EP1470149B1 - Procede d'extraction et de purification de phytosterols et de phytostanols extraits de poix de tall oil - Google Patents
Procede d'extraction et de purification de phytosterols et de phytostanols extraits de poix de tall oil Download PDFInfo
- Publication number
- EP1470149B1 EP1470149B1 EP03701377A EP03701377A EP1470149B1 EP 1470149 B1 EP1470149 B1 EP 1470149B1 EP 03701377 A EP03701377 A EP 03701377A EP 03701377 A EP03701377 A EP 03701377A EP 1470149 B1 EP1470149 B1 EP 1470149B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- pitch
- phytosterols
- phytostanols
- water
- acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Revoked
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J9/00—Normal steroids containing carbon, hydrogen, halogen or oxygen substituted in position 17 beta by a chain of more than two carbon atoms, e.g. cholane, cholestane, coprostane
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B3/00—Refining fats or fatty oils
- C11B3/001—Refining fats or fatty oils by a combination of two or more of the means hereafter
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B3/00—Refining fats or fatty oils
- C11B3/02—Refining fats or fatty oils by chemical reaction
- C11B3/06—Refining fats or fatty oils by chemical reaction with bases
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11B—PRODUCING, e.g. BY PRESSING RAW MATERIALS OR BY EXTRACTION FROM WASTE MATERIALS, REFINING OR PRESERVING FATS, FATTY SUBSTANCES, e.g. LANOLIN, FATTY OILS OR WAXES; ESSENTIAL OILS; PERFUMES
- C11B3/00—Refining fats or fatty oils
- C11B3/12—Refining fats or fatty oils by distillation
Definitions
- This invention relates to the separation and purification of unsaponifiables, such as phytosterols and phytostanols, from tall oil pitch.
- Sterols are naturally occurring compounds that perform many critical cellular functions.
- Phytosterols such as campesterol, stigmasterol, campestanol and beta-sitosterol in plants, ergosterol in fungi and cholesterol in animals are each primary components of cellular and sub-cellular membranes in their respective cell types.
- the dietary source of phytosterols in humans comes form plant material i.e. vegetables and plant oils.
- the average Western diet contains about 60-80 mg of phytosterols per day, which can be contrasted with a vegetarian diet, which provides about 500 mg per day.
- these dietary plant sterols have received a great deal of attention because of their possible anti-cancer properties and their ability to decrease cholesterol levels when fed to a number of mammalian species, including humans.
- phytosterols offer a unique combination of long-term safety, efficacy, and versatility in human treatment.
- the ongoing challenges with respect to phytosterols is in their isolation, extraction and purification from plant sources, and in determining additional sources which are cost-effective and manageable on a large-scale.
- phytosterols have been isolated from sources such as corn oil, wheat germ oil, soya bean pitch and corn oil pitch.
- tall oil pitch which is obtained during the process of preparing paper from wood, particularly pine wood, has been used as a phytosterol source.
- wood chips are digested or cooked for 2 hours at 170° C in aqueous liquor containing sodium hydroxide and sodium sulfide. The digestion delignifies the wood chips and gives rise to cellulose pulp, sodium rosin soaps, sodium fatty soaps, lignin degradation products and a number of other chemicals.
- the sodium rosin soaps, sodium fatty soaps and other hydrophobic compounds, which remain the in cooking liquor, are separated out by concentrating the liquor causing them to skim or float to the surface (hence the term “skimmings").
- Skimmings generally comprise, along with sodium rosin soaps and sodium fatty soaps, hydrophobic compounds such as phytosterols, phytostanols, esters, fatty alcohols, waxes, and terpenes, collectively often referred to as the unsaponifiable fraction.
- hydrophobic compounds such as phytosterols, phytostanols, esters, fatty alcohols, waxes, and terpenes, collectively often referred to as the unsaponifiable fraction.
- the result is crude tall oil. This is then distilled to remove the volatile materials leaving a "pitch" as the residue.
- Phytosterols and their saturated analogues can be isolated from either the skimming or the pitch.
- Oy Kaukas AB in Finland has been practising the commercial extraction of phytosterols from skimming soap since 1981. Exemplary patents in this area include: US Patent No.
- United States Patent No. 3,840,570 to Jullan provides a process for preparing sterols from tall oil pitch by extraction in a water - alcohol - hydrocarbon mixture followed by saponification and subsequent purification.
- the starting material in this process is tall oil pitch from which are extracted phytosterols and various impurities. It is recognized that, in any tall oil pitch purification process, the long-chain alcohol and acid impurities are particularly difficult to separate from the sterols (which are, themselves, high molecular weight alcohols). This procedure is cumbersome as it involves several solvent extraction steps with different polar and non-polar solvents. Solvent recovery would be necessarily complex.
- the bottom fraction of the first evaporator is then fed into a second thin film evaporator in which the unsaponifiables, including phytosterols are removed as light ends and a molten soap is recovered as the bottom fraction.
- Lehtinen teaches the recovery of fatty acids and rosin acids by reacting the pitch with an alkali at 200-300° C, in the amount of 5 to 25% tall oil pitch, prior to vacuum distillation of the heated mixture to recover the fatty acids and rosin acids in the distillate fraction
- the present invention provides a process for isolating and purifying phytosterols and phytostanols from tall oil pitch which comprises:
- the present invention also comprises compositions of phytosterols and/or phytostanols prepared according to the process described herein.
- the present invention provides a unique method for the processing of tall oil pitch for the specific purpose of separating the phytosterols and phytostanols therefrom.
- the term "tall oil pitch” or "pitch”, hereinafter, is understood to mean the dark, tarry residue product of the distillation of crude tall oil, the latter having been produced by acidulation of skimming soap.
- Skimming soap in turn, is produced by the evaporation of black liquor, one of the products of the "Kraft Process" in which wood chips are digested with an alkaline solution.
- the steps of producing pitch from wood chips are well known and practised in the field and will not be elaborated upon herein.
- the wood chips from which the starting material (pitch) of the present invention originally derived may be from any hard wood or soft wood variety of tree including, but not limited to, fir, cedar, pine, spruce, oak, hemlock and poplar. Most preferably, the chips from which the pitch is produced are from any Pacific Northwest or Southeast American or European forest variety of woods.
- Tall oil pitch is known to include a variety of free and esterified phytosterols including, but not limited to sitosterol, stigmasterol, campesterol and their saturated equivalents.
- Free sterols and stanols, once extracted and purified from pitch can be used as is in pharmaceuticals, nutraceuticals, foods, beverages and the like or can be chemically modified to confer properties such as stability or solubility. Examples of such manipulation are provided in WO-A-01/00653 , WO-A-00/78789 , WO-A-01/32679 , WO-A-99/63841 , WO-A-01/66560 , and US Patent No. 6,087,353 , all of which are assigned to Forbes Medi-Tech Inc.
- the first step of the process of the present invention which is critical, is feeding the pitch into a first distillation column and thereafter distilling the pitch.
- This preliminary distillation step serves two purposes. Firstly, excess rosin acids and fatty acids are removed thereby making the subsequent saponification step more efficient in terms of conversion of steryl esters to free sterols. Secondly, this distillation reduces the amount of alkali metal base required for saponification and the amount of acid required for the subsequent neutralization.
- the pitch is distilled to achieve an acid value of less than 40, most preferably less than 30. This step is particularly important in pitch sources having high acid values.
- the distillation column may be selected from the group consisting of short path distillation columns, wiped film evaporation columns, thin film evaporation columns and molecular distillation columns.
- the distillation column used in this step is a wiped film evaporation column. No where in any of the prior processes is this pre-treatment of the pitch used.
- the second step of the process of the present invention is saponification of the distilled pitch by adding, an aqueous solution of one or more alkali metal bases.
- Preferred bases include sodium hydroxide, potassium hydroxide or combinations of both.
- the weight percentage of alkali metal to pitch should be in the range of 1% to 30%. More preferably, the range is 1 to 10% or 1 to 15% on an anhydrous basis.
- a more alkali metal base and longer reaction times must be used in order to attain satisfactory conversion of the steryl ester to free sterol.
- a pitch source with an acid value of 50 would require that the weight percentage of alkali metal to pitch be in the range of 20% to 25%.
- saponification is conducted at a temperature in the range of 100°C to 250°C for a period in the range of 60 to 300 minutes, more preferably 120-240 minutes.
- the third step is neutralizing the saponified pitch with one or more acids to achieve an ending pH of between 5.8 and 6.3. Achieving a pH within this range is critical. A higher pH will result in difficulty in the subsequent water removal steps. A lower pH will catalyze the reversion of the free sterols to their esterified form during storage and handling thereby significantly reducing end yield of free sterols.
- the neutralization is carried out at a temperature in the range of 90°C to 130°C, more preferably in the range of 100°C to 120°C, more preferably 101°C to 120°C and most preferably 105°C to 118°C for a period of 1 to 10 hours.
- Temperatures in the higher end of the range are preferred as this reduces viscosity and allows for more efficient mixing of the pitch and acid thereby facilitating more rapid neutralization.
- Acids which may be used include all organic and mineral acids, including, but not limited to sulfuric acid, hydrochloric acid, phosphoric acid or any combination thereof.
- the neutralization step is carried out under vigorous agitation and/or mixing. Both batch and continuous processes may be used. In the case of a continuous system, it is preferred to use a mixer at this stage. No disclosure of the preferred higher neutralization temperature is made in WO-A-99/42471 .
- the fourth step is allowing the neutralized pitch to settle for a period of at least 12 hours or until the water content of the pitch, on phase separation, is less than 15%, thereby forming a settled pitch and a water phase.
- water added during the prior steps of saponification and neutralization is removed, preferably by phase separation and evaporation (stripping). This settling can be achieved by holding the neutralized pitch in a vessel for the required period of time without agitation. Settling may occur in the same vessel as the prior saponification and neutralization steps or in a separate vessel.
- the water content on completion of this step must be less than 15%, more preferably less than 10%, otherwise the pitch will be too viscous after final water removal and to difficult to process further downstream due to the high neutralization salts content. It is preferred that the temperature for this settling step is maintained at the temperature of the prior neutralizing step. This type of settling step or an appreciation of its' benefits are not described in any of the prior publications.
- a second phase separation step wherein the water phase removed in the first settling process is transferred to another vessel for secondary phase separation.
- a "rag" layer or emulsion exists between the water phase and the organic phase, which should be removed to improve the quality of the pitch before the downstream distillation steps.
- the rag layer is allowed to separate by further “settling” without agitation. It is preferred that the temperature for this second phase separation step is maintained at the temperature of the prior settling step
- the pitch must be further dehydrated i.e. substantially all of the remaining water from the settled pitch must be removed.
- This may be achieved by any means which facilitates bulk disengagement of water from the organic phase. For example, heating at a sufficient temperature or heating under vacuum conditions.
- water may be removed using a pressure strip wherein the pressure is no greater than atmospheric and the temperature is maintained below 105°C in order to prevent reversion of the free sterols to steryl esters as the water is removed.
- the temperature of the pitch after this dehydration step be cooled to less than about 80°C, as above noted, to avoid reversion of the free sterols to steryl esters. Furthermore, if the dehydrated pitch is to be stored prior to the initiation of the downstream processing steps, it should be cooled and maintained at a temperature of approximately 60°C or less.
- modified pitch refers to a pitch which has, at the very least, been subject to:
- the "modified" pitch is in a form most appropriate and effective for the two subsequent vacuum distillation steps (the second and third of the process) which follow.
- Falling short path distillation columns with or without scraper, flat, rotary or others, short path distillation column centrifuges, multi-stage short path distillation columns, molecular distillation columns, wiped film evaporation columns, and thin film evaporation columns are all adequate for use within the present invention.
- wiped film evaporators or short path distillation columns are used. Distillation conditions are described in more detail below.
- a degasser may be operated just prior to the second stage of evaporative distillation to assist in the removal of residual water and light boiling point components.
- the light phase distillate resulting from the third distillation comprises free phytosterols and/or phytostanols. Subsequently, it is immediately dissolved in a solvent comprising at least one alcohol to produce a solution of phytosterols and/or phytostanols. The solution is then cooled to form a slurry with phytosterols and/or phytostanols crystallized therein and lastly, the slurry is washed and filtered to isolate the crystallized phytosterols and/or phytostanols from the filtrate.
- the solvent used to dissolve the light phase distillate comprises at least one low molecular weight monohydric alcohol.
- Suitable solvents include, but are not limited to, alcohols such as methanol, ethanol, isopropanol, and acetate esters thereof, ketones such as acetone, methyl ethyl ketone (MEK), methylisobutylketone (MIBK), C1 to C8 hydrocarbons or mixtures thereof.
- alcohols such as methanol, ethanol, isopropanol, and acetate esters thereof
- ketones such as acetone, methyl ethyl ketone (MEK), methylisobutylketone (MIBK), C1 to C8 hydrocarbons or mixtures thereof.
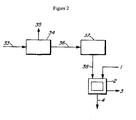
- Tall oil pitch 1 is introduced into a wiped film evaporator 2 and distilled to remove excess fatty acids and rosin acids 3, thereby producing distilled pitch 4.
- this evaporator operates in the range of 100 to 10,000 microns pressure and at a temperature in the range of 240° to 285°C. Generally, depending on the flow rate and the pitch source, it takes less than a minute to achieve the desired acid value of 40 or less.
- the acid value of the pitch may be assessed by taking a sample and titrating.
- Distilled pitch 4 is then added with an alkali metal base 5 into reactor 6.
- the amount of alkali metal base relative to the distilled pitch preferably should be sufficient to facilitate substantially complete or complete saponification of the distilled pitch.
- a water solution of an alkali metal base such as sodium hydroxide, potassium hydroxide or a combination whereof is preferred. These compounds or combinations will provide a relatively high alkalinity for a relatively reasonable cost. If such compounds or combinations are used, then the stoichiometric proportion of alkali metal base 5 to distilled pitch 4 theoretically that is required to achieve complete conversion typically may be approximately 1% by weight.
- Factors that may impact the precise amount of base to be used include the specific characteristics of tall oil pitch 1 and distilled pitch 4 (which characteristics may differ from batch to batch and source to source) and most importantly, the acid value of the tall oil pitch. For pitch sources which have a high acid value, more base and a longer reaction time are required in order to attain satisfactory conversion of the steryl esters to free sterols.
- the amount of base required at this saponification step is less than that required in prior saponification processes due to the fact that tall oil pitch 1 is distilled before saponification. This is due, in part, to the fact that, in prior known processes, a significant amount of the base is consumed in reaction with other components of the pitch such as rosin acids and fatty acids 3. A substantial portion of these unwanted components are removed in evaporator 2 freeing the base to react with the steryl esters.
- the proportion of alkali metal base 5 to distilled pitch 4 may be in the range of 1 to 15% by weight.
- distilled pitch 4 is saponified with 50% caustic (sodium hydroxide) diluted to a concentration of from between 7 to 12%, most preferably 9.5%.
- Mixing is sustained in a reactor 6 with sufficient vigor to maintain contact between distilled pitch 4 and alkali base metal 5.
- an operating temperature in the range of 100° to 250°C, more specifically from 120° to 160°C and most specifically 145°C for a period of time in the range of 120 to 240 minutes will suffice to facilitate the desired conversion.
- saponified pitch 7 is discharged into second reactor 9 for the neutralization step.
- Acid 8 is added to reactor 9 for this purpose.
- this neutralization step occurs in reactor 6 (acid 8 is added directly to reactor 6), the same vessel as used in the saponification step. Either way, it is critical that the ending pH of this step fall within the range of 5.8 to 6.3. A higher pH will result in difficulty in the subsequent water removal steps. A lower pH will catalyze the reversion of the free sterols to their esterified form during storage and handling thereby significantly reducing end yield of free sterols.
- the neutralization is carried out at a temperature in the range of 90°C to 130°C, more preferably in the range of 100°C to 120°C. for a period of 1 to 10 hours. It is most preferred that the temperature be greater than 100°C. Temperatures in the higher end of the range are preferred as this reduces viscosity and allows for more efficient mixing of the pitch and acid thereby facilitating more rapid neutralization. Continuous as opposed to batch neutralization is preferred.
- Acid 8 may be a simple organic acid such as acetic acid or formic acid, both of which are commercially practical.
- acid 8 may be a mineral acid such as sulphuric acid, hydrochloric acid or phosphoric acid. These are relatively strong mineral acids and are favoured over weaker acids such as boric acid.
- acid 8 is either 75% or 85% phosphoric acid or 93-98% sulphuric acid.
- Neutralized pitch 10 is introduced into settling vessel 11 and held with no agitation for a period of at least 12 hours or until the water content of the neutralized pitch is less than 15%, preferably less than 10% by weight thereby yielding settled pitch 13 and water phase 12, the latter of which is discarded.
- Water content may be measured by Krlki titration.
- this settling step may occur in either reactors 6 or 9.
- settled pitch 13 is subject to a secondary phase separation in receiving tank 14 to remove rag layer 15 and yield ultra-settled pitch 16.
- the pitch is held without agitation and for a period of 2 to 24 hours depending on the vessel size and geometry.
- ultra-settled pitch 16 is transferred to reactor 17 and is subject to either vacuum or atmospheric pressure stripping i.e. to strip or evaporate remaining water thereby yielding modified pitch 19.
- the temperature in reactor 17 is maintained below 105°C and water 18 is removed by atmospheric stripping. This may be batch or continous, although the latter is preferred. Temperatures below 105°C are preferred for this embodiment, as higher temperatures will cause reversion of some sterols to their ester form.
- water 18 is removed by vacuum stripping ultra-settled pitch 16 i.e.
- modified pitch 19 is immediately cooled to a temperature of less than about 80°C in order to prevent or minimize reversion of free sterols to steryl esters.
- Modified pitch 19 is introduced into a low pressure wiped film evaporator 21 for the removal of light ends 23. These light ends will comprise the fatty acids and rosin acids which were not removed in the pre-saponification distillation.
- the bottom fraction 22 contains the free phytosterols and is removed from evaporator 21 and moved into a second low pressure wiped film evaporator 24.
- Evaporator 24 serves to distill free phytosterols present in fraction 22 into light phase distillate 25.
- Distillate 25 also comprises fatty alcohols, fatty acids, rosin acids and high molecular weight wax esters.
- a bottom fraction 26 is stored and may be used as a fuel or feedstock for other industries.
- the specific reaction conditions within each of evaporators 21 and 24 will vary depending on the characteristics and type of the source pitch as well as on the type of evaporator employed. These conditions may also vary depending on whether the modified pitch is "degassed" prior to the introduction of modified pitch 19 into evaporator 21.
- a degasser is used (not shown in figures) to remove residual water and other light boiling point components. Generally, the degasser operates at approximately 1000 to 10,000 microns and 100°C to 175°C.
- the preferred range of distillation conditions within evaporator 21 are as follows: temperature from 190°C to 230°C and pressure from 1000 to 15,000 microns.
- the preferred range of distillation conditions within evaporator 24 are as follows: temperature from 250° to 315°C and pressure from 100 to 5,000 microns
- control of the feed rate into evaporators 21 and 24 is important due to the potential fouling of the cold traps within the evaporators that protect the vacuum systems. Too high a feed rate will foul the cold traps and cause operation interruptions. Too low a feed rate will make the process commercially non-viable. However, it is well within the purview of one skilled in the art to maximize these conditions.
- control of the sterol content in the distillation cuts produced in evaporators 21 and 24 may be measured by GC analysis and this information used to control temperature and feed rate. Once again, it is well within the purview of one skilled in the art to maximize these conditions.
- Light phase distillate 25 is introduced into a further reactor 27 where it is heated and stirred until dissolution has occurred in an added solvent 28.
- Solvent 28 may include alcohol and may include water. Effective dissolution of free sterols, has been found to occur at over 65°C, more preferably over 70°C and using a solvent to distillate ratio of 0.5-1.5. Other temperatures may be used, however the solubility of phytosterols will decrease as the temperature is lowered. To some extent, the preferred temperature depends on the solvent to distillate ratio. The more solvent that is used, the lower the distillation temperature and vice versa. However, a higher solvent amount negatively impacts yield so a balance must be sought. It is well within the purview of one skilled in the art to find this "balance".
- the temperature at which crystallization is effected may be in the range of 0 to 40°C (the more impurities, the higher temperature required for crystallization).
- solvent 31 is the same solvent as used in the crystallization step (solvent 28), the solvent wash is at ambient temperature and the wash ratio is 0.5:1 to 1.5-1 relative to the slurry flow rate to apparatus 30. Drying occurs on a continuous dryer yielding purified phytosterols and phytostanols 32 and spent filtrate 33.
- spent filtrate 33 is recovered, stripped of solvent 35 in reactor 34 (a solvent recycle system) yielding stripped filtrate 36, and then thermally treated to convert any free sterols to steryl esters in vessel 37.
- the product 38 of this recycling process can then be fed concurrently into pre-saponification distillation column 2.
- the neutralized mass was heated, with stirring, to 105°C where residual water began to boil,away. Temperature was gradually increased to 135°C during which approximately 100 ml water was collected. A vacuum was gradually pulled on the system by means of a mechanical pump. An end vacuum of 27 in. Hg. was attained during which an additional 25 ml of water was collected. The vacuum was relieved and products collected. A total of 131 g stripped water and 879 g modified pitch were obtained. GC analysis of the modified pitch showed 154 mg/g (15.4%) sterols as the free alcohol. The modified pitch thus obtained is suitable as a feed for high vacuum distillation equipment for the purpose of further enriching the sterol component.
- the modified pitch produced by the method of Example 1 was charged to the feed pot of a laboratory-scale short-path distillation apparatus (UIC model KD-6, stainless steel).
- This apparatus is well know to those practiced in the art and consists of a heated cylindrical evaporator and coaxial condenser.
- a mechanical vacuum pump provides the means to operate the system at reduced pressures in the range 0.01 mbar to 1,000 mbar.
- a wiper mechanism spreads the liquid feed material into a thin film on the evaporator surface thereby effecting a portion of the feed material to be evaporated.
- the vaporized feed material is conveyed by gaseous diffusion to the coaxial condenser surface where it is condensed to a liquid phase known heretofore as distillate.
- the non-vaporized portion of the feed flows by gravity and the pumping action of the wipers to a collection point where it is removed from the system by means of a mechanical pump. Similarly, the distillate is collected and removed.
- the thus described apparatus therefore provides a means of separating a multi-component feed into distillate and residue fractions based upon the various boiling points and gas-liquid equilibrium behavior of the components. Additional evaporation steps may be employed to separate the distillate and/or residue into further fractions. These additional steps may achieved by employing additional short-path distillation apparatus or by passing the desired fractions through a single short-path unit in batch-wise fashion.
- the optimal distillate to residue ratio of this initial separation is variable and depends upon the levels of these light-boilers in the feed.
- the relatively mild conditions and low mass fraction of distillate taken in the pre-cut results in only minimal sterol losses.
- the 95% residue fraction obtained in the pre-cut operation was again fed to the short-path distillation apparatus.
- Evaporator temperatures in the range 240 - 260°C and pressures in the range 0.5 - 1.5 mbar were employed to achieve a distillate to residue ratio of approximately 40:60.
- This nominal 40% distillate fraction is variable and must be determined experimentally for various tall oil pitch feedstocks.
- the nominal 40% distillate fraction was analyzed by GC to contain 28.2% free sterols.
- the thus obtained distillate is suitable for subsequent purification by solvent crystallization. It is to be understood that higher recoveries of sterol in the distillate fractions are possible, but higher levels of co-boiling impurities which may also be present in the distillate, may have a deleterious effect on the purity of the final product obtained.
- Example 2 In a 250 ml Erlenmeyer Flask, 40.0 g of the the nominal 40% distillate obtained in Example 2 were combined with 60.0 g of a solvent mixture comprised of 80%methanol and 20% iso-propanol. The mixture was heated on a hot plate to just below boiling (ca. 60 - 65°C) thereby effecting complete dissolution of the distillate into the solvent resulting in a clear caromel-colored liquid with no visible solids. The solution was allowed to cool to room temperature (ca. 20 - 25°C) with occasional swirling. Needle-like crystals began to form in the solution at approximately 55°C which rapidly attained a thickened slurry-like character.
- the remaining organic layer was sampled for water content and showed 4.2% water by Karl Fischer Titration.
- the organic layer was heated at atmospheric pressure by means of steam pressure on an internal coil to a final temperature of 235°F.
- the residual water was flashed off and condensed by means on an overhead condenser. Stripping time was approx. 2 hr.
- a final sample of Modified Pitch was taken and showed a water content of 0.98%. Further gas chromatigraphic analysis showed 16.84% total sterols (esters and alcohol combined) and 15.16% free sterols (alcohol form only).
- Modified Pitch such as that prepared in Example 4. was fed continuously to a two-stage short-path distillation system (UIC KD-1200, 12 m 2 evaporator surfaces).
- the system is connected serially, with the residue (heavy) stream from the first stage feeding forward to the second stage.
- the temperature of the evaporating surfaces is controlled by means of a hot-oil circulation system.
- the condensing surfaces for each stage may be controlled independently by means of a tempered-water circulation system. Vacuum may be drawn on either stage independently by means of mechanical vacuum pumps equipped with 2-stage Roots-type boosters. Residual water and dissolve gasses are disengaged from the feed prior to the first evaporator stage,by means of a flash tank.
- the temperature of the feed Prior to the flashing, the temperature of the feed may be heated above its storage temperature by means of a steam-heated shell and tube heat exchanger.
- the following process parameters represent average values over a 24 hr. continuous period: Feed Temp: 278°F Feed Rate: 33.5 lb/min. Evap. 1 Temp: 380°F Evap. 1 Pressure: 4.5 mm Hg abs. Cond. 1 Temp: 75°F Dist. 1 Flow: 3.4 lb/min Residue 1 Flow: 30.1 lb/min. Evap. 2 Temp: 511°F Evap. 2 Pressure: 1.6 mm Hg abs. Cond. 2 Temp: 113°F Dist. 2 Flow 13.6 Ib/min. Residue 2 Flow: 16.5 Ib/min.
- 20,000 lb. of sterol-enriched distillate such as produced in Example 5 was charged to a 6,500 gal. jacketed stainless steel stirred-tank reactor.
- 20,000 lb. of a solvent blend of composition 80% methanol and 20% iso-propanol was likewise charged.
- the mixture was heated by means of steam pressure on the jacket to a temperature of 160°F and held for 1 hr. under moderate agitation.
- the heated mixture was fed at 100 lb/min to a two-stage continuous scraped-surface crystallizer (Armstrong) of stainless steel construction.
- the first crystallizer stage was cooled by means of cooling-tower water of 78°F average temperature on its jacket.
- the second stage jacket was connected to a chilled-water system maintained at 45°F.
- Average interstage temperature of the crystallizer mass was 122°F. Average exit temperature was 85°F.
- the thus-crystallized slurry was collected in a 6,500gal. stainless steel buffer tank equipped with agitator and recirculation pump. The slurry was fed to a continuous, vapor-tight vacuum-belt filter (Pannevis) at an average rate of 35 lb/min. The vacuum action of the filter serves to disengage the sterol crystals from the mother liquor.
- Various devices such as overflow weirs and spray bars enable the filter cake to be washed with fresh solvent. The filter cake is washed and dried to an average moisture of 30-40% before being discharged from the filter via a rotary airlock valve.
- the discharged wet cake is fed directly to a continuous rotary tray dryer (Wyssmont Turbo-Dryer) whereby the residual solvent is removed to a level of ⁇ 1%.
- the dried sterol crystals are discharged from the dryer via a second rotary airlock valve where they may be collected in a suitable container or further processed. In the present case, 3088 lbs. of dried sterol crystalline powder was obtained. GC analysis showed 96.2% sterols. Melt point of the sterols was 138.4°C.
Claims (18)
- Procédé d'isolement et de purification de phytostérols et de phytostanols à partir de brai de tallol, ledit procédé comprenant :a) l'introduction du brai dans une première colonne de distillation ;b) la distillation du brai pour éliminer les acides de colophane et les acides gras en excès et obtenir un brai distillé ;c) la saponification du brai distillé par une solution aqueuse d'une ou de plusieurs bases métalliques alcalines pour obtenir un brai saponifié ;d) la neutralisation du brai saponifié par une quantité d'acide suffisante pour obtenir un pH final compris entre 5,8 et 6,3, formant ainsi un brai neutralisé ;e) la décantation du brai neutralisé pendant une durée d'au moins 12 heures ou jusqu'à ce que la teneur en eau du brai soit inférieure à 15 % après décantation, pour obtenir un brai décanté et une phase aqueuse ;f) l'élimination de la majorité de l'eau restante du brai décanté pour former un brai modifié ;g) la distillation du brai modifié dans une seconde colonne de distillation pour éliminer les fractions légères du brai modifié et obtenir une fraction basse comprenant des phytostérols et/ou des phytostanols libres ;h) la distillation de la fraction basse seule dans une troisième colonne de distillation pour obtenir un distillat de phase légère comprenant des phytostérols et/ou des phytostanols libres ;i) la dissolution du distillat de phase légère seul dans un solvant comprenant au moins un alcool pour obtenir une solution de phytostérols et/ou de phytostanols ;j) le refroidissement de la solution pour former une pulpe contenant des phytostérols et/ou des phytostanols cristallisés ; etk) le lavage, la filtration et le séchage de la pulpe pour isoler les phytostérols et/ou phytostanols cristallisés du filtrat.
- Procédé selon la revendication 1, dans lequel, dans l'étape b), le brai est distillé pour obtenir un indice d'acidité inférieur à 40.
- Procédé selon la revendication 1, dans laquel, dans l'étape b), le brai est distillé pour obtenir un indice d'acidité inférieur à 30.
- Procédé selon la revendication 1, dans lequel les colonnes de distillation des étapes b), g) et h) sont choisies au sein du groupe constitué par les colonnes de distillation de type court, les colonnes d'évaporation à film raclé, les colonnes d'évaporation à couche mince et les colonnes de distillation moléculaire.
- Procédé selon la revendication 1, dans lequel la colonne de distillation des étapes b), g) et h) est une colonne d'évaporation à film raclé.
- Procédé selon la revendication 1, dans lequel un courant d'alimentation supplémentaire opposé vient alimenter la première colonne de distillation, ledit courant d'alimentation étant constitué du filtrat de l'étape k), caractérisé en ce que le filtrat est prétraité pour en éliminer les solvants et convertir la majorité des stérols libres qu'il contient en esters stéryliques.
- Procédé selon la revendication 1, dans lequel, dans l'étape d), le brai saponifié est neutralisé à une température supérieure à 100 °C pendant une durée comprise entre 1 et 10 heures.
- Procédé selon la revendication 1, dans lequel les étapes c) et d) sont effectuées dans le même réacteur.
- Procédé selon la revendication 1, dans lequel l'étape d) est mise en oeuvre sous agitation vigoureuse.
- Procédé selon la revendication 1, dans lequel, dans l'étape d), l'acide et le brai sont mélangés.
- Procédé selon la revendication 1, dans lequel l'étape e) est mise en oeuvre sans agitation.
- Procédé selon la revendication 1, dans lequel une étape supplémentaire est pratiquée après l'étape e), ladite étape supplémentaire consistant en une seconde décantation de la phase aqueuse.
- Procédé selon la revendication 1, caractérisé en ce que l'étape d'élimination de la majorité de l'eau restante dans le brai décanté de l'étape f) pour obtenir un brai modifié implique l'utilisation d'un processus d'élimination de l'eau dont la pression est inférieure ou égale à la pression atmosphérique et dont la température est inférieure à 105 °C.
- Procédé selon la revendication 13, dans lequel :a) la température est laissée à diminuer jusqu'à 80°C ou moins après l'élimination de l'eau ; etb) la température est à nouveau laissée à diminuer jusqu'à 60°C ou moins si le brai modifié doit être stocké avant le début de l'étape g).
- Procédé suivant la revendication 1, dans lequel la base métallique alcaline est choisie au sein du groupe constitué par l'hydroxyde de sodium, l'hydroxyde de potassium ou leurs combinaisons.
- Procédé selon la revendication 1, dans lequel l'acide minéral est choisi au sein du groupe constitué par l'acide sulfurique, l'acide chlorhydrique, l'acide phosphorique ou leurs combinaisons quelconques.
- Procédé suivant la revendication 1, dans lequel le solvant de l'étape i) comprend un mono-alcool de faible masse moléculaire choisi au sein du groupe constitué par le méthanol, l'éthanol et l'isopropanol.
- Procédé selon la revendication 17, dans lequel le solvant de l'étape i) comprend en outre des esters acétates de méthanol, d'éthanol ou d'isopropanol, des cétones ou des hydrocarbures en C1-C8, ou leurs mélanges.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/060,022 US20030144536A1 (en) | 2002-01-28 | 2002-01-28 | Process of extracting and purifying phytosterols and phytostanols from tall oil pitch |
| US60022 | 2002-01-28 | ||
| PCT/CA2003/000098 WO2003064442A2 (fr) | 2002-01-28 | 2003-01-27 | Procede d'extraction et de purification de phytosterols et de phytostanols extraits de poix de tall oil |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1470149A2 EP1470149A2 (fr) | 2004-10-27 |
| EP1470149B1 true EP1470149B1 (fr) | 2009-11-11 |
Family
ID=27609947
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP03701377A Revoked EP1470149B1 (fr) | 2002-01-28 | 2003-01-27 | Procede d'extraction et de purification de phytosterols et de phytostanols extraits de poix de tall oil |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US20030144536A1 (fr) |
| EP (1) | EP1470149B1 (fr) |
| JP (1) | JP2005516069A (fr) |
| CN (1) | CN1639184A (fr) |
| AT (1) | ATE448239T1 (fr) |
| AU (1) | AU2003203072B2 (fr) |
| BR (1) | BR0307270A (fr) |
| CA (1) | CA2474592A1 (fr) |
| DE (1) | DE60329977D1 (fr) |
| ES (1) | ES2335754T3 (fr) |
| NZ (1) | NZ534369A (fr) |
| WO (1) | WO2003064442A2 (fr) |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR0106522A (pt) * | 2001-12-17 | 2003-09-09 | Resitec Ind Quimica Ltda | Processo para separar produtos valiosos insaponificáveis obtidos de matérias primas diversas |
| CN100334103C (zh) * | 2005-09-20 | 2007-08-29 | 南京工业大学 | 一种从植物油沥青中提取植物甾醇乙酸酯的方法 |
| US8309156B2 (en) * | 2005-12-20 | 2012-11-13 | Pharmachem Laboratories, Inc. | Compositions comprising one or more phytosterols and/or phytostanols, or derivatives thereof, and high HLB emulsifiers |
| US8318962B2 (en) * | 2006-12-29 | 2012-11-27 | Cognis Ip Management Gmbh | Process for recovering sterols from a crude source containing sterol esters |
| FI20070137A0 (fi) * | 2007-02-15 | 2007-02-15 | Raisio Benecol Oy | Menetelmä rasvahappojen, hartsihappojen ja sterolien eristämiseksi mäntyöljypiestä |
| FI20080174A0 (fi) † | 2008-02-29 | 2008-02-29 | Raisio Nutrition Ltd | Menetelmä sterolien ja happojen erottamiseksi mäntyöljypiestä |
| FI124101B (en) | 2012-05-14 | 2014-03-14 | Hankkija Maatalous Oy | Modified tall oil fatty acid |
| US8709238B2 (en) | 2012-05-18 | 2014-04-29 | Uop Llc | Process for separating crude tall oil for processing into fuels |
| FI124918B (fi) | 2013-05-14 | 2015-03-31 | Hankkija Maatalous Oy | Mäntyöljyrasvahappo |
| FI125051B (fi) | 2013-10-24 | 2015-05-15 | Hankkija Oy | Mäntyöljyrasvahappo |
| HUE050907T2 (hu) | 2013-11-13 | 2021-01-28 | Hankkija Oy | Gyantasavakat tartalmazó takarmány-kiegészítõ |
| CN103897009B (zh) * | 2014-04-14 | 2015-08-12 | 西安海斯夫生物科技有限公司 | 一种木浆浮油的分离方法 |
| MY185625A (en) * | 2015-07-02 | 2021-05-26 | Malaysian Palm Oil Board Mpob | Method of extracting and concentrating phytosterol from palm oil by-product |
| US11912874B2 (en) | 2016-06-10 | 2024-02-27 | A.L.M. Holding Company | Crude sterol as an additive in asphalt binder |
| CA3033251A1 (fr) | 2016-08-09 | 2018-02-15 | A.L.M Holding Company | Melanges de sterols utilises comme additif dans un liant d'asphalte |
| CN106433474A (zh) * | 2016-11-21 | 2017-02-22 | 无锡市伟丰印刷机械厂 | 一种松脂用高效溶解提纯设备 |
| WO2018144731A1 (fr) | 2017-02-02 | 2018-08-09 | A.L.M Holding Company | Émulsions bitumineuses contenant un additif stérolique pour chaussée asphaltée |
| US11427697B2 (en) | 2017-07-26 | 2022-08-30 | A.L.M. Holding Company | Use of sterols from animal waste as an additive in asphalt binder |
| CN107417759B (zh) * | 2017-08-11 | 2020-04-17 | 内蒙古昶辉生物科技股份有限公司 | 从纸浆浮油沥青中提炼植物甾醇以及甾醇酯的制备方法 |
| JP2021500496A (ja) | 2017-10-20 | 2021-01-07 | エー.エル.エム.ホールディング カンパニー | ステロール含有アスファルト乳剤表面処理 |
| CN112513234A (zh) * | 2018-06-01 | 2021-03-16 | 拉伊西奥营养有限公司 | 用于分离甾醇和富含脂肪酸和树脂酸的馏分的方法 |
| CN108912199A (zh) * | 2018-06-21 | 2018-11-30 | 江苏科鼐生物制品有限公司 | 一种木甾醇的制备方法及系统 |
| CN108912198A (zh) * | 2018-06-21 | 2018-11-30 | 江苏科鼐生物制品有限公司 | 一种高收率木甾醇的制备方法及系统 |
| CN108912196A (zh) * | 2018-06-21 | 2018-11-30 | 江苏科鼐生物制品有限公司 | 一种植物甾醇汽提脱溶剂的方法及系统 |
| CN115281250B (zh) * | 2022-09-20 | 2024-02-09 | 青岛葆福德生物医药科技有限公司 | 一种降血脂食用油配方及其制备工艺 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2835682A (en) * | 1955-08-08 | 1958-05-20 | Swift & Co | Sterol recovery process |
| US3649580A (en) * | 1969-03-13 | 1972-03-14 | Arizona Chem | Tall oil pitch tackifiers in ethylene-propylene terpolymer rubber |
| US3556827A (en) * | 1969-12-29 | 1971-01-19 | Kenneth E Mcconnaughay | Aggregate binder and paving composition |
| CA2230373A1 (fr) * | 1998-02-20 | 1999-08-20 | Forbes Medi-Tech Inc. | Methode de preparation de phytosterols a partir de poix de tall-oil |
| US7371876B2 (en) * | 1999-04-27 | 2008-05-13 | Sterol Technologies Ltd. | Process for the purification of sterols from hydrocarbon extracts using evaporative fractionation |
-
2002
- 2002-01-28 US US10/060,022 patent/US20030144536A1/en not_active Abandoned
-
2003
- 2003-01-27 AU AU2003203072A patent/AU2003203072B2/en not_active Ceased
- 2003-01-27 ES ES03701377T patent/ES2335754T3/es not_active Expired - Lifetime
- 2003-01-27 NZ NZ534369A patent/NZ534369A/en not_active IP Right Cessation
- 2003-01-27 BR BR0307270-3A patent/BR0307270A/pt not_active Application Discontinuation
- 2003-01-27 JP JP2003564062A patent/JP2005516069A/ja active Pending
- 2003-01-27 EP EP03701377A patent/EP1470149B1/fr not_active Revoked
- 2003-01-27 CN CN03804912.0A patent/CN1639184A/zh active Pending
- 2003-01-27 WO PCT/CA2003/000098 patent/WO2003064442A2/fr active Application Filing
- 2003-01-27 CA CA002474592A patent/CA2474592A1/fr not_active Abandoned
- 2003-01-27 AT AT03701377T patent/ATE448239T1/de not_active IP Right Cessation
- 2003-01-27 DE DE60329977T patent/DE60329977D1/de not_active Expired - Lifetime
Also Published As
| Publication number | Publication date |
|---|---|
| DE60329977D1 (de) | 2009-12-24 |
| CA2474592A1 (fr) | 2003-08-07 |
| EP1470149A2 (fr) | 2004-10-27 |
| WO2003064442A2 (fr) | 2003-08-07 |
| JP2005516069A (ja) | 2005-06-02 |
| CN1639184A (zh) | 2005-07-13 |
| WO2003064442A3 (fr) | 2003-10-02 |
| US20030144536A1 (en) | 2003-07-31 |
| BR0307270A (pt) | 2004-12-14 |
| AU2003203072B2 (en) | 2009-04-23 |
| NZ534369A (en) | 2006-07-28 |
| ES2335754T3 (es) | 2010-04-05 |
| ATE448239T1 (de) | 2009-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1470149B1 (fr) | Procede d'extraction et de purification de phytosterols et de phytostanols extraits de poix de tall oil | |
| AU2003203072A1 (en) | Distalliative process of extracting and purifying phytosterols and phytostanols from tall oil pitch | |
| US8338564B2 (en) | Method for the preparation of phytosterols from tall oil pitch | |
| WO2008099051A2 (fr) | Procédé d'isolation d'acides gras, d'acides résiniques et de stérols à partir d'une résine naturelle d'huile de pin | |
| EP1594834B1 (fr) | Procede pour obtenir des esters d'alkyle d'acide gras, des acides resiniques et des sterols a partir de tall oil brut | |
| EP1056767B1 (fr) | Procede pour la preparation de phytosterols a partir de poix de tall-oil | |
| CN113348160A (zh) | 鱼油胆固醇 | |
| EP1173463B1 (fr) | Procede de purification de sterols d'extraits d'hydrocarbures avec fractionnement par evaporation | |
| US10351588B2 (en) | Production of sterols | |
| US20040024175A1 (en) | Method for the preparation of phytosterols from tall oil pitch | |
| US7371876B2 (en) | Process for the purification of sterols from hydrocarbon extracts using evaporative fractionation | |
| CN114391018B (zh) | 从妥尔油沥青中提取植物甾醇和甾烷醇 | |
| US6770767B1 (en) | Method for the extraction and isolation of neutral substances from a soap | |
| RU2128662C1 (ru) | Способ получения ситостерина из таллового пека | |
| SU1076439A1 (ru) | Способ переработки сульфатного мыла | |
| EP1190025B1 (fr) | Procede d'extraction et d'isolement | |
| WO2000012533A1 (fr) | Isolation et purification de sterols de la fraction des neutres de poix de tall oil par cristallisation directe, phase duelle | |
| WO2000009535A1 (fr) | Isolement et purification de sterols a partir d'une fraction de substances chimiquement neutres de poix de tall oil par cristallisation directe en une etape | |
| CN112513234A (zh) | 用于分离甾醇和富含脂肪酸和树脂酸的馏分的方法 | |
| EP1389622A2 (fr) | Procédé de purification de stérols d'extraits d'hydrocarbures avec fractionnement par évaporation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20040729 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: PHYTO-SOURCE, L.P. |
|
| 17Q | First examination report despatched |
Effective date: 20080623 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: COGNIS IP MANAGEMENT GMBH |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LI LU MC NL PT SE SI SK TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 60329977 Country of ref document: DE Date of ref document: 20091224 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2335754 Country of ref document: ES Kind code of ref document: T3 |
|
| LTIE | Lt: invalidation of european patent or patent extension |
Effective date: 20091111 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20100311 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20100211 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100131 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20091111 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| 26 | Opposition filed |
Opponent name: RAISIO NUTRITION LTD. Effective date: 20100811 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100131 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20100212 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100131 |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20100127 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20110128 Year of fee payment: 9 Ref country code: FI Payment date: 20110112 Year of fee payment: 9 Ref country code: DE Payment date: 20110119 Year of fee payment: 9 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20110126 Year of fee payment: 9 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R064 Ref document number: 60329977 Country of ref document: DE Ref country code: DE Ref legal event code: R103 Ref document number: 60329977 Country of ref document: DE |
|
| RDAF | Communication despatched that patent is revoked |
Free format text: ORIGINAL CODE: EPIDOSNREV1 |
|
| RDAG | Patent revoked |
Free format text: ORIGINAL CODE: 0009271 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: PATENT REVOKED |
|
| 27W | Patent revoked |
Effective date: 20111124 |
|
| GBPR | Gb: patent revoked under art. 102 of the ep convention designating the uk as contracting state |
Effective date: 20111124 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: IT Payment date: 20120125 Year of fee payment: 10 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R107 Ref document number: 60329977 Country of ref document: DE Effective date: 20120705 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20120131 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20100512 Ref country code: LU Free format text: LAPSE BECAUSE OF REVOCATION BY EPO Effective date: 20100127 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: ES Payment date: 20120223 Year of fee payment: 10 |