WO2010056066A1 - Novel organometallic compounds for electroluminescence and organic electroluminescent device using the same - Google Patents
Novel organometallic compounds for electroluminescence and organic electroluminescent device using the same Download PDFInfo
- Publication number
- WO2010056066A1 WO2010056066A1 PCT/KR2009/006693 KR2009006693W WO2010056066A1 WO 2010056066 A1 WO2010056066 A1 WO 2010056066A1 KR 2009006693 W KR2009006693 W KR 2009006693W WO 2010056066 A1 WO2010056066 A1 WO 2010056066A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- halogen
- aryl
- tri
- group
- Prior art date
Links
- 238000005401 electroluminescence Methods 0.000 title description 10
- 150000002902 organometallic compounds Chemical class 0.000 title description 5
- 150000001875 compounds Chemical class 0.000 claims abstract description 66
- 150000002367 halogens Chemical class 0.000 claims description 56
- 125000006743 (C1-C60) alkyl group Chemical group 0.000 claims description 54
- 125000005104 aryl silyl group Chemical group 0.000 claims description 47
- 229910052736 halogen Inorganic materials 0.000 claims description 45
- -1 spirobifluorenyl Chemical group 0.000 claims description 44
- 125000006749 (C6-C60) aryl group Chemical group 0.000 claims description 42
- 125000003118 aryl group Chemical group 0.000 claims description 42
- 125000001072 heteroaryl group Chemical group 0.000 claims description 40
- 125000001424 substituent group Chemical group 0.000 claims description 37
- 239000010410 layer Substances 0.000 claims description 32
- 229910052739 hydrogen Inorganic materials 0.000 claims description 28
- 239000001257 hydrogen Substances 0.000 claims description 28
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 24
- 125000003545 alkoxy group Chemical group 0.000 claims description 23
- 125000003282 alkyl amino group Chemical group 0.000 claims description 23
- 125000001769 aryl amino group Chemical group 0.000 claims description 23
- 125000000217 alkyl group Chemical group 0.000 claims description 21
- 125000005103 alkyl silyl group Chemical group 0.000 claims description 19
- 125000005842 heteroatom Chemical group 0.000 claims description 19
- 125000006818 (C3-C60) cycloalkyl group Chemical group 0.000 claims description 18
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 18
- 125000004450 alkenylene group Chemical group 0.000 claims description 18
- 125000002947 alkylene group Chemical group 0.000 claims description 18
- 239000000126 substance Substances 0.000 claims description 18
- 125000004448 alkyl carbonyl group Chemical group 0.000 claims description 16
- 125000005129 aryl carbonyl group Chemical group 0.000 claims description 16
- 229910052751 metal Inorganic materials 0.000 claims description 16
- 239000002184 metal Substances 0.000 claims description 16
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 claims description 14
- 125000002723 alicyclic group Chemical group 0.000 claims description 14
- 125000002950 monocyclic group Chemical group 0.000 claims description 14
- 125000003367 polycyclic group Chemical group 0.000 claims description 14
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 13
- 125000006746 (C1-C60) alkoxy group Chemical group 0.000 claims description 12
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 claims description 12
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 11
- 125000006582 (C5-C6) heterocycloalkyl group Chemical group 0.000 claims description 10
- 125000004453 alkoxycarbonyl group Chemical group 0.000 claims description 10
- 150000001602 bicycloalkyls Chemical group 0.000 claims description 10
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 10
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 10
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 10
- 239000012044 organic layer Substances 0.000 claims description 10
- 125000006744 (C2-C60) alkenyl group Chemical group 0.000 claims description 9
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 claims description 9
- 125000001624 naphthyl group Chemical group 0.000 claims description 9
- 125000006820 (C1-C60) alkylthio group Chemical group 0.000 claims description 8
- 125000006745 (C2-C60) alkynyl group Chemical group 0.000 claims description 8
- 125000006752 (C6-C60) arylthio group Chemical group 0.000 claims description 8
- 239000002019 doping agent Substances 0.000 claims description 8
- 125000006751 (C6-C60) aryloxy group Chemical group 0.000 claims description 7
- 229910052717 sulfur Inorganic materials 0.000 claims description 7
- 125000000923 (C1-C30) alkyl group Chemical group 0.000 claims description 6
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 claims description 6
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 6
- 229910052760 oxygen Inorganic materials 0.000 claims description 6
- 125000006822 tri(C1-C30) alkylsilyl group Chemical group 0.000 claims description 6
- 125000003342 alkenyl group Chemical group 0.000 claims description 5
- 125000006736 (C6-C20) aryl group Chemical group 0.000 claims description 4
- 125000000304 alkynyl group Chemical group 0.000 claims description 4
- 229910052738 indium Inorganic materials 0.000 claims description 4
- 150000002739 metals Chemical class 0.000 claims description 4
- 229910052782 aluminium Inorganic materials 0.000 claims description 3
- 125000004104 aryloxy group Chemical group 0.000 claims description 3
- 239000003446 ligand Substances 0.000 claims description 3
- 229910052759 nickel Inorganic materials 0.000 claims description 3
- HCMJWOGOISXSDL-UHFFFAOYSA-N (2-isothiocyanato-1-phenylethyl)benzene Chemical compound C=1C=CC=CC=1C(CN=C=S)C1=CC=CC=C1 HCMJWOGOISXSDL-UHFFFAOYSA-N 0.000 claims description 2
- 125000006652 (C3-C12) cycloalkyl group Chemical group 0.000 claims description 2
- 125000000739 C2-C30 alkenyl group Chemical group 0.000 claims description 2
- 125000004414 alkyl thio group Chemical group 0.000 claims description 2
- 125000005110 aryl thio group Chemical group 0.000 claims description 2
- 229910052790 beryllium Inorganic materials 0.000 claims description 2
- 229910052791 calcium Inorganic materials 0.000 claims description 2
- 125000002091 cationic group Chemical group 0.000 claims description 2
- 229910052802 copper Inorganic materials 0.000 claims description 2
- 125000005105 dialkylarylsilyl group Chemical group 0.000 claims description 2
- 229910052733 gallium Inorganic materials 0.000 claims description 2
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 2
- 229910052747 lanthanoid Inorganic materials 0.000 claims description 2
- 229910052749 magnesium Inorganic materials 0.000 claims description 2
- 125000003003 spiro group Chemical group 0.000 claims description 2
- 229910052723 transition metal Inorganic materials 0.000 claims description 2
- 150000003624 transition metals Chemical class 0.000 claims description 2
- 125000004665 trialkylsilyl group Chemical group 0.000 claims description 2
- 125000005106 triarylsilyl group Chemical group 0.000 claims description 2
- 229910052725 zinc Inorganic materials 0.000 claims description 2
- 125000005843 halogen group Chemical group 0.000 claims 8
- 239000000463 material Substances 0.000 abstract description 44
- 238000004519 manufacturing process Methods 0.000 abstract description 4
- 150000002894 organic compounds Chemical class 0.000 description 44
- 0 COC(c1*(C[C@@](*)(C2(*)*)*3ccccc3-c(c(N)c3)c2cc3N)cccc1)O Chemical compound COC(c1*(C[C@@](*)(C2(*)*)*3ccccc3-c(c(N)c3)c2cc3N)cccc1)O 0.000 description 23
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 description 16
- 229910052805 deuterium Inorganic materials 0.000 description 16
- 125000004432 carbon atom Chemical group C* 0.000 description 15
- 125000005549 heteroarylene group Chemical group 0.000 description 11
- 238000002360 preparation method Methods 0.000 description 11
- 150000002431 hydrogen Chemical class 0.000 description 10
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 9
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- 125000006761 (C6-C60) arylene group Chemical group 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 230000005525 hole transport Effects 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 125000000732 arylene group Chemical group 0.000 description 5
- 238000001771 vacuum deposition Methods 0.000 description 5
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 4
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 4
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 4
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 4
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 4
- 239000004305 biphenyl Substances 0.000 description 4
- 235000010290 biphenyl Nutrition 0.000 description 4
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 125000002541 furyl group Chemical group 0.000 description 4
- 125000002883 imidazolyl group Chemical group 0.000 description 4
- 125000005956 isoquinolyl group Chemical group 0.000 description 4
- 125000001786 isothiazolyl group Chemical group 0.000 description 4
- 125000000842 isoxazolyl group Chemical group 0.000 description 4
- 125000001715 oxadiazolyl group Chemical group 0.000 description 4
- 125000002971 oxazolyl group Chemical group 0.000 description 4
- 125000004934 phenanthridinyl group Chemical group C1(=CC=CC2=NC=C3C=CC=CC3=C12)* 0.000 description 4
- 125000003373 pyrazinyl group Chemical group 0.000 description 4
- 125000003226 pyrazolyl group Chemical group 0.000 description 4
- 125000002098 pyridazinyl group Chemical group 0.000 description 4
- 125000004076 pyridyl group Chemical group 0.000 description 4
- 125000000714 pyrimidinyl group Chemical group 0.000 description 4
- 125000000168 pyrrolyl group Chemical group 0.000 description 4
- 125000005493 quinolyl group Chemical group 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- 125000001113 thiadiazolyl group Chemical group 0.000 description 4
- 125000000335 thiazolyl group Chemical group 0.000 description 4
- 125000001544 thienyl group Chemical group 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- CUJRVFIICFDLGR-UHFFFAOYSA-N acetylacetonate Chemical compound CC(=O)[CH-]C(C)=O CUJRVFIICFDLGR-UHFFFAOYSA-N 0.000 description 3
- 150000004770 chalcogenides Chemical class 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 238000000605 extraction Methods 0.000 description 3
- 125000002632 imidazolidinyl group Chemical group 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- IMKMFBIYHXBKRX-UHFFFAOYSA-M lithium;quinoline-2-carboxylate Chemical compound [Li+].C1=CC=CC2=NC(C(=O)[O-])=CC=C21 IMKMFBIYHXBKRX-UHFFFAOYSA-M 0.000 description 3
- 229910001507 metal halide Inorganic materials 0.000 description 3
- 150000005309 metal halides Chemical class 0.000 description 3
- 229910044991 metal oxide Inorganic materials 0.000 description 3
- 150000004706 metal oxides Chemical class 0.000 description 3
- 125000002757 morpholinyl group Chemical group 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N nickel Substances [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 3
- 229910052757 nitrogen Inorganic materials 0.000 description 3
- 125000004193 piperazinyl group Chemical group 0.000 description 3
- 125000003386 piperidinyl group Chemical group 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 3
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 2
- 150000001204 N-oxides Chemical class 0.000 description 2
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 2
- 229910052787 antimony Inorganic materials 0.000 description 2
- 229910052786 argon Inorganic materials 0.000 description 2
- XRWSZZJLZRKHHD-WVWIJVSJSA-N asunaprevir Chemical compound O=C([C@@H]1C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C)C(C)(C)C)OC1=NC=C(C2=CC=C(Cl)C=C21)OC)N[C@]1(C(=O)NS(=O)(=O)C2CC2)C[C@H]1C=C XRWSZZJLZRKHHD-WVWIJVSJSA-N 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 2
- 229910052797 bismuth Inorganic materials 0.000 description 2
- 125000006251 butylcarbonyl group Chemical group 0.000 description 2
- 125000004744 butyloxycarbonyl group Chemical group 0.000 description 2
- 229940125904 compound 1 Drugs 0.000 description 2
- 229940125961 compound 24 Drugs 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 150000004696 coordination complex Chemical class 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- ZTJBELXDHFJJEU-UHFFFAOYSA-N dimethylboron Chemical group C[B]C ZTJBELXDHFJJEU-UHFFFAOYSA-N 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 125000004672 ethylcarbonyl group Chemical group [H]C([H])([H])C([H])([H])C(*)=O 0.000 description 2
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 2
- 238000011049 filling Methods 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 229910052741 iridium Inorganic materials 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 229910052745 lead Inorganic materials 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 2
- 125000004674 methylcarbonyl group Chemical group CC(=O)* 0.000 description 2
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000005185 naphthylcarbonyl group Chemical group C1(=CC=CC2=CC=CC=C12)C(=O)* 0.000 description 2
- 230000001590 oxidative effect Effects 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 2
- 125000005003 perfluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 2
- 125000005004 perfluoroethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 2
- 229910052697 platinum Inorganic materials 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- LVTJOONKWUXEFR-FZRMHRINSA-N protoneodioscin Natural products O(C[C@@H](CC[C@]1(O)[C@H](C)[C@@H]2[C@]3(C)[C@H]([C@H]4[C@@H]([C@]5(C)C(=CC4)C[C@@H](O[C@@H]4[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@@H](O)[C@H](O[C@H]6[C@@H](O)[C@@H](O)[C@@H](O)[C@H](C)O6)[C@H](CO)O4)CC5)CC3)C[C@@H]2O1)C)[C@H]1[C@H](O)[C@H](O)[C@H](O)[C@@H](CO)O1 LVTJOONKWUXEFR-FZRMHRINSA-N 0.000 description 2
- 229910052761 rare earth metal Inorganic materials 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 229910052702 rhenium Inorganic materials 0.000 description 2
- 229910052703 rhodium Inorganic materials 0.000 description 2
- 150000003839 salts Chemical group 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 229910052814 silicon oxide Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- ILMRJRBKQSSXGY-UHFFFAOYSA-N tert-butyl(dimethyl)silicon Chemical group C[Si](C)C(C)(C)C ILMRJRBKQSSXGY-UHFFFAOYSA-N 0.000 description 2
- 229910052718 tin Inorganic materials 0.000 description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 2
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 2
- DXBHBZVCASKNBY-UHFFFAOYSA-N 1,2-Benz(a)anthracene Chemical class C1=CC=C2C3=CC4=CC=CC=C4C=C3C=CC2=C1 DXBHBZVCASKNBY-UHFFFAOYSA-N 0.000 description 1
- MNCMBBIFTVWHIP-UHFFFAOYSA-N 1-anthracen-9-yl-2,2,2-trifluoroethanone Chemical group C1=CC=C2C(C(=O)C(F)(F)F)=C(C=CC=C3)C3=CC2=C1 MNCMBBIFTVWHIP-UHFFFAOYSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 1
- VQGHOUODWALEFC-UHFFFAOYSA-N 2-phenylpyridine Chemical compound C1=CC=CC=C1C1=CC=CC=N1 VQGHOUODWALEFC-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 229910017107 AlOx Inorganic materials 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- PENCMTNCVDKNRG-GEFWVMAASA-N C/C=C\C=C(\C=Cc(c1c(c(-c2c(cccc3)c3ccc2)c2)O3)c2-c2cccc4c2cccc4)/C1=CN3s(cccc1C=CCc2c(cc3-c4c(cccc5)c5ccc4)-c4cccc5c4cccc5)c1-c2c3OC Chemical compound C/C=C\C=C(\C=Cc(c1c(c(-c2c(cccc3)c3ccc2)c2)O3)c2-c2cccc4c2cccc4)/C1=CN3s(cccc1C=CCc2c(cc3-c4c(cccc5)c5ccc4)-c4cccc5c4cccc5)c1-c2c3OC PENCMTNCVDKNRG-GEFWVMAASA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 229940126062 Compound A Drugs 0.000 description 1
- KOPBYBDAPCDYFK-UHFFFAOYSA-N Cs2O Inorganic materials [O-2].[Cs+].[Cs+] KOPBYBDAPCDYFK-UHFFFAOYSA-N 0.000 description 1
- NLDMNSXOCDLTTB-UHFFFAOYSA-N Heterophylliin A Natural products O1C2COC(=O)C3=CC(O)=C(O)C(O)=C3C3=C(O)C(O)=C(O)C=C3C(=O)OC2C(OC(=O)C=2C=C(O)C(O)=C(O)C=2)C(O)C1OC(=O)C1=CC(O)=C(O)C(O)=C1 NLDMNSXOCDLTTB-UHFFFAOYSA-N 0.000 description 1
- 239000002841 Lewis acid Substances 0.000 description 1
- FUJCRWPEOMXPAD-UHFFFAOYSA-N Li2O Inorganic materials [Li+].[Li+].[O-2] FUJCRWPEOMXPAD-UHFFFAOYSA-N 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N Magnesium oxide Chemical compound [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229910003564 SiAlON Inorganic materials 0.000 description 1
- XSTXAVWGXDQKEL-UHFFFAOYSA-N Trichloroethylene Chemical group ClC=C(Cl)Cl XSTXAVWGXDQKEL-UHFFFAOYSA-N 0.000 description 1
- 239000007983 Tris buffer Substances 0.000 description 1
- ZOIORXHNWRGPMV-UHFFFAOYSA-N acetic acid;zinc Chemical compound [Zn].CC(O)=O.CC(O)=O ZOIORXHNWRGPMV-UHFFFAOYSA-N 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001339 alkali metal compounds Chemical class 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- REDXJYDRNCIFBQ-UHFFFAOYSA-N aluminium(3+) Chemical compound [Al+3] REDXJYDRNCIFBQ-UHFFFAOYSA-N 0.000 description 1
- TVIVIEFSHFOWTE-UHFFFAOYSA-N aluminum;quinolin-8-ol Chemical compound [Al+3].C1=CN=C2C(O)=CC=CC2=C1.C1=CN=C2C(O)=CC=CC2=C1.C1=CN=C2C(O)=CC=CC2=C1 TVIVIEFSHFOWTE-UHFFFAOYSA-N 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 125000002078 anthracen-1-yl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C([*])=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 125000000748 anthracen-2-yl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C([H])=C([*])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 1
- 150000001454 anthracenes Chemical class 0.000 description 1
- 125000004653 anthracenylene group Chemical group 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 125000002047 benzodioxolyl group Chemical group O1OC(C2=C1C=CC=C2)* 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000005874 benzothiadiazolyl group Chemical group 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- DIMYTQPLZWDZFE-UHFFFAOYSA-L beryllium sulfate tetrahydrate Chemical compound [Be+2].O.O.O.O.[O-]S([O-])(=O)=O DIMYTQPLZWDZFE-UHFFFAOYSA-L 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910001634 calcium fluoride Inorganic materials 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- JNGZXGGOCLZBFB-IVCQMTBJSA-N compound E Chemical compound N([C@@H](C)C(=O)N[C@@H]1C(N(C)C2=CC=CC=C2C(C=2C=CC=CC=2)=N1)=O)C(=O)CC1=CC(F)=CC(F)=C1 JNGZXGGOCLZBFB-IVCQMTBJSA-N 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 238000005137 deposition process Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- AKUNKIJLSDQFLS-UHFFFAOYSA-M dicesium;hydroxide Chemical compound [OH-].[Cs+].[Cs+] AKUNKIJLSDQFLS-UHFFFAOYSA-M 0.000 description 1
- XUCJHNOBJLKZNU-UHFFFAOYSA-M dilithium;hydroxide Chemical compound [Li+].[Li+].[OH-] XUCJHNOBJLKZNU-UHFFFAOYSA-M 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 125000003914 fluoranthenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC=C4C1=C23)* 0.000 description 1
- 125000003838 furazanyl group Chemical group 0.000 description 1
- 230000009477 glass transition Effects 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- MILUBEOXRNEUHS-UHFFFAOYSA-N iridium(3+) Chemical compound [Ir+3] MILUBEOXRNEUHS-UHFFFAOYSA-N 0.000 description 1
- 125000001977 isobenzofuranyl group Chemical group C=1(OC=C2C=CC=CC12)* 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 150000007517 lewis acids Chemical class 0.000 description 1
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Inorganic materials [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910001635 magnesium fluoride Inorganic materials 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 150000002736 metal compounds Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 150000002910 rare earth metals Chemical class 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 1
- 235000019345 sodium thiosulphate Nutrition 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000002344 surface layer Substances 0.000 description 1
- 125000001935 tetracenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC=CC=C4C=C3C=C12)* 0.000 description 1
- 125000005247 tetrazinyl group Chemical group N1=NN=NC(=C1)* 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- UBOXGVDOUJQMTN-UHFFFAOYSA-N trichloroethylene Natural products ClCC(Cl)Cl UBOXGVDOUJQMTN-UHFFFAOYSA-N 0.000 description 1
- ODHXBMXNKOYIBV-UHFFFAOYSA-N triphenylamine Chemical compound C1=CC=CC=C1N(C=1C=CC=CC=1)C1=CC=CC=C1 ODHXBMXNKOYIBV-UHFFFAOYSA-N 0.000 description 1
- 125000003960 triphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C3=CC=CC=C3C12)* 0.000 description 1
- 238000002061 vacuum sublimation Methods 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000004246 zinc acetate Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/16—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D215/20—Oxygen atoms
- C07D215/24—Oxygen atoms attached in position 8
- C07D215/26—Alcohols; Ethers thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D221/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00
- C07D221/02—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00 condensed with carbocyclic rings or ring systems
- C07D221/04—Ortho- or peri-condensed ring systems
- C07D221/06—Ring systems of three rings
- C07D221/08—Aza-anthracenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F5/00—Compounds containing elements of Groups 3 or 13 of the Periodic Table
- C07F5/02—Boron compounds
- C07F5/027—Organoboranes and organoborohydrides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0814—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring is substituted at a C ring atom by Si
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/381—Metal complexes comprising a group IIB metal element, e.g. comprising cadmium, mercury or zinc
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Definitions
- the present invention relates to an electroluminescent compound comprising a metal complex exhibiting excellent electrical conductivity and high luminous efficiency, and an electroluminescent device comprising the same as a host material.
- electroluminescent material The most important factor that determines luminous efficiency of an OLED is the electroluminescent material . Atpresent, fluorescent materials are widely used for the electroluminescent material. But, phosphorescent materials are better when considering the electroluminescence mechanism. Theoretically, phosphorescent materials can improve the luminous efficiency by 4-fold.
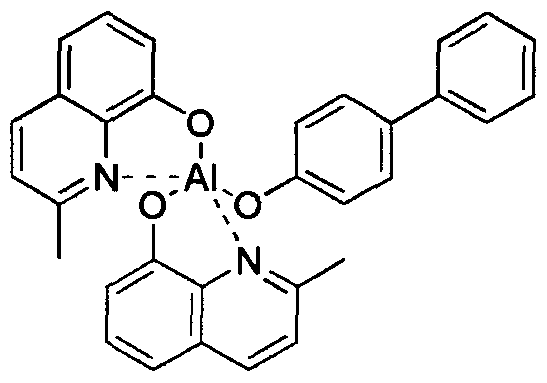
- iridium(III) complex-based phosphorescent materials are widely known. Such materials as (acac) Ir (btp) 2, Ir(ppy)3 and Firpic are known for red, green andblue colors, respectively. Recently, a lot of researches on phosphorescent materials are underway, especially in Japan, Europe and the US.
- CBP is the most widely known as a host material for a phosphorescent material.
- High-efficiency OLEDs using a hole blocking layer comprising BCP, BAIg, etc. are reported.
- High-performance OLEDs using BAIq derivatives as a host were reported by Pioneer (Japan) and others.
- the present invention provides an electroluminescent compound having a novel ligand metal complex with very superior electroluminescence characteristics and physical properties as compared to existing organic host materials as a backbone.
- the present invention provides an electroluminescent device comprising the electroluminescent compound as a host material.
- the present invention provides an organic solar cell comprising the electroluminescent compound.
- the present invention relates to an electroluminescent compound represented by Chemical Formula 1 and an electroluminescent device comprising the same as a host material:
- M represents a divalent or trivalent metal
- n represents the cationic valence of M
- Ri through R 8 independently represent hydrogen, (C1-C60) alkyl, halogen, cyano, (C3-C60) cycloalkyl, 5- or 6-memberedheterocycloalkyl containing one or more heteroatom (s) selected from N, O and S, (C7-C60)bicycloalkyl, adamantyl, (C2-C60) alkenyl, (C2-C60) alkynyl,
- aryl means an organic radical derived from an aromatic hydrocarbon by the removal of one hydrogen, and may include a 4- to 7-membered, particularly 5- or 6-membered, single ring or fused ring. Specific examples include phenyl, naphthyl, biphenyl, anthryl, indenyl, fluorenyl, phenanthryl, triphenylenyl, pyrenyl, perylenyl, chrysenyl, naphthacenyl, fluoranthenyl, etc., but not limited thereto.
- the naphthyl includes 1-naphthyl and 2-naphthyl
- the anthryl includes 1-anthryl, 2-anthryl and 9-anthryl
- the fluorenyl includes all of 1-fluorenyl, 2-fluorenyl, 3-fluorenyl, 4-fluorenyl and 9-fluorenyl.
- heteroaryl means an aryl group containing 1 to 4 heteroatom (s) selected from N, 0, S and Si as aromatic ring backbone atom(s), other remaining aromatic ring backbone atoms being carbon.
- the heteroaryl includes a divalent aryl group wherein the heteroatom (s) in the ring may be oxidized or quaternized to form, for example, N-oxide or quaternary salt .
- monocyclic heteroaryl such as furyl, thiophenyl, pyrrolyl, imidazolyl, pyrazolyl, thiazolyl, thiadiazolyl, isothiazolyl, isoxazolyl, oxazolyl, oxadiazolyl, triazinyl, tetrazinyl, triazolyl, tetrazolyl, furazanyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, etc., polycyclic heteroaryl such as benzofuranyl, benzothiophenyl, isobenzofuranyl, benzimidazolyl, benzothiazolyl, benzoisothiazolyl, benzoisoxazolyl, benzoxazolyl, isoindolyl, indolyl, indazolyl, benzothiadiazolyl, quinolyl, isoquinolyl, is
- the substituents including " (C1-C60) alkyl” moiety may have 1 to 60 carbon atoms, 1 to 20 carbon atoms, or 1 to 10 carbon atoms.
- the substituents including " (C6-C60) aryl” moiety may have 6 to 60 carbon atoms, 6 to 20 carbon atoms, or 6 to 12 carbon atoms.
- the substituents including " (C3-C60) heteroaryl” moiety may have 3 to 60 carbon atoms, 4 to 20 carbon atoms, or 4 to 12 carbon atoms.
- (C3-C60) cycloalkyl moiety may have 3 to 60 carbon atoms, 3 to 20 carbon atoms, or 3 to 7 carbon atoms.
- the substituents including " (C2-C60) alkenyl or alkynyl” moiety may have 2 to 60 carbon atoms, 2 to 20 carbon atoms, or 2 to 10 carbon atoms.
- Ri through R 8 may independently represent hydrogen, methyl, ethyl, n-propyl, i-propyl, n-butyl, i-butyl, t-butyl, trifluoromethyl, perfluoroethyl, perfluorobutyl, chloro, fluoro, cyano, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, pyrrolidinyl, imidazolidinyl, pyrazolidinyl, piperidinyl, piperazinyl, morpholinyl, thiomorpholinyl, pyridyl, furyl, thienyl, pyrrolyl, pyrazolyl, oxazolyl, isoxazolyl, thiazolyl, isothiazolyl, imidazolyl, oxadiazolyl,
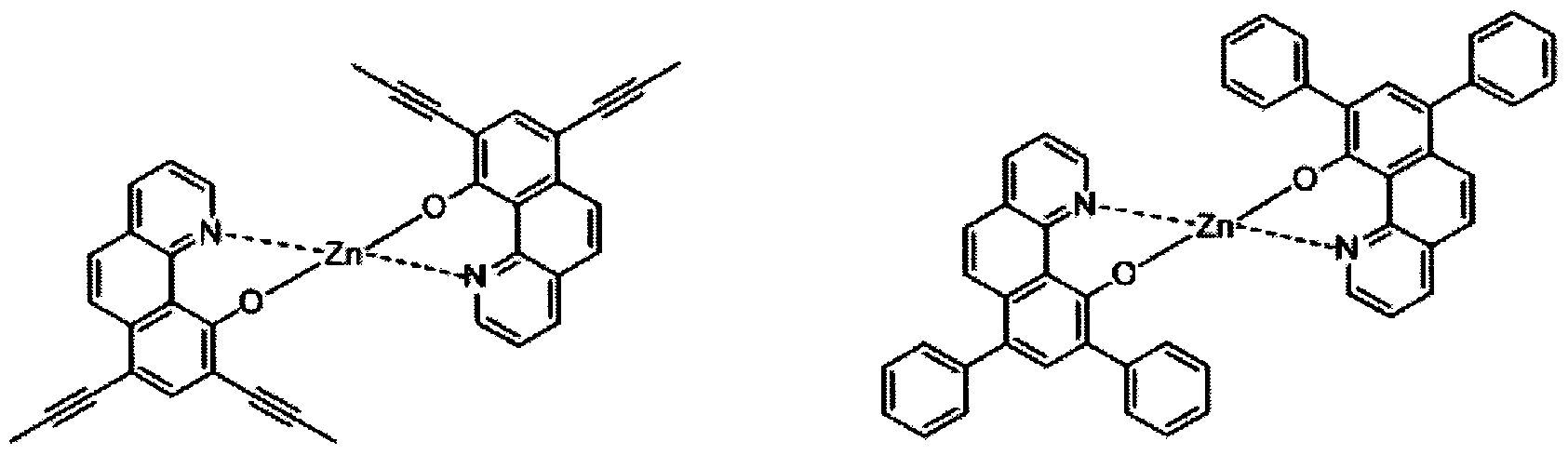
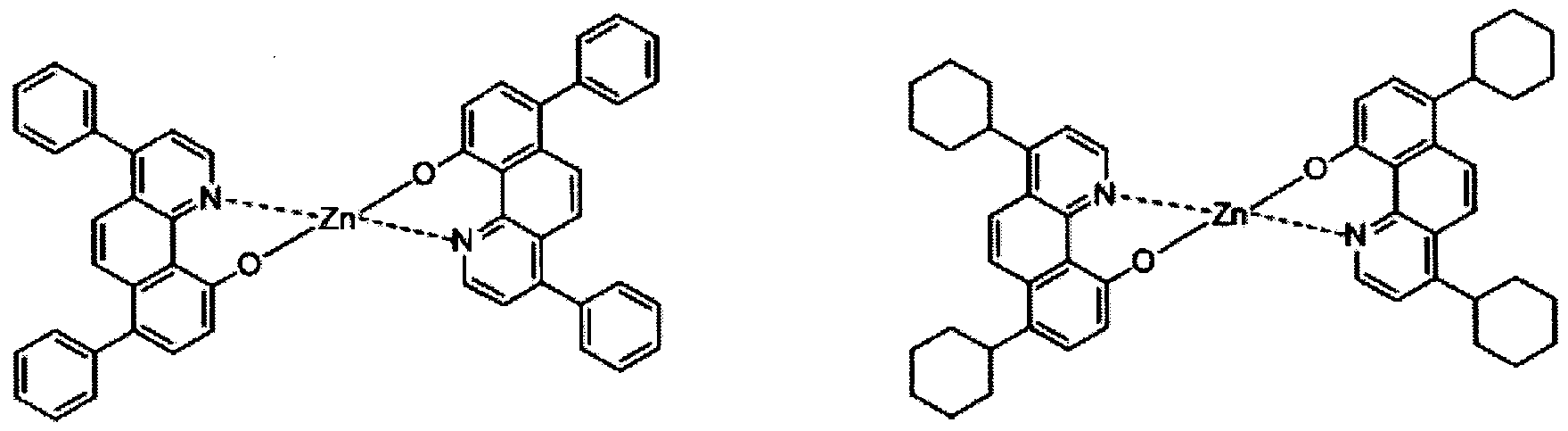
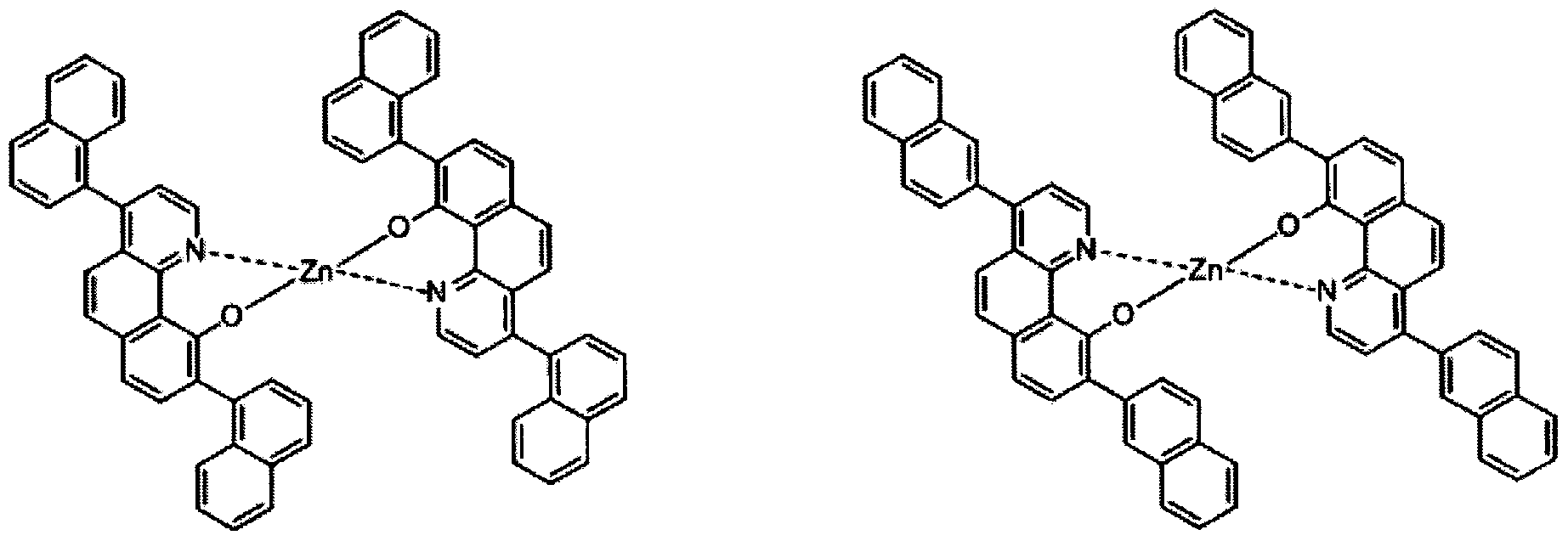
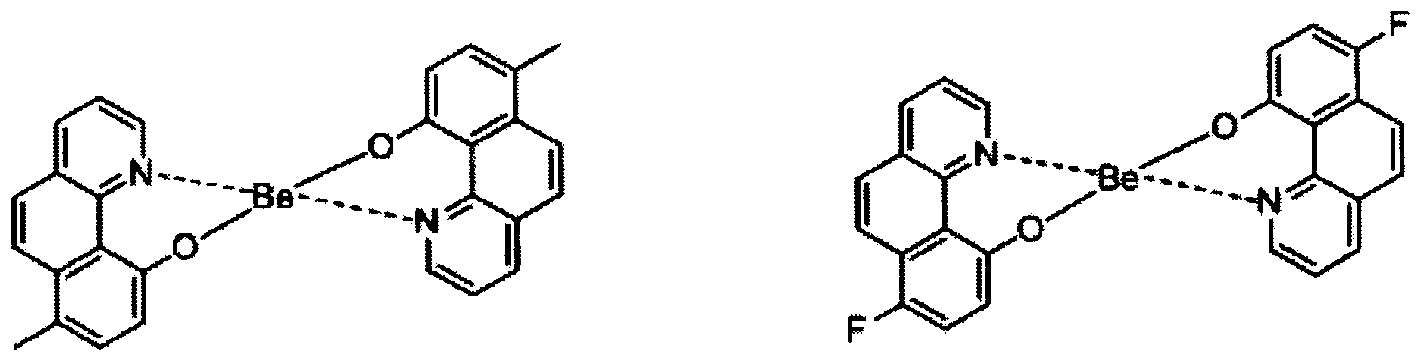
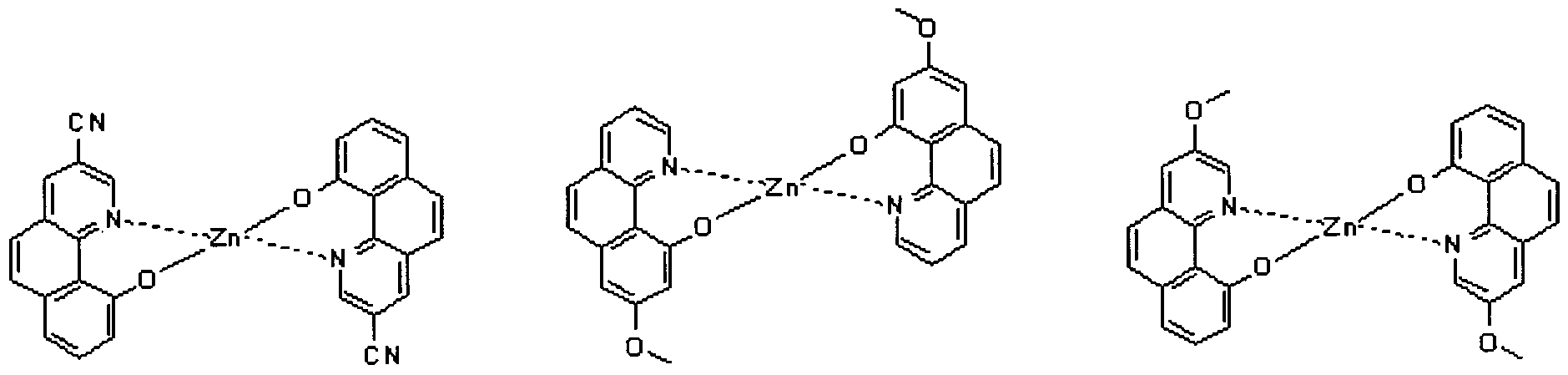
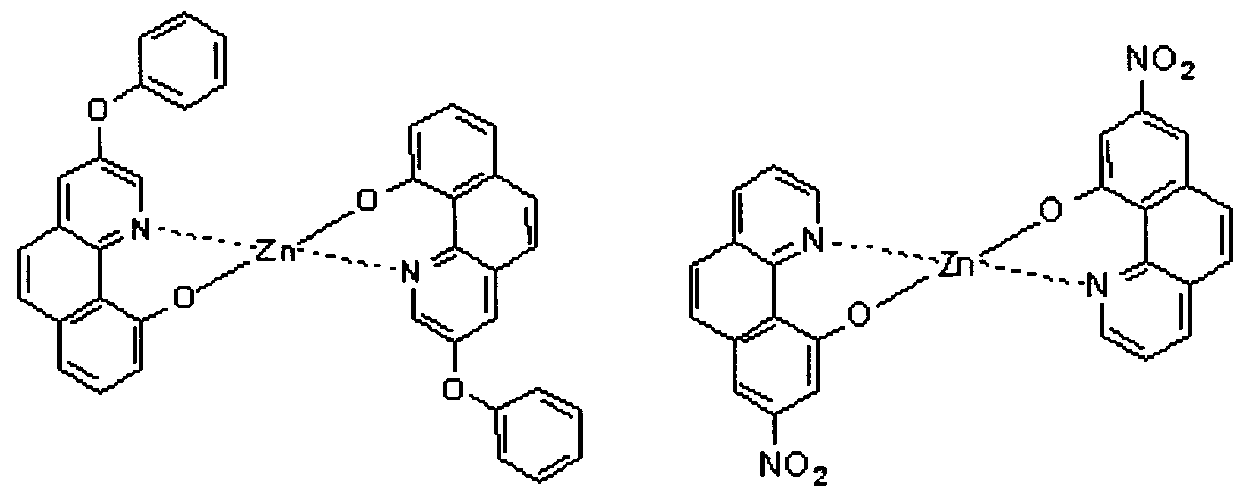
- electroluminescent compound represented by Chemical Formula 1 may be exemplified by following compounds, but not limited thereto:
- the present invention further provides an organic solar cell.
- the organic solar cell according to the present invention comprises one or more electroluminescent compound (s) represented by Chemical Formula 1.
- the present invention further provides an electroluminescent device.
- the electroluminescent device according to the present invention comprises a first electrode; a second electrode; and one or more organic layer (s) interposed between the first electrode and the second electrode, wherein the organic layer comprises one or more electroluminescent compound (s) represented by Chemical Formula 1.
- the organic layer further comprises an electroluminescent layer.
- the electroluminescent layer comprises one or more electroluminescent compound (s) represented by Chemical Formula 1, as an electroluminescent host, and one or more electroluminescent dopant (s) .
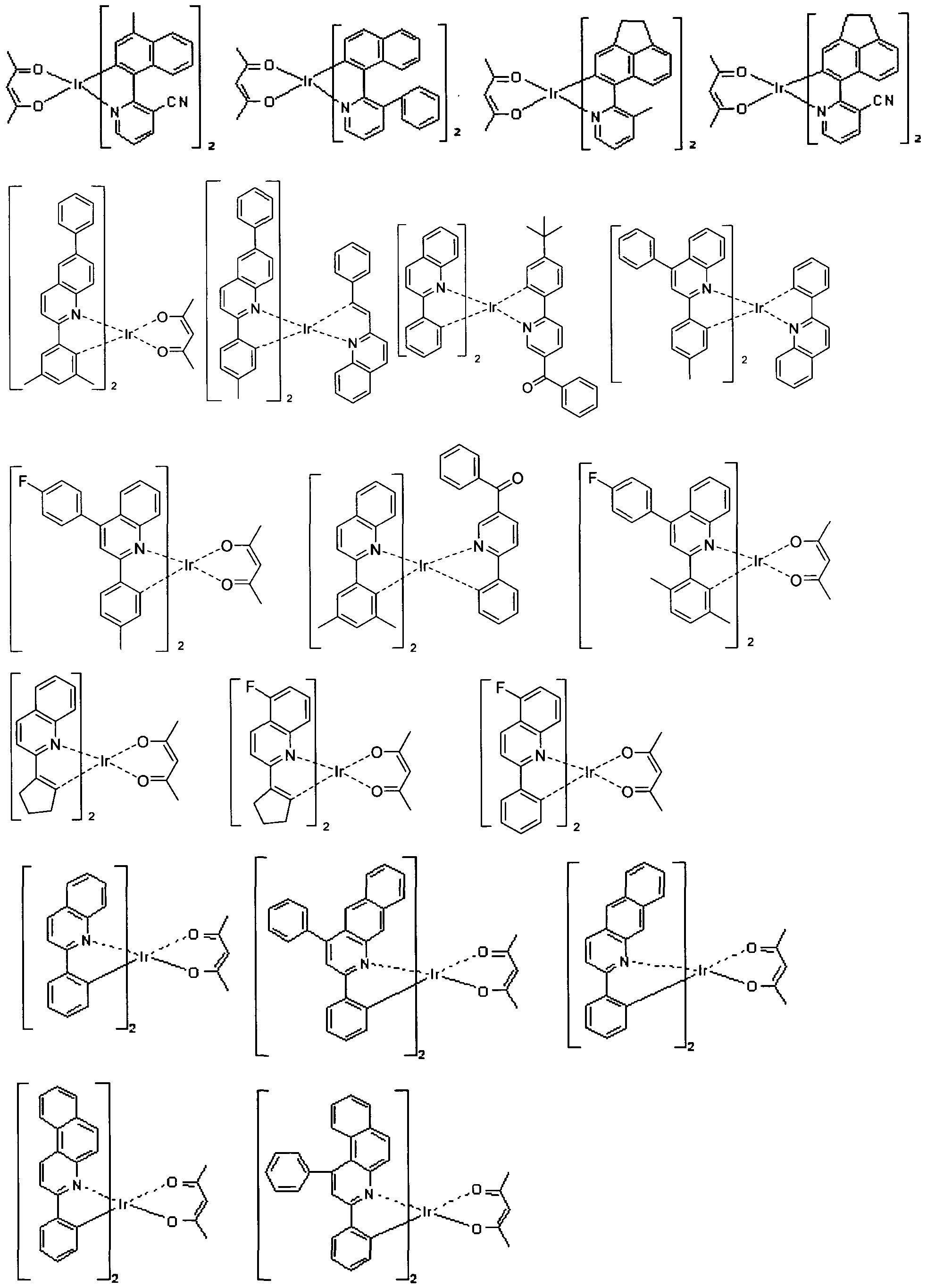
- the electroluminescent dopant used in the electroluminescent device according to the present invention is not particularly limited, but may be exemplified by the compounds represented by Chemical Formula 2:
- M 1 is a metal selected from a group consisting of Group 7, Group 8, Group 9, Group 10, Group 11, Group 13, Group 14, Group 15 and Group 16 metals;
- ligands L 101 , L 102 and L 103 are independently selected from the following structures:
- Rn through R 13 independently represent hydrogen, (C1-C60) alkyl with or without halogen substituent, (C6-C60)aryl with or without (C1-C60) alkyl substituent or halogen;
- Ri 4 through R 29 independently represent hydrogen, (C1-C60 ) alkyl, (Cl-C30)alkoxy, (C3-C60) cycloalkyl, (C2-C30) alkenyl, (C6-C60) aryl, mono- or di (C1-C30) alkylamino, mono- or di (C6-C30) arylamino, SF 5 , tri (C1-C30) alkylsilyl, di (C1-C30) alkyl (C6-C30) arylsilyl, tri (C6-C30) arylsilyl, cyano or halogen, and the alkyl, cycloalkyl, alkenyl or aryl of Ri 4 through R 29 may be further substituted by one or more substituent (s) selected from (C1-C60) alkyl, (C6-C60) aryl and halogen;
- R 30 through R 33 independently represent hydrogen, (C1-C60) alkyl with or without halogen substituent or (C6-C60)aryl with or without (C1-C60) alkyl substituent;
- R 34 and R 35 independently represent hydrogen, (C1-C60) alkyl, (C6-C60) aryl or halogen, OrR 34 and R 35 may be linked via (C3-C12) alkylene or (C3-C12) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring, and the alkyl or aryl of R 34 and R 35 , or the alicyclic ring or the monocyclic or polycyclic aromatic ring formed as they are linked via (C3-C12) alkylene or
- (C3-C12) alkenylene with or without a fused ring may be further substituted by one or more substituent (s) selected from (C1-C60) alkyl with or without halogen substituent, (C1-C30) alkoxy, halogen, tri (C1-C30) alkylsilyl, tri (C6-C30) arylsilyl and (C6-C60) aryl;
- R 36 represents (C1-C60) alkyl, (C6-C60) aryl, (C5-C60) heteroaryl containing one or more heteroatom (s) selected from N, 0 and S or halogen;
- R 37 through R 39 independently represent hydrogen, (C1-C60) alkyl,
- R 40 and R 41 independently represent hydrogen, (C1-C20) alkyl with or without halogen substituent, (C6-C20) aryl, halogen, cyano, tri (C1-C20) alkylsilyl, di (C1-C20) alkyl (C6-C20) arylsilyl, tri (C6-C20)arylsilyl, (C1-C20) alkoxy, (C1-C20) alkylcarbonyl, (C6-C20) arylcarbonyl, di (C1-C20) alkylamino or di (C6-C20) arylamino, or R 40 and R 4I may be linked via (C3-C12 ) alkylene or (C3-C12) alkenylene with or without
- (C3-C12) alkylene or (C3-C12) alkenylene with or without a fused ring may be further substituted by one or more substituent (s) selected from (C1-C20) alkyl with or without halogen substituent, halogen, cyano, tri (C1-C20) alkylsilyl, di (C1-C20) alkyl (C6-C20) arylsilyl, tri (C6-C20) arylsilyl, (C1-C20) alkoxy, (C1-C20) alkylcarbonyl, (C6-C20) arylcarbonyl, di (C1-C20) alkylamino, di (C6-C20) arylamino, phenyl, naphthyl, anthryl, fluorenyl and spirobifluorenyl, or may be further substituted by phenyl or fluorenyl with one or more substituent (s) selected
- R 42 through R 49 independently represent hydrogen, (C1-C20) alkyl with or without halogen substituent, (C1-C20) alkoxy, (C3-C12) cycloalkyl, halogen, cyano, (C6-C20) aryl, (C4-C20) heteroaryl, tri (C1-C20) alkylsilyl, di (Cl-C20)alkyl (C6-C20) arylsilyl or tri (C6-C20) arylsilyl; and
- R 5i through R 62 fe independently represent hydrogen, (C1-C60) alkyl with or without halogen substituent, (C1-C30) alkoxy, halogen, (C6-C60) aryl, cyano or (C5-C60) cycloalkyl, or each of R 51 through R 62 may be linked to an adjacent substituent via alkylene or alkenylene to form a (C5-C7)spiro ring or a (C5-C9) fused ring, or may be linked to Ri 7 or Ri 8 via alkylene or alkenylene to form a (C5-C7) fused ring.
- M 1 is selected from Ir, Pt, Pd, Rh, Re, Os, Tl, Pb, Bi, In, Sn, Sb, Te, Au and Ag.
- the compound represented by Chemical Formula 2 may be exemplified the following compounds, but not limited thereto:
- the electroluminescent device comprises the electroluminescent compound represented by Chemical Formula 1, and may further comprise one or more compound (s) selected from a group consisting of arylamine compounds and styrylarylamine compounds at the same time.
- the arylamine compound or the styrylarylamine compound may be a compound represented by Chemical Formula 3, but not limited thereto:
- Ari andAr 2 independently represent hydrogen, deuterium, halogen, (Cl-C ⁇ O)alkyl, (C6-C60) aryl, (C4-C60 ) heteroaryl, (C6-C60) arylamino, (C1-C60) alkylamino, 5- or 6-membered heterocycloalkyl containing one or more heteroatom (s) selected from N, 0 and S or (C3-C60) cycloalkyl, and Ari and Ar 2 may be linked via (C3-C60) alkylene or (C3-C60) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring;
- Ar 3 represents (C6-C60) aryl, (C4-C60) heteroaryl or a substituent selected from the following structures, when a is 1:
- Ar 3 represents (C6-C60) arylene, (C4-C60) heteroarylene or a substituent selected from the following structures, when a is 2:
- Ar 4 and Ar 5 independently represent (C6-C60 ) arylene or (C4-C60) heteroarylene;
- R 5 i f R 52 and R 53 independently represent hydrogen, halogen, deuterium, (C1-C60) alkyl or (C6-C60) aryl; b represents an integer from 1 to 4 ; c represents an integer 0 or 1; and the alkyl, aryl, heteroaryl, arylamino, alkylamino, cycloalkyl or heterocycloalkyl of Ari and A ⁇ 2l the aryl, heteroaryl, arylene or heteroarylene of Ar 3 , the arylene or heteroarylene of Ar 4 and Ars, or the alkyl or aryl of R 51 through R 53 may be further substituted by one or more substituent (s) selected from a group consisting of deuterium, halogen, (C1-C60) alkyl, (C6-C60) aryl, (C4-C60) heteroaryl, 5- or 6-membered heterocycloalkyl containing one or more heteroatom(s) selected from N, 0
- the organic layer may further comprise, in addition to the electroluminescent compound represented by Chemical Formula 1, one or more metal (s) selected from a group consisting of organic metals of Group 1, Group 2, 4th period and 5th period transition metals, lanthanide metals and d-transition elements .
- the organic layer may comprise an electroluminescent layer and a charge generating layer at the same time.
- An independent electroluminescence type electroluminescent device having a pixel structure in which the electroluminescent device according to the present invention comprising the electroluminescent compound represented by Chemical Formula 1 is used as a subpixel and one or more subpixel (s) comprising one or more metal compound (s) selected from a group consisting of Ir, Pt, Pd, Rh, Re, Os, Tl, Pb, Bi, In, Sn, Sb, Te, Au andAg is patterned in parallelmaybe manufactured.
- the organic layer may further comprise, in addition to the electroluminescent compound, one or more green- or blue-emitting organic compound (s) or organometallic compound (s) to form a white-emitting electroluminescent device.
- the green- or blue-emitting organic compound or organometallic compound may be exemplified by the compounds represented by Chemical Formulas 4 to 8, but not limited thereto:
- Arii andAr i2 independently represent (C1-C60) alkyl, (C6-C60) aryl, (C4-C60)heteroaryl, (C6-C60) arylamino, (C1-C60) alkylamino, 5- or 6-membered heterocycloalkyl containing one or more heteroatom (s) selected from N, 0 and S or (C3-C60) cycloalkyl, and Ar n and Ari 2 may be linked via (C3-C60) alkylene or (C3-C60) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring;
- Ar i3 represents (C6-C60) aryl, (C4-C60) heteroaryl or a substituent selected from the following structures, when d is 1:
- Ar i3 represents (C6-C60) arylene, (C4-C60) heteroarylene or a substituent selected from the following structures, when d is 2:
- Ar i4 and Ar 15 independently represent (C6-C60) arylene or (C4-C60) heteroarylene;
- R ⁇ i r R ⁇ 2 and R ⁇ j independently represent hydrogen, deuterium, (C1-C60) alkyl or (C6-C60) aryl; e represents an integer from 1 to 4; f represents an integer 0 or 1; the alkyl, aryl, heteroaryl, arylamino, alkylamino, cycloalkyl or heterocycloalkyl of Arn and AiTi 2 , the aryl, heteroaryl, arylene or heteroarylene of Ari 3 , the arylene or heteroarylene of Ari4 and Ar ⁇ 5 , or the alkyl or aryl of R 6 i through R 63 may be further substituted by one or more substituent (s) selected froma group consisting of deuterium, halogen, (C1-C60) alkyl, (C6-C60) aryl, (C4-C60) heteroaryl, 5- or
- R 7 i through R 74 independently represent hydrogen, deuterium, halogen, (C1-C60) alkyl, (C6-C60) aryl, (C4-C60) heteroaryl, 5- or 6-membered heterocycloalkyl containing one or more heteroatom (s) selected from N, 0 and S, (C3-C60) cycloalkyl, tri (C1-C60) alkylsilyl, di (C1-C60) alkyl (C6-C60) arylsilyl, tri (C6-C60) arylsilyl, adamantyl, (C7-C60)bicycloalkyl, (C2-C60) alkenyl, (C2-C60) alkynyl, cyano, (C1-C60) alkylamino, (C6-C60) arylamino, (C6-C60) ar (C1-C60) alkyl, (Cl-C ⁇ O)
- each of R 7 i through R 74 may be linked to an adjacent substituent via (C3-C60) alkylene or (C3-C60) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring; the alkyl, alkenyl, alkynyl, cycloalkyl, heterocycloalkyl, aryl, heteroaryl, arylsilyl, alkylsilyl, alkylamino or arylamino of R 7 i through R 74 , or the alicyclic ring or the monocyclic or polycyclic aromatic ring formed as they are linked to an adjacent substituent via (C3-C60) alkylene
- (C4-C60) heteroaryl 5- or 6-membered heterocycloalkyl containing one or more heteroatom (s) selected from N, O and S, (C3-C60) cycloalkyl, tri (C1-C60) alkylsilyl, di (C1-C60) alkyl (C6-C60) arylsilyl, tri (C6-C60) arylsilyl, adamantyl, (C7-C60) bicycloalkyl, (C2-C60) alkenyl, (C2-C60) alkynyl, cyano, (C1-C60) alkylamino, (C6-C60) arylamino, (C6-C60) ar (C1-C60) alkyl, (C1-C60) alkyloxy, (C1-C60) alkylthio, (C6-C60) aryloxy, (C6-C60) arylthio, (C
- Lu represents (C6-C60) arylene or (C4-C60) heteroarylene; Li 2 represent anthracenylene;
- Ar 2I through Rr 2A independently represent hydrogen, deuterium, (C1-C60) alkyl, (C1-C60) alkoxy, halogen, (C4-C60) heteroaryl, (C5-C60) cycloalkyl or (C6-C60) aryl, or the cycloalkyl, aryl or heteroaryl of Ar 2I through Ar 24 may be further substituted by one or more substituent (s) selected from a group consisting of (C6-C60)aryl or (C4-C60) heteroaryl with or without one or more substituent (s) selected from a group consisting of (C1-C60) alkyl with or without halogen substituent, (C1-C60) alkoxy, (C3-C60) cycloalkyl, halogen, cyano, tri (C1-C60) alkylsilyl, di (C1-C60) alkyl (C6-C60) arylsilyl and
- R 8 i and R 82 independently represent (C6-C60) aryl
- (C4-C60) heteroaryl 5- or 6-membered heterocycloalkyl containing one or more heteroatom (s) selected from N, 0 and S or (C3-C60) cycloalkyl, and the aryl or heteroaryl of R 8 i and R 82 may be further substituted by one or more substituent (s) selected from a group consisting of deuterium, (C1-C60) alkyl, halo (C1-C60) alkyl, (C1-C60) alkoxy,
- R 83 through R 86 independently represent hydrogen, deuterium, (Cl-C ⁇ O)alkyl, (C1-C60) alkoxy, halogen, (C4-C60) heteroaryl, (C5-C60) cycloalkyl or (C6-C60) aryl, and the heteroaryl, cycloalkyl or aryl of R 83 through R 86 may be further substituted by one or more substituent (s) selected from a group consisting of deuterium, (C1-C60) alkyl with or without halogen substituent, (C1-C60) alkoxy, (C3-C60) cycloalkyl, halogen, cyano, tri (C1-C60) alkylsilyl, di (C1-C60) alkyl (C6-C60) arylsilyl and tri (C6-C60) arylsilyl; Gi and G 2 independently represent a chemical bond or (C6-C60)
- Ar 3 I and Ar 32 represent (C4-C60) heteroaryl or aryl selected from the following structures:
- the aryl or heteroaryl of Ar 3I and Ar 32 may be substituted by one or more substituent (s) selected from deuterium, (C1-C60) alkyl, (C1-C60) alkoxy, (C6-C60)aryl and (C4-C60) heteroaryl;
- L 2I represents (C6-C60) arylene, (C4-C60) heteroarylene or a compound having the following structure:
- the arylene or heteroarylene of L 2 i may be substituted by one or more substituent (s) selected from deuterium, (C1-C60) alkyl, (C1-C60) alkoxy, (C6-C60) aryl, (C4-C60) heteroaryl and halogen;
- R 91A R 92 , R 93 and R 94 independently represent hydrogen, deuterium
- (C1-C60) alkyl or (C6-C60) aryl, or each of them may be linked to an adjacent substituent via (C3-C60) alkylene or (C3-C60) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring; and
- Rioi f R 102 , R 103 and R 104 independently represent hydrogen, deuterium, (C1-C60) alkyl, (C1-C60) alkoxy, (C6-C60) aryl, (C4-C60) heteroaryl or halogen, or each of them may be linked to an adjacent substituent via (C3-C60) alkylene or (C3-C60) alkenylene with or without a fused ring to form an alicyclic ring or a monocyclic or polycyclic aromatic ring.
- green- or blue-emitting organic compound or organometallic compound may be exemplified by the following compounds, but not limited thereto:
- a layer selected from a chalcogenide layer, a metal halide layer and a metal oxide layer may be placed on the inner surface of one or both electrode (s) among the pair of electrodes. More specifically, a chalcogenide (including oxide) layer of silicon or aluminum may be placed on the anode surface of the electroluminescent layer, and a metal halide layer or metal oxide layer may be placed on the cathode surface of the electroluminescent layer. A driving stability may be attained therefrom.
- the chalcogenide may be, for example, SiO x (1 ⁇ x ⁇ 2) , AlO x (1 ⁇ x ⁇ 1.5), SiON, SiAlON, etc.
- the metal halide may be, for example, LiF, MgF 2 , CaF 2 , a rare earth metal fluoride, etc.
- the metal oxide may be, for example, Cs 2 O, Li 2 O, MgO, SrO, BaO, CaO, etc.
- a mixed region of an electron transport compound and a reductive dopant or a mixed region of a hole transport compound and an oxidative dopant may be placed on the inner surface of one or both electrode (s) among the pair of electrodes.
- transport of electrons from the mixed region to the electroluminescent layer becomes easier, because the electron transport compound is reduced to an anion.
- transport of holes from the mixed region to the electroluminescent layer becomes easier, because the hole transport compound is oxidized to a cation.
- Preferred examples of the oxidative dopant include various Lewis acids and acceptor compounds .
- Preferred examples of the reductive dopant include alkali metals, alkali metal compounds, alkaline earth metals, rare earth metals and mixtures thereof.
- FIG. 1 is a cross-sectional view of an OLED device. ⁇ Description of symbols of significant parts of the drawing>
- Hole injecting layer 4 Hole transport layer
- Nickel (1.84 g, 2.8 mmol) was added to a 500 mL flask and argon gas was filled therein. After adding ether (50 mL) and Compound E (14.ImL, 42.15 mmol), the mixture was stirred for 30 minutes at room temperature. After adding Compound B (5.0 g, 14.0 mmol) dissolved in ether, the mixture was stirred for 48 hours under reflux. After cooling to room temperature, the mixture was poured to saturated ammonium chloride . After washing with water, followed by extraction with ethyl acetate, Compound F (2.0 g, 69%) was obtained by purification through silica column and drying.
- Electroluminescent compounds (Compounds 1 to 356) were prepared according to the method of Preparation Examples 1 and 2. Table 1 shows 1 H NMR and MS/FAB data of the prepared electroluminescent compounds. [Table 1]
- FIG. 1 An OLED device was manufactured using the electroluminescent compound according to the present invention as a host material.
- FIG 1 shows the cross-sectional view of the OLED device.
- the ITO substrate was mounted on a substrate holder of a vacuum deposition apparatus.
- a vacuum deposition apparatus After filling 4,4', 4"-tris (N, N- (2-naphthyl) -phenylamino) triphenylamine (2-TNATA) in a cell of the vacuum deposition apparatus, the pressure inside the chamber was reduced to 10 ⁇ 6 torr. Then, 2-TNATA was evaporatedby applying electrical current to the cell .
- Ahole injection layer 3 having a thickness of 60 nm was formed on the ITO substrate.
- NPB N 1 N 1 -bis ( ⁇ -naphthyl) -N, N' -diphenyl-4 , 4 '-diamine (NPB) in another cell of the vacuum deposition apparatus, NPB was evaporated by applying electrical current. A hole transport layer 4 having a thickness of 20 nm was formed on the hole injection layer.
- An electroluminescent layer 5 was formed on the hole transport layer as follows.
- the compound according to the present invention is
- an electroluminescent dopant e.g., (piq) 2 lr(acac)
- the two materials were evaporated at different speed, so that an electroluminescent layer 5 having a thickness of 30 nm was formed on the hole transport layer at 4 to 10 mol%.
- the electroluminescent compounds according to the present invention exhibited superior electroluminescence characteristics as compared to the existing material.
- Example 3 1) . Especially, the device of Example 3 was operated at a voltage
- Example 8 exhibited a driving voltage of 4.5 V and a power efficiency of 6.3 lm/W at 1000 cd/m 2 .
- the device using the electroluminescent compound according to the present invention as host material has excellent electroluminescence characteristics and power consumption can be improved by increasing power efficiency by 1.9-3.9 lm/W through decreased driving voltage.
- the electroluminescent compound according to the present invention When used for a host material of an electroluminescent material in an OLED device, the electroluminescent compound according to the present invention provides better red luminous efficiency as compared to existing host materials and excellent life property of the material . Therefore, it can be used tomanufacture OLEDs having very good operation life.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Electroluminescent Light Sources (AREA)
- Other In-Based Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2011536250A JP5690276B2 (en) | 2008-11-13 | 2009-11-13 | Novel organometallic compound for electroluminescence and electroluminescent device using the same as luminescent material |
| CN2009801454526A CN102216275A (en) | 2008-11-13 | 2009-11-13 | Novel organometallic compounds for electroluminescence and organic electroluminescent device using the same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2008-0112855 | 2008-11-13 | ||
| KR1020080112855A KR100901888B1 (en) | 2008-11-13 | 2008-11-13 | Novel organometalic compounds for electroluminescence and organic electroluminescent device using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2010056066A1 true WO2010056066A1 (en) | 2010-05-20 |
Family
ID=40982523
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2009/006693 WO2010056066A1 (en) | 2008-11-13 | 2009-11-13 | Novel organometallic compounds for electroluminescence and organic electroluminescent device using the same |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2202224A1 (en) |
| JP (1) | JP5690276B2 (en) |
| KR (1) | KR100901888B1 (en) |
| CN (1) | CN102216275A (en) |
| TW (1) | TWI428425B (en) |
| WO (1) | WO2010056066A1 (en) |
Cited By (86)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012069170A1 (en) * | 2010-11-22 | 2012-05-31 | Solvay Sa | A metal complex comprising a ligand having a combination of donor-acceptor substituents |
| JP2013080898A (en) * | 2011-09-22 | 2013-05-02 | Nippon Hoso Kyokai <Nhk> | Organic light-emitting element and display device using the same |
| US8716484B1 (en) | 2012-12-05 | 2014-05-06 | Universal Display Corporation | Hole transporting materials with twisted aryl groups |
| US8889864B2 (en) | 2006-02-10 | 2014-11-18 | Universal Display Corporation | Metal complexes of cyclometallated imidazo[1,2-f]phenanthridine and diimidazo[1,2-a:1′,2′-c]quinazoline ligands and isoelectronic and benzannulated analogs thereof |
| US9054344B2 (en) | 2010-01-20 | 2015-06-09 | Universal Display Corporation | Electroluminescent devices for lighting applications |
| US9196860B2 (en) | 2012-12-04 | 2015-11-24 | Universal Display Corporation | Compounds for triplet-triplet annihilation upconversion |
| US9252363B2 (en) | 2012-10-04 | 2016-02-02 | Universal Display Corporation | Aryloxyalkylcarboxylate solvent compositions for inkjet printing of organic layers |
| EP2915576A4 (en) * | 2012-11-05 | 2016-05-11 | Shanghai Inst Organic Chem | Extracting agent for separating lithium isotopes and use thereof |
| EP3056504A1 (en) | 2015-02-16 | 2016-08-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3061763A1 (en) | 2015-02-27 | 2016-08-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9450198B2 (en) | 2014-04-15 | 2016-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3098229A1 (en) | 2015-05-15 | 2016-11-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3101021A1 (en) | 2015-06-01 | 2016-12-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3124488A1 (en) | 2015-07-29 | 2017-02-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3159350A1 (en) | 2015-09-03 | 2017-04-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9653691B2 (en) | 2012-12-12 | 2017-05-16 | Universal Display Corporation | Phosphorescence-sensitizing fluorescence material system |
| EP3205658A1 (en) | 2016-02-09 | 2017-08-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3231809A2 (en) | 2016-04-11 | 2017-10-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3261147A1 (en) | 2016-06-20 | 2017-12-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3261146A2 (en) | 2016-06-20 | 2017-12-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3270435A2 (en) | 2016-06-20 | 2018-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9876173B2 (en) | 2013-12-09 | 2018-01-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3297051A1 (en) | 2016-09-14 | 2018-03-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3301088A1 (en) | 2016-10-03 | 2018-04-04 | Universal Display Corporation | Condensed pyridines as organic electroluminescent materials and devices |
| EP3305796A1 (en) | 2016-10-07 | 2018-04-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3321258A1 (en) | 2016-11-09 | 2018-05-16 | Universal Display Corporation | 4-phenylbenzo[g]quinazoline or 4-(3,5-dimethylphenylbenzo[g]quinazoline iridium complexes for use as near-infrared or infrared emitting materials in oleds |
| EP3323822A1 (en) | 2016-09-23 | 2018-05-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3345914A1 (en) | 2017-01-09 | 2018-07-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3354654A2 (en) | 2016-11-11 | 2018-08-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3381927A1 (en) | 2017-03-29 | 2018-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3401318A1 (en) | 2017-05-11 | 2018-11-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3418286A1 (en) | 2017-06-23 | 2018-12-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3444258A2 (en) | 2017-08-10 | 2019-02-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3489243A1 (en) | 2017-11-28 | 2019-05-29 | University of Southern California | Carbene compounds and organic electroluminescent devices |
| EP3492480A2 (en) | 2017-11-29 | 2019-06-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3492528A1 (en) | 2017-11-30 | 2019-06-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3613751A1 (en) | 2018-08-22 | 2020-02-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN111004290A (en) * | 2019-12-11 | 2020-04-14 | 吉林奥来德光电材料股份有限公司 | Organic phosphorus luminescent compound, preparation method thereof and organic electroluminescent device |
| DE102013200085B4 (en) | 2012-01-06 | 2020-06-18 | Universal Display Corporation | Highly efficient phosphorescent materials |
| EP3690973A1 (en) | 2019-01-30 | 2020-08-05 | University Of Southern California | Organic electroluminescent materials and devices |
| EP3689889A1 (en) | 2019-02-01 | 2020-08-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3709376A1 (en) | 2019-03-12 | 2020-09-16 | Universal Display Corporation | Oled with triplet emitter and excited state lifetime less than 200 ns |
| EP3715353A1 (en) | 2019-03-26 | 2020-09-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3750897A1 (en) | 2019-06-10 | 2020-12-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN112300140A (en) * | 2014-07-09 | 2021-02-02 | 环球展览公司 | Organic electroluminescent material and device |
| EP3771717A1 (en) | 2019-07-30 | 2021-02-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3778614A1 (en) | 2019-08-16 | 2021-02-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3816175A1 (en) | 2019-11-04 | 2021-05-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3823055A1 (en) | 2019-11-14 | 2021-05-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3845545A1 (en) | 2020-01-06 | 2021-07-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3858945A1 (en) | 2020-01-28 | 2021-08-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3937268A1 (en) | 2020-07-10 | 2022-01-12 | Universal Display Corporation | Plasmonic oleds and vertical dipole emitters |
| EP4001286A1 (en) | 2020-11-24 | 2022-05-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4001287A1 (en) | 2020-11-24 | 2022-05-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4016659A1 (en) | 2020-11-16 | 2022-06-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4019526A1 (en) | 2018-01-26 | 2022-06-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4039692A1 (en) | 2021-02-03 | 2022-08-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4053137A1 (en) | 2021-03-05 | 2022-09-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4056578A1 (en) | 2021-03-12 | 2022-09-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4059915A2 (en) | 2021-02-26 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4060758A2 (en) | 2021-02-26 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4059941A1 (en) | 2021-03-15 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4075530A1 (en) | 2021-04-14 | 2022-10-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4075531A1 (en) | 2021-04-13 | 2022-10-19 | Universal Display Corporation | Plasmonic oleds and vertical dipole emitters |
| EP4074723A1 (en) | 2021-04-05 | 2022-10-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4079743A1 (en) | 2021-04-23 | 2022-10-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4086266A1 (en) | 2021-04-23 | 2022-11-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4112701A2 (en) | 2021-06-08 | 2023-01-04 | University of Southern California | Molecular alignment of homoleptic iridium phosphors |
| DE102013214144B4 (en) | 2012-07-19 | 2023-02-16 | Universal Display Corp. | Diarylamino-substituted metal complexes |
| EP4151699A1 (en) | 2021-09-17 | 2023-03-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4185086A1 (en) | 2017-07-26 | 2023-05-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4212539A1 (en) | 2021-12-16 | 2023-07-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| DE102013003605B4 (en) | 2012-03-15 | 2023-08-17 | Universal Display Corporation | Secondary hole transport layer with tricarbazole compounds |
| EP4242285A1 (en) | 2022-03-09 | 2023-09-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4265626A2 (en) | 2022-04-18 | 2023-10-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4282863A1 (en) | 2022-05-24 | 2023-11-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4293001A1 (en) | 2022-06-08 | 2023-12-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4299693A1 (en) | 2022-06-28 | 2024-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4326030A1 (en) | 2022-08-17 | 2024-02-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362630A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362631A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362645A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4369898A1 (en) | 2022-10-27 | 2024-05-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4376583A2 (en) | 2022-10-27 | 2024-05-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4386065A1 (en) | 2022-12-14 | 2024-06-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| DE102014001468B4 (en) | 2013-02-08 | 2024-08-08 | Universal Display Corporation | HETEROLEPTIC PHENYLBENZIMIDAZOLE COMPLEX |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6511657B2 (en) * | 2014-09-03 | 2019-05-15 | 日本放送協会 | ORGANIC METAL COMPLEX, ORGANIC ELECTROLUMINESCENT DEVICE AND METHOD FOR MANUFACTURING THE SAME, DISPLAY DEVICE, LIGHTING DEVICE, ORGANIC THIN FILM SOLAR CELL |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06322362A (en) * | 1993-03-17 | 1994-11-22 | Sanyo Electric Co Ltd | Electroluminescent element |
| JP2000012222A (en) * | 1998-06-19 | 2000-01-14 | Toray Ind Inc | Light-emitting element |
| JP2003077671A (en) * | 2001-09-03 | 2003-03-14 | Canon Inc | Organic light emitting element |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5466392A (en) * | 1993-06-02 | 1995-11-14 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and compound having an aluminum complex structure |
| JPH0748385A (en) * | 1993-06-02 | 1995-02-21 | Idemitsu Kosan Co Ltd | Organic electroluminescent element and compound having aluminum complex structure |
| JP2875463B2 (en) * | 1993-11-10 | 1999-03-31 | 新光電気工業株式会社 | Light emitting material for EL element and EL element |
| JP3555253B2 (en) * | 1995-07-07 | 2004-08-18 | 東洋インキ製造株式会社 | Organic electroluminescent device material and organic electroluminescent device using the same |
| JPH1140355A (en) * | 1997-07-14 | 1999-02-12 | Toyo Ink Mfg Co Ltd | Organic electroluminescent element material and organic electroluminescent element using the material |
| US6406804B1 (en) * | 1998-04-09 | 2002-06-18 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device |
| US6656608B1 (en) * | 1998-12-25 | 2003-12-02 | Konica Corporation | Electroluminescent material, electroluminescent element and color conversion filter |
| JP3968933B2 (en) * | 1998-12-25 | 2007-08-29 | コニカミノルタホールディングス株式会社 | Electroluminescence element |
| JP2004091382A (en) * | 2002-08-30 | 2004-03-25 | Mitsubishi Chemicals Corp | Organometallic complex and organic electroluminescent element using the same |
| KR100773523B1 (en) * | 2003-11-06 | 2007-11-07 | 삼성에스디아이 주식회사 | Organometallic complexes and organic electroluminescence device using the same |
| KR100773524B1 (en) * | 2003-12-24 | 2007-11-07 | 삼성에스디아이 주식회사 | Binuclear organometallic complexes and organic electroluminescent display device using the same |
| KR20050122943A (en) * | 2004-06-26 | 2005-12-29 | 주식회사 코오롱 | Electroluminescence iridium compound and display device adopting as light-emitting dopant |
| KR100611852B1 (en) * | 2004-11-11 | 2006-08-11 | 에스케이씨 주식회사 | Phosphorescent red-emitting iridium complex and organic electroluminescent device comprising same |
| US8518558B2 (en) * | 2007-03-29 | 2013-08-27 | Konica Minolta Holdings, Inc. | Organic electroluminescent element |
| KR100850886B1 (en) * | 2007-09-07 | 2008-08-07 | (주)그라쎌 | Organometalic compounds for electroluminescence and organic electroluminescent device using the same |
-
2008
- 2008-11-13 KR KR1020080112855A patent/KR100901888B1/en not_active IP Right Cessation
-
2009
- 2009-03-30 EP EP09156568A patent/EP2202224A1/en not_active Withdrawn
- 2009-11-12 TW TW098138368A patent/TWI428425B/en not_active IP Right Cessation
- 2009-11-13 WO PCT/KR2009/006693 patent/WO2010056066A1/en active Application Filing
- 2009-11-13 JP JP2011536250A patent/JP5690276B2/en not_active Expired - Fee Related
- 2009-11-13 CN CN2009801454526A patent/CN102216275A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06322362A (en) * | 1993-03-17 | 1994-11-22 | Sanyo Electric Co Ltd | Electroluminescent element |
| JP2000012222A (en) * | 1998-06-19 | 2000-01-14 | Toray Ind Inc | Light-emitting element |
| JP2003077671A (en) * | 2001-09-03 | 2003-03-14 | Canon Inc | Organic light emitting element |
Non-Patent Citations (5)
Cited By (117)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8889864B2 (en) | 2006-02-10 | 2014-11-18 | Universal Display Corporation | Metal complexes of cyclometallated imidazo[1,2-f]phenanthridine and diimidazo[1,2-a:1′,2′-c]quinazoline ligands and isoelectronic and benzannulated analogs thereof |
| US9065063B2 (en) | 2006-02-10 | 2015-06-23 | Universal Display Corporation | Metal complexes of cyclometallated imidazo[1,2-f]phenanthridine and diimidazo[1,2-a:1′,2′-c]quinazoline ligands and isoelectronic and benzannulated analogs thereof |
| US9548462B2 (en) | 2006-02-10 | 2017-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9281483B2 (en) | 2006-02-10 | 2016-03-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9054344B2 (en) | 2010-01-20 | 2015-06-09 | Universal Display Corporation | Electroluminescent devices for lighting applications |
| JP2013545754A (en) * | 2010-11-22 | 2013-12-26 | ソルヴェイ(ソシエテ アノニム) | Metal complexes containing ligands with a combination of donor and acceptor substituents |
| WO2012069170A1 (en) * | 2010-11-22 | 2012-05-31 | Solvay Sa | A metal complex comprising a ligand having a combination of donor-acceptor substituents |
| JP2013080898A (en) * | 2011-09-22 | 2013-05-02 | Nippon Hoso Kyokai <Nhk> | Organic light-emitting element and display device using the same |
| DE102013200085B4 (en) | 2012-01-06 | 2020-06-18 | Universal Display Corporation | Highly efficient phosphorescent materials |
| DE102013003605B4 (en) | 2012-03-15 | 2023-08-17 | Universal Display Corporation | Secondary hole transport layer with tricarbazole compounds |
| DE102013214144B4 (en) | 2012-07-19 | 2023-02-16 | Universal Display Corp. | Diarylamino-substituted metal complexes |
| US9252363B2 (en) | 2012-10-04 | 2016-02-02 | Universal Display Corporation | Aryloxyalkylcarboxylate solvent compositions for inkjet printing of organic layers |
| EP2915576A4 (en) * | 2012-11-05 | 2016-05-11 | Shanghai Inst Organic Chem | Extracting agent for separating lithium isotopes and use thereof |
| US9196860B2 (en) | 2012-12-04 | 2015-11-24 | Universal Display Corporation | Compounds for triplet-triplet annihilation upconversion |
| US8716484B1 (en) | 2012-12-05 | 2014-05-06 | Universal Display Corporation | Hole transporting materials with twisted aryl groups |
| US9653691B2 (en) | 2012-12-12 | 2017-05-16 | Universal Display Corporation | Phosphorescence-sensitizing fluorescence material system |
| DE102014001468B4 (en) | 2013-02-08 | 2024-08-08 | Universal Display Corporation | HETEROLEPTIC PHENYLBENZIMIDAZOLE COMPLEX |
| US9876173B2 (en) | 2013-12-09 | 2018-01-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9450198B2 (en) | 2014-04-15 | 2016-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN112300140A (en) * | 2014-07-09 | 2021-02-02 | 环球展览公司 | Organic electroluminescent material and device |
| US11957047B2 (en) | 2014-07-09 | 2024-04-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3056504A1 (en) | 2015-02-16 | 2016-08-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3061763A1 (en) | 2015-02-27 | 2016-08-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3098229A1 (en) | 2015-05-15 | 2016-11-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3101021A1 (en) | 2015-06-01 | 2016-12-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3124488A1 (en) | 2015-07-29 | 2017-02-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3159350A1 (en) | 2015-09-03 | 2017-04-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3760635A1 (en) | 2015-09-03 | 2021-01-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3205658A1 (en) | 2016-02-09 | 2017-08-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3858842A1 (en) | 2016-02-09 | 2021-08-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4122941A1 (en) | 2016-04-11 | 2023-01-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3231809A2 (en) | 2016-04-11 | 2017-10-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3843171A1 (en) | 2016-06-20 | 2021-06-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3920254A1 (en) | 2016-06-20 | 2021-12-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3261146A2 (en) | 2016-06-20 | 2017-12-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4349935A2 (en) | 2016-06-20 | 2024-04-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3758084A1 (en) | 2016-06-20 | 2020-12-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3261147A1 (en) | 2016-06-20 | 2017-12-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3270435A2 (en) | 2016-06-20 | 2018-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3297051A1 (en) | 2016-09-14 | 2018-03-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3323822A1 (en) | 2016-09-23 | 2018-05-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3301088A1 (en) | 2016-10-03 | 2018-04-04 | Universal Display Corporation | Condensed pyridines as organic electroluminescent materials and devices |
| EP3858844A1 (en) | 2016-10-07 | 2021-08-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3305796A1 (en) | 2016-10-07 | 2018-04-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3321258A1 (en) | 2016-11-09 | 2018-05-16 | Universal Display Corporation | 4-phenylbenzo[g]quinazoline or 4-(3,5-dimethylphenylbenzo[g]quinazoline iridium complexes for use as near-infrared or infrared emitting materials in oleds |
| EP3789379A1 (en) | 2016-11-09 | 2021-03-10 | Universal Display Corporation | 4-phenylbenzo[g]quinazoline or 4-(3,5-dimethylphenylbenzo[g]quinazoline iridium complexes for use as near-infrared or infrared emitting materials in oleds |
| EP3354654A2 (en) | 2016-11-11 | 2018-08-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4092036A1 (en) | 2016-11-11 | 2022-11-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3345914A1 (en) | 2017-01-09 | 2018-07-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4212540A1 (en) | 2017-01-09 | 2023-07-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3689890A1 (en) | 2017-01-09 | 2020-08-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3985012A1 (en) | 2017-03-29 | 2022-04-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3730506A1 (en) | 2017-03-29 | 2020-10-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3381927A1 (en) | 2017-03-29 | 2018-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4141010A1 (en) | 2017-05-11 | 2023-03-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3401318A1 (en) | 2017-05-11 | 2018-11-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3418286A1 (en) | 2017-06-23 | 2018-12-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4185086A1 (en) | 2017-07-26 | 2023-05-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3444258A2 (en) | 2017-08-10 | 2019-02-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3783006A1 (en) | 2017-08-10 | 2021-02-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3878855A1 (en) | 2017-11-28 | 2021-09-15 | University of Southern California | Carbene compounds and organic electroluminescent devices |
| EP3489243A1 (en) | 2017-11-28 | 2019-05-29 | University of Southern California | Carbene compounds and organic electroluminescent devices |
| EP3492480A2 (en) | 2017-11-29 | 2019-06-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3492528A1 (en) | 2017-11-30 | 2019-06-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4019526A1 (en) | 2018-01-26 | 2022-06-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4206210A1 (en) | 2018-08-22 | 2023-07-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3613751A1 (en) | 2018-08-22 | 2020-02-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3690973A1 (en) | 2019-01-30 | 2020-08-05 | University Of Southern California | Organic electroluminescent materials and devices |
| EP3689889A1 (en) | 2019-02-01 | 2020-08-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4301117A2 (en) | 2019-02-01 | 2024-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3709376A1 (en) | 2019-03-12 | 2020-09-16 | Universal Display Corporation | Oled with triplet emitter and excited state lifetime less than 200 ns |
| EP4134371A2 (en) | 2019-03-26 | 2023-02-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3715353A1 (en) | 2019-03-26 | 2020-09-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3750897A1 (en) | 2019-06-10 | 2020-12-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4219515A1 (en) | 2019-07-30 | 2023-08-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3771717A1 (en) | 2019-07-30 | 2021-02-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3778614A1 (en) | 2019-08-16 | 2021-02-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3816175A1 (en) | 2019-11-04 | 2021-05-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3823055A1 (en) | 2019-11-14 | 2021-05-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN111004290B (en) * | 2019-12-11 | 2023-08-01 | 吉林奥来德光电材料股份有限公司 | Organic phosphorus luminescent compound, preparation method thereof and organic electroluminescent device |
| CN111004290A (en) * | 2019-12-11 | 2020-04-14 | 吉林奥来德光电材料股份有限公司 | Organic phosphorus luminescent compound, preparation method thereof and organic electroluminescent device |
| EP3845545A1 (en) | 2020-01-06 | 2021-07-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4151644A1 (en) | 2020-01-06 | 2023-03-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4294157A2 (en) | 2020-01-28 | 2023-12-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3858945A1 (en) | 2020-01-28 | 2021-08-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP3937268A1 (en) | 2020-07-10 | 2022-01-12 | Universal Display Corporation | Plasmonic oleds and vertical dipole emitters |
| EP4016659A1 (en) | 2020-11-16 | 2022-06-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4001286A1 (en) | 2020-11-24 | 2022-05-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4001287A1 (en) | 2020-11-24 | 2022-05-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4329463A2 (en) | 2020-11-24 | 2024-02-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4039692A1 (en) | 2021-02-03 | 2022-08-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4060758A2 (en) | 2021-02-26 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4059915A2 (en) | 2021-02-26 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4053137A1 (en) | 2021-03-05 | 2022-09-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4056578A1 (en) | 2021-03-12 | 2022-09-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4059941A1 (en) | 2021-03-15 | 2022-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4074723A1 (en) | 2021-04-05 | 2022-10-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4075531A1 (en) | 2021-04-13 | 2022-10-19 | Universal Display Corporation | Plasmonic oleds and vertical dipole emitters |
| EP4075530A1 (en) | 2021-04-14 | 2022-10-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4401530A2 (en) | 2021-04-14 | 2024-07-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4086266A1 (en) | 2021-04-23 | 2022-11-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4079743A1 (en) | 2021-04-23 | 2022-10-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4112701A2 (en) | 2021-06-08 | 2023-01-04 | University of Southern California | Molecular alignment of homoleptic iridium phosphors |
| EP4151699A1 (en) | 2021-09-17 | 2023-03-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4212539A1 (en) | 2021-12-16 | 2023-07-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4242285A1 (en) | 2022-03-09 | 2023-09-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4265626A2 (en) | 2022-04-18 | 2023-10-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4282863A1 (en) | 2022-05-24 | 2023-11-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4293001A1 (en) | 2022-06-08 | 2023-12-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4299693A1 (en) | 2022-06-28 | 2024-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4326030A1 (en) | 2022-08-17 | 2024-02-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362645A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4369898A1 (en) | 2022-10-27 | 2024-05-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4376583A2 (en) | 2022-10-27 | 2024-05-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362631A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4362630A2 (en) | 2022-10-27 | 2024-05-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP4386065A1 (en) | 2022-12-14 | 2024-06-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2202224A1 (en) | 2010-06-30 |
| JP5690276B2 (en) | 2015-03-25 |
| TW201035278A (en) | 2010-10-01 |
| TWI428425B (en) | 2014-03-01 |
| CN102216275A (en) | 2011-10-12 |
| KR100901888B1 (en) | 2009-06-09 |
| JP2012508732A (en) | 2012-04-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2202224A1 (en) | Novel Organometallic Compounds for Electroluminescence and Organic Electroluminescent Device Using the Same | |
| EP2182002B1 (en) | Novel compounds for electronic material and organic electronic device using the same | |
| KR101376530B1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| EP2085450A1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN102224217B (en) | Electroluminescent device using the electroluminescent compounds | |
| EP2066150B1 (en) | Novel red electroluminescent compounds and organic electroluminescent device using the same | |
| EP2062958B1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR101495547B1 (en) | Novel compounds for electronic material and organic electronic device using the same | |
| EP2098532A1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| JP5554914B2 (en) | Novel organic electroluminescent compound and organic electroluminescent device using the same | |
| EP2182038A1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| EP2161319A2 (en) | Benzo(c)phenanthrene derivatives and organic electroluminescent device using the same | |
| EP2103665A1 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| EP2281863A2 (en) | Fluorene-derivatives and organic electroluminescent device using the same | |
| EP2058322A2 (en) | Novel red electroluminescent compounds and organic electroluminescent device using the same | |
| EP2194110A1 (en) | Electroluminescent device using electroluminescent compounds | |
| WO2010050778A1 (en) | Novel compounds for organic electronic material and organic electronic device using the same | |
| JP2009173630A (en) | New organic light-emitting compound and organic light-emitting device using the same | |
| EP2161320A2 (en) | Fluorene derivatives and organic electroluminescent device using the same | |
| EP2103666A2 (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| EP2055709A2 (en) | Novel red electroluminescent compounds and organic electroluminescent device using the same | |
| JP2009249385A (en) | New organic electroluminescent compound, and organic electroluminescent element using the same | |
| EP2060579A1 (en) | Novel red electroluminescent compounds and organic electroluminescent device using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980145452.6 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09826298 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2011536250 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 09826298 Country of ref document: EP Kind code of ref document: A1 |