WO2022137913A1 - Maleimide resin, asymmetric bismaleimide compound, curable composition, cured object, semiconductor-encapsulating material, semiconductor-encapsulating device, prepreg, circuit board, and build-up film - Google Patents
Maleimide resin, asymmetric bismaleimide compound, curable composition, cured object, semiconductor-encapsulating material, semiconductor-encapsulating device, prepreg, circuit board, and build-up film Download PDFInfo
- Publication number
- WO2022137913A1 WO2022137913A1 PCT/JP2021/042356 JP2021042356W WO2022137913A1 WO 2022137913 A1 WO2022137913 A1 WO 2022137913A1 JP 2021042356 W JP2021042356 W JP 2021042356W WO 2022137913 A1 WO2022137913 A1 WO 2022137913A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- compound
- curable composition
- aromatic
- semiconductor
- Prior art date
Links
- 229920005989 resin Polymers 0.000 title claims abstract description 77
- 239000011347 resin Substances 0.000 title claims abstract description 77
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 title claims abstract description 67
- -1 bismaleimide compound Chemical class 0.000 title claims abstract description 48
- 239000000203 mixture Substances 0.000 title claims abstract description 36
- 239000000463 material Substances 0.000 title claims abstract description 28
- 229920003192 poly(bis maleimide) Polymers 0.000 title claims description 19
- 150000001875 compounds Chemical class 0.000 claims abstract description 78
- 125000003118 aryl group Chemical group 0.000 claims abstract description 69
- 239000011230 binding agent Substances 0.000 claims abstract description 20
- 229920000768 polyamine Polymers 0.000 claims abstract description 15
- 125000001931 aliphatic group Chemical group 0.000 claims description 43
- 125000003545 alkoxy group Chemical group 0.000 claims description 30
- 125000005843 halogen group Chemical group 0.000 claims description 30
- 239000000047 product Substances 0.000 claims description 30
- 125000004432 carbon atom Chemical group C* 0.000 claims description 29
- 239000004065 semiconductor Substances 0.000 claims description 26
- 125000001424 substituent group Chemical group 0.000 claims description 24
- 150000001491 aromatic compounds Chemical class 0.000 claims description 22
- 125000003302 alkenyloxy group Chemical group 0.000 claims description 16
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 16
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 10
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 9
- 125000005133 alkynyloxy group Chemical group 0.000 claims description 8
- 238000005538 encapsulation Methods 0.000 claims description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- 125000004430 oxygen atom Chemical group O* 0.000 claims description 6
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 5
- 229910052717 sulfur Inorganic materials 0.000 claims description 5
- 125000004434 sulfur atom Chemical group 0.000 claims description 5
- 239000007795 chemical reaction product Substances 0.000 claims description 4
- 125000000962 organic group Chemical group 0.000 claims description 4
- 239000003205 fragrance Substances 0.000 claims 1
- 238000006243 chemical reaction Methods 0.000 abstract description 23
- 238000002844 melting Methods 0.000 abstract description 21
- 230000008018 melting Effects 0.000 abstract description 21
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 54
- 238000005227 gel permeation chromatography Methods 0.000 description 25
- 239000000243 solution Substances 0.000 description 25
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 20
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 18
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 15
- 239000002904 solvent Substances 0.000 description 15
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 14
- 238000005259 measurement Methods 0.000 description 14
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 13
- 239000012153 distilled water Substances 0.000 description 13
- 125000004122 cyclic group Chemical group 0.000 description 12
- 238000000034 method Methods 0.000 description 12
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 11
- 150000008065 acid anhydrides Chemical class 0.000 description 10
- 150000001412 amines Chemical class 0.000 description 10
- 229920000767 polyaniline Polymers 0.000 description 10
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 10
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 238000000113 differential scanning calorimetry Methods 0.000 description 9
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 8
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 8
- 239000003822 epoxy resin Substances 0.000 description 8
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 8
- 229920000647 polyepoxide Polymers 0.000 description 8
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 8
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 8
- 125000000217 alkyl group Chemical group 0.000 description 7
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 6
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 230000002378 acidificating effect Effects 0.000 description 6
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 6
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 6
- LNEPOXFFQSENCJ-UHFFFAOYSA-N haloperidol Chemical compound C1CC(O)(C=2C=CC(Cl)=CC=2)CCN1CCCC(=O)C1=CC=C(F)C=C1 LNEPOXFFQSENCJ-UHFFFAOYSA-N 0.000 description 6
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 6
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 6
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 6
- 239000000523 sample Substances 0.000 description 6
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 5
- 125000005336 allyloxy group Chemical group 0.000 description 5
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 5
- 239000003054 catalyst Substances 0.000 description 5
- 229910052801 chlorine Inorganic materials 0.000 description 5
- 125000001309 chloro group Chemical group Cl* 0.000 description 5
- 238000001816 cooling Methods 0.000 description 5
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 5
- 239000003063 flame retardant Substances 0.000 description 5
- 229910052731 fluorine Inorganic materials 0.000 description 5
- 125000001153 fluoro group Chemical group F* 0.000 description 5
- 239000011521 glass Substances 0.000 description 5
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 239000011256 inorganic filler Substances 0.000 description 5
- 229910003475 inorganic filler Inorganic materials 0.000 description 5
- 125000001624 naphthyl group Chemical group 0.000 description 5
- 125000004998 naphthylethyl group Chemical group C1(=CC=CC2=CC=CC=C12)CC* 0.000 description 5
- 125000004923 naphthylmethyl group Chemical group C1(=CC=CC2=CC=CC=C12)C* 0.000 description 5
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 5
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 5
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 5
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 5
- MLPVBIWIRCKMJV-UHFFFAOYSA-N 2-ethylaniline Chemical compound CCC1=CC=CC=C1N MLPVBIWIRCKMJV-UHFFFAOYSA-N 0.000 description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 4
- 125000002490 anilino group Chemical group [H]N(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 4
- FPYJFEHAWHCUMM-UHFFFAOYSA-N maleic anhydride Chemical compound O=C1OC(=O)C=C1 FPYJFEHAWHCUMM-UHFFFAOYSA-N 0.000 description 4
- 238000002156 mixing Methods 0.000 description 4
- 239000003960 organic solvent Substances 0.000 description 4
- 238000003786 synthesis reaction Methods 0.000 description 4
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 4
- XQUPVDVFXZDTLT-UHFFFAOYSA-N 1-[4-[[4-(2,5-dioxopyrrol-1-yl)phenyl]methyl]phenyl]pyrrole-2,5-dione Chemical class O=C1C=CC(=O)N1C(C=C1)=CC=C1CC1=CC=C(N2C(C=CC2=O)=O)C=C1 XQUPVDVFXZDTLT-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- 235000002597 Solanum melongena Nutrition 0.000 description 3
- 229910021529 ammonia Inorganic materials 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 238000006297 dehydration reaction Methods 0.000 description 3
- 239000004744 fabric Substances 0.000 description 3
- 239000005350 fused silica glass Substances 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 238000002514 liquid chromatography mass spectrum Methods 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 239000005011 phenolic resin Substances 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- 239000002798 polar solvent Substances 0.000 description 3
- YLLIGHVCTUPGEH-UHFFFAOYSA-M potassium;ethanol;hydroxide Chemical compound [OH-].[K+].CCO YLLIGHVCTUPGEH-UHFFFAOYSA-M 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 235000017557 sodium bicarbonate Nutrition 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- RNFJDJUURJAICM-UHFFFAOYSA-N 2,2,4,4,6,6-hexaphenoxy-1,3,5-triaza-2$l^{5},4$l^{5},6$l^{5}-triphosphacyclohexa-1,3,5-triene Chemical compound N=1P(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP(OC=2C=CC=CC=2)(OC=2C=CC=CC=2)=NP=1(OC=1C=CC=CC=1)OC1=CC=CC=C1 RNFJDJUURJAICM-UHFFFAOYSA-N 0.000 description 2
- FOYHNROGBXVLLX-UHFFFAOYSA-N 2,6-diethylaniline Chemical compound CCC1=CC=CC(CC)=C1N FOYHNROGBXVLLX-UHFFFAOYSA-N 0.000 description 2
- UFFBMTHBGFGIHF-UHFFFAOYSA-N 2,6-dimethylaniline Chemical group CC1=CC=CC(C)=C1N UFFBMTHBGFGIHF-UHFFFAOYSA-N 0.000 description 2
- FPZWZCWUIYYYBU-UHFFFAOYSA-N 2-(2-ethoxyethoxy)ethyl acetate Chemical compound CCOCCOCCOC(C)=O FPZWZCWUIYYYBU-UHFFFAOYSA-N 0.000 description 2
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 2
- KWOLFJPFCHCOCG-UHFFFAOYSA-N Acetophenone Chemical compound CC(=O)C1=CC=CC=C1 KWOLFJPFCHCOCG-UHFFFAOYSA-N 0.000 description 2
- 229930185605 Bisphenol Natural products 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 2
- UFWIBTONFRDIAS-UHFFFAOYSA-N Naphthalene Chemical compound C1=CC=CC2=CC=CC=C21 UFWIBTONFRDIAS-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- 239000004793 Polystyrene Substances 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- 239000003377 acid catalyst Substances 0.000 description 2
- 238000007259 addition reaction Methods 0.000 description 2
- 239000002313 adhesive film Substances 0.000 description 2
- 229910000148 ammonium phosphate Inorganic materials 0.000 description 2
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Chemical compound C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000004760 aramid Substances 0.000 description 2
- 229920003235 aromatic polyamide Polymers 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 description 2
- 235000010290 biphenyl Nutrition 0.000 description 2
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 150000007973 cyanuric acids Chemical class 0.000 description 2
- MNNHAPBLZZVQHP-UHFFFAOYSA-N diammonium hydrogen phosphate Chemical compound [NH4+].[NH4+].OP([O-])([O-])=O MNNHAPBLZZVQHP-UHFFFAOYSA-N 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000010438 heat treatment Methods 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 150000002460 imidazoles Chemical class 0.000 description 2
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 2
- 125000005439 maleimidyl group Chemical group C1(C=CC(N1*)=O)=O 0.000 description 2
- 239000012046 mixed solvent Substances 0.000 description 2
- 125000004957 naphthylene group Chemical group 0.000 description 2
- 239000012454 non-polar solvent Substances 0.000 description 2
- 229920003986 novolac Polymers 0.000 description 2
- FDPIMTJIUBPUKL-UHFFFAOYSA-N pentan-3-one Chemical compound CCC(=O)CC FDPIMTJIUBPUKL-UHFFFAOYSA-N 0.000 description 2
- 150000002978 peroxides Chemical class 0.000 description 2
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 2
- 150000003018 phosphorus compounds Chemical class 0.000 description 2
- 229920001296 polysiloxane Polymers 0.000 description 2
- 229920002223 polystyrene Polymers 0.000 description 2
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 2
- 150000003222 pyridines Chemical class 0.000 description 2
- 230000003014 reinforcing effect Effects 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 150000003512 tertiary amines Chemical class 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 238000004448 titration Methods 0.000 description 2
- 238000005406 washing Methods 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- FYADHXFMURLYQI-UHFFFAOYSA-N 1,2,4-triazine Chemical class C1=CN=NC=N1 FYADHXFMURLYQI-UHFFFAOYSA-N 0.000 description 1
- XAZPKEBWNIUCKF-UHFFFAOYSA-N 1-[4-[4-[2-[4-[4-(2,5-dioxopyrrol-1-yl)phenoxy]phenyl]propan-2-yl]phenoxy]phenyl]pyrrole-2,5-dione Chemical class C=1C=C(OC=2C=CC(=CC=2)N2C(C=CC2=O)=O)C=CC=1C(C)(C)C(C=C1)=CC=C1OC(C=C1)=CC=C1N1C(=O)C=CC1=O XAZPKEBWNIUCKF-UHFFFAOYSA-N 0.000 description 1
- LHENQXAPVKABON-UHFFFAOYSA-N 1-methoxypropan-1-ol Chemical compound CCC(O)OC LHENQXAPVKABON-UHFFFAOYSA-N 0.000 description 1
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 1
- WJFKNYWRSNBZNX-UHFFFAOYSA-N 10H-phenothiazine Chemical compound C1=CC=C2NC3=CC=CC=C3SC2=C1 WJFKNYWRSNBZNX-UHFFFAOYSA-N 0.000 description 1
- MFGALGYVFGDXIX-UHFFFAOYSA-N 2,3-Dimethylmaleic anhydride Chemical compound CC1=C(C)C(=O)OC1=O MFGALGYVFGDXIX-UHFFFAOYSA-N 0.000 description 1
- BSYJHYLAMMJNRC-UHFFFAOYSA-N 2,4,4-trimethylpentan-2-ol Chemical compound CC(C)(C)CC(C)(C)O BSYJHYLAMMJNRC-UHFFFAOYSA-N 0.000 description 1
- OAYXUHPQHDHDDZ-UHFFFAOYSA-N 2-(2-butoxyethoxy)ethanol Chemical compound CCCCOCCOCCO OAYXUHPQHDHDDZ-UHFFFAOYSA-N 0.000 description 1
- XNWFRZJHXBZDAG-UHFFFAOYSA-N 2-METHOXYETHANOL Chemical compound COCCO XNWFRZJHXBZDAG-UHFFFAOYSA-N 0.000 description 1
- ZNQVEEAIQZEUHB-UHFFFAOYSA-N 2-ethoxyethanol Chemical compound CCOCCO ZNQVEEAIQZEUHB-UHFFFAOYSA-N 0.000 description 1
- SVONRAPFKPVNKG-UHFFFAOYSA-N 2-ethoxyethyl acetate Chemical compound CCOCCOC(C)=O SVONRAPFKPVNKG-UHFFFAOYSA-N 0.000 description 1
- CMLFRMDBDNHMRA-UHFFFAOYSA-N 2h-1,2-benzoxazine Chemical compound C1=CC=C2C=CNOC2=C1 CMLFRMDBDNHMRA-UHFFFAOYSA-N 0.000 description 1
- AYKYXWQEBUNJCN-UHFFFAOYSA-N 3-methylfuran-2,5-dione Chemical compound CC1=CC(=O)OC1=O AYKYXWQEBUNJCN-UHFFFAOYSA-N 0.000 description 1
- VPWNQTHUCYMVMZ-UHFFFAOYSA-N 4,4'-sulfonyldiphenol Chemical class C1=CC(O)=CC=C1S(=O)(=O)C1=CC=C(O)C=C1 VPWNQTHUCYMVMZ-UHFFFAOYSA-N 0.000 description 1
- VVQVMHASNBSOOC-UHFFFAOYSA-N 4-(hydroxymethyl)benzenesulfonic acid Chemical compound OCC1=CC=C(S(O)(=O)=O)C=C1 VVQVMHASNBSOOC-UHFFFAOYSA-N 0.000 description 1
- ULKLGIFJWFIQFF-UHFFFAOYSA-N 5K8XI641G3 Chemical compound CCC1=NC=C(C)N1 ULKLGIFJWFIQFF-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000004254 Ammonium phosphate Substances 0.000 description 1
- 239000004114 Ammonium polyphosphate Substances 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- 101100223811 Caenorhabditis elegans dsc-1 gene Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 102100021202 Desmocollin-1 Human genes 0.000 description 1
- 239000005696 Diammonium phosphate Substances 0.000 description 1
- 101000968043 Homo sapiens Desmocollin-1 Proteins 0.000 description 1
- 101000880960 Homo sapiens Desmocollin-3 Proteins 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- NTIZESTWPVYFNL-UHFFFAOYSA-N Methyl isobutyl ketone Chemical compound CC(C)CC(C)=O NTIZESTWPVYFNL-UHFFFAOYSA-N 0.000 description 1
- UIHCLUNTQKBZGK-UHFFFAOYSA-N Methyl isobutyl ketone Natural products CCC(C)C(C)=O UIHCLUNTQKBZGK-UHFFFAOYSA-N 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical compound OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- 239000006087 Silane Coupling Agent Substances 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- IKHGUXGNUITLKF-XPULMUKRSA-N acetaldehyde Chemical compound [14CH]([14CH3])=O IKHGUXGNUITLKF-XPULMUKRSA-N 0.000 description 1
- WEVYAHXRMPXWCK-UHFFFAOYSA-N acetonitrile Substances CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 1
- XFSBVAOIAHNAPC-WSORPINJSA-N acetylbenzoylaconine Chemical compound O([C@H]1[C@]2(O)C[C@H]3C45[C@@H]6[C@@H]([C@@]([C@H]31)(OC(C)=O)[C@@H](O)[C@@H]2OC)[C@H](OC)C4[C@]([C@@H](C[C@H]5OC)O)(COC)CN6CC)C(=O)C1=CC=CC=C1 XFSBVAOIAHNAPC-WSORPINJSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000000853 adhesive Substances 0.000 description 1
- 230000001070 adhesive effect Effects 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- LFVGISIMTYGQHF-UHFFFAOYSA-N ammonium dihydrogen phosphate Chemical compound [NH4+].OP(O)([O-])=O LFVGISIMTYGQHF-UHFFFAOYSA-N 0.000 description 1
- 229910000387 ammonium dihydrogen phosphate Inorganic materials 0.000 description 1
- ZRIUUUJAJJNDSS-UHFFFAOYSA-N ammonium phosphates Chemical compound [NH4+].[NH4+].[NH4+].[O-]P([O-])([O-])=O ZRIUUUJAJJNDSS-UHFFFAOYSA-N 0.000 description 1
- 235000019289 ammonium phosphates Nutrition 0.000 description 1
- 235000019826 ammonium polyphosphate Nutrition 0.000 description 1
- 229920001276 ammonium polyphosphate Polymers 0.000 description 1
- 239000000010 aprotic solvent Substances 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 150000004074 biphenyls Chemical class 0.000 description 1
- IISBACLAFKSPIT-UHFFFAOYSA-N bisphenol A Chemical compound C=1C=C(O)C=CC=1C(C)(C)C1=CC=C(O)C=C1 IISBACLAFKSPIT-UHFFFAOYSA-N 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 150000001639 boron compounds Chemical class 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 238000005266 casting Methods 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 239000004927 clay Substances 0.000 description 1
- 238000013329 compounding Methods 0.000 description 1
- 238000007906 compression Methods 0.000 description 1
- 230000006835 compression Effects 0.000 description 1
- 239000011231 conductive filler Substances 0.000 description 1
- 229910002026 crystalline silica Inorganic materials 0.000 description 1
- 239000004643 cyanate ester Substances 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 229910000388 diammonium phosphate Inorganic materials 0.000 description 1
- 235000019838 diammonium phosphate Nutrition 0.000 description 1
- DENRZWYUOJLTMF-UHFFFAOYSA-N diethyl sulfate Chemical compound CCOS(=O)(=O)OCC DENRZWYUOJLTMF-UHFFFAOYSA-N 0.000 description 1
- 238000001938 differential scanning calorimetry curve Methods 0.000 description 1
- RXKJFZQQPQGTFL-UHFFFAOYSA-N dihydroxyacetone Chemical class OCC(=O)CO RXKJFZQQPQGTFL-UHFFFAOYSA-N 0.000 description 1
- VAYGXNSJCAHWJZ-UHFFFAOYSA-N dimethyl sulfate Chemical compound COS(=O)(=O)OC VAYGXNSJCAHWJZ-UHFFFAOYSA-N 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000012776 electronic material Substances 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-M hexanoate Chemical compound CCCCCC([O-])=O FUZZWVXGSFPDMH-UHFFFAOYSA-M 0.000 description 1
- 238000005470 impregnation Methods 0.000 description 1
- 238000001746 injection moulding Methods 0.000 description 1
- 239000012796 inorganic flame retardant Substances 0.000 description 1
- 239000005453 ketone based solvent Substances 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- 229940098779 methanesulfonic acid Drugs 0.000 description 1
- 239000006082 mold release agent Substances 0.000 description 1
- 235000019837 monoammonium phosphate Nutrition 0.000 description 1
- 239000006012 monoammonium phosphate Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- 150000004002 naphthaldehydes Chemical class 0.000 description 1
- 239000004745 nonwoven fabric Substances 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000003973 paint Substances 0.000 description 1
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 229950000688 phenothiazine Drugs 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- ACVYVLVWPXVTIT-UHFFFAOYSA-N phosphinic acid Chemical class O[PH2]=O ACVYVLVWPXVTIT-UHFFFAOYSA-N 0.000 description 1
- VBQCHPIMZGQLAZ-UHFFFAOYSA-N phosphorane Chemical class [PH5] VBQCHPIMZGQLAZ-UHFFFAOYSA-N 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000003586 protic polar solvent Substances 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 239000010453 quartz Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 229920002545 silicone oil Polymers 0.000 description 1
- 229920002379 silicone rubber Polymers 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- FHYUCVWDMABHHH-UHFFFAOYSA-N toluene;1,2-xylene Chemical group CC1=CC=CC=C1.CC1=CC=CC=C1C FHYUCVWDMABHHH-UHFFFAOYSA-N 0.000 description 1
- 238000001721 transfer moulding Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D207/00—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D207/02—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D207/44—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having three double bonds between ring members or between ring members and non-ring members
- C07D207/444—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having three double bonds between ring members or between ring members and non-ring members having two doubly-bound oxygen atoms directly attached in positions 2 and 5
- C07D207/448—Heterocyclic compounds containing five-membered rings not condensed with other rings, with one nitrogen atom as the only ring hetero atom with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having three double bonds between ring members or between ring members and non-ring members having two doubly-bound oxygen atoms directly attached in positions 2 and 5 with only hydrogen atoms or radicals containing only hydrogen and carbon atoms directly attached to other ring carbon atoms, e.g. maleimide
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F22/00—Homopolymers and copolymers of compounds having one or more unsaturated aliphatic radicals each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides or nitriles thereof
- C08F22/36—Amides or imides
- C08F22/40—Imides, e.g. cyclic imides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G12/00—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen
- C08G12/02—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes
- C08G12/04—Condensation polymers of aldehydes or ketones with only compounds containing hydrogen attached to nitrogen of aldehydes with acyclic or carbocyclic compounds
- C08G12/06—Amines
- C08G12/08—Amines aromatic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/24—Impregnating materials with prepolymers which can be polymerised in situ, e.g. manufacture of prepregs
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/29—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the material, e.g. carbon
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01L—SEMICONDUCTOR DEVICES NOT COVERED BY CLASS H10
- H01L23/00—Details of semiconductor or other solid state devices
- H01L23/28—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection
- H01L23/31—Encapsulations, e.g. encapsulating layers, coatings, e.g. for protection characterised by the arrangement or shape
Definitions
- the present invention has a low melting point and softening point, excellent handleability, and a cured product having high heat resistance, and a maleimide resin or a maleimide compound that can be suitably used as a semiconductor encapsulating material, and a curability containing these.
- the present invention relates to a composition and a cured product thereof, a semiconductor encapsulating material, a semiconductor encapsulating device, a prepreg, a circuit board, and a build-up film.
- maleimide resin Since maleimide resin has extremely high heat resistance in cured products, its use is being considered as a resin material in fields that require particularly high heat resistance, such as encapsulation materials for power semiconductors, but it is currently on the market. Maleimide resin has a high melting point and softening point, and its low handleability as a material is a problem.
- a conventionally known maleimide resin for example, a 4,4'-diphenylmethanebismaleimide type compound is widely known, but as described above, the compound has a high melting point and is inferior in handleability as a material. (See, for example, Patent Document 1). Further, as a maleimide resin having relatively high handleability, a 2,2-bis [4- (4-maleimide phenoxy) phenyl] propane type compound is known, and the compound has a cured physical property such as heat resistance. , Did not meet the recent market demand (see, for example, Patent Document 2).
- the problem to be solved by the present invention is a maleimide resin or maleimide that has a low melting point and softening point and is excellent in handleability, and the cured product has high heat resistance and can be suitably used as a semiconductor encapsulating material or the like. It is an object of the present invention to provide a compound, a curable composition containing these and a cured product thereof, a semiconductor encapsulating material, a semiconductor encapsulating device, a prepreg, a circuit board, and a build-up film.
- a maleimide resin obtained by maleimizing a polyamine compound which is a reaction product of a plurality of aromatic monoamine compounds and a binder has a low melting point and softening point and is excellent in handleability.
- the cured product has high heat resistance and can be suitably used as a semiconductor encapsulating material, etc., and have completed the present invention.
- the present invention is a maleimide resin which is a maleimide product of a polyamine compound (C) which is a reaction product of a plurality of kinds of aromatic monoamine compounds (A) and a binder (B). Regarding.
- the present invention further relates to an asymmetric bismaleimide compound which is a maleimided product of an asymmetric diamine compound (C-1) in which two different aromatic monoamine compounds (A) are bound with a binder (B).
- the present invention further relates to a curable composition containing the maleimide resin or the asymmetrical bismaleimide compound.
- the present invention further relates to a cured product of the curable composition.
- the present invention further relates to a semiconductor encapsulation material using the curable composition.
- the present invention further relates to a semiconductor device using the semiconductor encapsulating material.
- the present invention further relates to a prepreg using the curable composition.
- the present invention further relates to a circuit board using the prepreg.
- the present invention further relates to a build-up film using the curable composition.
- a maleimide resin or a maleimide compound which has a low melting point and softening point and is excellent in handleability, has a high heat resistance of a cured product, and can be suitably used as a semiconductor encapsulation material, etc., is contained.
- a curable composition and a cured product thereof, a semiconductor encapsulating material, a semiconductor encapsulating device, a prepreg, a circuit board, and a build-up film can be provided.
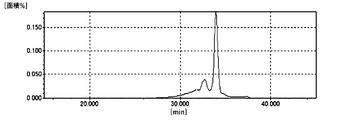
- FIG. 1 It is a GPC chart figure of the maleimide resin (1) obtained in Example 1.
- FIG. 2 is a GPC chart figure of the maleimide resin (2) obtained in Example 2.
- FIG. 2 is a GPC chart figure of the maleimide resin (3) obtained in Example 3.
- FIG. 2 is a GPC chart figure of the maleimide resin (4) obtained in Example 4.
- FIG. It is a GPC chart figure of the maleimide resin (5) obtained in Example 5.
- FIG. It is a GPC chart figure of the maleimide resin (6) obtained in Example 6.
- FIG. It is a differential scanning calorimetry (DSC) chart figure of the maleimide resin (1) obtained in Example 1.
- FIG. It is a differential scanning calorimetry (DSC) chart figure of the maleimide resin (2) obtained in Example 2.
- DSC differential scanning calorimetry
- the maleimide resin of the present invention is characterized by being a maleimide product of a polyamine compound (C) which is a reaction product of a plurality of aromatic monoamine compounds (A) and a binder (B).
- the aromatic monoamine compound (A) as long as it is a compound having one NH 2 on the aromatic ring, other specific structures are not particularly limited, and a wide variety of compounds can be used. Specifically, a compound having one NH 2 group on the aromatic ring of an aromatic compound such as benzene, naphthalene, and anthracene, and a compound having one or more other substituents in addition to the NH 2 group, etc. Can be mentioned. Examples of the other substituent include an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, a halogen atom, an aryl group, an aralkyl group, a hydroxyl group and the like.
- the aliphatic hydrocarbon group may have a linear type, a branched type, or a cyclic structure, and may have an unsaturated bond in the structure. Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group, a pentyl group, a hexyl group, a cyclohexyl group, a heptyl group, an octyl group and a nonyl group.
- the alkyloxy group include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Examples of the alkenyloxy group include an allyloxy group.
- Examples of the halogen atom include a fluorine atom, a chlorine atom and a bromine atom.
- Examples of the aryl group include a phenyl group, a naphthyl group, an anthryl group, and a structural site in which the aliphatic hydrocarbon group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- aralkyl group examples include a benzyl group, a phenylethyl group, a naphthylmethyl group, a naphthylethyl group, and a structural site in which the alkyl group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- the obtained maleimide resin has a low melting point and softening point and is excellent in handleability. Therefore, one or a plurality of the other substituents are added on the aniline or the aromatic nucleus of aniline.
- the compound to have is preferable.
- aniline, a compound having a substituent at the 2-position of aniline, and a compound having a substituent at the 2,6-position of aniline are particularly preferable.
- the type of the substituent of the compound having a substituent at the 2-position of aniline and the compound having a substituent at the 2,6-position of aniline is a maleimide resin having excellent heat resistance in a cured product, and therefore a carbon atom.
- An aliphatic hydrocarbon group having 1 to 4 carbon atoms is preferable, and an alkyl group having 1 to 4 carbon atoms is more preferable.
- the maleimide resin has a low melting point and softening point and is excellent in handleability while maintaining the high heat resistance characteristic of the maleimide resin.
- the number of the aromatic monoamine compound (A) to be used may be a plurality of kinds, that is, two or more kinds, and the upper limit is not particularly limited, but since it can be produced relatively easily, it is in the range of 2 to 5 kinds. It is preferable to use it, and it is more preferable to use two or three kinds in combination.
- the amount of each aromatic monoamine compound (A) used is at least 10 with respect to the total of the aromatic monoamine compounds (A) because the melting point and the softening point are low and the effect of excellent handleability is sufficiently exhibited. It is preferably 5% by mass or more, and more preferably 25% by mass or more. The upper limit thereof is preferably 90% or less, and more preferably 75% or less.
- the mass ratio of the two is preferably in the range of 10/90 to 90/10. More preferably, it is in the range of 20/80 to 80/20.
- the specific structure of the binder (B) is not particularly limited as long as it is a compound that reacts with the aromatic monoamine compound (A) to bind the aromatic rings of the aromatic monoamine compound (A) to each other. Compounds can be used. Further, the binder (B) may be used alone or in combination of two or more. Specific examples of the binder (B) include an aldehyde compound (B-1), a ketone compound (B-2), an aromatic compound (B-3) represented by the following general formula (B-3), and the following.
- Ar 1 represents an aromatic ring which may independently have a substituent.
- R 1 is independently a hydrogen atom or a methyl group.
- R 2 is an independently hydrogen atom or an aliphatic hydrocarbon group having 1 to 4 carbon atoms.
- R 3 is independently any of an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group and an aralkyl group, and l is an integer of 0 to 3.
- X is any of a hydroxyl group, a halogen atom, and an alkyloxy group.
- Y is any one of a single bond, a divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms, an oxygen atom, a sulfur atom, and a sulfonyl group.
- aldehyde compound (B-1) examples include aliphatic aldehyde compounds such as formaldehyde and acetaldehyde, and aromatic aldehyde compounds such as benzaldehyde and naphthaldehyde. One of these may be used alone, or two or more thereof may be used in combination.
- Examples of the ketone compound (B-2) include aliphatic ketone compounds such as acetone, methyl ethyl ketone and diethyl ketone, and aromatic ketone compounds such as acetophenone. One of these may be used alone, or two or more thereof may be used in combination.
- Ar 1 represents an aromatic ring which may independently have a substituent. Specific examples thereof include a phenylene group, a naphthylene group, and a structural site having one or a plurality of various substituents on these aromatic rings. Examples of the substituent include an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, a halogen atom, an aryl group, an aralkyl group, a hydroxyl group and the like.
- the aliphatic hydrocarbon group may have a linear type, a branched type, or a cyclic structure, and may have an unsaturated bond in the structure.
- Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group, a pentyl group, a hexyl group, a cyclohexyl group, a heptyl group, an octyl group and a nonyl group.
- Examples of the alkyloxy group include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Examples of the alkenyloxy group include an allyloxy group.
- the halogen atom include a fluorine atom, a chlorine atom and a bromine atom.
- Examples of the aryl group include a phenyl group, a naphthyl group, an anthryl group, and a structural site in which the aliphatic hydrocarbon group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- Examples of the aralkyl group include a benzyl group, a phenylethyl group, a naphthylmethyl group, a naphthylethyl group, and a structural site in which the alkyl group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- R 2 is an independent hydrogen atom or an aliphatic hydrocarbon group having 1 to 4 carbon atoms, respectively.
- the aliphatic hydrocarbon group having 1 to 4 carbon atoms may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure. Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group and the like.
- R 3 is independently composed of an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group and an aralkyl group. It is either, and l is an integer of 0 to 3.
- the aliphatic hydrocarbon group may have a linear type, a branched type, or a cyclic structure, and may have an unsaturated bond in the structure.
- Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group, a pentyl group, a hexyl group, a cyclohexyl group, a heptyl group, an octyl group and a nonyl group.
- Examples of the alkyloxy group include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Examples of the alkenyloxy group include an allyloxy group.
- the halogen atom include a fluorine atom, a chlorine atom and a bromine atom.
- Examples of the aryl group include a phenyl group, a naphthyl group, an anthryl group, and a structural site in which the aliphatic hydrocarbon group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- Examples of the aralkyl group include a benzyl group, a phenylethyl group, a naphthylmethyl group, a naphthylethyl group, and a structural site in which the alkyl group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- X is any of a hydroxyl group, a halogen atom, and an alkyloxy group.
- alkyloxy group examples include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Y is any one of a single bond, a divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms, an oxygen atom, a sulfur atom, and a sulfonyl group. ..
- the divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure.
- the reaction step of reacting the aromatic monoamine compound (A) with the binder (B) to obtain the polyamine compound (C) is, for example, a plurality of types of the aromatic monoamine compound (A) and the binder ( Examples thereof include a method of reacting with B) under acidic catalyst conditions.
- the reaction may be carried out in a solvent as appropriate. Further, the reaction can be efficiently promoted by heating to about 50 to 200 ° C.
- the polyamine compound (C) as an intermediate can be obtained by washing with an alkaline aqueous solution, distilled water or the like.
- the acidic catalyst examples include p-toluenesulfonic acid, dimethylsulfuric acid, diethylsulfuric acid, sulfuric acid, hydrochloric acid, oxalic acid, and activated clay. One of these may be used alone, or two or more thereof may be used in combination.
- the amount of the acid catalyst added is preferably in the range of 0.01 to 0.5 mol, preferably 0.1 to 0.3 mol, with respect to 2 mol of the aromatic monoamine compound (A).
- the range ratio is more preferable. When the number of moles cannot be defined, the ratio is preferably in the range of 1 wt% to 50 wt% with respect to the total amount of the aniline compound (A), the binder (B), the solvent and the acidic catalyst.
- the solvent examples include distilled water and organic solvents such as toluenexylene. These may be used alone or as a mixed solvent of two or more kinds.
- the amount of the solvent used is preferably in the range of 5 to 100% by mass with respect to the total mass of the aromatic monoamine compound (A) and the binder (B).
- Examples of the maleimideization reaction of the polyamine compound (C) include a method of reacting the polyamine compound (C) with an acid anhydride under acidic catalytic conditions.
- reaction control becomes easy, it is preferable to add the acid anhydride in portions to the polyamine compound (C), or to dissolve the acid anhydride in an appropriate solvent and drop the acid anhydride.
- the reaction may be carried out in a solvent as appropriate.
- the polyamine compound (C) and the acid anhydride are stirred at room temperature to obtain an amic acid intermediate.
- an acid catalyst is added and heated to 50 to 200 ° C., more preferably 70 to 150 ° C. to proceed the reaction. At this time, it is preferable to remove the water in the system.
- the desired maleimide resin can be obtained by washing with an alkaline aqueous solution or distilled water.
- acid anhydride examples include maleic anhydride, citraconic acid anhydride, 2,3-dimethylmaleic acid anhydride and the like. One of these may be used alone, or two or more thereof may be used in combination.
- the acidic catalyst examples include p-toluenesulfonic acid, hydroxy-p-toluenesulfonic acid, methanesulfonic acid, sulfuric acid, phosphoric acid and the like. One of these may be used alone, or two or more thereof may be used in combination.
- the amount of the acidic catalyst added is usually 0.01 to 10 mol, preferably 0.03 to 3 mol, based on 1 g / mol of the amino group equivalent of the polyaniline compound (C).
- the solvent may be any one that can dissolve the polyamine compound (C) and the acid anhydride.
- a mixed solvent of a non-polar solvent such as toluene and an aprotic polar solvent such as dimethylformamide is used. It is preferable to use.
- the non-polar solvent include xylene, chlorobenzene and the like in addition to toluene.
- the aprotic polar solvent include dimethylformaldehyde and methylethylketone. The compounding ratio of both and the amount of the solvent used are appropriately adjusted depending on the solvent solubility of the polyamine compound (C) and the acid anhydride.
- the mass ratio of the non-protic solvent to the aprotic solvent is in the range of 1/99 to 99/1, and the total solvent amount is the sum of the polyamine compound (C), the acid anhydride and the total solvent amount.
- the range is 0.5 to 80% can be mentioned.
- the molecular weight of the maleimide resin of the present invention is not particularly limited, and the reaction conditions and the like can be appropriately changed according to the intended use and adjusted to a preferable value.
- it when it is used as a semiconductor encapsulant material, it is represented by the following formula (1) because it is a resin having a low melting point and softening point and excellent handling property while maintaining high heat resistance in a cured product.
- Such dinuclear component, trinuclear component as represented by the following structural formula (2), tetranuclear component represented by the following formulas (3-1) and (3-2), etc. are relatively low. It preferably contains a component having a molecular weight.
- A is a structural moiety derived from the aromatic monoamine compound (A) and has a maleimide group
- B is a structural moiety derived from the binder (B).
- a and B in the formula may be the same or different.
- the maleimide resin contains a dinuclear component (bismaleimide compound).
- the ratio of the binuclear component (bismaleimide compound) in the maleimide resin is preferably 30% or more, more preferably 50% or more.
- the content of the binuclear body in the maleimide resin is a value calculated from the area ratio of the gel permeation chromatography (GPC) chart. Further, in the present invention, the measurement conditions of gel permeation chromatography (GPC) are described in Examples.
- the "number of nuclei” is the number of structural sites derived from the aromatic monoamine compound (A) in the molecule as shown in the formulas (1) to (3-2).
- the dinuclear component bismaleimide compound
- two different aromatic monoamine compounds are obtained because they are resins having low melting point and softening point and excellent handling property while maintaining high heat resistance in the cured product.
- An asymmetric bismaleimide compound which is a maleimided product of the asymmetric diamine compound (C-1) to which (A) is bound with the binder (B), is preferable.
- a compound using an aniline compound as the aromatic monoamine compound (A) is preferable, and an asymmetric bismaleimide compound represented by the following structural formula (4) is more preferable.
- the asymmetric maleimide compound may be isolated and purified before use.
- [Z in the formula is a divalent organic group having 1 to 200 carbon atoms.
- Each of R4 is independently an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group, or an aralkyl group. If there are multiple R4s in the equation, they may be the same or different.
- m is 0 or an integer of 1 to 4.
- the structural part ⁇ and the structural part ⁇ surrounded by the broken line in the formula have different structures from each other. ]
- Z in the structural formula (4) is a structural site derived from the binder (B).
- Z is a divalent organic group having 1 to 200 carbon atoms, but may be a structural moiety containing other atoms such as oxygen atom and halogen atom as long as the carbon atom number is in the range of 1 to 200. .. Above all, a divalent organic group having 1 to 20 carbon atoms is more preferable.
- Specific examples of the Z include structural parts represented by the following general formulas (Z-1) to (Z-8).
- Ar 1 represents an aromatic ring which may independently have a substituent.
- R 3 is independently any of an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group and an aralkyl group, and l is an integer of 0 to 3.
- R 5 represents an aromatic ring which may independently have a hydrogen atom, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, or a substituent.
- R 6 independently represents a hydrogen atom or an aliphatic hydrocarbon group having 1 to 4 carbon atoms.
- R 7 is either a divalent aliphatic hydrocarbon group other than the one represented by the general formula (Z-1), an aromatic group which may have a substituent, or a combination thereof.
- Y is any one of a single bond, a divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms, an oxygen atom, a sulfur atom, and a sulfonyl group.
- n is an integer of 1 or more.
- R 5 in the general formula (Z-1) represents an aromatic ring which may independently have a hydrogen atom, an aliphatic hydrocarbon group having 1 to 4 carbon atoms, or a substituent.
- the aliphatic hydrocarbon group having 1 to 4 carbon atoms may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure. Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group and the like.
- Examples of the aromatic ring that may have the substituent include a phenylene group, a naphthylene group, and a structural site having one or a plurality of various substituents on these aromatic rings.
- Examples of the substituent include an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, a halogen atom, an aryl group, an aralkyl group, a hydroxyl group and the like.
- the aliphatic hydrocarbon group may have a linear type, a branched type, or a cyclic structure, and may have an unsaturated bond in the structure.
- Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group, a pentyl group, a hexyl group, a cyclohexyl group, a heptyl group, an octyl group and a nonyl group.
- Examples of the alkyloxy group include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Examples of the alkenyloxy group include an allyloxy group.
- the halogen atom include a fluorine atom, a chlorine atom and a bromine atom.
- Examples of the aryl group include a phenyl group, a naphthyl group, an anthryl group, and a structural site in which the aliphatic hydrocarbon group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- Examples of the aralkyl group include a benzyl group, a phenylethyl group, a naphthylmethyl group, a naphthylethyl group, and a structural site in which the alkyl group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- Ar 1 in the general formulas (Z-2), (Z-3) and (Z-8) represents an aromatic ring which may independently have a substituent. Specific examples thereof include those similar to Ar 1 in the general formulas (B-3) to (B-6).
- R 6 in the general formulas (Z-2) and (Z-3) independently represent a hydrogen atom or an aliphatic hydrocarbon group having 1 to 4 carbon atoms.
- the aliphatic hydrocarbon group having 1 to 4 carbon atoms may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure. Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group and the like.
- Y in the general formula (Z-3) is any one of a single bond, a divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms, an oxygen atom, a sulfur atom, and a sulfonyl group.
- the divalent aliphatic hydrocarbon group having 1 to 6 carbon atoms may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure.
- R 3 in the general formula (Z-4) is independently any of an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group, and an aralkyl group. Is an integer from 0 to 3. Specific examples thereof include those similar to R3 in the general formulas (B - 7) and (B-8).
- R 7 in the general formula (Z-7) is a divalent aliphatic hydrocarbon group other than that represented by the general formula (Z-1), an aromatic group which may have a substituent, or an aromatic group. It is one of the combinations.
- the divalent aliphatic hydrocarbon group may have a linear type, a branched type or a cyclic structure, and may have an unsaturated bond in the structure.
- R 4 in the structural formula (4) is independently any one of an aliphatic hydrocarbon group, an alkyloxy group, an alkenyloxy group, an alkynyloxy group, a halogen atom, an aryl group and an aralkyl group.
- the aliphatic hydrocarbon group may have a linear type, a branched type, or a cyclic structure, and may have an unsaturated bond in the structure.
- Specific examples thereof include a methyl group, an ethyl group, a vinyl group, a propyl group, an allyl group, a butyl group, a pentyl group, a hexyl group, a cyclohexyl group, a heptyl group, an octyl group and a nonyl group.
- Examples of the alkyloxy group include a methoxy group, an ethoxy group, a propyloxy group, a butoxy group and the like.
- Examples of the alkenyloxy group include an allyloxy group.
- the halogen atom include a fluorine atom, a chlorine atom and a bromine atom.
- aryl group examples include a phenyl group, a naphthyl group, an anthryl group, and a structural site in which the aliphatic hydrocarbon group, an alkoxy group, a halogen atom and the like are substituted on these aromatic nuclei.
- aralkyl group examples include a benzyl group, a phenylethyl group, a naphthylmethyl group, a naphthylethyl group, and a structural site in which the alkyl group, an alkoxy group, a halogen atom and the like are substituted on the aromatic ring thereof. If there are multiple R4s in the equation, they may be the same or different.
- the obtained maleimide resin has a low melting point and softening point and is excellent in handleability, it is preferable to have a substituent on one or both of the carbon atoms adjacent to the carbon atom substituted by the maleimide group.
- the substituent is preferably an aliphatic hydrocarbon group having 1 to 4 carbon atoms, and more preferably an alkyl group having 1 to 4 carbon atoms.
- the curable composition of the present invention contains the maleimide resin or the asymmetrical bismaleimide compound.
- the maleimide resin or the asymmetrical bismaleimide compound may be used alone as the curable component, or one or a plurality of other curable compounds may be used in combination.
- Examples of the other curable compound include epoxy resin, phenol resin, amine compound, active ester resin, cyanate ester resin, benzoxazine resin, unsaturated bond-containing compound and the like.
- epoxy resin examples include various bisphenol type epoxy resins, various biphenyl type epoxy resins, various novolak type epoxy resins, dicyclopentadiene-phenol addition reaction type epoxy resins, and phenol aralkyl type epoxy resins. One of these may be used alone, or two or more thereof may be used in combination.
- phenol resin examples include various bisphenols, various biphenyls, various novolak resins, dicyclopentadiene-phenol addition reaction type resins, phenol aralkyl type resins, and various arylene ether resins. One of these may be used alone, or two or more thereof may be used in combination.
- the curable composition of the present invention may contain various additives such as a curing accelerator, a flame retardant, an inorganic filler, a silane coupling agent, a mold release agent, a pigment, and an emulsifier, if necessary.
- the curing accelerator examples include phosphorus-based compounds, peroxides, tertiary amines, imidazole compounds, pyridine compounds, organic acid metal salts, Lewis acids, amine complex salts and the like.
- triphenylphosphine for phosphorus-based compounds, dikmylperoxide for peroxides, and 1,8-diazabicyclo- [5] for tertiary amines are excellent in curability, heat resistance, electrical properties, moisture resistance reliability, etc.
- -Undecene (DBU) 2-ethyl-4-methylimidazole for imidazole compounds
- 4-dimethylaminopyridine for pyridine compounds are preferred.
- the flame retardant may be, for example, red phosphorus, monoammonium phosphate, diammonium phosphate, triammonium phosphate, ammonium phosphate such as ammonium polyphosphate, inorganic phosphorus compounds such as phosphate amide; phosphoric acid ester compounds, phosphonic acid.
- Cyclic organic phosphorus Organophosphorus compounds such as compounds and derivatives obtained by reacting them with compounds such as epoxy resins and phenol resins; nitrogen-based flame retardants such as triazine compounds, cyanuric acid compounds, isocyanuric acid compounds and phenothiazine; silicone oils, silicone rubbers and silicones.
- Silicone-based flame retardants such as resins; examples thereof include metal hydroxides, metal oxides, metal carbonate compounds, metal powders, boron compounds, and inorganic flame retardants such as low melting point glass. When these flame retardants are used, it is preferably in the range of 0.1 to 20% by mass with respect to the resin solid content of the curable composition.
- the inorganic filler is blended, for example, when the curable composition of the present invention is used for semiconductor encapsulation material applications.
- the inorganic filler include fused silica, crystalline silica, alumina, silicon nitride, aluminum hydroxide and the like. Above all, the molten silica is preferable because it is possible to blend a larger amount of the inorganic filler.
- the fused silica can be used in either a crushed form or a spherical shape, but in order to increase the blending amount of the fused silica and suppress the increase in the melt viscosity of the curable composition, a spherical one is mainly used. Is preferable.
- the filling rate is preferably in the range of 0.5 to 95 parts by mass in 100 parts by mass of the curable composition.
- a conductive filler such as silver powder or copper powder can be used.
- the curable composition of the present invention has a low melting point and softening point and is excellent in handleability, and since the cured product has high heat resistance, it can be particularly preferably used as a semiconductor encapsulating material. It can be widely used for electronic materials such as printed wiring boards and resist materials, and for paints, adhesives, molded products, and the like.
- the semiconductor encapsulation material can be prepared by mixing the formulations using, for example, an extruder, a kneader, a roll, or the like.
- the semiconductor encapsulating material is molded by casting or using a transfer molding machine, an injection molding machine, or the like, and further, the temperature is 50 to 250 ° C. A method of heating under the conditions for 1 to 10 hours can be mentioned, and a semiconductor device which is a molded product can be obtained by such a method.
- the curable composition of the present invention When the curable composition of the present invention is used for a printed wiring board application or a build-up adhesive film application, it is generally preferable to mix and dilute it with an organic solvent.
- the organic solvent include methyl ethyl ketone, acetone, dimethylformamide, methyl isobutyl ketone, methoxypropanol, cyclohexanone, methyl cellosolve, ethyl diglycol acetate, propylene glycol monomethyl ether acetate and the like.
- the type and blending amount of the organic solvent can be appropriately adjusted according to the usage environment of the curable composition. For example, in the case of printed wiring board applications, a polar solvent having a boiling point of 160 ° C.
- ketone solvents such as acetone, methyl ethyl ketone and cyclohexanone
- acetate solvents such as ethyl acetate, butyl acetate, cellosolve acetate, propylene glycol monomethyl ether acetate and carbitol acetate
- carbitol such as cellosolve and butyl carbitol.
- a solvent an aromatic hydrocarbon solvent such as toluene and xylene, dimethylformamide, dimethylacetamide, N-methylpyrrolidone and the like, and it is preferable to use the non-volatile content at a ratio of 30 to 60% by mass.
- the curable composition is impregnated into a reinforcing base material and cured to obtain a prepreg, which is then heat-bonded by overlapping.
- the method can be mentioned.
- the reinforcing base material include paper, glass cloth, glass non-woven fabric, aramid paper, aramid cloth, glass mat, and glass roving cloth.
- the impregnation amount of the curable composition is not particularly limited, but it is usually preferable to adjust the resin content in the prepreg to be 20 to 60% by mass.
- Example 1 Synthesis of maleimide resin (1)
- the reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene.
- the diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 109.48 g of the polyaniline compound (1).
- the amine equivalent of the polyaniline compound (1) was 146 eq / g.
- Each peak corresponds to an ammonia adduct of the following compound.
- the content of the dinuclear component (bismaleimide compound) calculated from the area ratio in the GPC chart was 96%.
- the GPC chart of the maleimide resin (1) is shown in FIG.
- Example 2 Synthesis of maleimide resin (2)
- a 500 mL eggplant flask attached to a rotary evaporator 52.11 g (0.43 mol) of 2-ethylaniline, 64.17 g (0.43 mol) of 2,6-diethylaniline, 22.14 g of distilled water and 22 p-toluenesulfonic acid. .73 g was charged and heated to 70 ° C. with stirring. After holding the ring at 70 ° C. for 30 minutes, 34.98 g (0.43 mol) of a 37% formalin solution was added in 4 portions over 1 hour and reacted for 4 hours.
- the reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene.
- the diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 119.02 g of the polyaniline compound (2).
- the amine equivalent of the polyaniline compound (2) was 165 eq / g.
- Each peak corresponds to an ammonia adduct of the following compound.
- the content of the dinuclear component (bismaleimide compound) calculated from the area ratio in the GPC chart was 56%.
- the GPC chart of the maleimide resin (2) is shown in FIG.
- reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene.
- the diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 106.11 g of the polyaniline compound (4).
- the amine equivalent was 147 eq / g.
- Example 4 to 6 Synthesis of maleimide resins (4) to (6)
- Maleimide resins (4) to (6) were synthesized in the same procedure as in Example 1 except that the type and number of moles of the aniline compound were changed as shown in Table 1 below.
- the GPC charts of the maleimide resins (4) to (6) are shown in FIGS. 4 to 6.
- Table 1 shows the content of the binuclear component (bismaleimide compound) of each maleimide resin calculated from the area ratio of the GPC chart. Moreover, it was confirmed from the MS spectrum of each maleimide resin that each of them contained an asymmetrical bismaleimide compound.
- Examples 7 to 12 Evaluation of maleimide resins (1) to (6)
- the melting point, softening point, Td5 of the cured product, and the coefficient of thermal expansion of the cured product of each maleimide resin were measured and evaluated in the following manner. The evaluation results are shown in Table 2.
- Td5 of cured product The maleimide resins obtained in Examples 1 to 6 were poured into molds of 11 cm ⁇ 5 cm ( ⁇ thickness of about 1 mm) and cured at 200 ° C. for 2 hours and further at 250 ° C. for 2 hours to obtain a cured product. .. Td5 of the obtained cured product was measured using TGA / DSC manufactured by METTLER TOLEDO CO., LTD. Measuring equipment: METTLER TOLEDO Co., Ltd.
- TGA / DSC 1 Measurement range: 40 ° C to 150 ° C to 600 ° C Temperature rise rate: 20 ° C / min (40 ° C ⁇ 150 ° C) Hold for 15 minutes (150 ° C) 5 ° C / min (150 ° C ⁇ 600 ° C) Atmosphere: Nitrogen
Abstract
Description
本発明のマレイミド樹脂は複数種の芳香族モノアミン化合物(A)と、結合剤(B)との反応生成物であるポリアミン化合物(C)のマレイミド化物であることを特徴とする。 Hereinafter, the present invention will be described in detail.
The maleimide resin of the present invention is characterized by being a maleimide product of a polyamine compound (C) which is a reaction product of a plurality of aromatic monoamine compounds (A) and a binder (B).
測定装置 :東ソー株式会社製「HLC-8320 GPC」、
カラム:東ソー株式会社製ガードカラム「HXL-L」
+東ソー株式会社製「TSK-GEL G2000HXL」
+東ソー株式会社製「TSK-GEL G2000HXL」
+東ソー株式会社製「TSK-GEL G3000HXL」
+東ソー株式会社製「TSK-GEL G4000HXL」
検出器: RI(示差屈折計)
データ処理:東ソー株式会社製「GPCワークステーション EcoSEC-WorkStation」
測定条件: カラム温度 40℃
展開溶媒 テトラヒドロフラン
流速 1.0ml/分
標準 : 前記「GPCワークステーション EcoSEC-WorkStation」の測定マニュアルに準拠して、分子量が既知の下記の単分散ポリスチレンを用いた。
(使用ポリスチレン)
東ソー株式会社製「A-500」
東ソー株式会社製「A-1000」
東ソー株式会社製「A-2500」
東ソー株式会社製「A-5000」
東ソー株式会社製「F-1」
東ソー株式会社製「F-2」
東ソー株式会社製「F-4」
東ソー株式会社製「F-10」
東ソー株式会社製「F-20」
東ソー株式会社製「F-40」
東ソー株式会社製「F-80」
東ソー株式会社製「F-128」
試料 : 樹脂固形分換算で1.0質量%のテトラヒドロフラン溶液をマイクロフィルターでろ過したもの(50μl)。 <Measurement conditions for gel permeation chromatography (GPC)>
Measuring device: "HLC-8320 GPC" manufactured by Tosoh Corporation,
Column: Guard column "HXL-L" manufactured by Tosoh Corporation
+ "TSK-GEL G2000HXL" manufactured by Tosoh Corporation
+ "TSK-GEL G2000HXL" manufactured by Tosoh Corporation
+ "TSK-GEL G3000HXL" manufactured by Tosoh Corporation
+ "TSK-GEL G4000HXL" manufactured by Tosoh Corporation
Detector: RI (Differential Refractometer)
Data processing: "GPC Workstation EcoSEC-WorkStation" manufactured by Tosoh Corporation
Measurement conditions: Column temperature 40 ° C
Developing solvent Tetrahydrofuran Flow rate 1.0 ml / min Standard: The following monodisperse polystyrene with a known molecular weight was used in accordance with the measurement manual of the above-mentioned "GPC workstation EcoSEC-WorkStation".
(Polystyrene used)
"A-500" manufactured by Tosoh Corporation
"A-1000" manufactured by Tosoh Corporation
"A-2500" manufactured by Tosoh Corporation
"A-5000" manufactured by Tosoh Corporation
"F-1" manufactured by Tosoh Corporation
"F-2" manufactured by Tosoh Corporation
"F-4" manufactured by Tosoh Corporation
"F-10" manufactured by Tosoh Corporation
"F-20" manufactured by Tosoh Corporation
"F-40" manufactured by Tosoh Corporation
"F-80" manufactured by Tosoh Corporation
"F-128" manufactured by Tosoh Corporation
Sample: A solution of 1.0% by mass in tetrahydrofuran in terms of resin solid content filtered through a microfilter (50 μl).
コントローラ:Agilent Technologies 1260 Infinity II
カラム:Agilent EC-C18(4.6×50mm、2.7μm)
カラム温度 :40℃
ポンプ流速 :1.0ml/分
溶離条件 :K1-水、K2-アセトニトリル
K1/K2=0/100→30/70(線形に濃度変化0-1.67分)
K1/K2=30/70(1.67-5分)
K1/K2=30/70→90/10(5-8分)
(比率は体積比)
検出波長 :UV254、275、300nm
MS :Agilent Technologies InfinityLab LC/MSD <Measurement conditions of high performance liquid chromatography (HPLC) and liquid chromatography mass spectrometry (LC-MS)>
Controller: Agilent Technologies 1260 Infinity II
Column: Agilent EC-C18 (4.6 x 50 mm, 2.7 μm)
Column temperature: 40 ° C
Pump flow rate: 1.0 ml / min Elution conditions: K1-water, K2-acetonitrile K1 / K2 = 0/100 → 30/70 (linear concentration change 0-1.67 min)
K1 / K2 = 30/70 (1.67-5 minutes)
K1 / K2 = 30/70 → 90/10 (5-8 minutes)
(Ratio is volume ratio)
Detection wavelength: UV254, 275, 300nm
MS: Agilent Technologies InfinityLab LC / MSD
500mL共栓付き三角フラスコに、試料であるポリアニリン化合物を約2.5g入れ、ピリジン7.5g、無水酢酸2.5g、トリフェニルホスフィン7.5gを精秤後、冷却管を装着し、120℃に設定したオイルバスにて150分加熱還流する。
混合物を冷却した後、蒸留水5.0mL、プロピレングリコールモノメチルエーテル100mL、テトラヒドロフラン75mL、0.5mol/L水酸化カリウム-エタノール溶液(~50mL)を加え、電位差滴定法により滴定した。同様の方法で空試験を行なって補正した。
アミン当量(g/eq.)=(S×2,000)/(Blank-A)
S:試料の量(g)
A:0.5mol/L水酸化カリウム-エタノール溶液の消費量(mL)
Blank:空試験における0.5mol/L水酸化カリウム-エタノール溶液の消費量(mL) <Measurement of amine equivalent>
Approximately 2.5 g of the sample polyaniline compound was placed in a 500 mL Erlenmeyer flask with a stopper, 7.5 g of pyridine, 2.5 g of acetic anhydride, and 7.5 g of triphenylphosphine were precisely weighed, and then a cooling tube was attached. Heat and reflux for 150 minutes in the oil bath set in.
After cooling the mixture, 5.0 mL of distilled water, 100 mL of propylene glycol monomethyl ether, 75 mL of tetrahydrofuran, and a 0.5 mol / L potassium hydroxide-ethanol solution (~ 50 mL) were added, and titration was performed by a potential difference titration method. A blank test was performed in the same manner to correct the problem.
Amine equivalent (g / eq.) = (S × 2,000) / (Blank-A)
S: Amount of sample (g)
A: Consumption (mL) of 0.5 mol / L potassium hydroxide-ethanol solution
Blank: Consumption (mL) of 0.5 mol / L potassium hydroxide-ethanol solution in blank test
ロータリーエバポレーターに取り付けた500mLナスフラスコに、2,6-キシリジン52.40g(0.43mol)、2,6-ジエチルアニリン64.52g(0.43mol)、蒸留水22.14gおよびp-トルエンスルホン酸22.73gを仕込み、攪拌しながら70℃まで加熱した。70℃で30分間保持した後、37%ホルマリン溶液34.98g(0.43mol)を1時間かけて4分割投入し、4時間反応させた。反応後、室温まで空冷し、反応溶液を2Lのセパラブルフラスコに移し、トルエン140gで希釈した。希釈溶液は10%水酸化ナトリウム水溶液100gで1回、蒸留水100gで4回洗浄し、減圧濃縮してポリアニリン化合物(1)109.48gを得た。ポリアニリン化合物(1)のアミン当量は146eq/gであった。 (Example 1: Synthesis of maleimide resin (1))
52.40 g (0.43 mol) of 2,6-xylidine, 64.52 g (0.43 mol) of 2,6-diethylaniline, 22.14 g of distilled water and p-toluenesulfonic acid in a 500 mL eggplant flask attached to a rotary evaporator. 22.73 g was charged and heated to 70 ° C. with stirring. After holding at 70 ° C. for 30 minutes, 34.98 g (0.43 mol) of a 37% formalin solution was added in 4 portions over 1 hour and reacted for 4 hours. After the reaction, the mixture was air-cooled to room temperature, the reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene. The diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 109.48 g of the polyaniline compound (1). The amine equivalent of the polyaniline compound (1) was 146 eq / g.
ロータリーエバポレーターに取り付けた500mLナスフラスコに、2-エチルアニリン52.11g(0.43mol)、2,6-ジエチルアニリン64.17g(0.43mol)、蒸留水22.14gおよびp-トルエンスルホン酸22.73gを仕込み、攪拌しながら70℃まで加熱した。70℃で30分環保持した後、37%ホルマリン溶液34.98g(0.43mol)を1時間かけて4分割投入し、4時間反応させた。反応後、室温まで空冷し、反応溶液を2Lのセパラブルフラスコに移し、トルエン140gで希釈した。希釈溶液は10%水酸化ナトリウム水溶液100gで1回、蒸留水100gで4回洗浄し、減圧濃縮してポリアニリン化合物(2)119.02gを得た。ポリアニリン化合物(2)のアミン当量は165eq/gであった。 (Example 2: Synthesis of maleimide resin (2))
In a 500 mL eggplant flask attached to a rotary evaporator, 52.11 g (0.43 mol) of 2-ethylaniline, 64.17 g (0.43 mol) of 2,6-diethylaniline, 22.14 g of distilled water and 22 p-toluenesulfonic acid. .73 g was charged and heated to 70 ° C. with stirring. After holding the ring at 70 ° C. for 30 minutes, 34.98 g (0.43 mol) of a 37% formalin solution was added in 4 portions over 1 hour and reacted for 4 hours. After the reaction, the mixture was air-cooled to room temperature, the reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene. The diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 119.02 g of the polyaniline compound (2). The amine equivalent of the polyaniline compound (2) was 165 eq / g.
ロータリーエバポレーターに取り付けた500mLナスフラスコに、2-エチルアニリン52.11g(0.43mol)、2,6-キシリジン52.11g(0.43mol)、蒸留水22.14gおよびp-トルエンスルホン酸22.73gを仕込み、攪拌しながら70℃まで加熱した。70℃で30分間保持した後、37%ホルマリン溶液34.98g(0.43mol)を1時間かけて4分割投入し、4時間反応させた。反応後、室温まで空冷し、反応溶液を2Lのセパラブルフラスコに移し、トルエン140gで希釈した。希釈溶液は10%水酸化ナトリウム水溶液100gで1回、蒸留水100gで4回洗浄し、減圧濃縮してポリアニリン化合物(4)106.11gを得た。アミン当量は147eq/gであった。 (Example 3: Synthesis of maleimide resin (3))
In a 500 mL eggplant flask attached to a rotary evaporator, 52.11 g (0.43 mol) of 2-ethylaniline, 52.11 g (0.43 mol) of 2,6-xylidine, 22.14 g of distilled water and 22. of p-toluenesulfonic acid. 73 g was charged and heated to 70 ° C. with stirring. After holding at 70 ° C. for 30 minutes, 34.98 g (0.43 mol) of a 37% formalin solution was added in 4 portions over 1 hour and reacted for 4 hours. After the reaction, the mixture was air-cooled to room temperature, the reaction solution was transferred to a 2 L separable flask, and diluted with 140 g of toluene. The diluted solution was washed once with 100 g of a 10% aqueous sodium hydroxide solution and four times with 100 g of distilled water, and concentrated under reduced pressure to obtain 106.11 g of the polyaniline compound (4). The amine equivalent was 147 eq / g.
アニリン化合物の種類とモル数を下記表1に示す通り変更した以外は、実施例1と同様の手順でマレイミド樹脂(4)~(6)を合成した。マレイミド樹脂(4)~(6)のGPCチャート図を図4~6に示す。
GPCチャート図の面積比から算出される各マレイミド樹脂の2核体成分(ビスマレイミド化合物)の含有量を表1に示す。
また、各マレイミド樹脂のMSスペクトルから、それぞれ非対称ビスマレイミド化合物を含有していることを確認した。 (Examples 4 to 6: Synthesis of maleimide resins (4) to (6))
Maleimide resins (4) to (6) were synthesized in the same procedure as in Example 1 except that the type and number of moles of the aniline compound were changed as shown in Table 1 below. The GPC charts of the maleimide resins (4) to (6) are shown in FIGS. 4 to 6.
Table 1 shows the content of the binuclear component (bismaleimide compound) of each maleimide resin calculated from the area ratio of the GPC chart.
Moreover, it was confirmed from the MS spectrum of each maleimide resin that each of them contained an asymmetrical bismaleimide compound.
下記の要領で各マレイミド樹脂の融点、軟化点、硬化物のTd5、及び硬化物の熱膨張率を測定し、評価した。評価結果を表2に示す。 (Examples 7 to 12: Evaluation of maleimide resins (1) to (6))
The melting point, softening point, Td5 of the cured product, and the coefficient of thermal expansion of the cured product of each maleimide resin were measured and evaluated in the following manner. The evaluation results are shown in Table 2.
実施例1~6で得られたマレイミド樹脂について、示差走査熱量測定(DSC)を用い、以下の条件にて測定したDSC曲線における融解ピークの頂点を融点とした。マレイミド樹脂(1)~(6)の示差走査熱量測定(DSC)チャート図を図7~12に示す。
測定装置 :メトラー・トレド株式会社製「DSC1」、
サンプル量:約5mg
温度条件 :10℃/分 <Measurement of melting point>
For the maleimide resins obtained in Examples 1 to 6, differential scanning calorimetry (DSC) was used, and the apex of the melting peak in the DSC curve measured under the following conditions was defined as the melting point. The differential scanning calorimetry (DSC) charts of the maleimide resins (1) to (6) are shown in FIGS. 7 to 12.
Measuring device: "DSC1" manufactured by METTLER TOLEDO Co., Ltd.,
Sample amount: Approximately 5 mg
Temperature condition: 10 ° C / min
実施例1~6で得られたマレイミド樹脂について、軟化点をJIS K7234(環球法)に準拠して測定した。 <Measurement of softening point>
The softening points of the maleimide resins obtained in Examples 1 to 6 were measured according to JIS K7234 (ring ball method).
実施例1~6で得られたマレイミド樹脂を、それぞれ、11cm×5cm(×厚み約1mm)の型に流し込み、200℃で2時間、更に250℃で2時間硬化させて、硬化物を得た。
得られた硬化物について、メトラー・トレド株式会社製 TGA/DSCを用い、Td5を測定した。
測定機器:メトラー・トレド株式会社 TGA/DSC 1
測定範囲:40℃~150℃~600℃
昇温速度:20℃/分(40℃→150℃)
15分保持(150℃)
5℃/分 (150℃→600℃)
雰囲気 :窒素 <Measurement of Td5 of cured product>
The maleimide resins obtained in Examples 1 to 6 were poured into molds of 11 cm × 5 cm (× thickness of about 1 mm) and cured at 200 ° C. for 2 hours and further at 250 ° C. for 2 hours to obtain a cured product. ..
Td5 of the obtained cured product was measured using TGA / DSC manufactured by METTLER TOLEDO CO., LTD.
Measuring equipment: METTLER TOLEDO Co., Ltd. TGA / DSC 1
Measurement range: 40 ° C to 150 ° C to 600 ° C
Temperature rise rate: 20 ° C / min (40 ° C → 150 ° C)
Hold for 15 minutes (150 ° C)
5 ° C / min (150 ° C → 600 ° C)
Atmosphere: Nitrogen
先と同様の条件で硬化物を得、株式会社日立ハイテクサイエンス社製TMA/SS6100を用いて熱膨張率を測定した。
測定機器 :TMA/SS6100(株式会社日立ハイテクサイエンス)
プローブ :石英製膨張・圧縮プローブ
測定荷重 :88.8mN
測定温度 :1st run r.t.~270℃
2nd run 0~270℃
昇温速度 :3℃/分
雰囲気 :窒素 Measurement of the coefficient of thermal expansion of the cured product The cured product was obtained under the same conditions as the above, and the coefficient of thermal expansion was measured using TMA / SS6100 manufactured by Hitachi High-Tech Science Co., Ltd.
Measuring equipment: TMA / SS6100 (Hitachi High-Tech Science Corporation)
Probe: Quartz expansion / compression probe Measurement load: 88.8 mN
Measurement temperature: 1st run r. t. ~ 270 ° C
2nd run 0-270 ° C
Temperature rise rate: 3 ° C / min Atmosphere: Nitrogen
比較対象として4,4’-ジフェニルメタンビスマレイミド(大和化成株式会社製「BMI-1000」)を用い、実施例と同様の方法にて、マレイミド樹脂の融点を測定した。融点は160℃であった。また、当該ビスマレイミド化合物は150℃では溶融しないことから、JIS K7234(環球法)に準拠しての軟化点は測定しなかった。 (Comparative Example 1)
Using 4,4'-diphenylmethanebismaleimide (“BMI-1000” manufactured by Daiwa Kasei Co., Ltd.) as a comparison target, the melting point of the maleimide resin was measured by the same method as in the examples. The melting point was 160 ° C. Moreover, since the bismaleimide compound does not melt at 150 ° C., the softening point was not measured according to JIS K7234 (ring ball method).
Claims (12)
- 複数種の芳香族モノアミン化合物(A)と、結合剤(B)との反応生成物であるポリアミン化合物(C)のマレイミド化物であることを特徴とするマレイミド樹脂。 A maleimide resin characterized by being a maleimide product of a polyamine compound (C) which is a reaction product of a plurality of types of aromatic monoamine compounds (A) and a binder (B).
- 前記結合剤(B)が、アルデヒド化合物(B-1)、ケトン化合物(B-2)、下記一般式(B-3)で表される芳香族化合物(B-3)、下記一般式(B-4)で表される芳香族化合物(B-4)、下記一般式(B-5)で表される芳香族化合物(B-5)、下記一般式(B-6)で表される芳香族化合物(B-6)、下記一般式(B-7)で表される芳香族化合物(B-7)、下記一般式(B-8)で表される芳香族化合物(B-8)のいずれか一種以上である請求項1記載のマレイミド樹脂。
- 異なる2種の芳香族モノアミン化合物(A)が結合剤(B)で結合された非対称ジアミン化合物(C-1)のマレイミド化物を含有する請求項1又は2記載のマレイミド樹脂。 The maleimide resin according to claim 1 or 2, which contains a maleimided product of an asymmetric diamine compound (C-1) in which two different aromatic monoamine compounds (A) are bound with a binder (B).
- 異なる2種の芳香族モノアミン化合物(A)が結合剤(B)で結合された非対称ジアミン化合物(C-1)のマレイミド化物である非対称ビスマレイミド化合物。 An asymmetric bismaleimide compound which is a maleimided product of an asymmetric diamine compound (C-1) in which two different aromatic monoamine compounds (A) are bound with a binder (B).
- 下記一般式(4)で表されることを特徴とする請求項4記載の非対称ビスマレイミド化合物。
- 請求項1~3のいずれか一つに記載のマレイミド樹脂、或いは請求項4又は5記載の非対称ビスマレイミド化合物を含有する硬化性組成物。 A curable composition containing the maleimide resin according to any one of claims 1 to 3 or the asymmetrical bismaleimide compound according to claim 4 or 5.
- 請求項6記載の硬化性組成物の硬化物。 A cured product of the curable composition according to claim 6.
- 請求項6記載の硬化性組成物を用いた半導体封止材料。 A semiconductor encapsulation material using the curable composition according to claim 6.
- 請求項8記載の半導体封止材料を用いた半導体装置。 A semiconductor device using the semiconductor encapsulating material according to claim 8.
- 請求項6記載の硬化性組成物を用いたプリプレグ。 A prepreg using the curable composition according to claim 6.
- 請求項10記載のプリプレグを用いた回路基板。 A circuit board using the prepreg according to claim 10.
- 請求項6記載の硬化性組成物を用いたビルドアップフィルム。 A build-up film using the curable composition according to claim 6.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020237022081A KR20230113611A (en) | 2020-12-22 | 2021-11-18 | Maleimide resin, asymmetric bismaleimide compound, curable composition, cured product, semiconductor encapsulation material, semiconductor encapsulation device, prepreg, circuit board, and build-up film |
| JP2022536593A JP7140307B1 (en) | 2020-12-22 | 2021-11-18 | Maleimide resin, asymmetric bismaleimide compound, curable composition, cured product, semiconductor sealing material, semiconductor sealing device, prepreg, circuit board, and build-up film |
| CN202180086105.1A CN116888098A (en) | 2020-12-22 | 2021-11-18 | Maleimide resin, asymmetric bismaleimide compound, curable composition, cured product, semiconductor sealing material, semiconductor sealing device, prepreg, circuit board, and laminated film |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2020212256 | 2020-12-22 | ||
| JP2020-212256 | 2020-12-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022137913A1 true WO2022137913A1 (en) | 2022-06-30 |
Family
ID=82159361
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2021/042356 WO2022137913A1 (en) | 2020-12-22 | 2021-11-18 | Maleimide resin, asymmetric bismaleimide compound, curable composition, cured object, semiconductor-encapsulating material, semiconductor-encapsulating device, prepreg, circuit board, and build-up film |
Country Status (5)
| Country | Link |
|---|---|
| JP (1) | JP7140307B1 (en) |
| KR (1) | KR20230113611A (en) |
| CN (1) | CN116888098A (en) |
| TW (1) | TW202231691A (en) |
| WO (1) | WO2022137913A1 (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5130290A (en) * | 1974-07-02 | 1976-03-15 | Ciba Geigy | |
| JPS52125161A (en) * | 1976-04-09 | 1977-10-20 | Ciba Geigy Ag | Preparation of maleimide |
| JPH02300223A (en) * | 1989-05-16 | 1990-12-12 | Mitsubishi Petrochem Co Ltd | Polyimide |
| JPH0431464A (en) * | 1990-05-25 | 1992-02-03 | Mitsubishi Petrochem Co Ltd | Curing resin composition for circuit and metal foil-clad resin board |
| KR20150002953A (en) * | 2013-06-27 | 2015-01-08 | 제일모직주식회사 | Hardmask composition, method of forming patterns using the hardmask composition and semiconductor integrated circuit device including the patterns including the patterns |
| WO2020217679A1 (en) * | 2019-04-26 | 2020-10-29 | Dic株式会社 | Maleimide, curable resin composition, and cured product |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0385937A1 (en) | 1989-02-28 | 1990-09-05 | Ciba-Geigy Ag | Curable blends based on aromatic bismaleinimides |
| JPH06128225A (en) | 1992-10-19 | 1994-05-10 | Mitsubishi Petrochem Co Ltd | Production of amorphous bismaleimide |
-
2021
- 2021-11-18 WO PCT/JP2021/042356 patent/WO2022137913A1/en active Application Filing
- 2021-11-18 CN CN202180086105.1A patent/CN116888098A/en active Pending
- 2021-11-18 KR KR1020237022081A patent/KR20230113611A/en unknown
- 2021-11-18 TW TW110142894A patent/TW202231691A/en unknown
- 2021-11-18 JP JP2022536593A patent/JP7140307B1/en active Active
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5130290A (en) * | 1974-07-02 | 1976-03-15 | Ciba Geigy | |
| JPS52125161A (en) * | 1976-04-09 | 1977-10-20 | Ciba Geigy Ag | Preparation of maleimide |
| JPH02300223A (en) * | 1989-05-16 | 1990-12-12 | Mitsubishi Petrochem Co Ltd | Polyimide |
| JPH0431464A (en) * | 1990-05-25 | 1992-02-03 | Mitsubishi Petrochem Co Ltd | Curing resin composition for circuit and metal foil-clad resin board |
| KR20150002953A (en) * | 2013-06-27 | 2015-01-08 | 제일모직주식회사 | Hardmask composition, method of forming patterns using the hardmask composition and semiconductor integrated circuit device including the patterns including the patterns |
| WO2020217679A1 (en) * | 2019-04-26 | 2020-10-29 | Dic株式会社 | Maleimide, curable resin composition, and cured product |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202231691A (en) | 2022-08-16 |
| CN116888098A (en) | 2023-10-13 |
| JPWO2022137913A1 (en) | 2022-06-30 |
| JP7140307B1 (en) | 2022-09-21 |
| KR20230113611A (en) | 2023-07-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP3464783B2 (en) | Phosphorus-containing curing agent and flame-retardant cured epoxy resin using the same | |
| JP5664817B2 (en) | Phenolic hydroxyl group-containing compound, phenol resin, curable composition, cured product thereof, semiconductor sealing material, and printed wiring board | |
| JP7068669B2 (en) | Active ester compound | |
| WO2018008411A1 (en) | Active ester resin composition and cured product of same | |
| KR102268344B1 (en) | Active ester composition and cured product thereof | |
| JP6332720B1 (en) | Active ester resin and its cured product | |
| JP6862701B2 (en) | Active ester resin and its cured product | |
| KR20190025607A (en) | Curable composition and its cured product | |
| JP5682805B1 (en) | Phenolic hydroxyl group-containing compound, phenol resin, curable composition, cured product thereof, semiconductor sealing material, and printed wiring board | |
| WO2022137913A1 (en) | Maleimide resin, asymmetric bismaleimide compound, curable composition, cured object, semiconductor-encapsulating material, semiconductor-encapsulating device, prepreg, circuit board, and build-up film | |
| WO2018008409A1 (en) | Active ester resin and cured product thereof | |
| KR101184292B1 (en) | Novel epoxy compounds and their flame-retarding epoxy resin compositions | |
| JP6863067B2 (en) | δ-Valerolactone skeleton-containing resin | |
| JP5682804B1 (en) | Phenolic hydroxyl group-containing compound, phenol resin, curable composition, cured product thereof, semiconductor sealing material, and printed wiring board | |
| WO2017145772A1 (en) | Epoxy resin, curable resin composition, and cured product thereof | |
| JP7465926B2 (en) | Epoxy resin, its manufacturing method, epoxy composition containing same, and uses thereof | |
| JP7276665B2 (en) | Active ester composition and semiconductor encapsulation material | |
| JP6332721B1 (en) | Active ester resin and its cured product | |
| JP2018193470A (en) | δ- VALEROLACTONE SKELETON-CONTAINING RESIN |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2022536593 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21910066 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180086105.1 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 20237022081 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 21910066 Country of ref document: EP Kind code of ref document: A1 |