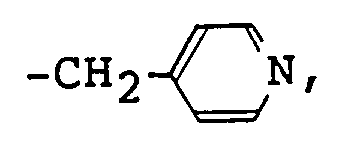EP0555864A1 - Composition for use in oral cavity - Google Patents
Composition for use in oral cavity Download PDFInfo
- Publication number
- EP0555864A1 EP0555864A1 EP19930102227 EP93102227A EP0555864A1 EP 0555864 A1 EP0555864 A1 EP 0555864A1 EP 19930102227 EP19930102227 EP 19930102227 EP 93102227 A EP93102227 A EP 93102227A EP 0555864 A1 EP0555864 A1 EP 0555864A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- group
- oral cavity
- ion
- composition
- carbon atoms
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/43—Guanidines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/41—Amines
- A61K8/416—Quaternary ammonium compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4906—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom
- A61K8/4926—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with one nitrogen as the only hetero atom having six membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/55—Phosphorus compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/63—Steroids; Derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/64—Proteins; Peptides; Derivatives or degradation products thereof
- A61K8/66—Enzymes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
Definitions
- the present invention relates to a composition for use in the oral cavity, which can disinfect bacteria in the oral cavity, has a reduced bitter or astringent taste, and is useful for the prevention or treatment of oral diseases such as dental caries, periodontosis, and the like.
- Dental caries and periodontosis which are typical diseases in the oral cavity are a type of bacterial infection.
- Dental caries is considered to be caused by Streptococcus mutans , gingivitis by Actinomyces viscosus, and periodontitis by Prophyromonas gingivilis, Capnocytophaga ochracea, and the like.
- compositions for oral cavity comprising an antibacterial agent for these bacteria had been developed for the prevention of dental caries and periodontosis, which are commercially available.
- examples of such compositions include dentifrices, mouthwashes, and the like, in which a cationic antibacterial compound (e.g., chlorhexidine hydrochloride, cetylpyridinium chloride), a nonionic antibacterial compound (e.g., triclosan), or the like, is incorporated.
- UK Patent No. 1,431,932 discloses a composition for use in the oral cavity for suppressing formation of dental calculus and dental stone, comprising an antibacterial compound which consists of 2 mols of a quaternary ammonium compound and 1 mol of a monoalkyl phosphate.
- This composition for use in the oral cavity exhibits no prolonged antibacterial effect and does not provide an improved taste.
- Japanese Patent Laid-open (kokai) No. 47542/1978 USP No.
- Japanese Patent Laid-open (kokai) No. 218605/1990 describes a composition comprising a quaternary ammonium compound and a salt of phosphoric ester and effective as an antibacterial and antiseptic agent for use in the skin or the hair.
- the specification does not suggest at all the possibility of applying the proposed composition to the oral cavity.
- An object of the present invention is to provide a composition for use in the oral cavity which exhibits an immediate and long-continued antibacterial activity to bacteria in the oral cavity, provides a good taste, and does not reduce its antibacterial activity even when a surfactant as a detergent is added.
- compositions comprising a specific antibacterial compound and a vehicle for use in the oral cavity provides little bitter or astringent taste, has an excellent antibacterial activity, and exhibits an immediate and long-lasting continued antibacterial effect, even if a surfactant is added. This finding has led to the completion of the present invention.
- composition for use in the oral cavity comprising:
- composition for use in the oral cavity comprising,
- Figure 1 is a chart showing the results of the disinfection test of the composition in Example 2.
- Figure 2 is a graph showing the rate of adsorption of antibacterial compounds in the compositions according to Example 3.
- antibacterial compounds of said component (A) used in the composition for use in the oral cavity of the present invention are the compounds represented by the following formulas (2) to (4), wherein at least one of R1, R2, or R3 is a linear or branched alkyl or alkenyl group with 8 to 30 carbon atoms, and others are individually a group selected from methyl group, ethyl group, benzyl group, and a group represented by one of the following formulas, -(CH2CH2O) n -H (wherein n is an integer of 1 to 15), or R4 represents a group -CH3, -CH2CH3, or -CH2CH2OH; and the counter anion X ⁇ represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to
- those having a linear or branched alkyl group with 8 to 14 carbon atoms e.g., 2-ethylhexyl group, decyl group, dodecyl group, or tetradecyl group or benzyl group, for at least one of R1 and R2, and methyl group and/or group for others are preferable.
- R5 is a linear or branched alkyl group with 10 to 20 carbon atoms
- X and X ⁇ may be the same or different and each individually represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or a monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms.
- Examples of linear or branched alkyl groups with 10 to 20 carbon atoms represented by R5 in general formula (3) include decyl group, dodecyl group, tetradecyl group, hexadecyl group, octadecyl group, eicosyl group, and the like.
- monoalkyl phosphate ion, monoalkenyl phosphate ion, monoalkyl phosphonate ion, or monoalkenyl phosphonate ion which are the counter anions represented by X ⁇ or X in formulas (1) to (4), are anions of the following formula (5) or (6), wherein R6 is a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms and R7 is a hydrogen atom, an alkali metal, an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue; and wherein R8 is a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms and R9 is a hydrogen atom, an alkali metal, an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue.
- an alkyl group with 10 to 20 carbon atoms is preferable, 12 to 16 carbon atoms more preferable, and 14 carbon atoms most preferable.
- Specific examples of such alkyl groups are lauryl, myristyl, and cetyl groups, and the like.
- R7 and R9 are normally hydrogen atoms.
- Some portion of counter anions may have an alkali metal (e.g., potassium, sodium), an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue for R7 or R9.
- the amount of the component (A) in the composition for use in the oral cavity of the present invention is 0.001 to 20% by weight (hereinafter % refers to % by weight), and preferably 0.01 to 1%. An amount outside the above range is not desirable, since if it is less than 0.001%, the antibacterial strength is weakened; if greater than 20%, the stability of the formulation is poor.
- Commonly used vehicles for the oral cavity such as those used in toothpaste, toothpowder, liquid dentifrice, mouthwash, gargle, mouth rinse, gum massage cream, troches, chewing gum, candies, or the like, can be used as vehicles for component (B) in the composition for use in the oral cavity of the present invention.
- Such vehicles for the oral cavity which can be used in the composition of the present invention include abrasives, thickening agents, moisturizers, surfactants, perfumes, sweeteners, preservatives, colorants, water, water-soluble fluorine-containing compounds, silicone, and other effective ingredients.
- abrasives examples include hydrogen calcium phosphate, calcium carbonate, calcium pyrophosphate, insoluble sodium metaphosphate, potassium metaphosphate, silicic acid anhydride, silicic acid hydrate, aluminum silicate, zirconium silicate, bentonite, zeolite, aluminum oxide, aluminum hydroxide, resins and mixtures of these.
- silicic acid anhydride, calcium carbonate, zeolite, silicic acid hydrate, and aluminum oxide are especially preferable.
- thickening agents examples include sodium carboxymethyl cellulose, methyl cellulose, hydroxyethyl cellulose, arginates, carrageenan, gum arabic, polyvinyl alcohol, gum tragacanth, starch, sodium polyacrylate, and the like.
- moisturizers examples include polyethylene glycol, propylene glycol, sorbitol, glycerine, maltitol, xylitol, and the like. Of these, glycerine, sorbitol, and the like are preferable.
- the surfactant is used as a foaming agent or a stabilizing agent for an oil-containing material.
- Various types of surfactants other than phosphates may be used.
- preferable surfactants include sodium alkyl sulfate, sodium alkylbenzenesulfonate, sodium N-acylsarcosinate, N-acylglutamates, polyoxyethylene hydrogenated castor oil, polyoxyethylene-polyoxypropylene block copolymers (pluronic type), fatty acid esters of sucrose, alkylglycosides, fatty acid esters of sorbitan, fatty acid esters of polyoxyethylenesorbitan, alkyldimethylamine oxide, carbobetaine, hydroxycarbobetaine, phosphobetaine, hydroxyphosphobetaine, sulfobetaine, hydroxysulfobetaine, and the like.
- water-soluble nonionic and amphoteric surfactants such as polyoxyethylene hydrogenated castor oil, polyoxyethylene-polyoxypropylene block copolymers, fatty acid esters of sucrose, alkylglycosides, fatty acid esters of sorbitan, and fatty acid esters of polyoxyethylenesorbitan, are particular preferable.
- perfumes examples include natural perfumes such as spearmint oil, peppermint oil, wintergreen oil, sassafras oil, clove oil, sage oil, eucalyptus oil, mayonara oil, cinnamon oil, thyme oil, lemon oil, orange oil, and the like; as well as synthetic perfumes such as 1-menthol, anethole, carvone, eugenol, thymol, methyl salicylate, and the like.
- natural perfumes such as spearmint oil, peppermint oil, wintergreen oil, sassafras oil, clove oil, sage oil, eucalyptus oil, mayonara oil, cinnamon oil, thyme oil, lemon oil, orange oil, and the like
- synthetic perfumes such as 1-menthol, anethole, carvone, eugenol, thymol, methyl salicylate, and the like.
- sweeteners examples include saccharin, saccharin sodium, stevioside, neohesperidyldihydrocarcone, beruraruchine, p-methoxycinnamic aldehyde, glycyrrhizinates, aspartame (methyl aspartylphenylalanine), and the like.
- preservatives include benzoic acid, sodium benzoate, parahydroxybenzoic esters, and the like.
- Water-soluble fluorine-containing compounds which can be used include sodium fluoride, sodium monofluorophosphate, and the like.
- Other effective ingredients include chlorophyll compounds, sodium chloride, vitamin C, vitamin E, nicotinic acid esters, allantoinchlorohydroxy aluminum, azulene, lysozyme chloride, hinokitiol, ⁇ -glycyrrhetinic acid, dipotassium glycyrrhizinate, protease, materials extracted from herbal medicines, and the like.
- any vehicles among the above-mentioned oral vehicles can be incorporated with the exception of the abrasive and the thickening agent.
- the ratio of alcohol and water is preferably 1:1 to 200:1, and more preferably 5:1 to 100:1.
- the amount of alcohol in the composition is preferably 1 to 20%.
- the composition is in the form of a paste, such as toothpaste or liquid dentifrice
- all of the above-mentioned vehicles can be incorporated.
- the abrasive is preferably 20 to 75% in the case of a toothpaste, and 10 to 30% in the case of a liquid dentifrice.
- a solid composition such as a toothpowder
- the solid ingredients among the above-mentioned vehicles for use in the oral cavity can be blended. Basically, it is preferable to incorporate 60 to 99% of the abrasive.
- the total content of the perfume and the sweetener in the oral vehicle be 0.01 to 5%.
- the pH of the composition of the present invention is in the range of 5 to 9.5, preferably, in the range of 6 to 8.
- the component (C) used in the present invention is a surfactant represented by the above general formula (7), wherein R10 is a linear or branched alkyl or alkenyl group with 8 to 30 carbon atoms, preferably an alkyl group with 10 to 20 carbon atoms, more preferably 12 to 16 carbon atoms, and most preferably 14 carbon atoms. Specific examples include decyl, lauryl, myristyl, and cetyl groups. Z1 and Z2 are preferably a partial neutralized compound having a neutralization degree of 0.8 to 2.0, especially 1.0 to 1.8.
- component (C) Given as specific examples of component (C) are 1 potassium monodecylphosphate, 1.2 sodium monolaurylphosphate, 1.3 potassium monomyristylphosphate, 1.5 triethanolamine monomyristylphosphate, 1 arginine monomyristylphosphate, 1.3 arginine monomyristylphosphate, 1.2 sodium monocetylphosphate, and the like.
- component (C) when added to a composition containing components (A) and (B), exhibit an action of further extending the period of time for which the composition maintains the antibacterial activity.
- amount of component (C) to be incorporated into the composition for the oral cavity of the present invention varies depending on the desired antibacterial activity and the types of cation (A m+ )
- the composition for use in the oral cavity can be prepared either by formulating components (A) and (B) or by formulating component (A'), instead of component (A), and the molar ratio of component (A') to component (C) satisfies (A')/(C) ⁇ 1/m.
- component (A') and (C) are formulated, it is considered that these components (A') and (C) react in the composition to form a salt.
- antibacterial compounds (8) of component (A') are components represented by the following general formulas (9), (10), and (11). wherein counter anions Y ⁇ and Y - 1 individually represent a halogen ion, Y2 is a halogen atom or a gluconic acid residue, and R1, R2, R3, R4, and R5 are the same as defined above.
- an excess amount of component (C) extends the period of time for which the composition maintains the antibacterial activity.
- the compounds represented by the general formula (1) used in the present invention can be prepared according to known methods.
- said compound (2) can easily be prepared from a commercially available quaternary ammonium salt, which is a compound represented by said general formula (9) with a halogen atom as the counter ion, by converting it into the OH-type quaternary ammonium salt, and neutralizing it by said compound having an anion residue.
- Said compound (3) can be prepared by a method comprising submitting a commercially available compound (10) to alkaline conditions, desalting it by electrodialysis, and neutralizing the product with said compound having an anion residue.
- An alkali metal salt, an amine salt, an organic amine salt, or a basic amino acid salt may be included during the neutralization to the extent that the neutralization is not hindered.
- Preferable formulas of the composition for use in the oral cavity of the present invention include liquid formulas such as mouthwash, mouth rinse, and gargle; paste formulas such as toothpaste, liquid dentifrice, and gum massage cream; and solid formulas such as toothpowder, and troche.
- a solid composition such as a toothpowder can normally be formulated by mechanically blending the above-mentioned solid ingredients.
- a composition in the form of a paste can normally be formulated by mechanically blending the various ingredients with deaeration.
- the composition is, in principle, preferably applied about 1-3 times per day, about five days a week.
- a mouth rinse is the most desirable. Disinfection can be effectively performed using about 10 ml of the mouth rinse of the present invention after brushing, holding in the mouth for about 30 seconds, then ejecting.
- composition for use in the oral cavity of the present invention exhibits a remarkablely rapid disinfection effect and continued antibacterial effect action against oral cavity bacteria while providing a good taste. Its disinfection effect is not reduced in the presence of surfactants.
- Antibacterial compounds in Table 1 were evaluated in terms of their taste.
- Evaluation was performed by a panel of 5 adult men and 5 adult women. 20 ml of a 1% ethanol solution containing 0.02% or 0.1% of one of the compounds in Table 1 was held in the mouth for 30 seconds to evaluate the taste while gargling and the aftertaste as one of three grades, O (not bad), D (rather bad), or X (bad). Each compound was graded as one of O, D, or X, to which the average of all panelists is closest. The results are shown in Table 1, which manifestly demonstrates a remarkable improvement in the taste of the composition of the present invention.
- the mixture was stirred for 1 minute at 37°C, suitably diluted with physiological saline, and cultured for 5 days at 37°C in BHI agar medium (goat blood hemin and menadione containing, a product of Difco Co.) to measure the number of living bacteria.
- BHI agar medium goat blood hemin and menadione containing, a product of Difco Co.
- Benzalkonium chloride (Sanizole, trade mark, a product of Kao Corp.), chlorhexidine gluconate, and cetylpyridinium chloride were selected as antibacterial compounds.
- An appropriate amount of arginine laurylphosphate (1.3 neutralization) was added to prepare aqueous solutions of antibacterial compound with a concentration of 0.01%.
- 1 g of hydroxyapatite powder (a product of Wako Pure Chemical Co.), which is a major component of the tooth was added to 20 ml of each solution. After stirring for 30 seconds, the amount the antibacterial compounds in the supernatant was quantitatively measured to calculate their adsorption to the hydroxyapatite.
- Each sample solution was evaluated in terms of the taste during mouthwash and the aftertaste by adult persons by holding 10 ml of the solution in the mouth for 30 seconds while gargling.
- the rate of bacterial metabolism suppression was determined by collecting saliva after 30 and 60 minutes and measuring its lactic acid producing capability, which is taken as the standard for the antibacterial activity.
- lactic acid producing capability 0.4 ml of saliva and 0.1 ml of 10% sucrose were reacted for 5 minutes at 37°C, and the reaction product was filtered and submitted to ion chromatography.
- the rate of suppression of lactic acid production was calculated by deducting the amount of lactic acid produced while unreacted as a blank to determine the lactic acid production.
- the results are shown in Table 4, which indicates that the comparative compounds had unfavorable tastes and hardly maintained their antibacterial effects, while the compounds of the present invention not only provided good taste, but also was proven to promote their prolonged antibacterial effects by the addition of a specific amount of arginine myristylphosphate.
- the compounds having the molar ratio of (antibacterial cation)/(counter anion) smaller than the reciprocal of the cation valency such as myristylphospholylated benzarkonium, myristylphospholylated cetylpyridinium, and dimyristylphospholylated chlorhexidine, exhibited both good tastes and excellent effects of the continued antibacterial action.
- Tooth paste compositions were prepared by blending and deaerating the components shown in Table 5.
- Concentrated mouthwash compositions 7 and 8 were prepared by blending the components shown in Table 6. The composition can be used after diluting to a volume of about 30 fold.
- Troche compositions with formulations shown in Table 7 were prepared by blending the components shown in Table 7 and molding the blend into a disk shape by a tabletting machine.
- Mouthwash compositions 11 and 12 were prepared by adding other components shown in Table 1 and blending the mixture.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Cosmetics (AREA)
Abstract
wherein Am+ is a nitrogen-containing antibacterial cation and the counter anion X⁻ represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or a monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms, and m is a valence of the cation A, and (B) a vehicle for use in the oral cavity. It exhibits a remarkablely rapid and continued disinfection effect action against oral cavity bacteria, while providing a good taste. Its disinfection effect is not reduced in the presence of surfactants. In addition, it does not color the tooth.
Description
- The present invention relates to a composition for use in the oral cavity, which can disinfect bacteria in the oral cavity, has a reduced bitter or astringent taste, and is useful for the prevention or treatment of oral diseases such as dental caries, periodontosis, and the like.
- Dental caries and periodontosis (e.g., gingivitis, periodontitis), which are typical diseases in the oral cavity are a type of bacterial infection. Dental caries is considered to be caused by Streptococcus mutans, gingivitis by Actinomyces viscosus, and periodontitis by Prophyromonas gingivilis, Capnocytophaga ochracea, and the like.
- Accordingly, compositions for oral cavity comprising an antibacterial agent for these bacteria had been developed for the prevention of dental caries and periodontosis, which are commercially available. Examples of such compositions include dentifrices, mouthwashes, and the like, in which a cationic antibacterial compound (e.g., chlorhexidine hydrochloride, cetylpyridinium chloride), a nonionic antibacterial compound (e.g., triclosan), or the like, is incorporated.
- However, many of the conventional cationic antibacterial agents give a bitter and astringent taste when incorporated in an amount effective for the disinfection. For example, Yamazaki, et al reports that a gargle comprising 0.04% by weight of benzalkonium chloride is not suitable for common use, since it provides an extremely bitter and astringent taste, even though its effect is remarkable ["Oral cavity disinfection effect of benzalkonium chloride", Journal of Dental Critics, 554, 227 (1988)]. Furthermore, these cationic antibacterial compounds decrease their disinfection effects in the presence of an anionic surfactant in a mouthwash composition. Therefore, their use in combination with a nonionic surfactant was proposed (e.g., Japanese Patent Laid-open (kokai) Nos. 101417/1984, 101418/1984, 282317/1990, 109315/1991). The proposed compositions, however, not only failed to improve the taste, but also provided insufficient disinfection effects due to the poor immediate antibacterial effect exhibited by conventional nonionic antibacterial compounds. Thus, the compositions had a drawback in that they could not exhibit sufficient antibacterial actions during use.
- UK Patent No. 1,431,932 discloses a composition for use in the oral cavity for suppressing formation of dental calculus and dental stone, comprising an antibacterial compound which consists of 2 mols of a quaternary ammonium compound and 1 mol of a monoalkyl phosphate. This composition for use in the oral cavity, however, exhibits no prolonged antibacterial effect and does not provide an improved taste. On the other hand, as compositions for use in the oral cavity to which a phosphate type surfactant is added, Japanese Patent Laid-open (kokai) No. 47542/1978 (USP No. 4,152,421) discloses a dentifrice comprising a monoalkyl phosphate as a foaming agent, which the inventors claim does not change taste of foods and beverages; Japanese Patent Laid-open (kohyo) No. 500061/1979 (USP No. 4,366,146) proposes a dentifrice for preventing formation of spots comprising a monoalkyl phosphate as a film forming material; and USP No. 4,036,950 discloses a composition for use in the oral cavity having a pH of above 6 and comprising an abrasive, a cationic antibacterial compound, and a polyoxyethylene-alkylether phosphate. Of these publications, Japanese Patent Laid-open (kokai) No. 47542/1978 and Japanese Patent Laid-open (kohyo) No. 500061/1979 does not mention the combined use of a cationic surfactant and a phosphate, and the composition proposed in USP No. 4,036,950 provides only insufficient improvement in its taste.
- Japanese Patent Laid-open (kokai) No. 218605/1990 describes a composition comprising a quaternary ammonium compound and a salt of phosphoric ester and effective as an antibacterial and antiseptic agent for use in the skin or the hair. The specification, however, does not suggest at all the possibility of applying the proposed composition to the oral cavity.
- An object of the present invention is to provide a composition for use in the oral cavity which exhibits an immediate and long-continued antibacterial activity to bacteria in the oral cavity, provides a good taste, and does not reduce its antibacterial activity even when a surfactant as a detergent is added.
- The inventors of the present invention have undertaken painstaking investigations and found that a composition comprising a specific antibacterial compound and a vehicle for use in the oral cavity provides little bitter or astringent taste, has an excellent antibacterial activity, and exhibits an immediate and long-lasting continued antibacterial effect, even if a surfactant is added. This finding has led to the completion of the present invention.
- Specifically, the above object is achieved in the present invention by the provision of a composition for use in the oral cavity comprising:
- (A) an antibacterial compound represented by the following general formula (1),
- (B) a vehicle for use in the oral cavity.
The above object is further achieved in the present invention by the provision of a composition for use in the oral cavity comprising, in addition to said components (A) and (B), - (C) a surfactant represented by the following general formula (7),
- The above object is still further achieved in the present invention by the provision of a composition for use in the oral cavity comprising,
- (A') an antibacterial compound represented by the following general formula (8),
- Other objects, features and advantages of the invention will hereinafter become more readily apparent from the following description.
- Figure 1 is a chart showing the results of the disinfection test of the composition in Example 2.
- Figure 2 is a graph showing the rate of adsorption of antibacterial compounds in the compositions according to Example 3.
- Given as examples of antibacterial compounds of said component (A) used in the composition for use in the oral cavity of the present invention are the compounds represented by the following formulas (2) to (4),
wherein at least one of R¹, R², or R³ is a linear or branched alkyl or alkenyl group with 8 to 30 carbon atoms, and others are individually a group selected from methyl group, ethyl group, benzyl group, and a group represented by one of the following formulas,
-(CH₂CH₂O)n-H (wherein n is an integer of 1 to 15),
or
R⁴ represents a group -CH₃, -CH₂CH₃, or -CH₂CH₂OH; and the counter anion X⁻ represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms. - Among compounds of general formula (2), those having a linear or branched alkyl group with 8 to 14 carbon atoms, e.g., 2-ethylhexyl group, decyl group, dodecyl group, or tetradecyl group or benzyl group, for at least one of R¹ and R², and methyl group and/or group for others are preferable.
wherein R⁵ is a linear or branched alkyl group with 10 to 20 carbon atoms, X and X⁻ may be the same or different and each individually represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or a monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms. - Examples of linear or branched alkyl groups with 10 to 20 carbon atoms represented by R⁵ in general formula (3) include decyl group, dodecyl group, tetradecyl group, hexadecyl group, octadecyl group, eicosyl group, and the like.
- Given as examples of the monoalkyl phosphate ion, monoalkenyl phosphate ion, monoalkyl phosphonate ion, or monoalkenyl phosphonate ion, which are the counter anions represented by X⁻ or X in formulas (1) to (4), are anions of the following formula (5) or (6),
wherein R⁶ is a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms and R⁷ is a hydrogen atom, an alkali metal, an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue; and
wherein R⁸ is a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms and R⁹ is a hydrogen atom, an alkali metal, an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue. - As R⁶ and R⁸ in the above general formula (5) or (6), an alkyl group with 10 to 20 carbon atoms is preferable, 12 to 16 carbon atoms more preferable, and 14 carbon atoms most preferable. Specific examples of such alkyl groups are lauryl, myristyl, and cetyl groups, and the like. R⁷ and R⁹ are normally hydrogen atoms. Some portion of counter anions may have an alkali metal (e.g., potassium, sodium), an alkyl ammonium group, an alkyl ammonium group with a substituted hydroxy group, or a basic amino acid group residue for R⁷ or R⁹.
- The amount of the component (A) in the composition for use in the oral cavity of the present invention is 0.001 to 20% by weight (hereinafter % refers to % by weight), and preferably 0.01 to 1%. An amount outside the above range is not desirable, since if it is less than 0.001%, the antibacterial strength is weakened; if greater than 20%, the stability of the formulation is poor.
- Commonly used vehicles for the oral cavity, such as those used in toothpaste, toothpowder, liquid dentifrice, mouthwash, gargle, mouth rinse, gum massage cream, troches, chewing gum, candies, or the like, can be used as vehicles for component (B) in the composition for use in the oral cavity of the present invention.
- Such vehicles for the oral cavity which can be used in the composition of the present invention include abrasives, thickening agents, moisturizers, surfactants, perfumes, sweeteners, preservatives, colorants, water, water-soluble fluorine-containing compounds, silicone, and other effective ingredients.
- Examples of abrasives include hydrogen calcium phosphate, calcium carbonate, calcium pyrophosphate, insoluble sodium metaphosphate, potassium metaphosphate, silicic acid anhydride, silicic acid hydrate, aluminum silicate, zirconium silicate, bentonite, zeolite, aluminum oxide, aluminum hydroxide, resins and mixtures of these. Among these, silicic acid anhydride, calcium carbonate, zeolite, silicic acid hydrate, and aluminum oxide are especially preferable.
- Examples of thickening agents include sodium carboxymethyl cellulose, methyl cellulose, hydroxyethyl cellulose, arginates, carrageenan, gum arabic, polyvinyl alcohol, gum tragacanth, starch, sodium polyacrylate, and the like.
- Examples of moisturizers include polyethylene glycol, propylene glycol, sorbitol, glycerine, maltitol, xylitol, and the like. Of these, glycerine, sorbitol, and the like are preferable.
- The surfactant is used as a foaming agent or a stabilizing agent for an oil-containing material. Various types of surfactants other than phosphates may be used. Examples of preferable surfactants include sodium alkyl sulfate, sodium alkylbenzenesulfonate, sodium N-acylsarcosinate, N-acylglutamates, polyoxyethylene hydrogenated castor oil, polyoxyethylene-polyoxypropylene block copolymers (pluronic type), fatty acid esters of sucrose, alkylglycosides, fatty acid esters of sorbitan, fatty acid esters of polyoxyethylenesorbitan, alkyldimethylamine oxide, carbobetaine, hydroxycarbobetaine, phosphobetaine, hydroxyphosphobetaine, sulfobetaine, hydroxysulfobetaine, and the like. Of these, water-soluble nonionic and amphoteric surfactants such as polyoxyethylene hydrogenated castor oil, polyoxyethylene-polyoxypropylene block copolymers, fatty acid esters of sucrose, alkylglycosides, fatty acid esters of sorbitan, and fatty acid esters of polyoxyethylenesorbitan, are particular preferable.
- Examples of perfumes include natural perfumes such as spearmint oil, peppermint oil, wintergreen oil, sassafras oil, clove oil, sage oil, eucalyptus oil, mayonara oil, cinnamon oil, thyme oil, lemon oil, orange oil, and the like; as well as synthetic perfumes such as 1-menthol, anethole, carvone, eugenol, thymol, methyl salicylate, and the like. Examples of sweeteners include saccharin, saccharin sodium, stevioside, neohesperidyldihydrocarcone, beruraruchine, p-methoxycinnamic aldehyde, glycyrrhizinates, aspartame (methyl aspartylphenylalanine), and the like. Examples of preservatives include benzoic acid, sodium benzoate, parahydroxybenzoic esters, and the like. Water-soluble fluorine-containing compounds which can be used include sodium fluoride, sodium monofluorophosphate, and the like. Other effective ingredients include chlorophyll compounds, sodium chloride, vitamin C, vitamin E, nicotinic acid esters, allantoinchlorohydroxy aluminum, azulene, lysozyme chloride, hinokitiol, β-glycyrrhetinic acid, dipotassium glycyrrhizinate, protease, materials extracted from herbal medicines, and the like.
- The amounts of these oral vehicles used in the composition of the present invention vary according to the formulation. In the case of a liquid composition, such as mouthrinse, mouthwash, and gargle, any vehicles among the above-mentioned oral vehicles can be incorporated with the exception of the abrasive and the thickening agent. In principle, it is desirable to incorporate 1 to 30% of the moisturizer, and 50 to 96% of alcohol and water. The ratio of alcohol and water is preferably 1:1 to 200:1, and more preferably 5:1 to 100:1. The amount of alcohol in the composition is preferably 1 to 20%.
- When the composition is in the form of a paste, such as toothpaste or liquid dentifrice, all of the above-mentioned vehicles can be incorporated. In principle, it is preferable to incorporate 10 to 75% of the abrasive, 0.5 to 5% of the thickening agent, and 10 to 85% of the moisturizer and water. The abrasive is preferably 20 to 75% in the case of a toothpaste, and 10 to 30% in the case of a liquid dentifrice.
- For a solid composition such as a toothpowder, the solid ingredients among the above-mentioned vehicles for use in the oral cavity can be blended. Basically, it is preferable to incorporate 60 to 99% of the abrasive.
- It is preferable that the total content of the perfume and the sweetener in the oral vehicle be 0.01 to 5%. The pH of the composition of the present invention is in the range of 5 to 9.5, preferably, in the range of 6 to 8.
- The component (C) used in the present invention is a surfactant represented by the above general formula (7), wherein R¹⁰ is a linear or branched alkyl or alkenyl group with 8 to 30 carbon atoms, preferably an alkyl group with 10 to 20 carbon atoms, more preferably 12 to 16 carbon atoms, and most preferably 14 carbon atoms. Specific examples include decyl, lauryl, myristyl, and cetyl groups. Z¹ and Z² are preferably a partial neutralized compound having a neutralization degree of 0.8 to 2.0, especially 1.0 to 1.8. Given as specific examples of component (C) are 1 potassium monodecylphosphate, 1.2 sodium monolaurylphosphate, 1.3 potassium monomyristylphosphate, 1.5 triethanolamine monomyristylphosphate, 1 arginine monomyristylphosphate, 1.3 arginine monomyristylphosphate, 1.2 sodium monocetylphosphate, and the like.
- These components (C), when added to a composition containing components (A) and (B), exhibit an action of further extending the period of time for which the composition maintains the antibacterial activity. Although the amount of component (C) to be incorporated into the composition for the oral cavity of the present invention varies depending on the desired antibacterial activity and the types of cation (Am+), a preferable amount, in terms of molar ratio, is in the range of (Am+)/(C)=1/2 to 1/100, with a more preferable amount being in the range of (Am+)/(C)=1/2 to 1/50. More specifically, in the case of the antibacterial compound of general formula (2), this amount is in the range of (Am+)/(C)=1/2 to 1/100, and preferably (Am+)/(C)=1/2 to 1/50; in the case of the antibacterial compound of general formula (3), it is in the range of (Am+)/(C)=1/2 to 1/20, and preferably (Am+)/(C)=1/2 to 1/10; and in the case of the antibacterial compound of general formula (4), it is in the range of (Am+)/(C)=1/2 to 1/50, and preferably (Am+)/(C)=1/2 to 1/20.
- In the present invention, the composition for use in the oral cavity can be prepared either by formulating components (A) and (B) or by formulating component (A'), instead of component (A), and the molar ratio of component (A') to component (C) satisfies (A')/(C)≦1/m. In the case of the composition in which components (A') and (C) are formulated, it is considered that these components (A') and (C) react in the composition to form a salt.
- Given as examples of antibacterial compounds (8) of component (A') are components represented by the following general formulas (9), (10), and (11).
wherein counter anions Y⁻ and Y - In the ratio of component (A') to component (C) satisfying the range defined above, an excess amount of component (C) extends the period of time for which the composition maintains the antibacterial activity. In the case where component (C) is added in excess, the molar ratio of (Am+)/(C) is preferably (Am+)/(C) =1/2 to 1/100, and more preferably (Am+)/(C)=1/2 to 1/50, although it depends on the types of the antibacterial cation (Am+). More specifically, in the case of the antibacterial compound of general formula (9), this molar ratio is in the range of (Am+)/(C)=1/2 to 1/100, and preferably (Am+)/(C)=1/2 to 1/50; in the case of the antibacterial compound of general formula (10), it is in the range of (Am+)/(C)=1/2 to 1/20, and preferably (Am+)/(C)=1/2 to 1/10; and in the case of the antibacterial compound of general formula (11), it is in the range of (Am+)/(C)=1/2 to 1/50, and preferably (Am+)/(C)=1/2 to 1/20.
- The compounds represented by the general formula (1) used in the present invention can be prepared according to known methods. For example, among compounds of the general formula (1), said compound (2) can easily be prepared from a commercially available quaternary ammonium salt, which is a compound represented by said general formula (9) with a halogen atom as the counter ion, by converting it into the OH-type quaternary ammonium salt, and neutralizing it by said compound having an anion residue. Said compound (3) can be prepared by a method comprising submitting a commercially available compound (10) to alkaline conditions, desalting it by electrodialysis, and neutralizing the product with said compound having an anion residue. An alkali metal salt, an amine salt, an organic amine salt, or a basic amino acid salt may be included during the neutralization to the extent that the neutralization is not hindered.
- Preferable formulas of the composition for use in the oral cavity of the present invention include liquid formulas such as mouthwash, mouth rinse, and gargle; paste formulas such as toothpaste, liquid dentifrice, and gum massage cream; and solid formulas such as toothpowder, and troche.
- Among the above compositions for use in the oral cavity of the present invention, a solid composition such as a toothpowder can normally be formulated by mechanically blending the above-mentioned solid ingredients. A composition in the form of a paste can normally be formulated by mechanically blending the various ingredients with deaeration.
- In order to disinfect the oral cavity by using the composition for use in the oral cavity of the present invention, the composition is, in principle, preferably applied about 1-3 times per day, about five days a week. Among the forms of the composition, a mouth rinse is the most desirable. Disinfection can be effectively performed using about 10 ml of the mouth rinse of the present invention after brushing, holding in the mouth for about 30 seconds, then ejecting.
- The composition for use in the oral cavity of the present invention exhibits a remarkablely rapid disinfection effect and continued antibacterial effect action against oral cavity bacteria while providing a good taste. Its disinfection effect is not reduced in the presence of surfactants.
- Other features of the invention will become apparent in the course of the following description of the exemplary embodiments which are given for illustration of the invention and are not intended to be limiting thereof.
- The following compounds were used in the Examples.
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- Antibacterial compounds in Table 1 were evaluated in terms of their taste.
- Evaluation was performed by a panel of 5 adult men and 5 adult women. 20 ml of a 1% ethanol solution containing 0.02% or 0.1% of one of the compounds in Table 1 was held in the mouth for 30 seconds to evaluate the taste while gargling and the aftertaste as one of three grades, O (not bad), D (rather bad), or X (bad). Each compound was graded as one of O, D, or X, to which the average of all panelists is closest. The results are shown in Table 1, which manifestly demonstrates a remarkable improvement in the taste of the composition of the present invention.
- A disinfection test was carried out on the compositions shown in Tables 2 and 3.
- Bacteria collected from saliva of male adults and bacteria causing diseases in the oral cavity, which are named in Table 2, were used for the test. The antibacterial compounds shown in Tables 2 and 3 were added to the test bacterial suspensions to a concentration of 0.01% in a anaerobic globe box (N₂:CO₂:H₂=80:10:10, manufactured by Hirasawa Co.), followed by the further addition of surfactants shown in Tables 2 and 3 to a concentration of 0.1%. The mixture was stirred for 1 minute at 37°C, suitably diluted with physiological saline, and cultured for 5 days at 37°C in BHI agar medium (goat blood hemin and menadione containing, a product of Difco Co.) to measure the number of living bacteria. The results are shown in Table 2 and Figure 1, which show that the compositions for the oral cavity of the present invention exhibited a remarkablly rapid disinfection effect which is not reduced in the presence of surfactants.
- Benzalkonium chloride (Sanizole, trade mark, a product of Kao Corp.), chlorhexidine gluconate, and cetylpyridinium chloride were selected as antibacterial compounds. An appropriate amount of arginine laurylphosphate (1.3 neutralization) was added to prepare aqueous solutions of antibacterial compound with a concentration of 0.01%. 1 g of hydroxyapatite powder (a product of Wako Pure Chemical Co.), which is a major component of the tooth was added to 20 ml of each solution. After stirring for 30 seconds, the amount the antibacterial compounds in the supernatant was quantitatively measured to calculate their adsorption to the hydroxyapatite.
- The results are shown in Figure 2, which demonstrates that the rate of adsorption is promoted by the addition of arginine laurylphosphate. It was further confirmed that especially good results could be obtained by adjusting the equivalent ratio of benzalkonium chloride and arginine laurylphosphate to 1:1 to 1:100, the molar ratio of chlorhexidine gluconate and arginine laurylphosphate to 1:1 to 50:1, and the equivalent ratio of cetylpyridinium chloride and arginine laurylphosphate to 1:1 to 1:20.
- The above results show that the use of the composition for the oral cavity of the present invention can effectively adsorb antibacterial compound to the tooth surface to continuously exhibit the antibacterial action.
- Water, 1 mM benzalkonium chloride, 0.5 mM myristylphospholylated di(benzarconium) (1 mM as benzarconium), 1 mM chlorhexidine gluconate, 1 mM cetylpyridinium chloride, 0.5 mM myristylphospholylated di(cetylpyridinium), and 1 mM monomyristylphospholylated chlorhexidine were used as comparative compounds, and 1 mM myristylphospholylated benzalkonium, 1 mM dimyristylphospholylated chlorhexidine, and 1 mM myristylphospholylated cetylpyridinium, to which an appropriate amount of arginine myristylphosphate was added (1.5 neutralization) were used as invention compounds.
- Each sample solution was evaluated in terms of the taste during mouthwash and the aftertaste by adult persons by holding 10 ml of the solution in the mouth for 30 seconds while gargling. The rate of bacterial metabolism suppression was determined by collecting saliva after 30 and 60 minutes and measuring its lactic acid producing capability, which is taken as the standard for the antibacterial activity. For the determination of the lactic acid producing capability 0.4 ml of saliva and 0.1 ml of 10% sucrose were reacted for 5 minutes at 37°C, and the reaction product was filtered and submitted to ion chromatography. The rate of suppression of lactic acid production, as the percentage against the amount of lactic acid produced when the person did not wash mouth, was calculated by deducting the amount of lactic acid produced while unreacted as a blank to determine the lactic acid production. The results are shown in Table 4, which indicates that the comparative compounds had unfavorable tastes and hardly maintained their antibacterial effects, while the compounds of the present invention not only provided good taste, but also was proven to promote their prolonged antibacterial effects by the addition of a specific amount of arginine myristylphosphate. Among the above compounds, those having the molar ratio of (antibacterial cation)/(counter anion) greater than the reciprocal of the cation valency, such as myristylphospholylated di(benzalkonium), myristylphospholylated di(cetylpyridinium), and monomyristylphospholylated chlorhexidine, exhibited both unfavorable tastes and poor effects of the continued antibacterial action. On the other hand, the compounds having the molar ratio of (antibacterial cation)/(counter anion) smaller than the reciprocal of the cation valency, such as myristylphospholylated benzarkonium, myristylphospholylated cetylpyridinium, and dimyristylphospholylated chlorhexidine, exhibited both good tastes and excellent effects of the continued antibacterial action.
-
-
-
- An aqueous solution of benzethonium chloride or cetylpyridinium chloride and an aqueous solution of sodium monoalkyl (C₁₂) phosphate (pH 7) were mixed to produce white precipitate. To the mixture were added ethanol and polyoxyethylene (EO=40) hydrogenated castor oil to solubilize the precipitate. Mouthwash compositions 11 and 12 were prepared by adding other components shown in Table 1 and blending the mixture.
-
Claims (10)
- A composition for use in the oral cavity comprising:(A) an antibacterial compound represented by the following general formula (1),
- A composition for use in the oral cavity according to Claim 1, wherein said antibacterial compound (A) is a compound represented by the following formula (2),
- A composition for use in the oral cavity according to Claim 1, wherein said antibacterial compound (A) is a compound represented by the following formula (3) or (4),
individually represents a monoalkyl phosphate ion, a monoalkenyl phosphate ion, a monoalkyl phosphonate ion, or a monoalkenyl phosphonate ion having a linear or branched alkyl or alkenyl group with 8 to 20 carbon atoms. - A composition for use in the oral cavity according to Claims 1-3, wherein said counter anion X⁻ is a phosphate ion represented by the following general formula (5),
- A composition for use in the oral cavity according to Claims 1-3, wherein said counter anion X⁻ is a phosphonate ion represented by the following general formula (6),
- A composition for use in the oral cavity according to Claim 1, further comprising,(C) a surfactant represented by the following general formula (7),
- A composition for use in the oral cavity comprising:(A') an antibacterial compound represented by the following general formula (8),
wherein the molar amount of component (C) is m times or more of that of component (A'). - A composition for use in the oral cavity according to Claim 7, wherein said antibacterial compound (A') is a compound represented by the following formula (9),
- A composition for use in the oral cavity according to Claim 7, wherein said antibacterial compound (A') is a compound represented by the following formula (10) or (11),
- Use of a composition comprising compound (A) of Claim 1 for the preparation of a medicament to be used in oral cavity.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP26746/92 | 1992-02-13 | ||
| JP2674692 | 1992-02-13 | ||
| JP12599892 | 1992-05-19 | ||
| JP125998/92 | 1992-05-19 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0555864A1 true EP0555864A1 (en) | 1993-08-18 |
| EP0555864B1 EP0555864B1 (en) | 1996-05-01 |
Family
ID=26364559
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP93102227A Expired - Lifetime EP0555864B1 (en) | 1992-02-13 | 1993-02-12 | Composition for use in oral cavity |
Country Status (3)
| Country | Link |
|---|---|
| US (1) | US5374418A (en) |
| EP (1) | EP0555864B1 (en) |
| DE (1) | DE69302413T2 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0569666A3 (en) * | 1992-05-15 | 1994-10-12 | Kao Corp | Composition comprising a monophosphate for use in the oral cavity. |
| US5451401A (en) * | 1993-09-29 | 1995-09-19 | The Procter & Gamble Company | Diphosphonic acid esters as tartar control agents |
| US5628986A (en) * | 1994-11-18 | 1997-05-13 | The Procter & Gamble Company | Oral compositions |
| WO2003003832A1 (en) * | 2001-07-06 | 2003-01-16 | Rnl Life Science Ltd. | Acid disinfectant composition comprising an anion surfactant |
| WO2010004361A2 (en) * | 2008-06-17 | 2010-01-14 | The Procter & Gamble Company | Compositions and methods for improving overall tooth health and appearance |
| GB2533527A (en) * | 2015-04-02 | 2016-06-22 | Byotrol Plc | Anti-microbial composition |
Families Citing this family (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5605676A (en) * | 1995-06-26 | 1997-02-25 | Colgate-Palmolive Company | Oral composition exhibiting improved uptake and retention of antibacterial compounds on dental tissue surfaces |
| US5827503A (en) * | 1996-08-08 | 1998-10-27 | Collagenex Pharmaceuticals, Inc. | Method and composition for treating periodontitis |
| US5800803A (en) * | 1997-02-10 | 1998-09-01 | Colgate-Palmolive Company | Oral composition exhibiting enhanced uptake by dental tissue of noncationic antibacterial agents |
| US6248309B1 (en) * | 1997-04-04 | 2001-06-19 | Optiva Corporation | Gums containing antimicrobial agents |
| US5939050A (en) * | 1997-04-04 | 1999-08-17 | Optiva Corp. | Antimicrobial compositions |
| US6035634A (en) | 1999-02-09 | 2000-03-14 | Latch-Tool Development Co. Llc | Compact, resistance regulated, multiple output hydraulic tool and seal valve arrangement |
| US8283135B2 (en) * | 2000-06-30 | 2012-10-09 | The Procter & Gamble Company | Oral care compositions containing combinations of anti-bacterial and host-response modulating agents |
| US20050048007A1 (en) * | 2000-11-02 | 2005-03-03 | Inobys Ltd. | Plaque reducing composition |
| RU2337734C2 (en) * | 2003-02-21 | 2008-11-10 | Родиа Инк. | Ultrasoft agent for hygienic oral cavity care to prevent sensitivity, caries, staining and dental deposit |
| US9241885B2 (en) * | 2004-01-29 | 2016-01-26 | The Procter & Gamble Company | Oral care compositions comprising increased bioavailable levels of quaternary ammonium antimicrobials |
| US9242125B2 (en) * | 2005-07-21 | 2016-01-26 | Coglate-Palmolive Company | Oral composition containing non-aggregated zinc nanoparticles |
| US20090202456A1 (en) * | 2008-02-08 | 2009-08-13 | Colgate-Palmolive Company | Novel salts and their uses |
| US8905760B2 (en) * | 2008-11-04 | 2014-12-09 | Duane C. Keller | Methods and systems for progressively treating and controlling oral periopathogens causing systemic inflammations |
| US8956161B2 (en) | 2008-11-04 | 2015-02-17 | Duane C Keller | Article and method for controlling oral-originated systemic disease |
| CA2773738C (en) * | 2009-09-11 | 2016-06-07 | The Procter & Gamble Company | Methods and compositions for hydrophobic modification of oral cavity surfaces |
| US8591229B2 (en) | 2010-12-16 | 2013-11-26 | Duane C. Keller | Devices and methods for creating a positive pressure environment for treatment of oral biofilms associated with periodontal disease |
| US20160228339A1 (en) | 2013-09-18 | 2016-08-11 | Glymur B.V. | Oral hygiene compositions |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2192801A1 (en) * | 1972-07-17 | 1974-02-15 | Unilever Nv | |
| DE2600708A1 (en) * | 1975-01-15 | 1976-07-22 | Colgate Palmolive Co | PREPARATIONS FOR ORAL AND DENTAL CARE |
| DE2744980A1 (en) * | 1976-10-12 | 1978-04-13 | Kao Corp | Dentifrice |
| EP0150374A2 (en) * | 1983-12-17 | 1985-08-07 | Henkel Kommanditgesellschaft auf Aktien | Mouth and tooth care compositions |
| EP0371542A2 (en) * | 1988-12-01 | 1990-06-06 | Unilever N.V. | Oral composition |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3864472A (en) * | 1972-11-06 | 1975-02-04 | Colgate Palmolive Co | Clear lemon-flavored mouthwash |
| US4064138A (en) * | 1975-11-12 | 1977-12-20 | General Mills, Inc. | Amino acid derivatives |
| US4256731A (en) * | 1978-03-23 | 1981-03-17 | Beecham, Inc. | Oral Compositions |
| US4536519A (en) * | 1981-06-15 | 1985-08-20 | Kao Soap Co., Ltd. | Emulsifying agent and emulsified cosmetics |
| US4391798A (en) * | 1982-03-15 | 1983-07-05 | Colgate-Palmolive Company | Non-irritating dentifrice |
| US5128122A (en) * | 1984-03-19 | 1992-07-07 | The Rockefeller University | Method and agents for preventing staining of teeth |
| US4670575A (en) * | 1984-06-05 | 1987-06-02 | Kao Corporation | Process for purification of phosphoric mono esters |
| JPS62258731A (en) * | 1986-05-06 | 1987-11-11 | Kao Corp | Oil-in-water-type emulsified composition |
| JPS63225312A (en) * | 1986-10-02 | 1988-09-20 | Kao Corp | Transparent or semitransparent jelly-like cosmetic |
| US5064640A (en) * | 1986-12-02 | 1991-11-12 | Purdue Research Foundation | Anticariogenic compositions |
| US4997672A (en) * | 1987-03-10 | 1991-03-05 | Virginia Commonwealth University | Salt taste enhancer |
| JPH0667818B2 (en) * | 1988-06-20 | 1994-08-31 | 花王株式会社 | Translucent emulsion cosmetics |
| JPH0825861B2 (en) * | 1989-04-24 | 1996-03-13 | サンスター株式会社 | Toothpaste composition |
-
1993
- 1993-02-11 US US08/016,234 patent/US5374418A/en not_active Expired - Fee Related
- 1993-02-12 EP EP93102227A patent/EP0555864B1/en not_active Expired - Lifetime
- 1993-02-12 DE DE69302413T patent/DE69302413T2/en not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2192801A1 (en) * | 1972-07-17 | 1974-02-15 | Unilever Nv | |
| DE2600708A1 (en) * | 1975-01-15 | 1976-07-22 | Colgate Palmolive Co | PREPARATIONS FOR ORAL AND DENTAL CARE |
| DE2744980A1 (en) * | 1976-10-12 | 1978-04-13 | Kao Corp | Dentifrice |
| EP0150374A2 (en) * | 1983-12-17 | 1985-08-07 | Henkel Kommanditgesellschaft auf Aktien | Mouth and tooth care compositions |
| EP0371542A2 (en) * | 1988-12-01 | 1990-06-06 | Unilever N.V. | Oral composition |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0569666A3 (en) * | 1992-05-15 | 1994-10-12 | Kao Corp | Composition comprising a monophosphate for use in the oral cavity. |
| US5451401A (en) * | 1993-09-29 | 1995-09-19 | The Procter & Gamble Company | Diphosphonic acid esters as tartar control agents |
| US5628986A (en) * | 1994-11-18 | 1997-05-13 | The Procter & Gamble Company | Oral compositions |
| WO2003003832A1 (en) * | 2001-07-06 | 2003-01-16 | Rnl Life Science Ltd. | Acid disinfectant composition comprising an anion surfactant |
| WO2010004361A2 (en) * | 2008-06-17 | 2010-01-14 | The Procter & Gamble Company | Compositions and methods for improving overall tooth health and appearance |
| WO2010004361A3 (en) * | 2008-06-17 | 2010-09-10 | The Procter & Gamble Company | Compositions and methods for improving overall tooth health and appearance |
| CN102056648A (en) * | 2008-06-17 | 2011-05-11 | 宝洁公司 | Compositions and methods for improving overall tooth health and appearance |
| CN102056648B (en) * | 2008-06-17 | 2014-11-05 | 宝洁公司 | Compositions and methods for improving overall tooth health and appearance |
| GB2533527A (en) * | 2015-04-02 | 2016-06-22 | Byotrol Plc | Anti-microbial composition |
| GB2533527B (en) * | 2015-04-02 | 2018-10-17 | Byotrol Plc | Anti-microbial composition |
Also Published As
| Publication number | Publication date |
|---|---|
| DE69302413D1 (en) | 1996-06-05 |
| EP0555864B1 (en) | 1996-05-01 |
| DE69302413T2 (en) | 1996-10-17 |
| US5374418A (en) | 1994-12-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0555864B1 (en) | Composition for use in oral cavity | |
| EP0569666B1 (en) | Composition comprising a monophosphate for use in the oral cavity | |
| US5015466A (en) | Anticalculus compositions using tartrate-succinates | |
| US5338537A (en) | Oral compositions | |
| US5019373A (en) | Oral composition | |
| US5158763A (en) | Non-staining anti-bacterial oral composition | |
| US3932606A (en) | Dentifrice | |
| EP0251591A1 (en) | Oral hygiene composition | |
| US4574081A (en) | Antiplaque dentifrice having improved flavor | |
| GB2057264A (en) | Plague inhibiting oral compositions | |
| JPS606609A (en) | Oral cavity product | |
| JPH10501266A (en) | Mouth rinse composition | |
| EP0539480B1 (en) | Anticalculus/antiplaque compositions using azacycloalkane diphosphonates | |
| GB2195535A (en) | Stable antiplaque dentifrice | |
| JPS63277612A (en) | Composition for oral cavity | |
| US3988435A (en) | Dihydrochalcone glycoside derivatives and pharmaceutical compositions containing same | |
| NZ231330A (en) | Antibacterial antiplaque oral composition containing an additive that prevents the staining of dental surfaces | |
| JP2534188B2 (en) | Oral composition | |
| JP2627709B2 (en) | Oral composition | |
| JPH05320032A (en) | Composition for oral cavity application | |
| EP0658340A1 (en) | Oral compositions | |
| JPH10324619A (en) | Composition for oral cavity | |
| GB2164255A (en) | Stable antiplaque dentifrice | |
| JPH07215830A (en) | Composition for oral cavity | |
| JPH05229923A (en) | Composition for oral cavity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): DE FR GB |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: HIOKI, YUICHI Inventor name: IWASAKI, TETSUJI Inventor name: EGUCHI, YASUTERU Inventor name: NAKAI, RYOZO Inventor name: YAMAGISHI, ATSUSHI Inventor name: OSHINO, KAZUHI |
|
| 17P | Request for examination filed |
Effective date: 19931123 |
|
| 17Q | First examination report despatched |
Effective date: 19950123 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB |
|
| REF | Corresponds to: |
Ref document number: 69302413 Country of ref document: DE Date of ref document: 19960605 |
|
| ET | Fr: translation filed | ||
| GRAH | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOS IGRA |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20070207 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20070208 Year of fee payment: 15 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20070208 Year of fee payment: 15 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20080212 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST Effective date: 20081031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080902 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080229 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20080212 |