WO2016125330A1 - Retinal regeneration promoting drug - Google Patents
Retinal regeneration promoting drug Download PDFInfo
- Publication number
- WO2016125330A1 WO2016125330A1 PCT/JP2015/074162 JP2015074162W WO2016125330A1 WO 2016125330 A1 WO2016125330 A1 WO 2016125330A1 JP 2015074162 W JP2015074162 W JP 2015074162W WO 2016125330 A1 WO2016125330 A1 WO 2016125330A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- granulin
- retinal
- grn1
- regeneration
- zebrafish
- Prior art date
Links
- 230000008929 regeneration Effects 0.000 title claims abstract description 38
- 238000011069 regeneration method Methods 0.000 title claims abstract description 38
- 230000002207 retinal effect Effects 0.000 title claims abstract description 38
- 230000001737 promoting effect Effects 0.000 title claims abstract description 7
- 239000003814 drug Substances 0.000 title abstract description 10
- 229940079593 drug Drugs 0.000 title abstract description 8
- 108060003393 Granulin Proteins 0.000 claims abstract description 64
- 102000017941 granulin Human genes 0.000 claims abstract description 64
- 210000000440 neutrophil Anatomy 0.000 claims abstract description 38
- YIEDSISPYKQADU-UHFFFAOYSA-N n-acetyl-n-[2-methyl-4-[(2-methylphenyl)diazenyl]phenyl]acetamide Chemical compound C1=C(C)C(N(C(C)=O)C(=O)C)=CC=C1N=NC1=CC=CC=C1C YIEDSISPYKQADU-UHFFFAOYSA-N 0.000 claims abstract description 37
- 239000012228 culture supernatant Substances 0.000 claims abstract description 12
- 239000012634 fragment Substances 0.000 claims abstract description 11
- 101100337797 Schizosaccharomyces pombe (strain 972 / ATCC 24843) grn1 gene Proteins 0.000 claims description 53
- 241000252212 Danio rerio Species 0.000 claims description 30
- 208000014674 injury Diseases 0.000 claims description 16
- 238000000034 method Methods 0.000 claims description 16
- 208000017442 Retinal disease Diseases 0.000 claims description 14
- 239000003795 chemical substances by application Substances 0.000 claims description 14
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 12
- 201000010099 disease Diseases 0.000 claims description 11
- 208000002780 macular degeneration Diseases 0.000 claims description 9
- 101001027324 Homo sapiens Progranulin Proteins 0.000 claims description 7
- 102000054121 human GRN Human genes 0.000 claims description 7
- 208000010412 Glaucoma Diseases 0.000 claims description 6
- 206010064930 age-related macular degeneration Diseases 0.000 claims description 6
- 208000034461 Progressive cone dystrophy Diseases 0.000 claims description 5
- 201000008615 cone dystrophy Diseases 0.000 claims description 5
- 201000004569 Blindness Diseases 0.000 claims description 4
- 206010005176 Blindness congenital Diseases 0.000 claims description 4
- 208000016615 Central areolar choroidal dystrophy Diseases 0.000 claims description 4
- 206010008025 Cerebellar ataxia Diseases 0.000 claims description 4
- 208000033810 Choroidal dystrophy Diseases 0.000 claims description 4
- 206010012689 Diabetic retinopathy Diseases 0.000 claims description 4
- 101001027325 Mus musculus Progranulin Proteins 0.000 claims description 4
- 208000007014 Retinitis pigmentosa Diseases 0.000 claims description 4
- 208000010112 Spinocerebellar Degenerations Diseases 0.000 claims description 4
- 208000027073 Stargardt disease Diseases 0.000 claims description 4
- 230000008733 trauma Effects 0.000 claims description 4
- 208000035719 Maculopathy Diseases 0.000 claims description 3
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 claims description 3
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 claims description 3
- 230000004393 visual impairment Effects 0.000 claims 1
- 210000001525 retina Anatomy 0.000 description 29
- 210000004027 cell Anatomy 0.000 description 26
- 101100110025 Danio rerio ascl1a gene Proteins 0.000 description 16
- 208000027418 Wounds and injury Diseases 0.000 description 12
- 230000006378 damage Effects 0.000 description 12
- 238000002360 preparation method Methods 0.000 description 11
- 230000008569 process Effects 0.000 description 10
- WOVKYSAHUYNSMH-RRKCRQDMSA-N 5-bromodeoxyuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(Br)=C1 WOVKYSAHUYNSMH-RRKCRQDMSA-N 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 8
- 108020004999 messenger RNA Proteins 0.000 description 8
- 210000000274 microglia Anatomy 0.000 description 8
- 210000000608 photoreceptor cell Anatomy 0.000 description 8
- 108090000623 proteins and genes Proteins 0.000 description 8
- 102000019204 Progranulins Human genes 0.000 description 7
- 108010012809 Progranulins Proteins 0.000 description 7
- 230000003247 decreasing effect Effects 0.000 description 7
- 230000004069 differentiation Effects 0.000 description 7
- 230000000694 effects Effects 0.000 description 7
- 210000000130 stem cell Anatomy 0.000 description 7
- 206010057430 Retinal injury Diseases 0.000 description 6
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 6
- 239000003550 marker Substances 0.000 description 6
- 238000003757 reverse transcription PCR Methods 0.000 description 6
- 108020005004 Guide RNA Proteins 0.000 description 5
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- 125000003275 alpha amino acid group Chemical group 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- -1 sorbit Chemical compound 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 4
- 210000005156 Müller Glia Anatomy 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 210000005252 bulbus oculi Anatomy 0.000 description 4
- 239000003889 eye drop Substances 0.000 description 4
- 210000004498 neuroglial cell Anatomy 0.000 description 4
- 230000035755 proliferation Effects 0.000 description 4
- WVDDGKGOMKODPV-UHFFFAOYSA-N Benzyl alcohol Chemical compound OCC1=CC=CC=C1 WVDDGKGOMKODPV-UHFFFAOYSA-N 0.000 description 3
- 208000037312 Familial drusen Diseases 0.000 description 3
- 239000012981 Hank's balanced salt solution Substances 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 241001465754 Metazoa Species 0.000 description 3
- 230000009471 action Effects 0.000 description 3
- 230000000692 anti-sense effect Effects 0.000 description 3
- 230000032459 dedifferentiation Effects 0.000 description 3
- 239000012153 distilled water Substances 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 238000012744 immunostaining Methods 0.000 description 3
- 238000007901 in situ hybridization Methods 0.000 description 3
- 239000007758 minimum essential medium Substances 0.000 description 3
- 108091008695 photoreceptors Proteins 0.000 description 3
- 238000003753 real-time PCR Methods 0.000 description 3
- 230000001172 regenerating effect Effects 0.000 description 3
- 239000000523 sample Substances 0.000 description 3
- 230000004304 visual acuity Effects 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 2
- 208000036443 AIPL1-related retinopathy Diseases 0.000 description 2
- 102000009027 Albumins Human genes 0.000 description 2
- 108010088751 Albumins Proteins 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 208000002339 Frontotemporal Lobar Degeneration Diseases 0.000 description 2
- 201000011240 Frontotemporal dementia Diseases 0.000 description 2
- 229930091371 Fructose Natural products 0.000 description 2
- 239000005715 Fructose Substances 0.000 description 2
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 206010038923 Retinopathy Diseases 0.000 description 2
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 2
- 239000002253 acid Substances 0.000 description 2
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 description 2
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 201000006754 cone-rod dystrophy Diseases 0.000 description 2
- 238000012790 confirmation Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- LVTYICIALWPMFW-UHFFFAOYSA-N diisopropanolamine Chemical compound CC(O)CNCC(C)O LVTYICIALWPMFW-UHFFFAOYSA-N 0.000 description 2
- 229940043276 diisopropanolamine Drugs 0.000 description 2
- 238000004520 electroporation Methods 0.000 description 2
- 239000008103 glucose Substances 0.000 description 2
- 238000003125 immunofluorescent labeling Methods 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 239000007951 isotonicity adjuster Substances 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 2
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 2
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 2
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 2
- 229960002216 methylparaben Drugs 0.000 description 2
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 2
- 210000005155 neural progenitor cell Anatomy 0.000 description 2
- 239000003002 pH adjusting agent Substances 0.000 description 2
- 239000002504 physiological saline solution Substances 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- 108090000765 processed proteins & peptides Proteins 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000002560 therapeutic procedure Methods 0.000 description 2
- 239000000811 xylitol Substances 0.000 description 2
- 235000010447 xylitol Nutrition 0.000 description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 2
- 229960002675 xylitol Drugs 0.000 description 2
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 102000014914 Carrier Proteins Human genes 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 241000699800 Cricetinae Species 0.000 description 1
- ZGTMUACCHSMWAC-UHFFFAOYSA-L EDTA disodium salt (anhydrous) Chemical compound [Na+].[Na+].OC(=O)CN(CC([O-])=O)CCN(CC(O)=O)CC([O-])=O ZGTMUACCHSMWAC-UHFFFAOYSA-L 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 108010028275 Leukocyte Elastase Proteins 0.000 description 1
- 102000016799 Leukocyte elastase Human genes 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 102000002274 Matrix Metalloproteinases Human genes 0.000 description 1
- 108010000684 Matrix Metalloproteinases Proteins 0.000 description 1
- 101100102907 Mus musculus Wdtc1 gene Proteins 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 241000286209 Phasianidae Species 0.000 description 1
- 229920002701 Polyoxyl 40 Stearate Polymers 0.000 description 1
- 108010076504 Protein Sorting Signals Proteins 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 229940050528 albumin Drugs 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000003146 anticoagulant agent Substances 0.000 description 1
- 229940127219 anticoagulant drug Drugs 0.000 description 1
- 235000019445 benzyl alcohol Nutrition 0.000 description 1
- 108091008324 binding proteins Proteins 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000005018 casein Substances 0.000 description 1
- BECPQYXYKAMYBN-UHFFFAOYSA-N casein, tech. Chemical compound NCCCCC(C(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(CC(C)C)N=C(O)C(CCC(O)=O)N=C(O)C(CC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(C(C)O)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=N)N=C(O)C(CCC(O)=O)N=C(O)C(CCC(O)=O)N=C(O)C(COP(O)(O)=O)N=C(O)C(CCC(O)=N)N=C(O)C(N)CC1=CC=CC=C1 BECPQYXYKAMYBN-UHFFFAOYSA-N 0.000 description 1
- 235000021240 caseins Nutrition 0.000 description 1
- 210000003169 central nervous system Anatomy 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 230000008045 co-localization Effects 0.000 description 1
- 238000011443 conventional therapy Methods 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 208000035475 disorder Diseases 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 210000000416 exudates and transudate Anatomy 0.000 description 1
- 210000001508 eye Anatomy 0.000 description 1
- 239000003885 eye ointment Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 238000000265 homogenisation Methods 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 101150108076 lin28a gene Proteins 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 230000001617 migratory effect Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000010369 molecular cloning Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 230000004770 neurodegeneration Effects 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 229930192033 plastin Natural products 0.000 description 1
- 108010049148 plastin Proteins 0.000 description 1
- 210000001778 pluripotent stem cell Anatomy 0.000 description 1
- 229940099429 polyoxyl 40 stearate Drugs 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 235000018102 proteins Nutrition 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 210000003994 retinal ganglion cell Anatomy 0.000 description 1
- 210000001164 retinal progenitor cell Anatomy 0.000 description 1
- 238000013341 scale-up Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 229940037001 sodium edetate Drugs 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 238000002636 symptomatic treatment Methods 0.000 description 1
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 1
- 238000000108 ultra-filtration Methods 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 230000004382 visual function Effects 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/14—Blood; Artificial blood
- A61K35/15—Cells of the myeloid line, e.g. granulocytes, basophils, eosinophils, neutrophils, leucocytes, monocytes, macrophages or mast cells; Myeloid precursor cells; Antigen-presenting cells, e.g. dendritic cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Definitions
- the present invention relates to a pharmaceutical effective for retina regeneration. Specifically, the present invention relates to a retina regeneration accelerator and its use.
- This application claims priority based on Japanese Patent Application No. 2015-018857 filed on February 2, 2015, the entire contents of which are incorporated by reference.
- the retina plays an important role in visual function, and the disorder causes various ophthalmic diseases such as glaucoma and age-related macular degeneration. Usually, since the retina does not regenerate, the treatment of ophthalmic diseases caused by retinal disorders must rely on symptomatic therapy.
- Human granulin is an 88 kDa precursor protein (progranulin) with 7.5 highly conserved granulin motifs (CX5-6CX5CCX8CCX6CCXDX2HCCPX4CX5-6C) containing 12 cysteine residues. (See FIG. 1).
- the signal peptide is cleaved into mature granulin, which is further cleaved to have peptides with various activities of about 6 kDa (granulin A, B, C, D, E, F, G, and P corresponding to 0.5). )become.
- enzymes that cleave granulin include matrix metalloproteinases and neutrophil elastase.
- Non-patent Document 1 Progranulin mutations are also involved in frontotemporal lobar degeneration (FTLD) and amyotrophic lateral sclerosis (ALS) and are important factors in neurodegenerative diseases (non- Patent Documents 2 and 3).
- FTLD frontotemporal lobar degeneration
- ALS amyotrophic lateral sclerosis
- TNF receptor has been identified as a binding protein for progranulin, and has attracted attention (Non-patent Document 4).
- An object of the present invention is to establish a new therapeutic means for ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders such as glaucoma and age-related macular degeneration (agent for promoting retinal regeneration) (Medicine) is to provide.
- Non-patent Document 5 Korean Patent Document 5
- Progranulin had a photoreceptor-protecting effect similar to adipose stem cells.
- the ratio of photoreceptor cells increased when the culture supernatant of adipose stem cells was added to primary retinal culture cells (announced at the 34th Annual Meeting of the Japanese Eye Pharmacology Society (Gifu, 2014.9.13-14)).
- the culture supernatant of adipose stem cells contains a factor that promotes differentiation into photoreceptor cells.
- the retina regeneration process roughly divided, it is necessary to proceed with two steps, namely, proliferation of retinal stem cells (retinal progenitor cells) and differentiation of retinal stem cells into photoreceptor cells. Therefore, promotion of differentiation into photoreceptor cells is important for regenerating the retina, but is not definitive.
- the present inventors focused on progranulin which was shown to promote differentiation into photoreceptor cells, and decided to investigate the details of its involvement in the regeneration process.
- mammals such as mice and humans do not regenerate the retina and central nervous system, and research on retinal regeneration is mainly performed using zebrafish, so we decided to use zebrafish to proceed with the study.
- zebrafish There are four subtypes of zebrafish: granulin A (grnA) with 10 granulin motifs, granulin B (grnB) with 9 granulins, and granulin 1 (grn1) and 2 (grn2) with 1.5 granulin motifs. .
- grnA is considered to be an ortholog of human progranulin and is expressed in microglia of the zebrafish retinal photopathy model.
- GrnA is also involved in the differentiation and regeneration of zebrafish muscle. It is known that when the zebrafish retina is damaged, the Muller glia cells in the retina are dedifferentiated and differentiated into various cells such as photoreceptors and retinal ganglion cells to compensate for the damaged site and regenerate. It has been.
- Retinal regeneration-promoting drug comprising a component selected from the group consisting of the following (1) and (2): (1) a granulin molecule, or a fragment thereof containing a granulin motif; (2) Neutrophils or culture supernatants thereof.
- the granulin molecule is zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin, and the fragment is granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G
- the retinal regeneration-promoting agent according to [1] which is granulin P.
- Glaucoma age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (cone rod dystrophy), Stargardt disease, macular
- the retina according to any one of [1] to [3], which is used for treatment of a disease selected from the group consisting of dystrophy, familial drusen, central ring-shaped choroidal dystrophy, and spinocerebellar degeneration type 7 Regeneration promoter.
- a therapeutic method comprising the step of administering a therapeutically effective amount of a component selected from the group consisting of the following (1) and (2) to a patient with an ophthalmic disease associated with a retinal disorder: (1) a granulin molecule, or a fragment thereof containing a granulin motif; (2) Neutrophils or culture supernatants thereof.
- the present invention relates to a drug (retinal regeneration promoting drug) that promotes retina regeneration and exerts a therapeutic effect.
- a drug receptor regeneration promoting drug
- a component constituting the drug of the present invention (1) a granulin molecule or a fragment thereof containing a granulin motif, or (2) a neutrophil or a culture supernatant thereof is used.
- the component (1) and the component (2) may be used in combination.
- the component (1) when the component (1) is administered to a living body, it acts on neutrophils, or acts on a factor secreted by neutrophils to increase the expression of ascl1, retina-specifically Muller glial cells, which are glial cells, proliferate, dedifferentiate into retinal neural progenitor cells, differentiate into photoreceptor cells, etc., and retina regeneration is promoted.
- the component (2) when the component (2) is administered to a living body, it can be expected to directly act to cause proliferation of Muller glial cells, dedifferentiation into retinal neural progenitor cells, differentiation into photoreceptor cells, etc. . If the component (1) and the component (2) are used in combination, in addition to the action effect mediated by neutrophils present in the living body, the action effect that does not involve neutrophils present in the living body can be exhibited, and the therapeutic effect I can expect an increase.
- zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin can be used as the granulin molecule.
- granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G or granulin P which are degradation products of human progranulin, can be used.
- granulin A and granulin F are particularly preferred components.
- the attached sequence listing shows the amino acid sequence of zebrafish grn1 (SEQ ID NO: 1) and the gene sequence encoding it (SEQ ID NO: 2), the amino acid sequence of zebrafish grn2 (SEQ ID NO: 3.
- the amino acid sequence of the variant is SEQ ID NO: 5 )
- the gene sequence encoding it SEQ ID NO: 4
- the amino acid sequence of human progranulin SEQ ID NO: 6
- the gene sequence encoding it SEQ ID NO: 7
- amino acid sequence of mouse progranulin mouse progranulin
- mouse grn1 SEQ ID NO: 8
- SEQ ID NO: 9 The attached sequence listing shows the amino acid sequence of zebrafish grn1 (SEQ ID NO: 1) and the gene sequence encoding it (SEQ ID NO: 2), the amino acid sequence of zebrafish grn2 (SEQ ID NO: 3.
- the amino acid sequence of the variant is SEQ ID NO: 5
- the gene sequence encoding it SEQ ID NO: 4
- Granulin molecules or fragments thereof can also be prepared by separating and purifying from a living body, but preferably as a recombinant (recombinant protein) from the viewpoint of homogenization of quality, preparation operation, scale-up, etc.
- a granulin molecule or fragment thereof is prepared.
- Preparation operations can be performed in a conventional manner, for example, Molecular Cloning (Third Edition, Cold Spring Harbor Laboratory Press, New York), Current protocols in molecular biology (edited by Frederick M. Ausubel et al., 1987) become.
- Neutrophils can be prepared by conventional methods. As specific examples of the method for preparing neutrophils, a method for preparing mouse active neutrophils and a method for preparing human neutrophils are described below.
- HBSS Horte' balanced salt solution
- culture may be performed at 37 ° C. and 5% CO 2 in RPMI1640 medium containing 10% FBS.
- the culture supernatant of neutrophils can be obtained by culturing the prepared neutrophils in vitro.
- prepared mouse or human neutrophils are seeded in a 10 cm petri dish (BD) or flask (25 cm 2 ) at 40,000 cells / ml, and cultured in HEPES-MEM or RPMI1640 without FBS for 12 to 24 hours. . Thereafter, the supernatant is collected, centrifuged at 300 ⁇ g for 5 minutes, and then sterilized by filtration using a 0.22 ⁇ m sterilizing filter (Millipore).
- the culture supernatant is concentrated by ultrafiltration (eg using Amicon Ultra-15 (Millipore) (fractionated molecular weight 3,000)).
- the retinal regeneration-promoting agent of the present invention can be prepared as an injection for intravitreal administration, an eye drop or an eye ointment according to a conventional method.
- An injection for intravitreal administration can be produced, for example, by suspending or dissolving the active ingredient in an appropriate solvent (for example, distilled water for injection or physiological saline).
- an appropriate solvent for example, distilled water for injection or physiological saline.
- isotonic agents such as mannitol, sodium chloride, glucose, sorbit, glycerol, xylitol, fructose, maltose, mannose, stabilizers such as albumin, preservatives such as benzyl alcohol and methyl parahydroxybenzoate
- an acid such as citric acid and a base such as diisopropanolamine can be added to the preparation as a pH adjuster.
- An eye drop can be produced, for example, by dissolving the above active ingredient in an appropriate solvent (for example, distilled water for injection or physiological saline).
- an appropriate solvent for example, distilled water for injection or physiological saline.
- isotonic agents such as mannitol, sodium chloride, glucose, sorbit, glycerol, xylitol, fructose, maltose, mannose, glycerin, stabilizers such as sodium edetate, albumin, benzyl alcohol, methyl parahydroxybenzoate, etc.
- a preservative, a surfactant such as polyoxyethylene monooleate, polyoxyl 40 stearate and the like can be added to the preparation.
- an acid such as citric acid and a base such as diisopropanolamine can be added to the preparation as a pH adjuster.
- the pH of the eye drop is not particularly limited as long as it is within the range acceptable for ophthalmic preparations, but is preferably 5.0 to 8.5.
- the retinal regeneration-promoting agent of the present invention is used for the treatment of various ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders (in other words, retinal regeneration has a therapeutic effect).
- ophthalmic disease to be treated with the retinal regeneration-promoting agent of the present invention glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, Cone dystrophy (cone rod dystrophy), Stargardt disease, macular dystrophy, familial drusen, central crested choroidal dystrophy and spinocerebellar degeneration 7 types.
- it is particularly effective for diseases in which degeneration occurs in photoreceptor cells.
- the dose (use amount) of the retinal regeneration-promoting agent of the present invention may be appropriately set in consideration of the patient's condition, age, weight, dosage form, and the like.
- an appropriate amount of the preparation containing 0.01 to 70% by weight of the active ingredient may be administered once a month.
- the preparation containing 0.01 to 70% by weight of the active ingredient may be instilled 1 to several times a day once to several drops.
- the treatment target of the retinal regeneration-promoting agent of the present invention is not particularly limited, and humans and non-human mammals (pet animals, domestic animals, laboratory animals, etc., specifically, for example, guinea pigs, hamsters, monkeys, cows, pigs, goats, Sheep, dogs, cats, chickens, quails, etc.).
- the retinal regeneration-promoting agent of the present invention is applied to humans.
- the present application includes glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (cone) (Body dystrophy), Stargardt disease, macular dystrophy, familial drusen, central annulus choroidal dystrophy, spinocerebellar degeneration 7 and the like, a therapeutically effective amount of the retinal regeneration promoter of the present invention is administered.
- a featured treatment is also provided.
- Method A 30G needle was inserted into the eyeball of a zebrafish (AB line), and the retina was damaged in 4 to 8 places. Thereafter, the eyeballs were removed between 6 hours and 7 days, and frozen sections were prepared. In addition, RNA was prepared after the retina was isolated. The frozen section was used to identify the expression site of grn1 mRNA by in situ hybridization. Simultaneously, immunostaining was performed using an anti-4C4 antibody (microglia marker) and an antiplastin antibody (microglia and neutrophil marker).
- RNA was used for measuring the expression levels of grn1, grn2, grnA, grnB and ascl1a mRNA by PCR.
- a morpholino oligo capable of specifically knocking down the expression of the zebrafish gene was prepared and introduced into the retina by electroporation. After introduction, BrdU was administered intraperitoneally to zebrafish. After this treatment, frozen sections were prepared and RNA was prepared. Using frozen sections, BrdU positive cells were quantified as cells in the regeneration process (using anti-BrdU antibody). RNA was used for quantification of the expression level of the gene ascl11, which is an indicator of zebrafish retinal regeneration.
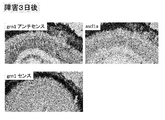
- grn1 Since the expression of grn1 was increased in the zebrafish retinal disorder model, it was decided to confirm the expression site by in situ hybridization. When examined using the grn1 antisense probe, positive cells with strong expression were observed around the damaged site 15 hours after the injury, and were confirmed even 24 hours later (FIG. 4). On the third day of injury, grn1 positivity was observed in Muller glia (FIG. 5). When immunostaining was performed using an anti-4C4 antibody that is a marker of microglia, colocalization with grn1-positive cells was hardly observed (FIG. 4). When the damaged site was immunostained with anti-4C4 antibody and antiplastin antibody (microglia and neutrophil markers), 4C4 negative / plastin positive cells were observed (FIG. 4). The shape of these cells resembled grn1-positive cells. From the above results, it was found that grn1 is highly expressed in retinal damage sites (especially neutrophils) in zebrafish.
- grn1 MO antisense morpholino-oligo
- RT-PCR was used to examine whether there was a change in the expression of ascl1a, which is essential for retinal regeneration. As a result, a decrease in ascl1a was observed in the retina into which grn1 MO was introduced 15 hours after injury (Fig. 6).
- grn1 is also expressed in retinal cells other than neutrophils including Muller glia, even if they are knocked down with grn1 MO, there was no change in the expression level of ascl1a. These results suggest that grn1 cleaved by neutrophil-specific enzymes may regulate the expression level of ascla1.
- zebrafish grn1 mRNA was cloned, a 6xHis tag sequence was added to the 3 'side, and then expressed in vitro to synthesize the grn1 protein. Purification was performed using a column for His tag and a column for buffer replacement to obtain recombinant grn1. 200 ng recombinant grn1 or vehicle was administered to the vitreous of the zebrafish retinal disorder model. Two days later, the eyeballs were removed, retinal mRNA was prepared, and the increase or decrease in retinal regeneration-related factors was confirmed by RT-PCR.
- BrdU was intraperitoneally administered, and the eyeball was removed to prepare a frozen section. Immunofluorescence staining was performed with an anti-BrdU antibody, and the presence or absence of BrdU positive cells was observed with a fluorescence microscope.
- the retinal regeneration-promoting agent of the present invention can be applied to the treatment of various ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders.
- Regenerative medicine using artificial pluripotent stem cells (iPS cells) or the like has attracted attention as a therapeutic means to replace conventional symptomatic treatment.
- the present invention increases technical options for regenerative medicine, and can be used and applied as an alternative or supplement to conventional therapies.
Abstract
The present invention addresses the problem of providing a drug capable of promoting retinal regeneration. Provided is a retinal regeneration promoting drug comprising a component selected from the group consisting of (1) a granulin molecule or a fragment thereof which contains a granulin motif and (2) a neutrophil or a culture supernatant thereof.
Description
本発明は網膜の再生に有効な医薬に関する。詳しくは、網膜の再生促進薬及びその用途に関する。本出願は、2015年2月2日に出願された日本国特許出願第2015-018857号に基づく優先権を主張するものであり、当該特許出願の全内容は参照により援用される。
The present invention relates to a pharmaceutical effective for retina regeneration. Specifically, the present invention relates to a retina regeneration accelerator and its use. This application claims priority based on Japanese Patent Application No. 2015-018857 filed on February 2, 2015, the entire contents of which are incorporated by reference.
網膜は視機能に関して重要な役割を果たしており、その障害は緑内障や加齢黄斑変性症をはじめとした様々な眼科疾患の原因となる。通常、網膜は再生しないことから、網膜の障害に起因する眼科疾患の治療は対症療法に頼らざるを得ないのが現状である。
The retina plays an important role in visual function, and the disorder causes various ophthalmic diseases such as glaucoma and age-related macular degeneration. Usually, since the retina does not regenerate, the treatment of ophthalmic diseases caused by retinal disorders must rely on symptomatic therapy.
ヒトグラニュリン(granulin; grn)は88kDaの前駆体蛋白質(プログラニュリン)であり、高度に保存された12個のシステイン残基を含んだグラニュリンモチーフ(CX5-6CX5CCX8CCX6CCXDX2HCCPX4CX5-6C)を7.5個有している(図1を参照)。シグナルペプチドが切断されて成熟したグラニュリンとなり、グラニュリンはさらに切断されて、約6kDaの様々な活性を有するペプチド(グラニュリンA, B, C, D, E, F, G,および0.5個に相当するP)になる。グラニュリンを切断する酵素としてマトリックスメタロプロテアーゼ群や好中球エラスターゼなどが知られている。これらのペプチドやインタクトなグラニュリンは成長因子として細胞成長を制御している。しかし、これらグラニュリンファミリーは細胞成長において刺激作用または阻害作用を発揮し、さらに両方の性質を有しているものもある。グラニュリンファミリーは通常の発生過程や、創傷治癒、がん、免疫応答などに関与している(非特許文献1)。また、プログラニュリンの変異は前頭側頭葉変性症(frontotemporal lobar degeneration;FTLD)や筋委縮性側索硬化症(ALS)に関与しており、神経変性疾患においても重要な因子である(非特許文献2、3)。近年ではプログラニュリンの結合蛋白質としてTNF受容体が同定されており、注目を集めている(非特許文献4)。
Human granulin (grn) is an 88 kDa precursor protein (progranulin) with 7.5 highly conserved granulin motifs (CX5-6CX5CCX8CCX6CCXDX2HCCPX4CX5-6C) containing 12 cysteine residues. (See FIG. 1). The signal peptide is cleaved into mature granulin, which is further cleaved to have peptides with various activities of about 6 kDa (granulin A, B, C, D, E, F, G, and P corresponding to 0.5). )become. Known examples of enzymes that cleave granulin include matrix metalloproteinases and neutrophil elastase. These peptides and intact granulin control cell growth as growth factors. However, these granulin families exert stimulating or inhibiting effects on cell growth, and some have both properties. The granulin family is involved in the normal development process, wound healing, cancer, immune response, etc. (Non-patent Document 1). Progranulin mutations are also involved in frontotemporal lobar degeneration (FTLD) and amyotrophic lateral sclerosis (ALS) and are important factors in neurodegenerative diseases (non- Patent Documents 2 and 3). In recent years, a TNF receptor has been identified as a binding protein for progranulin, and has attracted attention (Non-patent Document 4).
本発明の課題は、緑内障や加齢黄斑変性症等、網膜の障害に起因する又は網膜の障害を伴う眼科疾患に対する新たな治療手段を確立すべく、網膜の再生を促進する薬剤(網膜再生促進薬)を提供することにある。
An object of the present invention is to establish a new therapeutic means for ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders such as glaucoma and age-related macular degeneration (agent for promoting retinal regeneration) (Medicine) is to provide.
本発明者らの研究グループではマウスの脂肪幹細胞およびその培養上清に視細胞保護作用があることを見出し、培養上清中に豊富に存在する成分としてプログラニュリンを同定した(非特許文献5)。プログラニュリンは脂肪幹細胞と同様に視細胞保護作用を有していた。一方、脂肪幹細胞の培養上清を初代網膜培養細胞に添加すると、視細胞の割合が増えることを明らかにした(第34回日本眼薬理学会(岐阜、2014.9.13-14)で発表)。このことから、脂肪幹細胞の培養上清には視細胞への分化を促す因子が含まれていると考えられる。ここで、網膜の再生過程では、大別して二つのステップ、即ち、網膜幹細胞(網膜前駆細胞)の増殖と、網膜幹細胞の視細胞への分化、が進行する必要がある。従って、視細胞への分化促進は、網膜を再生させるために重要であるものの、決定的とはいえない。
Our research group found that mouse adipose stem cells and their culture supernatants have a protective effect on photoreceptors, and identified progranulin as an abundant component in the culture supernatant (Non-patent Document 5). ). Progranulin had a photoreceptor-protecting effect similar to adipose stem cells. On the other hand, it was clarified that the ratio of photoreceptor cells increased when the culture supernatant of adipose stem cells was added to primary retinal culture cells (announced at the 34th Annual Meeting of the Japanese Eye Pharmacology Society (Gifu, 2014.9.13-14)). From this, it is considered that the culture supernatant of adipose stem cells contains a factor that promotes differentiation into photoreceptor cells. Here, in the retina regeneration process, roughly divided, it is necessary to proceed with two steps, namely, proliferation of retinal stem cells (retinal progenitor cells) and differentiation of retinal stem cells into photoreceptor cells. Therefore, promotion of differentiation into photoreceptor cells is important for regenerating the retina, but is not definitive.
本発明者らは、視細胞への分化を促進することが示されたプログラニュリンに着目し、再生過程におけるその関与の詳細を調べることにした。通常、マウスやヒトなどの哺乳類では網膜や中枢神経の再生が起こらず、網膜再生の研究は主にゼブラフィッシュを用いて行われていることから、検討を進めるにあたってゼブラフィッシュを利用することにした。ゼブラフィッシュにはグラニュリンモチーフを10個有するグラニュリンA(grnA)、同9個有するグラニュリンB(grnB)、同1.5個有するグラニュリン1(grn1)及び2(grn2)の4種類のサブタイプが存在する。このうち、grnAはヒトプログラニュリンのオルソログであると考えられており、ゼブラフィッシュ網膜光障害モデルのミクログリアで発現が認められる。また、grnAはゼブラフィッシュの筋肉の分化や再生に関与している。尚、ゼブラフィッシュの網膜は障害を受けると網膜内のミュラーグリア細胞が脱分化を起こし、視細胞や網膜神経節細胞などの各種細胞へと分化して、損傷した部位を補い再生することが知られている。
The present inventors focused on progranulin which was shown to promote differentiation into photoreceptor cells, and decided to investigate the details of its involvement in the regeneration process. Usually, mammals such as mice and humans do not regenerate the retina and central nervous system, and research on retinal regeneration is mainly performed using zebrafish, so we decided to use zebrafish to proceed with the study. . There are four subtypes of zebrafish: granulin A (grnA) with 10 granulin motifs, granulin B (grnB) with 9 granulins, and granulin 1 (grn1) and 2 (grn2) with 1.5 granulin motifs. . Of these, grnA is considered to be an ortholog of human progranulin and is expressed in microglia of the zebrafish retinal photopathy model. GrnA is also involved in the differentiation and regeneration of zebrafish muscle. It is known that when the zebrafish retina is damaged, the Muller glia cells in the retina are dedifferentiated and differentiated into various cells such as photoreceptors and retinal ganglion cells to compensate for the damaged site and regenerate. It has been.
詳細な検討の結果、まず、ゼブラフィッシュの網膜再生過程においてグラニュリン1(grn1)遺伝子の発現が非常に初期段階から上昇することが見出され、特に好中球で強い発現が認められることが明らかとなった。一方、好中球のgrn1の発現を抑制すると、網膜再生に最も重要な因子であるascl1aの発現が抑制され、また再生の指標となるBrdU陽性細胞数の大幅な減少が認められた。この実験結果は、好中球で発現しているgrn1が網膜再生過程に深く関与し、非常に重要な役割を担っていることを示唆する。当該知見に加え、(1)網膜再生過程においてgrn1以外のグラニュリン分子の発現も上昇した事実(後述の実施例)、(2)グラニュリン分子は共通のモチーフを持つこと、(3)グラニュリンモチーフ単独でも活性を示すこと(非特許文献1、6)を考え合わせると、グラニュリン分子又はグラニュリンモチーフが網膜の再生に有効であることを期待できる。一方、網膜の再生過程に好中球が関与することは、これまでに報告のない新たな知見であり、その意義は大きく、好中球又はその分泌物によって網膜再生を促進できる可能性を強く示唆する。
以下の発明は主として以上の成果及び考察に基づく。
[1]以下の(1)及び(2)からなる群より選択される成分を含む、網膜再生促進薬:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。
[2]前記グラニュリン分子がゼブラフィッシュgrn1、ゼブラフィッシュgrn2、ヒトプログラニュリン又はマウスプログラニュリンであり、前記断片がグラニュリンA、グラニュリンB、グラニュリンC、グラニュリンD、グラニュリンE、グラニュリンF、グラニュリンG又はグラニュリンPである、[1]に記載の網膜再生促進薬。
[3]前記グラニュリン分子がリコンビナントタンパク質である、[1]又は[2]に記載の網膜再生促進薬。
[4]緑内障、加齢黄斑変性症、網膜色素変性症、糖尿病網膜症、外傷による視力低下又は失明、Leber先天盲、白点状眼底、錐体ジストロフィ(錐体杆体ジストロフィ)、Stargardt病、黄斑ジストロフィ、家族性ドルーゼン、中心性輪紋状脈絡膜ジストロフィ及び脊髄小脳変性症7型からなる群より選択される疾患の治療用である、[1]~[3]のいずれか一項に記載の網膜再生促進薬。
[5]網膜の障害を伴う眼科疾患の患者に対して、以下の(1)及び(2)からなる群より選択される成分を治療上有効量投与するステップを含む、治療法:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。 As a result of detailed examination, it was found that the expression of granulin 1 (grn1) gene increased from the very early stage in the retina regeneration process of zebrafish, and it was clear that strong expression was observed particularly in neutrophils. It became. On the other hand, when the expression of grn1 in neutrophils was suppressed, the expression of ascl1a, which is the most important factor for retinal regeneration, was suppressed, and the number of BrdU positive cells, which serve as an index of regeneration, was significantly reduced. This experimental result suggests that grn1 expressed in neutrophils is deeply involved in the retinal regeneration process and plays a very important role. In addition to the above findings, (1) the fact that the expression of granulin molecules other than grn1 was also increased during the retina regeneration process (examples described later), (2) the granulin molecules have a common motif, (3) the granulin motif alone However, considering that it shows activity (Non-patentDocuments 1 and 6), it can be expected that the granulin molecule or granulin motif is effective for retina regeneration. On the other hand, the involvement of neutrophils in the regeneration process of the retina is a new finding that has not been reported so far, and its significance is significant, and the possibility that retinal regeneration can be promoted by neutrophils or their secretions is strongly enhanced. Suggest.
The following invention is mainly based on the above results and considerations.
[1] Retinal regeneration-promoting drug comprising a component selected from the group consisting of the following (1) and (2):
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof.
[2] The granulin molecule is zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin, and the fragment is granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G Alternatively, the retinal regeneration-promoting agent according to [1], which is granulin P.
[3] The retinal regeneration-promoting agent according to [1] or [2], wherein the granulin molecule is a recombinant protein.
[4] Glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (cone rod dystrophy), Stargardt disease, macular The retina according to any one of [1] to [3], which is used for treatment of a disease selected from the group consisting of dystrophy, familial drusen, central ring-shaped choroidal dystrophy, andspinocerebellar degeneration type 7 Regeneration promoter.
[5] A therapeutic method comprising the step of administering a therapeutically effective amount of a component selected from the group consisting of the following (1) and (2) to a patient with an ophthalmic disease associated with a retinal disorder:
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof.
以下の発明は主として以上の成果及び考察に基づく。
[1]以下の(1)及び(2)からなる群より選択される成分を含む、網膜再生促進薬:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。
[2]前記グラニュリン分子がゼブラフィッシュgrn1、ゼブラフィッシュgrn2、ヒトプログラニュリン又はマウスプログラニュリンであり、前記断片がグラニュリンA、グラニュリンB、グラニュリンC、グラニュリンD、グラニュリンE、グラニュリンF、グラニュリンG又はグラニュリンPである、[1]に記載の網膜再生促進薬。
[3]前記グラニュリン分子がリコンビナントタンパク質である、[1]又は[2]に記載の網膜再生促進薬。
[4]緑内障、加齢黄斑変性症、網膜色素変性症、糖尿病網膜症、外傷による視力低下又は失明、Leber先天盲、白点状眼底、錐体ジストロフィ(錐体杆体ジストロフィ)、Stargardt病、黄斑ジストロフィ、家族性ドルーゼン、中心性輪紋状脈絡膜ジストロフィ及び脊髄小脳変性症7型からなる群より選択される疾患の治療用である、[1]~[3]のいずれか一項に記載の網膜再生促進薬。
[5]網膜の障害を伴う眼科疾患の患者に対して、以下の(1)及び(2)からなる群より選択される成分を治療上有効量投与するステップを含む、治療法:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。 As a result of detailed examination, it was found that the expression of granulin 1 (grn1) gene increased from the very early stage in the retina regeneration process of zebrafish, and it was clear that strong expression was observed particularly in neutrophils. It became. On the other hand, when the expression of grn1 in neutrophils was suppressed, the expression of ascl1a, which is the most important factor for retinal regeneration, was suppressed, and the number of BrdU positive cells, which serve as an index of regeneration, was significantly reduced. This experimental result suggests that grn1 expressed in neutrophils is deeply involved in the retinal regeneration process and plays a very important role. In addition to the above findings, (1) the fact that the expression of granulin molecules other than grn1 was also increased during the retina regeneration process (examples described later), (2) the granulin molecules have a common motif, (3) the granulin motif alone However, considering that it shows activity (Non-patent
The following invention is mainly based on the above results and considerations.
[1] Retinal regeneration-promoting drug comprising a component selected from the group consisting of the following (1) and (2):
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof.
[2] The granulin molecule is zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin, and the fragment is granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G Alternatively, the retinal regeneration-promoting agent according to [1], which is granulin P.
[3] The retinal regeneration-promoting agent according to [1] or [2], wherein the granulin molecule is a recombinant protein.
[4] Glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (cone rod dystrophy), Stargardt disease, macular The retina according to any one of [1] to [3], which is used for treatment of a disease selected from the group consisting of dystrophy, familial drusen, central ring-shaped choroidal dystrophy, and
[5] A therapeutic method comprising the step of administering a therapeutically effective amount of a component selected from the group consisting of the following (1) and (2) to a patient with an ophthalmic disease associated with a retinal disorder:
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof.
本発明は、網膜の再生を促して治療効果を発揮する薬剤(網膜再生促進薬)に関する。本発明の薬剤を構成する成分として、(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片、或いは(2)好中球又はその培養上清が用いられる。(1)の成分及び(2)の成分を併用してもよい。理論に拘泥する訳ではないが、(1)の成分を生体に投与した場合、好中球に作用し、もしくは好中球が分泌する因子と作用してascl1の発現が上昇し、網膜特異的グリア細胞であるミュラーグリア細胞の増殖、網膜神経前駆細胞への脱分化、視細胞への分化等が生じ、網膜の再生が促される。他方、(2)の成分を生体に投与した場合には、それが直接作用して、ミュラーグリア細胞の増殖、網膜神経前駆細胞への脱分化、視細胞への分化等が生ずることを期待できる。(1)の成分と(2)の成分を併用すれば、生体に存在する好中球を介在した作用効果に加え、生体に存在する好中球を介在しない作用効果も発揮でき、治療効果の増大を望める。
The present invention relates to a drug (retinal regeneration promoting drug) that promotes retina regeneration and exerts a therapeutic effect. As a component constituting the drug of the present invention, (1) a granulin molecule or a fragment thereof containing a granulin motif, or (2) a neutrophil or a culture supernatant thereof is used. The component (1) and the component (2) may be used in combination. Without being bound by theory, when the component (1) is administered to a living body, it acts on neutrophils, or acts on a factor secreted by neutrophils to increase the expression of ascl1, retina-specifically Muller glial cells, which are glial cells, proliferate, dedifferentiate into retinal neural progenitor cells, differentiate into photoreceptor cells, etc., and retina regeneration is promoted. On the other hand, when the component (2) is administered to a living body, it can be expected to directly act to cause proliferation of Muller glial cells, dedifferentiation into retinal neural progenitor cells, differentiation into photoreceptor cells, etc. . If the component (1) and the component (2) are used in combination, in addition to the action effect mediated by neutrophils present in the living body, the action effect that does not involve neutrophils present in the living body can be exhibited, and the therapeutic effect I can expect an increase.
グラニュリン分子として、例えば、ゼブラフィッシュgrn1、ゼブラフィッシュgrn2、ヒトプログラニュリン又はマウスプログラニュリン(マウスgrn1)を用いることができる。一方、グラニュリン分子の断片としては、ヒトプログラニュリンの分解産物である、グラニュリンA、グラニュリンB、グラニュリンC、グラニュリンD、グラニュリンE、グラニュリンF、グラニュリンG又はグラニュリンPを用いることができる。中でも、グラニュリンAおよびグラニュリンFは特に好ましい成分といえる。尚、添付の配列表にゼブラフィッシュgrn1のアミノ酸配列(配列番号1)及びそれをコードする遺伝子配列(配列番号2)、ゼブラフィッシュgrn2のアミノ酸配列(配列番号3。バリアントのアミノ酸配列は配列番号5)及びそれをコードする遺伝子配列(配列番号4)、ヒトプログラニュリンのアミノ酸配列(配列番号6)及びそれをコードする遺伝子配列(配列番号7)及びマウスプログラニュリン(マウスgrn1)のアミノ酸配列(配列番号8)及びそれをコードする遺伝子配列(配列番号9)を示す。
As the granulin molecule, for example, zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin (mouse grn1) can be used. On the other hand, as a fragment of the granulin molecule, granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G or granulin P, which are degradation products of human progranulin, can be used. Of these, granulin A and granulin F are particularly preferred components. The attached sequence listing shows the amino acid sequence of zebrafish grn1 (SEQ ID NO: 1) and the gene sequence encoding it (SEQ ID NO: 2), the amino acid sequence of zebrafish grn2 (SEQ ID NO: 3. The amino acid sequence of the variant is SEQ ID NO: 5 ) And the gene sequence encoding it (SEQ ID NO: 4), the amino acid sequence of human progranulin (SEQ ID NO: 6) and the gene sequence encoding it (SEQ ID NO: 7) and the amino acid sequence of mouse progranulin (mouse grn1) (SEQ ID NO: 8) and the gene sequence encoding it (SEQ ID NO: 9) are shown.
生体から分離、精製することによってもグラニュリン分子又はその断片を調製することが可能であるが、好ましくは、品質の均一化、調製操作、スケールアップなどの観点から、組換え体(リコンビナントタンパク質)としてグラニュリン分子又はその断片を調製する。調製操作は常法で行うことができ、例えば、Molecular Cloning(Third Edition, Cold Spring Harbor Laboratory Press, New York)、Current protocols in molecular biology(edited by Frederick M. Ausubel et al., 1987)等が参考になる。
Granulin molecules or fragments thereof can also be prepared by separating and purifying from a living body, but preferably as a recombinant (recombinant protein) from the viewpoint of homogenization of quality, preparation operation, scale-up, etc. A granulin molecule or fragment thereof is prepared. Preparation operations can be performed in a conventional manner, for example, Molecular Cloning (Third Edition, Cold Spring Harbor Laboratory Press, New York), Current protocols in molecular biology (edited by Frederick M. Ausubel et al., 1987) become.
好中球の調製は常法で行えばよい。好中球の調製法の具体例として、以下にマウス活性型好中球の調製法とヒト好中球の調製法を説明する。
<マウス活性型好中球の調製>
12%カゼイン2mLをマウス腹腔内投与5時間後、4℃にしておいたHBSS(Hanks’ balanced salt solution) 5mlにてperitoneal lavageにより腹腔浸出液(peritoneal exudate cells)を回収し、50 x gにて5分間遠心する。沈澄をよくほぐしてから4.5mlの蒸留水を加えすばやく撹持し残存赤血球を溶血し、20秒以内に10倍濃度のPBS(-)を加える。HBSSで2回洗浄した後、5mM HEPES-MEM (Eagles’ minimum essential medium)にて3×106/mlに調整する(活性型好中球液)。 Neutrophils can be prepared by conventional methods. As specific examples of the method for preparing neutrophils, a method for preparing mouse active neutrophils and a method for preparing human neutrophils are described below.
<Preparation of mouse activated neutrophils>
After 5 hours of intraperitoneal administration of 2 mL of 12% casein, collect peritoneal exudate cells by peritoneal lavage with 5 ml of HBSS (Hanks' balanced salt solution) kept at 4 ° C, 5 minutes at 50 xg Centrifuge. Thoroughly dissolve the precipitate, add 4.5 ml of distilled water and stir quickly to dissolve the remaining red blood cells, and add PBS (-) 10-fold in 20 seconds. After washing twice with HBSS, adjust to 3 × 10 6 / ml with 5 mM HEPES-MEM (Eagles' minimum essential medium) (active neutrophil solution).
<マウス活性型好中球の調製>
12%カゼイン2mLをマウス腹腔内投与5時間後、4℃にしておいたHBSS(Hanks’ balanced salt solution) 5mlにてperitoneal lavageにより腹腔浸出液(peritoneal exudate cells)を回収し、50 x gにて5分間遠心する。沈澄をよくほぐしてから4.5mlの蒸留水を加えすばやく撹持し残存赤血球を溶血し、20秒以内に10倍濃度のPBS(-)を加える。HBSSで2回洗浄した後、5mM HEPES-MEM (Eagles’ minimum essential medium)にて3×106/mlに調整する(活性型好中球液)。 Neutrophils can be prepared by conventional methods. As specific examples of the method for preparing neutrophils, a method for preparing mouse active neutrophils and a method for preparing human neutrophils are described below.
<Preparation of mouse activated neutrophils>
After 5 hours of intraperitoneal administration of 2 mL of 12% casein, collect peritoneal exudate cells by peritoneal lavage with 5 ml of HBSS (Hanks' balanced salt solution) kept at 4 ° C, 5 minutes at 50 xg Centrifuge. Thoroughly dissolve the precipitate, add 4.5 ml of distilled water and stir quickly to dissolve the remaining red blood cells, and add PBS (-) 10-fold in 20 seconds. After washing twice with HBSS, adjust to 3 × 10 6 / ml with 5 mM HEPES-MEM (Eagles' minimum essential medium) (active neutrophil solution).
<ヒト全血から好中球の調製>
抗凝固剤処理したヒト全血3.5mlをモノ・ポリ分離溶液(DS Pharma Biomedical)を使用して好中球層を単離する。 <Preparation of neutrophils from human whole blood>
Isolate 3.5 ml of anticoagulant-treated human whole blood using mono / poly separation solution (DS Pharma Biomedical).
抗凝固剤処理したヒト全血3.5mlをモノ・ポリ分離溶液(DS Pharma Biomedical)を使用して好中球層を単離する。 <Preparation of neutrophils from human whole blood>
Isolate 3.5 ml of anticoagulant-treated human whole blood using mono / poly separation solution (DS Pharma Biomedical).
尚、調製した好中球を維持するためには、例えば、10% FBSを含むRPMI1640培地にて37℃、5% CO2で培養すればよい。
In order to maintain the prepared neutrophils, for example, culture may be performed at 37 ° C. and 5% CO 2 in RPMI1640 medium containing 10% FBS.
好中球の培養上清は、調製した好中球をin vitroで培養することによって得ることができる。例えば、調製したマウス又はヒト好中球を10cmシャーレ(BD)またはフラスコ(25cm2)に40,000細胞/mlになるように播種し、FBSを含まないHEPES-MEM又はRPMI1640で12~24時間培養する。その後上清を回収し、300 x gで5分間遠心分離した後、0.22μm滅菌フィルター(Millipore)を用いて濾過滅菌を行う。限外ろ過(例えばアミコンウルトラ-15(Millipore)(分画分子量3,000)を使用する)により培養上清を濃縮する。
The culture supernatant of neutrophils can be obtained by culturing the prepared neutrophils in vitro. For example, prepared mouse or human neutrophils are seeded in a 10 cm petri dish (BD) or flask (25 cm 2 ) at 40,000 cells / ml, and cultured in HEPES-MEM or RPMI1640 without FBS for 12 to 24 hours. . Thereafter, the supernatant is collected, centrifuged at 300 × g for 5 minutes, and then sterilized by filtration using a 0.22 μm sterilizing filter (Millipore). The culture supernatant is concentrated by ultrafiltration (eg using Amicon Ultra-15 (Millipore) (fractionated molecular weight 3,000)).
本発明の網膜再生促進薬は、常法に従い、硝子体内投与用注射剤、点眼剤又は眼軟膏などとして調製することができる。硝子体内投与用注射剤は、例えば、上記有効成分を適当な溶媒(例えば注射用蒸留水、生理食塩水)に懸濁又は溶解させることによって製造することができる。適宜、マンニトール、塩化ナトリウム、グルコース、ソルビット、グリセロール、キシリトール、フルクトース、マルトース、マンノース等の等張化剤、アルブミン等の安定化剤、ベンジルアルコール、パラヒドロキシ安息香酸メチル等の保存剤等を製剤中に添加することができる。また、クエン酸等の酸、ジイソプロパノールアミン等の塩基をpH調整剤として製剤中に添加することもできる。
The retinal regeneration-promoting agent of the present invention can be prepared as an injection for intravitreal administration, an eye drop or an eye ointment according to a conventional method. An injection for intravitreal administration can be produced, for example, by suspending or dissolving the active ingredient in an appropriate solvent (for example, distilled water for injection or physiological saline). Appropriate preparation of isotonic agents such as mannitol, sodium chloride, glucose, sorbit, glycerol, xylitol, fructose, maltose, mannose, stabilizers such as albumin, preservatives such as benzyl alcohol and methyl parahydroxybenzoate Can be added. Further, an acid such as citric acid and a base such as diisopropanolamine can be added to the preparation as a pH adjuster.
点眼剤は、例えば、上記有効成分を適当な溶媒(例えば注射用蒸留水、生理食塩水)に溶解させることによって製造することができる。適宜、マンニトール、塩化ナトリウム、グルコース、ソルビット、グリセロール、キシリトール、フルクトース、マルトース、マンノース、グリセリン等の等張化剤、エデト酸ナトリウム、アルブミン等の安定化剤、ベンジルアルコール、パラヒドロキシ安息香酸メチル等の保存剤、ポリオキシエチレンモノオレート、ステアリン酸ポリオキシル40等の界面活性化剤等を製剤中に添加することができる。また、クエン酸等の酸、ジイソプロパノールアミン等の塩基をpH調整剤として製剤中に添加することもできる。点眼剤のpHは眼科製剤に許容される範囲内であれば特に限定されないが、5.0~8.5が好ましい。
An eye drop can be produced, for example, by dissolving the above active ingredient in an appropriate solvent (for example, distilled water for injection or physiological saline). As appropriate, isotonic agents such as mannitol, sodium chloride, glucose, sorbit, glycerol, xylitol, fructose, maltose, mannose, glycerin, stabilizers such as sodium edetate, albumin, benzyl alcohol, methyl parahydroxybenzoate, etc. A preservative, a surfactant such as polyoxyethylene monooleate, polyoxyl 40 stearate and the like can be added to the preparation. Further, an acid such as citric acid and a base such as diisopropanolamine can be added to the preparation as a pH adjuster. The pH of the eye drop is not particularly limited as long as it is within the range acceptable for ophthalmic preparations, but is preferably 5.0 to 8.5.
本発明の網膜再生促進薬は、網膜の障害に起因する又は網膜の障害を伴う(換言すれば、網膜の再生が治療効果をもたらすことになる)各種眼科疾患の治療に用いられる。本発明の網膜再生促進薬の治療対象となる眼科疾患としては、緑内障、加齢黄斑変性症、網膜色素変性症、糖尿病網膜症、外傷による視力低下又は失明、Leber先天盲、白点状眼底、錐体ジストロフィ(錐体杆体ジストロフィ)、Stargardt病、黄斑ジストロフィ、家族性ドルーゼン、中心性輪紋状脈絡膜ジストロフィ及び脊髄小脳変性症7型が挙げられる。中でも、視細胞に変性が起きる疾患に特に有効である。
The retinal regeneration-promoting agent of the present invention is used for the treatment of various ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders (in other words, retinal regeneration has a therapeutic effect). As an ophthalmic disease to be treated with the retinal regeneration-promoting agent of the present invention, glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, Cone dystrophy (cone rod dystrophy), Stargardt disease, macular dystrophy, familial drusen, central crested choroidal dystrophy and spinocerebellar degeneration 7 types. Among them, it is particularly effective for diseases in which degeneration occurs in photoreceptor cells.
本発明の網膜再生促進薬の投与量(使用量)は、患者の病態、年齢、体重、剤形等を考慮して適宜設定すればよい。硝子体内投与用注射剤として構成した場合には、例えば、有効成分を0.01~70重量%含有する当該製剤を1月に1回、適量を投与すればよい。点眼剤として構成した場合には、例えば、有効成分を0.01~70重量%含有する当該製剤を1日1~数回、1滴~数滴点眼すればよい。
The dose (use amount) of the retinal regeneration-promoting agent of the present invention may be appropriately set in consideration of the patient's condition, age, weight, dosage form, and the like. When configured as an injection for intravitreal administration, for example, an appropriate amount of the preparation containing 0.01 to 70% by weight of the active ingredient may be administered once a month. When configured as an eye drop, for example, the preparation containing 0.01 to 70% by weight of the active ingredient may be instilled 1 to several times a day once to several drops.
本発明の網膜再生促進薬の治療対象は特に限定されず、ヒト及びヒト以外の哺乳動物(ペット動物、家畜、実験動物など。具体的には例えばモルモット、ハムスター、サル、ウシ、ブタ、ヤギ、ヒツジ、イヌ、ネコ、ニワトリ、ウズラ等である)を含む。好ましくは、本発明の網膜再生促進薬はヒトに対して適用される。
The treatment target of the retinal regeneration-promoting agent of the present invention is not particularly limited, and humans and non-human mammals (pet animals, domestic animals, laboratory animals, etc., specifically, for example, guinea pigs, hamsters, monkeys, cows, pigs, goats, Sheep, dogs, cats, chickens, quails, etc.). Preferably, the retinal regeneration-promoting agent of the present invention is applied to humans.
以上の記述から自明な通り本出願は、緑内障、加齢黄斑変性症、網膜色素変性症、糖尿病網膜症、外傷による視力低下又は失明、Leber先天盲、白点状眼底、錐体ジストロフィ(錐体杆体ジストロフィ)、Stargardt病、黄斑ジストロフィ、家族性ドルーゼン、中心性輪紋状脈絡膜ジストロフィ及び脊髄小脳変性症7型等の患者に対して本発明の網膜再生促進薬を治療上有効量投与することを特徴とする治療法も提供する。
As is obvious from the above description, the present application includes glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, decreased visual acuity or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (cone) (Body dystrophy), Stargardt disease, macular dystrophy, familial drusen, central annulus choroidal dystrophy, spinocerebellar degeneration 7 and the like, a therapeutically effective amount of the retinal regeneration promoter of the present invention is administered. A featured treatment is also provided.
視細胞分化促進因子としてプログラニュリンに着目し、ゼブラフィッシュ網膜障害モデルを利用して以下の検討を行った。
1.方法
ゼブラフィッシュ(AB line)の眼球に30Gの針を刺し、網膜に4か所から8か所の障害を与えた。その後、6時間から7日後の間で眼球を摘出し、凍結切片を作製した。また、網膜を単離後にRNAを調製した。凍結切片はin situハイブリダイゼーション法によりgrn1 mRNAの発現部位を特定するために用いた。また、同時に抗4C4抗体(ミクログリアのマーカー)及び抗プラスチン(plastin)抗体(ミクログリア及び好中球のマーカー)を用いて免疫染色した。RNAは、PCR法によるgrn1、grn2、grnA、grnB及びascl1a mRNA発現量の測定に使用した。一方、ゼブラフィッシュ遺伝子の発現を特異的にノックダウンできるモルフォリノオリゴを作製してエレクトロポレーション法により網膜内に導入した。導入後、ゼブラフィッシュの腹腔内にBrdUを投与した。この処理の後、凍結切片を作製し、また、RNAを調製した。凍結切片を用い、BrdU陽性細胞を再生過程の細胞として定量した(抗BrdU抗体を使用)。また、RNAは、ゼブラフィッシュ網膜再生の指標となる遺伝子ascl11の発現量の定量に用いた。 Focusing on progranulin as a photoreceptor differentiation promoting factor, the following examination was performed using a zebrafish retinopathy model.
1. Method A 30G needle was inserted into the eyeball of a zebrafish (AB line), and the retina was damaged in 4 to 8 places. Thereafter, the eyeballs were removed between 6 hours and 7 days, and frozen sections were prepared. In addition, RNA was prepared after the retina was isolated. The frozen section was used to identify the expression site of grn1 mRNA by in situ hybridization. Simultaneously, immunostaining was performed using an anti-4C4 antibody (microglia marker) and an antiplastin antibody (microglia and neutrophil marker). RNA was used for measuring the expression levels of grn1, grn2, grnA, grnB and ascl1a mRNA by PCR. On the other hand, a morpholino oligo capable of specifically knocking down the expression of the zebrafish gene was prepared and introduced into the retina by electroporation. After introduction, BrdU was administered intraperitoneally to zebrafish. After this treatment, frozen sections were prepared and RNA was prepared. Using frozen sections, BrdU positive cells were quantified as cells in the regeneration process (using anti-BrdU antibody). RNA was used for quantification of the expression level of the gene ascl11, which is an indicator of zebrafish retinal regeneration.
1.方法
ゼブラフィッシュ(AB line)の眼球に30Gの針を刺し、網膜に4か所から8か所の障害を与えた。その後、6時間から7日後の間で眼球を摘出し、凍結切片を作製した。また、網膜を単離後にRNAを調製した。凍結切片はin situハイブリダイゼーション法によりgrn1 mRNAの発現部位を特定するために用いた。また、同時に抗4C4抗体(ミクログリアのマーカー)及び抗プラスチン(plastin)抗体(ミクログリア及び好中球のマーカー)を用いて免疫染色した。RNAは、PCR法によるgrn1、grn2、grnA、grnB及びascl1a mRNA発現量の測定に使用した。一方、ゼブラフィッシュ遺伝子の発現を特異的にノックダウンできるモルフォリノオリゴを作製してエレクトロポレーション法により網膜内に導入した。導入後、ゼブラフィッシュの腹腔内にBrdUを投与した。この処理の後、凍結切片を作製し、また、RNAを調製した。凍結切片を用い、BrdU陽性細胞を再生過程の細胞として定量した(抗BrdU抗体を使用)。また、RNAは、ゼブラフィッシュ網膜再生の指標となる遺伝子ascl11の発現量の定量に用いた。 Focusing on progranulin as a photoreceptor differentiation promoting factor, the following examination was performed using a zebrafish retinopathy model.
1. Method A 30G needle was inserted into the eyeball of a zebrafish (AB line), and the retina was damaged in 4 to 8 places. Thereafter, the eyeballs were removed between 6 hours and 7 days, and frozen sections were prepared. In addition, RNA was prepared after the retina was isolated. The frozen section was used to identify the expression site of grn1 mRNA by in situ hybridization. Simultaneously, immunostaining was performed using an anti-4C4 antibody (microglia marker) and an antiplastin antibody (microglia and neutrophil marker). RNA was used for measuring the expression levels of grn1, grn2, grnA, grnB and ascl1a mRNA by PCR. On the other hand, a morpholino oligo capable of specifically knocking down the expression of the zebrafish gene was prepared and introduced into the retina by electroporation. After introduction, BrdU was administered intraperitoneally to zebrafish. After this treatment, frozen sections were prepared and RNA was prepared. Using frozen sections, BrdU positive cells were quantified as cells in the regeneration process (using anti-BrdU antibody). RNA was used for quantification of the expression level of the gene ascl11, which is an indicator of zebrafish retinal regeneration.
2.結果
まず、ゼブラフィッシュの網膜障害時にグラニュリンファミリーのどの分子種が増加するかをRT-PCR法にて検討した。結果を図2に示す。grn1の発現が障害初期から顕著に増加していた。grn2も同様に増加していたが、grn1と比較して発現量は少なかった。一方、grnAは障害2日後に発現が増加したが、発現量は少ない。grnBは発現の低下が認められた。
より詳細に検討するために定量的PCR(qPCR)を行ったところ、grn1の発現量は未処置群と比較して障害15時間後に300倍の増加を認めた(図3)。一方、ゼブラフィッシュの網膜障害時に発現の増加が認められ、網膜の再生に必要不可欠な因子であるascl1aも同様に発現量が増加したが、発現量のピークはgrn1より遅く24時間から4日後の間であった(図3)。 2. Results First, the RT-PCR method was used to determine which molecular species of the granulin family increased during retinal damage in zebrafish. The results are shown in FIG. The expression of grn1 was significantly increased from the early stage of injury. grn2 was also increased, but the expression level was lower than that of grn1. On the other hand, the expression of grnA increased 2 days after the injury, but the expression level was small. The expression of grnB was decreased.
When quantitative PCR (qPCR) was performed to examine in more detail, the expression level of grn1 was increased by a factor of 300 after 15 hours of injury compared to the untreated group (FIG. 3). On the other hand, an increase in expression was observed at the time of retinal damage in zebrafish, and the expression level of ascl1a, an essential factor for retinal regeneration, also increased, but the peak expression level was later than grn1 24 hours to 4 days later. (Fig. 3).
まず、ゼブラフィッシュの網膜障害時にグラニュリンファミリーのどの分子種が増加するかをRT-PCR法にて検討した。結果を図2に示す。grn1の発現が障害初期から顕著に増加していた。grn2も同様に増加していたが、grn1と比較して発現量は少なかった。一方、grnAは障害2日後に発現が増加したが、発現量は少ない。grnBは発現の低下が認められた。
より詳細に検討するために定量的PCR(qPCR)を行ったところ、grn1の発現量は未処置群と比較して障害15時間後に300倍の増加を認めた(図3)。一方、ゼブラフィッシュの網膜障害時に発現の増加が認められ、網膜の再生に必要不可欠な因子であるascl1aも同様に発現量が増加したが、発現量のピークはgrn1より遅く24時間から4日後の間であった(図3)。 2. Results First, the RT-PCR method was used to determine which molecular species of the granulin family increased during retinal damage in zebrafish. The results are shown in FIG. The expression of grn1 was significantly increased from the early stage of injury. grn2 was also increased, but the expression level was lower than that of grn1. On the other hand, the expression of grnA increased 2 days after the injury, but the expression level was small. The expression of grnB was decreased.
When quantitative PCR (qPCR) was performed to examine in more detail, the expression level of grn1 was increased by a factor of 300 after 15 hours of injury compared to the untreated group (FIG. 3). On the other hand, an increase in expression was observed at the time of retinal damage in zebrafish, and the expression level of ascl1a, an essential factor for retinal regeneration, also increased, but the peak expression level was later than grn1 24 hours to 4 days later. (Fig. 3).
ゼブラフィッシュ網膜障害モデルにおいてgrn1の発現の増加が認められたことから、in situハイブリダイゼーション法により発現部位を確認することにした。grn1アンチセンスプローブを用いて検討したところ、障害15時間後から障害部位周辺に発現の強い陽性細胞が認められ、24時間後にも確認された(図4)。障害3日目ではミュラーグリアにgrn1陽性が認められた(図5)。ミクログリアのマーカーである抗4C4抗体を用いて免疫染色をしたところ、grn1陽性細胞との共局在はほとんど認められなかった(図4)。障害部位を抗4C4抗体及び抗プラスチン抗体(ミクログリア及び好中球のマーカー)を用いて免疫染色したところ、4C4陰性/プラスチン陽性細胞が認められた(図4)。また、これらの細胞の形状はgrn1陽性細胞に酷似していた。以上の結果から、ゼブラフィッシュの網膜障害部位(特に好中球)においてgrn1が高発現していることが判明した。
Since the expression of grn1 was increased in the zebrafish retinal disorder model, it was decided to confirm the expression site by in situ hybridization. When examined using the grn1 antisense probe, positive cells with strong expression were observed around the damaged site 15 hours after the injury, and were confirmed even 24 hours later (FIG. 4). On the third day of injury, grn1 positivity was observed in Muller glia (FIG. 5). When immunostaining was performed using an anti-4C4 antibody that is a marker of microglia, colocalization with grn1-positive cells was hardly observed (FIG. 4). When the damaged site was immunostained with anti-4C4 antibody and antiplastin antibody (microglia and neutrophil markers), 4C4 negative / plastin positive cells were observed (FIG. 4). The shape of these cells resembled grn1-positive cells. From the above results, it was found that grn1 is highly expressed in retinal damage sites (especially neutrophils) in zebrafish.
ゼブラフィッシュ網膜障害モデルの再生過程にgrn1がどの様に関与するかを調べるため、grn1のアンチセンスモルフォリノオリゴ(MO)による発現量のノックダウンを用いて検討した。Grn1 MOを障害直後(0h)、15時間後、48時間後にエレクトロポレーション法で網膜細胞及び好中球などの障害後に遊走してくる細胞に導入した。障害直後および48時間後では遊走細胞は認められないため、15時間後のみ遊走細胞にもMOを導入できる。はじめに、網膜の再生に必要不可欠であるascl1aの発現に変化があるか否かをRT-PCR法で検討したところ、障害15時間後にgrn1 MOを導入した網膜でascl1aの減少が認められた(図6)。一方、障害直後及び48時間後にgrn1 MOを導入した網膜ではascl1aの発現量に影響はなかった(図6)。このことから、好中球のgrn1がascl1aの発現量を正に調節していることが明らかとなった。そこで、網膜の再生に影響するか否かBrdU陽性細胞を指標として検討したところ、障害15時間後にgrn1 MOを導入した網膜では対照用のMOを導入した網膜と比較して、BrdU陽性細胞が減少していた(図7)。以上の結果より、好中球で発現するgrn1は網膜障害時にascl1aの発現を増加させ再生を促していることが示唆された。grn1はミュラーグリアをはじめとする好中球以外の網膜細胞にも発現が認められるにもかかわらず、それらをgrn1 MOでノックダウンしてもascl1aの発現量に変化が認められなかったことから、好中球特異的な酵素などにより切断されたgrn1がascla1の発現量を調節している可能性が示唆された。
In order to examine how grn1 is involved in the regeneration process of the zebrafish retinal disorder model, we examined the expression level of grn1 using antisense morpholino-oligo (MO). Grn1 MO was introduced into cells that migrated after injury, such as retinal cells and neutrophils, by electroporation immediately after injury (0h), 15 hours, and 48 hours later. Since migratory cells are not observed immediately after injury and 48 hours later, MO can be introduced into migrating cells only after 15 hours. First, RT-PCR was used to examine whether there was a change in the expression of ascl1a, which is essential for retinal regeneration. As a result, a decrease in ascl1a was observed in the retina into which grn1 MO was introduced 15 hours after injury (Fig. 6). On the other hand, the expression level of ascl1a was not affected in the retina into which grn1 MO was introduced immediately after the injury and 48 hours later (FIG. 6). This revealed that neutrophil grn1 positively regulates the expression level of ascl1a. Therefore, when BrdU positive cells were used as an index to determine whether or not they affected retina regeneration, BrdU positive cells decreased in retinas introduced with grn1 MO 15 hours after injury compared to retinas introduced with control MOs. (Figure 7). These results suggest that grn1 expressed in neutrophils increases ascl1a expression and promotes regeneration during retinal damage. Although grn1 is also expressed in retinal cells other than neutrophils including Muller glia, even if they are knocked down with grn1 MO, there was no change in the expression level of ascl1a. These results suggest that grn1 cleaved by neutrophil-specific enzymes may regulate the expression level of ascla1.
grn1の作用機序を更に検討するため、以下の実験を行った。まず、ゼブラフィッシュgrn1のmRNAをクローニングし、3'側に6xHisタグ配列を付加した後、in vitroで発現させ、grn1蛋白質を合成した。Hisタグ用のカラム及びバッファー置換用のカラムを用いて精製し、組換え体grn1を得た。200ngの組換え体grn1又は溶媒をゼブラフィッシュ網膜障害モデルの硝子体に投与した。2日後に眼球を摘出して網膜のmRNAを調製し、網膜再生関連因子の増減をRT-PCR法にて確認した。また、4日後にBrdUを腹腔内投与し、眼球を摘出して凍結切片を作製した。抗BrdU抗体で免疫蛍光染色して、BrdU陽性細胞の有無を蛍光顕微鏡で観察した。
The following experiment was conducted to further investigate the mechanism of action of grn1. First, zebrafish grn1 mRNA was cloned, a 6xHis tag sequence was added to the 3 'side, and then expressed in vitro to synthesize the grn1 protein. Purification was performed using a column for His tag and a column for buffer replacement to obtain recombinant grn1. 200 ng recombinant grn1 or vehicle was administered to the vitreous of the zebrafish retinal disorder model. Two days later, the eyeballs were removed, retinal mRNA was prepared, and the increase or decrease in retinal regeneration-related factors was confirmed by RT-PCR. Four days later, BrdU was intraperitoneally administered, and the eyeball was removed to prepare a frozen section. Immunofluorescence staining was performed with an anti-BrdU antibody, and the presence or absence of BrdU positive cells was observed with a fluorescence microscope.
RT-PCRの結果、網膜再生時に増加する因子(ascl1a、c-mycb、lin28、socs3)の発現増加が観察された(図8右)。また、免疫蛍光染色の結果、grn1-his投与群ではBrdU陽性細胞が確認された(図8左)。これらの結果から、grn1はゼブラフィッシュのミュラーグリア細胞の脱分化・増殖を促進していることが明らかとなった。また、grn1がascl1aの上流の因子である可能性が示唆された。以上の通り、grn1の直接的な作用が確認され、grn1はミュラーグリア細胞の脱分化・増殖を単独で促進できる因子であることが示された。
As a result of RT-PCR, increased expression of factors (ascl1a, c-mycb, lin28, socs3) increased during retina regeneration was observed (right side of FIG. 8). Further, as a result of immunofluorescence staining, BrdU positive cells were confirmed in the grn1-his administration group (FIG. 8 left). From these results, it was revealed that grn1 promotes dedifferentiation and proliferation of zebrafish Muller glia cells. It was also suggested that grn1 may be an upstream factor of ascl1a. As described above, the direct action of grn1 was confirmed, and it was shown that grn1 is a factor that can independently promote the dedifferentiation and proliferation of Muller glial cells.
本発明の網膜再生促進薬は、網膜の障害に起因する又は網膜の障害を伴う各種眼科疾患の治療に適用可能である。従来の対症療法に代わる治療手段として、人工多能性幹細胞(iPS細胞)等を利用した再生医療が注目されている。本発明は再生医療の技術的選択肢を増やすものであり、従来の治療法を代替又は補足するものとしてその利用・応用が図られる。
The retinal regeneration-promoting agent of the present invention can be applied to the treatment of various ophthalmic diseases caused by retinal disorders or accompanied by retinal disorders. Regenerative medicine using artificial pluripotent stem cells (iPS cells) or the like has attracted attention as a therapeutic means to replace conventional symptomatic treatment. The present invention increases technical options for regenerative medicine, and can be used and applied as an alternative or supplement to conventional therapies.
この発明は、上記発明の実施の形態及び実施例の説明に何ら限定されるものではない。特許請求の範囲の記載を逸脱せず、当業者が容易に想到できる範囲で種々の変形態様もこの発明に含まれる。本明細書の中で明示した論文、公開特許公報、及び特許公報などの内容は、その全ての内容を援用によって引用することとする。
The present invention is not limited to the description of the embodiments and examples of the above invention. Various modifications may be included in the present invention as long as those skilled in the art can easily conceive without departing from the description of the scope of claims. The contents of papers, published patent gazettes, patent gazettes, and the like specified in this specification are incorporated by reference in their entirety.
Claims (5)
- 以下の(1)及び(2)からなる群より選択される成分を含む、網膜再生促進薬:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。 Retinal regeneration promoting agent comprising a component selected from the group consisting of the following (1) and (2):
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof. - 前記グラニュリン分子がゼブラフィッシュgrn1、ゼブラフィッシュgrn2、ヒトプログラニュリン又はマウスプログラニュリンであり、前記断片がグラニュリンA、グラニュリンB、グラニュリンC、グラニュリンD、グラニュリンE、グラニュリンF、グラニュリンG又はグラニュリンPである、請求項1に記載の網膜再生促進薬。 The granulin molecule is zebrafish grn1, zebrafish grn2, human progranulin or mouse progranulin, and the fragment is granulin A, granulin B, granulin C, granulin D, granulin E, granulin F, granulin G or granulin P. The retinal regeneration-promoting agent according to claim 1, wherein
- 前記グラニュリン分子がリコンビナントタンパク質である、請求項1又は2に記載の網膜再生促進薬。 The retinal regeneration-promoting agent according to claim 1 or 2, wherein the granulin molecule is a recombinant protein.
- 緑内障、加齢黄斑変性症、網膜色素変性症、糖尿病網膜症、外傷による視力低下又は失明、Leber先天盲、白点状眼底、錐体ジストロフィ(錐体杆体ジストロフィ)、Stargardt病、黄斑ジストロフィ、家族性ドルーゼン、中心性輪紋状脈絡膜ジストロフィ及び脊髄小脳変性症7型からなる群より選択される疾患の治療用である、請求項1~3のいずれか一項に記載の網膜再生促進薬。 Glaucoma, age-related macular degeneration, retinitis pigmentosa, diabetic retinopathy, visual loss or blindness due to trauma, Leber congenital blindness, white spotted fundus, cone dystrophy (Star cone dystrophy), Stargardt disease, macular dystrophy, family The retinal regeneration-promoting agent according to any one of claims 1 to 3, which is used for treatment of a disease selected from the group consisting of sex drusen, central ring-shaped choroidal dystrophy, and spinocerebellar degeneration type 7.
- 網膜の障害を伴う眼科疾患の患者に対して、以下の(1)及び(2)からなる群より選択される成分を治療上有効量投与するステップを含む、治療法:
(1)グラニュリン分子、又はグラニュリンモチーフを含むその断片;
(2)好中球又はその培養上清。 A treatment method comprising the step of administering a therapeutically effective amount of a component selected from the group consisting of the following (1) and (2) to a patient with an ophthalmic disease associated with a retinal disorder:
(1) a granulin molecule, or a fragment thereof containing a granulin motif;
(2) Neutrophils or culture supernatants thereof.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015-018857 | 2015-02-02 | ||
| JP2015018857 | 2015-02-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016125330A1 true WO2016125330A1 (en) | 2016-08-11 |
Family
ID=56563689
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2015/074162 WO2016125330A1 (en) | 2015-02-02 | 2015-08-27 | Retinal regeneration promoting drug |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2016125330A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111032075A (en) * | 2017-06-15 | 2020-04-17 | 芝加哥大学 | Methods and compositions for treating cancer |
| EP4048799A4 (en) * | 2019-10-22 | 2023-11-15 | Applied Genetic Technologies Corporation | Adeno-associated virus (aav) systems for treatment of progranulin associated neurodegenerative diseases or disorders |
-
2015
- 2015-08-27 WO PCT/JP2015/074162 patent/WO2016125330A1/en active Application Filing
Non-Patent Citations (2)
| Title |
|---|
| KURIMOTO ET AL.: "Neutrophils Express Oncomodulin and Promote Optic Nerve Regeneration", THE JOURNAL OF NEUROSCIENCE, vol. 33, no. 37, 11 September 2013 (2013-09-11), pages 14816 - 14824 * |
| TSURUMA ET AL.: "Progranulin, a Major Secreted Protein of Mouse Adipose-Derived Stem Cells, Inhibits Light-Induced Retinal Degeneration", STEM CELLS TRANS MED, vol. 3, no. 1, 2014, pages 42 - 53 * |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111032075A (en) * | 2017-06-15 | 2020-04-17 | 芝加哥大学 | Methods and compositions for treating cancer |
| JP2020524141A (en) * | 2017-06-15 | 2020-08-13 | ザ・ユニバーシティ・オブ・シカゴThe University Of Chicago | Methods and compositions for treating cancer |
| JP7189160B2 (en) | 2017-06-15 | 2022-12-13 | ザ・ユニバーシティ・オブ・シカゴ | Methods and compositions for treating cancer |
| EP4048799A4 (en) * | 2019-10-22 | 2023-11-15 | Applied Genetic Technologies Corporation | Adeno-associated virus (aav) systems for treatment of progranulin associated neurodegenerative diseases or disorders |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Zhang et al. | RGMa mediates reactive astrogliosis and glial scar formation through TGFβ1/Smad2/3 signaling after stroke | |
| Tönges et al. | Hepatocyte growth factor protects retinal ganglion cells by increasing neuronal survival and axonal regeneration in vitro and in vivo | |
| Alizadeh et al. | Neuregulin‐1 positively modulates glial response and improves neurological recovery following traumatic spinal cord injury | |
| Khalin et al. | Targeted delivery of brain-derived neurotrophic factor for the treatment of blindness and deafness | |
| Wu et al. | Astrocytic YAP protects the optic nerve and retina in an experimental autoimmune encephalomyelitis model through TGF-β signaling | |
| Machalińska et al. | Long-term neuroprotective effects of NT-4–engineered mesenchymal stem cells injected intravitreally in a mouse model of acute retinal injury | |
| JP2004532809A (en) | Method for stimulating nervous system regeneration and repair by inhibiting phosphodiesterase type 4 | |
| KR20180096733A (en) | A long-acting GLF-1R agonist as a treatment for the nervous system and neurodegenerative conditions | |
| Sivan et al. | Stem cell therapy for treatment of ocular disorders | |
| CN111148755A (en) | Recombinant modified fibroblast growth factor and therapeutic uses thereof | |
| JP7452798B2 (en) | Disease therapeutics based on mesenchymal stem cell mobilization | |
| JP2011513290A (en) | New use of VEGFxxxb | |
| US11622964B2 (en) | Method for destroying cellular mechanical homeostasis and promoting regeneration and repair of tissues and organs, and use thereof | |
| WO2016125330A1 (en) | Retinal regeneration promoting drug | |
| JPWO2006077824A1 (en) | Drugs for nerve cell regeneration | |
| Heo et al. | Intraperitoneal administration of adipose tissue‐derived stem cells for the rescue of retinal degeneration in a mouse model via indigenous CNTF up‐regulation by IL‐6 | |
| ES2552587B1 (en) | PEPTIDE DERIVED FROM SOCS1 FOR USE IN CHRONIC COMPLICATIONS OF THE DIABETES | |
| Chen et al. | Neural stem cells in the treatment of Alzheimer’s disease: Current status, challenges, and future prospects | |
| JP6440107B2 (en) | Cell sheet production method, composition, cell culture aid, and cell culture method | |
| CN109937053B (en) | Pharmaceutical composition for the treatment of macular degeneration comprising an mTOR inhibitor | |
| JP2021522277A (en) | A pharmaceutical composition for the prevention or treatment of degenerative brain diseases containing ibrutinib as an active ingredient. | |
| WO2022131322A1 (en) | Neuron activator | |
| Quan et al. | Hydralazine plays an immunomodulation role of pro-regeneration in a mouse model of spinal cord injury | |
| Trofimova | Molecular Mechanisms of Retina Pathology and Ways of Its Correction | |
| JP2017514896A (en) | Use of peptides to treat angiogenesis-related diseases |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15881147 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15881147 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |