EP0102246B1 - Verpackung und Verdichtung eines teilchenförmigen Materials - Google Patents
Verpackung und Verdichtung eines teilchenförmigen Materials Download PDFInfo
- Publication number
- EP0102246B1 EP0102246B1 EP19830304974 EP83304974A EP0102246B1 EP 0102246 B1 EP0102246 B1 EP 0102246B1 EP 19830304974 EP19830304974 EP 19830304974 EP 83304974 A EP83304974 A EP 83304974A EP 0102246 B1 EP0102246 B1 EP 0102246B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- bellows
- container
- bellows container
- canister
- refractory
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired
Links
- 238000000280 densification Methods 0.000 title claims description 21
- 239000011236 particulate material Substances 0.000 title claims description 8
- 238000000034 method Methods 0.000 claims description 41
- 239000000463 material Substances 0.000 claims description 29
- 238000003825 pressing Methods 0.000 claims description 24
- 239000011435 rock Substances 0.000 claims description 23
- 238000010438 heat treatment Methods 0.000 claims description 21
- 230000006698 induction Effects 0.000 claims description 15
- 238000007906 compression Methods 0.000 claims description 14
- 230000006835 compression Effects 0.000 claims description 14
- 230000015572 biosynthetic process Effects 0.000 claims description 8
- 238000007731 hot pressing Methods 0.000 claims description 8
- 239000002901 radioactive waste Substances 0.000 claims description 8
- 238000003860 storage Methods 0.000 claims description 8
- 239000002243 precursor Substances 0.000 claims description 7
- 239000002002 slurry Substances 0.000 claims description 6
- 229910052751 metal Inorganic materials 0.000 claims description 4
- 239000002184 metal Substances 0.000 claims description 4
- 239000002245 particle Substances 0.000 claims description 3
- 239000011819 refractory material Substances 0.000 claims description 3
- 238000007789 sealing Methods 0.000 claims description 3
- 230000009471 action Effects 0.000 claims description 2
- 238000005507 spraying Methods 0.000 claims description 2
- 238000005192 partition Methods 0.000 claims 3
- 239000002699 waste material Substances 0.000 description 19
- 230000008569 process Effects 0.000 description 11
- 239000002927 high level radioactive waste Substances 0.000 description 7
- 230000002285 radioactive effect Effects 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 229910000831 Steel Inorganic materials 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 230000007774 longterm Effects 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- 239000000243 solution Substances 0.000 description 4
- 239000010959 steel Substances 0.000 description 4
- 229910002651 NO3 Inorganic materials 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 3
- 239000007789 gas Substances 0.000 description 3
- 229910052500 inorganic mineral Inorganic materials 0.000 description 3
- 239000011707 mineral Substances 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 2
- 239000000470 constituent Substances 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 239000001257 hydrogen Substances 0.000 description 2
- 229910052739 hydrogen Inorganic materials 0.000 description 2
- 238000002386 leaching Methods 0.000 description 2
- 230000033001 locomotion Effects 0.000 description 2
- 150000002823 nitrates Chemical class 0.000 description 2
- 239000006104 solid solution Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- VXUYXOFXAQZZMF-UHFFFAOYSA-N titanium(IV) isopropoxide Chemical compound CC(C)O[Ti](OC(C)C)(OC(C)C)OC(C)C VXUYXOFXAQZZMF-UHFFFAOYSA-N 0.000 description 2
- 229910001093 Zr alloy Inorganic materials 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 229910000963 austenitic stainless steel Inorganic materials 0.000 description 1
- 239000005388 borosilicate glass Substances 0.000 description 1
- BSDOQSMQCZQLDV-UHFFFAOYSA-N butan-1-olate;zirconium(4+) Chemical compound [Zr+4].CCCC[O-].CCCC[O-].CCCC[O-].CCCC[O-] BSDOQSMQCZQLDV-UHFFFAOYSA-N 0.000 description 1
- 238000002144 chemical decomposition reaction Methods 0.000 description 1
- 238000005056 compaction Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000004031 devitrification Methods 0.000 description 1
- 239000010419 fine particle Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000005484 gravity Effects 0.000 description 1
- 230000003100 immobilizing effect Effects 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 238000005342 ion exchange Methods 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000003758 nuclear fuel Substances 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- RVTZCBVAJQQJTK-UHFFFAOYSA-N oxygen(2-);zirconium(4+) Chemical compound [O-2].[O-2].[Zr+4] RVTZCBVAJQQJTK-UHFFFAOYSA-N 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000011343 solid material Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000011885 synergistic combination Substances 0.000 description 1
- 229910001928 zirconium oxide Inorganic materials 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G21—NUCLEAR PHYSICS; NUCLEAR ENGINEERING
- G21F—PROTECTION AGAINST X-RADIATION, GAMMA RADIATION, CORPUSCULAR RADIATION OR PARTICLE BOMBARDMENT; TREATING RADIOACTIVELY CONTAMINATED MATERIAL; DECONTAMINATION ARRANGEMENTS THEREFOR
- G21F9/00—Treating radioactively contaminated material; Decontamination arrangements therefor
- G21F9/28—Treating solids
- G21F9/34—Disposal of solid waste
Definitions
- the nuclear reactor waste is incorporated into the crystal lattices of the synthetic rock in the form of a dilute solid solution and therefore should be safely immobilised.
- a dense, compact, mechanically strong block of the synthetic rock incorporating the nuclear waste is produced by pressure and heat in a densification process and the block may then be safely disposed of in a suitable geological formation.
- the high working temperatures for the densification step are best achieved by the use of induction heating and therefore typically it takes many hours for the contents of the bellows container to come to a uniform working temperature. Therefore preheating of the bellows container to bring the contents up to a uniform temperature suitable for the densification step is a major advantage. Not only can the production rate for given capital cost be maximized but furthermore a substantial further advantage is that bringing the contents of the bellows container to the uniform densification temperature aids reliable and uniform densification thereby ensuring reliable axial compression of the bellows container which facilitates its later handling and storage.
- the bellows container is typically of a heat resistant steel and preferably a stainless steel. Inevitably the mechanical strength of the steel is reduced at the high densification temperatures in the region of 1100 to 1200°C.
- Such a method may be defined as consisting in a method for the containment of particulate waste material, the method comprising pouring the waste material into bellows containers of generally cylindrical form with a side wall including a bellows-like formation and of heat and decay resistant material, closing each bellows container with a lid, preheating in series the bellows containers to bring the contents thereof to a substantially uniform elevated temperature, placing each bellows container in turn on an upwardly displaceable ram and displacing the ram upwardly to insert the bellows container into a cylindrical canister and applying pressure and maintaining a sufficiently elevated temperature for sufficient time to cause densification of the contents of the bellows container with axial compression of the bellows container and relatively slight outward expansion thereof to cause the bellows container to grip the interior wall of the cylindrical canister, and when the canister has been filled with a series of such bellows containers, sealing the canister and removing the canister for storage.
- the invention is particularly useful in relation to the incorporation of high level radioactive waste in synthetic rock of the type described by A. E. Ringwood (and referred to above), the invention can also be applied to other synthetic rock arrangements and furthermore can also be applicable to other materials which require storage and are capable of compaction under heat and pressure.
- One example of such other material would be shredded waste zirconium alloy nuclear fuel rod tubes and similar waste components.
- the invention consists in a combination of steps which cooperate together in an advantageous relationship which permits efficient, economic, and convenient operations in a hot cell.
- the apparatus used can be relatively simple, and this can contribute greatly to the reliability and acceptability of the system due to simplicity of servicing and intrinsic reliability.
- the material is preferably provided in the form of well graded fine particles up to about 2 mm maximum dimension whereby a readily pourable material is provided which can be easily densified in the process.
- a preferred embodiment of the invention can also provide further means for safeguarding the cylindrical canister from outward deformation under the pressure of expanding bellows containers within the canister.
- This is achievable by the use of a block of refractory material having a slightly tapered bore which at its narrowest diameter just fits over the canister, the refractory block being adapted to be moved downwardly in a series of steps corresponding to bellows container locations, the slightly tapered bores permitting release of the block even if some outward deformation of the canister has taken place in a step of densification and compression of the bellows container.
- the refractory block is formed so as to embrace the induction heating coil for surrounding the canister.
- the refractory block comprises a series of interlocking refractory segments arranged to be mounted inside a cylindrical containment shroud which absorbs any expansion forces applied from the canister.
- apparatus for encapsulating particulate supply material in bellows containers within a cylindrical canister comprising means for pouring the particulate material into a bellows container, means for sealing the bellows container with a lid, means for moving bellows containers in sequence to a pressing station at which the container is heated and axial pressure is applied to the container causing the axial compression of the container, characterized in that said pressing station comprising an upwardly displaceable ram for receiving a bellows container, means for mounting a cylindrical canister with an open end directed downwardly towards said ram, means for upwardly pressing a bellows container supported on the ram into the canister, upper refractory support means to act as a fixed abutment for the canister and the bellows container pressed upwardly therein by said ram, heating means for maintaining an elevated temperature in said bellows container whilst said pressure is applied to cause densification of said supply material in the bellows container and to expand slightly the bell
- the process has a preliminary mixing stage 21 in which synthetic rock precursor from supply 20 is formed into a slurry with high level radioactive waste from waste supply 19 which is in the form of a nitrate solution, and the slurry is passed along line 22 to be sprayed into the elevated temperature end of a rotary kiln, at which a maximum temperature in the range 700-800 0 is maintained.
- the spraying step immediately vaporises the water content of the slurry sprayed into the rotary kiln and causes chemical decomposition of the radioactive nitrates and will cause the mineral components of the synthetic rock to start to form with the radioactive elements starting to go into mineral phases.
- the particulate material produced by the rotary kiln is fed into a titanium mixing stage 24 which receives metallic titanium powder from a hopper 25 whereby the mixture poured into a bellows container 20 has about 2% titanium metal powder by weight.
- the precursor material is a product which possesses a very high surface area and functions as an effective ion exchange medium, which is mixed with additives containing Ca, Ba, and AI in solution and mixed in a hot cell with high level nuclear waste (HLW) in the form of nitrate solution to form a thick homogeneous slurry at mixing stage 21.
- HMW high level nuclear waste
- up to about 20% by weight of the solid content of the slurry may comprise the high level wastes.
- the bellows container 20 is of a heat resisting steel such as an austenitic stainless steel, for example Sandvik grade 253MA which retains reasonable mechanical strength even at the elevated temperatures used in the process, although at these temperatures the container is relatively ductile.
- a thin perforated metal liner 26 is located within the bellows container and the space between the liner and bellows wall is filled with zirconium oxide powder 27.
- a stainless steel cap 29 is used to seal the bellows container which is then placed between a pair of pistons 30 for a cold pressing operation which can increase the density of particulate material from about 25% of the theoretical maximum density to about 36%.
- each bellows container 20 is fed in sequence into a vertical induction furnace 31, each bellows container being supported on a refractory disk 32, the lowermost refractory disk being supported by a retractable latch 33. Over a period of several hours the temperature gradually increases up to about 1200°C.
- a first water cooled ram 34 having a top spigot on which a refractory plate 35 is located is adapted to support and lower one at a time the bellows containers from the furnace for horizontal movement across a support table 36 to a pressing station having a second water cooled ram 37 of similar form.
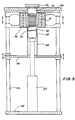
- Figure 2 shows the ram 37 both in the lower receiving position and also in the upwardly displaced pressing position inside a metal canister 38 mounted on a support 39 and having its top sealed and in abutment against a fixed refractory block 40, vertically displaceable induction heating coils 41 being provided outside the canister 38.
- the left hand side of the section of a bellows container 20a is shown in its configuration before hot pressing and the right hand side of the section shows a bellows container 20b as it would be after pressing.
- the bellows container slightly expands to become an interference fit within the canister 38 as shown by bellows 20c at the top of the canister 38.
- the refractory plate 32 upon which each of the bellows containers is supported is removed after the pressing stage, the plate 32 being lowered on the water cooled ram 37 and then pushed onto a receiving table from which the plate can be recycled for further use.
- Refractory plates will wear in use and must be replaced and an important advantage of the design illustrated in Figure 2 is a very simple and easily serviced arrangement made possible by the use of an upward pressing technique; this permits the replaceable refractory top plate 35 simply to sit on the head of each water cooled ram. Just a simple spigot and socket engagement is provided so that manipulators can readily remove a worn refractory plate and insert a new one.
- the apparatus further includes a base plate 42 with a set of upstanding tubular guides 43 on which sliding mounts for the support 39 and the induction furnace unit 41 are slidably mounted but adapted to be clamped at any selected position.
- the canister 38 is urged upwardly against the refractory block 40 which is supported by a top cap 44 adapted to be bolted to a top plate 45.
- Figure 3 shows the parts in slightly exploded view for clarity.
- the induction heating coil 41 is shown embedded within a refractory block 46 having a tapered bore, the drawing showing a greatly exaggerated taper and clearance between the bore and the container 38.
- the object of the tapered bore of the refractory block 46 is that any small expansion of the canister 38 causes the canister to be supported against further outward deformation by the refractory block but by virtue of the taper the refractory block can be released by downward motion to the next location for the succeeding bellows container.
- the refractory block is assembled from refractory segments comprising outer refractory segments 46a of cylindrical profile and inner refractory elements 46b having an inner profile adapted to cooperate to form a tapered bore with circumferentially extending grooves for accommodating the turns of the induction coil 41.
- the refractory elements are contained within a steel outer support cylinder 47 which absorbs the forces of any outward expansion applied by the canister 38.
- Figure 5 shows in isometric view the refractory blocks 46a and 46b each having a semi-circular rib 46c on one side thereof and a corresponding cavity 46d on the other side for interengagement purposes.
Landscapes
- Engineering & Computer Science (AREA)
- Environmental & Geological Engineering (AREA)
- Physics & Mathematics (AREA)
- General Engineering & Computer Science (AREA)
- High Energy & Nuclear Physics (AREA)
- Processing Of Solid Wastes (AREA)
Claims (20)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT83304974T ATE30649T1 (de) | 1982-08-30 | 1983-08-30 | Verpackung und verdichtung eines teilchenfoermigen materials. |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU18163/83A AU552755B2 (en) | 1982-08-30 | 1982-08-30 | Containment of waste material |
| AU5670/82 | 1982-08-30 | ||
| AUPF567082 | 1982-08-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0102246A1 EP0102246A1 (de) | 1984-03-07 |
| EP0102246B1 true EP0102246B1 (de) | 1987-11-04 |
Family
ID=25617056
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19830304974 Expired EP0102246B1 (de) | 1982-08-30 | 1983-08-30 | Verpackung und Verdichtung eines teilchenförmigen Materials |
Country Status (1)
| Country | Link |
|---|---|
| EP (1) | EP0102246B1 (de) |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IT1176516B (it) * | 1984-07-31 | 1987-08-18 | Agip Spa | Procedimento per la immobilizzazione di elementi di prodotti di fissione e/o elementi transuranici contenuti in scorie liquide radioattive ed apparecchiatura atta allo scopo |
| EP0209339A3 (de) * | 1985-07-16 | 1988-06-08 | AUSTRALIAN NUCLEAR SCIENCE & TECHNOLOGY ORGANISATION | Induktionsheizgerät und Verfahren |
| EP0211533A1 (de) * | 1985-07-16 | 1987-02-25 | AUSTRALIAN NUCLEAR SCIENCE & TECHNOLOGY ORGANISATION | Anpressschuh für heiss gepresste Rückstände in Behältern |
| EP0228816B1 (de) * | 1985-11-29 | 1991-04-10 | Australian Nuclear Science And Technology Organisation | Vorrichtungen für Vibrationsbehandlung |
| EP0230732B1 (de) * | 1985-11-29 | 1991-05-02 | Australian Nuclear Science And Technology Organisation | Erzeugung von Keramiken |
| CN120426780B (zh) * | 2025-07-04 | 2025-09-02 | 江苏皓越真空设备有限公司 | 一种sps烧结设备用水冷压头 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3169647D1 (en) * | 1980-05-19 | 1985-05-09 | Asea Ab | Method for treating radioactive material and container for enclosing such material |
| EP0044692B1 (de) * | 1980-07-15 | 1986-10-08 | AUSTRALIAN NUCLEAR SCIENCE & TECHNOLOGY ORGANISATION | Vorrichtungen zum Einschliessen von Abfällen |
| DE3129852C2 (de) * | 1981-07-29 | 1985-05-23 | GNS Gesellschaft für Nuklear-Service mbH, 4300 Essen | Verfahren zur Paketierung von radioaktiven Abfallstoffen |
-
1983
- 1983-08-30 EP EP19830304974 patent/EP0102246B1/de not_active Expired
Also Published As
| Publication number | Publication date |
|---|---|
| EP0102246A1 (de) | 1984-03-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA1207968A (en) | Containment and densification of particulate material | |
| CA1270073A (en) | Encapsulation of waste material | |
| US4172807A (en) | Method for anchoring radioactive substances in a body resistant to leaching by water | |
| GB1588350A (en) | Method of anchoring radioactive waste from nuclear fuel in a body resistant to leaching by water | |
| EP0044692B1 (de) | Vorrichtungen zum Einschliessen von Abfällen | |
| EP2856472B1 (de) | Verfahren zur konsolidierung von radioaktiven materialien durch isostatisches heisspressen | |
| US4726916A (en) | Method for embedding and storing dangerous materials, such as radioactive materials in a monolithic container | |
| EP0102246B1 (de) | Verpackung und Verdichtung eines teilchenförmigen Materials | |
| GB1590108A (en) | Method of treating radioactive waste | |
| US4280921A (en) | Immobilization of waste material | |
| EP0230732B1 (de) | Erzeugung von Keramiken | |
| KR20140132267A (ko) | 분말야금기술을 이용한 방사성 폐기물의 감용처리 방법 | |
| Rusin et al. | Multibarrier waste forms. Part I. Development | |
| RU2176830C2 (ru) | Способ переработки твердых высокоактивных отходов | |
| Larker | Hot isostatic pressing for the consolidation and containment of radioactive waste | |
| CA1186818A (en) | Arrangements for containing waste material | |
| Hoenig et al. | Densification studies of Synroc-D for high-level defense waste | |
| EP0296855A2 (de) | Lagermethode für radioaktives Abfallmaterial | |
| JPS6412360B2 (de) | ||
| EP0209339A2 (de) | Induktionsheizgerät und Verfahren | |
| EP0230740A2 (de) | Anordnungen und Verfahren zur Heizung eines Behälters oder zum Sintern | |
| Berreth et al. | Post treatment of high-level nuclear fuel wastes | |
| EP0228816B1 (de) | Vorrichtungen für Vibrationsbehandlung | |
| EP0211533A1 (de) | Anpressschuh für heiss gepresste Rückstände in Behältern | |
| Campbell et al. | Incorporation of high-level wastes in SYNROC: Results from recent process engineering studies at Lawrence Livermore National Laboratory |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19840803 |
|
| RAP1 | Party data changed (applicant data changed or rights of an application transferred) |
Owner name: THE AUSTRALIAN NATIONAL UNIVERSITY Owner name: AUSTRALIAN ATOMIC ENERGY COMMISSION |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19871104 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED. Effective date: 19871104 Ref country code: AT Effective date: 19871104 |
|
| REF | Corresponds to: |
Ref document number: 30649 Country of ref document: AT Date of ref document: 19871115 Kind code of ref document: T |
|
| ET | Fr: translation filed | ||
| REF | Corresponds to: |
Ref document number: 3374358 Country of ref document: DE Date of ref document: 19871210 |
|
| NLV1 | Nl: lapsed or annulled due to failure to fulfill the requirements of art. 29p and 29m of the patents act | ||
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19880831 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| EAL | Se: european patent in force in sweden |
Ref document number: 83304974.5 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 20010807 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20010810 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20010820 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20010930 Year of fee payment: 19 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: BE Payment date: 20011016 Year of fee payment: 19 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: IF02 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20020828 Year of fee payment: 20 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020831 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020831 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020831 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20020831 |
|
| BERE | Be: lapsed |
Owner name: THE *AUSTRALIAN NATIONAL UNIVERSITY Effective date: 20020831 Owner name: *AUSTRALIAN ATOMIC ENERGY COMMISSION Effective date: 20020831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20030301 |
|
| EUG | Se: european patent has lapsed | ||
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20030430 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20030829 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 |