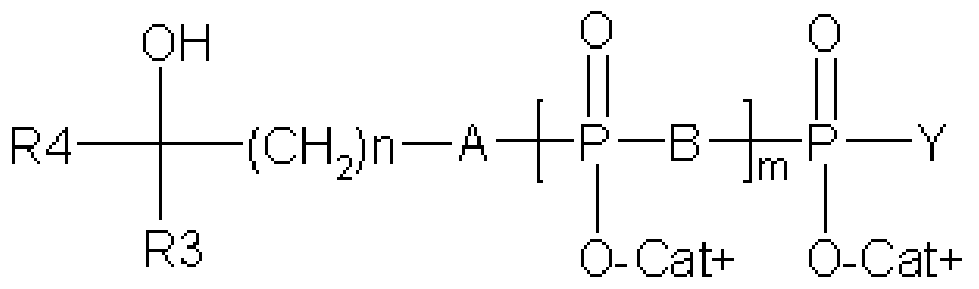WO2010049438A2 - Improved methods of using phosphoantigens for the treatment of diseases - Google Patents
Improved methods of using phosphoantigens for the treatment of diseases Download PDFInfo
- Publication number
- WO2010049438A2 WO2010049438A2 PCT/EP2009/064188 EP2009064188W WO2010049438A2 WO 2010049438 A2 WO2010049438 A2 WO 2010049438A2 EP 2009064188 W EP2009064188 W EP 2009064188W WO 2010049438 A2 WO2010049438 A2 WO 2010049438A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- cell
- cell activator
- ifnα
- disease
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/662—Phosphorus acids or esters thereof having P—C bonds, e.g. foscarnet, trichlorfon
- A61K31/663—Compounds having two or more phosphorus acid groups or esters thereof, e.g. clodronic acid, pamidronic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/19—Cytokines; Lymphokines; Interferons
- A61K38/21—Interferons [IFN]
- A61K38/212—IFN-alpha
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
Definitions
- the present invention relates to compositions and methods for regulating an immune response in a subject, particularly a T cell response in a subject.
- the present invention more specifically discloses efficient methods of regulating the innate immunity in a subject, such as by regulating the activity of gamma-delta T cells in a subject.
- the invention discloses that particular combinations of particular agents, such as a type I interferon cytokine and a gamma-delta T cell activator, can produce a remarkable activation of gamma-delta ( ⁇ ) T cells in vivo, a remarkable replication inhibition of virus replication and a remarkable increase in a subject's immune defense, against an infection, a cancer or an immune disease.
- the invention can be used for therapeutic purposes, to produce, regulate or facilitate an immune response in a subject infected by a disease. It is particularly suited to regulate a protective immune response in subjects having a proliferative disease, an infectious disease or an autoimmune disease.
- T cells of the gamma-delta type are expressed by most mammalian species. They represent 1- 10% of total circulating lymphocytes in healthy adult human subjects and most non-human primates (NHP). Most human peripheral blood gamma-delta T cells express a gamma-delta TCR heterodimer encoded by Vgamma9/Vdelta2 genes, some NK-lineage receptors for MHC class I and almost no CD4 nor CD8. These cells have been shown to exhibit strong, non MHC-restricted, cytolytic activity against virus-infected cells (Poccia et al, J. Leukocyte Biology, 62, 1997, p.
- ⁇ T cells represent a very attractive candidate for immunotherapy.
- activated ⁇ T cells exert potent cytolytic activity and T n -I cytokine secretion, these cells represent an important resource of anti-infectious and antitumoral effectors. Accordingly, it would be highly valuable to have methods available for regulating the activity (including the expansion and/or cytolytic activity, for instance), of these cells in vivo, in a subject.
- V ⁇ 9/V ⁇ 2 + lymphocytes spontaneously recognize a structurally related set of nonpeptide antigens, referred to as natural phosphoantigens and alkylamines.
- natural phosphoantigens and alkylamines In B cell tumors, the nature of antigens for the ⁇ T cells remains unidentified.
- V ⁇ 9/V ⁇ 2 + lymphocytes are also responsive to a variety of virally infected-, activated- or tumoral cell types without prior exposure. Again, in these situations, the responsible antigens remain unknown. It has been shown that, in vitro, V ⁇ 9/V ⁇ 2 + lymphocytes respond to synthetic drugs such as therapeutic aminobisphosphonates (reviewed in Espinosa, J Biol Chem 2001, Vol. 276(21), pp. 18337-18344), leading to their in vitro activation.
- V ⁇ 9/V ⁇ 2 + lymphocyte activation by non-peptide antigens appears to require cell-to-cell contact (Lang, J. Immunol. 1995 (154) 5986; Morita, Immunity, Vol. 3, 495-507, 1995; Miyagawa, J. Immunol 2001, 166: 5508-5514; Rojas, Infection and Immunity, 2002, vol70(8)).
- the first set of clinical evidence for in vivo expansion of human V ⁇ 9/V ⁇ 2 + lymphocytes induced by phosphoantigen agonists came from the finding of increases of circulating ⁇ T cells within one to three weeks in human adults with multiple myeloma after therapeutic intravenous injection of 60-90 mg of pamidronate (Kunzmann,Blood, 2000, vol 96(2).
- the actual mode of action of pamidronate is unclear and might involve an indirect effect on accessory cells (Miyagawa, 2001) and no appropriate conditions were disclosed to allow optimized activation of ⁇ T cells using such compound.
- the inventors and/or their colleagues have previously characterized a number of immunomodulatory compounds capable of modulating the activity and proliferation of ⁇ T cells. These compounds (also called "phosphoantigens") generally share a common structure in that they are organophosphate compounds. The classes having greatest potency are more particularly phosphate esters and phospho-phosphoroamidate esters. The particular compounds, used by the inventors and their colleagues in a lead clinical trial, comprise a pyrophosphate moiety. The lead molecules of this series of compounds developed by the inventors and their colleagues are called PhosphostimTM, which is at present being evaluated in three Phase II clinical trials in oncology and PicostimTM currently at the preclinical stage. A number of other compounds which activate ⁇ T cells are known as well, although most of these act indirectly (e.g. act on other immune cells which in turn activate ⁇ T cells) or act directly on ⁇ T cells but are less potent.
- ⁇ T cells could represent an additional strategy to inhibit HCV replication, ⁇ T cells activated by phosphoantigens induce a non-cytolytic inhibition of subgenomic HCV replication.
- the present invention proposes a treatment setting useful for treating a disease wherein the administration of type I interferon (type I IFN) is needed.
- Type I IFN includes in particular IFN ⁇ and
- IFN ⁇ Infectious diseases such as HCV and HBV are currently treated with IFN ⁇ but any other disease where the administration of a type I IFN may be beneficial is encompassed by the present invention, for example other infections such as HIV, HTLV-I, HPV, HHV8.
- the present invention is also suited for the treatment of subjects having more than one infection and wherein the administration of a type I IFN may be beneficial for the treatment of at least one of the infections, e.g. co-infected subjects, i.e. HCV/ AIDS co-infected subjects, HCV/Tuberculosis co-infected subjects and the like.
- the present invention is also suited for the treatment a proliferative disease, in particular a cancer.
- Cancers such as melanoma or kidney cancer are currently treated with IFN ⁇ but any other cancer where the administration of a type I IFN may be beneficial is encompassed by the present invention, for example, other cancers such as Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors.
- other cancers such as Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors.
- the method and compositions according to the present invention enable to strengthen the ⁇ T cell response thereby enhancing the potency of clearance of viruses, tumors and autoimmune wounds.
- the present invention provides a composition where IFN ⁇ sensitizes ⁇ T cells that are weak responders to ⁇ T cell activator stimulation.
- the combination of a type I IFN and a ⁇ T cell activator potentiates the cytokine release and activation of ⁇ T cells compared to single ⁇ T cell stimulation.
- the present invention thus provides a method to treat patients weakly responding to a ⁇ T cell activator by further administering an effective dose of a type I IFN.
- the immune response is only partial which leads to a partial control of the acute phase of the disease and a residual disease persists.
- a residual disease persists.
- some copies of the virus persist, in tumors, some tumor cells may persist as well.
- These residual disease markers may lead to a chronic disease or the redevelopment of said disease, it is therefore crucial to mount a total and effective immune response to lead to a total clearance of the disease and cure patients.
- the present invention provides a method to clear an acute phase infection comprising administering to a subject in need thereof an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN.
- Treg control immune responses to self- and foreign- antigens and play a major role in maintaining the balance between immunity and tolerance (Sakaguchi S. et al, Annu Rev Immunol. 2004;22:531-562, Tang Q et al , Nat Immunol. 2008;9:239-244).
- Treg have been shown to broadly suppress activation, proliferation and/or effector functions of various immune cell populations, in particular in ⁇ T cells (Li et al, Blood. 2008 Jun 15; 5629-36), thereby leading to an impaired response to therapy, i.e. lead to chronic infection when acute phase is not totally cleared.
- IL-2 administration can lead to an increase in the frequency of circulating CD4 + CD25 + regulatory T cells in cancer patients (Ahmadzadeh M, et al, Blood. 2006; 2409-2414).
- the inventors have established that the administration of a type I IFN, together with a ⁇ T cell activator and IL-2 leads to a decreased production of Treg, thereby improving the potency of ⁇ T cell response in a subject.
- the present invention provides also a method to reduce the proportion of Treg in a patient.
- the present invention provides a method to obtain a sustained response of successive ⁇ T cell activator administrations in a subject.
- the present invention provides a method to enhance cytokine secretion in a subject comprising administering an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN.
- the invention also provides a method for enhancing the cytokine secretion in a subject comprising contacting ⁇ T cells with an effective amount of a ⁇ T cell activator and an effective amount of type I IFN, especially IFN ⁇ . In an embodiment, said method is performed without further administering IL-2 to said subject.
- the present invention provides a method for enhancing the cytokine secretion of ⁇ T cells activated with a ⁇ T cell activator comprising contacting said activated ⁇ T cells with an effective amount of a type I IFN, especially IFN ⁇ . In an embodiment, said method is performed without further administering IL-2 to said subject.
- Type I IFN treatment is the induced toxicity in patients, reported undesirable side effects include headache, fever, flu like syndrome and the like. The effects are highly undesirable, particularly, in non symptomatic patients, who do not feel sick when infected but who may experience the listed undesirable side effects upon treatment.
- One object of the present invention is also to provide a treatment necessitating a smaller amount of type I IFN for the obtention of a similar therapeutic effect, and thus inducing less undesirable side effects than currently described monotherapy treatments.
- the present invention also provides a method of reducing an type I IFN treatment-related side effect in a subject receiving or in need of treatment with a type I IFN, comprising administering to a subject in need of treatment with a type I IFN, a pharmaceutical composition comprising type I IFN and a pharmaceutical composition comprising a ⁇ T cell activator in an amount which, together with the administered type I IFN results in an effective treatment having a reduced type I IFN treatment related side effect in comparison with the side effects that would occur upon sole effective type I IFN treatment.
- the present invention provides a method to inhibit viral replication comprising contacting supernatants of activated ⁇ T cells with a virus.
- the invention also provides a composition comprising IFN ⁇ and a ⁇ T cell activator for the treatment of an infectious disease, wherein the antiviral effect of the combined products is greater than each antiviral effect of each product alone.
- the invention also provides a composition comprising IFN ⁇ and a ⁇ T cell activator for the treatment of an infectious disease wherein the combination enables to clear chronic infection, in particular HCV infection.
- the present invention also provides a composition comprising IFN ⁇ and a ⁇ T cell activator to obtain a sustained virologic response (SVR) in virally infected patient, in particular HCV patients.
- SVR sustained virologic response
- the present invention is based on observations during (i) in vitro testing of activated ⁇ T cells ability to inhibit virus replication and (ii) studies in non-human primates human clinical trial using CHDMAPP (also referred to as PicostimTM), where it was observed that this compound, when used in certain therapeutic regimens with type I interferon leads to an improved ⁇ T cell activation and proliferation, including a strong cytokine secretion, an efficient ⁇ T cell activation. In viral diseases, the present invention also proves an efficient viral replication inhibition.
- the present invention therefore provides novel approaches and strategies for efficient regulation of ⁇ T cells in vivo and therefore disease management, in a subject, particularly a human subject or a non-human primate, wherein the administration of type I IFN can be beneficial.
- the disease is selected from a cancer, an infection or an autoimmune disease.
- the present invention now discloses particular compositions and methods that can be used to treat diseases, comprising inducing the inhibition of viruses in vivo, diminishing the tumor burden, or controlling an autoimmune disease, in a subject, through ⁇ T cell activation. These compositions and methods employ the conjoint treatment of a subject with a ⁇ T cell activator and type I IFN.
- the present invention provides a method for treating a disease comprising administering to a patient an effective amount of a ⁇ T cell activator in combination with an effective amount of a type I IFN.
- the present invention provides also a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN for the treatment of a disease.
- the present invention provides a method for the treatment of a disease wherein secretion of IFNy is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the present invention provides also a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein secretion of IFNy is needed
- the present invention provides a method for the treatment of a disease wherein an immune stimulation is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the present invention provides a method to strengthen the immune response in patients, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the present invention provides a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein an immune stimulation is needed
- the present invention also provides a method for the treatment of a disease wherein the stimulation of a ⁇ T cell is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the present invention also provides a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein the stimulation of a ⁇ T cell is needed
- the invention provides a method for the prevention of ⁇ T cell exhaustion comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the invention further provides a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN for the prevention of ⁇ T cell exhaustion.
- the invention provides a method for preventing or treating a chronic viral infection comprising administering to a patient an effective dose of a ⁇ T cell activator in combination with an effective dose of IFN ⁇ .
- the invention also provides a pharmaceutical composition comprising an effective amount of a ⁇ T cell activator and an effective amount of IFN ⁇ for the prevention or treatment of a chronic viral infection
- the invention provides a method for the prevention of Treg onset comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the invention also provides a method to reduce the proportion of Treg in a patient comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the invention further provides a method to obtain a sustained response of successive ⁇ T cell activator administrations in a subject comprising administering to a subject in need thereof an effective amount of a type I IFN together with a ⁇ T cell activator.
- the present invention provides a method of determining a suitable treatment, comprising: assessing a subject's response to ⁇ T cell activator, wherein a determination that a subject has a decreased response indicates that the subject will benefit from a ⁇ T cell activator and type I IFN combination treatment.
- the method comprises treating said subject with a ⁇ T cell activator and type I IFN combination treatment.
- the method according to the invention further comprises assessing a subject's response to ⁇ T cell activator by obtaining a biological sample comprising ⁇ T cells from the subject, and bringing the ⁇ T cells into contact with a ⁇ T cell activator and assessing the level of activation or proliferation of ⁇ T cells.
- the present invention also provides a method of administering a ⁇ T cell activator to an individual, comprising administering a predetermined amount of ⁇ T cell activator and type I IFN, where said predetermined amount is independent of an individual subject's ⁇ T cell responsiveness.
- the present invention also provides a method of producing a compound, comprising (a) determining whether a candidate ⁇ T cell activator has a synergistic cytokine induction or antiviral activity with a type I IFN, and (b) if said candidate ⁇ T cell activator is determined to have a synergistic cytokine induction or antiviral activity with a type I IFN, producing a quantity of said candidate ⁇ T cell activator, optionally for administration in combination with the type I IFN.
- the present invention also provides a method of potentiating cytokine release comprising administering to a patient an effective amount of a ⁇ T cell activator and an effective amount of a type I IFN.
- the present invention also provides a method of enhancing the cytokine response of a subject to a ⁇ T cell activator administration comprising further administering a type I IFN.
- the present invention also provides a method of activating ⁇ T cells in a patient being weakly responder to a ⁇ T cell treatment comprising further administering a type I IFN.
- the present invention also provides a method of clearing a chronic infection comprising administering a ⁇ T cell activator to an infected subject, together with a type I IFN.
- the present invention also provides a method to obtain a repeated cytokine secretion upon multiple ⁇ T cell activator stimulation comprising further administering a type I IFN.
- IL-2 is further administered to said patient.
- the present invention also provides a method to avoid ⁇ T cell amplification exhaustion or to obtain repeated cytokine release upon ⁇ T cell activator stimulation comprising administering to a subject in need thereof an effective amount of a ⁇ T cell activator, an effective amount of a type I IFN and an effective amount of IL-2.
- the present invention also provides a method of determining a treatment suitable for a subject, comprising: assessing the level of Treg response/level/activation in a subject wherein a determination that a subject has a increased Treg response/level/activation (e.g. compared to other subjects, compared to a predetermined level or threshold) indicates that the subject will benefit from a ⁇ T cell activator and type I IFN combination treatment.
- the subject prior to said assessment step, has been treated with IL-2 or IL-2 and a ⁇ T cell activator.
- said subject is administered a ⁇ T cell activator and type I IFN combination treatment.
- the ⁇ T cell activator is of formula I to II, more particularly, selected from the list consisting of A, B, C, D, E, F, G, and H.
- the ⁇ T cell activator is administered at a dose of about 0.1 to 30 mg/kg. In any of the embodiments listed above, the ⁇ T cell activator is administered subcutaneously. In any of the embodiments listed above, the type I IFN is IFN ⁇ . In any of the embodiments listed above, IFN ⁇ is administered at a dose comprised between 0.5 and 150 ⁇ g per week. In an aspect, IFN ⁇ dose is reduced compared to standard treatment. In any of the embodiments listed above, IFN ⁇ is administered subcutaneously.
- the disease is selected from the list consisting of a proliferative disease, an infectious disease or an autoimmune disease, in particular, the disease is a viral infection, especially HIV, HTLV, HPV, HHV.
- the viral infection is chronic; in particular, the chronic infection is HCV.
- the disease is a co infection, in particular, HCV/ AIDS or HCV/Tuberculosis.
- the disease is a cancer, in particular Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors.
- the ⁇ T cell activator administration occurs once a week for about 3 weeks.
- the IFN ⁇ administration occurs once a week for about 3 weeks.
- the method comprises further administering an antiviral agent. In any of the methods listed above, the method comprises further administering IL-2 at a low dose. DESCRIPTION OF THE FIGURES
- FIG. 1 Dose response potentiation effect of cytokine secretion (i.e. IFN ⁇ ) by CHDMAPP activated ⁇ T cells in the presence of IFN ⁇ .
- CHDMAPP dose dependant IFN ⁇ secretion by purified ⁇ T cells has been studied for various amounts of IFN ⁇ added in the culture medium (full dots: CHDMAPP alone, open squares: CHDMAPP + IFN ⁇ 2B , 10 IU, full triangles:
- CHDMAPP + IFNa 2B 100 IU, open lozenges: CHDMAPP + IFNa 2B , 1000 IU).
- the quantity of IFN ⁇ secreted points towards the potentiation of ⁇ T cell functions when cultured in the presence of IFN ⁇ .
- the CHDMAPP concentration (in nM) is represented in axis, in a log scale, the IFN ⁇ release (in IU/ml) is represented in ordinates.
- Figures 2A and 2B Dose response inhibitory effect of CHDMAPP stimulated PBMC- supernatants on HCV replicon model, in two representations.
- EC5 0 has been calculated at 1.68 nM
- ICi 0 has been calculated at 7.79 nM).
- Figures 3A and 3B Antiviral effect of supernatants of exogenous IFN ⁇ on HCV replicon model, in two representations.
- Figure 3B The IFN ⁇ concentration is represented in axis (in IU/ml, log scale), the concentration of HCV RNA copy (per ml, in millions) is represented in ordinates.
- EC 50 has been calculated at 0.217 nM, IC 90 has been calculated at 5.46 nM).
- FIGS. 4A and 4B Cytokine release profile (4A: MCP-I, IL-6, IL-l ⁇ , 4B: IFN ⁇ , TNF ⁇ , AUC (in pgxh/ml, left hand colunm) and Cmax (in pg/ml, right hand column)) in NHP during three cycles of treatment with CHDMAPP +/- IFN ⁇ .
- AUC is represented for cycle 1, cycle 2, cycle 3 and the last column represents the total of the three cycles
- Cmax is represented for cycle 1, cycle 2 and cycle 3).
- Animals have been treated for three cycles with CHDMAPP + IFN ⁇ (open squares) or with CHDMAPP only (full dots) as described in example 4.
- the cytokine release in CHDMAPP + IFN ⁇ treated animals is improved compared to CHDMAPP treated animals, thereby underlining the potentiating effect of the conjoint administration of IFN ⁇ and a ⁇ T cell activator.
- Figures 5A and 5B ⁇ T cell profile after multiple CHDMAPP activation, in the presence or absence of IFN ⁇ and/or IL-2 (5A). Treg profile after multiple CHDMAPP activation, in the presence or absence of IFN ⁇ and/or IL-2 (5B). Experiments have been conducted according to example 5. Time in days is represented in axis, absolute number of ⁇ T cells per ⁇ l of blood in ordinates.
- Figure 5A Despite multiple CHDMAPP treatment, the ⁇ T cell show an enhanced amplification in CHDMAPP+IFN ⁇ +IL-2 treated NHP (full square line) than in CHDMAPP-IL-2 treated NHP (dark grey and open dots line) or in IFN ⁇ alone treated NHP (grey triangles line).
- FIG. 6 IFN ⁇ release profile after multiple CHDMAPP activation, in the presence or absence of IFN ⁇ and/or IL-2.
- the sampling time is represented in axis (day, hour), the IFN ⁇ concentration (in pg/ml) in represented in ordinates.
- Multiple stimulation with CHDMAPP, IFN ⁇ and IL-2 full square line
- leads to an enhanced cytokine secretion here IFN ⁇
- CHDMAPP and IL-2 only treated NHP dark grey and open dots line
- a single treatment with IFN ⁇ grey triangles line
- This experiment underlines the long term potentiation of IFN ⁇ secretion by ⁇ T cells stimulated by CHDMAPP, IL-2 and IFN ⁇ in an in vivo model.
- FIG. 7 HCV RNA replication inhibition by sera of CHDMAPP treated NHP (non human primates), as described in example 3. HCV RNA copies (in million per ml) is represented in ordinates. Sera of 2 NHP (NHPl: dashed columns, NHP2: black columns) have been withdrawn before CHDMAPP administration (predose), and a few hours (e.g. 4-5 hours) after CHDMAPP administration and diluted (1/10, 1/20 and 1/30), collected sera have been tested in the replicon model described in example 3. Whereas untreated serum induces no viral inhibition (predose), serum collected after CHDMAPP administration induces a strong viral replication inhibition (dilution 1/10,
- Ribavirin 200 ⁇ M is provided as control.
- the level of inhibition between the two NHP is different, underlining that the inhibitory effect on viral replication of CHDMAPP is not only IFN ⁇ concentration dependent.
- FIG. 8 Intracellular staining of IFN ⁇ and TNF ⁇ of PBMC in different activation conditions.
- Cells have been stimulated in vitro according to example 6.
- Each dot plot represents a different activation conditions: 8A: medium, percentage of ⁇ T cells expressing IFN ⁇ and V ⁇ 2 chain (IFNy + V ⁇ 2 + cells): 0.4%; 8B: CHDMAPP, % IFNy + V ⁇ 2 + cells: 14.9%; 8C: IFN ⁇ , % IFNy + V ⁇ 2 + cells: 0.5%; 8D: CHDMAPP and IFN ⁇ , % IFNy + V ⁇ 2 + cells: 34.0%), IFN ⁇ is represented in ordinates, V ⁇ 2 in axis.
- FIG. 9 Crossed dose response IFN ⁇ /CHDMAPP on PBMCs: potentiation effect of cytokine secretion (i.e. IFN ⁇ ) by CHDMAPP activated ⁇ T cells in the presence of IFN ⁇ .
- CHDMAPP dependant IFN ⁇ secretion by purified ⁇ T cells was studied for various amounts of IFN ⁇ and CHDMAPP added in the culture medium.
- the quantity of IFN ⁇ secreted points towards the potentiation of ⁇ T cell functions when cultured in the presence of IFN ⁇ .
- Figure 9 A The histogram represents all the IFN ⁇ secretion registered for each CHDMAPP/IFN ⁇ dose couple experimented.
- Figure 9B is a three dimensional representation of the synergic effect between CHDMAPP and IFN ⁇
- FIG. 10 Cytokine release profile in NHP after multiple CHDMAPP activation, in the presence or absence of IFN ⁇ , for IFN ⁇ (10A) and MCP-I (10B). Experiments was conducted according to example 8. Time is represented in hours in axis. Cytokine release is represented in pg/ml in ordinates. In the absence of IFN ⁇ , the cytokine release gradually decreases over multiple
- CHDMAPP administrations (light grey; dots), on the contrary, the cytokine secretion level is maintained in the CHDMAPP+INF ⁇ group (dark grey; triangles).
- “Weekly” stands for “about once a week” (meaning that more than one treatment is made with an interval of about one week between treatments), the about here preferably meaning +/-1 day (that is, translating into “every 6 to 8 days”); most preferably, “weekly” stands for "once every 7 days”.
- the term “about” or “approximately” usually means within 20%, more preferably within
- the term “about” means within about a log (i.e., an order of magnitude) preferably within a factor of two of a given value.
- the expressions "regulating the activity of ⁇ T cells”, “stimulating the activity of ⁇ T cells”, “activating ⁇ T cells” and “regulating the activity of ⁇ T cells” designate causing or favoring an increase in the number and/or biological activity of such cells in a subject.
- Stimulating and regulating thus each include without limitation modulating (e.g., stimulating) expansion of such cells in a subject and/or, for instance, triggering of cytokine secretion (e.g., TNF ⁇ or IFN ⁇ ).
- ⁇ T cells normally represent between about 1-10% of total circulating lymphocytes in a healthy adult human subject.
- the present invention can be used to significantly increase the ⁇ T cells population in a subject, particularly to reach at least 30% of total circulating lymphocytes, typically 40%, more preferably at least 50% or 60%, or from 50% to 90%.
- Regulating also includes, in addition or in the alternative, modulating the biological activity of ⁇ T cells in a subject, particularly their cytolytic activity or their cytokine-secretion activity.
- the invention defines novel conditions and strategies for increasing the biological activity of ⁇ T cells towards target cells.
- the terms “conjoint”, “in combination” or “combination therapy”, used interchangeably, refer to the situation where two or more therapeutic agents affect the treatment or prevention of the same disease.
- the use of the terms “conjoint”, “in combination” or “combination therapy” does not restrict the order in which therapies (e.g., prophylactic or therapeutic agents) are administered to a subject with the disease.
- a first therapy can be administered prior to (e.g., 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks before), concomitantly with, or subsequent to (e.g., 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks after) the administration of a second therapy to a subject with a disease.
- the term “concomitantly” refers to the situation where two compounds are administered at the same time or within a few minutes i.e. one directly after the other.
- the term “conjointly” refer to the situation where two compounds are administered during the same day within 1, 2, 3, 4, 5, 6, 7, 8, 12, 18, 24 hours.
- the term “sequentially” refer to the situation where two compounds are administered within 1, 2, 3, 4, 6, 7 or more days, 1, 2, 3 or more weeks.
- the term "separately administered” indicates that the active ingredients are administered at a different site or through a different route or through a different schedule to the subject. Accordingly, the ingredients are generally not mixed together prior to administration, although they may be combined in a unique package in suitable separated containers.
- the term "Sustained virologic response" or "SVR” means that the virus (the viral load) remains undetectable in the blood for a long time, i.e. for at least six months after finishing the treatment.
- the viral load is measure by quantitative PCR (Polymerase Chain Reaction) from a biopsy (i.e. a blood extract) of an infected patient.
- the present invention is useful for the treatment of infectious disease.
- the invention is useful to treat chronic viral diseases, such a chronic HCV.
- the aim of a viral treatment can, for instance, be to obtain a satisfying SVR (sustained virologic response), which means that the virus cannot be detected in the patient's blood for a time length, i.e. at least 6 months.
- the term "subject's response” refers to the ability of a subject to amplify ⁇ T cells or release cytokines in response to stimulation i.e. by a ⁇ T cell activator or by a cytokine, in particular a type I IFN.
- the terms "weak response”, “weakly responding”, “decreased response” refers to an impaired ⁇ T cell response or an impaired cytokine release compared to standard response in other subject.
- a weak response or decrease response is for example 10%, 20%, 30%, 40%, 50% or more decreased compared to what is usually assessed in patients. For example, if a cytokine release is usually of lOOpg/ml upon a given dose of ⁇ T cell activator, a weak response would be a 60pg/ml of the same cytokine at the same ⁇ T cell activator dose. Ultimately, a 90% or 100% decreased response will be qualified as a non response, and the subject is nonresponder.
- the term “strengthen the immune response” refers to the improvement of the ⁇ T cell amplification or the cytokine release of a subject that usually present a weak or decreased response.
- the term “potentiate” or “potentiating” refers to the ability of a treatment, compound, method, to allow an enhanced results compared to what was usually obtained or expected based on previous experiments. For instance, a cytokine release is potentiated when one obtains a 10%, 20%, 30%, 40% or more increase in cytokine release compared to the cytokine level that was commonly observed before the present invention was made.
- the term “synergic”, “synergistically”, refers to the action of a combination wherein this action is superior to the mere addition of each effect detected separately.
- cancers can be treated using the methods and compositions according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFN ⁇ may be beneficial.
- the cancer to be treated is selected from melanoma, renal cell carcinoma, kidney cancer, NHL (Non Hodgkin Lymphoma), myeloma, CML (Chronic Myeloid Leukemia), hairy cell leukemia, cutaneous T cell leukemia, follicular lymphoma, squamous cell carcinoma, prostate carcinoma, bladder cell carcinoma, cervical dysplasia and colon carcinoma. Infectious diseases
- infectious diseases can be treated using the methods and compositions according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFN ⁇ , may be beneficial.
- the viral disease to be treated is selected from HCV (Hepatitis C virus), HBV (Hepatitis B virus), HIV (Human Immunodeficiency Virus), HPV (Human Papiloma Virus), HTLV-I (Human T cell lymphoma virus type 1), HHV (Human
- Herpes Virus Herpes Virus
- HHV8 condyloma acuminatum
- cytomegalovirus Epstein-Barr virus
- Kaposi syndrome Herpes Virus
- Hepatitis C is a blood- borne infectious disease that is caused by the Hepatitis C virus (HCV), affecting the liver.
- the Hepatitis C virus (HCV) is a small, enveloped, single-stranded, positive sense RNA virus.
- the hepatitis C virus (HCV) is spread by blood-to-blood contact.
- the symptoms of infection can be medically managed, and a proportion of patients can be cleared of the virus by a course of anti-viral medicines.
- An estimated 150-200 million people worldwide are infected with hepatitis C.
- the infection is often asymptomatic, people with HCV infection can experience mild symptoms, and consequently do not seek treatment.
- the virus is detectable in the blood within 1 to 3 weeks after infection, and antibodies are detectable within 3 to 12 weeks.
- 20-30% infected patients clear the virus during the acute phase, the other 70 to 80% patients develop chronic HCV e.g., an infection lasting more than 6 months.
- chronic infection can cause inflammation of the liver (chronic hepatitis). This condition can progress to scarring of the liver (fibrosis), and advanced scarring (cirrhosis). In some cases, those with cirrhosis will go on to develop liver failure or other complications of cirrhosis, including liver cancer.
- the object of the present invention is to provide a scheme of treatment that leads to the reduction of the residual disease in chronically HCV infected patients.
- the present invention provides a method of treatment, comprising: determining whether a subject has responded to a treatment comprising administration of type I IFN, and if said patient has demonstrated a decreased response or is non responder to IFN treatment, administering to the subject a ⁇ T cell activator and type I IFN combination treatment.
- Hepatitis B is a virus that infects the liver of hominoidae, including humans, and causes an inflammation called hepatitis. It is a DNA virus and one of many unrelated viruses that cause viral hepatitis. Symptoms of the acute illness caused by the virus include liver inflammation, vomiting, jaundice, and rarely, death.
- Hepatitis B virus infection may either be acute (self-limiting) or chronic (long-standing). Persons with self-limiting infection clear the infection spontaneously within weeks to months. Children are less likely than adults to clear the infection. More than 95% of people who become infected as adults or older children will stage a full recovery and develop protective immunity to the virus. However, only 5% of newborns that acquire the infection from their mother at birth will clear the infection. Of those infected between the age of one to six, 70% will clear the infection.
- Acute infection with hepatitis B virus is associated with acute viral hepatitis - an illness that begins with general ill-health, loss of appetite, nausea, vomiting, body aches, mild fever, dark urine, and then progresses to development of jaundice.
- Chronic infection with Hepatitis B virus may be either asymptomatic or may be associated with a chronic inflammation of the liver (chronic hepatitis), leading to cirrhosis over a period of several years. This type of infection dramatically increases the incidence of hepatocellular carcinoma (liver cancer).
- Autoimmune diseases Auto immune disease can also be treated using the methods and composition according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFN ⁇ may be beneficial. Such disease is for instance multiple sclerosis.
- Multiple sclerosis is an autoimmune condition in which the immune system attacks the central nervous system, leading to demyelination. Disease onset usually occurs in young adults, and it is more common in women. Common multiple sclerosis treatment is weekly intramuscular injection of interferon ⁇ -la (Avonex®, Rebif®) and one of interferon ⁇ -lb (Betaseron® or Betaferon®). Those medications are modestly effective at decreasing the number of attacks and slowing progression to disability, although they differ in their efficacy rate and studies of their long-term effects are still lacking. The present invention is also suited for the treatment of any other autoimmune disease where the administration of a type I IFN may be beneficial.
- ⁇ T cell activator designates a molecule, preferably artificially produced, which can activate ⁇ T lymphocytes. It is more preferably a ligand of the T receptor of ⁇ T lymphocytes.
- the ⁇ T cell activators may by of various natures, such as a peptide, lipid, small molecule, etc. It may be a purified or otherwise artificially produced (e.g., by chemical synthesis, or by microbiological process) endogenous ligand, or a fragment or derivative thereof, or an antibody having substantially the same antigenic specificity.
- a phosphoantigen that is a ⁇ T cell activator preferably increases the biological activity or causes the proliferation of ⁇ T cells, preferably increasing the activation of ⁇ T cells, particularly increasing cytokine secretion from ⁇ T cells or increasing the cytolytic activity of ⁇ T cells, with or without also stimulating the proliferation or expansion of ⁇ T cells.
- the ⁇ T cell activator is administered in an amount and under conditions sufficient to increase the activity ⁇ T cells in a subject, preferably in an amount and under conditions sufficient to increase cytokine secretion by ⁇ T cells and/or to increase the cytolytic activity of ⁇ T cells. Cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay (as described hereunder).
- cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay, or those provided in the examples herein.
- cytokine secretion can be determined according to the methods described in Espinosa et al. (/. Biol. Chem., 2001, Vol. 276, Issue 21, 18337-18344), describing measurement of TNF- ⁇ release in a bioassay using TNF ⁇ sensitive cells. Briefly, 10 4 ⁇ T cells/well were incubated with stimulus plus 25 units of IL2/well in 100 ⁇ l of culture medium during 24 h at 37 0 C.
- cytolytic activity is a 51 Cr release assay.
- the cytolytic activity of ⁇ T cells is measured against autologous normal and tumor target cell lines, or control sensitive target cell lines such as Daudi and control resistant target cell line such as Raji in 4h 51 Cr release assay.
- target cells were used in amounts of 2xlO 3 cells/well and labeled with lOO ⁇ Ci 51 Cr for 60 minutes. Effector/Target (E/T) ratio ranged from 30: 1 to 3.75: 1. Specific lysis (expressed as percentage) is calculated using the standard Formula
- phosphoantigen designates a T lymphocyte agonist, more particularly a T ⁇ lymphocyte agonist, whose activity depends on the presence of a phosphate moiety. It has been formerly described (see Espinosa et al, Microbes and Infections 2001, or Belmant et al, Drug discovery today 2005) that such compounds have a unique specificity to trigger a ⁇ T cell response.
- ⁇ T cell activator that is capable of stimulating ⁇ T cell activity.
- This stimulation can be by direct effect on ⁇ T cells as discussed below using compounds that can stimulate ⁇ T cells in a pure ⁇ T cell culture, or the stimulation can be by an indirect mechanism, such as treatment with pharmacological agents such as bisphosphonates which lead to IPP accumulation.
- a ⁇ T cell activator is a compound capable of regulating the activity of a ⁇ T cells in a population of ⁇ T cell clones in culture.
- the ⁇ T cell activator is capable of regulating the activity of a ⁇ T cell population of ⁇ T cell clones at millimolar concentration, preferably when the ⁇ T cell activator is present in culture at a concentration of less than 100 inM.
- a ⁇ T cell activator is capable of regulating the activity of a ⁇ T cell in a population of ⁇ T cell clones at millimolar concentration, preferably when the ⁇ T cell activator is present in culture at a concentration of less than 10 inM, or more preferably less than 1 mM.
- Regulating the activity of a ⁇ T cell can be assessed by any suitable means, preferably by assessing cytokine secretion, most preferably TNF-OC secretion as described herein.
- the ⁇ T cell activator is capable of causing at least a 20%, 50% or greater increase in the number of ⁇ T cells in culture, or more preferably at least a 2-fold increase in the number of ⁇ T cells in culture.
- the ⁇ T cell activator may be a chemical compound capable of selectively activating V ⁇ 9V ⁇ 2 T lymphocytes.
- Selective activation of V ⁇ 9V ⁇ 2 T lymphocytes indicates that the compound has a selective action towards specific cell populations, preferably increasing activation of V ⁇ 9V ⁇ 2 T cells at a greater rate or to a greater degree than other T cell types such as V ⁇ l T cells, or not substantially not activation other T cell types.
- selectivity can be assessed in vitro T cell activation assays.
- selectivity suggests that preferred compounds can cause a selective or targeted activation of the proliferation or biological activity of V ⁇ 9V ⁇ 2 T lymphocytes.
- ⁇ T cell proliferation can be detected by standard methods.
- One specific method for detecting ⁇ T cell amplification in vivo is described in Example 1.
- the ⁇ T cell activator may increase the biological activity of a ⁇ T cell, preferably increasing the activation of a ⁇ T cell, particularly increasing cytokine secretion from a ⁇ T cell or increasing the cytolytic activity of a cytotoxic ⁇ T cell, and/or stimulating the proliferation of a ⁇ T cell.
- Preferred ⁇ T cell activators include a composition comprising a compound of the Formula I, especially a ⁇ T cell activator according to Formula I, especially ⁇ T cell activator selected from the group consisting of BrHPP, CBrHPP, EpoxPP, HDMAPP, CHDMAPP,
- the ⁇ T cell activator is selected from the list consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP.
- a number of phosphoantigen compounds that are less potent ⁇ T cell activators are available and may be used in accordance with the invention.
- a bisphosphonate compounds such as pamidronate (Novartis, Nuernberg, Germany) or zoledronate may be used.
- ⁇ T cell activators for use in the present invention are phosphoantigens disclosed in WO 95/20673, isopentenyl pyrophosphate (IPP) (US 5,639,653), the disclosures of the two preceding documents being incorporated herein by reference, as well as alkylamines (such as ethylamine, iso-propyulamine, n-propylamine, n-butylamine and iso-butylamine, for instance).
- Isobutyl amine and 3-aminopropyl phosphonic acid are obtained from Aldrich (Chicago, IL).
- the ⁇ T cell activator is administered in an amount and under conditions sufficient to increase the activity ⁇ T cells in a subject, preferably in an amount and under conditions sufficient to increase cytokine secretion by ⁇ T cells and/or to increase the cytolytic activity of ⁇ T cells.
- a ⁇ T cell activator allows the cytokine secretion by ⁇ T cells to be increased at least 2, 3, 4, 10, 50, 100-fold, as determined in vitro.
- a phosphoantigen according to the present invention comprises a compound of Formula (I): Formula (I) wherein:
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including proton); • m is an integer from 1 to 3;
- B is O, NH, or any group capable to be hydrolyzed
- Y represents O " Cat + , a Ci-C 3 alkyl group, a group -A-R, or a radical selected from the group consisting of a nucleoside, an oligonucleotide, a nucleic acid, an amino acid, a peptide, a protein, a monosaccharide, an oligosaccharide, a polysaccharide, a fatty acid, a simple lipid, a complex lipid, a folic acid, a tetrahydrofolic acid, a phosphoric acid, an inositol, a vitamin, a co-enzyme, a flavonoid, an aldehyde, an epoxyde and a halohydrin;
- A is O, NH, CHF, CF 2 or CH 2 ;
- R is a linear, branched, or cyclic, aromatic or not, saturated or unsaturated, Ci-C 20 hydrocarbon group, optionally interrupted by at least one heteroatom, wherein said hydrocarbon group comprises an alkyl, an alkylenyl, or an alkynyl, preferably an alkyl or an alkylene, which can be substituted by one or several substituents selected from the group consisting of: an alkyl, an alkylenyl, an alkynyl, an epoxyalkyl, an aryl, an heterocycle, an alkoxy, an acyl, an alcohol, a carboxylic group (-C00H), an ester, an amine, an amino group (-NH 2 ), an amide (-CONH 2 ), an imine, a nitrile, an hydroxyl (-OH), a aldehyde group (- CHO), an halogen, an halogenoalkyl, a thiol (-SH), a thioalkyl
- the substituents are selected from the group consisting of: an (Ci-C 6 )alkyl, an (C 2 - Ce)alkylenyl, an (C 2 -Ce)alkynyl, an (C 2 -Ce)epoxyalkyl, an aryl, an heterocycle, an (Ci-C 6 )alkoxy, an (C 2 -Ce)acyl, an (Ci-C 6 )alcohol, a carboxylic group (-C00H), an (C 2 -Ce)ester, an (Ci-C 6 )amine, an amino group (-NH 2 ), an amide (-CONH 2 ), an (Ci-C 6 )imine, a nitrile, an hydroxyl (-OH), an aldehyde group (-CH0), an halogen, an (d-C 6 )halogenoalkyl, a thiol (-SH), a (d-C 6 )
- the substituents are selected from the group consisting of: an (Ci-C 6 )alkyl, an (C 2 -C 6 )epoxyalkyl, an (C 2 -C 6 )alkylenyl, an (d-C 6 )alkoxy, an (C 2 -C 6 )acyl, an (d-C 6 )alcohol, an (C 2 -C 6 )ester, an (Ci-C 6 )amine, an (Ci-C 6 )imine, an hydroxyl, a aldehyde group, an halogen, an (Ci- C 6 )halogenoalkyl and a combination thereof. Still more preferably, the substituents are selected from the group consisting of: an (C 3 -
- R is a (C 3 -C 25 )hydrocarbon group, more preferably a (C 5 -Ci 0 )hydrocarbon group.
- alkyl more specifically means a group such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl, eicosyl, heneicosyl, docosyl and the other isomeric forms thereof.
- (Ci-C 6 )alkyl more specifically means methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl and the other isomeric forms thereof.
- (Ci-C 3 )alkyl more specifically means methyl, ethyl, propyl, or isopropyl.
- alkenyl refers to an alkyl group defined hereinabove having at least one unsaturated ethylene bond and the term “alkynyl” refers to an alkyl group defined hereinabove having at least one unsaturated acetylene bond.
- (C 2 -Ce)alkylene includes a ethenyl, a propenyl (1-propenyl or 2-propenyl), a 1- or 2-methylpropenyl, a butenyl (1-butenyl, 2-butenyl, or 3-butenyl), a methylbutenyl, a 2-ethylpropenyl, a pentenyl (1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl), an hexenyl (1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl), and the other isomeric forms thereof.
- (C 2 -Ce)alkynyl includes ethynyl, 1-propynyl, 2-propynyl, 1-butynyl, 2-butynyl, 3-butynyl, 1- pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, or 5- hexynyl and the other isomeric forms thereof.
- epoxyalkyl refers to an alkyl group defined hereinabove having an epoxide group. More particularly, (C 2 -C 6 )epoxyalkyl includes epoxyethyl, epoxypropyl, epoxybutyl, epoxypentyl, epoxyhexyl and the other isomeric forms thereof. (C 2 -C 3 )epoxyalkyl includes epoxyethyl and epoxypropyl.
- aryl groups are mono-, bi- or tri-cyclic aromatic hydrocarbons having from 6 to 18 carbon atoms. Examples include a phenyl, ⁇ -naphthyl, ⁇ -naphthyl or anthracenyl group, in particular.
- Heterocycle groups are groups containing 5 to 8 rings comprising one or more heteroatoms, preferably 1 to 5 endocyclic heteroatoms. They may be mono-, bi- or tri-cyclic. They may be aromatic or not. Preferably, and more specifically for R 5 , they are aromatic heterocycles.
- aromatic heterocycles include pyridine, pyridazine, pyrimidine, pyrazine, furan, thiophene, pyrrole, oxazole, thiazole, isothiazole, imidazole, pyrazole, oxadiazole, triazole, thiadiazole and triazine groups.
- bicycles include in particular quinoline, isoquinoline and quinazoline groups (for two 6- membered rings) and indole, benzimidazole, benzoxazole, benzothiazole and indazole (for a 6- membered ring and a 5-membered ring).
- Nonaromatic heterocycles comprise in particular piperazine, piperidine, etc.
- Alkoxy groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -O- (ether) bond.
- (Ci-C 6 )alkoxy includes methoxy, ethoxy, propyloxy, butyloxy, pentyloxy, hexyloxy and the other isomeric forms thereof.
- (Ci-C 3 )alkoxy includes methoxy, ethoxy, propyloxy, and isopropyloxy.
- Alcyl groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -CO- (carbonyl) group.
- (C 2 -C 6 )acyl includes acetyl, propylacyl, butylacyl, pentylacyl, hexylacyl and the other isomeric forms thereof.
- (C 2 -C 3 )acyl includes acetyl, propylacyl and isopropylacyl.
- Alcohol correspond to the alkyl groups defined hereinabove containing at least one hydroxyl group. Alcohol can be primary, secondary or tertiary.
- (Ci-C 6 )alcohol includes methanol, ethanol, propanol, butanol, pentanol, hexanol and the other isomeric forms thereof.
- (Ci -C 3 ) alcohol includes methanol, ethanol, propanol and isopropanol.
- Ester groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -COO- (ester) bond.
- C 2 -C 6 )ester includes methylester, ethylester, propylester, butylester, pentylester and the other isomeric forms thereof.
- C 2 -C 3 )ester includes methylester and ethylester.
- “Amine” groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -N- (amine) bond.
- (Ci-C 6 )amine includes methylamine, ethylamine, propylamine, butylamine, pentylamine, hexylamine and the other isomeric forms thereof.
- (Ci-C 3 )amine includes methylamine, ethylamine, and propylamine.
- (Ci-C 6 )imine includes methylimine, ethylimine, propylimine, butylimine, pentylimine, hexylimine and the other isomeric forms thereof.
- (Ci-C 3 )imine includes methylimine, ethylimine, and propylimine.
- the halogen can be Cl, Br, I, or F, more preferably Br or F.
- Halogenoalkyl groups correspond to the alkyl groups defined hereinabove having at least one halogen.
- the groups can be monohalogenated or polyhalogenated containing the same or different halogen atoms.
- the group can be a trifluoroalkyl (CF 3 -R).
- (Ci-C 6 )halogenoalkyl includes halogenomethyl, halogenoethyl, halogenopropyl, halogenobutyl, halogenopentyl, halogenohexyl and the other isomeric forms thereof.
- Ci-C 3 )halogenoalkyl includes halogenomethyl, halogenoethyl, and halogenopropyl.
- Thioalkyl groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -S- (thioether) bond.
- (Ci-C 6 )thioalkyl includes thiomethyl, thioethyl, thiopropyl, thiobutyl, thiopentyl, thiohexyl and the other isomeric forms thereof.
- (Ci-C 3 )thioalkyl includes thiomethyl, thioethyl, and thiopropyl.
- Sulfone correspond to the alkyl groups defined hereinabove bonded to the molecule by an -SOO- (sulfone) bond.
- (C i-C ⁇ ) sulfone includes methylsulfone, ethylsulfone, propylsulfone, butylsulfone, pentylsulfone, hexylsulfone and the other isomeric forms thereof.
- (Ci -C 3 ) sulfone includes methylsulfone, ethylsulfone and propylsulfone.
- "Sulfoxyde” groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -SO- (sulfoxide) group.
- (Ci -C 6 ) sulfoxide includes methylsulf oxide, ethylsulf oxide, propylsulfoxide, butylsulfoxide, pentylsulfoxide, hexylsulfoxide and the other isomeric forms thereof.
- (Ci -C 3 )sulf oxide includes methylsulf oxide, ethylsulf oxide, propylsulfoxide and isopropylsulf oxide.
- Heteroatom denotes N, S, or O.
- Nucleoside refers to a compound composed of any pentose or modified pentose moiety attached to a specific position of a heterocycle or to the natural positions of a purine (9-position) or pyrimidine (1 -position) or to the equivalent position in an analog.
- the term nucleoside includes but is not limited to adenosine, thymine, uridine, cytidine and guanosine.
- the hydrocarbon group is a cycloalkylenyl such as a cyclopentadiene or a phenyl, or an heterocycle such as a furan, a pyrrole, a thiophene, a thiazole, an imidazole, a triazole, a pyridine, a pyrimidine, a pyrane, or a pyrazine.
- the cycloalkylenyl or the heterocycle is selected from the group consisting of a cyclopentadiene, a pyrrole or an imidazole.
- the cycloalkylenyl or the heterocycle is substituted by an alcohol.
- said alcohol is a (Ci-C 3 )alcohol.
- the hydrocarbon group is an alkylenyl with one or several double bonds.
- the alkylenyl group has one double bond.
- the alkylenyl group is a (C 3 - Cio)alkylenyl group, more preferably a (C 4 -C 7 )alkylenyl group.
- said alkylenyl group is substituted by at least one functional group.
- the functional group is selected from the group consisting of an hydroxy, an (Ci-C 3 )alkoxy, an aldehyde, an (C 2 -C 3 )acyl, or an (C 2 -C 3 )ester.
- the hydrocarbon group is butenyl substituted by a group -CH 2 OH.
- said alkenyl group can be the isoform trans (E) or cis (Z), more preferably a trans isoform (E).
- the alkylenyl group is the (E)-4-hydroxy-3-methyl-2 -butenyl.
- the alkylenyl group is an isopentenyl, a dimethylallyl or an hydroxydimethylallyl.
- the hydrocarbon group is an alkyl group substituted by an acyl.
- the hydrocarbon group is an (C 4 -C 7 )alkyl group substituted by an (Ci-C 3 )acyl.
- the phosphoantigen is of Formula (Ia):
- R 4 is an halogenated (Ci-C 3 )alkyl, a (Ci-C 3 )alkoxy-(Ci-C 3 )alkyl, an halogenated (C 2 - C 3 )acyl or a (C 1 -C 3 )alkoxy-(C 2 -C 3 )acyl,
- R 3 is (d-C 3 )alkyl group, • m is an integer from 1 to 3,
- n is an integer from 2 to 20
- B represents O, NH, or any group capable to be hydrolyzed
- A represents O, NH, CHF, CF 2 or CH 2 ,
- Y represents O " Cat + , a Ci-C 3 alkyl group, a group -A-R, or a radical selected from the group consisting of a nucleoside, an oligonucleotide, a nucleic acid, an amino acid, a peptide, a protein, a monosaccharide, an oligosaccharide, a polysaccharide, a fatty acid, a simple lipid, a complex lipid, a folic acid, a tetrahydrofolic acid, a phosphoric acid, an inositol, a vitamin, a co-enzyme, a flavonoid, an aldehyde, an epoxyde and a halohydrin;
- R is a linear, branched, or cyclic, aromatic or not, saturated or unsaturated, Ci-C 50 hydrocarbon group, optionally interrupted by at least one heteroatom, wherein said hydrocarbon group comprises an alkyl, an alkylenyl, or an alkynyl, preferably an alkyl or an alkylene, which can be substituted by one or several substituents selected from the group consisting of: an alkyl, an alkylenyl, an alkynyl, an epoxyalkyl, an aryl, an heterocycle, an alkoxy, an acyl, an alcohol, a carboxylic group (-C00H), an ester, an amine, an amino group (-NH 2 ), an amide (-CONH 2 ), an imine, a nitrile, an hydroxyl (-OH), a aldehyde group
- said phosphoantigen compound are ⁇ T cell activators, and
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including the proton).
- R 4 is an halogenated methyl (-CH 2 -X, X being an halogen), an halogenated (C 2 - C 3 )acetyl, or (Ci -C 3 ) alkoxy- acetyl.
- the halogenated methyl or acetyl can be mono-, di-, or tri- halogenated. More preferably, R 4 is a CH 2 -X group, X represents a halogen atom.
- X is selected from I, Br and Cl.
- R 3 is a methyl or ethyl group. More preferably, R 3 is a methyl.
- B is O and A is O or CH 2 .
- n is an integer from 2 to 10, or from 2 to 5. In a more preferred embodiment, n is 2.
- m is 1 or 2. More preferably, m is 1.
- Y is O " Cat + , or a nucleoside. More preferably, Y is O " Cat + .
- n is 2, R 3 is a methyl and R 4 is a halogenated methyl, more preferably a monohalogenated methyl, still more preferably a bromide methyl.
- n is 2, R 3 is a methyl, R 4 is a methyl bromide.
- R is 3-(bromomethyl)-3-butanol-l-yl.
- R 4 is a CH 2 -X group and A and B represent O.
- R 3 , X, n, m, Y and Cat + have the aforementioned meaning.
- R 3 is a methyl.
- n is 2.
- X is a bromide.
- R 4 is a CH 2 -X group and B represents O and A represents CH 2 .
- R 3 , X, n, m, Y and Cat + have the aforementioned meaning.
- R 3 is a methyl.
- n is 2.
- X is a bromide.
- a phosphoantigen comprises a compound of Formula (Ia3):
- X is an halogen (preferably selected from I, Br and Cl)
- R 3 is a methyl or ethyl group
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including the proton)
- n is an integer from 2 to 20.
- R 3 is a methyl.
- n is 2.
- X is a bromide.
- a phosphoantigen comprises a compound of Formula (A): OH O O
- x Cat + is 1 or 2 Na + .
- a phosphoantigen comprises a compound of Formula
- x Cat + is 1 or 2 Na + .
- the phosphoantigen is of Formula (Ib):
- n is an integer from 2 to 20
- m is an integer from 1 to 3
- R 5 is a methyl or ethyl group
- B represents O, NH, or any group capable to be hydrolyzed
- A represents O, NH, CHF, CF 2 or CH 2 ,
- Y is O " Cat + , a nucleoside, or a radical -A-R, wherein R has the aforementioned meaning
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including the proton).
- n is an integer from 2 to 10, or from 2 to 5. In a more preferred embodiment, n is 2.
- R 5 is a methyl.
- Y is O " Cat + , or a nucleoside. More preferably, Y is O " Cat + .
- A is O, NH or CH 2 . More preferably, B is O.
- B is O.
- m is 1 or 2. More preferably, m is 1.
- a phosphoantigen may comprise a compound of Formula (IbI) or (Ib2):
- a phosphoantigen comprises a compound of Formula (Ib3):
- R 5 is a methyl or ethyl group
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including the proton)
- n is an integer from 2 to 20.
- R 5 is a methyl.
- n is 2.
- a phosphoantigen comprises a compound of Formula (C):
- x Cat+ is 1 or 2 Na + .
- the phosphoantigen is of Formula (Ic):
- R 6 , R 7 , and R 8 represent, independently from each other, a hydrogen atom or a (Ci-C 3 )alkyl group,
- R 9 is an (C 2 -C 3 )acyl, an aldehyde, an (Ci-C 3 )alcohol, or an (C 2 -C 3 )ester,
- W is -CH-, -N- or -C-R 10 ,
- A is O, NH, CHF, CF 2 or CH 2 ,
- B represents O, NH, or any group capable to be hydrolyzed
- m is an integer from 1 to 3
- Y is O " Cat + , a nucleoside, or a radical -A-R, wherein R has the aforementioned meaning.
- Cat + represents one (or several, identical or different) organic or mineral cation(s) (including the proton),
- R 6 and R 8 are a methyl and R 7 is hydrogen. More preferably, R 9 is -CH 2 -OH, -CHO, -CO-CH 3 or -CO-OCH 3 .
- the double-bond between W and C is in conformation trans (E) or cis (Z). More preferably, the double-bond between W and C is in conformation trans (E).
- the group Y can permit the design of a prodrug. Therefore, Y is enzymolabile group which can be cleaved in particular regions of the subject. The group Y can also be targeting group.
- Y is O ⁇ Cat + , a group -B-R, or a radical selected from the group consisting of a nucleoside, a monosaccharide, an epoxyde and a halohydrin.
- Y is an enzymolabile group.
- Y is O ⁇ Cat + , a group -B-R, or a nucleoside.
- Y is O ⁇ Cat + .
- Y is a nucleoside.
- Cat + is H + , Na + , NH 4 + , K + , Li + , (CH 3 CH 2 ) 3 NH + .
- B is O or NH. More preferably, B is O.
- A is O, NH or CH 2 .
- n is 1 or 2. More preferably, m is 1.
- a phosphoantigen comprises a compound of Formula (IcI) or (Ic2):
- W is -CH-.
- R 6 and R 7 are hydrogen.
- R 8 is a methyl.
- R 9 is -CH 2 -OH.
- a phosphoantigen comprises a compound of Formula (D):
- a phosphoantigen comprises a compound of Formula
- a phosphoantigen comprises a compound of Formula
- phosphoantigen comprises a compound of Formula (Ic3):
- R 6 , R 7 and R 9 are hydrogen.
- Ri 0 is a methyl.
- R 8 is -CH 2 - OH.
- A is CH 2 , NH or O.
- a phosphoantigen comprises a compound named (Z)-5-hydroxy-3- methylpent-3-enyl pyrophosphonate, of Formula G:
- the ⁇ T cell activator is a compound named (E)-5-hydroxy-3- methylpent-3-enyl pyrophosphonate, of Formula H:
- compounds also include: (E)l-pyrophosphonobuta-l,3-diene; (E)I- pyrophosphonopenta-l,3-diene; (E)l-pyrophosphono-4-methylpenta-l,3-diene; (E,E)1- pyrophosphono-4,8 -dimethylnona- 1 ,3 ,7-triene; (E,E,E) 1 -pyrophosphono-4, 8 , 12-trimethyltrideca-
- the phosphoantigen can be selected from the group consisting of: 3-(halomethyl)-3-butanol-l-yl-diphosphate; 3-(halomethyl)-3-pentanol-l-yl- diphsophate; 4-(halomethyl)-4-pentanol-l -yl-diphosphate; 4-(halomethyl)-4-hexanol-l -yl- diphosphate ; 5 -(halomethyl) -5 -hexanol- 1 -yl-diphosphate ; 5 -(halomethyl) -5 -heptanol- 1 -yl- diphosphate; 6-(halomethyl)-6-heptanol-l -yl-diphosphate; 6-(halomethyl)-6-octanol-l -yl- diphosphate; 7-(halomethyl)-7-octanol-l -yl-diphosphate; 7-(halomethyl
- the phosphoantigen can be selected from the group consisting of: 3-
- the phosphoantigen can be selected from the group consisting of: 3,4-epoxy-3-methyl-l-butyl-diphosphate (Epox-PP); 3,4,-epoxy-3-methyl-l-butyl- triphosphate (Epox-PPP); ⁇ , ⁇ -di-3,4,-epoxy-3-methyl-l -butyl -triphosphate (di-Epox-TP); 3,4-epoxy- 3-ethyl-l -butyl -diphosphate; 4,5-epoxy-4-methyl- 1 -pentyl-diphosphate; 4,5-epoxy-4-ethyl- 1 -pentyl- diphosphate; 5,6-epoxy-5-methyl-l-hexyl-diphosphate; 5,6-epoxy-5-ethyl-l-hexyl -diphosphate; 6,7- epoxy-6-methyl-l-heptyl -d
- the phosphoantigen can be selected from the group consisting of: (E)-4-hydroxy-3-methyl-2-butenyl pyrophosphate (HDMAPP), (E)-5-hydroxy-4-methylpent-3-enyl pyrophosphonate (CHDMAPP) and (E)-5-hydroxy-3-methylpent-3-enyl pyrophosphonate (H- tiglylPP).
- HDMAPP high-methyl-2-butenyl pyrophosphate
- CHDMAPP -5-hydroxy-4-methylpent-3-enyl pyrophosphonate
- H- tiglylPP H- tiglylPP
- the phosphoantigen is a ⁇ T cell activator and is a compound described in any one of PCT publication nos. WO 00/12516, WO 00/12519, WO 03/050128, WO
- the phosphoantigen is a ⁇ T cell activator and is a compound selected from the group consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP.
- activators for use in the present invention are phosphoantigens disclosed in WO 95/20673, isopentenyl pyrophosphate (IPP) (U.S. Patent No. 5,639,653) and 3-methylbut-3-enyl pyrophosphonate (C-IPP).
- IPP isopentenyl pyrophosphate
- C-IPP 3-methylbut-3-enyl pyrophosphonate
- preferred ⁇ T cell activators are selected which increase the biological activity of ⁇ T cells, preferably increasing the activation or proliferation of ⁇ T cells, particularly increasing cytokine secretion from ⁇ T cells or increasing the cytolytic activity of ⁇ T cells, with or without also stimulating the expansion of ⁇ T cells.
- a ⁇ T cell activator allows the cytokine secretion by ⁇ T cells to be increased at least 2, 3, 4, 10, 50, 100-fold, as determined in vitro. Cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay, or those described herein.
- the present invention relates to methods for the treatment of a proliferative disease, an infectious disease or an autoimmune disease, where the ⁇ T cell activator, is of Formula I.
- Formula I designates all compounds derived from Formulas I: I, Ia, IaI, Ia2, Ia3, A, B, Ib, IbI, Ib2, Ib3, C, Ic, IcI, Ic2, Ic3, D, E, F, G and H.
- the ⁇ T cell activator selected from the group consisting of BrHPP, CBrHPP, EpoxPP, HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP is administered in an amount and under conditions sufficient to stimulate the expansion of the ⁇ T cell population in a subject, particularly to reach 30- 90% of total circulating lymphocytes, typically 40-90%, more preferably from 50-90%, or to induce an improved cytokine secretion, particularly to reach a 30-90%, more preferably a 50-90%, increase in cytokine release compared to the common cytokine secretion. Percentage of total circulating lymphocytes can be determined according to methods known in the art.
- a preferred method for determining the percentage of ⁇ T cells in total circulating lymphocytes is by flow cytometry, examples of appropriate protocols described in example 1.
- the present invention relates to an in vivo regimen for the treatment of proliferative disease, an infectious disease or an autoimmune disease, where a ⁇ T cell activator, especially a ⁇ T cell activator according to Formula I, especially ⁇ T cell activator selected from the group consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP, is administered to a warm- blooded animal, especially a human, in a dose that is higher (preferably at least 10%, 20%, 30% higher) than the single administration Efficient Concentration value giving half of the maximum effect (EC50) of ⁇ T cell biological activity or population expansion, or more preferably a dose that is at least 50%, or more preferably at least 60%, 75%, 85% or preferably between about 50% and 100% of the single administration Efficient Concentration value giving the maximum effect.
- EC50 maximum effect
- a compound of Formula I is administered in a dosage (single administration) between 1 ⁇ g/kg and about 100 mg/kg, more preferably between about 1 ⁇ g/kg and about 30 mg/kg.
- a compound of Formula Ia especially a compound of Formula A or B is administered in a dosage between about 1 mg/kg and about 100 mg/kg, preferably between about 10 mg/kg and about 80 mg/kg, more preferably between about 20 mg/kg and about 30 mg/kg.
- a compound of Formula Ic is administered in a dosage between about 1 ⁇ g/kg and about 100 mg/kg, preferably between about 1 ⁇ g/kg and about 20 mg/kg, more preferably between about 10 ⁇ g/kg and about 5 mg/kg.
- dosages and administration and examples of dose response experiments using ⁇ T cell activator in mice and primate models are provided in PCT Application WO 04/050 096, the disclosure of which is incorporated herein by reference.
- the dosages for administration to a warm blooded animal, particularly humans provided herein are indicated in pure form (anionic form) of the respective compound. Purity level for the active ingredient depending on the synthesis batch can be used to adjust the dosage from actual to anionic form and vice-versa.
- compounds are preferably administered in a dose sufficient to significantly increase the biological activity of ⁇ T cells or to significantly increase the ⁇ T cell population in a subject.
- Said dose is administered to the human by subcutaneous (s.c.) or intravenous (i.v.) route.
- Said dose is preferably administered to the human by subcutaneous (s.c.) route.
- said dose is preferably administered to the human by oral administration, in a solid or liquid dosage form, including but not limited to tablets, capsules, sachets, lozenges, powders, pills, or granules
- the solid dosage form can be, for example, a fast melt dosage form, chewable form, controlled release dosage form, lyophilized dosage form, delayed release dosage form, extended release dosage form, pulsatile release dosage form, mixed immediate release and controlled release dosage form, or a combination thereof.
- the method is based on the use of particular combinations of active agents, according to particular schedules.
- the invention more preferably uses a cytokine in combination with a ⁇ T cell activator, the cytokine being a type I IFN, preferably an interferon- ⁇ .
- Interferons are natural proteins produced by the cells of the immune system of most vertebrates in response to challenges by foreign agents such as viruses, parasites and tumor cells.
- Interferons belong to the large class of glycoproteins known as cytokines. Interferon- a
- Interferon alpha-2a (RoferonA®) binds to its specific cell-surface receptor, resulting in the transcription and translation of genes whose protein products have antiviral, antiproliferative, anticancer, and immune modulating effects.
- Peginterferon alpha-2a (Pegasys®, Roche), is a covalent conjugate of the protein interferon alpha-2a produced by recombinant DNA technology in E. coli with bis(monomethoxypoly ethylene glycol). The product has been approved by the FDA in October 2002.
- Interferon alpha 2b IFN ⁇ 2b (IntronA®) has a similar mode of action.
- PEG-interferon alfa- 2b (Peglntron®, FDA approved January 2001) is a covalent conjugate of recombinant interferon alpha, subtype 2b, and polyethylene glycol (PEG), used as an antiviral and antineoplastic agent.
- PEG polyethylene glycol
- the biological activity of this agent is derived from its interferon alpha-2b protein moiety.
- Interferons alpha bind to specific cell-surface receptors, leading to the transcription and translation of genes whose protein products mediate antiviral, antiproliferative, anticancer, and immune-modulating effects.
- the PEG moiety lowers the clearance of interferon alpha-2b, thereby extending the duration of its therapeutic effects, but may also reduce the interferon-mediated stimulation of an immune response.
- a standard treatment with peg IFN ⁇ 2b is from 50 to 150 ⁇ g per week.
- the present invention provides a method to treat a subject with a low dose (e.g. a dose that is at least 10%, 20%, 30%, 40%, 50% below the dose of IFN ⁇ required in absence of co-administering the ⁇ T cell activator which is between 50 and 150 ⁇ g per week).
- IFN ⁇ low dose is comprised between 0.1 and 50 ⁇ g/week, more preferably between 0.1 and 20 ⁇ g/week.
- IFN ⁇ is usually administered as a single subcutaneous injection, once a week for a period of time sufficient to obtain a response to the treatment. In most patients, the treatment lasts from 12 to 26 weeks.
- the present invention provides a method to treat a subject for a shorter time period (e.g. a time period shorter than what is usually prescribed, i.e. a time that is at least 10%, 20%, 30%, 40%, 50% or more reduced compared to the time of treatment with IFN ⁇ required in the absence of co-administering the ⁇ T cell activator).
- composition according to the invention can be carried out in a number of ways.
- the ⁇ T cell activator and type I IFN can be administered concomitantly, conjointly sequentially.
- both compounds are administered at the same time or within a few minutes, i.e. one directly after the other.
- the compounds are administered during the same day within 1, 2, 3, 4, 5, 6, 7, 8, 12, 18, 24 hours.
- the ⁇ T cell activator and type I IFN are administered sequentially, the compounds are administered within 1, 2, 3, 4, 6, 7 or more days, 1, 2, 3 or more weeks.
- the ⁇ T cell activator and the type I IFN are administered at concomitantly.
- the ⁇ T cell activator can be administered before or after the type I IFN. In an embodiment, the ⁇ T cell activator is administered after the beginning of the type I IFN treatment.
- a cycle comprises at least one administration of a ⁇ T cell activator and at least one administration of a type I IFN.
- a cycle comprises one administration of a ⁇ T cell activator and successive administrations of a type I IFN.
- each ⁇ T cell activator constitutes another treatment cycle.
- treatment cycles can be carried out in a number of ways.
- the treatment cycle comprises a single administration of a ⁇ T cell activator, and one, two, three, four or more successive administrations of the type I IFN, each type I IFN administration occurring once a week.
- the treatment cycle comprises a single administration of a ⁇ T cell activator and a single administration of a type I IFN.
- the administration of the ⁇ T cell activator can take place once (single treatment cycle) or many times (many treatment cycle).
- the administrations that give rise to a ⁇ T cell proliferation are separated by a period of time sufficient to prevent "exhaustion" of the ⁇ T cells.
- Exhaustion can be characterized by reduction in ability to produce cytokines or to proliferate in response to the ⁇ T cell activator, in comparison to that observed when the ⁇ T cells are treated with a first dose or a preceding dose of the ⁇ T cell activator.
- the ⁇ T cell rate (number of ⁇ T cells), is allowed to return to substantially basal rate prior to a second administration of the ⁇ T cell activator compound. At least about one week, two weeks, three weeks or up to eight weeks and more are required for a patient's ⁇ T cell rate to return to substantially basal rate.
- the ⁇ T cell activator is administered in multiple doses, the administration of successive doses of the ⁇ T cell activator takes place at the beginning of a treatment cycles, which are separated by at least 1, 2, 3 or 4 6, 8, 12, 24, or 36 weeks.
- one first dose of a ⁇ T cell activator is administered, and one or more (preferably at least two) further doses of ⁇ T cell activator are administered in different treatment cycles.
- compositions according to the invention may further comprise another therapeutic agent, including agents normally utilized for the particular therapeutic purpose for which the ⁇ T cell activator is being administered.
- the additional therapeutic agent will normally be present in the composition in amounts typically used for that agent in a monotherapy for the particular disease or condition being treated.
- therapeutic agents include, but are not limited to, therapeutic agents used in the treatment of cancers, infectious and autoimmune diseases.
- the invention uses further IL-2 in combination with a ⁇ T cell activator and type I IFN.
- the interleukin-2 polypeptide may be of human of animal origin, preferably of human origin. It may comprise the sequence of a wild-type human (or animal) IL-2 protein, or any biologically active fragment, variant or analogue thereof, i.e., any fragment, variant or analogue capable of binding to an IL-2 receptor and of inducing activation of ⁇ T cells in the method of this invention.
- the sequence of reference, wild-type human interleukin-2 proteins is available in the art, such as in Genbank, under accession numbers NP000577; AAK26665; PO1585; XP035511 for instance.
- the interleukin-2 is commercialized under the name ProleukinTM (aldesleukin).
- ProleukinTM which is described in its product information leaflet as follows: a human recombinant interleukin-2 product, is a highly purified protein with a molecular weight of approximately 15 300 daltons. The chemical name is des-alanyl-1, serine- 125 -human interleukin-2.
- ProleukinTM is a lymphokine, produced by recombinant DNA technology using a genetically engineered E.
- CoIi strain containing an analog to the human interleukin-2 gene Genetic engineering techniques were used to modify the human IL -2 gene, and the resulting expression clone encodes a modified human interleukin-2.
- the recombinant forms differs from native interleukin-2 in the following ways: a) ProleukinTM is not glycosylated because it is derived from E.
- ProleukinTM 1 1.1 mg protein.
- the dose of IL-2 to be administered is generally expressed per square meter.
- the body surface of a human subject lies between 1.5 to 2.1 m 2 depending on the size and weight of the subject.
- the body surface area (BSA) can be determined using different Formulas such as the Mosteller Formula or the Dubois and Dubois Formula.
- the Mosteller Formula BSA (m 2 ) ([Height(cm) x Weight(kg)]/ 3600 )'/ 2
- the DuBois and DuBois 2 Formula BSA (m 2 ) 0.20247 x Height(m) 0725 x Weight(kg) 0425
- the average body surface is of about 1.7 m 2 .
- the IL-2 polypeptide is preferably administered by injections of between 7 and 9 MIU total in human daily, over a period of 1 to 10 days.
- daily doses of 7.5 to 8.5 MIU total in human daily most preferably doses of 8 MIU total in human daily are being administered.
- the IL-2 polypeptide can be administered for example by injections of between 3.3 and 6 MIU/m 2 daily, over a period of 1 to 10 days.
- daily doses from 3.5 to 5.5 MIU/m 2 are being administered.
- the preferred dose for IL-2 expressed in international units, can be transposed to any available or commercial interleukin-2.
- the inventors have used the commercial interleukin-2 polypeptide ProleukinTM 1 commercialized by Chiron. It will be appreciated that any other interleukin-2 may be used, as long as the quantity of product to be administered is adjusted according to the dosage of the IL-2 chosen. Such dosage is easily carried on by the skilled artisan using standard analytical methods (see Hammerling et al, J. Pharm. And Biomed. Anal 1992, p. 547-553).
- IL-2 is preferably administered conjointly with the ⁇ T cell activator.
- IL-2 is administered more than once, daily, for at least 2, 3, 4, 5, 6 or more days after the first IL-2 administration.
- Dosages of IL-2 are proposed in PCT publication N° 2004/020096 incorporated by reference, especially page 30 to 33 "Co-treatment with cytokine”.
- the invention further comprises administering ribavirin with type I IFN , preferably IFN ⁇ , and the ⁇ T cell activator.
- Ribavirin Copegus; Rebetol; Ribasphere;
- Vilona,Virazole also generics from Sandoz, Teva, Warrick
- ribavirin is remarkable as a small molecule for its wide range of activity, including important activities against influenzas, flavi viruses and agents of many viral hemorrhagic fevers.
- protease inhibitors such as ITMN-191, InterMune & Roche; MK-7009, Merck; BI12202, Boehringer; TelaprevirTM, VX-950, N3S/4A inhibitor, Vertex Pharmaceuticals; Boceprevir, SCH-503034, NS3/4A inhibitor, Schering-Plough; TMC435350,
- nucleoside polymerase inhibitors such as: MK-0608, Merck ; R7128, Pharmasset & Roche ; PSI-6130, Pharmasset ; Rl 626, NS5B RNA polymerase inhibitor, Roche), cyclophilin inhibitors (such as DEBIO-025, Debiopharm; NIM-811, Novartis), nucleoside and nucleotide reverse transcriptase inhibitors (NRTI), non-nucleoside reverse transcriptase inhibitors
- nNRTI integrase inhibitors
- entry inhibitors or fusion inhibitors, i.e. maraviroc, enfuvirtide
- maturation inhibitors i.e. bevirimat, becon
- TLR7 agonists ANA 975, Anadys
- antivirals agents such as, but not limited to lamivudine, adefovir, tenofovir and entecavir, ritonavir, l'indinavir, nelfinavir, saquinavir, l'amprenavir and lopinavir can also be administered in the combination according to the present invention. Some of those compounds in development are listed in the publication Manns et al, Nature Drug Discovery 2007: 991-1000. Chemotherapeutic compounds - Anticancer agents
- the treatment comprises an additional step of administration of an anti-cancer agent.
- anti-cancer agents include, but are not limited to:
- spindle poisons such as mebendazole, colchicine
- mitotic inhibitors paclitaxel (Taxol), docetaxel (Taxotere); vinca alkaloids (i.e. vincristine, vinblastine, vinorelbine, vindesine));
- cytotoxic/antitumor antibiotics such as anthracyclines (i.e. doxorubicin, daunorubicin, adriamycine, idarubicin, epirubicin and mitoxantrone, valrubicin), streptomyces (i.e. actinomycin, bleomycin, mitomycin, plicamycin); • anti-metabolites (such as pyrimidine analogues (i.e. fluoropyrimidines analogs, 5-fluorouracil
- topoisomerase inhibitors i.e. camptothecins: irinotecan, topotecan, amsacrine, etoposide, etoposide phosphate, teniposide
- alkylating agents i.e. busulfan, cisplatin, oxaliplatin, BBR3464, carboplatin, chlorambucil, cyclophosphamide, aldophosphamide, ifosfamide, fotemustine, lomustine, streptozocin, dacarbazine, procarbazine, temozolomide, thioTEPA, Uramustine, mechlorethamine (nitrogen mustard), phosphor amide mustard, melphalan (Akeran®), temozolomide);
- alkylating agents i.e. busulfan, cisplatin, oxaliplatin, BBR3464, carboplatin, chlorambucil, cyclophosphamide, aldophosphamide, ifosfamide, fotemustine, lomustine, streptozocin, dacarbazine, procarbazine, temozolomide, thio
- DNA methyltransferase inhibitors 2'-deoxy-5-azacytidine (DAC), 5-azacytidine, 5-aza-2'- deoxycytidine, l- ⁇ -D-arabinofuranosyl-5-azacytosine, dihydro-5-azacytidine;
- DAC 2'-deoxy-5-azacytidine
- 5-azacytidine 5-aza-2'- deoxycytidine
- l- ⁇ -D-arabinofuranosyl-5-azacytosine dihydro-5-azacytidine
- vascular disrupting agents such as flavone acetic acid derivatives, 5,6-dimethylxanthenone-4- acetic acid (DMXAA) and flavone acetic acid (FAA);
- chemotherapeutic drugs such as aprepitant, bortezomib (Velcade®, Millenium Pharmaceuticals), imatinib mesylate (Gleevec®), carmustine (BCNU), lomustine (CCNU), tamoxifen, gefitinib, erlotinib, carboxyamidotriazole, efaproxiral, tirapazamine, xcytrin, thymalfasin, vinflunine.
- aprepitant bortezomib (Velcade®, Millenium Pharmaceuticals)
- imatinib mesylate Galeevec®
- carmustine BCNU
- lomustine CCNU
- tamoxifen gefitinib
- erlotinib carboxyamidotriazole
- efaproxiral efaproxiral
- tirapazamine xcytrin
- Example 1 Flow cytometry detection of ⁇ T cell amplification
- OptiLyse B reagent (Immunotech-Beckman-Coulter, Marseilles, France) according to the manufacturer's instructions.
- stained white blood cells were recovered by centrifugation and resuspended in 300 ⁇ l IX PBS + 0.2% PFA.
- Flow cytometry analysis on viable cells was performed on a FacsSCAN apparatus (Becton Dickinson) with the FacsDiva software.
- Example 2 Dose response potentiation effect of cytokine secretion (i.e. IFN ⁇ ) by CHDMAPP activated ⁇ T cells in the presence of IFN ⁇ .
- V ⁇ 9 ⁇ 2 T lymphocytes were purified and expanded according to described methods ⁇ Espinosa et al, J. Biol Chem 2001). The cells are then frozen.
- results The cells cultured with IFN ⁇ show a higher potential for IFN ⁇ secretion. The more IFN ⁇ , the higher the secretion of cytokines.
- the quantity of IFN ⁇ added in the medium does not significantly modify the EC50, which is similar for all settings, whereas the Cmax is increased, so one can assume that it is not the release potential of each individual ⁇ T cell that increases but rather that, upon IFN ⁇ stimulation, more ⁇ T cell become sensitive to CHDMAPP treatment, thereby increasing the cytokine release (i.e. here, IFN ⁇ ).
- IFN ⁇ is able to sensitize ⁇ T cells that are not or weakly responding to CHDMAPP stimulation.
- Co-treatment with a ⁇ T cell activator and a type I IFN can thus result in further activation and cytokine release by ⁇ T cells responding only weakly to a treatment.
- Example 3 Viral inhibition induced by CHDMAPP stimulated ⁇ T cells on a Replicon model.
- Huh7 cells are maintained at 37°C, 5% CO2, in DMEM supplemented with Lglutamine 2mM, non-essential amino acids, 10% Fetal Bovine Serum (FBS) and 500 ⁇ g/ml geneticin. Cells are sub-divided 1:3 or 1:4 every 2-3 days.
- FBS Fetal Bovine Serum
- Huh7 cells are collected, counted, plated in 96-well plates at 7500 cells/well in lOO ⁇ l standard maintenance medium and incubated in the conditions above.
- IFN ⁇ secretion (pg/ml) 1578 52.6 78.9 157.8
- IFN ⁇ secretion (pg/ml) 2197 73.2 109.9 219.7
- HCV RNA levels are measured by using TaqMan RT-PCR. Total cellular RNA is isolated and amplified by using ABBOTT RealTime HCV assay (Abbott mlOOOTM Automated Sample Preparation System and Abbott m2000rtTM instrument for reverse transcription, PCR amplification, and detection/quantitation, Abbott Molecular Inc., Des Plaines, IL). Results are reported in International Units/mL (IU/mL), and
- IFNy The content in IFNy has been dosed in supernatants of CHDMAPP stimulated ⁇ T cells in vitro, and values have been reported on the right axis. This IFNy axis also underlines that the production of cytokine by ⁇ T cells is essential for the replication inhibition.
- Example 4 NHP experiment on cytokine release upon repeated treatment with CHDMAPP +/-
- mice 8 naive cynomolgus macaque (M. fascicularis) (purchased from Noveprim) have been sorted in 2 groups.
- Group 1 4 animals have been treated with CHDMAPP alone, 3 mg/kg (subcutaneous s.c. injection).
- Group 2 4 animals have been treated with CHDMAPP 3 mg/kg (s.c.) and IFN ⁇ (Pegasys®), 27 ⁇ g/administration (s.c).
- Both groups are treated three times at one week interval that is to say at day 0, 7 and 14. Both compounds are administered conjointly (possibly at the same time or quickly one after the other). Blood samples have been collected at pre-dose, 30 min, 1 hour, 2, 3, 4, 5, 6, 8 and 24 hours after each administration. Serums are collected after centrifugation and cytokines are dosed. ILl- ⁇ , IL-6, MCP-I were analyzed by electrochemoluminescence (Sector 2400 apparatus), IFNy and TNF ⁇ were dosed by
- IFN ⁇ treated groups at the second cycle of treatment were treated with IFN ⁇ treated groups at the second cycle of treatment.
- Example 5 NHP experiment on ⁇ T cell and Treg population in NHP upon repeated treatment with CHDMAPP, IL-2 +/- IFN ⁇ .
- Experimental procedure 16 naive cynomolgus macaque (M. fascicularis) (purchased from Noveprim) have been sorted in 3 groups.
- Table 3 Treatment of NHP according to groups
- IFN ⁇ cytokine secretion
- CHDMAPP and IL-2 multiple stimulation with CHDMAPP, IFN ⁇ and IL-2 leads to an enhanced cytokine secretion (here IFN ⁇ ), compared to CHDMAPP and IL-2 only treated NHP, whereas a single treatment with IFN ⁇ induces no remarkable cytokine secretion.
- This experiment underlines the long term potentiation of the cytokine secretion (here IFN ⁇ ) by ⁇ T cells upon repeated dosages of a ⁇ T cell activator IL-2 and IFN ⁇ , in a relevant in vivo model.
- This experiment also underlines repeated ⁇ T cell amplification upon repeated dosages of a ⁇ T cell activator, IL-2 and IFN ⁇ , in a relevant in vivo model.
- Example 6 Intracellular staining of IFN ⁇ and TNF ⁇ of PBMC in different activation conditions.
- Fresh PBMC of a donor (containing 2% ⁇ T cells in total PBMC) were incubated for 4 hours in 96-U well plates with the following conditions:
- Activation of ⁇ T cells in medium or with type I IFN alone is negligible.
- Activation of ⁇ T cells with a ⁇ T cell activator, in particular CHDMAPP is moderate.
- activation of ⁇ T cells with a ⁇ T cell activator and a type I IFN is drastically enhanced compared to other conditions. This experiment underlines that the activation of ⁇ T cells is synergistically enhanced when ⁇ T cells are activated by both a ⁇ T cell activator and a type I IFN.
- Example 7 Crossed dose response effect of cytokine secretion (i.e. IFN ⁇ ) induced by activated ⁇ T cells using increasing amounts of CHDMAPP and increasing amounts of IFN ⁇ .
- IFN ⁇ cytokine secretion
- CHDMAPP ⁇ increasing doses of CHDMAPP: 0, 0.8, 2.5, 7.4, 22, 66.7, 200, 600 nM.
- IFNa 2B 0, 4, 12, 37, 111, 333, 1000 IU/ml (in the legend square on the right of figure 9A).
- the IFN ⁇ released in IU/ml is represented in ordinates in figure 9A.
- the IFN ⁇ release is increased when the CHDMAPP dose increases.
- the IFN ⁇ release is increased when the IFN ⁇ dose increases.
- Figure 9C is a graphical representation of the synergistic effect observed. Briefly, the theoretical effect of IFN ⁇ alone is calculated, the theoretical effect of CHDMAPP alone is calculated, these two effects are added to mimic a simple additive effect of IFN ⁇ and CHDMAPP ("additive effect"). Then this "additive effect” is subtracted from the obtained results.
- Figure 9C thus represents the synergistic effect obtained, e.g. the unforeseeable effect obtained on IFN ⁇ secretion when IFN ⁇ and CHDMAPP as used together in combination. The obtained results clearly underline the synergy observed between
- Example 8 Effect of CHDMAPP alone or in combination with interferon alpha on plasmatic cytokines release in cynomolgus monkey.
- CHDMAPP s.c, 3 mg/kg
- IFN ⁇ 2B s.c, 27 ⁇ g/administration
- FIG. 1OA and 1OB report the secretion of IFN ⁇ (Figure 10A) and MCP-I (Figure 10B) for each condition (CHDMAPP alone: light grey, dots, CHDMAPP+IFN ⁇ : dark grey, triangles).
- Figures 1OA and 1OB underline the fact that upon restimulation, the ability of ⁇ T cells to release cytokines is less impaired when the macaques are administered the combination of products rather than CHDMAPP alone. The combination treatment thus induces a more prolonged therapeutic effect.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Gastroenterology & Hepatology (AREA)
- Zoology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
The present invention provides novel approaches and strategies for efficient regulation of γδ T cells in vivo, in a subject, particularly a human subject or a non-human primate. The present invention now discloses particular compositions and methods that can be used to induce the activation of γδ T cells in vivo, in a subject. These compositions and methods employ the conjoint treatment of an individual with a γδ T cell activator and type I IFN and are particularly suited for immunotherapy in a subject, particularly in a subject having a cancer, an infectious or an autoimmune disease.
Description
IMPROVED METHODS OF USING PHOSPHOANTIGENS
FOR THE TREATMENT OF DISEASES
FIELD OF THE INVENTION The present invention relates to compositions and methods for regulating an immune response in a subject, particularly a T cell response in a subject. The present invention more specifically discloses efficient methods of regulating the innate immunity in a subject, such as by regulating the activity of gamma-delta T cells in a subject. The invention discloses that particular combinations of particular agents, such as a type I interferon cytokine and a gamma-delta T cell activator, can produce a remarkable activation of gamma-delta (γδ) T cells in vivo, a remarkable replication inhibition of virus replication and a remarkable increase in a subject's immune defense, against an infection, a cancer or an immune disease. The invention can be used for therapeutic purposes, to produce, regulate or facilitate an immune response in a subject infected by a disease. It is particularly suited to regulate a protective immune response in subjects having a proliferative disease, an infectious disease or an autoimmune disease.
BACKGROUND OF THE INVENTION
T cells of the gamma-delta type are expressed by most mammalian species. They represent 1- 10% of total circulating lymphocytes in healthy adult human subjects and most non-human primates (NHP). Most human peripheral blood gamma-delta T cells express a gamma-delta TCR heterodimer encoded by Vgamma9/Vdelta2 genes, some NK-lineage receptors for MHC class I and almost no CD4 nor CD8. These cells have been shown to exhibit strong, non MHC-restricted, cytolytic activity against virus-infected cells (Poccia et al, J. Leukocyte Biology, 62, 1997, p. 1-5), parasite-infected cells (Constant et al, Infection and Immunity, vol. 63, no. 12, Dec. 1995, p. 4628-4633), or tumor cells (Fournie et al, Bonneville, Res. Immunol, 66th FORUM IN IMMUNOLOGY, 147, p. 338-347).
These cells are also physiologically amplified in the context of several unrelated infectious diseases such as tuberculosis, malaria, tularemia, colibacillosis and also by B-cell tumors (for review see Hayday, 2000).
These cells are thus viewed as potent effectors of innate immunity. Because of their potent activity against tumor cells or infected cells, γδ T cells represent a very attractive candidate for immunotherapy. Notably, since activated γδ T cells exert potent cytolytic activity and Tn-I cytokine secretion, these cells represent an important resource of anti-infectious and antitumoral effectors. Accordingly, it would be highly valuable to have methods available for regulating the activity (including the expansion and/or cytolytic activity, for instance), of these cells in vivo, in a subject. In microbes, Vγ9/Vδ2+ lymphocytes spontaneously recognize a structurally related set of nonpeptide antigens, referred to as natural phosphoantigens and alkylamines. In B cell tumors, the
nature of antigens for the γδ T cells remains unidentified. Vγ9/Vδ2+ lymphocytes are also responsive to a variety of virally infected-, activated- or tumoral cell types without prior exposure. Again, in these situations, the responsible antigens remain unknown. It has been shown that, in vitro, Vγ9/Vδ2+ lymphocytes respond to synthetic drugs such as therapeutic aminobisphosphonates (reviewed in Espinosa, J Biol Chem 2001, Vol. 276(21), pp. 18337-18344), leading to their in vitro activation.
Recognition of natural non-peptide antigens is mediated by the γδ TCR, through amino acid residues located on both Vγ9- and Vδ2- CDR3 regions. Although neither processing nor presentation by CDl or MHC molecules is involved, Vγ9/Vδ2+ lymphocyte activation by non-peptide antigens appears to require cell-to-cell contact (Lang, J. Immunol. 1995 (154) 5986; Morita, Immunity, Vol. 3, 495-507, 1995; Miyagawa, J. Immunol 2001, 166: 5508-5514; Rojas, Infection and Immunity, 2002, vol70(8)).
The first set of clinical evidence for in vivo expansion of human Vγ9/Vδ2+ lymphocytes induced by phosphoantigen agonists came from the finding of increases of circulating γδ T cells within one to three weeks in human adults with multiple myeloma after therapeutic intravenous injection of 60-90 mg of pamidronate (Kunzmann,Blood, 2000, vol 96(2). However, the actual mode of action of pamidronate is unclear and might involve an indirect effect on accessory cells (Miyagawa, 2001) and no appropriate conditions were disclosed to allow optimized activation of γδ T cells using such compound.
The inventors and/or their colleagues have previously characterized a number of immunomodulatory compounds capable of modulating the activity and proliferation of γδ T cells. These compounds (also called "phosphoantigens") generally share a common structure in that they are organophosphate compounds. The classes having greatest potency are more particularly phosphate esters and phospho-phosphoroamidate esters. The particular compounds, used by the inventors and their colleagues in a lead clinical trial, comprise a pyrophosphate moiety. The lead molecules of this series of compounds developed by the inventors and their colleagues are called Phosphostim™, which is at present being evaluated in three Phase II clinical trials in oncology and Picostim™ currently at the preclinical stage. A number of other compounds which activate γδ T cells are known as well, although most of these act indirectly (e.g. act on other immune cells which in turn activate γδ T cells) or act directly on γδ T cells but are less potent.
For example in the treatment of HCV, many research teams have explored different possible schemes of treatment to control the onset of the chronic infection. For example, Larkin et al. (Journal of Interferon and Cytokine Research, 23:247-257), have proposed the action of combinations of Type I and Type II interferons on HCV virus replication. Up to date more than 70% of HCV infected subject do not clear totally the infection and remain chronically infected, the infection lasting more than 6 months. This long lasting infection can cause inflammation of the liver (chronic hepatitis). This condition can progress to scarring of the liver (fibrosis), and advanced scarring (cirrhosis). In some
cases, those with cirrhosis will go on to develop liver failure or other complications of cirrhosis, including liver cancer. Among untreated patients, 30% progress to liver failure or other complications of cirrhosis, including liver cancer, in less than 20 years.
Agrati et al, International Immunology, 2005, Vol. 18, No. 1, 11-18, , have suggested that γδ T cells could represent an additional strategy to inhibit HCV replication, γδ T cells activated by phosphoantigens induce a non-cytolytic inhibition of subgenomic HCV replication.
SUMMARY
The present invention proposes a treatment setting useful for treating a disease wherein the administration of type I interferon (type I IFN) is needed. Type I IFN includes in particular IFNα and
IFNβ. Infectious diseases such as HCV and HBV are currently treated with IFNα but any other disease where the administration of a type I IFN may be beneficial is encompassed by the present invention, for example other infections such as HIV, HTLV-I, HPV, HHV8. The present invention is also suited for the treatment of subjects having more than one infection and wherein the administration of a type I IFN may be beneficial for the treatment of at least one of the infections, e.g. co-infected subjects, i.e. HCV/ AIDS co-infected subjects, HCV/Tuberculosis co-infected subjects and the like. The present invention is also suited for the treatment a proliferative disease, in particular a cancer. Cancers such as melanoma or kidney cancer are currently treated with IFNα but any other cancer where the administration of a type I IFN may be beneficial is encompassed by the present invention, for example, other cancers such as Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors.
In patients, the functions of immune cells are usually impaired, thereby leading to a partial or non-efficient treatment of diseases. There is thus a need for a treatment enabling to strengthen the immune response in patients, in particular in patients having an impaired immune profile. The method and compositions according to the present invention enable to strengthen the γδ T cell response thereby enhancing the potency of clearance of viruses, tumors and autoimmune wounds. The present invention provides a composition where IFNα sensitizes γδ T cells that are weak responders to γδ T cell activator stimulation. The combination of a type I IFN and a γδ T cell activator potentiates the cytokine release and activation of γδ T cells compared to single γδ T cell stimulation. The present invention thus provides a method to treat patients weakly responding to a γδ T cell activator by further administering an effective dose of a type I IFN.
In another aspect, even in healthy subjects, sometimes the immune response is only partial which leads to a partial control of the acute phase of the disease and a residual disease persists. For example upon infection, some copies of the virus persist, in tumors, some tumor cells may persist as well. These residual disease markers may lead to a chronic disease or the redevelopment of said disease, it is therefore crucial to mount a total and effective immune response to lead to a total
clearance of the disease and cure patients. The present invention provides a method to clear an acute phase infection comprising administering to a subject in need thereof an effective amount of a γδ T cell activator and an effective amount of a type I IFN.
There is thus a need for a treatment leading to a boost of the immune system for the treatment of viral disease, cancer and autoimmune diseases.
It has been previously reported that successive administrations of a γδ T cell activator and IL- 2 induce a less vigorous expansions in γδ T cell number, suggesting a possible exhaustion of γδ T cells (Sicard et al, JI 2005). The inventors have established that the use of a type I IFN, together with a γδ T cell activator, in the presence of IL -2, decreases this exhaustion phenomenon, thereby providing a better amplification rate response and cellular activation upon multiple γδ T cell activation with IL-2.
Foxp3+ CD4+CD25+ regulatory T cells (Treg) control immune responses to self- and foreign- antigens and play a major role in maintaining the balance between immunity and tolerance (Sakaguchi S. et al, Annu Rev Immunol. 2004;22:531-562, Tang Q et al , Nat Immunol. 2008;9:239-244). Treg have been shown to broadly suppress activation, proliferation and/or effector functions of various immune cell populations, in particular in γδ T cells (Li et al, Blood. 2008 Jun 15; 5629-36), thereby leading to an impaired response to therapy, i.e. lead to chronic infection when acute phase is not totally cleared. Recent studies have shown that IL-2 administration can lead to an increase in the frequency of circulating CD4+CD25+ regulatory T cells in cancer patients (Ahmadzadeh M, et al, Blood. 2006; 2409-2414). The inventors have established that the administration of a type I IFN, together with a γδ T cell activator and IL-2 leads to a decreased production of Treg, thereby improving the potency of γδ T cell response in a subject. The present invention provides also a method to reduce the proportion of Treg in a patient. The present invention provides a method to obtain a sustained response of successive γδ T cell activator administrations in a subject. Whereas cytokine secretion has previously been reported only upon treatment with γδ T cell activator and IL-2 in healthy primates (Sicard, JI 2005, 175, pp5471-5480), the inventors have established that cytokine secretion occurs when cells are stimulated with a γδ T cell activator, even in the absence of IL-2 and that this cytokine secretion can be enhanced in the presence of a type I IFN. The present invention provides a method to enhance cytokine secretion in a subject comprising administering an effective amount of a γδ T cell activator and an effective amount of a type I IFN.
The invention also provides a method for enhancing the cytokine secretion in a subject comprising contacting γδ T cells with an effective amount of a γδ T cell activator and an effective amount of type I IFN, especially IFNα. In an embodiment, said method is performed without further administering IL-2 to said subject. The present invention provides a method for enhancing the cytokine secretion of γδ T cells activated with a γδ T cell activator comprising contacting said activated γδ T cells with an
effective amount of a type I IFN, especially IFNα. In an embodiment, said method is performed without further administering IL-2 to said subject.
A commonly reported drawback of Type I IFN treatment is the induced toxicity in patients, reported undesirable side effects include headache, fever, flu like syndrome and the like. The effects are highly undesirable, particularly, in non symptomatic patients, who do not feel sick when infected but who may experience the listed undesirable side effects upon treatment. One object of the present invention is also to provide a treatment necessitating a smaller amount of type I IFN for the obtention of a similar therapeutic effect, and thus inducing less undesirable side effects than currently described monotherapy treatments. The present invention also provides a method of reducing an type I IFN treatment-related side effect in a subject receiving or in need of treatment with a type I IFN, comprising administering to a subject in need of treatment with a type I IFN, a pharmaceutical composition comprising type I IFN and a pharmaceutical composition comprising a γδ T cell activator in an amount which, together with the administered type I IFN results in an effective treatment having a reduced type I IFN treatment related side effect in comparison with the side effects that would occur upon sole effective type I IFN treatment.
The present invention provides a method to inhibit viral replication comprising contacting supernatants of activated γδ T cells with a virus. The invention also provides a composition comprising IFNα and a γδ T cell activator for the treatment of an infectious disease, wherein the antiviral effect of the combined products is greater than each antiviral effect of each product alone.
The invention also provides a composition comprising IFNα and a γδ T cell activator for the treatment of an infectious disease wherein the combination enables to clear chronic infection, in particular HCV infection.
The present invention also provides a composition comprising IFNα and a γδ T cell activator to obtain a sustained virologic response (SVR) in virally infected patient, in particular HCV patients.
The present invention is based on observations during (i) in vitro testing of activated γδ T cells ability to inhibit virus replication and (ii) studies in non-human primates human clinical trial using CHDMAPP (also referred to as Picostim™), where it was observed that this compound, when used in certain therapeutic regimens with type I interferon leads to an improved γδ T cell activation and proliferation, including a strong cytokine secretion, an efficient γδ T cell activation. In viral diseases, the present invention also proves an efficient viral replication inhibition.
The present invention therefore provides novel approaches and strategies for efficient regulation of γδ T cells in vivo and therefore disease management, in a subject, particularly a human subject or a non-human primate, wherein the administration of type I IFN can be beneficial. In an embodiment, the disease is selected from a cancer, an infection or an autoimmune disease.
The present invention now discloses particular compositions and methods that can be used to treat diseases, comprising inducing the inhibition of viruses in vivo, diminishing the tumor burden, or controlling an autoimmune disease, in a subject, through γδ T cell activation. These compositions and methods employ the conjoint treatment of a subject with a γδ T cell activator and type I IFN. The present invention provides a method for treating a disease comprising administering to a patient an effective amount of a γδ T cell activator in combination with an effective amount of a type I IFN. The present invention provides also a pharmaceutical composition comprising an effective amount of a γδ T cell activator and an effective amount of a type I IFN for the treatment of a disease. In another embodiment, the present invention provides a method for the treatment of a disease wherein secretion of IFNy is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The present invention provides also a pharmaceutical composition comprising an effective amount of a γδ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein secretion of IFNy is needed
In another embodiment, the present invention provides a method for the treatment of a disease wherein an immune stimulation is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The present invention provides a method to strengthen the immune response in patients, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. In another embodiment, the present invention provides a pharmaceutical composition comprising an effective amount of a γδ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein an immune stimulation is needed
The present invention also provides a method for the treatment of a disease wherein the stimulation of a γδ T cell is needed, comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The present invention also provides a pharmaceutical composition comprising an effective amount of a γδ T cell activator and an effective amount of a type I IFN for the treatment of a disease wherein the stimulation of a γδ T cell is needed
In another aspect, the invention provides a method for the prevention of γδ T cell exhaustion comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The invention further provides a pharmaceutical composition comprising an effective amount of a γδ T cell activator and an effective amount of a type I IFN for the prevention of γδ T cell exhaustion.
In another aspect, the invention provides a method for preventing or treating a chronic viral infection comprising administering to a patient an effective dose of a γδ T cell activator in combination with an effective dose of IFNα. The invention also provides a pharmaceutical
composition comprising an effective amount of a γδ T cell activator and an effective amount of IFNα for the prevention or treatment of a chronic viral infection
In another aspect, the invention provides a method for the prevention of Treg onset comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The invention also provides a method to reduce the proportion of Treg in a patient comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. The invention further provides a method to obtain a sustained response of successive γδ T cell activator administrations in a subject comprising administering to a subject in need thereof an effective amount of a type I IFN together with a γδ T cell activator. In an embodiment, the present invention provides a method of determining a suitable treatment, comprising: assessing a subject's response to γδ T cell activator, wherein a determination that a subject has a decreased response indicates that the subject will benefit from a γδ T cell activator and type I IFN combination treatment. In a further embodiment, the method comprises treating said subject with a γδ T cell activator and type I IFN combination treatment. In a further embodiment, the method according to the invention further comprises assessing a subject's response to γδ T cell activator by obtaining a biological sample comprising γδ T cells from the subject, and bringing the γδ T cells into contact with a γδ T cell activator and assessing the level of activation or proliferation of γδ T cells.
The present invention also provides a method of administering a γδ T cell activator to an individual, comprising administering a predetermined amount of γδ T cell activator and type I IFN, where said predetermined amount is independent of an individual subject's γδ T cell responsiveness.
The present invention also provides a method of producing a compound, comprising (a) determining whether a candidate γδ T cell activator has a synergistic cytokine induction or antiviral activity with a type I IFN, and (b) if said candidate γδ T cell activator is determined to have a synergistic cytokine induction or antiviral activity with a type I IFN, producing a quantity of said candidate γδ T cell activator, optionally for administration in combination with the type I IFN.
The present invention also provides a method of potentiating cytokine release comprising administering to a patient an effective amount of a γδ T cell activator and an effective amount of a type I IFN. The present invention also provides a method of enhancing the cytokine response of a subject to a γδ T cell activator administration comprising further administering a type I IFN.
The present invention also provides a method of activating γδ T cells in a patient being weakly responder to a γδ T cell treatment comprising further administering a type I IFN.
The present invention also provides a method of clearing a chronic infection comprising administering a γδ T cell activator to an infected subject, together with a type I IFN.
The present invention also provides a method to obtain a repeated cytokine secretion upon multiple γδ T cell activator stimulation comprising further administering a type I IFN. In an embodiment, IL-2 is further administered to said patient.
The present invention also provides a method to avoid γδ T cell amplification exhaustion or to obtain repeated cytokine release upon γδ T cell activator stimulation comprising administering to a subject in need thereof an effective amount of a γδ T cell activator, an effective amount of a type I IFN and an effective amount of IL-2.
The present invention also provides a method of determining a treatment suitable for a subject, comprising: assessing the level of Treg response/level/activation in a subject wherein a determination that a subject has a increased Treg response/level/activation (e.g. compared to other subjects, compared to a predetermined level or threshold) indicates that the subject will benefit from a γδ T cell activator and type I IFN combination treatment. In an embodiment, the subject, prior to said assessment step, has been treated with IL-2 or IL-2 and a γδ T cell activator. In another embodiment, said subject is administered a γδ T cell activator and type I IFN combination treatment. In any of the embodiments listed above, the γδ T cell activator is of formula I to II, more particularly, selected from the list consisting of A, B, C, D, E, F, G, and H.
In any of the embodiments listed above, the γδ T cell activator is administered at a dose of about 0.1 to 30 mg/kg. In any of the embodiments listed above, the γδ T cell activator is administered subcutaneously. In any of the embodiments listed above, the type I IFN is IFNα. In any of the embodiments listed above, IFNα is administered at a dose comprised between 0.5 and 150μg per week. In an aspect, IFNα dose is reduced compared to standard treatment. In any of the embodiments listed above, IFNα is administered subcutaneously.
In any of the embodiments listed above, the disease is selected from the list consisting of a proliferative disease, an infectious disease or an autoimmune disease, in particular, the disease is a viral infection, especially HIV, HTLV, HPV, HHV. In another aspect, the viral infection is chronic; in particular, the chronic infection is HCV. In another aspect, the disease is a co infection, in particular, HCV/ AIDS or HCV/Tuberculosis. In any of the embodiments listed above, the disease is a cancer, in particular Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors. In any of the embodiments listed above, the γδ T cell activator administration occurs once a week for about 3 weeks. In any of the embodiments listed above, the IFNα administration occurs once a week for about 3 weeks.
In any of the methods listed above, the method comprises further administering an antiviral agent. In any of the methods listed above, the method comprises further administering IL-2 at a low dose.
DESCRIPTION OF THE FIGURES
Figure 1: Dose response potentiation effect of cytokine secretion (i.e. IFNγ) by CHDMAPP activated γδ T cells in the presence of IFNα. CHDMAPP dose dependant IFNγ secretion by purified γδT cells has been studied for various amounts of IFNα added in the culture medium (full dots: CHDMAPP alone, open squares: CHDMAPP + IFNα2B, 10 IU, full triangles:
CHDMAPP + IFNa2B, 100 IU, open lozenges: CHDMAPP + IFNa2B, 1000 IU). The quantity of IFNγ secreted points towards the potentiation of γδ T cell functions when cultured in the presence of IFNα. The CHDMAPP concentration (in nM) is represented in axis, in a log scale, the IFNγ release (in IU/ml) is represented in ordinates. Figures 2A and 2B: Dose response inhibitory effect of CHDMAPP stimulated PBMC- supernatants on HCV replicon model, in two representations. Supernatants of human PBMC stimulated by increasing doses of CHDMAPP, as detailed in example 3, decrease HCV RNA level in a dose dependant way. The decrease of HCV replication is inversely correlated with the amount of IFNγ released in the supernatants. Figure 2B: The CHDMAPP concentration is represented in axis (in nM, log scale), the concentration of HCV RNA copy (per ml, in millions) is represented in ordinates.
EC50 has been calculated at 1.68 nM, ICi0 has been calculated at 7.79 nM).
Figures 3A and 3B: Antiviral effect of supernatants of exogenous IFNγ on HCV replicon model, in two representations. Figure 3B: The IFNγ concentration is represented in axis (in IU/ml, log scale), the concentration of HCV RNA copy (per ml, in millions) is represented in ordinates. EC50 has been calculated at 0.217 nM, IC90 has been calculated at 5.46 nM).
Figures 4A and 4B: Cytokine release profile (4A: MCP-I, IL-6, IL-lβ, 4B: IFNγ, TNFα, AUC (in pgxh/ml, left hand colunm) and Cmax (in pg/ml, right hand column)) in NHP during three cycles of treatment with CHDMAPP +/- IFNα. AUC, is represented for cycle 1, cycle 2, cycle 3 and the last column represents the total of the three cycles, Cmax is represented for cycle 1, cycle 2 and cycle 3). Animals have been treated for three cycles with CHDMAPP + IFNα (open squares) or with CHDMAPP only (full dots) as described in example 4. The cytokine release in CHDMAPP + IFNα treated animals is improved compared to CHDMAPP treated animals, thereby underlining the potentiating effect of the conjoint administration of IFNα and a γδ T cell activator.
Figures 5A and 5B: γδ T cell profile after multiple CHDMAPP activation, in the presence or absence of IFNα and/or IL-2 (5A). Treg profile after multiple CHDMAPP activation, in the presence or absence of IFNα and/or IL-2 (5B). Experiments have been conducted according to example 5. Time in days is represented in axis, absolute number of γδ T cells per μl of blood in ordinates. Figure 5A: Despite multiple CHDMAPP treatment, the γδ T cell show an enhanced amplification in CHDMAPP+IFNα+IL-2 treated NHP (full square line) than in CHDMAPP-IL-2 treated NHP (dark grey and open dots line) or in IFNα alone treated NHP (grey
triangles line). The exhaustion of γδ T cell amplification observed with CHDMAPP-IL-2 is statistically less marked in the CHDMAPP + IFNα + IL-2 group. Figure 5B: Whereas classical γδ T cell amplification with CHDMAPP and IL-2 increases the number of CD4+CD25+ T cells (Treg) (dark grey and open dots line), in CHDMAPP + IFNα + IL-2 treated NHP (full square line), the Treg number is well controlled and remains acceptable upon multiple γδ T cell stimulation.
Figure 6: IFNγ release profile after multiple CHDMAPP activation, in the presence or absence of IFNα and/or IL-2. Experiments have been conducted as described in example 5. The sampling time is represented in axis (day, hour), the IFNγ concentration (in pg/ml) in represented in ordinates. Multiple stimulation with CHDMAPP, IFNα and IL-2 (full square line) leads to an enhanced cytokine secretion (here IFNγ), compared to CHDMAPP and IL-2 only treated NHP (dark grey and open dots line), whereas a single treatment with IFNα (grey triangles line) induces no remarkable cytokine secretion. This experiment underlines the long term potentiation of IFNγ secretion by γδ T cells stimulated by CHDMAPP, IL-2 and IFNα in an in vivo model.
Figure 7: HCV RNA replication inhibition by sera of CHDMAPP treated NHP (non human primates), as described in example 3. HCV RNA copies (in million per ml) is represented in ordinates. Sera of 2 NHP (NHPl: dashed columns, NHP2: black columns) have been withdrawn before CHDMAPP administration (predose), and a few hours (e.g. 4-5 hours) after CHDMAPP administration and diluted (1/10, 1/20 and 1/30), collected sera have been tested in the replicon model described in example 3. Whereas untreated serum induces no viral inhibition (predose), serum collected after CHDMAPP administration induces a strong viral replication inhibition (dilution 1/10,
1/20 and 1/30). Ribavirin (200 μM) is provided as control. Moreover, for an equivalent amount of plasmatic IFNγ (table 2), the level of inhibition between the two NHP is different, underlining that the inhibitory effect on viral replication of CHDMAPP is not only IFNγ concentration dependent.
Figure 8: Intracellular staining of IFNα and TNFα of PBMC in different activation conditions. Cells have been stimulated in vitro according to example 6. Each dot plot represents a different activation conditions: 8A: medium, percentage of γδ T cells expressing IFNγ and Vδ2 chain (IFNy+ Vδ2+ cells): 0.4%; 8B: CHDMAPP, % IFNy+ Vδ2+ cells: 14.9%; 8C: IFNα, % IFNy+ Vδ2+ cells: 0.5%; 8D: CHDMAPP and IFNα, % IFNy+ Vδ2+ cells: 34.0%), IFNγ is represented in ordinates, Vδ2 in axis. Whereas activation of γδ T cells in medium or with type I IFN alone is negligible and activation of γδ T cells with CHDMAPP is only moderate, activation of γδ T cells with CHDMAPP and IFNα drastically enhanced compared to other conditions. This experiment underlines that the activation of γδ T cells is synergistically enhanced when γδ T cells are activated by both a γδ T cell activator and a type I IFN.
Figure 9: Crossed dose response IFNα/CHDMAPP on PBMCs: potentiation effect of cytokine secretion (i.e. IFNγ) by CHDMAPP activated γδ T cells in the presence of IFNα.
CHDMAPP dependant IFNγ secretion by purified γδT cells was studied for various amounts of IFNα and CHDMAPP added in the culture medium. The quantity of IFNγ secreted points towards the potentiation of γδ T cell functions when cultured in the presence of IFNα. Figure 9 A: The histogram represents all the IFNγ secretion registered for each CHDMAPP/IFNα dose couple experimented. Figure 9B is a three dimensional representation of the synergic effect between CHDMAPP and IFNα
(total effect, minus as simple extrapolated additive effect of the combination treatment).
Figure 10: Cytokine release profile in NHP after multiple CHDMAPP activation, in the presence or absence of IFNα, for IFNγ (10A) and MCP-I (10B). Experiments was conducted according to example 8. Time is represented in hours in axis. Cytokine release is represented in pg/ml in ordinates. In the absence of IFNα, the cytokine release gradually decreases over multiple
CHDMAPP administrations (light grey; dots), on the contrary, the cytokine secretion level is maintained in the CHDMAPP+INFα group (dark grey; triangles). These data show that the co administration of IFNα together with CHDMAPP provides a sustained cytokine release, leading to a better management of the disease.
DETAILED DESCRIPTION DEFINITIONS
Where "comprising" is used, this can preferably be replaced by "consisting essentially of, more preferably by "consisting of. As used in the specification, "a" or "an" may mean one or more. As used in the claim(s), when used in conjunction with the word "comprising", the words "a" or "an" may mean one or more than one. As used herein "another" may mean at least a second or more.
Where hereinbefore and hereinafter numerical terms are used, they are meant to include the numbers representing the upper and lower limits. For example, "between 1 and 3" stands for a range "from and including 1 up to and including 3", and "in the range from 1 to 3" would stand for "from and including 1 up to and including 3". The same is true where instead of numbers (e.g. 3) words denoting numbers are used (e.g. "three").
"Weekly" stands for "about once a week" (meaning that more than one treatment is made with an interval of about one week between treatments), the about here preferably meaning +/-1 day (that is, translating into "every 6 to 8 days"); most preferably, "weekly" stands for "once every 7 days".
"3-weekly" or "three-weekly" stands for "about once every three weeks" (meaning that more than one treatment is made with an interval of about three weeks between treatments), the about here preferably meaning +/-3 days (that is, translating into every 18 to 24 days); most preferably, "weekly" stands for "once every 21 days" (=every third week).
The term "about" or "approximately" usually means within 20%, more preferably within
10%, and most preferably still within 5% of a given value or range. Alternatively, especially in biological systems (e.g., when measuring an immune response), the term "about" means within about a log (i.e., an order of magnitude) preferably within a factor of two of a given value. Within the context of the present invention, the expressions "regulating the activity of γδ T cells", "stimulating the activity of γδ T cells", "activating γδ T cells" and "regulating the activity of γδ T cells" designate causing or favoring an increase in the number and/or biological activity of such cells in a subject. Stimulating and regulating thus each include without limitation modulating (e.g., stimulating) expansion of such cells in a subject and/or, for instance, triggering of cytokine secretion (e.g., TNFα or IFNγ). γδ T cells normally represent between about 1-10% of total circulating lymphocytes in a healthy adult human subject. The present invention can be used to significantly increase the γδ T cells population in a subject, particularly to reach at least 30% of total circulating lymphocytes, typically 40%, more preferably at least 50% or 60%, or from 50% to 90%. Regulating also includes, in addition or in the alternative, modulating the biological activity of γδ T cells in a subject, particularly their cytolytic activity or their cytokine-secretion activity. The invention defines novel conditions and strategies for increasing the biological activity of γδ T cells towards target cells.
In accordance with the present invention there may be employed conventional molecular biology, microbiology, and recombinant DNA techniques within the skill of the art. Such techniques are well-known and are explained fully in the literature. See, e.g., Sambrook, Fritsch and Maniatis, Molecular Cloning: A Laboratory Manual, Second Edition (1989) Cold Spring Harbor Laboratory
Press, Cold Spring Harbor, New York (herein "Sambrook et al, 1989"); DNA Cloning: A Practical Approach, Volumes I and II (D. N. Glover ed. 1985); Oligonucleotide Synthesis (M. J. Gait ed. 1984); Nucleic Acid Hybridization, B. D. Hames & S. J. Higgins eds. (1985); Transcription And Translation, B. D. Hames & S. J. Higgins, eds. (1984); Animal Cell Culture, R. I. Freshney, ed. (1986); Immobilized Cells And Enzymes, IRL Press, (1986); B. Perbal, A Practical Guide To Molecular
Cloning (1984); F. M. Ausubel et al. (eds.), Current Protocols in Molecular Biology, John Wiley & Sons, Inc. (1994).
As used herein, the terms "conjoint", "in combination" or "combination therapy", used interchangeably, refer to the situation where two or more therapeutic agents affect the treatment or prevention of the same disease. The use of the terms "conjoint", "in combination" or "combination therapy" does not restrict the order in which therapies (e.g., prophylactic or therapeutic agents) are administered to a subject with the disease. A first therapy can be administered prior to (e.g., 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks before), concomitantly with, or subsequent to (e.g., 5 minutes, 15 minutes, 30 minutes, 45 minutes, 1 hour, 2 hours, 4 hours, 6 hours, 12 hours, 24 hours, 48 hours, 72 hours, 96 hours, 1 week, 2
weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 8 weeks, or 12 weeks after) the administration of a second therapy to a subject with a disease. As used herein, the term "concomitantly" refers to the situation where two compounds are administered at the same time or within a few minutes i.e. one directly after the other. As used herein, the term "conjointly" refer to the situation where two compounds are administered during the same day within 1, 2, 3, 4, 5, 6, 7, 8, 12, 18, 24 hours. As used herein, the term "sequentially" refer to the situation where two compounds are administered within 1, 2, 3, 4, 6, 7 or more days, 1, 2, 3 or more weeks.
Within the context of the present invention, the term "separately administered" indicates that the active ingredients are administered at a different site or through a different route or through a different schedule to the subject. Accordingly, the ingredients are generally not mixed together prior to administration, although they may be combined in a unique package in suitable separated containers.
As used herein, the term "Sustained virologic response" or "SVR" means that the virus (the viral load) remains undetectable in the blood for a long time, i.e. for at least six months after finishing the treatment. The viral load is measure by quantitative PCR (Polymerase Chain Reaction) from a biopsy (i.e. a blood extract) of an infected patient. In an embodiment, the present invention is useful for the treatment of infectious disease. In a particular embodiment, the invention is useful to treat chronic viral diseases, such a chronic HCV. The aim of a viral treatment can, for instance, be to obtain a satisfying SVR (sustained virologic response), which means that the virus cannot be detected in the patient's blood for a time length, i.e. at least 6 months.
As used herein, the term "subject's response" refers to the ability of a subject to amplify γδ T cells or release cytokines in response to stimulation i.e. by a γδ T cell activator or by a cytokine, in particular a type I IFN.
As used herein, the terms "weak response", "weakly responding", "decreased response" refers to an impaired γδ T cell response or an impaired cytokine release compared to standard response in other subject. A weak response or decrease response is for example 10%, 20%, 30%, 40%, 50% or more decreased compared to what is usually assessed in patients. For example, if a cytokine release is usually of lOOpg/ml upon a given dose of γδ T cell activator, a weak response would be a 60pg/ml of the same cytokine at the same γδ T cell activator dose. Ultimately, a 90% or 100% decreased response will be qualified as a non response, and the subject is nonresponder.
As used herein the term "strengthen the immune response" refers to the improvement of the γδ T cell amplification or the cytokine release of a subject that usually present a weak or decreased response.
As used herein, the term "potentiate" or "potentiating" refers to the ability of a treatment, compound, method, to allow an enhanced results compared to what was usually obtained or expected based on previous experiments. For instance, a cytokine release is potentiated when one obtains a
10%, 20%, 30%, 40% or more increase in cytokine release compared to the cytokine level that was commonly observed before the present invention was made.
As used herein, the term "synergic", "synergistically", refers to the action of a combination wherein this action is superior to the mere addition of each effect detected separately.
Diseases Cancers
A variety of cancers can be treated using the methods and compositions according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFNα may be beneficial. In an embodiment, the cancer to be treated is selected from melanoma, renal cell carcinoma, kidney cancer, NHL (Non Hodgkin Lymphoma), myeloma, CML (Chronic Myeloid Leukemia), hairy cell leukemia, cutaneous T cell leukemia, follicular lymphoma, squamous cell carcinoma, prostate carcinoma, bladder cell carcinoma, cervical dysplasia and colon carcinoma. Infectious diseases
A variety of infectious diseases can be treated using the methods and compositions according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFNα, may be beneficial. In an embodiment, the viral disease to be treated is selected from HCV (Hepatitis C virus), HBV (Hepatitis B virus), HIV (Human Immunodeficiency Virus), HPV (Human Papiloma Virus), HTLV-I (Human T cell lymphoma virus type 1), HHV (Human
Herpes Virus), HHV8, condyloma acuminatum, cytomegalovirus, Epstein-Barr virus and Kaposi syndrome.
The present invention is particularly suited for the treatment of HCV. Hepatitis C is a blood- borne infectious disease that is caused by the Hepatitis C virus (HCV), affecting the liver. The Hepatitis C virus (HCV) is a small, enveloped, single-stranded, positive sense RNA virus. The hepatitis C virus (HCV) is spread by blood-to-blood contact. The symptoms of infection can be medically managed, and a proportion of patients can be cleared of the virus by a course of anti-viral medicines. An estimated 150-200 million people worldwide are infected with hepatitis C. The infection is often asymptomatic, people with HCV infection can experience mild symptoms, and consequently do not seek treatment. The virus is detectable in the blood within 1 to 3 weeks after infection, and antibodies are detectable within 3 to 12 weeks. 20-30% infected patients clear the virus during the acute phase, the other 70 to 80% patients develop chronic HCV e.g., an infection lasting more than 6 months. Once established, chronic infection can cause inflammation of the liver (chronic hepatitis). This condition can progress to scarring of the liver (fibrosis), and advanced scarring (cirrhosis). In some cases, those with cirrhosis will go on to develop liver failure or other complications of cirrhosis, including liver cancer. Among untreated patients, 30% progress to liver
failure or other complications of cirrhosis, including liver cancer, in less than 20 years. The current treatment of HCV patients is a combination of IFNα and ribavirin for treatment cycles of about 18 to 24 months. Although the effect of each of the products has not been established so far, this combination is proven effective in approximately 15-25% of the patients. Many patients are unresponsive to the treatment or develop a chronic infection. In an embodiment, the object of the present invention is to provide a scheme of treatment that leads to the reduction of the residual disease in chronically HCV infected patients. In another embodiment, the present invention provides a method of treatment, comprising: determining whether a subject has responded to a treatment comprising administration of type I IFN, and if said patient has demonstrated a decreased response or is non responder to IFN treatment, administering to the subject a γδ T cell activator and type I IFN combination treatment.
The present invention is also particularly suited for the treatment of HBV. Hepatitis B is a virus that infects the liver of hominoidae, including humans, and causes an inflammation called hepatitis. It is a DNA virus and one of many unrelated viruses that cause viral hepatitis. Symptoms of the acute illness caused by the virus include liver inflammation, vomiting, jaundice, and rarely, death.
Chronic hepatitis B may eventually cause liver cirrhosis and liver cancer, a fatal disease with very poor response to current chemotherapy. Hepatitis B virus infection may either be acute (self-limiting) or chronic (long-standing). Persons with self-limiting infection clear the infection spontaneously within weeks to months. Children are less likely than adults to clear the infection. More than 95% of people who become infected as adults or older children will stage a full recovery and develop protective immunity to the virus. However, only 5% of newborns that acquire the infection from their mother at birth will clear the infection. Of those infected between the age of one to six, 70% will clear the infection. Acute infection with hepatitis B virus is associated with acute viral hepatitis - an illness that begins with general ill-health, loss of appetite, nausea, vomiting, body aches, mild fever, dark urine, and then progresses to development of jaundice. Chronic infection with Hepatitis B virus may be either asymptomatic or may be associated with a chronic inflammation of the liver (chronic hepatitis), leading to cirrhosis over a period of several years. This type of infection dramatically increases the incidence of hepatocellular carcinoma (liver cancer). Autoimmune diseases Auto immune disease can also be treated using the methods and composition according to the invention. Those diseases are selected among diseases where the administration of type I IFN, in particular IFNβ may be beneficial. Such disease is for instance multiple sclerosis.
Multiple sclerosis is an autoimmune condition in which the immune system attacks the central nervous system, leading to demyelination. Disease onset usually occurs in young adults, and it is more common in women. Common multiple sclerosis treatment is weekly intramuscular injection of interferon β-la (Avonex®, Rebif®) and one of interferon β-lb (Betaseron® or Betaferon®). Those
medications are modestly effective at decreasing the number of attacks and slowing progression to disability, although they differ in their efficacy rate and studies of their long-term effects are still lacking. The present invention is also suited for the treatment of any other autoimmune disease where the administration of a type I IFN may be beneficial.
γδ T cell activators
The term "γδ T cell activator" designates a molecule, preferably artificially produced, which can activate γδ T lymphocytes. It is more preferably a ligand of the T receptor of γδ T lymphocytes. The γδ T cell activators may by of various natures, such as a peptide, lipid, small molecule, etc. It may be a purified or otherwise artificially produced (e.g., by chemical synthesis, or by microbiological process) endogenous ligand, or a fragment or derivative thereof, or an antibody having substantially the same antigenic specificity.
A phosphoantigen that is a γδ T cell activator preferably increases the biological activity or causes the proliferation of γδ T cells, preferably increasing the activation of γδ T cells, particularly increasing cytokine secretion from γδ T cells or increasing the cytolytic activity of γδ T cells, with or without also stimulating the proliferation or expansion of γδ T cells. Accordingly, the γδ T cell activator is administered in an amount and under conditions sufficient to increase the activity γδ T cells in a subject, preferably in an amount and under conditions sufficient to increase cytokine secretion by γδ T cells and/or to increase the cytolytic activity of γδ T cells. Cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay (as described hereunder).
Method for detecting activation of a γδ T cell will be carried out according to standard methods. For example, cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay, or those provided in the examples herein. For example, cytokine secretion can be determined according to the methods described in Espinosa et al. (/. Biol. Chem., 2001, Vol. 276, Issue 21, 18337-18344), describing measurement of TNF-α release in a bioassay using TNFα sensitive cells. Briefly, 104 γδ T cells/well were incubated with stimulus plus 25 units of IL2/well in 100 μl of culture medium during 24 h at 37 0C. Then, 50 μl of supernatant were added to 50 μl of WEHI cells plated at 3 x 104 cells/well in culture medium plus actinomycin D (2 μg/ml) and LiCl (40 inM) and incubated for 20 h at 37 0C. Viability of the FNF-o -sensitive cells and measured with a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. 50 μl of 3-(4,5-dimethylthiazol-
2-yl)-2,5-diphenyltetrazolium bromide (Sigma; 2.5 ing/ml in phosphate-buffered saline) per well were added, and after 4 h of incubation at 37 0C, 50 μl of solubilization buffer (20% SDS, 66% dimethyl formamide, pH 4.7) were added, and absorbance (570 nm) was measured. Levels of TNF-α release were then calculated from a standard curve obtained using purified human rTNF-α (PeproTech, Inc., Rocky Hill, NJ). Interferon-γ released by activated T cells was measured by a sandwich enzyme-
linked immunosorbent assay. 5 x 104 γδ T cells/well were incubated with stimulus plus 25 units of
IL2/well in 100 μl of culture medium during 24 h at 37 0C. Then, 50 μl of supernatant were harvested for enzyme -linked immunosorbent assay using mouse monoclonal antibodies (BIOSOURCE, Camarillo, CA). A preferred assay for cytolytic activity is a 51Cr release assay. In exemplary assays, the cytolytic activity of γδ T cells is measured against autologous normal and tumor target cell lines, or control sensitive target cell lines such as Daudi and control resistant target cell line such as Raji in 4h 51Cr release assay. In a specific example, target cells were used in amounts of 2xlO3 cells/well and labeled with lOOμCi 51Cr for 60 minutes. Effector/Target (E/T) ratio ranged from 30: 1 to 3.75: 1. Specific lysis (expressed as percentage) is calculated using the standard Formula
[(experimental-spontaneous release / total-spontaneous release) xlOO].
The term phosphoantigen designates a T lymphocyte agonist, more particularly a Tγδ lymphocyte agonist, whose activity depends on the presence of a phosphate moiety. It has been formerly described (see Espinosa et al, Microbes and Infections 2001, or Belmant et al, Drug discovery today 2005) that such compounds have a unique specificity to trigger a γδ T cell response.
As discussed, the methods of the invention can generally be carried out with any γδ T cell activator that is capable of stimulating γδ T cell activity. This stimulation can be by direct effect on γδ T cells as discussed below using compounds that can stimulate γδ T cells in a pure γδ T cell culture, or the stimulation can be by an indirect mechanism, such as treatment with pharmacological agents such as bisphosphonates which lead to IPP accumulation. Preferably, a γδ T cell activator is a compound capable of regulating the activity of a γδ T cells in a population of γδ T cell clones in culture. The γδ T cell activator is capable of regulating the activity of a γδ T cell population of γδ T cell clones at millimolar concentration, preferably when the γδ T cell activator is present in culture at a concentration of less than 100 inM. Optionally a γδ T cell activator is capable of regulating the activity of a γδ T cell in a population of γδ T cell clones at millimolar concentration, preferably when the γδ T cell activator is present in culture at a concentration of less than 10 inM, or more preferably less than 1 mM. Regulating the activity of a γδ T cell can be assessed by any suitable means, preferably by assessing cytokine secretion, most preferably TNF-OC secretion as described herein. Methods for obtaining a population of pure γδ T cell clones is described in Davodeau et al, (J.Immunol.1993 (151)1214) and Moreau et al, (1986), the disclosures of which are incorporated herein by reference. Preferably the γδ T cell activator is capable of causing at least a 20%, 50% or greater increase in the number of γδ T cells in culture, or more preferably at least a 2-fold increase in the number of γδ T cells in culture.
In one embodiment, the γδ T cell activator may be a chemical compound capable of selectively activating Vγ9Vδ2 T lymphocytes. Selective activation of Vγ9Vδ2 T lymphocytes
indicates that the compound has a selective action towards specific cell populations, preferably increasing activation of Vγ9Vδ2 T cells at a greater rate or to a greater degree than other T cell types such as Vγl T cells, or not substantially not activation other T cell types. Such selectivity can be assessed in vitro T cell activation assays. Such selectivity, as disclosed in the present application, suggests that preferred compounds can cause a selective or targeted activation of the proliferation or biological activity of Vγ9Vδ2 T lymphocytes.
Detection of γδ T cell proliferation can be detected by standard methods. One specific method for detecting γδ T cell amplification in vivo is described in Example 1.
Preferred phosphoantigens In a preferred aspect, the γδ T cell activator may increase the biological activity of a γδ T cell, preferably increasing the activation of a γδ T cell, particularly increasing cytokine secretion from a γδ T cell or increasing the cytolytic activity of a cytotoxic γδ T cell, and/or stimulating the proliferation of a γδ T cell. Preferred γδ T cell activators include a composition comprising a compound of the Formula I, especially a γδ T cell activator according to Formula I, especially γδ T cell activator selected from the group consisting of BrHPP, CBrHPP, EpoxPP, HDMAPP, CHDMAPP,
NHDMAPP, H-angelylPP and H-tiglylPP. Most preferably, the γδ T cell activator is selected from the list consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP. However, it will appreciated that a number of phosphoantigen compounds that are less potent γδ T cell activators are available and may be used in accordance with the invention. For example, in one variant, a bisphosphonate compounds such as pamidronate (Novartis, Nuernberg, Germany) or zoledronate may be used. Other γδ T cell activators for use in the present invention are phosphoantigens disclosed in WO 95/20673, isopentenyl pyrophosphate (IPP) (US 5,639,653), the disclosures of the two preceding documents being incorporated herein by reference, as well as alkylamines (such as ethylamine, iso-propyulamine, n-propylamine, n-butylamine and iso-butylamine, for instance). Isobutyl amine and 3-aminopropyl phosphonic acid are obtained from Aldrich (Chicago, IL).
The γδ T cell activator is administered in an amount and under conditions sufficient to increase the activity γδ T cells in a subject, preferably in an amount and under conditions sufficient to increase cytokine secretion by γδ T cells and/or to increase the cytolytic activity of γδ T cells. In typical embodiments, a γδ T cell activator allows the cytokine secretion by γδ T cells to be increased at least 2, 3, 4, 10, 50, 100-fold, as determined in vitro.
In one aspect, a phosphoantigen according to the present invention comprises a compound of Formula (I):
Formula (I) wherein:
• Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including proton); • m is an integer from 1 to 3;
• B is O, NH, or any group capable to be hydrolyzed;
• Y represents O"Cat+, a Ci-C3 alkyl group, a group -A-R, or a radical selected from the group consisting of a nucleoside, an oligonucleotide, a nucleic acid, an amino acid, a peptide, a protein, a monosaccharide, an oligosaccharide, a polysaccharide, a fatty acid, a simple lipid, a complex lipid, a folic acid, a tetrahydrofolic acid, a phosphoric acid, an inositol, a vitamin, a co-enzyme, a flavonoid, an aldehyde, an epoxyde and a halohydrin;
• A is O, NH, CHF, CF2 or CH2; and,
• R is a linear, branched, or cyclic, aromatic or not, saturated or unsaturated, Ci-C20 hydrocarbon group, optionally interrupted by at least one heteroatom, wherein said hydrocarbon group comprises an alkyl, an alkylenyl, or an alkynyl, preferably an alkyl or an alkylene, which can be substituted by one or several substituents selected from the group consisting of: an alkyl, an alkylenyl, an alkynyl, an epoxyalkyl, an aryl, an heterocycle, an alkoxy, an acyl, an alcohol, a carboxylic group (-C00H), an ester, an amine, an amino group (-NH2), an amide (-CONH2), an imine, a nitrile, an hydroxyl (-OH), a aldehyde group (- CHO), an halogen, an halogenoalkyl, a thiol (-SH), a thioalkyl, a sulfone, a sulfoxide, and a combination thereof. Most preferably, said phosphoantigen compound are γδ T cell activators.
In a particular embodiment of any of the Formulas disclosed for use in accordance with the invention, the substituents as defined above are substituted by at least one of the substituents as specified above.
Preferably, the substituents are selected from the group consisting of: an (Ci-C6)alkyl, an (C2- Ce)alkylenyl, an (C2-Ce)alkynyl, an (C2-Ce)epoxyalkyl, an aryl, an heterocycle, an (Ci-C6)alkoxy, an (C2-Ce)acyl, an (Ci-C6)alcohol, a carboxylic group (-C00H), an (C2-Ce)ester, an (Ci-C6)amine, an amino group (-NH2), an amide (-CONH2), an (Ci-C6)imine, a nitrile, an hydroxyl (-OH), an aldehyde group (-CH0), an halogen, an (d-C6)halogenoalkyl, a thiol (-SH), a (d-C6)thioalkyl, a (Cr C6)sulfone, a (Ci-C6)sulfoxide, and a combination thereof.
More preferably, the substituents are selected from the group consisting of: an (Ci-C6)alkyl, an (C2-C6)epoxyalkyl, an (C2-C6)alkylenyl, an (d-C6)alkoxy, an (C2-C6)acyl, an (d-C6)alcohol, an (C2-C6)ester, an (Ci-C6)amine, an (Ci-C6)imine, an hydroxyl, a aldehyde group, an halogen, an (Ci- C6)halogenoalkyl and a combination thereof. Still more preferably, the substituents are selected from the group consisting of: an (C3-
C6)epoxyalkyl, an (Ci-C3)alkoxy, an (C2-C3)acyl, an (Ci-C3)alcohol, an (C2-C3)ester, an (Ci-C3)amine, an (Ci-C3)imine, an hydroxyl, an halogen, an (Ci-C3)halogenoalkyl, and a combination thereof. Preferably, R is a (C3-C25)hydrocarbon group, more preferably a (C5-Ci0)hydrocarbon group.
In the context of the present invention, the term "alkyl" more specifically means a group such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, tridecyl, tetradecyl, pentadecyl, hexadecyl, heptadecyl, octadecyl, nonadecyl, eicosyl, heneicosyl, docosyl and the other isomeric forms thereof. (Ci-C6)alkyl more specifically means methyl, ethyl, propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, hexyl and the other isomeric forms thereof. (Ci-C3)alkyl more specifically means methyl, ethyl, propyl, or isopropyl. The term "alkenyl" refers to an alkyl group defined hereinabove having at least one unsaturated ethylene bond and the term "alkynyl" refers to an alkyl group defined hereinabove having at least one unsaturated acetylene bond. (C2-Ce)alkylene includes a ethenyl, a propenyl (1-propenyl or 2-propenyl), a 1- or 2-methylpropenyl, a butenyl (1-butenyl, 2-butenyl, or 3-butenyl), a methylbutenyl, a 2-ethylpropenyl, a pentenyl (1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl), an hexenyl (1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl), and the other isomeric forms thereof. (C2-Ce)alkynyl includes ethynyl, 1-propynyl, 2-propynyl, 1-butynyl, 2-butynyl, 3-butynyl, 1- pentynyl, 2-pentynyl, 3-pentynyl, 4-pentynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4-hexynyl, or 5- hexynyl and the other isomeric forms thereof.
The term "epoxyalkyl" refers to an alkyl group defined hereinabove having an epoxide group. More particularly, (C2-C6)epoxyalkyl includes epoxyethyl, epoxypropyl, epoxybutyl, epoxypentyl, epoxyhexyl and the other isomeric forms thereof. (C2-C3)epoxyalkyl includes epoxyethyl and epoxypropyl.
The "aryl" groups are mono-, bi- or tri-cyclic aromatic hydrocarbons having from 6 to 18 carbon atoms. Examples include a phenyl, α-naphthyl, β-naphthyl or anthracenyl group, in particular. "Heterocycle" groups are groups containing 5 to 8 rings comprising one or more heteroatoms, preferably 1 to 5 endocyclic heteroatoms. They may be mono-, bi- or tri-cyclic. They may be aromatic or not. Preferably, and more specifically for R5, they are aromatic heterocycles. Examples of aromatic heterocycles include pyridine, pyridazine, pyrimidine, pyrazine, furan, thiophene, pyrrole, oxazole, thiazole, isothiazole, imidazole, pyrazole, oxadiazole, triazole, thiadiazole and triazine groups. Examples of bicycles include in particular quinoline, isoquinoline and quinazoline groups (for two 6- membered rings) and indole, benzimidazole, benzoxazole, benzothiazole and indazole (for a 6-
membered ring and a 5-membered ring). Nonaromatic heterocycles comprise in particular piperazine, piperidine, etc.
"Alkoxy" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -O- (ether) bond. (Ci-C6)alkoxy includes methoxy, ethoxy, propyloxy, butyloxy, pentyloxy, hexyloxy and the other isomeric forms thereof. (Ci-C3)alkoxy includes methoxy, ethoxy, propyloxy, and isopropyloxy.
"Alcyl" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -CO- (carbonyl) group. (C2-C6)acyl includes acetyl, propylacyl, butylacyl, pentylacyl, hexylacyl and the other isomeric forms thereof. (C2-C3)acyl includes acetyl, propylacyl and isopropylacyl. "Alcohol" groups correspond to the alkyl groups defined hereinabove containing at least one hydroxyl group. Alcohol can be primary, secondary or tertiary. (Ci-C6)alcohol includes methanol, ethanol, propanol, butanol, pentanol, hexanol and the other isomeric forms thereof. (Ci -C3) alcohol includes methanol, ethanol, propanol and isopropanol.
"Ester" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -COO- (ester) bond. (C2-C6)ester includes methylester, ethylester, propylester, butylester, pentylester and the other isomeric forms thereof. (C2-C3)ester includes methylester and ethylester.
"Amine" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -N- (amine) bond. (Ci-C6)amine includes methylamine, ethylamine, propylamine, butylamine, pentylamine, hexylamine and the other isomeric forms thereof. (Ci-C3)amine includes methylamine, ethylamine, and propylamine.
"Imine" groups correspond to the alkyl groups defined hereinabove having a (-C=N-) bond. (Ci-C6)imine includes methylimine, ethylimine, propylimine, butylimine, pentylimine, hexylimine and the other isomeric forms thereof. (Ci-C3)imine includes methylimine, ethylimine, and propylimine. The halogen can be Cl, Br, I, or F, more preferably Br or F.
"Halogenoalkyl" groups correspond to the alkyl groups defined hereinabove having at least one halogen. The groups can be monohalogenated or polyhalogenated containing the same or different halogen atoms. For example, the group can be a trifluoroalkyl (CF3-R). (Ci-C6)halogenoalkyl includes halogenomethyl, halogenoethyl, halogenopropyl, halogenobutyl, halogenopentyl, halogenohexyl and the other isomeric forms thereof. (Ci-C3)halogenoalkyl includes halogenomethyl, halogenoethyl, and halogenopropyl.
"Thioalkyl" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -S- (thioether) bond. (Ci-C6)thioalkyl includes thiomethyl, thioethyl, thiopropyl, thiobutyl, thiopentyl, thiohexyl and the other isomeric forms thereof. (Ci-C3)thioalkyl includes thiomethyl, thioethyl, and thiopropyl.
99 "Sulfone" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -SOO- (sulfone) bond. (C i-Cβ) sulfone includes methylsulfone, ethylsulfone, propylsulfone, butylsulfone, pentylsulfone, hexylsulfone and the other isomeric forms thereof. (Ci -C3) sulfone includes methylsulfone, ethylsulfone and propylsulfone. "Sulfoxyde" groups correspond to the alkyl groups defined hereinabove bonded to the molecule by an -SO- (sulfoxide) group. (Ci -C6) sulfoxide includes methylsulf oxide, ethylsulf oxide, propylsulfoxide, butylsulfoxide, pentylsulfoxide, hexylsulfoxide and the other isomeric forms thereof. (Ci -C3)sulf oxide includes methylsulf oxide, ethylsulf oxide, propylsulfoxide and isopropylsulf oxide. "Heteroatom" denotes N, S, or O. "Nucleoside" refers to a compound composed of any pentose or modified pentose moiety attached to a specific position of a heterocycle or to the natural positions of a purine (9-position) or pyrimidine (1 -position) or to the equivalent position in an analog. The term nucleoside includes but is not limited to adenosine, thymine, uridine, cytidine and guanosine.
In a particular embodiment, the hydrocarbon group is a cycloalkylenyl such as a cyclopentadiene or a phenyl, or an heterocycle such as a furan, a pyrrole, a thiophene, a thiazole, an imidazole, a triazole, a pyridine, a pyrimidine, a pyrane, or a pyrazine. Preferably, the cycloalkylenyl or the heterocycle is selected from the group consisting of a cyclopentadiene, a pyrrole or an imidazole. In a preferred embodiment, the cycloalkylenyl or the heterocycle is substituted by an alcohol. Preferably, said alcohol is a (Ci-C3)alcohol. In another embodiment, the hydrocarbon group is an alkylenyl with one or several double bonds. Preferably, the alkylenyl group has one double bond. Preferably, the alkylenyl group is a (C3- Cio)alkylenyl group, more preferably a (C4-C7)alkylenyl group. Preferably, said alkylenyl group is substituted by at least one functional group. More preferably, the functional group is selected from the group consisting of an hydroxy, an (Ci-C3)alkoxy, an aldehyde, an (C2-C3)acyl, or an (C2-C3)ester. In a more preferred embodiment, the hydrocarbon group is butenyl substituted by a group -CH2OH.
Optionally, said alkenyl group can be the isoform trans (E) or cis (Z), more preferably a trans isoform (E). In a most preferred embodiment, the alkylenyl group is the (E)-4-hydroxy-3-methyl-2 -butenyl. In an other preferred embodiment, the alkylenyl group is an isopentenyl, a dimethylallyl or an hydroxydimethylallyl. In an additional embodiment, the hydrocarbon group is an alkyl group substituted by an acyl.
More preferably, the hydrocarbon group is an (C4-C7)alkyl group substituted by an (Ci-C3)acyl. In a further preferred embodiment, the phosphoantigen is of Formula (Ia):
(Ia)
in which:
• R4 is an halogenated (Ci-C3)alkyl, a (Ci-C3)alkoxy-(Ci-C3)alkyl, an halogenated (C2- C3)acyl or a (C1-C3)alkoxy-(C2-C3)acyl,
• R3 is (d-C3)alkyl group, • m is an integer from 1 to 3,
• n is an integer from 2 to 20,
• B represents O, NH, or any group capable to be hydrolyzed
• A represents O, NH, CHF, CF2 or CH2,
• Y represents O"Cat+, a Ci-C3 alkyl group, a group -A-R, or a radical selected from the group consisting of a nucleoside, an oligonucleotide, a nucleic acid, an amino acid, a peptide, a protein, a monosaccharide, an oligosaccharide, a polysaccharide, a fatty acid, a simple lipid, a complex lipid, a folic acid, a tetrahydrofolic acid, a phosphoric acid, an inositol, a vitamin, a co-enzyme, a flavonoid, an aldehyde, an epoxyde and a halohydrin;
• R is a linear, branched, or cyclic, aromatic or not, saturated or unsaturated, Ci-C50 hydrocarbon group, optionally interrupted by at least one heteroatom, wherein said hydrocarbon group comprises an alkyl, an alkylenyl, or an alkynyl, preferably an alkyl or an alkylene, which can be substituted by one or several substituents selected from the group consisting of: an alkyl, an alkylenyl, an alkynyl, an epoxyalkyl, an aryl, an heterocycle, an alkoxy, an acyl, an alcohol, a carboxylic group (-C00H), an ester, an amine, an amino group (-NH2), an amide (-CONH2), an imine, a nitrile, an hydroxyl (-OH), a aldehyde group
(-CH0), an halogen, an halogenoalkyl, a thiol (-SH), a thioalkyl, a sulfone, a sulfoxide, and a combination thereof. Most preferably, said phosphoantigen compound are γδ T cell activators, and
• Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including the proton).
Preferably, R4 is an halogenated methyl (-CH2-X, X being an halogen), an halogenated (C2- C3)acetyl, or (Ci -C3) alkoxy- acetyl. The halogenated methyl or acetyl can be mono-, di-, or tri- halogenated. More preferably, R4 is a CH2-X group, X represents a halogen atom. Preferably, X is selected from I, Br and Cl. Preferably, R3 is a methyl or ethyl group. More preferably, R3 is a methyl.
Preferably, B is O and A is O or CH2. Preferably, n is an integer from 2 to 10, or from 2 to 5. In a more preferred embodiment, n is 2. Preferably, m is 1 or 2. More preferably, m is 1. Preferably, Y is O"Cat+, or a nucleoside. More preferably, Y is O"Cat+.
In a most preferred embodiment, n is 2, R3 is a methyl and R4 is a halogenated methyl, more preferably a monohalogenated methyl, still more preferably a bromide methyl. In a particularly
preferred embodiment, n is 2, R3 is a methyl, R4 is a methyl bromide. In a most preferred embodiment, R is 3-(bromomethyl)-3-butanol-l-yl.
In another embodiment, R4 is a CH2-X group and A and B represent O.
wherein R3, X, n, m, Y and Cat+ have the aforementioned meaning. Preferably, R3 is a methyl. Preferably, n is 2. Preferably, X is a bromide.
In another embodiment R4 is a CH2-X group and B represents O and A represents CH2.
Preferably, R3 is a methyl. Preferably, n is 2. Preferably, X is a bromide.
In one preferred embodiment, a phosphoantigen comprises a compound of Formula (Ia3):
in which X is an halogen (preferably selected from I, Br and Cl), R3 is a methyl or ethyl group, Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including the proton), and n is an integer from 2 to 20. Preferably, R3 is a methyl. Preferably, n is 2. Preferably, X is a bromide.
In a most preferred embodiment, a phosphoantigen comprises a compound of Formula (A): OH O O
+
Br CH2- -(CH2)2-O— P O -O" ,x Cat
CH3 O" O" (A) BrHPP
Preferably x Cat+ is 1 or 2 Na+.
In another most preferred embodiment, a phosphoantigen comprises a compound of Formula
(B):
OH O O
Br CH2- ■ (CH2)2- CH2- -o- -O" ,x Cat+
CH3 O" O" (B) CBrHPP
Preferably x Cat+ is 1 or 2 Na+.
In a further preferred embodiment, the phosphoantigen is of Formula (Ib):
R5 is a methyl or ethyl group,
B represents O, NH, or any group capable to be hydrolyzed,
A represents O, NH, CHF, CF2 or CH2,
Y is O"Cat+, a nucleoside, or a radical -A-R, wherein R has the aforementioned meaning, and
Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including the proton).
Preferably, n is an integer from 2 to 10, or from 2 to 5. In a more preferred embodiment, n is 2. Preferably, R5 is a methyl. Preferably, Y is O"Cat+, or a nucleoside. More preferably, Y is O"Cat+. Preferably, A is O, NH or CH2. More preferably, B is O. Preferably, B is O. Preferably, m is 1 or 2. More preferably, m is 1.
For example, a phosphoantigen may comprise a compound of Formula (IbI) or (Ib2):
wherein R5, n, m, Y and Cat+ have the above mentioned meaning.
In another preferred embodiment, a phosphoantigen comprises a compound of Formula (Ib3):
in which R5 is a methyl or ethyl group, Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including the proton), and n is an integer from 2 to 20. Preferably, R5 is a methyl. Preferably, n is 2.
In another preferred embodiment, a phosphoantigen comprises a compound of Formula (C):
O" O" (C) EpoxPP
Preferably x Cat+ is 1 or 2 Na+.
In a further preferred embodiment, the phosphoantigen is of Formula (Ic):
R6, R7, and R8 represent, independently from each other, a hydrogen atom or a (Ci-C3)alkyl group,
R9 is an (C2-C3)acyl, an aldehyde, an (Ci-C3)alcohol, or an (C2-C3)ester,
W is -CH-, -N- or -C-R10,
A is O, NH, CHF, CF2 or CH2,
B represents O, NH, or any group capable to be hydrolyzed, m is an integer from 1 to 3,
Y is O"Cat+, a nucleoside, or a radical -A-R, wherein R has the aforementioned meaning.
Cat+ represents one (or several, identical or different) organic or mineral cation(s) (including the proton),
More preferably, R6 and R8 are a methyl and R7 is hydrogen. More preferably, R9 is -CH2-OH, -CHO, -CO-CH3 or -CO-OCH3. Optionally, the double-bond between W and C is in conformation trans (E) or cis (Z). More preferably, the double-bond between W and C is in conformation trans (E).
The group Y can permit the design of a prodrug. Therefore, Y is enzymolabile group which can be cleaved in particular regions of the subject. The group Y can also be targeting group. In a preferred embodiment, Y is O~Cat+, a group -B-R, or a radical selected from the group consisting of a nucleoside, a monosaccharide, an epoxyde and a halohydrin. Preferably, Y is an enzymolabile group.
Preferably, Y is O~Cat+, a group -B-R, or a nucleoside. In a first preferred embodiment, Y is O~Cat+. In a second preferred embodiment, Y is a nucleoside.
In a preferred embodiment, Cat+ is H+, Na+, NH4 +, K+, Li+, (CH3CH2)3NH+.
In a preferred embodiment, B is O or NH. More preferably, B is O.
In a preferred embodiment, A is O, NH or CH2.
In a preferred embodiment, m is 1 or 2. More preferably, m is 1.
In another example, a phosphoantigen comprises a compound of Formula (IcI) or (Ic2):
In a preferred embodiment, W is -CH-. Preferably, R6 and R7 are hydrogen. Preferably, R8 is a methyl. Preferably, R9 is -CH2-OH.
In another preferred embodiment, a phosphoantigen comprises a compound of Formula (D):
In yet another preferred embodiment, a phosphoantigen comprises a compound of Formula
(E):
(F):
In another example, phosphoantigen comprises a compound of Formula (Ic3):
Preferably, R6, R7 and R9 are hydrogen. Preferably, Ri0 is a methyl. Preferably, R8 is -CH2- OH. Preferably, A is CH2, NH or O. In a preferred embodiment, a phosphoantigen comprises a compound named (Z)-5-hydroxy-3- methylpent-3-enyl pyrophosphonate, of Formula G:
In further embodiments, the γδ T cell activator is a compound named (E)-5-hydroxy-3- methylpent-3-enyl pyrophosphonate, of Formula H:
Specific examples of compounds also include: (E)l-pyrophosphonobuta-l,3-diene; (E)I- pyrophosphonopenta-l,3-diene; (E)l-pyrophosphono-4-methylpenta-l,3-diene; (E,E)1- pyrophosphono-4,8 -dimethylnona- 1 ,3 ,7-triene; (E,E,E) 1 -pyrophosphono-4, 8 , 12-trimethyltrideca-
1,3,7,11-tetraene; (E,E)l-triphosphono-4,8-dimethylnona-l,3,7-triene; 4- triphosphono-2- methylbutene ; α,β -di- [3 -methylpent-3 -enyl] -pyrophosphonate ; 1 -pyrophosphono-3 -methylbut-2-ene ;
α, β-di- [3 -methylbut-2-enyl] -triphosphonate ; α, β -di- [3 -methylbut-2-enyl] -pyrophosphonate ; allyl- pyrophosphonate; allyl-triphosphonate; α, γ-di-allyl-pyrophosphonate; α,β-di-allyl-triphosphonate; (E,E)4-[(5'-pyrophosphono-6' -methyl -penta-2',4'-dienyloxymethyl)-phenyl]-phenyl-methanone; (E,E)4-[(5 ' -triphosphono-6' -methyl -penta-2' ,4' -dienyloxymethyl) -phenyl] -phenyl-methanone; (E,E,E)[4-(9'-pyrophosphono-2',6'-dimethyl-nona-2',6',8'-trienyloxymethyl)-phenyl]-phenyl- methanone; (E,E,E)[4-(9'-pyrophosphono-2' ,6' ,8' -trimethyl-nona-2' ,6' ,8' -trienyloxymethyl) -phenyl] - phenyl-methanone; 5-pyrophosphono-2-methypentene; 5-triphosphono-2-methypentene; α,γ-di-[4- methylpent-4-enyl] -triphosphonate ; 5 -pyrophosphono-2-methypent-2-ene ; 5 -triphosphono-2- methypent-2-ene; 9-pyrophosphono-2,6-dimethynona-2,6-diene; 9-triphosphono-2,6-dimethynona- 2,6-diene; α,γ-di-[4,8-dimethylnona-2,6-dienyl]-triphosphonate; 4-pyrophosphono-2-methybutene; 4- methyl-2-oxa-pent-4-enyloxymethylpyrophosphate; 4-methyl-2-oxa-pent-4- enyloxymethyltriphosphate; α,β-di-[4-methyl-2-oxa-pent-4-enyloxymethyl] -pyrophosphate; and α,γ- di-[4-methyl-2-oxa-pent-4-enyloxymethyl]-triphosphate.
In other particular embodiments, the phosphoantigen can be selected from the group consisting of: 3-(halomethyl)-3-butanol-l-yl-diphosphate; 3-(halomethyl)-3-pentanol-l-yl- diphsophate; 4-(halomethyl)-4-pentanol-l -yl-diphosphate; 4-(halomethyl)-4-hexanol-l -yl- diphosphate ; 5 -(halomethyl) -5 -hexanol- 1 -yl-diphosphate ; 5 -(halomethyl) -5 -heptanol- 1 -yl- diphosphate; 6-(halomethyl)-6-heptanol-l -yl-diphosphate; 6-(halomethyl)-6-octanol-l -yl- diphosphate; 7-(halomethyl)-7-octanol-l -yl-diphosphate; 7-(halomethyl)-7-nonanol-l -yl-diphosphate; 8-(halomethyl)-8-nonanol-l-yl-diphosphate; 8-(halomethyl)-8-decanol-l -yl-diphosphate; 9-
(halomethyl) -9 -decanol-1 -yl-diphosphate; 9-(halomethyl)-9-undecanol-l -yl-diphosphate; 10- (halomethyl) - 10-undecanol- 1 -yl-diphosphate ; 10-(halomethyl) - 10-dodecanol- 1 -yl-diphosphate ; 11- (halomethyl)-l 1-dodecanol-l -yl-diphosphate; 11 -(halomethyl) -11-tridecanol-l -yl-diphosphate; 12- (halomethyl) - 12-tridecanol- 1 -yl-diphosphate ; 12-(halomethyl) - 12-tetradecanol- 1 -yl-diphosphate ; 13- (halomethyl)- 13 -tetradecanol- 1 -yl-diphosphate; 13 -(halomethyl)- 13-pentadecanol- 1 -yl-diphosphate;
14-(halomethyl) - 14-pentadecanol- 1 -yl-diphosphate ; 14-(halomethyl) - 14-hexadecanol- 1 -yl- diphosphate; 15-(halomethyl)- 15-hexadecanol- 1 -yl-diphosphate; 15-(halomethyl)-l 5-heptadecanol- 1 - yl-diphosphate; 16-(halomethyl)-16-heptadecanol-l -yl-diphosphate; 16-(halomethyl)-16-octadecanol- 1 -yl-diphosphate ; 17-(halomethyl)- 17-octadecanol- 1 -yl-diphosphate; 17-(halomethyl)- 17- nonadecanol-1 -yl-diphosphate; 18-(halomethyl)-l 8 -nonadecanol-1 -yl-diphosphate; 18-(halomethyl)-
18-eicosanol-l -yl-diphosphate; 19-(halomethyl)-19-eicosanol-l-yl-diphosphate; 19-(halomethyl)-19- heneicosanol- 1 -yl-diphosphate; 20-(halomethyl)-20-heneicosanol- 1 -yl-diphosphate ; 20-(halomethyl)- 20-docosanol-l -yl-diphosphate; 21-(halomethyl)-21-docosanol-l-yl-diphosphate; and 21- (halomethyl)-21-tricosanol-l-yl -diphosphate. More particularly, the phosphoantigen can be selected from the group consisting of: 3-
(bromomethyl) -3 -butanol-1 -yl-diphosphate (BrHPP); 5-bromo-4-hydroxy-4-methylpentyl
pyrophosphonate (CBrHPP); 3-(iodomethyl)-3-butanol-l-yl-diphosphate (IHPP); 3-(chloromethyl)-3- butanol-1-yl-diphosphate (ClHPP); 3-(bromomethyl)-3-butanol-l-yl-triphosphate (BrHPPP); 3- (iodomethyl) -3 -butanol- 1 -yl-triphosphate (IHPPP) ; α,γ-di- [3 -(bromomethyl) -3 -butanol- 1 -yl] - triphosphate (diBrHTP); and α,γ-di-[3-(iodomethyl)-3-butanol-l-yl]-triphosphate (dilHTP). In another particular embodiment, the phosphoantigen can be selected from the group consisting of: 3,4-epoxy-3-methyl-l-butyl-diphosphate (Epox-PP); 3,4,-epoxy-3-methyl-l-butyl- triphosphate (Epox-PPP); α,γ-di-3,4,-epoxy-3-methyl-l -butyl -triphosphate (di-Epox-TP); 3,4-epoxy- 3-ethyl-l -butyl -diphosphate; 4,5-epoxy-4-methyl- 1 -pentyl-diphosphate; 4,5-epoxy-4-ethyl- 1 -pentyl- diphosphate; 5,6-epoxy-5-methyl-l-hexyl-diphosphate; 5,6-epoxy-5-ethyl-l-hexyl -diphosphate; 6,7- epoxy-6-methyl-l-heptyl -diphosphate; 6,7-epoxy-6-ethyl-l-heptyl-diphosphate; 7,8-epoxy-7-methyl-
1-octyl -diphosphate; 7,8-epoxy-7-ethyl-l-octyl-diphosphate; 8,9-epoxy-8-methyl-l-nonyl- diphosphate ; 8 ,9-epoxy- 8 -ethyl- 1 -nonyl-diphosphate ; 9,10-epoxy-9-methyl- 1 -decyl -diphosphate ; 9,10-epoxy-9-ethyl-l-decyl-diphosphate; 10,1 l-epoxy-10-methyl-l-undecyl-diphosphate; 10,11- epoxy-10-ethyl-l-undecyl-diphosphate; 11,12-epoxy-l 1-methyl-l-dodecyl-diphosphate; 11,12-epoxy- 11-ethyl-l-dodecyl-diphosphate; 12,13-epoxy-12-methyl-l-tridecyl-diphosphate; 12,13-epoxy-12- ethyl- 1 -tridecyl-diphosphate ; 13,14-epoxy- 13 -methyl- 1 -tetr adecyl-diphosphate ; 13,14-epoxy- 13- ethyl- 1 -tetradecyl-diphosphate ; 14,15 -epoxy- 14-methyl- 1 -pentadecyl-diphosphate ; 14,15 -epoxy- 14- ethyl- 1 -pentadecyl-diphosphate ; 15,16-epoxy- 15 -methyl- 1 -hexadecyl-diphosphate ; 15,16-epoxy- 15- ethyl- 1 -hexadecyl-diphosphate ; 16, 17-epoxy- 16 -methyl- 1 -heptadecyl-diphosphate ; 16, 17-epoxy- 16- ethyl- 1 -heptadecyl-diphosphate ; 17,18 -epoxy- 17-methyl- 1 -octadecyl-diphosphate ; 17,18 -epoxy- 17- ethyl- 1 -octadecyl-diphosphate ; 18, 19-epoxy- 18 -methyl- 1 -nonadecyl-diphosphate ; 18, 19-epoxy- 18- ethyl- 1 -nonadecyl-diphosphate ; 19 ,20-epoxy- 19-methyl- 1 -eicosyl-diphosphate ; 19 ,20-epoxy- 19- ethyl- 1 -eicosyl-diphosphate; 20,21 -epoxy-20-methyl- 1 -heneicosyl-diphosphate ; 20,21 -epoxy-20- ethyl- 1 -heneicosyl-diphosphate ; 21 ,22-epoxy-21 -methyl- 1 -docosyl-diphosphate ; and 21 ,22-epoxy-21 - ethyl- 1 -docosyl-diphosphate.
In a preferred embodiment, the phosphoantigen can be selected from the group consisting of: (E)-4-hydroxy-3-methyl-2-butenyl pyrophosphate (HDMAPP), (E)-5-hydroxy-4-methylpent-3-enyl pyrophosphonate (CHDMAPP) and (E)-5-hydroxy-3-methylpent-3-enyl pyrophosphonate (H- tiglylPP). These compounds may be produced according to various techniques known per se in the art, some of which being disclosed in PCT Publications nos. WO 00/12516, WO 00/12519, WO 03/050128, WO 02/083720 and WO 03/009855, the disclosures of which are incorporated herein by reference.
In one preferred embodiment, the phosphoantigen is a γδ T cell activator and is a compound described in any one of PCT publication nos. WO 00/12516, WO 00/12519, WO 03/050128, WO
02/083720, WO 03/009855, WO 05/054258, WO 06/103 568 and WO 07/039 635, the disclosures of
which Formulas and specific structures as well as synthesis methods are incorporated herein by reference. In another preferred embodiment, the phosphoantigen is a γδ T cell activator and is a compound selected from the group consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP.
Alternatively, although less potent in their functions as a γδ T cell activator, other activators for use in the present invention are phosphoantigens disclosed in WO 95/20673, isopentenyl pyrophosphate (IPP) (U.S. Patent No. 5,639,653) and 3-methylbut-3-enyl pyrophosphonate (C-IPP).
The disclosures of both references are incorporated herein by reference. Also encompassed are compounds that contain a phosphate moiety and act as γδ T cell inhibitors; one example is a compound disclosed in U.S. Patent No. 6,624,151 Bl, the disclosure of which is incorporated herein by reference.
Each of the foregoing references relating to compounds and their synthesis are incorporated herein by reference.
Dosage of preferred γδ T cell activators As discussed, preferred compounds are selected which increase the biological activity of γδ T cells, preferably increasing the activation or proliferation of γδ T cells, particularly increasing cytokine secretion from γδ T cells or increasing the cytolytic activity of γδ T cells, with or without also stimulating the expansion of γδ T cells. For example, a γδ T cell activator allows the cytokine secretion by γδ T cells to be increased at least 2, 3, 4, 10, 50, 100-fold, as determined in vitro. Cytokine secretion and cytolytic activity can be assessed using any appropriate in vitro assay, or those described herein.
Optionally, in another aspect, the present invention relates to methods for the treatment of a proliferative disease, an infectious disease or an autoimmune disease, where the γδ T cell activator, is of Formula I. As used herein, the expression "Formula I", designates all compounds derived from Formulas I: I, Ia, IaI, Ia2, Ia3, A, B, Ib, IbI, Ib2, Ib3, C, Ic, IcI, Ic2, Ic3, D, E, F, G and H. Preferably, the γδ T cell activator selected from the group consisting of BrHPP, CBrHPP, EpoxPP, HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP, is administered in an amount and under conditions sufficient to stimulate the expansion of the γδ T cell population in a subject, particularly to reach 30- 90% of total circulating lymphocytes, typically 40-90%, more preferably from 50-90%, or to induce an improved cytokine secretion, particularly to reach a 30-90%, more preferably a 50-90%, increase in cytokine release compared to the common cytokine secretion. Percentage of total circulating lymphocytes can be determined according to methods known in the art. A preferred method for determining the percentage of γδ T cells in total circulating lymphocytes is by flow cytometry, examples of appropriate protocols described in example 1.
In a further aspect, the present invention relates to an in vivo regimen for the treatment of proliferative disease, an infectious disease or an autoimmune disease, where a γδ T cell activator, especially a γδ T cell activator according to Formula I, especially γδ T cell activator selected from the group consisting of HDMAPP, CHDMAPP, NHDMAPP and H-tiglylPP, is administered to a warm- blooded animal, especially a human, in a dose that is higher (preferably at least 10%, 20%, 30% higher) than the single administration Efficient Concentration value giving half of the maximum effect (EC50) of γδ T cell biological activity or population expansion, or more preferably a dose that is at least 50%, or more preferably at least 60%, 75%, 85% or preferably between about 50% and 100% of the single administration Efficient Concentration value giving the maximum effect. It will be appreciated that the above dosages related to a group of compounds, and that each particular compound may vary in optimal doses, as further described herein for exemplary compounds. Nevertheless, compounds are preferably administered in a dose sufficient to significantly increase the biological activity of γδ T cells or to significantly increase the γδ T cell population in a subject. In preferred exemplary compounds, a compound of Formula I is administered in a dosage (single administration) between 1 μg/kg and about 100 mg/kg, more preferably between about 1 μg/kg and about 30 mg/kg. In the framework of the present invention, the expression "Formula I", designate all compounds derived from Formula I: I, Ia, IaI, Ia2, Ia3, A, B, Ib, IbI, Ib2, Ib3, C, Ic, IcI, Ic2, Ic3, D, E, F, G and H. In preferred exemplary compounds, a compound of Formula Ia, especially a compound of Formula A or B is administered in a dosage between about 1 mg/kg and about 100 mg/kg, preferably between about 10 mg/kg and about 80 mg/kg, more preferably between about 20 mg/kg and about 30 mg/kg. In preferred exemplary compounds, a compound of Formula Ic, especially, a compound of Formula D, E, F, G or H, is administered in a dosage between about 1 μg/kg and about 100 mg/kg, preferably between about 1 μg/kg and about 20 mg/kg, more preferably between about 10 μg/kg and about 5 mg/kg. Further detail on dosages and administration and examples of dose response experiments using γδ T cell activator in mice and primate models are provided in PCT Application WO 04/050 096, the disclosure of which is incorporated herein by reference.
Unless otherwise indicated, the dosages for administration to a warm blooded animal, particularly humans provided herein are indicated in pure form (anionic form) of the respective compound. Purity level for the active ingredient depending on the synthesis batch can be used to adjust the dosage from actual to anionic form and vice-versa.
It will be appreciated that the above dosages related to a group of compounds, and that each particular compound may vary in optimal doses, as further described herein for exemplary compounds. Nevertheless, compounds are preferably administered in a dose sufficient to significantly increase the biological activity of γδ T cells or to significantly increase the γδ T cell population in a
subject. Said dose is administered to the human by subcutaneous (s.c.) or intravenous (i.v.) route. Said dose is preferably administered to the human by subcutaneous (s.c.) route.
In another embodiment, said dose is preferably administered to the human by oral administration, in a solid or liquid dosage form, including but not limited to tablets, capsules, sachets, lozenges, powders, pills, or granules, and the solid dosage form can be, for example, a fast melt dosage form, chewable form, controlled release dosage form, lyophilized dosage form, delayed release dosage form, extended release dosage form, pulsatile release dosage form, mixed immediate release and controlled release dosage form, or a combination thereof.
Type I IFN
As indicated above, the method is based on the use of particular combinations of active agents, according to particular schedules. The invention more preferably uses a cytokine in combination with a γδ T cell activator, the cytokine being a type I IFN, preferably an interferon-α.
Interferons are natural proteins produced by the cells of the immune system of most vertebrates in response to challenges by foreign agents such as viruses, parasites and tumor cells.
Interferons belong to the large class of glycoproteins known as cytokines. Interferon- a
Interferon alpha-2a (RoferonA®) binds to its specific cell-surface receptor, resulting in the transcription and translation of genes whose protein products have antiviral, antiproliferative, anticancer, and immune modulating effects. Peginterferon alpha-2a (Pegasys®, Roche), is a covalent conjugate of the protein interferon alpha-2a produced by recombinant DNA technology in E. coli with bis(monomethoxypoly ethylene glycol). The product has been approved by the FDA in October 2002.
Interferon alpha 2b (IFN α 2b (IntronA®) has a similar mode of action. PEG-interferon alfa- 2b (Peglntron®, FDA approved January 2001) is a covalent conjugate of recombinant interferon alpha, subtype 2b, and polyethylene glycol (PEG), used as an antiviral and antineoplastic agent. The biological activity of this agent is derived from its interferon alpha-2b protein moiety. Interferons alpha bind to specific cell-surface receptors, leading to the transcription and translation of genes whose protein products mediate antiviral, antiproliferative, anticancer, and immune-modulating effects. The PEG moiety lowers the clearance of interferon alpha-2b, thereby extending the duration of its therapeutic effects, but may also reduce the interferon-mediated stimulation of an immune response. A standard treatment with peg IFNα 2b is from 50 to 150 μg per week. In an embodiment, the present invention provides a method to treat a subject with a low dose (e.g. a dose that is at least 10%, 20%, 30%, 40%, 50% below the dose of IFNα required in absence of co-administering the γδ T cell activator which is between 50 and 150μg per week). For example, IFNα low dose is comprised between 0.1 and 50 μg/week, more preferably between 0.1 and 20 μg/week.
IFNα is usually administered as a single subcutaneous injection, once a week for a period of time sufficient to obtain a response to the treatment. In most patients, the treatment lasts from 12 to 26 weeks. In an embodiment, the present invention provides a method to treat a subject for a shorter time period (e.g. a time period shorter than what is usually prescribed, i.e. a time that is at least 10%, 20%, 30%, 40%, 50% or more reduced compared to the time of treatment with IFNα required in the absence of co-administering the γδ T cell activator).
Treatment cycles - administration scheme
Administration of the composition according to the invention can be carried out in a number of ways. The γδ T cell activator and type I IFN can be administered concomitantly, conjointly sequentially.
When the γδ T cell activator and type I IFN are administered concomitantly, both compounds are administered at the same time or within a few minutes, i.e. one directly after the other. When the γδ T cell activator and type I IFN are administered conjointly, the compounds are administered during the same day within 1, 2, 3, 4, 5, 6, 7, 8, 12, 18, 24 hours. When the γδ T cell activator and type I IFN are administered sequentially, the compounds are administered within 1, 2, 3, 4, 6, 7 or more days, 1, 2, 3 or more weeks. In an embodiment, the γδ T cell activator and the type I IFN are administered at concomitantly.
The γδ T cell activator can be administered before or after the type I IFN. In an embodiment, the γδ T cell activator is administered after the beginning of the type I IFN treatment.
In an embodiment, the γδ T cell activator and the type I IFN can be administered many times, each administration of both compounds constituting a treatment cycle. Generally, a cycle comprises at least one administration of a γδ T cell activator and at least one administration of a type I IFN. In an embodiment, a cycle comprises one administration of a γδ T cell activator and successive administrations of a type I IFN. In an embodiment, each γδ T cell activator constitutes another treatment cycle.
Treatment cycles can be carried out in a number of ways. In an embodiment, the treatment cycle comprises a single administration of a γδ T cell activator, and one, two, three, four or more successive administrations of the type I IFN, each type I IFN administration occurring once a week. In an embodiment, the treatment cycle comprises a single administration of a γδ T cell activator and a single administration of a type I IFN.
The administration of the γδ T cell activator can take place once (single treatment cycle) or many times (many treatment cycle). Preferably, if multiple administrations of the γδT cell activator are provided, the administrations that give rise to a γδT cell proliferation are separated by a period of time sufficient to prevent "exhaustion" of the γδT cells. Exhaustion can be characterized by reduction
in ability to produce cytokines or to proliferate in response to the γδT cell activator, in comparison to that observed when the γδT cells are treated with a first dose or a preceding dose of the γδT cell activator. Generally, the γδT cell rate (number of γδT cells), is allowed to return to substantially basal rate prior to a second administration of the γδT cell activator compound. At least about one week, two weeks, three weeks or up to eight weeks and more are required for a patient's γδT cell rate to return to substantially basal rate.
In another aspect, the γδT cell activator is administered in multiple doses, the administration of successive doses of the γδT cell activator takes place at the beginning of a treatment cycles, which are separated by at least 1, 2, 3 or 4 6, 8, 12, 24, or 36 weeks. In preferred aspects, one first dose of a γδT cell activator is administered, and one or more (preferably at least two) further doses of γδT cell activator are administered in different treatment cycles.
COMBINATION WITH OTHER THERAPEUTIC TREATMENTS
According to another embodiment, the compositions according to the invention may further comprise another therapeutic agent, including agents normally utilized for the particular therapeutic purpose for which the γδ T cell activator is being administered. The additional therapeutic agent will normally be present in the composition in amounts typically used for that agent in a monotherapy for the particular disease or condition being treated. Such therapeutic agents include, but are not limited to, therapeutic agents used in the treatment of cancers, infectious and autoimmune diseases. When one or more agents are used in combination in a therapeutic regimen, there is no requirement for the combined results to be additive of the effects observed when each treatment is conducted separately. Although at least additive effects are generally desirable, any increased effect, for example an anti-viral effect, above one of the single therapies would be of benefit. Also, there is no particular requirement for the combined treatment to exhibit synergistic effects, although this is certainly possible and advantageous.
IL-2
In an embodiment, the invention uses further IL-2 in combination with a γδ T cell activator and type I IFN.
The interleukin-2 polypeptide may be of human of animal origin, preferably of human origin. It may comprise the sequence of a wild-type human (or animal) IL-2 protein, or any biologically active fragment, variant or analogue thereof, i.e., any fragment, variant or analogue capable of binding to an IL-2 receptor and of inducing activation of γδ T cells in the method of this invention.
The sequence of reference, wild-type human interleukin-2 proteins is available in the art, such as in Genbank, under accession numbers NP000577; AAK26665; PO1585; XP035511 for instance. The interleukin-2 is commercialized under the name Proleukin™ (aldesleukin). Proleukin™, which is described in its product information leaflet as follows: a human recombinant interleukin-2
product, is a highly purified protein with a molecular weight of approximately 15 300 daltons. The chemical name is des-alanyl-1, serine- 125 -human interleukin-2. Proleukin™ is a lymphokine, produced by recombinant DNA technology using a genetically engineered E. CoIi strain containing an analog to the human interleukin-2 gene. Genetic engineering techniques were used to modify the human IL -2 gene, and the resulting expression clone encodes a modified human interleukin-2. The recombinant forms differs from native interleukin-2 in the following ways: a) Proleukin™ is not glycosylated because it is derived from E. CoIi; b) the molecule has no N-terminal alanine; the codon for this amino acid was deleted during the genetic engineering procedure; c) the molecule has serine substituted for cysteine at amino acid position 125; this was accomplished by site specific manipulation during the genetic engineering procedure; and d) the aggregation state of Proleukin™1 is likely to be different from that of native interleukin-2. Proleukin™1 biological potency is determined by a lymphocyte proliferation assay and is expressed in international units (IU) as established by the World Health Organization 1st international strand for interleukin-2 (human). The relationship between potency and protein mass is as follows: 18 MIU Proleukin™1 = 1.1 mg protein. The dose of IL-2 to be administered is generally expressed per square meter. The body surface of a human subject lies between 1.5 to 2.1 m2 depending on the size and weight of the subject. The body surface area (BSA) can be determined using different Formulas such as the Mosteller Formula or the Dubois and Dubois Formula.
The Mosteller Formula BSA (m2) = ([Height(cm) x Weight(kg)]/ 3600 )'/2 The DuBois and DuBois2 Formula BSA (m2) = 0.20247 x Height(m)0725 x Weight(kg)0425
The average body surface is of about 1.7 m2.
The IL-2 polypeptide is preferably administered by injections of between 7 and 9 MIU total in human daily, over a period of 1 to 10 days. Preferably, daily doses of 7.5 to 8.5 MIU total in human daily, most preferably doses of 8 MIU total in human daily are being administered. With respect to the body surface area equivalence and the variability, the IL-2 polypeptide can be administered for example by injections of between 3.3 and 6 MIU/m2 daily, over a period of 1 to 10 days. Preferably, daily doses from 3.5 to 5.5 MIU/m2 are being administered.
The preferred dose for IL-2, expressed in international units, can be transposed to any available or commercial interleukin-2. In the present inventions, the inventors have used the commercial interleukin-2 polypeptide Proleukin™1 commercialized by Chiron. It will be appreciated that any other interleukin-2 may be used, as long as the quantity of product to be administered is adjusted according to the dosage of the IL-2 chosen. Such dosage is easily carried on by the skilled artisan using standard analytical methods (see Hammerling et al, J. Pharm. And Biomed. Anal 1992, p. 547-553). IL-2 is preferably administered conjointly with the γδ T cell activator. In an embodiment, IL-2 is administered more than once, daily, for at least 2, 3, 4, 5, 6 or more days after the first IL-2
administration. Dosages of IL-2 are proposed in PCT publication N° 2004/020096 incorporated by reference, especially page 30 to 33 "Co-treatment with cytokine". Ribavirin
In an embodiment, the invention further comprises administering ribavirin with type I IFN , preferably IFNα, and the γδ T cell activator. Ribavirin (Copegus; Rebetol; Ribasphere;
Vilona,Virazole, also generics from Sandoz, Teva, Warrick) is an anti-viral drug which is active against a number of DNA and RNA viruses. It is a member of the nucleoside antimetabolite drugs that interfere with duplication of viral genetic material. Though not effective against all viruses, ribavirin is remarkable as a small molecule for its wide range of activity, including important activities against influenzas, flavi viruses and agents of many viral hemorrhagic fevers.
Viral inhibiting enzymes
Other compounds can also be used in combination with the treatment according to the invention. Such compounds include: protease inhibitors (such as ITMN-191, InterMune & Roche; MK-7009, Merck; BI12202, Boehringer; Telaprevir™, VX-950, N3S/4A inhibitor, Vertex Pharmaceuticals; Boceprevir, SCH-503034, NS3/4A inhibitor, Schering-Plough; TMC435350,
Tibotech & Medivir, NS3/41 protease inhibitor BILN 2061, NS3/4A protease inhibitor ACH 806, valopicitabine, HCV 796), nucleoside polymerase inhibitors (such as: MK-0608, Merck ; R7128, Pharmasset & Roche ; PSI-6130, Pharmasset ; Rl 626, NS5B RNA polymerase inhibitor, Roche), cyclophilin inhibitors (such as DEBIO-025, Debiopharm; NIM-811, Novartis), nucleoside and nucleotide reverse transcriptase inhibitors (NRTI), non-nucleoside reverse transcriptase inhibitors
(nNRTI), integrase inhibitors (i.e. raltegravir), entry inhibitors (or fusion inhibitors, i.e. maraviroc, enfuvirtide), maturation inhibitors (i.e. bevirimat, vivecon), other immunomodulating agents such as TLR7 agonists (ANA 975, Anadys).
Other antivirals agents such as, but not limited to lamivudine, adefovir, tenofovir and entecavir, ritonavir, l'indinavir, nelfinavir, saquinavir, l'amprenavir and lopinavir can also be administered in the combination according to the present invention. Some of those compounds in development are listed in the publication Manns et al, Nature Drug Discovery 2007: 991-1000. Chemotherapeutic compounds - Anticancer agents
According to the present invention, the treatment comprises an additional step of administration of an anti-cancer agent. Such anti-cancer agents include, but are not limited to:
• spindle poisons such as mebendazole, colchicine;
• mitotic inhibitors (taxanes (paclitaxel (Taxol), docetaxel (Taxotere); vinca alkaloids (i.e. vincristine, vinblastine, vinorelbine, vindesine));
• cytotoxic/antitumor antibiotics: such as anthracyclines (i.e. doxorubicin, daunorubicin, adriamycine, idarubicin, epirubicin and mitoxantrone, valrubicin), streptomyces (i.e. actinomycin, bleomycin, mitomycin, plicamycin);
• anti-metabolites (such as pyrimidine analogues (i.e. fluoropyrimidines analogs, 5-fluorouracil
(5-FU), floxuridine (FUDR), Cytosine arabinoside (Cytarabine), Gemcitabine (Gemzar®), capecitabine; purine analogues (i.e. azathioprine, mercaptopurine, thioguanine, fludarabine, pentostatin, cladribine, capecitabine, clofarabine); folic acid analogues (i.e. methotrexate, folic acid, pemetrexed, aminopterin, raltitrexed, trimethoprim, pyrimethamine);
• topoisomerase inhibitors (i.e. camptothecins: irinotecan, topotecan, amsacrine, etoposide, etoposide phosphate, teniposide);
• alkylating agents (i.e. busulfan, cisplatin, oxaliplatin, BBR3464, carboplatin, chlorambucil, cyclophosphamide, aldophosphamide, ifosfamide, fotemustine, lomustine, streptozocin, dacarbazine, procarbazine, temozolomide, thioTEPA, Uramustine, mechlorethamine (nitrogen mustard), phosphor amide mustard, melphalan (Akeran®), temozolomide);
• DNA methyltransferase inhibitors: 2'-deoxy-5-azacytidine (DAC), 5-azacytidine, 5-aza-2'- deoxycytidine, l-β-D-arabinofuranosyl-5-azacytosine, dihydro-5-azacytidine;
• vascular disrupting agents, such as flavone acetic acid derivatives, 5,6-dimethylxanthenone-4- acetic acid (DMXAA) and flavone acetic acid (FAA);
• also other chemotherapeutic drugs such as aprepitant, bortezomib (Velcade®, Millenium Pharmaceuticals), imatinib mesylate (Gleevec®), carmustine (BCNU), lomustine (CCNU), tamoxifen, gefitinib, erlotinib, carboxyamidotriazole, efaproxiral, tirapazamine, xcytrin, thymalfasin, vinflunine.
EXAMPLES
Example 1: Flow cytometry detection of γδ T cell amplification
Blood samples (4ml) were withdrawn into EDTA containing tubes. Tubes were shipped overnight at room temperature (RT) before flow cytometry analyses. Peripheral lymphocytes were analyzed by flow cytometry on total blood, after triple staining with anti-Vδ2FITC, anti-CD3PE and anti-CD25PC5 antibodies (V52-FITC: IMMU389 clone, Immunotech-Beckman-Coulter, Marseilles, France; CD3-PE: UCHTl clone, Immunotech-Beckman- Coulter; CD25PC5: M-A251 clone, Becton Dickinson, Le Pont de Claix, France).
Briefly, lOOμl patient blood was incubated 15 min at RT with lOμl anti-Vdelta2-FITC, lOμl anti-CD3-PE and lOμl anti-CD25PC5 antibodies. Antibodies were washed with 3ml IX PBS, centrifuged for 4 min at 1300rpm at RT and supernatant was discarded. Red cells were lysed with the
OptiLyse B reagent (Immunotech-Beckman-Coulter, Marseilles, France) according to the manufacturer's instructions. At the final step, stained white blood cells were recovered by centrifugation and resuspended in 300μl IX PBS + 0.2% PFA. Flow cytometry analysis on viable cells was performed on a FacsSCAN apparatus (Becton Dickinson) with the FacsDiva software.
Example 2: Dose response potentiation effect of cytokine secretion (i.e. IFNγ) by CHDMAPP activated γδ T cells in the presence of IFNα.
Expansion of purified γδ T-cells (thereafter called "bulk γδ"): Vγ9δ2 T lymphocytes were purified and expanded according to described methods {Espinosa et al, J. Biol Chem 2001). The cells are then frozen.
Stimulation of bulk γδ by CHDMAPP, the cells have been thawed and incubated in γδ medium 2 hours at 37°C, then centrifuged lOmin 1400rpm, resuspended and counted. Purified γδ (0.3 x 106 in 1 ml : 24 wells) were stimulated 24h with:
■ increasing doses of CHDMAPP.
■ increasing doses of CHDMAPP + a fixed dose of 10 IU of IFNa2B-
■ increasing doses of CHDMAPP + a fixed dose of 100 IU of IFNa2B-
■ increasing doses of CHDMAPP + a fixed dose of 1000 IU of IFNα2B . After 24h supernatants were centrifuged (1500rpm lOmin) and dosed by ELISA.
Results: The cells cultured with IFNα show a higher potential for IFNγ secretion. The more IFNα, the higher the secretion of cytokines.
Table 1: Calculated EC50 and Cmax for IFNγ release
The quantity of IFNα added in the medium does not significantly modify the EC50, which is similar for all settings, whereas the Cmax is increased, so one can assume that it is not the release potential of each individual γδ T cell that increases but rather that, upon IFNα stimulation, more γδ T cell become sensitive to CHDMAPP treatment, thereby increasing the cytokine release (i.e. here, IFNγ). These results underline that IFNα is able to sensitize γδ T cells that are not or weakly responding to CHDMAPP stimulation. Co-treatment with a γδ T cell activator and a type I IFN can thus result in further activation and cytokine release by γδ T cells responding only weakly to a treatment.
Example 3: Viral inhibition induced by CHDMAPP stimulated γδ T cells on a Replicon model.
The results shown in Figure 2 and 3 have been obtained using the hepatoma cell line Huh7, a subclone derived from cell line 9-13 expressing HCV genotype Ib replicon I377/NS3-3' (accession No. AJ242652). This HCV subgenomic replicon, stably maintained in Huh7 hepatoma cells, was created by Lohmann and al. (Lohmann V, Korner F, Koch JO, Herian U, Theilmann L and
Bartenschlager R. 1999. Science. 285: 110-113.). The replicon-containing cell culture, designated
GS4.3, was obtained from Dr. Christoph Seeger of the Institute for Cancer Research, Fox Chase Cancer Center, Philadelphia, Pennsylvania (Zhu Q, Guo JT and Seeger C. 2003. Replication of Hepatitis C virus subgenomic in nonhepatic epithelial and mouse hepatoma cells. J. Virol. 77(17): 9204-10.).
Experimental procedure:
In vitro PBMC setting: Culture of Huh7 cells: Huh7 cells are maintained at 37°C, 5% CO2, in DMEM supplemented with Lglutamine 2mM, non-essential amino acids, 10% Fetal Bovine Serum (FBS) and 500μg/ml geneticin. Cells are sub-divided 1:3 or 1:4 every 2-3 days.
24hr prior to the assay, Huh7 cells are collected, counted, plated in 96-well plates at 7500 cells/well in lOOμl standard maintenance medium and incubated in the conditions above.
To initiate the assay, culture medium is removed; cells are washed once with PBS. For control compounds only, 90μl assay medium (DMEM, Lglutamine, NEAA, 10% FBS) are added. Application of the control compounds and γδ Bulk supernatants: Supernatants of γδ bulk (stimulated or not with CHDMAPP) and IFNγ are added to duplicate wells and single wells respectively within the following range : 0.03, 0.1, 0.3, 1, 3, 10 and 30 IU/ml. Plates are rocked to mix, and incubated for 72 hr.
Ex vivo NHP setting: Plasma of cynomolgus was withdrawn a few hours (4 to 5 hours) after CHDMAPP treatment and was contacted with the replicon model described above where viral replication was measured. The sera were diluted and dosed for their content in IFNγ. Table 2: IFNγ content and HCA RNA copies evolution in NHP sera
Predose After CHDMAPP treatment
NHPl Serum dilution - 1/30 1/20 1/10
IFNγ secretion (pg/ml) 1578 52.6 78.9 157.8
HCV RNA copies/ml 106 1.803 1.149 0.706 0.364
Predose After CHDMAPP treatment
NHP2 Serum dilution - 1/10 1/20 1/30
IFNγ secretion (pg/ml) 2197 73.2 109.9 219.7
HCV RNA copies/ml 106 1.477 0.209 0.178 0.068
Quantification of HCV levels: Quantification is done by RT-PCR for assays #1 and #2 or Neomycin phosphotransferase II Elisa kit, purchased from AGDIA (PSP 73000). HCV RNA levels are measured by using TaqMan RT-PCR. Total cellular RNA is isolated and amplified by using ABBOTT RealTime HCV assay (Abbott mlOOOTM Automated Sample Preparation System and Abbott m2000rtTM instrument for reverse transcription, PCR amplification, and detection/quantitation,
Abbott Molecular Inc., Des Plaines, IL). Results are reported in International Units/mL (IU/mL), and
1 IU/ml = 4.3 copies/ml.
Results: Using increasing doses of CHDMAPP-stimulated γδ T cells an inhibition of RNA replication is observed. This inhibition is dose dependently related to the amount of CHDMAPP used to stimulate γδ T cells. The concentration of CHDMAPP at which the HCV RNA level in the replicon cells is reduced by 50% (IC50) is 1.68 nM. This low concentration underlines the efficiency of CHDMAPP for the inhibition of HCV replication in this model.
The content in IFNy has been dosed in supernatants of CHDMAPP stimulated γδ T cells in vitro, and values have been reported on the right axis. This IFNy axis also underlines that the production of cytokine by γδ T cells is essential for the replication inhibition.
Regarding the NHP experiment, it is noticeable that for an equivalent amount of plasmatic IFNy, the level of inhibition between the two NHP is different, underlining that the inhibitory effect on viral replication of CHDMAPP is not only IFNy concentration dependent (i.e. dilution 1/20 for NHPl versus dilution 1/30 for NHP2).
Example 4: NHP experiment on cytokine release upon repeated treatment with CHDMAPP +/-
IFNα
Experimental procedure: 8 naive cynomolgus macaque (M. fascicularis) (purchased from Noveprim) have been sorted in 2 groups. Group 1: 4 animals have been treated with CHDMAPP alone, 3 mg/kg (subcutaneous s.c. injection).
Group 2: 4 animals have been treated with CHDMAPP 3 mg/kg (s.c.) and IFNα (Pegasys®), 27 μg/administration (s.c).
Both groups are treated three times at one week interval that is to say at day 0, 7 and 14. Both compounds are administered conjointly (possibly at the same time or quickly one after the other). Blood samples have been collected at pre-dose, 30 min, 1 hour, 2, 3, 4, 5, 6, 8 and 24 hours after each administration. Serums are collected after centrifugation and cytokines are dosed. ILl-β, IL-6, MCP-I were analyzed by electrochemoluminescence (Sector 2400 apparatus), IFNy and TNFα were dosed by
ELISA.
Results: The cytokine release in CHDMAPP + IFNα treated animals is improved compared to CHDMAPP treated animals, especially cytokine exhaustion seems more moderate in CHDMAPP +
IFNα treated groups at the second cycle of treatment.
Example 5: NHP experiment on γδ T cell and Treg population in NHP upon repeated treatment with CHDMAPP, IL-2 +/- IFNα. Experimental procedure: 16 naive cynomolgus macaque (M. fascicularis) (purchased from Noveprim) have been sorted in 3 groups.
Table 3: Treatment of NHP according to groups
Administration of the three active compounds was made subcutaneously in one shot (bolus injection), concomitantly (i.e. one injection in a few minutes after each other) on the mentioned days. Blood samples were collected and analyzed for γδ T cell, Treg number and IFNγ content in the supernatants according to known procedures (i.e. as set forth in example 1).
Results: Despite multiple CHDMAPP treatment, the γδ T cells show an enhanced amplification in CHDMAPP+IFNα+IL-2 treated NHP compared to CHDAMPP+IL-2 or IFNα alone treated NHP. Whereas classical γδ T cell amplification with CHDMAPP+IL-2 increases the number of Treg, in CHDMAPP+IFNα+IL-2 treated NHP, the Treg number is well controlled and remains acceptable upon multiple γδ T cell stimulation.
Multiple stimulation with CHDMAPP, IFNα and IL-2 leads to an enhanced cytokine secretion (here IFNγ), compared to CHDMAPP and IL-2 only treated NHP, whereas a single treatment with IFNα induces no remarkable cytokine secretion. This experiment underlines the long term potentiation of the cytokine secretion (here IFNγ) by γδ T cells upon repeated dosages of a γδ T cell activator IL-2 and IFNα, in a relevant in vivo model. This experiment also underlines repeated γδ T cell amplification upon repeated dosages of a γδ T cell activator, IL-2 and IFNα, in a relevant in vivo model.
Example 6: Intracellular staining of IFNα and TNFα of PBMC in different activation conditions.
Fresh PBMC of a donor (containing 2% γδ T cells in total PBMC) were incubated for 4 hours in 96-U well plates with the following conditions:
• Medium (dot plot 8A)
• CHDMAPP (200 nM) (dot plot 8B) • IFN-OC2B (1000 IU/ml) (dot plot 8C)
• CHDMAPP (200 nM) + IFN- α2B (1000 IU/ml) (dot plot 8D)
Intracellular staining was performed with IFNγ-PE / TNFα-APC/ Vd2 FITC on a FACSCanto™. The results of staining have been gated on γδ T cells and γδ T cells represented for their content in IFNα and TNFα. The dot plots corresponding to each condition are enclosed as Figure 8. Table 4: Percentage of γδ T cells containing both IFNα and TNFα
Activation of γδ T cells in medium or with type I IFN alone is negligible. Activation of γδ T cells with a γδ T cell activator, in particular CHDMAPP is moderate. On the contrary, activation of γδ T cells with a γδ T cell activator and a type I IFN is drastically enhanced compared to other conditions. This experiment underlines that the activation of γδ T cells is synergistically enhanced when γδ T cells are activated by both a γδ T cell activator and a type I IFN.
Example 7: Crossed dose response effect of cytokine secretion (i.e. IFNγ) induced by activated γδ T cells using increasing amounts of CHDMAPP and increasing amounts of IFNα.
The experiment has been conducted similarly to example 2 using the following doses:
■ increasing doses of CHDMAPP: 0, 0.8, 2.5, 7.4, 22, 66.7, 200, 600 nM.
■ increasing doses of IFNa2B: 0, 4, 12, 37, 111, 333, 1000 IU/ml (in the legend square on the right of figure 9A).
The IFNγ released in IU/ml is represented in ordinates in figure 9A.
For a given IFNα dose, the IFNγ release is increased when the CHDMAPP dose increases. For a given CHDMAPP dose, the IFNγ release is increased when the IFNα dose increases.
Figure 9C is a graphical representation of the synergistic effect observed. Briefly, the theoretical effect of IFNα alone is calculated, the theoretical effect of CHDMAPP alone is calculated, these two effects are added to mimic a simple additive effect of IFNα and CHDMAPP ("additive effect"). Then this "additive effect" is subtracted from the obtained results. Figure 9C thus represents the synergistic effect obtained, e.g. the unforeseeable effect obtained on IFNγ secretion when IFNα and CHDMAPP as used together in combination. The obtained results clearly underline the synergy observed between
IFNα and a γδ T cell activator in terms of IFNγ secretion.
Example 8: Effect of CHDMAPP alone or in combination with interferon alpha on plasmatic cytokines release in cynomolgus monkey.
8 naive cynomolgus macaques have been treated with CHDMAPP with or without IFNα successively at day 0, 7 and 14. Group 1: CHDMAPP (s.c, 3 mg/kg)
Group 2: CHDMAPP (s.c, 3 mg/kg, IFNα2B (s.c, 27 μg/administration).
Blood was withdrawn pre-dose, 30 min, Ih, h, 3h, 4h, 5h, 6h, 8h, 24h for each administration and serum content of various cytokines (including IFNα and MCP-I) has been assessed by flow cytometry. Figure 1OA and 1OB report the secretion of IFNγ (Figure 10A) and MCP-I (Figure 10B) for each condition (CHDMAPP alone: light grey, dots, CHDMAPP+IFNα: dark grey, triangles). Figures 1OA and 1OB underline the fact that upon restimulation, the ability of γδ T cells to release cytokines is less impaired when the macaques are administered the combination of products rather than CHDMAPP alone. The combination treatment thus induces a more prolonged therapeutic effect.
All publications and patent applications cited in this specification are herein incorporated by reference in their entireties as if each individual publication or patent application were specifically and individually indicated to be incorporated by reference. Although the foregoing invention has been described in some detail by way of illustration and example for purposes of clarity of understanding, it will be readily apparent to one of ordinary skill in the art in light of the teachings of this invention that certain changes and modifications may be made thereto without departing from the spirit or scope of the appended claims.
Claims
1. A method for treating a disease comprising administering to a patient an effective amount of a γδ T cell activator in combination with an effective amount of a type I IFN.
2. The method according to claim 1 , wherein said γδ T cell activator is of formula I to II.
3. The method according to claim 1 or 2, wherein said γδ T cell activator is selected from the group consisting of A, B, C, D, E, F, G, and H.
4. The method according to any of the above claims, wherein said γδ T cell activator is administered at a dose of between about 0.1mg/kg to about 30 mg/kg.
5. The method according toany of the above claims, wherein said γδ T cell activator is administered subcutaneously.
6. The method according to claim 1, wherein said type I IFN is IFNα.
7. The method according to any of the above claims, wherein IFNα is administered at a dose of between about 0.5μg and about 150μg.
8. The method according to any of the above claims, wherein IFNα dose is reduced compared to standard treatment.
9. The method according to any of the above claims, wherein IFNα is administered subcutaneously.
10. The method according to any of the above claims, wherein said disease is selected from the group consisting of a proliferative disease, an infectious disease or an autoimmune disease.
11. The method according to any of the above claims, wherein said disease is a viral infection.
12. The method according to any of the above claims, wherein said disease is a HIV, HTLV-I, HPV, HHV.
13. The method according to any of the above claims, wherein said viral infection is chronic.
14. The method according to any of the above claims, wherein said chronic infection is HCV.
15. The method according to any of the above claims, wherein said disease is a co infection
16. The method according to any of the above claims, wherein said co infection is HCV/ AIDS or HCV/Tuberculosis.
17. The method according to any of the above claims, wherein said disease is a cancer.
18. The method according to any of the above claims, wherein said cancer is selected from Kaposi disease, NLH, myeloma, CML, hairy cell leukemia, neuro-endocrinous tumors.
19. The method according to any of the above claims, wherein said γδ T cell activator administration occurs once a week for about 3 weeks.
20. The method according to any of the above claims, wherein said IFNα administration occurs once a week for about 3 weeks.
21. The method according to any of the above claims, comprising further administering an antiviral agent.
22. The method according to any of the above claims, comprising further administering IL-2 at a low dose.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10968908P | 2008-10-30 | 2008-10-30 | |
| US61/109,689 | 2008-10-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2010049438A2 true WO2010049438A2 (en) | 2010-05-06 |
| WO2010049438A3 WO2010049438A3 (en) | 2010-12-02 |
Family
ID=41527839
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2009/064188 WO2010049438A2 (en) | 2008-10-30 | 2009-10-28 | Improved methods of using phosphoantigens for the treatment of diseases |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2010049438A2 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2012123381A1 (en) * | 2011-03-11 | 2012-09-20 | Assistance Publique - Hôpitaux De Paris | Use of low dose il-2 for treating autoimmune - related or inflammatory disorders |
| WO2013034306A1 (en) * | 2011-09-08 | 2013-03-14 | Philip Morris Products S.A. | Reconstituted tobacco plug for a smoking article |
| US8466159B2 (en) | 2011-10-21 | 2013-06-18 | Abbvie Inc. | Methods for treating HCV |
| US8492386B2 (en) | 2011-10-21 | 2013-07-23 | Abbvie Inc. | Methods for treating HCV |
| US8809265B2 (en) | 2011-10-21 | 2014-08-19 | Abbvie Inc. | Methods for treating HCV |
| US8853176B2 (en) | 2011-10-21 | 2014-10-07 | Abbvie Inc. | Methods for treating HCV |
| US9669071B2 (en) | 2011-03-11 | 2017-06-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Method for stimulating T lymphocytes with IL-2 |
| WO2020243371A1 (en) * | 2019-05-28 | 2020-12-03 | Massachusetts Institute Of Technology | Methods and compositions for modulating immune responses |
| JP2021529786A (en) * | 2018-07-04 | 2021-11-04 | ユニバーシティ カレッジ カーディフ コンサルタンツ リミテッド | Phosphoantigen prodrug compound |
| US11192914B2 (en) | 2016-04-28 | 2021-12-07 | Emory University | Alkyne containing nucleotide and nucleoside therapeutic compositions and uses related thereto |
| WO2023275025A1 (en) * | 2021-06-28 | 2023-01-05 | Byondis B.V. | Conjugates comprising phosphoantigens and their use in therapy |
| US11926641B2 (en) | 2018-03-19 | 2024-03-12 | University Of Iowa Research Foundation | Phosphonamidate butyrophilin ligands |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5885957A (en) * | 1985-03-16 | 1999-03-23 | Glaxo Wellcome Inc. | Treatment of HTLV-I infections |
| WO2001022990A2 (en) * | 1999-09-27 | 2001-04-05 | Coley Pharmaceutical Group, Inc. | Methods related to immunostimulatory nucleic acid-induced interferon |
| WO2004050096A2 (en) * | 2002-12-02 | 2004-06-17 | Innate Pharma | Phosphoantigens for regulating an immune response |
-
2009
- 2009-10-28 WO PCT/EP2009/064188 patent/WO2010049438A2/en active Application Filing
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5885957A (en) * | 1985-03-16 | 1999-03-23 | Glaxo Wellcome Inc. | Treatment of HTLV-I infections |
| WO2001022990A2 (en) * | 1999-09-27 | 2001-04-05 | Coley Pharmaceutical Group, Inc. | Methods related to immunostimulatory nucleic acid-induced interferon |
| WO2004050096A2 (en) * | 2002-12-02 | 2004-06-17 | Innate Pharma | Phosphoantigens for regulating an immune response |
Non-Patent Citations (3)
| Title |
|---|
| BONNAFOUS CECILE ET AL: "V gamma 9V delta 2 T CELLS ACTIVATED BY IPH1201 (SYNTHETIC PHOSPHOANTIGEN) AS POSSIBLE IMMUNOTHERAPY FOR CHRONIC HCV INFECTION AND ITS RATIONALE FOR COMBINATION WITH IFN alpha" HEPATOLOGY, vol. 48, no. 4, Suppl. S, 24 September 2008 (2008-09-24), pages 1150A-1151A, XP008124036 ISSN: 0270-9139 * |
| STAGNO FABIO ET AL: "Successful treatment of granulocytic sarcoma with alpha-interferon and disodium pamidronate at presentation of chronic myeloid leukemia" BLOOD, vol. 96, no. 11 Part 2, 16 November 2000 (2000-11-16), page 258B, XP008124038 & 42ND ANNUAL MEETING OF THE AMERICAN SOCIETY OF HEMATOLOGY; SAN FRANCISCO, CALIFORNIA, USA; DECEMBER 01-05, 2000 ISSN: 0006-4971 * |
| VIEY EMILIE ET AL: "Phosphostim-activated gammadelta T cells kill autologous metastatic renal cell carcinoma" JOURNAL OF IMMUNOLOGY, vol. 174, no. 3, 1 February 2005 (2005-02-01), pages 1338-1347, XP002590675 ISSN: 0022-1767 * |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2012228418B2 (en) * | 2011-03-11 | 2016-09-29 | Assistance Publique - Hopitaux De Paris | Use of low dose IL-2 for treating autoimmune - related or inflammatory disorders |
| US10293028B2 (en) | 2011-03-11 | 2019-05-21 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of low dose IL-2 for treating autoimmune related or inflammatory disorders |
| WO2012123381A1 (en) * | 2011-03-11 | 2012-09-20 | Assistance Publique - Hôpitaux De Paris | Use of low dose il-2 for treating autoimmune - related or inflammatory disorders |
| EP2683395B1 (en) | 2011-03-11 | 2018-08-01 | Assistance Publique - Hôpitaux de Paris | Use of low dose il-2 for treating type 1 diabetes |
| EP3766513A1 (en) * | 2011-03-11 | 2021-01-20 | Assistance Publique Hôpitaux de Paris | Use of low dosage il-2 for treating vasculitis |
| US11559566B2 (en) | 2011-03-11 | 2023-01-24 | Inserm (Institut National De La Sante Et De La Recherche Medicale) | Use of low dose IL-2 for treating autoimmune-related or inflammatory disorders |
| JP2014510725A (en) * | 2011-03-11 | 2014-05-01 | アシスタンス ピュブリク−オピトー ドゥ パリ | Use of low dose IL-2 for the treatment of autoimmune related disorders or inflammatory disorders |
| US10765723B2 (en) | 2011-03-11 | 2020-09-08 | INSERM (Institut National de la Santé et de la Recherche Médicale | Use of low dose IL-2 for treating autoimmune—related or inflammatory disorders |
| EP3443979B1 (en) | 2011-03-11 | 2020-05-27 | Assistance Publique - Hôpitaux De Paris | Il-2 dosage regimen for treating systemic lupus erythematosus |
| AU2020204023B2 (en) * | 2011-03-11 | 2022-06-02 | Assistance Publique - Hopitaux De Paris | Use of low dose IL-2 for treating autoimmune-related or inflammatory disorders |
| EP3443979A1 (en) * | 2011-03-11 | 2019-02-20 | Assistance Publique - Hôpitaux De Paris | Il-2 dosage regimen for treating systemic lupus erythematosus |
| US9669071B2 (en) | 2011-03-11 | 2017-06-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Method for stimulating T lymphocytes with IL-2 |
| WO2013034306A1 (en) * | 2011-09-08 | 2013-03-14 | Philip Morris Products S.A. | Reconstituted tobacco plug for a smoking article |
| US8466159B2 (en) | 2011-10-21 | 2013-06-18 | Abbvie Inc. | Methods for treating HCV |
| US9452194B2 (en) | 2011-10-21 | 2016-09-27 | Abbvie Inc. | Methods for treating HCV |
| US8993578B2 (en) | 2011-10-21 | 2015-03-31 | Abbvie Inc. | Methods for treating HCV |
| US8969357B2 (en) | 2011-10-21 | 2015-03-03 | Abbvie Inc. | Methods for treating HCV |
| US8853176B2 (en) | 2011-10-21 | 2014-10-07 | Abbvie Inc. | Methods for treating HCV |
| US8809265B2 (en) | 2011-10-21 | 2014-08-19 | Abbvie Inc. | Methods for treating HCV |
| US8685984B2 (en) | 2011-10-21 | 2014-04-01 | Abbvie Inc. | Methods for treating HCV |
| US8680106B2 (en) | 2011-10-21 | 2014-03-25 | AbbVic Inc. | Methods for treating HCV |
| US8492386B2 (en) | 2011-10-21 | 2013-07-23 | Abbvie Inc. | Methods for treating HCV |
| US11192914B2 (en) | 2016-04-28 | 2021-12-07 | Emory University | Alkyne containing nucleotide and nucleoside therapeutic compositions and uses related thereto |
| US11926641B2 (en) | 2018-03-19 | 2024-03-12 | University Of Iowa Research Foundation | Phosphonamidate butyrophilin ligands |
| JP2021529786A (en) * | 2018-07-04 | 2021-11-04 | ユニバーシティ カレッジ カーディフ コンサルタンツ リミテッド | Phosphoantigen prodrug compound |
| WO2020243371A1 (en) * | 2019-05-28 | 2020-12-03 | Massachusetts Institute Of Technology | Methods and compositions for modulating immune responses |
| WO2023275025A1 (en) * | 2021-06-28 | 2023-01-05 | Byondis B.V. | Conjugates comprising phosphoantigens and their use in therapy |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2010049438A3 (en) | 2010-12-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2010049438A2 (en) | Improved methods of using phosphoantigens for the treatment of diseases | |
| Costantini et al. | A reappraisal of thymosin alpha1 in cancer therapy | |
| US8158606B2 (en) | β-L-2′-deoxynucleosides for the treatment of resistant HBV strains and combination therapies | |
| Rizza et al. | Role of type I interferon in inducing a protective immune response: perspectives for clinical applications | |
| US20150259645A1 (en) | Method for inducing il-2-free proliferation of gamma delta t cells | |
| US20230285509A1 (en) | Interferon-based disease treatment method | |
| US8921384B2 (en) | Spiro[2.4]heptanes for treatment of flaviviridae infections | |
| AU2016349685A1 (en) | Combination therapy of an HBV capsid assembly inhibitor and an interferon | |
| JP2018512428A5 (en) | ||
| US20230044958A1 (en) | Method of treating virus infection using a tlr7 agonist | |
| EP2739301B1 (en) | Hcv immunotherapy | |
| Sun et al. | Investigational drugs with dual activity against HBV and HIV | |
| Cholongitas et al. | Novel therapeutic options for chronic hepatitis C | |
| Guilhot et al. | Interferon in chronic myeloid leukaemia: past and future | |
| AU2012358994A1 (en) | Pharmaceutical compositions and methods for treating gastrointestinal infections and disorders | |
| Kast | Ribavirin in cancer immunotherapies: controlling nitric oxide augments cytotoxic lymphocyte function | |
| US20100189681A1 (en) | Methods of Using Phosphoantigens Together with Interleukin-2 for the Treatment of Cancer | |
| Borah et al. | Antiviral drugs and vaccines | |
| Billiau | Interferons: the pathways of discovery: II. Immunomodulatory, in vivo and applied aspects | |
| Anaya et al. | The Use of Interleukin‐2 in Human Immunodeficiency Virus Infection | |
| Masihi | C7 Anti-infective activity of immunomodulators | |
| Araújo et al. | Therapeutic perspectives for hepatitis C | |
| WO2013128022A1 (en) | Method for preventing or treating a hepatitis c virus infection or a hepatitis b virus infection by administration of suicide gene-modified lymphocytes | |
| US20140242029A1 (en) | Combination therapy for treating hcv infection in an hcv-hiv coinfected patient population | |
| NZ620689B2 (en) | Hcv immunotherapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09748750 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase in: |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 09748750 Country of ref document: EP Kind code of ref document: A2 |