WO2014189123A1 - Sample pre-treatment method for measuring the quantity of toxic elements in agricultural produce samples - Google Patents
Sample pre-treatment method for measuring the quantity of toxic elements in agricultural produce samples Download PDFInfo
- Publication number
- WO2014189123A1 WO2014189123A1 PCT/JP2014/063639 JP2014063639W WO2014189123A1 WO 2014189123 A1 WO2014189123 A1 WO 2014189123A1 JP 2014063639 W JP2014063639 W JP 2014063639W WO 2014189123 A1 WO2014189123 A1 WO 2014189123A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sample
- conventional method
- extract
- amount
- present
- Prior art date
Links
Images
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/02—Food
- G01N33/10—Starch-containing substances, e.g. dough
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N1/00—Sampling; Preparing specimens for investigation
- G01N1/28—Preparing specimens for investigation including physical details of (bio-)chemical methods covered elsewhere, e.g. G01N33/50, C12Q
- G01N1/40—Concentrating samples
- G01N1/4055—Concentrating samples by solubility techniques
- G01N2001/4061—Solvent extraction
Definitions
- the present invention relates to a sample pretreatment method for measuring on-site the amount of harmful elements such as cadmium in crop samples such as rice, soybeans, sesame, etc., and particularly reduces work time and labor compared to conventional methods. And a method for improving the extraction rate of the measurement element.
- the amount of harmful elements contained in agricultural products is generally measured using an analytical instrument such as an ICP emission spectrometer or an atomic absorption photometer.
- an analytical instrument such as an ICP emission spectrometer or an atomic absorption photometer.
- measurement using an ICP emission spectroscopic analyzer or an atomic absorption photometer is employed.
- the measurement using these instruments required not only very expensive analytical instruments and specialized pretreatment, but also a long processing time and labor.
- the measurement cannot be performed near the site, and it is necessary to send the sample to a facility where an analytical instrument is installed and perform the measurement there.
- Patent Document 1 has been proposed as a method capable of measuring the amount of cadmium in a crop near the site.
- This method can be measured in a shorter time than the above method, but when the sample is a grain, legume or seed, it is necessary to finely pulverize with a miller at the time of pretreatment. In this case, there are problems such as troublesome washing of the miller and frequent occurrence of failure of the miller due to the long time for fine grinding.
- cadmium is extracted by increasing the contact ratio with hydrochloric acid by finely pulverizing the sample.
- the mobility of cadmium ions from the plant cell structure cannot be ensured sufficiently, the amount of cadmium extracted is significantly reduced. There was a problem that a small number of samples occurred.
- the present invention has been devised in view of the above-described problems of the prior art, and its purpose is to provide cadmium, arsenic, zinc, manganese, copper, lead, and cereals in grains, beans, and seed crop samples.
- Another object of the present invention is to provide a method capable of performing pretreatment of a sample when measuring the amount of chromium element simply and accurately in a short time with a high extraction rate.
- the present inventor has intensively studied a pretreatment method capable of completely extracting a measurement element by a simple method without performing pulverization with a miller.
- the sample is coarsely crushed so that the crusher used in the measuring device can be used, ⁇ -starch is converted to ⁇ -starch by adding water to this and heating, and ⁇ -starch is converted to sugar by adding an enzyme.
- the problem of fine pulverization by Mirther can be solved and a high extraction rate of the measurement elements from the sample can be achieved. As a result, the present invention has been completed.
- the present invention has the following configurations (1) to (7).
- (1) Measure the amount of at least one element selected from the group consisting of cadmium, arsenic, zinc, manganese, copper, lead and chromium in a crop sample selected from grains, beans or seeds.
- a sample pretreatment method for (I) a step of coarsely crushing the sample; (Ii) adding water to the coarsely pulverized sample and heating to convert ⁇ starch contained in the sample into ⁇ starch; (Iii) adding an enzyme to the sample to convert alpha starch in the sample into sugar; (Iv) adding hydrochloric acid to the sample to extract the measurement element in the sample, and (v) removing the solid matter from the extracted liquid.
- the pretreatment method of the present invention does not pulverize the sample in order to increase the contact ratio with hydrochloric acid, it is not necessary to use a pulverizer such as a miller. There is no burden of replacement costs. Since coarse pulverization is sufficient instead of fine pulverization, the coarse pulverized sample generally used for measuring the moisture content of agricultural products can be used as it is, and the sample weighing and pulverization can be omitted. Further, as a result of these, the overall measurement time is short and the measurement cost is low. Furthermore, due to water content in the plant cell structure and effective destruction of the plant cell structure that restrains the measurement element, the measurement element has high mobility, and extraction by chlorine complexation is sufficiently performed. Therefore, the measurement element is completely extracted from the sample, and the measurement accuracy is high.
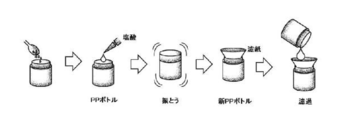
- FIG. 1 is a schematic diagram showing the procedure of the pretreatment method of the present invention used in the examples.
- FIG. 2 is a schematic diagram showing the procedure of the conventional method B used in the examples.
- the present invention is a sample pretreatment method for measuring the amount of a specific harmful element of a specific crop sample, and basically (i) a coarse pulverization step, (ii) a water addition & heating step, ( It consists of iii) enzyme addition step, (iv) hydrochloric acid addition step, and (v) solid matter removal step.
- the crop sample targeted by the method of the present invention is selected from grains, beans, or seeds, and these crops are protected by the cellulose layer on the surface, so that they are in the state at the time of harvest.
- the treatment liquid such as hydrochloric acid does not reach the starch layer inside.
- grains include rice, wheat, and buckwheat
- examples of beans include soybeans, red beans, and peanuts
- examples of seeds include sesame and rapeseed.
- the element to be measured in the method of the present invention is at least one element selected from the group consisting of cadmium, arsenic, zinc, manganese, copper, lead, and chromium. Since these elements have a harmful effect on the human body, it is necessary to monitor the content in crops before distribution.
- coarse pulverization refers to pulverization in which individual crushed pieces of a sample can be visually confirmed, and it is sufficient if the sample is divided into at least two pieces. Fine pulverization is not required so that individual crushed pieces of the sample cannot be visually confirmed. This is because, if divided into at least two pieces, the starch layer inside the sample is exposed, and the processing solution reaches the starch layer.
- the method of coarse pulverization is not particularly limited, and examples thereof include a method of hitting with a mallet or hammer or crushing with a hard material.
- a coarsely pulverized sample can be used as it is when measuring the moisture content of a crop sample. Thereby, the trouble of rough crushing or weighing can be omitted.
- the pulverizer can be easily cleaned with an air gun, etc. in a short time compared to fine pulverization, and the number of treatments per hour when processing a large number of samples is reduced. Increase significantly.
- the miller since the miller is not used for coarse pulverization, it is not necessary to consider the failure or replacement of the miller, which is a device having a relatively short service life.
- water is added to the coarsely pulverized sample and heated to convert ⁇ starch contained in the sample into ⁇ starch. Since the coarsely pulverized sample is ⁇ starch having low hydrophilicity as it is as a large pulverized piece, the measurement element cannot be extracted with hydrochloric acid. Therefore, in this process, water is added to the coarsely ground sample and heated to wipe the sample, thereby converting ⁇ starch into ⁇ starch and making it colloidal. As a result, the hydrophilicity of the sample is increased, and the measurement element can be sufficiently extracted with hydrochloric acid.
- the water to be added may be ordinary tap water, but it is preferable to use ion exchange water.
- the amount of water to be added may be equal to or greater than the sample weight. There is no upper limit to the amount of water, but if it is too much, the amount to be removed later increases.
- the heating method is not particularly limited, and the container in which water is added to the sample may be heated as it is. However, it is preferable because it is simple, efficient, and preferably heated by a microwave in a microwave oven or the like. In the case of heating with microwaves, for example, it is sufficient to perform the heating at about 500 W for about 30 seconds. When there is no heating equipment on site such as outdoors, it can be heated by heat of dissolution generated by adding calcium chloride or calcium carbonate.
- an enzyme is added to the sample to convert ⁇ starch in the sample into sugar.
- the sample In the above-described (ii) water addition & heating step, the sample is in a colloidal (gel) form, and thus cannot be filtered later. Therefore, in this step, the starch in the sample is decomposed into sugar by addition of the enzyme to change it into a smooth liquid.
- the enzyme is basically added in a neutral range in order to exert its ability. Further, after the enzyme is added to the sample, it is preferable to shake the sample in order to sufficiently infiltrate the sample into the sample.
- the enzyme is not particularly limited as long as the above object can be achieved.
- amylase for example, amylase, protease, and cellulase are used.
- Amylase has the effect of degrading starch into sugar
- protease and cellulase have the effect of degrading the surface layer bran when the sample is, for example, rice.
- the amount of the enzyme added is sufficient if it is converted to sugar, and generally only needs to be equal to or greater than the volume of the sample weight.
- hydrochloric acid is added to the sample to extract the measurement element in the sample.
- hydrochloric acid a hydrochloric acid solution having a concentration of preferably 0.002 to 2M, more preferably 0.05 to 0.5M may be used.
- the use ratio of the hydrochloric acid solution is generally 5 to 100% by volume of the sample, more preferably 10 to 50% by volume. If the amount used is too small, a sufficient chlorine complex cannot be formed, and if the amount used is too large, impurities in the sample increase.
- the hydrochloric acid solution may be added directly to the sample, or the sample may be immersed in the hydrochloric acid solution.
- a method that allows hydrochloric acid to sufficiently permeate the sample may be employed. Regardless of which method is employed, it is preferable to vigorously shake the sample in order to sufficiently permeate the sample with hydrochloric acid.
- the measurement element moved from the sample forms a chlorine complex.
- all the measurement elements constrained in the sample in the steps (i) to (iii) move so as to be able to react with chlorine, complex formation is possible for all the measurement elements.
- solids such as protein, crude fat, and ash are removed from the extracted liquid by filtration or centrifugation.
- Filtration is, for example, no.
- the filter paper of No. 2 may be used, and the centrifugation may be performed with the normal setting of the centrifuge. This significantly improves the applicability of the amount of measurement element to the measuring device.
- a sample prepared through the pretreatment method including the steps (i) to (v) of the present invention can measure the amount of the measurement element by any conventionally known method.
- the measuring method for example, an ICP method, a fluorescent X-ray method, an immunoassay method, a voltammetry method, an absorption method or the like is used.
- atomic absorption analysis (AAS), inductively coupled plasma atomic emission spectrometry (ICP- AES), inductively coupled plasma mass spectrometry (ICP-MS) or the like is used.
- Example 1 After roughly pulverizing 50 brown rice samples with a near-infrared moisture meter (Ketto Scientific Laboratory Co., Ltd., rice wheat moisture meter SP-1D3 type), according to the following extraction operation (the pretreatment method of the present invention), An extract was prepared. Extraction operation: (I) 1 g of brown rice sample coarsely pulverized with a moisture meter is put into a PP bottle (polypropylene bottle), and then 2.5 mL of ion-exchanged water is added (see (i) in FIG. 1).
- nitric acid was added to 50 specimens of the same brown rice and heated on a hot plate for about 24 hours, and an extraction solution was prepared according to an extraction operation (conventional method A) in which hydrogen peroxide was added to complete decomposition just before the decomposition.
- the conventional method A can be said to have an extraction rate of the measurement element of 100%, it requires a long-time pretreatment and is not used in the field.
- This conventional method A is implemented as a comparative index of the extraction ratio of the measurement element of the pretreatment method of the present invention.
- the amount of each element of cadmium, manganese, and zinc using ICP-OES (VISTA-MPX manufactured by SII) Using a device (atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation) with an atomic absorption photometer added with a hydride generator The amount of total arsenic was measured. Then, the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- ICP-OES VISTA-MPX manufactured by SII
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- Table 1 shows the amounts and recovery rates of each measurement element (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each brown rice specimen (sample numbers 1 to 50). Shown in
- the sample obtained by the pretreatment method of the present invention can be processed in a short time, but in the same manner as the conventional method A that requires a long time, cadmium, manganese, It is recognized that the measurement elements of zinc and arsenic are completely extracted.
- Comparative Example 1 After pulverizing 50 brown rice samples one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), an extract of conventional method B is prepared according to the following extraction procedure (conventional method B). did. Extraction operation: 1 g of the finely ground brown rice sample is put into a PP bottle (polypropylene bottle), and 10 mL of 0.1N hydrochloric acid is subsequently added. The PP bottle was shaken vigorously up and down for about 1 minute by hand, 2 Put in a new PP bottle with filter paper and filter the whole volume (see Fig. 2).
- an extract of the conventional method A was prepared from the same 50 brown rice samples in the same manner as in Example 1 according to the conventional method A described above.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the conventional method B with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Example 2 After 20 soybean samples were beaten in an agate mortar and coarsely pulverized, an extract of the method of the present invention was prepared according to the pretreatment method of the present invention in the same manner as in Example 1.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 20 samples of the same soybean according to the conventional method A described above.
- the amount of each element of cadmium, manganese, and zinc using ICP-OES (VISTA-MPX manufactured by SII) Using a device (atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation) with an atomic absorption photometer The amount of total arsenic was measured. Then, the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- ICP-OES VISTA-MPX manufactured by SII
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- Table 3 below shows the amounts and recovery rates of each measurement element (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each soybean sample (sample numbers 1 to 20). Shown in
- the sample obtained by the pretreatment method of the present invention can be processed in a short time, but in the same manner as in the conventional method A that requires a long time, cadmium, manganese, It is recognized that the measurement elements of zinc and arsenic are completely extracted.
- Comparative Example 2 Twenty soybean samples are pulverized one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), and then 10 samples are baked on a hot plate for 30 minutes until the color becomes dark. A sample was prepared (note: in the conventional method B soybean measurement, solid-liquid separation by filtration is not possible without baking treatment). From this sample, an extract of conventional method B was prepared according to the same extraction procedure as in conventional method B described in Comparative Example 1 (however, the shaking time was changed to 30 minutes).
- an extract of conventional method A was prepared in the same manner as in Example 1 from 20 samples of the same soybean.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the conventional method B with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Example 3 After 20 sesame samples were beaten in an agate mortar and coarsely pulverized, the extract of the method of the present invention was prepared according to the pretreatment method of the present invention in the same manner as in Example 1.
- the amount of each element of cadmium, manganese, and zinc using ICP-OES (VISTA-MPX manufactured by SII) Using a device (atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation) with an atomic absorption photometer added with a hydride generator The amount of total arsenic was measured. Then, the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- ICP-OES VISTA-MPX manufactured by SII
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- Table 5 shows the amounts and recovery rates of the respective measurement elements (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each sesame sample (sample numbers 1 to 20). Shown in
- the sample obtained by the pretreatment method of the present invention can be treated in a short time, but in the same way as the conventional method A that requires a long time, cadmium, manganese, It is recognized that the measurement elements of zinc and arsenic are completely extracted.
- Comparative Example 3 After pulverizing 20 sesame samples one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), an extract of conventional method B was prepared according to conventional method B as in Comparative Example 1. did.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the conventional method B with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Comparative Example 4 Example of the extraction operation of the pretreatment method of the present invention of Example 1 using 10 brown rice samples except that the addition of water shown in (i) and the heating by the microwave oven shown in (ii) were not performed. In the same manner as in Example 1, an extract of Comparative Method A was prepared.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 10 samples of the same brown rice.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the comparative method A with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Comparative Example 5 Extraction of Comparative Method B in the same manner as in Example 1 except that the enzyme shown in (ii) was not added in the extraction operation of the pretreatment method of the present invention in Example 1 using 10 brown rice samples. A liquid was created.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 10 samples of the same brown rice.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the comparative method B with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Table 8 shows the amounts and recovery rates of the measurement elements (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the comparative method B for each brown rice specimen (sample numbers 1 to 10).
- Comparative Example 6 Using the 10 brown rice samples, an extract of Comparative Method C was prepared in the same manner as in Example 1 except that coarse pulverization was not performed using the moisture meter in the extraction operation of the pretreatment method of the present invention of Example 1. did.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 10 samples of the same brown rice.
- the amount of each element of cadmium, manganese, and zinc was measured using ICP-OES (Vista-MPX manufactured by SII).
- ICP-OES Via-MPX manufactured by SII
- a device in which a hydride generator is added to the atomic absorption photometer atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation
- the amount was measured.
- the ratio (%) of the quantity of the measurement element obtained by the comparative method C with respect to the quantity of the measurement element obtained by the conventional method A was evaluated as a recovery rate.
- Example 4 Of the extraction operation of the pretreatment method of the present invention of Example 1 using 10 brown rice samples, Example 1 except that the brown rice was divided into two with a mallet instead of performing coarse pulverization with a moisture meter. Similarly, an extract of the method of the present invention was prepared.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 10 samples of the same brown rice.
- the amount of each element of cadmium, manganese, and zinc using ICP-OES (VISTA-MPX manufactured by SII) Using a device (atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation) with an atomic absorption photometer added with a hydride generator The amount of total arsenic was measured. Then, the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- ICP-OES VISTA-MPX manufactured by SII
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- Table 10 shows the amounts and recovery rates of the measurement elements (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each brown rice specimen (sample numbers 1 to 10). Shown in
- Example 5 In the extraction operation of the pretreatment method of the present invention in Example 1 using 10 brown rice samples, Example 1 except that brown rice was broken up to about 20 pieces with a mallet instead of performing coarse pulverization with a moisture meter. In the same manner, an extract of the present invention was prepared.
- an extract of conventional method A was prepared in the same manner as in Example 1 from 10 samples of the same brown rice.
- the amount of each element of cadmium, manganese, and zinc using ICP-OES (VISTA-MPX manufactured by SII) Using a device (atomic absorption photometer: AA-7000 manufactured by Shimadzu Corporation, hydride generator: HVG-1 manufactured by Shimadzu Corporation) with an atomic absorption photometer added with a hydride generator The amount of total arsenic was measured. Then, the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- ICP-OES VISTA-MPX manufactured by SII
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- Table 11 below shows the amounts and recovery rates of each measurement element (cadmium, manganese, zinc, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each brown rice specimen (sample numbers 1 to 10). Shown in
- Example 6 After roughly pulverizing 38 specimens of brown rice with a near-infrared moisture meter (Ketto Scientific Laboratory Co., Ltd., rice wheat moisture meter SP-1D3 type), in the same manner as in Example 1, according to the pretreatment method of the present invention, An extract was prepared.
- an extract of the conventional method A was prepared in the same manner as in Example 1 from 38 samples of the same brown rice.
- the amount of the measured element in the extract of the method of the present invention and the amount of the measured element of the extract of the conventional method A are compared, and the amount of the measured element obtained in the conventional method A is obtained by the pretreatment method of the present invention.
- the ratio (%) of the amount of the measured element obtained was evaluated as the recovery rate. The closer the value of the recovery rate is to 100%, the more the extract obtained by the pretreatment method of the present invention extracts the measurement element from the plant cell structure.
- the amount of each measurement element (cadmium, manganese, zinc, copper, lead, total chromium, total arsenic) obtained by the conventional method A and the pretreatment method of the present invention for each brown rice specimen (sample numbers 1 to 38), and The recovery is shown in Table 12 below.
- the sample obtained by the pretreatment method of the present invention can be processed in a short time, but as in the conventional method A that requires a long time, cadmium, manganese, It can be seen that the measurement elements of zinc, copper, lead, chromium and arsenic are completely extracted.
- the pretreatment method of the present invention is a simple and short time pretreatment of a sample for measuring the amount of cadmium, arsenic, zinc and / or manganese elements in a crop sample of grains, beans or seeds. Therefore, it is extremely useful as a pretreatment method performed near the site.
Abstract
Description
(1)穀粒類、豆類または種子類から選択される農作物試料中の、カドミウム、砒素、亜鉛、マンガン、銅、鉛、及びクロムからなる群から選択される少なくとも一種の元素の量を測定するための試料の前処理方法であって、
(i)試料を粗粉砕する工程、
(ii)粗粉砕された試料に水を添加して加熱し、試料中に含まれるβデンプンをαデンプンに変換する工程、
(iii)試料に酵素を添加して試料中のαデンプンを糖に変換する工程、
(iv)試料に塩酸を添加して試料中の測定元素を抽出する工程、及び
(v)抽出された液体から固形物を除去する工程
を含むことを特徴とする方法。
(2)試料の粗粉砕が、試料の水分率測定時に行なわれる粗粉砕であることを特徴とする(1)に記載の方法。
(3)粗粉砕された試料の加熱がマイクロ波によって行なわれることを特徴とする(1)または(2)に記載の方法。
(4)酵素として、アミラーゼ、プロテアーゼ及びセルラーゼを使用することを特徴とする(1)~(3)のいずれかに記載の方法。
(5)試料への酵素添加後及び塩酸添加後にそれぞれ試料を振とうすることを特徴とする(1)~(4)のいずれかに記載の方法。
(6)穀粒類が米、麦または蕎麦であり、豆類が大豆または落花生であり、種子類がゴマまたはナタネであることを特徴とする(1)~(5)のいずれかに記載の方法。
(7)(1)~(6)のいずれかに記載の方法を実施するための器具及び試薬を含むことを特徴とするキット。 That is, the present invention has the following configurations (1) to (7).
(1) Measure the amount of at least one element selected from the group consisting of cadmium, arsenic, zinc, manganese, copper, lead and chromium in a crop sample selected from grains, beans or seeds. A sample pretreatment method for
(I) a step of coarsely crushing the sample;
(Ii) adding water to the coarsely pulverized sample and heating to convert β starch contained in the sample into α starch;
(Iii) adding an enzyme to the sample to convert alpha starch in the sample into sugar;
(Iv) adding hydrochloric acid to the sample to extract the measurement element in the sample, and (v) removing the solid matter from the extracted liquid.
(2) The method according to (1), wherein the coarse pulverization of the sample is coarse pulverization performed at the time of measuring the moisture content of the sample.
(3) The method according to (1) or (2), wherein the coarsely pulverized sample is heated by microwaves.
(4) The method according to any one of (1) to (3), wherein amylase, protease and cellulase are used as the enzyme.
(5) The method according to any one of (1) to (4), wherein the sample is shaken after the enzyme is added to the sample and after the hydrochloric acid is added.
(6) The method according to any one of (1) to (5), wherein the grains are rice, wheat or buckwheat, the beans are soybeans or peanuts, and the seeds are sesame or rapeseed .
(7) A kit comprising a device and a reagent for performing the method according to any one of (1) to (6).
本発明の方法の対象とする測定元素は、カドミウム、砒素、亜鉛、マンガン、銅、鉛、及びクロムからなる群から選択される少なくとも一種の元素である。これらの元素は、人体にとって有害な影響を及ぼすので、農作物中の含有量を流通前に監視することが必要である。 The crop sample targeted by the method of the present invention is selected from grains, beans, or seeds, and these crops are protected by the cellulose layer on the surface, so that they are in the state at the time of harvest. Is characterized in that the treatment liquid such as hydrochloric acid does not reach the starch layer inside. Examples of grains include rice, wheat, and buckwheat, examples of beans include soybeans, red beans, and peanuts, and examples of seeds include sesame and rapeseed.
The element to be measured in the method of the present invention is at least one element selected from the group consisting of cadmium, arsenic, zinc, manganese, copper, lead, and chromium. Since these elements have a harmful effect on the human body, it is necessary to monitor the content in crops before distribution.
玄米50検体を近赤外水分計(株式会社ケツト科学研究所製、米麦水分計SP-1D3型)で粗粉砕した後、以下の抽出操作(本発明の前処理方法)に従って本発明法の抽出液を作成した。
抽出操作:
(i)水分計で粗粉砕した玄米試料1gをPPボトル(ポリプロピレンボトル)に入れ、続けてイオン交換水を2.5mL加える(図1の(i)参照)。
(ii)キャップをせずにPPボトルを電子レンジに入れて500W、30秒程度加熱した後、酵素溶液(アミラーゼ、プロテアーゼ、及びセルラーゼの等量混合溶液:10重量%濃度)2.5mLを加えてPPボトルのキャップを閉め、手で上下に10秒程度軽く振とうする(図1の(ii)参照)。
(iii)PPボトルのキャップをはずし、0.2N塩酸溶液を5mL加えてキャップをしっかりと閉めてから、手で上下に1分程度激しく振とうする(図1の(iii)参照)。
(iv)新しいPPボトルにNo.2濾紙をセットして、(iii)の溶液を全量濾過する(図1の(iv)参照)。 Example 1
After roughly pulverizing 50 brown rice samples with a near-infrared moisture meter (Ketto Scientific Laboratory Co., Ltd., rice wheat moisture meter SP-1D3 type), according to the following extraction operation (the pretreatment method of the present invention), An extract was prepared.
Extraction operation:
(I) 1 g of brown rice sample coarsely pulverized with a moisture meter is put into a PP bottle (polypropylene bottle), and then 2.5 mL of ion-exchanged water is added (see (i) in FIG. 1).
(Ii) Put PP bottle in microwave oven without cap and heat for about 500W for 30 seconds, then add 2.5mL of enzyme solution (Mixed solution of amylase, protease and cellulase: 10% by weight concentration) Close the cap of the PP bottle and shake it up and down by hand for about 10 seconds (see (ii) in FIG. 1).
(Iii) Remove the cap of the PP bottle, add 5 mL of 0.2N hydrochloric acid solution, close the cap tightly, and shake it up and down by hand for about 1 minute (see (iii) in FIG. 1).
(Iv) No. for new PP bottle. 2 Set filter paper and filter the whole solution of (iii) (see (iv) in FIG. 1).
玄米50検体を1検体づつラボ用微粉砕機(大阪ケミカル株式会社製、ラボミルサープラス LM-PLUS)で微粉砕した後、以下の抽出操作(従来法B)に従って従来法Bの抽出液を作成した。
抽出操作:
微粉砕した玄米試料1gをPPボトル(ポリプロピレンボトル)に入れ、続けて0.1N塩酸を10mL加える。このPPボトルを手で上下に1分程度激しく振とうした後、この溶液を、No.2濾紙をセットした新しいPPボトルに入れて全量濾過する(図2参照)。 Comparative Example 1
After pulverizing 50 brown rice samples one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), an extract of conventional method B is prepared according to the following extraction procedure (conventional method B). did.
Extraction operation:
1 g of the finely ground brown rice sample is put into a PP bottle (polypropylene bottle), and 10 mL of 0.1N hydrochloric acid is subsequently added. The PP bottle was shaken vigorously up and down for about 1 minute by hand, 2 Put in a new PP bottle with filter paper and filter the whole volume (see Fig. 2).
大豆20検体をメノウ乳鉢でたたいて粗粉砕した後、実施例1と同様に本発明の前処理方法に従って本発明法の抽出液を作成した。 Example 2
After 20 soybean samples were beaten in an agate mortar and coarsely pulverized, an extract of the method of the present invention was prepared according to the pretreatment method of the present invention in the same manner as in Example 1.
大豆20検体を1検体ずつラボ用微粉砕機(大阪ケミカル株式会社製、ラボミルサープラス LM-PLUS)で微粉砕した後、10検体ずつホットプレート上で30分程度きつね色になるまで焼成して試料とした(注釈:従来法Bの大豆測定では、焼成処理を行わないと、濾過による固液分離ができない)。この試料から、比較例1に記載の従来法Bと同様の抽出操作(ただし、振とう時間は30分に変更した)に従って従来法Bの抽出液を作成した。 Comparative Example 2
Twenty soybean samples are pulverized one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), and then 10 samples are baked on a hot plate for 30 minutes until the color becomes dark. A sample was prepared (note: in the conventional method B soybean measurement, solid-liquid separation by filtration is not possible without baking treatment). From this sample, an extract of conventional method B was prepared according to the same extraction procedure as in conventional method B described in Comparative Example 1 (however, the shaking time was changed to 30 minutes).
ゴマ20検体をメノウ乳鉢でたたいて粗粉砕した後、実施例1と同様に本発明法の前処理方法に従って本発明法の抽出液を作成した。 Example 3
After 20 sesame samples were beaten in an agate mortar and coarsely pulverized, the extract of the method of the present invention was prepared according to the pretreatment method of the present invention in the same manner as in Example 1.
ゴマ20検体を1検体ずつラボ用微粉砕機(大阪ケミカル株式会社製、ラボミルサープラス LM-PLUS)で微粉砕した後、比較例1と同様に従来法Bに従って従来法Bの抽出液を作成した。 Comparative Example 3
After pulverizing 20 sesame samples one by one with a laboratory pulverizer (labor mill surplus LM-PLUS, manufactured by Osaka Chemical Co., Ltd.), an extract of conventional method B was prepared according to conventional method B as in Comparative Example 1. did.
玄米試料10検体を用いて、実施例1の本発明の前処理方法の抽出操作のうち、(i)に示す水の添加と(ii)に示す電子レンジによる加熱を行わなかった以外は実施例1と同様にして比較法Aの抽出液を作成した。 Comparative Example 4
Example of the extraction operation of the pretreatment method of the present invention of Example 1 using 10 brown rice samples except that the addition of water shown in (i) and the heating by the microwave oven shown in (ii) were not performed. In the same manner as in Example 1, an extract of Comparative Method A was prepared.
玄米試料10検体を用いて、実施例1の本発明の前処理方法の抽出操作のうち、(ii)に示す酵素の添加を行わなかった以外は実施例1と同様にして比較法Bの抽出液を作成した。 Comparative Example 5
Extraction of Comparative Method B in the same manner as in Example 1 except that the enzyme shown in (ii) was not added in the extraction operation of the pretreatment method of the present invention in Example 1 using 10 brown rice samples. A liquid was created.
玄米試料10検体を用いて、実施例1の本発明の前処理方法の抽出操作のうち、水分計による粗粉砕を行わなかった以外は実施例1と同様にして比較法Cの抽出液を作成した。 Comparative Example 6
Using the 10 brown rice samples, an extract of Comparative Method C was prepared in the same manner as in Example 1 except that coarse pulverization was not performed using the moisture meter in the extraction operation of the pretreatment method of the present invention of Example 1. did.
玄米試料10検体を用いて、実施例1の本発明の前処理方法の抽出操作のうち、水分計による粗粉砕を行なう代わりに、木槌により玄米を2つに割った以外は実施例1と同様にして本発明法の抽出液を作成した。 Example 4
Of the extraction operation of the pretreatment method of the present invention of Example 1 using 10 brown rice samples, Example 1 except that the brown rice was divided into two with a mallet instead of performing coarse pulverization with a moisture meter. Similarly, an extract of the method of the present invention was prepared.
玄米試料10検体を用いて、実施例1の本発明の前処理方法の抽出操作のうち、水分計による粗粉砕を行なう代わりに、木槌により玄米を20片程度まで割った以外は実施例1と同様にして本発明の抽出液を作成した。 Example 5
In the extraction operation of the pretreatment method of the present invention in Example 1 using 10 brown rice samples, Example 1 except that brown rice was broken up to about 20 pieces with a mallet instead of performing coarse pulverization with a moisture meter. In the same manner, an extract of the present invention was prepared.
玄米38検体を近赤外水分計(株式会社ケツト科学研究所製、米麦水分計SP-1D3型)で粗粉砕した後、実施例1と同様に本発明の前処理方法に従って本発明法の抽出液を作成した。 Example 6
After roughly pulverizing 38 specimens of brown rice with a near-infrared moisture meter (Ketto Scientific Laboratory Co., Ltd., rice wheat moisture meter SP-1D3 type), in the same manner as in Example 1, according to the pretreatment method of the present invention, An extract was prepared.
Claims (7)
- 穀粒類、豆類または種子類から選択される農作物試料中の、カドミウム、砒素、亜鉛、マンガン、銅、鉛、及びクロムからなる群から選択される少なくとも一種の元素の量を測定するための試料の前処理方法であって、
(i)試料を粗粉砕する工程、
(ii)粗粉砕された試料に水を添加して加熱し、試料中に含まれるβデンプンをαデンプンに変換する工程、
(iii)試料に酵素を添加して試料中のαデンプンを糖に変換する工程、
(iv)試料に塩酸を添加して試料中の測定元素を抽出する工程、及び
(v)抽出された液体から固形物を除去する工程
を含むことを特徴とする方法。 Sample for measuring the amount of at least one element selected from the group consisting of cadmium, arsenic, zinc, manganese, copper, lead and chromium in a crop sample selected from grains, beans or seeds The pre-processing method of
(I) a step of coarsely crushing the sample;
(Ii) adding water to the coarsely pulverized sample and heating to convert β starch contained in the sample into α starch;
(Iii) adding an enzyme to the sample to convert alpha starch in the sample into sugar;
(Iv) adding hydrochloric acid to the sample to extract the measurement element in the sample, and (v) removing the solid matter from the extracted liquid. - 試料の粗粉砕が、試料の水分率測定時に行なわれる粗粉砕であることを特徴とする請求項1に記載の方法。 2. The method according to claim 1, wherein the coarse pulverization of the sample is coarse pulverization performed at the time of measuring the moisture content of the sample.
- 粗粉砕された試料の加熱がマイクロ波によって行なわれることを特徴とする請求項1または2に記載の方法。 The method according to claim 1 or 2, wherein the coarsely pulverized sample is heated by microwaves.
- 酵素として、アミラーゼ、プロテアーゼ及びセルラーゼを使用することを特徴とする請求項1~3のいずれかに記載の方法。 The method according to any one of claims 1 to 3, wherein amylase, protease and cellulase are used as the enzyme.
- 試料への酵素添加後及び塩酸添加後にそれぞれ試料を振とうすることを特徴とする請求項1~4のいずれかに記載の方法。 5. The method according to claim 1, wherein the sample is shaken after the enzyme is added to the sample and after the hydrochloric acid is added.
- 穀粒類が米、麦または蕎麦であり、豆類が大豆または落花生であり、種子類がゴマまたはナタネであることを特徴とする請求項1~5のいずれかに記載の方法。 The method according to any one of claims 1 to 5, wherein the grains are rice, wheat or buckwheat, the beans are soybeans or peanuts, and the seeds are sesame or rapeseed.
- 請求項1~6のいずれかに記載の方法を実施するための器具及び試薬を含むことを特徴とするキット。 A kit comprising an instrument and a reagent for carrying out the method according to any one of claims 1 to 6.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2014551865A JP5666070B1 (en) | 2013-05-24 | 2014-05-23 | Sample pretreatment method for measuring the amount of harmful elements in crop samples |
| CN201480002580.6A CN105452841A (en) | 2013-05-24 | 2014-05-23 | Sample pre-treatment method for measuring the quantity of toxic elements in agricultural produce samples |
| HK16109434.7A HK1221284A1 (en) | 2013-05-24 | 2016-08-08 | Sample pre-treatment method for measuring the quantity of toxic elements in agricultural produce samples |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013-109980 | 2013-05-24 | ||
| JP2013109980 | 2013-05-24 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014189123A1 true WO2014189123A1 (en) | 2014-11-27 |
Family
ID=51933674
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2014/063639 WO2014189123A1 (en) | 2013-05-24 | 2014-05-23 | Sample pre-treatment method for measuring the quantity of toxic elements in agricultural produce samples |
Country Status (4)
| Country | Link |
|---|---|
| JP (1) | JP5666070B1 (en) |
| CN (1) | CN105452841A (en) |
| HK (1) | HK1221284A1 (en) |
| WO (1) | WO2014189123A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111579349A (en) * | 2020-06-09 | 2020-08-25 | 福建天甫电子材料有限公司 | Method for preparing ICP-MS sample by concentration |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2016057301A (en) * | 2014-09-08 | 2016-04-21 | 株式会社住化分析センター | Method for preparing sample for analysis of hazardous substance, kit for preparation, and method for analysis of hazardous substance |
| JP6764160B2 (en) * | 2016-09-24 | 2020-09-30 | 学校法人中部大学 | Preparation method of sample for hazard analysis |
| CN109164220B (en) * | 2018-09-26 | 2021-07-09 | 贵州省三好食品开发有限公司 | Method for detecting heavy metal content of bean product raw material |
| CN109507247B (en) * | 2018-12-11 | 2021-09-24 | 邯郸学院 | Method for identifying millet producing area based on nonlinear chemical fingerprint spectrum |
| CN113866224B (en) * | 2021-12-01 | 2022-02-15 | 广东利诚检测技术有限公司 | Method for detecting content of harmful metal elements in food based on magnetoelectric coupling method |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5750658A (en) * | 1980-09-12 | 1982-03-25 | Mitsubishi Chem Ind Ltd | Rapid wet-decomposing method of sample |
| JP2005265523A (en) * | 2004-03-17 | 2005-09-29 | Japan Atom Energy Res Inst | Pretreatment method for analyzing metal in food using fibrous solid-phase extraction material |
| JP2006226986A (en) * | 2005-01-24 | 2006-08-31 | Central Res Inst Of Electric Power Ind | Cadmium measuring method and pretreatment device therefor |
| JP2009219437A (en) * | 2008-03-17 | 2009-10-01 | National Agriculture & Food Research Organization | Method for eliminating allergen from food raw material originating in grain seeds |
| JP2010133949A (en) * | 2008-11-06 | 2010-06-17 | Sumika Chemical Analysis Service Ltd | Pretreatment method for cadmium measurement sample, method for separating cadmium, column device for separating cadmium and use of the same |
| JP2011145288A (en) * | 2009-12-15 | 2011-07-28 | Sumika Chemical Analysis Service Ltd | Impurity removal method for heavy metal measuring sample, impurity removing agent, and measuring method of heavy metal |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS57152888A (en) * | 1981-03-14 | 1982-09-21 | Mitsui Eng & Shipbuild Co Ltd | Alcoholic fermentation of raw potato by enzymatic process |
| US4645745A (en) * | 1984-02-27 | 1987-02-24 | Hach Company | Digestion process |
| CN101041699B (en) * | 2006-03-22 | 2010-05-12 | 中国农业科学院农产品加工研究所 | Sweet potato pectin and production technique thereof |
| WO2008139485A1 (en) * | 2007-05-11 | 2008-11-20 | Tata Steel Limited | A process for the determination of hexavalent chromium in organic reductant treated chromite materials |
| CN102183392B (en) * | 2010-03-29 | 2012-08-15 | 天津师范大学 | Optimum digestion method in measuring heavy metals in waste rubber granules |
| CN102246914B (en) * | 2011-07-07 | 2013-01-02 | 暨南大学 | Method for leaching heavy metals in protein foods by using diluted acid and application thereof |
| CN102539742B (en) * | 2011-12-13 | 2014-12-03 | 暨南大学 | Method for detecting heavy metals in carbohydrate agricultural products and application thereof |
-
2014
- 2014-05-23 CN CN201480002580.6A patent/CN105452841A/en active Pending
- 2014-05-23 WO PCT/JP2014/063639 patent/WO2014189123A1/en active Application Filing
- 2014-05-23 JP JP2014551865A patent/JP5666070B1/en active Active
-
2016
- 2016-08-08 HK HK16109434.7A patent/HK1221284A1/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5750658A (en) * | 1980-09-12 | 1982-03-25 | Mitsubishi Chem Ind Ltd | Rapid wet-decomposing method of sample |
| JP2005265523A (en) * | 2004-03-17 | 2005-09-29 | Japan Atom Energy Res Inst | Pretreatment method for analyzing metal in food using fibrous solid-phase extraction material |
| JP2006226986A (en) * | 2005-01-24 | 2006-08-31 | Central Res Inst Of Electric Power Ind | Cadmium measuring method and pretreatment device therefor |
| JP2009219437A (en) * | 2008-03-17 | 2009-10-01 | National Agriculture & Food Research Organization | Method for eliminating allergen from food raw material originating in grain seeds |
| JP2010133949A (en) * | 2008-11-06 | 2010-06-17 | Sumika Chemical Analysis Service Ltd | Pretreatment method for cadmium measurement sample, method for separating cadmium, column device for separating cadmium and use of the same |
| JP2011145288A (en) * | 2009-12-15 | 2011-07-28 | Sumika Chemical Analysis Service Ltd | Impurity removal method for heavy metal measuring sample, impurity removing agent, and measuring method of heavy metal |
Non-Patent Citations (1)
| Title |
|---|
| KAORU ABE ET AL.: "Nogyo ni Kakawaru Kankyo no Choki Monitering Immunochromato Kit o Mochiita Nosanbutsu no Cadmium Nodo no Kan'i Sokuteiho", NATIONAL INSTITUTE FOR AGRO-ENVIRONMENTAL SCIENCES, vol. 27, 31 March 2011 (2011-03-31), pages 70 - 71 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111579349A (en) * | 2020-06-09 | 2020-08-25 | 福建天甫电子材料有限公司 | Method for preparing ICP-MS sample by concentration |
Also Published As
| Publication number | Publication date |
|---|---|
| HK1221284A1 (en) | 2017-05-26 |
| JPWO2014189123A1 (en) | 2017-02-23 |
| CN105452841A (en) | 2016-03-30 |
| JP5666070B1 (en) | 2015-02-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP5666070B1 (en) | Sample pretreatment method for measuring the amount of harmful elements in crop samples | |
| Jia et al. | Impact of microplastics on bioaccumulation of heavy metals in rape (Brassica napus L.) | |
| Chao et al. | Effect of biochar from peanut shell on speciation and availability of lead and zinc in an acidic paddy soil | |
| Lamble et al. | Microwave digestion procedures for environmental matrices. Critical Review | |
| Yafa et al. | A comparative study of acid-extractable and total digestion methods for the determination of inorganic elements in peat material by inductively coupled plasma-optical emission spectrometry | |
| Stylianou et al. | Effect of acid treatment on the removal of heavy metals from sewage sludge | |
| Andrade et al. | Comparison of ICP OES and LIBS analysis of medicinal herbs rich in flavonoids from Eastern Europe | |
| Khan et al. | Hazardous impact and translocation of vanadium (V) species from soil to different vegetables and grasses grown in the vicinity of thermal power plant | |
| Radulescu et al. | Studies concerning heavy metals accumulation of Carduus nutans L. and Taraxacum officinale as potential soil bioindicator species | |
| CN103575583A (en) | Method for rapidly and mildly extracting heavy metals from cereals | |
| Pinto et al. | Assessment of metal (loid) s phytoavailability in intensive agricultural soils by the application of single extractions to rhizosphere soil | |
| WO2012137314A1 (en) | Method and apparatus for measuring cadmium in food | |
| Kim et al. | Phytoavailability-based threshold values for cadmium in soil for safer crop production | |
| Jena et al. | Study of heavy metal distribution in medicinal plant basil | |
| Khalili Sadrabad et al. | Heavy metal accumulation in soybeans cultivated in Iran, 2015-2016 | |
| JP5319485B2 (en) | Method and apparatus for measuring cadmium in food | |
| Belay et al. | Validation of a method for determining heavy metals in some ethiopian spices by dry ashing using atomic absorption spectroscopy | |
| WO2022187974A1 (en) | Processes for extracting muscimol from amanita muscaria | |
| CN104198513A (en) | Quick determination method for cadmium element in grains with X-ray fluorescent spectrometry | |
| Busato et al. | Humic substances isolated from residues of sugar cane industry as root growth promoter | |
| CN104181182A (en) | Method for rapidly measuring arsenic element in grain by X fluorescent spectrometry | |
| Bhadkariya et al. | Remediation of cadmium by Indian mustard (Brassica juncea L.) from cadmium contaminated soil: a phytoextraction study | |
| CN110813230A (en) | Composite biochar and preparation method and application thereof | |
| CN104181183A (en) | Method for rapidly measuring lead element in grain by X fluorescent spectrometry | |
| CN104198665A (en) | Method for rapidly determining cadmium in cereal crops |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201480002580.6 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 2014551865 Country of ref document: JP Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 14800868 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 14800868 Country of ref document: EP Kind code of ref document: A1 |