OBJECT OF THE INVENTION
-
The present invention deals with a replenishment process for metal electrodeposition baths,
i.e. with a step of metal plating processes in which the electrolyte is enriched in metal
cations to maintain substantially constant the metal cations concentration of the
electrodeposition baths. More particularly, the invention relates to the replenishment step in
plating processes from Copper-containing alkaline pyrophosphate or acid sulphate baths
and from Zinc-containing acid sulphate baths, using insoluble anodes.
-
In these processes the concentration of Cu2+ and Zn2+ is kept constant by recirculating the
working solution in an enrichment cell, or replenishment cell.
STATE OF THE ART
-
In almost all industrial processes involving metal electroplating, and particularly in Cu and
Zn electroplating, it is required that the concentration of the metal cations, namely Cu2+
and Zn2+, is kept constant.
-
This is presently achieved, for Cu plating baths, in the following ways:
- i. Chemical dissolution method. Cu metal slabs are plunged into the plating solution, which
performs a corrosive action on the metal, this method is far too slow for an efficient
industrial application.
- ii. Addition of CuO powder. CuO powder can be poured directly into the working
electrolyte and dissolves forming Cu2+ ed OH-. This method is very handy and has the
advantage of carrying out an alkalinising action (see Detailed description of the Invention
in the following). Nevertheless dissolution is not quantitative (some very fine powder
remains suspended in the plating bath and cannot be filtered out by standard continuous
filtration plants, this powder can codeposit and spoil the properties of the plated layer). In
addition, this reagent is generally too expensive for large-scale industrial applications.
-
-
A field were the need of plating processes with constant Cu2+ and Zn2+ is specially strong,
is that relevant to the production of steel-wire reinforced rubber items for applications such
as: tyres, pressure tubes, carrier- and drive-belts. Tyres are commonly reinforced with
brass-coated high-strength steel plaits. In order to achieve an optimal behaviour of the
reinforced system, it is necessary to guarantee a high adhesion between rubber and
steelcord. The mechanism which ensures such adhesion is generally the chemical bonding
obtained through the formation of sulphide bonds between a metal, which easily undergoes
sulfidation, and the rubber; this reaction occurs mainly during the vulcanisation process.
Alloys of interest for such applications are brass, Zn-Co and Zn-Mn. Such alloys also
enhance the corrosion resistance of the steelcord. Such coatings are generally applied to
steel wires whose diameter is larger than that of the final steelcord, such wires are therefore
drawn after electroplating, the coating thus performs also a lubricating action during the
drawing process.
-
The standard brass-coating process of steel wire typically consists in the following unit
operations.
- 1) Hot water rinse at 140÷180°F.
- 2) Sulphuric or chloridric acid pickling, in order to dissolve surface oxides.
- 3) Water rinse.
- 4) Electrodeposition of the Cu layer (typical thickness 0.5 µm) from an alkaline
pyrophosphate bath.
- 5) Electrodeposition of the Cu layer (typical thickness 0.5 µm) from an acid sulphate bath.
- 6) Water rinse.
- 7) Electrodeposition of the Zn layer (typical thickness 0.5 µm) from an acid sulphate bath.
- 8) Water rinse.
- 9) After the diffusion heat-treatment the brass-coated wire is pickled in a dilute phosphoric
acid solution at room temperature.
-
-
Step # 5 could be omitted by having a thickness of Cu layer in step # 4 of about 1 arm.
-
In the baths for the electrodeposition of Cu and Zn, the concentration of the Cu2+ e Zn2+
cations is generally kept constant by the oxidation of soluble Cu and Zn anodes,
respectively.
-
EP-A-0508212, in the name of The Goodyear Tyre and Rubber Co., discloses a process of
keeping the Cu concentration constant with a system comprising insoluble anodes and an
cation-exchange membrane-based replenishment device. According to this process, there is
provided a replenishment, or enrichment, cell wherein a Cu anode is dissolved and the
selective membrane avoids the Cu ions to migrate to the cathode, be reduced and redeposit
at the cathode. The selective membrane acts as a chemical and selective separation means
for Cu ions present in the enrichment bath.
-
The drawbacks of this process are that the membranes are expensive and lean to clog, and
that they require maintenance and a pretreatment before they can be used in the process.
SUMMARY OF THE INVENTION
-
The present invention deals with a peculiar procedure for the supply of metal cations, and
especially of Cu2+ and Zn2+, for keeping the concentration of these cations constant in
applications of alkaline pyrophosphate Cu and of acid sulphate Cu or Zn electroplating
processes carried out with insoluble anodes.
-
The use of insoluble anodes shows marked advantages in comparison with the traditional
soluble-anode process:
- i. dimensional stability of current density distribution, with enhanced reproducibility of the
product characteristics,
- ii. savings of labour costs related to the substitution if soluble anodes,
- iii. avoidance of reworking/recycling processes for partially dissolved anodes.
-
-
Cu2+ and Zn2+ supply to the working (i.e. deposition) cell is achieved by circulating a given
amount of the working electrolyte from the working cell to the enrichment cell where
dissolution of suitable anodes takes place: metal deposition at the cathode of the
enrichment cell is reduced or substantially avoided, in the absence of the above disclosed
selective membrane or of other cation chemical separation means located between anode
and cathode of said enrichment cell, by carrying out cathodic reduction reactions that
produce or consume a gas.
-
Said reduction reaction can be divided into two main groups.
i. Anodic Cu or Zn dissolution with simultaneous limiting current density
electrodeposition of Cu (cathodic efficiency less than 100%)
-
The electrolyte in the enrichment cell is in contact with a large-area Cu or Zn anode and a
small-area cathode (e.g., but with no limitation, a metal wire of suitable mechanical
properties). These catodes and anodes are known in the art. The circulation of an electric
current is imposed to the system. The anode tends to dissolve with an anodic efficiency
close to 100% (the only technically relevant anodic reaction being the oxidation of Cu to
Cu2+ and of Zn to Zn2+), at the cathode the reduction of Cu2+ or Zn2+ to metallic Cu or Zn
occurs under limiting-current conditions (the cathodic kinetics is controlled by mass-transport
to the cathode) together with a side reaction: the reduction of H+ to H2 (cathodic
current efficiency less that 100%). The difference between the cathodic and anodic current
efficiencies is the enrichment factor for the solution. The enrichment factor can be
optimized by acting on the cell geometry, the hydrodynamic conditions of the electrolyte
and the current density imposed to the electrodic system. The limiting-current
electrodeposition brings about the formation of an incoherent and powdery cathodic
deposit, suitable means of powder removal will therefore be used. Notwithstanding the
possibility of engineering optimisation, any feasible implementation of the process leads to
an effective enrichment in Cu2+ of the solution.
ii. Anodic Cu dissolution with an oxygen cathode
-
The electrolyte in the enrichment cell is brought into contact with a Cu or Zn anode and a
suitable cathode. Preferably the cathode is a porous electrode with a catalytic performance
for the reaction of oxygen gas reduction to hydroxide anion in alkaline aqueous
environment, such as an oxygen fuel-cell electrode; the alkaline environment is favourable
in that most commercially available porous gas electrodes can be adopted. At the cathode
triple contact is achieved among electrolyte, oxygen-rich gas (either pure oxygen or
compressed air of the suitable pressure to achieve the required oxygen activity) and solid
material which acts both as current-carrier and electrocatalyst for oxygen reduction.
Alternatively - and especially with acid electrolytes - the reaction can be carried out in a
packed bed cathode system made of catalytic spheroids (e.g. silver or graphite activated
with platinum nuclei) through which the oxygen-rich gas flows in contact with the ion
solution to be enriched.
-
Both the above-mentioned implementations (porous electrode and packed-sphere bed
electrode) should be meant as examples and should not, in any way, be meant as limiting
the applications of the combination of oxygen-reduction cathodic reaction and anodic
dissolution of Cu or Zn. An electric current circulation is imposed to the system. The anode
tends to dissolve with an anodic efficiency close to 100% (the only technologically relevant
anodic reaction being the oxidation of Cu to Cu2+), at the cathode the reduction of O2 to
OH- occurs, this reaction is thermodynamically favoured over the reduction of Cu2+ to
metallic Cu and of Zn2+ to Zn. The reduction of metal - which would be a loss term as far
as cation-enrichment of the working solution is concerned and could damage the gas
electrode - can occur only if O2 reduction takes place under limiting current density
conditions. The enrichment factor can be optimized by acting on the type of the cell
(porous-electrode cell or packed sphere bed electrode) on the cell geometry, on the
hydrodynamic conditions of the electrolyte and on the current density imposed to the cell.
Even though engineering optimisations are possible, any feasible implementation of the
process produces an efficient Cu2+ or Zn2+ enrichment of the solution.
DETAILED DESCRIPTION OF THE INVENTION
-
The invention is further disclosed by reference to the following non-limiting drawings:
- Fig. 1 is a diagramic view of a device of the invention including working cell and
enrichment cell;
- fig. 2 is a schematic view of the enrichment cell for a limiting current process;
- fig. 3 is a schematic view of an enrichment cell for the gas-cathode process;
- fig. 4 is a top view of the cell of fig. 2; and
- fig. 5 is a schematic sectional view of a gas cathode for the gas-cathode cell.
-
The hereinbelow disclosed process specifically refers to plating of steel wires, but this
should not be intended to limit the invention scope to that use only.
-
In the application of this invention (see Figure 1) steel wire 1 is coated with a layer of a few
µm (typically 0,5÷5 µm and preferably 1÷2 µm) in a deposition, or working, cell 2. Wire 1
is polarised cathodically by generator 3, generally operated in galvanostatic (current
control) conditions (a successful application of potentiostatic, i.e. voltage control,
conditions is possible too), via contact pulleys 4, known in the art. Steel wire 1 is
contacting an aqueous solution of Cu2+-pyrophosphate, or Cu2+ acid sulphate or Zn2+ acid
sulphate 5 while flowing through the cell. Solution 5 is also in contact with an insoluble
anode 6 and electrically connected to generator 3. Given the limited aggressivity of the
electrolyte, most of the commercial insoluble anodes can be efficiently used (e.g. titanium-based
and Pt, Pt/Ir coated, commonly commercially available, see above cited EP-A-0508212
for more details).
-
The working solution displays a cation concentration typically in the range 0.10÷0.25
ozs./gall. for Cu2+ in pyrophosphate baths, 9÷12 ozs/gall for Cu2+ in acid sulphate baths,
and 10÷13 ozs/gall for Zn2+ in acid sulphate baths. Pyrophosphate anion concentration is
typically in the range 0.80÷1.5 ozs./gall and sulfuric acid concentration in Cu and Zn cation
solutions is typically in the range 8.0÷10.0 ozs/gall and the quantity necessary to reach a pH
of about 3, respectively. The solution pH is generally controlled in the following intervals:
Cu2+ pyrophosphate baths 7.5÷9.5 and preferably close to 8.5, Zn2+- sulphate baths
2.75÷3.25 and preferably close to 3.0.
-
The temperature of working solution 5 is regulated in the range 100÷150°F and preferably
close to 120°F. Generator 3 is controlled in such a way that it gives cathodic current
densities in the interval 0.25÷1.5 A/sq.in.. The steel wire crosses the electrodeposition cell
at a velocity suitable in order to guarantee - for a given cathodic current density - an
optimal residence time of the wire in the cation solution according to the desired coating
thickness.
-
As the electrodeposition process proceeds, the Cu2+ or Zn2+ concentration of working
solution 5 diminishes (in an approximately linear way as a function of time) and it is
necessary to replenish the solution with metal cations in order to keep the optimal
concentration constant against time. This aim is achieved by recirculating (Q=Q1) the
working solution present in the cell 5, or, alternatively, blending (Q>Q1) a portion of it with
a suitably Cu2+ or Zn2+-enriched solution produced in the enrichment device 7. The
enrichment device is connected in series to the recirculation system (recirculation pumps 8,
recirculation tubing 9, working electrolyte storage tank 10, the latter can be equipped with
pH and Cu2+/Zn2+ concentration controls), which is generally available in electrodeposition
systems of this kind, allowing the treatment of a flow Q of electrolyte.
-
Usually, a portion Q1 (Q>Q1) is derived from the recirculation flow Q and is delivered to
the enrichment device 7, comprising an enrichment cell 11 and possibly a storage and
diluting tank for the treated electrolyte 12, a dilution system 13 supplying demineralised
water to tank 12 and the relevant pumping systems 14 and pipelines 15. The treated flow
Q1 is added to the working electrolyte storage tank 10, from which the flow Q is pumped to
the wire-coating cell.
-
As above mentioned, enrichment cell 11 can be implemented according to one of two
embodiments of the process: i. (see Figure 2) anodic Cu or Zn dissolution with
simultaneous limiting-current electrodeposition of Cu or Zn (cathodic efficiency less than
100%); ii. (see Figure 3) anodic dissolution of Cu or Zn with oxygen cathode.
-
In the limiting-current enrichment cell (Figure 2) the Cu2+ or Zn2+-enriched solution is
contacted with at least one Cu or Zn anode la whose surface area is much larger than the
surface of cathode 3a. It is generally advisable to use Cu or Zn spheroids (nuggets) or
scraps with typical dimension of a few inches which can be conveniently contained in one
or two suitable commercial anode basket (e.g. made of Ti, see above cited EP application
for details); if two anode baskets are present, the wire cathode is usually located between
them. The anodic polarisation is obtained by connecting the Cu or Zn anode to the positive
terminal of the generator 2a (of the same characteristics as the generator 3 of Figure 1). The
anodic metal is oxidised to the relevant cation; as far as the material balance is concerned,
it is a very good approximation to consider this as the only anodic reaction (100% anodic
current efficiency) taking place in the enrichment cell. The anodic Cu or Zn is dissolved
during the operation of the enrichment cell and it is therefore necessary to add Cu
spheroids or scarps - typically batchwise - to the anode basket(s).
-
The thus enriched anolyte is in contact with a metal cathods 3a (e.g. a steel wire) whose
surface area is much smaller than the anodic one. The cathodic polarisation is achieved by
connecting the cathodic wire to the negative terminal of generator 2a (of the same
characteristics as the generator 3 of Figure 1). The thermodynamically favoured cathodic
reaction in the system at hand is Cu2+ or Zn2+ reduction to Cu metal or Zn metal. The rate
of such reaction is limited by mass transport of the cations to the cathode.
-
A first approximation to the maximum reduction rate of Cu2+ to Cu and Zn2+ to Zn (a loss
term for the enrichment process) - expressed as the maximum cathodic current density
(A/sq.in.) for Cu discharge imax Cu and for Zn discharge imax Zn is given by the following
equations:
imax Cu=0.0154·[Cu2+]/δ
imax Zn=12.61·[Zn2+]/δ
where [Cu2+] and [Zn2+] are the cation concentrations expressed in ozs./gall, imax is
expressed in A/sq.in and δ is the concentration boundary layer thickness (typically of the
order of a few hundreds of µm), expressed in µm.
-
The imposed current density is larger than these values and the exceeding current density is
used up for the next competitive side reaction of hydrogen evolution from water. It is worth
noting that the loss term for electrodeposition of Cu or Zn can be limited to very low
values.
-
Several technical features can be adopted, alone or combined together, to enhance the
limiting current conditions.
-
A large area anode and a reduced area cathode can be used, as disclosed in figures 2 and 4.
-
Another feature is to minimize cation migration to the cathode by keeping the portion of
electrolyte solution around the cathode in still conditions; to obtain this result, a porous
container 16 is provided. Container 16 is made of porous ceramic, fabric or similar material
suitable to let the electrolyte flow through its pores and reach the cathode: this will avoid
the commotion due to pumping in and out the enrichment cell to reach the cathode area,
while enabling the process to proceed. A further advantage is obtained through the use of
container 16: the flow from the cell to within container 16 being reduced, a lesser amount
of CU cations reach the cathode, thus enhancing the limiting current conditions.
-
It should be noticed that container 16 is not a cation exchange membrane as disclosed in
prior art. In that case the membrane does not allow for Cu ions to flow through it: in
present case porous container acts as a resistance to the flow of Cu ions, that is reduced but
not impeded. The porous container can be dispensed with and a porous diaphragm, having
analogous characteristics and defining a cathode area with respect to the anode(s) area(s),
can be used. The overall effect of porous diaphragm or container is to reduce the effective
diffusion coefficiency.
-
A further way of enhancing limiting current conditions is by controlling the electrolyte
temperature; temperature should be lowered to the lowest acceptable value that does not
jeopardize the cell efficiency.
-
The cathodic hydrogen-evolution reaction, besides allowing cation enrichment of the
solution, brings about a beneficial alkalinising; as a matter of fact, the working electrolyte
tends to acidify in the wire-coating cell because of the anodic reaction at the insoluble
anodes bringing about the consumption of hydroxide anions supporting the anodic oxygen
gas evolution; in the case of analyses performed under this research, it was observed that
the alkalinisation in the enrichment cell can compensate for the electrolyte acidification in
the wire-coating cell perfectly; it is anyway advisable to provide the system with an
automatic pH-control device.
-
As far as the cathodic hydrogen gas evolution is concerned, also in the case of intensive
industrial-scale applications, the amount of discharged hydrogen is relatively limited and
cannot be judged a safety hazard if released to the atmosphere; nevertheless special care
must be devoted in the design of industrial cells to avoid any hold-up of cathodic gas or
build-up of hydrogen partial pressure in the plant. It is always advisable to avoid the use of
free flames in the neighbourhood of the cathodic gas outlet of limiting-current enrichment
cells.
-
As mentioned above, the limiting-current electrodeposition of Cu or Zn - necessary for the
operation of the cathode of the limiting-current enrichment cell - can easily lead to the
formation of dendritic or powdery deposits, it is therefore advisable to periodically clean
the cathode. Such cleaning operation can be easily performed manually by simply wiping
the cathode with a cloth. In the case of a cathodic wire, the cleaning operation can be very
easily performed without interrupting the cell operation, e.g. with a device of the kind
illustrated in Figure 4.
-
In this figure a top view of the limiting-current enrichment cell, shown in Figure 2, is given,
one can note the large-surface-area anode 1a, the cathodic wire 3a and generator 2a. The
cathodic wire forms a loop which is tensioned by two pulleys 19a and 19b and is connected
to the cell through two seals 17 and 18 consisting of a disposable rigid plastic insert on the
solution-side of the cell. The pulleys can be rotated periodically, e.g. by use of crank 20.
-
The movement of the wire through the plastic inserts can detach the loose deposit which
falls in a suitable container, e.g. above mentioned porous container 16, or can be removed
from the bottom of the cells as a mud.
-
In the enrichment cell with an oxygen cathode (Figure 3) the enriched Cu2+-pyrophosphate,
Cu2+-sulphate or Zn2+-sulphate solutions are in contact with at least one Cu or Zn anode 1b
with the same properties mentioned with reference to the limiting-current enrichment cell,
except the requirement of a high surface area. The anodic polarisation is obtained by
connecting the Cu or Zn anode to the positive terminal of generator 2b (of the same
characteristics as the generator 3 of Figure 1). Cu metal and Zn metal oxidise to Cu2+ and
Zn2+ , respectively; it is a sound technical approximation to judge this reaction as the only
relevant one at the anode of the enrichment cell.
-
The anolyte enriched in Cu2+ or Zn2+ is in contact with a bed of packed spheres or to some
other sort of gas electrode 3b fed with a gas containing O2 of suitable activity. The cathodic
polarisation is achieved by connecting the gas electrode to the negative terminal of
generator 2b (of the same characteristics as the generator 3 of Figure 1). The
thermodynamically favoured cathodic reaction in the system of interest is the reduction of
O2 to OH-. The rate of such reaction is limited by the mass transport of O2 to the
electroactive regions of the cathode.
-
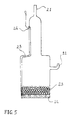
Figure 5 shows a gas-cathode suitable for the invention process.
-
The cathode is made of Pyrex glass and is equipped with gas inlet 21 and outlet 22. The
electrically active region of the electrode consists of a packed sphere bed of Ag beads 23 of
average diameter 200 µm, placed on a bored plate 24 of Ag, fixed to the Pyrex cylinder.
-
Electrical contact is provided by a Pt wire 25 brazed to the glass in position 26. Pure
oxygen is supplied with a low overpressure with respect to the atmosphere, in oredr to keep
the solution level at a level which can guarantee the three-phase contact electrolyte - gas -
Ag spheres. If air is used, it must be compressed to obtain the required oxygen activity.
-
Similarly to the limiting-current embodiment of the invention process, also the cathodic
reaction of oxygen reduction of hydroxide anions, besides allowing Cu2+ or Zn2+
enrichment of the solution, brings about a beneficial alkalinising; as a matter of fact, the
working electrolyte tends to acidify in the wire-coating cell because of the anodic reaction
at the insoluble anodes bringing about the consumption of hydroxide anions supporting the
anodic oxygen gas evolution; in the case of analyses performed under this research, it was
observed that the alkalinisation in the enrichment cell can compensate for the electrolyte
acidification in the wire-coating cell perfectly; it is anyway advisable to provide the system
with an automatic pH-control device.
-
It ought to be noted that - in the present detailed description of the invention - typical
implementations of the inventions were presented, such implementations, even though
considerably flexible, must be meant as illustrative, any possibility of varying or optimising
the design of the plant, by simplifying or complicating it, do not alter the scope and the
nature of the invention. The present invention will be described from an application-oriented
point of view in the following examples. These examples have a merely
illustrative purpose and must not, in ant way, be judges as limiting the extension or the
applicability of the invention or of the patented principles in devices showing a different
composition or layout.
EXAMPLE 1 - EXPERIMENT SHOWING THE PROCESS OF COPPER ANODIC
DISSOLUTION WITH SIMULTANEOUS LIMITING-CURRENT COPPER ELECTRODEPOSITION
-
In this experiment the enrichment of a Cu
2+-pyrophosphate solution is described, with the
limiting-current process, object of the present invention. The cell consists in a PVC tank of
dimensions 8 × 8 × 16 cu.in., equipped with a temperature-control system with plunged
resistance and thermocouple and a recirculation system (magnetic PTFE pump) providing a
maximum recirculation flow of Q=7 gall./h and a limited agitation of the electolyte (as far
as the hydrodynamics of the cell are concerned, a stagnant electrolyte is optimal close to
the cathode); the solution is removed from the upper part of the cell (1.5 in. below the free
surface of the solution) and fed to the enrichment cell in the lower part (3 in. from the
bottom of the cell). The anode consists in two Ti anode baskets of dimensions 1 × 6 × 12
cu.in. hanging from opposite walls of the cell. The anode baskets contains Cu spheroids of
diameter 0.5 in.. The cathode is a fixed steel wire of of diameter 0.02 in., placed 8 in.
above the bottom of the cell and centrally with respect to the two anodes, about 1.5 in. from
each one of them. The cell is powered with a laboratory generator, the experiments were
carried out under current control. The analysis of Cu concentration was performed by
atomic absorption on discontinuously sampled portions of the electrolyte. The pH was
monitored continuously with a glass-electrode pH-meter. The experimental results are
reported in Table 1 for tests carried out with a starting Cu
2+ concentration of 0.1155
ozs./gall., starting pyrophosphate 0.99 ozs./gall., starting pH 8.0, temperature 122°F.
| Variations of Cu2+ concentrations and pH for a demonstration run of a limiting-current pilot-plant enrichment cell (†pH corrected with discontinuous additions of H2SO4 in order to avoid basic-salt precipitations) |
| time (hours) | I=1 A
Cu2+ ozs/gall | I=1 A
pH | I=5 A
Cu2+ ozs/gall | I=5 A
pH | I=7,5 A
Cu2+ ozs/gall | I=7,5 A
pH |
| 1 | 0.1157 | 8,02 | 0.1154 | 8,08 | 0.1307 | 8,07 |
| 2 | 0.1212 | 8,03 | 0.1289 | 8,15 | 0.1352 | 8,32 |
| 5 | 0.1185 | 8,08 | 0.1387 | 8,35 | 0.1434 | 8,80 |
| 10 | 0.1307 | 8,16 | 0.1434 | 8,78 | 0.1592 | 9,51 |
| 24 | 0.1359 | 8,30 | 0.1563 | 9,48 | 0.2141 | ∼10,00† |
| 48 | 0.1457 | 8,78 | 0.2045 | ∼10,00† | 0.2871 | ∼10,00† |
EXAMPLE 2 - EXPERIMENT SHOWING THE PROCESS OF COPPER ANODIC
DISSOLUTION WITH OXYGEN CATHODE
-
In this experiment the enrichment of a Cu
2+-pyrophosphate solution is described, with the
oxygen-cathode process, object of the present invention. The cell was the same as
described in Example 1. The anode was only one of the anode baskets used in Example 1.
The gas cathode used in this Example is shown in Figure 6. This cathode was placed
vertically in a central position in the enrichment cell. The cell is electrically connected as
in Example 1, the experiment was carried out under current control. The analysis of Cu
concentration and of pH were performed as in Example 1. The experimental results are
reported in Table 2 for tests carried out with a starting Cu
2+ concentration of 0.1155
ozs./gall., starting pyrophosphate 0.99 ozs./gall., starting pH 8.0, temperature 122°F.
| Variations of Cu2+ concentrations and pH for a demonstration run of a oxygen-cathode pilot-plant enrichment cell |
| time (hours) | I=1 A
Cu2+ ozs/gall | I=1 A
pH | I=1 A
Cu2+ ozs/gall | I=1 A
pH |
| 1 | 0.1137 | 8,02 | 0.1179 | 8,06 |
| 2 | 0.1163 | 8,02 | 0.1197 | 8,15 |
| 5 | 0.1179 | 8,09 | 0.1216 | 8,22 |
| 10 | 0.1175 | 8,14 | 0.1274 | 8,39 |
| 24 | 0.1252 | 8,35 | 0.1371 | 8,88 |
| 48 | 0.1382 | 8,92 | 0.1598 | 9,53 |
EXAMPLE 3 - EXPERIMENT SHOWING THE PROCESS OF COPPER ANODIC
DISSOLUTION IN A Cu2+-SULPHATE SOLUTION WITH SIMULTANEOUS
LIMITING-CURRENT COPPER ELECTRODEPOSITION
-
In this experiment the enrichment of a Cu2+-sulphate solution is described, with the
limiting-current process, object of the present invention.
-
The same cell described in Example 1 is adopted. The experimental results are reported in
Table 3 for tests carried out with a starting Cu
2+ concentration of 10.25ozs./gall., H
2SO
4
concentration of 9 ozs./gall., starting conductivity of 0.1912 S, temperature 120°F.
| Variations of Cu2+ concentrations and conductivity for a demonstration run of a limiting-current pilot-plant enrichment cell with an acidic copper sulphate electrolyte |
| time (hours) | I=1 A
Cu2+ ozs/gall | I=1 A
conduct. S | I=2 A
Cu2+ ozs/gall | I=2 A
conduct. S | I=5 A
Cu2+ ozs/gall | I=5 A
conduct. S |
| 1 | 10.42 | 0.1887 | 10.61 | 0.1876 | 11.06 | 0.1815 |
| 2 | 10.59 | 0.1872 | 10.88 | 0.1854 | 11.85 | 0.1784 |
| 5 | 11.07 | 0.1861 | 11.66 | 0.1804 | precipitation | - |
| 10 | 11.34 | 0.1757 | precipitation | - | - | - |
| 24 | precipitation | - | - | - | - | - |
EXAMPLE 4 - EXPERIMENT SHOWING THE PROCESS OF COPPER ANODIC
DISSOLUTION IN A Cu2+-SULPHATE SOLUTION WITH OXYGEN CATHODE
-
In this experiment the enrichment of a Cu
2+-sulphate solution is described, with the
oxygen-cathode process, object of the present invention. The experimental system was the
same as described in Example 2. The experimental results are reported in Table 4 for tests
carried out with a starting Cu
2+ concentration of 10.25 ozs./gall., H
2SO
4 concentration of 9
ozs./gall., starting conductivity of 0.1912 S, temperature 120 °F,
| Variations of Cu2+ concentrations and conductivity for a demonstration run of a oxygen-cathode pilot-plant enrichment cell with an acidic copper sulphate electrolyte |
| time (hours) | I=1 A
Cu2+ ozs/gall | I=1 A
conduct. S | I=2 A
Cu2+ ozs/gall | I=2 A
conduct. S |
| 1 | 10.40 | 0.1899 | 10.72 | 0.1885 |
| 2 | 10.47 | 0.1886 | 11.03 | 0.1865 |
| 5 | 10.95 | 0.1851 | 11.70 | 0.1807 |
| 10 | 12.35 | 0.1787 | precipitation | - |
EXAMPLE 5 - EXPERIMENT SHOWING THE PROCESS OF ZINC DISSOLUTION IN
A Zn2+-SULPHATE SOLUTION WITH SIMULTANEOUS LIMITING-CURRENT ZINC
ELECTRODEPOSITION
-
In this experiment the enrichment of a Zn2+-sulphate solution is described, with the
limiting-current process, object of the present invention.
-
The same cell described in Example 1 is adopted. The experimental results are reported in
Table 5 for tests carried out with a starting Zn
2+ concentration of 11.85 ozs./gall., starting
pH 3, room temperature.
| Variations of Zn2+ concentrations and pH for a demonstration run of a limiting-current pilot-plant enrichment cell with an acidic zinc sulphate electrolyte |
| time (hours) | I=1 A
Zn2+ ozs/gall | I=1 A
pH | I=2 A
Zn2+ ozs/gall | I=2 A
pH | I=5 A
Zn2+ ozs/gall | I=5 A
pH |
| 1 | 12.04 | 3.02 | 12.25 | 3.04 | 12.87 | 3.10 |
| 2 | 12.24 | 3.05 | 12.65 | 3.08 | 14.04 | 3.27 |
| 5 | 12.84 | 3.15 | 13.93 | 3.31 | 16.91 | 3.69 |
| 10 | 13.87 | 3.32 | 15.97 | 3.52 | 22.67 | 4.43 |
| 24 | 16.62 | 3.85 | 21.70 | 4.23 | precipitation | - |
| 48 | 21.41 | 4.30 | 29.46 | 5.40 | - | - |
EXAMPLE 6 - EXPERIMENT SHOWING THE PROCESS OF ZINC ANODIC
DISSOLUTION IN A Zn2+-SULPHATE SOLUTION WITH OXYGEN CATHODE
-
In this experiment the enrichment of a Zn
2+-sulphate solution is described, with the oxygen-cathode
process, object of the present invention. The experimental system was the same as
described in Example 2. The experimental results are reported in Table 6 for tests carried
out with a starting Zn
2+ concentration of 11.85 ozs./gall., starting
pH 3, room temperature.
| Variations of Zn2+ concentrations and pH for a demonstration run of a oxygen-cathode pilot-plant enrichment cell with an acidic zinc sulphate electrolyte |
| time (hours) | I=1 A
Zn2+ ozs/gall | I=1 A
pH | I=2 A
Zn2+ ozs/gall | I=2 A
pH |
| 1 | 11.98 | 3.01 | 12.16 | 3.02 |
| 2 | 12.09 | 3.06 | 12.71 | 3.07 |
| 5 | 12.96 | 3.12 | 13.92 | 3.29 |
| 10 | 13.99 | 3.39 | 15.82 | 3.63 |
| 24 | 16.19 | 4.12 | 21.49 | 4.15 |
| 48 | 22.35 | 4.42 | 29.18 | 5.10 |