US9526798B2 - Pyrrolobenzodiazepines and targeted conjugates - Google Patents
Pyrrolobenzodiazepines and targeted conjugates Download PDFInfo
- Publication number
- US9526798B2 US9526798B2 US14/351,168 US201214351168A US9526798B2 US 9526798 B2 US9526798 B2 US 9526798B2 US 201214351168 A US201214351168 A US 201214351168A US 9526798 B2 US9526798 B2 US 9526798B2
- Authority
- US
- United States
- Prior art keywords
- group
- attachment
- point
- unit
- groups
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 [10*]N1C/C=C(OCCCC)\C(OC)=C/CC(=O)N2C=C(C3=CC=C(N)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(OC)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(OCCCN(C)C)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCCC)=C2)C(=O)N2C=C(C3=CC=CS3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(OC)C(OCC)=C2)C(=O)N2/C=C(/C3=CC=C(OCCCNC)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(OC)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(N)C=C3)C[C@@]2([H])C1[11*] Chemical compound [10*]N1C/C=C(OCCCC)\C(OC)=C/CC(=O)N2C=C(C3=CC=C(N)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(OC)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(OCCCN(C)C)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(C)C(OCCCC)=C2)C(=O)N2C=C(C3=CC=CS3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(OC)C(OCC)=C2)C(=O)N2/C=C(/C3=CC=C(OCCCNC)C=C3)C[C@@]2([H])C1[11*].[10*]N1C2=C(C=C(OC)C(OCCC)=C2)C(=O)N2C=C(C3=CC=C(N)C=C3)C[C@@]2([H])C1[11*] 0.000 description 58
- RZXMPPFPUUCRFN-UHFFFAOYSA-N CC1=CC=C(N)C=C1 Chemical compound CC1=CC=C(N)C=C1 RZXMPPFPUUCRFN-UHFFFAOYSA-N 0.000 description 3
- CHLICZRVGGXEOD-UHFFFAOYSA-N COC1=CC=C(C)C=C1 Chemical compound COC1=CC=C(C)C=C1 CHLICZRVGGXEOD-UHFFFAOYSA-N 0.000 description 3
- BAQWAJKPAKTEQJ-UHFFFAOYSA-N CC(=O)CCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O Chemical compound CC(=O)CCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O BAQWAJKPAKTEQJ-UHFFFAOYSA-N 0.000 description 2
- JWMHCBPAAFXCGZ-UHFFFAOYSA-N CCCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O Chemical compound CCCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O JWMHCBPAAFXCGZ-UHFFFAOYSA-N 0.000 description 2
- WBCQROCIEMMGJF-QOXWQENKSA-N C.C.C.C.C.CC1(C)OB(C2=CC=C(O)C=C2)OC1(C)C.COCCOCCOCCOCCBr.COCCOCCOCCOCCOC.COCCOCCOCCOCCOC1=CC=C(B2OC(C)(C)C(C)(C)O2)C=C1.[H][C@@]12C=NC3=CC(OCCCCCOC4=C(OC)C=C5C(=O)N6C=C(C7=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C[C@]6([H])C=NC5=C4)=C(OC)C=C3C(=O)N1C=C(C1=CC=C(C)C=C1)C2.[H][C@@]12C=NC3=CC(OCCCCCOC4=C(OC)C=C5C(=O)N6C=C(C7=CC=C(OCCOCCOCCOCCOC)C=C7)C[C@]6([H])C=NC5=C4)=C(OC)C=C3C(=O)N1C=C(C1=CC=C(N)C=C1)C2.[H][C@]12CC(C3=CC=C(N)C=C3)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(OC)C=C4C(=O)N5C=C(C6=CC=C(OCCOCCOCCOCCOC)C=C6)C[C@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(OC)C=C4C(=O)N5C=C(C6=CC=C(N)C=C6)C[C@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C=C1N(COCC[Si](C)(C)C)C2=O Chemical compound C.C.C.C.C.CC1(C)OB(C2=CC=C(O)C=C2)OC1(C)C.COCCOCCOCCOCCBr.COCCOCCOCCOCCOC.COCCOCCOCCOCCOC1=CC=C(B2OC(C)(C)C(C)(C)O2)C=C1.[H][C@@]12C=NC3=CC(OCCCCCOC4=C(OC)C=C5C(=O)N6C=C(C7=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C[C@]6([H])C=NC5=C4)=C(OC)C=C3C(=O)N1C=C(C1=CC=C(C)C=C1)C2.[H][C@@]12C=NC3=CC(OCCCCCOC4=C(OC)C=C5C(=O)N6C=C(C7=CC=C(OCCOCCOCCOCCOC)C=C7)C[C@]6([H])C=NC5=C4)=C(OC)C=C3C(=O)N1C=C(C1=CC=C(N)C=C1)C2.[H][C@]12CC(C3=CC=C(N)C=C3)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(OC)C=C4C(=O)N5C=C(C6=CC=C(OCCOCCOCCOCCOC)C=C6)C[C@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(OC)C=C4C(=O)N5C=C(C6=CC=C(N)C=C6)C[C@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C=C1N(COCC[Si](C)(C)C)C2=O WBCQROCIEMMGJF-QOXWQENKSA-N 0.000 description 1
- JWUOPHPJBBOVMB-UHFFFAOYSA-N CC(=O)CC1=CC=C(N2C(=O)C=CC2=O)C=C1 Chemical compound CC(=O)CC1=CC=C(N2C(=O)C=CC2=O)C=C1 JWUOPHPJBBOVMB-UHFFFAOYSA-N 0.000 description 1
- LFCBWFJNOGWLFB-UHFFFAOYSA-N CC(=O)CC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CC(=O)CC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 LFCBWFJNOGWLFB-UHFFFAOYSA-N 0.000 description 1
- XTSLYHFFUACHDH-UHFFFAOYSA-N CC(=O)CCOCCNC(=O)CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 Chemical compound CC(=O)CCOCCNC(=O)CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 XTSLYHFFUACHDH-UHFFFAOYSA-N 0.000 description 1
- DEPVUHNJOZEXRA-UHFFFAOYSA-N CC(=O)CCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CC(=O)CCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 DEPVUHNJOZEXRA-UHFFFAOYSA-N 0.000 description 1
- DDXFQOZQGRXYAD-UHFFFAOYSA-N CC(=O)CCOCCNC(=O)CCN1C(=O)C=CC1=O Chemical compound CC(=O)CCOCCNC(=O)CCN1C(=O)C=CC1=O DDXFQOZQGRXYAD-UHFFFAOYSA-N 0.000 description 1
- ZSGAKFQBDCKAQV-UHFFFAOYSA-N CC(=O)CN1C(=O)C=CC1=O Chemical compound CC(=O)CN1C(=O)C=CC1=O ZSGAKFQBDCKAQV-UHFFFAOYSA-N 0.000 description 1
- FHVGOGGHHZGKOT-UHFFFAOYSA-N CC(=O)CN1C(=O)CC(C(C)C)C1=O Chemical compound CC(=O)CN1C(=O)CC(C(C)C)C1=O FHVGOGGHHZGKOT-UHFFFAOYSA-N 0.000 description 1
- IWKGMMOJPFLURZ-QRWLVFNGSA-N CC(=O)OCC1=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(C)C)C(C)C)C=C1 Chemical compound CC(=O)OCC1=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(C)C)C(C)C)C=C1 IWKGMMOJPFLURZ-QRWLVFNGSA-N 0.000 description 1
- MAAHDDYKDAWRRC-PXJZQJOASA-N CC(=O)OCC1=CC=C(NC(=O)[C@H](CCCCNC(=O)OC(C)(C)C)CC(=O)[C@H](CC2=CC=CC=C2)NC(C)C)C=C1 Chemical compound CC(=O)OCC1=CC=C(NC(=O)[C@H](CCCCNC(=O)OC(C)(C)C)CC(=O)[C@H](CC2=CC=CC=C2)NC(C)C)C=C1 MAAHDDYKDAWRRC-PXJZQJOASA-N 0.000 description 1
- KHHZIGVTJRTCMP-AMUDCUPNSA-N CC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN1C(=O)CC(C(C)C)C1=O)C(C)C Chemical compound CC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN1C(=O)CC(C(C)C)C1=O)C(C)C KHHZIGVTJRTCMP-AMUDCUPNSA-N 0.000 description 1
- PRCUDJXDCWUEDT-UHFFFAOYSA-O CC(C)=C(C)O.CC(C)=C(C)[O-].[H+].[H+].[H]C(C)(C)C(C)=O Chemical compound CC(C)=C(C)O.CC(C)=C(C)[O-].[H+].[H+].[H]C(C)(C)C(C)=O PRCUDJXDCWUEDT-UHFFFAOYSA-O 0.000 description 1
- LUOKEEIOPMUJLA-UHFFFAOYSA-N CC(C)C1CC(=O)N(C)C1=O Chemical compound CC(C)C1CC(=O)N(C)C1=O LUOKEEIOPMUJLA-UHFFFAOYSA-N 0.000 description 1
- WTEXJEVWSRVCQA-MOPGFXCFSA-N CC(C)N[C@@H](CC1=CC=CC=C1)C(=O)C[C@@H](CCCCN)C(=O)N(C)C Chemical compound CC(C)N[C@@H](CC1=CC=CC=C1)C(=O)C[C@@H](CCCCN)C(=O)N(C)C WTEXJEVWSRVCQA-MOPGFXCFSA-N 0.000 description 1
- GEGDCUSIMMLNQZ-YPMHNXCESA-N CC(C)N[C@H](C(=O)C[C@@H](C)C(=O)N(C)C)C(C)C Chemical compound CC(C)N[C@H](C(=O)C[C@@H](C)C(=O)N(C)C)C(C)C GEGDCUSIMMLNQZ-YPMHNXCESA-N 0.000 description 1
- JFOXQDLSTDJWQZ-UHFFFAOYSA-N CC(C)SC1CC(=O)N(C)C1=O Chemical compound CC(C)SC1CC(=O)N(C)C1=O JFOXQDLSTDJWQZ-UHFFFAOYSA-N 0.000 description 1
- KDCGJUBEDPXANZ-SKURDQRSSA-N CC(C)[C@H](CC(=O)CCCCCN1C(=O)C=CC1=O)C(=O)N[C@@H](CCCNC(N)=O)C(=O)CC1=CC=C(COC(=O)OC2=CC=C([N+](=O)[O-])C=C2)C=C1.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4C=C(C6=CC=C(CCC(=O)OCC7=CC=C(CC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](CC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C=C6)C[C@@]4([H])C=N5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 Chemical compound CC(C)[C@H](CC(=O)CCCCCN1C(=O)C=CC1=O)C(=O)N[C@@H](CCCNC(N)=O)C(=O)CC1=CC=C(COC(=O)OC2=CC=C([N+](=O)[O-])C=C2)C=C1.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4C=C(C6=CC=C(CCC(=O)OCC7=CC=C(CC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](CC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C=C6)C[C@@]4([H])C=N5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 KDCGJUBEDPXANZ-SKURDQRSSA-N 0.000 description 1
- HMTSWYPNXFHGEP-UHFFFAOYSA-N CC1=CC=C(CN)C=C1 Chemical compound CC1=CC=C(CN)C=C1 HMTSWYPNXFHGEP-UHFFFAOYSA-N 0.000 description 1
- KWLPQJGMKNLTSR-UHFFFAOYSA-N CC1=CC=C(OCCCN(C)C)C=C1 Chemical compound CC1=CC=C(OCCCN(C)C)C=C1 KWLPQJGMKNLTSR-UHFFFAOYSA-N 0.000 description 1
- JHKXVSGKKYHYJR-UHFFFAOYSA-N CC1=CC=C(OCCCN)C=C1 Chemical compound CC1=CC=C(OCCCN)C=C1 JHKXVSGKKYHYJR-UHFFFAOYSA-N 0.000 description 1
- XQQBUAPQHNYYRS-UHFFFAOYSA-N CC1=CC=CS1 Chemical compound CC1=CC=CS1 XQQBUAPQHNYYRS-UHFFFAOYSA-N 0.000 description 1
- PMCALBQELZDXNF-UHFFFAOYSA-N CC1=CN(C(C)C)N=N1.CC1=CN=NN1C(C)C Chemical compound CC1=CN(C(C)C)N=N1.CC1=CN=NN1C(C)C PMCALBQELZDXNF-UHFFFAOYSA-N 0.000 description 1
- ONQOJDTXLDAYDR-UHFFFAOYSA-N CC1=NN=NC1 Chemical compound CC1=NN=NC1 ONQOJDTXLDAYDR-UHFFFAOYSA-N 0.000 description 1
- FHVHFKZQDVQILM-UHFFFAOYSA-N CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 Chemical compound CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 FHVHFKZQDVQILM-UHFFFAOYSA-N 0.000 description 1
- BWJGIKVTQUUIDU-UHFFFAOYSA-N CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 BWJGIKVTQUUIDU-UHFFFAOYSA-N 0.000 description 1
- MATQILKGIZOQCB-UHFFFAOYSA-N CCCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CCCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 MATQILKGIZOQCB-UHFFFAOYSA-N 0.000 description 1
- VACJKMWLHOIONG-UHFFFAOYSA-N CCCC1=CC=C(N2C(=O)CC(SC(C)C)C2=O)C=C1 Chemical compound CCCC1=CC=C(N2C(=O)CC(SC(C)C)C2=O)C=C1 VACJKMWLHOIONG-UHFFFAOYSA-N 0.000 description 1
- MZTNVLOCZNFIKM-UHFFFAOYSA-N CCCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CCCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 MZTNVLOCZNFIKM-UHFFFAOYSA-N 0.000 description 1
- DRQFFVSVJKQTNR-UHFFFAOYSA-N CCCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(SC(C)C)C2=O)C=C1 Chemical compound CCCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(SC(C)C)C2=O)C=C1 DRQFFVSVJKQTNR-UHFFFAOYSA-N 0.000 description 1
- WDPMEBXSGXQWJO-UHFFFAOYSA-N CCCCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O Chemical compound CCCCOCCNC(=O)CCN1C(=O)CC(C(C)C)C1=O WDPMEBXSGXQWJO-UHFFFAOYSA-N 0.000 description 1
- YIDYXANUZXRJFH-UHFFFAOYSA-N CCCCOCCNC(=O)CCN1C(=O)CC(SC(C)C)C1=O Chemical compound CCCCOCCNC(=O)CCN1C(=O)CC(SC(C)C)C1=O YIDYXANUZXRJFH-UHFFFAOYSA-N 0.000 description 1
- MTNDQCKAFUJMQV-UHFFFAOYSA-N CCCN1C(=O)CC(SC(C)C)C1=O Chemical compound CCCN1C(=O)CC(SC(C)C)C1=O MTNDQCKAFUJMQV-UHFFFAOYSA-N 0.000 description 1
- YKYONYBAUNKHLG-UHFFFAOYSA-N CCCOC(C)=O Chemical compound CCCOC(C)=O YKYONYBAUNKHLG-UHFFFAOYSA-N 0.000 description 1
- JENTVNSCQYNIQX-UHFFFAOYSA-N CCCOCCNC(=O)CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 Chemical compound CCCOCCNC(=O)CCC1=CC=C(N2C(=O)C=CC2=O)C=C1 JENTVNSCQYNIQX-UHFFFAOYSA-N 0.000 description 1
- WYPPASTYOKMLHF-UHFFFAOYSA-N CCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 Chemical compound CCCOCCNC(=O)CCC1=CC=C(N2C(=O)CC(C(C)C)C2=O)C=C1 WYPPASTYOKMLHF-UHFFFAOYSA-N 0.000 description 1
- ASWGLSDBMYYAJM-UHFFFAOYSA-N CCCOCCNC(=O)CCN1C(=O)C=CC1=O Chemical compound CCCOCCNC(=O)CCN1C(=O)C=CC1=O ASWGLSDBMYYAJM-UHFFFAOYSA-N 0.000 description 1
- HDFGOPSGAURCEO-UHFFFAOYSA-N CCN1C(=O)C=CC1=O Chemical compound CCN1C(=O)C=CC1=O HDFGOPSGAURCEO-UHFFFAOYSA-N 0.000 description 1
- XOBKSJJDNFUZPF-UHFFFAOYSA-N CCOC Chemical compound CCOC XOBKSJJDNFUZPF-UHFFFAOYSA-N 0.000 description 1
- IHMZQQVGQIKENS-UHFFFAOYSA-N CN1C(=O)C2=CC=CC=C2C1=O.CN1C(=O)C=CC1=O.CN1C(=O)CCC1=O Chemical compound CN1C(=O)C2=CC=CC=C2C1=O.CN1C(=O)C=CC1=O.CN1C(=O)CCC1=O IHMZQQVGQIKENS-UHFFFAOYSA-N 0.000 description 1
- SEEYREPSKCQBBF-UHFFFAOYSA-N CN1C(=O)C=CC1=O Chemical compound CN1C(=O)C=CC1=O SEEYREPSKCQBBF-UHFFFAOYSA-N 0.000 description 1
- XRYKNRLGZZNWEE-UHFFFAOYSA-N CN1C(=O)N=NC1=O Chemical compound CN1C(=O)N=NC1=O XRYKNRLGZZNWEE-UHFFFAOYSA-N 0.000 description 1
- CXCMQFLZQKLNQA-UHFFFAOYSA-N CNCCCOC1=CC=C(C)C=C1 Chemical compound CNCCCOC1=CC=C(C)C=C1 CXCMQFLZQKLNQA-UHFFFAOYSA-N 0.000 description 1
- KFGJLGGSRWRFST-RQCNNRJASA-N COC(=O)C1O[C@@H](OC2=CC=C(COC(=O)OC3=CC=C([N+](=O)[O-])C=C3)C=C2NC(=O)CCNC(=O)OCC2C3=C(C=CC=C3)C3=C2C=CC=C3)C(OC(C)=O)[C@@H](C)[C@@H]1C.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2O Chemical compound COC(=O)C1O[C@@H](OC2=CC=C(COC(=O)OC3=CC=C([N+](=O)[O-])C=C3)C=C2NC(=O)CCNC(=O)OCC2C3=C(C=CC=C3)C3=C2C=CC=C3)C(OC(C)=O)[C@@H](C)[C@@H]1C.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2O KFGJLGGSRWRFST-RQCNNRJASA-N 0.000 description 1
- KXKVLQRXCPHEJC-UHFFFAOYSA-N COC(C)=O Chemical compound COC(C)=O KXKVLQRXCPHEJC-UHFFFAOYSA-N 0.000 description 1
- SLDZFENVULXRDD-UHFFFAOYSA-N COCCOCCOCCOCCOC1=CC=C(C)C=C1 Chemical compound COCCOCCOCCOCCOC1=CC=C(C)C=C1 SLDZFENVULXRDD-UHFFFAOYSA-N 0.000 description 1
- FTOVHPYLYYMJFU-UHFFFAOYSA-N C[IH][IH]c(cc(CO)cc1)c1N Chemical compound C[IH][IH]c(cc(CO)cc1)c1N FTOVHPYLYYMJFU-UHFFFAOYSA-N 0.000 description 1
- VLVCDUSVTXIWGW-UHFFFAOYSA-N Nc(cc1)ccc1I Chemical compound Nc(cc1)ccc1I VLVCDUSVTXIWGW-UHFFFAOYSA-N 0.000 description 1
- NMHMQYSITIFCSQ-UHFFFAOYSA-N O=C(CCN(C(C=C1)=O)C1=O)NCCCOCCI Chemical compound O=C(CCN(C(C=C1)=O)C1=O)NCCCOCCI NMHMQYSITIFCSQ-UHFFFAOYSA-N 0.000 description 1
- IZYKWRJGIYKBSF-BRGYPHDKSA-M O=S(=O)=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(C)C=C5)C[C@@]3([H])C([Na])C4)C(C)=C1)CC2S(=O)(=O)O[Na] Chemical compound O=S(=O)=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(C)C=C5)C[C@@]3([H])C([Na])C4)C(C)=C1)CC2S(=O)(=O)O[Na] IZYKWRJGIYKBSF-BRGYPHDKSA-M 0.000 description 1
- NDYJOZAQSPMQHB-UHFFFAOYSA-N [H]N1C(=O)N(C)C(=O)N1C(C)C Chemical compound [H]N1C(=O)N(C)C(=O)N1C(C)C NDYJOZAQSPMQHB-UHFFFAOYSA-N 0.000 description 1
- FJUUBTIXTHLSOQ-SOVOJBPTSA-N [H][C@@]12CC(C)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(C)C=C4C(=O)N5C=C(OS(=O)(=O)C(F)(F)F)C[C@@]5([H])C(C)N(C)C4=C3)C=C1N(C)C2C Chemical compound [H][C@@]12CC(C)=CN1C(=O)C1=CC(OC)=C(OCCCCCOC3=C(C)C=C4C(=O)N5C=C(OS(=O)(=O)C(F)(F)F)C[C@@]5([H])C(C)N(C)C4=C3)C=C1N(C)C2C FJUUBTIXTHLSOQ-SOVOJBPTSA-N 0.000 description 1
- YWUBZVCHJRPPHC-QWLYLMPRSA-N [H][C@@]12CC(C3=CC=C(CN)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O Chemical compound [H][C@@]12CC(C3=CC=C(CN)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O YWUBZVCHJRPPHC-QWLYLMPRSA-N 0.000 description 1
- JFYPJSYTYQKORH-YXSGLDHLSA-N [H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)OCC=C)C(C)C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(CN)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O Chemical compound [H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)OCC=C)C(C)C)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=C(C=C(OCCCCCOC3=C(OC)C=C4C(=O)N5/C=C(/C6=CC=C(CN)C=C6)C[C@@]5([H])C(=O)N(COCC[Si](C)(C)C)C4=C3)C(OC)=C1)N(COCC[Si](C)(C)C)C2=O JFYPJSYTYQKORH-YXSGLDHLSA-N 0.000 description 1
- CDLXDVQRAIYOOY-OAPJLYHOSA-N [H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4C=C(C6=CC=C(CN)C=C6)C[C@@]4([H])C=N5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 Chemical compound [H][C@@]12CC(C3=CC=C(OC)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3C=C(C5=CC=C(CN)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4C=C(C6=CC=C(CN)C=C6)C[C@@]4([H])C=N5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 CDLXDVQRAIYOOY-OAPJLYHOSA-N 0.000 description 1
- UPZACQBDAOPPDC-ANAIXNRLSA-N [H][C@@]12CC(C3=CC=C(OCCCN(C)C)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(N)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(N)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 Chemical compound [H][C@@]12CC(C3=CC=C(OCCCN(C)C)C=C3)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(N)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@@]12CC(OS(=O)(=O)C(F)(F)F)=CN1C(=O)C1=CC(C)=C(OCCCCCOC3=CC4=C(C=C3OC)C(=O)N3/C=C(/C5=CC=C(N)C=C5)C[C@@]3([H])C(=O)N4COCC[Si](C)(C)C)C=C1N(COCC[Si](C)(C)C)C2=O.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 UPZACQBDAOPPDC-ANAIXNRLSA-N 0.000 description 1
- DIYIJQZXBLFKSJ-VXKWHMMOSA-N [H][C@]12C=NC3=C(C=C(C)C(OCCCOC4=CC5=C(C=C4OC)C(=O)N4CC(=C)C[C@@]4([H])C=N5)=C3)C(=O)N1CC(=C)C2 Chemical compound [H][C@]12C=NC3=C(C=C(C)C(OCCCOC4=CC5=C(C=C4OC)C(=O)N4CC(=C)C[C@@]4([H])C=N5)=C3)C(=O)N1CC(=C)C2 DIYIJQZXBLFKSJ-VXKWHMMOSA-N 0.000 description 1
- NMDZFSZULAOTLD-RUWZBFSUSA-N [H][C@]12C=NC3=C(C=C(OC)C(OCCCCCOC4=C(OC)C=C5C(=O)N6/C=C(/C7=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C[C@@]6([H])/C=N\C5=C4)=C3)C(=O)N1C=C(C1=CC=C(OC)C=C1)C2.[H][C@]12C=NC3=C(C=C(OC)C(OCCCCCOC4=C(OC)C=C5C(=O)N6/C=C(/C7=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)OCC=C)C(C)C)C=C7)C[C@@]6([H])/C=N\C5=C4)=C3)C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 Chemical compound [H][C@]12C=NC3=C(C=C(OC)C(OCCCCCOC4=C(OC)C=C5C(=O)N6/C=C(/C7=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)CCCCCN8C(=O)C=CC8=O)C(C)C)C=C7)C[C@@]6([H])/C=N\C5=C4)=C3)C(=O)N1C=C(C1=CC=C(OC)C=C1)C2.[H][C@]12C=NC3=C(C=C(OC)C(OCCCCCOC4=C(OC)C=C5C(=O)N6/C=C(/C7=CC=C(CCC(=O)[C@H](C)NC(=O)[C@@H](CC(=O)OCC=C)C(C)C)C=C7)C[C@@]6([H])/C=N\C5=C4)=C3)C(=O)N1C=C(C1=CC=C(OC)C=C1)C2 NMDZFSZULAOTLD-RUWZBFSUSA-N 0.000 description 1
- WLQFQGMEWVRAPC-HEVIKAOCSA-N [H][C@]12C=NC3=C(C=C(OC)C(OCCCOC4=CC5=C(C=C4C)C(=O)N4/C=C(/C6=CC=C(C)C=C6)C[C@@]4([H])/C=N\5)=C3)C(=O)N1C=C(C1=CC=C(C)C=C1)C2 Chemical compound [H][C@]12C=NC3=C(C=C(OC)C(OCCCOC4=CC5=C(C=C4C)C(=O)N4/C=C(/C6=CC=C(C)C=C6)C[C@@]4([H])/C=N\5)=C3)C(=O)N1C=C(C1=CC=C(C)C=C1)C2 WLQFQGMEWVRAPC-HEVIKAOCSA-N 0.000 description 1
- APCGBLBDFBFPPS-RALGLRHXSA-N [H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(NC(=O)CCCCCN7C(=O)C=CC7=O)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 Chemical compound [H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(NC(=O)CCCCCN7C(=O)C=CC7=O)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 APCGBLBDFBFPPS-RALGLRHXSA-N 0.000 description 1
- LQRUOWKROKFCSX-PWEGVWNHSA-N [H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN7C(=O)C=CC7=O)C(C)C)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 Chemical compound [H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(N)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2.[H][C@]12C=NC3=CC(OCCCCCOC4=CC5=C(C=C4OC)C(=O)N4/C=C(/C6=CC=C(NC(=O)[C@H](C)CC(=O)[C@@H](NC(=O)CCCCCN7C(=O)C=CC7=O)C(C)C)C=C6)C[C@@]4([H])/C=N\5)=C(C)C=C3C(=O)N1C=C(C1=CC=C(OCCCN(C)C)C=C1)C2 LQRUOWKROKFCSX-PWEGVWNHSA-N 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A61K47/48384—
-
- A61K47/481—
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/55—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound the modifying agent being also a pharmacologically or therapeutically active agent, i.e. the entire conjugate being a codrug, i.e. a dimer, oligomer or polymer of pharmacologically or therapeutically active compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6889—Conjugates wherein the antibody being the modifying agent and wherein the linker, binder or spacer confers particular properties to the conjugates, e.g. peptidic enzyme-labile linkers or acid-labile linkers, providing for an acid-labile immuno conjugate wherein the drug may be released from its antibody conjugated part in an acidic, e.g. tumoural or environment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- the present invention relates to pyrrolobenzodiazepines (PBDs), in particular pyrrolobenzodiazepine dimers having a C2-C3 double bond and an aryl group at the C2 position in each monomer unit, and their inclusion in targeted conjugates.
- PBDs pyrrolobenzodiazepines
- pyrrolobenzodiazepine dimers having a C2-C3 double bond and an aryl group at the C2 position in each monomer unit, and their inclusion in targeted conjugates.
- PBDs pyrrolobenzodiazepines
- Family members include abbeymycin (Hochlowski, et al., J. Antibiotics, 40, 145-148 (1987)), chicamycin (Konishi, et al., J. Antibiotics, 37, 200-206 (1984)), DC-81 (Japanese Patent 58-180 487; Thurston, et al., Chem. Brit., 26, 767-772 (1990); Bose, et al., Tetrahedron, 48, 751-758 (1992)), mazethramycin (Kuminoto, et al., J.
- PBDs are of the general structure:
- the PBD dimers are thought to form sequence-selective DNA lesions such as the palindromic 5′-Pu-GATC-Py-3′ interstrand cross-link (Smellie, M., et al, Biochemistry, 42, 8232-8239 (2003); Martin, C., et al., Biochemistry, 44, 4135-4147) which is thought to be mainly responsible for their biological activity.
- sequence-selective DNA lesions such as the palindromic 5′-Pu-GATC-Py-3′ interstrand cross-link (Smellie, M., et al, Biochemistry, 42, 8232-8239 (2003); Martin, C., et al., Biochemistry, 44, 4135-4147) which is thought to be mainly responsible for their biological activity.
- PBD dimmer SG2000 (SJG-136):
- WO 2010/043880 discloses unsymmetrical dimeric PBD compound bearing aryl groups in the C2 position of each monomer, where one of these aryl groups bears a substituent designed to provide an anchor for linking the compound to another moiety.
- the present inventors have developed further specific unsymmetrical dimeric PBD compounds for inclusion in targeted conjugates. These compounds may have advantages in their preparation and use, particularly in their biological properties and the synthesis of conjugates, and the biological properties of these conjugates.
- the present invention comprises a compound, or a pharmaceutically acceptable salt or solvate thereof, selected from the group consisting of:
- a second aspect of the present invention provides the use of a compound of the first aspect of the invention in the manufacture of a medicament for treating a proliferative disease.
- the second aspect also provides a compound of the first aspect of the invention for use in the treatment of a proliferative disease.
- a third aspect of the present invention comprises a compound according to the first aspect of the invention, except where either:
- a fourth aspect of the present invention comprises a method of making a compound of the first aspect, or a pharmaceutically acceptable salt or solvate thereof, from a compound of the third aspect, or a pharmaceutically acceptable salt or solvate thereof, by deprotection of the imine bond.
- a fifth aspect of the present invention provides a method of making a compound of the first or third aspect of the invention, comprising at least one of the method steps set out below.
- the present invention relates to Conjugates comprising dimers of PBDs linked to a targeting agent, wherein the PBD dimer is a compound as described herein, or a pharmaceutically acceptable salt or solvate thereof (supra).
- the Conjugates have the following formula IV: L-(LU-D) p (IV) or a pharmaceutically acceptable salt or solvate thereof, wherein L is a Ligand unit (i.e., a targeting agent), LU is a Linker unit and D is a Drug unit that is a PBD dimer (see below).
- the subscript p is from 1 to 20.
- the Conjugates comprise a Ligand unit covalently linked to at least one Drug unit by a Linker unit.
- the Ligand unit described more fully below, is a targeting agent that binds to a target moiety.
- the Ligand unit can, for example, specifically bind to a cell component (a Cell Binding Agent) or to other target molecules of interest.
- the present invention also provides methods for the treatment of, for example, various cancers and autoimmune disease. These methods encompass the use of the Conjugates wherein the Ligand unit is a targeting agent that specifically binds to a target molecule.
- the Ligand unit can be, for example, a protein, polypeptide or peptide, such as an antibody, an antigen-binding fragment of an antibody, or other binding agent, such as an Fc fusion protein.
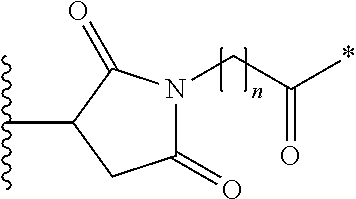
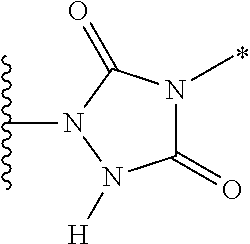
- the PBD dimer D is selected from the group consisting of:
- R 10 and R 11 are as defined in the first aspect, and the asterix indicates the point of attachment to the Linker Unit.
- the drug loading is represented by p, the number of drug molecules per Ligand unit (e.g., an antibody). Drug loading may range from 1 to 20 Drug units (D) per Ligand unit (e.g., Ab or mAb).
- D Drug units
- p represents the average drug loading of the Conjugates in the composition, and p ranges from 1 to 20.
- p is from about 1 to about 8 Drug units per Ligand unit. In some embodiments, p is 1. In some embodiments, p is 2. In some embodiments, p is from about 2 to about 8 Drug units per Ligand unit. In some embodiments, p is from about 2 to about 6, 2 to about 5, or 2 to about 4 Drug units per Ligand unit. In some embodiments, p is about 2, about 4, about 6 or about 8 Drug units per Ligand unit.
- the average number of Drugs units per Ligand unit in a preparation from a conjugation reaction may be characterized by conventional means such as mass spectroscopy, ELISA assay, and HPLC.
- the quantitative distribution of Conjugates in terms of p may also be determined.
- separation, purification, and characterization of homogeneous Conjugates, where p is a certain value, from Conjugates with other drug loadings may be achieved by means such as reverse phase HPLC or electrophoresis.
- the present invention relates to Linker-Drug compounds (i.e., Drug-Linkers) comprising dimers of PBDs (see above) linked to a linking unit.
- Drug-linkers can be used as intermediates for the synthesis of Conjugates comprising dimers of PBDs linked to a targeting agent.
- Drug-Linkers have the following formula V: LU-D (V) or a pharmaceutically acceptable salt or solvate thereof, wherein LU is a Linker unit and D is a Drug unit that is a PBD dimer, as defined in the sixth aspect of the invention.
- FIG. 1 shows the effect on mean tumour volume following treatment with two conjugates of the present invention.
- the pharmaceutically acceptable cation may be inorganic or organic.
- Examples of pharmaceutically acceptable monovalent inorganic cations include, but are not limited to, alkali metal ions such as Na + and K + .
- Examples of pharmaceutically acceptable divalent inorganic cations include, but are not limited to, alkaline earth cations such as Ca 2+ and Mg 2+ .
- Examples of pharmaceutically acceptable organic cations include, but are not limited to, ammonium ion (i.e. NH 4 + ) and substituted ammonium ions (e.g. NH 3 R + , NH 2 R 2 + , NHR 3 + , NR 4 + ).
- Examples of some suitable substituted ammonium ions are those derived from: ethylamine, diethylamine, dicyclohexylamine, triethylamine, butylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine, benzylamine, phenylbenzylamine, choline, meglumine, and tromethamine, as well as amino acids, such as lysine and arginine.
- An example of a common quaternary ammonium ion is N(CH 3 ) 4 + .
- saturated C 1-4 alkyl refers to a monovalent moiety obtained by removing a hydrogen atom from a carbon atom of a hydrocarbon compound having from 1 to 4 carbon atoms, which may be aliphatic or alicyclic.
- saturated C 1-2 alkyl as used herein, pertains to a monovalent moiety obtained by removing a hydrogen atom from a carbon atom of a hydrocarbon compound having from 1 to 2 carbon atoms, i.e. methyl or ethyl.
- saturated alkyl groups include, but are not limited to, methyl (C 1 ), ethyl (C 2 ), propyl (C 3 ), and butyl (C 4 )
- saturated linear alkyl groups include, but are not limited to, methyl (C 1 ), ethyl (C 2 ), n-propyl (C 3 ) and n-butyl (C 4 ).
- saturated branched alkyl groups include iso-propyl (C 3 ), iso-butyl (C 4 ), sec-butyl (C 4 ) and tert-butyl (C 4 ).
- Oxygen protecting group refers to a moiety which masks a hydroxy group, and these are well known in the art. A large number of suitable groups are described on pages 23 to 200 of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3 rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes. Classes of particular interest include silyl ethers (e.g. TMS, TBDMS), substituted methyl ethers (e.g. THP) and esters (e.g. acetate).
- silyl ethers e.g. TMS, TBDMS
- substituted methyl ethers e.g. THP
- esters e.g. acetate
- Carbamate nitrogen protecting group pertains to a moiety which masks the nitrogen in the imine bond, and these are well known in the art. These groups have the following structure:
- R′ 10 is R as defined below.
- suitable groups are described on pages 503 to 549 of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3 rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
- Hemi-aminal nitrogen protecting group pertains to a group having the following structure:
- R′ 10 is R as defined below.
- suitable groups are described on pages 633 to 647 as amide protecting groups of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3 rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
- R is selected from optionally substituted C 1-12 alkyl, C 3-20 heterocyclyl and C 5-20 aryl groups.
- substituted refers to a parent group which bears one or more substituents.
- substituted is used herein in the conventional sense and refers to a chemical moiety which is covalently attached to, or if appropriate, fused to, a parent group.
- substituents are well known, and methods for their formation and introduction into a variety of parent groups are also well known.
- C 1-12 alkyl refers to a monovalent moiety obtained by removing a hydrogen atom from a carbon atom of a hydrocarbon compound having from 1 to 12 carbon atoms, which may be aliphatic or alicydic, and which may be saturated or unsaturated (e.g. partially unsaturated, fully unsaturated).
- C 1-4 alkyl as used herein, pertains to a monovalent moiety obtained by removing a hydrogen atom from a carbon atom of a hydrocarbon compound having from 1 to 4 carbon atoms, which may be aliphatic or alicyclic, and which may be saturated or unsaturated (e.g. partially unsaturated, fully unsaturated).
- C 1-2 alkyl as used herein, pertains to a monovalent moiety obtained by removing a hydrogen atom from a carbon atom of a hydrocarbon compound having from 1 to 2 carbon atoms, i.e. methyl or ethyl.
- alkyl includes the sub-classes alkenyl, alkynyl, cycloalkyl, etc., discussed below.
- saturated alkyl groups include, but are not limited to, methyl (C 1 ), ethyl (C 2 ), propyl (C 3 ), butyl (C 4 ), pentyl (C5), hexyl (C 6 ) and heptyl (C 7 ).
- saturated linear alkyl groups include, but are not limited to, methyl (C 1 ), ethyl (C 2 ), n-propyl (C 3 ), n-butyl (C 4 ), n-pentyl (amyl) (C 5 ), n-hexyl (C 6 ) and n-heptyl (C 7 ).
- saturated branched alkyl groups include iso-propyl (C 3 ), iso-butyl (C 4 ), sec-butyl (C 4 ), tert-butyl (C 4 ), iso-pentyl (C 5 ), and neo-pentyl (C 5 ).
- C 2-12 Alkenyl The term “C 2-12 alkenyl” as used herein, pertains to an alkyl group having one or more carbon-carbon double bonds.
- unsaturated alkenyl groups include, but are not limited to, ethenyl (vinyl, —CH ⁇ CH 2 ), 1-propenyl (—CH ⁇ CH—CH 3 ), 2-propenyl (allyl, —CH—CH ⁇ CH 2 ), isopropenyl (1-methylvinyl, —C(CH 3 ) ⁇ CH 2 ), butenyl (C 4 ), pentenyl (C 5 ), and hexenyl (C 6 ).
- C 2-12 alkynyl The term “C 2-12 alkynyl” as used herein, pertains to an alkyl group having one or more carbon-carbon triple bonds.
- unsaturated alkynyl groups include, but are not limited to, ethynyl (—C ⁇ CH) and 2-propynyl (propargyl, —CH 2 —C ⁇ CH).
- C 3-12 cycloalkyl refers to an alkyl group which is also a cyclyl group; that is, a monovalent moiety obtained by removing a hydrogen atom from an alicyclic ring atom of a cyclic hydrocarbon (carbocyclic) compound, which moiety has from 3 to 7 carbon atoms, including from 3 to 7 ring atoms.
- cycloalkyl groups include, but are not limited to, those derived from:
- C 3-20 heterocyclyl refers to a monovalent moiety obtained by removing a hydrogen atom from a ring atom of a heterocyclic compound, which moiety has from 3 to 20 ring atoms, of which from 1 to 10 are ring heteroatoms.
- each ring has from 3 to 7 ring atoms, of which from 1 to 4 are ring heteroatoms.
- the prefixes e.g. C 3-20 , C 3-7 , C 5-6 , etc.
- the term “C 5-6 heterocyclyl”, as used herein, pertains to a heterocyclyl group having 5 or 6 ring atoms.
- monocyclic heterocyclyl groups include, but are not limited to, those derived from:
- substituted monocyclic heterocyclyl groups include those derived from saccharides, in cyclic form, for example, furanoses (C 5 ), such as arabinofuranose, lyxofuranose, ribofuranose, and xylofuranse, and pyranoses (C 5 ), such as allopyranose, altropyranose, glucopyranose, mannopyranose, gulopyranose, idopyranose, galactopyranose, and talopyranose.
- furanoses such as arabinofuranose, lyxofuranose, ribofuranose, and xylofuranse
- pyranoses C 5
- allopyranose altropyranose
- glucopyranose glucopyranose
- mannopyranose gulopyranose
- idopyranose idopyranose
- galactopyranose galactopyranose
- C 5-20 aryl refers to a monovalent moiety obtained by removing a hydrogen atom from an aromatic ring atom of an aromatic compound, which moiety has from 3 to 20 ring atoms.
- C 5-7 aryl pertains to a monovalent moiety obtained by removing a hydrogen atom from an aromatic ring atom of an aromatic compound, which moiety has from 5 to 7 ring atoms and the term “C 5-10 aryl”, as used herein, pertains to a monovalent moiety obtained by removing a hydrogen atom from an aromatic ring atom of an aromatic compound, which moiety has from 5 to 10 ring atoms.
- each ring has from 5 to 7 ring atoms.
- the prefixes e.g. C 3-20 , C 5-7 , C 5-6 , C 5-10 , etc.
- the term “C 5-6 aryl” as used herein, pertains to an aryl group having 5 or 6 ring atoms.
- the ring atoms may be all carbon atoms, as in “carboaryl groups”.
- carboaryl groups include, but are not limited to, those derived from benzene (i.e. phenyl) (C 6 ), naphthalene (C 10 ), azulene (C 10 ), anthracene (C 14 ), phenanthrene (C 14 ), naphthacene (C 18 ), and pyrene (C 16 ).
- benzene i.e. phenyl
- C 10 naphthalene
- azulene C 10
- anthracene C 14
- phenanthrene C 14
- naphthacene C 18
- pyrene C 16
- aryl groups which comprise fused rings include, but are not limited to, groups derived from indane (e.g. 2,3-dihydro-1H-indene) (C 9 ), indene (C 9 ), isoindene (C 9 ), tetraline (1,2,3,4-tetrahydronaphthalene (C 10 ), acenaphthene (C 12 ), fluorene (C 13 ), phenalene (C 13 ), acephenanthrene (C 15 ), and aceanthrene (C 16 ).
- indane e.g. 2,3-dihydro-1H-indene
- indene C 9
- isoindene C 9
- tetraline (1,2,3,4-tetrahydronaphthalene C 10
- acenaphthene C 12
- fluorene C 13
- phenalene C 13
- acephenanthrene C 15
- aceanthrene
- the ring atoms may include one or more heteroatoms, as in “heteroaryl groups”.
- heteroaryl groups include, but are not limited to, those derived from:
- heteroaryl which comprise fused rings, include, but are not limited to:
- Halo —F, —Cl, —Br, and —I.
- Ether —OR, wherein R is an ether substituent, for example, a C 1-7 alkyl group (also referred to as a C 1-7 alkoxy group, discussed below), a C 3-20 heterocyclyl group (also referred to as a C 3-20 heterocyclyloxy group), or a C 5-20 aryl group (also referred to as a C 5-20 aryloxy group), preferably a C 1-7 alkyl group.
- R is an ether substituent, for example, a C 1-7 alkyl group (also referred to as a C 1-7 alkoxy group, discussed below), a C 3-20 heterocyclyl group (also referred to as a C 3-20 heterocyclyloxy group), or a C 5-20 aryl group (also referred to as a C 5-20 aryloxy group), preferably a C 1-7 alkyl group.
- Alkoxy —OR, wherein R is an alkyl group, for example, a C 1-7 alkyl group.
- C 1-7 alkoxy groups include, but are not limited to, —OMe (methoxy), —OEt (ethoxy), —O(nPr) (n-propoxy), —O(iPr) (isopropoxy), —O(nBu) (n-butoxy), —O(sBu) (sec-butoxy), —O(iBu) (isobutoxy), and —O(tBu) (tert-butoxy).
- Acetal —CH(OR 1 )(OR 2 ), wherein R 1 and R 2 are independently acetal substituents, for example, a C 1-7 alkyl group, a C 3-20 heterocycyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group, or, in the case of a “cyclic” acetal group, R 1 and R 2 , taken together with the two oxygen atoms to which they are attached, and the carbon atoms to which they are attached, form a heterocyclic ring having from 4 to 8 ring atoms.
- acetal groups include, but are not limited to, —CH(OMe) 2 , —CH(OEt) 2 , and —CH(OMe)(OEt).
- Hemiacetal —CH(OH)(OR 1 ), wherein R 1 is a hemiacetal substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R 1 is a hemiacetal substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- hemiacetal groups include, but are not limited to, —CH(OH)(OMe) and —CH(OH)(OEt).
- Ketal —CR(OR 1 )(OR 2 ), where R 1 and R 2 are as defined for acetals, and R is a ketal substituent other than hydrogen, for example, a C 1-7 alkyl group, a C 3-20 heterocycyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- ketal groups include, but are not limited to, —C(Me)(OMe) 2 , —C(Me)(OEt) 2 , —C(Me)(OMe)(OEt), —C(Et)(OMe) 2 , —C(Et)(OEt) 2 , and —C(Et)(OMe)(OEt).
- Hemiketal —CR(OH)(OR 1 ), where R 1 is as defined for hemiacetals, and R is a hemiketal substituent other than hydrogen, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- hemiacetal groups include, but are not limited to, —C(Me)(OH)(OMe), —C(Et)(OH)(OMe), —C(Me)(OH)(OEt), and —C(Et)(OH)(OEt).
- Imino (imine): ⁇ NR wherein R is an imino substituent, for example, hydrogen, C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably hydrogen or a C 1-7 alkyl group.
- ester groups include, but are not limited to, ⁇ NH, ⁇ NMe, ⁇ NEt, and ⁇ NPh.
- R is an acyl substituent, for example, a C 1-7 alkyl group (also referred to as C 1-7 alkylacyl or C 1-7 alkanoyl), a C 3-20 heterocyclyl group (also referred to as C 3-20 heterocyclylacyl), or a C 5-20 aryl group (also referred to as C 5-20 arylacyl), preferably a C 1-7 alkyl group.
- R is an acyl substituent, for example, a C 1-7 alkyl group (also referred to as C 1-7 alkylacyl or C 1-7 alkanoyl), a C 3-20 heterocyclyl group (also referred to as C 3-20 heterocyclylacyl), or a C 5-20 aryl group (also referred to as C 5-20 arylacyl), preferably a C 1-7 alkyl group.
- acyl groups include, but are not limited to, —C( ⁇ O)CH 3 (acetyl), —C( ⁇ O)CH 2 CH 3 (propionyl), —C( ⁇ O)C(CH 3 ) 3 (t-butyryl), and —C( ⁇ O)Ph (benzoyl, phenone).
- Thiolocarboxy thiolocarboxylic acid: —C( ⁇ O)SH.
- Imidic acid —C( ⁇ NH)OH.
- Ester (carboxylate, carboxylic acid ester, oxycarbonyl): —C( ⁇ O)OR, wherein R is an ester substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- ester groups include, but are not limited to, —C( ⁇ O)OCH 3 , —C( ⁇ O)OCH 2 CH 3 , —C( ⁇ O)OC(CH 3 ) 3 , and —C( ⁇ O)OPh.
- R is an acyloxy substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- acyloxy groups include, but are not limited to, —OC( ⁇ O)CH 3 (acetoxy), —OC( ⁇ O)CH 2 CH 3 , —OC( ⁇ O)C(CH 3 ) 3 , —OC( ⁇ O)Ph, and —OC( ⁇ O)CH 2 Ph.
- Oxycarboyloxy —OC( ⁇ O)OR, wherein R is an ester substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- ester groups include, but are not limited to, —OC( ⁇ O)OCH 3 , —OC( ⁇ O)OCH 2 CH 3 , —OC( ⁇ O)OC(CH 3 ) 3 , and —OC( ⁇ O)OPh.
- R 1 and R 2 are independently amino substituents, for example, hydrogen, a C 1-7 alkyl group (also referred to as C 1-7 alkylamino or di-C 1-7 alkylamino), a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably H or a C 1-7 alkyl group, or, in the case of a “cyclic” amino group, R 1 and R 2 , taken together with the nitrogen atom to which they are attached, form a heterocyclic ring having from 4 to 8 ring atoms.
- R 1 and R 2 are independently amino substituents, for example, hydrogen, a C 1-7 alkyl group (also referred to as C 1-7 alkylamino or di-C 1-7 alkylamino), a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably H or a C 1-7 alkyl group, or, in the case of a “cyclic” amino group, R 1 and R 2 ,
- Amino groups may be primary (—NH 2 ), secondary (—NHR 1 ), or tertiary (—NHR 1 R 2 ), and in cationic form, may be quaternary (— + NR 1 R 2 R 3 ).
- Examples of amino groups include, but are not limited to, —NH 2 , —NHCH 3 , —NHC(CH 3 ) 2 , —N(CH 3 ) 2 , —N(CH 2 CH 3 ) 2 , and —NHPh.
- Examples of cyclic amino groups include, but are not limited to, aziridino, azetidino, pyrrolidino, piperidino, piperazino, morpholino, and thiomorpholino.
- amido groups include, but are not limited to, —C( ⁇ O)NH 2 , —C( ⁇ O)NHCH 3 , —C( ⁇ O)N(CH 3 ) 2 , —C( ⁇ O)NHCH 2 CH 3 , and —C( ⁇ O)N(CH 2 CH 3 ) 2 , as well as amido groups in which R 1 and R 2 , together with the nitrogen atom to which they are attached, form a heterocylic structure as in, for example, piperidinocarbonyl, morpholinocarbonyl, thiomorpholinocarbonyl, and piperazinocarbonyl.
- Thioamido (thiocarbamyl) —C( ⁇ S)NR 1 R 2 , wherein R 1 and R 2 are independently amino substituents, as defined for amino groups.
- amido groups include, but are not limited to, —C( ⁇ S)NH 2 , —C( ⁇ S)NHCH 3 , —C( ⁇ S)N(CH 3 ) 2 , and —C( ⁇ S)NHCH 2 CH 3 .
- acylamide groups include, but are not limited to, —NHC( ⁇ O)CH 3 , —NHC( ⁇ O)CH 2 CH 3 , and —NHC( ⁇ O)Ph.
- R 1 and R 2 may together form a cyclic structure, as in, for example, succinimidyl, maleimidyl, and phthalimidyl:
- Aminocarbonyloxy —OC( ⁇ O)NR 1 R 2 , wherein R 1 and R 2 are independently amino substituents, as defined for amino groups.
- Examples of aminocarbonyloxy groups include, but are not limited to, —OC( ⁇ O)NH 2 , —OC( ⁇ O)NHMe, —OC( ⁇ O)NMe 2 , and —OC( ⁇ O)NEt 2 .
- R 2 and R 3 are independently amino substituents, as defined for amino groups, and R 1 is a ureido substituent, for example, hydrogen, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably hydrogen or a C 1-7 alkyl group.
- ureido groups include, but are not limited to, —NHCONH 2 , —NHCONHMe, —NHCONHEt, —NHCONMe 2 , —NHCONEt 2 , —NMeCONH 2 , —NMeCONHMe, —NMeCONHEt, —NMeCONMe 2 , and —NMeCONEt 2 .
- Tetrazolyl a five membered aromatic ring having four nitrogen atoms and one carbon atom
- Imino ⁇ NR, wherein R is an imino substituent, for example, for example, hydrogen, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably H or a C 1-7 alkyl group.
- imino groups include, but are not limited to, ⁇ NH, ⁇ NMe, and ⁇ NEt.
- amidine groups include, but are not limited to, —C( ⁇ NH)NH 2 , —C( ⁇ NH)NMe 2 , and —C( ⁇ NMe)NMe 2 .
- Nitroso —NO.
- C 1-7 alkylthio groups include, but are not limited to, —SCH 3 and —SCH 2 CH 3 .
- Disulfide —SS—R, wherein R is a disulfide substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group (also referred to herein as C 1-7 alkyl disulfide).
- R is a disulfide substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group (also referred to herein as C 1-7 alkyl disulfide).
- C 1-7 alkyl disulfide groups include, but are not limited to, —SSCH 3 and —SSCH 2 CH 3 .
- R is a sulfine substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfine groups include, but are not limited to, —S( ⁇ O)CH 3 and —S( ⁇ O)CH 2 CH 3 .
- sulfone groups include, but are not limited to, —S( ⁇ O) 2 CH 3 (methanesulfonyl, mesyl), —S( ⁇ O) 2 CF 3 (triflyl), —S( ⁇ O) 2 CH 2 CH 3 (esyl), —S( ⁇ O) 2 C 4 F 9 (nonaflyl), —S( ⁇ O) 2 CH 2 CF 3 (tresyl), —S( ⁇ O) 2 CH 2 CH 2 NH 2 (tauryl), —S( ⁇ O) 2 Ph (phenylsulfonyl, besyl), 4-methylphenylsulfonyl (tosyl), 4-chlorophenylsulfonyl (closyl), 4-bromophenylsulfonyl (brosyl), 4-nitrophenyl (nosyl), 2-naphthalenesulfonate (napsyl), and 5-dimethylamino-naphthalen
- Sulfinate (sulfinic acid ester): —S( ⁇ O)OR; wherein R is a sulfinate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R is a sulfinate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfinate groups include, but are not limited to, —S( ⁇ O)OCH 3 (methoxysulfinyl; methyl sulfinate) and —S( ⁇ O)OCH 2 CH 3 (ethoxysulfinyl; ethyl sulfinate).
- Sulfonate (sulfonic acid ester): —S( ⁇ O) 2 OR, wherein R is a sulfonate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R is a sulfonate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfonate groups include, but are not limited to, —S( ⁇ O) 2 OCH 3 (methoxysulfonyl; methyl sulfonate) and —S( ⁇ O) 2 OCH 2 CH 3 (ethoxysulfonyl; ethyl sulfonate).
- Sulfinyloxy —OS( ⁇ O)R, wherein R is a sulfinyloxy substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R is a sulfinyloxy substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfinyloxy groups include, but are not limited to, —OS( ⁇ O)CH 3 and —OS( ⁇ O)CH 2 CH 3 .
- Sulfonyloxy —OS( ⁇ O) 2 R, wherein R is a sulfonyloxy substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R is a sulfonyloxy substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfonyloxy groups include, but are not limited to, —OS( ⁇ O) 2 CH 3 (mesylate) and —OS( ⁇ O) 2 CH 2 CH 3 (esylate).
- Sulfate —OS( ⁇ O) 2 OR; wherein R is a sulfate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R is a sulfate substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfate groups include, but are not limited to, —OS( ⁇ O) 2 OCH 3 and —SO( ⁇ O) 2 OCH 2 CH 3 .
- Sulfamyl (sulfamoyl; sulfinic acid amide; sulfinamide): —S( ⁇ O)NR 1 R 2 , wherein R 1 and R 2 are independently amino substituents, as defined for amino groups.
- R 1 and R 2 are independently amino substituents, as defined for amino groups.
- sulfamyl groups include, but are not limited to, —S( ⁇ O)NH 2 , —S( ⁇ O)NH(CH 3 ), —S( ⁇ O)N(CH 3 ) 2 , —S( ⁇ O)NH(CH 2 CH 3 ), —S( ⁇ O)N(CH 2 CH 3 ) 2 , and —S( ⁇ O)NHPh.
- Sulfonamido (sulfinamoyl; sulfonic acid amide; sulfonamide): —S( ⁇ O) 2 NR 1 R 2 , wherein R 1 and R 2 are independently amino substituents, as defined for amino groups.
- sulfonamido groups include, but are not limited to, —S( ⁇ O) 2 NH 2 , —S( ⁇ O) 2 NH(CH 3 ), —S( ⁇ O) 2 N(CH 3 ) 2 , —S( ⁇ O) 2 NH(CH 2 CH 3 ), —S( ⁇ O) 2 N(CH 2 CH 3 ) 2 , and —S( ⁇ O) 2 NHPh.
- Sulfamino —NR 1 S( ⁇ O) 2 OH, wherein R 1 is an amino substituent, as defined for amino groups.
- R 1 is an amino substituent, as defined for amino groups.
- sulfamino groups include, but are not limited to, —NHS( ⁇ O) 2 OH and —N(CH 3 )S( ⁇ O) 2 OH.
- Sulfonamino —NR 1 S( ⁇ O) 2 R, wherein R 1 is an amino substituent, as defined for amino groups, and R is a sulfonamino substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R 1 is an amino substituent, as defined for amino groups
- R is a sulfonamino substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfonamino groups include, but are not limited to, —NHS( ⁇ O) 2 CH 3 and —N(CH 3 )S( ⁇ O) 2 C 6 H 5 .
- Sulfinamino —NR 1 S( ⁇ O)R, wherein R 1 is an amino substituent, as defined for amino groups, and R is a sulfinamino substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- R 1 is an amino substituent, as defined for amino groups
- R is a sulfinamino substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group.
- sulfinamino groups include, but are not limited to, —NHS( ⁇ O)CH 3 and —N(CH 3 )S( ⁇ O)C 6 H 5 .
- R is a phosphino substituent, for example, —H, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- Examples of phosphino groups include, but are not limited to, —PH 2 , —P(CH 3 ) 2 , —P(CH 2 CH 3 ) 2 , —P(t-Bu) 2 , and —P(Ph) 2 .
- Phospho —P( ⁇ O) 2 .
- Phosphinyl phosphine oxide: —P( ⁇ O)R 2 , wherein R is a phosphinyl substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group or a C 5-20 aryl group.
- R is a phosphinyl substituent, for example, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably a C 1-7 alkyl group or a C 5-20 aryl group.
- Examples of phosphinyl groups include, but are not limited to, —P( ⁇ O)(CH 3 ) 2 , —P( ⁇ O)(CH 2 CH 3 ) 2 , —P( ⁇ O)(t-Bu) 2 , and —P( ⁇ O)(Ph) 2 .
- Phosphonic acid (phosphono) —P( ⁇ O)(OH) 2 .
- R is a phosphonate substituent, for example, —H, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- Examples of phosphonate groups include, but are not limited to, —P( ⁇ O)(OCH 3 ) 2 , —P( ⁇ O)(OCH 2 CH 3 ) 2 , —P( ⁇ O)(O-t-Bu) 2 , and —P(
- Phosphoric acid —OP( ⁇ O)(OH) 2 .
- Phosphate (phosphonooxy ester) —OP( ⁇ O)(OR) 2 , where R is a phosphate substituent, for example, —H, a C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- phosphate groups include, but are not limited to, —OP( ⁇ O)(OCH 3 ) 2 , —OP( ⁇ O)(OCH 2 CH 3 ) 2 , —OP( ⁇ O)(O-t-Bu) 2 , and —OP( ⁇ O)(OPh) 2 .
- Phosphorous acid —OP(OH) 2 .
- Phosphite —OP(OR) 2 , where R is a phosphite substituent, for example, —H, a C 1-7 alkyl group, a C 3-20 heterocycyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- phosphite groups include, but are not limited to, —OP(OCH 3 ) 2 , —OP(OCH 2 CH 3 ) 2 , —OP(O-t-Bu) 2 , and —OP(OPh) 2 .
- Phosphoramidite —OP(OR 1 )—NR 2 2 , where R 1 and R 2 are phosphoramidite substituents, for example, —H, a (optionally substituted) C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- Examples of phosphoramidite groups include, but are not limited to, —OP(OCH 2 CH 3 )—N(CH 3 ) 2 , —OP(OCH 2 CH 3 )—N(i-Pr) 2 , and —OP(OCH 2 CH 2 CN)—N(i-Pr) 2 .
- Phosphoramidate —OP( ⁇ O)(OR 1 )—NR 2 2 , where R 1 and R 2 are phosphoramidate substituents, for example, —H, a (optionally substituted) C 1-7 alkyl group, a C 3-20 heterocyclyl group, or a C 5-20 aryl group, preferably —H, a C 1-7 alkyl group, or a C 5-20 aryl group.
- Examples of phosphoramidate groups include, but are not limited to, —OP( ⁇ O)(OCH 2 CH 3 )—N(CH 3 ) 2 , —OP( ⁇ O)(OCH 2 CH 3 )—N(i-Pr) 2 , and —OP( ⁇ O)(OCH 2 CH 2 CN)—N(i-Pr) 2 .
- the present invention provides Conjugates comprising a PBD dimer connected to a Ligand unit via a Linker unit.
- the Linker unit includes a Stretcher unit (A), a Specificity unit (L 1 ), and a Spacer unit (L 2 ).
- the Linker unit is connected at one end to the Ligand unit (L) and at the other end to the PBD dimer compound (D).
- the Conjugate has the formula: L-(A 1 a -L 1 s -L 2 y -D) p L-(A 1 a -L s 1 -D) p , L-(A 1 -L 1 -D) p , or L-(A 1 -D) p
- the Ligand unit (L) is a Cell Binding Agent (CBA) that specifically binds to a target molecule on the surface of a target cell.
- CBA Cell Binding Agent
- the Ligand unit (L) is a Cell Binding Agent (CBA) that specifically binds to a target molecule on the surface of a target cell.
- CBA Cell Binding Agent
- L 1 can be a cleavable Specificity unit, and may be referred to as a “trigger” that when cleaved activates a self-immolative group (or self-immolative groups) L 2 , when a self-immolative group(s) is present.
- the Specificity unit L 1 is cleaved, or the linkage (i.e., the covalent bond) between L 1 and L 2 is cleaved, the self-immolative group releases the Drug unit (D).
- the Ligand unit (L) is a Cell Binding Agent (CBA) that specifically binds to a target molecule on the surface of a target cell.
- CBA Cell Binding Agent
- L 1 and L 2 can vary widely. These groups are chosen on the basis of their characteristics, which may be dictated in part, by the conditions at the site to which the conjugate is delivered.
- the Specificity unit L 1 is cleavable, the structure and/or sequence of L 1 is selected such that it is cleaved by the action of enzymes present at the target site (e.g., the target cell).
- L 1 units that are cleavable by changes in pH (e.g. acid or base labile), temperature or upon irradiation (e.g. photolabile) may also be used.
- L 1 units that are cleavable under reducing or oxidising conditions may also find use in the Conjugates.
- L 1 may comprise one amino acid or a contiguous sequence of amino acids.
- the amino acid sequence may be the target substrate for an enzyme.
- L 1 is cleavable by the action of an enzyme.
- the enzyme is an esterase or a peptidase.
- L 1 may be cleaved by a lysosomal protease, such as a cathepsin.
- L 2 is present and together with —C( ⁇ O)O— forms a self-immolative group or self-immolative groups. In some embodiments, —C( ⁇ O)O— also is a self-immolative group.
- the enzyme cleaves the bond between L 1 and L 2 , whereby the self-immolative group(s) release the Drug unit.
- L 1 and L 2 where present, may be connected by a bond selected from:
- An amino group of L 1 that connects to L 2 may be the N-terminus of an amino acid or may be derived from an amino group of an amino acid side chain, for example a lysine amino acid side chain.
- a carboxyl group of L 1 that connects to L 2 may be the C-terminus of an amino acid or may be derived from a carboxyl group of an amino acid side chain, for example a glutamic acid amino acid side chain.
- a hydroxy group of L 1 that connects to L 2 may be derived from a hydroxy group of an amino acid side chain, for example a serine amino acid side chain.
- —C( ⁇ O)O— and L 2 together form the group:
- Y is NH
- n is 0 or 1. Preferably, n is 0.
- PABC p-aminobenzylcarbonyl linker
- the Drug unit i.e., the asymmetric PBD
- —C( ⁇ O)O— and L 2 together form a group selected from:
- —C( ⁇ O)O— and L 2 together form a group selected from:
- D is N.
- D is CH.
- E is O or S.
- F is CH.
- the covalent bond between L 1 and L 2 is a cathepsin labile (e.g., cleavable) bond.
- L 1 comprises a dipeptide.
- the amino acids in the dipeptide may be any combination of natural amino acids and non-natural amino acids.
- the dipeptide comprises natural amino acids.
- the linker is a cathepsin labile linker
- the dipeptide is the site of action for cathepsin-mediated cleavage. The dipeptide then is a recognition site for cathepsin.
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO— is selected from:
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO— is selected from:
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO—, is -Phe-Lys-, Val-Cit or -Val-Ala-.
- dipeptide combinations of interest include:
- dipeptide combinations may be used, including those described by Dubowchik et al., which is incorporated herein by reference in its entirety and for all purposes.
- the amino acid side chain is chemically protected, where appropriate.
- the side chain protecting group may be a group as discussed below.
- Protected amino acid sequences are cleavable by enzymes. For example, a dipeptide sequence comprising a Boc side chain-protected Lys residue is cleavable by cathepsin.
- —X 2 — is connected indirectly to the Drug unit.
- the Spacer unit L 2 is present.
- —X 2 — is connected directly to the Drug unit.
- the Spacer unit L 2 is absent.
- the dipeptide is used in combination with a self-immolative group(s) (the Spacer unit).
- the self-immolative group(s) may be connected to —X 2 —.
- —X 2 — is connected directly to the self-immolative group.
- —X 2 — is connected to the group Y of the self-immolative group.
- the group —X 2 —CO— is connected to Y, where Y is NH.
- —X 1 — is connected directly to A 1 .
- the group NH—X 1 — (the amino terminus of X 1 ) is connected to A 1 .
- a 1 may comprise the functionality —CO— thereby to form an amide link with —X 1 —.
- L 1 and L 2 together with —OC( ⁇ O)— comprise the group —X 1 —X 2 -PABC-.
- the PABC group is connected directly to the Drug unit.
- the self-immolative group and the dipeptide together form the group -Phe-Lys-PABC-, which is illustrated below:
- the self-immolative group and the dipeptide together form the group -Val-Ala-PABC-, which is illustrated below:
- L 1 and L 2 together with —OC( ⁇ O)— represent:
- E is selected such that the group is susceptible to cleavage, e.g., by light or by the action of an enzyme.
- E may be —NO 2 or glucuronic acid (e.g., ⁇ -glucuronic acid).
- the former may be susceptible to the action of a nitroreductase, the latter to the action of a ⁇ -glucuronidase.
- the group Y may be a covalent bond.
- the group Y may be a functional group selected from:
- the group Y is preferably —NH—, —CH 2 —, —O—, and —S—.
- L 1 and L 2 together with —OC( ⁇ O)— represent:
- L 1 and L 2 together represent:
- the Specificity unit L 1 and the Ligand unit are indirectly connected via the Stretcher unit.
- L 1 and A 1 may be connected by a bond selected from:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- the group A 1 is:
- connection between the Ligand unit and A 1 is through a thiol residue of the Ligand unit and a maleimide group of A 1 .
- connection between the Ligand unit and A 1 is:
- the maleimide-derived group is replaced with the group:
- the maleimide-derived group is replaced with a group, which optionally together with a Ligand unit (e.g., a Cell Binding Agent), is selected from:
- a Ligand unit e.g., a Cell Binding Agent
- the maleimide-derived group is replaced with a group, which optionally together with the Ligand unit, is selected from:
- the Stretcher unit A 1 is present, the Specificity unit L 1 is present and Spacer unit L 2 is absent.
- L 1 and the Drug unit are directly connected via a bond.
- L 2 is a bond.
- L 1 and D may be connected by a bond selected from:
- L 1 and D are preferably connected by a bond:
- L 1 comprises a dipeptide and one end of the dipeptide is linked to D.
- the amino acids in the dipeptide may be any combination of natural amino acids and non-natural amino acids.
- the dipeptide comprises natural amino acids.
- the linker is a cathepsin labile linker
- the dipeptide is the site of action for cathepsin-mediated cleavage. The dipeptide then is a recognition site for cathepsin.
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO— is selected from:
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO— is selected from:
- the group —X 1 —X 2 — in dipeptide, —NH—X 1 —X 2 —CO—, is -Phe-Lys- or -Val-Ala-.
- dipeptide combinations of interest include:
- dipeptide combinations may be used, including those described above.
- L 1 -D is:
- the dipeptide is valine-alanine and L 1 -D is:
- the dipeptide is phenylalnine-lysine and L 1 -D is:
- the dipeptide is valine-citrulline.
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the group L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the groups L-A 1 -L 1 are:
- the Stretcher unit is an acetamide unit, having the formula: -CH 2 —CO—N-*
- Linker-Drug compounds are provided for conjugation to a Ligand unit.
- the Linker-Drug compounds are designed for connection to a Cell Binding Agent.
- the Drug Linker compound has the formula:
- the Drug Linker compound has the formula: G 1 -L 1 -L 2 -.
- L 1 and L 2 are as defined above. References to connection to A 1 can be construed here as referring to a connection to G 1 .
- L 1 comprises an amino acid
- the side chain of that amino acid may be protected. Any suitable protecting group may be used.
- the side chain protecting groups are removable with other protecting groups in the compound, where present.
- the protecting groups may be orthogonal to other protecting groups in the molecule, where present.
- Suitable protecting groups for amino acid side chains include those groups described in the Novabiochem Catalog 2006/2007. Protecting groups for use in a cathepsin labile linker are also discussed in Dubowchik et al.
- the group L 1 includes a Lys amino acid residue.
- the side chain of this amino acid may be protected with a Boc or Alloc protected group.
- a Boc protecting group is most preferred.
- the functional group G 1 forms a connecting group upon reaction with a Ligand unit (e.g., a cell binding agent.
- a Ligand unit e.g., a cell binding agent.
- the functional group G 1 is or comprises an amino, carboxylic acid, hydroxy, thiol, or maleimide group for reaction with an appropriate group on the Ligand unit.
- G 1 comprises a maleimide group.
- the group G 1 is an alkyl maleimide group. This group is suitable for reaction with thiol groups, particularly cysteine thiol groups, present in the cell binding agent, for example present in an antibody.
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the group G 1 is:
- the maleimide-derived group is replaced with the group:
- the maleimide group is replaced with a group selected from:
- —C( ⁇ O)CH 2 X may be preferred, especially when the carbonyl group is bound to —NH—.
- L 1 is present, and G 1 is —NH 2 , —NHMe, —COOH, —OH or —SH.
- G 1 is —NH 2 or —NHMe. Either group may be the N-terminal of an L 1 amino acid sequence.
- L 1 is present and G 1 is —NH 2 , and L 1 is an amino acid sequence —X 1 —X 2 —, as defined above.
- L 1 is present and G 1 is COOH. This group may be the C-terminal of an L 1 amino acid sequence.
- L 1 is present and G 1 is OH.
- L 1 is present and G 1 is SH.
- the group G 1 may be convertable from one functional group to another.
- L 1 is present and G 1 is —NH 2 .
- This group is convertable to another group G 1 comprising a maleimide group.
- the group —NH 2 may be reacted with an acids or an activated acid (e.g., N-succinimide forms) of those G 1 groups comprising maleimide shown above.
- the group G 1 may therefore be converted to a functional group that is more appropriate for reaction with a Ligand unit.
- L 1 is present and G 1 is —NH 2 , —NHMe, —COOH, —OH or —SH.
- these groups are provided in a chemically protected form.
- the chemically protected form is therefore a precursor to the linker that is provided with a functional group.
- G 1 is —NH 2 in a chemically protected form.
- the group may be protected with a carbamate protecting group.
- the carbamate protecting group may be selected from the group consisting of:
- G 1 is —NH 2 , it is protected with an Fmoc group.
- the protecting group is the same as the carbamate protecting group of the capping group.
- the protecting group is not the same as the carbamate protecting group of the capping group. In this embodiment, it is preferred that the protecting group is removable under conditions that do not remove the carbamate protecting group of the capping group.
- the chemical protecting group may be removed to provide a functional group to form a connection to a Ligand unit.
- this functional group may then be converted to another functional group as described above
- the active group is an anine.
- This amine is preferably the N-terminal amine of a peptide, and may be the N-terminal amine of the preferred dipeptides of the invention.
- the active group may be reacted to yield the functional group that is intended to form a connection to a Ligand unit.
- the Linker unit is a precursor to the Linker unit having an active group.
- the Linker unit comprises the active group, which is protected by way of a protecting group. The protecting group may be removed to provide the Linker unit having an active group.
- the protecting group may be an amine protecting group, such as those described in Green and Wuts.
- the protecting group is preferably orthogonal to other protecting groups, where present, in the Linker unit.
- the protecting group is orthogonal to the capping group.
- the active group protecting group is removable whilst retaining the capping group.
- the protecting group and the capping group is removable under the same conditions as those used to remove the capping group.
- the Linker unit is:
- the Linker unit is:
- the Ligand Unit may be of any kind, and include a protein, polypeptide, peptide and a non-peptidic agent that specifically binds to a target molecule.
- the Ligand unit may be a protein, polypeptide or peptide.
- the Ligand unit may be a cyclic polypeptide.
- These Ligand units can include antibodies or a fragment of an antibody that contains at least one target molecule-binding site, lymphokines, hormones, growth factors, or any other cell binding molecule or substance that can specifically bind to a target.
- the ligand Unit is also referred to herein as a “binding agent” or “targeting agent”.
- the terms “specifically binds” and “specific binding” refer to the binding of an antibody or other protein, polypeptide or peptide to a predetermined molecule (e.g., an antigen).
- a predetermined molecule e.g., an antigen
- the antibody or other molecule binds with an affinity of at least about 1 ⁇ 10 7 M ⁇ 1 , and binds to the predetermined molecule with an affinity that is at least two-fold greater than its affinity for binding to a non-specific molecule (e.g., BSA, casein) other than the predetermined molecule or a closely-related molecule.
- Ligand units include those agents described for use in WO 2007/085930, which is incorporated herein in its entirety and for all purposes.
- the Ligand unit is a Cell Binding Agent that binds to an extracellular target on a cell.
- a Cell Binding Agent can be a protein, polypeptide, peptide or a non-peptidic agent.
- the Cell Binding Agent may be a protein, polypeptide or peptide.
- the Cell Binding Agent may be a cyclic polypeptide.
- the Cell Binding Agent also may be antibody or an antigen-binding fragment of an antibody.
- the present invention provides an antibody-drug conjugate (ADC).
- ADC antibody-drug conjugate
- the antibody is a monoclonal antibody; chimeric antibody; humanized antibody; fully human antibody; or a single chain antibody.
- the antibody is a fragment of one of these antibodies having biological activity. Examples of such fragments include Fab, Fab′, F(ab′) 2 and Fv fragments.
- the antibody may be a diabody, a domain antibody (DAB) or a single chain antibody.
- DAB domain antibody
- the antibody is a monoclonal antibody.
- Antibodies for use in the present invention include those antibodies described in WO 2005/082023 which is incorporated by reference herein in its entirety and for all purposes. Particularly preferred are those antibodies for tumour-associated antigens. Examples of those antigens known in the art include, but are not limited to, those tumour-associated antigens set out in WO 2005/082023. See, for instance, pages 41-55.
- the conjugates are designed to target tumour cells via their cell surface antigens.
- the antigens may be cell surface antigens which are either over-expressed or expressed at abnormal times or cell types.
- the target antigen is expressed only on proliferative cells (preferably tumour cells); however this is rarely observed in practice.
- target antigens are usually selected on the basis of differential expression between proliferative and healthy tissue.
- Antibodies have been raised to target specific tumour related antigens including:
- the Ligand unit is connected to the Linker unit. In one embodiment, the Ligand unit is connected to A, where present, of the Linker unit.
- connection between the Ligand unit and the Linker unit is through a thioether bond.
- connection between the Ligand unit and the Linker unit is through a disulfide bond.
- connection between the Ligand unit and the Linker unit is through an amide bond.
- connection between the Ligand unit and the Linker unit is through an ester bond.
- connection between the Ligand unit and the Linker is formed between a thiol group of a cysteine residue of the Ligand unit and a maleimide group of the Linker unit.
- the cysteine residues of the Ligand unit may be available for reaction with the functional group of the Linker unit to form a connection.
- the thiol groups of the antibody may participate in interchain disulfide bonds. These interchain bonds may be converted to free thiol groups by e.g. treatment of the antibody with DTT prior to reaction with the functional group of the Linker unit.
- the cysteine residue is introduced into the heavy or light chain of an antibody.
- Positions for cysteine insertion by substitution in antibody heavy or light chains include those described in Published U.S. Application No. 2007-0092940 and International Patent Publication WO2008/070593, which are herein incorporated by reference in their entirety and for all purposes.
- the compounds and conjugates of the present invention may be used in a method of therapy.
- a method of treatment comprising administering to a subject in need of treatment a therapeutically-effective amount of a compound or conjugate disclosed herein.
- therapeutically effective amount is an amount sufficient to show benefit to a patient. Such benefit may be at least amelioration of at least one symptom.
- the actual amount administered, and rate and time-course of administration, will depend on the nature and severity of what is being treated. Prescription of treatment, e.g. decisions on dosage, is within the responsibility of general practitioners and other medical doctors.
- a compound or conjugate may be administered alone or in combination with other treatments, either simultaneously or sequentially dependent upon the condition to be treated.
- treatments and therapies include, but are not limited to, chemotherapy (the administration of active agents, including, e.g. drugs; surgery; and radiation therapy.
- compositions according to the present invention may comprise, in addition to the active ingredient, i.e. a compound or conjugate disclosed herein, a pharmaceutically acceptable excipient, carrier, buffer, stabiliser or other materials well known to those skilled in the art. Such materials should be non-toxic and should not interfere with the efficacy of the active ingredient.
- a pharmaceutically acceptable excipient e.g. cutaneous, subcutaneous, or intravenous.
- compositions for oral administration may be in tablet, capsule, powder or liquid form.
- a tablet may comprise a solid carrier or an adjuvant.
- Liquid pharmaceutical compositions generally comprise a liquid carrier such as water, petroleum, animal or vegetable oils, mineral oil or synthetic oil. Physiological saline solution, dextrose or other saccharide solution or glycols such as ethylene glycol, propylene glycol or polyethylene glycol may be included.
- a capsule may comprise a solid carrier such a gelatin.
- the active ingredient will be in the form of a parenterally acceptable aqueous solution which is pyrogen-free and has suitable pH, isotonicity and stability.
- a parenterally acceptable aqueous solution which is pyrogen-free and has suitable pH, isotonicity and stability.
- isotonic vehicles such as Sodium Chloride Injection. Ringer's Injection, Lactated Ringer's Injection. Preservatives, stabilisers, buffers, antioxidants and/or other additives may be included, as required.
- the Compounds and Conjugates can be used to treat proliferative disease and autoimmune disease.
- proliferative disease pertains to an unwanted or uncontrolled cellular proliferation of excessive or abnormal cells which is undesired, such as, neoplastic or hyperplastic growth, whether in vitro or in vivo.
- proliferative conditions include, but are not limited to, benign, pre-malignant, and malignant cellular proliferation, including but not limited to, neoplasms and tumours (e.g., histocytoma, glioma, astrocyoma, osteoma), cancers (e.g.
- lung cancer small cell lung cancer, gastrointestinal cancer, bowel cancer, colon cancer, breast carinoma, ovarian carcinoma, prostate cancer, testicular cancer, liver cancer, kidney cancer, bladder cancer, pancreatic cancer, brain cancer, sarcoma, osteosarcoma, Kaposi's sarcoma, melanoma), leukemias, psoriasis, bone diseases, fibroprcliferative disorders (e.g. of connective tissues), and atherosclerosis.
- cancers of interest include, but are not limited to, haematological; malignancies such as leukemias and lymphomas, such as non-Hodgkin lymphoma, and subtypes such as DLBCL, marginal zone, mantle zone, and follicular, Hodgkin lymphoma, AML, and other cancers of B or T cell origin.
- malignancies such as leukemias and lymphomas, such as non-Hodgkin lymphoma
- subtypes such as DLBCL, marginal zone, mantle zone, and follicular, Hodgkin lymphoma, AML, and other cancers of B or T cell origin.
- autoimmune disease examples include the following: rheumatoid arthritis, autoimmune demyelinative diseases (e.g., multiple sclerosis, allergic encephalomyelitis), psoriatic arthritis, endocrine ophthalmopathy, uveoretinitis, systemic lupus erythematosus, myasthenia gravis, Graves' disease, glomerulonephritis, autoimmune hepatological disorder, inflammatory bowel disease (e.g., Crohn's disease), anaphylaxis, allergic reaction, Sjögren's syndrome, type I diabetes mellitus, primary biliary cirrhosis, Wegener's granulomatosis, fibromyalgia, polymyositis, dermatomyositis, multiple endocrine failure, Schmidt's syndrome, autoimmune uveitis, Addison's disease, adrenalitis, thyroiditis, Hashimoto's thyroiditis, autoimmune thyroid disease
- the autoimmune disease is a disorder of B lymphocytes (e.g., systemic lupus erythematosus, Goodpasture s syndrome, rheumatoid arthritis, and type I diabetes), Th1-lymphocytes (e.g., rheumatoid arthritis, multiple sclerosis, psoriasis, Sjögren's syndrome, Hashimoto's thyroiditis, Graves' disease, primary biliary cirrhosis, Wegener's granulomatosis, tuberculosis, or graft versus host disease), or Th2-lymphocytes (e.g., atopic dermatitis, systemic lupus erythematosus, atopic asthma, rhinoconjunctivitis, allergic rhinitis, Omenn's syndrome, systemic sclerosis, or chronic graft versus host disease).
- disorders involving dendritic cells involve disorders of Th1-lymphocyte
- the amount of the Conjugate administered ranges from about 0.01 to about 10 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.01 to about 5 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.05 to about 5 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.1 to about 5 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.1 to about 4 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.05 to about 3 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.1 to about 3 mg/kg per dose. In some embodiments, the amount of the Conjugate administered ranges from about 0.1 to about 2 mg/kg per dose.
- a reference to carboxylic acid also includes the anionic (carboxylate) form (—COO ⁇ ), a salt or solvate thereof, as well as conventional protected forms.
- a reference to an amino group includes the protonated form (—N + HR 1 R 2 ), a salt or solvate of the amino group, for example, a hydrochloride salt, as well as conventional protected forms of an amino group.
- a reference to a hydroxyl group also includes the anionic form (—O ⁇ ), a salt or solvate thereof, as well as conventional protected forms.
- a corresponding salt of the active compound for example, a pharmaceutically-acceptable salt.
- a pharmaceutically-acceptable salt examples are discussed in Berge, et al., J. Pharm. Sci., 66, 1-19 (1977).
- a salt may be formed with a suitable cation.
- suitable inorganic cations include, but are not limited to, alkali metal ions such as Na + and K + , alkaline earth cations such as Ca 2+ and Mg 2+ , and other cations such as Al +3 .
- suitable organic cations include, but are not limited to, ammonium ion (i.e. NH 4 + ) and substituted ammonium ions (e.g. NH 3 R + , NH 2 R 2 + , NHR 3 + , NR 4 + ).
- Examples of some suitable substituted ammonium ions are those derived from: ethylamine, diethylamine, dicyclohexylamine, triethylamine, butylamine, ethylenediamine, ethanolamine, diethanolamine, piperazine, benzylamine, phenylbenzylamine, choline, meglumine, and tromethamine, as well as amino acids, such as lysine and arginine.
- An example of a common quaternary ammonium ion is N(CH 3 ) 4 + .
- a salt may be formed with a suitable anion.
- suitable inorganic anions include, but are not limited to, those derived from the following inorganic acids: hydrochloric, hydrobromic, hydroiodic, sulfuric, sulfurous, nitric, nitrous, phosphoric, and phosphorous.
- Suitable organic anions include, but are not limited to, those derived from the following organic acids: 2-acetyoxybenzoic, acetic, ascorbic, aspartic, benzoic, camphorsulfonic, cinnamic, citric, edetic, ethanedisulfonic, ethanesulfonic, fumaric, glucheptonic, gluconic, glutamic, glycolic, hydroxymaleic, hydroxynaphthalene carboxylic, isethionic, lactic, lactobionic, lauric, maleic, malic, methanesulfonic, mucic, oleic, oxalic, palmitic, pamoic, pantothenic, phenylacetic, phenylsulfonic, propionic, pyruvic, salicylic, stearic, succinic, sulfanilic, tartaric, toluenesulfonic, and valeric.
- solvate is used herein in the conventional sense to refer to a complex of solute (e.g. active compound, salt of active compound) and solvent. If the solvent is water, the solvate may be conveniently referred to as a hydrate, for example, a mono-hydrate, a di-hydrate, a tri-hydrate, etc.
- the invention includes compounds where a solvent adds across the imine bond of the PBD moiety, which is illustrated below where the solvent is water or an alcohol (R A OH, where R A is C 1-4 alkyl):
- carbinolamine and carbinolamine ether forms of the PBD can be called the carbinolamine and carbinolamine ether forms of the PBD.
- the balance of these equilibria depend on the conditions in which the compounds are found, as well as the nature of the moiety itself.
- Certain compounds may exist in one or more particular geometric, optical, enantiomeric, diasteriomeric, epimeric, atropic, stereoisomeric, tautomeric, conformational, or anomeric forms, including but not limited to, cis- and trans-forms; E- and Z-forms; c-, t-, and r-forms; endo- and exo-forms; R-, S-, and meso-forms; D- and L-forms; d- and l-forms: (+) and ( ⁇ ) forms; keto-, enol-, and enolate-forms; syn- and anti-forms; synclinal- and anticlinal-forms; ⁇ - and ⁇ -forms; axial and equatorial forms; boat-, chair-, twist-, envelope-, and halfchair-forms; and combinations thereof, hereinafter collectively referred to as “isomers” (or “isomeric forms”).
- isomers are structural (or constitutional) isomers (i.e. isomers which differ in the connections between atoms rather than merely by the position of atoms in space).
- a reference to a methoxy group, —OCH 3 is not to be construed as a reference to its structural isomer, a hydroxymethyl group, —CH 2 OH.
- a reference to ortho-chlorophenyl is not to be construed as a reference to its structural isomer, meta-chlorophenyl.
- a reference to a class of structures may well include structurally isomeric forms falling within that class (e.g. C 1-7 alkyl includes n-propyl and iso-propyl; butyl includes n-, iso-, sec-, and tert-butyl; methoxyphenyl includes ortho-, meta-, and para-methoxyphenyl).
- C 1-7 alkyl includes n-propyl and iso-propyl
- butyl includes n-, iso-, sec-, and tert-butyl
- methoxyphenyl includes ortho-, meta-, and para-methoxyphenyl
- keto/enol (illustrated below), imine/enamine, amide/imino alcohol, amidine/amidine, nitroso/oxime, thioketone/enethiol, N-nitroso/hyroxyazo, and nitro/aci-nitro.
- H may be in any isotopic form, including 1 H, 2 H (D), and 3 H (T); C may be in any isotopic form, including 12 C, 13 C, and 14 C; O may be in any isotopic form, including 16 O and 18 O; and the like.
- a reference to a particular compound includes all such isomeric forms, including (wholly or partially) racemic and other mixtures thereof.
- Methods for the preparation (e.g. asymmetric synthesis) and separation (e.g. fractional crystallisation and chromatographic means) of such isomeric forms are either known in the art or are readily obtained by adapting the methods taught herein, or known methods, in a known manner.
- R 2 and R 12 represent the C2 aromatic groups of the compounds of the present invention, as shown in the table below:
- Prot N is a nitrogen protecting group for synthesis and Prot O is a protected oxygen group for synthesis or an oxo group, by deprotecting the imine bond by standard methods.
- the compound produced may be in its carbinolamine or carbinolamine ether form depending on the solvents used.
- Prot N is Troc and Prot O is an oxygen protecting group for synthesis
- the deprotection is carried out using a Cd/Pb couple to yield the compound of the present invention.
- Prot N is SEM, or an analogous group
- Prot O is an oxo group
- the oxo group can be removed by reduction, which leads to a protected carbinolamine intermediate, which can then be treated to remove the SEM protecting group, followed by the elimination of water.
- the reduction of the compound of Formula 2 can be accomplished by, for example, superhydride or lithium tetraborohydride, whilst a suitable means for removing the SEM protecting group is treatment with silica gel.
- R 2 , Prot N and Prot O are as defined for compounds of formula 2, by coupling an organometallic derivative comprising R 12 , such as an organoboron derivative.
- organoboron derivative may be a boronate or boronic acid.
- R 12 , Prot N and Prot O are as defined for compounds of formula 2, by coupling an organometallic derivative comprising R 2 , such as an organoboron derivative.
- organoboron derivative may be a boronate or boronic acid.
- Prot N and Prot O are as defined for compounds of formula 2, by coupling about a single equivalent (e.g. 0.9 or 1 to 1.1 or 1.2) of an organometallic derivative, such as an organoboron derivative, comprising R 2 or R 12 .
- an organometallic derivative such as an organoboron derivative
- the couplings described above are usually carried out in the presence of a palladium catalyst, for example Pd(PPh 3 ) 4 , Pd(OCOCH 3 ) 2 , PdCl 2 , Pd 2 (dba) 3 .
- a palladium catalyst for example Pd(PPh 3 ) 4 , Pd(OCOCH 3 ) 2 , PdCl 2 , Pd 2 (dba) 3 .
- the coupling may be carried out under standard conditions, or may also be carried out under microwave conditions.
- the two coupling steps are usually carried out sequentially. They may be carried out with or without purification between the two steps. If no purification is carried out, then the two steps may be carried out in the same reaction vessel. Purification is usually required after the second coupling step. Purification of the compound from the undesired by-products may be carried out by column chromatography or ion-exchange separation.
- R 10 and R 10′ are H and R 11 and R 11′ are SO z M
- R 10 and R 11 form a nitrogen-carbon double bond between the nitrogen and carbon atoms to which they are bound, by the addition of the appropriate bisulphite salt or sulphinate salt, followed by an appropriate purification step. Further methods are described in GB 2 053 894, which is herein incorporated by reference.
- the compounds of Formula 2 are used in the synthesis of the drug linker compounds.
- the removal of the N10/C11 protecting groups may occur during the synthesis of the drug linker compounds.
- Nitrogen protecting groups for synthesis are well known in the art.
- the protecting groups of particular interest are carbamate nitrogen protecting groups and hemi-aminal nitrogen protecting groups.
- Carbamate nitrogen protecting groups have the following structure:
- R′ 10 is R as defined above.
- suitable groups are described on pages 503 to 549 of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3 rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
- Particularly preferred protecting groups include Troc, Teoc, Fmoc, BOC, Doc, Hoc, TcBOC, 1-Adoc and 2-Adoc.
- nitrobenzyloxycarbonyl e.g. 4-nitrobenzyloxycarbonyl
- 2-(phenylsulphonyl)ethoxycarbonyl e.g. 4-nitrobenzyloxycarbonyl
- nitrobenzyloxycarbonyl e.g. 4-nitrobenzyloxycarbonyl
- 2-(phenylsulphonyl)ethoxycarbonyl e.g. 4-nitrobenzyloxycarbonyl
- Hemi-aminal nitrogen protecting groups have the following structure:
- R′ 10 is R as defined above.
- suitable groups are described on pages 633 to 647 as amide protecting groups of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3 rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
- the groups disclosed herein can be applied to compounds of the present invention. Such groups include, but are not limited to, SEM, MOM, MTM, MEM, BOM, nitro or methoxy substituted BOM, Cl 3 CCH 2 OCH 2 —.
- Protected Oxygen Group for Synthesis include, but are not limited to, SEM, MOM, MTM, MEM, BOM, nitro or methoxy substituted BOM, Cl 3 CCH 2 OCH 2 —.
- Classes of particular interest include silyl ethers, methyl ethers, alkyl ethers, benzyl ethers, esters, acetates, benzoates, carbonates, and sulfonates.
- Preferred oxygen protecting groups include acetates, TBS and THP.
- Conjugates comprising PBD dimers as described herein can be prepared using the knowledge of the skilled artisan in combination with the teachings provided herein.
- linkers are described in U.S. Pat. No. 6,214,345, U.S. Pat. No. 7,498,298 as well as WO 2009/0117531, each of which is incorporated herein by reference in their entirety and for all purposes.
- Other linkers can be prepared according to the references cited herein or as known to the skilled artisan.
- Linker-Drug compounds can be prepared according to methods known in the art in combination with the teachings provided herein. For example, linkage of amine-based X substituents (of the PBD dimer Drug unit) to active groups of the Linker units can be performed according to methods generally described in U.S. Pat. Nos. 6,214,345 and 7,498,298; and WO 2009-0117531, or as otherwise known to the skilled artisan.
- Antibodies can be conjugated to Linker-Drug compounds as described in Doronina et al., Nature Biotechnology, 2003, 21, 778-784). Briefly, antibodies (4-5 mg/mL) in PBS containing 50 mM sodium borate at pH 7.4 are reduced with tris(carboxyethyl)phosphine hydrochloride (TCEP) at 37° C. The progress of the reaction, which reduces interchain disulfides, is monitored by reaction with 5,5′-dithiobis(2-nitrobenzoic acid) and allowed to proceed until the desired level of thiols/mAb is achieved. The reduced antibody is then cooled to 0° C. and alkylated with 1.5 equivalents of maleimide drug-linker per antibody thiol.
- TCEP tris(carboxyethyl)phosphine hydrochloride
- the reaction is quenched by the addition of 5 equivalents of N-acetyl cysteine. Quenched drug-linker is removed by gel filtration over a PD-10 column. The ADC is then sterile-filtered through a 0.22 ⁇ m syringe filter. Protein concentration can be determined by spectral analysis at 280 nm and 329 nm, respectively, with correction for the contribution of drug absorbance at 280 nm. Size exclusion chromatography can be used to determine the extent of antibody aggregation, and RP-HPLC can be used to determine the levels of remaining NAC-quenched drug-linker.
- Antibodies with introduced cysteine residues can be conjugated to Linker-Drug compounds as described in International Patent Publication WO2008/070593, which is incorporated herein by reference in its entirety and for all purposes or as follows.
- Antibodies containing an introduced cysteine residue in the heavy chain are fully reduced by adding 10 equivalents of TCEP and 1 mM EDTA and adjusting the pH to 7.4 with 1M Tris buffer (pH 9.0). Following a 1 hour incubation at 37° C., the reaction is cooled to 22° C. and 30 equivalents of dehydroascorbic acid is added to selectively reoxidize the native disulfides, while leaving the introduced cysteine in the reduced state.
- the pH is adjusted to 6.5 with 1M Tris buffer (pH 3.7) and the reaction is allowed to proceed for 1 hour at 22° C.
- the pH of the solution is then raised again to 7.4 by addition of 1 M Tris buffer (pH 9.0).
- 3.5 equivalents of the PBD drug linker in DMSO is placed in a suitable container for dilution with propylene glycol prior to addition to the reaction.
- the antibody itself is first diluted with propylene glycol to a final concentration of 33% (e.g., if the antibody solution was in a 60 mL reaction volume, 30 mL of propylene glycol was added).
- This same volume of propylene glycol (30 mL in this example) is added to the PBD drug linker as a diluent. After mixing, the solution of PBD drug linker in propylene glycol is added to the antibody solution to effect the conjugation; the final concentration of propylene glycol is 50%. The reaction is allowed to proceed for 30 minutes and then quenched by addition of 5 equivalents of N-acetyl cysteine. The ADC is purified by ultrafiltration through a 30 kD membrane. (Note that the concentration of propylene glycol used in the reaction can be reduced for any particular PBD, as its sole purpose is to maintain solubility of the drug linker in the aqueous media.)
- conjugation can be performed generally as follows. To a solution of reduced and reoxidized antibodies (having introduced cysteines in the heavy chain) in 10 mM Tris (pH 7.4), 50 mM NaCl, and 2 mM DTPA is added 0.5 volumes of propylene glycol. A 10 mM solution of acetamide-based Linker-Drug compound in dimethylacetamide is prepared immediately prior to conjugation. An equivalent amount of propylene glycol as added to the antibody solution is added to a 6-fold molar excess of the Linker-Drug compound.
- the dilute Linker-Drug solution is added to the antibody solution and the pH is adjusted to 8-8.5 using 1 M Tris (pH 9).
- the conjugation reaction is allowed to proceed for 45 minutes at 37° C.
- the conjugation is verified by reducing and denaturing reversed phase PLRP-S chromatography.
- Excess Linker-Drug compound is removed with Quadrasil MP resin and the buffer is exchanged into 10 mM Tris (pH 7.4), 50 mM NaCl, and 5% propylene glycol using a PD-10 desalting column.
- R 2 ′ represents the part of R 2 (as defined above) which links the PBD core to the NH 2 group (for compound C1, the NH 2 group is replaced by NHMe).
- n is as defined above.
- the glucuronide linker intermediate S1 (reference: Jeffrey et al., Bioconjugate Chemistry, 2006, 17, 831-840) can be treated with diphosgene in dichloromethane at ⁇ 78° C. to afford the glucuronide chloroformate, which is then reacted with the PBD dimer S2 dissolved in CH 2 Cl 2 by dropwise addition. Warming the reaction to 0° C. over 2 hours followed by extraction will yield the compound S3. Treating a solution of S3 in an equal solvent mixture of MeOH, tetrahydrofuran, and water (cooled to 0° C.) with lithium hydroxide monohydrate for 4 hours, followed by reaction with glacial acetic acid will yield the compound S4. Adding maleimidocaproyl NHS ester to a solution of S4 in DMF, followed by diisopropylethylamine and stirring at room temperature under nitrogen for 2 hours will yield the desired drug linker S5.
- R 10 is carbamate nitrogen protecting group, it may preferably be Teoc, Fmoc and Troc, and may more preferably be Troc.
- Prot O may preferably be TBS or THP, and may more preferably be TBS.
- R 10 is a hemi-aminal nitrogen protecting group, it may preferably be MOM, BOM or SEM, and may more preferably be SEM.
- Conjugates of the present invention include, for example, those of the formula: CBA-A 1 -L 1 -*
- Conjugates of the present invention include, for example, those of the formula: CBA-A 1 -L 1 -*
- Preferred conjugates of the present invention include any of those described in (a) and (b) wherein A 1 is
- Preferred conjugates of the present invention include those wherein the Linker Unit is
- the wavy line indicates the point of attachment to the Ligand unit (e.g., antibody) and the asterisk indicates the point of attachment to D.
- connection between the antibody and the Linker unit is formed between a thiol group of a cysteine residue of the antibody and a maleimide group of the Linker unit.
- the antibody is a monoclonal antibody that specifically binds to the Cripto antigen, CD19 antigen, CD20 antigen, CD22 antigen, CD30 antigen, CD33 antigen, Glycoprotein NMB, CanAg antigen, Her2 (ErbB2/Neu) antigen, CD56 (NCAM) antigen, CD70 antigen, CD79 antigen, CD138 antigen, PSCA, PSMA (prostate specific membrane antigen), BCMA, E-selectin, EphB2, Melanotransferin, Muc16 antigen or TMEFF2 antigen.
- the Cripto antigen CD19 antigen, CD20 antigen, CD22 antigen, CD30 antigen, CD33 antigen, Glycoprotein NMB, CanAg antigen, Her2 (ErbB2/Neu) antigen, CD56 (NCAM) antigen, CD70 antigen, CD79 antigen, CD138 antigen, PSCA, PSMA (prostate specific membrane antigen), BCMA, E-selectin, EphB2, Mel
- Optical rotations were measured on an ADP 220 polarimeter (Bellingham Stanley Ltd.) and concentrations (c) are given in g/100 mL. Melting points were measured using a digital melting point apparatus (Electrothermal). IR spectra were recorded on a Perkin-Elmer Spectrum 1000 FT IR Spectrometer. 1 H and 13 C NMR spectra were acquired at 300 K using a Bruker Avance NMR spectrometer at 400 and 100 MHz, respectively.
- Waters Micromass ZQ parameters used were: Capillary (kV), 3.38; Cone (V), 35; Extractor (V), 3.0; Source temperature (° C.), 100; Desolvation Temperature (° C.), 200; Cone flow rate (L/h), 50; De-solvation flow rate (L/h), 250.
- HRMS High-resolution mass spectroscopy
- HRMS High-resolution mass spectroscopy
- TLC Thin Layer Chromatography
- Tetrakis-tris-phenylphosphine palladium complex 38 mg, 3.23 ⁇ 10 ⁇ 5 mol, 0.02 eq was added to a stirred, degassed mixture of 1,1′-[[(Pentane-1,5-diyl)dioxy]bis(11aS)-7-methoxy-2-[[(trifluoromethyl)sulfonyl]oxy]-10-((2-(trimethylsilyl)ethoxy)methyl)-1,10,11,11a-tetrahydro-5H-pyrrolo[2,1-c][1,4]-benzodiazepin-5,11-dione](1)(Compound 8b in WO 2010/043880) (185 mg, 1.62 mmol, 1.0 eq.), 4-methoxyphenyl boronic acid (234 mg, 1.54 mmol, 0.95 eq.) and Na 2 CO 3 (274 mg, 2.59 mmol, 1.6 eq.) in toluene
- Tetrakis-tris-phenylphosphine palladium complex (30 mg, 2.5 ⁇ 10 ⁇ 5 mol, 0.04 eq) was added to a stirred, degassed mixture of the methoxy triflate (2) (700 mg, 0.63 mmol, 1.0 eq.), 4-aminomethylphenyl boronic acid (190 mg, 1.015 mmol, 1.6 eq.) and Na 2 CO 3 (303 mg, 2.85 mmol, 4.5 eq.) in toluene/ethanol/water (20 mL/10 mL/10 mL). The reaction mixture was heated at 75° C. under an argon atmosphere for 3 hours.
- Analytical thin layer chromatography was performed on silica gel 60 F254 aluminum sheets (EMD Chemicals, Gibbstown, N.J.). Radial chromatography was performed on Chromatotron apparatus (Harris Research, Palo Alto, Calif.). Analytical HPLC was performed on a Varian ProStar 210 solvent delivery system configured with a Varian ProStar 330 PDA detector. Samples were eluted over a C12 Phenomenex Synergi 2.0 ⁇ 150 mm, 4 ⁇ m, 80 ⁇ reverse-phase column.
- the acidic mobile phase consisted of acetonitrile and water both containing either 0.05% trifluoroacetic acid or 0.1% formic acid (denoted for each compound).
- LC-MS was performed on a ZMD Micromass mass spectrometer interfaced to an HP Agilent 1100 HPLC instrument equipped with a C12 Phenomenex Synergi 2.0 ⁇ 150 mm, 4 ⁇ m, 80 ⁇ reverse phase column.
- Preparative HPLC was carried out on a Varian ProStar 210 solvent delivery system configured with a Varian ProStar 330 PDA detector. Products were purified over a C12 Phenomenex Synergi 10.0 ⁇ 250 mm, 4 ⁇ m, 80 ⁇ reverse phase column eluting with 0.1% formic acid in water (solvent A) and 0.1% formic acid in acetonitrile (solvent B).
- the purification method consisted of the following gradient of solvent A to solvent B: 90:10 from 0 to 5 min; 90:10 to 10:90 from 5 min to 80 min; followed by isocratic 10:90 for 5 min.
- the flow rate was 4.6 mL/min with monitoring at 254 nm.
- NMR spectral data were collected on a Varian Mercury 400 MHz spectrometer. Coupling constants (J) are reported in hertz.
- the combined organics were washed with brine, dried over sodium sulfate, and concentrated to dryness.
- the residue obtained was dissolved in chloroform (0.75 mL), ethanol (2 mL, and water (0.3 mL), and 800 mg of silica gel was added, providing a thick slurry that was sealed and stirred at room temperature for four days. TLC analysis at that time revealed conversion to imine 5.
- the silica gel was then filtered and washed multiple times with 10% methanol in chloroform until no further PBD absorbance was observed in the filtrate.
- the combined organics were then washed with brine, dried over sodium sulfate, and concentrated to dryness.
- the acidic mobile phase consisted of acetonitrile and water both containing either 0.05% trifluoroacetic acid or 0.1% formic acid (denoted for each compound).
- LC-MS was performed on a ZMD Micromass mass spectrometer interfaced to an HP Agilent 1100 HPLC instrument equipped with a C12 Phenomenex Synergi 2.0 ⁇ 150 mm, 4 ⁇ m, 80 ⁇ reverse phase column.
- Preparative HPLC was carried out on a Varian ProStar 210 solvent delivery system configured with a Varian ProStar 330 PDA detector. Products were purified over a C12 Phenomenex Synergi 10.0 ⁇ 250 mm, 4 ⁇ m, 80 ⁇ reverse phase column eluting with 0.1% formic acid in water (solvent A) and 0.1% formic acid in acetonitrile (solvent B).
- the purification method consisted of the following gradient of solvent A to solvent B: 90:10 from 0 to 5 min; 90:10 to 10:90 from 5 min to 80 min; followed by isocratic 10:90 for 5 min.
- the flow rate was 4.6 mL/min with monitoring at 254 nm.
- NMR spectral data were collected on a Varian Mercury 400 MHz spectrometer. Coupling constants (J) are reported in hertz.
- the reaction was quenched through the addition of water (3 mL) and allowed to warm to room temperature, then diluted with brine (25 mL) and extracted three times with dichloromethane (25 mL). The combined organics were washed with brine (25 mL), dried over sodium sulfate, and evaporated to dryness. The residue was dissolved in a mixture of dichloromethane (2.4 mL), ethanol (6.2 mL), and water (0.9 mL), and silica gel (2.4 g) was added. The resulting slurry was stirred at room temperature for 3 days.
- the reaction was quenched through the addition of water (1 mL) and allowed to warm to room temperature, then diluted with brine (25 mL) and extracted three times with dichloromethane (25 mL). The combined organics were washed with brine (25 mL), dried over sodium sulfate, and evaporated to dryness.
- the SEM carbinolamine 10 (39 mg) was carried forward without further purification.
- Activated glucuronide linker 11 (Jeffrey et al., Bioconjugate Chemistry, 2006, 17, 831-840) (38 mg, 42 ⁇ mol, 1.1 eq) was dissolved in anhydrous dimethylformamide (0.6 mL) and added to a flask containing 10 (39 mg, 37 ⁇ mol, 1 eq). Diisopropylethylamine (13 ⁇ L, 74 ⁇ mol, 2 eq) was added and the reaction was stirred at room temperature under nitrogen; after 1.5 hours LC-MS revealed conversion to the coupled product. The reaction was diluted with brine (25 mL) and extracted three times with dichloromethane (25 mL).
- Protected glucuronide linker 12 (25 mg, 16 ⁇ mol, 1 eq) was dissolved in methanol (0.5 mL) and tetrahydrofuran (0.5 mL), and the cooled to 0° C. Lithium hydroxide monohydrate (4 mg, 96 ⁇ mol, 6 eq) was dissolved in water (0.5 mL) and added dropwise to the reaction, which was then allowed to warm to room temperature and monitored by LC-MS. Additional LiOH (3.2 mg, 76 ⁇ mol, 4.8 eq) in 0.4 mL of water was added to the reaction after 2 h to further drive conversion to product.
- glucuronide linker 13 (2 mg, 1.7 ⁇ mol, 1 eq), maleimidocaproic acid N-hydroxysuccinimide ester (0.8 mg, 2.6 ⁇ mol, 1.5 eq), and anhydrous dimethylformamide (85 ⁇ L).
- Diisopropylethylamine (1.5 uL, 8.5 ⁇ mol, 5 eq) was added, the reaction was then stirred at room temperature under nitrogen. After 2 hours HPLC revealed conversion to product. The reaction was diluted in dimethylsulfoxide and purified by preparative HPLC to provide PBD glucuronide linker 14 (1.4 mg, 61%).
- LC-MS t R 12.30 min, m/z (ES + ) found 1373.7 (M+H) + .
- the reaction was quenched through the addition of water (1 mL) and allowed to warm to room temperature, then diluted brine (25 mL) and extracted three times with dichloromethane (25 mL). The combined organics were washed with brine (25 mL), dried over sodium sulfate, and evaporated to dryness. The residue was dissolved in a mixture of dichloromethane (1.2 mL), ethanol (3.2 mL), and water (0.5 mL), and silica gel (1.6 g) was added. The resulting slurry was stirred at room temperature for 4 days.
- a flame-dried flask was charged with maleimidocaproic acid (5.2 mg, 25 ⁇ mol, 1.5 eq) dissolved in 0.33 mL of 5% methanol in anhydrous dichloromethane.
- the acid was pre-activated by addition of N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (7.3 mg, 30 ⁇ mol, 1.8 eq), followed by stirring at room temperature under nitrogen for 15 minutes.
- the activated acid was then added to a flame-dried flask containing PBD dimer 17 (13.5 mg, 16 ⁇ mol. 1 eq). The reaction was stirred for 4 h at room temperature under nitrogen, at which time LC-MS revealed conversion to product.
- a flame-dried flask was charged with maleimidocaproyl-valine-alanine linker (Compound 36 in Example 13 of WO 2011/130613 A1) (9 mg, 24 ⁇ mol, 1.5 eq) dissolved in 0.33 mL of 5% methanol in anhydrous dichloromethane.
- the acid was pre-activated by addition of N-ethoxycarbonyl-2-ethoxy-1,2-dihydroquinoline (7.1 mg, 29 ⁇ mol, 1.8 eq), followed by stirring at room temperature under nitrogen for 15 minutes.
- the activated acid was then added to a flame-dried flask containing PBD dimer 17 (13 mg, 16 ⁇ mol, 1 eq).
- Antibodies with introduced cysteines Antibodies to CD70 containing a cysteine residue at position 239 of the heavy chain were fully reduced by adding 10 equivalents of TCEP and 1 mM EDTA and adjusting the pH to 7.4 with 1M Tris buffer (pH 9.0). Following a 1 hour incubation at 37° C., the reaction was cooled to 22° C. and 30 equivalents of dehydroascorbic acid were added to selectively reoxidize the native disulfides, while leaving cysteine 239 in the reduced state. The pH was adjusted to 6.5 with 1M Tris buffer (pH 3.7) and the reaction was allowed to proceed for 1 hour at 22° C.
- the pH of the solution was then raised again to 7.4 by addition of 1 M Tris buffer (pH 9.0).
- 3.5 equivalents of the PBD drug linker in DMSO were placed in a suitable container for dilution with propylene glycol prior to addition to the reaction.
- the antibody itself was first diluted with propylene glycol to a final concentration of 33% (e.g., if the antibody solution was in a 60 mL reaction volume, 30 mL of propylene glycol was added). This same volume of propylene glycol (30 mL in this example) was then added to the PBD drug linker as a diluent.
- the solution of PBD drug linker in propylene glycol was added to the antibody solution to effect the conjugation; the final concentration of propylene glycol is 50%.
- the reaction was allowed to proceed for 30 minutes and then quenched by addition of 5 equivalents of N-acetyl cysteine.
- the ADC was then purified by ultrafiltration through a 30 kD membrane. (Note that the concentration of propylene glycol used in the reaction can be reduced for any particular PBD, as its sole purpose is to maintain solubility of the drug linker in the aqueous media.)
- the in vitro cytotoxic activity of the selected antibody drug conjugate was assessed using a resazurin (Sigma, St. Louis, Mo., USA) reduction assay (reference: Doronina et al., Nature Biotechnology, 2003, 21, 778-784).
- the antibody drug conjugate was prepared as described above.
- 96-hour assay cells cultured in log-phase growth were seeded for 24 h in 96-well plates containing 150 ⁇ L RPMI 1640 supplemented with 20% FBS. Serial dilutions of ADC in cell culture media were prepared at 4 ⁇ working concentration; 50 ⁇ L of each dilution was added to the 96-well plates. Following addition of ADC, the cells were incubated with test articles for 4 days at 37° C. Resazurin was then added to each well to achieve a 50 ⁇ M final concentration, and the plates were incubated for an additional 4 hours at 37° C.
- the plates were then read for the extent of dye reduction on a Fusion HT plate reader (Packard Instruments, Meridien, Conn., USA) with excitation and emission wavelengths of 530 and 590 nm, respectively.
- the IC 50 value, determined in triplicate, is defined here as the concentration that results in a 50% reduction in cell growth relative to untreated controls.
- h1F6 is the humanized anti-CD70 antibody described below.
- the antibody used was a CD70 antibody, humanized h1F6 IgG1 (WO2006/113909 incorporated by reference in its entirety and for all purposes), with a point mutation substituting cysteine for serine at position 239. Conjugation to the Drug Unit is through the introduced cysteine at position 239. An average of 2 drugs is loaded per antibody.
- FIG. 1 the results of a treatment study using an h1F6-compound 18 and h1F6-compound 19 conjugate in a CD70+ renal cell carcinoma model are shown. Dosing was carried out at q7d ⁇ 2. In the figure, is untreated, ⁇ is treatment with h1F63c-18 at 0.1 mg/kg, ⁇ is treatment with h1F63c-18 at 0.3 mg/kg, ⁇ is treatment with h1F63c-19 at 1 mg/kg, ⁇ is treatment with h1F63c-19 at 3 mg/kg,
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Cell Biology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Peptides Or Proteins (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Saccharide Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
has recently entered Phase II clinical trials in the oncology area (Gregson, S., et al, J. Med. Chem., 44, 737-748 (2001); Alley, M. C., et al., Cancer Research, 64, 6700-6706 (2004); Hartley, J. A., et al., Cancer Research, 64, 6693-6699 (2004)).
- (a) R10 is H, and R11 is OH or ORA, where RA is saturated C1-4 alkyl; or
- (b) R10 and R11 form a nitrogen-carbon double bond between the nitrogen and carbon atoms to which they are bound; or
- (c) R10 is H and R11 is SOzM, where z is 2 or 3 and M is a monovalent pharmaceutically acceptable cation, or both M together are a divalent pharmaceutically acceptable cation.
- (a) R10 is carbamate nitrogen protecting group, and R11 is O-ProtO, wherein ProtO is an oxygen protecting group; or
- (b) R10 is a hemi-aminal nitrogen protecting group and R11 is an oxo group.
L-(LU-D)p (IV)
or a pharmaceutically acceptable salt or solvate thereof, wherein L is a Ligand unit (i.e., a targeting agent), LU is a Linker unit and D is a Drug unit that is a PBD dimer (see below). The subscript p is from 1 to 20. Accordingly, the Conjugates comprise a Ligand unit covalently linked to at least one Drug unit by a Linker unit. The Ligand unit, described more fully below, is a targeting agent that binds to a target moiety. The Ligand unit can, for example, specifically bind to a cell component (a Cell Binding Agent) or to other target molecules of interest. Accordingly, the present invention also provides methods for the treatment of, for example, various cancers and autoimmune disease. These methods encompass the use of the Conjugates wherein the Ligand unit is a targeting agent that specifically binds to a target molecule. The Ligand unit can be, for example, a protein, polypeptide or peptide, such as an antibody, an antigen-binding fragment of an antibody, or other binding agent, such as an Fc fusion protein.
or a pharmaceutically acceptable salt or solvate thereof, where R10 and R11 are as defined in the first aspect, and the asterix indicates the point of attachment to the Linker Unit.
LU-D (V)
or a pharmaceutically acceptable salt or solvate thereof, wherein LU is a Linker unit and D is a Drug unit that is a PBD dimer, as defined in the sixth aspect of the invention.
- Oxo (keto, -one): ═O.
wherein R′10 is R as defined below. A large number of suitable groups are described on pages 503 to 549 of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
wherein R′10 is R as defined below. A large number of suitable groups are described on pages 633 to 647 as amide protecting groups of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
R
-
- saturated monocyclic hydrocarbon compounds:
cyclopropane (C3), cyclobutane (C4), cyclopentane (C5), cyclohexane (C6), cycloheptane (C7), methylcyclopropane (C4), dimethylcyclopropane (C5), methylcyclobutane (C5), dimethylcyclobutane (C6), methylcyclopentane (C6), dimethylcyclopentane (C7) and methylcyclohexane (C7); - unsaturated monocyclic hydrocarbon compounds:
cyclopropene (C3), cyclobutene (C4), cyclopentene (C5), cyclohexene (C6), methylcyclopropene (C4), dimethylcyclopropene (C5), methylcyclobutene (C5), dimethylcyclobutene (C6), methylcyclopentene (C6), dimethylcyclopentene (C7) and methylcyclohexene (C7); and - saturated polycyclic hydrocarbon compounds:
norcarane (C7), norpinane (C7), norbornane (C7).
- saturated monocyclic hydrocarbon compounds:
- N1: aziridine (C3), azetidine (C4), pyrrolidine (tetrahydropyrrole) (C5), pyrroline (e.g., 3-pyrroline, 2,5-dihydropyrrole) (C5), 2H-pyrrole or 3H-pyrrole (isopyrrole, isoazole) (C5), piperidine (C6), dihydropyridine (C6), tetrahydropyridine (C6), azepine (C7);
- O1: oxirane (C3), oxetane (C4), oxolane (tetrahydrofuran) (C5), oxole (dihydrofuran) (C5), oxane (tetrahydropyran) (C6), dihydropyran (C6), pyran (C6), oxepin (C7);
- S1: thiirane (C3), thietane (C4), thiolane (tetrahydrothiophene) (C5), thiane (tetrahydrothiopyran) (C6), thiepane (C7);
- O2: dioxolane (C5), dioxane (C6), and dioxepane (C7);
- O3: trioxane (C6);
- N2: imidazolidine (C5), pyrazolidine (diazolidine) (C5), imidazoline (C5), pyrazoline (dihydropyrazole) (C5), piperazine (C6);
- N1O1: tetrahydrooxazole (C5), dihydrooxazole (C5), tetrahydroisoxazole (C5), dihydroisoxazole (C5), morpholine (C6), tetrahydrooxazine (C6), dihydrooxazine (C6), oxazine (C6);
- N1S1: thiazoline (C5), thiazolidine (C5), thiomorpholine (C6);
- N2O1: oxadiazine (C6);
- O1S1: oxathiole (C5) and oxathiane (thioxane) (C6); and,
- N1O1S1: oxathiazine (C6).
- N1: pyrrole (azole) (C5), pyridine (azine) (C6);
- O1: furan (oxole) (C5);
- S1: thiophene (thiole) (C5);
- N1O1: oxazole (C5), isoxazole (C5), isoxazine (C6);
- N2O1: oxadiazole (furazan) (C5);
- N3O1: oxatriazole (C5);
- N1S1: thiazole (C6), isothiazole (C5);
- N2: imidazole (1,3-diazole) (C5), pyrazole (1,2-diazole) (C5), pyridazine (1,2-diazine) (C6), pyrimidine (1,3-diazine) (C6) (e.g., cytosine, thymine, uracil), pyrazine (1,4-diazine) (C6);
- N3: triazole (C5), triazine (C6); and,
- N4: tetrazole (C5).
-
- C9 (with 2 fused rings) derived from benzofuran (O1), isobenzofuran (O1), indole (N1), isoindole (N1), indolizine (N1), indoline (N1), isoindoline (N1), purine (N4) (e.g., adenine, guanine), benzimidazole (N2), indazole (N2), benzoxazole (N1O1), benzisoxazole (N1O1), benzodioxole (O2), benzofurazan (N2O1), benzotriazole (N3), benzothiofuran (S1), benzothiazole (N1S1), benzothiadiazole (N2S);
- C10 (with 2 fused rings) derived from chromene (O1), isochromene (O1), chroman (O1), isochroman (O1), benzodioxan (O2), quinoline (N1), isoquinoline (N1), quinolizine (N1), benzoxazine (N1O1), benzodiazine (N2), pyridopyridine (N2), quinoxaline (N2), quinazoline (N2), cinnoline (N2), phthalazine (N2), naphthyridine (N2), pteridine (N4);
- C11 (with 2 fused rings) derived from benzodiazepine (N2);
- C13 (with 3 fused rings) derived from carbazole (N1), dibenzofuran (O1), dibenzothiophene (S1), carboline (N2), perimidine (N2), pyridoindole (N2); and,
- C14 (with 3 fused rings) derived from acridine (N1), xanthene (O1), thioxanthene (S1), oxanthrene (O2), phenoxathiin (O1S1), phenazine (N2), phenoxazine (N1O1), phenothiazine (N1S1), thianthrene (S2), phenanthridine (N1), phenanthroline (N2), phenazine (N2).
L-(A1 a-L1 s-L2 y-D)p (IVa)
-
- or a pharmaceutically acceptable salt or solvate thereof, wherein:
- L is the Ligand unit; and
- -A1 a-L1 s-L2 y- is a Linker unit (LU), wherein:
- -A1- is a Stretcher unit,
- a is 1 or 2,
- -L1- is a Specificity unit,
- s is an integer ranging from 0 to 12,
- -L2- is a Spacer unit,
- y is 0, 1 or 2;
- -D is a PBD dimer; and
- p is from 1 to 20.
-
- or a pharmaceutically acceptable salt or solvate thereof, wherein:
- L is the Ligand unit; and
- -A1 a-L1 s(L2 y)- is a Linker unit (LU), wherein:
- -A1- is a Stretcher unit linked to a Stretcher unit (L2),
- a is 1 or 2,
- -L1- is a Specificity unit linked to a Stretcher unit (L2),
- s is an integer ranging from 0 to 12,
- -L2- is a Spacer unit,
- y is 0, 1 or 2;
- -D is a PBD dimer; and
- p is from 1 to 20.
Preferences
L-(A1 a-L1 s-L2 y-D)p
L-(A1 a-Ls 1-D)p,
L-(A1-L1-D)p, or
L-(A1-D)p
-
- or a pharmaceutically acceptable salt or solvate thereof, wherein L, A1, a, L1, s, L2, D, y and p are as described above.
-
- where the asterisk indicates the point of attachment to the Drug unit (D), CBA is the Cell Binding Agent, L1 is a Specificity unit, A1 is a Stretcher unit connecting L1 to the Cell Binding Agent, L2 is a Spacer unit, which is a covalent bond, a self-immolative group or together with —OC(═O)— forms a self-immolative group, and L2 is optional. —OC(═O)— may be considered as being part of L1 or L2, as appropriate.
CBA-A1 a-L1 s-L2 y-*
-
- where the asterisk indicates the point of attachment to the Drug unit (D), CBA is the Cell Binding Agent, L1 is a Specificity unit, A1 is a Stretcher unit connecting L1 to the Cell Binding Agent, L2 is a Spacer unit which is a covalent bond or a self-immolative group, and a is 1 or 2, s is 0, 1 or 2, and y is 0 or 1 or 2.
-
- where the asterisk indicates the point of attachment to the Drug (D), CBA is the Cell Binding Agent, L1 is a Specificity unit connected to L2, A1 is a Stretcher unit connecting L2 to the Cell Binding Agent, L2 is a self-immolative group, and a is 1 or 2, s is 1 or 2, and y is 1 or 2.
-
- —C(═O)NH—,
- —C(═O)O—,
- —NHC(═O)—,
- —OC(═O)—,
- —OC(═O)O—,
- —NHC(═O)O—,
- —OC(═O)NH—,
- —NHC(═O)NH, and
- —O— (a glycosidic bond).
-
- where the asterisk indicates the point of attachment to the Drug unit, the wavy line indicates the point of attachment to the L1, Y is —N(H)—, —O—, —C(═O)N(H)— or —C(═O)O—, and n is 0 to 3. The phenylene ring is optionally substituted with one, two or three substituents as described herein.
-
- where the asterisk indicates the attachment to the Drug, L* is the activated form of the remaining portion of the linker and the released Drug unit is not shown. These groups have the advantage of separating the site of activation from the Drug.
-
- where the asterisk, the wavy line, Y, and n are as defined above. Each phenylene ring is optionally substituted with one, two or three substituents as described herein. In one embodiment, the phenylene ring having the Y substituent is optionally substituted and the phenylene ring not having the Y substituent is unsubstituted.
-
- where the asterisk, the wavy line, Y, and n are as defined above, E is O, S or NR, D is N, CH, or CR, and F is N, CH, or CR.
-
- -Phe-Lys-,
- -Val-Ala-,
- -Val-Lys-,
- -Ala-Lys-,
- -Val-Cit-,
- -Phe-Cit-,
- -Leu-Cit-,
- -Ile-Cit-,
- -Phe-Arg-, and
- -Trp-Cit-;
where Cit is citrulline. In such a dipeptide, —NH— is the amino group of X1, and CO is the carbonyl group of X2.
-
- -Phe-Lys-,
- -Val-Ala-,
- -Val-Lys-,
- -Ala-Lys-, and
- -Val-Cit-.
-
- -Gly-Gly-,
- -Pro-Pro-, and
- -Val-Glu-.
-
- Arg: Z, Mtr, Tos;
- Asn: Trt, Xan;
- Asp: Bzl, t-Bu;
- Cys: Acm, Bzl, Bzl-OMe, Bzl-Me, Trt;
- Glu: Bzl, t-Bu;
- Gln: Trt, Xan;
- His: Boc, Dnp, Tos, Trt;
- Lys: Boc, Z—Cl, Fmoc, Z;
- Ser: Bzl, TBDMS, TBDPS;
- Thr: Bz;
- Trp: Boc;
- Tyr: Bzl, Z, Z—Br.
-
- where the asterisk indicates the point of attachment to the Drug unit, and the wavy line indicates the point of attachment to the remaining portion of L1 or the point of attachment to A1. Preferably, the wavy line indicates the point of attachment to A1.
-
- where the asterisk and the wavy line are as defined above.
-
- where the asterisk indicates the point of attachment to the Drug unit, the wavy line indicates the point of attachment to A1, Y is a covalent bond or a functional group, and E is a group that is susceptible to cleavage thereby to activate a self-immolative group.
-
- —C(═O)—
- —NH-1
- —O—
- —C(═O)NH—,
- —C(═O)O—,
- —NHC(═O)—,
- —OC(═O)—,
- —OC(═O)O—,
- —NHC(═O)O—,
- —OC(═O)NH—,
- —NHC(═O)NH—,
- —NHC(═O)NH,
- —C(═O)NHC(═O)—,
- SO2, and
- —S—.
-
- where the asterisk indicates the point of attachment to the Drug unit, the wavy line indicates the point of attachment to A, Y is a covalent bond or a functional group and E is glucuronic acid (e.g., (β-glucuronic acid). Y is preferably a functional group selected from —NH—.
-
- where the asterisk indicates the point of attachment to the remainder of L2 or the Drug unit, the wavy line indicates the point of attachment to A1, Y is a covalent bond or a functional group and E is glucuronic acid (e.g., β-glucuronic acid). Y is preferably a functional group selected from —NH—, —CH2—, —O—, and —S—.
In some further embodiments, Y is a functional group as set forth above, the functional group is linked to an amino acid, and the amino acid is linked to the Stretcher unit A1. In some embodiments, amino acid is β-alanine. In such an embodiment, the amino acid is equivalently considered part of the Stretcher unit.
- where the asterisk indicates the point of attachment to the remainder of L2 or the Drug unit, the wavy line indicates the point of attachment to A1, Y is a covalent bond or a functional group and E is glucuronic acid (e.g., β-glucuronic acid). Y is preferably a functional group selected from —NH—, —CH2—, —O—, and —S—.
-
- —C(═O)NH—,
- —C(═O)O—,
- —NHC(═O)—,
- —OC(═O)—,
- —OC(═O)O—,
- —NHC(═O)O—,
- —OC(═O)NH—, and
- —NHC(═O)NH—.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to the remaining portion of A1, L1, L2 or D, and the wavy line indicates the point of attachment to the remaining portion of the Ligand unit. In this embodiment, the S atom is typically derived from the Ligand unit.
-
- where the wavy line indicates the point of attachment to the Ligand unit as before, and the asterisk indicates the bond to the remaining portion of the A1 group, or to L1, L2 or D.
-
- where the wavy line indicates point of attachment to the Ligand unit, and the asterisk indicates the bond to the remaining portion of the A1 group, or to L1, L2 or D.
-
- —C(═O)NH—,
- —C(═O)O—,
- —NHC(═O)—,
- —OC(═O)—,
- —OC(═O)O—.
- —NHC(═O)O—,
- —OC(═O)NH—,
- —NHC(═O)NH—,
- —NHC(═O)NH,
- —C(═O)NHC(═O)—,
- —S—,
- —S—S—,
- —CH2C(═O)—
- —C(═O)CH2—,
- ═N—NH—, and
- —NH—N═.
-
- where the wavy line indicates either the point of attachment to the Ligand unit or the bond to the remaining portion of the A1 group, and the asterisk indicates the other of the point of attachment to the Ligand unit or the bond to the remaining portion of the A1 group.
-
- —C(═O)N<,
- —OC(═O)N<, and
- —NHC(═O)N<,
where N< is part of D.
-
- —C(═O)N<.
-
- -Phe-Lys-,
- -Val-Ala-,
- -Val-Lys-,
- -Ala-Lys-,
- -Val-Cit-,
- -Phe-Cit-,
- -Leu-Cit-,
- -Ile-Cit-,
- -Phe-Arg-, and
- -Trp-Cit-;
where Cit is citrulline. In such a dipeptide, —NH— is the amino group of X1, and CO is the carbonyl group of X2.
-
- -Phe-Lys-,
- -Val-Ala-,
- -Val-Lys-,
- -Ala-Lys-, and
- -Val-Cit-.
-
- -Gly-Gly-,
- -Pro-Pro-, and
- -Val-Glu-.
-
- —NH—X1—X2—CO—N<*
- where —NH—X1—X2—CO is the dipeptide, —N< is part of the Drug unit, the asterisk indicates the points of attachment to the remainder of the Drug unit, and the wavy line indicates the point of attachment to the remaining portion of L1 or the point of attachment to A1. Preferably, the wavy line indicates the point of attachment to A1.
-
- where the asterisks, —N< and the wavy line are as defined above.
-
- where the asterisks, —N< and the wavy line are as defined above.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is I and m is 0 to 10, 1 to 7, preferably 3 to 7, most preferably 3 or 7.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, S is a sulfur group of the Ligand unit, the wavy line indicates the point of attachment to the rest of the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, S is a sulfur group of the Ligand unit, the wavy line indicates the point of attachment to the remainder of the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, S is a sulfur group of the Ligand unit, the wavy line indicates the point of attachment to the remainder of the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 7, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the remainder of the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the remainder of the Ligand unit, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the remainder of the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L2 or D, the wavy line indicates the point of attachment to the remainder of the Ligand unit, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to the remainder of the Stretcher unit, L1 or D, and the wavy line indicates the point of attachment to the Ligand unit.
Linker-Drugs
- where the asterisk indicates the point of attachment to the remainder of the Stretcher unit, L1 or D, and the wavy line indicates the point of attachment to the Ligand unit.
-
- where the asterisk indicates the point of attachment to the Drug unit (D, as defined above), G1 is a Stretcher group (A1) to form a connection to a Ligand unit, L1 is a Specificity unit, L2 (a Spacer unit) is a covalent bond or together with —OC(═O)— forms a self-immolative group(s).
G1-L1-L2-.
-
- where the asterisk indicates the point of attachment to the Drug unit (D), G1 is a Stretcher unit (A1) to form a connection to a Ligand unit, L1 is a Specificity unit, L2 (a Spacer unit) is a covalent bond or a self-immolative group(s).
-
- where the asterisk indicates the point of attachment to L1, L2 or D, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is I and m is 0 to 10, 1 to 2, preferably 4 to 8, and most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L1, L2 or D, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is I and m is 0 to 10, 1 to 8, preferably 4 to 8, and most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, and n is 0 to 6. In one embodiment, n is 5.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 2, preferably 4 to 8, and most preferably 4 or 8.
-
- where the asterisk indicates the point of attachment to L1, L2 or D, n is 0 or 1, and m is 0 to 30. In a preferred embodiment, n is 1 and m is 0 to 10, 1 to 8, preferably 4 to 8, and most preferably 4 or 8.
-
- where the asterisk indicates the bond to the remaining portion of the G group.
-
- where the asterisk indicates the bond to the remaining portion of the G group.
-
- —C(═O)OH,
- —OH,
- —NH2,
- —SH,
- —C(═O)CH2X, where X is Cl, Br or I,
- —CHO,
- —NHNH2
- —C≡CH, and
- —N3(azide).
-
- Alloc, Fmoc, Boc, Troc, Teoc, Cbz and PNZ.
Preferably, where G1 is —NH2, it is protected with an Alloc or Fmoc group.
- Alloc, Fmoc, Boc, Troc, Teoc, Cbz and PNZ.
-
- where the asterisk indicates the point of attachment to the Drug unit, and the wavy line indicates the point of attachment to the remaining portion of the Linker unit, as applicable or the point of attachment to G1. Preferably, the wavy line indicates the point of attachment to G1.
-
- Cripto, CD19, CD20, CD22, CD30, CD33, Glycoprotein NMB, CanAg, Her2 (ErbB2/Neu), CD56 (NCAM), CD70, CD79, CD138, PSCA, PSMA (prostate specific membrane antigen), BCMA, E-selectin, EphB2, Melanotransferin, Muc16 and TMEFF2. In any of the embodiments provided herein, the Ligand unit can be a monoclonal antibody that specifically binds to the Cripto antigen, CD19 antigen, CD20 antigen, CD22 antigen, CD30 antigen, CD33 antigen, Glycoprotein NMB, CanAg antigen, Her2 (ErbB2/Neu) antigen, CD56 (NCAM) antigen, CD70 antigen, CD79 antigen, CD138 antigen, PSCA, PSMA (prostate specific membrane antigen), BCMA, E-selectin, EphB2, Melanotransferin, Muc16 antigen or TMEFF2 antigen.
- a) WO 00/12508 (pages 14 to 30);
- b) WO 2005/023814 (pages 3 to 10);
- c) WO 2004/043963 (pages 28 to 29); and
- d) WO 2005/085251 (
pages 30 to 39).
Synthesis Route
where n is 0 or 1, and R2 and R12 represent the C2 aromatic groups of the compounds of the present invention, as shown in the table below:
where R2, ProtN and ProtO are as defined for compounds of formula 2, by coupling an organometallic derivative comprising R12, such as an organoboron derivative. The organoboron derivative may be a boronate or boronic acid.
where R12, ProtN and ProtO are as defined for compounds of formula 2, by coupling an organometallic derivative comprising R2, such as an organoboron derivative. The organoboron derivative may be a boronate or boronic acid.
where ProtN and ProtO are as defined for compounds of formula 2, by coupling about a single equivalent (e.g. 0.9 or 1 to 1.1 or 1.2) of an organometallic derivative, such as an organoboron derivative, comprising R2 or R12.
wherein R′10 is R as defined above. A large number of suitable groups are described on pages 503 to 549 of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes.
wherein R′10 is R as defined above. A large number of suitable groups are described on pages 633 to 647 as amide protecting groups of Greene, T. W. and Wuts, G. M., Protective Groups in Organic Synthesis, 3rd Edition, John Wiley & Sons, Inc., 1999, which is incorporated herein by reference in its entirety and for all purposes. The groups disclosed herein can be applied to compounds of the present invention. Such groups include, but are not limited to, SEM, MOM, MTM, MEM, BOM, nitro or methoxy substituted BOM, Cl3CCH2OCH2—.
Protected Oxygen Group for Synthesis
CBA-A1-L1-*
-
- where the asterisk indicates the point of attachment to the PBD dimer (D), or the Spacer Unit, CBA is the Cell Binding Agent, L1 is a Specificity unit that is cleavable by the action of an enzyme, and A1 is a Stretcher unit connecting L1 to the Cell Binding Agent.
CBA-A1-L1-*
-
- where the asterisk indicates the point of attachment to the PBD dimer (D), CBA is the Cell Binding Agent, A1 is a Stretcher unit connecting L1 to the Cell Binding Agent and L1 is a Specificity unit that is cleavable by the action of cathepsin, L1 is a dipeptide, L1 is a dipeptide that is cleavable by the action of cathepsin or L1 is a dipeptide selected from -Phe-Lys-, -Val-Ala-, -Val-Lys-, -Ala-Lys-, and -Val-Cit-.
-
- where the asterisk indicates the point of attachment to L1, the wavy line indicates the point of attachment to CBA, and n is 0 to 6 (preferably n is 5).
wherein the wavy line indicates the point of attachment to the Ligand unit (e.g., antibody) and the asterisk indicates the point of attachment to D.
| TABLE 1 |
| IC50 in pM following 48 hours treatment |
| compound | 786-O | Caki-1 | HL60 | HEL9217 |
| 17 | 7 | 2 | 3 | 2 |
| 23 | 100 | 100 | 40 | 100 |
| TABLE 2 |
| IC50 in pM following 96 hours treatment |
| antigen-negative | ||||
| ADC | drugs/Ab | 786-O | Caki-1 | cell line |
| h1F6ec-6 | 1.8 | 260 | 12 | 28,000 |
| h1F6ec-9 | 1.5 | 120 | 4.9 | 60,000 |
| h1F6ec-14 | 1.8 | 380 | 24 | 20,000 |
| TABLE 3 |
| IC50 in pM following 96 hours treatment |
| antigen-negative | |||||
| ADC | drugs/Ab | 786-O | Caki-1 | UMRC3 | cell line |
| h1F6ec-18 | 1.9 | 540 | 63 | 1000 | 4000 |
| h1F6ec-19 | 1.9 | 13 | 3.8 | 25 | 2000 |
| h1F6ec-24 | 2.0 | 30 | 8 | — | 3000 |
Claims (4)
L-(LU-D)p (IV)
LU-D (V)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/351,168 US9526798B2 (en) | 2011-10-14 | 2012-10-12 | Pyrrolobenzodiazepines and targeted conjugates |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161547192P | 2011-10-14 | 2011-10-14 | |
| PCT/US2012/059867 WO2013055990A1 (en) | 2011-10-14 | 2012-10-12 | Pyrrolobenzodiazepines and targeted conjugates |
| US14/351,168 US9526798B2 (en) | 2011-10-14 | 2012-10-12 | Pyrrolobenzodiazepines and targeted conjugates |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2012/059867 A-371-Of-International WO2013055990A1 (en) | 2011-10-14 | 2012-10-12 | Pyrrolobenzodiazepines and targeted conjugates |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/181,900 Continuation US9713647B2 (en) | 2011-10-14 | 2016-06-14 | Pyrrolobenzodiazepines and targeted conjugates |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20140286970A1 US20140286970A1 (en) | 2014-09-25 |
| US9526798B2 true US9526798B2 (en) | 2016-12-27 |
Family
ID=48082465
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/351,168 Active US9526798B2 (en) | 2011-10-14 | 2012-10-12 | Pyrrolobenzodiazepines and targeted conjugates |
| US15/181,900 Active US9713647B2 (en) | 2011-10-14 | 2016-06-14 | Pyrrolobenzodiazepines and targeted conjugates |
| US15/635,063 Active US10328084B2 (en) | 2011-10-14 | 2017-06-27 | Pyrrolobenzodiazepines and targeted conjugates |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/181,900 Active US9713647B2 (en) | 2011-10-14 | 2016-06-14 | Pyrrolobenzodiazepines and targeted conjugates |
| US15/635,063 Active US10328084B2 (en) | 2011-10-14 | 2017-06-27 | Pyrrolobenzodiazepines and targeted conjugates |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US9526798B2 (en) |
| EP (2) | EP2755642B1 (en) |
| JP (2) | JP6393617B2 (en) |
| CN (2) | CN108164551A (en) |
| CA (1) | CA2850373C (en) |
| ES (2) | ES2945932T3 (en) |
| HK (2) | HK1200091A1 (en) |
| WO (1) | WO2013055990A1 (en) |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10188746B2 (en) | 2014-09-10 | 2019-01-29 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10335497B2 (en) | 2012-10-12 | 2019-07-02 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10392393B2 (en) | 2016-01-26 | 2019-08-27 | Medimmune Limited | Pyrrolobenzodiazepines |
| US10420777B2 (en) | 2014-09-12 | 2019-09-24 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10543279B2 (en) | 2016-04-29 | 2020-01-28 | Medimmune Limited | Pyrrolobenzodiazepine conjugates and their use for the treatment of cancer |
| US10561739B2 (en) | 2010-04-15 | 2020-02-18 | Seattle Genetics Inc. | Targeted pyrrolobenzodiazapine conjugates |
| US10695439B2 (en) | 2016-02-10 | 2020-06-30 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US10799595B2 (en) | 2016-10-14 | 2020-10-13 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11160872B2 (en) | 2017-02-08 | 2021-11-02 | Adc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugates |
| US11352324B2 (en) | 2018-03-01 | 2022-06-07 | Medimmune Limited | Methods |
| US11370801B2 (en) | 2017-04-18 | 2022-06-28 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11517626B2 (en) | 2016-02-10 | 2022-12-06 | Medimmune Limited | Pyrrolobenzodiazepine antibody conjugates |
| US11524969B2 (en) | 2018-04-12 | 2022-12-13 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof as antitumour agents |
| US11612665B2 (en) | 2017-02-08 | 2023-03-28 | Medimmune Limited | Pyrrolobenzodiazepine-antibody conjugates |
| US11649250B2 (en) | 2017-08-18 | 2023-05-16 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
Families Citing this family (127)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB0819095D0 (en) | 2008-10-17 | 2008-11-26 | Spirogen Ltd | Pyrrolobenzodiazepines |
| CN102971329B (en) | 2010-04-15 | 2016-06-29 | 麦迪穆有限责任公司 | Pyrrolobenzodiazepines for treating proliferative disease is tall and erect |
| SG11201400770SA (en) | 2011-09-20 | 2014-04-28 | Spirogen Sarl | Pyrrolobenzodiazepines as unsymmetrical dimeric pbd compounds for inclusion in targeted conjugates |
| ES2945932T3 (en) | 2011-10-14 | 2023-07-10 | Seagen Inc | Pyrrolobenzodiazepines and targeted conjugates |
| EA036202B1 (en) | 2011-10-14 | 2020-10-14 | Сиэтл Дженетикс, Инк. | Pyrrolobenzodiazepines and targeted conjugates |
| US9399073B2 (en) | 2011-10-14 | 2016-07-26 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines |
| EP2751110B1 (en) * | 2011-10-14 | 2017-04-19 | MedImmune Limited | Asymmetrical bis-(5H-Pyrrolo[2,1-c][1,4]benzodiazepin-5-one) derivatives for the treatment of proliferative and autoimmune diseases |
| BR112015008251B1 (en) | 2012-10-12 | 2023-09-26 | Medimmune Limited | PYRROLOBENZODIAZEPINES, CONJUGATES THEREOF, COMPOSITION AND PHARMACEUTICAL COMPOSITION COMPRISING THE CONJUGATES, USE OF THE CONJUGATES FOR THE TREATMENT OF A PROLIFERATIVE DISEASE AND METHOD FOR PREPARING THE CONJUGATES |
| RS57694B1 (en) | 2012-10-12 | 2018-11-30 | Adc Therapeutics Sa | Pyrrolobenzodiazepine - anti-psma antibody conjugates |
| HUE039329T2 (en) | 2012-10-12 | 2018-12-28 | Adc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugates |
| ES2680153T3 (en) | 2012-10-12 | 2018-09-04 | Adc Therapeutics Sa | Anti-PSMA-pyrrolobenzodiazepine antibody conjugates |
| AU2013328673B2 (en) * | 2012-10-12 | 2017-07-13 | Medimmune Limited | Synthesis and intermediates of pyrrolobenzodiazepine derivatives for conjugation |
| MX364327B (en) | 2012-10-12 | 2019-04-23 | Medimmune Ltd | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates. |
| JP6392763B2 (en) | 2012-10-12 | 2018-09-19 | エイディーシー・セラピューティクス・エス・アーAdc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugate |
| ES2660029T3 (en) | 2012-10-12 | 2018-03-20 | Medimmune Limited | Antibody-pyrrolobenzodiazepine conjugates |
| US9353150B2 (en) | 2012-12-04 | 2016-05-31 | Massachusetts Institute Of Technology | Substituted pyrazino[1′,2′:1 ,5]pyrrolo[2,3-b]-indole-1,4-diones for cancer treatment |
| ES2658888T5 (en) | 2012-12-21 | 2021-10-19 | Medimmune Ltd | Pyrrolobenzodiazepines and their conjugates |
| WO2014096365A1 (en) | 2012-12-21 | 2014-06-26 | Spirogen Sàrl | Unsymmetrical pyrrolobenzodiazepines-dimers for use in the treatment of proliferative and autoimmune diseases |
| US9453046B2 (en) | 2013-03-13 | 2016-09-27 | Seattle Genetics, Inc. | Activated carbon filtration for purification of benzodiazepine ADCs |
| KR102401525B1 (en) | 2013-03-13 | 2022-05-24 | 씨젠 인크. | Cyclodextrin and antibody-drug conjugate formulations |
| NZ710745A (en) | 2013-03-13 | 2019-03-29 | Genentech Inc | Pyrrolobenzodiazepines and conjugates thereof |
| JP6340019B2 (en) | 2013-03-13 | 2018-06-06 | メドイミューン・リミテッドMedImmune Limited | Pyrrolobenzodiazepine and its conjugates |
| US10570151B2 (en) | 2013-03-15 | 2020-02-25 | Regeneron Pharmaceuticals, Inc. | Biologically active molecules, conjugates thereof, and therapeutic uses |
| US10208125B2 (en) | 2013-07-15 | 2019-02-19 | University of Pittsburgh—of the Commonwealth System of Higher Education | Anti-mucin 1 binding agents and uses thereof |
| CN105530942B (en) | 2013-08-26 | 2019-10-11 | 瑞泽恩制药公司 | A kind of pharmaceutical composition comprising macrolides diastereomer, preparation method and use |
| US9950078B2 (en) | 2013-10-11 | 2018-04-24 | Medimmune Limited | Pyrrolobenzodiazepine-antibody conjugates |
| GB201317982D0 (en) | 2013-10-11 | 2013-11-27 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| GB201317981D0 (en) | 2013-10-11 | 2013-11-27 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015052532A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US9956299B2 (en) | 2013-10-11 | 2018-05-01 | Medimmune Limited | Pyrrolobenzodiazepine—antibody conjugates |
| KR20240034882A (en) | 2013-10-15 | 2024-03-14 | 씨젠 인크. | Pegylated drug-linkers for improved ligand-drug conjugate pharmacokinetics |
| KR102405762B1 (en) | 2013-12-16 | 2022-06-07 | 제넨테크, 인크. | Peptidomimetic compounds and antibody-drug conjugates thereof |
| KR20240010081A (en) | 2013-12-19 | 2024-01-23 | 씨젠 인크. | Methylene carbamate linkers for use with targeted-drug conjugates |
| JP6632984B2 (en) | 2014-03-11 | 2020-01-22 | リジェネロン・ファーマシューティカルズ・インコーポレイテッドRegeneron Pharmaceuticals, Inc. | Anti-EGFRvIII antibodies and uses thereof |
| RU2711485C2 (en) | 2014-04-11 | 2020-01-17 | МЕДИММЬЮН, ЭлЭлСи | Conjugated compounds containing cysteine-constructed antibodies |
| KR101628872B1 (en) * | 2014-05-28 | 2016-06-09 | 주식회사 레고켐 바이오사이언스 | Compounds comprising self-immolative group |
| EA201790545A1 (en) | 2014-09-12 | 2017-07-31 | Дженентек, Инк. | ANTIBODIES AND IMMUNOCONJUGATES AGAINST HER2 |
| MX2017006770A (en) | 2014-11-25 | 2018-02-09 | Adc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugates. |
| EA201791550A1 (en) | 2015-01-14 | 2017-11-30 | Бристол-Маерс Сквибб Компани | DYMERAS OF BENZODIAZEPINS, THEIR CONJUGATES AND METHODS OF THEIR RECEPTION AND APPLICATION |
| ES2747386T3 (en) | 2015-01-14 | 2020-03-10 | Bristol Myers Squibb Co | Heteroarylene-linked benzodiazepine dimers, conjugates thereof and methods of preparation and use |
| WO2016160615A1 (en) | 2015-03-27 | 2016-10-06 | Regeneron Pharmaceuticals, Inc. | Maytansinoid derivatives, conjugates thereof, and methods of use |
| GB201506411D0 (en) | 2015-04-15 | 2015-05-27 | Bergenbio As | Humanized anti-axl antibodies |
| GB201506402D0 (en) | 2015-04-15 | 2015-05-27 | Berkel Patricius H C Van And Howard Philip W | Site-specific antibody-drug conjugates |
| KR20180019592A (en) | 2015-06-23 | 2018-02-26 | 브리스톨-마이어스 스큅 컴퍼니 | Macrocycline benzodiazepine dimer, conjugate thereof, preparation method and uses |
| MX2017017117A (en) | 2015-07-06 | 2018-03-06 | Regeneron Pharma | Multispecific antigen-binding molecules and uses thereof. |
| MA43345A (en) | 2015-10-02 | 2018-08-08 | Hoffmann La Roche | PYRROLOBENZODIAZEPINE ANTIBODY-DRUG CONJUGATES AND METHODS OF USE |
| JP6412906B2 (en) | 2015-11-03 | 2018-10-24 | 財團法人工業技術研究院Industrial Technology Research Institute | Compound, linker-drug and ligand-drug complex |
| EP3380125A4 (en) | 2015-11-25 | 2019-08-28 | LegoChem Biosciences, Inc. | Conjugates comprising peptide groups and methods related thereto |
| US11173214B2 (en) | 2015-11-25 | 2021-11-16 | Legochem Biosciences, Inc. | Antibody-drug conjugates comprising branched linkers and methods related thereto |
| CN112451682A (en) | 2015-11-25 | 2021-03-09 | 乐高化学生物科学股份有限公司 | Conjugates comprising self-degrading groups and methods relating thereto |
| US11793880B2 (en) | 2015-12-04 | 2023-10-24 | Seagen Inc. | Conjugates of quaternized tubulysin compounds |
| EP4074328A1 (en) | 2015-12-04 | 2022-10-19 | Seagen Inc. | Intermediates of conjugates of quaternized tubulysin compounds |
| GB201521709D0 (en) * | 2015-12-09 | 2016-01-20 | Kings College London And Sec Dep For Health The | PBD Antibacterial agents |
| AU2016366521A1 (en) | 2015-12-11 | 2018-06-21 | Regeneron Pharmaceuticals, Inc. | Methods for reducing or preventing growth of tumors resistant to EGFR and/or ErbB3 blockade |
| KR20180099892A (en) | 2016-01-25 | 2018-09-05 | 리제너론 파아마슈티컬스, 인크. | Maytansinoid derivatives, conjugates thereof, and methods of use |
| EP3433278A4 (en) | 2016-03-25 | 2019-11-06 | Seattle Genetics, Inc. | Process for the preparation of pegylated drug-linkers and intermediates thereof |
| MA45328A (en) | 2016-04-01 | 2019-02-06 | Avidity Biosciences Llc | NUCLEIC ACID-POLYPEPTIDE COMPOSITIONS AND USES THEREOF |
| WO2017190079A1 (en) | 2016-04-28 | 2017-11-02 | Regeneron Pharmaceuticals, Inc. | Methods of making multispecific antigen-binding molecules |
| WO2017197045A1 (en) | 2016-05-11 | 2017-11-16 | Movassaghi Mohammad | Convergent and enantioselective total synthesis of communesin analogs |
| EP3458069B1 (en) | 2016-05-18 | 2023-07-05 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepines and conjugates thereof |
| EP3475284B1 (en) | 2016-06-24 | 2022-11-02 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepines and conjugates thereof |
| TW201811376A (en) | 2016-07-01 | 2018-04-01 | 英商葛蘭素史密斯克藍智慧財產權有限公司 | Antibody-drug conjugates and therapeutic methods using the same |
| BR112019005670A2 (en) | 2016-09-23 | 2019-06-04 | Regeneron Pharma | anti-muc16 antibodies (mucin 16) |
| SG10202102617QA (en) | 2016-09-23 | 2021-04-29 | Regeneron Pharma | Anti-steap2 antibodies, antibody-drug conjugates, and bispecific antigen-binding molecules that bind steap2 and cd3, and uses thereof |
| AU2017343565A1 (en) | 2016-10-10 | 2019-05-30 | Cellerant Therapeutics, Inc. | Isoquinolidinobenzodiazepine (IQB)-1(chloromethyl)-2,3-dihydro-1H-benzo[e]indole (CBI) dimers |
| US10711032B2 (en) | 2016-11-08 | 2020-07-14 | Regeneron Pharmaceuticals, Inc. | Steroids and protein-conjugates thereof |
| TWI782930B (en) | 2016-11-16 | 2022-11-11 | 美商再生元醫藥公司 | Anti-met antibodies, bispecific antigen binding molecules that bind met, and methods of use thereof |
| GB201619490D0 (en) * | 2016-11-17 | 2017-01-04 | Medimmune Ltd | Pyrrolobenzodiazepine conjugates |
| CN110381980A (en) | 2017-01-06 | 2019-10-25 | 艾维迪提生物科学有限责任公司 | The method that nucleic acid-peptide composition and inducing exon are skipped |
| WO2018160683A1 (en) | 2017-02-28 | 2018-09-07 | Seattle Genetics, Inc. | Cysteine mutated antibodies for conjugation |
| US11730822B2 (en) | 2017-03-24 | 2023-08-22 | Seagen Inc. | Process for the preparation of glucuronide drug-linkers and intermediates thereof |
| CA3058360A1 (en) | 2017-03-29 | 2018-10-04 | Legochem Biosciences, Inc. | Pyrrolobenzodiazepine dimer prodrug and ligand-linker conjugate compound of the same |
| KR20190141666A (en) | 2017-04-20 | 2019-12-24 | 에이디씨 테라퓨틱스 에스에이 | Combination Therapy with Anti-AXL Antibody-Drug Conjugates |
| WO2018209239A1 (en) | 2017-05-11 | 2018-11-15 | Massachusetts Institute Of Technology | Potent agelastatin derivatives as modulators for cancer invasion and metastasis |
| CA3063871A1 (en) | 2017-05-18 | 2018-11-22 | Regeneron Pharmaceuticals, Inc. | Cyclodextrin protein drug conjugates |
| WO2018213082A1 (en) | 2017-05-18 | 2018-11-22 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamides and protein conjugates thereof |
| AU2018285562B2 (en) | 2017-06-14 | 2024-01-18 | Adc Therapeutics Sa | Dosage regimes for the administration of an anti-CD19 ADC |
| CA3067829A1 (en) | 2017-06-23 | 2018-12-27 | VelosBio Inc. | Ror1 antibody immunoconjugates |
| GB201711809D0 (en) | 2017-07-21 | 2017-09-06 | Governors Of The Univ Of Alberta | Antisense oligonucleotide |
| GB201714115D0 (en) * | 2017-09-04 | 2017-10-18 | Femtogenix Ltd | Cytotoxic agents |
| TW202404648A (en) | 2017-09-29 | 2024-02-01 | 日商第一三共股份有限公司 | Antibody-pyrrolobenzodiazepine derivative conjugate |
| JP2021500862A (en) | 2017-10-04 | 2021-01-14 | アビディティー バイオサイエンシーズ,インク. | Nucleic acid polypeptide composition and its use |
| US10640508B2 (en) | 2017-10-13 | 2020-05-05 | Massachusetts Institute Of Technology | Diazene directed modular synthesis of compounds with quaternary carbon centers |
| CA3080857A1 (en) | 2017-11-07 | 2019-05-16 | Regeneron Pharmaceuticals, Inc. | Hydrophilic linkers for antibody drug conjugates |
| WO2019096788A1 (en) | 2017-11-14 | 2019-05-23 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11638760B2 (en) | 2017-11-27 | 2023-05-02 | Mersana Therapeutics, Inc. | Pyrrolobenzodiazepine antibody conjugates |
| MX2020005860A (en) | 2017-12-06 | 2020-09-09 | Avidity Biosciences Inc | Compositions and methods of treating muscle atrophy and myotonic dystrophy. |
| JP2021506883A (en) | 2017-12-21 | 2021-02-22 | メルサナ セラピューティクス インコーポレイテッド | Pyrrolobenzodiazepine antibody conjugate |
| KR20200108002A (en) | 2018-01-08 | 2020-09-16 | 리제너론 파마슈티칼스 인코포레이티드 | Steroids and antibody-conjugates thereof |
| MX2020011487A (en) | 2018-04-30 | 2020-12-07 | Regeneron Pharma | Antibodies, and bispecific antigen-binding molecules that bind her2 and/or aplp2, conjugates, and uses thereof. |
| JP2021523147A (en) | 2018-05-09 | 2021-09-02 | レゲネロン ファーマシューティカルス,インコーポレーテッド | Anti-MSR1 antibody and how to use it |
| BR112020022299A2 (en) | 2018-05-09 | 2021-02-23 | Legochem Biosciences, Inc. | compositions and methods related to anti-cd19 antibody-drug conjugates |
| AU2019269685A1 (en) | 2018-05-17 | 2020-12-03 | Regeneron Pharmaceuticals, Inc. | Anti-CD63 antibodies, conjugates, and uses thereof |
| EP3883926A1 (en) | 2018-11-20 | 2021-09-29 | Regeneron Pharmaceuticals, Inc. | Bis-octahydrophenanthrene carboxamide derivatives and protein conjugates thereof for use as lxr agonists |
| IL297818B1 (en) | 2018-12-21 | 2024-05-01 | Avidity Biosciences Inc | Anti-transferrin receptor antibodies and uses thereof |
| EP3897841A1 (en) | 2018-12-21 | 2021-10-27 | Regeneron Pharmaceuticals, Inc. | Rifamycin analogs and antibody-drug conjugates thereof |
| CN118027137A (en) | 2018-12-21 | 2024-05-14 | 里珍纳龙药品有限公司 | Tubulysin and protein-tubulysin conjugates |
| KR20200084802A (en) | 2019-01-03 | 2020-07-13 | 주식회사 레고켐 바이오사이언스 | Pyrrolobenzodiazepine dimer compounds with improved safety and its use |
| BR112021013464A2 (en) | 2019-01-08 | 2021-09-21 | Regeneron Pharmaceuticals, Inc. | TRACELESS BINDERS AND PROTEIN CONJUGATES THEREOF |
| GB201901197D0 (en) | 2019-01-29 | 2019-03-20 | Femtogenix Ltd | G-A Crosslinking cytotoxic agents |
| CN113711036A (en) | 2019-01-30 | 2021-11-26 | 真和制药有限公司 | anti-GAL 3 antibodies and uses thereof |
| CA3128519A1 (en) | 2019-02-21 | 2020-08-27 | Regeneron Pharmaceuticals, Inc. | Methods of treating ocular cancer using anti-met antibodies and bispecific antigen binding molecules that bind met |
| US11535634B2 (en) | 2019-06-05 | 2022-12-27 | Massachusetts Institute Of Technology | Compounds, conjugates, and compositions of epipolythiodiketopiperazines and polythiodiketopiperazines and uses thereof |
| EP3980436A4 (en) | 2019-06-06 | 2023-12-20 | Avidity Biosciences, Inc. | Nucleic acid-polypeptide compositions and uses thereof |
| KR20210028544A (en) | 2019-09-04 | 2021-03-12 | 주식회사 레고켐 바이오사이언스 | Antibody-drug conjugate comprising antibody binding to antibody against human ROR1 and its use |
| KR20220063185A (en) | 2019-09-16 | 2022-05-17 | 리제너론 파마슈티칼스 인코포레이티드 | Radiolabeled MET binding protein for immuno-PET imaging |
| US11814428B2 (en) | 2019-09-19 | 2023-11-14 | Regeneron Pharmaceuticals, Inc. | Anti-PTCRA antibody-drug conjugates and uses thereof |
| WO2021080608A1 (en) | 2019-10-25 | 2021-04-29 | Medimmune, Llc | Branched moiety for use in conjugates |
| EP4093440A1 (en) | 2020-01-24 | 2022-11-30 | Regeneron Pharmaceuticals, Inc. | Protein-antiviral compound conjugates |
| AU2021228225A1 (en) | 2020-02-28 | 2022-09-01 | Regeneron Pharmaceuticals, Inc. | Bispecific antigen binding molecules that bind HER2, and methods of use thereof |
| AU2021237465A1 (en) | 2020-03-19 | 2022-10-13 | Avidity Biosciences, Inc. | Compositions and methods of treating Facioscapulohumeral muscular dystrophy |
| US11446387B2 (en) | 2020-03-27 | 2022-09-20 | Avidity Biosciences, Inc. | Compositions and methods of treating muscle dystrophy |
| JP2023521885A (en) | 2020-04-16 | 2023-05-25 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | Diels-Alder conjugation method |
| KR20230028325A (en) | 2020-06-24 | 2023-02-28 | 리제너론 파마슈티칼스 인코포레이티드 | Tubulysins and protein-tubulin conjugates |
| JP2023534437A (en) | 2020-07-13 | 2023-08-09 | リジェネロン ファーマシューティカルズ,インク. | Camptothecin analogs conjugated to glutamine residues in proteins and uses thereof |
| KR20230070337A (en) | 2020-09-14 | 2023-05-22 | 리제너론 파마슈티칼스 인코포레이티드 | Antibody-drug conjugates containing GLP1 peptidomimetics and uses thereof |
| AU2021366691A1 (en) | 2020-10-22 | 2023-03-23 | Regeneron Pharmaceuticals, Inc. | Anti-FGFR2 antibodies and methods of use thereof |
| EP4370555A1 (en) | 2021-07-13 | 2024-05-22 | TrueBinding, Inc. | Methods of preventing protein aggregation |
| IL307947A (en) | 2021-07-28 | 2023-12-01 | Regeneron Pharma | Protein-antiviral compound conjugates |
| AU2022345098A1 (en) | 2021-09-16 | 2024-04-04 | Avidity Biosciences, Inc. | Compositions and methods of treating facioscapulohumeral muscular dystrophy |
| WO2023122347A2 (en) | 2021-12-23 | 2023-06-29 | Mirecule, Inc. | Compositions for delivery of polynucleotides |
| US20230414775A1 (en) | 2021-12-29 | 2023-12-28 | Regeneron Pharmaceuticals, Inc. | Tubulysins and protein-tubulysin conjugates |
| US20230287138A1 (en) | 2022-01-12 | 2023-09-14 | Regneron Pharmaceuticals, Inc. | Protein-drug conjugates comprising camptothecin analogs and methods of use thereof |
| WO2023137443A1 (en) | 2022-01-14 | 2023-07-20 | Regeneron Pharmaceuticals, Inc. | Verrucarin a derivatives and antibody drug conjugates thereof |
| WO2023173132A1 (en) | 2022-03-11 | 2023-09-14 | Regeneron Pharmaceuticals, Inc. | Anti-glp1r antibody-drug conjugates comprising glp1 peptidomimetics and uses thereof |
| WO2024020164A2 (en) | 2022-07-21 | 2024-01-25 | Firefly Bio, Inc. | Glucocorticoid receptor agonists and conjugates thereof |
| WO2024026474A1 (en) | 2022-07-29 | 2024-02-01 | Regeneron Pharmaceuticals, Inc. | Compositions and methods for transferrin receptor (tfr)-mediated delivery to the brain and muscle |
Citations (112)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3361742A (en) | 1964-12-07 | 1968-01-02 | Hoffmann La Roche | 5-oxo-1h-pyrrolo-[2, 1-c][1, 4]-benzodiazepin-2-crylamides |
| US3523941A (en) | 1967-03-06 | 1970-08-11 | Hoffmann La Roche | Benzodiazepine compounds and process for their preparation |
| US3524849A (en) | 1967-10-27 | 1970-08-18 | Hoffmann La Roche | Process for the preparation of pyrrolo-benzodiazepine acrylamides and intermediates useful therein |
| FR2027356A1 (en) | 1968-12-30 | 1970-09-25 | Fujisawa Pharmaceutical Co | Benzodiazepinone antibiotics |
| GB1299198A (en) | 1968-12-30 | 1972-12-06 | Fujisawa Pharmaceutical Co | Pyrrolobenzodiazepinone derivatives |
| US3794644A (en) | 1969-06-26 | 1974-02-26 | Fujisawa Pharmaceutical Co | Pyrrolo-benzodiazepinone compounds |
| JPS5382792U (en) | 1976-12-10 | 1978-07-08 | ||
| US4185016A (en) | 1977-12-27 | 1980-01-22 | The Green Cross Corporation | Pyrrolo benzodiazepine compounds |
| GB2053894A (en) | 1979-07-17 | 1981-02-11 | Green Cross Corp | Benzodiazepines processes for producing them and compositions containing them |
| JPS57131791U (en) | 1981-02-06 | 1982-08-17 | ||
| US4353827A (en) | 1981-02-27 | 1982-10-12 | Hoffmann-La Roche Inc. | Diazepine derivatives |
| US4382032A (en) | 1981-02-27 | 1983-05-03 | Hoffmann-La Roche Inc. | Diazepine derivatives |
| US4386028A (en) | 1981-02-27 | 1983-05-31 | Hoffmann-La Roche Inc. | Imidazo[1,5a][1,4]benzodiazepines |
| JPS58180487U (en) | 1982-05-28 | 1983-12-02 | 松下電工株式会社 | Light beam alarm assembly |
| US4427588A (en) | 1982-11-08 | 1984-01-24 | Bristol-Myers Company | Process for conversion of oxotomaymycin to tomaymycin |
| US4427587A (en) | 1982-11-10 | 1984-01-24 | Bristol-Myers Company | Total synthesis of antitumor antibiotics BBM-2040A and BBM-2040B |
| FR2586683A1 (en) | 1985-08-29 | 1987-03-06 | Centre Nat Rech Scient | New neothramycin derivatives, process for their preparation and their application as medicaments |
| US4701325A (en) | 1983-02-17 | 1987-10-20 | The Green Cross Corporation | Injectable aqueous solution containing hydrogensulfite and/or sulfite and anticancerous benzodiazepine compound |
| US4923984A (en) | 1988-08-05 | 1990-05-08 | Shionogi & Co., Ltd. | Pyrrolo(1,4)benzodiazepine derivatives |
| WO1992019620A1 (en) | 1991-05-07 | 1992-11-12 | Centre National De La Recherche Scientifique (Cnrs) | Novel derivatives of pyrrolo [1, 4]-benzodiazepines, method of preparation and medicaments containing them |
| WO1993018045A1 (en) | 1992-03-09 | 1993-09-16 | Cancer Research Campaign Technology Limited | Anti-cancer pyrrolobenzodiazepine derivatives |
| WO1995004718A1 (en) | 1993-08-04 | 1995-02-16 | Zeneca Limited | 4-methyleneproline derivatives as fungicides |
| US5418241A (en) | 1992-09-28 | 1995-05-23 | Synthelabo | Piperidine derivatives, their preparation and their application in therapeutics |
| WO2000012508A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
| WO2000012506A3 (en) | 1998-08-27 | 2000-06-29 | Univ Portsmouth | Collections of compounds |
| WO2000003291A9 (en) | 1998-07-08 | 2000-07-27 | E Ink Corp | Methods for achieving improved color in microencapsulated electrophoretic devices |
| WO2000012509B1 (en) | 1998-08-27 | 2000-08-10 | Univ Portsmouth | Collections of compounds |
| WO2000012507A8 (en) | 1998-08-27 | 2000-10-19 | Univ Portsmouth | Pyrrolobenzodiazepines |
| WO2001016104A1 (en) | 1999-08-27 | 2001-03-08 | Spirogen Limited | Cyclopropylindole derivatives |
| US6362331B1 (en) | 2001-03-30 | 2002-03-26 | Council Of Scientific And Industrial Research | Process for the preparation of antitumor agents |
| US6660742B2 (en) | 2000-09-19 | 2003-12-09 | Taiho Pharmaceutical Co. Ltd. | Compositions and methods of the use thereof achiral analogues of CC-1065 and the duocarmycins |
| US6660856B2 (en) | 2002-03-08 | 2003-12-09 | Kaohsiung Medical University | Synthesis of pyrrolo[2,1-c][1,4]benzodiazepine analogues |
| WO2004043963A1 (en) | 2002-11-14 | 2004-05-27 | Spirogen Limited | Pyrrolobenzodiazepines |
| US20040138269A1 (en) | 2002-10-11 | 2004-07-15 | Sugen, Inc. | Substituted pyrroles as kinase inhibitors |
| WO2005023814A1 (en) | 2003-09-11 | 2005-03-17 | Spirogen Limited | Synthesis of protected pyrrolobenzodiazepines |
| US6884799B2 (en) | 2003-03-31 | 2005-04-26 | Council Of Scientific And Industrial Research | Non-cross-linking pyrrolo[2,1-c][1,4]benzodiazepines and process thereof |
| WO2005042535A1 (en) | 2003-10-22 | 2005-05-12 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2005040170A3 (en) | 2003-10-22 | 2005-06-30 | Us Gov Health & Human Serv | Pyrrolobenzodiazepine derivatives, compositions comprising the same and methods related thereto |
| WO2005085251A1 (en) | 2004-03-01 | 2005-09-15 | Spirogen Limited | 11-hydroxy-5h-pyrrolo[2,1-c][1,4]benzodiazepin-5-one derivatives as key intermediates for the preparation of c2 substituted pyrrolobenzodiazepines |
| WO2005085260A1 (en) | 2004-03-09 | 2005-09-15 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2005085250A8 (en) | 2004-03-01 | 2005-10-27 | Spirogen Ltd | C8, c8' linked 5-oxo-1,2,3,11a-tetrahydro-5h-pyrrolo[2,1-c][1,4]benzodiazepine dimers with 1h-pyrrole-dicarboxylic acid amide linkers and oligomeric analogs therof as well as related compounds for the treatment of proliferative diseases |
| WO2005085177A3 (en) | 2004-03-01 | 2005-11-17 | Spirogen Ltd | Biaryl amino acids and their use in dna binding oligomers |
| WO2005082023A3 (en) | 2004-02-23 | 2005-12-22 | Genentech Inc | Heterocyclic self-immolative linkers and conjugates |
| WO2005085259A3 (en) | 2004-03-01 | 2006-01-05 | Spirogen Ltd | Pyrrolobenzodiazepines as key intermediates in the synthesis of dimeric cytotoxic pyrrolobenzodiazepines |
| WO2005110423A3 (en) | 2004-05-13 | 2006-01-19 | Spirogen Ltd | Pyrrolobenzodiazepine therapeutic agents useful in the treatment of leukaemias |
| WO2006111759A1 (en) | 2005-04-21 | 2006-10-26 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2005105113A3 (en) | 2004-04-27 | 2007-02-22 | Sod Conseils Rech Applic | Therapeutic compositions containing at least one pyrrolobenzodiazepine derivative and fludarabine |
| WO2007039752A1 (en) | 2005-10-05 | 2007-04-12 | Spirogen Limited | Alkyl 4- [4- (5-oxo-2, 3, 5, 11a-tetrahyd0-5h-pyrr0l0 [2, 1-c] [1, 4] benzodiazepine-8-yloxy) -butyrylamino]-1h-pyrrole-2-carboxylate derivatives and related compounds for the treatment of a proliferative disease |
| US20070092940A1 (en) | 2004-09-23 | 2007-04-26 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| US20070154906A1 (en) | 2005-10-05 | 2007-07-05 | Spirogen Ltd. | Methods to identify therapeutic candidates |
| EP1813614A1 (en) | 2006-01-25 | 2007-08-01 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives |
| US20070185336A1 (en) | 2004-03-03 | 2007-08-09 | Degussa Ag | Process for preparing n-protected 4-ketoproline derivatives |
| WO2008010101A2 (en) | 2006-07-18 | 2008-01-24 | Sanofi-Aventis | Antagonist antibody against epha2 for the treatment of cancer |
| US20080092940A1 (en) | 2004-12-24 | 2008-04-24 | Showa Denko K.K. | Production Method of Thermoelectric Semiconductor Alloy, Thermoelectric Conversion Module and Thermoelectric Power Generating Device |
| WO2008050140A2 (en) | 2006-10-27 | 2008-05-02 | Spirogen Limited | Compounds for treatment of parasitic infection |
| WO2008047242A3 (en) | 2006-10-19 | 2008-06-26 | Sanofi Aventis | Novel anti-cd38 antibodies for the treatment of cancer |
| US20080213289A1 (en) | 2002-07-31 | 2008-09-04 | Seattle Genetics, Inc. | Recombinant anti-cd30 antibodies and uses thereof |
| WO2008070593A3 (en) | 2006-12-01 | 2008-10-30 | Seattle Genetics Inc | Variant target binding agents and uses thereof |
| WO2009016516A2 (en) | 2007-07-19 | 2009-02-05 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2009060215A1 (en) | 2007-11-09 | 2009-05-14 | Spirogen Limited | Polyamides |
| WO2009060208A1 (en) | 2007-11-09 | 2009-05-14 | Spirogen Limited | Pyrrolobenzodiazepines |
| US20090148942A1 (en) | 2005-04-19 | 2009-06-11 | Mcdonagh Charlotte | Humanized anti-cd70 binding agents and uses thereof |
| WO2010010347A1 (en) | 2008-07-22 | 2010-01-28 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2010043877A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2010043880A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| WO2010091150A1 (en) | 2009-02-05 | 2010-08-12 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| WO2011023883A1 (en) | 2009-08-25 | 2011-03-03 | Sanofi-Aventis | Conjugates of pyrrolo[1,4]benzodiazepine dimers as anticancer agents |
| WO2011038159A2 (en) | 2009-09-24 | 2011-03-31 | Seattle Genetics, Inc. | Dr5 ligand drug conjugates |
| WO2011100227A1 (en) | 2010-02-09 | 2011-08-18 | Bristol-Myers Squibb Company | Benzylpyrrolidinone derivatives as modulators of chemokine receptor activity |
| US20110256157A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines and conjugates thereof |
| WO2011128650A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Intermediates useful for the synthesis of pyrrolobenzodiazepines |
| WO2011130616A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2011130613A1 (en) | 2010-04-15 | 2011-10-20 | Seattle Genetics, Inc. | Targeted pyrrolobenzodiazapine conjugates |
| WO2012112708A1 (en) | 2011-02-15 | 2012-08-23 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives and methods of preparation |
| WO2013041606A1 (en) | 2011-09-20 | 2013-03-28 | Spirogen Sàrl | Pyrrolobenzodiazepines as unsymmetrical dimeric pbd compounds for inclusion in targeted conjugates |
| WO2013053872A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Synthesis method and intermediates useful in the preparation of pyrrolobenzodiazepines |
| WO2013053873A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013055990A1 (en) | 2011-10-14 | 2013-04-18 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| WO2013053871A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013055993A1 (en) | 2011-10-14 | 2013-04-18 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| WO2013055987A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2013164593A1 (en) | 2012-04-30 | 2013-11-07 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013164592A1 (en) | 2012-04-30 | 2013-11-07 | Ucl Business Plc | Pyrrolobenzodiazepines |
| WO2014011518A1 (en) | 2012-07-09 | 2014-01-16 | Genentech, Inc. | Immunoconjugates comprising anti-cd22 antibodies |
| WO2014011519A1 (en) | 2012-07-09 | 2014-01-16 | Genentech, Inc. | Immunoconjugates comprising anti-cd79b antibodies |
| WO2014022679A2 (en) | 2012-08-02 | 2014-02-06 | Genentech, Inc. | Anti-etbr antibodies and immunoconjugates |
| WO2014057074A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014057118A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| WO2014057115A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-her2 antibody conjugates |
| WO2014057072A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Synthesis and intermediates of pyrrolobenzodiazepine derivatives for conjugation |
| WO2014057122A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| WO2014057117A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2014057114A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-psma antibody conjugates |
| WO2014057120A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2014057073A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014057113A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine - anti-psma antibody conjugates |
| WO2014057119A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2014096365A1 (en) | 2012-12-21 | 2014-06-26 | Spirogen Sàrl | Unsymmetrical pyrrolobenzodiazepines-dimers for use in the treatment of proliferative and autoimmune diseases |
| WO2014096368A1 (en) | 2012-12-21 | 2014-06-26 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014130879A2 (en) | 2013-02-22 | 2014-08-28 | Stem Centrx, Inc. | Novel antibody conjugates and uses thereof |
| WO2014140174A1 (en) | 2013-03-13 | 2014-09-18 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014140862A2 (en) | 2013-03-13 | 2014-09-18 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| US20140294868A1 (en) | 2013-03-13 | 2014-10-02 | Genentech, Inc. | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014174111A1 (en) | 2013-04-26 | 2014-10-30 | Pierre Fabre Medicament | Axl antibody-drug conjugate and its use for the treatment of cancer |
| WO2015052535A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052533A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052532A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052534A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052321A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015052322A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015095124A1 (en) | 2013-12-16 | 2015-06-25 | Genentech Inc. | Peptidomimetic compounds and antibody-drug conjugates thereof |
| WO2015159076A1 (en) | 2014-04-15 | 2015-10-22 | Cancer Research Technology Limited | Humanized anti-tn-muc1 antibodies and their conjugates |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6053033B2 (en) | 1976-12-28 | 1985-11-22 | 財団法人微生物化学研究会 | New anticancer antibiotic mazethramycin and its production method |
| JPS57131791A (en) | 1980-12-31 | 1982-08-14 | Fujisawa Pharmaceut Co Ltd | Benzodiazepine derivative and its preparation |
| JPS58180487A (en) | 1982-04-16 | 1983-10-21 | Kyowa Hakko Kogyo Co Ltd | Antibiotic dc-81 and its preparation |
| US6214345B1 (en) | 1993-05-14 | 2001-04-10 | Bristol-Myers Squibb Co. | Lysosomal enzyme-cleavable antitumor drug conjugates |
| SG195524A1 (en) | 2003-11-06 | 2013-12-30 | Seattle Genetics Inc | Monomethylvaline compounds capable of conjugation to ligands |
| US8609105B2 (en) | 2008-03-18 | 2013-12-17 | Seattle Genetics, Inc. | Auristatin drug linker conjugates |
-
2012
- 2012-10-12 ES ES18168997T patent/ES2945932T3/en active Active
- 2012-10-12 CA CA2850373A patent/CA2850373C/en active Active
- 2012-10-12 ES ES12840661.8T patent/ES2687246T3/en active Active
- 2012-10-12 CN CN201810169400.3A patent/CN108164551A/en active Pending
- 2012-10-12 EP EP12840661.8A patent/EP2755642B1/en active Active
- 2012-10-12 JP JP2014535897A patent/JP6393617B2/en active Active
- 2012-10-12 US US14/351,168 patent/US9526798B2/en active Active
- 2012-10-12 WO PCT/US2012/059867 patent/WO2013055990A1/en active Application Filing
- 2012-10-12 CN CN201280062295.4A patent/CN103987384A/en active Pending
- 2012-10-12 EP EP18168997.7A patent/EP3388435B1/en active Active
-
2015
- 2015-01-15 HK HK15100506.0A patent/HK1200091A1/en unknown
-
2016
- 2016-06-14 US US15/181,900 patent/US9713647B2/en active Active
-
2017
- 2017-06-27 US US15/635,063 patent/US10328084B2/en active Active
-
2018
- 2018-06-04 JP JP2018107170A patent/JP7123635B2/en active Active
- 2018-12-06 HK HK18115628.8A patent/HK1256557A1/en unknown
Patent Citations (184)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3361742A (en) | 1964-12-07 | 1968-01-02 | Hoffmann La Roche | 5-oxo-1h-pyrrolo-[2, 1-c][1, 4]-benzodiazepin-2-crylamides |
| US3523941A (en) | 1967-03-06 | 1970-08-11 | Hoffmann La Roche | Benzodiazepine compounds and process for their preparation |
| US3524849A (en) | 1967-10-27 | 1970-08-18 | Hoffmann La Roche | Process for the preparation of pyrrolo-benzodiazepine acrylamides and intermediates useful therein |
| FR2027356A1 (en) | 1968-12-30 | 1970-09-25 | Fujisawa Pharmaceutical Co | Benzodiazepinone antibiotics |
| GB1299198A (en) | 1968-12-30 | 1972-12-06 | Fujisawa Pharmaceutical Co | Pyrrolobenzodiazepinone derivatives |
| US3794644A (en) | 1969-06-26 | 1974-02-26 | Fujisawa Pharmaceutical Co | Pyrrolo-benzodiazepinone compounds |
| JPS5382792U (en) | 1976-12-10 | 1978-07-08 | ||
| US4185016A (en) | 1977-12-27 | 1980-01-22 | The Green Cross Corporation | Pyrrolo benzodiazepine compounds |
| US4239683A (en) | 1977-12-27 | 1980-12-16 | The Green Cross Corporation | Pyrrolo benzodiazepine compounds |
| GB2053894A (en) | 1979-07-17 | 1981-02-11 | Green Cross Corp | Benzodiazepines processes for producing them and compositions containing them |
| US4309437A (en) | 1979-07-17 | 1982-01-05 | The Green Cross Corporation | Pyrrolo-benzodiazepine compounds and anti-tumor composition thereof |
| JPS57131791U (en) | 1981-02-06 | 1982-08-17 | ||
| US4353827A (en) | 1981-02-27 | 1982-10-12 | Hoffmann-La Roche Inc. | Diazepine derivatives |
| US4382032A (en) | 1981-02-27 | 1983-05-03 | Hoffmann-La Roche Inc. | Diazepine derivatives |
| US4386028A (en) | 1981-02-27 | 1983-05-31 | Hoffmann-La Roche Inc. | Imidazo[1,5a][1,4]benzodiazepines |
| US4405516A (en) | 1981-02-27 | 1983-09-20 | Hoffmann-La Roche Inc. | Benzodiazepine derivatives |
| US4405517A (en) | 1981-02-27 | 1983-09-20 | Hoffmann-La Roche Inc. | Azeto-diazepines |
| US4407752A (en) | 1981-02-27 | 1983-10-04 | Hoffmann-La Roche Inc. | Azeto-imidazo-diazepines |
| JPS58180487U (en) | 1982-05-28 | 1983-12-02 | 松下電工株式会社 | Light beam alarm assembly |
| US4427588A (en) | 1982-11-08 | 1984-01-24 | Bristol-Myers Company | Process for conversion of oxotomaymycin to tomaymycin |
| US4427587A (en) | 1982-11-10 | 1984-01-24 | Bristol-Myers Company | Total synthesis of antitumor antibiotics BBM-2040A and BBM-2040B |
| US4701325A (en) | 1983-02-17 | 1987-10-20 | The Green Cross Corporation | Injectable aqueous solution containing hydrogensulfite and/or sulfite and anticancerous benzodiazepine compound |
| FR2586683A1 (en) | 1985-08-29 | 1987-03-06 | Centre Nat Rech Scient | New neothramycin derivatives, process for their preparation and their application as medicaments |
| US4923984A (en) | 1988-08-05 | 1990-05-08 | Shionogi & Co., Ltd. | Pyrrolo(1,4)benzodiazepine derivatives |
| WO1992019620A1 (en) | 1991-05-07 | 1992-11-12 | Centre National De La Recherche Scientifique (Cnrs) | Novel derivatives of pyrrolo [1, 4]-benzodiazepines, method of preparation and medicaments containing them |
| WO1993018045A1 (en) | 1992-03-09 | 1993-09-16 | Cancer Research Campaign Technology Limited | Anti-cancer pyrrolobenzodiazepine derivatives |
| US5418241A (en) | 1992-09-28 | 1995-05-23 | Synthelabo | Piperidine derivatives, their preparation and their application in therapeutics |
| WO1995004718A1 (en) | 1993-08-04 | 1995-02-16 | Zeneca Limited | 4-methyleneproline derivatives as fungicides |
| WO2000003291A9 (en) | 1998-07-08 | 2000-07-27 | E Ink Corp | Methods for achieving improved color in microencapsulated electrophoretic devices |
| US6562806B1 (en) | 1998-08-27 | 2003-05-13 | Spirogen Limited | Pyrrolobenzodiazepines |
| US7049311B1 (en) | 1998-08-27 | 2006-05-23 | Spirogen Limited | Pyrrolbenzodiazepines |
| WO2000012509B1 (en) | 1998-08-27 | 2000-08-10 | Univ Portsmouth | Collections of compounds |
| WO2000012507A8 (en) | 1998-08-27 | 2000-10-19 | Univ Portsmouth | Pyrrolobenzodiazepines |
| US20040198722A1 (en) | 1998-08-27 | 2004-10-07 | Spirogen Limited | Collections of compounds |
| US7265105B2 (en) | 1998-08-27 | 2007-09-04 | Spirogen Limited | Pyrrolobenzodiazepines |
| US7704924B2 (en) | 1998-08-27 | 2010-04-27 | Spirogen Limited | Library of compounds comprising pyrrolobenzodiazepine moieties |
| US6608192B1 (en) | 1998-08-27 | 2003-08-19 | Spirogen Limited | Collections of compounds |
| US20030195196A1 (en) | 1998-08-27 | 2003-10-16 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2000012506A3 (en) | 1998-08-27 | 2000-06-29 | Univ Portsmouth | Collections of compounds |
| WO2000012508A2 (en) | 1998-08-27 | 2000-03-09 | Spirogen Limited | Pyrrolbenzodiazepines |
| US7067511B2 (en) | 1998-08-27 | 2006-06-27 | Spirogen Limited | Pyrrolobenzodiazepines |
| US6747144B1 (en) | 1998-08-27 | 2004-06-08 | Spirogen Limited | Collections of compounds |
| US6909006B1 (en) | 1999-08-27 | 2005-06-21 | Spirogen Limited | Cyclopropylindole derivatives |
| WO2001016104A1 (en) | 1999-08-27 | 2001-03-08 | Spirogen Limited | Cyclopropylindole derivatives |
| US6660742B2 (en) | 2000-09-19 | 2003-12-09 | Taiho Pharmaceutical Co. Ltd. | Compositions and methods of the use thereof achiral analogues of CC-1065 and the duocarmycins |
| US6362331B1 (en) | 2001-03-30 | 2002-03-26 | Council Of Scientific And Industrial Research | Process for the preparation of antitumor agents |
| US6660856B2 (en) | 2002-03-08 | 2003-12-09 | Kaohsiung Medical University | Synthesis of pyrrolo[2,1-c][1,4]benzodiazepine analogues |
| US20080213289A1 (en) | 2002-07-31 | 2008-09-04 | Seattle Genetics, Inc. | Recombinant anti-cd30 antibodies and uses thereof |
| US20040138269A1 (en) | 2002-10-11 | 2004-07-15 | Sugen, Inc. | Substituted pyrroles as kinase inhibitors |
| US7407951B2 (en) | 2002-11-14 | 2008-08-05 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2004043963A1 (en) | 2002-11-14 | 2004-05-27 | Spirogen Limited | Pyrrolobenzodiazepines |
| US6884799B2 (en) | 2003-03-31 | 2005-04-26 | Council Of Scientific And Industrial Research | Non-cross-linking pyrrolo[2,1-c][1,4]benzodiazepines and process thereof |
| US7429658B2 (en) | 2003-09-11 | 2008-09-30 | Spirogen Limited | Synthesis of protected pyrrolobenzodiazepines |
| WO2005023814A1 (en) | 2003-09-11 | 2005-03-17 | Spirogen Limited | Synthesis of protected pyrrolobenzodiazepines |
| WO2005042535A1 (en) | 2003-10-22 | 2005-05-12 | Spirogen Limited | Pyrrolobenzodiazepines |
| US7244724B2 (en) | 2003-10-22 | 2007-07-17 | United States Of America, Represented By The Secretary, Department Of Health And Human Services | Pyrrolobenzodiazepines |
| WO2005040170A3 (en) | 2003-10-22 | 2005-06-30 | Us Gov Health & Human Serv | Pyrrolobenzodiazepine derivatives, compositions comprising the same and methods related thereto |
| WO2005082023A3 (en) | 2004-02-23 | 2005-12-22 | Genentech Inc | Heterocyclic self-immolative linkers and conjugates |
| US20110160192A1 (en) | 2004-03-01 | 2011-06-30 | Spirogen Limited | Biaryl amino acids and their use in dna binding oligomers |
| WO2005085250A8 (en) | 2004-03-01 | 2005-10-27 | Spirogen Ltd | C8, c8' linked 5-oxo-1,2,3,11a-tetrahydro-5h-pyrrolo[2,1-c][1,4]benzodiazepine dimers with 1h-pyrrole-dicarboxylic acid amide linkers and oligomeric analogs therof as well as related compounds for the treatment of proliferative diseases |
| US20070249591A1 (en) | 2004-03-01 | 2007-10-25 | Spirogen Limited | Biaryl Amino Acids and Their Use in Dna Binding Oligomers |
| US7557099B2 (en) | 2004-03-01 | 2009-07-07 | Spirogen Limited | Pyrrolobenzodiazepines as key intermediates in the synthesis of dimeric cytotoxic pyrrolobenzodiazepines |
| WO2005085177A3 (en) | 2004-03-01 | 2005-11-17 | Spirogen Ltd | Biaryl amino acids and their use in dna binding oligomers |
| WO2005085251A1 (en) | 2004-03-01 | 2005-09-15 | Spirogen Limited | 11-hydroxy-5h-pyrrolo[2,1-c][1,4]benzodiazepin-5-one derivatives as key intermediates for the preparation of c2 substituted pyrrolobenzodiazepines |
| US7741319B2 (en) | 2004-03-01 | 2010-06-22 | Spirogen Limited | 11-hydroxy-5h-pyrrolo[2,1-c][1,4] benzodiazepin-5-one derivatives as key intermediates for the preparation of c2 substituted pyrrolobenzodiazepines |
| US20070191349A1 (en) | 2004-03-01 | 2007-08-16 | Spirogen Limited | C8, c8' linked 5-oxo-1,2,3,11a-tetrahydro-5h-pyrrolo[2,1-c][1,4] benzodiazepine dimers with 1h-pyrrole-dicarboxylic acid amide linkers and oligomeric analogs therof as well as related compounds for the treatment of proliferative diseases |
| WO2005085259A3 (en) | 2004-03-01 | 2006-01-05 | Spirogen Ltd | Pyrrolobenzodiazepines as key intermediates in the synthesis of dimeric cytotoxic pyrrolobenzodiazepines |
| US20070185336A1 (en) | 2004-03-03 | 2007-08-09 | Degussa Ag | Process for preparing n-protected 4-ketoproline derivatives |
| US7528126B2 (en) | 2004-03-09 | 2009-05-05 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2005085260A1 (en) | 2004-03-09 | 2005-09-15 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2005105113A3 (en) | 2004-04-27 | 2007-02-22 | Sod Conseils Rech Applic | Therapeutic compositions containing at least one pyrrolobenzodiazepine derivative and fludarabine |
| US8034808B2 (en) | 2004-04-27 | 2011-10-11 | Ipsen Pharma S.A.S. | Therapeutic compositions containing at least one pyrrolobenzodiazepine derivative and fludarabine |
| US20070232592A1 (en) | 2004-04-27 | 2007-10-04 | Societe De Conseils Recherches Et D'applications Scientifiques | Therapeutic Compositions Containing at Least one Pyrrolobenzodiazepine Derivative and Fludarabine |
| US20080090812A1 (en) | 2004-05-13 | 2008-04-17 | Spirogen Limited | Pyrrolobenzodiazepine Therapeutic Agents Useful in the Treatment of Leukemias |
| WO2005110423A3 (en) | 2004-05-13 | 2006-01-19 | Spirogen Ltd | Pyrrolobenzodiazepine therapeutic agents useful in the treatment of leukaemias |
| US20070092940A1 (en) | 2004-09-23 | 2007-04-26 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| US20080092940A1 (en) | 2004-12-24 | 2008-04-24 | Showa Denko K.K. | Production Method of Thermoelectric Semiconductor Alloy, Thermoelectric Conversion Module and Thermoelectric Power Generating Device |
| US20090148942A1 (en) | 2005-04-19 | 2009-06-11 | Mcdonagh Charlotte | Humanized anti-cd70 binding agents and uses thereof |
| US7612062B2 (en) | 2005-04-21 | 2009-11-03 | Spirogen Limited | Pyrrolobenzodiazepines |
| US8633185B2 (en) | 2005-04-21 | 2014-01-21 | Spirogen Sarl | Pyrrolobenzodiazepines |
| WO2006111759A1 (en) | 2005-04-21 | 2006-10-26 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2007039752A1 (en) | 2005-10-05 | 2007-04-12 | Spirogen Limited | Alkyl 4- [4- (5-oxo-2, 3, 5, 11a-tetrahyd0-5h-pyrr0l0 [2, 1-c] [1, 4] benzodiazepine-8-yloxy) -butyrylamino]-1h-pyrrole-2-carboxylate derivatives and related compounds for the treatment of a proliferative disease |
| US8637664B2 (en) | 2005-10-05 | 2014-01-28 | Spirogen Sarl | Alkyl 4- [4- (5-oxo-2,3,5, 11a-tetrahydo-5H-pyrrolo [2, 1-c] [1,4] benzodiazepine-8-yloxy)-butyrylamino]-1H-pyrrole-2-carboxylate derivatives and related compounds for the treatment of a proliferative disease |
| US20070154906A1 (en) | 2005-10-05 | 2007-07-05 | Spirogen Ltd. | Methods to identify therapeutic candidates |
| EP1813614A1 (en) | 2006-01-25 | 2007-08-01 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives |
| US8163736B2 (en) | 2006-01-25 | 2012-04-24 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives |
| WO2007085930A1 (en) | 2006-01-25 | 2007-08-02 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2008010101A2 (en) | 2006-07-18 | 2008-01-24 | Sanofi-Aventis | Antagonist antibody against epha2 for the treatment of cancer |
| WO2008047242A3 (en) | 2006-10-19 | 2008-06-26 | Sanofi Aventis | Novel anti-cd38 antibodies for the treatment of cancer |
| WO2008050140A2 (en) | 2006-10-27 | 2008-05-02 | Spirogen Limited | Compounds for treatment of parasitic infection |
| WO2008070593A3 (en) | 2006-12-01 | 2008-10-30 | Seattle Genetics Inc | Variant target binding agents and uses thereof |
| WO2009016516A2 (en) | 2007-07-19 | 2009-02-05 | Sanofi-Aventis | Cytotoxic agents comprising new tomaymycin derivatives and their therapeutic use |
| WO2009060215A1 (en) | 2007-11-09 | 2009-05-14 | Spirogen Limited | Polyamides |
| WO2009060208A1 (en) | 2007-11-09 | 2009-05-14 | Spirogen Limited | Pyrrolobenzodiazepines |
| WO2010010347A1 (en) | 2008-07-22 | 2010-01-28 | Spirogen Limited | Pyrrolobenzodiazepines |
| US8501934B2 (en) | 2008-07-22 | 2013-08-06 | Spirogen Sarl | Pyrrolobenzodiazepines |
| WO2010043880A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| US20110196148A1 (en) | 2008-10-17 | 2011-08-11 | Spirogen Limited | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| US20110201803A1 (en) | 2008-10-17 | 2011-08-18 | Spirogen Limited | Pyrrolobenzodiazepines |
| US8940733B2 (en) | 2008-10-17 | 2015-01-27 | Spirogen Sarl | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| US20150111880A1 (en) | 2008-10-17 | 2015-04-23 | Spirogen Sarl | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| US8592576B2 (en) | 2008-10-17 | 2013-11-26 | Spirogen Sarl | Unsymmetrical pyrrolobenzodiazepine-dimers for treatment of proliferative diseases |
| WO2010043877A1 (en) | 2008-10-17 | 2010-04-22 | Spirogen Limited | Pyrrolobenzodiazepines |
| US8487092B2 (en) | 2008-10-17 | 2013-07-16 | Spirogen Developments Sarl | Pyrrolobenzodiazepines |
| WO2010091150A1 (en) | 2009-02-05 | 2010-08-12 | Immunogen, Inc. | Novel benzodiazepine derivatives |
| WO2011023883A1 (en) | 2009-08-25 | 2011-03-03 | Sanofi-Aventis | Conjugates of pyrrolo[1,4]benzodiazepine dimers as anticancer agents |
| WO2011038159A2 (en) | 2009-09-24 | 2011-03-31 | Seattle Genetics, Inc. | Dr5 ligand drug conjugates |
| WO2011100227A1 (en) | 2010-02-09 | 2011-08-18 | Bristol-Myers Squibb Company | Benzylpyrrolidinone derivatives as modulators of chemokine receptor activity |
| US20130028917A1 (en) | 2010-04-15 | 2013-01-31 | Spirogen Developments Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2011130613A1 (en) | 2010-04-15 | 2011-10-20 | Seattle Genetics, Inc. | Targeted pyrrolobenzodiazapine conjugates |
| US20110256157A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US8829184B2 (en) | 2010-04-15 | 2014-09-09 | Spirogen Sarl | Intermediates useful for the synthesis of pyrrolobenzodiazepines |
| US20130028919A1 (en) | 2010-04-15 | 2013-01-31 | Spirogen Developments Sàrl | Targeted pyrrolobenzodiazapine conjugates |
| US20140066435A1 (en) | 2010-04-15 | 2014-03-06 | Spirogen Sarl | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2011130598A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines and conjugates thereof |
| WO2011128650A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Intermediates useful for the synthesis of pyrrolobenzodiazepines |
| US8697688B2 (en) | 2010-04-15 | 2014-04-15 | Seattle Genetics Inc. | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2011130616A1 (en) | 2010-04-15 | 2011-10-20 | Spirogen Limited | Pyrrolobenzodiazepines used to treat proliferative diseases |
| WO2012128868A1 (en) | 2011-02-15 | 2012-09-27 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives |
| WO2012112708A1 (en) | 2011-02-15 | 2012-08-23 | Immunogen, Inc. | Cytotoxic benzodiazepine derivatives and methods of preparation |
| US20150158869A1 (en) | 2011-09-20 | 2015-06-11 | Spirogen Sàrl | Pyrrolobenzodiazepines as unsymmetrical dimeric pbd compounds for inclusion in targeted conjugates |
| WO2013041606A1 (en) | 2011-09-20 | 2013-03-28 | Spirogen Sàrl | Pyrrolobenzodiazepines as unsymmetrical dimeric pbd compounds for inclusion in targeted conjugates |
| US9102704B2 (en) | 2011-10-14 | 2015-08-11 | Spirogen Sarl | Synthesis method and intermediates useful in the preparation of pyrrolobenzodiazepines |
| WO2013055993A1 (en) | 2011-10-14 | 2013-04-18 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| US20140234346A1 (en) | 2011-10-14 | 2014-08-21 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013053871A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013053873A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013053872A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Synthesis method and intermediates useful in the preparation of pyrrolobenzodiazepines |
| US20140274907A1 (en) | 2011-10-14 | 2014-09-18 | Spirogen Sarl | Pyrrolobenzodiazepines |
| US20140302066A1 (en) | 2011-10-14 | 2014-10-09 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| US20130266595A1 (en) | 2011-10-14 | 2013-10-10 | Genentech, Inc. | Pyrrolobenzodiazepines and conjugates thereof |
| WO2013055987A1 (en) | 2011-10-14 | 2013-04-18 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2013055990A1 (en) | 2011-10-14 | 2013-04-18 | Seattle Genetics, Inc. | Pyrrolobenzodiazepines and targeted conjugates |
| WO2013164593A1 (en) | 2012-04-30 | 2013-11-07 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| WO2013164592A1 (en) | 2012-04-30 | 2013-11-07 | Ucl Business Plc | Pyrrolobenzodiazepines |
| US20150133435A1 (en) | 2012-04-30 | 2015-05-14 | Spirogen Sàrl | Pyrrolobenzodiazepines |
| US20150126495A1 (en) | 2012-04-30 | 2015-05-07 | Ucl Business Plc | Pyrrolobenzodiazepines |
| US20140030279A1 (en) | 2012-07-09 | 2014-01-30 | Spirogen Sarl | Anti-cd22 antibodies and immunoconjugates |
| WO2014011518A1 (en) | 2012-07-09 | 2014-01-16 | Genentech, Inc. | Immunoconjugates comprising anti-cd22 antibodies |
| US20140030280A1 (en) | 2012-07-09 | 2014-01-30 | Genentech, Inc. | Anti-cd79b antibodies and immunoconjugates |
| WO2014011519A1 (en) | 2012-07-09 | 2014-01-16 | Genentech, Inc. | Immunoconjugates comprising anti-cd79b antibodies |
| US20150183883A1 (en) | 2012-08-02 | 2015-07-02 | Genentech, Inc. | Anti-etbr antibodies and immunoconjugates |
| WO2014022679A2 (en) | 2012-08-02 | 2014-02-06 | Genentech, Inc. | Anti-etbr antibodies and immunoconjugates |
| US20140120118A1 (en) | 2012-10-12 | 2014-05-01 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014057117A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US20150297746A1 (en) | 2012-10-12 | 2015-10-22 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US20150283258A1 (en) | 2012-10-12 | 2015-10-08 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine - anti-psma antibody conjugates |
| WO2014057119A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US20150283263A1 (en) | 2012-10-12 | 2015-10-08 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2014057113A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine - anti-psma antibody conjugates |
| US20150283262A1 (en) | 2012-10-12 | 2015-10-08 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US20150273077A1 (en) | 2012-10-12 | 2015-10-01 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-her2 antibody conjugates |
| WO2014057073A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| US20150274737A1 (en) | 2012-10-12 | 2015-10-01 | Spirogen Sàrl | Synthesis and intermediates of pyrrolobenzodiazepine derivatives for conjugation |
| WO2014057120A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| US20150273078A1 (en) | 2012-10-12 | 2015-10-01 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-psma antibody conjugates |
| WO2014057114A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-psma antibody conjugates |
| US20150265722A1 (en) | 2012-10-12 | 2015-09-24 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| WO2014057074A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014057118A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sarl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| WO2014057115A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-her2 antibody conjugates |
| WO2014057072A1 (en) | 2012-10-12 | 2014-04-17 | Spirogen Sàrl | Synthesis and intermediates of pyrrolobenzodiazepine derivatives for conjugation |
| WO2014057122A1 (en) | 2012-10-12 | 2014-04-17 | Adc Therapeutics Sàrl | Pyrrolobenzodiazepine-anti-cd22 antibody conjugates |
| US20140127239A1 (en) | 2012-10-12 | 2014-05-08 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| US20150344482A1 (en) | 2012-12-21 | 2015-12-03 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| US20150315196A1 (en) | 2012-12-21 | 2015-11-05 | Spirogen Sàrl | Unsymmetrical pyrrolobenzodiazepines-dimers for use in the treatment of proliferative and autoimmune diseases |
| WO2014096365A1 (en) | 2012-12-21 | 2014-06-26 | Spirogen Sàrl | Unsymmetrical pyrrolobenzodiazepines-dimers for use in the treatment of proliferative and autoimmune diseases |
| WO2014096368A1 (en) | 2012-12-21 | 2014-06-26 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014130879A2 (en) | 2013-02-22 | 2014-08-28 | Stem Centrx, Inc. | Novel antibody conjugates and uses thereof |
| US20160015828A1 (en) | 2013-02-22 | 2016-01-21 | Spirogen Sàrl | Novel antibody conjugates and uses thereof |
| WO2014140862A2 (en) | 2013-03-13 | 2014-09-18 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014159981A3 (en) | 2013-03-13 | 2015-04-09 | Spirogen Sarl | Pyrrolobenzodiazepines and conjugates thereof |
| US20140294868A1 (en) | 2013-03-13 | 2014-10-02 | Genentech, Inc. | Pyrrolobenzodiazepines and conjugates thereof |
| US20160031887A1 (en) | 2013-03-13 | 2016-02-04 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014140174A1 (en) | 2013-03-13 | 2014-09-18 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2014174111A1 (en) | 2013-04-26 | 2014-10-30 | Pierre Fabre Medicament | Axl antibody-drug conjugate and its use for the treatment of cancer |
| WO2015052533A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052535A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052532A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052534A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepine-antibody conjugates |
| WO2015052321A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015052322A1 (en) | 2013-10-11 | 2015-04-16 | Spirogen Sàrl | Pyrrolobenzodiazepines and conjugates thereof |
| WO2015095124A1 (en) | 2013-12-16 | 2015-06-25 | Genentech Inc. | Peptidomimetic compounds and antibody-drug conjugates thereof |
| WO2015159076A1 (en) | 2014-04-15 | 2015-10-22 | Cancer Research Technology Limited | Humanized anti-tn-muc1 antibodies and their conjugates |
Non-Patent Citations (169)
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10561739B2 (en) | 2010-04-15 | 2020-02-18 | Seattle Genetics Inc. | Targeted pyrrolobenzodiazapine conjugates |
| US10335497B2 (en) | 2012-10-12 | 2019-07-02 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10646584B2 (en) | 2012-10-12 | 2020-05-12 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10994023B2 (en) | 2012-10-12 | 2021-05-04 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US11701430B2 (en) | 2012-10-12 | 2023-07-18 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10188746B2 (en) | 2014-09-10 | 2019-01-29 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10420777B2 (en) | 2014-09-12 | 2019-09-24 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof |
| US10392393B2 (en) | 2016-01-26 | 2019-08-27 | Medimmune Limited | Pyrrolobenzodiazepines |
| US11517626B2 (en) | 2016-02-10 | 2022-12-06 | Medimmune Limited | Pyrrolobenzodiazepine antibody conjugates |
| US10695439B2 (en) | 2016-02-10 | 2020-06-30 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US10543279B2 (en) | 2016-04-29 | 2020-01-28 | Medimmune Limited | Pyrrolobenzodiazepine conjugates and their use for the treatment of cancer |
| US10799595B2 (en) | 2016-10-14 | 2020-10-13 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11612665B2 (en) | 2017-02-08 | 2023-03-28 | Medimmune Limited | Pyrrolobenzodiazepine-antibody conjugates |
| US11160872B2 (en) | 2017-02-08 | 2021-11-02 | Adc Therapeutics Sa | Pyrrolobenzodiazepine-antibody conjugates |
| US11813335B2 (en) | 2017-02-08 | 2023-11-14 | Medimmune Limited | Pyrrolobenzodiazepine-antibody conjugates |
| US11370801B2 (en) | 2017-04-18 | 2022-06-28 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11649250B2 (en) | 2017-08-18 | 2023-05-16 | Medimmune Limited | Pyrrolobenzodiazepine conjugates |
| US11352324B2 (en) | 2018-03-01 | 2022-06-07 | Medimmune Limited | Methods |
| US11524969B2 (en) | 2018-04-12 | 2022-12-13 | Medimmune Limited | Pyrrolobenzodiazepines and conjugates thereof as antitumour agents |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2945932T3 (en) | 2023-07-10 |
| ES2687246T3 (en) | 2018-10-24 |
| US20170290924A1 (en) | 2017-10-12 |
| CA2850373C (en) | 2019-07-16 |
| CA2850373A1 (en) | 2013-04-18 |
| EP2755642A1 (en) | 2014-07-23 |
| WO2013055990A1 (en) | 2013-04-18 |
| EP2755642B1 (en) | 2018-07-18 |
| JP6393617B2 (en) | 2018-09-19 |
| EP3388435B1 (en) | 2023-05-03 |
| US10328084B2 (en) | 2019-06-25 |
| US9713647B2 (en) | 2017-07-25 |
| JP2014528480A (en) | 2014-10-27 |
| CN108164551A (en) | 2018-06-15 |
| EP3388435A1 (en) | 2018-10-17 |
| JP7123635B2 (en) | 2022-08-23 |
| HK1256557A1 (en) | 2019-09-27 |
| US20160289239A1 (en) | 2016-10-06 |
| EP2755642A4 (en) | 2015-06-24 |
| HK1200091A1 (en) | 2015-07-31 |
| JP2018150371A (en) | 2018-09-27 |
| US20140286970A1 (en) | 2014-09-25 |
| CN103987384A (en) | 2014-08-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10328084B2 (en) | Pyrrolobenzodiazepines and targeted conjugates | |
| US10329352B2 (en) | Pyrrolobenzodiazepines and targeted conjugates | |
| US10561739B2 (en) | Targeted pyrrolobenzodiazapine conjugates | |
| US9732084B2 (en) | Pyrrolobenzodiazepines used to treat proliferative diseases | |
| US9388187B2 (en) | Pyrrolobenzodiazepines | |
| US9399073B2 (en) | Pyrrolobenzodiazepines | |
| US9399641B2 (en) | Pyrrolobenzodiazepines as unsymmetrical dimeric PBD compounds for inclusion in targeted conjugates | |
| NZ623216B2 (en) | Pyrrolobenzodiazepines and targeted conjugates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: SPIROGEN SARL, SWITZERLAND Free format text: MERGER;ASSIGNOR:SPIROGEN DEVELOPMENTS SARL;REEL/FRAME:032747/0332 Effective date: 20121105 Owner name: SPIROGEN LIMITED, UNITED KINGDOM Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:HOWARD, PHILIP WILSON;REEL/FRAME:032745/0537 Effective date: 20120828 Owner name: SPIROGEN DEVELOPMENTS SARL, SWITZERLAND Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:SPIROGEN LIMITED;REEL/FRAME:032745/0586 Effective date: 20120831 Owner name: SEATTLE GENETICS, INC., WASHINGTON Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:JEFFREY, SCOTT C.;BURKE, PATRICK J.;SIGNING DATES FROM 20120914 TO 20120917;REEL/FRAME:032746/0071 |
|
| AS | Assignment |
Owner name: MEDIMMUNE LIMITED, ENGLAND Free format text: CONFIRMATORY ASSIGNMENT;ASSIGNOR:SPIROGEN SARL;REEL/FRAME:036932/0278 Effective date: 20141013 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| AS | Assignment |
Owner name: MEDIMMUNE LIMITED, ENGLAND Free format text: CORRECTIVE ASSIGNMENT TO CORRECT THE POSTAL CODE OF THE ASSIGNEE PREVIOUSLY RECORDED AT REEL: 036932 FRAME: 0278. ASSIGNOR(S) HEREBY CONFIRMS THE ASSIGNMENT;ASSIGNOR:SPIROGEN SARL;REEL/FRAME:042242/0389 Effective date: 20141013 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 4TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1551); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 4 |
|
| AS | Assignment |
Owner name: SEAGEN INC., WASHINGTON Free format text: CHANGE OF NAME;ASSIGNOR:SEATTLE GENETICS, INC.;REEL/FRAME:055362/0182 Effective date: 20201006 |