WO2016150614A1 - A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof - Google Patents
A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof Download PDFInfo
- Publication number
- WO2016150614A1 WO2016150614A1 PCT/EP2016/052969 EP2016052969W WO2016150614A1 WO 2016150614 A1 WO2016150614 A1 WO 2016150614A1 EP 2016052969 W EP2016052969 W EP 2016052969W WO 2016150614 A1 WO2016150614 A1 WO 2016150614A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- jeffamine
- curing agent
- process according
- bis
- aminomethyl
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/50—Amines
- C08G59/504—Amines containing an atom other than nitrogen belonging to the amine group, carbon and hydrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
- C08G59/40—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing characterised by the curing agents used
- C08G59/50—Amines
- C08G59/5026—Amines cycloaliphatic
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B3/00—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties
- H01B3/18—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances
- H01B3/30—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes
- H01B3/40—Insulators or insulating bodies characterised by the insulating materials; Selection of materials for their insulating or dielectric properties mainly consisting of organic substances plastics; resins; waxes epoxy resins
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F30/00—Fixed transformers not covered by group H01F19/00
- H01F30/06—Fixed transformers not covered by group H01F19/00 characterised by the structure
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01F—MAGNETS; INDUCTANCES; TRANSFORMERS; SELECTION OF MATERIALS FOR THEIR MAGNETIC PROPERTIES
- H01F38/00—Adaptations of transformers or inductances for specific applications or functions
- H01F38/20—Instruments transformers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2650/00—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule
- C08G2650/28—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the polymer type
- C08G2650/50—Macromolecular compounds obtained by reactions forming an ether link in the main chain of the macromolecule characterised by the polymer type containing nitrogen, e.g. polyetheramines or Jeffamines(r)
Definitions
- the present invention relates to a process for the preparation of insulation systems for electrical engineering, wherein a multiple component thermosetting epoxy resin composition is used.
- the insulation encased articles obtained by the process according to the present invention exhibit good mechanical, electrical and dielectrical properties and can be used as, for example, insulators, bushings, switchgears and instrument transformers.
- Epoxy resin compositions are commonly used for the preparation of insulation systems for electrical engineering. However, most of these epoxy resin compositions utilize anhydrides as curing agents. Due to the developing regulatory framework for chemicals, it is expected that the use of anhydrides in epoxy resins will be restricted in the near future, because of their R42 label (respiratory sensitizer). Therefore, some anhydrides are already on the SVHC candidate list (substances of very high concern) of the REACH regulation. Therefore, it is likely that in some years these substances may no longer be used without special authorisation. As all known anhydrides are R42-labeled and even yet unknown anhydrides would be expected by toxicologists to be also R42-labeled, a solution that is free of anhydrides is desirable.
- Amines as curing agents for epoxy resins are well known, in particular, for the preparation of composite materials. However, amine curing agents are often too reactive to be processable in electrical potting or encapsulation applications. As the mass of the epoxy resin
- thermoset the composition to be processed increases, control of the exotherm becomes vital.
- the uncontrolled release of heat from the curing of the thermoset due to its mass may result in the degradation of the thermoset's mechanical properties, or even to thermal decomposition of the thermoset. Also degradation of the mechanical properties of the structural parts in contact with the thermoset is likely to occur.
- APG automatic pressure gelation process
- compositions is inappropriate and the exotherm is too high for application in APG, when amines are used as curing agents.
- aromatic amines In order to cope with the problem of an inappropriate cure profile of epoxy resins containing amine curing agents, the use of aromatic amines was suggested. However, today the aromatic amines considered are on the banned substance list which prevents their use in potting or casting applications.
- other amines such as aliphatic amines, are too reactive and do not provide an acceptable gelation profile in APG, which is suitable for the casting of big parts with low shrinkage and low exotherm.
- some properties of the cured products are not competitive with anhydride cured thermosets, such as long term aging, tracking resistance, arc resistance, dielectric properties after humid conditioning. Accordingly, there is a need for new thermosetting, anhydride-free epoxy compositions which advantageously can be used in potting or encapsulation applications for manufacturing of electrical insulation systems having improved properties, which are suitable for switchgear, transformer and other applications.
- an object of the present invention to provide a process for the preparation of insulation systems for electrical engineering by automatic pressure gelation (APG), wherein anhydride-free, thermosetting epoxy compositions can be used, and the cure profile can be controlled in the desired manner.
- Still another object of the present invention is to provide the encased articles obtained from the inventive process which exhibit excellent mechanical, electrical and dielectrical properties and can be used, for example, as insulators, bushings, switchgears and instrument transformers in electrical engineering.
- the present invention relates to a process for the preparation of insulation systems for electrical engineering by automatic pressure gelation (APG), wherein
- thermosetting resin composition comprising
- insulation systems are prepared by casting, potting, encapsulation, and impregnation processes such as gravity casting, vacuum casting, automatic pressure gelation (APG), vacuum pressure gelation (VPG), infusion, and the like.
- a typical process for making insulation systems for electrical engineering, such as cast resin epoxy insulators, is the automatic pressure gelation process (APG process).
- the APG process allows for the preparation of a casting product made of an epoxy resin in a short period of time by hardening and forming the epoxy resin.
- an APG apparatus to carry out the APG process includes a pair of molds (herafter called mold), a resin mixing tank connected to the mold through a pipe, and an opening and closing system for opening and closing the mold.
- the components of the curable composition comprising the epoxy resin and the curing agent have to be prepared for injection.
- components are combined and transferred into a mixer and mixed at elevated temperature and reduced pressure to degas the formulation.
- the degassed mixture is subsequently injected into the hot mold.
- the epoxy resin component and the curing agent component are typically mixed individually with the filler at elevated temperature and reduced pressure to prepare the pre-mixture of the resin and the curing agent.
- further additives may be added beforehand.
- the two components are combined to form the final reactive mixture, typically by mixing at elevated temperature and reduced pressure. Subsequently, the degassed mixture is injected into the mold.
- a metal conductor or an insert which is pre-heated and dried, is placed into the mold located in a vacuum chamber.
- the epoxy resin composition is injected into the mold from an inlet located at the bottom of the mold by applying pressure to the resin mixing tank.
- the resin composition is normally held at a moderate temperature of 40 to 60 C to ensure an appropriate pot life (usable time of the epoxy resin), while the temperature of the mold is kept at around 120 C or above to obtain the casting products within a reasonably short time.
- the resin composition cures while the pressure applied to the epoxy resin in the resin mixing tank is kept at about 0.1 to 0.5 MPa.
- casting products made of more than 10 kg of resin may be produced conveniently by the APG process within a short time, for example, of from 20 to 60 minutes. Normally, the casting product released from the mold is post cured in a separate curing oven to complete the reaction of the epoxy resin.
- the at least one epoxy resin (A) is a compound containing at least one vicinal epoxy group, preferably more than one vicinal epoxy group, for example, two or three vicinal epoxy groups.
- the epoxy resin may be saturated or unsaturated, aliphatic, cycloaliphatic, aromatic or heterocyclic and may be substituted.
- the epoxy resin may also be a monomeric or a polymeric compound.
- the epoxy resins used in embodiments disclosed herein for component (A) of the present invention, may vary and include conventional and commercially available epoxy resins, which may be used alone or in combinations of two or more. In choosing epoxy resins for the compositions disclosed herein, consideration should not only be given to properties of the final product, but also to viscosity and other properties that may influence the processing of the resin composition.
- Particularly suitable epoxy resins known to the skilled worker are based on reaction products of polyfunctional alcohols, phenols, cycloaliphatic carboxylic acids, aromatic amines, or aminophenols with epichlorohydrin.
- Aliphatic alcohols which come into consideration for reaction with epichlorhydrin to form suitable polyglycidyl ethers are, for example, ethylene glycol and poly(oxyethylene)glycols such as diethylene glycol and triethylene glycol, propylene glycol and poly(oxypropylene)- glycols, propane-1 ,3-diol, butane-1 ,4-diol, pentane-1 ,5-diol, hexane-1 ,6-diol, hexane-2,4,6- triol, glycerol, 1 ,1 ,1 -trimethylolpropane, and pentaerythritol.
- ethylene glycol and poly(oxyethylene)glycols such as diethylene glycol and triethylene glycol
- propylene glycol and poly(oxypropylene)- glycols propane-1 ,3-diol, butane-1 ,4-diol, pentane-1
- Cycloaliphatic alcohols which come into consideration for reaction with epichlorhydrin to form suitable polyglycidyl ethers are, for example, 1 ,4-cyclohexanediol (quinitol), 1 ,1 - bis(hydroxymethyl)cyclohex-3-ene, bis(4-hydroxycyclohexyl)methane, and 2,2-bis(4- hydroxycyclohexyl)-propane.
- Alcohols containing aromatic nuclei which come into consideration for reaction with epichlorhydrin to form suitable polyglycidyl ethers are, for example, N,N-bis-(2- hydroxyethyl)aniline and 4,4'-bis(2-hydroxyethylamino)diphenylmethane.
- the polyglycidyl ethers are derived from substances containing two or more phenolic hydroxy groups per molecule, for example, resorcinol, catechol, hydroquinone, bis(4-hydroxyphenyl)methane (bisphenol F), 1 ,1 ,2,2-tetrakis(4-hydroxyphenyl)ethane, 4,4'- dihydroxydiphenyl, bis(4-hydroxyphenyl)sulphone (bisphenol S), 1 ,1 -bis(4-hydroxylphenyl)-1 - phenyl ethane (bisphenol AP), 1 ,1 -bis(4-hydroxylphenyl)ethylene (bisphenol AD), phenol- formaldehyde or cresol-formaldehyde novolac resins, 2,2-bis(4-hydroxyphenyl)propane (bisphenol A), and 2,2-bis(3,5-dibromo-4-hydroxyphenyl)propane.
- bisphenol F bis(4-hydroxyphenyl)
- Another few non-limiting embodiments include, for example, triglycidyl ethers of para- aminophenols. It is also possible to use a mixture of two or more epoxy resins.
- the at least one epoxy resin component (A) is either commercially available or can be prepared according to processes known per se.
- Commercially available products are, for example, D.E.R. 330, D.E.R. 331 , D.E.R.332, D.E.R. 334, D.E.R. 354, D.E.R. 580, D.E.N. 431 , D.E.N. 438, D.E.R. 736, or D.E.R. 732 available from The Dow Chemical Company, or ARALDITE ® MY 740 or ARALDITE ® CY 228 from Huntsman Corporation.
- the amount of epoxy resin (A) in the final composition is, for example, of from 30 weight percent (wt %) to 92 wt %, based on the total weight of components (A) and (B) in the composition. In one embodiment, the amount of epoxy resin (A) is, for example, of from 45 wt % to 87 wt %, based on the total weight of components (A) and (B). In another embodiment, the amount of the epoxy resin (A) is, for example, of from 50 wt % to 82 wt %, based on the total weight of components (A) and (B). ln a preferred embodiment of the present invention the at least one epoxy resin (A) is a diglycidylether of bisphenol A.
- the at least one curing agent component (b1 ) is a cycioaiiphatic amine.
- cycioaiiphatic amine denotes cycioaiiphatic amines and mixed cycioaiiphatic-aromatic amine derivatives, for example, methylene bridged aminobenzyl-cyclohexylamines.
- cyclohexylamines examples include 1 ,2-diaminocyclohexane, 1 ,4-diaminocyclohexane, bis(4- aminocyclohexyl)methane, bis(3-methyl-4-aminocyclohexyl)methane, bis(3,5-methyl-4- aminocyclohexyl)methane, 2,4-bis(4-aminocyclohexylmethyl)cyclohexylamine, 2,2-bis(4- aminocyclohexyl)propane, 4,4'-bis(4-cyclohexylmethyl)dicyclohexylamine, 2,2-bis(4-amino-3- methylcylohexyl)propane, 3-aminomethyl-3,5.5-trimethylcyclohexylamine (isophorone diamine), 1 ,4-bis(aminomethyl)cyclohexane, 1 ,3-bis(aminomethyl)cycl
- bicyclo[2.2.1]heptanebis(methylamine) (norbornane diamine), 3,3.5-trimethyl-N-(propan-2- yl)-5-[(propan-2-ylamino)methyl]cyclohexylamine, Jefflink JL 754 available from Huntsman Corporation, 4-aminocyclohexyl-4-hydroxycyclohexylmethane, N-aminoethylpiperazine.
- mixed cycioaiiphatic-aromatic amines include 4-(4'- aminobenzyl)cyclohexylamine, 2,4-bis(4-aminocyclohexylmethyl)aniline, and, partially hydrogenated trimethylenetetraaniline and analogs thereof and hydrogenated bisaniline A and hydrogenated bisaniline P.
- the amount of curing agent component (b1 ) in the final composition is, for example, of from 1 weight percent (wt %) to 30 wt %, based on the total weight of components (A) and (B) in the composition. In one embodiment, the amount of curing agent component (b1 ) is, for example, of from 2 wt % to 20 wt %, based on the total weight of components (A) and (B). In another embodiment, the amount of curing agent component (b1 ) is, for example, of from 3 wt % to 15 wt %, based on the total weight of components (A) and (B).
- Preferred cycioaiiphatic amines include 1 ,2-diaminocyclohexane, bis(4- aminocyclohexyl)methane, 3-aminomethyl-3,5,5-trimethylcyclohexylamine (isophorone diamine), 1 ,3-bis(aminomethyl)cyclohexane, bicyclo[2.2.1 ]heptanebis(methylamine)
- the at least one curing agent component (b1 ) is 3-aminomethyl-3,5,5-trimethylcyclohexylamine (isophorone diamine) denoted IPD.
- the cycloaliphatic amine may be used alone, or, alternatively, mixtures of at least two, for example, two, three or four different cycloaliphatic amines may be used.
- the at least one curing agent component (b2) is a polyetheramine.
- the polyetheramine is, for example, a polyether polyamine, such as a polyether triamine, or a polyether diamine.
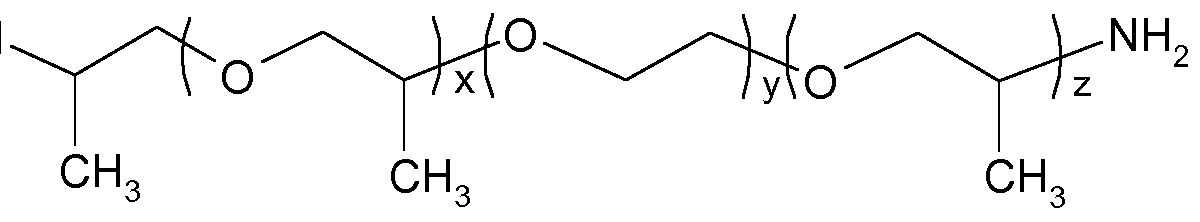
- Useful polyether diamines include polyoxyalkylene diamines such as polyethylene oxide- polypropylene oxide copolymers that are co-terminated by amine groups. Such polyether diamines may have the formula H 2 N(PO) x (EO)y(PO) z NH 2 , wherein x is a number of from 0 to 10, y is a number of from 0 to 40 and z is a number of from 0 to 10, EO is ethylene oxide and PO is propylene oxide.
- the polyether polyamines may also be other polyethylene oxide or polypropylene oxide polymers co-terminated by amine groups.
- Representative polyether diamines using ethylene oxide (EO) and propylene oxide (PO) include the polyether diamines of the following formulae
- polyether polyamines or ether oligomers may be used.
- Any primary polyamine having a hydrocarbon chain with some ether oxygen atoms included may be used. The oxygen atoms may be spaced at regular intervals, so that the polyether polyamine has a single repeating monomer unit, or the oxygen atoms may be spaced at differing intervals, which may be random or distributed according to a repeating pattern.
- the polyether polyamine may be a diamine of an ether copolymer, which may be random, block, repeating, or alternating, or of an ether multipolymer having three or more different ether monomer units.
- the polyether polyamines may have primary or secondary amines.
- the oxygen atoms of the polyether component of the polyether polyamine may be replaced, altogether or in part, with other electronegative species such as sulfur.
- a polythioether polyamine may be used.
- polyether polyamines examples include JEFFAMINE ® polyetheramines that are commercially available from Huntsman Corporation.
- the ether units of these amines are ethylene oxide units, propylene oxide units or mixtures thereof.
- JEFFAMINE ® polyetheramines typically have oxypropylene units or mixtures of oxyethylene and oxypropylene units.
- JEFFAMINE ® polyetheramines which are preferred as curing agent component (b2) are JEFFAMINE ® D-, JEFFAMINE ® ED-, JEFFAMINE ® T-, and JEFFAMINE ® XTJ-series polyetheramines.
- the JEFFAMINE ® D-series polyetheramines are amine terminated polypropylene glycols (PPG) of the general formula
- x is a number of from 2 to 8, in particular x is -2.5 for JEFFAMINE ® D-230, or x is about 6.1 for JEFFAMINE ® D-400.
- the JEFFAMINE ® ED-series polyetheramines are polyether diamines based on a predominantly polyethylene glycol (PEG) backbone of the general formula (2 ) wherein y is a number of from 5 to 40 and the sum of x + z is a number of from 3 to 8, in particular y is about 9.0 and x + z is about 3.6 for JEFFAMINE ® ED-600, or y is about 12.5 and x + z is about 6.0 for JEFFAMINE ® ED-900.
- PEG polyethylene glycol
- the JEFFAMINE ® T-series polyetheramines are amine terminated polypropylene glycols (PEG) of the general formula
- R is hydrogen, CH 3 or C 2 H 5 , n is a number 0, 1 or 2, and x + y + z is a number of from 3 to 100, in particular R is C 2 H 5 , n is 1 , and x + y + z is a number of from 5 to 6 for JEFFAMINE ® T-403.
- the JEFFAMINE ® XTJ-series polyetheramines are slower amines analogous to
- JEFFAMINE ® XTJ-568 is preferred.
- JEFFAMINE ® XT J polyetheramines are primary amines prepared by amination of butylene oxide capped alcohols. The reaction results in primary amines with the terminal end group of the formula
- the polyether polyamines may be used alone, or, alternatively, mixtures of at least two, for example, two, three or four different polyether polyamines may be used.
- the amount of curing agent component (b2) in the final composition is, for example, of from 2 weight percent (wt %) to 40 wt %, based on the total weight of components (A) and (B) in the composition. In one embodiment, the amount of curing agent component (b2) is, for example, of from 5 wt % to 30 wt %, based on the total weight of components (A) and (B). In another embodiment, the amount of curing agent component (b2) is, for example, of from 5 wt % to 20 wt %, based on the total weight of components (A) and (B).
- Particular JEFFAMINE ® polyetheramines that may be used as curing agent component (b2) in accordance with the process of the present invention include JEFFAMINE ® D-230, JEFFAMINE ® D-400, JEFFAMINE ® T-403, JEFFAMINE ® XTJ-568, JEFFAMINE ® ED-600, and JEFFAMINE ® ED-900, especially preferred are JEFFAMINE ® D-230, JEFFAMINE ® D- 400, JEFFAMINE ® T-403 and JEFFAMINE ® XTJ-568.
- a process according to the present invention is preferred wherein the said resin composition comprises
- a polyetheramine selected from the group JEFFAMINE ® D-230, JEFFAMINE ® D-400, JEFFAMINE ® T-403, JEFFAMINE ® XTJ-568.
- the multiple component thermosetting resin composition according to the process of the present invention may contain one or more fillers generally used in electrical insulations which are selected from the group consisting of metal powder, wood flour, glass powder, glass beads, semi-metal oxides, metal oxides, metal hydroxides, semi-metal and metal nitrides, semi-metal and metal carbides, metal carbonates, metal sulfates, and natural or synthetic minerals.
- fillers generally used in electrical insulations which are selected from the group consisting of metal powder, wood flour, glass powder, glass beads, semi-metal oxides, metal oxides, metal hydroxides, semi-metal and metal nitrides, semi-metal and metal carbides, metal carbonates, metal sulfates, and natural or synthetic minerals.
- Preferred fillers are selected from the group consisting of quartz sand, silanised quartz powder, silica, aluminium oxide, titanium oxide, zirconium oxide, Mg(OH) 2 , AI(OH) 3 , dolomite [CaMg (C0 3 ) 2 ], silanised AI(OH) 3 , AIO(OH), silicon nitride, boron nitrides, aluminium nitride, silicon carbide, boron carbides, dolomite, chalk, CaC0 3 , barite, gypsum, hydromagnesite, zeolites, talcum, mica, kaolin and wollastonite. Especially preferred is silica, wollastonite or calcium carbonate.
- the filler material may optionally be coated for example with a silane or a siloxane known for coating filler materials, e.g. dimethylsiloxanes which may be cross linked, or other known coating materials.
- the amount of filler in the final composition is, for example of from 30 weight percent (wt %) to 75 wt %, based on the total weight of the thermosetting epoxy resin composition. In one embodiment, the amount of filler is, for example, of from 40 wt % to 75 wt %, based on the total weight of the thermosetting epoxy resin composition. In another embodiment, the amount of filler is, for example, of from 50 wt % to 70 wt %, based on the total weight of the thermosetting epoxy resin composition. In still another embodiment, the amount of filler is, for example, of from 60 wt % to 70 wt %, based on the total weight of the thermosetting epoxy resin composition.
- Further additives may be selected from processing aids to improve the rheological properties of the liquid mix resin, hydrophobic compounds including silicones, wetting/dispersing agents, plasticizers, reactive or non-reactive diluents, flexibilizers, accelerators, antioxidants, light absorbers, pigments, flame retardants, fibers and other additives generally used in electrical applications. These additives are known to the person skilled in the art.
- the present invention also refers to the use of a multiple component thermosetting resin composition comprising
- APG automatic pressure gelation
- Preparation of insulation systems for electrical engineering is often carried out by Automatic Pressure Gelation (APG) or Vacuum Casting.
- APG Automatic Pressure Gelation
- Vacuum Casting When using known epoxy resin compositions based on anhydride cure, such processes typically include a curing step in the mold for a time sufficient to shape the epoxy resin composition into its final infusible three dimensional structures, typically up to ten hours, and a post-curing step of the demolded article at elevated temperature to develop the ultimate physical and mechanical properties of the cured epoxy resin composition.
- Such a post-curing step may take, depending on the shape and size of the article, up to thirty hours.
- the cure profile and shrinkage can advantageously be controlled in the desired manner, when carrying out the inventive process.
- shorter curing times and lower mold and curing temperatures can be applied.
- the post-cure time can be substantially shortened and the post-cure temperature lowered, all of which safes process time and energy.
- a post-cure treatment may even be omitted.
- the pot life of the thermosetting epoxy resin composition according to the inventive process is sufficient to use common application techniques known in the art.
- thermosetting epoxy resin composition according to the inventive process are distinguished by less odor emission.
- a lower exothermic peak temperature to control the cure profile, i.e. gelation front within the mold, is provided by the process according to the present invention, which is similar to processes carried out with known epoxy resin compositions based on anhydride cure.
- the process according to the present invention is useful for the preparation of encased articles exhibiting good mechanical, electrical and dielectrical properties.
- the present invention refers to an insulation system article obtained by the process according to the present invention.
- the glass transition temperature of the article is in the same range as for known high temperature cure anhydride based thermosetting epoxy resin compositions.
- insulation system articles prepared according to the present invention are dry-type transformers, particularly cast coils for dry type distribution transformers, especially vacuum cast dry distribution transformers, which within the resin structure contain electrical conductors; medium and high- voltage insulations for indoor and outdoor use, like breakers or switchgear applications; medium and high voltage bushings; as long-rod, composite and cap-type insulators, and also for base insulators in the medium-voltage sector, in the production of insulators associated with outdoor power switches, measuring transducers, leadthroughs, and overvoltage protectors, in switchgear constructions, in power switches, and electrical machines, as coating materials for transistors and other
- the articles prepared in accordance with the inventive process are used for medium and high voltage switchgear applications and instrument transformers (6 kV to 72 kV).
- thermosetting resin composition is prepared by using as the epoxy resin component (A) 100 parts of ARALDITE ® MY 740, and as the curing agent component (B) 28 parts of a mixture containing, as component (b1 ), 8 parts of isophorone diamine and, as component (b2), 20 parts of JEFFAMINE ® XTJ-568.
- a total of 192 parts of Silica W12 (available from Quarzwerke) are used as the filler (60 wt% based on the total weight of the thermosetting epoxy resin composition).
- Components (A) and (B) are pre-mixed individually with the appropriate quantity of the filler.
- the premixes of filled components (A) and (B) are feeded into a batch mixer at a temperature of 40°C and injected into the mold preheated to 1 10 to 120°C and mold temperature is kept at this temperature for 2h at a maximum temperature of 120°C.
- the exotherm as determined by Differential Scanning Calorimetry on a Mettler SC 822 e is 126 J/g.
- Example 1 is repeated. However, a total of 238 parts of Silica W12 are used as the filler (65 wt% based on the total weight of the thermosetting epoxy resin composition), instead of 192 parts. Components (A) and (B) are pre-mixed individually with the appropriate quantity of the filler and processed as given in Example 1.
- ARALDITE ® casting resin contains 100 parts of ARALDITE ® CY 228 (diglycidylether of bisphenol A), 85 parts of Hardener HY 918 (anhydride hardener), 0.8 parts of Accelerator DY 062 (tertiary amine accelerator) and 340 parts of Silica W12 (65 wt% based on the total weight of the thermosetting epoxy resin composition).
- the individual components are mixed with the appropriate quantities of fillers and additives.
- the premixes are feeded into a batch mixer at a temperature of 60°C and injected into the mold preheated to 135°C and mold temperature is kept at this temperature until curing is completed. Cure time is 10h at a maximum temperature of 140°C.
- the exotherm as determined by Differential Scanning Calorimetry on a Mettler SC 822 e is 120 J/g.
- APG trails with the compositions prepared in accordance with Examples 1 and 2, and Comparative Example are carried out by using as a mold a cylinder (length: 300 mm, diameter 60 mm).
- a release agent (QZ 66 available from Huntsman Corporation) is used.
- the total weight of the thermosetting casting resin composition injected under external pressure (about 3 bar) into the mold is approximately 1 .1 kg.
- thermosetting resin composition *wt% based on the total weight of the thermosetting resin composition
- the pot life of the thermosetting epoxy resin composition of Example 1 is sufficient to use common application techniques known in the art, as demonstrated by the data given in Table 1 .
- the composition of Example 1 is distinguished by low odor emission. Odor emission of the composition of Comparative Example is much more intense.
- shorter curing times and lower curing temperatures can be applied in case of the composition of Example 1 , as demonstrated by the gel time data given in Table 2.
- the post-cure time can be substantially shortened and the post-cure temperature lowered, which is demonstrated by the corresponding data in Table 3.
- the data given in Table 3 demonstrate that the glass transition temperatures before and after post cure and shrinkage after post cure of the composition of Example 1 are in the same range as the properties of the known composition according to the Comparative Example.
Abstract
Description
Claims
Priority Applications (10)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2017012199A MX2017012199A (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof. |
| PL16704432.0T PL3275002T5 (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof |
| JP2017545303A JP6683721B2 (en) | 2015-03-26 | 2016-02-12 | Method of manufacturing insulation systems for electrical engineering, products obtained therefrom, and their use |
| US15/561,918 US20180112031A1 (en) | 2015-03-26 | 2016-02-12 | A Process for the Preparation of Insulation Systems for Electrical Engineering, the Articles Obtained Therefrom and the Use Thereof |
| ES16704432T ES2739688T5 (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical equipment, the articles obtained therefrom and their use |
| KR1020177022791A KR102580662B1 (en) | 2015-03-26 | 2016-02-12 | Method for manufacturing insulating systems for electrical engineering, articles obtained thereby, and uses thereof |
| CA2976825A CA2976825C (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof |
| CN201680018184.1A CN107531884B (en) | 2015-03-26 | 2016-02-12 | Method for producing an electrical engineering insulation system, products obtained therefrom and use thereof |
| EP16704432.0A EP3275002B2 (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical engineering, and the use thereof |
| HRP20191244TT HRP20191244T4 (en) | 2015-03-26 | 2019-07-11 | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15161029.2 | 2015-03-26 | ||
| EP15161029 | 2015-03-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016150614A1 true WO2016150614A1 (en) | 2016-09-29 |
Family
ID=52736936
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2016/052969 WO2016150614A1 (en) | 2015-03-26 | 2016-02-12 | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US20180112031A1 (en) |
| EP (1) | EP3275002B2 (en) |
| JP (1) | JP6683721B2 (en) |
| KR (1) | KR102580662B1 (en) |
| CN (1) | CN107531884B (en) |
| CA (1) | CA2976825C (en) |
| ES (1) | ES2739688T5 (en) |
| HR (1) | HRP20191244T4 (en) |
| MX (1) | MX2017012199A (en) |
| PL (1) | PL3275002T5 (en) |
| TW (1) | TWI777917B (en) |
| WO (1) | WO2016150614A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108189423A (en) * | 2017-12-29 | 2018-06-22 | 江苏神马电力股份有限公司 | Compound insulation tube liner and its preparation method and application |
| CN108215233A (en) * | 2017-12-11 | 2018-06-29 | 湖北耐创新材料洁具有限公司 | A kind of pressure molding of resin and mineral mixing pouring piece |
| WO2018140576A1 (en) * | 2017-01-26 | 2018-08-02 | Huntsman Advanced Materials Licensing (Switzerland) Gmbh | A thermosetting epoxy resin composition for the preparation of articles for electrical engineering, and the articles obtained therefrom |
| CN109923145A (en) * | 2017-02-06 | 2019-06-21 | 亨斯迈石油化学有限责任公司 | Curing agent for epoxy resin |
| RU2787124C1 (en) * | 2022-08-29 | 2022-12-28 | Российская Федерация, от имени которой выступает Государственная корпорация по атомной энергии "Росатом" (Госкорпорация "Росатом") | Electrical insulating pilling and impregnation compound |
| WO2023114201A3 (en) * | 2021-12-14 | 2023-07-27 | Huntsman Petrochemical Llc | Polyurethane composition |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0839865A1 (en) * | 1996-10-30 | 1998-05-06 | Ciba SC Holding AG | Curable epoxy resin compositions |
| WO2009062543A1 (en) * | 2007-11-13 | 2009-05-22 | Abb Research Ltd | Fiber-reinforced composite system as electrical insulation |
| WO2010010048A1 (en) * | 2008-07-22 | 2010-01-28 | Basf Se | Blends containing epoxy resins and mixtures of amines with guanidine derivatives |
| WO2013124251A2 (en) * | 2012-02-22 | 2013-08-29 | Basf Se | Blends for composites |
Family Cites Families (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BE755079A (en) | 1969-08-21 | 1971-02-22 | Ciba Geigy | Method and device for impregnating articles or parts, in particular electrical windings, by means of hardenable masses of cast resin |
| DE59610415D1 (en) | 1995-04-04 | 2003-06-12 | Vantico Ag | Curable epoxy resin mixture containing wollastonite |
| US5965673A (en) * | 1997-04-10 | 1999-10-12 | Raytheon Company | Epoxy-terminated prepolymer of polyepoxide and diamine with curing agent |
| DE60104449T2 (en) * | 2000-11-29 | 2005-07-28 | Huntsman Advanced Materials (Switzerland) Gmbh | FILLED EPOXY RESIN SYSTEM WITH HIGH MECHANICAL STRENGTH |
| DE602006010872D1 (en) * | 2005-07-15 | 2010-01-14 | Huntsman Adv Mat Switzerland | HARDENED COMPOSITION |
| EP2230267B1 (en) * | 2009-03-20 | 2014-08-13 | ABB Research Ltd. | Method of producing a curable epoxy resin composition |
| EP2470583A1 (en) | 2009-08-27 | 2012-07-04 | ABB Research Ltd. | Curable epoxy resin composition |
| EP2480587A1 (en) * | 2009-09-25 | 2012-08-01 | Dow Global Technologies LLC | Curable epoxy resin compositions and composites made therefrom |
| WO2011097009A2 (en) | 2010-02-02 | 2011-08-11 | Dow Global Technologies Llc | Curable epoxy resin compositions |
| US20110315916A1 (en) * | 2010-06-29 | 2011-12-29 | Dow Global Technologies Inc. | Curable composition |
| CN102040805A (en) * | 2010-12-23 | 2011-05-04 | 东方电气集团东方汽轮机有限公司 | Epoxy resin system for manufacturing blade mould of wind-driven generator and preparation method thereof |
| JP5825734B2 (en) * | 2011-07-13 | 2015-12-02 | コニシ株式会社 | Model material or model manufacturing method |
| US9193862B2 (en) * | 2012-02-22 | 2015-11-24 | Basf Se | Blends for composite materials |
| JP2014118576A (en) * | 2012-12-18 | 2014-06-30 | Air Products And Chemicals Inc | Epoxy resin composition including solvated solid |
| JP6461475B2 (en) * | 2013-06-27 | 2019-01-30 | 三菱電機株式会社 | Epoxy resin composition for cast molding, and method for producing molded product for high voltage equipment using the same |
| US9862798B2 (en) * | 2013-09-30 | 2018-01-09 | Evonik Degussa Gmbh | Epoxy liquid curing agent compositions |
-
2016
- 2016-02-12 PL PL16704432.0T patent/PL3275002T5/en unknown
- 2016-02-12 WO PCT/EP2016/052969 patent/WO2016150614A1/en active Application Filing
- 2016-02-12 CN CN201680018184.1A patent/CN107531884B/en active Active
- 2016-02-12 KR KR1020177022791A patent/KR102580662B1/en active IP Right Grant
- 2016-02-12 MX MX2017012199A patent/MX2017012199A/en unknown
- 2016-02-12 US US15/561,918 patent/US20180112031A1/en active Pending
- 2016-02-12 EP EP16704432.0A patent/EP3275002B2/en active Active
- 2016-02-12 CA CA2976825A patent/CA2976825C/en active Active
- 2016-02-12 JP JP2017545303A patent/JP6683721B2/en active Active
- 2016-02-12 ES ES16704432T patent/ES2739688T5/en active Active
- 2016-03-24 TW TW105109160A patent/TWI777917B/en active
-
2019
- 2019-07-11 HR HRP20191244TT patent/HRP20191244T4/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0839865A1 (en) * | 1996-10-30 | 1998-05-06 | Ciba SC Holding AG | Curable epoxy resin compositions |
| WO2009062543A1 (en) * | 2007-11-13 | 2009-05-22 | Abb Research Ltd | Fiber-reinforced composite system as electrical insulation |
| WO2010010048A1 (en) * | 2008-07-22 | 2010-01-28 | Basf Se | Blends containing epoxy resins and mixtures of amines with guanidine derivatives |
| WO2013124251A2 (en) * | 2012-02-22 | 2013-08-29 | Basf Se | Blends for composites |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2018140576A1 (en) * | 2017-01-26 | 2018-08-02 | Huntsman Advanced Materials Licensing (Switzerland) Gmbh | A thermosetting epoxy resin composition for the preparation of articles for electrical engineering, and the articles obtained therefrom |
| CN110214156A (en) * | 2017-01-26 | 2019-09-06 | 亨斯迈先进材料许可(瑞士)有限公司 | It is used to prepare the thermosetting epoxy resin composition of electrical engineering product, and the product obtained by it |
| KR20190104423A (en) * | 2017-01-26 | 2019-09-09 | 훈츠만 어드밴스트 머티리얼스 라이센싱 (스위처랜드) 게엠베하 | Thermosetting epoxy resin composition and product obtained therefrom for the manufacture of electrical engineering products |
| EP3574034A4 (en) * | 2017-01-26 | 2020-11-18 | Huntsman Advanced Materials Licensing (Switzerland) GmbH | A thermosetting epoxy resin composition for the preparation of articles for electrical engineering, and the articles obtained therefrom |
| US11525054B2 (en) | 2017-01-26 | 2022-12-13 | Huntsman Advanced Materials Licensing (CH) GmbH | Thermosetting epoxy resin composition for the preparation of articles for electrical engineering, and the articles obtained therefrom |
| KR102530214B1 (en) * | 2017-01-26 | 2023-05-10 | 훈츠만 어드밴스트 머티리얼스 라이센싱 (스위처랜드) 게엠베하 | Thermosetting epoxy resin composition for the manufacture of products for electrical engineering and products obtained therefrom |
| CN109923145A (en) * | 2017-02-06 | 2019-06-21 | 亨斯迈石油化学有限责任公司 | Curing agent for epoxy resin |
| CN108215233A (en) * | 2017-12-11 | 2018-06-29 | 湖北耐创新材料洁具有限公司 | A kind of pressure molding of resin and mineral mixing pouring piece |
| CN108189423A (en) * | 2017-12-29 | 2018-06-22 | 江苏神马电力股份有限公司 | Compound insulation tube liner and its preparation method and application |
| WO2023114201A3 (en) * | 2021-12-14 | 2023-07-27 | Huntsman Petrochemical Llc | Polyurethane composition |
| RU2787124C1 (en) * | 2022-08-29 | 2022-12-28 | Российская Федерация, от имени которой выступает Государственная корпорация по атомной энергии "Росатом" (Госкорпорация "Росатом") | Electrical insulating pilling and impregnation compound |
Also Published As
| Publication number | Publication date |
|---|---|
| PL3275002T5 (en) | 2023-06-19 |
| MX2017012199A (en) | 2017-12-15 |
| HRP20191244T1 (en) | 2019-10-04 |
| JP2018512471A (en) | 2018-05-17 |
| EP3275002B1 (en) | 2019-06-12 |
| PL3275002T3 (en) | 2020-01-31 |
| KR20170130369A (en) | 2017-11-28 |
| EP3275002A1 (en) | 2018-01-31 |
| EP3275002B2 (en) | 2022-01-19 |
| ES2739688T5 (en) | 2022-05-11 |
| KR102580662B1 (en) | 2023-09-21 |
| JP6683721B2 (en) | 2020-04-22 |
| HRP20191244T4 (en) | 2022-03-04 |
| CA2976825A1 (en) | 2016-09-29 |
| CN107531884A (en) | 2018-01-02 |
| ES2739688T3 (en) | 2020-02-03 |
| CA2976825C (en) | 2023-04-18 |
| CN107531884B (en) | 2021-04-20 |
| TW201638204A (en) | 2016-11-01 |
| US20180112031A1 (en) | 2018-04-26 |
| TWI777917B (en) | 2022-09-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3274390B1 (en) | A thermosetting epoxy resin composition for the preparation of outdoor articles, and the articles obtained therefrom | |
| CA2976825C (en) | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof | |
| CA2983679C (en) | A curing agent for thermosetting epoxy resins, and a process for the preparation of insulation systems for electrical engineering | |
| CA3016634A1 (en) | A process for the preparation of insulation systems for electrical engineering, the articles obtained therefrom and the use thereof | |
| KR102530214B1 (en) | Thermosetting epoxy resin composition for the manufacture of products for electrical engineering and products obtained therefrom |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 16704432 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2976825 Country of ref document: CA Ref document number: 20177022791 Country of ref document: KR Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 2017545303 Country of ref document: JP Kind code of ref document: A |
|
| REEP | Request for entry into the european phase |
Ref document number: 2016704432 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2017/012199 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15561918 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |