WO2019094548A1 - Compositions and methods for inhibiting viral vector-induced inflammatory responses - Google Patents
Compositions and methods for inhibiting viral vector-induced inflammatory responses Download PDFInfo
- Publication number
- WO2019094548A1 WO2019094548A1 PCT/US2018/059756 US2018059756W WO2019094548A1 WO 2019094548 A1 WO2019094548 A1 WO 2019094548A1 US 2018059756 W US2018059756 W US 2018059756W WO 2019094548 A1 WO2019094548 A1 WO 2019094548A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- viral genome
- seq
- recombinant viral
- inhibitory
- nucleotide sequence
- Prior art date
Links
- 230000002401 inhibitory effect Effects 0.000 title claims abstract description 262
- 238000000034 method Methods 0.000 title claims description 106
- 230000028709 inflammatory response Effects 0.000 title claims description 49
- 239000000203 mixture Substances 0.000 title description 14
- 239000013603 viral vector Substances 0.000 title description 13
- 108091034117 Oligonucleotide Proteins 0.000 claims abstract description 300
- 230000003612 virological effect Effects 0.000 claims abstract description 277
- 239000002773 nucleotide Substances 0.000 claims description 150
- 125000003729 nucleotide group Chemical group 0.000 claims description 150
- 230000001225 therapeutic effect Effects 0.000 claims description 117
- 108091035539 telomere Proteins 0.000 claims description 82
- 102000055501 telomere Human genes 0.000 claims description 82
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 claims description 78
- 230000014509 gene expression Effects 0.000 claims description 69
- 150000007523 nucleic acids Chemical class 0.000 claims description 63
- 102000002689 Toll-like receptor Human genes 0.000 claims description 58
- 108020000411 Toll-like receptor Proteins 0.000 claims description 58
- 102000039446 nucleic acids Human genes 0.000 claims description 56
- 108020004707 nucleic acids Proteins 0.000 claims description 56
- 102000008235 Toll-Like Receptor 9 Human genes 0.000 claims description 50
- 108010060818 Toll-Like Receptor 9 Proteins 0.000 claims description 50
- 108090000623 proteins and genes Proteins 0.000 claims description 36
- 230000000692 anti-sense effect Effects 0.000 claims description 34
- 230000011664 signaling Effects 0.000 claims description 31
- 230000004913 activation Effects 0.000 claims description 28
- 230000002757 inflammatory effect Effects 0.000 claims description 27
- 108020004414 DNA Proteins 0.000 claims description 24
- 210000003205 muscle Anatomy 0.000 claims description 19
- 238000011144 upstream manufacturing Methods 0.000 claims description 18
- 241000702421 Dependoparvovirus Species 0.000 claims description 13
- 108091028043 Nucleic acid sequence Proteins 0.000 claims description 12
- 102000004169 proteins and genes Human genes 0.000 claims description 12
- 108091032973 (ribonucleotides)n+m Proteins 0.000 claims description 11
- 241000701022 Cytomegalovirus Species 0.000 claims description 10
- 230000000295 complement effect Effects 0.000 claims description 10
- 239000003112 inhibitor Substances 0.000 claims description 9
- 230000001404 mediated effect Effects 0.000 claims description 9
- 229940046168 CpG oligodeoxynucleotide Drugs 0.000 claims description 8
- 241000701161 unidentified adenovirus Species 0.000 claims description 7
- 208000007514 Herpes zoster Diseases 0.000 claims description 6
- 241000700584 Simplexvirus Species 0.000 claims description 6
- 230000003213 activating effect Effects 0.000 claims description 6
- 241000701041 Human betaherpesvirus 7 Species 0.000 claims description 5
- 241000701044 Human gammaherpesvirus 4 Species 0.000 claims description 5
- 241001502974 Human gammaherpesvirus 8 Species 0.000 claims description 5
- 241000702617 Human parvovirus B19 Species 0.000 claims description 5
- 241000829111 Human polyomavirus 1 Species 0.000 claims description 5
- 241000701460 JC polyomavirus Species 0.000 claims description 5
- 108091026898 Leader sequence (mRNA) Proteins 0.000 claims description 5
- 241000700627 Monkeypox virus Species 0.000 claims description 5
- 241000700647 Variola virus Species 0.000 claims description 5
- 208000002672 hepatitis B Diseases 0.000 claims description 5
- 108020005345 3' Untranslated Regions Proteins 0.000 claims description 4
- 208000026350 Inborn Genetic disease Diseases 0.000 claims description 4
- 208000016361 genetic disease Diseases 0.000 claims description 4
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 4
- 108091023045 Untranslated Region Proteins 0.000 claims description 3
- 206010061218 Inflammation Diseases 0.000 abstract description 40
- 230000004054 inflammatory process Effects 0.000 abstract description 40
- 238000001415 gene therapy Methods 0.000 abstract description 12
- 210000001508 eye Anatomy 0.000 description 78
- 239000013598 vector Substances 0.000 description 74
- 210000003411 telomere Anatomy 0.000 description 58
- 229940046166 oligodeoxynucleotide Drugs 0.000 description 57
- 210000004027 cell Anatomy 0.000 description 52
- 241001465754 Metazoa Species 0.000 description 44
- 238000002347 injection Methods 0.000 description 34
- 239000007924 injection Substances 0.000 description 34
- 241000699670 Mus sp. Species 0.000 description 31
- 102000004127 Cytokines Human genes 0.000 description 28
- 108090000695 Cytokines Proteins 0.000 description 28
- 230000006378 damage Effects 0.000 description 28
- 239000013607 AAV vector Substances 0.000 description 26
- 239000005090 green fluorescent protein Substances 0.000 description 26
- 210000004185 liver Anatomy 0.000 description 24
- 210000001525 retina Anatomy 0.000 description 24
- 238000001727 in vivo Methods 0.000 description 22
- 238000012014 optical coherence tomography Methods 0.000 description 22
- 230000000770 proinflammatory effect Effects 0.000 description 19
- 239000003981 vehicle Substances 0.000 description 18
- 210000000234 capsid Anatomy 0.000 description 17
- 239000002245 particle Substances 0.000 description 17
- 238000004519 manufacturing process Methods 0.000 description 16
- KDWFDOFTPHDNJL-TUBOTVQJSA-N odn-2006 Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=S)O[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)O[C@@H]2[C@H](O[C@H](C2)N2C(NC(=O)C(C)=C2)=O)COP(O)(=O)O[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=S)O[C@H]2[C@H]([C@@H](O[C@@H]2COP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H]([C@@H](O)[C@@H](O2)N2C3=C(C(NC(N)=N3)=O)N=C2)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H]([C@@H](O)[C@@H](O2)N2C3=C(C(NC(N)=N3)=O)N=C2)OP(S)(=O)OC[C@@H]2[C@H]([C@@H](O)[C@@H](O2)N2C(N=C(N)C=C2)=O)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)OP(O)(=O)OC[C@@H]2[C@H](C[C@@H](O2)N2C(NC(=O)C(C)=C2)=O)O)N2C3=C(C(NC(N)=N3)=O)N=C2)O)N2C(N=C(N)C=C2)=O)O)N2C3=C(C(NC(N)=N3)=O)N=C2)O)N2C3=C(C(NC(N)=N3)=O)N=C2)O)N2C(N=C(N)C=C2)=O)O)[C@@H](O)C1 KDWFDOFTPHDNJL-TUBOTVQJSA-N 0.000 description 16
- 241001164825 Adeno-associated virus - 8 Species 0.000 description 15
- 230000009528 severe injury Effects 0.000 description 15
- 238000010186 staining Methods 0.000 description 15
- 108700019146 Transgenes Proteins 0.000 description 14
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 13
- 230000000694 effects Effects 0.000 description 13
- 230000002207 retinal effect Effects 0.000 description 13
- 239000011780 sodium chloride Substances 0.000 description 13
- 102100022641 Coagulation factor IX Human genes 0.000 description 12
- 108010076282 Factor IX Proteins 0.000 description 12
- 241000282887 Suidae Species 0.000 description 12
- 108091036066 Three prime untranslated region Proteins 0.000 description 12
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 11
- 241000702423 Adeno-associated virus - 2 Species 0.000 description 11
- 230000028993 immune response Effects 0.000 description 11
- 230000007170 pathology Effects 0.000 description 11
- 210000001519 tissue Anatomy 0.000 description 11
- 108020003589 5' Untranslated Regions Proteins 0.000 description 10
- 238000011740 C57BL/6 mouse Methods 0.000 description 10
- 229960004222 factor ix Drugs 0.000 description 10
- 230000008595 infiltration Effects 0.000 description 10
- 238000001764 infiltration Methods 0.000 description 10
- 230000002829 reductive effect Effects 0.000 description 10
- 238000012360 testing method Methods 0.000 description 10
- 210000001744 T-lymphocyte Anatomy 0.000 description 9
- 241000700605 Viruses Species 0.000 description 9
- 238000003780 insertion Methods 0.000 description 9
- 230000037431 insertion Effects 0.000 description 9
- 108091008695 photoreceptors Proteins 0.000 description 9
- 241000282414 Homo sapiens Species 0.000 description 8
- 241000699666 Mus <mouse, genus> Species 0.000 description 8
- 230000005867 T cell response Effects 0.000 description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 8
- 229960000027 human factor ix Drugs 0.000 description 8
- 230000015788 innate immune response Effects 0.000 description 8
- 238000010255 intramuscular injection Methods 0.000 description 8
- 239000007927 intramuscular injection Substances 0.000 description 8
- 230000000670 limiting effect Effects 0.000 description 8
- 238000003753 real-time PCR Methods 0.000 description 8
- 230000002441 reversible effect Effects 0.000 description 8
- 108010071690 Prealbumin Proteins 0.000 description 7
- 238000011529 RT qPCR Methods 0.000 description 7
- 102000009190 Transthyretin Human genes 0.000 description 7
- 201000010099 disease Diseases 0.000 description 7
- 230000006698 induction Effects 0.000 description 7
- 230000001939 inductive effect Effects 0.000 description 7
- 210000005228 liver tissue Anatomy 0.000 description 7
- 239000000243 solution Substances 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 102000004889 Interleukin-6 Human genes 0.000 description 6
- 108090001005 Interleukin-6 Proteins 0.000 description 6
- 206010038848 Retinal detachment Diseases 0.000 description 6
- 108010048367 enhanced green fluorescent protein Proteins 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- 239000000463 material Substances 0.000 description 6
- 210000000274 microglia Anatomy 0.000 description 6
- 230000004264 retinal detachment Effects 0.000 description 6
- 239000000523 sample Substances 0.000 description 6
- 210000003462 vein Anatomy 0.000 description 6
- 102000053602 DNA Human genes 0.000 description 5
- 101000669402 Homo sapiens Toll-like receptor 7 Proteins 0.000 description 5
- 108010050904 Interferons Proteins 0.000 description 5
- 102000014150 Interferons Human genes 0.000 description 5
- 108091081062 Repeated sequence (DNA) Proteins 0.000 description 5
- 108020004682 Single-Stranded DNA Proteins 0.000 description 5
- 206010047663 Vitritis Diseases 0.000 description 5
- 238000004422 calculation algorithm Methods 0.000 description 5
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 5
- 238000001514 detection method Methods 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 239000002158 endotoxin Substances 0.000 description 5
- 208000013653 hyalitis Diseases 0.000 description 5
- 210000002865 immune cell Anatomy 0.000 description 5
- 238000001990 intravenous administration Methods 0.000 description 5
- 238000003475 lamination Methods 0.000 description 5
- 238000004806 packaging method and process Methods 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 230000004044 response Effects 0.000 description 5
- 210000000964 retinal cone photoreceptor cell Anatomy 0.000 description 5
- 239000004055 small Interfering RNA Substances 0.000 description 5
- 238000001356 surgical procedure Methods 0.000 description 5
- 238000010361 transduction Methods 0.000 description 5
- 230000026683 transduction Effects 0.000 description 5
- 102100025248 C-X-C motif chemokine 10 Human genes 0.000 description 4
- 108091008102 DNA aptamers Proteins 0.000 description 4
- 101000858088 Homo sapiens C-X-C motif chemokine 10 Proteins 0.000 description 4
- 101000831567 Homo sapiens Toll-like receptor 2 Proteins 0.000 description 4
- 208000017442 Retinal disease Diseases 0.000 description 4
- 102100024333 Toll-like receptor 2 Human genes 0.000 description 4
- 102100039390 Toll-like receptor 7 Human genes 0.000 description 4
- 102100040247 Tumor necrosis factor Human genes 0.000 description 4
- 238000002835 absorbance Methods 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 108010006025 bovine growth hormone Proteins 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- 231100000673 dose–response relationship Toxicity 0.000 description 4
- 238000003114 enzyme-linked immunosorbent spot assay Methods 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 238000003384 imaging method Methods 0.000 description 4
- 238000000338 in vitro Methods 0.000 description 4
- 229940079322 interferon Drugs 0.000 description 4
- 238000007918 intramuscular administration Methods 0.000 description 4
- 238000005259 measurement Methods 0.000 description 4
- 239000002105 nanoparticle Substances 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 230000019491 signal transduction Effects 0.000 description 4
- 210000004988 splenocyte Anatomy 0.000 description 4
- 239000006228 supernatant Substances 0.000 description 4
- 238000012546 transfer Methods 0.000 description 4
- 206010002091 Anaesthesia Diseases 0.000 description 3
- 108050003620 Arrestin-C Proteins 0.000 description 3
- 102100026440 Arrestin-C Human genes 0.000 description 3
- 108091033409 CRISPR Proteins 0.000 description 3
- 241000283707 Capra Species 0.000 description 3
- 241000282552 Chlorocebus aethiops Species 0.000 description 3
- 238000002965 ELISA Methods 0.000 description 3
- 238000011510 Elispot assay Methods 0.000 description 3
- 241000283074 Equus asinus Species 0.000 description 3
- 101150066002 GFP gene Proteins 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 102000001398 Granzyme Human genes 0.000 description 3
- 108060005986 Granzyme Proteins 0.000 description 3
- 108010074328 Interferon-gamma Proteins 0.000 description 3
- 238000011050 LAL assay Methods 0.000 description 3
- 206010025421 Macule Diseases 0.000 description 3
- 102000003945 NF-kappa B Human genes 0.000 description 3
- 108010057466 NF-kappa B Proteins 0.000 description 3
- 108020004459 Small interfering RNA Proteins 0.000 description 3
- 208000004350 Strabismus Diseases 0.000 description 3
- 229930006000 Sucrose Natural products 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 206010046851 Uveitis Diseases 0.000 description 3
- 230000037005 anaesthesia Effects 0.000 description 3
- 238000000137 annealing Methods 0.000 description 3
- 210000001742 aqueous humor Anatomy 0.000 description 3
- 210000004369 blood Anatomy 0.000 description 3
- 239000008280 blood Substances 0.000 description 3
- 201000004709 chorioretinitis Diseases 0.000 description 3
- 210000003161 choroid Anatomy 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 210000004087 cornea Anatomy 0.000 description 3
- 230000016396 cytokine production Effects 0.000 description 3
- 230000007423 decrease Effects 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- 238000001476 gene delivery Methods 0.000 description 3
- 230000009368 gene silencing by RNA Effects 0.000 description 3
- 230000036541 health Effects 0.000 description 3
- 238000003364 immunohistochemistry Methods 0.000 description 3
- 239000002054 inoculum Substances 0.000 description 3
- 238000010253 intravenous injection Methods 0.000 description 3
- 239000003446 ligand Substances 0.000 description 3
- 108020004999 messenger RNA Proteins 0.000 description 3
- 206010030875 ophthalmoplegia Diseases 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- 230000001105 regulatory effect Effects 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 210000000952 spleen Anatomy 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 239000005720 sucrose Substances 0.000 description 3
- RYYWUUFWQRZTIU-UHFFFAOYSA-K thiophosphate Chemical group [O-]P([O-])([O-])=S RYYWUUFWQRZTIU-UHFFFAOYSA-K 0.000 description 3
- 238000013518 transcription Methods 0.000 description 3
- 230000035897 transcription Effects 0.000 description 3
- 230000003442 weekly effect Effects 0.000 description 3
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 2
- FHJATBIERQTCTN-UHFFFAOYSA-N 1-[4-amino-2-(ethylaminomethyl)imidazo[4,5-c]quinolin-1-yl]-2-methylpropan-2-ol Chemical compound C1=CC=CC2=C(N(C(CNCC)=N3)CC(C)(C)O)C3=C(N)N=C21 FHJATBIERQTCTN-UHFFFAOYSA-N 0.000 description 2
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 2
- 108700028369 Alleles Proteins 0.000 description 2
- 201000002862 Angle-Closure Glaucoma Diseases 0.000 description 2
- 206010053781 Anterior chamber cell Diseases 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- 208000002177 Cataract Diseases 0.000 description 2
- 108010012236 Chemokines Proteins 0.000 description 2
- 102000019034 Chemokines Human genes 0.000 description 2
- 208000033825 Chorioretinal atrophy Diseases 0.000 description 2
- 208000033810 Choroidal dystrophy Diseases 0.000 description 2
- 208000006992 Color Vision Defects Diseases 0.000 description 2
- 206010012689 Diabetic retinopathy Diseases 0.000 description 2
- 206010013801 Duchenne Muscular Dystrophy Diseases 0.000 description 2
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 2
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 108020005004 Guide RNA Proteins 0.000 description 2
- 101000800479 Homo sapiens Toll-like receptor 9 Proteins 0.000 description 2
- 230000005353 IP-10 production Effects 0.000 description 2
- 102100026720 Interferon beta Human genes 0.000 description 2
- 102100037850 Interferon gamma Human genes 0.000 description 2
- 108090000467 Interferon-beta Proteins 0.000 description 2
- 206010048804 Kearns-Sayre syndrome Diseases 0.000 description 2
- 108700011259 MicroRNAs Proteins 0.000 description 2
- 208000029578 Muscle disease Diseases 0.000 description 2
- 101710163270 Nuclease Proteins 0.000 description 2
- 208000022873 Ocular disease Diseases 0.000 description 2
- 241000243985 Onchocerca volvulus Species 0.000 description 2
- 206010030348 Open-Angle Glaucoma Diseases 0.000 description 2
- 108050001704 Opsin Proteins 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- 208000004788 Pars Planitis Diseases 0.000 description 2
- QGMRQYFBGABWDR-UHFFFAOYSA-M Pentobarbital sodium Chemical compound [Na+].CCCC(C)C1(CC)C(=O)NC(=O)[N-]C1=O QGMRQYFBGABWDR-UHFFFAOYSA-M 0.000 description 2
- 108700008625 Reporter Genes Proteins 0.000 description 2
- 108091027568 Single-stranded nucleotide Proteins 0.000 description 2
- 108091027967 Small hairpin RNA Proteins 0.000 description 2
- RYYWUUFWQRZTIU-UHFFFAOYSA-N Thiophosphoric acid Chemical group OP(O)(S)=O RYYWUUFWQRZTIU-UHFFFAOYSA-N 0.000 description 2
- 108010018242 Transcription Factor AP-1 Proteins 0.000 description 2
- 102100023118 Transcription factor JunD Human genes 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 241001492404 Woodchuck hepatitis virus Species 0.000 description 2
- 230000004308 accommodation Effects 0.000 description 2
- 150000001413 amino acids Chemical group 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 210000002159 anterior chamber Anatomy 0.000 description 2
- 206010003246 arthritis Diseases 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 210000004204 blood vessel Anatomy 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 210000003169 central nervous system Anatomy 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 201000007254 color blindness Diseases 0.000 description 2
- 238000004590 computer program Methods 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 239000008121 dextrose Substances 0.000 description 2
- 238000007847 digital PCR Methods 0.000 description 2
- 238000006471 dimerization reaction Methods 0.000 description 2
- -1 e.g. Proteins 0.000 description 2
- 239000012636 effector Substances 0.000 description 2
- 210000000744 eyelid Anatomy 0.000 description 2
- 230000004438 eyesight Effects 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 230000004927 fusion Effects 0.000 description 2
- 229940124670 gardiquimod Drugs 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- ACGUYXCXAPNIKK-UHFFFAOYSA-N hexachlorophene Chemical compound OC1=C(Cl)C=C(Cl)C(Cl)=C1CC1=C(O)C(Cl)=CC(Cl)=C1Cl ACGUYXCXAPNIKK-UHFFFAOYSA-N 0.000 description 2
- 230000006801 homologous recombination Effects 0.000 description 2
- 238000002744 homologous recombination Methods 0.000 description 2
- 102000045710 human TLR9 Human genes 0.000 description 2
- 210000000987 immune system Anatomy 0.000 description 2
- 230000002055 immunohistochemical effect Effects 0.000 description 2
- 238000010348 incorporation Methods 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 208000017532 inherited retinal dystrophy Diseases 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 230000010468 interferon response Effects 0.000 description 2
- 230000017306 interleukin-6 production Effects 0.000 description 2
- 210000005229 liver cell Anatomy 0.000 description 2
- 208000002780 macular degeneration Diseases 0.000 description 2
- 239000002679 microRNA Substances 0.000 description 2
- 239000003094 microcapsule Substances 0.000 description 2
- 210000000663 muscle cell Anatomy 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 239000002088 nanocapsule Substances 0.000 description 2
- 239000002077 nanosphere Substances 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- 230000001124 posttranscriptional effect Effects 0.000 description 2
- 238000004321 preservation Methods 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 210000001747 pupil Anatomy 0.000 description 2
- 230000009257 reactivity Effects 0.000 description 2
- 230000004256 retinal image Effects 0.000 description 2
- 231100000241 scar Toxicity 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 238000004904 shortening Methods 0.000 description 2
- 150000003431 steroids Chemical class 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- 238000001890 transfection Methods 0.000 description 2
- 201000008827 tuberculosis Diseases 0.000 description 2
- 102000003390 tumor necrosis factor Human genes 0.000 description 2
- 230000006433 tumor necrosis factor production Effects 0.000 description 2
- 238000011870 unpaired t-test Methods 0.000 description 2
- 229960005486 vaccine Drugs 0.000 description 2
- 230000007502 viral entry Effects 0.000 description 2
- 210000000605 viral structure Anatomy 0.000 description 2
- 238000001262 western blot Methods 0.000 description 2
- HSTOKWSFWGCZMH-UHFFFAOYSA-N 3,3'-diaminobenzidine Chemical compound C1=C(N)C(N)=CC=C1C1=CC=C(N)C(N)=C1 HSTOKWSFWGCZMH-UHFFFAOYSA-N 0.000 description 1
- 239000012103 Alexa Fluor 488 Substances 0.000 description 1
- 239000012110 Alexa Fluor 594 Substances 0.000 description 1
- 239000012114 Alexa Fluor 647 Substances 0.000 description 1
- 102100022524 Alpha-1-antichymotrypsin Human genes 0.000 description 1
- 201000009487 Amblyopia Diseases 0.000 description 1
- 208000005598 Angioid Streaks Diseases 0.000 description 1
- 108091023037 Aptamer Proteins 0.000 description 1
- 201000007978 Argyll Robertson pupil Diseases 0.000 description 1
- 208000002109 Argyria Diseases 0.000 description 1
- 108090000328 Arrestin Proteins 0.000 description 1
- 102000003916 Arrestin Human genes 0.000 description 1
- 206010003694 Atrophy Diseases 0.000 description 1
- 229930003347 Atropine Natural products 0.000 description 1
- 201000005943 Barth syndrome Diseases 0.000 description 1
- 201000006935 Becker muscular dystrophy Diseases 0.000 description 1
- 208000020941 Benign concentric annular macular dystrophy Diseases 0.000 description 1
- 206010061692 Benign muscle neoplasm Diseases 0.000 description 1
- 206010005152 Blepharochalasis Diseases 0.000 description 1
- 201000004569 Blindness Diseases 0.000 description 1
- 102100021943 C-C motif chemokine 2 Human genes 0.000 description 1
- 102100032367 C-C motif chemokine 5 Human genes 0.000 description 1
- 210000001266 CD8-positive T-lymphocyte Anatomy 0.000 description 1
- 238000010354 CRISPR gene editing Methods 0.000 description 1
- 102000005701 Calcium-Binding Proteins Human genes 0.000 description 1
- 108010045403 Calcium-Binding Proteins Proteins 0.000 description 1
- 102000016289 Cell Adhesion Molecules Human genes 0.000 description 1
- 108010067225 Cell Adhesion Molecules Proteins 0.000 description 1
- 208000016615 Central areolar choroidal dystrophy Diseases 0.000 description 1
- 208000003569 Central serous chorioretinopathy Diseases 0.000 description 1
- 201000005072 Chorioretinal scar Diseases 0.000 description 1
- 208000033379 Chorioretinopathy Diseases 0.000 description 1
- 208000024304 Choroidal Effusions Diseases 0.000 description 1
- 206010008783 Choroidal detachment Diseases 0.000 description 1
- 206010008786 Choroidal haemorrhage Diseases 0.000 description 1
- 208000008515 Choroidal sclerosis Diseases 0.000 description 1
- 208000002691 Choroiditis Diseases 0.000 description 1
- 208000031404 Chromosome Aberrations Diseases 0.000 description 1
- 208000032544 Cicatrix Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 208000021089 Coats disease Diseases 0.000 description 1
- 108091026890 Coding region Proteins 0.000 description 1
- 201000003101 Coloboma Diseases 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 108010003730 Cone Opsins Proteins 0.000 description 1
- 208000004117 Congenital Myasthenic Syndromes Diseases 0.000 description 1
- 206010010741 Conjunctivitis Diseases 0.000 description 1
- 206010010984 Corneal abrasion Diseases 0.000 description 1
- 208000028006 Corneal injury Diseases 0.000 description 1
- 206010055665 Corneal neovascularisation Diseases 0.000 description 1
- 230000004543 DNA replication Effects 0.000 description 1
- 102000004163 DNA-directed RNA polymerases Human genes 0.000 description 1
- 108090000626 DNA-directed RNA polymerases Proteins 0.000 description 1
- 206010011841 Dacryoadenitis acquired Diseases 0.000 description 1
- 101100447432 Danio rerio gapdh-2 gene Proteins 0.000 description 1
- 241001128004 Demodex Species 0.000 description 1
- 201000004624 Dermatitis Diseases 0.000 description 1
- 208000003556 Dry Eye Syndromes Diseases 0.000 description 1
- 206010013774 Dry eye Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 238000008157 ELISA kit Methods 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 241000909851 Epiphora Species 0.000 description 1
- 208000001351 Epiretinal Membrane Diseases 0.000 description 1
- 208000001692 Esotropia Diseases 0.000 description 1
- 108700039887 Essential Genes Proteins 0.000 description 1
- 108091029865 Exogenous DNA Proteins 0.000 description 1
- 201000005538 Exotropia Diseases 0.000 description 1
- 206010015901 Exudative retinopathy Diseases 0.000 description 1
- 206010015995 Eyelid ptosis Diseases 0.000 description 1
- 208000037149 Facioscapulohumeral dystrophy Diseases 0.000 description 1
- 108010073385 Fibrin Proteins 0.000 description 1
- 102000009123 Fibrin Human genes 0.000 description 1
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 1
- 208000001640 Fibromyalgia Diseases 0.000 description 1
- 201000006353 Filariasis Diseases 0.000 description 1
- 208000014260 Fungal keratitis Diseases 0.000 description 1
- 101150112014 Gapdh gene Proteins 0.000 description 1
- 208000034826 Genetic Predisposition to Disease Diseases 0.000 description 1
- 208000010412 Glaucoma Diseases 0.000 description 1
- 208000007698 Gyrate Atrophy Diseases 0.000 description 1
- 108091005904 Hemoglobin subunit beta Proteins 0.000 description 1
- 102100021519 Hemoglobin subunit beta Human genes 0.000 description 1
- 208000032843 Hemorrhage Diseases 0.000 description 1
- 208000032087 Hereditary Leber Optic Atrophy Diseases 0.000 description 1
- 206010019899 Hereditary retinal dystrophy Diseases 0.000 description 1
- 208000009889 Herpes Simplex Diseases 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 101000678026 Homo sapiens Alpha-1-antichymotrypsin Proteins 0.000 description 1
- 101000785755 Homo sapiens Arrestin-C Proteins 0.000 description 1
- 101000897480 Homo sapiens C-C motif chemokine 2 Proteins 0.000 description 1
- 101000797762 Homo sapiens C-C motif chemokine 5 Proteins 0.000 description 1
- 101000899111 Homo sapiens Hemoglobin subunit beta Proteins 0.000 description 1
- 101000959820 Homo sapiens Interferon alpha-1/13 Proteins 0.000 description 1
- 101000729271 Homo sapiens Retinoid isomerohydrolase Proteins 0.000 description 1
- 101000831496 Homo sapiens Toll-like receptor 3 Proteins 0.000 description 1
- 101000800483 Homo sapiens Toll-like receptor 8 Proteins 0.000 description 1
- 101000611183 Homo sapiens Tumor necrosis factor Proteins 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- RKUNBYITZUJHSG-UHFFFAOYSA-N Hyosciamin-hydrochlorid Natural products CN1C(C2)CCC1CC2OC(=O)C(CO)C1=CC=CC=C1 RKUNBYITZUJHSG-UHFFFAOYSA-N 0.000 description 1
- 206010020565 Hyperaemia Diseases 0.000 description 1
- 206010020675 Hypermetropia Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010021531 Impetigo Diseases 0.000 description 1
- 208000032578 Inherited retinal disease Diseases 0.000 description 1
- 102000002227 Interferon Type I Human genes 0.000 description 1
- 108010014726 Interferon Type I Proteins 0.000 description 1
- 102100040019 Interferon alpha-1/13 Human genes 0.000 description 1
- 102000008070 Interferon-gamma Human genes 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- PIWKPBJCKXDKJR-UHFFFAOYSA-N Isoflurane Chemical compound FC(F)OC(Cl)C(F)(F)F PIWKPBJCKXDKJR-UHFFFAOYSA-N 0.000 description 1
- 206010062353 Keratitis fungal Diseases 0.000 description 1
- 208000009319 Keratoconjunctivitis Sicca Diseases 0.000 description 1
- 201000002287 Keratoconus Diseases 0.000 description 1
- YQEZLKZALYSWHR-UHFFFAOYSA-N Ketamine Chemical compound C=1C=CC=C(Cl)C=1C1(NC)CCCCC1=O YQEZLKZALYSWHR-UHFFFAOYSA-N 0.000 description 1
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 1
- 201000010743 Lambert-Eaton myasthenic syndrome Diseases 0.000 description 1
- 201000003533 Leber congenital amaurosis Diseases 0.000 description 1
- 201000000639 Leber hereditary optic neuropathy Diseases 0.000 description 1
- 208000004554 Leishmaniasis Diseases 0.000 description 1
- 206010024214 Lenticular opacities Diseases 0.000 description 1
- 206010024229 Leprosy Diseases 0.000 description 1
- 208000002041 Loiasis Diseases 0.000 description 1
- 208000009564 MELAS Syndrome Diseases 0.000 description 1
- 201000009035 MERRF syndrome Diseases 0.000 description 1
- 208000001344 Macular Edema Diseases 0.000 description 1
- 208000031471 Macular fibrosis Diseases 0.000 description 1
- 206010025415 Macular oedema Diseases 0.000 description 1
- 208000035719 Maculopathy Diseases 0.000 description 1
- 206010063341 Metamorphopsia Diseases 0.000 description 1
- 201000002169 Mitochondrial myopathy Diseases 0.000 description 1
- 208000014844 Mitochondrial neurogastrointestinal encephalomyopathy Diseases 0.000 description 1
- 101000574441 Mus musculus Alkaline phosphatase, germ cell type Proteins 0.000 description 1
- 208000021642 Muscular disease Diseases 0.000 description 1
- 206010028372 Muscular weakness Diseases 0.000 description 1
- 208000000112 Myalgia Diseases 0.000 description 1
- 206010028424 Myasthenic syndrome Diseases 0.000 description 1
- 206010069825 Myoclonic epilepsy and ragged-red fibres Diseases 0.000 description 1
- 201000004458 Myoma Diseases 0.000 description 1
- 201000002481 Myositis Diseases 0.000 description 1
- 206010061533 Myotonia Diseases 0.000 description 1
- 208000010316 Myotonia congenita Diseases 0.000 description 1
- 206010068871 Myotonic dystrophy Diseases 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 208000001140 Night Blindness Diseases 0.000 description 1
- 206010067013 Normal tension glaucoma Diseases 0.000 description 1
- 102100021010 Nucleolin Human genes 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 206010030043 Ocular hypertension Diseases 0.000 description 1
- 208000009702 Optic Disk Drusen Diseases 0.000 description 1
- 208000036584 Optic disc drusen Diseases 0.000 description 1
- 206010061323 Optic neuropathy Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 208000030852 Parasitic disease Diseases 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 102000035195 Peptidases Human genes 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 206010057249 Phagocytosis Diseases 0.000 description 1
- 206010034944 Photokeratitis Diseases 0.000 description 1
- 208000007048 Polymyalgia Rheumatica Diseases 0.000 description 1
- 208000003971 Posterior uveitis Diseases 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 241000589517 Pseudomonas aeruginosa Species 0.000 description 1
- 101100022899 Pseudomonas aeruginosa merE gene Proteins 0.000 description 1
- 201000004360 Pthirus pubis infestation Diseases 0.000 description 1
- LCTONWCANYUPML-UHFFFAOYSA-M Pyruvate Chemical compound CC(=O)C([O-])=O LCTONWCANYUPML-UHFFFAOYSA-M 0.000 description 1
- 108091034057 RNA (poly(A)) Proteins 0.000 description 1
- 238000002123 RNA extraction Methods 0.000 description 1
- 238000010802 RNA extraction kit Methods 0.000 description 1
- 238000012228 RNA interference-mediated gene silencing Methods 0.000 description 1
- 102000018120 Recombinases Human genes 0.000 description 1
- 108010091086 Recombinases Proteins 0.000 description 1
- 208000037111 Retinal Hemorrhage Diseases 0.000 description 1
- 208000032430 Retinal dystrophy Diseases 0.000 description 1
- 206010038910 Retinitis Diseases 0.000 description 1
- 208000007014 Retinitis pigmentosa Diseases 0.000 description 1
- 102100031176 Retinoid isomerohydrolase Human genes 0.000 description 1
- 206010038923 Retinopathy Diseases 0.000 description 1
- 206010038926 Retinopathy hypertensive Diseases 0.000 description 1
- 206010038933 Retinopathy of prematurity Diseases 0.000 description 1
- 208000032458 Retinopathy solar Diseases 0.000 description 1
- 206010039020 Rhabdomyolysis Diseases 0.000 description 1
- 239000006146 Roswell Park Memorial Institute medium Substances 0.000 description 1
- 241000593989 Scardinius erythrophthalmus Species 0.000 description 1
- 206010039705 Scleritis Diseases 0.000 description 1
- 208000034189 Sclerosis Diseases 0.000 description 1
- 206010039729 Scotoma Diseases 0.000 description 1
- 238000000692 Student's t-test Methods 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- 206010042742 Sympathetic ophthalmia Diseases 0.000 description 1
- 230000024932 T cell mediated immunity Effects 0.000 description 1
- 238000010459 TALEN Methods 0.000 description 1
- 208000003217 Tetany Diseases 0.000 description 1
- 208000035954 Thomsen and Becker disease Diseases 0.000 description 1
- 208000031789 Thygeson superficial punctate keratitis Diseases 0.000 description 1
- 102100024324 Toll-like receptor 3 Human genes 0.000 description 1
- 102100033110 Toll-like receptor 8 Human genes 0.000 description 1
- 241000223996 Toxoplasma Species 0.000 description 1
- 108010043645 Transcription Activator-Like Effector Nucleases Proteins 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 229920004890 Triton X-100 Polymers 0.000 description 1
- 239000013504 Triton X-100 Substances 0.000 description 1
- BGDKAVGWHJFAGW-UHFFFAOYSA-N Tropicamide Chemical compound C=1C=CC=CC=1C(CO)C(=O)N(CC)CC1=CC=NC=C1 BGDKAVGWHJFAGW-UHFFFAOYSA-N 0.000 description 1
- 206010064996 Ulcerative keratitis Diseases 0.000 description 1
- COQLPRJCUIATTQ-UHFFFAOYSA-N Uranyl acetate Chemical compound O.O.O=[U]=O.CC(O)=O.CC(O)=O COQLPRJCUIATTQ-UHFFFAOYSA-N 0.000 description 1
- 108700005077 Viral Genes Proteins 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- 208000034699 Vitreous floaters Diseases 0.000 description 1
- 208000025749 Vogt-Koyanagi-Harada disease Diseases 0.000 description 1
- 208000034705 Vogt-Koyanagi-Harada syndrome Diseases 0.000 description 1
- 241000282485 Vulpes vulpes Species 0.000 description 1
- 206010048211 Xanthelasma Diseases 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 108010017070 Zinc Finger Nucleases Proteins 0.000 description 1
- 201000000761 achromatopsia Diseases 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 230000008649 adaptation response Effects 0.000 description 1
- 230000003044 adaptive effect Effects 0.000 description 1
- 230000033289 adaptive immune response Effects 0.000 description 1
- 102000035181 adaptor proteins Human genes 0.000 description 1
- 108091005764 adaptor proteins Proteins 0.000 description 1
- 206010064930 age-related macular degeneration Diseases 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 230000001668 ameliorated effect Effects 0.000 description 1
- 208000008303 aniridia Diseases 0.000 description 1
- 206010002537 anisometropia Diseases 0.000 description 1
- 230000003092 anti-cytokine Effects 0.000 description 1
- 229940124599 anti-inflammatory drug Drugs 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 230000002785 anti-thrombosis Effects 0.000 description 1
- 230000000840 anti-viral effect Effects 0.000 description 1
- 239000000427 antigen Substances 0.000 description 1
- 210000000612 antigen-presenting cell Anatomy 0.000 description 1
- 230000007503 antigenic stimulation Effects 0.000 description 1
- 108091007433 antigens Proteins 0.000 description 1
- 102000036639 antigens Human genes 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 101150010487 are gene Proteins 0.000 description 1
- 239000000607 artificial tear Substances 0.000 description 1
- 238000003149 assay kit Methods 0.000 description 1
- 201000009310 astigmatism Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000037444 atrophy Effects 0.000 description 1
- 229960000396 atropine Drugs 0.000 description 1
- RKUNBYITZUJHSG-SPUOUPEWSA-N atropine Chemical compound O([C@H]1C[C@H]2CC[C@@H](C1)N2C)C(=O)C(CO)C1=CC=CC=C1 RKUNBYITZUJHSG-SPUOUPEWSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 229940033687 beuthanasia Drugs 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 239000012620 biological material Substances 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 208000002352 blister Diseases 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 230000036772 blood pressure Effects 0.000 description 1
- 230000004378 blood-retinal barrier Effects 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- 229910052792 caesium Inorganic materials 0.000 description 1
- TVFDJXOCXUVLDH-UHFFFAOYSA-N caesium atom Chemical compound [Cs] TVFDJXOCXUVLDH-UHFFFAOYSA-N 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 208000003571 choroideremia Diseases 0.000 description 1
- 238000011260 co-administration Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 201000000159 corneal neovascularization Diseases 0.000 description 1
- 201000007717 corneal ulcer Diseases 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 201000004400 dacryoadenitis Diseases 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 210000001787 dendrite Anatomy 0.000 description 1
- 239000005547 deoxyribonucleotide Substances 0.000 description 1
- 125000002637 deoxyribonucleotide group Chemical group 0.000 description 1
- 230000000779 depleting effect Effects 0.000 description 1
- 201000001981 dermatomyositis Diseases 0.000 description 1
- HRLIOXLXPOHXTA-NSHDSACASA-N dexmedetomidine Chemical compound C1([C@@H](C)C=2C(=C(C)C=CC=2)C)=CN=C[N]1 HRLIOXLXPOHXTA-NSHDSACASA-N 0.000 description 1
- 229960004253 dexmedetomidine Drugs 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 201000003079 ectropion Diseases 0.000 description 1
- 238000000635 electron micrograph Methods 0.000 description 1
- 210000001163 endosome Anatomy 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000012854 evaluation process Methods 0.000 description 1
- 208000001936 exophthalmos Diseases 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000013604 expression vector Substances 0.000 description 1
- 208000030533 eye disease Diseases 0.000 description 1
- 208000008570 facioscapulohumeral muscular dystrophy Diseases 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 229950003499 fibrin Drugs 0.000 description 1
- 239000013020 final formulation Substances 0.000 description 1
- 238000002073 fluorescence micrograph Methods 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 201000006321 fundus dystrophy Diseases 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 108091006104 gene-regulatory proteins Proteins 0.000 description 1
- 102000034356 gene-regulatory proteins Human genes 0.000 description 1
- 238000010362 genome editing Methods 0.000 description 1
- 238000002873 global sequence alignment Methods 0.000 description 1
- 230000002641 glycemic effect Effects 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 210000003494 hepatocyte Anatomy 0.000 description 1
- 208000008025 hordeolum Diseases 0.000 description 1
- 210000002287 horizontal cell Anatomy 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 230000005745 host immune response Effects 0.000 description 1
- 102000045715 human TLR7 Human genes 0.000 description 1
- 201000006318 hyperopia Diseases 0.000 description 1
- 230000004305 hyperopia Effects 0.000 description 1
- 201000001948 hypertensive retinopathy Diseases 0.000 description 1
- 210000004713 immature microglia Anatomy 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 230000006450 immune cell response Effects 0.000 description 1
- 230000002163 immunogen Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 238000002991 immunohistochemical analysis Methods 0.000 description 1
- 238000011532 immunohistochemical staining Methods 0.000 description 1
- 229940088592 immunologic factor Drugs 0.000 description 1
- 239000000367 immunologic factor Substances 0.000 description 1
- 230000006054 immunological memory Effects 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 229940125721 immunosuppressive agent Drugs 0.000 description 1
- 229940124589 immunosuppressive drug Drugs 0.000 description 1
- 230000001976 improved effect Effects 0.000 description 1
- 208000023692 inborn mitochondrial myopathy Diseases 0.000 description 1
- 238000009540 indirect ophthalmoscopy Methods 0.000 description 1
- 230000002458 infectious effect Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229960003130 interferon gamma Drugs 0.000 description 1
- 229960001388 interferon-beta Drugs 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- NBQNWMBBSKPBAY-UHFFFAOYSA-N iodixanol Chemical compound IC=1C(C(=O)NCC(O)CO)=C(I)C(C(=O)NCC(O)CO)=C(I)C=1N(C(=O)C)CC(O)CN(C(C)=O)C1=C(I)C(C(=O)NCC(O)CO)=C(I)C(C(=O)NCC(O)CO)=C1I NBQNWMBBSKPBAY-UHFFFAOYSA-N 0.000 description 1
- 229960004359 iodixanol Drugs 0.000 description 1
- PGHMRUGBZOYCAA-UHFFFAOYSA-N ionomycin Natural products O1C(CC(O)C(C)C(O)C(C)C=CCC(C)CC(C)C(O)=CC(=O)C(C)CC(C)CC(CCC(O)=O)C)CCC1(C)C1OC(C)(C(C)O)CC1 PGHMRUGBZOYCAA-UHFFFAOYSA-N 0.000 description 1
- PGHMRUGBZOYCAA-ADZNBVRBSA-N ionomycin Chemical compound O1[C@H](C[C@H](O)[C@H](C)[C@H](O)[C@H](C)/C=C/C[C@@H](C)C[C@@H](C)C(/O)=C/C(=O)[C@@H](C)C[C@@H](C)C[C@@H](CCC(O)=O)C)CC[C@@]1(C)[C@@H]1O[C@](C)([C@@H](C)O)CC1 PGHMRUGBZOYCAA-ADZNBVRBSA-N 0.000 description 1
- 201000004614 iritis Diseases 0.000 description 1
- 229960002725 isoflurane Drugs 0.000 description 1
- 206010023332 keratitis Diseases 0.000 description 1
- 229960003299 ketamine Drugs 0.000 description 1
- 210000001865 kupffer cell Anatomy 0.000 description 1
- 208000016747 lacrimal apparatus disease Diseases 0.000 description 1
- 206010023683 lagophthalmos Diseases 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 208000019423 liver disease Diseases 0.000 description 1
- 201000002978 low tension glaucoma Diseases 0.000 description 1
- 230000002934 lysing effect Effects 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- 201000010230 macular retinal edema Diseases 0.000 description 1
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 1
- 239000001095 magnesium carbonate Substances 0.000 description 1
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 1
- 238000013178 mathematical model Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 239000011859 microparticle Substances 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 108010054609 middle-wavelength opsin Proteins 0.000 description 1
- 230000002438 mitochondrial effect Effects 0.000 description 1
- 238000009126 molecular therapy Methods 0.000 description 1
- 208000008588 molluscum contagiosum Diseases 0.000 description 1
- 238000001964 muscle biopsy Methods 0.000 description 1
- 201000006938 muscular dystrophy Diseases 0.000 description 1
- 230000036473 myasthenia Effects 0.000 description 1
- 206010028417 myasthenia gravis Diseases 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 208000001491 myopia Diseases 0.000 description 1
- 230000004379 myopia Effects 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 208000018360 neuromuscular disease Diseases 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 244000309711 non-enveloped viruses Species 0.000 description 1
- 230000006780 non-homologous end joining Effects 0.000 description 1
- 108010044762 nucleolin Proteins 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 238000013536 ocular coherence tomography Methods 0.000 description 1
- 201000005111 ocular hyperemia Diseases 0.000 description 1
- 208000003177 ocular onchocerciasis Diseases 0.000 description 1
- 229940118344 ocusoft Drugs 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 208000002042 onchocerciasis Diseases 0.000 description 1
- 210000003733 optic disk Anatomy 0.000 description 1
- 210000001328 optic nerve Anatomy 0.000 description 1
- 208000020911 optic nerve disease Diseases 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 208000014380 ornithine aminotransferase deficiency Diseases 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 230000020477 pH reduction Effects 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 102000007863 pattern recognition receptors Human genes 0.000 description 1
- 108010089193 pattern recognition receptors Proteins 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 229960002275 pentobarbital sodium Drugs 0.000 description 1
- 201000003841 peripheral retinal degeneration Diseases 0.000 description 1
- 230000008782 phagocytosis Effects 0.000 description 1
- 239000008177 pharmaceutical agent Substances 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- SONNWYBIRXJNDC-VIFPVBQESA-N phenylephrine Chemical compound CNC[C@H](O)C1=CC=CC(O)=C1 SONNWYBIRXJNDC-VIFPVBQESA-N 0.000 description 1
- 229960001802 phenylephrine Drugs 0.000 description 1
- 229960002790 phenytoin sodium Drugs 0.000 description 1
- 150000004713 phosphodiesters Chemical class 0.000 description 1
- 238000013310 pig model Methods 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 102000040430 polynucleotide Human genes 0.000 description 1
- 108091033319 polynucleotide Proteins 0.000 description 1
- 239000002157 polynucleotide Substances 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 230000036515 potency Effects 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 201000010041 presbyopia Diseases 0.000 description 1
- 210000000063 presynaptic terminal Anatomy 0.000 description 1
- 238000009516 primary packaging Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 230000000750 progressive effect Effects 0.000 description 1
- 201000003004 ptosis Diseases 0.000 description 1
- 210000003314 quadriceps muscle Anatomy 0.000 description 1
- 229920013730 reactive polymer Polymers 0.000 description 1
- 230000010837 receptor-mediated endocytosis Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000029058 respiratory gaseous exchange Effects 0.000 description 1
- 210000003583 retinal pigment epithelium Anatomy 0.000 description 1
- 201000007714 retinoschisis Diseases 0.000 description 1
- 238000003757 reverse transcription PCR Methods 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 230000037387 scars Effects 0.000 description 1
- 210000003786 sclera Anatomy 0.000 description 1
- 238000007789 sealing Methods 0.000 description 1
- 230000003248 secreting effect Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000002864 sequence alignment Methods 0.000 description 1
- 108091006024 signal transducing proteins Proteins 0.000 description 1
- 102000034285 signal transducing proteins Human genes 0.000 description 1
- 210000002027 skeletal muscle Anatomy 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 1
- FJPYVLNWWICYDW-UHFFFAOYSA-M sodium;5,5-diphenylimidazolidin-1-ide-2,4-dione Chemical compound [Na+].O=C1[N-]C(=O)NC1(C=1C=CC=CC=1)C1=CC=CC=C1 FJPYVLNWWICYDW-UHFFFAOYSA-M 0.000 description 1
- 201000000824 solar retinopathy Diseases 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 208000002320 spinal muscular atrophy Diseases 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 229940021506 stye Drugs 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 230000008093 supporting effect Effects 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 208000018657 thygeson superficial punctate keratopathy Diseases 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- 108091006106 transcriptional activators Proteins 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 238000004627 transmission electron microscopy Methods 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 229960004791 tropicamide Drugs 0.000 description 1
- 238000005199 ultracentrifugation Methods 0.000 description 1
- 230000024275 uncoating of virus Effects 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 210000003934 vacuole Anatomy 0.000 description 1
- 230000004095 viral genome expression Effects 0.000 description 1
- 230000029812 viral genome replication Effects 0.000 description 1
- 230000009385 viral infection Effects 0.000 description 1
- 210000002845 virion Anatomy 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
- 108010047303 von Willebrand Factor Proteins 0.000 description 1
- 102100036537 von Willebrand factor Human genes 0.000 description 1
- 229960001134 von willebrand factor Drugs 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- 208000005494 xerophthalmia Diseases 0.000 description 1
- 201000009482 yaws Diseases 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
- A61K48/0058—Nucleic acids adapted for tissue specific expression, e.g. having tissue specific promoters as part of a contruct
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/0075—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the delivery route, e.g. oral, subcutaneous
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/117—Nucleic acids having immunomodulatory properties, e.g. containing CpG-motifs
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2710/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA dsDNA viruses
- C12N2710/00011—Details
- C12N2710/10011—Adenoviridae
- C12N2710/10311—Mastadenovirus, e.g. human or simian adenoviruses
- C12N2710/10341—Use of virus, viral particle or viral elements as a vector
- C12N2710/10343—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14143—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- viruses have evolved to become highly efficient at nucleic acid delivery to specific cell types while avoiding immuno surveillance by an infected host. These properties make viruses attractive gene-delivery vehicles, or vectors, for gene therapy.
- retrovirus adenovirus
- AAV adeno-associated virus
- herpes simplex virus have been modified in the laboratory for use in gene therapy (Robbins, PD et al. Pharmol Ther,
- viral molecular therapy vectors that inhibit nucleic acid-mediated inflammatory responses while boosting expression of a desired therapeutic molecule (e.g., a therapeutic gene of interest).
- the vectors of the present disclosure include a recombinant viral genome linked in cis to an inhibitory oligonucleotide sequence that prevents virally-induced production of proinflammatory cytokines.
- the inhibitory oligonucleotide inhibits nucleic acid-mediated activation of toll-like receptors and/or inhibits nucleic acid-mediated toll-like receptor (TLR) signaling (e.g., TLR9).
- TLR nucleic acid-mediated toll-like receptor
- inclusion of the inhibitory oligonucleotide in the viral genome not only inhibits the inflammatory response, but it also increases transduction efficiency and/or efficacy of the therapeutic nucleotide sequence and/or expression levels of expression products encoded by nucleic acids, as appropriate.
- the amount of recombinant viral genome needed to be therapeutically effective is less than the amount needed with conventional viral vector delivery systems that do not include an inhibitory oligonucleotide.
- some aspects of the present disclosure provide recombinant viral genomes comprising a therapeutic nucleic acid (e.g., DNA encoding a gene (e.g., Cas9) or gene fragment (e.g., a replacement exon of interest) and an inhibitory oligonucleotide that inhibits the production of proinflammatory cytokines.
- methods comprising administering to a subject a recombinant viral genome that comprises a therapeutic nucleic acid (e.g., DNA) and an inhibitory oligonucleotide that inhibits the production of proinflammatory cytokines.
- the recombinant viral genomes are administered intramuscularly.
- the recombinant viral genomes are administered intravenously.
- the recombinant viral genomes are administered to the eye (e.g., intravitreally).
- aspects of the present disclosure provide recombinant viral genomes comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that inhibits nucleic acid- sensing TLR activation and/or signaling. Also provided herein, in some aspects, are methods comprising administering to a subject a recombinant viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that inhibits nucleic acid-sensing TLR activation and/or signaling.
- the TLR is TLR9.
- the inhibitory oligonucleotide binds to the TLR.
- the inhibitory oligonucleotide binds to inflammatory nucleic acids.
- the inflammatory nucleic acids comprise
- the inhibitory oligonucleotide comprises a
- FIGS. 1A-1C includes data showing the impact of exemplary inhibitory oligonucleotides on TLR9 activation in vitro.
- HEK293-TLR9 reporter cells were treated with the indicated constructs (single-stranded DNA with a phosphorothioate backbone) at the indicated constructs (single-stranded DNA with a phosphorothioate backbone) at the indicated constructs (single-stranded DNA with a phosphorothioate backbone) at the indicated
- FIG. 1A TLR9 activating nucleic acid (ODN 2006) linked to each of the indicated sequences on the X-axis was tested in the HEK293-TLR9 reporter system.
- the left graph in FIG. 1A shows results using 0.5 ⁇ of the constructs.
- the right graph in FIG. 1A shows results using 5 ⁇ of the constructs.
- IB TLR9 activating nucleic acid (ODN 2006) linked through a AAAAA (SEQ ID NO: 8) linker to each of the indicated sequences on the X-axis was tested in the HEK293-TLR9 reporter system.
- FIG. 1C TLR9 activating nucleic acid (ODN 2006) linked to each of the sequences indicated on the X-axis was tested in the HEK293-TLR9 reporter system.
- FIGS. 2A-2B show muscle tissue data relating to the impact of three copies of the telomere oligonucleotide ("3xtelomere” SEQ ID NO: 35) on the inflammatory response elicited by a linked self-complementary adeno-associated virus (scAAV) genome and on AAV transgene expression in infected mice.
- FIG. 2A shows data relating to the impact of indicated constructs or control saline on expression of proinflammatory cytokines, using qRT-PCR.
- Adult C57BL/6 mice were infected with indicated AAV2 viruses (10 11 vg per mouse) by intramuscular injection in the quadricep.
- FIG. 2B shows data relating to the impact of indicated AAV vectors on GFP expression.
- Adult C57BL/6 mice were infected with indicated AAV2 viruses similar to FIG. 2A and a piece of the quadricep was analyzed for GFP gene expression by qRT-PCR 28 d later.
- scAAV-eGFP infection was set to 1-fold expression for GFP.
- FIGS. 3A-3C includes liver tissue data relating to the impact of three copies of telomere ("3xtelomere” SEQ ID NO: 35) and three copies of c41 (3 X c4 ⁇ oligonucleotides on the inflammatory response elicited by a linked self-complementary adeno-associated virus (scAAV) in infected mice. All three panels show data relating to the impact of indicated constructs or control saline on expression of proinflammatory cytokines, using qRT-PCR.
- Adult C57BL/6 mice were infected with indicated AAV2 viruses (10 11 vg per mouse) by tail vein injections.
- FIG. 3D includes liver tissue data relating to the impact of the inhibitory oligonucleotide sequence or a control sequence on the inflammatory response elicited by a linked scAAV genome in infected mice.
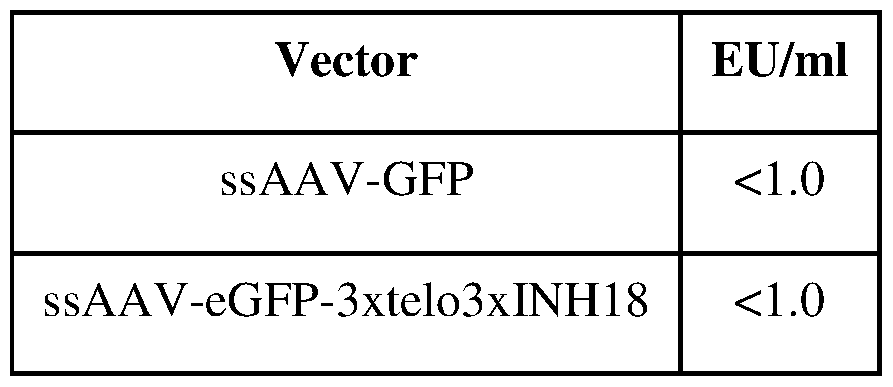
- FIG. 3E includes liver tissue data relating to the impact of three copies of telomere oligonucleotide ("3xtelomere" SEQ ID NO: 35) on scAAV transgene expression in infected mice.
- FIG. 4 is a schematic representation of a single-stranded AAV vector encoding GFP (ssAAV-eGFP). Two engineered forms are shown. ssAAV-eGFP-5xtelomere carries an insertion of 5 copies of telomere oligonucleotide (5 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of another 5 copies with AAAAA linkers (in anti-sense orientation).
- ssAAV-eGFP-5xtelomere carries an insertion of 5 copies of telomere oligonucleotide (5 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of another 5 copies with AAAAA linkers (in anti-sense orientation).
- ssAAV-eGFP-3xtelo3xINH18 carries an insertion of 3 copies of telomere oligonucleotide (3 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of 3 copies of INH-18 (3 X SEQ ID NO: 5) with AAAAA (SEQ ID NO: 8) linkers (in anti-sense orientation) ("3xtelo3xINH18 SEQ ID NO 39).
- LpA poly A signal.
- FIG. 5 includes a graph showing inflammatory responses to various inhibitory oligonucleotides in HEK293-TLR9 reporter cells in vitro.
- HEK293-TLR9 reporter cells were treated with 0.02 ⁇ of the indicated oligonucleotides (see Example 1) for 18 hours.
- a 50 ⁇ sample of cell supernatant was incubated with 150 ⁇ HEK-Blue Detection and then analyzed on a plate reader for absorbance at 630 nm.
- Control a random 24 nucleotide strand
- ODN 2006 SEQ ID NO: 26
- ODN TTAGGG SEQ ID NO: 6
- ODN 2006 co-administered with ODN TTAGGG in trans.
- FIGS. 6A- 6B includes data showing the impact of exemplary inhibitory
- HEK293-TLR7 or HEK293-TLR2 reporter cells were treated with or without a high concentration of 5 ⁇ of the indicated constructs (single-stranded DNA with a phosphorothioate backbone), along with the appropriate TLR7 stimulant (1 ⁇ of Gardiquimod) or TLR2 stimulant (100 ng/ml of FSL-2), for 18 hours.
- TTAGGG in FIGS. 6A-6B refers to ODN TTAGGG (SEQ ID NO: 6).
- FIG. 6A shows results using HEK293-TLR7 reporter cells and FIG. 6B shows results using HEK293-TLR2 reporter cells.
- FIGS. 7A-7E show the effects of incorporation of three copies of the telomere sequence ("3xtelomere" SEQ ID NO: 35) into a self-complementary AAV vector encoding human factor IX on innate immune response and transgene expression in vivo in liver tissue.
- FIGS. 7A shows a schematic diagram of vector organization of scAAV-FIX (the original "wild-type" vector, SEQ ID NO: 33) and scAAV-FIX-3xtelomere (SEQ ID NO: 34).
- AAV vectors were packaged in an AAV8 capsid.
- ITR inverted terminal repeat
- TTR transthyretin promoter
- hFIX human factor IX
- bGH bovine growth hormone poly(A) signal
- TRS terminal resolution site.
- FIGS. 8A-8D include data showing the effects of multiple copies of inhibitory oligonucleotides (multiple copies of telomere (multiple copies of SEQ ID NO: 9) and multiple copies of INH-18 (multiple copies of SEQ ID NO: 5)) on AAV-induced T cell reactivity and T cell infiltration.
- FIG. 8A includes data where mice received indicated doses of AAVrh32.33 vectors (single- stranded AAV) via intramuscular injections and 21 d later, splenocytes were subject to IFN- ⁇ ELISPOT assays to quantify CD8+ T cell responses to an immunodominant epitope of rh32.33 capsid.
- FIGS. 8B-8C show the number of CD8+ T cells and CD8+ Granzyme B+ T cells in the muscle sections (four fields examined per sample) for PBS and 1 xlO 10 vg rh32.33 vectors 21 dpi. (FIG.
- FIGS. 9A-9B show characterization of AAV8 vectors used in a pig study.
- ssAAV-eGFP vector is provided as SEQ ID NO: 36.
- ssAAV-eGFP-3xtelo3xINH18 vector is provided as SEQ ID NO: 36.
- FIG. 9A is a silver staining comparison of 1 x 10 vg AAV8 vectors.
- FIG. 9B is a series of transmission electron microscopy images showing negatively stained AAV, and representative empty particles are shown with white arrows. Representative images are shown for each vector. Scale bar: 100 nm.
- FIGS. lOA-lOC show that engineered vector evades photoreceptor pathology and immune responses in subretinal-injected pig eyes.
- ssAAV-eGFP is provided as SEQ ID NO: 36.
- ssAAV-eGFP-3xtelo3xINH18 is provided as SEQ ID NO: 37.
- FIG. 10A shows
- FIG. 10B shows microglia proliferation and activation in the retina indicated by anti-Ibal staining.
- FIG. IOC shows cytotoxic T cell infiltration into the retina indicated by anti-CD8 staining. Scale bars, 50 ⁇ .
- ONL outer nuclear layer
- Ibal ionized calcium-binding adaptor protein 1.
- FIG. 11 shows that engineered vector comprising an inhibitory oligonucleotide evades photoreceptor pathology in subretinal-injected pig eyes.
- ssAAV-eGFP is provided as SEQ ID NO: 36.
- ssAAV- eGFP-3xtelo3xINH18 is provided as SEQ ID NO: 37.
- FIG. 12 shows a schematic representation of a single- stranded AAV vector encoding GFP (ssAAV-eGFP).
- ssAAV-eGFP-3xtelo3xINH18 (SEQ ID NO: 37) carries an insertion of 3 copies of telomere oligonucleotide (3 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of 3 copies of INH-18 (3 X SEQ ID NO: 5) with AAAAA linkers (in anti-sense orientation, i.e., anti-sense orientation of SEQ ID NO: 8) in the 3' untranslated region (between the polyA signal and right ITR).
- ssAAV-eGFP-double (SEQ ID NO: 38) carries the same insertion, but additionally carries an insertion of 3 copies of 4084-F (3 X SEQ ID NO: 3) oligonucleotide with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of 3 copies of 2088 (3 X SEQ ID NO: 2) oligonucleotide with AAAAA linkers (in anti-sense orientation, i.e., anti-sense orientation of SEQ ID NO: 8) in the 5' untranslated region (between the left ITR and the promoter).
- LpA poly A signal.
- FIG. 13 shows a schematic representation of a different single-stranded AAV vector encoding GFP (ssAAV-eGFP-WPRE).
- ssAAV-eGFP-WPRE- 3xtelo3xINH18 (SEQ ID NO: 42) carries an insertion of 3 copies of telomere oligonucleotide (3 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by reverse complementary sequences of 3 copies of INH-18 (3 X SEQ ID NO: 5) with AAAAA linkers (in anti-sense orientation, i.e., anti-sense orientation of SEQ ID NO: 8) in the 3' untranslated region (between the polyA signal and right ITR).
- WPRE Woodchuck Hepatitis Virus Posttranscriptional Regulatory Element.
- LpA polyA signal.
- gene therapy with viral vectors often includes systemic treatment with an immunosuppressive agent.
- immunosuppressive and anti-inflammatory drugs can compromise the patient's immune system during treatment and patients often still develop neutralizing antibodies or T cells to exogenous biological materials (e.g., against AAV capsid), precluding future re- administration or leading to destruction of transduced cells.
- recombinant viral genomes that may be used as delivery vehicles, without inducing a substantial inflammatory response.
- the recombinant viral genomes induce less of an inflammatory response than would otherwise be induced without the presence of the inhibitory oligonucleotide.
- the recombinant viral genomes of the present disclosure include an inhibitory oligonucleotide that inhibits production of proinflammatory cytokines, thus, inhibiting the inflammatory response.
- the inhibitory oligonucleotide inhibits an inflammatory response induced by the toll-like receptor (TLR) pathway, for example, by inhibiting (preventing) TLR activation and/or inhibiting TLR signaling.
- TLR toll-like receptor
- Viral vectors are often studied in experimental and clinical models as agents for gene therapy. Recent generations of viral vectors have the majority of viral genes removed and result in vectors with a large carrying capacity, reduced host immune responses and improved gene transfer efficiency. Some viral vectors, such as adenovirus vectors and adeno-associated virus vectors, however, still activate innate immune responses following administration in vivo.
- the innate response to viral vectors is independent of
- PAMPs pattern-associated molecular patterns
- This response results in inflammation of transduced tissues and can reduce viral transduction efficiency.
- Viral infection can activate a number of signaling pathways following cell entry that ultimately lead to expression of inflammatory (proinflammatory) genes.
- inflammatory proinflammatory
- cytokines, chemokines and leukocyte adhesion molecules are induced by the viral vector in a wide range of cell types providing a molecular basis for the inflammatory properties of these vectors. See Liu, Q. et al. Gene Therapy 2003;10:935-940.
- TLR toll-like receptor
- TLR9 is immune pattern- recognition receptors that detect pathogens and damaged cells.
- TLR9 amino acid sequences can be found in publically-available gene databases, such as GenBank and UnitProtKB.
- the amino acid sequence of wild-type human TLR9 can be identified as UniProtKB entryQ9NR96 (TLR9_Human).
- TLR9 is generally located on endosomal membranes in immune cells.
- TLR9 is an exemplary nucleic acid-sensing TLRs that detects exogenous nucleic acids (inflammatory nucleic acids) that have entered a cell (see, e.g. Takeda, K et al., Semin Immunol. 2004;16(1):3- 9; Lee, J et al. Proc Natl Acad Sci USA. 2011; 108(34): 14055-60).
- Nucleic acids recognized by TLR9 include those that originate from bacteria, viruses or even endogenous nucleic acids.
- TLRs' are TLRs that can bind to nucleic acids, such as ssRNA, dsRNA, and DNA containing unmethylated CpG (cytosine-phosphate-guanine) motifs. This binding typically results in the dimerization of TLR and activation of TLR signaling, which results in the production of (expression of and/or activation of) proinflammatory molecules, such as proinflammatory cytokines.
- TLR9 recognizes nucleic acids with unmethylated CpGs (Kumagai, Y, et al. Adv Drug Deliv Rev. 2008;60(7)795-804).
- 'inflammatory nucleic acids' are nucleic acids that activate TLR signaling (e.g., bind to TLR to activate TLR signaling).
- the inflammatory nucleic acids comprise deoxycytidyl-deoxyguanosine (CpG) oligodeoxynucleotides.
- CpG oligodeoxynucleo tides are sequences that comprise at least one unmethylated CpG motif and activate an immune response. See, e.g., Krieg, AM et al. Nature. 1995; 374(6522):546-9.
- the inflammatory nucleic acids activate TLR9 signaling (e.g., by binding to TLR9).
- TLR9 signaling e.g., by binding to TLR9
- An exemplary inflammatory nucleic acid is provided in Example 1 (ODN 2006).
- TLR signaling results in an inflammatory response characterized by gene expression of antiviral molecules and proinflammatory cytokines, including type I interferons and NF-kB (p25-RelA complex) target genes.
- TLR signaling may be used as means to determine the impact of an inhibitory oligonucleotide on the inflammatory response.
- a TLR reporter cell line may be used to assess the inhibitory nature of an oligonucleotide, for example, by determining the level of cytokine (e.g. IL6, CXCL10 and/or TNF) production, which reflects the level of TLR signaling (see, e.g., Example 1).
- cytokine e.g. IL6, CXCL10 and/or TNF
- the level of a particular cytokine is measured using quantitative PCR with primers targeting the cytokine of interest (see, e.g., Examples 3 and 4). Additional methods of measuring cytokine levels include enzyme-linked immunosorbent assay (ELISA) and Western blot analysis with an anti-cytokine antibody.
- ELISA enzyme-linked immunosorbent assay
- Inhibition of an inflammatory response may be measured as a decrease in TLR signaling.
- a decrease in cytokine activity level or expression level e.g., a 2-fold, 5-fold, 10- fold, 50-fold reduction
- a decrease in cytokine activity level or expression level e.g., a 2-fold, 5-fold, 10- fold, 50-fold reduction
- a control may indicate inhibition (partial or complete inhibition) of the inflammatory response (see, e.g., Examples 3 and 4).
- An inhibitory oligonucleotide is an oligonucleotide that, when co-delivered in vivo with another nucleic acid (such as a viral genome, a single-stranded RNA, or a single-stranded DNA), inhibits the production of proinflammatory cytokines, relative to proinflammatory cytokine production in the absence of the inhibitory oligonucleotide.
- another nucleic acid such as a viral genome, a single-stranded RNA, or a single-stranded DNA
- the inhibitory oligonucleotides of the present disclosure comprise at least two nucleotides covalently linked together, and in some instances, may contain phosphodiester bonds (e.g., a phosphodiester "backbone"). In some instances, the oligonucleotide may contain phosphorothioate bonds (e.g., a phosphorothioate backbone).
- the length of an inhibitory oligonucleotide may vary, but is should be understood that the length of an inhibitory oligonucleotide is typically 4 to 200 nucleotides.
- an inhibitory oligonucleotide has a length of 4 to 100 nucleotides. In some embodiments, an inhibitory oligonucleotide has a length of 4 to 10, 4 to 20, 4 to 30, 4 to 50, 4 to 60, 4 to 70, 4 to 80, or 4 to 90 nucleotides. In some embodiments, an inhibitory oligonucleotide has a length of 5 to 10, 5 to 20, 5 to 30, 5 to 50, 5 to 60, 5 to 70, 5 to 80, 5 to 90, or 5 to 100 nucleotides.
- an inhibitory oligonucleotide has a length of 6 to 10, 6 to 20, 6 to 30, 6 to 50, 6 to 60, 6 to 70, 6 to 80, 6 to 90, or 6 to 100 nucleotides. In some embodiments, an inhibitory oligonucleotide has a length of 7 to 10, 7 to 20, 7 to 30, 7 to 50, 7 to 60, 7 to 70, 7 to 80, 7 to 90, or 7 to 100 nucleotides. In some embodiments, an inhibitory oligonucleotide has a length of 8 to 10, 8 to 20, 8 to 30, 8 to 50, 8 to 60, 8 to 70, 8 to 80, 8 to 90, or 8 to 100 nucleotides.
- an inhibitory oligonucleotide has a length of 9 to 10, 9 to 20, 9 to 30, 9 to 50, 9 to 60, 9 to 70, 9 to 80, 9 to 90, or 9 to 100 nucleotides. In some embodiments, an inhibitory oligonucleotide has a length of 10 to 10, 10 to 20, 10 to 30, 10 to 50, 10 to 60, 10 to 70, 10 to 80, 10 to 90, or 10 to 100 nucleotides. Inhibitory oligonucleotides may be produced recombinantly or synthetically, for example.
- the inhibitory oligonucleotide comprises or consists of deoxyribonucleotides.
- the inhibitory oligonucleotide is an inhibitory DNA oligonucleotide.
- the inhibitory oligonucleotide does not include RNA. It should be understood that the definition of inhibitory oligonucleotides, as provided herein, specifically excludes RNA interference molecules (RNAi), such as short interfering RNA (siRNA) molecules.
- RNAi RNA interference molecules
- siRNA short interfering RNA
- the inhibitory oligonucleotides inhibit the activation of nucleic acid-sensing TLRs.
- the inhibitory oligonucleotides may act as molecular scavengers and bind to (and sequester) inflammatory nucleic acids, thus preventing the inflammatory nucleic acids from binding the TLR and activating TLR signaling.
- the inhibitory oligonucleotides may prevent dimerization of a TLR.
- the inhibitory oligonucleotides inhibit TLR signaling (to downstream molecules).
- the inhibitory oligonucleotides may bind indirectly or directly to a TLR (e.g., TLR9) to block TLR-mediated production of proinflammatory cytokines (e.g., induction of proinflammatory cytokine activity and/or expression).
- TLR e.g., TLR9
- proinflammatory cytokines e.g., induction of proinflammatory cytokine activity and/or expression.
- the inhibitor oligonucleotide competes for receptor-mediated endocytosis or phagocytosis. In some embodiments, the inhibitor oligonucleotide inhibits TLR9 trafficking. In some embodiments, the inhibitor oligonucleotide inhibits TLR9 processing into a functionally active product. In some embodiments, the inhibitor oligonucleotide inhibits endosomal acidification or activity of key proteases in endosomes. In some embodiments, the inhibitor oligonucleotide blocks signaling proteins downstream of TLR9.
- an inhibitory oligonucleotide may reduce nucleic acid- sensing TLR activation and/or signaling by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95%, relative to control (nucleic acid-sensing TLR activation and/or signaling in the absence of the inhibitory oligonucleotide).
- the inhibitory oligonucleotides of the present disclosure inhibit production of proinflammatory cytokines.
- proinflammatory cytokines include interleukins (e.g. , IL- 1, IL-6, IL- 17 and IL-18), interferons (IFNs, e.g. , interferon a (IFNa), interferon ⁇ (IFNP), and interferon ⁇ (IFNy)), tumor necrosis factors (TNFs) (e.g. , TNF- a) and chemokines (e.g. , CCL2, CXCL10 and CCL5).
- IFNs interferons
- TNFs tumor necrosis factors
- chemokines e.g. , CCL2, CXCL10 and CCL5
- the inhibitory oligonucleotide inhibits IL-6, CXCL10 and/or TNF production. In some embodiments, the inhibitory oligonucleotide inhibits IL-6 production. In some embodiments, the inhibitory oligonucleotide inhibits CXCL10 production. In some embodiments, the inhibitory
- oligonucleotide inhibits TNF production.
- the level of inflammatory cytokine production may be measured using Western blot analysis, quantitative PCR and/or enzyme-linked immunosorbent assay. Other assays for assessing the inflammatory response are known and may be used as provided herein.
- an inhibitory oligonucleotide reduces production of (activity of and/or expression of) proinflammatory cytokines by at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, or at least 95%, relative to control (inflammatory cytokine production in the absence of the inhibitory oligonucleotide).
- the inhibitory oligonucleotides of the present disclosure may include different motifs that contribute to anti-inflammatory properties of the inhibitory oligonucleotide.
- the inhibitory oligonucleotide comprises at least one CCx(not-C)(not-C)xxGGG motif, wherein x is any nucleic acid (e.g., A, T, C or G (but not C where specified)). See, e.g., Ashman, RF et al. Int Immunol. 2011 ; 23(3):203- 14.
- Non-limiting examples of inhibitory oligonucleotides that comprise at least one CCx(not-C)(not-C)xxGGG motif include ODN 4228 (see, e.g.
- SEQ ID NO: 2 SEQ ID NO: 3
- SEQ ID NO: 4 SEQ ID NO: 5
- SEQ ID NO: 7 SEQ ID NO: 16
- SEQ ID NO: 17 SEQ ID NO: 18
- SEQ ID NO: 19 SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO: 23, and SEQ ID NO: 24.
- an inhibitory oligonucleotide comprises at least one TTAGGG motif (SEQ ID NO: 61).
- an inhibitory oligonucleotide may comprise at least 2, 3, 4, 5, 6, 7, 8, 9 or 10 TTAGGG motifs.
- the inhibitory oligonucleotide includes two TTAGGG motifs.
- the inhibitory oligonucleotide includes three TTAGGG motifs.
- the inhibitory oligonucleotide includes four TTAGGG motifs.
- Exemplary inhibitory oligonucleotides comprising at least one TTAGGG motif include those identified by SEQ ID NO:6 and/or SEQ ID NO: 9.
- the inhibitory oligonucleotide includes at least one sequence that is identical to a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the inhibitory oligonucleotide includes a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 1. In some embodiments, the inhibitory oligonucleotide includes a nucleotide sequence that is identical to SEQ ID NO: 6. In some embodiments, the inhibitory oligonucleotide includes a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 9.
- the inhibitory oligonucleotide includes multiple tandem repeats of a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- tandem repeats are sequences that that follow one another.
- the tandem repeats may be directly next to one another (e.g., TTAGGGTTAGGGTTAGGG (repeated sequence underlined)).
- the tandem repeats may be separated by another sequence (e.g., a linker sequence) (e.g., TTAGGG-linker-TTAGGG-linker-TTAGGG).
- the inhibitory oligonucleotide includes multiple tandem repeat sequences (e.g. two, three, four or five tandem repeats).
- the inhibitory oligonucleotide includes multiple tandem repeats (e.g. two, three, four or five repeats) of SEQ ID NO: 1. In some embodiments, the inhibitory oligonucleotide includes multiple tandem repeats (e.g. two, three, four or five repeats) of SEQ ID NO: 9. In some embodiments, the inhibitory oligonucleotide includes multiple tandem repeats (e.g. two, three, four or five repeats) of SEQ ID NO: 6.
- the multiple tandem repeat sequences in an inhibitory are in some embodiments.
- oligonucleotide are separated by a linker.
- the linker may be oriented in the sense or antisense direction.
- a linker may be oriented in the sense direction if it is separating multiple tandem repeat sequences that are also oriented in the sense direction.
- a linker is oriented in the antisense direction when it is separating multiple tandem repeat sequences that are also oriented in the antisense direction.
- the linker is a polyA linker (a string of "A" nucleotides).
- the polyA linker comprises at least one nucleotide sequence that is identified by SEQ ID NO: 8.
- the polyA linker comprises two nucleotide sequences that is identified by SEQ ID NO: 8.
- the polyA linker comprises three nucleotide sequences that is identified by SEQ ID NO: 8.
- oligonucleotide may have three tandem repeats of the sequence in SEQ ID NO: 1 and each of the tandem repeats may be separated by a linker identified by SEQ ID NO: 8.
- the linker comprises at least one nucleotide sequence (e.g. , at least 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotide sequences) that is the antisense sequence of SEQ ID NO: 8 (i.e. , SEQ ID NO: 8 oriented in the antisense direction).
- an inhibitory oligonucleotide may have three tandem repeats of the sequence in SEQ ID NO: 9 or SEQ ID NO: 6 and each of the tandem repeats may be separated by a linker identified by SEQ ID NO: 8.
- the viral genome comprises a sequence that is at least 90% (e.g. , at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or is 100%) identical to SEQ ID NO: 35.
- a recombinant viral genome is single- stranded and comprises the sequence in SEQ ID NO: 35 that is located upstream (5') of a therapeutic nucleotide sequence (e.g. , Factor IX) (e.g. , in the 5' UTR).
- a therapeutic nucleotide sequence e.g. , Factor IX
- a recombinant viral genome is single- stranded and comprises the sequence in SEQ ID NO: 35 that is located upstream (5') of a therapeutic nucleotide sequence (e.g. , Factor IX) (e.g. , in the 5' UTR).
- a therapeutic nucleotide sequence e.g. , Factor IX
- the recombinant viral genome is single-stranded and comprises the sequence in SEQ ID NO: 35 that is located downstream (3 ') of a therapeutic nucleotide sequence (e.g. , Factor IX) (e.g. , downtream from (3') a polyA tail linked to the therapeutic nucleic acid, including in the 3 ' UTR).
- the viral genome comprises a sequence that is at least 90% (e.g. , at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or is 100%) identical to SEQ ID NO: 34.
- the inhibitory oligonucleotide includes a combination of nucleotide sequences of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the inhibitory oligonucleotide may include at least one copy (e.g. , one, two, three, four, five, six or seven copies) of a nucleotide sequence of of any one of SEQ ID NOS: 1-7, 9-25, or 27-32 combined with at least one copy (e.g. , one, two, three, four, five, six or seven copies) of a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the inhibitory oligonucleotide includes three copies of SEQ ID NO: 9 and three copies of SEQ ID NO: 5 (e.g., SEQ ID NO: 39). In some embodiments, the inhibitory oligonucleotide comprises three copies of SEQ ID NO: 3 and three copies of SEQ ID NO: 2. In some embodiments, the inhibitory oligonucleotide includes five copies of SEQ ID NO: 1 and three copies of SEQ ID NO: 9. See also Example 8 and Materials and Methods section of the Examples below.
- a single expression construct may include more than one (e.g. , at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 15, or at least 20) type of inhibitory oligonucleotides, one type (one or more copies, e.g., 2, 3, 4 or 5 tandem copies) in the sense orientation and another type (one or more copies, e.g., 2, 3, 4 or 5 tandem copies) in the antisense orientation.
- inclusion of two different types of inhibitory oligonucleotides may prevent self-annealing between the inhibitory oligonucleotides and formation of unwanted hairpin structures.
- a recombinant single- stranded viral genome comprising an inhibitory oligonucleotide in the sense orientation and another inhibitory oligonucleotide in the antisense direction increases the probability that each packaged viral genome comprises at least one inhibitory oligonucleotide in the correct orientation.
- an inhibitory oligonucleotide may have three tandem repeats of the sequence in SEQ ID NO: 9 or SEQ ID NO: 6 and three tandem repeats of the sequence in SEQ ID NO: 5.
- the tandem repeats of the sequence in SEQ ID NO: 9 or SEQ ID NO: 6 may be oriented in the opposite direction of the tandem repeats of the sequence in SEQ ID NO: 5.
- the tandem repeats of the sequence in SEQ ID NO: 9 or SEQ ID NO: 6 may be oriented in the sense direction and the tandem repeats of the sequence in SEQ ID NO: 5 may be oriented in the antisense direction or vice versa.
- the tandem repeats of SEQ ID NO: 9 or SEQ ID NO: 6 may be upstream (5') or downstream (3 ') of the tandem repeats of the sequence in SEQ ID NO: 5.
- Each of the tandem repeats (e.g. , repeats of SEQ ID NOS: 5, 6, 9, or any combination thereof) may be separated by a linker identified by SEQ ID NO: 8 oriented in the sense or antisense direction.
- the viral genome comprises a sequence that is at least 90% (e.g. , at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or is 100%) identical to SEQ ID NO: 39.
- a recombinant viral genome is single- stranded and comprises the sequence in SEQ ID NO: 39 that is located upstream (5') (e.g. , in the 5' UTR) of a therapeutic nucleotide sequence.
- a recombinant viral genome is single- stranded and comprises the sequence in SEQ ID NO: 39 that is located downstream (3 ') (e.g. , downtream from (3') a polyA tail linked to the therapeutic nucleic acid, including in the 3 ' UTR) of a therapeutic nucleotide sequence.
- an inhibitory oligonucleotide may have three tandem repeats of the sequence in SEQ ID NO: 3 and three tandem repeats of the sequence in SEQ ID NO: 2.
- the tandem repeats of the sequence in SEQ ID NO: 3 may be oriented in the opposite direction of the tandem repeats of the sequence in SEQ ID NO: 2.
- the tandem repeats of the sequence in SEQ ID NO: 3 may be oriented in the sense direction and the tandem repeats of the sequence in SEQ ID NO: 2 may be oriented in the antisense direction or vice versa.
- the tandem repeats of SEQ ID NO: 3 may be upstream (5') or downstream (3 ') of the tandem repeats of the sequence in SEQ ID NO: 2.
- Each of the tandem repeats e.g.
- repeats of SEQ ID NOS: 2, 3, or any combination thereof may be separated by a linker identified by SEQ ID NO: 8 that is oriented in the sense or antisense direction.
- the viral genome comprises a sequence that is at least 90% (e.g. , at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or is 100%) identical to SEQ ID NO: 40.
- a recombinant viral genome is single-stranded and comprises the sequence in SEQ ID NO: 40 that is located upstream (5') (e.g.
- a recombinant viral genome is single-stranded and comprises the sequence in SEQ ID NO: 40 that is located downstream (3 ') (e.g. , downtream from (3') a polyA tail linked to the therapeutic nucleic acid, including in the 3 ' UTR) of a therapeutic nucleotide sequence.
- inhibitory oligonucleotide such as those described herein, as well as inhibitory oligonucleotides that share a certain degree of sequence identity (percent identity) with a reference inhibitory
- oligonucleotide e.g. , SEQ ID NO: l, SEQ ID NO:6, or SEQ ID NO: 9
- Percent identity refers to a relationship between the sequences of two or more polynucleotides (nucleic acids), as determined by comparing the sequences. Identity measures the percent of identical matches between the smaller of two or more sequences with gap alignments (if any) addressed by a particular mathematical model or computer program (e.g., "algorithms"). Identity of related molecules can be readily calculated by known methods.
- Percent (%) identity as it applies to nucleic acid sequences is defined as the percentage of nucleic acid residues in the candidate nucleic acid sequence that are identical with the residues in the nucleic acid sequence of a second sequence after aligning the sequences and introducing gaps, if necessary, to achieve the maximum percent identity. Identity depends on a calculation of percent identity but may differ in value due to gaps and penalties introduced in the calculation. Variants of a particular inhibitory oligonucleotide may have at least 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% but less than 100% sequence identity to that particular reference inhibitory oligonucleotide sequence, as determined by sequence alignment programs and parameters described herein and known to those skilled in the art.
- the inhibitory oligonucleotide includes at least one sequence that is at least 80% identical to a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32. In some embodiments, the inhibitory oligonucleotide includes at least one sequence that is at least 90% identical to a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm.
- Techniques for determining identity are codified in publicly available computer programs.
- Exemplary computer software to determine homology between two sequences include, but are not limited to, the GCG program package (Devereux, J. et al. Nucleic Acids Research, 12(1): 387, 1984), the BLAST suite (Altschul, S. F. et al. Nucleic Acids Res. 25: 3389, 1997), and FASTA (Altschul, S. F. et al. J. Molec. Biol. 215: 403, 1990).
- Other techniques include: the Smith-Waterman algorithm (Smith, T.F. et al. J. Mol. Biol.
- the present disclosure provides recombinant viral genomes in which an (at least one) inhibitory oligonucleotide is included in the viral genome.
- an (at least one) inhibitory oligonucleotide is included in the viral genome.
- two or more inhibitory oligonucleotide is included in the viral genome.
- Multipe inhibitory oligonucleotides may be located in different locations throughout the viral genome (relative to each other).
- the viral genomes typically include a therapeutic nucleotide sequence and an inhibitory
- the inhibitory oligonucleotide may be located, for example, in the 3' untranslated region (UTR) of the viral genome (see, e.g., scAAV-eGFP-3xc41 and scAAV- eGFP-3x telomere in Example 2 described below).
- the inhibitory oligonucleotide is downstream (3') relative to the therapeutic nucleotide sequence.
- the inhibitory oligocnuleotide is located downtream from (3') a polyA tail linked to the therapeutic nucleic acid.
- the inhibitory oligonucleotide is located in the 5' UTR of the viral genome.
- the inhibitory oligonucleotide is located upstream (5') of a promoter operably linked to the therapeutic nucleotide sequence. In some instances, the inhibitory oligonucleotide is located upstream (5') relative to the therapeutic nucleotide sequence.
- a recombinant viral genome may comprise an inhibitory oligonucleotide located downstream (3') (e.g. , downtream from (3') a polyA tail linked to the therapeutic nucleic acid, including in the 3' UTR) and an inhibitory oligonucleotide located upstream (5') (e.g. , in the 5' UTR) of the therapeutic nucleotide sequence.
- the recombinant viral genome may comprise inflammatory nucleic acids (e.g. , CpG oligodeoxynucleotides).
- the inflammatory nucleic acids may be located anywhere in the viral genome (e.g. , the viral ITR, the promoter, the intron, the transgene, the 5' UTR, the 3' UTR, etc. ).
- the therapeutic nucleotide sequence may comprise inflammatory nucleic acids (e.g. , CpG oligodeoxynucleotide).
- the inhibitory oligonucleotides of the present disclosure may be oriented in the sense direction and/or antisense direction in the viral genome.
- a viral genome includes 1, 2, 3, 4, or 5 copies of an inhibitory oligonucleotide (e.g., 1, 2, 3, 4, or 5 copies of SEQ ID NO: 9) in the sense direction.
- the same viral genome includes 1, 2, 3, 4, or 5 copies of the same inhibitory oligonucleotide (e.g., 1, 2, 3, 4, or 5 of reverse complement of SEQ ID NO:9) or a different oligonucleotide (e.g., reverse complement of SEQ ID NO: 1 or SEQ ID NO:5) in the antisense direction. See also Example 6 and Materials and Methods section of the Examples below.
- the recombinant viral genomes may be used, in some embodiments, to deliver (to a subject) a therapeutic nucleotide sequence of interest (e.g., a therapeutic DNA, a therapeutic RNA, and/or a therapeutic protein encoded by the nucleotide sequence).
- a therapeutic nucleotide sequence of interest e.g., a therapeutic DNA, a therapeutic RNA, and/or a therapeutic protein encoded by the nucleotide sequence.
- the recombinant viral genomes of the present disclosure are gene delivery vectors.
- the therapeutic nucleotide sequence is a gene encoding a therapeutic protein, as discussed elsewhere herein.
- a recombinant viral genome is a viral genome that is not naturally occurring.
- the viral genomes may be from adeno-associated virus (AAV), adenovirus, herpes simplex virus, varicella, variola virus, hepatitis B, cytomegalovirus, JC polyomavirus, BK polyomavirus, monkeypox virus, Herpes Zoster, Epstein-Barr virus, human herpes virus 7, Kaposi's sarcoma- associated herpesvirus, or human parvovirus B 19.
- AAV adeno-associated virus
- adenovirus herpes simplex virus
- varicella varicella
- variola virus varicella
- variola virus hepatitis B
- JC polyomavirus cytomegalovirus
- BK polyomavirus monkeypox virus
- Herpes Zoster Epstein-Barr virus
- human herpes virus 7 Kaposi's sarcoma- associated her
- a viral genome is an AAV genome.
- AAV is a small, non- enveloped virus that packages a single- stranded linear DNA genome that is approximately 5 kb long, and has been adapted for use as a gene transfer vehicle (Samulski, RJ et al., Annu Rev Virol. 2014;1(1):427-51).
- the coding regions of AAV are flanked by inverted terminal repeats (ITRs), which act as the origins for DNA replication and serve as the primary packaging signal (McLaughlin, SK et al. Virol. 1988;62(6): 1963-73; Hauswirth, WW et al. 1977;78(2):488-99).
- ITRs inverted terminal repeats
- Both positive and negative strands are packaged into virions equally well and capable of infection (Zhong, L et al. Mol Ther. 2008;16(2):290-5; Zhou, X et al. Mol Ther. 2008;16(3):494- 9; Samulski, RJ et al. Virol. 1987;61(10):3096-101).
- a small deletion in one of the two ITRs allows packaging of self-complementary vectors, in which the genome self-anneals after viral uncoating. This results in more efficient transduction of cells but reduces the coding capacity by half (McCarty, DM et al. Mol Ther. 2008;16(10): 1648-56; McCarty, DM et al. Gene Ther. 2001;8(16): 1248-54).
- the recombinant viral genomes of the present disclosure include a single-stranded nucleotide sequence.
- the viral genome is self- complementary.
- a self-complementary viral genome is a viral genome that forms an
- the viral genome is a single- stranded nucleotide sequence (e.g., ssAAV). In some embodiments, the single-stranded viral genome does not form an
- a recombinant viral genome is a single- stranded viral genome comprising an inhibitory oligonucleotide downstream (3 ') of the therapeutic nucleotide sequence (e.g. , downtream from (3') a polyA tail linked to the therapeutic nucleic acid, including in the 3 ' UTR of the viral genome).
- a recombinant viral genome is a single-stranded viral genome comprising an inhibitory oligonucleotide upstream (5') of the therapeutic nucleotide sequence (e.g. , in the 5' UTR of the viral genome).
- a single-stranded viral genome comprising an inhibitory oligonucleotide that is located upstream (5') of a therapeutic nucleotide sequence (e.g. , Factor IX) is provided as SEQ ID NO: 34.
- a therapeutic nucleotide sequence e.g. , Factor IX
- recombinant viral genome is a single-stranded viral genome comprising an inhibitory oligonucleotide upstream (5') of the therapeutic nucleotide sequence (e.g. , in the 5' UTR of the viral genome) and an inhibitory oligonucleotide downstream (3 ') of the therapeutic nucleotide sequence (e.g. , in the 3 ' UTR of the viral genome).
- a recombinant viral genome may comprise at least two different inhibitory oligonucleotides located upstream (5') of a therapeutic nucleotide sequence and at least two different inhibitory oligonucleotides sequences located downstream (3 ') of the therapeutic nucleotide sequence.
- all inhibitory oligonucleotides sequences are different in a recombinant viral genome.
- a recombinant viral genome may comprise inhibitory oligonucleotides that each comprise different multiple tandem repeats of a sequence that is at least 90% identical to a sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- an inhibitory oligonucleotide comprises multiple tandem repeats of two different sequences selected from to a sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- a recombinant viral genome comprises 1) an inhibitory
- oligonucleotide sequence that is at least 90% (e.g., at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or is identical) to SEQ ID NO: 39 located upstream (5') of a therapeutic nucleotide sequence (e.g. , in the 5' UTR) and 2) an inhibitory oligonucleotide that is at least 90% (e.g., at least 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or is identical) to SEQ ID NO: 40 that is located downstream (3 ') of a therapeutic nucleotide sequence (e.g. , in the 3 ' UTR).
- the inhibitory oligonucleotide that is at least 90% identical to SEQ ID NO: 39 may be located downstream (3 ') of a therapeutic nucleotide sequence (e.g. , in the 3 ' UTR) and the inhibitory oligonucleotide that is at least 90% identical to SEQ ID: 40 may be located upstream (5') of a therapeutic nucleotide sequence (e.g. , in the 5' UTR).
- Exemplary methods for making a single-stranded viral genome with limited self- annealing are provided in Example 8 and in the Materials and Methods section of the Examples below.
- the recombinant viral genomes of the present disclosure may comprise a therapeutic nucleotide sequence.
- a therapeutic nucleotide sequence is a nucleotide sequence (e.g., RNA or DNA) that confers a therapeutic benefit or encodes a molecule (e.g., protein) that confers a therapeutic benefit to a subject when administered in vivo.
- the therapeutic nucleotide sequence is a therapeutic RNA sequence (e.g., an RNAi molecule).
- the therapeutic nucleotide sequence is a therapeutic DNA sequence (e.g. a DNA aptamer that binds a target).
- the therapeutic nucleotide sequence encodes a therapeutic protein or peptide.
- the therapeutic nucleotide sequence may encode a wild-type (unmodified) protein to compensate for a modified (e.g., mutated or truncated) version of the protein present in a subject or to compensate for a protein the subject lacks.
- modified proteins encoded by a therapeutic nucleotide sequence include antibodies, enzymes, hormones, growth factors, cytokines and fusion proteins.
- the therapeutic nucleotide sequence is configured to replace a disease allele.
- the therapeutic nucleotide sequence could be designed to facilitate nonhomologous end joining or homologous recombination.
- the therapeutic gene sequence is a programmable nuclease.
- programmable nucleases include Cas9, Cpfl, C2c2, zinc finger, zinc finger nucleases, TALEs, TALENs, meganucleases, and fusions thereof to effector domains. Effector domains include transcriptional activators, transcription repressors, transposes, recombinases and deaminases.
- the therapeutic nucleotide sequence encodes a guide RNA (e.g. , for gene editing) or a DNA template (e.g. , for homologous recombination).
- the therapeutic nucleotide sequence itself is a therapeutic molecule.
- the nucleotide sequence is a DNA aptamer that binds a molecular target (e.g., protein target).
- a process termed SELEX systematic evolution of ligands by exponential enrichment is frequently used to select oligonucleotides from a DNA library that bind strongly to a target (Zhou J et al. Ther Nucleic Acids. 2014;3:el69).
- DNA aptamers include AS 1411, which binds to the cellular protein nucleolin and has been tested as an anticancer agent (Bates PJ et al. Exp Mol Pathol. 2009;86(3): 151-64; Soundararajan S et al. Cancer Res.
- the therapeutic nucleotide sequence encodes a sequence that is capable of reducing expression of a disease gene.
- the therapeutic nucleotide sequence is complementary to a mRNA encoding a disease gene.
- the therapeutic nucleotide sequence may be a guide RNA (e.g. , for use in CRISPR systems), a siRNA, a microRNA (miRNA), or a short hairpin RNA (shRNA).
- a therapeutic nucleotide sequence targets a mutant allele.
- the recombinant viral genome comprises a promoter operably linked to the therapeutic nucleotide sequence.
- a promoter is a control region of a nucleic acid sequence at which initiation and rate of transcription of the remainder of a nucleic acid sequence are controlled.
- a promoter may also contain sub-regions at which regulatory proteins and molecules may bind, such as RNA polymerase and other transcription factors.
- a promoter drives expression or drives transcription of the nucleic acid sequence that it regulates.
- a promoter is considered to be Operably linked' to a nucleotide sequence when it is in a correct functional location and orientation in relation to the nucleotide sequence to control ('drive') transcriptional initiation and/or expression of that sequence. Promoters may be constitutive or inducible.
- An inducible promoter is a promoter that is regulated (e.g., activated or inactivated) by the presence or absence of a particular factor.
- a suitable host cell line e.g. , HEK293T, HeLa cells and Sf9 insect cells
- One or more expression vectors e.g. viral vectors
- encoding viral components, at least one therapeutic nucleotide sequence and at least one inhibitory oligonucleotide described herein may be introduced into the suitable host cells, which can then be cultured under suitable conditions allowing for production of the viral particles.
- a helper virus can be used to facilitate replication and/or assembly of the viral particles.
- a host cell line producing one or more of essential viral components for viral genome replication and/or viral particle assembly may be used. The supernatant of the cell culture may be collected and the viral particles contained therein can be collected via routine methodology.
- a method for AAV production is provided in the Materials and Methods section below.
- the recombinant viral genomes as provided herein may be administered by intravenous, intramuscular, subretinal, intravitreal, intrathecal, intraparenchymal, and intracranial injections.
- the recombinant viral genomes is delivered by intramuscular injection.
- the recombinant viral genomes is delivered by intravenous injection.
- the recombinant viral genomes are used to transduce cells in the liver, skeletal muscle, cardiac muscle, eye (e.g., retina), central nervous system or any combination thereof.
- compositions comprising any of the recombinant viral genomes as disclosed herein.
- the compositions further comprise a pharmaceutically-acceptable excipient.
- pharmaceutically-acceptable excipients include water, saline, dextrose, glycerol, ethanol and combinations thereof. The excipient may be selected on the basis of the mode and route of administration, and standard pharmaceutical practice.
- Recombinant viral genomes may be formulated in a delivery vehicle.
- delivery vehicles include nanoparticles, such as nanocapsules and nanospheres. See, e.g., Sing, R et al. Exp Mol Pathol. 2009;86(3):215-223.
- a nanocapsule is often comprised of a polymeric shell encapsulating a drug (e.g., recombinant viral genome of the present disclosure).
- Nanospheres are often comprised of a solid polymeric matrix throughout which the drug (e.g. recombinant viral genome) is dispersed.
- the nanoparticle is a lipid particle, such as a liposome.
- nanoparticle also encompasses microparticles, such as microcapsules and microspheres.
- compositions comprising any of the recombinant viral genomes disclosed herein may be found, for example, in Remington's Pharmaceutical Sciences, 18th edition, Mack Publishing Co., Easton, Pa (1990) (incorporated herein by reference in its entirety).
- any of the recombinant viral genomes or compositions disclosed herein may be administered to a subject (e.g., mammalian subject, such as a human, mouse, rabbit, goat, or pig) to inhibit the inflammatory response (e.g., inhibit induction of the inflammatory response).
- a subject e.g., mammalian subject, such as a human, mouse, rabbit, goat, or pig
- the subject is in need of gene therapy.
- the subject may have a genetic disorder (e.g. , characterized by chromosomal abnormality and/or gene defects including mutation, truncation, insertion and deletion).
- the subject may have, may be suspected of having, or may at risk for a disease.
- the disease is an ocular disease.
- an "ocular disease” or “eye disease” is a disease or condition of the eye (e.g. , retinal disease).
- Non-limiting examples of conditions that affect the eye include Ectropion, Lagophthalmos, Blepharochalasis, Ptosis, Stye, Xanthelasma, Dermatitis, Demodex, leishmaniasis, loiasis, onchocerciasis, phthiriasis, (herpes simplex), leprosy, molluscum contagiosum, tuberculosis, yaws, zoster, impetigo,
- Dacryoadenitis Dacryoadenitis, Epiphora, exophthalmos, Conjunctivitis, Scleritis, Keratitis, Corneal ulcer /
- Choroidal haemorrhage Choroidal haemorrhage, NOS (Not Otherwise Specified), Choroidal detachment, Chorioretinal, Chorioretinal inflammation, infectious and parasitic diseases, Chorioretinitis, syphilitic, toxoplasma, tuberculosis, chorioretinal, Retinal detachment, distorted vision,
- Retinoschisis Hypertensive retinopathy, Diabetic retinopathy, Retinopathy, Retinopathy of prematurity, Age-related macular degeneration, macula, Macular degeneration, Bull's Eye Maculopathy, Epiretinal membrane, Peripheral retinal degeneration, Hereditary retinal dystrophy, Retinitis pigmentosa, Retinal haemorrhage, retinal layers, Central serous retinopathy, Retinal detachment, retinal disorders, Macular edema, macula, Retinal disorder, Diabetic retinopathy, Glaucoma, optic neuropathy, ocular hypertension, open-angle glaucoma, angle- closure glaucoma, Normal Tension glaucoma, open-angle glaucoma, angle-closure glaucoma, Floaters, Leber's hereditary optic neuropathy, Optic disc drusen, Strabismus, Ophthalmoparesis,
- the disease affects muscle.
- muscle diseases include Barth syndrome, Duchenne muscular dystrophy, Becker muscular dystrophy, myotonic dystrophy, facioscapulohumeral muscular dystrophy, mitochondrial
- Kearns-Sayre syndrome myalgia, fibromyalgia, polymyalgia rheumatica, myoma, myositis, dermatomyositis, neuromuscular disease, Kearns-Sayre syndrome, muscular dystrophy, myasthenia, congenital myasthenic syndrome, Lambert-Eaton myasthenic syndrome, myasthenia gravis, myotonia, myotonia congenita, spinal muscular atrophy, tetany,
- Suitable routes of administration include parenterally, by injection, for example, intravenously, subcutaneously, intramuscularly intrathecally, intraperitoneally,
- Oral formulations may include normally employed incipients such as, for example, pharmaceutical grades of saccharine, cellulose, magnesium carbonate and the like. These compositions may take the form of solutions, suspensions, tablets, pills, capsules, sustained release formulations or powders.
- a recombinant viral genome comprising an inhibitory
- the intramuscularly administered recombinant viral genome comprises an adeno-associated viral genome, which comprises a therapeutic nucleotide sequence and an inhibitory nucleotide sequence.
- the inhibitory nucleotide sequence comprises a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the adeno-associated viral genome administered intramuscularly comprises a therapeutic nucleotide sequence and three tandem repeats of SEQ ID NO: 1.
- the adeno-associated viral genome comprises adeno-associated viral genome
- administered intramuscularly comprises a therapeutic nucleotide sequence and three tandem repeats of SEQ ID NO: 9.
- the viral genome administered intramuscularly is expressed in muscle cells.
- the recombinant viral genomes of the present disclosure are administered intravenously to a subject. In some embodiments, the recombinant viral genomes are administered peritoneally to a subject. In some embodiments, the intravenously or peritoneally administered recombinant viral genome comprises an adeno-associated viral genome, which includes a therapeutic nucleotide sequence and an inhibitory nucleotide sequence. In some embodiments, the inhibitory nucleotide sequence comprises a nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- the adeno-associated viral genome administered intravenously or peritoneally comprises a therapeutic nucleotide sequence and three tandem repeats of SEQ ID NO: 1. In some embodiments, the adeno-associated viral genome administered intravenously or peritoneally comprises a therapeutic nucleotide sequence and three tandem repeats of SEQ ID NO: 9. In some
- the recombinant viral genome administered intravenously or peritoneally is expressed in the liver cells of a subject.
- An inflammatory response may be assessed by measuring the level of cytokine activity and/or expression in a subject.
- CXCLIO is measured.
- the level of cytokine expression and/or activity correlates with the degree of the inflammatory response.
- a subject who has received a recombinant viral genome of the present disclosure may have a reduction of or undetectable expression and/or activity levels of certain cytokines, indicative of a reduced or no inflammatory response, compared to a subject who has received a recombinant viral genome that does not include an inhibitory oligonucleotide.
- control inflammatory response for comparison is the inflammatory response elicited by a viral genome that does not comprise an inhibitory oligonucleotide as determined by the same or a substantially similar assay under the same or substantially similar conditions.
- exemplary control viral genomes for adeno-associated virus include scAAV-eGFP-3xcontrol and scAAV-eGFP used in Examples 2, 3, 4 and 5 below.
- a recombinant viral genome of the present disclosure elicits an inflammatory response in the subject that is at least 2-fold lower than a control.
- a recombinant viral genome may elicit an inflammatory response in the subject that is at least 3- fold, 4-fold, 5-fold, 6-fold, 7-fold, 8-fold, 9-fold, 10-fold, 25-fold or 50-fold lower than a control.
- a recombinant viral genome elicits an inflammatory response in the subject that is at least 10% lower than a control.
- a recombinant viral genome may elicit an inflammatory response in the subject that is at least 15%, 20%, 25%, 30%, 35%, 40%, 45%, 55%, 60%, 70%, 80% or 90% lower than a control.
- a recombinant viral genome may inhibit induction of an inflammatory response compared to a control, such that the inflammatory response is undetectable.
- a control in some embodiments, is an inflammatory response elicited in a subject by a viral genome that does not comprise an inhibitory oligonucleotide.
- a recombinant viral genome of the present disclosure reduces AAV-induced pathology in the eye of a subject compared to control.
- a control in some embodiments, is the pathology, including tissue damage and alteration in morphology, elicited in an organ by a viral genome that does not comprise an inhibitory oligonucleotide.
- a recombinant viral genome of the present disclosure may reduce loss of cone outer segments, reduce shortening of cone outer segments, or alter the morphology of cone outer segments less than a control.
- an eye receiving a recombinant viral genome of the present disclosure may have better preservation of cone outer segments and appeared morphologically closer to an eye receiving no viral genome.
- Morphology of the eye or tissues of the eye can be determined using methods known in the art, including cone arrestin staining and opsin staining.
- Retinal images from in vivo optical coherence tomography (OCT) b-scans may be used to determine damage to outer retinal lamination.
- the lengths of various types of damage e.g., retinal detachment, non-severe laminar disruption, or severe laminar damage
- OCT optical coherence tomography
- a recombinant viral genome of the present disclosure elicits less or no severe laminar damage compared to a viral genome without an inhibitory oligonucleotide.
- a recombinant viral genome (and thus the therapeutic nucleotide sequence of the recombinant viral genome) of the present disclosure is expressed in cells of the subject at a level that is at least 2-fold greater than a control.
- a recombinant viral genome may be expressed in cells of the subject at a level that is at least 3-fold, 4-fold, 5-fold, 6- fold, 7-fold, 8-fold, 9-fold, 10-fold, 25-fold or 50-fold higher than a control.
- a recombinant viral genome of the present disclosure is expressed in cells of the subject at a level that is at least 10% higher than a control.
- a recombinant viral genome may be expressed in cells of the subject at a level that is at least 15%, 20%, 25%, 30%, 35%, 40%, 45%, 55%, 60%, 70%, 80% or 90% higher than a control.
- a control in some embodiments, is the expression level of a viral genome that does not comprise an inhibitory oligonucleotide.
- a therapeutically effective amount of a recombinant viral genome of the present disclosure is administered to a subject to treat a genetic disorder, such as a muscle disorder or a liver disorder.
- a therapeutically effective amount in some embodiments, is an amount of a therapeutic nucleotide sequence (and/or a recombinant viral genomes) required to confer therapeutic effect on the subject.
- a therapeutically effective amount is an amount of inhibitory oligonucleotide required to inhibit induction of an
- a recombinant viral genome comprising a therapeutic nucleotide sequence and an inhibitory nucleotide sequence
- Effective amounts vary, as recognized by those skilled in the art, depending on the route of administration, excipient usage, and co-usage with other active agents. Effective amounts depend on the subject to be treated, including, for example, the weight, sex and age of the subject as well as the strength of the subject's immune system and/or genetic predisposition. Suitable dosage ranges are readily determinable by one skilled in the art.
- the effective amount (and thus the dosage and/or dosing schedule) of the compositions disclosed herein may also depend on the type of the viral genome, the type of therapeutic nucleotide sequence, and/or the type of inhibitory oligonucleotide.
- the therapeutically effective amount of a recombinant viral genome of the present disclosure is at least 20% lower than the therapeutically effective amount of a viral genome not comprising an inhibitory oligonucleotide.
- the therapeutically effective amount of a recombinant viral genome of the present disclosure may be at least at least 25%, 30%, 40%, 50% or 60% (but less than 100%) lower than the therapeutically effective amount of a viral genome not comprising an inhibitory oligonucleotide.
- therapeutically effective amount results in expression of an encoded therapeutic molecule at a level that is equal to or greater than (e.g., at least 5%, 10%, 20%, 30%, 40%, or 50% greater than) expression of the same encoded therapeutic molecule from a viral genome not comprising an inhibitory oligonucleotide sequence.
- a method comprising administering to a subject a recombinant viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that inhibits nucleic acid- sensing toll-like receptor (TLR) activation and/or signaling, wherein the recombinant viral genome inhibits inflammatory response to the recombinant viral genome in a target tissue.
- TLR toll-like receptor
- the inflammatory nucleic acids comprise CpG oligodeoxynucleotides .
- inhibitory oligonucleotide comprises a CCx(not-C)(not-C)xxGGG motif, wherein x is any nucleic acid.
- inhibitory oligonucleotide comprises a nucleotide sequence that is at least 90% identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- inhibitory oligonucleotide comprises at least two tandem repeats of the TTAGGG motif.
- inhibitory oligonucleotide comprises at least three tandem repeats of the TTAGGG motif.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 6 or SEQ ID NO: 9.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 1.
- inhibitory oligonucleotide comprises multiple tandem repeats of the nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 1.
- the viral genome is from adeno- associated virus (AAV), adenovirus, herpes simplex virus, varicella, variola virus, hepatitis B, cytomegalovirus, JC polyomavirus, BK polyomavirus, monkeypox virus, Herpes Zoster, Epstein-Barr virus, human herpes virus 7, Kaposi's sarcoma-associated herpesvirus, or human parvovirus B 19.
- AAV adeno- associated virus
- adenovirus herpes simplex virus
- varicella varicella
- variola virus varicella
- variola virus hepatitis B
- JC polyomavirus cytomegalovirus
- BK polyomavirus monkeypox virus
- Herpes Zoster Herpes Zoster
- Epstein-Barr virus human herpes virus 7
- Kaposi's sarcoma-associated herpesvirus or human parvovirus B 19.
- the therapeutic molecule is a therapeutic RNA or therapeutic DNA. 38. The method of paragraph 36, wherein the therapeutic molecule is a therapeutic protein or peptide.
- the therapeutically effective amount is reduced by at least 20% relative to the therapeutically effective amount of a control, wherein the control is a recombinant viral genome that does not comprise an inhibitory oligonucleotide that inhibits nucleic acid-sensing TLR activation and/or signaling.
- the therapeutic molecule is expressed in cells of the subject at a level that is equal to or greater than the expression level of a control, wherein the control is the therapeutic molecule encoded by a viral genome that does not comprise an an inhibitory oligonucleotide that inhibits nucleic acid-sensing TLR activation and/or signaling.
- a recombinant viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that inhibits nucleic acid- sensing toll-like receptor (TLR) activation and/or signaling.
- TLR toll-like receptor
- TLR is TLR3, TLR7, TLR8 or TLR9.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- inhibitory oligonucleotide comprises at least three tandem repeats of the TTAGGG motif.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 6 or SEQ ID NO: 9.
- inhibitory oligonucleotide comprises a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: l.
- inhibitory oligonucleotide comprises multiple tandem repeats of the nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 1.
- recombinant viral genome of any one of paragraphs 42-57 wherein the viral genome is from adeno-associated virus (AAV), adenovirus, herpes simplex virus, varicella, variola virus, hepatitis B, cytomegalovirus, JC polyomavirus, BK polyomavirus, monkeypox virus, Herpes Zoster, Epstein-Barr virus, human herpes virus 7, Kaposi's sarcoma-associated herpesvirus, or human parvovirus B 19.
- AAV adeno-associated virus
- adenovirus herpes simplex virus
- varicella varicella
- variola virus varicella
- variola virus hepatitis B
- JC polyomavirus cytomegalovirus
- BK polyomavirus monkeypox virus
- Herpes Zoster Herpes Zoster
- Epstein-Barr virus human herpes virus 7
- a method of treating a condition in a subject comprising administering by intramuscular injection or intravenous injection to the subject a therapeutically effective amount of a recombinant adeno-associated viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that comprises a nucleotide sequence that is identical to the nucleotide sequence identified by SEQ ID NO: 1, SEQ ID NO: 2, SEQ ID NO: 3, SEQ ID NO: 4, SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7, SEQ ID NO: 9, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 20, SEQ ID NO: 21, SEQ ID NO: 22, SEQ ID NO: 23 or SEQ ID NO: 24, to treat the condition in the subject without eliciting an inflammatory response.
- a recombinant adeno-associated viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that comprises a nucleotide sequence that is identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- a method comprising administering to a subject a recombinant viral genome comprising a therapeutic nucleic acid and an inhibitory oligonucleotide that inhibits the production of inflammatory cytokines.
- a recombinant viral genome comprising a therapeutic nucleic acid and an inhibitory oligonucleotide that inhibits the production of inflammatory cytokines.
- the recombinant viral genome further comprises at least one other inhibitory oligonucleotide that inhibits nucleic acid-sensing TLR activation and/or signaling.
- each of the inhibitory oligonucleotides comprises a nucleotide sequence that is at least 90% identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- oligonucleotides comprises SEQ ID NO: 9.
- oligonucleotides comprises SEQ ID NO: 5.
- each of the inhibitory oligonucleotides comprises a nucleotide sequence that is at least 90% identical to the nucleotide sequence of any one of SEQ ID NOS: 1-7, 9-25, or 27-32.
- a method comprising administering to an eye of a subject a recombinant viral genome comprising a therapeutic nucleotide sequence and an inhibitory oligonucleotide that inhibits nucleic acid-sensing toll-like receptor (TLR) activation and/or signaling, wherein the
- recombinant viral genome inhibits an inflammatory response to the recombinant viral genome in the eye, optionally wherein the recombinant viral genome is administered intravitreally.
- Example 1 Impact of oligonucleotides on TLR9 activation in vitro.
- This example provides data indicating that incorporating TLR9 inhibitory
- oligonucleotides into DNA can reduce TLR9-mediated inflammation.
- oligonucleotides were covalently linked to a short nucleic acid that normally activates TLR signaling to determine whether the inhibitory oligonucleotides would block TLR9 activation by the short nucleic acid.
- the inhibitory oligonucleotides tested included: c41 oligonucleotide (SEQ ID NO: 1), ODN 2088 (SEQ ID NO: 2), ODN 4084-F (SEQ ID NO: 3), ODN INH- 1 (SEQ ID NO: 4), ODN INH- 18 (SEQ ID NO: 5), ODN TTAGGG (SEQ ID NO: 6), G-ODN (SEQ ID NO: 7), ODN 2114 (SEQ ID NO: 16), ODN 4024 (SEQ ID NO: 17), ODN INH-4 (SEQ ID NO: 18), ODN INH-13 (SEQ ID NO: 19), ODN Poly-G (SEQ ID NO: 20), ODN GpG (SEQ ID NO: 21), ODN IRS-869
- oligonucleotides were generated in which ODN 2006, a CpG-containing oligonucleotide known to strongly activate TLR9, is immediately followed by a control or experimental oligonucleotide (i.e. ODN 2006 is on the 5' end). All oligonucleotides were synthesized with a phosphorothioate backbone for increased stability. A HEK293 -based reporter cell line that constitutively expresses TLR9 to measure TLR9-mediated inflammation was used. When oligonucleotides were applied at the low concentration of 0.5 ⁇ , both oligonucleotides containing control sequences induced robust inflammation compared to mock treatment (FIG.
- Oligonucleotides with an AAAAA linker (SEQ ID NO: 8) between ODN 2006 and the following sequence were also tested, and similar results were observed with reduction in inflammation (FIG. IB), suggesting the TLR9 inhibitory sequence can be distal to the inflammatory sequence.
- oligonucleotide ODN TTAGGG was also able to reduce inflammation (FIG. 1C). Taken together, these results show that incorporation of a TLR9 inhibitory oligonucleotide in a DNA inhibits inflammatory responses.
- ODN 2006 covalently linked to a control sequence gave comparable inflammation as ODN 2006 and the control ODN co-administered at the same concentration (in trans), and observed that ODN 2006-TTAGGG (fusion of SEQ ID NOS: 26 and SEQ ID NO: 6) blocked -80% of induced inflammation while co-administration of ODN 2006 (SEQ ID NO: 26) and ODN TTAGGG (SEQ ID NO: 6) only inhibited -35% of induced inflammation (FIG. 5).
- linking a TLR9 inhibitory sequence to an otherwise inflammatory nucleic acid (in cis) can be more effective at preventing inflammation than administering the TLR9 inhibitory sequence as an independent molecule (in trans).
- ODN TTAGGG did not reduce inflammatory responses by TLR7 stimulation (FIG. 6 A) or TLR2 stimulation (FIG. 6B) even at high concentration of 5uM, supporting its specificity for TLR9.
- Example 2 Engineering a self -complementary AAV vector.
- sc self-complementary AAV vector encoding enhanced green fluorescent protein (eGFP) was used, and 3 copies of c41 oligonucleotide (3 X SEQ ID NO: 1) or telomere (SEQ ID NO: 9), derived from bacteria and mammalian telomeres respectively.
- sc AAV vectors were used as they have been shown to be more efficient at triggering TLR9 activation and inducing more inflammation in the mouse liver than single-stranded (ss) AAV vectors.
- AAAAA SEQ ID NO: 8
- 3xc41 and 3x telomere sequences were placed after the polyA sequence and upstream of the right inverted terminal repeat (ITR) so they would be present in the DNA genome during viral entry, but would be absent from subsequent mRNA transcripts upon successful transduction (scAAV-eGFP-3xc41 and scAAV-eGFP-3x telomere).
- Example 3 Impact of three copies of telomere oligonucleotide (“3xtelomere” SEQ ID NO: 35) on the inflammatory response elicited by a self-complementary adeno-associated virus (scAAV) genome and on transgene expression in muscle tissue of mice in vivo.
- scAAV self-complementary adeno-associated virus
- telomere is an important tissue target for gene therapy.
- the scAAV-eGFP-3x telomere was selected for in vivo characterization as the telomere oligonucleotide is derived from human sequences and might be preferable for clinical use.
- 3xtelomere SEQ ID NO: 35
- telomere oligonucleotide could also increase viral genome expression.
- Example 4 Impact of telomere and c41 oligonucleotides on inflammatory response elicited by a self-complementary adeno-associated virus (scAAV) genome in liver tissue of mice in vivo.
- scAAV self-complementary adeno-associated virus
- Intravenous delivery of AAV is often used to transduce hepatocytes for gene therapy.
- Kupffer cells resident hepatic antigen-presenting cells
- inflammatory and innate immune responses 1-9 h later
- proinflammatory cytokines such as TNF and IL6
- immune cells such as neutrophils, macrophages and natural killer (NK) cells infiltrate the liver 2 h after AAV administration.
- scAAV-eGFP or scAAV-eGFP-3x telomere was administered via tail vein injection.
- scAAV-eGFP stimulated increased Tnf and 116 expression in the liver (approximately 3 to 10 fold, compared to saline), indicating inflammation (FIG. 3A).
- scAAV-eGFP-3x telomere showed little to no increase in inflammatory markers (FIG. 3A).
- mice were tested in subsequent experiments and scAAV-eGFP stimulated statistically significant Tnf induction in the liver compared to saline, while scAAV- eGFP-3x telomere and scAAV-eGFP-3xc41 did not (FIGS. 3B-3C), demonstrating their ability to evade eliciting inflammation in the liver. Finally, it was determined that scAAV-eGFP- 3xcontrol is not able to prevent inflammation in the liver compared to scAAV-eGFP, demonstrating that the inhibitory oligonucleotide sequence plays a role in preventing
- Example 5 Impact of three copies of telomere oligonucleotide on transgene expression by a scAAV in vivo in liver tissue.
- scAAV-eGFP or scAAV-eGFP-3x telomere was administered to mice via intravenous injection and GFP gene expression was measured in the liver 14 d later. 10.6x higher gene expression of GFP by scAAV-eGFP-3x telomere was observed (FIG. 3E), suggesting that the engineered vectors also increase transgene expression in the liver.
- Example 6 Engineering a self -complementary AAV vector encoding human factor IX (FIX) and the impact of three copies of telomere oligonucleotide on innate immune response and transgene expression in vivo in liver tissue.
- FIX human factor IX
- SEQ ID NO: 9 Three copies of SEQ ID NO: 9 were inserted into a plasmid harboring a vector genome for a self-complementary AAV vector expressing human FIX (FIG. 7A).
- An AAAAA (SEQ ID NO: 8) linker was inserted between copies of the inhibitory oligonucleotide (FIG. 7A).
- the 3x telomere sequences were placed after the left inverted terminal repeat (ITR) but before the transthyretin (TTR) promoter so they would be present in the DNA genome during viral entry, but would be absent from subsequent mRNA transcripts upon successful transduction.
- scAAV-FIX or scAAV-FIX-3x telomere (1 x 10 11 vg of each, or 1 x 10 10 vg of each, in an AAV8 capsid) was administered via tail vein injection.
- scAAV-FIX stimulated increased interferon (Ifnbl and Ifnal3) gene expression in the liver compared to saline 2 h after administration, indicating innate immune responses (FIG. 7B).
- scAAV-FIX-3x telomere showed little to no increase in interferon expression (FIG. 7B).
- mice expressed more human FIX protein in plasma compared to scAAV-FIX treatment 14 d and 28 d later (FIG. 7D).
- a 10-fold lower dose of AAV vectors did not result in interferon responses (FIG. 7C) or differences in human FIX protein expression in plasma (FIG. 7E), suggesting that the two vectors do not have inherently different potencies and that evading interferon responses enhances transgene expression.
- Example 7 Intramuscular injection of engineered single-stranded AAV vector in vivo in mice.
- AAVrh32.33 capsid and intramuscularly injected into the quadriceps muscle of mice. While dose-dependent immune responses are well-appreciated in clinical use, robust immune responses are generally not observed in mice upon AAV administration, especially ss AAV vectors which are more widely used and have a larger coding capacity. Thus, the more clinically relevant immunogenic condition was modeled in mice by utilizing AAVrh32.33 capsid (hereafter referred to as rh32.33) and intramuscular delivery.
- Tlr9 'A mice showed substantially reduced T cell responses against rh32.33 capsid and maintained stable transgene expression, and similar benefits were observed in wild-type mice by partially depleting the AAV vector genome of CpG motifs (Faust et al, J Clin Invest 123, 2994-3001 (2013)).
- ssAAV-eGFP triggered a range of CD8+ T cell responses against rh32.33 capsid, with 7 of 10 animals showing positive T cell reactivity (FIG. 8A).
- ssAAV-eGFP-3xtelo3xINH18 which harbors the inhibitory oligonucleotide designed for single-stranded vectors, showed nearly no CD8+ T cell response against rh32.33 capsid (1 of 10 animals positive) and was not statistically different from PBS treatment (FIG. 8A).
- both vectors elicited extremely high capsid-directed T cell responses (-800 - 1000 SFU/million splenocytes); while not statistically significant, the engineered vector elicited a modestly weaker T cell capsid response than the parental vector, consistent with the idea that the inhibitory oligonucleotide functions in an AAV dose-dependent manner (FIG. 8A).
- Cytotoxic T cell infiltrates have been observed in muscle biopsies of patients receiving intramuscular AAV gene therapy (Ferreira et ah , Front Immunol 5, 82 (2014); Flotte et ah , Hum Gene Ther 22, 1239- 1247 (2011)).
- Example 8 Engineering a single-stranded AAV vector.
- a single- stranded AAV vector, ssAAV-eGFP was modified by inserting 5 copies of the telomere sequence (5 X SEQ ID NO: 9) with an AAAAA (SEQ ID NO: 8) linker between each copy, followed by another 5 copies of the telomere sequence (5 X SEQ ID NO: 9) in anti-sense direction with a linker between each copy, giving ssAAV-eGFP-5xtelomere (FIG. 4). Since both positive and negative strands of the viral genome are equally likely to be packaged into a viral particle, this ensures that each packaged viral genome would have 5 copies of the telomere sequence (5 X SEQ ID NO: 9) in the correct orientation.
- ssAAV-eGFP-3xtelo3xINH18 which carries 3 copies of the telomere sequence (3 X SEQ ID NO: 9) with AAAAA (SEQ ID NO: 8) linkers, followed by 3 copies of INH-18 (3 X SEQ ID NO: 5) with linkers but in anti-sense direction was also engineered (FIG. 4).
- INH-18 (SEQ ID NO: 5) was chosen as it showed potent inhibition of TLR9-mediated inflammation in the oligonucleotide assays described above (Fig. 1A on the right).
- telomere sequence 3 X SEQ ID NO: 9 in the sense direction followed by 3 copies of INH- 18 (3 X SEQ ID NO: 5) in the anti-sense direction avoids the possibility of self-annealing via complementarity, which may hamper viral packaging.
- this design increases the probability that each packaged viral genome will have either 3 copies of the telomere sequence (3 X SEQ ID NO: 9) in the correct orientation or 3 copies of INH-18 (3 X SEQ ID NO: 5) in the correct orientation.
- the three vectors were produced as purified AAV8 viruses and total yield of viruses was measured via PCR of viral genomes.
- ssAAV-eGFP and ssAAV-eGFP-3xtelo3xINH18 gave similar yields of 3.97 x 10 13 vg and 3.60 x 10 13 vg, while ssAAV-eGFP-5xtelomere gave -10- fold lower yield of 3.11 x 10 12 vg (Table 1).
- Table 1 includes yields of indicated AAV8 viruses produced after triple transfection and purification and yield is shown as total amount of viral genomes (vg) obtained for each vector.
- ssAAV-eGFP-5xtelomere may have packaging issues and results in lower viral yields compared to the parental ssAAV-eGFP vector, while ssAAV-eGFP-3xtelo3xINH18 gives comparable viral yields.
- Example 9 Subretinal injection of engineered single-stranded AAV vector in vivo in outbred pigs.
- the eye is often described as "immune-privileged" due in part to the presence of a blood- retina barrier that limits the entry of immune cells and immune factors.
- innate and adaptive immune responses have been reported in both large animal studies and clinical trials following subretinal AAV administration with dose-dependent severity (Bainbridge et al., N Engl J Med 372, 1887-1897 (2015); Ramachandran et al., Hum Gene Ther 28, 154-167 (2017); Reichel et al., Mol Ther 25, 2648-2660 (2017)).
- AAV8 vectors dose of 4 x 10 vg per eye or with vehicle control.
- the animals received clinical examinations at weekly intervals and OCT imaging at 2 wpi and 6 wpi, and were euthanized 6 wpi. wpi, weeks post-injection; OD, oculus dextrus (right eye); OS, oculus sinister (left eye); N.A., not applicable.
- ssAAV-eGFP and ssAAV-eGFP-3xtelo3xINH18 were used and packaged in an AAV8 capsid (FIG. 4, Tables 2-4 and FIGs. 9A-9B).
- Endotoxin testing of AAV8 vectors using a limulus amebocyte lysate assay showed that both vectors were endotoxin free ( ⁇ 1 EU/ml) (Table 3).
- the percentage of full particles was determined by negatively staining AAV with 0.5% uranyl acetate and viewing under a transmission electron microscope. Empty particles show an electron-dense circle in the middle of the capsid (example shown with arrow in FIG. 9B).
- OCT optical coherence tomography
- RPE indicates retinal pigment epithelium
- IS/OS indicates inner segment/outer segment
- wpi indicates weeks post injection.
- Representative OCT b-scan measurements in a vehicle-treated eye and from two different pigs whose left eye was injected with ssAAV-eGFP (OS) and whose right eye was injected with ssAAV-eGFP-3xtelo3xINH18 (OD) are shown in Table 5.
- the outermost hyper-reflective band (located at the bottom of each b-scan, data not shown) represents the choroid/sclera. Moving inward, the next hyper-reflective band represents the RPE and the third band the IS/OS of the photoreceptors (data not shown). Severe damage were areas where both the photoreceptor and RPE layers were disrupted, non- severe damage were areas where the hyper-reflective bands for one or both of these layers were thinner and less well defined. Severe damage always surrounded the retinotomy and non-severe damage always surrounded areas of severe damage. Areas outside of the calipers had normal retinal lamination. The length of each type of damage (mm) is indicated in Table 5.
- Severe damage always surrounded the retinotomy and was defined as loss of the hyper-reflective outer retinal bands that represent the RPE and photoreceptor inner/outer segments.
- Non-severe damage was usually found surrounding areas of severe damage and was defined as areas where the outer retinal hyper-reflective bands were thinner and/or more poorly defined.
- the fluorescence images of each eye cup were used and the GFP+ boundary was then superimposed on the fundus images. This analysis demonstrated that the areas of damage correspond to the GFP+ areas.
- the area of severe damage decreased between 2 and 6 wpi (-84% smaller).
- the area with severe damage tended to remain the same.
- outer retinal laminar pathology measured by OCT was consistent with retinal histology and that engineered vector comprising inhibitory oligonucleotide sequences ameliorated outer retina laminar pathology in subretinal-injected pig eyes.
- the vehicle-injected eye showed a small area of non- severe damage surrounding a similarly sized area of severe damage around the retinotomy site and the areas of damage were reduced between 2 wpi and 6 wpi (e.g. , compare 2 wpi measurements with 6 wpi measurements for Vehicle Injection: 23588 OD in Table 5 and fundus images described above).
- retinal detachment created by the subretinal injection was noted.
- Microglia cells are the resident innate immune cells of the retina and various reports in the literature suggest that CNS microglia can respond to CpG ligands (Olson et ah , J Immunol 173, 3916-3924 (2004)).
- Ibal staining of uninjected and vehicle-injected eyes showed a ramified staining pattern outside the outer nuclear layer (ONL), consistent with resting microglia (FIG. 10B). It was found that Ibal signal increased significantly in all AAV-injected eyes (FIG.
- the engineered vector carrying the inhibitory oligonucleotide can evade eliciting undesirable innate immune and adaptive immune cell responses in the retina compared to the parental vector.
- Example 10 Engineering a single-stranded AAV vector and testing intravitreal administration.
- Single-stranded AAV vectors ssAAV2-eGFP-WPRE (SEQ ID NO: 41) and ssAAV2- eGFP-WPRE-3xtelo3xINH18 (SEQ ID NO: 42) (FIG. 14) were administered intravitreally to the eyes of African Green monkey (one vector per eye).
- the engineered vector with the inhibitory oligonucleotide reduced the immune response relative to the control vector.
- Animal A983 which received ssAAV-eGFP-WPRe in its OS eye had several signs of ocular inflammation by opthalmic examination, while the vehicle-injected OD eye did not (Table 7).
- Table 7 Non-limiting example of inflammation as measured by clinical examinations of the
- the eye with control treatment showed a moderate anterior chamber cell score with 11-20 cells, mild aqueous flare that was just detectable (i.e., the Tyndall effect was barely discernable; the intensity of the light beam in the anterior chamber was less than the intensity of the slit beam as it passes through the lens), moderated fibrin strands (large fibrinous strands/clots, or more than 5 small fibrinous strands), mild injection of tertiary vessels and minimal to moderate injection of the secondary vessels observed for the iris hyperemia score, moderate lens capsule deposits, and mild lens opacity or vacuoles /clefts between the lens fibers, and fundus detail was visible.
- Example 11 Additional engineering of single-stranded AAV vector and testing by intramuscular injection in vivo in mice.
- ssAAV-eGFP-3xtelo3xINH18 (packaged in rh32.33 capsid) showed reduced immune responses compared to ssAAV-eGFP and this occurs in an AAV dose-dependent fashion (FIG. 8A).
- 3 copies of 4084-F (3 X SEQ ID NO: 3) were added with AAAAA (SEQ ID NO: 8) linkers followed by 3 copies of 2088 ( 3 X SEQ ID NO: 2) with linkers but in the anti- sense direction between the left ITR and promoter (the 5' untranslated region) of ssAAV-eGFP-3xtelo3xINH18 and term this ssAAV-eGFP-double.
- This vector packaged in rh32.33 capsid, showed further reduction immune responses in mice upon intramuscular injection.
- inhibitory oligonucleotides in various locations of the vector genome to reduce immune responses, and it is possible to further reduce immune responses by using more inhibitory oligonucleotides in combination.
- mice Male, 7-9 weeks old were purchased from the Jackson Laboratory.
- scAAV-eGFP was purchased from Cell Biolabs (VPK-430) and has been previously described (Gray, JT et al., Methods Mol. Biol. 2011 ;807:25-46).
- eGFP enhanced green fluorescent protein
- CMV cytomegalovirus
- c41 oligonucleotide (5'- TGGCGCGCACCCACGGCCTG-3'; SEQ ID NO: 1) derived from Pseudomonas aeruginosa and telomere oligonucleotide (5 '-TTTAGGGTTAGGGTTAGGGTTAGGG-3 '; SEQ ID NO: 9; initial T nucleotide is optional for function) derived from mammalian telomeres have been described (Gursel, I et al., J Immunol. 2003;171(3): 1393-400; Kaminski, JJ et al., J Immunol.
- telomere oligonucleotide manufactured by Invivogen, catalog code: tlrl-ttag harbored an additional T (in bold) compared to published studies and thus was included in the sequence.
- Invivogen removed the additional T in their manufactured telomere oligonucleotide (catalog code: tlrl-ttagl51).
- control (5'- GCTAGATGTTAGCGT-3 '; SEQ ID NO: 34) was used as a negative control sequence that does not inhibit TLR9 activation (Invivogen, catalog code: tlrl-2088c).
- scAAV-eGFP sequences were inserted into the unique Spel site found immediately 5' of the right ITR.
- a unique Clal site was created immediately 5' of the inserted sequences, thus allowing Clal/Spel sub-cloning of sequences.
- 3 copies of c41, telomere, or control oligonucleotide were inserted, separated by AAAAA (SEQ ID NO: 8) linkers, giving scAAV-eGFP-3xc41, scAAV-eGFP-3x telomere and scAAV-eGFP-3x control, respectively.
- one copy of telomere was inserted, with an AAAAA linker (SEQ ID NO: 8), giving scAAV-eGFP-lx telomere (data not shown).
- Self-complementary vectors were packaged into AAV2 (Vigene Biosciences) by triple transfection of HEK293 cells and purified using iodixanol gradient ultracentrifugation and then concentrated to 500ul using Amicon Ultra-15 columns in PBS.
- the purified viruses were titered by qPCR using primers derived from ITR and an AAV standard. The final yield of the viruses ranged from 0.5 - 3 x 10 13 vg.
- scAAV.FIX (Martino, AT et al. Blood 2011;117(24):6459-68) expressed human factor IX (FIX) under the control of a liver-specific transthyretin (TTR) mouse promoter and included a bovine growth hormone (bGH) polyA sequence.
- TTR liver-specific transthyretin
- bGH bovine growth hormone
- GTVC Gene Transfer Vector Core
- MEEI Ear Infirmary
- ssAAV-eGFP The single- stranded AAV vector ssAAV-eGFP has been previously described (Xiong, W. et al., Clin Invest, 2015; 125(4): 1433-45) and was originally obtained from the Harvard DF/HCC DNA Resource Core (clone ID: EvNO00061595).
- ssAAV-eGFP contained a CMV enhancer/promoter, human ⁇ -globin intron, eGFP, and ⁇ -globin polyA sequence.
- Kpnl-5x telomere(sense)-5x telomere(anti-sense)-NheI was inserted immediately 5' of the Xbal site adjacent to the right ITR.
- AAAAA SEQ ID NO: 8 was used as a linker between copies of the telomere sequence (SEQ ID NO: 9).
- telomere sequence SEQ ID NO: 9 or INH-18 (SEQ ID NO: 5) was added as single- stranded AAV vectors have an equal chance of packaging positive or negative strands of the viral genome, thus ensuring that all packaged AAV genomes will carry copies of the telomere sequence (SEQ ID NO: 9) or INH-18 (SEQ ID NO: 5) in the right orientation.
- Spel-3x 4084-F(sense)-3x2088(anti-sense)-SpeI were additional inserted into the unique Spel site found after the left ITR and before the promoter in ssAAV- eGFP-3xtelo3xINH18, and this vector was termed ssAAV-eGFP-double (FIG. 12).
- Single- stranded vectors were packaged into AAV8 (subretinal pig studies) or AAVrh32.33 (intramuscular mouse studies) and purified by the core facility Gene Transfer Vector Core (GTVC) at Massachusetts Eye and Ear Infirmary (MEEI).
- GTVC Gene Transfer Vector Core
- the viral titers were determined by digital PCR using primers against the CMV promoter and total yield for each vector was calculated by multiplying viral titer (vg/ml) by volume.
- the purity of vector preps was evaluated by running 1 x 10 10 vg (viral genome) on an SDS-PAGE gel.
- vector preps had ⁇ lEU/ml of endotoxin using a limulus amebocyte lysate assay (ToxinSensor Chromogenic LAL Endotoxin Assay Kit, Genscript). No significant differences in viral yield (viral titer x volume) were observed between parental vectors and corresponding engineered vectors for >20 purifications, suggesting that insertion of the described sequences do not hamper viral packaging.
- ssAAV-eGFP-WPRE contains a CMV promoter and woodchuck hepatitis virus posttranscriptional regulatory element (WPRE) for eGFP expression.
- WPRE woodchuck hepatitis virus posttranscriptional regulatory element
- Kpnl-3x telomere(sense)-3xINH-18(anti-sense) was inserted immediately 5' of the Xhol site just upstream of the right ITR (FIG. 13).
- the parental vector and engineered vector were packaged into AAV2 and purified by cesium purification.
- a HEK293-based reporter cell line stably expressing human TLR9 and an inducible SEAP (secreted embryonic alkaline phosphatase) reporter gene was obtained (HEK-Blue hTLR9, Invivogen).
- the SEAP gene is under the control of the IFN- ⁇ minimal promoter fused to five NF-kB and AP-1 binding sites. Stimulation with a TLR9 ligand such as ODN 2006 activates NF-kB and AP-1 and therefore induces the production of SEAP, which can subsequently be measured to determine the amount of inflammation.
- All designed single-stranded DNA oligonucleotides were synthesized with a phosphorothioate backbone for increased stability (IDT).
- ODN 2006 was directly linked to indicated sequence with no intervening nucleotides, or with a AAAAA (SEQ ID NO: 8) linker sequence.
- ODN Control (15nt) and ODN Control (24nt) were 5' - TCC TGA GCT TGA AGT - 3' (SEQ ID NO: 43) and 5' - TTA TTA TTA TTA TTA TTA TTA - 3' (SEQ ID NO: 44) respectively, and the two control sequences were selected to match the approximate range of lengths of the various TLR9 inhibitory oligonucleotides.
- oligonucleotides were incubated with 6xl0 4 HEK293-TLR9 cells in 200ul of DMEM growth media per well in 96-well flat bottom plates for 18 h, and 50ul media was aspirated and incubated with lOOul HEK-Blue Detection media (Invivogen) for 4-6 h at 37°C and then absorbance at 639nm was read on a plate reader.
- HEK293 reporter cell lines stably expressing inducible SEAP reporter gene and human TLR7 (1 x 10 5 cells) or TLR2 (6 x 10 4 cells) (both from Invivogen) were stimulated with 1 g/ml of Gardiquimod or 100 ng/ml of FSL-2 (both from Invivogen) respectively with or without control oligonucleotides or TLR9 inhibitory oligonucleotides for 18 h and SEAP activity was measured.
- RNAlater solution (Thermo Scientific). The muscle tissues were subjected to RNA extraction, reverse transcription, and qPCR as described in the liver studies. For GFP expression studies, the quadricep was harvested 28 d later.
- mice were injected intramuscularly with 50ul PBS or AAVrh32.33 viruses (10 10 or 10 11 vg per animal, single- stranded vector) in the quadriceps. 21 d later, the animals were sacrified and spleens were harvested (see ELISPOT below) and muscle tisses were fixed in 10% formalin overnight and transferred to 70% ethanol. For histology, muscle samples were processed at DF/HCC Specialized Histopathology Services (SHS) and Beth Israel
- BIDMC Deaconess Medical Center histology core facilities, embedded in paraffin, and stained for CD8 (1:500, D4W2Z, CST) and granzyme B (1:500, polyclonal, catalog # AF1865, R&D).
- Antibodies were tagged with AlexaFluor 488 Tyramide (B40957, Thermo) or AlexaFluor 647 Tyramide (B40958, Thermo). All immunohistochemistry was performed on the Leica Bond automated staining platform using the Leica Biosystems Refine Detection Kit with citrate antigen retrieval. Muscle sections were also stained for GFP by immunohistochemistory (IHC) with IHC chromogen substrate DAB (3,3'-diaminobenzidine).
- IHC immunohistochemistory
- DAB 3,3'-diaminobenzidine
- Spleens were harvested from C57BL/6 mice injected i.m. with AAVrh32.33 single- stranded vectors 21 d post-injection. Spleens were passed through a 70 ⁇ cell strainer
- ELISPOT plates were evaluated in blinded fashion (ZellNet Consulting, Inc., Fort Lee, NJ) using an automated Elispot reader system (KS ELISpot reader, Zeiss, Thornwood, USA) with KS
- RNAlater solution (OMEGA Bio-Tek).
- RNA-to-cDNA kit Similar amounts of RNA were reverse transcribed into cDNA with a high-capacity RNA-to-cDNA kit (Thermo Scientific) and similar amounts of cDNA were assayed with quantitative PCR (qPCR) using TaqMan Fast Advanced Master Mix (Thermo Scientific) and commercially available pre-designed primers/probes with FAM reporter dye for the indicated target genes (IDT).
- qPCR quantitative PCR
- qPCR quantitative PCR
- IDT FAM reporter dye for the indicated target genes
- Expression level for each gene was calculated by normalizing against the housekeeping genes Actb or Gapdh using the AACT method and expressed as fold levels compared to saline-injected mice. All qPCR reactions were run on a realplex4 Mastercycle (Eppendorf). For GFP expression studies, the liver was harvested 14 d later.
- FIX expression For human FIX expression, plasma (EDTA) were obtained 14 d and 28 d after AAV administration and human FIX expression was quantified by analyzing diluted plasma samples using an ELISA kit specific for human factor IX (abl88393, Abeam). Plasma from PBS-injected mice gave signals similar to that of a blank control, demonstrating specificity of the kit for human factor IX.
- the kit had a sensitivity of at least 0.78 ng/ml.
- IV line inserted in the ear vein was used to deliver IV fluids (Lactated Ringers Solution with or without 5% dextrose; 10 - 15 mL/kg/h) to maintain blood pressure and normal glycemic levels (60 - 140 mg/dL).
- IV fluids Lacated Ringers Solution with or without 5% dextrose; 10 - 15 mL/kg/h
- body temperature was monitored every 30 min with a rectal thermometer and maintained via a heated procedure table.
- Heart and respiration rates and oxygen saturation were recorded every 10 min throughout the procedures and anesthesia adjusted to maintain a normal range for these physiological parameters.
- a vitreoretinal surgical approach was used to gain access to the subretinal space (between the retina and the pigment epithelium at the back of the eye) and to deposit an inoculum containing AAV in final formulation buffer (FFB) or FFB alone (vehicle).
- FFB final formulation buffer
- FFB alone vehicle
- a lateral canthotomy was performed to increase exposure in the surgical field.
- two 25 g trocars were placed at 1.5 mm posterior to the limbus; one in superior-nasal and the other in inferior-nasal quadrant.
- An anterior chamber fluid paracentesis was performed to make space for the injected volume.
- a light pipe was inserted into one trochar to help visualize the retina.
- a 41 gauge subretinal cannula needle was placed through the other trochar and used to make a local retinal detachment (bleb) followed by injection of inoculum (-75 ⁇ ).
- Either AAV8.GFP.io2 or AAV8.GFP was injected into OD or OS of two pigs (4 eyes) in each of three surgery sessions. After the injection, the light pipe, needle and the trochars were removed (they are self-sealing). The lateral canthotomy was sutured closed with 4-0 Nylon. Antibiotic and steroid ointment was placed topically at the end of the surgery.
- each eye was scored for inflammation of the eye/retina using the SUN classification 10 .
- the retinal surgeon performing the injections was blinded to the test article, and similarly, clinical examinations and scoring of inflammation were performed blinded.
- OCT ocular coherence tomography
- the shape, location and size of these two types of damage were measured using the software provided with the OCT system. Specifically, calipers were placed over severe and flanking non-severe damage in b-scans across the fundus and their areas were computed and summed over the entire extent of damage. These areas were then superimposed on the fundus image and the areas of damage compared to the areas of GFP expression (see below).
- Beuthanasia (390 mg pentobarbital sodium, 50 mg phenytoin sodium/ml; 1 mL/5 kg), and their eyes were enucleated.
- the cornea and lens were removed and the eyecup was dissected and fixed in 4% paraformaldehyde in PBS for 1 h at room temperature and then washed in PBS.
- Wholemount retinas were examined using a low power fluorescent microscope (Olympus MVX10) and the region of GFP+ expression was located and images acquired and plotted on the fundus images relative to the blood vessels and optic nerve head.
- the retina was dissected so that the piece used for histology included all of the GFP+ region, as well as GFP- flanking regions.
- the pig retinal tissue was cryoprotected in graded sucrose solutions up to 30% sucrose in PBS, then embedded in a 1: 1 mixture of 30% sucrose and optimal cutting temperature (OCT) compound (Tissue-Tek) followed by cryosectioning on a Leica
- CM3050S Leica Microsystems. Transverse sections of retinal tissue were cut at 20 ⁇ . For immunohistochemistry, tissue sections were first blocked with 5% donkey serum (if the secondary antibody was donkey-origin) or 5% goat serum (if the secondary antibody was goat-origin) in PBS with 0.1% Triton X-100 for 1 h at room temperature.
- Sections were then stained overnight at 4 °C in blocking solution with primary antibodies against red- green opsin (1:600, AB5405, EMD Millipore), human cone arrestin 11 (1: 10000), Ibal (1:200, ab5076, Abeam) and CD8 (1:200, MCA1223GA, Bio-Rad), followed by staining for 2 h at room temperature with goat anti-rabbit, donkey anti-mouse, or donkey anti-goat AlexaFluor 594-labeled secondary antibodies (111-585-144, 715-585-150 and 705-586- 147, all from Jackson ImmunoResearch) used at 1: 1000 in PBS.
- Tissues were lastly stained with 4',6-diamidino-2-phenylindole (DAPI) for 5 min and mounted using Fluoromount-G (Southern Biotech).
- DAPI 4',6-diamidino-2-phenylindole
- the slides were examined using a LSM710 laser scanning confocal microscope (Zeiss) with a 40x oil-immersion objective, and image processing was
- African Green monkeys received intravitreal injections of 100 ul of ssAAV-eGFP- WPRE or ssAAV-eGFP-WPRE-3xtelo3xINH18 (both packaged in AAV2 capsid) in one eye, and the vehicle control (buffer) in the other eye. Ophthalmic examinations by slit lamp biomicroscopy and retinoscopy, as well as fundus imaging (both color photos and fluorescence photos), were performed at various time points. Vitreous haze was graded on a scale of 0 to 4 using the Nussenblatt scale, and anterior chamber cells and aqueous flare were graded using a modified Packett-McDonald scoring system.
- OCT optical coherence tomography
- ODN TTAGGG ODN TTAGGG
- AAAAA SEQ ID NO: 8
- ODN 4348 ODN 4348:
- TCGTATCCTGGAGGGGAAG SEQ ID NO: 27.
- TAATATCCTGGAGGGGAAG SEQ ID NO: 28.
- ODN A CCTATCCTGGAGGGGAAG (SEQ ID NO: 29).
- ssAAV-eGFP-3xtelo3xINH18 inhibitory oligonucleotide in underlined and bold text: tgcgcgctcgctcactgaggccgcccgggcaaagcccgggcgtcgggcgacctttggtcgcccggcctcagtgagcgagcgag cgcgcagagagggagtggccaactccatcactaggggttccttgtagttaatgattaacccgccatgctacttatctacgtagccatgctcta gcggcctctctgcataaataaaaaattagtcagccatgagcttggcccattgcatacgttgtgtatccatatcataatatgtacatttatat tggctcatgtccaa
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Molecular Biology (AREA)
- Organic Chemistry (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Plant Pathology (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Virology (AREA)
- Immunology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201880072584.XA CN111511918A (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| AU2018364542A AU2018364542A1 (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| EP18842597.9A EP3707262A1 (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| US16/762,356 US11981911B2 (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| JP2020525991A JP2021502381A (en) | 2017-11-08 | 2018-11-08 | Compositions and Methods for Inhibiting Viral Vector-Induced Inflammatory Responses |
| KR1020207015916A KR20200085812A (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory response |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762583449P | 2017-11-08 | 2017-11-08 | |
| US62/583,449 | 2017-11-08 | ||
| US201762595433P | 2017-12-06 | 2017-12-06 | |
| US62/595,433 | 2017-12-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019094548A1 true WO2019094548A1 (en) | 2019-05-16 |
Family
ID=65244583
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2018/059756 WO2019094548A1 (en) | 2017-11-08 | 2018-11-08 | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11981911B2 (en) |
| EP (1) | EP3707262A1 (en) |
| JP (1) | JP2021502381A (en) |
| KR (1) | KR20200085812A (en) |
| CN (1) | CN111511918A (en) |
| AU (1) | AU2018364542A1 (en) |
| WO (1) | WO2019094548A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11339396B2 (en) | 2016-06-08 | 2022-05-24 | President And Fellows Of Harvard College | Engineered viral vector reduces induction of inflammatory and immune responses |
| WO2022187679A1 (en) * | 2021-03-04 | 2022-09-09 | Kriya Therapeutics, Inc. | Viral vector constructs incorporating dna for inhibiting toll like receptors and methods of using the same |
| US11981911B2 (en) | 2017-11-08 | 2024-05-14 | President And Fellows Of Harvard College | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20230076269A (en) | 2021-11-24 | 2023-05-31 | 김채린 | Remove sulfur dioxide using eggshells |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050239733A1 (en) * | 2003-10-31 | 2005-10-27 | Coley Pharmaceutical Gmbh | Sequence requirements for inhibitory oligonucleotides |
| US20110070241A1 (en) * | 2009-06-30 | 2011-03-24 | Duke University | Methods for modulating immune responses to aav gene therapy vectors |
Family Cites Families (82)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL104314A0 (en) | 1992-01-07 | 1993-05-13 | Novo Nordisk As | Human kunitz-type protease inhibitor and variants thereof,their production and pharmaceutical compositions containing them |
| US5489508A (en) | 1992-05-13 | 1996-02-06 | University Of Texas System Board Of Regents | Therapy and diagnosis of conditions related to telomere length and/or telomerase activity |
| US5643890A (en) | 1995-01-31 | 1997-07-01 | The Board Of Regents Of The University Of Nebraska | Synthetic oligonucleotides which mimic telomeric sequences for use in treatment of cancer and other diseases |
| CA2291483C (en) | 1997-06-06 | 2012-09-18 | Dynavax Technologies Corporation | Immunostimulatory oligonucleotides, compositions thereof and methods of use thereof |
| US6551795B1 (en) | 1998-02-18 | 2003-04-22 | Genome Therapeutics Corporation | Nucleic acid and amino acid sequences relating to pseudomonas aeruginosa for diagnostics and therapeutics |
| PT1181304E (en) | 1999-04-08 | 2008-01-23 | Antisoma Res Ltd | Antiproliferative activity of g-righ oligonucleotides and method of using same to bind to nucleolin |
| MXPA02003108A (en) | 1999-09-25 | 2003-10-14 | Univ Iowa Res Found | Immunostimulatory nucleic acids. |
| MXPA00011713A (en) | 2000-11-28 | 2002-05-31 | Tgt Lab S A De C V | Re-combining viral and non-viral vectors containing the human gene of the urokinase plasminogen activator and its benefit in the treatment of diverse types of hepatic, renal, pulmonary, pancreatic and cardiac fibrosis and hypertrophic scars. |
| AU2002241952A1 (en) | 2001-01-22 | 2002-07-30 | Genta Incorporated | Cell-proliferative disorder treatments using cre decoy oligomers, bcl-2 antisense and hybrid oligomers |
| WO2003027313A2 (en) | 2001-09-24 | 2003-04-03 | The Government Of The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | SUPPRESSORS OF CpG OLIGONUCLEOTIDES AND METHODS OF USE |
| EP2368431A1 (en) | 2002-04-04 | 2011-09-28 | Coley Pharmaceutical GmbH | Immunostimulatory G,U-containing oligoribonucleotides |
| US7357928B2 (en) | 2002-04-08 | 2008-04-15 | University Of Louisville Research Foundation, Inc. | Method for the diagnosis and prognosis of malignant diseases |
| AU2003243409A1 (en) | 2002-06-05 | 2003-12-22 | Coley Pharmaceutical Group, Inc. | Method for treating autoimmune or inflammatory diseases with combinations of inhibitory oligonucleotides and small molecule antagonists of immunostimulatory cpg nucleic acids |
| AU2003263963A1 (en) | 2002-08-01 | 2004-02-23 | The Government Of The United States Of America As Represented By The Secretary Of The Department Of | Method of treating inflammatory arthropathies with suppressors of cpg oligonucleotides |
| US8043622B2 (en) | 2002-10-08 | 2011-10-25 | The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Method of treating inflammatory lung disease with suppressors of CpG oligonucleotides |
| US7217807B2 (en) | 2002-11-26 | 2007-05-15 | Rosetta Genomics Ltd | Bioinformatically detectable group of novel HIV regulatory genes and uses thereof |
| US20050013812A1 (en) | 2003-07-14 | 2005-01-20 | Dow Steven W. | Vaccines using pattern recognition receptor-ligand:lipid complexes |
| WO2005059517A2 (en) | 2003-12-16 | 2005-06-30 | University Of Massachusetts | Toll-like receptor assays |
| CN1989439B (en) | 2004-05-06 | 2010-12-29 | 美国政府健康及人类服务部 | Methods and compositions for the treatment of uveitis |
| CA2566519C (en) | 2004-05-14 | 2020-04-21 | Rosetta Genomics Ltd. | Micrornas and uses thereof |
| CN101031658A (en) | 2004-05-26 | 2007-09-05 | 罗塞塔金诺米克斯有限公司 | Viral and viral associated mirnas and uses thereof |
| NZ553581A (en) | 2004-09-01 | 2011-04-29 | Dynavax Tech Corp | Methods and compositions for inhibition of innate immune responses and autoimmunity using immunoregulatory polynucleotides with a TGC sequence |
| JP2008000001A (en) | 2004-09-30 | 2008-01-10 | Osaka Univ | Immune stimulating oligonucleotide and use in pharmaceutical |
| CA2591582A1 (en) | 2004-12-17 | 2006-06-22 | Dynavax Technologies Corporation | Methods and compositions for induction or promotion of immune tolerance |
| US20070077231A1 (en) | 2005-09-30 | 2007-04-05 | Contag Christopher H | Immune effector cells pre-infected with oncolytic virus |
| WO2007047396A2 (en) | 2005-10-12 | 2007-04-26 | Idera Pharmaceuticals, Inc. | Immune regulatory oligonucleotide (iro) compounds to modulate toll-like receptor based immune response |
| WO2007120368A2 (en) | 2006-01-09 | 2007-10-25 | The Regents Of The University Of California | Immunostimulatory combinations for vaccine adjuvants |
| US8298818B2 (en) | 2006-04-28 | 2012-10-30 | University Of Florida Research Foundation, Inc. | Self-complementary adeno-associated virus having a truncated CMV-chicken β-actin promoter |
| JP2009540017A (en) | 2006-06-13 | 2009-11-19 | ベイヒル セラピューティクス インコーポレーティッド | Polynucleotide therapy |
| US8377898B2 (en) | 2006-10-12 | 2013-02-19 | Idera Pharmaceuticals, Inc. | Immune regulatory oligonucleotide (IRO) compounds to modulate toll-like receptor based immune response |
| US20110033448A1 (en) | 2006-12-15 | 2011-02-10 | Michel Gilliet | Inhibitors of LL-37 Mediated Immune Reactivity to Self Nucleic Acids |
| EP2126116A2 (en) | 2007-03-01 | 2009-12-02 | Rosetta Genomics Ltd | Methods for distingushing between lung squamous carcinoma and other non smallcell lung cancers |
| WO2009006141A2 (en) | 2007-07-05 | 2009-01-08 | Bausch & Lomb Incorporated | Authority to read as follows: compositions and methods for treating or controlling infections of the eye a sequelae thereof |
| US7879812B2 (en) * | 2007-08-06 | 2011-02-01 | University Of Iowa Research Foundation | Immunomodulatory oligonucleotides and methods of use therefor |
| AU2008286735A1 (en) | 2007-08-15 | 2009-02-19 | Idera Pharmaceuticals, Inc. | Toll like receptor modulators |
| EP2209896B1 (en) | 2007-10-26 | 2017-03-01 | Dynavax Technologies Corporation | Methods and compositions for inhibition of immune responses and autoimmunity |
| WO2009089401A2 (en) | 2008-01-10 | 2009-07-16 | Bausch & Lomb Incorporated | Compositions comprising toll-like receptor or coreceptor antagonists and methods for treating or controlling ocular allergy using same |
| WO2009089399A2 (en) | 2008-01-10 | 2009-07-16 | Bausch & Lomb Incorporated | Compositions comprising toll-like receptor or coreceptor antagonists and methods for ocular neuroprotection |
| WO2009143292A2 (en) | 2008-05-21 | 2009-11-26 | The Government Of The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Method of treating pneumoconiosis with oligodeoxynucleotides |
| JP5415724B2 (en) | 2008-08-05 | 2014-02-12 | 長春華普生物技術有限公司 | A kind of oligonucleotide and its application |
| US8030289B2 (en) | 2008-08-06 | 2011-10-04 | Changchun Huapu Biotechnology Co., Ltd. | Oligonucleotide and use thereof |
| US8933011B2 (en) | 2008-09-24 | 2015-01-13 | The Provost, Fellows And Scholars Of The College Of The Holy And Undivided Trinity Of Queen Elizabeth Near Dublin | Treatment of preterm labor with toll-like receptor 9 antagonists |
| GB0906130D0 (en) | 2008-10-03 | 2009-05-20 | Procrata Biosystems Ltd | Transcription factor decoys |
| MX2011003625A (en) | 2008-10-06 | 2011-05-31 | Idera Pharmaceuticals Inc | Use of inhibitors of toll-like receptors in the prevention and treatment of hypercholesterolemia and hyperlipidemia and diseases related thereto. |
| US8053422B2 (en) | 2008-12-04 | 2011-11-08 | The United States Of America As Represented By The Department Of Health And Human Services | Anti-cancer oligodeoxynucleotides |
| US8728458B2 (en) | 2009-04-30 | 2014-05-20 | The Regents Of The University Of California | Combination anti-HIV vectors, targeting vectors, and methods of use |
| JP6275380B2 (en) | 2009-06-01 | 2018-02-07 | イデラ ファーマシューティカルズ インコーポレイテッドIdera Pharmaceuticals, Inc. | Enhancement of autoimmune and inflammatory disease treatment by immunoregulatory oligonucleotide (IRO) antagonists of TLR7 and TLR9 |
| EP2451974A2 (en) | 2009-07-08 | 2012-05-16 | Idera Pharmaceuticals, Inc. | Oligonucleotide-based compounds as inhibitors of toll-like receptors |
| CN101712957B (en) | 2009-11-25 | 2011-11-23 | 中国人民解放军第三军医大学第一附属医院 | Toll-like receptor 9 (TLR9) blocking agent and application thereof |
| US8771669B1 (en) | 2010-02-09 | 2014-07-08 | David Gordon Bermudes | Immunization and/or treatment of parasites and infectious agents by live bacteria |
| EP3444346B1 (en) | 2010-04-23 | 2022-07-27 | University of Massachusetts | Aav-based treatment of cholesterol-related disorders |
| SG186380A1 (en) | 2010-06-16 | 2013-01-30 | Dynavax Tech Corp | Methods of treatment using tlr7 and/or tlr9 inhibitors |
| JP5921535B2 (en) | 2010-06-16 | 2016-05-24 | ディナバックス テクノロジーズ コーポレイション | Methods of treatment using TLR7 and / or TLR9 inhibitors |
| GB201014026D0 (en) | 2010-08-20 | 2010-10-06 | Ucl Business Plc | Treatment |
| WO2012051491A1 (en) | 2010-10-14 | 2012-04-19 | The United States Of America, As Represented By The Secretary National Institutes Of Health | Compositions and methods for controlling neurotropic viral pathogenesis by micro-rna targeting |
| CN107012149A (en) | 2010-11-19 | 2017-08-04 | 艾德拉药物股份有限公司 | Regulate and control immunomodulatory oligonucleotide (IRO) compound of the immune response based on TOLL sample acceptors |
| CN103889441A (en) * | 2011-09-29 | 2014-06-25 | 伊丽莎白女王在都柏林附近神圣不可分割的三一学院教务长、研究员、学者及董事会其他成员 | Compositions and methods for the treatment of degenerative retinal conditions |
| US20140378531A1 (en) | 2012-02-01 | 2014-12-25 | New York University | Inhibition of pattern recognition receptors in pancreatic cancer treatment using tlr inhibitors |
| AR091569A1 (en) | 2012-06-28 | 2015-02-11 | Intervet Int Bv | TOLL TYPE RECEIVERS |
| EP3738974A1 (en) | 2012-09-28 | 2020-11-18 | The University of North Carolina at Chapel Hill | Aav vectors targeted to oligodendrocytes |
| CN102925485B (en) | 2012-11-09 | 2014-07-09 | 中国农业科学院兰州兽医研究所 | Preparation method of dually-expressed AAV (Adeno Associated Virus) recombinant virus |
| AU2012395641B2 (en) | 2012-11-29 | 2019-10-03 | Sbi Biotech Co., Ltd. | Inhibitory oligonucleotide and use thereof |
| WO2014105870A1 (en) | 2012-12-27 | 2014-07-03 | Sierra Sciences, Inc. | Enhancing health in mammals using telomerase reverse transcriptase gene therapy |
| US9260719B2 (en) | 2013-01-08 | 2016-02-16 | Idera Pharmaceuticals, Inc. | Immune regulatory oligonucleotide (IRO) compounds to modulate toll-like receptor based immune response |
| US20140271550A1 (en) | 2013-03-14 | 2014-09-18 | The Trustees Of The University Of Pennsylvania | Constructs and Methods for Delivering Molecules via Viral Vectors with Blunted Innate Immune Responses |
| WO2015089114A1 (en) | 2013-12-09 | 2015-06-18 | Bullet Biotechnology, Inc. | Specific virus-like particle-cpg oligonucleotide vaccines and uses thereof |
| US10441654B2 (en) | 2014-01-24 | 2019-10-15 | Children's Hospital Of Eastern Ontario Research Institute Inc. | SMC combination therapy for the treatment of cancer |
| WO2015183943A2 (en) | 2014-05-27 | 2015-12-03 | Trustees Of Boston University | Inhibitors of fibroproliferative disorders and cancer |
| EP2982756A1 (en) | 2014-08-04 | 2016-02-10 | Berlin Cures Holding AG | Aptamers for use against autoantibody-associated diseases |
| US20170333565A1 (en) | 2014-10-31 | 2017-11-23 | H. Lee Moffitt Cancer Center And Research Institute, Inc. | Tlr9 targeted cytotoxic agents |
| WO2016130832A1 (en) | 2015-02-13 | 2016-08-18 | Idera Pharmaceuticals, Inc. | Toll-like receptor 9 antagonist and methods of use thereof |
| US11156599B2 (en) | 2015-03-19 | 2021-10-26 | The Johns Hopkins University | Assay for telomere length regulators |
| WO2016168656A1 (en) | 2015-04-15 | 2016-10-20 | Ji Hoon Lee | Aptasensor and method of detecting target material |
| WO2016183370A1 (en) | 2015-05-13 | 2016-11-17 | The United States Of America, As Represented By The Secretary, Department Of Health & Human Services | Synthetic immunosuppressive oligodeoxynucleotides reduce ischemic tissue damage |
| EP3329004B1 (en) | 2015-07-29 | 2020-01-15 | IFOM Fondazione Istituto Firc di Oncologia Molecolare | Therapeutic oligonucleotides |
| EP3377095B1 (en) | 2015-11-18 | 2020-01-01 | INSERM - Institut National de la Santé et de la Recherche Médicale | Methods and pharmaceutical compositions for treating rhabdomyolysis |
| KR20180089531A (en) | 2015-12-25 | 2018-08-08 | 에스비아이 바이오테크 가부시키가이샤 | TLR inhibitory oligonucleotides and uses thereof |
| US10124062B2 (en) | 2016-02-26 | 2018-11-13 | The United States Of America As Represented By The Department Of Veterans Affairs | Methods of treating, inhibiting and/or preventing an auditory impairment |
| US11339396B2 (en) | 2016-06-08 | 2022-05-24 | President And Fellows Of Harvard College | Engineered viral vector reduces induction of inflammatory and immune responses |
| CN117653654A (en) | 2016-11-23 | 2024-03-08 | 柏林制药股份有限公司 | Aptamer for inhibiting and/or suppressing TLR9 activation |
| WO2019094548A1 (en) | 2017-11-08 | 2019-05-16 | President And Fellows Of Harvard College | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| WO2020232211A1 (en) | 2019-05-15 | 2020-11-19 | President And Fellows Of Harvard College | Compositions and methods for inhibiting nucleic acid vector- induced inflammatory responses |
-
2018
- 2018-11-08 WO PCT/US2018/059756 patent/WO2019094548A1/en unknown
- 2018-11-08 KR KR1020207015916A patent/KR20200085812A/en not_active Application Discontinuation
- 2018-11-08 EP EP18842597.9A patent/EP3707262A1/en active Pending
- 2018-11-08 US US16/762,356 patent/US11981911B2/en active Active
- 2018-11-08 JP JP2020525991A patent/JP2021502381A/en active Pending
- 2018-11-08 AU AU2018364542A patent/AU2018364542A1/en active Pending
- 2018-11-08 CN CN201880072584.XA patent/CN111511918A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050239733A1 (en) * | 2003-10-31 | 2005-10-27 | Coley Pharmaceutical Gmbh | Sequence requirements for inhibitory oligonucleotides |
| US20110070241A1 (en) * | 2009-06-30 | 2011-03-24 | Duke University | Methods for modulating immune responses to aav gene therapy vectors |
Non-Patent Citations (60)
| Title |
|---|
| "Microcapsules and Nanoparticles in Medicine and Pharmacy", 1992, CRC PRESS |
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING CO. |
| ALTSCHUL, S. F. ET AL., J. MOLEC. BIOL., vol. 215, 1990, pages 403 |
| ALTSCHUL, S. F. ET AL., NUCLEIC ACIDS RES, vol. 25, 1997, pages 3389 |
| ASHMAN, RF ET AL., INT IMMUNOL, vol. 23, no. 3, 2011, pages 203 - 14 |
| BAINBRIDGE ET AL., N ENGL J MED, vol. 372, 2015, pages 1887 - 1897 |
| BATES PJ ET AL., EXP MOL PATHOL, vol. 86, no. 3, 2009, pages 151 - 64 |
| CHAKRABORTY, A. ET AL., SCI REP., vol. 3, 2013, pages 1746 |
| DEVEREUX, J. ET AL., NUCLEIC ACIDS RESEARCH, vol. 12, no. 1, 1984, pages 387 |
| DIMOPOULOS ET AL., AM J OPHTHALMOL, vol. 193, September 2018 (2018-09-01), pages 130 - 142 |
| FAUST ET AL., J CLIN INVEST, vol. 123, 2013, pages 2994 - 3001 |
| FERREIRA ET AL., FRONT IMMUNOL, vol. 5, 2014, pages 82 |
| FLOTTE ET AL., HUM GENE THER, vol. 22, 2011, pages 1239 - 1247 |
| GRAY, JT ET AL., METHODS MOL. BIOL., vol. 807, 2011, pages 25 - 46 |
| GURSEL, I ET AL., J IMMUNOL, vol. 171, no. 3, 2003, pages 1393 - 400 |
| HUANG X ET AL: "Targeting the TLR9MyD88 pathway in the regulation of adaptive immune responses", 1 August 2010, EXPERT OPINION ON THERAPEUTIC TAR, INFORMA HEALTHCARE, UK, PAGE(S) 787 - 796, ISSN: 1472-8222, XP009155283 * |
| KAMINSKI, JJ ET AL., J IMMUNOL, vol. 191, no. 7, 2013, pages 3876 - 83 |
| KRIEG AM, NAT REV DRUG DISCOV, vol. 5, no. 6, 2006, pages 471 - 84 |
| KRIEG, AM ET AL., NATURE, vol. 374, no. 6522, 1995, pages 546 - 9 |
| KUMAGAI, Y ET AL., ADV DRUG DELIV REV, vol. 60, no. 7, 2008, pages 795 - 804 |
| LEE, J ET AL., PROC NATL ACAD SCI USA., vol. 108, no. 34, 2011, pages 14055 - 60 |
| LENERT, P ET AL., ARTHRITIS RES THER, vol. 11, no. 3, 2009, pages R79 |
| LENERT, P ET AL., DNA CELL BIOL, vol. 22, no. 10, 2003, pages 621 - 31 |
| LENERT, PS ET AL., ARTHRITIS RES THER, vol. 8, no. 1, 2006, pages 203 |
| LENERT, PS, MEDIATORS INFLAMM, vol. 2010, 2010, pages 986596 |
| LI, Y ET AL., VACCINE, vol. 29, no. 11, 2011, pages 2193 - 8 |
| LIU, Q. ET AL., GENE THERAPY, vol. 10, 2003, pages 935 - 940 |
| MARKUS HS ET AL., STROKE, vol. 42, no. 8, 2011, pages 2149 - 53 |
| MARTINO, AT ET AL., BLOOD, vol. 117, no. 24, 2011, pages 6459 - 68 |
| MATHIOWITZ ET AL., J. APPL. POLYMER SCI., vol. 35, 1988, pages 755 - 774 |
| MATHIOWITZ ET AL., REACTIVE POLYMERS, vol. 6, 1987, pages 275 - 283 |
| MATHIOWITZ; LANGER, J. CONTROLLED RELEASE, vol. 5, 1987, pages 13 - 22 |
| MAYS ET AL., J IMMUNOL, vol. 182, 2009, pages 6051 - 6060 |
| MCCARTY, DM ET AL., GENE THER, vol. 8, no. 16, 2001, pages 1248 - 54 |
| MCCARTY, DM ET AL., MOL THER, vol. 16, no. 10, 2008, pages 1648 - 56 |
| MCLAUGHLIN, SK ET AL., J VIROL, vol. 62, no. 6, 1988, pages 1963 - 73 |
| NEEDLEMAN, S.B. ET AL., J. MOL. BIOL., vol. 48, 1970, pages 443 |
| NELSON S YEW ET AL: "Reducing the immunostimulatory activity of CpG-containing plasmid DNA vectors for non-viral gene therapy", EXPERT OPINION ON DRUG DELIVERY, vol. 1, no. 1, 1 November 2004 (2004-11-01), GB, pages 115 - 125, XP055474595, ISSN: 1742-5247, DOI: 10.1517/17425247.1.1.115 * |
| OHTO, U ET AL., NATURE, vol. 520, no. 7549, 2015, pages 702 - 5 |
| OLSON ET AL., J IMMUNOL, vol. 173, 2004, pages 3916 - 3924 |
| PETER, M ET AL., IMMUNOLOGY, vol. 123, no. l, 2008, pages 118 - 28 |
| PURI, A ET AL., CRIT REV THER DRUG CARRIER SYST, vol. 26, no. 6, 2009, pages 523 - 80 |
| RAMACHANDRAN ET AL., HUM GENE THER, vol. 28, 2017, pages 154 - 167 |
| REICHEL ET AL., MOL THER, vol. 25, 2017, pages 2648 - 2660 |
| ROBBINS, PD ET AL., PHARMOL THER, vol. 80, no. 1, 1998, pages 35 - 47 |
| SAMULSKI, RJ ET AL., ANNU REV VIROL, vol. 1, no. 1, 2014, pages 427 - 51 |
| SAMULSKI, RJ ET AL., J VIROL, vol. 61, no. 10, 1987, pages 3096 - 101 |
| SANCHEZ ET AL., GRAEFES ARCH CLIN EXP OPHTHALMOL, vol. 249, 2011, pages 475 - 482 |
| SHIROTA, H ET AL., J IMMUNOL, vol. 174, no. 8, 2005, pages 4579 - 83 |
| SING, R ET AL., EXP MOL PATHOL, vol. 86, no. 3, 2009, pages 215 - 223 |
| SMITH, T.F. ET AL., J. MOL. BIOL., vol. 147, 1981, pages 195 |
| SOUNDARARAJAN S ET AL., CANCER RES, vol. 68, no. 7, 2008, pages 2358 - 65 |
| STUNZ, LL ET AL., EUR J IMMUNOL, vol. 32, no. 5, 2002, pages 1212 - 22 |
| TAKEDA, K ET AL., SEMIN IMMUNOL, vol. 16, no. 1, 2004, pages 3 - 9 |
| VINCENZO CERULLO ET AL: "Toll-like Receptor 9 Triggers an Innate Immune Response to Helper-dependent Adenoviral Vectors", MOLECULAR THERAPY, vol. 15, no. 2, 1 February 2007 (2007-02-01), GB, pages 378 - 385, XP055580038, ISSN: 1525-0016, DOI: 10.1038/sj.mt.6300031 * |
| XIONG, W. ET AL., J CLIN INVEST, vol. 125, no. 4, 2015, pages 1433 - 45 |
| XUE ET AL., NAT MED, vol. 24, no. 10, October 2018 (2018-10-01), pages 1507 - 1512 |
| ZHONG, L ET AL., MOL THER, vol. 16, no. 2, 2008, pages 290 - 5 |
| ZHOU J ET AL., THER NUCLEIC ACIDS, vol. 3, 2014, pages el69 |
| ZHOU, X ET AL., MOL THER, vol. 16, no. 3, 2008, pages 494 - 9 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11339396B2 (en) | 2016-06-08 | 2022-05-24 | President And Fellows Of Harvard College | Engineered viral vector reduces induction of inflammatory and immune responses |
| US11981911B2 (en) | 2017-11-08 | 2024-05-14 | President And Fellows Of Harvard College | Compositions and methods for inhibiting viral vector-induced inflammatory responses |
| WO2022187679A1 (en) * | 2021-03-04 | 2022-09-09 | Kriya Therapeutics, Inc. | Viral vector constructs incorporating dna for inhibiting toll like receptors and methods of using the same |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20200085812A (en) | 2020-07-15 |
| EP3707262A1 (en) | 2020-09-16 |
| CN111511918A (en) | 2020-08-07 |
| US20200270637A1 (en) | 2020-08-27 |
| AU2018364542A1 (en) | 2020-04-16 |
| US11981911B2 (en) | 2024-05-14 |
| JP2021502381A (en) | 2021-01-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220111015A1 (en) | Treatment of ocular neovascularization using anti-vegf proteins | |
| US11981911B2 (en) | Compositions and methods for inhibiting viral vector-induced inflammatory responses | |
| Hauswirth et al. | Treatment of leber congenital amaurosis due to RPE65 mutations by ocular subretinal injection of adeno-associated virus gene vector: short-term results of a phase I trial | |
| JP6290185B2 (en) | Viral vectors for treating retinal dysplasia | |
| JP2023509177A (en) | Methods and compositions for treating Usher's syndrome | |
| JP2020535184A (en) | Treatment of eye diseases with post-translationally modified fully human anti-VEGF Fab | |
| KR20190038536A (en) | Compositions and methods for reducing ocular angiogenesis | |
| US20210260168A1 (en) | Compositions and methods of fas inhibition | |
| JP7289306B2 (en) | Compositions and methods for treating retinal disorders | |
| US20220259599A1 (en) | Compositions and methods for inhibiting nucleic acid vector-induced inflammatory responses | |
| WO2023280157A1 (en) | Construction and use of anti-vegf antibody in-vivo expression system | |
| AU2022253065A1 (en) | Compositions and methods for ocular transgene expression | |
| AU2020212026A1 (en) | Highly efficient transduction and lateral spread in the retina by a novel AAV virus enhanced by rational design |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18842597 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2018364542 Country of ref document: AU Date of ref document: 20181108 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2020525991 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 20207015916 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2018842597 Country of ref document: EP Effective date: 20200608 |