WO2015110930A1 - Modified interleukin 21 receptor proteins - Google Patents
Modified interleukin 21 receptor proteins Download PDFInfo
- Publication number
- WO2015110930A1 WO2015110930A1 PCT/IB2015/050169 IB2015050169W WO2015110930A1 WO 2015110930 A1 WO2015110930 A1 WO 2015110930A1 IB 2015050169 W IB2015050169 W IB 2015050169W WO 2015110930 A1 WO2015110930 A1 WO 2015110930A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino acid
- seq
- modified
- ecd
- acid sequence
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/715—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons
- C07K14/7155—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons for interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Definitions
- the present invention relates to novel modified IL21 R ECD proteins and fusion proteins thereof having, among other properties, improved stability, activity, and increased expression.
- the invention further relates to use of these proteins to treat diseases or disorders mediated by interaction of IL21 with IL21 R.
- Human interleukin 21 is a cytokine that shares sequence homology with IL2, IL4 and IL15 (Parrish-Novak et al. (2000) Nature 408:57-63).
- Human IL21 receptor (IL21 R) is a class I cytokine receptor that is expressed in lymphoid tissues, particularly on T cells, B cells, natural killer (NK) cells, dendritic cells (DC) and macrophages (Parrish-Novak et al. (2000) supra), which allows these cells to respond to IL21 (Leonard and Spolski (2005) Nat. Rev. Immunol. 5:688-98).
- the widespread lymphoid distribution of IL21 R indicates that IL21 plays an important role in immune regulation.
- IL21 significantly modulates the function of B cells, CD4 + and CD8 + T cells, and NK cells (Parrish-Novak et al. (2000) supra; Kasaian et al. (2002) Immunity 16:559-69). Recent evidence suggests that IL21 - mediated signaling can have antitumor activity (Sivakumar et al. (2004) Immunology 1 12: 177- 82), and that IL21 can prevent antigen-induced asthma in mice (Shang et al. (2006) Cell.
- IL21 and IL21 R The amino acid sequences of IL21 and IL21 R, and the nucleotide sequences encoding them, are described in WO00/53761 , WO01/85792, and WO2003/028630. IL21 and IL21 R antagonists, including antibodies and fusion proteins, are also described in these applications. Despite these previous advances, there remains a long-felt need for improved IL21 R proteins, including IL21 R fusion proteins with improved therapeutic characteristics. The present invention meets this need.
- This application discloses modified IL21 R extracellular domain (ECD) proteins or polypeptides, as well as associated reagents, compositions and methods.
- ECD extracellular domain
- Embodiment 1 (E1).
- a modified IL21 R ECD comprising a modification selected from the group consisting of: one or more amino acid substitutions at or within four amino acids residues of position 148 of the amino acid sequence set forth as SEQ ID NO:1 ; one or more amino acid insertions at amino acid position 148 of the amino acid sequence set forth as SEQ ID NO:1 ; and deletion of the tryptophan residue at position 148 (W148) of the amino acid sequence set forth as SEQ ID NO:1.
- E1 Described below are a number of embodiments (E) of this first aspect of the invention where, for convenience, E1 is identical thereto.
- the modified IL21 R ECD of E2 wherein the substituted or inserted amino acid is selected from the group consisting of glycine, alanine, leucine, methionine, phenylalanine, lysine, glutamine, glutamic acid, serine, proline, valine, isoleucine, cysteine, tyrosine, histidine, arginine, asparagine, aspartic acid, and threonine.
- the modified IL21 R ECD of E1 further comprising at least one amino acid substitution at amino acid position 147, 149, and 150 of the amino acid sequence set forth as SEQ ID NO: 1.
- the modified IL21 R ECD of E1 comprising serine at position 148 (SEQ ID NO:6).
- the modified IL21 R ECD of claim 5 comprising glycine at position 147, serine at position
- the modified IL21 R ECD of E9 comprising alanine at position 122 (SEQ ID NO:8).
- E1 1.
- the modified IL21 R ECD of E9 comprising isoleucine at position 122 (SEQ ID NO:9).
- the modified IL21 R ECD of E9 comprising tryptophan at position 122 (SEQ ID NO: 10).
- E13. The modified IL21 R ECD of E9, comprising phenylalanine at position 122 (SEQ ID NO:
- E14 The modified IL21 R ECD of E9, comprising tyrosine at position 122 (SEQ ID NO:12).
- E15 A fusion protein comprising a modified IL21 R ECD according to any one of E1-E14, fused to a heterologous amino acid sequence.
- E16 The fusion protein of E15, wherein the IL21 R ECD is fused to the heterologous amino acid sequence via a linker.
- E17 The fusion protein of E16, wherein the linker is a peptidyl linker comprising a sequence selected from: GSGEGEGSEGSG (SEQ ID NO:13); GGSEGEGSEGGS (SEQ ID NO:13);
- E20 The fusion protein of E15, wherein the heterologous amino acid sequence comprises a human lgG1 Fc domain.
- E21 The fusion protein of E20, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:16.
- E22 The fusion protein of E20, wherein the Fc domain is modified to alter effector function of the Fc domain.
- E23 The fusion protein of E22, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:19.
- E24. The fusion protein of E20, wherein the Fc domain is modified to enhance the half-life of the fusion protein.
- E25 The fusion protein of E24, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO: 17.
- E26 The fusion protein of E24, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:18.
- E27 The fusion protein of E15 comprising an amino acid sequence selected from the group consisting of the sequence set forth as SEQ ID NO:20, SEQ ID NO:21 , SEQ ID NO:22, SEQ ID NO:23, SEQ ID NO:24, SEQ ID NO:25, SEQ ID NO:26, SEQ ID NO:27, SEQ ID NO:28 and SEQ ID NO:29.
- E28 A fusion protein comprising the amino acid sequence of SEQ ID NO:20.
- a pharmaceutical composition comprising the modified IL21 R ECD of any one of El- El 4, or the fusion protein of any one of E15-E28, and a pharmaceutically acceptable agent.
- E30 The pharmaceutical composition according to E29, wherein the composition comprises 0.5 - 200 mg/mL of the modified IL21 R ECD of any one of E1-E17 or the fusion protein of any one of E18-E31 , 10-50 mM tromethamine, 0.01 - 0.10 mg/mL EDTA, 20 - 120 mg/mL sucrose, and 0.01 - 0.4 mg/mL polysorbate 80.
- composition according to E30 wherein the composition comprises 100 mg/ml of the fusion protein comprising the amino acid sequence set forth as SEQ ID NO:20, and further comprises 40mM tromethamine Tris, 0.05 mg/mL EDTA, 100mg/ml sucrose, and 0.2 mg/ml polysorbate 80.
- E32 An isolated nucleic acid encoding the modified IL21 R ECD of any one of E1-E14, or the fusion protein of any one of E15-E28.
- E33 An isolated nucleic acid comprising the nucleic acid sequence of SEQ ID NO:31.
- E34 A vector comprising the nucleic acid of E33.
- E35 A host cell comprising the nucleic acid of claim 33.
- E36 A host cell comprising the vector of claim 34.
- E38 The method of E36, further comprising isolating the protein.
- E40. A modified IL21 R-Fc fusion protein comprising the amino acid sequence encoded by the nucleic acid insert comprised by the vector deposited as ATCC Accession No. PTA- 120480.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:20.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:21.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:22.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:23.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:24.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:25.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:26.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:27.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:28.
- a modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:29.
- a method of treating a disease or disorder mediated by interaction of IL21 with IL21 R comprising administering to a patient in need thereof, the IL21 R ECD protein of any one of claims E1-E14, the pharmaceutical composition of any one of claims E29-E31 , or the fusion protein of any one of claims E15-E28 or E39-E50.
- E52 The method of E51 , wherein the disease or disorder is an inflammatory or autoimmune disease or disorder.
- E53 The method of 51 , wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
- GVHD graft-versus-host disease
- multiple sclerosis allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis
- spondyloarthropathy multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
- E55 The use of E54, wherein the disease or disorder is an inflammatory or autoimmune
- E56 The use of E54, wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
- GVHD graft-versus-host disease
- multiple sclerosis allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis
- spondyloarthropathy multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
- E50 for treatment of a disease or disorder mediated by interaction of IL21 with IL21 R in a patient in need thereof
- E58 The use of E57, wherein the disease or disorder is an inflammatory or autoimmune
- E59 The use of E57, wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
- GVHD graft-versus-host disease
- multiple sclerosis allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis
- spondyloarthropathy multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
- E61 The pharmaceutical composition of E60, wherein the disease or disorder is an
- E62 The pharmaceutical composition of E60, wherein the inflammatory disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis, spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma, scleroderma, vasculitis, and Behcet's Disease.
- Figure 1 depicts a diagram showing the X-ray crystal structure of two chains of IL21 R ECD at 2.8 Angstrom resolution, represented in ribbon diagram in grey shading (chain A) and black (chain B).
- the respective IL21 cytokines complexed to the receptor chains are
- the inset provides a close-up view of a flexible loop of the C-terminal domain of the receptor.
- the tryptophan (Trp or W) at position 148 of each chain is represented using sticks and the two tryptophans are shown interlocked through a pi stacking interaction which, without being limited to any particular theory, helps to stabilize the homodimer of the IL21 receptor.
- Figure 2 depicts a graph illustrating data from a differential scanning calorimetry thermogram demonstrating enhanced thermal stability of modified IL21 R ECD produced by substituting aspartate (Asp or D) at position 122 (D122) with para-acetyl phenylalanine (paF), relative to other positions within the IL21 R ECD.
- Figure 3 depicts an image of a Western blot showing various transiently expressed modified IL21 R ECD proteins comprising a His tag from HEK 293 cells. The cells were transiently transfected with each construct designated at the bottom of the gel and were cultured for five days before supernatant harvest. A western blot analysis was performed on the culture medium of the various mutants.
- the nitrocellulose membranes were probed with mouse anti-Histidine IgG (Qiagen, cat#: 34670) as primary antibody followed by horseradish peroxidase-conjugated rabbit anti-mouse IgG (Pierce, cat#: 32430) as secondary antibody.
- Each lane is loaded with an aliquot of conditioned medium obtained from a sample comprising an equal number of cells (hereinafter "equal cell count").
- ECD expression is directly compared to three different IL21 R ECD variants ECD1 , ECD2 and ECD3, which comprise mutations at and surrounding Trp 148 (see Table 1 below). This western blot analysis shows the enhanced expression level of modified IL21 R ECD proteins relative to wild type IL21 R ECD protein.
- Figure 4 is an image of a Western blot showing expression of various modified IL21 R ECD constructs from transiently transfected HEK 293 cells. Experimental methods were as described for Figure 3, supra.
- Figure 5 shows a graph depicting a differential scanning calorimetry thermogram demonstrating the thermal stability enhancement of modified IL21 R ECD constructs ECD1 , ECD2, and ECD4 as compared with wild type IL21 R ECD (WTECD).
- Figure 6 depicts a graph showing results from an analytical size exclusion
- Figure 7 depicts a graph showing the results of an analytical size exclusion chromatograph demonstrating enhanced resistance to aggregation of modified IL21 R constructs ECD1 and ECD2 compared with wild type ECD.
- the chromatograms were collected after subjecting the protein samples to an overnight incubation at 4°C (panel 7A showing no aggregation for any of the proteins) then subjecting the samples to a stress temperature of 40°C (panel 7B). After 40°C incubation overnight, the peak demonstrating 16% protein aggregation for wild type ECD is not present in the chromatograms for ECD1 or ECD2.
- Figure 8 is a diagram depicting a structural representation of an IL21 RFc fusion protein as disclosed herein.
- One IL21 R-Fc fusion protein is shown in black and the other IL21 R-Fc is shown in light gray.
- the two fusion proteins are shown as a homodimer stabilized by two disulfide bridges formed by two cysteines in each hinge region of the Fc of each fusion peptide.
- Figure 9 shows the results of an isothermal titration calorimetry (ITC) experiment comparing IL21 cytokine being titrated to a monomer version of IL21 R (ECD1) and to a homodimer ECD1 fusion construct comprising a linker and an Fc domain (ECD1-L3-FC3).
- Figure 9A presents the experimental thermogram of IL-21 titration to the monomer ECD1.
- Figure 9B shows the fitted curve of the data in 9A for molar ratio (N) determination.
- Figure 9C presents the experimental thermogram of IL-21 titration to the homodimer ECD1 Fc fusion construct ECD1-L3-FC3.
- Figure 9D shows the fitted curve of the data in 9C for molar ratio (N) determination.
- Figure 10 depicts the results of a BIAcore IL21 titration study demonstrating that linker L1 design allows the engagement of two IL21 cytokine per
- FIG. 10A depicts an experimental sensorgram showing saturation of the Response signal as IL-21 is titrated in to higher concentrations.
- ECD1-L1-FC1 is coated on the BIAcore chip for a theoretical Rmax response of 50.83RU.
- IL-21 cytokine is titrated progressively to saturation and the Rmax is recorded at 49.5RU as seen in the fitted curve of the Figure 10B. This observed value represents 97.4% of a full stoichiometry of two cytokine bound per IL-21 RFc homodimer on the chip.
- Figure 11 is an illustration showing the modifications of Linker L2 (bottom sequence: GGSEGEGSEGGS) from the amino acid sequence of Linker L1 (top sequence:
- Figure 12 is an image of a Western blot showing the results of transient expression of various modified IL21 RFc constructs ECD1-L1-FC1 , ECD1-L1-FC2, ECD7-L2-FC1 , and ECD1- L2-FC1 in cells.
- NCM nitrocellulose membrane
- the NCM were developed with a horseradish peroxidase-conjugated goat anti-human IgG (Sigma, cat#: A0170) antibody to detect human Fc.
- Each sample identified at the bottom of the gel was loaded in duplicate and each lane was loaded with an aliquot of conditioned medium normalized to equal number of cells.
- the far right lane contains a molecular weight standard with weights in kD.
- the migration position of the I L21 Fc fusion constructs is at approximately 150kD.
- a product-related smear of higher molecular weight species is created by heterogeneous O-linked glycosylated species.
- These post translational modifications have been characterized and located on specific serine residues in the linker L1 sequence.
- Comparison of the lanes for ECD1-L1-FC1 to ECD1- L1-FC2 shows identical banding profiles for both molecules. This demonstrates that the R445K mutation in the FC from FC1 to FC2 did not modify the SDS-PAGE properties of the molecule.
- Comparison of the lanes for ECD1-L1-FC1 to ECD1-L2-FC1 shows a noticeable reduction of the smear species highlighted by the accolade. This demonstrates that the linker L2 design with 2 serine position permutations did not support as efficiently if at all the O-linked glycosylation of the linker. Comparison of the lanes for ECD1-L2-FC1 to ECD7-L2-FC1 shows a very distinct improvement of the banding pattern for the ECD7 construct. Virtually all high molecular weight smears above the 150kD band have been removed. This demonstrates that the D122 mutation contained in the ECD7 design, through its stabilization effect and in conjunction with the L2 linker design, allows the production of a much more homogeneous molecule with reduced high molecular weight species.
- Figure 13 comprising panels A and B, shows an image of an isoelectric focusing (IEF) gel ( Figure 13A) and an image of a SDS PAGE gel ( Figure 13B) of modified IL21 RFc fusion construct ECD1-L1-FC1 to demonstrate the isoelectric point (pi)
- IEF isoelectric focusing
- Figure 13B SDS PAGE gel
- the empirical pi by IEF gel is visualized by a tight group of bands around the 4.2 pi standard.
- the sequence calculated pi for this construct is approximately 5.44.
- the difference is attributed to 6 N-linked glycosylation sites occupied and capped with sialic acids rendering the molecule significantly more acidic than its amino acid sequence predicted.
- the SDS PAGE gel presents both non-reduced and reduced conditions and highlights the purity of the material tested as well as the disulfide driven dimer nature of the molecule than can be fully reduced to monomer with dithiothreitol (DTT) treatment in the denaturing condition of this gel.
- DTT dithiothreitol
- Figure 14 shows a graph depicting the results of a differential scanning calorimetry thermogram demonstrating thermal stability enhancement of modified IL21 RFc fusion proteins ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1-FC1 , and ECD11-L1-FC1 compared to wild type IL21 RFc (ECD1). All constructs were identical in design comprising the same linker (L1) and Fc construct (FC1), varying only at the amino acid at position 122 of the IL21 R ECD component of the fusion protein. Wild type IL21 R ECD carried an Asp at position 122 (D122).
- ECD7, 8, 9, 10 and 1 1 carry at position 122, respectively, Ala, lie, Trp, Phe and Tyr.
- the CH2 and CH3 domain transition midpoints (Tm) are unmodified by these mutations.
- the wild type ECD (having D122) has a lower Tm at 50.22 degrees while the hydrophobic mutants for the 122 position overlap with an average gain of over 4 degrees to a higher Tm around approximately 54 degrees. This demonstrates the thermal stability enhancement brought by mutating D122 to a hydrophobic residue.
- FIG. 15 shows a diagram depicting the results of BIAcore studies of the binding of various modified IL21 RFc fusion proteins to FcRn.
- Solid lines are for ECD1-L1-FC3.
- This FC3 molecule carries the triple (Winter) mutation (3m) for reduced effector function (i.e., LLGL has been mutated to AAGA).
- the dashed lines represent ECD1-L1-FC1.
- This FC1 molecule is designed for extended in vivo half life and carries both the 3m mutations and the LS double mutant for enhanced binding to FcRn (see Table 4).
- the BIAcore experimental sensogram of this figure demonstrates the over 10 fold higher in vitro affinity of the FC1 construct vs.
- Figure 16 shows a graph depicting the pharmacokinetic (PK) profiles of various modified IL21 RFc fusion constructs in non-human primate administered intravenously (IV) or
- ECD1-L1-FC3 IV is shown by solid circles; ECD1- L1-FC1 IV is shown by solid squares; ECD1-L1-FC1 SC is shown by open squares; and WT ECD-L3-Fc3 IV is shown by open diamonds. Measuring the serum concentration of the molecules over time, this figure illustrates the progressive improvements of in vivo PK properties obtained with different IL21 RFc constructs.
- WTECD-L3-FC3 to ECD1-L1- FC3 demonstrates the combined advantage of the ECD1 mutation at position 148 and L1 linker, over wild type ECD and Linker L3.
- Figure 17 shows a graph depicting the results of a primary B cell proliferation assay and the ability of different IL21 RFc constructs to inhibit proliferation.
- the control human lgG1 molecule did not show neutralization activity in the range tested -.up to 50nM.
- Both ECD1-L1- FC1 and WTECD-L1-FC3 showed equal neutralization potency, slightly under 10nM. This demonstrates that the stabilizing mutations introduced in the design of ECD1 and the use of the LS mutations in FC1 and FC3 did not affect the in vitro biological function of the IL-21 receptor.
- Figure 18 shows a graph depicting the results of an I L21 -dependent primary T cell assay using modified IL-21 RFc constructs ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1- FC1 , and ECD1 1-L1-FC1.
- the cell proliferation was measured in counts per minute.
- the control human lgG1 in solid black line showed no neutralization activity in the range tested up to 1000nM. All other molecules, wild type ECD and D122 ECD mutants, showed equal
- modified IL21 R ECD proteins with improved properties, including greater in vitro expression levels, improved stability, decreased aggregation, and increased half-life compared to wild type unmodified IL21 R proteins.
- the half-life of the modified IL21 R ECD proteins can be further improved by fusing heterologous proteins, such as an Fc region of an antibody, to the IL21 R ECD protein.
- the fusion proteins can be further modified by substituting amino acids of the Fc region of the fusion protein.
- polypeptide encompasses native or artificial proteins, protein fragments and polypeptide analogs of a protein sequence.
- a polypeptide may be monomeric or polymeric.
- isolated protein is a protein, polypeptide or antibody that by virtue of its origin or source of derivation has one to four of the following: (1) is not associated with naturally associated components that accompany it in its native state, (2) is free of other proteins from the same species, (3) is expressed by a cell from a different species, or (4) does not occur in nature.
- a polypeptide that is chemically synthesized or synthesized in a cellular system different from the cell from which it naturally originates will be “isolated” from its naturally associated components.
- a protein may also be rendered substantially free of naturally associated components by isolation, using protein purification techniques well known in the art.
- a protein or polypeptide is “substantially pure,” “substantially homogeneous,” or
- substantially purified when at least about 60 to 75% of a sample exhibits a single species of polypeptide.
- the polypeptide or protein may be monomeric or multimeric.
- a substantially pure polypeptide or protein will typically comprise about 50%, 60%, 70%, 80% or 90% W/W of a protein sample, more usually about 95%, and preferably will be over 99% pure. Protein purity or homogeneity may be indicated by a number of means well known in the art, such as
- polyacrylamide gel electrophoresis of a protein sample followed by visualizing a single polypeptide band upon staining the gel with a stain well known in the art.
- higher resolution may be provided by using HPLC or other means well known in the art for purification.
- polypeptide fragment refers to a polypeptide that has an amino-terminal and/or carboxy-terminal deletion, but where the remaining amino acid sequence is identical to the corresponding positions in the full-length naturally-occurring sequence.
- fragments according to the invention may be made by truncation, e.g., by removal of one or more amino acids from the N and/or C-terminal ends of a polypeptide. Up to 10, up to 20, up to 30, up to 40 or more amino acids may be removed from the N and/or C terminal in this way. Fragments may also be generated by one or more internal deletions. In some embodiments, fragments are at least 5, 6, 8 or 10 amino acids long.
- the fragments are at least 14, at least 20, at least 50, or at least 70, 80, 90, 100, 150 or 200 amino acids long.
- amino acid substitutions of a protein or portion thereof are those which: (1) reduce susceptibility to proteolysis, (2) reduce susceptibility to oxidation, (3) alter binding affinity for forming protein complexes, or (4) confer or modify other physicochemical or functional properties.
- single or multiple amino acid substitutions may be made in the normally-occurring sequence.
- a conservative amino acid substitution should not substantially change the structural characteristics of the parent sequence.
- Examples of art-recognized polypeptide secondary and tertiary structures are described in Proteins, Structures and Molecular Principles (Creighton, Ed., W. H. Freeman and Company, New York (1984)); Introduction to Protein Structure (C. Branden and J. Tooze, eds., Garland Publishing, New York, N.Y. (1991)); and Thornton et al., Nature 354:105 (1991), which are each incorporated herein by reference.
- binding affinity is intended to refer to the dissociation rate of a particular antigen-antibody interaction.
- the K D is the ratio of the rate of dissociation, also called the “off-rate (k off )", to the association rate, or "on-rate (k on )".
- K D equals k off / k on and is expressed as a molar concentration (M). It follows that the smaller the K D , the stronger the affinity of binding. Therefore, a K D of 1 ⁇ indicates weaker binding affinity compared to a KD of 1 nM.
- KD values for antibodies can be determined using methods well established in the art. One method for determining the KD of an antibody is by using surface plasmon resonance, typically using a biosensor system such as a BIAcore® system.
- SPR surface plasmon resonance
- polynucleotide as referred to herein means a polymeric form of nucleotides of at least 10 bases in length, either ribonucleotides or deoxynbonucleotides or a modified form of either type of nucleotide.
- the term includes single and double stranded forms.
- isolated polynucleotide as used herein means a polynucleotide of genomic, cDNA, or synthetic origin or some combination thereof, which by virtue of its origin or source of derivation, the "isolated polynucleotide” has one to three of the following: (1) is not associated with all or a portion of a polynucleotides with which the "isolated polynucleotide” is found in nature, (2) is operably linked to a polynucleotide to which it is not linked in nature, or (3) does not occur in nature as part of a larger sequence.
- oligonucleotide as used herein includes naturally occurring, and modified nucleotides linked together by naturally occurring and non-naturally occurring oligonucleotide linkages. Oligonucleotides are a polynucleotide subset generally comprising a length of 200 bases or fewer. Preferably oligonucleotides are 10 to 60 bases in length and most preferably 12, 13, 14, 15, 16, 17, 18, 19, or 20 to 40 bases in length. Oligonucleotides are usually single stranded, e.g. for primers and probes; although oligonucleotides may be double stranded, e.g. for use in the construction of a gene mutant. Oligonucleotides of the invention can be either sense or antisense oligonucleotides.
- nucleotides as used herein includes deoxyribonucleotides and ribonucleotides.
- modified nucleotides as used herein includes nucleotides with modified or substituted sugar groups and the like.
- oligonucleotide linkages referred to herein includes oligonucleotides linkages such as phosphorothioate, phosphorodithioate, phosphoroselenoate, phosphorodiselenoate, phosphoroanilothioate, phoshoraniladate, phosphoroamidate, and the like. See e.g., LaPlanche et al., Nucl. Acids Res.
- oligonucleotide can include a label for detection, if desired.
- Operably linked sequences include both expression control sequences that are contiguous with the gene of interest and expression control sequences that act in trans or at a distance to control the gene of interest.
- expression control sequence means polynucleotide sequences that are necessary to effect the expression and processing of coding sequences to which they are ligated. Expression control sequences include appropriate transcription initiation, termination, promoter and enhancer sequences; efficient RNA processing signals such as splicing and polyadenylation signals; sequences that stabilize cytoplasmic mRNA; sequences that enhance translation efficiency (i.e., Kozak consensus sequence); sequences that enhance protein stability; and when desired, sequences that enhance protein secretion.
- control sequences differs depending upon the host organism; in prokaryotes, such control sequences generally include promoter, ribosomal binding site, and transcription termination sequence; in eukaryotes, generally, such control sequences include promoters and transcription termination sequence.
- control sequences is intended to include, at a minimum, all components whose presence is essential for expression and processing, and can also include additional components whose presence is advantageous, for example, leader sequences and fusion partner sequences.
- vector means a nucleic acid molecule capable of transporting another nucleic acid to which it has been linked.
- the vector is a plasmid, i.e., a circular double stranded DNA loop into which additional DNA segments may be ligated.
- the vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome.
- the vectors are capable of autonomous replication in a host cell into which they are introduced (e.g. , bacterial vectors having a bacterial origin of replication and episomal mammalian vectors).
- the vectors e.g., non-episomal mammalian vectors
- the vectors can be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome.
- certain vectors are capable of directing the expression of genes to which they are operatively linked. Such vectors are referred to herein as “recombinant expression vectors" (or simply, “expression vectors”).
- recombinant host cell means a cell into which an exogenous nucleic acid and/or recombinant vector has been introduced. It should be understood that “recombinant host cell” and “host cell” mean not only the particular subject cell but also the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term “host cell” as used herein.
- selectively hybridize means to detectably and specifically bind.
- Polynucleotides, oligonucleotides and fragments thereof in accordance with the invention selectively hybridize to nucleic acid strands under hybridization and wash conditions that minimize appreciable amounts of detectable binding to nonspecific nucleic acids.
- “High stringency” or “highly stringent” conditions can be used to achieve selective hybridization conditions as known in the art and discussed herein.
- high stringency or “highly stringent” conditions is the incubation of a polynucleotide with another polynucleotide, wherein one polynucleotide may be affixed to a solid surface such as a membrane, in a hybridization buffer of 6X SSPE or SSC, 50% formamide, 5X Denhardt's reagent, 0.5% SDS, 100 ⁇ g ml denatured, fragmented salmon sperm DNA at a hybridization temperature of 42°C for 12-16 hours, followed by twice washing at 55°C using a wash buffer of 1X SSC, 0.5% SDS. See also Sambrook et a/., supra, pp. 9.50-9.55.
- percent sequence identity in the context of nucleic acid sequences means the percent of residues when a first contiguous sequence is compared and aligned for maximum correspondence to a second contiguous sequence.
- the length of sequence identity comparison may be over a stretch of at least about nine nucleotides, usually at least about 18 nucleotides, more usually at least about 24 nucleotides, typically at least about 28 nucleotides, more typically at least about 32 nucleotides, and preferably at least about 36, 48 or more nucleotides.
- polynucleotide sequences can be compared using FASTA, Gap or Bestfit, which are programs in Wisconsin Package Version 10.0, Genetics Computer Group (GCG), Madison, Wisconsin.
- FASTA which includes, e.g., the programs FASTA2 and FAST A3, provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences (Pearson, Methods Enzymol. 183:63-98 (1990); Pearson, Methods Mol. Biol. 132:185-219 (2000); Pearson, Methods Enzymol. 266:227-258 (1996); Pearson, J. Mol. Biol. 276:71-84 (1998); herein incorporated by reference). Unless otherwise specified, default parameters for a particular program or algorithm are used.
- percent sequence identity between nucleic acid sequences can be determined using FASTA with its default parameters (a word size of 6 and the NOPAM factor for the scoring matrix) or using Gap with its default parameters as provided in GCG Version 6.1 , herein incorporated by reference.
- a reference to a nucleotide sequence encompasses its complement unless otherwise specified.
- a reference to a nucleic acid having a particular sequence should be understood to encompass its complementary strand, with its complementary sequence.
- percent sequence identity means a ratio, expressed as a percent of the number of identical residues over the number of residues compared.
- nucleic acid or fragment thereof when referring to a nucleic acid or fragment thereof, means that when optimally aligned with appropriate nucleotide insertions or deletions with another nucleic acid (or its complementary strand), there is nucleotide sequence identity in at least about 85%, preferably at least about 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98% or 99% of the nucleotide bases, as measured by any well- known algorithm of sequence identity, such as FASTA, BLAST or Gap, as discussed above.
- the term "substantial identity” means that two peptide sequences, when optimally aligned, such as by the programs GAP or BESTFIT using default gap weights, as supplied with the programs, share at least 70%, 75%, 80% or 85% sequence identity, preferably at least 90%, 91 %, 92%, 93%, 94% 95%, 96%, 97%, 98% or 99% sequence identity. In certain embodiments, residue positions that are not identical differ by conservative amino acid substitutions.
- a “conservative amino acid substitution” is one in which an amino acid residue is substituted by another amino acid residue having a side chain R group with similar chemical properties (e.g., charge or hydrophobicity).
- a conservative amino acid substitution will not substantially change the functional properties of a protein.
- the percent sequence identity may be adjusted upwards to correct for the conservative nature of the substitution. Means for making this adjustment are well-known to those of skill in the art. See, e.g., Pearson, Methods Mol. Biol. 243:307-31 (1994).
- Examples of groups of amino acids that have side chains with similar chemical properties include 1) aliphatic side chains: glycine, alanine, valine, leucine, and isoleucine; 2) aliphatic-hydroxyl side chains: serine and threonine; 3) amide-containing side chains: asparagine and glutamine; 4) aromatic side chains:
- amino acids substitution groups are:
- valine-leucine-isoleucine phenylalanine-tyrosine, lysine-arginine, alanine-valine,
- glutamate-aspartate glutamate-aspartate, and asparagine-glutamine.
- a conservative substitution or replacement is any change having a positive value in the PAM250 log-likelihood matrix disclosed in Gonnet et al., Science 256: 1443-45 (1992), herein incorporated by reference.
- a “moderately conservative” replacement is any change having a nonnegative value in the PAM250 log-likelihood matrix.
- Sequence identity for polypeptides is typically measured using sequence analysis software. Protein analysis software matches sequences using measures of similarity assigned to various substitutions, deletions and other modifications, including conservative amino acid substitutions.
- GCG contains programs such as "Gap” and "Bestfit” which can be used with default parameters, as specified with the programs, to determine sequence homology or sequence identity between closely related polypeptides, such as homologous polypeptides from different species of organisms or between a wild type protein and a mutein thereof. See, e.g., GCG Version 6.1. Polypeptide sequences also can be compared using FASTA using default or recommended parameters, see GCG Version 6.1.
- FASTA e.g., FASTA2 and FASTA3
- FASTA2 and FASTA3 provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences (Pearson, Methods Enzymol. 183:63-98 (1990); Pearson, Methods Mol. Biol. 132: 185-219 (2000)).
- Another preferred algorithm when comparing a sequence of the invention to a database containing a large number of sequences from different organisms is the computer program BLAST, especially blastp or tblastn, using default parameters, as supplied with the programs. See, e.g., Altschul et al., J. Mol. Biol. 215:403-410 (1990); Altschul et al., Nucleic Acids Res. 25:3389-402 (1997).
- the length of polypeptide sequences compared for homology will generally be at least about 16 amino acid residues, usually at least about 20 residues, more usually at least about 24 residues, typically at least about 28 residues, and preferably more than about 35 residues.
- potency is a measurement of biological activity and may be designated as IC 50 , or effective concentration of a protein needed to inhibit 50% of a biological activity in a cell which activity is mediated by the protein.
- phrases “effective amount” or “therapeutically effective amount” as used herein refers to an amount necessary (at dosages and for periods of time and for the means of
- An effective amount is at least the minimal amount, but less than a toxic amount, of an active agent which is necessary to impart therapeutic benefit to a subject.
- the term "compete”, as used herein with regard to polypeptide, means that a first polypeptide binds to an epitope in a manner sufficiently similar to the binding of a second polypeptide such that the result of binding of the first polypeptide with its cognate epitope is detectably decreased in the presence of the second polypeptide compared to the binding of the first polypeptide in the absence of the second polypeptide.
- the alternative, where the binding of the second polypeptide to its epitope is also detectably decreased in the presence of the first polypeptide can, but need not be the case. That is, a first polypeptide can inhibit the binding of a second polypeptide to its epitope without that second polypeptide inhibiting the binding of the first polypeptide to its respective epitope.
- each polypeptide detectably inhibits the binding of the other polypeptide with its cognate epitope or ligand, whether to the same, greater, or lesser extent, the polypeptides are said to "cross-compete" with each other for binding of their respective epitope(s).
- Both competing and cross-competing polypeptides are encompassed by the present invention. Regardless of the mechanism by which such competition or cross-competition occurs (e.g., steric hindrance, conformational change, or binding to a common epitope, or portion thereof), the skilled artisan would appreciate, based upon the teachings provided herein, that such competing and/or cross-competing polypeptides are encompassed and can be useful for the methods disclosed herein.
- IgG immunoglobulin gamma gene
- this class comprises lgG1 , lgG2, lgG3, and lgG4.
- mice this class comprises lgG1 , lgG2a, lgG2b, lgG3.
- immunoglobulin (lg) herein is meant a protein consisting of one or more polypeptides substantially encoded by immunoglobulin genes. Immunoglobulins include but are not limited to antibodies. Immunoglobulins may have a number of structural forms, including but not limited to full length antibodies, antibody fragments, and individual immunoglobulin domains.
- immunoglobulin (lg) domain herein is meant a region of an immunoglobulin that exists as a distinct structural entity as ascertained by one skilled in the art of protein structure, lg domains typically have a characteristic folding topology.
- the known lg domains in the IgG class of antibodies are the variable heavy chain domain (VH), the heavy chain constant domains - Cy1 , Cv2, Cy3 - together comprising the Cy domain which includes the hinge region between Cy1 and Cy2 , the variable domain of the light chain (VL), and the constant domain of the light chain (CL), which in humans comprises either the kappa (CK) or lambda (CA) light chain constant domain.
- Fc region is used to define a C-terminal region of an immunoglobulin heavy chain.
- the "Fc region” (also known as the "fragment crystallizable” or “tail” region) may be a native sequence Fc region or a variant Fc region.
- the human IgG heavy chain Fc region is usually defined to stretch from an amino acid residue at position Cys226, or from Pro230, to the carboxyl-terminus thereof. For all heavy chain constant region amino acid positions discussed in the present invention, numbering is according to the Eu index first described in Edelman et al., 1969, Proc. Natl. Acad. Sci.
- EU index as set forth in Kabat refers to the amino acid residue numbering system based on the human lgG1 EU antibody of Edelman et al. as set forth in Kabat 1991.
- the Fc region of an immunoglobulin generally comprises two constant domains, CH2 and CH3.
- an "Fc polypeptide,” as the term is used herein, comprises a CH2 and a CH3 domain and can include at least a portion of the hinge domain, but does not usually include the entire CH1 domain.
- an Fc region can be present in dimeric or monomeric form.
- Fc receptor and “FcR” describe a receptor that binds to the Fc region of an antibody.
- the preferred FcR is a native sequence human FcR.
- a preferred FcR is one which binds an IgG antibody (a gamma receptor) and includes receptors of the FcvRI, FcvRII, and FcvRIII subclasses, including allelic variants and alternatively spliced forms of these receptors.
- FcvRII receptors include FcvRIIA (an “activating receptor”) and FcvRIIB (an “inhibiting receptor”), which have similar amino acid sequences that differ primarily in the cytoplasmic domains thereof.
- FcRs are reviewed in Ravetch and Kinet, 1991 , Ann. Rev. Immunol., 9:457-92; Capel et al., 1994, Immunomethods, 4:25-34; and de Haas et al., 1995, J. Lab. Clin. Med., 126:330-41.
- FcR also includes the neonatal receptor, FcRn, which is responsible for the transfer of maternal IgGs to the fetus (Guyer et al., 1976, J. Immunol., 117:587; and Kim et al., 1994, J. Immunol., 24:249).
- compositions comprising such carriers are formulated by well known conventional methods (see, for example, Remington's Pharmaceutical Sciences, 18th edition, A. Gennaro, ed., Mack Publishing Co., Easton, PA, 1990; and Remington, The Science and Practice of Pharmacy 20th Ed. Mack Publishing, 2000).
- treating means reversing, alleviating, inhibiting the progress of, delaying the progression of, delaying the onset of, or preventing the disorder or condition to which such term applies, or one or more symptoms of such disorder or condition.
- treatment refers to the act of treating as “treating” is defined immediately above.
- treating also includes adjuvant and neo-adjuvant treatment of a subject.
- reference herein to “treatment” includes reference to curative, palliative and prophylactic treatment.
- This application discloses novel modified IL21 R ECD proteins having substantially improved useful characteristics. Such proteins, and fusion proteins thereof, may be used, for example, as antagonists of IL21 cytokine activity.
- Modified IL21 R proteins were designed by mutating the amino acid at or around W148, that is, within four amino acid residues of W148 using SEQ ID NO:1 as a reference, to produce constructs having at least one characteristic of greater stability, in vitro expression, reduced aggregation, and increased half- life in order to develop a therapeutic protein with improved characteristics over wild type proteins, making them improved therapeutics with decreased cost of goods making them also commercially desirable.
- modified IL21 R ECD proteins having one or more amino acid substitutions at or within four amino acid residues on either side, i.e., "flanking", W148 with respect to SEQ ID NO: 1.
- the substitution at W148 or within four amino acids residues on either side of W148 may be any naturally occurring amino acid, including glycine, alanine, leucine, methionine, phenylalanine, lysine, glutamine, glutamic acid, serine, proline, valine, isoleucine, cysteine, tyrosine, histidine, arginine, asparagine, aspartic acid, and threonine.
- W148 is substituted with a serine or asparagine.
- the substitution may be at position 49 with respect to SEQ ID NO:1. In further embodiments, the substitutions may be made at one or more of positions 49, 147, 148, 149 or 150.
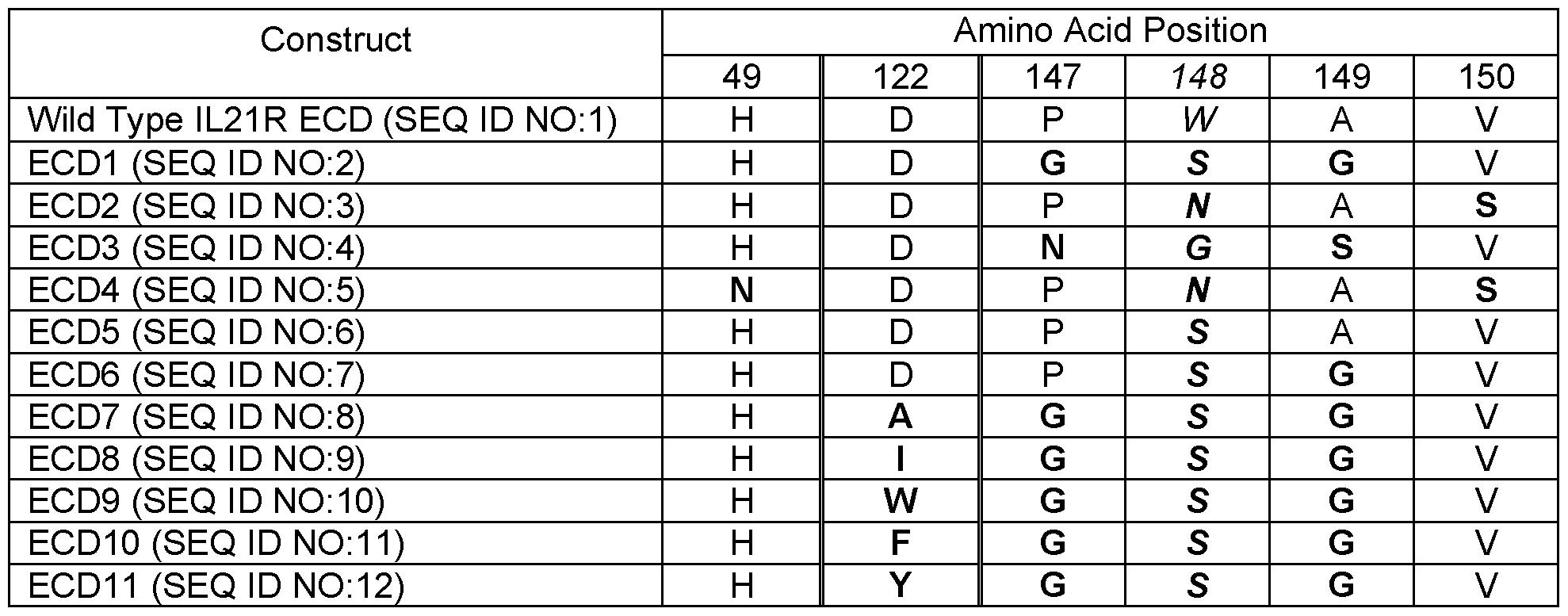
- the modified IL21 R ECD may comprise substitutions selected from the group consisting of: a. Glycine at position 147, serine at position 148, glycine at position 149 (SEQ ID NO:2) ;
- Serine at position 148 Serine at position 148, glycine at position 149 (SEQ ID NO:7); and e. Asparagine at position 49, asparagine at position 148, and serine at position 150 (SEQ ID NO:5).
- the modified IL21 R ECD protein may further comprise a substitution of the aspartic acid residue at position 122 (D122).
- D122 may be substituted for by alanine, isoleucine, tryptophan, phenylalanine, or tyrosine.
- Table 1 provides a summary of the various exemplary IL21 R ECD mutations disclosed herein.
- Table 2 provides the complete sequences of various IL21 R ECD proteins.
- Table 1 Summary of Amino Acid Substitutions in Modified IL21 R ECD Proteins Relative to Wild
- Type IL21 R Protein (mutated positions are in bold)
- This application also discloses fusion proteins of the modified IL21 R ECD proteins disclosed herein.
- IL21 R fusion polypeptide or “IL21 R fusion protein” refers to a fusion of one or more amino acid residues (such as a heterologous protein or peptide) at the N- terminus or C-terminus of any modified IL21 R ECD protein described herein.
- fusion protein refers to a protein or polypeptide that has an amino acid sequence derived from two or more proteins. The fusion protein may also include linking regions of amino acids between amino acid portions derived from separate proteins.
- Heterologous peptides and polypeptides include, but are not limited to, an epitope (e.g., FLAG) or a tag sequence (e.g., His 6 , and the like) to allow for the detection and/or isolation of an IL21 R polypeptide mutant; a transmembrane receptor protein or a portion thereof, such as an extracellular domain or a transmembrane and intracellular domain; a ligand or a portion thereof which binds to a transmembrane receptor protein; an enzyme or portion thereof which is catalytically active; a polypeptide or peptide which promotes oligomerization, such as a leucine zipper domain; a polypeptide or peptide which increases stability, such as an immunoglobulin constant region (e.g., an Fc domain); a half life-extending sequence comprising a combination of two or more (e.g., 2, 5, 10, 15, 20, 25, etc) naturally occurring or non-naturally occurring charged and/or uncharged amino acids
- IL21 R fusion proteins can be made by fusing heterologous sequences at either the N- terminus or at the C-terminus of a modified IL21 R ECD protein.
- a heterologous sequence can be an amino acid sequence or a non-amino acid-containing polymer.
- Heterologous sequences can be fused either directly to the modified IL21 R ECD protein either chemically or by recombinant expression from a single polynucleotide or they may be joined via a linker or adapter molecule.
- a peptidyl linker or adapter molecule can be one or more amino acid residues (or -mers), e.g., 1 , 2, 3, 4, 5, 6, 7, 8, or 9 residues (or -mers), preferably from 10 to 50 amino acid residues (or -mers), e.g., 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, or 50 residues (or -mers), and more preferably from 15 to 35 amino acid residues (or -mers).
- a linker or adapter molecule can also be designed with a cleavage site for a DNA restriction endonuclease or for a protease to allow for the separation of the fused moieties.
- a linker can, but need not, be employed.
- the linker can be made up of amino acids linked together by peptide bonds, i.e., a peptidyl linker.
- the linker is made up of from 1 to 20 or more amino acids linked by peptide bonds, wherein the amino acids are selected from the 20 naturally occurring amino acids.
- the amino acids are selected from the amino acids glycine, serine, and glutamate.
- suitable linkers include: GSGEGEGSEGSG (SEQ ID NO:13); GGSEGEGSEGGS (SEQ ID NO:14); and GGGS (SEQ ID NO:15). While a linker of 12 amino acid residues has been found to work with the modified IL21 RFc fusion proteins disclosed herein, the present invention contemplates linkers of any length or composition. Exemplary linkers are shown in Table 3. Table 3: Linker Sequences
- linkers described herein are exemplary, and linkers that are much longer and which include other residues are contemplated by the present invention b) Fc Proteins
- the modified IL21 R ECD proteins are fused to an Fc domain, e.g., one or more domains of an Fc region of a human IgG.
- Antibodies comprise two functionally independent parts, a variable domain known as "Fab,” that binds an antigen, and a constant domain known as "Fc,” that is involved in, among other things, effector functions such as complement activation and attack by phagocytic cells.
- An Fc has a long serum half-life, whereas a Fab is short-lived (Capon et ah, 1989, Nature 337: 525-31) such that when joined together with a therapeutic protein, an Fc domain can provide longer half-life or incorporate such effector functions as Fc receptor binding, protein A binding, complement fixation, and other characteristics that are desirable in a therapeutic protein.
- the modified ECD Fc fusion protein comprises the amino acid sequence encoded by the polynucleotide insert of the vector deposited with the ATCC on July 17, 2013, as ECD1-L1- FC1 with the ATCC Accession Number PTA-120480.
- the resulting modified IL21 RFc fusion protein can be purified, for example, by the use of a Protein A affinity column. Peptides and proteins fused to an Fc region have been found to exhibit a substantially greater half-life in vivo than the unfused counterpart. Also, a fusion to an Fc region allows for dimerization/multimerization of the fusion polypeptide.
- the Fc region can be a naturally occurring Fc region, such as an lgG1 , lgG2, lgG3 or lgG4 Fc.
- an Fc region is a human lgG1 Fc, e.g., SEQ ID NO: 16.
- the Fc region can also be altered to improve certain qualities, such as modification to reduce effector function, e.g., SEQ ID NO: 19, or modified to improve therapeutic qualities, such as increased circulation time (half-life), e.g., SEQ ID NO: 17.
- compositions comprising modified IL21 R ECD proteins, or fusion proteins thereof, are within the scope of the present invention, and are specifically contemplated in light of the identification of several modified IL21 R ECD sequences and fusion proteins exhibiting enhanced properties.
- Such pharmaceutical compositions can comprise a
- a modified IL21 R ECD protein, or fusion protein thereof in admixture with a pharmaceutically or physiologically acceptable formulation agent selected for suitability with the mode of administration.
- Acceptable formulation agents preferably are nontoxic to recipients at the dosages and concentrations employed.
- the pharmaceutical composition can contain formulation agent(s) for modifying, maintaining, or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption, or penetration of the composition.
- formulation agent(s) for modifying, maintaining, or preserving for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption, or penetration of the composition.
- Suitable formulation agents include, but are not limited to, amino acids (such as glycine, glutamine, asparagine, arginine, or lysine), antimicrobials, antioxidants (such as ascorbic acid, sodium sulfite, methionine or sodium hydrogen-sulfite), buffers (such as borate, bicarbonate, Tris-HCI, histidine, citrates, phosphates, or other organic acids), bulking agents (such as mannitol or glycine), chelating agents (such as ethylenediamine tetraacetic acid (EDTA)), complexing agents (such as caffeine, polyvinylpyrrolidone, beta-cyclodextrin, or hydroxypropyl-beta-cyclodextrin), fillers, monosaccharides, disaccharides, and other carbohydrates (such as glucose, mannose, or dextrins), proteins (such as serum albumin, gelatin, or immunoglobulins), coloring, flavoring and di
- compositions will be determined by a skilled artisan depending upon, for example, the intended route of administration, delivery format, and desired dosage (see, e.g., Remington's Pharmaceutical Sciences, supra). Such compositions can influence the physical state, stability, rate of in vivo release, and rate of in vivo clearance of the modified IL21 R ECD protein or fusion protein thereof.
- the primary vehicle or carrier in a pharmaceutical composition can be either aqueous or non-aqueous in nature.
- a suitable vehicle or carrier for injection can be water, physiological saline solution, or artificial cerebrospinal fluid, possibly supplemented with other materials common in compositions for parenteral administration.
- Neutral buffered saline or saline mixed with serum albumin are further exemplary vehicles.
- Other exemplary vehicles are further exemplary vehicles.
- compositions comprise Histidine or Tris buffer of about pH 6.0-8.5, which can further include sorbitol or a suitable substitute.
- IL21 RFc fusion protein compositions can be prepared for storage by mixing the selected composition having the desired degree of purity with optional formulation agents (Remington's Pharmaceutical Sciences, supra) in the form of a an aqueous solution.
- the pharmaceutical compositions can be selected for parenteral delivery. Alternatively, the compositions can be selected for inhalation or for delivery through the digestive tract, such as orally.
- the preparation of such pharmaceutically acceptable compositions is within the skill of the art.
- the formulation components are present in concentrations that are acceptable to the site of administration. For example, buffers are used to maintain the composition at
- physiological pH typically within a pH range of from about 6 to about 8.
- the therapeutic compositions for use in this invention can be in the form of a pyrogen-free, parenterally acceptable, aqueous solution comprising the desired modified IL21 R ECD protein, or fusion protein thereof, in a
- a particularly suitable vehicle for parenteral injection is sterile distilled water in which a modified IL21 R ECD protein, or fusion protein thereof, is formulated as a sterile, isotonic solution, properly preserved.
- Yet another preparation can involve the formulation of the desired molecule with an agent, such as injectable microspheres, bio-erodible particles, polymeric compounds (such as polylactic acid or polyglycolic acid), beads, or liposomes, that provides for the controlled or sustained release of the product which can then be delivered via a depot injection.
- Hyaluronic acid can also be used, and this can have the effect of promoting sustained duration in the circulation.
- Other suitable means for the introduction of the desired molecule include implantable drug delivery devices.
- a pharmaceutical composition can be formulated for inhalation.
- the pharmaceutical composition can be formulated as a dry powder for inhalation.
- Inhalation solutions can also be formulated with a propellant for aerosol delivery. In yet another embodiment, solutions can be nebulized. Pulmonary administration is further described in International Publication No. WO94/20069, which describes the pulmonary delivery of chemically modified proteins.
- formulations that are administered in this fashion can be formulated with or without those carriers customarily used in the compounding of solid dosage forms such as tablets and capsules.
- a capsule can be designed to release the active portion of the formulation at the point in the gastrointestinal tract when bioavailability is maximized and pre-systemic degradation is minimized.
- Additional agents can be included to facilitate absorption. Diluents, flavorings, low melting point waxes, vegetable oils, lubricants, suspending agents, tablet disintegrating agents, and binders can also be employed.
- Another pharmaceutical composition can involve an effective quantity of modified IL21 R ECD protein, or a fusion protein thereof, in a mixture with non-toxic excipients that are suitable for the manufacture of tablets.
- excipients include, but are not limited to, inert diluents, such as calcium carbonate, sodium carbonate or bicarbonate, lactose, or calcium phosphate; or binding agents, such as starch, gelatin, or acacia; or lubricating agents such as magnesium stearate, stearic acid, or talc.
- sustained- or controlled-delivery formulations including formulations involving modified IL21 R ECD proteins, or fusion proteins thereof, in sustained- or controlled-delivery formulations.
- Techniques for formulating a variety of other sustained- or controlled-delivery means such as liposome carriers, bio-erodible microparticles or porous beads and depot injections, are also known to those skilled in the art (see, e.g., International Publication No. W093/15722, which describes the controlled release of porous polymeric microparticles for the delivery of pharmaceutical compositions, and Wischke & Schwendeman, 2008, Int. J. Pharm. 364: 298-327, and Freiberg & Zhu, 2004, Int. J. Pharm. 282: 1-18, which discuss microsphere/microparticle preparation and use).
- a hydrogel is an example of a sustained- or controlled-delivery formulation.
- sustained-release preparations include semipermeable polymer matrices in the form of shaped articles, e.g. films, or microcapsules.
- Sustained release matrices can include polyesters, hydrogels, polylactides (U.S. Patent No. 3,773,919 and European Patent No. 0 058 481), copolymers of L-glutamic acid and gamma ethyl-L-glutamate (Sidman et ah, 1983, Biopolymers 22: 547-56), poly(2-hydroxyethyl-methacrylate) (Langer et ah, 1981 , J. Biomed. Mater. Res. 15: 167-277 and Langer, 1982, Chem.
- Sustained-release compositions can also include liposomes, which can be prepared by any of several methods known in the art. See, e.g., Epstein et ah, 1985, Proc. Natl. Acad. Sci. U.S.A. 82: 3688-92; and European Patent Nos. 0 036 676, 0 088 046, and 0 143 949.
- the pharmaceutical composition to be used for in vivo administration typically should be sterile. This can be accomplished by filtration through sterile filtration membranes. Where the composition is lyophilized, sterilization using this method can be conducted either prior to, or following, lyophilization and reconstitution.
- the composition for parenteral administration can be stored in lyophilized form or in a solution.
- parenteral compositions generally are placed into a container having a sterile access port, for example, an intravenous solution bag or vial having a stopper pierceable by a hypodermic injection needle.
- the parenteral composition can be diluted into parenterally acceptable diluents (e.g., saline and 5% Dextrose).
- the pharmaceutical composition can be stored in sterile vials as a solution, suspension, gel, emulsion, solid, or as a dehydrated or lyophilized powder.
- Such formulations can be stored either in a ready-to-use form or in a form (e.g., lyophilized) requiring reconstitution prior to administration.
- kits for producing a single-dose administration unit can each contain both a first container having a dried protein and a second container having an aqueous formulation. Also included within the scope of this invention are kits containing single and multi-chambered pre-filled syringes (e.g., liquid syringes and dual chamber syringes).
- the present invention is directed to a pharmaceutical composition
- a pharmaceutical composition comprising a modified IL21 RFc fusion protein formulated as a powder for injection after reconstitution to a solution for injection.
- the formulation components may range in
- the composition upon reconstitution , will contain 100 mg/ml of IL21 RFc fusion protein, 40 mM tromethamine (Tris base), 0.05 mg/ml EDTA, 100 mg/ml sucrose, 0.2 mg/ml polysorbate 80.
- Selecting an administration regimen for a therapeutic depends on several factors, including the serum or tissue turnover rate of the entity, the level of symptoms, the
- an administration regimen maximizes the amount of therapeutic delivered to the patient consistent with an acceptable level of side effects. Accordingly, the amount of biologic delivered depends in part on the particular entity and the severity of the condition being treated. Guidance in selecting appropriate doses of antibodies, Fc fusion therapeutic proteins, cytokines, and small molecules are available (see, e.g., Wawrzynczak, 1996, Antibody Therapy, Bios Scientific Pub.
- Determination of the appropriate dose is made by the clinician, e.g., using parameters or factors known or suspected in the art to affect treatment or predicted to affect treatment. Generally, the dose begins with an amount somewhat less than the optimum dose and it is increased by small increments thereafter until the desired or optimum effect is achieved relative to any negative side effects.
- Important diagnostic measures include those of symptoms of, e.g., the inflammation or level of inflammatory cytokines produced.
- Actual dosage levels of the active ingredients in the pharmaceutical compositions of the present disclosure may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient.
- the selected dosage level will depend upon a variety of pharmacokinetic factors including the activity of the particular compositions of the present disclosure employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compositions employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
- compositions comprising modified IL21 R ECD of the disclosure can be provided by continuous infusion, or by doses at intervals of, e.g., one day, one week, or 1-7 times per week.
- Doses may be provided intravenously, subcutaneously, topically, orally, nasally, rectally, intramuscular, intracerebrally, or by inhalation.
- a specific dose protocol is one involving the maximal dose or dose frequency that avoids significant undesirable side effects.
- a total weekly dose may be at least 0.05 ⁇ g kg body weight, at least 0.2 ⁇ g kg, at least 0.5 ⁇ g kg, at least 1 ⁇ g kg, at least 10 ⁇ g kg, at least 100 ⁇ g kg, at least 0.2 mg/kg, at least 1.0 mg/kg, at least 2.0 mg/kg, at least 10 mg/kg, at least 15 mg/kg, at least 20 mg/kg, at least 25 mg/kg, or at least 50 mg/kg (see, e.g., Yang, et al., 2003, New Engl. J. Med. 349:427-434; Herold, et al., 2002, New Engl. J. Med. 346:1692-1698; Liu, et al., 1999, J. Neurol. Neurosurg. Psych. 67:451-456;
- the dose may be at least 15 ⁇ g, at least 20 ⁇ g, at least 25 ⁇ g, at least 30 ⁇ g, at least 35 ⁇ g, at least 40 ⁇ g, at least 45 ⁇ g, at least 50 ⁇ g, at least 55 ⁇ g, at least 60 ⁇ g, at least 65 ⁇ g, at least 70 ⁇ g, at least 75 ⁇ g, at least 80 ⁇ g, at least 85 ⁇ g, at least 90 ⁇ g, at least 95 ⁇ g, or at least 100 ⁇ g.
- the doses administered to a subject may number at least 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , or 12, or more.
- the dosage administered to a patient may be 0.0001 mg/kg to 100 mg/kg of the patient's body weight.
- the dosage may be between 0.0001 mg/kg and 20 mg/kg, 0.0001 mg/kg and 10 mg/kg, 0.0001 mg/kg and 5 mg/kg, 0.0001 and 2 mg/kg, 0.0001 and 1 mg/kg, 0.0001 mg/kg and 0.75 mg/kg, 0.0001 mg/kg and 0.5 mg/kg, 0.0001 mg/'kg to 0.25 mg/kg, 0.0001 to 0.15 mg/kg, 0.0001 to 0.10 mg/kg, 0.001 to 0.5 mg/kg, 0.01 to 0.25 mg/kg or 0.01 to 0.10 mg/kg of the patient's body weight.
- the dosage of the therapeutic protein of the disclosure may be calculated using the patient's weight in kilograms (kg) multiplied by the dose to be administered in mg/kg.
- the dosage of the proteins of the disclosure may be 150 ⁇ g/kg or less, 125 ⁇ g/kg or less, 100 ⁇ g/kg or less, 95 ⁇ g/kg or less, 90 ⁇ g/kg or less, 85 ⁇ /kg or less, 80 ⁇ /kg or less, 75 ⁇ /kg or less, 70 ⁇ /kg or less, 65 ⁇ /kg or less, 60 ⁇ /kg or less, 55 ⁇ /kg or less, 50 ⁇ /kg or less, 45 ⁇ /kg or less, 40 ⁇ /kg or less, 35 ⁇ /kg or less, 30 ⁇ /kg or less, 25 ⁇ /kg or less, 20 ⁇ /kg or less, 15 ⁇ /kg or less, 10 ⁇ /kg or less, 5 ⁇ /kg or less, 2.5 ⁇ /kg or less, 2 ⁇ /kg or less, 1.5 ⁇ /kg or
- Unit dose of the therapeutic proteins of the disclosure may be 0.1 mg to 20 mg, 0.1 mg to 15 mg, 0.1 mg to 12 mg, 0.1 mg to 10 mg, 0.1 mg to 8 mg, 0.1 mg to 7 mg, 0.1 mg to 5 mg, 0.1 to 2.5 mg, 0.25 mg to 20 mg, 0.25 to 15 mg, 0.25 to 12 mg, 0.25 to 10 mg, 0.25 to 8 mg, 0.25 mg to 7 m g, 0.25 mg to 5 mg, 0.5 mg to 2.5 mg, 1 mg to 20 mg, 1 mg to 15 mg, 1 mg to 12 mg, 1 mg to 10 mg, 1 mg to 8 mg, 1 mg to 7 mg, 1 mg to 5 mg, or 1 mg to 2.5 mg.
- the dosage of the therapeutic proteins of the disclosure may achieve a serum titer of at least 0.1 ⁇ g/ml, at least 0.5 ⁇ g/ml, at least 1 ⁇ g/ml, at least 2 ⁇ g/ml, at least 5 ⁇ g/ml, at least 6 ⁇ g/ml, at least 10 ⁇ g/ml, at least 15 ⁇ g/ml, at least 20 ⁇ g/ml, at least 25 ⁇ g/ml, at least 50 ⁇ g/ml, at least 100 ⁇ g/ml, at least 125 ⁇ g/ml, at least 150 v, at least 175 ⁇ g/ml, at least 200 ⁇ g/ml, at least 225 g/ml, at least 250 g/ml, at least 275 g/ml, at least 300 g/ml, at least 325 g/ml, at least 350 g/ml, at least 375 g/ml /ml, or at least 400
- the dosage of the antibodies of the disclosure may achieve a serum titer of at least 0.1 g/ml, at least 0.5 g/ml, at least 1 g/ml, at least, 2 g/ml, at least 5 g/ml, at least 6 g/ml, at least 10 g/ml, at least 15 g/ml, at least 20 g/ml, at least 25 g/ml, at least 50 g/ml, at least 100 g/ml, at least 125 g/ml, at least 150 g/ml, at least 175 g/ml, at least 200 g/ml, at least 225 g/ml, at least 250 g/ml, at least 275 g/ml, at least 300 g/ml, at least 325 g/ml, at least 350 g/ml, at least 375 g/ml, or at least 400 g/ml in the subject.
- Doses of therapeutic proteins of the disclosure may be repeated and the administrations may be separated by at least 1 day, 2 days, 3 days, 5 days, 10 days, 15 days, 30 days, 45 days, 2 months, 75 days, 3 months, or at least 6 months.
- An effective amount for a particular patient may vary depending on factors such as the condition being treated, the overall health of the patient, the method route and dose of administration and the severity of side effects (see, e.g., Maynard, et al., 1996, A Handbook of SOPs for Good Clinical Practice, Interpharm Press, Boca Raton, Fla.; Dent, 2001 , Good Laboratory and Good Clinical Practice, Urch Publ, London, UK).
- the route of administration may be by, e.g., topical or cutaneous application, injection or infusion by intravenous, intraperitoneal, intracerebral, intramuscular, intraocular, intraarterial, intracerebrospinal, intralesional, or by sustained release systems or an implant (see, e.g.,
- the composition may also include a solubilizing agent and a local anesthetic such as lidocaine to ease pain at the site of the injection.
- pulmonary administration can also be employed, e.g., by use of an inhaler or nebulizer, and formulation with an aerosolizing agent.
- an inhaler or nebulizer e.g., a pressurized gas, a pressurized gas, or a pressurized gas.
- an engineered antibody or engineered antibody conjugate, combination therapy, or a composition of the disclosure is administered using Alkermes AIRTM pulmonary drug delivery technology (Alkermes, Inc., Cambridge, Mass.).
- the frequency of dosing will depend upon the pharmacokinetic parameters of the modified IL21 R ECD protein or fusion protein thereof in the formulation being used. Typically, a clinician will administer the composition until a dosage is reached that achieves the desired effect.
- the composition can therefore be administered as a single dose, as two or more doses (which may or may not contain the same amount of the desired molecule) over time, or as a continuous infusion via an implantation device or catheter. Further refinement of the appropriate dosage is routinely made by those of ordinary skill in the art and is within the ambit of tasks routinely performed by them. Appropriate dosages can be ascertained through use of appropriate dose-response data.
- the route of administration of the pharmaceutical composition is in accord with known methods, e.g., orally; through injection by subcutaneous, intravenous, intraperitoneal, intracerebral (intraparenchymal), intracerebroventncular, intramuscular, intraocular, intraarterial, intraportal, or intralesional routes; by sustained release systems (which may also be injected); or by implantation devices.
- the compositions can be administered by bolus injection or continuously by infusion, or by implantation device.
- the composition can be administered locally via implantation of a membrane, sponge, or other appropriate material onto which the desired molecule has been absorbed or encapsulated.
- the device can be implanted into any suitable tissue or organ, and delivery of the desired molecule can be via diffusion, timed-release bolus, or continuous administration.
- drug e.g., a modified IL21 R ECD protein, or fusion protein thereof as disclosed herein
- a variety of different approaches can be employed.
- a hydrogel comprising a polymer such as a gelatin (e.g., bovine gelatin, human gelatin, or gelatin from another source) or a naturally-occurring or a synthetically generated polymer can be employed.
- Any percentage of polymer e.g., gelatin
- a hydrogel such as 5, 10, 15 or 20%.
- concentration can depend on a variety of factors, such as the therapeutic profile desired and the pharmacokinetic profile of the therapeutic molecule.
- polymers that can be incorporated into a hydrogel include polyethylene glycol (“PEG”), polyethylene oxide, polyethylene oxide-co-polypropylene oxide, co-polyethylene oxide block or random copolymers, polyvinyl alcohol, polyvinyl pyrrolidinone), poly(amino acids), dextran, heparin, polysaccharides, polyethers and the like.
- PEG polyethylene glycol
- polyethylene oxide polyethylene oxide-co-polypropylene oxide
- co-polyethylene oxide block or random copolymers polyvinyl alcohol, polyvinyl pyrrolidinone), poly(amino acids), dextran, heparin, polysaccharides, polyethers and the like.
- PEG polyethylene glycol
- polyethylene oxide polyethylene oxide-co-polypropylene oxide
- polyvinyl alcohol polyvinyl pyrrolidinone
- poly(amino acids) poly(amin
- cross- linking can be achieved via a methacrylation reaction involving methacrylic anhydride.
- a high degree of cross-linking may be desirable while in other situations a lower degree of crosslinking is preferred.
- a higher degree of crosslinking provides a longer sustained release.
- a higher degree of crosslinking may provide a firmer hydrogel and a longer period over which drug is delivered.
- Any ratio of polymer to crosslinking agent e.g., methacrylic anhydride
- the ratio of polymer to crosslinker can be, e.g., 8: 1 , 16: 1 , 24: 1 , or 32: 1.
- the hydrogel polymer is gelatin and the crosslinker is methacrylate
- ratios of 8: 1 , 16: 1 , 24:1 , or 32: 1 methyacrylic anhydride:gelatin can be employed.
- Modified IL21 R ECD proteins, fusion proteins thereof, and pharmaceutical compositions comprising the modified IL21 R ECD proteins or fusion proteins thereof can be used to regulate at least one IL21-mediated or IL21 R-mediated immune response, such as one or more of cell proliferation, cytokine expression or secretion, chemokine secretion, and cytolytic activity, of T cells, B cells, NK cells, macrophages, or synovial cells.
- IL21-mediated or IL21 R-mediated immune response such as one or more of cell proliferation, cytokine expression or secretion, chemokine secretion, and cytolytic activity, of T cells, B cells, NK cells, macrophages, or synovial cells.
- the proteins of the invention can be used to inhibit the activity (e.g., proliferation, differentiation, and/or survival) of an immune or hematopoietic cell (e.g., a cell of myeloid, lymphoid, or erythroid lineage, or precursor cells thereof), and, thus, can be used to treat a variety of diseases or disorders mediated by interaction of IL21 with IL21 R.
- an immune or hematopoietic cell e.g., a cell of myeloid, lymphoid, or erythroid lineage, or precursor cells thereof
- the modified IL21 R ECD proteins, or fusion proteins thereof, or pharmaceutical compositions thereof, of this disclosure can used in the treatment or prevention of IL21 or IL21 R-mediated disorders.
- the invention provides for use of the modified IL21 R ECD proteins, or fusion proteins thereof, or
- compositions thereof, of this disclosure in the manufacture of a medicament for use in treatment or prevention of IL21 or IL21 R-mediated disorders.
- this application discloses modified IL21 R ECD proteins, or fusion proteins thereof, or pharmaceutical compositions thereof, for use in treatment of IL21 or I L21 R-mediated disorders.
- this application discloses pharmaceutical compositions comprising the modified IL21 R ECD proteins, or fusion proteins thereof, of this disclosure for use in treating or preventing IL21 or I L21 R-mediated diseases.
- immune disorders examples include, but are not limited to, transplant rejection, graft-versus-host disease (GVHD), allergies (for example, atopic allergy), and autoimmune diseases, including diabetes mellitus, arthritic disorders (including rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, and ankylosing spondylitis), spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis (including atopic dermatitis and eczematous dermatitis), psoriasis, Sjogren's syndrome, IBD (including Crohn's disease and ulcerative colitis), asthma (including intrinsic asthma and allergic asthma), scleroderma, vasculitis, and Behcet's Disease.
- GVHD
- a disorder or condition mediated by the interaction between IL21 and IL21 R can be treated by administering modified IL21 R ECD protein, or fusion protein thereof, or a pharmaceutical composition thereof, as described herein, to a patient in need thereof in the amount of a therapeutically effective dose.
- the administration can be performed as described herein, such as by IV injection, intraperitoneal injection, intramuscular injection, or orally in the form of a tablet or liquid formation.
- a desired dosage can be determined by a clinician, as described herein, and can represent a therapeutically effective dose of modified IL21 R ECD or fusion protein.
- a therapeutically effective dose will depend, inter alia, upon the administration schedule, the unit dose of agent administered, whether the composition is administered in combination with other therapeutic agents, the immune status and the health of the recipient.
- therapeutically effective dose means that amount of modified IL21 R ECD protein or fusion protein thereof that elicits the biological or medicinal response in a tissue system, animal, or human being sought by a researcher, medical doctor, or other clinician, which includes alleviation of the symptoms of the disease or disorder being treated.
- Vector EDC1-L1-FC1 having ATCC Accession No. PTA-120480, comprises a DNA insert encoding modified IL21 R ECD1 , linker 1 , and Fc variant designated FC1.
- the deposit was made under the provisions of the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purpose of Patent Procedure and Regulations thereunder (Budapest Treaty). This assures maintenance of a viable culture of the deposit for 30 years from the date of deposit.
- IL21 R protein complexed with its cytokine was solved. More specifically, IL21 and IL21 R ECD were expressed in CHO cells and purified
- cytokine and receptor were then mixed in a one to one ratio and applied to a SourceQ anion exchanger chromatography to further purify the complex. Selected fractions were concentrated to 7.3 mg/ml and a hanging drop crystallization screen was realized. After optimization, good quality crystals were obtained from 1720mM Ammonium Sulfate, 10mM Cobaltous Chloride and 100mM MES buffer at pH 6.43. The crystals were frozen and data was collected at the National Synchrotron Light Source (NSLS, Brookhaven, NY) produced diffraction to 2.6A resolution. The symmetry space group was 14122 and the structure was solved through molecular replacement technique using the IL4/IL4R complex structure available from the protein data bank.
- Figure 1 Working from the X-ray structure of the IL21 receptor ( Figure 1), various mutants of human IL21 R ECD with a C- terminal hexa histidine tag were designed.
- the tryptophan at position 148 is structurally in a solvent exposed flexible loop and may create an unfavorable conformation due to its highly hydrophobic nature.
- D122 aspartate residue at position 122
- paF para-acetyl phenylalanine
- Additional D122 position mutants substituted with hydrophobic residues were designed to mimic the stabilizing effect observed with the substitution of the non-natural amino acid paF.
- Table 2 lists various IL21 R ECD mutants and their amino acid sequences. Bolded and underlined residues in each of the modified IL21 R ECD constructs were mutated relative to wild type IL21 R ECD (SEQ ID NO:1).
- IL21 R ECD constructs as listed in Table 2 were transiently expressed as IL21 R-ECD-His6 constructs in HEK293 cells.
- HEK293F cells obtained from the [American Type Culture Collection (ATCC, Manassas,
- VA were cultured in freestyle 293 medium (Invitrogen, Grand Island, NY). Cells were grown and maintained in a humidified incubator with 7% C0 2 at 37°C. Conditioned media for the constructs were produced from a standard large-scale transient HEK293 transfection process.
- the conditioned media were filtered at 0.22 micron prior to purification.
- the secreted proteins from the conditioned media were analyzed by Western Blot as follows. Serum-free conditioned media samples were resolved by SDS PAGE on precast
- FIG. 3 and Figure 4 depict a Western blot analysis demonstrating that expression of modified IL21 R ECD constructs ECD1 , ECD2, ECD3, and ECD4 was greater than expression of wild type IL21 R ECD.
- each of the modified constructs has improved expression in the HEK293 cells as compared to the wild type protein.
- Conditioned media from transient HEK293 or CHO stable cultures were typically diafiltered against PBS pH 7.2 before loading onto an appropriately sized Nickel NTA Superflow (Qiagen) chromatography column equilibrated in 300mM NaCI, 50mM NaHPO pH 8. The column was washed with 10CV of 300mM NaCI, 50mM NaHPO, 15mM Imidazole pH 8 before being resolved by a 5CV linear gradient to 300mM NaCI, 50mM NaHPO, 250mM Imidazole pH8. The protein peak pool eluted from the Nickel NTA step was loaded on a Superdex 200 size exclusion chromatography column (GE Healthcare) equilibrated and developed in PBS. Finally, the SEC Fractions with high purity were pooled, concentrated and frozen for long term storage at -80°C. Biophysical characteristics of the purified modified IL21 R ECD constructs were further investigated as discussed below.
- Thermal denaturation experiments were performed on a Capillary-DSC instrument (MicroCal, Northampton, MA). Protein samples were dialyzed into PBS buffer, pH 7.2 at 4°C, normalized at a concentration of 1 mg/ml and extensively degassed before analysis. They were scanned from 20 to 90°C, at a scan rate of 1 °C min "1 against the dialysis buffer contained in the reference cell. The thermograms generated with the protein samples were analyzed using Origin 7.0 software (MicroCal Software). Data were converted to excess heat capacity, reference trace-subtracted, normalized for concentration and analyzed for Tm determination.
- Figure 5 depicts a differential scanning calorimetry thermogram demonstrating thermal stability enhancement of modified IL21 R ECD constructs ECD1 , ECD2, and ECD4 as compared to wild type IL21 R ECD.
- the T m for the mutated monomeric IL21 R ECDs was increased by about 6-8°C, clearly demonstrating a positive stabilization effect of the mutations over the wild type sequence.
- the formatting of IL21 R ECD1 as an Fc fusion preserved the thermal stability gain by 4°C (data not shown).
- T m for the mutated monomeric IL21 R ECD constructs was increased over the wild type protein demonstrating that the substitution of W148 and flanking residues at positions 147 and 149 (PWA, respectively) with GSG conferred greater stability upon the modified IL21 R ECD1.
- IL21 R ECD constructs were analyzed for purity by analytical size exclusion chromatography on an Agilent 1200 system using a Superdex 200 10/300GL (GE Healthcare) chromatography column. Typically 20 to 100 microgram of sample was loaded through the autosampler and injected on the column developed in PBS, pH6.8 at a flow rate of 0.75 ml/min.
- Figure 6 depicts an analytical size exclusion chromatograph that demonstrates reduced aggregations levels for modified IL21 R construct ECD1 as compared to wild type IL21 R following Nickel NTA initial affinity capture purification. Molecules were analyzed by analytical SEC immediately after the IMAC capture and imidazole elution. The IL21 R wild type molecule presents approximately 30% high molecular weight (HMW) species eluting ahead of the major peak of interest visible at 20 minute. In identical conditions, the ECD1 construct has a significantly decreased amount of HMW species, down to 10%, approximately 20% less high molecular weight aggregates than its wild type version. This not only improved the overall production process yields but also facilitated the final formulation and increased the stability of the product.
- HMW high molecular weight
- Figure 7 depicts results from an analytical size exclusion chromatograph that demonstrates reduced aggregations levels in modified IL21 R constructs using ECD1 , ECD2, and ECD4 compared with wild type.
- Each construct is purified to homogeneity and freed of high molecular weight species by Nickel affinity capture and size exclusion chromatography.
- the wild type, ECD1 and ECD2 molecules were incubated overnight in PBS buffer at the two different temperatures of 4°C and 40°C. Immediately following this incubation, the protein samples were analyzed by SEC. The 4°C samples were all consistent with the quality of the starting material with no visible HMW species detectable by SEC. Conversely, the 40°C samples presented a differential profile between the WT and ECD1/ECD2.
- the high temperature incubation originated aggregation only for the wild type sample representing up to 16% of the total integrated OD280nm signal.
- Both modified IL21 R constructs ECD1 and ECD2 demonstrated resistance to aggregation properties compared to wild type IL21 R in temperature stress condition.
- FIG. 8 shows a structural representation of the modified IL21 RFc fusion proteins.
- the modified linker sequence L1 allowed for full engagement of IL21 , with one IL21 RFc homodimeric molecule able to trap two free IL21 cytokine molecules.
- the results of a BIAcore study depicted in Figure 10 demonstrate full 2: 1 stoichiometry of binding of I L21 to lL21 RFc. Briefly, I L21 R Fc is coated on the BIAcore chip for a theoretical Rmax of 50.83 RU. IL21 cytokine is titrated progressively to saturation and the Rmax is recorded at 49.5RU. This observed value represents 97.4% of a full stoichiometry of two cytokine bound per IL21 RFc homodimer on the chip.
- Linker L1 modifications were made to Linker L1 to eliminate O-linked glycosylation by switching S-G residues at the ends of the linkers, as illustrated in Figure 11.
- the resulting linker is designated Linker L2, as shown in Table 3.
- Linker L1 sequence supported the post- translational modification of two serines by O-linked glycosylation.
- the heterogeneity of the species was accentuated by the partial nature of O-linked glycan occupancy and its variable size.
- the first and last serines carry the O-linked modification.
- IL21 RFc fusion proteins As one of the desired properties of IL21 RFc fusion proteins is to trap and clear plasma and tissue circulating IL21 cytokine, native Fc functions such as antibody-dependent cell- mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) are generally not preferred.
- ADCC antibody-dependent cell- mediated cytotoxicity
- CDC complement-dependent cytotoxicity
- effector functions were engineered out of human lgG1 Fc by mutating the positions Leu242, Leu243 and Gly245 to alanines (234, 235 and 237, respectively, per Kabat antibody numbering (see Table 4). These mutations from LLGL to AAGA are expected to reduce Fc effector functions including Fc mediated cell depletion.
- Increased serum residence time was also engineered in by mutating two residues at the interface between the CH2 and CH3 in the Fc domain. That is, the mutations M436L and N442S (428 and 434 respective to Eu antibody Kabat numbering (see Table 4) have been shown to increase the binding affinity to FcRn and the in-vivo half life as described in International Patent Publication No. WO 2009/086320.
- Example 1 1 BIAcore studies demonstrated that these mutations applied to modified IL21 RFc fusion proteins disclosed herein, such as ECD1-L1-FC1 , increased by a ten-fold factor the affinity to human FcRn as directly compared to its Fc wild type homologue molecule.
- the actual in-vivo NHP PK profile benefitted significantly from these two mutations, doubling the drug exposure and affecting positively both the terminal half life, which was increased three-fold, as well as the clearance rate, which was reduced to one half, compared with the wild type protein WTECD-L1-FC1.
- modified IL21 RFc fusion proteins were designed, and several are listed in Table 5. As will be apparent to persons skilled in the art, many other combinations of modified IL21 RFc fusion proteins may be generated by combining any of the modified IL21 R ECDs disclosed above, with any one of Linker 1 or Linker 2, or any other appropriate linker, with any of the human lgG1 Fc proteins disclosed above, or any other appropriate Fc proteins.
- Example 8 Expression and Purification of Modified IL21 RFc Fusion Proteins
- Figure 12 depicts expression of the modified IL21 RFc constructs ECD1-L1-FC1 , ECD1- L1-FC2, ECD-L2-FC1 , and ECD7-L2-FC1 as listed in Table 5 as analyzed using Western blotting.
- the culture medium supernatant was run by non-reducing SDS gel electrophoresis.
- the NCM were developed with a horseradish peroxidase-conjugated goat anti-human IgG (Sigma, cat. #: A0170) antibody. Each lane was loaded with an equal cell count.
- ECD1-L1-FC1 and ECD1-L1-FC2 demonstrated identical banding patterns by Western blot and the main migration band of the non-reduced IL-21 RFc is indicated by the arrow and which presented marked smearing immediately above it. This distinct smearing pattern is likely associated with the heterogeneous O-linked glycosylation post-translational modification of the L1 linker sequence.
- the banding pattern of ECD1-L2-FC1 showed a marked decrease in this smearing
- ECD7-L2-FC1 presents even further improved and cleaner banding pattern with no high molecular weight smearing above the IL-21 RFc band.
- This demonstrates the additional beneficial effect on aggregation obtained through the ECD7 engineering which comprises the substitution of GSG at amino acid residues 147, 148 and 149, respectively, and the additional substitution of D122A.
- conditioned media from transient or stable CHO cultures were typically loaded neat onto an appropriately sized protein A chromatography column equilibrated in PBS pH 7.2 (rmpProteinA Sepharose Fast Flow, GE Healthcare).
- the column was then washed with 5 column volumes (CV) of PBS before being resolved by a 10 CV linear gradient to 20 mM citric acid 150 mM NaCI pH2.5.
- the protein A peak pool was diluted 1 to 3 with 20mM Tris pH8.6 for binding to source 15Q column (GE Healthcare) and eluted with a 15 CV linear gradient to 20mM Tris 1 M NaCI pH8.6.
- the Source 15Q peak containing the IL21 R.Fc was then pooled and applied over a Superdex200 size exclusion chromatography column (GE Healthcare) equilibrated and developed in PBS. The final fractions were 0.2 micron filtered and concentrated to variable concentrations before freezing for long term storage at -80°C. Proteins were further characterized as described below.
- Modified IL21 RFc fusion protein construct ECD1-L1-FC1 was analyzed for purity by SDS PAGE as described above, and by isoelectric focusing (IEF) to confirm pi. IEF gel
- IL21 R ECD has five occupied N-linked glycosylation sites creating heterogeneity in the molecular charge as illustrated by the diffuse band migrating around the 4.2 pi marker of the IEF gel.
- the SDS PAGE demonstrates a high purity of material with no other band detected in the reduced condition.
- the non-reduced condition shows predominantly the disulfide bridged covalent IL21 RFc homodimer fusion protein migrating as expected around 140 kD, as well as a minor component of non-covalently bound equivalent to its reduced monomeric form migrating at about 70 kD.
- DSC was performed as described above on modified IL21 RFc fusion proteins ECD7-L1- FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1-FC1 , and ECD1 1-L1-FC1 as listed in Table 5, and compared to ECD1-L1-FC1 fusion protein.
- the DSC thermogram is depicted in Figure 14.
- Table 7 is a summary of the Tm temperatures of the various constructs from the thermogram in Figure 14.
- BIAcore analysis was performed to determine the steady-state affinity (KD) for different IL21 RFc engineered molecules for binding human FcRn.
- BIAcore technology utilizes changes in the refractive index at the surface layer of a sensor upon binding of the IL21 RFc molecules to human FcRn protein immobilized on the layer. Binding is detected by surface plasmon resonance (SPR) of laser light refracting from the surface.
- SPR surface plasmon resonance
- Human FcRn was specifically biotinylated through an engineered Avi-tag using the BirA reagent (Catalog #: BIRA500, Avidity, LLC, Aurora, Colorado) and immobilized onto a streptavidin (SA) sensor chip to enable uniform orientation of the FcRn protein on the sensor.
- the BIAcore results shown in Figure 15 illustrate the binding kinetic effect of engineering FcRn binding enhancement in the Fc domain.
- the FC1 containing IL21 RFc fusion protein which contains mutations in the Fc component to both enhance FcRn binding and in vivo half-life, demonstrated a 10-fold improved steady-state KD to FcRn over the non engineered FC3 containing molecule.
- test article was captured by recombinant human IL-21-FLAG
- FIG. 16 depicts PK profiles of non-human primates injected with modified IL21 RFc fusion protein constructs ECD1-L1-FC1 , ECD1-L1-FC3 and wild type IL21 RFc.
- Table 8 is a summary of the PK profiles depicted in Figure 16.
- Table 8 Summary of PK Profiles of Various IL21 RFc Constructs
- the wild type ECD formatted as Fc fusion linked by a linker comprising four glycine and a serine demonstrated a poor PK profile and low drug exposure presumed to result from the low bioavailability, low average terminal half-life as well as the unexpectedly high clearance rate.
- the first pass of construct engineering introducing both ECD1 and linker L1 optimization, brought the PK profile back to numbers expected for typical Fc fusion molecules with a clearance rate below 1 ml/hr/kg.
- the pharmacokinetic properties were then further improved by two-fold overall exposure gain through the introduction of the FC1 engineering with FcRn binding enhancement mutations as discussed in Example 6 and 1 1 above.
- ECD1-L1-FC1 demonstrated increased bioavailability at 83.6%.
- Example 13 Human Primary B Cell Proliferation Assays Using Modified IL21 RFc Fusion Constructs
- Buffy coat cells from healthy human donors were incubated with a B cell enrichment cocktail (RosetteSep; StemCell Technologies, Vancouver, British Columbia, Canada), and isolated by the negative selection method described in the manufacturer's instructions. The purity of the isolated B cells was measured by flow cytometry after staining with anti-CD19. The enriched cells were approximately 60 - 80 % CD19+ B cells.
- Enriched human B cells were cultured in 0.2 mL RPMI containing 10% FBS, 50 U/mL penicillin, 50 ug/mL streptomycin, and 2 mM L-glutamine at 1 X 10 5 /well in 96-well flat bottom plates. B cells were incubated with serially diluted anti-human IL21 receptor antibodies, 1 ug/mL anti-CD40 mAb (BD Bisosciences) and 20 ng/mL IL21 cytokine for 3 days in a 37C° incubator adjusted to 5% C0 2 .
- FIG. 17 is a graph depicting the results of a primary IL21 dependent B cell proliferation assay using modified IL21 RFc construct ECD1-L1-FC1 and wild type IL21 RFc positive control WTECD-L1-FC3.
- the human lgG1 control demonstrated no effect on B cell proliferation.
- Both mutant and wild type IL21 RFc proteins showed equal potency in this assay with an IC50 around 10nM demonstrating that the GSG mutations of the ECD1 do not affect the biological function of the IL21 receptor in recognizing the cytokine.
- Human CD4+ T cells were isolated from buffy coats with a ROSETTESEPTM CD4+ T cell enrichment cocktail (Stem Cell Technologies, Vancouver, BC, Canada) according to the manufacturer's instructions. Cells (80-90% CD4+/CD3+ T cells) were activated for 3 days with anti-CD3/anti-CD28-coated microspheres (prepared in house) in RPMI containing 10% FBS, 100 U/ml penicillin, 100 g/ml streptomycin, 2 mM L-glutamine, and HEPES (37°C, 5% C0 2 ). Microspheres were then removed, and the cells were washed once prior to resting overnight at ⁇ 1 x 10 6 cells/ml in culture medium and once before addition to assay plates.
- ROSETTESEPTM CD4+ T cell enrichment cocktail Stem Cell Technologies, Vancouver, BC, Canada
- Test articles were diluted in culture medium in flat-bottomed 96-well plates, followed by sequential addition of human IL-21 (25 ng/ml final concentration) and 10 5 cells/well. After 72 hr, cells were pulsed with 1 pCi/well 3H-thymidine (Perkin Elmer (NEN)) for 6 hr and harvested onto glass fiber filter mats for liquid scintillation counting.
- human IL-21 25 ng/ml final concentration
- Figure 18 shows a graph depicting the results of an IL21 dependent primary T cell proliferation assay using modified IL21 RFc constructs ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1- FC1 , ECD10-L1-FC1 , and ECD1 1-L1-FC1.
- Table 9 is a summary of the IC50 of each of the constructs. Table 9: IC50 of Various IL21 RFc Constructs in Primary T-Cell Assay
- a concentration of 25ng/ml of IL21 cytokine maintained proliferation of the T cells in this assay.
- all 5 mutants showed good potency in neutralization of T cell proliferation.
- the differences in IC50 ranged from 19.6 nM to 28.3 nM and the differences are believed not to be significant.
- Example 15 Characterization of Modified IL21 RFc Constructs with Fc Variant T445K
- ECD1-L1-FC1 and ECD1-L1-FC2 were characterized and compared in various side by side experiments. Both proteins expressed to comparable levels and purified equally well following the same protocol. Analytical characterization by SDS PAGE, IEF, size exclusion, ion exchange and hydrophobic interaction chromatographies demonstrated that both ECD1-L1-FC1 and ECD1-L1-FC2 molecules were similar. The differential scanning calorimetry result highlighted minor destabilization of the Fc CH2 and CH3 domains of ECD1- L1-FC2, but this was considered within the norm of variation observed with molecules containing the same human lgG1 Fc fragment. In addition, no significant differences in activity in functional assays for IL21 neutralization, IL21 binding, and FcRn binding, were observed between ECD1-L1-FC1 and ECD1-L1-FC2.
- Modified IL21 RFc construct ECD1-L1-FC1 was formulated as a powder for injection in a
- ECD1-L1-FC1 Active Ingredient 140 100 mg/mL
Abstract
Disclosed herein are modified interleukin 21 receptor extracellular domain (IL21R ECD) proteins comprising a modification selected from: one or more amino acid substitutions at or within 4 amino acid residues of the tryptophan at position 148of the amino acid sequence of SEQ ID NO:1, an insertion of one or more amino acids at position 148of the amino acid sequence of SEQ ID NO:1, or deletion of the tryptophan at position 148of the amino acid sequence of SEQ ID NO:1. Additional substitutions are also contemplated. Also disclosed are fusion proteins comprising the modified IL21R ECD proteins, pharmaceutical compositions comprising the modified IL21R ECD proteins and fusion proteins, and methods of treatment and uses relating to the modified IL21R ECD proteins and fusion proteins.
Description
MODIFIED INTERLEUKIN 21 RECEPTOR PROTEINS
FIELD OF THE INVENTION
The present invention relates to novel modified IL21 R ECD proteins and fusion proteins thereof having, among other properties, improved stability, activity, and increased expression. The invention further relates to use of these proteins to treat diseases or disorders mediated by interaction of IL21 with IL21 R.
BACKGROUND OF THE INVENTION
Human interleukin 21 (IL21) is a cytokine that shares sequence homology with IL2, IL4 and IL15 (Parrish-Novak et al. (2000) Nature 408:57-63). Human IL21 receptor (IL21 R) is a class I cytokine receptor that is expressed in lymphoid tissues, particularly on T cells, B cells, natural killer (NK) cells, dendritic cells (DC) and macrophages (Parrish-Novak et al. (2000) supra), which allows these cells to respond to IL21 (Leonard and Spolski (2005) Nat. Rev. Immunol. 5:688-98). The widespread lymphoid distribution of IL21 R indicates that IL21 plays an important role in immune regulation. In vitro studies have shown that IL21 significantly modulates the function of B cells, CD4+ and CD8+ T cells, and NK cells (Parrish-Novak et al. (2000) supra; Kasaian et al. (2002) Immunity 16:559-69). Recent evidence suggests that IL21 - mediated signaling can have antitumor activity (Sivakumar et al. (2004) Immunology 1 12: 177- 82), and that IL21 can prevent antigen-induced asthma in mice (Shang et al. (2006) Cell.
Immunol. 241 :66-74).
In autoimmunity, disruption of the IL21 gene and injection of recombinant IL21 have been shown to modulate the progression of experimental autoimmune myasthenia gravis (EAMG) and experimental autoimmune encephalomyelitis (EAE), respectively (King et al. (2004) Cell 1 17:265-77; Ozaki et al. (2004) Immunol. 173:5361-71 ; Vollmer et al. (2005) Immunol. 174:2696-2701 ; Liu et al. (2006) Immunol. 176:5247-54)). In these experimental systems, it has been suggested that the manipulation of IL21-mediated signaling directly altered the function of CD8+ cells, B cells, T helper cells, and NK cells. Animal studies using surrogate murine IL21 RFc fusion protein in a lupus-prone mouse model have demonstrated a pathogenic role of IL21 in the disease model and reduction in disease progression by administering the surrogate fusion protein. (Herber et al. (201 1) J. Immunol. 178:3822-3830).
The amino acid sequences of IL21 and IL21 R, and the nucleotide sequences encoding them, are described in WO00/53761 , WO01/85792, and WO2003/028630. IL21 and IL21 R
antagonists, including antibodies and fusion proteins, are also described in these applications. Despite these previous advances, there remains a long-felt need for improved IL21 R proteins, including IL21 R fusion proteins with improved therapeutic characteristics. The present invention meets this need.
SUMMARY OF THE INVENTION
This application discloses modified IL21 R extracellular domain (ECD) proteins or polypeptides, as well as associated reagents, compositions and methods.
Embodiment 1 (E1). According to a first aspect of the invention, there is provided a modified IL21 R ECD comprising a modification selected from the group consisting of: one or more amino acid substitutions at or within four amino acids residues of position 148 of the amino acid sequence set forth as SEQ ID NO:1 ; one or more amino acid insertions at amino acid position 148 of the amino acid sequence set forth as SEQ ID NO:1 ; and deletion of the tryptophan residue at position 148 (W148) of the amino acid sequence set forth as SEQ ID NO:1.
Described below are a number of embodiments (E) of this first aspect of the invention where, for convenience, E1 is identical thereto.
E.2. The modified IL21 R ECD of E1 , wherein the one or more substituted or inserted amino acid is a naturally-occurring amino acid.
E3. The modified IL21 R ECD of E2, wherein the substituted or inserted amino acid is selected from the group consisting of glycine, alanine, leucine, methionine, phenylalanine, lysine, glutamine, glutamic acid, serine, proline, valine, isoleucine, cysteine, tyrosine, histidine, arginine, asparagine, aspartic acid, and threonine.
E4. The modified IL21 R ECD of E1 , wherein the substituted amino acid at position 148 is
selected from the group consisting of serine or asparagine.
E5. The modified IL21 R ECD of E1 further comprising at least one amino acid substitution at amino acid position 147, 149, and 150 of the amino acid sequence set forth as SEQ ID NO: 1.
E6. The modified IL21 R ECD of E5, wherein the amino acid substitution is selected from the group consisting of:
a. Glycine at position 147, serine at position 148, and glycine at position 149 (SEQ ID
NO:2);
b. Asparagine at position 148 and serine at position 150 (SEQ ID NO: 3);
c. Asparagine at position 148, glycine at position 149, and serine at position 150 (SEQ ID NO:4); and
d. Serine at position 148 and glycine at position 149 (SEQ ID NO:7).
E7. The modified IL21 R ECD of E1 comprising serine at position 148 (SEQ ID NO:6).
E8. The modified IL21 R ECD of E5 comprising asparagine at position 148, serine at position
150, and further comprising asparagine at position 49 (SEQ ID NO:5).
E9. The modified IL21 R ECD of claim 5 comprising glycine at position 147, serine at position
148, glycine at position 149, and further comprising an amino acid substitution at position 122 all relative to the amino acid sequence set forth as SEQ ID NO:1.
E10. The modified IL21 R ECD of E9, comprising alanine at position 122 (SEQ ID NO:8). E1 1. The modified IL21 R ECD of E9, comprising isoleucine at position 122 (SEQ ID NO:9). E12. The modified IL21 R ECD of E9, comprising tryptophan at position 122 (SEQ ID NO: 10). E13. The modified IL21 R ECD of E9, comprising phenylalanine at position 122 (SEQ ID
NO:1 1).
E14. The modified IL21 R ECD of E9, comprising tyrosine at position 122 (SEQ ID NO:12). E15. A fusion protein comprising a modified IL21 R ECD according to any one of E1-E14, fused to a heterologous amino acid sequence.
E16. The fusion protein of E15, wherein the IL21 R ECD is fused to the heterologous amino acid sequence via a linker.
E17. The fusion protein of E16, wherein the linker is a peptidyl linker comprising a sequence selected from: GSGEGEGSEGSG (SEQ ID NO:13); GGSEGEGSEGGS (SEQ ID
NO: 14); and GGGGS (SEQ ID NO:15).
E18. The fusion protein of E17, wherein the linker comprises the amino acid sequence
GSGEGEGSEGSG (SEQ ID NO:13).
E19. The fusion protein of E17, wherein the linker comprises the amino acid sequence
GGSEGEGSEGGS (SEQ ID NO:14).
E20. The fusion protein of E15, wherein the heterologous amino acid sequence comprises a human lgG1 Fc domain.
E21. The fusion protein of E20, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:16.
E22. The fusion protein of E20, wherein the Fc domain is modified to alter effector function of the Fc domain.
E23. The fusion protein of E22, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:19.
E24. The fusion protein of E20, wherein the Fc domain is modified to enhance the half-life of the fusion protein.
E25. The fusion protein of E24, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO: 17.
E26. The fusion protein of E24, wherein the Fc domain comprises the amino acid sequence set forth as SEQ ID NO:18.
E27. The fusion protein of E15 comprising an amino acid sequence selected from the group consisting of the sequence set forth as SEQ ID NO:20, SEQ ID NO:21 , SEQ ID NO:22, SEQ ID NO:23, SEQ ID NO:24, SEQ ID NO:25, SEQ ID NO:26, SEQ ID NO:27, SEQ ID NO:28 and SEQ ID NO:29.
E28. A fusion protein comprising the amino acid sequence of SEQ ID NO:20.
E29. A pharmaceutical composition comprising the modified IL21 R ECD of any one of El- El 4, or the fusion protein of any one of E15-E28, and a pharmaceutically acceptable agent.
E30. The pharmaceutical composition according to E29, wherein the composition comprises 0.5 - 200 mg/mL of the modified IL21 R ECD of any one of E1-E17 or the fusion protein of any one of E18-E31 , 10-50 mM tromethamine, 0.01 - 0.10 mg/mL EDTA, 20 - 120 mg/mL sucrose, and 0.01 - 0.4 mg/mL polysorbate 80.
E31. The pharmaceutical composition according to E30, wherein the composition comprises 100 mg/ml of the fusion protein comprising the amino acid sequence set forth as SEQ ID NO:20, and further comprises 40mM tromethamine Tris, 0.05 mg/mL EDTA, 100mg/ml sucrose, and 0.2 mg/ml polysorbate 80.
E32. An isolated nucleic acid encoding the modified IL21 R ECD of any one of E1-E14, or the fusion protein of any one of E15-E28.
E33. An isolated nucleic acid comprising the nucleic acid sequence of SEQ ID NO:31.
E34. A vector comprising the nucleic acid of E33.
E35. A host cell comprising the nucleic acid of claim 33.
E36. A host cell comprising the vector of claim 34.
E37. A method of producing a modified IL21 R protein comprising growing the host cell of E34-
E35 under conditions where the protein encoded by the nucleic acid is expressed.
E38. The method of E36, further comprising isolating the protein.
E39. A modified IL21 RFc fusion protein encoded by the nucleic acid insert comprised in the vector having ATCC Accession No. PTA-120480.
E40. A modified IL21 R-Fc fusion protein comprising the amino acid sequence encoded by the nucleic acid insert comprised by the vector deposited as ATCC Accession No. PTA- 120480.
E41. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:20.
E42. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:21.
E43. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:22.
E44. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:23.
E45. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:24.
E46. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:25.
E47. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:26.
E48. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:27.
E49. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:28.
E50. A modified IL21 R Fc fusion protein comprising an amino acid sequence consisting of the amino acid sequence of SEQ ID NO:29.
E51. A method of treating a disease or disorder mediated by interaction of IL21 with IL21 R comprising administering to a patient in need thereof, the IL21 R ECD protein of any one of claims E1-E14, the pharmaceutical composition of any one of claims E29-E31 , or the fusion protein of any one of claims E15-E28 or E39-E50.
E52. The method of E51 , wherein the disease or disorder is an inflammatory or autoimmune disease or disorder.
E53. The method of 51 , wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic
lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
scleroderma, vasculitis, and Behcet's Disease.
E54. Use of the modified IL21 R ECD of any one of E1-E14, or the fusion protein of any one of claims E15-E28 or E39-E50, in the manufacture of a medicament for treatment of a disease or disorder mediated by interaction of IL21 with IL21 R.
E55. The use of E54, wherein the disease or disorder is an inflammatory or autoimmune
disease or disorder.
E56. The use of E54, wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
scleroderma, vasculitis, and Behcet's Disease.
E57. Use of the IL21 R ECD protein of any one of E1-E14, the pharmaceutical composition of any one of claims E29-E31 , or the fusion protein of any one of claims E15-E28 or E39-
E50, for treatment of a disease or disorder mediated by interaction of IL21 with IL21 R in a patient in need thereof
E58. The use of E57, wherein the disease or disorder is an inflammatory or autoimmune
disease or disorder
E59. The use of E57, wherein the disease or disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis,
spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma,
scleroderma, vasculitis, and Behcet's Disease.
E60. The IL21 R ECD protein of any one of claims E1-E14, the pharmaceutical composition of any one of E29-E31 , or the fusion protein of any one of E15-E28 or E39-E50, for use in treating a disease or disorder mediated by interaction of IL21 with IL21 R.
E61. The pharmaceutical composition of E60, wherein the disease or disorder is an
inflammatory or autoimmune disorder.
E62. The pharmaceutical composition of E60, wherein the inflammatory disorder is selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis, spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma, scleroderma, vasculitis, and Behcet's Disease.
BRIEF DESCRIPTION OF THE DRAWINGS
The foregoing summary, as well as the following detailed description of the invention, will be better understood when read in conjunction with the appended drawings. For the purpose of illustrating the invention there are shown in the drawings embodiment(s) which are presently preferred. It should be understood, however, that the invention is not limited to the precise arrangements and instrumentalities shown.
In the drawings:
Figure 1 depicts a diagram showing the X-ray crystal structure of two chains of IL21 R ECD at 2.8 Angstrom resolution, represented in ribbon diagram in grey shading (chain A) and black (chain B). The respective IL21 cytokines complexed to the receptor chains are
represented by surface density models. The inset provides a close-up view of a flexible loop of the C-terminal domain of the receptor. The tryptophan (Trp or W) at position 148 of each chain is represented using sticks and the two tryptophans are shown interlocked through a pi stacking interaction which, without being limited to any particular theory, helps to stabilize the homodimer of the IL21 receptor.
Figure 2 depicts a graph illustrating data from a differential scanning calorimetry thermogram demonstrating enhanced thermal stability of modified IL21 R ECD produced by substituting aspartate (Asp or D) at position 122 (D122) with para-acetyl phenylalanine (paF), relative to other positions within the IL21 R ECD.
Figure 3 depicts an image of a Western blot showing various transiently expressed modified IL21 R ECD proteins comprising a His tag from HEK 293 cells. The cells were transiently transfected with each construct designated at the bottom of the gel and were cultured for five days before supernatant harvest. A western blot analysis was performed on the culture medium of the various mutants. More specifically, following transfer, the nitrocellulose membranes were probed with mouse anti-Histidine IgG (Qiagen, cat#: 34670) as primary antibody followed by horseradish peroxidase-conjugated rabbit anti-mouse IgG (Pierce, cat#: 32430) as secondary antibody. Each lane is loaded with an aliquot of conditioned medium obtained from a sample comprising an equal number of cells (hereinafter "equal cell count"). The wild type ECD expression is directly compared to three different IL21 R ECD variants ECD1 , ECD2 and ECD3, which comprise mutations at and surrounding Trp 148 (see Table 1 below). This western blot analysis shows the enhanced expression level of modified IL21 R ECD proteins relative to wild type IL21 R ECD protein.
Figure 4 is an image of a Western blot showing expression of various modified IL21 R ECD constructs from transiently transfected HEK 293 cells. Experimental methods were as described for Figure 3, supra.
Figure 5 shows a graph depicting a differential scanning calorimetry thermogram demonstrating the thermal stability enhancement of modified IL21 R ECD constructs ECD1 , ECD2, and ECD4 as compared with wild type IL21 R ECD (WTECD).
Figure 6 depicts a graph showing results from an analytical size exclusion
chromatograph demonstrating reduced aggregation level of modified IL21 R construct ECD1 compared with wild type ECD following Nickel NTA initial affinity capture purification.
Figure 7, comprising panels A and B, depicts a graph showing the results of an analytical size exclusion chromatograph demonstrating enhanced resistance to aggregation of modified IL21 R constructs ECD1 and ECD2 compared with wild type ECD. The chromatograms were collected after subjecting the protein samples to an overnight incubation at 4°C (panel 7A showing no aggregation for any of the proteins) then subjecting the samples to a stress temperature of 40°C (panel 7B). After 40°C incubation overnight, the peak demonstrating 16% protein aggregation for wild type ECD is not present in the chromatograms for ECD1 or ECD2.
Figure 8 is a diagram depicting a structural representation of an IL21 RFc fusion protein as disclosed herein. One IL21 R-Fc fusion protein is shown in black and the other IL21 R-Fc is shown in light gray. The two fusion proteins are shown as a homodimer stabilized by two disulfide bridges formed by two cysteines in each hinge region of the Fc of each fusion peptide.
Figure 9, comprising panels A through D, shows the results of an isothermal titration calorimetry (ITC) experiment comparing IL21 cytokine being titrated to a monomer version of IL21 R (ECD1) and to a homodimer ECD1 fusion construct comprising a linker and an Fc domain (ECD1-L3-FC3). Figure 9A presents the experimental thermogram of IL-21 titration to the monomer ECD1. Figure 9B shows the fitted curve of the data in 9A for molar ratio (N) determination. Figure 9C presents the experimental thermogram of IL-21 titration to the homodimer ECD1 Fc fusion construct ECD1-L3-FC3. Figure 9D shows the fitted curve of the data in 9C for molar ratio (N) determination. The resulting molar ratio of N=0.829 for the monomer ECD1 (panel 9B) reaches approximately the expected binding stoichiometry of N=1 for one cytokine binding one receptor. In the case of the Fc fusion molecule, comprising the linker L3, the measured molar ratio N=0.888 (panel 9D) stays unexpectedly at the same approximate level of N=1. An Fc fusion molecule contains two receptors and a result closer to N=2 was anticipated.
Figure 10, comprising panels A and B, depicts the results of a BIAcore IL21 titration study demonstrating that linker L1 design allows the engagement of two IL21 cytokine per
IL21 RFc fusion protein homodimer. Figure 10A depicts an experimental sensorgram showing saturation of the Response signal as IL-21 is titrated in to higher concentrations. ECD1-L1-FC1 is coated on the BIAcore chip for a theoretical Rmax response of 50.83RU. IL-21 cytokine is titrated progressively to saturation and the Rmax is recorded at 49.5RU as seen in the fitted curve of the Figure 10B. This observed value represents 97.4% of a full stoichiometry of two cytokine bound per IL-21 RFc homodimer on the chip.
Figure 11 is an illustration showing the modifications of Linker L2 (bottom sequence: GGSEGEGSEGGS) from the amino acid sequence of Linker L1 (top sequence:
GSGEGEGSEGSG).
Figure 12 is an image of a Western blot showing the results of transient expression of various modified IL21 RFc constructs ECD1-L1-FC1 , ECD1-L1-FC2, ECD7-L2-FC1 , and ECD1- L2-FC1 in cells. Following protein transfer from the gel to a nitrocellulose membrane (NCM), the NCM were developed with a horseradish peroxidase-conjugated goat anti-human IgG (Sigma, cat#: A0170) antibody to detect human Fc. Each sample identified at the bottom of the gel was loaded in duplicate and each lane was loaded with an aliquot of conditioned medium normalized to equal number of cells. The far right lane contains a molecular weight standard with weights in kD. The migration position of the I L21 Fc fusion constructs is at approximately 150kD. Immediately above the position of the fusion constructs is a product-related smear of higher molecular weight species. The smear is created by heterogeneous O-linked glycosylated
species. These post translational modifications have been characterized and located on specific serine residues in the linker L1 sequence. Comparison of the lanes for ECD1-L1-FC1 to ECD1- L1-FC2 shows identical banding profiles for both molecules. This demonstrates that the R445K mutation in the FC from FC1 to FC2 did not modify the SDS-PAGE properties of the molecule. Comparison of the lanes for ECD1-L1-FC1 to ECD1-L2-FC1 shows a noticeable reduction of the smear species highlighted by the accolade. This demonstrates that the linker L2 design with 2 serine position permutations did not support as efficiently if at all the O-linked glycosylation of the linker. Comparison of the lanes for ECD1-L2-FC1 to ECD7-L2-FC1 shows a very distinct improvement of the banding pattern for the ECD7 construct. Virtually all high molecular weight smears above the 150kD band have been removed. This demonstrates that the D122 mutation contained in the ECD7 design, through its stabilization effect and in conjunction with the L2 linker design, allows the production of a much more homogeneous molecule with reduced high molecular weight species.
Figure 13, comprising panels A and B, shows an image of an isoelectric focusing (IEF) gel (Figure 13A) and an image of a SDS PAGE gel (Figure 13B) of modified IL21 RFc fusion construct ECD1-L1-FC1 to demonstrate the isoelectric point (pi) The empirical pi by IEF gel is visualized by a tight group of bands around the 4.2 pi standard. The sequence calculated pi for this construct is approximately 5.44. The difference is attributed to 6 N-linked glycosylation sites occupied and capped with sialic acids rendering the molecule significantly more acidic than its amino acid sequence predicted. The SDS PAGE gel presents both non-reduced and reduced conditions and highlights the purity of the material tested as well as the disulfide driven dimer nature of the molecule than can be fully reduced to monomer with dithiothreitol (DTT) treatment in the denaturing condition of this gel.
Figure 14 shows a graph depicting the results of a differential scanning calorimetry thermogram demonstrating thermal stability enhancement of modified IL21 RFc fusion proteins ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1-FC1 , and ECD11-L1-FC1 compared to wild type IL21 RFc (ECD1). All constructs were identical in design comprising the same linker (L1) and Fc construct (FC1), varying only at the amino acid at position 122 of the IL21 R ECD component of the fusion protein. Wild type IL21 R ECD carried an Asp at position 122 (D122). ECD7, 8, 9, 10 and 1 1 carry at position 122, respectively, Ala, lie, Trp, Phe and Tyr. The CH2 and CH3 domain transition midpoints (Tm) are unmodified by these mutations. The wild type ECD (having D122) has a lower Tm at 50.22 degrees while the hydrophobic mutants for the 122 position overlap with an average gain of over 4 degrees to a higher Tm around approximately
54 degrees. This demonstrates the thermal stability enhancement brought by mutating D122 to a hydrophobic residue.
Figure 15 shows a diagram depicting the results of BIAcore studies of the binding of various modified IL21 RFc fusion proteins to FcRn. Solid lines are for ECD1-L1-FC3. This FC3 molecule carries the triple (Winter) mutation (3m) for reduced effector function (i.e., LLGL has been mutated to AAGA). The dashed lines represent ECD1-L1-FC1. This FC1 molecule is designed for extended in vivo half life and carries both the 3m mutations and the LS double mutant for enhanced binding to FcRn (see Table 4). The BIAcore experimental sensogram of this figure demonstrates the over 10 fold higher in vitro affinity of the FC1 construct vs. the FC3 construct to human FcRn (ECD1-L1-FC3 average KD (nM) = 21 14.5, sd 102.5 vs. ECD1-L1- FC1 average KD (nM) = 194.1 , sd 17.1).
Figure 16 shows a graph depicting the pharmacokinetic (PK) profiles of various modified IL21 RFc fusion constructs in non-human primate administered intravenously (IV) or
subcutaneously (SC) at a dose of 2 mg/kg. ECD1-L1-FC3 IV is shown by solid circles; ECD1- L1-FC1 IV is shown by solid squares; ECD1-L1-FC1 SC is shown by open squares; and WT ECD-L3-Fc3 IV is shown by open diamonds. Measuring the serum concentration of the molecules over time, this figure illustrates the progressive improvements of in vivo PK properties obtained with different IL21 RFc constructs. The comparison of WTECD-L3-FC3 to ECD1-L1- FC3 demonstrates the combined advantage of the ECD1 mutation at position 148 and L1 linker, over wild type ECD and Linker L3. This design allows for a 18-fold better clearance rate (CL) from 15.9 mL/hr/Kg to 0.882 mL/hr/Kg. The comparison of ECD1-L1-FC3 to ECD1-L1-FC1 also demonstrates the direct beneficial effect of the LS mutations contained in FC1. Without wishing to be bound by any particular theory, the enhancement in FcRn binding apparently mediates a 3.5-fold improved terminal half-life to 247 hr and reduced by half the clearance rate to 0.241 mL/hr/Kg. Additionally, the intravenous (IV) versus subcutaneous (SC) data for ECD1-L1-FC1 demonstrate the good bioavailability of this molecule at about 84%.
Figure 17 shows a graph depicting the results of a primary B cell proliferation assay and the ability of different IL21 RFc constructs to inhibit proliferation. The control human lgG1 molecule did not show neutralization activity in the range tested -.up to 50nM. Both ECD1-L1- FC1 and WTECD-L1-FC3 showed equal neutralization potency, slightly under 10nM. This demonstrates that the stabilizing mutations introduced in the design of ECD1 and the use of the LS mutations in FC1 and FC3 did not affect the in vitro biological function of the IL-21 receptor.
Figure 18 shows a graph depicting the results of an I L21 -dependent primary T cell assay using modified IL-21 RFc constructs ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1-
FC1 , and ECD1 1-L1-FC1. The cell proliferation was measured in counts per minute. The control human lgG1 in solid black line showed no neutralization activity in the range tested up to 1000nM. All other molecules, wild type ECD and D122 ECD mutants, showed equal
neutralization potency around 25nM. This demonstrates that the mutation of position D122 to a hydrophobic residue fully preserves biological function of the IL-21 receptor protein.
DETAILED DESCRIPTION OF THE INVENTION
This application discloses modified IL21 R ECD proteins with improved properties, including greater in vitro expression levels, improved stability, decreased aggregation, and increased half-life compared to wild type unmodified IL21 R proteins. The half-life of the modified IL21 R ECD proteins can be further improved by fusing heterologous proteins, such as an Fc region of an antibody, to the IL21 R ECD protein. The fusion proteins can be further modified by substituting amino acids of the Fc region of the fusion protein. Definitions and General Techniques
Unless otherwise defined herein, scientific and technical terms used in connection with the present invention shall have the meanings that are commonly understood by those of ordinary skill in the art. Further, unless otherwise required by context, singular terms shall include pluralities and plural terms shall include the singular. Generally, nomenclatures used in connection with, and techniques of, cell and tissue culture, molecular biology, immunology, microbiology, genetics and protein and nucleic acid chemistry and hybridization described herein are those well known and commonly used in the art.
The methods and techniques of the present invention are generally performed according to conventional methods well known in the art and as described in various general and more specific references that are cited and discussed throughout the present specification unless otherwise indicated. See, e.g., Sambrook et al., Molecular Cloning: A Laboratory Manual, 2d ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (1989) and Ausubel et al., Current Protocols in Molecular Biology, Greene Publishing Associates (1992), and Harlow and Lane Antibodies: A Laboratory Manual, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y. (1990), which are incorporated herein by reference. Enzymatic reactions and purification techniques are performed according to manufacturer's specifications, as commonly accomplished in the art or as described herein. The nomenclatures used in connection with, and the laboratory procedures and techniques of, analytical chemistry, synthetic organic chemistry, and medicinal and pharmaceutical chemistry described herein are those well known
and commonly used in the art. Standard techniques are used for chemical syntheses, chemical analyses, pharmaceutical preparation, formulation, and delivery, and treatment of patients.
The following terms, unless otherwise indicated, shall be understood to have the following meanings:
The term "polypeptide" encompasses native or artificial proteins, protein fragments and polypeptide analogs of a protein sequence. A polypeptide may be monomeric or polymeric.
The term "isolated protein", "isolated polypeptide" or "isolated antibody" is a protein, polypeptide or antibody that by virtue of its origin or source of derivation has one to four of the following: (1) is not associated with naturally associated components that accompany it in its native state, (2) is free of other proteins from the same species, (3) is expressed by a cell from a different species, or (4) does not occur in nature. Thus, a polypeptide that is chemically synthesized or synthesized in a cellular system different from the cell from which it naturally originates will be "isolated" from its naturally associated components. A protein may also be rendered substantially free of naturally associated components by isolation, using protein purification techniques well known in the art.
A protein or polypeptide is "substantially pure," "substantially homogeneous," or
"substantially purified" when at least about 60 to 75% of a sample exhibits a single species of polypeptide. The polypeptide or protein may be monomeric or multimeric. A substantially pure polypeptide or protein will typically comprise about 50%, 60%, 70%, 80% or 90% W/W of a protein sample, more usually about 95%, and preferably will be over 99% pure. Protein purity or homogeneity may be indicated by a number of means well known in the art, such as
polyacrylamide gel electrophoresis of a protein sample, followed by visualizing a single polypeptide band upon staining the gel with a stain well known in the art. For certain purposes, higher resolution may be provided by using HPLC or other means well known in the art for purification.
The term "polypeptide fragment" as used herein refers to a polypeptide that has an amino-terminal and/or carboxy-terminal deletion, but where the remaining amino acid sequence is identical to the corresponding positions in the full-length naturally-occurring sequence. Also, fragments according to the invention may be made by truncation, e.g., by removal of one or more amino acids from the N and/or C-terminal ends of a polypeptide. Up to 10, up to 20, up to 30, up to 40 or more amino acids may be removed from the N and/or C terminal in this way. Fragments may also be generated by one or more internal deletions. In some embodiments, fragments are at least 5, 6, 8 or 10 amino acids long. In other embodiments, the fragments are at least 14, at least 20, at least 50, or at least 70, 80, 90, 100, 150 or 200 amino acids long.
In certain embodiments, amino acid substitutions of a protein or portion thereof are those which: (1) reduce susceptibility to proteolysis, (2) reduce susceptibility to oxidation, (3) alter binding affinity for forming protein complexes, or (4) confer or modify other physicochemical or functional properties. For example, single or multiple amino acid substitutions (preferably conservative amino acid substitutions) may be made in the normally-occurring sequence.
A conservative amino acid substitution should not substantially change the structural characteristics of the parent sequence. Examples of art-recognized polypeptide secondary and tertiary structures are described in Proteins, Structures and Molecular Principles (Creighton, Ed., W. H. Freeman and Company, New York (1984)); Introduction to Protein Structure (C. Branden and J. Tooze, eds., Garland Publishing, New York, N.Y. (1991)); and Thornton et al., Nature 354:105 (1991), which are each incorporated herein by reference.
The term "binding affinity (KD)" as used herein, is intended to refer to the dissociation rate of a particular antigen-antibody interaction. The KD is the ratio of the rate of dissociation, also called the "off-rate (koff)", to the association rate, or "on-rate (kon)". Thus, KD equals koff / kon and is expressed as a molar concentration (M). It follows that the smaller the KD, the stronger the affinity of binding. Therefore, a KD of 1 μΜ indicates weaker binding affinity compared to a KD of 1 nM. KD values for antibodies can be determined using methods well established in the art. One method for determining the KD of an antibody is by using surface plasmon resonance, typically using a biosensor system such as a BIAcore® system.
The term "surface plasmon resonance" (SPR), as used herein, refers to an optical phenomenon that allows for the analysis of real-time biospecific interactions by detection of alterations in protein concentrations within a biosensor matrix, for example using the
BIACORE™ system (formerly Pharmacia Biosensor AB, Uppsala, Sweden acquired by GE Healthcare, Little Chalfont, UK). For further descriptions, see Jonsson U. et al., Ann. Biol. Clin. 51 : 19-26 (1993); Jonsson U. et al., Biotechniques 1 1 :620-627 (1991); Jonsson B. et al., J. Mol. Recognit. 8:125-131 (1995); and Johnsson B. et al., Anal. Biochem. 198:268-277 (1991).
As used herein, the twenty naturally occurring amino acids and their abbreviations follow conventional usage. See Immunology - A Synthesis (2nd Edition, E.S. Golub and D.R. Gren, Eds., Sinauer Associates, Sunderland, Mass. (1991)), which is incorporated herein by reference.
The term "polynucleotide" as referred to herein means a polymeric form of nucleotides of at least 10 bases in length, either ribonucleotides or deoxynbonucleotides or a modified form of either type of nucleotide. The term includes single and double stranded forms.
The term "isolated polynucleotide" as used herein means a polynucleotide of genomic, cDNA, or synthetic origin or some combination thereof, which by virtue of its origin or source of derivation, the "isolated polynucleotide" has one to three of the following: (1) is not associated with all or a portion of a polynucleotides with which the "isolated polynucleotide" is found in nature, (2) is operably linked to a polynucleotide to which it is not linked in nature, or (3) does not occur in nature as part of a larger sequence.
The term "oligonucleotide" as used herein includes naturally occurring, and modified nucleotides linked together by naturally occurring and non-naturally occurring oligonucleotide linkages. Oligonucleotides are a polynucleotide subset generally comprising a length of 200 bases or fewer. Preferably oligonucleotides are 10 to 60 bases in length and most preferably 12, 13, 14, 15, 16, 17, 18, 19, or 20 to 40 bases in length. Oligonucleotides are usually single stranded, e.g. for primers and probes; although oligonucleotides may be double stranded, e.g. for use in the construction of a gene mutant. Oligonucleotides of the invention can be either sense or antisense oligonucleotides.
The term "naturally occurring nucleotides" as used herein includes deoxyribonucleotides and ribonucleotides. The term "modified nucleotides" as used herein includes nucleotides with modified or substituted sugar groups and the like. The term "oligonucleotide linkages" referred to herein includes oligonucleotides linkages such as phosphorothioate, phosphorodithioate, phosphoroselenoate, phosphorodiselenoate, phosphoroanilothioate, phoshoraniladate, phosphoroamidate, and the like. See e.g., LaPlanche et al., Nucl. Acids Res. 14:9081 (1986); Stec et al., J. Am. Chem. Soc. 106:6077 (1984); Stein et al., Nucl. Acids Res. 16:3209 (1988); Zon et al., Anti-Cancer Drug Design 6:539 (1991); Zon et al., Oligonucleotides and Analogues: A Practical Approach, pp. 87-108 (F. Eckstein, Ed., Oxford University Press, Oxford England (1991)); U.S. Patent No. 5,151 ,510; Uhlmann and Peyman, Chemical Re views 90:543 (1990), the disclosures of which are hereby incorporated by reference. An oligonucleotide can include a label for detection, if desired.
Operably linked" sequences include both expression control sequences that are contiguous with the gene of interest and expression control sequences that act in trans or at a distance to control the gene of interest. The term "expression control sequence" as used herein means polynucleotide sequences that are necessary to effect the expression and processing of coding sequences to which they are ligated. Expression control sequences include appropriate transcription initiation, termination, promoter and enhancer sequences; efficient RNA processing signals such as splicing and polyadenylation signals; sequences that stabilize cytoplasmic mRNA; sequences that enhance translation efficiency (i.e., Kozak consensus sequence);
sequences that enhance protein stability; and when desired, sequences that enhance protein secretion. The nature of such control sequences differs depending upon the host organism; in prokaryotes, such control sequences generally include promoter, ribosomal binding site, and transcription termination sequence; in eukaryotes, generally, such control sequences include promoters and transcription termination sequence. The term "control sequences" is intended to include, at a minimum, all components whose presence is essential for expression and processing, and can also include additional components whose presence is advantageous, for example, leader sequences and fusion partner sequences.
The term "vector", as used herein, means a nucleic acid molecule capable of transporting another nucleic acid to which it has been linked. In some embodiments, the vector is a plasmid, i.e., a circular double stranded DNA loop into which additional DNA segments may be ligated. In some embodiments, the vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome. In some embodiments, the vectors are capable of autonomous replication in a host cell into which they are introduced (e.g. , bacterial vectors having a bacterial origin of replication and episomal mammalian vectors). In other
embodiments, the vectors (e.g., non-episomal mammalian vectors) can be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome. Moreover, certain vectors are capable of directing the expression of genes to which they are operatively linked. Such vectors are referred to herein as "recombinant expression vectors" (or simply, "expression vectors").
The term "recombinant host cell" (or simply "host cell"), as used herein, means a cell into which an exogenous nucleic acid and/or recombinant vector has been introduced. It should be understood that "recombinant host cell" and "host cell" mean not only the particular subject cell but also the progeny of such a cell. Because certain modifications may occur in succeeding generations due to either mutation or environmental influences, such progeny may not, in fact, be identical to the parent cell, but are still included within the scope of the term "host cell" as used herein.
The term "selectively hybridize" referred to herein means to detectably and specifically bind. Polynucleotides, oligonucleotides and fragments thereof in accordance with the invention selectively hybridize to nucleic acid strands under hybridization and wash conditions that minimize appreciable amounts of detectable binding to nonspecific nucleic acids. "High stringency" or "highly stringent" conditions can be used to achieve selective hybridization conditions as known in the art and discussed herein. One example of "high stringency" or "highly stringent" conditions is the incubation of a polynucleotide with another polynucleotide,
wherein one polynucleotide may be affixed to a solid surface such as a membrane, in a hybridization buffer of 6X SSPE or SSC, 50% formamide, 5X Denhardt's reagent, 0.5% SDS, 100 μg ml denatured, fragmented salmon sperm DNA at a hybridization temperature of 42°C for 12-16 hours, followed by twice washing at 55°C using a wash buffer of 1X SSC, 0.5% SDS. See also Sambrook et a/., supra, pp. 9.50-9.55.
The term "percent sequence identity" in the context of nucleic acid sequences means the percent of residues when a first contiguous sequence is compared and aligned for maximum correspondence to a second contiguous sequence. The length of sequence identity comparison may be over a stretch of at least about nine nucleotides, usually at least about 18 nucleotides, more usually at least about 24 nucleotides, typically at least about 28 nucleotides, more typically at least about 32 nucleotides, and preferably at least about 36, 48 or more nucleotides. There are a number of different algorithms known in the art which can be used to measure nucleotide sequence identity. For instance, polynucleotide sequences can be compared using FASTA, Gap or Bestfit, which are programs in Wisconsin Package Version 10.0, Genetics Computer Group (GCG), Madison, Wisconsin. FASTA, which includes, e.g., the programs FASTA2 and FAST A3, provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences (Pearson, Methods Enzymol. 183:63-98 (1990); Pearson, Methods Mol. Biol. 132:185-219 (2000); Pearson, Methods Enzymol. 266:227-258 (1996); Pearson, J. Mol. Biol. 276:71-84 (1998); herein incorporated by reference). Unless otherwise specified, default parameters for a particular program or algorithm are used. For instance, percent sequence identity between nucleic acid sequences can be determined using FASTA with its default parameters (a word size of 6 and the NOPAM factor for the scoring matrix) or using Gap with its default parameters as provided in GCG Version 6.1 , herein incorporated by reference.
A reference to a nucleotide sequence encompasses its complement unless otherwise specified. Thus, a reference to a nucleic acid having a particular sequence should be understood to encompass its complementary strand, with its complementary sequence.
The term "percent sequence identity" means a ratio, expressed as a percent of the number of identical residues over the number of residues compared.
The term "substantial similarity" or "substantial sequence similarity," when referring to a nucleic acid or fragment thereof, means that when optimally aligned with appropriate nucleotide insertions or deletions with another nucleic acid (or its complementary strand), there is nucleotide sequence identity in at least about 85%, preferably at least about 90%, 91 %, 92%,
93%, 94%, 95%, 96%, 97%, 98% or 99% of the nucleotide bases, as measured by any well- known algorithm of sequence identity, such as FASTA, BLAST or Gap, as discussed above.
As applied to polypeptides, the term "substantial identity" means that two peptide sequences, when optimally aligned, such as by the programs GAP or BESTFIT using default gap weights, as supplied with the programs, share at least 70%, 75%, 80% or 85% sequence identity, preferably at least 90%, 91 %, 92%, 93%, 94% 95%, 96%, 97%, 98% or 99% sequence identity. In certain embodiments, residue positions that are not identical differ by conservative amino acid substitutions.
A "conservative amino acid substitution" is one in which an amino acid residue is substituted by another amino acid residue having a side chain R group with similar chemical properties (e.g., charge or hydrophobicity). In general, a conservative amino acid substitution will not substantially change the functional properties of a protein. In cases where two or more amino acid sequences differ from each other by conservative substitutions, the percent sequence identity may be adjusted upwards to correct for the conservative nature of the substitution. Means for making this adjustment are well-known to those of skill in the art. See, e.g., Pearson, Methods Mol. Biol. 243:307-31 (1994). Examples of groups of amino acids that have side chains with similar chemical properties include 1) aliphatic side chains: glycine, alanine, valine, leucine, and isoleucine; 2) aliphatic-hydroxyl side chains: serine and threonine; 3) amide-containing side chains: asparagine and glutamine; 4) aromatic side chains:
phenylalanine, tyrosine, and tryptophan; 5) basic side chains: lysine, arginine, and histidine; 6) acidic side chains: aspartic acid and glutamic acid; and 7) sulfur-containing side chains:
cysteine and methionine. Conservative amino acids substitution groups are:
valine-leucine-isoleucine, phenylalanine-tyrosine, lysine-arginine, alanine-valine,
glutamate-aspartate, and asparagine-glutamine.
Alternatively, a conservative substitution or replacement, as the terms are used interchangeably herein, is any change having a positive value in the PAM250 log-likelihood matrix disclosed in Gonnet et al., Science 256: 1443-45 (1992), herein incorporated by reference. A "moderately conservative" replacement is any change having a nonnegative value in the PAM250 log-likelihood matrix.
Sequence identity for polypeptides, is typically measured using sequence analysis software. Protein analysis software matches sequences using measures of similarity assigned to various substitutions, deletions and other modifications, including conservative amino acid substitutions. For instance, GCG contains programs such as "Gap" and "Bestfit" which can be used with default parameters, as specified with the programs, to determine sequence homology
or sequence identity between closely related polypeptides, such as homologous polypeptides from different species of organisms or between a wild type protein and a mutein thereof. See, e.g., GCG Version 6.1. Polypeptide sequences also can be compared using FASTA using default or recommended parameters, see GCG Version 6.1. (University of Wisconsin Wl) FASTA (e.g., FASTA2 and FASTA3) provides alignments and percent sequence identity of the regions of the best overlap between the query and search sequences (Pearson, Methods Enzymol. 183:63-98 (1990); Pearson, Methods Mol. Biol. 132: 185-219 (2000)). Another preferred algorithm when comparing a sequence of the invention to a database containing a large number of sequences from different organisms is the computer program BLAST, especially blastp or tblastn, using default parameters, as supplied with the programs. See, e.g., Altschul et al., J. Mol. Biol. 215:403-410 (1990); Altschul et al., Nucleic Acids Res. 25:3389-402 (1997).
The length of polypeptide sequences compared for homology will generally be at least about 16 amino acid residues, usually at least about 20 residues, more usually at least about 24 residues, typically at least about 28 residues, and preferably more than about 35 residues.
When searching a database containing sequences from a large number of different organisms, it is preferable to compare amino acid sequences.
The term "potency" is a measurement of biological activity and may be designated as IC50, or effective concentration of a protein needed to inhibit 50% of a biological activity in a cell which activity is mediated by the protein.
The phrase "effective amount" or "therapeutically effective amount" as used herein refers to an amount necessary (at dosages and for periods of time and for the means of
administration) to achieve the desired therapeutic result. An effective amount is at least the minimal amount, but less than a toxic amount, of an active agent which is necessary to impart therapeutic benefit to a subject.
The term "inhibit" or "neutralize" as used herein with respect to bioactivity of a polypeptide of the invention means the ability of the polypeptide to substantially antagonize, prohibit, prevent, restrain, slow, disrupt, eliminate, stop, reduce or reverse e.g. progression or severity of that which is being inhibited including, but not limited to, a biological activity.
The term "compete", as used herein with regard to polypeptide, means that a first polypeptide binds to an epitope in a manner sufficiently similar to the binding of a second polypeptide such that the result of binding of the first polypeptide with its cognate epitope is detectably decreased in the presence of the second polypeptide compared to the binding of the first polypeptide in the absence of the second polypeptide. The alternative, where the binding of
the second polypeptide to its epitope is also detectably decreased in the presence of the first polypeptide, can, but need not be the case. That is, a first polypeptide can inhibit the binding of a second polypeptide to its epitope without that second polypeptide inhibiting the binding of the first polypeptide to its respective epitope. However, where each polypeptide detectably inhibits the binding of the other polypeptide with its cognate epitope or ligand, whether to the same, greater, or lesser extent, the polypeptides are said to "cross-compete" with each other for binding of their respective epitope(s). Both competing and cross-competing polypeptides are encompassed by the present invention. Regardless of the mechanism by which such competition or cross-competition occurs (e.g., steric hindrance, conformational change, or binding to a common epitope, or portion thereof), the skilled artisan would appreciate, based upon the teachings provided herein, that such competing and/or cross-competing polypeptides are encompassed and can be useful for the methods disclosed herein.
By "IgG" as used herein is meant a polypeptide belonging to the class of antibodies that are substantially encoded by a recognized immunoglobulin gamma gene. In humans this class comprises lgG1 , lgG2, lgG3, and lgG4. In mice this class comprises lgG1 , lgG2a, lgG2b, lgG3. By "immunoglobulin (lg)" herein is meant a protein consisting of one or more polypeptides substantially encoded by immunoglobulin genes. Immunoglobulins include but are not limited to antibodies. Immunoglobulins may have a number of structural forms, including but not limited to full length antibodies, antibody fragments, and individual immunoglobulin domains. By
"immunoglobulin (lg) domain" herein is meant a region of an immunoglobulin that exists as a distinct structural entity as ascertained by one skilled in the art of protein structure, lg domains typically have a characteristic folding topology. The known lg domains in the IgG class of antibodies are the variable heavy chain domain (VH), the heavy chain constant domains - Cy1 , Cv2, Cy3 - together comprising the Cy domain which includes the hinge region between Cy1 and Cy2 , the variable domain of the light chain (VL), and the constant domain of the light chain (CL), which in humans comprises either the kappa (CK) or lambda (CA) light chain constant domain.
As known in the art, the term "Fc region" is used to define a C-terminal region of an immunoglobulin heavy chain. The "Fc region" (also known as the "fragment crystallizable" or "tail" region) may be a native sequence Fc region or a variant Fc region. Although the boundaries of the Fc region of an immunoglobulin heavy chain might vary, the human IgG heavy chain Fc region is usually defined to stretch from an amino acid residue at position Cys226, or from Pro230, to the carboxyl-terminus thereof. For all heavy chain constant region amino acid positions discussed in the present invention, numbering is according to the Eu index
first described in Edelman et al., 1969, Proc. Natl. Acad. Sci. USA 63(1):78-85, describing the amino acid sequence of myeloma protein Eu, which is the first human lgG1 sequenced. The EU index of Edelman et al. is also set forth in Kabat et al., Sequences of Proteins of
Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md., 1991. Thus, the "EU index as set forth in Kabat" or "EU index of Kabat" refers to the amino acid residue numbering system based on the human lgG1 EU antibody of Edelman et al. as set forth in Kabat 1991.
The Fc region of an immunoglobulin generally comprises two constant domains, CH2 and CH3. Typically, an "Fc polypeptide," as the term is used herein, comprises a CH2 and a CH3 domain and can include at least a portion of the hinge domain, but does not usually include the entire CH1 domain. As is known in the art, an Fc region can be present in dimeric or monomeric form.
As used in the art, "Fc receptor" and "FcR" describe a receptor that binds to the Fc region of an antibody. The preferred FcR is a native sequence human FcR. Moreover, a preferred FcR is one which binds an IgG antibody (a gamma receptor) and includes receptors of the FcvRI, FcvRII, and FcvRIII subclasses, including allelic variants and alternatively spliced forms of these receptors. FcvRII receptors include FcvRIIA (an "activating receptor") and FcvRIIB (an "inhibiting receptor"), which have similar amino acid sequences that differ primarily in the cytoplasmic domains thereof. FcRs are reviewed in Ravetch and Kinet, 1991 , Ann. Rev. Immunol., 9:457-92; Capel et al., 1994, Immunomethods, 4:25-34; and de Haas et al., 1995, J. Lab. Clin. Med., 126:330-41. "FcR" also includes the neonatal receptor, FcRn, which is responsible for the transfer of maternal IgGs to the fetus (Guyer et al., 1976, J. Immunol., 117:587; and Kim et al., 1994, J. Immunol., 24:249).
As used herein, "pharmaceutically acceptable carrier" or "pharmaceutical acceptable excipient" includes any material which, when combined with an active ingredient, allows the ingredient to retain biological activity and is non-reactive with the subject's immune system. Compositions comprising such carriers are formulated by well known conventional methods (see, for example, Remington's Pharmaceutical Sciences, 18th edition, A. Gennaro, ed., Mack Publishing Co., Easton, PA, 1990; and Remington, The Science and Practice of Pharmacy 20th Ed. Mack Publishing, 2000).
The term "treating", as used herein, unless otherwise indicated, means reversing, alleviating, inhibiting the progress of, delaying the progression of, delaying the onset of, or preventing the disorder or condition to which such term applies, or one or more symptoms of such disorder or condition. The term "treatment", as used herein, unless otherwise indicated,
refers to the act of treating as "treating" is defined immediately above. The term "treating" also includes adjuvant and neo-adjuvant treatment of a subject. For the avoidance of doubt, reference herein to "treatment" includes reference to curative, palliative and prophylactic treatment.
Throughout this specification and claims, the word "comprise," or variations such as
"comprises" or "comprising," will be understood to imply the inclusion of a stated integer or group of integers but not the exclusion of any other integer or group of integers.
Modified IL21 R ECD Proteins
This application discloses novel modified IL21 R ECD proteins having substantially improved useful characteristics. Such proteins, and fusion proteins thereof, may be used, for example, as antagonists of IL21 cytokine activity.
By examining the X-ray crystal structure of human wild type IL21 R ECD protein complexed to IL21 cytokine, the tryptophan residue at position 148 (also referred to as Trp148 or W148) of wild type IL21 R protein was identified at the interface of two IL21 R monomers in a dimer configuration of the IL21 R-IL21 cytokine complex. Modified IL21 R proteins were designed by mutating the amino acid at or around W148, that is, within four amino acid residues of W148 using SEQ ID NO:1 as a reference, to produce constructs having at least one characteristic of greater stability, in vitro expression, reduced aggregation, and increased half- life in order to develop a therapeutic protein with improved characteristics over wild type proteins, making them improved therapeutics with decreased cost of goods making them also commercially desirable.
Accordingly, disclosed herein are modified IL21 R ECD proteins having one or more amino acid substitutions at or within four amino acid residues on either side, i.e., "flanking", W148 with respect to SEQ ID NO: 1. The substitution at W148 or within four amino acids residues on either side of W148 may be any naturally occurring amino acid, including glycine, alanine, leucine, methionine, phenylalanine, lysine, glutamine, glutamic acid, serine, proline, valine, isoleucine, cysteine, tyrosine, histidine, arginine, asparagine, aspartic acid, and threonine. In a specific embodiment, W148 is substituted with a serine or asparagine.
In other embodiments, additional amino acids to W148, or within four amino acid residues surrounding W148, may also be substituted. In some embodiments, the substitution may be at position 49 with respect to SEQ ID NO:1. In further embodiments, the substitutions may be made at one or more of positions 49, 147, 148, 149 or 150. In exemplary embodiments, the modified IL21 R ECD may comprise substitutions selected from the group consisting of:
a. Glycine at position 147, serine at position 148, glycine at position 149 (SEQ ID NO:2) ;
b. Asparagine at position 148, serine at position 150 (SEQ ID NO: 3); c. Asparagine at position 148, glycine at position 149, serine at position 150 (SEQ ID NO:4);
d. Serine at position 148, glycine at position 149 (SEQ ID NO:7); and e. Asparagine at position 49, asparagine at position 148, and serine at position 150 (SEQ ID NO:5).
In a further embodiment, the modified IL21 R ECD protein may further comprise a substitution of the aspartic acid residue at position 122 (D122). In specific embodiments, D122 may be substituted for by alanine, isoleucine, tryptophan, phenylalanine, or tyrosine.
Table 1 provides a summary of the various exemplary IL21 R ECD mutations disclosed herein. Table 2 provides the complete sequences of various IL21 R ECD proteins. Table 1 : Summary of Amino Acid Substitutions in Modified IL21 R ECD Proteins Relative to Wild
Type IL21 R Protein (mutated positions are in bold)
Table 2: Modified IL21R ECD Protein Sequences
Modified IL21 R ECD8 CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 9
EATSCSLHRSAHNATHATYTCHMDVFHFMADDIFSVNITDQ SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRIDY EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP
Modified IL21 R ECD9 CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 10
EATSCSLHRSAHNATHATYTCHMDVFHFMADDIFSVNITDQ SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSWY EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP
Modified IL21 R ECD10 CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 11
EATSCSLHRSAHNATHATYTCHMDVFHFMADDIFSVNITDQ SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSFY EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP
Modified IL21 R ECD11 CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 12
EATSCSLHRSAHNATHATYTCHMDVFHFMADDIFSVNITDQ SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSYY EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP
Modified IL21 R Fusion Proteins
This application also discloses fusion proteins of the modified IL21 R ECD proteins disclosed herein.
As used herein, the term "IL21 R fusion polypeptide" or "IL21 R fusion protein" refers to a fusion of one or more amino acid residues (such as a heterologous protein or peptide) at the N- terminus or C-terminus of any modified IL21 R ECD protein described herein. Thus, the term "fusion protein" refers to a protein or polypeptide that has an amino acid sequence derived from two or more proteins. The fusion protein may also include linking regions of amino acids between amino acid portions derived from separate proteins.
Heterologous peptides and polypeptides include, but are not limited to, an epitope (e.g., FLAG) or a tag sequence (e.g., His6, and the like) to allow for the detection and/or isolation of an IL21 R polypeptide mutant; a transmembrane receptor protein or a portion thereof, such as an extracellular domain or a transmembrane and intracellular domain; a ligand or a portion thereof which binds to a transmembrane receptor protein; an enzyme or portion thereof which is catalytically active; a polypeptide or peptide which promotes oligomerization, such as a leucine zipper domain; a polypeptide or peptide which increases stability, such as an immunoglobulin
constant region (e.g., an Fc domain); a half life-extending sequence comprising a combination of two or more (e.g., 2, 5, 10, 15, 20, 25, etc) naturally occurring or non-naturally occurring charged and/or uncharged amino acids (e.g., Serine, Glycine, Glutamic or Aspartic Acid) designed to form a predominantly hydrophilic or predominantly hydrophobic fusion partner for a modified IL21 R ECD protein; a functional or non-functional antibody, or a heavy or light chain thereof; and a polypeptide which has an activity, such as a therapeutic activity, different from the modified IL21 R ECD proteins of the present invention.
IL21 R fusion proteins can be made by fusing heterologous sequences at either the N- terminus or at the C-terminus of a modified IL21 R ECD protein. As described herein, a heterologous sequence can be an amino acid sequence or a non-amino acid-containing polymer. Heterologous sequences can be fused either directly to the modified IL21 R ECD protein either chemically or by recombinant expression from a single polynucleotide or they may be joined via a linker or adapter molecule. A peptidyl linker or adapter molecule can be one or more amino acid residues (or -mers), e.g., 1 , 2, 3, 4, 5, 6, 7, 8, or 9 residues (or -mers), preferably from 10 to 50 amino acid residues (or -mers), e.g., 10, 1 1 , 12, 13, 14, 15, 16, 17, 18, 19, 20, 25, 30, 35, 40, 45, or 50 residues (or -mers), and more preferably from 15 to 35 amino acid residues (or -mers). A linker or adapter molecule can also be designed with a cleavage site for a DNA restriction endonuclease or for a protease to allow for the separation of the fused moieties. a) Linkers
When forming the fusion proteins of the present invention, a linker can, but need not, be employed. The linker can be made up of amino acids linked together by peptide bonds, i.e., a peptidyl linker. In some embodiments of the present invention, the linker is made up of from 1 to 20 or more amino acids linked by peptide bonds, wherein the amino acids are selected from the 20 naturally occurring amino acids. In some embodiments, the amino acids are selected from the amino acids glycine, serine, and glutamate. In some embodiments, suitable linkers include: GSGEGEGSEGSG (SEQ ID NO:13); GGSEGEGSEGGS (SEQ ID NO:14); and GGGS (SEQ ID NO:15). While a linker of 12 amino acid residues has been found to work with the modified IL21 RFc fusion proteins disclosed herein, the present invention contemplates linkers of any length or composition. Exemplary linkers are shown in Table 3.
Table 3: Linker Sequences
The linkers described herein are exemplary, and linkers that are much longer and which include other residues are contemplated by the present invention b) Fc Proteins
In some embodiments of the present invention, the modified IL21 R ECD proteins are fused to an Fc domain, e.g., one or more domains of an Fc region of a human IgG. Antibodies comprise two functionally independent parts, a variable domain known as "Fab," that binds an antigen, and a constant domain known as "Fc," that is involved in, among other things, effector functions such as complement activation and attack by phagocytic cells. An Fc has a long serum half-life, whereas a Fab is short-lived (Capon et ah, 1989, Nature 337: 525-31) such that when joined together with a therapeutic protein, an Fc domain can provide longer half-life or incorporate such effector functions as Fc receptor binding, protein A binding, complement fixation, and other characteristics that are desirable in a therapeutic protein.
In vivo pharmacokinetic analysis indicated that wild type human IL21 R has a short half- life. Therefore, to extend the half-life of IL21 RFc, various Fc sequences were fused to the modified IL21 R ECD proteins disclosed herein. The fusion of the Fc sequence to wild type IL21 R ECD did not extend the half-life as much as was desired, however this observation led to the identification of modified IL21 R ECDs (as disclosed herein and illustrated above in Table 2 ) and modified IL21 RFc fusion proteins with enhanced half-life as exemplified in Example 16. These and other IL21 RFc fusion proteins form embodiments of the present invention. Table 4 below illustrates some of the Fc modifications exemplified in this application, and Table 5 below illustrates some of the modified IL21 RFc fusion protein constructs. In one embodiment, the modified ECD Fc fusion protein comprises the amino acid sequence encoded by the polynucleotide insert of the vector deposited with the ATCC on July 17, 2013, as ECD1-L1- FC1 with the ATCC Accession Number PTA-120480.
Table 4: Human lgG1 Fc Sequences
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD1-L1-FC2 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSDY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYKQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 22
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD1-L1-FC3 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSDY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 23
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD1-L2-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSDY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GGSEGEGSEGGSKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 24
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD7-L1-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSAY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 25
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD7-L2-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSAY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GGSEGEGSEGGSKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 26
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD8-L1-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSIY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 27
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD9-L1-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSWY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD 28
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD10-L1-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSFY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
Full Length CPDLVCYTDYLQTVICILEMWNLHPSTLTLTWQDQYEELKD
IL21 RFC EATSCSLHRSAHNATHATYTCHMDVFHFMADDI FSVNITDQ
ECD11-L1-FC1 SGNYSQECGSFLLAESIKPAPPFNVTVTFSGQYNISWRSYY
EDPAFYMLKGKLQYELQYRNRGDGSGVSPRRKLISVDSRSV SLLPLEFRKDSSYELQVRAGPMPGSSYQGTWSEWSDPVI FQ TQSEELKEGWNP GSGEGEGSEGSGKTHTCPPCPAPEAAGAP SVFLFPPKPKDTLMISRTPEVTCVVVDVSHEDPEVKFNWYV DGVEVHNAKTKPREEQYNSTYRWSVLTVLHQDWLNGKEYK CKVSNKALPAPIEKTISKAKGQPREPQVYTLPPSREEMTKN QVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDG SFFLYSKLTVDKSRWQQGNVFSCSVLHEALHSHYTQKSLSL SPGK
The resulting modified IL21 RFc fusion protein can be purified, for example, by the use of a Protein A affinity column. Peptides and proteins fused to an Fc region have been found to exhibit a substantially greater half-life in vivo than the unfused counterpart. Also, a fusion to an Fc region allows for dimerization/multimerization of the fusion polypeptide. The Fc region can be a naturally occurring Fc region, such as an lgG1 , lgG2, lgG3 or lgG4 Fc. In one example, an Fc region is a human lgG1 Fc, e.g., SEQ ID NO: 16. The Fc region can also be altered to improve certain qualities, such as modification to reduce effector function, e.g., SEQ ID NO: 19, or modified to improve therapeutic qualities, such as increased circulation time (half-life), e.g., SEQ ID NO: 17.
Pharmaceutical Compositions
Pharmaceutical compositions comprising modified IL21 R ECD proteins, or fusion proteins thereof, are within the scope of the present invention, and are specifically contemplated in light of the identification of several modified IL21 R ECD sequences and fusion proteins exhibiting enhanced properties. Such pharmaceutical compositions can comprise a
therapeutically effective amount of a modified IL21 R ECD protein, or fusion protein thereof, in admixture with a pharmaceutically or physiologically acceptable formulation agent selected for suitability with the mode of administration. Acceptable formulation agents preferably are nontoxic to recipients at the dosages and concentrations employed.
The pharmaceutical composition can contain formulation agent(s) for modifying, maintaining, or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption, or penetration of the composition. Suitable formulation agents include, but are not limited to, amino acids (such as glycine, glutamine, asparagine, arginine, or lysine), antimicrobials, antioxidants (such as
ascorbic acid, sodium sulfite, methionine or sodium hydrogen-sulfite), buffers (such as borate, bicarbonate, Tris-HCI, histidine, citrates, phosphates, or other organic acids), bulking agents (such as mannitol or glycine), chelating agents (such as ethylenediamine tetraacetic acid (EDTA)), complexing agents (such as caffeine, polyvinylpyrrolidone, beta-cyclodextrin, or hydroxypropyl-beta-cyclodextrin), fillers, monosaccharides, disaccharides, and other carbohydrates (such as glucose, mannose, or dextrins), proteins (such as serum albumin, gelatin, or immunoglobulins), coloring, flavoring and diluting agents, emulsifying agents, hydrophilic polymers (such as polyvinylpyrrolidone), low molecular weight polypeptides, salt- forming counterions (such as sodium), preservatives (such as benzalkonium chloride, benzoic acid, salicylic acid, thimerosal, phenethyl alcohol, methylparaben, propylparaben, chlorhexidine, sorbic acid, or hydrogen peroxide), solvents (such as glycerin, propylene glycol, or polyethylene glycol), sugar alcohols (such as mannitol or sorbitol), suspending agents, surfactants or wetting agents (such as pluronics; PEG; sorbitan esters; polysorbates such as polysorbate 20 or polysorbate 80; triton; tromethamine; lecithin; cholesterol or tyloxapal), stability enhancing agents (such as sucrose or sorbitol), tonicity enhancing agents (such as alkali metal halides - preferably sodium or potassium chloride - or mannitol sorbitol), delivery vehicles, diluents, excipients and/or pharmaceutical adjuvants (see, e.g., Remington's Pharmaceutical Sciences (18th Ed., A.R. Gennaro, ed., Mack Publishing Company 1990), and subsequent editions of the same, incorporated herein by reference for any purpose).
The optimal pharmaceutical composition will be determined by a skilled artisan depending upon, for example, the intended route of administration, delivery format, and desired dosage (see, e.g., Remington's Pharmaceutical Sciences, supra). Such compositions can influence the physical state, stability, rate of in vivo release, and rate of in vivo clearance of the modified IL21 R ECD protein or fusion protein thereof.
The primary vehicle or carrier in a pharmaceutical composition can be either aqueous or non-aqueous in nature. For example, a suitable vehicle or carrier for injection can be water, physiological saline solution, or artificial cerebrospinal fluid, possibly supplemented with other materials common in compositions for parenteral administration. Neutral buffered saline or saline mixed with serum albumin are further exemplary vehicles. Other exemplary
pharmaceutical compositions comprise Histidine or Tris buffer of about pH 6.0-8.5, which can further include sorbitol or a suitable substitute. In one embodiment of the present invention, IL21 RFc fusion protein compositions can be prepared for storage by mixing the selected composition having the desired degree of purity with optional formulation agents (Remington's Pharmaceutical Sciences, supra) in the form of a an aqueous solution.
The pharmaceutical compositions can be selected for parenteral delivery. Alternatively, the compositions can be selected for inhalation or for delivery through the digestive tract, such as orally. The preparation of such pharmaceutically acceptable compositions is within the skill of the art. The formulation components are present in concentrations that are acceptable to the site of administration. For example, buffers are used to maintain the composition at
physiological pH or at a slightly lower pH, typically within a pH range of from about 6 to about 8.
When parenteral administration is contemplated, the therapeutic compositions for use in this invention can be in the form of a pyrogen-free, parenterally acceptable, aqueous solution comprising the desired modified IL21 R ECD protein, or fusion protein thereof, in a
pharmaceutically acceptable vehicle. A particularly suitable vehicle for parenteral injection is sterile distilled water in which a modified IL21 R ECD protein, or fusion protein thereof, is formulated as a sterile, isotonic solution, properly preserved. Yet another preparation can involve the formulation of the desired molecule with an agent, such as injectable microspheres, bio-erodible particles, polymeric compounds (such as polylactic acid or polyglycolic acid), beads, or liposomes, that provides for the controlled or sustained release of the product which can then be delivered via a depot injection. Hyaluronic acid can also be used, and this can have the effect of promoting sustained duration in the circulation. Other suitable means for the introduction of the desired molecule include implantable drug delivery devices.
In one embodiment, a pharmaceutical composition can be formulated for inhalation. For example, the pharmaceutical composition can be formulated as a dry powder for inhalation.
Inhalation solutions can also be formulated with a propellant for aerosol delivery. In yet another embodiment, solutions can be nebulized. Pulmonary administration is further described in International Publication No. WO94/20069, which describes the pulmonary delivery of chemically modified proteins.
It is also contemplated that certain formulations can be administered orally. In one embodiment of the present invention, formulations that are administered in this fashion can be formulated with or without those carriers customarily used in the compounding of solid dosage forms such as tablets and capsules. For example, a capsule can be designed to release the active portion of the formulation at the point in the gastrointestinal tract when bioavailability is maximized and pre-systemic degradation is minimized. Additional agents can be included to facilitate absorption. Diluents, flavorings, low melting point waxes, vegetable oils, lubricants, suspending agents, tablet disintegrating agents, and binders can also be employed.
Another pharmaceutical composition can involve an effective quantity of modified IL21 R ECD protein, or a fusion protein thereof, in a mixture with non-toxic excipients that are suitable
for the manufacture of tablets. By dissolving the tablets in sterile water, or another appropriate vehicle, solutions can be prepared in unit-dose form. Suitable excipients include, but are not limited to, inert diluents, such as calcium carbonate, sodium carbonate or bicarbonate, lactose, or calcium phosphate; or binding agents, such as starch, gelatin, or acacia; or lubricating agents such as magnesium stearate, stearic acid, or talc.
Additional pharmaceutical compositions will be evident to those skilled in the art, including formulations involving modified IL21 R ECD proteins, or fusion proteins thereof, in sustained- or controlled-delivery formulations. Techniques for formulating a variety of other sustained- or controlled-delivery means, such as liposome carriers, bio-erodible microparticles or porous beads and depot injections, are also known to those skilled in the art (see, e.g., International Publication No. W093/15722, which describes the controlled release of porous polymeric microparticles for the delivery of pharmaceutical compositions, and Wischke & Schwendeman, 2008, Int. J. Pharm. 364: 298-327, and Freiberg & Zhu, 2004, Int. J. Pharm. 282: 1-18, which discuss microsphere/microparticle preparation and use). As described herein, a hydrogel is an example of a sustained- or controlled-delivery formulation.
Additional examples of sustained-release preparations include semipermeable polymer matrices in the form of shaped articles, e.g. films, or microcapsules. Sustained release matrices can include polyesters, hydrogels, polylactides (U.S. Patent No. 3,773,919 and European Patent No. 0 058 481), copolymers of L-glutamic acid and gamma ethyl-L-glutamate (Sidman et ah, 1983, Biopolymers 22: 547-56), poly(2-hydroxyethyl-methacrylate) (Langer et ah, 1981 , J. Biomed. Mater. Res. 15: 167-277 and Langer, 1982, Chem. Tech. 12: 98-105), ethylene vinyl acetate (Langer et al, supra) or poly-D(-)-3-hydroxybutyric acid (European Patent No. 0 133 988). Sustained-release compositions can also include liposomes, which can be prepared by any of several methods known in the art. See, e.g., Epstein et ah, 1985, Proc. Natl. Acad. Sci. U.S.A. 82: 3688-92; and European Patent Nos. 0 036 676, 0 088 046, and 0 143 949.
The pharmaceutical composition to be used for in vivo administration typically should be sterile. This can be accomplished by filtration through sterile filtration membranes. Where the composition is lyophilized, sterilization using this method can be conducted either prior to, or following, lyophilization and reconstitution. The composition for parenteral administration can be stored in lyophilized form or in a solution. In addition, parenteral compositions generally are placed into a container having a sterile access port, for example, an intravenous solution bag or vial having a stopper pierceable by a hypodermic injection needle. The parenteral composition can be diluted into parenterally acceptable diluents (e.g., saline and 5% Dextrose).
Once the pharmaceutical composition has been formulated, it can be stored in sterile vials as a solution, suspension, gel, emulsion, solid, or as a dehydrated or lyophilized powder. Such formulations can be stored either in a ready-to-use form or in a form (e.g., lyophilized) requiring reconstitution prior to administration.
In one embodiment, the present invention is directed to kits for producing a single-dose administration unit. The kits can each contain both a first container having a dried protein and a second container having an aqueous formulation. Also included within the scope of this invention are kits containing single and multi-chambered pre-filled syringes (e.g., liquid syringes and dual chamber syringes).
In one embodiment, the present invention is directed to a pharmaceutical composition comprising a modified IL21 RFc fusion protein formulated as a powder for injection after reconstitution to a solution for injection. The formulation components may range in
concentration from 0.5 - 200 mg/mL of the modified IL21 R ECDs or the fusion proteins disclosed herein, 10-50 mM tromethamine, 0.01 - 0.10 mg/mL EDTA, 20 - 120 mg/mL sucrose, and 0.01 - 0.4 mg/mL polysorbate 80. In a specific embodiment, upon reconstitution , the composition will contain 100 mg/ml of IL21 RFc fusion protein, 40 mM tromethamine (Tris base), 0.05 mg/ml EDTA, 100 mg/ml sucrose, 0.2 mg/ml polysorbate 80.
Selecting an administration regimen for a therapeutic depends on several factors, including the serum or tissue turnover rate of the entity, the level of symptoms, the
immunogenicity of the entity, and the accessibility of the target cells in the biological matrix. In certain embodiments, an administration regimen maximizes the amount of therapeutic delivered to the patient consistent with an acceptable level of side effects. Accordingly, the amount of biologic delivered depends in part on the particular entity and the severity of the condition being treated. Guidance in selecting appropriate doses of antibodies, Fc fusion therapeutic proteins, cytokines, and small molecules are available (see, e.g., Wawrzynczak, 1996, Antibody Therapy, Bios Scientific Pub. Ltd, Oxfordshire, UK; Kresina (ed.), 1991 , Monoclonal Antibodies, Cytokines and Arthritis, Marcel Dekker, New York, N.Y.; Bach (ed.), 1993, Monoclonal Antibodies and Peptide Therapy in Autoimmune Diseases, Marcel Dekker, New York, N. Y.; Baert, et al., 2003, New Engl. J. Med. 348:601-608; Milgrom, et al., 1999, New Engl. J. Med. 341 :1966-1973;
Slamon, et al., 2001 , New Engl. J. Med. 344:783-792; Beniaminovitz, et al., 2000, New Engl. J. Med. 342:613-619; Ghosh, et al., 2003, New Engl. J. Med. 348:24-32; Lipsky, et al., 2000, New Engl. J. Med. 343:1594-1602).
Determination of the appropriate dose is made by the clinician, e.g., using parameters or factors known or suspected in the art to affect treatment or predicted to affect treatment.
Generally, the dose begins with an amount somewhat less than the optimum dose and it is increased by small increments thereafter until the desired or optimum effect is achieved relative to any negative side effects. Important diagnostic measures include those of symptoms of, e.g., the inflammation or level of inflammatory cytokines produced.
Actual dosage levels of the active ingredients in the pharmaceutical compositions of the present disclosure may be varied so as to obtain an amount of the active ingredient which is effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient. The selected dosage level will depend upon a variety of pharmacokinetic factors including the activity of the particular compositions of the present disclosure employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compositions employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
Compositions comprising modified IL21 R ECD of the disclosure can be provided by continuous infusion, or by doses at intervals of, e.g., one day, one week, or 1-7 times per week. Doses may be provided intravenously, subcutaneously, topically, orally, nasally, rectally, intramuscular, intracerebrally, or by inhalation. A specific dose protocol is one involving the maximal dose or dose frequency that avoids significant undesirable side effects. A total weekly dose may be at least 0.05 μg kg body weight, at least 0.2 μg kg, at least 0.5 μg kg, at least 1 μg kg, at least 10 μg kg, at least 100 μg kg, at least 0.2 mg/kg, at least 1.0 mg/kg, at least 2.0 mg/kg, at least 10 mg/kg, at least 15 mg/kg, at least 20 mg/kg, at least 25 mg/kg, or at least 50 mg/kg (see, e.g., Yang, et al., 2003, New Engl. J. Med. 349:427-434; Herold, et al., 2002, New Engl. J. Med. 346:1692-1698; Liu, et al., 1999, J. Neurol. Neurosurg. Psych. 67:451-456;
Portielji, et al., 2003, Cancer. Immunol. Immunother. 52: 133-144). The dose may be at least 15 μg, at least 20 μg, at least 25 μg, at least 30 μg, at least 35 μg, at least 40 μg, at least 45 μg, at least 50 μg, at least 55 μg, at least 60 μg, at least 65 μg, at least 70 μg, at least 75 μg, at least 80 μg, at least 85 μg, at least 90 μg, at least 95 μg, or at least 100 μg. The doses administered to a subject may number at least 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 1 1 , or 12, or more.
For therapeutic modified IL21 R proteins of the disclosure, the dosage administered to a patient may be 0.0001 mg/kg to 100 mg/kg of the patient's body weight. The dosage may be between 0.0001 mg/kg and 20 mg/kg, 0.0001 mg/kg and 10 mg/kg, 0.0001 mg/kg and 5 mg/kg, 0.0001 and 2 mg/kg, 0.0001 and 1 mg/kg, 0.0001 mg/kg and 0.75 mg/kg, 0.0001 mg/kg and 0.5
mg/kg, 0.0001 mg/'kg to 0.25 mg/kg, 0.0001 to 0.15 mg/kg, 0.0001 to 0.10 mg/kg, 0.001 to 0.5 mg/kg, 0.01 to 0.25 mg/kg or 0.01 to 0.10 mg/kg of the patient's body weight.
The dosage of the therapeutic protein of the disclosure may be calculated using the patient's weight in kilograms (kg) multiplied by the dose to be administered in mg/kg. The dosage of the proteins of the disclosure may be 150 μg/kg or less, 125 μg/kg or less, 100 μg/kg or less, 95 μg/kg or less, 90 μg/kg or less, 85 μ/kg or less, 80 μ/kg or less, 75 μ/kg or less, 70 μ/kg or less, 65 μ/kg or less, 60 μ/kg or less, 55 μ/kg or less, 50 μ/kg or less, 45 μ/kg or less, 40 μ/kg or less, 35 μ/kg or less, 30 μ/kg or less, 25 μ/kg or less, 20 μ/kg or less, 15 μ/kg or less, 10 μ/kg or less, 5 μ/kg or less, 2.5 μ/kg or less, 2 μ/kg or less, 1.5 μ/kg or less, 1 μ/kg or less, 0.5 μ/kg or less, or 0.1 μ/kg or less of a patient's body weight.
Unit dose of the therapeutic proteins of the disclosure may be 0.1 mg to 20 mg, 0.1 mg to 15 mg, 0.1 mg to 12 mg, 0.1 mg to 10 mg, 0.1 mg to 8 mg, 0.1 mg to 7 mg, 0.1 mg to 5 mg, 0.1 to 2.5 mg, 0.25 mg to 20 mg, 0.25 to 15 mg, 0.25 to 12 mg, 0.25 to 10 mg, 0.25 to 8 mg, 0.25 mg to 7 m g, 0.25 mg to 5 mg, 0.5 mg to 2.5 mg, 1 mg to 20 mg, 1 mg to 15 mg, 1 mg to 12 mg, 1 mg to 10 mg, 1 mg to 8 mg, 1 mg to 7 mg, 1 mg to 5 mg, or 1 mg to 2.5 mg.
The dosage of the therapeutic proteins of the disclosure may achieve a serum titer of at least 0.1 μg/ml, at least 0.5 μg/ml, at least 1 μg/ml, at least 2 μg/ml, at least 5 μg/ml, at least 6 μg/ml, at least 10 μg/ml, at least 15 μg/ml, at least 20 μg/ml, at least 25 μg/ml, at least 50 μg/ml, at least 100 μg/ml, at least 125 μg/ml, at least 150 v, at least 175 μg/ml, at least 200 μg/ml, at least 225 g/ml, at least 250 g/ml, at least 275 g/ml, at least 300 g/ml, at least 325 g/ml, at least 350 g/ml, at least 375 g/ml /ml, or at least 400 g/ml /ml in a subject. Alternatively, the dosage of the antibodies of the disclosure may achieve a serum titer of at least 0.1 g/ml, at least 0.5 g/ml, at least 1 g/ml, at least, 2 g/ml, at least 5 g/ml, at least 6 g/ml, at least 10 g/ml, at least 15 g/ml, at least 20 g/ml, at least 25 g/ml, at least 50 g/ml, at least 100 g/ml, at least 125 g/ml, at least 150 g/ml, at least 175 g/ml, at least 200 g/ml, at least 225 g/ml, at least 250 g/ml, at least 275 g/ml, at least 300 g/ml, at least 325 g/ml, at least 350 g/ml, at least 375 g/ml, or at least 400 g/ml in the subject.
Doses of therapeutic proteins of the disclosure may be repeated and the administrations may be separated by at least 1 day, 2 days, 3 days, 5 days, 10 days, 15 days, 30 days, 45 days, 2 months, 75 days, 3 months, or at least 6 months.
An effective amount for a particular patient may vary depending on factors such as the condition being treated, the overall health of the patient, the method route and dose of administration and the severity of side effects (see, e.g., Maynard, et al., 1996, A Handbook of
SOPs for Good Clinical Practice, Interpharm Press, Boca Raton, Fla.; Dent, 2001 , Good Laboratory and Good Clinical Practice, Urch Publ, London, UK).
The route of administration may be by, e.g., topical or cutaneous application, injection or infusion by intravenous, intraperitoneal, intracerebral, intramuscular, intraocular, intraarterial, intracerebrospinal, intralesional, or by sustained release systems or an implant (see, e.g.,
Sidman et al., 1983, Biopolymers 22:547-556; Langer, et al., 1981 , J. Biomed. Mater. Res. 15: 167-277; Langer, 1982, Chem. Tech. 12:98-105; Epstein, et al., 1985, Proc. Natl. Acad. Sci. USA 82:3688-3692; Hwang, et al., 1980, Proc. Natl. Acad. Sci. USA 77:4030-4034; U.S. Pat. Nos. 6,350466 and 6,316,024). Where necessary, the composition may also include a solubilizing agent and a local anesthetic such as lidocaine to ease pain at the site of the injection. In addition, pulmonary administration can also be employed, e.g., by use of an inhaler or nebulizer, and formulation with an aerosolizing agent. See, e.g., U.S. Pat. Nos. 6,019,968, 5,985,320, 5,985,309, 5,934,272, 5,874,064, 5,855,913, 5,290,540, and 4,880,078; and PCT Publication Nos. WO 92/19244, WO 97/32572, WO 97/44013, WO 98/31346, and WO
99/66903, each of which is incorporated herein by reference their entirety. In one embodiment, an engineered antibody or engineered antibody conjugate, combination therapy, or a composition of the disclosure is administered using Alkermes AIR™ pulmonary drug delivery technology (Alkermes, Inc., Cambridge, Mass.).
The frequency of dosing will depend upon the pharmacokinetic parameters of the modified IL21 R ECD protein or fusion protein thereof in the formulation being used. Typically, a clinician will administer the composition until a dosage is reached that achieves the desired effect. The composition can therefore be administered as a single dose, as two or more doses (which may or may not contain the same amount of the desired molecule) over time, or as a continuous infusion via an implantation device or catheter. Further refinement of the appropriate dosage is routinely made by those of ordinary skill in the art and is within the ambit of tasks routinely performed by them. Appropriate dosages can be ascertained through use of appropriate dose-response data.
The route of administration of the pharmaceutical composition is in accord with known methods, e.g., orally; through injection by subcutaneous, intravenous, intraperitoneal, intracerebral (intraparenchymal), intracerebroventncular, intramuscular, intraocular, intraarterial, intraportal, or intralesional routes; by sustained release systems (which may also be injected); or by implantation devices. Where desired, the compositions can be administered by bolus injection or continuously by infusion, or by implantation device.
Alternatively or additionally, the composition can be administered locally via implantation of a membrane, sponge, or other appropriate material onto which the desired molecule has been absorbed or encapsulated. Where an implantation device is used, the device can be implanted into any suitable tissue or organ, and delivery of the desired molecule can be via diffusion, timed-release bolus, or continuous administration. In order to deliver drug, e.g., a modified IL21 R ECD protein, or fusion protein thereof as disclosed herein, at a predetermined rate such that the drug concentration can be maintained at a desired therapeutically effective level over an extended period, a variety of different approaches can be employed. In one example, a hydrogel comprising a polymer such as a gelatin (e.g., bovine gelatin, human gelatin, or gelatin from another source) or a naturally-occurring or a synthetically generated polymer can be employed. Any percentage of polymer (e.g., gelatin) can be employed in a hydrogel, such as 5, 10, 15 or 20%. The selection of an appropriate concentration can depend on a variety of factors, such as the therapeutic profile desired and the pharmacokinetic profile of the therapeutic molecule.
Examples of polymers that can be incorporated into a hydrogel include polyethylene glycol ("PEG"), polyethylene oxide, polyethylene oxide-co-polypropylene oxide, co-polyethylene oxide block or random copolymers, polyvinyl alcohol, polyvinyl pyrrolidinone), poly(amino acids), dextran, heparin, polysaccharides, polyethers and the like.
Another factor that can be considered when generating a hydrogel formulation is the degree of crosslinking in the hydrogel and the crosslinking agent. In one embodiment, cross- linking can be achieved via a methacrylation reaction involving methacrylic anhydride. In some situations, a high degree of cross-linking may be desirable while in other situations a lower degree of crosslinking is preferred. In some cases a higher degree of crosslinking provides a longer sustained release. A higher degree of crosslinking may provide a firmer hydrogel and a longer period over which drug is delivered. Any ratio of polymer to crosslinking agent (e.g., methacrylic anhydride) can be employed to generate a hydrogel with desired properties. For example, the ratio of polymer to crosslinker can be, e.g., 8: 1 , 16: 1 , 24: 1 , or 32: 1. For example, when the hydrogel polymer is gelatin and the crosslinker is methacrylate, ratios of 8: 1 , 16: 1 , 24:1 , or 32: 1 methyacrylic anhydride:gelatin can be employed.
Methods of Treatment
Modified IL21 R ECD proteins, fusion proteins thereof, and pharmaceutical compositions comprising the modified IL21 R ECD proteins or fusion proteins thereof, can be used to regulate at least one IL21-mediated or IL21 R-mediated immune response, such as one or more of cell
proliferation, cytokine expression or secretion, chemokine secretion, and cytolytic activity, of T cells, B cells, NK cells, macrophages, or synovial cells. Accordingly, the proteins of the invention can be used to inhibit the activity (e.g., proliferation, differentiation, and/or survival) of an immune or hematopoietic cell (e.g., a cell of myeloid, lymphoid, or erythroid lineage, or precursor cells thereof), and, thus, can be used to treat a variety of diseases or disorders mediated by interaction of IL21 with IL21 R. Accordingly, the modified IL21 R ECD proteins, or fusion proteins thereof, or pharmaceutical compositions thereof, of this disclosure can used in the treatment or prevention of IL21 or IL21 R-mediated disorders. In addition, the invention provides for use of the modified IL21 R ECD proteins, or fusion proteins thereof, or
pharmaceutical compositions thereof, of this disclosure in the manufacture of a medicament for use in treatment or prevention of IL21 or IL21 R-mediated disorders. In another embodiment, this application discloses modified IL21 R ECD proteins, or fusion proteins thereof, or pharmaceutical compositions thereof, for use in treatment of IL21 or I L21 R-mediated disorders. In a further embodiment, this application discloses pharmaceutical compositions comprising the modified IL21 R ECD proteins, or fusion proteins thereof, of this disclosure for use in treating or preventing IL21 or I L21 R-mediated diseases.
Examples of immune disorders that can be treated include, but are not limited to, transplant rejection, graft-versus-host disease (GVHD), allergies (for example, atopic allergy), and autoimmune diseases, including diabetes mellitus, arthritic disorders (including rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, and ankylosing spondylitis), spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis (including atopic dermatitis and eczematous dermatitis), psoriasis, Sjogren's syndrome, IBD (including Crohn's disease and ulcerative colitis), asthma (including intrinsic asthma and allergic asthma), scleroderma, vasculitis, and Behcet's Disease.
In application, a disorder or condition mediated by the interaction between IL21 and IL21 R can be treated by administering modified IL21 R ECD protein, or fusion protein thereof, or a pharmaceutical composition thereof, as described herein, to a patient in need thereof in the amount of a therapeutically effective dose. The administration can be performed as described herein, such as by IV injection, intraperitoneal injection, intramuscular injection, or orally in the form of a tablet or liquid formation. In most situations, a desired dosage can be determined by a clinician, as described herein, and can represent a therapeutically effective dose of modified IL21 R ECD or fusion protein. It will be apparent to those of skill in the art that a therapeutically effective dose will depend, inter alia, upon the administration schedule, the unit dose of agent
administered, whether the composition is administered in combination with other therapeutic agents, the immune status and the health of the recipient. The term "therapeutically effective dose," as used herein, means that amount of modified IL21 R ECD protein or fusion protein thereof that elicits the biological or medicinal response in a tissue system, animal, or human being sought by a researcher, medical doctor, or other clinician, which includes alleviation of the symptoms of the disease or disorder being treated.
Biological Deposit
Representative materials of the present invention were deposited in the American Type Culture Collection, 10801 University Boulevard, Manassas, Va. 201 10-2209, USA, on July 17, 2013. Vector EDC1-L1-FC1 , having ATCC Accession No. PTA-120480, comprises a DNA insert encoding modified IL21 R ECD1 , linker 1 , and Fc variant designated FC1. The deposit was made under the provisions of the Budapest Treaty on the International Recognition of the Deposit of Microorganisms for the Purpose of Patent Procedure and Regulations thereunder (Budapest Treaty). This assures maintenance of a viable culture of the deposit for 30 years from the date of deposit. The deposit will be made available by ATCC under the terms of the Budapest Treaty, and subject to an agreement between Pfizer Inc. and ATCC, which assures permanent and unrestricted availability of the progeny of the culture of the deposit to the public upon issuance of the pertinent U.S. patent or upon laying open to the public of any U.S. or foreign patent application, whichever comes first, and assures availability of the progeny to one determined by the U.S. Commissioner of Patents and Trademarks to be entitled thereto according to 35 U.S.C. Section 122 and the Commissioner's rules pursuant thereto (including 37 C.F.R. Section 1.14 with particular reference to 886 OG 638).
The assignee of the present application has agreed that if a culture of the materials on deposit should die or be lost or destroyed when cultivated under suitable conditions, the materials will be promptly replaced on notification with another of the same. Availability of the deposited material is not to be construed as a license to practice the invention in contravention of the rights granted under the authority of any government in accordance with its patent laws.
Exemplary Embodiments
The invention is further described in detail by reference to the following experimental examples. These examples are provided for purposes of illustration only, and are not intended to be limiting unless otherwise specified. Thus, the invention should in no way be construed as
being limited to the following examples, but rather, should be construed to encompass any and all variations which become evident as a result of the teaching provided herein.
EXAMPLES
Example 1. X-ray Crystal Structure of Wild Type Human IL21 R ECD
The X-ray structure of the human IL21 R protein complexed with its cytokine was solved. More specifically, IL21 and IL21 R ECD were expressed in CHO cells and purified
independently. Both cytokine and receptor were then mixed in a one to one ratio and applied to a SourceQ anion exchanger chromatography to further purify the complex. Selected fractions were concentrated to 7.3 mg/ml and a hanging drop crystallization screen was realized. After optimization, good quality crystals were obtained from 1720mM Ammonium Sulfate, 10mM Cobaltous Chloride and 100mM MES buffer at pH 6.43. The crystals were frozen and data was collected at the National Synchrotron Light Source (NSLS, Brookhaven, NY) produced diffraction to 2.6A resolution. The symmetry space group was 14122 and the structure was solved through molecular replacement technique using the IL4/IL4R complex structure available from the protein data bank.
The crystal structure identified tryptophan as position 148 (W148) of IL21 R ECD as a critical contributor to a dimer interface between two monomers of the receptor (Figure 1).
Example 2. Generating Modified IL21 R ECD Proteins
Wild type human IL21 R ECD has a relatively low thermal stability measured at Tm=51 °C by DSC with an onset of melting at 40°C. When formatted as an Fc fusion, the wild type IL21 R ECD specific transition midpoint is further reduced down to 48°C. Working from the X-ray structure of the IL21 receptor (Figure 1), various mutants of human IL21 R ECD with a C- terminal hexa histidine tag were designed. In particular, the tryptophan at position 148 is structurally in a solvent exposed flexible loop and may create an unfavorable conformation due to its highly hydrophobic nature. Surprisingly, mutation of this amino acid, and optionally its flanking residues, to a different residue, including more hydrophilic residues, stabilized the flexible loop of the IL21 R ECD and decreased possible negative entropic effects associated with the high solvation penalty from the native tryptophan.
In addition, the aspartate residue at position 122 (D122) was identified as a residue that, when mutated, provided significant and unexpected stabilization of modified IL21 R ECD constructs. D122 sits in the middle of a hydrophobic structural environment. As shown in
Figure 2, substitution of D122 with para-acetyl phenylalanine (paF) significantly enhanced thermal stability of the modified IL21 RFc ECD relative to other paF mutants at other solvent exposed sites on the protein. Table 6 sets forth the Tm values for the various modified IL21 R ECDs from Figure 2.
Table 6: Tm Values of Variant IL21 R ECDs
Additional D122 position mutants substituted with hydrophobic residues were designed to mimic the stabilizing effect observed with the substitution of the non-natural amino acid paF. Table 2 lists various IL21 R ECD mutants and their amino acid sequences. Bolded and underlined residues in each of the modified IL21 R ECD constructs were mutated relative to wild type IL21 R ECD (SEQ ID NO:1).
Example 3: Expression of Modified IL21 R ECD Constructs
Various modified IL21 R ECD constructs as listed in Table 2 were transiently expressed as IL21 R-ECD-His6 constructs in HEK293 cells.
HEK293F cells obtained from the [American Type Culture Collection (ATCC, Manassas,
VA) were cultured in freestyle 293 medium (Invitrogen, Grand Island, NY). Cells were grown and maintained in a humidified incubator with 7% C02 at 37°C. Conditioned media for the constructs were produced from a standard large-scale transient HEK293 transfection process.
The conditioned media were filtered at 0.22 micron prior to purification.
The secreted proteins from the conditioned media were analyzed by Western Blot as follows. Serum-free conditioned media samples were resolved by SDS PAGE on precast
NuPAGE Bis-Tris mini gels 4-12% gradient (Invitrogen) according to the manufacturer's protocol. Further, each well was loaded with an identical aliquot of conditioned medium obtained from a sample comprising a equal number of cells compared with the other lanes of the gel.
Gels are stained for protein visualization using Coomassie Brilliant Blue R-250 and the gels were transferred under 250 milliamps current onto nitrocellulose membranes for 0.5 h at 4°C. In
the case of monomelic His tagged proteins, Western membranes were probed with mouse anti- Histidine IgG (Qiagen, cat#: 34670) as primary antibody followed by horseradish peroxidase- conjugated rabbit anti-mouse IgG (Pierce, cat#: 32430) as secondary antibody. For Western blot on Fc fusion proteins, the nitrocellulose membranes were developed with a single horseradish peroxidase-conjugated goat anti-human IgG (Sigma, cat#: A0170) antibody.
Figure 3 and Figure 4 depict a Western blot analysis demonstrating that expression of modified IL21 R ECD constructs ECD1 , ECD2, ECD3, and ECD4 was greater than expression of wild type IL21 R ECD. Thus, each of the modified constructs has improved expression in the HEK293 cells as compared to the wild type protein.
Example 4: Purification of Modified IL21 R ECD Constructs Comprising a His tag
Conditioned media from transient HEK293 or CHO stable cultures were typically diafiltered against PBS pH 7.2 before loading onto an appropriately sized Nickel NTA Superflow (Qiagen) chromatography column equilibrated in 300mM NaCI, 50mM NaHPO pH 8. The column was washed with 10CV of 300mM NaCI, 50mM NaHPO, 15mM Imidazole pH 8 before being resolved by a 5CV linear gradient to 300mM NaCI, 50mM NaHPO, 250mM Imidazole pH8. The protein peak pool eluted from the Nickel NTA step was loaded on a Superdex 200 size exclusion chromatography column (GE Healthcare) equilibrated and developed in PBS. Finally, the SEC Fractions with high purity were pooled, concentrated and frozen for long term storage at -80°C. Biophysical characteristics of the purified modified IL21 R ECD constructs were further investigated as discussed below.
Example 5: Differential scanning calorimetry
Thermal denaturation experiments were performed on a Capillary-DSC instrument (MicroCal, Northampton, MA). Protein samples were dialyzed into PBS buffer, pH 7.2 at 4°C, normalized at a concentration of 1 mg/ml and extensively degassed before analysis. They were scanned from 20 to 90°C, at a scan rate of 1 °C min"1 against the dialysis buffer contained in the reference cell. The thermograms generated with the protein samples were analyzed using Origin 7.0 software (MicroCal Software). Data were converted to excess heat capacity, reference trace-subtracted, normalized for concentration and analyzed for Tm determination.
Figure 5 depicts a differential scanning calorimetry thermogram demonstrating thermal stability enhancement of modified IL21 R ECD constructs ECD1 , ECD2, and ECD4 as compared to wild type IL21 R ECD. The Tm for the mutated monomeric IL21 R ECDs was increased by about 6-8°C, clearly demonstrating a positive stabilization effect of the mutations over the wild
type sequence. The formatting of IL21 R ECD1 as an Fc fusion preserved the thermal stability gain by 4°C (data not shown). Again, Tm for the mutated monomeric IL21 R ECD constructs was increased over the wild type protein demonstrating that the substitution of W148 and flanking residues at positions 147 and 149 (PWA, respectively) with GSG conferred greater stability upon the modified IL21 R ECD1.
Example 6: Analytical size exclusion chromatography
Various modified IL21 R ECD constructs were analyzed for purity by analytical size exclusion chromatography on an Agilent 1200 system using a Superdex 200 10/300GL (GE Healthcare) chromatography column. Typically 20 to 100 microgram of sample was loaded through the autosampler and injected on the column developed in PBS, pH6.8 at a flow rate of 0.75 ml/min.
Figure 6 depicts an analytical size exclusion chromatograph that demonstrates reduced aggregations levels for modified IL21 R construct ECD1 as compared to wild type IL21 R following Nickel NTA initial affinity capture purification. Molecules were analyzed by analytical SEC immediately after the IMAC capture and imidazole elution. The IL21 R wild type molecule presents approximately 30% high molecular weight (HMW) species eluting ahead of the major peak of interest visible at 20 minute. In identical conditions, the ECD1 construct has a significantly decreased amount of HMW species, down to 10%, approximately 20% less high molecular weight aggregates than its wild type version. This not only improved the overall production process yields but also facilitated the final formulation and increased the stability of the product.
Figure 7 depicts results from an analytical size exclusion chromatograph that demonstrates reduced aggregations levels in modified IL21 R constructs using ECD1 , ECD2, and ECD4 compared with wild type. Each construct is purified to homogeneity and freed of high molecular weight species by Nickel affinity capture and size exclusion chromatography. The wild type, ECD1 and ECD2 molecules were incubated overnight in PBS buffer at the two different temperatures of 4°C and 40°C. Immediately following this incubation, the protein samples were analyzed by SEC. The 4°C samples were all consistent with the quality of the starting material with no visible HMW species detectable by SEC. Conversely, the 40°C samples presented a differential profile between the WT and ECD1/ECD2. That is, the high temperature incubation originated aggregation only for the wild type sample representing up to 16% of the total integrated OD280nm signal. Both modified IL21 R constructs ECD1 and ECD2
demonstrated resistance to aggregation properties compared to wild type IL21 R in temperature stress condition.
Example 7: Generation of Modified IL21 RFc Constructs
Various modified IL21 RFc fusion proteins were designed based on the modified IL21 R
ECD constructs described above in Table 2. Linkers and human lgG1 Fc proteins were selected as described below. Figure 8 shows a structural representation of the modified IL21 RFc fusion proteins.
a) Linker Design
By exploring linker structure options it was determined that the desired stoichiometry of 2 cytokine molecules to 1 Fc dimeric fusion molecule was not easily achieved . That is, the classic (Gly4Ser)n motif typically used in protein engineering to genetically link protein domains did not adequately support the desired target engagement stoichiometry. Isothermal titration calorimetry was employed to investigate the stoichiometry of IL21 cytokine binding of an IL21 RFc fusion protein construct. As demonstrated by the graph in Figure 9, the IL21 R monomer molecule demonstrated approximately the correct binding stoichiometry of N=1 with one cytokine binding one monomeric receptor. In the case of the homodimeric Fc fusion molecule, the use of a G4S motif (four glycines and one serine) to link the IL21 receptor to the Fc domain did not allow the expected N=2 binding stoichiometry. Instead, only a stoichiometry of approximately N=1 is measured. To be noted, longer lengths of the linker in multiples of the G4S motif did not help improve the stoichiometry back to the expected N=2.
The modified linker sequence L1 , as shown in Table 3, allowed for full engagement of IL21 , with one IL21 RFc homodimeric molecule able to trap two free IL21 cytokine molecules. The results of a BIAcore study depicted in Figure 10 demonstrate full 2: 1 stoichiometry of binding of I L21 to lL21 RFc. Briefly, I L21 R Fc is coated on the BIAcore chip for a theoretical Rmax of 50.83 RU. IL21 cytokine is titrated progressively to saturation and the Rmax is recorded at 49.5RU. This observed value represents 97.4% of a full stoichiometry of two cytokine bound per IL21 RFc homodimer on the chip.
In addition, modifications were made to Linker L1 to eliminate O-linked glycosylation by switching S-G residues at the ends of the linkers, as illustrated in Figure 11. The resulting linker is designated Linker L2, as shown in Table 3. Upon fine characterization by mass spectrometry and glycoanalysis, it was concluded that the Linker L1 sequence supported the post- translational modification of two serines by O-linked glycosylation. The heterogeneity of the species was accentuated by the partial nature of O-linked glycan occupancy and its variable
size. In the specific Linker sequence L1 GSGEGEGSEGSG (SEQ ID NO: 13), the first and last serines carry the O-linked modification. As SG motifs have been associated with higher potential for O-linked occupancy (Mann et al., J Biol Chem. 1990 Mar 25;265(9):5317-23), the sequence was modified from SG to GS to disrupt this O-linked glycosylation motif. The resulting new linker sequence - GGSEGEGSEGGS - demonstrated better homogeneity by Western blot, as discussed in Example 8 below. b) Human lgG1 Fc Design
As one of the desired properties of IL21 RFc fusion proteins is to trap and clear plasma and tissue circulating IL21 cytokine, native Fc functions such as antibody-dependent cell- mediated cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC) are generally not preferred. Using a set of mutations as described by Winter et al. in US Patent Nos. 5,624,821 and 5,648,260, effector functions were engineered out of human lgG1 Fc by mutating the positions Leu242, Leu243 and Gly245 to alanines (234, 235 and 237, respectively, per Kabat antibody numbering (see Table 4). These mutations from LLGL to AAGA are expected to reduce Fc effector functions including Fc mediated cell depletion.
Increased serum residence time was also engineered in by mutating two residues at the interface between the CH2 and CH3 in the Fc domain. That is, the mutations M436L and N442S (428 and 434 respective to Eu antibody Kabat numbering (see Table 4) have been shown to increase the binding affinity to FcRn and the in-vivo half life as described in International Patent Publication No. WO 2009/086320.
As disclosed in Example 1 1 below, BIAcore studies demonstrated that these mutations applied to modified IL21 RFc fusion proteins disclosed herein, such as ECD1-L1-FC1 , increased by a ten-fold factor the affinity to human FcRn as directly compared to its Fc wild type homologue molecule. As further disclosed in Example 12, the actual in-vivo NHP PK profile benefitted significantly from these two mutations, doubling the drug exposure and affecting positively both the terminal half life, which was increased three-fold, as well as the clearance rate, which was reduced to one half, compared with the wild type protein WTECD-L1-FC1.
Various modified IL21 RFc fusion proteins were designed, and several are listed in Table 5. As will be apparent to persons skilled in the art, many other combinations of modified IL21 RFc fusion proteins may be generated by combining any of the modified IL21 R ECDs disclosed above, with any one of Linker 1 or Linker 2, or any other appropriate linker, with any of the human lgG1 Fc proteins disclosed above, or any other appropriate Fc proteins.
Example 8: Expression and Purification of Modified IL21 RFc Fusion Proteins
Figure 12 depicts expression of the modified IL21 RFc constructs ECD1-L1-FC1 , ECD1- L1-FC2, ECD-L2-FC1 , and ECD7-L2-FC1 as listed in Table 5 as analyzed using Western blotting. The culture medium supernatant was run by non-reducing SDS gel electrophoresis. Following electro-transfer of the proteins from the gel to nitrocellulose membranes (NCM), the NCM were developed with a horseradish peroxidase-conjugated goat anti-human IgG (Sigma, cat. #: A0170) antibody. Each lane was loaded with an equal cell count. Both ECD1-L1-FC1 and ECD1-L1-FC2 demonstrated identical banding patterns by Western blot and the main migration band of the non-reduced IL-21 RFc is indicated by the arrow and which presented marked smearing immediately above it. This distinct smearing pattern is likely associated with the heterogeneous O-linked glycosylation post-translational modification of the L1 linker sequence. The banding pattern of ECD1-L2-FC1 showed a marked decrease in this smearing
demonstrating the beneficial effect of the optimized L2 linker sequence which eliminated the O- linked glycosylation motif of the L1 linker. ECD7-L2-FC1 presents even further improved and cleaner banding pattern with no high molecular weight smearing above the IL-21 RFc band. This demonstrates the additional beneficial effect on aggregation obtained through the ECD7 engineering which comprises the substitution of GSG at amino acid residues 147, 148 and 149, respectively, and the additional substitution of D122A. To further purify the modified IL21 RFc fusion proteins, conditioned media from transient or stable CHO cultures were typically loaded neat onto an appropriately sized protein A chromatography column equilibrated in PBS pH 7.2 (rmpProteinA Sepharose Fast Flow, GE Healthcare). The column was then washed with 5 column volumes (CV) of PBS before being resolved by a 10 CV linear gradient to 20 mM citric acid 150 mM NaCI pH2.5. The protein A peak pool was diluted 1 to 3 with 20mM Tris pH8.6 for binding to source 15Q column (GE Healthcare) and eluted with a 15 CV linear gradient to 20mM Tris 1 M NaCI pH8.6. The Source 15Q peak containing the IL21 R.Fc was then pooled and applied over a Superdex200 size exclusion chromatography column (GE Healthcare) equilibrated and developed in PBS. The final fractions were 0.2 micron filtered and concentrated to variable concentrations before freezing for long term storage at -80°C. Proteins were further characterized as described below.
Example 9: SDS PAGE and IEF gel electrophoresis
Modified IL21 RFc fusion protein construct ECD1-L1-FC1 was analyzed for purity by SDS PAGE as described above, and by isoelectric focusing (IEF) to confirm pi. IEF gel
electrophoresis was performed on precast vertical Novex pH 3-10 IEF 5% polyacrylamide gels
(Invitrogen) according to the manufacturer's protocol. Gels were stained for protein visualization using Coomassie Brilliant Blue R-250. Figure 13 depicts the SDS and IEF gels.
IL21 R ECD has five occupied N-linked glycosylation sites creating heterogeneity in the molecular charge as illustrated by the diffuse band migrating around the 4.2 pi marker of the IEF gel.
The SDS PAGE demonstrates a high purity of material with no other band detected in the reduced condition. The non-reduced condition shows predominantly the disulfide bridged covalent IL21 RFc homodimer fusion protein migrating as expected around 140 kD, as well as a minor component of non-covalently bound equivalent to its reduced monomeric form migrating at about 70 kD.
Example 10: Differential Scanning Calorimetry (DSQ of Modified IL21 RFc Constructs
DSC was performed as described above on modified IL21 RFc fusion proteins ECD7-L1- FC1 , ECD8-L1-FC1 , ECD9-L1-FC1 , ECD10-L1-FC1 , and ECD1 1-L1-FC1 as listed in Table 5, and compared to ECD1-L1-FC1 fusion protein. The DSC thermogram is depicted in Figure 14. Table 7 is a summary of the Tm temperatures of the various constructs from the thermogram in Figure 14.
Table 7: Thermal Transition Midpoints (Tm) of Various IL21 RFc Constructs by DSC
For each of the modified IL21 RFc constructs, the DSC transition midpoint of the mutated IL21 R domain was extended from 3.2 to 4.6 °C relative to the native D122 native molecule. These lower values compared to the 6 degree gain on the monomer (see Figure 2) are attributed to both the different structural contribution of the natural amino acids compared to paF as well as the destabilizing effect of the Fc formatting.
Example 11 : BIAcore Studies on Modified IL21 RFc Fusion Constructs Binding to FcRn
BIAcore analysis was performed to determine the steady-state affinity (KD) for different IL21 RFc engineered molecules for binding human FcRn. BIAcore technology utilizes changes in the refractive index at the surface layer of a sensor upon binding of the IL21 RFc molecules to human FcRn protein immobilized on the layer. Binding is detected by surface plasmon resonance (SPR) of laser light refracting from the surface. Human FcRn was specifically biotinylated through an engineered Avi-tag using the BirA reagent (Catalog #: BIRA500, Avidity, LLC, Aurora, Colorado) and immobilized onto a streptavidin (SA) sensor chip to enable uniform orientation of the FcRn protein on the sensor. Next, various concentrations of the IL21 RFc molecules in 20mM MES (2-(N-morpholino)ethanesulfonic acid pH 6.0, with 150 mM NaCI, 3 mM EDTA (ethylenediaminetetraacetic acid), 0.5% Surfactant P20 (MES-EP) were injected over the chip surface. The surface was regenerated using HBS-EP + 0.05% Surfactant P20 (GE Healthcare, Piscataway, NJ), pH 7.4, between injection cycles. The steady-state binding affinities were determined for the modified IL21 RFc constructs and these were compared to each other. The data is averaged from a minimum of two experiments on a minimum of two different surfaces. The BIAcore results shown in Figure 15 illustrate the binding kinetic effect of engineering FcRn binding enhancement in the Fc domain. The FC1 containing IL21 RFc fusion protein, which contains mutations in the Fc component to both enhance FcRn binding and in vivo half-life, demonstrated a 10-fold improved steady-state KD to FcRn over the non engineered FC3 containing molecule.
Example 12: Non-Human Primate Pharmacokinetic (PK) Profiles of Various Modified IL21 RFc Fusion Proteins
In this assay, test article was captured by recombinant human IL-21-FLAG
(DYKDDDDK) tag and detected using biotinylated anti-human IgG antibody. Streptavidin Poly- 80 horseradish peroxidase (HRP) enzyme conjugate and substrate ABTS (2,2' azino-di(3-ethyl- benzthiazoline-6-sulfonate) were used to produce a colored end product and optical density was measured spectrophotometrically at a wavelength of 405 nm. Sample concentrations were determined by interpolation from a standard curve that was fitted using a 4-parameter logistic equation. The lower limit of quantitation was 15-40 ng/mL.
A single 2 mg/kg dose of various modified IL21 RFc fusions proteins was administered IV or SC to female naive cynomolgus monkeys. Figure 16 depicts PK profiles of non-human primates injected with modified IL21 RFc fusion protein constructs ECD1-L1-FC1 , ECD1-L1-FC3 and wild type IL21 RFc. Table 8 is a summary of the PK profiles depicted in Figure 16.
Table 8: Summary of PK Profiles of Various IL21 RFc Constructs
The wild type ECD formatted as Fc fusion linked by a linker comprising four glycine and a serine (G4S; designated as L3) demonstrated a poor PK profile and low drug exposure presumed to result from the low bioavailability, low average terminal half-life as well as the unexpectedly high clearance rate. The first pass of construct engineering, introducing both ECD1 and linker L1 optimization, brought the PK profile back to numbers expected for typical Fc fusion molecules with a clearance rate below 1 ml/hr/kg. The pharmacokinetic properties were then further improved by two-fold overall exposure gain through the introduction of the FC1 engineering with FcRn binding enhancement mutations as discussed in Example 6 and 1 1 above. Furthermore, ECD1-L1-FC1 demonstrated increased bioavailability at 83.6%.
Example 13: Human Primary B Cell Proliferation Assays Using Modified IL21 RFc Fusion Constructs
Buffy coat cells from healthy human donors were incubated with a B cell enrichment cocktail (RosetteSep; StemCell Technologies, Vancouver, British Columbia, Canada), and isolated by the negative selection method described in the manufacturer's instructions. The purity of the isolated B cells was measured by flow cytometry after staining with anti-CD19. The enriched cells were approximately 60 - 80 % CD19+ B cells.
Enriched human B cells were cultured in 0.2 mL RPMI containing 10% FBS, 50 U/mL penicillin, 50 ug/mL streptomycin, and 2 mM L-glutamine at 1 X 105/well in 96-well flat bottom plates. B cells were incubated with serially diluted anti-human IL21 receptor antibodies, 1 ug/mL anti-CD40 mAb (BD Bisosciences) and 20 ng/mL IL21 cytokine for 3 days in a 37C° incubator adjusted to 5% C02. On day 3, cultures were pulsed with 0.5 uCi/well [3H]thymidine (Perkin- Elmer NEN, Boston, MA) and harvested 5 h later onto glass fiber filter mats. [3H]Thymidine incorporation was determined by liquid scintillation counting. The raw data was graphed in Excel / XLFIT4.
Figure 17 is a graph depicting the results of a primary IL21 dependent B cell proliferation assay using modified IL21 RFc construct ECD1-L1-FC1 and wild type IL21 RFc positive control WTECD-L1-FC3. The human lgG1 control demonstrated no effect on B cell proliferation. Both mutant and wild type IL21 RFc proteins showed equal potency in this assay with an IC50 around 10nM demonstrating that the GSG mutations of the ECD1 do not affect the biological function of the IL21 receptor in recognizing the cytokine.
Example 14: Human T Cell Proliferation Assays Using Modified IL21 RFc Fusion Constructs
Human CD4+ T cells were isolated from buffy coats with a ROSETTESEP™ CD4+ T cell enrichment cocktail (Stem Cell Technologies, Vancouver, BC, Canada) according to the manufacturer's instructions. Cells (80-90% CD4+/CD3+ T cells) were activated for 3 days with anti-CD3/anti-CD28-coated microspheres (prepared in house) in RPMI containing 10% FBS, 100 U/ml penicillin, 100 g/ml streptomycin, 2 mM L-glutamine, and HEPES (37°C, 5% C02). Microspheres were then removed, and the cells were washed once prior to resting overnight at ~1 x 106 cells/ml in culture medium and once before addition to assay plates. Test articles were diluted in culture medium in flat-bottomed 96-well plates, followed by sequential addition of human IL-21 (25 ng/ml final concentration) and 105 cells/well. After 72 hr, cells were pulsed with 1 pCi/well 3H-thymidine (Perkin Elmer (NEN)) for 6 hr and harvested onto glass fiber filter mats for liquid scintillation counting.
Figure 18 shows a graph depicting the results of an IL21 dependent primary T cell proliferation assay using modified IL21 RFc constructs ECD7-L1-FC1 , ECD8-L1-FC1 , ECD9-L1- FC1 , ECD10-L1-FC1 , and ECD1 1-L1-FC1. Table 9 is a summary of the IC50 of each of the constructs. Table 9: IC50 of Various IL21 RFc Constructs in Primary T-Cell Assay
A concentration of 25ng/ml of IL21 cytokine maintained proliferation of the T cells in this assay. Compared to a human lgG1 anti-tetanus toxin negative control antibody and the monomeric IL21 R as positive control, all 5 mutants showed good potency in neutralization of T cell proliferation. The differences in IC50 ranged from 19.6 nM to 28.3 nM and the differences are believed not to be significant.
Example 15: Characterization of Modified IL21 RFc Constructs with Fc Variant T445K
Modified IL21 RFc constructs ECD1-L1-FC1 and ECD1-L1-FC2 were characterized and compared in various side by side experiments. Both proteins expressed to comparable levels and purified equally well following the same protocol. Analytical characterization by SDS PAGE, IEF, size exclusion, ion exchange and hydrophobic interaction chromatographies demonstrated that both ECD1-L1-FC1 and ECD1-L1-FC2 molecules were similar. The differential scanning calorimetry result highlighted minor destabilization of the Fc CH2 and CH3 domains of ECD1- L1-FC2, but this was considered within the norm of variation observed with molecules containing the same human lgG1 Fc fragment. In addition, no significant differences in activity in functional assays for IL21 neutralization, IL21 binding, and FcRn binding, were observed between ECD1-L1-FC1 and ECD1-L1-FC2.
Example 16: Formulation of IL21 RFc Constructs
Modified IL21 RFc construct ECD1-L1-FC1 was formulated as a powder for injection in a
100 mg/mL dosage strength after reconstitution. Total protein content per reconstituted vial was 140 mg (1.4 mL) of ECD1-L1-FC1 but 100 mg (1.0 mL) was withdrawn from the vial. That is, to ensure that 1.0 mL can be withdrawn from the vial, there is an overfill of 0.4 mL. The components of one embodiment of the formulation composition are listed in Table 10.
Table 10. Composition of ECD1-L1-FC1 Powder for Injection, 100 mg/Vial
(mg/vial) Formula
ECD1-L1-FC1 Active Ingredient 140 100 mg/mL
Hydrochloric Acid To adjust pH q.s to pH 7.4 pH 7.4
Water for injection Solvent q.s to 1.0 ml_ 1.4 mL/vial
Although the disclosed teachings have been described with reference to various applications, methods, kits, and compositions, it will be appreciated that various changes and modifications can be made without departing from the teachings herein and the claimed invention below. The foregoing examples are provided to better illustrate the disclosed teachings and are not intended to limit the scope of the teachings presented herein. While the present teachings have been described in terms of these exemplary embodiments, the skilled artisan will readily understand that numerous variations and modifications of these exemplary embodiments are possible without undue experimentation. All such variations and modifications are within the scope of the current teachings.
All references cited herein, including patents, patent applications, papers, text books, and the like, and the references cited therein, to the extent that they are not already, are hereby incorporated by reference in their entirety. In the event that one or more of the incorporated literature and similar materials differs from or contradicts this application, including but not limited to defined terms, term usage, described techniques, or the like, this application controls.
The foregoing description and Examples detail certain specific embodiments of the invention and describes the best mode contemplated by the inventors. It will be appreciated, however, that no matter how detailed the foregoing may appear in text, the invention may be practiced in many ways and the invention should be construed in accordance with the appended claims and any equivalents thereof.
Claims
1. A modified interleukin 21 receptor (IL21 R) protein extracellular domain (ECD)
comprising a modification selected from the group consisting of: a. one or more amino acid substitutions at or within four amino acid residues of tryptophan at position 148 (W148) of the amino acid sequence set forth as SEQ ID NO:1 ; b. one or more amino acid insertions at amino acid position 148 of the amino acid sequence of SEQ ID NO:1 ; and c. deletion of the tryptophan residue at position 148 of the amino acid sequence of SEQ ID NO: 1 ; and further wherein the substituted or inserted amino acid is a naturally-occurring amino acid selected from the group consisting of glycine, alanine, leucine, methionine, phenylalanine, lysine, glutamine, glutamic acid, serine, proline, valine, isoleucine, cysteine, tyrosine, histidine, arginine, asparagine, aspartic acid, and threonine.
2. The modified IL21 R ECD of claim 1 , wherein a. the substituted amino acid at position 148 is selected from serine or asparagine; b. the ECD further comprises at least one amino acid substitution at amino acid
position 147, 149, and 150 of the amino acid sequence set forth as SEQ ID NO: 1 ; c. the amino acid substitution comprises glycine at position 147, serine at position 148, and glycine at position 149 (SEQ ID NO:2);
d. the amino acid substitution comprises asparagine at position 148 and serine at position 150 (SEQ ID NO: 3);
e. the amino acid substitution comprises asparagine at position 148, glycine at
position 149, and serine at position 150 (SEQ ID NO:4);
f. the amino acid substitution comprises serine at position 148 and glycine at position 149 (SEQ ID NO:7);
g. the modified IL21 R ECD comprises serine at position 148 (SEQ ID NO:6);
h. the modified IL21 R ECD comprises an amino acid substitution at amino acid position 147, an amino acid substitution at amino acid position 149, asparagine at position 148, serine at position 150, and further comprises asparagine at position 49 (SEQ ID NO:5).
i. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and an amino acid substitution at position 122 all relative to the amino acid sequence of SEQ ID NO: 1.
j. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and alanine at position 122 all relative to the amino acid sequence of SEQ ID NO: 1. k. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and isoleucine at position 122 all relative to the amino acid sequence of SEQ ID NO: 1.
I. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and tryptophan at position 122 all relative to the amino acid sequence of SEQ ID NO: 1. m. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and phenylalanine at position 122 all relative to the amino acid sequence of SEQ ID NO: 1.
n. the modified IL21 R ECD comprises glycine at position 147, serine at position 148, glycine at position 149, an amino acid substitution at amino acid position 150, and tyrosine at position 122 all relative to the amino acid sequence of SEQ ID NO: 1.
A fusion protein comprising a modified IL21 R ECD according to claim 2 fused to a heterologous amino acid sequence via a peptidyl linker, wherein the linker comprises a sequence selected from: GSGEGEGSEGSG (SEQ ID NO:13), GGSEGEGSEGGS (SEQ ID NO:14), and GGGGS (SEQ ID NO:15).
The fusion protein of claim 3, wherein the heterologous amino acid sequence:
a. comprises a human lgG1 Fc domain comprising the amino acid sequence set forth as SEQ ID NO:16.
b. comprises a human lgG1 Fc domain comprising the amino acid sequence set forth as SEQ ID NO:19
c. comprises a human lgG1 Fc domain comprising the amino acid sequence set forth as SEQ ID NO:17;
d. comprises a human lgG1 Fc domain comprising the amino acid sequence set forth as SEQ ID NO:18;
The fusion protein of claim 4 comprising an amino acid sequence selected from the group consisting of the sequence set forth as SEQ ID NO:20S SEQ ID NO:21 , SEQ ID NO:22, SEQ ID NO:23, SEQ ID NO:24, SEQ ID NO:25, SEQ ID NO:26, SEQ ID NO:27, SEQ ID NO:28 and SEQ ID NO:29.
A fusion protein consisting of the amino acid sequence of SEQ ID NO:20.
A pharmaceutical composition comprising the fusion protein of claim 5, and a
pharmaceutically acceptable agent.
The pharmaceutical composition according to claim 7, wherein the composition comprises 0.5 - 200 mg/mL of the fusion protein of claim 5, 10-50 mM tromethamine, 0.01 - 0.10 mg/mL EDTA, 20 - 120 mg/mL sucrose, and 0.01 - 0.4 mg/mL polysorbate 80.
An isolated nucleic acid encoding the modified IL21 R ECD of claim 2.
An isolated nucleic acid encoding the fusion protein of claim 5.
An isolated nucleic acid comprising the nucleic acid sequence of SEQ ID NO:31.
A vector comprising the nucleic acid of claim 10.
A host cell comprising the nucleic acid of claim 12.
A method of producing a fusion protein comprising a modified IL21 R ECD comprising growing the host cell of claim 16 under conditions where the fusion protein is expressed.
A modified IL21 R-Fc fusion protein encoded by the nucleic acid insert comprised in the vector having ATCC Accession No. PTA-120480.
A modified IL21 R-Fc fusion protein comprising the amino acid sequence encoded by the nucleic acid insert comprised by the vector deposited as ATCC Accession No. PTA- 120480.
A method of treating a disease or disorder mediated by interaction of IL21 with IL21 R comprising administering to a patient in need thereof, the fusion protein of claim 5.
The method of claim 17, wherein the disease or disorder is an inflammatory or autoimmune disease or disorder selected from the group consisting of transplant rejection, graft-versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis, osteoarthritis, psoriatic arthritis, ankylosing spondylitis, spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma, scleroderma, vasculitis, and Behcet's Disease.
Use of the fusion protein of claim 5 in the manufacture of a medicament for treatment of a disease or disorder mediated by interaction of IL21 with IL21 R.
The use of claim 19, wherein the disease or disorder is an inflammatory or autoimmune disease or disorder selected from the group consisting of transplant rejection, graft- versus-host disease (GVHD), multiple sclerosis, allergies, atopic allergy, diabetes mellitus, arthritic disorders, rheumatoid arthritis, juvenile rheumatoid arthritis,
osteoarthritis, psoriatic arthritis, ankylosing spondylitis, spondyloarthropathy, multiple sclerosis, encephalomyelitis, myasthenia gravis, systemic lupus erythematosus, cutaneous lupus erythematosus, autoimmune thyroiditis, dermatitis, atopic dermatitis, eczematous dermatitis, psoriasis, Sjogren's syndrome, IBD, Crohn's disease, ulcerative colitis, asthma, intrinsic asthma, allergic asthma, scleroderma, vasculitis, and Behcet's Disease.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201461931438P | 2014-01-24 | 2014-01-24 | |
| US61/931,438 | 2014-01-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015110930A1 true WO2015110930A1 (en) | 2015-07-30 |
Family
ID=52629618
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2015/050169 WO2015110930A1 (en) | 2014-01-24 | 2015-01-09 | Modified interleukin 21 receptor proteins |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW201531482A (en) |
| WO (1) | WO2015110930A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020023702A1 (en) * | 2018-07-25 | 2020-01-30 | AskGene Pharma, Inc. | Novel il-21 prodrugs and methods of use thereof |
| US11634485B2 (en) | 2019-02-18 | 2023-04-25 | Eli Lilly And Company | Therapeutic antibody formulation |
| US11845801B2 (en) | 2019-06-12 | 2023-12-19 | AskGene Pharma, Inc. | IL-15 prodrugs and methods of use thereof |
Citations (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3773919A (en) | 1969-10-23 | 1973-11-20 | Du Pont | Polylactide-drug mixtures |
| EP0036676A1 (en) | 1978-03-24 | 1981-09-30 | The Regents Of The University Of California | Method of making uniformly sized liposomes and liposomes so made |
| EP0058481A1 (en) | 1981-02-16 | 1982-08-25 | Zeneca Limited | Continuous release pharmaceutical compositions |
| EP0088046A2 (en) | 1982-02-17 | 1983-09-07 | Ciba-Geigy Ag | Lipids in the aqueous phase |
| EP0133988A2 (en) | 1983-08-02 | 1985-03-13 | Hoechst Aktiengesellschaft | Regulating peptide-containing pharmaceutical preparations with retarded release, and process for their preparation |
| EP0143949A1 (en) | 1983-11-01 | 1985-06-12 | TERUMO KABUSHIKI KAISHA trading as TERUMO CORPORATION | Pharmaceutical composition containing urokinase |
| US4880078A (en) | 1987-06-29 | 1989-11-14 | Honda Giken Kogyo Kabushiki Kaisha | Exhaust muffler |
| US5151510A (en) | 1990-04-20 | 1992-09-29 | Applied Biosystems, Inc. | Method of synethesizing sulfurized oligonucleotide analogs |
| WO1992019244A2 (en) | 1991-05-01 | 1992-11-12 | Henry M. Jackson Foundation For The Advancement Of Military Medicine | A method for treating infectious respiratory diseases |
| WO1993015722A1 (en) | 1992-02-07 | 1993-08-19 | Syntex (Usa) Inc. | Controlled delivery of pharmaceuticals from preformed porous microparticles |
| WO1994020069A1 (en) | 1993-03-08 | 1994-09-15 | Amgen Inc. | Pulmonary administration of granulocyte colony stimulating factor |
| US5624821A (en) | 1987-03-18 | 1997-04-29 | Scotgen Biopharmaceuticals Incorporated | Antibodies with altered effector functions |
| WO1997032572A2 (en) | 1996-03-04 | 1997-09-12 | The Penn State Research Foundation | Materials and methods for enhancing cellular internalization |
| WO1997044013A1 (en) | 1996-05-24 | 1997-11-27 | Massachusetts Institute Of Technology | Aerodynamically light particles for pulmonary drug delivery |
| WO1998031346A1 (en) | 1997-01-16 | 1998-07-23 | Massachusetts Institute Of Technology | Preparation of particles for inhalation |
| US5855913A (en) | 1997-01-16 | 1999-01-05 | Massachusetts Instite Of Technology | Particles incorporating surfactants for pulmonary drug delivery |
| US5934272A (en) | 1993-01-29 | 1999-08-10 | Aradigm Corporation | Device and method of creating aerosolized mist of respiratory drug |
| US5985309A (en) | 1996-05-24 | 1999-11-16 | Massachusetts Institute Of Technology | Preparation of particles for inhalation |
| WO1999066903A2 (en) | 1998-06-24 | 1999-12-29 | Advanced Inhalation Research, Inc. | Large porous particles emitted from an inhaler |
| US6019968A (en) | 1995-04-14 | 2000-02-01 | Inhale Therapeutic Systems, Inc. | Dispersible antibody compositions and methods for their preparation and use |
| WO2000053761A2 (en) | 1999-03-09 | 2000-09-14 | Zymogenetics, Inc. | Human cytokine as ligand of the zalpha receptor and uses thereof |
| US6316024B1 (en) | 1996-10-11 | 2001-11-13 | Sequus Pharmaceuticals, Inc. | Therapeutic liposome composition and method of preparation |
| WO2001085792A2 (en) | 2000-05-11 | 2001-11-15 | Genetics Institute, Llc. | Mu-1, member of the cytokine receptor family |
| US6350466B1 (en) | 1994-08-05 | 2002-02-26 | Targesome, Inc. | Targeted polymerized liposome diagnostic and treatment agents |
| WO2003028630A2 (en) | 2001-10-04 | 2003-04-10 | Genetics Institute Llc. | Methods and compositions for modulating interleukin-21 receptor activity |
| WO2006113331A1 (en) * | 2005-04-14 | 2006-10-26 | Wyeth | Methods for treating and preventing fibrosis by il-21 / il-21r antagonists |
| WO2009086320A1 (en) | 2007-12-26 | 2009-07-09 | Xencor, Inc | Fc variants with altered binding to fcrn |
| WO2009100035A2 (en) * | 2008-02-01 | 2009-08-13 | Wyeth | Interleukin-21 (il-21) and il-21 receptor (il-21r) modulation of regulatory t cells and forkhead box p3 (foxp3) |
-
2015
- 2015-01-09 WO PCT/IB2015/050169 patent/WO2015110930A1/en active Application Filing
- 2015-01-23 TW TW104102405A patent/TW201531482A/en unknown
Patent Citations (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3773919A (en) | 1969-10-23 | 1973-11-20 | Du Pont | Polylactide-drug mixtures |
| EP0036676A1 (en) | 1978-03-24 | 1981-09-30 | The Regents Of The University Of California | Method of making uniformly sized liposomes and liposomes so made |
| EP0058481A1 (en) | 1981-02-16 | 1982-08-25 | Zeneca Limited | Continuous release pharmaceutical compositions |
| EP0088046A2 (en) | 1982-02-17 | 1983-09-07 | Ciba-Geigy Ag | Lipids in the aqueous phase |
| EP0133988A2 (en) | 1983-08-02 | 1985-03-13 | Hoechst Aktiengesellschaft | Regulating peptide-containing pharmaceutical preparations with retarded release, and process for their preparation |
| EP0143949A1 (en) | 1983-11-01 | 1985-06-12 | TERUMO KABUSHIKI KAISHA trading as TERUMO CORPORATION | Pharmaceutical composition containing urokinase |
| US5624821A (en) | 1987-03-18 | 1997-04-29 | Scotgen Biopharmaceuticals Incorporated | Antibodies with altered effector functions |
| US5648260A (en) | 1987-03-18 | 1997-07-15 | Scotgen Biopharmaceuticals Incorporated | DNA encoding antibodies with altered effector functions |
| US4880078A (en) | 1987-06-29 | 1989-11-14 | Honda Giken Kogyo Kabushiki Kaisha | Exhaust muffler |
| US5151510A (en) | 1990-04-20 | 1992-09-29 | Applied Biosystems, Inc. | Method of synethesizing sulfurized oligonucleotide analogs |
| US5290540A (en) | 1991-05-01 | 1994-03-01 | Henry M. Jackson Foundation For The Advancement Of Military Medicine | Method for treating infectious respiratory diseases |
| WO1992019244A2 (en) | 1991-05-01 | 1992-11-12 | Henry M. Jackson Foundation For The Advancement Of Military Medicine | A method for treating infectious respiratory diseases |
| WO1993015722A1 (en) | 1992-02-07 | 1993-08-19 | Syntex (Usa) Inc. | Controlled delivery of pharmaceuticals from preformed porous microparticles |
| US5934272A (en) | 1993-01-29 | 1999-08-10 | Aradigm Corporation | Device and method of creating aerosolized mist of respiratory drug |
| WO1994020069A1 (en) | 1993-03-08 | 1994-09-15 | Amgen Inc. | Pulmonary administration of granulocyte colony stimulating factor |
| US6350466B1 (en) | 1994-08-05 | 2002-02-26 | Targesome, Inc. | Targeted polymerized liposome diagnostic and treatment agents |
| US6019968A (en) | 1995-04-14 | 2000-02-01 | Inhale Therapeutic Systems, Inc. | Dispersible antibody compositions and methods for their preparation and use |
| WO1997032572A2 (en) | 1996-03-04 | 1997-09-12 | The Penn State Research Foundation | Materials and methods for enhancing cellular internalization |
| US5985320A (en) | 1996-03-04 | 1999-11-16 | The Penn State Research Foundation | Materials and methods for enhancing cellular internalization |
| WO1997044013A1 (en) | 1996-05-24 | 1997-11-27 | Massachusetts Institute Of Technology | Aerodynamically light particles for pulmonary drug delivery |
| US5874064A (en) | 1996-05-24 | 1999-02-23 | Massachusetts Institute Of Technology | Aerodynamically light particles for pulmonary drug delivery |
| US5985309A (en) | 1996-05-24 | 1999-11-16 | Massachusetts Institute Of Technology | Preparation of particles for inhalation |
| US6316024B1 (en) | 1996-10-11 | 2001-11-13 | Sequus Pharmaceuticals, Inc. | Therapeutic liposome composition and method of preparation |
| WO1998031346A1 (en) | 1997-01-16 | 1998-07-23 | Massachusetts Institute Of Technology | Preparation of particles for inhalation |
| US5855913A (en) | 1997-01-16 | 1999-01-05 | Massachusetts Instite Of Technology | Particles incorporating surfactants for pulmonary drug delivery |
| WO1999066903A2 (en) | 1998-06-24 | 1999-12-29 | Advanced Inhalation Research, Inc. | Large porous particles emitted from an inhaler |
| WO2000053761A2 (en) | 1999-03-09 | 2000-09-14 | Zymogenetics, Inc. | Human cytokine as ligand of the zalpha receptor and uses thereof |
| WO2001085792A2 (en) | 2000-05-11 | 2001-11-15 | Genetics Institute, Llc. | Mu-1, member of the cytokine receptor family |
| WO2003028630A2 (en) | 2001-10-04 | 2003-04-10 | Genetics Institute Llc. | Methods and compositions for modulating interleukin-21 receptor activity |
| WO2006113331A1 (en) * | 2005-04-14 | 2006-10-26 | Wyeth | Methods for treating and preventing fibrosis by il-21 / il-21r antagonists |
| WO2009086320A1 (en) | 2007-12-26 | 2009-07-09 | Xencor, Inc | Fc variants with altered binding to fcrn |
| WO2009100035A2 (en) * | 2008-02-01 | 2009-08-13 | Wyeth | Interleukin-21 (il-21) and il-21 receptor (il-21r) modulation of regulatory t cells and forkhead box p3 (foxp3) |
Non-Patent Citations (79)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING COMPANY |
| A. GENNARO: "Remington's Pharmaceutical Sciences, 18th. ed.", 1990, MACK PUBLISHING CO. |
| ALTSCHUL ET AL., J. MOL. BIOL., vol. 215, 1990, pages 403 - 410 |
| ALTSCHUL ET AL., NUCLEIC ACIDS RES., vol. 25, 1997, pages 3389 - 402 |
| AUSUBEL ET AL.: "Current Protocols in Molecular Biology", 1992, GREENE PUBLISHING ASSOCIATES |
| BACH: "Monoclonal Antibodies and Peptide Therapy in Autoimmune Diseases", 1993, MARCEL DEKKER |
| BAERT ET AL., NEW ENGL. J. MED., vol. 348, 2003, pages 601 - 608 |
| BENIAMINOVITZ ET AL., NEW ENGL. J. MED., vol. 342, 2000, pages 613 - 619 |
| C. BRANDEN AND J. TOOZE: "Introduction to Protein Structure", 1991, GARLAND PUBLISHING |
| CAPEL ET AL., IMMUNOMETHODS, vol. 4, 1994, pages 25 - 34 |
| CAPON, NATURE, vol. 337, 1989, pages 525 - 31 |
| CREIGHTON: "Proteins, Structures and Molecular Principles", 1984, W. H. FREEMAN AND COMPANY |
| DATABASE Protein [online] NCBI; 29 May 2013 (2013-05-29), "predicted interleukin-21 receptor", XP002738768, Database accession no. xp_004623194 * |
| DATABASE UniProt [online] 1 October 2000 (2000-10-01), "RecName: Full=Interleukin-21 receptor; Short=IL-21 receptor; Short=IL-21R; AltName: Full=Lymphocyte receptor beta; Short=LR-beta; AltName: Full=Novel cytokine receptor 8; Short=NR8;", XP002738766, retrieved from EBI accession no. UNIPROT:Q9JHX3 Database accession no. Q9JHX3 * |
| DATABASE UniProt [online] 22 February 2012 (2012-02-22), "SubName: Full=Uncharacterized protein {ECO:0000313|Ensembl:ENSOGAP00000003422}; Flags: Fragment;", XP002738767, retrieved from EBI accession no. UNIPROT:H0WNL4 Database accession no. H0WNL4 * |
| DE HAAS ET AL., J. LAB. CLIN. MED., vol. 126, 1995, pages 330 - 41 |
| DENT: "Good Laboratory and Good Clinical Practice", 2001, URCH PUBL |
| E.S. GOLUB AND D.R. GREN: "Immunology - A Synthesis, 2nd ed.", 1991, SINAUER ASSOCIATES |
| EDELMAN ET AL., PROC. NATL. ACAD. SCI. USA, vol. 63, no. 1, 1969, pages 78 - 85 |
| EPSTEIN ET AL., PROC. NATL. ACAD. SCI. USA, vol. 82, 1985, pages 3688 - 3692 |
| EPSTEIN, PROC. NATL. ACAD. SCI. U.S.A., vol. 82, 1985, pages 3688 - 92 |
| F. ECKSTEIN: "Oligonucleotides and Analogues: A Practical Approach", 1991, OXFORD UNIVERSITY PRESS, article ZON ET AL., pages: 87 - 108 |
| FREIBERG; ZHU, INT. J. PHARM., vol. 282, 2004, pages 1 - 18 |
| GHOSH ET AL., NEW ENGL. J. MED., vol. 348, 2003, pages 24 - 32 |
| GONNET ET AL., SCIENCE, vol. 256, 1992, pages 1443 - 45 |
| GUYER ET AL., J. IMMUNOL., vol. 117, 1976, pages 587 |
| HAMMING OLE J ET AL: "Crystal Structure of Interleukin-21 Receptor (IL-21R) Bound to IL-21 Reveals That Sugar Chain Interacting with WSXWS Motif Is Integral Part of IL-21R", JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 287, no. 12, March 2012 (2012-03-01), pages 9454 - 9460, XP002738765 * |
| HARLOW; LANE: "Antibodies: A Laboratory Manual", 1990, COLD SPRING HARBOR LABORATORY PRESS |
| HERBER ET AL., J. IMMUNOL., vol. 178, 2011, pages 3822 - 3830 |
| HEROLD ET AL., NEW ENGL. J. MED., vol. 346, 2002, pages 1692 - 1698 |
| HWANG ET AL., PROC. NATL. ACAD. SCI. USA, vol. 77, 1980, pages 4030 - 4034 |
| JOHNSSON B., ANAL. BIOCHEM., vol. 198, 1991, pages 268 - 277 |
| JONSSON B. ET AL., J. MOL. RECOGNIT, vol. 8, 1995, pages 125 - 131 |
| JONSSON U. ET AL., ANN. BIOL. CLIN, vol. 51, 1993, pages 19 - 26 |
| JONSSON U., BIOTECHNIQUES, vol. 11, 1991, pages 620 - 627 |
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest, 5th ed.", 1991, NATIONAL INSTITUTES OF HEALTH |
| KASAIAN ET AL., IMMUNITY, vol. 16, 2002, pages 559 - 69 |
| KIM ET AL., J. IMMUNOL., vol. 24, 1994, pages 249 |
| KING ET AL., CELL, vol. 117, 2004, pages 265 - 77 |
| KRESINA: "Monoclonal Antibodies, Cytokines and Arthritis", 1991, MARCEL DEKKER |
| LANGER ET AL., J. BIOMED. MATER. RES, vol. 15, 1981, pages 167 - 277 |
| LANGER, CHEM. TECH, vol. 12, 1982, pages 98 - 105 |
| LANGER, J. BIOMED. MATER. RES., vol. 15, 1981, pages 167 - 277 |
| LAPLANCHE ET AL., NUCL. ACIDS RES., vol. 14, 1986, pages 9081 |
| LEONARD; SPOLSKI, NAT. REV. IMMUNOL., vol. 5, 2005, pages 688 - 98 |
| LIPSKY ET AL., NEW ENGL. J. MED., vol. 343, 2000, pages 1594 - 1602 |
| LIU ET AL., IMMUNOL., vol. 176, 2006, pages 5247 - 54 |
| LIU ET AL., J. NEUROL. NEUROSURG. PSYCH, vol. 67, 1999, pages 451 - 456 |
| MANN ET AL., J BIOL CHEM., vol. 265, no. 9, 25 March 1990 (1990-03-25), pages 5317 - 23 |
| MAYNARD ET AL.: "A Handbook of SOPs for Good Clinical Practice", 1996, INTERPHARM PRESS |
| MILGROM ET AL., NEW ENGL. J. MED., vol. 341, 1999, pages 1966 - 1973 |
| OZAKI ET AL., IMMUNOL, vol. 173, 2004, pages 5361 - 71 |
| OZAKI KATSUTOSHI ET AL: "Cloning of a type I cytokine receptor most related to the IL-2 receptor beta chain", PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES, vol. 97, no. 21, 10 October 2000 (2000-10-10), NATIONAL ACADEMY OF SCIENCES, US, pages 11439 - 11444, XP002186603, ISSN: 0027-8424, DOI: 10.1073/PNAS.200360997 * |
| PARRISH-NOVAK ET AL., NATURE, vol. 408, 2000, pages 57 - 63 |
| PARRISH-NOVAK JULIA ET AL: "Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function", NATURE, vol. 408, no. 6808, 2 November 2000 (2000-11-02), NATURE PUBLISHING GROUP, UNITED KINGDOM, pages 57 - 63, XP002186602, ISSN: 0028-0836, DOI: 10.1038/35040504 * |
| PEARSON, J. MOL. BIOL., vol. 276, 1998, pages 71 - 84 |
| PEARSON, METHODS ENZYMOL., vol. 183, 1990, pages 63 - 98 |
| PEARSON, METHODS ENZYMOL., vol. 266, 1996, pages 227 - 258 |
| PEARSON, METHODS MOL. BIOL, vol. 132, 2000, pages 185 - 219 |
| PEARSON, METHODS MOL. BIOL., vol. 132, 2000, pages 185 - 219 |
| PEARSON, METHODS MOL. BIOL., vol. 243, 1994, pages 307 - 31 |
| PORTIELJI ET AL., CANCER. IMMUNOL. IMMUNOTHER, vol. 52, 2003, pages 133 - 144 |
| RAVETCH; KINET, ANN. REV. IMMUNOL., vol. 9, 1991, pages 457 - 92 |
| REMINGTON: "The Science and Practice of Pharmacy, 20th ed.", 2000, MACK PUBLISHING |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY PRESS |
| SHANG ET AL., CELL. IMMUNOL, vol. 241, 2006, pages 66 - 74 |
| SIDMAN ET AL., BIOPOLYMERS, vol. 22, 1983, pages 547 - 556 |
| SIDMAN, BIOPOLYMERS, vol. 22, 1983, pages 547 - 56 |
| SIVAKUMAR ET AL., IMMUNOLOGY, vol. 112, 2004, pages 177 - 82 |
| SLAMON ET AL., NEW ENGL. J. MED., vol. 344, 2001, pages 783 - 792 |
| STEC ET AL., J. AM. CHEM. SOC., vol. 106, 1984, pages 6077 |
| STEIN ET AL., NUCL. ACIDS RES., vol. 16, 1988, pages 3209 |
| THORNTON ET AL., NATURE, vol. 354, 1991, pages 105 |
| UHLMANN; PEYMAN, CHEMICAL REVIEWS, vol. 90, 1990, pages 543 |
| VOLLMER ET AL., IMMUNOL., vol. 174, 2005, pages 2696 - 2701 |
| WAWRZYNCZAK: "Antibody Therapy", 1996, BIOS SCIENTIFIC PUB. LTD |
| WISCHKE; SCHWENDEMAN, INT. J. PHARM., vol. 364, 2008, pages 298 - 327 |
| YANG ET AL., NEW ENGL. J. MED., vol. 349, 2003, pages 427 - 434 |
| ZON ET AL., ANTI-CANCER DRUG DESIGN, vol. 6, 1991, pages 539 |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020023702A1 (en) * | 2018-07-25 | 2020-01-30 | AskGene Pharma, Inc. | Novel il-21 prodrugs and methods of use thereof |
| CN112534052A (en) * | 2018-07-25 | 2021-03-19 | 奥美药业有限公司 | Novel IL-21 prodrugs and methods of use |
| US11634485B2 (en) | 2019-02-18 | 2023-04-25 | Eli Lilly And Company | Therapeutic antibody formulation |
| US11845801B2 (en) | 2019-06-12 | 2023-12-19 | AskGene Pharma, Inc. | IL-15 prodrugs and methods of use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201531482A (en) | 2015-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20210122805A1 (en) | Fc FUSION PROTEINS COMPRISING NOVEL LINKERS OR ARRANGEMENTS | |
| CN103168048B (en) | dimeric VSTM3 fusion proteins and related compositions and methods | |
| JP6621741B2 (en) | CD86 variant with improved affinity for CTLA-4 | |
| EP1765872B1 (en) | Glycosylated immunoglobulin and immunoadhesin comprising the same | |
| US11505595B2 (en) | TIM-3 targeted heterodimeric fusion proteins containing IL-15/IL-15RA Fc-fusion proteins and TIM-3 antigen binding domains | |
| JP2019500862A (en) | Heterodimeric antibodies that bind to CD3 and PSMA | |
| IL263211B2 (en) | Compositions and methods related to engineered fc constructs | |
| US20230279071A1 (en) | LAG-3 TARGETED HETERODIMERIC FUSION PROTEINS CONTAINING IL-15/IL-15RA Fc-FUSION PROTEINS AND LAG-3 ANTIGEN BINDING DOMAINS | |
| JP2015536317A (en) | Immune regulatory protein | |
| KR102602539B1 (en) | Fc-FUSED HIGH AFFINITY IgE RECEPTOR α-CHAIN | |
| WO2015110930A1 (en) | Modified interleukin 21 receptor proteins | |
| CA3127144A1 (en) | Cd3-specific binding molecules | |
| US20240025968A1 (en) | LAG-3 TARGETED HETERODIMERIC FUSION PROTEINS CONTAINING IL-15/IL-15RA Fc-FUSION PROTEINS AND LAG-3 ANTIGEN BINDING DOMAINS | |
| CA3157042A1 (en) | Methods of cancer treatment using anti-ox40 antibodies in combination with anti-tigit antibodies | |
| US20240067727A1 (en) | Bifunctional anti-pd1/il-7 molecules | |
| US20230303648A1 (en) | Bifunctional molecules comprising an il-7 variant | |
| WO2024038198A1 (en) | Multi-domain binding molecules | |
| US20220332847A1 (en) | RECOMBINANT IgG Fc MULTIMERS FOR THE TREATMENT OF IMMUNE COMPLEX-MEDIATED KIDNEY DISORDERS | |
| WO2023073599A1 (en) | Engineered fc variants | |
| EP4301471A1 (en) | Application of apoptosis inhibitor 5 (api5) for epithelial restitution | |
| KR20130127366A (en) | Double-targeting protein comprising igg antibody specific to vegf and protein specific to dll4 and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15708271 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15708271 Country of ref document: EP Kind code of ref document: A1 |