WO2011005925A1 - Core/shell rubbers for use in electrical laminate compositions - Google Patents
Core/shell rubbers for use in electrical laminate compositions Download PDFInfo
- Publication number
- WO2011005925A1 WO2011005925A1 PCT/US2010/041311 US2010041311W WO2011005925A1 WO 2011005925 A1 WO2011005925 A1 WO 2011005925A1 US 2010041311 W US2010041311 W US 2010041311W WO 2011005925 A1 WO2011005925 A1 WO 2011005925A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- epoxy
- epoxy resin
- silicone
- composition
- compositions
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L63/00—Compositions of epoxy resins; Compositions of derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G59/00—Polycondensates containing more than one epoxy group per molecule; Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups
- C08G59/18—Macromolecules obtained by polymerising compounds containing more than one epoxy group per molecule using curing agents or catalysts which react with the epoxy groups ; e.g. general methods of curing
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/01—Use of inorganic substances as compounding ingredients characterized by their specific function
- C08K3/016—Flame-proofing or flame-retarding additives
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/16—Halogen-containing compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L51/00—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L51/04—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers grafted on to rubbers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L51/00—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L51/08—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving unsaturated carbon-to-carbon bonds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L51/00—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers
- C08L51/08—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving unsaturated carbon-to-carbon bonds
- C08L51/085—Compositions of graft polymers in which the grafted component is obtained by reactions only involving carbon-to-carbon unsaturated bonds; Compositions of derivatives of such polymers grafted on to macromolecular compounds obtained otherwise than by reactions only involving unsaturated carbon-to-carbon bonds on to polysiloxanes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D163/00—Coating compositions based on epoxy resins; Coating compositions based on derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09J—ADHESIVES; NON-MECHANICAL ASPECTS OF ADHESIVE PROCESSES IN GENERAL; ADHESIVE PROCESSES NOT PROVIDED FOR ELSEWHERE; USE OF MATERIALS AS ADHESIVES
- C09J163/00—Adhesives based on epoxy resins; Adhesives based on derivatives of epoxy resins
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2207/00—Properties characterising the ingredient of the composition
- C08L2207/53—Core-shell polymer
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2666/00—Composition of polymers characterized by a further compound in the blend, being organic macromolecular compounds, natural resins, waxes or and bituminous materials, non-macromolecular organic substances, inorganic substances or characterized by their function in the composition
- C08L2666/02—Organic macromolecular compounds, natural resins, waxes or and bituminous materials
- C08L2666/24—Graft or block copolymers according to groups C08L51/00, C08L53/00 or C08L55/02; Derivatives thereof
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/582—Recycling of unreacted starting or intermediate materials
Definitions
- Embodiments disclosed herein relate to epoxy compositions. More specifically, embodiments disclosed herein relate to epoxy compositions useful in electrical laminates. More specifically, embodiments disclosed herein relate to low dielectric constant epoxy compositions formed from epoxy resins and core/shell toughening agents, such as a silicone-acrylate core/shell rubber.
- Thermosettable compositions useful in high-performance electrical applications must meet a set of demanding property requirements.
- such materials optimally have good high-temperature properties such as high glass transition temperatures (e.g., above 200 0 C) and low water absorption at elevated temperature (e.g., less than 4% water adsorption).
- Such materials must also exhibit stable solubility in organic solvents, such as acetone, as the preparation of electrical laminates conventionally involves impregnation of a porous glass web with a solution of the thermosettable resin.
- the uncured material will ideally have a low melting temperature (e.g., below 12O 0 C) and a wide temperature range of processable viscosity (a wide "processing window").
- Epoxy resins are one of the most widely used engineering resins, and are well-known for their use in electrical laminates. Epoxy resins have been used as materials for electrical/electronics equipment, such as materials for electrical laminates because of their superiority in heat resistance, chemical resistance, insulation property, dimensional stability, adhesiveness and the like.
- a second trend is that the speed of electronic devices is increasing.
- printed circuit boards with improved dielectric properties e.g., a lower dielectric constant (D k ) and dissipation factor (D f ) are needed.
- compositions having desirable toughness, dielectric properties, and thermal properties, that are useful in electrical laminates.
- composition comprising, consisting of, or consisting essentially of: an epoxy resin; a curing agent; and a silicone-acrylate core/shell rubber.
- a process comprising, consisting of, or consisting essentially of: dispersing a silicone- acrylate core/shell rubber in a solvent; admixing the dispersed silicone- acrylate core/shell rubber with an epoxy resin and one or more of a hardener, a catalyst, and additional solvent to form a curable composition.
- Embodiments disclosed herein relate to epoxy compositions. More specifically, embodiments disclosed herein relate to epoxy compositions useful in electrical laminates. More specifically, embodiments disclosed herein relate to low dielectric constant epoxy compositions formed from epoxy resins and core/shell toughening agents, such as a silicone-acrylate core/shell rubber.
- compositions disclosed herein may include at least one epoxy resin, at least one hardener or curing agent, and a silicone-acrylate core/shell rubber toughening agent. Such compositions are useful in electrical laminates, for example, due to the resulting thermoset resin having desirable electrical properties and physical properties, including impact resistance.
- curable compositions may be formed by dispersing a silicone-acrylate core/shell rubber toughening agent in a liquid epoxy resin. In other embodiments, curable compositions may be formed by dispersing a silicone-acrylate core/shell rubber toughening agent in a solvent, and then admixing the dispersion with an epoxy resin and one or more of a hardener, a catalyst, and additional solvent, to form a curable composition.
- Thermoset compositions may be formed as a reaction product of the above-described curable compositions including at least one epoxy resin, at least one hardener, and a silicone-acrylate core/shell rubber. Such thermoset compositions are useful in electrical laminates, among other applications.
- embodiments disclosed herein include various components, including epoxy resins, silicone-acrylate core/shell rubbers, and hardeners.
- Embodiments of compositions described herein may also include catalysts and various additives. Examples of each of these components are described in more detail below. EPOXY RESINS
- the epoxy resins used in embodiments disclosed herein may vary and include conventional and commercially available epoxy resins, which may be used alone or in combinations of two or more, including, for example, novolac resins, isocyanate modified epoxy resins, and carboxylate adducts, among others.
- novolac resins novolac resins
- isocyanate modified epoxy resins and carboxylate adducts, among others.
- the epoxy resin component may be any type of epoxy resin useful in molding compositions, including any material containing one or more reactive oxirane groups, referred to herein as "epoxy groups” or "epoxy functionality.”
- Epoxy resins useful in embodiments disclosed herein may include mono- functional epoxy resins, multi- or poly-functional epoxy resins, and combinations thereof.
- Monomeric and polymeric epoxy resins may be aliphatic, cycloaliphatic, aromatic, or heterocyclic epoxy resins.
- the polymeric epoxies include linear polymers having terminal epoxy groups (a diglycidyl ether of a polyoxyalkylene glycol, for example), polymer skeletal oxirane units (polybutadiene polyepoxide, for example) and polymers having pendant epoxy groups (such as a glycidyl methacrylate polymer or copolymer, for example).
- the epoxies may be pure compounds, but are generally mixtures or compounds containing one, two or more epoxy groups per molecule.
- epoxy resins may also include reactive -OH groups, which may react at higher temperatures with anhydrides, organic acids, amino resins, phenolic resins, or with epoxy groups (when catalyzed) to result in additional crosslinking.
- the epoxy resins may be glycidyl ethers, cycloaliphatic resins, epoxidized oils, and so forth.
- Illustrative polyepoxide compounds useful in embodiments disclosed herein are described in the 2 nd chapter of "Epoxy Resins" by Clayton A. May, published in 1988 by Marcel Dekker, Inc., New York, and U.S. Patent No. 4,066,628.
- the glycidyl ethers are frequently the reaction product of epichlorohydrin and a phenol or polyphenolic compound such as bisphenol A (commercially available as D.E.R.TM 383 or D.E.R.TM 330 from The Dow Chemical Company, Midland, Michigan); pyrocatechol, resorcinol, hydroquinone, 4,4'-dihydroxydiphenyl methane (or bisphenol F), 4,4'-dihydroxy-3,3'-dimethyldiphenyl methane, 4,4'- dihydroxydiphenyl dimethyl methane (or bisphenol A), 4,4'- dihydroxydiphenyl methyl methane, 4,4'-dihydroxydiphenyl cyclohexane, 4,4'-dihydroxy-3,3'-dimethyldiphenyl propane, 4,4'-dihydroxydiphenyl sulfone, and tris(4-hydroxyphenyl)methane; chlorinated or brominated products of the above-ment
- such materials typically contain small amounts of oligomers derived from condensation of the phenolic starting material with the glycidyl ether product.
- 'Advanced' resins are prepared by reacting a polyepoxide with a polyphenol.
- Such oligomers are useful in the formulation to achieve useful rheology and cure characteristics. Specific examples include the condensation products of bisphenol A diglycidyl ether with bisphenol A, tetrabromobisphenol A or the condensation products of the diglycidyl ether of tetrabromobisphenol A with bisphenol A or tetrabromobisphenol A.
- aromatic isocyanates such as methylene diisocyanate or toluene diisocyanate may be added during these advancement reactions to give oligomers that contain oxazolidinone heterocycles in the backbone of the chains.
- aromatic isocyanates such as methylene diisocyanate or toluene diisocyanate may be added during these advancement reactions to give oligomers that contain oxazolidinone heterocycles in the backbone of the chains.
- D.E.R.TM 592 and D.E.R.TM 593 each available from The Dow Chemical Company, Midland Michigan.lt is common to add the glycidyl ethers of novolacs, which are polyphenols derived from condensation of formaldehyde or other aldehyde with a phenol. Specific examples include the novolacs of phenol, cresol, dimethylphenols, p- hydroxybiphenyl, naphthol, and bromophenols.
- epoxy resins are derived from epoxidation of olefins, typically with peracids or hydrogen peroxide.
- the olefins may be contained within a linear or cyclic chain.
- the epoxy resin may include glycidyl ether type; glycidyl-ester type; alicyclic type; heterocyclic type, and halogenated epoxy resins, etc.
- suitable epoxy resins may include cresol novolac epoxy resin, phenolic novolac epoxy resin, biphenyl epoxy resin, hydroquinone epoxy resin, stilbene epoxy resin, and mixtures and combinations thereof.
- Suitable polyepoxy compounds may include resorcinol diglycidyl ether (l,3-bis-(2,3-epoxypropoxy)benzene), diglycidyl ether of bisphenol A (2,2- bis(p-(2,3-epoxypropoxy)phenyl)propane), triglycidyl p-aminophenol (4-(2,3- epoxypropoxy)-N,N-bis(2,3-epoxypropyl)aniline), diglycidyl ether of bromobisphenol A (2,2-bis(4-(2,3-epoxypropoxy)3-bromo-phenyl)propane), diglycidyl ether of bisphenol F (2,2-bis(p-(2,3- epoxypropoxy)phenyl)methane), triglycidyl ether of meta- and/or para- aminophenol (3-(2,3-epoxypropoxy)N,N-bis(2,3-epoxypropyl)aniline
- Epoxy resins include polyepoxy compounds based on aromatic amines and epichlorohydrin, such as N,N'-diglycidyl-aniline; N 5 N'- dimethyl-N,N'-diglycidyl-4,4'-diaminodiphenyl methane; N 5 N 5 N' ,N'- tetraglycidyl-4,4'-diaminodiphenyl methane; N-diglycidyl-4-aminophenyl glycidyl ether; and N,N,N',N'-tetraglycidyl-l,3-propylene bis-4- aminobenzoate.
- Epoxy resins may also include glycidyl derivatives of one or more of: aromatic diamines, aniline and substituted derivatives, aminophenols, polyhydric phenols, polyhydric alcohols, polycarboxylic acids.
- Useful epoxy resins include, for example, polyglycidyl ethers of polyhydric polyols, such as ethylene glycol, Methylene glycol, 1,2-propylene glycol, 1,5-pentanediol, 1,2,6-hexanetriol, glycerol, and 2,2-bis(4-hydroxy cyclohexyl)propane; polyglycidyl ethers of aliphatic and aromatic polycarboxylic acids, such as, for example, oxalic acid, succinic acid, glutaric acid, terephthalic acid, 2,6-napthalene dicarboxylic acid, and dimerized linoleic acid; polyglycidyl ethers of polyphenols, such as, for example, bis- phenol A, bis-phenol F, l,l-bis(4-hydroxyphenyl)ethane, l,l-bis(4- hydroxyphenyl)isobutane, and 1,5-dihydroxy
- the epoxy compounds may be cycloaliphatic or alicyclic epoxides.
- cycloaliphatic epoxides include diepoxides of cycloaliphatic esters of dicarboxylic acids such as bis(3,4-epoxycyclohexylmethyl)oxalate, bis(3,4-epoxycyclohexylmethyl)adipate, bis(3,4-epoxy-6- methylcyclohexylmethyl)adipate, bis(3,4-epoxycyclohexylmethyl)pimelate; vinylcyclohexene diepoxide; limonene diepoxide; dicyclopentadiene diepoxide; and the like.
- Other suitable diepoxides of cycloaliphatic esters of dicarboxylic acids are described, for example, in U.S. Patent No. 2,750,395.
- cycloaliphatic epoxides include 3,4-epoxycyclohexylmethyl-3,4- epoxycyclohexane carboxylates such as 3,4-epoxycyclohexylmethyl-3,4- epoxycyclohexane carboxylate; 3,4-epoxy-l-methylcyclohexyl-methyl-3,4- epoxy-1-methylcyclohexane carboxylate; 6-methyl-3,4- epoxycyclohexylmethylmethyl-6-methyl-3,4-epoxycyclohexane carboxylate; 3,4-epoxy-2-methylcyclohexylmethyl-3,4-epoxy-2-methylcyclohexane carboxylate; S ⁇ -epoxy-S-methylcyclohexyl-methyl-S ⁇ -epoxy-S- methylcyclohexane carboxylate; 3,4-epoxy-5-methylcyclohexyl-methyl-3,4- epoxy-5-methylcyclohexane carboxylate carboxy
- epoxy-containing materials which are particularly useful include those based on glycidyl ether monomers.
- examples are di- or polyglycidyl ethers of polyhydric phenols obtained by reacting a polyhydric phenol with an excess of chlorohydrin such as epichlorohydrin.
- Such polyhydric phenols include resorcinol, bis(4-hydroxyphenyl)methane (known as bisphenol F), 2,2- bis(4-hydroxyphenyl)propane (known as bisphenol A), 2,2-bis(4'-hydroxy- 3',5'-dibromophenyl)propane, l,l,2,2-tetrakis(4'-hydroxy-phenyl)ethane or condensates of phenols with formaldehyde that are obtained under acid conditions such as phenol novolacs and cresol novolacs. Examples of this type of epoxy resin are described in U.S. Patent No. 3,018,262.
- di- or polyglycidyl ethers of polyhydric alcohols such as 1,4- butanediol
- polyalkylene glycols such as polypropylene glycol
- di- or polyglycidyl ethers of cycloaliphatic polyols such as 2,2-bis(4- hydroxycyclohexyl)propane.
- monofunctional resins such as cresyl glycidyl ether or butyl glycidyl ether.
- Another class of epoxy compounds are polyglycidyl esters and poly(beta-methylglycidyl) esters of polyvalent carboxylic acids such as phthalic acid, terephthalic acid, tetrahydrophthalic acid or hexahydrophthalic acid.
- a further class of epoxy compounds are N-glycidyl derivatives of amines, amides and heterocyclic nitrogen bases such as N,N-diglycidyl aniline, N,N-diglycidyl toluidine, N,N,N',N'-tetraglycidyl bis(4- aminophenyl)methane, triglycidyl isocyanurate, N,N'-diglycidyl ethyl urea,
- N,N'-diglycidyl-5,5-dimethylhydantoin and N,N'-diglycidyl-5- isopropylhydantoin.
- Still other epoxy-containing materials are copolymers of acrylic acid esters of glycidol such as glycidyl acrylate and glycidyl methacrylate with one or more copolymerizable vinyl compounds. Examples of such copolymers are
- Epoxy compounds that are readily available include octadecylene oxide; glycidylmethacrylate; diglycidyl ether of bisphenol A; D.E.R.TM 331 (bisphenol A liquid epoxy resin) and D.E.R.TM 332 (diglycidyl ether of bisphenol A) available from The Dow Chemical Company, Midland, Michigan; vinylcyclohexene dioxide; 3,4-epoxycyclohexylmethyl-3,4- epoxycyclohexane carboxylate; 3,4-epoxy-6-methylcyclohexyl-methyl-3,4- epoxy-6-methylcyclohexane carboxylate; bis(3,4-epoxy-6- methylcyclohexylmethyl) adipate; bis(2,3-epoxycyclopentyl) ether; aliphatic epoxy modified with polypropylene glycol; dipentene dioxide; epoxidized polybutadiene; silicone resin containing epoxy functionality; flame retardant epoxy resins
- Epoxy resins may also include isocyanate modified epoxy resins.
- Polyepoxide polymers or copolymers with isocyanate or polyisocyanate functionality may include epoxy-polyurethane copolymers. These materials may be formed by the use of a polyepoxide prepolymer having one or more oxirane rings to give a 1,2-epoxy functionality and also having open oxirane rings, which are useful as the hydroxyl groups for the dihydroxyl-containing compounds for reaction with diisocyanate or polyisocyanates. The isocyanate moiety opens the oxirane ring and the reaction continues as an isocyanate reaction with a primary or secondary hydroxyl group. There is sufficient epoxide functionality on the polyepoxide resin to enable the production of an epoxy polyurethane copolymer still having effective oxirane rings. Linear polymers may be produced through reactions of diepoxides and diisocyanates.
- the di- or polyisocyanates may be aromatic or aliphatic in some embodiments.
- Silicone-acrylate core/shell rubber toughening agents may be used to prevent the composites disclosed herein from becoming brittle when the epoxy resin cures.
- silicone-acrylate core/shell rubber toughening agents may be a rubber compound including a silicone rubber core and an acrylate polymer shell.
- silicone- acrylate core/shell rubber toughening agents used in embodiments disclosed herein functions by forming a secondary phase within the epoxy polymer matrix.
- This secondary phase is rubbery and hence is capable of crack growth arrestment, providing improved toughness.
- Silicone-acrylate core/shell rubbers useful in embodiments disclosed herein may contain particulate, highly cross-linked silicone rubber particles of an average diameter (dso) of from 0.1 to 3 microns, in particular from 0.1 to 1 micron, and gel contents greater than 60 wt-%, in particular greater than 80 wt-% (where particle size is as measured by light scattering techniques, and gel content is measured by solvent dissolution techniques).
- the acrylate rubber which is grafted on to the silicone rubber particles is present in the silicone/acrylate core/shell rubbers preferably in quantities of 50 wt-% or less, in particular in quantities of from 30 to 5 wt-% and may have gel contents >70 wt-%, in particular >85 wt-%.
- the acrylate rubber moiety of the silicone- acrylate core/shell rubbers is polymerized on to the silicone rubber particles; the following can thus form: graft polymers in the sense of covalent compounds of silicone rubber and acrylate rubber, cross-linked acrylate rubber moieties which encase the silicone rubber particles in a manner more or less mechanical, and optionally small quantities of soluble acrylate rubbers.
- silicone-acrylate core/shell rubbers designate the reaction products which are obtained by polymerization of acrylate in the presence of silicone rubber particles, irrespective of the actual extent of grafting.
- the silicone rubber backbone in some embodiments, may also be a cross-linked silicone rubber.
- the silicone rubbers contain groups which can be rendered capable of radical addition or transfer reaction.
- groups may include vinyl, allyl, chloroalkyl and mercapto groups, in quantities of from 2 to 10 mole %, calculated on the radicals R.
- the acrylate rubber polymer b) grafted on to the silicone rubber core a) represents a partially to highly cross-linked acrylate rubber and is a polymer of from 100 to 60 weight percent alkyl acrylate, from 60 to 0 weight percent of other monomers which are copolymerizable with alkyl acrylate, and, if necessary, from 0.1 to 10 weight percent, calculated on the sum of alkyl acrylate and other monomers, of a cross-linking monomer having at least two vinyl and/or allyl groups in the molecule.
- Alkyl acrylates may include C 4 to Ci 4 alkyl acrylates, such as, for example, methyl, ethyl, butyl, octyl and 2-ethylhexyl acrylate, chloroethyl acrylate, benzyl acrylate, phenethyl acrylate, such as Ci to Ce alkyl esters, including butyl acrylate.
- Ci 4 alkyl acrylates such as, for example, methyl, ethyl, butyl, octyl and 2-ethylhexyl acrylate, chloroethyl acrylate, benzyl acrylate, phenethyl acrylate, such as Ci to Ce alkyl esters, including butyl acrylate.
- Monomers that are copolymerizable with the alkyl acrylates may include styrene, alpha-methylstyrene, halostyrene, methoxystyrene, acrylonitrile, methacrylonitrile, Ci to Cg alkyl methacrylates which may be substituted in the alkyl radical optionally by functional groups such as hydroxyl, epoxy or amine groups, for example methyl methacrylate, cyclohexyl methacrylate, glycidyl methacrylate, hydroxyethyl methacrylate, hydroxypropyl methacrylate, (meth)acrylic acid, maleic acid (ester), fumaric acid, itaconic acid, (meth)acrylamides, vinyl acetate, vinyl propionate or N- methylol compounds of (meth)acrylamides.
- Cross-linking monomers may include esters of unsaturated carboxylic acids having a polyol (preferably from 2 to 20 carbon atoms in the ester group), such as ethylene glycol dimethacrylate, esters of a polyfunctional carboxylic acid having an unsaturated alcohol (preferably from 8 to 30 carbon atoms in the ester radical), such as triallyl cyanurate, triallyl isocyanurate; divinyl compounds such as divinylbenzene; esters of unsaturated carboxylic acids having unsaturated alcohols (preferably from 6 to 12 carbon atoms in the ester radical) such as allyl methacrylate; phosphoric acid esters, for example triallyl phosphate and 1,3,5-triacrylolylhexahydro-s-triazine.
- esters of unsaturated carboxylic acids having a polyol preferably from 2 to 20 carbon atoms in the ester group
- esters of a polyfunctional carboxylic acid having an unsaturated alcohol preferably from 8 to
- the silicone-acrylate core/shell rubbers may be prepared, for example, in aqueous emulsion in the following manner: in a first stage, the silicone rubber, that is to say the core a), is first prepared by emulsion polymerizing a silicone oligomer.
- the monomers (alkyl acrylate, optionally cross-linking monomers and optionally further monomers) which form the acrylate rubber b) are then graft polymerized in the presence of the silicone rubber emulsion of the first stage. Formation of new particles should be as far as possible suppressed during this graft polymerization.
- An emulsion stabilizer is present in the quantity necessary for covering the surface of the particles.
- Graft polymerization is preferably accomplished within the temperature range 3O 0 C to 9O 0 C, and is initiated by known radical initiators, for example, azo- initiators, peroxides, peresters, persulphates, perphosphates or by redox initiator systems.
- stable aqueous emulsions of the silicone rubber/acrylate rubber particles arise, normally with polymer solids contents within the range 20 to 50 wt-%.
- silicone-acrylate core/shell rubber toughening agents used in the curable compositions described herein may depend on a variety of factors including the equivalent weight of the polymers, as well as the desired properties of the products made from the composition. In general, the amount of silicone-acrylate core/shell rubber may be used in an amount ranging from
- Another component, which may be added to the compositions disclosed herein, is a solvent or a blend of solvents.
- the solvent used in the epoxy resin composition may be miscible with the other components in the resin composition.
- the solvent used may be selected from those typically used in making electrical laminates. Examples of suitable solvents employed in the present invention include, for example, ketones, ethers, acetates, aromatic hydrocarbons, cyclohexanone, dimethylformamide, glycol ethers, and combinations thereof.
- Solvents for the catalyst and the inhibitor may include polar solvents.
- Lower alcohols having from 1 to 20 carbon atoms such as, for example, methanol, provide good solubility and volatility for removal from the resin matrix when prepregs are formed.
- Other useful solvents may include, for example, acetone, methyl ethyl ketone, DOWANOL PMA, N-methyl-2- pyrrolidone, dimethyl sulfoxide, dimethyl formamide, tetrahydrofuran, 1, 2- propane diol, ethylene glycol and glycerine.
- the total amount of solvent used in the curable epoxy resin composition generally may range from about 1 to about 65 weight percent in some embodiments. In other embodiments, the total amount of solvent may range from 2 to 60 weight percent; from 3 to 50 weight percent in other embodiments; and from 5 to 40 weight percent in yet other embodiments.
- Catalysts may include imidazole compounds including compounds having one imidazole ring per molecule, such as imidazole, 2- methylimidazole, 2-ethyl-4-methylimidazole, 2-undecylimidazole, 2- heptadecylimidazole, 2-phenylimidazole, 2-phenyl-4-methylimidazole, 1- benzyl-2-methylimidazole, 2-ethylimidazole, 2-isopropylimidazole, 2-phenyl-4- benzylimidazole, 1 -cyanoethyl-2-methylimidazole, 1 -cyanoethyl-2-ethyl-4- methylimidazole, 1 -cyanoethyl-2-undecylimidazole, 1 -cyanoethyl-2- isopropylimidazole,
- suitable catalysts may include amine catalysts such as N-alkylmorpholines, N-alkylalkanolamines, N 5 N- dialkylcyclohexylamines, and alkylamines where the alkyl groups are methyl, ethyl, propyl, butyl and isomeric forms thereof, and heterocyclic amines.
- Non-amine catalysts may also be used.
- Organometallic compounds of bismuth, lead, tin, titanium, iron, antimony, uranium, cadmium, cobalt, thorium, aluminum, mercury, zinc, nickel, cerium, molybdenum, vanadium, copper, manganese, and zirconium may be used.
- Illustrative examples include bismuth nitrate, lead 2-ethylhexoate, lead benzoate, ferric chloride, antimony trichloride, stannous acetate, stannous octoate, and stannous 2-ethylhexoate.
- Other catalysts that may be used are disclosed in, for example, PCT Publication No.
- suitable catalysts may include nucleophilic amines and phosphines, especially nitrogen heterocycles such as alkylated imidazoles: 2-phenyl imidazole, 2-methyl imidazole, 1 -methyl imidazole, 2- methyl-4-ethyl imidazole; other heterocycles such as diazabicycloundecene (DBU), diazabicyclooctene, hexamethylenetetramine, morpholine, piperidine; trialkylamines such as triethylamine, trimethylamine, benzyldimethyl amine; phosphines such as triphenylphosphine, tritolylphosphine, triethylphosphine; quaternary salts such as triethylammonium chloride, tetraethylammonium chloride, tetraethylammonium acetate, triphenylphosphonium acetate
- a hardener or curing agent may be provided for promoting crosslinking of the curable composition to form a thermoset composition.
- the hardeners and curing agents may be used individually or as a mixture of two or more.
- hardeners may include dicyandiamide (dicy) or phenolic curing agents such as novolacs, resoles, bisphenols.
- Other hardeners may include advanced (oligomeric) epoxy resins, some of which are disclosed above. Examples of advanced epoxy resin hardeners may include, for example, epoxy resins prepared from bisphenol A diglycidyl ether (or the diglycidyl ether of tetrabromobisphenol A) and an excess of bisphenol or (tetrabromobisphenol).
- Curing agents may also include primary and secondary polyamines and adducts thereof, anhydrides, and polyamides.
- polyfunctional amines may include aliphatic amine compounds such as diethylene triamine (D.E.H.TM 20, available from The Dow Chemical Company, Midland, Michigan), Methylene tetramine (D.E.H.TM 24, available from The Dow
- Aromatic amines such as metaphenylene diamine and diamine diphenyl sulfone, aliphatic polyamines, such as amino ethyl piperazine and polyethylene polyamine, and aromatic polyamines, such as metaphenylene diamine, diamino diphenyl sulfone, and diethyltoluene diamine, may also be used.
- Anhydride curing agents may include, for example, nadic methyl anhydride, hexahydrophthalic anhydride, trimellitic anhydride, dodecenyl succinic anhydride, phthalic anhydride, methyl hexahydrophthalic anhydride, tetrahydrophthalic anhydride, and methyl tetrahydrophthalic anhydride, among others.
- the hardener or curing agent may include a phenol-derived or substituted phenol-derived novolac or an anhydride.
- suitable hardeners include phenol novolac hardener, cresol novolac hardener, dicyclopentadiene bisphenol hardener, limonene type hardener, anhydrides, and mixtures thereof.
- the phenol novolac hardener may contain a biphenyl or naphthyl moiety.
- the phenolic hydroxy groups may be attached to the biphenyl or naphthyl moiety of the compound.
- One method of preparing a hardener containing a biphenyl moiety may be prepared by reacting phenol with bismethoxy-methylene biphenyl.
- curing agents may include dicyandiamide, boron trifluoride monoethylamine, and diaminocyclohexane. Curing agents may also include imidazoles, their salts, and adducts. These epoxy curing agents are typically solid at room temperature. Examples of suitable imidazole curing agents include but are not limited to imidazole, 2-methylimidazole, 2- propylimidazole, 4-(hydroxymethyl)imidazole, 2-phenylimidazole, 2-benzyl-4- methylimizdazole, and benzimidazole. Other curing agents include phenolic, benzoxazine, aromatic amines, amido amines, aliphatic amines, anhydrides, and phenols.
- the curing agents may be polyamides or an amino compound having a molecular weight up to 500 per amino group, such as an aromatic amine or a guanidine derivative.
- amino curing agents include 4-chlorophenyl-N,N-dimethyl-urea and 3,4-dichlorophenyl-N,N- dimethyl-urea.
- curing agents useful in embodiments disclosed herein include: 3,3'- and 4,4'-diaminodiphenylsulfone; methylenedianiline; bis(4- amino-3,5-dimethyl-phenyl)-l,4-diisopropylbenzene available as EPON 1062 from Shell Chemical Co.; and bis(4-aminophenyl)-l,4-diisopropylbenzene available as EPON 1061 from Hexion Chemical Co.
- Thiol curing agents for epoxy compounds may also be used.
- "thiol” also includes polythiol or polymercaptan curing agents.
- Illustrative thiols include aliphatic thiols such as methanedithiol, propanedithiol, cyclohexanedithiol, 2-mercaptoethyl-2,3-dimercapto-succinate, 2,3-dimercapto- l-propanol(2-mercaptoacetate), diethylene glycol bis(2-mercaptoacetate), 1,2- dimercaptopropyl methyl ether, bis(2-mercaptoethyl)ether, trimethylolpropane tris(thioglycolate), pentaerythritol tetra(mercaptopropionate), pentaerythritol tetra(thioglycolate), ethyleneglycol dithioglycolate, trimethylolpropane tris
- the curing agent may also be a nucleophilic substance such as an amine, a tertiary phosphine, a quaternary ammonium salt with a nucleophilic anion, a quaternary phosphonium salt with a nucleophilic anion, an imidazole, a tertiary arsenium salt with a nucleophilic anion, and a tertiary sulfonium salt with a nucleophilic anion.
- a nucleophilic substance such as an amine, a tertiary phosphine, a quaternary ammonium salt with a nucleophilic anion, a quaternary phosphonium salt with a nucleophilic anion, an imidazole, a tertiary arsenium salt with a nucleophilic anion, and a tertiary sulfonium salt with a nucleophilic anion.
- Aliphatic polyamines that are modified by adduction with epoxy resins, acrylonitrile, or methacrylates may also be utilized as curing agents.
- various Mannich bases can be used.
- Aromatic amines wherein the amine groups are directly attached to the aromatic ring may also be used.
- Quaternary ammonium salts with a nucleophilic anion useful as a curing agent in embodiments disclosed herein may include tetraethyl ammonium chloride, tetrapropyl ammonium acetate, hexyl trimethyl ammonium bromide, benzyl trimethyl ammonium cyanide, cetyl triethyl ammonium azide, N 5 N- dimethylpyrrolidinium isocyanate, N-methylpyrridinium phenolate, N-methyl- o-chloropyridinium chloride, methyl viologen dichloride and the like.
- the suitability of the curing agent for use herein may be determined by reference to manufacturer specifications or routine experimentation. Manufacturer specifications may be used to determine if the curing agent is an amorphous solid or a crystalline solid at the desired temperatures for mixing with the liquid or solid epoxy. Alternatively, the solid curing agent may be tested using differential scanning calorimetry (DSC) to determine the amorphous or crystalline nature of the solid curing agent and the suitability of the curing agent for mixing with the resin composition in either liquid or solid form.
- DSC differential scanning calorimetry
- the curable compositions described herein may be used in formulations that contain halogenated and non-halogenated flame retardants, including brominated and non-brominated flame retardants.
- brominated additives include tetrabromobisphenol A (TBBA) and materials derived therefrom: TBBA-diglycidyl ether, reaction products of bisphenol A or TBBA with TBBA-diglycidyl ether, and reaction products of bisphenol A diglycidyl ether with TBBA.
- Non-brominated flame retardants include the various materials derived from DOP (9,10-dihydro-9-oxa-10-phosphaphenanthrene 10-oxide) such as
- DOP-hydroquinone (10-(2',5'-dihydroxyphenyl)-9,10-dihydro-9-oxa-10- phosphaphenanthrene 10-oxide)
- condensation products of DOP with glycidyl ether derivatives of novolacs and inorganic flame retardants such as aluminum trihydrate and aluminum phosphinite.
- compositions disclosed herein may optionally include synergists, and conventional additives and fillers.
- Synergists may include, for example, magnesium hydroxide, zinc borate, and metallocenes), solvents (e.g., acetone, methyl ethyl ketone, and DOWANOLTM PMA).
- Additives and fillers may include, for example, silica, glass, talc, metal powders, titanium dioxide, wetting agents, pigments, coloring agents, mold release agents, coupling agents, ion scavengers, UV stabilizers, flexibilizing agents, and tackifying agents.
- Additives and fillers may also include fumed silica, aggregates such as glass beads, polytetrafluoroethylene, polyol resins, polyester resins, phenolic resins, graphite, molybdenum disulfide, abrasive pigments, viscosity reducing agents, boron nitride, mica, nucleating agents, and stabilizers, among others.
- Fillers may include functional or non-functional particulate fillers that may have an average particle size ranging from 5 nm to 100 microns and may include, for example, alumina trihydrate, aluminum oxide, aluminum hydroxide oxide, metal oxides, and nano tubes). Fillers and modifiers may be preheated to drive off moisture prior to addition to the epoxy resin composition. Additionally, these optional additives may have an effect on the properties of the composition, before and/or after curing, and should be taken into account when formulating the composition and the desired reaction product.
- compositions disclosed herein may include additional toughening agents.
- Toughening agents function by forming a secondary phase within the polymer matrix. This secondary phase is rubbery and hence is capable of crack growth arrestment, providing improved impact toughness.
- Toughening agents may include polysulfones, silicon-containing elastomeric polymers, polysiloxanes, and other rubber toughening agents known in the art.
- minor amounts of higher molecular weight, relatively non-volatile monoalcohols, polyols, and other epoxy- or isocyanato- reactive diluents may be used, if desired, to serve as plasticizers in the curable and thermoset compositions disclosed herein.
- isocyanates, isocyanurates, cyanate esters, allyl containing molecules or other ethylenically unsaturated compounds, and acrylates may be used in some embodiments.
- non-reactive thermoplastic resins include polyphenylsulfones, polysulfones, polyethersulfones, polyvinylidene fluoride, polyetherimide, polyphthalimide, polybenzimidazole, acrylics, phenoxy, and urethane.
- compositions disclosed herein may also include adhesion promoters such as modified organosilanes (epoxidized, methacryl, amino), acetylacetonates, and sulfur containing molecules.
- compositions disclosed herein may include wetting and dispersing aids, for example, modified organosilanes, BYK 900 series and W 9010, and modified fluorocarbons.
- compositions disclosed herein may include air release additives, for example, BYK A530, BYK A525, BYK A555, and BYK A560.
- Embodiments disclosed herein may also include surface modifiers (e.g., slip and gloss additives) and mold release agents (e.g., waxes), and other functional additives or prereacted products to improve polymer properties.
- Some embodiments may include other co-reactants that may be incorporated to obtain specific properties of the curable and electrical laminate compositions disclosed herein. Mixtures of co-reactants and/or one or more of the above described additives may also be used.
- thermosetting compositions disclosed herein may include fibrous reinforcement materials, such as continuous and/or chopped fibers.
- the fibrous reinforcement material may include glass fibers, carbon fibers, or organic fibers such as polyamide, polyimide, and polyester.
- the concentration of fibrous reinforcements used in embodiments of the thermosetting compositions may be between about 1 percent to about 95 percent by weight, based on the total weight of the composition; between about 5 percent and 90 percent by weight in other embodiments; between about 10 percent and 80 percent in other embodiments; between about 20 percent and 70 percent in other embodiments; and between 30 percent and 60 percent in yet other embodiments.
- compositions disclosed herein may include nanofillers.
- Nanofillers may include inorganic, organic, or metallic, and may be in the form of powders, whiskers, fibers, plates or films.
- the nanofillers may be generally any filler or combination of fillers having at least one dimension (length, width, or thickness) from about 0.1 to about 100 nanometers.
- the at least one dimension may be characterized as the grain size; for whiskers and fibers, the at least one dimension is the diameter; and for plates and films, the at least one dimension is the thickness.
- Clays for example, may be dispersed in an epoxy resin-based matrix, and the clays may be broken down into very thin constituent layers when dispersed in the epoxy resin under shear.
- Nanofillers may include clays, organo-clays, carbon nanotubes, nanowhiskers (such as SiC), SiO 2 , elements, anions, or salts of one or more elements selected from the s, p, d, and f groups of the periodic table, metals, metal oxides, and ceramics.
- concentration of any of the above described additives, when used in the thermosetting compositions described herein, may be between about 1 percent and 95 percent, based on the total weight of the composition; between
- Curable or hardenable compositions, or varnishes prepared therefrom disclosed herein may include at least one epoxy resin, at least one curing agent, and at least one silicone-acrylate core/shell rubber toughening agent.
- curable compositions and/or varnishes disclosed herein may additionally include a catalyst.
- curable compositions and/or varnishes disclosed herein may include a reinforcing agent. Curable compositions and/or varnishes may be formed, in some embodiments, by admixing the above components.
- curable compositions and/or varnishes may include from about 30 to about 98 volume percent epoxy resin. In other embodiments, curable compositions and/or varnishes may include 65 to 95 volume percent epoxy resin; from 70 to 90 volume percent epoxy resin in other embodiments; from 30 to 65 volume percent epoxy resin in other embodiments; and from 40 to 60 volume percent epoxy resin in yet other embodiments.
- Compositions may include from about 0.1 to about 30 volume percent of the silicone-acrylate core/shell rubber toughening agent in some embodiments.
- curable compositions may include from about 1 to about 25 volume percent silicone-acrylate core/shell rubber toughening agent; and from about 2 to about 20 volume percent silicone-acrylate core/shell rubber toughening agent in yet other embodiments.
- the amount of reinforcing material in the composition may vary depending on the type and form of the reinforcing material and the expected end product.
- Curable compositions may include from about 20 to about 70 volume percent reinforcing materials in some embodiments. In other embodiments, curable compositions may include from about 30 to about 65 volume percent reinforcing materials; and from 40 to 60 volume percent reinforcing materials in yet other embodiments.
- compositions may include from about 0.1 to about 50 volume percent optional additives in some embodiments. In other embodiments, curable compositions may include from about 0.1 to about 5 volume percent optional additives; and from 0.5 to 2.5 volume percent optional additives in yet other embodiments.
- the amount of catalyst used may vary from 0.1 to 20 parts per hundred parts epoxy resin, by weight, in some embodiments. In other embodiments, catalyst may be used in an amount ranging from 1 to 15 parts per hundred parts epoxy resin, by weight; and from 2 to 10 parts per hundred parts epoxy resin, by weight, in yet other embodiments.
- the specific amount of catalyst used for a given system should be determined experimentally to develop the optimum in properties desired.
- the specific amount of curing agent used for a given system should be determined experimentally to develop the optimum in properties desired.
- Variables to consider in selecting a curing agent and an amount of curing agent may include, for example, the epoxy resin composition (if a blend), the desired properties of the cured composition (flexibility, electrical properties, etc.), desired cure rates, as well as the number of reactive groups per catalyst molecule, such as the number of active hydrogens in an amine.
- the amount of curing agent used may vary from 0.1 to 150 parts per hundred parts epoxy resin, by weight, in some embodiments. In other embodiments, the curing agent may be used in an amount ranging from 5 to 95 parts per hundred parts epoxy resin, by weight; and the curing agent may be used in an amount ranging from 10 to 90 parts per hundred parts epoxy resin, by weight, in yet other embodiments.
- curable compositions may depend, in part, upon the properties desired in the electrical laminate composition or coating to be produced, the desired cure response of the composition, and the desired storage stability of the composition (desired shelf life).
- curable compositions may be formed by admixing the epoxidized cycloaliphatic olefin polymer, one or more epoxy resins, one or more hardeners, and other components as desired, where the relative amounts of the components may depend upon the desired properties of the electrical laminate composition.
- the epoxidized cycloaliphatic olefin polymer may be present in curable compositions disclosed herein in an amount range from 0.1 to 5 weight percent of the curable composition. In other embodiments, the epoxidized cycloaliphatic olefin polymer may be present in curable compositions disclosed herein in an amount range from 0.5 to 2.5 weight percent of the curable composition; and from about 1.0 to 2.0 weight percent of the curable composition in other embodiments.
- the epoxy resin may be present in an amount range from 0.1 to 99 weight percent of the curable composition. In other embodiments, the epoxy resin may range from 5 to 90 weight percent of the curable composition; from 10 to 80 weight percent in other embodiments; and from 10 to 50 weight percent in yet other embodiments.
- the proportions of other components may also depend, in part, upon the properties desired in the electrical laminate composition or coating to be produced.
- variables to consider in selecting curing agents and amounts of curing agents may include the epoxy composition (if a blend), the desired properties of the electrical laminate composition (T g , T d , flexibility, electrical properties (D k , D f ), etc.), desired cure rates, and the number of reactive groups per catalyst molecule, such as the number of active hydrogens in an amine.
- the amount of curing agent used may vary from 0.1 to 150 parts per hundred parts epoxy resin, by weight.
- the curing agent may be used in an amount ranging from 5 to 95 parts per hundred parts epoxy resin, by weight; and the curing agent may be used in an amount ranging from 10 to 90 parts per hundred parts epoxy resin, by weight, in yet other embodiments. In yet other embodiments, the amount of curing agent may depend on components other than the epoxy resin.
- thermoset resins formed from the above described curable compositions may have a glass transition temperature, as measured using differential scanning calorimetry, of at least 14O 0 C. In other embodiments, thermoset resins formed from the above described curable compositions may have a glass transition temperature, as measured using differential scanning calorimetry, of at least 145 0 C; at least 15O 0 C in other embodiments; at least 175 0 C in other embodiments; and at least 200 0 C in yet other embodiments.
- curable compositions described above may be disposed on or impregnated in a substrate and cured.
- substrates may include metals, such as stainless steel, iron, steel, copper, zinc, tin, aluminum, alumite and the like; alloys of such metals, and sheets which are plated with such metals and laminated sheets of such metals.
- substrates may also include polymers, glass, and various fibers, such as, for example, carbon/graphite; boron; quartz; aluminum oxide; glass such as E glass, S glass,
- fibers may include: organic fibers, such as KEVLAR; aluminum oxide-containing fibers, such as
- the substrate may be coated with a compatibilizer to improve the adhesion of the electrical laminate composition to the substrate.
- COMPOSITES AND COATED STRUCTURES COMPOSITES AND COATED STRUCTURES
- composites may be formed by curing the electrical laminate compositions disclosed herein.
- composites may be formed by applying a curable epoxy resin composition to a substrate or a reinforcing material, such as by impregnating or coating the substrate or reinforcing material, and curing the electrical laminate composition.
- the varnish After the varnish has been produced, as described above, it may be disposed on, in, or between the above described substrates, before, during, or after cure of an electrical laminate composition.
- a composite may be formed by coating a substrate with a varnish. Coating may be performed by various procedures, including spray coating, curtain flow coating, coating with a roll coater or a gravure coater, brush coating, and dipping or immersion coating.
- the substrate may be monolayer or multi-layer.
- the substrate may be a composite of two alloys, a multi-layered polymeric article, and a metal-coated polymer, among others, for example.
- one or more layers of the curable composition may be disposed on a substrate.
- Other multi-layer composites, formed by various combinations of substrate layers and electrical laminate composition layers are also envisaged herein.
- the heating of the varnish may be localized, such as to avoid overheating of a temperature-sensitive substrate, for example.
- the heating may include heating the substrate and the curable composition.
- Curing of the curable compositions and/or varnishes disclosed herein may require a temperature of at least about 3O 0 C, up to about 25O 0 C, for periods of minutes up to hours, depending on the epoxy resin, curing agent, and catalyst, if used. In other embodiments, curing may occur at a temperature of at least 100 0 C, for periods of minutes up to hours. Post- treatments may be used as well, such post-treatments ordinarily being at temperatures between about 100 0 C and 25O 0 C.
- curing may be staged to prevent undesirable temperature excursions due to reaction exotherms.
- Staging includes curing for a period of time at a temperature followed by curing for a period of time at a higher temperature.
- Staged curing may include two or more curing stages, and may commence at temperatures below about 18O 0 C in some embodiments, and below about 15O 0 C in other embodiments.
- curing temperatures may range from a lower limit of 3O 0 C, 4O 0 C, 5O 0 C, 6O 0 C, 7O 0 C, 8O 0 C, 9O 0 C, 100 0 C, HO 0 C, 12O 0 C, 13O 0 C, 14O 0 C, 15O 0 C, 16O 0 C, 17O 0 C, or 18O 0 C to an upper limit of 25O 0 C, 24O 0 C, 23O 0 C, 22O 0 C, 21O 0 C, 200 0 C, 19O 0 C, 18O 0 C, 17O 0 C, 16O 0 C, where the range may be from any lower limit to any upper limit.
- the curable compositions disclosed herein may be useful in composites containing high strength filaments or fibers such as carbon (graphite), glass, boron, and the like.
- Composites may contain from about 30% to about 70%, in some embodiments, and from 40% to 70% in other embodiments, of these fibers based on the total volume of the composite.
- Fiber reinforced composites may be formed by hot melt prepregging.
- the prepregging method is characterized by impregnating bands or fabrics of continuous fiber with a thermosetting composition as described herein in molten form to yield a prepreg, which is laid up and cured to provide a composite of fiber and epoxy resin.
- thermoset compositions formed according to embodiments disclosed herein may have a glass transition temperature, T g , as measured using differential scanning calorimetry of at least 165 0 C and a fracture toughness, ki c , as measured according to ASTM D-5045, of at least 1.0 mPa m 05 .
- the thermoset compositions may have a glass transition temperature, as measured using differential scanning calorimetry of at least 17O 0 C; 175 0 C in yet other embodiments.
- thermoset compositions formed according to embodiments disclosed herein may have a 5% decomposition temperature, T d , as measured using thermogravimetric analysis (TGA) of at least 365 0 C.
- TGA thermogravimetric analysis
- the thermoset compositions may have a T d , as measured using TGA of at least
- silicone-acrylate core/shell rubber toughening agents used in thermoset resins according to embodiments disclosed herein may result in improved flame retardancy resulting from a synergistic effect between silicone, contained in the toughening agent, and bromine, contained in an added flame retardant.
- Flammability ratings are obtained by testing under UL-94 which requires the exposure of a defined test sample of material to a defined flame for a specified period of time. Ratings of V-O, V-I, and V-2 are obtained according to a number of criteria, including flame time, afterglow time, and cotton-igniting drips.
- Thermoset resins according to embodiments disclosed herein may have UL-94 vertical burn ratings of V-O, indicating that burning stops within 10 seconds after two applications of ten seconds each of a flame to a test bar, with no flaming drips.
- the average time elapsed for the burning to stop (flame extinguishing time) during a first burn may be less than 0.9 seconds; less than 0.7 seconds in other embodiments.
- curable compositions and composites described herein may be useful as adhesives, structural and electrical laminates, coatings, marine coatings, composites, powder coatings, adhesives, castings, structures for the aerospace industry, and as circuit boards and the like for the electronics industry.
- the curable compositions and resulting thermoset resins may be used in composites, coatings, adhesives, or sealants that may be disposed on, in, or between various substrates.
- the curable compositions may be applied to a substrate to obtain an epoxy based prepreg.
- the substrates include, for example, glass cloth, a glass fiber, glass paper, paper, and similar substrates of polyethylene and polypropylene.
- the obtained prepreg may be cut into a desired size.
- An electrical conductive layer may be formed on the laminate / prepreg with an electrical conductive material.
- suitable electrical conductive materials include electrical conductive metals such as copper, gold, silver, platinum and aluminum. Such electrical laminates may be used, for example, as multi-layer printed circuit boards for electrical or electronics equipment.
- Samples and Comparative Sample are analyzed for the thermal and mechanical characterization (including differential scanning calorimetry (DSC), thermomechanical analysis (TMA), dynamic mechanical thermal analysis (DMTA), thermogravimetric analysis (TGA), and mechanical testing (fracture toughness and tensile properties)).
- DSC differential scanning calorimetry
- TMA thermomechanical analysis
- DMTA dynamic mechanical thermal analysis
- TGA thermogravimetric analysis
- mechanical testing fracture toughness and tensile properties
- DSC Differential scanning calorimetry
- T g Reported glass transition temperature
- TMA Thermomechanical analysis
- T260 is the time required for a laminate to begin to delaminate when heated to 26O 0 C.
- T288 measures the delamination time at 288 0 C.
- T260 and T288 are also determined by thermogravimetric analysis (TMA). The sample is heated to 26O 0 C and held at that temperature until such time as a measureable change in sample thickness, as a result of thermal decomposition, is detected. T288 is measured in the same way, except the sample is heated to 288 0 C.
- DMTA Dynamic mechanical thermal analysis
- ARES LS rheometer (Rheometric Scientific, Piscataway, NJ) equipped with an environmental controlled oven chamber and rectangular plate fixtures. For 1.75 inch by 0.5 inch by 0.125 inch samples, a 0.1% strain is applied at 1 Hz while ramping to 25O 0 C at 3°C/min.
- TGA Thermogravimetric analyses
- T d degradation temperatures
- Fracture toughness (k lc and G lc ) testing of the samples is performed in accordance with ASTM D-5045.
- the samples are cut using a water-jet cutter to minimize cracking and residual stress.
- a minimum of five analyses are performed and averaged.
- thermoset plaques were cut into 0.5 inch by 2.75 inch pieces with a
- Water uptake is measured by molding the sample powders (the powders of the epoxy resin compositions) to form test pieces with a thickness of 3 mm and a diameter of 50 mm. After being post-cured at 175 0 C, the test pieces are put into a constant temperature humidity chamber, which was set at a temperature of 85 0 C and a relative humidity of 85%, for 72 hours. A variation in weight is measured before and after the chamber to calculate the water uptake.
- D.E.R.TM 560 is a brominated epoxy resin of the tetrabromobisphenol A epichlorohydrin type having an epoxide equivalent weight of about 450 grams per equivalent, also available from The Dow Chemical Company, Midland, Michigan.
- ReziCureTM 3026 is a phenolic novolac hardener (epoxy curative / co-reactant) available from SI Group.
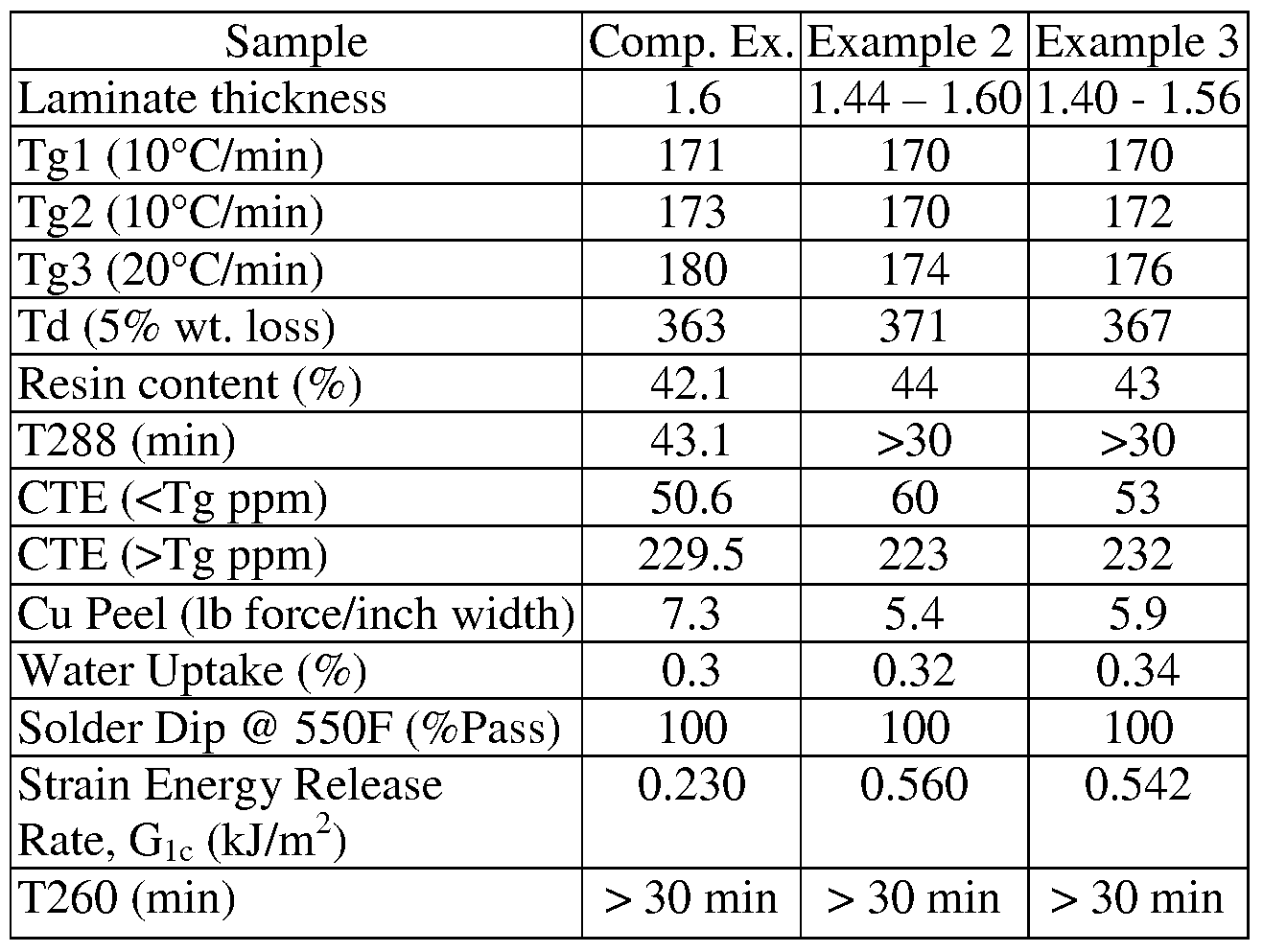
- Example 1 Properties of the laminates formed in Example 1 are compared to a control sample (Comparative Example) in Table IA.
- the Comparative Example is the same formulation described in Table 1, without the dispersed METABLEN, with the formulation of the Comparative Example given in Table IA.
- a varnish is prepared in a similar manner to that described for Example
- the toughening agent used for this example is METABLEN SX-005, a silicone-acrylate core/shell rubber available from Mitsubishi Rayon.
- Example 3 A varnish is prepared in a similar manner to that described for Example
- SX-006 is a silicone-acrylate core/shell rubber available from Mitsubishi Rayon.
- D.E.R.TM 592 is a brominated epoxy resin having an epoxide equivalent weight of about 360 grams per equivalent, available from The Dow Chemical Company, Midland, Michigan.
- Flammability test measurements (vertical burning test) are presented in
- thermoset compositions including epoxy resins and a core/shell rubber toughening agent.
- the resulting thermoset compositions may have dielectric properties suitable for use in high speed electronic parts, such as printed circuit boards. While this invention has been described in detail for the purpose of illustration, it should not be construed as limited thereby but intended to cover all changes and modifications within the spirit and scope thereof.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Materials Engineering (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Epoxy Resins (AREA)
- Laminated Bodies (AREA)
- Adhesives Or Adhesive Processes (AREA)
- Paints Or Removers (AREA)
- Macromonomer-Based Addition Polymer (AREA)
- Reinforced Plastic Materials (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/320,560 US20120095133A1 (en) | 2009-07-10 | 2010-07-08 | Core/shell rubbers for use in electrical laminate compositions |
| CN2010800309636A CN102471561A (en) | 2009-07-10 | 2010-07-08 | Core/shell rubbers for use in electrical laminate compositions |
| JP2012519713A JP2012532963A (en) | 2009-07-10 | 2010-07-08 | Core / shell rubber for use in electrical laminate compositions |
| EP10731901A EP2451872A1 (en) | 2009-07-10 | 2010-07-08 | Core/shell rubbers for use in electrical laminate compositions |
| SG2012002085A SG177615A1 (en) | 2009-07-10 | 2010-07-08 | Core/shell rubbers for use in electrical laminate compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US22457209P | 2009-07-10 | 2009-07-10 | |
| US61/224,572 | 2009-07-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011005925A1 true WO2011005925A1 (en) | 2011-01-13 |
Family
ID=42989637
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/041311 WO2011005925A1 (en) | 2009-07-10 | 2010-07-08 | Core/shell rubbers for use in electrical laminate compositions |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20120095133A1 (en) |
| EP (1) | EP2451872A1 (en) |
| JP (2) | JP2012532963A (en) |
| KR (1) | KR20120044969A (en) |
| CN (1) | CN102471561A (en) |
| SG (1) | SG177615A1 (en) |
| TW (1) | TWI600698B (en) |
| WO (1) | WO2011005925A1 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20120136094A1 (en) * | 2010-11-29 | 2012-05-31 | Li-Chun Chen | Varnish composition, and pre-impregnated manufacture thereof |
| WO2014039063A1 (en) * | 2012-09-07 | 2014-03-13 | Dow Global Technologies Llc | Toughening masterblends |
| WO2014043048A2 (en) * | 2012-09-17 | 2014-03-20 | 3M Innovative Properties Company | Powder coating epoxy compositions, methods, and articles |
| WO2014043047A2 (en) * | 2012-09-17 | 2014-03-20 | 3M Innovative Properties Company | Liquid epoxy coating compositions, methods, and articles |
| WO2014084226A1 (en) * | 2012-11-28 | 2014-06-05 | 三菱瓦斯化学株式会社 | Resin composition, prepreg, laminated plate, metal foil-clad laminated plate, and printed circuit board |
| WO2014123849A1 (en) * | 2013-02-07 | 2014-08-14 | Trillion Science, Inc. | One part epoxy resin including acrylic block copolymer |
| US20140299270A1 (en) * | 2010-11-19 | 2014-10-09 | Ppg Industries Ohio, Inc. | Adhesive compositions containing graphenic carbon particles |
| US20150252183A1 (en) * | 2012-10-19 | 2015-09-10 | Dow Global Technologies Llc | Polymer particle dispersions with epoxy hardeners |
| WO2016209864A1 (en) * | 2015-06-25 | 2016-12-29 | Dow Global Technologies Llc | Novel epoxy resin system for making carbon fiber composites |
| US10106711B2 (en) * | 2010-03-31 | 2018-10-23 | 3M Intellectual Property Company | Epoxy adhesive compositions comprising an adhesion promoter |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2385962B1 (en) * | 2009-01-06 | 2015-09-02 | Dow Global Technologies LLC | Metal stabilizers for epoxy resins and advancement process |
| US20140150970A1 (en) | 2010-11-19 | 2014-06-05 | Ppg Industries Ohio, Inc. | Structural adhesive compositions |
| RU2015118587A (en) * | 2012-10-19 | 2016-12-10 | БЛЮ КЬЮБ АйПи ЭлЭлСи | SHOCK-RESISTANT EPOXY THERMORACTOPLASTES CONTAINING RUBBERS TYPE Kernel-Shell and POLYOLS |
| WO2014149074A1 (en) * | 2013-03-15 | 2014-09-25 | Dow Global Technologies Llc | Toughened epoxy thermosets containing core shell rubbers and polyols |
| JP6251557B2 (en) * | 2013-12-05 | 2017-12-20 | デクセリアルズ株式会社 | Compound, thermosetting resin composition, and thermosetting sheet |
| CN105874014B (en) * | 2013-12-20 | 2018-05-25 | 巴斯夫涂料有限公司 | Prepare method, water soluble electrocoating, its purposes, cataphoresis painting method and the coated product of pigment paste |
| CN105196562A (en) * | 2015-08-28 | 2015-12-30 | 湖北省华昌达能新材料有限公司 | Glass fiber composite lamination flat plate |
| US10377928B2 (en) | 2015-12-10 | 2019-08-13 | Ppg Industries Ohio, Inc. | Structural adhesive compositions |
| US10351661B2 (en) | 2015-12-10 | 2019-07-16 | Ppg Industries Ohio, Inc. | Method for producing an aminimide |
| CN105754533B (en) * | 2016-03-18 | 2018-07-20 | 黑龙江省科学院石油化学研究院 | A kind of composite structure repairing high-performance epoxy adhesive and preparation method thereof |
| JP6694057B2 (en) * | 2016-03-31 | 2020-05-13 | デンカ株式会社 | Composition |
| WO2018037874A1 (en) * | 2016-08-26 | 2018-03-01 | ダイキン工業株式会社 | Powder coating material, laminate and pipe |
| EP3626777A4 (en) * | 2017-05-19 | 2021-04-07 | Kaneka Corporation | Solvent composition and production method therefor |
| JP7382937B2 (en) * | 2017-12-21 | 2023-11-17 | サイテック インダストリーズ インコーポレイテッド | Reinforced adhesive and bonding method using the same |
| CN114616303A (en) * | 2019-11-01 | 2022-06-10 | 塞特工业公司 | Primer composition for adhesive bonding and method of using the same |
| CN115477910B (en) * | 2022-09-20 | 2023-11-24 | 江苏矽时代材料科技有限公司 | Single-component high-performance insulating epoxy adhesive and preparation method and application thereof |
Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2750395A (en) | 1954-01-05 | 1956-06-12 | Union Carbide & Carbon Corp | Diepoxides |
| US2890194A (en) | 1956-05-24 | 1959-06-09 | Union Carbide Corp | Compositions of epoxides and polycarboxylic acid compounds |
| US3018262A (en) | 1957-05-01 | 1962-01-23 | Shell Oil Co | Curing polyepoxides with certain metal salts of inorganic acids |
| US4066628A (en) | 1976-08-02 | 1978-01-03 | Mitsubishi Chemical Industries Ltd. | Oxazolidone catalyst |
| DE3938157A1 (en) * | 1988-11-16 | 1990-05-17 | Sunstar Engineering Inc | DISPERSION AGENT FOR A POLYMER AND EPOXY RESIN |
| WO2000015690A1 (en) | 1998-09-10 | 2000-03-23 | The Dow Chemical Company | Polyols useful for preparing water blown rigid polyurethane foam |
| WO2003048235A1 (en) * | 2001-12-06 | 2003-06-12 | Vantico Ag | Heat-curable resin composition |
| JP2004204171A (en) * | 2002-12-26 | 2004-07-22 | Wintech Polymer Ltd | Flame-retardant resin composition for sheathed electric wire |
| JP2005281465A (en) * | 2004-03-29 | 2005-10-13 | Wintech Polymer Ltd | Flame-retardant resin composition |
| JP2006016446A (en) * | 2004-06-30 | 2006-01-19 | Toray Ind Inc | Resin composition and molded article comprised of the same |
| JP2006016447A (en) * | 2004-06-30 | 2006-01-19 | Toray Ind Inc | Resin composition and molded article comprised of the same |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6183233A (en) * | 1984-09-29 | 1986-04-26 | Sumitomo Bakelite Co Ltd | Production of epoxy reisn laminated sheet |
| JP3288185B2 (en) | 1994-10-07 | 2002-06-04 | 日立化成工業株式会社 | Epoxy resin molding material for sealing electronic parts and semiconductor device using the same |
| JPH0977844A (en) * | 1995-09-13 | 1997-03-25 | Mitsubishi Rayon Co Ltd | Epoxy resin composition and fiber-reinforced resin tubular article using the same |
| JP4092862B2 (en) * | 2000-08-10 | 2008-05-28 | 新神戸電機株式会社 | Prepreg manufacturing method, prepreg, printed wiring board and laminate |
| JP2003055533A (en) * | 2001-08-10 | 2003-02-26 | Mitsubishi Rayon Co Ltd | Epoxy resin composition for sealing |
| JP4804671B2 (en) * | 2001-08-22 | 2011-11-02 | 京セラケミカル株式会社 | Prepreg |
| JP2003231828A (en) * | 2002-02-08 | 2003-08-19 | Hitachi Chem Co Ltd | Method for producing laminate for electric wiring board and varnish for prepreg |
| JP4287162B2 (en) * | 2003-01-27 | 2009-07-01 | 株式会社カネカ | Two-component curable composition |
| WO2006019041A1 (en) * | 2004-08-18 | 2006-02-23 | Kaneka Corporation | Epoxy resin composition for semiconductor sealing agents and epoxy resin molding material |
| DE102004047708A1 (en) | 2004-09-30 | 2006-04-06 | Wacker Chemie Ag | Core-shell particle-containing composition and its preparation |
| JP4059244B2 (en) * | 2004-11-24 | 2008-03-12 | 松下電工株式会社 | Epoxy resin composition, prepreg, laminated board, printed wiring board |
| JP2006282958A (en) * | 2005-04-05 | 2006-10-19 | Shin Etsu Chem Co Ltd | Semiconductor-sealing epoxy resin composition and semiconductor device |
| WO2008102853A1 (en) * | 2007-02-23 | 2008-08-28 | Panasonic Electric Works Co., Ltd. | Epoxy resin composition, prepreg, laminates and printed wiring boards |
| DE102007024967A1 (en) * | 2007-05-30 | 2008-12-04 | Wacker Chemie Ag | Reaction resins containing core-shell particles and process for their preparation and their use |
| JP2009073933A (en) * | 2007-09-20 | 2009-04-09 | Toto Kasei Co Ltd | Epoxy resin composition having thermal degradation resistance |
| JP2009275107A (en) * | 2008-05-14 | 2009-11-26 | Nitto Denko Corp | Semiconductor-sealing epoxy resin composition and semiconductor device using the same |
| BRPI1009422B1 (en) * | 2009-03-10 | 2019-10-01 | Elastagen Pty Ltd | INJECTABLE BIOMATERIALS |
-
2010
- 2010-07-08 EP EP10731901A patent/EP2451872A1/en not_active Withdrawn
- 2010-07-08 US US13/320,560 patent/US20120095133A1/en not_active Abandoned
- 2010-07-08 CN CN2010800309636A patent/CN102471561A/en active Pending
- 2010-07-08 JP JP2012519713A patent/JP2012532963A/en active Pending
- 2010-07-08 KR KR1020127000565A patent/KR20120044969A/en not_active Application Discontinuation
- 2010-07-08 WO PCT/US2010/041311 patent/WO2011005925A1/en active Application Filing
- 2010-07-08 SG SG2012002085A patent/SG177615A1/en unknown
- 2010-07-09 TW TW099122685A patent/TWI600698B/en not_active IP Right Cessation
-
2015
- 2015-01-15 JP JP2015005457A patent/JP2015129287A/en active Pending
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2750395A (en) | 1954-01-05 | 1956-06-12 | Union Carbide & Carbon Corp | Diepoxides |
| US2890194A (en) | 1956-05-24 | 1959-06-09 | Union Carbide Corp | Compositions of epoxides and polycarboxylic acid compounds |
| US3018262A (en) | 1957-05-01 | 1962-01-23 | Shell Oil Co | Curing polyepoxides with certain metal salts of inorganic acids |
| US4066628A (en) | 1976-08-02 | 1978-01-03 | Mitsubishi Chemical Industries Ltd. | Oxazolidone catalyst |
| DE3938157A1 (en) * | 1988-11-16 | 1990-05-17 | Sunstar Engineering Inc | DISPERSION AGENT FOR A POLYMER AND EPOXY RESIN |
| WO2000015690A1 (en) | 1998-09-10 | 2000-03-23 | The Dow Chemical Company | Polyols useful for preparing water blown rigid polyurethane foam |
| WO2003048235A1 (en) * | 2001-12-06 | 2003-06-12 | Vantico Ag | Heat-curable resin composition |
| JP2004204171A (en) * | 2002-12-26 | 2004-07-22 | Wintech Polymer Ltd | Flame-retardant resin composition for sheathed electric wire |
| JP2005281465A (en) * | 2004-03-29 | 2005-10-13 | Wintech Polymer Ltd | Flame-retardant resin composition |
| JP2006016446A (en) * | 2004-06-30 | 2006-01-19 | Toray Ind Inc | Resin composition and molded article comprised of the same |
| JP2006016447A (en) * | 2004-06-30 | 2006-01-19 | Toray Ind Inc | Resin composition and molded article comprised of the same |
Non-Patent Citations (5)
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10106711B2 (en) * | 2010-03-31 | 2018-10-23 | 3M Intellectual Property Company | Epoxy adhesive compositions comprising an adhesion promoter |
| US20140299270A1 (en) * | 2010-11-19 | 2014-10-09 | Ppg Industries Ohio, Inc. | Adhesive compositions containing graphenic carbon particles |
| US9562175B2 (en) * | 2010-11-19 | 2017-02-07 | Ppg Industries Ohio, Inc. | Adhesive compositions containing graphenic carbon particles |
| US8663803B2 (en) * | 2010-11-29 | 2014-03-04 | Iteq Corporation | Varnish composition, and pre-impregnated manufacture thereof |
| US20120136094A1 (en) * | 2010-11-29 | 2012-05-31 | Li-Chun Chen | Varnish composition, and pre-impregnated manufacture thereof |
| WO2014039063A1 (en) * | 2012-09-07 | 2014-03-13 | Dow Global Technologies Llc | Toughening masterblends |
| WO2014043047A2 (en) * | 2012-09-17 | 2014-03-20 | 3M Innovative Properties Company | Liquid epoxy coating compositions, methods, and articles |
| WO2014043047A3 (en) * | 2012-09-17 | 2015-02-26 | 3M Innovative Properties Company | Liquid epoxy coating compositions, methods, and articles |
| RU2605985C2 (en) * | 2012-09-17 | 2017-01-10 | 3М Инновейтив Пропертиз Компани | Powdered epoxy coating compositions, methods and articles |
| WO2014043048A3 (en) * | 2012-09-17 | 2014-05-08 | 3M Innovative Properties Company | Powder coating epoxy compositions, methods, and articles |
| WO2014043048A2 (en) * | 2012-09-17 | 2014-03-20 | 3M Innovative Properties Company | Powder coating epoxy compositions, methods, and articles |
| US20150252183A1 (en) * | 2012-10-19 | 2015-09-10 | Dow Global Technologies Llc | Polymer particle dispersions with epoxy hardeners |
| WO2014084226A1 (en) * | 2012-11-28 | 2014-06-05 | 三菱瓦斯化学株式会社 | Resin composition, prepreg, laminated plate, metal foil-clad laminated plate, and printed circuit board |
| JPWO2014084226A1 (en) * | 2012-11-28 | 2017-01-05 | 三菱瓦斯化学株式会社 | Resin composition, prepreg, laminate, metal foil-clad laminate, and printed wiring board |
| US10178767B2 (en) | 2012-11-28 | 2019-01-08 | Mitsubishi Gas Chemical Company, Inc. | Resin composition, prepreg, laminate, metallic foil clad laminate, and printed circuit board |
| WO2014123849A1 (en) * | 2013-02-07 | 2014-08-14 | Trillion Science, Inc. | One part epoxy resin including acrylic block copolymer |
| WO2016209864A1 (en) * | 2015-06-25 | 2016-12-29 | Dow Global Technologies Llc | Novel epoxy resin system for making carbon fiber composites |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2451872A1 (en) | 2012-05-16 |
| TWI600698B (en) | 2017-10-01 |
| US20120095133A1 (en) | 2012-04-19 |
| CN102471561A (en) | 2012-05-23 |
| TW201111437A (en) | 2011-04-01 |
| JP2012532963A (en) | 2012-12-20 |
| SG177615A1 (en) | 2012-03-29 |
| JP2015129287A (en) | 2015-07-16 |
| KR20120044969A (en) | 2012-05-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20120095133A1 (en) | Core/shell rubbers for use in electrical laminate compositions | |
| US20150315432A1 (en) | Metallic compounds in non-brominated flame retardant epoxy resins | |
| EP2147034B1 (en) | Epoxy thermoset compositions comprising excess epoxy resin and process for the preparation thereof | |
| KR101318456B1 (en) | Homogeneous bismaleimide-triazine-epoxy compositions useful for the manufacture of electrical laminates | |
| KR101150311B1 (en) | Dimethylformamide-free formulations using dicyandiamide as curing agent for thermosetting epoxy resins | |
| EP2385970B1 (en) | Metal stabilizers for epoxy resins | |
| EP2229416A1 (en) | High tg epoxy systems for composite application | |
| WO2009129088A1 (en) | Epoxy-imidazole catalysts useful for powder coating applications | |
| JP2014208839A (en) | Metal compound in non-brominated frame retardant epoxy resin | |
| JP5878568B2 (en) | Metal stabilizer for epoxy resin |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 201080030963.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10731901 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 13320560 Country of ref document: US |
|
| REEP | Request for entry into the european phase |
Ref document number: 2010731901 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010731901 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012519713 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 20127000565 Country of ref document: KR Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1201000074 Country of ref document: TH |