WO2010127303A1 - Organisms for the production of isopropanol, n-butanol, and isobutanol - Google Patents
Organisms for the production of isopropanol, n-butanol, and isobutanol Download PDFInfo
- Publication number
- WO2010127303A1 WO2010127303A1 PCT/US2010/033271 US2010033271W WO2010127303A1 WO 2010127303 A1 WO2010127303 A1 WO 2010127303A1 US 2010033271 W US2010033271 W US 2010033271W WO 2010127303 A1 WO2010127303 A1 WO 2010127303A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- coa
- microbial organism
- encoded
- naturally occurring
- group
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P17/00—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms
- C12P17/16—Preparation of heterocyclic carbon compounds with only O, N, S, Se or Te as ring hetero atoms containing two or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/52—Genes encoding for enzymes or proenzymes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/0004—Oxidoreductases (1.)
- C12N9/0006—Oxidoreductases (1.) acting on CH-OH groups as donors (1.1)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/10—Transferases (2.)
- C12N9/1025—Acyltransferases (2.3)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/16—Hydrolases (3) acting on ester bonds (3.1)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/88—Lyases (4.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/93—Ligases (6)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/02—Preparation of oxygen-containing organic compounds containing a hydroxy group
- C12P7/04—Preparation of oxygen-containing organic compounds containing a hydroxy group acyclic
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P7/00—Preparation of oxygen-containing organic compounds
- C12P7/02—Preparation of oxygen-containing organic compounds containing a hydroxy group
- C12P7/04—Preparation of oxygen-containing organic compounds containing a hydroxy group acyclic
- C12P7/16—Butanols
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E50/00—Technologies for the production of fuel of non-fossil origin
- Y02E50/10—Biofuels, e.g. bio-diesel
Definitions
- the present invention relates generally to biosynthetic processes and organisms capable of producing organic compounds. More specifically, the invention relates to non-naturally occurring organisms that can produce the commodity chemicals isopropanol, w-butanol, or isobutanol.
- Isopropanol is a colorless, flammable, three-carbon alcohol that mixes completely with most solvents, including water.
- the largest use for isopropanol is as a solvent, including its well known yet small use as "rubbing alcohol,” which is a mixture of isopropanol and water.
- rubbing alcohol is a mixture of isopropanol and water.
- isopropanol is found in many everyday products such as paints, lacquers, thinners, inks, adhesives, general-purpose cleaners, disinfectants, cosmetics, toiletries, de-icers, and pharmaceuticals. Low-grade isopropanol is also used in motor oils.
- the second largest use is as a chemical intermediate for the production of isopropylamines (e.g.
- isopropylethers in agricultural products
- isopropylethers in agricultural products
- isopropyl esters is manufactured by two petrochemical routes. The predominant process entails the hydration of propylene either with or without sulfuric acid catalysis. Secondarily, isopropanol is produced via hydrogenation of acetone, which is a byproduct formed in the production of phenol and propylene oxide. High-priced propylene is currently driving costs up and margins down throughout the chemical industry motivating the need for an expanded range of low cost feedstocks.
- Isobutanol is another colorless, flammable, four carbon alcohol that is being aggressively pursued as a biofuel.
- Isobutyl acetate a common solvent used in the production of lacquer and coatings and also as a flavoring agent in the food industry.
- Isobutyl esters are used in plastics, rubbers, and other dispersions. Additional applications for isobutanol include its use as a solvent in paint, varnish removers, and inks.
- Methods for isobutanol synthesis from petroleum derived feedstocks include oxo synthesis (Weber et al., Industrial & Engineering Chemistry Research, 62:33-37 (1970)) and Guerbet condensation of methanol with w-propanol (Carlini et al., J. of Molecular Catalysis A: Chemical, 220:215-220 (2004);Carlini et al., J. of Molecular Catalysis A: Chemical, 184:273-280 (2002);Carlini et al., J. of Molecular Catalysis A: Chemical, 200:137-146 (2003);Carlini et al., J. of Molecular Catalysis A: Chemical, 206:409-418 (2003)).
- the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having an isopropanol pathway having at least one exogenous nucleic acid encoding an isopropanol pathway enzyme expressed in a sufficient amount to produce isopropanol.
- the isopropanol pathway includes an enzyme selected from the group consisting of a 4-hydroxybutyryl-CoA dehydratase, a crotonase, a 3 -hydroxybutyryl-CoA dehydrogenase, an acetoacetyl-CoA synthetase, an acetyl-CoA:acetoacetate-CoA transferase, an acetoacetyl-CoA hydrolase, an acetoacetate decarboxylase, and an acetone reductase.
- the present invention provides a method for producing w-butanol comprising culturing a non-naturally occurring microbial organism having an w-butanol pathway, under conditions and for a sufficient period of time to produce w-butanol.
- the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having an isobutanol pathway having at least one exogenous nucleic acid encoding an isobutanol pathway enzyme expressed in a sufficient amount to produce isobutanol.
- the present invention provides a method for producing isobutanol that includes culturing a non-naturally occurring microbial organism having an isobutanol pathway, under conditions and for a sufficient period of time to produce isobutanol.
- This invention is directed, in part, to non-naturally occurring microorganisms that express genes encoding enzymes that catalyze isopropanol, «-butanol, or isobutanol production.
- Pathways for the production of isopropanol, w-butanol, or isobutanol disclosed herein are based on 4- hydroxybutyryl-CoA as a starting material as shown in Figure 1.
- Successfully engineering these pathways entails identifying an appropriate set of enzymes with sufficient activity and specificity, cloning their corresponding genes into a production host, optimizing fermentation conditions, and assaying for product formation following fermentation.
- a benefit of the present invention is that it bypasses the first three steps of this traditional butanol production pathway (i.e., acetyl-CoA acetyltransferase, 3-hydroxybutyryl-CoA dehydrogenase, and crotonase) which form one molecule of crotonoyl-CoA from two acetyl-CoA molecules.
- acetyl-CoA acetyltransferase 3-hydroxybutyryl-CoA dehydrogenase
- crotonase Any or all of these enzymes represent potential bottlenecks to production. For example, although recombinant strains of E.
- the traditional Clostridial route requires that two reducing equivalents per w-butanol are extracted from the conversion of pyruvate to acetyl-CoA (i.e., two pyruvate molecules must be oxidized to two acetyl-CoA molecules per butanol produced).
- the production pathway disclosed herein needs only one pyruvate to be oxidized to acetyl-CoA per w-butanol produced.
- the additional reducing equivalent is generated by the conversion of isocitrate to alpha-ketoglutarate by isocitrate dehydrogenase.
- the theoretical yield of isobutanol via the 4-hydroxybutyryl-CoA pathway is about 1.00 mol/mol assuming glucose as the feedstock.
- One benefit of the current invention is that it bypasses the acetolactate synthase, acetohydroxy acid isomeroreductase, acetohydroxy acid dehydratase, and branched chain alpha-keto acid dehydrogenase steps of the conversion pathway from pyruvate to isobutyryl-CoA to isobutanol described in Donaldson et al., U.S 20070092957.
- the butyryl-CoA intermediate is bypassed completely by converting 4-hydroxybutyryl-CoA to 3-hydroxyisobutyryl-CoA, which is subsequently dehydrated and reduced to isobutyryl-CoA.
- this invention is also directed, in part, to methods for producing isopropanol, w-butanol, or isobutanol through culturing of these no n- naturally occurring microbial organisms.
- any of the strains disclosed herein can be cultured under appropriate conditions, for a sufficient period of time to provide the commodity chemicals isopropanol, w-butanol, or isobutanol.
- non-naturally occurring when used in reference to a microbial organism or microorganism of the invention is intended to mean that the microbial organism has at least one genetic alteration not normally found in a naturally occurring strain of the referenced species, including wild-type strains of the referenced species.
- Genetic alterations include, for example, modifications introducing expressible nucleic acids encoding metabolic polypeptides, other nucleic acid additions, nucleic acid deletions and/or other functional disruption of the microbial genetic material. Such modifications include, for example, coding regions and functional fragments thereof, for heterologous, homologous or both heterologous and homologous polypeptides for the referenced species.
- Additional modifications include, for example, non-coding regulatory regions in which the modifications alter expression of a gene or operon.
- Exemplary metabolic polypeptides include enzymes or proteins within an isopropanol, M-butanol, or isobutanol biosynthetic pathway.
- a metabolic modification refers to a biochemical reaction that is altered from its naturally occurring state. Therefore, non-naturally occurring microorganisms can have genetic modifications to nucleic acids encoding metabolic polypeptides or, functional fragments thereof. Exemplary metabolic modifications are disclosed herein.
- isolated when used in reference to a microbial organism is intended to mean an organism that is substantially free of at least one component as the referenced microbial organism is found in nature.
- the term includes a microbial organism that is removed from some or all components as it is found in its natural environment.
- the term also includes a microbial organism that is removed from some or all components as the microbial organism is found in non-naturally occurring environments. Therefore, an isolated microbial organism is partly or completely separated from other substances as it is found in nature or as it is grown, stored or subsisted in non-naturally occurring environments.
- Specific examples of isolated microbial organisms include partially pure microbes, substantially pure microbes and microbes cultured in a medium that is non-naturally occurring.
- microbial As used herein, the terms "microbial,” “microbial organism” or “microorganism” is intended to mean any organism that exists as a microscopic cell that is included within the domains of archaea, bacteria or eukarya. Therefore, the term is intended to encompass prokaryotic or eukaryotic cells or organisms having a microscopic size and includes bacteria, archaea and eubacteria of all species as well as eukaryotic microorganisms such as yeast and fungi. The term also includes cell cultures of any species that can be cultured for the production of a biochemical.
- CoA or "coenzyme A” is intended to mean an organic cofactor or prosthetic group (nonprotein portion of an enzyme) whose presence is required for the activity of many enzymes (the apoenzyme) to form an active enzyme system.
- Coenzyme A functions in certain condensing enzymes, acts in acetyl or other acyl group transfer and in fatty acid synthesis and oxidation, pyruvate oxidation and in other acetylation.
- the term "substantially anaerobic" when used in reference to a culture or growth condition is intended to mean that the amount of oxygen is less than about 10% of saturation for dissolved oxygen in liquid media.
- the term also is intended to include sealed chambers of liquid or solid medium maintained with an atmosphere of less than about 1% oxygen.
- "Exogenous” as it is used herein is intended to mean that the referenced molecule or the referenced activity is introduced into the host microbial organism.
- the molecule can be introduced, for example, by introduction of an encoding nucleic acid into the host genetic material such as by integration into a host chromosome or as non-chromosomal genetic material such as a plasmid.
- the term as it is used in reference to expression of an encoding nucleic acid refers to introduction of the encoding nucleic acid in an expressible form into the microbial organism.
- the term refers to an activity that is introduced into the host reference organism.
- the source can be, for example, a homologous or heterologous encoding nucleic acid that expresses the referenced activity following introduction into the host microbial organism. Therefore, the term “endogenous” refers to a referenced molecule or activity that is present in the host.
- the term when used in reference to expression of an encoding nucleic acid refers to expression of an encoding nucleic acid contained within the microbial organism.
- heterologous refers to a molecule or activity derived from a source other than the referenced species whereas “homologous” refers to a molecule or activity derived from the host microbial organism. Accordingly, exogenous expression of an encoding nucleic acid of the invention can utilize either or both a heterologous or homologous encoding nucleic acid.
- the no n- naturally occurring microbal organisms of the invention can contain stable genetic alterations, which refers to microorganisms that can be cultured for greater than five generations without loss of the alteration.
- stable genetic alterations include modifications that persist greater than 10 generations, particularly stable modifications will persist more than about 25 generations, and more particularly, stable genetic modifications will be greater than 50 generations, including indefinitely.
- E. coli metabolic modifications are described with reference to a suitable host organism such as E. coli and their corresponding metabolic reactions or a suitable source organism for desired genetic material such as genes for a desired metabolic pathway.
- a suitable host organism such as E. coli and their corresponding metabolic reactions or a suitable source organism for desired genetic material such as genes for a desired metabolic pathway.
- desired genetic material such as genes for a desired metabolic pathway.
- the E. coli metabolic alterations exemplified herein can readily be applied to other species by incorporating the same or analogous encoding nucleic acid from species other than the referenced species.
- Such genetic alterations include, for example, genetic alterations of species homologs, in general, and in particular, orthologs, paralogs or nonorthologous gene displacements.
- ortholog is a gene or genes that are related by vertical descent and are responsible for substantially the same or identical functions in different organisms.
- mouse epoxide hydrolase and human epoxide hydrolase can be considered orthologs for the biological function of hydrolysis of epoxides.
- Genes are related by vertical descent when, for example, they share sequence similarity of sufficient amount to indicate they are homologous, or related by evolution from a common ancestor.
- Genes can also be considered orthologs if they share three-dimensional structure but not necessarily sequence similarity, of a sufficient amount to indicate that they have evolved from a common ancestor to the extent that the primary sequence similarity is not identifiable.
- Genes that are orthologous can encode proteins with sequence similarity of about 25% to 100% amino acid sequence identity.
- Genes encoding proteins sharing an amino acid similarity less that 25% can also be considered to have arisen by vertical descent if their three-dimensional structure also shows similarities.
- Members of the serine protease family of enzymes, including tissue plasminogen activator and elastase, are considered to have arisen by vertical descent from a common ancestor.
- Orthologs include genes or their encoded gene products that through, for example, evolution, have diverged in structure or overall activity. For example, where one species encodes a gene product exhibiting two functions and where such functions have been separated into distinct genes in a second species, the three genes and their corresponding products are considered to be orthologs. For the production of a biochemical product, those skilled in the art will understand that the orthologous gene harboring the metabolic activity to be introduced or disrupted is to be chosen for construction of the non-naturally occurring microorganism.
- An example of orthologs exhibiting separable activities is where distinct activities have been separated into distinct gene products between two or more species or within a single species.
- a specific example is the separation of elastase proteolysis and plasminogen proteolysis, two types of serine protease activity, into distinct molecules as plasminogen activator and elastase.
- a second example is the separation of mycoplasma 5 '-3' exo nuclease and Drosophila DNA polymerase III activity.
- the DNA polymerase from the first species can be considered an ortholog to either or both of the exo nuclease or the polymerase from the second species and vice versa.
- paralogs are homologs related by, for example, duplication followed by evolutionary divergence and have similar or common, but not identical functions.
- Paralogs can originate or derive from, for example, the same species or from a different species.
- microsomal epoxide hydrolase epoxide hydrolase I
- soluble epoxide hydrolase epoxide hydrolase II
- Paralogs are proteins from the same species with significant sequence similarity to each other indicating that they are homologous, or related through co-evolution from a common ancestor.
- Groups of paralogous protein families include HipA homologs, luciferase genes, peptidases, and others.
- a nonorthologous gene displacement is a nonorthologous gene from one species that can substitute for a referenced gene function in a different species. Substitution includes, for example, being able to perform substantially the same or a similar function in the species of origin compared to the referenced function in the different species.
- a nonorthologous gene displacement will be identifiable as structurally related to a known gene encoding the referenced function, less structurally related but functionally similar genes and their corresponding gene products nevertheless will still fall within the meaning of the term as it is used herein.
- Functional similarity requires, for example, at least some structural similarity in the active site or binding region of a nonorthologous gene product compared to a gene encoding the function sought to be substituted. Therefore, a nonorthologous gene includes, for example, a paralog or an unrelated gene.
- Orthologs, paralogs and nonorthologous gene displacements can be determined by methods well known to those skilled in the art. For example, inspection of nucleic acid or amino acid sequences for two polypeptides will reveal sequence identity and similarities between the compared sequences. Based on such similarities, one skilled in the art can determine if the similarity is sufficiently high to indicate the proteins are related through evolution from a common ancestor. Algorithms well known to those skilled in the art, such as Align, BLAST, Clustal W and others compare and determine a raw sequence similarity or identity, and also determine the presence or significance of gaps in the sequence which can be assigned a weight or score. Such algorithms also are known in the art and are similarly applicable for determining nucleotide sequence similarity or identity.
- Parameters for sufficient similarity to determine relatedness are computed based on well known methods for calculating statistical similarity, or the chance of finding a similar match in a random polypeptide, and the significance of the match determined.
- a computer comparison of two or more sequences can, if desired, also be optimized visually by those skilled in the art.
- Related gene products or proteins can be expected to have a high similarity, for example, 25% to 100% sequence identity. Proteins that are unrelated can have an identity which is essentially the same as would be expected to occur by chance, if a database of sufficient size is scanned (about 5%). Sequences between 5% and 24% may or may not represent sufficient homology to conclude that the compared sequences are related. Additional statistical analysis to determine the significance of such matches given the size of the data set can be carried out to determine the relevance of these sequences.
- Exemplary parameters for determining relatedness of two or more sequences using the BLAST algorithm can be as set forth below.
- amino acid sequence alignments can be performed using BLASTP version 2.0.8 (Jan-05-1999) and the following parameters: Matrix: 0 BLOSUM 62; gap open: 11; gap extension: 1; x_dropoff: 50; expect: 10.0; wordsize: 3; filter: on.
- Nucleic acid sequence alignments can be performed using BLASTN version 2.0.6 (Sept-16-1998) and the following parameters: Match: 1 ; mismatch: -2; gap open: 5; gap extension: 2; x_dropoff: 50; expect: 10.0; wordsize: 11; filter: off.
- Those skilled in the art will know what modifications can be made to the above parameters to either increase or decrease the stringency of the comparison, for example, and determine the relatedness of two or more sequences.
- the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having an isopropanol pathway having at least one exogenous nucleic acid encoding an isopropanol pathway enzyme expressed in a sufficient amount to produce isopropanol.
- the isopropanol pathway includes an enzyme selected from the group consisting of a 4-hydroxybutyryl-CoA dehydratase, a crotonase, a 3 -hydroxybutyryl-CoA dehydrogenase, an acetoacetyl-CoA synthetase, an acetyl-CoA:acetoacetate-CoA transferase, an acetoacetyl-CoA hydrolase, an acetoacetate decarboxylase, and an acetone reductase.
- the microbial organism includes two exogenous nucleic acids, each encoding an isopropanol pathway enzyme, while in other embodiments the microbial organism includes three exogenous nucleic acids, each encoding an isopropanol pathway enzyme. In some embodiments, the microbial organism includes four exogenous nucleic acids, each encoding an isopropanol pathway enzyme. In further embodiments, the microbial organism includes five exogenous nucleic acids, each encoding an isopropanol pathway enzyme. In yet further embodiments, the microbial organism includes six exogenous nucleic acids, each encoding an isopropanol pathway enzyme.
- the microbial organism can also include seven exogenous nucleic acids, each encoding an isopropanol pathway enzyme. Finally, the microbial organism can include eight exogenous nucleic acids, each encoding an isopropanol pathway enzyme. Any of the aforementioned genes that are inserted into the host organism can be a heterologous nucleic acid. In some embodiments, the non-naturally occurring microbial organism is in a substantially anaerobic culture medium.
- the present invention provides a 4-hydroxybutyryl-CoA to isopropanol pathway that provides a nucleic acid encoding an enzyme that carries out the dehydration of 4-hydroxybutyryl-CoA to form crotonoyl-CoA as shown in step A of Figure 1.
- Crotonase subsequently hydrates crotonoyl-CoA to 3-hydroxybutyryl-CoA (step B) which, in turn, is oxidized to acetoacetyl-CoA by 3 -hydro xybutyryl-CoA dehydrogenase (step C).
- Acetoacetyl-CoA is converted to acetoacetate by a synthetase, transferase, or hydrolase (steps D, E, or F).
- the final two steps involve the decarboxylation of acetoacetate to form acetone (step G) and its subsequent reduction to isopropanol (step H).
- the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having a w-butanol pathway having at least one exogenous nucleic acid encoding a w-butanol pathway enzyme expressed in a sufficient amount to produce w-butanol.
- the w-butanol pathway includes an enzyme selected from the group consisting of a 4-hydroxybutyryl-CoA dehydratase, a crotonoyl-CoA reductase, a butyryl-CoA reductase (aldehyde forming), a butyraldehyde reductase, and a butyryl-CoA reductase (alcohol forming).
- the microbial organism includes two exogenous nucleic acids, each encoding an w-butanol pathway enzyme, while in other embodiments, the microbial organism includes three exogenous nucleic acids, each encoding an w-butanol pathway enzyme. In further embodiments, the microbial organism includes four exogenous nucleic acids, each encoding an M-butanol pathway enzyme. Any of the aforementioned nucleic acids can be provided as a heterologous nucleic acid. Such non-naturally occurring microbial organism can be grown in a substantially anaerobic culture medium.
- the invention provides a non-naturally occurring microbial organism having a w-butanol pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of 4-hydroxybutyryl-CoA to crotonoyl- CoA, crotonoyl-CoA to butyryl-CoA, butyryl-CoA to w-butanol, butyryl-CoA to butyraldehyde, and butyraldehyde to w-butanol.
- the 4-hydroxybutyryl-CoA to w-butanol pathway begins with the dehydration of 4- hydroxybutyryl-CoA to crotonoyl-CoA as shown in step A of Figure 1, which is then reduced to butyryl-CoA (step I).
- Butyryl-CoA then undergoes two reductions carried out either by two separate enzymes, steps J and K, or a single dual-function enzyme as shown in step L.
- the present invention provides a non-naturally occurring microbial organism that includes a microbial organism having an isobutanol pathway having at least one exogenous nucleic acid encoding an isobutanol pathway enzyme expressed in a sufficient amount to produce isobutanol.
- the isobutanol pathway includes an enzyme selected from the group consisting of a 4-hydroxybutyryl-CoA dehydratase, a crotonoyl-CoA reductase, an isobutyryl-CoA mutase, a 4-hydroxybutyryl-CoA mutase, a 3-hydroxyisobutyryl-CoA dehydratase, a methacrylyl-CoA-reductase, an isobutyryl-CoA reductase (aldehyde forming), an isobutyraldehyde reductase, and an isobutyryl-CoA reductase (alcohol forming).
- the microbial organism includes two exogenous nucleic acids, each encoding an isobutanol pathway enzyme, while in other embodiments, the microbial organism includes three exogenous nucleic acids, each encoding an isobutanol pathway enzyme. In other embodiments, the microbial organism includes four exogenous nucleic acids, each encoding an isobutanol pathway enzyme. In still further embodiments, the microbial organism includes five exogenous nucleic acids, each encoding an isobutanol pathway enzyme. Any of the exogenous nucleic acid can be a heterologous nucleic acid. Such non-naturally occurring microbial organism can be grown in a substantially anaerobic culture medium.
- the non-naturally occurring organism has a set of isobutanol pathway enzymes that includes a 4-hydroxybutyryl-CoA dehydratase, a crotonoyl-CoA reductase, an isobutyryl-CoA mutase, an isobutyryl-CoA reductase (aldehyde forming), and an isobutyraldehyde reductase.
- Such organisms can have one, two three, four, five, up to all nucleic acids encoding isobutanol pathway enzymes provided as exogenous nucleic acids.
- the non-naturally occurring organism has a set of isobutanol pathway enzymes comprises a 4-hydroxybutyryl-CoA dehydratase, a crotonoyl-CoA reductase, an isobutyryl-CoA mutase, and an isobutyryl-CoA reductase (alcohol forming).
- Such organisms can have one, two three, four, up to all nucleic acids encoding isobutanol pathway enzymes provided as exogenous nucleic acids.
- the non-naturally occurring organism has a set of isobutanol pathway enzymes comprises a 4-hydroxybutyryl-CoA mutase, a 3 -hydro xyisobutyryl-CoA dehydratase, a methacrylyl-CoA-reductase, an isobutyryl-CoA reductase (aldehyde forming), and an isobutyraldehyde reductase.
- Such organisms can have one, two three, four, five, up to all nucleic acids encoding isobutanol pathway enzymes provided as exogenous nucleic acids.
- the non-naturally occurring organism has a set of isobutanol pathway enzymes comprises a 4-hydroxybutyryl-CoA mutase, a 3 -hydro xyisobutyryl-CoA dehydratase, a methacrylyl-CoA-reductase, and an isobutyryl-CoA reductase (alcohol forming).
- Such organisms can have one, two three, four, up to all nucleic acids encoding isobutanol pathway enzymes provided as exogenous nucleic acids.
- the invention provides a non-naturally occurring microbial organism having an isobutanol pathway, wherein the non-naturally occurring microbial organism comprises at least one exogenous nucleic acid encoding an enzyme or protein that converts a substrate to a product selected from the group consisting of 4-hydroxybutyryl-CoA to crotonoyl-CoA, crotonoyl-CoA to butyryl-CoA, butyryl-CoA to isobutyryl-CoA, 4- hydroxybutyryl-CoA to 3-hydroxyisobutyryl-CoA, 3-hydroxyisobutyryl-CoA to methacrylyl- CoA, methacrylyl-CoA to isobutyryl-CoA, isobutyryl-CoA to isobutanol, isobutyryl-CoA to isobutyraldehyde, and isobutryaldehyde to isobutanol.
- Isobutyryl-CoA can be formed from 4-hydroxybutyryl-CoA via a dehydration, reduction, and carbon backbone rearrangement as shown in steps A, I, and M, of Figure 1.
- this intermediate can be obtained via first carbon backbone rearrangement, then dehydration and reduction as shown in steps Q, R, and S of Figure 1.
- Isobutyryl-CoA then undergoes two reductions to form isobutanol.
- the reductions are carried out either by two enzymes, steps N and O or a single dual- function enzyme as shown in step P.
- the invention is described herein with general reference to the metabolic reaction, reactant or product thereof, or with specific reference to one or more nucleic acids or genes encoding an enzyme associated with or catalyzing, or a protein associated with, the referenced metabolic reaction, reactant or product. Unless otherwise expressly stated herein, those skilled in the art will understand that reference to a reaction also constitutes reference to the reactants and products of the reaction. Similarly, unless otherwise expressly stated herein, reference to a reactant or product also references the reaction, and reference to any of these metabolic constituents also references the gene or genes encoding the enzymes that catalyze or proteins involved in the referenced reaction, reactant or product.
- Table 1 shows the enzyme types useful to convert common central metabolic intermediates into isopropanol, «-butanol, or isobutanol.
- the first three digits of each label correspond to the first three Enzyme Commission number digits which denote the general type of transformation independent of substrate specificity.
- Exemplary genes encoding enzymes that catalyze the conversion of an aldehyde to alcohol include air A encoding a medium- chain alcohol dehydrogenase for C2-C14 (Tani et al, Appl. Environ. Microdot, 66:5231-5235 (2000)), ADH2 from Saccharomyces cerevisiae (Atsumi et al., Nature, 451 :86-89 (2008)), jg/zD from E. coli which has preference for molecules longer than C3 (Sulzenbacher et al., J.
- ADHl from Zymomonas mobilis has been demonstrated to have activity on a number of aldehydes including formaldehyde, acetaldehyde, propionaldehyde, butyraldehyde, and acrolein (Kinoshita et al., Appl Microbiol Biotechnol, 22:249-254 (1985)).
- Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 2.
- Step C catalyzing the formation of acetoacetyl-CoA from 3-hydroxybutyryl-CoA participates in the acetyl-CoA fermentation pathway to butyrate in several species of Clostridia and has been studied in detail (Jones and Woods, Microbiol. Rev. 50:484-524 (1986).
- coli encoded by fadB and fadJ, function as 3-hydroxyacyl-CoA dehydrogenases (Binstock and Schulz, Meth. Enzymol. 71 Pt C, 403-411 (1981).
- Yet other genes demonstrated to catalyze this reversible transformation are phbB from Zoogloea ramigera (Ploux et al., Eur. J. Biochem. 174:177-182 (1988) and phaB from Rhodobacter sphaeroides (Alber et al., MoI. Microbiol. 61 :297-309 (2006).
- the former gene is NADPH-dependent, its nucleotide sequence has been determined (Peoples and Sinskey, MoI.
- alcohol dehydrogenases that convert a ketone to a hydroxyl functional group.
- Two such enzymes from E. coli are encoded by malate dehydrogenase (mdh) and lactate dehydrogenase (idhA).
- mdh malate dehydrogenase
- idhA lactate dehydrogenase
- lactate dehydrogenase from Ralstonia eutropha has been shown to demonstrate high activities on substrates of various chain lengths such as lactate, 2-oxobutyrate, 2-oxopentanoate and 2-oxoglutarate Steinbuchel et al., Eur. J. Biochem., 130:329-334 (1983)).
- Transformations in Figure 1 also rely on the two-step reduction of acyl-CoA to the corresponding alcohol.
- step L in the butanol pathway and step P in the isobutanol pathway rely on this transformation.
- Exemplary two-step oxidoreductases that convert an acyl- CoA to alcohol include those that transform substrates such as acetyl-CoA to ethanol (e.g., adhE irom E. coli (Kessler et al., FEBS. Lett., 281 :59-63 (1991)) and butyryl-CoA to butanol (e.g. adhE2 from C acetobutylicum (Fontaine et al., J.
- Another exemplary enzyme can convert malonyl-CoA to 3 -HP.
- An NADPH-dependent enzyme with this activity has characterized in Chloroflexus aurantiacus where it participates in the 3- hydroxypropionate cycle (Hugler et al., J. Bacteriol, 184:2404-2410 (2002); Strauss et al., Eur. J. Biochem., 215:633-643 (1993)).
- This enzyme with a mass of 300 kDa, is highly substrate- specific and shows little sequence similarity to other known oxidoreductases (Hugler et al., supra).
- Enzymes in other organisms including Roseiflexus castenholzii, Erythrobacter sp. NAPl and marine gamma proteobacterium HTCC2080 can be inferred by sequence similarity. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 9.
- acyl-CoA molecules can be reduced by enzymes such as the jojoba (Simmondsia chinensis) FAR which encodes an alcohol- forming fatty acyl-CoA reductase. Its overexpression in E. coli resulted in FAR activity and the accumulation of fatty alcohol (Metz et al., Plant Physiology, 122:635-644 (2000)) (FAR, AAD38039.1, Simmondsia chinensis).
- jojoba Simmondsia chinensis
- the pathways disclosed herein also involve oxidoreductase-type transformations that convert an acyl-CoA to an aldehyde.
- Steps J and N catalyze the reduction of butytyl-CoA to butyraldehyde and isobutyryl-CoA to isobutyraldehyde respectively.
- acyl-CoA dehydrogenases are capable of reducing an acyl-CoA to its corresponding aldehyde.
- genes that encode such enzymes include the Acinetobacter calcoaceticus acrl encoding a fatty acyl-CoA reductase (Reiser et al., Journal of Bacteriology , 179:2969-2975 (1997)), the Acinetobacter sp. M-I fatty acyl-CoA reductase (Ishige et al., Environ. Microbiol, 68:1192- 1195 (2002)), and a CoA- and NADP- dependent succinate semialdehyde dehydrogenase encoded by the sucD gene in Clostridium kluyveri (Sohling et al., J. Bacteriol. 178:871-880 (1996)).
- SucD of P. gingivalis is another succinate semialdehyde dehydrogenase (Takahashi et al., J. Bacteriol 182:4704-4710 (2000)).
- the enzyme acylating acetaldehyde dehydrogenase in Pseudomonas sp, encoded by bphG, is yet another enzyme demonstrated to oxidize and acylate acetaldehyde, propionaldehyde, butyraldehyde, isobutyraldehyde and formaldehyde (Powlowski et al., J. Bacteriol. 175:377-385 (1993).
- malonyl-CoA reductase which transforms malonyl-CoA to malonic semialdehyde.
- Malonyl-CoA reductase is a key enzyme in autotrophic carbon fixation via the 3-hydroxypropionate cycle in thermoacidophilic archaeal bacteria (Berg et al., Science 318:1782-1786 (2007); Thauer Science 318: 1732-1733 (2007)).
- the enzyme utilizes NADPH as a cofactor and has been characterized in Metallosphaera and Sulfolobus spp (Alber et al., supra; Hugler et al., J.
- the enzyme is encoded by Msed_0709 in Metallosphaera sedula (Alber et al., supra; Berg et al., supra).
- a gene encoding a malonyl-CoA reductase from Sulfolobus tokodaii was cloned and heterologously expressed in E. coli ((Alber et al., supra). This enzyme has also been shown to catalyze the conversion of methylmalonyl-CoA to its corresponding aldehyde.
- aldehyde dehydrogenase functionality of these enzymes is similar to the bifunctional dehydrogenase from Chloroflexus aurantiacus, there is little sequence similarity.
- malonyl-CoA reductase enzymes have high sequence similarity to aspartate-semialdehyde dehydrogenase, an enzyme catalyzing the reduction and concurrent dephosphorylation of aspartyl-4-phosphate to aspartate semialdehyde. Additional genes can be found by sequence homology to proteins in other organisms including Sulfolobus solfataricus and Sulfolobus acidocaldarius and have been listed below.
- Yet another enzyme for CoA-acylating aldehyde dehydrogenase is the aid gene from Clostridium beijerinckii (Toth et al., Appl. Environ
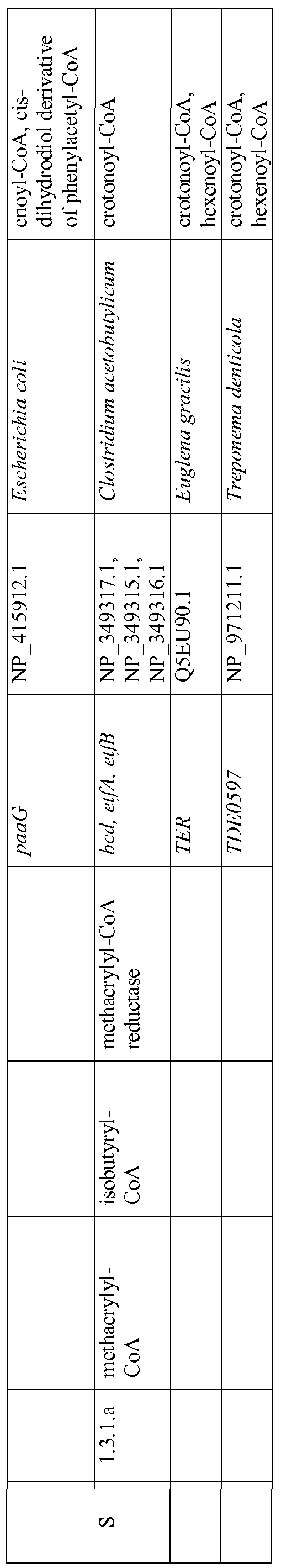
- step I refers to the conversion of crotonyl-CoA to butyryl-CoA by crotonyl-CoA reductase and step S refers to the conversion of methacryl-CoA to isobutyryl-CoA by methacrylyl-CoA reductase.
- Enoyl-CoA reductase enzymes are enzymes that can carry out either step.
- One exemplary enoyl-CoA reductase is the gene product o ⁇ bcd from C. acetobutylicum (Boynton et al., J. Bacteriol. 178:3015-3024 (1996); Atsumi et al., Metab. Eng.
- enoyl-CoA reductase is the mitochondrial enoyl-CoA reductase from E. gracilis (Hoffmeister et al., J. Biol. Chem. 280:4329-4338 (2005)). A construct derived from this sequence following the removal of its mitochondrial targeting leader sequence was cloned in E. coli resulting in an active enzyme (Hoffmeister et al., supra).
- Isobutyryl-CoA dehydrogenase is another enzyme for step S of Figure 1, though it naturally catalyzes the oxidation of isobutyryl-CoA to methacrylyl-CoA.
- the crystal structure of the human isobutyryl-CoA dehydrogenase with and without the bound substrate has been determined (Battaile et al., J. Biol. Chem. 279: 16526-16534 (2004)).
- Additional isobutyryl- CoA dehydrogenases from Mus musculus and Rhodopseudomonas palustris can be inferred by sequence similarity. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 13.
- 2-enoate reductases (EC 1.3.1.31) that are known to catalyze the NADH-dependent reduction of a wide variety of ⁇ , ⁇ -unsaturated carboxylic acids and aldehydes (Rohdich et al., J. Biol. Chem. 276:5779-5787 (2001)).
- 2-enoate reductase is encoded by enr in several species of Clostridia (Giesel, et al. Arch. Microbiol. 135:51-57 (1983)) including C tyrobutyricum, and C thermoaceticum (now called Moorella thermoaceticum) (Rohdich et al., supra).
- thermoaceticum enr gene has also been expressed in an enzymatically active form in E. coli (Rohdich et al., supra).
- GenBank accession numbers shown in Table 14.
- Additional exemplary transferase transformations are catalyzed by the gene products oicatl, cat2, and cat3 of Clostridium kluyveri which have been shown to exhibit succinyl-CoA, 4- hydroxybutyryl-CoA, and butyryl-CoA acetyltransferase activity, respectively (Aberthart et al., J. Chem. Soc. 6: 1404-1406 (1979); Agnihotri et al. Med. Chem. 11 :9-20 (2003)).
- Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 16.
- the genes encoding this enzyme are gctA and gctB.
- This enzyme has reduced but detectable activity with other CoA derivatives including glutaryl-CoA, 2-hydroxyglutaryl-CoA, adipyl-CoA and acrylyl-CoA (Buckel et al., supra).
- the enzyme has been cloned and expressed in is. coli (Mack et al., Eur. J. Biochem. 226:41-51 (1994)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 17.
- Additional enzymes capable of converting acetoacetyl-CoA to acetoacetate include succinyl- CoA:3-ketoacid CoA transferases which utilize succinate as the CoA acceptor.
- succinyl-CoA:3:ketoacid-CoA transferases are present in Helicobacter pylori (Corthesy-Theulaz et al., J. Biol. Chem. 272:25659-25667 (1997)) and Bacillus subtilis (Stols et al., Protein. Expr. Purif 53:396-403 (2007)).
- GenBank accession numbers shown in Table 18.
- Step F of Figure 1 refers to the conversion of acetoacetyl-CoA to acetoacetate by acetoacetyl- CoA hydrolase.
- Such activity has been detected in Rattus norvegicus (Patel et al., Biochem. J. Biochem. J. 176:951-958 (1978)), Bos taurus (Drummond et al., J. Biol. Chem. 235:318-325 (I960)), and Homo sapiens (Rous, Biochem. Biophys. Res. Commun. 69:74-78, (1976)), although the gene sequences encoding the corresponding enzymes are not known.
- acetyl-CoA hydrolases (EC 3.1.2.1) have broad substrate specificity and thus represent suitable enzymes for hydrolyzing acetoacetyl-CoA.
- the enzyme from Rattus norvegicus brain, acotl2 (NP_570103.1) (Robinson, Jr. et al., Biochem. Biophys. Res. Comm. 71 :959-965 (1976)) can react with butyryl-CoA, hexanoyl-CoA and malonyl-CoA.
- Additional hydrolase enzymes include 3-hydroxyisobutyryl-CoA hydrolase which has been described to efficiently catalyze the conversion of 3 -hydroxyisobutyryl-CoA to 3- hydroxyisobutyrate during valine degradation (Shimomura et al., J. Biol. Chem. 269:14248- 14253 (1994)). Genes encoding this enzyme include hibch of Rattus norvegicus (Shimomura supra; Shimomura et al, 2000) and Homo sapiens (Shimomura et al., Methods Enzymol. 324:229-240 (2000)). Genes identified by sequence homology include hibch of Saccharomyces cerevisiae and BCJ2292 of Bacillus cereus. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 19.
- Yet another hydrolase is the human dicarboxylic acid thioesterase, acot8, which exhibits activity on glutaryl-CoA, adipyl-CoA, suberyl-CoA, sebacyl-CoA, and dodecanedioyl-CoA (Westin et al., J. Biol. Chem. 280:38125-38132 (2005)) and the closest E. coli homolog, tesB, which can also hydrolyze a broad range of CoA thiolesters (Naggert et al., J. Biol. Chem. 266:11044-11050 (1991)).
- a similar enzyme has also been characterized in the rat liver (Deana, Biochem. Int. 26:767-773 (1992)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 20.
- E. coli thiolester hydrolases include the gene products of tesA (Bonner et al., J. Biol. Chem. 247:3123-3133 (1972)), ybgC (Kuznetsova et al., FEMS Microbiol. Rev. 29:263- 279 (2005); Zhuang et al., FEBS Lett. 516:161-163 (2002)), paal (Song et al., J. Biol. Chem. 281 : 11028-11038 (2006)), and jM5 (Leduc et al., J. Bacteriol. 189: 7112-7126 (2007)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 21.
- step G acetoacetate is decarboxylated to form acetone.
- This reaction can be catalyzed by acetoacetate decarboxylase (EC 4.1.1.4), an enzyme studied for its role in bacterial solventogenesis.
- acetoacetate decarboxylase EC 4.1.1.4
- Exemplary bacterial enzymes have been characterized from Clostridium acetobutylicum (Benner et al., J. Am. Chem. So. 103:993-994 (1981); HIghbarger et al., Biochemistry 35:41-46 (1996); Petersen et al., Appl. Environ. Microbiol. 56:3491-3498 (1990); Rozzel et al. J. Am. Chem.
- Clostridium beijerinckii Clostridium beijerinckii (Ravagnani et al. MoI. Microbiol. 37:1172-1185 (2000)). Acetoacetate decarboxylase activity has also been demonstrated in Pseudomonas putida and Bacillus polymyxa but genes are not associated with this activity to date (Matiasek et al., Curr. Microbiol. 42: 276-281 (2001)). Bacterial genes in other organisms such as Clostridium botulinum and Bacillus amyloliquefaciens FZB42 can be identified by sequence homology.
- acetoacetate decarboxylase catalyzes the final step of the ketone-body pathway (Kalapos, Biochim. Biophys. Acta 1621 : 122- 139 (2003)), but genes associated with this activity have not been identified to date. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 22.
- decarboxylase enzymes include pyruvate decarboxylase (EC 4.1.1.1) and benzoylformate decarboxylase (EC 4.1.1.7).
- Pyruvate decarboxylase (PDC) also termed keto- acid decarboxylase, is a key enzyme in alcoholic fermentation, catalyzing the decarboxylation of pyruvate to acetaldehyde.
- the enzyme from Saccharomyces cerevisiae has a broad substrate range for aliphatic 2-keto acids including 2-ketobutyrate, 2-ketovalerate, 3-hydroxypyruvate and 2-phenylpyruvate (Li et al., Biochemistry 38:10004-10012 (1999)).
- benzoylformate decarboxylase (EC 4.1.1.7) has a broad substrate range and has been the target of enzyme engineering studies.
- the enzyme from Pseudomonas putida has been extensively studied and crystal structures of this enzyme are available (Polovnikova et al., Biochemistry 42: 1820-1830 (2003); Hasson et al., Biochemistry 37:9918-9930 (1998)).
- Site- directed mutagenesis of two residues in the active site of the Pseudomonas putida enzyme altered the affinity (Km) of naturally and non-naturally occuring substrates (Siegert et al., Protein Eng. Des. SeI, 18:345-357 (2005)).
- This enzyme has been further modified by directed engineering (Lingen et al., Chembiochem 4:721-726 (2003); Lingen et al., Protein Eng. 15:585-593 (2002)).
- a third enzyme capable of decarboxylating 2-oxoacids is alpha-ketoglutarate decarboxylase (KGD).
- the substrate range of this class of enzymes has not been studied to date.
- the KDC from Mycobacterium tuberculosis (Tian et al., Proc. Natl. Acad. Sci. U.S.A. 102:10670-10675 (2005)) has been cloned and has been functionally expressed in E. coli at Genomatica.
- KDC enzyme activity has been detected in several species oiRhizobia including Bradyrhizobium japonicum and Mesorhizobium loti (Green et al., J. Bacteriol. 182:2838-2844 (2000)).
- MTYKAPVKDVKFLLDKVFKV (Shigeoka et al., supra).
- the gene can be identified by testing genes containing this w-terminal sequence for KDC activity. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 25.
- step B Hydration of crotonyl-CoA to form 3-hydroxybutyryl-CoA (step B, Figure 1) is catalyzed by a crotonase (EC 4.2.1.55).
- crotonase EC 4.2.1.55
- These enzymes are part of the pathways for w-butanol formation in some organisms, particularly Clostridial species, and also comprise one step of the 3- hydroxypropionate/4-hydroxybutyrate cycle in thermoacidophilic Archaea of the genera Sulfolobus, Acidianus, and Metallosphaera.
- Exemplary genes encoding crotonase enzymes can be found in C acetobutylicum (Boynton et al., Journal of Bacteriology 178:3015-3024 (1996)), C kluyveri (Hillmer et al., FEBS Lett. 21 :351-354 (1972)), and Metallosphaera sedula (Berg et al., Archaea. Science. 318:1782-1786 (2007)) though the sequence of the latter gene is not known.
- Enoyl-CoA hydratases which are involved in fatty acid beta-oxidation and/or the metabolism of various amino acids, can also catalyze the hydration of crotonyl-CoA to form 3- hydroxybutyryl-CoA (Agnihotri et al., Med. Chem., 11 :9-20 (2003); Conrad et al., J Bacteriol. 118: 103-111 (1974)).
- the enoyl-CoA hydratases, phaA and phaB, of P. putida have been indicated to carry out the hydroxylation of double bonds during phenylacetate catabolism (Olivera et al., Proc. Natl. Acad.
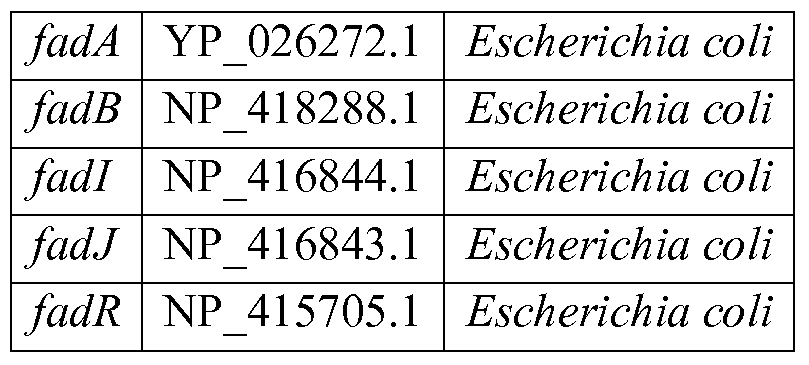
- the E. coli gene products oifadA and fadB encode a multienzyme complex involved in fatty acid oxidation that exhibits enoyl-CoA hydratase activity (Haller et al., Biochemistry, 39:4622-4629 (2000), Martinez-Carrion et al., J. Biol. Chem. 240:3538-3546 (1965) and Matties et al., Appl. Environ. Microbiol., 58:1435-1439 (1992)). Knocking out a negative regulator encoded by fadR can be utilized to activate the fadB gene product (Jeng et al. Biochem. 13:2898-2903 (1974)).
- fadl and fadJ genes encode similar functions and are naturally expressed under anaerobic conditions (Atsumi et al., Nature 451 :86-89 (2008)). Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 27.
- the abfD genes from C. aminobutyrium and C. kluyveri match exactly with these n- terminal amino acid sequences, and have been indicated to encode the 4-hydroxybutyrul-CoA dehydratases/vinylacetyl-CoA ⁇ -isomerase activities.
- the abfD gene from Porphyromonas gingivalis ATCC 33277 is identified through homology from genome projects. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 27.
- step R Dehydration of 3-hydroxyisobutyryl-CoA to methacrylyl-CoA (step R) can be accomplished by a reversible 3-hydroxyacyl-CoA dehydratase such as crotonase (also called 3-hydroxybutyryl- CoA dehydratase, EC 4.2.1.55) or enoyl-CoA hydratase (also called 3-hydroxyacyl-CoA dehydratase, EC 4.2.1.17). These enzymes are generally reversible (Moskowitz et al., Biochemistry 8:2748-2755 (1969); Durre et al., FEMS Microbiol Rev 17:251-262 (1995)). Exemplary enzymes are listed above. 3-hydroxyisobutyryl-CoA is not a natural substrate of these enzymes, but it is similar in structure to the native substrate, 3 -hydroxybutyryl-CoA.
- 3-hydroxyisobutyryl-CoA is not a natural substrate of these enzymes, but it is similar
- step M is carried out by isobutyryl-CoA mutase (ICM), a cobalamin- dependent methylmutase that reversibly rearranges the carbon backbone of butyryl-CoA into isobutyryl-CoA (Ratnatilleke et al., J Biol Chem 274:31679-31685 (1999)).
- ICM isobutyryl-CoA mutase
- Such an enzyme is also suitable for catalyzing the conversion of 4-hydroxybutyryl-CoA to 3-hydroxyisobutyryl- CoA, described by step Q of Figure 1.
- Genes encoding a heterodimeric ICM include icm and icmB of Streptomyces cinnamonensis (Ratnatilleke et al., supra; Vrijbloed et al., J Bacteriol 181 :5600-5605 (1999); Zerbe-Burkhardt et al., J Biol Chem 273:6508-6517 (1998)).
- Homologous genes in Streptomyces avermitilis MA-4680 likely catalyze the same or similar transformations.
- MCM is encoded by genes scpA in Escherichia coli (Haller et al., supra; Bobik et al., Anal Bioanal Chem 375:344-349 (2003)) and mutA in Homo sapiens (Padovani et al., Biochemistry 45:9300-9306 (2006)).
- MCM contains alpha and beta subunits and is encoded by two genes. Exemplary genes encoding the two-subunit protein are Propionibacterium fredenreichii sp.
- sequences can be used to identify homologue proteins in GenBank or other databases through sequence similarity searches (for example, BLASTp).
- sequence similarity searches for example, BLASTp.
- the resulting homologue proteins and their corresponding gene sequences provide additional exogenous DNA sequences for transformation into E. coli or other suitable host microorganisms to generate production hosts.
- Additional genes include the following, which were identified based on high homology to the E. coli spcA gene product. Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 30.
- M. extorquens forms a complex with methylmalonyl-CoA mutase, stimulates in vitro mutase activity, and possibly protects it from irreversible inactivation (Korotkova et al., supra).
- the M. extorquens meaB gene product is highly similar to the product of the E. coli argK gene (BLASTp: 45% identity, e-value: 4e-67), which is adjacent to scpA on the chromosome. No sequence for a meaB homolog in P.
- Propionibacterium acnes KPA171202 gene product YP_055310.1
- YP_055310.1 is 51% identical to the M. extorquens meaB protein and its gene is also adjacent to the methylmalonyl-CoA mutase gene on the chromosome.
- Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 31.
- E. coli can synthesize adenosylcobalamin, a necessary cofactor for this reaction, when supplied with the intermediates cobinamide or cobalamin (Lawrence et al., J Bacteriol 177:6371-6380 (1995); Lawrence et al., Genetics 142:11-24 (1996)).
- the ability to synthesize cobalamins de novo has been conferred upon E. coli following the expression of heterologous genes (Raux et al., J Bacteriol 178:753-767 (1996)).
- Step D in Figure 1 refers to an acid-thiol ligase which catalyzes the conversion of acetoacetyl- CoA to acetoacetate.
- An exemplary acid-thiol ligase is the enzyme encoded by sucCD of is. coli which catalyzes the formation of succinyl-CoA from succinate with the concomitant consumption of one ATP, a reaction which is reversible in vivo (Buck et al., Biochemistry 24:6245-6252 (1985)).
- Additional enzymes are acetoacetyl-CoA synthetases from Mus musculus (Hasegawa et al., Biochim Biophys Acta 1779:414-419 (2008)) and Homo sapiens (Ohgami et al., Biochem Pharmacol 65:989-994 (2003)) which naturally catalyze the ATP-dependant conversion of acetoacetate into acetoacetyl-CoA.
- Such enzymes can convert acetoacetyl-CoA to acetoacetate should they exhibit acetoacetyl-CoA hydrolase activity.
- Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 32. Table 32
- ADP-forming acetyl-CoA synthetase (ACD, EC 6.2.1.13) is another enzyme that couples the conversion of acyl-CoA esters to their corresponding acids with the concurrent synthesis of ATP. Although this enzyme has not been shown to react with acetoacetyl-CoA as a substrate, several enzymes with broad substrate specificities have been described in the literature.
- Haloarcula marismortui annotated as a succinyl-CoA synthetase accepts propionate, butyrate, and branched-chain acids (isovalerate and isobutyrate) as substrates, and was shown to operate in the forward and reverse directions (Brasen et al., Arch Microbiol 182:277-287 (2004)).
- the ACD encoded by PAE3250 from hyperthermophilic crenarchaeon Pyrobaculum aerophilum showed the broadest substrate range of all characterized ACDs, reacting with acetyl-CoA, isobutyryl-CoA (preferred substrate) and phenylacetyl-CoA (Brasen et al., supra).
- the enzymes from A. fulgidus, H. marismortui and P. aerophilum have all been cloned, functionally expressed, and characterized in E. coli (Musfeldt et al., swpr ⁇ ;Brasen et al., supra).
- the net transformation depicted by step D of Figure 1 can also be carried out by two enzymes such as acetate kinase and phosptransacetylase or butyrate kinase and phosphotransbutyrylase.
- Data related to the sequences for each of these exemplary gene products can be found using the following GenBank accession numbers shown in Table 33.
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include a 4-hydroxybutyryl-CoA dehydratase encoded by one or more genes selected from the group consisting oifumA,fumB,fumC,fumH,fuml, MmcB, MmcC, hmd, BACCAP '_02294, ANACOLJJ2527, NtherDRAFT_2368, dmclA, dmclB, crt, crtl,paaA, paaB,phaA,phaB, maoC, paaF, paaG, abfl), and Msed_l 220.
- the A- hydroxybutyryl-CoA dehydratase is encoded by abfl).
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include a crotonase encoded by one or more genes selected from the group consisting oifumA,fumB,fumC,fumH,fuml, MmcB, MmcC, hmd, BACCAP _ 02294, ANACOL_02527, NtherDRAFT_2368, dmdA, dmdB, crt, crtl, paaA,paaB,phaA,phaB, maoC, paaF,paaG, abfD, and Msed_1220.
- the crotonase is encoded by one or more genes selected from the group consisting of crt, crtl,paaA, paaB,phaA,phaB, maoC, paaF, and paaG.
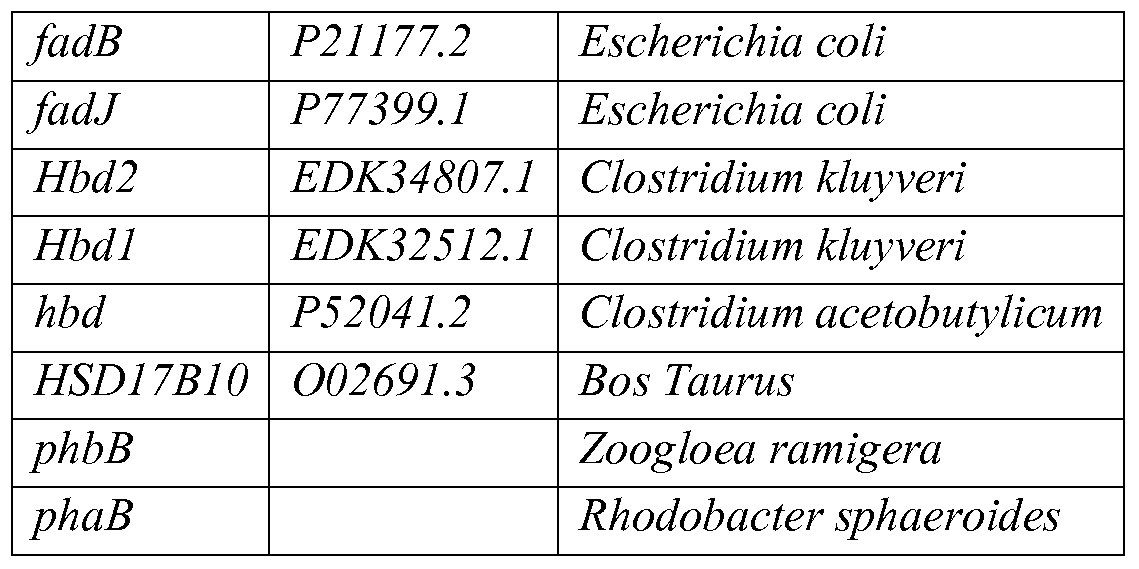
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include a 3-hydroxybutyryl-CoA dehydrogenase encoded by one or more genes selected from the group consisting oithrA, akthr2, homo, homl, hom2,fadB,fadJ, Hbd2, Hbdl, hbd, HSD17B10, phbB,phaB, Msed_1423, Msed_0399, Msed_0389, Msed_1993, adh, adhA, adh-A, mdh, idhA, Idh, and bdh.
- the 3-hydroxybutyryl- CoA dehydrogenase is encoded by one or more genes selected from the group consisting o ⁇ hbd, Hbd2, Hbdl, Msed_1423, Msed_0399, Msed_0389, Msed_1993,fadB, and fadJ.
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include an acetoacetyl-CoA synthetase encoded by one or more genes selected from the group consisting of sucC, sucD, AACS, AF1211, scs, and PAE3250.
- the acetoacetyl-CoA synthetase is encoded by one or more genes selected from the group consisting of sucC, sucD, AACS, and AF1211.
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include an acetyl-CoA:acetoacetate-CoA transferase encoded by one or more genes selected from the group consisting of atoA, atoD, actA, cgO592, ctfA, ctfB, catl, cat2, cat3, gctA, gctB, HPAGl _0676, HPAGl _0677, ScoA, and ScoB.
- the acetyl-CoA:acetoacetate-CoA transferase is encoded by one or more genes selected from the group consisting oiatoA, atoD, actA, cgO592, ctfA, ctfB, HPAGl _0676, HPAGl _0677, ScoA, and ScoB.
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can also include an acetoacetate decarboxylase encoded by one or more genes selected from the group consisting of Adc, cbei_3835, CLL_A2135, RBAM_030030.
- the non-naturally occurring microbial organism capable of producing isopropanol from A- hydroxybutyryl-CoA can include an acetone reductase encoded by one or more genes selected from the group consisting oithrA, akthr2, homo, homl, hom2,fadB,fadJ, Hbd2, Hbdl, hbd, HSD17B10, phbB,phaB, Msed_1423, Msed_0399, Msed_0389, Msed_1993, adh, adhA, adh-A, mdh, idhA, Idh, and bdh.
- the acetone reductase is encoded by one or more genes selected from the group consisting o ⁇ adh, adhA, and adh-A.
- the non-naturally occurring microbial organism capable of producing w-butanol from A- hydroxybutyryl-CoA can include a 4-hydroxybutyryl-CoA dehydratase encoded by one or more genes selected from the group consisting oifumA,fumB,fumC,fumH,fuml, MmcB, MmcC, hmd, BACCAP '_02294, ANACOLJJ2527, NtherDRAFT_2368, dmdA, dmdB, crt, crtl,paaA, paaB,phaA,phaB, maoC, paaF, paaG, abfl), and Msed_l 220.
- the A- hydroxybutyryl-CoA dehydratase is encoded by abfl).
- the non-naturally occurring microbial organism capable of producing w-butanol from A- hydroxybutyryl-CoA can include a crotonoyl-CoA reductase encoded by one or more genes selected from the group consisting of bed, etfA, et ⁇ , Ter, TDE0597, IBD, RPA3448, FadH, and enr.
- the crotonoyl-CoA reductase is encoded by one or more genes selected from the group consisting o ⁇ bcd, etfA, etfB, Ter, and TDE0597.
- the non-naturally occurring microbial organism capable of producing w-butanol from A- hydroxybutyryl-CoA can include a butyraldehyde reductase encoded by one or more genes selected from the group consisting oialrA, ADH2, yqhD, bdh I, bdh II, adhA, 4hbd, adhl, P84067, mmsb, dhat, and 3hidh.
- the butyraldehyde reductase is encoded by one or more genes selected from the group consisting oialrA, ADH2, yqhD, bdh I, bdh II, 4hbd, adhl, and mmsb.
- the non-naturally occurring microbial organism capable of producing w-butanol from A- hydroxybutyryl-CoA can include a butyryl-CoA reductase (alcohol forming) encoded by one or more genes selected from the group consisting oiadhE, adhE2, mcr, Rcas_2929, NAPl_02720, MGP2080_00535, and FAR.
- the butyryl-CoA reductase (alcohol forming) is encoded by one or more genes selected from the group consisting oiadhE2, mcr, and FAR.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a 4-hydroxybutyryl-CoA dehydratase encoded by one or more genes selected from the group consisting oifumA,fumB,fumC,fumH,fuml, MmcB, MmcC, hmd, BACCAF '_02294, ANACOL_02527, NtherDRAFT_2368, dmdA, dmdB, crt, crtl,paaA, paaB,phaA,phaB, maoC, paaF , paaG, abfD, and Msed_l 220.
- the 4-hydroxybutyryl-CoA dehydratase is encoded by crt, crtl,paaA, paaB, phaA, phaB, maoC, paaF, and paa G.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a crotonoyl-CoA reductase encoded by one or more genes selected from the group consisting of bed, etfA, et ⁇ , Ter, TDE0597, IBD, RP A3448, FadH, and enr.
- the crotonoyl-CoA reductase is encoded by one or more genes selected from the group consisting of bed, etfA, etfB, Ter, and TDE0597.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a isobutyryl-CoA mutase encoded by one or more genes selected from the group consisting oi icm, icmB, icmA, Mpe_B0538, Mpe_B0541, scpA, mutA, mutB, mcmA, mcmB, sbm, SARI_04585, YfreAJ)! 000861, argK, PPA0597, and meaB.
- the isobutyryl-CoA mutase is encoded by one or more genes selected from the group consisting oiicmB, icmA, Mpe_B0538, and Mpe_B0541.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a 3-hydroxyisobutyryl-CoA dehydratase encoded by one or more genes selected from the group consisting oifumA,fumB,fumC,fumH,fuml, MmcB, MmcC, hmd, BACCAP _02294, ANACOL_02527, NtherDRAFT_2368, dmdA, dmdB, crt, crtl, paaA, paaB, phaA, phaB, maoC, paaF, paaG, abfD, and Msed_1220.
- the 3-hydroxyisobutyryl-CoA dehydratase is encoded by one or more genes selected from the group consisting of crt, crtl,paaA, paaB, phaA, phaB, maoC, paaF, and paaG.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a methacrylyl-CoA reductase encoded by one or more genes selected from the group consisting of bed, etfA, et ⁇ , Ter, TDE0597, IBD, RP A3448, FadH, and enr.
- the methacrylyl-CoA reductase is encoded by one or more genes selected from the group consisting o ⁇ bcd, etfA, et ⁇ , Ter, and TDE0597.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a isobutyryl-CoA reductase (aldehyde forming) encoded by one or more genes selected from the group consisting oiacrl, sucD, bphG, adhE, Msed_0709, mcr, asd-2, Saci_2370, Aid, and eutE.
- the isobutyryl-CoA reductase (aldehyde forming) is encoded by one or more genes selected from the group consisting of sucD, bphG, Msed_0709, mcr, and Aid.
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include a isobutyraldehyde reductase encoded by one or more genes selected from the group consisting oialrA, ADH2, yqhD, bdh I, bdh II, adhA, 4hbd, adhl,
- the non-naturally occurring microbial organism capable of producing isobutanol from A- hydroxybutyryl-CoA can include an isobutyryl-CoA reductase (alcohol forming) encoded by one or more genes selected from the group consisting oiadhE, adhE2, mcr, Rcas_2929, NAPl_02720, MGP2080_00535, and FAR.
- the isobutyryl-CoA reductase (alcohol forming) is encoded by one or more genes selected from the group consisting oiadhE2, mcr, and FAR.
- the non-naturally occurring microbial organisms of the invention can be produced by introducing expressible nucleic acids encoding one or more of the enzymes or proteins participating in one or more isopropanol, M-butanol, or isobutanol biosynthetic pathways.
- nucleic acids for some or all of a particular isopropanol, M-butanol, or isobutanol biosynthetic pathway can be expressed. For example, if a chosen host is deficient in one or more enzymes or proteins for a desired biosynthetic pathway, then expressible nucleic acids for the deficient enzyme(s) or protein(s) are introduced into the host for subsequent exogenous expression.
- a non-naturally occurring microbial organism of the invention can be produced by introducing exogenous enzyme or protein activities to obtain a desired biosynthetic pathway or a desired biosynthetic pathway can be obtained by introducing one or more exogenous enzyme or protein activities that, together with one or more endogenous enzymes or proteins, produces a desired product such as isopropanol, M-butanol, or isobutanol.
- the non-naturally occurring microbial organisms of the invention will include at least one exogenously expressed isopropanol, M-butanol, or isobutanol pathway-encoding nucleic acid and up to all encoding nucleic acids for one or more isopropanol, M-butanol, or isobutanol biosynthetic pathways.
- isopropanol, M-butanol, or isobutanol biosynthesis can be established in a host deficient in a pathway enzyme or protein through exogenous expression of the corresponding encoding nucleic acid.
- exogenous expression of all enzyme or proteins in the pathway can be included, although it is understood that all enzymes or proteins of a pathway can be expressed even if the host contains at least one of the pathway enzymes or proteins.
- exogenous expression of all enzymes or proteins in a pathway for production of isopropanol, «-butanol, or isobutanol can be included.
- a non-naturally occurring microbial organism of the invention can have one, two, three, four, five, up to all nucleic acids encoding the enzymes or proteins constituting an isopropanol, w-butanol, or isobutanol biosynthetic pathway disclosed herein.
- the non-naturally occurring microbial organisms also can include other genetic modifications that facilitate or optimize isopropanol, w-butanol, or isobutanol biosynthesis or that confer other useful functions onto the host microbial organism.
- One such other functionality can include, for example, augmentation of the synthesis of one or more of the isopropanol, «-butanol, or isobutanol pathway precursors such as acetyl-CoA.
- a host microbial organism is selected such that it produces the precursor of an isopropanol, w-butanol, or isobutanol pathway, either as a naturally produced molecule or as an engineered product that either provides de novo production of a desired precursor or increased production of a precursor naturally produced by the host microbial organism.
- acetyl-CoA is produced naturally in a host organism such as E. coli.
- a host organism can be engineered to increase production of a precursor, as disclosed herein.
- a microbial organism that has been engineered to produce a desired precursor can be used as a host organism and further engineered to express enzymes or proteins of an isopropanol, w-butanol, or isobutanol pathway.
- a non-naturally occurring microbial organism of the invention is generated from a host that contains the enzymatic capability to synthesize isopropanol, n- butanol, or isobutanol.
- it can be useful to increase the synthesis or accumulation of an isopropanol, «-butanol, or isobutanol pathway product to, for example, drive isopropanol, w-butanol, or isobutanol pathway reactions toward isopropanol, w-butanol, or isobutanol production.
- Increased synthesis or accumulation can be accomplished by, for example, overexpression of nucleic acids encoding one or more of the above-described isopropanol, w-butanol, or isobutanol pathway enzymes or proteins.
- Over expression the enzyme or enzymes and/or protein or proteins of the isopropanol, w-butanol, or isobutanol pathway can occur, for example, through exogenous expression of the endogenous gene or genes, or through exogenous expression of the heterologous gene or genes.
- naturally occurring organisms can be readily generated to be non-naturally occurring microbial organisms of the invention, for example, producing isopropanol, «-butanol, or isobutanol, through overexpression of one, two, three, four, five, that is, up to all nucleic acids encoding isopropanol, w-butanol, or isobutanol biosynthetic pathway enzymes or proteins.
- a non-naturally occurring organism can be generated by mutagenesis of an endogenous gene that results in an increase in activity of an enzyme in the isopropanol, «-butanol, or isobutanol biosynthetic pathway.
- exogenous expression of the encoding nucleic acids is employed.
- Exogenous expression confers the ability to custom tailor the expression and/or regulatory elements to the host and application to achieve a desired expression level that is controlled by the user.
- endogenous expression also can be utilized in other embodiments such as by removing a negative regulatory effector or induction of the gene's promoter when linked to an inducible promoter or other regulatory element.
- an endogenous gene having a naturally occurring inducible promoter can be up-regulated by providing the appropriate inducing agent, or the regulatory region of an endogenous gene can be engineered to incorporate an inducible regulatory element, thereby allowing the regulation of increased expression of an endogenous gene at a desired time.
- an inducible promoter can be included as a regulatory element for an exogenous gene introduced into a non-naturally occurring microbial organism.
- a non- naturally occurring microbial organism having isopropanol, «-butanol, or isobutanol biosynthetic pathway can comprise at least two exogenous nucleic acids encoding desired enzymes or proteins.
- any combination of two or more enzymes or proteins of a biosynthetic pathway can be included in a non-naturally occurring microbial organism of the invention.
- any combination of three or more enzymes or proteins of a biosynthetic pathway can be included in a non-naturally occurring microbial organism of the invention and so forth, as desired, so long as the combination of enzymes and/or proteins of the desired biosynthetic pathway results in production of the corresponding desired product.
- any combination of four, or more enzymes or proteins of a biosynthetic pathway as disclosed herein can be included in a non-naturally occurring microbial organism of the invention, as desired, so long as the combination of enzymes and/or proteins of the desired biosynthetic pathway results in production of the corresponding desired product.
- non-naturally occurring microbial organisms and methods of the invention also can be utilized in various combinations with each other and with other microbial organisms and methods well known in the art to achieve product biosynthesis by other routes.
- the isopropanol, n- butanol, or isobutanol pathway intermediate can then be used as a substrate for a second microbial organism that converts the isopropanol, «-butanol, or isobutanol pathway intermediate to isopropanol, «-butanol, or isobutanol.
- isopropanol, «-butanol, or isobutanol can be accomplished by constructing a microbial organism that contains biosynthetic pathways for conversion of one pathway intermediate to another pathway intermediate or the product.
- isopropanol, «-butanol, or isobutanol also can be biosynthetically produced from microbial organisms through co-culture or co- fermentation using two organisms in the same vessel, where the first microbial organism produces isopropanol, «-butanol, or isobutanol intermediate and the second microbial organism converts the intermediate to isopropanol, w-butanol, or isobutanol.
- Sources of encoding nucleic acids for isopropanol, w-butanol, or isobutanol pathway enzyme or protein can include, for example, any species where the encoded gene product is capable of catalyzing the referenced reaction.
- Such species include both prokaryotic and eukaryotic organisms including, but not limited to, bacteria, including archaea and eubacteria, and eukaryotes, including yeast, plant, insect, animal, and mammal, including human.
- Exemplary species for such sources include, for example, Escherichia coli, as well as other exemplary species disclosed herein or available as source organisms for corresponding genes.
- the metabolic alterations enabling biosynthesis of isopropanol, «-butanol, or isobutanol described herein with reference to a particular organism such as E. coli can be readily applied to other microorganisms, including prokaryotic and eukaryotic organisms alike. Given the teachings and guidance provided herein, those skilled in the art will know that a metabolic alteration exemplified in one organism can be applied equally to other organisms.
- isopropanol, w-butanol, or isobutanol biosynthetic pathway exists in an unrelated species
- isopropanol, w-butanol, or isobutanol biosynthesis can be conferred onto the host species by, for example, exogenous expression of a paralog or paralogs from the unrelated species that catalyzes a similar, yet non-identical metabolic reaction to replace the referenced reaction. Because certain differences among metabolic networks exist between different organisms, those skilled in the art will understand that the actual gene usage between different organisms can differ.
- Host microbial organisms can be selected from, and the non-naturally occurring microbial organisms generated in, for example, bacteria, yeast, fungus or any of a variety of other microorganisms applicable to fermentation processes.
- Exemplary bacteria include species selected from Escherichia coli, Klebsiella oxytoca, Anaerobiospirillum succiniciproducens, Actinobacillus succinogenes, Mannheimia succiniciproducens, Rhizobium etli, Bacillus subtilis, Corynebacterium glutamicum, Gluconobacter oxydans, Zymomonas mobilis, Lactococcus lactis, Lactobacillus plantarum, Streptomyces coelicolor, Clostridium acetobutylicum, Pseudomonas fluorescens, and Pseudomonas putida.
- Exemplary yeasts or fungi include species selected from Saccharomyces cerevisiae, Schizosaccharomyces pombe, Kluyveromyces lactis, Kluyveromyces marxianus, Aspergillus terreus, Aspergillus niger and Pichia pastoris .
- E. coli is a particularly useful host organism since it is a well characterized microbial organism suitable for genetic engineering.
- Other particularly useful host organisms include yeast such as Saccharomyces cerevisiae.
- Methods for constructing and testing the expression levels of a non-naturally occurring isopropanol, w-butanol, or isobutanol-producing host can be performed, for example, by recombinant and detection methods well known in the art. Such methods can be found described in, for example, Sambrook et al., Molecular Cloning: A Laboratory Manual, Third Ed., Cold Spring Harbor Laboratory, New York (2001); and Ausubel et al., Current Protocols in Molecular Biology, John Wiley and Sons, Baltimore, MD (1999).
- Exogenous nucleic acid sequences involved in a pathway for production of isopropanol, n- butanol, or isobutanol can be introduced stably or transiently into a host cell using techniques well known in the art including, but not limited to, conjugation, electroporation, chemical transformation, transduction, transfection, and ultrasound transformation.
- some nucleic acid sequences in the genes or cDNAs of eukaryotic nucleic acids can encode targeting signals such as an w-terminal mitochondrial or other targeting signal, which can be removed before transformation into prokaryotic host cells, if desired. For example, removal of a mitochondrial leader sequence led to increased expression in is.
- genes can be expressed in the cytosol without the addition of leader sequence, or can be targeted to mitochondrion or other organelles, or targeted for secretion, by the addition of a suitable targeting sequence such as a mitochondrial targeting or secretion signal suitable for the host cells.
- a suitable targeting sequence such as a mitochondrial targeting or secretion signal suitable for the host cells.
- Selection control sequences can include constitutive and inducible promoters, transcription enhancers, transcription terminators, and the like which are well known in the art.
- both nucleic acids can be inserted, for example, into a single expression vector or in separate expression vectors.
- the encoding nucleic acids can be operationally linked to one common expression control sequence or linked to different expression control sequences, such as one inducible promoter and one constitutive promoter.
- exogenous nucleic acid sequences involved in a metabolic or synthetic pathway can be confirmed using methods well known in the art. Such methods include, for example, nucleic acid analysis such as Northern blots or polymerase chain reaction (PCR) amplification of mRNA, or immunob lotting for expression of gene products, or other suitable analytical methods to test the expression of an introduced nucleic acid sequence or its corresponding gene product. It is understood by those skilled in the art that the exogenous nucleic acid is expressed in a sufficient amount to produce the desired product, and it is further understood that expression levels can be optimized to obtain sufficient expression using methods well known in the art and as disclosed herein.
- nucleic acid analysis such as Northern blots or polymerase chain reaction (PCR) amplification of mRNA, or immunob lotting for expression of gene products, or other suitable analytical methods to test the expression of an introduced nucleic acid sequence or its corresponding gene product.
- PCR polymerase chain reaction
- the invention provides a method for producing isopropanol, w-butanol, or isobutanol that includes culturing the non-naturally occurring microbial organism disclosed herein, under conditions and for a sufficient period of time to produce isopropanol, w-butanol, or isobutanol, including organisms that incorporate one, two, three, four, five, six, seven, eight, up to all exogenous nucleic acids encoding enzymes that complete a isopropanol, «-butanol, or isobutanol pathway.
- at least one exogenous nucleic acid is a heterologous nucleic acid.
- Suitable purification and/or assays to test for the production of isopropanol, w-butanol, or isobutanol can be performed using well known methods. Suitable replicates such as triplicate cultures can be grown for each engineered strain to be tested. For example, product and byproduct formation in the engineered production host can be monitored. The final product and intermediates, and other organic compounds, can be analyzed by methods such as HPLC (High Performance Liquid Chromatography), GC-MS (Gas Chromatography-Mass Spectroscopy) and LC-MS (Liquid Chromatography-Mass Spectroscopy) or other suitable analytical methods using routine procedures well known in the art. The release of product in the fermentation broth can also be tested with the culture supernatant.
- HPLC High Performance Liquid Chromatography
- GC-MS Gas Chromatography-Mass Spectroscopy
- LC-MS Liquid Chromatography-Mass Spectroscopy
- Byproducts and residual glucose can be quantified by HPLC using, for example, a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng. 90:775-779 (2005)), or other suitable assay and detection methods well known in the art.
- the individual enzyme or protein activities from the exogenous DNA sequences can also be assayed using methods well known in the art (see, for example, WO/2008/115840 and Hanai et al., Appl Environ Microbiol 73:7814-7818 (2007)).
- the isopropanol, «-butanol, or isobutanol can be separated from other components in the culture using a variety of methods well known in the art. Such separation methods include, for example, extraction procedures as well as methods that include continuous liquid-liquid extraction, pervaporation, membrane filtration, membrane separation, reverse osmosis, electrodialysis, distillation, crystallization, centrifugation, extractive filtration, ion exchange chromatography, size exclusion chromatography, adsorption chromatography, and ultrafiltration. All of the above methods are well known in the art. Any of the non- naturally occurring microbial organisms described herein can be cultured to produce and/or secrete the biosynthetic products of the invention. For example, the isopropanol, «-butanol, or isobutanol producers can be cultured for the biosynthetic production of isopropanol, w-butanol, or isobutanol.
- the recombinant strains are cultured in a medium with carbon source and other essential nutrients. It is highly desirable to maintain anaerobic conditions in the fermenter to reduce the cost of the overall process. Such conditions can be obtained, for example, by first sparging the medium with nitrogen and then sealing the flasks with a septum and crimp-cap. For strains where growth is not observed anaerobically, microaerobic conditions can be applied by perforating the septum with a small hole for limited aeration. Exemplary anaerobic conditions have been described previously and are well-known in the art. Exemplary aerobic and anaerobic conditions are described, for example, in United State Patent application serial No. 11/891,602, filed August 10, 2007. Fermentations can be performed in a batch, fed-batch or continuous manner, as disclosed herein.
- the pH of the medium can be maintained at a desired pH, in particular neutral pH, such as a pH of around 7 by addition of a base, such as NaOH or other bases, or acid, as needed to maintain the culture medium at a desirable pH.
- the growth rate can be determined by measuring optical density using a spectrophotometer (600 nm), and the glucose uptake rate by monitoring carbon source depletion over time.
- the isopropanol, w-butanol, or isobutanol microbial organisms of the invention also can be modified for growth on syngas as its source of carbon.
- one or more proteins or enzymes are expressed in the isopropanol, w-butanol, or isobutanol producing organisms to provide a metabolic pathway for utilization of syngas or other gaseous carbon source.
- Organisms of the present invention can utilize, for example, any carbohydrate source which can supply a source of carbon to the non-naturally occurring microorganism.
- Such sources include, for example, sugars such as glucose, xylose, arabinose, galactose, mannose, fructose and starch.
- Other sources of carbohydrate include, for example, renewable feedstocks and biomass.
- Exemplary types of biomasses that can be used as feedstocks in the methods of the invention include cellulosic biomass, hemicellulosic biomass and lignin feedstocks or portions of feedstocks.
- Such biomass feedstocks contain, for example, carbohydrate substrates useful as carbon sources such as glucose, xylose, arabinose, galactose, mannose, fructose and starch.
- carbohydrate substrates useful as carbon sources such as glucose, xylose, arabinose, galactose, mannose, fructose and starch.
- a non-naturally occurring microbial organism can be produced that secretes the biosynthesized compounds of the invention when grown on a carbon source such as , syngas, CO and/or CO2.
- a carbon source such as , syngas, CO and/or CO2.
- Such compounds include, for example, isopropanol, w-butanol, or isobutanol and any of the intermediate metabolites in the isopropanol, w-butanol, or isobutanol pathway.
- the non-naturally occurring microbial organisms of the invention are constructed using methods well known in the art as exemplified herein to exogenously express at least one nucleic acid encoding an isopropanol, w-butanol, or isobutanol pathway enzyme or protein in sufficient amounts to produce isopropanol, w-butanol, or isobutanol. It is understood that the microbial organisms of the invention are cultured under conditions sufficient to produce isopropanol, n- butanol, or isobutanol.
- the non-naturally occurring microbial organisms of the invention can achieve biosynthesis of isopropanol, n- butanol, or isobutanol resulting in intracellular concentrations between about 0.1-2000 mM or more.
- the intracellular concentration of isopropanol, «-butanol, or isobutanol is between about 3-1800 mM, particularly between about 5-1700 mM and more particularly between about 8-1600 mM, including about 100 mM, 200 mM, 500 mM, 800 mM, or more.
- Intracellular concentrations between and above each of these exemplary ranges also can be achieved from the non-naturally occurring microbial organisms of the invention.
- culture conditions include anaerobic or substantially anaerobic growth or maintenance conditions.
- Exemplary anaerobic conditions have been described previously and are well known in the art.
- Exemplary anaerobic conditions for fermentation processes are described herein and are described, for example, in U.S. patent application No. US 2009/0047719, filed August 10, 2007. Any of these conditions can be employed with the non- naturally occurring microbial organisms as well as other anaerobic conditions well known in the art.
- the isopropanol, «-butanol, or isobutanol producers can synthesize isopropanol, «-butanol, or isobutanol at intracellular concentrations of 5-10 mM or more as well as all other concentrations exemplified herein. It is understood that, even though the above description refers to intracellular concentrations, isopropanol, «-butanol, or isobutanol producing microbial organisms can produce isopropanol, «-butanol, or isobutanol intracellular Iy and/or secrete the product into the culture medium.
- the culture conditions can include, for example, liquid culture procedures as well as fermentation and other large scale culture procedures. As described herein, particularly useful yields of the biosynthetic products of the invention can be obtained under anaerobic or substantially anaerobic culture conditions.
- one exemplary growth condition for achieving biosynthesis of isopropanol, «-butanol, or isobutanol includes anaerobic culture or fermentation conditions.
- the non-naturally occurring microbial organisms of the invention can be sustained, cultured or fermented under anaerobic or substantially anaerobic conditions.
- anaerobic conditions refers to an environment devoid of oxygen.
- substantially anaerobic conditions include, for example, a culture, batch fermentation or continuous fermentation such that the dissolved oxygen concentration in the medium remains between 0 and 10% of saturation.
- Substantially anaerobic conditions also includes growing or resting cells in liquid medium or on solid agar inside a sealed chamber maintained with an atmosphere of less than 1% oxygen.
- the percent of oxygen can be maintained by, for example, sparging the culture with an N2/CO2 mixture or other suitable non-oxygen gas or gases.
- the culture conditions described herein can be scaled up and grown continuously for manufacturing of isopropanol, w-butanol, or isobutanol.
- Exemplary growth procedures include, for example, fed-batch fermentation and batch separation; fed-batch fermentation and continuous separation, or continuous fermentation and continuous separation. All of these processes are well known in the art. Fermentation procedures are particularly useful for the biosynthetic production of commercial quantities of isopropanol, w-butanol, or isobutanol.
- the continuous and/or near- continuous production of isopropanol, w-butanol, or isobutanol will include culturing a non- naturally occurring isopropanol, «-butanol, or isobutanol producing organism of the invention in sufficient nutrients and medium to sustain and/or nearly sustain growth in an exponential phase.
- Continuous culture under such conditions can be include, for example, 1 day, 2, 3, 4, 5, 6 or 7 days or more.
- continuous culture can include 1 week, 2, 3, 4 or 5 or more weeks and up to several months.
- organisms of the invention can be cultured for hours, if suitable for a particular application.
- the continuous and/or near- continuous culture conditions also can include all time intervals in between these exemplary periods. It is further understood that the time of culturing the microbial organism of the invention is for a sufficient period of time to produce a sufficient amount of product for a desired purpose.
- Fermentation procedures are well known in the art. Briefly, fermentation for the biosynthetic production of isopropanol, w-butanol, or isobutanol can be utilized in, for example, fed-batch fermentation and batch separation; fed-batch fermentation and continuous separation, or continuous fermentation and continuous separation. Examples of batch and continuous fermentation procedures are well known in the art.
- the isopropanol, w-butanol, or isobutanol producers also can be, for example, simultaneously subjected to chemical synthesis procedures to convert the product to other compounds or the product can be separated from the fermentation culture and sequentially subjected to chemical conversion to convert the product to other compounds, if desired.
- syngas can be used as a carbon feedstock.
- Important process considerations for a syngas fermentation are high biomass concentration and good gas-liquid mass transfer (Bredwell et al, Biotechnol Prog. 15:834-844 (1999).
- the solubility of CO in water is somewhat less than that of oxygen.
- Continuously gas-sparged fermentations can be performed in controlled fermenters with constant off-gas analysis by mass spectrometry and periodic liquid sampling and analysis by GC and HPLC.
- the liquid phase can function in batch mode.
- Fermentation products such as alcohols, organic acids, and residual glucose along with residual methanol are quantified by HPLC (Shimadzu, Columbia MD), for example, using an Aminex® series of HPLC columns (for example, HPX-87 series) (BioRad, Hercules CA), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids.
- the growth rate is determined by measuring optical density using a spectrophotometer (600 nm). All piping in these systems is glass or metal to maintain anaerobic conditions.
- the gas sparging is performed with glass frits to decrease bubble size and improve mass transfer. Various sparging rates are tested, ranging from about 0.1 to 1 wm (vapor volumes per minute). To obtain accurate measurements of gas uptake rates, periodic challenges are performed in which the gas flow is temporarily stopped, and the gas phase composition is monitored as a function of time.
- One method to increase the microbial concentration is to recycle cells via a tangential flow membrane from a sidestream.
- Repeated batch culture can also be used, as previously described for production of acetate by Moorella (Sakai et al., J Biosci.Bioeng 99:252- 258 (2005)).
- Various other methods can also be used (Bredwell et al., Biotechnol Prog. 15:834- 844 (1999); Datar et al., Biotechnol Bioeng 86:587-594 (2004)). Additional optimization can be tested such as overpressure at 1.5 atm to improve mass transfer (Najafpour and Younesi, Enzyme and Microbial Technology 38[l-2], 223-228 (2006)).
- a typical impurity profile is 4.5% CH 4 , 0.1% C 2 H 2 , 0.35% C 2 H 6 , 1.4% C 2 H 4 , and 150 ppm nitric oxide (Datar et al., Biotechnol Bioeng 86:587-594 (2004)).
- Tars represented by compounds such as benzene, toluene, ethylbenzene, p-xylene, o-xylene, and naphthalene, are added at ppm levels to test for any effect on production.
- One class of enzymes in the pathways disclosed herein is the oxidoreductases that interconvert ketones or aldehydes to alcohols (1.1.1). Enzymes in this class that can operate on a wide range of substrates.
- Table 34 shows the activity of the enzyme and its K m on different alcohols. The enzyme is reversible and has very high activity on several aldehydes also as shown in Table 34.
- Lactate dehydrogenase (1.1.1.27) from Ralstonia eutropha is another enzyme that has been demonstrated to have high activities on several 2-oxoacids such as 2-oxobutyrate, 2- oxopentanoate and 2-oxoglutarate (a C5 compound analogous to 2-oxoadipate) (Steinbuchel et al., Eur. J. Biochem. 130:329-334 (1983)).
- Column 2 in Table 36 shows the activities o ⁇ ldhA from R. eutropha (formerly A eutrophus) on different substrates (Steinbuchel et al., supra).
- Table 36 shows the activities o ⁇ ldhA from R. eutropha (formerly A eutrophus) on different substrates (Steinbuchel et al., supra).
- Oxidoreductases that can convert 2-oxoacids to their acyl-CoA counterparts (1.2.1) have been shown to accept multiple substrates as well.
- branched-chain 2-keto-acid dehydrogenase complex also known as 2-oxoisovalerate dehydrogenase (1.2.1.25)
- BCKAD branched-chain 2-keto-acid dehydrogenase complex
- 2-oxoisovalerate dehydrogenase (1.2.1.25)
- 2-keto acids derivatives of valine, leucine and isoleucine to their acyl-CoA derivatives and CO2.
- Rattus norvegicus Paxton et al., Biochem. J.
- CoA transferases (2.8.3) have been demonstrated to have the ability to act on more than one substrate. Specifically, a CoA transferase was purified from Clostridium acetobutylicum and was reported to have the highest activities on acetate, propionate, and butyrate. It also had significant activities with valerate, isobutyrate, and crotonate (Wiesenborn et al., Appl. Environ. Microbiol. 55:323-329 (1989)). In another study, the E.
- acyl-CoA:acetate-CoA transferase also known as acetate-CoA transferase (EC 2.8.3.8) has been shown to transfer the CoA moiety to acetate from a variety of branched and linear acyl-CoA substrates, including isobutyrate (Matthies et al., Appl Environ Microbiol 58:1435-1439 (1992)), valerate (Vanderwinkel et al., Biochem. Biophys. Res Commun. 33:902-908 (1968)) and butanoate (Vanderwinkel et al., supra).
- L-rhamnose isomerase from Pseudomonas stutzeri catalyzes the isomerization between various aldoalses and ketoses (Yoshida et al., J. MoI. Biol. 365:1505- 1516 (2007)). These include isomerization between L-rhamnose and L-rhamnulose, L-mannose and L-fructose, L-xylose and L-xylulose, D-ribose and D-ribulose, and D-allose and D-psicose.
- the phosphotransferases (2.7.1), the homoserine kinase (2.7.1.39) from E. coli that converts L-homoserine to L-homoserine phosphate was found to phosphorylate numerous homoserine analogs.
- the carboxyl functional group at the R-position had been replaced by an ester or by a hydroxymethyl group (Huo et al., Biochemistry 35: 16180-16185 (1996)).
- Table 37 demonstrates the broad substrate specificity of this kinase.
- malonyl CoA synthetase (6.3.4.9) from Rhizobium trifolii could convert several diacids, namely, ethyl-, propyl-, allyl-, isopropyl-, dimethyl-, cyclopropyl-, cyclopropylmethylene-, cyclobutyl-, and benzyl-malonate into their corresponding monothio esters (Pohl et al., J. Am. Chem. Soc. 123:5822-5823 (2001)).
- decarboxylases (4.1.1) with broad substrate ranges.
- Pyruvate decarboxylase also termed keto-acid decarboxylase
- keto-acid decarboxylase is a key enzyme in alcoholic fermentation, catalyzing the decarboxylation of pyruvate to acetaldehyde.
- the enzyme isolated from Saccharomyces cerevisiae has a broad substrate range for aliphatic 2-keto acids including 2-ketobutyrate, 2-keto valerate, and 2-phenylpyruvate (Li et al., Biochemistry 38:10004-10012 (1999)).
- benzoylformate decarboxylase has a broad substrate range and has been the target of enzyme engineering studies.
- Lactococcus lactis has been characterized on a variety of branched and linear substrates including 2-oxobutanoate, 2-oxohexanoate, 2-oxopentanoate, 3-methyl-2- oxobutanoate, 4-methyl-2-oxobutanoate and isocaproate (Smit et al., Appl Environ Microbiol 71 :303-311 (2005)).
- enzymes known to have one dominant activity have also been indicated to catalyze a very different function.
- the cofactor-dependent phosphoglycerate mutase (5.4.2.1) from Bacillus stearothermophilus and Bacillus subtilis is known to function as a phosphatase as well (Rigden et al., Protein ScL 10:1835-1846 (2001)).
- the enzyme from B. stearothermophilus is known to have activity on several substrates, including 3- phosphoglycerate, alpha-napthylphosphate, p-nitrophenylphosphate, AMP, fructose-6- phosphate, ribose-5 -phosphate and CMP.
- triosephosphate isomerase enzyme was improved up to 19-fold from 1.3 fold (Hermes et al., Proc. Natl. Acad. Sci. U. S. A 87:696-700 (1990)). This enhancement in specific activity was accomplished by using random mutagenesis over the whole length of the protein and the improvement could be traced back to mutations in six amino acid residues.
- a case in point is the dihydroflavonol 4-reductase for which a single amino acid was changed in the presumed substrate-binding region that could preferentially reduce dihydrokaempferol (Johnson et al., Plant J. 25:325-333 (2001)).
- the substrate specificity of a very specific isocitrate dehydrogenase from Escherichia coli was changed form isocitrate to isopropylmalate by changing one residue in the active site (Doyle et al., Biochemistry 40:4234-4241 (2001)).
- a fucosidase was evolved from a galactosidase in E. coli by DNA shuffling and screening (Zhang et al., Proc Natl Acad Sci US. A 94:4504-4509 (1997)).
- aspartate aminotransferase from E. coli was converted into a tyrosine aminotransferase using homology modeling and site-directed mutagenesis (Onuffer et al., Protein Sci. 4:1750-1757 (1995)).
- CCP Cytochrome c peroxidase
- mutants were isolated which possessed a 300-fold increased activity against guaiacol and up to 1000-fold increased specificity for this substrate relative to that for the natural substrate (Iffland et al., Biochemistry 39:10790-10798 (2000)).
- enzymes with different substrate preferences than both the parent enzymes have been obtained.
- biphenyl-dioxygenase-mediated degradation of polychlorinated biphenyls was improved by shuffling genes from two bacteria, Pseudomonas pseudoalcaligens and Burkholderia cepacia (Kumamaru et al., Nat. Biotechnol 16, 663-666 (1998)).
- the resulting chimeric biphenyl oxygenases showed different substrate preferences than both the parental enzymes and enhanced the degradation activity towards related biphenyl compounds and single aromatic ring hydrocarbons such as toluene and benzene which were originally poor substrates for the enzyme.
- a final example of directed evolution shows the extensive modifications to which an enzyme can be subjected to achieve a range of desired functions.
- the enzyme, lactate dehydrogenase from Bacillus stearothermophilus was subjected to site-directed mutagenesis, and three amino acid substitutions were made at sites that were believed to determine the specificity towards different hydroxyacids (Clarke et al., Biochem. Biophys. Res. Commun. 148:15-23 (1987)). After these mutations, the specificity for oxaloacetate over pyruvate was increased to 500 in contrast to the wild type enzyme that had a catalytic specificity for pyruvate over oxaloacetate of 1000.
- This enzyme was further engineered using site-directed mutagenesis to have activity towards branched-chain substituted pyruvates (Wilks et al., Biochemistry 29:8587-8591 (1990)). Specifically, the enzyme had a 55-fold improvement in K cat for alpha-ketoisocaproate. Three structural modifications were made in the same enzyme to change its substrate specificity from lactate to malate. The enzyme was highly active and specific towards malate (Wilks et al., Science 242:1541-1544 (1988)). The same enzyme from B.
- stearothermophilus was subsequently engineered to have high catalytic activity towards alpha-keto acids with positively charged side chains, such as those containing ammonium groups (Hogan et al., Biochemistry 34:4225-4230 (1995)). Mutants with acidic amino acids introduced at position 102 of the enzyme favored binding of such side chain ammonium groups. The results obtained proved that the mutants showed up to 25 -fold improvements in kc a t/K m values for omega-amino-alpha-keto acid substrates.
- This enzyme was also structurally modified to function as a phenyllactate dehydrogenase instead of a lactate dehydrogenase (Wilks et al., Biochemistry 31 :7802-7806 (1992)). Restriction sites were introduced into the gene for the enzyme which enabled a region of the gene to be excised. This region coded for a mobile surface loop of polypeptide (residues 98-110) which normally seals the active site vacuole from bulk solvent and is a major determinant of substrate specificity. The variable length and sequence loops were inserted into the cut gene and used to synthesize hydroxyacid dehydrogenases with altered substrate specificities.
- directed evolution is a powerful approach that involves the introduction of mutations targeted to a specific gene in order to improve and/or alter the properties of an enzyme.
- Improved and/or altered enzymes can be identified through the development and implementation of sensitive high-throughput screening assays that allow the automated screening of many enzyme variants (e.g., >10 4 ). Iterative rounds of mutagenesis and screening typically are performed to afford an enzyme with optimized properties.
- Computational algorithms that can help to identify areas of the gene for mutagenesis also have been developed and can significantly reduce the number of enzyme variants that need to be generated and screened.
- Enzyme characteristics that have been improved and/or altered by directed evolution technologies include, for example, selectivity/specificity - for conversion of non-natural substrates; temperature stability - for robust high temperature processing; pH stability - for bioprocessing under lower or higher pH conditions; substrate or product tolerance - so that high product titers can be achieved; binding (K m ) - broadens substrate binding to include non-natural substrates; inhibition (K 1 ) - to remove inhibition by products, substrates, or key intermediates; activity (kcat) - increases enzymatic reaction rates to achieve desired flux; expression levels - increases protein yields and overall pathway flux; oxygen stability - for operation of air sensitive enzymes under aerobic conditions; and anaerobic activity - for operation of an aerobic enzyme in the absence of oxygen.
- EpPCR (Pritchard et al., J Theor.Biol 234:497-509 (2005)) introduces random point mutations by reducing the fidelity of DNA polymerase in PCR reactions by the addition Of Mn 2+ ions, by biasing dNTP concentrations, or by other conditional variations.
- the five step cloning process to confine the mutagenesis to the target gene of interest involves: 1) error-prone PCR amplification of the gene of interest; 2) restriction enzyme digestion; 3) gel purification of the desired DNA fragment; 4) ligation into a vector; 5) transformation of the gene variants into a suitable host and screening of the library for improved performance.
- This method can generate multiple mutations in a single gene simultaneously, which can be useful.
- a high number of mutants can be generated by EpPCR, so a high-throughput screening assay or a selection method (especially using robotics) is useful to identify those with desirable characteristics.
- Error-prone Rolling Circle Amplification (epRCA) (Fujii et al., Nucleic Acids Res 32:el45 (2004); and Fujii et al., Nat.Protoc. 1 :2493-2497 (2006)) has many of the same elements as epPCR except a whole circular plasmid is used as the template and random 6-mers with exonuclease resistant thiophosphate linkages on the last 2 nucleotides are used to amplify the plasmid followed by transformation into cells in which the plasmid is re-circularized at tandem repeats. Adjusting the Mn 2+ concentration can vary the mutation rate somewhat.
- This technique uses a simple error-prone, single-step method to create a full copy of the plasmid with 3 - 4 mutations/kbp. No restriction enzyme digestion or specific primers are required. Additionally, this method is typically available as a kit.
- DNA or Family Shuffling typically involves digestion of 2 or more variant genes with nucleases such as Dnase I or EndoV to generate a pool of random fragments that are reassembled by cycles of annealing and extension in the presence of DNA polymerase to create a library of chimeric genes. Fragments prime each other and recombination occurs when one copy primes another copy (template switch). This method can be used with >lkbp DNA sequences.
- this method introduces point mutations in the extension steps at a rate similar to error-prone PCR.
- the method can be used to remove deleterious random neutral mutations that might confer antigenicity.
- Staggered Extension (StEP) (Zhao et al., Nat.Biotechnol 16:258-261 (1998)) entails template priming followed by repeated cycles of 2 step PCR with denaturation and very short duration of annealing/extension (as short as 5 sec). Growing fragments anneal to different templates and extend further, which is repeated until full-length sequences are made. Template switching means most resulting fragments have multiple parents. Combinations of low- fidelity polymerases (T aq and Mutazyme) reduce error-prone biases because of opposite mutational spectra.
- Random Priming Recombination random sequence primers are used to generate many short DNA fragments complementary to different segments of the template.
- Base misincorporation and mispriming via epPCR give point mutations. Short DNA fragments prime one another based on homology and are recombined and reassembled into full-length by repeated thermocycling. Removal of templates prior to this step assures low parental recombinants. This method, like most others, can be performed over multiple iterations to evolve distinct properties. This technology avoids sequence bias, is independent of gene length, and requires very little parent DNA for the application.
- Random Chimeragenesis on Transient Templates (RACHITT) (Coco et al., Nat.Biotechnol 19:354-359 (2001)) employs Dnase I fragmentation and size fractionation of ssDNA. Homologous fragments are hybridized in the absence of polymerase to a complementary ssDNA scaffold. Any overlapping unhybridized fragment ends are trimmed down by an exonuclease. Gaps between fragments are filled in, and then ligated to give a pool of full-length diverse strands hybridized to the scaffold (that contains U to preclude amplification). The scaffold then is destroyed and is replaced by a new strand complementary to the diverse strand by PCR amplification.
- the method involves one strand (scaffold) that is from only one parent while the priming fragments derive from other genes; the parent scaffold is selected against. Thus, no reannealing with parental fragments occurs. Overlapping fragments are trimmed with an exonuclease. Otherwise, this is conceptually similar to DNA shuffling and StEP. Therefore, there should be no siblings, few inactives, and no unshuffled parentals. This technique has advantages in that few or no parental genes are created and many more crossovers can result relative to standard DNA shuffling.
- Recombined Extension on Truncated templates entails template switching of unidirectionally growing strands from primers in the presence of unidirectional ssDNA fragments used as a pool oftemplates.
- Recombined Extension on Truncated templates entails template switching of unidirectionally growing strands from primers in the presence of unidirectional ssDNA fragments used as a pool oftemplates.
- No DNA endonucleases are used. Unidirectional ssDNA is made by by DNA polymerase with random primers or serial deletion with exonuclease. Unidirectional ssDNA are only templates and not primers.
- Random priming and exonucleases don't introduce sequence bias as true of enzymatic cleavage of DNA shuffling/RACHITT.
- RETT can be easier to optimize than StEP because it uses normal PCR conditions instead of very short extensions. Recombination occurs as a component of the PCR steps— no direct shuffling. This method can also be more
- ITCHY Incremental Truncation for the Creation of Hybrid Enzymes
- THIO-ITCHY Thio-Incremental Truncation for the Creation of Hybrid Enzymes
- ITCHY Thio-Incremental Truncation for the Creation of Hybrid Enzymes
- phosphothioate dNTPs are used to generate truncations.
- Lutz, S., M. Ostermeier, and S. J. Benkovic, 2001 Rapid generation of incremental truncation libraries for protein engineering using alpha-phosphothioate nucleotides. Nucleic Acids Res 29:E16.
- Relative to ITCHY, THIO-ITCHY can be easier to optimize, provide more reproducibility, and adjustability.
- SCRATCHY - ITCHY combined with DNA shuffling is a combination of DNA shuffling and ITCHY; therefore, allowing multiple crossovers. (Lutz et al., Proc Natl Acad Sci U S.A 98:11248-11253 (2001)).
- SCRATCHY combines the best features of ITCHY and DNA shuffling. Computational predictions can be used in optimization. SCRATCHY is more effective than DNA shuffling when sequence identity is below 80%.
- RNDM Random Drift Mutagenesis
- Sequence Saturation Mutagenesis is a random mutagenesis method that: 1) generates pool of random length fragments using random incorporation of a phosphothioate nucleotide and cleavage; this pool is used as a template to 2) extend in the presence of "universal" bases such as inosine; 3) replication of a inosine-containing complement gives random base incorporation and, consequently, mutagenesis.
- overlapping oligonucleotides are designed to encode "all genetic diversity in targets" and allow a very high diversity for the shuffled progeny.
- Nat.Biotechnol 20: 1251-1255 (2002)
- sequence/codon biases to make more distantly related sequences recombine at rates approaching more closely related sequences and it doesn't require possessing the template genes physically.
- Nucleotide Exchange and Excision Technology NexT exploits a combination of dUTP incorporation followed by treatment with uracil DNA glycosylase and then piperidine to perform endpoint DNA fragmentation.
- the gene is reassembled using internal PCR primer extension with proofreading polymerase.
- the sizes for shuffling are directly controllable using varying dUPT::dTTP ratios. This is an end point reaction using simple methods for uracil incorporation and cleavage.
- One can use other nucleotide analogs such as 8-oxo-guanine with this method. Additionally, the technique works well with very short fragments (86 bp) and has a low error rate. Chemical cleavage of DNA means very few unshuffled clones.
- SHIPREC Sequence Homology-Independent Protein Recombination
- GSSM Gene Site Saturation Mutagenesis
- the starting materials are a supercoiled dsDNA plasmid with insert and 2 primers degenerate at the desired site for mutations.
- Primers carry the mutation of interest and anneal to the same sequence on opposite strands of DNA; mutation in the middle of the primer and ⁇ 20 nucleotides of correct sequence flanking on each side.
- Dpnl is used to digest dam-methylated DNA to eliminate the wild-type template.
- CMCM Combinatorial Multiple Cassette Mutagenesis
- conditional ts mutator plasmids allow increases of 20- to 4000- X in random and natural mutation frequency during selection and to block accumulation of deleterious mutations when selection is not required.
- This technology is based on a plasmid-derived mutD5 gene, which encodes a mutant subunit of DNA polymerase III. This subunit binds to endogenous DNA polymerase III and compromises the proofreading ability of polymerase III in any of the strain that harbors the plasmid. A broad-spectrum of base substitutions and frameshift mutations occur.
- mutator plasmid In order for effective use, the mutator plasmid should be removed once the desired phenotype is achieved; this is accomplished through a temperature sensitive origin of replication, which allows plasmid curing at 41 0 C. It should be noted that mutator strains have been explored for quite some time (e.g., see Winter and coworkers, 1996, J. MoL Biol. 260, 359- 3680. In this technique very high spontaneous mutation rates are observed. The conditional property minimizes non-desired background mutations. This technology could be combined with adaptive evolution to enhance mutagenesis rates and more rapidly achieve desired phenotypes.
- LTM Look-Through Mutagenesis
- Gene Reassembly is a DNA shuffling method that can be applied to multiple genes at one time or to creating a large library of chimeras (multiple mutations) of a single gene, (on the worldwide web at www.verenium.com/Pages/Technology/EnzymeTech/TechEnzyTGR.html)
- this technology is used in combination with ultra-high-throughput screening to query the represented sequence space for desired improvements.
- This technique allows multiple gene recombination independent of homology. The exact number and position of cross-over events can be pre-determined using fragments designed via bioinformatic analysis.
- This technology leads to a very high level of diversity with virtually no parental gene reformation and a low level of inactive genes.
- GSSM a large range of mutations can be tested for improved activity.
- the method allows "blending" and "fine tuning" of DNA shuffling, e.g. codon usage can be optimized.
- Silico Protein Design Automation PDA is an optimization algorithm that anchors the structurally defined protein backbone possessing a particular fold, and searches sequence space for amino acid substitutions that can stabilize the fold and overall protein energetics.
- This technology allows in silico structure-based entropy predictions in order to search for structural tolerance toward protein amino acid variations.
- Statistical mechanics is applied to calculate coupling interactions at each position - structural tolerance toward amino acid substitution is a measure of coupling.
- this technology is designed to yield desired modifications of protein properties while maintaining the integrity of structural characteristics.
- ISM Iterative Saturation Mutagenesis
- Modeling can be utilized to optimize growth conditions. Modeling can also be used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and U.S. Patent No. 7,127,379). Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of isopropanol, n- butanol, or isobutanol.
- OptKnock is a metabolic modeling and simulation program that suggests gene deletion strategies that result in genetically stable microorganisms which overproduce the target product.
- the framework examines the complete metabolic and/or biochemical network of a microorganism in order to suggest genetic manipulations that force the desired biochemical to become an obligatory byproduct of cell growth.
- OptKnock is a term used herein to refer to a computational method and system for modeling cellular metabolism.
- the OptKnock program relates to a framework of models and methods that incorporate particular constraints into flux balance analysis (FBA) models. These constraints include, for example, qualitative kinetic information, qualitative regulatory information, and/or DNA microarray experimental data.
- OptKnock also computes solutions to various metabolic problems by, for example, tightening the flux boundaries derived through flux balance models and subsequently probing the performance limits of metabolic networks in the presence of gene additions or deletions.
- OptKnock computational framework allows the construction of model formulations that enable an effective query of the performance limits of metabolic networks and provides methods for solving the resulting mixed-integer linear programming problems.
- the metabolic modeling and simulation methods referred to herein as OptKnock are described in, for example, U.S. 2002/0168654, WO 2002/055995, and U.S. 2009/0047719.
- SimPheny® Another computational method for identifying and designing metabolic alterations favoring biosynthetic production of a product is a metabolic modeling and simulation system termed SimPheny®. This computational method and system is described in, for example, U.S.
- metabolic modeling and simulation methods include, for example, the computational systems exemplified above as SimPheny® and OptKnock.
- SimPheny® and OptKnock For illustration of the invention, some methods are described herein with reference to the OptKnock computation framework for modeling and simulation.
- OptKnock computation framework for modeling and simulation.
- Those skilled in the art will know how to apply the identification, design and implementation of the metabolic alterations using OptKnock to any of such other metabolic modeling and simulation computational frameworks and methods well known in the art.
- the methods described above will provide one set of metabolic reactions to disrupt. Elimination of each reaction within the set or metabolic modification can result in a desired product as an obligatory product during the growth phase of the organism. Because the reactions are known, a solution to the bilevel OptKnock problem also will provide the associated gene or genes encoding one or more enzymes that catalyze each reaction within the set of reactions. Identification of a set of reactions and their corresponding genes encoding the enzymes participating in each reaction is generally an automated process, accomplished through correlation of the reactions with a reaction database having a relationship between enzymes and encoding genes.
- the set of reactions that are to be disrupted in order to achieve production of a desired product are implemented in the target cell or organism by functional disruption of at least one gene encoding each metabolic reaction within the set.
- One particularly useful means to achieve functional disruption of the reaction set is by deletion of each encoding gene.
- These latter aberrations, resulting in less than total deletion of the gene set can be useful, for example, when rapid assessments of the coupling of a product are desired or when genetic reversion is less likely to occur.
- an optimization method termed integer cuts. This method proceeds by iteratively solving the OptKnock problem exemplified above with the incorporation of an additional constraint referred to as an integer cut at each iteration. Integer cut constraints effectively prevent the solution procedure from choosing the exact same set of reactions identified in any previous iteration that obligatorily couples product biosynthesis to growth. For example, if a previously identified growth-coupled metabolic modification specifies reactions 1, 2, and 3 for disruption, then the following constraint prevents the same reactions from being simultaneously considered in subsequent solutions.
- the integer cut method is well known in the art and can be found described in, for example, Burgard et al., Biotechnol. Prog. 17:791-797 (2001). As with all methods described herein with reference to their use in combination with the OptKnock computational framework for metabolic modeling and simulation, the integer cut method of reducing redundancy in iterative computational analysis also can be applied with other computational frameworks well known in the art including, for example, SimPheny®.
- the methods exemplified herein allow the construction of cells and organisms that biosynthetically produce a desired product, including the obligatory coupling of production of a target biochemical product to growth of the cell or organism engineered to harbor the identified genetic alterations. Therefore, the computational methods described herein allow the identification and implementation of metabolic modifications that are identified by an in silico method selected from OptKnock or SimPheny®.
- the set of metabolic modifications can include, for example, addition of one or more biosynthetic pathway enzymes and/or functional disruption of one or more metabolic reactions including, for example, disruption by gene deletion.
- An in silico stoichiometric model of E. coli metabolism can be employed to identify essential genes for metabolic pathways as exemplified previously and described in, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and in U.S. Patent No. 7,127,379.
- the OptKnock mathematical framework can be applied to pinpoint gene deletions leading to the growth-coupled production of a desired product. Further, the solution of the bilevel OptKnock problem provides only one set of deletions.
- integer cuts an optimization technique, termed integer cuts. This entails iteratively solving the OptKnock problem with the incorporation of an additional constraint referred to as an integer cut at each iteration, as discussed above.
- This example describes the generation of a microbial organism capable of producing isopropanol from 4-hydroxybutyryl-CoA.
- Escherichia coli is used as a target organism to engineer the isopropanol pathway shown in Figure 1 that starts from 4-hydroxybutyryl-CoA.
- E. coli provides a good host for generating a non-naturally occurring microorganism capable of producing isopropanol.
- E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
- nucleic acids encoding the enzymes utilized in the pathway are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999).
- sucD YP_001396394
- 4hbd YP_001396393
- bukl Q45829
- an ⁇ ptb NP_349676
- succinic semialdehyde dehydrogenase CoA-dependent
- A- hydroxybutyrate dehydrogenase 4-hydroxybutyrate kinase
- phosphotransbutyrylase activities are cloned into an expression vector or integrated into the chromosome as described in Burk et al. (U.S. publication 2009/0075351).
- abfD P55792
- crtl NP_349318.1 genes encoding 4-hydroxybutyryl-CoA dehydratase, vinylacetyl-CoA ⁇ - isomerase, and enoyl-CoA hydratase activities, respectively, are cloned into the pZE13 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- hbd NP_349314.1
- atoAD P76459.1, P76458.1
- pZS23 vector Expressys, Ruelzheim, Germany
- adc NP_149328.1
- adh AAA23199.2 genes encoding acetoacetate decarboxylase and acetone reductase activities are cloned into the pZS13 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- pZS23 is obtained by replacing the ampicillin resistance module of the pZS13 vector (Expressys, Ruelzheim, Germany) with a kanamycin resistance module by well- known molecular biology techniques.
- the three sets of plasmids are transformed into a 4- hydroxybutyryl-CoA producing strain of E. coli to express the proteins and enzymes required for isopropanol synthesis from 4-hydroxybutyryl-CoA.
- the resulting genetically engineered organism is cultured in glucose-containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). Cobalamin is also supplied to the medium to ensure activity of the mutase enzyme unless the host strain of E. coli is engineered to synthesize cobalamin de novo (see, for example, Raux et al., J. Bacteriol. 178:753-767 (1996)).
- the expression of the isopropanol synthesis genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including for example, Northern blots, PCR amplification of mRNA, immunoblotting, and the like.
- Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individual activities.
- the ability of the engineered E. coli strain to produce isopropanol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) and/or liquid chromatography-mass spectrometry (LCMS).
- Microbial strains engineered to have a functional isopropanol synthesis pathway are further augmented by optimization for efficient utilization of the pathway. Briefly, the engineered strain is assessed to determine whether any of the exogenous genes are expressed at a rate limiting level. Expression is increased for any enzymes expressed at low levels that can limit the flux through the pathway by, for example, introduction of additional gene copy numbers.
- Modeling is also used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and U.S. Patent No. 7,127,379).
- Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of isopropanol.
- One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer.
- Adaptive evolution also can be used to generate better producers of, for example, the 4-hydroxybutyryl-CoA intermediate of the isopropanol product. Adaptive evolution is performed to improve both growth and production characteristics (Fong and Palsson, Nat. Genet. 36:1056-1058 (2004); Alper et al., Science 314: 1565-1568 (2006)). Based on the results, subsequent rounds of modeling, genetic engineering and adaptive evolution can be applied to the isopropanol producer to further increase production.
- the above organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing the culture vessel, for example, flasks can be sealed with a septum and crimp-cap. Microaerobic conditions also can be utilized by providing a small hole in the septum for limited aeration. The pH of the medium is maintained at a pH of around 7 by addition of an acid, such as H 2 SO 4 .
- an acid such as H 2 SO 4 .
- the growth rate is determined by measuring optical density using a spectrophotometer (600 nm) and the glucose uptake rate by monitoring carbon source depletion over time.
- Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu, Columbia MD), for example, using an Aminex® series of HPLC columns (for example, HPX-87 series) (BioRad, Hercules CA), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng. 775-779 (2005)).
- Escherichia coli is used as a target organism to engineer the butanol pathway shown in Figure 1 that starts from 4-hydroxybutyryl-CoA.
- E. coli provides a good host for generating a non- naturally occurring microorganism capable of producing butanol.
- E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
- nucleic acids encoding the enzymes utilized in the pathway are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999).
- sucD (YP_001396394), 4hbd (YP_001396393), bukl (Q45829), zn ⁇ ptb (NP_349676) genes encoding succinic semialdehyde dehydrogenase (CoA-dependent), 4- hydroxybutyrate dehydrogenase, 4-hydroxybutyrate kinase, and phosphotransbutyrylase activities, respectively, are cloned into an expression vector or integrated into the chromosome as described in Burk et al. (U.S. publication 2009/0075351).
- the abfD (P55792) gene encoding 4-hydroxybutyryl-CoA dehydratase and vinylacetyl-CoA ⁇ -isomerase activities is cloned into the pZE13 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- the bed (NP_349317.1) and etfAB (NP_349315.1, NPJ49316.1) genes encoding crotonyl-CoA reductase activity are cloned into the pZS23 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- the aid (AAT66436) and adhl (AAR91477.1) genes encoding butyryl-CoA reductase (aldehyde forming) and butyraldehyde reductase activities are cloned into the pZS13 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- pZS23 is obtained by replacing the ampicillin resistance module of the pZS13 vector (Expressys, Ruelzheim, Germany) with a kanamycin resistance module by well- known molecular biology techniques.
- the three sets of plasmids are transformed into a 4- hydroxybutyryl-CoA producing strain of is. coli to express the proteins and enzymes required for butanol synthesis from 4-hydroxybutyryl-CoA.
- the resulting genetically engineered organism is cultured in glucose-containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). Cobalamin is also supplied to the medium to ensure activity of the mutase enzyme unless the host strain of is. coli is engineered to synthesize cobalamin de novo (see, for example, Raux et al., J. Bacteriol. 178:753-767 (1996)).
- the expression of the butanol synthesis genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including for example, Northern blots, PCR amplification of mRNA, immunoblotting, and the like.
- Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individual activities.
- the ability of the engineered E. coli strain to produce butanol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) and/or liquid chromatography-mass spectrometry (LCMS).
- GCMS gas chromatography-mass spectrometry
- LCMS liquid chromatography-mass spectrometry
- Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of butanol.
- One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer. 84:647-657 (2003)), which is applied to select gene knockouts that collectively result in better production of butanol.
- Adaptive evolution also can be used to generate better producers of, for example, the 4-hydroxybutyryl-CoA intermediate of the butanol product.
- Adaptive evolution is performed to improve both growth and production characteristics (Fong and Palsson, Nat. Genet. 36:1056-1058 (2004); Alper et al., Science 314:1565-1568 (2006)). Based on the results, subsequent rounds of modeling, genetic engineering and adaptive evolution can be applied to the butanol producer to further increase production.
- the above organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing the culture vessel, for example, flasks can be sealed with a septum and crimp-cap. Microaerobic conditions also can be utilized by providing a small hole in the septum for limited aeration. The pH of the medium is maintained at a pH of around 7 by addition of an acid, such as H 2 SO 4 .
- an acid such as H 2 SO 4 .
- the growth rate is determined by measuring optical density using a spectrophotometer (600 nm) and the glucose uptake rate by monitoring carbon source depletion over time.
- Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu, Columbia MD), for example, using an Aminex® series of HPLC columns (for example, HPX-87 series) (BioRad, Hercules CA), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng. 775-779 (2005)).
- This example describes the generation of a microbial organism capable of producing isobutanol from 4-hydroxybutyryl-CoA.
- Escherichia coli is used as a target organism to engineer the isobutanol pathway shown in Figure 1 that starts from 4-hydroxybutyryl-CoA.
- E. coli provides a good host for generating a non- naturally occurring microorganism capable of producing isobutanol.
- E. coli is amenable to genetic manipulation and is known to be capable of producing various products, like ethanol, acetic acid, formic acid, lactic acid, and succinic acid, effectively under anaerobic or microaerobic conditions.
- nucleic acids encoding the enzymes utilized in the pathway are expressed in E. coli using well known molecular biology techniques (see, for example, Sambrook, supra, 2001; Ausubel supra, 1999).
- sucD YP_001396394
- 4hbd YP_001396393
- bukl Q45829
- an ⁇ ptb NP_349676
- succinic semialdehyde dehydrogenase CoA-dependent
- A- hydroxybutyrate dehydrogenase 4-hydroxybutyrate kinase
- phosphotransbutyrylase activities are cloned into an expression vector or integrated into the chromosome as described in Burk et al. (U.S. publication 2009/0075351).
- abfD P55792
- icm AAC08713.1
- icmB CAB59633.1
- the bed (NP_349317.1) and etfAB (NP_349315.1, NP_349316.1) genes encoding crotonyl-CoA reductase activity are cloned into the pZS23 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- the adhE (AAV66076.1) gene encoding isobutyryl-CoA reductase (alcohol forming) activity is cloned into the pZS13 vector (Expressys, Ruelzheim, Germany) under the PAl/lacO promoter.
- pZS23 is obtained by replacing the ampicillin resistance module of the pZS13 vector (Expressys, Ruelzheim, Germany) with a kanamycin resistance module by well-known molecular biology techniques.
- the three sets of plasmids are transformed into a A- hydroxybutyryl-CoA producing strain of is. coli to express the proteins and enzymes required for isobutanol synthesis from 4-hydroxybutyryl-CoA.
- the resulting genetically engineered organism is cultured in glucose-containing medium following procedures well known in the art (see, for example, Sambrook et al., supra, 2001). Cobalamin is also supplied to the medium to ensure activity of the mutase enzyme unless the host strain of E. coli is engineered to synthesize cobalamin de novo (see, for example, Raux et al., J. Bacteriol. 178:753-767 (1996)).
- the expression of the isobutanol synthesis genes is corroborated using methods well known in the art for determining polypeptide expression or enzymatic activity, including for example, Northern blots, PCR amplification of mRNA, immunoblotting, and the like.
- Enzymatic activities of the expressed enzymes are confirmed using assays specific for the individual activities.
- the ability of the engineered E. coli strain to produce isobutanol is confirmed using HPLC, gas chromatography-mass spectrometry (GCMS) and/or liquid chromatography-mass spectrometry (LCMS).
- Microbial strains engineered to have a functional isobutanol synthesis pathway are further augmented by optimization for efficient utilization of the pathway. Briefly, the engineered strain is assessed to determine whether any of the exogenous genes are expressed at a rate limiting level. Expression is increased for any enzymes expressed at low levels that can limit the flux through the pathway by, for example, introduction of additional gene copy numbers.
- Modeling is also used to design gene knockouts that additionally optimize utilization of the pathway (see, for example, U.S. patent publications US 2002/0012939, US 2003/0224363, US 2004/0029149, US 2004/0072723, US 2003/0059792, US 2002/0168654 and US 2004/0009466, and U.S. Patent No. 7,127,379).
- Modeling analysis allows reliable predictions of the effects on cell growth of shifting the metabolism towards more efficient production of isobutanol.
- One modeling method is the bilevel optimization approach, OptKnock (Burgard et al., Biotechnol. Bioengineer.
- the above organism is cultured in a fermenter using a medium known in the art to support growth of the organism under anaerobic conditions. Fermentations are performed in either a batch, fed-batch or continuous manner. Anaerobic conditions are maintained by first sparging the medium with nitrogen and then sealing the culture vessel, for example, flasks can be sealed with a septum and crimp-cap. Microaerobic conditions also can be utilized by providing a small hole in the septum for limited aeration. The pH of the medium is maintained at a pH of around 7 by addition of an acid, such as H2SO4.
- an acid such as H2SO4.
- the growth rate is determined by measuring optical density using a spectrophotometer (600 nm) and the glucose uptake rate by monitoring carbon source depletion over time.
- Byproducts such as undesirable alcohols, organic acids, and residual glucose can be quantified by HPLC (Shimadzu, Columbia MD), for example, using an Aminex® series of HPLC columns (for example, HPX-87 series) (BioRad, Hercules CA), using a refractive index detector for glucose and alcohols, and a UV detector for organic acids (Lin et al., Biotechnol. Bioeng. 775-779 (2005)).
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Plant Pathology (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Virology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2010242849A AU2010242849A1 (en) | 2009-04-30 | 2010-04-30 | Organisms for the production of isopropanol, n-butanol, and isobutanol |
| JP2012508799A JP2012525156A (en) | 2009-04-30 | 2010-04-30 | Microorganisms for the production of isopropanol, n-butanol, and isobutanol |
| EP10770455A EP2425006A4 (en) | 2009-04-30 | 2010-04-30 | Organisms for the production of isopropanol, n-butanol, and isobutanol |
| BRPI1013505A BRPI1013505A2 (en) | 2009-04-30 | 2010-04-30 | organisms for the production of isopropanol, n-butanol, and isobutanol |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US17448209P | 2009-04-30 | 2009-04-30 | |
| US61/174,482 | 2009-04-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010127303A1 true WO2010127303A1 (en) | 2010-11-04 |
Family
ID=43032589
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2010/033271 WO2010127303A1 (en) | 2009-04-30 | 2010-04-30 | Organisms for the production of isopropanol, n-butanol, and isobutanol |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8993285B2 (en) |
| EP (1) | EP2425006A4 (en) |
| JP (1) | JP2012525156A (en) |
| KR (1) | KR20120068763A (en) |
| AU (1) | AU2010242849A1 (en) |
| BR (1) | BRPI1013505A2 (en) |
| WO (1) | WO2010127303A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2373781A1 (en) * | 2008-12-16 | 2011-10-12 | Genomatica, Inc. | Microorganisms and methods for conversion of syngas and other carbon sources to useful products |
| WO2011057871A3 (en) * | 2009-11-11 | 2012-01-26 | Evonik Röhm Gmbh | Use of a protein homologous to a meab protein for increasing the enzymatic activity of a 3-hydroxycarboxylic acid-coa mutase |
| US8728804B2 (en) | 2010-10-29 | 2014-05-20 | Novozymes A/S | Polypeptides having succinyl-CoA: acetoacetate transferase activity and polynucleotides encoding same |
| WO2014152434A2 (en) | 2013-03-15 | 2014-09-25 | Genomatica, Inc. | Microorganisms and methods for producing butadiene and related compounds by formate assimilation |
| WO2015084633A1 (en) | 2013-12-03 | 2015-06-11 | Genomatica, Inc. | Microorganisms and methods for improving product yields on methanol using acetyl-coa synthesis |
| WO2016044713A1 (en) | 2014-09-18 | 2016-03-24 | Genomatica, Inc. | Non-natural microbial organisms with improved energetic efficiency |
| WO2019152375A1 (en) | 2018-01-30 | 2019-08-08 | Genomatica, Inc. | Fermentation systems and methods with substantially uniform volumetric uptake rate of a reactive gaseous component |
| US10597684B2 (en) | 2013-12-27 | 2020-03-24 | Genomatica, Inc. | Methods and organisms with increased carbon flux efficiencies |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8129154B2 (en) | 2008-06-17 | 2012-03-06 | Genomatica, Inc. | Microorganisms and methods for the biosynthesis of fumarate, malate, and acrylate |
| US20100184173A1 (en) * | 2008-11-14 | 2010-07-22 | Genomatica, Inc. | Microorganisms for the production of methyl ethyl ketone and 2-butanol |
| WO2010144746A2 (en) | 2009-06-10 | 2010-12-16 | Genomatica, Inc. | Microorganisms and methods for carbon-efficient biosynthesis of mek and 2-butanol |
| CN109136161A (en) | 2009-12-10 | 2019-01-04 | 基因组股份公司 | Synthesis gas or other gaseous carbon sources and methanol are converted into the method and organism of 1,3 butylene glycol |
| US8445244B2 (en) | 2010-02-23 | 2013-05-21 | Genomatica, Inc. | Methods for increasing product yields |
| WO2012116338A1 (en) * | 2011-02-25 | 2012-08-30 | The Ohio State University Research Foundation | Autotrophic hydrogen bacteria and uses thereof |
| WO2012177601A2 (en) * | 2011-06-22 | 2012-12-27 | Genomatica, Inc. | Microorganisms for producing isobutanol and methods related thereto |
| WO2012177599A2 (en) * | 2011-06-22 | 2012-12-27 | Genomatica, Inc. | Microorganisms for producing n-propanol 1, 3-propanediol, 1,2-propanediol or glycerol and methods related thereto |
| US9914947B2 (en) * | 2011-07-27 | 2018-03-13 | Alliance For Sustainable Energy, Llc | Biological production of organic compounds |
| EP3404094A1 (en) | 2012-03-06 | 2018-11-21 | Lygos, Inc. | Recombinant host cells for the production of malonate |
| JP6487325B2 (en) | 2012-10-08 | 2019-03-20 | キャリスタ, インコーポレイテッド | GAS-FED fermentation system |
| WO2014071286A1 (en) | 2012-11-05 | 2014-05-08 | Genomatica, Inc. | Microorganisms for enhancing the availability of reducing equivalents in the presence of methanol, and for producing 1,2-propanediol |
| US9469860B2 (en) * | 2013-01-18 | 2016-10-18 | Synata Bio, Inc. | Method for production of n-butanol from syngas using syntrophic co-cultures of anaerobic microorganisms |
| US20140206066A1 (en) * | 2013-01-18 | 2014-07-24 | Coskata, Inc. | Syntrophic co-culture of anaerobic microorganism for production of n-butanol from syngas |
| US11541105B2 (en) | 2018-06-01 | 2023-01-03 | The Research Foundation For The State University Of New York | Compositions and methods for disrupting biofilm formation and maintenance |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080293125A1 (en) * | 2007-04-18 | 2008-11-27 | Gevo, Inc. | Engineered microorganisms for producing isopropanol |
| US20090047718A1 (en) * | 2007-05-17 | 2009-02-19 | Blaschek Hans P | Methods and compositions for producing solvents |
| US20090075351A1 (en) * | 2007-03-16 | 2009-03-19 | Burk Mark J | Compositions and methods for the biosynthesis of 1,4-butanediol and its precursors |
Family Cites Families (141)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3513209A (en) | 1968-08-19 | 1970-05-19 | Du Pont | Method of making 1,4-cyclohexadiene |
| US4076948A (en) | 1968-10-10 | 1978-02-28 | El Paso Products Company | Process for treatment of adipic acid mother liquor |
| GB1230276A (en) | 1968-12-09 | 1971-04-28 | ||
| US3965182A (en) | 1969-10-02 | 1976-06-22 | Ethyl Corporation | Preparation of aniline from phenol and ammonia |
| JPS506776Y1 (en) | 1969-12-27 | 1975-02-26 | ||
| JPS4831084B1 (en) | 1970-09-04 | 1973-09-26 | ||
| GB1344557A (en) | 1972-06-23 | 1974-01-23 | Mitsubishi Petrochemical Co | Process for preparing 1,4-butanediol |
| JPS5543759B2 (en) | 1972-06-28 | 1980-11-07 | ||
| DE2455617C3 (en) | 1974-11-23 | 1982-03-18 | Basf Ag, 6700 Ludwigshafen | Process for the production of butanediol and / or tetrahydrofuran via the intermediate stage of γ-butyrolactone |
| DE2501499A1 (en) | 1975-01-16 | 1976-07-22 | Hoechst Ag | PROCESS FOR THE PRODUCTION OF BUTANDIOL- (1.4) |
| US4190495A (en) | 1976-09-27 | 1980-02-26 | Research Corporation | Modified microorganisms and method of preparing and using same |
| US4301077A (en) | 1980-12-22 | 1981-11-17 | Standard Oil Company | Process for the manufacture of 1-4-butanediol and tetrahydrofuran |
| IT1190783B (en) | 1981-04-29 | 1988-02-24 | Davy Mckee Oil & Chem | PROCESS FOR HYDROGENOLYSIS OF ESTERS OF CARBOXYLIC ACIDS |
| JPS6070088A (en) * | 1983-09-26 | 1985-04-20 | Agency Of Ind Science & Technol | Production of isopropanol |
| JPS60114197A (en) | 1983-11-25 | 1985-06-20 | Agency Of Ind Science & Technol | Preparation of dicarboxylic acid by bacterium |
| US4871667A (en) | 1984-11-26 | 1989-10-03 | Agency Of Industrial Science & Technology | Process for preparing muconic acid |
| US4652685A (en) | 1985-11-15 | 1987-03-24 | General Electric Company | Hydrogenation of lactones to glycols |
| EP0249773B1 (en) | 1986-06-11 | 1992-12-16 | Michigan Biotechnology Institute | A process for the production of succinic acid by anaerobic fermentation |
| US5143834A (en) | 1986-06-11 | 1992-09-01 | Glassner David A | Process for the production and purification of succinic acid |
| US5168055A (en) | 1986-06-11 | 1992-12-01 | Rathin Datta | Fermentation and purification process for succinic acid |
| US5182199A (en) | 1987-05-27 | 1993-01-26 | Hartley Brian S | Thermophilic ethanol production in a two-stage closed system |
| EP0633319B1 (en) | 1988-04-27 | 1999-03-17 | Daicel Chemical Industries, Ltd. | Process for producing optically active 1,3-butanediol |
| EP0470172B1 (en) | 1989-04-27 | 1995-06-28 | BioControl Systems, Incorporated | Precipitate test for microorganisms |
| WO1991013997A1 (en) | 1990-03-09 | 1991-09-19 | Basf K&F Corporation | Process for the production of natural long-chain alcohols |
| US5192673A (en) | 1990-04-30 | 1993-03-09 | Michigan Biotechnology Institute | Mutant strain of C. acetobutylicum and process for making butanol |
| US5079143A (en) | 1990-05-02 | 1992-01-07 | The Upjohn Company | Method of indentifying compounds useful as antiparasitic drugs |
| US5173429A (en) | 1990-11-09 | 1992-12-22 | The Board Of Trustees Of The University Of Arkansas | Clostridiumm ljungdahlii, an anaerobic ethanol and acetate producing microorganism |
| IL100572A (en) | 1991-01-03 | 1997-01-10 | Lepetit Spa | Amides of antibiotic ge 2270 factors their preparation and pharmaceutical compositions containing them |
| US5416020A (en) | 1992-09-29 | 1995-05-16 | Bio-Technical Resources | Lactobacillus delbrueckii ssp. bulgaricus strain and fermentation process for producing L-(+)-lactic acid |
| US5807722A (en) | 1992-10-30 | 1998-09-15 | Bioengineering Resources, Inc. | Biological production of acetic acid from waste gases with Clostridium ljungdahlii |
| US6136577A (en) | 1992-10-30 | 2000-10-24 | Bioengineering Resources, Inc. | Biological production of ethanol from waste gases with Clostridium ljungdahlii |
| FR2702492B1 (en) | 1993-03-12 | 1995-05-24 | Rhone Poulenc Chimie | Production process by fermentation of itaconic acid. |
| US5487987A (en) | 1993-09-16 | 1996-01-30 | Purdue Research Foundation | Synthesis of adipic acid from biomass-derived carbon sources |
| US5521075A (en) | 1994-12-19 | 1996-05-28 | Michigan Biotechnology Institute | Method for making succinic acid, anaerobiospirillum succiniciproducens variants for use in process and methods for obtaining variants |
| US5504004A (en) | 1994-12-20 | 1996-04-02 | Michigan Biotechnology Institute | Process for making succinic acid, microorganisms for use in the process and methods of obtaining the microorganisms |
| US5700934A (en) | 1995-03-01 | 1997-12-23 | Dsm N.V. | Process for the preparation of epsilon-caprolactam and epsilon-caprolactam precursors |
| US5478952A (en) | 1995-03-03 | 1995-12-26 | E. I. Du Pont De Nemours And Company | Ru,Re/carbon catalyst for hydrogenation in aqueous solution |
| US5863782A (en) | 1995-04-19 | 1999-01-26 | Women's And Children's Hospital | Synthetic mammalian sulphamidase and genetic sequences encoding same |
| US5686276A (en) | 1995-05-12 | 1997-11-11 | E. I. Du Pont De Nemours And Company | Bioconversion of a fermentable carbon source to 1,3-propanediol by a single microorganism |
| FR2736927B1 (en) | 1995-07-18 | 1997-10-17 | Rhone Poulenc Fibres & Polymer | ENZYMES HAVING AMIDASE ACTIVITY, GENETIC TOOLS AND HOST MICROORGANISMS FOR OBTAINING SAME AND HYDROLYSIS PROCESS USING THE SAME |
| US5573931A (en) | 1995-08-28 | 1996-11-12 | Michigan Biotechnology Institute | Method for making succinic acid, bacterial variants for use in the process, and methods for obtaining variants |
| US5770435A (en) | 1995-11-02 | 1998-06-23 | University Of Chicago | Mutant E. coli strain with increased succinic acid production |
| US5869301A (en) | 1995-11-02 | 1999-02-09 | Lockhead Martin Energy Research Corporation | Method for the production of dicarboxylic acids |
| EP0883683A1 (en) | 1996-02-27 | 1998-12-16 | Michigan State University | Cloning and expression of the gene encoding thermoanaerobacter ethanolicus 39e secondary-alcohol dehydrogenase and enzyme biochemical characterization |
| US5958745A (en) | 1996-03-13 | 1999-09-28 | Monsanto Company | Methods of optimizing substrate pools and biosynthesis of poly-β-hydroxybutyrate-co-poly-β-hydroxyvalerate in bacteria and plants |
| DE69638265D1 (en) | 1996-07-01 | 2010-11-11 | Emmaus Foundation Inc | BIOLOGICAL PREPARATION OF ACETIC ACID FROM EXHAUST GASES |
| KR100459818B1 (en) | 1996-09-02 | 2004-12-03 | 이. 아이. 두퐁 드 느무르 앤드 컴퍼니 | Process for the preparation of epsilon-caprolactam |
| US6117658A (en) | 1997-02-13 | 2000-09-12 | James Madison University | Methods of making polyhydroxyalkanoates comprising 4-hydroxybutyrate monomer units |
| KR100516986B1 (en) | 1997-02-19 | 2005-09-26 | 코닌클리즈케 디에스엠 엔.브이. | Process for the preparation of caprolactam in the absence of catalysts by contacting 6-aminocaproic acid derivatives with superheated steam |
| US6274790B1 (en) | 1997-04-14 | 2001-08-14 | The University Of British Columbia | Nucleic acids encoding a plant enzyme involved in very long chain fatty acid synthesis |
| KR19990013007A (en) | 1997-07-31 | 1999-02-25 | 박원훈 | Transformed Escherichia Coli S373 (BTCC 8818 P) and Production Method of Succinic Acid Using the Same |
| JPH11103863A (en) | 1997-10-08 | 1999-04-20 | Nippon Shokubai Co Ltd | Maleate isomerase gene |
| US6280986B1 (en) | 1997-12-01 | 2001-08-28 | The United States Of America As Represented By The Secretary Of Agriculture | Stabilization of pet operon plasmids and ethanol production in bacterial strains lacking lactate dehydrogenase and pyruvate formate lyase activities |
| MY121380A (en) | 1998-04-13 | 2006-01-28 | Univ Georgia | Pyruvate carboxylase overexpression for enhanced production of oxaloacetate-derived biochemicals in microbial cells |
| US20030087381A1 (en) | 1998-04-13 | 2003-05-08 | University Of Georgia Research Foundation, Inc. | Metabolically engineered organisms for enhanced production of oxaloacetate-derived biochemicals |
| US6159738A (en) | 1998-04-28 | 2000-12-12 | University Of Chicago | Method for construction of bacterial strains with increased succinic acid production |
| US6432686B1 (en) | 1998-05-12 | 2002-08-13 | E. I. Du Pont De Nemours And Company | Method for the production of 1,3-propanediol by recombinant organisms comprising genes for vitamin B12 transport |
| US6444784B1 (en) | 1998-05-29 | 2002-09-03 | Exxonmobil Research & Engineering Company | Wax crystal modifiers (LAW657) |
| DE19856136C2 (en) | 1998-12-04 | 2002-10-24 | Pasteur Institut | Method and device for the selection of accelerated proliferation of living cells in suspension |
| EP1147229A2 (en) | 1999-02-02 | 2001-10-24 | Bernhard O. Palsson | Methods for identifying drug targets based on genomic sequence data |
| US6686310B1 (en) | 1999-02-09 | 2004-02-03 | E. I. Du Pont De Nemours And Company | High surface area sol-gel route prepared hydrogenation catalysts |
| US6365376B1 (en) | 1999-02-19 | 2002-04-02 | E. I. Du Pont De Nemours And Company | Genes and enzymes for the production of adipic acid intermediates |
| WO2000052183A1 (en) | 1999-03-05 | 2000-09-08 | Monsanto Technology Llc | Multigene expression vectors for the biosynthesis of products via multienzyme biological pathways |
| CA2374482C (en) | 1999-05-21 | 2012-09-18 | Cargill Dow Llc | Methods and materials for the synthesis of organic products |
| ATE338139T1 (en) | 1999-08-30 | 2006-09-15 | Wisconsin Alumni Res Found | PRODUCTION OF 3-HYDROXYPROPIONIC ACID IN RECOMBINANT ORGANISMS |
| US6852517B1 (en) | 1999-08-30 | 2005-02-08 | Wisconsin Alumni Research Foundation | Production of 3-hydroxypropionic acid in recombinant organisms |
| US6660857B2 (en) | 2000-02-03 | 2003-12-09 | Dsm N.V. | Process for the preparation of ε-caprolactam |
| US6878861B2 (en) | 2000-07-21 | 2005-04-12 | Washington State University Research Foundation | Acyl coenzyme A thioesterases |
| BR0112251B1 (en) | 2000-07-25 | 2013-04-09 | Continuous methods for ethanol production from anaerobic bacterial fermentation of a gaseous substrate. | |
| MXPA03004324A (en) | 2000-11-20 | 2004-01-26 | Cargill Inc | 3-hydroxypropionic acid and other organic compounds. |
| CN1955299A (en) | 2000-11-22 | 2007-05-02 | 内特尔沃克公司 | Methods and materials for the synthesis of organic products |
| CN1358841A (en) | 2000-12-11 | 2002-07-17 | 云南省微生物研究所 | Yunnan streptin |
| CA2433529A1 (en) | 2000-12-28 | 2002-07-11 | Toyota Jidosha Kabushiki Kaisha | Process for producing prenyl alcohol |
| JP4776146B2 (en) | 2001-01-10 | 2011-09-21 | ザ・ペン・ステート・リサーチ・ファンデーション | Method and system for modeling cellular metabolism |
| US7127379B2 (en) | 2001-01-31 | 2006-10-24 | The Regents Of The University Of California | Method for the evolutionary design of biochemical reaction networks |
| EP1381860A4 (en) | 2001-03-01 | 2008-10-15 | Univ California | Models and methods for determining systemic properties of regulated reaction networks |
| US6743610B2 (en) | 2001-03-30 | 2004-06-01 | The University Of Chicago | Method to produce succinic acid from raw hydrolysates |
| ATE393137T1 (en) | 2001-05-07 | 2008-05-15 | Cargill Inc | METHOD FOR PRODUCING CARBOXYLIC ACIDS AND THEIR DERIVATIVES |
| WO2003010322A1 (en) | 2001-07-20 | 2003-02-06 | Youming Zhang | Improved rect or recet cloning and subcloning method |
| CA2356540A1 (en) | 2001-08-30 | 2003-02-28 | Emory University | Expressed dna sequences involved in mitochondrial functions |
| US7256016B2 (en) | 2001-11-02 | 2007-08-14 | Rice University | Recycling system for manipulation of intracellular NADH availability |
| JP4532116B2 (en) | 2002-01-18 | 2010-08-25 | ノボザイムズ エー/エス | Alanine 2,3-aminomutase |
| EP1473368A4 (en) | 2002-02-06 | 2006-01-25 | Showa Denko Kk | Alpha-substituted-alpha, beta-unsaturated carbonyl compound reductase gene |
| US20030224363A1 (en) | 2002-03-19 | 2003-12-04 | Park Sung M. | Compositions and methods for modeling bacillus subtilis metabolism |
| AU2003222128A1 (en) | 2002-03-29 | 2003-10-13 | Genomatica, Inc. | Human metabolic models and methods |
| WO2003095651A1 (en) | 2002-05-10 | 2003-11-20 | Kyowa Hakko Kogyo Co., Ltd. | Process for producing mevalonic acid |
| US7856317B2 (en) | 2002-06-14 | 2010-12-21 | Genomatica, Inc. | Systems and methods for constructing genomic-based phenotypic models |
| ATE362544T1 (en) | 2002-06-14 | 2007-06-15 | Dsm Ip Assets Bv | POLYPEPTIDES WITH ALPHA-H ALPHA AMINO ACID AMIDE RACEMASE ACTIVITY AND NUCLEIC ACIDS CODING THERETO |
| US7826975B2 (en) | 2002-07-10 | 2010-11-02 | The Penn State Research Foundation | Method for redesign of microbial production systems |
| US8027821B2 (en) | 2002-07-10 | 2011-09-27 | The Penn State Research Foundation | Method for determining gene knockouts |
| US7413878B2 (en) | 2002-07-15 | 2008-08-19 | Kosan Biosciences, Inc. | Recombinant host cells expressing atoAD and capable of making a polyketide using a starter unit |
| KR101148255B1 (en) | 2002-10-04 | 2012-08-08 | 다니스코 유에스 인크. | Process for the biological production of 1,3-propanediol with high yield |
| EP1552472A4 (en) | 2002-10-15 | 2008-02-20 | Univ California | Methods and systems to identify operational reaction pathways |
| WO2004044210A2 (en) | 2002-11-06 | 2004-05-27 | University Of Florida | Materials and methods for the efficient production of acetate and other products |
| US7432091B2 (en) | 2003-02-24 | 2008-10-07 | Research Institute Of Innovative Technology For The Earth | Highly efficient hydrogen production method using microorganism |
| RU2268300C2 (en) | 2003-04-07 | 2006-01-20 | Закрытое акционерное общество "Научно-исследовательский институт Аджиномото-Генетика" | METHOD FOR PREPARING L-AMINO ACIDS BY USING MICROORGANISMS POSSESSING ENHANCED EXPRESSION OF pckA GENE |
| US7927859B2 (en) | 2003-08-22 | 2011-04-19 | Rice University | High molar succinate yield bacteria by increasing the intracellular NADH availability |
| CN1852978A (en) | 2003-09-17 | 2006-10-25 | 三菱化学株式会社 | Process for producing non-amino organic acid |
| WO2005026338A1 (en) | 2003-09-18 | 2005-03-24 | Ciba Specialty Chemicals Holding Inc. | Alcohol dehydrogenases with increased solvent and temperature stability |
| FR2862068B1 (en) | 2003-11-06 | 2007-10-12 | Metabolic Explorer Sa | OPTIMIZED MICROORGANISM STRAINS FOR NADPH CONSUMPTION BIOSYNTHESIS PATHWAYS |
| US7244610B2 (en) | 2003-11-14 | 2007-07-17 | Rice University | Aerobic succinate production in bacteria |
| FR2864967B1 (en) | 2004-01-12 | 2006-05-19 | Metabolic Explorer Sa | ADVANCED MICROORGANISM FOR THE PRODUCTION OF 1,2-PROPANEDIOL |
| EP2194139A1 (en) | 2004-01-19 | 2010-06-09 | DSM IP Assets B.V. | Biochemical synthesis of 6 amino caproic acid |
| US7608700B2 (en) | 2004-03-08 | 2009-10-27 | North Carolina State University | Lactobacillus acidophilus nucleic acid sequences encoding stress-related proteins and uses therefor |
| DE102004031177A1 (en) | 2004-06-29 | 2006-01-19 | Henkel Kgaa | New odoriferous gene products from Bacillus licheniformis and improved biotechnological production processes based on them |
| US7262046B2 (en) | 2004-08-09 | 2007-08-28 | Rice University | Aerobic succinate production in bacteria |
| WO2006031424A2 (en) | 2004-08-27 | 2006-03-23 | Rice University | Mutant e. coli strain with increased succinic acid production |
| ES2444785T3 (en) | 2004-09-09 | 2014-02-26 | Research Institute Of Innovative Technology For The Earth | DNA fragment that has a promoter function |
| WO2006034156A2 (en) | 2004-09-17 | 2006-03-30 | Rice University | High succinate producing bacteria |
| US7569380B2 (en) | 2004-12-22 | 2009-08-04 | Rice University | Simultaneous anaerobic production of isoamyl acetate and succinic acid |
| JP2006204255A (en) | 2005-01-31 | 2006-08-10 | Canon Inc | ACETYL-CoA ACYLTRANSFERASE GENE-BROKEN POLYHYDROXYALKANOATE-PRODUCING MICROORGANISM, AND METHOD FOR PRODUCING POLYHYDROXYALKANOATE THEREWITH |
| US20090068207A1 (en) | 2005-04-15 | 2009-03-12 | Vascular Biogenics Ltd. | Compositions Containing Beta 2-Glycoprotein I-Derived Peptides for the Prevention and/or Treatment of Vascular Disease |
| US8187842B2 (en) | 2005-06-20 | 2012-05-29 | Archer Daniels Midland Company | Altered glyoxylate shunt for improved production of aspartate-derived amino acids and chemicals |
| KR100679638B1 (en) | 2005-08-19 | 2007-02-06 | 한국과학기술원 | Microorganisms transformed with gene encoding formate ddehydrogenase d or e and method for preparing succinic acid using the same |
| KR100676160B1 (en) | 2005-08-19 | 2007-02-01 | 한국과학기술원 | Microorganisms transformed with gene encoding malic enzyme and method for preparing succinic acid using the same |
| JP2009507493A (en) | 2005-09-09 | 2009-02-26 | ジェノマティカ・インコーポレイテッド | Methods and organisms for growth-linked succinate production |
| US9297028B2 (en) | 2005-09-29 | 2016-03-29 | Butamax Advanced Biofuels Llc | Fermentive production of four carbon alcohols |
| UA96928C2 (en) | 2005-10-26 | 2011-12-26 | Э.И. Дю Пон Де Немур Энд Компани | Fermentive production of four carbon alcohols |
| JP2010524428A (en) | 2006-05-01 | 2010-07-22 | ユニバーシティー オブ フロリダ リサーチ ファンデーション, インク. | Ethanol production in non-recombinant hosts |
| US8962298B2 (en) | 2006-05-02 | 2015-02-24 | Butamax Advanced Biofuels Llc | Recombinant host cell comprising a diol dehydratase |
| DE102006025821A1 (en) | 2006-06-02 | 2007-12-06 | Degussa Gmbh | An enzyme for the production of Mehylmalonatsemialdehyd or Malonatsemialdehyd |
| RU2311231C1 (en) | 2006-08-15 | 2007-11-27 | ООО "Объединенный центр исследований и разработок" | Catalyst for production of acrylic acid esters according to metathesis reaction of dialkyl malates (variants) and a catalytic composition based thereof |
| BRPI0716212A2 (en) | 2006-08-30 | 2013-10-15 | Cargill Inc | BETA-ALANINE / ALPHA KETOGLUTARATE AMINOTRANSFERASE FOR PRODUCTION OF 3-HYDROXIPROPIONIC ACID |
| US8017364B2 (en) | 2006-12-12 | 2011-09-13 | Butamax(Tm) Advanced Biofuels Llc | Solvent tolerant microorganisms |
| US20100062505A1 (en) | 2006-12-21 | 2010-03-11 | Gevo, Inc. | Butanol production by metabolically engineered yeast |
| US20080274522A1 (en) | 2007-05-02 | 2008-11-06 | Bramucci Michael G | Method for the production of 2-butanone |
| US8426174B2 (en) | 2007-05-02 | 2013-04-23 | Butamax(Tm) Advanced Biofuels Llc | Method for the production of 2-butanol |
| BRPI0812515A2 (en) | 2007-06-15 | 2014-12-30 | Evonik Degussa Gmbh | MICRO-ORGANISM WITH UNREGULATED VITAMIN B12 SYSTEM |
| EP2017344A1 (en) | 2007-07-20 | 2009-01-21 | Nederlandse Organisatie voor toegepast- natuurwetenschappelijk onderzoek TNO | Production of itaconic acid |
| US7947483B2 (en) | 2007-08-10 | 2011-05-24 | Genomatica, Inc. | Methods and organisms for the growth-coupled production of 1,4-butanediol |
| EP2182051A4 (en) * | 2007-08-29 | 2010-12-29 | Res Inst Innovative Tech Earth | Transformants capable of producing isopropanol |
| KR101042242B1 (en) | 2007-09-07 | 2011-06-17 | 한국과학기술원 | Mutants having a producing ability of 1,4-butanediol and method for preparing 1,4-butanediol using the same |
| CN101903530A (en) * | 2007-10-12 | 2010-12-01 | 加利福尼亚大学董事会 | Microorganism engineered to produce isopropanol |
| WO2009094485A1 (en) | 2008-01-22 | 2009-07-30 | Genomatica, Inc. | Methods and organisms for utilizing synthesis gas or other gaseous carbon sources and methanol |
| WO2009103026A1 (en) | 2008-02-15 | 2009-08-20 | Gevo, Inc. | Engineered microorganisms for producing isopropanol |
| TWI787575B (en) | 2008-03-11 | 2022-12-21 | 美商吉諾瑪蒂卡股份有限公司 | PREPARATION OF 6-AMINOCAPROIC ACID FROM α-KETOPIMELIC ACID |
| MY177321A (en) | 2008-03-11 | 2020-09-11 | Genomatica Inc | Adipate (ester or thioester) synthesis |
| CN103555643B (en) | 2008-03-27 | 2016-08-10 | 基因组股份公司 | For producing the microorganism of adipic acid and other compounds |
| KR20110021797A (en) | 2008-04-25 | 2011-03-04 | 자이단호우진 치큐칸쿄 산교기쥬츠 켄큐키코 | Genetically modified coryneform bacteria capable of producing isopropanol |
| US8129154B2 (en) | 2008-06-17 | 2012-03-06 | Genomatica, Inc. | Microorganisms and methods for the biosynthesis of fumarate, malate, and acrylate |
| US8357826B2 (en) | 2008-10-16 | 2013-01-22 | Karl Kharas | Methods and apparatus for synthesis of alcohols from syngas |
-
2010
- 2010-04-30 BR BRPI1013505A patent/BRPI1013505A2/en not_active IP Right Cessation
- 2010-04-30 EP EP10770455A patent/EP2425006A4/en not_active Withdrawn
- 2010-04-30 JP JP2012508799A patent/JP2012525156A/en active Pending
- 2010-04-30 WO PCT/US2010/033271 patent/WO2010127303A1/en active Application Filing
- 2010-04-30 KR KR1020117028541A patent/KR20120068763A/en not_active Application Discontinuation
- 2010-04-30 AU AU2010242849A patent/AU2010242849A1/en not_active Abandoned
- 2010-04-30 US US12/772,094 patent/US8993285B2/en not_active Expired - Fee Related
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090075351A1 (en) * | 2007-03-16 | 2009-03-19 | Burk Mark J | Compositions and methods for the biosynthesis of 1,4-butanediol and its precursors |
| US20080293125A1 (en) * | 2007-04-18 | 2008-11-27 | Gevo, Inc. | Engineered microorganisms for producing isopropanol |
| US20090047718A1 (en) * | 2007-05-17 | 2009-02-19 | Blaschek Hans P | Methods and compositions for producing solvents |
Non-Patent Citations (5)
| Title |
|---|
| GERHARDT ET AL.: "Fermentation of 4-aminobutyrate by Clostridium aminobutyricum : cloning of two genes involved in the formation and dehydration of 4-hydroxybutyryl-CoA.", ARCH MICROBIOL, vol. 174, 2000, pages 189 - 199, XP008109904 * |
| MATTA ET AL.: "Interactions of the Antizyme AtoC with Regulatory Elements of the Escherichia coli atoDAEB Operon.", JOURNAL OF BACTERIOLOGY, vol. 189, no. 17, September 2007 (2007-09-01), pages 6324 - 6332, XP008129549 * |
| See also references of EP2425006A4 * |
| STEEN ET AL.: "Metabolic engineering of Saccharomyces cerevisiae for the production of n-butanol.", MICROBIAL CELL FACTORIES, vol. 7, no. 36, 2008, pages 6, XP021049875 * |
| ZOU ET AL.: "Metabolic Engineering for Microbial Production and Applications of Copolyesters Consisting of 3-Hydroxybutyrate and Medium-Chain-Length 3-Hydroxyalkanoates.", MACROMOL. BIOSCI., vol. 7, 2007, pages 174 - 182, XP008155010 * |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2373781A4 (en) * | 2008-12-16 | 2012-10-10 | Genomatica Inc | Microorganisms and methods for conversion of syngas and other carbon sources to useful products |
| EP2373781A1 (en) * | 2008-12-16 | 2011-10-12 | Genomatica, Inc. | Microorganisms and methods for conversion of syngas and other carbon sources to useful products |
| WO2011057871A3 (en) * | 2009-11-11 | 2012-01-26 | Evonik Röhm Gmbh | Use of a protein homologous to a meab protein for increasing the enzymatic activity of a 3-hydroxycarboxylic acid-coa mutase |
| US8980594B2 (en) | 2009-11-11 | 2015-03-17 | Evonik Roehm Gmbh | Use of a protein homologous to a MeaB protein for increasing the enzymatic activity of a 3-hydroxycarboxylic acid-CoA mutase |
| US8728804B2 (en) | 2010-10-29 | 2014-05-20 | Novozymes A/S | Polypeptides having succinyl-CoA: acetoacetate transferase activity and polynucleotides encoding same |
| WO2014152434A2 (en) | 2013-03-15 | 2014-09-25 | Genomatica, Inc. | Microorganisms and methods for producing butadiene and related compounds by formate assimilation |
| US10808262B2 (en) | 2013-12-03 | 2020-10-20 | Genomatica, Inc. | Microorganisms and methods for improving product yields on methanol using acetyl-CoA synthesis |
| WO2015084633A1 (en) | 2013-12-03 | 2015-06-11 | Genomatica, Inc. | Microorganisms and methods for improving product yields on methanol using acetyl-coa synthesis |
| EP4296364A2 (en) | 2013-12-03 | 2023-12-27 | Genomatica, Inc. | Microorganisms and methods for improving product yields on methanol using acetyl-coa synthesis |
| EP3967747A1 (en) | 2013-12-03 | 2022-03-16 | Genomatica, Inc. | Microorganisms and methods for improving product yields on methanol using acetyl-coa synthesis |
| EP3744830A1 (en) | 2013-12-27 | 2020-12-02 | Genomatica, Inc. | Methods and organisms with increased carbon flux efficiencies |
| US10597684B2 (en) | 2013-12-27 | 2020-03-24 | Genomatica, Inc. | Methods and organisms with increased carbon flux efficiencies |
| EP4407037A2 (en) | 2013-12-27 | 2024-07-31 | Genomatica, Inc. | Methods and organisms with increased carbon flux efficiencies |
| EP3741865A1 (en) | 2014-09-18 | 2020-11-25 | Genomatica, Inc. | Non-natural microbial organisms with improved energetic efficiency |
| WO2016044713A1 (en) | 2014-09-18 | 2016-03-24 | Genomatica, Inc. | Non-natural microbial organisms with improved energetic efficiency |
| EP4421181A2 (en) | 2014-09-18 | 2024-08-28 | Genomatica, Inc. | Non-natural microbial organisms with improved energetic efficiency |
| WO2019152375A1 (en) | 2018-01-30 | 2019-08-08 | Genomatica, Inc. | Fermentation systems and methods with substantially uniform volumetric uptake rate of a reactive gaseous component |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2425006A4 (en) | 2013-01-23 |
| KR20120068763A (en) | 2012-06-27 |
| US20100323418A1 (en) | 2010-12-23 |
| AU2010242849A1 (en) | 2011-11-24 |
| EP2425006A1 (en) | 2012-03-07 |
| US8993285B2 (en) | 2015-03-31 |
| BRPI1013505A2 (en) | 2018-02-14 |
| JP2012525156A (en) | 2012-10-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20220290192A1 (en) | Organisms for the production of 1,3-butanediol | |
| US8993285B2 (en) | Organisms for the production of isopropanol, n-butanol, and isobutanol | |
| US8715971B2 (en) | Microorganisms and methods for the co-production of isopropanol and 1,4-butanediol | |
| WO2012177619A2 (en) | Microorganisms for producing 1,3-butanediol and methods related thereto | |
| WO2012177601A2 (en) | Microorganisms for producing isobutanol and methods related thereto | |
| AU2013202629B2 (en) | Organisms for the production of 1,3-butanediol | |
| AU2013202621A1 (en) | Organisms for the production of isopropanol, n-butanol, and isobutanol | |
| AU2013203166A1 (en) | Microorganisms for producing 1,3-butanediol and methods related thereto |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10770455 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2012508799 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2010242849 Country of ref document: AU Date of ref document: 20100430 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20117028541 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010770455 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: PI1013505 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: PI1013505 Country of ref document: BR Kind code of ref document: A2 Effective date: 20111031 |