WO2013191270A1 - Glass and optical element production method - Google Patents
Glass and optical element production method Download PDFInfo
- Publication number
- WO2013191270A1 WO2013191270A1 PCT/JP2013/067050 JP2013067050W WO2013191270A1 WO 2013191270 A1 WO2013191270 A1 WO 2013191270A1 JP 2013067050 W JP2013067050 W JP 2013067050W WO 2013191270 A1 WO2013191270 A1 WO 2013191270A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- glass
- melting
- optical
- sample
- atmosphere
- Prior art date
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03B—MANUFACTURE, SHAPING, OR SUPPLEMENTARY PROCESSES
- C03B3/00—Charging the melting furnaces
- C03B3/02—Charging the melting furnaces combined with preheating, premelting or pretreating the glass-making ingredients, pellets or cullet
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03B—MANUFACTURE, SHAPING, OR SUPPLEMENTARY PROCESSES
- C03B5/00—Melting in furnaces; Furnaces so far as specially adapted for glass manufacture
- C03B5/16—Special features of the melting process; Auxiliary means specially adapted for glass-melting furnaces
- C03B5/167—Means for preventing damage to equipment, e.g. by molten glass, hot gases, batches
-
- C—CHEMISTRY; METALLURGY
- C03—GLASS; MINERAL OR SLAG WOOL
- C03B—MANUFACTURE, SHAPING, OR SUPPLEMENTARY PROCESSES
- C03B5/00—Melting in furnaces; Furnaces so far as specially adapted for glass manufacture
- C03B5/16—Special features of the melting process; Auxiliary means specially adapted for glass-melting furnaces
- C03B5/235—Heating the glass
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P40/00—Technologies relating to the processing of minerals
- Y02P40/50—Glass production, e.g. reusing waste heat during processing or shaping
- Y02P40/57—Improving the yield, e-g- reduction of reject rates
Definitions
- the present invention relates to a glass having excellent transmittance and a method of manufacturing an optical element.
- the high refractive index optical glass usually contains a large amount of high refractive index components such as Ti, Nb, W and Bi as glass components. These components are easily reduced in the melting process of the glass, and these reduced components absorb light on the short wavelength side of the visible light range, so the glass may be colored (hereinafter referred to as "reduced color" ) Increases.
- the high refractive index component which is easily reduced is reacted (oxidized) with a noble metal material such as platinum which is widely used as a crucible material, and causes a noble metal ion generated by oxidizing the noble metal to be dissolved in the molten glass.
- a noble metal material such as platinum which is widely used as a crucible material
- Noble metal ions dissolved in the molten glass absorb visible light, thereby increasing the color of the glass.
- Patent Document 1 proposes a technique of bubbling non-oxidizing gas during glass melting and a technique of reheating and heat-treating the glass once obtained.
- oxygen in the atmosphere may react with a noble metal material such as platinum which is a material of the melting container.
- a noble metal material such as platinum which is a material of the melting container.
- platinum dioxide PtO 2
- Pt 4+ platinum ions
- Patent Document 1 the technique of bubbling non-oxidizing gas as in Patent Document 1 alone can not sufficiently suppress the dissolution of a noble metal such as platinum in the glass, and still significantly reduces the coloring of the high refractive index optical glass. It was difficult.
- This invention is made in view of such a situation, and an object of this invention is to provide the manufacturing method of the glass excellent in the transmittance
- the gist of the present invention for the purpose of solving such problems is as follows. [1] A glass raw material containing at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 is heated and melted in a melting vessel in a melting step (i) to obtain a molten glass, The manufacturing method of the glass which performs operation which raises the moisture content in molten glass.
- a step (ii) of flowing the molten glass out of the melting vessel The method further includes the step (iii) of forming the molten glass, The method for producing glass according to any one of the above [1] to [5], wherein at least one of the step (ii) and the step (iii) is performed in an oxidizing atmosphere.
- the method further includes the step (iv) of heat-treating the glass, The method for producing glass according to any one of the above [1] to [6], wherein the step (iv) is performed in an oxidizing atmosphere.
- the transmittance of glass can be dramatically improved.
- the amount of the noble metal such as platinum dissolved in the glass can be significantly reduced.
- the method for producing glass of the present embodiment heats and melts a glass material containing at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 in a melting vessel to obtain a molten glass
- a melting vessel to obtain a molten glass
- an operation to increase the amount of water in the molten glass is performed.
- the operation to increase the moisture in the molten glass is preferably to supply moisture into the melting vessel (hereinafter, first embodiment), or the glass material contains moisture.
- the melting container is substantially sealed (second embodiment).
- the operation which raises the moisture content in molten glass is raising the moisture content in molten glass rather than the moisture content in molten glass when not performing such operation. If the operation to increase the water content in the molten glass is not performed, the water content in the molten glass decreases with the passage of time. An operation of reducing and suppressing the reduction of the amount of water in the molten glass is also included in the operation of increasing the amount of water in the molten glass.
- the glass produced by the production method of the present embodiment includes a high refractive index component (at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 ), and the glass is strongly colored. Even in this case, the coloration of the glass can be significantly reduced by heat treating the glass in an oxidizing atmosphere in a later step. That is, after heat treatment, the glass obtained by the manufacturing method of the present embodiment has little color and has extremely excellent transmittance.
- the present inventors estimate that the reason why such an effect can be obtained is as follows.
- the molten glass Melting on the reduction side can suppress dissolution of metal ions (ions of the metal constituting the melting vessel) into the molten glass.
- the melting vessel is alloyed as described above.
- the degree of coloring of the glass is enhanced by reduction of the high refractive index component even if the molten glass is not excessively reduced, the degree of reduction of coloring is small even if the glass is subjected to heat treatment in a later step.
- the coloring can be greatly reduced by heat-treating the glass once obtained while forming a state in which the metal material constituting the melting vessel is ionized and is not dissolved in the molten glass. You can make a glass.
- H + is considered to be suitable as such an ion, but to make H + more mobile, introduce OH ⁇ into the glass structure so that H + can hop from OH ⁇ . It is believed that this can increase the oxidation rate during heat treatment.
- a raw material component having a high water content may be used as a glass raw material, such as a hydroxide such as orthophosphoric acid, aluminum hydroxide or calcium hydroxide.
- a glass raw material such as a hydroxide such as orthophosphoric acid, aluminum hydroxide or calcium hydroxide.
- Moisture evaporates in the process of melting into molten glass and in the state of high temperature molten glass.
- the batch raw material is initially contained in the batch raw material The contained water is lost during culletization, and the water is also lost during the remelting process in the melting vessel. Therefore, regardless of which method is employed to produce the glass, the water content of the glass is usually extremely low. As a result, this glass can not be heat-treated to significantly reduce its color.
- the melting vessel may be substantially sealed in the heating and melting process while using a glass material containing water. Also in this case, since the airtightness of the melting vessel is enhanced, when the glass raw material is heated and melted in the melting vessel, the moisture contained in the glass raw material can be suppressed from transpiration to the outside of the melting vessel. Therefore, as a result, it is easy to increase the water content of the glass.
- the glass raw material containing water can be obtained using orthophosphoric acid, a hydroxide, etc. as mentioned above.
- the glass raw material containing water can be obtained also by introduce
- the cullet raw material glass raw material containing water
- the water adhering to the surface is removed when the cullet is dried.
- the water taken into the cullet contributes to the increase of the water content in the molten glass obtained by the remelting (heating and melting process).
- a glass component having a strong action of taking in and holding water in the glass that is, a phosphoric acid component (eg, orthophosphate, metaphosphate, pyrophosphate etc.) It is also effective to use
- a glass material containing a phosphoric acid component it is particularly preferable to use a glass material containing a phosphoric acid component.
- the phosphoric acid component include orthophosphoric acid (H 3 PO 4 ), and in addition, pyrophosphoric acid (H 4 P 2 O 7 ) and various phosphates (especially hydration) )
- various known phosphoric acid compounds may be used, and two or more kinds may be used in combination.
- the water content of the obtained glass is high, and as a result, when the glass is heat-treated, it is possible to significantly reduce the coloration.
- the water content of the glass is higher than when the second embodiment is performed alone. Can be further enhanced, and the color reduction effect is also high.
- the operation of increasing the water content in the molten glass is performed by supplying the water into the melting container in the heating and melting step.
- water can be supplied to the molten glass from the outside, and as a result, the water content of the glass can be increased.
- the manner of supplying water into the melting vessel is not particularly limited, but the first supplying manner of supplying water vapor into the atmosphere near the liquid surface of the molten glass, the second supplying of water vapor while bubbling water vapor into the molten glass It is preferable that it is any selected from the supply aspect and the 3rd supply aspect which combined the 1st supply aspect and the 2nd supply aspect.
- Moisture is usually preferably supplied in the form of water vapor (gas), but for example, it is a glass raw material powder (solid) having a high water content, the remaining components excluding water being substantially the same components as glass. It may be supplied in a form.
- the operation of increasing the water content in the molten glass is performed by using a glass material containing water and by substantially sealing the melting container in the heating and melting step. That is, the molten glass containing water is sealed in a very narrow space called a melting vessel. For this reason, it is possible to suppress the evaporation of water by increasing the water vapor partial pressure in the atmosphere in the vicinity of the liquid surface of the molten glass. As a result, the water content of the glass can be increased.
- the method for substantially sealing the melting container is not particularly limited, for example, if the melting container is a container having an opening, the opening of the melting container may be covered. In this case, the heating and melting process is performed in a state where the molten glass is confined in a sealed space surrounded by the melting vessel and the lid. The lid may simply be placed on the melting vessel so as to close the opening of the melting vessel. In this case, if the pressure in the sealed melting container is increased, it is possible to ensure a substantially sealed state in which the gas in the melting container gradually leaks to the outside. However, after closing the opening of the melting vessel, a pressing force is applied so that the lid is in pressure contact with the opening so that the lid is strongly fixed to the melting vessel, or the opening is sealed, etc. And the airtightness in the melting vessel may be further enhanced.
- the opening is opened other than the embodiment in which the opening of the melting container is directly covered or the opening is directly sealed.
- the melting container in the state where the part is open is disposed in a closed melting container storage chamber in the melting furnace. Then, the embodiments (1) to (3) may be combined appropriately.
- the operation of increasing the amount of water in the molten glass is carried out according to the first embodiment in the method for producing glass of this embodiment. That is, it is preferable to perform at least one of a process of adding water vapor to the melting atmosphere and a process of bubbling a gas containing water vapor in the molten material as the operation of increasing the water content in the molten glass.
- the method of adding water vapor to the melting atmosphere is not particularly limited.
- a connecting pipe is inserted into the crucible through an opening provided in the melting apparatus, and a gas containing water vapor is optionally passed through this pipe.
- the method of supplying to the space in a cage etc. are mentioned.
- the flow rate of the gas containing water vapor supplied to the space in the crucible is not particularly limited, and can be adjusted based on the measurement result of ⁇ OH of the experimentally produced glass.
- a relatively small amount of steam can be supplied to obtain a glass having a desired ⁇ OH.
- the volume in the glass melting furnace becomes larger than the volume in the crucible, so to set ⁇ OH to a desired value, A relatively large amount of water vapor will be supplied into the glass melting furnace.
- the flow rate of gas, the flow rate of steam, the additional flow rate of atmosphere, and the feed rate of steam are values converted to 25 ° C. and 1 atm.
- a well-known method can be used.
- a platinum or platinum alloy pipe is inserted into the melt in the melting vessel, and a gas containing water vapor is blown into the melt through the pipe, near the bottom of the melting vessel consisting of the same material as the material of the melting vessel.
- the bubble diameter of the gas containing water vapor blown into the melt is preferably 0.01 to 100 mm in diameter, more preferably 0.1 to 30 mm. By setting it as the said range, it is thought that the moisture content in molten glass can be raised effectively. In addition, when the bubble diameter is too small, there is a problem that the bubbling pipe inserted into the melt tends to be clogged.
- the flow rate of the gas containing water vapor blown into the melt is not particularly limited, and can be adjusted based on the measurement result of ⁇ OH of the experimentally produced glass.
- the ⁇ OH of experimentally produced glass is measured, and if the measurement result is smaller than the desired value, the gas flow rate is increased, and conversely, if the measurement result is larger than the desired ⁇ OH value, the gas flow rate Make adjustments to reduce
- the ⁇ OH of glass may be determined experimentally and the flow rate of gas may be adjusted from the measurement result.
- the glass having the desired ⁇ OH can be produced by feeding back the supply amount of water vapor, that is, the flow rate of the gas, to the next production based on the measured value of ⁇ OH of the experimentally produced glass.
- the content of water vapor in the gas containing water vapor is preferably 10% by volume or more, more preferably 20% by volume or more, still more preferably 30% by volume or more, more preferably 40% by volume or more, still more preferably 50% by volume
- the content is more preferably 60% by volume or more, still more preferably 70% by volume or more, particularly preferably 80% by volume or more, and still more preferably 90% by volume or more.
- the content of water vapor is preferably as high as possible, and by setting the content to the above range, in particular, in the finally obtained glass, the color reduction effect can be enhanced, the content of noble metal can be reduced, and the clarity is improved. can do.
- the gas containing water vapor may be one produced or a commercially available one, and may be a mixed gas with another gas.
- another gas air etc. are mentioned, for example.
- the molten material may be stirred in a melting atmosphere to which water vapor is added.
- the operation of increasing the water content in the molten glass is the first operation. It is particularly preferred to carry out the embodiment in combination with the second embodiment.
- the melting vessel is platinum (Pt)
- Pt platinum
- melting of glass is performed in the atmosphere, and oxygen in the atmosphere may react with a noble metal material such as platinum which is a material of the melting vessel.
- a noble metal material such as platinum which is a material of the melting vessel.
- platinum dioxide PtO 2
- Pt 4+ platinum ions
- Noble metal ions dissolved in the molten glass absorb visible light, so the coloring of the glass tends to increase.
- dissolution of metal ions into the molten glass can be suppressed by performing an operation to increase the amount of water in the molten glass.
- the partial pressure of oxygen in the melting atmosphere is reduced by the operation of increasing the amount of water in the molten glass (for example, supplying water into the melting container, etc.), and a platinum material which is a material such as a melting container Etc. to be oxidized.
- platinum dioxide and platinum ions (Pt 4+ ) formed by the reaction of oxygen in the melting atmosphere with a platinum material and the like can be effectively prevented from melting into the molten material (glass), and platinum is obtained in the obtained glass
- the amount of penetration of (Pt) is further reduced.
- the glass manufactured by the manufacturing method of this embodiment has very little content of noble metals, such as Pt originating in manufacturing tools, such as a fusion container. Therefore, there is little coloring of the glass by ultraviolet irradiation called solarization. Therefore, when such a glass is used as an optical element, for example, the secular change of the transmittance is small. Moreover, when fixing an optical element using an ultraviolet curing adhesive, even if it irradiates an ultraviolet-ray to an optical element, the effect that a transmittance
- the content of the noble metal in the obtained glass is 4 ppm or less from the viewpoints of reduction of coloring of the glass due to the noble metal ion, improvement of transmittance, reduction of solarization, reduction of foreign metal particles, and the like.
- the lower limit value of the content of the noble metal is preferably as low as possible, and 3 ppm, 2.7 ppm, 2.5 ppm, 2.2 ppm, 2.0 ppm, 1.8 ppm, 1.6 ppm, 1.4 ppm, 1.2 ppm, 1.1 ppm, It is even more preferable that the upper limit value is lower in the order of 1.0 ppm and 0.9 ppm.
- the lower limit of the content of the noble metal is not particularly limited, it is unavoidably contained in the order of 0.001 ppm.
- noble metals include simple metals such as Pt, Au, Rh and Ir, and alloys such as Pt alloys, Au alloys, Rh alloys, and Ir alloys.
- Pt or a Pt alloy which is excellent in heat resistance and corrosion resistance, is preferable as the melting vessel material and the melting tool material. Therefore, with regard to a glass produced by using a Pt or Pt alloy melting container or a melting tool, the content of Pt contained in the glass is preferably 4 ppm or less.
- the more preferable upper limit of the content of Pt is the same as the more preferable upper limit of the content of the noble metal contained in the glass.
- the lower limit of the content of Pt is not particularly limited, but unavoidably, about 0.001 ppm is included.
- the clarity can be significantly improved.
- a homogeneous, low bubble glass is required.
- a fining step for releasing (defoaming) the dissolved gas in the molten glass, but the fining property of the glass is determined in the molten glass. It depends on the amount of dissolved gas.
- Such amount of dissolved gas is greatly influenced by the composition of the glass (especially the type of raw material), the melting time of the glass and the number of times of melting.
- dissolved gas can be supplemented in the melting process, the problem of clarity is solved.
- the glass manufactured by the manufacturing method of the present embodiment is to increase the amount of dissolved gas in the molten glass by performing an operation to increase the amount of water in the molten glass (for example, supplying water into the melting container, etc.) It is believed that That is, the moisture (for example, water vapor) actively introduced into the molten glass is considered to play a role as a dissolved gas and to improve the clarity of the glass.
- the glass manufacturing method of the present embodiment since the glass has excellent clarity, the time required for the clarification step can be shortened, and the productivity is improved.
- the clarification tank which performs a clarification process is also comprised with metal materials, such as platinum and a platinum alloy. Therefore, as the fining step takes a long time, problems such as the deterioration of the transmittance due to the dissolution of platinum ions into the melt become remarkable.
- the glass manufacturing method of the present embodiment since the time required for the fining step can be shortened, the contact time between the molten glass and the melting vessel or the like can be reduced, and a melt of noble metal ions such as platinum can be obtained. Penetration of is also considered to be able to reduce more.
- the use of the glass obtained by the manufacturing method of this embodiment is not specifically limited, For example, it can use suitably as optical glass.
- the optical glass containing a large amount of high refractive index components (Ti, Nb, W, Bi, etc.) has a remarkable problem of coloration (reduction color) of the glass, but the manufacturing method of this embodiment According to this, it is possible to easily obtain an optical glass having an excellent transmittance while having a high refractive index.
- the optical glass obtained by the manufacturing method of the present embodiment contains a large amount of the high refractive index component as described above, the reduced color can be efficiently reduced by heat treatment. Furthermore, even when a noble metal material such as platinum is used as a melting vessel or the like, it is possible to effectively suppress the dissolution of the noble metal material into the molten glass, and the color originating from the noble metal ion is extremely small.
- the optical glass obtained by the manufacturing method of such this embodiment has the outstanding transmittance
- the amount of dissolved gas in the molten glass can be increased by the operation of increasing the amount of water in the molten glass, and the clarity can be significantly improved. As a result, homogeneous optical glass with less bubbles can be obtained in a short time.
- the glass manufacturing method of the present embodiment publicly known methods can be appropriately adopted for the glass raw material preparation method, the glass raw material heating method, the melting method, and the molten glass forming method except for the matters described in the present specification.
- well-known materials can be appropriately used for the glass raw materials used in the method for producing glass of the present embodiment and the material constituting the melting vessel.
- a material which constitutes a fusion vessel etc. used at the time of preparation of glass a material (for example, metal material, quartz material, etc.) which usually has heat resistance and corrosion resistance in temperature and atmosphere which melts fusion glass is mentioned. It can be used appropriately.
- a molten product exhibiting significant corrosion may be formed, or the molten glass may react with the material constituting the melting vessel or the like to melt the melting vessel. Therefore, when selecting the material which comprises a fusion
- phosphate glass containing a high refractive index component glass comprising P 2 O 5 and at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3
- a molten product is produced that exhibits significant attack. Since such molten products tend to attack even corrosion resistant materials such as platinum, precious metal materials such as platinum are attacked by the above molten products, dissolve in the melt and are formed as foreign matter. And the problem of increasing the coloration of the glass.
- the melting vessel for heating and melting batch materials is selected from materials such as a melting vessel separately from the latter half of the melting process, the clarification process, etc. It is preferable to do.
- a melting container etc. which are used when heating and melting a batch raw material, containers and apparatus made from refractories, such as quartz, are suitable. Refractories such as quartz are corroded by the above-mentioned melt products, but even if they are corroded and mixed in the melt, they become part of the glass composition, so there are few problems like noble metal materials.
- a precious metal container or device made of platinum or platinum alloy since the problem that the molten product corrodes the precious metal material is small, it is preferable to use a precious metal container or device made of platinum or platinum alloy.
- B 2 O 3 and a borate glass containing a high refractive index component (a glass containing at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 )
- a molten product such as the above-mentioned phosphate glass also attacks noble metal materials
- refractory containers such as quartz tend to be severely attacked. Therefore, as a melting container etc., it is suitable to use containers and instruments made of precious metals such as platinum and platinum alloy which are not easily corroded in the process of manufacturing glass.
- the method for producing glass of the present embodiment it is preferable to use a metal material as a material for forming a melting vessel used for producing glass.
- the metal material it is preferable to use at least one selected from noble metals and noble metal alloys.
- the water content in the molten glass is maintained high as compared with a general method for producing glass.
- the molten glass is in a redox state in which the above-mentioned noble metal or noble metal alloy is not corroded by the molten glass, and the reduced high refractive index component is not alloyed with the material constituting the melting container. Can be maintained.
- platinum, gold or the like as the noble metal and a platinum alloy, gold alloy or the like as the noble metal alloy.
- the heating and melting step is usually a refining step for promoting the defoaming of the molten glass, in addition to the melting step for melting the molten glass by heating the glass raw material to make the molten glass; It is preferable to include a homogenization step in which the temperature of the molten glass after fining is lowered to a viscosity suitable for molding and the mixture is stirred and homogenized.
- a raw material corresponding to the glass component can be weighed and sufficiently mixed to obtain an optical glass having desired characteristics, and a mixed raw material (batch raw material) obtained by mixing and a mixed cullet can be used.
- a culletizing step (rough melt step) of rough melting and batching the batch material is carried out before the melting step (remelt step).
- the cullet is preferably pre-measured for refractive index measurement. If the measured value of the refractive index equals the desired value, the cullet is taken as the formulated cullet and if the measured value of the refractive index deviates from the desired value, the cullet with the refractive index higher than the desired value and the desired value The cullet having a low refractive index is mixed to form a compound cullet.
- the cullet is made of glass, it does not have to be homogeneous glass.
- the cullet may also contain air bubbles.
- the raw material of a batch raw material may be included.
- the composition and optical properties of cullet eg, refractive index, Abbe's number, etc.
- the composition and optical properties of cullet are remelted from cullet to form a homogeneous, bubble-free glass, and the composition and optical properties of this glass are respectively the composition of cullet and the optical properties Do.
- the melting vessel is made of a metal material
- the heating temperature of the glass during the heating and melting process is 800 to 1500 ° C. from the viewpoint of suppressing the ionization of the metal material and securing the water content in the glass. It is preferable to maintain it, more preferably, it is maintained at 1400 ° C. or less, more preferably, 1300 ° C. or less.
- the heating temperature of the glass during the heating and melting step is the highest in the fining step It is preferable to set to, that is, to melt the glass below the fining temperature.
- the time from the start to the end of the heating and melting process is extended, the reduction of high refractive index components promotes ionization of the metal material when the melting vessel is made of a metal material, and the moisture content in the glass tends to decrease. Will be shown. Therefore, it is preferable to set the time from the start to the end of the heating and melting process within 100 hours. The time from the start to the end of the heating and melting process may be appropriately adjusted depending on the size of the volume of the melting container and the like.
- the glass production method of the present embodiment is more preferably carried out by the rough melt-remelt method. That is, the method for producing glass of this embodiment is preferably It has a rough melting process to melt the compounded material to obtain cullet, and a remelt process to remelt the cullet to obtain glass, In at least one of the rough melting step and the remelting step, an operation to increase the amount of water in the molten glass is performed.
- the melting temperature (rough melting temperature) of the batch material at the time of rough melting is preferably in the range of 800 to 1400.degree.
- the temperature of the melt in the rough melt process is the same as the melt temperature of the cullet (remelt temperature) in the remelt process, in order to enhance the refining effect.
- the temperature is preferably lower than the melting temperature of cullet, and particularly preferably lower than the fining temperature in the remelt process.
- the dissolution time in the rough melt process can be appropriately adjusted in consideration of the volume of the crucible and the input amount of the batch material to the crucible, and for example, the dissolution time is 0.1 to 100 hours, more preferably 0.1. It may be in the range of -20 hours.
- the melting temperature (remelting temperature) of the prepared cullet in the remelt step is preferably in the range of 800 to 1,500 ° C. However, in order to further enhance the fining effect, it is preferable to make the remelting temperature lower than the fining temperature.
- the dissolution time in the remelt process can be appropriately adjusted in consideration of the volume of the crucible and the amount of the mixed cullet to be introduced into the crucible. For example, the dissolution time at the time of remelting is preferably 0.1 to 100 hours, more preferably 2 to It may be in the range of 20 hours.
- the atmosphere at the time of melting is not particularly limited, but water vapor is added to the melting atmosphere from the viewpoint of effectively increasing the amount of water in the molten glass. Is preferred.
- Melting atmosphere starts melting in melting atmosphere other than water vapor such as air atmosphere or nitrogen atmosphere first, and water vapor may be added to the melting atmosphere in the middle by operation to increase the amount of water in the molten glass, or melting atmosphere May be previously adjusted to a water vapor atmosphere.
- water vapor may be added to the melting atmosphere in the middle by operation to increase the amount of water in the molten glass, or melting atmosphere May be previously adjusted to a water vapor atmosphere.
- the partial pressure of water vapor in the melting atmosphere when performing the operation to increase the amount of water in the molten glass is higher than the partial pressure of water vapor in the atmosphere, and more preferably higher than the partial pressure of oxygen.
- the upper limit of the water vapor partial pressure is not particularly limited, and for example, the melting atmosphere can be entirely replaced with water vapor.
- the high partial pressure of water vapor in the melting atmosphere throughout the melting process can effectively prevent oxygen from reacting with the melting vessel made of a noble metal material such as platinum, and the amount of dissolution of Pt or the like in the glass can be reduced. It is possible to reduce and effectively prevent the deterioration (decrease) of the transmittance. Furthermore, by maintaining the amount of dissolved gas until just before the clarification step, the improvement effect of the clarity is enhanced.
- the melting step can also involve stirring of the melt for the purpose of homogenization of the melt.
- a well-known method can be used as a stirring method, For example, the method of bubbling gas to a molten material, the method of stirring by a stirring rod, etc. are mentioned.
- bubbling using a gas containing water vapor and stirring of the melt in a melting atmosphere to which water vapor is added are preferable from the viewpoint of homogenizing the melt and increasing the amount of water in the molten glass.
- a glass material containing at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 is heated and melted in a melting vessel.
- the method further includes a step (ii) of flowing the molten glass out of the melting container, and a step (iii) of forming the molten glass.
- the clarified and homogenized molten glass flows out from the glass outflow pipe attached to the bottom of the melting vessel.
- the temperature of the glass outflow pipe is adjusted and maintained so that the viscosity is suitable for forming in a temperature range where the flowing molten glass does not devitrify.
- any known forming method can be used as long as the molten glass in the melting container can be formed into a predetermined shape.
- molten glass may be poured into a mold to form a block, or a linear molten glass flow made to flow down from a pipe may be cut at a certain length (certain amount) to form a glass block.
- the shapes of the individual glasses obtained through the forming process may be largely dispersed.
- the obtained glass may be deeply colored, and the coloring can be reduced by performing heat treatment in a later step.
- the method for producing glass of the present embodiment it is preferable to perform at least one of the outflow step (ii) and the forming step (iii) in an oxidizing atmosphere. Thereby, the reduced color of the glass can be efficiently reduced.
- the reduced color derived from the high refractive index component can be reduced by heat treating the glass in an oxidizing atmosphere.
- the oxidation of Ti, Nb, W and Bi tends to proceed more rapidly as the temperature of the glass is higher.
- the glass as hot as possible ie the glass of the melting step (i)
- the glass of the melting step (i) is exposed to an oxidizing atmosphere.
- the melting vessel, the fining tank, etc. is made of a precious metal material etc.
- the glass of the melting step (i) is in contact with the precious metal material, so when the melting atmosphere is an oxidizing atmosphere, the precious metal material is an atmosphere Reacts with oxygen in the inside, and there are problems such as dissolution of precious metal ions in the glass.
- the glass in the outflow step (ii) and the forming step (iii) is lower in temperature than the glass in the melting step (i), but still kept at a sufficiently high temperature as compared to the glass cooled after forming It can be said. Therefore, even in these steps, the effect of reducing the color of the glass by exposing the glass to an oxidizing atmosphere can be fully expected. Furthermore, in the outflow step (ii) and the forming step (iii), since the glass does not come in contact with the noble metal material constituting the melting vessel or the like, it is considered that the above-mentioned problems hardly occur.
- the reduced color can be efficiently reduced without concern for the penetration of the noble metal material or the like into the molten glass. it can.
- the reduction color can be reduced more efficiently.
- the reduced color of the glass is reduced in at least one of the outflow step (ii) and the forming step (iii), so that bubbles and precipitates are formed in the glass after the forming step (iii).
- the manufacturing method of the glass of this embodiment preferably has a heat treatment process (iv) which heat-processes the manufactured glass.
- the heat treatment is preferably performed in an oxidizing atmosphere. This makes it possible to significantly reduce the coloration of the glass obtained.
- the glass obtained through the heat treatment step is less colored and highly transparent, that is, it has a high transmittance in the visible range.
- the heat treatment temperature and the heat treatment time may be appropriately set so as to obtain desired optical characteristics.
- the heat treatment temperature is preferably a temperature (Tg-100 ° C.) lower than the softening point of the glass and 100 ° C. lower than the glass transition temperature Tg.
- heat processing time can be shortened. Further, the heat treatment time can be shortened even if the oxygen partial pressure in the oxidizing atmosphere is increased. As described above, although the heat treatment time changes depending on the heat treatment temperature and the oxygen partial pressure in the oxidizing atmosphere, it may be set so that the coloration of the glass becomes a desired level.
- the heat treatment time is preferably typically 0.1 hour to 100 hours.
- the oxidizing atmosphere is an air atmosphere or an atmosphere having a higher oxygen partial pressure than air, preferably oxygen than air. There is a high partial pressure atmosphere.
- the method for setting it as an oxidative atmosphere is not specifically limited, For example, the method of supplying oxidative atmosphere gas etc. are mentioned.
- the oxidizing atmosphere gas may be a gas containing oxygen, and the oxygen concentration may be, for example, about the same as or higher than that of air.
- an oxidizing atmosphere gas for example, air, a gas obtained by adding oxygen to air, a gas substantially consisting only of oxygen, and the like can be mentioned.
- the glass obtained by the manufacturing method of the present embodiment contains a large amount of water by the operation of increasing the water content in the molten glass, and as described above, after passing through the heat treatment step (iv), the coloring is reduced and excellent Have a good transmittance.
- Moisture contained in such glass is, OH - as the infrared absorption due to, it is possible to quantitatively grasp a spectrophotometer.
- the water content in glass can be grasped
- ⁇ OH ⁇ [ln (B / A)] / t (1)
- t represents the thickness (mm) of the glass used to measure the external transmittance
- A represents a wavelength of 2500 nm when light is incident on the glass in parallel with the thickness direction
- B represents the external transmittance (%) at a wavelength of 2900 nm when light is incident on the glass in parallel with its thickness direction.
- ln is a natural logarithm.
- the unit of ⁇ OH is mm ⁇ 1 .
- external transmittance refers to the ratio of the intensity Iout of transmitted light transmitted through the glass to the intensity Iin of incident light incident on the glass (Iout / Iin), that is, the transmittance in consideration of surface reflection on the surface of the glass.
- the “internal transmittance” described later is the transmittance when there is no surface reflection on the surface of the glass (that is, the transmittance of the glass material itself constituting the glass). Each transmittance is obtained by measuring the transmission spectrum using a spectrophotometer.
- the ⁇ OH represented by the above formula (1) means the absorbance due to the hydroxyl group. Therefore, the concentration of water (and / or hydroxide ion, hereinafter, simply referred to as "water”) contained in the glass can be evaluated by evaluating ⁇ OH. That is, the higher the ⁇ OH, the higher the water content in the glass.
- the ⁇ OH of glass varies depending on the glass composition, production conditions, and the like.
- a glass having a glass composition that easily takes in water tends to have a higher ⁇ OH than a glass having a glass composition that does not easily take in water.
- it compares with the same glass composition by performing operation which raises the moisture content in molten glass, it exists in the tendency for (beta) OH to become high.
- the glass obtained by the manufacturing method of the present embodiment when evaluating whether the water content in the glass is increased, it is necessary to compare the ease with which water is taken in at a similar level.
- the value of ⁇ OH of glass is not particularly limited as long as it can be adjusted, but from the viewpoint of enhancing effects such as color reduction of glass and reduction of noble metal content in glass, The higher the value of ⁇ OH, the better.
- the glass produced by the manufacturing method of the present embodiment satisfies the following formula (1-2). ⁇ OHOH0.4891 ⁇ ln (1 / HR) +2.48 (1-2)
- ln is a natural logarithm.
- HR represents the total amount (mol%) of the content of each component of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 in the glass.
- the unit of ⁇ OH is mm ⁇ 1 .
- the result of whether the glass satisfies these formulas can also be an index for determining whether an operation to increase the amount of water in the molten glass has been performed.
- the glass produced by the production method of the present embodiment satisfies the following formula (1-3). ⁇ OH ⁇ 181.39 ⁇ nd -3 -325.75 ⁇ nd -2 + 194.85 ⁇ nd -1 -38.1 ⁇ (1-3)
- nd represents the refractive index of the glass.
- the unit of ⁇ OH is mm ⁇ 1 .
- the production method of the present embodiment it is preferable to carry out an operation to increase the amount of water in the molten glass to such an extent that ⁇ OH of the obtained glass satisfies the above equation (1-3).
- the partial pressure of oxygen in the melting atmosphere is sufficiently reduced, and even when the melting container is made of a noble metal material or the like, reaction between oxygen in the melting atmosphere and the noble metal material can be effectively prevented.
- the noble metal content in the obtained glass can be further reduced.
- the result of whether the glass satisfies these formulas can also be an index for determining whether an operation to increase the amount of water in the molten glass has been performed.
- the upper limit of ⁇ OH of glass obtained by the method for producing glass of the present embodiment is also different depending on the type of glass and production conditions, and is not particularly limited as long as it can be adjusted. Since the amount of volatile matter from the molten glass tends to increase as the ⁇ OH is increased, the ⁇ OH is preferably 10 mm -1 or less, more preferably 8 mm -1 or less, from the viewpoint of suppressing volatilization from the molten glass. preferably 6 mm -1 or less, more preferably 5 mm -1 or less, even more preferably 4 mm -1 or less, even more preferably 3 mm -1 or less, even more preferably to a 2 mm -1 or less.
- the ⁇ OH of the glass can be evaluated regardless of the presence or absence of coloring of the glass (presence or absence of reduced color). Further, since the heat treatment step (iv) is usually performed at a temperature lower than the softening point of the glass, the value of ⁇ OH of the glass does not substantially change before and after that, and it is measured before or after the heat treatment step (iv) May be Therefore, the ⁇ OH of the glass may be measured on any of the transparent glass having undergone the heat treatment step (iv) and the strongly colored glass not having undergone the heat treatment step (iv).
- the refractive index nd of the glass obtained by the manufacturing method of the glass of this embodiment is 1.75 or more.
- the lower limit of the refractive index nd is more preferably 1.80, still more preferably 1.85, and particularly preferably 1.90.
- the upper limit of the refractive index nd is not limited as long as glass can be obtained, but can be, for example, about 2.5.
- the optical system can be made compact and highly functional. From such a viewpoint, the higher the refractive index nd, the better.
- the upper limit of the refractive index nd is preferably 2.4, more preferably 2.3.
- the content of the glass component, the total content, and the content of the additive are represented by mol% in terms of oxide.
- the glass obtained by the manufacturing method of the present embodiment is at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 as a glass component (hereinafter, “high refractive index Component) is contained.
- the total content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 contained in the glass is 20% or more, more preferably 25% or more, still more preferably 30% or more More preferably, it is 35% or more.
- TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 exceeds 85%, the devitrification resistance tends to deteriorate, so from the viewpoint of maintaining the devitrification resistance, TiO 2 , Nb
- the total content of 2 O 5 , WO 3 and Bi 2 O 3 is preferably 85% or less, more preferably 80% or less, and still more preferably 75% or less.
- the obtained glass is preferably a P 2 O 5 -containing glass.
- the H + transfer rate during heat treatment is fast, and coloring can be reduced by heat treatment for a short time as compared with other composition systems.
- the glass obtained by the manufacturing method of the present embodiment is preferably a phosphate glass containing a high refractive index component. That is, as a glass component, at least one oxide selected from TiO 2, Nb 2 O 5, WO 3 and Bi 2 O 3, and more preferably an oxide glass containing a P 2 O 5.
- the glass having a P 2 O 5 content greater than the SiO 2 content and a B 2 O 3 content greater than the content of P 2 O 5 in terms of mol% Mention may be made of glasses which are higher than the total content of SiO 2 and B 2 O 3 .
- This embodiment can be applied to a glass composition containing a known composition in which the content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 is in the above range in addition to the compositions exemplified in the examples. .
- the preferable glass composition in this embodiment is demonstrated.
- P 2 O 5 is a glass network forming component and serves to maintain the thermal stability of the glass.
- the content of P 2 O 5 is less than 7%, the thermal stability tends to decrease, so the content of P 2 O 5 is preferably 7% or more.
- the content of P 2 O 5 is preferably in the range of 7 to 40%.
- the lower limit of the content of P 2 O 5 is more preferably 10%, still more preferably 12%, still more preferably 15%, still more preferably 18%.
- the upper limit of the content of P 2 O 5 is more preferably 35%, still more preferably 33%, still more preferably 30%, still more preferably 28%.
- the content of SiO 2 is preferably smaller than the content (M) of P 2 O 5 . More preferably, the range of the content of SiO 2 is 0% to 0.8 ⁇ M [%] in terms of the relationship between the content of SiO 2 and the above-mentioned M (content [%] of P 2 O 5 ). Further preferable range is 0% to 0.5 ⁇ M [%], more preferable range is 0% to 0.3 ⁇ M [%], and still more preferable range is 0% to 0.15 ⁇ M [%] is there.
- B 2 O 3 works to improve the devitrification resistance by containing a small amount.
- the preferable range of the content of B 2 O 3 is 0% or more and less than M [%] More preferable range is 0% to 0.9 ⁇ M [%], more preferable range is 0% to 0.7 ⁇ M [%], and more preferable range is 0% to 0.6 ⁇ M [%], An even more preferable range is 0% to 0.5 ⁇ M [%], a still more preferable range is 0% to 0.4 ⁇ M [%], and a still more preferable range is 0% to 0.35 ⁇ M [%] It is.
- TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 are components that function to increase the refractive index and also to increase the dispersion, and to improve the chemical durability.
- the devitrification resistance tends to deteriorate as the contents of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 increase.
- the upper limit of the content of TiO 2 is preferably 40%, more preferably 35%, still more preferably 33%, and still more preferably 30%. From the top to obtain the effect of introducing TiO 2, preferable lower limit of the content of TiO 2 1%, more preferred lower limit is 3%.
- the content of TiO 2 can also be made 0%.
- the upper limit of the content of Nb 2 O 5 is preferably 45%, more preferably 40%, and still more preferably 35%. From the top to obtain the effect of introducing Nb 2 O 5, preferable lower limit is 5% of the content of Nb 2 O 5, more preferred lower limit is 8%, more preferred lower limit is 11%.
- the content of Nb 2 O 5 can also be made 0%.
- the preferred range of the content of WO 3 is 0 to 30%. From the viewpoint of obtaining the introduction effect of WO 3 described above, the lower limit of the content of WO 3 is preferably 1%, more preferably 3%, and still more preferably 5%. On the other hand, from the viewpoint of maintaining the devitrification resistance, the upper limit of the content of WO 3 is preferably 27%, more preferably 24%, still more preferably 20%, and still more preferably 18%. The content of WO 3 can also be made 0%.
- the preferred range of the content of Bi 2 O 3 is 0 to 35%. From the viewpoint of obtaining the effect of introducing the Bi 2 O 3, Bi 2 O 3 preferred lower limit is 1% of the content of, and more preferable lower limit is 3%, more preferred lower limit is 5%. On the other hand, from the viewpoint of maintaining the devitrification resistance, the upper limit of the content of Bi 2 O 3 is preferably 30%, more preferably 28%, and still more preferably 24%. The content of Bi 2 O 3 can also be 0%.
- the divalent metal components such as BaO, SrO, CaO, MgO and ZnO work to improve the meltability of the glass and to reduce the coloration of the glass. In addition, if it is an appropriate amount, it works to improve the devitrification resistance.
- the content of BaO, SrO, CaO, MgO and ZnO is preferably 0 to 40% in total, since the refractive index tends to be lowered and the devitrification resistance is deteriorated due to the inclusion of an excessive amount, 0 It is more preferable that it is ⁇ 32%.
- the upper limit of the total content of BaO, SrO, CaO, MgO and ZnO is preferably 30%, more preferably 27%, still more preferably 25%.
- the lower limit of the total content of BaO, SrO, CaO, MgO and ZnO is preferably 0.1%, more preferably 0.5%, still more preferably 1%.
- BaO is an effective component for maintaining a high refractive index, so the content of BaO is preferably in the range of 0 to 40%, preferably in the range of 0 to 32%. It is more preferable to do.
- the upper limit of the content of BaO is preferably 30%, more preferably 27%, and still more preferably 25%.
- the lower limit of the content of BaO is preferably 0.1%, more preferably 0.5%, and still more preferably 1%.
- the content of BaO can also be made 0%.
- Alkali metal oxides such as Li 2 O, Na 2 O and K 2 O work to improve the meltability of the glass and to reduce the coloration of the glass. It also lowers the glass transition temperature and the softening temperature and lowers the heat treatment temperature of the glass. However, the inclusion of an excessive amount tends to lower the refractive index and deteriorate the devitrification resistance, so the total content of Li 2 O, Na 2 O and K 2 O is preferably 0 to 40%. 0 to 35% is more preferable, 0 to 32% is more preferable, and 0 to 30% is more preferable. The content of each of Li 2 O, Na 2 O and K 2 O can also be made 0%.
- the content is more than 0% and less than 10%, more preferably 0%, from the viewpoint of obtaining a high refractive index glass. More preferably, it is more than 9%, and more preferably more than 0% and 8% or less.
- the preferable range of the content of Al 2 O 3 is 0 to 12%, more preferable range is 0 to 7%, and further preferable range is 0 to 3%.
- ZrO 2 works to increase the refractive index, and a small amount works to improve the devitrification resistance.
- the preferable range of the content of ZrO 2 is 0 to 16%, more preferably 0 to 12%, and still more preferably 0. A range of -7%, more preferably 0-3%.
- GeO 2 works to increase the refractive index while maintaining resistance to devitrification. Also, GeO 2 works to increase the refractive index, but unlike TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 , it does not increase the coloration of the glass. However, since it is a very expensive component as compared with other components, the content of GeO 2 should be as low as possible in order to reduce the manufacturing cost of the glass. Therefore, in order to widely spread high refractive index glass products, it is desirable to provide a high refractive index glass with excellent transmittance while reducing the content of GeO 2 . According to this embodiment, by setting the total content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 to 20% or more, excellent transmittance can be obtained without using a large amount of GeO 2. High refractive index glass can be provided.
- the preferable range of the content of GeO 2 is 0 to 10%, more preferably 0 to 5%, still more preferably 0 to 3%, still more preferably 0 to 2%, and still more preferable
- the range is 0 to 1%, the still more preferable range is 0 to 0.5%, and GeO 2 may not be contained.
- a manufacturing cost is not considered, it can use suitably in an effective amount.
- TeO 2 works to increase the refractive index while maintaining resistance to devitrification.
- the content of TeO 2 is preferably in the range of 0 to 10%, more preferably in the range of 0 to 5%, still more preferably in the range of 0 to 3%, and still more preferably in the range of 0 to 2.
- a still more preferable range is 0 to 1%, and a still more preferable range is 0 to 0.5%, and TeO 2 may not be contained.
- Sb 2 O 3 has an oxidizing action and functions to suppress the reduction of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . Therefore, the addition of antimony oxide has conventionally been used to suppress the reduction of the high refractive index component during melting.
- Sb 2 O 3 itself absorbs light in the visible region and oxidizes the melting vessel made of a noble metal by its oxidation action to promote the dissolution of noble metal ions into the molten glass, so the coloration of the glass finally obtained Cause.
- the glass manufacturing method of the present embodiment it is preferable to prepare the glass raw material so that the content of antimony oxide becomes less than 1000 ppm in terms of Sb 2 O 3 in the obtained glass. According to the method for producing glass of the present embodiment, it is possible to reduce the coloration of the glass obtained by heat treating the glass in an oxidizing atmosphere without using the oxidation action of antimony oxide.
- the preferable range of the content of Sb 2 O 3 is 0 ppm or more and less than 1000 ppm.
- the upper limit of the content of Sb 2 O 3 is more preferably as small as possible in the order of 900 ppm, 800 ppm, 700 ppm, 600 ppm, 500 ppm, 400 ppm, 300 ppm, 200 ppm, 100 ppm. It is not necessary to contain Sb 2 O 3 .
- the total content of SrO, BaO, ZnO, Li 2 O, Na 2 O, K 2 O, Al 2 O 3 , ZrO 2 , GeO 2 , TeO 2 and Sb 2 O 3 is 90% or more. , 92% or more, more preferably 95% or more, still more preferably 96% or more, still more preferably 97% or more, still more preferably 98% or more More preferably, it is more preferably 99% or more.
- the total content may be 100%.
- Ta 2 O 5 , Y 2 O 3 , La 2 O 3 , Gd 2 O 3 , Yb 2 O 3 , In 2 O 3 , Ga 2 O 3 , SnO 2 , CeO 2 , F, etc. are also contained if they are in small amounts be able to.
- the total content of Ta 2 O 5 , Y 2 O 3 , La 2 O 3 , Gd 2 O 3 , Yb 2 O 3 , In 2 O 3 , Ga 2 O 3 and F is preferably 0 to 10%. , 0 to 7% is more preferable, 0 to 5% is more preferable, 0 to 3% is more preferable, 0 to 1% is still more preferable, and 0 to 0. Even more preferably, it is 5%.
- F is not a component to be contained in a large amount.
- a preferable range of the content of F is 0 to 3%, a more preferable range is 0 to 1%, a further preferable range is 0 to 0.5%, and it is more preferable that substantially no F is contained.
- a substance or additive that has absorption in the visible range such as Cu, Cr, Mn, Fe, Co, Ni, V, Mo, Nd, Eu, Er, Tb, Ho, Pr, etc. It is preferable not to contain it.
- the inclusion of unavoidable impurities is not excluded. Therefore, although it is most preferable that the above-mentioned components be completely used in the glass raw material, it is sufficient that the used amount is substantially zero, that is, even when trace amounts are inevitably mixed in the glass raw material as impurities. preferable.
- a glass raw material according to a glass component, an oxide, phosphoric acid, phosphate (polyphosphate, metaphosphate, pyrophosphate etc.), boric acid, boric anhydride, boric acid anhydride, carbonate, nitrate, sulfuric acid
- Known glass materials such as salts and hydroxides can be used.
- the application of the glass obtained by the production method of the present embodiment is not particularly limited, for example, it can be suitably used as an optical glass.
- a known method may be applied. For example, molten glass is molded to produce a glass material for press molding. Next, this glass material is reheated and press-molded to produce an optical element blank. Furthermore, it processes by the process including grinding
- molten glass is molded to produce a glass material for press molding, and this glass material is heated and precision press molded to produce an optical element.
- a molten glass may be molded to produce a glass molded body, and the glass molded body may be processed to produce a glass material for press molding.
- a molten glass is molded to produce a glass molded body, and the molded body is processed to produce an optical element.
- the optical functional surface of the produced optical element may be coated with an antireflective film, a total reflection film or the like according to the purpose of use.
- an optical element various lenses, such as a spherical lens, an aspheric lens, a macro lens, and a lens array, a prism, a diffraction grating, etc. can be illustrated.
- the manufacturing method of the following optical glass is one of the preferable another form, Comprising:
- the manufacturing method of the glass of this invention is not limited to the manufacturing method of the following optical glass.
- the material for an optical glass produced by using the method of producing a glass of the first embodiment and / or the method of producing a glass of the second embodiment is oxidized
- An optical glass having a refractive index nd of 1.9 or more is produced through at least a heat treatment step (iv) of heat treatment in a thermal atmosphere.
- the “material for optical glass” is a glass produced through a forming step of forming the molten glass in the melting container into a predetermined shape, and the glass in a deeply colored state before being subjected to heat treatment Means
- “optical glass” means the glass which heat-processed the raw material for optical glass of the state colored strongly. That is, “optical glass” is glass whose coloring is reduced by heat treatment as compared with “material for optical glass”.
- materials for optical glass” and “optical glass” “glass materials for press molding” manufactured using “materials for optical glass” or “optical glass”, “optical elements” and “other glass”
- the articles are all amorphous glass and not crystallized glass.
- the oxidizing atmosphere gas may be any gas containing oxygen, and the oxygen concentration may be, for example, about the same as or higher than that of air.
- an oxidizing atmosphere gas for example, air, a gas obtained by adding oxygen to air, a gas substantially consisting only of oxygen, and the like can be mentioned.
- the heat treatment temperature and the heat treatment time may be set appropriately so as to obtain desired optical characteristics.
- the optical glass obtained through a heat treatment process has a high less transparent coloring, the water contained in the optical glass, OH - in the infrared absorption attributable, to quantitatively grasp a spectrophotometer Is possible.
- the water content in the optical glass is the water content in the material for optical glass and the material for optical glass It is apparent that the water content is proportional to or strongly correlated with the water content in the molten glass at the same time.
- suitable manufacturing conditions for producing the material for optical glass that is, the method and amount of supplying water into the melting container, and as the glass raw material It becomes extremely easy to grasp the kind of material used and the compounding amount (in particular, the compounding amount of the phosphoric acid component). Therefore, by measuring and grasping the optical properties (especially the transmittance) and the water content of the optical glass, it is possible to easily determine suitable manufacturing conditions for manufacturing the material for optical glass based on the results.
- the water content in optical glass can be grasped
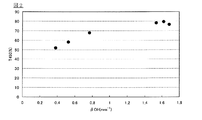
- FIG. 2 shows No. 1 shown in Table 1.
- No. 1 used for measurement of the graph shown in FIG. The optical glass having the composition of No.
- the raw material for optical glass which consists of a composition of 1 is heat-processed at 600 degreeC in air
- FIG. 1 The external transmittance at a wavelength of 450 nm when light is incident on the optical glass having a thickness of 5 mm in parallel to the thickness direction with respect to the ⁇ OH value when the ⁇ OH value of the optical glass having the composition of 3 is changed It is the graph which showed the change of (T450).
- the optical glass having the composition of No. 2 has no.
- the raw material for optical glass which consists of 2 compositions is heat-processed at 570 degreeC in air
- the ⁇ OH value, the refractive index nd and the Abbe number ⁇ ⁇ d shown in FIG. 2, FIG. 3 and Table 1 are values measured in the state of the optical glass after heat treatment.
- the glass composition of No. 4 has a total content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 of 30 mol% or more, and further contains P 2 O 5 .
- the ⁇ OH values at a plurality of points shown in FIG. 2 are values set by adjusting the amount of water vapor per unit time when water vapor is introduced near the liquid surface of the molten glass in the production of the material for optical glass. is there. At this time, all production conditions other than the amount of water vapor were the same. That is, the ⁇ OH value was increased or decreased by the increase or decrease of the amount of water vapor.
- This point also applies to the ⁇ OH values of a plurality of points shown in FIG. As apparent from FIGS. 2 and 3, it can be seen that the external transmittance (T450) also increases as the ⁇ OH value increases.
- the coloring degree of the optical glass can be quantified by ⁇ 80 which is an index indicating the coloring degree.
- ⁇ 80 is the thickness of the optical glass based on the measured internal transmittance after measuring the internal transmittance in the wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with the thickness direction Means the wavelength (nm) at which the internal transmittance (internal transmittance ⁇ ) calculated assuming that L is 10 mm is 80%. In other words, it means that the internal transmittance at a thickness of 10 mm is 80% or more in the wavelength range of ⁇ 80 or more in the wavelength range of 280 to 700 nm.
- T1 is a surface reflection loss measured in a wavelength range of 280 nm to 1550 nm when light is incident in parallel with the thickness direction for the first sample having a thickness of d1 (mm)
- a transmittance (%) including T2 is a wavelength of 280 nm when light is incident in parallel to the thickness direction of a second sample having a thickness of d2 (mm) made of the same glass as the first sample It represents transmission (%) including surface reflection loss measured in the range of ⁇ 1550 nm.
- ⁇ 80 is calculated using the result of transmittance measurement at wavelengths of 280 to 700 nm, the transmittances T1 and T2 may be measured in the range of wavelengths of 280 to 700 nm.
- ⁇ d represents a difference d2-d1 (mm) between the thickness d1 and the thickness d2, and the thickness d1 and the thickness d2 satisfy the relationship of d1 ⁇ d2.
- ⁇ 80 increases as the total content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 increases.
- the total content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 in mol% is X
- the relationship between X and ⁇ 80 is as shown in the following equation (3). Therefore, it is difficult to significantly improve ⁇ 80.
- a represents a constant (1.8359 nm / mol%)
- b represents a constant (351.06 nm).
- the heat treatment step is performed using a material having a high water content for optical glass, and therefore, it is easy to satisfy the following formula (4).
- the heat treatment temperature and the heat treatment time in the heat treatment step can be appropriately set so as to satisfy the following equation (4). ⁇ 80 ⁇ aX + b (4)
- a and b are the same as those shown in Formula (3).
- an optical glass satisfying the following Formula (5) or the following Formula (6) can be produced by appropriately selecting the heat treatment temperature and the heat treatment time in the heat treatment step.
- a is the same as that shown in the formula (3), and c represents a constant (348.06 nm).
- d represents a constant (345.06 nm).
- the heat treatment process for the purpose of color reduction of performing the heat treatment in an oxidative atmosphere (for the purpose of reducing the coloration of the material for optical glass) using the manufactured material for optical glass
- the manufactured material for optical glass It is also possible to produce a glass material for press molding or an optical element containing an optical glass having a ratio nd of 1.9 or more.
- post-steps carried out as necessary other than the heat treatment step for the purpose of color reduction for example, forming steps by pouring into a press or a mold, cutting steps, polishing steps, heat treatment steps (but heat treatment for the purpose of color reduction)
- the various known processes (aside from the process) can be appropriately carried out before and after the heat treatment process for the purpose of color reduction.
- the glass material for press molding is a glass material for obtaining a press-formed product, specifically, an optical element blank or an optical element.
- a method of producing a glass material for press molding for example, (1) a flowing molten glass flow is separated to form a molten glass mass, and the glass material for press molding finally obtained in the process of cooling the molten glass mass
- a method of producing a glass material for press molding by performing a heat treatment step for reducing color on the material for optical glass after molding the material for optical glass of the same shape and size, (2) a molten glass as a mold And casting the glass block (material for optical glass) into a glass block, and producing a glass material for press molding in a post-processing step including a heat treatment step for the purpose of reducing the color of the glass block.
- Examples of the optical element include various lenses such as a spherical lens and an aspheric lens, and a prism.
- Examples of the method of producing an optical element include the methods shown in the following (1) to (3).
- the present invention is not limited to the embodiment at all, and it is needless to say that the present invention can be practiced in various modes without departing from the scope of the present invention. .
- an optical glass is exemplified, but any glass product in which coloring by a reducing component causes a problem can be suitably used for manufacturing various glass products regardless of the optical element.
- a glass product an optical window material, glass for solar cells, a cover glass etc. are mentioned, for example.
- the glass which concerns on this embodiment is suitable as a material for optical elements, it is preferable that it is amorphous (amorphous) glass.
- a method of producing an optical element made of glass for example, there is a method of heating and softening a glass material to form it.
- the crystallized glass in which the crystal phase is dispersed in the glassy material is unsuitable for the above forming method.
- the crystal phase in the crystallized glass may scatter light to reduce the performance as an optical element.
- Amorphous glass does not have such a problem.
- this embodiment exemplifies the method of melting a raw material mainly using a crucible as an example of the manufacturing method of optical glass, as a melting container, the tube made of quartz etc. which both ends opened is used. It is also good.
- a tube made of quartz or the like is fixed in an inclined state in a glass melting furnace.
- an opening is provided at a position corresponding to the lower end of the lower end of the tube.
- Raw material batch material or cullet
- the melt flows slowly in the tube and flows out one after another from the lower open side of the tube.
- the effluent passes through the opening at the bottom of the furnace and is dropped one after another into water in a water tank previously disposed under the opening at the bottom of the glass melting furnace to form cullet.
- the raw material is melted using a tube made of quartz or the like, but instead of the tube, a crucible made of quartz or the like may be used.
- a crucible made of quartz or the like may be used.
- raw materials are put in a crucible made of quartz etc., heated and melted to form a melt, and then the melt is cast in water or poured out on a cooled heat-resistant plate to produce cullet. Good.
- Example 1 [Preparation of batch material] First, when producing optical glass with desired characteristics, phosphoric acid, barium metametaphosphate, titanium oxide, niobium oxide, tungsten oxide, bismuth oxide, boric acid, barium carbonate, barium carbonate, sodium carbonate, potassium carbonate as raw materials of glass And silicon oxide were prepared respectively. Next, the above raw materials are appropriately selected and weighed so that the glass composition of the finally obtained optical glass becomes the oxide compositions I to VIII shown in Table 2, and sufficiently mixed to produce batch raw materials I to VIII. did.
- the cullet taken out of the water is dried, a part of the cullet is sampled for measuring the refractive index, put in a platinum crucible and melted, and the obtained glass melt is clarified and homogenized, and then cast in a mold After molding and holding at a temperature near the glass transition temperature, cooling was performed at a temperature decrease rate of 30 ° C./hour.
- the refractive index nd of the sample for refractive index measurement thus obtained was measured by the refractive index measurement method defined by the Japan Optical Glass Industrial Standard.
- a cullet was prepared so as to obtain a desired refractive index to obtain a prepared cullet for producing an optical glass.
- the temperature of the crucible was raised to a clarifying temperature (range of 900 to 1450 ° C.) to clarify (refining step). Subsequently, the temperature of the crucible was lowered to the homogenization temperature, and the mixture was homogenized by stirring with a stirrer (homogenization step).
- a clarifying temperature range of 900 to 1450 ° C.
- the volume in the melting furnace (the volume of the space in the furnace made of a refractory that stores the crucible), and the residence time of the melting material in the melting furnace (after the cullet is introduced into the platinum melting container, The time until the molten glass flows out is shown in Table 3.
- a platinum pipe is inserted from outside the melting furnace into a platinum crucible disposed in the furnace, and water vapor (H 2 O 100% by volume) is introduced into the space in the platinum crucible through the platinum pipe. And supplied.
- water vapor H 2 O 100% by volume
- the flow rate of the supplied water vapor is shown in Table 3.
- steam shown in Table 3 is the value converted into the flow volume in normal temperature and normal pressure, and a unit is a liter / minute.
- the molten glass thus homogenized flows out of the platinum glass outflow pipe attached to the bottom of the crucible in an air atmosphere (effluence process) and is poured into a mold disposed below the outflow pipe, whereby a long length is obtained.
- the glass block (width 150 mm ⁇ thickness 10 mm) was molded (molding step).
- the optical glass sample was processed to prepare a cylindrical measurement sample (diameter 5 mm, height 20 mm).
- the glass transition temperature Tg of the obtained measurement sample was measured using a thermomechanical analyzer (TMA) at a temperature rising rate of + 10 ° C./min.
- ln is a natural logarithm
- the thickness t corresponds to the distance between the two planes.
- the external transmittance also includes the reflection loss on the surface of the glass sample, and is the ratio of the intensity of the transmitted light to the intensity of the incident light incident on the glass sample (transmitted light intensity / incident light intensity). Also, the higher the value of ⁇ OH, the more water is contained in the glass. The results are shown in Table 7.
- T450 (H) The optical glass sample was heat-treated by raising the temperature at a rate of + 100 ° C./hour, holding it at a predetermined holding temperature for 100 hours, and decreasing the temperature at a rate of ⁇ 30 ° C./hour in an air atmosphere.
- holding temperature changes according to a composition, it was set as the temperature shown in Table 5 according to the oxide composition of each optical glass sample.
- the heat-treated optical glass sample was processed to prepare a plate-like glass sample having a thickness of 10 mm, which was optically polished parallel to each other and flat on both sides.
- the external transmittance T450 (H) at 450 nm of the plate-like glass sample thus obtained was determined using a spectrophotometer. The larger the value of T450 (H), the better the transmittance, meaning that the coloration of the glass is reduced. The results are shown in Table 7.
- the optical glass sample was heat-treated under the same conditions as in the case of T450 (H).
- the heat-treated optical glass sample was processed to prepare a plate-like glass sample having a thickness of 10 mm ⁇ 0.1 mm, which was optically polished parallel to each other and flat on both sides.
- T450 (L) 0.5 to 0.7 cc of molten glass which has been subjected to the homogenization process when producing an optical glass sample, and a mold for floating molding (concave portion receiving the molten glass is formed of a porous body, a porous body It was poured into the recess of the mold having a structure in which the gas spouted from the surface of the recess, the gas was spouted from the recess, an upward wind pressure was applied to the molten glass block on the recess, and the glass block was formed in a floating state.
- the temperature of the above glass gob is raised at a rate of + 100 ° C./hour, held at a predetermined holding temperature and holding time, and lowered at a rate of -30 ° C./hour to obtain a spherical optical glass sample after heat treatment.
- holding temperature and holding time change according to a composition, it was set as the temperature and time which are shown in Table 6 according to the oxide composition of each optical glass sample.
- the obtained spherical optical glass sample was processed to prepare a plate-like glass sample having a thickness of 5 mm, which was optically polished on both sides parallel to each other and flat.
- the external transmittance T450 (L) at 450 nm of the plate-like glass sample thus obtained was determined using a spectrophotometer. The larger the value of T450 (L), the better the transmittance, and the shorter the heat treatment, the less the coloration of the glass.
- Bubble breakage 40 cc of molten glass (glass melt) before starting the clarifying process in preparing an optical glass sample is collected and clarified in another platinum crucible in the atmosphere for a fixed time, and the glass melt is It was cooled in a platinum crucible and solidified. In this process, the color was reduced to such an extent that the number of bubbles contained in the glass could be counted. The solidified glass was then removed from the platinum crucible.
- the inside of the glass was subjected to magnified observation (100 ⁇ ) using an optical microscope (magnification: 20 to 100 ⁇ ), and the number of bubbles contained in the glass was counted.
- the same observation was carried out for each of the measurement samples having different clarifying times, and the clarifying time of the measurement sample, in which the number of bubbles remaining in the glass was 100 / kg or less, was evaluated as the defoaming time. The shorter the defoaming time, the better the clarity.
- Table 7 The results are shown in Table 7.
- an optical glass sample having a high ⁇ OH can be obtained (Sample 13 to Sample 16, Sample 24 to Sample 26, Sample 33 to Sample 35, Sample 43 to sample 46, sample 53 to sample 56, sample 63 to sample 66, sample 72, sample 73, and sample 82 to sample 84). That is, it is expected that the optical glass sample produced by the production method of the present invention has a high water content in the glass.
- the values of T450 (H) and T450 (L), which are values indicating the transmittance of the optical glass sample after heat treatment, are It is large and the value of ⁇ 70 (or ⁇ 80) is small. From these results, it was confirmed that the optical glass sample produced by the production method of the present invention has excellent transmittance after heat treatment.
- the transmittance is dramatically improved by a short time heat treatment, and the thickness of the measurement sample is In view of the above, it was confirmed that the transmittance was equivalent to the transmittance (the value of T450 (H)) after the heat treatment for 100 hours.
- the optical glass sample produced by the production method of the present invention also has a significantly reduced Pt content. This result is considered to be that the optical glass sample produced by the production method of the present invention is prevented from deterioration of the transmittance due to Pt ions, and the optical glass sample produced by the production method of the present invention is excellent in transmittance It agrees with having.
- the optical glass sample produced by the production method corresponding to the comparative example of the present invention is produced with the same oxide composition by the production method of the present invention because the operation to increase the water content in the molten glass is not performed. It was confirmed that the value of ⁇ OH is smaller than that of the optical glass sample (Sample 11, Sample 12, Sample 21 to Sample 23, Sample 31, Sample 32, Sample 41, Sample 42, Sample 51, Sample 52, Sample 61, sample 62, sample 71, and sample 81). That is, it is expected that the optical glass sample produced by the production method corresponding to the comparative example of the present invention has a lower water content in the glass than the optical glass sample produced by the production method of the present invention.
- Such an optical glass sample produced by the production method corresponding to the comparative example of the present invention has a transmission of glass after heat treatment compared to an optical glass sample produced with the same oxide composition according to the production method of the present invention It was confirmed that the rate was low, the amount of dissolved Pt was large, and the time required for defoaming was also increased.
- an optical glass having excellent transmittance after heat treatment can be obtained. Further, according to the manufacturing method of the present invention, since a good optical glass can be obtained even if the time required for the clarification step and the heat treatment step is shortened, the production cost can be significantly reduced and the productivity is also improved.
- Example 2 Optical glass samples were produced under the same conditions as Samples 51 to 56 of Example 1 except that antimony oxide (Sb 2 O 3 ) was added to batch raw material V as a glass raw material (Samples 51a to 56a).
- the amount of antimony oxide added is shown in Table 8. In addition, a unit is ppm with respect to 100 mass% of batch raw materials.
- Example 3 The molten glass (molten glass just before starting the outflow step) manufactured by the same method as the sample 55 of Example 1 is allowed to flow out (outflow step) in the atmosphere, and cast into a mold to be formed (forming step) ), And annealing (annealing step) at a temperature decrease rate of ⁇ 100 ° C./hour to produce an optical glass sample (sample 55 b).
- each treatment atmosphere at the time of performing the outflow step and the forming step is changed to the air atmosphere to form an oxidizing atmosphere (an atmosphere in which oxygen is added to air to increase the oxygen partial pressure).
- Another optical glass sample (sample 55c) was produced in the same manner.
- the oxidizing atmosphere was adjusted as follows.
- the molten glass was covered with a cover at a zone where the molten glass flowed out and formed, oxygen gas was supplied into the cover, and the oxygen partial pressure in the atmosphere, ie, the atmosphere where the molten glass flowed out and formed, was higher than the oxygen partial pressure in the atmosphere.
- the volume ratio of oxygen in the atmosphere for flowing out and forming the molten glass was approximately 30% to 40%.
- the refractive index nd, the Abbe number dd, and the glass transition temperature Tg were substantially the same as the values shown in the oxide composition V of Example 1. Further, ⁇ OH was 1.31 mm ⁇ 1 .
- the sample 55c produced by setting the oxygen partial pressure of the atmosphere at the time of flowing out and forming the molten glass higher than the oxygen partial pressure in the atmosphere flows out and forms the molten glass in the atmosphere. It was confirmed that the transmittance in the visible range is high and the color is low compared to the sample 55b produced.
- Example 4 The optical glass samples (glass blocks) produced in Examples 1 to 3 were divided, and further processed if necessary, to obtain glass materials for press molding corresponding to the respective optical glasses.
- the glass material for press molding obtained in this manner was heated, softened and pressed in the atmosphere to produce an optical element blank having a lens shape.
- the obtained optical element blank was annealed in the atmosphere, and was further processed by grinding, polishing and the like to produce glass optical elements such as lenses and prisms corresponding to the samples of Examples 1 to 3. .
- the temperature-fall rate at the time of annealing was set so that the refractive index of an optical element might turn into a desired value.
- Optical glass sample (Sample 13 to Sample 16, Sample 24 to Sample 26, Sample 33 to Sample 35, Sample 43 to Sample 46, Sample 53 to Sample 56, Sample 63 to Sample 66, Samples prepared by the manufacturing method of the present invention 72, Sample 73, Sample 82 to Sample 84, Sample 53a to Sample 56a, Sample 55b and Sample 55c), the optical element produced in the process from forming of the molten glass to processing of the optical element blank, etc. It was confirmed that the coloring was significantly reduced by the heat treatment in the oxidizing atmosphere of
- optical glass sample glass (Sample 11, Sample 12, Sample 21 to Sample 23, Sample 31, Sample 32, Sample 41, Sample 42, Sample 51, Sample 52) manufactured by the manufacturing method corresponding to the comparative example of the present invention.
- the optical element manufactured using the sample 61, the sample 62, the sample 71, the sample 81, the sample 51b, and the sample 52b) has an oxidizing atmosphere such as the atmosphere between molding of the molten glass and processing of the optical element blank. Even after the heat treatment in, it was confirmed that the coloring remained and the reduction effect of the coloring was low.
- Examples A1 to A6 and Examples B1 to B6) The raw materials of the batch are roughly melted to prepare cullet, and the cullet is put into a platinum crucible, heated, melted and shaped. 1 to No.
- Each optical glass of the composition of 4 was produced in the following procedures. First, phosphate, orthophosphoric acid, oxides, carbonates, nitrates and sulfates were weighed and thoroughly mixed to prepare a raw material (batch raw material). Next, this batch raw material was put into a container made of quartz. 1 and No. The glass No. 2 was heated in the range of the liquidus temperature LT to 1400 ° C. 3 and No. The glass No. 4 was heated at a liquidus temperature LT to 1300 ° C. to form a molten glass, and this molten glass was dropped into water to prepare a cullet raw material.
- the cullet raw material was reconstituted, and was put into a platinum crucible (fusion container) and a platinum lid was placed.
- the cullet raw material in the platinum crucible was heated within the range of the liquidus temperature LT to 1300 ° C. of the glass composition of the cullet raw material to melt the cullet raw material and melt vitrify (melting step).
- the temperature of the liquidus temperature LT to 1400 ° C. to clarify
- the temperature of the liquidus temperature LT to 1300 ° C. lowers and stirs and homogenizes (homogenization) Step)
- the clarified and homogenized molten glass is drained from the glass outflow pipe and cast in a mold to form a glass block (material for optical glass).
- the water vapor flow rate per unit time supplied into the platinum crucible is shown in Table 9 and Table 10.
- the water vapor flow rate shown in Table 9 and Table 10 is a value converted to the flow rate at normal temperature, and the unit is liter / minute.
- the platinum crucible is sealed with a platinum lid without an opening, and the platinum crucible is sealed from the melting process through the clarification process to the homogenization process. It controlled the transpiration of water from cullet raw material and molten glass in the process of melting.
- each glass block (material for optical glass) consisting of 2 glasses is raised to 600 ° C from 25 ° C over 2 hours in the atmosphere, and annealing (heat treatment) is performed at 600 ° C to form a glass block (material for optical glass) An operation to reduce the color was performed. Thereafter, the glass block was cooled to normal temperature at a temperature lowering rate of -30 ° C / hour.
- maintained at 600 degreeC is 1 hour.
- the temperature of each glass block (material for optical glass) consisting of 4 glasses is raised to 570 ° C. from 25 ° C.
- the measured values of the refractive index nd and the Abbe number dd shown in Table 1 are values measured using a sample cooled at a cooling rate of 30 ° C. per hour, and the measured values of the liquidus temperature LT After reheating and holding for 2 hours, it is cooled to room temperature, and the presence or absence of crystal precipitation inside the glass is confirmed by an optical microscope, and the lowest temperature at which no crystal is observed is taken as the liquidus temperature.
- Examples A1 to A3 in Table 9 are data on optical glass prepared from a platinum pipe without introducing water vapor into the melting vessel, and Examples A4 to A6 are samples from platinum pipes to the melting vessel. It is data about the optical glass produced by introducing water vapor.
- the use of a phosphoric acid raw material and the improvement of the airtightness of the melting container introduce water into the molten glass and suppresses the dissipation of water vapor from the melting container.
- the water vapor partial pressure in the melting vessel is also positively increased.
- Example A4 Comparing the T450 and ⁇ 80 of the optical glass of Example A1 to Example A3 with the T450 and ⁇ 80 of the optical glass of Example A4 to Example A6, Example A4 in which the water vapor partial pressure in the melting vessel is positively increased
- the optical glass of Example A6 also has a larger ⁇ OH value. Then, it is understood from the results of visual observation of the glass block before and after heat treatment described later that the degree of coloring is significantly reduced.
- Examples B4 to B6 are from platinum pipes to a melting vessel It is data about the optical glass produced by introducing water vapor.
- Example B1 to Example B3 by using a phosphoric acid raw material and by enhancing the airtightness of the melting vessel, water is introduced into the molten glass and the dissipation of water vapor from the melting vessel is suppressed.
- Example B4 to Example B6 the water vapor partial pressure in the melting vessel is also positively increased.
- Example B4 Comparing the T450 and ⁇ 80 of the optical glass of Example B1 to Example B3 with the T450 and ⁇ 80 of the optical glass of Example B4 to Example B6, Example B4 in which the water vapor partial pressure in the melting vessel is positively increased
- the optical glass of Example B6 also has a larger ⁇ OH value. Then, it is understood from the results of visual observation of the glass block before and after heat treatment described later that the degree of coloring is significantly reduced.
- Examples A1 to A6 and Examples B1 to B6 platinum crucibles were used as the melting vessel, but materials for optical glass are manufactured using platinum alloy crucibles, gold crucibles, and gold alloy crucibles. Even when the obtained material for optical glass was heat-treated in an oxidizing atmosphere, the coloration thereof could be significantly reduced, and as a result, an optical glass with less coloration could be obtained. Furthermore, in Examples A4 to A6 and Examples B4 to B6, water vapor was supplied through a pipe into a covered platinum crucible, but similar effects may be obtained by bubbling water vapor into molten glass in the platinum crucible. You can get the effect. The composition of optical glass to be produced is shown in Table 1 2 or No. Even when the glass composition is changed to No. 4, no. 1 and No. Similar results are obtained with the glass composition of 3.
- Examples A4 to A6 and B4 to B6 steam obtained by boiling water using a boiler was used as steam supplied into the platinum crucible.
- water vapor obtained by other methods can also be appropriately used.
- water is sprayed in the form of a mist into a refractory glass melting furnace that accommodates a melting vessel such as a platinum crucible and steamed to increase the partial pressure of water vapor in the atmosphere inside the glass melting furnace and inside the melting vessel Good.
- water may be supplied into the glass melting furnace using a pump, and the water in the melting furnace may be boiled to vaporize the water, and the partial pressure of water vapor in the glass melting atmosphere may be increased, or other methods. May be used.
- the moisture content in the optical glass material can be increased also by using these methods.
- Example A1 A glass block (raw material for optical glass) was produced in the same manner as in Examples A1 to A3 except that the melting vessel was not opened but in an open state, and then heat treatment was performed in the same manner as in Examples A1 to A6. However, the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples A1 to A6.
- No. 1 described in Table 1 is a glass composition.
- No. 1 instead of the composition of No. 1 A glass block (material for optical glass) was produced and heat-treated in the same manner as in Comparative Example A1 except that the composition of No. 2 was used.
- the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples A1 to A6.
- Comparative Example B1 A glass block (material for optical glass) was produced in the same manner as in Examples B1 to B3 except that the melting vessel was not opened but in an open state, and then heat treatment was performed in the same manner as in Examples B1 to B6. However, the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples B1 to B6.
- No. 1 described in Table 1 is a glass composition.
- No. 3 instead of No. 3 A glass block (material for optical glass) was produced and heat-treated in the same manner as in Comparative Example B1 except that the composition No. 4 was used. However, the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples B1 to B6.
- Comparative Example A2 A glass block (material for optical glass) was produced in the same manner as in Examples A4 to A6 except that nitrogen gas was introduced instead of water vapor into the melting vessel, and then heat treatment was performed in the same manner as in Examples A1 to A6. . However, the degree of coloring of the heat-treated glass block (optical glass) was much larger than that of the glass block (optical glass) of Comparative Example A1.
- No. 1 described in Table 1 is a glass composition.
- No. 1 instead of the composition of No. 1 A glass block (material for optical glass) was produced in the same manner as in Comparative Example 2 except that the composition of No. 2 was used, and was heat-treated. However, the results were similar to Comparative Example A2.
- Comparative Example B2 A glass block (material for optical glass) was produced in the same manner as in Examples B4 to B6 except that nitrogen gas was introduced instead of water vapor into the melting vessel, and then heat treatment was performed in the same manner as in Examples B1 to B6. . However, the coloring degree of the heat treated glass block (optical glass) was much larger than the glass block (optical glass) of Comparative Example B1.
- No. 1 described in Table 1 is a glass composition.
- No. 3 instead of No. 3 A glass block (material for optical glass) was produced and heat-treated in the same manner as in Comparative Example B2, except that the composition No. 4 was used. However, the results were similar to Comparative Example B2.
- Example 3 A glass block (material for optical glass) is produced in the same manner as in Examples A4 to A6 except that a reducing gas such as carbon monoxide gas is introduced into the melting vessel in place of water vapor, and then Examples A1 to A6 and Heat treatment was performed in the same manner. However, the coloring degree of the heat-treated glass block (optical glass) was much larger than that of the glass block (optical glass) of Comparative Example 1.
- the concentration of the reducing gas is increased, the reduced glass component is alloyed with the platinum crucible and breakage of the crucible occurs. This corresponds to No. 1 described in Table 1 for the glass composition. 2 to No. The same applies to the case of changing to the composition of 4.
- a glass block (optical glass) is colored, but the sheet positioned below the glass block (optical glass) has sufficient transparency (medium transparency).
- Example 7 The optical glasses produced in Examples A1 to A6 and Examples B1 to B6 were processed into glass materials for press molding, heated, softened, and press molded to produce optical element blanks. Further, the optical element blank was processed to produce an optical element such as a spherical lens or a prism. Furthermore, the lens surface and the prism surface were coated with an antireflective film to obtain a final product. No. 1 described in Table 1 2 and No. The glass material for press molding, an optical element blank, and an optical element were similarly produced about the optical glass of 4.
- the preferable thing of another form of this embodiment is a heating and melting process of heating and melting a glass material in a melting vessel in order to obtain a molten glass,
- a heating and melting process of heating and melting a glass material in a melting vessel in order to obtain a molten glass
- An oxide glass comprising at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 as a glass component, wherein TiO 2 , Nb 2 O 5 , WO 3 and Bi 2
- the material for optical glass which is 30 mol% or more in total content of O 3 is manufactured, It is a manufacturing method of the material for optical glass characterized by supplying moisture in the fusion container in the above-mentioned heating and melting process.
- the manner of supplying water into the melting vessel is a first supplying manner of supplying water vapor into the atmosphere near the liquid surface of the molten glass, and a method of supplying water vapor while bubbling water vapor into the molten glass It is preferable that it is any selected from the 2nd supply aspect and the 3rd supply aspect which combined the 1st supply aspect and the 2nd supply aspect.
- the glass material preferably contains a phosphoric acid component.
- the preferable thing of another form of this embodiment is a heating and melting process of heating and melting a glass material in a melting vessel in order to obtain a molten glass,
- a forming step of forming the molten glass in the melting container into a predetermined shape An oxide glass comprising at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 as a glass component, wherein TiO 2 , Nb 2 O 5 , WO 3 and Bi 2
- the material for optical glass which is 30 mol% or more in total content of O 3 is manufactured, While the said glass-making feedstock contains water
- the glass material preferably contains a phosphoric acid component.
- the optical glass material further includes P 2 O 5 , and
- the content of P 2 O 5 contained in the material for optical glass is preferably in the range of 10 mol% to 35 mol%.
- the material constituting the melting vessel is preferably at least one metal material selected from noble metals and noble metal alloys.
- a material for optical glass is produced by the method for producing a material for optical glass according to any one of the above, and a heat treatment step of heat treating the material for optical glass in an oxidizing atmosphere
- the optical glass manufacturing method is characterized in that an optical glass having a refractive index nd of at least 1.9 is produced.
- ⁇ 80 of the optical glass satisfies the following expression (4).
- ⁇ 80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
- a material for optical glass is produced by the method for producing a material for optical glass according to any one of the above, and a heat treatment step of heat treating the material for optical glass in an oxidizing atmosphere
- a method of manufacturing a glass material for press molding comprising: manufacturing a glass material for press molding made of optical glass having a refractive index nd of 1.9 or more.
- ⁇ 80 of the optical glass satisfies the following expression (4).
- ⁇ 80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
- a material for optical glass is produced by the method for producing a material for optical glass according to any one of the above, and a heat treatment step of heat treating the material for optical glass in an oxidizing atmosphere Manufacturing an optical element made of an optical glass having a refractive index nd of 1.9 or more.
- ⁇ 80 of the optical glass satisfies the following expression (4).
- ⁇ 80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
Abstract
Description
〔1〕 TiO2、Nb2O5、WO3およびBi2O3の少なくとも一種以上の成分を含むガラス原料を熔融容器内にて加熱、熔融し、熔融ガラスを得る熔融工程(i)において、熔融ガラス中の水分量を高める操作を行うガラスの製造方法。 The gist of the present invention for the purpose of solving such problems is as follows.
[1] A glass raw material containing at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 is heated and melted in a melting vessel in a melting step (i) to obtain a molten glass, The manufacturing method of the glass which performs operation which raises the moisture content in molten glass.
前記熔融ガラスを成形する工程(iii)をさらに有し、
前記工程(ii)および前記工程(iii)の少なくともいずれか一つの工程を酸化性雰囲気下で行う上記〔1〕~〔5〕のいずれかに記載のガラスの製造方法。 [6] a step (ii) of flowing the molten glass out of the melting vessel;
The method further includes the step (iii) of forming the molten glass,
The method for producing glass according to any one of the above [1] to [5], wherein at least one of the step (ii) and the step (iii) is performed in an oxidizing atmosphere.
前記工程(iv)を酸化性雰囲気下で行う上記〔1〕~〔6〕のいずれかに記載のガラスの製造方法。 [7] The method further includes the step (iv) of heat-treating the glass,
The method for producing glass according to any one of the above [1] to [6], wherein the step (iv) is performed in an oxidizing atmosphere.
前記ガラスを、さらに成形または加工する工程と、を有する光学素子の製造方法。 [10] A process for producing glass by the production method according to any one of the above [1] to [9],
And d) further molding or processing the glass.
すなわち、本実施形態のガラスの製造方法は、好ましくは、
調合材料を熔融してカレットを得るラフメルト工程と、前記カレットを再熔融してガラスを得るリメルト工程と、を有し、
前記ラフメルト工程および前記リメルト工程のうち少なくともいずれか一方において、熔融ガラス中の水分量を高める操作を行うことを特徴とする。 The glass production method of the present embodiment is more preferably carried out by the rough melt-remelt method.
That is, the method for producing glass of this embodiment is preferably
It has a rough melting process to melt the compounded material to obtain cullet, and a remelt process to remelt the cullet to obtain glass,
In at least one of the rough melting step and the remelting step, an operation to increase the amount of water in the molten glass is performed.
βOH=-[ln(B/A)]/t ・・・(1) The water content in glass can be grasped | ascertained by the (beta) OH value shown, for example to the following Formula (1).
βOH = − [ln (B / A)] / t (1)
βOH≧0.4891×ln(1/HR)+2.48 ・・・(1-2) In particular, from the viewpoint of improving the transmittance of glass after heat treatment, it is preferable that the glass produced by the manufacturing method of the present embodiment satisfies the following formula (1-2).
βOHOH0.4891 × ln (1 / HR) +2.48 (1-2)
βOH≧181.39×nd-3-325.75×nd-2+194.85×nd-1-38.1 ・・・(1-3) In particular, from the viewpoint of reducing the noble metal content in the glass, it is preferable that the glass produced by the production method of the present embodiment satisfies the following formula (1-3).
βOH ≧ 181.39 × nd -3 -325.75 × nd -2 + 194.85 × nd -1 -38.1 ··· (1-3)
以下、特記しない限り、ガラス成分の含有量、合計含有量、添加剤の含有量は酸化物換算のモル%で表示する。 About glass composition Hereinafter, unless otherwise stated, the content of the glass component, the total content, and the content of the additive are represented by mol% in terms of oxide.
次に、本実施態様における好ましいガラス組成について説明する。 This embodiment can be applied to a glass composition containing a known composition in which the content of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 is in the above range in addition to the compositions exemplified in the examples. .
Next, the preferable glass composition in this embodiment is demonstrated.
本実施形態の製造方法により得られるガラスの用途は、特に限定されるものではないが、例えば光学ガラスとして好適に用いることができる。本実施形態の製造方法により作製した光学ガラスを使用して光学素子を作るには、公知の方法を適用すればよい。例えば、熔融ガラスを成形してプレス成形用ガラス素材を作製する。次に、このガラス素材を再加熱、プレス成形して光学素子ブランクを作製する。さらに光学素子ブランクの研磨を含む工程により加工して光学素子を作製する。
あるいは、熔融ガラスを成形してプレス成形用ガラス素材を作製し、このガラス素材を加熱、精密プレス成形して光学素子を作製する。 Production of Optical Element Although the application of the glass obtained by the production method of the present embodiment is not particularly limited, for example, it can be suitably used as an optical glass. In order to produce an optical element using the optical glass produced by the production method of this embodiment, a known method may be applied. For example, molten glass is molded to produce a glass material for press molding. Next, this glass material is reheated and press-molded to produce an optical element blank. Furthermore, it processes by the process including grinding | polishing of an optical element blank, and an optical element is produced.
Alternatively, molten glass is molded to produce a glass material for press molding, and this glass material is heated and precision press molded to produce an optical element.
あるいは、熔融ガラスを成形してガラス成形体を作製し、この成形体を加工して光学素子を作製する。 In each of the above-described steps, a molten glass may be molded to produce a glass molded body, and the glass molded body may be processed to produce a glass material for press molding.
Alternatively, a molten glass is molded to produce a glass molded body, and the molded body is processed to produce an optical element.
光学素子としては、球面レンズ、非球面レンズ、マクロレンズ、レンズアレイなどの各種レンズ、プリズム、回折格子などを例示することができる。 The optical functional surface of the produced optical element may be coated with an antireflective film, a total reflection film or the like according to the purpose of use.
As an optical element, various lenses, such as a spherical lens, an aspheric lens, a macro lens, and a lens array, a prism, a diffraction grating, etc. can be illustrated.
以下、本実施形態の実施の別形態として、光学ガラスの製造方法を説明する。なお、下記の光学ガラスの製造方法は、好ましい別形態の一つであって、本発明のガラスの製造方法は、下記の光学ガラスの製造方法に限定されるものではない。 Method of Producing Optical Glass Hereinafter, a method of producing optical glass will be described as another embodiment of the present embodiment. In addition, the manufacturing method of the following optical glass is one of the preferable another form, Comprising: The manufacturing method of the glass of this invention is not limited to the manufacturing method of the following optical glass.
logτ=-(logT1-logT2)×10/Δd ・・・(2) The coloring degree of the optical glass can be quantified by λτ80 which is an index indicating the coloring degree. λτ80 is the thickness of the optical glass based on the measured internal transmittance after measuring the internal transmittance in the wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with the thickness direction Means the wavelength (nm) at which the internal transmittance (internal transmittance τ) calculated assuming that L is 10 mm is 80%. In other words, it means that the internal transmittance at a thickness of 10 mm is 80% or more in the wavelength range of λτ80 or more in the wavelength range of 280 to 700 nm. Here, the internal transmittance τ is the transmittance excluding the surface reflection loss on the incident side and the emission side, and the transmittance T1 and T2 including the surface reflection loss of each sample using two samples having different thicknesses. That is, it is a value calculated based on the following equation (2) using the external transmittances T1 and T2 in the wavelength range of 280 nm to 1550 nm, and using those measured values.
log τ = − (log T1−log T2) × 10 / Δd (2)
λτ80>aX+b ・・・(3)
なお、式(3)中、aは、定数(1.8359nm/mol%)を表し、bは、定数(351.06nm)を表す。
λτ80> aX + b (3)
In addition, in Formula (3), a represents a constant (1.8359 nm / mol%), b represents a constant (351.06 nm).
λτ80<aX+b ・・・(4)
なお、式(4)中、aおよびbは式(3)に示したものと同様である。 On the other hand, in the method for producing an optical glass according to the present embodiment, the heat treatment step is performed using a material having a high water content for optical glass, and therefore, it is easy to satisfy the following formula (4). Here, the heat treatment temperature and the heat treatment time in the heat treatment step can be appropriately set so as to satisfy the following equation (4).
λτ80 <aX + b (4)
In Formula (4), a and b are the same as those shown in Formula (3).
λτ80<aX+c ・・・(5)
λτ80<aX+d ・・・(6)
ここで式(5)中、aは式(3)に示したものと同様であり、cは定数(348.06nm)を表す。また、式(6)中、aは式(3)に示したものと同様であり、dは定数(345.06nm)を表す。 In addition, it is more preferable to satisfy the following Formula (5), and it is more preferable to satisfy the following Formula (6). In this case, an optical glass satisfying the following Formula (5) or the following Formula (6) can be produced by appropriately selecting the heat treatment temperature and the heat treatment time in the heat treatment step.
λτ80 <aX + c (5)
λτ80 <aX + d (6)
Here, in the formula (5), a is the same as that shown in the formula (3), and c represents a constant (348.06 nm). Further, in the formula (6), a is the same as that shown in the formula (3), and d represents a constant (345.06 nm).
[バッチ原料の調製]
まず、所望の特性を備えた光学ガラスを作製するにあたり、ガラスの原材料として、リン酸、メタリン酸バリウム、酸化チタン、酸化ニオブ、酸化タングステン、酸化ビスマス、ホウ酸、炭酸バリウム、炭酸ナトリウム、炭酸カリウムおよび酸化ケイ素をそれぞれ準備した。次に、最終的に得られる光学ガラスのガラス組成が、表2に示す酸化物組成I~VIIIとなるように、上記原材料を適宜選択、秤量し、十分混合してバッチ原料I~VIIIを作製した。 Example 1
[Preparation of batch material]
First, when producing optical glass with desired characteristics, phosphoric acid, barium metametaphosphate, titanium oxide, niobium oxide, tungsten oxide, bismuth oxide, boric acid, barium carbonate, barium carbonate, sodium carbonate, potassium carbonate as raw materials of glass And silicon oxide were prepared respectively. Next, the above raw materials are appropriately selected and weighed so that the glass composition of the finally obtained optical glass becomes the oxide compositions I to VIII shown in Table 2, and sufficiently mixed to produce batch raw materials I to VIII. did.
調合されたバッチ原料I~VIIIを、各光学ガラスのガラス原料とした。このガラス原料を石英製坩堝に投入し、大気雰囲気中で900~1350℃で熔解して熔融物を得た。このようにして得られた熔融物を水中に滴下してカレットを得た。 [Preparation of cullet and compound cullet (rough melt process)]
The prepared batch materials I to VIII were used as glass materials of each optical glass. The glass raw material was put into a quartz crucible and melted at 900 to 1350 ° C. in an air atmosphere to obtain a molten material. The melt thus obtained was dropped into water to obtain cullet.
次に、調合カレットを白金製坩堝(熔融容器)に投入し、800~1350℃の範囲内で白金製坩堝内の調合カレットを加熱、熔融し、熔融ガラスとした(熔融工程)。 [Preparation of optical glass (remelt process)]
Next, the prepared cullet was introduced into a platinum crucible (melting vessel), and the prepared cullet in the platinum crucible was heated and melted at 800 ° C. to 1350 ° C. to form a molten glass (melting step).
得られた光学ガラスサンプル(試料11~試料84)の各種物性は、以下のように測定、評価した。 [Evaluation of optical glass]
Various physical properties of the obtained optical glass samples (samples 11 to 84) were measured and evaluated as follows.
光学ガラスサンプルを適量採取し、これを酸およびアルカリ処理し、誘導結合プラズマ質量分析法(ICP-MS法)、イオンクロマトグラフフィー法を用いて、各成分の含有量を定量することで測定し、酸化物組成I~VIIIと一致していることを確認した。 [1] Glass composition An appropriate amount of optical glass sample is collected, treated with acid and alkali, and the content of each component is quantified using inductively coupled plasma mass spectrometry (ICP-MS method) and ion chromatography method. It was confirmed that the oxide compositions I to VIII were in agreement.
光学ガラスサンプルを作製する際の、均質化工程を経た熔融ガラスを、鋳型に鋳込んで成形し、ガラス転移温度付近の温度で保持した後、10℃/時の降温速度で冷却し、測定用試料を作製した。得られた測定用試料について、日本光学硝子工業会規格で定められた屈折率測定法により、屈折率nd、ng、nF、ncを測定した。さらに、これら屈折率の測定値より、アッベ数νdを算出した。 [2] refractive index nd, Abbe number d d and glass transition temperature Tg
The molten glass that has passed through the homogenization process in making the optical glass sample is cast in a mold and molded, held at a temperature near the glass transition temperature, and then cooled at a temperature decrease rate of 10 ° C / hour for measurement A sample was made. The refractive indexes nd, ng, nF, and nc were measured for the obtained measurement samples by the refractive index measurement method defined by the Japan Optical Glass Industrial Standard. Furthermore, the Abbe number νd was calculated from the measured values of the refractive index.
光学ガラスサンプルを加工して、両面が互いに平行かつ平坦に光学研磨された厚さ1mmの板状ガラス試料を準備した。この板状ガラス試料の研磨面に垂直方向から光を入射して、波長2500nmにおける外部透過率Aおよび波長2900nmにおける外部透過率Bを、分光光度計を用いてそれぞれ測定し、下記式(1)により、βOHを算出した。
βOH=-[ln(B/A)]/t ・・・(1) [3] βOH
The optical glass sample was processed to prepare a plate-like glass sample having a thickness of 1 mm, which was optically polished so that both sides were parallel to each other and flat. Light is perpendicularly incident on the polished surface of this plate-like glass sample, and the external transmittance A at a wavelength of 2500 nm and the external transmittance B at a wavelength of 2900 nm are respectively measured using a spectrophotometer, and the following formula (1) ΒOH was calculated by
βOH = − [ln (B / A)] / t (1)
光学ガラスサンプルを、大気雰囲気中で、+100℃/時の速度で昇温し、所定の保持温度で100時間保持して、-30℃/時の速度で降温して、熱処理した。なお、保持温度は、組成に応じて異なるため、それぞれの光学ガラスサンプルの酸化物組成に応じて、表5に示す温度とした。 [4] T450 (H)
The optical glass sample was heat-treated by raising the temperature at a rate of + 100 ° C./hour, holding it at a predetermined holding temperature for 100 hours, and decreasing the temperature at a rate of −30 ° C./hour in an air atmosphere. In addition, since holding temperature changes according to a composition, it was set as the temperature shown in Table 5 according to the oxide composition of each optical glass sample.
光学ガラスサンプルを適量採取し、これをアルカリ融解して、Ptを分離する処理した後、ICP-MS法によりガラス中のPt量を定量した。結果を表7に示す。 [5] Pt Content An appropriate amount of an optical glass sample was collected and alkali-melted to separate Pt, and then the amount of Pt in the glass was quantified by ICP-MS. The results are shown in Table 7.
まず、光学ガラスサンプルを、T450(H)の場合と同様の条件で熱処理した。
熱処理後の光学ガラスサンプルを加工して、両面が互いに平行かつ平坦に光学研磨された厚さ10mm±0.1mmの板状ガラス試料を準備した。この板状ガラス試料の研磨面に垂直方向から光を入射して、波長280nm~700nmの範囲で表面反射損失を含む分光透過率を、分光光度計を用いて測定し、分光透過率(外部透過率)が80%および70%になる波長を、それぞれ着色度λ80およびλ70とした。λ80およびλ70の値は、いずれも小さいほど、ガラスの着色が少ないことを意味する。結果を表7に示す。なお、λ80により評価した試料については、表7に示す結果に下線を付した。 [6] Degree of coloring λ80 and λ70
First, the optical glass sample was heat-treated under the same conditions as in the case of T450 (H).
The heat-treated optical glass sample was processed to prepare a plate-like glass sample having a thickness of 10 mm ± 0.1 mm, which was optically polished parallel to each other and flat on both sides. Light is perpendicularly incident on the polished surface of this plate-like glass sample, and the spectral transmittance including surface reflection loss in the wavelength range of 280 nm to 700 nm is measured using a spectrophotometer, and the spectral transmittance (external transmission The wavelengths at which the ratio) reaches 80% and 70% were taken as the coloring degrees .lambda.80 and .lambda.70, respectively. The smaller the values of λ80 and λ70, the lower the coloration of the glass. The results are shown in Table 7. The results shown in Table 7 are underlined for the samples evaluated by λ80.
光学ガラスサンプルを作製する際の、均質化工程を経た熔融ガラスを、0.5~0.7cc採取し、浮上成形用の鋳型(熔融ガラスを受ける凹部が多孔質体で形成され、多孔質体を通して凹部表面からガスが噴出する構造になっている鋳型)の凹部に流し込み、凹部からガスを噴出し、凹部上の熔融ガラス塊に上向きの風圧を加え、ガラス塊を浮上状態で成形した。 [7] T450 (L)
0.5 to 0.7 cc of molten glass which has been subjected to the homogenization process when producing an optical glass sample, and a mold for floating molding (concave portion receiving the molten glass is formed of a porous body, a porous body It was poured into the recess of the mold having a structure in which the gas spouted from the surface of the recess, the gas was spouted from the recess, an upward wind pressure was applied to the molten glass block on the recess, and the glass block was formed in a floating state.
光学ガラスサンプルを作製する際の、清澄工程を開始する前の熔融ガラス(ガラス融液)を40cc採取し、大気中で別の白金坩堝で一定時間清澄し、ガラス融液を白金坩堝中で冷却し、固化させた。この過程で、ガラス中に含まれる泡の数をカウントできる程度に着色を低減した。次に固化したガラスを白金坩堝から取り出した。 [8]
ガラス原料として、バッチ原料Vに酸化アンチモン(Sb2O3)を添加した以外は、実施例1の試料51~試料56と同様の条件で光学ガラスサンプルを作製した(試料51a~試料56a)。酸化アンチモンの添加量を表8に示す。なお、単位は、バッチ原料100質量%に対するppmである。 (Example 2)
Optical glass samples were produced under the same conditions as Samples 51 to 56 of Example 1 except that antimony oxide (Sb 2 O 3 ) was added to batch raw material V as a glass raw material (Samples 51a to 56a). The amount of antimony oxide added is shown in Table 8. In addition, a unit is ppm with respect to 100 mass% of batch raw materials.
得られた光学ガラスサンプル(試料51a~試料56a)の各種物性は、実施例1の場合と同様の条件により測定、評価した。 [Evaluation of optical glass]
Various physical properties of the obtained optical glass samples (Samples 51a to 56a) were measured and evaluated under the same conditions as in Example 1.
実施例1の試料55と同様の方法で作製された熔融ガラス(流出工程を開始する直前の熔融ガラス)を、大気雰囲気中で、流出し(流出工程)、鋳型に流し込んで成形し(成形工程)、-100℃/時の降温速度でアニール(アニール工程)し、光学ガラスサンプル(試料55b)を作製した。 (Example 3)
The molten glass (molten glass just before starting the outflow step) manufactured by the same method as the sample 55 of Example 1 is allowed to flow out (outflow step) in the atmosphere, and cast into a mold to be formed (forming step) ), And annealing (annealing step) at a temperature decrease rate of −100 ° C./hour to produce an optical glass sample (sample 55 b).
[9]分光透過率
光学ガラスサンプルを加工して、両面が互いに平行かつ平坦に光学研磨された同じ厚みの板状ガラス試料を準備した。この板状ガラス試料の研磨面(板厚方向)に垂直方向から光を入射して、波長280nm~700nmの範囲で表面反射損失を含む分光透過率を、分光光度計を用いて測定した。結果を図4に示す。図4の横軸は波長、縦軸は外部透過率である。 Furthermore, the following measurements were performed on sample 55b and sample 55c.
[9] Spectral Transmittance The optical glass sample was processed to prepare a plate-like glass sample of the same thickness optically polished on both sides parallel to each other and flat. Light was perpendicularly incident on the polished surface (plate thickness direction) of this plate-like glass sample, and the spectral transmittance including surface reflection loss was measured using a spectrophotometer in the wavelength range of 280 nm to 700 nm. The results are shown in FIG. The horizontal axis in FIG. 4 is the wavelength, and the vertical axis is the external transmittance.
実施例1~3で作製した光学ガラスサンプル(ガラスブロック)を分割し、必要に応じて、さらに加工を施し、各光学ガラスに対応するプレス成形用ガラス素材を得た。 (Example 4)
The optical glass samples (glass blocks) produced in Examples 1 to 3 were divided, and further processed if necessary, to obtain glass materials for press molding corresponding to the respective optical glasses.
(実施例A1~A6および実施例B1~B6)
バッチ原料を粗熔解してカレットを作製し、カレットを白金製坩堝に入れて加熱、熔融、成形して、表1に示すNo.1~No.4の組成の各光学ガラスを以下の手順で作製した。まず最初に、リン酸塩、正リン酸、酸化物、炭酸塩、硝酸塩、硫酸塩を秤量し、十分混合して調合した原料(バッチ原料)とした。次に、このバッチ原料を石英製容器に入れて、No.1およびNo.2のガラスについては液相温度LT~1400℃の範囲で加熱し、No.3およびNo.4のガラスについては液相温度LT~1300℃の範囲で加熱することで、熔融ガラスとし、この熔融ガラスを水中に滴下してカレット原料を作製した。 Next, an example according to another embodiment of the present invention will be described.
(Examples A1 to A6 and Examples B1 to B6)
The raw materials of the batch are roughly melted to prepare cullet, and the cullet is put into a platinum crucible, heated, melted and shaped. 1 to No. Each optical glass of the composition of 4 was produced in the following procedures. First, phosphate, orthophosphoric acid, oxides, carbonates, nitrates and sulfates were weighed and thoroughly mixed to prepare a raw material (batch raw material). Next, this batch raw material was put into a container made of quartz. 1 and No. The glass No. 2 was heated in the range of the liquidus temperature LT to 1400 ° C. 3 and No. The glass No. 4 was heated at a liquidus temperature LT to 1300 ° C. to form a molten glass, and this molten glass was dropped into water to prepare a cullet raw material.
熔融容器に蓋をせず開放状態とした以外は実施例A1~A3と同様にしてガラスブロック(光学ガラス用素材)を作製した後、実施例A1~A6と同様にして熱処理を行った。しかしながら、熱処理されたガラスブロック(光学ガラス)の着色度合は、実施例A1~A6よりも大きかった。 (Comparative Example A1)
A glass block (raw material for optical glass) was produced in the same manner as in Examples A1 to A3 except that the melting vessel was not opened but in an open state, and then heat treatment was performed in the same manner as in Examples A1 to A6. However, the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples A1 to A6.
熔融容器に蓋をせず開放状態とした以外は実施例B1~B3と同様にしてガラスブロック(光学ガラス用素材)を作製した後、実施例B1~B6と同様にして熱処理を行った。しかしながら、熱処理されたガラスブロック(光学ガラス)の着色度合は、実施例B1~B6よりも大きかった。 (Comparative Example B1)
A glass block (material for optical glass) was produced in the same manner as in Examples B1 to B3 except that the melting vessel was not opened but in an open state, and then heat treatment was performed in the same manner as in Examples B1 to B6. However, the degree of coloring of the heat-treated glass block (optical glass) was greater than in Examples B1 to B6.
熔融容器内に水蒸気の代わりに窒素ガスを導入した以外は実施例A4~A6と同様にしてガラスブロック(光学ガラス用素材)を作製した後、実施例A1~A6と同様にして熱処理を行った。しかしながら、熱処理したガラスブロック(光学ガラス)の着色度合は、比較例A1のガラスブロック(光学ガラス)よりも非常に大きくなった。 (Comparative Example A2)
A glass block (material for optical glass) was produced in the same manner as in Examples A4 to A6 except that nitrogen gas was introduced instead of water vapor into the melting vessel, and then heat treatment was performed in the same manner as in Examples A1 to A6. . However, the degree of coloring of the heat-treated glass block (optical glass) was much larger than that of the glass block (optical glass) of Comparative Example A1.
熔融容器内に水蒸気の代わりに窒素ガスを導入した以外は実施例B4~B6と同様にしてガラスブロック(光学ガラス用素材)を作製した後、実施例B1~B6と同様にして熱処理を行った。しかしながら、熱処理したガラスブロック(光学ガラス)の着色度合は、比較例B1のガラスブロック(光学ガラス)よりも非常に大きくなった。 (Comparative Example B2)
A glass block (material for optical glass) was produced in the same manner as in Examples B4 to B6 except that nitrogen gas was introduced instead of water vapor into the melting vessel, and then heat treatment was performed in the same manner as in Examples B1 to B6. . However, the coloring degree of the heat treated glass block (optical glass) was much larger than the glass block (optical glass) of Comparative Example B1.
熔融容器内に水蒸気の代わりに一酸化炭素ガスなどの還元性ガスを導入した以外は実施例A4~A6と同様にしてガラスブロック(光学ガラス用素材)を作製した後、実施例A1~A6と同様にして熱処理を行った。しかしながら、熱処理したガラスブロック(光学ガラス)の着色度合は、比較例1のガラスブロック(光学ガラス)よりも非常に大きくなった。 (Comparative example 3)
A glass block (material for optical glass) is produced in the same manner as in Examples A4 to A6 except that a reducing gas such as carbon monoxide gas is introduced into the melting vessel in place of water vapor, and then Examples A1 to A6 and Heat treatment was performed in the same manner. However, the coloring degree of the heat-treated glass block (optical glass) was much larger than that of the glass block (optical glass) of Comparative Example 1.
表11に、各実施例および比較例で作製したガラスブロックの熱処理前後の着色度合の観察結果を示す。なお、着色度合は、白色の用紙上に、平面形状が略円形状のガラスブロックを配置して、室内光下にて目視観察することにより評価した。なお、観察に用いたいずれの実施例および比較例のガラスブロックも厚みはほぼ同じである。また、表11中に示す透明度の評価基準は以下の通りである。A:ガラスブロック(光学ガラス)が薄く着色しているものの、ガラスブロック(光学ガラス)の下方に位置する用紙の白さも十分に認識できる程に透明度が高い(高透明度)。B:ガラスブロック(光学ガラス)が着色しているが、ガラスブロック(光学ガラス)の下方に位置する用紙は十分に認識できる程度の透明度はある(中透明度)。C:ガラスブロック(光学ガラス)が濃く着色しており、ガラスブロック(光学ガラス)の下方に位置する用紙が僅かに認識できる程度の低い透明度しかない(低透明度)。D:ガラスブロック(光学ガラス)は完全に不透明であり、ガラスブロック(光学ガラス)の下方に位置する用紙の存在は全く認識できない(不透明)。 (Details of the observation result of the degree of coloring of the glass block before and after heat treatment)
In Table 11, the observation result of the coloring degree before and behind heat processing of the glass block produced by each Example and the comparative example is shown. The degree of coloring was evaluated by disposing a glass block having a substantially circular planar shape on a white paper and visually observing it under room light. In addition, the thickness of the glass block of any Example used for observation and a comparative example is substantially the same. Moreover, the evaluation criteria of the transparency shown in Table 11 are as follows. A: Although the glass block (optical glass) is lightly colored, the transparency is high (high transparency) so that the whiteness of the sheet located below the glass block (optical glass) can be sufficiently recognized. B: The glass block (optical glass) is colored, but the sheet positioned below the glass block (optical glass) has sufficient transparency (medium transparency). C: The glass block (optical glass) is strongly colored and there is only low transparency (low transparency) to which the sheet located below the glass block (optical glass) is slightly recognizable. D: The glass block (optical glass) is completely opaque, and the presence of a sheet located below the glass block (optical glass) can not be recognized at all (opaque).
実施例A1~A6、B1~B6および比較例A1、A2、B1、B2、3で用いた熱処理後のガラスブロックのうち、透明度の評価がDのものを除いてガラスブロックの内部を光学顕微鏡により観察した。その結果、いずれのガラスブロックにおいてもその内部に、混入した白金異物および析出した結晶は確認されなかった。また、実施例A1~A6、B1~B6および比較例A1、A2、B1、B2、3で用いたガラスブロック中の白金溶解量をICP発光分光法により測定したところ、いずれも2ppm未満であった。
(実施例7)
実施例A1~A6および実施例B1~B6で作製した光学ガラスをプレス成形用ガラス素材に加工し、加熱、軟化してプレス成形し、光学素子ブランクを作製した。さらに光学素子ブランクを加工して球面レンズ、プリズムなどの光学素子を作製した。さらにレンズ表面、プリズム表面に反射防止膜をコートして最終製品を得た。表1に記載のNo.2およびNo.4の光学ガラスについても同様にしてプレス成形用ガラス素材、光学素子ブランク、光学素子を作製した。 (Confirmation of platinum contamination etc.)
Of the glass blocks after heat treatment used in Examples A1 to A6 and B1 to B6 and Comparative Examples A1, A2, B1 and B2, 3, the evaluation of the transparency is the inside of the glass block by an optical microscope except for those of D. I observed it. As a result, no platinum foreign matter and precipitated crystals were found in the interior of any glass block. In addition, the amount of dissolved platinum in the glass blocks used in Examples A1 to A6 and B1 to B6 and Comparative Examples A1, A2, B1 and B2 and 3 was measured by ICP emission spectroscopy, and all were less than 2 ppm. .
(Example 7)
The optical glasses produced in Examples A1 to A6 and Examples B1 to B6 were processed into glass materials for press molding, heated, softened, and press molded to produce optical element blanks. Further, the optical element blank was processed to produce an optical element such as a spherical lens or a prism. Furthermore, the lens surface and the prism surface were coated with an antireflective film to obtain a final product. No. 1 described in Table 1 2 and No. The glass material for press molding, an optical element blank, and an optical element were similarly produced about the optical glass of 4.
本実施の別形態の好ましいものは、熔融ガラスを得るために、熔融容器内にてガラス原料を加熱・熔融する加熱・熔融工程と、
前記熔融容器内の前記熔融ガラスを所定の形状に成形する成形工程とを、少なくとも経ることにより、
ガラス成分として、TiO2、Nb2O5、WO3およびBi2O3から選択される少なくとも1種の酸化物を含む酸化物ガラスであり、TiO2、Nb2O5、WO3およびBi2O3の合計含有量が30mol%以上である光学ガラス用素材を製造し、
前記加熱・熔融工程において、前記熔融容器内に水分を供給することを特徴とする光学ガラス用素材の製造方法である。 The following is a summary.
The preferable thing of another form of this embodiment is a heating and melting process of heating and melting a glass material in a melting vessel in order to obtain a molten glass,
By at least a forming step of forming the molten glass in the melting container into a predetermined shape,
An oxide glass comprising at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 as a glass component, wherein TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 The material for optical glass which is 30 mol% or more in total content of O 3 is manufactured,
It is a manufacturing method of the material for optical glass characterized by supplying moisture in the fusion container in the above-mentioned heating and melting process.
前記熔融容器内の前記熔融ガラスを所定の形状に成形する成形工程とを、少なくとも経ることにより、
ガラス成分として、TiO2、Nb2O5、WO3およびBi2O3から選択される少なくとも1種の酸化物を含む酸化物ガラスであり、TiO2、Nb2O5、WO3およびBi2O3の合計含有量が30mol%以上である光学ガラス用素材を製造し、
前記ガラス原料が水分を含むとともに、
前記加熱・熔融工程において、前記熔融容器が略密閉されることを特徴とする光学ガラス用素材の製造方法である。 The preferable thing of another form of this embodiment is a heating and melting process of heating and melting a glass material in a melting vessel in order to obtain a molten glass,
By at least a forming step of forming the molten glass in the melting container into a predetermined shape,
An oxide glass comprising at least one oxide selected from TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 as a glass component, wherein TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 The material for optical glass which is 30 mol% or more in total content of O 3 is manufactured,
While the said glass-making feedstock contains water | moisture content,
In the heating and melting step, the melting vessel is substantially sealed.
前記光学ガラス用素材に含まれるP2O5の含有量が、10mol%~35mol%の範囲内であることが好ましい。 In the present embodiment, the optical glass material further includes P 2 O 5 , and
The content of P 2 O 5 contained in the material for optical glass is preferably in the range of 10 mol% to 35 mol%.
λτ80<aX+b ・・・(4)
〔式(4)中、λτ80は、前記光学ガラスに対してその厚み方向と平行に光を入射した際の波長280~700nmの範囲における内部透過率を測定した後、当該測定された内部透過率に基づいて前記光学ガラスの厚みが10mmであると仮定して計算した内部透過率が、80%となる波長(nm)を表し、aは、定数(1.8359nm/mol%)を表し、bは、定数(351.06nm)を表し、Xは、TiO2、Nb2O5、WO3およびBi2O3の合計含有量(mol%)を表す。〕 In the present embodiment, it is preferable to perform the heat treatment step such that λτ 80 of the optical glass satisfies the following expression (4).
λτ80 <aX + b (4)
[In the formula (4), λτ80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
λτ80<aX+b ・・・(4)
〔式(4)中、λτ80は、前記光学ガラスに対してその厚み方向と平行に光を入射した際の波長280~700nmの範囲における内部透過率を測定した後、当該測定された内部透過率に基づいて前記光学ガラスの厚みが10mmであると仮定して計算した内部透過率が、80%となる波長(nm)を表し、aは、定数(1.8359nm/mol%)を表し、bは、定数(351.06nm)を表し、Xは、TiO2、Nb2O5、WO3およびBi2O3の合計含有量(mol%)を表す。〕 In the present embodiment, it is preferable to perform the heat treatment step such that λτ 80 of the optical glass satisfies the following expression (4).
λτ80 <aX + b (4)
[In the formula (4), λτ80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
λτ80<aX+b ・・・(4)
〔式(4)中、λτ80は、前記光学ガラスに対してその厚み方向と平行に光を入射した際の波長280~700nmの範囲における内部透過率を測定した後、当該測定された内部透過率に基づいて前記光学ガラスの厚みが10mmであると仮定して計算した内部透過率が、80%となる波長(nm)を表し、aは、定数(1.8359nm/mol%)を表し、bは、定数(351.06nm)を表し、Xは、TiO2、Nb2O5、WO3およびBi2O3の合計含有量(mol%)を表す。〕 In the present embodiment, it is preferable to perform the heat treatment step such that λτ 80 of the optical glass satisfies the following expression (4).
λτ80 <aX + b (4)
[In the formula (4), λτ80 is the internal transmittance measured in a wavelength range of 280 to 700 nm when light is incident on the optical glass in parallel with its thickness direction, and then the measured internal transmittance Represents the wavelength (nm) at which the internal transmittance calculated based on the assumption that the thickness of the optical glass is 10 mm is 80%, a represents a constant (1.8359 nm / mol%), b Represents a constant (351.06 nm), and X represents the total content (mol%) of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 . ]
Claims (10)
- TiO2、Nb2O5、WO3およびBi2O3の少なくとも一種以上の成分を含むガラス原料を熔融容器内にて加熱、熔融し、熔融ガラスを得る熔融工程(i)において、熔融ガラス中の水分量を高める操作を行うガラスの製造方法。 Glass raw materials containing at least one or more components of TiO 2 , Nb 2 O 5 , WO 3 and Bi 2 O 3 are heated and melted in a melting container to obtain molten glass, and in the melting step (i) A method of producing glass, which performs an operation to increase the amount of water.
- 前記ガラス原料が、TiO2、Nb2O5、WO3およびBi2O3を合計で20モル%以上含む請求項1に記載のガラスの製造方法。 The glass raw material, TiO 2, Nb 2 O 5 , a method of manufacturing glass according to claim 1 comprising WO 3 and Bi 2 O 3 in total 20 mol% or more.
- 前記熔融ガラス中の水分量を高める操作が、熔融雰囲気に水蒸気を付加する処理および熔融物内に水蒸気を含むガスをバブリングする処理の少なくともいずれか一方により行われる請求項1または2に記載のガラスの製造方法。 The glass according to claim 1 or 2, wherein the operation of increasing the water content in the molten glass is performed by at least one of a treatment of adding water vapor to the melting atmosphere and a treatment of bubbling a gas containing water vapor in the melt. Manufacturing method.
- 前記熔融容器が金属材料からなる請求項1~3のいずれかに記載のガラスの製造方法。 The method for producing glass according to any one of claims 1 to 3, wherein the melting vessel is made of a metal material.
- 前記熔融工程(i)における熔融容器内の酸素分圧が、大気中の酸素分圧よりも低い請求項1~4のいずれかに記載のガラスの製造方法。 The method according to any one of claims 1 to 4, wherein the partial pressure of oxygen in the melting vessel in the melting step (i) is lower than the partial pressure of oxygen in the atmosphere.
- 前記熔融ガラスを前記熔融容器外に流出する工程(ii)、
前記熔融ガラスを成形する工程(iii)をさらに有し、
前記工程(ii)および前記工程(iii)の少なくともいずれか一つの工程を酸化性雰囲気下で行う請求項1~5のいずれかに記載のガラスの製造方法。 Draining the molten glass out of the melting vessel (ii)
The method further includes the step (iii) of forming the molten glass,
The method for producing glass according to any one of claims 1 to 5, wherein at least one of the step (ii) and the step (iii) is performed in an oxidizing atmosphere. - 前記ガラスを熱処理する工程(iv)をさらに有し、
前記工程(iv)を酸化性雰囲気下で行う請求項1~6のいずれかに記載のガラスの製造方法。 Further comprising the step (iv) of heat treating the glass;
The method for producing glass according to any one of claims 1 to 6, wherein the step (iv) is performed in an oxidizing atmosphere. - 前記酸化性雰囲気が、大気雰囲気、または大気よりも酸素分圧が高い雰囲気である請求項6または7に記載のガラスの製造方法。 The method according to claim 6 or 7, wherein the oxidizing atmosphere is an air atmosphere or an atmosphere having an oxygen partial pressure higher than the air.
- 前記ガラスが、リン酸ガラスである請求項1~8のいずれかに記載のガラスの製造方法。 The method for producing a glass according to any one of claims 1 to 8, wherein the glass is a phosphate glass.
- 請求項1~9のいずれかに記載の製造方法によりガラスを製造する工程と、
前記ガラスを、さらに成形または加工する工程と、を有する光学素子の製造方法。 A process of producing a glass by the production method according to any one of claims 1 to 9,
And d) further molding or processing the glass.
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201380033009.6A CN104395247B (en) | 2012-06-22 | 2013-06-21 | The manufacture method of glass and optical element |
| US14/410,519 US9604872B2 (en) | 2012-06-22 | 2013-06-21 | Glass and optical element production method |
| KR1020147035452A KR102186021B1 (en) | 2012-06-22 | 2013-06-21 | Glass and optical element production method |
Applications Claiming Priority (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012-141452 | 2012-06-22 | ||
| JP2012-141453 | 2012-06-22 | ||
| JP2012-141454 | 2012-06-22 | ||
| JP2012141452 | 2012-06-22 | ||
| JP2012141454 | 2012-06-22 | ||
| JP2012141453 | 2012-06-22 | ||
| JP2012-240954 | 2012-10-31 | ||
| JP2012240953A JP2014024748A (en) | 2012-06-22 | 2012-10-31 | Optical glass, glass raw material for press molding, and optical element |
| JP2012-240953 | 2012-10-31 | ||
| JP2012240954A JP2014024749A (en) | 2012-06-22 | 2012-10-31 | Optical glass, glass raw material for press molding, and optical element |
| JP2012240955 | 2012-10-31 | ||
| JP2012-240955 | 2012-10-31 | ||
| JP2013094501 | 2013-04-26 | ||
| JP2013-094501 | 2013-04-26 | ||
| JP2013-094498 | 2013-04-26 | ||
| JP2013094498 | 2013-04-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013191270A1 true WO2013191270A1 (en) | 2013-12-27 |
Family
ID=49768862
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/067051 WO2013191271A1 (en) | 2012-06-22 | 2013-06-21 | Glass, optical glass, glass raw material for press molding, and optical element |
| PCT/JP2013/067050 WO2013191270A1 (en) | 2012-06-22 | 2013-06-21 | Glass and optical element production method |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2013/067051 WO2013191271A1 (en) | 2012-06-22 | 2013-06-21 | Glass, optical glass, glass raw material for press molding, and optical element |
Country Status (1)
| Country | Link |
|---|---|
| WO (2) | WO2013191271A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9834465B2 (en) | 2013-09-30 | 2017-12-05 | Hoya Corporation | Optical glass and method for producing the same |
| JP2020117411A (en) * | 2019-01-18 | 2020-08-06 | Hoya株式会社 | Method for promoting improvement of glass transmittance, method for manufacturing glass, and glass |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6734630B2 (en) * | 2015-05-12 | 2020-08-05 | 株式会社オハラ | Optical glass |
| JP6755712B2 (en) * | 2016-05-20 | 2020-09-16 | Hoya株式会社 | Manufacturing method of optical products |
| CN113135653B (en) * | 2017-07-20 | 2022-11-01 | Hoya株式会社 | Optical glass and optical element |
| JP6517411B2 (en) * | 2017-07-20 | 2019-05-22 | Hoya株式会社 | Optical glass and optical element |
| JP2023030503A (en) * | 2021-08-23 | 2023-03-08 | 日本電気硝子株式会社 | Method for manufacturing glass material and glass material |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05178638A (en) * | 1991-06-26 | 1993-07-20 | Hoya Corp | Faraday rotation glass |
| JP2005132713A (en) * | 2003-10-10 | 2005-05-26 | Nippon Electric Glass Co Ltd | Method for producing alkali-free glass and alkali-free glass |
| JP2010057893A (en) * | 2008-08-06 | 2010-03-18 | Nippon Electric Glass Co Ltd | Sealing glass |
| JP2011042556A (en) * | 2009-07-24 | 2011-03-03 | Nippon Electric Glass Co Ltd | Method for manufacturing optical glass |
| JP2011046550A (en) * | 2009-08-26 | 2011-03-10 | Nippon Electric Glass Co Ltd | Method for producing sealing glass and sealing glass |
-
2013
- 2013-06-21 WO PCT/JP2013/067051 patent/WO2013191271A1/en active Application Filing
- 2013-06-21 WO PCT/JP2013/067050 patent/WO2013191270A1/en active Application Filing
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05178638A (en) * | 1991-06-26 | 1993-07-20 | Hoya Corp | Faraday rotation glass |
| JP2005132713A (en) * | 2003-10-10 | 2005-05-26 | Nippon Electric Glass Co Ltd | Method for producing alkali-free glass and alkali-free glass |
| JP2010057893A (en) * | 2008-08-06 | 2010-03-18 | Nippon Electric Glass Co Ltd | Sealing glass |
| JP2011042556A (en) * | 2009-07-24 | 2011-03-03 | Nippon Electric Glass Co Ltd | Method for manufacturing optical glass |
| JP2011046550A (en) * | 2009-08-26 | 2011-03-10 | Nippon Electric Glass Co Ltd | Method for producing sealing glass and sealing glass |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9834465B2 (en) | 2013-09-30 | 2017-12-05 | Hoya Corporation | Optical glass and method for producing the same |
| JP2020117411A (en) * | 2019-01-18 | 2020-08-06 | Hoya株式会社 | Method for promoting improvement of glass transmittance, method for manufacturing glass, and glass |
| JP7433764B2 (en) | 2019-01-18 | 2024-02-20 | Hoya株式会社 | Method for promoting improvement of transmittance of glass, method for manufacturing glass, and glass |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2013191271A1 (en) | 2013-12-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6270350B2 (en) | Glass and optical element manufacturing method | |
| JP6009709B1 (en) | Optical glass and manufacturing method thereof | |
| JP7320100B2 (en) | Optical glasses and optical elements | |
| WO2013191270A1 (en) | Glass and optical element production method | |
| JP5826428B1 (en) | Glass, optical glass, glass material for press molding and optical element | |
| JP5826429B1 (en) | Glass, optical glass, glass material for press molding and optical element | |
| JP6283512B2 (en) | Method for producing glass and method for producing optical element |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13806170 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 20147035452 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14410519 Country of ref document: US |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13806170 Country of ref document: EP Kind code of ref document: A1 |