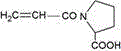CN109970904B - Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof - Google Patents
Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof Download PDFInfo
- Publication number
- CN109970904B CN109970904B CN201910268969.XA CN201910268969A CN109970904B CN 109970904 B CN109970904 B CN 109970904B CN 201910268969 A CN201910268969 A CN 201910268969A CN 109970904 B CN109970904 B CN 109970904B
- Authority
- CN
- China
- Prior art keywords
- sodium polyacrylate
- amino acid
- high blood
- acrylamide
- acrylic acid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/04—Acids; Metal salts or ammonium salts thereof
- C08F220/06—Acrylic acid; Methacrylic acid; Metal salts or ammonium salts thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/10—Esters
- C08F220/26—Esters containing oxygen in addition to the carboxy oxygen
- C08F220/28—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety
- C08F220/285—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety
- C08F220/286—Esters containing oxygen in addition to the carboxy oxygen containing no aromatic rings in the alcohol moiety and containing a polyether chain in the alcohol moiety and containing polyethylene oxide in the alcohol moiety, e.g. methoxy polyethylene glycol (meth)acrylate
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
- C08F220/60—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing nitrogen in addition to the carbonamido nitrogen
- C08F220/603—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide containing nitrogen in addition to the carbonamido nitrogen and containing oxygen in addition to the carbonamido oxygen and nitrogen
Abstract
The invention relates to the technical field of high-absorptivity resin, in particular to sodium polyacrylate high-absorptivity resin with high blood absorption performance and a preparation method thereof. The sodium polyacrylate high-absorptivity resin with high blood absorption performance comprises sodium polyacrylate high-absorptivity resin and functional monomer N-amino acid base acrylic amide. The functional monomer introduced by the sodium polyacrylate high-absorptivity resin with high blood absorption performance has a natural amino acid structure, has excellent compatibility with protein, can effectively solve the problem of protein adsorption, has a carboxyl group, is similar to an acrylic acid structure on one hand, has good compatibility, can perform a neutralization reaction like acrylic acid on the other hand, has excellent water solubility, and hardly influences the water absorption and saline absorption of the final resin.
Description
Technical Field
The invention relates to the technical field of high-absorptivity resin, in particular to sodium polyacrylate high-absorptivity resin with high blood absorption performance and a preparation method thereof.
Background
With the improvement of living standard of people, the performance requirements of blood absorbing materials adopted in the fields of female sanitary towels, pet pads, operation blood absorption and the like are higher and higher, and the blood absorbing materials have extremely strong blood absorbing performance so as to meet the requirements on ultrathin performance, lateral leakage prevention, reverse osmosis prevention and the like. The high molecular super absorbent resin as the super absorbent material is a main material, but because the material focuses on the water absorption performance in structural design, the composition of human blood and animal blood is very complex, besides most of water, the high molecular super absorbent resin also contains various organic components such as protein, and the traditional super absorbent resin cannot realize the super absorption performance on blood components.
Because more than 90% of water in blood, 6-9% of protein and the rest about 2% of electrolyte and small molecular organic substances, namely the protein is the most main macromolecular substance in blood, and the rest are small molecular substances, the general high-absorptivity resin has strong absorption capacity for small molecular substances such as water, electrolyte and the like, and because the protein is a macromolecular substance, the effective volume is large, and the protein is difficult to enter pores of the high-absorptivity resin, the problem of protein adsorption must be solved to improve the blood absorption performance.
At present, in order to improve the blood absorption performance of a sodium polyacrylate super absorbent resin, people generally aim to change the pore structure of the resin through a surface modification technology on the basis of the sodium polyacrylate super absorbent resin with an original formula so as to improve the blood absorption performance, for example, a multistage surface modification technology is adopted in a Chinese patent ZL201710757755.X, a gradual change type hierarchical structure is formed on the surface of the sodium polyacrylate super absorbent resin, so that the liquid absorption speed and the blood absorption capacity of the water absorbent resin are improved, a polyvalent metal salt is adopted for surface modification in a U.S. Pat. No. 6,20170144130, and the high blood absorption resin is prepared in a manner of copolymerizing polyethylene glycol monoacrylate and acrylic acid. However, since the protein in blood is a macromolecule and its volume is very large, it is difficult to enter the pore of super absorbent resin like water, electrolyte and other small molecules, and the blood absorption performance can not be substantially improved.
Disclosure of Invention
The invention provides a sodium polyacrylate high-absorptivity resin with high blood absorption performance and a preparation method thereof, which solve the problems in the prior art.
The technical scheme adopted by the invention for solving the technical problems is as follows:
a high-absorptivity resin of sodium polyacrylate with high blood-sucking power is prepared from high-absorptivity sodium polyacrylate and N-amino acid-base acrylic amide.
The N-amino acid-based acrylic acid amide accounts for 0.5-5%, preferably 0.7-4.9% of the weight of the sodium polyacrylate high-absorptivity resin.
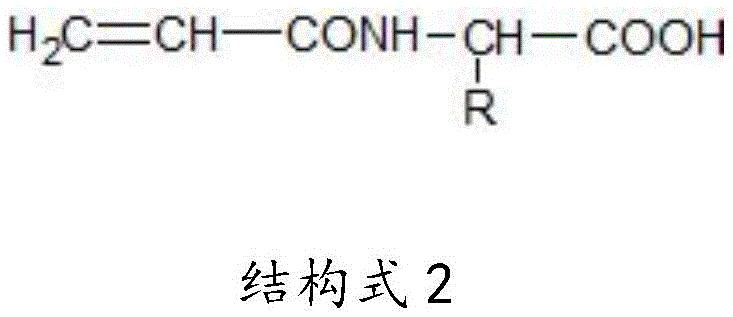
The N-amino acid based acrylic amide is amide obtained by reacting carboxyl of acrylic acid with alpha amino of natural amino acid, and has the following structure:
Acrylamide with proline as structural formula 1;
the structural formula 2 is acrylamide of 19 other natural amino acids.
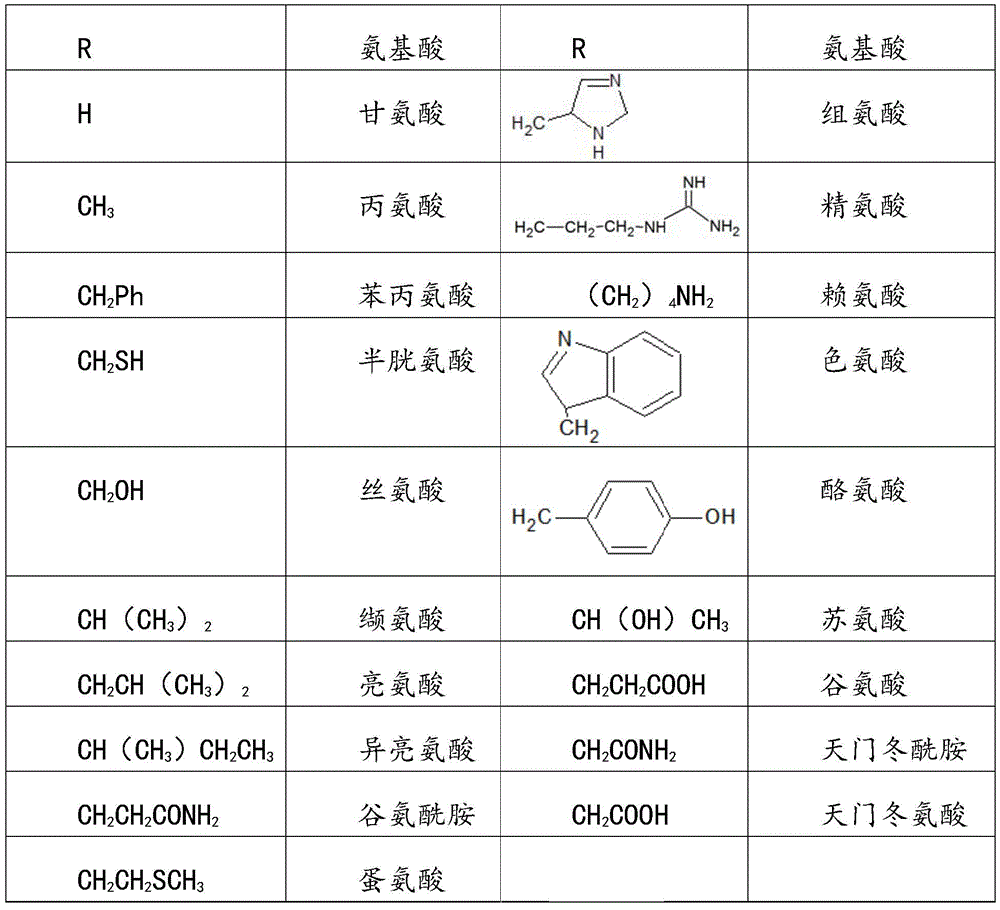
In the structural formula 2, R group is glycine, alanine, phenylalanine, cysteine, serine, valine, leucine, isoleucine, glutamine, methionine, histidine, arginine, lysine, tryptophan, tyrosine, threonine, glutamic acid, asparagine and aspartic acid.
The R groups in formula 2 and the natural amino acids represented by them are as follows:
wherein the R groups and the natural amino acids represented by them are as follows in Table 1:
table 1.
Preferably, the sodium polyacrylate high-absorptivity resin with high blood absorption performance comprises the following raw materials in parts by weight:
300-400 parts of acrylic acid, 10-50 parts of N-amino acid acrylic amide, 5-10 parts of a cross-linking agent, 500-700 parts of deionized water, 180-240 parts of sodium hydroxide and 26-39 parts of an initiator.
The cross-linking agent is one or a mixture of diacetone acrylamide and polyethylene glycol diacrylate; the initiator is prepared from 4-6 parts of oxidant, 2-3 parts of reducing agent and 20-30 parts of deionized water.
The oxidant is one of ammonium persulfate, potassium persulfate, hydrogen peroxide and tert-butyl hydroperoxide; the reducing agent is one of sodium bisulfite, sodium sulfite and ascorbic acid.
The neutralization degree of the acrylic acid and the N-amino acid-based acrylic acid amide is 75-85%.
The preparation method of the sodium polyacrylate high-absorptivity resin with high blood absorption performance comprises the following operation steps:
(1) neutralization reaction
Carrying out neutralization reaction on acrylic acid, N-amino acid based acrylic acid amide and a sodium hydroxide solution to obtain a neutralized solution;
(2) polymerisation reaction
Stirring and mixing the neutralized liquid obtained in the step (1) with a cross-linking agent and an initiator to obtain polymer soft colloid;
(3) drying and pulverizing
And (3) drying and crushing the polymer soft colloid in the step (2).
The N-amino acid based acrylic acid amide in the step (1) is amide obtained by reacting carboxyl of acrylic acid with alpha amino of natural amino acid; the cross-linking agent in the step (2) is one or a mixture of diacetone acrylamide and polyethylene glycol diacrylate.
The polymerization reaction condition of the step (2) is polymerization reaction for 10-20min at the temperature of 20-60 ℃.
Preferably, the preparation method of the sodium polyacrylate high-absorptivity resin with high blood absorption performance according to the parts by weight comprises the following operation steps:
(1) and (3) neutralization reaction:
firstly, preparing a sodium hydroxide aqueous solution, adding the rest deionized water and acrylic acid into a neutralization kettle, dissolving N-amino acid base acrylic acid amide in the neutralization kettle, and uniformly stirring; dropwise adding the sodium hydroxide aqueous solution into a neutralization kettle, controlling the reaction temperature to be lower than 50 ℃, stirring for 20-30min after dropwise adding, and naturally cooling;
(2) polymerisation reaction
Adding the obtained neutralized solution into a polymerization reaction kettle, adding the aqueous solution of a cross-linking agent and an initiator under stirring, and carrying out polymerization reaction for 10-20min under stirring at the temperature of 20-60 ℃ to obtain a polymer soft colloid;
(3) drying and pulverizing
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
The invention has the beneficial effects that:
the invention adds functional monomer containing natural amino acid structure into the polymerized monomer, because the important organic matter in blood is protein, the most fundamental problem of solving the blood-sucking property of the blood-sucking high molecular resin is that the polymer resin has strong absorption property to the water and electrolyte in blood and strong adsorption property to the protein in the blood. And proteins are polymers of various natural amino acids. The functional monomer introduced in the invention has a natural amino acid structure, has excellent compatibility with protein, can effectively solve the problem of protein adsorption, and simultaneously has a carboxyl group, has a structure similar to that of acrylic acid, has good compatibility, can perform neutralization reaction like acrylic acid, and has excellent water solubility, so that the introduction of the monomer hardly influences the water absorption and saline water absorption of the final resin.
Detailed Description
In order to clearly illustrate the technical features of the present invention, the present invention is explained in detail by the following embodiments.
Example 1
Weighing 180kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; adding 360kg of acrylic acid, 20kg of N-glycine-based acrylamide and 100kg of deionized water into a neutralization kettle, stirring and dissolving uniformly, then dropwise adding the prepared sodium hydroxide aqueous solution, controlling the temperature of the neutralization kettle to be lower than 50 ℃, and after dropwise adding, stirring and reacting for 20-30 minutes.
Weighing 4.4kg of ammonium persulfate, 2.2kg of sodium sulfite and 6kg of diacetone acrylamide, dissolving in 25kg of deionized water, adding into a polymerization kettle, adding the obtained neutralized solution of acrylic acid and functional monomer N-glycine-based acrylamide, and carrying out polymerization reaction for 15 minutes at 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 1
The same procedure as in example 1 was repeated except that the N-glycinylacrylamide in example 1 was replaced with the same amount of acrylic acid.
Example 2
Weighing 200kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; 380kg of acrylic acid, 18kg of N-glutamyl acrylamide and 80kg of deionized water are added into a neutralization kettle, after uniform stirring and dissolution, the prepared sodium hydroxide aqueous solution is dripped, the temperature of the neutralization kettle is controlled to be lower than 50 ℃, and after dripping is finished, stirring and reacting for 20-30 min.
Weighing 4.8kg of ammonium persulfate, 2.4kg of sodium sulfite and 6.2kg of diacetone acrylamide, dissolving in 28kg of deionized water, adding into a polymerization kettle, adding the obtained neutralization solution of acrylic acid and functional monomer N-glutamic acrylamide, and carrying out polymerization reaction for 15 minutes at 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 2
The same procedure as in example 2 was repeated except that the N-glutamyl acrylamide in example 2 was changed to the same amount of acrylic acid.
Example 3
Weighing 180kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; adding 350kg of acrylic acid, 15kg of N-lysine acrylamide and 100kg of deionized water into a neutralization kettle, stirring and dissolving uniformly, then dropwise adding the prepared sodium hydroxide aqueous solution, controlling the temperature of the neutralization kettle to be lower than 50 ℃, and after dropwise adding, stirring and reacting for 20-30 min.
Weighing 4.5kg of ammonium persulfate, 2.3kg of sodium sulfite and 6kg of polyethylene glycol diacrylate, dissolving in 25kg of deionized water, adding into a polymerization kettle, adding the obtained acrylic acid and the neutralization solution of the functional monomer N-lysine acrylamide, and carrying out polymerization reaction for 15min at the temperature of 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 3
The same procedure as in example 3 was repeated except that the N-lysine based acrylamide in example 3 was replaced with the same amount of acrylic acid.
Example 4
Weighing 200kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; 370kg of acrylic acid, 22kg of N-alanyl acrylamide and 80kg of deionized water are added into a neutralization kettle, after uniform stirring and dissolution, the prepared sodium hydroxide aqueous solution is dripped, the temperature of the neutralization kettle is controlled to be lower than 50 ℃, and after dripping is finished, stirring and reacting for 20-30 min.
4.8kg of potassium persulfate, 2.2kg of sodium bisulfite and 6kg of diacetone acrylamide are weighed and dissolved in 28kg of deionized water, added into a polymerization kettle, and the obtained neutralized solution of acrylic acid and functional monomer N-alanyl acrylamide thereof is added, and the polymerization reaction is carried out for 15 minutes at the temperature of 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 4
The same procedure as in example 4 was repeated except that the N-alanylacrylamide in example 4 was replaced with the same amount of acrylic acid.
Example 5
Weighing 200kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; 370kg of acrylic acid, 16kg of N-serine acrylamide and 90kg of deionized water are added into a neutralization kettle, after uniform stirring and dissolution, the prepared sodium hydroxide aqueous solution is dripped, the temperature of the neutralization kettle is controlled to be lower than 50 ℃, and after dripping is finished, stirring and reacting for 20-30 min.
Weighing 4.8kg of ammonium persulfate, 2.4kg of ascorbic acid and 6.2kg of polyethylene glycol diacrylate, dissolving in 30kg of deionized water, adding into a polymerization kettle, adding the obtained neutralized solution of acrylic acid and functional monomers thereof, and carrying out polymerization reaction for 15min at the temperature of 20-60 ℃ to obtain the super absorbent resin soft colloid.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 5
The same procedure as in example 5 was repeated except that the N-serine acrylamide in example 5 was replaced with the same amount of acrylic acid.
Example 6
Weighing 190kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; 380kg of acrylic acid, 20kg of N-methionine acrylamide and 90kg of deionized water are added into a neutralization kettle, after uniform stirring and dissolution, the prepared sodium hydroxide aqueous solution is dripped, the temperature of the neutralization kettle is controlled to be lower than 50 ℃, and after dripping is finished, stirring and reacting for 20-30 min.
Weighing 4.6kg of ammonium persulfate, 2.3kg of sodium sulfite and 6.2kg of diacetone acrylamide, dissolving in 28kg of deionized water, adding into a polymerization kettle, adding the obtained neutralized solution of acrylic acid and functional monomers thereof, and carrying out polymerization reaction for 15min at the temperature of 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 6
The same procedure as in example 6 was repeated except that the N-methionoacrylamide in example 6 was replaced with the same amount of acrylic acid.
Example 7
Weighing 200kg of sodium hydroxide, preparing into a 30% aqueous solution, and standing for cooling; 380kg of acrylic acid, 15kg of N-glutamine acrylamide and 80kg of deionized water are added into a neutralization kettle, after uniform stirring and dissolution, the prepared sodium hydroxide aqueous solution is dropwise added, the temperature of the neutralization kettle is controlled to be lower than 50 ℃, and after the dropwise addition is finished, the stirring reaction is carried out for 20-30 min.
Weighing 4.8kg of ammonium persulfate, 2.4kg of sodium sulfite and 6.2kg of diacetone acrylamide, dissolving in 28kg of deionized water, adding into a polymerization kettle, adding the obtained neutralized solution of acrylic acid and functional monomers thereof, and carrying out polymerization reaction for 15min at the temperature of 20-60 ℃ to obtain the soft colloid of the super absorbent resin.
Drying the obtained soft colloid at 100-180 deg.C for 50-120min, and pulverizing into granular material.
Comparative example 7
The same procedure as in example 7 was repeated except that the N-glutamylaminoacrylamide in example 7 was replaced with the same amount of acrylic acid.
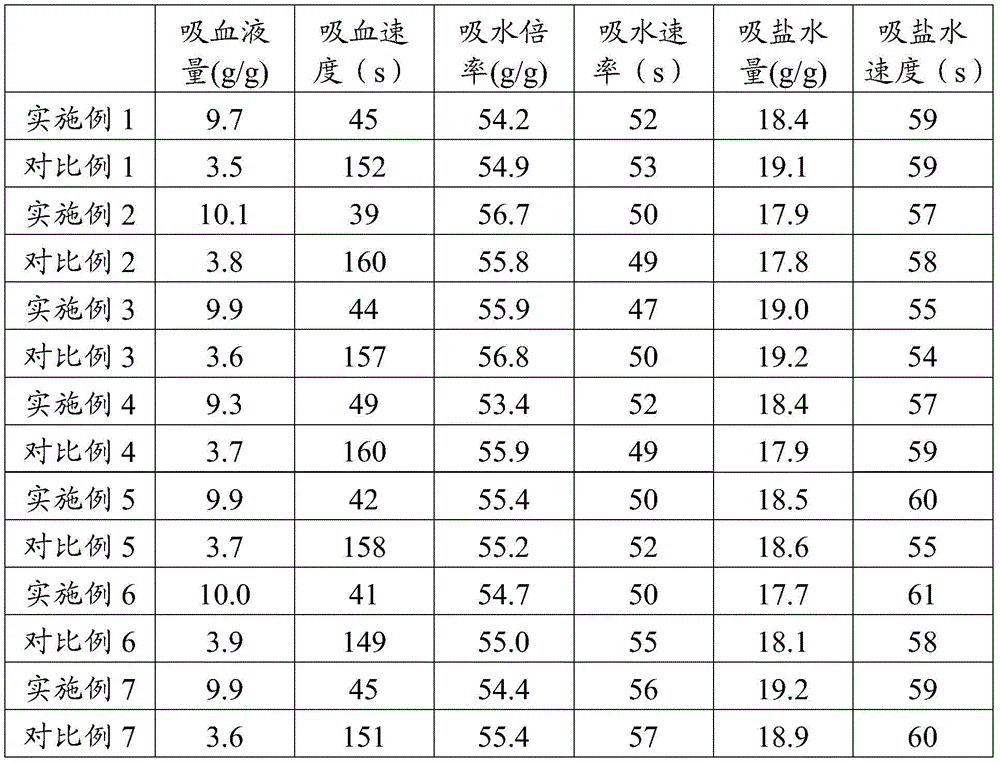
TABLE 2 Properties of absorbent resins obtained in examples and comparative examples
The test of the blood sucking amount and the blood sucking speed is carried out according to the method of the national standard GB/T22875-2008; testing the water absorption multiplying power and the water absorption rate according to a method of national standard GB 22905-2008; the saline uptake and rate of saline uptake were performed according to the method of GB22905-2008 except that water was replaced with 0.1% normal saline.
As is apparent from the data shown in Table 2, the outstanding advantage of the present invention is that it has excellent blood-sucking performance, and the blood-sucking amount and the blood-sucking rate are significantly higher than those of the comparative example, while the water absorption amount, the saline absorption amount and the absorption rate are comparable to those of the comparative example.
The above-described embodiments should not be construed as limiting the scope of the invention, and any alternative modifications or alterations to the embodiments of the present invention will be apparent to those skilled in the art.
The present invention is not described in detail, but is known to those skilled in the art.
Claims (10)
1. A high-absorptivity resin of sodium polyacrylate with high blood-sucking power is prepared from sodium polyacrylate and water
A sodium acrylate structural unit and a structural unit derived from a functional monomer, an N-amino acid based acrylamide;
the N-amino acid based acrylic amide is amide obtained by reacting carboxyl of acrylic acid with alpha amino of natural amino acid, and has the following structure:
Structural formula 1 structural formula 2
Acrylamide with proline as structural formula 1;
the structural formula 2 is acrylamide of 19 other natural amino acids.
2. The sodium polyacrylate superabsorbent resin with high blood absorption performance of claim 1, wherein the N-amino acid based acrylamide accounts for 0.5 to 5 percent of the weight of the sodium polyacrylate superabsorbent resin.
3. The sodium polyacrylate superabsorbent resin with high blood absorption performance of claim 2, wherein the N-amino acid based acrylamide accounts for 0.7 to 4.9 percent of the weight of the sodium polyacrylate superabsorbent resin.
4. The sodium polyacrylate high-absorptivity resin with high blood absorption performance as claimed in claim 1, which is characterized by comprising the following raw materials in parts by weight:
300-400 parts of acrylic acid, 10-50 parts of N-amino acid acrylic amide, 5-10 parts of a cross-linking agent, 500-700 parts of deionized water, 180-240 parts of sodium hydroxide and 26-39 parts of an initiator.
5. The sodium polyacrylate superabsorbent resin with high blood absorption performance of claim 4, wherein the cross-linking agent is one of diacetone acrylamide, polyethylene glycol diacrylate or a mixture thereof; the initiator is prepared from 4-6 parts of oxidant, 2-3 parts of reducing agent and 20-30 parts of deionized water.
6. The sodium polyacrylate superabsorbent resin with high blood absorption performance of claim 5, wherein the oxidant is one of ammonium persulfate, potassium persulfate, hydrogen peroxide and tert-butyl hydrogen peroxide; the reducing agent is one of sodium bisulfite, sodium sulfite and ascorbic acid.
7. The sodium polyacrylate superabsorbent resin with high blood absorption capacity of claim 4, wherein the neutralization degree of acrylic acid and N-amino acid based acrylamide is 75% to 85%.
8. A preparation method of a sodium polyacrylate high-absorptivity resin with high blood-sucking performance is characterized in that the sodium polyacrylate high-absorptivity resin with high blood-sucking performance comprises a sodium acrylate structural unit and a structural unit derived from a functional monomer N-amino acid base acrylic amide;
the N-amino acid based acrylic amide is amide obtained by reacting carboxyl of acrylic acid with alpha amino of natural amino acid, and has the following structure:
Structural formula 1 structural formula 2
Acrylamide with proline as structural formula 1;
the structural formula 2 is acrylamide of 19 other natural amino acids;
the preparation method comprises the following operation steps:
(1) neutralization reaction
Carrying out neutralization reaction on acrylic acid, N-amino acid based acrylic acid amide and a sodium hydroxide solution to obtain a neutralized solution;
(2) polymerisation reaction
Stirring and mixing the neutralization solution obtained in the step (1) with a cross-linking agent and an initiator to obtain a polymer soft colloid;
(3) drying and pulverizing
And (3) drying and crushing the polymer soft colloid in the step (2).
9. The method for preparing sodium polyacrylate superabsorbent resin having high blood absorption according to claim 8, wherein the N-amino acid based acrylic acid amide of step (1) is an amide obtained by reacting a carboxyl group of acrylic acid with an alpha amino group of a natural amino acid; the cross-linking agent in the step (2) is one or a mixture of diacetone acrylamide and polyethylene glycol diacrylate.
10. The method for preparing sodium polyacrylate superabsorbent resin having high blood absorption according to claim 8, wherein the polymerization reaction condition in the step (2) is polymerization at 20 to 60 ℃ for 10 to 20 min.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910268969.XA CN109970904B (en) | 2019-04-03 | 2019-04-03 | Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910268969.XA CN109970904B (en) | 2019-04-03 | 2019-04-03 | Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN109970904A CN109970904A (en) | 2019-07-05 |
| CN109970904B true CN109970904B (en) | 2021-04-27 |
Family
ID=67082887
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910268969.XA Active CN109970904B (en) | 2019-04-03 | 2019-04-03 | Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN109970904B (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114380936B (en) * | 2021-12-08 | 2023-07-11 | 山东昊月新材料股份有限公司 | High-blood-absorption sodium polyacrylate resin containing polypeptide structure and preparation method thereof |
| CN114522027A (en) * | 2021-12-30 | 2022-05-24 | 福建恒安集团有限公司 | Super-soft high blood absorption type composite core and sanitary towel |
| CN114805662B (en) * | 2022-04-20 | 2023-07-14 | 山东昊月新材料股份有限公司 | High-absorption resin with in-situ antibacterial property and synthesis method thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100336563C (en) * | 2002-10-24 | 2007-09-12 | 大日本油墨化学工业株式会社 | Absorbent material and absorbent article |
| JP2011213718A (en) * | 2010-03-17 | 2011-10-27 | Nippon Zoki Pharmaceut Co Ltd | Manufacturing method of medicine containing amino acid derivative and derivative of the same |
| CN102659974A (en) * | 2012-05-21 | 2012-09-12 | 天津工业大学 | pH response type gel modified by L-amino acid |
-
2019
- 2019-04-03 CN CN201910268969.XA patent/CN109970904B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100336563C (en) * | 2002-10-24 | 2007-09-12 | 大日本油墨化学工业株式会社 | Absorbent material and absorbent article |
| JP2011213718A (en) * | 2010-03-17 | 2011-10-27 | Nippon Zoki Pharmaceut Co Ltd | Manufacturing method of medicine containing amino acid derivative and derivative of the same |
| CN102659974A (en) * | 2012-05-21 | 2012-09-12 | 天津工业大学 | pH response type gel modified by L-amino acid |
Non-Patent Citations (1)
| Title |
|---|
| Poly(acrylic acid)–cysteine for oral vitamin B12 delivery;Federica Sarti等;《Analytical Biochemistry》;20110910;第13-19页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109970904A (en) | 2019-07-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109970904B (en) | Sodium polyacrylate high-absorptivity resin with high blood absorption performance and preparation method thereof | |
| KR910008293B1 (en) | High absorptive resin | |
| CN102153693B (en) | Salt-tolerant acrylic absorbent resin and method for preparing same | |
| JP2003206305A (en) | Manufacturing method of water-absorbable resin | |
| CA1262004A (en) | Process for producing highly absorptive resin | |
| CN104804130A (en) | Hydrogel applicable to seawater desalination and preparation method thereof | |
| CN104693362A (en) | Method for preparing cellulose graft copolymer super absorbent resin | |
| CN103224588B (en) | A kind of flocculation agent | |
| CN1296401C (en) | Method for preparing grouting material of modified polyacrylamide | |
| JP3259143B2 (en) | Method for producing water absorbent resin | |
| CN109535307B (en) | Reversed phase suspension polymerization preparation process of water-absorbent resin | |
| CN109608569A (en) | A method of preparing ethylene-acrylic acid copolymer | |
| CN108610459A (en) | A kind of preparation method of quick absorption-type super absorbent resin | |
| CN106492659A (en) | A kind of temperature and pH double-bang firecrackers answer PVDF semi-interpenetrating network polymer films and preparation method thereof | |
| CN104744623B (en) | A kind of concrete workability conditioning agent and preparation method thereof | |
| CN114380936B (en) | High-blood-absorption sodium polyacrylate resin containing polypeptide structure and preparation method thereof | |
| CN103374104B (en) | Preparation method of super absorbent polymer (SAP) | |
| JP4918952B2 (en) | Water-absorbing material | |
| Qu et al. | Preparation of sodium carboxymethylcellulose/poly (methyl acrylate) IPN hydrogels and their application for adsorption | |
| JPH01292003A (en) | Purification of water-absorbable resin | |
| JPS58154710A (en) | Production of amphoteric, highly water-absorptive resin | |
| CN106589441A (en) | Preparation method for water absorbent with high strength and high water absorption rate | |
| JPS578210A (en) | Production of water-absorbing resin | |
| CN109851965A (en) | A kind of nano combined high absorbency material and preparation method thereof | |
| CN107754006B (en) | Application of temperature-responsive supramolecular copolymer hydrogel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |