WO2012125973A2 - Potent and selective inhibitors of nav1.3 and nav1.7 - Google Patents
Potent and selective inhibitors of nav1.3 and nav1.7 Download PDFInfo
- Publication number
- WO2012125973A2 WO2012125973A2 PCT/US2012/029537 US2012029537W WO2012125973A2 WO 2012125973 A2 WO2012125973 A2 WO 2012125973A2 US 2012029537 W US2012029537 W US 2012029537W WO 2012125973 A2 WO2012125973 A2 WO 2012125973A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- gptx
- amide
- composition
- matter
- Prior art date
Links
- 230000003389 potentiating effect Effects 0.000 title description 15
- 229940124639 Selective inhibitor Drugs 0.000 title description 3
- 108090000765 processed proteins & peptides Proteins 0.000 claims abstract description 330
- 102000004196 processed proteins & peptides Human genes 0.000 claims abstract description 119
- 239000000203 mixture Substances 0.000 claims abstract description 107
- 208000002193 Pain Diseases 0.000 claims abstract description 58
- 229920001184 polypeptide Polymers 0.000 claims abstract description 54
- 230000036407 pain Effects 0.000 claims abstract description 48
- 150000007523 nucleic acids Chemical class 0.000 claims abstract description 33
- 238000000034 method Methods 0.000 claims abstract description 31
- 102000039446 nucleic acids Human genes 0.000 claims abstract description 30
- 108020004707 nucleic acids Proteins 0.000 claims abstract description 30
- 239000013604 expression vector Substances 0.000 claims abstract description 11
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 4
- 125000000539 amino acid group Chemical group 0.000 claims description 116
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 89
- 235000001014 amino acid Nutrition 0.000 claims description 63
- JDHILDINMRGULE-LURJTMIESA-N N(pros)-methyl-L-histidine Chemical compound CN1C=NC=C1C[C@H](N)C(O)=O JDHILDINMRGULE-LURJTMIESA-N 0.000 claims description 59
- 125000003295 alanine group Chemical group N[C@@H](C)C(=O)* 0.000 claims description 58
- 239000000863 peptide conjugate Substances 0.000 claims description 51
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 claims description 50
- 150000001413 amino acids Chemical group 0.000 claims description 50
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 claims description 49
- 229940024606 amino acid Drugs 0.000 claims description 49
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 claims description 48
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 claims description 47
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 claims description 47
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 claims description 46
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 claims description 45
- 230000007935 neutral effect Effects 0.000 claims description 44
- SNDPXSYFESPGGJ-UHFFFAOYSA-N 2-aminopentanoic acid Chemical compound CCCC(N)C(O)=O SNDPXSYFESPGGJ-UHFFFAOYSA-N 0.000 claims description 43
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 claims description 43
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims description 43
- -1 2-Abu Chemical compound 0.000 claims description 42
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims description 42
- 230000002209 hydrophobic effect Effects 0.000 claims description 42
- NMDDZEVVQDPECF-LURJTMIESA-N (2s)-2,7-diaminoheptanoic acid Chemical compound NCCCCC[C@H](N)C(O)=O NMDDZEVVQDPECF-LURJTMIESA-N 0.000 claims description 41
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 claims description 41
- QUOGESRFPZDMMT-YFKPBYRVSA-N L-homoarginine Chemical compound OC(=O)[C@@H](N)CCCCNC(N)=N QUOGESRFPZDMMT-YFKPBYRVSA-N 0.000 claims description 41
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims description 41
- BRMWTNUJHUMWMS-UHFFFAOYSA-N 3-Methylhistidine Natural products CN1C=NC(CC(N)C(O)=O)=C1 BRMWTNUJHUMWMS-UHFFFAOYSA-N 0.000 claims description 40
- QUOGESRFPZDMMT-UHFFFAOYSA-N L-Homoarginine Natural products OC(=O)C(N)CCCCNC(N)=N QUOGESRFPZDMMT-UHFFFAOYSA-N 0.000 claims description 40
- 125000003130 L-selenocysteinyl group Chemical group O=C([*])[C@@](N([H])[H])([H])C([H])([H])[Se][H] 0.000 claims description 40
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 claims description 40
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 claims description 40
- 229960003104 ornithine Drugs 0.000 claims description 40
- JULROCUWKLNBSN-UHFFFAOYSA-N seleno-DL-cystine Natural products OC(=O)C(N)C[Se][Se]CC(N)C(O)=O JULROCUWKLNBSN-UHFFFAOYSA-N 0.000 claims description 40
- LUTLAXLNPLZCOF-UHFFFAOYSA-N 1-Methylhistidine Natural products OC(=O)C(N)(C)CC1=NC=CN1 LUTLAXLNPLZCOF-UHFFFAOYSA-N 0.000 claims description 39
- PQNASZJZHFPQLE-UHFFFAOYSA-N N(6)-methyllysine Chemical compound CNCCCCC(N)C(O)=O PQNASZJZHFPQLE-UHFFFAOYSA-N 0.000 claims description 39
- NTWVQPHTOUKMDI-YFKPBYRVSA-N N-Methyl-arginine Chemical compound CN[C@H](C(O)=O)CCCN=C(N)N NTWVQPHTOUKMDI-YFKPBYRVSA-N 0.000 claims description 39
- JCZLABDVDPYLRZ-AWEZNQCLSA-N biphenylalanine Chemical compound C1=CC(C[C@H](N)C(O)=O)=CC=C1C1=CC=CC=C1 JCZLABDVDPYLRZ-AWEZNQCLSA-N 0.000 claims description 38
- BVAUMRCGVHUWOZ-ZETCQYMHSA-N (2s)-2-(cyclohexylazaniumyl)propanoate Chemical compound OC(=O)[C@H](C)NC1CCCCC1 BVAUMRCGVHUWOZ-ZETCQYMHSA-N 0.000 claims description 36
- SNDPXSYFESPGGJ-BYPYZUCNSA-N L-2-aminopentanoic acid Chemical compound CCC[C@H](N)C(O)=O SNDPXSYFESPGGJ-BYPYZUCNSA-N 0.000 claims description 36
- WAMWSIDTKSNDCU-ZETCQYMHSA-N (2s)-2-azaniumyl-2-cyclohexylacetate Chemical compound OC(=O)[C@@H](N)C1CCCCC1 WAMWSIDTKSNDCU-ZETCQYMHSA-N 0.000 claims description 35
- 125000000510 L-tryptophano group Chemical group [H]C1=C([H])C([H])=C2N([H])C([H])=C(C([H])([H])[C@@]([H])(C(O[H])=O)N([H])[*])C2=C1[H] 0.000 claims description 35
- NTNWOCRCBQPEKQ-YFKPBYRVSA-N N(omega)-methyl-L-arginine Chemical compound CN=C(N)NCCC[C@H](N)C(O)=O NTNWOCRCBQPEKQ-YFKPBYRVSA-N 0.000 claims description 34
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 33
- 108060003951 Immunoglobulin Proteins 0.000 claims description 32
- 102000018358 immunoglobulin Human genes 0.000 claims description 32
- NCHSYZVVWKVWFQ-BYPYZUCNSA-N Ng-amino-l-arginine Chemical compound NNC(N)=NCCC[C@H](N)C(O)=O NCHSYZVVWKVWFQ-BYPYZUCNSA-N 0.000 claims description 31
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 claims description 30
- 230000002378 acidificating effect Effects 0.000 claims description 30
- 239000004475 Arginine Substances 0.000 claims description 27
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 claims description 27
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 claims description 26
- 235000009697 arginine Nutrition 0.000 claims description 26
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Chemical compound CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 claims description 25
- 239000004471 Glycine Substances 0.000 claims description 24
- 235000004279 alanine Nutrition 0.000 claims description 17
- BZQFBWGGLXLEPQ-REOHCLBHSA-N phosphoserine Chemical compound OC(=O)[C@@H](N)COP(O)(O)=O BZQFBWGGLXLEPQ-REOHCLBHSA-N 0.000 claims description 16
- 229950006137 dexfosfoserine Drugs 0.000 claims description 15
- 125000005647 linker group Chemical group 0.000 claims description 15
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 claims description 14
- BZQFBWGGLXLEPQ-UHFFFAOYSA-N O-phosphoryl-L-serine Natural products OC(=O)C(N)COP(O)(O)=O BZQFBWGGLXLEPQ-UHFFFAOYSA-N 0.000 claims description 14
- UHBYWPGGCSDKFX-VKHMYHEASA-N gamma-carboxy-L-glutamic acid Chemical compound OC(=O)[C@@H](N)CC(C(O)=O)C(O)=O UHBYWPGGCSDKFX-VKHMYHEASA-N 0.000 claims description 14
- DCWXELXMIBXGTH-UHFFFAOYSA-N phosphotyrosine Chemical compound OC(=O)C(N)CC1=CC=C(OP(O)(O)=O)C=C1 DCWXELXMIBXGTH-UHFFFAOYSA-N 0.000 claims description 14
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 claims description 13
- 239000002253 acid Substances 0.000 claims description 13
- 125000003118 aryl group Chemical group 0.000 claims description 13
- 229930182817 methionine Natural products 0.000 claims description 13
- 235000006109 methionine Nutrition 0.000 claims description 13
- NRCSJHVDTAAISV-QMMMGPOBSA-N (2s)-2-amino-3-(3,4-dichlorophenyl)propanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(Cl)C(Cl)=C1 NRCSJHVDTAAISV-QMMMGPOBSA-N 0.000 claims description 11
- CRFFPDBJLGAGQL-QMMMGPOBSA-N (2s)-2-amino-3-[4-(trifluoromethyl)phenyl]propanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(C(F)(F)F)C=C1 CRFFPDBJLGAGQL-QMMMGPOBSA-N 0.000 claims description 11
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 11
- 150000003839 salts Chemical class 0.000 claims description 11
- CVZZNRXMDCOHBG-UHFFFAOYSA-N 2-azaniumyl-3-(2-chlorophenyl)propanoate Chemical compound OC(=O)C(N)CC1=CC=CC=C1Cl CVZZNRXMDCOHBG-UHFFFAOYSA-N 0.000 claims description 10
- NIGWMJHCCYYCSF-UHFFFAOYSA-N Fenclonine Chemical compound OC(=O)C(N)CC1=CC=C(Cl)C=C1 NIGWMJHCCYYCSF-UHFFFAOYSA-N 0.000 claims description 10
- 125000003282 alkyl amino group Chemical group 0.000 claims description 10
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 claims description 9
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 claims description 9
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 claims description 9
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 claims description 9
- 201000011384 erythromelalgia Diseases 0.000 claims description 9
- 229960000310 isoleucine Drugs 0.000 claims description 9
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 claims description 9
- 239000004474 valine Substances 0.000 claims description 9
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 claims description 8
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 claims description 8
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 8
- 229960001230 asparagine Drugs 0.000 claims description 8
- 235000009582 asparagine Nutrition 0.000 claims description 8
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 claims description 8
- 235000004554 glutamine Nutrition 0.000 claims description 8
- 235000004400 serine Nutrition 0.000 claims description 8
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 claims description 7
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 claims description 7
- 239000004473 Threonine Substances 0.000 claims description 7
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 claims description 7
- 125000001314 canonical amino-acid group Chemical group 0.000 claims description 7
- 235000008521 threonine Nutrition 0.000 claims description 7
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 7
- 229960004441 tyrosine Drugs 0.000 claims description 7
- CNMAQBJBWQQZFZ-LURJTMIESA-N (2s)-2-(pyridin-2-ylamino)propanoic acid Chemical compound OC(=O)[C@H](C)NC1=CC=CC=N1 CNMAQBJBWQQZFZ-LURJTMIESA-N 0.000 claims description 6
- WTKYBFQVZPCGAO-LURJTMIESA-N (2s)-2-(pyridin-3-ylamino)propanoic acid Chemical compound OC(=O)[C@H](C)NC1=CC=CN=C1 WTKYBFQVZPCGAO-LURJTMIESA-N 0.000 claims description 6
- 208000000094 Chronic Pain Diseases 0.000 claims description 6
- PMMYEEVYMWASQN-DMTCNVIQSA-N Hydroxyproline Chemical compound O[C@H]1CN[C@H](C(O)=O)C1 PMMYEEVYMWASQN-DMTCNVIQSA-N 0.000 claims description 6
- 208000037674 Primary erythromelalgia Diseases 0.000 claims description 6
- 125000000291 glutamic acid group Chemical group N[C@@H](CCC(O)=O)C(=O)* 0.000 claims description 6
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 claims description 6
- 229960005190 phenylalanine Drugs 0.000 claims description 6
- 229920001223 polyethylene glycol Polymers 0.000 claims description 6
- 208000017692 primary erythermalgia Diseases 0.000 claims description 6
- QVAZQYUQYQZVJV-LURJTMIESA-N (2s)-2-(piperidin-4-ylamino)propanoic acid Chemical compound OC(=O)[C@H](C)NC1CCNCC1 QVAZQYUQYQZVJV-LURJTMIESA-N 0.000 claims description 5
- SAAQPSNNIOGFSQ-LURJTMIESA-N (2s)-2-(pyridin-4-ylamino)propanoic acid Chemical compound OC(=O)[C@H](C)NC1=CC=NC=C1 SAAQPSNNIOGFSQ-LURJTMIESA-N 0.000 claims description 5
- 230000001154 acute effect Effects 0.000 claims description 5
- 235000003704 aspartic acid Nutrition 0.000 claims description 5
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 claims description 5
- PMMYEEVYMWASQN-UHFFFAOYSA-N dl-hydroxyproline Natural products OC1C[NH2+]C(C([O-])=O)C1 PMMYEEVYMWASQN-UHFFFAOYSA-N 0.000 claims description 5
- 229960002591 hydroxyproline Drugs 0.000 claims description 5
- 230000002085 persistent effect Effects 0.000 claims description 5
- SCIFESDRCALIIM-VIFPVBQESA-N N-methyl-L-phenylalanine Chemical compound C[NH2+][C@H](C([O-])=O)CC1=CC=CC=C1 SCIFESDRCALIIM-VIFPVBQESA-N 0.000 claims description 4
- 208000019865 paroxysmal extreme pain disease Diseases 0.000 claims description 4
- FGMPLJWBKKVCDB-UHFFFAOYSA-N trans-L-hydroxy-proline Natural products ON1CCCC1C(O)=O FGMPLJWBKKVCDB-UHFFFAOYSA-N 0.000 claims description 4
- YJKWJVAAWIUIFA-MRVPVSSYSA-N (2r)-2-azaniumyl-2-pyridin-2-ylpropanoate Chemical compound OC(=O)[C@@](N)(C)C1=CC=CC=N1 YJKWJVAAWIUIFA-MRVPVSSYSA-N 0.000 claims description 3
- LRHRHAWNXCGABU-UHFFFAOYSA-N 2-(cyclopentylazaniumyl)acetate Chemical compound OC(=O)CNC1CCCC1 LRHRHAWNXCGABU-UHFFFAOYSA-N 0.000 claims description 3
- YXDGRBPZVQPESQ-QMMMGPOBSA-N 4-[(2s)-2-amino-2-carboxyethyl]benzoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(C(O)=O)C=C1 YXDGRBPZVQPESQ-QMMMGPOBSA-N 0.000 claims description 3
- ZGUNAGUHMKGQNY-ZETCQYMHSA-N L-alpha-phenylglycine zwitterion Chemical compound OC(=O)[C@@H](N)C1=CC=CC=C1 ZGUNAGUHMKGQNY-ZETCQYMHSA-N 0.000 claims description 3
- SCIFESDRCALIIM-UHFFFAOYSA-N N-Me-Phenylalanine Natural products CNC(C(O)=O)CC1=CC=CC=C1 SCIFESDRCALIIM-UHFFFAOYSA-N 0.000 claims description 3
- AKCRVYNORCOYQT-YFKPBYRVSA-N N-methyl-L-valine Chemical compound CN[C@@H](C(C)C)C(O)=O AKCRVYNORCOYQT-YFKPBYRVSA-N 0.000 claims description 3
- 125000000729 N-terminal amino-acid group Chemical group 0.000 claims description 3
- 206010028980 Neoplasm Diseases 0.000 claims description 3
- 208000005298 acute pain Diseases 0.000 claims description 3
- 201000011510 cancer Diseases 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 3
- 125000001165 hydrophobic group Chemical group 0.000 claims description 3
- DZLNHFMRPBPULJ-UHFFFAOYSA-N thioproline Chemical compound OC(=O)C1CSCN1 DZLNHFMRPBPULJ-UHFFFAOYSA-N 0.000 claims description 3
- 208000006561 Cluster Headache Diseases 0.000 claims description 2
- 108091006905 Human Serum Albumin Proteins 0.000 claims description 2
- 102000008100 Human Serum Albumin Human genes 0.000 claims description 2
- 108010071690 Prealbumin Proteins 0.000 claims description 2
- 238000002512 chemotherapy Methods 0.000 claims description 2
- XJODGRWDFZVTKW-ZCFIWIBFSA-N n-methylleucine Chemical compound CN[C@@H](C(O)=O)CC(C)C XJODGRWDFZVTKW-ZCFIWIBFSA-N 0.000 claims description 2
- 238000001356 surgical procedure Methods 0.000 claims description 2
- 229950001139 timonacic Drugs 0.000 claims description 2
- 206010044652 trigeminal neuralgia Diseases 0.000 claims description 2
- 125000000998 L-alanino group Chemical group [H]N([*])[C@](C([H])([H])[H])([H])C(=O)O[H] 0.000 claims 11
- 125000000393 L-methionino group Chemical group [H]OC(=O)[C@@]([H])(N([H])[*])C([H])([H])C(SC([H])([H])[H])([H])[H] 0.000 claims 6
- 208000023890 Complex Regional Pain Syndromes Diseases 0.000 claims 2
- 208000006820 Arthralgia Diseases 0.000 claims 1
- 208000032131 Diabetic Neuropathies Diseases 0.000 claims 1
- 206010016059 Facial pain Diseases 0.000 claims 1
- 208000005741 Failed Back Surgery Syndrome Diseases 0.000 claims 1
- 208000001640 Fibromyalgia Diseases 0.000 claims 1
- 206010061218 Inflammation Diseases 0.000 claims 1
- 208000005615 Interstitial Cystitis Diseases 0.000 claims 1
- 208000008930 Low Back Pain Diseases 0.000 claims 1
- 208000019695 Migraine disease Diseases 0.000 claims 1
- KZNQNBZMBZJQJO-UHFFFAOYSA-N N-glycyl-L-proline Natural products NCC(=O)N1CCCC1C(O)=O KZNQNBZMBZJQJO-UHFFFAOYSA-N 0.000 claims 1
- 208000005890 Neuroma Diseases 0.000 claims 1
- 208000000450 Pelvic Pain Diseases 0.000 claims 1
- 239000002202 Polyethylene glycol Substances 0.000 claims 1
- 206010057239 Post laminectomy syndrome Diseases 0.000 claims 1
- 206010036376 Postherpetic Neuralgia Diseases 0.000 claims 1
- 102000007584 Prealbumin Human genes 0.000 claims 1
- 208000008765 Sciatica Diseases 0.000 claims 1
- 125000000217 alkyl group Chemical group 0.000 claims 1
- 125000000267 glycino group Chemical group [H]N([*])C([H])([H])C(=O)O[H] 0.000 claims 1
- KZNQNBZMBZJQJO-YFKPBYRVSA-N glyclproline Chemical compound NCC(=O)N1CCC[C@H]1C(O)=O KZNQNBZMBZJQJO-YFKPBYRVSA-N 0.000 claims 1
- 108010077515 glycylproline Proteins 0.000 claims 1
- 230000004054 inflammatory process Effects 0.000 claims 1
- 208000014674 injury Diseases 0.000 claims 1
- 206010027599 migraine Diseases 0.000 claims 1
- 201000008482 osteoarthritis Diseases 0.000 claims 1
- 208000005877 painful neuropathy Diseases 0.000 claims 1
- 230000008733 trauma Effects 0.000 claims 1
- 239000003112 inhibitor Substances 0.000 abstract description 18
- 101000684820 Homo sapiens Sodium channel protein type 3 subunit alpha Proteins 0.000 abstract description 2
- 102100031367 Sodium channel protein type 9 subunit alpha Human genes 0.000 abstract description 2
- VHPXWPAGHGHDCK-UHFFFAOYSA-N 5-chloro-4-[(3,4-dichlorophenoxy)methyl]-2-fluoro-n-methylsulfonylbenzamide Chemical compound C1=C(F)C(C(=O)NS(=O)(=O)C)=CC(Cl)=C1COC1=CC=C(Cl)C(Cl)=C1 VHPXWPAGHGHDCK-UHFFFAOYSA-N 0.000 abstract 1
- 101000654386 Homo sapiens Sodium channel protein type 9 subunit alpha Proteins 0.000 abstract 1
- 229940124777 Nav1.7 inhibitor Drugs 0.000 abstract 1
- 102100023720 Sodium channel protein type 3 subunit alpha Human genes 0.000 abstract 1
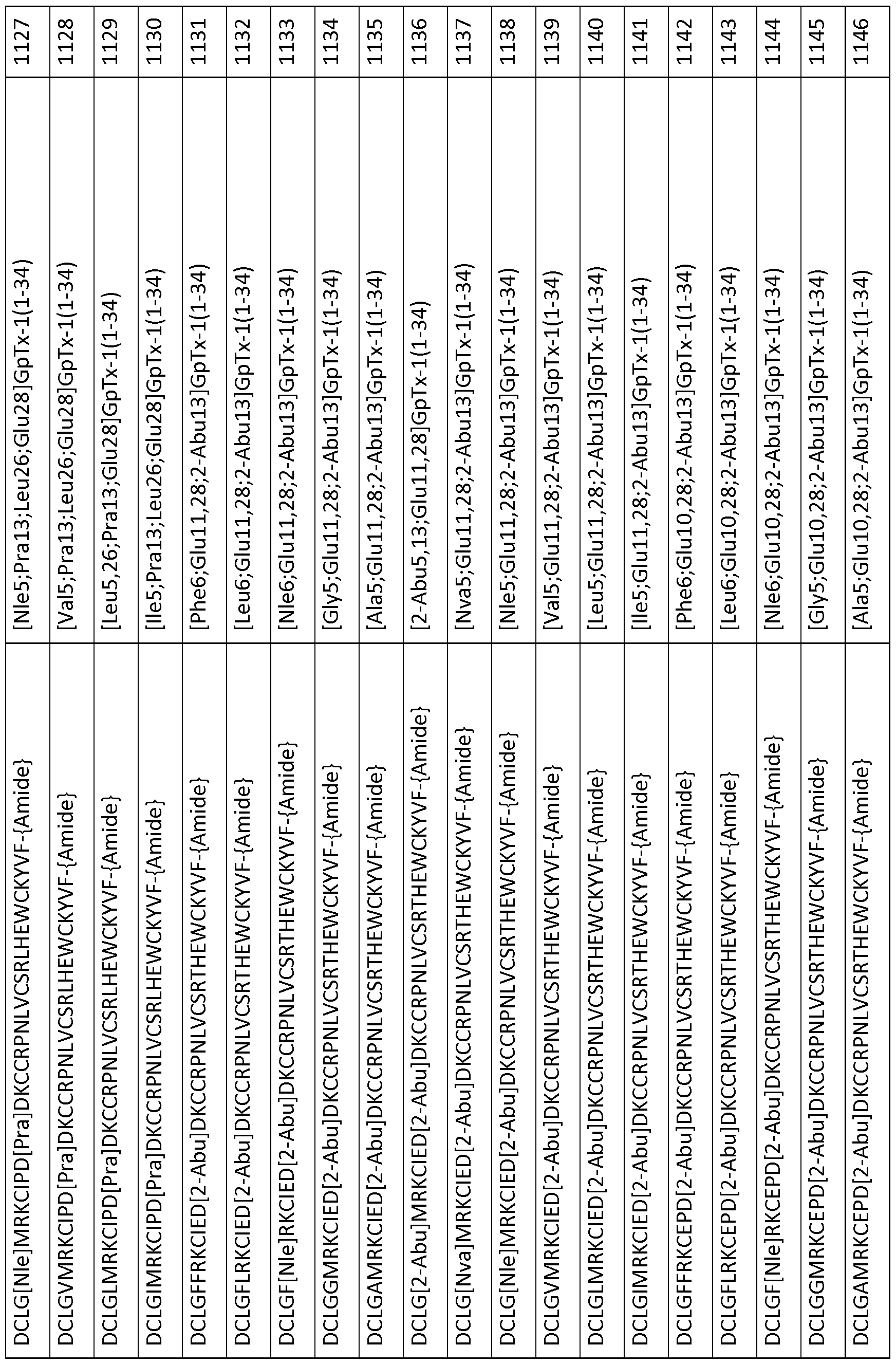
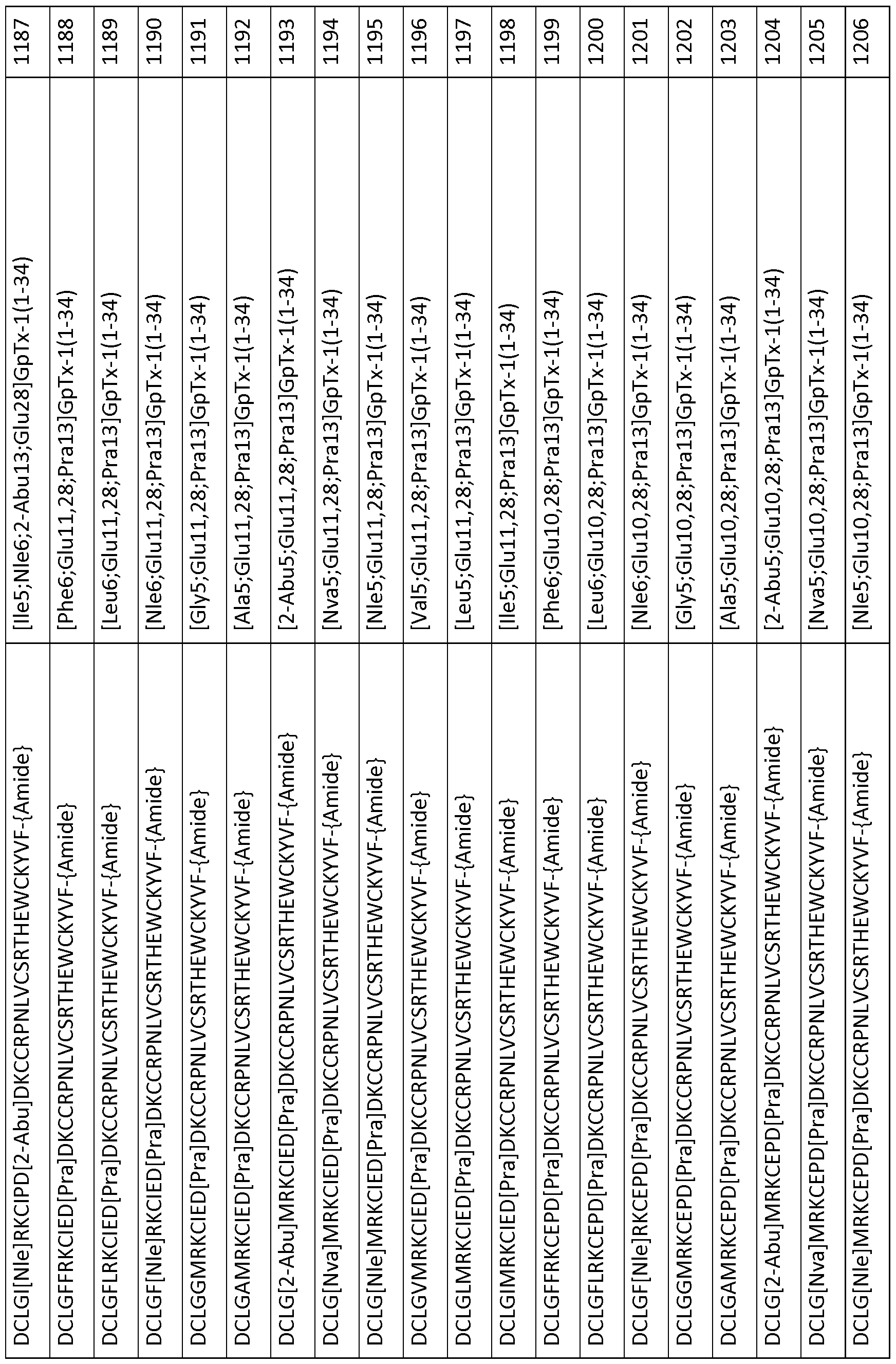
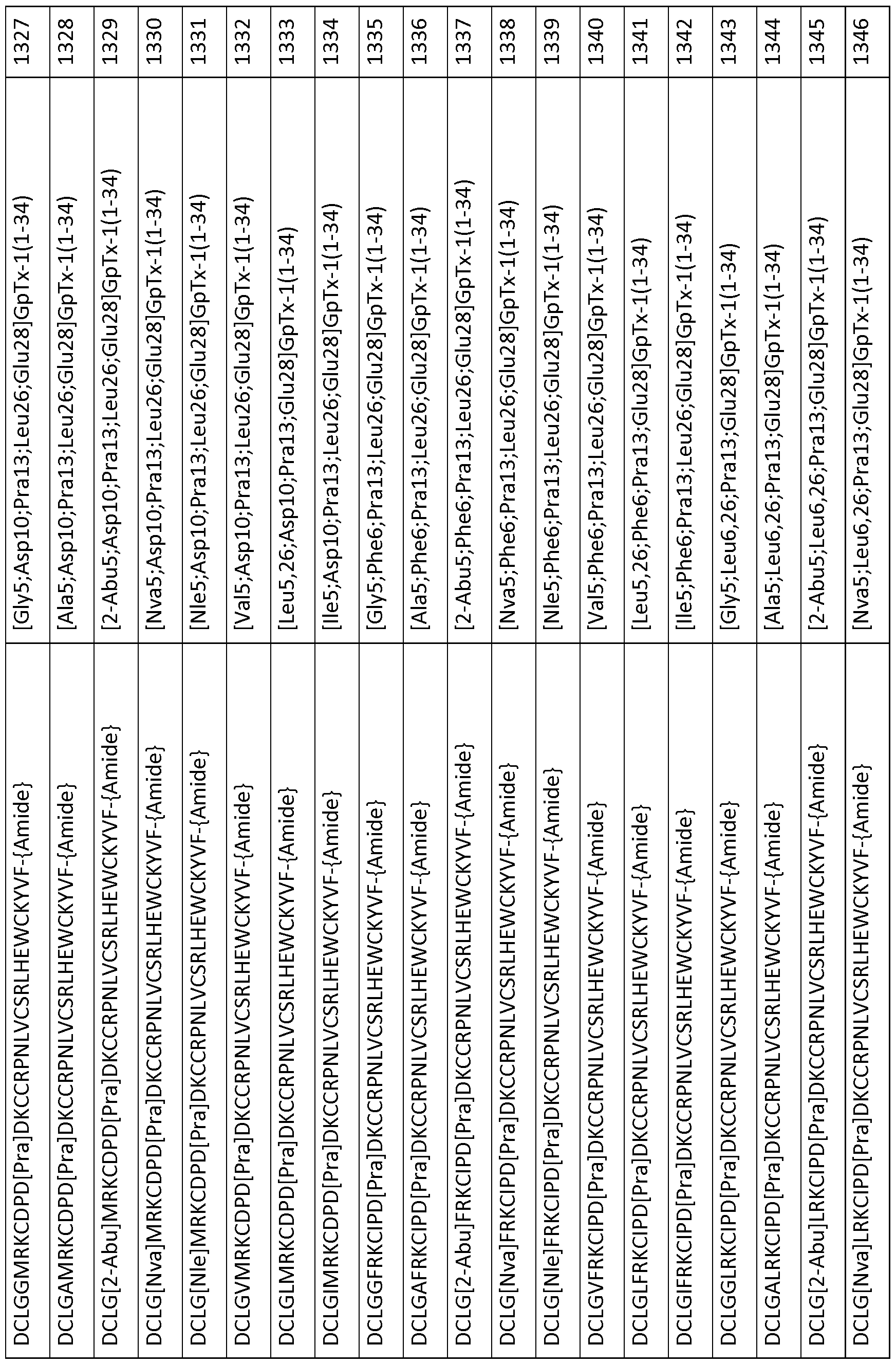
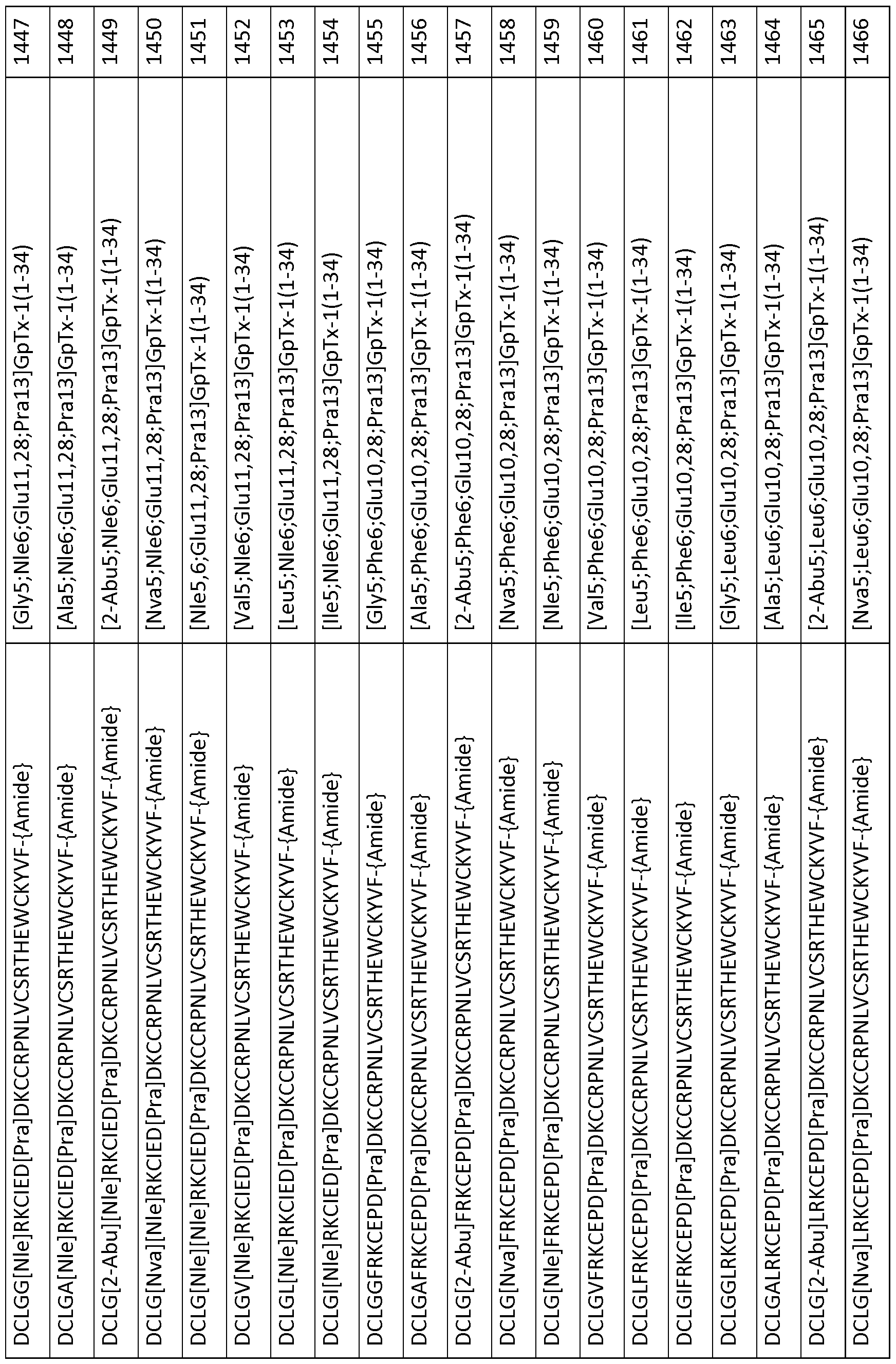
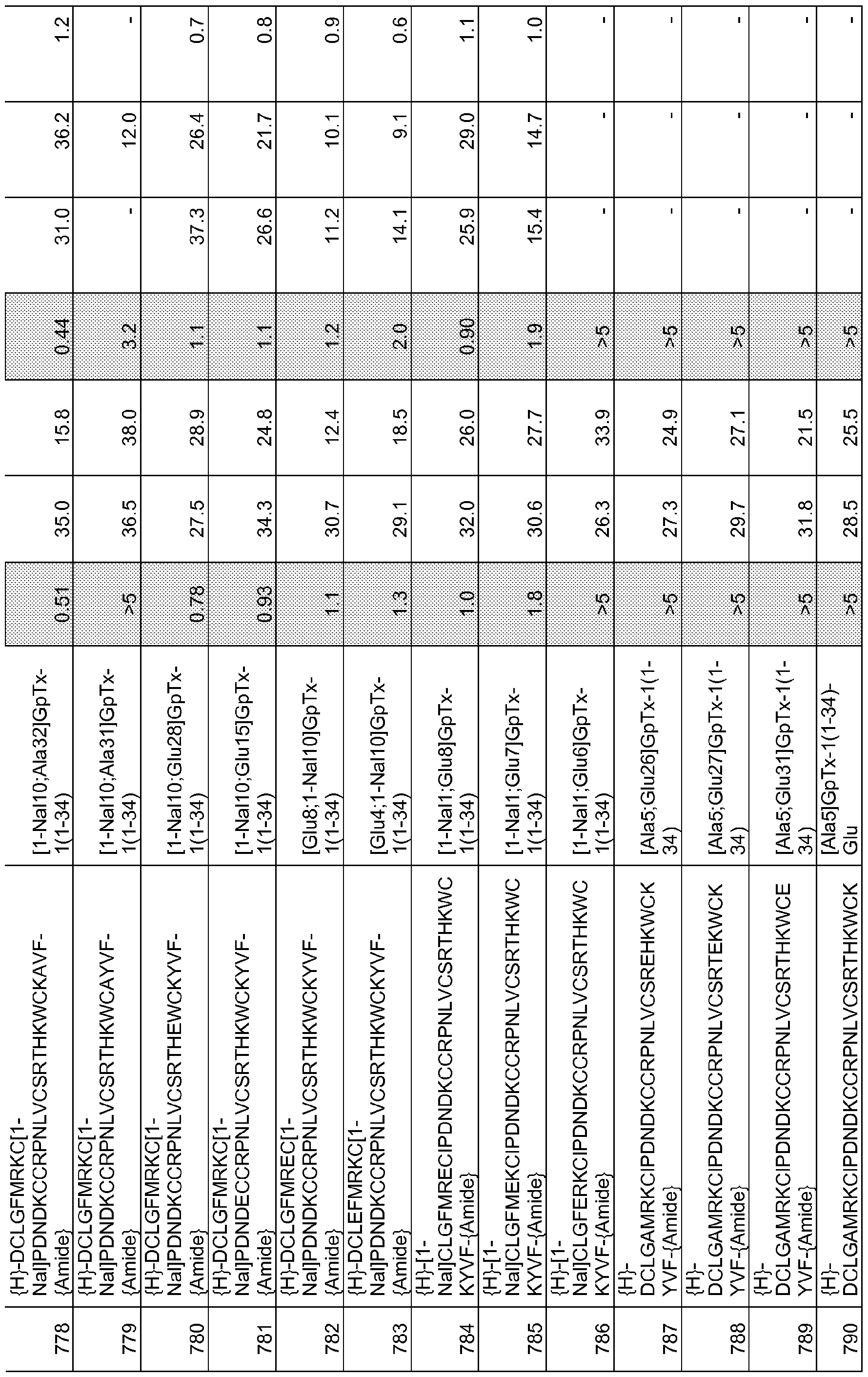
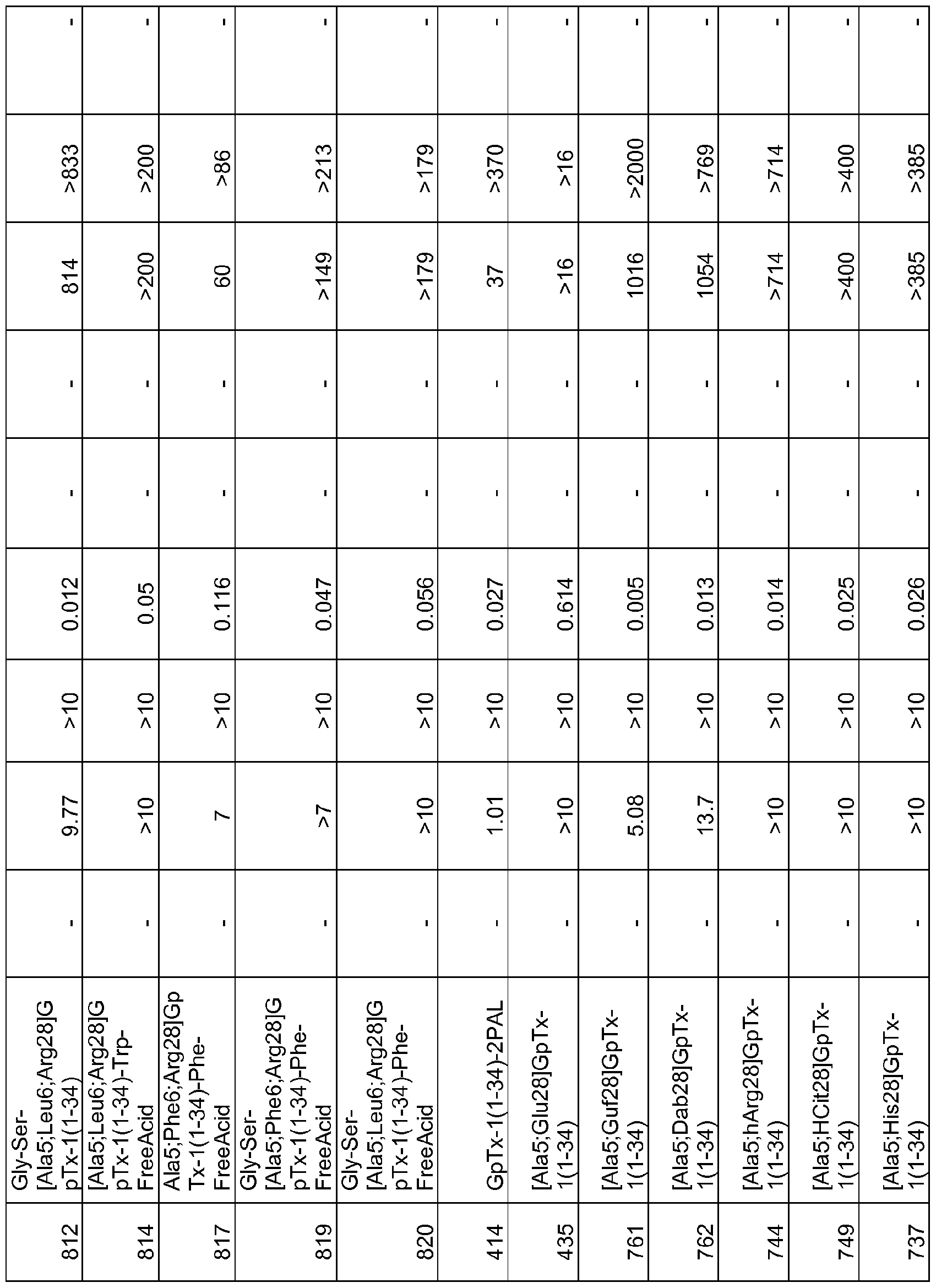
- 101000844072 Grammostola rosea Toxin GTx1-15 Proteins 0.000 description 378
- 150000001408 amides Chemical class 0.000 description 356
- 239000003053 toxin Substances 0.000 description 119
- 210000004027 cell Anatomy 0.000 description 116
- 231100000765 toxin Toxicity 0.000 description 111
- 108700012359 toxins Proteins 0.000 description 111
- 108090000623 proteins and genes Proteins 0.000 description 105
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 92
- 230000000694 effects Effects 0.000 description 89
- 102000004169 proteins and genes Human genes 0.000 description 84
- 235000018102 proteins Nutrition 0.000 description 79
- 238000007920 subcutaneous administration Methods 0.000 description 79
- 239000000562 conjugate Substances 0.000 description 73
- 108091006146 Channels Proteins 0.000 description 72
- CFMYXEVWODSLAX-QOZOJKKESA-N tetrodotoxin Chemical compound O([C@@]([C@H]1O)(O)O[C@H]2[C@@]3(O)CO)[C@H]3[C@@H](O)[C@]11[C@H]2[C@@H](O)N=C(N)N1 CFMYXEVWODSLAX-QOZOJKKESA-N 0.000 description 62
- CFMYXEVWODSLAX-UHFFFAOYSA-N tetrodotoxin Natural products C12C(O)NC(=N)NC2(C2O)C(O)C3C(CO)(O)C1OC2(O)O3 CFMYXEVWODSLAX-UHFFFAOYSA-N 0.000 description 62
- 229950010357 tetrodotoxin Drugs 0.000 description 62
- 239000011734 sodium Substances 0.000 description 53
- BQJCRHHNABKAKU-KBQPJGBKSA-N morphine Chemical compound O([C@H]1[C@H](C=C[C@H]23)O)C4=C5[C@@]12CCN(C)[C@@H]3CC5=CC=C4O BQJCRHHNABKAKU-KBQPJGBKSA-N 0.000 description 52
- 241000699670 Mus sp. Species 0.000 description 51
- 238000002203 pretreatment Methods 0.000 description 50
- 239000012071 phase Substances 0.000 description 44
- 125000003275 alpha amino acid group Chemical group 0.000 description 43
- 230000001965 increasing effect Effects 0.000 description 42
- 238000007792 addition Methods 0.000 description 41
- 238000002347 injection Methods 0.000 description 40
- 239000007924 injection Substances 0.000 description 40
- 239000003981 vehicle Substances 0.000 description 38
- 102000018674 Sodium Channels Human genes 0.000 description 34
- 108010052164 Sodium Channels Proteins 0.000 description 34
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 32
- 238000006467 substitution reaction Methods 0.000 description 32
- 102000053602 DNA Human genes 0.000 description 30
- 210000002569 neuron Anatomy 0.000 description 30
- 210000002683 foot Anatomy 0.000 description 29
- 239000000178 monomer Substances 0.000 description 27
- 108020004414 DNA Proteins 0.000 description 26
- 229960005181 morphine Drugs 0.000 description 26
- 230000014509 gene expression Effects 0.000 description 23
- 210000003594 spinal ganglia Anatomy 0.000 description 23
- 231100000611 venom Toxicity 0.000 description 23
- 239000000306 component Substances 0.000 description 22
- 230000002779 inactivation Effects 0.000 description 22
- 230000033001 locomotion Effects 0.000 description 22
- 239000002435 venom Substances 0.000 description 22
- 210000001048 venom Anatomy 0.000 description 22
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 20
- BRMWTNUJHUMWMS-LURJTMIESA-N N(tele)-methyl-L-histidine Chemical group CN1C=NC(C[C@H](N)C(O)=O)=C1 BRMWTNUJHUMWMS-LURJTMIESA-N 0.000 description 20
- 238000003776 cleavage reaction Methods 0.000 description 20
- 230000006742 locomotor activity Effects 0.000 description 20
- 230000000384 rearing effect Effects 0.000 description 20
- 230000007017 scission Effects 0.000 description 20
- 102000035195 Peptidases Human genes 0.000 description 19
- 108091005804 Peptidases Proteins 0.000 description 19
- 210000004899 c-terminal region Anatomy 0.000 description 17
- 230000004927 fusion Effects 0.000 description 17
- 230000009467 reduction Effects 0.000 description 17
- 238000006722 reduction reaction Methods 0.000 description 17
- 230000002829 reductive effect Effects 0.000 description 17
- 108010053752 Voltage-Gated Sodium Channels Proteins 0.000 description 16
- 102000016913 Voltage-Gated Sodium Channels Human genes 0.000 description 16
- 231100000673 dose–response relationship Toxicity 0.000 description 16
- 125000003729 nucleotide group Chemical group 0.000 description 16
- 239000013641 positive control Substances 0.000 description 16
- 239000004365 Protease Substances 0.000 description 15
- 230000006399 behavior Effects 0.000 description 15
- 235000018417 cysteine Nutrition 0.000 description 15
- 230000004048 modification Effects 0.000 description 15
- 238000012986 modification Methods 0.000 description 15
- 239000000243 solution Substances 0.000 description 15
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 14
- 241000699666 Mus <mouse, genus> Species 0.000 description 14
- 238000005481 NMR spectroscopy Methods 0.000 description 14
- 238000006243 chemical reaction Methods 0.000 description 14
- 230000036470 plasma concentration Effects 0.000 description 14
- 102000004310 Ion Channels Human genes 0.000 description 13
- 108090000862 Ion Channels Proteins 0.000 description 13
- 206010030113 Oedema Diseases 0.000 description 13
- 230000027455 binding Effects 0.000 description 13
- XOAUGYVLRSCGBG-UHFFFAOYSA-N protx ii Chemical compound O=C1NC(CCC(N)=O)C(=O)NC(CCCCN)C(=O)NC(CC=2C3=CC=CC=C3NC=2)C(=O)NC(CCSC)C(=O)NC(CC=2C3=CC=CC=C3NC=2)C(=O)NC(C(C)O)C(=O)NC(C(NC(CC(O)=O)C(=O)NC(CO)C(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NC(CCCCN)C(=O)NC(CSSCC(NC(=O)C(CC=2C3=CC=CC=C3NC=2)NC(=O)C(CC(C)C)NC(=O)C(CCCNC(N)=N)NC2=O)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CCCCN)C(=O)NC(CC(C)C)C(=O)NC(CC=3C4=CC=CC=C4NC=3)C(O)=O)C(=O)N3)=O)CSSCC2NC(=O)C(C(C)C)NC(=O)C(CCSC)NC(=O)CNC(=O)C(CCC(O)=O)NC(=O)C3CSSCC1NC(=O)C(N)CC1=CC=C(O)C=C1 XOAUGYVLRSCGBG-UHFFFAOYSA-N 0.000 description 13
- 108091028043 Nucleic acid sequence Proteins 0.000 description 12
- 241000239226 Scorpiones Species 0.000 description 12
- 101000844259 Thrixopelma pruriens Beta/omega-theraphotoxin-Tp2a Proteins 0.000 description 12
- 230000004913 activation Effects 0.000 description 12
- 239000007787 solid Substances 0.000 description 12
- 108091026890 Coding region Proteins 0.000 description 11
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 11
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 11
- 239000002773 nucleotide Substances 0.000 description 11
- 229910052708 sodium Inorganic materials 0.000 description 11
- VLPIATFUUWWMKC-SNVBAGLBSA-N (2r)-1-(2,6-dimethylphenoxy)propan-2-amine Chemical compound C[C@@H](N)COC1=C(C)C=CC=C1C VLPIATFUUWWMKC-SNVBAGLBSA-N 0.000 description 10
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 10
- ODKSFYDXXFIFQN-BYPYZUCNSA-N L-arginine Chemical compound OC(=O)[C@@H](N)CCCN=C(N)N ODKSFYDXXFIFQN-BYPYZUCNSA-N 0.000 description 10
- 239000004472 Lysine Substances 0.000 description 10
- 241000700159 Rattus Species 0.000 description 10
- 108700019146 Transgenes Proteins 0.000 description 10
- 230000021615 conjugation Effects 0.000 description 10
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 10
- 239000003814 drug Substances 0.000 description 10
- 229960003404 mexiletine Drugs 0.000 description 10
- 230000035772 mutation Effects 0.000 description 10
- 102000040430 polynucleotide Human genes 0.000 description 10
- 108091033319 polynucleotide Proteins 0.000 description 10
- 239000002157 polynucleotide Substances 0.000 description 10
- UQDJGEHQDNVPGU-UHFFFAOYSA-N serine phosphoethanolamine Chemical compound [NH3+]CCOP([O-])(=O)OCC([NH3+])C([O-])=O UQDJGEHQDNVPGU-UHFFFAOYSA-N 0.000 description 10
- 239000000126 substance Substances 0.000 description 10
- 229960002685 biotin Drugs 0.000 description 9
- 235000020958 biotin Nutrition 0.000 description 9
- 239000011616 biotin Substances 0.000 description 9
- 230000006287 biotinylation Effects 0.000 description 9
- 238000007413 biotinylation Methods 0.000 description 9
- 108020001507 fusion proteins Proteins 0.000 description 9
- 102000037865 fusion proteins Human genes 0.000 description 9
- 125000001360 methionine group Chemical group N[C@@H](CCSC)C(=O)* 0.000 description 9
- 238000013518 transcription Methods 0.000 description 9
- 230000035897 transcription Effects 0.000 description 9
- 101100228200 Caenorhabditis elegans gly-5 gene Proteins 0.000 description 8
- 108020004705 Codon Proteins 0.000 description 8
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 8
- 108091034117 Oligonucleotide Proteins 0.000 description 8
- 230000000903 blocking effect Effects 0.000 description 8
- 230000006870 function Effects 0.000 description 8
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 8
- 235000014304 histidine Nutrition 0.000 description 8
- 235000018977 lysine Nutrition 0.000 description 8
- 101710179441 Huwentoxin-IV Proteins 0.000 description 7
- 101800003415 Mu-conotoxin KIIIA Proteins 0.000 description 7
- 238000002835 absorbance Methods 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 7
- 210000000170 cell membrane Anatomy 0.000 description 7
- 150000001875 compounds Chemical class 0.000 description 7
- 238000012217 deletion Methods 0.000 description 7
- 230000037430 deletion Effects 0.000 description 7
- 108010042252 huwentoxin I Proteins 0.000 description 7
- MJMLBAPXMAOKDU-UHFFFAOYSA-N huwentoxin iv Chemical compound C1SSCC(C(NC(CSSCC(NC(=O)C(N)CCC(O)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(C(C)CC)C(=O)NC(CC=2C=CC=CC=2)C(=O)NC(CCCCN)C(=O)NC(C)C(=O)N2)C(=O)NC(CCCCN)C(=O)NC(CO)C(=O)NC(CO)C(=O)NC(CCCCN)C(=O)NC(CC(C)C)C(=O)NC(C(=O)N3)C(C)C)=O)NC(=O)C(CCC(N)=O)NC(=O)C(CC(O)=O)NC(=O)C(CC(N)=O)NC(=O)C(CO)NC(=O)C4CCCN4C(=O)C(CC(N)=O)NC(=O)C2CSSCC3C(=O)NC(CO)C(=O)NC(CCCNC(N)=N)C(=O)NC(CCCCN)C(=O)NC(C(C)O)C(=O)NC(CCCNC(N)=N)C(=O)NC(CC=2C3=CC=CC=C3NC=2)C(=O)NC1C(=O)NC(CCCCN)C(=O)NC(C(=O)NC(CCC(N)=O)C(=O)NC(C(C)CC)C(N)=O)CC1=CC=C(O)C=C1 MJMLBAPXMAOKDU-UHFFFAOYSA-N 0.000 description 7
- 210000003000 inclusion body Anatomy 0.000 description 7
- 230000005764 inhibitory process Effects 0.000 description 7
- 208000004296 neuralgia Diseases 0.000 description 7
- 235000019419 proteases Nutrition 0.000 description 7
- 230000004044 response Effects 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 241000242759 Actiniaria Species 0.000 description 6
- 241000124008 Mammalia Species 0.000 description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- 102000003729 Neprilysin Human genes 0.000 description 6
- 108090000028 Neprilysin Proteins 0.000 description 6
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 6
- 239000005557 antagonist Substances 0.000 description 6
- 238000003556 assay Methods 0.000 description 6
- UCMIRNVEIXFBKS-UHFFFAOYSA-N beta-alanine Chemical compound NCCC(O)=O UCMIRNVEIXFBKS-UHFFFAOYSA-N 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 238000005341 cation exchange Methods 0.000 description 6
- 230000007423 decrease Effects 0.000 description 6
- 239000003398 denaturant Substances 0.000 description 6
- 206010015037 epilepsy Diseases 0.000 description 6
- 238000002474 experimental method Methods 0.000 description 6
- 230000021824 exploration behavior Effects 0.000 description 6
- 230000003993 interaction Effects 0.000 description 6
- 238000007912 intraperitoneal administration Methods 0.000 description 6
- 230000001537 neural effect Effects 0.000 description 6
- 208000021722 neuropathic pain Diseases 0.000 description 6
- 238000010647 peptide synthesis reaction Methods 0.000 description 6
- 210000002381 plasma Anatomy 0.000 description 6
- 102000005962 receptors Human genes 0.000 description 6
- 108020003175 receptors Proteins 0.000 description 6
- 238000011160 research Methods 0.000 description 6
- 239000002795 scorpion venom Substances 0.000 description 6
- 238000010186 staining Methods 0.000 description 6
- 239000000758 substrate Substances 0.000 description 6
- 238000003786 synthesis reaction Methods 0.000 description 6
- 229940124597 therapeutic agent Drugs 0.000 description 6
- 238000011282 treatment Methods 0.000 description 6
- 241000239290 Araneae Species 0.000 description 5
- 108090001008 Avidin Proteins 0.000 description 5
- 241000894006 Bacteria Species 0.000 description 5
- 102000004190 Enzymes Human genes 0.000 description 5
- 108090000790 Enzymes Proteins 0.000 description 5
- 241001034728 Grammostola porteri Species 0.000 description 5
- 241001157778 Haplopelma schmidti Species 0.000 description 5
- 101710138657 Neurotoxin Proteins 0.000 description 5
- 241000239299 Orthochirus scrobiculosus Species 0.000 description 5
- 108010029485 Protein Isoforms Proteins 0.000 description 5
- 102000001708 Protein Isoforms Human genes 0.000 description 5
- 108010076504 Protein Sorting Signals Proteins 0.000 description 5
- 108020004511 Recombinant DNA Proteins 0.000 description 5
- 101150080511 Scn9a gene Proteins 0.000 description 5
- 241000239292 Theraphosidae Species 0.000 description 5
- FVECELJHCSPHKY-UHFFFAOYSA-N Veratridine Natural products C1=C(OC)C(OC)=CC=C1C(=O)OC1C2(O)OC34CC5(O)C(CN6C(CCC(C)C6)C6(C)O)C6(O)C(O)CC5(O)C4CCC2C3(C)CC1 FVECELJHCSPHKY-UHFFFAOYSA-N 0.000 description 5
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 5
- 230000036982 action potential Effects 0.000 description 5
- QWCKQJZIFLGMSD-UHFFFAOYSA-N alpha-aminobutyric acid Chemical compound CCC(N)C(O)=O QWCKQJZIFLGMSD-UHFFFAOYSA-N 0.000 description 5
- 230000004071 biological effect Effects 0.000 description 5
- 210000003169 central nervous system Anatomy 0.000 description 5
- 239000003623 enhancer Substances 0.000 description 5
- 229940088598 enzyme Drugs 0.000 description 5
- 210000001723 extracellular space Anatomy 0.000 description 5
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical class OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 description 5
- 239000000499 gel Substances 0.000 description 5
- 229930195712 glutamate Natural products 0.000 description 5
- 239000000710 homodimer Substances 0.000 description 5
- 239000002581 neurotoxin Substances 0.000 description 5
- 231100000618 neurotoxin Toxicity 0.000 description 5
- 230000003040 nociceptive effect Effects 0.000 description 5
- 239000002953 phosphate buffered saline Substances 0.000 description 5
- 229910052700 potassium Inorganic materials 0.000 description 5
- 230000036515 potency Effects 0.000 description 5
- 150000003141 primary amines Chemical class 0.000 description 5
- 125000001554 selenocysteine group Chemical group [H][Se]C([H])([H])C(N([H])[H])C(=O)O* 0.000 description 5
- 238000000926 separation method Methods 0.000 description 5
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 5
- 239000007790 solid phase Substances 0.000 description 5
- FVECELJHCSPHKY-JLSHOZRYSA-N veratridine Chemical compound C1=C(OC)C(OC)=CC=C1C(=O)O[C@@H]1[C@@]2(O)O[C@]34C[C@@]5(O)[C@H](CN6[C@@H](CC[C@H](C)C6)[C@@]6(C)O)[C@]6(O)[C@@H](O)C[C@@]5(O)[C@@H]4CC[C@H]2[C@]3(C)CC1 FVECELJHCSPHKY-JLSHOZRYSA-N 0.000 description 5
- QJYNZEYHSMRWBK-NIKIMHBISA-N 1,2,3,4,6-pentakis-O-galloyl-beta-D-glucose Chemical compound OC1=C(O)C(O)=CC(C(=O)OC[C@@H]2[C@H]([C@H](OC(=O)C=3C=C(O)C(O)=C(O)C=3)[C@@H](OC(=O)C=3C=C(O)C(O)=C(O)C=3)[C@H](OC(=O)C=3C=C(O)C(O)=C(O)C=3)O2)OC(=O)C=2C=C(O)C(O)=C(O)C=2)=C1 QJYNZEYHSMRWBK-NIKIMHBISA-N 0.000 description 4
- 206010001497 Agitation Diseases 0.000 description 4
- 241000238860 Chrysiogenetes <class> Species 0.000 description 4
- LEVWYRKDKASIDU-QWWZWVQMSA-N D-cystine Chemical compound OC(=O)[C@H](N)CSSC[C@@H](N)C(O)=O LEVWYRKDKASIDU-QWWZWVQMSA-N 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- 241000588724 Escherichia coli Species 0.000 description 4
- 108010051041 HC toxin Proteins 0.000 description 4
- 241000282412 Homo Species 0.000 description 4
- RHGKLRLOHDJJDR-BYPYZUCNSA-N L-citrulline Chemical compound NC(=O)NCCC[C@H]([NH3+])C([O-])=O RHGKLRLOHDJJDR-BYPYZUCNSA-N 0.000 description 4
- 241001465754 Metazoa Species 0.000 description 4
- NQTADLQHYWFPDB-UHFFFAOYSA-N N-Hydroxysuccinimide Chemical compound ON1C(=O)CCC1=O NQTADLQHYWFPDB-UHFFFAOYSA-N 0.000 description 4
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 4
- 102000004257 Potassium Channel Human genes 0.000 description 4
- 206010060862 Prostate cancer Diseases 0.000 description 4
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 4
- 108090000631 Trypsin Proteins 0.000 description 4
- 102000004142 Trypsin Human genes 0.000 description 4
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 4
- 230000000202 analgesic effect Effects 0.000 description 4
- 229940127131 antibody-peptide-conjugate Drugs 0.000 description 4
- 239000007864 aqueous solution Substances 0.000 description 4
- 229940009098 aspartate Drugs 0.000 description 4
- 230000001413 cellular effect Effects 0.000 description 4
- 238000001311 chemical methods and process Methods 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- 229960003067 cystine Drugs 0.000 description 4
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 4
- 238000000855 fermentation Methods 0.000 description 4
- 230000004151 fermentation Effects 0.000 description 4
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 4
- 238000001727 in vivo Methods 0.000 description 4
- 230000002401 inhibitory effect Effects 0.000 description 4
- 238000003780 insertion Methods 0.000 description 4
- 230000037431 insertion Effects 0.000 description 4
- 150000002500 ions Chemical class 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 210000004379 membrane Anatomy 0.000 description 4
- 239000003607 modifier Substances 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 239000011591 potassium Substances 0.000 description 4
- 108020001213 potassium channel Proteins 0.000 description 4
- 238000012545 processing Methods 0.000 description 4
- 235000019833 protease Nutrition 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 239000011541 reaction mixture Substances 0.000 description 4
- 238000004007 reversed phase HPLC Methods 0.000 description 4
- RPQXVSUAYFXFJA-HGRQIUPRSA-N saxitoxin Chemical compound NC(=O)OC[C@@H]1N=C(N)N2CCC(O)(O)[C@@]22N=C(N)N[C@@H]12 RPQXVSUAYFXFJA-HGRQIUPRSA-N 0.000 description 4
- RPQXVSUAYFXFJA-UHFFFAOYSA-N saxitoxin hydrate Natural products NC(=O)OCC1N=C(N)N2CCC(O)(O)C22NC(N)=NC12 RPQXVSUAYFXFJA-UHFFFAOYSA-N 0.000 description 4
- 235000016491 selenocysteine Nutrition 0.000 description 4
- 239000003195 sodium channel blocking agent Substances 0.000 description 4
- 230000002269 spontaneous effect Effects 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 238000001890 transfection Methods 0.000 description 4
- 238000013519 translation Methods 0.000 description 4
- 239000012588 trypsin Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- JUIKUQOUMZUFQT-UHFFFAOYSA-N 2-bromoacetamide Chemical group NC(=O)CBr JUIKUQOUMZUFQT-UHFFFAOYSA-N 0.000 description 3
- PECYZEOJVXMISF-UHFFFAOYSA-N 3-aminoalanine Chemical compound [NH3+]CC(N)C([O-])=O PECYZEOJVXMISF-UHFFFAOYSA-N 0.000 description 3
- BXRLWGXPSRYJDZ-UHFFFAOYSA-N 3-cyanoalanine Chemical compound OC(=O)C(N)CC#N BXRLWGXPSRYJDZ-UHFFFAOYSA-N 0.000 description 3
- 101100289894 Caenorhabditis elegans lys-7 gene Proteins 0.000 description 3
- 108090000317 Chymotrypsin Proteins 0.000 description 3
- 241001638933 Cochlicella barbara Species 0.000 description 3
- 241000168499 Conus kinoshitai Species 0.000 description 3
- 101000844746 Drosophila melanogaster Drosomycin Proteins 0.000 description 3
- 102000005593 Endopeptidases Human genes 0.000 description 3
- 108010059378 Endopeptidases Proteins 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 108091029865 Exogenous DNA Proteins 0.000 description 3
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 3
- 241000238631 Hexapoda Species 0.000 description 3
- 238000006736 Huisgen cycloaddition reaction Methods 0.000 description 3
- 208000004454 Hyperalgesia Diseases 0.000 description 3
- 206010065390 Inflammatory pain Diseases 0.000 description 3
- DGYHPLMPMRKMPD-UHFFFAOYSA-N L-propargyl glycine Natural products OC(=O)C(N)CC#C DGYHPLMPMRKMPD-UHFFFAOYSA-N 0.000 description 3
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 3
- 102000012479 Serine Proteases Human genes 0.000 description 3
- 108010022999 Serine Proteases Proteins 0.000 description 3
- 238000012867 alanine scanning Methods 0.000 description 3
- 230000006229 amino acid addition Effects 0.000 description 3
- 125000003277 amino group Chemical group 0.000 description 3
- 230000001580 bacterial effect Effects 0.000 description 3
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 238000012512 characterization method Methods 0.000 description 3
- 229960002376 chymotrypsin Drugs 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- 239000005547 deoxyribonucleotide Substances 0.000 description 3
- 125000002637 deoxyribonucleotide group Chemical group 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 239000000539 dimer Substances 0.000 description 3
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 3
- 229940125436 dual inhibitor Drugs 0.000 description 3
- 239000000975 dye Substances 0.000 description 3
- 238000005194 fractionation Methods 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 230000002068 genetic effect Effects 0.000 description 3
- 230000013595 glycosylation Effects 0.000 description 3
- 238000006206 glycosylation reaction Methods 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 3
- 210000004962 mammalian cell Anatomy 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 230000001404 mediated effect Effects 0.000 description 3
- 108020004999 messenger RNA Proteins 0.000 description 3
- 210000005036 nerve Anatomy 0.000 description 3
- 230000000269 nucleophilic effect Effects 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 230000001590 oxidative effect Effects 0.000 description 3
- 230000037361 pathway Effects 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 229940125422 potassium channel blocker Drugs 0.000 description 3
- 239000003450 potassium channel blocker Substances 0.000 description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 3
- 125000001500 prolyl group Chemical group [H]N1C([H])(C(=O)[*])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 3
- 125000006239 protecting group Chemical group 0.000 description 3
- 230000017854 proteolysis Effects 0.000 description 3
- 238000011084 recovery Methods 0.000 description 3
- 230000003248 secreting effect Effects 0.000 description 3
- 210000002027 skeletal muscle Anatomy 0.000 description 3
- 238000010532 solid phase synthesis reaction Methods 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- DLFVBJFMPXGRIB-UHFFFAOYSA-N thioacetamide Natural products CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 3
- 230000009466 transformation Effects 0.000 description 3
- 150000003852 triazoles Chemical class 0.000 description 3
- 239000013638 trimer Substances 0.000 description 3
- 239000013598 vector Substances 0.000 description 3
- BPKIMPVREBSLAJ-QTBYCLKRSA-N ziconotide Chemical compound C([C@H]1C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]2C(=O)N[C@@H]3C(=O)N[C@H](C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@H](C(N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CSSC2)C(N)=O)=O)CSSC[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@H](CCCCN)NC(=O)CNC(=O)[C@H](CCCCN)NC(=O)[C@@H](N)CSSC3)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(N1)=O)CCSC)[C@@H](C)O)C1=CC=C(O)C=C1 BPKIMPVREBSLAJ-QTBYCLKRSA-N 0.000 description 3
- WMBWREPUVVBILR-WIYYLYMNSA-N (-)-Epigallocatechin-3-o-gallate Chemical compound O([C@@H]1CC2=C(O)C=C(C=C2O[C@@H]1C=1C=C(O)C(O)=C(O)C=1)O)C(=O)C1=CC(O)=C(O)C(O)=C1 WMBWREPUVVBILR-WIYYLYMNSA-N 0.000 description 2
- YCHNJQXUYMPHNB-BYPYZUCNSA-N (2s)-2-amino-3-(2h-triazol-4-yl)propanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CNN=N1 YCHNJQXUYMPHNB-BYPYZUCNSA-N 0.000 description 2
- DQLHSFUMICQIMB-VIFPVBQESA-N (2s)-2-amino-3-(4-methylphenyl)propanoic acid Chemical compound CC1=CC=C(C[C@H](N)C(O)=O)C=C1 DQLHSFUMICQIMB-VIFPVBQESA-N 0.000 description 2
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 2
- AYQMNFRCBKOMIA-UHFFFAOYSA-N 1-benzazepin-2-one Chemical class O=C1C=CC=C2C=CC=CC2=N1 AYQMNFRCBKOMIA-UHFFFAOYSA-N 0.000 description 2
- OGNSCSPNOLGXSM-UHFFFAOYSA-N 2,4-diaminobutyric acid Chemical compound NCCC(N)C(O)=O OGNSCSPNOLGXSM-UHFFFAOYSA-N 0.000 description 2
- ODHCTXKNWHHXJC-VKHMYHEASA-N 5-oxo-L-proline Chemical compound OC(=O)[C@@H]1CCC(=O)N1 ODHCTXKNWHHXJC-VKHMYHEASA-N 0.000 description 2
- 101100512078 Caenorhabditis elegans lys-1 gene Proteins 0.000 description 2
- 108010076667 Caspases Proteins 0.000 description 2
- 102000011727 Caspases Human genes 0.000 description 2
- 241000239328 Centruroides noxius Species 0.000 description 2
- 241000239282 Centruroides suffusus Species 0.000 description 2
- 241000237970 Conus <genus> Species 0.000 description 2
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 2
- 108010005843 Cysteine Proteases Proteins 0.000 description 2
- 102000005927 Cysteine Proteases Human genes 0.000 description 2
- 241000196324 Embryophyta Species 0.000 description 2
- 241000206602 Eukaryota Species 0.000 description 2
- 108010049003 Fibrinogen Proteins 0.000 description 2
- 102000008946 Fibrinogen Human genes 0.000 description 2
- 102000003688 G-Protein-Coupled Receptors Human genes 0.000 description 2
- 108090000045 G-Protein-Coupled Receptors Proteins 0.000 description 2
- WMBWREPUVVBILR-UHFFFAOYSA-N GCG Natural products C=1C(O)=C(O)C(O)=CC=1C1OC2=CC(O)=CC(O)=C2CC1OC(=O)C1=CC(O)=C(O)C(O)=C1 WMBWREPUVVBILR-UHFFFAOYSA-N 0.000 description 2
- 241000237858 Gastropoda Species 0.000 description 2
- ZRALSGWEFCBTJO-UHFFFAOYSA-N Guanidine Chemical compound NC(N)=N ZRALSGWEFCBTJO-UHFFFAOYSA-N 0.000 description 2
- WZUVPPKBWHMQCE-UHFFFAOYSA-N Haematoxylin Chemical compound C12=CC(O)=C(O)C=C2CC2(O)C1C1=CC=C(O)C(O)=C1OC2 WZUVPPKBWHMQCE-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 241000257303 Hymenoptera Species 0.000 description 2
- 208000035154 Hyperesthesia Diseases 0.000 description 2
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 description 2
- RHGKLRLOHDJJDR-UHFFFAOYSA-N Ndelta-carbamoyl-DL-ornithine Natural products OC(=O)C(N)CCCNC(N)=O RHGKLRLOHDJJDR-UHFFFAOYSA-N 0.000 description 2
- 101100281510 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) met-6 gene Proteins 0.000 description 2
- 108010038807 Oligopeptides Proteins 0.000 description 2
- 102000015636 Oligopeptides Human genes 0.000 description 2
- 101100242312 Oryza sativa subsp. japonica OSK1 gene Proteins 0.000 description 2
- 101100533605 Oryza sativa subsp. japonica SKP1 gene Proteins 0.000 description 2
- 108090000526 Papain Proteins 0.000 description 2
- 101710124951 Phospholipase C Proteins 0.000 description 2
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 2
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 2
- 108091028664 Ribonucleotide Proteins 0.000 description 2
- 241000235088 Saccharomyces sp. Species 0.000 description 2
- 241000270295 Serpentes Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 108090000190 Thrombin Proteins 0.000 description 2
- 101000994422 Tityus discrepans Potassium channel toxin alpha-KTx 15.6 Proteins 0.000 description 2
- 108091023040 Transcription factor Proteins 0.000 description 2
- 102000040945 Transcription factor Human genes 0.000 description 2
- 241000700605 Viruses Species 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-N acetic acid Substances CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 2
- 150000001294 alanine derivatives Chemical class 0.000 description 2
- 150000001345 alkine derivatives Chemical class 0.000 description 2
- 230000000692 anti-sense effect Effects 0.000 description 2
- 239000001961 anticonvulsive agent Substances 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 229940000635 beta-alanine Drugs 0.000 description 2
- 230000000975 bioactive effect Effects 0.000 description 2
- 230000008827 biological function Effects 0.000 description 2
- 239000004202 carbamide Substances 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 230000000747 cardiac effect Effects 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 210000004978 chinese hamster ovary cell Anatomy 0.000 description 2
- 229960002173 citrulline Drugs 0.000 description 2
- 235000013477 citrulline Nutrition 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 238000004891 communication Methods 0.000 description 2
- 108050003126 conotoxin Proteins 0.000 description 2
- 239000000356 contaminant Substances 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 230000006735 deficit Effects 0.000 description 2
- YSMODUONRAFBET-UHFFFAOYSA-N delta-DL-hydroxylysine Natural products NCC(O)CCC(N)C(O)=O YSMODUONRAFBET-UHFFFAOYSA-N 0.000 description 2
- 238000001212 derivatisation Methods 0.000 description 2
- VEVRNHHLCPGNDU-MUGJNUQGSA-O desmosine Chemical compound OC(=O)[C@@H](N)CCCC[N+]1=CC(CC[C@H](N)C(O)=O)=C(CCC[C@H](N)C(O)=O)C(CC[C@H](N)C(O)=O)=C1 VEVRNHHLCPGNDU-MUGJNUQGSA-O 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 125000002228 disulfide group Chemical group 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 229940030275 epigallocatechin gallate Drugs 0.000 description 2
- YSMODUONRAFBET-UHNVWZDZSA-N erythro-5-hydroxy-L-lysine Chemical compound NC[C@H](O)CC[C@H](N)C(O)=O YSMODUONRAFBET-UHNVWZDZSA-N 0.000 description 2
- 229940012952 fibrinogen Drugs 0.000 description 2
- 230000002538 fungal effect Effects 0.000 description 2
- 235000013922 glutamic acid Nutrition 0.000 description 2
- 239000004220 glutamic acid Substances 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- 238000009396 hybridization Methods 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 230000000977 initiatory effect Effects 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 231100000518 lethal Toxicity 0.000 description 2
- 230000001665 lethal effect Effects 0.000 description 2
- 229960004194 lidocaine Drugs 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- 230000004807 localization Effects 0.000 description 2
- 210000001161 mammalian embryo Anatomy 0.000 description 2
- 238000013507 mapping Methods 0.000 description 2
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- 230000000813 microbial effect Effects 0.000 description 2
- 238000010369 molecular cloning Methods 0.000 description 2
- 238000002703 mutagenesis Methods 0.000 description 2
- 231100000350 mutagenesis Toxicity 0.000 description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N n-hexanoic acid Natural products CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 2
- 239000006225 natural substrate Substances 0.000 description 2
- 230000004770 neurodegeneration Effects 0.000 description 2
- 210000000929 nociceptor Anatomy 0.000 description 2
- 108091008700 nociceptors Proteins 0.000 description 2
- 210000004940 nucleus Anatomy 0.000 description 2
- 230000008058 pain sensation Effects 0.000 description 2
- 229940055729 papain Drugs 0.000 description 2
- 235000019834 papain Nutrition 0.000 description 2
- 230000006320 pegylation Effects 0.000 description 2
- 210000002856 peripheral neuron Anatomy 0.000 description 2
- 230000001766 physiological effect Effects 0.000 description 2
- 239000013612 plasmid Substances 0.000 description 2
- 230000008488 polyadenylation Effects 0.000 description 2
- 230000004481 post-translational protein modification Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 239000002336 ribonucleotide Substances 0.000 description 2
- 125000002652 ribonucleotide group Chemical group 0.000 description 2
- FSYKKLYZXJSNPZ-UHFFFAOYSA-N sarcosine Chemical compound C[NH2+]CC([O-])=O FSYKKLYZXJSNPZ-UHFFFAOYSA-N 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 210000000413 sensory ganglia Anatomy 0.000 description 2
- 150000003384 small molecules Chemical class 0.000 description 2
- 208000020431 spinal cord injury Diseases 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- 230000009885 systemic effect Effects 0.000 description 2
- 230000008685 targeting Effects 0.000 description 2
- RAOIDOHSFRTOEL-UHFFFAOYSA-N tetrahydrothiophene Chemical group C1CCSC1 RAOIDOHSFRTOEL-UHFFFAOYSA-N 0.000 description 2
- 230000000542 thalamic effect Effects 0.000 description 2
- 229960004072 thrombin Drugs 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000005030 transcription termination Effects 0.000 description 2
- 238000010361 transduction Methods 0.000 description 2
- 230000026683 transduction Effects 0.000 description 2
- 230000001131 transforming effect Effects 0.000 description 2
- 210000000427 trigeminal ganglion Anatomy 0.000 description 2
- 229960002811 ziconotide Drugs 0.000 description 2
- 108091058549 μ-conotoxin Proteins 0.000 description 2
- PFWOJTLVNOZSGQ-RIMJLNMDSA-N (1R,2aS,4S,7S,10S,13S,19S,22S,25S,28S,31R,36R,39S,45R,48S,51S,54S,57R,60S,63S,66S,69S,72S,75S,78S,81S,84S,87S,90R,93S,96S,99S)-10,51,87-tris(4-aminobutyl)-31-[[(2S)-2-[[(2S)-2-amino-5-carbamimidamidopentanoyl]amino]-3-hydroxypropanoyl]amino]-75-(aminomethyl)-93-(3-amino-3-oxopropyl)-60,96-dibenzyl-19,28-bis[(2S)-butan-2-yl]-4,54,69-tris(3-carbamimidamidopropyl)-25-(carboxymethyl)-2a,22,39,48-tetrakis[(1R)-1-hydroxyethyl]-7,63,81-tris(hydroxymethyl)-72-[(4-hydroxyphenyl)methyl]-84-(1H-imidazol-5-ylmethyl)-99-methyl-66-(2-methylpropyl)-78-(2-methylsulfanylethyl)-1a,3,4a,6,9,12,18,21,24,27,30,38,41,44,47,50,53,56,59,62,65,68,71,74,77,80,83,86,89,92,95,98-dotriacontaoxo-6a,7a,10a,11a,33,34-hexathia-a,2,3a,5,8,11,17,20,23,26,29,37,40,43,46,49,52,55,58,61,64,67,70,73,76,79,82,85,88,91,94,97-dotriacontazatetracyclo[55.47.4.445,90.013,17]dodecahectane-36-carboxylic acid Chemical compound CC[C@H](C)[C@@H]1NC(=O)[C@H](CSSC[C@H](NC(=O)[C@@H](NC(=O)CNC(=O)[C@@H]2CSSC[C@@H]3NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](Cc4ccccc4)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](Cc4ccccc4)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc4ccc(O)cc4)NC(=O)[C@H](CN)NC(=O)[C@H](CCSC)NC(=O)[C@H](CO)NC(=O)[C@H](Cc4cnc[nH]4)NC(=O)[C@H](CCCCN)NC3=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(=O)N2)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC1=O)[C@@H](C)O)[C@@H](C)CC)[C@@H](C)O)[C@@H](C)O)C(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CCCNC(N)=N PFWOJTLVNOZSGQ-RIMJLNMDSA-N 0.000 description 1
- GMKMEZVLHJARHF-UHFFFAOYSA-N (2R,6R)-form-2.6-Diaminoheptanedioic acid Natural products OC(=O)C(N)CCCC(N)C(O)=O GMKMEZVLHJARHF-UHFFFAOYSA-N 0.000 description 1
- XXGPNEDZNRZWPK-QMMMGPOBSA-N (2s)-2-(carboxyamino)-3-phenylpropanoic acid Chemical compound OC(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 XXGPNEDZNRZWPK-QMMMGPOBSA-N 0.000 description 1
- HUOYGRNYKOUAFE-QMMMGPOBSA-N (2s)-2-(iodoamino)-3-phenylpropanoic acid Chemical compound OC(=O)[C@@H](NI)CC1=CC=CC=C1 HUOYGRNYKOUAFE-QMMMGPOBSA-N 0.000 description 1
- WRQSUCJAKAMYMQ-YFKPBYRVSA-N (2s)-2-(thiophen-3-ylamino)propanoic acid Chemical compound OC(=O)[C@H](C)NC=1C=CSC=1 WRQSUCJAKAMYMQ-YFKPBYRVSA-N 0.000 description 1
- NPDBDJFLKKQMCM-SCSAIBSYSA-N (2s)-2-amino-3,3-dimethylbutanoic acid Chemical compound CC(C)(C)[C@H](N)C(O)=O NPDBDJFLKKQMCM-SCSAIBSYSA-N 0.000 description 1
- PECGVEGMRUZOML-AWEZNQCLSA-N (2s)-2-amino-3,3-diphenylpropanoic acid Chemical compound C=1C=CC=CC=1C([C@H](N)C(O)=O)C1=CC=CC=C1 PECGVEGMRUZOML-AWEZNQCLSA-N 0.000 description 1
- CSJZKSXYLTYFPU-NSHDSACASA-N (2s)-2-amino-3-(4-tert-butylphenyl)propanoic acid Chemical compound CC(C)(C)C1=CC=C(C[C@H](N)C(O)=O)C=C1 CSJZKSXYLTYFPU-NSHDSACASA-N 0.000 description 1
- FYMNTAQFDTZISY-QMMMGPOBSA-N (2s)-2-amino-3-[4-(diaminomethylideneamino)phenyl]propanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N=C(N)N)C=C1 FYMNTAQFDTZISY-QMMMGPOBSA-N 0.000 description 1
- PDRJLZDUOULRHE-ZETCQYMHSA-N (2s)-2-amino-3-pyridin-2-ylpropanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=N1 PDRJLZDUOULRHE-ZETCQYMHSA-N 0.000 description 1
- VWTFNYVAFGYEKI-QMMMGPOBSA-N (2s)-2-azaniumyl-3-(3,4-dimethoxyphenyl)propanoate Chemical compound COC1=CC=C(C[C@H](N)C(O)=O)C=C1OC VWTFNYVAFGYEKI-QMMMGPOBSA-N 0.000 description 1
- FMUMEWVNYMUECA-LURJTMIESA-N (2s)-2-azaniumyl-5-methylhexanoate Chemical compound CC(C)CC[C@H](N)C(O)=O FMUMEWVNYMUECA-LURJTMIESA-N 0.000 description 1
- OPOTYPXOKUZEKY-QMMMGPOBSA-N (2s)-2-nitramido-3-phenylpropanoic acid Chemical compound [O-][N+](=O)N[C@H](C(=O)O)CC1=CC=CC=C1 OPOTYPXOKUZEKY-QMMMGPOBSA-N 0.000 description 1
- RYAUUSMBKDBGNL-BKLSDQPFSA-N (2s)-4-(diaminomethylideneamino)pyrrolidine-2-carboxylic acid Chemical compound NC(=N)NC1CN[C@H](C(O)=O)C1 RYAUUSMBKDBGNL-BKLSDQPFSA-N 0.000 description 1
- PJDINCOFOROBQW-LURJTMIESA-N (3S)-3,7-diaminoheptanoic acid Chemical compound NCCCC[C@H](N)CC(O)=O PJDINCOFOROBQW-LURJTMIESA-N 0.000 description 1
- DUVVFMLAHWNDJD-VIFPVBQESA-N (3S)-3-Amino-4-(1H-indol-3-yl)butanoic acid Chemical compound C1=CC=C2C(C[C@@H](CC(O)=O)N)=CNC2=C1 DUVVFMLAHWNDJD-VIFPVBQESA-N 0.000 description 1
- OFVBLKINTLPEGH-VIFPVBQESA-N (3S)-3-Amino-4-phenylbutanoic acid Chemical compound OC(=O)C[C@@H](N)CC1=CC=CC=C1 OFVBLKINTLPEGH-VIFPVBQESA-N 0.000 description 1
- VNWXCGKMEWXYBP-YFKPBYRVSA-N (3s)-3-amino-6-(diaminomethylideneamino)hexanoic acid Chemical compound OC(=O)C[C@@H](N)CCCNC(N)=N VNWXCGKMEWXYBP-YFKPBYRVSA-N 0.000 description 1
- UKRWCEJTOPSTEC-RDTXWAMCSA-N (6ar,9r)-4-acetyl-n-ethyl-7-methyl-6,6a,8,9-tetrahydroindolo[4,3-fg]quinoline-9-carboxamide Chemical compound C1=CC(C=2[C@H](N(C)C[C@@H](C=2)C(=O)NCC)C2)=C3C2=CN(C(C)=O)C3=C1 UKRWCEJTOPSTEC-RDTXWAMCSA-N 0.000 description 1
- UJOYFRCOTPUKAK-MRVPVSSYSA-N (R)-3-ammonio-3-phenylpropanoate Chemical compound OC(=O)C[C@@H](N)C1=CC=CC=C1 UJOYFRCOTPUKAK-MRVPVSSYSA-N 0.000 description 1
- XJLSEXAGTJCILF-RXMQYKEDSA-N (R)-nipecotic acid zwitterion Chemical compound OC(=O)[C@@H]1CCCNC1 XJLSEXAGTJCILF-RXMQYKEDSA-N 0.000 description 1
- VWTFNYVAFGYEKI-UHFFFAOYSA-N (S)-3,4-dimethoxyphenylalanine Natural products COC1=CC=C(CC(N)C(O)=O)C=C1OC VWTFNYVAFGYEKI-UHFFFAOYSA-N 0.000 description 1
- FTSVTCPUMUXYJS-UHFFFAOYSA-N 1,2,3,4-tetrahydroisoquinoline Chemical compound C1=C=C[C]2CNCCC2=C1 FTSVTCPUMUXYJS-UHFFFAOYSA-N 0.000 description 1
- LEBVLXFERQHONN-UHFFFAOYSA-N 1-butyl-N-(2,6-dimethylphenyl)piperidine-2-carboxamide Chemical compound CCCCN1CCCCC1C(=O)NC1=C(C)C=CC=C1C LEBVLXFERQHONN-UHFFFAOYSA-N 0.000 description 1
- MKFDYOISVJDNKS-UHFFFAOYSA-N 2-(2,3-dihydro-1h-inden-1-ylamino)acetic acid Chemical compound C1=CC=C2C(NCC(=O)O)CCC2=C1 MKFDYOISVJDNKS-UHFFFAOYSA-N 0.000 description 1
- RIJNIVWHYSNSLQ-UHFFFAOYSA-N 2-(2,3-dihydro-1h-inden-2-ylazaniumyl)acetate Chemical compound C1=CC=C2CC(NCC(=O)O)CC2=C1 RIJNIVWHYSNSLQ-UHFFFAOYSA-N 0.000 description 1
- CVZZNRXMDCOHBG-QMMMGPOBSA-N 2-Chloro-L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1Cl CVZZNRXMDCOHBG-QMMMGPOBSA-N 0.000 description 1
- XJFPXLWGZWAWRQ-UHFFFAOYSA-N 2-[[2-[[2-[[2-[[2-[(2-azaniumylacetyl)amino]acetyl]amino]acetyl]amino]acetyl]amino]acetyl]amino]acetate Chemical compound NCC(=O)NCC(=O)NCC(=O)NCC(=O)NCC(=O)NCC(O)=O XJFPXLWGZWAWRQ-UHFFFAOYSA-N 0.000 description 1
- XNBJHKABANTVCP-UHFFFAOYSA-N 2-amino-3-(diaminomethylideneamino)propanoic acid Chemical compound OC(=O)C(N)CN=C(N)N XNBJHKABANTVCP-UHFFFAOYSA-N 0.000 description 1
- RDFMDVXONNIGBC-UHFFFAOYSA-N 2-aminoheptanoic acid Chemical compound CCCCCC(N)C(O)=O RDFMDVXONNIGBC-UHFFFAOYSA-N 0.000 description 1
- LRQKBLKVPFOOQJ-UHFFFAOYSA-N 2-aminohexanoic acid Chemical compound CCCCC(N)C(O)=O LRQKBLKVPFOOQJ-UHFFFAOYSA-N 0.000 description 1
- JUQLUIFNNFIIKC-UHFFFAOYSA-N 2-aminopimelic acid Chemical compound OC(=O)C(N)CCCCC(O)=O JUQLUIFNNFIIKC-UHFFFAOYSA-N 0.000 description 1
- OFYAYGJCPXRNBL-UHFFFAOYSA-N 2-azaniumyl-3-naphthalen-1-ylpropanoate Chemical compound C1=CC=C2C(CC(N)C(O)=O)=CC=CC2=C1 OFYAYGJCPXRNBL-UHFFFAOYSA-N 0.000 description 1
- 125000004042 4-aminobutyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])N([H])[H] 0.000 description 1
- XWHHYOYVRVGJJY-QMMMGPOBSA-N 4-fluoro-L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(F)C=C1 XWHHYOYVRVGJJY-QMMMGPOBSA-N 0.000 description 1
- PZNQZSRPDOEBMS-QMMMGPOBSA-N 4-iodo-L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(I)C=C1 PZNQZSRPDOEBMS-QMMMGPOBSA-N 0.000 description 1
- AGFIRQJZCNVMCW-UAKXSSHOSA-N 5-bromouridine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(Br)=C1 AGFIRQJZCNVMCW-UAKXSSHOSA-N 0.000 description 1
- 229940117976 5-hydroxylysine Drugs 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 101710113182 Alpha-toxin OD1 Proteins 0.000 description 1
- 101001023095 Anemonia sulcata Delta-actitoxin-Avd1a Proteins 0.000 description 1
- 108020000948 Antisense Oligonucleotides Proteins 0.000 description 1
- 101000641989 Araneus ventricosus Kunitz-type U1-aranetoxin-Av1a Proteins 0.000 description 1
- 102000004580 Aspartic Acid Proteases Human genes 0.000 description 1
- 108010017640 Aspartic Acid Proteases Proteins 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 101800001415 Bri23 peptide Proteins 0.000 description 1
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 1
- 101800000655 C-terminal peptide Proteins 0.000 description 1
- 102400000107 C-terminal peptide Human genes 0.000 description 1
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 1
- 101100228206 Caenorhabditis elegans gly-6 gene Proteins 0.000 description 1
- 101100285688 Caenorhabditis elegans hrg-7 gene Proteins 0.000 description 1
- 101100455752 Caenorhabditis elegans lys-3 gene Proteins 0.000 description 1
- 101100289888 Caenorhabditis elegans lys-5 gene Proteins 0.000 description 1
- 101100315624 Caenorhabditis elegans tyr-1 gene Proteins 0.000 description 1
- 108090000312 Calcium Channels Proteins 0.000 description 1
- 102000003922 Calcium Channels Human genes 0.000 description 1
- 102000005702 Calcium-Activated Potassium Channels Human genes 0.000 description 1
- 108010045489 Calcium-Activated Potassium Channels Proteins 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-N Caprylic acid Natural products CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 1
- 101001028691 Carybdea rastonii Toxin CrTX-A Proteins 0.000 description 1
- 101000685083 Centruroides infamatus Beta-toxin Cii1 Proteins 0.000 description 1
- 101000685085 Centruroides noxius Toxin Cn1 Proteins 0.000 description 1
- 206010008025 Cerebellar ataxia Diseases 0.000 description 1
- 101710091342 Chemotactic peptide Proteins 0.000 description 1
- 101001028688 Chironex fleckeri Toxin CfTX-1 Proteins 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 241000237976 Conus striatus Species 0.000 description 1
- 101000940362 Cyriopagopus schmidti Huwentoxin-IV Proteins 0.000 description 1
- 101000644407 Cyriopagopus schmidti U6-theraphotoxin-Hs1a Proteins 0.000 description 1
- FDKWRPBBCBCIGA-UWTATZPHSA-N D-Selenocysteine Natural products [Se]C[C@@H](N)C(O)=O FDKWRPBBCBCIGA-UWTATZPHSA-N 0.000 description 1
- 150000008574 D-amino acids Chemical class 0.000 description 1
- ODKSFYDXXFIFQN-SCSAIBSYSA-N D-arginine Chemical compound OC(=O)[C@H](N)CCCNC(N)=N ODKSFYDXXFIFQN-SCSAIBSYSA-N 0.000 description 1
- 229930028154 D-arginine Natural products 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- SNPQGCDJHZAVOB-SOMABOLJSA-N D-glucosyl-N-tetracosanoylsphinganine Chemical compound CCCCCCCCCCCCCCCCCCCCCCCC(=O)N[C@H]([C@H](O)CCCCCCCCCCCCCCC)COC1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O SNPQGCDJHZAVOB-SOMABOLJSA-N 0.000 description 1
- HMFHBZSHGGEWLO-SOOFDHNKSA-N D-ribofuranose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H]1O HMFHBZSHGGEWLO-SOOFDHNKSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 101100136092 Drosophila melanogaster peng gene Proteins 0.000 description 1
- 102100038796 E3 ubiquitin-protein ligase TRIM13 Human genes 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- 108010091443 Exopeptidases Proteins 0.000 description 1
- 102000018389 Exopeptidases Human genes 0.000 description 1
- 108091006020 Fc-tagged proteins Proteins 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- 101000664589 Homo sapiens E3 ubiquitin-protein ligase TRIM13 Proteins 0.000 description 1
- 101001123834 Homo sapiens Neprilysin Proteins 0.000 description 1
- 101001010792 Homo sapiens Transcriptional regulator ERG Proteins 0.000 description 1
- 101000867850 Homo sapiens Voltage-dependent T-type calcium channel subunit alpha-1G Proteins 0.000 description 1
- YZJSUQQZGCHHNQ-UHFFFAOYSA-N Homoglutamine Chemical compound OC(=O)C(N)CCCC(N)=O YZJSUQQZGCHHNQ-UHFFFAOYSA-N 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- LCWXJXMHJVIJFK-UHFFFAOYSA-N Hydroxylysine Natural products NCC(O)CC(N)CC(O)=O LCWXJXMHJVIJFK-UHFFFAOYSA-N 0.000 description 1
- 208000007599 Hyperkalemic periodic paralysis Diseases 0.000 description 1
- XQFRJNBWHJMXHO-RRKCRQDMSA-N IDUR Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(I)=C1 XQFRJNBWHJMXHO-RRKCRQDMSA-N 0.000 description 1
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 1
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 1
- 102000013463 Immunoglobulin Light Chains Human genes 0.000 description 1
- 108010065825 Immunoglobulin Light Chains Proteins 0.000 description 1
- UGQMRVRMYYASKQ-KQYNXXCUSA-N Inosine Chemical class O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=NC=NC(O)=C2N=C1 UGQMRVRMYYASKQ-KQYNXXCUSA-N 0.000 description 1
- 208000004404 Intractable Pain Diseases 0.000 description 1
- 108091092195 Intron Proteins 0.000 description 1
- 108060005987 Kallikrein Proteins 0.000 description 1
- 102000001399 Kallikrein Human genes 0.000 description 1
- AGPKZVBTJJNPAG-UHNVWZDZSA-N L-allo-Isoleucine Chemical compound CC[C@@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-UHNVWZDZSA-N 0.000 description 1
- 150000008575 L-amino acids Chemical class 0.000 description 1
- 229930064664 L-arginine Natural products 0.000 description 1
- 235000014852 L-arginine Nutrition 0.000 description 1
- XIGSAGMEBXLVJJ-YFKPBYRVSA-N L-homocitrulline Chemical compound NC(=O)NCCCC[C@H]([NH3+])C([O-])=O XIGSAGMEBXLVJJ-YFKPBYRVSA-N 0.000 description 1
- FFFHZYDWPBMWHY-VKHMYHEASA-N L-homocysteine Chemical compound OC(=O)[C@@H](N)CCS FFFHZYDWPBMWHY-VKHMYHEASA-N 0.000 description 1
- JTTHKOPSMAVJFE-VIFPVBQESA-N L-homophenylalanine Chemical compound OC(=O)[C@@H](N)CCC1=CC=CC=C1 JTTHKOPSMAVJFE-VIFPVBQESA-N 0.000 description 1
- HXEACLLIILLPRG-YFKPBYRVSA-N L-pipecolic acid Chemical compound [O-]C(=O)[C@@H]1CCCC[NH2+]1 HXEACLLIILLPRG-YFKPBYRVSA-N 0.000 description 1
- ZKZBPNGNEQAJSX-REOHCLBHSA-N L-selenocysteine Chemical compound [SeH]C[C@H](N)C(O)=O ZKZBPNGNEQAJSX-REOHCLBHSA-N 0.000 description 1
- 101150118523 LYS4 gene Proteins 0.000 description 1
- 241000239268 Leiurus quinquestriatus Species 0.000 description 1
- 102000018697 Membrane Proteins Human genes 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 208000016285 Movement disease Diseases 0.000 description 1
- 101100083427 Mus musculus Pithd1 gene Proteins 0.000 description 1
- 101100084404 Mus musculus Prodh gene Proteins 0.000 description 1
- 101100260702 Mus musculus Tinagl1 gene Proteins 0.000 description 1
- 101100208034 Mus musculus Trpv4 gene Proteins 0.000 description 1
- 206010061533 Myotonia Diseases 0.000 description 1
- CZCIKBSVHDNIDH-NSHDSACASA-N N(alpha)-methyl-L-tryptophan Chemical compound C1=CC=C2C(C[C@H]([NH2+]C)C([O-])=O)=CNC2=C1 CZCIKBSVHDNIDH-NSHDSACASA-N 0.000 description 1
- CHJJGSNFBQVOTG-UHFFFAOYSA-N N-methyl-guanidine Natural products CNC(N)=N CHJJGSNFBQVOTG-UHFFFAOYSA-N 0.000 description 1
- 229940124634 N-type calcium channel blocker Drugs 0.000 description 1
- CZCIKBSVHDNIDH-UHFFFAOYSA-N Nalpha-methyl-DL-tryptophan Natural products C1=CC=C2C(CC(NC)C(O)=O)=CNC2=C1 CZCIKBSVHDNIDH-UHFFFAOYSA-N 0.000 description 1
- 102100028782 Neprilysin Human genes 0.000 description 1
- 208000028389 Nerve injury Diseases 0.000 description 1
- 108090000189 Neuropeptides Proteins 0.000 description 1
- 101100109292 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) arg-13 gene Proteins 0.000 description 1
- 101100404023 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) arg-14 gene Proteins 0.000 description 1
- 101100491597 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) arg-6 gene Proteins 0.000 description 1
- 101100109397 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) arg-8 gene Proteins 0.000 description 1
- 101100426732 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) cys-9 gene Proteins 0.000 description 1
- 101100205180 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) leu-6 gene Proteins 0.000 description 1
- 101100426589 Neurospora crassa (strain ATCC 24698 / 74-OR23-1A / CBS 708.71 / DSM 1257 / FGSC 987) trp-3 gene Proteins 0.000 description 1
- 102000019315 Nicotinic acetylcholine receptors Human genes 0.000 description 1
- 108050006807 Nicotinic acetylcholine receptors Proteins 0.000 description 1
- GEYBMYRBIABFTA-VIFPVBQESA-N O-methyl-L-tyrosine Chemical compound COC1=CC=C(C[C@H](N)C(O)=O)C=C1 GEYBMYRBIABFTA-VIFPVBQESA-N 0.000 description 1
- 108010067372 Pancreatic elastase Proteins 0.000 description 1
- 102000016387 Pancreatic elastase Human genes 0.000 description 1
- 108010067902 Peptide Library Proteins 0.000 description 1
- 229940083963 Peptide antagonist Drugs 0.000 description 1
- 108091093037 Peptide nucleic acid Proteins 0.000 description 1
- 101000679608 Phaeosphaeria nodorum (strain SN15 / ATCC MYA-4574 / FGSC 10173) Cysteine rich necrotrophic effector Tox1 Proteins 0.000 description 1
- CXOFVDLJLONNDW-UHFFFAOYSA-N Phenytoin Chemical compound N1C(=O)NC(=O)C1(C=1C=CC=CC=1)C1=CC=CC=C1 CXOFVDLJLONNDW-UHFFFAOYSA-N 0.000 description 1
- 208000025844 Prostatic disease Diseases 0.000 description 1
- ODHCTXKNWHHXJC-GSVOUGTGSA-N Pyroglutamic acid Natural products OC(=O)[C@H]1CCC(=O)N1 ODHCTXKNWHHXJC-GSVOUGTGSA-N 0.000 description 1
- PYMYPHUHKUWMLA-LMVFSUKVSA-N Ribose Natural products OC[C@@H](O)[C@@H](O)[C@@H](O)C=O PYMYPHUHKUWMLA-LMVFSUKVSA-N 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 108010077895 Sarcosine Proteins 0.000 description 1
- 206010070834 Sensitisation Diseases 0.000 description 1
- FKNQFGJONOIPTF-UHFFFAOYSA-N Sodium cation Chemical compound [Na+] FKNQFGJONOIPTF-UHFFFAOYSA-N 0.000 description 1
- 229940089973 Sodium channel antagonist Drugs 0.000 description 1
- 101710097939 Sodium channel protein type 9 subunit alpha Proteins 0.000 description 1
- 241000242730 Stichodactyla helianthus Species 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 102000003691 T-Type Calcium Channels Human genes 0.000 description 1
- 108090000030 T-Type Calcium Channels Proteins 0.000 description 1
- 102000003629 TRPC3 Human genes 0.000 description 1
- 102000003622 TRPC4 Human genes 0.000 description 1
- 229920002253 Tannate Polymers 0.000 description 1
- WDLRUFUQRNWCPK-UHFFFAOYSA-N Tetraxetan Chemical compound OC(=O)CN1CCN(CC(O)=O)CCN(CC(O)=O)CCN(CC(O)=O)CC1 WDLRUFUQRNWCPK-UHFFFAOYSA-N 0.000 description 1
- 241001572088 Thrixopelma pruriens Species 0.000 description 1
- 101000844260 Thrixopelma pruriens Beta/omega-theraphotoxin-Tp1a Proteins 0.000 description 1
- 102000009190 Transthyretin Human genes 0.000 description 1
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- 229930003756 Vitamin B7 Natural products 0.000 description 1
- 102000003734 Voltage-Gated Potassium Channels Human genes 0.000 description 1
- 108090000013 Voltage-Gated Potassium Channels Proteins 0.000 description 1
- 241000269368 Xenopus laevis Species 0.000 description 1
- 230000001594 aberrant effect Effects 0.000 description 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- ODHCTXKNWHHXJC-UHFFFAOYSA-N acide pyroglutamique Natural products OC(=O)C1CCC(=O)N1 ODHCTXKNWHHXJC-UHFFFAOYSA-N 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 210000001943 adrenal medulla Anatomy 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- HMFHBZSHGGEWLO-UHFFFAOYSA-N alpha-D-Furanose-Ribose Natural products OCC1OC(O)C(O)C1O HMFHBZSHGGEWLO-UHFFFAOYSA-N 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 150000001412 amines Chemical class 0.000 description 1
- 125000002344 aminooxy group Chemical group [H]N([H])O[*] 0.000 description 1
- 210000003484 anatomy Anatomy 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 231100000659 animal toxin Toxicity 0.000 description 1
- 230000003042 antagnostic effect Effects 0.000 description 1
- 230000003502 anti-nociceptive effect Effects 0.000 description 1
- 229940125681 anticonvulsant agent Drugs 0.000 description 1
- 230000000890 antigenic effect Effects 0.000 description 1
- 239000000074 antisense oligonucleotide Substances 0.000 description 1
- 238000012230 antisense oligonucleotides Methods 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 101150088826 arg1 gene Proteins 0.000 description 1
- 238000003491 array Methods 0.000 description 1
- 230000006793 arrhythmia Effects 0.000 description 1
- 206010003119 arrhythmia Diseases 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-L aspartate group Chemical group N[C@@H](CC(=O)[O-])C(=O)[O-] CKLJMWTZIZZHCS-REOHCLBHSA-L 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 210000003050 axon Anatomy 0.000 description 1
- 150000001540 azides Chemical class 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- SRSXLGNVWSONIS-UHFFFAOYSA-M benzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=CC=C1 SRSXLGNVWSONIS-UHFFFAOYSA-M 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- GONOPSZTUGRENK-UHFFFAOYSA-N benzyl(trichloro)silane Chemical compound Cl[Si](Cl)(Cl)CC1=CC=CC=C1 GONOPSZTUGRENK-UHFFFAOYSA-N 0.000 description 1
- ADSALMJPJUKESW-UHFFFAOYSA-N beta-Homoproline Chemical compound OC(=O)CC1CCCN1 ADSALMJPJUKESW-UHFFFAOYSA-N 0.000 description 1
- 150000001576 beta-amino acids Chemical class 0.000 description 1
- 238000013378 biophysical characterization Methods 0.000 description 1
- 229920001222 biopolymer Polymers 0.000 description 1
- 108091006004 biotinylated proteins Proteins 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 229960003150 bupivacaine Drugs 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- FFGPTBGBLSHEPO-UHFFFAOYSA-N carbamazepine Chemical compound C1=CC2=CC=CC=C2N(C(=O)N)C2=CC=CC=C21 FFGPTBGBLSHEPO-UHFFFAOYSA-N 0.000 description 1
- 229960000623 carbamazepine Drugs 0.000 description 1
- 125000001951 carbamoylamino group Chemical group C(N)(=O)N* 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- UHBYWPGGCSDKFX-UHFFFAOYSA-N carboxyglutamic acid Chemical compound OC(=O)C(N)CC(C(O)=O)C(O)=O UHBYWPGGCSDKFX-UHFFFAOYSA-N 0.000 description 1
- 210000005056 cell body Anatomy 0.000 description 1
- 210000001638 cerebellum Anatomy 0.000 description 1
- 210000003710 cerebral cortex Anatomy 0.000 description 1
- PRQROPMIIGLWRP-BZSNNMDCSA-N chemotactic peptide Chemical compound CSCC[C@H](NC=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 PRQROPMIIGLWRP-BZSNNMDCSA-N 0.000 description 1
- 210000003763 chloroplast Anatomy 0.000 description 1
- BHYOQNUELFTYRT-DPAQBDIFSA-N cholesterol sulfate Chemical class C1C=C2C[C@@H](OS(O)(=O)=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 BHYOQNUELFTYRT-DPAQBDIFSA-N 0.000 description 1
- 229940080277 cholesteryl sulfate Drugs 0.000 description 1
- 239000013611 chromosomal DNA Substances 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 210000000349 chromosome Anatomy 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000022371 chronic pain syndrome Diseases 0.000 description 1
- 208000018912 cluster headache syndrome Diseases 0.000 description 1
- 239000005515 coenzyme Substances 0.000 description 1
- 230000000295 complement effect Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 239000000039 congener Substances 0.000 description 1
- 230000001268 conjugating effect Effects 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000000875 corresponding effect Effects 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 210000004395 cytoplasmic granule Anatomy 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 238000000502 dialysis Methods 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- PGUYAANYCROBRT-UHFFFAOYSA-N dihydroxy-selanyl-selanylidene-lambda5-phosphane Chemical compound OP(O)([SeH])=[Se] PGUYAANYCROBRT-UHFFFAOYSA-N 0.000 description 1
- YGXMYKTZDMYISM-IDIVVRGQSA-J dimagnesium;[[[(2r,3s,4r,5r)-5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-oxidophosphoryl]oxy-oxidophosphoryl] phosphate Chemical compound [Mg+2].[Mg+2].C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O YGXMYKTZDMYISM-IDIVVRGQSA-J 0.000 description 1
- SWSQBOPZIKWTGO-UHFFFAOYSA-N dimethylaminoamidine Natural products CN(C)C(N)=N SWSQBOPZIKWTGO-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- NAGJZTKCGNOGPW-UHFFFAOYSA-K dioxido-sulfanylidene-sulfido-$l^{5}-phosphane Chemical compound [O-]P([O-])([S-])=S NAGJZTKCGNOGPW-UHFFFAOYSA-K 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 150000003959 diselenides Chemical group 0.000 description 1
- 238000010494 dissociation reaction Methods 0.000 description 1
- 230000005593 dissociations Effects 0.000 description 1
- 238000009826 distribution Methods 0.000 description 1
- 230000006334 disulfide bridging Effects 0.000 description 1
- 238000009510 drug design Methods 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 229940066758 endopeptidases Drugs 0.000 description 1
- 210000002472 endoplasmic reticulum Anatomy 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 210000003059 ependyma Anatomy 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 229940093476 ethylene glycol Drugs 0.000 description 1
- 210000003527 eukaryotic cell Anatomy 0.000 description 1
- 230000000763 evoking effect Effects 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 230000028023 exocytosis Effects 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000012634 fragment Substances 0.000 description 1
- 229940074391 gallic acid Drugs 0.000 description 1
- 235000004515 gallic acid Nutrition 0.000 description 1
- BTCSSZJGUNDROE-UHFFFAOYSA-N gamma-aminobutyric acid Chemical compound NCCCC(O)=O BTCSSZJGUNDROE-UHFFFAOYSA-N 0.000 description 1
- 238000012224 gene deletion Methods 0.000 description 1
- 108091006104 gene-regulatory proteins Proteins 0.000 description 1
- 102000034356 gene-regulatory proteins Human genes 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 230000004110 gluconeogenesis Effects 0.000 description 1
- 229960002743 glutamine Drugs 0.000 description 1
- 125000003630 glycyl group Chemical group [H]N([H])C([H])([H])C(*)=O 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 229960004198 guanidine Drugs 0.000 description 1
- 229960000789 guanidine hydrochloride Drugs 0.000 description 1
- PJJJBBJSCAKJQF-UHFFFAOYSA-N guanidinium chloride Chemical compound [Cl-].NC(N)=[NH2+] PJJJBBJSCAKJQF-UHFFFAOYSA-N 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 210000004295 hippocampal neuron Anatomy 0.000 description 1
- 210000001320 hippocampus Anatomy 0.000 description 1
- 230000006801 homologous recombination Effects 0.000 description 1
- 238000002744 homologous recombination Methods 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- QJHBJHUKURJDLG-UHFFFAOYSA-N hydroxy-L-lysine Natural products NCCCCC(NO)C(O)=O QJHBJHUKURJDLG-UHFFFAOYSA-N 0.000 description 1
- 230000002267 hypothalamic effect Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 238000007901 in situ hybridization Methods 0.000 description 1
- QNRXNRGSOJZINA-UHFFFAOYSA-N indoline-2-carboxylic acid Chemical compound C1=CC=C2NC(C(=O)O)CC2=C1 QNRXNRGSOJZINA-UHFFFAOYSA-N 0.000 description 1
- 239000003999 initiator Substances 0.000 description 1
- 230000010354 integration Effects 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000002780 ion channel assay Methods 0.000 description 1
- 231100000666 ion channel toxin Toxicity 0.000 description 1
- 230000002427 irreversible effect Effects 0.000 description 1
- 238000010829 isocratic elution Methods 0.000 description 1
- RGXCTRIQQODGIZ-UHFFFAOYSA-O isodesmosine Chemical compound OC(=O)C(N)CCCC[N+]1=CC(CCC(N)C(O)=O)=CC(CCC(N)C(O)=O)=C1CCCC(N)C(O)=O RGXCTRIQQODGIZ-UHFFFAOYSA-O 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 230000000155 isotopic effect Effects 0.000 description 1
- 125000000468 ketone group Chemical group 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 101150036274 kil gene Proteins 0.000 description 1
- 238000011813 knockout mouse model Methods 0.000 description 1
- HXEACLLIILLPRG-RXMQYKEDSA-N l-pipecolic acid Natural products OC(=O)[C@H]1CCCCN1 HXEACLLIILLPRG-RXMQYKEDSA-N 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- PYZRQGJRPPTADH-UHFFFAOYSA-N lamotrigine Chemical compound NC1=NC(N)=NN=C1C1=CC=CC(Cl)=C1Cl PYZRQGJRPPTADH-UHFFFAOYSA-N 0.000 description 1
- 229960001848 lamotrigine Drugs 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 108020001756 ligand binding domains Proteins 0.000 description 1
- KXGCNMMJRFDFNR-WDRJZQOASA-N linaclotide Chemical compound C([C@H](NC(=O)[C@@H]1CSSC[C@H]2C(=O)N[C@H]3CSSC[C@H](N)C(=O)N[C@H](C(N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N2)=O)CSSC[C@H](NC(=O)[C@H](C)NC(=O)[C@@H]2CCCN2C(=O)[C@H](CC(N)=O)NC3=O)C(=O)N[C@H](C(NCC(=O)N1)=O)[C@H](O)C)C(O)=O)C1=CC=C(O)C=C1 KXGCNMMJRFDFNR-WDRJZQOASA-N 0.000 description 1
- 108010024409 linaclotide Proteins 0.000 description 1
- 229960000812 linaclotide Drugs 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 208000004731 long QT syndrome Diseases 0.000 description 1
- 230000004777 loss-of-function mutation Effects 0.000 description 1
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 238000007726 management method Methods 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 210000003071 memory t lymphocyte Anatomy 0.000 description 1
- GMKMEZVLHJARHF-SYDPRGILSA-N meso-2,6-diaminopimelic acid Chemical compound [O-]C(=O)[C@@H]([NH3+])CCC[C@@H]([NH3+])C([O-])=O GMKMEZVLHJARHF-SYDPRGILSA-N 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 230000008883 metastatic behaviour Effects 0.000 description 1
- 208000010658 metastatic prostate carcinoma Diseases 0.000 description 1
- 229960004452 methionine Drugs 0.000 description 1
- 239000000693 micelle Substances 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 210000001700 mitochondrial membrane Anatomy 0.000 description 1
- 108091005601 modified peptides Proteins 0.000 description 1
- 230000004001 molecular interaction Effects 0.000 description 1
- 238000000302 molecular modelling Methods 0.000 description 1
- ZLVYMPOQNJTFSG-QMMMGPOBSA-N monoiodotyrosine Chemical compound OC(=O)[C@@H](NI)CC1=CC=C(O)C=C1 ZLVYMPOQNJTFSG-QMMMGPOBSA-N 0.000 description 1
- 210000001611 motor endplate Anatomy 0.000 description 1
- 230000000869 mutational effect Effects 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 210000000107 myocyte Anatomy 0.000 description 1
- DUWWHGPELOTTOE-UHFFFAOYSA-N n-(5-chloro-2,4-dimethoxyphenyl)-3-oxobutanamide Chemical compound COC1=CC(OC)=C(NC(=O)CC(C)=O)C=C1Cl DUWWHGPELOTTOE-UHFFFAOYSA-N 0.000 description 1
- OFYAYGJCPXRNBL-LBPRGKRZSA-N naphthalen-2-yl-3-alanine Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CC=CC2=C1 OFYAYGJCPXRNBL-LBPRGKRZSA-N 0.000 description 1
- 230000008764 nerve damage Effects 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- 230000000955 neuroendocrine Effects 0.000 description 1
- 210000004412 neuroendocrine cell Anatomy 0.000 description 1
- 208000015706 neuroendocrine disease Diseases 0.000 description 1
- 108010087904 neutravidin Proteins 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- CQYBNXGHMBNGCG-RNJXMRFFSA-N octahydroindole-2-carboxylic acid Chemical compound C1CCC[C@H]2N[C@H](C(=O)O)C[C@@H]21 CQYBNXGHMBNGCG-RNJXMRFFSA-N 0.000 description 1
- 210000000287 oocyte Anatomy 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 230000008052 pain pathway Effects 0.000 description 1
- 230000037324 pain perception Effects 0.000 description 1
- 238000004091 panning Methods 0.000 description 1
- TVIDEEHSOPHZBR-AWEZNQCLSA-N para-(benzoyl)-phenylalanine Chemical compound C1=CC(C[C@H](N)C(O)=O)=CC=C1C(=O)C1=CC=CC=C1 TVIDEEHSOPHZBR-AWEZNQCLSA-N 0.000 description 1
- 230000001769 paralizing effect Effects 0.000 description 1
- 230000001314 paroxysmal effect Effects 0.000 description 1
- MLBYLEUJXUBIJJ-UHFFFAOYSA-N pent-4-ynoic acid Chemical compound OC(=O)CCC#C MLBYLEUJXUBIJJ-UHFFFAOYSA-N 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N pentanoic acid group Chemical group C(CCCC)(=O)O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- 239000000816 peptidomimetic Substances 0.000 description 1
- 210000001428 peripheral nervous system Anatomy 0.000 description 1
- 210000005223 peripheral sensory neuron Anatomy 0.000 description 1
- 210000001322 periplasm Anatomy 0.000 description 1
- 239000012660 pharmacological inhibitor Substances 0.000 description 1
- 229960002036 phenytoin Drugs 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- PTMHPRAIXMAOOB-UHFFFAOYSA-N phosphoramidic acid Chemical compound NP(O)(O)=O PTMHPRAIXMAOOB-UHFFFAOYSA-N 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000002360 preparation method Methods 0.000 description 1
- 229940020463 prialt Drugs 0.000 description 1
- 125000001325 propanoyl group Chemical group O=C([*])C([H])([H])C([H])([H])[H] 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- 230000006337 proteolytic cleavage Effects 0.000 description 1
- 229940024999 proteolytic enzymes for treatment of wounds and ulcers Drugs 0.000 description 1
- 238000011002 quantification Methods 0.000 description 1
- 239000000700 radioactive tracer Substances 0.000 description 1
- 238000003259 recombinant expression Methods 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 238000009877 rendering Methods 0.000 description 1
- 230000000284 resting effect Effects 0.000 description 1
- 108091008146 restriction endonucleases Proteins 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 210000003705 ribosome Anatomy 0.000 description 1
- 229940043230 sarcosine Drugs 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 210000004739 secretory vesicle Anatomy 0.000 description 1
- 229940055619 selenocysteine Drugs 0.000 description 1
- ZKZBPNGNEQAJSX-UHFFFAOYSA-N selenocysteine Natural products [SeH]CC(N)C(O)=O ZKZBPNGNEQAJSX-UHFFFAOYSA-N 0.000 description 1
- JRPHGDYSKGJTKZ-UHFFFAOYSA-K selenophosphate Chemical compound [O-]P([O-])([O-])=[Se] JRPHGDYSKGJTKZ-UHFFFAOYSA-K 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 230000008313 sensitization Effects 0.000 description 1
- 238000002864 sequence alignment Methods 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 150000003385 sodium Chemical class 0.000 description 1
- 229940125794 sodium channel blocker Drugs 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 229910001415 sodium ion Inorganic materials 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- RPENMORRBUTCPR-UHFFFAOYSA-M sodium;1-hydroxy-2,5-dioxopyrrolidine-3-sulfonate Chemical compound [Na+].ON1C(=O)CC(S([O-])(=O)=O)C1=O RPENMORRBUTCPR-UHFFFAOYSA-M 0.000 description 1
- 230000007928 solubilization Effects 0.000 description 1
- 238000005063 solubilization Methods 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000002708 spider venom Substances 0.000 description 1
- 210000000278 spinal cord Anatomy 0.000 description 1
- 210000001032 spinal nerve Anatomy 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 1
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- UWYZHKAOTLEWKK-UHFFFAOYSA-N tetrahydro-isoquinoline Natural products C1=CC=C2CNCCC2=C1 UWYZHKAOTLEWKK-UHFFFAOYSA-N 0.000 description 1
- NQRYJNQNLNOLGT-UHFFFAOYSA-N tetrahydropyridine hydrochloride Natural products C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 1
- 108010093171 tetrodotoxin-binding protein Proteins 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- YUKQRDCYNOVPGJ-UHFFFAOYSA-N thioacetamide Chemical compound CC(N)=S YUKQRDCYNOVPGJ-UHFFFAOYSA-N 0.000 description 1
- 125000003396 thiol group Chemical group [H]S* 0.000 description 1
- 150000003573 thiols Chemical class 0.000 description 1
- RYYWUUFWQRZTIU-UHFFFAOYSA-K thiophosphate Chemical compound [O-]P([O-])([O-])=S RYYWUUFWQRZTIU-UHFFFAOYSA-K 0.000 description 1
- YSMODUONRAFBET-WHFBIAKZSA-N threo-5-hydroxy-L-lysine Chemical compound NC[C@@H](O)CC[C@H](N)C(O)=O YSMODUONRAFBET-WHFBIAKZSA-N 0.000 description 1
- 125000000341 threoninyl group Chemical group [H]OC([H])(C([H])([H])[H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 108010030159 thrombin receptor peptide 14 Proteins 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000024033 toxin binding Effects 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 230000002103 transcriptional effect Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- ILJSQTXMGCGYMG-UHFFFAOYSA-N triacetic acid Chemical compound CC(=O)CC(=O)CC(O)=O ILJSQTXMGCGYMG-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 229960004799 tryptophan Drugs 0.000 description 1
- PSVXZQVXSXSQRO-UHFFFAOYSA-N undecaethylene glycol Chemical compound OCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO PSVXZQVXSXSQRO-UHFFFAOYSA-N 0.000 description 1
- 241001515965 unidentified phage Species 0.000 description 1
- 210000001170 unmyelinated nerve fiber Anatomy 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 235000011912 vitamin B7 Nutrition 0.000 description 1
- 239000011735 vitamin B7 Substances 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 102000038650 voltage-gated calcium channel activity Human genes 0.000 description 1
- 108091023044 voltage-gated calcium channel activity Proteins 0.000 description 1
- 102000008538 voltage-gated sodium channel activity proteins Human genes 0.000 description 1
- 108040002416 voltage-gated sodium channel activity proteins Proteins 0.000 description 1
- JPZXHKDZASGCLU-LBPRGKRZSA-N β-(2-naphthyl)-alanine Chemical compound C1=CC=CC2=CC(C[C@H](N)C(O)=O)=CC=C21 JPZXHKDZASGCLU-LBPRGKRZSA-N 0.000 description 1
- 108091058550 ω-conotoxin Proteins 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/43504—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from invertebrates
- C07K14/43513—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from invertebrates from arachnidae
- C07K14/43518—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from invertebrates from arachnidae from spiders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/6811—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a protein or peptide, e.g. transferrin or bleomycin
- A61K47/6817—Toxins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
- A61P29/02—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID] without antiinflammatory effect
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/44—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material not provided for elsewhere, e.g. haptens, metals, DNA, RNA, amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/35—Valency
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2319/00—Fusion polypeptide
- C07K2319/30—Non-immunoglobulin-derived peptide or protein having an immunoglobulin constant or Fc region, or a fragment thereof, attached thereto
Definitions
- the present invention relates to the biochemical arts, in particular to therapeutic peptides and conjugates.
- Voltage-gated sodium channels are glycoprotein complexes responsible for initiation and propagation of action potentials in excitable cells such as central and peripheral neurons, cardiac and skeletal muscle myocytes, and
- Mammalian sodium channels are heterotrimers, composed of a central, pore-forming alpha (a) subunit and auxiliary beta ( ⁇ ) subunits. Mutations in alpha subunit genes have been linked to paroxysmal disorders such as epilepsy, long QT syndrome, and hyperkalemic periodic paralysis in humans, and motor endplate disease and cerebellar ataxia in mice. (Isom, Sodium channel beta subunits: anything but auxiliary, Neuroscientist 7(l):42-54 (2001)). The ⁇ -subunit modulates the localization, expression and functional properties of a-subunits in VGSCs.
- Voltage gated sodium channels comprise a family consisting of 9 different subtypes (Na v l . l-Na v 1.9). As shown in Table 1, these subtypes show tissue specific localization and functional differences (See, Goldin, A. L., Resurgence of sodium channel research, Annu Rev Physiol 63: 871-94 (2001); Wilson et al,
- compositions useful as inhibitors of voltage-gated ion channels US 2005/0187217 Al.
- Three members of the gene family (Na v l .8, 1.9, 1.5) are resistant to block by the well- known sodium channel blocker tetrodotoxin (TTX), demonstrating subtype specificity within this gene family.
- TTX sodium channel blocker tetrodotoxin
- Mutational analysis has identified glutamate 387 as a critical residue for TTX binding (See, Noda, M., H. Suzuki, et al, A single point mutation confers tetrodotoxin and saxitoxin insensitivity on the sodium channel ⁇ " FEBS Lett 259(1): 213-6 (1989)).
- Na v s voltage-gated sodium channels
- Antagonists of Na v channels can attenuate these pain signals and are useful for treating a variety of pain conditions, including but not limited to acute, chronic, inflammatory, and neuropathic pain.
- Known Nay antagonists such as TTX, lidocaine, bupivacaine, phenytoin, lamotrigine, and carbamazepine, have been shown to be useful for attenuating pain in humans and animal models. (See, Mao, J. and L. L.
- the a-subunits of TTX-sensitive, voltage-gated Na v 1.7 channels are encoded by the SCN9A gene.
- the Na v l .7 channels are preferentially expressed in peripheral sensory neurons of the dorsal root ganglia, some of which are involved in the perception of pain.
- mutations in the SCN9A gene have shown a critical role for this gene in pain pathways.
- a role for the Nayl .7 channel in pain perception was established by recent clinical gene-linkage analyses that revealed gain- of-function mutations in the SCN9A gene as the etiological basis of inherited pain syndromes such as primary erythermalgia (PE), inherited erythromelalgia (IEM), and paroxysmal extreme pain disorder (PEPD).
- PE primary erythermalgia
- IEM inherited erythromelalgia
- PEPD paroxysmal extreme pain disorder
- Loss-of-function mutations of the SCN9A gene result in a complete inability of an otherwise healthy individual to sense any form of pain, (e.g., Ahmad et al., A stop codon mutation in SCN9A causes lack of pain sensation, Hum. Mol. Genet. 16(17):2114-21 (2007)).
- a cell-specific deletion of the SCN9A gene in conditional knockout mice reduces their ability to perceive mechanical, thermal or inflammatory pain.
- Nassar et al. Nociceptor-specific gene deletion reveals a major role for Nayl .7 (PN1) in acute and inflammatory pain, Proc. Natl. Acad. Sci, U S A. 101(34): 12706-12711 (2004)).
- TTX-sensitive, voltage-gated Na v 1.3 channels are encoded by the SCN3A gene.
- SCN3A gene The a-subunits of TTX-sensitive, voltage-gated Na v 1.3 channels are encoded by the SCN3A gene.
- Expression of Na v 1.3 has been shown to be upregulated within DRG neurons following nerve injury and in thalamic neurons following spinal cord injury.
- Toxin peptides produced by a variety of organisms have evolved to target ion channels. Snakes, scorpions, spiders, bees, snails and sea anemones are a few examples of organisms that produce venom that can serve as a rich source of small bioactive toxin peptides or "toxins" that potently and selectively target ion channels and receptors. In most cases, these toxin peptides have evolved as potent antagonists or inhibitors of ion channels, by binding to the channel pore and physically blocking the ion conduction pathway. In some other cases, as with some of the tarantula toxin peptides, the peptide is found to antagonize channel function by binding to a region outside the pore (e.g., the voltage sensor domain).
- a region outside the pore e.g., the voltage sensor domain
- Native toxin peptides are usually between about 20 and about 80 amino acids in length, contain 2-5 disulfide linkages and form a very compact structure.
- Toxin peptides e.g., from the venom of scorpions, sea anemones and cone snails
- Toxin peptides have been isolated and characterized for their impact on ion channels.
- Such peptides appear to have evolved from a relatively small number of structural frameworks that are particularly well suited to addressing the critical issues of potency, stability, and selectivity.
- Dauplais et al On the convergent evolution of animal toxins: conservation of a diad of functional residues in potassium channel-blocking toxins with unrelated structures, J. Biol.
- scorpion and Conus toxin peptides for example, contain 10-40 amino acids and up to five disulfide bonds, forming extremely compact and constrained structures (microproteins) often resistant to proteolysis.
- the conotoxin and scorpion toxin peptides can be divided into a number of superfamilies based on their disulfide connections and peptide folds.
- ⁇ x- conotoxins have well-defined four cysteine/two disulfide loop structures (Loughnan, 2004) and inhibit nicotinic acetylcholine receptors.
- ⁇ -conotoxins have six cysteine/three disulfide loop consensus structures (Nielsen, 2000) and block calcium channels. Structural subsets of toxins have evolved to inhibit either voltage-gated or calcium-activated potassium channels.
- Spider venoms contain many peptide toxins that target voltage-gated ion channels, including Kv, Cav, and Nav channels. A number of these peptides are gating modifiers that conform to the inhibitory cystine knot (ICK) structural motif.
- ICK inhibitory cystine knot
- spider toxins In contrast to some scorpion and sea anemone toxins, many spider toxins do not affect the rate of inactivation but inhibit channel activity by restricting the movement of the voltage sensor into the open channel conformation, shifting their voltage dependence of activation to a more positive potential. Many of these spider toxins are promiscuous within and across voltage-gated ion channel families.
- site 1 toxins the ⁇ -conotoxins
- site 3 toxins including the a- scorpion toxins, some sea anemone toxins and ⁇ -conotoxins, bind to the S3-S4 linker of domain IV and slow channel inactivation
- site 4 toxins including the ⁇ -scorpion toxins, bind to the S3-S4 linker in domain II and facilitate channel activation.
- ⁇ -Conotoxin KIIIA (SEQ ID NO:530), a site 1 toxin originally isolated from Conus kinoshitai, is a C-terminally amidated peptide 16 amino acids in length that contains 6 cysteine residues engaged in 3 intramolecular disulfide bonds. It was initially characterized as an inhibitor of tetrodotoxin (TTX)-resistant sodium channels in amphibian dorsal root ganglion (DRG) neurons.
- TTX tetrodotoxin
- KIIIA has been found to block cloned mammalian (rodent) channels expressed in Xenopus laevis oocytes with the following rank order potency: Na v 1.2 > Na v 1.4 > Na v 1.6 > Na v 1.7 > Na v 1.3 > Na v 1.5.
- Intraperitoneal injection of KIIIA has demonstrated analgesic activity in a formalin-induced pain assay in mice with an ED 50 of 1.6 nmol/mouse (0.1 mg/kg) without observed motor impairment; some motor impairment but not paralytic acitity was observed at a higher dose (10 nmol). (See, Zhang et al, 2007).
- the NMR solution structure of KIIIA places these residues within or adjacent to an a-helix near the C-terminus of the molecule.
- Khoo et al. Structure of the analgesic ⁇ -conotoxin KIIIA and effects on the structure and function of disulfide deletion, Biochem. 48(6): 1210-1219, (2009).
- the disulfide bond between Cysl and Cys9 may be removed by substitution of alanine (KIIIA[C1A,C9A]) without greatly reducing the activity of the compound.
- KIIIA binds to the neurotoxin receptor site 1 in the outer vestibule of the conducting pore of the VGSCs and blocks the channel in an all-or-none manner. Recent studies have shown that some analogs of KIIIA only partially inhibit the sodium current and may be able to bind simultaneously with TTX and saxitoxin (STX). (See, Zhang et al, Cooccupancy of the outer vestibule of voltage-gated sodium channels by ⁇ -conotoxin KIIIA and saxitoxin or tetrodotoxin, J. Neurophys.
- OD1 (SEQ ID NO:589) is an a-like toxin isolated from the venom of the
- Huwentoxin-IV(HWTX-IV; SEQ ID NO:528) is a 35 residue C- terminal peptide amide with 3 disulfide bridges between 6 cysteine residues isolated from the venom of the Chinese bird spider, Selenocosmia huwena.
- TTX neuronal tetrodotoxin
- HWTX-IV inhibits TTX-sensitive sodium currents in adult rat DRG neurons with an IC 50 value of 30 nM but has no effect on TTX-resistant VGSCs at up to a 100 nM concentration.
- HWTX-IV was also 12-fold less potent against central neuronal sodium channels in rat hippocampus neurons, suggesting that it may be selective toward Nayl .7.
- Huwentoxin-I Huwentoxin-I (HWTX-I; SEQ ID NO:529), a related family member is less potent against VGSCs but is active against N- Type Ca v channels.
- huwentoxin-I a selective N-type calcium channel blocker
- Toxicon 45(1): 15-20, (2005) Liang et al, Properties and amino acid sequence of huwentoxin-I, a neurotoxin purified from the venom of the Chinese bird spider Selenocosmia huwena, Toxicon 31(8):969-78, (1993)).
- Thixopelma pruriens is a 30 amino acid polypeptide with a C-terminal free acid and 6 cysteine residues that form 3 disulfide bonds. It differs from other members of the ICK family because it contains only three residues between the fifth and sixth cysteine residues instead of the normal 4-11.
- ProTx-II is a potent inhibitor of several Nay channel sub-types including Na v 1.2, Nayl .7 (IC 50 ⁇ 1 nM), Nayl .5, and Nayl .8, as well as Cav3.1 channels but not Ky channels. (See, Middleton et al., Two tarantula peptides inhibit activation of multiple sodium channels, Biochem.
- ProTx-II gating modifiers of voltage-gated sodium channels, Toxicon 49(2): 194-201, (2007); Edgerton et al., Inhibition of the activation pathway of the T-type calcium channel CaV3.1 by ProTxII, Toxicon 56(4):624-636, (2010)).
- the "alanine scan" analogs of ProTx-II were tested against Navl .5, identifying Met6, Trp7, Argl3, Metl9, Val20, Arg22, Leu23, Trp24, Lys27, Leu29, and Trp30 as being important for activity.
- ProTx-II were well tolerated in rats, but 1 mg/kg doses were lethal. ProTx-II was not efficacious in a mechanical hyperalgesia study. (See, Schmalhofer et al, ProTx-II, a selective inhibitor of NaV1.7 sodium channels, blocks action potential propagation in nociceptors, Mol. Pharm. 74(5): 1476-1484, (2008)). Intrathecal administration was lethal at 0.1 mg/kg and not effective in the hyperalgesia study at lower doses. ProTx-II application to desheathed cutaneous nerves completely blocked the C-fiber compound action potential but had little effect on action potential propagation of the intact nerve. (See, Schmalhofer et al, 2008).
- ProTx-II is believed to bind to the S3-S4 linker of domain II of Na v l .7 to inhibit channel activation but may also interact with the domain IV voltage sensor and affect sodium channel activation at higher concentrations.
- the tarantula toxins ProTx-II and huwentoxin-IV differentially interact with human Navl .7 voltage sensors to inhibit channel activation and inactivation, Mol. Pharm. 78(6): 1 124-1134, (2010); Sokolov et al., Inhibition of sodium channel gating by trapping the domain II voltage sensor with protoxin II, Mol. Pharm.
- toxin peptides are complex processes in venomous organisms, and is an even more complex process synthetically. Due to their conserved disulfide structures and need for efficient oxidative refolding, toxin peptides present challenges to synthesis. (See, Steiner and Bulaj, Optimization of oxidative folding methods for cysteine-rich peptides: a study of conotoxins containing three disulfide bridges, J. Pept. Sci.
- Tetrodotoxin is a non-CNS-penetrant inhibitor of sodium channels including Na v l .3 and Na v l .7; although it cannot be used systemically due to lack of selectivity among sodium channel subtypes, its efficacy provides further validation for treating chronic pain syndromes with inhibitors of Nayl .7 and / or Nayl .3 in peripheral neurons.
- Polypeptides typically exhibit the advantage of greater target selectivity than is characteristic of small molecules.
- Non-CNS penetrant toxin peptides and peptide analogs selective for Navl .7 and/or Navl .3 are desired, and are provided by the present invention.
- the present invention is directed to a composition of matter comprising an isolated polypeptide, which is a peripherally-restricted Nayl .7 inhibitor.
- the isolated polypeptide is a dual inhibitor of Na v 1.7 and Na v 1.3 having the amino acid sequence DCLGFMR CIPDNDKCCRPNLVCSRTHKWCKYVF-NH 2 (SEQ ID NO: l), which is a peptide we have designated "GpTx-1", and which was isolated from the venom of the tarantula Grammostola porteri, as described herein.
- Other embodiments of the present invention comprise a peptide analog of GpTx-1.
- the present invention is directed to a composition of matter comprising an isolated polypeptide comprising the amino acid sequence of the formula:
- X aa 2 is any amino acid residue; or X ⁇ 1 is absent and X aa 2 is any amino acid residue;
- X aa 3 is Cys, if X aa 18 is Cys; or X aa 3 is SeCys, if X aa 18 is SeCys; or X aa 3 is an alkyl amino acid residue, if X aa 18 is an alkyl amino acid residue;
- X aa 4 is an acidic, hydrophobic, basic, or neutral hydrophilic amino acid residue
- X aa 5 is a Gly, Ala, hydrophobic, or basic amino acid residue
- X aa 6 is a Gly, Ala, 2-Abu, Nle, Nva, or hydrophobic amino acid residue
- X aa 7 is a Gly, Ala, aromatic, or hydrophobic amino acid residue
- X aa 8 is a basic, acidic, or neutral hydrophilic amino acid residue, or an Ala residue;
- X aa 9 is a basic or neutral hydrophilic amino acid residue
- X aa 10 is Cys if X aa 24 is Cys; or X aa 10 is SeCys if X aa 24 is SeCys;
- Xaa 11 is any amino acid residue
- X aa 12 is a Pro, acidic, neutral, or hydrophobic amino acid residue
- X aa is a basic, neutral hydrophilic, or acidic amino acid residue, or an Ala residue;
- X aa 17 is a Cys if X ⁇ 31 is Cys; or X aa 17 is a SeCys if X ⁇ 31 is SeCys;
- X aa 18 is a Cys, SeCys, or an alkyl amino acid residue
- X aa 19 is any amino acid residue
- X aa 20 is a Pro, Gly, basic, or neutral hydrophilic residue
- X aa 21 is a basic, hydrophobic, or neutral hydrophilic amino acid residue
- X aa 22 is a hydrophobic or basic amino acid residue
- X aa 23 is a hydrophobic, basic, or neutral hydrophilic amino acid residue
- X aa 24 is a Cys or SeCys residue
- X aa 25 is a Ser, Ala, or a neutral hydrophilic amino acid residue
- X aa 26 is an Ala, acidic, basic, or neutral hydrophilic amino acid residue
- X aa 27 is an acidic, basic, neutral hydrophilic or hydrophobic residue
- X aa 28 is an aromatic or basic amino acid residue
- X aa 29 is acidic, basic, or neutral hydrophilic amino acid residue
- X aa 30 is a Trp, 5-bromoTrp, 6-bromoTrp, 5-chloroTrp, 6-chloroTrp, 1-Nal, 2-
- X aa 31 is a Cys or SeCys
- X aa 33 is a hydrophobic or aromatic amino acid residue
- X aa 34 is any amino acid residue
- X aa 35 is a hydrophobic amino acid residue
- each of X aa 36 , X aa 37 , and X aa 38 is independently absent or is independently a neutral, basic, or hydrophobic amino acid residue;
- amino-terminal residue is optionally acetylated, biotinylated, 4- pentynoylated, or PEGylated;
- the carboxy-terminal residue is optionally amidated.
- composition of matter comprises an amino acid sequence selected from SEQ ID NOS: 3-30, 32-72, 74-134, 136-178, 180-211, 218-239, 241-305, 307-363, 366-386, 388-432, 434-474, 515-527, 532-588, 590, 591, 593-775, 777, 778, 780-788, 790-1049, and 1062-3086.
- the present invention includes a composition of matter, comprising an amino acid sequence selected from SEQ ID NOS: 3-134, 136-305, 307-386, 388-474, 515-527, 532-588, 590, 591, 593-1049, and 1062-3086.
- the present invention also encompasses a nucleic acid (e.g., DNA or
- compositions of the invention provide an effective method of treating, or preventing, pain, for example acute, persistent, or chronic pain.
- a dual inhibitor of Navl .7 and Navl .3 can be effective in some forms of chronic pain, particularly in cases where Navl .7 inhibition alone is insufficient.
- compositions of the present invention provide such selectivity against Navl .5 and Navl .4.
- the GpTx-1 peptide SEQ ID NO: 1
- the inhibition of Navl .4 is strongly state dependent, such that GpTx-1 exerts negligible effects on the physiological, non- inactivated states of Navl .4.
- the present invention is also directed to a pharmaceutical composition or a medicament containing the inventive composition of matter, and a pharmaceutically acceptable carrier.
- GpTx-1 and GpTx-1 peptide analogs are also useful as research tools.
- Figure 1 shows the reversed phase (RP) HPLC separation of crude venom extracted from Grammostola porteri. The tick marks along the x-axis represent time slices of fractionation.
- Figure 1 shows the activity of the isolated venom fraction (percent of control, POC) in the Nay 1.7 Ion Works® Quatrro (IWQ) assay.
- Fraction 31 indicated with rectangular box
- Figure 2 shows the high resolution electrospray ionization-mass spectrometry (ESI-MS) analysis of the indicated fraction (31) from the initial separation of Grammostola porteri venom.
- the arrows indicate the (m/z) ratios observed for the different ionization states of a peptide with a molecular weight of 4071 Da that was eventually identifed as GpTx-1. Additional peaks in the mass spectrum indicate that the venom fraction is a mixture of at least four distinct peptides.
- Figure 3 shows the matrix-assisted laser desorption ionization time-of- flight mass spectrometry (MALDI-TOF MS) analysis of fraction 31 from the separation of Grammostola porteri venom.
- the inset shows the low and mid mass ranges.
- the peak with an m/z ratio of 4074.9 was eventually identified as GpTx-1, but the fraction is a mixture of at least four distinct peptides.
- Figure 4 shows the RP-HPLC separation (sub-fractionation) of the material in fraction 31 from the intial separation of Grammostola porteri venom.
- the tick marks along the x-axis represent time slices of the sub-fractionation.
- Figure 4 shows the activity of the isolated sub-fractions (POC) in the Nayl .7 Ion Works® Quatrro (IWQ) assay.
- the major peak in the RP-HPLC chromatogram was the most active in the Na v l .7 assay and was deconvoluted to identify GpTx-1.
- Figure 5 shows LC-MS analysis of synthetic GpTx-1.
- the top panel shows the RP-HPLC chromatogram of UV absorbance at 214 nm.
- the bottom panel shows the total ion count (TIC) of the ESI-MS detector.
- Figure 6 shows the ESI-MS analysis of the peak with the retention time
- Figure 9 shows a ribbon representation of the peptide backbone and disulfide bonds (with the numbers corresponding to the cysteine residues) from the structural determination of GpTx-1 by NMR spectroscopy.
- Figure 10 shows GpTx- 1 oriented with the hydrophobic and putative binding face formed by the C-terminal ⁇ -strand and residues Phe5 and Met6 oriented toward the reader.
- On the right is a ribbon representation of the peptide backbone with the side chains of key residues depicted, and the left shows a partially transparent surface rendering of the molecule.
- Figure 11 shows the hydrophobic and putative binding face of
- GpTx-1 with an opaque surface.
- the molecule On the right, the molecule has been rotated clockwise by 90° to show the topological contrast between the flat hydrophobic face and the hydrophilic (solvent-exposed) face of the peptide.
- Figure 12A-N shows schematic structures of some embodiments of a composition of the invention that include one or more units of a pharmacologically active toxin, e.g., GpTx-1, or toxin (e.g., GpTx-1) peptide analog (squiggle) fused, via an optional peptidyl linker moiety such as but not limited to L5 or L10 described herein, with one or more domains of an immunoglobulin.
- Figure 12A represents a monovalent heterodimeric Fc-toxin peptide analog fusion or conjugate with the toxin peptide analog fused or conjugated to the C-terminal end of one of the immunoglobulin Fc domain monomers.
- Figure 12B represents a bivalent homodimeric Fc-toxin peptide analog fusion or conjugate, with toxin peptide analogs fused to the C-terminal ends of both of the immunoglobulin Fc domain monomers.
- Figure 12C represents a monovalent heterodimeric toxin peptide analog-Fc fusion or conjugate with the toxin peptide analog fused or chemically conjugated to the N-terminal end of one of the immunoglobulin Fc domain monomers.
- Figure 12D represents a bivalent homodimeric toxin peptide analog-Fc fusion or conjugate, with toxin peptide analogs fused to the N-terminal ends of both of the immunoglobulin Fc domain monomers.
- Figure 12E represents a monovalent heterotrimeric Fc-toxin peptide analog/ Ab comprising an immunoglobulin heavy chain (HC) + immunoglobulin light chain (LC) + an immunoglobulin Fc monomer with a toxin peptide analog fused to its C-terminal end.
- Figure 12 F represents a monovalent heterotetrameric (HT) antibody HC-toxin peptide analog fusion or conjugate, with a toxin peptide analog fused to the C-terminal end of one of the HC monomers.
- Figure 12G represents a bivalent HT antibody Ab HC-toxin peptide analog fusion or conjugate having toxin peptide analogs on the C-terminal ends of both HC monomers.
- Figure 12H represents a monovalent HT toxin peptide analog-LC Ab, with the toxin peptide analog fused to the N-terminal end of one of the LC monomers.
- Figure 121 represents a monovalent HT toxin peptide analog-HC Ab, with the toxin peptide analog fused to the N-terminal end of one of the HC monomers.
- Figure 12J represents a monovalent HT Ab LC-toxin peptide analog fusion or conjugate (i.e., LC- toxin peptide analog fusion or conjugate + LC + 2(HC)), with the toxin peptide analog fused to the C-terminal end of one of the LC monomers.
- Figure 12K represents a bivalent HT Ab LC-toxin peptide analog fusion or conjugate (i.e., 2(LC-toxin peptide analog fusion or conjugate) + 2(HC)), with toxin peptide analogs fused to the C- terminal end of both of the LC monomers.
- Figure 12 L represents a trivalent HT Ab LC-toxin peptide analog/HC-toxin peptide analog (i.e., 2(LC-toxin peptide analog fusion or conjugate) + HC-toxin peptide analog fusion or conjugate + HC), with the toxin peptide analogs fused to the C-terminal ends of both of the LC monomers and one of the HC monomers.
- Figure 12M represents a bivalent antibody with a toxin peptide analog moiety inserted into an internal loop of the immunoglobulin Fc domain of each HC monomer.
- Figure 12N represents a monovalent antibody with a toxin peptide analog moiety inserted into an internal loop of the immunoglobulin Fc domain of one of the HC monomers. Dimers or trimers will form spontaneously in certain host cells upon expression of a deoxyribonucleic acid (DNA) construct encoding a single chain. In other host cells, the cells can be placed in conditions favoring formation of dimers/trimers or the dimers/trimers can be formed in vitro. If more than one HC monomer, LC monomer, or immunoglobulin Fc domain monomer is part of a single embodiment, the individual monomers can be, if desired, identical or different from each other.
- DNA deoxyribonucleic acid
- Figure 13 A-F illustrates in situ hybridization studies detecting expression of Nay 1.7 message RNA in human trigeminal ganglia (Figure 13 A-B) and in human dorsal root ganglia (Figure 13C-F).
- Figure 13 A, Figure 13C, Figure 13E show
- Navl .7-specific staining also was detected in myenteric plexes of stomach and small intestine. No Na v 1.7-specific staining was detected in human cerebellum, cerebral cortex, adrenal medulla, or pituitary, and only light staining was detected in
- hypothalamic nuclei Staining from spinal cord was restricted to light staining in ventral motor areas and to spinal ependyma (epithelial cells lining the central canal). All staining was done under RNAse-free conditions on formalin-fixed, paraffin- embedded tissue.
- Probe was verified specific for Na v l .7 by verifying a positive signal on HEK-293T cell lines expressing cloned human Nayl .7 and a lack of signal on cell lines expressing any of the following sodium channels: hNayl .l (HEK-293T), hNavl .2 (CHO), hNavl .3 (CHO), hNa v 1.4 (HEK-293T), hNa v 1.5 (HEK-293T), hNa v 1.6 (HEK- 293T), rNayl .3 (HEK-293T), and on the parental 293T cell lines.
- Figure 14A-E shows that individual neurons acutely isolated from rat sensory ganglia express a heterogeneous population of sodium currents. Individual neurons expressed either fast, tetrodotoxin-sensitive sodium currents (likely Nay 1.7 and Na v 1.3), slow, tetrodotoxin-resistant sodium currents (likely Na v 1.8), or a mixed population (no one current type encodes > 90% of the total sodium current).
- Figure 14A-C show examples of a neuron expressing fast TTX-S ( Figure 14A), mixed ( Figure 14B), or slow TTX-R (Figure 14C) sodium currents.
- Figure 14D-E illustrate that neurons isolated from sensory ganglia of rats that have undergone spinal nerve ligation ("SNL") surgery ( Figure 14E) express a far greater proportion of fast TTX-S currents than control rats ( Figure 14D).
- Figure 15A-B shows sequence alignments of KIIIA (SEQ ID NO:530),
- GpTx-l(l-34) (SEQ ID NO: l), Huwentoxin-IV(SEQ ID NO:528), Huwentoxin-I (SEQ ID NO:529), and ProTx II (SEQ ID NO:531).
- Figure 16 shows the concentration-time profile of GpTx-1 in mouse plasma following a 1 mg/kg subcutaneous (“s.c") administration.
- the GpTx-1 concentration was below the lower limit of quantitation in two of the three animals, and the profile could not be obtained.
- Figure 17A shows the concentration-time profile of [Ala5]GpTx-l (SEQ
- Figure 17B shows the average plasma concentration-time profiles of
- peptide concentrations in the plasma were sustained at about 0.6 ⁇ , approximately 40-fold over the in vitro Navl .7 IC 50 by PatchXpress® (PX), for 3 h.
- Figure 18 shows the dose response relationship for GpTx-1 (SEQ ID NO:
- Figure 19A shows the dose response relationship for GpTx-1 (SEQ ID NO:
- Figure 19B shows the dose response relationship for [Ala5]GpTx-l
- Figure 20 shows the effect of GpTx-1 (SEQ ID NO: l) on hNavl .7 channels.
- Cells were held at -140 mV and peak inward hNavl .7 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .7 current before GpTx-1
- ⁇ 00 nM GpTx- trace shows hNavl .7 current after GpTx-1 addition.
- Figure 21 shows a dose-response curve of GpTx- 1 (SEQ ID NO : 1 ) against hNavl .7 channels.
- Peak inward hNavl .7 currents were measured at -10 mV in the presence of increasing concentrations of GpTx-1; cells were held at -140 mV. Currents were normalized with 100 representing Navl .7 current with no peptide addition and 0 representing Navl .7 current following complete block.
- the IC 50 of GpTx-1 against hNavl .7 channels was 7.40 nM.
- Figure 22 shows a time course of increasing concentrations of GpTx-1
- Figure 23 shows the effect of GpTx-1 (SEQ ID NO: l) on hNavl .3 channels.
- Cells were held at -140 mV and peak inward hNavl .3 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .3 current before GpTx-1
- '300 nM GpTx- trace shows hNavl .3 current after GpTx-1 addition.
- Figure 24 shows a dose-response curve of GpTx-1 (SEQ ID NO: l) against hNavl .3 channels. Peak inward hNavl .3 currents were measured at -10 mV in the presence of increasing concentrations of GpTx-1; cells were held at -140 mV.
- Figure 25 shows a time course of increasing concentrations of GpTx-1
- Figure 26 shows the effect of GpTx-1 (SEQ ID NO: l) on hNavl .4 channels.
- Cells were held at -140 mV and peak inward hNavl .4 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .4 current before GpTx-1
- '3 ⁇ GpTx- trace shows hNavl .4 current after GpTx-1 addition.
- Figure 27 shows a dose-response curve of GpTx-1 (SEQ ID NO: l) against hNavl .4 channels. Peak inward hNavl .4 currents were measured at -10 mV in the presence of increasing concentrations of GpTx-1; cells were held at -140 mV.
- Figure 28 shows a dose-response curve of GpTx-1 (SEQ ID NO: l) against hNavl .4 channels.
- Peak inward hNavl .4 currents were measured at -10 mV in the presence of increasing concentrations of GpTx-1; cells were held at a potential yielding approximately 20% inactivation.
- GpTx-1 is more potent against hNavl .4 when channels are partially inactivated. Currents were normalized with 100 representing Navl .4 current with no peptide addition and 0 representing Navl .4 current following complete block.
- the IC 50 of GpTx-1 against partially inactivated hNavl .4 channels was 0.26 ⁇ .
- Figure 29 shows a time course of increasing concentrations of GpTx-1
- Figure 30 shows a time course of increasing concentrations of GpTx-1
- Figure 31 shows the effect of GpTx-1 (SEQ ID NO: l) on hNavl .5 channels.
- Cells were held at -140 mV and peak inward hNavl .5 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .5 current before GpTx-1
- '3 ⁇ GpTx- trace shows hNavl .5 current after GpTx-1 addition.
- Figure 32 shows a dose-response curve of GpTx-1 (SEQ ID NO: l) against hNavl .5 channels. Peak inward hNavl .5 currents were measured at -10 mV in the presence of increasing concentrations of GpTx-1; cells were held at -140 mV.
- Figure 33 shows a time course of increasing concentrations of GpTx-1
- Figure 34 shows the effect of [Ala5]GpTx-l (SEQ ID NO:22) on hNavl .7 channels.
- Cells were held at -140 mV and peak inward hNavl .7 currents were measured at -10 mV.
- 'Baseline" trace shows hNavl .7 current before [Ala5]GpTx-l addition
- "300 nM [Ala5]GpTx-l” trace shows hNavl .7 current after [Ala5]GpTx-l addition.
- Cells were held at a potential yielding approximately 20% inactivation.
- Figure 35 shows a dose-response curve of [Ala5]GpTx-l (SEQ ID NO: 1]
- Peak inward hNavl .7 currents were measured at -10 mV in the presence of increasing concentrations of [Ala5]GpTx-l; cells were held at a potential yielding approximately 20% inactivation. Currents were normalized with 100 representing Navl .7 current with no peptide addition and 0 representing Navl .7 current following complete block.
- the IC 50 of [Ala5]GpTx-l against partially inactivated hNavl .7 channels was 13.2 nM.
- Figure 36 shows a time course of increasing concentrations of
- [Ala5]GpTx-l (SEQ ID NO:22) against hNavl .7 channels. Peak inward hNavl .7 currents were measured at -10 mV every 10 seconds in the presence of increasing concentrations of [Ala5]GpTx-l; cells were held at -140 mV (squares) or a potential yielding approximately 20% inactivation (circles). "Baseline” indicates hNavl .7 current in the absence of [Ala5]GpTx-l and "Wash" indicates hNavl .7 current following removal of [Ala5]GpTx-l .
- Figure 37 shows a time course of increasing concentrations of GpTx-1
- Figure 38 shows a representative dose-response curve of GpTx-1 (SEQ ID NO: 1).
- Figure 39 shows the effect of a single concentration of GpTx-1 (SEQ ID NO:
- Figure 40 shows the time course of increasing concentrations of GpTx-1
- Control indicates Nav current in the absence of GpTx-1
- TTX indicates Nav current in the presence of 300 nM TTX
- Wash indicates Nav current following removal of GpTx-1 and TTX. Note that GpTx-1 blocked all TTX-sensitive Nav current, since 300 nM TTX gave no additional current block.
- Figure 41 shows a representative dose-response curve of GpTx-1 (SEQ ID NO:
- Figure 42 shows the effect of GpTx-1 (SEQ ID NO: l) on TTX-sensitive
- Nav channels in mouse DRG neurons were held at a potential yielding approximately 20% inactivation and peak inward Nav currents were measured at -10 mV.
- "Baseline” trace shows Nav current before GpTx-1 addition
- "200 nM GpTx- 1" trace shows Nav current after GpTx-1 addition.
- Figure 43 shows the time course of increasing concentrations of
- [Ala5]GpTx-l(l-34) (SEQ ID NO:22)
- " ⁇ '” indicates Nav current in the presence of 300 nM TTX
- “Wash” indicates Nav current following removal of [Ala5]GpTx- 1(1-34) (SEQ ID NO:22) and TTX. Note that [Ala5]GpTx-l(l-34) (SEQ ID NO:22) blocked nearly all TTX-sensitive Nav current.
- Figure 44 shows a representative dose-response curve of [Ala5]GpTx-
- Figure 45 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) on
- TTX-sensitive Nav channels in mouse DRG neurons were held at -140 mV and peak inward Nav currents were measured at -10 mV.
- "Baseline" trace shows Nav current before addition of [Ala5]GpTx-l(l-34) (SEQ ID NO:22), "300 nM SEQ ID NO:22” trace shows Nav current after SEQ ID NO:22 addition, and "300 nM SEQ ID NO:22 + 300 nM TTX” trace shows Nav current after SEQ ID NO:22 and TTX addition. Note that [Ala5]GpTx-l(l-34) selectively blocked TTX-sensitive Nav current but not TTX-resistant Nav current.
- Figure 46 shows a time course of increasing concentrations of a homodimeric conjugate (Homodimeric Conjugate 1) of peptide monomer
- Figure 47 shows a representative dose-response curve of Homodimeric
- Figure 48 shows the recovery of hNavl .7 currents following washout of
- hNavl .7 currents were measured at -10 mV every 10 seconds in the presence of 3 nM or 300 nM Homodimeric Conjugate 1; cells were held at a -140 mV.
- Control indicates hNavl .7 current in the absence of Homodimeric Conjugate 1
- Wash indicates hNavl .7 current following removal of Homodimeric Conjugate 1. Note that hNavl .7 currents partially recover following washout of 3 nM Homodimeric Conjugate 1 but do not recover following washout of 300 nM of Homodimeric Conjugate 1.
- Figure 49 shows the effect of Homodimeric Conjugate 1 on hNavl .7 channels.
- Cells were held at -140 mV and peak inward hNavl .7 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .7 current before Homodimeric Conjugate 1 addition
- ' 10 nM Homodimeric Conjugate trace shows hNavl .7 current after Homodimeric Conjugate 1 addition.
- Figure 50 shows a time course of increasing concentrations of
- Homodimeric Conjugate 1 against hNavl .7 channels Peak inward hNavl .7 currents were measured at -10 mV every 10 seconds in the presence of increasing concentrations of Homodimeric Conjugate 1; cells were held at a potential yielding approximately 20% inactivation. "Control” indicates hNavl .7 current in the absence of Homodimeric Conjugate 1 and "Wash” indicates hNavl .7 current following removal of Homodimeric Conjugate 1.
- Figure 51 shows a representative dose-response curve of Homodimeric
- Figure 52 shows the effect of Homodimeric Conjugate 1 on hNavl .7 channels.
- Cells were held at a potential yielding approximately 20% inactivation and peak inward hNavl .7 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .7 current before Homodimeric Conjugate 1 addition
- "3 nM Homodimeric Conjugate 1" trace shows hNavl .7 current after Homodimeric Conjugate 1 addition.
- Figure 53 shows a time course of increasing concentrations of
- Homodimeric Conjugate 1 against hNavl .5 channels Peak inward hNavl .5 currents were measured at -10 mV every 10 seconds in the presence of increasing concentrations of Homodimeric Conjugate 1 ; cells were held at a -140 mV. "Control” indicates hNavl .5 current in the absence of Homodimeric Conjugate 1 and "Wash” indicates hNavl .5 current following removal of Homodimeric Conjugate 1. Note that hNavl .5 currents partially recover following washout of Homodimeric Conjugate 1.
- Figure 54 shows a representative dose-response curve of Homodimeric
- Figure 56 shows the time course of increasing concentrations of
- Figure 57 shows a representative dose-response curve of Homodimeric
- Figure 58 shows the effect of Homodimeric Conjugate 1 on hNavl .5 channels.
- Cells were held at a potential yielding approximately 20% inactivation and peak inward hNavl .5 currents were measured at -10 mV.
- "Baseline" trace shows hNavl .5 current before Homodimeric Conjugate 1 addition
- "2 uM Homodimeric Conjugate 1" trace shows hNavl .5 current after Homodimeric Conjugate 1 addition.
- Figure 59 shows the timecourse of the effect of [Ala5 ]GpTx-l(l-34)
- Figure 60 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the formalin pain model in male CD-I mice with a 1-hour pre-treatment dose of 5 mg/kg s.c. during the first phase (0-5 minutes post formalin injection). Neither the peptide nor the morphine positive control significantly reduced the time spent lifting and/or licking the affected paw during the first phase. Pharmacological reductions in flinching during this first phase generally reflect nonspecific effects on animal movement, consciousness, or health rather than an actual reduction in pain.
- Figure 61 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the formalin pain model in male CD-I mice with a 1-hour pre-treatment dose of 5 mg/kg s.c. during the second phase (5-40 minutes post formalin injection).
- the peptide but not the morphine control significantly reduced the time spent lifting and/or licking the affected paw during the second phase.
- the lack of effect of the morphine positive control does not reflect a "failed" assay, but the variability of mouse responses to this concentration of morphine. Morphine doses for future experiments were increased to 3 mg/kg.
- Figure 62 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) on paw edema in the formalin pain model in male CD-I mice with a 1-hour pre-treatment dose of 5 mg/kg s.c. Neither the peptide nor the morphine positive control significantly reduced the paw edema caused by formalin injection relative to the vehicle (PBS).
- Figure 63 shows the timecourse of the effect of [Ala5]GpTx-l(l-34)
- the lower peptide doses (1.67 and 0.5 mg/ kg s.c.) had no effect relative to the vehicle in the second phase.
- Terminal exposures peptide plasma concentrations at 45 min post formalin injection
- Figure 64 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the first phase of the formalin pain model in male CD-I mice with 1-hour pre-treatment doses of 5, 1.67, and 0.5 mg/kg s.c.
- the peptide had no effect at any dose in the first phase (0-5 minutes post formalin injection) relative to the vehicle, but the positive control, a 3 mg/kg s.c. dose of morphine, did significantly reduce the time spent lifting/licking the affected paw in the first phase.
- Figure 65 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the second phase of the formalin pain model in male CD-I mice with 1-hour pre- treatment doses of 5, 1.67, and 0.5 mg/kg s.c.
- the 5 mg/kg s.c. dose of peptide demonstrated a significant reduction of the time spent lifting/licking in the second phase of the study, as did the morphine positive control.
- the lower peptide doses (1.67 and 0.5 mg/ kg s.c.) had no effect relative to the vehicle in the second phase.
- Figure 66 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) on paw edema in the formalin pain model in male CD-I mice with a 1-hour pre-treatment doses of 5, 1.67, and 0.5 mg/kg s.c. Neither the peptide nor the morphine control significantly reduced the paw edema caused by formalin injection relative to the vehicle (PBS).
- Figure 67 shows the timecourse of the effect of [Ala5]GpTx-l(l-34)
- Figure 68 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the first phase of the formalin pain model in male CD-I mice with 1-hour pre-treatment doses of 5, 3, and 1 mg/kg s.c.
- the 5 mg/kg peptide dose and the 3 mg/kg s.c. dose of morphine had no effect in the first phase (0-5 minutes post formalin injection).
- Figure 69 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) in the second phase of the formalin pain model in male CD-I mice with 1 hour pre- treatment doses of 5, 3, and 1 mg/kg s.c.
- the 5 mg/kg s.c. dose of peptide demonstrated a significant reduction of the time spent lifting/licking in the second phase of the study, as did the morphine positive control.
- the lower peptide doses did not show a reduction in lifting/licking behavior relative to the vehicle control in the second phase.
- the 1 mg/kg s.c. group was not significantly different from the vehicle control, whereas the 3 mg/kg s.c. group appeared to significantly increase the time spent lifting/licking in the second phase.
- Figure 70 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) on paw edema in the formalin pain model in male CD-I mice with a 1-hour pre-treatment doses of 5, 3, and 1 mg/kg s.c. Neither the peptide nor the morphine control significantly reduced the paw edema caused by formalin injection relative to the vehicle (PBS).
- Figure 71 shows the effects of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at 3 and 5 mg/kg s.c. doses with a 1-hour pre-treatment time on the total basic movement component of locomotor activity in male CD-I mice. No doses of peptide significantly decreased exploratory behavior in relation to the vehicle control. Terminal exposures (peptide plasma concentrations at 2 h post peptide injection) were 1.79 ⁇ 0.38 and 0.89 ⁇ 0.33 ⁇ for the 5 and 3 mg/kg s.c. doses, respectively.
- Figure 72 shows the effects of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at 3 and 5 mg/kg s.c. doses with a 1-hour pre-treatment time on fine movement component of locomotor activity in male CD-I mice. At the 3 mg/kg dose, fine movement was not significantly affected. At the 5mg/kg dose, peptide significantly reduced the total fine movement relative to the vehicle.
- Figure 73 shows the effects of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at 3 and 5 mg/kg s.c. doses with a 1-hour pre-treatment time on the total rearing component of locomotor activity in male CD-I mice. No doses of peptide significantly decreased exploratory behavior in relation to the vehicle control.
- Figure 74 shows the effects of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at 3 and 5 mg/kg s.c. doses with a 1-hour pre-treatment time on the total time rearing component of locomotor activity in male CD-I mice. At the 3 mg/kg dose, total time rearing was not significantly affected. At the 5 mg/kg dose, peptide significantly reduced the total time rearing relative to the vehicle.
- Figure 75 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time and mexiletine at a 30 mg/kg intraperitoneal ("i.p.") dose with a 30 minute pre-treatment time on the veratridine- induced (I ⁇ g injection in the dorsal paw) spontaneous nociceptive behaviors in male CD-I mice measured for 20 minutes after veratridine injection. Overall there was a good paw lifting response from the vehicle group that was significantly reversed with mexiletine. At 5 mg/kg, [Ala5]GpTx-l(l-34) (SEQ ID NO:22) did not significantly reduce the lifting behavior. There is no reduction of paw edema across all groups.
- Figure 76 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time and mexiletine at a 30 mg/kg i.p. dose with a 30 minute pre-treatment time on the veratridine-induced (1 ⁇ g injection in the dorsal paw) paw edema in male CD-I mice. There is no reduction of paw edema across all groups.
- Figure 77 shows the time course of the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time and mexiletine at a 30 mg/kg i.p. dose with a 30 minute pre-treatment time on the veratridine-induced (1 ⁇ g injection in the dorsal paw) spontaneous nociceptive behaviors in male CD-I mice. Overall there was a good paw lifting response from the vehicle group that was significantly reversed with mexiletine throughout the course of the experiment.
- [Ala5]GpTx-l(l-34) (SEQ ID NO:22) was unable to significantly reduce the lifting behavior during the course of the entire experiment, though it did trend toward a reduction in paw lifting and licking during the 10-15 and 15-20 minute time bins.
- Terminal exposure (peptide plasma concentration at 1.5 h post peptide injection) was
- Figure 79 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at 1 and 5 mg/kg s.c. doses with a 1-hour pre-treatment time and mexiletine at a 30 mg/kg
- Figure 80 shows the time course of the effects of [Ala5]GpTx- 1(1-34) (SEQ ID NO:22) at 1 and 5 mg/kg s.c. doses with a 1 hour pre-treatment time and mexiletine at a 30 mg/kg i.p. dose with a 30 minute pre-treatment time on the veratridine -induced (1 ⁇ g injection in the dorsal paw) spontaneous nociceptive behaviors in male CD-I mice. Overall there was a good paw lifting response from the vehicle group that was significantly reversed with mexiletine throughout the course of the experiment.
- [Ala5]GpTx-l(l-34) did not significantly reduce the lifting behavior at any point during the course of the experiment.
- Terminal exposures peptide plasma concentration at 1.5 h post peptide injection
- Figure 81 A shows the reaction of alkyne-containing peptide [Phe6; Pral3]GpTx-l(l-34) (SEQ ID NO: 1049) with a 2 kDa PEG bis-azide via a copper catalyzed 1,3-dipolar cycloaddition reaction to obtain the site-specific homodimeric peptide with two triazole linkages by converting the propargylglycine or Pra residues in the sequences to 3-(l,2,3-triazol-4-yl)alanine or Atz residues.
- Figure 81 B shows the two step sequence of first reacting an alkyne- containing peptide [Ala5,Phe6,Pral3,Leu26,Arg28]GpTx-l(l-34) (SEQ ID NO:640) with an azido-PEGl 1-bromoacetamide linker via a copper catalyzed 1,3 -dipolar cycloaddition reaction to obtain the site-specifically PEGylated peptide-linker construct (SEQ ID NO: 1062) with a triazole linkage by converting the propargylglycine or Pra residue in the peptide sequence to a 3-(l,2,3-triazol-4-yl)alanine or Atz residue.
- immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088 react with the bromoacetamide functionality in peptide- linker to form a site-specific immunoglobulin-peptide conjugate with a stable thioacetamide linkage (Immunoglobulin-peptide Conjugate 1). If one cysteine reacts, then the result is a monovalent immunoglobulin-peptide conjugate as represented, but if both cysteines react, then the result is a bivalent immunoglobulin-peptide conjugate.
- Figure 82 shows a reducing SDS-PAGE gel of the site specific conjugation reaction mixture of peptide-linker construct (SEQ ID NO: 1062) with anti- DNP mAb (E273C) hlgGl (comprising immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088) prior to purification on the SP- column.
- Approximately 75% of mAb heavy chain (HC) shows a 5-kD shift, indicating conjugation of a single GpTX-1 peptide analog. The higher molecular weight band indicates higher order conjugate due to over reduction.
- Figure 83 shows the results from the purification of the site specific conjugation reaction mixture of peptide-linker construct (SEQ ID NO: 1062) with anti- DNP mAb (E273C) hlgGl (comprising immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088).
- the left panel shows the cation exchange chromatogram of UV absorbance at 280 nm of the conjugation reaction using a 5-mL HiTrap SP-HP column.
- the right panel shows the non-reducing SDS- PAGE gel (left) of the peaks from the cation exchange chromatogram indicating that the internal disulfide bonds of the antibody-peptide conjugates are intact and the reducing SDS-PAGE gel (right) of the peaks from the cation exchange chromatogram.
- Peak 1 shows the heavy chain (HC) of the non-reacted antibody.
- Peak 2 shows approximately 50% of the HC shifted indicating a monovalent antibody- GpTx-1 peptide analog conjugate.
- Peak 3 shows nearly 100% HC shifted indicating bivalent antibody- GpTx-1 peptide analog conjugate.
- Figure 84 shows the results from the purification of the site specific conjugation reaction mixture of peptide-linker construct (SEQ ID NO: 1062) with anti- DNP mAb (E273C) hlgGl Fc domain (homodimer of SEQ ID NO:3089).
- the left panel shows the cation exchange chromatogram of UV absorbance at 280 nm of the conjugation reaction using a 5 mL HiTrap SP-HP column.
- the right panel shows the non-reducing SDS-PAGE gel (left) of the peaks from the cation exchange
- Peak 1 shows approximately 50% of the HC shifted indicating a monovalent antibody-peptide conjugate.
- Peak 2 shows near 100% HC shifted indicating bivalent antibody-peptide conjugate.
- Figure 85 shows the analytical size exclusion chromatogram of UV absorbance at 280 nm for the (top panel) unmodified anti-DNP mAb (E273C) hlgGl (comprising SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:
- Figure 86 shows the results from the deconvo luted LC-MS TOF of the bivalent peptide conjugate of peptide-linker construct (SEQ ID NO: 1062) with anti- DNP mAb (E273C) hlgGl (comprising immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088), which is Immunoglobulin- peptide Conjugate 2. The presence of multiple peaks indicates different glycosylation iso forms.
- Figure 87 shows the results from the deconvo luted LC-MS TOF of the bivalent peptide conjugate of peptide-linker construct (SEQ ID NO: 1062) with anti- DNP mAb (E52C) hlgGl Fc domain (homodimer of SEQ ID NO:3089), which is Fc- peptide Conjugate 2.
- the presence of multiple peaks indicates different glycosylation iso forms.
- Figure 88 shows a schematic representation of Fc-peptide Conjugate 1 for illustrative purposes only.
- a homology model of the immunoglobulin anti-DNP mAb (E52C) hlgGl Fc domain (homodimer of SEQ ID NO:3089) was constructed from an immunoglobulin crystal structure (lHZH.pdb) and is depicted as a solid ribbon. Cys52 of SEQ ID 3089, the site of conjugation, is rendered in CPK format. The PEGl 1 linker is depicted as a solid tube, in an arbitrary conformation in this embodiment, connecting the C52 residue in the immunoglobulin Fc domain to the Atzl3 residue in the peptide.
- a homology model of the peptide (SEQ ID NO: 1046) was constructed from the NMR structure of GpTx-1 and is displayed as a solid ribbon and shown in this embodiment in an arbitrary relative orientation to the immunoglobulin Fc domain.
- FIG. 89 shows a schematic representation of Fc-peptide Conjugate 2 for illustrative purposes only.
- a homology model of the anti-DNP mAb (E52C) hlgGl Fc domain (homodimer of SEQ ID NO:3089) was constructed from an immunoglobulin crystal structure (lHZH.pdb) and is depicted as a solid ribbon. Cys52 residues of both Fc domain monomers (SEQ ID 3089), were the sites of conjugation, and are rendered in CPK format.
- the PEGl 1 linkers are depicted as solid tubes in this embodiment in an arbitrary conformation connecting the C52 residues in the immunoglobulin Fc domain to the Atzl3 residues in the peptides.
- Homology models of the peptide were constructed from the NMR structure of GpTx-1 and are displayed as a solid ribbon and shown in arbitrary relative orientations to the immunoglobulin Fc domain. Two peptides are shown to reflect the bivalent nature of Fc-peptide Conjugate 2.
- Figure 90 shows a schematic representation of Immunoglobulin-peptide Conjugate 1 for illustrative purposes only.
- a homology model of the anti-DNP mAb (E273C) hlgGl (comprising immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088) was constructed from an
- immunoglobulin crystal structure (lHZH.pdb) and is depicted as a solid ribbon.
- Cys273, the site of conjugation is rendered in CPK format.
- the PEGl 1 linker is depicted as a solid tube in an arbitrary conformation connecting the C273 residue in the immunoglobulin to the Atzl3 residue in the peptide.
- a homology model of the peptide (SEQ ID NO: 1046) was constructed from the NMR structure of GpTx-1 and is displayed as a solid ribbon and shown in an arbitrary relative orientation to the immunoglobulin in this embodiment.
- Figure 91 shows a schematic representation of Immunoglobulin-peptide Conjugate 2 for illustrative purposes only.
- a homology model of the anti-DNP mAb (E273C) hlgGl (comprising immunoglobulin monomers SEQ ID NO:3087; SEQ ID NO:3088; SEQ ID NO:3087; SEQ ID NO:3088) was constructed from an
- immunoglobulin crystal structure (lHZH.pdb) and is depicted as a solid ribbon. Both Cys273 residues, the sites of conjugation, are rendered in CPK format.
- the PEGl 1 linkers are depicted as solid tubes in an arbitrary conformation connecting the C273 residues in the immunoglobulin to the Atzl3 residues in the peptides.
- Homology models of the peptide (SEQ ID NO: 1046) were constructed from the NMR structure of GpTx-1 and are displayed as a solid ribbon and shown in arbitrary relative orientations to the immunoglobulin in this embodiment. Two peptides are shown to reflect the bivalent nature of Immunoglobulin-peptide Conjugate 2.
- Figure 94 shows the effects of peptide [Ala5]GpTx-l (SEQ ID NO:22) at 500 nM on fast sodium current and voltage-gated potassium currents. Traces are voltage-clamp recordings from a neuron enzymatically isolated from rat dorsal root ganglia. Inward sodium and outward potassium currents combined were evoked simultaneously by a 20 ms step to 0 mV from a holding voltage of -90 mV. Trace at right was taken approximately 160 seconds after addition of 500 nM peptide.
- [Ala5]GpTx-l (SEQ ID NO:22) at 500 nM blocked fast sodium current but not voltage- gated potassium currents.
- Figure 95 shows the timecourse of the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO: 30) in the formalin pain model in male CD-I mice with a 1-hour pre- treatment dose of 5 mg/kg s.c.
- [Glu29]GpTx-l(l-34) did not demonstrate a significant reduction of the time spent lifting/licking in the second phase of the study (5-40 minutes post formalin injection), but the morphine positive control did significantly reduce the time spent lifting/licking in the second phase.
- Terminal exposure peptide plasma concentration at 45 min post formalin injection was 0.502 ⁇ 0.271 ⁇ for the 5 mg/kg peptide dose.
- Figure 96 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) in the first phase of the formalin pain model in male CD-I mice with a 1-hour pretreatment dose of 5 mg/kg s.c.
- the 5 mg/kg peptide dose and the 3 mg/kg s.c. dose of morphine had no effect in the first phase (0-5 minutes post formalin injection).
- FIG. 97 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) in the second phase of the formalin pain model (5-40 minutes post formalin injection) in male CD-I mice with a 1 hour pre -treatment dose of 5 mg/kg s.c.
- the 5 mg/kg s.c. dose of peptide did not demonstrate a significant reduction of the time spent lifting/licking in the second phase of the study relative to the vehicle control.
- the morphine positive control did significantly reduce the time spent lifting/licking in the second phase.
- Figure 98 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) on paw edema in the formalin pain model in male CD-I mice with a 1-hour pre-treatment dose 5 mg/kg s.c. Neither the peptide nor the morphine control significantly reduced the paw edema caused by formalin injection relative to the vehicle (PBS).
- Figure 99 shows a repeat of the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total basic movement component of locomotor activity in male CD-I mice. Total basic movement was significantly reduced by the 5 mg/kg peptide dose. Terminal exposure (peptide plasma concentration at 2 h post peptide injection) was 2.1 ⁇ 0.47 ⁇ .
- Figure 100 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the fine movement component of locomotor activity in male CD-I mice. At the 5mg/kg dose, peptide significantly reduced the total fine movement relative to the vehicle.
- Figure 101 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre -treatment time on the total rearing component of locomotor activity in male CD-I mice. At the 5mg/kg dose, peptide significantly reduced total rearing relative to the vehicle control.
- Figure 102 shows the effect of [Ala5]GpTx-l(l-34) (SEQ ID NO:22) at a 5 mg/kg s.c. dose with a 1-hour pre -treatment time on the total time rearing component of locomotor activity in male CD-I mice. At the 5 mg/kg dose, peptide significantly reduced the total time rearing relative to the vehicle control.
- Figure 103 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total basic movement component of locomotor activity in male CD-I mice.
- the peptide dose did not significantly decrease exploratory behavior or total basic movement relative to the vehicle control.
- Terminal exposure peptide plasma concentration at 2 h post peptide injection
- Figure 104 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the fine movement component of locomotor activity in male CD-I mice. At the 5 mg/kg dose, fine movement was not significantly affected relative to the vehicle control.
- Figure 105 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) at 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total rearing component of locomotor activity in male CD-I mice.
- the peptide dose did not significantly decrease exploratory behavior or total rearing in relation to the vehicle control.
- Figure 106 shows the effect of [Glu29]GpTx-l(l-34) (SEQ ID NO:30) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total time rearing component of locomotor activity in male CD-I mice. At the 5 mg/kg dose, total time rearing was not significantly affected relative to the vehicle control.
- Figure 107 shows the effect of [Glu28]GpTx-l(l-34) (SEQ ID NO: 153) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total basic movement component of locomotor activity in male CD-I mice. Total basic movement was significantly reduced by the 5 mg/kg peptide dose. Terminal exposure (peptide plasma concentration at 2 h post peptide injection) was 1.28 ⁇ 0.403 ⁇ .
- Figure 108 shows the effect of [Glu28]GpTx- 1(1 -34) (SEQ ID NO: 153) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the fine movement component of locomotor activity in male CD-I mice. At the 5mg/kg dose, peptide significantly reduced the total fine movement relative to the vehicle.
- Figure 109 shows the effect of [Glu28]GpTx-l(l-34) (SEQ ID NO: 153) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total rearing component of locomotor activity in male CD-I mice. At the 5mg/kg dose, peptide significantly reduced total rearing relative to the vehicle control.
- Figure 110 shows the effect of [Glu28]GpTx- 1(1 -34) (SEQ ID NO: 153) at a 5 mg/kg s.c. dose with a 1-hour pre-treatment time on the total time rearing component of locomotor activity in male CD-I mice. At the 5 mg/kg dose, peptide significantly reduced the total time rearing relative to the vehicle control.
- Figure 111 shows the effects of 10 mg/kg s.c. doses of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2 with a 1-hour pre-treatment time on the total basic movement component of locomotor activity in male CD-I mice.
- the peptide conjugate doses did not significantly decrease exploratory behavior or total basic movement relative to the vehicle control.
- Terminal exposures peptide conjugate plasma concentrations at 24.5 h post peptide conjugate injection
- Terminal exposures were 0.034 ⁇ 0.009 ⁇ and 0.051 ⁇ 0.007 ⁇ for the 10 mg/kg s.c. doses of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2, respectively.
- Figure 112 shows the effects of 10 mg/kg s.c. doses of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2 with a 1-hour pre-treatment time on the fine movement component of locomotor activity in male CD-I mice. At the 10 mg/kg doses, fine movement was not significantly affected by either peptide conjugate relative to the vehicle control.
- Figure 113 shows the effects of 10 mg/kg s.c. doses of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2 with a 1-hour pre-treatment time on the total rearing component of locomotor activity in male CD-I mice.
- the peptide conjugate doses did not significantly decrease exploratory behavior or total rearing in relation to the vehicle control.
- Figure 114 shows the effects of 10 mg/kg s.c. doses of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2 with a 1-hour pre-treatment time on the total time rearing component of locomotor activity in male CD-I mice. At the 10 mg/kg doses, total time rearing was not significantly affected by either peptide conjugate relative to the vehicle control.
- Figure 115 shows the timecourse of the effects of Fc-peptide Conjugate 2 and Immunoglobulin-peptide Conjugate 2 in the formalin pain model in male CD-I mice with 1-hour pre-treatment doses of 10 mg/kg s.c.
- Figure 116 shows the effects of Fc-peptide Conjugate 2 and
- Immunoglobulin-peptide Conjugate 2 did not demonstrate a significant reduction of the time spent lifting/licking in the second phase of the study relative to the vehicle control.
- the morphine positive control did significantly reduce the time spent lifting/licking in the second phase.
- a cell includes populations of a plurality of cells.
- Polypeptide and “protein” are used interchangeably herein and include a molecular chain of two or more amino acids linked covalently through peptide bonds. The terms do not refer to a specific length of the product. Thus, “peptides,” and “oligopeptides,” are included within the definition of polypeptide. The terms include post-translational modifications of the polypeptide, for example, glycosylations, acetylations, biotinylations, 4-pentynoylations, PEGylations, phosphorylations and the like. In addition, protein fragments, analogs, mutated or variant proteins, fusion proteins and the like are included within the meaning of polypeptide.
- the terms also include molecules in which one or more amino acid analogs or non-canonical or unnatural amino acids are included as can be expressed recombinantly using known protein engineering techniques.
- fusion proteins can be derivatized as described herein by well-known organic chemistry techniques.
- a composition of the present invention that includes a peptide or polypeptide of the invention covalently linked, attached, or bound, either directly or indirectly through a linker moiety, to another peptide or polypeptide of the invention or to a half-life extending moiety is a "conjugate” or “conjugated” molecule, whether conjugated by chemical means (e.g., post-translationally or post-synthetically) or by recombinant fusion.
- Biotin is a water-soluble B-complex vitamin, i.e., vitamin B7, that is composed of an ureido (tetrahydroimidizalone) ring fused with a tetrahydrothiophene ring (See, Formula I).
- a valeric acid substituent is attached to one of the carbon atoms of the
- biotin is a coenzyme in the metabolism of fatty acids and leucine, and it plays a role in vivo in gluconeogenesis.
- Biotin binds very tightly to the tetrameric protein avidin (e.g., Chicken avidin, bacterial streptavidin, and neutravidin), with a dissociation equilibrium constant K D in the order of 10 14 M to 10 "1 ' M, which is one of the strongest known protein-ligand interactions, approaching the covalent bond in strength.
- avidin e.g., Chicken avidin, bacterial streptavidin, and neutravidin
- biotin-avidin non- covalent interaction is often used in different biotechnological applications. (See, Laitinen et al., Genetically engineered avidins and streptavidins, Cell Mol Life Sci. 63 (24): 2992-30177 (2006)).
- Biotinylated means that a substance is covalently conjugated to one or more biotin moieties.
- Biotinylated peptides useful in practicing the invention can be purchased commercially (e.g., Midwest Bio-Tech Inc.) or can be readily synthesized and biotinylated.
- Biotinylation of compounds, such as peptides can be by any known chemical technique. These include primary amine biotinylation, sulfhydryl
- biotinylation and carboxyl biotinylation.
- amine groups on the peptide which are present as lysine side chain epsilon-amines and N-terminal a-amines, are common targets for primary amine biotinylation biotinylation.
- Amine-reactive biotinylation reagents can be divided into two groups based on water solubility.
- N-hydroxysuccinimide (NHS)-esters of biotin have poor solubility in aqueous solutions. For reactions in aqueous solution, they must first be dissolved in an organic solvent, then diluted into the aqueous reaction mixture.
- organic solvents for this purpose are dimethyl sulfoxide (DMSO) and dimethyl formamide (DMF), which are compatible with most proteins at low concentrations.
- Sulfo-NHS-esters of biotin are more soluble in water, and are dissolved in water just before use because they hydrolyze easily.
- the water solubility of sulfo- NHS-esters stems from their sulfonate group on the N-hydroxysuccinimide ring and eliminates the need to dissolve the reagent in an organic solvent.
- NHS- and sulfo-NHS-esters have a half-life of several hours at pH 7, but only a few minutes at pH 9.
- the conditions for conjugating NHS- esters to primary amines of peptides include incubation temperatures in the range 4- 37°C, reaction pH values in the range 7-9, and incubation times from a few minutes to about 12 hours. Buffers containing amines (such as Tris or glycine) must be avoided because they compete with the reaction.
- the HABA dye (2-(4-hydroxyazobenzene) benzoic acid) method can be used to determine the extent of biotinylation.
- HABA dye is bound to avidin and yields a characteristic absorbance.
- biotin in the form of biotinylated protein or other molecule, it displaces the dye, resulting in a change in absorbance at 500 nm.
- the absorbance change is directly proportional to the level of biotin in the sample.
- 4-pentynoylation of an amino acid residue is typically by coupling 4- pentynoic acid via a standard amide bond reaction via the N-terminal or at a side chain.
- 4-pentynoylation can alternatively employ an alkyne in the copper-catalyzed 1,3 -dipolar cycloaddition reaction (the so- called “Click” reaction) to react with the azide in the azido-PEG molecule to link the peptide and the PEG via a triazole.
- An "isolated polypeptide” is a polypeptide molecule that is purified or separated from at least one contaminant polypeptide molecule with which it is ordinarily associated in the natural source of the polypeptide.
- An isolated polypeptide molecule is other than in the form or setting in which it is found in nature.
- "Toxin peptides” include peptides and polypeptides having the same amino acid sequence of a naturally occurring pharmacologically active peptide or polypeptide that can be isolated from a venom, and also include modified peptide analogs of such naturally occurring molecules.
- GpTx-1 (SEQ ID NO: l); GpTx-2 (SEQ ID NO:467); GpTx-3 (SEQ ID NO:468); GpTx-4 (SEQ ID NO:469); GpTx-5 (SEQ ID NO:470); GpTx-6(l- 35) (SEQ ID NO:471); and GpTx-7(l-35) (SEQ ID NO:473) are examples of toxin peptides.
- toxins that inhibit voltage-gated sodium channels include Huwentoxin-IV (ECLEI FKACN PSNDQ CCKSS KLVCS RKTRW CKYQI- NH 2 // SEQ ID NO:528) and Huwentoxin-I (ACKGV FDACT PGKNE CCPNR
- VCSDK HKWCK WKL //SEQ ID NO:529) isolated from the venom of tarantula Ornithoctonus huwena
- KIIIA CNCS SKWCR DHSRC C-NH 2 //SEQ ID NO:530
- ProTxII YCQKW MWTCD SERKC CEGMV CRLWC KKKLW //SEQ ID NO:531 isolated from the venom of tarantula Thrixopelma pruriens.
- alpha toxin OD1 GVRDAYIADD KNCVYTCASN GYCNTECTKN GAESGYCQWl GRYGNACWCl KLPDEVPIRIPGKCR-NH 2 //SEQ ID NO:589
- OSKl also known as OsKl
- a toxin peptide isolated from Orthochirus scrobiculosus scorpion venom e.g., Mouhat et al, K+ channel types targeted by synthetic OSKl, a toxin from Orthochirus scrobiculosus scorpion venom, Biochem. J.
- the toxin peptides are usually between about 20 and about 80 amino acids in length, contain 2-5 disulfide linkages and form a very compact structure.
- Toxin peptides e.g., from the venom of scorpions, sea anemones and cone snails
- scorpions and Conus toxin peptides contain 10-40 amino acids and up to five disulfide bonds, forming extremely compact and constrained structure (microproteins) often resistant to proteolysis.
- the conotoxin and scorpion toxin peptides can be divided into a number of superfamilies based on their disulfide connections and peptide folds.
- the solution structure of many toxin peptides has been determined by NMR spectroscopy, illustrating their compact structure and verifying conservation of family folding patterns. (E.g., Jerusalem et al., Ionisation behaviour and solution properties of the potassium-channel blocker ShK toxin, Eur. J. Biochem.
- peptide analog refers to a peptide having a sequence that differs from a peptide sequence existing in nature by at least one amino acid residue substitution, internal addition, or internal deletion of at least one amino acid, and/or amino- or carboxy- terminal end truncations or additions, and/or carboxy-terminal amidation.
- An "internal deletion” refers to absence of an amino acid from a sequence existing in nature at a position other than the N- or C-terminus.
- an “internal addition” refers to presence of an amino acid in a sequence existing in nature at a position other than the N- or C-terminus.
- Embodiments of the inventive composition of matter includes a toxin peptide analog, or a pharmaceutically acceptable salt thereof.
- “Toxin peptide analogs” contain modifications of a native toxin peptide sequence of interest (e.g., amino acid residue substitutions, internal additions or insertions, internal deletions, and/or amino- or carboxy- terminal end truncations, or additions as previously described above) relative to a native toxin peptide sequence of interest, such as GpTx-1
- Toxin peptide analogs of the present invention are 20 to about 80 amino acid residues long and, in relation to SEQ ID NO: l, have C ⁇ C 4 , C 2 -C 5 and C 3 -C 6 disulfide (or diselenide) bonding in which, C 1 , C 2 , C 3 , C 4 , C 5 and C 6 represent the order of cysteine (or selenocysteine) residues appearing in the primary sequence of the toxin peptide stated conventionally with the N-terminus of the peptide(s) on the left, with the first and sixth cysteines (or selenocysteines) in the amino acid sequence forming a disulfide bond (or diselenide bond, if SeCys), the second and fourth cysteines (or selenocysteines) forming a disulfide bond (or dis
- physiologically acceptable salt of the composition of matter, for example a salt of the toxin peptide analog, is meant any salt or salts that are known or later discovered to be pharmaceutically acceptable.
- pharmaceutically acceptable salts are: acetate; trifluoroacetate; hydrohalides, such as hydrochloride and hydrobromide; sulfate; citrate; maleate; tartrate; glycolate; gluconate; succinate; mesylate; besylate; salts of gallic acid esters (gallic acid is also known as 3,4, 5 trihydroxybenzoic acid) such as PentaGalloylGlucose (PGG) and epigallocatechin gallate (EGCG), salts of cholesteryl sulfate, pamoate, tannate and oxalate salts.
- PSG PentaGalloylGlucose
- EGCG epigallocatechin gallate
- fusion protein indicates that the protein includes polypeptide components derived from more than one parental protein or polypeptide.
- a fusion protein is expressed from a fusion gene in which a nucleotide sequence encoding a polypeptide sequence from one protein is appended in frame with, and optionally separated by a linker from, a nucleotide sequence encoding a polypeptide sequence from a different protein.
- the fusion gene can then be expressed by a recombinant host cell as a single protein.
- peptide mimetic refers to a peptide or protein having biological activity comparable to a naturally occurring protein of interest, for example, but not limited to, a toxin peptide molecule. These terms further include peptides that indirectly mimic the activity of a naturally occurring peptide molecule, such as by potentiating the effects of the naturally occurring molecule.
- peptide antagonist refers to a peptide that blocks or in some way interferes with the biological activity of a receptor of interest, or has biological activity comparable to a known antagonist or inhibitor of a receptor of interest, such as, but not limited to, an ion channel (e.g., Kvl .3) or a G-Protein Coupled Receptor (GPCR).
- GPCR G-Protein Coupled Receptor
- a “domain” of a protein is any portion of the entire protein, up to and including the complete protein, but typically comprising less than the complete protein.
- a domain can, but need not, fold independently of the rest of the protein chain and/or be correlated with a particular biological, biochemical, or structural function or location (e.g., a ligand binding domain, or a cytosolic, transmembrane or extracellular domain).
- soluble when in reference to a protein produced by recombinant DNA technology in a host cell is a protein that exists in aqueous solution; if the protein contains a twin-arginine signal amino acid sequence the soluble protein is exported to the periplasmic space in gram negative bacterial hosts, or is secreted into the culture medium by eukaryotic host cells capable of secretion, or by bacterial host possessing the appropriate genes (e.g., the kil gene).
- a soluble protein is a protein which is not found in an inclusion body inside the host cell.
- a soluble protein is a protein which is not found integrated in cellular membranes.
- an insoluble protein is one which exists in denatured form inside cytoplasmic granules (called an inclusion body) in the host cell, or again depending on the context, an insoluble protein is one which is present in cell membranes, including but not limited to, cytoplasmic membranes, mitochondrial membranes, chloroplast membranes, endoplasmic reticulum membranes, etc.
- proteins which are "soluble” i.e. , dissolved or capable of being dissolved
- the toxin peptide analog is synthesized by the host cell and segregated in an insoluble form within cellular inclusion bodies, which can then be purified from other cellular components in a cell extract with relative ease, and the toxin peptide analog can in turn be solubilized, refolded and/or further purified.
- soluble protein i.e. , a protein which when expressed in a host cell is produced in a soluble form
- a "solubilized” protein An insoluble recombinant protein found inside an inclusion body or found integrated in a cell membrane may be solubilized (i.e., rendered into a soluble form) by treating purified inclusion bodies or cell membranes with denaturants such as guanidine hydrochloride, urea or sodium dodecyl sulfate (SDS). These denaturants must then be removed from the solubilized protein preparation to allow the recovered protein to renature (refold).
- denaturants such as guanidine hydrochloride, urea or sodium dodecyl sulfate (SDS).
- inventive compositions can be refolded in active form, not all proteins will refold into an active conformation after solubilization in a denaturant and removal of the denaturant. Many proteins precipitate upon removal of the denaturant.
- SDS may be used to solubilize inclusion bodies and cell membranes and will maintain the proteins in solution at low concentration. However, dialysis will not always remove all of the SDS (SDS can form micelles which do not dialyze out); therefore, SDS-solubilized inclusion body protein and SDS-solubilized cell membrane protein is soluble but not refolded.
- a "secreted" protein refers to those proteins capable of being directed to the ER, secretory vesicles, or the extracellular space as a result of a secretory signal peptide sequence, as well as those proteins released into the extracellular space without necessarily containing a signal sequence. If the secreted protein is released into the extracellular space, the secreted protein can undergo extracellular processing to produce a "mature" protein. Release into the extracellular space can occur by many
- the toxin peptide analog can be synthesized by the host cell as a secreted protein, which can then be further purified from the extracellular space and/or medium.
- the term "recombinant” indicates that the material (e.g., a nucleic acid or a polypeptide) has been artificially or synthetically (i.e., non-naturally) altered by human intervention. The alteration can be performed on the material within, or removed from, its natural environment or state.
- a "recombinant nucleic acid” is one that is made by recombining nucleic acids, e.g., during cloning, DNA shuffling or other well known molecular biological procedures. Examples of such molecular biological procedures are found in Maniatis et al., Molecular Cloning. A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y. (1982).
- a “recombinant DNA molecule,” is comprised of segments of DNA joined together by means of such molecular biological techniques.
- the term “recombinant protein” or “recombinant polypeptide” as used herein refers to a protein molecule which is expressed using a recombinant DNA molecule.
- a “recombinant host cell” is a cell that contains and/or expresses a recombinant nucleic acid.
- polynucleotide or “nucleic acid” includes both single- stranded and double-stranded nucleotide polymers containing two or more nucleotide residues.
- nucleotide residues comprising the polynucleotide can be ribonucleotides or deoxyribonucleotides or a modified form of either type of nucleotide.
- Said modifications include base modifications such as bromouridine and inosine derivatives, ribose modifications such as 2',3'-dideoxyribose, and internucleotide linkage modifications such as phosphorothioate, phosphorodithioate, phosphoroselenoate, phosphorodiselenoate, phosphoroanilothioate, phosphoraniladate and
- oligonucleotide means a polynucleotide comprising 200 or fewer nucleotide residues. In some embodiments, oligonucleotides are 10 to 60 bases in length. In other embodiments, oligonucleotides are 12, 13, 14, 15, 16, 17, 18, 19, or 20 to 40 nucleotides in length. Oligonucleotides may be single stranded or double stranded, e.g., for use in the construction of a mutant gene. Oligonucleotides may be sense or antisense oligonucleotides.
- An oligonucleotide can include a label, including an isotopic label (e.g., 125 1, 14 C, 13 C, 35 S, 3 H, 2 H, 13 N, 15 N, 18 0, 17 0, etc.), for ease of quantification or detection, a fluorescent label, a hapten or an antigenic label, for detection assays.
- Oligonucleotides may be used, for example, as PCR primers, cloning primers or hybridization probes.
- a "polynucleotide sequence” or “nucleotide sequence” or “nucleic acid sequence,” as used interchangeably herein, is the primary sequence of nucleotide residues in a polynucleotide, including of an oligonucleotide, a DNA, and RNA, a nucleic acid, or a character string representing the primary sequence of nucleotide residues, depending on context. From any specified polynucleotide sequence, either the given nucleic acid or the complementary polynucleotide sequence can be determined. Included are DNA or RNA of genomic or synthetic origin which may be single- or double-stranded, and represent the sense or antisense strand.
- the left-hand end of any single-stranded polynucleotide sequence discussed herein is the 5 ' end; the left-hand direction of double-stranded polynucleotide sequences is referred to as the 5' direction.
- the direction of 5' to 3' addition of nascent RNA transcripts is referred to as the transcription direction; sequence regions on the DNA strand having the same sequence as the RNA transcript that are 5' to the 5' end of the R A transcript are referred to as "upstream sequences;" sequence regions on the DNA strand having the same sequence as the RNA transcript that are 3' to the 3' end of the RNA transcript are referred to as "downstream sequences.”
- an "isolated nucleic acid molecule” or “isolated nucleic acid sequence” is a nucleic acid molecule that is either (1) identified and separated from at least one contaminant nucleic acid molecule with which it is ordinarily associated in the natural source of the nucleic acid or (2) cloned, amplified, tagged, or otherwise distinguished from background nucleic acids such that the sequence of the nucleic acid of interest can be determined.
- An isolated nucleic acid molecule is other than in the form or setting in which it is found in nature.
- an isolated nucleic acid molecule includes a nucleic acid molecule contained in cells that ordinarily express a polypeptide (e.g., an oligopeptide or antibody) where, for example, the nucleic acid molecule is in a chromosomal location different from that of natural cells.
- a polypeptide e.g., an oligopeptide or antibody
- nucleic acid molecule encoding refers to the order or sequence of deoxyribonucleotides along a strand of deoxyribonucleic acid.
- the order of these deoxyribonucleotides determines the order of ribonucleotides along the mRNA chain, and also determines the order of amino acids along the polypeptide (protein) chain.
- the DNA sequence thus codes for the RNA sequence and for the amino acid sequence.
- gene is used broadly to refer to any nucleic acid associated with a biological function. Genes typically include coding sequences and/or the regulatory sequences required for expression of such coding sequences. The term “gene” applies to a specific genomic or recombinant sequence, as well as to a cDNA or mRNA encoded by that sequence.
- a "fusion gene” contains a coding region that encodes a polypeptide with portions from different proteins that are not naturally found together, or not found naturally together in the same sequence as present in the encoded fusion protein (i.e., a chimeric protein). Genes also include non-expressed nucleic acid segments that, for example, form recognition sequences for other proteins.
- Non- expressed regulatory sequences including transcriptional control elements to which regulatory proteins, such as transcription factors, bind, resulting in transcription of adjacent or nearby sequences.
- "Expression of a gene” or "expression of a nucleic acid” means transcription of DNA into RNA (optionally including modification of the RNA, e.g., splicing), translation of RNA into a polypeptide (possibly including subsequent post- translational modification of the polypeptide), or both transcription and translation, as indicated by the context.
- coding region or "coding sequence” when used in reference to a structural gene refers to the nucleotide sequences which encode the amino acids found in the nascent polypeptide as a result of translation of an mRNA molecule.
- the coding region is bounded, in eukaryotes, on the 5' side by the nucleotide triplet "ATG” which encodes the initiator methionine and on the 3' side by one of the three triplets which specify stop codons (i.e., TAA, TAG, TGA).
- control sequence or "control signal” refers to a
- control sequences for prokaryotes may include a promoter, a ribosomal binding site, and a transcription termination sequence.
- control sequences for eukaryotes may include promoters comprising one or a plurality of recognition sites for transcription factors, transcription enhancer sequences or elements, polyadenylation sites, and transcription termination sequences.
- Control sequences can include leader sequences and/or fusion partner sequences.
- Promoters and enhancers consist of short arrays of DNA that interact specifically with cellular proteins involved in transcription (Maniatis, et al., Science 236: 1237 (1987)). Promoter and enhancer elements have been isolated from a variety of eukaryotic sources including genes in yeast, insect and mammalian cells and viruses (analogous control elements, i.e., promoters, are also found in prokaryotes). The selection of a particular promoter and enhancer depends on what cell type is to be used to express the protein of interest. Some eukaryotic promoters and enhancers have a broad host range while others are functional in a limited subset of cell types (for review see Voss, et al, Trends Biochem.
- vector means any molecule or entity (e.g., nucleic acid, plasmid, bacteriophage or virus) used to transfer protein coding information into a host cell.
- expression vector refers to a recombinant DNA molecule containing a desired coding sequence and appropriate nucleic acid control sequences necessary for the expression of the operably linked coding sequence in a particular host cell.
- An expression vector can include, but is not limited to, sequences that affect or control transcription, translation, and, if introns are present, affect RNA splicing of a coding region operably linked thereto.
- Nucleic acid sequences necessary for expression in prokaryotes include a promoter, optionally an operator sequence, a ribosome binding site and possibly other sequences. Eukaryotic cells are known to utilize promoters, enhancers, and termination and polyadenylation signals.
- a secretory signal peptide sequence can also, optionally, be encoded by the expression vector, operably linked to the coding sequence of interest, so that the expressed polypeptide can be secreted by the recombinant host cell, for more facile isolation of the polypeptide of interest from the cell, if desired.
- Such techniques are well known in the art. (E.g., Goodey, Andrew R.; et al, Peptide and DNA sequences, U.S. Patent No. 5,302,697; Weiner et al, Compositions and methods for protein secretion, U.S. Patent No. 6,022,952 and U.S. Patent No. 6,335,178; Uemura et al, Protein expression vector and utilization thereof, U.S. Patent No. 7,029,909; Ruben et al, 27 human secreted proteins, US 2003/0104400 Al).
- operable combination refers to the linkage of nucleic acid sequences in such a manner that a nucleic acid molecule capable of directing the transcription of a given gene and/or the synthesis of a desired protein molecule is produced.
- the term also refers to the linkage of amino acid sequences in such a manner so that a functional protein is produced.
- a control sequence in a vector that is "operably linked" to a protein coding sequence is ligated thereto so that expression of the protein coding sequence is achieved under conditions compatible with the transcriptional activity of the control sequences.
- the term "host cell” means a cell that has been transformed, or is capable of being transformed, with a nucleic acid and thereby expresses a gene of interest.
- the term includes the progeny of the parent cell, whether or not the progeny is identical in morphology or in genetic make-up to the original parent cell, so long as the gene of interest is present. Any of a large number of available and well-known host cells may be used in the practice of this invention. The selection of a particular host is dependent upon a number of factors recognized by the art. These include, for example, compatibility with the chosen expression vector, toxicity of the peptides encoded by the DNA molecule, rate of transformation, ease of recovery of the peptides, expression characteristics, bio-safety and costs.
- useful microbial host cells in culture include bacteria (such as Escherichia coli sp.), yeast (such as Saccharomyces sp.) and other fungal cells, insect cells, plant cells, mammalian (including human) cells, e.g., CHO cells and HEK-293 cells. Modifications can be made at the DNA level, as well.
- the peptide-encoding DNA sequence may be changed to codons more compatible with the chosen host cell. For E. coli, optimized codons are known in the art.
- Codons can be substituted to eliminate restriction sites or to include silent restriction sites, which may aid in processing of the DNA in the selected host cell.
- the transformed host is cultured and purified.
- Host cells may be cultured under conventional fermentation conditions so that the desired compounds are expressed. Such fermentation conditions are well known in the art.
- transfection means the uptake of foreign or exogenous DNA by a cell, and a cell has been "transfected" when the exogenous DNA has been introduced inside the cell membrane.
- transfection techniques are well known in the art and are disclosed herein. See, e.g., Graham et al, 1973, Virology 52:456; Sambrook et al., 2001, Molecular Cloning: A Laboratory Manual, supra; Davis et al, 1986, Basic Methods in Molecular Biology, Elsevier; Chu et al, 1981, Gene 13 : 197.
- Such techniques can be used to introduce one or more exogenous DNA moieties into suitable host cells.
- transformation refers to a change in a cell's genetic characteristics, and a cell has been transformed when it has been modified to contain new DNA or RNA.
- a cell is transformed where it is genetically modified from its native state by introducing new genetic material via transfection, transduction, or other techniques.
- the transforming DNA may recombine with that of the cell by physically integrating into a chromosome of the cell, or may be maintained transiently as an episomal element without being replicated, or may replicate independently as a plasmid.
- a cell is considered to have been "stably transformed” when the transforming DNA is replicated with the division of the cell.
- transgene refers to an isolated nucleotide sequence, originating in a different species from the host, that may be inserted into one or more cells of a mammal or mammalian embryo.
- the transgene optionally may be operably linked to other genetic elements (such as a promoter, poly A sequence and the like) that may serve to modulate, either directly, or indirectly in conjunction with the cellular machinery, the transcription and/or expression of the transgene.
- the transgene may be linked to nucleotide sequences that aid in integration of the transgene into the chromosomal DNA of the mammalian cell or embryo nucleus (as for example, in homologous recombination).
- the transgene may be comprised of a nucleotide sequence that is either homologous or heterologous to a particular nucleotide sequence in the mammal's endogenous genetic material, or is a hybrid sequence (i.e. one or more portions of the transgene are homologous, and one or more portions are heterologous to the mammal's genetic material).
- the transgene nucleotide sequence may encode a polypeptide or a variant of a polypeptide, found endogenously in the mammal, it may encode a polypeptide not naturally occurring in the mammal (i.e. an exogenous polypeptide), or it may encode a hybrid of endogenous and exogenous polypeptides.
- the promoter may be homologous or heterologous to the mammal and/or to the transgene.
- the promoter may be a hybrid of endogenous and exogenous promoter elements (enhancers, silencers, suppressors, and the like).
- Peptides Recombinant DNA- and/or RNA-mediated protein expression and protein engineering techniques, or any other methods of preparing peptides, are applicable to the making of the inventive polypeptides, e.g., toxin peptide analogs and fusion protein conjugates thereof (e.g., fusion proteins containing a toxin peptide analog and an immunoglobulin Fc domain, transthyretin, or human serum albumin).
- the peptides can be made in transformed host cells. Briefly, a recombinant DNA molecule, or construct, coding for the peptide is prepared. Methods of preparing such DNA molecules are well known in the art.
- sequences encoding the peptides can be excised from DNA using suitable restriction enzymes.
- Any of a large number of available and well-known host cells may be used in the practice of this invention. The selection of a particular host is dependent upon a number of factors recognized by the art. These include, for example, compatibility with the chosen expression vector, toxicity of the peptides encoded by the DNA molecule, rate of transformation, ease of recovery of the peptides, expression characteristics, bio-safety and costs. A balance of these factors must be struck with the understanding that not all hosts may be equally effective for the expression of a particular DNA sequence.
- useful microbial host cells in culture include bacteria (such as Escherichia coli sp.), yeast (such as Saccharomyces sp.) and other fungal cells, insect cells, plant cells, mammalian (including human) cells, e.g., CHO cells and HEK293 cells. Modifications can be made at the DNA level, as well.
- the peptide-encoding DNA sequence may be changed to codons more compatible with the chosen host cell.
- optimized codons are known in the art. Codons can be substituted to eliminate restriction sites or to include silent restriction sites, which may aid in processing of the DNA in the selected host cell.
- the transformed host is cultured and purified.
- Host cells may be cultured under conventional fermentation conditions so that the desired compounds are expressed. Such fermentation conditions are well known in the art.
- the DNA optionally further encodes, 5' to the coding region of a fusion protein, a signal peptide sequence (e.g., a secretory signal peptide) operably linked to the expressed toxin peptide analog.
- Peptide compositions of the present invention can also be made by synthetic methods.
- Solid phase synthesis is the preferred technique of making individual peptides since it is the most cost-effective method of making small peptides.
- well known solid phase synthesis techniques include the use of protecting groups, linkers, and solid phase supports, as well as specific protection and deprotection reaction conditions, linker cleavage conditions, use of scavengers, and other aspects of solid phase peptide synthesis. Suitable techniques are well known in the art. (E.g., Merrifield (1973), Chem. Polypeptides, pp. 335-61 (Katsoyannis and Panayotis eds.); Merrifield (1963), J. Am. Chem. Soc.
- amino acid substitution in an amino acid sequence is typically designated herein with a one-letter abbreviation for the amino acid residue in a particular position, followed by the numerical amino acid position relative to a native sequence of interest, which is then followed by the one-letter symbol for the amino acid residue substituted in.
- T30D symbolizes a substitution of a threonine residue by an aspartate residue at amino acid position 30, relative to the native sequence of interest.
- Non-canonical amino acid residues can be incorporated into a peptide within the scope of the invention by employing known techniques of protein engineering that use recombinantly expressing cells. (See, e.g., Link et al, Non- canonical amino acids in protein engineering, Current Opinion in Biotechnology, 14(6):603-609 (2003)).
- the term "non-canonical amino acid residue” refers to amino acid residues in D- or L-form that are not among the 20 canonical amino acids generally incorporated into naturally occurring proteins, for example, ⁇ -amino acids, homoamino acids, cyclic amino acids and amino acids with derivatized side chains. Examples include (in the L-form or D-form) ⁇ -alanine, ⁇ -aminopropionic acid, piperidinic acid, aminocaprioic acid, aminoheptanoic acid, aminopimelic acid, desmosine,
- diaminopimelic acid N"-ethylglycine, N"-ethylaspargine, hydroxylysine, allo- hydroxylysine, isodesmosine, allo-isoleucine, ⁇ -methylarginine, N"-methylglycine, N"-methylisoleucine, N"-methylvaline, , ⁇ -carboxyglutamate, ⁇ - ⁇ , ⁇ , ⁇ -trimethyllysine, ⁇ - ⁇ -acetyllysine, O-phosphoserine, N"-acetylserine, N"-formylmethionine, 3- methylhistidine, 5 -hydroxylysine, and other similar amino acids, and those listed in Table 3 below, and derivatized forms of any of these as described herein.
- Table 3 contains some exemplary non-canonical amino acid residues that are useful in accordance with the present invention and associated abbreviations as typically used herein, although the skilled practitioner will understand that different abbreviations and nomenclatures may be applicable to the same substance and appear interchangeably herein.
- Some amino acid sequences, as recited herein may include " ⁇ H ⁇ -" at the N- terminal, which represents an N-terminal amino group, and/or may include "- ⁇ Free Acid ⁇ " at the C-terminal, which represents a C-terminal carboxy group.
- Table 3 Useful non-canonical amino acids for amino acid addition, insertion, or substitution into peptide sequences in accordance with the present invention.
- N "-methy lleucine N"-MeL; NMeL; NMeLeu;
- the one or more useful modifications to peptide domains of the inventive compositions can include amino acid additions or insertions, amino acid deletions, peptide truncations, amino acid substitutions, and/or chemical derivatization of amino acid residues, accomplished by known chemical techniques.
- the thusly modified amino acid sequence includes at least one amino acid residue inserted or substituted therein, relative to the amino acid sequence of the native sequence of interest, in which the inserted or substituted amino acid residue has a side chain comprising a nucleophilic or electrophilic reactive functional group by which the peptide is covalently conjugated to a linker and/or half-life extending moiety.
- nucleophilic or electrophilic reactive functional group examples include, but are not limited to, a thiol, a primary amine, a seleno, a hydrazide, an aldehyde, a carboxylic acid, a ketone, an aminooxy, a masked (protected) aldehyde, or a masked (protected) keto functional group.
- amino acid residues having a side chain comprising a nucleophilic reactive functional group include, but are not limited to, a lysine residue, a homolysine, an ⁇ , ⁇ - diaminopropionic acid residue, an ⁇ , ⁇ -diaminobutyric acid residue, an ornithine residue, a cysteine, a homocysteine, a glutamic acid residue, an aspartic acid residue, or a selenocysteine ("SeCys”) residue.
- Amino acid residues are commonly categorized according to different chemical and/or physical characteristics.
- the term “acidic amino acid residue” refers to amino acid residues in D- or L-form having side chains comprising acidic groups.
- R a is independently, at each instance, H or R b ; and R b is independently, at each instance Ci_ 6 alkyl substituted by 0, 1, 2 or 3 substituents selected from halo, Ci_ 4 alk, Ci_ 3 haloalk, -OCi_ 4 alk, -NH 2 , -NHCi_ 4 alk, and -N(Ci_ 4 alk)Ci_ 4 alk; or any protonated form thereof, including alanine, valine, leucine, isoleucine, proline, serine, threon
- aromatic amino acid residue refers to amino acid residues in D- or L-form having side chains comprising aromatic groups.
- exemplary aromatic residues include tryptophan, tyrosine, 3-(l-naphthyl)alanine, histidine, or phenylalanine residues.
- basic amino acid residue refers to amino acid residues in D- or L-form having side chains comprising basic groups.
- Exemplary basic amino acid residues include histidine, lysine, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, and homoarginine (hR) residues.
- the term "hydrophilic amino acid residue” refers to amino acid residues in D- or L-form having side chains comprising polar groups.
- Exemplary hydrophilic residues include cysteine, serine, threonine, histidine, lysine, asparagine, aspartate, glutamate, glutamine, and citrulline (Cit) residues.
- lipophilic amino acid residue refers to amino acid residues in D- or L-form having sidechains comprising uncharged, aliphatic or aromatic groups.
- exemplary lipophilic sidechains include phenylalanine, isoleucine, leucine, methionine, valine, tryptophan, and tyrosine.
- Alanine (A) is amphiphilic— it is capable of acting as a hydrophilic, or lipophilic (i.e., hydrophobic), residue. Alanine, therefore, is included within the definition of both "lipophilic” (i.e., "hydrophobic") residue and "hydrophilic” residue.
- nonfunctional or “neutral” amino acid residue refers to amino acid residues in D- or L-form having side chains that lack acidic, basic, or aromatic groups.
- exemplary neutral amino acid residues include methionine, glycine, alanine, valine, isoleucine, leucine, and norleucine (Nle) residues.
- toxin peptide analogs can result from conservative modifications of the amino acid sequences of the toxin polypeptides disclosed herein. Conservative modifications will produce half-life extending moiety- conjugated peptides having functional, physical, and chemical characteristics similar to those of the conjugated (e.g., PEG-conjugated) peptide from which such modifications are made. Such conservatively modified forms of the conjugated toxin peptide analogs disclosed herein are also contemplated as being an embodiment of the present invention.
- substantial modifications in the functional and/or chemical characteristics of peptides may be accomplished by selecting substitutions in the amino acid sequence that differ significantly in their effect on maintaining (a) the structure of the molecular backbone in the region of the substitution, for example, as an a-helical conformation, (b) the charge or hydrophobicity of the molecule at the target site, or (c) the size of the molecule.
- a “conservative amino acid substitution” may involve a substitution of a native amino acid residue with a nonnative residue such that there is little or no effect on the polarity or charge of the amino acid residue at that position.
- any native residue in the polypeptide may also be substituted with alanine, as has been previously described for "alanine scanning mutagenesis” (see, for example, MacLennan et al, Acta Physiol. Scand. SuppL, 643:55-67 (1998); Sasaki et al, 1998, Adv. Biophys. 35: 1-24 (1998), which discuss alanine scanning mutagenesis).
- the amino acid sequence of the toxin peptide is modified in one or more ways relative to a native toxin peptide sequence of interest, such as, but not limited to, a native GpTx-1 sequence (SEQ ID NO: l), a peptide analog of GpTx-1, or any other toxin peptides having amino acid sequences as set for in Table 5 A, Table 5B, and Table 31, or elsewhere herein.
- the one or more useful modifications can include amino acid additions or insertions, amino acid deletions, peptide truncations, amino acid
- substitutions, and/or chemical derivatization of amino acid residues accomplished by known chemical techniques. Such modifications can be, for example, for the purpose of enhanced potency, selectivity, and/or proteolytic stability, or the like.
- Those skilled in the art are aware of techniques for designing peptide analogs with such enhanced properties, such as alanine scanning, rational design based on alignment mediated mutagenesis using known toxin peptide sequences and/or molecular modeling.
- protease is synonymous with “peptidase”.
- Proteases comprise two groups of enzymes: the endopeptidases which cleave peptide bonds at points within the protein, and the exopeptidases, which remove one or more amino acids from either N- or C-terminus respectively.
- proteinase is also used as a synonym for endopeptidase.
- the four mechanistic classes of proteinases are: serine proteinases, cysteine proteinases, aspartic proteinases, and metallo-proteinases. In addition to these four mechanistic classes, there is a section of the enzyme nomenclature which is allocated for proteases of unidentified catalytic mechanism. This indicates that the catalytic mechanism has not been identified.
- Cleavage subsite nomenclature is commonly adopted from a scheme created by Schechter and Berger (Schechter I. & Berger A., On the size of the active site in proteases. I. Papain, Biochemical and Biophysical Research Communication, 27: 157 (1967); Schechter I. & Berger A., On the active site of proteases. 3. Mapping the active site of papain; specific inhibitor peptides of papain, Biochemical and Biophysical Research Communication, 32:898 (1968)). According to this model, amino acid residues in a substrate undergoing cleavage are designated PI, P2, P3, P4 etc. in the N- terminal direction from the cleaved bond.
- the residues in the C-terminal direction are designated ⁇ , ⁇ 2', ⁇ 3', P4', etc.
- the skilled artisan is aware of a variety of tools for identifying protease binding or protease cleavage sites of interest.
- the PeptideCutter software tool is available through the ExPASy (Expert Protein Analysis System) proteomics server of the Swiss Institute of Bioinformatics (SIB;
- PeptideCutter searches a protein sequence from the SWISS-PROT and/or TrEMBL databases or a user-entered protein sequence for protease cleavage sites. Single proteases and chemicals, a selection or the whole list of proteases and chemicals can be used. Different forms of output of the results are available: tables of cleavage sites either grouped alphabetically according to enzyme names or sequentially according to the amino acid number. A third option for output is a map of cleavage sites. The sequence and the cleavage sites mapped onto it are grouped in blocks, the size of which can be chosen by the user. Other tools are also known for determining protease cleavage sites. (E.g., Turk, B. et al., Determination of protease cleavage site motifs using mixture-based oriented peptide libraries, Nature
- the serine proteinases include the chymo trypsin family, which includes mammalian protease enzymes such as chymotrypsin, trypsin or elastase or kallikrein.
- the serine proteinases exhibit different substrate specificities, which are related to amino acid substitutions in the various enzyme subsites interacting with the substrate residues. Some enzymes have an extended interaction site with the substrate whereas others have a specificity restricted to the PI substrate residue.
- a proline residue in position ⁇ normally exerts a strong negative influence on trypsin cleavage.
- the positioning of R and K in ⁇ results in an inhibition, as well as negatively charged residues in positions P2 and PI * .
- Chymotrypsin preferentially cleaves at a W, Y or F in position PI (high specificity) and to a lesser extent at L, M or H residue in position PI . (Keil, 1992). Exceptions to these rules are the following: When W is found in position PI, the cleavage is blocked when M or P are found in position ⁇ at the same time.
- a proline residue in position ⁇ nearly fully blocks the cleavage independent of the amino acids found in position PI .
- the cleavage is blocked by the presence of a Y residue in position ⁇ .
- H is located in position PI, the presence of a D, M or W residue also blocks the cleavage.
- Membrane metallo-endopeptidase (NEP; neutral endopeptidase, kidney- brush-border neutral proteinase, enkephalinase, EC 3.4.24.11) cleaves peptides at the amino side of hydrophobic amino acid residues. (Connelly, JC et al, Neutral
- Endopeptidase 24.11 in Human Neutrophils Cleavage of Chemotactic Peptide, PNAS, 82(24):8737-8741 (1985)).
- Thrombin preferentially cleaves at an R residue in position P 1. (Keil, 1992).
- the natural substrate of thrombin is fibrinogen.
- Optimum cleavage sites are when an R residue is in position PI and Gly is in position P2 and position ⁇ .
- Caspases are a family of cysteine proteases bearing an active site with a conserved amino acid sequence and which cleave peptides specifically following D residues. (Earnshaw WC et al, Mammalian caspases: Structure, activation, substrates, and functions during apoptosis, Annual Review of Biochemistry, 68:383-424 (1999)).
- Arg-C proteinase preferentially cleaves at an R residue in position PI .
- the cleavage behavior seems to be only moderately affected by residues in position ⁇ . (Keil, 1992).
- the Asp-N endopeptidase cleaves specifically bonds with a D residue in position ⁇ . (Keil, 1992).
- Desired amino acid substitutions can be determined by those skilled in the art at the time such substitutions are desired.
- amino acid substitutions can be used to identify important residues of the peptide sequence, or to increase or decrease the affinity of the peptide or vehicle-conjugated peptide molecules described herein.
- Naturally occurring residues may be divided into classes based on common side chain properties:
- Conservative amino acid substitutions may involve exchange of a member of one of these classes with another member of the same class.
- Conservative amino acid substitutions may encompass non-naturally occurring amino acid residues, which are typically incorporated by chemical peptide synthesis rather than by synthesis in biological systems. These include peptidomimetics and other reversed or inverted forms of amino acid moieties.
- Non-conservative substitutions may involve the exchange of a member of one of these classes for a member from another class. Such substituted residues may be introduced into regions of the toxin peptide analog.
- the hydropathic index of amino acids may be considered. Each amino acid has been assigned a hydropathic index on the basis of its hydrophobicity and charge
- threonine (-0.7); serine (-0.8); tryptophan (-0.9); tyrosine (-1.3); proline (-1.6); histidine (-3.2); glutamate (-3.5); glutamine (-3.5); aspartate (-3.5); asparagine (-3.5); lysine (- 3.9); and arginine (-4.5).
- hydropathic amino acid index in conferring interactive biological function on a protein is understood in the art (see, for example, Kyte et al., 1982, J. Mol. Biol. 157: 105-131). It is known that certain amino acids may be substituted for other amino acids having a similar hydropathic index or score and still retain a similar biological activity. In making changes based upon the hydropathic index, in certain embodiments, the substitution of amino acids whose hydropathic indices are within ⁇ 2 is included. In certain embodiments, those that are within ⁇ 1 are included, and in certain embodiments, those within ⁇ 0.5 are included.
- hydrophilicity values have been assigned to these amino acid residues: arginine (+3.0); lysine (+3.0); aspartate (+3.0 ⁇ 1); glutamate (+3.0 ⁇ 1); serine (+0.3); asparagine (+0.2); glutamine (+0.2); glycine (0); threonine (-0.4); proline (-0.5 ⁇ 1); alanine (-0.5); histidine (-0.5); cysteine (-1.0); methionine (-1.3); valine (- 1.5); leucine (-1.8); isoleucine (-1.8); tyrosine (-2.3); phenylalanine (-2.5) and tryptophan (-3.4).
- the substitution of amino acids whose hydrophilicity values are within ⁇ 2 is included, in certain embodiments, those that are within ⁇ 1 are included, and in certain embodiments, those within ⁇ 0.5 are included.
- Examples of conservative substitutions include the substitution of one non-polar (hydrophobic) amino acid residue such as isoleucine, valine, leucine norleucine, alanine, or methionine for another, the substitution of one polar
- (hydrophilic) amino acid residue for another such as between arginine and lysine, between glutamine and asparagine, between glycine and serine, the substitution of one basic amino acid residue such as lysine, arginine or histidine for another, or the substitution of one acidic residue, such as aspartic acid or glutamic acid for another.
- the phrase "conservative amino acid substitution” also includes the use of a chemically derivatized residue in place of a non-derivatized residue, provided that such polypeptide displays the requisite bioactivity.
- Other exemplary amino acid substitutions that can be useful in accordance with the present invention are set forth in Table 4 below.
- composition of matter comprising an isolated polypeptide comprising the amino acid sequence of the formula:
- [00221 ] is absent; or Xaa 1 is any amino acid residue and X aa 2 is any amino acid residue; or X ⁇ 1 is absent and X aa 2 is any amino acid residue;
- X aa is Cys, if X aa is Cys; or X aa is SeCys, if X aa is SeCys; or X aa is an alkyl amino acid residue, if X aa 18 is an alkyl amino acid residue (for example, X aa 3 and X aa 18 can be, independently, Ala or 2-Abu residues);
- X aa 4 is an acidic, hydrophobic, basic, or neutral hydrophilic amino acid residue (e.g., X aa 4 can be selected from Ala, Glu, Asp, phosphoserine, phosphotyrosine, gamma- carboxyglutamic acid, Phe, Pro, He, Leu, Met, Val, Trp, Tyr, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, co-methyl- arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl- lysine, ⁇ - ⁇ - methyl lysine, Dab, cyclohexylglycine (Chg), cyclohexylalanine (Cha), glycine, norleucine, norvaline, 1-Nal, 2-Nal, 4-phenyl-phenylalanine (Bip), Gin,
- X aa 5 is a Gly, Ala, hydrophobic, or basic amino acid residue (e.g., X aa 5 can be selected from Ala, Phe, He, Leu, Met, Val, Trp, Tyr, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, glycine, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, cyclohexylglycine (Chg),
- X aa 6 is a Gly, Ala, 2-Abu, norleucine (Nle), norvaline (Nva), or hydrophobic amino acid residue (e.g., X aa 6 can be selected from Ala, Phe, He, Leu, Met, Val, Trp, Tyr, proline, thiaproline, methionine, glycine, 1-Nal, 2-Nal, l 'NMe-Trp, cyclopentylglycine (Cpg), phenylglycine, N-methylleucine, N-methylphenylalanine, N-methylvaline,
- cyclohexylglycine Chg
- cyclohexylalanine Cha
- 2-chloro-phenylalanine 4-chloro- phenylalanine, 3,4-dichlorophenylalanine, 4-trifluoromethyl-phenylalanine, and 4- phenyl-phenylalanine (Bip) residues
- cyclohexylglycine Chg
- cyclohexylalanine Cha
- X aa 7 is a Gly, Ala, aromatic, or hydrophobic amino acid residue (e.g., X aa 7 can be selected from Gly, Ala, Phe, He, Leu, Met, Val, Trp, Tyr, Pro, 2-pyridinylalanine, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, cyclohexylglycine (Chg),
- X aa 8 is a basic, acidic, or neutral hydrophilic amino acid residue, or an Ala residue (e.g., X aa 8 can be selected from Ala, Arg, Lys, His, homolysine, ornithine, arginine, N- methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl- histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, Ser, Thr, Asn, Gin, Cit, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, Asp, and Glu residues);
- X aa 9 is a basic or neutral hydrophilic amino acid residue (e.g., X aa 9 can be selected from Ala, Pro, Met, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, co- aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine,
- X aa 10 is Cys if X aa 24 is Cys; or X aa 10 is SeCys if X aa 24 is SeCys;
- Xaa 11 is any amino acid residue (e.g., X aa n can be selected from Ala, Asp, Glu, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, Pra, Atz, homolysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1- methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2-Nal, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4-phenyl-phenylalanine (Bip) residues);
- X aa 12 is a Pro, acidic, neutral, or hydrophobic amino acid residue (e.g., X aa 12 can be selected from alanine, valine, leucine, isoleucine, proline, serine, threonine, aspartic acid, glutamatic acid, glycine, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, Cha, and 4-phenyl-phenylalanine (Bip), cyclohexylglycine (Chg), cyclohexylalanine (Cha), asparagine, glutamine, methionine, hydroxyproline, phenylalanine, tryptophan, and tyrosine);
- X aa 13 is any amino acid residue (e.g., X aa 13 can be selected from Ala, Asp, Glu, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, Pra, Atz, homo lysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1- methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2-Nal, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4-phenyl-phenylalanine (Bip) residues);
- X aa 14 is any amino acid residue (e.g., X aa 14 can be selected from Ala, Asp, Glu, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, Pra, Atz, homo lysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1- methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2-Nal, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4-phenyl-phenylalanine (Bip) residues);
- X aa 16 is a basic, neutral hydrophilic, or acidic amino acid residue, or an Ala residue (e.g., X aa 16 can be selected from Ala, Pro, Met, Arg, Lys, His, Pra, Atz, homo lysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl- histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, Gin, Asn, Ser, Thr, Cit, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, Asp, and Glu residues);
- X aa 17 is a Cys if X ⁇ 31 is Cys; or X aa 17 is a SeCys if X ⁇ 31 is SeCys;
- X aa 18 is a Cys, SeCys, or an alkyl amino acid residue
- X aa 19 is any amino acid residue (e.g., X aa 19 can be selected from Ala, Asp, Glu, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, homolysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2-Nal, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4- phenyl-phenylalanine (Bip) residues);
- X aa 20 is a Pro, Gly, basic, or neutral hydrophilic residue (e.g., X ⁇ 20 can be selected from Ala, Pro, Met, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, co- aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, Gin, Asn, Ser, Thr, and Cit residues);
- X ⁇ 20 can be selected from Ala, Pro, Met, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, co- aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, Gin
- X aa 21 is a basic, hydrophobic, or neutral hydrophilic amino acid residue (e.g., X aa 21 can be selected from Ala, Phe, Pro, He, Leu, Met, Val, Trp, Tyr, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl- histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, cyclohexylglycine (Chg), cyclohexylalanine (Cha), glycine, norleucine, norvaline, 1- Nal, 2-Nal, 4-phenyl-phenylalanine (Bip), Gin, Asn, Ser, Thr, and Cit residues);
- X aa 21 can be selected from Ala, Phe, Pro, He,
- X aa 22 is a hydrophobic or basic amino acid residue (e.g., X aa 22 can be selected from Ala, Phe, He, Leu, Met, Val, Trp, Tyr, Arg, Lys, His, homolysine, ornithine, arginine, N- methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl- histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, glycine, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4-phenyl-phenylalanine (Bip) residues);
- X aa 23 is a hydrophobic, basic, or neutral hydrophilic amino acid residue (e.g., X aa 23 can be selected from Ala, Phe, Pro, He, Leu, Met, Val, Trp, Tyr, Arg, Lys, His, Pra, Atz, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, co-methyl- arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ - methyl lysine, Dab, cyclohexylglycine (Chg), cyclohexylalanine (Cha), glycine, norleucine, norvaline, 1-Nal, 2-Nal, 4-phenyl-phenylalanine (Bip), Gin, Asn, Ser, Thr, and Cit residues);
- X aa 23 can be selected from Ala, P
- X aa 24 is a Cys or SeCys residue
- X aa 25 is a Ser, Ala, or a neutral hydrophilic amino acid residue (e.g., X aa 25 is selected from Ala, Gly, Pro, Met, glycine, Gin, Asn, Ser, Thr, and Cit residues);
- X aa 26 is an Ala, acidic, basic, or neutral hydrophilic amino acid residue (e.g., X aa 26 can be selected from Ala, Pro, Met, Arg, Lys, His, homolysine, ornithine, arginine, N- methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl- histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, glycine, Asp, Glu, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, Gin, Asn, Ser, Thr, and Cit residues);
- X aa 27 is an acidic, basic, neutral hydrophilic, or hydrophobic residue (e.g., X aa 27 can be selected from Thr, Leu, He, Val, Ser, Met, Gin, Asn, Phe, Tyr, Trp, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, ⁇ -aminoarginine, co-methyl- arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl- lysine, ⁇ - ⁇ - methyl lysine, Dab, Asp, Glu, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, cyclohexylglycine (Chg), cyclohexylalanine (Cha), norleucine, norvaline, 1-Nal,
- X aa 28 is an aromatic or basic amino acid residue (e.g., X aa 28 can be selected fromPhe, Trp, Tyr, Arg, Lys, His, homolysine, ornithine, arginine, N-methyl-arginine, co- aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine,
- X aa 29 is an acidic, basic, or neutral hydrophilic amino acid residue (e.g., X aa 29 can be selected from Ala, Asp, Glu, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, Phe, Gly, His, Lys, Asn, Pro, Gin, Arg, Ser, Thr, Tyr, Pra, Atz, homolysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, and Dab residues).
- X aa 29 can be selected from Ala, Asp, Glu, phosphoserine, phosphotyrosine, gamma-carboxyglutamic acid, Phe, Gly, His, Lys, Asn, Pro, Gin, Arg
- X ⁇ 29 is an acidic or neutral hydrophilic residue (e.g., an Ala, Asp, Glu, Gly, Asn, Gin, Ser, Thr, phosphoserine, phosphotyrosine, or gamma- carboxyglutamic acid residue [e.g., SEQ ID NOS: 1071-2798]);
- an acidic or neutral hydrophilic residue e.g., an Ala, Asp, Glu, Gly, Asn, Gin, Ser, Thr, phosphoserine, phosphotyrosine, or gamma- carboxyglutamic acid residue [e.g., SEQ ID NOS: 1071-2798]
- X aa 30 is a Trp, 5-bromoTrp, 6-bromoTrp, 5-chloroTrp, 6-chloroTrp, 1-Nal, 2-Nal, or thioTrp residue;
- X aa 31 is a Cys or SeCys
- X aa 33 is a hydrophobic or aromatic amino acid residue (e.g., X aa 33 can be selected from Phe, He, Leu, Met, Val, Trp, Tyr, norleucine, norvaline, 1-Nal, 2-Nal, 2-pyridyl-alanine, 3-pyridyl-alanine, 4-pyridyl-alanine, 4-piperidinyl-alanine, l 'NMe-Trp,
- cyclohexylglycine Chg
- cyclohexylalanine Cha
- 2-chloro-phenylalanine 4-chloro- phenylalanine, 3,4-dichlorophenylalanine, 4-trifluoromethyl-phenylalanine, and 4- phenyl-phenylalanine (Bip) residues
- cyclohexylglycine Chg
- cyclohexylalanine Cha
- X aa 34 is any amino acid residue (e.g., X aa 34 can be selected from Ala, Asp, Glu, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, Pra, Atz, homolysine, ornithine, N-methyl-arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1- methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, cyclohexylglycine (Chg), cyclohexylalanine (Cha), 2-chloro-phenylalanine, 4-chloro-phenylalanine, 3,4- dichlorophenylalanine, 4-trifluor
- X aa 35 is a hydrophobic amino acid residue (e.g., Xaa 35 can be selected from Phe, He, Leu, Met, Val, Trp, Tyr, norleucine, norvaline, 1-Nal, 2-Nal, l 'NMe-Trp, cyclohexylglycine (Chg), cyclohexylalanine (Cha), 2-chloro-phenylalanine, 4-chloro-phenylalanine, 3,4- dichlorophenylalanine, 4-trifluoromethyl-phenylalanine, and 4-phenyl-phenylalanine (Bip) residues);
- each of X aa , X aa , and X aa is independently absent or is independently a neutral
- each of Xaa , X aa , and X aa can be independently absent or independently selected from Ala, Phe, Gly, His, He, Lys, Leu, Met, Asn, Pro, Gin, Arg, Ser, Thr, Val, Trp, Tyr, homolysine, ornithine, N-methyl- arginine, ⁇ -aminoarginine, ⁇ -methyl-arginine, 1-methyl-histidine, 3-methyl-histidine, homoarginine, N-methyl-lysine, ⁇ - ⁇ -methyl lysine, Dab, norleucine, norvaline, 1-Nal, 2- Nal, cyclohexylglycine (Chg), cyclohexylalanine (Cha), and 4-phenyl-phenylalanine (Bip) residues);
- residue X aa and residue X aa are both SeCys residues, there is a diselenide bond between residue X aa 17 and residue X aa 31 ;
- amino-terminal residue is optionally acetylated, biotinylated, 4- pentynoylated, or PEGylated;
- the carboxy-terminal residue is optionally amidated.
- Many particular examples of such C-terminally amidated appear in Table 5A and Table 5B.
- Another example of the inventive composition described immediately above is the composition of matter wherein theisolated polypeptide comprises the amino acid sequence of the formula:
- composition of matter comprise an Ala, Gly, 2-Abu, Nle, or Nva residue at position X ⁇ of SEQ ID NO:475, with some of these embodiments also having a Glu residue at position X of SEQ ID NO:475, for example, [Ala5, Glu26]GpTx-l, [Gly5, Glu26]GpTx-l, [2-Abu5, Glu26]GpTx-l, [Nle5, Glu26]GpTx-l, or [Nva5,
- Glu26]GpTx-l peptide analogs which, optionally, can also have additional substituted amino acid residues at other amino acid positions and/or additional residues at the N- terminal and/or C-terminal end.
- Other useful examples comprise an Asp, Glu, or Gin residue at position X 9 of SEQ ID NO: 475 or X aa 29 of SEQ ID NO:476, with or without a Glu residue at position X aa 27 of SEQ ID NO:475, e.g., [Ala5, Asp28]GpTx-l, [Ala5, Glu28]GpTx-l, [Ala5, Gln28]GpTx-l, [Ala5, Glu26, Asp28]GpTx-l, [Ala5, Glu26, Asp28]GpTx-l, [Ala5, Glu26, Glu28]GpTx-l, [Ala5, Glu26, Gln28]GpTx-l, [G
- inventive compositions are unconjugated and conjugated peptide analogs of GpTx-1 having one of the amino acid sequences as set forth in Table 5A, Table 5B, and Table 31.
- inventive isolated polypeptides include those having an amino acid sequence selected from SEQ ID NOS: 3-134, 136-305, 307-386, 388-474, 515-527, 532-588, 590, 591, 593-1049, and 1062-3086, and more particularly an amino acid sequence selected from SEQ ID NOS: 3-30, 32-72, 74-134, 136-178, 180-211, 218-239, 241-305, 307- 363, 366-386, 388-432, 434-474, 515-527, 532-588, 590, 591, 593-775, 777, 778, 780- 788, 790-1049, and 1062-3086. Any of these further comprising an optional linker moiety and a pharmaceutically acceptable
- composition comprising any of these polypeptides (with or without a covalently linked half-life extending moiety) and a pharmaceutically acceptable carrier is also encompassed within the present invention.
- Trp-GpTx-1 (1-34) ⁇ Amide ⁇ 106
- GpTx-1 (1-34)- ⁇ H ⁇ -DCLGFMRKCIPDNDKCCRPNLVCSRTHKWCKYVF- FreeAcid ⁇ FreeAcid ⁇ 107
- GpTx-1 (1-34)-Trp- DCLGFMRKCIPDNDKCCRPNLVCSRTHKWCKYVFW ⁇ Free FreeAcid Acid ⁇ 133
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Genetics & Genomics (AREA)
- Insects & Arthropods (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Gastroenterology & Hepatology (AREA)
- Toxicology (AREA)
- Immunology (AREA)
- Zoology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Tropical Medicine & Parasitology (AREA)
- Pain & Pain Management (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Rheumatology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
Claims
Priority Applications (18)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2013558223A JP2014509859A (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of 17NAV1.3 and NAV1.7 |
| AU2012228990A AU2012228990B2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of Nav1.3 and Nav1.7 |
| KR1020137027301A KR20140145947A (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| EA201391331A EA201391331A1 (en) | 2011-03-16 | 2012-03-16 | ACTIVE AND SELECTIVE INHIBITORS NAV1.3 AND NAV1.7 |
| MX2013010497A MX2013010497A (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7. |
| SG2013068770A SG193434A1 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| BR112013023674A BR112013023674A2 (en) | 2011-03-16 | 2012-03-16 | potent and selective inhibitors of nav1.3 and nav1.7 |
| EP12711312.4A EP2686340A2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| CA2830065A CA2830065A1 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| NZ615242A NZ615242B2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| US14/005,135 US9340590B2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of NaV1.3 and NaV1.7 |
| AP2013007173A AP2013007173A0 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of NAV1.3 and NAV1.7 |
| CN201280023655.XA CN103930437A (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of Nav1.3 and Nav1.7 |
| TNP2013000361A TN2013000361A1 (en) | 2012-03-07 | 2013-09-10 | Potent and selective inhibitors of nav1.3 and nav1.7 |
| ZA2013/06920A ZA201306920B (en) | 2011-03-16 | 2013-09-13 | POTENT AND SELECTIVE INHIBITORS OF NAv1.3 AND NAv1.7 |
| MA36308A MA35385B1 (en) | 2011-03-16 | 2013-10-08 | Strong and selective inhibitors of nav1.3 and nav1.7 |
| HK14111760.9A HK1198173A1 (en) | 2011-03-16 | 2014-11-21 | Potent and selective inhibitors of nav1.3 and nav1.7 nav13 nav17 |
| US15/134,305 US9796766B2 (en) | 2011-03-16 | 2016-04-20 | Potent and selective inhibitors of NAV1.3 and NAV1.7 |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161453492P | 2011-03-16 | 2011-03-16 | |
| US61/453,492 | 2011-03-16 | ||
| US201261608088P | 2012-03-07 | 2012-03-07 | |
| US61/608,088 | 2012-03-07 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/005,135 A-371-Of-International US9340590B2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of NaV1.3 and NaV1.7 |
| US15/134,305 Continuation US9796766B2 (en) | 2011-03-16 | 2016-04-20 | Potent and selective inhibitors of NAV1.3 and NAV1.7 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012125973A2 true WO2012125973A2 (en) | 2012-09-20 |
| WO2012125973A3 WO2012125973A3 (en) | 2013-01-03 |
Family
ID=45895479
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2012/029537 WO2012125973A2 (en) | 2011-03-16 | 2012-03-16 | Potent and selective inhibitors of nav1.3 and nav1.7 |
Country Status (23)
| Country | Link |
|---|---|
| US (2) | US9340590B2 (en) |
| EP (1) | EP2686340A2 (en) |
| JP (2) | JP2014509859A (en) |
| KR (1) | KR20140145947A (en) |
| CN (1) | CN103930437A (en) |
| AP (1) | AP2013007173A0 (en) |
| AR (1) | AR088413A1 (en) |
| AU (1) | AU2012228990B2 (en) |
| BR (1) | BR112013023674A2 (en) |
| CA (1) | CA2830065A1 (en) |
| CL (1) | CL2013002673A1 (en) |
| CO (1) | CO6821886A2 (en) |
| CR (1) | CR20130533A (en) |
| EA (1) | EA201391331A1 (en) |
| HK (1) | HK1198173A1 (en) |
| MA (1) | MA35385B1 (en) |
| MX (1) | MX2013010497A (en) |
| PE (1) | PE20140593A1 (en) |
| SG (2) | SG193434A1 (en) |
| TW (1) | TW201300407A (en) |
| UY (1) | UY33959A (en) |
| WO (1) | WO2012125973A2 (en) |
| ZA (1) | ZA201306920B (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014165277A2 (en) | 2013-03-12 | 2014-10-09 | Amgen Inc. | POTENT AND SELECTIVE INHIBITORS OF Nav1.7 |
| WO2014144911A3 (en) * | 2013-03-15 | 2014-12-04 | Capon Daniel J | Hybrid immunoglobulin containing non-peptidyl linkage |
| US9636418B2 (en) | 2013-03-12 | 2017-05-02 | Amgen Inc. | Potent and selective inhibitors of NAV1.7 |
| CN111187338A (en) * | 2014-06-12 | 2020-05-22 | Ra制药公司 | Modulation of complement activity |
| US11066459B2 (en) | 2014-03-14 | 2021-07-20 | Biomolecular Holdings Llc | Hybrid immunoglobulin containing non-peptidyl linkage |
| WO2022066767A2 (en) | 2020-09-23 | 2022-03-31 | Aldevron, Llc | Potent and selective inhibitors of nav1.7 |
| WO2023205468A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205778A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205465A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205463A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2024123815A1 (en) | 2022-12-06 | 2024-06-13 | Vertex Pharmaceuticals Incorporated | Process for the synthesis of substituted tetrahydrofuran modulators of sodium channels |
| EP4194562A4 (en) * | 2020-08-05 | 2024-08-14 | Aist | Method for screening for polypeptide that acts on target protein |
Families Citing this family (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105348392B (en) * | 2015-11-18 | 2019-08-02 | 北京华金瑞清生物医药技术有限公司 | A kind of Nav1.7 inhibitor and its remodeling method |
| US20170239183A1 (en) * | 2016-02-23 | 2017-08-24 | PixarBio Corporation | COMPOSITIONS COMPRISING NAv1.7 SELECTIVE INHIBITORS FOR TREATING ACUTE, POST-OPERATIVE, OR CHRONIC PAIN AND METHODS OF USING THE SAME |
| US20190218255A1 (en) * | 2016-09-16 | 2019-07-18 | Olipass Corporation | Scn9a antisense oligonucleotides |
| CN110536895B (en) * | 2017-01-24 | 2023-10-20 | 奥利通公司 | SCN9A antisense analgesic |
| CN109517041B (en) * | 2018-11-14 | 2019-09-17 | 青海芬陀利华生物科技有限公司 | Gptx-1 toxin and its application |
| CN113429463B (en) * | 2021-05-18 | 2022-08-23 | 湖南百尔泰克生物医药有限公司 | Polypeptide with analgesic effect and application thereof |
Citations (89)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3691016A (en) | 1970-04-17 | 1972-09-12 | Monsanto Co | Process for the preparation of insoluble enzymes |
| US3941763A (en) | 1975-03-28 | 1976-03-02 | American Home Products Corporation | PGlu-D-Met-Trp-Ser-Tyr-D-Ala-Leu-Arg-Pro-Gly-NH2 and intermediates |
| US3969287A (en) | 1972-12-08 | 1976-07-13 | Boehringer Mannheim Gmbh | Carrier-bound protein prepared by reacting the protein with an acylating or alkylating compound having a carrier-bonding group and reacting the product with a carrier |
| US4083368A (en) | 1976-09-01 | 1978-04-11 | Freezer Winthrop J | Inhaler |
| US4195128A (en) | 1976-05-03 | 1980-03-25 | Bayer Aktiengesellschaft | Polymeric carrier bound ligands |
| US4229537A (en) | 1978-02-09 | 1980-10-21 | New York University | Preparation of trichloro-s-triazine activated supports for coupling ligands |
| US4247642A (en) | 1977-02-17 | 1981-01-27 | Sumitomo Chemical Company, Limited | Enzyme immobilization with pullulan gel |
| US4330440A (en) | 1977-02-08 | 1982-05-18 | Development Finance Corporation Of New Zealand | Activated matrix and method of activation |
| US4351337A (en) | 1973-05-17 | 1982-09-28 | Arthur D. Little, Inc. | Biodegradable, implantable drug delivery device, and process for preparing and using the same |
| WO1988001649A1 (en) | 1986-09-02 | 1988-03-10 | Genex Corporation | Single polypeptide chain binding molecules |
| US4847325A (en) | 1988-01-20 | 1989-07-11 | Cetus Corporation | Conjugation of polymer to colony stimulating factor-1 |
| US4849224A (en) | 1987-11-12 | 1989-07-18 | Theratech Inc. | Device for administering an active agent to the skin or mucosa |
| US4906169A (en) | 1986-12-29 | 1990-03-06 | Rutgers, The State University Of New Jersey | Transdermal estrogen/progestin dosage unit, system and process |
| US4911916A (en) | 1986-12-22 | 1990-03-27 | Cygnus Research Corporation | Diffusion matrix for transdermal drug administration and transdermal drug delivery devices including same |
| US4925673A (en) | 1986-08-18 | 1990-05-15 | Clinical Technologies Associates, Inc. | Delivery systems for pharmacological agents encapsulated with proteinoids |
| US4925677A (en) | 1988-08-31 | 1990-05-15 | Theratech, Inc. | Biodegradable hydrogel matrices for the controlled release of pharmacologically active agents |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US4983395A (en) | 1987-11-12 | 1991-01-08 | Theratech Inc. | Device for administering an active agent to the skin or mucosa |
| US4994439A (en) | 1989-01-19 | 1991-02-19 | California Biotechnology Inc. | Transmembrane formulations for drug administration |
| US5002936A (en) | 1985-04-12 | 1991-03-26 | Seymour Lieberman | Lipophilic complexes of pharmacologically active inorganic mineral acid esters of organic compounds |
| US5013556A (en) | 1989-10-20 | 1991-05-07 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US5023084A (en) | 1986-12-29 | 1991-06-11 | Rutgers, The State University Of New Jersey | Transdermal estrogen/progestin dosage unit, system and process |
| US5166322A (en) | 1989-04-21 | 1992-11-24 | Genetics Institute | Cysteine added variants of interleukin-3 and chemical modifications thereof |
| US5171264A (en) | 1990-02-28 | 1992-12-15 | Massachusetts Institute Of Technology | Immobilized polyethylene oxide star molecules for bioapplications |
| US5206344A (en) | 1985-06-26 | 1993-04-27 | Cetus Oncology Corporation | Interleukin-2 muteins and polymer conjugation thereof |
| WO1993011161A1 (en) | 1991-11-25 | 1993-06-10 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US5252714A (en) | 1990-11-28 | 1993-10-12 | The University Of Alabama In Huntsville | Preparation and use of polyethylene glycol propionaldehyde |
| US5260203A (en) | 1986-09-02 | 1993-11-09 | Enzon, Inc. | Single polypeptide chain binding molecules |
| US5284656A (en) | 1991-03-15 | 1994-02-08 | Amgen Inc. | Pulmonary administration of granulocyte colony stimulating factor |
| US5302697A (en) | 1988-07-23 | 1994-04-12 | Delta Biotechnology Limited | Peptide and DNA sequences |
| US5346701A (en) | 1993-02-22 | 1994-09-13 | Theratech, Inc. | Transmucosal delivery of macromolecular drugs |
| WO1995013312A1 (en) | 1993-11-12 | 1995-05-18 | Shearwater Polymers, Inc. | Water soluble active sulfones of poly(ethylene glycol) |
| US5460820A (en) | 1993-08-03 | 1995-10-24 | Theratech, Inc. | Methods for providing testosterone and optionally estrogen replacement therapy to women |
| EP0473084B1 (en) | 1990-08-28 | 1995-11-15 | Sumitomo Pharmaceuticals Company, Limited | Polyethylene glycol derivatives, their modified peptides, methods for producing them and use of the modified peptides |
| WO1996011953A1 (en) | 1994-10-12 | 1996-04-25 | Amgen Inc. | N-terminally chemically modified protein compositions and methods |
| WO1996032478A1 (en) | 1995-04-14 | 1996-10-17 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US5605702A (en) | 1994-03-28 | 1997-02-25 | Laboratoires D'hygiene Et De Dietetique | Eva-based transdermal matrix system for the administration of an estrogen and/or a progestogen |
| US5643575A (en) | 1993-10-27 | 1997-07-01 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5662925A (en) | 1994-12-21 | 1997-09-02 | Theratech, Inc. | Transdermal delivery system with adhesive overlay and peel seal disc |
| WO1997034631A1 (en) | 1996-03-18 | 1997-09-25 | Board Of Regents, The University Of Texas System | Immunoglobin-like domains with increased half lives |
| US5714142A (en) | 1994-02-23 | 1998-02-03 | Blaney; Jeffrey M. | Method and compositions for increasing the serum half-life of pharmacologically active agents by binding to transthyretin-selective ligands |
| US5739277A (en) | 1995-04-14 | 1998-04-14 | Genentech Inc. | Altered polypeptides with increased half-life |
| US5770559A (en) | 1992-10-14 | 1998-06-23 | The Regents Of The University Of Colorado | Solubilization of pharmaceutical substances in an organic solvent and preparation of pharmaceutical powders using the same |
| US5783208A (en) | 1996-07-19 | 1998-07-21 | Theratech, Inc. | Transdermal drug delivery matrix for coadministering estradiol and another steroid |
| US5792451A (en) | 1994-03-02 | 1998-08-11 | Emisphere Technologies, Inc. | Oral drug delivery compositions and methods |
| US5919455A (en) | 1993-10-27 | 1999-07-06 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5932462A (en) | 1995-01-10 | 1999-08-03 | Shearwater Polymers, Inc. | Multiarmed, monofunctional, polymer for coupling to molecules and surfaces |
| US5990237A (en) | 1997-05-21 | 1999-11-23 | Shearwater Polymers, Inc. | Poly(ethylene glycol) aldehyde hydrates and related polymers and applications in modifying amines |
| US6022952A (en) | 1998-04-01 | 2000-02-08 | University Of Alberta | Compositions and methods for protein secretion |
| US6077680A (en) | 1996-11-27 | 2000-06-20 | The University Of Florida | ShK toxin compositions and methods of use |
| WO2000038652A1 (en) | 1998-12-23 | 2000-07-06 | Amgen Inc. | Polyol/oil suspensions for the sustained release of proteins |
| WO2000038651A1 (en) | 1998-12-23 | 2000-07-06 | Amgen Inc. | BIODEGRADABLE pH/THERMOSENSITIVE HYDROGELS FOR SUSTAINED DELIVERY OF BIOLOGICALLY ACTIVE AGENTS |
| US6273086B1 (en) | 1998-02-06 | 2001-08-14 | Unisia Jecs Corporation | Inhalant medicator |
| US6342225B1 (en) | 1993-08-13 | 2002-01-29 | Deutshces Wollforschungsinstitut | Pharmaceutical active conjugates |
| WO2002011801A1 (en) | 2000-08-04 | 2002-02-14 | The Technology Partnership Plc | Dry powder inhaler |
| US20020119946A1 (en) | 2001-02-28 | 2002-08-29 | Mg Pharmacy Ltd. | Formative agent of protein complex |
| US6451986B1 (en) | 1998-06-22 | 2002-09-17 | Immunex Corporation | Site specific protein modification |
| US20030069395A1 (en) | 2001-03-09 | 2003-04-10 | Sato Aaron K. | Serum albumin binding moieties |
| US6548644B1 (en) | 1997-03-10 | 2003-04-15 | Immunex Corporation | Site protected protein modification |
| US6552170B1 (en) | 1990-04-06 | 2003-04-22 | Amgen Inc. | PEGylation reagents and compounds formed therewith |
| EP0575545B1 (en) | 1991-03-15 | 2003-05-21 | Amgen Inc. | Pegylation of polypeptides |
| US20030104400A1 (en) | 1999-03-18 | 2003-06-05 | Human Genome Sciences, Inc. | 27 human secreted proteins |
| US6602498B2 (en) | 2000-02-22 | 2003-08-05 | Shearwater Corporation | N-maleimidyl polymer derivatives |
| US20030191056A1 (en) | 2002-04-04 | 2003-10-09 | Kenneth Walker | Use of transthyretin peptide/protein fusions to increase the serum half-life of pharmacologically active peptides/proteins |
| US6660843B1 (en) | 1998-10-23 | 2003-12-09 | Amgen Inc. | Modified peptides as therapeutic agents |
| US6689749B1 (en) | 1996-12-19 | 2004-02-10 | Suntory Limited | Neuropeptides originating in scorpion |
| WO2004017918A2 (en) | 2002-08-21 | 2004-03-04 | Norton Healthcare Ltd. | Method of preparing dry powder inhalation compositions |
| WO2004110472A2 (en) | 2003-06-12 | 2004-12-23 | Eli Lilly And Company | Fusion proteins |
| US20050054051A1 (en) | 2001-04-12 | 2005-03-10 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| US6887487B2 (en) | 2001-10-19 | 2005-05-03 | Idexx Laboratories, Inc. | Injectable compositions for the controlled delivery of pharmacologically active compound |
| US6887470B1 (en) | 1999-09-10 | 2005-05-03 | Conjuchem, Inc. | Protection of endogenous therapeutic peptides from peptidase activity through conjugation to blood components |
| US6892728B2 (en) | 2000-03-21 | 2005-05-17 | Astrazeneca Ab | Inhalation device |
| US6900317B2 (en) | 2002-02-19 | 2005-05-31 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Salts of the CGRP antagonist BIBN4096 and inhalable powdered medicaments containing them |
| US6926898B2 (en) | 2000-04-12 | 2005-08-09 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| US6932962B1 (en) | 1994-12-22 | 2005-08-23 | Astrazeneca Ab | Aerosol drug formulations containing hydrofluoroalkanes and alkyl saccharides |
| US20050187217A1 (en) | 2003-08-05 | 2005-08-25 | Wilson Dean M. | Compositions useful as inhibitors of voltage-gated ion channels |
| WO2005105057A1 (en) | 2004-04-23 | 2005-11-10 | Amgen Inc. | Sustained release formulations |
| WO2006000116A1 (en) | 2004-06-23 | 2006-01-05 | Huber+Suhner Ag | Broadband patch antenna |
| WO2006002850A2 (en) | 2004-06-25 | 2006-01-12 | Cellpep Sa | OsK1 DERIVATIVES |
| US7029909B1 (en) | 1998-11-20 | 2006-04-18 | Fuso Pharmaceutical Industries, Ltd. | Protein expression vector and utilization thereof |
| WO2006042151A2 (en) | 2004-10-07 | 2006-04-20 | The Regents Of The University Of California | Analogs of shk toxin and their uses in selective inhibition of kv1.3 potassium channels |
| US20060199812A1 (en) | 2005-01-24 | 2006-09-07 | Amgen Inc. | Method of conjugating aminothiol containing molecules to vehicles |
| WO2006116156A2 (en) | 2005-04-22 | 2006-11-02 | Amgen Inc. | Toxin peptides with extended blood halflife |
| WO2007109324A2 (en) | 2006-03-21 | 2007-09-27 | Xenon Pharmaceuticals, Inc. | Potent and selective nav 1.7 sodium channel blockers |
| WO2008088422A2 (en) | 2006-10-25 | 2008-07-24 | Amgen Inc. | Toxin peptide therapeutic agents |
| US7442778B2 (en) | 2004-09-24 | 2008-10-28 | Amgen Inc. | Modified Fc molecules |
| WO2009033027A2 (en) | 2007-09-05 | 2009-03-12 | Medtronic, Inc. | Suppression of scn9a gene expression and/or function for the treatment of pain |
| WO2010108154A2 (en) | 2009-03-20 | 2010-09-23 | Amgen Inc. | Selective and potent peptide inhibitors of kv1.3 |
Family Cites Families (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH0640945A (en) | 1992-07-23 | 1994-02-15 | Kureha Chem Ind Co Ltd | Fc fragment binding antitumor agent |
| US5514774A (en) | 1993-06-29 | 1996-05-07 | University Of Utah Research Foundation | Conotoxin peptides |
| US5432155A (en) | 1993-06-29 | 1995-07-11 | The Salk Institute For Biological Studies | Conotoxins I |
| US5776896A (en) | 1996-01-03 | 1998-07-07 | Zeneca Limited | Analgesic peptides from venom of grammostola spatulata and use thereof |
| US5756663A (en) | 1996-01-03 | 1998-05-26 | Zeneca Limited | Antiarrhythmic peptide from venom of spider Grammostola spatulata |
| AUPP589598A0 (en) | 1998-09-14 | 1998-10-08 | University Of Queensland, The | Novel peptides |
| WO2001038564A2 (en) | 1999-11-26 | 2001-05-31 | Mcgill University | Loci for idiopathic generalized epilepsy, mutations thereof and method using same to assess, diagnose, prognose or treat epilepsy |
| CA2393616A1 (en) | 2000-01-31 | 2001-08-02 | Human Genome Sciences, Inc. | Nucleic acids, proteins, and antibodies |
| US7125847B1 (en) | 2000-04-07 | 2006-10-24 | The Research Foundation Of State University Of New York At Buffalo | Mechanically activated channel blocker |
| CN1133461C (en) | 2000-04-11 | 2004-01-07 | 厦门北大之路生物工程有限公司 | Application of tigroid spider toxic extract in preparing analgesic |
| EP1280895B1 (en) | 2000-05-10 | 2005-08-03 | Centre National De La Recherche Scientifique (Cnrs) | Polypeptide inhibiting a proton-gated na+ channel |
| GB0021617D0 (en) | 2000-09-02 | 2000-10-18 | Imp College Innovations Ltd | Diagnosis and treatment of cancer |
| US7393657B2 (en) | 2001-04-12 | 2008-07-01 | Imperial College Innovations Limited | Diagnosis and treatment of cancer: I |
| GB0120238D0 (en) | 2001-08-20 | 2001-10-10 | Univ College Of London | Sodium channel regulators and modulators |
| CA2474751A1 (en) | 2002-02-25 | 2003-09-04 | Vanderbilt University | Expression system for human brain-specific voltage-gated sodium channel, type 1 |
| DK2226316T3 (en) | 2002-05-30 | 2016-04-11 | Scripps Research Inst | Copper catalyzed ligation of azides and acetylenes |
| WO2004085647A1 (en) | 2003-03-26 | 2004-10-07 | Pharmadesign, Inc. | Low-molecular weight peptides inhibiting ion channel activity |
| CA2539227A1 (en) | 2003-08-08 | 2005-02-17 | Vertex Pharmaceuticals Incorporated | Heteroarylaminosulfonylphenyl derivatives for use as sodium or calcium channel blockers in the treatment of pain |
| US7615563B2 (en) | 2003-08-08 | 2009-11-10 | Gonzalez Iii Jesus E | Compositions useful as inhibitors of voltage-gated sodium channels |
| US7060723B2 (en) | 2003-08-29 | 2006-06-13 | Allergan, Inc. | Treating neurological disorders using selective antagonists of persistent sodium current |
| US7125908B2 (en) | 2003-08-29 | 2006-10-24 | Allergan, Inc. | Treating pain using selective antagonists of persistent sodium current |
| JP2007518422A (en) | 2004-01-21 | 2007-07-12 | ユニバーシティ・オブ・ユタ・リサーチ・ファウンデーション | Mutant sodium channel Naν1.7 and methods related thereto |
| WO2005118614A1 (en) | 2004-04-08 | 2005-12-15 | Avigen, Inc. | Methods and compositions for treating neuropathic pain |
| US7259145B2 (en) | 2004-07-07 | 2007-08-21 | The Research Foundation Of State University Of New York | Mechanically activated channel blocker |
| JP2008519034A (en) | 2004-11-03 | 2008-06-05 | バーテックス ファーマシューティカルズ インコーポレイテッド | Pyrimidine derivatives as ion channel modulators and methods of use |
| WO2006101629A2 (en) | 2005-02-17 | 2006-09-28 | Vertex Pharmaceuticals Incorporated | SODIUM CHANNEL PROTEIN TYPE III α-SUBUNIT SPLICE VARIANT |
| GB0517487D0 (en) | 2005-08-26 | 2005-10-05 | Isis Innovation | Antibodies |
| US7972813B2 (en) | 2005-09-30 | 2011-07-05 | Vertex Pharmaceuticals Incorporated | Tetrodotoxin-resistant sodium channel alpha subunit |
| KR20140130524A (en) | 2005-11-08 | 2014-11-10 | 애더리스 래버러토리즈 | Mu-conotoxin peptides and use thereof as a local anesthetic |
| JP4742345B2 (en) * | 2006-06-20 | 2011-08-10 | 独立行政法人産業技術総合研究所 | Polypeptide derived from Gramostra spatula that blocks calcium channel and gene thereof |
| EP2628749A3 (en) | 2006-06-20 | 2013-12-04 | Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center | Antimicrobial kinocidin compostions and methods of use |
| CN101480393A (en) * | 2008-01-07 | 2009-07-15 | 云南昊邦制药有限公司 | Application of bulleyaconitine A in preparing medicament for treating Nav1.7 ache disease |
| EP2249856B1 (en) | 2008-01-30 | 2013-11-06 | Baylor College Of Medicine | Peptides that target dorsal root ganglion neurons |
| CA2751159A1 (en) | 2009-02-02 | 2010-08-05 | Chromocell Corporation | Cell lines expressing nav and methods of using them |
| WO2010104115A1 (en) * | 2009-03-10 | 2010-09-16 | 独立行政法人産業技術総合研究所 | Method for preparing polypeptide specifically recognizing membrane protein |
| JP5717143B2 (en) * | 2009-03-10 | 2015-05-13 | 独立行政法人産業技術総合研究所 | Methods for preparing polypeptide libraries |
| BR112012005860A2 (en) * | 2009-09-15 | 2017-01-31 | Alomone Preclinical Ltd | peptides isolated from spider venom, and uses thereof |
| GB0922434D0 (en) | 2009-12-22 | 2010-02-03 | Ucb Pharma Sa | antibodies and fragments thereof |
| GB0922435D0 (en) | 2009-12-22 | 2010-02-03 | Ucb Pharma Sa | Method |
| JP6095368B2 (en) | 2009-10-27 | 2017-03-15 | ユセベ ファルマ ソシエテ アノニム | Nav1.7 antibody whose function is modified |
| US8871996B2 (en) | 2010-06-09 | 2014-10-28 | Regeneron Pharmaceuticals, Inc. | Mice expressing human voltage-gated sodium channels |
| US9279003B2 (en) | 2010-07-07 | 2016-03-08 | Purdue Pharma L.P. | Analogs of sodium channel peptide toxin |
| CN110041420A (en) | 2012-05-18 | 2019-07-23 | 詹森生物科技公司 | Bird catching arachinid toxin I V variant and its application method |
-
2012
- 2012-03-16 CA CA2830065A patent/CA2830065A1/en not_active Abandoned
- 2012-03-16 AP AP2013007173A patent/AP2013007173A0/en unknown
- 2012-03-16 UY UY0001033959A patent/UY33959A/en not_active Application Discontinuation
- 2012-03-16 SG SG2013068770A patent/SG193434A1/en unknown
- 2012-03-16 TW TW101109226A patent/TW201300407A/en unknown
- 2012-03-16 EP EP12711312.4A patent/EP2686340A2/en not_active Withdrawn
- 2012-03-16 PE PE2013002050A patent/PE20140593A1/en not_active Application Discontinuation
- 2012-03-16 AU AU2012228990A patent/AU2012228990B2/en active Active
- 2012-03-16 WO PCT/US2012/029537 patent/WO2012125973A2/en active Application Filing
- 2012-03-16 JP JP2013558223A patent/JP2014509859A/en active Pending
- 2012-03-16 EA EA201391331A patent/EA201391331A1/en unknown
- 2012-03-16 BR BR112013023674A patent/BR112013023674A2/en not_active IP Right Cessation
- 2012-03-16 KR KR1020137027301A patent/KR20140145947A/en not_active Application Discontinuation
- 2012-03-16 SG SG10201601789TA patent/SG10201601789TA/en unknown
- 2012-03-16 MX MX2013010497A patent/MX2013010497A/en unknown
- 2012-03-16 CN CN201280023655.XA patent/CN103930437A/en active Pending
- 2012-03-16 US US14/005,135 patent/US9340590B2/en active Active
- 2012-03-19 AR ARP120100896A patent/AR088413A1/en unknown
-
2013
- 2013-09-13 ZA ZA2013/06920A patent/ZA201306920B/en unknown
- 2013-09-16 CL CL2013002673A patent/CL2013002673A1/en unknown
- 2013-10-08 MA MA36308A patent/MA35385B1/en unknown
- 2013-10-11 CO CO13242679A patent/CO6821886A2/en unknown
- 2013-10-16 CR CR20130533A patent/CR20130533A/en unknown
-
2014
- 2014-11-21 HK HK14111760.9A patent/HK1198173A1/en unknown
-
2016
- 2016-04-20 US US15/134,305 patent/US9796766B2/en active Active
- 2016-08-22 JP JP2016161682A patent/JP2017031157A/en active Pending
Patent Citations (114)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3691016A (en) | 1970-04-17 | 1972-09-12 | Monsanto Co | Process for the preparation of insoluble enzymes |
| US3969287A (en) | 1972-12-08 | 1976-07-13 | Boehringer Mannheim Gmbh | Carrier-bound protein prepared by reacting the protein with an acylating or alkylating compound having a carrier-bonding group and reacting the product with a carrier |
| US4351337A (en) | 1973-05-17 | 1982-09-28 | Arthur D. Little, Inc. | Biodegradable, implantable drug delivery device, and process for preparing and using the same |
| US3941763A (en) | 1975-03-28 | 1976-03-02 | American Home Products Corporation | PGlu-D-Met-Trp-Ser-Tyr-D-Ala-Leu-Arg-Pro-Gly-NH2 and intermediates |
| US4195128A (en) | 1976-05-03 | 1980-03-25 | Bayer Aktiengesellschaft | Polymeric carrier bound ligands |
| US4083368A (en) | 1976-09-01 | 1978-04-11 | Freezer Winthrop J | Inhaler |
| US4330440A (en) | 1977-02-08 | 1982-05-18 | Development Finance Corporation Of New Zealand | Activated matrix and method of activation |
| US4247642A (en) | 1977-02-17 | 1981-01-27 | Sumitomo Chemical Company, Limited | Enzyme immobilization with pullulan gel |
| US4229537A (en) | 1978-02-09 | 1980-10-21 | New York University | Preparation of trichloro-s-triazine activated supports for coupling ligands |
| US5002936A (en) | 1985-04-12 | 1991-03-26 | Seymour Lieberman | Lipophilic complexes of pharmacologically active inorganic mineral acid esters of organic compounds |
| US5206344A (en) | 1985-06-26 | 1993-04-27 | Cetus Oncology Corporation | Interleukin-2 muteins and polymer conjugation thereof |
| US4925673A (en) | 1986-08-18 | 1990-05-15 | Clinical Technologies Associates, Inc. | Delivery systems for pharmacological agents encapsulated with proteinoids |
| WO1988001649A1 (en) | 1986-09-02 | 1988-03-10 | Genex Corporation | Single polypeptide chain binding molecules |
| US5260203A (en) | 1986-09-02 | 1993-11-09 | Enzon, Inc. | Single polypeptide chain binding molecules |
| US4911916A (en) | 1986-12-22 | 1990-03-27 | Cygnus Research Corporation | Diffusion matrix for transdermal drug administration and transdermal drug delivery devices including same |
| US4906169A (en) | 1986-12-29 | 1990-03-06 | Rutgers, The State University Of New Jersey | Transdermal estrogen/progestin dosage unit, system and process |
| US5023084A (en) | 1986-12-29 | 1991-06-11 | Rutgers, The State University Of New Jersey | Transdermal estrogen/progestin dosage unit, system and process |
| US4946778A (en) | 1987-09-21 | 1990-08-07 | Genex Corporation | Single polypeptide chain binding molecules |
| US4849224A (en) | 1987-11-12 | 1989-07-18 | Theratech Inc. | Device for administering an active agent to the skin or mucosa |
| US4983395A (en) | 1987-11-12 | 1991-01-08 | Theratech Inc. | Device for administering an active agent to the skin or mucosa |
| US4847325A (en) | 1988-01-20 | 1989-07-11 | Cetus Corporation | Conjugation of polymer to colony stimulating factor-1 |
| US5302697A (en) | 1988-07-23 | 1994-04-12 | Delta Biotechnology Limited | Peptide and DNA sequences |
| US4925677A (en) | 1988-08-31 | 1990-05-15 | Theratech, Inc. | Biodegradable hydrogel matrices for the controlled release of pharmacologically active agents |
| US4994439A (en) | 1989-01-19 | 1991-02-19 | California Biotechnology Inc. | Transmembrane formulations for drug administration |
| US5166322A (en) | 1989-04-21 | 1992-11-24 | Genetics Institute | Cysteine added variants of interleukin-3 and chemical modifications thereof |
| EP0668354A1 (en) | 1989-04-21 | 1995-08-23 | Genetics Institute, Inc. | Cysteine added variants of G-CSF and chemical modifications thereof |
| EP0668353A1 (en) | 1989-04-21 | 1995-08-23 | Genetics Institute, Inc. | Cysteine added variants of EPO and chemical modifications thereof |
| EP0469074B1 (en) | 1989-04-21 | 1996-07-31 | Genetics Institute, Inc. | Cysteine added variants il-3 and chemical modifications thereof |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US5013556A (en) | 1989-10-20 | 1991-05-07 | Liposome Technology, Inc. | Liposomes with enhanced circulation time |
| US5171264A (en) | 1990-02-28 | 1992-12-15 | Massachusetts Institute Of Technology | Immobilized polyethylene oxide star molecules for bioapplications |
| US6552170B1 (en) | 1990-04-06 | 2003-04-22 | Amgen Inc. | PEGylation reagents and compounds formed therewith |
| EP0473084B1 (en) | 1990-08-28 | 1995-11-15 | Sumitomo Pharmaceuticals Company, Limited | Polyethylene glycol derivatives, their modified peptides, methods for producing them and use of the modified peptides |
| US5252714A (en) | 1990-11-28 | 1993-10-12 | The University Of Alabama In Huntsville | Preparation and use of polyethylene glycol propionaldehyde |
| US5284656A (en) | 1991-03-15 | 1994-02-08 | Amgen Inc. | Pulmonary administration of granulocyte colony stimulating factor |
| EP0575545B1 (en) | 1991-03-15 | 2003-05-21 | Amgen Inc. | Pegylation of polypeptides |
| WO1993011161A1 (en) | 1991-11-25 | 1993-06-10 | Enzon, Inc. | Multivalent antigen-binding proteins |
| US5981474A (en) | 1992-10-14 | 1999-11-09 | University Technology Corporation | Solubilization of pharmaceutical substances in an organic solvent and preparation of pharmaceutical powders using the same |
| US5770559A (en) | 1992-10-14 | 1998-06-23 | The Regents Of The University Of Colorado | Solubilization of pharmaceutical substances in an organic solvent and preparation of pharmaceutical powders using the same |
| US5516523A (en) | 1993-02-22 | 1996-05-14 | Theratech, Inc. | Transmucosal delivery of macromolecular drugs |
| US5346701A (en) | 1993-02-22 | 1994-09-13 | Theratech, Inc. | Transmucosal delivery of macromolecular drugs |
| US5460820B1 (en) | 1993-08-03 | 1999-08-03 | Theratech Inc | Method for providing testosterone and optionally estrogen replacement therapy to women |
| US5460820A (en) | 1993-08-03 | 1995-10-24 | Theratech, Inc. | Methods for providing testosterone and optionally estrogen replacement therapy to women |
| US6342225B1 (en) | 1993-08-13 | 2002-01-29 | Deutshces Wollforschungsinstitut | Pharmaceutical active conjugates |
| US5643575A (en) | 1993-10-27 | 1997-07-01 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5919455A (en) | 1993-10-27 | 1999-07-06 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5900461A (en) | 1993-11-12 | 1999-05-04 | Shearwater Polymers, Inc. | Isolatable, water soluble, and hydrolytically stable active sulfones of poly(ethylene glycol) and related polymers for modification of surfaces and molecules |
| US6894025B2 (en) | 1993-11-12 | 2005-05-17 | Nektar Therapeutics Al, Corp. | Biologically active molecules having thiol moiety conjugated to polymers containing ethyl sulfone moiety |
| US5446090A (en) | 1993-11-12 | 1995-08-29 | Shearwater Polymers, Inc. | Isolatable, water soluble, and hydrolytically stable active sulfones of poly(ethylene glycol) and related polymers for modification of surfaces and molecules |
| US5739208A (en) | 1993-11-12 | 1998-04-14 | Shearwater Polymers, Inc. | Isolatable, water soluble, and hydrolytically stable active sulfones of poly(ethylene glycol) and related polymers for modification of surfaces and molecules |
| WO1995013312A1 (en) | 1993-11-12 | 1995-05-18 | Shearwater Polymers, Inc. | Water soluble active sulfones of poly(ethylene glycol) |
| US6610281B2 (en) | 1993-11-12 | 2003-08-26 | Shearwater Corporation | Water soluble activated polymers containing an active ethyl sulfone moiety for modification of surfaces and molecules |
| US5714142A (en) | 1994-02-23 | 1998-02-03 | Blaney; Jeffrey M. | Method and compositions for increasing the serum half-life of pharmacologically active agents by binding to transthyretin-selective ligands |
| US5792451A (en) | 1994-03-02 | 1998-08-11 | Emisphere Technologies, Inc. | Oral drug delivery compositions and methods |
| US5605702A (en) | 1994-03-28 | 1997-02-25 | Laboratoires D'hygiene Et De Dietetique | Eva-based transdermal matrix system for the administration of an estrogen and/or a progestogen |
| US5824784A (en) | 1994-10-12 | 1998-10-20 | Amgen Inc. | N-terminally chemically modified protein compositions and methods |
| WO1996011953A1 (en) | 1994-10-12 | 1996-04-25 | Amgen Inc. | N-terminally chemically modified protein compositions and methods |
| US5985265A (en) | 1994-10-12 | 1999-11-16 | Amgen Inc. | N-terminally chemically modified protein compositions and methods |
| US5662925A (en) | 1994-12-21 | 1997-09-02 | Theratech, Inc. | Transdermal delivery system with adhesive overlay and peel seal disc |
| US6932962B1 (en) | 1994-12-22 | 2005-08-23 | Astrazeneca Ab | Aerosol drug formulations containing hydrofluoroalkanes and alkyl saccharides |
| US5932462A (en) | 1995-01-10 | 1999-08-03 | Shearwater Polymers, Inc. | Multiarmed, monofunctional, polymer for coupling to molecules and surfaces |
| WO1996032478A1 (en) | 1995-04-14 | 1996-10-17 | Genentech, Inc. | Altered polypeptides with increased half-life |
| US5739277A (en) | 1995-04-14 | 1998-04-14 | Genentech Inc. | Altered polypeptides with increased half-life |
| WO1997034631A1 (en) | 1996-03-18 | 1997-09-25 | Board Of Regents, The University Of Texas System | Immunoglobin-like domains with increased half lives |
| US5783208A (en) | 1996-07-19 | 1998-07-21 | Theratech, Inc. | Transdermal drug delivery matrix for coadministering estradiol and another steroid |
| US6077680A (en) | 1996-11-27 | 2000-06-20 | The University Of Florida | ShK toxin compositions and methods of use |
| US6689749B1 (en) | 1996-12-19 | 2004-02-10 | Suntory Limited | Neuropeptides originating in scorpion |
| US6548644B1 (en) | 1997-03-10 | 2003-04-15 | Immunex Corporation | Site protected protein modification |
| US5990237A (en) | 1997-05-21 | 1999-11-23 | Shearwater Polymers, Inc. | Poly(ethylene glycol) aldehyde hydrates and related polymers and applications in modifying amines |
| US6273086B1 (en) | 1998-02-06 | 2001-08-14 | Unisia Jecs Corporation | Inhalant medicator |
| US6335178B1 (en) | 1998-04-01 | 2002-01-01 | University Of Alberta | Compositions and methods for protein secretion |
| US6022952A (en) | 1998-04-01 | 2000-02-08 | University Of Alberta | Compositions and methods for protein secretion |
| US6451986B1 (en) | 1998-06-22 | 2002-09-17 | Immunex Corporation | Site specific protein modification |
| US6660843B1 (en) | 1998-10-23 | 2003-12-09 | Amgen Inc. | Modified peptides as therapeutic agents |
| US7029909B1 (en) | 1998-11-20 | 2006-04-18 | Fuso Pharmaceutical Industries, Ltd. | Protein expression vector and utilization thereof |
| US6245740B1 (en) | 1998-12-23 | 2001-06-12 | Amgen Inc. | Polyol:oil suspensions for the sustained release of proteins |
| WO2000038652A1 (en) | 1998-12-23 | 2000-07-06 | Amgen Inc. | Polyol/oil suspensions for the sustained release of proteins |
| WO2000038651A1 (en) | 1998-12-23 | 2000-07-06 | Amgen Inc. | BIODEGRADABLE pH/THERMOSENSITIVE HYDROGELS FOR SUSTAINED DELIVERY OF BIOLOGICALLY ACTIVE AGENTS |
| US20030104400A1 (en) | 1999-03-18 | 2003-06-05 | Human Genome Sciences, Inc. | 27 human secreted proteins |
| US6887470B1 (en) | 1999-09-10 | 2005-05-03 | Conjuchem, Inc. | Protection of endogenous therapeutic peptides from peptidase activity through conjugation to blood components |
| US6602498B2 (en) | 2000-02-22 | 2003-08-05 | Shearwater Corporation | N-maleimidyl polymer derivatives |
| US6892728B2 (en) | 2000-03-21 | 2005-05-17 | Astrazeneca Ab | Inhalation device |
| US6926898B2 (en) | 2000-04-12 | 2005-08-09 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| WO2002011801A1 (en) | 2000-08-04 | 2002-02-14 | The Technology Partnership Plc | Dry powder inhaler |
| US20020119946A1 (en) | 2001-02-28 | 2002-08-29 | Mg Pharmacy Ltd. | Formative agent of protein complex |
| US20030069395A1 (en) | 2001-03-09 | 2003-04-10 | Sato Aaron K. | Serum albumin binding moieties |
| US20050054051A1 (en) | 2001-04-12 | 2005-03-10 | Human Genome Sciences, Inc. | Albumin fusion proteins |
| US6887487B2 (en) | 2001-10-19 | 2005-05-03 | Idexx Laboratories, Inc. | Injectable compositions for the controlled delivery of pharmacologically active compound |
| US6900317B2 (en) | 2002-02-19 | 2005-05-31 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Salts of the CGRP antagonist BIBN4096 and inhalable powdered medicaments containing them |
| US20030191056A1 (en) | 2002-04-04 | 2003-10-09 | Kenneth Walker | Use of transthyretin peptide/protein fusions to increase the serum half-life of pharmacologically active peptides/proteins |
| US20030195154A1 (en) | 2002-04-04 | 2003-10-16 | Kenneth Walker | Use of transthyretin peptide/protein fusions to increase the serum half-life of pharmacologically active peptides/proteins |
| WO2004017918A2 (en) | 2002-08-21 | 2004-03-04 | Norton Healthcare Ltd. | Method of preparing dry powder inhalation compositions |
| WO2004110472A2 (en) | 2003-06-12 | 2004-12-23 | Eli Lilly And Company | Fusion proteins |
| US20050187217A1 (en) | 2003-08-05 | 2005-08-25 | Wilson Dean M. | Compositions useful as inhibitors of voltage-gated ion channels |
| WO2005105057A1 (en) | 2004-04-23 | 2005-11-10 | Amgen Inc. | Sustained release formulations |
| WO2006000116A1 (en) | 2004-06-23 | 2006-01-05 | Huber+Suhner Ag | Broadband patch antenna |
| WO2006002850A2 (en) | 2004-06-25 | 2006-01-12 | Cellpep Sa | OsK1 DERIVATIVES |
| US7662931B2 (en) | 2004-09-24 | 2010-02-16 | Amgen Inc. | Modified Fc molecules |
| US7750127B2 (en) | 2004-09-24 | 2010-07-06 | Amgen Inc. | Modified Fc molecules |
| US7750128B2 (en) | 2004-09-24 | 2010-07-06 | Amgen Inc. | Modified Fc molecules |
| US7655765B2 (en) | 2004-09-24 | 2010-02-02 | Amgen Inc. | Modified Fc molecules |
| US7655764B2 (en) | 2004-09-24 | 2010-02-02 | Amgen Inc. | Modified Fc molecules |
| US7645861B2 (en) | 2004-09-24 | 2010-01-12 | Amgen Inc. | Modified Fc molecules |
| US7442778B2 (en) | 2004-09-24 | 2008-10-28 | Amgen Inc. | Modified Fc molecules |
| WO2006042151A2 (en) | 2004-10-07 | 2006-04-20 | The Regents Of The University Of California | Analogs of shk toxin and their uses in selective inhibition of kv1.3 potassium channels |
| US20060199812A1 (en) | 2005-01-24 | 2006-09-07 | Amgen Inc. | Method of conjugating aminothiol containing molecules to vehicles |
| US20070071764A1 (en) | 2005-04-22 | 2007-03-29 | Sullivan John K | Toxin peptide therapeutic agents |
| WO2006116156A2 (en) | 2005-04-22 | 2006-11-02 | Amgen Inc. | Toxin peptides with extended blood halflife |
| US7833979B2 (en) | 2005-04-22 | 2010-11-16 | Amgen Inc. | Toxin peptide therapeutic agents |
| WO2007109324A2 (en) | 2006-03-21 | 2007-09-27 | Xenon Pharmaceuticals, Inc. | Potent and selective nav 1.7 sodium channel blockers |
| WO2008088422A2 (en) | 2006-10-25 | 2008-07-24 | Amgen Inc. | Toxin peptide therapeutic agents |
| WO2009033027A2 (en) | 2007-09-05 | 2009-03-12 | Medtronic, Inc. | Suppression of scn9a gene expression and/or function for the treatment of pain |
| WO2010108154A2 (en) | 2009-03-20 | 2010-09-23 | Amgen Inc. | Selective and potent peptide inhibitors of kv1.3 |
| WO2010108153A2 (en) | 2009-03-20 | 2010-09-23 | Amgen Inc. | Carrier immunoglobulins and uses thereof |
Non-Patent Citations (173)
| Title |
|---|
| "Biochemical Nomenclature and Related Documents, 2nd edition,", 1992, PORTLAND PRESS, pages: 39 - 69 |
| "Fmoc Solid Phase Peptide Synthesis, A Practical Approach", 2000, OXFORD PRESS |
| "Remington's Pharmaceutical Sciences", 1990 |
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING CO., pages: 1435 - 1712 |
| "Sequence Analysis Primer", 1991, M. STOCKTON PRESS |
| (PAUL, W.,: "Fundamental Immunology, 2nd ed.", 1989, RAVEN PRESS, N.Y. |
| A.M. FELIX ET AL.: "PEGylated Peptides IV: Enhanced biological activity of site-directed PEGylated GRF analogs", INT. J. PEPTIDE PROTEIN RES., vol. 46, 1995, pages 253 - 264 |
| A.M. FELIX: "Site-specific poly(ethylene glycol)ylation of peptides", vol. 680, 1997, ACS SYMPOSIUM SERIES, pages: 218 - 238 |
| ABUCHOWSKI; DAVIS: "Soluble Polymer-Enzyme Adducts, Enzymes as Drugs", 1981, WILEY-INTERSCIENCE, NEW YORK, pages: 367 - 83 |
| ADAMS, DRUG DEVELOP. RES., vol. 46, 1999, pages 219 - 34 |
| ADJEI ET AL., INTERNATL. J. PHARMACEUTICS, vol. 63, 1990, pages 135 - 44 |
| ADJEI ET AL., PHARMA. RES., vol. 7, 1990, pages 565 - 9 |
| AHMAD ET AL., HUM MOL GENET, vol. 16, 2007, pages 2114 - 2121 |
| AHMAD ET AL.: "A stop codon mutation in SCN9A causes lack of pain sensation", HUM. MOL. GENET., vol. 16, no. 17, 2007, pages 2114 - 21 |
| ALESSANDRI-HABER ET AL.: "Mapping the functional anatomy of BgK on Kvl .1, Kvl .2, and Kvl.3", J. BIOL. CHCM., vol. 274, no. 50, 1999, pages 35653 - 61 |
| AMINO ACIDS AND PEPTIDES, vol. 16, 1985, pages 387 - 410 |
| BACKONJA, M. M.: "Use of anticonvulsants for treatment of neuropathic pain", NEUROLOGY, vol. 59, no. 5, 2002, pages 14 - 7 |
| BARRETT A. ET AL.: "Handbook of proteolytic enzymes", 1998, ACADEMIC PRESS |
| BEETON ET AL.: "Targeting effector memory T cells with a selective peptide inhibitor of Kvl .3 channels for therapy of autoimmune diseases", MOLEC. PHARMACOL., vol. 67, no. 4, 2005, pages 1369 - 81 |
| BEETON ET AL.: "Targeting effector memory T cells with a selective peptide inhibitor of Kvl .3 channnels for therapy of autoimmune diseases", MOLEC. PHARMACOL., vol. 67, no. 4, 2005, pages 1369 - 81 |
| BHATNAGAR ET AL., J. MED. CHEM., vol. 39, 1996, pages 3814 - 3819 |
| BILLEN ET AL.: "Animal peptides targeting voltage-activated sodium channels", CUR. PHARM. DES., vol. 14, 2008, pages 2492 - 2502 |
| BIOCHEM. J., vol. 219, 1984, pages 345 - 373 |
| BIRD ET AL., SCIENCE, vol. 242, 1988, pages 423 - 426 |
| BRAQUET ET AL., J. CARDIOVASC. PHARMACOL., vol. 13, no. 5, 1989, pages 143 - 146 |
| BULAJ ET AL.: "Novel conotoxins from Conus striatus and Conus kinoshitai selectively block TTX-resistant sodium channels", BIOCHEM., vol. 44, no. 19, 2005, pages 7259 - 7265 |
| C. DELGADO ET AL.: "The uses and properties of PEG-linked proteins", CRIT. REV. THERAP. DRUG CARRIER SYSTEMS, vol. 9, 1992, pages 249 - 304 |
| CARILLO ET AL., SIAM J. APPLIED MATH., vol. 48, 1988, pages 1073 |
| CHEN ET AL.: "Antinociceptive effects of intrathecally administered huwentoxin-I, a selective N-type calcium channel blocker, in the formalin test in conscious rats", TOXICON, vol. 45, no. 1, 2005, pages 15 - 20 |
| CHOTHIA ET AL., J. MOL.BIOL., vol. 196, 1987, pages 901 - 917 |
| CHOTHIA ET AL., NATURE, vol. 342, 1989, pages 878 - 883 |
| CHOTHIA; LESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| CHU ET AL., GENE, vol. 13, 1981, pages 197 |
| CONNELLY, JC ET AL.: "Neutral Endopeptidase 24.11 in Human Neutrophils: Cleavage of Chemotactic Peptide", PNAS, vol. 82, no. 24, 1985, pages 8737 - 8741 |
| COX ET AL., NATURE, vol. 444, 2006, pages 894 - 898 |
| CREIGHTON: "Proteins: Structure and Molecule Properties", 1983, W. H. FREEMAN & CO., SAN FRANCISCO, pages: 79 - 86 |
| DAUPLAIS ET AL.: "On the convergent evolution of animal toxins: conservation of a diad of functional residues in potassium channel-blocking toxins with unrelated structures", J. BIOL. CHEM., vol. 272, no. 7, 1997, pages 4302 - 09 |
| DAVIS ET AL., BIOCHEM. INTL., vol. 10, 1985, pages 394 - 414 |
| DAVIS ET AL.: "Basic Methods in Molecular Biology", 1986, ELSEVIER |
| DAYHOFF ET AL., ATLAS OF PROTEIN SEQUENCE AND STRUCTURE, vol. 5, no. 3, 1978 |
| DAYHOFF ET AL.: "Atlas of Protein Sequence and Structure", vol. 5, 1978, pages: 345 - 352 |
| DEBS ET AL., J. IMMUNOL., vol. 140, 1988, pages 3482 - 8 |
| DEL RIO-PORTILLO ET AL.: "NMR solution structure of Cnl2, a novel peptide from the Mexican scorpion Centruroides noxius with a typical beta-toxin sequence but with alpha-like physiological activity", EUR. J. BIOCHEM., vol. 271, no. 12, 2004, pages 2504 - 16 |
| DEVEREUX ET AL., NUCL. ACID RES., vol. 12, 1984, pages 387 |
| DISS ET AL.: "A potential novel marker for human prostate cancer: voltage-gated sodium channel expression in vivo", PROSTATE CANCER AND PROSTATIC DISEASES, vol. 8, 2005, pages 266 - 73 |
| EARNSHAW WC ET AL.: "Mammalian caspases: Structure, activation, substrates, and functions during apoptosis", ANNUAL REVIEW OF BIOCHEMISTRY, vol. 68, 1999, pages 383 - 424 |
| EDGERTON ET AL.: "Evidence for multiple effects of ProTxII on activation gating in NaV 1.5", TOXICON, vol. 52, no. 3, 2008, pages 489 - 500 |
| EDGERTON ET AL.: "Inhibition of the activation pathway of the T-type calcium channel CaV3.1 by ProTxII", TOXICON, vol. 56, no. 4, 2010, pages 624 - 636 |
| ERICKSON ET AL.: "The Proteins(3rd ed.)", vol. 2, 1976, pages: 257 - 527 |
| ESTACION ET AL.: "A sodium channel mutation linked to epilepsy increases ramp and persistent current of Nav1.3 and induces hyperexcitability in hippocampal neurons", EXPERIMENTAL NEUROLOGY, vol. 224, no. 2, 2010, pages 362 - 36R |
| ESTACION ET AL.: "Navl.7 gain-of-function mutations as a continuum: A1632E displays physiological changes associated with erythromelalgia and paroxysmal extreme pain disorder mutations and produces symptoms of both disorders", J. NEUROSCI., vol. 28, no. 43, 2008, pages 11079 - 88 |
| EUR. J. BIOCHEM., vol. 138, 1984, pages 9 - 37 |
| EUR. J. BIOCHEM., vol. 213, 1993, pages 2 |
| FERTLEMAN ET AL., NEUROLOGY, vol. 69, 2007, pages 586 - 595 |
| FINN ET AL.: "The Proteins", vol. 2, 1976, pages: 105 - 253 |
| FRENCH ET AL.: "The tetrodotoxin receptor of voltage-gated sodium channels - perspectives from interactions with p-conotoxins", MARINE DRUGS, vol. 8, 2010, pages 2153 - 2161 |
| G. A. GRANT,: "Synthetic Peptides, A User's Guide", 1992, W.H. FREEMAN & COMPANY, NEW YORK |
| G. B. FIELDS ET AL.: "Synthetic Peptides: A User's Guide", 1990, pages: 77 - 183 |
| G.E. MEANS ET AL.: "Chemical modification of proteins", 1971, HOLDEN DAY, INC., article "Selected techniques for the modification of protein side chains", pages: 219 |
| G6NGORA-BENITEZ ET AL.: "Optimized Fmoc solid-phase synthesis of the cysteine-rich peptide Linaclotide", BIOPOLYMERS PEPT. SCI., vol. 96, no. 1, 2011, pages 69 - 80 |
| GOLDBERG ET AL., CLIN GENET, vol. 71, 2007, pages 311 - 319 |
| GRAHAM ET AL., VIROLOGY, vol. 52, 1973, pages 456 |
| GREGORIADIS ET AL.: "Improving the therapeutic efficacy of peptides and proteins: a role for polysialic acids", INTL. J. PHARMACEUTICS, vol. 300, 2005, pages 125 - 30 |
| GRIFFIN, A. M., AND GRIFFIN, H. G.: "Computer Analysis of Sequence Data", 1994, HUMANA PRESS |
| HAGCN, J PAIN SYMP MANAG, vol. 34, 2007, pages 171 - 182 |
| HAINS ET AL., J NEUROSCI, vol. 24, 2004, pages 4832 - 4839 |
| HAINS ET AL.: "Changes in electrophysiological properties and sodium channel Nav1.3 expression in thalamic neurons after spinal cord injury", BRAIN, vol. 128, 2005, pages 2359 - 71 |
| HAN ET AL.: "Disulfide-depleted selenoconopeptides: simplified oxidative folding of cysteine-rich peptides", ACS MED. CHEM. LETT., vol. 1, no. 4, 2010, pages 140 - 144 |
| HAN ET AL.: "Structurally minimized p-conotoxin analogs as sodium channel blockers: implications for designing conopeptide-based therapeutics", CHEMMEDCHEM, vol. 4, no. 3, 2009, pages 406 - 414 |
| HARTY ET AL.: "Navl .7 mutant A863P in erythromelalgia: effects of altered activation and steady-state inactivation on excitability of nociceptive dorsal root ganglion neurons", J. NEUROSCI., vol. 26, no. 48, 2006, pages 12566 - 75 |
| HENIKOFF ET AL., PROC. NATL. ACAD. SCI. U.S.A., vol. 89, 1992, pages 10915 - 10919 |
| HOLLINGER ET AL., PROC. NATL. ACAD. SCI. USA, vol. 90, 1993, pages 6444 - 6448 |
| HOYT ET AL.: "Bcnzazcpinonc Navl .7 blockcrs: Potential treatments for ncuropathic pain", BIOORG. MED. CHEM. LETT., vol. 17, no. 22, 2007, pages 6172 - 77 |
| HOYT ET AL.: "Discovery of a novel class of benzazepinone Na(v)1.7 blockers: potential treatments for neuropathic pain", BIOORG. MED. CHEM. LETT., vol. 17, no. 16, 2007, pages 4630 - 34 |
| HUBBARD ET AL.: "Annals Int. Med.", vol. 3, 1989, pages: 206 - 12 |
| HUSTON ET AL., PROC. ATATL ACAD. SCI. USA, vol. 85, 1988, pages 5879 - 5883 |
| IIDA ET AL.: "Two mechanisms of the enhanced antibody-dependent cellular cytotoxicity (ADCC) efficacy of non-fucosylated therapeutic antibodies in human blood", BMC CANCER, vol. 9, 2009, pages 58 |
| INTERNAT. J. PEPT. PROT. RES., vol. 24, 1984, pages 84 |
| ISOM: "Sodium channel beta subunits: anything but auxiliary", NEUROSCIENTIST, vol. 7, no. 1, 2001, pages 42 - 54 |
| J. BIOL. CHEM., vol. 260, 1985, pages 14 - 42 |
| JALALI ET AL.: "OD1, the first toxin isolated from the venom of the scorpion Odonthobuthus doriae active on voltage-gated Na+ channels", FEBS LETT., vol. 579, no. 19, 2005, pages 4181 - 4186 |
| JARAVINE ET AL.: "Three-dimensional structure of toxin OSK1 from Orthochirus scrobiculosus scorpion venom", BIOCHEM., vol. 36, no. 6, 1997, pages 1223 - 32 |
| JENSEN, T. S.: "Anticonvulsants in neuropathic pain: rationale and clinical evidence", EUR J PAIN, vol. 6, no. A, 2002, pages 61 - 68 |
| KABAT: "Sequences of Proteins of Immunological Interest", 1987, NIH |
| KALMAN ET AL.: "ShK-Dap22, a potent Kvl .3-specific immunosuppressive polypeptide", J. BIOL. CHEM., vol. 273, no. 49, 1998, pages 32697 - 707 |
| KEIL, B.: "Specificity of proteolysis", 1992, SPRINGER-VERLAG BERLIN-HEIDELBERG-NEWYORK, pages: 335 |
| KHOO ET AL.: "Lactam-stabilized helical analogues of the analgesic µ-conotoxin KIIIA", J. MED. CHEM., vol. 54, 2011, pages 7558 - 7566 |
| KHOO ET AL.: "Structure of the analgesic p-conotoxin KIIIA and effects on the structure and function of disulfide deletion", BIOCHEM., vol. 48, no. 6, 2009, pages 1210 - 1219 |
| KYTE ET AL., J. MOL. BIOL., vol. 157, 1982, pages 105 - 131 |
| LAITINCN: "Genetically engineered avidins and streptavidins", CELL MOL LIFE SCI., vol. 63, no. 24, 2006, pages 2992 - 30177 |
| LAITINEN ET AL.: "Genetically engineered avidins and streptavidins", CELL MOL LIFE SCI., vol. 63, no. 24, 2006, pages 2992 - 30177 |
| LESK, A. M.,: "Computational Molecular Biology", 1988, OXFORD UNIVERSITY PRESS |
| LIANG ET AL.: "Properties and amino acid sequence of huwentoxin-I, a neurotoxin purified from the venom of the Chinese bird spider Selenocosmia huwena", TOXICON, vol. 31, no. 8, 1993, pages 969 - 78 |
| LINK ET AL.: "Non-canonical amino acids in protein engineering", CURRENT OPINION IN BIOTECHNOLOGY, vol. 14, no. 6, 2003, pages 603 - 609 |
| M. BODANSZKY AND A. BODANSZKY,: "The Practice of Peptide Synthesis, 2nd cd.", 1994, SPRINGER-VERLAG |
| M. BODANSZKY,: "Principles of Peptide Synthesis, 2nd ed.,", 1993, SPRINGCR-VCRLAG |
| M. MORPURGO ET AL.: "Preparation and characterization of poly(ethylene glycol) vinyl sulfone", BIOCONJ. CHEM., vol. 7, 1996, pages 363 - 368 |
| MACLENNAN ET AL., ACTA PHYSIOL. SCAND., vol. 643, 1998, pages 55 - 67 |
| MAERTENS ET AL.: "Potent modulation of the voltage-gated sodium channel Nav1.7 by OD1, a toxin from the scorpion Odonthobuthus doriae", MOL. PHARM., vol. 70, no. 1, 2006, pages 405 - 414 |
| MANIATIS ET AL., SCIENCE, vol. 236, 1987, pages 1237 |
| MANIATIS ET AL.: "Molecular Cloning. A Laboratory Manual.", 1982, COLD SPRING HARBOR LABORATORY |
| MAO, J.; L. L. CHEN: "Systemic lidocaine for neuropathic pain relief", PAIN, vol. 87, no. 1, 2000, pages 7 - 17 |
| MARSHALL, K.: "Modem Pharmaceutics", 1979 |
| MCARTHUR ET AL.: "Interactions of key charged residues contributing to selective block of neuronal sodium channels by p-conotoxin KIIIA", MOL. PHARM., vol. 80, no. 4, 2011, pages 573 - 584 |
| MERRIFIELD, J. AM. CHEM. SOC., vol. 85, 1963, pages 2149 |
| MERRIFIELD: "Chem. Polypeptides", 1973, pages: 335 - 61 |
| MIDDLETON ET AL.: "Two tarantula peptides inhibit activation of multiple sodium channels", BIOCHEM., vol. 41, no. 50, 2002, pages 14734 - 14747 |
| MOLEC. IMMUNOL., vol. 29, no. 5, 1992, pages 633 - 9 |
| MOUHAT ET AL.: "K+ channel types targeted by synthetic OSK1, a toxin from Orthochirus scrobiculosus scorpion venom", BIOCHEM. J., vol. 385, 2005, pages 95 - 104 |
| MOUHAT ET AL.: "Pharmacological profiling of Orthochirus scrobiculosus toxin 1 analogs with a trimmed N-terminal domain", MOLEC. PHARMACOL., vol. 69, 2006, pages 354 - 62 |
| NASSAR ET AL.: "Nociceptor-specific gene deletion reveals a major role for Navl.7 (PN1) in acute and inflammatory pain", PROC. NATL. ACAD. SCI, U S A., vol. 101, no. 34, 2004, pages 12706 - 12711 |
| NEEDLEMAN ET AL., J. MOL. BIOL., vol. 48, 1970, pages 443 - 453 |
| NEWMARK ET AL., J. APPL. BIOCHEM., vol. 4, 1982, pages 185 - 9 |
| NICKE ET AL., EUR. J. BIOCHEM., vol. 271, 2004, pages 2305 - 19 |
| NN, vol. 152, 1985, pages 1 |
| NODA, M.; H. SUZUKI ET AL.: "A single point mutation confers tetrodotoxin and saxitoxin insensitivity on the sodium channel II", FEBS LETT, vol. 259, no. 1, 1989, pages 213 - 6 |
| NORTON ET AL.: "The cystine knot structure of ion channel toxins and related polypeptides", TOXICON, vol. 36, no. 11, 1998, pages 1573 - 1583 |
| OSWEIN ET AL.: "Aerosolization of Proteins", PROC. SYMP. RESP. DRUG DELIVERY II, March 1990 (1990-03-01) |
| P. J. KOCIENSKI,: "Protecting Groups", 1994, GEORG THIEME VERLAG |
| PALLAGHY ET AL.: "A common structural motif incorporating a cystine knot and a triple-stranded ?-sheet in toxic and inhibitory polypeptidcs", PROT. SCI., vol. 3, no. 10, 1994, pages 1833 - 6 |
| PENG ET AL.: "Function and solution structure of huwentoxin - IV , a potent neuronal tetrodotoxin (TTX)-sensitive sodium channel antagonist from chinese bird spider Selenocosmia huwena", J. BIOL. CHEM., vol. 277, no. 49, 2002, pages 47564 - 47571 |
| PENNINGTON ET AL.: "Engineering a stable and selective peptide blocker of the Kvl.3 channel in T lymphocytes", MOLECULAR PHARMACOLOGY FAST FORWARD, 2 January 2009 (2009-01-02) |
| PENNINGTON ET AL.: "Role of disulfide bonds in the structure and potassium channel blocking activity of ShK toxin", BIOCHEM., vol. 38, no. 44, 1999, pages 14549 - 58 |
| PLUCKTHUN: "The Pharmacology of Monoclonal Antibodies", vol. 113, 1994, SPRINGER-VERLAG, NEW YORK, pages: 269 - 315 |
| PRIEST ET AL.: "ProTx-I and ProTx-TT: gating modifiers of voltage-gated sodium channels", TOXICON, vol. 49, no. 2, 2007, pages 194 - 201 |
| PROCHNICKA-CHALUFOUR ET AL.: "Solution structure of discrepin, a new K+-channel blocking peptide from the alpha-KTxl5 subfamily", BIOCHEM., vol. 45, no. 6, 2006, pages 1795 - 1804 |
| PROCHNICKA-CHALUFOUR ET AL.: "Solution structure of discrepin, a new K+-channel blocking peptide from the alpha-KTxlS subfamily", BIOCHEM., vol. 45, no. 6, 2006, pages 1795 - 1804 |
| PURE APPL. CHEM., vol. 56, 1984, pages 595 - 624 |
| R. GREENWALD ET AL.: "Poly(ethylene glycol) conjugated drugs and prodrugs: a comprehensive review", CRITICAL REVIEWS IN THERAPEUTIC DRUG CARRIER SYSTEMS, vol. 17, 2000, pages 101 - 161 |
| R.J. GOODSON; N.V. KATRE: "Site-directed pegylation of recombinant interleukin-2 at its glycosylation site", BIOTECHNOLOGY, vol. 8, 1990, pages 343 - 346 |
| ROTH, D.B.; CRAIG, N.L., CELL, vol. 94, 1998, pages 411 - 414 |
| ROZEN, T. D.: "Antiepileptic drugs in the management of cluster headache and trigeminal neuralgia", HEADACHE, vol. 41, no. 1, 2001, pages 25 - 32 |
| S. HERMAN ET AL.: "Poly(ethylene glycol) with reactive endgroups: 1. Modification of proteins", J. BIOACTIVE COMPATIBLE POLYMERS, vol. 10, 1995, pages 145 - 187 |
| S. HERMAN ET AL.: "Poly(ethylene glycol) with reactive endgroups: I. Modification of proteins", J. BIOACTIVE COMPATIBLE POLYMERS, vol. 10, 1995, pages 145 - 187 |
| S. HERMAN: "Poly(ethylene glycol) with Reactive Endgroups: I. Modification of Proteins", J. BIOACTIVE AND COMPATIBLE POLYMERS, vol. 10, 1995, pages 145 - 187 |
| S. ZALIPSKY ET AL.: "Poly(ethylene glycol) chemistry: Biotechnical and biomedical applications", 1992, PLENUM PRESS: NEW YORK, article "Use of functionalized poly(ethylene glycol)s for modification of polypeptides", pages: 347 - 370 |
| S. ZALIPSKY: "Chemistry of Polyethylene Glycol Conjugates with Biologically Active Molecules", ADVANCED DRUG DELIVERY REVIEWS, vol. 16, 1995, pages 157 - 182 |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual", 2001 |
| SANDLER; KARO: "Polymer Synthesis", vol. 3, ACADEMIC PRESS, NEW YORK, pages: 138 - 161 |
| SASAKI ET AL., ADV. BIOPHYS, vol. 35, 1998, pages 1 - 24 |
| SCC, GOLDIN, A. L.: "Resurgence of sodium channel research", ANNU REV PHYSIOL, vol. 63, 2001, pages 871 - 94 |
| SCHECHTER 1.; BERGER A.: "On the active site of proteases. 3. Mapping the active site of papain; specific inhibitor peptides of papain", BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATION, vol. 32, 1968, pages 898 |
| SCHECHTER I.; BERGER A.: "On the size of the active site in proteases. I. Papain", BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATION, vol. 27, 1967, pages 157 |
| SCHMALHOFER ET AL.: "ProTx-II, a selective inhibitor of NaV 1.7 sodium channels, blocks action potential propagation in nociceptors", MOL. PHARM., vol. 74, no. 5, 2008, pages 1476 - 1484 |
| SMITH ET AL., J. CLIN. INVEST., vol. 84, 1989, pages 1145 - 6 |
| SMITH ET AL.: "Differential phospholipid binding by site 3 and site 4 toxins: implications for structural variability between voltage-sensitive sodium channel comains", J. BIOL. CHEM., vol. 280, no. 12, 2005, pages 11127 - 11133 |
| SMITH ET AL.: "Molecular interactions of the gating modifier toxin ProTx-II with Navl .5: implied existence of a novel toxin binding site coupled to activation", J. BIOL. CHEM., vol. 282, no. 17, 2007, pages 12687 - 12697 |
| SMITH, D. W.,: "Biocomputing Informatics and Genome Projects", 1993, ACADEMIC PRESS |
| SOKOLOV ET AL.: "Inhibition of sodium channel gating by trapping the domain 11 voltage sensor with protoxin 11", MOL. PHARM., vol. 73, no. 3, 2008, pages 1020 - 1028 |
| STEINER; BULAJ: "Optimization of oxidative folding methods for cysteine-rich peptides: a study of conotoxins containing three disulfide bridges", J. PEPT. SCI., vol. 17, no. 1, 2011, pages 1 - 7 |
| T. W. GREENE AND P. G. M. WUTS,: "Protecting Groups in Organic Synthesis,3rd Edition,", 1999, JOHN WILEY & SONS, INC. |
| THIMMAPAYA ET AL.: "Distribution and functional characterization of human Navl.3 splice variants", EUR. J. NEUROSCI., vol. 22, 2005, pages 1 - 9 |
| TUDOR ET AL.: "Ionisation behaviour and solution properties of the potassium-channel blocker ShK toxin", EUR. J. BIOCHEM., vol. 251, no. 1-2, 1998, pages 133 - 41 |
| TUDOR: "Ionisation behaviour and solution properties of the potassium-channel blocker ShK toxin", EUR. J. BIOCHEM., vol. 251, no. 1-2, 1998, pages 133 - 41 |
| TURK, B. ET AL.: "Determination of protease cleavage site motifs using mixture-based oriented peptide libraries", NATURE BIOTECHNOLOGY, vol. 19, 2001, pages 661 - 667 |
| UYSAL-ONGANER ET AL.: "Epidermal growth factor potentiates in vitro metastatic behavior human prostate cancer PC-3M cells: involvement of voltage-gated sodium channel", MOLEC. CANCER, vol. 6, 2007, pages 76 |
| VON HEINJE, G.: "Sequence Analysis in Molecular Biology", 1987, ACADEMIC PRESS |
| VOSS ET AL., TRENDS BIOCHEM. SCI., vol. 11, 1986, pages 287 |
| W. D. BENNET, J. W. CHRISTENSEN, L. K. HAMAKER, M. L. PETERSON, M. R. RHODES, AND H. H.SANEII: "Advanced Chemtech Handbook of Combinatorial & Solid Phase Organic Chemistry", 1998, ADVANCED CHEMTECH |
| WANG ET AL.: "The cross channel activities of spider neurotoxin huwentoxin I on rat dorsal root ganglion neurons", BIOCHEM. BIOPHYS. RES. COMM., vol. 357, no. 3, 2007, pages 579 - 583 |
| WOOLF; MA, NEURON, vol. 55, 2007, pages 353 - 364 |
| XIAO ET AL.: "Synthesis and characterization of huwentoxin-IV, a neurotoxin inhibiting central neuronal sodium channels", TOXICON, vol. 51, no. 2, 2008, pages 230 - 239 |
| XIAO ET AL.: "Tarantula huwentoxin-IV inhibits neuronal sodium channels by binding to receptor site 4 and trapping the domain II voltage sensor in the closed configuration", J. BIOL. CHEM., vol. 283, no. 40, 2008, pages 27300 - 27313 |
| XIAO ET AL.: "The tarantula toxins ProTx-II and huwentoxin-IV differentially interact with human Navl .7 voltage sensors to inhibit channel activation and inactivation", MOL. PHARM., vol. 78, no. 6, 2010, pages 1124 - 1134 |
| Y. LU ET AL.: "PEGylated peptides III: Solid-phase synthesis with PEGylating reagents of varying molecular weight: synthesis of multiply PEGylated pcptidcs", REACTIVE POLYMERS, vol. 22, 1994, pages 221 - 229 |
| YANG: "Mutations in SCN9A, encoding a sodium channel alpha subunit, in patients with primary erythermalgia", J. MED. GENET., vol. 41, 2004, pages 171 - 174 |
| YEOMANS ET AL.: "Decrease in inflammatory hyperalgesia by herpes vector-mediated knockdown of Navl.7 sodium channels in primary afferents", HUM. GENE THER., vol. 16, no. 2, 2005, pages 271 - 7 |
| ZALIPSKY, S. ET AL.: "poly(ethylene glycol) chemistry: Biotechnical and biomedical applications", 1992, PLENUM PRESS: NEW YORK., article "Use of functionalized poly(ethylene glycol)s for modification of polypeptides", pages: 347 - 370 |
| ZAPATA ET AL., PROTEIN ENG., vol. B, no. 10, 1995, pages 1057 - 1062 |
| ZHANG ET AL.: "Cooccupancy of the outer vestibule of voltage-gated sodium channels by µ-conotoxin KIIIA and saxitoxin or tetrodotoxin", J. NEUROPHYS, vol. 104, no. 1, 2010, pages 88 - 97 |
| ZHANG ET AL.: "p-Conotoxin KIIIA derivatives with divergent affinities versus efficacies in blocking voltage-gated sodium channels", BIOCHEM., vol. 49, no. 23, 2010, pages 4804 - 4812 |
| ZHANG ET AL.: "Structure/function characterization of p-conotoxin KIIIA , an analgesic, nearly irreversible blocker of mammalian neuronal sodium channels", J. BIOL. CHEM., vol. 282, no. 42, 2007, pages 30699 - 30706 |
| ZHANG: "Syncrgistic and antagonistic interactions between tetrodotoxin and g-conotoxin in blocking voltage-gated sodium channels", CHANNELS, vol. 3, no. 1, 2009, pages 32 - 38 |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9636418B2 (en) | 2013-03-12 | 2017-05-02 | Amgen Inc. | Potent and selective inhibitors of NAV1.7 |
| WO2014165277A2 (en) | 2013-03-12 | 2014-10-09 | Amgen Inc. | POTENT AND SELECTIVE INHIBITORS OF Nav1.7 |
| WO2014165277A3 (en) * | 2013-03-12 | 2015-02-26 | Amgen Inc. | POTENT AND SELECTIVE INHIBITORS OF Nav1.7 |
| US10344060B2 (en) | 2013-03-12 | 2019-07-09 | Amgen Inc. | Potent and selective inhibitors of Nav1.7 |
| US11220556B2 (en) * | 2013-03-15 | 2022-01-11 | Biomolecular Holdings Llc | Hybrid immunoglobulin containing non-peptidyl linkage |
| US20160024226A1 (en) * | 2013-03-15 | 2016-01-28 | Daniel J. Capon | Hybrid immunoglobulin containing non-peptidyl linkage |
| WO2014144911A3 (en) * | 2013-03-15 | 2014-12-04 | Capon Daniel J | Hybrid immunoglobulin containing non-peptidyl linkage |
| US11066459B2 (en) | 2014-03-14 | 2021-07-20 | Biomolecular Holdings Llc | Hybrid immunoglobulin containing non-peptidyl linkage |
| CN111187338A (en) * | 2014-06-12 | 2020-05-22 | Ra制药公司 | Modulation of complement activity |
| EP4194562A4 (en) * | 2020-08-05 | 2024-08-14 | Aist | Method for screening for polypeptide that acts on target protein |
| WO2022066767A2 (en) | 2020-09-23 | 2022-03-31 | Aldevron, Llc | Potent and selective inhibitors of nav1.7 |
| WO2022066767A3 (en) * | 2020-09-23 | 2022-07-07 | Aldevron, Llc | Potent and selective inhibitors of nav1.7 |
| WO2023205468A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205778A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205465A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2023205463A1 (en) | 2022-04-22 | 2023-10-26 | Vertex Pharmaceuticals Incorporated | Heteroaryl compounds for the treatment of pain |
| WO2024123815A1 (en) | 2022-12-06 | 2024-06-13 | Vertex Pharmaceuticals Incorporated | Process for the synthesis of substituted tetrahydrofuran modulators of sodium channels |
Also Published As
| Publication number | Publication date |
|---|---|
| MA35385B1 (en) | 2014-09-01 |
| EP2686340A2 (en) | 2014-01-22 |
| JP2017031157A (en) | 2017-02-09 |
| KR20140145947A (en) | 2014-12-24 |
| TW201300407A (en) | 2013-01-01 |
| SG193434A1 (en) | 2013-10-30 |
| BR112013023674A2 (en) | 2016-12-13 |
| JP2014509859A (en) | 2014-04-24 |
| SG10201601789TA (en) | 2016-04-28 |
| US9796766B2 (en) | 2017-10-24 |
| PE20140593A1 (en) | 2014-05-10 |
| CN103930437A (en) | 2014-07-16 |
| US9340590B2 (en) | 2016-05-17 |
| EA201391331A1 (en) | 2014-02-28 |
| CR20130533A (en) | 2014-01-09 |
| US20140073577A1 (en) | 2014-03-13 |
| AR088413A1 (en) | 2014-06-11 |
| CA2830065A1 (en) | 2012-09-20 |
| ZA201306920B (en) | 2014-05-28 |
| AU2012228990A1 (en) | 2013-09-19 |
| NZ615242A (en) | 2016-03-31 |
| CO6821886A2 (en) | 2013-12-31 |
| US20160304570A1 (en) | 2016-10-20 |
| WO2012125973A3 (en) | 2013-01-03 |
| CL2013002673A1 (en) | 2014-07-25 |
| HK1198173A1 (en) | 2015-03-13 |
| UY33959A (en) | 2012-09-28 |
| AP2013007173A0 (en) | 2013-10-31 |
| MX2013010497A (en) | 2013-12-16 |
| AU2012228990B2 (en) | 2017-04-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9796766B2 (en) | Potent and selective inhibitors of NAV1.3 and NAV1.7 | |
| AU2010226392B9 (en) | Selective and potent peptide inhibitors of Kv1.3 | |
| EP2970408B1 (en) | Potent and selective inhibitors of nav1.7 | |
| US7825093B2 (en) | Methods of using OSK1 peptide analogs | |
| US9636418B2 (en) | Potent and selective inhibitors of NAV1.7 | |
| NZ615242B2 (en) | Potent and selective inhibitors of nav1.3 and nav1.7 | |
| Murray et al. | Potent and selective inhibitors of NA V 1.7 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12711312 Country of ref document: EP Kind code of ref document: A2 |
|
| ENP | Entry into the national phase |
Ref document number: 2830065 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2013558223 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 002050-2013 Country of ref document: PE Ref document number: MX/A/2013/010497 Country of ref document: MX |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2013002673 Country of ref document: CL Ref document number: 1301005083 Country of ref document: TH |
|
| ENP | Entry into the national phase |
Ref document number: 2012228990 Country of ref document: AU Date of ref document: 20120316 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15256595 Country of ref document: CO Ref document number: 13242679 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: A201312087 Country of ref document: UA Ref document number: 201391331 Country of ref document: EA |
|
| ENP | Entry into the national phase |
Ref document number: 20137027301 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012711312 Country of ref document: EP Ref document number: CR2013-000533 Country of ref document: CR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 14005135 Country of ref document: US |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013023674 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112013023674 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130916 |