WO2015009889A1 - Deuterated intedanib derivatives and their use for the treatment of proliferative disorders - Google Patents
Deuterated intedanib derivatives and their use for the treatment of proliferative disorders Download PDFInfo
- Publication number
- WO2015009889A1 WO2015009889A1 PCT/US2014/046951 US2014046951W WO2015009889A1 WO 2015009889 A1 WO2015009889 A1 WO 2015009889A1 US 2014046951 W US2014046951 W US 2014046951W WO 2015009889 A1 WO2015009889 A1 WO 2015009889A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- deuterium
- hydrogen
- alkyl
- formula
- Prior art date
Links
- 0 CC*C(*)(OC(C)=O)OP(OCc1ccccc1)(OCc1ccccc1)=O Chemical compound CC*C(*)(OC(C)=O)OP(OCc1ccccc1)(OCc1ccccc1)=O 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/30—Indoles; Hydrogenated indoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to carbon atoms of the hetero ring
- C07D209/32—Oxygen atoms
- C07D209/34—Oxygen atoms in position 2
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B59/00—Introduction of isotopes of elements into organic compounds ; Labelled organic compounds per se
- C07B59/002—Heterocyclic compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- ADME absorption, distribution, metabolism and/or excretion
- ADME limitation that affects many medicines is the formation of toxic or biologically reactive metabolites.
- some patients receiving the drug may experience toxicities, or the safe dosing of such drugs may be limited such that patients receive a suboptimal amount of the active agent.
- modifying dosing intervals or formulation approaches can help to reduce clinical adverse effects, but often the formation of such undesirable metabolites is intrinsic to the metabolism of the compound.
- a metabolic inhibitor will be co-administered with a drug that is cleared too rapidly.
- a drug that is cleared too rapidly.
- the FDA recommends that these drugs be co-dosed with ritonavir, an inhibitor of cytochrome P450 enzyme 3A4 (CYP3A4), the enzyme typically responsible for their metabolism (see Kempf, D.J. et al., Antimicrobial agents and chemotherapy, 1997, 41(3): 654-60).
- CYP3A4 cytochrome P450 enzyme 3A4
- Ritonavir causes adverse effects and adds to the pill burden for HIV patients who must already take a combination of different drugs.
- the CYP2D6 inhibitor quinidine has been added to dextromethorphan for the purpose of reducing rapid CYP2D6 metabolism of dextromethorphan in a treatment of pseudobulbar affect.
- Quinidine has unwanted side effects that greatly limit its use in potential combination therapy (see Wang, L et al., Clinical Pharmacology and Therapeutics, 1994, 56(6 Pt 1): 659-67; and FDA label for quinidine at www.accessdata.fda.gov).
- a potentially attractive strategy for improving a drug's metabolic properties is deuterium modification.
- Deuterium is a safe, stable, nonradioactive isotope of hydrogen. Compared to hydrogen, deuterium forms stronger bonds with carbon. In select cases, the increased bond strength imparted by deuterium can positively impact the ADME properties of a drug, creating the potential for improved drug efficacy, safety, and/or tolerability.
- the size and shape of deuterium are essentially identical to those of hydrogen, replacement of hydrogen by deuterium would not be expected to affect the biochemical potency and selectivity of the drug as compared to the original chemical entity that contains only hydrogen.
- This invention relates to novel derivatives of intedanib.
- This invention also provides compositions comprising a compound of this invention and the use of such compositions in methods of treating cancer, such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme; and/or fibrosis, such as pulmonary fibrosis, such as IPF.
- cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme
- fibrosis such as pulmonary fibrosis, such as IPF.
- Intedanib (BIBF-1120), also referred to as nintedanib, is an inhibitor of vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet derived growth factor receptor (PDGFR) developed by Boehringer-Ingelheim currently being investigated in phase III clinical trials for cancer as well as idiopathic pulmonary fibrosis (IPF).
- VEGFR vascular endothelial growth factor receptor
- FGFR fibroblast growth factor receptor
- PDGFR platelet derived growth factor receptor
- intedanib undergoes rapid first-pass metabolism via N-demethylation N- demethylintedanib (BIBF-1053) followed by ester hydrolysis.
- a metabolite formed from glucuronidation of the acid formed from the ester hydrolysis was detected in urine. Oxidation of the piperazine ring is also observed as a minor metabolite.
- treat means decrease, suppress, attenuate, diminish, arrest, or stabilize the development or progression of a disease (e.g., a disease or disorder delineated herein), lessen the severity of the disease or improve the symptoms associated with the disease.
- a disease e.g., a disease or disorder delineated herein
- Disease means any condition or disorder that damages or interferes with the normal function of a cell, tissue, or organ.
- alkyl refers to a monovalent saturated hydrocarbon group.
- Ci-C 6 alkyl is an alkyl having from 1 to 6 carbon atoms. An alkyl may be linear or branched. Examples of alkyl groups include methyl; ethyl; propyl, including n-propyl and isopropyl; butyl, including n-butyl, isobutyl, sec-butyl, and t-butyl; pentyl, including, for example, n-pentyl, isopentyl, and neopentyl; and hexyl, including, for example, n-hexyl and 2-methylpentyl.
- heterocycloalkyl refers to a monocyclic or bicyclic monovalent saturated or non-aromatic unsaturated hydrocarbon ring system having from one to three heteroatoms independently selected from nitrogen, oxygen and sulfur.
- 3-8 membered heterocycloalkyl refers to a heterocycloalkyl wherein the number of atoms in the ring is from 3-8.
- Bicyclic ring systems include fused, bridged, and spirocyclic ring systems.
- 3-8 membered heterocycloalkyl groups include azetidinyl, carbazoyl, dioxolanyl, perhydroazepinyl, piperidinyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, imidazolinyl, imidazolidinyl, dihydropyridinyl, tetrahydropyridinyl, oxazolinyl, oxazolidinyl, isoxazolidinyl, morpholinyl, thiazolinyl, thiazolidinyl, quinuclidinyl, isothiazolidinyl, tetrahydrofuryl, tetrahydropyranyl, thiamorpholinyl, thiamorpholinyl sulfox
- the nitrogen, phosphorus, carbon or sulfur atoms may be optionally oxidized to various oxidation states.
- the group -S(O) 0-2 - refers to -S-(sulfide), -S(0)-(sulfoxide), and -S0 2 - (sulfone) respectively.
- heterocycle refers to a monocyclic saturated or non-aromatic unsaturated hydrocarbon ring system having from one to three heteroatoms independently selected from nitrogen, oxygen and sulfur.
- heterocycle refers to a heterocycle wherein the number of atoms in the ring is from 3-8.
- Aryl refers to a monovalent aromatic hydrocarbon group having the stated number of carbon atoms (i.e., C 6 -Cio means from 5 to 14 carbon atoms).
- Typical aryl groups include groups derived from azulene, benzene, naphthalene, , and the like.
- the aryl group is phenyl or naphthyl, which may be 2-naphthyl or 1-naphthyl.
- any atom not specifically designated as a particular isotope is meant to represent any stable isotope of that atom.
- a position is designated specifically as “H” or “hydrogen”
- the position is understood to have hydrogen at its natural abundance isotopic composition.
- a position is designated specifically as “D” or “deuterium”
- the position is understood to have deuterium at an abundance that is at least 3000 times greater than the natural abundance of deuterium, which is 0.015% (i.e., at least 45% incorporation of deuterium).
- isotopic enrichment factor means the ratio between the isotopic abundance and the natural abundance of a specified isotope.
- a compound of this invention has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium
- incorporation at each designated deuterium atom at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5%) deuterium incorporation).
- isotopologue refers to a species in which the chemical structure differs from a specific compound of this invention only in the isotopic composition thereof.
- a compound represented by a particular chemical structure containing indicated deuterium atoms will also contain lesser amounts of isotopologues having hydrogen atoms at one or more of the designated deuterium positions in that structure.
- the relative amount of such isotopologues in a compound of this invention will depend upon a number of factors including the isotopic purity of deuterated reagents used to make the compound and the efficiency of incorporation of deuterium in the various synthesis steps used to prepare the compound.
- the relative amount of such isotopologues in toto will be less than 55% of the compound. In other embodiments, the relative amount of such isotopologues in toto will be less than 50%, less than 47.5%, less than 40%, less than 32.5%, less than 25%, less than 17.5%, less than 10%>, less than 5%, less than 3%, less than 1%, or less than 0.5%> of the compound.
- the invention also provides salts of the compounds of the invention.
- a salt of a compound of this invention is formed between an acid and a basic group of the compound, such as an amino functional group, or a base and an acidic group of the compound, such as a carboxyl functional group.
- the compound is a pharmaceutically acceptable acid addition salt.
- pharmaceutically acceptable refers to a component that is, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and other mammals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable salt means any non-toxic salt that, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention.
- pharmaceutically acceptable counterion is an ionic portion of a salt that is not toxic when released from the salt upon administration to a recipient.
- Acids commonly employed to form pharmaceutically acceptable salts include inorganic acids such as hydrogen bisulfide, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid and phosphoric acid, as well as organic acids such as para- toluenesulfonic acid, salicylic acid, tartaric acid, bitartaric acid, ascorbic acid, maleic acid, besylic acid, fumaric acid, gluconic acid, glucuronic acid, formic acid, glutamic acid, methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, lactic acid, oxalic acid, para-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid, as well as related inorganic and organic acids.
- organic acids such as para- toluenesulfonic acid, salicylic acid, tartaric acid, bitartaric acid, ascorbic acid
- salts thus include sulfate, pyrosulfate, bisulfate, sulfite, bisulfite, phosphate, monohydrogenphosphate, dihydrogenphosphate, metaphosphate, pyrophosphate, chloride, bromide, iodide, acetate, propionate, decanoate, caprylate, acrylate, formate, isobutyrate, caprate, heptanoate, propiolate, oxalate, malonate, succinate, suberate, sebacate, fumarate, maleate, butyne-l,4-dioate, hexyne-l,6-dioate, benzoate, chlorobenzoate, methylbenzoate, dinitrobenzoate, hydroxybenzoate, methoxybenzoate, phthalate, terephthalate, sulfonate, xylene sulfonate, phenylacetate, phenylpropionate
- the compounds of the present invention may contain an asymmetric carbon atom, for example, as the result of deuterium substitution or otherwise.
- compounds of this invention can exist as either individual enantiomers, or mixtures of the two enantiomers.
- a compound of the present invention may exist as either a racemic mixture or a scalemic mixture, or as individual respective stereoisomers that are substantially free from another possible stereoisomer.
- substantially free of other stereoisomers as used herein means less than 25% of other stereoisomers, preferably less than 10% of other stereoisomers, more preferably less than 5% of other stereoisomers and most preferably less than 2% of other stereoisomers are present.
- stable compounds refers to compounds which possess stability sufficient to allow for their manufacture and which maintain the integrity of the compound for a sufficient period of time to be useful for the purposes detailed herein (e.g., formulation into therapeutic products, intermediates for use in production of therapeutic compounds, isolatable or storable intermediate compounds, treating a disease or condition responsive to therapeutic agents).
- a group is "substituted with” a substituent when one or more hydrogen atoms of the group are replaced with a corresponding number of substituent atoms (if the substituent is an atom) or groups (if the substituent is a group).
- substituted with deuterium refers to the replacement of one or more hydrogen atoms with a corresponding number of deuterium atoms.
- variable may be referred to generally (e.g., "each R") or may be referred to specifically (e.g., R 1 , R 2 , R 3 , etc.). Unless otherwise indicated, when a variable is referred to generally, it is meant to include all specific embodiments of that particular variable.
- each of R 1 and R 3 is independently selected from CH 3 , CH 2 D, CHD 2 and CD 3 ;
- R 2 is CH 3 , CH 2 D, CHD 2 , CD 3 , hydrogen, or -C(0)OR 4 ;
- R 4 is -C(X 1 )(X 2 )OP(0)(OH) 2 or -C(X 1 )(X 2 )OC(0)X 7 ;
- X 7 is 3- to 8-membered heterocycloalkyl or Ci-C 6 alkyl optionally substituted with one or more R 9 ;
- each R 9 is independently OR 5 , NR 6 R 7 , or C 6 -Cio aryl wherein the C 6 -Cio aryl is optionally substituted with OH;
- R 5 is hydrogen, Ci-C 6 alkyl or Ci-C 6 alkyl-C(O);
- R 6 and R 7 are each independently hydrogen, Ci-C 6 alkyl or Ci-C 6 alkyl-C(O);
- X 1 and X 2 are each independently hydrogen, deuterium or Ci-C 6 alkyl
- each Y 1 is the same and is hydrogen or deuterium
- each Y 2 is the same and is hydrogen or deuterium
- each Y 3 is the same and is hydrogen or deuterium
- each Y 1 is hydrogen
- each Y 2 is hydrogen
- R 1 is CH 3
- R 3 is CH 3
- R 2 is not -C(0)OR 4 , then each Y 3 is deuterium.
- each of R 1 , R 3 and R 2 is independently selected from CH 3 and CD 3 .
- R 1 is CD 3 . In one aspect of this embodiment, R 3 is CD 3 . In another aspect of this embodiment, R 3 is CH 3 . In one example of any of the foregoing aspects, R 2 is CD 3 . In another example of any of the foregoing aspects, R 2 is CH 3 . In a more particular example of any of the foregoing aspects, R 2 is CD 3 and each Y 1 is deuterium. In another more particular example of any of the foregoing aspects, R 2 is CH 3 and each Y 1 is deuterium. In another more particular example of any of the foregoing aspects, R 2 is CD 3 and each Y 1 is hydrogen.
- R 2 is CH 3 and each Y 1 is hydrogen.
- R 1 is CH 3 .
- R 3 is CD 3 .
- R 3 is CH 3 .
- R 2 is CD 3 .
- R 2 is CH 3 .
- R 2 is CD 3 and each Y 1 is deuterium.
- R 2 is CH 3 and each Y 1 is deuterium.
- R 2 is CD 3 and each Y 1 is hydrogen.
- R 2 is CH 3 and each Y 1 is hydrogen.
- R 2 is CH 3 and each Y 1 is hydrogen.
- R 3 is CD 3 .
- R 2 is CD 3 .
- R 2 is CH 3 .
- each Y 1 is deuterium.
- each Y 1 is hydrogen.
- R 3 is CH 3 .
- R 2 is CD 3 .
- R 2 is CH 3 .
- each Y 1 is deuterium.
- each Y 1 is hydrogen.
- R 2 is CD 3 .
- each Y 1 is deuterium.
- each Y 1 is hydrogen.
- R 2 is CH 3 .
- each Y 1 is deuterium.
- each Y 1 is hydrogen.
- each Y 1 is hydrogen. In one aspect of this embodiment, each Y 2 is hydrogen. In another aspect of this embodiment, each Y 2 is deuterium.
- each Y 1 is deuterium.
- each Y 2 is hydrogen. In another aspect of this embodiment, each Y 2 is deuterium.
- R 2 is -C(0)OR 4 .
- R 4 is -C(X 1 )(X 2 )OP(0)(OH) 2 .
- X 1 and X 2 are each hydrogen. In another example of this aspect, X 1 and X 2 are each deuterium.
- R 4 is
- X 1 and X 2 are each hydrogen and X 7 is Ci-C 6 alkyl.
- X 1 and X 2 are each hydrogen and X 7 is CH 3 or C 2 H 5 .
- X 1 and X 2 are each deuterium and X 7 is Ci-C 6 alkyl.
- X 1 and X 2 are each deuterium and X 7 is CH 3 or C 2 H 5 .
- X 1 and X 2 are each hydrogen and X 7 is Ci-C 6 alkyl substituted with OR 5 .
- X 1 and X 2 are each hydrogen, R 5 is hydrogen and X 7 is Ci-C 6 alkyl substituted with OH such as CH 2 OH or CH(OH)CH 3 .
- X 1 and X 2 are each hydrogen
- R 5 is Ci-C 6 alkyl-C(O)
- X 7 is Ci-C 6 alkyl substituted with Ci-C 6 alkyl- C(0)0 such as CH 2 0(CO)CH 3 or CH(CH 3 )OC(0)CH 3 ).
- R 4 is -C(X 1 )(X 2 )OC(0)X 7
- X 1 and X 2 are each deuterium and X 7 is Ci-C 6 alkyl substituted with OR 5 .
- X 1 and X 2 are each deuterium
- R 5 is hydrogen
- X 7 is Ci-C 6 alkyl substituted with OH such as CH 2 OH or CH(OH)CH 3
- X 1 and X 2 are each deuterium
- R 5 is CH 3 C(0)
- X 7 is C C 6 alkyl substituted with C C 6 alkyl-C(0)0 such as CH 2 0(CO)CH 3 or CH(CH 3 )OC(0)CH 3 ).
- X 7 is Ci-C 6 alkyl substituted with NR 6 R 7 and_optionally with C 6 - C 10 aryl wherein the C 6 -Cio aryl is optionally substituted with OH.
- R 4 is -C(X 1 )(X 2 )OC(0)X 7
- X 1 and X 2 are each hydrogen and X 7 is Ci-C 6 alkyl substituted with NR 6 R 7 .
- X 1 and X 2 are each hydrogen
- R 6 and R 7 are each hydrogen
- X 7 is Ci-C 6 alkyl substituted with NH 2 such as CH 2 NH 2 or CH(NH 2 )CH 3 .
- X 1 and X 2 are each hydrogen, R 6 is hydrogen and R 7 is Ci-C 6 alkyl-C(O) and X 7 is Ci-C 6 alkyl substituted with Ci-C 6 alkyl-C(0)NH such as CH 2 NH(CO)CH 3 or CH(CH 3 )NHC(0)CH 3 ).
- X 1 and X 2 are each deuterium and X 7 is Ci-C 6 alkyl substituted with NR 6 R 7 .
- X 1 and X 2 are each deuterium
- R 6 and R 7 are each hydrogen
- X 7 is Ci-C 6 alkyl substituted with NH 2 such as CH 2 NH 2 or CH(NH 2 )CH 3 .
- X 1 and X 2 are each deuterium, R 6 is hydrogen and R 7 is Ci-C 6 alkyl-C(O) and X 7 is Ci-C 6 alkyl substituted with Ci-C 6 alkyl-C(0)NH such as CH 2 NH(CO)CH 3 or
- R 4 is -C(X 1 )(X 2 )OC(0)X 7
- X 1 and X 2 are each hydrogen and X 7 is Ci-C 6 alkyl substituted with NR 6 R 7 and with C 6 -Ci 0 aryl wherein the C 6 -Cio aryl is optionally substituted with OH.
- X 1 and X 2 are each hydrogen and X 7 is CH 3 or C 2 3 ⁇ 4 wherein X 7 is substituted either with (a) NH 2 and phenyl or (b) NH 2 and 4-hydroxyphenyl.
- X 1 and X 2 are each deuterium and X 7 is Ci-C 6 alkyl substituted with NR 6 R 7 and with C 6 -Cio aryl wherein the C 6 -Cio aryl is optionally substituted with OH.
- X 1 and X 2 are each deuterium and X 7 is CH 3 or C 2 H 5 wherein X 7 is substituted either with (a) NH 2 and phenyl or (b) NH 2 and 4-hydroxyphenyl.
- X 1 and X 2 are each hydrogen and X 7 is 3- to 8-membered heterocycloalkyl.
- X 1 and X 2 are each hydrogen and X 7 is pyrrolidinyl.
- the pyrrolydinyl may be, for example, 2-(S)-pyrrolidinyl or 2-(R)-pyrrolidinyl.
- X 1 and X 2 are each deuterium and X 7 is 3- to 8-membered heterocycloalkyl.
- X 1 and X 2 are each deuterium and X 7 is pyrrolidinyl.
- the pyrrolydinyl may be, for example, 2-(S)-pyrrolidinyl or 2-(R)-pyrroliydinyl.
- any atom not designated as deuterium in any of the embodiments, aspects, or examples set forth above is present at its natural isotopic abundance.
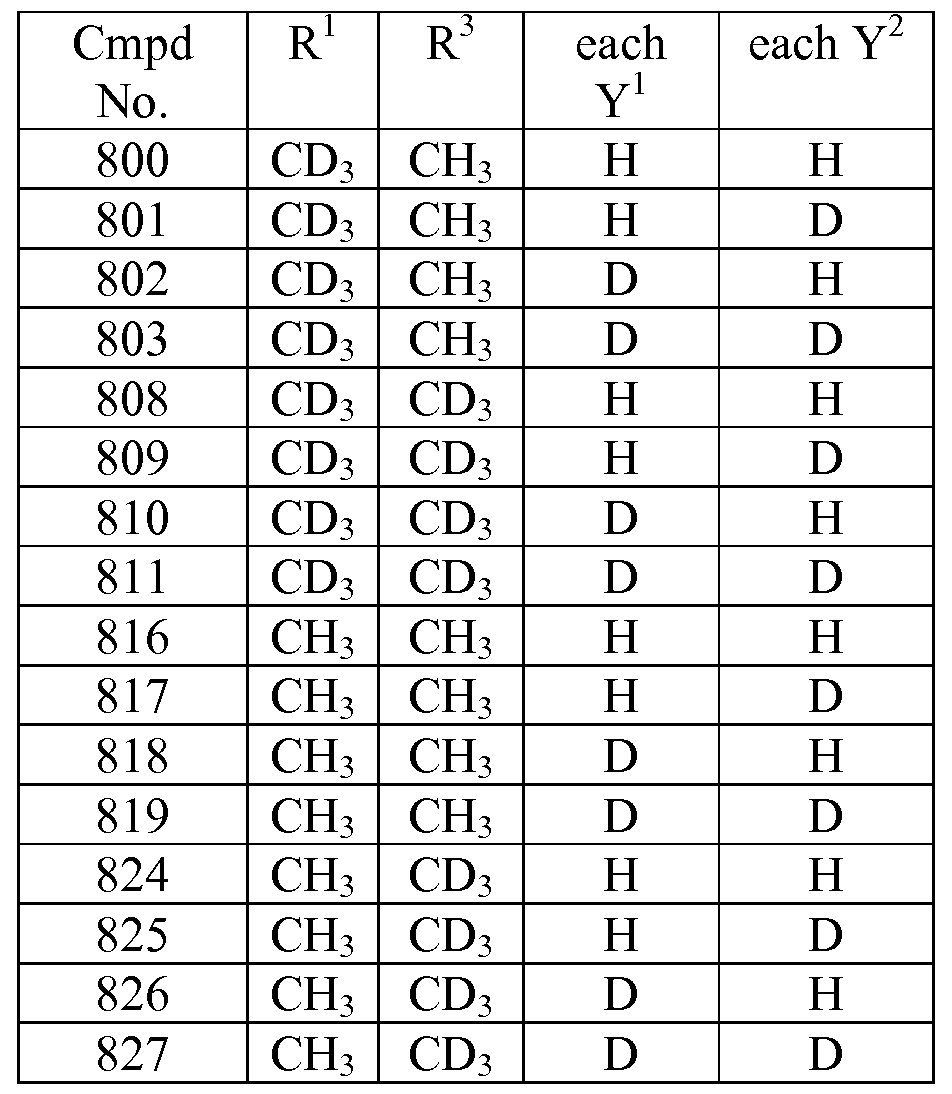
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 1 (below), wherein each Y 3 is hydrogen:
- any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 2 (below), wherein each Y 3 is deuterium:
- any atom not designated as deuterium is present at its natural isotopic abundance.
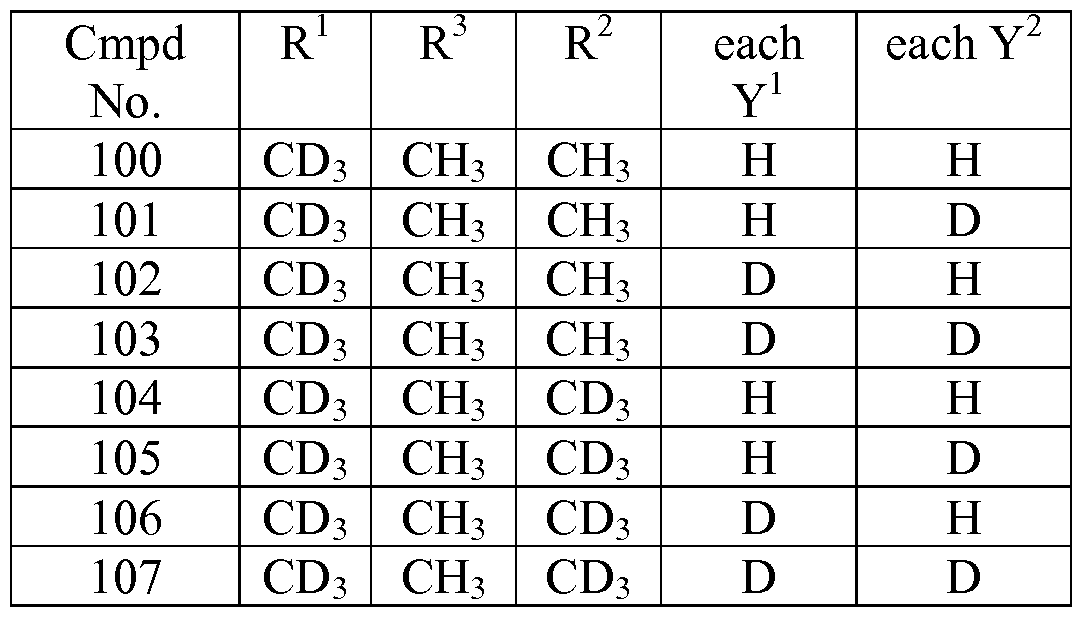
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 3 (below), wherein each Y 3 is hydrogen and R 2 is C(0)OCH 2 OP(0)(OH) 2 :
- Table 3 Exemplary Embodiments of Formula I or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 4 (below), wherein each Y 3 is deuterium and R 2 is C(0)OCH 2 OP(0)(OH) 2 :
- any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 5 (below), wherein each Y 3 is hydrogen and R 2 is C(0)OCH 2 OC(0)CH(CH 3 )OH:
- the compound is the (R) enantiomer of any one of the compounds in Table 5. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 5.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 6 (below), wherein each Y 3 is deuterium and R 2 is C(0)OCH 2 OC(0)CH(CH 3 )OH:
- any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is the (R) enantiomer of any one of the compounds in Table 6. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 6. [63] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 7 (below), wherein each Y 3 is hydrogen and R 2 is C(0)OCH 2 OC(0)CH(CH 3 )NH 2 :
- any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is the (R) enantiomer of any one of the compounds in Table 7. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 7.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 8 (below), wherein each Y 3 is deuterium and R 2 is C(0)OCH 2 OC(0)CH(CH 3 )NH 2 :
- Table 8 Exemplary Embodiments of Formula I or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is the (R) enantiomer of any one of the compounds in Table 8. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 8.
- the compound is selected from any one of the compounds (Cmpd) set forth in Table 9 (below), wherein each Y 3 is deuterium and R 2 is C(0)OCH 2 OC(0)CH(CH 2 C 6 H 5 )NH 2 :
- any atom not designated as deuterium is present at its natural isotopic abundance.
- the compound is the (R) enantiomer of any one of the compounds in Table 9. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 9.
- any atom not designated as deuterium in any of the embodiments, aspects, or examples set forth above is present at its natural isotopic abundance.
- Such methods can be carried out utilizing corresponding deuterated and optionally, other isotope-containing reagents and/or intermediates to synthesize the compounds delineated herein, or invoking standard synthetic protocols known in the art for introducing isotopic atoms to a chemical structure.
- 6a may be prepared from 10a, which is commercially available from CDN isotopes, by treating 10a with an alkyl iodide R 2 I and treating the resulting tertiary amine with HCI, as disclosed in U.S. patent publication 2009-0149399 .
- Intermediate 6b may be similarly prepared from 10b, which is prepared as described in J. Labelled Compounds and Radiopharmaceuticals, 25(4), 359-367, by treating 10b with R 2 I and treating the resulting tertiary amine with HCI.
- Intermediates 6c (where R 2 is either CH 3 or CD 3 , as discussed below) may be prepared from 10b, whjch is prepared as described above.
- 10b is treated sequentially with benzyl bromide, lithium aluminum hydride (or lithium aluminum deuteride) and H 2 , Pd/C to provide 6c where R 2 is CH 3 (when lithium aluminum hydride is used) or R 2 is CD 3 (when lithium aluminum deuteride is used).
- a compound of formula I wherein R 2 is H - obtained for example, as disclosed in Scheme 1 - is treated with 21, which may be obtained as disclosed in Scheme 6 below, to provide, after a deprotection step if necessary, the compound of formula I wherein R 2 is COOR 4 and R 4 is -C(X 1 )(X 2 )OC(0)X 7 .
- X 1 and X 2 of formula I are each hydrogen, and X 7 is Ci-C 6 alkyl optionally substituted with one or more R 9 .
- a compound of formula I wherein R 2 is H - obtained for example, as disclosed in Scheme 1 - is treated with 17, which may be obtained as disclosed in Scheme 7 below, to provide, after removal of the Cbz protecting group by treatment with hydrogen and Pd(OH) 2 /C, the compound of formula I wherein R 2 is COOR 4 and R 4 is -C(X 1 )(X 2 )OC(0)X 7 .
- X 1 and X 2 of formula I are each hydrogen
- X 7 is C1-C5 alkyl substituted with NH 2 .
- "Co" is intended to mean the absence of an alkyl substituent, so that for that particular embodiment 17 has the structure:
- Scheme 5 may be readily modified by replacing 17, for example, with 47: so as to provide a compound of formula I in which X is Ci-C 6 alkyl, such as ethyl, substituted with NR 6 R 7 and with C 6 -Ci 0 aryl.
- X is Ci-C 6 alkyl, such as ethyl, substituted with NR 6 R 7 and with C 6 -Ci 0 aryl.
- the C 6 -Ci 0 aryl may be optionally substituted with OH as disclosed herein.
- Compound 21 may be prepared by treating 18 with acid 22 to give 23; and reacting 23 with sulfuryl chloride to afford 21.
- Compound 17 may be prepared by treating chloromethyl chloroformate with NaSEt to provide 18; treating 18 with protected amino acid 19 to give 20; and reacting 20 with sulfuryl chloride to afford 17.
- a variety of naturally occurring amino acids may be envisioned for the formation of 18, such as valine, in which case the corresponding structure of 17 resulting from Scheme 7 is
- Scheme 8b depicts an alternate method for producing compounds of Formula I where R 2 is COOR 4 and R 4 is -C(X 1 )(X 2 )OP(0)(OH) 2 .
- a compound of Formula I wherein R 2 is hydrogen is treated with chloromethyl chloroformate to provide compound 26.
- Treatment with di-tert-butylphosphate potassium salt and tetrabutylammonium iodide (TBAI) provides compound 27.
- TBAI tetrabutylammonium iodide
- Removal of the t-butyl groups via treatment with trifluoroacetic acid provides compounds of Formula I wherein R 2 is COOR 4 and R 4 is -C(X 1 )(X 2 )OP(0)(OH) 2 .
- the disodium or calcium salt forms of the compound of formula I thus obtained may be accessed via treatment with sodium hydroxide or with calcium hydroxide or calcium acetate or calcium chloride.
- transformations and protecting group methodologies useful in synthesizing the applicable compounds are known in the art and include, for example, those described in Larock R, Comprehensive Organic Transformations, VCH Publishers (1989); Greene, TW et al, Protective Groups in Organic Synthesis, 3 rd Ed., John Wiley and Sons (1999); Fieser, L et al., Fieser and Fieser's Reagents for Organic Synthesis, John Wiley and Sons (1994); and Paquette, L, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995) and subsequent editions thereof.
- the invention also provides pharmaceutical compositions comprising an effective amount of a compound of Formula I (e.g., including any of the formulae herein), or a pharmaceutically acceptable salt of said compound; and a pharmaceutically acceptable carrier.
- a pharmaceutically acceptable carrier e.g., including any of the formulae herein
- the carrier(s) are "acceptable" in the sense of being compatible with the other ingredients of the formulation and, in the case of a pharmaceutically acceptable carrier, not deleterious to the recipient thereof in an amount used in the medicament.
- Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polyoxypropylene -block polymers, polyethylene glycol and wool fat. If required, the solubility and bioavailability of the compounds of the present invention in pharmaceutical compositions may be enhanced by methods well- known in the art.
- Another known method of enhancing bioavailability is the use of an amorphous form of a compound of this invention optionally formulated with a poloxamer, such as LUTROLTM and PLURONICTM (BASF Corporation), or block copolymers of ethylene oxide and propylene oxide. See United States patent 7,014,866; and United States patent publications 20060094744 and 20060079502.
- compositions of the invention include those suitable for oral, rectal, nasal, topical (including buccal and sublingual), vaginal or parenteral (including subcutaneous, intramuscular, intravenous and intradermal) administration.
- the compound of the formulae herein is administered transdermally (e.g., using a transdermal patch or iontophoretic techniques).
- Other formulations may conveniently be presented in unit dosage form, e.g., tablets, sustained release capsules, and in liposomes, and may be prepared by any methods well known in the art of pharmacy. See, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins, Baltimore, MD (20th ed. 2000).
- Such preparative methods include the step of bringing into association with the molecule to be administered ingredients such as the carrier that constitutes one or more accessory ingredients.
- ingredients such as the carrier that constitutes one or more accessory ingredients.
- the compositions are prepared by uniformly and intimately bringing into association the active ingredients with liquid carriers, liposomes or finely divided solid carriers, or both, and then, if necessary, shaping the product.
- compositions of the present invention suitable for oral administration may be presented as discrete units such as capsules, sachets, or tablets each containing a predetermined amount of the active ingredient; a powder or granules; a solution or a suspension in an aqueous liquid or a non-aqueous liquid; an oil-in-water liquid emulsion; a water-in-oil liquid emulsion; packed in liposomes; or as a bolus, etc.
- Soft gelatin capsules can be useful for containing such suspensions, which may beneficially increase the rate of compound absorption.
- carriers that are commonly used include lactose and corn starch.
- Lubricating agents such as magnesium stearate, are also typically added.
- useful diluents include lactose and dried cornstarch.
- aqueous suspensions are administered orally, the active ingredient is combined with emulsifying and suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
- compositions suitable for oral administration include lozenges comprising the ingredients in a flavored basis, usually sucrose and acacia or tragacanth; and pastilles comprising the active ingredient in an inert basis such as gelatin and glycerin, or sucrose and acacia.
- compositions suitable for parenteral administration include aqueous and nonaqueous sterile injection solutions which may contain anti-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents.
- the formulations may be presented in unit- dose or multi-dose containers, for example, sealed ampules and vials, and may be stored in a freeze dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example water for injections, immediately prior to use.
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets.
- Such injection solutions may be in the form, for example, of a sterile injectable aqueous or oleaginous suspension.
- This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example, as a solution in 1,3-butanediol.
- the acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions.
- These oil solutions or suspensions may also contain a long- chain alcohol diluent or dispersant.
- the pharmaceutical compositions of this invention may be administered in the form of suppositories for rectal administration. These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components.
- suitable non-irritating excipient include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
- compositions of this invention may be administered by nasal aerosol or inhalation.
- Such compositions are prepared according to techniques well- known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art. See, e.g.: Rabinowitz JD and Zaffaroni AC, US Patent 6,803,031, assigned to Alexza Molecular Delivery Corporation.
- Topical administration of the pharmaceutical compositions of this invention is especially useful when the desired treatment involves areas or organs readily accessible by topical application.
- the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier.
- Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax, and water.
- the pharmaceutical composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier.
- Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2- octyldodecanol, benzyl alcohol, and water.
- the pharmaceutical compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches and iontophoretic administration are also included in this invention.
- Application of the subject therapeutics may be local, so as to be administered at the site of interest.
- Various techniques can be used for providing the subject
- compositions at the site of interest such as injection, use of catheters, trocars, projectiles, pluronic gel, stents, sustained drug release polymers or other device which provides for internal access.
- the compounds of this invention may be incorporated into compositions for coating an implantable medical device, such as prostheses, artificial valves, vascular grafts, stents, or catheters.
- an implantable medical device such as prostheses, artificial valves, vascular grafts, stents, or catheters.
- Suitable coatings and the general preparation of coated implantable devices are known in the art and are exemplified in US Patents 6,099,562; 5,886,026; and 5,304,121.
- the coatings are typically biocompatible polymeric materials such as a hydrogel polymer,
- Coatings for invasive devices are to be included within the definition of pharmaceutically acceptable carrier, adjuvant or vehicle, as those terms are used herein.
- the invention provides a method of coating an implantable medical device comprising the step of contacting said device with the coating composition described above. It will be obvious to those skilled in the art that the coating of the device will occur prior to implantation into a mammal.
- the invention provides a method of impregnating an implantable drug release device comprising the step of contacting said drug release device with a compound or composition of this invention.
- Implantable drug release devices include, but are not limited to, biodegradable polymer capsules or bullets, non-degradable, diffusible polymer capsules and biodegradable polymer wafers.
- the invention provides an implantable medical device coated with a compound or a composition comprising a compound of this invention, such that said compound is therapeutically active.
- the invention provides an implantable drug release device impregnated with or containing a compound or a composition comprising a compound of this invention, such that said compound is released from said device and is therapeutically active.
- an implantable drug release device impregnated with or containing a compound or a composition comprising a compound of this invention, such that said compound is released from said device and is therapeutically active.
- a composition of this invention may be painted onto the organ, or a composition of this invention may be applied in any other convenient way.
- a composition of this invention further comprises a second therapeutic agent.
- the second therapeutic agent may be selected from any compound or therapeutic agent known to have or that demonstrates advantageous properties when administered with a compound having the same mechanism of action as intedanib.
- Such agents include those indicated as being useful in combination with intedanib, including but not limited to, those described in PCT patent publication
- the second therapeutic agent is an agent useful in the treatment or prevention of a disease or condition selected from cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme, and fibrosis, such as pulmonary fibrosis, such as IPF.
- cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme
- fibrosis such as pulmonary fibrosis, such as IPF.
- the second therapeutic agent is an anti-cancer agent.
- the invention provides separate dosage forms of a compound of this invention and one or more of any of the above-described second therapeutic agents, wherein the compound and second therapeutic agent are associated with one another.
- association with one another means that the separate dosage forms are packaged together or otherwise attached to one another such that it is readily apparent that the separate dosage forms are intended to be sold and administered together (within less than 24 hours of one another, consecutively or simultaneously).
- the compound of the present invention is present in an effective amount.
- effective amount refers to an amount which, when administered in a proper dosing regimen, is sufficient to treat the target disorder.
- an effective amount of a compound of this invention can range from 10 to 1200 mg, such as 20 to 1200 mg; 10 to 600 mg; or 20 to 600 mg.. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 1200 mg, such as 200 to 600 mg; 400 to 1200 mg; or 400 to 600 mg.. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 1000 mg, such as 400 to 1000 mg; 200 to 500 mg; or 400 to 500 mg..
- an effective amount of a compound of this invention can range from 20 to 1200 mg, administered once a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 400 to 1200 mg, administered once a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 400 to 1000 mg, administered once a day.
- an effective amount of a compound of this invention can range from 10 to 600 mg, administered twice a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 600 mg, administered twice a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 500 mg, administered twice a day.
- an effective amount of a compound of this invention can range from 10 to 600 mg, administered once a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 600 mg, administered once a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 500 mg, administered once a day.
- the dosage of the compound of this invention is selected to produce a target tumor/plasma ratio of > 0.33.
- Effective doses will also vary, as recognized by those skilled in the art, depending on the diseases treated, the severity of the disease, the route of administration, the sex, age and general health condition of the subject, excipient usage, the possibility of co- usage with other therapeutic treatments such as use of other agents and the judgment of the treating physician.
- an effective amount of the second therapeutic agent is between about 20% and 100% of the dosage normally utilized in a monotherapy regime using just that agent.
- an effective amount is between about 70% and 100% of the normal monotherapeutic dose.
- the normal monotherapeutic dosages of these second therapeutic agents are well known in the art. See, e.g., Wells et al, eds., Pharmacotherapy Handbook, 2nd Edition,
- the invention provides a method of inhibiting the activity of one or more of vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet derived growth factor receptor (PDGFR) in a cell, comprising contacting a cell with one or more compounds of Formula I herein.
- VEGFR vascular endothelial growth factor receptor
- FGFR fibroblast growth factor receptor
- PDGFR platelet derived growth factor receptor
- the type III receptor tyrosine kinase is selected from PDGFR-B, c- KIT and Flt3.
- the invention provides a method of treating cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme, and fibrosis, such as pulmonary fibrosis, such as IPF in a subject in need thereof, comprising the step of administering to the subject an effective amount of a compound or a composition of this invention.
- cancer such as colorectal cancer, small cell lung cancer (SCLC) and acute myeloid leukemia (AML).
- Identifying a subject in need of such treatment can be in the judgment of a subject or a health care professional and can be subjective (e.g. opinion) or objective (e.g.
- the subject is a patient.
- any of the above methods of treatment comprises the further step of co-administering to the subject in need thereof one or more second therapeutic agents.
- the choice of second therapeutic agent may be made from any second therapeutic agent known to be useful for co-administration with intedanib.
- the choice of second therapeutic agent is also dependent upon the particular disease or condition to be treated. Examples of second therapeutic agents that may be employed in the methods of this invention are those set forth above for use in combination
- compositions comprising a compound of this invention and a second therapeutic agent.
- the method of treating a cancer in a subject with a compound or composition of this invention comprises the additional step of co-administering to the subject in need thereof a second anti-cancer agent or subjecting the subject to radiation therapy.
- the second anti-cancer agent is selected from
- the second anti-cancer agent is bevacizumab and the cancer is selected from non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme.
- co-administered means that the second therapeutic agent may be administered together with a compound of this invention as part of a single dosage form (such as a composition of this invention comprising a compound of the invention and an second therapeutic agent as described above) or as separate, multiple dosage forms.
- the additional agent may be administered prior to, consecutively with, or following the administration of a compound of this invention.
- both the compounds of this invention and the second therapeutic agent(s) are administered by conventional methods.
- composition of this invention comprising both a compound of the invention and a second therapeutic agent, to a subject does not preclude the separate administration of that same therapeutic agent, any other second therapeutic agent or any compound of this invention to said subject at another time during a course of treatment.
- the effective amount of the compound of this invention is less than its effective amount would be where the second therapeutic agent is not
- the effective amount of the second therapeutic agent is less than its effective amount would be where the compound of this invention is not administered. In this way, undesired side effects associated with high doses of either agent may be minimized.
- Other potential advantages including without limitation improved dosing regimens and/or reduced drug cost) will be apparent to those of skill in the art.
- the invention provides a method of treating a disease or condition associated with or complicated with immune suppression in a subject in need thereof, comprising the step of administering to the subject an effective amount of a compound or a composition of this invention.
- the invention provides the use of a compound of Formula I alone or together with one or more of the above-described second therapeutic agents in the manufacture of a medicament, either as a single composition or as separate dosage forms, for treatment or prevention in a subject of a disease, disorder or symptom set forth above.
- Another aspect of the invention is a compound of Formula I for use in the treatment or prevention in a subject of a disease, disorder or symptom thereof delineated herein.
- Microsomal Assay Human liver microsomes (20 mg/mL) are obtained from Xenotech, LLC (Lenexa, KS). ⁇ -nicotinamide adenine dinucleotide phosphate, reduced form (NADPH), magnesium chloride (MgCl 2 ), and dimethyl sulfoxide (DMSO) are purchased from Sigma- Aldrich.
- 7.5 mM stock solutions of test compounds are prepared in DMSO.
- the 7.5 mM stock solutions are diluted to 12.5-50 ⁇ in acetonitrile (ACN).
- ACN acetonitrile
- the 20 mg/mL human liver microsomes are diluted to 0.625 mg/mL in 0.1 M potassium phosphate buffer, pH 7.4, containing 3 mM MgCl 2 .
- the diluted microsomes are added to wells of a 96-well deep-well polypropylene plate in triplicate.
- a 10 aliquot of the 12.5-50 ⁇ test compound is added to the microsomes and the mixture is pre-warmed for 10 minutes. Reactions are initiated by addition of pre-warmed NADPH solution.
- the final reaction volume is 0.5 mL and contains 0.5 mg/mL human liver microsomes, 0.25-1.0 ⁇ test compound, and 2 mM NADPH in 0.1 M potassium phosphate buffer, pH 7.4, and 3 mM MgCl 2 .
- the reaction mixtures are incubated at 37 °C, and 50 ⁇ , aliquots are removed at 0, 5, 10, 20, and 30 minutes and added to shallow-well 96-well plates which contain 50 ⁇ ⁇ of ice-cold ACN with internal standard to stop the reactions.
- the plates are stored at 4 °C for 20 minutes after which 100 ⁇ ⁇ of water is added to the wells of the plate before centrifugation to pellet precipitated proteins.
Abstract
The present invention provides a deuterated compound of Formula I: or a pharmaceutically acceptable salt thereof, wherein the variables shown in Formula I are as defined in the specification. The compound of Formula I is useful for the treatment of cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme, and/or fibrosis, such as pulmonary fibrosis, including IPF.
Description
DEUTERATED INTEDANIB DERIVATIVES
AND THEIR USE FOR THE TREATMENT OF PROLIFERATIVE DISORDERS
Cross-Reference to Related Applications
This application claims the benefit of U.S. Provisional Application Serial No. 61/847,723, filed July 18, 2013. The disclosure of the prior application is considered part of (and is incorporated by reference in) the disclosure of this application.
Background of the Invention
[1] Many current medicines suffer from poor absorption, distribution, metabolism and/or excretion (ADME) properties that prevent their wider use or limit their use in certain indications. Poor ADME properties are also a major reason for the failure of drug candidates in clinical trials. While formulation technologies and prodrug strategies can be employed in some cases to improve certain ADME properties, these approaches often fail to address the underlying ADME problems that exist for many drugs and drug candidates. One such problem is rapid metabolism that causes a number of drugs, which otherwise would be highly effective in treating a disease, to be cleared too rapidly from the body. A possible solution to rapid drug clearance is frequent or high dosing to attain a sufficiently high plasma level of drug. This, however, introduces a number of potential treatment problems such as poor patient compliance with the dosing regimen, side effects that become more acute with higher doses, and increased cost of treatment. A rapidly metabolized drug may also expose patients to undesirable toxic or reactive metabolites.
[2] Another ADME limitation that affects many medicines is the formation of toxic or biologically reactive metabolites. As a result, some patients receiving the drug may experience toxicities, or the safe dosing of such drugs may be limited such that patients receive a suboptimal amount of the active agent. In certain cases, modifying dosing intervals or formulation approaches can help to reduce clinical adverse effects, but often the formation of such undesirable metabolites is intrinsic to the metabolism of the compound.
[3] In some select cases, a metabolic inhibitor will be co-administered with a drug that is cleared too rapidly. Such is the case with the protease inhibitor class of drugs that are used to treat HIV infection. The FDA recommends that these drugs be co-dosed with
ritonavir, an inhibitor of cytochrome P450 enzyme 3A4 (CYP3A4), the enzyme typically responsible for their metabolism (see Kempf, D.J. et al., Antimicrobial agents and chemotherapy, 1997, 41(3): 654-60). Ritonavir, however, causes adverse effects and adds to the pill burden for HIV patients who must already take a combination of different drugs. Similarly, the CYP2D6 inhibitor quinidine has been added to dextromethorphan for the purpose of reducing rapid CYP2D6 metabolism of dextromethorphan in a treatment of pseudobulbar affect. Quinidine, however, has unwanted side effects that greatly limit its use in potential combination therapy (see Wang, L et al., Clinical Pharmacology and Therapeutics, 1994, 56(6 Pt 1): 659-67; and FDA label for quinidine at www.accessdata.fda.gov).
[4] In general, combining drugs with cytochrome P450 inhibitors is not a satisfactory strategy for decreasing drug clearance. The inhibition of a CYP enzyme's activity can affect the metabolism and clearance of other drugs metabolized by that same enzyme. CYP inhibition can cause other drugs to accumulate in the body to toxic levels.
[5] A potentially attractive strategy for improving a drug's metabolic properties is deuterium modification. In this approach, one attempts to slow the CYP -mediated metabolism of a drug or to reduce the formation of undesirable metabolites by replacing one or more hydrogen atoms with deuterium atoms. Deuterium is a safe, stable, nonradioactive isotope of hydrogen. Compared to hydrogen, deuterium forms stronger bonds with carbon. In select cases, the increased bond strength imparted by deuterium can positively impact the ADME properties of a drug, creating the potential for improved drug efficacy, safety, and/or tolerability. At the same time, because the size and shape of deuterium are essentially identical to those of hydrogen, replacement of hydrogen by deuterium would not be expected to affect the biochemical potency and selectivity of the drug as compared to the original chemical entity that contains only hydrogen.
[6] Over the past 35 years, the effects of deuterium substitution on the rate of metabolism have been reported for a very small percentage of approved drugs (see, e.g., Blake, MI et al, J Pharm Sci, 1975, 64:367-91; Foster, AB, Adv Drug Res 1985, 14: 1-40 ("Foster"); Kushner, DJ et al, Can J Physiol Pharmacol 1999, 79-88; Fisher, MB et al, Curr Opin Drug Discov Devel, 2006, 9: 101-09 ("Fisher")). The results have been variable and unpredictable. For some compounds deuteration caused decreased
metabolic clearance in vivo. For others, there was no change in metabolism. Still others demonstrated increased metabolic clearance. The variability in deuterium effects has also led experts to question or dismiss deuterium modification as a viable drug design strategy for inhibiting adverse metabolism (see Foster at p. 35 and Fisher at p. 101).
[7] The effects of deuterium modification on a drug's metabolic properties are not predictable even when deuterium atoms are incorporated at known sites of metabolism. Only by actually preparing and testing a deuterated drug can one determine if and how the rate of metabolism will differ from that of its non-deuterated counterpart. See, for example, Fukuto et al. (J. Med. Chem. 1991, 34, 2871-76). Many drugs have multiple sites where metabolism is possible. The site(s) where deuterium substitution is required and the extent of deuteration necessary to see an effect on metabolism, if any, will be different for each drug.
[8] This invention relates to novel derivatives of intedanib. This invention also provides compositions comprising a compound of this invention and the use of such compositions in methods of treating cancer, such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme; and/or fibrosis, such as pulmonary fibrosis, such as IPF.
[9] Intedanib (BIBF-1120), also referred to as nintedanib, is an inhibitor of vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet derived growth factor receptor (PDGFR) developed by Boehringer-Ingelheim currently being investigated in phase III clinical trials for cancer as well as idiopathic pulmonary fibrosis (IPF). A 52 week phase II trial for IPF indicated that treatment with oral intedanib 150 mg BID was associated with a 68% reduction in the rate of annual decline in forced vital capacity. As discussed in Xenobiotica, 2011; 41(4): 297-311, intedanib undergoes rapid first-pass metabolism via N-demethylation N- demethylintedanib (BIBF-1053) followed by ester hydrolysis. A metabolite formed from glucuronidation of the acid formed from the ester hydrolysis was detected in urine. Oxidation of the piperazine ring is also observed as a minor metabolite.
[10] There are no deuterated examples of intedanib reported in the literature; however one example is commercially available:
(commercially available).
[11] Despite the potential beneficial activities of intedanib, there is a continuing need for new compounds to treat the aforementioned diseases and conditions.
Definitions
[12] The term "treat" means decrease, suppress, attenuate, diminish, arrest, or stabilize the development or progression of a disease (e.g., a disease or disorder delineated herein), lessen the severity of the disease or improve the symptoms associated with the disease.
[13] "Disease" means any condition or disorder that damages or interferes with the normal function of a cell, tissue, or organ.
[14] "The term "alkyl" refers to a monovalent saturated hydrocarbon group. Ci-C 6 alkyl is an alkyl having from 1 to 6 carbon atoms. An alkyl may be linear or branched. Examples of alkyl groups include methyl; ethyl; propyl, including n-propyl and isopropyl; butyl, including n-butyl, isobutyl, sec-butyl, and t-butyl; pentyl, including, for example, n-pentyl, isopentyl, and neopentyl; and hexyl, including, for example, n-hexyl and 2-methylpentyl.
[15] The term "heterocycloalkyl" refers to a monocyclic or bicyclic monovalent saturated or non-aromatic unsaturated hydrocarbon ring system having from one to three
heteroatoms independently selected from nitrogen, oxygen and sulfur. The term "3-8 membered heterocycloalkyl" refers to a heterocycloalkyl wherein the number of atoms in the ring is from 3-8. Bicyclic ring systems include fused, bridged, and spirocyclic ring systems. More particular examples of 3-8 membered heterocycloalkyl groups include azetidinyl, carbazoyl, dioxolanyl, perhydroazepinyl, piperidinyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolidinyl, 4-piperidonyl, pyrrolidinyl, pyrazolidinyl, imidazolinyl, imidazolidinyl, dihydropyridinyl, tetrahydropyridinyl, oxazolinyl, oxazolidinyl, isoxazolidinyl, morpholinyl, thiazolinyl, thiazolidinyl, quinuclidinyl, isothiazolidinyl, tetrahydrofuryl, tetrahydropyranyl, thiamorpholinyl, thiamorpholinyl sulfoxide, thiamorpholinyl sulfone, dioxaphospholanyl and
tetrahydrofuranyl. In the above heterocycloalkyl substituents, the nitrogen, phosphorus, carbon or sulfur atoms may be optionally oxidized to various oxidation states. In a specific example, the group -S(O)0-2-, refers to -S-(sulfide), -S(0)-(sulfoxide), and -S02- (sulfone) respectively.
[16] The term "heterocycle" refers to a monocyclic saturated or non-aromatic unsaturated hydrocarbon ring system having from one to three heteroatoms independently selected from nitrogen, oxygen and sulfur. The term "3-8 membered heterocycle" refers to a heterocycle wherein the number of atoms in the ring is from 3-8.
[17] "Aryl" refers to a monovalent aromatic hydrocarbon group having the stated number of carbon atoms (i.e., C6-Cio means from 5 to 14 carbon atoms). Typical aryl groups include groups derived from azulene, benzene, naphthalene, , and the like. In a specific embodiment, the aryl group is phenyl or naphthyl, which may be 2-naphthyl or 1-naphthyl.
[18] It will be recognized that some variation of natural isotopic abundance occurs in a synthesized compound depending upon the origin of chemical materials used in the synthesis. Thus, a preparation of intedanib will inherently contain small amounts of deuterated isotopologues. The concentration of naturally abundant stable hydrogen and carbon isotopes, notwithstanding this variation, is small and immaterial as compared to the degree of stable isotopic substitution of compounds of this invention. See, for instance, Wada, E et al, Seikagaku, 1994, 66: 15; Gannes, LZ et al, Comp Biochem Physiol Mol Integr Physiol, 1998, 119:725.
[19] In the compounds of this invention any atom not specifically designated as a particular isotope is meant to represent any stable isotope of that atom. Unless otherwise stated, when a position is designated specifically as "H" or "hydrogen", the position is understood to have hydrogen at its natural abundance isotopic composition. Also unless otherwise stated, when a position is designated specifically as "D" or "deuterium", the position is understood to have deuterium at an abundance that is at least 3000 times greater than the natural abundance of deuterium, which is 0.015% (i.e., at least 45% incorporation of deuterium).
[20] The term "isotopic enrichment factor" as used herein means the ratio between the isotopic abundance and the natural abundance of a specified isotope.
[21] In other embodiments, a compound of this invention has an isotopic enrichment factor for each designated deuterium atom of at least 3500 (52.5% deuterium
incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), at least 4500 (67.5% deuterium incorporation), at least 5000 (75% deuterium), at least 5500 (82.5% deuterium incorporation), at least 6000 (90% deuterium incorporation), at least 6333.3 (95% deuterium incorporation), at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5%) deuterium incorporation).
[22] The term "isotopologue" refers to a species in which the chemical structure differs from a specific compound of this invention only in the isotopic composition thereof.
[23] The term "compound," when referring to a compound of this invention, refers to a collection of molecules having an identical chemical structure, except that there may be isotopic variation among the constituent atoms of the molecules. Thus, it will be clear to those of skill in the art that a compound represented by a particular chemical structure containing indicated deuterium atoms, will also contain lesser amounts of isotopologues having hydrogen atoms at one or more of the designated deuterium positions in that structure. The relative amount of such isotopologues in a compound of this invention will depend upon a number of factors including the isotopic purity of deuterated reagents used to make the compound and the efficiency of incorporation of deuterium in the various synthesis steps used to prepare the compound. However, as set forth above the relative amount of such isotopologues in toto will be less than 55% of the compound. In
other embodiments, the relative amount of such isotopologues in toto will be less than 50%, less than 47.5%, less than 40%, less than 32.5%, less than 25%, less than 17.5%, less than 10%>, less than 5%, less than 3%, less than 1%, or less than 0.5%> of the compound.
[24] The invention also provides salts of the compounds of the invention.
[25] A salt of a compound of this invention is formed between an acid and a basic group of the compound, such as an amino functional group, or a base and an acidic group of the compound, such as a carboxyl functional group. According to another
embodiment, the compound is a pharmaceutically acceptable acid addition salt.
[26] The term "pharmaceutically acceptable," as used herein, refers to a component that is, within the scope of sound medical judgment, suitable for use in contact with the tissues of humans and other mammals without undue toxicity, irritation, allergic response and the like, and are commensurate with a reasonable benefit/risk ratio. A
"pharmaceutically acceptable salt" means any non-toxic salt that, upon administration to a recipient, is capable of providing, either directly or indirectly, a compound of this invention. A "pharmaceutically acceptable counterion" is an ionic portion of a salt that is not toxic when released from the salt upon administration to a recipient.
[27] Acids commonly employed to form pharmaceutically acceptable salts include inorganic acids such as hydrogen bisulfide, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid and phosphoric acid, as well as organic acids such as para- toluenesulfonic acid, salicylic acid, tartaric acid, bitartaric acid, ascorbic acid, maleic acid, besylic acid, fumaric acid, gluconic acid, glucuronic acid, formic acid, glutamic acid, methanesulfonic acid, ethanesulfonic acid, benzenesulfonic acid, lactic acid, oxalic acid, para-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid, as well as related inorganic and organic acids. Such
pharmaceutically acceptable salts thus include sulfate, pyrosulfate, bisulfate, sulfite, bisulfite, phosphate, monohydrogenphosphate, dihydrogenphosphate, metaphosphate, pyrophosphate, chloride, bromide, iodide, acetate, propionate, decanoate, caprylate, acrylate, formate, isobutyrate, caprate, heptanoate, propiolate, oxalate, malonate, succinate, suberate, sebacate, fumarate, maleate, butyne-l,4-dioate, hexyne-l,6-dioate, benzoate, chlorobenzoate, methylbenzoate, dinitrobenzoate, hydroxybenzoate,
methoxybenzoate, phthalate, terephthalate, sulfonate, xylene sulfonate, phenylacetate, phenylpropionate, phenylbutyrate, citrate, lactate, β-hydroxybutyrate, glycolate, maleate, tartrate, methanesulfonate, propanesulfonate, naphthalene- 1 -sulfonate, naphthalene-2- sulfonate, mandelate and other salts. In one embodiment, pharmaceutically acceptable acid addition salts include those formed with mineral acids such as hydrochloric acid and hydrobromic acid, and especially those formed with organic acids such as maleic acid.
[28] The compounds of the present invention (e.g., compounds of Formula I), may contain an asymmetric carbon atom, for example, as the result of deuterium substitution or otherwise. As such, compounds of this invention can exist as either individual enantiomers, or mixtures of the two enantiomers. Accordingly, a compound of the present invention may exist as either a racemic mixture or a scalemic mixture, or as individual respective stereoisomers that are substantially free from another possible stereoisomer. The term "substantially free of other stereoisomers" as used herein means less than 25% of other stereoisomers, preferably less than 10% of other stereoisomers, more preferably less than 5% of other stereoisomers and most preferably less than 2% of other stereoisomers are present. Methods of obtaining or synthesizing an individual enantiomer for a given compound are known in the art and may be applied as practicable to final compounds or to starting material or intermediates.
[29] Unless otherwise indicated, when a disclosed compound is named or depicted by a structure without specifying the stereochemistry and has one or more chiral centers, it is understood to represent all possible stereoisomers of the compound.
[30] The term "stable compounds," as used herein, refers to compounds which possess stability sufficient to allow for their manufacture and which maintain the integrity of the compound for a sufficient period of time to be useful for the purposes detailed herein (e.g., formulation into therapeutic products, intermediates for use in production of therapeutic compounds, isolatable or storable intermediate compounds, treating a disease or condition responsive to therapeutic agents).
[31] "D" and "d" both refer to deuterium. "dx_y" refers to substitution with from x to y number of deuterium atoms. "Stereoisomer" refers to both enantiomers and
diastereomers. "Tert" and "t-" each refer to tertiary. "US" refers to the United States of America.
[32] A group is "substituted with" a substituent when one or more hydrogen atoms of the group are replaced with a corresponding number of substituent atoms (if the substituent is an atom) or groups (if the substituent is a group). For example, "substituted with deuterium" refers to the replacement of one or more hydrogen atoms with a corresponding number of deuterium atoms.
[33] Throughout this specification, a variable may be referred to generally (e.g., "each R") or may be referred to specifically (e.g., R1, R2, R3, etc.). Unless otherwise indicated, when a variable is referred to generally, it is meant to include all specific embodiments of that particular variable.
Therapeutic Compounds
The present invention in one embodiment provides a compound of Formula I
Formula I,
or a pharmaceutically acceptable salt thereof, wherein:
each of R1 and R3 is independently selected from CH3, CH2D, CHD2 and CD3;
R2 is CH3, CH2D, CHD2, CD3, hydrogen, or -C(0)OR4;
R4 is -C(X1)(X2)OP(0)(OH)2 or -C(X1)(X2)OC(0)X7;
X7 is 3- to 8-membered heterocycloalkyl or Ci-C6 alkyl optionally substituted with one or more R9;
each R9 is independently OR5, NR6R7, or C6-Cio aryl wherein the C6-Cio aryl is optionally substituted with OH;
R5 is hydrogen, Ci-C6 alkyl or Ci-C6 alkyl-C(O);
R6 and R7 are each independently hydrogen, Ci-C6 alkyl or Ci-C6 alkyl-C(O);
X1 and X2 are each independently hydrogen, deuterium or Ci-C6 alkyl;
or X1 and X2 taken together with the carbon they are joined to forms a C3-Cg carbocycle or a 3- to 8-membered heterocycle containing one or two heteroatoms independently selected from S, O, and N;
each Y1 is the same and is hydrogen or deuterium;
each Y2 is the same and is hydrogen or deuterium;
and
each Y3 is the same and is hydrogen or deuterium;
provided that when each Y1 is hydrogen, each Y2 is hydrogen; R1 is CH3; R3 is CH3; and R2 is not -C(0)OR4, then each Y3 is deuterium.
[35] In one set of embodiments of the compound of Formula I, each of R1, R3 and R2 is independently selected from CH3 and CD3.
[36] In one embodiment of the compound of Formula I, R1 is CD3. In one aspect of this embodiment, R3 is CD3. In another aspect of this embodiment, R3 is CH3. In one example of any of the foregoing aspects, R2 is CD3. In another example of any of the foregoing aspects, R2 is CH3. In a more particular example of any of the foregoing aspects, R2 is CD3 and each Y1 is deuterium. In another more particular example of any of the foregoing aspects, R2 is CH3 and each Y1 is deuterium. In another more particular example of any of the foregoing aspects, R2 is CD3 and each Y1 is hydrogen. In another more particular example of any of the foregoing aspects, R2 is CH3 and each Y1 is hydrogen. In another embodiment of the compound of Formula I, R1 is CH3. In one aspect of this embodiment, R3 is CD3. In another aspect of this embodiment, R3 is CH3. In one example of any of the foregoing aspects, R2 is CD3. In another example of any of
the foregoing aspects, R2 is CH3. In a more particular example of any of the foregoing aspects, R2 is CD3 and each Y1 is deuterium. In another more particular example of any of the foregoing aspects, R2 is CH3 and each Y1 is deuterium. In another more particular example of any of the foregoing aspects, R2 is CD3 and each Y1 is hydrogen. In another more particular example of any of the foregoing aspects, R2 is CH3 and each Y1 is hydrogen.
[37] In one embodiment of the compound of Formula I, R3 is CD3. In one aspect of this embodiment, R2 is CD3. In another aspect of this embodiment, R2 is CH3. In one example of any of the foregoing aspects, each Y1 is deuterium. In another example of any of the foregoing aspects, each Y1 is hydrogen.
[38] In another embodiment of the compound of Formula I, R3 is CH3. In one aspect of this embodiment, R2 is CD3. In another aspect of this embodiment, R2 is CH3. In one example of any of the foregoing aspects, each Y1 is deuterium. In another example of any of the foregoing aspects, each Y1 is hydrogen.
[39] In one embodiment of the compound of Formula I, R2 is CD3. In one aspect of this embodiment, each Y1 is deuterium. In another aspect of this embodiment, each Y1 is hydrogen.
[40] In one embodiment of the compound of Formula I, R2 is CH3. In one aspect of this embodiment, each Y1 is deuterium. In another aspect of this embodiment, each Y1 is hydrogen.
[41] In one embodiment of the compound of Formula I, each Y1 is hydrogen. In one aspect of this embodiment, each Y2 is hydrogen. In another aspect of this embodiment, each Y2 is deuterium.
[42] In one embodiment of the compound of Formula I, each Y1 is deuterium. In one aspect of this embodiment, each Y2 is hydrogen. In another aspect of this embodiment, each Y2 is deuterium.
[43] In one embodiment of the compound of Formula I, R2 is -C(0)OR4.
[44] In one aspect of this embodiment, R4 is -C(X1)(X2)OP(0)(OH)2.
[45] In an example of this aspect, X1 and X2 are each hydrogen. In another example of
this aspect, X1 and X2 are each deuterium.
[46] In another aspect of the embodiment where R2 is -C(0)OR4, R4 is
-C(X1)(X2)OC(0)X7.
[47] In an example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each hydrogen and X7 is Ci-C6 alkyl. In more specific examples, X1 and X2 are each hydrogen and X7 is CH3 or C2H5. In another example of this aspect, X1 and X2 are each deuterium and X7 is Ci-C6 alkyl. In more specific examples, X1 and X2 are each deuterium and X7 is CH3 or C2H5.
[48] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each hydrogen and X7 is Ci-C6 alkyl substituted with OR5. In more specific examples, X1 and X2 are each hydrogen, R5 is hydrogen and X7 is Ci-C6 alkyl substituted with OH such as CH2OH or CH(OH)CH3. In other more specific examples, X1 and X2 are each hydrogen, R5 is Ci-C6 alkyl-C(O) and X7 is Ci-C6 alkyl substituted with Ci-C6 alkyl- C(0)0 such as CH20(CO)CH3 or CH(CH3)OC(0)CH3).
[49] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each deuterium and X7 is Ci-C6 alkyl substituted with OR5. In more specific examples, X1 and X2 are each deuterium, R5 is hydrogen and X7 is Ci-C6 alkyl substituted with OH such as CH2OH or CH(OH)CH3. In other more specific examples, X1 and X2 are each deuterium, R5 is CH3C(0) and X7 is C C6 alkyl substituted with C C6 alkyl-C(0)0 such as CH20(CO)CH3 or CH(CH3)OC(0)CH3).
[50] In other aspects, X7 is Ci-C6 alkyl substituted with NR6R7 and_optionally with C6- C10 aryl wherein the C6-Cio aryl is optionally substituted with OH. Thus, in another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each hydrogen and X7 is Ci-C6 alkyl substituted with NR6R7. In more specific examples, X1 and X2 are each hydrogen, R6 and R7 are each hydrogen and X7 is Ci-C6 alkyl substituted with NH2 such as CH2NH2 or CH(NH2)CH3. In other more specific examples, X1 and X2 are each hydrogen, R6 is hydrogen and R7 is Ci-C6 alkyl-C(O) and X7 is Ci-C6 alkyl substituted with Ci-C6 alkyl-C(0)NH such as CH2NH(CO)CH3 or CH(CH3)NHC(0)CH3).
[51] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each deuterium and X7 is Ci-C6 alkyl substituted with NR6R7. In more specific examples, X1 and X2 are each deuterium, R6 and R7 are each hydrogen and X7 is Ci-C6 alkyl
substituted with NH2 such as CH2NH2 or CH(NH2)CH3. In other more specific examples, X1 and X2 are each deuterium, R6 is hydrogen and R7 is Ci-C6 alkyl-C(O) and X7 is Ci-C6 alkyl substituted with Ci-C6 alkyl-C(0)NH such as CH2NH(CO)CH3 or
CH(CH3)NHC(0)CH3).
[52] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each hydrogen and X7 is Ci-C6 alkyl substituted with NR6R7 and with C6-Ci0 aryl wherein the C6-Cio aryl is optionally substituted with OH. In more specific examples, X1 and X2 are each hydrogen and X7 is CH3 or C2¾ wherein X7 is substituted either with (a) NH2 and phenyl or (b) NH2 and 4-hydroxyphenyl. In another example of this aspect, X1 and X2 are each deuterium and X7 is Ci-C6 alkyl substituted with NR6R7 and with C6-Cio aryl wherein the C6-Cio aryl is optionally substituted with OH. In more specific examples, X1 and X2 are each deuterium and X7 is CH3 or C2H5 wherein X7 is substituted either with (a) NH2 and phenyl or (b) NH2 and 4-hydroxyphenyl.
[53] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each hydrogen and X7 is 3- to 8-membered heterocycloalkyl. In a more specific example, X1 and X2 are each hydrogen and X7 is pyrrolidinyl. The pyrrolydinyl may be, for example, 2-(S)-pyrrolidinyl or 2-(R)-pyrrolidinyl.
[54] In another example of the aspect where R4 is -C(X1)(X2)OC(0)X7, X1 and X2 are each deuterium and X7 is 3- to 8-membered heterocycloalkyl. In a more specific example, X1 and X2 are each deuterium and X7 is pyrrolidinyl. The pyrrolydinyl may be, for example, 2-(S)-pyrrolidinyl or 2-(R)-pyrroliydinyl.
[55] In another set of embodiments, any atom not designated as deuterium in any of the embodiments, aspects, or examples set forth above is present at its natural isotopic abundance.
[56] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 1 (below), wherein each Y3 is hydrogen:
Table 1 : Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[57] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 2 (below), wherein each Y3 is deuterium:
Table 2: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[58] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 3 (below), wherein each Y3 is hydrogen and R2 is C(0)OCH2OP(0)(OH)2:
Table 3: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[59] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 4 (below), wherein each Y3 is deuterium and R2 is C(0)OCH2OP(0)(OH)2:
Table 4: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[60] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 5 (below), wherein each Y3 is hydrogen and R2 is C(0)OCH2OC(0)CH(CH3)OH:
Table 5: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
[61] In one embodiment, the compound is the (R) enantiomer of any one of the compounds in Table 5. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 5.
[62] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 6 (below), wherein each Y3 is deuterium and R2 is C(0)OCH2OC(0)CH(CH3)OH:
Table 6: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
In one embodiment, the compound is the (R) enantiomer of any one of the compounds in Table 6. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 6.
[63] In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 7 (below), wherein each Y3 is hydrogen and R2 is C(0)OCH2OC(0)CH(CH3)NH2 :
Table 7: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
In one embodiment, the compound is the (R) enantiomer of any one of the compounds in Table 7. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 7.
In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 8 (below), wherein each Y3 is deuterium and R2 is C(0)OCH2OC(0)CH(CH3)NH2 :
Table 8: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
In one embodiment, the compound is the (R) enantiomer of any one of the compounds in Table 8. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 8.
In yet another embodiment, the compound is selected from any one of the compounds (Cmpd) set forth in Table 9 (below), wherein each Y3 is deuterium and R2 is C(0)OCH2OC(0)CH(CH2C6H5)NH2 :
Table 9: Exemplary Embodiments of Formula I
or a pharmaceutically acceptable salt thereof, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
In one embodiment, the compound is the (R) enantiomer of any one of the compounds in Table 9. In one embodiment, the compound is the (S) enantiomer of any one of the compounds in Table 9.
[64] In another set of embodiments, any atom not designated as deuterium in any of the embodiments, aspects, or examples set forth above is present at its natural isotopic abundance.
[65] The synthesis of compounds of Formula I may be readily achieved by synthetic chemists of ordinary skill by reference to the Exemplary Synthesis and Examples disclosed herein. Relevant procedures analogous to those of use for the preparation of compounds of Formula I and intermediates thereof are disclosed, for instance in Pandey, A et al, J Med Chem 2002, 45(17), 3772-3793; Knesl, P et al, Molecules, 11(4), 286-297, 2006, and PCT patent publication Nos WO2002016351; WO2002036587 and
WO2007012042.
[66] Such methods can be carried out utilizing corresponding deuterated and optionally, other isotope-containing reagents and/or intermediates to synthesize the compounds delineated herein, or invoking standard synthetic protocols known in the art for introducing isotopic atoms to a chemical structure.
Exemplary Synthesis
[67] Scheme 1 : Preparation of compounds of Formula I wherein R2 is other than COOR4
Formula I
[68] As shown in Scheme 1 , 2 may be treated with an amine m a manner analogous to that described in patent publication WO2009144473 Al to give 3.
Acylation of 3 with 4b in a manner analogous to that described in U.S. Patent No.
7,713.994 affords 5, the bromine atom of which may be displaced by piperazine 6
(prepared as disclosed in Scheme 2 below) in a manner analogous to that described in Roth et al, J. Med. Chem., 2009, 52, 4466-4480. Reduction of the nitro group of 7 with H2 and Pd/C in a manner analogous to that described in Roth et al. yields 8. Treatment of 8 with 9 (prepared as disclosed in Scheme 3 below) in a manner analogous to that described in Roth et al. gives a compound of Formula I. Preparation of examples of intermediate 6
X=H then R2=CH3
X=D then R2=CD3
[70] As shown in Scheme 2, 6a may be prepared from 10a, which is commercially available from CDN isotopes, by treating 10a with an alkyl iodide R2I and treating the resulting tertiary amine with HCI, as disclosed in U.S. patent publication 2009-0149399 . Intermediate 6b may be similarly prepared from 10b, which is prepared as described in J. Labelled Compounds and Radiopharmaceuticals, 25(4), 359-367, by treating 10b with R2I and treating the resulting tertiary amine with HCI. Intermediates 6c (where R2 is either CH3 or CD3, as discussed below) may be prepared from 10b, whjch is prepared as described above. 10b is treated sequentially with benzyl bromide, lithium aluminum hydride (or lithium aluminum deuteride) and H2, Pd/C to provide 6c where R2 is CH3
(when lithium aluminum hydride is used) or R2 is CD3 (when lithium aluminum deuteride is used).
15
As shown in Scheme 3, 11 is treated with thionyl chloride and R^H to give 12.
Treatment of 12 with methyl chloroacetate in a manner analogous to that described in Roth, et al. gives 13. Following a procedure analogous to that described in Roth, et al, reduction of the nitro group of 13 with ¾ and Pd/C affords 14; acylation of 14 with acetic anhydride gives 15; and treatment of 15 with PhC(OMe)3 yields 9.
[72] Scheme 4. General Synthesis of Compounds of Formula I where R2 is COOR4 and R4 is -C(X1 (X2 OC(0 X7
As shown in scheme 4, a compound of formula I wherein R2 is H - obtained, for example, as disclosed in Scheme 1 - is treated with 21, which may be obtained as disclosed in Scheme 6 below, to provide, after a deprotection step if necessary, the compound of formula I wherein R2 is COOR4 and R4 is -C(X1)(X2)OC(0)X7. In exemplary Scheme 4, X1 and X2 of formula I are each hydrogen, and X7 is Ci-C6 alkyl optionally substituted with one or more R9.
[73] Scheme 5. Synthesis of Compounds of Formula I where R2 is COOR4 , R4 is -C(X1 (X2 OC(0 X7 and X7 is Ci-Cfi alkyl substituted with NR6R7 and optionally with Cfi-Cui aryl wherein the Q-Ci o aryl is optionally substituted with OH.
As shown in scheme 5, a compound of formula I wherein R2 is H - obtained, for example, as disclosed in Scheme 1 - is treated with 17, which may be obtained as disclosed in Scheme 7 below, to provide, after removal of the Cbz protecting group by treatment with hydrogen and Pd(OH)2/C, the compound of formula I wherein R2 is COOR4 and R4 is -C(X1)(X2)OC(0)X7. In exemplary Scheme 5, X1 and X2 of formula I are each hydrogen, and X7 is C1-C5 alkyl substituted with NH2. In the C0-C5 alkyl group shown for 17 in Scheme 5, "Co" is intended to mean the absence of an alkyl substituent, so that for that particular embodiment 17 has the structure:
Scheme 5 may be readily modified by replacing 17, for example, with 47:
so as to provide a compound of formula I in which X is Ci-C6 alkyl, such as ethyl, substituted with NR6R7 and with C6-Ci0 aryl. The C6-Ci0 aryl may be optionally substituted with OH as disclosed herein.
Scheme 6. Preparation of 21.
HO. .X7
J 0L T 22
O EtS. .0. ,O. .X7
EtS O CI Π Π
18 C¾C°3 ° 23 °
S02CI2
Ck ^CL „x7
BF3-oEt2 γ ^γ
O O 21
As shown in Scheme 6, Compound 21 may be prepared by treating 18 with acid 22 to give 23; and reacting 23 with sulfuryl chloride to afford 21.
Scheme 7. Preparation of 17.
18
As shown in Scheme 7, Compound 17 may be prepared by treating chloromethyl chloroformate with NaSEt to provide 18; treating 18 with protected amino acid 19 to give 20; and reacting 20 with sulfuryl chloride to afford 17. A variety of naturally occurring amino acids may be envisioned for the formation of 18, such as valine, in which case the corresponding structure of 17 resulting from Scheme 7 is
[74] Scheme 8a. General Synthesis of Compounds of Formula I where R2 is COOR4 and R4 is -C(X1 (X2OP(Q(OH7
[75] As shown in scheme 8a, a compound of formula I wherein R2 is H - obtained, for example, as disclosed in Scheme 1 - is treated with 24 optionally in the presence of a base such as diisopropylethylamine to provide 25 which, following reductive debenzylation, affords the compound of formula I wherein R2 is COOR4 and R4 is
-C(X1)(X2)OP(0)(OH)2.. An example of 24 is the known
(bis(benzyloxy)phosphoryloxy)methyl carbonochloridate (wherein X1 and X2 are both hydrogen - see PCT publication WO2007050732A1 and European Patent publication EP747385). The disodium or calcium salt forms of the compound of formula I thus obtained may be accessed via treatment with sodium hydroxide or with calcium hydroxide or calcium acetate or calcium chloride.
Scheme 8b. Alternative general Synthesis of Compounds of Formula I where R2 is COOR4 and R4 is -CX^PfOYOHY?
[76] Scheme 8b depicts an alternate method for producing compounds of Formula I where R2 is COOR4 and R4 is -C(X1)(X2)OP(0)(OH)2. A compound of Formula I wherein R2 is hydrogen is treated with chloromethyl chloroformate to provide compound 26. Treatment with di-tert-butylphosphate potassium salt and tetrabutylammonium iodide (TBAI) provides compound 27. Removal of the t-butyl groups via treatment with trifluoroacetic acid provides compounds of Formula I wherein R2 is COOR4 and R4 is -C(X1)(X2)OP(0)(OH)2. The disodium or calcium salt forms of the compound of formula I thus obtained may be accessed via treatment with sodium hydroxide or with calcium hydroxide or calcium acetate or calcium chloride.
[77] The specific approaches and compounds shown above are not intended to be limiting. The chemical structures in the schemes herein depict variables that are hereby defined commensurately with chemical group definitions (moieties, atoms, etc.) of the corresponding position in the compound formulae herein, whether identified by the same variable name (i.e., R1, R2, R3, etc.) or not. The suitability of a chemical group in a compound structure for use in the synthesis of another compound is within
the knowledge of one of ordinary skill in the art.
[78] Additional methods of synthesizing compounds of Formula I and their synthetic precursors, including those within routes not explicitly shown in schemes herein, are within the means of chemists of ordinary skill in the art. Synthetic chemistry
transformations and protecting group methodologies (protection and deprotection) useful
in synthesizing the applicable compounds are known in the art and include, for example, those described in Larock R, Comprehensive Organic Transformations, VCH Publishers (1989); Greene, TW et al, Protective Groups in Organic Synthesis, 3rd Ed., John Wiley and Sons (1999); Fieser, L et al., Fieser and Fieser's Reagents for Organic Synthesis, John Wiley and Sons (1994); and Paquette, L, ed., Encyclopedia of Reagents for Organic Synthesis, John Wiley and Sons (1995) and subsequent editions thereof.
[79] Combinations of substituents and variables envisioned by this invention are only those that result in the formation of stable compounds.
Compositions
[80] The invention also provides pharmaceutical compositions comprising an effective amount of a compound of Formula I (e.g., including any of the formulae herein), or a pharmaceutically acceptable salt of said compound; and a pharmaceutically acceptable carrier. The carrier(s) are "acceptable" in the sense of being compatible with the other ingredients of the formulation and, in the case of a pharmaceutically acceptable carrier, not deleterious to the recipient thereof in an amount used in the medicament.
[81] Pharmaceutically acceptable carriers, adjuvants and vehicles that may be used in the pharmaceutical compositions of this invention include, but are not limited to, ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polyoxypropylene -block polymers, polyethylene glycol and wool fat.If required, the solubility and bioavailability of the compounds of the present invention in pharmaceutical compositions may be enhanced by methods well- known in the art. One method includes the use of lipid excipients in the formulation. See "Oral Lipid-Based Formulations: Enhancing the Bioavailability of Poorly Water-Soluble Drugs (Drugs and the Pharmaceutical Sciences)," David J. Hauss, ed. Informa
Healthcare, 2007; and "Role of Lipid Excipients in Modifying Oral and Parenteral Drug
Delivery: Basic Principles and Biological Examples," Kishor M. Wasan, ed. Wiley- Interscience, 2006.
[82] Another known method of enhancing bioavailability is the use of an amorphous form of a compound of this invention optionally formulated with a poloxamer, such as LUTROL™ and PLURONIC™ (BASF Corporation), or block copolymers of ethylene oxide and propylene oxide. See United States patent 7,014,866; and United States patent publications 20060094744 and 20060079502.
[83] The pharmaceutical compositions of the invention include those suitable for oral, rectal, nasal, topical (including buccal and sublingual), vaginal or parenteral (including subcutaneous, intramuscular, intravenous and intradermal) administration. In certain embodiments, the compound of the formulae herein is administered transdermally (e.g., using a transdermal patch or iontophoretic techniques). Other formulations may conveniently be presented in unit dosage form, e.g., tablets, sustained release capsules, and in liposomes, and may be prepared by any methods well known in the art of pharmacy. See, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins, Baltimore, MD (20th ed. 2000).
[84] Such preparative methods include the step of bringing into association with the molecule to be administered ingredients such as the carrier that constitutes one or more accessory ingredients. In general, the compositions are prepared by uniformly and intimately bringing into association the active ingredients with liquid carriers, liposomes or finely divided solid carriers, or both, and then, if necessary, shaping the product.
[85] In certain embodiments, the compound is administered orally. Compositions of the present invention suitable for oral administration may be presented as discrete units such as capsules, sachets, or tablets each containing a predetermined amount of the active ingredient; a powder or granules; a solution or a suspension in an aqueous liquid or a non-aqueous liquid; an oil-in-water liquid emulsion; a water-in-oil liquid emulsion; packed in liposomes; or as a bolus, etc. Soft gelatin capsules can be useful for containing such suspensions, which may beneficially increase the rate of compound absorption.
[86] In the case of tablets for oral use, carriers that are commonly used include lactose and corn starch. Lubricating agents, such as magnesium stearate, are also typically added. For oral administration in a capsule form, useful diluents include lactose and
dried cornstarch. When aqueous suspensions are administered orally, the active ingredient is combined with emulsifying and suspending agents. If desired, certain sweetening and/or flavoring and/or coloring agents may be added.
[87] Compositions suitable for oral administration include lozenges comprising the ingredients in a flavored basis, usually sucrose and acacia or tragacanth; and pastilles comprising the active ingredient in an inert basis such as gelatin and glycerin, or sucrose and acacia.
[88] Compositions suitable for parenteral administration include aqueous and nonaqueous sterile injection solutions which may contain anti-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents. The formulations may be presented in unit- dose or multi-dose containers, for example, sealed ampules and vials, and may be stored in a freeze dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example water for injections, immediately prior to use. Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets.
[89] Such injection solutions may be in the form, for example, of a sterile injectable aqueous or oleaginous suspension. This suspension may be formulated according to techniques known in the art using suitable dispersing or wetting agents (such as, for example, Tween 80) and suspending agents. The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally-acceptable diluent or solvent, for example, as a solution in 1,3-butanediol. Among the acceptable vehicles and solvents that may be employed are mannitol, water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose, any bland fixed oil may be employed including synthetic mono- or diglycerides. Fatty acids, such as oleic acid and its glyceride derivatives are useful in the preparation of injectables, as are natural pharmaceutically-acceptable oils, such as olive oil or castor oil, especially in their polyoxyethylated versions. These oil solutions or suspensions may also contain a long- chain alcohol diluent or dispersant.
[90] The pharmaceutical compositions of this invention may be administered in the form of suppositories for rectal administration. These compositions can be prepared by mixing a compound of this invention with a suitable non-irritating excipient which is solid at room temperature but liquid at the rectal temperature and therefore will melt in the rectum to release the active components. Such materials include, but are not limited to, cocoa butter, beeswax and polyethylene glycols.
[91] The pharmaceutical compositions of this invention may be administered by nasal aerosol or inhalation. Such compositions are prepared according to techniques well- known in the art of pharmaceutical formulation and may be prepared as solutions in saline, employing benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, fluorocarbons, and/or other solubilizing or dispersing agents known in the art. See, e.g.: Rabinowitz JD and Zaffaroni AC, US Patent 6,803,031, assigned to Alexza Molecular Delivery Corporation.
[92] Topical administration of the pharmaceutical compositions of this invention is especially useful when the desired treatment involves areas or organs readily accessible by topical application. For topical application topically to the skin, the pharmaceutical composition should be formulated with a suitable ointment containing the active components suspended or dissolved in a carrier. Carriers for topical administration of the compounds of this invention include, but are not limited to, mineral oil, liquid petroleum, white petroleum, propylene glycol, polyoxyethylene polyoxypropylene compound, emulsifying wax, and water. Alternatively, the pharmaceutical composition can be formulated with a suitable lotion or cream containing the active compound suspended or dissolved in a carrier. Suitable carriers include, but are not limited to, mineral oil, sorbitan monostearate, polysorbate 60, cetyl esters wax, cetearyl alcohol, 2- octyldodecanol, benzyl alcohol, and water. The pharmaceutical compositions of this invention may also be topically applied to the lower intestinal tract by rectal suppository formulation or in a suitable enema formulation. Topically-transdermal patches and iontophoretic administration are also included in this invention.
[93] Application of the subject therapeutics may be local, so as to be administered at the site of interest. Various techniques can be used for providing the subject
compositions at the site of interest, such as injection, use of catheters, trocars, projectiles,
pluronic gel, stents, sustained drug release polymers or other device which provides for internal access.
[94] Thus, according to yet another embodiment, the compounds of this invention may be incorporated into compositions for coating an implantable medical device, such as prostheses, artificial valves, vascular grafts, stents, or catheters. Suitable coatings and the general preparation of coated implantable devices are known in the art and are exemplified in US Patents 6,099,562; 5,886,026; and 5,304,121. The coatings are typically biocompatible polymeric materials such as a hydrogel polymer,
polymethyldisiloxane, polycaprolactone, polyethylene glycol, polylactic acid, ethylene vinyl acetate, and mixtures thereof. The coatings may optionally be further covered by a suitable topcoat of fluorosilicone, polysaccharides, polyethylene glycol, phospholipids or combinations thereof to impart controlled release characteristics in the composition. Coatings for invasive devices are to be included within the definition of pharmaceutically acceptable carrier, adjuvant or vehicle, as those terms are used herein.
[95] According to another embodiment, the invention provides a method of coating an implantable medical device comprising the step of contacting said device with the coating composition described above. It will be obvious to those skilled in the art that the coating of the device will occur prior to implantation into a mammal.
[96] According to another embodiment, the invention provides a method of impregnating an implantable drug release device comprising the step of contacting said drug release device with a compound or composition of this invention. Implantable drug release devices include, but are not limited to, biodegradable polymer capsules or bullets, non-degradable, diffusible polymer capsules and biodegradable polymer wafers.
[97] According to another embodiment, the invention provides an implantable medical device coated with a compound or a composition comprising a compound of this invention, such that said compound is therapeutically active.
[98] According to another embodiment, the invention provides an implantable drug release device impregnated with or containing a compound or a composition comprising a compound of this invention, such that said compound is released from said device and is therapeutically active.
[99] Where an organ or tissue is accessible because of removal from the subject, such organ or tissue may be bathed in a medium containing a composition of this invention, a composition of this invention may be painted onto the organ, or a composition of this invention may be applied in any other convenient way.
[100] In another embodiment, a composition of this invention further comprises a second therapeutic agent. The second therapeutic agent may be selected from any compound or therapeutic agent known to have or that demonstrates advantageous properties when administered with a compound having the same mechanism of action as intedanib. Such agents include those indicated as being useful in combination with intedanib, including but not limited to, those described in PCT patent publication
WO2002016351, WO2006002422 and WO2007014943.
[101] Preferably, the second therapeutic agent is an agent useful in the treatment or prevention of a disease or condition selected from cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme, and fibrosis, such as pulmonary fibrosis, such as IPF.
[102] In one embodiment, the second therapeutic agent is an anti-cancer agent.
[103] In another embodiment, the invention provides separate dosage forms of a compound of this invention and one or more of any of the above-described second therapeutic agents, wherein the compound and second therapeutic agent are associated with one another. The term "associated with one another" as used herein means that the separate dosage forms are packaged together or otherwise attached to one another such that it is readily apparent that the separate dosage forms are intended to be sold and administered together (within less than 24 hours of one another, consecutively or simultaneously).
[104] In the pharmaceutical compositions of the invention, the compound of the present invention is present in an effective amount. As used herein, the term "effective amount" refers to an amount which, when administered in a proper dosing regimen, is sufficient to treat the target disorder.
[105] The interrelationship of dosages for animals and humans (based on milligrams per meter squared of body surface) is described in Freireich et al, Cancer Chemother. Rep, 1966, 50: 219. Body surface area may be approximately determined from height and
weight of the subject. See, e.g., Scientific Tables, Geigy Pharmaceuticals, Ardsley, N.Y., 1970, 537.
[106] In one embodiment, an effective amount of a compound of this invention can range from 10 to 1200 mg, such as 20 to 1200 mg; 10 to 600 mg; or 20 to 600 mg.. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 1200 mg, such as 200 to 600 mg; 400 to 1200 mg; or 400 to 600 mg.. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 1000 mg, such as 400 to 1000 mg; 200 to 500 mg; or 400 to 500 mg..
[107] In one embodiment, an effective amount of a compound of this invention can range from 20 to 1200 mg, administered once a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 400 to 1200 mg, administered once a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 400 to 1000 mg, administered once a day.
[108] In one embodiment, an effective amount of a compound of this invention can range from 10 to 600 mg, administered twice a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 600 mg, administered twice a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 500 mg, administered twice a day.
[109] In one embodiment, an effective amount of a compound of this invention can range from 10 to 600 mg, administered once a day. In one aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 600 mg, administered once a day. In an alternate aspect of this embodiment an effective amount of a compound of this invention can range from 200 to 500 mg, administered once a day.
[110] In certain embodiments, the dosage of the compound of this invention is selected to produce a target tumor/plasma ratio of > 0.33.
[Ill] Effective doses will also vary, as recognized by those skilled in the art, depending on the diseases treated, the severity of the disease, the route of administration, the sex, age and general health condition of the subject, excipient usage, the possibility of co-
usage with other therapeutic treatments such as use of other agents and the judgment of the treating physician.
[112] For pharmaceutical compositions that comprise a second therapeutic agent, an effective amount of the second therapeutic agent is between about 20% and 100% of the dosage normally utilized in a monotherapy regime using just that agent. Preferably, an effective amount is between about 70% and 100% of the normal monotherapeutic dose. The normal monotherapeutic dosages of these second therapeutic agents are well known in the art. See, e.g., Wells et al, eds., Pharmacotherapy Handbook, 2nd Edition,
Appleton and Lange, Stamford, Conn. (2000); PDR Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000, Deluxe Edition, Tarascon Publishing, Loma Linda, Calif. (2000), each of which references are incorporated herein by reference in their entirety.
[113] It is expected that some of the second therapeutic agents referenced above will act synergistically with the compounds of this invention. When this occurs, it will allow the effective dosage of the second therapeutic agent and/or the compound of this invention to be reduced from that required in a monotherapy. This has the advantage of minimizing toxic side effects of either the second therapeutic agent of a compound of this invention, synergistic improvements in efficacy, improved ease of administration or use and/or reduced overall expense of compound preparation or formulation.
Methods of Treatment
[114] In another embodiment, the invention provides a method of inhibiting the activity of one or more of vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet derived growth factor receptor (PDGFR) in a cell, comprising contacting a cell with one or more compounds of Formula I herein. In a more specific aspect, the type III receptor tyrosine kinase is selected from PDGFR-B, c- KIT and Flt3.
[115] According to another embodiment, the invention provides a method of treating cancer such as non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme, and fibrosis, such as pulmonary fibrosis, such as IPF in a subject in need thereof, comprising the step of administering to the subject an effective amount of a compound or a composition of this invention.
[116] According to another embodiment, the invention provides a method of treating cancer such as colorectal cancer, small cell lung cancer (SCLC) and acute myeloid leukemia (AML).
[117] Identifying a subject in need of such treatment can be in the judgment of a subject or a health care professional and can be subjective (e.g. opinion) or objective (e.g.
measurable by a test or diagnostic method). In one embodiment the subject is a patient.
[118] In another embodiment, any of the above methods of treatment comprises the further step of co-administering to the subject in need thereof one or more second therapeutic agents. The choice of second therapeutic agent may be made from any second therapeutic agent known to be useful for co-administration with intedanib. The choice of second therapeutic agent is also dependent upon the particular disease or condition to be treated. Examples of second therapeutic agents that may be employed in the methods of this invention are those set forth above for use in combination
compositions comprising a compound of this invention and a second therapeutic agent.
[119] In one embodiment, the method of treating a cancer in a subject with a compound or composition of this invention comprises the additional step of co-administering to the subject in need thereof a second anti-cancer agent or subjecting the subject to radiation therapy. In one aspect, the second anti-cancer agent is selected from
bevacizumab and temozolomide. In a more specific embodiment, the second anti-cancer agent is bevacizumab and the cancer is selected from non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, and glioblastoma multiforme.
[120] The term "co-administered" as used herein means that the second therapeutic agent may be administered together with a compound of this invention as part of a single dosage form (such as a composition of this invention comprising a compound of the invention and an second therapeutic agent as described above) or as separate, multiple dosage forms. Alternatively, the additional agent may be administered prior to, consecutively with, or following the administration of a compound of this invention. In such combination therapy treatment, both the compounds of this invention and the second therapeutic agent(s) are administered by conventional methods. The administration of a composition of this invention, comprising both a compound of the invention and a second therapeutic agent, to a subject does not preclude the separate administration of that same
therapeutic agent, any other second therapeutic agent or any compound of this invention to said subject at another time during a course of treatment.
[121] Effective amounts of these second therapeutic agents are well known to those skilled in the art and guidance for dosing may be found in patents and published patent applications referenced herein, as well as in Wells et al., eds., Pharmacotherapy
Handbook, 2nd Edition, Appleton and Lange, Stamford, Conn. (2000); PDR
Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000, Deluxe Edition, Tarascon Publishing, Loma Linda, Calif. (2000), and other medical texts. However, it is well within the skilled artisan's purview to determine the second therapeutic agent's optimal effective-amount range.
[122] In one embodiment of the invention, where a second therapeutic agent is administered to a subject, the effective amount of the compound of this invention is less than its effective amount would be where the second therapeutic agent is not
administered. In another embodiment, the effective amount of the second therapeutic agent is less than its effective amount would be where the compound of this invention is not administered. In this way, undesired side effects associated with high doses of either agent may be minimized. Other potential advantages (including without limitation improved dosing regimens and/or reduced drug cost) will be apparent to those of skill in the art.
[123] In another embodiment, the invention provides a method of treating a disease or condition associated with or complicated with immune suppression in a subject in need thereof, comprising the step of administering to the subject an effective amount of a compound or a composition of this invention.
[124] In yet another aspect, the invention provides the use of a compound of Formula I alone or together with one or more of the above-described second therapeutic agents in the manufacture of a medicament, either as a single composition or as separate dosage forms, for treatment or prevention in a subject of a disease, disorder or symptom set forth above. Another aspect of the invention is a compound of Formula I for use in the treatment or prevention in a subject of a disease, disorder or symptom thereof delineated herein.
Example 1. Evaluation of Metabolic Stability
[125] Microsomal Assay: Human liver microsomes (20 mg/mL) are obtained from Xenotech, LLC (Lenexa, KS). β -nicotinamide adenine dinucleotide phosphate, reduced form (NADPH), magnesium chloride (MgCl2), and dimethyl sulfoxide (DMSO) are purchased from Sigma- Aldrich.
[126] Determination of Metabolic Stability: 7.5 mM stock solutions of test compounds are prepared in DMSO. The 7.5 mM stock solutions are diluted to 12.5-50 μΜ in acetonitrile (ACN). The 20 mg/mL human liver microsomes are diluted to 0.625 mg/mL in 0.1 M potassium phosphate buffer, pH 7.4, containing 3 mM MgCl2. The diluted microsomes are added to wells of a 96-well deep-well polypropylene plate in triplicate. A 10 aliquot of the 12.5-50 μΜ test compound is added to the microsomes and the mixture is pre-warmed for 10 minutes. Reactions are initiated by addition of pre-warmed NADPH solution. The final reaction volume is 0.5 mL and contains 0.5 mg/mL human liver microsomes, 0.25-1.0 μΜ test compound, and 2 mM NADPH in 0.1 M potassium phosphate buffer, pH 7.4, and 3 mM MgCl2. The reaction mixtures are incubated at 37 °C, and 50 μΐ, aliquots are removed at 0, 5, 10, 20, and 30 minutes and added to shallow-well 96-well plates which contain 50 μΐ^ of ice-cold ACN with internal standard to stop the reactions. The plates are stored at 4 °C for 20 minutes after which 100 μΐ^ of water is added to the wells of the plate before centrifugation to pellet precipitated proteins. Supernatants are transferred to another 96-well plate and analyzed for amounts of parent remaining by LC-MS/MS using an Applied Bio-systems API 4000 mass spectrometer. The same procedure is followed for the non-deuterated counterpart of the compound of Formula I and the positive control, 7-ethoxycoumarin (1 μΜ). Testing is done in triplicate.
[127] Data analysis: The in vitro ty2s for test compounds are calculated from the slopes of the linear regression of % parent remaining (In) vs incubation time relationship,
in vitro t ½ = 0.693/k
k = -[slope of linear regression of % parent remaining(ln) vs incubation time]
[128] Data analysis is performed using Microsoft Excel Software.
[129] Without further description, it is believed that one of ordinary skill in the art can, using the preceding description and the illustrative examples, make and utilize the
compounds of the present invention and practice the claimed methods. It should be understood that the foregoing discussion and examples merely present a detailed description of certain preferred embodiments. It will be apparent to those of ordinary skill in the art that various modifications and equivalents can be made without departing from the spirit and scope of the invention.
Claims
A compound of Formula I
Formula I
, or a pharmaceutically acceptable salt thereof, wherein:
each of R1 and R3 is independently selected from CH3, CH2D, CHD2 and CD3;
R2 is CH3, CH2D, CHD2, CD3, hydrogen, or -C(0)OR4;
R4 is -C(X1)(X2)OP(0)(OH)2 or -C(X1)(X2)OC(0)X7;
X7 is 3- to 8-membered heterocycloalkyl or Ci-C6 alkyl optionally substituted with one or more R9;
each R9 is independently OR5, NR6R7, or C6-Cio aryl wherein the C6-Cio aryl is optionally substituted with OH;
R5 is hydrogen, C C6 alkyl or C C6 alkyl-C(O);
R6 and R7 are each independently hydrogen, Ci-C6 alkyl or Ci-C6 alkyl-C(O);
X1 and X2 are each independently hydrogen, deuterium or Ci-C6 alkyl;
or X1 and X2 taken together with the carbon they are joined to forms a C3-Cs carbocycle or a 3- to 8-membered heterocycle containing one or two heteroatoms
independently selected from S, O, and N;
each Y1 is the same and is hydrogen or deuterium;
each Y2 is the same and is hydrogen or deuterium;
and
each Y3 is the same and is hydrogen or deuterium;
provided that when each Y1 is hydrogen, each Y2 is hydrogen; R1 is CH3; R3 is CH3; and R2 is not -C(0)OR4,
then each Y3 is deuterium.
2. The compound of claim 1, wherein each of R1, R3 and R2 is independently selected from CH3 and CD3.
3. The compound of claim 1 or 2, wherein R1 is CD3.
4. The compound of claim 1 or 2, wherein R1 is CH3.
5. The compound of claim 1, 2 3, or 4, wherein R3 is CD3.
6. The compound of claim 1, 2, 3 or 4, wherein R3 is CH3.
7. The compound of any one of claims 1 to 6, wherein R2 is CD3.
8. The compound of any one of claims 1 to 6, wherein R2 is CH3.
9. The compound of any one of claims 1 to 6, wherein R2 is -C(0)OR4.
10. The compound of any one of claims 1 to 9, wherein each Y1 is deuterium.
11. The compound of any one of claims 1 to 9, wherein each Y1 is hydrogen.
12. The compound of any one of claims 1 to 11, wherein each Y2 is deuterium.
13. The compound of any one of claims 1 to 11, wherein each Y2 is hydrogen.
14. The compound of claim 1, wherein the compound is selected from any one of the compounds (Cmpd) set forth in the table below and wherein each Y3 is hydrogen:
15. The compound of claim 1, wherein the compound is selected from any one of the compounds (Cmpd) set forth in the table below and wherein each Y3 is deuterium:
16. The compound of any one of the preceding claims, wherein any atom not designated as deuterium is present at its natural isotopic abundance.
17. A pharmaceutical composition comprising the compound of claim 1; and a pharmaceutically acceptable carrier.
18. A method of treating cancer, fibrosis, or a combination thereof in a subject, comprising administering to the subject in need thereof a pharmaceutical composition of claim 17.
19. The method of claim 18, wherein the method is a method of treating pulmonary fibrosis.
20. The method of claim 19, wherein the fibrosis is IPF.
21. The method of claim 18, wherein the cancer is non-small cell lung cancer, ovarian cancer, renal cell carcinoma, hepatocellular carcinoma, or glioblastoma multiforme.
22. The method of claim 18, wherein the cancer is colorectal cancer, small cell lung cancer (SCLC) or acute myeloid leukemia (AML).
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361847723P | 2013-07-18 | 2013-07-18 | |
| US61/847,723 | 2013-07-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2015009889A1 true WO2015009889A1 (en) | 2015-01-22 |
Family
ID=51293161
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2014/046951 WO2015009889A1 (en) | 2013-07-18 | 2014-07-17 | Deuterated intedanib derivatives and their use for the treatment of proliferative disorders |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2015009889A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112574094A (en) * | 2020-12-14 | 2021-03-30 | 成都大学 | Indolone derivatives and pharmaceutical use thereof |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5304121A (en) | 1990-12-28 | 1994-04-19 | Boston Scientific Corporation | Drug delivery system making use of a hydrogel polymer coating |
| EP0747385A1 (en) | 1995-06-06 | 1996-12-11 | Bristol-Myers Squibb Company | Prodrugs of paclitaxel derivatives |
| US5886026A (en) | 1993-07-19 | 1999-03-23 | Angiotech Pharmaceuticals Inc. | Anti-angiogenic compositions and methods of use |
| US6099562A (en) | 1996-06-13 | 2000-08-08 | Schneider (Usa) Inc. | Drug coating with topcoat |
| WO2001027081A1 (en) * | 1999-10-13 | 2001-04-19 | Boehringer Ingelheim Pharma Kg | 6-position substituted indoline, production and use thereof as a medicament |
| WO2002016351A1 (en) | 2000-08-18 | 2002-02-28 | Cor Therapeutics, Inc. | Quinazoline derivatives as kinase inhibitors |
| WO2002036587A2 (en) | 2000-11-01 | 2002-05-10 | Cor Therapeutics, Inc. | Process for the production of 4-quinazolinylpiperazin-1-carboxylic acid phenylamides |
| US20040176392A1 (en) * | 2002-07-24 | 2004-09-09 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 3-Z-[1-(4-(N-((4-methyl-piperazin-1-yl)-methylcarbonyl)-N-methyl-amino)-anilino)-1-phenyl-methylene]-6-methoxycarbonyl-2-indolinone-monoethanesulphonate and the use thereof as a pharmaceutical composition |
| US6803031B2 (en) | 2001-05-24 | 2004-10-12 | Alexza Molecular Delivery Corporation | Delivery of erectile dysfunction drugs through an inhalation route |
| WO2006002422A2 (en) | 2004-06-24 | 2006-01-05 | Novartis Vaccines And Diagnostics Inc. | Compounds for immunopotentiation |
| US7014866B2 (en) | 2001-05-03 | 2006-03-21 | Hoffmann-La Roche Inc. | High dose solid unit oral pharmaceutical dosage form of amorphous nelfinavir mesylate and process for making same |
| US20060079502A1 (en) | 1999-11-02 | 2006-04-13 | Steffen Lang | Pharmaceutical compositions |
| US20060094744A1 (en) | 2004-09-29 | 2006-05-04 | Maryanoff Cynthia A | Pharmaceutical dosage forms of stable amorphous rapamycin like compounds |
| WO2006067165A2 (en) * | 2004-12-24 | 2006-06-29 | Boehringer Ingelheim International Gmbh | Indolidone derivatives for the treatment or prevention of fibrotic diseases |
| WO2007012042A1 (en) | 2005-07-20 | 2007-01-25 | Millennium Pharmaceuticals, Inc. | New crystal forms of 4-[6-methoxy-7-(3-piperidin-1-yl-propoxy)quinazolin-4-yl]piperazine-1-carboxylic acid(4-isopropoxyphenyl)-amide |
| WO2007014943A2 (en) | 2005-08-01 | 2007-02-08 | Ares Trading S.A. | Therapy for neurological diseases |
| WO2007050732A1 (en) | 2005-10-28 | 2007-05-03 | Abbott Laboratories | INDAZOLE DERIVATIVES THAT INHIBIT TRPVl RECEPTOR |
| US20090149399A1 (en) | 2007-12-10 | 2009-06-11 | Concert Pharmaceuticals Inc. | Heterocyclic kinase inhibitors |
| WO2009144473A1 (en) | 2008-05-29 | 2009-12-03 | F2G Limited | Antifungal combination therapy |
| US7713994B2 (en) | 2005-12-22 | 2010-05-11 | Wyeth Llc | Substituted isoquinoline-1,3(2H,4H)-diones, 1-thioxo,1,4-dihydro-2H-isoquinoline-3-ones and 1,4-dihyro-3 (2H)-isoquinolones and methods of use thereof |
-
2014
- 2014-07-17 WO PCT/US2014/046951 patent/WO2015009889A1/en active Application Filing
Patent Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5304121A (en) | 1990-12-28 | 1994-04-19 | Boston Scientific Corporation | Drug delivery system making use of a hydrogel polymer coating |
| US5886026A (en) | 1993-07-19 | 1999-03-23 | Angiotech Pharmaceuticals Inc. | Anti-angiogenic compositions and methods of use |
| EP0747385A1 (en) | 1995-06-06 | 1996-12-11 | Bristol-Myers Squibb Company | Prodrugs of paclitaxel derivatives |
| US6099562A (en) | 1996-06-13 | 2000-08-08 | Schneider (Usa) Inc. | Drug coating with topcoat |
| WO2001027081A1 (en) * | 1999-10-13 | 2001-04-19 | Boehringer Ingelheim Pharma Kg | 6-position substituted indoline, production and use thereof as a medicament |
| US20060079502A1 (en) | 1999-11-02 | 2006-04-13 | Steffen Lang | Pharmaceutical compositions |
| WO2002016351A1 (en) | 2000-08-18 | 2002-02-28 | Cor Therapeutics, Inc. | Quinazoline derivatives as kinase inhibitors |
| WO2002036587A2 (en) | 2000-11-01 | 2002-05-10 | Cor Therapeutics, Inc. | Process for the production of 4-quinazolinylpiperazin-1-carboxylic acid phenylamides |
| US7014866B2 (en) | 2001-05-03 | 2006-03-21 | Hoffmann-La Roche Inc. | High dose solid unit oral pharmaceutical dosage form of amorphous nelfinavir mesylate and process for making same |
| US6803031B2 (en) | 2001-05-24 | 2004-10-12 | Alexza Molecular Delivery Corporation | Delivery of erectile dysfunction drugs through an inhalation route |
| US20040176392A1 (en) * | 2002-07-24 | 2004-09-09 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | 3-Z-[1-(4-(N-((4-methyl-piperazin-1-yl)-methylcarbonyl)-N-methyl-amino)-anilino)-1-phenyl-methylene]-6-methoxycarbonyl-2-indolinone-monoethanesulphonate and the use thereof as a pharmaceutical composition |
| WO2006002422A2 (en) | 2004-06-24 | 2006-01-05 | Novartis Vaccines And Diagnostics Inc. | Compounds for immunopotentiation |
| US20060094744A1 (en) | 2004-09-29 | 2006-05-04 | Maryanoff Cynthia A | Pharmaceutical dosage forms of stable amorphous rapamycin like compounds |
| WO2006067165A2 (en) * | 2004-12-24 | 2006-06-29 | Boehringer Ingelheim International Gmbh | Indolidone derivatives for the treatment or prevention of fibrotic diseases |
| WO2007012042A1 (en) | 2005-07-20 | 2007-01-25 | Millennium Pharmaceuticals, Inc. | New crystal forms of 4-[6-methoxy-7-(3-piperidin-1-yl-propoxy)quinazolin-4-yl]piperazine-1-carboxylic acid(4-isopropoxyphenyl)-amide |
| WO2007014943A2 (en) | 2005-08-01 | 2007-02-08 | Ares Trading S.A. | Therapy for neurological diseases |
| WO2007050732A1 (en) | 2005-10-28 | 2007-05-03 | Abbott Laboratories | INDAZOLE DERIVATIVES THAT INHIBIT TRPVl RECEPTOR |
| US7713994B2 (en) | 2005-12-22 | 2010-05-11 | Wyeth Llc | Substituted isoquinoline-1,3(2H,4H)-diones, 1-thioxo,1,4-dihydro-2H-isoquinoline-3-ones and 1,4-dihyro-3 (2H)-isoquinolones and methods of use thereof |
| US20090149399A1 (en) | 2007-12-10 | 2009-06-11 | Concert Pharmaceuticals Inc. | Heterocyclic kinase inhibitors |
| WO2009144473A1 (en) | 2008-05-29 | 2009-12-03 | F2G Limited | Antifungal combination therapy |
Non-Patent Citations (30)
| Title |
|---|
| "Encyclopedia ofreagentsfor Organic Synthesis", 1995, JOHN WILEY AND SONS |
| "Oral Lipid-Based Formulations: Enhancing the Bioavailability of Poorly Water-Soluble Drugs (Drugs and the Pharmaceutical Sciences", 2007, INFORMA HEALTHCARE |
| "PDR Pharmacopoeia, Tarascon Pocket Pharmacopoeia 2000", 2000, TARASCON PUBLISHING |
| "PDR Pharmacopoeia, Tarascon Pocket Pharmacopoeia", 2000, TARASCON PUBLISHING |
| "Pharmacotherapy Handbook", 2000, APPLETON AND LANGE |
| "Remington: The Science and Practice of Pharmacy", 2000, LIPPINCOTT WILLIAMS & WILKINS |
| "Role of Lipid Excipients in Modifying Oral and Parenteral Drug Delivery: Basic Principles and Biological Examples", 2006, WILEY-INTERSCIENCE |
| "Scientific Tables, Geigy Pharmaceuticals", 1970, ARDSLEY, pages: 537 |
| BLAKE, MI ET AL., J PHARM SCI, vol. 64, 1975, pages 367 - 91 |
| DATABASE REGISTRY [online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; 22 October 2010 (2010-10-22), XP002729126, retrieved from STN Database accession no. 1246833-43-1 * |
| DUMONT ET AL: "Prospects in the use of deuterated molecules as therapeutic agents", REVUE IRE, INSTITUT NATIONAL DES RADIOELEMENTS, BELGIUM, vol. 6, no. 4, 1 January 1982 (1982-01-01), pages 2 - 10, XP009125461, ISSN: 0770-1160 * |
| FIESER, L ET AL.: "Fieser and Fieser's Reagentsfor Organic Synthesis", 1994, JOHN WILEY AND SONS |
| FISHER, MB ET AL., CURR OPIN DRUG DISCOV DEVEL, vol. 9, 2006, pages 101 - 09 |
| FOSTER A B: "Deuterium isotope effects in the metabolism of drugs and xenobiotics: implications for drug design", ADVANCES IN DRUG RESEARCH, ACADEMIC PRESS, LONDON, GB, vol. 14, 1 January 1985 (1985-01-01), pages 1 - 40, XP009086953, ISSN: 0065-2490 * |
| FOSTER, AB, ADV DRUG RES, vol. 14, 1985, pages 1 - 40 |
| FREIREICH ET AL., CANCER CHEMOTHER. REP, vol. 50, 1966, pages 219 |
| FUKUTO ET AL., J. MED. CHEM., vol. 34, 1991, pages 2871 - 76 |
| GANNES, LZ ET AL., COMP BIOCHEM PHYSIOL MOL INTEGR PHYSIOL, vol. 119, 1998, pages 725 |
| GREENE, TW ET AL.: "Protective Groups in Organic Synthesis", 1999, JOHN WILEY AND SONS |
| J. LABELLED COMPOUNDS AND RADIOPHARMACEUTICALS, vol. 25, no. 4, pages 359 - 367 |
| KEMPF, D.J. ET AL., ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, vol. 41, no. 3, 1997, pages 654 - 60 |
| KNESL, P ET AL., MOLECULES, vol. 11, no. 4, 2006, pages 286 - 297 |
| KUSHNER DJ ET AL: "Pharmacological uses and perspectives of heavy water and deuterated compounds", CANADIAN JOURNAL OF PHYSIOLOGY AND PHARMACOLOGY, OTTAWA, ONT, CA, vol. 77, no. 2, 1 February 1999 (1999-02-01), pages 79 - 88, XP009086918 * |
| KUSHNER, DJ ET AL., CAN J PHYSIOL PHARMACOL, 1999, pages 79 - 88 |
| LAROCK R: "Comprehensive Organic Transformations", 1989, VCH PUBLISHERS |
| PANDEY, A ET AL., J MED CHEM, vol. 45, no. 17, 2002, pages 3772 - 3793 |
| ROTH ET AL., J. MED. CHEM., vol. 52, 2009, pages 4466 - 4480 |
| WADA, E ET AL., SEIKAGAKU, vol. 66, 1994, pages 15 |
| WANG, L ET AL., CLINICAL PHARMACOLOGY AND THERAPEUTICS, vol. 56, 1994, pages 659 - 67 |
| XENOBIOTICA, vol. 41, no. 4, 2011, pages 297 - 311 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112574094A (en) * | 2020-12-14 | 2021-03-30 | 成都大学 | Indolone derivatives and pharmaceutical use thereof |
| WO2022126441A1 (en) * | 2020-12-14 | 2022-06-23 | 成都大学 | Oxindole derivative and pharmaceutical use thereof |
| CN112574094B (en) * | 2020-12-14 | 2022-07-01 | 成都大学 | Indolone derivatives and pharmaceutical use thereof |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2014235462B2 (en) | Deuterated palbociclib | |
| WO2012151361A1 (en) | Carbamoylpyridone derivatives | |
| US9776973B2 (en) | Deuterated momelotinib | |
| WO2014011971A2 (en) | Deuterated carfilzomib | |
| EP2935251A1 (en) | Deuterated alk inhibitors | |
| WO2011017612A1 (en) | Substituted diphenylpyrazine derivatives | |
| WO2018005328A1 (en) | Deuterated bictegravir | |
| US9676790B2 (en) | Substituted thienotriazolodiazapines | |
| WO2011091035A1 (en) | Aminoquinoline derivatives | |
| EP2968268A1 (en) | Inhibitors of the enzyme udp-glucose: n-acyl-sphingosine glucosyltransferase | |
| EP2970213A1 (en) | Deuterated pacritinib | |
| WO2015009889A1 (en) | Deuterated intedanib derivatives and their use for the treatment of proliferative disorders | |
| US10683305B2 (en) | Deuterated OTX-015 | |
| WO2012129381A1 (en) | Deuterated preladenant | |
| WO2010068480A1 (en) | Deuterated derivatives of dimeboline | |
| US20110201678A1 (en) | Xanthenone-4-Acetic Acid Derivatives | |
| WO2011159920A1 (en) | [5,6]-dihydro-2h-pyran-2-one derivatives | |
| WO2018013686A1 (en) | Deuterated idalopirdine | |
| EP2804857A1 (en) | Deuterated alpha-lipoic acid | |
| WO2014150044A1 (en) | Amine reuptake inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14748042 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14748042 Country of ref document: EP Kind code of ref document: A1 |