WO2012118819A2 - Nitric oxide-releasing s-nitrosothiol-modified silica particles and methods of making the same - Google Patents
Nitric oxide-releasing s-nitrosothiol-modified silica particles and methods of making the same Download PDFInfo
- Publication number
- WO2012118819A2 WO2012118819A2 PCT/US2012/026960 US2012026960W WO2012118819A2 WO 2012118819 A2 WO2012118819 A2 WO 2012118819A2 US 2012026960 W US2012026960 W US 2012026960W WO 2012118819 A2 WO2012118819 A2 WO 2012118819A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nitrosothiol
- particles
- thiol
- functionalized
- silica particles
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2/00—Methods or apparatus for disinfecting or sterilising materials or objects other than foodstuffs or contact lenses; Accessories therefor
- A61L2/16—Methods or apparatus for disinfecting or sterilising materials or objects other than foodstuffs or contact lenses; Accessories therefor using chemical substances
- A61L2/18—Liquid substances or solutions comprising solids or dissolved gases
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0834—Compounds having one or more O-Si linkage
- C07F7/0838—Compounds with one or more Si-O-Si sequences
- C07F7/0872—Preparation and treatment thereof
- C07F7/0874—Reactions involving a bond of the Si-O-Si linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/10—Compounds having one or more C—Si linkages containing nitrogen having a Si-N linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/26—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen nitrogen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/28—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen sulfur-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/28—Nitrogen-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/30—Sulfur-, selenium- or tellurium-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/02—Elements
- C08K2003/023—Silicon
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
Definitions
- the present invention relates to nitric oxide-releasing particles. More particularly, the present application relates to S-nitrosothiol-modified silica particles.
- RSNOs S-Nitrosothiols
- RSNOs S-nitroso-glutathione (GSNO), S-nitroso-iV-acetylcysteine (SNAC), and S-nitroso-N-acetyl-penicillamine (SNAP)
- GSNO S-nitroso-glutathione
- SNAC S-nitroso-iV-acetylcysteine
- SNAP S-nitroso-N-acetyl-penicillamine
- NO donor precursors have been conjugated to larger scaffolds (e.g., proteins, dendrimers, and nanoparticles), thus enabling high NO storage per delivery vehicle and release profiles similar to their small molecule analogues.
- Silica particles are among the most widely employed macromolecular scaffolds for biomedical applications due to facile synthetic strategies and minimal cytotoxicity.
- the surface of fumed silica particles (7-10 nm diameter) have been grafted with SNAP, SNAC, and S-nitrosocysteine (CysNO) to create S-nitrosothiol-modified silica particles.
- NO storage was limited to 0.021-0.138 ⁇ mg " ' because the thiol functionalization was restricted to the exterior of the particle. Additionally, these systems are not able to tune particle size to fit a therapeutic system of interest.
- the hydrolysis and co-condensation of organosilane and tetraalkoxysilane precursors via sol-gel chemistry may represent a method for preparing a silica network with a higher concentration of organic functionalites.
- the Stober process (sol-gel chemistry with an alcohol solvent and an ammonia catalyst) has proven effective for synthesizing N-diazeniumdiolate- modified silica particles of diverse size and NO storage capacity. See, for example, U.S. Publication No. 2009/0214618 (Schoenfisch et al.), which is herein incorporated by reference in its entirety.
- the advantage of the Stober method over surface grafting is that the co- condensation provides uniform incorporation of the organic (i.e., NO donor) functionality throughout the resulting silica network as opposed to restricted functionalization at the surface alone. As a result, such particles may exhibit significantly increased NO storage.
- a first aspect of the present invention comprises a method of forming
- S-nitrosothiol-functionalized co-condensed silica particles comprising:
- thiol-containing silane and a backbone alkoxysilane in a sol precursor solution that comprises water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles comprise a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix;
- a second aspect of the present invention comprises S-nitrosothiol-functionalized monodisperse co-condensed silica particles having an average particle diameter in a range of about 10 nni to about 100 ⁇ .
- a further aspect of the present invention comprises S-nitrosothiol-functionalized co- condensed silica particles having an NO storage in a range of about 0.01 ⁇ to about 10 ⁇ NO per mg particle.
- Another aspect of the present invention comprises a S-nitrosothiol-functionalized co- condensed silica particle comprising tertiary nitrosothiol functional groups.
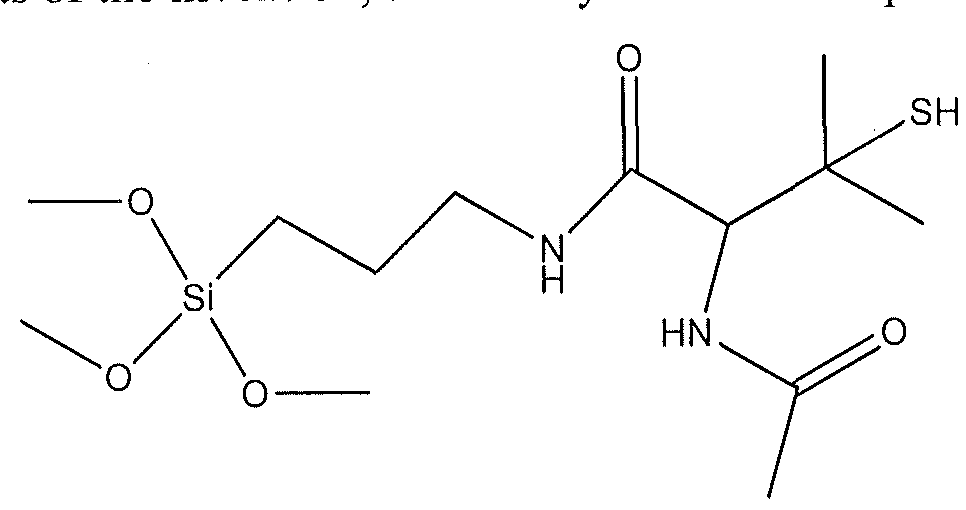
- a further aspect of the present invention comprises a compound having the following structure:
- R, R' and R" are each independently alkyl and n is 0 in a range of 0 to 10.
- Figure 1 provides a scheme for the synthesis of N-acetylpenicillamine
- NAPTMS propyltrimethoxysilane
- Figure 2 provides an ⁇ NMR spectrum of the NAPTMS compound.
- Figure 3 shows a solid-state cross polarization/magic angle spinning (CP/MAS) "Si NMR spectra of silica synthesized with (A) 0, (B) 25, (C) 40, (D) 60, (E) 75, and (F) 85 mol % MPTMS (balance TMOS). The Q and T bands have been designated.
- CP/MAS cross polarization/magic angle spinning
- Figure 4 shows the scanning electron micrographs of (A) 25, (B) 40, (C) 60, (D) 75 and (E) 85 mol% MPTMS (balance TMOS) and (F) 75 and (G) 85 mol % MPTMS (balance TEOS) particles synthesized with 16.0 M water, 5.5 M ammonia, and 0.1 M silane.
- Figure 5 shows the scanning electron micrographs of (A) 25, (B) 40, (C) 60, (D) 75 and (E) 85 mol% MPTMS (balance TMOS) and (F) 75 and (G) 85 mol % MPTMS (balance TEOS) particles synthesized via a semi-batch process with a silane feed rate of 0.5 rriL min "1 .
- Figure 6 shows the scanning electron micrographs of 75 mol% MPTMS (balance TEOS) synthesized with (A) 47.0, (B) 42.0, (C) 40.6, (D) 36.5, (E) 32.5, and (F) 24.9 M water,
- Figure 7 shows the scanning electron micrographs of (A-B) 25, (C-D) 40, (E-F) 60, (G-H) 75, and (I-J) 85 mol% MPTMS (balance TMOS) and (K-L) 85 mol % MPTMS (balance TEOS) particles synthesized with (A,C,E,F,G,I,K) 32.5 and (B, D, F, H, J, L) 24.9
- Figure 8 shows a schematic for the S-nitrosothiol decomposition pathways.
- Figure 9 shows the nitric oxide release from RSNO-modified 75 mol% MPTMS
- Figure 10 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention prior to sonication.
- Figure 11 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention after 30 minutes sonication followed by nitrosation.
- Figure 12 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention after 60 minutes sonication followed by nitrosation.
- alkyl refers to C 1-2 o inclusive, linear (i.e., "straight-chain"), branched, or cyclic, saturated or at least partially and in some cases fully unsaturated (i.e., alkenyl and alkynyl) hydrocarbon chains, including for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, octyl, ethenyl, propenyl, butenyl, pentenyl, hexenyl, octenyl, butadienyl, propynyl, butynyl, pentynyl, hexynyl, heptynyl, and allenyl groups.
- Branched refers to an alkyl group in which a lower alkyl group, such as methyl, ethyl or propyl, is attached to a linear alkyl chain.
- exemplary branched alkyl groups include, but are not limited to, isopropyl, isobutyl, tert-butyl.
- “Lower alkyl” refers to an alkyl group having 1 to about 8 carbon atoms (i.e., a Ci -8 alkyl), e.g., 1 , 2, 3, 4, 5, 6, 7, or 8 carbon atoms.
- “Higher alkyl” refers to an alkyl group having about 10 to about 20 carbon atoms, e.g., 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 carbon atoms. In certain
- alkyl refers, in particular, to C1-5 straight-chain alkyls. In other embodiments, “alkyl” refers, in particular, to C 1-5 branched-chain alkyls.
- mercapto or "thio” refers to the -SH group.
- thiol-functionalized co-condensed silica particles that include reacting a thiol- containing silane and a backbone alkoxysilane in a sol precursor solution that includes water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles include a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix.
- the methods further include reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent to provide the S-nitrosothiol-functionalized co-condensed silica particles.
- any suitable thiol-containing silane may be used.
- the thiol- containing silane includes a primary thiol, in some embodiments, a secondary thiol, and in some embodiments, a tertiary thiol. Combinations of different silanes may also be used.
- a suitable thiol-containing silane will be a silane that will allow for particle formation, and in some embodiments, monodisperse particle formation. Thus, some thiol-containing silanes may be suitable with some backbone alkoxysilanes and not suitable with others.
- the primary thiol-containing silane is mercaptopropyltrimethoxysilane.
- the tertiary thiol alkoxysilane has the following structure:
- tertiary thiol alkoxysilane has the structure:
- R, R' and R" are each independently H, alkyl or substituted alkyl and n is 0-10. In some embodiments, R, R' and R" are each independently alkyl and n is 0-5. Furthermore, in particular embodiments of the invention, the tertiary thiol is a compound having the structure:
- backbone alkoxysilane refers to an alkoxysilane that does not contain a thiol functional group. Examples include tetramethoxysilane, tetraethoxysilane, methyltrimethoxysilane, methyltriethoxysilane and butyltrimethoxysilane.
- a suitable backbone silane will be a silane that will allow for particle formation, and in some embodiments, monodisperse particle formation.
- the water concentration in the sol precursor solution is in a range of about 8 M to about 32.5 M.
- a catalyst such as an ammonia catalyst, is included in the sol precursor solution. Any suitable concentration of catalyst may be used. However, in some embodiments, an ammonia catalyst is included in the sol precursor solution, in some embodiments, at a concentration in a range of about 1.9 M to about 5.5 M.
- mercaptopropyltrimethoxysilane and tetramethoxysilane are reacted in the presence of water and an ammonia catalyst to form thiol-functionalized co-condensed silica particles.
- the reaction occurs in a solution that includes mercaptopropyltrimethoxysilane and tetramethoxysilane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 25 to about 85 mol % mercaptopropyltrimethoxysilane.
- water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 M to about 5.5 M.
- mercaptopropyltrimethoxysilane and tetraethoxysilane are reacted in the presence of water and an ammonia catalyst to form thiol- functionalized co-condensed silica particles.
- the reaction occurs in a solution that includes mercaptopropyltrimethoxysilane and tetraethoxysilane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 75 to about 85 mol % mercaptopropyltrimethoxysilane.
- water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 M to about 5.5M.
- methods of forming thiol-functionalized co-condensed silica particles include reacting a tertiary thiol-containing silane and a backbone alkoxysilane in the presences of water and an ammonia catalyst to form thiol-functionalized co-condensed silica particles.
- the reaction occurs in a solution that includes tertiary thiol- containing silane and alkoxysiiane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 25 to about 85 mol % tertiary thiol-containing silane.
- water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 to about 5.5 M.
- the sol precursor solution may also be stirred/agitated as known to those of skill in the art, and other additives or silane monomers used in sol chemistry may be included in some embodiments of the invention.
- methods provided herein may be used to form nitrosothiol-functionalized co-condensed silica particles, which in some embodiments, are monodisperse.
- monodisperse refers to particles having a uniform particle size, in some embodiments, having an average particle diameter ⁇ 100 nm as measured from electron micrographs; a Z-average ⁇ 60 nm as measured from dynamic light scattering; and/or having a polydispersity index ⁇ 0.1 as measured via dynamic light scattering.
- the methods described herein provide monodisperse particles having an average particle diameter of less than 100 microns, and in some embodiments, less than 1 micron.
- the methods used herein may provide monodisperse particles having an average particle diameter in a range of about 10 nm to about 100 ⁇ . In some embodiments, the particles have an average particle diameter in a range of about 200 to about 700 nm.
- any suitable method of nitrosating the thiol-functionalized co-condensed silica particles may be used. Further, any suitable nitrosating agent may be used. However, in some embodiments, the nitrosating agent includes acidified sodium nitrite, alkyl nitrites, including tertbutyl nitrite and isopentyl nitrite, and various nitrogen oxides including nitrous oxide, N 2 0 3 , N 0 4 and N0 2 . Examples of nitrosation may be found in Williams, D. L. H. Acc. Che . Res. 1999, 32, 869, the contents of which are incorporated herein by reference in their entirety.
- the nitrosation chemistry conserves particle size integrity and yields monodisperse S-nitrosothiol-functionalized co-condensed silica particles. No changes in particle size have been observed following addition of the nitric oxide functionality to the macromolecular structure, a drawback that has been observed with other nitrosothiol-modified macromolecular donors. Furthermore, as shown below in the Examples, the thiol-functionalized co-condensed silica particles may be sonicated prior to nitrosation without deleteriously affecting the NO storage and/or morphology of the particles.
- the co-condensed silica particles may include S-nitrosothiol groups throughout the particle, and as such, may provide enhanced NO storage properties.
- S-nitrosothiol-functionalized co-condensed silicas particles that have an NO storage in a range of about 0.01 ⁇ to about 10 ⁇ NO per mg particle, and in some embodiments, 0.09 ⁇ to about 4.40 ⁇ NO per mg particle.
- S-nitrosothiol groups throughout the interior of the silica particle structure may also afford unexpected stability.
- Glutathione and other thiols are known to one skilled in the art to be a vial trigger for RSNO decomposition and release a variety of NOx species.
- the low porosity of the S- nitrosothiol-functionalized co-condensed silica particles protect the RSNO donors from premature decomposition by glutathione or other blood components, adding a level of nitric oxide stability when used in drug delivery applications.
- NAP N-Acetyl Penicillamme
- NAPTMS N-Acetyl Penicillamme Propyltrimethoxysilane
- NAP thiolactone was thus synthesized to design such a precursor for the synthesis of NO-releasing xerogels. Penicillamine was reacted in the presence of acetic anhydride to generate the NAP thiolactone in situ. After characterization by ⁇ and l 3 CNMR, the NAP thiolactone was directly coupled with APTMS to result in a tertiary thiol-bearing silane, referred to as NAPTMS (see Figure 1). Successful synthesis of this tertiary thiol-bearing silane was verified via 'HNMR characterization ( Figure 2).
- Ratios of mercaptosilane and alkoxysilane were added either as a bolus injection or dropwise via a Kent Scientific Genie Plus syringe pump at a flow rate of 0.25-3.0 mL/min through an 18.5 gauge needle to a solution of ethanol, water, and ammonium hydroxide. Solution was stirred for 2 h at room temperature, collected via centrifugation at 4500 rpm (10 mins), washed twice with 40 n L EtOH, recollected, and dried overnight at ambient conditions.
- TMOS Tetramethoxysilane
- MPMDMS 3-mercaptopropylmethyldimethoxysilane
- Solid-state cross polarization/magic angle spinning (CP/MAS) 29Si (71.548 MHz frequency) nuclear magnetic resonance (NMR) spectroscopy was performed on a Bruker 360 MHz DMX spectrometer (Billerica, MA). Particles were packed into 4 mm rotors and spun at 8.0 kHz. Spectra were collected at 5000 scans with the determination of chemical shifts in parts per million relative to an external TMS standard. Nitric oxide release was measured in real time (1 sec intervals) using 5 a Sievers NOATM 280i Chemiluminescence Nitric Oxide Analyzer (NOA) (Boulder, CO).
- NOA Chemiluminescence Nitric Oxide Analyzer
- Copper-initiated NO release was studied by adding the particles to 25 n L of 10 or 25 ⁇ CuBr 2 (aq).
- the NOA sample flask was shielded from light with aluminum foil for experiments where light was not the intended initiator of NO release.
- Particle size was determined using a Zetasizer Nano ZS Particle Size and Zeta Potential Dynamic Light Scattering (DLS) Instrument (Malvern, UK). Samples were suspended in PBS at a concentration of 1 mg mL- 1 and sonicated for 15 min prior to analysis. Scanning electron micrographs were recorded on a Hitachi S-4700 Scanning Electron Microscope (Pleasanton, CA).
- CP/MAS NMR solid-state Si cross polarization/magic angle spinning nuclear magnetic resonance
- Figure 3 shows silica synthesized with (A) 0, (B) 25, (C) 40, (D) 60, (E) 75, and (F) 85 mol % MPTMS (balance TMOS). Particles synthesized entirely from TMOS exhibited only Q bands. With increasing MPTMS concentration in the solution used to prepare the particles, the T bands increased relative to the Q bands, confirming greater incorporation of MPTMS in the silica particle.
- Sulfur weight percent of each composition was determined using elemental analysis and further corroborated the covalent incorporation of the mercaptosilane.
- the weight percent of sulfur in the silica was 4.92, 7.1 1 , 11.67, 13.56 and 17.30 for the 25, 40, 60, 75 and 85 mol% MPTMS (balance TMOS) compositions, respectively.
- the TEOS-based particles were found to have sulfur weight percents of 16.15 and 19.34 for 75 and 85 mol% MPTMS, respectively.
- the weight percent of sulfur increased linearly with increasing MPTMS concentration in the initial solution.
- Example 4 Variation of water, ammonia and silane concentrations and feed rate
- FIG. 4 depicts the resulting particles as the concentration of MPTMS was increased from 25-85 mol% and backbone alkoxysilane varied between TMOS and TEOS.
- the particles were synthesized with 16.0 M water, 5.5 M ammonia, and 0.1 M silane.
- the product yield increased to -170 mg for these two compositions and can be attributed to the greater concentration of the larger MPTMS in the particles.
- the 75 mol% MPTMS (balance TEOS) particles were chosen as a model system to examine the effect of the water concentration on particle size and morphology.
- 75 mol% MPTMS (balance TEOS) was synthesized with (A) 47.0, (B) 42.0, (C) 40.6, (D) 36.5, (E) 32.5, and (F) 24.9 M water.
- Water concentrations >40.6 M favored rapid silane hydrolysis and condensation kinetics, leading to a highly condensed network rather than discrete, spherical particles.
- a water concentration of 36.5 M discrete particles were formed, but with morphologies featuring excessive aggregation.
- Monodisperse particles (333 ⁇ 48 nm) were first observed at a slightly lower water concentration (32.5 M). Particle size increased with decreasing water concentrations (456 ⁇ 68 nm and 635 ⁇ 63 nm for 24.9 and 16.2 M, respectively). Furthermore, the smaller particle sizes were accompanied with slightly lower yields for each composition.
- the yields for 75 mol% MPTMS (balance TEOS) particles were ⁇ 65, 150, and 170 mg for water concentrations of 32.5, 24,9, and 16.2 M, respectively.
- the differences in yield may be factors of the efficiency of particle collection (i.e., centrifugation rpm and duration) for the smaller particles rather than chemical differences.
- the appropriate water concentrations (32.5 and 24.9 M) were next used to tune particle size and reduce the bimodal distribution characteristic of the 60, 75, and 85 mol% MPTMS (balance TMOS) particles.
- the intermediate water concentration (24.9 M) yielded particles with sizes of 179 ⁇ 22 and 196 ⁇ 25 nm for the 25 and 40 mol % MPTMS (balance TMOS) compositions, respectively.
- the greater water concentration (32.5 M) drastically increased the reaction kinetics for the mostly TMOS-based systems, resulting in highly-fused silica networks.
- the TEOS-based counterpart to this system behaved similarly by yielding only discrete particles (506 ⁇ 77 nm) when synthesized with the higher water concentration. At lower water amounts, the formation of aggregated particles was noted. The trend of decreasing particle yield with increasing water content that was observed for the 75 mol% MPTMS (balance TEOS) composition was mirrored for all other compositions evaluated. The yields for the 75 mol% (balance TMOS) particles decreased from -120 to 60 mg upon increasing the water content from 24.9 to 32.5 M.
- the 25 mol% MPTMS (balance TMOS) particle yield decreased from -90 to 20 mg while the 40 mol% MPTMS system exhibited a decrease from -50 to 9 mg upon increasing the water concentration from 16.2 to 24.9 M.
- the yields for both 85 mol% MPTMS compositions (i.e, TMOS and TEOS balance) at a water concentration of 32.5 M were -160 mg.
- the elevated water concentrations used to synthesize the thiol-modified particles successfully resolved the bimodal nature of certain compositions not resolvable using a semi-batch process alone.
- 60 mol% MPTMS (balance TMOS) was the only composition that consistently yielded particles of a bimodal nature. Increasing the water content regardless of addition method (bolus vs. semi-batch) resulted in a highly-fused silica network.
- Particle sizes were also measured by DLS to corroborate particle monodispersity and size measured using SEM. As shown in Table 1, the DLS measurements were in agreement with the sizes calculated from the SEM images. The slightly increased average diameters observed with DLS may be attributed to particle hydration (DLS measurements conducted in solution). Like SEM, the DLS measurements indicated a narrow size distribution, as evidenced by low polydispersity indices for each composition.
- PBS was used as a dispersant for compositions with a large concentration of MPTMS.
- smaller particles with a large degree of inorganic character i.e., ⁇ 40 mol% MPTMS
- This aggregation may be attributed to a large surface density of protonated silanol groups leading to unfavorable particle interaction.
- ethanol was a viable alternative dispersant as evidenced by the correlation between DLS and SEM measurements. Elemental analysis was used to characterize the composition of the particles. As expected, the weight percentages of sulfur in the particles increased accordingly with the MPTMS mol% used to make the particles indicating incorporation of the thiol functionality (Table 2).
- TMOS/ethanol mixture (71.9 ⁇ , TMOS and 200 ⁇ , ethanol) via syringe pump at a rate of 1.0 mL/min.
- Particles were collected by centrifugation at 4500 rpm (5 min), washed with 40 mL chilled 500 ⁇ DTPA(aq), recollected, washed with 40 mL chilled MeOH, recollected, and vacuum dried for 30 min while shielded from light. Particles were stored at -20 °C in vacuo until further study.
- the MPTMS-modified particles were nitrosated to enable NO storage and release. Briefly, the particles were treated with acidified sodium nitrite, generating nitrous acid, a nitrosating agent that reacts with thiols to form RSNOs (see Eq 1).
- Transnitrosation between a thiol and an RSNO may also occur, resulting in the transfer of the nitroso functionality and formation of a new RSNO species that may decompose via the aforementioned pathways.
- RSNO-modified particles ( ⁇ 2 mg) were added to 500 ⁇ DTP A (pH 7.4 PBS) at a temperature of 0 °C, while measuring the ensuing NO release as a function of photolytic decomposition.
- RSNO-modified silica particles exhibited photo-initiated NO release upon exposure to broadband, white light. Greater irradiation levels (i.e., power) resulted in elevated NO release from the particles.
- low levels of NO release ( ⁇ 15 ppb mg "1 s "1 ) were observed at 0 °C and in the dark
- the discrepancy may be attributed to inability to measure NO at low levels beyond 48 h and/or loss of NO through its reaction with oxygen present in the soak solutions. As evident by a pinlc hue, the particles still contained a portion of their NO payload even after 48 h of release.
- Example 8 Influence of particle sonication before/after nitrosation
- SEM images of the particles formed in Example 7 A are provided in Figure 10.
- SEM images of the particles formed in Example 7C are provided in Figure 11.
- SEM images of the particles formed in Example 7D are shown in Figure 12.
Abstract
Provided according to some embodiments of the invention are methods of forming co-condensed silica particles. In some embodiments, the methods include reacting a thiol-containing silane and a backbone alkoxysilane in a reaction solution that comprises water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles include a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix; and reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent to provide the S-nitrosothiol-functionalized co-condensed silica particles. In some embodiments, provided are S-nitrosothiol-functionalized co-condensed silica particles.
Description
NITRIC OXIDE-RELEASING S-NITROSOTHIOL-MODIFIED SILICA
PARTICLES AND METHODS OF MAKING THE SAME
CROSS-REFERENCE TO RELATED APPLICATIONS
This application claims priority from U.S. Provisional Application Serial No.
61/447,368, filed February 28, 2011, and U.S. Provisional Application No. 61/565,694, filed December 1, 2011, the disclosure of each of which is hereby incorporated by reference herein in its entirety.
STATEMENT OF GOVERNMENT SUPPORT
The present invention was funded in part by government support under grant number 5-R01 -EB000708 from the National Institutes of Health. The United States Government has certain rights in this invention.
FIELD OF THE INVENTION
The present invention relates to nitric oxide-releasing particles. More particularly, the present application relates to S-nitrosothiol-modified silica particles.
BACKGROUND OF THE INVENTION
Since the discovery of the physiological roles of nitric oxide (NO), much research has focused on the synthesis of NO-releasing materials/vehicles to elicit NO's characteristics as an antimicrobial agent, mediator of wound repair, or angiogenic cofactor. S-Nitrosothiols (RSNOs) are one class of endogenous NO donor believed to store/transport the majority of the body's natural reservoir of NO. As such, a large body of work has utilized low molecular weight RSNOs (e.g., S-nitroso-glutathione (GSNO), S-nitroso-iV-acetylcysteine (SNAC), and S-nitroso-N-acetyl-penicillamine (SNAP)) as donors to spontaneously release NO. Although promising, the clinical application of low molecular weight NO donors has been slow due to both lack of tissue specific targeting and uncontrollable NO release kinetics. To address such shortcomings, NO donor precursors have been conjugated to larger scaffolds (e.g., proteins, dendrimers, and nanoparticles), thus enabling high NO storage per delivery vehicle and release profiles similar to their small molecule analogues.
Silica particles are among the most widely employed macromolecular scaffolds for biomedical applications due to facile synthetic strategies and minimal cytotoxicity.
Previously, the surface of fumed silica particles (7-10 nm diameter) have been grafted with
SNAP, SNAC, and S-nitrosocysteine (CysNO) to create S-nitrosothiol-modified silica particles. However, the NO storage was limited to 0.021-0.138 μπιοΐ mg"' because the thiol functionalization was restricted to the exterior of the particle. Additionally, these systems are not able to tune particle size to fit a therapeutic system of interest. Alternatively, the hydrolysis and co-condensation of organosilane and tetraalkoxysilane precursors via sol-gel chemistry may represent a method for preparing a silica network with a higher concentration of organic functionalites. Indeed, the Stober process (sol-gel chemistry with an alcohol solvent and an ammonia catalyst) has proven effective for synthesizing N-diazeniumdiolate- modified silica particles of diverse size and NO storage capacity. See, for example, U.S. Publication No. 2009/0214618 (Schoenfisch et al.), which is herein incorporated by reference in its entirety. The advantage of the Stober method over surface grafting is that the co- condensation provides uniform incorporation of the organic (i.e., NO donor) functionality throughout the resulting silica network as opposed to restricted functionalization at the surface alone. As a result, such particles may exhibit significantly increased NO storage.
SUMMARY OF THE INVENTION
A first aspect of the present invention comprises a method of forming
S-nitrosothiol-functionalized co-condensed silica particles comprising:
reacting a thiol-containing silane and a backbone alkoxysilane in a sol precursor solution that comprises water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles comprise a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix; and
reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent to provide the S-nitrosothiol-functionalized co-condensed silica particles.
A second aspect of the present invention comprises S-nitrosothiol-functionalized monodisperse co-condensed silica particles having an average particle diameter in a range of about 10 nni to about 100 μηι.
A further aspect of the present invention comprises S-nitrosothiol-functionalized co- condensed silica particles having an NO storage in a range of about 0.01 μηιοΐ to about 10 μιτιοΐ NO per mg particle.
Another aspect of the present invention comprises a S-nitrosothiol-functionalized co- condensed silica particle comprising tertiary nitrosothiol functional groups.
wherein R, R' and R" are each independently alkyl and n is 0 in a range of 0 to 10.
BRIEF DESCRIPTION OF THE DRAWINGS
The accompanying drawings, which are included to provide a further understanding of the invention and are incorporated in and constitute a part of this application, illustrate certain embodiment(s) of the invention.
Figure 1 provides a scheme for the synthesis of N-acetylpenicillamine
propyltrimethoxysilane (NAPTMS).
Figure 2 provides an Ή NMR spectrum of the NAPTMS compound.
Figure 3 shows a solid-state cross polarization/magic angle spinning (CP/MAS) "Si NMR spectra of silica synthesized with (A) 0, (B) 25, (C) 40, (D) 60, (E) 75, and (F) 85 mol % MPTMS (balance TMOS). The Q and T bands have been designated.
Figure 4 shows the scanning electron micrographs of (A) 25, (B) 40, (C) 60, (D) 75 and (E) 85 mol% MPTMS (balance TMOS) and (F) 75 and (G) 85 mol % MPTMS (balance TEOS) particles synthesized with 16.0 M water, 5.5 M ammonia, and 0.1 M silane.
Figure 5 shows the scanning electron micrographs of (A) 25, (B) 40, (C) 60, (D) 75 and (E) 85 mol% MPTMS (balance TMOS) and (F) 75 and (G) 85 mol % MPTMS (balance TEOS) particles synthesized via a semi-batch process with a silane feed rate of 0.5 rriL min"1.
Figure 6 shows the scanning electron micrographs of 75 mol% MPTMS (balance TEOS) synthesized with (A) 47.0, (B) 42.0, (C) 40.6, (D) 36.5, (E) 32.5, and (F) 24.9 M water,
Figure 7 shows the scanning electron micrographs of (A-B) 25, (C-D) 40, (E-F) 60, (G-H) 75, and (I-J) 85 mol% MPTMS (balance TMOS) and (K-L) 85 mol % MPTMS (balance TEOS) particles synthesized with (A,C,E,F,G,I,K) 32.5 and (B, D, F, H, J, L) 24.9
M water.
Figure 8 shows a schematic for the S-nitrosothiol decomposition pathways.
Figure 9 shows the nitric oxide release from RSNO-modified 75 mol% MPTMS
(balance TEOS) particles in the presence of (A) 0 (— ), 60 (- -), 100 (· · ·), and 200 (- ·) W
irradiation at 0 °C and (B) 0 (— ), 10 (- -), and 25 (· · ·) μΜ CuBr2/PBS solution at 0 °C. Note: 0 μΜ CuBr2 is 500 μΜ DTPA (pH 7.4 PBS). The inset of A provides an enlarged view of NO release profile without irradiation.
Figure 10 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention prior to sonication.
Figure 11 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention after 30 minutes sonication followed by nitrosation.
Figure 12 shows SEM images of tertiary thiol-functionalized co-condensed silica particles according to some embodiments of the invention after 60 minutes sonication followed by nitrosation.
DETAILED DESCRIPTION OF EMBODIMENTS OF THE INVENTION
The foregoing and other aspects of the present invention will now be described in more detail with respect to the description and methodologies provided herein. It should be appreciated that the invention can be embodied in different forms and should not be construed as limited to the embodiments set forth herein. Rather, these embodiments are provided so that this disclosure will be thorough and complete, and will fully convey the scope of the invention to those skilled in the art.
The terminology used in the description of the invention herein is for the purpose of describing particular embodiments only and is not intended to be limiting of the invention. As used in the description of the embodiments of the invention and the appended claims, the singular forms "a", "an" and "the" are intended to include the plural forms as well, unless the context clearly indicates otherwise. Also, as used herein, "and/or" refers to and encompasses any and all possible combinations of one or more of the associated listed items. Furthermore, the term "about," as used herein when referring to a measurable value such as an amount of a compound, dose, time, temperature, and the like, is meant to encompass variations of 20%, 10%), 5%>, 1%, 0.5%), or even 0.1%o of the specified amount. It will be further understood that the terms "comprises" and/or "comprising," when used in this specification, specify the presence of stated features, integers, steps, operations, elements, and/or components, but do not preclude the presence or addition of one or more other features, integers, steps, operations, elements, components, and/or groups thereof. Unless otherwise defined, all terms, including technical and scientific terms used in the description, have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
All patents, patent applications and publications referred to herein are incorporated by reference in their entirety. In the event of conflicting terminology, the present specification is controlling.
The embodiments described in one aspect of the present invention are not limited to the aspect described. The embodiments may also be applied to a different aspect of the invention as long as the embodiments do not prevent these aspects of the invention from operating for its intended purpose.
Chemical Definitions
As used herein the term "alkyl" refers to C1-2o inclusive, linear (i.e., "straight-chain"), branched, or cyclic, saturated or at least partially and in some cases fully unsaturated (i.e., alkenyl and alkynyl) hydrocarbon chains, including for example, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, octyl, ethenyl, propenyl, butenyl, pentenyl, hexenyl, octenyl, butadienyl, propynyl, butynyl, pentynyl, hexynyl, heptynyl, and allenyl groups. "Branched" refers to an alkyl group in which a lower alkyl group, such as methyl, ethyl or propyl, is attached to a linear alkyl chain. Exemplary branched alkyl groups include, but are not limited to, isopropyl, isobutyl, tert-butyl. "Lower alkyl" refers to an alkyl group having 1 to about 8 carbon atoms (i.e., a Ci-8 alkyl), e.g., 1 , 2, 3, 4, 5, 6, 7, or 8 carbon atoms. "Higher alkyl" refers to an alkyl group having about 10 to about 20 carbon atoms, e.g., 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 carbon atoms. In certain
embodiments, "alkyl" refers, in particular, to C1-5 straight-chain alkyls. In other embodiments, "alkyl" refers, in particular, to C1-5 branched-chain alkyls.
The term "mercapto" or "thio" refers to the -SH group.
Provided herein according to some embodiments of the invention are methods of forming co-condensed silica particles via the Stober process. See, e.g., Stober, W.; Fink, A.; Bohn, E. J. Colloid Interface Sci. 1968, 26, 62 (incorporated by reference herein in its entirety). Particle formation under the Stober process proceeds upon hydrolysis and condensation of silane precursors where the relative hydrolysis rates for the precursors dictate both the speed of particle growth and the likelihood of each silane' s incorporation into the silica network. Excessive disparities between reaction rates of different silanes may lead to absence of particle formation upon attempted co-condensation.
Provided according to some embodiments of the invention are methods of forming S- nitrosothiol-functionalized co-condensed silica particles that include reacting a thiol-
containing silane and a backbone alkoxysilane in a sol precursor solution that includes water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles include a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix. In some embodiments of the invention, the methods further include reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent to provide the S-nitrosothiol-functionalized co-condensed silica particles.
Any suitable thiol-containing silane may be used. In some embodiments, the thiol- containing silane includes a primary thiol, in some embodiments, a secondary thiol, and in some embodiments, a tertiary thiol. Combinations of different silanes may also be used. A suitable thiol-containing silane will be a silane that will allow for particle formation, and in some embodiments, monodisperse particle formation. Thus, some thiol-containing silanes may be suitable with some backbone alkoxysilanes and not suitable with others. In some embodiments, the primary thiol-containing silane is mercaptopropyltrimethoxysilane. In some embodiments, the tertiary thiol alkoxysilane has the following structure:
(OR)(OR')(OR")Si(Rx), wherein R, R' and R" are each independently H, alkyl or substituted alkyl and Rx is functional group that comprises a tertiary thiol group. In particular embodiments, the tertiary thiol alkoxysilane has the structure:
wherein R, R' and R" are each independently H, alkyl or substituted alkyl and n is 0-10. In some embodiments, R, R' and R" are each independently alkyl and n is 0-5. Furthermore, in particular embodiments of the invention, the tertiary thiol is a compound having the structure:
"backbone alkoxysilane" refers to an alkoxysilane that does not contain a thiol functional group. Examples include tetramethoxysilane, tetraethoxysilane, methyltrimethoxysilane, methyltriethoxysilane and butyltrimethoxysilane. A suitable backbone silane will be a silane that will allow for particle formation, and in some embodiments, monodisperse particle formation.
Any suitable concentration of water may be used. In some embodiments, the water concentration in the sol precursor solution is in a range of about 8 M to about 32.5 M.
In some embodiments, a catalyst, such as an ammonia catalyst, is included in the sol precursor solution. Any suitable concentration of catalyst may be used. However, in some embodiments, an ammonia catalyst is included in the sol precursor solution, in some embodiments, at a concentration in a range of about 1.9 M to about 5.5 M.
In particular embodiments of the invention, mercaptopropyltrimethoxysilane and tetramethoxysilane are reacted in the presence of water and an ammonia catalyst to form thiol-functionalized co-condensed silica particles. In some embodiments, the reaction occurs in a solution that includes mercaptopropyltrimethoxysilane and tetramethoxysilane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 25 to about 85 mol % mercaptopropyltrimethoxysilane. In some embodiments, water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 M to about 5.5 M.
In some embodiments of the invention, mercaptopropyltrimethoxysilane and tetraethoxysilane are reacted in the presence of water and an ammonia catalyst to form thiol- functionalized co-condensed silica particles. In some embodiments, the reaction occurs in a solution that includes mercaptopropyltrimethoxysilane and tetraethoxysilane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 75 to about 85 mol % mercaptopropyltrimethoxysilane. In some embodiments, water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 M to about 5.5M.
In some embodiments, methods of forming thiol-functionalized co-condensed silica particles include reacting a tertiary thiol-containing silane and a backbone alkoxysilane in the presences of water and an ammonia catalyst to form thiol-functionalized co-condensed silica particles. In some embodiments, the reaction occurs in a solution that includes tertiary thiol-
containing silane and alkoxysiiane at a total silane monomer concentration in a range of about 0.1 M to about 0.4 M, wherein the silane monomer includes about 25 to about 85 mol % tertiary thiol-containing silane. In some embodiments, water is present in the solution at a concentration in a range of about 8.0 to about 32.5 M and the ammonia catalyst is present at a concentration in a range of about 1.9 to about 5.5 M.
The sol precursor solution may also be stirred/agitated as known to those of skill in the art, and other additives or silane monomers used in sol chemistry may be included in some embodiments of the invention.
In some embodiments of the invention, methods provided herein may be used to form nitrosothiol-functionalized co-condensed silica particles, which in some embodiments, are monodisperse. As used herein, the term "monodisperse" refers to particles having a uniform particle size, in some embodiments, having an average particle diameter ± 100 nm as measured from electron micrographs; a Z-average ± 60 nm as measured from dynamic light scattering; and/or having a polydispersity index <0.1 as measured via dynamic light scattering. In some embodiments, the methods described herein provide monodisperse particles having an average particle diameter of less than 100 microns, and in some embodiments, less than 1 micron. In particular embodiments, the methods used herein may provide monodisperse particles having an average particle diameter in a range of about 10 nm to about 100 μηι. In some embodiments, the particles have an average particle diameter in a range of about 200 to about 700 nm.
Any suitable method of nitrosating the thiol-functionalized co-condensed silica particles may be used. Further, any suitable nitrosating agent may be used. However, in some embodiments, the nitrosating agent includes acidified sodium nitrite, alkyl nitrites, including tertbutyl nitrite and isopentyl nitrite, and various nitrogen oxides including nitrous oxide, N203, N 04 and N02. Examples of nitrosation may be found in Williams, D. L. H. Acc. Che . Res. 1999, 32, 869, the contents of which are incorporated herein by reference in their entirety.
In some embodiments of the invention, the nitrosation chemistry conserves particle size integrity and yields monodisperse S-nitrosothiol-functionalized co-condensed silica particles. No changes in particle size have been observed following addition of the nitric oxide functionality to the macromolecular structure, a drawback that has been observed with other nitrosothiol-modified macromolecular donors. Furthermore, as shown below in the Examples, the thiol-functionalized co-condensed silica particles may be sonicated prior to nitrosation without deleteriously affecting the NO storage and/or morphology of the particles.
The co-condensed silica particles may include S-nitrosothiol groups throughout the particle, and as such, may provide enhanced NO storage properties. For example, in some embodiments of the invention, provided are S-nitrosothiol-functionalized co-condensed silicas particles that have an NO storage in a range of about 0.01 μπιοΐ to about 10 μηιοΐ NO per mg particle, and in some embodiments, 0.09 μιηοΐ to about 4.40 μιηοΐ NO per mg particle.
The incorporation of the S-nitrosothiol groups throughout the interior of the silica particle structure may also afford unexpected stability. Glutathione and other thiols are known to one skilled in the art to be a vial trigger for RSNO decomposition and release a variety of NOx species. In some embodiments of the invention, the low porosity of the S- nitrosothiol-functionalized co-condensed silica particles protect the RSNO donors from premature decomposition by glutathione or other blood components, adding a level of nitric oxide stability when used in drug delivery applications.
EXAMPLES
Preparation of NAPTMS
Synthesis ofN-Acetyl Penicillamme (NAP) Thiolactone. Acetic anhydride (96 mmol, 9. 80 g) was added dropwise to a well stirred solution of D-(-) penicillamine (40 mmol, 5. 97 g) in pyridine (50 mL) at 0 °C. After 30 min, the flask was removed from ice and allowed to stir at room temperature for 15 h. The resultant orange solution was partitioned between chloroform and dilute HCl and washed 4x with dilute HCl. After drying over MgS04, the organic phase was evaporated to yield an orange residue. The residue was first dissolved in absolute ethanol (20 mL), and then precipitated in pentane at -78 °C. The light yellow crystalline product was isolated by filtration (2. 07 g, 30%). Ή NMR (CDC13) δΐ . 65 (s, C¾), 1. 86 (s, C/J3), 2. 05 (s, NHCOC¾), 5. 68-5. 70 (d, C/J(CH3)2), 6. 56 (N COCH3). 13C NMR (CDC13) δ 22. 52 (NHCO H3), 26. 20 ( H(CH3)2), 30. 22 (CH(CH3)2), 51. 23 (CH), 169. 37 (NHCOCH3), 192. 21 (SCO).
Synthesis ofN-Acetyl Penicillamme Propyltrimethoxysilane (NAPTMS). APTMS (10 mmol, 1. 78g). was added to a stirring solution of NAP thiolactone (10 mmol, 1. 72 g) in methylene chloride (20 mL). The light yellow solution was stirred for 4 h at room
temperature before distillation of the methylene chloride to yield NAPTMS as a viscous clear oil. Ή NMR (CDC13) δ 0. 54 (t, SiCH2), 1. 24 and 1. 39 (s, CH(C/ 3)2SH), 1. 54 (m,
SiCH2CH2), 1 . 96 (s, NHCOCH3), 2. 96 and 3. 21 (m, SiCH2CH2CH2), 3. 44 (s, Si(OC/J3)3), 4. 63 (d, CHC(CH3)2SH), 6. 99 (d, CHNHCOCH3), 7. 70 (t, CH2NHCOCH). , 3C NMR
(CDCI3) δ D 6. 59 (Si H2), 22. 42 and 22. 97 (CH(C¾)2SH), 28. 64 (NHCOCH3), 30. 80 (SiCH2CH2), 41 . 93 (CHC(CH3)2SH), 46. 23 (SiCH2CH2 H2), 50. 35 (Si(OCH3)3), 60. 32 (CHC(CH3)2SH), 169. 64 (CHNHCOCH3), 170. 17 (CHCONH).
The preparation of tertiary thiol-based precursors was investigated for the
development of biomedical devices/therapeutics with continuous and photoactivatable NO release. A NAP thiolactone was thus synthesized to design such a precursor for the synthesis of NO-releasing xerogels. Penicillamine was reacted in the presence of acetic anhydride to generate the NAP thiolactone in situ. After characterization by Ή and l 3CNMR, the NAP thiolactone was directly coupled with APTMS to result in a tertiary thiol-bearing silane, referred to as NAPTMS (see Figure 1). Successful synthesis of this tertiary thiol-bearing silane was verified via 'HNMR characterization (Figure 2).
Example 1 : MPTMS
Ratios of mercaptosilane and alkoxysilane (25-85 mol% MPTMS, balance TMOS or TEOS) were added either as a bolus injection or dropwise via a Kent Scientific Genie Plus syringe pump at a flow rate of 0.25-3.0 mL/min through an 18.5 gauge needle to a solution of ethanol, water, and ammonium hydroxide. Solution was stirred for 2 h at room temperature, collected via centrifugation at 4500 rpm (10 mins), washed twice with 40 n L EtOH, recollected, and dried overnight at ambient conditions.
Our initial attempt to synthesize thiol-containing silica particles was based on a bolus injection of 3-mercaptopropyltrimethoxysilane (MPTMS) and alkoxysilane into
EtOH/NH4OH solution. The resulting concentrations of ammonia, water and total silane were 3.3, 8.0, and 0.2 M, respectively. Tetramethoxysilane (TMOS) proved to be a sufficient backbone silane for co-condensation with MPTMS as their combination (at various mole percentages) resulted in the formation of a white precipitate. (~300 mg yield).
As indicated by solution turbidity, a marked increase in reaction time was observed upon increasing the concentration of MPTMS up to 85 mol%. At this concentration, the time to form a visible product after combining the silanes was roughly 15 min. Product formation at MPTMS concentrations >85 mol% was not observed, The inability to form particles at greater MPTMS concentrations may be attributed to the disparate hydrolysis rates between
the silanes, suggesting that co-condensation requires a minimum concentration of the more readily hydrolyzable silane (i.e., TMOS) to initiate particle growth,
Materials synthesized via the co-condensation of MPTMS and tetraethoxysilane (TEOS) formed only in the concentration range of 75-85 mol% MPTMS. In contrast to the TMOS system, products with lower concentrations of MPTMS (e.g., 25 mol%) did not form using TEOS as a backbone, even at prolonged reaction times (up to 48 h).
Example 2: MPMDMS
Another thiol-functionalized monomer, 3-mercaptopropylmethyldimethoxysilane (MPMDMS), was also investigated. Unfortunately, the product yield (~5 mg) formed using MPMDMS with either TMOS or TEOS was significantly lower than MPTMS. The substitution of one of the hydrolyzable methoxy groups with a nonhydrolyzable methyl linkage in MPMDMS (vs. MPTMS) appears to decrease the resulting hydrolysis rate under basic conditions, possibly due to the inductive effect of electron density donation to the Si atom. As a result, the reaction with hydroxide anion to hydrolyze the silane may be inhibited. Particle formation may even be further limited as each MPMDMS molecule is capable of forming only two siloxane bridges. Consequently, particle formation using MPMDMS was unsuccessful.
Example 3: Characterization of first generation
mercaptosilane-based silica particles
Solid-state cross polarization/magic angle spinning (CP/MAS) 29Si (71.548 MHz frequency) nuclear magnetic resonance (NMR) spectroscopy was performed on a Bruker 360 MHz DMX spectrometer (Billerica, MA). Particles were packed into 4 mm rotors and spun at 8.0 kHz. Spectra were collected at 5000 scans with the determination of chemical shifts in parts per million relative to an external TMS standard. Nitric oxide release was measured in real time (1 sec intervals) using 5 a Sievers NOATM 280i Chemiluminescence Nitric Oxide Analyzer (NOA) (Boulder, CO). Calibration of the NOA was performed with both air passed through a Sievers NO zero filter and 26.39 ppm NO gas (balance N2). Nitric oxide-releasing particles were immersed in 25 mL of deoxygenated solution and sparged with an 80 mL min- 1 N2 stream. Additional N2 was supplied to the reaction flask to match the collection rate of the NOA at 200 mL min-1.
Temperature control was maintained using a water bath at 37 °C. Thermal and photo- initiated NO release were studied by conducting the experiments in 500 μΜ DTP A (pH 7.4 PBS) to chelate trace copper and illuminating the sample flask with 60, 100, and 200 W incandescent bulbs, respectively. Copper-initiated NO release was studied by adding the particles to 25 n L of 10 or 25 μΜ CuBr2(aq). The NOA sample flask was shielded from light with aluminum foil for experiments where light was not the intended initiator of NO release. Particle size was determined using a Zetasizer Nano ZS Particle Size and Zeta Potential Dynamic Light Scattering (DLS) Instrument (Malvern, UK). Samples were suspended in PBS at a concentration of 1 mg mL-1 and sonicated for 15 min prior to analysis. Scanning electron micrographs were recorded on a Hitachi S-4700 Scanning Electron Microscope (Pleasanton, CA).
To confirm the incorporation of mercaptosilane within the silica network and compare various compositions, solid-state Si cross polarization/magic angle spinning nuclear magnetic resonance (CP/MAS NMR) was used to characterize the MPTMS/TMOS products as a function of MPTMS concentration. Silicon atoms of tetraalkoxysilanes appear in the NMR spectra as Q" bands while those of organotrialkoxysilanes appear as Tn bands. In both cases, n denotes the number of siloxane bonds attached to the Si atom. The greater number of siloxane bonds to the Si atom, the further the NMR band shifts upfield. Figure 3 shows silica synthesized with (A) 0, (B) 25, (C) 40, (D) 60, (E) 75, and (F) 85 mol % MPTMS (balance TMOS). Particles synthesized entirely from TMOS exhibited only Q bands. With increasing MPTMS concentration in the solution used to prepare the particles, the T bands increased relative to the Q bands, confirming greater incorporation of MPTMS in the silica particle.
Sulfur weight percent of each composition was determined using elemental analysis and further corroborated the covalent incorporation of the mercaptosilane. The weight percent of sulfur in the silica was 4.92, 7.1 1 , 11.67, 13.56 and 17.30 for the 25, 40, 60, 75 and 85 mol% MPTMS (balance TMOS) compositions, respectively. The TEOS-based particles were found to have sulfur weight percents of 16.15 and 19.34 for 75 and 85 mol% MPTMS, respectively. As expected, the weight percent of sulfur increased linearly with increasing MPTMS concentration in the initial solution.
Dynamic light scattering (DLS) measurements indicated that the sample was too polydisperse to accurately measure the particle size. Scanning electron micrographs (SEMs)
further indicated that the thiol-containing silica was polydisperse and exhibited nonspherical morphology more indicative of colloidal silica than individual particles, (data not shown).
Example 4: Variation of water, ammonia and silane concentrations and feed rate
We systematically varied synthetic parameters (i.e., water, ammonia, and silane concentrations) to tune the resulting particle morphology and achieve a more spherical shape. The composition of 25 mol % MPTMS (balance TMOS) was chosen as the model system for comparison due to minimal organic character.
For MPTMS particles, we found that increasing the water content from 8.0 to 16.2 M promoted the formation of spherical particles and prevented aggregation/fusion. Lower ammonia concentrations were shown to result in particles that lacked spherical shape and aggregated. Thus, we discovered that the ratio of water and ammonia to silane was a critical factor during particle synthesis. Upon considering all the data, we determined that the most spherical and monodisperse particles were the 25 mol% MPTMS (balance TMOS) particles were formed using 5.5 M ammonia, 0.1 M total silane, and 16.2 M water. Of note, the product yield (~70 mg) with this synthesis was lower than that obtained for the polydisperse colloidal silica. The decreased yield was due to the 4-fold decrease in the silane concentration used in the optimized synthesis.
Next, the concentration of MPTMS in the solution was increased to enhance the degree of thiol functionality and potential NO storage of the particles. Figure 4 depicts the resulting particles as the concentration of MPTMS was increased from 25-85 mol% and backbone alkoxysilane varied between TMOS and TEOS. The particles were synthesized with 16.0 M water, 5.5 M ammonia, and 0.1 M silane.
As with the polydisperse colloidal silica system, the formation of particles was not observed for 25-60 mol% MPTMS (balance TEOS). Only 75 and 85 mol% MPTMS concentrations yielded particles with TEOS, illustrating how disparities in hydrolysis and condensation kinetics adversely affect and hinder particle formation. The 75 mol% MPTMS (balance TEOS) particles formed in a narrow size distribution and exhibited spherical morphologies (Fig. 4F). In contrast, 85 mol% MPTMS (balance TEOS) particles appeared aggregated (Fig. 4G). When using TMOS, 25 mol% MPTMS was the only concentration that yielded spherical, monodisperse particles (Fig. 4A). Particles with >40 mol% MPTMS
(balance TMOS) exhibited ideal morphologies, but with concomitant bimodal size distributions (Fig. 4B-E).
To remedy the bimodal size distribution that was observed for certain MPTMS compositions, we evaluated the effect of a silane feed rate of 0.5 mL min"1 on particle morphologies throughout the range of compositions (Figure 5). The slower feed rate improved the dispersity of the already narrow size distribution for 25 mol % MPTMS (Fig 5A). A pronounced improvement in the monodispersity was also noted for 40 mol %
MPTMS (balance TMOS, Fig. 5B), with SEM indicating a particle diameter of 293 ± 24 nm. Slower silane feed rates (e.g., 0.25 mL min"1) resulted in slight monodispersity improvements (data not shown), but at lower yields (e.g., -40 vs. 70 mg for 40 mol% MPTMS (balance TMOS) composition). Thus, 0.5 mL min"1 was determined to be the optimal feed rate as it allowed for a balance between sufficient particle yield and monodispersity. Similar to 25 mol% MPTMS (balance TMOS), the monodispersity of 75 mol% MPTMS (balance TEOS) improved, while the 85 mol% MPTMS (balance TEOS) system remained aggregated (Fig 5F and 5G, respectively).
Additionally, the product yield increased to -170 mg for these two compositions and can be attributed to the greater concentration of the larger MPTMS in the particles.
Unfortunately, the semi-batch process proved problematic for 60, 75, and 85 mol% MPTMS (balance TMOS) particles. As shown in Figure 5C-E, the slowed silane addition resulted in both aggregation and the formation of a large silica network rather than monodisperse, spherical particles. To examine this phenomenon further, silane feed rates were varied (0.25- 3.0 mL min"1) for 60 mol% MPTMS (balance TMOS). Feed rates <2.0 mL min"1 resulted in polydisperse, aggregated silica, while faster feed rates (2.0-3.0 mL min"1) produced particles of a bimodal size (data not shown).
We then attempted to decrease the size of the particles to improve particle
monodispersity. The 75 mol% MPTMS (balance TEOS) particles were chosen as a model system to examine the effect of the water concentration on particle size and morphology. As shown in Figure 6, 75 mol% MPTMS (balance TEOS) was synthesized with (A) 47.0, (B) 42.0, (C) 40.6, (D) 36.5, (E) 32.5, and (F) 24.9 M water. Water concentrations >40.6 M favored rapid silane hydrolysis and condensation kinetics, leading to a highly condensed network rather than discrete, spherical particles. At a water concentration of 36.5 M, discrete particles were formed, but with morphologies featuring excessive aggregation. Monodisperse particles (333 ± 48 nm) were first observed at a slightly lower water concentration (32.5 M).
Particle size increased with decreasing water concentrations (456 ± 68 nm and 635 ± 63 nm for 24.9 and 16.2 M, respectively). Furthermore, the smaller particle sizes were accompanied with slightly lower yields for each composition. The yields for 75 mol% MPTMS (balance TEOS) particles were ~65, 150, and 170 mg for water concentrations of 32.5, 24,9, and 16.2 M, respectively. The differences in yield may be factors of the efficiency of particle collection (i.e., centrifugation rpm and duration) for the smaller particles rather than chemical differences.
The appropriate water concentrations (32.5 and 24.9 M) were next used to tune particle size and reduce the bimodal distribution characteristic of the 60, 75, and 85 mol% MPTMS (balance TMOS) particles. As shown in Figure 7, the intermediate water concentration (24.9 M) yielded particles with sizes of 179 ± 22 and 196 ± 25 nm for the 25 and 40 mol % MPTMS (balance TMOS) compositions, respectively. The greater water concentration (32.5 M) drastically increased the reaction kinetics for the mostly TMOS-based systems, resulting in highly-fused silica networks. Increasing the concentration of MPTMS (75 mol%) yielded monodisperse, spherical particles of 363 ± 51 and 279 ± 49 nm using 24.9 and 32.5 M water, respectively. Aggregated and fused particles were formed for the greatest MPTMS concentration (85 mol%) when using 24.9 M water. However, monodisperse particles (440 ± 84 nm) were formed when synthesized with 32.5 M water.
The TEOS-based counterpart to this system behaved similarly by yielding only discrete particles (506 ± 77 nm) when synthesized with the higher water concentration. At lower water amounts, the formation of aggregated particles was noted. The trend of decreasing particle yield with increasing water content that was observed for the 75 mol% MPTMS (balance TEOS) composition was mirrored for all other compositions evaluated. The yields for the 75 mol% (balance TMOS) particles decreased from -120 to 60 mg upon increasing the water content from 24.9 to 32.5 M. Likewise, the 25 mol% MPTMS (balance TMOS) particle yield decreased from -90 to 20 mg while the 40 mol% MPTMS system exhibited a decrease from -50 to 9 mg upon increasing the water concentration from 16.2 to 24.9 M. The yields for both 85 mol% MPTMS compositions (i.e, TMOS and TEOS balance) at a water concentration of 32.5 M were -160 mg. Perhaps of greatest significance, the elevated water concentrations used to synthesize the thiol-modified particles successfully resolved the bimodal nature of certain compositions not resolvable using a semi-batch process alone. Of note, 60 mol% MPTMS (balance TMOS) was the only composition that
consistently yielded particles of a bimodal nature. Increasing the water content regardless of addition method (bolus vs. semi-batch) resulted in a highly-fused silica network.
Particle sizes were also measured by DLS to corroborate particle monodispersity and size measured using SEM. As shown in Table 1, the DLS measurements were in agreement with the sizes calculated from the SEM images. The slightly increased average diameters observed with DLS may be attributed to particle hydration (DLS measurements conducted in solution). Like SEM, the DLS measurements indicated a narrow size distribution, as evidenced by low polydispersity indices for each composition.
TABLE 1
Particle composition Water content Particle size3 Z-Average sizeb Polydispersity
(mol % MPTMS) (M) (nm) (nm) index
75% (balance TEOS) 32.5 333 + 48 416.2 ± 23.4 0.027
75% (balance TEOS) 24.9 456 ± 68 529.6 ± 23.7 0.018
75% (balance TEOS) 16.2 635 ± 63 718.0 + 51.7 0.046
85% (balance TEOS) 32.5 506 ± 77 668.7 ± 46.0 0.040
25% (balance TMOS) 24.9 179 ± 22 258.4 ± 15.1° 0.031
25% (balance TMOS) 16.2 252 ± 20 469.0 ± 24.8° 0.025
40% (balance TMOS) 24.9 196 ± 25 240.7 ± 17.9° 0.064
40% (balance TMOS) 16.2 293 ± 24 404.8 ± 28.2 0.045
75% (balance TMOS) 32.5 279 ± 49 431 .2 ± 29.5 0.043
75% (balance TMOS) 24.9 363 ± 51 507.6 ± 30.8 0.032
85% (balance TMOS) 32.5 440 ± 84 696.2 ± 44.4 0.042
aSlze calculated from scanning electron micrographs of n = 120 particles
bSizes acquired from dynamic light scattering measurements in pH 7.4 PBS for n
"Ethanol used as dlspersant
Of note, PBS was used as a dispersant for compositions with a large concentration of MPTMS. However, smaller particles with a large degree of inorganic character (i.e., <40 mol% MPTMS) rapidly aggregated in this dispersant and caused erratic DLS measurements. This aggregation may be attributed to a large surface density of protonated silanol groups leading to unfavorable particle interaction. While basic conditions resulted in inconsistent DLS measurements due to particle dissolution, ethanol was a viable alternative dispersant as evidenced by the correlation between DLS and SEM measurements.
Elemental analysis was used to characterize the composition of the particles. As expected, the weight percentages of sulfur in the particles increased accordingly with the MPTMS mol% used to make the particles indicating incorporation of the thiol functionality (Table 2).
TABLE 2
Particle com position W ater content S ulfur content3 (mol % MPTMS) (M) (wt % )
75% (balance TEOS) 32 5 13.83 ± 3.01
75% (balance TEOS) 24 9 16.01 ± 1.71
75% (balance TEOS) 16 2 15.62 ± 1.90
85% (balance TEOS) 32 5 20.02 ± 3.88
25% (balance TMOS) 24 9 < 0.0
25% (balance TMOS) 16 2 0.51 ± 0.36
40% (balance TMOS) 24 9 1.09 ± 0.58
40% (balance TMOS) 16 2 3.08 ± 2.57
75% (balance TMOS) 32 5 18.29 ± 5.34
75% (balance TMOS) 24 9 15.30 ± 5.32
85% (balance TMOS) 32 5 20.55 ± 5.70
"Average weight percents are calculated from n = 3 syntheses
Weig t percent was less than ins trument lim it of detection
Syntheses promoting the formation of discrete, spherical particles tended to be preferentially derived from one precursor as evidenced by a large gap in the transition from 40 to 75 mol% MPTMS (wt% 3.08 ± 2.57 and 15.62 ± 1.90, respectively). These values were in marked contrast to the sulfur wt% of the colloidal silica. Although the increased sulfur wt% were more linearly proportional for the colloidal silica, the lack of discrete, spherical particles was not ideal. The comparison of the two systems (colloidal vs. discrete particles) and syntheses reveals that a balance exists between silane incorporation and certain design criteria.
Example 5: Synthesis of Particles with NAPTMS
Procedure for 25% NAPTMS balance TMOS/TEOS:
1. Dissolved 85.4 mg NAPTMS (tertiary precursor) in 3.95 mL of ethanol by vortexing
2. To the reaction mixture added 4.09 mL of water then added TMOS/ethanol mixture (71.9 μΐ, TMOS and 200 μί, ethanol) via syringe pump at a rate of 1.0 mL/min.
3. Added 6 mL of 5M HCL and let sonicate (120%) for lhour.
4. Added 4 mL of ammonium hydroxide and allowed to sonicate (120%) for 30 minutes,
Final Concentrations TMOS and TEOS Particles:
[Silane] = 0.0352 M [Ethanol] = 3.85 M
[Water] = 42.8 M [NH3] = 3.43 M
[HC1] = 1.57 M
Size Characterization:
Example 6: Nitrosation of mercaptosilane-based silica particles
Thiols within the particles were nitrosated via reaction with nitrous acid.12 Particles (-200 mg) were first added to 4 mL methanol (MeOH). While stirring, 2 mL of hydrochloric acid (5 M) was added to the suspension. A 2 mL aqueous solution containing sodium nitrite (2x molar excess to thiol) and DTPA (500 μΜ) was then added to the particle suspension, and the mixture stirred for 2 h in the dark and on ice. Particles were collected by centrifugation at 4500 rpm (5 min), washed with 40 mL chilled 500 μΜ DTPA(aq), recollected, washed with 40 mL chilled MeOH, recollected, and vacuum dried for 30 min while shielded from light. Particles were stored at -20 °C in vacuo until further study.
The MPTMS-modified particles were nitrosated to enable NO storage and release. Briefly, the particles were treated with acidified sodium nitrite, generating nitrous acid, a nitrosating agent that reacts with thiols to form RSNOs (see Eq 1).
RSH + HN02♦± RSNO + H20 (1)
Since S-nitrosothiols prepared from primary thiols absorb light at 330-350 and 550— 600 nm, successful RSNO formation was confirmed by the resulting red color of the particles after nitrosation. Furthermore, the intensity of the color increased with MPTMS mol% indicating greater RSNO formation.
As widely known, S-nitrosothiols decompose via a multitude of pathways (Figure 8). Both photo and thermal irradiation of RSNOs result in homolytic cleavage of the S-N bond, yielding NO and a thiyl radical. The thiyl radical may subsequently react with an RSNO to generate a disulfide and an additional equivalent of NO. Cu(I), resulting from the reduction of Cu(II) via trace thiolate ions, has been shown to be active in a catalytic RSNO
decomposition mechanism. Transnitrosation between a thiol and an RSNO may also occur, resulting in the transfer of the nitroso functionality and formation of a new RSNO species that may decompose via the aforementioned pathways.
To assess the NO storage and release, RSNO-modified particles (~2 mg) were added to 500 μΜ DTP A (pH 7.4 PBS) at a temperature of 0 °C, while measuring the ensuing NO release as a function of photolytic decomposition. As shown in Figure 9 A, RSNO-modified silica particles exhibited photo-initiated NO release upon exposure to broadband, white light. Greater irradiation levels (i.e., power) resulted in elevated NO release from the particles. Of note, low levels of NO release (~15 ppb mg"1 s"1) were observed at 0 °C and in the dark
(Figure 9A inset). Others have shown that oxygen may react with NO to form dinitrogen trioxide (N203), an oxidant that also decomposes RSNOs. Elimination of oxygen from the storage environment of the RSNO-modified particles would thus be expected to increase the NO storage stability of the particles. Indeed, no significant loss in NO release capacity was measured upon storing the particles for 2 months at -20 °C in vacuo and in the dark.
Due to the rapid kinetics of the photo-initiated decomposition, total NO storage of the particles was assessed by exposing the particles to 200 W of broadband light. Indeed, >95 % of the NO stored was released after 5 h of irradiation at 200 W. As given in Table 3, the total NO released from the particles ranged from 0.09-4.39 μιηοΐ mg"1. These levels of NO storage are an order of magnitude larger than previously reported RSNO-modified silica particles. Using the average sulfur weight percents in conjunction with the average NO storage values, the percent conversion of thiol to RSNO for the different particle compositions was calculated to be 58-78% for the 75 and 85 mol% MPTMS/TMOS and MPTMS/TEOS systems. The 25 and 40 mol% MPTMS particles were found to have lower thiol to RSNO conversions (54-63%).
TABLE 3
Particle composition Water content Total NO released3
(mol % MPTMS) (M) (μηιοΙ mg-1)
75% (balance TEOS) 32.5 3.24 ± 0.61
75% (balance TEOS) 24.9 3.58 ± 0.39
75% (balance TEOS) 16.2 3.15 ± 0.60
85% (balance TEOS) 32.5 3.95 ± 0.66
25% (balance TMOS) 24.9 0.09 ± 0.02
25% (balance TMOS) 16.2 0.10 ± 0.02
40% (balance TMOS) 24.9 0.34 ± 0.02
40% (balance TMOS) 16.2 0.52 ± 0.22
75% (balance TMOS) 32.5 3.31 ± 0.85
75% (balance TMOS) 24.9 3.73 ± 0.62
85% (balance TMOS) 32.5 4.39 ± 0.02
"Averages are calculated from n = 3 syntheses and after 5 h of 200 W irradiation
The effect of copper on NO release was investigated as a function of copper concentration. These assays were performed using Cu(II) via CuBr2 due to the insolubility of Cu(I) compounds in aqueous solutions. As expected, the NO release from the RSNO- modified particles correlated with the copper concentration (Fig 9B) with the greatest copper concentration examined (25 μΜ) generating the maximum NO release (-45 ppb mg"1 s"1).
The use of RSNO-modified particles for biomedical application likely necessitates an NO release trigger other than light or large concentrations of free copper ions. We thus evaluated NO release from the particles via thermal degradation at 37 °C using 75 mol% MPTMS (balance TEOS, 718.0 ± 51.7 nm) as a model system. Particles were introduced into 500 μΜ DTP A (pH 7.4 PBS), maintained at 37 °C and shielded from external light while monitoring NO release over 48 h (Table 4). Under these conditions, the particles released a total of 1.17 μιηοΐ NO mg"1 with a corresponding half life of 2.95 h. When compared to the total amount released after 5 h using 200 W irradiation (3.15 μιηοΐ mg"1 , Table 3), the discrepancy may be attributed to inability to measure NO at low levels beyond 48 h and/or loss of NO through its reaction with oxygen present in the soak solutions. As evident by a pinlc hue, the particles still contained a portion of their NO payload even after 48 h of release.
TABLE 4
Time Instantaneous NO release
(h) (ppb mg-1 S"1)a
0 1205.7 ± 22.4
0.5 481.2 ± 7.7
1 355.7 ± 7.7
6 74.9 ± 0.7
12 33.2 ± 0.4
24 12.6 ± 0.2
48 2.50 ± 0.07
aAverages are calculated from n = 3 syntheses
Example 7: Thermal Initiated NO Release Characterization of Primary and Tertiary
RSNO Particles
For each particle composition, approximately 3 mg of particles were added to the collection flask containing PBS (500μΜ DTP A) and the NO release monitored over 75 min. The NO storage and release characteristics are shown in Table 5.
TABLE 5
The NO storage and release characteristics of the 25% NAPTMS sample while under irradiation were also investigated. Using 200 W illumination, and 0.3 m distance, the results are shown in Table 6.
TABLE 6
The results shown in Tables 5 and 6 shown that NO stability of the particles can be significantly increased by using a tertiary nitrosothiol-functionalized silica particles.
Example 8: Influence of particle sonication before/after nitrosation
Experiment 8A: Nitrosated particles no Sonication
1. Nitrosate 15 mg of particles in methanol, 5M HC1, and 2 mol X (vs. thiol) of
NaNO2/500 uM DTP A.
2. Collect and wash with cold dtpa and cold methanol. Dry under vacuum for 45 min in dark (covered with foil).
3. Add 1 mg of nitrosated particles to 5 mL PBS (DTPA)
4. Expose to 200 W illumination (30 cm from inside bottom of box)
Experiment 8B: Particle nitrosation then Sonication
1. Nitrosate 15 mg of particles in methanol, 5M HC1, and 2 mol X (vs. thiol) of
NaNO2/500 uM DTPA.
2. Collect and wash with cold dtpa and cold methanol. Dry under vacuum for 45 min in dark (covered with foil).
3. Add 1 mg of nitrosated particles to 5 mL of PBS (DTPA) and sonicate for 30 min on ice at amplitude = 50%.
4. Expose to 200 W illumination (30 cm from inside bottom of box)
Experiment 8C: Particle sonication (30 min at amplitude = 50%) then nitrosation
1. Sonicate 15 mg of non-nitrosated particles in 4 mL of Methanol on ice for 30 minutes on ice at amplitude = 50%.
2. Nitrosatesonicated particles in 4 mL of methanol, 5M HCl, and 2 mol X (vs. thiol) of NaNO2/500uM DTPA.
3. Collect and wash with cold dtpa and cold methanol. Dry under vacuum for 45 min in dark (covered with foil).
4. Add 1 mg of nitrosated particles to 5 mL PBS (DTPA)
5. Expose to 200 W illumination (30 cm from inside bottom of box)
Experiment 8D: Particle sonication (60 min at amplitude = 50%o) then nitrosation
1 . Sonicate 15 mg of non-nitrosated particles in 4 mL of Methanol on ice for 60 minutes on ice at amplitude = 50%>.
2. Nitrosatesonicated particles in 4 mL of methanol, 5M HCl, and 2 mol X (vs. thiol) of NaNO2/500uM DTPA.
3. Collect and wash with cold dtpa and cold methanol. Dry under vacuum for 45 min in dark (covered with foil).
4. Add 1 mg of nitrosated particles to 5 mL PBS (DTPA)
5. Expose to 200 W illumination (30 cm from inside bottom of box)
The results of Experiments 8A-8D are shown in
SEM images of the particles formed in Example 7 A are provided in Figure 10. SEM images of the particles formed in Example 7C are provided in Figure 11. SEM images of the particles formed in Example 7D are shown in Figure 12. These results show that particle morphology and nitric oxide storage is not significantly influenced by the sonication procedure. Thus, sonicating particles may be used to narrow size distribution and/or making smaller particles.
In the drawings and specification, there have been disclosed embodiments of the invention and, although specific terms are employed, they are used in a generic and descriptive sense only and not for purposes of limitation, the scope of the invention being set forth in the following claims.
Claims
1 . A method of forming S-nitrosothiol-functionalized co-condensed silica particles comprising:
reacting a thiol-containing silane and a backbone alkoxysilane in a sol precursor solution that comprises water to form thiol-functionalized co-condensed silica particles, wherein the thiol-functionalized co-condensed silica particles comprise a polysiloxane matrix and at least some of thiol groups are present within the polysiloxane matrix; and
reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent to provide the S-nitrosothiol-functionalized co-condensed silica particles.
2. The method of claim 1 , wherein the thiol-containing silane comprises a primary thiol-containing silane.
3. The method of claim 2, wherein the primary thiol-containing silane comprises mercaptopropyltrimethoxysilane and the backbone alkoxysilane comprises
tetramethoxysilane.
4. The method of claim 3, wherein the sol precursor solution comprises an ammonia catalyst at a concentration in a range of about 1.9 to about 5.5 M;
wherein the total silane monomer concentration in the sol precursor solution is in a range of about 0.1 M to about 0.4 M;
wherein the total silane monomer concentration comprises about 25 to about 85 mol % mercaptopropyltrimethoxysilane; and
wherein the water is present in the sol precursor solution at a concentration in a range of about 8.0 to about 32.5 M.
5. The method of claim 2, wherein the primary thiol-containing silane is mercaptopropyltrimethoxysilane and the backbone alkoxysilane is tetraethoxysilane.
6. The method of claim 5, wherein the sol precursor solution comprises an ammonia catalyst at a concentration in a range of about 1.9 to about 5.5 M;
wherein the total silane monomer concentration in the sol precursor solution is in a range of about 0.1 M to about 0.4 M; wherein the total silane monomer concentration comprises about 75 to about 85 mol % mercaptopropyltrimethoxysilane; and
wherein the water is present in the sol precursor solution a concentration in a range of about 8.0 to about 32.5 M.
7. The method of claim 1, wherein the thiol-containing silane comprises a tertiary thiol-containing silane.
8. The method of claim 7, wherein the tertiary thiol-containing silane comprises a tertiary thiol having the following structure:
wherein R, R' and R" are each independently alkyl and n is in a range of 0 to 10.
9. The method of claim 8, wherein the tertiary thiol-containing silane has the following structure:
10. The method of claim 8, wherein the backbone alkoxysilane comprises tetraethoxysilane.
1 1. The method of claim 10, wherein the sol precursor solution comprises an ammonia catalyst at a concentration in a range of about 1.9 to about 5.5 M; wherein the total silane monomer concentration in the sol precursor solution is in a range of about 0.1 M to about 0.4 M;
wherein the total silane monomer concentration comprises about 25 to about 85 mol % tertiary thiol-containing silane; and
wherein the water is present in the sol precursor solution at a concentration in a range of about 8.0 to about 32.5 M.
12. The method of claim 1 , wherein the thiol-functionalized co-condensed silica particles are sonicated prior to reacting the thiol-functionalized co-condensed silica particles with a nitrosating agent.
13. S-nitrosothiol-functionalized monodisperse co-condensed silica particles having an average particle diameter in a range of about 10 nra to about 100 μπι.
14. The S-nitrosothiol-functionalized monodisperse co-condensed silica particles of claim 13, wherein the particles have an average particle diameter in a range of about 200 nm to about 700 nm.
15. The S-nitrosothiol-functionalized co-condensed silica particles of claim 13, wherein the particles have at least some nitrosothiol functional groups distributed within a polysiloxane matrix of the particles.
16. The S-nitrosothiol-functionalized co-condensed silica particles of claim 13, wherein the S-nitrosothiol functional groups comprise primary nitrosothiol functional groups.
17. The S-nitrosothiol-functionalized co-condensed silica particles of claim 13, wherein the S-nitrosothiol functional groups comprise tertiary nitrosothiol functional groups.
18. S-nitrosothiol-functionalized co-condensed silica particles having an NO storage in a range of about 0.01 μιτιοΐ to about 10 μΐΏθΙ NO per mg particle.
19. The S-nitrosothiol-functionalized co-condensed silica particles of claim 18, wherein the particles have an NO storage in a range of about 0.09 μτηοΐ to about 4.40 μιηοΐ NO per mg particle.
20. The S-nitrosothiol-functionalized co-condensed silica particles of claim 18, wherein the particles have at least some nitrosothiol functional groups distributed within a polysiloxane matrix of the particles.
21. The S-nitrosothiol-functionalized co-condensed silica particles of claim 18, wherein the S-nitrosothiol functional groups comprise primary nitrosothiol functional groups.
22. The S-nitrosothiol-functionalized co-condensed silica particles of claim 18, wherein the S-nitrosothiol functional groups comprise tertiary nitrosothiol functional groups.
23. A S-nitrosothiol-functionalized co-condensed silica particle comprising tertiary nitrosothiol functional groups.
24. A compound having the following structure:
wherein R, R' and R" are each independently alkyl and n is 0 in a range of 0 to 10.
25. The compound of claim 24, wherein the compound has the following structure:
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP12752627.5A EP2681286B1 (en) | 2011-02-28 | 2012-02-28 | Nitric oxide-releasing s-nitrosothiol-modified silica particles and methods of making the same |
| ES12752627T ES2695173T3 (en) | 2011-02-28 | 2012-02-28 | Silica particles modified with S-nitrosothiol that release nitric oxide and methods of manufacturing them |
| US13/975,930 US9713652B2 (en) | 2011-02-28 | 2013-08-26 | Nitric oxide-releasing S-nitrosothiol-modified silica particles and methods of making the same |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201161447368P | 2011-02-28 | 2011-02-28 | |
| US61/447,368 | 2011-02-28 | ||
| US201161565694P | 2011-12-01 | 2011-12-01 | |
| US61/565,694 | 2011-12-01 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/975,930 Continuation US9713652B2 (en) | 2011-02-28 | 2013-08-26 | Nitric oxide-releasing S-nitrosothiol-modified silica particles and methods of making the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2012118819A2 true WO2012118819A2 (en) | 2012-09-07 |
| WO2012118819A3 WO2012118819A3 (en) | 2014-04-17 |
Family
ID=46758457
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2012/026972 WO2012118829A2 (en) | 2011-02-28 | 2012-02-28 | Tertiary s-nitrosothiol-modified nitricoxide-releasing xerogels and methods of using the same |
| PCT/US2012/026960 WO2012118819A2 (en) | 2011-02-28 | 2012-02-28 | Nitric oxide-releasing s-nitrosothiol-modified silica particles and methods of making the same |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2012/026972 WO2012118829A2 (en) | 2011-02-28 | 2012-02-28 | Tertiary s-nitrosothiol-modified nitricoxide-releasing xerogels and methods of using the same |
Country Status (4)
| Country | Link |
|---|---|
| US (2) | US9713652B2 (en) |
| EP (1) | EP2681286B1 (en) |
| ES (1) | ES2695173T3 (en) |
| WO (2) | WO2012118829A2 (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2517443A (en) * | 2013-08-20 | 2015-02-25 | Henkel IP & Holding GmbH | Improved bonding or vulcanisation compositions |
| US8981139B2 (en) | 2011-02-28 | 2015-03-17 | The University Of North Carolina At Chapel Hill | Tertiary S-nitrosothiol-modified nitric—oxide-releasing xerogels and methods of using the same |
| CN107454849A (en) * | 2015-03-27 | 2017-12-08 | 萨卢庞特咨询有限公司 | The sterilizing methods of S nitrosothiolses |
| US10322082B2 (en) | 2014-07-11 | 2019-06-18 | Novan, Inc. | Topical antiviral compositions and methods of using the same |
| US10322081B2 (en) | 2014-07-11 | 2019-06-18 | Novan, Inc. | Topical antiviral compositions and methods of using the same |
| WO2020180705A1 (en) * | 2019-03-01 | 2020-09-10 | The University Of North Carolina At Chapel Hill | Extended nitric oxide-releasing polymers via functionalized mesoporous silica nanoparticles |
| US10849864B2 (en) | 2015-07-28 | 2020-12-01 | Novan, Inc. | Combinations and methods for the treatment and/or prevention of fungal infections |
| US10925689B2 (en) | 2014-07-14 | 2021-02-23 | Novan, Inc. | Nitric oxide releasing nail coating compositions, nitric oxide releasing nail coatings, and methods of using the same |
| US11077194B2 (en) | 2012-03-14 | 2021-08-03 | Novan, Inc. | Nitric oxide releasing pharmaceutical compositions |
| US11285171B2 (en) | 2018-03-01 | 2022-03-29 | Novan, Inc. | Nitric oxide releasing suppositories and methods of use thereof |
| US11534382B2 (en) | 2017-06-19 | 2022-12-27 | Novan, Inc. | Topical compositions and methods of using the same |
Families Citing this family (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PL1871433T3 (en) | 2005-03-24 | 2009-09-30 | Nolabs Ab | Cosmetic treatment with nitric oxide, device for performing said treatment and manufacturing method therefor |
| US9259535B2 (en) | 2006-06-22 | 2016-02-16 | Excelsior Medical Corporation | Antiseptic cap equipped syringe |
| US11229746B2 (en) | 2006-06-22 | 2022-01-25 | Excelsior Medical Corporation | Antiseptic cap |
| CN101801521B (en) | 2007-05-14 | 2015-06-17 | 纽约州立大学研究基金会 | Induction of a physiological dispersion response in bacterial cells in a biofilm |
| US9078992B2 (en) | 2008-10-27 | 2015-07-14 | Pursuit Vascular, Inc. | Medical device for applying antimicrobial to proximal end of catheter |
| EP2467127B1 (en) | 2009-08-21 | 2023-08-02 | Novan, Inc. | Topical gels |
| EP2729131B1 (en) | 2011-07-05 | 2020-04-15 | Novan, Inc. | Topical compositions |
| WO2013006613A1 (en) | 2011-07-05 | 2013-01-10 | Novan, Inc. | Methods of manufacturing topical compositions and apparatus for same |
| CN103796704B (en) | 2011-07-12 | 2016-12-07 | 博讯瓦勒公司 | For the device that antimicrobial is delivered in percutaneous catheter |
| WO2013169538A1 (en) * | 2012-05-08 | 2013-11-14 | Albert Einstein College Of Medicine Of Yeshiva University | Nanoparticle delivery vehicle for s-nitroso-n-acetyl cysteine and uses thereof |
| EP2953660B1 (en) | 2013-02-07 | 2020-04-01 | The Regents Of The University Of Michigan | Thromboresistant/bactericidal s-nitroso-n-acetylpenicillamine (snap)-doped nitric oxide release polymers with enhanced stability |
| US9855211B2 (en) | 2013-02-28 | 2018-01-02 | Novan, Inc. | Topical compositions and methods of using the same |
| CA2913952C (en) | 2013-05-30 | 2021-12-07 | National Research Council Of Canada | Conductors comprising a functionalized organosiloxane network and methods for the preparation thereof |
| US10206947B2 (en) | 2013-08-08 | 2019-02-19 | Novan, Inc. | Topical compositions and methods of using the same |
| KR102321169B1 (en) | 2013-08-08 | 2021-11-02 | 노반, 인크. | Topical compositions and methods of using the same |
| CN103773227B (en) * | 2013-12-23 | 2016-04-06 | 安徽省金盾涂料有限责任公司 | A kind of high-strength wearable coating |
| EP2982386A1 (en) | 2014-08-05 | 2016-02-10 | Christian Schrank | Breast implant comprising a nitric oxide releasing material |
| WO2016182822A1 (en) | 2015-05-08 | 2016-11-17 | Icu Medical, Inc. | Medical connectors configured to receive emitters of therapeutic agents |
| US20160367620A1 (en) | 2015-06-19 | 2016-12-22 | Harry B. Demopoulos | Glutathione |
| TWI638668B (en) * | 2015-09-09 | 2018-10-21 | 國立中興大學 | Surface modification method and surface modification structure for improving blood compatibility of biomedical metal substrate |
| CN108271347B (en) | 2015-09-15 | 2021-04-20 | 萨维奇医疗股份有限公司 | Device and method for anchoring a sheath in a tissue cavity |
| WO2017100629A1 (en) | 2015-12-09 | 2017-06-15 | The Research Foundation For The State University Of New York | Mixed transition metal oxides silica xerogels as antifouling/fouling release surfaces |
| JP7090549B2 (en) | 2016-03-02 | 2022-06-24 | ノヴァン,インコーポレイテッド | Therapeutic compositions for inflammation and their treatment methods |
| KR102426006B1 (en) | 2016-04-13 | 2022-07-29 | 노반, 인크. | Compositions, systems, kits, and methods for treating infections |
| KR101888763B1 (en) * | 2016-05-13 | 2018-09-20 | 광운대학교 산학협력단 | Nanofiber for storage and delivery of nitric oxide |
| JP7005609B2 (en) | 2016-10-14 | 2022-02-10 | アイシーユー・メディカル・インコーポレーテッド | Purification cap for medical connectors |
| WO2018191415A1 (en) | 2017-04-11 | 2018-10-18 | Colorado State University Research Foundation | Functionalization of metal-organic frameworks |
| WO2018204206A2 (en) | 2017-05-01 | 2018-11-08 | Icu Medical, Inc. | Medical fluid connectors and methods for providing additives in medical fluid lines |
| WO2019014348A1 (en) * | 2017-07-11 | 2019-01-17 | Colorado State University Research Foundation | Metal-organic framework-chitosan composite material |
| TW201915519A (en) * | 2017-09-26 | 2019-04-16 | 鴻海精密工業股份有限公司 | Gas permeable ophthalmic lens material and gas permeable ophthalmic lens of such material |
| AU2019254237A1 (en) | 2018-04-16 | 2020-12-03 | Onquality Pharmaceuticals China Ltd. | Method for preventing or treating side effects of cancer therapy |
| US11541105B2 (en) | 2018-06-01 | 2023-01-03 | The Research Foundation For The State University Of New York | Compositions and methods for disrupting biofilm formation and maintenance |
| US20190365797A1 (en) * | 2018-06-05 | 2019-12-05 | Zylo Therapeutics, Inc. | Systems and Methods for Delivery of Nitric Oxide |
| US11400195B2 (en) | 2018-11-07 | 2022-08-02 | Icu Medical, Inc. | Peritoneal dialysis transfer set with antimicrobial properties |
| US11534595B2 (en) | 2018-11-07 | 2022-12-27 | Icu Medical, Inc. | Device for delivering an antimicrobial composition into an infusion device |
| US11541221B2 (en) | 2018-11-07 | 2023-01-03 | Icu Medical, Inc. | Tubing set with antimicrobial properties |
| US11517732B2 (en) | 2018-11-07 | 2022-12-06 | Icu Medical, Inc. | Syringe with antimicrobial properties |
| US11541220B2 (en) | 2018-11-07 | 2023-01-03 | Icu Medical, Inc. | Needleless connector with antimicrobial properties |
| AU2019384564B2 (en) | 2018-11-21 | 2023-11-23 | Icu Medical, Inc. | Antimicrobial device comprising a cap with ring and insert |
| CN114206948B (en) * | 2019-06-10 | 2023-11-03 | 密歇根大学董事会 | catheter insertion device |
| WO2021188651A1 (en) | 2020-03-19 | 2021-09-23 | Friedman Joel M | Novel approach to sustained production and delivery of nitric oxide and s-nitrosothiols |
| CN112341896A (en) * | 2020-08-28 | 2021-02-09 | 四川仁智新材料科技有限责任公司 | Performance regulating material for pipeline preparation and preparation method thereof |
| AU2021373070B2 (en) | 2020-11-09 | 2023-08-24 | Albert Einstein College Of Medicine | Transdermal delivery formulations |
| JP2024500319A (en) | 2020-12-07 | 2024-01-09 | アイシーユー・メディカル・インコーポレーテッド | Peritoneal dialysis cap, system, and method |
| US11786712B2 (en) | 2021-08-30 | 2023-10-17 | Albert Einstein College Of Medicine | Nitric oxide-releasing device |
| WO2023205125A1 (en) * | 2022-04-20 | 2023-10-26 | Sterile State, Llc | Functionalized polymeric material for forming nitric oxide and methods therefor |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090214618A1 (en) | 2005-05-27 | 2009-08-27 | Schoenfisch Mark H | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
Family Cites Families (401)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4558120A (en) | 1983-01-07 | 1985-12-10 | The Dow Chemical Company | Dense star polymer |
| US4631337A (en) | 1983-01-07 | 1986-12-23 | The Dow Chemical Company | Hydrolytically-stable dense star polyamine |
| US4568737A (en) | 1983-01-07 | 1986-02-04 | The Dow Chemical Company | Dense star polymers and dendrimers |
| US4507466A (en) | 1983-01-07 | 1985-03-26 | The Dow Chemical Corporation | Dense star polymers having core, core branches, terminal groups |
| US4737550A (en) | 1983-01-07 | 1988-04-12 | The Dow Chemical Company | Bridged dense star polymers |
| US4587329A (en) | 1984-08-17 | 1986-05-06 | The Dow Chemical Company | Dense star polymers having two dimensional molecular diameter |
| US4871779A (en) | 1985-12-23 | 1989-10-03 | The Dow Chemical Company | Ion exchange/chelation resins containing dense star polymers having ion exchange or chelate capabilities |
| US4694064A (en) | 1986-02-28 | 1987-09-15 | The Dow Chemical Company | Rod-shaped dendrimer |
| US4713975A (en) | 1986-05-30 | 1987-12-22 | The Dow Chemical Company | Dense star polymers for calibrating/characterizing sub-micron apertures |
| US4857599A (en) | 1988-02-08 | 1989-08-15 | The Dow Chemical Company | Modified dense star polymers |
| US5061487A (en) | 1988-05-09 | 1991-10-29 | Dow Corning Corporation | Antimicrobial superabsorbent compositions and methods |
| US5035892A (en) | 1988-05-09 | 1991-07-30 | Dow Corning Corporation | Antimicrobial superabsorbent compositions and methods |
| US4990338A (en) | 1988-05-09 | 1991-02-05 | Dow Corning Corporation | Antimicrobial superabsorbent compositions and methods |
| US4985023A (en) | 1988-05-09 | 1991-01-15 | Dow Corning Corporation | Antimicrobial superabsorbent articles |
| US5045322A (en) | 1988-05-09 | 1991-09-03 | Dow Corning Corporation | Antimicrobial superabsorbent sanitary napkin |
| US5079004A (en) | 1990-08-06 | 1992-01-07 | Dow Corning Corporation | Antimicrobial superabsorbent compositions and method |
| US5380758A (en) | 1991-03-29 | 1995-01-10 | Brigham And Women's Hospital | S-nitrosothiols as smooth muscle relaxants and therapeutic uses thereof |
| DE69231666T2 (en) | 1991-11-14 | 2001-05-10 | Brigham & Womens Hospital | NITROSYLATION OF ENZYM-SH GROUPS AS A THERAPEUTIC MEASURE |
| US6291424B1 (en) | 1991-11-14 | 2001-09-18 | Brigham And Women's Hospital | Nitrosated and nitrosylated heme proteins |
| SE9200564L (en) | 1992-02-26 | 1993-03-15 | Perstorp Ab | DENDRITIC MACROMOLECYLE OF POLYESTER TYPE, PROCEDURES FOR PRODUCING THEREOF AND USING THEREOF |
| US5814666A (en) | 1992-04-13 | 1998-09-29 | The United States As Represented By The Department Of Health And Human Services | Encapsulated and non-encapsulated nitric oxide generators used as antimicrobial agents |
| US5910316A (en) | 1992-08-24 | 1999-06-08 | The United States Of America, As Represented By The Department Of Health And Human Services | Use of nitric oxide-releasing agents to treat impotency |
| US6200558B1 (en) | 1993-09-14 | 2001-03-13 | The United States Of America As Represented By The Department Of Health And Human Services | Biopolymer-bound nitric oxide-releasing compositions, pharmaceutical compositions incorporating same and methods of treating biological disorders using same |
| US5632981A (en) | 1992-08-24 | 1997-05-27 | The United States Of America As Represented By The Department Of Health And Human Services | Biopolymer-bound nitric oxide-releasing compositions, pharmaceutical compositions incorporating same and methods of treating biological disorders using same |
| US5650447A (en) | 1992-08-24 | 1997-07-22 | The United States Of America As Represented By The Secretary Of The Department Of Health And Human Services | Nitric oxide-releasing polymers to treat restenosis and related disorders |
| US5405919A (en) | 1992-08-24 | 1995-04-11 | The United States Of America As Represented By The Secretary Of Health And Human Services | Polymer-bound nitric oxide/nucleophile adduct compositions, pharmaceutical compositions and methods of treating biological disorders |
| US5525357A (en) | 1992-08-24 | 1996-06-11 | The United States Of America As Represented By The Department Of Health And Human Services | Polymer-bound nitric oxide/nucleophile adduct compositions, pharmaceutical compositions incorporating same and methods of treating biological disorders using same |
| US5691423A (en) | 1992-08-24 | 1997-11-25 | The United States Of America As Represented By The Department Of Health And Human Services | Polysaccharide-bound nitric oxide-nucleophile adducts |
| US5891459A (en) | 1993-06-11 | 1999-04-06 | The Board Of Trustees Of The Leland Stanford Junior University | Enhancement of vascular function by modulation of endogenous nitric oxide production or activity |
| US5852058A (en) | 1993-06-11 | 1998-12-22 | The Board Of Trustees Of The Leland Stanford Junior University | Intramural delivery of nitric oxide enhancer for inhibiting lesion formation after vascular injury |
| US5428070A (en) | 1993-06-11 | 1995-06-27 | The Board Of Trustees Of The Leland Stanford Junior University | Treatment of vascular degenerative diseases by modulation of endogenous nitric oxide production of activity |
| US5861168A (en) | 1993-06-11 | 1999-01-19 | The Board Of Trustees Of The Leland Stanford Junior University | Intramural delivery of nitric oxide enhancer for inhibiting lesion formation after vascular injury |
| US6087479A (en) | 1993-09-17 | 2000-07-11 | Nitromed, Inc. | Localized use of nitric oxide-adducts to prevent internal tissue damage |
| US6255277B1 (en) | 1993-09-17 | 2001-07-03 | Brigham And Women's Hospital | Localized use of nitric oxide-adducts to prevent internal tissue damage |
| DE69433478T2 (en) | 1993-09-17 | 2004-11-25 | Brigham And Women's Hospital, Boston | USE OF STAINOXIDE ADDUCTS TO PREVENT THROMBOSES ON ARTIFICIAL AND VASCULAR SURFACES |
| US5840759A (en) | 1993-10-08 | 1998-11-24 | The United States Of America As Represented By The Department Of Health And Human Services | Use of nitric oxide releasing compounds to protect noncancerous cells from chemotherapeutic agents |
| WO1995010267A1 (en) | 1993-10-08 | 1995-04-20 | The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Use of nitric oxide-releasing compounds as hypoxic cell radiation sensitizers |
| CA2175467A1 (en) | 1993-11-02 | 1995-05-11 | David A. Wink, Jr. | Use of nitric oxide releasing compounds as protective agents in ischemia reperfusion injury |
| US5786332A (en) | 1995-03-06 | 1998-07-28 | Trega Biosciences, Inc. | Cytokine restraining agents and methods of use in pathologies and conditions associated with altered cytokine levels |
| US5726156A (en) | 1995-03-06 | 1998-03-10 | Trega Biosciences, Inc. | Cytokine regulatory agents and methods of use in pathologies and conditions associated with altered cytokine levels |
| US5599984A (en) | 1994-01-21 | 1997-02-04 | The Picower Institute For Medical Research | Guanylhydrazones and their use to treat inflammatory conditions |
| US6008255A (en) | 1994-01-21 | 1999-12-28 | The Picower Institute For Medical Research | Guanylhydrazones and their use to treat inflammatory conditions |
| US6709681B2 (en) | 1995-02-17 | 2004-03-23 | Aberdeen University | Acidified nitrite as an antimicrobial agent |
| EP0746327B1 (en) | 1994-02-21 | 2004-04-21 | Aberdeen University | Acidified nitrite as an antimicrobial agent |
| US5935924A (en) | 1994-04-15 | 1999-08-10 | Genentech, Inc. | Treatment of congestive heart failure |
| US5504117A (en) | 1994-05-27 | 1996-04-02 | Neptune Pharmaceutical Corporation | Pharmacologic preparation for the treatment of anal disorders |
| PT719145E (en) | 1994-05-27 | 2001-01-31 | Cellegy Pharma Inc | NITRIC OXIDE DERIVATIVE COMPOSITION FOR TREATMENT OF ANAL DISORDERS |
| US6190704B1 (en) | 1994-09-23 | 2001-02-20 | New York Society For The Ruptured And Crippled Maintaining The Hospital For Special Surgery | Regulation of wound healing by nitric oxide |
| US6747062B2 (en) | 1994-09-26 | 2004-06-08 | New York Society For The Ruptured And Crippled Maintaining The Hospital For Special Surgery | Regulation of wound healing by nitric oxide |
| US5519020A (en) | 1994-10-28 | 1996-05-21 | The University Of Akron | Polymeric wound healing accelerators |
| US5629322A (en) | 1994-11-15 | 1997-05-13 | Merck & Co., Inc. | Cyclic amidine analogs as inhibitors of nitric oxide synthase |
| US5700830A (en) | 1994-11-22 | 1997-12-23 | The United States Of America As Represented By The Department Of Health And Human Services | Use of nitric oxide-releasing agents for reducing metastasis risk |
| US5961466A (en) | 1995-01-03 | 1999-10-05 | Omnicorder Technologies, Inc. | Method of detection of cancerous lesions by their effect on the spatial distribution of modulation of temperature and homogeneity of tissue |
| US5999843A (en) | 1995-01-03 | 1999-12-07 | Omnicorder Technologies, Inc. | Detection of cancerous lesions by their effect on the spatial homogeneity of skin temperature |
| US6035225A (en) | 1995-01-03 | 2000-03-07 | Omnicorder Technologies | Detection of cancerous lesions by measuring nitric oxide concentrations in tissue |
| US5810010A (en) | 1995-01-03 | 1998-09-22 | Anbar; Michael | Detection of cancerous lesions by their effect on the periodic modulation of perfusion in the surrounding tissues |
| JPH10511957A (en) | 1995-01-05 | 1998-11-17 | ザ ボード オブ リージェンツ オブ ザ ユニヴァーシティ オブ ミシガン | Surface-modified nanoparticles and methods for their production and use |
| US5665077A (en) | 1995-04-24 | 1997-09-09 | Nitrosci Pharmaceuticals Llc | Nitric oxide-releasing nitroso compositions and methods and intravascular devices for using them to prevent restenosis |
| US6043358A (en) | 1995-11-01 | 2000-03-28 | Merck & Co., Inc. | Hexahydro-5-imino-1,4-heteroazepine derivatives as inhibitors of nitric oxide synthases |
| DE69637676D1 (en) | 1995-11-09 | 2008-10-23 | Univ R | Use of locally administered lysines to IMPROVE VASCULAR FUNCTION |
| US5821261A (en) | 1995-12-08 | 1998-10-13 | Merck & Co., Inc. | Substituted saturated aza heterocycles as inhibitors of nitric oxide synthase |
| US20020061879A1 (en) | 1996-02-02 | 2002-05-23 | Garvey David S. | Nitrosated and nitrosylated alpha-adrenergic receptor antagonist compounds, compositions and their uses |
| US20050065161A1 (en) | 1996-02-02 | 2005-03-24 | Nitromed, Inc. | Nitrosated and nitrosylated alpha-adrenergic receptor antagonist compounds, compositions and their uses |
| US6323211B1 (en) | 1996-02-02 | 2001-11-27 | Nitromed, Inc. | Compositions and methods for treating sexual dysfunctions |
| US5994294A (en) | 1996-02-02 | 1999-11-30 | Nitromed, Inc. | Nitrosated and nitrosylated α-adrenergic receptor antagonist compounds, compositions and their uses |
| US20020143007A1 (en) | 1996-02-02 | 2002-10-03 | Garvey David S. | Nitrosated and nitrosylated alpha-adrenergic receptor antagonists, compositions and methods of use |
| US6294517B1 (en) | 1996-02-02 | 2001-09-25 | Nitromed, Inc. | Compositions and kits comprising alpha-adrenergic receptor antagonists and nitric oxide donors and methods of use |
| US5932538A (en) | 1996-02-02 | 1999-08-03 | Nitromed, Inc. | Nitrosated and nitrosylated α-adrenergic receptor antagonist compounds, compositions and their uses |
| US6469065B1 (en) | 1996-02-02 | 2002-10-22 | Nitromed, Inc. | Nitrosated and nitrosylated α-adrenergic receptor antagonist, compositions and methods of use |
| IT1282686B1 (en) | 1996-02-26 | 1998-03-31 | Nicox Sa | COMPOUNDS ABLE TO REDUCE DRUG TOXICITY |
| AU2990497A (en) | 1996-03-15 | 1997-10-01 | Yale University | Methods and means of detecting nitric oxide synthase |
| US6143037A (en) | 1996-06-12 | 2000-11-07 | The Regents Of The University Of Michigan | Compositions and methods for coating medical devices |
| US5770645A (en) | 1996-08-02 | 1998-06-23 | Duke University Medical Center | Polymers for delivering nitric oxide in vivo |
| US6232434B1 (en) | 1996-08-02 | 2001-05-15 | Duke University Medical Center | Polymers for delivering nitric oxide in vivo |
| AU3986097A (en) | 1996-08-27 | 1998-03-19 | University Of Akron, The | Lipophilic polyamine esters for the site specific delivery of nitric oxide in pharmaceutical use |
| US5797887A (en) | 1996-08-27 | 1998-08-25 | Novovasc Llc | Medical device with a surface adapted for exposure to a blood stream which is coated with a polymer containing a nitrosyl-containing organo-metallic compound which releases nitric oxide from the coating to mediate platelet aggregation |
| US20030093143A1 (en) | 1999-03-01 | 2003-05-15 | Yiju Zhao | Medical device having surface depressions containing nitric oxide releasing compound |
| DE69731758T2 (en) | 1996-09-27 | 2005-12-15 | The United States Government As Represented By The Department Of Health And Human Services | O2-ARYLATE OR O2 GLYCOSYIATE 1-SUBSTITUTED DIAZEN-1-IUM-1,2-DIOLATE AND O2-SUBSTITUTED 1 (2-CARBOXYLATE) PYROLIDIN-1-YL DIAZEN-1-IUM-1,2-DIOLATE |
| GB9622997D0 (en) | 1996-11-05 | 1997-01-08 | Univ St Andrews | Nitric oxide donor drugs |
| US5958427A (en) | 1996-11-08 | 1999-09-28 | Salzman; Andrew L. | Nitric oxide donor compounds and pharmaceutical compositions for pulmonary hypertension and other indications |
| US6372796B1 (en) | 1996-11-13 | 2002-04-16 | Cold Spring Harbor Laboratory | Therapeutic uses for nitric oxide inhibitors |
| US5891472A (en) | 1996-11-19 | 1999-04-06 | Meri Charmyne Russell | Treatment of equine laminitis |
| US6896899B2 (en) | 1996-12-31 | 2005-05-24 | Antioxidant Pharmaceuticals Corp. | Pharmaceutical preparations of glutathione and methods of administration thereof |
| CA2276183C (en) | 1996-12-31 | 2009-06-09 | Antioxidant Pharmaceuticals Corporation | Pharmaceutical preparations of glutathione and methods of administration thereof |
| US6070928A (en) | 1997-01-17 | 2000-06-06 | Cascade Engineering, Inc. | Mat with integrally molded door |
| EP1001677A4 (en) | 1997-02-04 | 2003-01-08 | Gen Hospital Corp | A novel method for treating epidermal or dermal conditions |
| US6034752A (en) | 1997-03-22 | 2000-03-07 | Kent Displays Incorporated | Display device reflecting visible and infrared radiation |
| IL120531A (en) | 1997-03-26 | 2006-12-31 | Yissum Res Dev Co | Nitric oxide donors and pharmaceutical compositions containing them |
| US6171232B1 (en) | 1997-06-26 | 2001-01-09 | Cordis Corporation | Method for targeting in vivo nitric oxide release |
| ATE499343T1 (en) | 1997-07-03 | 2011-03-15 | Us Gov Health & Human Serv | NITROGEN OXIDE RELEASE AMIDINE AND ENAMINE RELATED DIAZENIUM DIOLATES, PREPARATIONS AND USES THEREOF AND METHOD FOR THE PRODUCTION THEREOF |
| US6796966B2 (en) | 1997-10-15 | 2004-09-28 | Jeffrey E. Thomas | Apparatus, and kits for preventing of alleviating vasoconstriction or vasospasm in a mammal |
| JP2001519386A (en) | 1997-10-15 | 2001-10-23 | トーマス・ジェファーソン・ユニバーシティ | Nitric oxide donor compositions, methods, devices and kits for preventing or reducing vasoconstriction or vasospasm in mammals |
| US5994444A (en) | 1997-10-16 | 1999-11-30 | Medtronic, Inc. | Polymeric material that releases nitric oxide |
| CA2219867A1 (en) | 1997-10-31 | 1999-04-30 | Jiangping Wu | The use of proteasome inhibitors for treating cancer, inflammation, autoimmune disease, graft rejection and septic shock |
| US6180082B1 (en) | 1997-11-24 | 2001-01-30 | Board Of Supervisors Of Louisiana State University And Agricultural And Mechanical College | Method to enhance tissue accumulation of radiolabeled compounds |
| EP1060174B1 (en) | 1997-12-23 | 2004-09-22 | Amersham Health AS | Nitric oxide releasing chelating agents and their therapeutic use |
| GB9801398D0 (en) | 1998-01-22 | 1998-03-18 | Anggard Erik E | Chemical compounds |
| IT1298338B1 (en) | 1998-03-05 | 1999-12-20 | Nicox Sa | PHARMACEUTICAL COMPOSITIONS FOR THE ULCER |
| US6560478B1 (en) | 1998-03-16 | 2003-05-06 | The Research Foundation Of City University Of New York | Method and system for examining biological materials using low power CW excitation Raman spectroscopy |
| US6151522A (en) | 1998-03-16 | 2000-11-21 | The Research Foundation Of Cuny | Method and system for examining biological materials using low power CW excitation raman spectroscopy |
| US5962520A (en) | 1998-04-02 | 1999-10-05 | The University Of Akron | Hydrolytically unstable, biocompatible polymer |
| US6103275A (en) | 1998-06-10 | 2000-08-15 | Nitric Oxide Solutions | Systems and methods for topical treatment with nitric oxide |
| US6207855B1 (en) | 1998-06-23 | 2001-03-27 | Duke University Medical Center | Stable no-delivering compounds |
| US20020068365A1 (en) | 1998-07-28 | 2002-06-06 | Eric H. Kuhrts | Controlled release nitric oxide producing agents |
| US20040043068A1 (en) | 1998-09-29 | 2004-03-04 | Eugene Tedeschi | Uses for medical devices having a lubricious, nitric oxide-releasing coating |
| US6379691B1 (en) | 1998-09-29 | 2002-04-30 | Medtronic/Ave, Inc. | Uses for medical devices having a lubricious, nitric oxide-releasing coating |
| US6299980B1 (en) | 1998-09-29 | 2001-10-09 | Medtronic Ave, Inc. | One step lubricious coating |
| US20070086954A1 (en) | 1998-11-23 | 2007-04-19 | Miller Christopher C | Method and apparatus for treatment of respiratory infections by nitric oxide inhalation |
| CA2254645A1 (en) | 1998-11-23 | 2000-05-23 | Pulmonox Medical Corporation | Method and apparatus for treatment of respiratory infections by nitric oxide inhalation |
| US20070275100A1 (en) | 1998-11-23 | 2007-11-29 | Miller Christopher C | Use of gaseous nitric oxide as an anti-cancer agent |
| US6261594B1 (en) | 1998-11-25 | 2001-07-17 | The University Of Akron | Chitosan-based nitric oxide donor compositions |
| US7179475B1 (en) | 1998-12-04 | 2007-02-20 | Johnson & Johnson Consumer Companies, Inc. | Anhydrous topical skin preparations |
| US6238683B1 (en) | 1998-12-04 | 2001-05-29 | Johnson & Johnson Consumer Companies, Inc. | Anhydrous topical skin preparations |
| GB9905425D0 (en) | 1999-03-09 | 1999-05-05 | Queen Mary & Westfield College | Pharmaceutical composition |
| US20050142218A1 (en) | 2000-03-09 | 2005-06-30 | Queen Mary & Westfield College | Pharmaceutical composition containing nitrate source and an acidifying agent for treating skin ischaemia |
| US6312663B1 (en) | 1999-03-19 | 2001-11-06 | Joseph V. Boykin, Jr. | Prediction of diabetes impaired wound healing by urinary nitrate assay |
| US6562785B1 (en) | 1999-03-23 | 2003-05-13 | Howard M. Shapiro | Method for overcoming bacterial antibiotic resistance |
| WO2000063462A1 (en) | 1999-04-14 | 2000-10-26 | University Of Cincinnati | Silane coatings for adhesion promotion |
| US7655423B2 (en) | 1999-06-14 | 2010-02-02 | Henry Ford Health System | Nitric oxide donors for inducing neurogenesis |
| US7135498B1 (en) | 1999-06-14 | 2006-11-14 | Henry Ford Health System | Nitric oxide donors for inducing neurogenesis |
| EP1233670B1 (en) | 1999-06-14 | 2008-09-17 | Henry Ford Health System | Nitric oxide donors for inducing neurogenesis |
| US20010012851A1 (en) | 1999-07-29 | 2001-08-09 | Kristin M. Lundy | Nitric oxide releasing oxindole prodrugs for anagesic, anti-inflammatory and disease-modifying use |
| US20020049157A1 (en) | 1999-08-25 | 2002-04-25 | Jiangping Wu | Use of proteasome inhibitors for treating cancer, inflammation, autoimmune disease, graft rejection and septic shock |
| IL143305A0 (en) | 1999-09-02 | 2002-04-21 | Rice University | Nitric oxide-producing hydrogel materials |
| US7052711B2 (en) | 1999-09-02 | 2006-05-30 | Rice University | Nitric oxide-producing hydrogel materials |
| US7279176B1 (en) | 1999-09-02 | 2007-10-09 | Rice University | Nitric oxide-producing hydrogel materials |
| US6737447B1 (en) | 1999-10-08 | 2004-05-18 | The University Of Akron | Nitric oxide-modified linear poly(ethylenimine) fibers and uses thereof |
| IL150361A0 (en) | 1999-12-20 | 2002-12-01 | Majeste La Reine Du Cheff Du C | Method and composition for treatment and/or prevention of antibiotic-resistant microorganism infections |
| US6492405B2 (en) | 1999-12-30 | 2002-12-10 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Nitric oxide donors and pharmaceutical compositions containing them |
| US20020132234A1 (en) | 2000-01-24 | 2002-09-19 | Moskowitz David W. | Nitric oxide synthase gene diagnostic polymorphisms |
| US6758214B2 (en) | 2000-01-28 | 2004-07-06 | Cyterra Corporation | Simple nitric oxide generator for ambulatory and/or bedside inhaled no treatment |
| US6699846B2 (en) | 2000-03-17 | 2004-03-02 | Corixa Corporation | Mono- and disaccharides for the treatment of nitric oxide related disorders |
| MXPA02009236A (en) | 2000-03-20 | 2004-04-05 | Novovascular Inc | Matrices containing nitric oxide donors and reducing agents and their use. |
| US20010038832A1 (en) | 2000-04-11 | 2001-11-08 | Benjamin Bonavida | Nitric oxide and analogues thereof effectuate sensitization of neoplasm and immunologically undesired tissues to cytotoxicity |
| US7678391B2 (en) | 2000-04-26 | 2010-03-16 | Queen's University At Kingston | Formulations and methods of using nitric oxide mimetics against a malignant cell phenotype |
| US20050142217A1 (en) | 2000-04-26 | 2005-06-30 | Adams Michael A. | Formulations and methods of using nitric oxide mimetics against a malignant cell phenotype |
| JP2003531179A (en) | 2000-04-26 | 2003-10-21 | クイーンズ ユニバーシティ アット キングストン | Formulations and methods of using nitric oxide mimetics for malignant cell phenotype |
| US20010053772A1 (en) | 2000-04-28 | 2001-12-20 | Benjamin Bonavida | Aza analogues of alkyl lysophospholipids exert immunomodulatory effects |
| US6270779B1 (en) | 2000-05-10 | 2001-08-07 | United States Of America | Nitric oxide-releasing metallic medical devices |
| AU2001264729A1 (en) | 2000-05-22 | 2001-12-03 | Nitromed, Inc. | Thromboxane inhibitors, compositions and methods of use related applications |
| US6538033B2 (en) | 2000-08-29 | 2003-03-25 | Huntington Medical Research Institutes | Nitric oxide donor compounds |
| GB0021317D0 (en) | 2000-08-30 | 2000-10-18 | Queen Mary & Westfield College | Transdermal pharmaceutical delivery composition |
| JP4841066B2 (en) | 2000-09-01 | 2011-12-21 | ライスユニバーシティ | Nitric oxide-forming hydrogel materials |
| GB0022084D0 (en) | 2000-09-08 | 2000-10-25 | Univ Aberdeen | Treatment of multiply antibiotic-resistant organisms |
| US6410622B1 (en) | 2000-09-11 | 2002-06-25 | Gregory W. Endres | Method of preventing fouling organisms in marine environments and polymer-bound nitric oxide/nitric oxide-releasing compositions usable therefor |
| EP1333823B1 (en) | 2000-10-16 | 2010-03-31 | Duke University | Therapeutic use of aerosolized s-nitrosoglutathione in cystic fibrosis |
| CA2425196A1 (en) | 2000-10-17 | 2002-04-25 | Board Of Regents, The University Of Texas System | A method to incorporate n-(4-hydroxyphenyl) retinamide in liposomes |
| US6359182B1 (en) | 2000-10-26 | 2002-03-19 | Duke University | C-nitroso compounds and use thereof |
| US7049308B2 (en) | 2000-10-26 | 2006-05-23 | Duke University | C-nitroso compounds and use thereof |
| KR100383149B1 (en) | 2000-12-12 | 2003-05-12 | 한국생명공학연구원 | A novel use of diterpene compounds as therapeutic agents of inflammation, immune disease and cancer |
| DE10063937A1 (en) | 2000-12-20 | 2002-07-18 | Bayer Ag | Process for the preparation of trimethylol compounds and formic acid |
| US6780849B2 (en) | 2000-12-21 | 2004-08-24 | Scimed Life Systems, Inc. | Lipid-based nitric oxide donors |
| US7329412B2 (en) | 2000-12-22 | 2008-02-12 | The Trustees Of Columbia University In The City Of New York | Antimicrobial medical devices containing chlorhexidine free base and salt |
| US20040009238A1 (en) | 2002-07-09 | 2004-01-15 | Chris Miller | Exogenenous nitric oxide gas (gNO) therapy in wound healing |
| US7122018B2 (en) | 2000-12-26 | 2006-10-17 | Sensormedics Corporation | Device and method for treatment of wounds with nitric oxide |
| US6432077B1 (en) | 2000-12-26 | 2002-08-13 | Sensormedics Corporation | Device and method for treatment of surface infections with nitric oxide |
| US7335383B2 (en) | 2001-01-16 | 2008-02-26 | The Regents Of The University Of Michigan | Generation of nitric oxide in vivo from nitrite, nitrate or nitrosothiols endogenous in blood |
| EP1357936B1 (en) | 2001-01-16 | 2008-10-01 | The Regents of The University of Michigan | Biocatalytic and biomimetic generation of nitric oxide in situ at substrate/blood interfaces |
| US7128904B2 (en) | 2001-01-16 | 2006-10-31 | The Regents Of The University Of Michigan | Material containing metal ion ligand complex producing nitric oxide in contact with blood |
| US6706274B2 (en) | 2001-01-18 | 2004-03-16 | Scimed Life Systems, Inc. | Differential delivery of nitric oxide |
| ITMI20010426A1 (en) | 2001-03-01 | 2002-09-01 | Consiglio Nazionale Ricerche | NITROGEN OXIDE DONORS BASED ON METAL CENTERS |
| US7012098B2 (en) | 2001-03-23 | 2006-03-14 | Pharmacia Corporation | Inhibitors of inducible nitric oxide synthase for chemoprevention and treatment of cancers |
| US20020138051A1 (en) | 2001-03-26 | 2002-09-26 | Hole Douglas R. | System and method for the prevention and treatment of animal wound infections using nitric oxide |
| US7122027B2 (en) | 2001-05-25 | 2006-10-17 | Medtronic, Inc. | Implantable medical device with controllable gaseous agent release system |
| ES2180446B1 (en) | 2001-07-02 | 2004-01-16 | Esteve Labor Dr | EMPLOYMENT OF 2,5-DIHYDROXIBENCENOSULPHONIC ACID DERIVATIVES IN THE PREPARATION OF A MEDICINAL PRODUCT TO POWER THE EFFECT OF OTHER PHARMACOS IN THE TREATMENT OF ERECTILE DYSFUNCTION. |
| US7122529B2 (en) | 2001-07-12 | 2006-10-17 | The Johns Hopkins University | Compounds that release nitric oxide at controlled rates upon photolysis |
| GB0119011D0 (en) | 2001-08-03 | 2001-09-26 | Univ Aberdeen | Treatment of nail infections |
| CA2459647A1 (en) | 2001-08-20 | 2003-02-27 | University Of Virginia Patent Foundation | Use of s-nitrosothiol signaling to treat disordered control of breathing |
| US6841166B1 (en) | 2001-08-21 | 2005-01-11 | The Regents Of The University Of Michigan | Nitric oxide-releasing polymers incorporating diazeniumdiolated silane derivatives |
| US7135189B2 (en) | 2001-08-23 | 2006-11-14 | Boston Scientific Scimed, Inc. | Compositions and techniques for localized therapy |
| CA2459891C (en) | 2001-09-05 | 2014-02-04 | Cyterra Corporation | Method and apparatus for nitric oxide generation |
| US6673338B1 (en) | 2001-09-10 | 2004-01-06 | The United States Of America As Represented By The Department Of Health And Human Services | Nitric oxide-releasing imidate and thioimidate diazeniumdiolates, compositions, uses thereof and method of making same |
| WO2003026717A1 (en) | 2001-09-26 | 2003-04-03 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Nitric oxide-releasing coated medical devices and method of preparing same |
| US6703046B2 (en) | 2001-10-04 | 2004-03-09 | Medtronic Ave Inc. | Highly cross-linked, extremely hydrophobic nitric oxide-releasing polymers and methods for their manufacture and use |
| EP1300083A1 (en) | 2001-10-04 | 2003-04-09 | Societe Des Produits Nestle S.A. | Milk-based snack |
| GB0125222D0 (en) | 2001-10-19 | 2001-12-12 | Barts & London Nhs Trust | Composition for the treatment of microbial infections |
| US6627602B2 (en) | 2001-11-13 | 2003-09-30 | Duke University | Preventing desensitization of receptors |
| US6472390B1 (en) | 2001-11-13 | 2002-10-29 | Duke University | Use of therapeutic dosages for nitric oxide donors which do not significantly lower blood pressure or pulmonary artery pressure |
| US20040110691A1 (en) | 2001-11-13 | 2004-06-10 | Stamler Jonathan S. | Thiol reactive agents as a therapeutic modality |
| CN1612804A (en) | 2001-12-03 | 2005-05-04 | C·R·巴德公司 | Microbe-resistant medical device, microbe-resistant polymeric coating and methods for producing same |
| WO2003059339A1 (en) | 2002-01-15 | 2003-07-24 | Trustees Of Dartmouth College | Tricyclic-bis-enone derivatives and methods of use thereof |
| US8404665B2 (en) | 2002-03-21 | 2013-03-26 | University Of Utah Research Foundation | In vivo use of glutathione S-transferase activated nitric oxide donors |
| CA2480832A1 (en) | 2002-04-05 | 2003-10-23 | Nitromed, Inc. | Nitric oxide donors, compositions and methods of use |
| EP1501563A1 (en) | 2002-05-03 | 2005-02-02 | Duke University Medical Center | Carbon nanotubules for storage of nitric oxide |
| US7328706B2 (en) | 2002-05-06 | 2008-02-12 | Dynamic Mouth Devices Llc | Therapeutic and protective dental device useful as an intra-oral delivery system |
| EP1507526A2 (en) | 2002-05-07 | 2005-02-23 | THE GOVERNMENT OF THE UNITED STATES OF AMERICA, as represented by THE SECRETARY, DEPARTMENT OF HEALTH AND HUMAN SERVICES | Polydiazeniumdiolated cyclic polyamines with polyphasic nitric oxide release and related compounds, compositions comprising same and methods of using same |
| JP2006515859A (en) | 2002-05-13 | 2006-06-08 | トラスティーズ オブ ダートマス カレッジ | Inhibitors and uses thereof |
| US7070798B1 (en) | 2002-06-21 | 2006-07-04 | Advanced Cardiovascular Systems, Inc. | Coatings for implantable medical devices incorporating chemically-bound polymers and oligomers of L-arginine |
| PL375161A1 (en) | 2002-06-21 | 2005-11-28 | University Of Pittsburgh Of The Commonwealth System Of Higher Education | Pharmaceutical use of nitric oxide, heme oxygenase-1 and products of heme degradation |
| US6949530B2 (en) | 2002-07-18 | 2005-09-27 | The United States Of America As Represented By The Department Of Health And Human Services | Nitric oxide-releasing amidine diazeniumdiolates, compositions and uses thereof and method of making same |
| WO2004009253A1 (en) | 2002-07-19 | 2004-01-29 | University Of North Carolina At Chapel Hill | Surface-localized release of an anti-biofouling agent via micropatterning |
| EP1527045A1 (en) | 2002-07-26 | 2005-05-04 | Merck Frosst Canada & Co. | Nitric oxide releasing prodrugs of diaryl-2-(5h)-furanones as cyclooxygenase-2 inhibitors |
| JP2005534485A (en) | 2002-08-02 | 2005-11-17 | アメリカ合衆国 | Cross-linked nitric oxide-releasing polyamine-coated substrate, composition containing it and method for its production |
| US20040076582A1 (en) | 2002-08-30 | 2004-04-22 | Dimatteo Kristian | Agent delivery particle |
| US20040033480A1 (en) | 2002-08-15 | 2004-02-19 | Wong Norman C.W. | Use of resveratrol to regulate expression of apolipoprotein A1 |
| US20050080024A1 (en) | 2002-08-15 | 2005-04-14 | Joseph Tucker | Nitric oxide donating derivatives for the treatment of cardiovascular disorders |
| US20050080021A1 (en) | 2002-08-15 | 2005-04-14 | Joseph Tucker | Nitric oxide donating derivatives of stilbenes, polyphenols and flavonoids for the treatment of cardiovascular disorders |
| US6951902B2 (en) | 2002-08-16 | 2005-10-04 | Michigan Biotechnology Institute | Two dimensional polymer that generates nitric oxide |
| US7674482B2 (en) | 2002-08-27 | 2010-03-09 | Targeted Medical Pharma Inc. | Method and compositions for potentiating pharmaceuticals with amino acid based medical foods |
| US20030039697A1 (en) | 2002-09-12 | 2003-02-27 | Yi-Ju Zhao | Matrices containing nitric oxide donors and reducing agents and their use |
| CA2503063A1 (en) | 2002-10-22 | 2004-05-06 | Merck Frosst Canada & Co./Merck Frosst Canada & Cie | Nitric oxide releasing selective cyclooxygenase-2 inhibitors |
| US20080317679A1 (en) | 2002-10-25 | 2008-12-25 | Foamix Ltd. | Foamable compositions and kits comprising one or more of a channel agent, a cholinergic agent, a nitric oxide donor, and related agents and their uses |
| WO2004046309A2 (en) | 2002-11-18 | 2004-06-03 | Phoenix Pharmacologics, Inc. | Methods for inhibiting viral replication in vivo |
| AU2003293529A1 (en) | 2002-12-16 | 2004-07-29 | Nitromed, Inc. | Nitrosated and nitrosylated rapamycin compounds, compositions and methods of use |
| WO2004064767A2 (en) | 2003-01-23 | 2004-08-05 | Kahn Nighat N | Nitric oxide inducing agents |
| FR2850578B1 (en) | 2003-02-03 | 2006-07-28 | Oreal | USE OF N-ARYLMETHYLENE ETHYLENEDIAMINETRIACETATES, N-ARYLMETHYLENEIMINODIACETATES OR N, N'-DIARYLMETHYLENE ETHYLENEDIAMINEACETATES AS DONORS OF NO |
| ATE432258T1 (en) | 2003-03-05 | 2009-06-15 | Merck Frosst Company | NITROGEN MONOXIDE RELEASING PRODRUGS OF DIARYL-2-(5H)-FURANONES AS INHIBITORS OF CYCLOOXYGENASE-2 |
| CA2518506A1 (en) | 2003-03-13 | 2004-11-18 | Nitromed, Inc. | Nitrosated and nitrosylated compounds, compositions and methods of use |
| SE0300971D0 (en) | 2003-04-03 | 2003-04-03 | Aga Ab | Nitric oxide in the treatment of inflammation |
| US20040254419A1 (en) | 2003-04-08 | 2004-12-16 | Xingwu Wang | Therapeutic assembly |
| US20050079132A1 (en) | 2003-04-08 | 2005-04-14 | Xingwu Wang | Medical device with low magnetic susceptibility |
| US7972137B2 (en) | 2003-06-30 | 2011-07-05 | Rosen Gerald M | Anti-microbial dental formulations for the prevention and treatment of oral mucosal disease |
| PL1648826T3 (en) | 2003-07-03 | 2018-02-28 | The University Court Of The University Of St. Andrews | Zeolites for delivery of nitric oxide |
| US7234079B2 (en) | 2003-07-11 | 2007-06-19 | Agency For Science, Technology & Research | Method and system for enabling recovery of data stored in a computer network; a method and a system for recovering data stored in a computer network |
| US20050054714A1 (en) | 2003-07-17 | 2005-03-10 | Benito Munoz | Nitric oxide releasing drugs for Alzheimer's disease |
| EP1648527A4 (en) | 2003-07-25 | 2008-04-23 | Univ Akron | Stabilization and ionic triggering of nitric oxide release |
| US7364585B2 (en) | 2003-08-11 | 2008-04-29 | Boston Scientific Scimed, Inc. | Medical devices comprising drug-loaded capsules for localized drug delivery |
| US7314857B2 (en) | 2003-08-25 | 2008-01-01 | Kane Biotech Inc. | Synergistic antimicrobial compositions and methods of inhibiting biofilm formation |
| AU2004270161A1 (en) | 2003-08-28 | 2005-03-17 | Nicox S.A. | Nitrosated and nitrosylated cardiovascular compounds, compositions and methods of use |
| JP2007518697A (en) | 2003-09-26 | 2007-07-12 | ニトロメッド インコーポレーティッド | Nitrosated glutamic acid compounds, compositions, and methods of use |
| AU2004275868A1 (en) | 2003-09-26 | 2005-04-07 | David R. Whitlock | Methods of using ammonia oxidizing bacteria |
| US7485324B2 (en) | 2003-09-29 | 2009-02-03 | Pulmonox Technologies Corporation | Use of exogenous gaseous nitric oxide in the treatment and disinfection of biofilms |
| US20050074506A1 (en) | 2003-10-02 | 2005-04-07 | Brainsgate Ltd. | Targeted release of nitric oxide in the CNS circulation for modulating the BBB and treating disorders |
| EP1691856A2 (en) | 2003-10-14 | 2006-08-23 | Cube Medical A/S | Medical device with electrospun nanofibers |
| US20060286159A1 (en) | 2003-10-17 | 2006-12-21 | Calvert Murrell George A | Treatment of persistent active tendinopathy using transdermal glyceryl trinitrate providing durability of effect |
| US20070053966A1 (en) | 2003-10-17 | 2007-03-08 | Robert Ang | Medicated orthopedic support structures for treatment of damaged musculoskeletal tissue |
| US20070059351A1 (en) | 2003-10-17 | 2007-03-15 | Murrell George A C | Transdermal patches containing a nitric oxide-donor and a second active agent and associated methods |
| US20050171199A1 (en) | 2003-10-17 | 2005-08-04 | New York Society For The Ruptured And Crippled Maintaining The Hospital For Special Surgery | Treatment of overuse tendinopathy using transdermal nitric oxide-generating agents |
| US20060286158A1 (en) | 2003-10-17 | 2006-12-21 | Calvert Murrell George A | Treatment of overuse tendinopathy using transdermal nitric oxide-generating agents |
| AU2004296174A1 (en) | 2003-12-15 | 2005-06-23 | Nitricare Hb | Device and method for administering therapeutic agents |
| CA2552735C (en) | 2004-01-07 | 2012-09-11 | Noxilizer, Inc. | Sterilization system and device |
| US8017074B2 (en) | 2004-01-07 | 2011-09-13 | Noxilizer, Inc. | Sterilization system and device |
| JP2007526911A (en) | 2004-01-22 | 2007-09-20 | ザ ユニバーシティ オブ アクロン | Polymer NO Donor Predrug Nanofiber Coating for Medical Devices and Therapy |
| WO2005070006A2 (en) | 2004-01-22 | 2005-08-04 | Nitromed, Inc. | Nitrosated and/or nitrosylated compounds, compositions and methods of use |
| AU2005206229A1 (en) | 2004-01-27 | 2005-08-04 | Merck Frosst Company | Nitric oxide releasing prodrugs of diaryl-2-(5H)-furanones as cyclooxygenase-2 inhibitors |
| CA2554334A1 (en) | 2004-01-27 | 2005-08-04 | Merck Frosst Company | Nitric oxide releasing prodrugs of diaryl-2-(5h)-furanones as cyclooxygenase-2 inhibitors |
| US20050165452A1 (en) | 2004-01-28 | 2005-07-28 | Medtronic, Inc. | Antithrombogenic medical device |
| US7569559B2 (en) | 2004-02-09 | 2009-08-04 | Noxilizer, Inc. | Nitric oxide-releasing molecules |
| US7829553B2 (en) | 2004-02-09 | 2010-11-09 | Amulet Pharmaceuticals, Inc. | Nitric oxide-releasing polymers |
| US20070166227A1 (en) | 2004-02-10 | 2007-07-19 | Shuang Liu | Crowned dithiocarbamate metal complexes and methods for their use |
| EP1732577B1 (en) | 2004-02-23 | 2013-04-10 | Strategic Science & Technologies, LLC | Topical delivery of l-arginine to improve body and skin appearance |
| US20070191377A1 (en) | 2004-03-31 | 2007-08-16 | Nitromed, Inc. | Methods for treating blood disorders with nitric oxide donor compounds |
| CA2563678A1 (en) | 2004-04-19 | 2005-11-03 | Strategic Science & Technologies, Llc | Beneficial effects of increasing local blood flow |
| WO2005115440A2 (en) | 2004-04-29 | 2005-12-08 | Kahn Nighat N | Compositions and method for modulating insulin-activated nitric oxide synthase |
| KR20070040332A (en) | 2004-05-03 | 2007-04-16 | 네오실 인코퍼레이티드 | Polycationic antimicrobial therapeutic |
| WO2005112954A1 (en) | 2004-05-14 | 2005-12-01 | William Marsh Rice University | Nitric oxide releasing compositions and associated methods |
| US8791073B2 (en) | 2004-05-14 | 2014-07-29 | William Marsh Rice University | Peptide-modified polyurethane compositions and associated methods |
| US7582623B2 (en) | 2004-05-20 | 2009-09-01 | The Regents Of The University Of California | Photoactive metal nitrosyls for blood pressure regulation and cancer therapy |
| DE602005018222D1 (en) | 2004-06-10 | 2010-01-21 | Nippon Kayaku Kk | |
| US20060008529A1 (en) | 2004-07-12 | 2006-01-12 | Meyerhoff Mark E | Use of additive sites to control nitric oxide release from nitric oxide donors contained within polymers |
| US20060039950A1 (en) | 2004-08-23 | 2006-02-23 | Zhengrong Zhou | Multi-functional biocompatible coatings for intravascular devices |
| EP1794195B1 (en) | 2004-09-27 | 2014-12-17 | Government of the United States of America, Represented by the Secretary, Department of Health and Human Services | Nitric oxide-releasing diazeniumdiolated acrylonitrile-based polymers, and compositions, medical devices, and uses thereof |
| US20070154570A1 (en) | 2004-09-29 | 2007-07-05 | Miller Christopher C | Use of nitric oxide in the treatment and disinfection of biofilms |
| WO2006041855A2 (en) | 2004-10-04 | 2006-04-20 | Nitromed, Inc. | Compositions and methods using apocynin compounds and nitric oxide donors |
| FR2877004B1 (en) | 2004-10-21 | 2007-03-09 | Oreal | ESTERS AND SILANIC AMIDES OF 2-OXOTHIAZOLIDINE-4-CARBOXYLIC ACID AND THEIR COSMETIC USES. |
| US8388677B2 (en) | 2004-10-29 | 2013-03-05 | Boston Scientific Scimed, Inc. | Anti-thrombogenic and anti-restenotic vascular medical devices |
| US20080275093A1 (en) | 2004-11-15 | 2008-11-06 | Nitromed, Inc. | Diuretic Compounds Comprising Heterocyclic Nitric Oxide Donor Groups, Compositions and Methods of Use |
| EP1827332A4 (en) | 2004-11-29 | 2010-03-17 | Univ Akron | Topical nitric oxide donor devices and methods for their therapeutic use |
| EP1681068A1 (en) | 2004-12-16 | 2006-07-19 | NOLabs AB | Anti-pathogen and anti-cancer filtering device and method comprising nitric oxide |
| WO2006064056A2 (en) | 2004-12-16 | 2006-06-22 | Nolabs Ab | Anti-pathogen and anti-cancer filtering device and method comprising nitric oxide |
| JP2008525321A (en) | 2004-12-24 | 2008-07-17 | ザ ユニバーシティー オブ クイーンズランド | Pain therapy |
| AU2006210952B2 (en) | 2005-01-31 | 2011-08-04 | Mylan Laboratories, Inc. | Pharmaceutical composition comprising hydroxylated nebivolol |
| EP1846009A2 (en) | 2005-02-11 | 2007-10-24 | NOLabs AB | Improved device for application of medicaments, manufacturing method therefor, and method of treatment |
| JP5175107B2 (en) | 2005-02-11 | 2013-04-03 | ノーラブズ エービー | Nitrogen oxide neuropathy treatment device, method and use |
| EP1690557A1 (en) | 2005-02-11 | 2006-08-16 | NOLabs AB | Device for treatment of rectal disorders, and manufacturing process for the same |
| WO2006084913A2 (en) | 2005-02-11 | 2006-08-17 | Nolabs Ab | Device for treatment of rectal disorders, and manufacturing process for the same, involving nitric oxide |
| EP1690532A1 (en) | 2005-02-11 | 2006-08-16 | NOLabs AB | Device for gastric treatment and manufacturing process for the same |
| EP1690554A1 (en) | 2005-02-11 | 2006-08-16 | NOLabs AB | Device for treatment of infections, including dermatophytosis and onychomycosis |
| EP1690558A1 (en) | 2005-02-11 | 2006-08-16 | NOLabs AB | Device for treatment of diabetic disorders |
| PT1861130E (en) | 2005-02-11 | 2008-12-02 | Nolabs Ab | Device and method for treatment of dermatomycosis, and in particular onychomycosis |
| EP1757278A1 (en) | 2005-08-23 | 2007-02-28 | NOLabs AB | Device, system, and method comprising microencapsulated liquid for release of nitric oxide from a polymer |
| EP1700611A1 (en) | 2005-02-11 | 2006-09-13 | NOLabs AB | Device for treatment of disorders in the oral cavity, and manufacturing process for the same |
| WO2006084914A2 (en) | 2005-02-11 | 2006-08-17 | Nolabs Ab | Device for gastric treatment and manufacturing process for the same |
| JP2008530226A (en) | 2005-02-16 | 2008-08-07 | ニトロメッド インコーポレーティッド | Organic nitric oxide donor salts, compositions, and methods of use of antimicrobial compounds |
| WO2006091716A2 (en) | 2005-02-24 | 2006-08-31 | Nitromed, Inc. | Nitric oxide enhancing diuretic compounds, compositions and methods of use |
| US8119840B2 (en) | 2005-03-04 | 2012-02-21 | The University Of Akron | Ethambutol based nitric oxide donors |
| JP2008533031A (en) | 2005-03-09 | 2008-08-21 | ニトロメッド インコーポレーティッド | Organic nitric oxide enhancing salts, compositions and methods of use of angiotensin II antagonists |
| GB0505035D0 (en) | 2005-03-11 | 2005-04-20 | Insense Ltd | Improvements relating to skin dressings |
| WO2006097348A1 (en) | 2005-03-15 | 2006-09-21 | L'oreal | Use of agents such as nonpolymeric nitric oxide donors for making the lips full again and/or colouring the lips |
| BRPI0611552A2 (en) | 2005-03-24 | 2010-11-23 | Nolabs Ab | intravascular, interstitial or intra-organ medical access device, process for making an intravascular, interstitial or intra-organ medical access device, use of an eluting nitric oxide polymer (no) and therapeutic method for preventing infection and / or obtaining antithrombotic effect |
| EP1704879A1 (en) | 2005-03-24 | 2006-09-27 | NOLabs AB | Intravascular, interstitial or intraorgan medical device comprising a nitric oxide eluting polymer |
| EP1704876A1 (en) | 2005-03-24 | 2006-09-27 | NOLabs AB | Cosmetic treatment, device for performing said treatment and manufacturing method thereof |
| PL1871433T3 (en) | 2005-03-24 | 2009-09-30 | Nolabs Ab | Cosmetic treatment with nitric oxide, device for performing said treatment and manufacturing method therefor |
| EP1704877A1 (en) | 2005-03-24 | 2006-09-27 | NOLabs AB | A wound care device comprising a nitric oxide eluting polymer |
| WO2006100155A1 (en) | 2005-03-24 | 2006-09-28 | Nolabs Ab | Device for wound care, and manufacturing method thereof, involving the use of nitric oxide |
| US20080214646A1 (en) | 2005-05-16 | 2008-09-04 | Knaus Edward E | Novel Nsaids Possessing a Nitric Oxide Donor Diazen-1-Ium-1,2-Diolate Moiety |
| CA2605269C (en) | 2005-05-17 | 2012-12-18 | Syntach Ag | A device and kit for treatment of disorders in the heart rhythm regulation system |
| US20090048219A1 (en) | 2005-05-23 | 2009-02-19 | Nitromed Inc. | Organic nitric oxide donor salts of nonsteroidal antiinflammatory compounds, compositions and methods of use |
| EP1899451B1 (en) | 2005-05-24 | 2016-10-12 | NewSouth Innovations Pty Limited | Methods and compositions for regulating biofilm development |
| US20100016790A1 (en) | 2005-06-01 | 2010-01-21 | Tor Peters | Treatment And Pre-Treatment Device, And Manufacturing Method Therefor, Involving Nitric Oxide |
| WO2006128743A1 (en) | 2005-06-01 | 2006-12-07 | Nolabs Ab | Feedstuff |
| EP1731176A1 (en) | 2005-06-01 | 2006-12-13 | NOLabs AB | Pre-treatment device comprising nitric oxide |
| EP1728438A1 (en) | 2005-06-01 | 2006-12-06 | NOLabs AB | Feedstuff |
| US8106080B2 (en) | 2005-06-09 | 2012-01-31 | Gu-Qi Wang | Compositions and methods for enhancing nitric oxide delivery |
| WO2007005758A2 (en) | 2005-06-30 | 2007-01-11 | Mc3, Inc. | Methods , compositions and devices for promoting angiogenesis |
| CA2613108A1 (en) | 2005-06-30 | 2007-01-11 | Mc3, Inc. | Nitric oxide coatings for medical devices |
| US20080139450A1 (en) | 2005-07-01 | 2008-06-12 | Srinivasa Madhyastha | Antimicrobial Compositions and Uses Thereof |
| AU2006265707B2 (en) | 2005-07-01 | 2012-06-14 | Kane Biotech Inc. | Antimicrobial compositions for inhibiting growth and proliferation of a microbial biofilm on medical devices |
| BRPI0503201B8 (en) | 2005-07-28 | 2021-05-25 | Sci Tech Produtos Medicos Ltda | stents coated with hydrophilic polymer blends, eluting nitric oxide and s-nitrosothiols |
| US20090018091A1 (en) | 2005-08-02 | 2009-01-15 | Nitromed, Inc. | Nitric Oxide Enhancing Antimicrobial Compounds, Compositions and Methods of Use |
| US8021679B2 (en) | 2005-08-25 | 2011-09-20 | Medtronic Vascular, Inc | Nitric oxide-releasing biodegradable polymers useful as medical devices and coatings therefore |
| ATE510464T1 (en) | 2005-08-26 | 2011-06-15 | Nestec Sa | COMPOSITIONS AND METHODS FOR IMPROVING FUNCTIONAL VASCULAR INTEGRITY, CELL SURVIVAL AND REDUCING APOPTOSIS AFTER AN ISSUE OF BRAIN |
| US20070048344A1 (en) | 2005-08-31 | 2007-03-01 | Ali Yahiaoui | Antimicrobial composition |
| US20070053952A1 (en) | 2005-09-07 | 2007-03-08 | Medtronic Vascular, Inc. | Nitric oxide-releasing polymers derived from modified polymers |
| EP1764119A1 (en) | 2005-09-09 | 2007-03-21 | NOLabs AB | Implants with improved osteointegration |
| US20090010989A1 (en) | 2005-09-12 | 2009-01-08 | N0Labs Ab | Coating For Implants and Implants With Improved Osteointegration, and Manufacturing Method |
| US20070088345A1 (en) | 2005-10-13 | 2007-04-19 | Ust Inc. | Applications of HIFU and chemotherapy |
| US7531164B2 (en) | 2005-10-21 | 2009-05-12 | Duke University | Preventing bacterial or viral infectivity and composition containing infection preventing additive |
| AU2006309212B2 (en) | 2005-10-31 | 2011-09-15 | Government Of The United States Of America, Represented By The Secretary Department Of Health And Human Services | Polysaccharide-derived nitric oxide-releasing carbon-bound diazeniumdiolates |
| WO2007053578A2 (en) | 2005-10-31 | 2007-05-10 | Amulet Pharmaceuticals, Inc. | Multi-phasic nitric oxide and drug co-eluting stent coatings |
| EP1790335A1 (en) | 2005-11-14 | 2007-05-30 | NOLabs AB | Composition and its use for the manufacture of a medicament for treating, prophylactically treating, preventing cancer and/or infections in the urinary tract |
| WO2007059311A2 (en) | 2005-11-16 | 2007-05-24 | Nitromed, Inc. | Furoxan compounds, compositions and methods of use |
| WO2007057763A2 (en) | 2005-11-18 | 2007-05-24 | Pulmonox Technologies Corporation | Nitric oxide as an anti-viral agent, vaccine and vaccine adjuvant |
| JP2009518069A (en) | 2005-12-02 | 2009-05-07 | ザ・リージェンツ・オブ・ザ・ユニバーシティ・オブ・ミシガン | Compositions, coatings and devices, and methods for making and using them |
| US20070129690A1 (en) | 2005-12-02 | 2007-06-07 | Joel Rosenblatt | Catheter with polymeric coating |
| AU2006322035A1 (en) | 2005-12-06 | 2007-06-14 | Amulet Pharmaceuticals, Inc. | Nitric oxide-releasing polymers |
| JP2009521481A (en) | 2005-12-21 | 2009-06-04 | ナイトロックス, エルエルシー | Stable S-nitrosothiol formulation |
| US9801902B2 (en) | 2006-01-17 | 2017-10-31 | The University Of Akron | Debridement method using topical nitric oxide donor devices and compositions |
| WO2007087631A2 (en) | 2006-01-26 | 2007-08-02 | Bebaas, Inc. | Cobalamin compositions for the treatment of cancer |
| EP1983975B1 (en) | 2006-02-03 | 2015-07-29 | Giulio Cossu | Method of treatment for muscular dystrophy |
| US20070202155A1 (en) | 2006-02-03 | 2007-08-30 | Cure Therapeutics, Inc. | Low dose no donor-containing transdermal patch |
| JP5745210B2 (en) | 2006-02-03 | 2015-07-08 | ニコックス サイエンス アイルランド | Use of nitrooxy derivatives as therapeutic agents for muscular dystrophy |
| US20070219208A1 (en) | 2006-02-27 | 2007-09-20 | Balaraman Kalyanaraman | Methods for Treating Cancer |
| US20070203242A1 (en) | 2006-02-27 | 2007-08-30 | Calwood Nutritionals, Inc. | Method for treating gastric reflux |
| US7645748B2 (en) | 2006-04-03 | 2010-01-12 | Forbes Medi-Tech Inc. | Sterol/stanol phosphorylnitroderivatives and use thereof |
| US7645749B2 (en) | 2006-04-03 | 2010-01-12 | Forbes Medi-Tech Inc. | Sterol/stanol nitroderivatives and use thereof |
| AU2007244903B2 (en) | 2006-04-24 | 2012-02-02 | The Government Of The United States Of America As Represented By The Secretary Department Of Health And Human Services | Diazeniumdiolated non-steroidal anti-inflammatory drugs, compositions thereof, and related methods |
| US8241619B2 (en) | 2006-05-15 | 2012-08-14 | Medtronic Vascular, Inc. | Hindered amine nitric oxide donating polymers for coating medical devices |
| US20080171351A1 (en) | 2006-05-22 | 2008-07-17 | University Of Notre Dame Du Lac | Probes for anionic cell surface detection |
| US8455255B2 (en) * | 2006-06-08 | 2013-06-04 | The University Of Tokushima | Method for production of novel nano silica particle and use of the nano silica particle |
| US20090263416A1 (en) | 2006-06-16 | 2009-10-22 | Dawson Karl A | Reduction of antibiotic resistance in bacteria |
| WO2007149520A2 (en) | 2006-06-21 | 2007-12-27 | Albert Einstein College Of Medicine Of Yeshiva University | Compositions for sustained release of nitric oxide, methods of preparing same and uses thereof |
| US20080025972A1 (en) | 2006-07-26 | 2008-01-31 | Duke University | Treating sex steriod responsive disorders |
| EP2051935B1 (en) | 2006-07-26 | 2016-09-07 | STMicroelectronics Srl | Use of nitroaniline derivatives for the production of nitric oxide |
| GB0616350D0 (en) | 2006-08-17 | 2006-09-27 | Univ St Andrews | Adsorption and release of nitric oxide in metal organic frameworks |
| EP1895012A1 (en) | 2006-08-30 | 2008-03-05 | Universitätsklinikum Freiburg | Method for inducing tumor apoptosis by increasing nitric oxide levels |
| GB0618711D0 (en) | 2006-09-22 | 2006-11-01 | Univ Exeter | Agricultural treatment |
| GB0623531D0 (en) | 2006-11-25 | 2007-01-03 | Univ St Andrews | Nitric oxide storage and production |
| US8221690B2 (en) | 2007-10-30 | 2012-07-17 | The Invention Science Fund I, Llc | Systems and devices that utilize photolyzable nitric oxide donors |
| US7862598B2 (en) | 2007-10-30 | 2011-01-04 | The Invention Science Fund I, Llc | Devices and systems that deliver nitric oxide |
| US8642093B2 (en) | 2007-10-30 | 2014-02-04 | The Invention Science Fund I, Llc | Methods and systems for use of photolyzable nitric oxide donors |
| US20090110933A1 (en) | 2007-10-30 | 2009-04-30 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems and devices related to nitric oxide releasing materials |
| US20090112197A1 (en) | 2007-10-30 | 2009-04-30 | Searete Llc | Devices configured to facilitate release of nitric oxide |
| US20080175881A1 (en) | 2007-01-18 | 2008-07-24 | Boston Scientific Scimed, Inc. | Blood-contacting medical devices for the release of nitric oxide and anti-restenotic agents |
| WO2008094275A1 (en) | 2007-01-30 | 2008-08-07 | New York University | Peptides for treatment of conditions associated with nitric oxide |
| CA2677387C (en) | 2007-02-05 | 2016-06-21 | Nicox S.A. | Nitric oxide donor compounds |
| US20080193385A1 (en) | 2007-02-08 | 2008-08-14 | Todd Maibach | Compositions and methods for treating neuropathy |
| US20080193566A1 (en) | 2007-02-09 | 2008-08-14 | Miller Christopher C | Use of high dose concentrations of gaseous nitric oxide |
| WO2008100591A2 (en) | 2007-02-14 | 2008-08-21 | The General Hospital Corporation | Modulation of nitric oxide signaling to normalize tumor vasculature |
| US7811600B2 (en) | 2007-03-08 | 2010-10-12 | Medtronic Vascular, Inc. | Nitric oxide donating medical devices and methods of making same |
| BRPI0721507A2 (en) | 2007-03-27 | 2013-01-08 | Nolabs Ab | Typical Nitric Oxide Dispensing Device, Kit, Nitric Oxide Usage, and Typical Nitric Oxide Dispensing and Treatment Methods |
| US8598368B2 (en) | 2007-04-20 | 2013-12-03 | Duke University | Stable neutral nitric oxide source |
| WO2008150505A1 (en) | 2007-06-01 | 2008-12-11 | Amulet Pharmaceuticals, Inc. | Compounds, polymers and methods for treating gastrointestinal dysfunction |
| WO2008157393A1 (en) | 2007-06-15 | 2008-12-24 | Ikaria, Inc. | Compositions comprising sulfide alone or in combination with nitric oxide and their use to |
| US8273828B2 (en) | 2007-07-24 | 2012-09-25 | Medtronic Vascular, Inc. | Methods for introducing reactive secondary amines pendant to polymers backbones that are useful for diazeniumdiolation |
| WO2009014616A1 (en) | 2007-07-26 | 2009-01-29 | Duke University | Measuring amount of bound and combined nitric oxide in blood |
| GB0715554D0 (en) | 2007-08-09 | 2007-09-19 | Insense Ltd | Improvements relating to skin dressings |
| GB0715556D0 (en) | 2007-08-09 | 2007-09-19 | Insense Ltd | Improvements relating to skin dressings |
| CA2599082A1 (en) | 2007-08-27 | 2009-02-27 | Ping I. Lee | Supramacromolecular polymer complexes providing controlled nitric oxide release for healing wounds |
| WO2009071990A2 (en) | 2007-08-31 | 2009-06-11 | Topigen Pharmaceuticals Inc. | No-donating cordicosteroid with improved pharmacokinetic, anti-inflammatory and vasodilatory properties |
| WO2009036571A1 (en) | 2007-09-21 | 2009-03-26 | Enox Biopharma, Inc. | Antimicrobial gas-releasing ear drainage tubes |
| US8399005B2 (en) | 2007-10-12 | 2013-03-19 | University Of North Carolina At Chapel Hill | Use of nitric oxide to enhance the efficacy of silver and other topical wound care agents |
| US7897399B2 (en) | 2007-10-30 | 2011-03-01 | The Invention Science Fund I, Llc | Nitric oxide sensors and systems |
| US8877508B2 (en) | 2007-10-30 | 2014-11-04 | The Invention Science Fund I, Llc | Devices and systems that deliver nitric oxide |
| US20090112193A1 (en) | 2007-10-30 | 2009-04-30 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems and devices that utilize photolyzable nitric oxide donors |
| US7846400B2 (en) | 2007-10-30 | 2010-12-07 | The Invention Science Fund I, Llc | Substrates for nitric oxide releasing devices |
| US20090112055A1 (en) | 2007-10-30 | 2009-04-30 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Sleeves configured to facilitate release of nitric oxide |
| WO2009064861A2 (en) | 2007-11-13 | 2009-05-22 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Nitric oxide-releasing diazeniumdiolated polymers, compositions, medical devices, and uses thereof |
| WO2009073643A2 (en) | 2007-12-03 | 2009-06-11 | Cleveland State University | Nitric oxide release coatings incorporating nitric oxide synthase enzyme |
| JP5709292B2 (en) * | 2007-12-06 | 2015-04-30 | 国立大学法人徳島大学 | Nano-functional silica particles and method for producing the same |
| BRPI0705396B1 (en) | 2007-12-12 | 2021-10-05 | Universidade Estadual De Campinas - Unicamp | PHTHLIMID DERIVED COMPOUNDS |
| IE20070934A1 (en) | 2007-12-21 | 2009-06-24 | Trinity College Dublin | Efficient aspirin prodrugs |
| AU2008345034A1 (en) | 2007-12-27 | 2009-07-09 | Aires Pharmaceuticals, Inc. | Aerosolized nitrite and nitric oxide - donating compounds and uses thereof |
| CA2994586C (en) | 2008-01-04 | 2020-07-21 | Nestec S.A. | Compositions comprising unsaturated fatty acids and nitric oxide releasing compounds and use thereof for enhancing cognitive and related functions |
| US8720436B2 (en) | 2008-01-31 | 2014-05-13 | Genosys, Inc. | Nitric oxide gel apparatus and method |
| JP5623916B2 (en) | 2008-01-31 | 2014-11-12 | バンダービルト・ユニバーシティVanderbilt University | Pharmaceutical composition for aneurysmal subarachnoid hemorrhage and vasospasm |
| WO2009098113A1 (en) | 2008-02-07 | 2009-08-13 | Nicox S.A. | Nitric oxide donor compounds |
| US20090222088A1 (en) | 2008-02-29 | 2009-09-03 | Medtronic Vascular, Inc. | Secondary Amine Containing Nitric Oxide Releasing Polymer Composition |
| US20090232863A1 (en) | 2008-03-17 | 2009-09-17 | Medtronic Vascular, Inc. | Biodegradable Carbon Diazeniumdiolate Based Nitric Oxide Donating Polymers |
| US20090232868A1 (en) | 2008-03-17 | 2009-09-17 | Medtronic Vascular, Inc. | Nitric Oxide Releasing Polymer Composition |
| WO2009124379A1 (en) | 2008-04-09 | 2009-10-15 | Enox Biopharma, Inc. | Biofilm-inhibiting catheters and tubings |
| JP2011518222A (en) | 2008-04-21 | 2011-06-23 | スリーエム イノベイティブ プロパティズ カンパニー | Nitric oxide releasing composition, device and method |
| WO2009155690A1 (en) | 2008-06-24 | 2009-12-30 | Micropharma Limited | Nitric oxide compositions and devices and methods for cosmesis |
| US8852640B2 (en) | 2008-07-03 | 2014-10-07 | Ecole Polytechnique Federale De Lausanne (Epfl) | Micelles for delivery of nitric oxide |
| US20100040703A1 (en) | 2008-08-13 | 2010-02-18 | Chris Miller | Use of nitric oxide |
| JP5495337B2 (en) | 2008-09-19 | 2014-05-21 | ノースウエスタン ユニバーシティ | Biodegradable nitric oxide generating polymer and related medical devices |
| US7807716B2 (en) | 2008-09-24 | 2010-10-05 | Oral Delivery Technology Ltd. | Nitric oxide amino acid ester compound, compositions for increasing nitric oxide levels and method of use |
| US20100086530A1 (en) | 2008-10-06 | 2010-04-08 | Martinov Norman P | Cancer-tumor necrosis by artery nutrient-oxygen blocking |
| CA2740421C (en) | 2008-10-15 | 2018-08-28 | The University Of Memphis Research Foundation | Method and device for detection of bioavailable drug concentration in a fluid sample |
| US20120003293A1 (en) | 2008-10-30 | 2012-01-05 | Chris Miller | Nitric oxide-releasing dressings |
| US20100166603A1 (en) | 2008-10-31 | 2010-07-01 | Noxilizer, Inc. | Powder Sterilization |
| US8158187B2 (en) | 2008-12-19 | 2012-04-17 | Medtronic Vascular, Inc. | Dry diazeniumdiolation methods for producing nitric oxide releasing medical devices |
| US20100197702A1 (en) | 2009-02-04 | 2010-08-05 | Hellberg Mark R | Ophthalmic composition with nitric oxide donor compound and method of forming and using same |
| US8062653B2 (en) | 2009-02-18 | 2011-11-22 | Bezwada Biomedical, Llc | Controlled release of nitric oxide and drugs from functionalized macromers and oligomers |
| EP2398761A4 (en) | 2009-02-18 | 2015-11-04 | Bezwada Biomedical Llc | Controlled release of nitric oxide and drugs from functionalized macromers and oligomers |
| US9216198B2 (en) | 2009-03-30 | 2015-12-22 | The Board Of Regents, The University Of Texas System | Compositions and medical devices for controlled release of nitric oxide and methods of production thereof |
| US8236341B2 (en) | 2009-04-02 | 2012-08-07 | Medtronic Vascular, Inc. | Poly(tetrafluoroethylene) polymer with nitric oxide donating surface |
| US8709465B2 (en) | 2009-04-13 | 2014-04-29 | Medtronic Vascular, Inc. | Diazeniumdiolated phosphorylcholine polymers for nitric oxide release |
| EP2445508B1 (en) | 2009-06-22 | 2018-11-14 | VERO Biotech LLC | Nitric oxide therapies |
| US20110059189A1 (en) | 2009-09-08 | 2011-03-10 | Cisne Enterprises Inc. | Method and composition for treating cancer, effecting apoptosis and treating retroviral infections |
| ES2695173T3 (en) | 2011-02-28 | 2019-01-02 | Novan Inc | Silica particles modified with S-nitrosothiol that release nitric oxide and methods of manufacturing them |
-
2012
- 2012-02-28 ES ES12752627T patent/ES2695173T3/en active Active
- 2012-02-28 WO PCT/US2012/026972 patent/WO2012118829A2/en active Application Filing
- 2012-02-28 EP EP12752627.5A patent/EP2681286B1/en active Active
- 2012-02-28 WO PCT/US2012/026960 patent/WO2012118819A2/en active Application Filing
-
2013
- 2013-08-26 US US13/975,930 patent/US9713652B2/en active Active
- 2013-08-26 US US13/975,995 patent/US8981139B2/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090214618A1 (en) | 2005-05-27 | 2009-08-27 | Schoenfisch Mark H | Nitric oxide-releasing particles for nitric oxide therapeutics and biomedical applications |
Non-Patent Citations (5)
| Title |
|---|
| FROST ET AL., JOURNAL OF BIOMEDICAL MATERIALS RESEARCH, vol. 72A, 2005, pages 409 - 419 |
| RICCIO ET AL., BIOMATERIALS, vol. 30, 2009, pages 4494 - 4502 |
| See also references of EP2681286A4 |
| STOBER, W.; FINK, A.; BOHN, E. J., COLLOID INTERFACE SCI., vol. 26, 1968, pages 62 |
| WILLIAMS, D. L. H., ACC. CHE . RES., vol. 32, 1999, pages 869 |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8981139B2 (en) | 2011-02-28 | 2015-03-17 | The University Of North Carolina At Chapel Hill | Tertiary S-nitrosothiol-modified nitric—oxide-releasing xerogels and methods of using the same |
| US9713652B2 (en) | 2011-02-28 | 2017-07-25 | The University Of North Carolina At Chapel Hill | Nitric oxide-releasing S-nitrosothiol-modified silica particles and methods of making the same |
| US11077194B2 (en) | 2012-03-14 | 2021-08-03 | Novan, Inc. | Nitric oxide releasing pharmaceutical compositions |
| GB2517443A (en) * | 2013-08-20 | 2015-02-25 | Henkel IP & Holding GmbH | Improved bonding or vulcanisation compositions |
| US10322082B2 (en) | 2014-07-11 | 2019-06-18 | Novan, Inc. | Topical antiviral compositions and methods of using the same |
| US10322081B2 (en) | 2014-07-11 | 2019-06-18 | Novan, Inc. | Topical antiviral compositions and methods of using the same |
| US10736839B2 (en) | 2014-07-11 | 2020-08-11 | Novan, Inc. | Topical antiviral compositions, delivery systems, and methods of using the same |
| US11723858B2 (en) | 2014-07-11 | 2023-08-15 | Novan, Inc. | Topical antiviral compositions, delivery systems, and methods of using the same |
| US11040006B2 (en) | 2014-07-11 | 2021-06-22 | Novan, Inc. | Topical antiviral compositions, delivery systems, and methods of using the same |
| US10925689B2 (en) | 2014-07-14 | 2021-02-23 | Novan, Inc. | Nitric oxide releasing nail coating compositions, nitric oxide releasing nail coatings, and methods of using the same |
| CN107454849A (en) * | 2015-03-27 | 2017-12-08 | 萨卢庞特咨询有限公司 | The sterilizing methods of S nitrosothiolses |
| US10849864B2 (en) | 2015-07-28 | 2020-12-01 | Novan, Inc. | Combinations and methods for the treatment and/or prevention of fungal infections |
| US11534382B2 (en) | 2017-06-19 | 2022-12-27 | Novan, Inc. | Topical compositions and methods of using the same |
| US11285171B2 (en) | 2018-03-01 | 2022-03-29 | Novan, Inc. | Nitric oxide releasing suppositories and methods of use thereof |
| CN113507924A (en) * | 2019-03-01 | 2021-10-15 | 北卡罗来纳大学教堂山分校 | Nitric oxide extended release polymers via functionalized mesoporous silica nanoparticles |
| WO2020180705A1 (en) * | 2019-03-01 | 2020-09-10 | The University Of North Carolina At Chapel Hill | Extended nitric oxide-releasing polymers via functionalized mesoporous silica nanoparticles |
Also Published As
| Publication number | Publication date |
|---|---|
| US8981139B2 (en) | 2015-03-17 |
| EP2681286A4 (en) | 2015-08-05 |
| WO2012118829A3 (en) | 2014-04-17 |
| WO2012118819A3 (en) | 2014-04-17 |
| US20130344334A1 (en) | 2013-12-26 |
| US9713652B2 (en) | 2017-07-25 |
| EP2681286A2 (en) | 2014-01-08 |
| ES2695173T3 (en) | 2019-01-02 |
| EP2681286B1 (en) | 2018-08-15 |
| US20140017121A1 (en) | 2014-01-16 |
| WO2012118829A2 (en) | 2012-09-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9713652B2 (en) | Nitric oxide-releasing S-nitrosothiol-modified silica particles and methods of making the same | |
| US20220402944A1 (en) | Functionalized Mesoporous Silica via an Aminosilane Surfactant Ion Exchange Reaction: Controlled Scaffold Design and Nitric Oxide Release | |
| US10435357B2 (en) | Tunable nitric oxide-releasing macromolecules having multiple nitric oxide donor structures | |
| US9017643B2 (en) | Particles comprising a releasable dopant therein | |
| Michelot et al. | Spectroscopic characterisation of hydroxyapatite and nanocrystalline apatite with grafted aminopropyltriethoxysilane: nature of silane–surface interaction | |
| EP3105281B1 (en) | Materials and methods relating to stabilised polymeric silicate compositions | |
| Cao et al. | Fabrication of autofluorescent porous silica nanoparticles for redox-responsive drug release | |
| US9815910B2 (en) | Surface modification of cellulose nanocrystals | |
| Mandal et al. | Vibrational spectroscopic investigation on interaction of sago starch capped silver nanoparticles with collagen: a comparative physicochemical study using FT-IR and FT-Raman techniques | |
| US20220152219A1 (en) | Extended nitric oxide-releasing polymers via functionalized mesoporous silica nanoparticles | |
| CN112516321B (en) | Controlled release nano-drug carrier and preparation method and application thereof | |
| Garren et al. | Derivatization of graphene oxide nanosheets with tunable nitric oxide release for antibacterial biomaterials | |
| Zhou et al. | Highly functionalized bridged silsesquioxanes | |
| RU2421397C1 (en) | Organo-inorganic molecular silica sols and preparation method thereof | |
| Laird et al. | Mesoporous organosilicas with thiol functionalised pores: multifunctional dendrimers as sacrificial building block and template | |
| Riccio | S-nitrosothiol-derived nitric oxide delivery vehicles: Synthesis and detection | |
| US20230355662A1 (en) | Sulfur functionalized monoliths and particles derived from the same as nitric oxide carriers for pharmaceutical and cosmetic applications | |
| KR102495579B1 (en) | Core-corona type organic-inorganic hybridized nanoparticles having super water repellency, antifouling properties, antibacterial properties, and oil repellency, and a method for manufacturing the same | |
| US20230312342A1 (en) | Method of preparation of zinc-oxygen-based nanoparticles, zinc peroxide nanoparticles obtained by this method and their use | |
| AU2006257727B2 (en) | Particles comprising a releasable dopant therein | |
| Niembro et al. | Catalytically active palladium nanoparticles embedded in an organic-inorganic fluorinated hybrid material | |
| Dell'Arciprete et al. | Optimal Silicon-based nanomaterials for biological applications | |
| PL241213B1 (en) | Method of producing and controlling the size of silica nano-objects containing a fluorescent dye in their structure | |
| Bordianu et al. | Synthesis and characterization of a silsesquioxane nanocomposite with photoluminescence properties | |
| Rieter | Development of inorganic-organic hybrid nanomaterials for biological and biomedical applications |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12752627 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012752627 Country of ref document: EP |