WO2022231003A1 - Kneaded meat product with improved quality - Google Patents
Kneaded meat product with improved quality Download PDFInfo
- Publication number
- WO2022231003A1 WO2022231003A1 PCT/JP2022/019450 JP2022019450W WO2022231003A1 WO 2022231003 A1 WO2022231003 A1 WO 2022231003A1 JP 2022019450 W JP2022019450 W JP 2022019450W WO 2022231003 A1 WO2022231003 A1 WO 2022231003A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- phospholipase
- heating
- weight
- yield
- meat
- Prior art date
Links
- 235000013622 meat product Nutrition 0.000 title abstract 5
- 235000013372 meat Nutrition 0.000 claims abstract description 91
- 102000011420 Phospholipase D Human genes 0.000 claims abstract description 78
- 108090000553 Phospholipase D Proteins 0.000 claims abstract description 78
- 239000002994 raw material Substances 0.000 claims abstract description 46
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 claims abstract description 40
- 229960001231 choline Drugs 0.000 claims abstract description 39
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 claims abstract description 37
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 claims abstract description 37
- 229960000367 inositol Drugs 0.000 claims abstract description 37
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 claims abstract description 37
- 238000000034 method Methods 0.000 claims abstract description 32
- 238000010438 heat treatment Methods 0.000 claims description 97
- 102000004190 Enzymes Human genes 0.000 claims description 84
- 108090000790 Enzymes Proteins 0.000 claims description 78
- 229940023462 paste product Drugs 0.000 claims description 41
- 108090001060 Lipase Proteins 0.000 claims description 34
- 102000004882 Lipase Human genes 0.000 claims description 34
- 239000004367 Lipase Substances 0.000 claims description 34
- 235000019421 lipase Nutrition 0.000 claims description 34
- 108060008539 Transglutaminase Proteins 0.000 claims description 31
- 102000003601 transglutaminase Human genes 0.000 claims description 31
- 244000144972 livestock Species 0.000 claims description 28
- 230000003625 amylolytic effect Effects 0.000 claims description 21
- 150000002327 glycerophospholipids Chemical class 0.000 claims description 6
- 150000003904 phospholipids Chemical class 0.000 claims description 6
- 241001446247 uncultured actinomycete Species 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 abstract description 70
- 238000010411 cooking Methods 0.000 abstract 3
- 229940088598 enzyme Drugs 0.000 description 82
- 230000000052 comparative effect Effects 0.000 description 61
- 238000012360 testing method Methods 0.000 description 40
- 235000013580 sausages Nutrition 0.000 description 26
- 108010028144 alpha-Glucosidases Proteins 0.000 description 24
- 102100024295 Maltase-glucoamylase Human genes 0.000 description 23
- 238000010304 firing Methods 0.000 description 22
- 239000000047 product Substances 0.000 description 17
- 238000001816 cooling Methods 0.000 description 16
- 238000011156 evaluation Methods 0.000 description 12
- 239000000243 solution Substances 0.000 description 12
- 238000004364 calculation method Methods 0.000 description 11
- 235000015220 hamburgers Nutrition 0.000 description 11
- 235000020995 raw meat Nutrition 0.000 description 11
- 241000287828 Gallus gallus Species 0.000 description 10
- 235000015278 beef Nutrition 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 235000013330 chicken meat Nutrition 0.000 description 10
- 238000001035 drying Methods 0.000 description 10
- 235000013305 food Nutrition 0.000 description 8
- 238000005259 measurement Methods 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 235000020991 processed meat Nutrition 0.000 description 8
- SXAMGRAIZSSWIH-UHFFFAOYSA-N 2-[3-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-1,2,4-oxadiazol-5-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C1=NOC(=N1)CC(=O)N1CC2=C(CC1)NN=N2 SXAMGRAIZSSWIH-UHFFFAOYSA-N 0.000 description 7
- WWSJZGAPAVMETJ-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-3-ethoxypyrazol-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C(=NN(C=1)CC(=O)N1CC2=C(CC1)NN=N2)OCC WWSJZGAPAVMETJ-UHFFFAOYSA-N 0.000 description 7
- FYELSNVLZVIGTI-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-5-ethylpyrazol-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C=NN(C=1CC)CC(=O)N1CC2=C(CC1)NN=N2 FYELSNVLZVIGTI-UHFFFAOYSA-N 0.000 description 7
- CONKBQPVFMXDOV-QHCPKHFHSA-N 6-[(5S)-5-[[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]methyl]-2-oxo-1,3-oxazolidin-3-yl]-3H-1,3-benzoxazol-2-one Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C[C@H]1CN(C(O1)=O)C1=CC2=C(NC(O2)=O)C=C1 CONKBQPVFMXDOV-QHCPKHFHSA-N 0.000 description 7
- 230000002255 enzymatic effect Effects 0.000 description 7
- 238000000465 moulding Methods 0.000 description 7
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 7
- 230000000694 effects Effects 0.000 description 6
- 238000002156 mixing Methods 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- HMUNWXXNJPVALC-UHFFFAOYSA-N 1-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)C(CN1CC2=C(CC1)NN=N2)=O HMUNWXXNJPVALC-UHFFFAOYSA-N 0.000 description 5
- YJLUBHOZZTYQIP-UHFFFAOYSA-N 2-[5-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-1,3,4-oxadiazol-2-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C1=NN=C(O1)CC(=O)N1CC2=C(CC1)NN=N2 YJLUBHOZZTYQIP-UHFFFAOYSA-N 0.000 description 5
- 241000234282 Allium Species 0.000 description 5
- 235000002732 Allium cepa var. cepa Nutrition 0.000 description 5
- HEMHJVSKTPXQMS-UHFFFAOYSA-M sodium hydroxide Inorganic materials [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 238000002835 absorbance Methods 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 238000007872 degassing Methods 0.000 description 4
- 235000014113 dietary fatty acids Nutrition 0.000 description 4
- 229930195729 fatty acid Natural products 0.000 description 4
- 239000000194 fatty acid Substances 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 239000004615 ingredient Substances 0.000 description 4
- 235000020997 lean meat Nutrition 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- 235000018102 proteins Nutrition 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 150000003839 salts Chemical class 0.000 description 4
- 239000012086 standard solution Substances 0.000 description 4
- WZFUQSJFWNHZHM-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)CC(=O)N1CC2=C(CC1)NN=N2 WZFUQSJFWNHZHM-UHFFFAOYSA-N 0.000 description 3
- ZRPAUEVGEGEPFQ-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]pyrazol-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C=NN(C=1)CC(=O)N1CC2=C(CC1)NN=N2 ZRPAUEVGEGEPFQ-UHFFFAOYSA-N 0.000 description 3
- NEAQRZUHTPSBBM-UHFFFAOYSA-N 2-hydroxy-3,3-dimethyl-7-nitro-4h-isoquinolin-1-one Chemical compound C1=C([N+]([O-])=O)C=C2C(=O)N(O)C(C)(C)CC2=C1 NEAQRZUHTPSBBM-UHFFFAOYSA-N 0.000 description 3
- 102000003914 Cholinesterases Human genes 0.000 description 3
- 108090000322 Cholinesterases Proteins 0.000 description 3
- 102000008186 Collagen Human genes 0.000 description 3
- 108010035532 Collagen Proteins 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 238000011088 calibration curve Methods 0.000 description 3
- 229940048961 cholinesterase Drugs 0.000 description 3
- 229920001436 collagen Polymers 0.000 description 3
- 235000013601 eggs Nutrition 0.000 description 3
- 238000006911 enzymatic reaction Methods 0.000 description 3
- 150000004665 fatty acids Chemical class 0.000 description 3
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 3
- 235000015277 pork Nutrition 0.000 description 3
- 239000012488 sample solution Substances 0.000 description 3
- 230000000391 smoking effect Effects 0.000 description 3
- 238000010025 steaming Methods 0.000 description 3
- AZQWKYJCGOJGHM-UHFFFAOYSA-N 1,4-benzoquinone Chemical compound O=C1C=CC(=O)C=C1 AZQWKYJCGOJGHM-UHFFFAOYSA-N 0.000 description 2
- KZEVSDGEBAJOTK-UHFFFAOYSA-N 1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-2-[5-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]-1,3,4-oxadiazol-2-yl]ethanone Chemical compound N1N=NC=2CN(CCC=21)C(CC=1OC(=NN=1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)=O KZEVSDGEBAJOTK-UHFFFAOYSA-N 0.000 description 2
- JQMFQLVAJGZSQS-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]piperazin-1-yl]-N-(2-oxo-3H-1,3-benzoxazol-6-yl)acetamide Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)N1CCN(CC1)CC(=O)NC1=CC2=C(NC(O2)=O)C=C1 JQMFQLVAJGZSQS-UHFFFAOYSA-N 0.000 description 2
- 241000186361 Actinobacteria <class> Species 0.000 description 2
- 108010000659 Choline oxidase Proteins 0.000 description 2
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 2
- 206010016807 Fluid retention Diseases 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 102000003992 Peroxidases Human genes 0.000 description 2
- 108010009736 Protein Hydrolysates Proteins 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- 235000010724 Wisteria floribunda Nutrition 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 108090000637 alpha-Amylases Proteins 0.000 description 2
- 239000005018 casein Substances 0.000 description 2
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 2
- 235000021240 caseins Nutrition 0.000 description 2
- 238000004040 coloring Methods 0.000 description 2
- 238000004945 emulsification Methods 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 2
- 229940050410 gluconate Drugs 0.000 description 2
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 description 2
- 230000003301 hydrolyzing effect Effects 0.000 description 2
- 244000005700 microbiome Species 0.000 description 2
- 108040007629 peroxidase activity proteins Proteins 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 238000006276 transfer reaction Methods 0.000 description 2
- 238000009461 vacuum packaging Methods 0.000 description 2
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 235000015579 Alternanthera sessilis Nutrition 0.000 description 1
- 240000002930 Alternanthera sessilis Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 108010082495 Dietary Plant Proteins Proteins 0.000 description 1
- 239000004278 EU approved seasoning Substances 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 108010073178 Glucan 1,4-alpha-Glucosidase Proteins 0.000 description 1
- 102100022624 Glucoamylase Human genes 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 108010024636 Glutathione Proteins 0.000 description 1
- 108010068370 Glutens Proteins 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 108010028688 Isoamylase Proteins 0.000 description 1
- 102400000471 Isomaltase Human genes 0.000 description 1
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 1
- 235000006439 Lemna minor Nutrition 0.000 description 1
- 244000242291 Lemna paucicostata Species 0.000 description 1
- 101710098556 Lipase A Proteins 0.000 description 1
- 101710099648 Lysosomal acid lipase/cholesteryl ester hydrolase Proteins 0.000 description 1
- 102100026001 Lysosomal acid lipase/cholesteryl ester hydrolase Human genes 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 239000004368 Modified starch Substances 0.000 description 1
- 229920000881 Modified starch Polymers 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- 108010026867 Oligo-1,6-Glucosidase Proteins 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010034203 Pectus Carinatum Diseases 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- 102000015439 Phospholipases Human genes 0.000 description 1
- 108010064785 Phospholipases Proteins 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 235000014680 Saccharomyces cerevisiae Nutrition 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 102000014384 Type C Phospholipases Human genes 0.000 description 1
- 108010079194 Type C Phospholipases Proteins 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 125000002252 acyl group Chemical group 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 239000013566 allergen Substances 0.000 description 1
- 102000004139 alpha-Amylases Human genes 0.000 description 1
- WQZGKKKJIJFFOK-DVKNGEFBSA-N alpha-D-glucose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-DVKNGEFBSA-N 0.000 description 1
- 102000016679 alpha-Glucosidases Human genes 0.000 description 1
- 229940024171 alpha-amylase Drugs 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 108010019077 beta-Amylase Proteins 0.000 description 1
- 235000012813 breadcrumbs Nutrition 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical class OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 239000013065 commercial product Substances 0.000 description 1
- 108010032220 cyclomaltodextrinase Proteins 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 229960002433 cysteine Drugs 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 235000013364 duck meat Nutrition 0.000 description 1
- 235000014103 egg white Nutrition 0.000 description 1
- 210000000969 egg white Anatomy 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- -1 fatty acid esters Chemical class 0.000 description 1
- 235000013312 flour Nutrition 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 235000011194 food seasoning agent Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 239000006479 glucose peptone medium Substances 0.000 description 1
- 125000000404 glutamine group Chemical group N[C@@H](CCC(N)=O)C(=O)* 0.000 description 1
- 229960003180 glutathione Drugs 0.000 description 1
- 235000003969 glutathione Nutrition 0.000 description 1
- 235000021312 gluten Nutrition 0.000 description 1
- 235000015275 goose meat Nutrition 0.000 description 1
- 235000020993 ground meat Nutrition 0.000 description 1
- 239000002054 inoculum Substances 0.000 description 1
- 150000004698 iron complex Chemical class 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 235000015255 meat loaf Nutrition 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- HOVAGTYPODGVJG-ZFYZTMLRSA-N methyl alpha-D-glucopyranoside Chemical compound CO[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O HOVAGTYPODGVJG-ZFYZTMLRSA-N 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 235000019426 modified starch Nutrition 0.000 description 1
- 108010035855 neopullulanase Proteins 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 150000003905 phosphatidylinositols Chemical class 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 238000005554 pickling Methods 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 230000001376 precipitating effect Effects 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 239000003531 protein hydrolysate Substances 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 235000021067 refined food Nutrition 0.000 description 1
- 238000005096 rolling process Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 235000002639 sodium chloride Nutrition 0.000 description 1
- 235000011121 sodium hydroxide Nutrition 0.000 description 1
- 235000013599 spices Nutrition 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 235000014122 turkey meat Nutrition 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 229940100445 wheat starch Drugs 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L13/00—Meat products; Meat meal; Preparation or treatment thereof
- A23L13/60—Comminuted or emulsified meat products, e.g. sausages; Reformed meat from comminuted meat product
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L35/00—Food or foodstuffs not provided for in groups A23L5/00 – A23L33/00; Preparation or treatment thereof
Definitions
- the present invention relates to a livestock meat paste product with improved quality, specifically to a livestock meat paste product with improved yield after heating during production.
- meat paste products after molding the dough obtained by mixing minced meat and other raw materials, it is heated (for example, baked) to make the final product, but the yield after heating (relative to the weight before heating (before baking) If the ratio of the weight after heating (after baking) is high, the meat juice is trapped inside and is considered to be a desirable quality.
- Patent Literature 1 discloses a method for producing a processed meat food in which lipase is added to the food so that the fatty acid content per 1 mg of the food is 13 ⁇ g or more and 80 ⁇ g or less.
- Patent Document 2 describes a method for producing a processed meat food having a salt content of 0.1% to 1% by weight, comprising adding transglutaminase and gluconate or lipase to the processed meat food;
- a method for producing a processed meat food is disclosed, which comprises adding transglutaminase, gluconate and lipase to the processed meat food.
- Patent Document 3 in the production of processed meat products, by using phospholipase C and / or phospholipase D, texture such as hardness and suppleness of processed meat products is not deteriorated during storage of processed meat products. It is disclosed that separation of water can be suppressed.
- the purpose of the present invention is to provide a livestock meat paste product with improved yield after heating during production.
- the present inventors have made intensive studies to solve the above problems, and found new findings that meat paste products such as meatballs containing choline and inositol, which are free products from phospholipids, have a high yield after heating. Found it. Based on the above findings, the present inventors have made further intensive studies and completed the present invention.
- a livestock meat paste product containing either one or both of the following (1) and (2). (1) 65 to 9000 ppm by weight of choline; (2) 230 to 380 ppm by weight of inositol; [3] The meat paste product according to [1] or [2] above, wherein the choline and inositol are derived from phospholipids contained in raw materials of the meat paste product. [4] A method for improving the post-heating yield of a livestock meat paste product, comprising allowing phospholipase D to act on livestock meat that is a raw material (in this specification, "raw meat” is sometimes referred to as "raw material meat”).
- Phospholipase D is Sigma-Aldrich Phospholipase D Streptomyces derived (trade name), Nagase Chemtech Denazym PMD-P1 (trade name) and Asahi Kasei Pharma PHOSPHOLIPASE D [PLDP (T-39) ] (Glycerophospholipid specific) (trade name).
- the meat paste product of the present invention has an improved yield after heating during production, and is expected to have improved texture and taste (for example, improved juiciness).
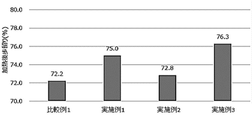
- FIG. 1 shows the results of calculation of the post-heating yield in the production of meatballs of Examples 1 to 3 and Comparative Example 1 in Test Example 1.
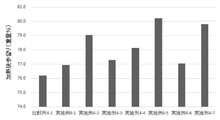
- FIG. FIG. 2 shows the results of calculation of the post-heating yield in the production of meatballs of Examples and Comparative Examples in Test Example 2.
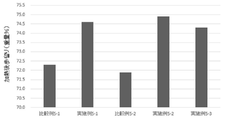
- FIG. 3 shows the results of calculation of the post-heating yield during production of the meatballs of Examples and Comparative Examples in Test Example 3.
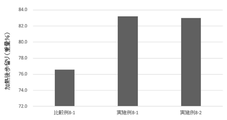
- FIG. 4 shows the results of calculation of post-heating yields in the production of coarsely ground sausages of Examples and Comparative Examples in Test Example 4.
- FIG. FIG. 5 shows the results of calculation of the post-heating yield in the production of meatballs of Examples and Comparative Examples in Test Example 5.
- FIG. FIG. 1 shows the results of calculation of the post-heating yield in the production of meatballs of Examples 1 to 3 and Comparative Example 1 in Test Example 1.
- FIG. 3 shows the results of calculation of the post-heating yield during production of the meatballs of Examples and Comparative Examples in Test Example 3.
- FIG. 6 shows the results of calculation of the post-heating yield in the production of meatballs of Examples and Comparative Examples in Test Example 6.
- FIG. 7 shows the results of calculation of the post-heating yield during production of the meatballs of Examples and Comparative Examples in Test Example 7.
- FIG. 8 shows the results of calculation of post-heating yields in the production of hamburger steaks of Examples and Comparative Examples in Test Example 8.
- FIG. 9 shows the results of calculation of post-heating yields in the production of coarsely ground sausages of Examples and Comparative Examples in Test Example 9.
- FIG. 10 shows the results of calculation of post-heating yields in the production of chicken meatballs of Examples and Comparative Examples in Test Example 10.
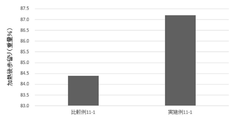
- FIG. 11 shows the results of calculation of post-heating yields during production of coarsely ground sausages of Examples and Comparative Examples in Test Example 11.
- FIG. 11 shows the results of calculation of post-heating yields during production of coarsely ground sausages of Examples and Comparative Examples in Test Example 11.
- the livestock meat paste product of the present invention is characterized by containing either one or both of (1) and (2) below.
- Examples of the livestock meat paste product of the present invention include a livestock meat paste product containing the above (1) and (2).
- Examples of the livestock meat paste product of the present invention include the following (i) to (iv).
- the content of choline and inositol in the livestock meat paste product can be measured by the method described in Test Example 1 below.
- the choline and inositol are preferably choline and inositol derived from phospholipids (phosphatidylcholine, phosphatidylinositol) contained in raw materials of the meat paste product.
- the "phospholipid contained in the raw material of the meat paste product” includes not only those derived from the raw meat of the meat paste product, but also other raw materials (e.g., eggs) that may optionally be contained in the meat paste product. It also includes those derived from The raw meat is the same as those exemplified in the description below.
- the choline and inositol may be added in the product manufacturing process (for example, the raw material mixing process) so that the choline and inositol contents in the final product are within the above ranges.
- the meat paste product of the present invention is a food made from ground meat as a main raw material, and examples thereof include meatballs, sausages (eg, coarsely ground sausages), hamburgers, chicken meatballs, ham, and meatloaf.
- the livestock meat paste product of the present invention contains livestock meat as a raw material.
- the raw meat include beef, pork, horse meat, mutton, goat meat, rabbit meat, chicken, turkey meat, duck meat, goose meat, and quail meat.
- the content of raw livestock meat is, for example, 0.1 to 99% by weight, preferably 1 to 97% by weight, more preferably 10 to 90% by weight, based on the raw materials (total weight) of the product. is.
- the meat paste product of the present invention contains ingredients commonly used in meat paste products (e.g., proteins such as onions, bread crumbs, eggs, vegetable proteins, gluten, egg whites, gelatin, and casein) and food additives.
- ingredients commonly used in meat paste products e.g., proteins such as onions, bread crumbs, eggs, vegetable proteins, gluten, egg whites, gelatin, and casein
- Substances e.g.
- Phospholipases are enzymes that have the activity of hydrolyzing phospholipids.
- Phospholipase D activity units herein are measured and defined as follows. 0.1 mL of the enzyme solution was mixed with 0.9 mL of the substrate solution containing phosphatidylcholine, reacted at 37° C. for 30 minutes, and after stopping the reaction, 50 ⁇ L of the reaction solution was added to 1 mL of the coloring solution containing choline oxidase, peroxidase, etc. and reacted for 5 minutes. After stopping the reaction, the amount of dye produced from choline is measured. 1 U (unit) is defined as the amount of enzyme that liberates 1 ⁇ mole of choline per minute at 37° C. using phosphatidylcholine as a substrate.
- the amount of phospholipase D added is preferably 0.001 U or more, more preferably 0.01 to 100 U, and still more preferably 0.1 to 50 U per 1 g of the raw material of the meat paste product.
- the action time (reaction time) of phospholipase D is not particularly limited as long as it is a time during which the enzyme can act on meat, which is a substrate substance. 10 minutes or longer, 20 minutes or longer, and 30 minutes or longer. Further, for example, 72 hours or less, 48 hours or less, 24 hours or less, 12 hours or less, 6 hours or less, 3 hours or less, 2 hours or less, and 1 hour or less. A realistic action time is preferably 0 to 72 hours, more preferably 30 minutes to 72 hours.
- the action temperature (reaction temperature) is not particularly limited as long as the enzyme maintains its activity. The enzymatic reaction can be terminated, for example, by heating at 70-75° C. for 5-10 minutes.
- At least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme is further added to the raw animal meat and allowed to act. is preferred.
- the order of addition may be in any order, and all may be added simultaneously or sequentially with a time lag. is desirable.
- at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme is allowed to further act on raw meat, the action time, action temperature, and method for terminating the enzymatic reaction are determined by the action time of phospholipase D described above.
- the following enzymes (I) to (VIII) are given as the enzymes to act on the raw meat.
- (I) Phospholipase D II) phospholipase D and transglutaminase
- III phospholipase D and lipase
- IV phospholipase D and amylolytic enzyme (eg ⁇ -glucosidase)
- V phospholipase D, transglutaminase and lipase
- VI phospholipase D, transglutaminase and amylolytic enzymes (e.g.
- VII Phospholipase D, lipase and amylolytic enzyme (eg ⁇ -glucosidase)
- VIII Phospholipase D, transglutaminase, lipase and amylolytic enzyme (eg ⁇ -glucosidase)
- the transglutaminase used in the present invention is an enzyme having the activity of catalyzing an acyl transfer reaction using a glutamine residue in a protein or peptide as a donor and a lysine residue as an acceptor, and is derived from mammals, for example. , those derived from fish, those derived from microorganisms, etc. are known.
- the origin of the transglutaminase used in the present invention is not particularly limited as long as it has the above-mentioned activity, and transglutaminase of any origin can be used, and recombinant enzymes can also be used.
- the transglutaminase used in the present invention may be a commercial product, and as a specific example, a microorganism-derived transglutaminase commercially available from Ajinomoto Co., Inc. under the trade name of "Activa" TG can be used alone or in combination. can.
- the enzymatic activity of transglutaminase is defined by allowing transglutaminase to act in a reaction system using benzyloxycarbonyl-L-glutamylglycine and hydroxylamine as substrates in a Tris buffer at a temperature of 37° C. and a pH of 6.0.
- the lipase used in the present invention is an enzyme that catalyzes the reaction of hydrolyzing fatty acid esters into fatty acids and glycerol.
- Enzymes commercially available from Amano Enzyme Co., Ltd. under the trade names of "Lipase A 'Amano'6" and “Lipase AY 'Amano'” are examples of lipases.
- the enzymatic activity of lipase was measured by emulsifying 100 ml of olive oil and 150 ml of a 2% PVA test solution as a substrate, mixing 5 ml of the substrate, 4 ml of McIlvaine buffer (pH 7.0) and 1 ml of the enzyme solution, followed by mixing at 37° C. for 60 minutes. After the reaction is terminated, the produced fatty acid is measured by a titration method. 1 U (unit) is defined as the acid liberating activity corresponding to 1 ⁇ mol of liberated oleic acid.
- an amylolytic enzyme is an enzyme that catalyzes the decomposition reaction and transfer reaction of sugar chains containing starch.
- amylolytic enzymes used in the present invention include ⁇ -glucosidase, ⁇ -amylase, ⁇ -amylase, glucoamylase, isomaltase, pullulanase, sucrose- ⁇ -glucosidase, ⁇ -glucosyltransferase, cyclomaltodextrinase, Examples include isoamylase and neopullulanase, with ⁇ -glucosidase being preferred.
- the ⁇ -glucosidase used in the present invention is an enzyme that hydrolyzes a non-reducing terminal ⁇ -1,4-glucosidic bond to produce ⁇ -glucose.
- transglucosidase is preferred.
- An enzyme commercially available from Amano Enzyme Co., Ltd. under the trade name of "Transglucosidase L 'Amano'" is an example of ⁇ -glucosidase.
- the enzymatic activity of ⁇ -glucosidase was measured by adding 1 ml of 0.02 M acetate buffer (pH 5.0) to 1 ml of 1 mM ⁇ -methyl-D-glucoside, adding 0.5 ml of the enzyme solution, and precipitating at 40° C. for 60 minutes.
- the amount of enzyme that produces 1 ⁇ g of glucose in 2.5 ml of reaction solution when acted upon is defined as 1 U (unit).
- the amount of transglutaminase to be added is such that the enzymatic activity is, for example, 0.005 to 3.0 U, preferably 0.01 to 1.0 U, per 1 g of the raw material for the meat paste product. 0 U, more preferably 0.05 to 0.5 U, still more preferably 0.1 to 0.3 U.
- the amount of lipase to be added is such that the enzymatic activity is, for example, 0.1 to 30.0 U, preferably 0.3 to 15.0 U, preferably 0.3 to 15.0 U, per 1 g of the raw material of the meat paste product. It is more preferably 0.5 to 10.0U, still more preferably 1.0 to 3.0U.
- the amount of the amylolytic enzyme (eg, ⁇ -glucosidase) added is such that the enzymatic activity per 1 g of the raw material of the meat paste product is, for example, It is 0.0005 to 0.1 U, preferably 0.001 to 0.05 U, more preferably 0.002 to 0.02 U, still more preferably 0.005 to 0.01 U.
- the meat paste product of the present invention produced by the above-described production method of the present invention has an improved yield after heating during production, and is expected to have an improved texture and taste.
- the present invention also relates to a method for improving the post-heating yield of livestock meat paste products, which comprises allowing phospholipase D to act on raw livestock meat.
- the phospholipase D is Sigma-Aldrich's phospholipase D actinomycete-derived (trade name), Nagase Chemtech's Denazym PMD-P1 (trade name), and Asahi Kasei Pharma's PHOSPHOLIPASE D [PLDP ( T-39)] (Glycerophospholipid specific) (trade names) are preferred.
- At least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme may be further acted on the raw meat.
- the following enzymes (I) to (VIII) are listed as enzymes to act on raw meat.
- VII Phospholipase D, lipase and amylolytic enzyme (eg ⁇ -glucosidase)
- VIII Phospholipase D, transglutaminase, lipase and amylolytic enzyme (eg ⁇ -glucosidase)
- examples of meat paste products examples of raw meat, amounts used, definitions, examples, addition amounts, and addition of phospholipase D, transglutaminase, lipase, and amylolytic enzyme ( ⁇ -glucosidase)
- the method, optional ingredients, additives, etc. are described in the method for producing the livestock meat paste product of the present invention, examples of livestock meat paste products, examples of raw meat paste products, amounts used, phospholipase D, transglutaminase, lipase and It is the same as the definition, exemplification, addition amount, addition method, optionally added components, additives, etc. of the amylolytic enzyme ( ⁇ -glucosidase).
- the post-heating yield of the meat paste product refers to the weight after heating (after baking) relative to the weight before heating (before baking) of the dough (putty) obtained by mixing the raw materials at the time of manufacturing the meat paste product. refers to the ratio (% by weight) of
- the improved yield after heating of the meat paste product means that the yield after heating is higher than that of the meat paste product produced without the addition of enzymes.
- Test Example 1 (Production of meatballs of Examples 1 to 3 and Comparative Example 1)
- the beef rib lean meat and beef Kenne fat shown in Table 1 were minced to 3 mm by a meat grinder (chopper).
- the onion was chopped into pieces of about 2 mm.
- Each phospholipase D shown in Table 2 was added to the raw material of the composition shown in Table 1 so as to be 3.0 U per 1 g of raw material, and a standard mixer ("KitchenAid KSM5WH", manufactured by Whirlpool Corporation) was added. and mixed for 3 minutes at a setting speed of 1 to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, baked at 250 ° C.
- cholinesterase kit-NC 289-75181, Fujifilm Wako Pure Chemical Industries, Ltd.
- a calibration curve was prepared from the absorbance, and the content of free choline in meatballs was determined. The results are shown in Table 5-2.
- the choline content in the meatball (estimated value: 60 ppm by weight) is the amount of choline originally contained in the raw material.
- the choline content in the meatballs of Examples 2-1 to 2-5 and Comparative Examples 2-2 to 2-4 shown in Tables 7-1 and 7-2 is the choline originally contained in the raw materials. It is the sum of the amount (estimated value: 60 ppm by weight) and the added amount of choline. (Evaluation of yield after heating) In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production.
- Test Example 3 Inositol addition test (production of meatballs of Examples 3-1 to 3-3 and Comparative Examples 3-2 to 3-7)
- the beef rib lean meat and beef kenne fat shown in Table 6 of Test Example 2 were minced to 3 mm by a meat grinder (chopper). The onion was chopped into pieces of about 2 mm.
- a standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) was prepared using raw materials with the formulations shown in Table 6 of Test Example 2 and inositol in the amounts shown in Tables 8-1 and 8-2. was mixed for 3 minutes at a setting speed of 1 to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres.
- the inositol content in the meatball of Comparative Example 3-1 shown in Table 8-1 is the inositol content in the meatball of Comparative Example 1 of Test Example 1 produced by the same method (213 ppm by weight, Table 5- 3). Since inositol was not added in Comparative Example 3-1, the content of inositol in the meatball (estimated value: 213 ppm by weight) is the amount of inositol originally contained in the raw material.
- the inositol content in the meatballs of Examples 3-1 to 3-3 and Comparative Examples 3-2 to 3-7 shown in Tables 8-1 and 8-2 is the inositol originally contained in the raw materials.
- the collagen casing manufactured by Devro Co., Ltd.
- coarsely ground sausages of Examples 4-1 to 4-7 were produced.
- a coarsely ground sausage of Comparative Example 4-1 was obtained in the same manner as in Examples 4-1 to 4-7 except that no enzyme was added.
- evaluation of yield after heating In order to evaluate the post-heating yield, the weight before drying and the weight after cooling (that is, after the drying-smoking-steaming-cooling process) were measured during the production of the coarsely ground sausage.
- a standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) was used and mixed at a setting speed of 1 for 3 minutes to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, it was allowed to stand in a refrigerator for 1 hour. Baking at 250 ° C. for 1 minute with an impinger (FGJOA 9L, manufactured by Fujimac) and baking at 200 ° C. and RH 50% for 6 minutes in a steam convection oven (FSCC WE 61G, manufactured by Fujimac), Examples 5-1 to 5 -3 meatballs were obtained.
- FGJOA 9L an impinger
- FSCC WE 61G steam convection oven
- Example 5-1 produced by adding phospholipase D
- Examples 5-2 and 5-3 produced by adding phospholipase D and transglutaminase were Compared to Comparative Example 5-1 without enzyme addition and Comparative Example 5-2 without phospholipase D addition, the post-heating yield was higher, and an improvement in post-heating yield was observed.
- Example 6-1 produced by adding phospholipase D and Examples 6-2 to 6-4 produced by adding phospholipase D and ⁇ -glucosidase , Compared to Comparative Example 6-1 without enzyme addition and Comparative Example 6-2 without phospholipase D addition, the yield after heating was high, and an improvement in yield after heating was observed.
- Example 7 Enzyme addition test (combined use of phospholipase D, ⁇ -glucosidase and lipase (production of meatballs of Example 7-1) A meatball of Example 7-1 was obtained in the same manner as in Example 5-1, etc., except that the enzyme was changed to the enzyme and the amount shown in Table 14.
- Enzymes include phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Co., Ltd.), ⁇ -glucosidase (Transglucosidase L "Amano”, manufactured by Amano Enzyme Co., Ltd.) and lipase (Lipase AY "Amano", manufactured by Amano Enzyme Co., Ltd.). )It was used.
- Production of meatballs in Comparative Example 7-1 A meatball of Comparative Example 7-1 was obtained in the same manner as in Example 7-1 except that no enzyme was added.
- evaluation of yield after heating In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. The post-heating yield was calculated in the same manner as in Test Example 5. The results are shown in Table 14 and FIG.
- Example 7-1 produced by adding phospholipase D, ⁇ -glucosidase and lipase had a higher yield after heating than Comparative Example 7-1 without enzyme addition. was high, and an improvement in yield was observed after heating.
- the dough was weighed to 150 g, and after the air was removed, it was formed into an oval shape (major axis: 130 mm, minor axis: 90 mm, thickness: 12 mm).
- the hamburger dough is baked on a griddle (FGFT60601TC, Fujimac Co., Ltd.) at 230 ° C. for 1 minute each on both sides, and after the surface is baked, baked at 250 ° C. for 4 minutes with an impinger (FGJOA9L, Fujimac Co., Ltd.).
- FGJOA9L Fujimac Co., Ltd.
- Example 9-1 produced by adding phospholipase D has a higher yield after heating than Comparative Example 9-1 without enzyme addition, and the yield after heating is improvement was observed.
- Example 10-1 produced by adding phospholipase D had a higher yield after heating than Comparative Example 10-1 without enzyme addition, and a lower yield after heating. Improvement was observed.
- Example 11-1 produced by adding phospholipase D has a high yield after heating and a high yield after heating compared to Comparative Example 11-1 without enzyme addition. improvement was observed.
Abstract
Description
また、畜肉練り製品においては、挽肉及びその他の原材料を混合して得られた生地を成形後、加熱(例えば焼成)して最終製品とするが、加熱後歩留り(加熱前(焼成前)の重量に対する加熱後(焼成後)の重量の割合)が高いと、肉汁が内部に閉じ込められており好ましい品質と考えられる。 Normally, when producing meat paste products such as sausages and meatballs, raw minced meat and salts are mixed to elute salt-soluble proteins to promote emulsification. Promoting emulsification affects the hardness and water retention of meat paste products, but existing emulsification-promoting ingredients such as emulsifiers, eggs, and milk casein cannot be used due to the need for labeling on final products and from the perspective of allergens. was difficult.
In addition, in meat paste products, after molding the dough obtained by mixing minced meat and other raw materials, it is heated (for example, baked) to make the final product, but the yield after heating (relative to the weight before heating (before baking) If the ratio of the weight after heating (after baking) is high, the meat juice is trapped inside and is considered to be a desirable quality.
特許文献1には、食品にリパーゼを添加し、食品1mgあたりの脂肪酸含有量を13μg以上、80μg以下とする、食肉加工食品の製造方法が開示されている。
特許文献2には、食塩の含有量が0.1重量%~1重量%である食肉加工食品の製造方法であって、トランスグルタミナーゼとグルコン酸塩もしくはリパーゼとを食肉加工食品に添加すること、またはトランスグルタミナーゼとグルコン酸塩およびリパーゼとを食肉加工食品に添加することを含む、食肉加工食品の製造方法が開示されている。
特許文献3には、畜肉加工製品の製造において、ホスホリパーゼC及び/又はホスホリパーゼDを用いることにより、畜肉加工製品の硬さ、しなやかさ等の食感を劣化させることなく、畜肉加工製品の保存時の離水を抑制することができることが開示されている。 For the purpose of improving the hardness and water retention of livestock meat paste products, methods using enzymes during production have been investigated.
Patent Literature 1 discloses a method for producing a processed meat food in which lipase is added to the food so that the fatty acid content per 1 mg of the food is 13 µg or more and 80 µg or less.
Patent Document 2 describes a method for producing a processed meat food having a salt content of 0.1% to 1% by weight, comprising adding transglutaminase and gluconate or lipase to the processed meat food; Alternatively, a method for producing a processed meat food is disclosed, which comprises adding transglutaminase, gluconate and lipase to the processed meat food.
In Patent Document 3, in the production of processed meat products, by using phospholipase C and / or phospholipase D, texture such as hardness and suppleness of processed meat products is not deteriorated during storage of processed meat products. It is disclosed that separation of water can be suppressed.
上記知見に基づいて、本発明者らは、さらに鋭意検討をして、本発明を完成させた。 The present inventors have made intensive studies to solve the above problems, and found new findings that meat paste products such as meatballs containing choline and inositol, which are free products from phospholipids, have a high yield after heating. Found it.
Based on the above findings, the present inventors have made further intensive studies and completed the present invention.
[1]下記(1)、(2)のいずれか一方又は両方を含有する、畜肉練り製品。
(1)65~9000重量ppmのコリン
(2)230~380重量ppmのイノシトール
[2]前記(1)及び(2)を含有する、上記[1]記載の畜肉練り製品。
[3]前記コリン及びイノシトールが、畜肉練り製品の原材料に含まれるリン脂質に由来する、上記[1]又は[2]に記載の畜肉練り製品。
[4]原材料である畜肉(本明細書において「原材料である畜肉」を「原料畜肉」と称する場合がある。)にホスホリパーゼDを作用させることを含む、畜肉練り製品の加熱後歩留り向上方法。
[5]ホスホリパーゼDが、シグマ アルドリッチ(Sigma-Aldrich)製ホスホリパーゼD 放線菌由来(商品名)、ナガセケムテック製デナチームPMD-P1(商品名)及び旭化成ファーマ製PHOSPHOLIPASE D[PLDP(T-39)](Glycerophospholipid specific)(商品名)からなる群から選択される、上記[4]記載の方法。
[6]トランスグルタミナーゼ、リパーゼ、及び澱粉分解酵素からなる群から選択される少なくとも1つの酵素をさらに作用させることを含む、上記[4]または[5]に記載の方法。
[7]原材料である畜肉に、シグマ アルドリッチ製ホスホリパーゼD 放線菌由来(商品名)、ナガセケムテック製デナチームPMD-P1(商品名)及び旭化成ファーマ製PHOSPHOLIPASE D[PLDP(T-39)](Glycerophospholipid specific)(商品名)からなる群から選択されるホスホリパーゼDを作用させることを含む、上記[1]~[3]のいずれかに記載の畜肉練り製品の製造方法。
[8]トランスグルタミナーゼ、リパーゼ、及び澱粉分解酵素からなる群から選択される少なくとも1つの酵素をさらに作用させることを含む、上記[7]記載の方法。
[9]原材料である畜肉に、シグマ アルドリッチ製ホスホリパーゼD 放線菌由来(商品名)、ナガセケムテック製デナチームPMD-P1(商品名)及び旭化成ファーマ製PHOSPHOLIPASE D[PLDP(T-39)](Glycerophospholipid specific)(商品名)からなる群から選択されるホスホリパーゼDを作用させることを含む、畜肉練り製品の製造方法。
[10]トランスグルタミナーゼ、リパーゼ、及び澱粉分解酵素からなる群から選択される少なくとも1つの酵素をさらに作用させることを含む、上記[9]記載の方法。 That is, the present invention provides the following.
[1] A livestock meat paste product containing either one or both of the following (1) and (2).
(1) 65 to 9000 ppm by weight of choline; (2) 230 to 380 ppm by weight of inositol;
[3] The meat paste product according to [1] or [2] above, wherein the choline and inositol are derived from phospholipids contained in raw materials of the meat paste product.
[4] A method for improving the post-heating yield of a livestock meat paste product, comprising allowing phospholipase D to act on livestock meat that is a raw material (in this specification, "raw meat" is sometimes referred to as "raw material meat").
[5] Phospholipase D is Sigma-Aldrich Phospholipase D Streptomyces derived (trade name), Nagase Chemtech Denazym PMD-P1 (trade name) and Asahi Kasei Pharma PHOSPHOLIPASE D [PLDP (T-39) ] (Glycerophospholipid specific) (trade name).
[6] The method according to [4] or [5] above, which further comprises allowing at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme to act.
[7] For livestock meat, which is a raw material, Phospholipase D derived from actinomycetes (trade name) manufactured by Sigma-Aldrich, Denathym PMD-P1 (trade name) manufactured by Nagase Chemtech, and PHOSPHOLIPASE D [PLDP (T-39)] (Glycerophospholipid) manufactured by Asahi Kasei Pharma specific) (trade name).
[8] The method according to [7] above, which further comprises allowing at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme to act.
[9] For livestock meat, which is a raw material, Phospholipase D derived from actinomycetes (trade name) manufactured by Sigma-Aldrich, Denathym PMD-P1 (trade name) manufactured by Nagase Chemtech, and PHOSPHOLIPASE D [PLDP (T-39)] (Glycerophospholipid) manufactured by Asahi Kasei Pharma specific) (trade name).
[10] The method according to [9] above, which further comprises allowing at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme to act.
本発明の畜肉練り製品は、以下(1)、(2)のいずれか一方又は両方を含有することを特徴とする。
(1)65~9000重量ppm(好ましくは80~7000重量ppm、より好ましくは100~5000重量ppm、さらに好ましくは260~2060重量ppm)のコリン
(2)230~380重量ppm(好ましくは235~363重量ppm、より好ましくは240~320重量ppm、さらに好ましくは243~273重量ppm)のイノシトール The present invention will be described in detail below.
The livestock meat paste product of the present invention is characterized by containing either one or both of (1) and (2) below.
(1) 65 to 9000 ppm by weight (preferably 80 to 7000 ppm by weight, more preferably 100 to 5000 ppm by weight, more preferably 260 to 2060 ppm by weight) of choline; 363 ppm by weight, more preferably 240 to 320 ppm by weight, more preferably 243 to 273 ppm by weight) of inositol
本発明の畜肉練り製品としては、例えば、以下(i)~(iv)が挙げられる。
(i)(1)65~9000重量ppmのコリン及び(2)230~380重量ppmのイノシトールを含有する、畜肉練り製品。
(ii)(1)80~7000重量ppmのコリン及び(2)235~363重量ppmのイノシトールを含有する、畜肉練り製品。
(iii)(1)100~5000重量ppmのコリン及び(2)240~320重量ppmのイノシトールを含有する、畜肉練り製品。
(iv)(1)260~2060重量ppmのコリン及び(2)243~273重量ppmのイノシトールを含有する、畜肉練り製品。 Examples of the livestock meat paste product of the present invention include a livestock meat paste product containing the above (1) and (2).
Examples of the livestock meat paste product of the present invention include the following (i) to (iv).
(i) A meat paste containing (1) 65-9000 ppm by weight choline and (2) 230-380 ppm by weight inositol.
(ii) A meat paste containing (1) 80-7000 ppm by weight choline and (2) 235-363 ppm by weight inositol.
(iii) A livestock meat paste product containing (1) 100 to 5000 ppm by weight of choline and (2) 240 to 320 ppm by weight of inositol.
(iv) A meat paste containing (1) 260-2060 ppm by weight choline and (2) 243-273 ppm by weight inositol.
本発明の畜肉練り製品において、前記コリン、イノシトールは、最終製品中のコリン、イノシトール含有量が上記した範囲となるように、製品の製造工程(例えば、原材料の混合工程)で添加してもよい。 In the meat paste product of the present invention, the choline and inositol are preferably choline and inositol derived from phospholipids (phosphatidylcholine, phosphatidylinositol) contained in raw materials of the meat paste product. In the present invention, the "phospholipid contained in the raw material of the meat paste product" includes not only those derived from the raw meat of the meat paste product, but also other raw materials (e.g., eggs) that may optionally be contained in the meat paste product. It also includes those derived from The raw meat is the same as those exemplified in the description below.
In the livestock meat paste product of the present invention, the choline and inositol may be added in the product manufacturing process (for example, the raw material mixing process) so that the choline and inositol contents in the final product are within the above ranges.
本発明の畜肉練り製品において、原料畜肉の含有量は、製品の原材料(合計重量)に対して、例えば0.1~99重量%、好ましくは1~97重量%、より好ましくは10~90重量%である。 The livestock meat paste product of the present invention contains livestock meat as a raw material. Examples of the raw meat include beef, pork, horse meat, mutton, goat meat, rabbit meat, chicken, turkey meat, duck meat, goose meat, and quail meat.
In the livestock meat paste product of the present invention, the content of raw livestock meat is, for example, 0.1 to 99% by weight, preferably 1 to 97% by weight, more preferably 10 to 90% by weight, based on the raw materials (total weight) of the product. is.
本明細書においてホスホリパーゼDの活性単位は、次のように測定され、かつ、定義される。
ホスファチジルコリンを含む基質溶液0.9mLに酵素溶液0.1mLを混和し、37℃にて30分間反応させ、反応停止後、コリンオキシダーゼ、ペルオキシダーゼ等を含む発色溶液1mLに反応液50μLを加え5分反応させ、反応停止後、コリンより生成した色素量を測定する。ホスファチジルコリンを基質として37℃、1分間に1μモルのコリンを遊離する酵素量を1U(ユニット)と定義する。 Phospholipases are enzymes that have the activity of hydrolyzing phospholipids.
Phospholipase D activity units herein are measured and defined as follows.
0.1 mL of the enzyme solution was mixed with 0.9 mL of the substrate solution containing phosphatidylcholine, reacted at 37° C. for 30 minutes, and after stopping the reaction, 50 μL of the reaction solution was added to 1 mL of the coloring solution containing choline oxidase, peroxidase, etc. and reacted for 5 minutes. After stopping the reaction, the amount of dye produced from choline is measured. 1 U (unit) is defined as the amount of enzyme that liberates 1 μmole of choline per minute at 37° C. using phosphatidylcholine as a substrate.
複数の酵素を添加する場合の添加順序は、いかなる順序でもよく、全てを同時に添加しても、時間差をつけて順に添加しても構わないが、簡便性の観点から、全てを同時に添加するのが望ましい。
トランスグルタミナーゼ、リパーゼ、及び澱粉分解酵素からなる群から選択される少なくとも1つの酵素をさらに原料畜肉に作用させる場合の、作用時間、作用温度、酵素反応の終了方法は、上記したホスホリパーゼDの作用時間、作用温度、酵素反応の終了方法と同じである。
本発明の製造方法において、原料畜肉に作用させるための酵素としては、以下(I)~(VIII)が挙げられる。
(I)ホスホリパーゼD
(II)ホスホリパーゼD及びトランスグルタミナーゼ
(III)ホスホリパーゼD及びリパーゼ
(IV)ホスホリパーゼD及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(V)ホスホリパーゼD、トランスグルタミナーゼ及びリパーゼ
(VI)ホスホリパーゼD、トランスグルタミナーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(VII)ホスホリパーゼD、リパーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(VIII)ホスホリパーゼD、トランスグルタミナーゼ、リパーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ) In the above-described production method of the present invention, in addition to the above-described phospholipase D, at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme is further added to the raw animal meat and allowed to act. is preferred.
When a plurality of enzymes are added, the order of addition may be in any order, and all may be added simultaneously or sequentially with a time lag. is desirable.
When at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme is allowed to further act on raw meat, the action time, action temperature, and method for terminating the enzymatic reaction are determined by the action time of phospholipase D described above. , the working temperature, and the termination method of the enzymatic reaction.
In the production method of the present invention, the following enzymes (I) to (VIII) are given as the enzymes to act on the raw meat.
(I) Phospholipase D
(II) phospholipase D and transglutaminase (III) phospholipase D and lipase (IV) phospholipase D and amylolytic enzyme (eg α-glucosidase)
(V) phospholipase D, transglutaminase and lipase (VI) phospholipase D, transglutaminase and amylolytic enzymes (e.g. α-glucosidase)
(VII) Phospholipase D, lipase and amylolytic enzyme (eg α-glucosidase)
(VIII) Phospholipase D, transglutaminase, lipase and amylolytic enzyme (eg α-glucosidase)
本明細書において、トランスグルタミナーゼの酵素活性は、温度37℃、pH6.0のトリス緩衝液中、ベンジルオキシカルボニル-L-グルタミルグリシンおよびヒドロキシルアミンを基質とする反応系で、トランスグルタミナーゼを作用せしめ、生成したヒドロキサム酸をトリクロロ酢酸存在下で鉄錯体を形成させた後、525nmにおける吸光度を測定し、ヒドロキサム酸量を検量線により求め、1分間に1μモルのヒドロキサム酸を生成せしめる酵素量を1ユニット(1U)と定義する(特開昭64-27471号公報参照)。 The transglutaminase used in the present invention is an enzyme having the activity of catalyzing an acyl transfer reaction using a glutamine residue in a protein or peptide as a donor and a lysine residue as an acceptor, and is derived from mammals, for example. , those derived from fish, those derived from microorganisms, etc. are known. The origin of the transglutaminase used in the present invention is not particularly limited as long as it has the above-mentioned activity, and transglutaminase of any origin can be used, and recombinant enzymes can also be used. The transglutaminase used in the present invention may be a commercial product, and as a specific example, a microorganism-derived transglutaminase commercially available from Ajinomoto Co., Inc. under the trade name of "Activa" TG can be used alone or in combination. can.
As used herein, the enzymatic activity of transglutaminase is defined by allowing transglutaminase to act in a reaction system using benzyloxycarbonyl-L-glutamylglycine and hydroxylamine as substrates in a Tris buffer at a temperature of 37° C. and a pH of 6.0. After forming an iron complex with the produced hydroxamic acid in the presence of trichloroacetic acid, the absorbance at 525 nm is measured, the amount of hydroxamic acid is obtained from a calibration curve, and the amount of enzyme that produces 1 μmole of hydroxamic acid per minute is 1 unit. (1U) (see JP-A-64-27471).
本明細書において、リパーゼの酵素活性は、オリーブ油100mlと2%PVA試液150mlを乳化させ基質とし、基質5ml、マッキルベイン緩衝液(pH7.0)4ml及び酵素液1mlを混和し、37℃にて60分間反応させ、反応停止後、生成した脂肪酸を滴定法で測定する。遊離したオレイン酸1μmolに相当する酸を遊離させる活性を1U(ユニット)と定義する。 The lipase used in the present invention is an enzyme that catalyzes the reaction of hydrolyzing fatty acid esters into fatty acids and glycerol. Enzymes commercially available from Amano Enzyme Co., Ltd. under the trade names of "Lipase A 'Amano'6" and "Lipase AY 'Amano'" are examples of lipases.
In the present specification, the enzymatic activity of lipase was measured by emulsifying 100 ml of olive oil and 150 ml of a 2% PVA test solution as a substrate, mixing 5 ml of the substrate, 4 ml of McIlvaine buffer (pH 7.0) and 1 ml of the enzyme solution, followed by mixing at 37° C. for 60 minutes. After the reaction is terminated, the produced fatty acid is measured by a titration method. 1 U (unit) is defined as the acid liberating activity corresponding to 1 μmol of liberated oleic acid.
本明細書において、α-グルコシダーゼの酵素活性は、1mM α-メチル-D-グルコシド1mlに0.02M酢酸バッファー(pH5.0)1mlを加え、酵素溶液0.5ml添加して、40℃60分間作用させたときに、反応液2.5ml中に1μgのブドウ糖を生成する酵素量を1U(ユニット)と定義する。 The α-glucosidase used in the present invention is an enzyme that hydrolyzes a non-reducing terminal α-1,4-glucosidic bond to produce α-glucose. Among α-glucosidases, transglucosidase is preferred. An enzyme commercially available from Amano Enzyme Co., Ltd. under the trade name of "Transglucosidase L 'Amano'" is an example of α-glucosidase.
As used herein, the enzymatic activity of α-glucosidase was measured by adding 1 ml of 0.02 M acetate buffer (pH 5.0) to 1 ml of 1 mM α-methyl-D-glucoside, adding 0.5 ml of the enzyme solution, and precipitating at 40° C. for 60 minutes. The amount of enzyme that produces 1 μg of glucose in 2.5 ml of reaction solution when acted upon is defined as 1 U (unit).
本発明の、該加熱後歩留り向上方法において、ホスホリパーゼDは、シグマ アルドリッチ製ホスホリパーゼD 放線菌由来(商品名)、ナガセケムテック製デナチームPMD-P1(商品名)及び旭化成ファーマ製PHOSPHOLIPASE D[PLDP(T-39)](Glycerophospholipid specific)(商品名)からなる群から選択されるホスホリパーゼDが好ましい。 The present invention also relates to a method for improving the post-heating yield of livestock meat paste products, which comprises allowing phospholipase D to act on raw livestock meat.
In the method for improving the post-heating yield of the present invention, the phospholipase D is Sigma-Aldrich's phospholipase D actinomycete-derived (trade name), Nagase Chemtech's Denazym PMD-P1 (trade name), and Asahi Kasei Pharma's PHOSPHOLIPASE D [PLDP ( T-39)] (Glycerophospholipid specific) (trade names) are preferred.
本発明の、該加熱後歩留り向上方法において、原料畜肉に作用させるための酵素としては、以下(I)~(VIII)が挙げられる。
(I)ホスホリパーゼD
(II)ホスホリパーゼD及びトランスグルタミナーゼ
(III)ホスホリパーゼD及びリパーゼ
(IV)ホスホリパーゼD及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(V)ホスホリパーゼD、トランスグルタミナーゼ及びリパーゼ
(VI)ホスホリパーゼD、トランスグルタミナーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(VII)ホスホリパーゼD、リパーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ)
(VIII)ホスホリパーゼD、トランスグルタミナーゼ、リパーゼ及び澱粉分解酵素(例えば、α-グルコシダーゼ) In the method for improving the post-heating yield of the present invention, in addition to the above-described phospholipase D, at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme may be further acted on the raw meat. preferable.
In the method for improving yield after heating according to the present invention, the following enzymes (I) to (VIII) are listed as enzymes to act on raw meat.
(I) Phospholipase D
(II) phospholipase D and transglutaminase (III) phospholipase D and lipase (IV) phospholipase D and amylolytic enzyme (eg α-glucosidase)
(V) phospholipase D, transglutaminase and lipase (VI) phospholipase D, transglutaminase and amylolytic enzymes (e.g. α-glucosidase)
(VII) Phospholipase D, lipase and amylolytic enzyme (eg α-glucosidase)
(VIII) Phospholipase D, transglutaminase, lipase and amylolytic enzyme (eg α-glucosidase)
本発明において、畜肉練り製品の加熱後歩留り向上とは、酵素無添加で製造した畜肉練り製品と比較して、加熱後歩留まりが高いことをいう。 In this specification, the post-heating yield of the meat paste product refers to the weight after heating (after baking) relative to the weight before heating (before baking) of the dough (putty) obtained by mixing the raw materials at the time of manufacturing the meat paste product. refers to the ratio (% by weight) of
In the present invention, the improved yield after heating of the meat paste product means that the yield after heating is higher than that of the meat paste product produced without the addition of enzymes.
(実施例1~3、比較例1のミートボールの製造)
表1に示した牛バラ赤身、牛ケンネ脂を肉挽き機(チョッパー)により3mmのミンチとした。玉ねぎは約2mmのみじん切りとした。
表1に示した配合の原材料に、表2に示した各ホスホリパーゼDを原材料1gあたり3.0Uとなるよう添加し、スタンダードミキサー(「キッチンエイド(KitchenAid)KSM5WH」、ワールプール・コーポレーション製)を用い、設定速度1にて3分間混合し、ミートボール生地を調製した。空気抜きした後に30gの球形に手で成型した。成型後、インピンジャー(FGJOA9L、フジマック製)にて250℃で1分間焼成及びスチームコンベクションオーブン(FSCC WE 61G、フジマック製)にて200℃、RH50%で6分間焼成し、実施例1~3のミートボールを得た。
また、ホスホリパーゼDを添加しない以外は同様の方法で、比較例1のミートボールを得た。
得られた実施例1~3、比較例1のミートボールについて、後述の(1)加熱後歩留りの評価、及び(2)ミートボール中遊離コリン及び遊離イノシトール測定を行った。加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。
表2に示す各ホスホリパーゼDのメーカー、酵素活性を表3に示す。 [Test Example 1]
(Production of meatballs of Examples 1 to 3 and Comparative Example 1)
The beef rib lean meat and beef Kenne fat shown in Table 1 were minced to 3 mm by a meat grinder (chopper). The onion was chopped into pieces of about 2 mm.
Each phospholipase D shown in Table 2 was added to the raw material of the composition shown in Table 1 so as to be 3.0 U per 1 g of raw material, and a standard mixer ("KitchenAid KSM5WH", manufactured by Whirlpool Corporation) was added. and mixed for 3 minutes at a setting speed of 1 to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, baked at 250 ° C. for 1 minute with an impinger (FGJOA9L, manufactured by Fujimac) and baked at 200 ° C. for 6 minutes at RH 50% in a steam convection oven (FSCC WE 61G, manufactured by Fujimac). Got meatballs.
Moreover, meatballs of Comparative Example 1 were obtained in the same manner except that phospholipase D was not added.
The meatballs obtained in Examples 1 to 3 and Comparative Example 1 were subjected to (1) evaluation of post-heating yield and (2) measurement of free choline and free inositol in meatballs, which will be described later. In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production.
Table 3 shows the manufacturer and enzymatic activity of each phospholipase D shown in Table 2.
一般的に、挽き肉加工食品においては、加熱後歩留りが高いと、肉汁が内部に閉じ込められており、好ましい品質であると考えられる。
実施例1~3、比較例1のミートボールの製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表4及び図1に示す。 (1) Evaluation of post-heating yield In general, when the minced meat processed food has a high post-heating yield, meat juice is trapped inside, and it is considered to be of desirable quality.
The post-heating yield was calculated according to the following formula from the weight before baking and the weight after baking when the meatballs of Examples 1 to 3 and Comparative Example 1 were produced.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Table 4 and FIG.
(2-1)サンプルの調製
実施例1~3、比較例1のミートボールについて、アルミパウチに入れて外側から手でつぶし、凍結乾燥機を用いて凍結乾燥した。凍結乾燥による減量割合を表5-1に示す。乾燥後、麺棒でたたいて粉砕し、ミートボール粉末を得た。得られたミートボール粉末を、下記の(2-2)ミートボール中遊離コリン測定、(2-3)ミートボール中遊離イノシトール測定のサンプルとして用いた。 (2) Measurement of free choline and free inositol in meatballs (2-1) Preparation of samples The meatballs of Examples 1 to 3 and Comparative Example 1 were placed in an aluminum pouch, crushed by hand from the outside, and then using a freeze dryer. and lyophilized. Table 5-1 shows the weight loss rate due to freeze-drying. After drying, it was pulverized by hitting with a rolling pin to obtain meatball powder. The obtained meatball powder was used as a sample for the following (2-2) measurement of free choline in meatballs and (2-3) measurement of free inositol in meatballs.
(測定前処理(抽出)方法)
(i)0.5gのミートボール粉末に3.0mlの水を加えた。
(ii)氷冷しながら10000rpmで2分間、ポリトロンで粉砕した。
(iii)1.5mlチューブ2本に全量を均等に分注し、10000xg,10度,1分で遠心分離した。
(iv)上清を2.5mlシリンジに移し、0.45μmクロマトディスクを通してサンプル液を得た。
(遊離コリン測定方法)
コリンオキシダーゼ、ペルオキシダーゼ等を含む発色溶液(コリンエステラーゼキット-NC、289-75181、富士フイルム和光純薬株式会社)1mLにサンプル液50μLを加え5分反応させ、赤色キノン色素を生成した。その後、反応停止液(コリンエステラーゼキット-NC、289-75181、富士フイルム和光純薬株式会社)にて反応停止後、10分間室温にて放置した。その後、波長505nmにおける吸光度を測定した。基準液(コリンエステラーゼキット-NC、289-75181、富士フイルム和光純薬株式会社)を用いて、0、0.75、1、1.25、1.5及び1.75mMコリン標準液を作成し、上記と同様の操作にて吸光度を測定した。吸光度より検量線を作成し、ミートボール中遊離コリン含有量を求めた。結果を表5-2に示す。 (2-2) Measurement of free choline in meatballs (measurement pretreatment (extraction) method)
(i) 3.0 ml of water was added to 0.5 g of meatball powder.
(ii) pulverized with a polytron at 10,000 rpm for 2 minutes while cooling on ice;
(iii) The total amount was evenly dispensed into two 1.5 ml tubes and centrifuged at 10000 xg, 10°C for 1 minute.
(iv) The supernatant was transferred to a 2.5 ml syringe and passed through a 0.45 μm chromatodisk to obtain a sample solution.
(Method for measuring free choline)
50 μL of the sample solution was added to 1 mL of a coloring solution containing choline oxidase, peroxidase, etc. (Cholinesterase Kit-NC, 289-75181, Fuji Film Wako Pure Chemical Industries, Ltd.) and allowed to react for 5 minutes to generate a red quinone dye. After that, the reaction was terminated with a reaction termination solution (cholinesterase kit-NC, 289-75181, Fuji Film Wako Pure Chemical Industries, Ltd.) and allowed to stand at room temperature for 10 minutes. After that, absorbance at a wavelength of 505 nm was measured. 0, 0.75, 1, 1.25, 1.5 and 1.75 mM choline standard solutions were prepared using a standard solution (cholinesterase kit-NC, 289-75181, Fujifilm Wako Pure Chemical Industries, Ltd.), Absorbance was measured in the same manner as above. A calibration curve was prepared from the absorbance, and the content of free choline in meatballs was determined. The results are shown in Table 5-2.
(測定前処理(抽出)方法)
(i)1~2gのミートボール粉末に60mlの水を加えた。
(ii)30分振とう後、1mol/L水酸化ナトリウム溶液及び1mol/L塩酸を用いてpH5.0に調整した。
(iii)水で100mLに定容し、ろ過し、サンプル液を得た。
(遊離イノシトール測定方法)
Saccharomyces cerevisiae ATCC 9080をブドウ糖ペプトン培地「ニッスイ」(日水製薬株式会社)を用い30℃±1℃、20時間±3時間で前培養し、接種菌液を調製した。滅菌したイノシトール分析培地に標準溶液又はサンプル液を塗布し、30℃±1℃、19時間±3時間で培養し、波長600nmで測定した。標準溶液(イノシトール0.5mg/ml、25%エタノール溶液)を用いて検量線を作成し、ミートボール中遊離イノシトール含有量を求めた。結果を表5-3に示す。 (2-3) Measurement of free inositol in meatballs (measurement pretreatment (extraction) method)
(i) 60 ml of water was added to 1-2 g of meatball powder.
(ii) After shaking for 30 minutes, the pH was adjusted to 5.0 using 1 mol/L sodium hydroxide solution and 1 mol/L hydrochloric acid.
(iii) The volume was adjusted to 100 mL with water and filtered to obtain a sample liquid.
(Method for measuring free inositol)
Saccharomyces cerevisiae ATCC 9080 was pre-cultured at 30° C.±1° C. for 20 hours±3 hours using a glucose peptone medium "Nissui" (Nissui Pharmaceutical Co., Ltd.) to prepare an inoculum solution. A standard solution or a sample solution was applied to a sterilized inositol analysis medium, cultured at 30° C.±1° C. for 19 hours±3 hours, and measured at a wavelength of 600 nm. A calibration curve was prepared using a standard solution (0.5 mg/ml inositol, 25% ethanol solution) to determine the content of free inositol in meatballs. The results are shown in Table 5-3.
(実施例2-1~実施例2-5、比較例2-2~比較例2-4のミートボールの製造)
表6に示した牛バラ赤身、牛ケンネ脂を肉挽き機(チョッパー)により3mmのミンチとした。玉ねぎは約2mmのみじん切りとした。
表6に示した配合の原材料および表7-1、7-2に記載の添加量のコリンを使用し、スタンダードミキサー(「キッチンエイド(KitchenAid)KSM5WH」、ワールプール・コーポレーション製)を用い、設定速度1にて3分間混合し、ミートボール生地を調製した。空気抜きした後に30gの球形に手で成型した。成型後、1時間冷蔵庫で静置した。インピンジャー(FGJOA9L、フジマック製)にて250℃で1分間焼成及びスチームコンベクションオーブン(FSCC WE 61G、フジマック製)にて200℃、RH50%で6分間焼成し、実施例2-1~実施例2-5、比較例2-2~比較例2-4のミートボールを得た。
(比較例2-1のミートボールの製造)
コリンを添加しない以外は実施例2-1等と同じ方法で、比較例2-1のミートボールを得た。
(ミートボール中コリン含有量)
表7-1に示す、比較例2-1のミートボール中コリン含有量は、同様の方法で製造した試験例1の比較例1のミートボール中のコリン含有量(60重量ppm、表5-2参照)に基づく推定値である。比較例2-1はコリンを添加していないので、ミートボール中コリン含有量(推定値:60重量ppm)は、原材料中に元々含まれるコリン量である。
表7-1、7-2に示す、実施例2-1~実施例2-5、比較例2-2~比較例2-4のミートボール中コリン含有量は、原材料中に元々含まれるコリン量(推定値:60重量ppm)とコリン添加量との合計である。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表7-1、7-2及び図2に示す。 [Test Example 2] Choline addition test (production of meatballs of Examples 2-1 to 2-5 and Comparative Examples 2-2 to 2-4)
The beef rib lean meat and beef Kenne fat shown in Table 6 were minced to 3 mm by a meat grinder (chopper). The onion was chopped into pieces of about 2 mm.
A standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) was used using raw materials with the formulations shown in Table 6 and choline in the amounts shown in Tables 7-1 and 7-2. Mix for 3 minutes at speed 1 to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, it was allowed to stand in a refrigerator for 1 hour. Baking at 250 ° C. for 1 minute with an impinger (FGJOA 9L, manufactured by Fujimac) and baking at 200 ° C. and RH 50% for 6 minutes in a steam convection oven (FSCC WE 61G, manufactured by Fujimac), Examples 2-1 to 2 -5, meatballs of Comparative Examples 2-2 to 2-4 were obtained.
(Production of meatballs of Comparative Example 2-1)
Meatballs of Comparative Example 2-1 were obtained in the same manner as in Example 2-1, etc., except that choline was not added.
(choline content in meatball)
The choline content in the meatball of Comparative Example 2-1 shown in Table 7-1 is the choline content in the meatball of Comparative Example 1 of Test Example 1 produced by the same method (60 ppm by weight, Table 5-1). 2). Since choline was not added in Comparative Example 2-1, the choline content in the meatball (estimated value: 60 ppm by weight) is the amount of choline originally contained in the raw material.
The choline content in the meatballs of Examples 2-1 to 2-5 and Comparative Examples 2-2 to 2-4 shown in Tables 7-1 and 7-2 is the choline originally contained in the raw materials. It is the sum of the amount (estimated value: 60 ppm by weight) and the added amount of choline.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. From the weight before firing and the weight after firing at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Tables 7-1, 7-2 and FIG.
(実施例3-1~実施例3-3、比較例3-2~比較例3-7のミートボールの製造)
試験例2の表6に示した牛バラ赤身、牛ケンネ脂を肉挽き機(チョッパー)により3mmのミンチとした。玉ねぎは約2mmのみじん切りとした。
試験例2の表6に示した配合の原材料および表8-1、8-2に記載の添加量のイノシトールを使用し、スタンダードミキサー(「キッチンエイド(KitchenAid)KSM5WH」、ワールプール・コーポレーション製)を用い設定速度1にて3分間混合し、ミートボール生地を調製した。空気抜きした後に30gの球形に手で成型した。成型後、1時間冷蔵庫で静置した。インピンジャー(FGJOA9L、フジマック製)にて250℃で1分間焼成及びスチームコンベクションオーブン(FSCC WE 61G、フジマック製)にて200℃、RH50%で6分間焼成し、実施例3-1~実施例3-3、比較例3-2~比較例3-7のミートボールを得た。
(比較例3-1のミートボールの製造)
イノシトールを添加しない以外は実施例3-1等と同じ方法で、比較例3-1のミートボールを得た。
(ミートボール中イノシトール含有量)
表8-1に示す、比較例3-1のミートボール中イノシトール含有量は、同様の方法で製造した試験例1の比較例1のミートボール中のイノシトール含有量(213重量ppm、表5-3参照)に基づく推定値である。比較例3-1はイノシトールを添加していないので、ミートボール中イノシトール含有量(推定値:213重量ppm)は、原材料中に元々含まれるイノシトール量である。
表8-1、8-2に示す、実施例3-1~実施例3-3、比較例3-2~比較例3-7のミートボール中イノシトール含有量は、原材料中に元々含まれるイノシトール量(推定値:213重量ppm)とイノシトール添加量との合計である。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表8-1、8-2及び図3に示す。 [Test Example 3] Inositol addition test (production of meatballs of Examples 3-1 to 3-3 and Comparative Examples 3-2 to 3-7)
The beef rib lean meat and beef kenne fat shown in Table 6 of Test Example 2 were minced to 3 mm by a meat grinder (chopper). The onion was chopped into pieces of about 2 mm.
A standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) was prepared using raw materials with the formulations shown in Table 6 of Test Example 2 and inositol in the amounts shown in Tables 8-1 and 8-2. was mixed for 3 minutes at a setting speed of 1 to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, it was allowed to stand in a refrigerator for 1 hour. Baking at 250 ° C. for 1 minute with an impinger (FGJOA 9L, manufactured by Fujimac) and baking at 200 ° C. and RH 50% for 6 minutes in a steam convection oven (FSCC WE 61G, manufactured by Fujimac), Examples 3-1 to 3 -3, meatballs of Comparative Examples 3-2 to 3-7 were obtained.
(Production of meatballs in Comparative Example 3-1)
Meatballs of Comparative Example 3-1 were obtained in the same manner as in Example 3-1, etc., except that inositol was not added.
(Inositol content in meatballs)
The inositol content in the meatball of Comparative Example 3-1 shown in Table 8-1 is the inositol content in the meatball of Comparative Example 1 of Test Example 1 produced by the same method (213 ppm by weight, Table 5- 3). Since inositol was not added in Comparative Example 3-1, the content of inositol in the meatball (estimated value: 213 ppm by weight) is the amount of inositol originally contained in the raw material.
The inositol content in the meatballs of Examples 3-1 to 3-3 and Comparative Examples 3-2 to 3-7 shown in Tables 8-1 and 8-2 is the inositol originally contained in the raw materials. It is the sum of the amount (estimated value: 213 ppm by weight) and the added amount of inositol.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. From the weight before firing and the weight after firing at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Tables 8-1, 8-2 and FIG.
(実施例4-1~実施例4-7の粗挽きソーセージの製造)
表9中の群Aの原材料を肉挽き機(チョッパー)により5mmのミンチとし、ニーダー(「卓上ニーダーPNV-5」、株式会社入江商会製)にて、前記ミンチに表9中の群Bの原材料および水を添加し、更に表10に記載のユニット量となるようにホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)およびリパーゼ(リパーゼAY「アマノ」、天野エンザイム株式会社製)を加えて10分間混合した後、冷蔵庫にて4時間静置し塩漬処理をした。
次いで、表9中の群Cの原材料を添加してスタンダードミキサー(「キッチンエイド(KitchenAid) KSM5WH」、ワールプール・コーポレーション製)にて2速で1分間混合した後、真空包装した。
次に、コラーゲンケーシング(デブロ(Devro)株式会社製)に充填し、60℃で30分間乾燥した後、燻煙(60℃、10分)および蒸煮(75℃、30分)を行い、一晩冷却して、実施例4-1~実施例4-7の粗挽きソーセージを製造した。
(比較例4-1の粗挽きソーセージの製造)
酵素を添加しない以外は実施例4-1~実施例4-7と同じ方法で、比較例4-1の粗挽きソーセージを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、粗挽きソーセージ製造時において、上記乾燥前の重量と、冷却後(すなわち、乾燥-燻煙-蒸煮-冷却工程の後)の重量を測定した。製造時における乾燥前の重量と冷却後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(冷却後の重量/乾燥前の重量)×100
結果を表10及び図4に示す。 [Test Example 4] Enzyme addition test (combination of phospholipase D and lipase)
(Production of coarsely ground sausages of Examples 4-1 to 4-7)
The raw materials of Group A in Table 9 are minced to 5 mm by a meat grinder (chopper), and the minced meat of Group B in Table 9 is minced with a kneader ("Desktop Kneader PNV-5", manufactured by Irie Shokai Co., Ltd.). Raw materials and water were added, and phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) and lipase (Lipase AY "Amano", manufactured by Amano Enzyme Co., Ltd.) were added so that the unit amounts shown in Table 10 were obtained. After the mixture was added and mixed for 10 minutes, it was allowed to stand in a refrigerator for 4 hours for pickling.
Next, the raw materials of group C in Table 9 were added and mixed in a standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) at 2nd speed for 1 minute, followed by vacuum packaging.
Next, the collagen casing (manufactured by Devro Co., Ltd.) was filled and dried at 60°C for 30 minutes, followed by smoking (60°C, 10 minutes) and steaming (75°C, 30 minutes) overnight. After cooling, coarsely ground sausages of Examples 4-1 to 4-7 were produced.
(Production of coarsely ground sausage of Comparative Example 4-1)
A coarsely ground sausage of Comparative Example 4-1 was obtained in the same manner as in Examples 4-1 to 4-7 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the post-heating yield, the weight before drying and the weight after cooling (that is, after the drying-smoking-steaming-cooling process) were measured during the production of the coarsely ground sausage. From the weight before drying and the weight after cooling at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after cooling/weight before drying) x 100
The results are shown in Table 10 and FIG.
(実施例5-1~実施例5-3のミートボールの製造)
表11に示した牛バラ赤身、牛ケンネ脂を肉挽き機(チョッパー)により3mmのミンチとした。玉ねぎは約2mmのみじん切りとした。
表11に示した配合の原材料および表12に記載のユニット量となるようにホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)およびトランスグルタミナーゼ(「アクティバ」TG、味の素株式会社製)を使用し、スタンダードミキサー(「キッチンエイド(KitchenAid)KSM5WH」、ワールプール・コーポレーション製)を用い設定速度1にて3分間混合し、ミートボール生地を調製した。空気抜きした後に30gの球形に手で成型した。成型後、1時間冷蔵庫で静置した。インピンジャー(FGJOA9L、フジマック製)にて250℃で1分間焼成及びスチームコンベクションオーブン(FSCC WE 61G、フジマック製)にて200℃、RH50%で6分間焼成し、実施例5-1~実施例5-3のミートボールを得た。
(比較例5-1、5-2のミートボールの製造)
酵素を添加しない以外は実施例5-1等と同じ方法で、比較例5-1のミートボールを得た。ホスホリパーゼDを添加しない以外は実施例5-1等と同じ方法で、比較例5-2のミートボールを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表12及び図5に示す。 [Test Example 5] Enzyme addition test (combination of phospholipase D and transglutaminase)
(Production of meatballs of Examples 5-1 to 5-3)
The beef rib lean meat and beef kenne fat shown in Table 11 were minced to 3 mm by a meat grinder (chopper). The onion was chopped into pieces of about 2 mm.
Phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) and transglutaminase ("Activa" TG, manufactured by Ajinomoto Co., Inc.) are added to the raw materials of the formulation shown in Table 11 and the unit amounts shown in Table 12. A standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) was used and mixed at a setting speed of 1 for 3 minutes to prepare a meatball dough. After degassing, it was manually molded into 30 g spheres. After molding, it was allowed to stand in a refrigerator for 1 hour. Baking at 250 ° C. for 1 minute with an impinger (FGJOA 9L, manufactured by Fujimac) and baking at 200 ° C. and RH 50% for 6 minutes in a steam convection oven (FSCC WE 61G, manufactured by Fujimac), Examples 5-1 to 5 -3 meatballs were obtained.
(Production of meatballs of Comparative Examples 5-1 and 5-2)
Meatballs of Comparative Example 5-1 were obtained in the same manner as in Example 5-1, etc., except that no enzyme was added. A meatball of Comparative Example 5-2 was obtained in the same manner as in Example 5-1, etc., except that phospholipase D was not added.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. From the weight before firing and the weight after firing at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Table 12 and FIG.
(実施例6-1~実施例6-4のミートボールの製造)
酵素を表13に示す酵素及び配合量に変更した以外は、実施例5-1等と同じ方法で、実施例6-1~実施例6-4のミートボールを得た。酵素はホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)およびα―グルコシダーゼ(トランスグルコシダーゼL「アマノ」」、天野エンザイム株式会社製)を使用した。
(比較例6-1、6-2のミートボールの製造)
酵素を添加しない以外は実施例6-1等と同じ方法で、比較例6-1のミートボールを得た。ホスホリパーゼDを添加しない以外は実施例6-1等と同じ方法で、比較例6-2のミートボールを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。試験例5と同じ方法で、加熱後歩留りを算出した。結果を表13及び図6に示す。 [Test Example 6] Enzyme addition test (combination of phospholipase D and α-glucosidase)
(Production of meatballs of Examples 6-1 to 6-4)
Meatballs of Examples 6-1 to 6-4 were obtained in the same manner as in Example 5-1, etc., except that the enzyme was changed to the enzyme and the blending amount shown in Table 13. Phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) and α-glucosidase (Transglucosidase L "Amano", manufactured by Amano Enzyme Co., Ltd.) were used as enzymes.
(Production of meatballs of Comparative Examples 6-1 and 6-2)
A meatball of Comparative Example 6-1 was obtained in the same manner as in Example 6-1, etc., except that no enzyme was added. A meatball of Comparative Example 6-2 was obtained in the same manner as in Example 6-1, etc., except that phospholipase D was not added.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. The post-heating yield was calculated in the same manner as in Test Example 5. The results are shown in Table 13 and FIG.
(実施例7-1のミートボールの製造)
酵素を表14に示す酵素及び配合量に変更した以外は、実施例5-1等と同じ方法で、実施例7-1のミートボールを得た。酵素はホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)、α―グルコシダーゼ(トランスグルコシダーゼL「アマノ」」、天野エンザイム株式会社製)およびリパーゼ(リパーゼAY「アマノ」、天野エンザイム株式会社製)を使用した。
(比較例7-1のミートボールの製造)
酵素を添加しない以外は実施例7-1と同じ方法で、比較例7-1のミートボールを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ミートボール製造時において、上記焼成前の重量と焼成後の重量を測定した。試験例5と同じ方法で、加熱後歩留りを算出した。結果を表14及び図7に示す。 [Test Example 7] Enzyme addition test (combined use of phospholipase D, α-glucosidase and lipase (production of meatballs of Example 7-1)
A meatball of Example 7-1 was obtained in the same manner as in Example 5-1, etc., except that the enzyme was changed to the enzyme and the amount shown in Table 14. Enzymes include phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Co., Ltd.), α-glucosidase (Transglucosidase L "Amano", manufactured by Amano Enzyme Co., Ltd.) and lipase (Lipase AY "Amano", manufactured by Amano Enzyme Co., Ltd.). )It was used.
(Production of meatballs in Comparative Example 7-1)
A meatball of Comparative Example 7-1 was obtained in the same manner as in Example 7-1 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of meatball production. The post-heating yield was calculated in the same manner as in Test Example 5. The results are shown in Table 14 and FIG.
(実施例8-1、8-2のハンバーグの製造)
表15に示した肉原料(牛バラ、豚ウデ)を肉挽き機(チョッパー)により3mmのミンチとした。
表15に示した配合の各原料に、ホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)を対原材料1gあたり0.4Uもしくは4.0Uとなるよう添加し、スタンダードミキサー(「キッチンエイド(KitchenAid)KSM5WH」、ワールプール・コーポレーション製)を用い、設定速度1にて3分間混合し、ハンバーグ生地を調製した。
生地を150gに計量し、空気抜きした後に小判形(長径130mm、短径90mm、厚さ12mm)に成型した。成型後にハンバーグ生地を230℃のグリドル(FGFT60601TC、フジマック社製)にて両面1分間ずつ焼成し、表面に焼き目を付けた後、インピンジャー(FGJOA9L、フジマック製)にて250℃で4分間焼成し、実施例8-1、8-2のハンバーグを得た。
(比較例8-1のハンバーグの製造)
酵素を添加しない以外は実施例8-1、8-2と同じ方法で、比較例8-1のハンバーグを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、ハンバーグ製造時において、上記焼成前の重量と焼成後の重量を測定した。製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表16及び図8に示す。 [Test Example 8] Phospholipase D addition test (hamburger)
(Production of hamburgers of Examples 8-1 and 8-2)
The meat raw materials (beef ribs, pork ribs) shown in Table 15 were minced to 3 mm by a meat grinder (chopper).
Phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) was added to each raw material of the formulation shown in Table 15 so as to be 0.4 U or 4.0 U per 1 g of the raw material, and a standard mixer ("KitchenAid (KitchenAid) KSM5WH", manufactured by Whirlpool Corporation) was mixed at a setting speed of 1 for 3 minutes to prepare a hamburger dough.
The dough was weighed to 150 g, and after the air was removed, it was formed into an oval shape (major axis: 130 mm, minor axis: 90 mm, thickness: 12 mm). After molding, the hamburger dough is baked on a griddle (FGFT60601TC, Fujimac Co., Ltd.) at 230 ° C. for 1 minute each on both sides, and after the surface is baked, baked at 250 ° C. for 4 minutes with an impinger (FGJOA9L, Fujimac Co., Ltd.). Then, hamburgers of Examples 8-1 and 8-2 were obtained.
(Production of hamburger in Comparative Example 8-1)
A hamburger of Comparative Example 8-1 was obtained in the same manner as in Examples 8-1 and 8-2 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the yield after heating, the weight before baking and the weight after baking were measured at the time of hamburger production. From the weight before firing and the weight after firing at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Table 16 and FIG.
(実施例9-1の粗挽きソーセージの製造)
表17中の群Aの原材料を肉挽き機(チョッパー)により5mmのミンチとし、ニーダー(「卓上ニーダーPNV-5」、株式会社入江商会製)にて、前記ミンチに表17中の群Bの原材料および水を添加し、更に表18に記載の配合量でホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)を加えて10分間混合した後、冷蔵庫にて4時間静置し塩漬処理をした。
次いで、表17中の群Cの原材料を添加してスタンダードミキサー(「キッチンエイド(KitchenAid) KSM5WH」、ワールプール・コーポレーション製)にて2速で1分間混合した後、真空包装した。
次に、コラーゲンケーシング(デブロ(Devro)株式会社製)に充填し、60℃で30分間乾燥した後、燻煙(60℃、10分)および蒸煮(75℃、30分)を行い、一晩冷却して、実施例9-1の粗挽きソーセージを製造した。
(比較例9-1の粗挽きソーセージの製造)
酵素を添加しない以外は実施例9-1と同じ方法で、比較例9-1の粗挽きソーセージを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、粗挽きソーセージ製造時において、上記乾燥前の重量と、冷却後(すなわち、乾燥-燻煙-蒸煮-冷却工程の後)の重量を測定した。製造時における乾燥前の重量と冷却後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(冷却後の重量/乾燥前の重量)×100
結果を表18及び図9に示す。 [Test Example 9] Phospholipase D addition test (sausage)
(Production of coarsely ground sausage of Example 9-1)
The raw materials of Group A in Table 17 were minced to 5 mm by a meat grinder (chopper), and the minced meat of Group B in Table 17 was minced with a kneader ("Desktop Kneader PNV-5", manufactured by Irie Shokai Co., Ltd.). Raw materials and water are added, and phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) is added in the amount shown in Table 18 and mixed for 10 minutes, then left for 4 hours in a refrigerator and salted. processed.
Next, the raw materials of group C in Table 17 were added and mixed in a standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) at 2nd speed for 1 minute, followed by vacuum packaging.
Next, the collagen casing (manufactured by Devro Co., Ltd.) was filled and dried at 60°C for 30 minutes, followed by smoking (60°C, 10 minutes) and steaming (75°C, 30 minutes) overnight. After cooling, coarsely ground sausages of Example 9-1 were produced.
(Production of coarsely ground sausage of Comparative Example 9-1)
A coarsely ground sausage of Comparative Example 9-1 was obtained in the same manner as in Example 9-1 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the post-heating yield, the weight before drying and the weight after cooling (that is, after the drying-smoking-steaming-cooling process) were measured during the production of the coarsely ground sausage. From the weight before drying and the weight after cooling at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after cooling/weight before drying) x 100
The results are shown in Table 18 and FIG.
(実施例10-1の鶏つみれの製造)
表19中の肉原料(鶏ムネ肉、豚脂肪)を肉挽き機(チョッパー)により5mmのミンチとし、スタンダードミキサー(「キッチンエイド(KitchenAid) KSM5WH」、ワールプール・コーポレーション製)にて、前記ミンチに表19中の各原料を添加し、更に表20に記載の配合量でホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)を加えて2速3分30秒間混合した。
生地を15gに計量し、冷蔵庫にて2時間静置した。成型後にスチームコンベクションオーブン(FSCC WE 61G、フジマック製)にて100℃、RH100%で10分間焼成し、実施例10-1の鶏つみれを得た。
(比較例10-1の鶏つみれの製造)
酵素を添加しない以外は実施例10-1と同じ方法で、比較例10-1の鶏つみれを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、鶏つみれ製造時において、上記焼成前の重量と焼成後の重量を測定した。製造時における焼成前の重量と焼成後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(焼成後の重量/焼成前の重量)×100
結果を表20及び図10に示す。 [Test Example 10] Phospholipase D addition test (chicken fishball)
(Production of chicken fishballs in Example 10-1)
The meat raw materials (chicken breast meat, pork fat) in Table 19 were minced to 5 mm by a meat grinder (chopper), and the minced meat was obtained with a standard mixer ("KitchenAid KSM5WH", manufactured by Whirlpool Corporation). In addition, each raw material in Table 19 was added to the mixture, and phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) was added in the amount shown in Table 20 and mixed at 2nd speed for 3 minutes and 30 seconds.
The dough was weighed to 15 g and allowed to stand in the refrigerator for 2 hours. After molding, it was baked in a steam convection oven (FSCC WE 61G, manufactured by Fujimac) at 100° C. and RH 100% for 10 minutes to obtain chicken meatballs of Example 10-1.
(Production of chicken fishballs in Comparative Example 10-1)
Chicken meatballs of Comparative Example 10-1 were obtained in the same manner as in Example 10-1 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the post-heating yield, the weight before baking and the weight after baking were measured during the production of chicken fishballs. From the weight before firing and the weight after firing at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after firing/weight before firing) x 100
The results are shown in Table 20 and FIG.
(実施例11-1の粗挽きソーセージの製造)
表21中の群Aの原材料を肉挽き機(チョッパー)により5mmのミンチとし、ニーダー(「卓上ニーダーPNV-5」、株式会社入江商会製)にて、前記ミンチに表21中の群Bの原材料および水を添加し、更に表22に記載の配合量でホスホリパーゼD(デナチーム PMD-P1、ナガセケムテックス株式会社製)を加えて10分間混合した後、冷蔵庫にて4時間静置し塩漬処理をした。
次いで、表21中の群Cの原材料を添加してスタンダードミキサー(「キッチンエイド(KitchenAid) KSM5WH」、ワールプール・コーポレーション製)にて2速で1分間混合した後、真空包装した。
次に、コラーゲンケーシング(デブロ(Devro)株式会社製)に充填し、60℃で30分間乾燥した後、燻煙(60℃、10分)および蒸煮(75℃、30分)を行い、一晩冷却して、実施例11―1の粗挽きソーセージを製造した。
(比較例11-1の粗挽きソーセージの製造)
酵素を添加しない以外は実施例11-1と同じ方法で、比較例11-1の粗挽きソーセージを得た。
(加熱後歩留りの評価)
加熱後歩留りの評価のために、粗挽きソーセージ製造時において、上記乾燥前の重量と、冷却後(すなわち、乾燥-燻煙-蒸煮-冷却工程の後)の重量を測定した。製造時における乾燥前の重量と冷却後の重量から、下記の式に従って、加熱後歩留りを算出した。
(式)
加熱後歩留り(重量%)=(冷却後の重量/乾燥前の重量)×100
結果を表22及び図11に示す。 [Test Example 11] Phospholipase D addition test (high-fat sausage)
(Production of coarsely ground sausage of Example 11-1)
The raw materials of Group A in Table 21 were minced to 5 mm by a meat grinder (chopper), and the minced meat of Group B in Table 21 was minced with a kneader ("Desktop Kneader PNV-5", manufactured by Irie Shokai Co., Ltd.). Raw materials and water are added, and phospholipase D (Denazyme PMD-P1, manufactured by Nagase ChemteX Corporation) is added in the amount shown in Table 22 and mixed for 10 minutes, then left for 4 hours in a refrigerator and salted. processed.
Next, the raw materials of Group C in Table 21 were added and mixed in a standard mixer (“KitchenAid KSM5WH”, manufactured by Whirlpool Corporation) at 2nd speed for 1 minute, and then vacuum-packaged.
Next, the collagen casing (manufactured by Devro Co., Ltd.) was filled and dried at 60°C for 30 minutes, followed by smoking (60°C, 10 minutes) and steaming (75°C, 30 minutes) overnight. After cooling, coarsely ground sausages of Example 11-1 were produced.
(Production of coarsely ground sausage of Comparative Example 11-1)
A coarsely ground sausage of Comparative Example 11-1 was obtained in the same manner as in Example 11-1 except that no enzyme was added.
(Evaluation of yield after heating)
In order to evaluate the post-heating yield, the weight before drying and the weight after cooling (that is, after the drying-smoking-steaming-cooling process) were measured during the production of the coarsely ground sausage. From the weight before drying and the weight after cooling at the time of production, the post-heating yield was calculated according to the following formula.
(formula)
Yield after heating (% by weight) = (weight after cooling/weight before drying) x 100
The results are shown in Table 22 and FIG.
Claims (6)
- 下記(1)、(2)のいずれか一方又は両方を含有する、畜肉練り製品。
(1)65~9000重量ppmのコリン
(2)230~380重量ppmのイノシトール A livestock meat paste product containing either one or both of the following (1) and (2).
(1) 65-9000 ppm by weight choline (2) 230-380 ppm by weight inositol - 前記(1)及び(2)を含有する、請求項1記載の畜肉練り製品。 The meat paste product according to claim 1, which contains the above (1) and (2).
- 前記コリン及びイノシトールが、畜肉練り製品の原材料に含まれるリン脂質に由来する、請求項1又は2に記載の畜肉練り製品。 The meat paste product according to claim 1 or 2, wherein the choline and inositol are derived from phospholipids contained in raw materials of the meat paste product.
- 原材料である畜肉にホスホリパーゼDを作用させることを含む、畜肉練り製品の加熱後歩留り向上方法。 A method for improving the post-heating yield of livestock meat paste products, which includes allowing phospholipase D to act on livestock meat, which is a raw material.
- ホスホリパーゼDが、シグマ アルドリッチ製ホスホリパーゼD 放線菌由来(商品名)、ナガセケムテック製デナチームPMD-P1(商品名)及び旭化成ファーマ製PHOSPHOLIPASE D[PLDP(T-39)](Glycerophospholipid specific)(商品名)からなる群から選択される、請求項4記載の方法。 Phospholipase D is Sigma-Aldrich's Phospholipase D Actinomycete-derived (trade name), Nagase Chemtech's Denazym PMD-P1 (trade name), and Asahi Kasei Pharma's PHOSPHOLIPASE D [PLDP (T-39)] (Glycerophospholipid specific) (trade name) 5. The method of claim 4, wherein the method is selected from the group consisting of:
- トランスグルタミナーゼ、リパーゼ、及び澱粉分解酵素からなる群から選択される少なくとも1つの酵素をさらに作用させることを含む、請求項4または5に記載の方法。 The method according to claim 4 or 5, further comprising allowing at least one enzyme selected from the group consisting of transglutaminase, lipase, and amylolytic enzyme to act.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112023022575A BR112023022575A2 (en) | 2021-04-30 | 2022-04-28 | MASHED MEAT PRODUCT, AND, METHOD FOR IMPROVING YIELD AFTER HEATING A MASHED MEAT PRODUCT |
| JP2023517634A JPWO2022231003A1 (en) | 2021-04-30 | 2022-04-28 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2021-077257 | 2021-04-30 | ||
| JP2021077257 | 2021-04-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022231003A1 true WO2022231003A1 (en) | 2022-11-03 |
Family
ID=83846897
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2022/019450 WO2022231003A1 (en) | 2021-04-30 | 2022-04-28 | Kneaded meat product with improved quality |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JPWO2022231003A1 (en) |
| BR (1) | BR112023022575A2 (en) |
| WO (1) | WO2022231003A1 (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11346719A (en) * | 1998-06-03 | 1999-12-21 | Izu Rice Center:Kk | Preparation of lye using phyllostachys pubescens and modification of meat using the same |
| WO2006075771A1 (en) * | 2005-01-13 | 2006-07-20 | Ajinomoto Co., Inc. | Processed meat product or fish paste product and process for producing the same |
| WO2010140708A1 (en) * | 2009-06-05 | 2010-12-09 | 味の素株式会社 | Enzyme preparation for reforming processed meat products and manufacturing method for processed meat products |
| WO2013172447A1 (en) * | 2012-05-17 | 2013-11-21 | ナガセケムテックス株式会社 | Enzyme preparation for modifying food material |
| CN107593874A (en) * | 2017-08-24 | 2018-01-19 | 安徽靖童科技农业发展有限公司 | A kind of meat product processing stabilizers |
| JP2020145977A (en) * | 2019-03-14 | 2020-09-17 | 奥野製薬工業株式会社 | Texture-improving agent and texture-improving method of meat product |
-
2022
- 2022-04-28 BR BR112023022575A patent/BR112023022575A2/en unknown
- 2022-04-28 WO PCT/JP2022/019450 patent/WO2022231003A1/en unknown
- 2022-04-28 JP JP2023517634A patent/JPWO2022231003A1/ja active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11346719A (en) * | 1998-06-03 | 1999-12-21 | Izu Rice Center:Kk | Preparation of lye using phyllostachys pubescens and modification of meat using the same |
| WO2006075771A1 (en) * | 2005-01-13 | 2006-07-20 | Ajinomoto Co., Inc. | Processed meat product or fish paste product and process for producing the same |
| WO2010140708A1 (en) * | 2009-06-05 | 2010-12-09 | 味の素株式会社 | Enzyme preparation for reforming processed meat products and manufacturing method for processed meat products |
| WO2013172447A1 (en) * | 2012-05-17 | 2013-11-21 | ナガセケムテックス株式会社 | Enzyme preparation for modifying food material |
| CN107593874A (en) * | 2017-08-24 | 2018-01-19 | 安徽靖童科技农业发展有限公司 | A kind of meat product processing stabilizers |
| JP2020145977A (en) * | 2019-03-14 | 2020-09-17 | 奥野製薬工業株式会社 | Texture-improving agent and texture-improving method of meat product |
Also Published As
| Publication number | Publication date |
|---|---|
| BR112023022575A2 (en) | 2024-01-16 |
| JPWO2022231003A1 (en) | 2022-11-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6984588B2 (en) | Manufacturing method of food containing plant protein | |
| JP6642617B2 (en) | Method for producing starch-containing food and enzyme preparation for modifying starch-containing food | |
| Renzetti et al. | Oxidative and proteolytic enzyme preparations as promising improvers for oat bread formulations: rheological, biochemical and microstructural background | |
| CN101573037B (en) | Method of preparing a cake using phospholipase | |
| RU2473246C2 (en) | Method for manufacture of treated meat product ot seafood product and enzyme preparation for improvement of treated meat product or seafood product | |
| EA018153B1 (en) | Lipid acyltransferase and food or feed composition comprising the same | |
| US20140087052A1 (en) | Oil- or fat-processed starch and method for producing same | |
| NZ247925A (en) | Fat substitute composition comprising a cereal hydrolysate and a hydrocolloid gum in an amount to form a thermo-irreversible gel; low fat (emulsified) meat products | |
| Yüceer et al. | Texture, rheology, storage stability, and sensory evaluation of meringue's prepared from lipase enzyme‐modified liquid egg white | |
| US5294457A (en) | Thermo-irreversible gel particles for use in foods | |
| US20120135111A1 (en) | Enzyme preparation for reforming processed meat products and manufacturing method for processed meat products | |
| JP6066016B2 (en) | Noodle manufacturing method and noodle loosening improver | |
| US20100323058A1 (en) | Method for producing fish paste product, and enzyme preparation for fish paste products | |
| WO2022231003A1 (en) | Kneaded meat product with improved quality | |
| JP4800911B2 (en) | Lysified egg yolk and food using the same | |
| JP2000513231A (en) | Use of deamidase in baking | |
| JPWO2020004506A1 (en) | Waxy seed starch and its manufacturing method | |
| JP2017093299A (en) | Bakery food mixture | |
| EP4241570A1 (en) | Production method for meat-like processed food product | |
| JP2019510466A (en) | Improved Enzymatic Modification of Wheat Phospholipids in Bread Application | |
| JP2023066140A (en) | Powder composition for food production | |
| WO2020145371A1 (en) | Method for manufacturing starch-containing food | |
| JP6203018B2 (en) | Bread | |
| JP2012065554A (en) | Food texture-improving agent | |
| JP2790838B2 (en) | Anti-aging agent for starch, starchy raw material and starch-containing food containing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22795916 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2023517634 Country of ref document: JP Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023022575 Country of ref document: BR |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 112023022575 Country of ref document: BR Kind code of ref document: A2 Effective date: 20231027 |