JP5064225B2 - 抗体精製法 - Google Patents
抗体精製法 Download PDFInfo
- Publication number
- JP5064225B2 JP5064225B2 JP2007537850A JP2007537850A JP5064225B2 JP 5064225 B2 JP5064225 B2 JP 5064225B2 JP 2007537850 A JP2007537850 A JP 2007537850A JP 2007537850 A JP2007537850 A JP 2007537850A JP 5064225 B2 JP5064225 B2 JP 5064225B2
- Authority
- JP
- Japan
- Prior art keywords
- protein
- group
- separation matrix
- antibody
- matrix
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 - B01D15/36
- B01D15/3804—Affinity chromatography
- B01D15/3828—Ligand exchange chromatography, e.g. complexation, chelation or metal interaction chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J41/00—Anion exchange; Use of material as anion exchangers; Treatment of material for improving the anion exchange properties
- B01J41/04—Processes using organic exchangers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/32—Bonded phase chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/32—Bonded phase chromatography
- B01D15/325—Reversed phase
- B01D15/327—Reversed phase with hydrophobic interaction
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/36—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving ionic interaction
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/36—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving ionic interaction
- B01D15/361—Ion-exchange
- B01D15/363—Anion-exchange
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 - B01D15/36
- B01D15/3804—Affinity chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 - B01D15/36
- B01D15/3804—Affinity chromatography
- B01D15/3809—Affinity chromatography of the antigen-antibody type, e.g. protein A, G, L chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D15/00—Separating processes involving the treatment of liquids with solid sorbents; Apparatus therefor
- B01D15/08—Selective adsorption, e.g. chromatography
- B01D15/26—Selective adsorption, e.g. chromatography characterised by the separation mechanism
- B01D15/38—Selective adsorption, e.g. chromatography characterised by the separation mechanism involving specific interaction not covered by one or more of groups B01D15/265 - B01D15/36
- B01D15/3847—Multimodal interactions
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/281—Sorbents specially adapted for preparative, analytical or investigative chromatography
- B01J20/286—Phases chemically bonded to a substrate, e.g. to silica or to polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3242—Layers with a functional group, e.g. an affinity material, a ligand, a reactant or a complexing group
- B01J20/3244—Non-macromolecular compounds
- B01J20/3246—Non-macromolecular compounds having a well defined chemical structure
- B01J20/3248—Non-macromolecular compounds having a well defined chemical structure the functional group or the linking, spacer or anchoring group as a whole comprising at least one type of heteroatom selected from a nitrogen, oxygen or sulfur, these atoms not being part of the carrier as such
- B01J20/3253—Non-macromolecular compounds having a well defined chemical structure the functional group or the linking, spacer or anchoring group as a whole comprising at least one type of heteroatom selected from a nitrogen, oxygen or sulfur, these atoms not being part of the carrier as such comprising a cyclic structure not containing any of the heteroatoms nitrogen, oxygen or sulfur, e.g. aromatic structures
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/30—Processes for preparing, regenerating, or reactivating
- B01J20/32—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating
- B01J20/3231—Impregnating or coating ; Solid sorbent compositions obtained from processes involving impregnating or coating characterised by the coating or impregnating layer
- B01J20/3242—Layers with a functional group, e.g. an affinity material, a ligand, a reactant or a complexing group
- B01J20/3285—Coating or impregnation layers comprising different type of functional groups or interactions, e.g. different ligands in various parts of the sorbent, mixed mode, dual zone, bimodal, multimodal, ionic or hydrophobic, cationic or anionic, hydrophilic or hydrophobic
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J41/00—Anion exchange; Use of material as anion exchangers; Treatment of material for improving the anion exchange properties
- B01J41/20—Anion exchangers for chromatographic processes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/14—Extraction; Separation; Purification
- C07K1/16—Extraction; Separation; Purification by chromatography
- C07K1/165—Extraction; Separation; Purification by chromatography mixed-mode chromatography
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/14—Extraction; Separation; Purification
- C07K1/36—Extraction; Separation; Purification by a combination of two or more processes of different types
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/06—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies from serum
- C07K16/065—Purification, fragmentation
Landscapes
- Chemical & Material Sciences (AREA)
- Analytical Chemistry (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Molecular Biology (AREA)
- General Health & Medical Sciences (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Biochemistry (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Immunology (AREA)
- Peptides Or Proteins (AREA)
- Treatment Of Liquids With Adsorbents In General (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Description
用語「抗体」及び「免疫グロブリン」は、本明細書中で互換的に使用される。
図1において、a)はプロトタイプマルチモーダルリガンドである2−アミノベンズイミダゾールを示し、b)はプロトタイプマルチモーダルリガンドであるチオミカミンを示し、c)はビーズ形態の担体に固定されたプロトタイプマルチモーダルリガンドであるN−ベンジル−N−メチルエタノールアミンを示し、d)はプロトタイプマルチモーダルリガンドであるN,N−ジメチルベンジルアミンを示す。実験の部で、プロトタイプリガンドを6%アガロースマトリックスであるSepharose(商標)6FFにカップリングする。
非結合条件下で、約50mgのmAb1を含む試料を、プロトタイプ901035A(N−ベンジル−N−メチルエタノールアミン)及び901035B(N,N−ジメチルベンジルアミン)上に約5及び12mS/cmで注入した。5、10及び15カラム体積(CV)時に通過液画分(FT)を集めた。溶出ピークからの画分をプールした。FT画分を、HCP及びプロテインAの含有量について分析した。
カラム及びゲルは、スウェーデン、Uppsala、GE Healthcareから入手した。
使用する化学薬品は全て分析級とした。水はMilliQで濾過した。
対照マトリックスは、Q Sepharose(商標)Fast Flow(FF)(スウェーデン、Uppsala、GE Healthcare)とした。マルチモーダル分離マトリックスのプロトタイプは、下表1に記載した通りのリガンドを所持した。
A.マトリックスへのアリル基の導入
Sepharose(商標)6 Fast Flow(スウェーデン、Uppsala、GE Healthcare)をアリルグリシジルエーテルで次のように活性化した。即ち、100mlのSepharose(商標)6 Fast Flowを吸引乾燥し、0.3gのNaBH4、12gのNa2SO4及び35mlの50%NaOH水溶液と混合した。混合物を50℃で1時間撹拌した。100mlのアリルグリシジルエーテルを添加した後、懸濁液を激しく撹拌しながら更に16時間50℃に保持した。混合物を濾過した後、ゲルを500mlの蒸留水、500mlのエタノール、200mlの蒸留水、200mlの0.2M酢酸及び500mlの蒸留水で順次洗浄した。
50mlのアリル活性化Sepharose(商標)6 Fast Flow(アリル基0.22ミリモル/ml排水ゲル)、1gの酢酸ナトリウム及び15mlの蒸留水からなる撹拌懸濁液に、臭素を黄色が持続するまで添加した。次いで、ギ酸ナトリウムを懸濁液が完全に脱色されるまで添加した。反応混合物を濾過し、ゲルを500mlの蒸留水で洗浄した。次いで、活性化ゲルを反応容器に直ちに移送し、N−ベンジル−N−メチルエタノールアミンと更に反応させた。
アミン基の窒素原子を経由してマトリックスにアミン基を直接導入した。典型的な手順において、マトリックスへのカップリングは、アリル基の臭素化及び塩基性条件下での求核置換により実現された。25mlの臭素活性化ゲル(アリル基0.22ミリモル/ml排水ゲル)を、N−ベンジル−N−メチルエタノールアミン(16.0ml)の溶液を含む反応ガラス瓶に移した。5mlの水を添加し、反応溶液のpHを水酸化ナトリウム溶液で12.0に調整した。反応物を撹拌下に16時間50℃で保持した。反応混合物を濾過した後、ゲルを、10mlの蒸留水で3回、10mlの0.5HCl水で3回、最後に10mlの蒸留水で3回、順次洗浄した。BMEA Sepharose(商標)Fast Flowゲルが、アミン0.15ミリモル/mlゲルの置換度で得られた。
MAb1及びMAb2と表示され、それぞれ1.46及び1.50の吸光係数を有する2つの異なるヒト化IgG抗体、サブクラス1を使用した。双方の抗体とも、CHO培養で発現させ、続いて本発明の実験に先立って従来からのプロテインAアフィニティークロマトグラフィーを使用して精製した。
式中、
A280は280nmでの吸光度であり、
ε(mL・mg−1・cm−1)は特定のタンパク質に対する吸光係数であり、
C(mg/mL)はタンパク質の濃度であり、
l(cm)は光路長である。
緩衝液は、25mM Bis−Tris、pH6.0又は6.5とした。所望される導電率、約5又は12mS/cmに応じて、35又は100mMのNaClを含めた。プロトタイプ901035A及び901035Bの場合、溶出緩衝液(B−緩衝液)は、25mM Bis−Tris、0.5M NaCl、pH6.5とした。リガンドとしてチオミカミン及びABIを用いるプロトタイプの場合、溶出緩衝液(B−緩衝液)は、0.5M酢酸ナトリウム、pH4.0とした。流速は0.5mL/分(150cm/時間)とした。
A−緩衝液は25mM Bis−Tris、pH6.0とした。導電率は、50mM NaClを添加して約7mS/cmとし、B−緩衝液は、0.5M酢酸ナトリウム、pH4.0とした。流速は0.5mL/分(150cm/時間)とした。試料濃度は、MAbを4mg/mL、rPrAを0.04mg/mL(1%(w/w)に相当)とした。
選択した画分を、800μLのSPA試料希釈液+200μL試料の比率でSPA試料希釈液と混合した。混合後、画分を加熱ブロック上、99℃で10分間加熱し、次いで、再混合した。次いで、試料を組換えプロテインAについて分析した。
試料(最小で600μL)をHCP含有量について分析した。検出下限は10ng/mLである。
プロトタイプリガンドN−ベンジル−N−メチルエタノールアミン(901035A)及びN,N−ジメチルベンジルアミン(901035B)で精製したMAb−1含有試料
実施例1では、50mgのMAb1を含む試料を、Sepharose(商標)6FFに固定化したN−ベンジル−N−メチルエタノールアミン(901035A)、Sepharose(商標)6FFに固定化したN,N−ジメチルベンジルアミン(901035B)及び対照マトリックスQ Sepharose(商標)FFに、25mM Bis−Tris、100mM NaCl(〜12mS/cm)、pH6.5で適用した。溶出は、25mM Bis−Tris、0.5M NaCl、pH6.5で実施した。
プロトタイプリガンド、チオミカミン及び2−アミノベンズイミダゾールで精製したMAb−1含有試料
この実施例では、20mgのMAb1を含む試料を、プロトタイプ及び対照分離マトリックスに注入した。緩衝液は、平衡化及び注入の場合、25mM Bis−Tris、35mM NaCl(〜5mS/cm)、pH6.5とした。溶出緩衝液は、0.5M 酢酸ナトリウム、pH4.0とした。a)チオミカミン、65μモル/mL(1282004)、b)チオミカミン128μモル/mL(1282002)、c)Q Sepharose(商標)FF、d)2−アミノベンズイミダゾール(ABI)、65μモル/mL(1282045)及びe)2−アミノベンズイミダゾール(ABI)、146μモル/mL(1282032)である。HCP及びプロテインA分析の結果を下表4及び5に示す。
プロトタイプリガンド、チオミカミン及び2−アミノベンズイミダゾールで精製したMAb−2含有試料
20mgのMAb2を含む試料をプロトタイプ及び対照に適用した。緩衝液は、25mM Bis−Tris、100mM NaCl(〜12mS/cm)、pH6.0とした。溶出は、0.5M酢酸ナトリウム、pH4.0で実施した。得られたクロマトグラムを図3に示す。図3a)は、チオミカミン65μモル/mL(1282004、緑)、チオミカミン128μモル/mL(1282002、青)及びQ Sepharose(商標)FF(黒)。図3b)は、2−アミノベンズイミダゾール65μモル/mL(1282045、青)、2−アミノベンズイミダゾール146μモル/mL(1282032、緑)及びQ Sepharose(商標)FF(黒)である。分析SECを使用して、下表6及び7に示すようなHCP及びプロテインA分析のための画分を選択した。
プロトタイプリガンド、N−ベンジル−N−メチルエタノールアミン、N,N−ジメチルベンジルアミン、チオミカミン及び2−アミノベンズイミダゾールでの、MAb1及び組換えプロテインA(rPrA)を含む試料からのMAb1の精製
この実施例では、Ab1−rプロテインAを含む試料についてのプロトタイプでのクロマトグラフィーを実施した。A−緩衝液は、25mM Bis−Tris、50mM NaCl、pH6.0とした。導電率は約7mS/cmであった。B−緩衝液は、0.5M酢酸ナトリウム、pH4.0とした。流速は0.5mL/分(150cm/時間)であった。試料は、mAb1が4mg/mL、rプロテインAが1%(w/w)の濃度の、10mgのmAb1、0.10mgのrPrAとした。結果を図4に示す。
Q Phenyl Sepharose 6 Fast Flow配置での抗体精製
非結合条件下で、約50mgのmAbを含む試料をプロトタイプQ Phenyl Sepharose(商標)6 Fast Flowに注入した。5、10及び15カラム容積(CV)時に通過液画分(FT)を採取した。溶出ピークからの画分を分析した。
カラム及びPhenyl Sepharose(商標)6 Fast Flowは、スウェーデン、Uppsala、GE Healthcareから入手した。
HR 5/5(商標) カタログ番号18−0338−01 CV=1mL。
クロマトグラフィー装置 AKTAExplorer(商標)10
分光計 Spectra MAX plus。
使用する化学薬品は、全て分析級とした。水はMiLLiQで濾過した。
架橋アガロースゲル(Phenyl Sepharose(商標)6 Fast Flow(高置換)、スウェーデン、Uppsala、GE Healthcare)から出発する、本発明による分離マトリックスを調製するための1つの方法を次に例示する。
Q−基(−N(CH3)3)は、次のように、グリシジルトリメチルアンモニウムクロリド(G−MAC)との反応によってPhenyl Sepharose(商標)6 Fast Flow(高置換)に導入した。即ち、15gの吸引乾燥Phenyl Sepharose(商標)6 Fast Flow(高置換)を、5mlの水、5mlの50%NaOH水溶液、0.02gのNaBH4及び40mlのG−MACと混合した。混合物を30℃で16時間撹拌した。混合物を濾過した後、ゲルを、100mlの蒸留水、100mlのエタノール及び100mlの蒸留水で順次洗浄した。
使用するモノクロナール抗体は、CHO培養で発現させ、続いて、当面の実験に先立って従来からのプロテインAアフィニティークロマトグラフィーを使用して精製した。
mAb試料を緩衝液で10倍に希釈した。試料溶液の2つの控えをA280で測定した。平均値を使用し、Lambert−Beerの法則に従って濃度を計算した。即ち、
C=A/(l×ε)
C=IgGの濃度
A=280nmでの吸光度
l=光路長
ε=mAbに対するモル吸光係数、mg−1ml=1.46。
宿主細胞タンパク質及びプロテインAからのmAbの分離を、非結合条件下で試験した。カラムに適用する試料はMabSelectで精製したmAbとした。流速は、0.5ml/分(150cm/時間)とした。ラン中は終始、280nmでの吸光度を検出した。2つの異なる緩衝液(下記参照)を試験した。各ランの前にA−緩衝液への緩衝液交換を実施した。試料体積に応じてHiPrep脱塩及びHiTrap脱塩カラムを使用した。
緩衝液 A−緩衝液:25mM Tris/HCl、pH8.0
B−緩衝液:25mM Tris/HCl、0.5M NaCl、pH8.0
A−緩衝液:25mMリン酸塩緩衝液、pH7.0
B−緩衝液:25mMリン酸緩衝液、0.5M NaCl、pH7.0
方法:出発材料として、MabSelectからのpHを調整した溶出液を使用した。
選択した画分を、800μlのSPA試料希釈液+200μlの試料の比率でSPA試料希釈剤と混合した。混合した後、画分を加熱ブロック上、99℃で10分間加熱し、次いで再混合した。次いで、試料を組換えプロテインAについて分析した。
試料(最小で600μl)をHCP含有量について分析した。検出下限は10ng/mLである。
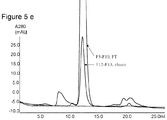
非結合条件下で、約50mgのmAbを、Q Phenyl Sepharose(商標)6 Fast Flowを充填したHR5/5カラムに2つの異なるpH(pH7.0及び8.0)で注入した。通過液画分を図1に従って5、10、15カラム容積(CV)時に採取した。表8及び9は、通過液画分のプロテインA及びHCP分析の結果を示す。画分中にプロテインAの残余は検出できなかった。更に、8.0の試料pHを使用した場合に、FT1及びFT2中に宿主細胞タンパク質は検出できなかった。7.0の試料pHを使用した場合には、少量の宿主細胞タンパク質が観察されたが、HCPは、試料中のHCP含有量に比べて約50分の1に低下した。又、図6は、勾配溶出でのクロマトグラムで極めて小さなピークのみが観察されるので(図6)、モノクロナール抗体分子がQ Phenyl Sepharose(商標)6 Fast Flowに吸着されないことを示している。
Claims (9)
- 液体試料中の1種以上の抗体を1種以上の他の化合物から分離する方法であって、当該方法が、液体試料を含む移動相をマルチモーダル分離マトリックスと接触させ、抗体を移動相中に遊離させた状態で、1種以上の目標化合物を吸着することを含んでおり、上記マルチモーダル分離マトリックスが、目標化合物の負に荷電した部位と相互作用できる第1の基と目標化合物と電荷−電荷相互作用以外の1種以上の相互作用ができる芳香族基を含む第2の基とを含む、分離方法。
- 前記液体試料が粗供給液を含む、請求項1記載の方法。
- 前記目標化合物が宿主細胞タンパク質であり、80%以上のタンパク質がマルチモーダル分離マトリックスに吸着される、請求項2記載の方法。
- 前記液体試料が、分離マトリックスからの溶出液を含む、請求項1記載の方法。
- 溶出液が得られる分離マトリックスが、タンパク質リガンドを含む、請求項4記載の方法。
- 第1の基が第4級アミンである、請求項1乃至請求項5のいずれか1項記載の方法。
- 第2の基が、芳香族又はヘテロ芳香族の環構造を含む疎水性基である、請求項1乃至請求項6のいずれか1項記載の方法。
- 前記分離マトリックスが粒状であり、第1の基を含むリガンドが固定された第1粒子と第2の基を含むリガンドが固定された第2粒子との混合物を含む、請求項1乃至請求項7のいずれか1項記載の方法。
- 分離マトリックスが、第1の基を含む第1リガンドと第2の基を含む第2リガンドとの混合物が固定されたフィルターである、請求項1乃至請求項8のいずれか1項記載の方法。
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SE0402558A SE0402558D0 (sv) | 2004-10-21 | 2004-10-21 | A process for the purification of antibodies |
| SE0402558-1 | 2004-10-21 | ||
| SE0402910 | 2004-11-26 | ||
| SE0402910-4 | 2004-11-26 | ||
| PCT/SE2005/001591 WO2006043895A1 (en) | 2004-10-21 | 2005-10-21 | A method of antibody purification |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2008517906A JP2008517906A (ja) | 2008-05-29 |
| JP2008517906A5 JP2008517906A5 (ja) | 2008-11-13 |
| JP5064225B2 true JP5064225B2 (ja) | 2012-10-31 |
Family
ID=36203226
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2007537850A Active JP5064225B2 (ja) | 2004-10-21 | 2005-10-21 | 抗体精製法 |
| JP2007537851A Active JP4831436B2 (ja) | 2004-10-21 | 2005-10-21 | クロマトグラフィーリガンド |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2007537851A Active JP4831436B2 (ja) | 2004-10-21 | 2005-10-21 | クロマトグラフィーリガンド |
Country Status (12)
| Country | Link |
|---|---|
| US (5) | US7867784B2 (ja) |
| EP (2) | EP1827691B1 (ja) |
| JP (2) | JP5064225B2 (ja) |
| KR (2) | KR101243601B1 (ja) |
| AU (2) | AU2005296351B2 (ja) |
| BR (2) | BRPI0518216C8 (ja) |
| CA (2) | CA2583578C (ja) |
| DK (1) | DK1827691T3 (ja) |
| ES (1) | ES2612572T3 (ja) |
| PL (1) | PL1827691T3 (ja) |
| RU (3) | RU2396246C2 (ja) |
| WO (2) | WO2006043895A1 (ja) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8350946B2 (en) | 2005-01-31 | 2013-01-08 | The Invention Science Fund I, Llc | Viewfinder for shared image device |
| US8606383B2 (en) | 2005-01-31 | 2013-12-10 | The Invention Science Fund I, Llc | Audio sharing |
| US8681225B2 (en) | 2005-06-02 | 2014-03-25 | Royce A. Levien | Storage access technique for captured data |
| US8902320B2 (en) | 2005-01-31 | 2014-12-02 | The Invention Science Fund I, Llc | Shared image device synchronization or designation |
| US8988537B2 (en) | 2005-01-31 | 2015-03-24 | The Invention Science Fund I, Llc | Shared image devices |
| US9001215B2 (en) | 2005-06-02 | 2015-04-07 | The Invention Science Fund I, Llc | Estimating shared image device operational capabilities or resources |
| US9041826B2 (en) | 2005-06-02 | 2015-05-26 | The Invention Science Fund I, Llc | Capturing selected image objects |
| US9082456B2 (en) | 2005-01-31 | 2015-07-14 | The Invention Science Fund I Llc | Shared image device designation |
| US9093121B2 (en) | 2006-02-28 | 2015-07-28 | The Invention Science Fund I, Llc | Data management of an audio data stream |
| US9124729B2 (en) | 2005-01-31 | 2015-09-01 | The Invention Science Fund I, Llc | Shared image device synchronization or designation |
| US9325781B2 (en) | 2005-01-31 | 2016-04-26 | Invention Science Fund I, Llc | Audio sharing |
| US9451200B2 (en) | 2005-06-02 | 2016-09-20 | Invention Science Fund I, Llc | Storage access technique for captured data |
| US9489717B2 (en) | 2005-01-31 | 2016-11-08 | Invention Science Fund I, Llc | Shared image device |
| US9819490B2 (en) | 2005-05-04 | 2017-11-14 | Invention Science Fund I, Llc | Regional proximity for shared image device(s) |
| US9910341B2 (en) | 2005-01-31 | 2018-03-06 | The Invention Science Fund I, Llc | Shared image device designation |
| US9942511B2 (en) | 2005-10-31 | 2018-04-10 | Invention Science Fund I, Llc | Preservation/degradation of video/audio aspects of a data stream |
| WO2018092691A1 (ja) * | 2016-11-18 | 2018-05-24 | Jnc株式会社 | 抗体の精製方法 |
| US10003762B2 (en) | 2005-04-26 | 2018-06-19 | Invention Science Fund I, Llc | Shared image devices |
Families Citing this family (109)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4801598B2 (ja) | 2004-01-20 | 2011-10-26 | ポール・コーポレーション | 生理的イオン強度でタンパク質を吸着させるためのクロマトグラフィー材料 |
| US7867784B2 (en) * | 2004-10-21 | 2011-01-11 | Ge Healthcare Bio-Science Ab | Chromatography ligand |
| TW200639190A (en) * | 2005-02-04 | 2006-11-16 | Showa Denko Kk | Packing material for ion chromatography |
| NZ571479A (en) | 2006-04-05 | 2012-10-26 | Abbott Biotech Ltd | Antibody purification |
| AU2007272494B2 (en) | 2006-07-14 | 2011-07-28 | Wisconsin Alumni Research Foundation | Adsorptive membranes for trapping viruses |
| US7999085B2 (en) * | 2007-01-09 | 2011-08-16 | Bio-Rad Laboratories, Inc. | Enhanced capacity and purification of protein by mixed mode chromatography in the presence of aqueous-soluble nonionic organic polymers |
| US7691980B2 (en) * | 2007-01-09 | 2010-04-06 | Bio-Rad Laboratories, Inc. | Enhanced capacity and purification of antibodies by mixed mode chromatography in the presence of aqueous-soluble nonionic organic polymers |
| WO2008085116A1 (en) * | 2007-01-10 | 2008-07-17 | Ge Healthcare Bio-Sciences Ab | Mult i -modal ion exchange chromotography resins |
| US10562007B2 (en) | 2007-07-25 | 2020-02-18 | Ge Healthcare Bioprocess R&D Ab | Separation matrix |
| EP2719705B1 (en) * | 2007-10-26 | 2020-07-29 | Asahi Kasei Kabushiki Kaisha | Method for purifying protein |
| WO2009058769A1 (en) * | 2007-10-30 | 2009-05-07 | Schering Corporation | Purification of antibodies containing hydrophobic variants |
| JP5999899B2 (ja) * | 2008-04-08 | 2016-09-28 | バイオ−ラッド ラボラトリーズ インコーポレーティッド | 抗体のクロマトグラフィー精製法 |
| DE102008018732B4 (de) * | 2008-04-14 | 2022-06-09 | Sartorius Stedim Biotech Gmbh | Verfahren zur Stofftrennung unter Verwendung einer Cellulosehydrat-Membran in der Größenausschlusschromatographie |
| DE102008055821A1 (de) * | 2008-04-14 | 2009-10-15 | Sartorius Stedim Biotech Gmbh | Cellulosehydrat-Membran, Verfahren zur ihrer Herstellung und Verwendung davon |
| DE102008018734B4 (de) * | 2008-04-14 | 2013-03-21 | Sartorius Stedim Biotech Gmbh | Hydrophobe Cellulose-Membran, Verfahren zu ihrer Herstellung und ihre Verwendung in der hydrophoben Interaktionschromatographie |
| FR2931838B1 (fr) * | 2008-06-02 | 2010-06-11 | Millipore Corp | Installation pour traiter un liquide biologique. |
| US20110166332A1 (en) * | 2008-09-12 | 2011-07-07 | Ge Healthcare Bio-Sciences Ab | Enhanced antibody aggregate removal with capto adhere in the presence of protein-excluded zwitterions |
| US8497358B2 (en) | 2008-12-19 | 2013-07-30 | Takeda Pharmaceutical Company Limited | Antibody purification method |
| FR2940145B1 (fr) | 2008-12-24 | 2011-03-25 | Millipore Corp | Chariot et installation de traitement d'un liquide biologique |
| JP2010158624A (ja) * | 2009-01-08 | 2010-07-22 | Asahi Kasei Chemicals Corp | 多孔質吸着膜、及び当該多孔質吸着膜を用いたたんぱく質の精製方法 |
| FR2941385B1 (fr) | 2009-01-23 | 2011-04-01 | Millipore Corp | Procede pour fournir un circuit pour liquide biologique et circuit obtenu. |
| WO2011001963A1 (ja) * | 2009-07-03 | 2011-01-06 | 旭化成ケミカルズ株式会社 | 多孔質基材に固定されたグラフト鎖に結合しているアミノ基及びアルキル基を有する多孔膜を用いた抗体の精製方法 |
| EA201290076A1 (ru) | 2009-07-28 | 2012-08-30 | Инстракцион Гмбх | Специфичный сорбент для связывания белков и пептидов и способ выделения с его использованием |
| US8277649B2 (en) | 2009-12-14 | 2012-10-02 | General Electric Company | Membranes and associated methods for purification of antibodies |
| FR2955119B1 (fr) | 2010-01-13 | 2012-12-28 | Millipore Corp | Circuit pour liquide biologique |
| MX344248B (es) | 2010-05-25 | 2016-12-09 | Genentech Inc * | Metodos de purificacion de polipeptidos. |
| FR2960795B1 (fr) | 2010-06-08 | 2012-07-27 | Millipore Corp | Dispositif pour une installation de traitement de liquide biologique |
| FR2960796B1 (fr) | 2010-06-08 | 2014-01-24 | Millipore Corp | Dispositif pour une installation de traitement de liquide biologique |
| FR2960794B1 (fr) | 2010-06-08 | 2012-07-27 | Millipore Corp | Dispositif pour une installation de traitement de liquide biologique |
| FR2961711B1 (fr) | 2010-06-23 | 2012-08-17 | Millipore Corp | Poche pour circuit d'une installation de traitement de liquide biologique |
| FR2961713B1 (fr) | 2010-06-23 | 2012-08-10 | Millipore Corp | Poche pour circuit d'une installation de traitement de liquide biologique |
| KR101997543B1 (ko) | 2010-07-30 | 2019-07-09 | 이엠디 밀리포어 코포레이션 | 크로마토그래피 매질 및 방법 |
| FR2963573B1 (fr) | 2010-08-03 | 2012-08-31 | Millipore Corp | Chariot de pompage pour une installation de traitement de liquide biologique |
| CN103201003B (zh) * | 2010-09-20 | 2016-04-13 | Abbvie公司 | 采用模拟移动床色谱纯化抗体 |
| EP2655412B1 (en) | 2010-12-21 | 2018-01-17 | F. Hoffmann-La Roche AG | Isoform enriched antibody preparation and method for obtaining it |
| FR2973396B1 (fr) | 2011-03-28 | 2013-05-10 | Millipore Corp | Installation de traitement de liquide biologique |
| EP2702077A2 (en) | 2011-04-27 | 2014-03-05 | AbbVie Inc. | Methods for controlling the galactosylation profile of recombinantly-expressed proteins |
| EP2570185A1 (en) * | 2011-09-15 | 2013-03-20 | InstrAction GmbH | Sorbent comprising an aromatic ring system on its surface for the purification of organic molecules |
| EP2570184A1 (en) * | 2011-09-15 | 2013-03-20 | InstrAction GmbH | Sorbent comprising on its surface an aromatic ring system having an anionic or deprotonizable group for the purification of organic molecules |
| US8802448B2 (en) | 2011-07-27 | 2014-08-12 | Pall Corporation | Mixed mode ligands |
| EP2578286A1 (en) * | 2011-10-04 | 2013-04-10 | Merck Patent GmbH | Method and apparatus for chromatographic purification |
| US9309282B2 (en) * | 2011-10-19 | 2016-04-12 | Bio-Rad Laboratories, Inc. | Solid phase for mixed-mode chromatographic purification of proteins |
| ES2767135T3 (es) * | 2011-10-19 | 2020-06-16 | Novimmune Sa | Métodos para purificar anticuerpos |
| US20140288278A1 (en) * | 2011-10-31 | 2014-09-25 | Joseph Nti-Gyabaah | Chromatography process for resolving heterogeneous antibody aggregates |
| EP2782925B1 (en) * | 2011-11-23 | 2024-03-06 | Sanofi | Protein purification using bis-tris buffer |
| WO2013075740A1 (en) * | 2011-11-23 | 2013-05-30 | Sanofi | Antibody purification method |
| CN104812491B (zh) | 2012-03-08 | 2018-06-19 | 生物辐射实验室股份有限公司 | 阴离子交换-疏水性混合模式 |
| SG10201701224UA (en) * | 2012-03-12 | 2017-04-27 | Merck Patent Gmbh | Removal of protein aggregates from biopharmaceutical preparations in a flowthrough mode |
| US9150645B2 (en) | 2012-04-20 | 2015-10-06 | Abbvie, Inc. | Cell culture methods to reduce acidic species |
| WO2013158273A1 (en) | 2012-04-20 | 2013-10-24 | Abbvie Inc. | Methods to modulate c-terminal lysine variant distribution |
| US9067990B2 (en) | 2013-03-14 | 2015-06-30 | Abbvie, Inc. | Protein purification using displacement chromatography |
| US9249182B2 (en) | 2012-05-24 | 2016-02-02 | Abbvie, Inc. | Purification of antibodies using hydrophobic interaction chromatography |
| RU2501008C1 (ru) * | 2012-06-14 | 2013-12-10 | Федеральное государственное бюджетное учреждение "Научно-исследовательский институт клинической иммунологии" Сибирского отделения Российской академии медицинских наук (ФГБУ "НИИКИ" СОРАМН) | СПОСОБ АФФИННОГО ВЫДЕЛЕНИЯ АУТОАНТИТЕЛ КЛАССА IgG К ИММУНОРЕГУЛЯТОРНОМУ ЦИТОКИНУ TNF |
| FR2993572B1 (fr) | 2012-07-23 | 2016-04-15 | Emd Millipore Corp | Circuit pour liquide biologique comportant une vanne a pincement |
| US9512214B2 (en) | 2012-09-02 | 2016-12-06 | Abbvie, Inc. | Methods to control protein heterogeneity |
| US9206390B2 (en) | 2012-09-02 | 2015-12-08 | Abbvie, Inc. | Methods to control protein heterogeneity |
| JP6335785B2 (ja) * | 2012-09-03 | 2018-05-30 | 株式会社カネカ | ミックスモード抗体アフィニティー分離マトリックスとそれを用いた精製方法および標的分子 |
| ES2868093T3 (es) | 2012-09-17 | 2021-10-21 | Grace W R & Co | Medio y dispositivos de cromatografía |
| WO2014077762A1 (en) | 2012-11-13 | 2014-05-22 | Ge Healthcare Bio-Sciences Ab | Multimodal anion exchange matrices |
| ES2912930T3 (es) | 2013-02-26 | 2022-05-30 | Merck Millipore Ltd | Membranas de cromatografía en modo mixto |
| RU2662929C2 (ru) | 2013-03-08 | 2018-07-31 | Джензим Корпорейшн | Интегрированное непрерывное производство терапевтических белковых лекарственных веществ |
| WO2014143205A1 (en) | 2013-03-12 | 2014-09-18 | Abbvie Inc. | Human antibodies that bind human tnf-alpha and methods of preparing the same |
| US10023608B1 (en) * | 2013-03-13 | 2018-07-17 | Amgen Inc. | Protein purification methods to remove impurities |
| US9017687B1 (en) | 2013-10-18 | 2015-04-28 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same using displacement chromatography |
| US8921526B2 (en) | 2013-03-14 | 2014-12-30 | Abbvie, Inc. | Mutated anti-TNFα antibodies and methods of their use |
| US9499614B2 (en) | 2013-03-14 | 2016-11-22 | Abbvie Inc. | Methods for modulating protein glycosylation profiles of recombinant protein therapeutics using monosaccharides and oligosaccharides |
| DK2970378T3 (da) | 2013-03-15 | 2021-08-23 | Biogen Ma Inc | Hydrofob interaktionsproteinkromatografi under saltfrie betingelser |
| EP3301115B1 (en) * | 2013-05-06 | 2021-11-10 | Sanofi | Continuous multistep process for purifying antibodies |
| US10519195B2 (en) | 2013-09-17 | 2019-12-31 | Kaneka Corporation | Antibody purification method, antibody obtained therefrom, novel antibody purification method using cation exchanger, and antibody obtained therefrom |
| US9598667B2 (en) | 2013-10-04 | 2017-03-21 | Abbvie Inc. | Use of metal ions for modulation of protein glycosylation profiles of recombinant proteins |
| US9085618B2 (en) | 2013-10-18 | 2015-07-21 | Abbvie, Inc. | Low acidic species compositions and methods for producing and using the same |
| US8946395B1 (en) | 2013-10-18 | 2015-02-03 | Abbvie Inc. | Purification of proteins using hydrophobic interaction chromatography |
| US9181337B2 (en) | 2013-10-18 | 2015-11-10 | Abbvie, Inc. | Modulated lysine variant species compositions and methods for producing and using the same |
| WO2015073884A2 (en) | 2013-11-15 | 2015-05-21 | Abbvie, Inc. | Glycoengineered binding protein compositions |
| SG10201804200WA (en) | 2013-12-12 | 2018-07-30 | Emd Millipore Corp | Protein separations using an acrylamide containing filter |
| EP3094390B1 (en) | 2014-01-16 | 2021-07-07 | W.R. Grace & CO. - CONN. | Affinity chromatography media and chromatography devices |
| EP3094391A4 (en) * | 2014-01-17 | 2017-09-20 | Repligen Corporation | Sterilizing chromatography columns |
| TWI709570B (zh) | 2014-01-17 | 2020-11-11 | 美商健臻公司 | 無菌層析法及製法 |
| TWI709569B (zh) | 2014-01-17 | 2020-11-11 | 美商健臻公司 | 無菌層析樹脂及其用於製造方法的用途 |
| US20240100450A9 (en) * | 2014-01-17 | 2024-03-28 | Repligen Corporation | Sterilizing chromatography columns |
| US10583429B2 (en) | 2014-03-14 | 2020-03-10 | Bio-Rad Laboratories, Inc. | Mixed mode ligands |
| US11389783B2 (en) | 2014-05-02 | 2022-07-19 | W.R. Grace & Co.-Conn. | Functionalized support material and methods of making and using functionalized support material |
| US10155799B2 (en) * | 2014-07-21 | 2018-12-18 | Merck Sharp & Dohme Corp. | Chromatography process for purification of insulin and insulin analogs |
| JP6665184B2 (ja) | 2014-12-08 | 2020-03-13 | イー・エム・デイー・ミリポア・コーポレイシヨン | 混床イオン交換吸着剤 |
| US10695744B2 (en) | 2015-06-05 | 2020-06-30 | W. R. Grace & Co.-Conn. | Adsorbent biprocessing clarification agents and methods of making and using the same |
| US10864512B2 (en) * | 2016-04-06 | 2020-12-15 | Cytiva Bioprocess R&D Ab | Chromatography matrix |
| EA201990317A1 (ru) | 2016-08-16 | 2019-07-31 | Ридженерон Фармасьютикалз, Инк. | Способы определения количества индивидуальных антител из смеси |
| ES2924060T3 (es) | 2016-10-25 | 2022-10-04 | Regeneron Pharma | Procedimientos y sistema para análisis de datos de cromatografía |
| JP7479151B2 (ja) * | 2017-01-30 | 2024-05-08 | リジェネロン・ファーマシューティカルズ・インコーポレイテッド | クロマトグラフィーにおけるバイオバーデンを低減するための組成物および方法 |
| US11161128B2 (en) | 2017-11-14 | 2021-11-02 | General Electric Company | Spray nozzle device for delivering a restorative coating through a hole in a case of a turbine engine |
| US11534780B2 (en) | 2017-11-14 | 2022-12-27 | General Electric Company | Spray nozzle device for delivering a restorative coating through a hole in a case of a turbine engine |
| EP3749697A4 (en) | 2018-02-05 | 2021-11-03 | Bio-Rad Laboratories, Inc. | CHROMATOGRAPHIC RESIN WITH A LIGAND WITH ANION EXCHANGE-HYDROPHOBIC MIXED MODE |
| US20210106974A1 (en) | 2018-03-05 | 2021-04-15 | Chiral Technologies Europe Sas | Composite materials for bioseparations |
| JP7344232B2 (ja) | 2018-03-05 | 2023-09-13 | キラル テクノロジーズ ヨーロッパ エスアーエス | バイオセパレーションのための複合材料 |
| WO2019173731A1 (en) * | 2018-03-08 | 2019-09-12 | Bio-Rad Laboratories, Inc. | Anionic exchange-hydrophobic mixed mode chromatography resin |
| WO2019175412A2 (en) * | 2018-03-15 | 2019-09-19 | Klawego Gmbh & Co. Kg | Composite materials for the depletion of contaminants from solutions |
| TW202005694A (zh) | 2018-07-02 | 2020-02-01 | 美商里珍納龍藥品有限公司 | 自混合物製備多肽之系統及方法 |
| EP3843869A1 (en) | 2018-08-31 | 2021-07-07 | Genzyme Corporation | Sterile chromatography resin and use thereof in manufacturing processes |
| US11045773B2 (en) | 2018-08-31 | 2021-06-29 | Pall Corporation | Salt tolerant porous medium |
| US10737259B2 (en) | 2018-08-31 | 2020-08-11 | Pall Corporation | Salt tolerant anion exchange medium |
| KR20210143767A (ko) | 2019-03-27 | 2021-11-29 | 씨티바 스웨덴 에이비 | 생체분자의 분리 방법 |
| CN114040815A (zh) * | 2019-09-05 | 2022-02-11 | 生物辐射实验室股份有限公司 | 阴离子交换-疏水混合模式色谱树脂 |
| EP4019125A1 (en) | 2020-12-22 | 2022-06-29 | Sartorius Stedim Biotech GmbH | Chromatographic material and method of producing same |
| GB202110014D0 (en) | 2021-07-12 | 2021-08-25 | Cytiva Bioprocess R & D Ab | A method for separating adeno-associated virus capsids, compositions obtained by said method and uses thereof |
| CA3236293A1 (en) | 2021-12-07 | 2023-06-15 | Cytiva Bioprocess R&D Ab | Separation matrix and methods for separating target molecules |
| GB202202777D0 (en) | 2022-03-01 | 2022-04-13 | Cytiva Bioprocess R & D Ab | A method for separating supercoiled plasmid DNA |
| GB202206123D0 (en) | 2022-04-27 | 2022-06-08 | Cytiva Bioprocess R & D Ab | A pre-screening method and a method for separating adeno-associated virus capsids |
| GB202214280D0 (en) | 2022-09-29 | 2022-11-16 | Cytiva Vioprocess R&D Ab | Chromatography ligand and chromatography material, and uses thereof |
| GB202215814D0 (en) | 2022-10-26 | 2022-12-07 | Cytiva Bioprocess R & D Ab | A chromatography device, system, and use thereof for analytic separation |
Family Cites Families (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2405555A (en) * | 1946-08-13 | Manufacture of heterocyclic bases | ||
| US3467656A (en) * | 1967-09-06 | 1969-09-16 | Hoffmann La Roche | 4-aryl-3,1-benzoxazine-2-thione |
| SE8204810L (sv) | 1982-08-23 | 1984-02-24 | Pharmacia Fine Chemicals Ab | Hybrid-dna-molekyl, transformerad mikroorganism och sett att framstella protein a |
| US5151350A (en) | 1982-10-27 | 1992-09-29 | Repligen Corporation | Cloned genes encoding recombinant protein a |
| US4983722A (en) | 1988-06-08 | 1991-01-08 | Miles Inc. | Removal of protein A from antibody preparations |
| US5322859A (en) * | 1993-02-08 | 1994-06-21 | University Of Iowa Research Foundation | Antiglaucoma drug composition and method |
| US5429746A (en) * | 1994-02-22 | 1995-07-04 | Smith Kline Beecham Corporation | Antibody purification |
| US5652348A (en) | 1994-09-23 | 1997-07-29 | Massey University | Chromatographic resins and methods for using same |
| US6117996A (en) | 1995-09-20 | 2000-09-12 | Novo Nordisk A/S | Triazine based ligands and use thereof |
| GB9519197D0 (en) * | 1995-09-20 | 1995-11-22 | Affinity Chromatography Ltd | Novel affinity ligands and their use |
| SE9601368D0 (sv) | 1996-04-11 | 1996-04-11 | Pharmacia Biotech Ab | Process for the production of a porous cross-linked polysaccharide gel |
| EP1386660B1 (en) | 1996-08-30 | 2009-09-02 | Upfront Chromatography A/S | Isolation of immunoglobulins |
| SE9700383D0 (sv) * | 1997-02-04 | 1997-02-04 | Pharmacia Biotech Ab | An adsorption/separation method and a medium for adsorption/separation |
| US5908960A (en) | 1997-05-07 | 1999-06-01 | Smithkline Beecham Corporation | Compounds |
| JP4737903B2 (ja) * | 1999-07-07 | 2011-08-03 | ザイモジェネティクス, インコーポレイテッド | ヒトサイトカイン受容体 |
| SE9904197D0 (sv) * | 1999-11-22 | 1999-11-22 | Amersham Pharm Biotech Ab | A method for anion exchange adsorption on matrices carrying mixed mode ligands |
| DE60032520T2 (de) * | 2000-06-08 | 2007-10-04 | Kaneka Corp. | Verfahren zur herstellung von sulfonsäureestern |
| SE0004932D0 (sv) | 2000-12-31 | 2000-12-31 | Apbiotech Ab | A method for mixed mode adsorption and mixed mode adsorbents |
| WO2002053256A1 (en) * | 2001-01-05 | 2002-07-11 | Pro-Chem, Inc. | Devices and methods for purification |
| US6652853B2 (en) * | 2001-03-08 | 2003-11-25 | Ludwig Institute For Cancer Research | Method for treating cancer using A33 specific antibodies and chemotherapeutic agents |
| DE10155984A1 (de) | 2001-11-15 | 2003-05-28 | Boehringer Ingelheim Pharma | Verfahren zur Reduktion des Liganden-leakage von Affinitätschromatographie-Matrices |
| DK1601697T3 (da) * | 2003-02-28 | 2007-10-01 | Lonza Biologics Plc | Oprensning af antistof ved protein A- og ionbytningskromatografi |
| AU2005216847B2 (en) * | 2004-02-27 | 2010-04-01 | Cytiva Bioprocess R&D Ab | A process for the purification of antibodies |
| BRPI0515586B1 (pt) | 2004-09-22 | 2015-11-03 | Ge Healthcare Bio Sciences Ab | método de preparar uma matriz cromatográfica de polissacarídeo reticulado, matriz cromatográfica porosa de polissacarídeo reticulado, e, uso de uma matriz cromatográfica |
| US9266041B2 (en) * | 2004-10-21 | 2016-02-23 | Ge Healthcare Bio-Sciences Ab | Chromatography ligand |
| US7867784B2 (en) * | 2004-10-21 | 2011-01-11 | Ge Healthcare Bio-Science Ab | Chromatography ligand |
| US7691980B2 (en) * | 2007-01-09 | 2010-04-06 | Bio-Rad Laboratories, Inc. | Enhanced capacity and purification of antibodies by mixed mode chromatography in the presence of aqueous-soluble nonionic organic polymers |
-
2005
- 2005-10-21 US US11/577,208 patent/US7867784B2/en active Active
- 2005-10-21 RU RU2007112990/04A patent/RU2396246C2/ru active
- 2005-10-21 RU RU2010110437/04A patent/RU2541429C2/ru active
- 2005-10-21 JP JP2007537850A patent/JP5064225B2/ja active Active
- 2005-10-21 CA CA2583578A patent/CA2583578C/en active Active
- 2005-10-21 CA CA2582113A patent/CA2582113C/en active Active
- 2005-10-21 US US11/577,203 patent/US7714112B2/en active Active
- 2005-10-21 WO PCT/SE2005/001591 patent/WO2006043895A1/en active Application Filing
- 2005-10-21 PL PL05796563T patent/PL1827691T3/pl unknown
- 2005-10-21 RU RU2007112991/15A patent/RU2389552C2/ru active
- 2005-10-21 BR BRPI0518216A patent/BRPI0518216C8/pt active IP Right Grant
- 2005-10-21 KR KR1020077009037A patent/KR101243601B1/ko active IP Right Grant
- 2005-10-21 DK DK05796563.4T patent/DK1827691T3/en active
- 2005-10-21 JP JP2007537851A patent/JP4831436B2/ja active Active
- 2005-10-21 AU AU2005296351A patent/AU2005296351B2/en active Active
- 2005-10-21 EP EP05796563.4A patent/EP1827691B1/en active Active
- 2005-10-21 ES ES05796563.4T patent/ES2612572T3/es active Active
- 2005-10-21 EP EP05796462A patent/EP1807205B1/en active Active
- 2005-10-21 AU AU2005296352A patent/AU2005296352B2/en active Active
- 2005-10-21 KR KR1020077009033A patent/KR101243425B1/ko active IP Right Grant
- 2005-10-21 WO PCT/SE2005/001592 patent/WO2006043896A1/en active Application Filing
- 2005-10-21 BR BRPI0518207-7A patent/BRPI0518207B1/pt active IP Right Grant
-
2011
- 2011-01-04 US US12/984,132 patent/US8895710B2/en active Active
-
2014
- 2014-11-17 US US14/542,816 patent/US9878266B2/en active Active
-
2018
- 2018-01-29 US US15/882,998 patent/US10751645B2/en active Active
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9910341B2 (en) | 2005-01-31 | 2018-03-06 | The Invention Science Fund I, Llc | Shared image device designation |
| US9489717B2 (en) | 2005-01-31 | 2016-11-08 | Invention Science Fund I, Llc | Shared image device |
| US9082456B2 (en) | 2005-01-31 | 2015-07-14 | The Invention Science Fund I Llc | Shared image device designation |
| US8902320B2 (en) | 2005-01-31 | 2014-12-02 | The Invention Science Fund I, Llc | Shared image device synchronization or designation |
| US8988537B2 (en) | 2005-01-31 | 2015-03-24 | The Invention Science Fund I, Llc | Shared image devices |
| US8606383B2 (en) | 2005-01-31 | 2013-12-10 | The Invention Science Fund I, Llc | Audio sharing |
| US9019383B2 (en) | 2005-01-31 | 2015-04-28 | The Invention Science Fund I, Llc | Shared image devices |
| US9124729B2 (en) | 2005-01-31 | 2015-09-01 | The Invention Science Fund I, Llc | Shared image device synchronization or designation |
| US8350946B2 (en) | 2005-01-31 | 2013-01-08 | The Invention Science Fund I, Llc | Viewfinder for shared image device |
| US9325781B2 (en) | 2005-01-31 | 2016-04-26 | Invention Science Fund I, Llc | Audio sharing |
| US10003762B2 (en) | 2005-04-26 | 2018-06-19 | Invention Science Fund I, Llc | Shared image devices |
| US9819490B2 (en) | 2005-05-04 | 2017-11-14 | Invention Science Fund I, Llc | Regional proximity for shared image device(s) |
| US9001215B2 (en) | 2005-06-02 | 2015-04-07 | The Invention Science Fund I, Llc | Estimating shared image device operational capabilities or resources |
| US9451200B2 (en) | 2005-06-02 | 2016-09-20 | Invention Science Fund I, Llc | Storage access technique for captured data |
| US9041826B2 (en) | 2005-06-02 | 2015-05-26 | The Invention Science Fund I, Llc | Capturing selected image objects |
| US9967424B2 (en) | 2005-06-02 | 2018-05-08 | Invention Science Fund I, Llc | Data storage usage protocol |
| US8681225B2 (en) | 2005-06-02 | 2014-03-25 | Royce A. Levien | Storage access technique for captured data |
| US9942511B2 (en) | 2005-10-31 | 2018-04-10 | Invention Science Fund I, Llc | Preservation/degradation of video/audio aspects of a data stream |
| US9093121B2 (en) | 2006-02-28 | 2015-07-28 | The Invention Science Fund I, Llc | Data management of an audio data stream |
| WO2018092691A1 (ja) * | 2016-11-18 | 2018-05-24 | Jnc株式会社 | 抗体の精製方法 |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5064225B2 (ja) | 抗体精製法 | |
| JP4776615B2 (ja) | 抗体精製 | |
| KR101150050B1 (ko) | 항체 정제 방법 | |
| US9266041B2 (en) | Chromatography ligand | |
| CN101043947B (zh) | 一种纯化抗体的方法 | |
| JP4739233B2 (ja) | 免疫グロブリンの精製 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20080919 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20080919 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20081008 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20110114 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20110114 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20110628 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20110928 |
|
| A602 | Written permission of extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A602 Effective date: 20111005 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20111227 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20120619 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120620 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20120710 |
|
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20120808 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 5064225 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| FPAY | Renewal fee payment (event date is renewal date of database) |
Free format text: PAYMENT UNTIL: 20150817 Year of fee payment: 3 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313113 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| S533 | Written request for registration of change of name |
Free format text: JAPANESE INTERMEDIATE CODE: R313533 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |