WO2011011799A2 - Thiophene azo dyes and laundry care compositions containing the same - Google Patents
Thiophene azo dyes and laundry care compositions containing the same Download PDFInfo
- Publication number
- WO2011011799A2 WO2011011799A2 PCT/US2010/056508 US2010056508W WO2011011799A2 WO 2011011799 A2 WO2011011799 A2 WO 2011011799A2 US 2010056508 W US2010056508 W US 2010056508W WO 2011011799 A2 WO2011011799 A2 WO 2011011799A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- moiety
- alkyl
- laundry care
- care composition
- formula
- Prior art date
Links
- 0 *c1c(*)[s]c(N=N*)c1* Chemical compound *c1c(*)[s]c(N=N*)c1* 0.000 description 3
- PCPRCUZOHHJGET-UHFFFAOYSA-N CCN(CC(c1ccccc1)OCCOCCO)c(cc1)cc(C)c1N Chemical compound CCN(CC(c1ccccc1)OCCOCCO)c(cc1)cc(C)c1N PCPRCUZOHHJGET-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/40—Dyes ; Pigments
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B29/00—Monoazo dyes prepared by diazotising and coupling
- C09B29/06—Monoazo dyes prepared by diazotising and coupling from coupling components containing amino as the only directing group
- C09B29/08—Amino benzenes
- C09B29/0801—Amino benzenes containing acid groups, e.g. COOH, SO3H, PO3H2, OSO3H, OPO3H2; SO2NHSO2R or salts thereof, R being hydrocarbonyls
- C09B29/0802—Amino benzenes containing acid groups, e.g. COOH, SO3H, PO3H2, OSO3H, OPO3H2; SO2NHSO2R or salts thereof, R being hydrocarbonyls containing COOH
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09B—ORGANIC DYES OR CLOSELY-RELATED COMPOUNDS FOR PRODUCING DYES, e.g. PIGMENTS; MORDANTS; LAKES
- C09B29/00—Monoazo dyes prepared by diazotising and coupling
- C09B29/06—Monoazo dyes prepared by diazotising and coupling from coupling components containing amino as the only directing group
- C09B29/08—Amino benzenes
- C09B29/0801—Amino benzenes containing acid groups, e.g. COOH, SO3H, PO3H2, OSO3H, OPO3H2; SO2NHSO2R or salts thereof, R being hydrocarbonyls
- C09B29/0803—Amino benzenes containing acid groups, e.g. COOH, SO3H, PO3H2, OSO3H, OPO3H2; SO2NHSO2R or salts thereof, R being hydrocarbonyls containing SO3H, OSO3H
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/50—Perfumes
- C11D3/502—Protected perfumes
- C11D3/505—Protected perfumes encapsulated or adsorbed on a carrier, e.g. zeolite or clay
Definitions
- This application relates to thiophene azo dyes for use as hueing agents, laundry care compositions comprising such thiophene azo dyes, processes for making such thiophene azo dyes and laundry care compositions and methods of using the same.
- the aforementioned thiophene azo dyes contain a formally charged moiety and are generally comprised of at least two components: at least one chromophore component and at least one polymeric component. Suitable chromophore components generally fluoresce blue, red, violet, or purple color when exposed to ultraviolet light, or they may absorb light to reflect these same shades.
- thiophene azo dyes are advantageous in providing a hueing effect, for example, a whitening effect to fabrics, while not building up over time and causing undesirable blue discoloration to the treated fabrics.
- the thiophene azo dyes are also generally stable to bleaching agents used in laundry care compositions.
- hueing agents are generally to visually brighten these textile substrates and counteract the fading and yellowing of the substrates.
- hueing agents may be found in laundry detergents, fabric softeners, or rinse aids and are therefore applied to textile substrates during the laundering process.
- hueing agents function to visually brighten treated textile substrates without causing undesirable staining of the textile substrates.
- Cellulosic substrates in particular, tend to exhibit a yellow hue after exposure to light, air, and/or soiling. This yellowness is often difficult to reverse by normal laundering procedures.
- the hueing agents disclosed herein and the laundry care compositions comprising same offer advantages over previous efforts in this area, as, unlike previous thiophene azo hueing agents, the present hueing agents comprise a formally charged moiety that allows such hueing agents to provide enhanced deposition, removal and hue angle when used in compositions such as laundry care compositions.
- the present hueing agents offer improved stability in wash environments that contain bleaching agents, for example bleach boosters. While not being bound by theory, Applicants believe that such enhanced stability is, at least in part, due to the improved partitioning behavior of such hueing agents.
- the aforementioned benefits are offered over a range of fabric types. In short, Applicants recognized the source of the current hueing deficiencies and herein provide the solution to such problem.
- the hueing compounds disclosed herein also absorb light at a wavelength appropriate to visually neutralize the yellowness of substrates, including textile substrates. These compounds function ideally as hueing agents for substrates, including textile substrates, and may be incorporated into laundry care compositions for use by consumers.
- This application relates to thiophene azo dyes for use as hueing agents, laundry care compositions comprising such thiophene azo dyes that may serve as hueing agents, processes for making such thiophene azo dyes and laundry care compositions and methods of using the same.
- the aforementioned thiophene azo dyes contain a formally charged moiety and are generally comprised of at least two components: at least one chromophore component and at least one polymeric component. Suitable chromophore components generally fluoresce blue, red, violet, or purple color when exposed to ultraviolet light, or they may absorb light to reflect these same shades.
- thiophene azo dyes are advantageous in providing a hueing effect, for example, a whitening effect to fabrics, while not building up over time and causing undesirable blue discoloration to the treated fabrics.
- the thiophene azo dyes are also generally stable to bleaching agents used in laundry care compositions.
- alkoxy is intended to include C 1 -C 8 alkoxy and alkoxy derivatives of polyols having repeating units such as butylene oxide, glycidol oxide, ethylene oxide or propylene oxide.
- alkyl and “alkyl capped” are intended to include C 1 -C 18 alkyl groups, and in one aspect, C 1 -C 6 alkyl groups.
- aryl is intended to include C 3 -C 12 aryl groups.
- arylalkyl is intended to include C 1 -C 18 alkyl groups and, in one aspect, C 1 -C 6 alkyl groups.
- formally charged moiety means a moiety having at least one formal positive charge or at least one formal negative charge in aqueous solution at a pH in the range from 7 to 11.
- ethylene oxide "propylene oxide” and “butylene oxide” may be shown herein by their typical designation of “EO,” “PO” and “BO,” respectively.
- laundry care composition includes, unless otherwise indicated, granular, powder, liquid, gel, paste, unit dose bar form and/or flake type washing agents and/or fabric treatment compositions.
- fabric treatment composition includes, unless otherwise indicated, fabric softening compositions, fabric enhancing compositions, fabric freshening compositions and combinations there of. Such compositions may be, but need not be rinse added compositions.
- cellulosic substrates are intended to include any substrate which comprises at least a majority by weight of cellulose.
- Cellulose may be found in wood, cotton, linen, jute, and hemp.
- Cellulosic substrates may be in the form of powders, fibers, pulp and articles formed from powders, fibers and pulp.
- Cellulosic fibers include, without limitation, cotton, rayon (regenerated cellulose), acetate (cellulose acetate), triacetate (cellulose triacetate), and mixtures thereof.
- Articles formed from cellulosic fibers include textile articles such as fabrics.
- Articles formed from pulp include paper.
- maximum extinction coefficient is intended to describe the molar extinction coefficient at the maximum wavelength in the range of 400 nanometers to 750 nanometers.
- the thiophene azo dyes disclosed herein may contain a distribution of repeating units in their polymeric moiety. Accordingly, in one aspect, the molecular weight of the thiophene azo dyes disclosed herein may be reported as an average molecular weight, as determined by its molecular weight distribution.
- test methods disclosed in the Test Methods Section of the present application should be used to determine the respective values of the parameters of Applicants' inventions.
- component or composition levels are in reference to the active portion of that component or composition, and are exclusive of impurities, for example, residual solvents or by-products, which may be present in commercially available sources of such components or compositions.
- Laundry Care Composition Comprising Suitable Hueing Agents
- a laundry care composition comprising a hueing agent and one or more laundry care ingredients is disclosed.
- Suitable hueing agents include all the hueing agents listed below.
- the hueing agents of the present invention include thiophene azo dyes which contain a formally charged moiety. However, these thiophene azo dyes do not comprise a meta-bis(2- hydroxy-3-trimethylammonium propyl) amino tolyl group.
- the aforementioned thiophene azo dyes comprise a thiophene moiety, an azo moiety and a coupler moiety, said thiophene moiety being covalently bound to said azo moiety and said coupler being covalently bound to said azo moiety, at least one of said thiophene and/or azo moieties comprising a formally charged moiety.
- the hueing agents of the present invention may be dyes, pigments, or polymeric colorants generally comprising a chromophore constituent and a polymeric constituent.
- the chromophore constituent is characterized in that it emits or absorbs wavelength in the range of blue, red, violet, purple, or combinations thereof upon exposure to light.
- the chromophore constituent exhibits an absorbance spectrum maximum in the wavelength range of about 400 nanometers to about 750 nanometers, in another aspect of about 520 nanometers to about 650 nanometers, in yet another aspect of about 540 nanometers to about 630 nanometers, in another aspect of about 560 nanometers to about 610 nanometers, in another aspect of about 565 nanometers to about 580 nanometers in methanol solution.
- suitable polymeric constituents include polyoxyalkylene chains having multiple repeating units.
- the polymeric constituents include polyoxyalkylene chains having from 2 to about 30 repeating units, from 2 to about 20 repeating units, from 2 to about 10 repeating units or even from about 3 or 4 to about 6 repeating units.
- Non-limiting examples of polyoxyalkylene chains include ethylene oxide, propylene oxide, glycidol oxide, butylene oxide and mixtures thereof.
- the thiophene azo dye contains a formally charged moiety, with the proviso that the dye does not comprise a meta-bis(2-hydroxy-3- trimethylammoniumpropyl) amino tolyl group.
- the thiophene azo dye exhibits, in the wavelength range of about 400 nm to about 750 nm in methanol solution, or of about 520 nm to about 650 nm in methanol solution, or of about 540 nm to about 630 nm in methanol solution, or of about 560 nm to about 610 nm in methanol solution, or of about 565 nm to about 580 nm in methanol solution, a maximum extinction coefficient from about 1000 to about 1,000,000 liter/mol/cm, or from about 5,000 to about 750,000 liter/mol/cm, or from about 10,000 to about 500,000 liter/mol/cm, or from about 20,000 to about 250,000 liter/mol/cm.
- the thiophene azo dye exhibits a molecular weight from greater than 300 daltons, or from about 300 daltons to about 5000 daltons, or from about 350 daltons to about 3000 daltons, or from about 400 daltons to about 1500 daltons.
- the thiophene azo dye exhibits an aqueous partition value from about 10% to 100% or from about 20% to 100% or from about 30% to 100% or from about 40% to 100%, said dye comprising a non-covalently bound charge balancing counterion.
- the thiophene azo dye further exhibits an aqueous partition value from 0% to about 40%, from 0% to about 30%, from 0% to about 20%, or from about 1% to about 10%, said dye comprising a covalently bound charge balancing counterion.
- the thiophene azo dye of the present invention may be represented by general Formula (I):
- R 1 , R 2 and R 3 are each independently selected from hydrogen, electron- withdrawing moieties, and electron-donating moieties, provided that at least one of R 1 , R 2 and R 3 is an electron- withdrawing moiety; in another aspect, Ri is an electron-withdrawing moiety; in yet another aspect, Ri and R 3 are electron- withdrawing moieties; and b.) wherein X is an organic moiety having a molecular weight from about 65 daltons to about 4855 daltons, or from about 150 daltons to about 2855 daltons, or from about 193 daltons to about 1355 daltons, or from about 300 daltons to about 855 daltons, or from about 400 daltons to about 600 daltons, or from about 420 daltons to about 575 daltons.
- each R 1 , R 2 and R 3 may be independently selected from hydrogen, (C 1 -CzO-alkyl, (C 3 -C 1 o)-aryl, carboxylate, cyano, sulfonate, phosphonate, sulfate, acetate, nitro, (C 1 -CzO-alkyl ester, halogen or amino moiety, or each R 1 , R 2 and R 3 may be independently selected from hydrogen, nitro, cyano, (C 1 -CzO-alkyl ester or (C 1 -C 4 )-alkyl.
- the X may be a moiety having Formula (II) below:
- R 4 is selected from a moiety having Formula (III) below
- Each R 8 is independently selected from hydrogen, C 1 - Cs alkyl optionally substituted with a hydroxy, or acetyl;
- m is an integer from 0 to 10;
- Y is selected from a sulfonate, carboxylate, a phosphonate or quaternary ammonium species selected from an imidazolium, pyridinium, morpholinium, piperidinium, or a moiety having Formula (IV) below:
- R 9 is a C 1 -Cg alkyl moiety optionally substituted with -OH,
- Rio is selected from C 1 -C 18 alkyl moiety optionally substituted with -OH, or C 2 -Cg alkyl substituted with sulfonate, or C 1 -C 8 alkyl substituted with carboxylate,
- R 4 is selected from a moiety having Formula (V) below:
- Rn and Ri 2 is independently selected from hydrogen, C 1 -Cs alkyl, aryl, acetyl or hydroxyl moiety; m and n are independent and are integers from 0 to 10, ii.) Y is as described above;
- R 4 is selected from a moiety having Formula (VI) below:
- Rn is selected from an aryl moiety, arylalkyl moiety such as a benzyl moiety, C 1 -C 18 alkyl moiety, or a siloxane moiety;
- Each Ri 4 is independently selected from hydrogen, C 1 -
- R 5 can be the same as R 4 or selected from C 1 -C 12 alkyl moiety, aryl moiety or arylalkyl moiety such as a benzyl moiety; wherein the index a is an integer from 0 to 4, or from 0 to 3, or from 0 to 2, and each R 6 may be independently selected from a C 1 -C 6 alkyl, a C 1 -C 4 alkoxy, a nitro, a hydroxyl, a halogen, or -NHC(O)R 22 wherein R 22 is selected from H, -NH 2 , C 1 -C 6 alkyl, phenyl, -(CH 2 ) S OR 23 where the index s is 1 or 2 and R 23 is selected from Me, phenyl, and - CO 2 CH 2 CN; - NHSO 2 R 24 wherein R 24 is
- X is a moiety having Formula VII below:
- each R 4 and R 5 can independently be selected from:
- R' is selected from the group consisting of H, CH 3 , CH 2 O(CH 2 CH 2 O) z Ri 5 , and mixtures thereof;
- R" is selected from the group consisting of H, CH 2 O(CH 2 CH 2 O) Z Ri 5 , and mixtures thereof;
- z 0 to 10;
- each R i5 is independently selected from -H and -CH 2 CHRi 8 N + Ri 9 R 2 oR 2 i wherein Ri 8 is selected from -H and -CH 3 ;
- each Ri 9 and R 20 is independently selected from C 1 -C 4 alkyl optionally substituted with -OH;
- R 2 i is independently selected from C 1 -C 12 optionally -OH substituted alkyl or (CH 2 ) r O p Q;
- the index r is an integer from 1 to 8;
- the index p is 0 or 1; and
- Q is an anionic group selected from
- R 4 is selected from a moiety having Formula (III) below
- Rg is a hydrogen, C 1 -C 4 alkyl moiety or aryl moiety
- Y is a quaternary ammonium species selected from a group consisting of an imidazolium, or a moiety having
- R 9 is a C 1 -C 2 alkyl moiety
- Rio is selected from C 1 -C 8 alkyl moiety optionally substituted with -OH, or C 2 -C 4 alkyl substituted with sulfonate, or C 1 -C 4 alkyl substituted with carboxylate,
- R 4 is selected from a moiety having Formula (V) below:
- Rn and Ri 2 is independently selected from hydrogen, C 1 -C 4 alkyl or aryl moiety; m and n are independent and are integers from 0 to 5,
- R 4 is selected from a moiety having Formula (VI) below:
- Rn is selected from an aryl moiety, benzyl moiety, or a
- Each R 14 is independently selected from hydrogen or -
- m is an integer from 0 to 10,
- R 5 can be the same as R 4 or selected from C 1 -C 6 alkyl moiety or benzyl moiety;
- each R 6 may be independently selected from methyl, methoxy, or acetamido moiety.
- each R 1 , R 2 and R 3 may be independently selected from hydrogen, (C 1 -C 4 )-alkyl, (C 3 -C 1 o)-aryl, carboxylate, cyano, sulfonate, phosphonate, sulfate, acetate, nitro, (C 1 -C 4 )-alkyl ester, halogen or amino moiety, or each R 1 , R 2 and R 3 may be independently selected from hydrogen, nitro, cyano, (C 1 -C 4 )- alkyl ester or (C 1 -C 4 )-alkyl.
- the thiophene azo dye of the present invention may be represented by Formula (VIII):
- Formula VIII wherein the A moiety is selected from the group consisting of Table 1 A Moieties Nos. 1 - 118, or Table 1 A Moieties Nos. 6 - 11, 15, 21 - 23, 30 - 31, 33 - 39, 41, 43, 46 - 48, 50 - 55, 57 - 58, 64 - 65, 70 - 73, 77 - 78, 82 - 86, 88 - 90, 93 - 95, 99 - 100, 104 - 106, and 110 - 118, or Table 1 A Moieties Nos.
- the thiophene azo dye of the present invention may be represented by t tVhife f fnoililnowwiinngcr f foorrmmuullaa *:
- moiety A is selected from Table 1 A Moieties Nos. 1 - 118, or from Table 1 A Moieties Nos. 6 - 11, 15, 21 - 23, 30 - 31, 33 - 39, 41, 43, 46 - 48, 50 - 55, 57 - 58, 64 - 65, 70 - 73, 77 - 78, 82 - 86, 88 - 90, 93 - 95, 99 - 100, 104 - 106, and 110 - 118, or from Table 1 A Moieties Nos.
- R 6 is selected from Table 2 R 6 Substituent Identity and Position Nos. 1 - 40, or from Table 2 R 6 Substituent Identity and Position Nos. 1, 3, 5, 7 - 9, 11 - 14, 21, 23 - 24, 31, 33 - 34, 36 and 40, or from Table 2 R 6 Substituent Identity and Position Nos. 1, 3, 5, 7, 12, 13, 14, 31, 36 and 40; and R 4 and R 5 grouping is selected from Table 3 R 4 and R 5 Groupings Nos.
- a moieties may be selected from the moieties shown in Table 1 :
- Re may be selected from the substituents shown in Table 2:
- R 4 and R 5 groupings may be selected from the groupings shown in Table 3:
- X moieties may be selected from the moieties shown in Table 4:
- the thiophene azo dye having a formally charged moiety may comprise any one of the A moieties selected from Table 1, any one of the R 6 substituents selected from Table 2, any one of the R 4 and R 5 groupings selected from Table 3, and any one of the X moieties selected from Table 4.
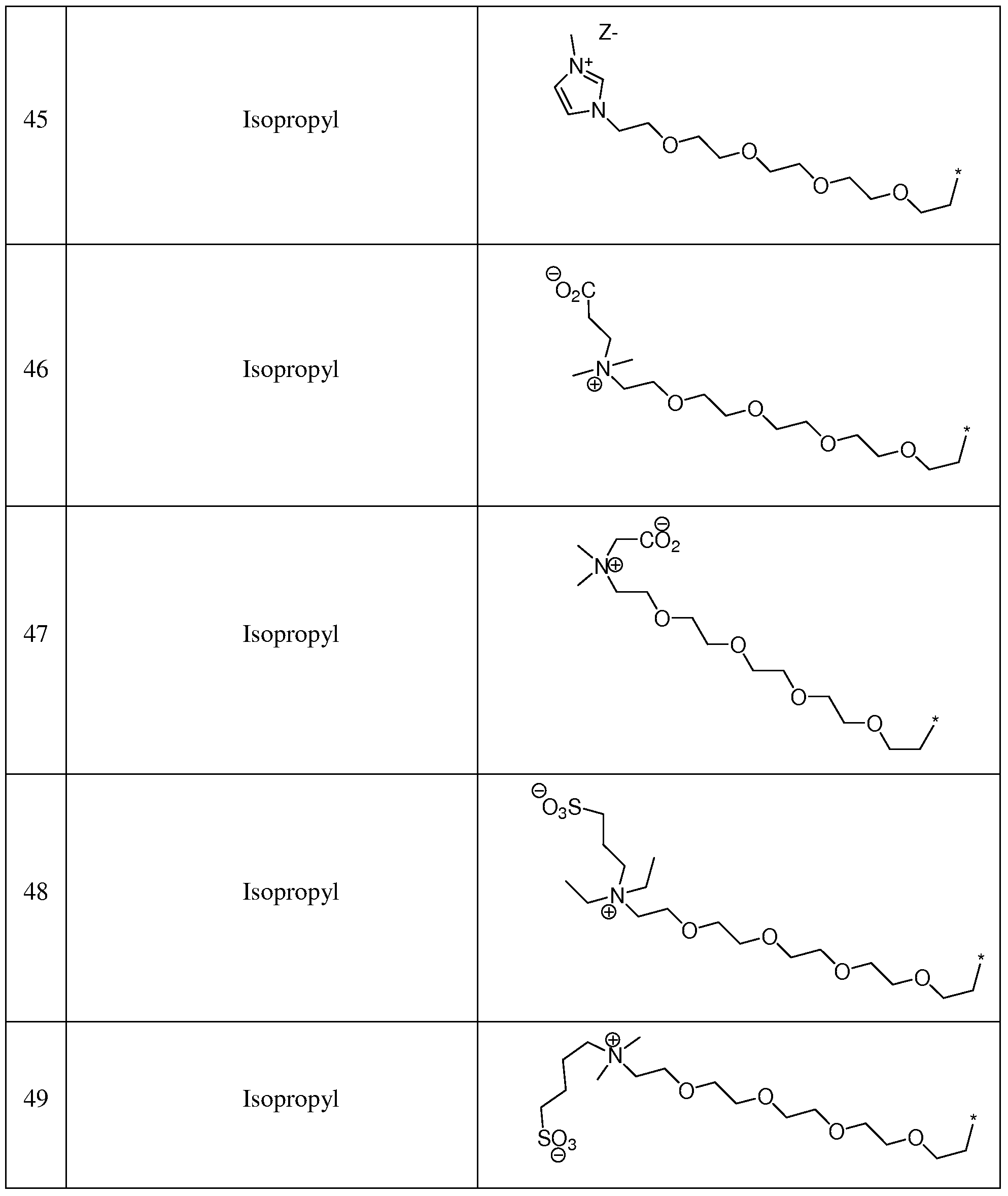
- suitable thiophene azo dyes include, but are not limited to, the structures shown in Table 5:
- hueing agents described in the present specification may be incorporated into laundry care compositions including but not limited to laundry detergents and fabric care compositions.
- the laundry care compositions including laundry detergents may be in solid or liquid form, including a gel form, and/or unit does forms, including multicompartment unit dose forms.
- Such compositions may comprise one or more of said hueing agents and a laundry care ingredient.
- said laundry care composition may comprise, based on total laundry care composition weight, less than 15% builder, less than 10% builder, or even less than 5% builder.
- said laundry care composition may comprise, based on total laundry care composition weight, a total of no more than 20% water; a total of no more than 15% water; a total of no more than 10% water; or even a total of no more than 5% water.
- said laundry care composition may comprise, rising, based on total laundry care composition weight, from about 10% to about 70% of a water-miscible organic solvent having a molecular weight of greater than 70 Daltons.
- said laundry care composition may comprise, based on total laundry care composition weight, a perfume microcapsule comprising a core and a shell that encapsulates said core, said perfume microcapsule having a D[4,3] average particle of from about 0.01 microns to about 200 microns and optionally a formaldehyde scavenger that is supplied via the addition of the microcapsules (contained in a perfume microcapsule slurry that is added to the laundry care ingredient) and/or added directly to the laundry care composition.
- the shell of said perfume microcapsules may be made of any material, including materials selected from the group consisting of polyethylenes, polyamides, polystyrenes, polyisoprenes, polycarbonates, polyesters, polyacrylates, polyureas, polyurethanes, polyolefins, polysaccharides, epoxy resins, vinyl polymers, and mixtures thereof.
- useful shell materials include materials that are sufficiently impervious to the core material and the materials in the environment in which the perfume microcapsule will be employed, to permit the delivery perfume to be obtained.
- Suitable impervious shell materials include materials selected from the group consisting of reaction products of one or more amines with one or more aldehydes, such as urea cross-linked with formaldehyde or gluteraldehyde, melamine cross-linked with formaldehyde; gelatin-polyphosphate coacervates optionally cross-linked with gluteraldehyde; gelatin-gum Arabic coacervates; cross-linked silicone fluids; polyamine reacted with polyisocyanates and mixtures thereof.
- the shell material comprises melamine cross-linked with formaldehyde and/or a polyacrylate.
- Suitable perfume microcapsules may be obtained from Appleton Papers of Appleton Wisconsin, USA.
- suitable formaldehyde scavengers include materials selected from the group consisting of sodium bisulfite, urea, ethylene urea, cysteine, cysteamine, lysine, glycine, serine, carnosine, histidine, glutathione, 3,4-diaminobenzoic acid, allantoin, glycouril, anthranilic acid, methyl anthranilate, methyl 4-aminobenzoate, ethyl acetoacetate, acetoacetamide, malonamide, ascorbic acid, 1,3-dihydroxyacetone dimer, biuret, oxamide, benzoguanamine, pyroglutamic acid, pyrogallol, methyl gallate, ethyl gallate, propyl gallate, Methanol amine, succinamide, thiabendazole, benzotriazol, triazole, indoline, sulfanilic acid, oxamide, sorbi
- Such formaldehyde scavengers are typically combined with a slurry containing said perfume microcapsules, at a level, based on total slurry weight, of from about 2 wt.% to about 18 wt.%, from about 3.5 wt.% to about 14 wt.% or even from about 5 wt.% to about 13 wt.%.
- such formaldehyde scavengers may be combined with a product containing a perfume microcapsule, said scavengers being combined with said product at a level, based on total product weight, of from about 0.005% to about 0.8%, alternatively from about 0.03% to about 0.5%, alternatively from about 0.065% to about 0.25% of the product formulation,.
- such formaldehyde scavengers may be combined with a slurry containing said perfume microcapsules, at a level, based on total slurry weight, of from about 2 wt.% to about 14 wt.%, from about 3.5 wt.% to about 14 wt.% or even from about 5 wt.% to about 14 wt.% and said slurry may be added to a product matrix to which addition an identical or different scavenger may be added at a level, based on total product weight, of from about 0.005% to about 0.5%, alternatively from about 0.01% to about 0.25%, alternatively from about 0.05% to about 0.15% of the product formulation,
- one or more of the aforementioned formaldehyde scavengers may be combined with a liquid fabric enhancing product containing perfume microcapsules at a level, based on total liquid fabric enhancing product weight, of from 0.005% to about 0.8%, alternatively from about 0.03% to about 0.4%, alternatively from about 0.06% to about 0.25% of the product formulation
- such formaldehyde scavengers may be combined with a liquid laundry detergent product containingperfume microcapsules, said scavengers being selected from the group consisting of sodium bisulfite, urea, ethylene urea, cysteine, cysteamine, lysine, glycine, serine, carnosine, histidine, glutathione, 3,4-diaminobenzoic acid, allantoin, glycouril, anthranilic acid, methyl anthranilate, methyl 4-aminobenzoate, ethyl acetoacetate,
- the hueing agents may be added to substrates using a variety of application techniques.
- the hueing agent may be included as a component of a laundry detergent.
- the hueing agent may be present in the laundry detergent composition in an amount from about 0.000001% to about 10% by weight of the composition, from about 0.00001% to about 10% by weight of the composition, from about 0.0001% to about 5% by weight of the composition, and even from about 0.0001% to about 1% by weight of the composition.
- the laundry detergent composition typically comprises a surfactant in an amount sufficient to provide desired cleaning properties.
- the laundry detergent composition may comprise, based on total laundry detergent composition weight, from about 0.5% to about 99% of the surfactant; from about 1% to about 95% of the surfactant; from about 5% to about 90% of the surfactant, from about 5% to about 70% of the surfactant, or even from about 5% to about 40% of the surfactant.
- the surfactant may comprise anionic, nonionic, cationic, zwitterionic and/or amphoteric surfactants.
- the detergent composition comprises anionic surfactant, nonionic surfactant, or mixtures thereof.
- Fabric care compositions are typically added in the rinse cycle, which is after the detergent solution has been used and replaced with the rinsing solution in typical laundering processes.
- the fabric care compositions disclosed herein may be comprise a rinse added fabric softening active and a suitable hueing agent as disclosed in the present specification.
- the fabric care composition may comprise, based on total fabric care composition weight, from about 1% to about 90%, or from about 5% to about 50% fabric softening active.
- the hueing agent may be present in the fabric care composition in an amount from about 0.5 ppb to about 50 ppm, or from about 0.5 ppm to about 30 ppm.
- laundry care ingredients While not essential for the purposes of the present invention, the non-limiting list of laundry care ingredients illustrated hereinafter are suitable for use in the laundry care compositions and may be desirably incorporated in certain aspects of the invention, for example to assist or enhance performance, for treatment of the substrate to be cleaned, or to modify the aesthetics of the composition as is the case with perfumes, colorants, dyes or the like. It is understood that such ingredients are in addition to the components that were previously listed for any particular aspect. The total amount of such adjuncts may range, once the amount of dye is taken into consideration from about 90% to about 99.99999995% by weight of the laundry care composition. The precise nature of these additional components, and levels of incorporation thereof, will depend on the physical form of the composition and the nature of the operation for which it is to be used.
- Suitable laundry care ingredients include, but are not limited to, fabric softening actives, polymers, for example cationic polymers, surfactants, builders, chelating agents, dye transfer inhibiting agents, dispersants, enzymes, and enzyme stabilizers, catalytic materials, bleach activators, polymeric dispersing agents, clay soil removal/anti-redeposition agents, brighteners, suds suppressors, dyes, perfume(s) including quadrant perfumes and additional perfume delivery systems including perfume loaded zeolites, starch encapsuled accords, and Schiff base pro-perfumes, structure elasticizing agents, fabric softeners, carriers, hydrotropes, processing aids and/or pigments.
- suitable examples of such other adjuncts and levels of use are found in U.S. Patent Nos. 5,576,282, 6,306,812 Bl and 6,326,348 Bl that are incorporated by reference.
- the laundry care ingredients are not essential to Applicants' laundry care compositions.
- certain aspects of Applicants' compositions do not contain one or more of the following adjuncts materials: fabric softening actives, bleach activators, surfactants, builders, chelating agents, dye transfer inhibiting agents, dispersants, enzymes, and enzyme stabilizers, catalytic metal complexes, polymeric dispersing agents, clay and soil removal/anti-redeposition agents, brighteners, suds suppressors, dyes, additional perfumes and perfume delivery systems, structure elasticizing agents, fabric softeners, carriers, hydrotropes, processing aids and/or pigments.
- one or more adjuncts may be present as detailed below:
- Suitable anionic surfactants useful herein can comprise any of the conventional anionic surfactant types typically used in liquid detergent products. These include the alkyl benzene sulfonic acids and their salts as well as alkoxylated or non-alkoxylated alkyl sulfate materials. Exemplary anionic surfactants are the alkali metal salts of C 1O -C 16 alkyl benzene sulfonic acids, or C 11 -C 14 alkyl benzene sulfonic acids. In one aspect, the alkyl group is linear and such linear alkyl benzene sulfonates are known as "LAS". Alkyl benzene sulfonates, and particularly LAS, are well known in the art.
- Such surfactants and their preparation are described for example in U.S. Pat. Nos. 2,220,099 and 2,477,383.
- Especially useful are the sodium and potassium linear straight chain alkylbenzene sulfonates in which the average number of carbon atoms in the alkyl group is from about 11 to 14.
- Sodium Cn-C 14 e.g., C 12
- LAS is a specific example of such surfactants.
- anionic surfactant comprises ethoxylated alkyl sulfate surfactants.
- Such materials also known as alkyl ether sulfates or alkyl polyethoxylate sulfates, are those which correspond to the formula: R'—O— (C 2 H 4 O) n -SO 3 M wherein R' is a Cg-C 2O alkyl group, n is from about 1 to 20, and M is a salt-forming cation.
- R' is C 1O -C 18 alkyl, n is from about 1 to 15, and M is sodium, potassium, ammonium, alkylammonium, or alkanolammonium.
- R' is a C 12 -C 16 , n is from about 1 to 6 and M is sodium.
- non-alkoxylated, e.g., non-ethoxylated, alkyl ether sulfate surfactants are those produced by the sulfation of higher C 8 -C 2 o fatty alcohols.
- Conventional primary alkyl sulfate surfactants have the general formula: ROSO 3 -M + wherein R is typically a linear Cg-C 2O hydrocarbyl group, which may be straight chain or branched chain, and M is a water-solubilizing cation.
- R is a C 1O -C 15 alkyl
- M is alkali metal, more specifically R is C 12 -C 14 and M is sodium.
- anionic surfactants useful herein include: a) C 11 -C 18 alkyl benzene sulfonates (LAS); b) C 10 -C 20 primary, branched-chain and random alkyl sulfates (AS); c) C 1O -C 18 secondary (2,3) alkyl sulfates having formulae (I) and (II): wherein M in formulae (I) and (II) is hydrogen or a cation which provides charge neutrality, and all M units, whether associated with a surfactant or adjunct ingredient, can either be a hydrogen atom or a cation depending upon the form isolated by the artisan or the relative pH of the system wherein the compound is used, with non-limiting examples of suitable cations including sodium, potassium, ammonium, and mixtures thereof, and x is an integer of at least about 7, or at least about 9, and y is an integer of at least 8, or at least about 9; d) C 1O -C 18 alkyl benzene
- MLAS modified alkylbenzene sulfonate
- MES methyl ester sulfonate
- AOS alpha-olefin sulfonate
- Suitable nonionic surfactants useful herein can comprise any of the conventional nonionic surfactant types typically used in liquid detergent products. These include alkoxylated fatty alcohols and amine oxide surfactants. In one aspect, for use in the liquid detergent products herein are those nonionic surfactants which are normally liquid.
- Suitable nonionic surfactants for use herein include the alcohol alkoxylate nonionic surfactants.
- Alcohol alkoxylates are materials which correspond to the general formula: RZ(C m H 21n O) n OH wherein R 1 is a Cs-C 16 alkyl group, m is from 2 to 4, and n ranges from about 2 to 12.
- R 1 is an alkyl group, which may be primary or secondary, that comprises from about 9 to 15 carbon atoms, or from about 10 to 14 carbon atoms.
- the alkoxylated fatty alcohols will also be ethoxylated materials that contain from about 2 to 12 ethylene oxide moieties per molecule, or from about 3 to 10 ethylene oxide moieties per molecule.
- alkoxylated fatty alcohol materials useful in the liquid detergent compositions herein will frequently have a hydrophilic-lipophilic balance (HLB) which ranges from about 3 to 17 from about 6 to 15, or from about 8 to 15.
- HLB hydrophilic-lipophilic balance
- Alkoxylated fatty alcohol nonionic surfactants have been marketed under the tradenames Neodol and Dobanol by the Shell Chemical Company.
- Nonionic surfactant useful herein comprises the amine oxide surfactants.
- Amine oxides are materials which are often referred to in the art as "semi-polar" nonionics. Amine oxides have the formula: R(EO) x (PO) y (BO) z N(O)(CH 2 R') 2 .qH 2 O.
- R is a relatively long-chain hydrocarbyl moiety which can be saturated or unsaturated, linear or branched, and can contain from 8 to 20, 10 to 16 carbon atoms, or is a C 12 -C 16 primary alkyl.
- R' is a short-chain moiety, in one aspect R' may be selected from hydrogen, methyl and -CH 2 OH. When x+y+z is different from 0, EO is ethyleneoxy, PO is propyleneneoxy and BO is butyleneoxy. Amine oxide surfactants are illustrated by C 12-14 alkyldimethyl amine oxide.
- Non-limiting examples of nonionic surfactants include: a) C 12 -C 18 alkyl ethoxylates, such as, NEODOL® nonionic surfactants from Shell; b) C 6 -C 12 alkyl phenol alkoxylates wherein the alkoxylate units are a mixture of ethyleneoxy and propyleneoxy units; c) C 12 -C 1S alcohol and C 6 -C 12 alkyl phenol condensates with ethylene oxide/propylene oxide block polymers such as Pluronic® from BASF; d) C 14 -C 22 mid-chain branched alcohols, BA, as discussed in U.S. Pat. No.
- the detersive surfactant component may comprise combinations of anionic and nonionic surfactant materials.
- the weight ratio of anionic to nonionic will typically range from 10:90 to 90:10, more typically from 30:70 to 70:30.
- Cationic surfactants are well known in the art and non-limiting examples of these include quaternary ammonium surfactants, which can have up to 26 carbon atoms. Additional examples include a) alkoxylate quaternary ammonium (AQA) surfactants as discussed in U.S. Pat. No. 6,136,769; b) dimethyl hydroxyethyl quaternary ammonium as discussed in U.S. Pat. No. 6,004,922; c) polyamine cationic surfactants as discussed in WO 98/35002, WO 98/35003, WO 98/35004, WO 98/35005, and WO 98/35006; d) cationic ester surfactants as discussed in U.S. Pat.
- AQA alkoxylate quaternary ammonium
- Non-limiting examples of zwitterionic surfactants include derivatives of secondary and tertiary amines, derivatives of heterocyclic secondary and tertiary amines, or derivatives of quaternary ammonium, quaternary phosphonium or tertiary sulfonium compounds. See U.S. Pat. No. 3,929,678 to Laughlin et al., issued Dec.
- betaine including alkyl dimethyl betaine and cocodimethyl amidopropyl betaine, Cg to C 18 (in one aspect C 12 to C 18 ) amine oxides and sulfo and hydroxy betaines, such as N-alkyl-N,N-dimethylammino-1- propane sulfonate where the alkyl group can be C 8 to C 18 , or C 1 O to C 14 .
- Non-limiting examples of ampholytic surfactants include aliphatic derivatives of secondary or tertiary amines, or aliphatic derivatives of heterocyclic secondary and tertiary amines in which the aliphatic radical can be straight- or branched-chain.
- One of the aliphatic substituents comprises at least about 8 carbon atoms, typically from about 8 to about 18 carbon atoms, and at least one comprises an anionic water-solubilizing group, e.g. carboxy, sulfonate, sulfate. See U.S. Pat. No. 3,929,678 to Laughlin et al., issued Dec. 30, 1975 at column 19, lines 18-35, for examples of ampholytic surfactants.
- Aqueous, non-surface active liquid carrier Aqueous, non-surface active liquid carrier
- the laundry care compositions may be in the form of a solid, either in tablet or particulate form, including, but not limited to particles, flakes, sheets, or the like, or the compositions may be in the form of a liquid.
- the liquid detergent compositions may comprise an aqueous, non-surface active liquid carrier.
- the amount of the aqueous, non-surface active liquid carrier employed in the compositions herein will be effective to solubilize, suspend or disperse the composition components.
- the liquid detergent compositions may comprise, based on total liquid detergent composition weight, from about 5% to about 90%, from about 10% to about 70%, or from about 20% to about 70% of the aqueous, non-surface active liquid carrier.
- aqueous, non-surface active liquid carrier is typically water. Accordingly, the aqueous, non-surface active liquid carrier component will generally be mostly, if not completely, comprised of water. While other types of water-miscible liquids, such alkanols, diols, other polyols, ethers, amines, and the like, have been conventionally been added to liquid detergent compositions as co-solvents or stabilizers, for purposes of the present invention, the utilization of such water-miscible liquids typically is minimized to hold down composition cost. Accordingly, the aqueous liquid carrier component of the liquid detergent products herein will generally comprise water present in concentrations ranging from about 5% to about 90%, or from about 5% to about 70%, by weight of the liquid detergent composition.
- the cleaning compositions of the present invention may comprise one or more bleaching agents. Suitable bleaching agents other than bleaching catalysts include photobleaches, bleach activators, hydrogen peroxide, sources of hydrogen peroxide, preformed peracids and mixtures thereof. In general, when a bleaching agent is used, the compositions of the present invention may comprise from about 0.1% to about 50% or even from about 0.1% to about 25% bleaching agent by weight of the subject cleaning
- bleaching agents include:
- photobleaches for example sulfonated zinc phthalocyanine
- Suitable preformed peracids include, but are not limited to, compounds selected from the group consisting of percarboxylic acids and salts, percarbonic acids and salts, perimidic acids and salts, peroxymonosulfuric acids and salts, for example, Oxzone ®, and mixtures thereof.
- inorganic perhydrate salts including alkali metal salts such as sodium salts of perborate (usually mono- or tetra-hydrate), percarbonate, persulphate, perphosphate, persilicate salts and mixtures thereof.
- the inorganic perhydrate salts are selected from the group consisting of sodium salts of perborate, percarbonate and mixtures thereof.
- inorganic perhydrate salts are typically present in amounts of from 0.05 to 40 wt%, or 1 to 30 wt% of the overall composition and are typically incorporated into such compositions as a crystalline solid that may be coated. Suitable coatings include, inorganic salts such as alkali metal silicate, carbonate or borate salts or mixtures thereof, or organic materials such as water- soluble or dispersible polymers, waxes, oils or fatty soaps; and
- suitable leaving groups are benzoic acid and derivatives thereof - especially benzene sulphonate.
- Suitable bleach activators include dodecanoyl oxybenzene sulphonate, decanoyl oxybenzene sulphonate, decanoyl oxybenzoic acid or salts thereof, 3,5,5-trimethyl hexanoyloxybenzene sulphonate, tetraacetyl ethylene diamine (TAED) and nonanoyloxybenzene sulphonate (NOBS).
- TAED tetraacetyl ethylene diamine
- NOBS nonanoyloxybenzene sulphonate
- Suitable bleach activators are also disclosed in WO 98/17767. While any suitable bleach activator may be employed, in one aspect of the invention the subject cleaning composition may comprise NOBS, TAED or mixtures thereof.
- the peracid and/or bleach activator is generally present in the composition in an amount of from about 0.1 to about 60 wt%, from about 0.5 to about 40 wt % or even from about 0.6 to about 10 wt% based on the composition.
- One or more hydrophobic peracids or precursors thereof may be used in combination with one or more hydrophilic peracid or precursor thereof.
- the amounts of hydrogen peroxide source and peracid or bleach activator may be selected such that the molar ratio of available oxygen (from the peroxide source) to peracid is from 1:1 to 35:1, or even 2:1 to 10:1.
- Bleach Boosting Compounds may comprise one or more bleach boosting compounds.
- Bleach boosting compounds provide increased bleaching effectiveness in lower temperature applications.
- the bleach boosters act in conjunction with conventional peroxygen bleaching sources to provide increased bleaching effectiveness. This is normally accomplished through in situ formation of an active oxygen transfer agent such as a dioxirane, an oxaziridine, or an oxaziridinium. Alternatively, preformed dioxiranes, oxaziridines and oxaziridiniums may be used.
- Suitable bleach boosting compounds for use in accordance with the present invention are cationic imines, zwitterionic imines, anionic imines and/or polyionic imines having a net charge of from about +3 to about -3, and mixtures thereof.
- These imine bleach boosting compounds of the present invention include those of the general structure: where RI - R ⁇ may be a hydrogen or an unsubstituted or substituted radical selected from the group consisting of phenyl, aryl, heterocyclic ring, alkyl and cycloalkyl radicals.
- Suitable bleach boosting compounds include zwitterionic bleach boosters zwitterionic bleach boosters, which are described in U.S. Patent Nos. 5,576,282 and 5,718,614.
- Other bleach boosting compounds include cationic bleach boosters described in U.S. Patent Nos. 5,360,569; 5,442,066; 5,478,357; 5,370,826; 5,482,515; 5,550,256; and WO 95/13351, WO 95/13352, and WO 95/13353.
- Peroxygen sources are well-known in the art and the peroxygen source employed in the present invention may comprise any of these well known sources, including peroxygen compounds as well as compounds, which under consumer use conditions, provide an effective amount of peroxygen in situ.
- the peroxygen source may include a hydrogen peroxide source, the in situ formation of a peracid anion through the reaction of a hydrogen peroxide source and a bleach activator, preformed peracid compounds or mixtures of suitable peroxygen sources.
- the bleach boosting compounds when present, are typically employed in conjunction with a peroxygen source in the bleaching systems of the present invention.
- Enzyme Bleaching - Enzymatic systems may be used as bleaching agents.
- the hydrogen peroxide may also be present by adding an enzymatic system (i.e. an enzyme and a substrate therefore) which is capable of generating hydrogen peroxide at the beginning or during the washing and/or rinsing process.
- an enzymatic system i.e. an enzyme and a substrate therefore
- Such enzymatic systems are disclosed in EP Patent Application 91202655.6 filed October 9, 1991.
- the present invention compositions and methods may utilize alternative bleach systems such as ozone, chlorine dioxide and the like.
- Bleaching with ozone may be accomplished by introducing ozone-containing gas having ozone content from about 20 to about 300 g/m 3 into the solution that is to contact the fabrics. The gas:liquid ratio in the solution should be maintained from about 1:2.5 to about 1:6.
- U.S. Patent No. 5,346, 588 describes a process for the utilization of ozone as an alternative to conventional bleach systems and is herein incorporated
- the fabric softening active is a quaternary ammonium compound suitable for softening fabric in a rinse step.
- the FSA is formed from a reaction product of a fatty acid and an aminoalcohol obtaining mixtures of mono-, di-, and, in one aspect, triester compounds.
- the FSA comprises one or more softener quaternary ammonium compounds such, but not limited to, as a monoalkyquaternary ammonium compound, a diamido quaternary compound and a diester quaternary ammonium compound, or a combination thereof.
- the FSA comprises a diester quaternary ammonium (hereinafter "DQA") compound composition.
- DQA compounds compositions also encompasses a description of diamido FSAs and FSAs with mixed amido and ester linkages as well as the aforementioned diester linkages, all herein referred to as DQA.
- DQA (I) DQA (I)
- FSA FSA in the present CFSC
- each R substituent is either hydrogen, a short chain C ⁇ -Cg, for example C ⁇ - C3 alkyl or hydroxyalkyl group, e.g., methyl, ethyl, propyl, hydroxyethyl, and the like, poly (C2-3 alkoxy), for example, polyethoxy, group, benzyl, or mixtures thereof; each m is 2 or 3; each n is from 1 to about 4, or 2; each Y is -0-(O)C-, -C(O)-O-, -NR-C(O)-, or -C(O)-NR- and it is acceptable for each Y to be the same or different; the sum of carbons in each R.1, plus one when Y is -0-(O)C- or -NR-C(O) -, is Q ⁇ - ⁇ -22, or C14-C20 with each RI being a hydrocarbyl, or substituted hydrocarbyl group; it is acceptable for R ⁇ to be unsatur
- Suitable DQA compounds are typically made by reacting alkanolamines such as MDEA (methyldiethanolamine) and TEA (triethanolamine) with fatty acids.
- alkanolamines such as MDEA (methyldiethanolamine) and TEA (triethanolamine)
- Some materials that typically result from such reactions include N,N-di(acyl-oxyethyl)-N,N-dimethylammonium chloride or N,N-di(acyl-oxyethyl)-N,N-methylhydroxyethylammonium methylsulfate
- the acyl group is derived from animal fats, unsaturated, and polyunsaturated, fatty acids, e.g., tallow, hardended tallow, oleic acid, and/or partially hydrogenated fatty acids, derived from vegetable oils and/or partially hydrogenated vegetable oils, such as, canola oil, safflower oil, peanut oil, sunflower oil, corn oil, soybean oil, tall oil, rice
- the FSA comprises other actives in addition to DQA (1) or DQA.
- the FSA comprises only DQA (1) or DQA and is free or essentially free of any other quaternary ammonium compounds or other actives.
- the FSA comprises the precursor amine that is used to produce the DQA.
- the FSA comprises a compound, identified as DTTMAC comprising the formula:
- each RI is a Cg-C22 > or C14-C20 but no more than one being less than about C ⁇ 2 an d then the other is at least about 16, hydrocarbyl, or substituted hydrocarbyl substituent, for example, C10-C20 alkyl or alkenyl (unsaturated alkyl, including polyunsaturated alkyl, also referred to sometimes as "alkylene"), in one aspect C] ⁇ Ci 8 alkyl or alkenyl, and branch or unbranched.
- the Iodine Value (IV) of the FSA is from about 1 to 70; each R is H or a short chain C ⁇ -Cg, or C1-C3 alkyl or hydroxyalkyl group, e.g., methyl, ethyl, propyl, hydroxyethyl, and the like, benzyl, or (R.2 O)2-4.H where each R2 is a C ⁇ .g alkylene group; and A " is a softener compatible anion, suitable anions include chloride, bromide, methylsulfate, ethylsulfate, sulfate, phosphate, or nitrate; in one aspect the anions are chloride or methyl sulfate.
- FSAs include dialkydimethylammonium salts and dialkylenedimethylammonium salts such as ditallowdimethylammonium and ditallowdimethylammonium methylsulfate.
- dialkylenedimethylammonium salts such as ditallowdimethylammonium and ditallowdimethylammonium methylsulfate.
- dialkylenedimethylammonium salts usable in the present invention are di-hydrogenated tallow dimethyl ammonium chloride and ditallowdimethyl ammonium chloride available from Degussa under the trade names Adogen® 442 and Adogen® 470 respectively.
- the FSA comprises other actives in addition to DTTMAC.
- the FSA comprises only compounds of the DTTMAC and is free or essentially free of any other quaternary ammonium compounds or other actives.
- the FSA comprises an FSA described in U.S. Pat. Pub. No. 2004/0204337 Al, published Oct. 14, 2004 to Corona et al., from paragraphs 30 - 79.
- the FSA is one described in U.S. Pat. Pub. No. 2004/0229769 Al, published Nov. 18, 2005, to Smith et al., on paragraphs 26 - 31; or U.S. Pat. No. 6,494,920, at column 1, line 51 et seq. detailing an "esterquat" or a quaternized fatty acid triethanolamine ester salt.
- the FSA is chosen from at least one of the following: ditallowoyloxyethyl dimethyl ammonium chloride, dihydrogenated-tallowoyloxyethyl dimethyl ammonium chloride, ditallow dimethyl ammonium chloride, ditallowoyloxyethyl dimethyl ammonium methyl sulfate, dihydrogenated-tallowoyloxyethyl dimethyl ammonium chloride, dihydrogenated-tallowoyloxyethyl dimethyl ammonium chloride, or combinations thereof.
- the FSA may also include amide containing compound compositions.
- diamide comprising compounds may include but not limited to methyl- bis(tallowamidoethyl)-2-hydroxyethylammonium methyl sulfate (available from Degussa under the trade names Varisoft 110 and Varisoft 222).
- An example of an amide-ester containing compound is N-[3-(stearoylamino)propyl]-N-[2-(stearoyloxy)ethoxy)ethyl)]-N- methylamine.
- Another aspect of the invention provides for a rinse added fabric softening composition further comprising a cationic starch.
- Cationic starches are disclosed in US 2004/0204337 Al.
- the rinse added fabric softening composition comprises from about 0.1% to about 7% of cationic starch by weight of the fabric softening composition.
- the cationic starch is HCP401 from National Starch.
- compositions of the present invention can comprise one or more detergent builders or builder systems. When present, the compositions will typically comprise at least about 1% builder, or from about 5% or 10% to about 80%, 50%, or even 30% by weight, of said builder.
- Builders include, but are not limited to, the alkali metal, ammonium and alkanolammonium salts of polyphosphates, alkali metal silicates, alkaline earth and alkali metal carbonates, aluminosilicate builders polycarboxylate compounds.
- ether hydroxypolycarboxylates copolymers of maleic anhydride with ethylene or vinyl methyl ether, 1,3,5-trihydroxybenzene-2,4,6-trisulphonic acid, and carboxymethyl-oxysuccinic acid
- the various alkali metal, ammonium and substituted ammonium salts of polyacetic acids such as ethylenediamine tetraacetic acid and nitrilotriacetic acid
- polycarboxylates such as mellitic acid, succinic acid, oxydisuccinic acid, polymaleic acid, benzene 1,3,5- tricarboxylic acid, carboxymethyloxysuccinic acid, and soluble salts thereof.
- compositions herein may also optionally contain one or more copper, iron and/or manganese chelating agents. If utilized, chelating agents will generally comprise from about 0.1% by weight of the compositions herein to about 15%, or even from about 3.0% to about 15% by weight of the compositions herein.
- compositions of the present invention may also include one or more dye transfer inhibiting agents.
- Suitable polymeric dye transfer inhibiting agents include, but are not limited to, polyvinylpyrrolidone polymers, polyamine N-oxide polymers, copolymers of N-vinylpyrrolidone and N-vinylimidazole, polyvinyloxazolidones and polyvinylimidazoles or mixtures thereof.
- the dye transfer inhibiting agents are present at levels from about 0.0001%, from about 0.01%, from about 0.05% by weight of the cleaning compositions to about 10%, about 2%, or even about 1% by weight of the cleaning compositions.
- compositions of the present invention can also contain dispersants.
- Suitable water-soluble organic materials are the homo- or co-polymeric acids or their salts, in which the polycarboxylic acid may comprise at least two carboxyl radicals separated from each other by not more than two carbon atoms.
- Enzymes - The compositions can comprise one or more detergent enzymes which provide cleaning performance and/or fabric care benefits.
- suitable enzymes include, but are not limited to, hemicellulases, peroxidases, proteases, cellulases, xylanases, lipases, phospholipases, esterases, cutinases, pectinases, keratanases, reductases, oxidases, phenoloxidases, lipoxygenases, ligninases, pullulanases, tannases, pentosanases, malanases, ⁇ -glucanases, arabinosidases, hyaluronidase, chondroitinase, laccase, and amylases, or mixtures thereof.
- a typical combination is a cocktail of conventional applicable enzymes like protease, lipase, cutinase and/or cellulase in conjunction with amylase.
- Enzyme Stabilizers - Enzymes for use in compositions for example, detergents can be stabilized by various techniques.
- the enzymes employed herein can be stabilized by the presence of water-soluble sources of calcium and/or magnesium ions in the finished compositions that provide such ions to the enzymes.
- laundry care compositions of the present invention can be formulated into any suitable form and prepared by any process chosen by the formulator, non-limiting examples of which are described in Applicants' examples and in U.S. 5,879,584; U.S. 5,691,297; U.S. 5,574,005; U.S. 5,569,645; U.S. 5,565,422; U.S. 5,516,448; U.S. 5,489,392; U.S. 5,486,303 all of which are incorporated herein by reference.
- the liquid detergent compositions may be in the form of an aqueous solution or uniform dispersion or suspension of surfactant, hueing agent, and certain optional other ingredients, some of which may normally be in solid form, that have been combined with the normally liquid components of the composition, such as the liquid alcohol ethoxylate nonionic, the aqueous liquid carrier, and any other normally liquid optional ingredients.
- a solution, dispersion or suspension will be acceptably phase stable and will typically have a viscosity which ranges from about 100 to 600 cps, or from about 150 to 400 cps. For purposes of this invention, viscosity is measured with a Brookfield LVDV-II+ viscometer apparatus using a #21 spindle.
- the liquid detergent compositions herein can be prepared by combining the components thereof in any convenient order and by mixing, e.g., agitating, the resulting component combination to form a phase stable liquid detergent composition.
- a liquid matrix is formed containing at least a major proportion, or even substantially all, of the liquid components, e.g., nonionic surfactant, the non-surface active liquid carriers and other optional liquid components, with the liquid components being thoroughly admixed by imparting shear agitation to this liquid combination.
- the liquid components e.g., nonionic surfactant, the non-surface active liquid carriers and other optional liquid components
- shear agitation for example, rapid stirring with a mechanical stirrer may usefully be employed. While shear agitation is maintained, substantially all of any anionic surfactants and the solid form ingredients can be added.
- Agitation of the mixture is continued, and if necessary, can be increased at this point to form a solution or a uniform dispersion of insoluble solid phase particulates within the liquid phase.
- particles of any enzyme material to be included e.g., enzyme prills, are incorporated.
- one or more of the solid components may be added to the agitated mixture as a solution or slurry of particles premixed with a minor portion of one or more of the liquid components.
- agitation of the mixture is continued for a period of time sufficient to form compositions having the requisite viscosity and phase stability characteristics. Frequently this will involve agitation for a period of from about 30 to 60 minutes.
- the hueing agent is first combined with one or more liquid components to form a hueing agent premix, and this hueing agent premix is added to a composition formulation containing a substantial portion, for example more than 50% by weight, more specifically, more than 70% by weight, and yet more specifically, more than 90% by weight, of the balance of components of the laundry detergent composition.
- a composition formulation containing a substantial portion for example more than 50% by weight, more specifically, more than 70% by weight, and yet more specifically, more than 90% by weight, of the balance of components of the laundry detergent composition.
- both the hueing agent premix and the enzyme component are added at a final stage of component additions.
- the hueing agent is encapsulated prior to addition to the detergent composition, the encapsulated hueing agent is suspended in a structured liquid, and the suspension is added to a composition formulation containing a substantial portion of the balance of components of the laundry detergent composition.
- the detergent compositions may be in a solid form. Suitable solid forms include tablets and particulate forms, for example, granular particles, flakes or sheets. Various techniques for forming detergent compositions in such solid forms are well known in the art and may be used herein.
- the hueing agent is provided in particulate form, optionally including additional but not all components of the laundry detergent composition.
- the hueing agent particulate is combined with one or more additional particulates containing a balance of components of the laundry detergent composition.
- the hueing agent, optionally including additional but not all components of the laundry detergent composition may be provided in an encapsulated form, and the hueing agent encapsulate is combined with particulates containing a substantial balance of components of the laundry detergent composition.
- compositions of this invention can be used to form aqueous washing solutions for use in the laundering of fabrics.
- an effective amount of such compositions is added to water, for example in a conventional fabric laundering automatic washing machine, to form such aqueous laundering solutions.
- the aqueous washing solution so formed is then contacted, typically under agitation, with the fabrics to be laundered therewith.
- An effective amount of the liquid detergent compositions herein added to water to form aqueous laundering solutions can comprise amounts sufficient to form from about 500 to 7,000 ppm of composition in aqueous washing solution, or from about 1,000 to 3,000 ppm of the detergent compositions herein will be provided in aqueous washing solution.
- Certain of the consumer products disclosed herein can be used to clean or treat a situs inter alia a surface or fabric.
- a situs is contacted with an embodiment of Applicants' consumer product, in neat form or diluted in a liquor, for example, a wash liquor and then the situs may be optionally washed and/or rinsed.
- a situs is optionally washed and/or rinsed, contacted with an aspect of the consumer product and then optionally washed and/or rinsed.
- washing includes but is not limited to, scrubbing, and mechanical agitation.
- the fabric may comprise most any fabric capable of being laundered or treated in normal consumer use conditions.
- Liquors that may comprise the disclosed compositions may have a pH of from about 3 to about 11.5. Such compositions are typically employed at concentrations of from about 500 ppm to about 15,000 ppm in solution.
- the wash solvent is water
- the water temperature typically ranges from about 5 °C to about 90 °C and, when the situs comprises a fabric, the water to fabric ratio is typically from about 1:1 to about 30:1.
- Employing one or more of the aforementioned methods results in a treated situs.
- method of treating and/or cleaning a surface or fabric comprising the steps of optionally washing and/or rinsing said surface or fabric, contacting said surface or fabric with any laundry care composition disclosed in this specification, then optionally washing and/or rinsing said surface and/or fabric then optionally letting said surface or fabric to dry and/or actively drying said surface or fabric, is disclosed.
- the prepared diazonium salt solution was slowly added into the above solution for coupling reaction. Care was taken to not allow the temperature to rise above 10°C. After complete addition of diazonium salt solution, the reaction was allowed to slowly reach room temperature over an hour. The mixture was then neutralized with sodium hydroxide and phase separated. The product layer was then dissolved with methanol and filtered to remove any excess salts. The filtrate was evaporated and the product of this reaction can be used at this point or further diluted with water to a lower viscosity.
- Example 1 was prepared via the Intermediate Type 2 procedures, wherein only 2 moles of ethylene oxide were added to the initial material and the tosylated material was quaternized by using 2 molar equivalents N-methyl imidazole. Colorant synthesis was as described in Example 12.
- Example 2 was prepared as Example 1, except the initial alkoxylation was done using m- toluidine.
- Example 7 was prepared via the Intermediate Type 1 procedures, wherein only 1 mole of ethylene oxide was added to the N-ethyl- aniline and the tosylated material was quaternized by using 1 molar equivalent triethylamine. Colorant synthesis was as described in Example 12.
- Example 13
- Example 13 was prepared as Example 12, except quaternization was done with N-methyl imidazole.
- Example 14 was prepared as Example 12, except quaternization was done with N 5 N- Dimethyl-glycine.
- Example 15 was prepared as Example 12, except quaternization was done with N 5 N- Dimethylpropyl sulfonate.
- Example 18 was prepared via the Intermediate Type 2 procedures and colorant synthesis was as described in Example 12.
- Example 19 was prepared via the Intermediate Type 2 procedures, wherein the initial alkoxylation was done using 2,5-dimethoxyaniline and colorant synthesis was as described in Example 12.
- Example 21 was prepared via the Intermediate Type 2 procedures, wherein quaternization was done using dimethylethanolamine and colorant synthesis was as described in Example 12.
- Example 22 was prepared via the Intermediate Type 2 procedures, wherein quaternization was done using triethanolamine and colorant synthesis was as described in Example 12.
- Example 35 was prepared via the Intermediate Type 2 procedures, wherein alkoxylation was done using m-toluidine and quaterinzation was done using triethylamine. Colorant synthesis was as described in Example 12.
- Example 36 was prepared as Example 1, except that the initial alkoxylation was done using m-toluidine and quaternization was done using triethylamine. Color synthesis was as described in Example 12.
- Aqueous Partition Value (APV) (A f ZA 1 ) x 100% wherein A 1 is the initial solution absorbance at ⁇ max and A f is the final solution absorbance at
- the hueing efficiency, DE* eff of the dye is assessed by determining the L*, a*, and b* value measurements of each swatch using a Hunter LabScan XE reflectance spectrophotometer with D65 illumination, 10° observer and UV filter excluded. The hueing efficiency of the dye is then calculated using the following equation:
- DE* eff ((L* c - I ⁇ ) 2 + (a* c - a* s ) 2 + (b* c - b* s ) 2 ) 1/2 wherein the subscripts c and s respectively refer to the L*, a*, and b* values measured for the control, i.e., the fabric sample washed in detergent with no dye, and the fabric sample washed in detergent containing the dye to be screened.
- Formulations Ia - 11 Liquid Detergent Formulations Tables 7A and 7B provide examples of liquid detergent formulations which include at least one thiophene azo dye of the present invention as a hueing agent. The formulations are shown in Table 7 A as Formulations Ia through If and in Table 7B as Formulations Ig through 11.
- Table 8 provides examples of granular detergent formulations which include at least one thiophene azo dye of the present invention as a hueing agent.
- the formulations are shown in Table 8 as Formulations 2a through 2e.
- Hhiophene azo hueing agent from Table 5, Examples 1-21, preferably with hueing efficiency
- Formulations 3a - 3d Liquid Fabric Care Compositions
- Table 9 provides examples of liquid fabric care compositions which include at least one thiophene azo dye of the present invention as a hueing agent.
- the compositions are shown in Table 9 as Formulations 3a through 3d.
- Cationic starch based on common maize starch or potato starch, containing 25% to 95% amylose and a degree of substitution of from 0.02 to 0.09, and having a viscosity measured as Water Fluidity having a value from 50 to 84.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Dispersion Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112013011851A BR112013011851A2 (en) | 2010-11-12 | 2010-11-12 | "laundry care composition comprising azothiophene dyes and method for treating and / or cleaning a surface or fabric" |
| CN201080070050.7A CN103210073B (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dye and the laundry care composition comprising them |
| MX2013005276A MX2013005276A (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dyes and laundry care compositions containing the same. |
| CA2817718A CA2817718C (en) | 2010-11-12 | 2010-11-12 | Laundry care compositions comprising charged thiophene azo dyes |
| EP10781555.7A EP2638142B1 (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dyes and laundry care compositions containing the same |
| JP2013538698A JP5833133B2 (en) | 2010-11-12 | 2010-11-12 | Thiopheneazo dye and laundry care composition containing the same |
| PCT/US2010/056508 WO2011011799A2 (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dyes and laundry care compositions containing the same |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/US2010/056508 WO2011011799A2 (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dyes and laundry care compositions containing the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2011011799A2 true WO2011011799A2 (en) | 2011-01-27 |
| WO2011011799A3 WO2011011799A3 (en) | 2011-10-06 |
Family
ID=43499689
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/056508 WO2011011799A2 (en) | 2010-11-12 | 2010-11-12 | Thiophene azo dyes and laundry care compositions containing the same |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP2638142B1 (en) |
| JP (1) | JP5833133B2 (en) |
| CN (1) | CN103210073B (en) |
| BR (1) | BR112013011851A2 (en) |
| CA (1) | CA2817718C (en) |
| MX (1) | MX2013005276A (en) |
| WO (1) | WO2011011799A2 (en) |
Cited By (144)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05260462A (en) * | 1991-08-21 | 1993-10-08 | Sgs Thomson Microelectron Sa | Predictor |
| WO2012159778A1 (en) | 2011-05-26 | 2012-11-29 | Unilever Plc | Liquid laundry composition |
| WO2012166768A1 (en) * | 2011-06-03 | 2012-12-06 | The Procter & Gamble Company | Laundry care compositions containing dyes |
| WO2012163871A1 (en) * | 2011-06-01 | 2012-12-06 | Unilever Plc | Liquid detergent composition containing dye polymer |
| WO2012166699A1 (en) * | 2011-06-03 | 2012-12-06 | The Procter & Gamble Company | Thiophene azo carboxylate dyes and laundry care compositions containing the same |
| EP2540825A2 (en) | 2011-06-30 | 2013-01-02 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants reference to a sequence listing |
| WO2013011071A1 (en) | 2011-07-21 | 2013-01-24 | Unilever Plc | Liquid laundry composition |
| EP2551335A1 (en) | 2011-07-25 | 2013-01-30 | The Procter & Gamble Company | Enzyme stabilized liquid detergent composition |
| WO2013025742A1 (en) | 2011-08-15 | 2013-02-21 | The Procter & Gamble Company | Detergent compositions containing pyridinol-n-oxide compounds |
| WO2013006871A3 (en) * | 2012-02-13 | 2013-03-14 | Milliken & Company | Laundry care compositions containing dyes |
| CN102972395A (en) * | 2012-12-06 | 2013-03-20 | 扬州大学 | 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing lygocoris lucorum and method for preparing 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing and treating lygocoris lucorum |
| WO2013043805A1 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising primary surfactant systems comprising highly branched surfactants especially isoprenoid - based surfactants |
| WO2013043803A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising specific blend ratios of isoprenoid-based surfactants |
| WO2013043852A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Easy-rinse detergent compositions comprising isoprenoid-based surfactants |
| WO2013043855A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | High suds detergent compositions comprising isoprenoid-based surfactants |
| WO2013043857A1 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising sustainable surfactant systems comprising isoprenoid-derived surfactants |
| CN103230766A (en) * | 2013-05-08 | 2013-08-07 | 天津城市建设学院 | Preparation and application of self-adhesive double-wall essence microcapsule |
| WO2013189640A1 (en) | 2012-06-21 | 2013-12-27 | Unilever Plc | Liquid laundry detergent |
| WO2014012921A1 (en) | 2012-07-17 | 2014-01-23 | Unilever Plc | Bright detergent composition |
| WO2014012923A1 (en) | 2012-07-17 | 2014-01-23 | Unilever Plc | Bright detergent composition |
| WO2014018309A1 (en) | 2012-07-26 | 2014-01-30 | The Procter & Gamble Company | Low ph liquid cleaning compositions with enzymes |
| EP2712915A1 (en) | 2012-10-01 | 2014-04-02 | The Procter and Gamble Company | Methods of treating a surface and compositions for use therein |
| WO2014066309A1 (en) | 2012-10-24 | 2014-05-01 | The Procter & Gamble Company | Anti foam compositions comprising partly phenyl bearing polyorganosilicons |
| WO2014066308A1 (en) | 2012-10-24 | 2014-05-01 | The Procter & Gamble Company | Anti foam compositions comprising aryl bearing polyorganosilicons |
| US8715368B2 (en) | 2010-11-12 | 2014-05-06 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| EP2740785A1 (en) | 2012-12-06 | 2014-06-11 | The Procter and Gamble Company | Use of composition to reduce weeping and migration through a water soluble film |
| WO2014089386A1 (en) | 2012-12-06 | 2014-06-12 | The Procter & Gamble Company | Soluble pouch comprising hueing dye |
| EP2767582A1 (en) | 2013-02-19 | 2014-08-20 | The Procter and Gamble Company | Method of laundering a fabric |
| EP2767581A1 (en) | 2013-02-19 | 2014-08-20 | The Procter & Gamble Company | Method of laundering a fabric |
| EP2767579A1 (en) | 2013-02-19 | 2014-08-20 | The Procter and Gamble Company | Method of laundering a fabric |
| WO2014160821A1 (en) | 2013-03-28 | 2014-10-02 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine, a soil release polymer, and a carboxymethylcellulose |
| WO2014168776A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Hydroxyl polymer fiber structures comprising ammonium alkylsulfonate salts and methods for making same |
| WO2014168942A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Fibrous structures comprising polysaccharide filaments |
| WO2014168775A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Fibrous structures exhibiting improved whiteness index values |
| EP2808372A1 (en) | 2013-05-28 | 2014-12-03 | The Procter and Gamble Company | Surface treatment compositions comprising photochromic dyes |
| JP2015500888A (en) * | 2011-06-03 | 2015-01-08 | ミリケン・アンド・カンパニーMilliken & Company | Thiophene azocarboxylate dyes and laundry care compositions containing them |
| EP2828340A1 (en) * | 2012-03-19 | 2015-01-28 | The Procter & Gamble Company | Laundry care compositions containing dyes |
| EP2832842A1 (en) | 2013-07-30 | 2015-02-04 | The Procter & Gamble Company | Method of making granular detergent compositions comprising surfactants |
| EP2832843A1 (en) | 2013-07-30 | 2015-02-04 | The Procter & Gamble Company | Method of making granular detergent compositions comprising polymers |
| WO2015041887A3 (en) * | 2013-09-18 | 2015-05-14 | Milliken & Company | Laundry care composition comprising carboxylate dye |
| EP2899260A1 (en) | 2014-01-22 | 2015-07-29 | Unilever PLC | Process to manufacture a liquid detergent formulation |
| WO2015112341A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015112338A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| WO2015112339A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015112340A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| WO2015126547A1 (en) | 2014-02-19 | 2015-08-27 | Milliken & Company | Composition comprising benefit agent and aprotic solvent |
| WO2015127004A1 (en) | 2014-02-19 | 2015-08-27 | The Procter & Gamble Company | Composition comprising benefit agent and aprotic solvent |
| WO2015130669A1 (en) | 2014-02-25 | 2015-09-03 | The Procter & Gamble Company | A process for making renewable surfactant intermediates and surfactants from fats and oils and products thereof |
| WO2015130653A1 (en) | 2014-02-25 | 2015-09-03 | The Procter & Gamble Company | A process for making renewable surfactant intermediates and surfactants from fats and oils and products thereof |
| WO2015148360A1 (en) | 2014-03-27 | 2015-10-01 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2015148361A1 (en) | 2014-03-27 | 2015-10-01 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2015168194A1 (en) | 2014-04-29 | 2015-11-05 | The Procter & Gamble Company | Fabric care compositions comprising polyurethane, polyurea and/or polyurethaneurea polymers |
| WO2015187757A1 (en) | 2014-06-06 | 2015-12-10 | The Procter & Gamble Company | Detergent composition comprising polyalkyleneimine polymers |
| EP2987848A1 (en) | 2014-08-19 | 2016-02-24 | The Procter & Gamble Company | Method of laundering a fabric |
| EP2987849A1 (en) | 2014-08-19 | 2016-02-24 | The Procter and Gamble Company | Method of Laundering a Fabric |
| WO2016032991A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016032992A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016032995A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Method of treating a fabric |
| WO2016032993A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016044200A1 (en) | 2014-09-15 | 2016-03-24 | The Procter & Gamble Company | Detergent compositions containing salts of polyetheramines and polymeric acid |
| WO2016049387A1 (en) | 2014-09-26 | 2016-03-31 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2016049388A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Fabric care compositions containing a polyetheramine |
| WO2016048969A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Detergent compositions containing a polyetheramine and an anionic soil release polymer |
| WO2016048674A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2016081437A1 (en) | 2014-11-17 | 2016-05-26 | The Procter & Gamble Company | Benefit agent delivery compositions |
| JP2016530392A (en) * | 2013-09-18 | 2016-09-29 | ザ プロクター アンド ギャンブル カンパニー | Laundry care composition containing a thiophene azocarboxylate dye |
| EP3088503A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| EP3088502A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| EP3088505A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| EP3088506A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Detergent composition |
| EP3088504A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| JP2016536433A (en) * | 2013-09-18 | 2016-11-24 | ザ プロクター アンド ギャンブル カンパニー | Laundry care composition comprising a carboxylate dye |
| WO2016188693A1 (en) | 2015-05-27 | 2016-12-01 | Unilever Plc | Laundry detergent composition |
| WO2016192905A1 (en) | 2015-06-02 | 2016-12-08 | Unilever Plc | Laundry detergent composition |
| JP2016538350A (en) * | 2013-09-23 | 2016-12-08 | ザ プロクター アンド ギャンブル カンパニー | particle |
| WO2016200440A1 (en) | 2015-06-11 | 2016-12-15 | The Procter & Gamble Company | Device and methods for applying compositions to surfaces |
| EP2992054B1 (en) | 2013-05-03 | 2017-01-04 | Unilever PLC | Alkoxylated bis azo dyes |
| WO2017055205A1 (en) | 2015-10-01 | 2017-04-06 | Unilever Plc | Powder laundry detergent composition |
| EP3173467A1 (en) | 2015-11-26 | 2017-05-31 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2017140391A1 (en) | 2016-02-17 | 2017-08-24 | Unilever Plc | Whitening composition |
| WO2017140392A1 (en) | 2016-02-17 | 2017-08-24 | Unilever Plc | Whitening composition |
| WO2017198438A1 (en) | 2016-05-17 | 2017-11-23 | Unilever Plc | Liquid laundry detergent compositions |
| WO2017198574A1 (en) | 2016-05-17 | 2017-11-23 | Unilever Plc | Liquid laundry detergent compositions |
| EP3301162A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| EP3301164A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| EP3301163A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| WO2018060139A1 (en) | 2016-09-27 | 2018-04-05 | Unilever Plc | Domestic laundering method |
| WO2018072979A1 (en) | 2016-10-18 | 2018-04-26 | Unilever Plc | Whitening composition |
| EP3357994A1 (en) | 2017-02-01 | 2018-08-08 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants |
| WO2019008036A1 (en) | 2017-07-07 | 2019-01-10 | Unilever Plc | Whitening composition |
| WO2019008035A1 (en) | 2017-07-07 | 2019-01-10 | Unilever Plc | Laundry cleaning composition |
| WO2019105675A1 (en) | 2017-11-30 | 2019-06-06 | Unilever Plc | Detergent composition comprising protease |
| WO2019162135A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Process of preparing a solid composition comprising aminopolycarboxylate |
| WO2019192813A1 (en) | 2018-04-03 | 2019-10-10 | Unilever N.V. | Dye granule |
| WO2019219531A1 (en) | 2018-05-17 | 2019-11-21 | Unilever Plc | Cleaning composition |
| WO2019219302A1 (en) | 2018-05-17 | 2019-11-21 | Unilever Plc | Cleaning composition comprising rhamnolipid and alkyl ether carboxylate surfactants |
| WO2020016097A1 (en) | 2018-07-17 | 2020-01-23 | Unilever Plc | Use of a rhamnolipid in a surfactant system |
| WO2020058024A1 (en) | 2018-09-17 | 2020-03-26 | Unilever Plc | Detergent composition |
| WO2020104158A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104156A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104157A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104159A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104155A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| US10676703B2 (en) | 2016-10-03 | 2020-06-09 | The Procter & Gamble Company | Process for preparing a spray-dried laundry detergent particle |
| WO2020186030A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2020186028A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2020186052A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Method for treating cotton |
| EP3750978A1 (en) | 2019-06-12 | 2020-12-16 | Unilever N.V. | Laundry detergent composition |
| EP3750979A1 (en) | 2019-06-12 | 2020-12-16 | Unilever N.V. | Use of laundry detergent composition |
| WO2020259947A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020259948A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020259949A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020260040A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020260038A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020264552A1 (en) | 2019-06-24 | 2020-12-30 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants |
| WO2020260006A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent compositions |
| WO2021032833A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021043764A1 (en) | 2019-09-02 | 2021-03-11 | Unilever Global Ip Limited | Detergent composition |
| WO2021069516A1 (en) | 2019-10-07 | 2021-04-15 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021133701A1 (en) | 2019-12-23 | 2021-07-01 | The Procter & Gamble Company | Compositions comprising enzymes |
| WO2021185956A1 (en) | 2020-03-19 | 2021-09-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021185870A1 (en) | 2020-03-19 | 2021-09-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021249927A1 (en) | 2020-06-08 | 2021-12-16 | Unilever Ip Holdings B.V. | Method of improving protease activity |
| WO2022023250A1 (en) | 2020-07-27 | 2022-02-03 | Unilever Ip Holdings B.V. | Use of an enzyme and surfactant for inhibiting microorganisms |
| WO2022043042A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022043045A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022042977A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022042989A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Surfactant and detergent composition |
| WO2022043138A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Surfactant and detergent composition |
| WO2022094163A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning composition comprising alginate lyase enzymes |
| WO2022128786A1 (en) | 2020-12-17 | 2022-06-23 | Unilever Ip Holdings B.V. | Use and cleaning composition |
| WO2022128781A1 (en) | 2020-12-17 | 2022-06-23 | Unilever Ip Holdings B.V. | Cleaning composition |
| EP4060010A2 (en) | 2021-03-15 | 2022-09-21 | The Procter & Gamble Company | Cleaning compositions containing polypeptide variants |
| WO2022235720A1 (en) | 2021-05-05 | 2022-11-10 | The Procter & Gamble Company | Methods for making cleaning compositions and detecting soils |
| EP4108767A1 (en) | 2021-06-22 | 2022-12-28 | The Procter & Gamble Company | Cleaning or treatment compositions containing nuclease enzymes |
| WO2022268657A1 (en) | 2021-06-24 | 2022-12-29 | Unilever Ip Holdings B.V. | Unit dose cleaning composition |
| EP4112707A1 (en) | 2021-06-30 | 2023-01-04 | The Procter & Gamble Company | Fabric treatment |
| WO2023017794A1 (en) | 2021-08-10 | 2023-02-16 | 株式会社日本触媒 | Polyalkylene-oxide-containing compound |
| WO2023041694A1 (en) | 2021-09-20 | 2023-03-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2023067075A1 (en) | 2021-10-21 | 2023-04-27 | Unilever Ip Holdings B.V. | Detergent compositions |
| WO2023144071A1 (en) | 2022-01-28 | 2023-08-03 | Unilever Ip Holdings B.V. | Laundry composition |
| EP4273210A1 (en) | 2022-05-04 | 2023-11-08 | The Procter & Gamble Company | Detergent compositions containing enzymes |
| US12031113B2 (en) | 2020-03-02 | 2024-07-09 | Milliken & Company | Composition comprising hueing agent |
| EP4410941A1 (en) | 2023-02-01 | 2024-08-07 | The Procter & Gamble Company | Detergent compositions containing enzymes |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3047007B1 (en) * | 2013-09-18 | 2018-04-11 | The Procter and Gamble Company | Laundry care composition comprising carboxylate dye |
| US10155868B2 (en) * | 2015-10-13 | 2018-12-18 | Milliken & Company | Whitening agents for cellulosic substrates |
| US11351106B2 (en) * | 2020-09-14 | 2022-06-07 | Milliken & Company | Oxidative hair cream composition containing thiophene azo colorant |
| US11344492B2 (en) | 2020-09-14 | 2022-05-31 | Milliken & Company | Oxidative hair cream composition containing polymeric colorant |
Citations (61)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2220099A (en) | 1934-01-10 | 1940-11-05 | Gen Aniline & Flim Corp | Sulphonic acids |
| US2477383A (en) | 1946-12-26 | 1949-07-26 | California Research Corp | Sulfonated detergent and its method of preparation |
| US3929678A (en) | 1974-08-01 | 1975-12-30 | Procter & Gamble | Detergent composition having enhanced particulate soil removal performance |
| US4228042A (en) | 1978-06-26 | 1980-10-14 | The Procter & Gamble Company | Biodegradable cationic surface-active agents containing ester or amide and polyalkoxy group |
| US4239660A (en) | 1978-12-13 | 1980-12-16 | The Procter & Gamble Company | Detergent composition comprising a hydrolyzable cationic surfactant and specific alkalinity source |
| US4260529A (en) | 1978-06-26 | 1981-04-07 | The Procter & Gamble Company | Detergent composition consisting essentially of biodegradable nonionic surfactant and cationic surfactant containing ester or amide |
| US4483780A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions containing polyglycoside and polyethoxylate detergent surfactants |
| US4483779A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions comprising polyglycoside and polyethoxylate surfactants and anionic fluorescer |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| WO1992006162A1 (en) | 1990-09-28 | 1992-04-16 | The Procter & Gamble Company | Detergent containing alkyl sulfate and polyhydroxy fatty acid amide surfactants |
| WO1993019038A1 (en) | 1992-03-26 | 1993-09-30 | The Procter & Gamble Company | Process for reducing the levels of fatty acid contaminants in polyhydroxy fatty acid amide surfactants |
| WO1993019146A1 (en) | 1992-03-16 | 1993-09-30 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| WO1994009099A1 (en) | 1992-10-13 | 1994-04-28 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| US5332528A (en) | 1990-09-28 | 1994-07-26 | The Procter & Gamble Company | Polyhydroxy fatty acid amides in soil release agent-containing detergent compositions |
| US5346588A (en) | 1989-10-30 | 1994-09-13 | Lenzing Aktiengesellschaft | Process for the chlorine-free bleaching of cellulosic materials with ozone |
| US5360569A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Activation of bleach precursors with catalytic imine quaternary salts |
| US5370826A (en) | 1993-11-12 | 1994-12-06 | Lever Brothers Company, Division Of Conopco, Inc. | Quaternay oxaziridinium salts as bleaching compounds |
| WO1995013352A1 (en) | 1993-11-12 | 1995-05-18 | Unilever N.V. | Imine quaternary salts as bleach catalysts |
| WO1995013353A1 (en) | 1993-11-12 | 1995-05-18 | Unilever N.V. | Activation of bleach precursors with imine quaternary salts |
| US5482515A (en) | 1993-11-12 | 1996-01-09 | Lever Brothers Company, Division Of Conopco, Inc. | Imine quaternary salts as bleach catalysts |
| US5486303A (en) | 1993-08-27 | 1996-01-23 | The Procter & Gamble Company | Process for making high density detergent agglomerates using an anhydrous powder additive |
| US5489392A (en) | 1994-09-20 | 1996-02-06 | The Procter & Gamble Company | Process for making a high density detergent composition in a single mixer/densifier with selected recycle streams for improved agglomerate properties |
| US5516448A (en) | 1994-09-20 | 1996-05-14 | The Procter & Gamble Company | Process for making a high density detergent composition which includes selected recycle streams for improved agglomerate |
| US5565422A (en) | 1995-06-23 | 1996-10-15 | The Procter & Gamble Company | Process for preparing a free-flowing particulate detergent composition having improved solubility |
| US5569645A (en) | 1995-04-24 | 1996-10-29 | The Procter & Gamble Company | Low dosage detergent composition containing optimum proportions of agglomerates and spray dried granules for improved flow properties |
| US5574005A (en) | 1995-03-07 | 1996-11-12 | The Procter & Gamble Company | Process for producing detergent agglomerates from high active surfactant pastes having non-linear viscoelastic properties |
| US5576282A (en) | 1995-09-11 | 1996-11-19 | The Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US5691297A (en) | 1994-09-20 | 1997-11-25 | The Procter & Gamble Company | Process for making a high density detergent composition by controlling agglomeration within a dispersion index |
| US5718614A (en) | 1995-08-28 | 1998-02-17 | Electro-Matic Products Company | Control apparatus for grinder |
| WO1998017767A1 (en) | 1996-10-18 | 1998-04-30 | The Procter & Gamble Company | Detergent compositions |
| US5759990A (en) | 1996-10-21 | 1998-06-02 | The Procter & Gamble Company | Concentrated fabric softening composition with good freeze/thaw recovery and highly unsaturated fabric softener compound therefor |
| WO1998035003A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Detergent compound |
| WO1998035006A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Liquid cleaning composition |
| WO1998035004A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Solid detergent compositions |
| WO1998035002A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Cleaning compositions |
| WO1998035005A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | A cleaning composition |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999005084A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Process for making alkylbenzenesulfonate surfactants from alcohols and products thereof |
| WO1999005241A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Cleaning products comprising improved alkylarylsulfonate surfactants prepared via vinylidene olefins and processes for preparation thereof |
| WO1999005082A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved processes for making alkylbenzenesulfonate surfactants and products thereof |
| WO1999007656A2 (en) | 1997-08-08 | 1999-02-18 | The Procter & Gamble Company | Improved processes for making surfactants via adsorptive separation and products thereof |
| US5879584A (en) | 1994-09-10 | 1999-03-09 | The Procter & Gamble Company | Process for manufacturing aqueous compositions comprising peracids |
| US6004922A (en) | 1996-05-03 | 1999-12-21 | The Procter & Gamble Company | Laundry detergent compositions comprising cationic surfactants and modified polyamine soil dispersents |
| US6008181A (en) | 1996-04-16 | 1999-12-28 | The Procter & Gamble Company | Mid-Chain branched Alkoxylated Sulfate Surfactants |
| US6020303A (en) | 1996-04-16 | 2000-02-01 | The Procter & Gamble Company | Mid-chain branched surfactants |
| US6022844A (en) | 1996-03-05 | 2000-02-08 | The Procter & Gamble Company | Cationic detergent compounds |
| WO2000023549A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| WO2000023548A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| US6060443A (en) | 1996-04-16 | 2000-05-09 | The Procter & Gamble Company | Mid-chain branched alkyl sulfate surfactants |
| US6093856A (en) | 1996-11-26 | 2000-07-25 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| WO2000047708A1 (en) | 1999-02-10 | 2000-08-17 | The Procter & Gamble Company | Low density particulate solids useful in laundry detergents |
| US6136769A (en) | 1996-05-17 | 2000-10-24 | The Procter & Gamble Company | Alkoxylated cationic detergency ingredients |
| US6150322A (en) | 1998-08-12 | 2000-11-21 | Shell Oil Company | Highly branched primary alcohol compositions and biodegradable detergents made therefrom |
| US6221825B1 (en) | 1996-12-31 | 2001-04-24 | The Procter & Gamble Company | Thickened, highly aqueous liquid detergent compositions |
| WO2001042408A2 (en) | 1999-12-08 | 2001-06-14 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| US6482994B2 (en) | 1997-08-02 | 2002-11-19 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| US6494920B1 (en) | 1999-02-04 | 2002-12-17 | Cognis Deutschland Gmbh & Co. Kg | Detergent mixtures |
| US20040204337A1 (en) | 2003-03-25 | 2004-10-14 | The Procter & Gamble Company | Fabric care compositions comprising cationic starch |
| US20040229769A1 (en) | 2000-12-27 | 2004-11-18 | Colgate-Palmolive Company | Thickened fabric conditioners |
Family Cites Families (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1389266A (en) * | 1972-05-18 | 1975-04-03 | Ici Ltd | Basic monoazo dyestuffs |
| US4459229A (en) * | 1981-10-09 | 1984-07-10 | Eastman Kodak Company | Heterocyclic azo dyes with couplers having sulfoaralkyl groups |
| JPS59170147A (en) * | 1983-03-18 | 1984-09-26 | Mitsubishi Chem Ind Ltd | Basic disazo dye |
| DE3528759A1 (en) * | 1985-08-10 | 1987-02-19 | Basf Ag | HETEROCYCLIC AZO DYES |
| DE3707715A1 (en) * | 1987-03-11 | 1988-09-22 | Basf Ag | DYES WITH N- (2-AMINOETHYL) PIPERAZINE GROUPS |
| DE4215495A1 (en) * | 1992-05-12 | 1993-11-18 | Basf Ag | Azo dyes with an acetalized diazo component from the thiophene or thiazole series |
| CN101370925B (en) * | 2006-01-23 | 2014-03-26 | 美利肯公司 | Laundry care compositions with thiazolium dye |
| PL2192169T3 (en) * | 2007-01-19 | 2012-10-31 | Procter & Gamble | Laundry care composition comprising a whitening agents for cellulosic substrates |
| CN101910394B (en) * | 2008-01-10 | 2012-06-27 | 荷兰联合利华有限公司 | Shading composition |
| ATE539141T1 (en) * | 2008-06-13 | 2012-01-15 | Procter & Gamble | MULTI-CHAMBER BAGS |
| EP2163608A1 (en) * | 2008-09-12 | 2010-03-17 | The Procter & Gamble Company | Laundry particle made by extrusion comprising a hueing dye and fatty acid soap |
| ES2385762T3 (en) * | 2008-09-30 | 2012-07-31 | The Procter & Gamble Company | Composition comprising microcapsules |
| JP2010209294A (en) * | 2009-03-12 | 2010-09-24 | Lion Corp | Detergent composition |
| EP2638113B1 (en) * | 2010-11-12 | 2017-01-04 | Milliken & Company | Thiophene azo dyes and laundry care compositions containing the same |
-
2010
- 2010-11-12 WO PCT/US2010/056508 patent/WO2011011799A2/en active Application Filing
- 2010-11-12 EP EP10781555.7A patent/EP2638142B1/en active Active
- 2010-11-12 MX MX2013005276A patent/MX2013005276A/en unknown
- 2010-11-12 CN CN201080070050.7A patent/CN103210073B/en not_active Expired - Fee Related
- 2010-11-12 JP JP2013538698A patent/JP5833133B2/en not_active Expired - Fee Related
- 2010-11-12 BR BR112013011851A patent/BR112013011851A2/en not_active IP Right Cessation
- 2010-11-12 CA CA2817718A patent/CA2817718C/en not_active Expired - Fee Related
Patent Citations (67)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2220099A (en) | 1934-01-10 | 1940-11-05 | Gen Aniline & Flim Corp | Sulphonic acids |
| US2477383A (en) | 1946-12-26 | 1949-07-26 | California Research Corp | Sulfonated detergent and its method of preparation |
| US3929678A (en) | 1974-08-01 | 1975-12-30 | Procter & Gamble | Detergent composition having enhanced particulate soil removal performance |
| US4228042A (en) | 1978-06-26 | 1980-10-14 | The Procter & Gamble Company | Biodegradable cationic surface-active agents containing ester or amide and polyalkoxy group |
| US4260529A (en) | 1978-06-26 | 1981-04-07 | The Procter & Gamble Company | Detergent composition consisting essentially of biodegradable nonionic surfactant and cationic surfactant containing ester or amide |
| US4239660A (en) | 1978-12-13 | 1980-12-16 | The Procter & Gamble Company | Detergent composition comprising a hydrolyzable cationic surfactant and specific alkalinity source |
| US4565647A (en) | 1982-04-26 | 1986-01-21 | The Procter & Gamble Company | Foaming surfactant compositions |
| US4483779A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions comprising polyglycoside and polyethoxylate surfactants and anionic fluorescer |
| US4483780A (en) | 1982-04-26 | 1984-11-20 | The Procter & Gamble Company | Detergent compositions containing polyglycoside and polyethoxylate detergent surfactants |
| US4565647B1 (en) | 1982-04-26 | 1994-04-05 | Procter & Gamble | Foaming surfactant compositions |
| US5346588A (en) | 1989-10-30 | 1994-09-13 | Lenzing Aktiengesellschaft | Process for the chlorine-free bleaching of cellulosic materials with ozone |
| WO1992006162A1 (en) | 1990-09-28 | 1992-04-16 | The Procter & Gamble Company | Detergent containing alkyl sulfate and polyhydroxy fatty acid amide surfactants |
| US5332528A (en) | 1990-09-28 | 1994-07-26 | The Procter & Gamble Company | Polyhydroxy fatty acid amides in soil release agent-containing detergent compositions |
| WO1993019146A1 (en) | 1992-03-16 | 1993-09-30 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| WO1993019038A1 (en) | 1992-03-26 | 1993-09-30 | The Procter & Gamble Company | Process for reducing the levels of fatty acid contaminants in polyhydroxy fatty acid amide surfactants |
| WO1994009099A1 (en) | 1992-10-13 | 1994-04-28 | The Procter & Gamble Company | Fluid compositions containing polyhydroxy fatty acid amides |
| US5486303A (en) | 1993-08-27 | 1996-01-23 | The Procter & Gamble Company | Process for making high density detergent agglomerates using an anhydrous powder additive |
| US5550256A (en) | 1993-11-12 | 1996-08-27 | Lever Brothers Company, Division Of Conopco, Inc. | Imine quaternary salts as bleach catalysts |
| WO1995013351A1 (en) | 1993-11-12 | 1995-05-18 | Unilever N.V. | Quarternary oxaziridinium salts as bleaching compounds |
| WO1995013352A1 (en) | 1993-11-12 | 1995-05-18 | Unilever N.V. | Imine quaternary salts as bleach catalysts |
| WO1995013353A1 (en) | 1993-11-12 | 1995-05-18 | Unilever N.V. | Activation of bleach precursors with imine quaternary salts |
| US5442066A (en) | 1993-11-12 | 1995-08-15 | Lever Brothers Company, Division Of Conopco, Inc. | Quaternary oxaziridinium salts as bleaching compounds |
| US5478357A (en) | 1993-11-12 | 1995-12-26 | Lever Brothers Company, Division Of Conopco, Inc. | Activation of bleach precursors with imine quaternary salts |
| US5482515A (en) | 1993-11-12 | 1996-01-09 | Lever Brothers Company, Division Of Conopco, Inc. | Imine quaternary salts as bleach catalysts |
| US5360569A (en) | 1993-11-12 | 1994-11-01 | Lever Brothers Company, Division Of Conopco, Inc. | Activation of bleach precursors with catalytic imine quaternary salts |
| US5370826A (en) | 1993-11-12 | 1994-12-06 | Lever Brothers Company, Division Of Conopco, Inc. | Quaternay oxaziridinium salts as bleaching compounds |
| US5879584A (en) | 1994-09-10 | 1999-03-09 | The Procter & Gamble Company | Process for manufacturing aqueous compositions comprising peracids |
| US5489392A (en) | 1994-09-20 | 1996-02-06 | The Procter & Gamble Company | Process for making a high density detergent composition in a single mixer/densifier with selected recycle streams for improved agglomerate properties |
| US5516448A (en) | 1994-09-20 | 1996-05-14 | The Procter & Gamble Company | Process for making a high density detergent composition which includes selected recycle streams for improved agglomerate |
| US5691297A (en) | 1994-09-20 | 1997-11-25 | The Procter & Gamble Company | Process for making a high density detergent composition by controlling agglomeration within a dispersion index |
| US5574005A (en) | 1995-03-07 | 1996-11-12 | The Procter & Gamble Company | Process for producing detergent agglomerates from high active surfactant pastes having non-linear viscoelastic properties |
| US5569645A (en) | 1995-04-24 | 1996-10-29 | The Procter & Gamble Company | Low dosage detergent composition containing optimum proportions of agglomerates and spray dried granules for improved flow properties |
| US5565422A (en) | 1995-06-23 | 1996-10-15 | The Procter & Gamble Company | Process for preparing a free-flowing particulate detergent composition having improved solubility |
| US5718614A (en) | 1995-08-28 | 1998-02-17 | Electro-Matic Products Company | Control apparatus for grinder |
| US5576282A (en) | 1995-09-11 | 1996-11-19 | The Procter & Gamble Company | Color-safe bleach boosters, compositions and laundry methods employing same |
| US6022844A (en) | 1996-03-05 | 2000-02-08 | The Procter & Gamble Company | Cationic detergent compounds |
| US6060443A (en) | 1996-04-16 | 2000-05-09 | The Procter & Gamble Company | Mid-chain branched alkyl sulfate surfactants |
| US6020303A (en) | 1996-04-16 | 2000-02-01 | The Procter & Gamble Company | Mid-chain branched surfactants |
| US6008181A (en) | 1996-04-16 | 1999-12-28 | The Procter & Gamble Company | Mid-Chain branched Alkoxylated Sulfate Surfactants |
| US6004922A (en) | 1996-05-03 | 1999-12-21 | The Procter & Gamble Company | Laundry detergent compositions comprising cationic surfactants and modified polyamine soil dispersents |
| US6136769A (en) | 1996-05-17 | 2000-10-24 | The Procter & Gamble Company | Alkoxylated cationic detergency ingredients |
| WO1998017767A1 (en) | 1996-10-18 | 1998-04-30 | The Procter & Gamble Company | Detergent compositions |
| US5759990A (en) | 1996-10-21 | 1998-06-02 | The Procter & Gamble Company | Concentrated fabric softening composition with good freeze/thaw recovery and highly unsaturated fabric softener compound therefor |
| US6153577A (en) | 1996-11-26 | 2000-11-28 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| US6093856A (en) | 1996-11-26 | 2000-07-25 | The Procter & Gamble Company | Polyoxyalkylene surfactants |
| US6221825B1 (en) | 1996-12-31 | 2001-04-24 | The Procter & Gamble Company | Thickened, highly aqueous liquid detergent compositions |
| WO1998035006A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Liquid cleaning composition |
| WO1998035003A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Detergent compound |
| WO1998035005A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | A cleaning composition |
| WO1998035002A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Cleaning compositions |
| WO1998035004A1 (en) | 1997-02-11 | 1998-08-13 | The Procter & Gamble Company | Solid detergent compositions |
| WO1999005241A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Cleaning products comprising improved alkylarylsulfonate surfactants prepared via vinylidene olefins and processes for preparation thereof |
| WO1999005082A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved processes for making alkylbenzenesulfonate surfactants and products thereof |
| WO1999005243A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Detergent compositions containing mixtures of crystallinity-disrupted surfactants |
| WO1999005084A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Process for making alkylbenzenesulfonate surfactants from alcohols and products thereof |
| WO1999005242A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkylbenzenesulfonate surfactants |
| WO1999005244A1 (en) | 1997-07-21 | 1999-02-04 | The Procter & Gamble Company | Improved alkyl aryl sulfonate surfactants |
| US6482994B2 (en) | 1997-08-02 | 2002-11-19 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| WO1999007656A2 (en) | 1997-08-08 | 1999-02-18 | The Procter & Gamble Company | Improved processes for making surfactants via adsorptive separation and products thereof |
| US6150322A (en) | 1998-08-12 | 2000-11-21 | Shell Oil Company | Highly branched primary alcohol compositions and biodegradable detergents made therefrom |
| WO2000023548A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| WO2000023549A1 (en) | 1998-10-20 | 2000-04-27 | The Procter & Gamble Company | Laundry detergents comprising modified alkylbenzene sulfonates |
| US6494920B1 (en) | 1999-02-04 | 2002-12-17 | Cognis Deutschland Gmbh & Co. Kg | Detergent mixtures |
| WO2000047708A1 (en) | 1999-02-10 | 2000-08-17 | The Procter & Gamble Company | Low density particulate solids useful in laundry detergents |
| WO2001042408A2 (en) | 1999-12-08 | 2001-06-14 | The Procter & Gamble Company | Ether-capped poly(oxyalkylated) alcohol surfactants |
| US20040229769A1 (en) | 2000-12-27 | 2004-11-18 | Colgate-Palmolive Company | Thickened fabric conditioners |
| US20040204337A1 (en) | 2003-03-25 | 2004-10-14 | The Procter & Gamble Company | Fabric care compositions comprising cationic starch |
Cited By (233)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05260462A (en) * | 1991-08-21 | 1993-10-08 | Sgs Thomson Microelectron Sa | Predictor |
| US9487740B2 (en) | 2010-11-12 | 2016-11-08 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| US8979946B2 (en) | 2010-11-12 | 2015-03-17 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| US10655091B2 (en) | 2010-11-12 | 2020-05-19 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| US10435651B2 (en) | 2010-11-12 | 2019-10-08 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| US8715368B2 (en) | 2010-11-12 | 2014-05-06 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| US9856439B2 (en) | 2010-11-12 | 2018-01-02 | The Procter & Gamble Company | Thiophene azo dyes and laundry care compositions containing the same |
| WO2012159778A1 (en) | 2011-05-26 | 2012-11-29 | Unilever Plc | Liquid laundry composition |
| WO2012163871A1 (en) * | 2011-06-01 | 2012-12-06 | Unilever Plc | Liquid detergent composition containing dye polymer |
| EP4134424A1 (en) | 2011-06-01 | 2023-02-15 | Unilever IP Holdings B.V. | Liquid detergent composition containing dye polymer |
| EP3354792A1 (en) | 2011-06-01 | 2018-08-01 | Unilever PLC, a company registered in England and Wales under company no. 41424 of | Liquid detergent composition containing dye polymer |
| EP2714985B1 (en) | 2011-06-01 | 2018-02-28 | Unilever PLC | Liquid detergent composition containing dye polymer |
| RU2598853C2 (en) * | 2011-06-03 | 2016-09-27 | Дзе Проктер Энд Гэмбл Компани | Composition for washing clothes containing dyes |
| CN103582696A (en) * | 2011-06-03 | 2014-02-12 | 宝洁公司 | Laundry care compositions containing dyes |
| US8888865B2 (en) | 2011-06-03 | 2014-11-18 | The Procter & Gamble Company | Thiophene azo carboxylate dyes and laundry care compositions containing the same |
| JP2015500888A (en) * | 2011-06-03 | 2015-01-08 | ミリケン・アンド・カンパニーMilliken & Company | Thiophene azocarboxylate dyes and laundry care compositions containing them |
| WO2012166768A1 (en) * | 2011-06-03 | 2012-12-06 | The Procter & Gamble Company | Laundry care compositions containing dyes |
| JP2014520177A (en) * | 2011-06-03 | 2014-08-21 | ザ プロクター アンド ギャンブル カンパニー | Thiophene azocarboxylate dye and laundry care composition containing the same |
| WO2012166699A1 (en) * | 2011-06-03 | 2012-12-06 | The Procter & Gamble Company | Thiophene azo carboxylate dyes and laundry care compositions containing the same |
| US9752104B2 (en) | 2011-06-03 | 2017-09-05 | The Procter & Gamble Company | Thiophene AZO carboxylate dyes and laundry care compositions containing the same |
| CN103703117A (en) * | 2011-06-03 | 2014-04-02 | 宝洁公司 | Thiophene azo carboxylate dyes and laundry care compositions containing the same |
| CN103703117B (en) * | 2011-06-03 | 2015-11-25 | 宝洁公司 | Thiophene azo carboxylate dyes and laundry care compositions containing the same |
| US8858651B2 (en) | 2011-06-03 | 2014-10-14 | The Procter & Gamble Company | Laundry care compositions containing dyes |
| CN103582696B (en) * | 2011-06-03 | 2015-11-25 | 宝洁公司 | Comprise the laundry care composition of dyestuff |
| EP2540825A2 (en) | 2011-06-30 | 2013-01-02 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants reference to a sequence listing |
| EP3121270A2 (en) | 2011-06-30 | 2017-01-25 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants reference to a sequence listing |
| WO2013011071A1 (en) | 2011-07-21 | 2013-01-24 | Unilever Plc | Liquid laundry composition |
| WO2013016368A1 (en) | 2011-07-25 | 2013-01-31 | The Procter & Gamble Company | Detergent compositions |
| EP2551335A1 (en) | 2011-07-25 | 2013-01-30 | The Procter & Gamble Company | Enzyme stabilized liquid detergent composition |
| EP2551336A1 (en) | 2011-07-25 | 2013-01-30 | The Procter & Gamble Company | Detergent compositions |
| WO2013025742A1 (en) | 2011-08-15 | 2013-02-21 | The Procter & Gamble Company | Detergent compositions containing pyridinol-n-oxide compounds |
| WO2013043857A1 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising sustainable surfactant systems comprising isoprenoid-derived surfactants |
| WO2013043805A1 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising primary surfactant systems comprising highly branched surfactants especially isoprenoid - based surfactants |
| WO2013043855A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | High suds detergent compositions comprising isoprenoid-based surfactants |
| WO2013043852A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Easy-rinse detergent compositions comprising isoprenoid-based surfactants |
| WO2013043803A2 (en) | 2011-09-20 | 2013-03-28 | The Procter & Gamble Company | Detergent compositions comprising specific blend ratios of isoprenoid-based surfactants |
| WO2013006871A3 (en) * | 2012-02-13 | 2013-03-14 | Milliken & Company | Laundry care compositions containing dyes |
| JP2015514141A (en) * | 2012-03-19 | 2015-05-18 | ザ プロクター アンド ギャンブルカンパニー | Laundry care compositions containing dyes |
| EP2828340A1 (en) * | 2012-03-19 | 2015-01-28 | The Procter & Gamble Company | Laundry care compositions containing dyes |
| WO2013189640A1 (en) | 2012-06-21 | 2013-12-27 | Unilever Plc | Liquid laundry detergent |
| WO2013189615A1 (en) | 2012-06-21 | 2013-12-27 | Unilever Plc | Liquid laundry detergent |
| US20150159121A1 (en) * | 2012-06-21 | 2015-06-11 | Conopco, Inc., D/B/A Unilever | Liquid laundry detergent |
| US9249379B2 (en) | 2012-06-21 | 2016-02-02 | Conopco, Inc. | Liquid laundry detergent |
| CN104379718A (en) * | 2012-06-21 | 2015-02-25 | 荷兰联合利华有限公司 | Liquid laundry detergent |
| WO2014012923A1 (en) | 2012-07-17 | 2014-01-23 | Unilever Plc | Bright detergent composition |
| EP2875112B1 (en) | 2012-07-17 | 2017-02-08 | Unilever PLC | Bright detergent composition |
| CN104428405B (en) * | 2012-07-17 | 2018-03-27 | 荷兰联合利华有限公司 | bright detergent composition |
| US20150175942A1 (en) * | 2012-07-17 | 2015-06-25 | Conopco, Inc., D/B/A Unilever | Bright detergent composition |
| WO2014012921A1 (en) | 2012-07-17 | 2014-01-23 | Unilever Plc | Bright detergent composition |
| US9200240B2 (en) | 2012-07-17 | 2015-12-01 | Conopco, Inc. | Bright detergent composition |
| US20150141317A1 (en) * | 2012-07-17 | 2015-05-21 | Conopco, Inc., D/B/A Unilever | Bright detergent composition |
| CN104471049A (en) * | 2012-07-17 | 2015-03-25 | 荷兰联合利华有限公司 | Bright detergent composition |
| US9193941B2 (en) | 2012-07-17 | 2015-11-24 | Conopco, Inc. | Bright detergent composition |
| CN104428405A (en) * | 2012-07-17 | 2015-03-18 | 荷兰联合利华有限公司 | Bright detergent composition |
| CN104471049B (en) * | 2012-07-17 | 2017-07-21 | 荷兰联合利华有限公司 | Bright detergent composition |
| WO2014018309A1 (en) | 2012-07-26 | 2014-01-30 | The Procter & Gamble Company | Low ph liquid cleaning compositions with enzymes |
| WO2014055245A1 (en) | 2012-10-01 | 2014-04-10 | The Procter & Gamble Company | Methods of treating a surface and compositions for use therein |
| EP2712915A1 (en) | 2012-10-01 | 2014-04-02 | The Procter and Gamble Company | Methods of treating a surface and compositions for use therein |
| US9133421B2 (en) | 2012-10-24 | 2015-09-15 | The Procter & Gamble Company | Compositions comprising anti-foams |
| WO2014066309A1 (en) | 2012-10-24 | 2014-05-01 | The Procter & Gamble Company | Anti foam compositions comprising partly phenyl bearing polyorganosilicons |
| WO2014066308A1 (en) | 2012-10-24 | 2014-05-01 | The Procter & Gamble Company | Anti foam compositions comprising aryl bearing polyorganosilicons |
| US9095787B2 (en) | 2012-10-24 | 2015-08-04 | The Procter & Gamble Company | Compositions comprising anti-foams |
| CN102972395B (en) * | 2012-12-06 | 2014-09-10 | 扬州大学 | 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing lygocoris lucorum and method for preparing 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing and treating lygocoris lucorum |
| WO2014089270A1 (en) | 2012-12-06 | 2014-06-12 | The Procter & Gamble Company | Use of composition to reduce weeping and migration through a water soluble film |
| EP2740785A1 (en) | 2012-12-06 | 2014-06-11 | The Procter and Gamble Company | Use of composition to reduce weeping and migration through a water soluble film |
| JP2016504440A (en) * | 2012-12-06 | 2016-02-12 | ザ プロクター アンド ギャンブルカンパニー | Use of compositions to reduce migration and leakage through water-soluble films |
| CN102972395A (en) * | 2012-12-06 | 2013-03-20 | 扬州大学 | 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing lygocoris lucorum and method for preparing 2, 5-bithiophene-2-ethinyl thiophene nano microcapsule preparation for preventing and treating lygocoris lucorum |
| WO2014089386A1 (en) | 2012-12-06 | 2014-06-12 | The Procter & Gamble Company | Soluble pouch comprising hueing dye |
| WO2014130509A1 (en) | 2013-02-19 | 2014-08-28 | The Procter & Gamble Company | Method of laundering a fabric |
| WO2014130512A1 (en) | 2013-02-19 | 2014-08-28 | The Procter & Gamble Company | Method of laundering a fabric |
| EP2767581A1 (en) | 2013-02-19 | 2014-08-20 | The Procter & Gamble Company | Method of laundering a fabric |
| EP2767579A1 (en) | 2013-02-19 | 2014-08-20 | The Procter and Gamble Company | Method of laundering a fabric |
| WO2014130508A1 (en) | 2013-02-19 | 2014-08-28 | The Procter & Gamble Company | Method of laundering a fabric |
| EP2767582A1 (en) | 2013-02-19 | 2014-08-20 | The Procter and Gamble Company | Method of laundering a fabric |
| WO2014160821A1 (en) | 2013-03-28 | 2014-10-02 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine, a soil release polymer, and a carboxymethylcellulose |
| WO2014160820A1 (en) | 2013-03-28 | 2014-10-02 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2014168776A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Hydroxyl polymer fiber structures comprising ammonium alkylsulfonate salts and methods for making same |
| WO2014168775A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Fibrous structures exhibiting improved whiteness index values |
| WO2014168942A1 (en) | 2013-04-12 | 2014-10-16 | The Procter & Gamble Company | Fibrous structures comprising polysaccharide filaments |
| EP2992054B1 (en) | 2013-05-03 | 2017-01-04 | Unilever PLC | Alkoxylated bis azo dyes |
| CN103230766A (en) * | 2013-05-08 | 2013-08-07 | 天津城市建设学院 | Preparation and application of self-adhesive double-wall essence microcapsule |
| CN103230766B (en) * | 2013-05-08 | 2014-12-10 | 天津城市建设学院 | Preparation and application of self-adhesive double-wall essence microcapsule |
| EP3699256A1 (en) | 2013-05-28 | 2020-08-26 | The Procter & Gamble Company | Surface treatment compositions comprising photochromic dyes |
| WO2014193859A1 (en) | 2013-05-28 | 2014-12-04 | The Procter & Gamble Company | Surface treatment compositions comprising photochromic dyes |
| EP2808372A1 (en) | 2013-05-28 | 2014-12-03 | The Procter and Gamble Company | Surface treatment compositions comprising photochromic dyes |
| EP2832842A1 (en) | 2013-07-30 | 2015-02-04 | The Procter & Gamble Company | Method of making granular detergent compositions comprising surfactants |
| EP2832843A1 (en) | 2013-07-30 | 2015-02-04 | The Procter & Gamble Company | Method of making granular detergent compositions comprising polymers |
| WO2015041887A3 (en) * | 2013-09-18 | 2015-05-14 | Milliken & Company | Laundry care composition comprising carboxylate dye |
| JP2016536433A (en) * | 2013-09-18 | 2016-11-24 | ザ プロクター アンド ギャンブル カンパニー | Laundry care composition comprising a carboxylate dye |
| US9834682B2 (en) | 2013-09-18 | 2017-12-05 | Milliken & Company | Laundry care composition comprising carboxylate dye |
| JP2016530392A (en) * | 2013-09-18 | 2016-09-29 | ザ プロクター アンド ギャンブル カンパニー | Laundry care composition containing a thiophene azocarboxylate dye |
| JP2016538350A (en) * | 2013-09-23 | 2016-12-08 | ザ プロクター アンド ギャンブル カンパニー | particle |
| WO2015112340A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| WO2015112341A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015112338A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Method of treating textile fabrics |
| EP2899260A1 (en) | 2014-01-22 | 2015-07-29 | Unilever PLC | Process to manufacture a liquid detergent formulation |
| WO2015112339A1 (en) | 2014-01-22 | 2015-07-30 | The Procter & Gamble Company | Fabric treatment composition |
| WO2015110444A1 (en) | 2014-01-22 | 2015-07-30 | Unilever Plc | Process to manufacture a liquid detergent formulation |
| WO2015127004A1 (en) | 2014-02-19 | 2015-08-27 | The Procter & Gamble Company | Composition comprising benefit agent and aprotic solvent |
| WO2015126547A1 (en) | 2014-02-19 | 2015-08-27 | Milliken & Company | Composition comprising benefit agent and aprotic solvent |
| WO2015130669A1 (en) | 2014-02-25 | 2015-09-03 | The Procter & Gamble Company | A process for making renewable surfactant intermediates and surfactants from fats and oils and products thereof |
| WO2015130653A1 (en) | 2014-02-25 | 2015-09-03 | The Procter & Gamble Company | A process for making renewable surfactant intermediates and surfactants from fats and oils and products thereof |
| WO2015148360A1 (en) | 2014-03-27 | 2015-10-01 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2015148361A1 (en) | 2014-03-27 | 2015-10-01 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| US9574161B2 (en) | 2014-04-29 | 2017-02-21 | The Procter & Gamble Company | Fabric care compositions comprising polyurethane, polyurea and/or polyurethaneurea polymers |
| WO2015168194A1 (en) | 2014-04-29 | 2015-11-05 | The Procter & Gamble Company | Fabric care compositions comprising polyurethane, polyurea and/or polyurethaneurea polymers |
| WO2015187757A1 (en) | 2014-06-06 | 2015-12-10 | The Procter & Gamble Company | Detergent composition comprising polyalkyleneimine polymers |
| EP2987849A1 (en) | 2014-08-19 | 2016-02-24 | The Procter and Gamble Company | Method of Laundering a Fabric |
| EP2987848A1 (en) | 2014-08-19 | 2016-02-24 | The Procter & Gamble Company | Method of laundering a fabric |
| WO2016032991A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016032992A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016032993A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Detergent composition comprising a cationic polymer |
| WO2016032995A1 (en) | 2014-08-27 | 2016-03-03 | The Procter & Gamble Company | Method of treating a fabric |
| WO2016044200A1 (en) | 2014-09-15 | 2016-03-24 | The Procter & Gamble Company | Detergent compositions containing salts of polyetheramines and polymeric acid |
| WO2016048969A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Detergent compositions containing a polyetheramine and an anionic soil release polymer |
| WO2016049388A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Fabric care compositions containing a polyetheramine |
| WO2016048674A1 (en) | 2014-09-25 | 2016-03-31 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2016049387A1 (en) | 2014-09-26 | 2016-03-31 | The Procter & Gamble Company | Cleaning compositions containing a polyetheramine |
| WO2016081437A1 (en) | 2014-11-17 | 2016-05-26 | The Procter & Gamble Company | Benefit agent delivery compositions |
| WO2016176296A1 (en) | 2015-04-29 | 2016-11-03 | The Procter & Gamble Company | Method of laundering a fabric |
| EP3088504A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| EP3674387A1 (en) | 2015-04-29 | 2020-07-01 | The Procter & Gamble Company | Method of treating a fabric |
| WO2016176282A1 (en) | 2015-04-29 | 2016-11-03 | The Procter & Gamble Company | Method of treating a fabric |
| WO2016176240A1 (en) | 2015-04-29 | 2016-11-03 | The Procter & Gamble Company | Method of treating a fabric |
| EP3088505A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| EP3088506A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Detergent composition |
| EP3088502A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| WO2016176241A1 (en) | 2015-04-29 | 2016-11-03 | The Procter & Gamble Company | Detergent composition |
| WO2016176280A1 (en) | 2015-04-29 | 2016-11-03 | The Procter & Gamble Company | Method of treating a fabric |
| EP3088503A1 (en) | 2015-04-29 | 2016-11-02 | The Procter and Gamble Company | Method of treating a fabric |
| WO2016188693A1 (en) | 2015-05-27 | 2016-12-01 | Unilever Plc | Laundry detergent composition |
| WO2016192905A1 (en) | 2015-06-02 | 2016-12-08 | Unilever Plc | Laundry detergent composition |
| WO2016200440A1 (en) | 2015-06-11 | 2016-12-15 | The Procter & Gamble Company | Device and methods for applying compositions to surfaces |
| WO2017055205A1 (en) | 2015-10-01 | 2017-04-06 | Unilever Plc | Powder laundry detergent composition |
| WO2017091674A1 (en) | 2015-11-26 | 2017-06-01 | The Procter & Gamble Company | Liquid detergent compositions comprising protease and encapsulated lipase |
| EP3173467A1 (en) | 2015-11-26 | 2017-05-31 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2017140392A1 (en) | 2016-02-17 | 2017-08-24 | Unilever Plc | Whitening composition |
| WO2017140391A1 (en) | 2016-02-17 | 2017-08-24 | Unilever Plc | Whitening composition |
| WO2017198574A1 (en) | 2016-05-17 | 2017-11-23 | Unilever Plc | Liquid laundry detergent compositions |
| WO2017198438A1 (en) | 2016-05-17 | 2017-11-23 | Unilever Plc | Liquid laundry detergent compositions |
| WO2018060139A1 (en) | 2016-09-27 | 2018-04-05 | Unilever Plc | Domestic laundering method |
| WO2018067486A1 (en) * | 2016-10-03 | 2018-04-12 | The Procter & Gamble Company | Low ph laundry detergent composition |
| WO2018067487A1 (en) * | 2016-10-03 | 2018-04-12 | The Procter & Gamble Company | Low ph laundry detergent composition |
| RU2709518C1 (en) * | 2016-10-03 | 2019-12-18 | Дзе Проктер Энд Гэмбл Компани | DETERGENT COMPOSITION FOR WASHING WITH LOW pH |
| CN109715774A (en) * | 2016-10-03 | 2019-05-03 | 宝洁公司 | Low pH laundry detergent composition |
| WO2018067488A1 (en) * | 2016-10-03 | 2018-04-12 | The Procter & Gamble Company | Low ph laundry detergent composition |
| US10676703B2 (en) | 2016-10-03 | 2020-06-09 | The Procter & Gamble Company | Process for preparing a spray-dried laundry detergent particle |
| EP3301162A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| CN109715774B (en) * | 2016-10-03 | 2021-10-01 | 宝洁公司 | Low pH laundry detergent compositions |
| EP3301164A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| EP3301163A1 (en) * | 2016-10-03 | 2018-04-04 | The Procter & Gamble Company | Low ph laundry detergent composition |
| WO2018072979A1 (en) | 2016-10-18 | 2018-04-26 | Unilever Plc | Whitening composition |
| EP3357994A1 (en) | 2017-02-01 | 2018-08-08 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants |
| WO2018144399A1 (en) | 2017-02-01 | 2018-08-09 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants |
| WO2019008036A1 (en) | 2017-07-07 | 2019-01-10 | Unilever Plc | Whitening composition |
| WO2019008035A1 (en) | 2017-07-07 | 2019-01-10 | Unilever Plc | Laundry cleaning composition |
| WO2019105675A1 (en) | 2017-11-30 | 2019-06-06 | Unilever Plc | Detergent composition comprising protease |
| WO2019162130A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Shaped detergent product comprising aminopolycarboxylate |
| WO2019162138A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Solid compositions comprising aminopolycarboxylate |
| WO2019162136A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Detergent solid composition comprising aminopolycarboxylate and organic acid |
| WO2019162132A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Detergent solid composition comprising aminopolycarboxylate and inorganic acid. |
| WO2019162134A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Solid compositions comprising aminopolycarboxylate |
| WO2019162133A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Shaped detergent product composition comprising aminopolycarboxylate |
| WO2019162137A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Water-soluble film comprising aminopolycarboxylate |
| WO2019162135A1 (en) | 2018-02-23 | 2019-08-29 | Unilever N.V. | Process of preparing a solid composition comprising aminopolycarboxylate |
| WO2019192813A1 (en) | 2018-04-03 | 2019-10-10 | Unilever N.V. | Dye granule |
| WO2019219531A1 (en) | 2018-05-17 | 2019-11-21 | Unilever Plc | Cleaning composition |
| WO2019219302A1 (en) | 2018-05-17 | 2019-11-21 | Unilever Plc | Cleaning composition comprising rhamnolipid and alkyl ether carboxylate surfactants |
| WO2020016097A1 (en) | 2018-07-17 | 2020-01-23 | Unilever Plc | Use of a rhamnolipid in a surfactant system |
| WO2020058024A1 (en) | 2018-09-17 | 2020-03-26 | Unilever Plc | Detergent composition |
| WO2020104155A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104159A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104157A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104156A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020104158A1 (en) | 2018-11-20 | 2020-05-28 | Unilever Plc | Detergent composition |
| WO2020186030A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2020186028A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Cleaning compositions comprising enzymes |
| WO2020186052A1 (en) | 2019-03-14 | 2020-09-17 | The Procter & Gamble Company | Method for treating cotton |
| EP3750979A1 (en) | 2019-06-12 | 2020-12-16 | Unilever N.V. | Use of laundry detergent composition |
| EP3750978A1 (en) | 2019-06-12 | 2020-12-16 | Unilever N.V. | Laundry detergent composition |
| WO2020264552A1 (en) | 2019-06-24 | 2020-12-30 | The Procter & Gamble Company | Cleaning compositions comprising amylase variants |
| WO2020259948A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020259949A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020260040A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020260038A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2020260006A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent compositions |
| WO2020259947A1 (en) | 2019-06-28 | 2020-12-30 | Unilever Plc | Detergent composition |
| WO2021032833A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021032818A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021032815A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | An embossed detergent solid |
| WO2021032834A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021032817A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021032816A1 (en) | 2019-08-21 | 2021-02-25 | Unilever Ip Holdings B.V. | Detergent solid composition |
| WO2021043764A1 (en) | 2019-09-02 | 2021-03-11 | Unilever Global Ip Limited | Detergent composition |
| WO2021069516A1 (en) | 2019-10-07 | 2021-04-15 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021133701A1 (en) | 2019-12-23 | 2021-07-01 | The Procter & Gamble Company | Compositions comprising enzymes |
| US12031113B2 (en) | 2020-03-02 | 2024-07-09 | Milliken & Company | Composition comprising hueing agent |
| WO2021185956A1 (en) | 2020-03-19 | 2021-09-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021185870A1 (en) | 2020-03-19 | 2021-09-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2021249927A1 (en) | 2020-06-08 | 2021-12-16 | Unilever Ip Holdings B.V. | Method of improving protease activity |
| WO2022023250A1 (en) | 2020-07-27 | 2022-02-03 | Unilever Ip Holdings B.V. | Use of an enzyme and surfactant for inhibiting microorganisms |
| WO2022042977A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022042989A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Surfactant and detergent composition |
| WO2022043138A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Surfactant and detergent composition |
| WO2022043042A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022043045A1 (en) | 2020-08-28 | 2022-03-03 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2022094163A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning composition comprising alginate lyase enzymes |
| WO2022094589A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning compositions containing alginase enzymes |
| WO2022094590A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning compositions containing alginate lyase enzymes |
| WO2022094588A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning compositions containing alginate lyase enzymes |
| WO2022094164A1 (en) | 2020-10-29 | 2022-05-05 | The Procter & Gamble Company | Cleaning composition comprising alginate lyase enzymes |
| WO2022128781A1 (en) | 2020-12-17 | 2022-06-23 | Unilever Ip Holdings B.V. | Cleaning composition |
| WO2022128786A1 (en) | 2020-12-17 | 2022-06-23 | Unilever Ip Holdings B.V. | Use and cleaning composition |
| WO2022197512A1 (en) | 2021-03-15 | 2022-09-22 | The Procter & Gamble Company | Cleaning compositions containing polypeptide variants |
| EP4060010A2 (en) | 2021-03-15 | 2022-09-21 | The Procter & Gamble Company | Cleaning compositions containing polypeptide variants |
| WO2022235720A1 (en) | 2021-05-05 | 2022-11-10 | The Procter & Gamble Company | Methods for making cleaning compositions and detecting soils |
| EP4095223A1 (en) | 2021-05-05 | 2022-11-30 | The Procter & Gamble Company | Methods for making cleaning compositions and for detecting soils |
| EP4108767A1 (en) | 2021-06-22 | 2022-12-28 | The Procter & Gamble Company | Cleaning or treatment compositions containing nuclease enzymes |
| WO2022272255A1 (en) | 2021-06-22 | 2022-12-29 | The Procter & Gamble Company | Cleaning or treatment compositions containing nuclease enzymes |
| WO2022268657A1 (en) | 2021-06-24 | 2022-12-29 | Unilever Ip Holdings B.V. | Unit dose cleaning composition |
| WO2022268728A1 (en) | 2021-06-24 | 2022-12-29 | Unilever Ip Holdings B.V. | Unit dose cleaning composition |
| WO2023278970A1 (en) | 2021-06-30 | 2023-01-05 | The Procter & Gamble Company | Fabric treatment |
| EP4112707A1 (en) | 2021-06-30 | 2023-01-04 | The Procter & Gamble Company | Fabric treatment |
| WO2023017794A1 (en) | 2021-08-10 | 2023-02-16 | 株式会社日本触媒 | Polyalkylene-oxide-containing compound |
| WO2023041694A1 (en) | 2021-09-20 | 2023-03-23 | Unilever Ip Holdings B.V. | Detergent composition |
| WO2023067073A1 (en) | 2021-10-21 | 2023-04-27 | Unilever Ip Holdings B.V. | Detergent compositions |
| WO2023067074A1 (en) | 2021-10-21 | 2023-04-27 | Unilever Ip Holdings B.V. | Detergent compositions |
| WO2023067075A1 (en) | 2021-10-21 | 2023-04-27 | Unilever Ip Holdings B.V. | Detergent compositions |
| WO2023144071A1 (en) | 2022-01-28 | 2023-08-03 | Unilever Ip Holdings B.V. | Laundry composition |
| EP4273210A1 (en) | 2022-05-04 | 2023-11-08 | The Procter & Gamble Company | Detergent compositions containing enzymes |
| WO2023215679A1 (en) | 2022-05-04 | 2023-11-09 | The Procter & Gamble Company | Detergent compositions containing enzymes |
| EP4410941A1 (en) | 2023-02-01 | 2024-08-07 | The Procter & Gamble Company | Detergent compositions containing enzymes |
| WO2024163695A1 (en) | 2023-02-01 | 2024-08-08 | The Procter & Gamble Company | Detergent compositions containing enzymes |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2011011799A3 (en) | 2011-10-06 |
| CN103210073A (en) | 2013-07-17 |
| JP5833133B2 (en) | 2015-12-16 |
| CA2817718C (en) | 2016-02-09 |
| CN103210073B (en) | 2016-06-08 |
| CA2817718A1 (en) | 2011-01-27 |
| BR112013011851A2 (en) | 2016-08-16 |
| EP2638142A2 (en) | 2013-09-18 |
| MX2013005276A (en) | 2013-06-03 |
| EP2638142B1 (en) | 2017-05-10 |
| JP2014501802A (en) | 2014-01-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2638142B1 (en) | Thiophene azo dyes and laundry care compositions containing the same | |
| EP2638113B1 (en) | Thiophene azo dyes and laundry care compositions containing the same | |
| EP2714815B1 (en) | Thiophene azo carboxylate dyes and laundry care compositions containing the same | |
| US10655091B2 (en) | Thiophene azo dyes and laundry care compositions containing the same | |
| EP2630197B1 (en) | Bis-azo colorants for use as bluing agents | |
| JP6005733B2 (en) | Thiophene azocarboxylate dye and laundry care composition containing the same | |
| US9556337B2 (en) | Thiophene azo dyes and laundry care compositions containing the same | |
| US20120101018A1 (en) | Bis-azo colorants for use as bluing agents | |
| WO2012054058A1 (en) | Bis-azo colorants for use as bluing agents | |
| US8962815B2 (en) | Bis-azo colorants for use as bluing agents | |
| EP3408368B1 (en) | Bis-azo colorants for use as bluing agents | |
| WO2017131852A1 (en) | Bis-azo colorants for use as bluing agents | |
| JP6141369B2 (en) | Thiopheneazo dye and laundry care composition containing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10781555 Country of ref document: EP Kind code of ref document: A2 |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010781555 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010781555 Country of ref document: EP |
|
| ENP | Entry into the national phase in: |
Ref document number: 2013538698 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2013/005276 Country of ref document: MX |
|
| ENP | Entry into the national phase in: |
Ref document number: 2817718 Country of ref document: CA |
|
| NENP | Non-entry into the national phase in: |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112013011851 Country of ref document: BR |
|
| ENP | Entry into the national phase in: |
Ref document number: 112013011851 Country of ref document: BR Kind code of ref document: A2 Effective date: 20130513 |