulting in the drastic deterioration of ductility. Si content may more preferably be not lower than 1.15% as the more preferred lower limit (still more preferably not lower than 1.20%) and not higher than 2.7% as the more preferred upper limit (still more preferably not higher than 2.5%).
- Mn is an element to stabilize austenite, and it contributes to an increase of retained austenite. To make such an effect exhibited, Mn may preferably be contained at 0.5% or higher. However, when Mn content becomes excessive, the formation of ferrite is prevented, thereby making it impossible to secure a prescribed fraction of ferrite, and therefore, Mn content may preferably be 2% or lower. In addition, a considerable improvement of austenite strength increases a hot rolling load, thereby making it difficult to produce steel sheets, and therefore, even from the viewpoint of productivity, it is not preferable that Mn is contained at higher than 2%. Mn content may more preferably be not lower than 0.7% as the more preferred lower limit (still more preferably not lower than 0.9%) and not higher than 1.8% as the more preferred higher limit (still more preferably not higher than 1.6%).
- P is an element unavoidably contained in steel and deteriorates ductility. Therefore, P content may preferably be reduced as low as possible. However, extreme reduction causes an increase of steel production cost, and reduction to 0% is difficult in the actual production. Therefore, P content may more preferably be controlled to 0.05% or lower (not including 0%). P content may more preferably be not higher than 0.045% as the more preferred upper limit (still more preferably not higher than 0.040%).
- S is also an element unavoidably contained in steel and deteriorates ductility, similarly to P. Therefore, S content may preferably be reduced as low as possible. However, extreme reduction causes an increase of steel production cost, and reduction to 0% is difficult in the actual production. Therefore, S content may preferably be controlled to 0.05% or lower (not including 0%). S content may more preferably be not higher than 0.045% as the more preferred upper limit (still more preferably not higher than 0.040%).
- Al is useful as a deoxidizing element and further useful for fixation of dissolved N in steel as AlN to improve ductility. To make such an effect effectively exhibited, Al content may preferably be controlled to 0.01% or higher. However, when Al content becomes higher than 0.1%, it results in the excessive formation of Al2O3 to deteriorate ductility. A1 content may more preferably be not lower than 0.013% as the more preferred lower limit (still more preferably not lower than 0.015%) and not higher than 0.08% as the more preferred upper limit (still more preferably not higher than 0.06%).
- N is an element unavoidably incorporated in steel, and a reduction of N content may be preferred, which has, however, a limitation in actual process. Therefore, the lower limit of N content was set to 0.001%. When N content becomes excessive, ductility is deteriorated by strain aging, or the addition of B causes deposition of N as BN, thereby lowering the effect of improving hardenability by solid solution of B. Therefore, the upper limit of N content was set to 0.01%. N content may more preferably be not higher than 0.008% as the more preferred upper limit (still more preferably not higher than 0.006%).
- The basic chemical components in the press-formed product of the present invention are as described above, and the remainder consists essentially of iron. The wording "consists essentially of iron" means that the press-formed product of the present invention can contain, in addition to iron, minor components (e.g., besides Mg, Ca, Sr, and Ba, REM such as La, and carbide-forming elements such as Zr, Hf, Ta, W, and Mo) in such a level that these minor components do not inhibit the characteristics of the steel sheet of the present invention, and can further contain unavoidable impurities (e.g., O, H) other than P, S, and N.
- It is also useful to allow the press-formed product of the present invention to contain additional elements, when needed; for example, (a) B at 0.01% or lower (not including 0%) and Ti at 0.1% or lower (not including 0%); (b) one or more selected from the group consisting of Cu, Ni, Cr, and Mo at 1% or lower (not including 0%) in total; and (c) V and/or Nb at 0.1% or lower (not including 0%) in total. The press-formed product may have further improved characteristics depending on the kinds of elements contained. When these elements are contained, their preferred ranges and grounds for limitation of their ranges are as follows:
- B is an element to prevent the formation of cementite during cooling after heating, thereby contributing to the securement of retained austenite. To make such an effect exhibited, B may preferably be contained at 0.0001% or higher, but even if B is contained beyond 0.01%, the effect is saturated. B content may more preferably be not lower than 0.0002% as the more preferred lower limit (still more preferably not lower than 0.0005%) and not higher than 0.008% as the more preferred upper limit (still more preferably not higher than 0.005%).
- On the other hand, Ti fixes N and maintains B in solid solution state, thereby exhibiting the effect of improving hardenability. To make such an effect exhibited, Ti may preferably be contained at least 4 times higher than N content. However, when Ti content becomes excessive beyond 0.1%, it results in excessive formation of TiC, thereby causing an increase of strength by precipitation enhancement but a deterioration of ductility. Ti content may more preferably be not lower than 0.05% as the more preferred lower limit (still more preferably not lower than 0.06%) and not higher than 0.09% as the more preferred higher limit (still more preferably not higher than 0.08%).
- Cu, Ni, Cr, and Mo prevent the formation of cementite during cooling after heating, and effectively act the securement of retained austenite. To make such an effect exhibited, these elements may preferably be contained at 0.01% or higher in total. Taking only characteristics into consideration, their content may be preferable when it is higher, but may preferably be controlled to 1% or lower in total because of a cost increase by alloy element addition. In addition, these elements have the action of considerably enhancing the strength of austenite, thereby increasing a hot rolling load so that the production of steel sheets becomes difficult. Therefore, even from the viewpoint of productivity, their content may preferably be controlled to 1% or lower. These elements' content may more preferably be not lower than 0.05% as the more preferred lower limit (still more preferably not lower than 0.06%) in total and not higher than 0.9% as the more preferred upper limit (still more preferably not higher than 0.8%) in total.
- V and Nb have the effect of forming fine carbide and make structure fine by pinning effect. To make such an effect exhibited, these elements may preferably be contained at 0.001% or higher in total. However, when these elements' content becomes excessive, it results in the formation of coarse carbide, which becomes the origin of fracture, thereby deteriorating ductility in contrast. Therefore, these elements' content may preferably be controlled to 0.1% or lower in total. These elements' content may more preferably be not lower than 0.005% as the more preferred lower limit (still more preferably not lower than 0.008%) in total and not higher than 0.08% as the more preferred upper limit (still more preferably not higher than 0.06%) in total.
- The thin steel sheet for hot press forming of the present invention may be either a non-plated steel sheet or a plated steel sheet. When it is a plated steel sheet, the type of plating may be either ordinary galvanization or aluminium coating. The method of plating may be either hot-dip plating or electroplating. After the plating, alloying heat treatment may be carried out, or additional plating may be carried out as multilayer plating.
- According to the present invention, the characteristics of formed products, such as strength and elongation, can be controlled by properly adjusting press forming conditions (heating temperature and cooling rate), and in addition, hot press-formed products having high ductility (retained ductility) can be obtained, so that they can be applied even to parts (e.g., energy-absorbing members), to which conventional hot press-formed products have hardly been applied; therefore, the present invention is extremely useful for extending the application range of hot press-formed products. The formed products, which can be obtained in the present invention, have further enhanced residual ductility as compared with formed products, of which structure was adjusted by ordinary annealing after cold press forming.
- The following will describe the advantageous effects of the present invention more specifically by way of Examples, but the present invention is not limited to the Examples described below. The present invention can be put into practice after appropriate modifications or variations within a range capable of meeting the gist described above and below, all of which are included in the technical scope of the present invention.
- The present application claims the benefit of priority based on Japanese Patent Application No.
2011-130636 filed on June 10, 2011 2011-130636 filed on June 10, 2011 - Steel materials having respective chemical element compositions shown in Table 1 below were formed into slabs for experimental use by a vacuum fusion method, after which the slabs were hot rolled, followed by cooling, and then wound. These rolled sheets were further cold rolled into thin steel sheets, followed by quench treatment so that they had respectively prescribed initial structures. In Table 1, Ac1 transformation point, Ac3 transformation point, Ms point, and (Bs - 100°C) were determined respectively on the basis of formulas (2) to (5) described below (see, e.g., the Japanese translation of "The Physical Metallurgy of Steels" originally written by William C. Leslie, published by Maruzen, 1985). Table 1 further shows the calculated values of (Ac1 transformation point x 0.3 + Ac3 transformation point x 0.7) (these calculated values may hereinafter be referred to as "A values").
- The steel sheets thus obtained were heated under the respective conditions shown in Table 2 below, and then subjected to forming and cooling treatment using a high speed heat treatment testing system for steel sheets (CAS series, available from ULVAC-RIKO, Inc.), which can control an average cooling rate. The steel sheets to be subjected to cooling treatment had a size of 190 mm x 70 mm (and a sheet thickness of 1.4 mm). Test Nos. 1 to 14, 17 to 19, and 21 to 25 were the cases where hot-rolled steel sheets were used as steel sheets for forming. Test Nos. 15, 16, and 20 were the cases where cold-rolled steel sheets were used steel sheets for forming. The term "cooling 1" shown in Table 2 indicates cooling from a heating temperature to a temperature of 700°C to 500°C. The term "cooling 2" shown in Table 2 indicates cooling from then to a temperature range of [(Bs - 100°C) to Ms point] (In Test Nos. 19 to 23, forming was started at this stage). When needed, the steel sheet was subjected to hot-dip galvanization to obtain a hot-dip galvanized steel sheet (Test No. 25).
- For the respective steel sheets after the above treatments (heating, forming, and cooling), measurement of tensile strength (TS) and elongation (total elongation EL), and observation of metallic structure (fraction of each structure), were carried out by the methods described below.
- JIS No. 5 specimens were used for tensile tests to measure tensile strength (TS) and elongation (EL). At that time, strain rate in the tensile tests was set to 10 mm/sec. In the present invention, the specimens were evaluated as "passing" when fulfilling any of the conditions that: (a) tensile strength (TS) is from 780 to 979 MPa and elongation (EL) is 25% or higher; and (b) tensile strength (TS) is from 980 to 1179 MPa and elongation (EL) is 15% or higher.
-
- (1) For ferrite and bainitic ferrite structures in the steel sheets, the steel sheets were each subjected to nital etching, and then observed by SEM (with a magnification of 1000x or 2000x), in which ferrite and bainitic ferrite were distinguished to determine their respective fractions (area fractions).
- (2) For the fraction (area fraction) of retained austenite in the steel sheets, the steel sheets were each measured by an X-ray diffraction method, after grinding to one-quarter thicknesses of the steel sheets and subsequent chemical polishing (see, e.g., ISJJ Int. Vol. 33 (1933), No. 7, p. 776).
- (3) For the area fraction of martensite (as-quenched martensite), the steel sheets were each subjected to repera etching, and assuming white contrast as a mixed structure of martensite (as-quenched martensite) and retained austenite by SEM observation, the area fraction of the mixed structure was measured. The fraction of as-quenched martensite was calculated by subtracting the fraction of retained austenite, which had been determined by an X-ray diffraction method, from the area fraction of the mixed structure.
- These results are shown in Table 3 below, together with the types of pre-forming steel sheets (fraction of ferrite, and reduction of cold-rolled steel sheet).
[Table 2] Test No. Steel grade Production conditions Plated or non-plated Steel sheet for forming Heating temperature (°C) Average cooling rate in cooling 1 (°C/sec) Cooling rate changing temperature from cooling 1 to cooling 2 (°C) Average cooling rate in cooling 2 (°C/sec) Retention time at [Bs - 100°C to Ms point] (sec) Forming finishing temperature (°C) Fraction of ferrite (% by area) Reduction (%) 1 A 60 - 800 40 - - 3.2 300 Non-plated 2 B 60 - 800 40 - - 3.0 300 Non-plated 3 C 60 - 800 40 - - 3.1 300 Non-plated 4 D 60 - 800 40 - - 3.3 300 Non-plated 5 E 60 - 800 40 - - 2.9 300 Non-plated 6 F 60 - 800 40 - - 3.0 300 Non-plated 7 G 60 - 800 40 - - 3.3 300 Non-plated 8 H 60 - 800 40 - - 3.0 300 Non-plated 9 I 60 - 800 40 - - 2.9 300 Non-plated 10 J 60 - 800 40 - - 3.0 300 Non-plated 11 K 60 - 800 40 - - 2.7 300 Non-plated 12 L 60 - 800 40 - - 3.2 300 Non-plated 13 M 50 - 800 40 - - 3.3 300 Non-plated 14 N 60 - 800 40 - - 2.9 300 Non-plated 15 C 30 50 800 40 - - 3.1 300 Non-plated 16 C 30 20 800 40 - - 3.1 300 Non-plated 17 C 60 - 720 40 - - 3.1 300 Non-plated 18 C 60 - 900 40 - - 3.1 300 Non-plated 19 C 60 - 900 5 600 40 3.1 300 Non-plated 20 C 30 20 900 5 600 40 3.1 300 Non-plated 21 C 60 - 800 40 450 3 13.3 300 Non-plated 22 C 60 - 900 15 600 40 3.1 300 Non-plated 23 C 60 - 900 5 450 40 3.1 300 Non-plated 24 C 60 - 800 40 - - 3.1 600 Non-plated 25 C 60 - 800 40 - - 3.1 300 Plated [Table 3] Test No. Steel grade Structure of formed product (% by area) Tensile strength TS (MPa) Elongation EL (%) Ferrite Bainitic ferrite Martensite Retained austenite Others* 1 A 41 25 26 8 994 17 2 B 43 29 22 6 1020 16 3 C 48 23 21 8 994 17 4 D 49 23 21 7 1002 17 5 E 49 24 20 7 1023 17 6 F 48 24 21 7 1031 17 7 G 44 25 23 8 1028 17 8 H 46 25 22 7 1011 17 9 I 45 24 23 8 1019 17 10 J 48 24 22 6 1018 17 11 K 49 26 24 1 1022 12 12 L 51 25 22 2 1302 14 13 M 36 28 28 8 1095 16 14 N 45 27 28 0 989 13 15 C 48 24 20 8 1023 17 16 C 25 45 25 5 1082 13 17 C 81 - 15 - θ:4 745 14 18 C - - 95 5 1523 10 19 C 65 8 20 7 984 17 20 C 62 9 22 7 999 17 21 C 43 26 22 9 1032 18 22 C 12 61 20 7 1233 12 23 C 83 17 - - 921 14 24 C 56 20 - - P:24 893 14 25 C 47 23 23 8 994 17 * θ and P indicate cementite and pearlite, respectively. - From these results, discussions can be made as follows: Test Nos. 1 to 10, 13, 15, 19 to 21, and 25 are Examples fulfilling the requirements defined in the present invention, thereby indicating that parts having satisfactory balance between strength and ductility were obtained.
- In contrast, Test Nos. 11 to 12, 14, 16 to 18, and 22 to 24 are Comparative Examples not fulfilling any of the requirements defined in the present invention, thereby deteriorating any of the characteristics. More specifically, Test No. 11 was the case where steel having insufficient C content (steel grade K shown in Table 1) was used, so that retained austenite was not secured, thereby obtaining only low elongation (EL). Test No. 12 was the case where steel having insufficient Si content (steel grade L shown in Table 1), so that retained austenite was not secured, thereby obtaining only low elongation (EL).
- Test No. 14 was intended for conventional 2MnB5 equivalent steel (steel grade N shown in Table 1), so that retained austenite was not secured, thereby obtaining only low elongation (EL), although high strength was obtained. Test No. 16 was the case where cold-rolled steel sheet having low reduction was used, so that the formed product had a structure containing ferrite at 25% by area, thereby lowering elongation (EL). Test No. 17 was the case where the heating temperature was lower than Ac1 transformation point, so that the formed product had a structure containing ferrite at 81% by area (the remainder was martensite and cementite) and retained austenite was not secured, thereby lowering elongation (EL) and tensile strength. Test No. 18 was the case where the heating temperature was higher than A value, so that ferrite and bainitic ferrite were not secured by excessive formation of martensite, thereby lowering elongation (EL).
- Test No. 22 was the case where the average cooling rate in cooling 1 was high, so that ferrite was not secured by the formation of bainitic ferrite, thereby lowering elongation (EL). Test No. 23 was the case where the average cooling rate in cooling 1 was low and the cooling rate changing temperature was low, so that the formed product had a structure containing ferrite at 83% by area (the remainder was bainitic ferrite) and retained austenite was not secured, thereby lowering elongation (EL). Test No. 24 was the case where the forming finishing temperature was high, so that pearlite was formed in the structure of the formed product and retained austenite was not secured, thereby lowing elongation (EL).
- The present invention makes it possible to provide a hot press-formed product, including a steel sheet formed by a hot press-forming method, and having a metallic structure that contains ferrite at 30% to 80% by area, bainitic ferrite at lower than 30% by area (not including 0%), martensite at 30% by area or lower (not including 0%), and retained austenite at 3% to 20% by area, whereby balance between strength and elongation can be controlled in a proper range and high ductility can be achieved.
-
- 1
- Punch
- 2
- Die
- 3
- Blank holder
- 4
- Steel sheet (Blank)
Claims (9)
- A hot press-formed product, comprising a steel sheet formed by a hot press-forming method, and having a metallic structure that contains ferrite at 30% to 80% by area, bainitic ferrite at lower than 30% by area not including 0% by area, martensite at 30% by area or lower not including 0% by area and retained austenite at 3% to 20% by area.
- The hot press-formed product according to claim 1, having the following chemical element composition:C at 0.1% to 0.3% where "%" means "% by mass", and the same applies to the below with respect to the chemical element compositionSi at 0.5% to 3%;Mn at 0.5% to 2%;P at 0.05% or lower not including 0% ;S at 0.05% or lower not including 0% ;Al at 0.01% to 0.1%; andN at 0.001% to 0.01%,and the remainder consisting of iron and unavoidable impurities.
- The hot press-formed product according to claim 2, further comprising, as additional elements, B at 0.01% or lower not including 0% and Ti at 0.1% or lower not including 0%.
- The hot press-formed product according to claim 2 or 3, further comprising, as additional elements, one or more selected from the group consisting of Cu, Ni, Cr, and Mo at 1% or lower not including 0% in total.
- The hot press-formed product according to claim 2 or 3, further comprising, as additional elements, V and/or Nb at 0.1% or lower not including 0% in total.
- A process for producing a hot press-formed product as set forth in claim 1, comprising:heating a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or heating a cold-rolled steel sheet at a reduction of 30% or higher, to a temperature not lower than Ac1 transformation point and not higher than Ac1 transformation point x 0.3 + Ac3 transformation point x 0.7; and thenstarting the forming of the hot-rolled steel sheet or the cold-rolled steel sheet with a press tool to produce the hot press-formed product, during which forming an average cooling rate of 20°C/sec or higher is kept in the press tool, and which forming is finished at a temperature not higher than bainite transformation starting temperature Bs - 100°.
- A process for producing a hot press-formed product as set forth in claim 1, comprising:heating a thin steel sheet to a temperature not lower than Ac3 transformation point and not higher than 1000°C;cooling the thin steel sheet to a temperature not higher than 700°C and not lower than 500°C at an average cooling rate of 10°C/sec or lower; and thenstarting the forming of the thin steel sheet with a press tool to produce the hot press-formed product, during which forming an average cooling rate of 20°C/sec or higher is kept in the press tool, and which forming is finished at a temperature not higher than bainite transformation starting temperature Bs - 100°.
- The process according to claim 6 or 7, wherein the forming finishing temperature is controlled in a temperature range of not higher than bainite transformation starting temperature Bs - 100°C and not lower than martensite transformation starting temperature Ms, in which temperature range the steel sheet is retained for 10 seconds or longer, followed by the forming.
- A thin steel sheet for hot press forming, which is intended for use in producing a hot press-formed product as set forth in claim 1, and which is a hot-rolled steel sheet having a metallic structure that contains ferrite at 50% by area or higher, or a cold-rolled steel sheet at a reduction of 30% or higher.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011130636 | 2011-06-10 | ||
| PCT/JP2012/064850 WO2012169639A1 (en) | 2011-06-10 | 2012-06-08 | Hot press molded article, method for producing same, and thin steel sheet for hot press molding |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP2719787A1 EP2719787A1 (en) | 2014-04-16 |
| EP2719787A4 EP2719787A4 (en) | 2015-08-19 |
| EP2719787B1 true EP2719787B1 (en) | 2016-04-13 |
Family
ID=47296191
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP12796568.9A Revoked EP2719787B1 (en) | 2011-06-10 | 2012-06-08 | Hot press molded article, method for producing same, and thin steel sheet for hot press molding |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20140056753A1 (en) |
| EP (1) | EP2719787B1 (en) |
| JP (1) | JP5883350B2 (en) |
| CN (1) | CN103597106B (en) |
| ES (1) | ES2569190T3 (en) |
| WO (1) | WO2012169639A1 (en) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2803747B1 (en) | 2012-01-13 | 2019-03-27 | Nippon Steel & Sumitomo Metal Corporation | Cold-rolled steel sheet and method for producing cold-rolled steel sheet |
| PL2803748T3 (en) | 2012-01-13 | 2018-08-31 | Nippon Steel & Sumitomo Metal Corporation | Hot stamp molded article, and method for producing hot stamp molded article |
| JP5890710B2 (en) | 2012-03-15 | 2016-03-22 | 株式会社神戸製鋼所 | Hot press-formed product and method for producing the same |
| ES2683149T3 (en) * | 2013-01-18 | 2018-09-25 | Kabushiki Kaisha Kobe Seiko Sho (Kobe Steel, Ltd.) | Manufacturing method for a hot pressed steel member |
| EP2982772B1 (en) | 2013-04-02 | 2018-10-10 | Nippon Steel & Sumitomo Metal Corporation | Hot-stamp-molded article, cold-rolled steel sheet, and method for manufacturing hot-stamp-molded article |
| JP6003837B2 (en) * | 2013-07-25 | 2016-10-05 | Jfeスチール株式会社 | Manufacturing method of high strength pressed parts |
| KR101827188B1 (en) | 2013-09-10 | 2018-02-07 | 가부시키가이샤 고베 세이코쇼 | Hot-pressing steel plate, press-molded article, and method for manufacturing press-molded article |
| KR101827187B1 (en) | 2013-09-10 | 2018-02-07 | 가부시키가이샤 고베 세이코쇼 | Hot-pressing steel plate, press-molded article, and method for manufacturing press-molded article |
| JP5728108B2 (en) * | 2013-09-27 | 2015-06-03 | 株式会社神戸製鋼所 | High-strength steel sheet with excellent workability and low-temperature toughness, and method for producing the same |
| KR101568549B1 (en) | 2013-12-25 | 2015-11-11 | 주식회사 포스코 | Steel sheet for hot press formed product having high bendability and ultra high strength, hot press formed product using the same and method for manufacturing the same |
| US10954578B2 (en) * | 2014-10-30 | 2021-03-23 | Jfe Steel Corporation | High-strength steel sheet and method for manufacturing same |
| JP6372633B1 (en) | 2016-11-16 | 2018-08-15 | Jfeスチール株式会社 | High strength steel plate and manufacturing method thereof |
| WO2018096387A1 (en) * | 2016-11-24 | 2018-05-31 | Arcelormittal | Hot-rolled and coated steel sheet for hot-stamping, hot-stamped coated steel part and methods for manufacturing the same |
| EP3327152B1 (en) * | 2016-11-29 | 2023-10-11 | Tata Steel UK Limited | Method for hot-forming a steel blank |
| MX2019004457A (en) | 2017-01-30 | 2019-06-24 | Nippon Steel & Sumitomo Metal Corp | Steel sheet. |
| JP7214973B2 (en) * | 2018-03-30 | 2023-01-31 | マツダ株式会社 | HOT PRESSING METHOD AND PROCESSING APPARATUS |
| JP7443635B2 (en) * | 2020-01-31 | 2024-03-06 | 株式会社神戸製鋼所 | Galvanized steel sheet for hot stamping, hot stamping parts, and method for manufacturing hot stamping parts |
| CN113584391A (en) * | 2021-08-03 | 2021-11-02 | 武汉科技大学 | 1700 MPa-grade hydrogen-induced delayed cracking resistant hot forming steel and preparation method thereof |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005126733A (en) | 2003-10-21 | 2005-05-19 | Nippon Steel Corp | Steel sheet for hot press having excellent hot workability, and automotive member |
| JP2006183138A (en) | 2004-11-30 | 2006-07-13 | Jfe Steel Kk | Thin steel sheet and its production method |
| JP2007016296A (en) | 2005-07-11 | 2007-01-25 | Nippon Steel Corp | Steel sheet for press forming with excellent ductility after forming, its forming method and automotive parts using the steel sheet for press forming |
| JP2007211276A (en) | 2006-02-08 | 2007-08-23 | Sumitomo Metal Ind Ltd | Plated steel sheet for hot-press use, its manufacturing method, and method for manufacturing hot press formed member |
| EP1939308A1 (en) | 2006-11-14 | 2008-07-02 | ThyssenKrupp Steel AG | Method for manufacturing a component through hot press hardening and highly rigid component with improved breaking strain |
| US20080286603A1 (en) | 2005-12-01 | 2008-11-20 | Posco | Steel Sheet for Hot Press Forming Having Excellent Heat Treatment and Impact Property, Hot Press Parts Made of It and the Method for Manufacturing Thereof |
| US20080308194A1 (en) | 2005-09-21 | 2008-12-18 | Arcelormittal France | Method for Making a Steel Part of Multiphase Microstructure |
| WO2009145563A2 (en) | 2008-05-29 | 2009-12-03 | 주식회사 포스코 | Ultra high strength steel sheet with an excellent heat treatment property for hot press forming, quenched member, and manufacturing method for same |
| US20110036465A1 (en) | 2008-02-08 | 2011-02-17 | Jfe Steel Corporation | High-strength galvanized steel sheet with excellent formability and method for manufacturing the same |
| WO2012048841A1 (en) | 2010-10-12 | 2012-04-19 | Tata Steel Ijmuiden B.V. | Method of hot forming a steel blank and the hot formed part |
| EP2557193A1 (en) | 2010-04-09 | 2013-02-13 | JFE Steel Corporation | High-strength steel sheet having excellent hot rolling workability, and process for production thereof |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5531839A (en) * | 1993-10-05 | 1996-07-02 | Nkk Corporation | Continously annealed cold-rolled steel sheet excellent in balance between deep drawability and resistance to secondary-work embrittlement and method for manufacturing same |
| JPH09143612A (en) * | 1995-11-21 | 1997-06-03 | Kobe Steel Ltd | High strength hot rolled steel plate member low in yield ratio |
| JP3582511B2 (en) * | 2001-10-23 | 2004-10-27 | 住友金属工業株式会社 | Surface-treated steel for hot press forming and its manufacturing method |
| JP4412727B2 (en) * | 2004-01-09 | 2010-02-10 | 株式会社神戸製鋼所 | Super high strength steel sheet with excellent hydrogen embrittlement resistance and method for producing the same |
| JP4673558B2 (en) * | 2004-01-26 | 2011-04-20 | 新日本製鐵株式会社 | Hot press molding method and automotive member excellent in productivity |
| CN100345639C (en) * | 2005-12-21 | 2007-10-31 | 攀钢集团攀枝花钢铁研究院 | Method for manufacturing hot rolled steel plate with ultra-fine grain thin specification and hot rolled steel plate manufactured thereby |
| JP4733522B2 (en) * | 2006-01-06 | 2011-07-27 | 新日本製鐵株式会社 | Method for producing high-strength quenched molded body with excellent corrosion resistance and fatigue resistance |
| JP5151246B2 (en) * | 2007-05-24 | 2013-02-27 | Jfeスチール株式会社 | High-strength cold-rolled steel sheet and high-strength hot-dip galvanized steel sheet excellent in deep drawability and strength-ductility balance and manufacturing method thereof |
| JP5347392B2 (en) * | 2008-09-12 | 2013-11-20 | Jfeスチール株式会社 | Hot press member excellent in ductility, steel plate for hot press member, and method for producing hot press member |
| JP5418168B2 (en) * | 2008-11-28 | 2014-02-19 | Jfeスチール株式会社 | High-strength cold-rolled steel sheet excellent in formability, high-strength hot-dip galvanized steel sheet, and production method thereof |
-
2012
- 2012-06-08 EP EP12796568.9A patent/EP2719787B1/en not_active Revoked
- 2012-06-08 WO PCT/JP2012/064850 patent/WO2012169639A1/en active Application Filing
- 2012-06-08 ES ES12796568.9T patent/ES2569190T3/en active Active
- 2012-06-08 US US14/113,761 patent/US20140056753A1/en not_active Abandoned
- 2012-06-08 JP JP2012131417A patent/JP5883350B2/en not_active Expired - Fee Related
- 2012-06-08 CN CN201280027597.8A patent/CN103597106B/en not_active Expired - Fee Related
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2005126733A (en) | 2003-10-21 | 2005-05-19 | Nippon Steel Corp | Steel sheet for hot press having excellent hot workability, and automotive member |
| JP2006183138A (en) | 2004-11-30 | 2006-07-13 | Jfe Steel Kk | Thin steel sheet and its production method |
| JP2007016296A (en) | 2005-07-11 | 2007-01-25 | Nippon Steel Corp | Steel sheet for press forming with excellent ductility after forming, its forming method and automotive parts using the steel sheet for press forming |
| US20080308194A1 (en) | 2005-09-21 | 2008-12-18 | Arcelormittal France | Method for Making a Steel Part of Multiphase Microstructure |
| US20080286603A1 (en) | 2005-12-01 | 2008-11-20 | Posco | Steel Sheet for Hot Press Forming Having Excellent Heat Treatment and Impact Property, Hot Press Parts Made of It and the Method for Manufacturing Thereof |
| JP2007211276A (en) | 2006-02-08 | 2007-08-23 | Sumitomo Metal Ind Ltd | Plated steel sheet for hot-press use, its manufacturing method, and method for manufacturing hot press formed member |
| EP1939308A1 (en) | 2006-11-14 | 2008-07-02 | ThyssenKrupp Steel AG | Method for manufacturing a component through hot press hardening and highly rigid component with improved breaking strain |
| US20110036465A1 (en) | 2008-02-08 | 2011-02-17 | Jfe Steel Corporation | High-strength galvanized steel sheet with excellent formability and method for manufacturing the same |
| WO2009145563A2 (en) | 2008-05-29 | 2009-12-03 | 주식회사 포스코 | Ultra high strength steel sheet with an excellent heat treatment property for hot press forming, quenched member, and manufacturing method for same |
| EP2557193A1 (en) | 2010-04-09 | 2013-02-13 | JFE Steel Corporation | High-strength steel sheet having excellent hot rolling workability, and process for production thereof |
| WO2012048841A1 (en) | 2010-10-12 | 2012-04-19 | Tata Steel Ijmuiden B.V. | Method of hot forming a steel blank and the hot formed part |
Non-Patent Citations (4)
| Title |
|---|
| "20Mn B5 Warmgewalztes Mittelband Datasheet", THYSSENKRUPP, July 2013 (2013-07-01), XP055340812 |
| "22Mn B5 Warmgewalztes Mittelband Datasheet", THYSSENKRUPP, August 2013 (2013-08-01), XP055340815 |
| "27Mn B5-2 Warmgewalztes Mittelband Datasheet", THYSSENKRUPP, August 2013 (2013-08-01), XP055340819 |
| MALEK NADERI: "Hot Stamping of Ultra High Strength Steels", VON DER FAKULTÄT FÜR GEORESSOURCEN UND MATERIALTECHNIK DER RHEINISCH-WESTFÄLISCHEN TECHNISCHEN HOCHSCHULE AACHEN ZUR ERLANGUNG DES AKADEMISCHEN GRADES EINES DOKTORS DER INGENIEURWISSENSCHAFTEN GENEHMIGTE DISSERTATION, 2008, XP055340807 |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2569190T3 (en) | 2016-05-09 |
| WO2012169639A1 (en) | 2012-12-13 |
| EP2719787A1 (en) | 2014-04-16 |
| EP2719787A4 (en) | 2015-08-19 |
| CN103597106B (en) | 2016-03-02 |
| CN103597106A (en) | 2014-02-19 |
| JP5883350B2 (en) | 2016-03-15 |
| JP2013014841A (en) | 2013-01-24 |
| US20140056753A1 (en) | 2014-02-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2703513B1 (en) | Hot press molded article, fabrication method therefor | |
| EP2719788B1 (en) | Hot press molded article, method for producing same, and thin steel sheet for hot press molding | |
| EP2719787B1 (en) | Hot press molded article, method for producing same, and thin steel sheet for hot press molding | |
| EP2719786B1 (en) | Process for producing a hot press-formed product. | |
| KR101609967B1 (en) | Steel sheet for hot pressing use, press-molded article, and method for producing press-molded article | |
| US9850554B2 (en) | Hot-press formed product and method for manufacturing same | |
| EP3431623A1 (en) | Hot-press formed product and method for manufacturing same | |
| EP3045554B1 (en) | Hot-pressing steel plate, press-molded article, and method for manufacturing press-molded article | |
| KR101609968B1 (en) | Steel sheet for hot pressing use, press-molded article, and method for producing press-molded article | |
| EP2824195A1 (en) | Process for producing press-formed product, and press-formed product | |
| KR101716624B1 (en) | Method for manufacturing press-molded article, and press-molded article | |
| TW201333221A (en) | Hot-rolled steel sheet for nitriding and cold-rolled steel sheet for nitriding with excellent fatigue strength and manufacturing method therefor, as well as automobile parts of excellent fatigue strength using same | |
| JP2004124123A (en) | Low yield ratio type high strength cold rolled steel sheet having excellent workability and shape fixability, and production method therefor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20131031 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| DAX | Request for extension of the european patent (deleted) | ||
| RA4 | Supplementary search report drawn up and despatched (corrected) |
Effective date: 20150717 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: C22C 38/00 20060101AFI20150713BHEP Ipc: B21D 22/20 20060101ALI20150713BHEP Ipc: C21D 9/00 20060101ALI20150713BHEP Ipc: C22C 38/60 20060101ALI20150713BHEP Ipc: C21D 1/18 20060101ALI20150713BHEP |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20151014 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: OKITA, KEISUKE Inventor name: NAITOU, JUNYA Inventor name: MURAKAMI, TOSHIO Inventor name: IKEDA, SHUSHI |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: KABUSHIKI KAISHA KOBE SEIKO SHO |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 790240 Country of ref document: AT Kind code of ref document: T Effective date: 20160415 Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2569190 Country of ref document: ES Kind code of ref document: T3 Effective date: 20160509 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602012017133 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 5 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20160413 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160713 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160816 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160714 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: T3 Ref document number: E 21449 Country of ref document: SK |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R026 Ref document number: 602012017133 Country of ref document: DE |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| 26 | Opposition filed |
Opponent name: SSAB TECHNOLOGY AB Effective date: 20170111 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160630 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160630 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160608 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20120608 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160608 Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 Ref country code: MT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20160630 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20160413 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: UEP Ref document number: 790240 Country of ref document: AT Kind code of ref document: T Effective date: 20160413 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20190528 Year of fee payment: 8 Ref country code: CZ Payment date: 20190517 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20190527 Year of fee payment: 8 Ref country code: FR Payment date: 20190510 Year of fee payment: 8 Ref country code: SE Payment date: 20190611 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SK Payment date: 20190502 Year of fee payment: 8 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20190701 Year of fee payment: 8 Ref country code: GB Payment date: 20190605 Year of fee payment: 8 Ref country code: AT Payment date: 20190528 Year of fee payment: 8 |
|
| RDAF | Communication despatched that patent is revoked |
Free format text: ORIGINAL CODE: EPIDOSNREV1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R064 Ref document number: 602012017133 Country of ref document: DE Ref country code: DE Ref legal event code: R103 Ref document number: 602012017133 Country of ref document: DE |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| 27W | Patent revoked |
Effective date: 20200725 |
|
| GBPR | Gb: patent revoked under art. 102 of the ep convention designating the uk as contracting state |
Effective date: 20200725 |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: MGE |
|
| REG | Reference to a national code |
Ref country code: SK Ref legal event code: MC4A Ref document number: E 21449 Country of ref document: SK Effective date: 20200725 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MA03 Ref document number: 790240 Country of ref document: AT Kind code of ref document: T Effective date: 20200725 |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: ECNC |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200609 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200609 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SK Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200725 |