EP2566445B1 - Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition - Google Patents
Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition Download PDFInfo
- Publication number
- EP2566445B1 EP2566445B1 EP11730359.4A EP11730359A EP2566445B1 EP 2566445 B1 EP2566445 B1 EP 2566445B1 EP 11730359 A EP11730359 A EP 11730359A EP 2566445 B1 EP2566445 B1 EP 2566445B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- equal
- mol
- less
- radical
- formula
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- XGVWAQBZVDLHKR-UHFFFAOYSA-N CCC(CC)(CC)CC1CCCC1 Chemical compound CCC(CC)(CC)CC1CCCC1 XGVWAQBZVDLHKR-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8141—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- A61K8/8158—Homopolymers or copolymers of amides or imides, e.g. (meth) acrylamide; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/345—Alcohols containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/062—Oil-in-water emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/04—Dispersions; Emulsions
- A61K8/06—Emulsions
- A61K8/064—Water-in-oil emulsions, e.g. Water-in-silicone emulsions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/58—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing oxygen in addition to the carbonamido oxygen, e.g. N-methylolacrylamide, N-(meth)acryloylmorpholine
- C08F220/585—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing oxygen in addition to the carbonamido oxygen, e.g. N-methylolacrylamide, N-(meth)acryloylmorpholine and containing other heteroatoms, e.g. 2-acrylamido-2-methylpropane sulfonic acid [AMPS]
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/06—Ethers; Acetals; Ketals; Ortho-esters
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/10—Esters; Ether-esters
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D133/00—Coating compositions based on homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides, or nitriles thereof; Coating compositions based on derivatives of such polymers
- C09D133/24—Homopolymers or copolymers of amides or imides
- C09D133/26—Homopolymers or copolymers of acrylamide or methacrylamide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/54—Polymers characterized by specific structures/properties
- A61K2800/544—Dendrimers, Hyperbranched polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q1/00—Make-up preparations; Body powders; Preparations for removing make-up
- A61Q1/14—Preparations for removing make-up
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/04—Topical preparations for affording protection against sunlight or other radiation; Topical sun tanning preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/002—Aftershave preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/004—Aftersun preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/005—Preparations for sensitive skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/008—Preparations for oily skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/04—Preparations for care of the skin for chemically tanning the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/06—Preparations for care of the skin for countering cellulitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/02—Preparations for cleaning the hair
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/20—Esters of polyhydric alcohols or phenols, e.g. 2-hydroxyethyl (meth)acrylate or glycerol mono-(meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/28—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety
- C08F220/285—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/28—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety
- C08F220/285—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety
- C08F220/286—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety and containing polyethylene oxide in the alcohol moiety, e.g. methoxy polyethylene glycol (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F222/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides, or nitriles thereof
- C08F222/36—Amides or imides
- C08F222/38—Amides
- C08F222/385—Monomers containing two or more (meth)acrylamide groups, e.g. N,N'-methylenebisacrylamide
Definitions
- the present application relates to self-reversing water-in-oil inverse latexes, their method of preparation and their application as a thickener and / or emulsifier for skin care products, scalp and hair or for the manufacture of preparations. cosmetics, dermopharmaceuticals or pharmaceuticals.
- Inverse polyelectrolyte latices including 2-methyl 2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid also known as 2-acrylamido-2-methylpropanesulfonic acid, ATBS or AMPS
- ATBS or AMPS 2-acrylamido-2-methylpropanesulfonic acid
- the inventors have therefore sought to develop reverse latices that do not have the aforementioned drawbacks.
- the radical R 1 represents a lauryl radical or a stearyl radical.
- the strong acid function of the monomers containing it is in particular the sulphonic acid function.
- Said monomers are, for example, partially or totally salified styrenesulphonic acid or 2-methyl-2 - [(1-oxo-2-propenyl) amino] 1-propanesulphonic acid (also known as 2-acrylamido-2-methyl propanesulfonic acid). or totally salified.
- the weak acid function of the monomers comprising them is in particular the partially salified carboxylic acid function.
- Said monomers are for example acrylic acid, methacrylic acid, itaconic acid, maleic acid or 3-methyl-3 - [(1-oxo-2-propenyl) amino] butanoic acid partially or totally salified. It is more particularly acrylic acid or partially salified methacrylic acid.
- the term salified indicates that it is alkali metal salts such as sodium or potassium salts, nitrogenous base salts such as, or the salt of ammonium.
- the invention also more particularly relates to a composition as defined above, for which the polyelectrolyte P comprises, in molar percentage, from 0.5% to 10% of a monomeric unit derived from the monomer of formula (I) such that previously defined.
- the emulsifier system (S 1 ) of water-in-oil (W / O) type consists of either a single surfactant or a mixture of surfactants provided that said surfactant or said mixture has a low enough HLB value to induce water-in-oil emulsions.
- sorbitan esters such as sorbitan oleate, such as that marketed by the company SEPPIC under the name MONTANE TM 80
- sorbitan isostearate such as that marketed by the company SEPPIC under the name MONTANE TM 70 or sorbitan sesquioleate such as that marketed by the company SEPPIC under the name Montane TM 83.
- polyethoxylated sorbitan esters for example pentaethoxylated sorbitan mono-oleate such as that marketed by the company SEPPIC under the name MONTANOX TM 81 or pentaethoxylated sorbitan isostearate such as that sold under the name MONTANOX TM 71 by the company SEPPIC.
- oleocetyl alcohol diethoxylated such as that marketed under the name SIMULSOL TM OC 72 by the company SEPPIC
- polyesters with a molecular weight between 1000 and 3000 products of condensation between a poly (isobutenyl) succinic acid or its anhydride and such as HYPERMER TM 2296 marketed by the company UNIQEMA or finally block copolymers of molecular weight between 2500 and 3500, such as HYPERMER TM B246 marketed by the company UNIQEMA or SIMALINE TM IE 200 marketed by the company SEPPIC.
- the oil-in-water (O / W) emulsifier system (S 2 ) comprises at least a non-zero amount of the surfactant composition (C) as defined above.
- non-zero amount more particularly denotes a proportion greater than or equal to 10% by mass and, more particularly greater than or equal to 50% by mass.
- said oil-in-water (O / W) emulsifier system (S 2 ) consists of 100% by weight of the surfactant composition (C) as defined above.
- the emulsifier system (S 2 ) of oil-in-water (O / W) type further comprises at least one emulsifying surfactant of the (O / W) type different from one or the other. other compounds constituting said surfactant composition (C) as defined above.
- the emulsifier system (S 2 ) of oil-in-water (O / W) type further comprises a non-zero mass proportion of at least one emulsifying agent of the chosen oil-in-water type. from polyethoxylated sorbitan oleate with 20 moles of ethylene oxide; polyethoxylated sorbitan laurate with 20 moles of ethylene oxide; polyethoxylated castor oil with 40 moles of ethylene oxide; decaethoxylated oleodecyl alcohol, heptaethoxylated lauric alcohol; or polyethoxylated sorbitan hexaoleate.
- crosslinked polyelectrolyte for (P), a nonlinear polyelectrolyte in the state of three-dimensional network insoluble in water, but swellable with water and thus leading to the production of a chemical gel.
- composition according to the invention may comprise crosslinked polyelectrolytes and / or branched polyelectrolytes.
- the polyelectrolyte (P) When the polyelectrolyte (P) is crosslinked, it is more particularly with a diethylenic or polyethylenic compound in a molar proportion expressed relative to the monomers used depending on the chemical nature of the crosslinking agent and generally less than or equal to 0.40 mol% , mainly less than 0.25 mol%, more particularly less than or equal to 0.05 mol% and very particularly between 0.005% and 0.01 mol%.
- the crosslinking agent and / or the branching agent is chosen from ethylene glycol dimethacrylate, diethylene glycol diacrylate and sodium diallyloxyacetate. ethylene glycol diacrylate, diallyl urea, triallylamine, trimethylol propanetriacrylate, methylenebis (acrylamide) or a mixture of these compounds.
- the invention more particularly relates to a composition as defined above, for which in the polyelectrolyte (P), the monomeric unit derived from the monomer of formula (I) is a monomeric unit derived from tetraethoxylated lauryl acrylate.
- the oil phase is constituted either by a commercial mineral oil containing saturated hydrocarbons such as paraffins, isoparaffins or cycloparaffins having, at ambient temperature, a density between 0.7 and 0.9 and a boiling point greater than about 250 ° C, such as for example MARCOL TM 52 or MARCOL TM 82, marketed by EXXON CHEMICAL, or by a vegetable oil such as squalane of vegetable origin, or by a synthetic oil such as hydrogenated polyisobutene or hydrogenated polydecene, or a mixture of several of these oils.
- MARCOL TM 52 is a commercial oil that meets the definition of vaseline oils from the French Codex.
- composition according to the invention may also contain various additives such as complexing agents, transfer agents or chain-limiting agents.
- the invention also relates to a topical cosmetic, dermopharmaceutical or pharmaceutical composition, characterized in that it comprises, as thickening and / or emulsifying agent, an effective amount of the composition as defined above.
- a topical composition according to the invention intended to be applied to the skin, the scalp or the mucous membranes of humans or animals, may consist of a topical emulsion comprising at least one aqueous phase and at least one phase oil, in the form of a water-in-oil emulsion, or oil in water, or water in oil in water, or oil in water in oil. More particularly, this topical emulsion may consist of a fluid emulsion, such as a milk or a fluid gel.
- the oil phase of the topical emulsion can consist of a mixture of one or more oils.
- the topical composition according to the invention comprises a sufficient amount of inverse latex according to the invention to cause a change in its rheology.
- the topical composition according to the invention usually comprises between 0.1% and 10% by weight of said self-invertible inverse latex defined above.
- the pH of the topical composition is generally between 3 and 9.
- the topical composition may further comprise compounds conventionally included in this type of composition, for example perfumes, preservatives, dyes, emollients or surfactants.
- the invention relates to the use of the novel thickening and / or emulsifying agent according to the invention, mentioned above, for thickening and emulsifying a topical composition comprising at least one aqueous phase.
- composition according to the invention is an interesting substitute to those sold under the names SEPIGEL TM 305, SEPIGEL TM 501, SIMULGEL TM EG, SIMULGEL TM NS or SIMUL-GEL TM 600 by the applicant, since it also has good compatibility with the other excipients used for the preparation of formulations such as milks, lotions, creams, soaps, baths, balms, shampoos or conditioners. It can also be implemented with said SEPIGEL or SIMULGEL.
- aqueous dispersions with a selected organopolysiloxane compound, for example from those described in US Pat. WO 93/05762 or in WO 93/21316 . It can also be used to form aqueous gels with cosmetically or physiologically acceptable acidic pH, such as those described in US Pat. WO 93/07856 ; it can also be used in combination with nonionic celluloses, for example to form styling gels such as those described in US Pat.
- EP 0 684 024 or in association with esters of fatty acids and sugar, to form compositions for the treatment of hair or skin such as those described in EP 0 603 019 or in shampoos or conditioners as described and claimed in WO 92/21316 or finally in combination with an anionic homopolymer such as CARBOPOL TM to form hair treatment products such as those described in DE 195 23596 or in combination with other thickening polymers.
- an anionic homopolymer such as CARBOPOL TM to form hair treatment products such as those described in DE 195 23596 or in combination with other thickening polymers.
- composition according to the invention is also compatible with the active principles such as, for example, self-tanning agents such as dihydroxyacetone (DHA) or anti-acne agents; it can therefore be introduced into self-tanning compositions such as those claimed in EP0 715 845 , EP 0 604 249 , EP 0 576 188 or in 93/07902 .
- self-tanning agents such as dihydroxyacetone (DHA) or anti-acne agents
- composition as defined above is intended for the treatment of the skin and / or the scalp and / or the mucous membranes, it more particularly comprises an inverse anionic polyelectrolyte latex object of the present invention.
- the inverse latexes of the present invention can be used as a thickener for textile printing pastes.
- EXAMPLE 1 Self-invertible inverse latex of the copolymer ATBS (Na salt) / HEA / (ALE-4 EO) [(ATBS / HEA / (ALE-4OE) 89.0 / 9.9 / 1.1 molar] cross-linked to MBA
- step f) 4% by weight of MONTANOX TM 20 is added in place of 4% by weight of the surfactant composition (C) and the self-invertible inverse latex (T1).
- EXAMPLE 2 (according to the invention) : Self-invertible inverse latex of the copolymer ATBS (Na salt) / HEA / (ALE-4OE) [(ATBS / HEA / (ALE-4OE) 89.0 / 9.9 / 1.1 molar] cross-linked to MBA
- step f) only 4% by weight of the surfactant composition (C) is added and the self-invertible inverse latex (2) is obtained. ).
- EXAMPLE 3 (According to the Invention): Self-Invertible Reverse Latex of the Copolymer AM / AA / (ALE-4OE) [(AM / AA / (Alfa-4OE) 24.7 / 74.1 / 1.2 molar] Cross-linked to MBA
- EXAMPLE 4 (according to the invention) : Self-invertible inverse latex of the copolymer ATBS (Na salt) / AA / HEA / THAM / (ALE-4OE) [ATBS / AA / HEA / THAM (ALE-4OE) 83.9 / 1.9 / 9.3 / 3.7 / 1.2 molar] cross-linked to the MBA
- Cyclomethicone 10% Self-reversing inverse latex (1): 0.8% MONTANOV TM 68: 4.5% Conservative: 0.65% Lysine: 0.025% EDTA (disodium salt): 0.05% Xanthan gum: 0.2% Glycerin: 3% Water: qsp 100%
- Cyclomethicone 10% Self-invertible inverse latex (3): 0.8% MONTANOV TM 68: 4.5% Perfluoropolymethylisopropyl ether: 0.5% Conservative: 0.65% Lysine: 0.025% EDTA (disodium salt): PEMULEN TM TR: 0.05% Glycerin: 3% Water: qsq 100%
- Example 18 moisturizing cream for oily skin
- Example 20 Cream with AHA for sensitive skin
- Example 33 Non-greasy tanning for face and body
- Example 40 Cream mask "rinse off” restructuring for stressed and weakened hair
- Self-invertible inverse latex (3) 4% Vegetal squalane: 5% Dimethicone: 1.5% SEPICONTROL TM A5: 4% Xanthan gum : 0.3% Water: qsp.100% Conservative, Perfume: qs.
- SIMULSOL TM 1293 is hydrogenated and polyethoxylated castor oil, with an ethoxylation index equal to 40, sold by the company SEPPIC.
- CAPIGEL TM 98 is a liquid thickener based on acrylate copolymer marketed by the company SEPPIC.
- KETROL TM T is xanthan gum marketed by the company KELCO.
- LANOL TM 99 is isononyl isononanoate marketed by the company SEPPIC.
- DC1501 is a mixture of cyclopentasiloxane and dimethiconol marketed by Dow Chemical.
- MONTANOV TM 82 is an emulsifier based on cetearyl alcohol and cocoylglucos ide.
- MONTANOV TM 68 cetearyl glucoside
- the MICROPEARL TM M 100 is an ultra-fine powder with a very soft touch and mattifying action marketed by MATSUMO.
- SEPICIDE TM CI imidazolidine urea, is a preservative marketed by the company SEPPIC.
- PEMULEN TM TR is an acrylic polymer marketed by GOODRICH.
- SIMULSOL TM 165 is self-emulsifiable glycerol stearate marketed by the company SEPPIC.
- LANOL TM 1688 is an emollient non-fat ester marketed by the company SEPPIC.
- LANOL TM 14M and LANOL S are consistency factors marketed by the company SEPPIC.
- SEPICIDE TM HB which is a mixture of phenoxy ethanol, methyl paraben, ethyl paraben, propyl paraben and butyl paraben, is a preservative marketed by the company SEPPIC.
- MONTEINE TM CA is a moisturizing agent marketed by the company SEPPIC.
- SCHERCEMOL TM OP is a non-greasy emollient ester.
- LANOL TM P is a stabilizing additive marketed by the company SEPPIC.
- PARSOL TM MCX is octyl (para-methoxy) cinnamate; marketed by GIVAUDAN.
- SEPIPERL TM N is a pearlescent agent, sold by the company SEPPIC, based on a mixture of alkyl polyglucosides such as those described in US Pat. WO 95/13863 .
- MICROPEARL TM SQL is a mixture of micro particles containing squalane which is released by the action of massage; it is marketed by MATSUMO.
- LANOL TM 99 is isononyl isononanoate marketed by the company SEPPIC.
- LANOL TM 37T is glycerol triheptanoate, marketed by the company SEPPIC.
- SOLAGUM TM L is a carrageenan marketed by the company SEPPIC.
- MARCOL TM 82 is a paraffin oil marketed by the company EXXON.
- LA-NOL TM 84D is dioctyl malate marketed by the company SEPPIC.
- PARSOL NOX TM is a sunscreen marketed by the company GIVAUDAN.
- EUSOLEX TM 4360 is a solar filter marketed by MERCK.
- DOW CORNING TM 245 Fluid is cyclomethicone sold by the company Dow Corning.
- LIPACIDE TM PVB is a hydrolyzate of acyl wheat proteins marketed by the company SEPPIC.
- MICROPEARL TM LM is a mixture of squalane, polymethylmethacrylate and menthol, marketed by the company SEPPIC.
- SEPICONTROL TM A5 is a mixture of capryloyl glycine, sarcosine and cinnamon zylanicum extract marketed by the company SEPPIC, such as those described in the international patent application. PCT / FR98 / 01313 filed on June 23, 1998 .
- LANOL TM 2681 is a caprylate mixture, copra caprate, marketed by the company SEPPIC.
- MONTANOV TM 202 is an APG / fatty alcohol composition as described in WO998 / 47610 marketed by the company SEPPIC.
Description
La présente demande concerne des latex inverse eau dans huile auto-inversibles, leur procédé de préparation et leur application en tant qu'épaississant et/ou émulsionnant de produits de soins de la peau, du cuir chevelu et des cheveux ou pour la fabrication de préparations cosmétiques, dermopharmaceutiques ou pharmaceutiques.The present application relates to self-reversing water-in-oil inverse latexes, their method of preparation and their application as a thickener and / or emulsifier for skin care products, scalp and hair or for the manufacture of preparations. cosmetics, dermopharmaceuticals or pharmaceuticals.
Les latex inverses à base de polyélectrolytes dont l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino]1-propanesulfonique (dénommé aussi acide 2-acrylamido 2-méthyl propanesulfonique, ATBS ou AMPS) partiellement ou totalement salifié, ainsi que leur utilisation en cosmétique et/ou pharmacie ont fait l'objet de nombreuses demandes de brevet. Cependant, la présence de quantités importantes d'eau et d'huile dans ces latex inverses représente un inconvénient non négligeable en termes de volume, de coût et parfois de risques accrus et/ou d'effets toxiques.Inverse polyelectrolyte latices including 2-methyl 2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid (also known as 2-acrylamido-2-methylpropanesulfonic acid, ATBS or AMPS) partially or totally salified , as well as their use in cosmetics and / or pharmacy have been the subject of many patent applications. However, the presence of significant amounts of water and oil in these inverse latexes represents a significant disadvantage in terms of volume, cost and sometimes increased risks and / or toxic effects.
Des solutions ont donc été développées pour augmenter la concentration en polyélectrolytes dans les latex inverses finaux par exemple en soumettant le milieu réactionnel, en fin de polymérisation, à une étape de distillation sous vide pour enlever une partie plus ou moins importante d'eau et d'huile. Cette distillation est cependant délicate à mettre en oeuvre car elle induit souvent une déstabilisation du latex inverse qu'il faut contrer par l'addition préalable d'agents stabilisants.Solutions have therefore been developed for increasing the concentration of polyelectrolytes in the final inverse latices, for example by subjecting the reaction medium, at the end of the polymerization, to a vacuum distillation stage to remove a greater or lesser portion of water and water. 'oil. This distillation is however difficult to implement because it often induces a destabilization of the inverse latex which must be countered by the prior addition of stabilizing agents.
Les demandes de brevet européen
L'inconvénient de ces polymères stabilisants est qu'ils contiennent des alcools ou des glycols pouvant induire des problèmes environnementaux. De plus il se produit parfois une prise en masse du milieu réactionnel lors de l'étape de distillation, sans que ce phénomène n'ait jamais vraiment été expliqué, mais dont la conséquence certaine est la destruction du lot de latex inverse en cours de fabrication et un nettoyage pénible et coûteux du réacteur utilisé. Enfin, même quand la distillation se déroule correctement, les latex inverses obtenus s'inversent souvent difficilement lors de leur mise en oeuvre dans une phase aqueuse. Ils présentent aussi une viscosité élevée et parfois en leur sein des micro-gels. Ces inconvénients interdisent donc leur utilisation dans la fabrication de formulations cosmétiques. Pour pallier à ces inconvénients, les inventeurs ont développé un latex inverse divulgué dans la demande de brevet français publiée sous le numéro
C(R1)(R3)=C(R2)-C(=O)-O-(CH2-CH2-O)n-R4 (A)
dans laquelle les radicaux R1, R2 et R3, identiques ou différents représentent indépendamment les uns des autres un atome d'hydrogène ou un radical alkyle linéaire ou ramifié comportant de 1 à 4 atomes de carbone, le radical R4 représente un radical aliphatique linéaire ou ramifié, saturé ou insaturé, comportant de 6 à 30 atomes de carbone et n représente un nombre compris entre 1 et 50.The disadvantage of these stabilizing polymers is that they contain alcohols or glycols which can induce environmental problems. In addition, it sometimes occurs that the reaction medium becomes solid during the distillation step, without this phenomenon having really been explained, but the undeniable consequence of which is the destruction of the batch of inverse latex during manufacture. and a painful and expensive cleaning of the reactor used. Finally, even when the distillation proceeds correctly, the inverse latexes obtained are often inverted with difficulty when they are used in an aqueous phase. They also have a high viscosity and sometimes within them micro-gels. These disadvantages therefore prohibit their use in the manufacture of cosmetic formulations. To overcome these drawbacks, the inventors have developed an inverse latex disclosed in the French patent application published under the number
C (R 1 ) (R 3 ) = C (R 2 ) -C (= O) -O- (CH 2 -CH 2 -O) n -R 4 (A)
in which the radicals R 1 , R 2 and R 3 , which are identical or different, represent, independently of each other, a hydrogen atom or a linear or branched alkyl radical containing from 1 to 4 carbon atoms, the R 4 radical represents an aliphatic radical; linear or branched, saturated or unsaturated, having from 6 to 30 carbon atoms and n represents a number inclusive between 1 and 50.
Cependant, lorsque l'on met en oeuvre ce latex inverse pour préparer une formulation épaissie, sa vitesse d'inversion dans la phase aqueuse, c'est-à-dire le temps nécessaire pour obtenir le développement maximal de la viscosité, reste assez faible ce qui signifie pour l'utilisateur une perte de temps qui est dommageable en phase industrielle. En effet il est bien connu que le temps d'inversion des latex inverses augmente considérablement en fonction de l'échelle d'utilisation. Par ailleurs la stabilité dans le temps des latex inverses décrits dans
Les inventeurs ont donc cherché à développer des latex inverses qui ne présentent pas les inconvénients précités.The inventors have therefore sought to develop reverse latices that do not have the aforementioned drawbacks.
Selon un premier aspect, l'invention a pour objet une composition sous forme d'un latex inverse auto-inversible comprenant pour 100% de sa masse :
- a) - De 50% massique à 70% massique d'un polyélectrolyte anionique (P) réticulé, obtenu par polymérisation :
- D'au moins un monomère neutre de formule (I) :
- D'au moins un monomère neutre choisi parmi l'acrylamide, le N,N-diméthyl acrylamide, le N-[2-hydroxy-1,1-bis(hydroxyméthyl)éthyl] propènamide [ou tris(hydroxyméthyl) acrylamidométhane ou N-[tris(hydroxyméthyl) méthyl] acrylamide dénommé aussi THAM] ou l'acrylate de (2-hydroxy éthyle) ; et
- D'au moins un monomère comportant une fonction acide fort et/ou d'au moins un monomère comportant une fonction acide faible ;
- D'au moins un monomère neutre de formule (I) :
- b) - De 4% massique à 10% massique d'un système émulsionnant (S1) de type eau dans huile (E/H) ;
- c) - De 1% massique à 10% massique d'un système émulsionnant (S2) de type huile dans eau (H/E) comprenant une proportion massique non nulle d'une composition tensioactive (C), ladite composition tensioactive (C) comprenant pour 100% molaire :
- 1) - Une proportion supérieure ou égale à 10% molaire et inférieure ou égale à 50% molaire d'une composition (CII) comprenant pour 100% molaire :
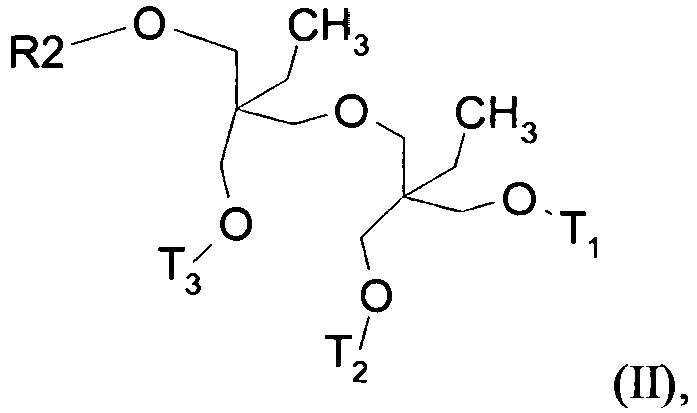
- α) - De 60% molaire à 100% molaire d'un composé de formule (II) :
- R2 représente un radical alkyle linéaire ou ramifié, comportant de 12 atomes de carbone,
- T1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m1-H dans lequel m1 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T2 identique ou différent de T1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m2-H dans lequel m2 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- T3 identique ou différent de T1 et de T2, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m3-H dans lequel m3 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- étant entendu que la somme m1 + m2 + m3 est supérieure à 0 et inférieure ou égale à dix ; β) - Optionnellement jusqu'à 40% molaire d'un composé de formule (II') :
- R'2 représente un radical alkyle linéaire ou ramifié, comportant 14 atomes de carbone,
- T'1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m1-H dans lequel m1 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T'2 identique ou différent de T'1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m2-H dans lequel m2 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- T'3 identique ou différent de T'1 et de T'2, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m3-H dans lequel m3 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- étant entendu, que la somme m1 + m2 + m3 est supérieure à 0 et inférieure ou égale à dix ; et γ) - Optionnellement jusqu'à 10% molaire d'un composé de formule (II") :
- R"2 représente un radical alkyle linéaire ou ramifié, comportant 16 atomes de carbone,
- T"1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m1-H dans lequel m1 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T"2 identique ou différent de T"1, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m2-H dans lequel m2 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- T"3 identique ou différent de T"1 et de T"2, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m3-H dans lequel m3 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- étant entendu que la somme m1 + m2 + m3 est supérieure à 0 et inférieure ou égale à dix ;
- α) - De 60% molaire à 100% molaire d'un composé de formule (II) :
- 2) - Une proportion supérieure ou égale à 50% molaire et inférieure ou égale à 90% molaire d'une composition (CIII) comprenant pour 100% molaire :
- α) - De 60% molaire à 100% molaire d'un composé de formule (III) :
formules (III) et (IV) dans lesquelles :- R2 représente un radical alkyle linéaire ou ramifié, comportant 12 atomes de carbone,
- T4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m4-H dans lequel m4 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T5 identique ou différent de T4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m5-H dans lequel m5 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T6 identique ou différent de T4 et de T5 représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m6-H dans lequel m6 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T7 identique ou différent de T4 de T5 et de T6, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m7-H dans lequel m7 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix étant entendu que la somme m4 + m5 + m6 + m7 est supérieure à 0 et inférieure ou égale à dix ;
- β) - Optionnellement jusqu'à 40% molaire composé de formule (III') :
formules (III') et (IV') dans lesquelles :- R'2 représente un radical alkyle linéaire ou ramifié, comportant 14 atomes de carbone,
- T'4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m4-H dans lequel m4 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T'5 identique ou différent de T'4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m5-H dans lequel m5 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T'6 identique ou différent de T'4 et de T'5, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m6-H dans lequel m6 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- T'7 identique ou différent de T'4, de T'5 et de T'6, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m7-H dans lequel m7 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- étant entendu que la somme m4 + m5 + m6 + m7 est supérieure à 0 et inférieure ou égale à dix ; et
- γ) - Optionnellement jusqu'à 10% molaire d'un composé de formule (III") :
formules (III") et (IV") dans lesquelles :- R"2 représente un radical alkyle linéaire ou ramifié, comportant 16 atomes de carbone,
- T"4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m4-H dans lequel m4 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T"5 identique ou différent de T"4, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m5-H dans lequel m5 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T"6 identique ou différent de T"4 et de T"5 représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m6-H dans lequel m6 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- T"7 identique ou différent de T"4, de T"5 et de T"6, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m7-H dans lequel m7 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- étant entendu que la somme m4 + m5 + m6 + m7 est supérieure à 0 et inférieure ou égale à dix ;
- d) - De 15% massique à 45% massique d'au moins une huile, et
- e) - De 0% massique à 5% massique d'eau.
- α) - De 60% molaire à 100% molaire d'un composé de formule (III) :
- 1) - Une proportion supérieure ou égale à 10% molaire et inférieure ou égale à 50% molaire d'une composition (CII) comprenant pour 100% molaire :
- a) - from 50% by weight to 70% by weight of a crosslinked anionic polyelectrolyte (P) obtained by polymerization:
- Of at least one neutral monomer of formula (I):
- At least one neutral monomer selected from acrylamide, N, N-dimethylacrylamide, N- [2-hydroxy-1,1-bis (hydroxymethyl) ethyl] propenamide [or tris (hydroxymethyl) acrylamidomethane or N- [tris (hydroxymethyl) methyl] acrylamide also referred to as THAM] or (2-hydroxyethyl acrylate); and
- At least one monomer comprising a strong acid function and / or at least one monomer comprising a weak acid function;
- Of at least one neutral monomer of formula (I):
- b) - from 4% by weight to 10% by weight of an emulsifier system (S 1 ) of water-in-oil (W / O) type;
- c) - from 1 wt% to 10 wt% of an oil-in-water (O / W) emulsifier system (S 2 ) comprising a non-zero mass proportion of a surfactant composition (C), said surfactant composition (C) ) comprising for 100 mol%:
- 1) - a proportion greater than or equal to 10 mol% and less than or equal to 50 mol% of a composition (C II ) comprising for 100 mol%:
- α) - from 60 mol% to 100 mol% of a compound of formula (II):
- R2 represents a linear or branched alkyl radical containing 12 carbon atoms,
- T 1 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 1 -H in which m 1 is an integer greater than or equal to zero and less than or equal to ten,
- T 2 identical or different from T 1 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m2 -H in which m2 is an integer greater than or equal to zero and less than or equal to ten , and
- T 3 identical or different from T 1 and T 2 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m3 -H in which m3 is an integer greater than or equal to zero and lower or equal to ten,
- it being understood that the sum m1 + m2 + m3 is greater than 0 and less than or equal to ten; β ) - Optionally up to 40 mol% of a compound of formula (II '):
- R ' 2 represents a linear or branched alkyl radical containing 14 carbon atoms,
- T ' 1 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 1 -H in which m 1 is an integer greater than or equal to zero and less than or equal to ten,
- T ' 2 identical or different from T' 1 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m2 -H in which m2 is an integer greater than or equal to zero and less than or equal to to ten, and
- T ' 3 identical or different from T' 1 and T ' 2 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m3 -H in which m3 is an integer greater than or equal to zero and less than or equal to ten,
- it being understood that the sum m1 + m2 + m3 is greater than 0 and less than or equal to ten; and γ) - optionally up to 10 mol% of a compound of formula (II "):
- R " 2 represents a linear or branched alkyl radical containing 16 carbon atoms,
- T " 1 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 1 -H in which m 1 is an integer greater than or equal to zero and less than or equal to ten,
- T " 2 identical or different from T" 1 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m2 -H in which m2 is an integer greater than or equal to zero and less than or equal to to ten, and
- T " 3 identical or different from T" 1 and T " 2 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m3 -H in which m3 is an integer greater than or equal to zero and less than or equal to ten,
- it being understood that the sum m1 + m2 + m3 is greater than 0 and less than or equal to ten;
- α) - from 60 mol% to 100 mol% of a compound of formula (II):
- 2) - a proportion greater than or equal to 50 mol% and less than or equal to 90 mol% of a composition (C III ) comprising for 100 mol%:
- α) - from 60 mol% to 100 mol% of a compound of formula (III):
formulas (III) and (IV) in which:- R2 represents a linear or branched alkyl radical containing 12 carbon atoms,
- T 4 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 4 -H in which m 4 is an integer greater than or equal to zero and less than or equal to ten,
- T 5 identical or different from T 4 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 5 -H in which m 5 is an integer greater than or equal to zero and less than or equal to ten ,
- T 6 identical or different from T 4 and T 5 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 6 -H in which m 6 is an integer greater than or equal to zero and lower or equal to ten,
- T 7 identical or different from T 4 of T 5 and T 6 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m7 -H wherein m7 is an integer greater than or equal to zero and less than or equal to ten, provided that the sum m4 + m5 + m6 + m7 is greater than 0 and less or equal to ten;
- β) - Optionally up to 40% molar compound of formula (III '):
formulas (III ') and (IV') in which:- R ' 2 represents a linear or branched alkyl radical containing 14 carbon atoms,
- T ' 4 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 4 -H in which m 4 is an integer greater than or equal to zero and less than or equal to ten,
- T ' 5 identical or different from T' 4 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 5 -H in which m 5 is an integer greater than or equal to zero and less than or equal to to ten,
- T ' 6 identical or different from T' 4 and T ' 5 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 6 -H in which m 6 is an integer greater than or equal to zero and less than or equal to ten, and
- T ' 7 identical or different from T' 4 , T ' 5 and T' 6 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m7 -H in which m7 is a number integer greater than or equal to zero and less than or equal to ten,
- it being understood that the sum m4 + m5 + m6 + m7 is greater than 0 and less than or equal to ten; and
- γ) - optionally up to 10 mol% of a compound of formula (III "):
formulas (III ") and (IV") in which:- R " 2 represents a linear or branched alkyl radical containing 16 carbon atoms,
- T " 4 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 4 -H in which m 4 is an integer greater than or equal to zero and less than or equal to ten,
- T " 5 identical or different from T" 4 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m5 -H wherein m5 is an integer greater than or equal to zero and less than or equal to to ten,
- T " 6 identical or different from T" 4 and T " 5 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 6 -H in which m 6 is an integer greater than or equal to zero and less than or equal to ten, and
- T " 7 identical or different from T" 4 , T " 5 and T" 6 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m7 -H in which m7 is a number integer greater than or equal to zero and less than or equal to ten,
- it being understood that the sum m4 + m5 + m6 + m7 is greater than 0 and less than or equal to ten;
- d) - from 15% by weight to 45% by weight of at least one oil, and
- e) - From 0% by weight to 5% by weight of water.
- α) - from 60 mol% to 100 mol% of a compound of formula (III):
- 1) - a proportion greater than or equal to 10 mol% and less than or equal to 50 mol% of a composition (C II ) comprising for 100 mol%:
Par radical aliphatique linéaire ou ramifié, saturé ou insaturé, comportant de 6 à 20 atomes de carbone, on désigne pour le radical R1 dans la formule (I) telle que définie ci-dessus, plus particulièrement les radicaux linéaires tels que, par exemple, les radicaux, hexyle, octyle, nonyle, décyle, undécyle, dodécyle, tétradécyle, hexadécyle, octadécyle ou eicosyleBy linear or branched, saturated or unsaturated aliphatic radical containing from 6 to 20 carbon atoms, for the radical R 1 in the formula (I) as defined above, is meant more particularly linear radicals such as, for example, radicals, hexyl, octyl, nonyl, decyl, undecyl, dodecyl, tetradecyl, hexadecyl, octadecyl or eicosyl
Selon un premier aspect particulier, dans la formule (I) telle que définie ci-dessus, le radical R1 représente un radical lauryle ou un radical stéaryle.According to a first particular aspect, in the formula (I) as defined above, the radical R 1 represents a lauryl radical or a stearyl radical.
Selon un autre aspect particulier, dans la formule (I) telle que définie ci-dessus, n est supérieur ou égal à deux et inférieur ou égal à vingt.According to another particular aspect, in the formula (I) as defined above, n is greater than or equal to two and less than or equal to twenty.
Selon un autre aspect particulier, dans le polyélectrolyte P de la composition objet de la présente invention, la fonction acide fort des monomères en comportant est notamment la fonction acide sulfonique. Lesdits monomères sont par exemple l'acide styrènesulfonique partiellement ou totalement salifié ou, l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique (dénommé aussi acide 2-acrylamido 2-méthyl propanesulfonique) partiellement ou totalement salifiée.According to another particular aspect, in the polyelectrolyte P of the composition which is the subject of the present invention, the strong acid function of the monomers containing it is in particular the sulphonic acid function. Said monomers are, for example, partially or totally salified styrenesulphonic acid or 2-methyl-2 - [(1-oxo-2-propenyl) amino] 1-propanesulphonic acid (also known as 2-acrylamido-2-methyl propanesulfonic acid). or totally salified.
Selon un autre aspect particulier, dans le polyélectrolyte P compris dans la composition objet de la présente invention, la fonction acide faible des monomères en comportant est notamment la fonction acide carboxylique partiellement salifiée. Lesdits monomères sont par exemple l'acide acrylique, l'acide méthacrylique, l'acide itaconique, l'acide maléique ou l'acide 3-méthyl 3-[(1-oxo 2-propènyl) amino] butanoïque partiellement ou totalement salifié. Il s'agit plus particulièrement de l'acide acrylique ou de l'acide méthacrylique partiellement salifié.According to another particular aspect, in the polyelectrolyte P included in the composition which is the subject of the present invention, the weak acid function of the monomers comprising them is in particular the partially salified carboxylic acid function. Said monomers are for example acrylic acid, methacrylic acid, itaconic acid, maleic acid or 3-methyl-3 - [(1-oxo-2-propenyl) amino] butanoic acid partially or totally salified. It is more particularly acrylic acid or partially salified methacrylic acid.
Pour les monomères à fonction acide fort ou à fonction acide faible, le terme salifié indique qu'il s'agit de sels de métaux alcalins tels que les sels de sodium ou de potassium, les sels de bases azotées comme, ou le sel d'ammonium.For the monomers with strong acid function or low acid function, the term salified indicates that it is alkali metal salts such as sodium or potassium salts, nitrogenous base salts such as, or the salt of ammonium.
L'invention a plus particulièrement pour objet, une composition telle que définie précédemment, pour laquelle dans le polyélectrolyte (P), les unités monomériques comportant une fonction acide fort sont issues de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique partiellement ou totalement salifié en sel de sodium, en sel de potassium, ou en sel d'ammonium et les unités monomériques comportant une fonction acide faible sont issues de l'acide acrylique ou de l'acide méthacrylique, partiellement salifié en sel de sodium, en sel de potassium, ou en sel d'ammonium.The invention more particularly relates to a composition as defined above, for which in the polyelectrolyte (P), the monomeric units comprising a strong acid function are derived from 2-methyl 2 - [(1-oxo 2) -propenyl) amino] 1-propanesulfonic acid partially or totally salified to sodium salt, potassium salt, or ammonium salt and the monomeric units having weak acid function are derived from acrylic acid or methacrylic acid partially salified with sodium salt, potassium salt or ammonium salt.
L'invention a aussi plus particulièrement pour objet une composition telle que définie précédemment, pour laquelle le polyélectrolyte P comprend, en pourcentage molaire, de 0,5% à 10% d'une unité monomérique issue du monomère de formule (I) telle que définie précédemment.The invention also more particularly relates to a composition as defined above, for which the polyelectrolyte P comprises, in molar percentage, from 0.5% to 10% of a monomeric unit derived from the monomer of formula (I) such that previously defined.
Selon un autre aspect particulier de la présente invention, ladite composition tensioactive (C) telle que définie précédemment, comprend en outre :
- 3) - Jusqu'à 5% molaire d'une composition (CV) comprenant pour 100% molaire
- α) - De 60% molaire à 100% molaire d'un composé de formule (V) :
- R2 représente un radical alkyle linéaire ou ramifié, comportant de 12 atomes de carbone,
- T8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m8-H dans lequel m8 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T9 identique ou différent de T8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m9-H dans lequel m9 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- étant entendu que la somme m8 + m9 est supérieure à 0 et inférieure ou égale à dix ;
- P) - Optionnellement jusqu'à 40% molaire d'un composé de formule (V')
- R'2 représente un radical alkyle linéaire ou ramifié, comportant 14 atomes de carbone,
- T'8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m8-H dans lequel m8 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T'9 identique ou différent de T'8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m9-H dans lequel m9 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- étant entendu que la somme m8 + m9 est supérieure à 0 et inférieure ou égale à dix ; et
- γ) - Optionnellement jusqu'à 10% molaire d'un composé de formule
- R"2 représente un radical alkyle linéaire ou ramifié, comportant 16 atomes de carbone,
- T"8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m8-H dans lequel m8 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix,
- T"9 identique ou différent de T"8, représente un atome d'hydrogène ou un radical (-CH2-CH2-O-)m9-H dans lequel m9 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix, et
- étant entendu que la somme m8 + m9 est supérieure à 0 et inférieure ou égale à dix.
- α) - De 60% molaire à 100% molaire d'un composé de formule (V) :
- 3) - Up to 5 mol% of a composition (C V ) comprising for 100 mol%
- α) - from 60 mol% to 100 mol% of a compound of formula (V):
- R2 represents a linear or branched alkyl radical containing 12 carbon atoms,
- T 8 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 8 -H in which m 8 is an integer greater than or equal to zero and less than or equal to ten,
- T 9 identical or different from T 8 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m9 -H wherein m9 is an integer greater than or equal to zero and less than or equal to ten, and
- it being understood that the sum m8 + m9 is greater than 0 and less than or equal to ten;
- P) - Optionally up to 40 mol% of a compound of formula (V ')
- R ' 2 represents a linear or branched alkyl radical containing 14 carbon atoms,
- T ' 8 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 8 -H in which m 8 is an integer greater than or equal to zero and less than or equal to ten,
- T ' 9 identical or different from T' 8 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m9 -H in which m9 is an integer greater than or equal to zero and less than or equal to to ten, and
- it being understood that the sum m8 + m9 is greater than 0 and less than or equal to ten; and
- γ) - optionally up to 10 mol% of a compound of formula
- R " 2 represents a linear or branched alkyl radical containing 16 carbon atoms,
- T " 8 represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 8 -H in which m 8 is an integer greater than or equal to zero and less than or equal to ten,
- T " 9 identical or different from T" 8 , represents a hydrogen atom or a radical (-CH 2 -CH 2 -O-) m 9 -H in which m 9 is an integer greater than or equal to zero and less than or equal to to ten, and
- it being understood that the sum m8 + m9 is greater than 0 and less than or equal to ten.
- α) - from 60 mol% to 100 mol% of a compound of formula (V):
Selon un autre aspect particulier de la présente invention, ladite composition tensioactive (C) telle que définie précédemment, comprend en outre :
- 4) - Jusqu'à 5% molaire d'une composition (CVI) comprenant pour 100% molaire :
- α) - De 60% molaire à 100% molaire d'un composé de formule (VI) :
R2-(-CH2-CH2-O-)m10-H (VI),
dans laquelle R2 représente un radical alkyle linéaire ou ramifié, comportant 12 atomes de carbone et m10 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix ; - P) - Optionnellement jusqu'à 40% molaire d'un composé de formule (VI') :
R'2-(-CH2-CH2-O-)m10-H (VI'),
dans laquelle R'2 représente un radical alkyle linéaire ou ramifié, comportant 14 atomes de carbone et m10 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix ; et - γ) - Optionnellement jusqu'à 10% molaire d'un composé de formule (VI") :
R"2-(-CH2-CH2-O-)m10-H (VI"),
dans laquelle R"2 représente un radical alkyle linéaire ou ramifié, comportant 16 atomes de carbone et m10 est un nombre entier supérieur ou égal à zéro et inférieur ou égal à dix.
- α) - De 60% molaire à 100% molaire d'un composé de formule (VI) :
- 4) - Up to 5 mol% of a composition (C VI ) comprising for 100 mol%:
- α) - from 60 mol% to 100 mol% of a compound of formula (VI):
R2 - (- CH 2 -CH 2 -O-) m10 -H (VI),
wherein R2 is a linear or branched alkyl radical having 12 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten; - P) - Optionally up to 40 mol% of a compound of formula (VI '):
R ' 2 - (- CH 2 -CH 2 -O-) m10 -H (VI'),
wherein R ' 2 represents a linear or branched alkyl radical having 14 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten; and - γ) - optionally up to 10 mol% of a compound of formula (VI "):
R " 2 - (- CH 2 -CH 2 -O-) m 10 -H (VI"),
wherein R " 2 represents a linear or branched alkyl radical having 16 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten.
- α) - from 60 mol% to 100 mol% of a compound of formula (VI):
Selon un aspect particulier de la présente invention, la dite composition tensioactive (C) telle que définie précédemment, comprend :
- 1) - Une proportion supérieure ou égale à 20% molaire et inférieure ou égale à 50% molaire d'une composition (CII) telle que définie précédemment ;
- 2) - Une proportion supérieure ou égale à 50% molaire et inférieure ou égale à 80% molaire d'une composition (CII) telle que définie précédemment.
- 1) - a proportion greater than or equal to 20 mol% and less than or equal to 50 mol% of a composition (C II ) as defined above;
- 2) - A proportion greater than or equal to 50 mol% and less than or equal to 80 mol% of a composition (C II ) as defined above.
Selon un autre aspect particulier de la présente invention :
- 1) - ladite composition (CII) comprend pour 100% molaire :
- α) - De 60% à 80% molaire du composé de formule (II),
- P) - De 15% à 30% molaire du composé de formule (II'), et
- γ) - Jusqu'à 10% molaire du composé de formule (II"), et
- 2) - ladite composition (CIII) comprend pour 100% molaire :
- α) - De 60% à 80% molaire du composé de formule (III), de son isomère de formule (IV) ou du mélange de ces isomères,
- P) - de 15% à 30% molaire du composé de formule (III'), de son isomère de formule (IV') ou du mélange de ces isomères, et
- γ) - Jusqu'à 10% molaire du composé de formule (III"), de son isomère de formule (IV") ou du mélange de ces isomères.
- 1) - said composition (C II ) comprises for 100 mol%:
- α) - from 60% to 80% molar of the compound of formula (II),
- P) - from 15% to 30% molar of the compound of formula (II '), and
- γ ) - up to 10 mol% of the compound of formula (II "), and
- 2) - said composition (C III ) comprises for 100 mol%:
- α) - from 60% to 80% molar of the compound of formula (III), of its isomer of formula (IV) or of the mixture of these isomers,
- P) - from 15% to 30% molar of the compound of formula (III '), of its isomer of formula (IV') or of the mixture of these isomers, and
- γ) - Up to 10 mol% of the compound of formula (III "), its isomer of formula (IV") or the mixture of these isomers.
Dans la composition telle que définie ci-dessus, le système émulsionnant (S1) de type eau dans huile (E/H) est constitué soit d'un seul tensioactif soit d'un mélange de tensioactifs à condition que ledit tensioactif ou ledit mélange ait une valeur de HLB suffisamment faible pour induire des émulsions eau dans huile. Comme agent émulsionnant de type eau - dans huile, il y a par exemple les esters de sorbitan, comme l'oléate de sorbitan, comme celui commercialisé par la société SEPPIC sous le nom MONTANE™ 80, l'isostéarate de sorbitan, comme celui commercialisé par la société SEPPIC sous le nom MONTANE™ 70 ou le sesquioléate de sorbitan comme celui commercialisé par la société SEPPIC sous le nom MONTANE™ 83. Il y aussi certains esters de sorbitan polyéthoxylés, par exemple le mono-oléate de sorbitan pentaéthoxylé comme celui commercialisé par la société SEPPIC sous le nom MONTANOX™ 81 ou l'isostéarate de sorbitan pentaéthoxylé comme celui commercialisé sous le nom MONTANOX™ 71 par la société SEPPIC. Il y a encore l'alcool oléocétylique diéthoxylé, comme celui commercialisé sous le nom SIMULSOL™ OC 72 par la société SEPPIC, les polyesters de poids moléculaire compris entre 1000 et 3000, produits de la condensation entre un acide poly(isobutènyl) succinique ou son anhydride et tels que l'HYPERMER™ 2296 commercialisé par la société UNIQEMA ou enfin les copolymères blocks de poids moléculaire compris entre 2500 et 3500, comme l'HYPERMER™ B246 commercialisé par la société UNIQEMA ou le SIMALINE™ IE 200 commercialisé par la société SEPPIC.In the composition as defined above, the emulsifier system (S 1 ) of water-in-oil (W / O) type consists of either a single surfactant or a mixture of surfactants provided that said surfactant or said mixture has a low enough HLB value to induce water-in-oil emulsions. As an emulsifier of the water-in-oil type, there are, for example, sorbitan esters, such as sorbitan oleate, such as that marketed by the company SEPPIC under the name MONTANE ™ 80, sorbitan isostearate, such as that marketed by the company SEPPIC under the name MONTANE ™ 70 or sorbitan sesquioleate such as that marketed by the company SEPPIC under the name Montane ™ 83. There are also certain polyethoxylated sorbitan esters, for example pentaethoxylated sorbitan mono-oleate such as that marketed by the company SEPPIC under the name MONTANOX ™ 81 or pentaethoxylated sorbitan isostearate such as that sold under the name MONTANOX ™ 71 by the company SEPPIC. There is still oleocetyl alcohol diethoxylated, such as that marketed under the name SIMULSOL ™ OC 72 by the company SEPPIC, polyesters with a molecular weight between 1000 and 3000, products of condensation between a poly (isobutenyl) succinic acid or its anhydride and such as HYPERMER ™ 2296 marketed by the company UNIQEMA or finally block copolymers of molecular weight between 2500 and 3500, such as HYPERMER ™ B246 marketed by the company UNIQEMA or SIMALINE ™ IE 200 marketed by the company SEPPIC.
Dans la composition objet de la présente invention, le système émulsionnant (S2) de type huile dans eau (H/E) comprend au moins une quantité non nulle de la composition tensioactive (C) telle que définie ci-dessus. Par quantité non nulle, on désigne plus particulièrement une proportion supérieure ou égale à 10 % massique et, tout particulièrement supérieure ou égale à 50% massique.In the composition that is the subject of the present invention, the oil-in-water (O / W) emulsifier system (S 2 ) comprises at least a non-zero amount of the surfactant composition (C) as defined above. By non-zero amount, more particularly denotes a proportion greater than or equal to 10% by mass and, more particularly greater than or equal to 50% by mass.
Selon un mode particulier de la présente invention, ledit système émulsionnant (S2) de type huile dans eau (H/E) consiste en 100% massique de la composition tensioactive (C) telle que définie précédemment.According to one particular embodiment of the present invention, said oil-in-water (O / W) emulsifier system (S 2 ) consists of 100% by weight of the surfactant composition (C) as defined above.
Selon un autre mode particulier de la présente invention le système émulsionnant (S2) de type huile dans eau (H/E) comprend en outre au moins un tensioactif émulsionnant du type (H/E) différent de l'un ou de l'autre des composés constituant ladite composition tensioactive (C) telle que définies précédemment.According to another particular embodiment of the present invention, the emulsifier system (S 2 ) of oil-in-water (O / W) type further comprises at least one emulsifying surfactant of the (O / W) type different from one or the other. other compounds constituting said surfactant composition (C) as defined above.
Par "agent émulsionnant du type huile dans eau", on désigne des agents émulsionnants possédant une valeur de HLB suffisamment élevée pour fournir des émulsions huile dans eau, tels que :
- Les esters de sorbitan éthoxylés comme l'oléate de sorbitan polyéthoxylé avec 20 moles d'oxyde d'éthylène commercialisé par la société SEPPIC sous le nom de MONTANOX™ 80 ou le laurate de sorbitan polyéthoxylé avec 20 moles d'oxyde d'éthylène commercialisé par la société SEPPIC sous le nom de MONTANOX™ 20 ;
- L'huile de ricin polyéthoxylée avec 40 moles d'oxyde d'éthylène commercialisé sous le nom SIMULSOL™ OL50 ;
- L'alcool oléodécylique décaéthoxylé, commercialisé par la société SEPPIC sous le nom SIMULSOL™ OC 710 ;
- L'alcool laurique heptaéthoxylé commercialisé sous le nom SIMULSOL™ P7 ;
- Ou les hexaoléates de sorbitan polyéthoxylés commercialisés par la société SEPPIC sous le nom SIMALINE™ IE 400.
- Ethoxylated sorbitan esters such as polyethoxylated sorbitan oleate with 20 moles of ethylene oxide marketed by the company SEPPIC under the name MONTANOX ™ 80 or polyethoxylated sorbitan laurate with 20 moles of ethylene oxide marketed by the company SEPPIC under the name MONTANOX ™ 20;
- Castor oil polyethoxylated with 40 moles of ethylene oxide sold under the name SIMULSOL ™ OL50;
- The oleodecyl alcohol decaethoxylated, sold by the company SEPPIC under the name SIMULSOL ™ OC 710;
- Heptaethoxylated lauryl alcohol marketed under the name SIMULSOL ™ P7;
- Or the polyethoxylated sorbitan hexaoleate marketed by SEPPIC under the name SIMALINE ™ IE 400.
Selon un mode particulier de la composition telle que définie précédemment, le système émulsionnant (S2) de type huile dans eau (H/E) comprend en outre une proportion massique non nulle d'au moins un agent émulsionnant du type huile dans eau choisi parmi l'oléate de sorbitan polyéthoxylé avec 20 moles d'oxyde d'éthylène ; le laurate de sorbitan polyéthoxylé avec 20 moles d'oxyde d'éthylène ; l'huile de ricin polyéthoxylée avec 40 moles d'oxyde d'éthylène ; l'alcool oléodécylique décaéthoxylé, l'alcool laurique heptaéthoxylé ; ou les hexaoléates de sorbitan polyéthoxylés.According to one particular embodiment of the composition as defined above, the emulsifier system (S 2 ) of oil-in-water (O / W) type further comprises a non-zero mass proportion of at least one emulsifying agent of the chosen oil-in-water type. from polyethoxylated sorbitan oleate with 20 moles of ethylene oxide; polyethoxylated sorbitan laurate with 20 moles of ethylene oxide; polyethoxylated castor oil with 40 moles of ethylene oxide; decaethoxylated oleodecyl alcohol, heptaethoxylated lauric alcohol; or polyethoxylated sorbitan hexaoleate.
Selon un mode tout particulier de la présente invention, ledit système émulsionnant (S2) de type huile dans eau (H/E) comprend pour 100% de sa masse :
- de 10% massique à 40% massique d'alcool laurique heptaéthoxylé et
- de 60% massique à 90% massique de ladite composition tensioactive (C).
- from 10% by mass to 40% by weight of lauric alcohol heptaethoxylated and
- from 60% to 90% by weight of said surfactant composition (C).
Par polyélectrolyte branché, on désigne pour (P), un polyélectrolyte non linéaire qui possède des chaînes pendantes de manière à obtenir, lorsque ce polyélectrolyte est mis en solution dans l'eau, un fort état d'enchevêtrement conduisant à des viscosités à bas gradient très importantes.By connected polyelectrolyte, is meant for (P), a nonlinear polyelectrolyte which has pendant chains so as to obtain, when this polyelectrolyte is dissolved in water, a high state of entanglement leading to low gradient viscosities very important.
Par polyélectrolyte réticulé on désigne pour (P), un polyélectrolyte non linéaire se présentant à l'état de réseau tridimensionnel insoluble dans l'eau, mais gonflable à l'eau et conduisant donc à l'obtention d'un gel chimique.For crosslinked polyelectrolyte is meant for (P), a nonlinear polyelectrolyte in the state of three-dimensional network insoluble in water, but swellable with water and thus leading to the production of a chemical gel.
La composition selon l'invention peut comporter des polyélectrolytes réticulés et/ou des polyélectrolytes branchés.The composition according to the invention may comprise crosslinked polyelectrolytes and / or branched polyelectrolytes.
Lorsque le polyélectrolyte (P) est réticulé, il l'est plus particulièrement avec un composé diéthylénique ou polyéthylènique dans une proportion molaire exprimée par rapport aux monomères mis en oeuvre dépendant la nature chimique du réticulant et généralement inférieure ou égale à 0,40% molaire, principalement inférieure à 0,25% molaire, plus particulièrement inférieure ou égale à 0,05% molaire et tout particulièrement comprise entre 0,005 % et 0,01 % molaire. De préférence, l'agent de réticulation et/ou l'agent de ramification est choisi parmi le diméthacrylate d'éthylèneglycol, le diacrylate de diéthylèneglycol, le diallyloxyacétate de sodium. le diacrylate d'éthylèneglycol, le diallyl urée, le triallylamine, le triméthylol propanetriacrylate, le méthylène-bis(acrylamide) ou un mélange de ces composés.When the polyelectrolyte (P) is crosslinked, it is more particularly with a diethylenic or polyethylenic compound in a molar proportion expressed relative to the monomers used depending on the chemical nature of the crosslinking agent and generally less than or equal to 0.40 mol% , mainly less than 0.25 mol%, more particularly less than or equal to 0.05 mol% and very particularly between 0.005% and 0.01 mol%. Preferably, the crosslinking agent and / or the branching agent is chosen from ethylene glycol dimethacrylate, diethylene glycol diacrylate and sodium diallyloxyacetate. ethylene glycol diacrylate, diallyl urea, triallylamine, trimethylol propanetriacrylate, methylenebis (acrylamide) or a mixture of these compounds.
L'invention a plus particulièrement pour objet, une composition telle que définie précédemment, pour laquelle dans le polyélectrolyte (P), l'unité monomérique issue du monomère de formule (I) est une unité monomérique issus de l'acrylate de lauryle tétraéthoxylé.The invention more particularly relates to a composition as defined above, for which in the polyelectrolyte (P), the monomeric unit derived from the monomer of formula (I) is a monomeric unit derived from tetraethoxylated lauryl acrylate.
Le polyélectrolyte (P) est alors de préférence choisi parmi :
- Les copolymères réticulés de l'acide acrylique partiellement salifié sous forme de sel de sodium ou de sel d'ammonium, de l'acrylamide et de l'acrylate de lauryle tétraéthoxylé ;
- Les copolymères réticulés de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique partiellement salifié sous forme de sel de sodium ou de sel d'ammonium, de l'acrylamide et de l'acrylate de lauryle tétraéthoxylé ;
- Les copolymères réticulés de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique partiellement salifié sous forme de sel de sodium ou de sel d'ammonium, de l'acrylate de 2-hydroxy éthyle et de l'acrylate de lauryle tétraéthoxylé ;
- Les copolymères réticulés de l'acrylamide, de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique, de l'acide acrylique partiellement salifiés sous forme de sel sodium ou de sel d'ammonium et de l'acrylate de lauryle tétraéthoxylé ;
- Les copolymères de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique partiellement salifié sous forme de sel de sodium ou de sel d'ammonium, de l'acrylamide, du vinyl pyrrolidone et de l'acrylate de lauryle tétraéthoxylé ; et
- Les copolymères réticulés de l'acide 2-méthyl 2-[(1-oxo 2-propènyl) amino] 1-propanesulfonique partiellement ou totalement salifié sous forme de sel de sodium, de l'acide acrylique partiellement salifiés sous forme de sel sodium ou de sel d'ammonium, de l'acrylate de 2-hydroxy éthyle, de tris(hydroxyméthyl)aminométhyl acrylamide et de l'acrylate de lauryle tétraéthoxylé.
- Crosslinked copolymers of partially salified acrylic acid in the form of sodium salt or ammonium salt, acrylamide and tetraethoxylated lauryl acrylate;
- Cross-linked copolymers of partially salified 2-methyl 2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid in the form of sodium salt or ammonium salt, acrylamide and acrylate tetraethoxylated lauryl;
- Cross-linked copolymers of partially salified 2-methyl 2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid in the form of sodium salt or ammonium salt, 2-hydroxyethyl acrylate and tetraethoxylated lauryl acrylate;
- Crosslinked copolymers of acrylamide, 2-methyl-2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid, partially salified acrylic acid in the form of sodium salt or ammonium salt and tetraethoxylated lauryl acrylate;
- Copolymers of partially salified 2-methyl-2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid in the form of sodium salt or ammonium salt, acrylamide, vinyl pyrrolidone and tetraethoxylated lauryl acrylate; and
- Crosslinked copolymers of 2-methyl-2 - [(1-oxo-2-propenyl) amino] 1-propanesulfonic acid partially or totally salified in the form of sodium salt, partially salified acrylic acid in sodium salt form or of ammonium salt, 2-hydroxyethyl acrylate, tris (hydroxymethyl) aminomethyl acrylamide and tetraethoxylated lauryl acrylate.
Selon un autre aspect particulier de la présente invention, le polyélectrolyte anionique (P) réticulé, comporte pour 100% de monomères mis en oeuvre :
- De 20% molaire à 80% molaire d'unités monomériques issues d'un monomère comportant soit une fonction acide fort, soit une fonction acide faible ;
- De 15% molaire à 75% molaire d'unités monomériques issues d'un monomère neutre différent du composé de formule (I) telle que définie précédemment ;
- De 0,5% à 5% molaire d'unités monomériques issues d'un monomère de formule (I) telle que définie précédemment.
- From 20 mol% to 80 mol% of monomeric units derived from a monomer comprising either a strong acid function or a weak acid function;
- From 15 mol% to 75 mol% of monomeric units derived from a neutral monomer different from the compound of formula (I) as defined above;
- 0.5% to 5% molar monomeric units derived from a monomer of formula (I) as defined above.
Selon un autre aspect particulier de la présente invention, le polyélectrolyte anionique (P) réticulé comporte pour 100% de monomères mis en oeuvre :
- De 40% molaire à 80% molaire d'unités monomériques issues d'un monomère comportant une fonction acide fort ;
- De 15% molaire à 55% molaire d'unités monomériques issues d'un monomère neutre différent du composé de formule (I) telle que définie précédemment ;
- De 1% à 5% molaire d'unités monomériques issues d'un monomère de formule (I) telle que définie précédemment.
- From 40 mol% to 80 mol% of monomeric units derived from a monomer comprising a strong acid function;
- From 15 mol% to 55 mol% of monomeric units derived from a neutral monomer different from the compound of formula (I) as defined above;
- 1% to 5% molar monomeric units derived from a monomer of formula (I) as defined above.
Dans la composition objet de la présente invention, la phase huile est constituée soit par une huile minérale commerciale contenant des hydrocarbures saturés comme les paraffines, les isoparaffines ou les cycloparaffines présentant à température ambiante, une densité entre 0,7 et 0,9 et un point d'ébullition supérieur à environ 250°C, telle que par exemple le MARCOL™ 52 ou le MARCOL™ 82, commercialisés par EXXON CHEMICAL, soit par une huile végétale comme le squalane d'origine végétale, soit par une huile de synthèse tel que le polyisobutène hydrogéné ou le polydécène hydrogéné, soit par un mélange de plusieurs de ces huiles. Le MARCOL™ 52 est une huile commerciale répondant à la définition des huiles de vaseline du Codex français. C'est une huile blanche minérale conforme aux réglementations FDA 21 CFR 172.878 et CFR 178.3620 (a) et elle est inscrite à la Pharmacopée des USA, US XXIII (1995) et à la Pharmacopée européenne (1993). La composition selon l'invention peut également contenir divers additifs tels que des agents complexants, des agents de transfert ou des agents limiteurs de chaîne.In the composition that is the subject of the present invention, the oil phase is constituted either by a commercial mineral oil containing saturated hydrocarbons such as paraffins, isoparaffins or cycloparaffins having, at ambient temperature, a density between 0.7 and 0.9 and a boiling point greater than about 250 ° C, such as for example MARCOL ™ 52 or MARCOL ™ 82, marketed by EXXON CHEMICAL, or by a vegetable oil such as squalane of vegetable origin, or by a synthetic oil such as hydrogenated polyisobutene or hydrogenated polydecene, or a mixture of several of these oils. MARCOL ™ 52 is a commercial oil that meets the definition of vaseline oils from the French Codex. It is a white mineral oil in accordance with FDA 21 CFR 172.878 and CFR 178.3620 (a) and is listed in the US Pharmacopeia, US XXIII (1995) and the European Pharmacopoeia (1993). The composition according to the invention may also contain various additives such as complexing agents, transfer agents or chain-limiting agents.
Selon un autre aspect de la présente invention, celle-ci a pour objet un procédé de préparation de la composition telle que définie précédemment, caractérisé en ce que :
- a) l'on émulsionne une phase aqueuse (A) contenant les monomères et les éventuels additifs hydrophiles, dans une phase organique (O) contenant, le système tensioactif (S1), un mélange constitué de l'huile destinée à être présente dans la composition finale et d'une huile volatile et les éventuels additifs hydrophobes,
- b) l'on amorce la réaction de polymérisation par introduction dans l'émulsion formée en a), d'un initiateur de radicaux libres puis on la laisse se dérouler, et
- c) l'on concentre par distillation le milieu réactionnel issu de l'étape b), jusqu'à élimination complète de ladite huile volatile ;
- d) l'on introduit dans le milieu concentré issu de l'étape c), ledit système émulsionnant (S2) de type huile dans eau (H/E) à une température inférieure ou égale à 70°C.
- a) an aqueous phase (A) containing the monomers and any hydrophilic additives is emulsified in an organic phase (O) containing, the surfactant system (S 1 ), a mixture consisting of the oil intended to be present in the final composition and a volatile oil and any hydrophobic additives,
- b) initiating the polymerization reaction by introducing into the emulsion formed in a), a free radical initiator and then allowing it to proceed, and
- c) the reaction medium resulting from step b) is concentrated by distillation until complete elimination of said volatile oil;
- d) is introduced into the concentrated medium from step c), said emulsifier system (S 2 ) oil-in-water (O / W) at a temperature of less than or equal to 70 ° C.
Les huiles volatiles appropriées à la mise en oeuvre du procédé tel que défini ci- dessus, sont par exemple des isoparaffines légères comportant de 8 à 13 atomes de carbone comme par exemple celles vendues sous les noms ISOPAR™ G, ISOPAR™ L ou ISOPAR™ H ou ISOPAR™ J.The volatile oils suitable for carrying out the process as defined above are, for example, light isoparaffins comprising from 8 to 13 carbon atoms, for example those sold under the names ISOPAR ™ G, ISOPAR ™ L or ISOPAR ™. H or ISOPAR ™ J.
Selon une mise en oeuvre préférée du procédé tel que défini précédemment, la réaction de polymérisation est amorcée par un couple oxydoréducteur, tel que le couple hydroperoxyde de cumène - métabisulfite de sodium, à une température inférieure ou égale à 10°C, puis conduite soit de manière quasi-adiabatique jusqu'à une température supérieure ou égale à 40°C, plus particulièrement supérieure ou égale à 50°C, soit en contrôlant l'évolution de la température.According to a preferred embodiment of the process as defined above, the polymerization reaction is initiated by a redox pair, such as cumene hydroperoxide-sodium metabisulphite pair, at a temperature of less than or equal to 10 ° C., and then conducted either in a quasi-adiabatic manner up to a temperature greater than or equal to 40 ° C, more particularly greater than or equal to 50 ° C, or by controlling the evolution of the temperature.
Le procédé de préparation de ladite composition tensioactive (C) telle que définie précédemment comprend les étapes successive suivantes :
- Une étape a) de réaction d'un mélange d'alcools comprenant pour 100% molaire :
- De 60% molaire à 100% molaire d'un composé de formule (VII),
R2-O-H (VII),
dans laquelle R2 représente un radical alkyle linéaire ou ramifié, comportant 12 atomes de carbone ; - Optionnellement jusqu'à 40% molaire d'un composé de formule (VII') :
R'2-O-H (VII'),
dans laquelle R'2 représente un radical alkyle linéaire ou ramifié, comportant 14 atomes de carbone, et - Optionnellement jusqu'à 10% molaire d'un composé de formule (VII") :
R"2-O-H (VII"),
dans laquelle R"2 représente un radical alkyle linéaire ou ramifié, comportant 16 atomes de carbone;
- De 60% molaire à 100% molaire d'un composé de formule (VII),
- α) - Le composé de formule (IX) :
- β) - Optionnellement le composé de formule (IX)' :
- γ) - Optionnellement le composé de formule (IX") :
- δ) - Le composé de formule (X) :
- ε) - Optionnellement le composé de formule (X') :
- ζ) - Optionnellement le composé de formule (X") :
- A reaction step a) of a mixture of alcohols comprising for 100 mol%:
- From 60 mol% to 100 mol% of a compound of formula (VII),
R2-OH (VII),
wherein R2 represents a linear or branched alkyl radical having 12 carbon atoms; - Optionally up to 40 mol% of a compound of formula (VII '):
R ' 2 -OH (VII'),
in which R ' 2 represents a linear or branched alkyl radical containing 14 carbon atoms, and - Optionally up to 10 mol% of a compound of formula (VII "):
R " 2 -OH (VII"),
wherein R " 2 represents a linear or branched alkyl radical having 16 carbon atoms;
- From 60 mol% to 100 mol% of a compound of formula (VII),
- α) - The compound of formula (IX):
- β) - Optionally the compound of formula (IX) ':
- γ) - Optionally the compound of formula (IX "):
- δ) - The compound of formula (X):
- ε) - Optionally the compound of formula (X '):
- ζ) - Optionally the compound of formula (X "):
Une étape b) de réaction de ladite composition (C') avec l'oxyde d'éthylène de formule (XII) :
L'invention a aussi pour objet l'utilisation de la composition telle que définie précédemment, comme agent épaississant et/ou émulsionnant, pour préparer une composition topique cosmétique, dermopharmaceutique ou pharmaceutique.The subject of the invention is also the use of the composition as defined above, as thickener and / or emulsifier, for preparing a topical cosmetic, dermopharmaceutical or pharmaceutical composition.
L'invention a aussi pour objet une composition topique cosmétique, dermopharmaceutique ou pharmaceutique, caractérisée en ce qu'elle comprend, comme agent épaississant et/ou émulsionnant, une quantité efficace de la composition telle que définie précédemment.The invention also relates to a topical cosmetic, dermopharmaceutical or pharmaceutical composition, characterized in that it comprises, as thickening and / or emulsifying agent, an effective amount of the composition as defined above.
Une composition topique selon l'invention, destinée à être appliquée sur la peau, sur le cuir chevelu ou les muqueuses de l'homme ou de l'animal, peut consister en une émulsion topique comprenant au moins une phase aqueuse et au moins une phase huile, se présentant sous la forme d'une émulsion eau dans huile, ou huile dans eau, ou eau dans huile dans eau, ou huile dans eau dans huile. Plus particulièrement, cette émulsion topique peut consister en une émulsion fluide, telle un lait ou un gel fluide. La phase huile de l'émulsion topique peut consister en un mélange d'une ou plusieurs huiles.A topical composition according to the invention, intended to be applied to the skin, the scalp or the mucous membranes of humans or animals, may consist of a topical emulsion comprising at least one aqueous phase and at least one phase oil, in the form of a water-in-oil emulsion, or oil in water, or water in oil in water, or oil in water in oil. More particularly, this topical emulsion may consist of a fluid emulsion, such as a milk or a fluid gel. The oil phase of the topical emulsion can consist of a mixture of one or more oils.
Une composition topique selon l'invention peut être destinée à une utilisation cosmétique ou être utilisée pour préparer un médicament destiné au traitement des maladies de la peau, du cuir chevelu et des muqueuses. Dans ce dernier cas, la composition topique comporte alors un principe actif qui peut par exemple consister en un agent anti-inflammatoire, un myorelaxant, un antifongique ou un antibactérien.A topical composition according to the invention may be intended for cosmetic use or be used to prepare a medicament for the treatment of diseases of the skin, scalp and mucous membranes. In the latter case, the topical composition then comprises an active ingredient which may for example consist of an anti-inflammatory agent, a muscle relaxant, an antifungal agent or an antibacterial agent.
Lorsque la composition topique est utilisée en tant que composition cosmétique destinée à être appliquée sur la peau, sur le cuir chevelu ou les muqueuses, elle peut ou non comporter un principe actif, par exemple un agent hydratant, un agent bronzant, un filtre solaire, un antirides, un agent à visée amincissante, un agent antiradicalaire, un agent antipelliculaire, un agent anti-acnéique ou un antifongique.When the topical composition is used as a cosmetic composition intended to be applied to the skin, the scalp or the mucous membranes, it may or may not comprise an active ingredient, for example a moisturizing agent, a tanning agent, a sunscreen, an anti-wrinkle, a slimming agent, an antiradical agent, an anti-dandruff agent, an anti-acne agent or an antifungal agent.
Par quantité efficace, on signifie que la composition topique selon l'invention comporte une quantité suffisante de latex inverse selon l'invention pour provoquer une modification de sa rhéologie. La composition topique selon l'invention comporte habituellement entre 0,1% et 10% en poids dudit latex inverse auto-inversible défini ci-dessus. Le pH de la composition topique est généralement compris entre 3 et 9.By effective amount, it is meant that the topical composition according to the invention comprises a sufficient amount of inverse latex according to the invention to cause a change in its rheology. The topical composition according to the invention usually comprises between 0.1% and 10% by weight of said self-invertible inverse latex defined above. The pH of the topical composition is generally between 3 and 9.
La composition topique peut en outre comporter des composés classiquement compris dans ce type de compositions, par exemple des parfums, des conservateurs, des colorants, des émollients ou des tensioactifs.The topical composition may further comprise compounds conventionally included in this type of composition, for example perfumes, preservatives, dyes, emollients or surfactants.
Selon encore un autre aspect, l'invention concerne l'utilisation du nouvel agent épaississant et/ou émulsionnant conforme à l'invention, mentionné ci-dessus, pour épaissir et émulsionner une composition topique comprenant au moins une phase aqueuse.According to yet another aspect, the invention relates to the use of the novel thickening and / or emulsifying agent according to the invention, mentioned above, for thickening and emulsifying a topical composition comprising at least one aqueous phase.
La composition selon l'invention est un substitut intéressant à celles vendues sous les noms SEPIGEL™ 305, SEPIGEL™ 501, SIMULGEL™ EG, SIMULGEL™ NS ou SIMUL-GEL™ 600 par la demanderesse, car elle présente aussi une bonne compatibilité avec les autres excipients utilisés pour la préparation de formulations telles que les laits, les lotions, les crèmes, les savons, les bains, les baumes, les shampooings ou les après- shampooings. Elle peut aussi être mise en oeuvre avec lesdits SEPIGEL ou SIMULGEL. Elle est notamment compatible avec les concentrés décrits et revendiqués dans les publications internationales
La composition selon l'invention est également compatible avec les principes actifs tels que par exemple, les agents auto-bronzants comme le dihydroxyacétone (DHA) ou les agents anti-acné ; elle peut donc être introduite dans des compositions auto-bronzantes comme celles revendiquées dans
Elle est également compatible avec les dérivés N-acylés d'aminoacides, ce qui permet son utilisation dans des compositions apaisantes notamment pour peau sensible, telles que celles décrites ou revendiquées dans
Lorsque la composition telle que définie précédemment est destinée au traitement de la peau et/ou du cuir chevelu et/ou des muqueuses, elle comprend plus particulièrement un latex inverse de polyélectrolyte anionique objet de la présente invention. Les latex inverse objet de la présente invention peuvent être utilisés comme épaississant de pâtes d'impression textile.When the composition as defined above is intended for the treatment of the skin and / or the scalp and / or the mucous membranes, it more particularly comprises an inverse anionic polyelectrolyte latex object of the present invention. The inverse latexes of the present invention can be used as a thickener for textile printing pastes.
Les exemples qui suivent ont pour but d'illustrer la présente invention sans toutefois la limiter.The following examples are intended to illustrate the present invention without limiting it.
21.100 g d'un mélange d'alcools gras comprenant de 65% à 75% massique d'alcanol comportant 12 atomes de carbone, de 21% à 28% massique d'alcanol comportant 14 atomes de carbone et de 4% à 8% massique d'alcanol comportant 16 atomes de carbone, préalablement chauffés, sont introduits dans un réacteur, maintenus sous agitation et séchés, On ajoute ensuite 326 grammes de trifluorure de bore à 50% dans le diéthyl-éther puis progressivement sous agitation pendant 4 heures, 32.600 g de 3-(hydroxyméthyl) 3-éthyl oxétane, en maintenant la température autour de 110°C. Le milieu réactionnel est alors laissé encore 11 heures à 115°C. On obtient alors la composition (C') attendue, caractérisée comme suit :
- Aspect à 25°C : Gel trouble
- Indice d'acide (en mg KOH/g ; NFT60-204) : 3,9
- Indice d'hydroxyle (en mg KOH/g) : 400,5
- Teneur massique en 3-(hydroxyméthyl) 3-éthyl oxétane libre (déterminée par chromatographie en phase gazeuse) : < 0.05%
- Teneur massique en alcanols libres (déterminée par chromatographie en phase gazeuse) : alcanol en C12 : 5,7% ; alcanol en C14 : 2,0% ; alcanol en C16 : 0,5%.
- Appearance at 25 ° C: cloudy gel
- Acid number (in mg KOH / g, NFT60-204): 3.9
- Hydroxyl value (in mg KOH / g): 400.5
- Mass content of free 3- (hydroxymethyl) 3-ethyloxetane (determined by gas chromatography): <0.05%
- Mass content of free alkanols (determined by gas chromatography): C12 alkanol: 5.7%; C14 alkanol: 2.0%; C16 alkanol: 0.5%.
50.000 g de la composition intermédiaire (C') obtenue au stade A1) précédent, sont introduits dans un autoclave d'une capacité de 0,1 m3, avec 75 g de potasse puis séchés à une température de 105°C. Une quantité de 35 000 g d'oxyde d'éthylène est ensuite progressivement introduite en régulant la température du mélange réactionnel à une valeur de 125°C. Une fois la quantité totale d'oxyde d'éthylène introduite, le mélange réactionnel est maintenu sous agitation à 125°C pendant une durée supplémentaire d'une heure. Le produit alors obtenu est ensuite refroidi à une température de 80°C et vidangé. On obtient alors la composition tensioactive (C) caractérisée comme suit :
- Aspect à 30°C : liquide limpide
- Couleur : 125 Alpha
- Indice d'hydroxyle (en mg KOH/g) : 252,5
- Indice d'acide (en mg KOH/g) (NFT60-204) : 0,08
- Teneur résiduelle en eau : 0.05%
- Point de trouble (NF EN 1890E) : 76°C
- Teneur massique en alcanols libres (chromatographie en phase gazeuse) : Alcanol en C12 : 1,2% ; alcanol en C14 : 0,4% ; alcanol en C16 : 0,1% soit au total 1.7% d'alcanols résiduels
- Viscosité à 25°C (Brookfield LVT Mobile 3 Vitesse 12) : 1.072 mPa.s
- Appearance at 30 ° C: clear liquid
- Color: 125 Alpha
- Hydroxyl value (in mg KOH / g): 252.5
- Acid number (in mg KOH / g) (NFT60-204): 0.08
- Residual water content: 0.05%
- Cloud point (NF EN 1890E): 76 ° C
- Mass content of free alkanols (gas chromatography): C12 Alcanol: 1.2%; C14 alkanol: 0.4%; C16 alkanol: 0.1%, ie a total of 1.7% of residual alkanols
- Viscosity at 25 ° C (Brookfield LVT Mobile 3 Speed 12): 1.072 mPa.s
-
a) - Dans un premier bêcher, on introduit successivement sous agitation :
- 672,5g d'une solution commerciale à 55% massique de sel de sodium de l'acide 2-acrylamido 2-méthyl propanesulfonique (ATBS Na),
- 20,8g d'acrylate de 2-hydroxy éthyle (HEA) ;
- 0,028g de méthylène bis(acrylamide) (MBA) ;et
- 1,0 g d'une solution commerciale à 40% massique de diéthylènetriaminepenta acétate de sodium.
Puis on y ajuste le pH à 4 en ajoutant si nécessaire la quantité requise d'acide 2-acrylamido 2-méthyl propane sulfonique et de l'eau permutée jusqu'à 700 g
- 672.5 g of a commercial solution containing 55% by weight of sodium salt of 2-acrylamido-2-methylpropanesulphonic acid (ATBS Na),
- 20.8g of 2-hydroxyethyl acrylate (HEA);
- 0.028g of methylene bis (acrylamide) (MBA), and
- 1.0 g of a 40% by weight commercial solution of diethylenetriaminepenta sodium acetate.
Then the pH is adjusted to 4, adding if necessary the required amount of 2-acrylamido-2-methylpropanesulphonic acid and permutated water up to 700 g
-
b) - Dans un deuxième bêcher, on introduit successivement sous agitation :
- 130 g de polyisobutène,
- 30 g de MARCOL™ 52,
- 90 g d'ISOPAR™ H,
- 17 g de MONTANE™ 70,
- 3 g d'HYPERMER™ 6212,
- 5 g de SIMALINE™IE 200,
- 7,2 g d'acrylate de lauryle tétraéthoxylé (commercial) (ALE-4OE),
- 0,36g de peroxyde de dilauroyle.
- 130 g of polyisobutene,
- 30 g of MARCOL ™ 52,
- 90 g of ISOPAR ™ H,
- 17 g MONTANE ™ 70,
- 3 g of HYPERMER ™ 6212,
- 5 g of SIMALINE ™ IE 200,
- 7.2 g of tetraethoxylated lauryl acrylate (commercial) (ALE-4OE),
- 0.36 g of dilauroyl peroxide.
- c) - La phase aqueuse est alors incorporée dans la phase organique sous agitation et puis la pré-émulsion ainsi obtenue est soumise à une agitation mécanique cisaillante à l'aide d'une turbine de type Silverson de manière à créer une émulsion fine sous barbotage d'azote. c) - The aqueous phase is then incorporated into the organic phase with stirring and then the pre-emulsion thus obtained is subjected to a shearing mechanical stirring using a Silverson type turbine so as to create a fine emulsion under bubbling nitrogen.
- d) - Après refroidissement à environ 8 °C, la réaction de polymérisation est initiée à l'aide du couple oxydoréducteur : hydroperoxyde de cumène / métabisulfite de sodium. d) - After cooling to about 8 ° C, the polymerization reaction is initiated using the oxidation-reduction pair: cumene hydroperoxide / sodium metabisulfite.
- e) - Une fois la réaction de polymérisation terminée, on retire par distillation sous vide l'ISOPAR™ H et la quasi-totalité de l'eau. e) - Once the polymerization reaction is complete, the ISOPAR ™ H and almost all of the water are distilled off under vacuum.
- f) - On obtient après introduction de 2% massique de Laureth-7 et de 4% massique de la composition tensioactive (C) obtenue à l'exemple A, le latex inverse auto-inversible (1) contenant environ 63% de polymère, qui est peu visqueux, qui s'inverse très rapidement dans l'eau et dont le pouvoir épaississant est important. De plus ce latex inverse est très stable car on n'observe aucun phénomène de synérèse, en ce qu'il ne relargue que de très peu d'huile et que polymère ne sédimente pas. Sa teneur en eau mesurée par une titrimétrie Karl-Fisher est de 1,8% en poids. f) - is obtained after introduction of 2% by weight of Laureth-7 and 4% by weight of the surfactant composition (C) obtained in Example A, the self-invertible inverse latex (1) containing about 63% of polymer, which is not very viscous, which is reversed very quickly in water and whose thickening power is important. Moreover, this inverse latex is very stable because no phenomenon of syneresis is observed, in that it releases only very little oil and that polymer does not sediment. Its water content measured by Karl-Fisher titrimetry is 1.8% by weight.
a) - On mesure la viscosité du latex inverse auto-inversible (1) obtenu comme indiqué au paragraphe 1, celle d'une solution aqueuse exempte de chlorure de sodium (Sol.1) et celles de solutions aqueuses contenant respectivement 0,1 % massique (Sol.2) et 1% massique (Sol.3) de chlorure de sodium, lesdites solutions aqueuses comprenant chacune 2% massique dudit latex inverse auto-inversible (1). Les résultats mesurés à l'aide d'un viscosimètre Brookfield RVT sont consignés dans le tableau suivant :
a) - The viscosity of the self-invertible inverse latex (1) obtained as indicated in paragraph 1 is measured, that of an aqueous solution free of sodium chloride (Sol.1) and those of aqueous solutions containing respectively 0.1% mass (Sol.2) and 1% by weight (Sol.3) of sodium chloride, said aqueous solutions each comprising 2% by weight of said self-invertible inverse latex (1). The results measured using a Brookfield RVT viscometer are recorded in the following table:
- a) - Le temps d'inversion est évalué en mesurant le temps nécessaire pour obtenir un gel lisse et homogène pour une solution aqueuse à 2% massiques de latex inverse auto-inversible (1) dans les conditions standard de mesure de cette viscosité c'est-à-dire en incorporant 16 g du latex inverse (1) dans 784 g d'eau, le tout étant placé dans un bécher de 1 litre de forme basse, puis en agitant le tout à l'aide d'une hélice type papillon tournant à 150 tours par minute. Le temps d'inversion est donc la durée évaluée entre la mise en route de l'agitateur et l'apparition d'un milieu lisse et homogène dans le bêcher. Dans le présent exemple, le temps d'inversion est de 50 secondes. a) - The inversion time is evaluated by measuring the time required to obtain a smooth and homogeneous gel for a 2% by weight aqueous solution of self-invertible inverse latex (1) under the standard conditions for measuring this viscosity. that is to say by incorporating 16 g of the inverse latex (1) into 784 g of water, all of which is placed in a 1-liter beaker of low form, then stirring the whole with the aid of a typical propeller butterfly turning 150 turns per minute. The inversion time is thus the evaluated time between the start of the stirrer and the appearance of a smooth and homogeneous medium in the beaker. In this example, the inversion time is 50 seconds.
- b) - La stabilité du latex inverse est évaluée en observant le temps d'apparition d'une couche huile en surface. Dans le présent exemple, le temps d'apparition de la couche d'huile en surface du latex inverse (1) est de deux semaines. b) - The stability of the inverse latex is evaluated by observing the time of appearance of an oil layer on the surface. In the present example, the time of appearance of the oil layer on the surface of the inverse latex (1) is two weeks.
On reproduit les étapes a) à d) de l'exemple 1. A l'étape f), on ajoute 4% massique de MONTANOX™ 20 à la place de 4% massique de la composition tensioactive (C) et l'on obtient le latex inverse auto-inversible (T1).The steps a) to d) of Example 1 are repeated. In step f), 4% by weight of MONTANOX ™ 20 is added in place of 4% by weight of the surfactant composition (C) and the self-invertible inverse latex (T1).
a) - On mesure la viscosité du latex inverse auto-inversible (T1) obtenu comme indiqué au paragraphe 1, celle d'une solution aqueuse exempte de chlorure de sodium (Sol.4) et celles de solutions aqueuses contenant respectivement 0,1% massique (Sol.5) et 1% massique (Sol.6) de chlorure de sodium, lesdites solutions aqueuses comprenant chacune 2% massique dudit latex inverse auto-inversible (T1). Les résultats mesurés à l'aide d'un viscosimètre Brookfield RVT sont consignés dans le tableau suivant :
a) - The viscosity of the self-invertible inverse latex (T1) obtained as indicated in paragraph 1 is measured, that of an aqueous solution free of sodium chloride (Sol.4) and those of aqueous solutions containing respectively 0.1% mass (Sol.5) and 1% by weight (Sol.6) of sodium chloride, said aqueous solutions each comprising 2% by weight of said self-invertible inverse latex (T1). The results measured using a Brookfield RVT viscometer are recorded in the following table:
- a) - Le temps d'inversion évalué de la même manière qu'à l'exemple précédent est de 2 minutes 20 secondes. a) - The inversion time evaluated in the same way as in the previous example is 2 minutes 20 seconds.
- b) - La stabilité du latex inverse (T1) est évaluée de la même manière qu'à l'exemple précédent. Un relargage important d'huile est observé au bout d'une semaine. b) - The stability of the inverse latex (T1) is evaluated in the same way as in the previous example. A significant release of oil is observed after one week.
On reproduit les étapes a) à d) de l'exemple 1. A l'étape f), on n'ajoute que 4% massique de la composition tensioactive (C) et l'on obtient le latex inverse auto-inversible (2).The steps a) to d) of Example 1 are repeated. In step f), only 4% by weight of the surfactant composition (C) is added and the self-invertible inverse latex (2) is obtained. ).
- a) - Les performances viscosimétriques du latex inverse (2) sont similaires à celles rapportées pour le latex inverse de l'exemple 1. a) - The viscometric performances of the inverse latex (2) are similar to those reported for the inverse latex of Example 1.
- b) - Le temps d'inversion du latex inverse (2), évalué de la même manière qu'à l'exemple 1, est d'environ 40 secondes. b) - The reversal time of the inverse latex (2), evaluated in the same manner as in Example 1, is about 40 seconds.
- c) - La stabilité du latex inverse (2) est évaluée de la même manière qu'à l'exemple 1. Le temps d'apparition de la couche d'huile est de quelques jours. c) - The stability of the inverse latex (2) is evaluated in the same manner as in Example 1. The time of appearance of the oil layer is a few days.
On reproduit les étapes a) à d) de l'exemple 1. A l'étape f), on ajoute 4% massique d'une composition (C"'), qui comprend pour 100% molaire :
- i) - Une proportion supérieure ou égale à 10% molaire et inférieure ou égale à 50% molaire d'un composé de formule (II"') correspondant à la formule (II) dans laquelle R2 représente un radical alkyle linéaire ou ramifié, comportant 10 atomes de carbone, et dans la quelle la somme m1 + m2 + m3 est égale à 5;
- ii) - Une proportion supérieure ou égale à 50% molaire et inférieure ou égale à 90% molaire d'un composé de formule (III"') ou de son isomère de formule (IV"') ou du mélange de ces deux isomères, formules (III"') et (IV"') correspondant respectivement aux formules (III) et (IV) dans lesquelles R2 représente un radical alkyle linéaire ou ramifié, comportant 10 atomes de carbone, et dans la quelle la somme m4 + m5 + m6 + m7 est égale à 5; et l'on obtient le latex inverse auto-inversible (T2).
- i) - A proportion greater than or equal to 10 mol% and less than or equal to 50 mol% of a compound of formula (II "') corresponding to formula (II) in which R2 represents a linear or branched alkyl radical, comprising 10 carbon atoms, and in which the sum m1 + m2 + m3 is equal to 5;
- ii) a proportion greater than or equal to 50 mol% and less than or equal to 90 mol% of a compound of formula (III "') or of its isomer of formula (IV"') or of the mixture of these two isomers, formulas (III "') and (IV"') respectively corresponding to formulas (III) and (IV) in which R2 represents a linear or branched alkyl radical containing 10 carbon atoms, and in which the sum m4 + m5 + m6 + m7 is equal to 5; and the self-invertible inverse latex (T2) is obtained.
- a) - Les performances viscosimétriques du latex inverse (T2) sont similaires à celles rapportées pour le latex inverse de l'exemple 1. a) - The viscometric performances of the inverse latex (T2) are similar to those reported for the inverse latex of Example 1.
- b) - Le temps d'inversion du latex inverse (T2), évalué de la même manière qu'à l'exemple 1, est d'environ 50 secondes.b) - The reversal time of the inverse latex (T2), evaluated in the same manner as in Example 1, is about 50 seconds.
- c) - La stabilité du latex inverse (T2) est évaluée de la même manière qu'à l'exemple 1. Le temps d'apparition de la couche d'huile est de quelques heures.c) - The stability of the inverse latex (T2) is evaluated in the same way as in Example 1. The time of appearance of the oil layer is a few hours.
-
a) - Dans un premier bêcher, on introduit successivement sous agitation :
- 106,5 g d'une solution commerciale d'acrylamide (AM) à 50% (massique)
- 162,0 g d'acide acrylique glacial (AA),
- 98,1 g d'une solution d'ammoniaque à 29,3% en poids,
- 0,047 g de méthylène bis(acrylamide) (MBA).
- 0,45 g d'une solution commerciale à 40% du diéthylènetriaminepentaacétate de sodium,
- de l'eau permutée jusqu'à 680 g.
- 106.5 g of a commercial solution of acrylamide (AM) at 50% (by mass)
- 162.0 g of glacial acrylic acid (AA),
- 98.1 g of a solution of ammonia at 29.3% by weight,
- 0.047 g methylene bis (acrylamide) (MBA).
- 0.45 g of a 40% commercial solution of sodium diethylenetriaminepentaacetate,
- permuted water up to 680 g.
-
b) - Dans un deuxième bêcher, on introduit successivement sous agitation :
- 121 g de polyisobutène,
- 28 g de MARCOL™ 52,
- 99 g d'ISOPAR™ H,
- 17 g de MONTANE™ 70,
- 3 g d'HYPERMER™ 2296,
- 5 g de SIMALINE™IE 200,
- 1,2 g d'acrylate de lauryle tétraéthoxylé (commercial) (ALE-4OE),
- 0,1 g d'AIBN.
- 121 g of polyisobutene,
- 28 g of MARCOL ™ 52,
- 99 g of ISOPAR ™ H,
- 17 g MONTANE ™ 70,
- 3 g of HYPERMER ™ 2296,
- 5 g of SIMALINE ™ IE 200,
- 1.2 g of tetraethoxylated lauryl acrylate (commercial) (ALE-4OE),
- 0.1 g of AIBN.
- c) - La phase aqueuse est alors incorporée dans la phase organique sous agitation et puis la pré-émulsion ainsi obtenue est soumise à une agitation mécanique cisaillante à l'aide d'une turbine de type Silverson de manière à créer une émulsion fine sous barbotage d'azote. c) - The aqueous phase is then incorporated into the organic phase with stirring and then the pre-emulsion thus obtained is subjected to a shearing mechanical stirring using a Silverson type turbine so as to create a fine emulsion under bubbling nitrogen.
- d) - Après refroidissement à environ 8 °C, la réaction de polymérisation est initiée à l'aide du couple oxydoréducteur : hydroperoxyde de cumène / métabisulfite de sodium. d) - After cooling to about 8 ° C, the polymerization reaction is initiated using the oxidation-reduction pair: cumene hydroperoxide / sodium metabisulfite.
- e) - Une fois la réaction de polymérisation terminée, on retire par distillation sous vide l'ISOPAR™ H et la quasi-totalité de l'eau. e) - Once the polymerization reaction is complete, the ISOPAR ™ H and almost all of the water are distilled off under vacuum.
- f) - On obtient après introduction de 4% de MONTANOX™ 20 et de 2% de Laureth 7, le latex inverse auto-inversible (T3), contenant environ 63% de polymère, qui est peu visqueux, qui s'inverse très rapidement dans l'eau et dont le pouvoir épaississant est important. Sa teneur en eau mesurée par une titrimétrie Karl-Fisher est de 1,8% en poids. f) - After the introduction of 4% of MONTANOX ™ 20 and 2% of Laureth 7, the self-invertible inverse latex (T3), containing about 63% of polymer, which is not very viscous, which is reversed very rapidly, is obtained in water and whose thickening power is important. Its water content measured by Karl-Fisher titrimetry is 1.8% by weight.
a) - On mesure la viscosité du latex inverse auto-inversible (T3) obtenu comme indiqué au paragraphe 1, celle d'une solution aqueuse exempte de chlorure de sodium (Sol.7) et celles de solutions aqueuses contenant respectivement 0,1 % massique (Sol.8) et 1% massique (Sol.9) de chlorure de sodium, lesdites solutions aqueuses comprenant chacune 2% massique dudit latex inverse auto-inversible (T3). Les résultats mesurés à l'aide d'un viscosimètre Brookfield RVT sont consignés dans le tableau suivant :
a) - The viscosity of the self-invertible inverse latex (T3) obtained as indicated in paragraph 1 is measured, that of an aqueous solution free of sodium chloride (Sol.7) and those of aqueous solutions respectively containing 0.1% mass (Sol.8) and 1% by mass (Sol.9) of sodium chloride, said aqueous solutions each comprising 2% by weight of said self-invertible inverse latex (T3). The results measured using a Brookfield RVT viscometer are recorded in the following table:
- b) - Le temps d'inversion du latex inverse (T3), évalué de la même manière qu'à l'exemple 1, est d'environ 2 minutes. b) - The reversal time of the inverse latex (T3), evaluated in the same manner as in Example 1, is about 2 minutes.
- c) - La stabilité du latex inverse du latex inverse (T3) est évaluée de la même manière qu'à l'exemple 1. Le temps d'apparition de la couche d'huile est de deux semaines. c) - The stability of the inverse latex of the inverse latex (T3) is evaluated in the same manner as in Example 1. The time of appearance of the oil layer is two weeks.
On reproduit les étapes a) à d) de l'exemple T3. A l'étape f), on ajoute 2% massique de Laureth 7 et 4% massique de la composition tensioactive (C) au lieu des de 4% de MONTA-NOX™ 20 et 2% de Laureth 7 dudit exemple T3 et l'on obtient le latex inverse auto-inversible (3).Steps a) to d) of Example T3 are repeated. In step f), 2% by weight of Laureth 7 and 4% by weight of the surfactant composition (C) are added instead of 4% of MONTA-NOX ™ 20 and 2% of Laureth 7 of said example T3 and the self-invertible inverse latex (3) is obtained.
- a) - Les performances viscosimétriques du latex inverse (3) sont similaires à celles rapportées pour le latex inverse de l'exemple T3. a) - The viscometric performances of the inverse latex (3) are similar to those reported for the inverse latex of Example T3.
- b) - Le temps d'inversion du latex inverse (3), évalué de la même manière qu'à l'exemple 1, est d'environ 30 secondes. b) - The reversal time of the inverse latex (3), evaluated in the same manner as in Example 1, is about 30 seconds.
- c) - La stabilité du latex inverse (3) est évaluée de la même manière qu'à l'exemple 1. Le temps d'apparition des premières gouttes d'huile est de trois semaines. c) - The stability of the inverse latex (3) is evaluated in the same manner as in Example 1. The time of appearance of the first drops of oil is three weeks.
-
a) - Dans un premier bêcher, on introduit successivement sous agitation :
- 672,5 g d'une solution commerciale à 55% (massique) du sel de sodium de l'acide 2 acrylamido 2 méthyl propanesulfonique (ATBSNa) ;
- 20,8 g d'acrylate de 2-hydroxy éthyle ;
- 12,6g de THAM;
- 2,6 g d'acide acrylique (AA)
- 0,041 g de méthylène bis(acrylamide) (MBA).
- 0,45 g d'une solution commerciale à 40% du diéthylènetriaminepentacétate de sodium,
Puis on y ajuste le pH à 4 en ajoutant si nécessaire la quantité requise d"acide 2 acrylamido 2 méthyl propane sulfonique et de l'eau permutée jusqu'à 700 g a) - In a first beaker, successively introduced with stirring:- 672.5 g of a commercial solution containing 55% (by weight) of the sodium salt of 2-acrylamido-2-methylpropanesulphonic acid (ATBSNa);
- 20.8 g of 2-hydroxyethyl acrylate;
- 12.6 g THAM;
- 2.6 g of acrylic acid (AA)
- 0.041 g methylene bis (acrylamide) (MBA).
- 0.45 g of a 40% commercial solution of sodium diethylenetriaminepentacetate,
Then the pH is adjusted to 4 by adding, if necessary, the required amount of 2-acrylamido-2-methyl propane sulfonic acid and the permutated water up to 700 g. -
b) - Dans un deuxième bêcher, on introduit successivement sous agitation :
- 130 g de polyisobutène,
- 30 g de MARCOL™ 52,
- 90 g d'ISOPAR™ H,
- 17 g de MONTANE™ 70,
- 5 g d'HYPERMER™ 6212,
- 3g de DEHYMULS PGPH (Polyhydroxy stéarate de polyglycérol),
- 7,4 g d'acrylate de lauryle tétraéthoxylé (commercial) (ALE-4OE),
- 0,14g de dilauroyle peroxyde
- 130 g of polyisobutene,
- 30 g of MARCOL ™ 52,
- 90 g of ISOPAR ™ H,
- 17 g MONTANE ™ 70,
- 5 g of HYPERMER ™ 6212,
- 3 g of DEHYMULS PGPH (polyglycerol polyhydroxy stearate),
- 7.4 g of tetraethoxylated lauryl acrylate (commercial) (ALE-4OE),
- 0.14g of dilauroyl peroxide
- c) - La phase aqueuse est alors introduite dans la phase organique sous agitation et puis la pré-émulsion ainsi obtenue est soumise à une agitation mécanique cisaillante à l'aide d'une turbine de type Silverson de manière à créer une émulsion fine sous barbotage d'azote.c) - The aqueous phase is then introduced into the organic phase with stirring and then the pre-emulsion thus obtained is subjected to a shearing mechanical stirring using a Silverson type turbine so as to create a fine emulsion under bubbling nitrogen.
- d) - Après refroidissement à environ 8 °C, la réaction de polymérisation est initiée à l'aide du couple oxydoréducteur : hydroperoxyde de cumène / métabisulfite de sodium.d) - After cooling to about 8 ° C, the polymerization reaction is initiated using the oxidation-reduction pair: cumene hydroperoxide / sodium metabisulfite.
- e) - Une fois la réaction de polymérisation terminée, on retire par distillation sous vide l'ISOPAR™ H et la quasi-totalité de l'eau.e) - Once the polymerization reaction is complete, the ISOPAR ™ H and almost all of the water are distilled off under vacuum.
- f) - On obtient après introduction de 2% massique de Laureth-7 et de 4% massique de la composition tensioactive (C) obtenue à l'exemple A, le latex inverse auto-inversible (4), contenant environ 63% de polymère, qui est peu visqueux, qui s'inverse très rapidement dans l'eau et dont le pouvoir épaississant est important. Sa teneur en eau mesurée par une titrimétrie Karl-Fisher est de 2,2% en poids.f) - is obtained after introduction of 2% by weight of Laureth-7 and 4% by weight of the surfactant composition (C) obtained in Example A, the self-invertible inverse latex (4), containing about 63% of polymer , which is not very viscous, which reverses very quickly in water and whose thickening power is important. Its water content measured by Karl-Fisher titrimetry is 2.2% by weight.
a) - On mesure la viscosité du latex inverse auto-inversible (4) obtenu comme indiqué au paragraphe 1, celle d'une solution aqueuse exempte de chlorure de sodium (sol.10) et celle d'une solution aqueuse contenant 0,1% massique (Sol.11) de chlorure de sodium, lesdites solutions aqueuses comprenant chacune 2% massique dudit latex inverse auto-inversible (4). Les résultats mesurés à l'aide d'une viscosimètre Brookfield RVT sont consignés dans le tableau suivant :
a) - The viscosity of the self-invertible inverse latex (4) obtained as indicated in paragraph 1 is measured, that of an aqueous solution free of sodium chloride (sol.10) and that of an aqueous solution containing 0.1 % mass (Sol.11) of sodium chloride, said aqueous solutions each comprising 2% by weight of said self-invertible inverse latex (4). The results measured using a Brookfield RVT viscometer are recorded in the following table:
- b) - Le temps d'inversion du latex inverse (4), évalué de la même manière qu'à l'exemple 1, est d'environ 30 secondes. b) - The reversal time of the inverse latex (4), evaluated in the same manner as in Example 1, is about 30 seconds.
- c) - La stabilité du latex inverse (4) est évaluée de la même manière qu'à l'exemple 1. Le temps d'apparition des premières gouttes d'huile est de trois semaines. c) - The stability of the inverse latex (4) is evaluated in the same manner as in Example 1. The time of appearance of the first drops of oil is three weeks.
Ajouter B dans A.Add B to A.
Ajouter C dans B, émulsionner B dans A à 70°C, puis ajouter D à 60°C puis E à 30°C.Add C to B, emulsify B in A at 70 ° C, then add D at 60 ° C and then E at 30 ° C.
Emulsionner B dans A à 70°C, ajouter C, puis ajouter D à 60°C puis E à 30°C.Emulsify B in A at 70 ° C, add C, then add D at 60 ° C and then E at 30 ° C.
Introduire B dans A vers 75°C ; ajouter C vers 60°C, puis D vers 45°CIntroduce B in A to 75 ° C; add C to 60 ° C, then D to 45 ° C
Introduire B dans A ; ajouter C puis D, puis E.Introduce B into A; add C then D, then E.
Emulsionner B dans A à 75°C puis ajouter C vers 60°C, puis D vers 30°C et ajuster le pH si nécessaireEmulsify B in A at 75 ° C then add C to 60 ° C, then D to 30 ° C and adjust the pH if necessary
Ajouter B dans A ; puis ajouter au mélange, C puis DAdd B to A; then add to the mixture, C then D
Préparer A ; additionner B, puis C, puis D.Prepare A; add B, then C, then D.
Préparer A ; additionner B, puis C, puis D.Prepare A; add B, then C, then D.
Fondre A à environ 75°C. Emulsionner B dans A à 75°C puis ajouter C vers 60°C, puis D.Melt A at about 75 ° C. Emulsify B in A at 75 ° C then add C to 60 ° C, then D.
Les définitions des produits commerciaux utilisés dans les exemples sont les suivantes : SIMULSOL™ 1293 est de l'huile de castor hydrogénée et polyéthoxylée, avec un indice d'éthoxylation égal à 40, commercialisé par la société SEPPIC.
CAPIGEL™ 98 est un épaississant liquide à base de copolymère acrylate commercialisé par la société SEPPIC.
KETROL™T est de la gomme de xanthane commercialisée par la société KELCO. LANOL™ 99 est de l'isononyl isononanoate commercialisé par la société SEPPIC.
DC1501 est un mélange de cyclopentasiloxane et de diméthiconol commercialisé par la société DOW CHEMICAL.
MONTANOV™ 82 est un agent émulsionnant à base d'alcool cétéarylique et de cocoylglucos ide.
Le MONTANOV™ 68 (cétéaryl glucoside), est une composition auto-émulsionnable telle que décrite dans
Le MICROPEARL™ M 100 est une poudre ultra fine au toucher très doux et à action matifiante commercialisée par la société MATSUMO.
Le SEPICIDE™ CI, imidazolidine urée, est un agent conservateur commercialisé par la société SEPPIC.
PEMULEN™ TR est un polymère acrylique commercialisé par GOODRICH.
Le SIMULSOL™165 est du stéarate de glycérol auto-émulsionnable commercialisée par la société SEPPIC.
Le LANOL™ 1688 est un ester émollient à effet non gras commercialisé par la société SEPPIC.
Le LANOL™ 14M et le LANOL S sont des facteurs de consistance commercialisés par la société SEPPIC.
Le SEPICIDE™ HB, qui est un mélange de phénoxy éthanol, de méthyl paraben, d'éthyl paraben, de propyl paraben et de butyl paraben, est un agent conservateur commercialisé par la société SEPPIC.
Le MONTEINE™ CA est un agent hydratant commercialisé par la société SEPPIC.
Le SCHERCEMOL™ OP est un ester émollient à effet non gras.
Le LANOL™ P est un additif à effet stabilisant commercialisé par la société SEPPIC.
Le PARSOL™ MCX est du (para-méthoxy) cinnamate d'octyle; commercialisé par la société GIVAUDAN.
Le SEPIPERL™ N est un agent nacrant, commercialisé par la société SEPPIC, à base d'un mélange d'alkyl polyglucosides tels que ceux décrits dans
Le MICROPEARL™ SQL est un mélange de micro particules renfermant du squalane qui se libère sous l'action du massage; il est commercialisé par la société MATSUMO.
Le LANOL™ 99 est de l'isononanoate d'isononyle commercialisé par la société SEPPIC.
Le LANOL™ 37T est du triheptanoate de glycérol, commercialisé par la société SEPPIC.
Le SOLAGUM™ L est un carraghénane commercialisé par la société SEPPIC.
Le MARCOL™ 82 est une huile de paraffine commercialisée par la société EXXON. Le LA-NOL™ 84D est du malate de dioctyle commercialisé par la société SEPPIC. Le PARSOL NOX ™ est un filtre solaire commercialisé par la société GIVAUDAN.
EUSOLEX™ 4360 est un filtre solaire commercialisé par la société MERCK.
Le DOW CORNING™ 245 Fluid est de la cyclométhicone, commercialisée par la société DOW CORNING.
Le LIPACIDE™ PVB, est un hydrolysat de protéines de blé acyle commercialisé par la société SEPPIC.
Le MICROPEARL™ LM est un mélange de squalane, de polyméthylméthacrylate et de menthol, commercialisé par la société SEPPIC.
Le SEPICONTROL™ A5 est un mélange capryloy glycine, sarcosine, extrait de cinnamon zylanicum, commercialisé par la société SEPPIC, tel que ceux décrits dans la demande internationale de brevet
Le LANOL™ 2681 est un mélange caprylate, caprate de coprah, commercialisé par la société SEPPIC.
Le MONTANOV™ 202, est une composition APG/alcools gras telle que décrite dans
CAPIGEL ™ 98 is a liquid thickener based on acrylate copolymer marketed by the company SEPPIC.
KETROL ™ T is xanthan gum marketed by the company KELCO. LANOL ™ 99 is isononyl isononanoate marketed by the company SEPPIC.
DC1501 is a mixture of cyclopentasiloxane and dimethiconol marketed by Dow Chemical.
MONTANOV ™ 82 is an emulsifier based on cetearyl alcohol and cocoylglucos ide.
MONTANOV ™ 68 (cetearyl glucoside), is a self-emulsifiable composition as described in
The MICROPEARL ™ M 100 is an ultra-fine powder with a very soft touch and mattifying action marketed by MATSUMO.
SEPICIDE ™ CI, imidazolidine urea, is a preservative marketed by the company SEPPIC.
PEMULEN ™ TR is an acrylic polymer marketed by GOODRICH.
SIMULSOL ™ 165 is self-emulsifiable glycerol stearate marketed by the company SEPPIC.
LANOL ™ 1688 is an emollient non-fat ester marketed by the company SEPPIC.
LANOL ™ 14M and LANOL S are consistency factors marketed by the company SEPPIC.
SEPICIDE ™ HB, which is a mixture of phenoxy ethanol, methyl paraben, ethyl paraben, propyl paraben and butyl paraben, is a preservative marketed by the company SEPPIC.
MONTEINE ™ CA is a moisturizing agent marketed by the company SEPPIC.
SCHERCEMOL ™ OP is a non-greasy emollient ester.
LANOL ™ P is a stabilizing additive marketed by the company SEPPIC.
PARSOL ™ MCX is octyl (para-methoxy) cinnamate; marketed by GIVAUDAN.
SEPIPERL ™ N is a pearlescent agent, sold by the company SEPPIC, based on a mixture of alkyl polyglucosides such as those described in US Pat.
MICROPEARL ™ SQL is a mixture of micro particles containing squalane which is released by the action of massage; it is marketed by MATSUMO.
LANOL ™ 99 is isononyl isononanoate marketed by the company SEPPIC.
LANOL ™ 37T is glycerol triheptanoate, marketed by the company SEPPIC.
SOLAGUM ™ L is a carrageenan marketed by the company SEPPIC.
MARCOL ™ 82 is a paraffin oil marketed by the company EXXON. LA-NOL ™ 84D is dioctyl malate marketed by the company SEPPIC. PARSOL NOX ™ is a sunscreen marketed by the company GIVAUDAN.
EUSOLEX ™ 4360 is a solar filter marketed by MERCK.
DOW CORNING ™ 245 Fluid is cyclomethicone sold by the company Dow Corning.
LIPACIDE ™ PVB is a hydrolyzate of acyl wheat proteins marketed by the company SEPPIC.
MICROPEARL ™ LM is a mixture of squalane, polymethylmethacrylate and menthol, marketed by the company SEPPIC.
SEPICONTROL ™ A5 is a mixture of capryloyl glycine, sarcosine and cinnamon zylanicum extract marketed by the company SEPPIC, such as those described in the international patent application.
LANOL ™ 2681 is a caprylate mixture, copra caprate, marketed by the company SEPPIC.
MONTANOV ™ 202, is an APG / fatty alcohol composition as described in
Claims (16)
- Composition in the form of a self-invertible inverse latex comprising, for 100 % of its weight:a) - from 50 wt.% to 70 wt.% of a cross-linked anionic polyelectrolyte (P) obtained by polymerisation:- of at least one neutral monomer of formula (I):
in which formulae (III) and (IV):- R2 represents a linear or branched alkyl radical comprising 12 carbon atoms,- T4 represents a hydrogen atom or a (-CH2-CH2-O-)m4-H radical in which m4 is an integer greater than or equal to zero and less than or equal to ten,- T5, which is identical to or different from T4, represents a hydrogen atom or a (-CH2-CH2-O-)m5-H radical in which m5 is an integer greater than or equal to zero and less than or equal to ten,- T6, which is identical to or different from T4 and T5, represents a hydrogen atom or a (-CH2-CH2-O-)m6-H radical in which m6 is an integer greater than or equal to zero and less than or equal to ten,- T7, which is identical to or different from T4, T5 and T6, represents a hydrogen atom or a (-CH2-CH2-O-)m7-H radical in which m7 is an integer greater than or equal to zero and less than or equal to ten, it being understood that the sum m4+m5+m6+m7 is greater than 0 and less than or equal to ten;β) - optionally up to 40 mol.% of a compound of formula (III'):
in which formulae (III') and (IV'):- R'2 represents a linear or branched alkyl radical comprising 14 carbon atoms,- T'4 represents a hydrogen atom or a (-CH2-CH2-O-)m4-H radical in which m4 is an integer greater than or equal to zero and less than or equal to ten,- T'5, which is identical to or different from T'4, represents a hydrogen atom or a (-CH2-CH2-O-)m5-H radical in which m5 is an integer greater than or equal to zero and less than or equal to ten,- T'6, which is identical to or different from T'4 and T'5, represents a hydrogen atom or a (-CH2-CH2-O-)m6-H radical in which m6 is an integer greater than or equal to zero and less than or equal to ten, and- T'7, which is identical to or different from T'4, T'5 and T'6, represents a hydrogen atom or a (-CH2-CH2-O-)m7-H radical in which m7 is an integer greater than or equal to zero and less than or equal to ten,- it being understood that the sum m4+m5+m6+m7 is greater than 0 and less than or equal to ten; andγ) - optionally up to 10 mol.% of a compound of formula (III"):
in which formulae (III") and (IV"):- R"2 represents a linear or branched alkyl radical comprising 16 carbon atoms,- T"4 represents a hydrogen atom or a (-CH2-CH2-O-)m4-H radical in which m4 is an integer greater than or equal to zero and less than or equal to ten,- T"5, which is identical to or different from T"4, represents a hydrogen atom or a (-CH2-CH2-O-)m5-H radical in which m5 is an integer greater than or equal to zero and less than or equal to ten,- T"6, which is identical to or different from T"4 and T"5, represents a hydrogen atom or a (-CH2-CH2-O-)m6-H radical in which m6 is an integer greater than or equal to zero and less than or equal to ten, and- T"7, which is identical to or different from T"4, T"5 and T"6, represents a hydrogen atom or a (-CH2-CH2-O-)m7-H radical in which m7 is an integer greater than or equal to zero and less than or equal to ten,- it being understood that the sum m4+m5+m6+m7 is greater than 0 and less than or equal to ten;d) - from 15 wt.% to 45 wt.% of at least one oil, ande) - from 0 wt.% to 5 wt.% of water. - Composition according to claim 1, wherein, in formula (I), the radical R1 represents a lauryl radical or a stearyl radical.
- Composition according to either claim 1 or claim 2, wherein, in formula (I), n is greater than or equal to two and less than or equal to twenty.
- Composition according to any of claims 1 to 3, wherein, in the polyelectrolyte P, the monomer units comprising a strong acid function result from 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid partially or completely salified as sodium salt, as potassium salt or as ammonium salt and the monomer units comprising a weak acid function result from acrylic acid or from methacrylic acid partially salified as sodium salt, as potassium salt or as ammonium salt.
- Composition according to any of claims 1 to 4, wherein the polyelectrolyte P comprises, in mole percent, from 0.5 % to 10 % of a monomer unit resulting from the monomer of formula (I) as defined above.
- Composition according to any of claims 1 to 5, wherein said surfactant composition (C) additionally comprises:3) - up to 5 mol.% of a composition (CV) comprising, per 100 mol.%:α) - from 60 mol.% to 100 mol.% of a compound of formula (V):
- Composition according to any of claims 1 to 6, wherein said surfactant composition (C) additionally comprises:4) - up to 5 mol.% of a composition (CVI) comprising, per 100 mol.%:α) - from 60 mol.% to 100 mol.% of a compound of formula (VI):
R2-(-CH2-CH2-O-)m10-H (VI),
in which R2 represents a linear or branched alkyl radical comprising 12 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten;β) - optionally up to 40 mol.% of a compound of formula (VI'):
R'2-(-CH2-CH2-O-)m10-H (VI'),
in which R'2 represents a linear or branched alkyl radical comprising 14 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten; andγ) - optionally up to 10 mol.% of a compound of formula (VI"):
R"2-(-CH2-CH2-O-)m10-H (VI"),
in which R"2 represents a linear or branched alkyl radical comprising 16 carbon atoms and m10 is an integer greater than or equal to zero and less than or equal to ten. - Composition according to any of claims 1 to 7, wherein said surfactant composition (C) comprises:1) - a proportion of greater than or equal to 20 mol.% and of less than or equal to 50 mol.% of a composition (CII) as defined above;2) - a proportion of greater than or equal to 50 mol.% and of less than or equal to 80 mol.% of a composition (CIII) as defined above.
- Composition according to any of claims 1 to 8, wherein, in said surfactant composition (C):1) - said composition (CII) comprises, per 100 mol.%:α) - from 60 mol.% to 80 mol.% of the compound of formula (II),β) - from 15 mol.% to 30 mol.% of the compound of formula (II'), andγ) - up to 10 mol.% of the compound of formula (II"), and2) - said composition (CIII) comprises, per 100 mol.%:α) - from 60 mol.% to 80 mol.% of the compound of formula (III), of its isomer of formula (IV) or of the mixture of these isomers,β) - from 15 mol.% to 30 mol.% of the compound of formula (III'), of its isomer of formula (IV') or of the mixture of these isomers, andγ) - up to 10 mol.% of the compound of formula (III"), of its isomer of formula (IV") or of the mixture of these isomers.
- Composition according to any of claims 1 to 9, wherein said emulsifying system (S2) of oil-in-water (O/W) type consists of 100 wt.% of said surfactant composition (C).
- Composition according to any of claims 1 to 10, wherein said emulsifying system (S2) of oil-in-water (O/W) type comprises, per 100 % of its weight:- from 10 wt.% to 40 wt.% of heptaethoxylated lauryl alcohol and- from 60 wt.% to 90 wt.% of said surfactant composition (C).
- Composition according to any of claims 1 to 11, wherein, in the polyelectrolyte (P), the monomer unit resulting from the monomer of formula (I) is a monomer unit resulting from tetraethoxylated lauryl acrylate.
- Composition according to claim 12, wherein the polyelectrolyte (P) is selected from:- cross-linked copolymers of acrylic acid partially salified in the sodium salt or ammonium salt form, of acrylamide and of tetraethoxylated lauryl acrylate;- cross-linked copolymers of 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid partially salified in the sodium salt or ammonium salt form, of acrylamide and of tetraethoxylated lauryl acrylate;- cross-linked copolymers of 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid partially salified in the sodium salt or ammonium salt form, of 2-hydroxyethyl acrylate and of tetraethoxylated lauryl acrylate;- cross-linked copolymers of acrylamide, of 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid, of acrylic acid partially salified in the sodium salt or ammonium salt form and of tetraethoxylated lauryl acrylate;- copolymers of 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid partially salified in the sodium salt or ammonium salt form, of acrylamide, of vinylpyrrolidone and of tetraethoxylated lauryl acrylate; and- cross-linked copolymers of 2-methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid partially or completely salified in the sodium salt form, of acrylic acid partially salified in the sodium salt or ammonium salt form, of 2-hydroxyethyl acrylate, of tris(hydroxymethyl)aminomethyl acrylamide and of tetraethoxylated lauryl acrylate.
- Composition according to any of claims 1 to 13, wherein the cross-linked anionic polyelectrolyte (P) comprises, per 100 % of monomers used:- from 40 mol.% to 80 mol.% of monomer units resulting from a monomer comprising a strong acid function;- from 15 mol.% to 55 mol.% of monomer units resulting from a neutral monomer other than the compound of formula (I) as defined above;- from 1 mol.% to 5 mol.% of monomer units resulting from a monomer of formula (I) as defined above.
- Use of the composition according to any of claims 1 to 14 as a thickening and/or emulsifying agent for preparing a cosmetic, dermopharmaceutical or pharmaceutical topical composition.
- Cosmetic, dermopharmaceutical or pharmaceutical topical composition, characterised in that it comprises, as a thickening and/or emulsifying agent, an effective amount of the composition according to any of claims 1 to 14.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PL11730359T PL2566445T3 (en) | 2010-05-06 | 2011-04-11 | Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1053534A FR2959746B1 (en) | 2010-05-06 | 2010-05-06 | NEW AUTO-INVERSIBLE REVERSE LATEX, ITS USE AS A THICKENING AGENT IN A COSMETIC COMPOSITION |
| PCT/FR2011/050816 WO2011138533A1 (en) | 2010-05-06 | 2011-04-11 | Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP2566445A1 EP2566445A1 (en) | 2013-03-13 |
| EP2566445B1 true EP2566445B1 (en) | 2015-02-25 |
Family
ID=42710324
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP11730359.4A Active EP2566445B1 (en) | 2010-05-06 | 2011-04-11 | Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US9101552B2 (en) |
| EP (1) | EP2566445B1 (en) |
| JP (1) | JP5727001B2 (en) |
| KR (1) | KR20130079377A (en) |
| CN (1) | CN102917686B (en) |
| BR (1) | BR112012028442B1 (en) |
| ES (1) | ES2535353T3 (en) |
| FR (1) | FR2959746B1 (en) |
| PL (1) | PL2566445T3 (en) |
| RU (1) | RU2012152456A (en) |
| WO (1) | WO2011138533A1 (en) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2959743B1 (en) * | 2010-05-06 | 2012-06-15 | Soc Dexploitation De Produits Pour Les Industries Chimiques Seppic | COMPOSITION OF POLYALCOXY DERIVATIVES OF TRIMETHYLOLPROPANE AND FATTY ALCOHOLS PREPARED FOR USE AND AS INVERTER USE IN SELF-INVERSIBLE REVERSE LATEXES |
| GB2567198B (en) * | 2017-10-05 | 2022-01-12 | Scott Bader Co | Inverse emulsion thickeners |
| FR3079837B1 (en) * | 2018-04-06 | 2020-03-13 | Societe D'exploitation De Produits Pour Les Industries Chimiques Seppic | SELF-REVERSIBLE REVERSE LATEX COMPRISING POLYGLYCEROL ESTERS, ITS USE AS A THICKENER AND COSMETIC COMPOSITIONS COMPRISING SAME |
| IT201800020872A1 (en) | 2018-12-21 | 2020-06-21 | Lamberti Spa | REVERSE EMULSIONS USEFUL AS THICKENERS FOR COSMETICS |
| FR3104166B1 (en) * | 2019-12-09 | 2021-12-10 | Soc Dexploitation De Produits Pour Les Industries Chimiques Seppic | Detergent composition comprising an inverse latex combining a particular sequestering agent and a polyelectrolyte comprising a weak acid function |
Family Cites Families (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1482515A (en) | 1974-07-24 | 1977-08-10 | Allied Colloids Ltd | Polymers |
| GB8309275D0 (en) | 1983-04-06 | 1983-05-11 | Allied Colloids Ltd | Dissolution of water soluble polymers in water |
| US4554018A (en) | 1984-02-01 | 1985-11-19 | Allied Colloids Limited | Production of polymeric thickeners and their use in printing |
| FR2668080B1 (en) | 1990-10-17 | 1993-08-13 | Seppic Sa | SELF-EMULSIONABLE COMPOSITIONS BASED ON FATTY ALCOHOLS, THEIR PREPARATION PROCESS AND THEIR USE FOR MAKING EMULSIONS. |
| FR2676923B1 (en) | 1991-06-03 | 1995-04-21 | Oreal | COMPOSITION FOR TREATING KERATINIC MATERIALS BASED ON SILICONE AND LATEX. |
| FR2676922B1 (en) | 1991-06-03 | 1995-01-20 | Givaudan Lavirotte | COSMETIC APPLICATIONS OF N-ACYL DERIVATIVES OF MIXTURES OF AMINO ACIDS DERIVED FROM VEGETABLE PROTEIN HYDROLYSATES. |
| FR2681245B1 (en) | 1991-09-17 | 1995-06-09 | Oreal | USE IN COSMETICS OR IN TOPICAL APPLICATION OF AN AQUEOUS DISPERSION BASED ON ORGANOPOLYSILOXANES AND A CROSSLINKED ACRYLAMIDE / ACID 2-ACRYLAMIDO 2-METHYLPROPANE SULFONIC SULFONIC COPOLYMER. |
| JP3555762B2 (en) | 1991-10-16 | 2004-08-18 | リチャードソン、ビックス、インコーポレーテッド | Low pH aqueous cosmetic gel containing nonionic polyacrylamide derivative |
| FR2682679B1 (en) | 1991-10-17 | 1995-06-16 | Seppic Sa | NOVEL ALKYLPOLYOSIDE DERIVATIVES, PROCESSES FOR THEIR PREPARATION AND APPLICATIONS AS SURFACE AGENTS. |
| US5532347A (en) | 1992-04-10 | 1996-07-02 | State Of Oregon, Acting By And Through The Oregon State Board Of Higher Education On Behalf Of The Oregon Health Sciences University, A Non-Profit Organization | DNA encoding α melanocyte stimulating hormone receptor |
| US5232688A (en) | 1992-06-17 | 1993-08-03 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Self-tanner cosmetic compositions |
| FR2698004B1 (en) | 1992-11-13 | 1995-01-20 | Oreal | Aqueous cosmetic or dermatological dispersion for the treatment of hair or skin, based on fatty acid esters of sugar or of alkyl sugar and crosslinked copolymers of acrylamide. |
| FR2698267B1 (en) | 1992-11-24 | 1995-02-10 | Oreal | Dihydroxyacetone-based compositions and their cosmetic use. |
| FR2705673B1 (en) | 1993-05-25 | 1995-07-28 | Givaudan Lavirotte | Compositions comprising amino acid derivatives, processes for their preparation and their uses. |
| FR2706299B1 (en) | 1993-06-10 | 1995-09-01 | Oreal | Cosmetic or dermatological oil-in-water emulsion containing a self-emulsifiable composition based on a fatty alcohol and an alkylpolyoside and a co-emulsifier. |
| FR2709679B1 (en) | 1993-08-06 | 1995-10-06 | Seppic Sa | Concentrated aqueous compositions of alkylpolyglycosides and their uses. |
| FR2712595B1 (en) | 1993-11-19 | 1995-12-22 | Seppic Sa | A concentrate comprising alkyl glycosides and its uses. |
| DE4414426C1 (en) | 1994-04-26 | 1995-09-21 | Zsuzsa Cserhati | Joint prosthesis e.g. for finger joint |
| FR2727626B1 (en) | 1994-12-05 | 1997-01-17 | Oreal | SELF-TANNING COSMETIC COMPOSITIONS BASED ON DIHYDROXYACETONE, ALKYLPOLYOSIDES AND FATTY ALCOHOLS |
| FR2734496B1 (en) | 1995-05-24 | 1997-07-04 | Seppic Sa | EMULSIFYING COMPOSITION BASED ON ALKYLPOLYGLYCOSIDES, AND USES THEREOF |
| DE19523596A1 (en) | 1995-07-03 | 1997-01-09 | Henkel Kgaa | Preparation for the treatment of keratinic fibers |
| JP2000517335A (en) | 1996-09-09 | 2000-12-26 | ソシエテ・デクスプロワタシオン・ド・プロデュイ・プール・レ・ザンデュストリ・シミーク―セピック | Cosmetic use of a compound having a lipoamino acid structure, and a cosmetic composition having some of these compounds and having relaxing activity |
| FR2756195B1 (en) | 1996-11-22 | 1999-02-12 | Seppic Sa | NEW COMPOSITIONS BASED ON ALKYLGLYCOSIDES AND FATTY ALCOHOLS |
| FR2762317B1 (en) | 1997-04-21 | 1999-07-09 | Seppic Sa | COMPOSITION BASED ON ALKYLPOLYGLYCOSIDES AND FATTY ALCOHOLS AND USES THEREOF |
| FR2773656B1 (en) | 1998-01-15 | 2000-02-11 | Alsthom Cge Alcatel | INTELLIGENT GATEWAY BETWEEN A SERVICE CONTROL POINT, AND A SIGNALING NETWORK |
| FR2783417B1 (en) | 1998-09-17 | 2002-06-28 | Oreal | TOPICAL COSMETIC OR DERMATOLOGICAL COMPOSITIONS COMPRISING DENDRITIC POLYESTERS |
| FR2794124B1 (en) * | 1999-05-28 | 2003-03-21 | Seppic Sa | NOVEL INVERSE AUTO INVERSIBLE LATEX, PREPARATION METHOD AND USE IN COSMETICS |
| FR2810883B1 (en) * | 2000-06-28 | 2006-07-28 | Seppic Sa | NOVEL AUTOINVERSIBLE INVERSE LATEX ON FATTY ACID ESTERS, COSMETIC, DERMOCOSMETIC, DERMOPHARMACEUTICAL OR PHARMACEUTICAL COMPOSITIONS COMPRISING THE SAME |
| FR2835252B1 (en) | 2002-01-25 | 2005-08-05 | Seppic Sa | USE OF A PROTEIN KINASE A INACTIFYING COMPOUND IN A COMPOSITION CONTAINING A COSMETICALLY ACCEPTABLE MEDIUM FOR LAMINATING THE SKIN |
| SE524461C2 (en) * | 2002-01-25 | 2004-08-10 | Perstorp Specialty Chem Ab | Chain extended dendritic polyether, composition and use thereof |
| DE10261750A1 (en) * | 2002-12-30 | 2004-07-15 | Basf Ag | Ampholytic copolymer and its use |
| EP1623697A4 (en) * | 2003-05-13 | 2008-03-19 | Nisshin Oillio Group Ltd | Oil-in-water emulsion cosmetics |
| FR2879607B1 (en) * | 2004-12-16 | 2007-03-30 | Seppic Sa | NOVEL CONCENTRATED REVERSE LATEX, PROCESS FOR PREPARING THE SAME, AND USE IN INDUSTRY |
| FR2886300B1 (en) * | 2005-05-25 | 2007-06-29 | Seppic Sa | NEW REVERSE LATEX OF COPOLYMERS OF AMPS AND NN-DIMETHYL ACRYLAMIDE; USE IN COSMETICS |
| EP1932515A1 (en) * | 2006-12-12 | 2008-06-18 | Societe D'exploitation De Produits Pour Les Industries Chimiques, S.E.P.P.I.C. | Inverse latex of anionic polyelectrolytes in silicone oils; cosmetic use thereof |
| FR2959743B1 (en) * | 2010-05-06 | 2012-06-15 | Soc Dexploitation De Produits Pour Les Industries Chimiques Seppic | COMPOSITION OF POLYALCOXY DERIVATIVES OF TRIMETHYLOLPROPANE AND FATTY ALCOHOLS PREPARED FOR USE AND AS INVERTER USE IN SELF-INVERSIBLE REVERSE LATEXES |
-
2010
- 2010-05-06 FR FR1053534A patent/FR2959746B1/en not_active Expired - Fee Related
-
2011
- 2011-04-11 PL PL11730359T patent/PL2566445T3/en unknown
- 2011-04-11 ES ES11730359.4T patent/ES2535353T3/en active Active
- 2011-04-11 US US13/641,969 patent/US9101552B2/en active Active
- 2011-04-11 RU RU2012152456/15A patent/RU2012152456A/en not_active Application Discontinuation
- 2011-04-11 JP JP2013508539A patent/JP5727001B2/en active Active
- 2011-04-11 BR BR112012028442-8A patent/BR112012028442B1/en not_active IP Right Cessation
- 2011-04-11 KR KR1020127028571A patent/KR20130079377A/en not_active Application Discontinuation
- 2011-04-11 CN CN201180022707.7A patent/CN102917686B/en active Active
- 2011-04-11 WO PCT/FR2011/050816 patent/WO2011138533A1/en active Application Filing
- 2011-04-11 EP EP11730359.4A patent/EP2566445B1/en active Active
Also Published As
| Publication number | Publication date |
|---|---|
| EP2566445A1 (en) | 2013-03-13 |
| WO2011138533A1 (en) | 2011-11-10 |
| US9101552B2 (en) | 2015-08-11 |
| JP5727001B2 (en) | 2015-06-03 |
| US20130035401A1 (en) | 2013-02-07 |
| FR2959746B1 (en) | 2012-06-15 |
| ES2535353T3 (en) | 2015-05-08 |
| PL2566445T3 (en) | 2015-08-31 |
| RU2012152456A (en) | 2014-06-20 |
| CN102917686B (en) | 2015-04-15 |
| JP2013527282A (en) | 2013-06-27 |
| FR2959746A1 (en) | 2011-11-11 |
| CN102917686A (en) | 2013-02-06 |
| KR20130079377A (en) | 2013-07-10 |
| BR112012028442B1 (en) | 2018-02-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1828256B1 (en) | Novel concentrated positive latex, method for the preparation thereof, and use thereof in industry | |
| EP2475351B1 (en) | Novel powdered polymer, preparation method thereof and use of same as a thickener | |
| EP1369435B1 (en) | Thickening latex, manufacturing process and cosmetic applications | |
| EP1903060A1 (en) | Novel concentrated inverse latex, production method and use thereof in industry | |
| FR2910899A1 (en) | Anionic polyelectrolyte, useful as thickener, emulsifier and/or a stabilizer e.g. for topical cosmetic composition, comprises terpolymer of monomer having salified free strong acid function with neutral monomer, and alkane-oxy monomer | |
| EP1152023B1 (en) | Inverse latex based on squalane or hydrogenated polyisobutene and cosmetic compositions, dermocosmetics, dermopharmaceuticals or pharmaceutical compositions containing them | |
| FR2886300A1 (en) | NEW REVERSE LATEX OF COPOLYMERS OF AMPS AND NN-DIMETHYL ACRYLAMIDE; USE IN COSMETICS | |
| EP1010708B1 (en) | Use in cosmetics of an inverse latex stable against electrolytes | |
| EP2293771B1 (en) | Novel inverse latices that are free of oxyethylene derivatives, and cosmetic, dermocosmetic, dermopharmaceutical or pharmaceutical compositions comprising same | |
| EP1152022A1 (en) | Inverse latex based on white mineral oils, squalane, hydrogenated polyisobutene, isohexadecane or isododecane, cosmetic compositions, dermocosmetics, dermopharmaceuticals or pharmaceutical compositions containing them | |
| FR2943677A1 (en) | NEW THICKENING POLYMER IN POWDER FORM | |
| WO2009156690A2 (en) | Novel inverse latices based on fatty alcohol ethers, and cosmetic, dermocosmetic, dermopharmaceutical or pharmaceutical compositions comprising same | |
| EP1166771A1 (en) | Invers autoinversible latices based on fatty acid esters and cosmetic, dermocosmetic, dermopharmaceutic or pharmaceutic compositions | |
| EP2566445B1 (en) | Novel self-reversible reverse latex, and use thereof as a thickening agent in a cosmetic composition | |
| EP2646510B1 (en) | Novel polymeric thickening agent free of acrylamide fragments, method for the preparation thereof, and composition containing same | |
| EP1149117A1 (en) | Novel alkanolamide-free thickening latex | |
| EP1371692B1 (en) | Auto inversible inverse Microlatex, process for its perparaion and its utilization | |
| WO2001035922A1 (en) | Novel positive latex and use in cosmetics | |
| FR2992323A1 (en) | NEW SURFACE-FREE REVERSE LATEX FREE OF SURFACTANT, ITS USE AS A THICKENING AGENT IN A COSMETIC COMPOSITION |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20121206 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| DAX | Request for extension of the european patent (deleted) | ||
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20140919 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602011014020 Country of ref document: DE Effective date: 20150409 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 711312 Country of ref document: AT Kind code of ref document: T Effective date: 20150415 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2535353 Country of ref document: ES Kind code of ref document: T3 Effective date: 20150508 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: VDEP Effective date: 20150225 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 711312 Country of ref document: AT Kind code of ref document: T Effective date: 20150225 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150525 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150526 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150625 |
|
| REG | Reference to a national code |
Ref country code: PL Ref legal event code: T3 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602011014020 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: LU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150411 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| 26N | No opposition filed |
Effective date: 20151126 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150411 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20110411 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: PL Payment date: 20170327 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20170329 Year of fee payment: 7 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150625 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20150430 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 8 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20150225 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20190521 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20190418 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180411 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200430 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200430 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20210901 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200412 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20180411 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20230421 Year of fee payment: 13 Ref country code: FR Payment date: 20230420 Year of fee payment: 13 Ref country code: DE Payment date: 20230420 Year of fee payment: 13 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230419 Year of fee payment: 13 |