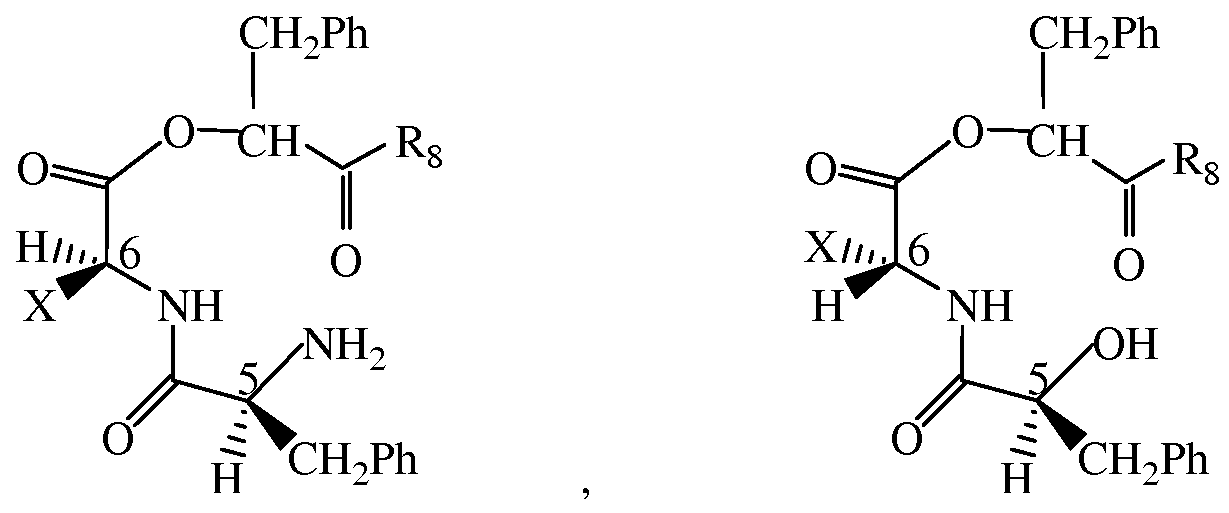WO2009099763A1 - Ester-based peptide prodrugs - Google Patents
Ester-based peptide prodrugs Download PDFInfo
- Publication number
- WO2009099763A1 WO2009099763A1 PCT/US2009/031593 US2009031593W WO2009099763A1 WO 2009099763 A1 WO2009099763 A1 WO 2009099763A1 US 2009031593 W US2009031593 W US 2009031593W WO 2009099763 A1 WO2009099763 A1 WO 2009099763A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino acid
- seq
- glucagon
- peptide
- group
- Prior art date
Links
- 0 CC(N*C(*)=O)=O Chemical compound CC(N*C(*)=O)=O 0.000 description 17
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/28—Insulins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/26—Glucagons
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Definitions
- Peptide-based drugs are highly effective medicines with relatively short duration of action and variable therapeutic index.
- the present disclosure is directed to peptide-based prodrugs wherein the prodrug derivative is designed to delay onset of action and extend the half life of the drug.
- the delayed onset of action is advantageous in that it allows systemic distribution of the prodrug prior to its activation. Accordingly, the administration of prodrugs eliminates complications caused by peak activities upon administration and increases the therapeutic index of the parent drug.
- Receptor recognition and subsequent processing of the peptide and protein agonists is the primary route of degradation of many peptide and protein-based drugs.
- prodrugs can be prepared to extend the peptide or protein's biological half life based on a strategy of inhibiting recognition of the prodrug by the corresponding receptor.
- the prodrugs disclosed herein will ultimately be chemically converted to structures that can be recognized by the receptor, wherein the speed of this chemical conversion will determine the time of onset and duration of in vivo biological action.
- the molecular design disclosed in this application relies upon an intramolecular chemical reaction that is not dependent upon additional chemical additives, or enzymes.
- the prodrug chemistry is broadly applicable to peptide and protein based drugs where an aliphatic hydroxyl group can be accommodated in the active site and when the chemically modified derivative yields a poorly active peptide or protein.
- a specific example to illustrate this point would be the formation of reversible serine esters, at the active site serine, in the family of serine proteases.
- Insulin is a spontaneous peptide hormone. It demonstrates unparalleled ability to lower glucose in virtually all forms of diabetes. Unfortunately, its pharmacology is not glucose sensitive and as such it is capable of excessive action that can lead to life-threatening hypoglycemia. Inconsistent pharmacology is a hallmark of insulin therapy such that it is extremely difficult to normalize blood glucose without occurrence of hypoglycemia.
- native insulin is of short duration of action and requires modification to render it suitable for use in control of basal glucose.
- Established approaches to delay the onset of insulin action include reduction in solubility, and albumin binding.
- Prodrug chemistry offers the opportunity to precisely control the onset and duration of insulin action after clearance from the site of administration and equilibration in the plasma at a highly defined concentration.
- Insulin is a two chain heterodimer that is biosynthetically derived from a low potency single chain proinsulin precursor through enzymatic processing.
- Human insulin is comprised of two peptide chains (an "A chain” (SEQ ID NO: 613) and "B chain” (SEQ ID NO: 614)) bound together by disulfide bonds and having a total of 51 amino acids.
- the C-terminal region of the B -chain and two terminal ends of the A-chain associate in three-dimensional structure to assembly a site for high affinity binding to the insulin receptor.
- the selective insertion of hydroxyl groups can be accommodated without loss in potency at multiple locations within this active site region. Chemical esterification of these active site hydroxyl groups with specific dipeptides would dramatically lessen activity and serve as suitable prodrugs.
- Pre-proglucagon is a 158 amino acid precursor polypeptide that is processed in different tissues to form a number of different progluc agon-derived peptides, including glucagon, glucagon- like peptide- 1 (GLP-I), glucagon- like peptide-2 (GLP-2) and oxyntomodulin (OXM), that are involved in a wide variety of physiological functions, including glucose homeostasis, insulin secretion, gastric emptying, and intestinal growth, as well as the regulation of food intake.
- GLP-I glucagon- like peptide- 1
- GLP-2 glucagon- like peptide-2
- OXM oxyntomodulin
- Glucagon is a 29-amino acid peptide (SEQ ID NO: 612 that corresponds to amino acids 33 through 61 of pre-proglucagon, while GLP-I is produced as a 37-amino acid peptide that corresponds to amino acids 72 through 108 of pre- proglucagon.
- GLP- 1(7-36) amide SEQ ID NO: 602; the C terminus is an arginine amide
- GLP- 1(7-37) acid SEQ ID NO: 601; C terminus is a glycine
- Glucagon is a life-saving medicine that is used in the acute treatment of severe hypoglycemia.
- Oxyntomodulin has been reported to have pharmacological ability to suppress appetite and lower body weight.
- Clinical studies with GLP-I like agonists have proven this family of peptides to be an effective treatment for Type II diabetes. In addition, it might be intrinsically safer than insulin therapy because of its glucose dependent action, thus eliminating the chances of hypoglycemia.
- Structure-activity relationship studies have shown that the N terminal histidine for each of these three peptides (glucagon, GLP- 1 and oxyntomodulin) is especially important for the full action and that N-terminally extended forms severely diminish biological potency.
- DPP-IV Dipeptidyl Peptidase IV
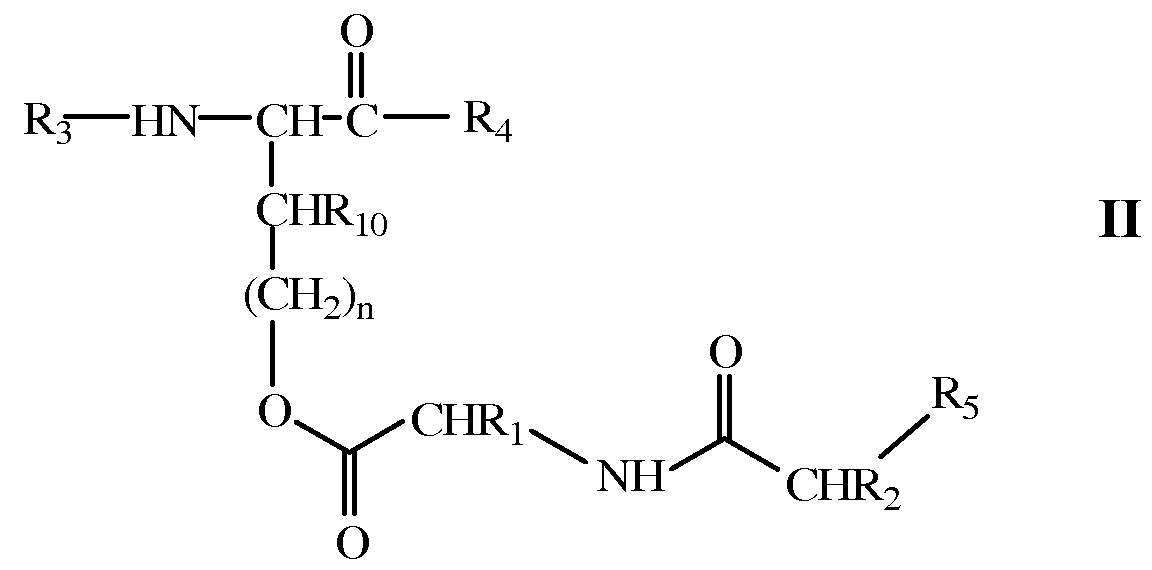
- a prodrug derivative of a bioactive polypeptide is prepared by covalently linking a dipeptide to the bioactive polypeptide via an ester linkage.
- the dipeptide is covalently bound to the bioactive polypeptide at a position that interferes with the bioactive polypeptide's ability to interact with its corresponding receptor or cofactor. Subsequent removal of the dipeptide, under physiological conditions and in the absence of enzymatic activity, restores full activity to the polypeptide.
- R 3 is selected from the group consisting of NH 2 , an amino acid sequence and
- R 4 is -OH, NH 2 or an amino acid sequence, Rio is selected from the group consisting of H, C]-C 4 alkyl, and (CH 2 ) n (C 6 -Cio aryl),
- W is C 6 -CiO aryl or a bond
- n is an integer from 0 to 3.
- Ri 2 is when R 3 is NH 2 or an amino acid sequence.
- R 3 or R 4 is an amino acid sequence of a bioactive peptide.
- R 3 is an amino acid sequence of the bioactive peptide which is N-terminal to an amino acid of the bioactive peptide comprising a side chain of structure
- Rl 2 and R 4 is an amino acid sequence of the bioactive peptide which is C-terminal to the amino acid of the bioactive peptide.
- R 7 is 0-amino acid
- R 7 is an amino acid sequence of the bioactive peptide which is N- terminal to an amino acid of the bioactive peptide comprising a side chain of structure
- Rl2 and R 4 is an amino acid sequence of the bioactive peptide which is C-terminal to the amino acid of the bioactive peptide.
- R 3 is and R 7 and R 4 represent amino acid sequences from glucagon, GLP-I or insulin peptides.
- R 3 is an N-terminal sequence selected from either glucagon, GLP-I or insulin peptides
- R 4 is a carboxy terminal sequence selected from either glucagon, GLP-I or insulin peptides
- Rj 2 is
- R 3 is H or the N-terminal amino acids of the bioactive protein (e.g., a glucagon, GLP-I or insulin peptide)
- R 4 represents OH or the C-terminal amino acids of the bioactive protein (e.g., a glucagon, GLP-I or insulin peptide)
- Rio is selected from the group consisting of H, C]-C 4 alkyl, and (CH 2 ) n (C 6 -Ci 0 aryl), and n is an integer from 0 to 3.
- Rj is selected from the group consisting of CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 aryl), and (C 0 -C 4 alkyl)(C 5 -C6 heteroaryl),
- R 2 is selected from the group consisting of (C 0 -C 4 alkyl)C]-C 4 alkyl, (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 aryl), and (C 0 -C 4 alkyl)(C 5 -C 6 heteroaryl)
- R 5 is OH or NH 2 .
- the dipeptide is linked to the bioactive polypeptide through the hydroxyl group of a serine or threonine residue (i.e., n is 0 and Rj 0 is H, or n is 0 and Rj 0 is CH 3 in the compound of Formula II). More particularly, the modified serine or threonine residue is located in a protein binding domain or active site, such that attachment of the dipeptide interferes with the bioactive polypeptide's ability to interact with its native receptor or substrate.
- a serine or threonine residue i.e., n is 0 and Rj 0 is H, or n is 0 and Rj 0 is CH 3 in the compound of Formula II.
- the modified serine or threonine residue is located in a protein binding domain or active site, such that attachment of the dipeptide interferes with the bioactive polypeptide's ability to interact with its native receptor or substrate.
- R 3 or R 4 represent amino acids of the bioactive peptide.
- R 3 and R 4 represent amino acids of the bioactive peptide.
- the prodrug comprises a general structure of A-B-Q, wherein A is a hydroxyl acid or amino acid covalently linked to B; B is an amino acid covalently linked to Q via an ester bond, and Q is a bioactive peptide, polypeptide, or protein, e.g., a glucagon superfamily peptide (e.g., a glucagon related analog peptide), wherein the chemical cleavage half-life (ti /2 ) of A-B from Q is no more than about 1 week in PBS under physiological conditions.
- A is a hydroxyl acid or amino acid covalently linked to B
- B is an amino acid covalently linked to Q via an ester bond
- Q is a bioactive peptide, polypeptide, or protein, e.g., a glucagon superfamily peptide (e.g., a glucagon related analog peptide), wherein the chemical cleavage half-life (ti /2 ) of
- a non-enzymatic self cleaving complex comprising the general structure A-B-Q
- Q is a known bioactive peptide and A-B represents a dipeptide. More particularly, A can be either an amino acid or a hydroxyl acid, and B is an amino acid.
- the dipeptide (A-B) is covalently bound to Q via an ester linkage at a position that interferes with Q's ability to interact with its corresponding receptor or cofactor.
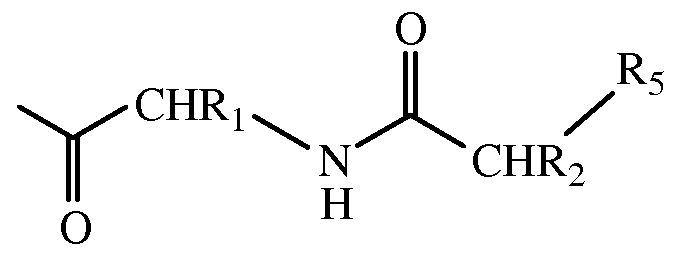
- Q is a bioactive peptide, polypeptide, or protein, e.g., a glucagon superfamily peptide such as a glucagon related analog peptide and A-B represents a dipeptide of the structure:
- R] and R 2 are independently selected from the group consisting of H, Ci-C 4 alkyl, (Ci-C 4 alkyl)OH, (Ci-C 4 alkyl)SH, (C 2 -C 3 alkyl)SCH 3 , (Ci-C 4 alkyl)CONH 2 , (Ci-C 4 alkyl)COOH, (Ci-C 4 alkyl)NH 2 , (Ci-C 4 alkyl)NHC(NH 2 + ) NH 2 , (C 4 -C 6 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Cio aryl)R 9 , and CH 2 (C 5 -C 9 heteroaryl),
- R 5 is OH or NH 2 .
- R 9 C 1 -C 4 alkyl, NH 2 , or OH, and wherein R 7 is -0-amino acid or O.
- A is an amino acid or a hydroxyl acid
- B is an amino acid covalently linked to Q via an ester bond
- Q is a bioactive peptide, polypeptide, or protein, e.g., a glucagon superfamily peptide such as a glucagon related analog peptide, wherein the dipeptide A-B further comprises a depot polymer linked to the side chain of A or B.
- the depot polymer is selected to be of a sufficient size that the complex A-B-Q is effectively sequestered at the site of injection or is otherwise incapable of interacting with its target (e.g., receptor).
- Chemical cleavage of A-B from Q produces a diketopiperazine or diketomorpholine and releases the active bioactive peptide, polypeptide, or protein to the patient in a controlled manner over a predetermined duration of time after administration.
- the bioactive polypeptide is selected from the group consisting of insulin, glucagon and GLP-I and derivatives of said polypeptides wherein the derivative polypeptide comprises 1 to 6 amino acid substitutions relative to the native sequence.
- the substituting amino acids can be either natural amino acids or synthetic amino acids, and in one embodiment the amino acid substitutions represent conservative amino acid substitutions.
- R 3 comprises the N-terminal amino acids of a bioactive protein (e.g., a glucagon, GLP-I or insulin peptide)
- R 4 comprises the C-terminal amino acids of the bioactive protein (e.g., a glucagon, GLP-I or insulin peptide)
- Ri and R 2 are independently selected from the group consisting of H, Ci-C 3 alkyl, CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Cio aryl), and CH 2 (Cs-C 9 heteroaryl)
- R 5 is OH or NH 2 wherein the a non- enzymatic activation tl/2 is between 1-100 hrs, and more typically between 12 and 72 hours, and in one embodiment the tl/2 is between 24-48 hrs.
- Ri is selected from the group consisting of CH 2 (CH 3 ) 2 , (C 4 - Cs)cycloalkyl, CH 2 (C 6 aryl), and CH 2 (Cs-C 6 heteroaryl)
- R 2 is selected from the group consisting of (C 4 -Cs)cycloalkyl, CH 2 (C 6 aryl) and CH 2 (Cs-C 6 heteroaryl)
- Rs is OH or NH 2 .
- the prodrug comprises the general structure of
- R 3 comprises the N-terminal amino acids of the bioactive protein which are N- terminal to an amino acid comprising a side chain structure
- R4 comprises the C-terminal amino acids of the bioactive protein which are C-terminal to the amino acid.
- the bioactive peptide, bioactive polypeptide, or bioactive protein is selected from a group consisting of a glucagon superfamily peptide (e.g., glucagon related analog peptide), osteocalcin, insulin, and an analog, derivative, or conjugate of one of the foregoing.
- the glucagon superfamily peptide is selected from the group consisting of Growth Hormone Releasing Hormone (GHRH; SEQ ID NO: 657), vasoactive intestinal peptide (VIP; SEQ ID NO: 658), Pituitary adenylate cyclase-activating polypeptide 27 (PACAP-27; SEQ ID NO: 659), peptide histidine methionine (PHM; SEQ ID NO: 660), or Secretin (SEQ ID NO: 661), glucagon (SEQ ID NO: 612), exendin-4 (SEQ ID NO: 662), Glucagon-like peptide -1 (GLP-I) (amino acids 7-37 provided as SEQ ID NO: 601), Glucagon-like peptide -2 (GLP-2) (SEQ ID NO: 663), GIP (SEQ ID NO: 664) or Oxyntomodulin (SEQ ID NO: 665), and an analog, derivative, or conjug
- a prodrug derivative of insulin wherein the insulin prodrug comprises an A chain comprising a sequence of SEQ ID NO: 626 and a B chain comprising a sequence of SEQ ID NO: 628 wherein a dipeptide of the formula
- R 1 and R 2 are independently selected from the group consisting of H, C 1 -C3 alkyl, CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 .
- n is an integer selected from 0 to 3 and m is an integer selected from 1-4
- R 3 is an amino acid sequence selected from the group consisting of X]X 2 X 3 G (SEQ ID NO: 621) and
- X is selected from the group consisting of histidine, hydroxy-histidine, homo-histidine, tyrosine and phenylalanine
- X 2 is an amino acid selected from the group consisting of glycine, alanine, serine, valine, d alanine, aminoisobutyric acid, N-methyl alanine and similar sized natural and synthetic amino acids
- X 3 is selected from the group consisting of glutamic acid, aspartic acid, glutamine and asparagine
- X 4 is selected from the group consisting of desaminohistidine, desaminohomo-histidine, desaminotyrosine and desaminophenylalanine
- R 4 is an amino acid sequence selected from the group consisting of
- FTSDVSSYLEGQAAKEFIAWLVKGRG SEQ ID NO: 603
- FTSDVSSYLEGQAAKEFIAWLVKGR-amide SEQ ID NO: 604
- FTSDVSSYLEGQAAKEFIAWLVKGX l4 PSSGAPPPS-amide SEQ ID NO: 605), wherein X] 4 is Arg or GIy
- FTSDYSKYLDSRRAQDFVQWLMNT SEQ ID NO: 618
- R 5 is NH 2 or HO; Re is H or
- R 7 is O or OX 4 X 2 X 3 G (SEQ ID NO: 634);
- R] and R 2 are independently selected from the group consisting of H, Ci -C 3 alkyl, CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 , with the proviso that when R 3 is
- R 6 is H, and when R 6 is
- R 3 is XiX 2 X 3 G-(SEQ ID NO: 621), further provided that when R 6 is H, R 3 is not X 1 X 2 X 3 G-(SEQ ID NO: 621).
- R] is selected from the group consisting of CH 2 (CH 3 ) 2 , (C 4 - Cs)cycloalkyl, CH 2 (C 6 -Ci O aryl), and CH 2 (Cs-C 9 heteroaryl) and R 2 is selected from the group consisting of (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci O aryl) and R 5 is OH or NH 2 .
- GLP- 1 prodrug comprising the structure of Formula III:
- R 3 is an amino acid sequence selected from the group consisting of R 5 HAQG (SEQ ID NO: 639), R 5 FAQG (SEQ ID NO: 640), R 5 YAQG (SEQ ID NO: 641);
- R 4 comprises an amino acid sequence selected from the group consisting of FTSDVSSYLEGQAAKEFIAWLVKGRG (SEQ ID NO: 603), FTSDVSSYLEGQAAKEFIAWLVKGR-amide (SEQ ID NO: 604), and FTSDVSSYLEGQAAKEFIAWLVKGX l4 PSSGAPPPS-amide (SEQ ID NO: 605), wherein Xi 4 is Arg or GIy;
- R 5 is NH 2 or HO
- Ri and R 2 are independently selected from the group consisting of H, Ci-C 3 alkyl, CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 .
- Ri is selected from the group consisting of CH 2 (CH 3 ) 2 , (C 4 -
- HO-FAEGTFTSDVSSYLEGQAAKEFIAWLVKGXPSSGAPPPS (SEQ ID NO: 607; HO- F 7 ,GLP(8-36)-CEX).
- Fig. 2 is a graph demonstrating the bioassay results for GLP-I analog HO-F7,GLP(8- 36)-CEX (O) relative to GLP-I (•).
- Fig. 3 is a synthetic scheme for preparing ester linked, amine nucleophile dipeptide prodrugs.
- Fig. 4 is a synthetic scheme for preparing ester linked, hydroxyl nucleophile dipeptide prodrugs.
- Figs 5A and 5B are schematic representations of the cleavage of the ester linkage by an amine nucleophile bearing prodrug (Fig. 5A) and by a hydroxyl nucleophile bearing prodrug (Fig. 5B) to generate the active compound and a diketopiperazine or diketomopholine, respectively.
- Fig. 6A-6D shows the structures of four prodrugs (6A: HO-F5dV6-O-F7,GLP(8-36)- CEX; 6B: F5V6-O-F7,GLP(8-36)-CEX; 6C: HO-F5F6-O-F7,GLP(8-36)-CEX and 6D: G 5 V 6 - O-F 7 ,GLP(8-36)CEX) and their respective tl/2 times.
- Fig. 7 is a bar graph representing bioassay data showing the relative potency of G 5 V 6 - O-F 7 ,GLP(8-36)CEX over time relative to appropriate GLP-I controls.
- Fig. 8 is a bar graph representing bioassay data showing the relative potency of HO- F 5 F 6 -O-F 7 ,GLP(8-36)-CEX over time relative to appropriate GLP-I controls.
- Fig. 9 is a graph demonstrating the bioassay results of internal serine/threonine GLP derivatives (F 7 Q 9 S 11 GLPCEX, ⁇ and F 7 Q 9 T 11 GLPCEX, O) relative to GLP-I ( ⁇ ).
- Fig. 10 is a graph plotting the logarithm of the concentrations of the prodrug and the drug over time, demonstrating the kinetic profile of the prodrug F 7 Q 9 -S ⁇ -( GIy-GIy)-GLP- CEX). The data shows the disappearance of a prodrug ( ⁇ ) and the appearance of a drug (D) over time. The results shown here are typical of the prodrugs disclosed herein.
- Fig. 1 IA-11C provide a synthetic scheme for the synthesis of (H7F),(E9Q),[T1 lS-O ⁇ -(Gly-Gly)],GLP(8-36)-CEX.
- Fig. 12 represents a graph of blood glucose (mg/dl) as a function of time (minutes) in response to a glucose injection in DIO mice followed by injection with PBS vehicle, a GLP-I -based peptide (Peptide B or D at 2 nmol/kg), or a prodrug of two different GLP-I -based peptides (Peptide A, C, or E at 2 nmol/kg).
- Fig. 13 represents a graph of blood glucose (mg/dl) as a function of time
- Fig. 14 represents a graph of blood glucose (mg/dl) as a function of time (minutes) in response to a glucose injection of DIO mice followed by an injection with PBS vehicle, a GLP-I -based peptide (Peptide D or F at 0.67 nmol/kg), or a prodrug of a GLP-I -based peptide (Peptide E or G at 0.67 nmol/kg).
- Fig. 15 represents a graph of blood glucose (mg/dl) as a function of time (minutes) in response to a glucose injection of DIO mice injected followed by an injection with vehicle control, a GLP-I -based pegylated peptide at 15 or 70 nmol/kg (Peptides Y and F-Peg), or a prodrug of a GLP-I -based peptide at 15 or 70 nmol/kg (Peptides (Z and G-Peg).
- Fig. 16 represents a graph of the total change in body weight of DIO mice injected with vehicle control, a GLP-I -based pegylated peptide at 15 or 70 nmol/kg (Peptides Y and F-Peg), or a prodrug of a GLP-I -based peptide at 15 or 70 nmol/kg (Peptides (Z and G-Peg).
- Fig. 17 represents a graph of the total food intake of DIO mice injected with vehicle control, a GLP-I -based pegylated peptide at 15 or 70 nmol/kg (Peptides Y and F-Peg), or a prodrug of a GLP-I -based peptide at 15 or 70 nmol/kg (Peptides Z and G- Peg).
- Fig. 18 is an alignment of amino acid sequences of certain glucagon superfamily peptides.
- prodrug is defined as any compound that undergoes biotransformation before exhibiting its pharmacological effects.
- bioactive polypeptide refers to polypeptides which are capable of exerting a biological effect in vitro and/or in vivo.
- a general reference to a peptide is intended to encompass peptides that have modified amino and carboxy termini.
- an amino acid chain comprising an amide group in place of the terminal carboxylic acid is intended to be encompassed by an amino acid sequence designating the standard amino acids.
- the term "O- amino acid” or "HO-amino acid” designates an amino acid wherein the native amino group at the N-terminus of an amino acid or an amino acid sequence has been replaced with an oxygen or hydroxyl group, respectively.
- “O-HAEG” or “HO-HAEG” is intended to designate an amino acid sequence (HAEG) wherein the native amino group at the N-terminus has been replaced with an oxygen or hydroxyl group, respectively.
- hydroxyl acid or "hydroxyl acid” as used herein refers to an amino acid wherein the native alpha amino group has been replaced with a hydroxyl group.
- the designation "-amino acid- amide” represents an amino acid sequence, wherein the native carboxy group has been replaced with an amide group.
- Designation of an amino acid without specifying its stereochemistry is intended to encompass either the L or D form of the amino acid or a racemic mixture.
- the term "pharmaceutically acceptable carrier” includes any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions such as an oil/water or water/oil emulsion, and various types of wetting agents. The term also encompasses any of the agents approved by a regulatory agency of the US Federal government or listed in the US Pharmacopeia for use in animals, including humans.
- pharmaceutically acceptable salt refers to salts of compounds that retain the biological activity of the parent compound, and which are not biologically or otherwise undesirable. Many of the compounds disclosed herein are capable of forming acid and/or base salts by virtue of the presence of amino and/or carboxyl groups or groups similar thereto.
- Pharmaceutically acceptable base addition salts can be prepared from inorganic and organic bases.
- Salts derived from inorganic bases include by way of example only, sodium, potassium, lithium, ammonium, calcium and magnesium salts.
- Salts derived from organic bases include, but are not limited to, salts of primary, secondary and tertiary amines.
- Salts derived from inorganic acids include hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like.
- Salts derived from organic acids include acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, malic acid, malonic acid, succinic acid, maleic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, p-toluene-sulfonic acid, salicylic acid, and the like.
- treating includes prophylaxis of the specific disorder or condition, or alleviation of the symptoms associated with a specific disorder or condition and/or preventing or eliminating said symptoms.
- treating diabetes will refer in general to maintaining glucose blood levels near normal levels and may include increasing or decreasing blood glucose levels depending on a given situation.
- an "effective" amount or a “therapeutically effective amount” of a prodrug refers to a nontoxic but sufficient amount of the prodrug to provide the desired effect.
- one desired effect would be the prevention or treatment of hyperglycemia.
- the amount that is "effective” will vary from subject to subject, depending on the age and general condition of the individual, mode of administration, and the like. Thus, it is not always possible to specify an exact “effective amount.” However, an appropriate “effective” amount in any individual case may be determined by one of ordinary skill in the art using routine experimentation.
- parenteral means not through the alimentary canal but by some other route such as subcutaneous, intramuscular, intraspinal, or intravenous.
- glucagon superfamily peptide refers to a group of peptides related in structure in their N-terminal and C-terminal regions (see, for example, Sherwood et al.,
- glucagon related analog peptides include all glucagon related analog peptides, as well as Growth Hormone Releasing Hormone (GHRH; SEQ ID NO: 657), vasoactive intestinal peptide (VIP; SEQ ID NO: 658), Pituitary adenylate cyclase- activating polypeptide 27 (PACAP-27; SEQ ID NO: 659), peptide histidine isoleucine (PHI), peptide histidine methionine (PHM) (SEQ ID NO: 660), and Secretin (SEQ ID NO: 661), and analogs, derivatives or conjugates thereof with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to the native peptide.
- Glucagon superfamily peptides preferably retain the ability to interact with its corresponding receptor.
- glucagon and glucagon analogs, derivatives, and conjugates thereof preferably retain the ability to interact with the glucagon receptor
- any reference to an amino acid position in a glucagon superfamily peptide refers to the position relative to the native glucagon amino acid sequence (SEQ ID NO: 612), see Fig. 18 for an alignment of representative glucagon superfamily peptides.
- glucagon related analog peptide refers to those peptides which have biological activity (as agonists or antagonists) at any one or more of the glucagon, GLP-I, GLP-2, and GIP receptors and comprise an amino acid sequence that shares at least 40% sequence identity (e.g., 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%) with at least one of native glucagon (SEQ ID NO: 612), native oxyntomodulin (SEQ ID NO: 665), native exendin-4 (SEQ ID NO: 662), native GLP-I (SEQ ID NOs: 601 and 602), native GLP- 2 (SEQ ID NO: 663), or native GIP (SEQ ID NO: 664).
- native glucagon SEQ ID NO: 612
- native oxyntomodulin SEQ ID NO: 665
- native exendin-4 SEQ ID NO: 662
- native GLP-I SEQ ID NOs:
- any reference to an amino acid position in a glucagon related analog peptide refers to the position relative to the native glucagon amino acid sequence (SEQ ID NO: 612).
- the term "identity" as used herein relates to the similarity between two or more sequences. Identity is measured by dividing the number of identical residues by the total number of residues and multiplying the product by 100 to achieve a percentage.
- GLP-I agonist refers to a compound that stimulates GLP-I receptor activity, as measured by cAMP production using a validated in vitro model assay, such as that described in Example 2.
- GLP-I is a generic term that designates GLP- 1(7- 36)amide (consisting of the sequence of SEQ ID NO: 602), GLP-l(7-37)acid (consisting of the sequence of SEQ ID NO: 601) or a mixture of those two compounds.
- GLP-I in the absence of any further designation is intended to mean native GLP- 1 , respectively.
- glucagon peptide is a generic term that designates the natural glucagon peptide of SEQ ID NO: 612 as well as modified derivatives having one or more amino acid substitutions at amino acid positions 1, 2, 5, 7, 8, 10, 12, 13, 14, 16, 17, 18, 24, 28 and 29, relative to the sequence of SEQ ID NO: 612.
- GLP-I peptide is a generic term that designates native GLP- 1 as well as modified derivatives having one or more amino acid substitutions at amino acid positions 1, 2, 3, 5, 8, 10, 12, 13, 14, 16, 17, 18, 24, 28 and 29, relative to the native GLP-I sequence.
- insulin peptide is a generic term that designates the 51 amino acid dimer comprising the A chain of SEQ ID NO: 613 and the B chain of SEQ ID NO: 614 as well as modified derivatives of the A chain and/or B chain, including one or more amino acid substitutions at positions selected from A5, A8, A9, AlO, A12, A14, A15, A17, A18, A21, Bl, B2, B3, B4, B5, B9, BlO, B13, B14, B17, B20, B21, B22, B23, B26, B27, B28, B29 and B30 or deletions of any or all of positions B 1-5 and B26-30.
- an amino acid "modification” refers to a substitution, addition or deletion of an amino acid, and includes substitution with or addition of any of the 20 amino acids commonly found in human proteins, as well as atypical or non- naturally occurring amino acids.
- Commercial sources of atypical amino acids include Sigma-Aldrich (Milwaukee, WI), ChemPep Inc. (Miami, FL), and Genzyme Pharmaceuticals (Cambridge, MA).
- Atypical amino acids may be purchased from commercial suppliers, synthesized de novo, or chemically modified or derivatized from naturally occurring amino acids.
- substitution refers to the replacement of one amino acid residue by a different amino acid residue.
- polyethylene glycol chain refers to mixtures of condensation polymers of ethylene oxide and water, in a branched or straight chain, represented by the general formula H(OCH 2 CH 2 ) n OH, wherein n is at least 9. Absent any further characterization, the term is intended to include polymers of ethylene glycol with an average total molecular weight selected from the range of 500 to 80,000 Daltons. "Polyethylene glycol chain” or “PEG chain” is used in combination with a numeric suffix to indicate the approximate average molecular weight thereof. For example, PEG-5,000 refers to polyethylene glycol chain having a total molecular weight average of about 5,000 Daltons.
- pegylated refers to a compound that has been modified from its native state by linking a polyethylene glycol chain to the compound.
- a “pegylated polypeptide” is a polypeptide that has a PEG chain covalently bound to the polypeptide.
- Linker is a bond, molecule or group of molecules that binds two separate entities to one another. Linkers may provide for optimal spacing of the two entities or may further supply a labile linkage that allows the two entities to be separated from each other. Labile linkages include photocleavable groups, acid-labile moieties, base-labile moieties and enzyme-cleavable groups.
- a "dimer” is a complex comprising two subunits covalently bound to one another via a linker.
- dimer when used absent any qualifying language, encompasses both homodimers and heterodimers.
- a homodimer comprises two identical subunits, whereas a heterodimer comprises two subunits that differ, although the two subunits are substantially similar to one another.
- Ci-C n alkyl wherein n can be from 1 through 6, as used herein, represents a branched or linear alkyl group having from one to the specified number of carbon atoms.
- Typical Ci-C 6 alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, iso-propyl, butyl, iso-butyl, sec-butyl, tert-butyl, pentyl, hexyl and the like.
- C 2 -C n alkenyl wherein n can be from 2 through 6, as used herein, represents an olefinically unsaturated branched or linear group having from 2 to the specified number of carbon atoms and at least one double bond.
- C 2 -C n alkynyl wherein n can be from 2 to 6, refers to an unsaturated branched or linear group having from 2 to n carbon atoms and at least one triple bond. Examples of such groups include, but are not limited to, 1-propynyl, 2-propynyl, 1-butynyl, 2-butynyl, 1-pentynyl, and the like.
- aryl refers to a mono- or bicyclic carbocyclic ring system having one or two aromatic rings including, but not limited to, phenyl, naphthyl, tetrahydronaphthyl, indanyl, indenyl, and the like.
- the size of the aryl ring and the presence of substituents or linking groups are indicated by designating the number of carbons present.
- (C 1 -C 3 alkyl)(C 6 -Cio aryl) refers to a 5 to 10 membered aryl that is attached to a parent moiety via a one to three membered alkyl chain.
- heteroaryl refers to a mono- or bi- cyclic ring system containing one or two aromatic rings and containing at least one nitrogen, oxygen, or sulfur atom in an aromatic ring.
- the size of the heteroaryl ring and the presence of substituents or linking groups are indicated by designating the number of carbons present.
- (Ci-C n alkyl)(Cs-C6 heteroaryl) refers to a 5 or 6 membered heteroaryl that is attached to a parent moiety via a one to "n" membered alkyl chain.
- charged amino acid refers to an amino acid that comprises a side chain that is negatively charged (i.e., de-protonated) or positively charged (i.e., protonated) in aqueous solution at physiological pH.
- negatively charged amino acids include aspartic acid, glutamic acid, cysteic acid, homocysteic acid, and homoglutamic acid
- positively charged amino acids include arginine, lysine and histidine.
- Charged amino acids include the charged amino acids among the 20 amino acids commonly found in human proteins, as well as atypical or non-naturally occurring amino acids.
- acidic amino acid refers to an amino acid that comprises a second acidic moiety, including for example, a carboxylic acid or sulfonic acid group.
- patient without further designation is intended to encompass any warm blooded vertebrate domesticated animal (including for example, but not limited to livestock, horses, cats, dogs and other pets) and humans.
- the present disclosure describes the formulation of prodrug derivatives of known bioactive polypeptides. More particularly, the prodrugs disclosed herein are formulated to enhance the half life of the parent bioactive peptide or protein, while allowing for activation of the prodrug via a non-enzymatic degradation mechanism.
- the ideal prodrug should be soluble in water at physiological conditions (for example, a pH of 7.2 and 37 0 C), and it should be stable in the powder form for long term storage. It should also be immunologically silent and exhibit a low activity relative to the parent drug.
- the prodrug will exhibit no more than 10% of the activity of the parent drug, in one embodiment the prodrug exhibits less than 10%, less than 5%, about 1%, or less than 1% activity relative to the parent drug. Furthermore, the prodrug, when injected in the body, should be quantitatively converted to the active drug within a defined period of time. As disclosed herein, applicants have provided a general technique for producing prodrugs of known bioactive polypeptides that meets each of these objectives.
- a chemoreversible prodrug comprising the sequence of a known bioactive polypeptide modified to have a dipeptide covalently bound to the bioactive polypeptide via an ester linkage.
- a prodrug is provided having a non- enzymatic activation half time (tl/2) of between 1-100 hrs under physiological conditions.
- Physiological conditions as disclosed herein are intended to include a temperature of about 35 to 4O 0 C and a pH of about 7.0 to about 7.4 and more typically include a pH of 7.2 to 7.4 and a temperature of 36 to 38 0 C.
- the rate of cleavage, and thus activation of the prodrug depends on the structure and stereochemistry of the dipeptide pro-moiety and also on the strength of the nucleophile.
- the prodrugs disclosed herein will ultimately be chemically converted to structures that can be recognized by the native receptor of the drug, wherein the speed of this chemical conversion will determine the time of onset and duration of in vivo biological action.
- the molecular design disclosed in this application relies upon an intramolecular chemical reaction that is not dependent upon additional chemical additives, or enzymes. The speed of conversion is controlled by the chemical nature of the dipeptide substituent and its cleavage under physiological conditions.
- prodrugs are provided wherein the bioactive polypeptides have extended half lives of at least 1 hour, and more typically greater than 20 hours but less than 100 hours, and are converted to the active form at physiological conditions through a non- enzymatic reaction driven by inherent chemical instability.
- a non- enzymatic activation tl/2 time of the prodrug is between 1-100 hrs, and more typically between 12 and 72 hours, and in one embodiment the tl/2 is between 24-48 hrs as measured by incubating the prodrug in a phosphate buffer solution (e.g., PBS) at 37°C and pH of 7.2.
- a phosphate buffer solution e.g., PBS
- activation of the prodrug occurs after cleavage of an ester bond linked dipeptide, and formation of a diketopiperazine or diketomorpholine, and the active bioactive polypeptide (see Figs. 5A & 5B).
- Specific dipeptides composed of natural or synthetic amino acids have been identified that facilitate intramolecular decomposition under physiological conditions to release bioactive peptides.
- the prodrug chemistry is broadly applicable to peptide and protein based drugs where an aliphatic hydroxyl group can be accommodated in the active site, and when the chemically modified derivative yields a poorly active (e.g., having 10% or less activity relative to the parent) peptide or protein.
- Medicinally important peptides and proteins where this prodrug technology can be employed include but are not limited to insulin, glucagon and glucagon related peptides (GLPs, oxyntomodulin), gastrin inhibitory peptide (GIP), growth hormone releasing hormone (GHRH), calcitonin, parathyroid hormone (PTH), neuropeptide Y (NPY), pancreatic polypeptide (PP), and related substances.
- GLPs glucagon and glucagon related peptides
- GLPs gastrin inhibitory peptide
- GHRH growth hormone releasing hormone
- calcitonin parathyroid hormone
- NPY neuropeptide Y
- PP pancreatic polypeptide
- Medicinally important proteins where this prodrug technology can be employed include but are not limited to growth hormone, erythropoietin (EPO), neutrophil stimulating growth factors (CGSF), interferons, blood coagulation enzymes, antibodies, leptin and the like.
- EPO erythropoietin
- CGSF neutrophil stimulating growth factors
- interferons blood coagulation enzymes
- antibodies leptin and the like.
- a prodrug derivative of glucagon, insulin, GLP-I, or a modified derivative of glucagon, insulin or GLP- 1 are provided.
- a modified derivative of glucagon, insulin or GLP-I includes, for example, a polypeptide that differs from the native sequence of glucagon, insulin or GLP, respectively, by 1, 2, 3, 4, 5, or 6 amino acid substitutions.
- an aliphatic hydroxyl group of the bioactive peptide or protein including for example the hydroxyl bearing side chain of a serine or threonine residue or the hydroxyl group of an N-terminal hydroxylated amino acid (HO- amino acid), is modified by the covalent linkage of the group:
- R] and R 2 are independently selected from the group consisting of H, C 1 -C 6 alkyl, (C 1 -C 4 alkyl)OH, (C 1 -C 4 alkyl)SH, (C 2 - C 3 alkyl)SCH 3 , (Ci-C 4 alkyl)CONH 2 , (Ci-C 4 alkyl)COOH, (Ci-C 4 alkyl)NH 2 , (Ci-C 4 alkyl)NHC(NH 2 + ) NH 2 , (C 4 -C 6 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Ci 0 aryl)R 9 , and CH 2 (C 5 -C 9 heteroaryl) and R 5 is OH or NH 2 .
- the bioactive peptide is a glucagon superfamily peptide (e.g., a glucagon related analog peptide), osteocalcin, or an analog, derivative, or conjugate of one of the foregoing.
- a glucagon superfamily peptide e.g., a glucagon related analog peptide
- osteocalcin e.g., osteocalcin, or an analog, derivative, or conjugate of one of the foregoing.
- bioactive protein is glucagon, GLP-I or insulin.
- Ri and R 2 are independently selected from the group consisting of
- Ci-C 3 alkyl CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 .
- Ri is selected from the group consisting of CH 2 (CH 3 ) 2 , (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl)
- R 2 is selected from the group consisting of (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl) and CH 2 (C 5 -C 9 heteroaryl)
- R 5 is OH or NH 2 .
- Rl2 wherein R 3 is selected from the group consisting of NH 2 , HO-, an amino acid sequence, and
- R 4 is -OH, NH 2 , or an amino acid sequence
- Rio is selected from the group consisting of H, Ci-C 4 alkyl, and (CH 2 ) n (C6-Cio aryl), W is C 6 -Ci O aryl or a bond, and n is an integer from 0 to 3,
- Ri and R 2 are independently selected from the group consisting of H, Ci-C 4 alkyl, (Ci-C 4 alkyl)OH, (Ci-C 4 alkyl)SH, (C 2 -C 3 alkyl)SCH 3 , (Ci-C 4 alkyl)CONH 2 , (Ci-C 4 alkyl)COOH, (Ci-C 4 alkyl)NH 2 , (Ci-C 4 alkyl)NHC (NH 2 + ) NH 2 , (C 4 -C 6 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Cio aryl)R 9 , and CH 2 (C 5 -C 9 heteroaryl), R 5 is OH or NH 2 and
- R 9 Ci-C 4 alkyl, NH 2 or OH, wherein R 7 is -O- amino acid or O, and Ri 2 is -OH or
- R 3 is NH 2 or an amino acid sequence.
- R 3 or R 4 is an amino acid sequence of a bioactive peptide.
- R 3 is an amino acid sequence of the bioactive peptide which is N-terminal to an amino acid of the bioactive peptide comprising a side chain of structure
- R 4 is an amino acid sequence of the bioactive peptide which is C -terminal to the amino acid of the bioactive peptide.
- R 7 is 0-amino acid
- R 7 is an amino acid sequence of the bioactive peptide which is N- terminal to an amino acid of the bioactive peptide comprising a side chain of structure
- Rl 2 and R 4 is an amino acid sequence of the bioactive peptide which is C -terminal to the amino acid of the bioactive peptide.
- R 3 is
- R 7 and R 4 represent amino acid sequences from glucagon, GLP-I or insulin peptides.
- R 3 is an N-terminal sequence selected from either glucagon, GLP-I or insulin peptides
- R 4 is a carboxy terminal sequence selected from either glucagon, GLP-I or insulin peptides
- Ri 2 is
- R 3 is H or the N-terminal amino acids of the bioactive protein (e.g., a glucagon,
- R 4 represents OH or the C-terminal amino acids of the bioactive protein (e.g., a glucagon, GLP-I or insulin peptide), R] 0 is selected from the group consisting of H, Ci-C 4 alkyl, and (CH 2 ) n (C6-Cio aryl), wherein n is an integer from 0 to 3
- Ri and R 2 are independently selected from the group consisting of H, Ci-C 4 alkyl,
- Ri is selected from the group consisting of CH(CH 3 ) 2 ,
- R 2 is selected from the group consisting of (C 0 -C 4 alkyl)Ci-C 4 alkyl, (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 aryl), and (C 0 -C 4 alkyl)(C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 .
- the dipeptide is linked to the bioactive polypeptide through the hydroxyl group of a serine or threonine residue (i.e., n is 0 and Ri 0 is H, or n is 0 and Ri 0 is CH 3 in the compound of Formula III). More particularly the modified serine or threonine residue is located in a protein binding domain or active site, such that attachment of the dipeptide interferes with the bioactive polypeptide's ability to interact with its native receptor or substrate.
- at least one of R 3 or R 4 represent amino acids of the bioactive peptide. In some embodiments, R 3 and R 4 represent amino acids of the bioactive peptide.
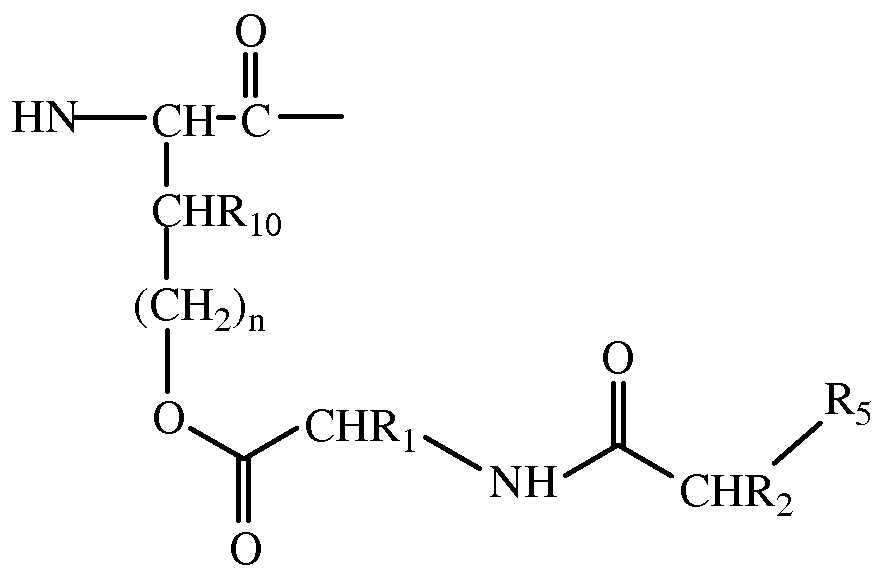
- polypeptide having the general structure of Formula II wherein R 3 is selected from the group consisting of H and an amino acid sequence,
- R 4 is -OH or an amino acid sequence
- Rio is selected from the group consisting of H, Ci-C 4 alkyl, and (CH 2 ) n (C6-Cio aryl), wherein n is an integer from 0 to 3 ;
- Ri is selected from the group consisting of CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 aryl), and (C 0 -C 4 alkyl)(C 5 -C 6 heteroaryl),
- R 2 is selected from the group consisting of (C 0 -C 4 alkyl)Ci-C 4 alkyl, (C 4 - C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 aryl), and (C 0 -C 4 alkyl)(C 5 -C 6 heteroaryl), and R 5 is OH or NH 2 .
- Ri 0 is H and n is 0.
- R 3 is H or the N-terminal amino acids of the bioactive protein located upstream of a serine/threonine residue present (either naturally or as an amino acid substitution) in the bioactive polypeptide
- R 4 represents OH or the C-terminal amino acids of the bioactive protein located downstream of a serine/theronine residue present (either naturally or as an amino acid substitution) in the bioactive polypeptide (acid or amide)
- R] 0 is H or CH 3 , wherein a serine/theronine moiety located within said bioactive protein has been modified by the covalent linkage of the group:
- Rj and R 2 are independently selected from the group consisting of H, C]-C 4 alkyl, (Ci-C 4 alkyl)OH, (Ci-C 4 alkyl)SH, (C 2 -C 3 alkyl)SCH 3 , (Ci-C 4 alkyl)CONH 2 , (Ci-C 4 alkyl)COOH, (Ci-C 4 alkyl)NH 2 , (Ci-C 4 alkyl)NHC (NH 2 + ) NH 2 , (C 4 -C 6 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Cio aryl)R 9 , and CH 2 (C 5 -C 9 heteroaryl),
- Ri is selected from the group consisting of CH(CH 3 ) 2 , CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Ci 0 aryl), and (C 0 - C 4 alkyl)(C 5 -C 9 heteroaryl),
- R 2 is selected from the group consisting of (C 0 -C 4 alkyl)Ci-C 4 alkyl, (C 4 -C 5 )cycloalkyl, (C 0 -C 4 alkyl)(C 6 -Ci 0 aryl), and (C 0 -C 4 alkyl)(C 5 -C 9 heteroaryl), and R 5 is OH or NH 2 .
- the serine/threonine residue selected for modification is located in a protein binding domain or active site, such that attachment of the dipeptide interferes with the
- R 3 or R 4 represents amino acids of the bioactive peptide.
- both of R 3 and R 4 represent amino acids of the bioactive peptide.
- R3 is an amino acid sequence of the bioactive peptide which is N-terminal to an amino acid of the bioactive peptide comprising a side chain of structure
- R 1 9 and R4 is an amino acid sequence of the bioactive peptide which is C-terminal to the amino acid of the bioactive peptide.
- the bioactive polypeptide is selected from the group consisting of insulin, glucagon, GLP-I and derivatives of those polypeptides wherein the derivatives differ by 1, 2, 3, 4, 5 or 6 amino acids relative to the native sequence.
- the insulin, glucagon, GLP-I derivative differs from the native sequence by 1, 2, 3, 4, 5 or 6 conservative amino acids substitutions.
- R 3 comprises the native N-terminal amino acids of a bioactive protein
- R 4 comprises the native C-terminal amino acids of the bioactive protein
- R] and R 2 are independently selected from the group consisting of H, C 1 -C3 alkyl, CH 2 CH(CH 3 ) 2 , CH(CH 3 )(CH 2 CH 3 ), (C 4 -C 5 )cycloalkyl, CH 2 (C 6 -Ci 0 aryl), and CH 2 (C 5 -C 9 heteroaryl)
- R 5 is OH or NH 2
- the a non-enzymatic activation tl/2 is between 10-100 hrs.
- the tl/2 of the prodrug is between 20-70 hrs.
- Ri is selected from the group consisting of CH 2 (CH 3 ) 2 , (C 4 - C 5 )cycloalkyl, CH 2 (C 6 aryl), and CH 2 (C 5 -C 6 heteroaryl)
- R 2 is selected from the group consisting of (C 4 -C 5 )cycloalkyl, CH 2 (C 6 aryl) and CH 2 (C 5 -C 6 heteroaryl)
- R 5 is OH or NH 2 .
- the prodrug comprises the general structure of
- R 3 comprises the N-terminal amino acids of the bioactive protein which are N- terminal to an amino acid of the bioactive protein comprising a side chain of structure
- X and R4 comprises C -terminal amino acids of the bioactive protein which are C -terminal to the amino acid of the bioactive protein.
- the bioactive polypeptide is selected from the group consisting of insulin, glucagon, GLP-I and derivatives of insulin, glucagon and GLP-I wherein the derivative polypeptides comprise 1 to 6 amino acid substitutions relative to the native sequence.
- the substituting amino acids can be either natural amino acids or synthetic amino acids, and in one embodiment the amino acid substitutions represent conservative amino acid substitutions.
- Bioactive Peptides, Polypeptides, and Proteins The bioactive peptide, bioactive polypeptide, or bioactive protein linked via an ester bond to the prodrug dipeptide moiety can be a glucagon superfamily peptide (e.g., a member of the glucagon superfamily (those described in Sherwood et al., 2000, supra), a glucagon related analog peptide), osteocalcin, insulin, or an analog, derivative, or conjugate thereof.
- glucagon superfamily peptide e.g., a member of the glucagon superfamily (those described in Sherwood et al., 2000, supra)
- glucagon related analog peptide e.g., osteocalcin, insulin, or an analog, derivative, or conjugate thereof.
- the bioactive peptide is osteocalcin or an analog or derivative thereof comprising an amino acid sequence which is at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identical to native osteocalcin over the length of the native peptide.
- the bioactive peptide may comprise an analog of osteocalcin with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to native osteocalcin. Insulin
- the bioactive peptide is insulin, or an analog or derivative thereof comprising an A chain and a B chain, each chain comprising an amino acid sequence which is at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identical to native A or B chain of insulin over the length of the native peptide.
- the insulin analog or derivative may comprise an analog of an insulin A or B chain with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to the native A or B insulin chain.
- the A chain of insulin comprises an amino acid sequence of SEQ ID NO: 613 or SEQ ID NO: 626, or an analog of derivative thereof (e.g., having at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identity to SEQ ID NO: 613 or 626 or having up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to SEQ ID NO: 613 or 626).
- the B chain of insulin can comprise an amino acid sequence of SEQ ID NO: 614, SEQ ID NO: 627, or SEQ ID NO: 628, or an analog or derivative thereof (e.g., having at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identity to SEQ ID NO: 614, SEQ ID NO: 627, or SEQ ID NO: 628 or having up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to SEQ ID NO: 614, SEQ ID NO: 627, or SEQ ID NO: 628.
- an amino acid sequence of SEQ ID NO: 614, SEQ ID NO: 627, or SEQ ID NO: 628 or an analog or derivative thereof (e.g., having at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identity to SEQ ID NO: 614, SEQ ID NO: 627, or SEQ
- the bioactive peptide is any of the glucagon superfamily peptides that are known in the art, including, for example, GHRH (SEQ ID NO: 657), VIP (SEQ ID NO: 658), PACAP-27 (SEQ ID NO: 659), PHM (SEQ ID NO: 660), Secretin (SEQ ID NO: 661), glucagon (SEQ ID NO: 612), exendin-4 (SEQ ID NO: 662), Glucagon-like peptide -1 (GLP-I) (e.g., amino acids 7-37 provided as SEQ ID NO: 601), Glucagon-like peptide -2 (GLP-2) (SEQ ID NO: 663), GIP (SEQ ID NO: 664) and Oxyntomodulin (SEQ ID NO: 665).
- GHRH SEQ ID NO: 657
- VIP SEQ ID NO: 658
- PACAP-27 SEQ ID NO: 659
- PHM SEQ ID NO: 660
- Glucagon superfamily peptides may have common structural characteristics, including but not limited to homology within the N-terminal amino acids and/or alpha-helical structure within the C-terminal portion. It is believed that the C-terminus generally functions in receptor binding and the N-terminus generally functions in receptor signaling. A few amino acids in the N-terminal portion and C-terminal portion, for example, Hisl, Gly4, Phe6, Phe22, Val23, Trp25, and Leu26 (according to the amino acid numbering of Glucagon), are highly conserved among members of the glucagon superfamily, such that the amino acids of the member of the glucagon superfamily at the corresponding positions show identity, are conservatively substituted or otherwise exhibit similarity in amino acid side chains.
- the glucagon superfamily peptide may be an analog or derivative of GHRH (SEQ ID NO: 657), VIP (SEQ ID NO: 658), PACAP-27 (SEQ ID NO: 659), PHM (SEQ ID NO: 660), Secretin (SEQ ID NO: 661), glucagon (SEQ ID NO: 612), exendin-4 (SEQ ID NO: 662), Glucagon-like peptide -1 (GLP-I) (e.g., amino acids 7-37 provided as SEQ ID NO: 601), Glucagon-like peptide -2 (GLP-2) (SEQ ID NO: 663), GIP (SEQ ID NO: 664) and Oxyntomodulin (SEQ ID NO: 665) comprising the native amino acid sequence with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications.
- GLP-I Glucagon-like peptide -1
- GLP-2 e.g., amino acids 7-37 provided as SEQ ID
- the glucagon superfamily peptide may comprise a C-terminus or a C- terminal amino acid sequence including but not limited to: COOH, CONH 2 , GPSSGAPPPS (SEQ ID NO: 624), GPSSGAPPPS-CONH 2 (SEQ ID NO: 723), a oxyntomodulin carboxy terminal extension, KRNRNNIA (SEQ ID NO: 625) or KGKKNDWKHNITQ (SEQ ID NO: 666).
- Additional, C-terminal amino acid sequences for glucagon superfamily peptides are further detailed below.
- the glucagon superfamily peptide can be an analog of GHRH
- the glucagon superfamily peptide may comprise an analog of GHRH with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications relative to native GHRH.
- the bioactive peptide can comprise an amino acid sequence which is a chimera of two or more native glucagon related analog peptide sequences.
- the bioactive peptide comprises an amino acid sequence that is at least about 50% identical to native glucagon (SEQ ID NO: 612) and retains the alpha-helix conformation of the amino acids corresponding to amino acids 12-29 of SEQ ID NO: 612.
- the bioactive peptide may be a conjugate of a glucagon superfamily peptide. Conjugates are further described herein with regard to glucagon related analog peptides. The teachings of conjugates in this context generally apply to the bioactive peptides which are not glucagon related analog peptides.
- the glucagon superfamily peptide is a glucagon related analog peptide, e.g. glucagon (SEQ ID NO: 612), oxyntomodulin (SEQ ID NO: 665), exendin-4 (SEQ ID NO: 662), Glucagon-like peptide -1 (GLP-I) (amino acids 7-37 provided as SEQ ID NO: 601), Glucagon-like peptide -2 (GLP-2) (SEQ ID NO: 663), GIP (SEQ ID NO: 664) or analogs, derivatives and conjugates of the foregoing.
- glucagon SEQ ID NO: 612
- oxyntomodulin SEQ ID NO: 665
- exendin-4 SEQ ID NO: 662
- GLP-I Glucagon-like peptide -1
- GLP-2 Glucagon-like peptide -2
- GIP GIP
- the glucagon related analog peptide comprises an amino acid sequence of native glucagon, native exendin- 4, native (7-37)GLP-l, native GLP-2, native GHRH, native VIP, native PACAP-27, native PHM, native Oxyntomodulin, native Secretin, or native GIP with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications.
- native glucagon native exendin- 4, native (7-37)GLP-l, native GLP-2, native GHRH, native VIP, native PACAP-27, native PHM, native Oxyntomodulin, native Secretin, or native GIP with up to 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 amino acid modifications.
- native peptides are known in the art.
- the glucagon related analog peptide comprises an amino acid sequence that is at least about 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% identical to the corresponding sequence of native glucagon, native oxyntomodulin, native exendin-4, native (7-37)GLP-l, native GLP-2, or native GIP over the length of the native peptide (or over the positions which correspond to glucagon, see e.g., Figure 18).
- the glucagon related analog peptide is a Class 1, 2, 3, 4 or 5 glucagon related analog peptide as detailed herein.
- glucagon related analog peptide refers to those peptides which have biological activity (as agonists or antagonists) at any one or more of the glucagon, oxyntomodulin, exendin-4, GLP-I, GLP-2, and GIP receptors and comprise an amino acid sequence that shares at least 40% sequence identity (e.g., 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%) with at least one of native glucagon, native oxyntomodulin, native exendin- 4, native GLP-I, native GLP-2, or native GIP.
- glucagon related analog peptides are contemplated, e.g. peptides which have biological activity (as agonists or antagonists) at any one or more of the glucagon or GLP-I or GIP receptors, together with all possible subsets of sequence identity to each listed native peptide, e.g., comprise an amino acid sequence that shares at least 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, or 95% sequence identity with native glucagon over the length of native glucagon.
- the glucagon related analog peptide is a peptide having glucagon receptor agonist activity, GIP receptor agonist activity, glucagon receptor/GLP-1 receptor co-agonist activity, glucagon receptor antagonist activity, or glucagon receptor antagonist & GLP-I receptor agonist activity.
- the peptide retains an alpha-helix conformation in the C-terminal half of the molecule.
- the peptide retains positions involved in receptor interaction or signaling, e.g. position 3 of glucagon, or position 7, 10, 12, 13, 15 or 17 of (1-37)GLP-1.
- the glucagon related analog peptide can be a peptide of Class 1, Class 2, Class 3, Class 4, and/or Class 5, each of which is further described herein.
- the glucagon related analog peptide can comprise the native glucagon amino acid sequence (SEQ ID NO: 612) with modifications.
- the glucagon related analog peptide may comprise a total of 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 amino acid modifications relative to the native glucagon sequence, e.g. conservative or non-conservative substitutions.
- Modifications and substitutions described herein are, in certain aspects made at specific positions within a glucagon related analog peptide wherein the numbering of the position corresponds to the numbering of glucagon (SEQ ID NO: 612).
- 1, 2, 3, 4 or 5 non- conservative substitutions are carried out at any of positions 2, 5, 7, 10, 11, 12, 13, 14, 17, 18, 19, 20, 21, 24, 27, 28 or 29 and up to 5 further conservative substitutions are carried out at any of these positions.
- 1, 2, or 3 amino acid modifications are carried out within amino acids at positions 1-16, and 1, 2 or 3 amino acid modifications are carried out within amino acids at positions 17-26.
- such glucagon related analog peptides retain at least 22, 23, 24, 25, 26, 27 or 28 of the naturally occurring amino acids at the corresponding positions in native glucagon (e.g. have 1-7, 1-5 or 1-3 modifications relative to naturally occurring glucagon).
- the glucagon related analog peptide comprises a modification at position 1 or 2 to reduce susceptibility to cleavage by dipeptidyl peptidase IV. More particularly, in some embodiments, position 1 of a glucagon related analog peptide (e.g., selected from those in Fig. 10) is substituted with an amino acid selected from the group consisting of D-histidine, alpha, alpha-dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha-methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl- histidine, acetyl-histidine and homo-histidine.
- DMIA alpha
- N-methyl histidine alpha-methyl imidiazole acetic acid
- imidazole acetic acid desaminohistidine, hydroxyl- histidine, acetyl-histidine and homo-histidine.
- position 2 of the glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-serine, D-alanine, valine, glycine, N-methyl serine, and aminoisobutyric acid.
- the glucagon related analog peptide (e.g., a Class 1 glucagon related analog peptide, Class 2 glucagon related analog peptide, Class 3 glucagon related analog peptide, Class 4 glucagon related analog peptides or Class 5 glucagon related analog peptide) is attached (covalently bonded) to a hydrophilic moiety.
- Hydrophilic moieties can be attached to the glucagon related analog peptide under any suitable conditions used to react a protein with an activated polymer molecule.
- Any means known in the art can be used, including via acylation, reductive alkylation, Michael addition, thiol alkylation or other chemoselective conjugation/ligation methods through a reactive group on the PEG moiety (e.g., an aldehyde, amino, ester, thiol, ⁇ -haloacetyl, maleimido or hydrazino group) to a reactive group on the target compound (e.g., an aldehyde, amino, ester, thiol, ⁇ -haloacetyl, maleimido or hydrazino group).
- a reactive group on the PEG moiety e.g., an aldehyde, amino, ester, thiol, ⁇ -haloacetyl, maleimido or hydrazino group
- a reactive group on the target compound e.g., an aldehyde, amino, ester, thiol, ⁇ -haloacety
- Activating groups which can be used to link the water soluble polymer to one or more proteins include without limitation sulfone, maleimide, sulfhydryl, thiol, triflate, tresylate, azidirine, oxirane and 5-pyridyl. If attached to the peptide by reductive alkylation, the polymer selected should have a single reactive aldehyde so that the degree of polymerization is controlled. See, for example, Kinstler et al., Adv. Drug. Delivery Rev. 54: 477-485 (2002); Roberts et al., Adv. Drug Delivery Rev. 54: 459-476 (2002); and Zalipsky et al., Adv. Drug Delivery Rev. 16: 157-182 (1995).
- Suitable hydrophilic moieties include polyethylene glycol (PEG), polypropylene glycol, polyoxyethylated polyols (e.g., POG), polyoxyethylated sorbitol, polyoxyethylated glucose, polyoxyethylated glycerol (POG), polyoxyalkylenes, polyethylene glycol propionaldehyde, copolymers of ethylene glycol/propylene glycol, monomethoxy- polyethylene glycol, mono-(Cl-ClO) alkoxy- or aryloxy-polyethylene glycol, carboxymethylcellulose, polyacetals, polyvinyl alcohol (PVA), polyvinyl pyrrolidone, poly-1, 3-dioxolane, poly-1, 3, 6-trioxane, ethylene/maleic anhydride copolymer, poly (.beta.-amino acids) (either homopolymers or random copolymers), poly (n- vinyl pyrrolidon
- Dextrans are polysaccharide polymers of glucose subunits, predominantly linked by ⁇ l-6 linkages. Dextran is available in many molecular weight ranges, e.g., about 1 kD to about 100 kD, or from about 5, 10, 15 or 20 kD to about 20, 30, 40, 50, 60, 70, 80 or 90 kD.
- the hydrophilic moiety is a polyethylene glycol (PEG) chain or other water soluble polymer that is covalently linked to the side chain of an amino acid residue at one or more of positions 16, 17, 21, 24, 29, 40 of said glucagon related analog peptide, within a C-terminal extension, or at the C-terminal amino acid.
- PEG polyethylene glycol
- the native amino acid at that position is substituted with an amino acid having a side chain suitable for crosslinking with hydrophilic moieties, to facilitate linkage of the hydrophilic moiety to the peptide.
- exemplary amino acids include Cys, Lys, Orn, homo-Cys, or acetyl phenylalanine (Ac-Phe).
- an amino acid modified to comprise a hydrophilic group is added to the peptide at the C-terminus.
- the hydrophilic moiety e.g., polyethylene glycol chain in accordance with some embodiments has a molecular weight selected from the range of about 500 to about 40,000 Daltons.
- the polyethylene glycol chain has a molecular weight selected from the range of about 500 to about 5,000 Daltons, or about 1,000 to about 5,000 Daltons.
- the hydrophilic moiety, e.g., polyethylene glycol chain has a molecular weight of about 10,000 to about 20,000 Daltons.
- the hydrophilic moiety, e.g. polyethylene glycol chain has a molecular weight of about 20,000 to about 40,000 Daltons.
- Linear or branched polymers are contemplated.
- Resulting preparations of conjugates may be essentially monodisperse or polydisperse, and may have about 0.5, 0.7, 1, 1.2, 1.5 or 2 polymer moieties per peptide.
- the glucagon related analog peptide (e.g. a Class 1 glucagon related analog peptide, Class 2 glucagon related analog peptide, Class 3 glucagon related analog peptide, Class 4 glucagon related analog peptide, Class 4 glucagon related analog peptides or Class 5 glucagon related analog peptide), is modified to comprise an acyl group.
- Acylation can be carried out at any position within the glucagon related analog peptide, including any of positions 1-29, a position within a C-terminal extension, or the C-terminal amino acid, provided that the activity exhibited by the non-acylated glucagon related analog peptide is retained upon acylation.
- the acylated peptide retains the glucagon agonist activity.
- the acylated peptide retains the glucagon antagonist activity.
- the acylated peptide retains GLP-I agonist activity.
- Nonlimiting examples include acylation at positions 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21, 24, 27, 28, or 29 (according to the amino acid numbering of wild type glucagon).
- the acyl group can be covalently linked directly to an amino acid of the glucagon related analog peptide, or indirectly to an amino acid of the glucagon related analog peptide via a spacer, wherein the spacer is positioned between the amino acid of the glucagon related analog peptide and the acyl group.
- Glucagon related analog peptides may be acylated at the same amino acid position where a hydrophilic moiety is linked, or at a different amino acid position.
- Nonlimiting examples include acylation at position 10 (according to the amino acid numbering of the wild type glucagon) and pegylation at one or more positions in the C- terminal portion of the glucagon peptide, e.g., position 24, 28 or 29 (according to the amino acid numbering of the wild type glucagon), within a C-terminal extension, or at the C- terminus (e.g., through adding a C-terminal Cys).
- the glucagon related analog peptide is modified to comprise an acyl group by direct acylation of an amine, hydroxyl, or thiol of a side chain of an amino acid of the glucagon related analog peptide.
- the glucagon related analog peptide is directly acylated through the side chain amine, hydroxyl, or thiol of an amino acid.
- acylation is at position 10, 20, 24, or 29 (according to the amino acid numbering of the wild type glucagon).
- the acylated glucagon related analog peptide can comprise the amino acid sequence of SEQ ID NO : 612, or a modified amino acid sequence thereof comprising one or more of the amino acid modifications described herein, with at least one of the amino acids at positions 10, 20, 24, and 29 (according to the amino acid numbering of the wild type glucagon) modified to any amino acid comprising a side chain amine, hydroxyl, or thiol.
- the direct acylation of the glucagon related analog peptide occurs through the side chain amine, hydroxyl, or thiol of the amino acid at position 10 (according to the amino acid numbering of the wild type glucagon).
- the amino acid comprising a side chain amine is an amino acid of Formula VIII: H H 2 N C COOH
- the amino acid of Formula VIII is the amino acid wherein n is 4 (Lys) or n is 3 (Orn).
- the amino acid comprising a side chain hydroxyl is an amino acid of Formula IX:
- the amino acid of Formula IX is the amino acid wherein n is 1 (Ser).
- the amino acid comprising a side chain thiol is an amino acid of Formula X:
- the amino acid of Formula X is the amino acid wherein n is 1 (Cys).
- the acylated glucagon related analog peptide comprises a spacer between the peptide and the acyl group.
- the glucagon related analog peptide is covalently bound to the spacer, which is covalently bound to the acyl group.
- the glucagon related analog peptide is modified to comprise an acyl group by acylation of an amine, hydroxyl, or thiol of a spacer, which spacer is attached to a side chain of an amino acid at position 10, 20, 24, or 29 (according to the amino acid numbering of the wild type glucagon), or at the C-terminal amino acid of the glucagon related analog peptide.
- the amino acid to which the spacer is attached can be any amino acid comprising a moiety which permits linkage to the spacer.
- an amino acid comprising a side chain NH2, -OH, or -COOH e.g., Lys, Orn, Ser, Asp, or GIu
- the acylated glucagon related analog peptide can comprise the amino acid sequence of SEQ ID NO: 612, or a modified amino acid sequence thereof comprising one or more of the amino acid modifications described herein, with at least one of the amino acids at positions 10, 20, 24, and 29 (according to the amino acid numbering of the wild type glucagon) modified to any amino acid comprising a side chain amine, hydroxyl, or carboxylate.
- the spacer is an amino acid comprising a side chain amine, hydroxyl, or thiol, or a dipeptide or tripeptide comprising an amino acid comprising a side chain amine, hydroxyl, or thiol.
- the acylation can occur through the alpha amine of the amino acid or a side chain amine.
- the spacer amino acid can be any amino acid.
- the spacer amino acid can be a hydrophobic amino acid, e.g., GIy, Ala, VaI, Leu, He, Trp, Met, Phe, Tyr.
- the spacer amino acid can be an acidic residue, e.g., Asp and GIu.
- the spacer amino acid is an amino acid comprising a side chain amine, e.g., an amino acid of Formula VIII (e.g., Lys or Orn).
- an amino acid of Formula VIII e.g., Lys or Orn
- both the alpha amine and the side chain amine of the spacer amino acid to be acylated, such that the glucagon peptide is diacylated.
- Embodiments of the invention include such diacylated molecules.
- the amino acid or one of the amino acids of the dipeptide or tripeptide can be an amino acid of Formula IX.
- the amino acid is Ser.
- the amino acid or one of the amino acids of the dipeptide or tripeptide can be an amino acid of Formula X.
- the amino acid is Cys.
- the spacer comprises a hydrophilic bifunctional spacer.
- the spacer comprises an amino poly(alkyloxy)carboxylate.
- the spacer can comprise, for example, NH 2 (CH 2 CH 2 O) n (CH 2 ) H iCOOH, wherein m is any integer from 1 to 6 and n is any integer from 2 to 12, such as, e.g., 8-amino-3,6- dioxaoctanoic acid, which is commercially available from Peptides International, Inc. (Louisville, KY). Suitable methods of peptide acylation via amines, hydroxy Is, and thiols are known in the art.
- the acyl group of the acylated glucagon related analog peptide can be of any size, e.g., any length carbon chain, and can be linear or branched.
- the acyl group is a C4 to C30 fatty acid.
- the acyl group can be any of a C4 fatty acid, C6 fatty acid, C8 fatty acid, ClO fatty acid, C12 fatty acid, C14 fatty acid, Cl 6 fatty acid, C18 fatty acid, C20 fatty acid, C22 fatty acid, C24 fatty acid, C26 fatty acid, C28 fatty acid, or a C30 fatty acid.
- the acyl group is a C8 to C20 fatty acid, e.g., a C14 fatty acid or a C16 fatty acid.
- the acyl group is a bile acid.
- the bile acid can be any suitable bile acid, including, but not limited to, cholic acid, chenodeoxycholic acid, deoxycholic acid, lithocholic acid, taurocholic acid, glycocholic acid, and cholesterol acid.
- acylated glucagon related analog peptides described herein can be further modified to comprise a hydrophilic moiety.
- the hydrophilic moiety can comprise a polyethylene glycol (PEG) chain.
- PEG polyethylene glycol
- the acylated glucagon related analog peptide can comprise SEQ ID NO: 612, including any of the modifications described herein, in which at least one of the amino acids at position 10, 20, 24, and 29 (according to the amino acid numbering of the wild type glucagon) comprise an acyl group and at least one of the amino acids at position 16, 17, 21, 24, or 29 (according to the amino acid numbering of the wild type glucagon), a position within a C-terminal extension, or the C-terminal amino acid are modified to a Cys, Lys, Orn, homo-Cys, or Ac-Phe, and the side chain of the amino acid is covalently bonded to a hydrophilic moiety (e.g., PEG).
- a hydrophilic moiety e.g., PEG
- the acyl group is attached to position 10 (according to the amino acid numbering of the wild type glucagon), optionally via a spacer comprising Cys, Lys, Orn, homo-Cys, or Ac-Phe, and the hydrophilic moiety is incorporated at a Cys residue at position 24.
- the acylated glucagon related analog peptide can comprise a spacer, wherein the spacer is both acylated and modified to comprise the hydrophilic moiety.
- suitable spacers include a spacer comprising one or more amino acids selected from the group consisting of Cys, Lys, Orn, homo-Cys, and Ac-Phe.
- the glucagon related analog peptide e.g., a Class 1 glucagon related analog peptide, Class 2 glucagon related analog peptide, Class 3 glucagon related analog peptide, Class 4 glucagon peptide, or Class 5 glucagion related peptide
- a Class 1 glucagon related analog peptide Class 2 glucagon related analog peptide
- Class 3 glucagon related analog peptide Class 4 glucagon peptide
- Class 5 glucagion related peptide is modified to comprise an alkyl group which is attached to the glucagon related analog peptide via an ether, thioether, or amino linkage for purposes of prolonging half- life in circulation and/or delaying the onset of and/or extending the duration of action and/or improving resistance to proteases such as DPP-IV.
- Alkylation can be carried out at any positions within the glucagon related analog peptide, including any of positions 1-29, a position within a C-terminal extension, or the C- terminal amino acid, provided that an agonist or antagonist activity of the glucagon related analog peptide with respect to glucagon, GLP-I or other glucagon related analog peptide receptor is retained.
- the alkylated peptide retains glucagon agonist activity is retained.
- the alkylated peptide retains glucagon antagonist activity.
- the alkylated peptide if the unalkylated peptide has GLP-I agonist activity, then the alkylated peptide retains GLP-I agonist activity.
- Nonlimiting examples include alkylation at positions 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21, 24, 27, 28, or 29 (according to the amino acid numbering of wild type glucagon).
- the alkyl group can be covalently linked directly to an amino acid of the glucagon related analog peptide, or indirectly to an amino acid of the glucagon related analog peptide via a spacer, wherein the spacer is positioned between the amino acid of the glucagon related analog peptide and the alkyl group.
- Glucagon related analog peptides may be alkylated at the same amino acid position where a hydrophilic moiety is linked, or at a different amino acid position.
- Nonlimiting examples include alkylation at position 10 (according to the amino acid numbering of wild type glucagon) and pegylation at one or more positions in the C- terminal portion of the glucagon related analog peptide, e.g., position 24, 28 or 29 (according to the amino acid numbering of wild type glucagon), within a C-terminal extension, or at the C-terminus (e.g., through adding a C-terminal Cys).
- the glucagon related analog peptide is modified to comprise an alkyl group by direct alkylation of an amine, hydroxyl, or thiol of a side chain of an amino acid of the glucagon related analog peptide.
- the glucagon related analog peptide is directly alkylated through the side chain amine, hydroxyl, or thiol of an amino acid.
- alkylation is at position 10, 20, 24, or 29 (according to the amino acid numbering of wild type glucagon).
- the alkylated glucagon related analog peptide can comprise the amino acid sequence of SEQ ID NO : 612, or a modified amino acid sequence thereof comprising one or more of the amino acid modifications described herein, with at least one of the amino acids at positions 10, 20, 24, and 29 (according to the amino acid numbering of wild type glucagon) modified to any amino acid comprising a side chain amine, hydroxyl, or thiol.
- the direct alkylation of the glucagon related analog peptide occurs through the side chain amine, hydroxyl, or thiol of the amino acid at position 10 (according to the amino acid numbering of wild type glucagon).
- the amino acid comprising a side chain amine is an amino acid of Formula VIII.
- the amino acid of Formula VIII is the amino acid wherein n is 4 (Lys) or n is 3 (Orn).
- the amino acid comprising a side chain hydroxyl is an amino acid of Formula IX.
- the amino acid of Formula IX is the amino acid wherein n is 1 (Ser).
- the amino acid comprising a side chain thiol is an amino acid of Formula X.
- the amino acid of Formula X is the amino acid wherein n is 1 (Cys).
- the alkylated glucagon related analog peptide comprises a spacer between the peptide and the alkyl group.
- the glucagon related analog peptide is covalently bound to the spacer, which is covalently bound to the alkyl group.
- the glucagon related analog peptide is modified to comprise an alkyl group by alkylation of an amine, hydroxyl, or thiol of a spacer, which spacer is attached to a side chain of an amino acid at position 10, 20, 24, or 29 (according to the amino acid numbering of wild type glucagon) of the glucagon related analog peptide.
- the amino acid to which the spacer is attached can be any amino acid comprising a moiety which permits linkage to the spacer.
- an amino acid comprising a side chain NH 2 , -OH, or -COOH e.g., Lys, Orn, Ser, Asp, or GIu
- the alkylated glucagon related analog peptide can comprise the amino acid sequence of SEQ ID NO: 612, or a modified amino acid sequence thereof comprising one or more of the amino acid modifications described herein, with at least one of the amino acids at positions 10, 20, 24, and 29 (according to the amino acid numbering of wild type glucagon) modified to any amino acid comprising a side chain amine, hydroxyl, or carboxylate.
- the spacer is an amino acid comprising a side chain amine, hydroxyl, or thiol or a dipeptide or tripeptide comprising an amino acid comprising a side chain amine, hydroxyl, or thiol.
- the alkylation can occur through the alpha amine of the amino acid or a side chain amine.
- the spacer amino acid can be any amino acid.
- the spacer amino acid can be a hydrophobic amino acid, e.g., GIy, Ala, VaI, Leu, He, Trp, Met, Phe, Tyr.
- the spacer amino acid can be an acidic residue, e.g., Asp and GIu.
- the spacer amino acid is an amino acid comprising a side chain amine, e.g., an amino acid of Formula VIII (e.g., Lys or Orn).
- a side chain amine e.g., an amino acid of Formula VIII (e.g., Lys or Orn).
- both the alpha amine and the side chain amine of the spacer amino acid to be alkylated, such that the glucagon peptide is dialkylated.
- Embodiments of the invention include such dialkylated molecules.
- the amino acid or one of the amino acids of the spacer can be an amino acid of Formula IX.
- the amino acid is Ser.
- the amino acid or one of the amino acids of the spacer can be an amino acid of Formula X.
- the amino acid is Cys.
- the spacer comprises a hydrophilic bifunctional spacer.
- the spacer comprises an amino poly(alkyloxy)carboxylate.
- the spacer can comprise, for example, NH 2 (CH 2 CH 2 O) n (CH 2 ) TO COOH, wherein m is any integer from 1 to 6 and n is any integer from 2 to 12, such as, e.g., 8-amino-3,6- dioxaoctanoic acid, which is commercially available from Peptides International, Inc. (Louisville, KY). Suitable methods of peptide alkylation via amines, hydroxy Is, and thiols are known in the art.
- a Williamson ether synthesis can be used to form an ether linkage between the glucagon related analog peptide and the alkyl group.
- a nucleophilic substitution reaction of the peptide with an alkyl halide can result in any of an ether, thioether, or amino linkage.
- the alkyl group of the alkylated glucagon related analog peptide can be of any size, e.g., any length carbon chain, and can be linear or branched. In some embodiments of the invention, the alkyl group is a C4 to C30 alkyl.
- the alkyl group can be any of a C4 alkyl, C6 alkyl, C8 alkyl, ClO alkyl, C12 alkyl, C14 alkyl, Cl 6 alkyl, Cl 8 alkyl, C20 alkyl, C22 alkyl, C24 alkyl, C26 alkyl, C28 alkyl, or a C30 alkyl.
- the alkyl group is a C8 to C20 alkyl, e.g., a C14 alkyl or a C16 alkyl.
- the alkyl group comprises a steroid moiety of a bile acid, e.g., cholic acid, chenodeoxycholic acid, deoxycholic acid, lithocholic acid, taurocholic acid, glycocholic acid, and cholesterol acid.
- a bile acid e.g., cholic acid, chenodeoxycholic acid, deoxycholic acid, lithocholic acid, taurocholic acid, glycocholic acid, and cholesterol acid.
- the alkylated glucagon related analog peptides described herein can be further modified to comprise a hydrophilic moiety.
- the hydrophilic moiety can comprise a polyethylene glycol (PEG) chain.
- PEG polyethylene glycol
- the alkylated glucagon related analog peptide can comprise SEQ ID NO: 612, or a modified amino acid sequence thereof comprising one or more of the amino acid modifications described herein, in which at least one of the amino acids at position 10, 20, 24, and 29 (according to the amino acid numbering of wild type glucagon) comprise an alkyl group and at least one of the amino acids at position 16, 17, 21, 24, and 29, a position within a C-terminal extension or the C-terminal amino acid are modified to a Cys, Lys, Orn, homo-Cys, or Ac-Phe, and the side chain of the amino acid is covalently bonded to a hydrophilic moiety (e.g., PEG).
- a hydrophilic moiety e.g., PEG
- the alkyl group is attached to position 10 (according to the amino acid numbering of wild type glucagon), optionally via a spacer comprising Cys, Lys, Orn, homo-Cys, or Ac-Phe, and the hydrophilic moiety is incorporated at a Cys residue at position 24.
- the alkylated glucagon related analog peptide can comprise a spacer, wherein the spacer is both alkylated and modified to comprise the hydrophilic moiety.
- suitable spacers include a spacer comprising one or more amino acids selected from the group consisting of Cys, Lys, Orn, homo-Cys, and Ac-Phe.
- an intramolecular bridge is formed between two amino acid side chains to stabilize the three dimensional structure of the carboxy terminal portion (e.g., amino acids 12-29 (according to the amino acid numbering of wild type glucagon)) of the glucagon related analog peptide.
- the two amino acid side chains can be linked to one another through hydrogen-bonding, ionic interactions, such as the formation of salt bridges, or by covalent bonds.
- the size of the linker is about 8 atoms, or about 7-9 atoms.
- the intramolecular bridge is formed between two amino acids that are two amino acids apart, e.g., amino acids at positions j and j+3, wherein j is any integer between 12 and 26 (e.g., 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, and 26) according to the amino acid numbering of wild type glucagon.
- j is any integer between 12 and 26 (e.g., 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, and 26) according to the amino acid numbering of wild type glucagon.
- ] is 17.
- the size of the linker is about 6 atoms, or about 5 to 7 atoms.
- the intramolecular bridge is formed between two amino acids that are 6 amino acids apart, e.g., amino acids at positions k and k+7, wherein k is any integer between 12 and 22 (e.g., 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, and 22) according to the amino acid numbering of wild type glucagon. In some specific embodiments, k is 12, 13, or 17. In an exemplary embodiment, k is 17.
- amino acid pairings that are capable of covalently bonding to form a seven-atom linking bridge include Orn-Glu (lactam ring); Lys-Asp (lactam); or Homoser- Homoglu (lactone).
- amino acid pairings that may form an eight-atom linker include Lys-Glu (lactam); Homolys-Asp (lactam); Orn-Homoglu (lactam); 4-aminoPhe-Asp (lactam); or Tyr-Asp (lactone).
- One of ordinary skill in the art can envision alternative pairings or alternative amino acid analogs, including chemically modified derivatives, that would create a stabilizing structure of similar size and desired effect.
- a homocysteine- homocysteine disulfide bridge is 6 atoms in length and may be further modified to provide the desired effect.
- the amino acid pairings described above or similar pairings that one of ordinary skill in the art can envision may also provide added stability to the alpha-helix through non-covalent bonds, for example, through formation of salt bridges or hydrogen-bonding interactions.
- the size of a lactam ring can vary depending on the length of the amino acid side chains, and in one embodiment the lactam is formed by linking the side chains of a lysine amino acid to a glutamic acid side chain.
- Further exemplary embodiments include the following pairings, optionally with a lactam bridge: GIu at position 12 with Lys at position 16; native Lys at position 12 with GIu at position 16; GIu at position 16 with Lys at position 20; Lys at position 16 with GIu at position 20; GIu at position 20 with Lys at position 24; Lys at position 20 with GIu at position 24; Lys at position 20 with GIu at position 24; GIu at position 24 with Lys at position 28; Lys at position 24 with GIu at position 28.
- a lactam ring can be formed between the side chains of a Lys 12 and a GIu 16 or alternatively between a GIu 12 and a Lys 16).
- Intramolecular bridges other than a lactam bridge can be used to stabilize the alpha helix of the glucagon related analog peptides.
- the intramolecular bridge is a hydrophobic bridge.
- the intramolecular bridge optionally is between the side chains of two amino acids that are part of the hydrophobic face of the alpha helix of the glucagon related analog peptide.
- one of the amino acids joined by the hydrophobic bridge can be the amino acid at position 10, 14, and 18 (according to the amino acid numbering of wild type glucagon).
- olefin metathesis is used to cross-link one or two turns of the alpha helix of the glucagon related analog peptide using an all-hydrocarbon cross-linking system.
- the glucagon related analog peptide in this instance can comprise ⁇ -methylated amino acids bearing olefinic side chains of varying length and configured with either R or S stereochemistry at the i and i+4 or i+7 positions.
- the olefinic side can comprise (CH 2 )n, wherein n is any integer between 1 to 6. In one embodiment, n is 3 for a cross-link length of 8 atoms. Suitable methods of forming such intramolecular bridges are described in the art.
- the glucagon peptide can comprise O-allyl Ser residues located on adjacent helical turns, which are bridged together via ruthenium-catalyzed ring closing metathesis.
- Such procedures of cross-linking are described in, for example, Blackwell et al., Angew, Chem., Int. Ed. 37: 3281-3284 (1998).
- lanthionine which has been widely adopted as a peptidomimetic of cystine
- Suitable methods of lanthionine -based cyclization are known in the art. See, for instance, Matteucci et al., Tetrahedron Letters 45: 1399-1401 (2004); Mayer et al., J. Peptide Res. 51 : 432-436 (1998); Polinsky et al., J. Med. Chem. 35: 4185-4194 (1992); Osapay et al., J. Med. Chem.
- ⁇ , ⁇ -diaminoalkane tethers e.g., 1 ,4-diaminopropane and 1,5- diaminopentane
- tethers lead to the formation of a bridge 9-atoms or more in length, depending on the length of the diaminoalkane tether. Suitable methods of producing peptides cross-linked with such tethers are described in the art. See, for example, Phelan et al., J. Am. Chem. Soc. 119: 455-460 (1997).
- a disulfide bridge is used to cross-link one or two turns of the alpha helix of the glucagon related analog peptide.
- a modified disulfide bridge in which one or both sulfur atoms are replaced by a methylene group resulting in an isosteric macrocyclization is used to stabilize the alpha helix of the glucagon related analog peptide. Suitable methods of modifying peptides with disulfide bridges or sulfur-based cyclization are described in, for example, Jackson et al., J. Am. Chem. Soc. 113: 9391-9392 (1991) and Rudinger and Jost, Experientia 20: 570-571 (1964).
- the alpha helix of the glucagon related analog peptide is stabilized via the binding of metal atom by two His residues or a His and Cys pair positioned at i and i+4.
- the metal atom can be, for example, Ru(III), Cu(II), Zn(II), or Cd(II).
- metal binding-based alpha helix stabilization are known in the art. See, for example, Andrews and Tabor, Tetrahedron 55: 11711-11743 (1999); Ghadiri et al., J. Am. Chem. Soc. 112: 1630-1632 (1990); and Ghadiri et al., J. Am. Chem. Soc.
- the alpha helix of the glucagon related analog peptide can alternatively be stabilized through other means of peptide cyclizing, which means are reviewed in Davies, J. Peptide. Sci. 9: 471-501 (2003).
- the alpha helix can be stabilized via the formation of an amide bridge, thioether bridge, thioester bridge, urea bridge, carbamate bridge, sulfonamide bridge, and the like.
- a thioester bridge can be formed between the C-terminus and the side chain of a Cys residue.
- a thioester can be formed via side chains of amino acids having a thiol (Cys) and a carboxylic acid (e.g., Asp, GIu).
- a cross- linking agent such as a dicarboxylic acid, e.g. suberic acid (octanedioic acid), etc. can introduce a link between two functional groups of an amino acid side chain, such as a free amino, hydroxyl, thiol group, and combinations thereof.
- the alpha helix of the glucagon related analog peptide is stabilized through the incorporation of hydrophobic amino acids at positions i and i+4.
- i can be Tyr and i+4 can be either VaI or Leu; i can be Phe and i+4 can be Cys or Met; I can be Cys and i+4 can be Met; or i can be Phe and i+4 can be He. It should be understood that, for purposes herein, the above amino acid pairings can be reversed, such that the indicated amino acid at position i could alternatively be located at i+4, while the i+4 amino acid can be located at the i position.
- glucagon related analog peptide is a peptide having glucagon agonist activity, GIP agonist activity, glucagon antagonist and GLP-I activity
- the alpha helix is stabilized through incorporation (either by amino acid substitution or insertion) of one or more alpha helix-stabilizing amino acids at the C-terminal portion of the glucagon related analog peptide (around amino acids 12-29 according to the numbering of the amino acid numbering of wild type glucagon).
- the alpha helix-stabilizing amino acid is an ⁇ , ⁇ -disubstituted amino acid, including, but not limited to any of amino iso-butyric acid (AIB), an amino acid disubstituted with the same or a different group selected from methyl, ethyl, propyl, and n- butyl, or with a cyclooctane or cycloheptane (e.g., 1 -aminocyclooctane- 1 -carboxylic acid).
- AIB amino iso-butyric acid
- an amino acid disubstituted with the same or a different group selected from methyl, ethyl, propyl, and n- butyl or with a cyclooctane or cycloheptane (e.g., 1 -aminocyclooctane- 1 -carboxylic acid).
- one, two, three, four or more of positions 16, 17, 18, 19, 20, 21, 24 or 29 of the glucagon related analog peptide is substituted with an ⁇ , ⁇ -disubstituted amino acid.
- one, two, three or all of positions 16, 20, 21, and 24 are substituted with AIB.
- the present disclosure also encompasses conjugates in which a glucagon related analog peptide (e.g. a Class 1 glucagon related analog peptide, Class 2 glucagon related analog peptide, Class 3 glucagon related analog peptide, Class 4 glucagon related analog peptide, or Class 5 glucagon related analog peptide), is linked, optionally via covalent bonding and optionally via a linker, to a conjugate moiety.
- Linkage can be accomplished by covalent chemical bonds, physical forces such electrostatic, hydrogen, ionic, van der Waals, or hydrophobic or hydrophilic interactions.
- non-covalent coupling systems including biotin-avidin, ligand/receptor, enzyme/substrate, nucleic acid/nucleic acid binding protein, lipid/lipid binding protein, cellular adhesion molecule partners; or any binding partners or fragments thereof which have affinity for each other.
- the glucagon related analog peptide can be linked to conjugate moieties via direct covalent linkage by reacting targeted amino acid residues of the peptide with an organic derivatizing agent that is capable of reacting with selected side chains or the N- or C-terminal residues of these targeted amino acids.
- Reactive groups on the peptide or conjugate moiety include, e.g., an aldehyde, amino, ester, thiol, ⁇ -haloacetyl, maleimido or hydrazino group.
- Derivatizing agents include, for example, maleimidobenzoyl sulfosuccinimide ester (conjugation through cysteine residues), N-hydroxysuccinimide (through lysine residues), glutaraldehyde, succinic anhydride or other agents known in the art.
- the conjugate moieties can be linked to the peptide indirectly through intermediate carriers, such as polysaccharide or polypeptide carriers. Examples of polysaccharide carriers include aminodextran.
- suitable polypeptide carriers include polylysine, polyglutamic acid, polyaspartic acid, co-polymers thereof, and mixed polymers of these amino acids and others, e.g., serines, to confer desirable solubility properties on the resultant loaded carrier.
- Cysteinyl residues most commonly are reacted with ⁇ -haloacetates (and corresponding amines), such as chloroacetic acid or chloroacetamide, to give carboxymethyl or carboxyamidomethyl derivatives. Cysteinyl residues also are derivatized by reaction with bromotrifluoroacetone, alpha-bromo- ⁇ -(5-imidozoyl)propionic acid, chloroacetyl phosphate, N-alkylmaleimides, 3-nitro-2-pyridyl disulfide, methyl 2-pyridyl disulfide, p- chloromercuribenzoate, 2-chloromercuri-4-nitrophenol, or chloro-7-nitrobenzo-2-oxa-l,3- diazole.
- Histidyl residues are derivatized by reaction with diethylpyrocarbonate at pH 5.5-7.0 because this agent is relatively specific for the histidyl side chain.
- Para-bromophenacyl bromide also is useful; the reaction is preferably performed in 0.1 M sodium cacodylate at pH 6.0.
- Lysinyl and amino-terminal residues are reacted with succinic or other carboxylic acid anhydrides. Derivatization with these agents has the effect of reversing the charge of the lysinyl residues.
- Other suitable reagents for derivatizing alpha-amino-containing residues include imidoesters such as methyl picolinimidate, pyridoxal phosphate, pyridoxal, chloroborohydride, trinitrobenzenesulfonic acid, O-methylisourea, 2,4-pentanedione, and transaminase-catalyzed reaction with glyoxylate.
- Arginyl residues are modified by reaction with one or several conventional reagents, among them phenylglyoxal, 2,3-butanedione, 1 ,2-cyclohexanedione, and ninhydrin. Derivatization of arginine residues requires that the reaction be performed in alkaline conditions because of the high pK a of the guanidine functional group. Furthermore, these reagents may react with the groups of lysine as well as the arginine epsilon-amino group.
- tyrosyl residues may be made, with particular interest in introducing spectral labels into tyrosyl residues by reaction with aromatic diazonium compounds or tetranitromethane.
- aromatic diazonium compounds or tetranitromethane Most commonly, N-acetylimidizole and tetranitromethane are used to form O-acetyl tyrosyl species and 3-nitro derivatives, respectively.
- R and R' are different alkyl groups, such as 1- cyclohexyl-3-(2-morpholinyl-4-ethyl) carbodiimide or l-ethyl-3-(4-azonia-4,4- dimethylpentyl) carbodiimide.
- aspartyl and glutamyl residues are converted to asparaginyl and glutaminyl residues by reaction with ammonium ions.
- Sugar(s) may be attached to (a) arginine and histidine, (b) free carboxyl groups, (c) free sulfhydryl groups such as those of cysteine, (d) free hydroxyl groups such as those of serine, threonine, or hydroxyproline, (e) aromatic residues such as those of tyrosine, or tryptophan, or (f) the amide group of glutamine.
- conjugate moieties that can be linked to any of the glucagon related analog peptides described herein include but are not limited to a heterologous peptide or polypeptide (including for example, a plasma protein), a targeting agent, an immunoglobulin or portion thereof (e.g. variable region, CDR, or Fc region), a diagnostic label such as a radioisotope, fluorophore or enzymatic label, a polymer including water soluble polymers, or other therapeutic or diagnostic agents.
- a conjugate comprising a glucagon related analog peptide of the present invention and a plasma protein, wherein the plasma protein is selected from the group consisting of albumin, transferin, fibrinogen and globulins.
- the plasma protein moiety of the conjugate is albumin or transferin.
- the linker comprises a chain of atoms from 1 to about 60, or 1 to 30 atoms or longer, 2 to 5 atoms, 2 to 10 atoms, 5 to 10 atoms, or 10 to 20 atoms long.
- the chain atoms are all carbon atoms.
- the chain atoms in the backbone of the linker are selected from the group consisting of C, O, N, and S. Chain atoms and linkers may be selected according to their expected solubility
- the linker provides a functional group that is subject to cleavage by an enzyme or other catalyst or hydrolytic conditions found in the target tissue or organ or cell.
- the length of the linker is long enough to reduce the potential for steric hindrance. If the linker is a covalent bond or a peptidyl bond and the conjugate is a polypeptide, the entire conjugate can be a fusion protein. Such peptidyl linkers may be any length. Exemplary linkers are from about 1 to 50 amino acids in length, 5 to 50, 3 to 5, 5 to 10, 5 to 15, or 10 to 30 amino acids in length.
- the glucagon related analog peptides are conjugated, e.g., fused to an immunoglobulin or portion thereof (e.g. variable region, CDR, or Fc region).
- immunoglobulins include IgG, IgA, IgE, IgD or IgM.
- the Fc region is a C-terminal region of an Ig heavy chain, which is responsible for binding to Fc receptors that carry out activities such as recycling (which results in prolonged half-life), antibody dependent cell-mediated cytotoxicity (ADCC), and complement dependent cytotoxicity (CDC).
- the human IgG heavy chain Fc region stretches from Cys226 to the C-terminus of the heavy chain.
- the "hinge region” generally extends from Glu216 to Pro230 of human IgGl (hinge regions of other IgG isotypes may be aligned with the IgGl sequence by aligning the cysteines involved in cysteine bonding).
- the Fc region of an IgG includes two constant domains, CH2 and CH3.
- the CH2 domain of a human IgG Fc region usually extends from amino acids 231 to amino acid 341.
- the CH3 domain of a human IgG Fc region usually extends from amino acids 342 to 447.
- the Fc region may comprise one or more native or modified constant regions from an immunoglobulin heavy chain, other than CHl, for example, the CH2 and CH3 regions of IgG and IgA, or the CH3 and CH4 regions of IgE.
- Suitable conjugate moieties include portions of immunoglobulin sequence that include the FcRn binding site.
- FcRn a salvage receptor, is responsible for recycling immunoglobulins and returning them to circulation in blood.
- the region of the Fc portion of IgG that binds to the FcRn receptor has been described based on X-ray crystallography (Burmeister et al. 1994, Nature 372:379).
- the major contact area of the Fc with the FcRn is near the junction of the CH2 and CH3 domains.
- Fc-FcRn contacts are all within a single Ig heavy chain.
- the major contact sites include amino acid residues 248, 250-257, 272, 285, 288, 290-291, 308-311, and 314 of the CH2 domain and amino acid residues 385-387, 428, and 433-436 of the CH3 domain.
- Fc ⁇ R are responsible for ADCC and CDC.
- positions within the Fc region that make a direct contact with Fc ⁇ R are amino acids 234-239 (lower hinge region), amino acids 265-269 (B/C loop), amino acids 297-299 (C7E loop), and amino acids 327-332 (F/G) loop (Sondermann et al., Nature 406: 267-273, 2000).
- the lower hinge region of IgE has also been implicated in the FcRI binding (Henry, et al., Biochemistry 36, 15568-15578, 1997).
- Amino acid modifications may be made to the Fc region of an immunoglobulin.
- Such variant Fc regions comprise at least one amino acid modification in the CH3 domain of the Fc region (residues 342-447) and/or at least one amino acid modification in the CH2 domain of the Fc region (residues 231-341).
- Mutations believed to impart an increased affinity for FcRn include T256A, T307A, E380A, and N434A (Shields et al. 2001, J. Biol. Chem. 276:6591).
- Other mutations may reduce binding of the Fc region to Fc ⁇ RI, Fc ⁇ RIIA, Fc ⁇ RIIB, and/or Fc ⁇ RIIIA without significantly reducing affinity for FcRn.
- substitution of the Asn at position 297 of the Fc region with Ala or another amino acid removes a highly conserved N-glycosylation site and may result in reduced immunogenicity with concomitant prolonged half- life of the Fc region, as well as reduced binding to Fc ⁇ Rs (Routledge et al. 1995, Transplantation 60:847; Friend et al. 1999, Transplantation 68:1632; Shields et al. 1995, J. Biol. Chem. 276:6591).
- Amino acid modifications at positions 233-236 of IgGl have been made that reduce binding to Fc ⁇ Rs (Ward and Ghetie 1995, Therapeutic Immunology 2:77 and Armour et al. 1999, Eur. J. Immunol. 29:2613).
- Some exemplary amino acid substitutions are described in US Patents 7,355,008 and 7,381,408, each incorporated by reference herein in its entirety.
- a glucagon related analog peptide may comprise a C-terminus or a C-terminal amino acid sequence including but not limited to: COOH, CONH 2 , GPSSGAPPPS (SEQ ID NO: 624), GPSSGAPPPS-CONH 2 (SEQ ID NO: 723), a oxyntomodulin carboxy terminal extension, KRNRNNIA (SEQ ID NO: 625) or KGKKNDWKHNITQ (SEQ ID NO: 662).
- the terminal ten amino acids of Exendin-4 i.e.
- SEQ ID NO: 624 the sequence of SEQ ID NO: 624 (GPSSGAPPPS) are linked to the carboxy terminus of the Class 1 glucagon related analog peptide, Class 2 glucagon related analog peptide, Class 3 glucagon related analog peptide, Class 4 glucagon related analog peptide, or Class 5 glucagon related analog peptide of the present disclosure.
- Oxyntomodulin is a 37 amino acid peptide (SEQ ID NO: 661) that contains the 29 amino acid sequence of glucagon followed by an 8 amino acid carboxy terminal extension of SEQ ID NO: 625 (KRNRNNIA). Accordingly, in one embodiment prodrug derivatives of glucagon related analog peptides are provided that further comprise the carboxy terminal extension of the sequence of SEQ ID NO: 625 or a four amino acid extension having the sequence KRNR.
- glucagon related analog peptides and prodrugs of this disclosed herein may be prepared by standard synthetic methods, recombinant DNA techniques, or any other methods of preparing peptides and fusion proteins. Although certain non-natural amino acids cannot be expressed by standard recombinant DNA techniques, techniques for their preparation are known in the art. Compounds of this invention that encompass non-pep tide portions may be synthesized by standard organic chemistry reactions, in addition to standard peptide chemistry reactions when applicable.
- glucagon related analog peptides are described in detail below. With respect to each of the sections of disclosure concerning class 1, 2, 3, 4 and 5 glucagon related analog peptides modifications are described with respect to the glucagon related analog peptide portion of a prodrug compound detailed above. Thus, structural elements described with regard to a class of glucagon related analog peptides are structural elements of a bioactive peptide, polypeptide, or protein which is then further modified to generate a prodrug compound as described above.
- the glucagon related analog peptide is a Class 1 glucagon related analog peptide, which is described herein and in International Patent Application Publication No. WO 2008/086086, published on July 17, 2008, and U.S. Provisional Application No. 61/090,415, the contents of which are incorporated by reference in their entirety.
- Class 1 glucagon peptides retain glucagon receptor activity relative to the native glucagon peptide (SEQ ID NO: 701).
- the glucagon peptide can retain at least 10%, 20%, 30%, 40%, 50%, 60%, 70%, 75% activity, 80% activity, 85% activity, or 90% of the activity of native glucagon (calculated as the inverse ratio of EC50s for the glucagon peptide vs. glucagon, e.g., as measured by cAMP production using the assay generally described in Example 2).
- the Class 1 glucagon related analog peptides have the same or greater activity (used synonymously with the term "potency" herein) than glucagon.
- any of the Class 1 glucagon related analog peptides described herein may exhibit an EC50 at the human glucagon receptor of about 100 nM, 75 nM, 50 nM, 40 nM, 30 nM, 20 nM, 10 nM, 5 nM, 1 nM or less when tested for cAMP induction in HEK293 cells over- expressing glucagon receptor, e.g. using the assay of Example 2.
- pegylated peptides will exhibit a higher EC50 compared to the unpegylated peptide.
- the Class 1 glucagon related analog peptides exhibit less than about 5%, 4%, 3%, 2% or 1% of the activity of native GLP-I at the GLP-I receptor and/or a greater than about 5-fold, 10-fold, or 15-fold selectivity for glucagon receptor compared to GLP-I receptor.
- the Class 1 glucagon related analog peptides exhibit less than 5% of the activity of native GLP-I at the GLP-I receptor and exhibit a greater than 5-fold selectivity for glucagon receptor compared to GLP-I receptor.
- Native glucagon exhibits poor solubility in aqueous solution, particularly at physiological pH, with a tendency to aggregate and precipitate over time.
- Class 1 glucagon related analog peptides in one embodiment exhibit at least 2-fold, 5 -fold, or even higher solubility compared to native glucagon at a pH between 6 and 8, or between 6 and 9, for example, at pH 7 after 24 hours at 25°C.
- a Class 1 glucagon related analog peptide has been modified relative to the wild type peptide of His-Ser-Gln-Gly-Thr-Phe- Thr-Ser-Asp-Tyr- Ser-Lys-Tyr-Leu-Asp-Ser- Arg-Arg-Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu- Met-Asn-Thr (SEQ ID NO: 701) to improve the peptide's solubility in aqueous solutions, particularly at a pH ranging from about 5.5 to about 8.0, while retaining the native peptide's biological activity.
- the solubility of any of the Class 1 glucagon related analog peptides described herein can be further improved by attaching a hydrophilic moiety to the peptide.
- Hydrophilic moieties are further described herein.
- solubility is improved by adding charge to the Class 1 glucagon related analog peptide by the substitution of native non-charged amino acids with charged amino acids selected from the group consisting of lysine, arginine, histidine, aspartic acid and glutamic acid, or by the addition of charged amino acids to the amino or carboxy terminus of the peptide.
- the Class 1 glucagon related analog peptide has improved solubility due to the fact that the peptide is modified by amino acid substitutions and/or additions that introduce a charged amino acid into the C -terminal portion of the peptide, and in one embodiment at a position C-terminal to position 27 of SEQ ID NO: 701.
- one, two or three charged amino acids may be introduced within the C-terminal portion, and in one embodiment C-terminal to position 27.
- the native amino acid(s) at positions 28 and/or 29 are substituted with a charged amino acid, and/or one to three charged amino acids are added to the C-terminus of the peptide, e.g. after position 27, 28 or 29.
- the Class 1 glucagon related analog peptide may comprise any one or two of the following modifications: substitution of N28 with E; substitution of N28 with D; substitution of T29 with D; substitution of T29 with E; insertion of E after position 27, 28 or 29; insertion of D after position 27, 28 or 29.
- substitution of N28 with E substitution of N28 with D
- substitution of T29 with D substitution of T29 with E
- substitution of T29 with E substitution of T29 with E
- insertion of E after position 27, 28 or 29 insertion of D after position 27, 28 or 29.
- the Class 1 glucagon related analog peptide comprises an amino acid sequence of SEQ ID NO: 711, or an analog thereof that contains 1 to 3 further amino acid modifications (described herein in reference to glucagon agonists) relative to native glucagon, or a glucagon agonist analog thereof.
- SEQ ID NO: 711 represents a modified Class 1 glucagon related analog peptide, wherein the asparagine residue at position 28 of the native protein has been substituted with an aspartic acid.
- the Class 1 glucagon related analog peptide comprises an amino acid sequence of SEQ ID NO: 738, wherein the asparagine residue at position 28 of the native protein has been substituted with glutamic acid.
- Other exemplary embodiments include Class 1 glucagon related analog peptides of SEQ ID NOS: 724, 725, 726, 733, 735, 736 and 737.
- Class 1 glucagon peptides of one embodiment retain glucagon activity and exhibit at least 2-fold, 5-fold, 10- fold, 15-fold, 25-fold, 30-fold or greater solubility relative to native glucagon at a given pH between about 5.5 and 8, e.g., pH 7, when measured after 24 hours at 25°C. Additional modifications, e.g. conservative substitutions, which modifications are further described herein, may be made to the Class 1 glucagon related analog peptide that still allow it to retain glucagon activity.
- Any of the Class 1 glucagon peptides may additionally exhibit improved stability and/or reduced degradation, for example, retaining at least 95% of the original peptide after 24 hours at 25°C.
- Any of the Class 1 glucagon related analog peptides disclosed herein may additionally exhibit improved stability at a pH within the range of 5.5 to 8, for example, retaining at least 75%, 80%, 90%, 95%, 96%, 97%, 98% or 99% of the original peptide after 24 hours at 25 T!.
- the Class 1 glucagon related analog peptides may include additional modifications that alter its pharmaceutical properties, e.g. increased potency, prolonged half- life in circulation, increased shelf- life, reduced precipitation or aggregation, and/or reduced degradation, e.g., reduced occurrence of cleavage or chemical modification after storage.
- any of the foregoing Class 1 glucagon related analog peptides can be further modified to improve stability by modifying the amino acid at position 15 of SEQ ID NO: 701 to reduce degradation of the peptide over time, especially in acidic or alkaline buffers.
- Asp at position 15 is substituted with a GIu, homo-Glu, cysteic acid, or homo-cysteic acid.
- any of the Class 1 glucagon related analog peptides described herein can be further modified to improve stability by modifying the amino acid at position 16 of SEQ ID NO: 701.
- Ser at position 16 is substituted with Thr or AIB, or any of the amino acids substitutions described herein with regard to Class 1 glucagon related analog peptides which enhance potency at the glucagon receptor.
- Such modifications reduce cleavage of the peptide bond between Aspl5-Serl6.
- any of the Class 1 glucagon related analog peptides described herein can be further modified to reduce degradation at various amino acid positions by modifying any one, two, three, or all four of positions 20, 21, 24, or 27.
- Exemplary embodiments include substitution of GIn at position 20 with Ala or AIB, substitution of Asp at position 21 with GIu, substitution of GIn at position 24 with Ala or AIB, substitution of Met at position 27 with Leu or NIe.
- Removal or substitution of methionine reduces degradation due to oxidation of the methionine.
- Removal or substitution of GIn or Asn reduces degradation due to deamidation of GIn or Asn.
- Removal or substitution of Asp reduces degradation that occurs through dehydration of Asp to form a cyclic succinimide intermediate followed by isomerization to iso-aspartate.
- Class 1 glucagon related analog peptides are provided that have enhanced potency at the glucagon receptor, wherein the peptides comprise an amino acid modification at position 16 of native glucagon (SEQ ID NO: 701).
- enhanced potency can be provided by substituting the naturally occurring serine at position 16 with glutamic acid or with another negatively charged amino acid having a side chain with a length of 4 atoms, or alternatively with any one of glutamine, homoglutamic acid, or homocysteic acid, or a charged amino acid having a side chain containing at least one heteroatom, (e.g. N, O, S, P) and with a side chain length of about 4 (or 3-5) atoms.
- heteroatom e.g. N, O, S, P
- Substitution of serine at position 16 with glutamic acid enhances glucagon activity at least 2-fold, 4-fold, 5 -fold and up to 10-fold greater at the glucagon receptor.
- the Class 1 glucagon related analog peptide retains selectivity for the glucagon receptor relative to the GLP-I receptors, e.g., at least 5-fold, 10-fold, or 15-fold selectivity.
- the Class 1 glucagon peptides disclosed herein are further modified at position 1 or 2 to reduce susceptibility to cleavage by dipeptidyl peptidase IV. More particularly, in some embodiments, position 1 and/or position 2 of the Class 1 glucagon related analog peptide is substituted with the DPP-IV resistant amino acid(s) described herein. In one embodiment, position 2 of the analog peptide is substituted with an amino isobutyric acid. In one embodiment, position 2 of the analog peptide is substituted with an amino acid selected from the group consisting of D-serine, D-alanine, glycine, N-methyl serine, and ⁇ -amino butyric acid. In another embodiment, position 2 of the Class 1 glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-serine, glycine, and aminoisobutyric acid.
- Reduction in glucagon activity upon modification of the amino acids at position 1 and/or position 2 of the glucagon peptide can be restored by stabilization of the alpha-helix structure in the C-terminal portion of the glucagon peptide (around amino acids 12-29).
- the alpha helix structure can be stabilized by, e.g., formation of a covalent or non-covalent intramolecular bridge (e.g., a lactam bridge between side chains of amino acids at positions "i" and "i+4", wherein i is an integer from 12 to 25), substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid (e.g., an ⁇ , ⁇ - disubstituted amino acid), as further described herein.
- a covalent or non-covalent intramolecular bridge e.g., a lactam bridge between side chains of amino acids at positions "i" and "i+4", wherein i is an integer from 12 to 25
- substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid e.g., an ⁇ , ⁇ - disubstituted amino acid
- Glucagon receptor activity can be reduced by an amino acid modification at position
- Enhanced activity at the GLP-I receptor is provided by replacing the carboxylic acid of the C-terminal amino acid with a charge-neutral group, such as an amide or ester. Conversely, retaining the native carboxylic acid at the C-terminus of the peptide maintains the relatively greater selectivity of the Class 1 glucagon related analog peptide for the glucagon receptor vs. the GLP-I receptor (e.g., greater than about 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20-fold).
- Class 1 glucagon related analog peptide may further increase solubility and/or stability and/or glucagon activity.
- the Class 1 glucagon related analog peptide may alternatively comprise other modifications that do not substantially affect solubility or stability, and that do not substantially decrease glucagon activity.
- the Class 1 glucagon related analog peptide may comprise a total of up to l l, or up to 12, or up to 13, or up to 14 amino acid modifications relative to the native glucagon sequence. For example, conservative or non-conservative substitutions, additions or deletions may be carried out at any of positions 2, 5, 7, 10, 11, 12, 13, 14, 17, 18, 19, 20, 21, 24, 27, 28 or 29.
- Exemplary modifications of the Class 1 glucagon related analog peptide include but are not limited to: (a) non-conservative substitutions, conservative substitutions, additions or deletions while retaining at least partial glucagon agonist activity, for example, conservative substitutions at one or more of positions 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21, 24, 27, 28 or 29, substitution of Tyr at position 10 with VaI or Phe, substitution of Lys at position 12 with Arg, substitution of one or more of these positions with Ala;
- exemplary modifications of the Class 1 glucagon related analog peptide include at least one amino acid modification selected from Group A and one or more amino acid modifications selected from Group B and/or Group C, wherein Group A is: substitution of Asn at position 28 with a charged amino acid; substitution of Asn at position 28 with a charged amino acid selected from the group consisting of Lys, Arg, His, Asp, GIu, cysteic acid, and homocysteic acid; substitution at position 28 with Asn, Asp, or GIu; substitution at position 28 with Asp; substitution at position 28 with GIu; substitution of Thr at position 29 with a charged amino acid; substitution of Thr at position 29 with a charged amino acid selected from the group consisting of Lys, Arg, His, Asp, GIu, cysteic acid, and homocysteic acid; substitution at position 29 with Asp, GIu, or Lys; substitution at position 29 with GIu; insertion of 1-3 charged amino acids after position 29; insertion after position 29 of
- Lys at position 12 is substituted with Arg.
- amino acids at positions 29 and/or 28, and optionally at position 27, are deleted.
- the glucagon peptide comprises (a) an amino acid modification at position 1 and/or 2 that confers DPP-IV resistance, e.g., substitution with DMIA at position 1, or AIB at position 2, (b) an intramolecular bridge within positions 12-29, e.g.
- amino acids at positions 16 and 20, or one or more substitutions of the amino acids at positions 16, 20, 21, and 24 with an ⁇ , ⁇ disubstituted amino acid optionally (c) linked to a hydrophilic moiety such as PEG, e.g., through Cys at position 24, 29 or at the C-terminal amino acid, optionally (d) an amino acid modification at position 27 that substitutes Met with, e.g., NIe, optionally (e) amino acid modifications at positions 20, 21 and 24 that reduce degradation, and optionally (f) linked to SEQ ID NO: 720.
- a hydrophilic moiety such as PEG, e.g., through Cys at position 24, 29 or at the C-terminal amino acid
- amino acid modification at position 27 that substitutes Met with, e.g., NIe
- amino acid modifications at positions 20, 21 and 24 that reduce degradation optionally (f) linked to SEQ ID NO: 720.
- the glucagon peptide comprises (a) Asp28Glu29, or Glu28Glu29, or Glu29Glu30, or Glu28Glu30 or Asp28Glu30, and optionally (b) an amino acid modification at position 16 that substitutes Ser with, e.g. Thr or AIB, and optionally (c) an amino acid modification at position 27 that substitutes Met with, e.g., NIe, and optionally (d) amino acid modifications at positions 20, 21 and 24 that reduce degradation.
- the glucagon peptide is Tl 6, A20, E21, A24, Nle27, D28, E29.
- the Class 1 glucagon related analog peptide comprises the amino acid sequence:
- Xl-X2-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Ser-Arg-Arg-Ala-Gln- Asp-Phe-Val-Gln-Trp-Leu-Met-Z (SEQ ID NO: 739) with 1 to 3 amino acid modifications thereto, wherein Xl and/or X2 is a non- native amino acid that reduces susceptibility of (or increases resistance of) the glucagon peptide to cleavage by dipeptidyl peptidase IV (DPP- IV), wherein Z is selected from the group consisting of -COOH (the naturally occurring C- terminal carboxylate), -Asn-COOH, Asn-Thr-COOH, and Y-COOH, wherein Y is 1 to 2 amino acids, and wherein an intramolecular bridge, preferably a covalent bond, connects the side chains of an amino acid
- the intramolecular bridge is a lactam bridge.
- the amino acids at positions i and i+4 of SEQ ID NO: 739 are Lys and GIu, e.g., Glul ⁇ and Lys20.
- Xl is selected from the group consisting of: D-His, N-methyl-His, alpha-methyl-His, imidazole acetic acid, des-amino-His, hydroxyl-His, acetyl-His, homo-His, and alpha, alpha-dimethyl imidiazole acetic acid (DMIA).
- X2 is selected from the group consisting of: D-Ser, D-AIa, GIy, N-methyl-Ser, VaI, and alpha, amino isobutyric acid (AIB).
- the glucagon peptide is covalently linked to a hydrophilic moiety at any of amino acid positions 16, 17, 20, 21, 24, 29, 40, within a C-terminal extension, or at the C-terminal amino acid.
- this hydrophilic moiety is covalently linked to a Lys, Cys, Orn, homocysteine, or acetyl-phenylalanine residue at any of these positions.
- Exemplary hydrophilic moieties include polyethylene glycol (PEG), for example, of a molecular weight of about 1,000 Daltons to about 40,000 Daltons, or about 20,000 Daltons to about 40,000 Daltons.
- the Class I glucagon related analog peptide comprises the amino acid sequence: Xl-X2-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Ser-Arg-Arg-Ala-Gln- Asp-Phe-Val-Gln-Trp-Leu-Met-Z (SEQ ID NO: 739), wherein Xl and/or X2 is a non- native amino acid that reduces susceptibility of (or increases resistance of) the glucagon peptide to cleavage by dipeptidyl peptidase IV (DPP- IV), wherein one, two, three, four or more of positions 16, 20, 21, and 24 of the glucagon peptide is substituted with an ⁇ , ⁇ -disubstituted amino acid, and wherein Z is selected from the group consisting of -COOH (the naturally occurring C- terminal carboxylate),
- Exemplary further amino acid modifications to the foregoing Class 1 glucagon related analog peptides or analogs include substitution of Lys at position 12 with Arg; substitution of Asp at position 15 with GIu; substitution of Ser at position 16 with Thr or AIB; substitution of GIn at position 20 with Ala or AIB; substitution of Asp at position 21 with GIu; substitution of GIn at position 24 with Ala or AIB; substitution of Met at position 27 with Leu or NIe; substitution of Asn at position 28 with a charged amino acid; substitution of Asn at position 28 with a charged amino acid selected from the group consisting of Lys, Arg, His, Asp, GIu, cysteic acid, and homocysteic acid; substitution at position 28 with Asn, Asp, or GIu; substitution at position 28 with Asp; substitution at position 28 with GIu; substitution of Thr at position 29 with a charged amino acid; substitution of Thr at position 29 with a charged amino acid selected from the group consisting of Lys, Arg, His,
- Class 1 glucagon related analog peptides can be prepared that retain at least 20% of the activity of native glucagon at the glucagon receptor, and which are soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, (e.g. pH 7), and optionally retain at least 95% of the original peptide (e.g. 5% or less of the original peptide is degraded or cleaved) after 24 hours at 25°C.
- high potency Class 1 glucagon peptides can be prepared that exhibit at least about 100%, 125%, 150%, 175%, 200%, 250%, 300%, 350%, 400%, 450%, 500%, 600%, 700%, 800%, 900% or 10-fold or more of the activity of native glucagon at the glucagon receptor, and optionally are soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, (e.g. pH 7), and optionally retains at least 95% of the original peptide (e.g. 5% or less of the original peptide is degraded or cleaved) after 24 hours at 25°C.
- the native glucagon peptide of SEQ ID NO: 701 is modified by the substitution of the native amino acid at position 28 and/or 29 with a negatively charged amino acid (e.g., aspartic acid or glutamic acid) and optionally the addition of a negatively charged amino acid (e.g., aspartic acid or glutamic acid) to the carboxy terminus of the peptide.
- a negatively charged amino acid e.g., aspartic acid or glutamic acid
- a negatively charged amino acid e.g., aspartic acid or glutamic acid
- the native glucagon peptide of SEQ ID NO: 701 is modified by the substitution of the native amino acid at position 29 with a positively charged amino acid (e.g., lysine, arginine or histidine) and optionally the addition of one or two positively charged amino acid (e.g., lysine, arginine or histidine) on the carboxy terminus of the peptide.
- a positively charged amino acid e.g., lysine, arginine or histidine
- one or two positively charged amino acid e.g., lysine, arginine or histidine
- a Class 1 glucagon related analog peptide having improved solubility and stability wherein the peptide comprises the amino acid sequence of SEQ ID NO: 734 with the proviso that at least one amino acids at position, 28, or 29 is substituted with an acidic amino acid and/or an additional acidic amino acid is added at the carboxy terminus of SEQ ID NO: 734.
- the acidic amino acids are independently selected from the group consisting of Asp, GIu, cysteic acid and homocysteic acid.
- a Class 1 glucagon related analog peptide having improved solubility and stability wherein the agonist comprises the amino acid sequence of SEQ ID NO: 733, wherein at least one of the amino acids at positions 27, 28 or 29 is substituted with a non-native amino acid residue (i.e. at least one amino acid present at position 27, 28 or 29 of the analog is an acid amino acid different from the amino acid present at the corresponding position in SEQ ID NO: 701).
- a Class 1 glucagon related analog peptide comprising the sequence of SEQ ID NO: 733 with the proviso that when the amino acid at position 28 is asparagine and the amino acid at position 29 is threonine, the peptide further comprises one to two amino acids, independently selected from the group consisting of Lys, Arg, His, Asp or GIu, added to the carboxy terminus of the glucagon peptide.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 733 wherein 1 to 6 amino acids, selected from positions 1, 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21 or 24 of the analog differ from the corresponding amino acid of SEQ ID NO: 701.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 733 is provided wherein 1 to 3 amino acids selected from positions 1, 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21 or 24 of the peptide differ from the corresponding amino acid of SEQ ID NO: 701.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 707, SEQ ID NO: 708 or SEQ ID NO: 734 is provided wherein 1 to 2 amino acids selected from positions 1, 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21 or 24 of the peptide differ from the corresponding amino acid of SEQ ID NO: 701 , and in a further embodiment those one to two differing amino acids represent conservative amino acid substitutions relative to the amino acid present in the native sequence (SEQ ID NO: 701).
- a Class 1 glucagon related analog peptide of SEQ ID NO: 711 or SEQ ID NO: 713 wherein the glucagon peptide further comprises one, two or three amino acid substitutions at positions selected from positions 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21, 24, 27 or 29. In one embodiment the substitutions at positions 2, 5, 7, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 27 or 29 are conservative amino acid substitutions.
- a Class 1 glucagon related analog peptide comprising an analog peptide of SEQ ID NO: 701 wherein the peptide differs from SEQ ID NO: 701 by having an amino acid other than serine at position 2 and by having an acidic amino acid substituted for the native amino acid at position 28 or 29 or an acidic amino acid added to the carboxy terminus of the peptide of SEQ ID NO: 701.
- the acidic amino acid is aspartic acid or glutamic acid.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 709, SEQ ID NO: 712, SEQ ID NO: 713 or SEQ ID NO: 732 wherein the peptide differs from the parent molecule by a substitution at position 2. More particularly, position 2 of the peptide is substituted with an amino acid selected from the group consisting of D-serine, alanine, D-alanine, glycine, n-methyl serine and amino isobutyric acid.
- a Class 1 glucagon related analog peptide comprising a peptide of SEQ ID NO: 701 wherein the peptide differs from SEQ ID NO: 701 by having an amino acid other than histidine at position 1 and by having an acidic amino acid substituted for the native amino acid at position 28 or 29 or an acidic amino acid added to the carboxy terminus of the peptide of SEQ ID NO: 701.
- the acidic amino acid is aspartic acid or glutamic acid.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 709, SEQ ID NO: 712, SEQ ID NO: 713 or SEQ ID NO: 732 wherein the peptide differs from the parent molecule by a substitution at position 1. More particularly, position 1 of the peptide is substituted with an amino acid selected from the group consisting of DMIA, D-histidine, desaminohistidine, hydroxyl-histidine, acetyl- histidine and homo-histidine.
- the Class 1 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 709, SEQ ID NO: 712, SEQ ID NO: 713 and SEQ ID NO: 732.
- a Class 1 glucagon related analog peptide is provided comprising a sequence of SEQ ID NO: 709, SEQ ID NO: 712, SEQ ID NO: 713 or SEQ ID NO: 732 further comprising one to two amino acids, added to the C-terminus of SEQ ID NO: 709, SEQ ID NO: 712, SEQ ID NO: 713 or SEQ ID NO: 732, wherein the additional amino acids are independently selected from the group consisting of Lys, Arg, His, Asp GIu, cysteic acid or homocysteic acid.
- the additional amino acids added to the carboxy terminus are selected from the group consisting of Lys, Arg, His, Asp or GIu or in a further embodiment the additional amino acids are
- the Class 1 glucagon related analog peptide comprises the sequence of SEQ ID NO: 707 or a glucagon agonist analog thereof.
- the peptide comprising a sequence selected from the group consisting of SEQ ID NO: 708, SEQ ID NO: 710, SEQ ID NO: 711, SEQ ID NO: 712 and SEQ ID NO: 713.
- the peptide comprising a sequence selected from the group consisting of SEQ ID NO: 708, SEQ ID NO: 710 and SEQ ID NO: 711.
- the Class 1 glucagon related analog peptide comprises the sequence of SEQ ID NO: 708, SEQ ID NO: 710 and SEQ ID NO: 711 further comprising an additional amino acid, selected from the group consisting of Asp and GIu, added to the C-terminus of the glucagon peptide.
- the Class 1 glucagon related analog peptide comprises the sequence of SEQ ID NO: 711 or SEQ ID NO: 713, and in a further embodiment the glucagon peptide comprises the sequence of SEQ ID NO: 711.
- a Class 1 glucagon related analog peptide comprising a modified glucagon peptide selected from the group consisting of:
- Xaa at position 15 is Asp, GIu, cysteic acid, homoglutamic acid or homocysteic acid
- the Xaa at position 28 is Asn or an acidic amino acid
- the Xaa at position 29 is Thr or an acidic amino acid
- R is an acidic amino acid, COOH or CONH 2 , with the proviso that an acidic acid residue is present at one of positions 28, 29 or 30.
- R is COOH
- R is CONH 2 .
- the Class 1 glucagon related analog peptide may be a fusion glucagon peptide comprising a glucagon agonist analog comprising a glucagon peptide NH 2 - His-Ser-Gln-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Xaa-Ser-Arg-Arg-Ala-Gln- Asp-Phe-Val-Gln-Trp-Leu-Xaa-Xaa-Xaa-R (SEQ ID NO: 734), wherein R is an acidic amino acid or a bond and an amino acid sequence of SEQ ID NO: 720 (GPSSGAPPPS), SEQ ID NO: 721 (KRNRNNIA) or SEQ ID NO: 722 (KRNR) linked to the carboxy terminal amino acid of the glucagon peptide sequence.
- a glucagon agonist analog comprising a glucagon peptid
- the glucagon peptide is selected from the group consisting of SEQ ID NO: 733, SEQ ID NO: 707 or SEQ ID NO: 708 further comprising an amino acid sequence of SEQ ID NO: 720 (GPSSGAPPPS), SEQ ID NO: 721 (KRNRNNIA) or SEQ ID NO: 722 (KRNR) linked to the carboxy terminal amino acid of the glucagon peptide sequence.
- the glucagon fusion peptide comprises SEQ ID NO: 702, SEQ ID NO: 703, SEQ ID NO: 704, SEQ ID NO: 705 and SEQ ID NO: 706 or a glucagon agonist analog thereof, further comprising an amino acid sequence of SEQ ID NO: 720 (GPSSGAPPPS), SEQ ID NO: 721 (KRNRNNIA) or SEQ ID NO: 722 (KRNR) linked to amino acid 29 of the glucagon fusion peptide sequence.
- the fusion peptide further comprises a PEG chain linked to an amino acid at position 16, 17, 21, 24, 29, within a C-terminal extension, or at the C- terminal amino acid, wherein the PEG chain is selected from the range of 500 to 40,000
- amino acid sequence of SEQ ID NO: 720 (GPSSGAPPPS), SEQ ID NO: 721 (KRNRNNIA) or SEQ ID NO: 722 (KRNR) is bound to amino acid 29 of the glucagon peptide through a peptide bond.
- the glucagon peptide portion of the glucagon fusion peptide comprises a sequence selected from the group consisting of SEQ ID NO: 710, SEQ ID NO: 711 and SEQ ID NO: 713.
- the glucagon peptide portion of the glucagon fusion peptide comprises the sequence of SEQ ID NO: 711 or SEQ ID NO: 713, wherein a PEG chain is linked at position 21, 24, 29, within a C-terminal extension or at the C-terminal amino acid, respectively.
- the glucagon peptide sequence of the fusion peptide comprises the sequence of SEQ ID NO: 711, further comprising an amino acid sequence of SEQ ID NO: 720 (GPSSGAPPPS), SEQ ID NO: 721 (KRNRNNIA) or SEQ ID NO: 722 (KRNR) linked to amino acid 29 of the glucagon peptide.
- the glucagon fusion peptide comprises a sequence selected from the group consisting of SEQ ID NO: 724, SEQ ID NO: 725 and SEQ ID NO: 726.
- the fusion peptides of the present invention will have a C-terminal amino acid with the standard carboxylic acid group.
- the fusion glucagon peptide comprises a glucagon agonist analog selected from the group consisting of SEQ ID NO: 710, SEQ ID NO: 711 and SEQ ID NO: 713, further comprising an amino acid sequence of SEQ ID NO: 723 (GPSSGAPPPS-CONH 2 ) linked to amino acid 29 of the glucagon peptide.
- Exemplary glucagon peptides are selected from the group consisting of SEQ ID NO: 702, SEQ ID NO: 703, SEQ ID NO: 704, SEQ ID NO: 705, SEQ ID NO: 706, SEQ ID NO: 707, SEQ ID NO: 708, SEQ ID NO: 709, SEQ ID NO: 710, SEQ ID NO: 711 and SEQ ID NO: 733, wherein amino acid 29 of the glucagon peptide is bound to a second peptide through a peptide bond, and said second peptide comprises the sequence of SEQ ID NO: 720, SEQ ID NO: 721 or SEQ ID NO: 722.
- the glucagon peptide is pegylated.
- the Class 1 glucagon related analog peptides described herein can be further modified to improve the peptide's solubility and stability in aqueous solutions while retaining the biological activity of the glucagon peptide.
- introduction of hydrophilic groups at one or more positions selected from positions 16, 17, 20, 21, 24 and 29 of the peptide of SEQ ID NO: 711, or a glucagon agonist analog thereof, are anticipated to improve the solubility and stability.
- the Class 1 glucagon related analog peptide of SEQ ID NO: 710, SEQ ID NO: 711, SEQ ID NO: 713, or SEQ ID NO: 732 is modified to comprise one or more hydrophilic groups covalently linked to the side chains of amino acids present at positions 21 and 24 of the glucagon peptide.
- the Class 1 glucagon related analog peptide of SEQ ID NO: 711 is modified to contain one or more amino acid substitution at positions 16, 17, 20, 21, 24 and/or 29, wherein the native amino acid is substituted with an amino acid having a side chain suitable for crosslinking with hydrophilic moieties, including for example, PEG.
- the native peptide can be substituted with a naturally occurring amino acid or a synthetic (non-naturally occurring) amino acid.
- Synthetic or non-naturally occurring amino acids refer to amino acids that do not naturally occur in vivo but which, nevertheless, can be incorporated into the peptide structures described herein.
- a Class 1 glucagon related analog peptide of SEQ ID NO: 710, SEQ ID NO: 711 or SEQ ID NO: 713 wherein the native glucagon peptide sequence has been modified to contain a naturally occurring or synthetic amino acid in at least one of positions 16, 17, 21, 24, 29, within a C-terminal extension or at the C-terminal amino acid of the native sequence, wherein the amino acid substitute further comprises a hydrophilic moiety.
- the substitution is at position 21 or 24, and in a further embodiment the hydrophilic moiety is a PEG chain.
- the Class 1 glucagon related analog peptide of SEQ ID NO: 711 is substituted with at least one cysteine residue, wherein the side chain of the cysteine residue is further modified with a thiol reactive reagent, including for example, maleimido, vinyl sulfone, 2-pyridylthio, haloalkyl, and haloacyl.
- thiol reactive reagents may contain carboxy, keto, hydroxyl, and ether groups as well as other hydrophilic moieties such as polyethylene glycol units.
- the native glucagon peptide is substituted with lysine, and the side chain of the substituting lysine residue is further modified using amine reactive reagents such as active esters (succinimido, anhydride, etc) of carboxylic acids or aldehydes of hydrophilic moieties such as polyethylene glycol.
- amine reactive reagents such as active esters (succinimido, anhydride, etc) of carboxylic acids or aldehydes of hydrophilic moieties such as polyethylene glycol.
- the Class 1 glucagon related analog peptide is selected form the group consisting of SEQ ID NO: 714, SEQ ID NO: 715, SEQ ID NO: 716, SEQ ID NO: 717, SEQ ID NO: 718 and SEQ ID NO: 719.
- the pegylated Class 1 glucagon related analog peptide comprises two or more polyethylene chains covalently bound to the glucagon peptide wherein the total molecular weight of the glucagon chains is about 1,000 to about 5,000 Daltons.
- the pegylated Class 1 glucagon related analog peptide comprises a peptide of SEQ ID NO: 706, wherein a PEG chain is covalently linked to the amino acid residue at position 21 and at position 24, and wherein the combined molecular weight of the two PEG chains is about 1,000 to about 5,000 Daltons.
- the pegylated Class 1 glucagon related analog peptide comprises a peptide of SEQ ID NO: 706, wherein a PEG chain is covalently linked to the amino acid residue at position 21 and at position 24, and wherein the combined molecular weight of the two PEG chains is about 5,000 to about 20,000 Daltons.
- the polyethylene glycol chain may be in the form of a straight chain or it may be branched. In accordance with one embodiment the polyethylene glycol chain has an average molecular weight selected from the range of about 500 to about 40,000 Daltons. In one embodiment the polyethylene glycol chain has a molecular weight selected from the range of about 500 to about 5,000 Daltons. In another embodiment the polyethylene glycol chain has a molecular weight of about 20,000 to about 40,000 Daltons.
- the glucagon agonist may be a peptide comprising the amino acid sequence of any of the amino acid sequences of SEQ ID NOs: 701-758, optionally with up to 1, 2, 3, 4, or 5 further modifications that retain glucagon agonist activity.
- the glucagon related analog peptide is a Class 2 glucagon related analog peptide, which is described herein and in U.S. Provisional Application No. 61/090,448, the contents of which are incorporated by reference in their entirety.
- Native glucagon does not activate the GIP receptor, and normally has about 1 % of the activity of native-GLP-1 at the GLP-I receptor.
- Modifications to the native glucagon sequence described herein produce Class 2 glucagon related analog peptides that can exhibit potent glucagon activity equivalent to or better than the activity of native glucagon (SEQ ID NO: 801), potent GIP activity equivalent to or better than the activity of native GIP (SEQ ID NO: 804), and/or potent GLP-I activity equivalent to or better than the activity of native GLP-I.
- Class 2 glucagon related analog peptide may be one of a glucagon/GIP co-agonist, glucagon/GIP/GLP- 1 tri-agonist, GIP/GLP-1 co-agonist, or a GIP agonist glucagon peptide, as further described herein.
- the Class 2 glucagon related analog peptides described herein exhibit an EC50 for GIP receptor activation activity of about 100 nM or less, or about 75, 50, 25, 10, 8, 6, 5, 4, 3, 2 or 1 nM or less. In some embodiments, the Class 2 glucagon related analog peptides exhibit an EC50 for glucagon receptor activation of about 100 nM or less, or about 75, 50, 25, 10, 8, 6, 5, 4, 3, 2 or 1 nM or less. In some embodiments, the Class 2 glucagon related analog peptides exhibit an EC50 for GLP-I receptor activation of about 100 nM or less, or about 75, 50, 25, 10, 8, 6, 5, 4, 3, 2 or 1 nM or less.
- Receptor activation can be measured by in vitro assays measuring cAMP induction in HEK293 cells over-expressing the receptor, e.g. assaying HEK293 cells co-transfected with DNA encoding the receptor and a luciferase gene linked to cAMP responsive element as described in Example 2.
- Class 2 glucagon related analog peptides exhibit at least about
- Class 2 glucagon related analog peptides exhibit at least about 1%, 5%, 10%, 20%, 30%, 40%, 50%, 60%, 75%, 100%, 125%, 150%, 175%, 200%, 250%, 300%, 350%, 400%, 450%, or 500% or higher activity at the glucagon receptor relative to native glucagon (glucagon potency).
- Class 2 glucagon related analog peptides exhibit at least about 0.1%, 0.2%, 0.3%, 0.4%, 0.5%, 0.6%, 0.7%, 0.8%, 0.9%, 1%, 5%, 10%, 20%, 30%, 40%, 50%, 60%, 75%, 100%, 125%, 150%, 175% or 200% or higher activity at the GLP-I receptor relative to native GLP- 1 (GLP-I potency).
- a Class 2 glucagon related analog peptide's activity at a receptor relative to a native ligand of the receptor is calculated as the inverse ratio of EC50s for the Class 2 glucagon related analog peptide vs. the native ligand.
- Class 2 glucagon related analog peptides exhibit activity at both the glucagon receptor and the GIP receptor ("glucagon/GIP co-agonists"). These Class 2 glucagon related analog peptides have lost native glucagon's selectivity for glucagon receptor compared to GIP receptor.
- the EC50 of the Class 2 glucagon related analog peptide at the GIP receptor is less than about 50-fold, 40-fold, 30-fold or 20-fold different (higher or lower) from its EC50 at the glucagon receptor.
- the GIP potency of the Class 2 glucagon related analog peptide is less than about 500-, 450-, 400-, 350-, 300-, 250-, 200-, 150-, 100-, 75-, 50-, 25-, 20-, 15-, 10-, or 5-fold different (higher or lower) from its glucagon potency.
- the ratio of the EC50 of the Class 2 glucagon related analog peptide at the GIP receptor divided by the EC50 of the Class 2 glucagon related analog peptide at the glucagon receptor is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5.
- the ratio of the GIP potency of the Class 2 glucagon related analog peptide compared to the glucagon potency of the Class 2 glucagon related analog peptide is less than about 500, 450, 400, 350, 300, 250, 200, 150, 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5.
- GLP-I activity have been significantly reduced or destroyed, e.g., by an amino acid modification at position 7.
- Class 2 glucagon related analog peptides exhibit activity at the glucagon, GIP and GLP-I receptors ("glucagon/GIP/GLP- 1 tri-agonists"). These Class 2 glucagon related analog peptides have lost native glucagon's selectivity for the glucagon receptor compared to both the GLP-I and GIP receptors.
- the EC50 of the Class 2 glucagon related analog peptide at the GIP receptor is less than about 50-fold, 40- fold, 30-fold or 20-fold different (higher or lower) from its respective EC50s at the glucagon and GLP-I receptors.
- the GIP potency of the Class 2 glucagon related analog peptide is less than about 500-, 450-, 400-, 350-, 300-, 250-, 200-, 150-, 100-, 75-, 50- , 25-, 20-, 15-, 10-, or 5-fold different (higher or lower) from its glucagon and GLP-I potencies.
- the ratio of the EC50 of the tri-agonist at the GIP receptor divided by the EC50 of the tri-agonist at the GLP-I receptor is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5.
- the ratio of the GIP potency of the tri- agonist compared to the GLP-I potency of the tri-agonist is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5. In related embodiments, the ratio of the EC50 of the tri-agonist at the GIP receptor divided by the EC50 of the tri-agonist at the glucagon receptor is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5. In some embodiments, the ratio of the GIP potency of the tri-agonist compared to the glucagon potency of the tri-agonist is less than about 500, 450, 400, 350, 300, 250, 200, 150, 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5.
- Class 2 glucagon related analog peptides exhibit activity at the GLP-I and GIP receptors, but in which the glucagon activity has been significantly reduced or destroyed (“GIP/GLP-1 co-agonists"), e.g., by an amino acid modification at position 3. For example, substitution at this position with an acidic, basic, or a hydrophobic amino acid (glutamic acid, ornithine, norleucine) reduces glucagon activity.
- the EC50 of the glucagon peptide at the GIP receptor is less than about 50-fold, 40-fold, 30-fold or 20-fold different (higher or lower) from its EC50 at the GLP-I receptor.
- the GIP potency of the Class 2 glucagon related analog peptide is less than about 25-, 20-, 15-, 10-, or 5-fold different (higher or lower) from its GLP-I potency.
- these Class 2 glucagon related analog peptides have about 10% or less of the activity of native glucagon at the glucagon receptor, e.g. about 1-10%, or about 0.1-10%, or greater than about 0.1% but less than about 10%.
- the ratio of the EC50 of the Class 2 glucagon related analog peptide at the GIP receptor divided by the EC50 of the Class 2 glucagon related analog peptide at the GLP-I receptor is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5. In some embodiments, the ratio of the GIP potency of the Class 2 glucagon related analog peptide compared to the GLP-I potency of the Class 2 glucagon related analog peptide is less than about 100, 75, 60, 50, 40, 30, 20, 15, 10, or 5.
- Class 2 glucagon related analog peptides exhibit activity at the GIP receptor, in which the glucagon and GLP-I activity have been significantly reduced or destroyed ("GIP agonist glucagon peptides"), e.g., by amino acid modifications at positions 3 and 7.
- GIP agonist glucagon peptides e.g., by amino acid modifications at positions 3 and 7.
- these Class 2 glucagon related analog peptides have about 10% or less of the activity of native glucagon at the glucagon receptor, e.g. about 1-10%, or about 0.1-10%, or greater than about 0.1%, 0.5%, or 1% but less than about 1%, 5%, or 10%.
- these Class 2 glucagon related analog peptides also have about 10% or less of the activity of native GLP-I at the GLP-I receptor, e.g. about 1-10%, or about 0.1- 10%, or greater than about 0.1%, 0.5%, or 1% but less than about 1%, 5%, or 10%.
- the EC50 of the Class 2 glucagon related analog peptide for GIP receptor activation is about 4, 2, 1 nM or less, or the analog has at least about 1%, 2%, 3%, 4% or 5% of the activity of native GIP at the GIP receptor.
- the EC50 of the unpegylated Class 2 glucagon related analog peptide for GLP- 1 receptor activation is about 4, 2, 1 nM or less or has at least about 1%, 2%, 3%, 4% or 5% of the activity of native GLP-I at the GLP-I receptor.
- the EC50 of the unpegylated Class 2 glucagon related analog peptide for glucagon receptor activation is about 4, 2, 1 nM or less, or at least about 5%, 10%, 15% or 20% of the activity of native glucagon at the glucagon receptor. In some embodiments, the unpegylated Class 2 glucagon related analog peptide has less than about 1% of the activity of native glucagon at the glucagon receptor. In other embodiments, the unpegylated Class 2 glucagon related analog peptide has less than about 10%, 5% or 1% of the activity of native GLP-I at the GLP-I receptor.
- the relative EC50s at one or more receptors may be higher.
- the EC50 of a pegylated analog for GIP receptor activation is about 10 nM or less, or the Class 2 glucagon related analog peptide has at least about 0.1%, 0.2%, 0.3%, 0.4% or 0.5% of the activity of native GIP at the GIP receptor.
- the EC50 of a pegylated Class 2 glucagon related analog peptide for GLP-I receptor activation is about 10 nM or less or has at least about 0.1%, 0.2%, 0.3%, 0.4% or 0.5% of the activity of native GLP-I at the GLP-I receptor. In yet other related embodiments, the EC50 of a pegylated Class 2 glucagon related analog peptide for glucagon receptor activation is about 10 nM or less, or at least about 0.5%, 1 %, 1.5% or 2% of the activity of native glucagon at the glucagon receptor.
- the Class 2 glucagon related analog peptide has less than about 1 % of the activity of native glucagon at the glucagon receptor. In other embodiments, the Class 2 glucagon related analog peptide has less than about 10%, 5% or 1% of the activity of native GLP-I at the GLP-I receptor.
- glucagon related analog peptide permit the manipulation of glucagon (SEQ ID NO: 801) to create glucagon peptides that exhibit increased GIP activity, glucagon activity, and/or GLP- 1 activity.
- Other modifications disclosed herein in reference to a Class 2 glucagon related analog peptide prolong the half-life, increase solubility, or increase stability of the resulting peptide.
- Yet other modifications disclosed herein in reference to a Class 2 glucagon related analog peptide have no effect on activity, or can be made without destroying the desired activity or activities. Any of the combinations in reference to a Class 2 glucagon related analog peptide that serve the same purpose (e.g.
- increasing GIP activity can be applied individually or in combination. Any of the single or sets of combinations in reference to a Class 2 glucagon related analog peptide that confer enhanced properties can be applied individually or in combination, e.g. increased GIP and/or GLP-I activity can be combined with increased half-life.
- 1, 2, 3, 4, 5, 6 or more of the amino acid modifications may be non- conservative substitutions, additions or deletions. In some embodiments, 1, 2, 3, 4, 5, 6 or more of the amino acid modifications may be conservative substitutions.
- Enhanced activity at the GIP receptor is provided by an amino acid modification at position 1.
- His at position 1 is substituted with a large, aromatic amino acid, optionally Tyr, Phe, Trp, amino-Phe, nitro-Phe, chloro-Phe, sulfo-Phe, 4-pyridyl-Ala, methyl- Tyr, or 3-amino Tyr.
- the combination of Tyr at position 1 with stabilization of the alpha helix within the region corresponding to amino acids 12-29 provided a Class 2 glucagon related analog peptide that activates the GIP receptor as well as the GLP-I receptor and the glucagon receptor.
- the alpha helix structure can be stabilized by, e.g., formation of a covalent or non-covalent intramolecular bridge, or substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid (e.g., an ⁇ , ⁇ - disubstituted amino acid).
- Enhanced activity at the GIP receptor is also provided by amino acid modifications at positions 27 and/or 28, and optionally at position 29.
- the Met at position 27 is substituted with a large aliphatic amino acid, optionally Leu
- the Asn at position 28 is substituted with a small aliphatic amino acid, optionally Ala
- the Thr at position 29 is substituted with a small aliphatic amino acid, optionally GIy.
- Substitution with LAG at positions 27-29 provides increased GIP activity relative to the native MNT sequence at those positions.
- Enhanced activity at the GIP receptor is also provided by an amino acid modification at position 12.
- position 12 is substituted with a large, aliphatic, nonpolar amino acid, optionally He.
- Enhanced activity at the GIP receptor is also provided by an amino acid modification at positions 17 and/or 18.
- position 17 is substituted with a polar residue, optionally GIn
- position 18 is substituted with a small aliphatic amino acid, optionally Ala.
- a substitution with QA at positions 17 and 18 provides increased GIP activity relative to the native RR sequence at those positions.
- an intramolecular bridge can be formed by a covalent bond between the side chains of two amino acids at positions i and i+4 or between positions j and j+3, or between positions k and k+7.
- the bridge is between positions 12 and 16, 16 and 20, 20 and 24, 24 and 28, or 17 and 20.
- non-covalent interactions such as salt bridges can be formed between positively and negatively charged amino acids at these positions.
- enhanced glucagon potency is provided by an amino acid modification at position 16 of native glucagon (SEQ ID NO: 801).
- such enhanced potency can be provided by substituting the naturally occurring serine at position 16 with glutamic acid or with another negatively charged amino acid having a side chain with a length of 4 atoms, or alternatively with any one of glutamine, homoglutamic acid, or homocysteic acid, or a charged amino acid having a side chain containing at least one heteroatom, (e.g. N, O, S, P) and with a side chain length of about 4 (or 3-5) atoms.
- the glucagon peptide retains its original selectivity for the glucagon receptor relative to the GLP-I receptors.
- Glucagon receptor activity can be reduced by an amino acid modification at position
- an intramolecular bridge can be formed by a covalent bond between the side chains of two amino acids at positions i and i+4 or between positions j and j+3, or between positions k and k+7.
- non-covalent interactions such as salt bridges can be formed between positively and negatively charged amino acids at these positions.
- Enhanced activity at the GLP-I receptor is provided by replacing the carboxylic acid of the C-terminal amino acid with a charge-neutral group, such as an amide or ester.
- Enhanced activity at the GLP- 1 receptor is also provided by stabilizing the alpha-helix structure in the C-terminal portion of glucagon (around amino acids 12-29), e.g., through formation of an intramolecular bridge between the side chains of two amino acids, or substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid (e.g., an ⁇ , ⁇ -disubstituted amino acid), as further described herein.
- an alpha helix- stabilizing amino acid e.g., an ⁇ , ⁇ -disubstituted amino acid
- the side chains of the amino acid pairs 12 and 16, 13 and 17, 16 and 20 , 17 and 21, 20 and 24 or 24 and 28 are linked to one another and thus stabilize the glucagon alpha helix.
- the bridge or linker is about 8 (or about 7-9) atoms in length, particularly when the bridge is between positions i and i+4. In some embodiments, the bridge or linker is about 6 (or about 5-7) atoms in length, particularly when the bridge is between positions j and j+3.
- intramolecular bridges are formed by (a) substituting the naturally occurring serine at position 16 with glutamic acid or with another negatively charged amino acid having a side chain with a length of 4 atoms, or alternatively with any one of glutamine, homoglutamic acid, or homocysteic acid, or a charged amino acid having a side chain containing at least one heteroatom, (e.g.
- the side chains of such amino acids at positions 16 and 20 can form a salt bridge or can be covalently linked.
- the two amino acids are bound to one another to form a lactam ring.
- stabilization of the alpha helix structure in the C -terminal portion of the glucagon peptide is achieved through the formation of an intramolecular bridge other than a lactam bridge.
- suitable covalent bonding methods include any one or more of olefin metathesis, lanthionine-based cyclization, disulfide bridge or modified sulfur-containing bridge formation, the use of ⁇ , ⁇ -diaminoalkane tethers, the formation of metal-atom bridges, and other means of peptide cyclization are used to stabilize the alpha helix.
- one or more ⁇ , ⁇ -disubstituted amino acids are inserted or substituted into this C-terminal portion (amino acids 12-29) at positions that retain the desired activity.
- amino acids 12-29 amino acids 12-29
- one, two, three or all of positions 16, 20, 21 or 24 are substituted with an ⁇ , ⁇ -disubstituted amino acid, e.g., AIB.
- Increased activity at the GLP-I receptor is provided by an amino acid modification at position 20 as described herein.
- Increased activity at the GLP-I receptor is provided by adding GPSSGAPPPS (SEQ ID NO: 895) or XGPSSGAPPPS (SEQ ID NO: 896) to the C-terminus.
- GLP-I activity in such analogs can be further increased by modifying the amino acid at position 18, 28 or 29, or at position 18 and 29, as described herein.
- GLP- 1 potency is provided by modifying the amino acid at position 10 to be a large, aromatic amino acid residue, optionally Trp.
- Reduced activity at the GLP-I receptor is provided, e.g., by an amino acid modification at position 7 as described herein.
- Potency at the GLP-I receptor can be further enhanced by an alanine substitution for the native arginine at position 18.
- the invention provides glucagon peptides that comprise modifications at position 16, at position 20, and at the C-terminal carboxylic acid group, optionally with a covalent bond between the amino acids at positions 16 and 20; glucagon peptides that comprise modifications at position 16 and at the C-terminal carboxylic acid group; glucagon peptides that comprise modifications at positions 16 and 20, optionally with a covalent bond between the amino acids at positions 16 and 20; and glucagon peptides that comprise modifications at position 20 and at the C-terminal carboxylic acid group.
- Modifications at position 1 and/or 2 can increase the peptide's resistance to dipeptidyl peptidase IV (DPP IV) cleavage.
- DPP IV dipeptidyl peptidase IV
- position 1 and/or position 2 may be substituted with a DPP-IV resistant amino acid as described herein.
- the amino acid at position 2 is substituted with N-methyl alanine. It was observed that modifications at position 2 (e.g.
- AIB at position 2) and in some cases modifications at position 1 may reduce glucagon activity, sometimes significantly; surprisingly, this reduction in glucagon activity can be restored by stabilizing the alpha-helix structure in the C-terminal portion of glucagon (around amino acids 12-29), e.g., through formation of a covalent bond between the side chains of two amino acids, as described herein.
- the covalent bond is between amino acids at positions "i" and "i+4", or positions "j" and "j+3", e.g., between positions 12 and 16, 16 and 20, 20 and 24, 24 and 28, or 17 and 20.
- this covalent bond is a lactam bridge between a glutamic acid at position 16 and a lysine at position 20. In some embodiments, this covalent bond is an intramolecular bridge other than a lactam bridge, as described herein.
- any of the Class 2 glucagon related analog peptides can be further modified to improve stability by modifying the amino acid at position 15 and/or 16 of SEQ ID NO: 801 to reduce degradation of the peptide over time, especially in acidic or alkaline buffers. Such modifications reduce cleavage of the Aspl5-Serl6 peptide bond.
- the amino acid modification at position 15 is a deletion or substitution of Asp with glutamic acid, homoglutamic acid, cysteic acid or homocysteic acid.
- the amino acid modification at position 16 is a deletion or substitution of Ser with Thr or AIB.
- Ser at position 16 is substituted with glutamic acid or with another negatively charged amino acid having a side chain with a length of 4 atoms, or alternatively with any one of glutamine, homoglutamic acid, or homocysteic acid.
- the methionine residue present at position 27 of the native peptide is modified, e.g. by deletion or substitution. Such modifications may prevent oxidative degradation of the peptide.
- the Met at position 27 is substituted with leucine, isoleucine or norleucine.
- Met at position 27 is substituted with leucine or norleucine.
- the GIn at position 20 and/or 24 is modified, e.g. by deletion or substitution. Such modifications can reduce degradation that occurs through deamidation of GIn.
- the GIn at position 20 and/or 24 is substituted with Ala or AIB.
- the GIn at position 20 and/or 24 is substituted with Lys, Arg, Orn, or Citrulline.
- the Asp at position 21 is modified, e.g. by deletion or substitution. Such modifications can reduce degradation that occurs through dehydration of Asp to form a cyclic succinimide intermediate followed by isomerization to iso-aspartate.
- position 21 is substituted with GIu, homoglutamic acid or homocysteic acid. In some specific embodiments, position 21 is substituted with GIu.
- Stabilization of the alpha-helix structure in the C-terminal portion of the Class 2 glucagon related analog peptide provides enhanced GLP-I and/or GIP activity and restores glucagon activity which has been reduced by amino acid modifications at positions 1 and/or 2.
- the alpha helix structure can be stabilized by, e.g., formation of a covalent or non-covalent intramolecular bridge, or substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid (e.g., an ⁇ , ⁇ -disubstituted amino acid).
- Stabilization of the alpha-helix structure of a GIP agonist may be accomplished as described herein.
- the glucagon peptides disclosed herein are modified to comprise an acyl group or alkyl group as described herein.
- Acylation or alkylation can increase the half-life of the glucagon peptides in circulation.
- Acylation or alkylation can advantageously delay the onset of action and/or extend the duration of action at the glucagon and/or GLP-I receptors and/or improve resistance to proteases such as DPP- IV and/or improve solubility.
- Activity at the glucagon and/or GLP-I and/or GIP receptors of the glucagon peptide may be maintained after acylation.
- the potency of the acylated glucagon peptides is comparable to the unacylated versions of the glucagon peptides.
- Class 2 glucagon related analog peptides may be acylated or alkylated at the same amino acid position where a hydrophilic moiety is linked, or at a different amino acid position, as described herein.
- the invention provides a glucagon peptide modified to comprise an acyl group or alkyl group covalently linked to the amino acid at position 10 of the glucagon peptide.
- the glucagon peptide may further comprise a spacer between the amino acid at position 10 of the glucagon peptide and the acyl group or alkyl group.
- the acyl group is a fatty acid or bile acid, or salt thereof, e.g.
- the spacer is any moiety with suitable reactive groups for attaching acyl or alkyl groups.
- the spacer comprises an amino acid, a dipeptide, or a tripeptide, or a hydrophilic bifunctional spacer.
- the spacer is selected from the group consisting of: Trp, GIu, Asp, Cys and a spacer comprising NH 2 (CH 2 CH 2 ⁇ )n(CH 2 )mCOOH, wherein m is any integer from 1 to 6 and n is any integer from 2 to 12.
- acylated or alkylated glucagon peptides may also further comprise a hydrophilic moiety, optionally a polyethylene glycol. Any of the foregoing glucagon peptides may comprise two acyl groups or two alkyl groups, or a combination thereof.
- the GIP agonist can be linked, optionally via covalent bonding and optionally via a linker, to a conjugate moiety as described herein.
- the second peptide is XGPSSGAPPPS (SEQ ID NO: 896), wherein X is selected from one of the 20 common amino acids, e.g., glutamic acid, aspartic acid or glycine.
- X represents an amino acid, for example Cys, that further comprises a hydrophilic moiety covalently linked to the side chain of that amino acid.
- Such C-terminal extensions improve solubility and also can improve GIP or GLP-I activity.
- the glucagon peptide further comprises a carboxy terminal extension, the carboxy terminal amino acid of the extension ends in an amide group or an ester group rather than a carboxylic acid.
- the threonine at position 29 of the native glucagon peptide is replaced with a glycine.
- a glucagon peptide having a glycine substitution for threonine at position 29 and comprising the C-terminal extension of GPSSGAPPPS (SEQ ID NO: 895) is four times as potent at the GLP-I receptor as native glucagon modified to comprise the same C-terminal extension.
- This T29G substitution can be used in conjunction with other modifications disclosed herein to enhance the affinity of the glucagon peptides for the GLP-I receptor.
- the T29G substitution can be combined with the S16E and N20K amino acid substitutions, optionally with a lactam bridge between amino acids 16 and 20, and optionally with addition of a PEG chain as described herein.
- an amino acid is added to the C-terminus, and the additional amino acid is selected from the group consisting of glutamic acid, aspartic acid and glycine.
- the solubility of any of the glucagon peptides can be improved by amino acid substitutions and/or additions that introduce a charged amino acid into the C-terminal portion of the peptide, preferably at a position C-terminal to position 27 of SEQ ID NO: 801.
- one, two or three charged amino acids may be introduced within the C-terminal portion, preferably C-terminal to position 27.
- the native amino acid(s) at positions 28 and/or 29 are substituted with one or two charged amino acids, and/or in a further embodiment one to three charged amino acids are also added to the C-terminus of the peptide.
- one, two or all of the charged amino acids are negatively charged.
- the negatively charged (acidic amino acid) is aspartic acid or glutamic acid.
- glucagon peptide may be made to the glucagon peptide that still allow it to retain GIP activity (and optionally GLP-I activity and/or glucagon activity).
- any of the modifications described above in reference to a Class 2 peptide which increase or decrease GIP activity, which increase or decrease glucagon receptor activity, and which increase GLP-I receptor activity can be applied individually or in combination. Any of the modifications described above in reference to a Class 2 glucagon related analog peptide can also be combined with other modifications that confer other desirable properties, such as increased solubility and/or stability and/or duration of action, as described herein with regard to Class 2 glucagon related analog peptides. Alternatively, any of the modifications described above in reference to Class 2 glucaton related peptides can be combined with other modifications described herein in reference to Class 2 glucagon related analog peptides that do not substantially affect solubility or stability or activity. Exemplary modifications include but are not limited to:
- (A) Improving solubility for example, by introducing one, two, three or more charged amino acid(s) to the C-terminal portion of native glucagon, preferably at a position C-terminal to position 27.
- a charged amino acid can be introduced by substituting a native amino acid with a charged amino acid, e.g. at positions 28 or 29, or alternatively by adding a charged amino acid, e.g. after position 27, 28 or 29.
- one, two, three or all of the charged amino acids are negatively charged.
- one, two, three or all of the charged amino acids are positively charged.
- Such modifications increase solubility, e.g. provide at least 2-fold, 5-fold, 10-fold, 15-fold, 25-fold, 30-fold or greater solubility relative to native glucagon at a given pH between about 5.5 and 8, e.g., pH 7, when measured after 24 hours at 25°C.
- (C) Increasing solubility and/or duration of action or half-life in circulation and/or delaying the onset of action by acylation or alkylation of the glucagon peptide, as described herein;
- (D) Increasing duration of action or half- life in circulation through introducing resistance to dipeptidyl peptidase IV (DPP IV) cleavage by modification of the amino acid at position 1 or 2 as described herein.
- DPP IV dipeptidyl peptidase IV
- (E) Increasing stability by modification of the Asp at position 15, for example, by deletion or substitution with glutamic acid, homoglutamic acid, cysteic acid or homocysteic acid. Such modifications can reduce degradation or cleavage at a pH within the range of 5.5 to 8, for example, retaining at least 75%, 80%, 90%, 95%, 96%, 97%, 98% or 99% of the original peptide after 24 hours at 25°C. Such modifications reduce cleavage of the peptide bond between Aspl5-Serl6.
- G Increasing stability by modification of the methionine at position 27, for example, by substitution with leucine or norleucine. Such modifications can reduce oxidative degradation. Stability can also be increased by modification of the GIn at position 20 or 24, e.g. by substitution with Ala or AIB. Such modifications can reduce degradation that occurs through deamidation of GIn. Stability can be increased by modification of Asp at position 21, e.g. by substitution with GIu. Such modifications can reduce degradation that occurs through dehydration of Asp to form a cyclic succinimide intermediate followed by isomerization to iso-aspartate.
- Non-conservative or conservative substitutions, additions or deletions that do not substantially affect activity for example, conservative substitutions at one or more of positions 2, 5, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 24, 27, 28 or 29; substitution of one or more of these positions with Ala; deletion of amino acids at one or more of positions 27, 28 or 29; or deletion of amino acid 29 optionally combined with a C-terminal amide or ester in place of the C-terminal carboxylic acid group; substitution of Lys at position 12 with Arg; substitution of Tyr at position 10 with VaI or Phe;
- GPSSGAPPPS SEQ ID NO: 895
- Some positions of the native glucagon peptide can be modified while retaining at least some of the activities of the parent peptide. Accordingly, applicants anticipate that one or more of the amino acids located at positions at positions 2, 5, 10, 11, 12, 13, 14, 17, 18, 19, 20, 21, 24, 27, 28 or 29 can be substituted with an amino acid different from that present in the native glucagon peptide, and still retain activity at the glucagon receptor.
- position 18 is substituted with an amino acid selected from the group consisting of Ala, Ser, or Thr.
- amino acid at position 20 is substituted with Lys, Arg, Orn, Citrulline or AIB.
- position 21 is substituted with GIu, homoglutamic acid or homocysteic acid.
- the glucagon peptide comprises 1 to 10 amino acid modifications selected from positions 16, 17, 18, 20, 21, 23, 24, 27, 28 and 29.
- the modifications are one or more amino acid substitutions selected from the group consisting of Glnl7, Alal8, Glu21, Ile23, Ala24, Val27 and Gly29.
- 1 to 2 amino acids selected from positions 17-26 differ from the parent peptide. In other embodiments, 1 to 2 amino acids selected from positions 17-22 differ from the parent peptide. In yet other embodiments, the modifications are Glnl7, Alal8, Glu21, Ile23 and Ala24.
- one or more amino acids is added to the carboxy terminus of the glucagon peptide.
- the amino acid is typically selected from one of the 20 common amino acids, and in some embodiments the amino acid has an amide group in place of the carboxylic acid of the native amino acid.
- the added amino acid is selected from the group consisting of glutamic acid and aspartic acid and glycine.
- the Class 2 glucagon related analog peptides disclosed herein are modified by truncation of the C-terminus by one or two amino acid residues yet retain similar activity and potency at the glucagon, GLP-I and/or GIP receptors.
- the amino acid at position 29 and/or 28 can be deleted.
- the Class 2 glucagon related analog peptide is an analog of glucagon (SEQ ID NO: 801) having GIP agonist activity, with the following modifications:
- the amino acid modification at position 1 is a substitution of His at position 1 with a large, aromatic amino acid, optionally Tyr, Phe, Trp, amino-Phe, nitro-Phe, chloro-Phe, sulfo-Phe, 4-pyridyl-Ala, methyl- Tyr, or 3 -amino Tyr,
- the lactam bridge is between the amino acids at positions 16 and 20, wherein one of the amino acids at positions 16 and 20 is substituted with GIu, and the other of the amino acids at positions 16 and 20 is substituted with Lys, or (ii) the ⁇ , ⁇ -disubstituted amino acid is AIB,
- Thr at position 29 is substituted with a small aliphatic amino acid, optionally GIy.
- the analog may comprise further modifications, including without limitation: (a) amino acid modification at position 12, optionally substitution with He,
- the Class 2 glucagon related analog peptide is covalently linked to a hydrophilic moiety at any of amino acid positions 16, 17, 20, 21, 24, or 29, after position 29 at an added amino acid (e.g., position 30) within a C-terminal extension, or at the C-terminal amino acid.
- this hydrophilic moiety is covalently linked to a Lys, Cys, Orn, homocysteine, or acetyl-phenylalanine residue at any of these positions.
- Exemplary hydrophilic moieties include polyethylene glycol (PEG), for example, of a molecular weight of about 1,000 Daltons to about 40,000 Daltons, or about 20,000 Daltons to about 40,000 Daltons.
- the GIP agonist may be a peptide comprising the amino acid sequence of any of the amino acid sequences, e.g., SEQ ID NOs: 805-894, optionally with up to 1, 2, 3, 4, or 5 further modifications that retain GIP agonist activity.
- the glucagon related analog peptide is a Class 3 glucagon related analog peptide, which is described herein and in International Patent Application Publication No. WO 2008/101017, published on August 21, 2008, and U.S. Provisional Application No. 61/090,412, the contents of which are incorporated by reference in their entirety.
- the Class 3 glucagon related analog peptide can be a peptide that exhibits increased activity at the glucagon receptor, and in further embodiments exhibits enhanced biophysical stability and/or aqueous solubility.
- the Class 3 glucagon related analog peptide has lost native glucagon's selectivity for the glucagon receptor verses the GLP-I receptor, and thus represents co-agonists of those two receptors. Selected amino acid modifications within the Class 3 glucagon related analog peptide can control the relative activity of the peptide at the GLP-I receptor verses the glucagon receptor.
- the Class 3 glucagon related analog peptide can be a glucagon/GLP- 1 co-agonist that has higher activity at the glucagon receptor versus the GLP- 1 receptor, a glucagon/GLP- 1 co-agonist that has approximately equivalent activity at both receptors, or a glucagon/GLP- 1 co-agonist that has higher activity at the GLP- 1 receptor versus the glucagon receptor.
- the latter category of co- agonist can be engineered to exhibit little or no activity at the glucagon receptor, and yet retain ability to activate the GLP-I receptor with the same or better potency than native GLP- 1. Any of these co-agonists may also include modifications that confer enhanced biophysical stability and/or aqueous solubility.
- Modifications of the Class 3 glucagon related analog peptide can be made to produce a glucagon peptide having anywhere from at least about 10% (including at least about 20%, 30%, 40%, 50%, 60%, 75%, 100%, 125%, 150%, 175%) to about 200% or higher activity at the GLP-I receptor relative to native GLP-I and anywhere from at least about 10% (including about 20%, 30%, 40%, 50%, 60%, 75%, 100%, 125%, 150%, 175%, 200%, 250%, 300%, 350%, 400%, 450%) to about 500% or higher activity at the glucagon receptor relative to native glucagon.
- amino acid sequence of native glucagon is SEQ ID NO: 1
- amino acid sequence of GLP-l(7-36)amide is SEQ ID NO: 52
- amino acid sequence of GLP-l(7-37)acid is SEQ ID NO: 50.
- a Class 3 glucagon related analog peptide may exhibit at least 10% of the activity of native glucagon at the glucagon receptor and at least 50% of the activity of native GLP-I at the GLP-I receptor, or at least 40% of the activity of native glucagon at the glucagon receptor and at least 40% of the activity of native GLP-I at the GLP-I receptor, or at least 60% of the activity of native glucagon at the glucagon receptor and at least 60% of the activity of native GLP-I at the GLP-I receptor.
- Selectivity of a Class 3 glucagon related analog peptide for the glucagon receptor versus the GLP-I receptor can be described as the relative ratio of glucagon/GLP- 1 activity (the peptide's activity at the glucagon receptor relative to native glucagon, divided by the peptide's activity at the GLP-I receptor relative to native GLP-I).
- a Class 3 glucagon related analog peptide that exhibits 60% of the activity of native glucagon at the glucagon receptor and 60% of the activity of native GLP- 1 at the GLP- 1 receptor has a 1 : 1 ratio of glucagon/GLP- 1 activity.
- Exemplary ratios of glucagon/GLP- 1 activity include about 1:1, 1.5: 1, 2:1, 3:1, 4:1, 5:1, 6:1, 7:1, 8:1, 9: 1 or 10: 1, or about 1 :10, 1:9, 1 :8, 1:7, 1:6, 1 :5,
- a glucagon/GLP- 1 activity ratio of 10:1 indicates a 10- fold selectivity for the glucagon receptor versus the GLP-I receptor.
- a GLP- 1/glucagon activity ratio of 10: 1 indicates a 10-fold selectivity for the GLP-I receptor versus the glucagon receptor.
- the Class 3 glucagon related analog peptides have about 10% or less of the activity of native glucagon at the glucagon receptor, e.g. about 1-10%, or about 0.1-10%, or greater than about 0.1% but less than about 10%, while exhibiting at least 20% of the activity of GLP-I at the GLP-I receptor.
- exemplary Class 3 glucagon related analog peptides described herein have about 0.5%, about 1% or about 7% of the activity of native glucagon, while exhibiting at least 20% of the activity of GLP-I at the GLP-I receptor.
- the Class 3 glucagon related analog peptide can be a glucagon peptide with increased or decreased activity at the glucagon receptor, or GLP-I receptor, or both.
- the Class 3 glucagon related analog peptide can be a glucagon peptide with altered selectivity for the glucagon receptor versus the GLP-I receptor.
- high potency Class 3 glucagon related analog peptides are provided that also exhibit improved solubility and/or stability.
- An exemplary high potency Class 3 glucagon related analog peptide exhibits at least about 200% of the activity of native glucagon at the glucagon receptor, and optionally is soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8, or between 6 and 9, or between 7 and 9 (e.g. pH 7), and optionally retains at least 95% of the original peptide (e.g. 5% or less of the original peptide is degraded or cleaved) after 24 hours at 25°C.
- an exemplary Class 3 glucagon related analog peptide exhibits greater than about 40% or greater than about 60% activity at both the glucagon and the GLP- 1 receptors (at a ratio between about 1 : 3 and 3:1, or between about 1:2 and 2: 1), is optionally soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, or between 7 and 9 (e.g. pH 7), and optionally retains at least 95% of the original peptide after 24 hours at 25°C.
- Another exemplary Class 3 glucagon related analog peptide exhibits about 175% or more of the activity of native glucagon at the glucagon receptor and about 20% or less of the activity of native GLP-I at the GLP-I receptor, is optionally soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, or between 7 and 9 (e.g. pH 7), and optionally retains at least 95% of the original peptide after 24 hours at 25°C.
- Yet another exemplary Class 3 glucagon related analog peptide exhibits about 10% or less of the activity of native glucagon at the glucagon receptor and at least about 20% of the activity of native GLP-I at the GLP-I receptor, is optionally soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, or between 7 and 9 (e.g. pH 7), and optionally retains at least 95% of the original peptide after 24 hours at 25°C.
- Yet another exemplary Class 3 glucagon related analog peptide exhibits about 10% or less but above 0.1% , 0.5% or 1% of the activity of native glucagon at the glucagon receptor and at least about 50%, 60%, 70%, 80%, 90% or 100% or more of the activity of native GLP-I at the GLP-I receptor, is optionally soluble at a concentration of at least 1 mg/mL at a pH between 6 and 8 or between 6 and 9, or between 7 and 9 (e.g. pH 7), and optionally retains at least 95% of the original peptide after 24 hours at 25°C.
- Class 3 glucagon related analog peptides retain at least 22, 23, 24, 25, 26, 27 or 28 of the naturally occurring amino acids at the corresponding positions in native glucagon (e.g. have 1-7, 1-5 or 1-3 modifications relative to naturally occurring glucagon). Modifications affecting glucagon activity
- the Class 3 glucagon related analog peptide is a glucagon agonist that has been modified relative to the wild type peptide of His-Ser-Gln-Gly-Thr-Phe- Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Ser- Arg-Arg- Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu- Met-Asn-Thr (SEQ ID NO: 1) to enhance the peptide's potency at the glucagon receptor.
- the normally occurring serine at position 16 of native glucagon can be substituted with select acidic amino acids to enhance the potency of glucagon, in terms of its ability to stimulate cAMP synthesis in a validated in vitro model assay (see Example T). More particularly, this substitution enhances the potency of the analog at least 2-fold, 4-fold, 5-fold, and up to 10-fold greater at the glucagon receptor. This substitution also enhances the analog's activity at the GLP-I receptor at least 5-fold, 10- fold, or 15 -fold relative to native glucagon, but selectivity is maintained for the glucagon receptor over the GLP-I receptor.
- such enhanced potency can be provided by substituting the naturally occurring serine at position 16 with glutamic acid or with another negatively charged amino acid having a side chain with a length of 4 atoms, or alternatively with any one of glutamine, homoglutamic acid, or homocysteic acid, or a charged amino acid having a side chain containing at least one heteroatom, (e.g. N, O, S, P) and with a side chain length of about 4 (or 3-5) atoms.
- heteroatom e.g. N, O, S, P
- the serine residue at position 16 of native glucagon is substituted with an amino acid selected from the group consisting of glutamic acid, glutamine, homoglutamic acid, homocysteic acid, threonine, or glycine.
- the serine residue at position 16 of native glucagon is substituted with an amino acid selected from the group consisting of glutamic acid, glutamine, homoglutamic acid and homocysteic acid, and in one embodiment the serine residue is substituted with glutamic acid.
- the enhanced potency Class 3 glucagon related analog peptide comprises a peptide of SEQ ID NO: 2, SEQ ID NO: 3, SEQ ID NO: 4, SEQ ID NO: 5, SEQ ID NO: 6, SEQ ID NO: 7 or a glucagon agonist analog of SEQ ID NO: 5.
- a Class 3 glucagon related analog peptide having enhanced potency at the glucagon receptor relative to wild type glucagon is provided wherein the peptide comprises the sequence of SEQ ID NO: 7, SEQ ID NO: 8, SEQ ID NO: 9 or SEQ ID NO: 10, wherein the glucagon peptide retains its selectivity for the glucagon receptor relative to the GLP-I receptors.
- the Class 3 glucagon related analog peptide having enhanced specificity for the glucagon receptor comprises the peptide of SEQ ID NO: 8, SEQ ID NO: 9, SEQ ID NO: 10 or a glucagon agonist analog thereof, wherein the carboxy terminal amino acid retains its native carboxylic acid group.
- a Class 3 glucagon related analog peptide comprises the sequence of NH 2 -His-Ser-Gln-Gly-Thr-Phe- Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Glu-Arg-Arg-Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu- Met-Asn-Thr-COOH (SEQ ID NO: 10), wherein the peptide exhibits approximately fivefold enhanced potency at the glucagon receptor, relative to native glucagon as measured by the in vitro cAMP assay of Example 2.
- Reduced activity at the glucagon receptor is provided, e.g., by an amino acid modification at position 3. Substitution at this position with an acidic, basic, or a hydrophobic amino acid (glutamic acid, ornithine, norleucine) has been shown to substantially reduce or destroy glucagon receptor activity.
- an acidic, basic, or a hydrophobic amino acid glutamic acid, ornithine, norleucine
- any of the Class 3 glucagon related analog peptides including glucagon analogs, glucagon agonist analogs, glucagon co-agonists, and glucagon/GLP-1 co-agonist molecules, described herein may be modified to contain a modification at position 3, e.g., GIn substituted with GIu, to produce a peptide with high selectivity, e.g., tenfold selectivity, for the GLP-I receptor as compared to the selectivity for the glucagon receptor.
- a modification at position 3 e.g., GIn substituted with GIu
- modifications at position 2 may reduce glucagon activity.
- This reduction in glucagon activity can be restored by stabilizing the alpha-helix in the C-terminal portion of glucagon, e.g. through means described herein, for example, through a covalent bond between amino acids at positions "i" and "i+4", e.g., 12 and 16, 16 and 20, or 20 and 24.
- this covalent bond is a lactam bridge between a glutamic acid at position 16 and a lysine at position 20.
- this covalent bond is an intramolecular bridge other than a lactam bridge.
- suitable covalent bonding methods include any one or more of olefin metathesis, lanthionine-based cyclization, disulfide bridge or modified sulfur-containing bridge formation, the use of ⁇ , ⁇ -diaminoalkane tethers, the formation of metal-atom bridges, and other means of peptide cyclization. Modifications affecting GLP-I activity
- Enhanced activity at the GLP- 1 receptor is provided by replacing the carboxylic acid of the C-terminal amino acid with a charge-neutral group, such as an amide or ester.
- these Class 3 glucagon related analog peptides comprise a sequence of SEQ ID NO: 20, wherein the carboxy terminal amino acid has an amide group in place of the carboxylic acid group found on the native amino acid.
- These Class 3 glucagon related analog peptides have strong activity at both the glucagon and GLP-I receptors and thus act as co- agonists at both receptors.
- the Class 3 glucagon related analog peptide is a glucagon and GLP-I receptor co-agonist, wherein the peptide comprises the sequence of SEQ ID NO: 20, wherein the amino acid at position 28 is Asn or Lys and the amino acid at position 29 is Thr-amide.
- Increased activity at the GLP-I receptor is provided by modifications that stabilize the alpha helix in the C-terminal portion of glucagon (e.g. around residues 12-29).
- such modifications permit formation of an intramolecular bridge between the side chains of two amino acids that are separated by three intervening amino acids (i.e., an amino acid at position "i” and an amino acid at position "i+4", wherein i is any integer between 12 and 25), by two intervening amino acids, i.e., an amino acid at position "j” and an amino acid at position "j+3,” wherein j is any integer between 12 and 27, or by six intervening amino acids, i.e., an amino acid at position "k” and an amino acid at position "k+7,” wherein k is any integer between 12 and 22.
- the bridge or linker is about 8 (or about 7-9) atoms in length and forms between side chains of amino acids at positions 12 and 16, or at positions 16 and 20, or at positions 20 and 24, or at positions 24 and 28.
- the two amino acid side chains can be linked to one another through hydrogen-bonding, ionic interactions, such as the formation of salt bridges, or by covalent bonds.
- the Class 3 glucagon related analog peptide exhibits glucagon/GLP-1 receptor co-agonist activity andcomprises an amino acid sequence selected from the group consisting of SEQ ID NO: 11 , 47, 48 and 49.
- the side chains are covalently bound to one another, and in one embodiment the two amino acids are bound to one another to form a lactam ring.
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 45, wherein at least one lactam ring is formed between the side chains of an amino acid pair selected from the group consisting of amino acid pairs 12 and
- the Class 3 glucagon related analog peptide comprises a glucagon peptide analog of SEQ ID NO: 20, wherein the peptide comprises an intramolecular lactam bridge formed between amino acid positions 12 and 16 or between amino acid positions 16 and 20.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 20, wherein an intramolecular lactam bridge is formed between amino acid positions 12 and 16, between amino acid positions 16 and 20, or between amino acid positions 20 and 24 and the amino acid at position 29 is glycine, wherein the sequence of SEQ ID NO: 29 is linked to the C-terminal amino acid of SEQ ID NO: 20.
- the amino acid at position 28 is aspartic acid.
- stabilization of the alpha helix structure in the C- terminal portion of the Class 3 glucagon related analog peptide is achieved through the formation of an intramolecular bridge other than a lactam bridge.
- suitable covalent bonding methods include any one or more of olefin metathesis, lanthionine-based cyclization, disulfide bridge or modified sulfur-containing bridge formation, the use of ⁇ , ⁇ - diaminoalkane tethers, the formation of metal- atom bridges, and other means of peptide cyclization are used to stabilize the alpha helix.
- enhanced activity at the GLP- 1 receptor may be achieved by stabilizing the alpha-helix structure in the C-terminal portion of the glucagon peptide (around amino acids 12-29) through introduction of one or more ⁇ , ⁇ -disubstituted amino acids at positions that retain the desired activity.
- stabilization of the alpha-helix is accomplished in this manner without introduction of an intramolecular bridge such as a salt bridge or covalent bond.
- an intramolecular bridge such as a salt bridge or covalent bond.
- glucagon peptide 17, 18, 19, 20, 21, 24 or 29 of a glucagon peptide is substituted with an ⁇ , ⁇ -disubstituted amino acid.
- substitution of position 16 of the Class 3 glucagon related analog peptide with amino iso-butyric acid (AIB) enhances GLP-I activity, in the absence of a salt bridge or lactam.
- AIB iso-butyric acid
- one, two, three or more of positions 16, 20, 21 or 24 are substituted with AIB.
- Enhanced activity at the GLP- 1 receptor may be achieved by an amino acid modification at position 20.
- the glutamine at position 20 is replaced with another hydrophilic amino acid having a side chain that is either charged or has an ability to hydrogen-bond, and is at least about 5 (or about 4-6) atoms in length, for example, lysine, citrulline, arginine, or ornithine.
- GLP-I activity at the GLP-I receptor is demonstrated in Class 3 glucagon related analog peptides comprising the C-terminal extension of SEQ ID NO: 26.
- GLP-I activity in such Class 3 glucagon related analog peptides comprising SEQ ID NO: 26 can be further increased by modifying the amino acid at position 18, 28 or 29, or at position 18 and 29, as described herein.
- a further modest increase in GLP-I potency may be achieved by modifying the amino acid at position 10 to be Trp.
- the Class 3 glucagon related analog peptides can comprise modifications at position 16, at position 20, and at the C-terminal carboxylic acid group, optionally with a covalent bond between the amino acids at positions 16 and 20; can comprise modifications at position 16 and at the C-terminal carboxylic acid group; can comprise modifications at positions 16 and 20, optionally with a covalent bond between the amino acids at positions 16 and 20; or can comprise modifications at position 20 and at the C-terminal carboxylic acid group; optionally with the proviso that the amino acid at position 12 is not Arg; or optionally with the proviso that the amino acid at position 9 is not GIu.
- the Class 3 glucagon related analog peptides can be further modified to improve the peptide's solubility and stability in aqueous solutions at physiological pH, while retaining the high biological activity relative to native glucagon.
- Hydrophilic moieties as discussed herein can be attached to the Class 3 glucagon related analog peptide as further discussed herein.
- hydrophilic groups at positions 17, 21, and 24 of the Class 3 glucagon related analog peptide comprising SEQ ID NO: 9 or SEQ ID NO: 10 are anticipated to improve the solubility and stability of the high potency glucagon analog in solutions having a physiological pH. Introduction of such groups also increases duration of action, e.g. as measured by a prolonged half-life in circulation.
- the Class 3 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18 and SEQ ID NO: 19, wherein the side chain of an amino acid residue at one of position 16, 17, 21 or 24 of said Class 3 glucagon related analog peptide further comprises a polyethylene glycol chain, having a molecular weight selected from the range of about 500 to about 40,000 Daltons.
- the polyethylene glycol chain has a molecular weight selected from the range of about 500 to about 5,000 Daltons.
- the polyethylene glycol chain has a molecular weight of about 10,000 to about 20,000 Daltons.
- the polyethylene glycol chain has a molecular weight of about 20,000 to about 40,000 Daltons.
- Suitable hydrophilic moieties include any water soluble polymers known in the art, including the hydrophilic moieties described herein, homo- or co-polymers of PEG, and a monomethyl-substituted polymer of PEG (mPEG).
- the hydrophilic group comprises a polyethylene (PEG) chain.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 6 or SEQ ID NO: 7 wherein a PEG chain is covalently linked to the side chains of amino acids present at positions 21 and 24 of the Class 3 glucagon related analog peptide and the carboxy terminal amino acid of the Class 3 glucagon related analog peptide has the carboxylic acid group.
- the polyethylene glycol chain has an average molecular weight selected from the range of about 500 to about 10,000 Daltons.
- the pegylated Class 3 glucagon related analog peptide comprises two or more polyethylene chains covalently bound to the Class 3 glucagon related analog peptide wherein the total molecular weight of the glucagon chains is about 1,000 to about 5,000 Daltons.
- the pegylated glucagon agonist comprises a peptide consisting of SEQ ID NO: 5 or a glucagon agonist analog of SEQ ID NO: 5, wherein a PEG chain is covalently linked to the amino acid residue at position 21 and at position 24, and wherein the combined molecular weight of the two PEG chains is about 1,000 to about 5,000 Daltons. Charged C-terminus
- the solubility of the Class 3 glucagon related analog peptide comprising SEQ ID NO: 20 can be further improved, for example, by introducing one, two, three or more charged amino acid(s) to the C-terminal portion of glucagon peptide of SEQ ID NO: 20, preferably at a position C-terminal to position 27.
- a charged amino acid can be introduced by substituting a native amino acid with a charged amino acid, e.g. at positions 28 or 29, or alternatively by adding a charged amino acid, e.g. after position 27, 28 or 29.
- one, two, three or all of the charged amino acids are negatively charged. Additional modifications, e.g.
- an analog of the Class 3 glucagon related analog peptide of SEQ ID NO: 20 is provided wherein the analog differs from SEQ ID NO: 20 by 1 to 2 amino acid substitutions at positions 17-26, and, in one embodiment, the analog differs from the peptide of SEQ ID NO: 20 by an amino acid substitution at position 20.
- the glucagon peptide is modified to comprise an acyl group, to prolong half-life in circulation and/or delay the onset of and/or extend the duration of action and/or improve resistance to proteases such as DPP-IV.
- the activity at the glucagon receptor and GLP-I receptor of the Class 3 glucagon related analog peptides is maintained after acylation. Further, the potency of the acylated analogs were comparable to the unacylated versions of the Class 3 glucagon related analog peptides.
- the invention provides a Class 3 glucagon related analog peptide modified to comprise an acyl group or alkyl group covalently linked to the amino acid at position 10 of the glucagon peptide.
- the glucagon peptide may further comprise a spacer between the amino acid at position 10 of the Class 3 glucagon related analog peptide and the acyl group or alkyl group.
- Any of the foregoing Class 3 glucagon related analog peptides may comprise two acyl groups or two alkyl groups, or a combination thereof.
- the acylated Class 3 glucagon related analog peptide comprises the amino acid sequence of any of SEQ ID NOs: 534-544 and 546-549. C-terminal truncation
- the Class 3 glucagon related analog peptides described herein are further modified by truncation or deletion of one or two amino acids of the C-terminus of the glucagon peptide (i.e., position 29 and/or 28) without affecting activity and/or potency at the glucagon and GLP-I receptors.
- the Class 3 glucagon related analog peptide can comprise amino acids 1-27 or 1-28 of the native glucagon peptide (SEQ ID NO: 1), optionally with one or more modifications described herein.
- the truncated Class 3 glucagon related analog peptide comprises SEQ ID NO: 550 or SEQ ID NO: 551.
- the truncated glucagon agonist peptide comprises SEQ ID NO: 552 or SEQ ID NO: 553.
- the Class 3 glucagon related analog peptides disclosed herein are modified by the addition of a second peptide to the carboxy terminus of the glucagon peptide, for example, SEQ ID NO: 26, SEQ ID NO: 27 or SEQ ID NO: 28.
- a Class 3 glucagon related analog peptide having a sequence selected from the group consisting of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19, SEQ ID NO: 66, SEQ ID NO: 67, SEQ ID NO: 68, and SEQ ID NO: 69 is covalently bound through a peptide bond to a second peptide, wherein the second peptide comprises a sequence selected from the group consisting of SEQ ID NO: 26, SEQ ID NO: 27 and SEQ ID NO: 28.
- Class 3 glucagon related analog peptides which comprise the C- terminal extension
- the threonine at position 29 of the native glucagon peptide is replaced with a glycine.
- a Class 3 glucagon related analog peptide having a glycine substitution for threonine at position 29 and comprising the carboxy terminal extension of SEQ ID NO: 26 is four times as potent at the GLP- 1 receptor as native glucagon modified to comprise the carboxy terminal extension of SEQ ID NO: 26. Potency at the GLP-I receptor can be further enhanced by an alanine substitution for the native arginine at position 18.
- Class 3 glucagon related analog peptide can have a carboxy terminal extension of SEQ ID NO: 27 (KRNRNNIA) or SEQ ID NO: 28.
- Class 3 glucagon related analog peptide comprising SEQ ID NO: 33 or SEQ ID NO: 20, further comprises the amino acid sequence of SEQ ID NO: 27 (KRNRNNIA) or SEQ ID NO: 28 linked to amino acid 29 of the glucagon peptide.
- the Class 3 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 13 SEQ ID NO: 14 and SEQ ID NO: 15, further comprising the amino acid sequence of SEQ ID NO: 27 (KRNRNNIA) or SEQ ID NO: 28 linked to amino acid 29 of the glucagon peptide.
- the glucagon peptide comprises a sequence selected from the group consisting of SEQ ID NO: 10, SEQ ID NO: 12, SEQ ID NO: 13 SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 66, SEQ ID NO: 67, SEQ ID NO: 68, SEQ ID NO: 69, SEQ ID NO: 55 and SEQ ID NO: 56 further comprising the amino acid sequence of SEQ ID NO: 26 (GPSSGAPPPS) or SEQ ID NO: 29 linked to amino acid 29 of the Class 3 glucagon related analog peptide.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 64.
- any of the modifications described above with regard to Class 3 glucagon related analog peptides which increase or decrease glucagon receptor activity and which increase GLP-I receptor activity can be applied individually or in combination. Combinations of the modifications that increase GLP-I receptor activity generally provide higher GLP-I activity than any of such modifications taken alone. Any of the modifications described above can also be combined with other modifications described herein in reference to Class 3 glucagon related analog peptides that confer other desirable properties, such as increased solubility and/or stability and/or duration of action. Alternatively, any of the modifications described above can be combined with other modifications described herein in reference to Class 3 glucagon related analog peptides that do not substantially affect solubility or stability or activity. Exemplary modifications include but are not limited to:
- (A) Improving solubility for example, by introducing one, two, three or more charged amino acid(s) to the C-terminal portion of native glucagon, preferably at a position C-terminal to position 27.
- a charged amino acid can be introduced by substituting a native amino acid with a charged amino acid, e.g. at positions 28 or 29, or alternatively by adding a charged amino acid, e.g. after position 27, 28 or 29.
- one, two, three or all of the charged amino acids are negatively charged.
- one, two, three or all of the charged amino acids are positively charged.
- Such modifications increase solubility, e.g. provide at least 2-fold, 5-fold, 10-fold, 15-fold, 25-fold, 30-fold or greater solubility relative to native glucagon at a given pH between about 5.5 and 8, e.g., pH 7, when measured after 24 hours at 25°C.
- (C) Increasing stability by modification of the aspartic acid at position 15, for example, by deletion or substitution with glutamic acid, homoglutamic acid, cysteic acid or homocysteic acid. Such modifications can reduce degradation or cleavage at a pH within the range of 5.5 to 8, especially in acidic or alkaline buffers, for example, retaining at least 75%, 80%, 90%, 95%, 96%, 97%, 98% or 99% of the original peptide after 24 hours at 25°C. .
- (D) Increasing stability by modification of the methionine at position 27, for example, by substitution with leucine or norleucine. Such modifications can reduce oxidative degradation. Stability can also be increased by modification of the GIn at position 20 or 24, e.g. by substitution with Ala or AIB. Such modifications can reduce degradation that occurs through deamidation of GIn. Stability can be increased by modification of Asp at position 21, e.g. by substitution with GIu. Such modifications can reduce degradation that occurs through dehydration of Asp to form a cyclic succinimide intermediate followed by isomerization to iso-aspartate.
- DPP IV dipeptidyl peptidase IV
- substitution of His at position 1 with a large, aromatic amino acid e.g., Tyr, Phe, Trp or amino-Phe
- Ser at position 2 with Ala substitution of Tyr at position 10 with VaI or Phe
- substitution of Lys at position 12 with Arg substitution of Asp at position 15 with GIu
- substitution of Ser at position 16 with Thr or AIB Class 3 glucagon related analog peptides with GLP-I activity that contain a non- conservative substitution of His at position 1 with a large, aromatic amino acid (e.g., Tyr) can retain GLP- 1 activity provided that the alpha-helix is stabilized via an intramolecular bridge, e.g., such as any of those described herein.
- Class 3 glucagon related analog peptide can be linked, optionally via covalent bonding and optionally via a linker, to a conjugate moiety.
- the Class 3 glucagon related analog peptide also can be part of a fusion peptide or protein wherein a second peptide or polypeptide has been fused to a terminus, e.g., the carboxy terminus of the Class 3 glucagon related analog peptide.
- the fusion Class 3 glucagon related analog peptide may comprise a glucagon agonist of SEQ ID NO: 55, SEQ ID NO: 9 or SEQ ID NO: 10 further comprising an amino acid sequence of SEQ ID NO: 26 (GPSSGAPPPS), SEQ ID NO: 27 (KRNRNNIA) or SEQ ID NO: 28 (KRNR) linked to amino acid 29 of the glucagon peptide.
- the amino acid sequence of SEQ ID NO: 26 is bound to amino acid 29 of the Class 3 glucagon related analog peptide through a peptide bond.
- SEQ ID NO: 26 GPSSGAPPPS
- SEQ ID NO: 27 KRNRNNIA
- SEQ ID NO: 28 KRNR
- substitution of the native threonine residue at position 29 with glycine dramatically increases GLP-I receptor activity.
- a Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 64.
- the Class 3 glucagon related analog peptide portion of the glucagon fusion peptide is selected from the group consisting of SEQ ID NO: 55, SEQ ID NO: 2, SEQ ID NO: 3, SEQ ID NO: 4, and SEQ ID NO: 5 wherein a PEG chain, when present at positions 17, 21, 24, or the C-terminal amino acid, or at both 21 and 24, is selected from the range of 500 to 40,000 Daltons. More particularly, in one embodiment, the Class 3 glucagon related analog peptide segment is selected from the group consisting of SEQ ID NO: 7, SEQ ID NO: 8, and SEQ ID NO: 63, wherein the PEG chain is selected from the range of 500 to 5,000.
- the Class 3 glucagon related analog peptide is a fusion peptide comprising the sequence of SEQ ID NO: 55 and SEQ ID NO: 65 wherein the peptide of SEQ ID NO: 65 is linked to the carboxy terminus of SEQ ID NO: 55.
- an additional chemical modification of the Class 3 glucagon related analog peptide of SEQ ID NO: 10 bestows increased GLP-I receptor potency to a point where the relative activity at the glucagon and GLP-I receptors is virtually equivalent.
- a Class 3 glucagon related analog peptide comprises a terminal amino acid comprisng an amide group in place of the carboxylic acid group that is present on the native amino acid.
- the relative activity of the Class 3 glucagon related analog peptide at the respective glucagon and GLP-I receptors can be adjusted by further modifications to the Class 3 glucagon related analog peptide to produce analogs demonstrating about 40% to about 500% or more of the activity of native glucagon at the glucagon receptor and about 20% to about 200% or more of the activity of native GLP- 1 at the GLP-I receptor, e.g. 50-fold, 100-fold or more increase relative to the normal activity of glucagon at the GLP-I receptor.
- a Class 3 glucagon related analog peptide is an analog of SEQ ID NO: 55, wherein said analog differs from SEQ ID NO: 55 by 1 to 3 amino acids, selected from positions 1, 2, 3, 5, 7, 10, 11, 13, 14, 17, 18, 19, 21, 24, 27, 28, and 29, wherein said Class 3 glucagon related analog peptide exhibits at least 20% of the activity of native GLP-I at the GLP-I receptor.
- the Class 3 glucagon related analog peptide comprises the sequence:
- Xaa at position 15 is selected from the group of amino acids consisting of Asp, GIu, cysteic acid, homoglutamic acid and homocysteic acid
- Xaa at position 16 is selected from the group of amino acids consisting of Ser, GIu, GIn, homoglutamic acid and homocysteic acid
- the Xaa at position 20 is GIn or Lys
- the Xaa at position 24 is GIn or GIu
- the Xaa at position 28 is Asn, Lys or an acidic amino acid
- the Xaa at position 15 is selected from the group of amino acids consisting of Asp, GIu, cysteic acid, homoglutamic acid and homocysteic acid
- Xaa at position 16 is selected from the group of amino acids consisting of Ser, GIu, GIn, homoglutamic acid and homocysteic acid
- the Xaa at position 20 is GIn or Lys
- the Xaa at position 24 is GIn or
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 33 wherein the amino acid at position 28 is aspartic acid and the amino acid at position 29 is glutamic acid.
- the amino acid at position 28 is the native asparagine
- the amino acid at position 29 is glycine
- the amino acid sequence of SEQ ID NO: 29 or SEQ ID NO: 65 is covalently linked to the carboxy terminus of SEQ ID NO: 33.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 33 wherein an additional acidic amino acid added to the carboxy terminus of the peptide.
- the carboxy terminal amino acid of the Class 3 glucagon related analog peptide has an amide in place of the carboxylic acid group of the natural amino acid.
- the Class 3 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 40, SEQ ID NO: 41, SEQ ID NO: 42, SEQ ID NO: 43 and SEQ ID NO: 44.
- the Class 3 glucagon related analog peptide is an analog comprising SEQ ID NO: 33, wherein said analog differs from SEQ ID NO: 33 by 1 to 3 amino acids, selected from positions 1, 2, 3, 5, 7, 10, 11, 13, 14, 17, 18, 19, 21 and 27, with the proviso that when the amino acid at position 16 is serine, either position 20 is lysine, or a lactam bridge is formed between the amino acid at position 24 and either the amino acid at - I l l -
- the analog differs from SEQ ID NO: 33 by 1 to 3 amino acids selected from positions 1, 2, 3, 21 and 27.
- the glucagon peptide analog of SEQ ID NO: 33 differs from that sequence by 1 to 2 amino acids, or in one embodiment by a single amino acid, selected form positions 1, 2, 3, 5, 7, 10, 11, 13, 14, 17, 18, 19, 21 and 27, with the proviso that when the amino acid at position 16 is serine, either position 20 is lysine, or a lactam bridge is formed between the amino acid at position 24 and either the amino acid at position 20 or position 28.
- the Class 3 glucagon related analog peptide is a relatively selective GLP-I receptor agonist comprising the sequence NH 2 -His-Ser-Xaa- Gly-Thr-Phe- Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Xaa-Xaa-Arg-Arg-Ala-Xaa-Asp-Phe-Val- Xaa-Trp-Leu-Met-Xaa-Xaa-R (SEQ ID NO: 53) wherein the Xaa at position 3 is selected from the group of amino acids consisting of GIu, Orn or NIe, the Xaa at position 15 is selected from the group of amino acids consisting of Asp, GIu, cysteic acid, homoglutamic acid and homocysteic acid, Xaa at position 16 is selected from the group of amino acids consisting of Ser, GIu, GIn, homoglutamic acid and homo
- the amino acid at position 3 is glutamic acid.
- the acidic amino acid substituted at position 28 and/or 29 is aspartic acid or glutamic acid.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 33 further comprising an additional acidic amino acid added to the carboxy terminus of the peptide.
- the carboxy terminal amino acid of the Class 3 glucagon related analog peptide has an amide in place of the carboxylic acid group of the natural amino acid.
- the Class 3 glucagon related analog peptide comprises a modified glucagon peptide selected from the group consisting of:
- Xaa at position 15 is selected from the group of amino acids consisting of Asp, GIu, cysteic acid, homoglutamic acid and homocysteic acid
- Xaa at position 16 is selected from the group of amino acids consisting of Ser, GIu, GIn, homoglutamic acid and homocysteic acid
- the Xaa at position 20 is GIn or Lys
- the Xaa at position 24 is GIn or GIu
- the Xaa at position 28 is Asn, Asp or Lys
- R is COOH or CONH 2 ,
- R is CONH 2
- the Xaa at position 15 is Asp
- the Xaa at position 16 is selected from the group of amino acids consisting of GIu, GIn, homoglutamic acid and homocysteic acid
- the Xaas at positions 20 and 24 are each GIn the Xaa at position 28 is Asn or Asp
- the Xaa at position 29 is Thr.
- the Xaa's at positions 15 and 16 are each GIu
- the Xaas at positions 20 and 24 are each GIn
- the Xaa at position 28 is Asn or Asp
- the Xaa at position 29 is Thr
- R is CONH 2 .
- the Class 3 glucagon related analog peptide has enhanced GLP- 1 activity and comprises (a) an amino acid substitution of His at position 1 with a large, aromatic amino acid and (b) an intramolecular bridge that stabilizes that alpha- helix in the C-terminal portion of the molecule (e.g. around positions 12-29).
- the amino acid at position 1 is Tyr, Phe, Trp, amino-Phe, nitro-Phe, chloro-Phe, sulfo-Phe, 4-pyridyl-Ala, methyl-Tyr, or 3-amino Tyr.
- the intramolecular bridge is between the side chains of two amino acids that are separated by three intervening amino acids, i.e., between the side chains of amino acids i and i+4.
- the intramolecular bridge is a lactam bridge.
- the Class 3 glucagon related analog peptide comprises a large, aromatic amino acid at position 1 and a lactam bridge between the amino acids at positions 16 and 20 of the peptide.
- Such a Class 3 glucagon related analog peptide may further comprise one or more (e.g., two, three, four, five or more) of the other modifications described herein.
- the Class 3 glucagon related analog peptide can comprise an amide in place of the C-terminal carboxylate. Accordingly, in one embodiment, the Class 3 glucagon related analog peptide comprises that amino acid sequence of SEQ ID NO: 555.
- the Class 3 glucagon related analog peptide has enhanced GLP-I activity and comprises (a) one or more substitutions within amino acid positions 12-29 with an ⁇ , ⁇ -disubstituted amino acid and optionally, (b) a C-terminal amide.
- such Class 3 glucagon related analog peptides specifically lack an intramolecular bridge that stabilizes the alpha-helix in the C-terminal portion of glucagon (around positions 12-29).
- one, two, three, four or more of positions 16, 17, 18, 19, 20, 21, 24 or 29 of the Class 3 glucagon related analog peptide is substituted with an ⁇ , ⁇ -disubstituted amino acid, e.g., amino iso-butyric acid (AIB), an amino acid disubstituted with the same or a different group selected from methyl, ethyl, propyl, and n-butyl, or with a cyclooctane or cycloheptane (e.g., 1-aminocyclooctane-l-carboxylic acid).
- an amino acid e.g., amino iso-butyric acid (AIB)
- AIB amino acid disubstituted with the same or a different group selected from methyl, ethyl, propyl, and n-butyl
- a cyclooctane or cycloheptane e.g., 1-aminocycl
- substitution of position 16 with AIB enhances GLP-I activity, in the absence of a salt bridge or lactam.
- one, two, three or more of positions 16, 20, 21 or 24 are substituted with AIB.
- Such a Class 3 glucagon related analog peptide may further comprise one or more of the other modifications described herein, including, but not limited to, acylation, alkylation, pegylation, deletion of 1-2 amino acids at the C-terminus, addition of and/or substitution with charged amino acids at the C-terminus, replacement of the C-terminal carboxylate with an amide, addition of a C-terminal extension, and conservative and/or non-conservative amino acid substitutions, such as substitution of Met at position 27 with Leu or NIe, substitution of Asp at position 15 with GIu (or like amino acid), substitution at position 1 and/or 2 with amino acids which achieve DPP-IV protease resistance, substitution of Ser at position 2 with Ala, substitution of Tyr at position 10 with VaI or Phe, substitution
- the foregoing Class 3 glucagon related analog peptide comprises a GIn or GIy at position 29 or addition of a C-terminal extension, e.g., GGPSSGAPPPS (SEQ ID NO: 26) C-terminal to the amino acid at position 28.
- the Class 3 glucagon related analog peptide comprises one or more of an amide group in place of the C-terminal carboxylate, an acyl group, e.g., a C 16 fatty acid, and a hydrophilic moiety, e.g., a polyethylene glycol (PEG).
- the Class 3 glucagon related analog peptide comprises the amino acid sequence of any of SEQ ID NOs: 1-25, 30-64, and 66-555 comprising no more than ten modifications relative to SEQ ID NO: 1 and comprising one or more amino acid substitutions with AIB at positions 16, 20, 21, and/or 24, wherein the peptide lacks an intramolecular bridge between the side chains of two amino acids of the peptide.
- the Class 3 glucagon related analog peptide comprises the amino acid sequence of any of SEQ ID NOs: 556-561. It has been reported that certain positions of the native glucagon peptide can be modified while retaining at least some of the activity of the parent peptide.
- one or more of the amino acids located at positions at positions 2, 5, 7, 10, 11, 12, 13, 14, 17, 18, 19, 20, 21, 24, 27, 28 or 29 of the Class 3 glucagon related analog peptide of SEQ ID NO: 11 can be substituted with an amino acid different from that present in the native glucagon peptide, and still retain activity at the glucagon receptor.
- the methionine residue present at position 27 of the native peptide is changed to leucine or norleucine to prevent oxidative degradation of the peptide.
- the amino acid at position 20 is substituted with Lys, Arg, Orn or Citrullene and/or position 21 is substituted with GIu, homoglutamic acid or homocysteic acid.
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 20 with an additional 1 to 6 amino acid changes, selected from positions 1, 2, 5, 7, 10, 11, 13, 14, 17, 18, 19, 21, 27, 28 or 29, differing from the corresponding amino acid of SEQ ID NO: 1, with the proviso that when the amino acid at position 16 is serine, position 20 is Lys, or alternatively when position 16 is serine the position 24 is GIu and either position 20 or position 28 is Lys.
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 20 with 1 to 3 amino acids selected from positions 1, 2, 5, 7, 10, 11, 13, 14, 17, 18, 19, 20, 21, 27, 28 or 29 differing from the corresponding amino acid of SEQ ID NO: 1.
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 8, SEQ ID NO: 9 or SEQ ID NO: 11 with 1 to 2 amino acids selected from positions 1, 2, 5, 7, 10, 11, 13, 14, 17, 18, 19, 20 or 21 differing from the corresponding amino acid of SEQ ID NO: 1, and in a further embodiment, the one to two differing amino acids represent conservative amino acid substitutions relative to the amino acid present in the native glucagon sequence (SEQ ID NO: 1).
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14 or SEQ ID NO: 15 further comprising one, two or three amino acid substitutions at positions selected from positions 2, 5, 7, 10, 11, 13, 14, 17, 18, 19, 20, 21, 27 or 29.
- the substitutions at positions 2, 5, 7, 10, 11, 13, 14, 16, 17, 18, 19, 20, 21, 27 or 29 are conservative amino acid substitutions.
- the Class 3 glucagon related analog peptide comprises a variant sequence of SEQ ID NO: 33, wherein 1 to 10 amino acids selected from positions 16, 17, 18, 20, 21, 23, 24, 27, 28 and 29, respectively, differ from the corresponding amino acid of SEQ ID NO: 1.
- the Class 3 glucagon related analog peptide differs from SEQ ID NO: 33 by one or more amino acid substitutions selected from the group consisting of Glnl7, Alal8, Glu21, Ile23, Ala24, Val27 and Gly29.
- the Class 3 glucagon related analog peptide comprises variant sequences of SEQ ID NO: 33, wherein 1 to 2 amino acids selected from positions 17- 26 differ from the corresponding amino acid of SEQ ID NO: 1.
- the Class 3 glucagon related analog peptide comprises a variant sequence of SEQ ID NO: 33, which differs from SEQ ID NO: 33 by an amino acid substitution selected from the group consisting of Glnl7, Alal8, Glu21, Ile23 and Ala24.
- the variant sequence of SEQ ID NO 33 differs from SEQ ID NO: 33 by an amino acid substitution at position 18 wherein the substituted amino acid is selected from the group consisting of Ala, Ser, Thr, and GIy. In accordance with one embodiment, the variant sequence of SEQ ID NO 33 differs from SEQ ID NO: 33 by an amino acid substitution of Ala at position 18. Such variations are encompassed by SEQ ID NO: 55.
- the Class 3 glucagon related analog peptide comprises a variant sequence of SEQ ID NO: 33, wherein 1 to 2 amino acids selected from positions 17-22 differ from the corresponding amino acid of SEQ ID NO: 1, and in a further embodiment, the variant sequence of SEQ ID NO: 33 variant differs from SEQ ID NO: 33 by 1 or 2 amino acid substitutions at positions 20 and 21.
- Class 3 glucagon related analog peptide comprises the sequence:
- R is CONH 2 .
- the Class 3 glucagon related analog peptide comprises a variant sequence of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 47, SEQ ID NO: 48 or SEQ ID NO: 49, which differs from said sequence by an amino acid substitution at position 20.
- the amino acid substitution is selected form the group consisting of Lys, Arg, Orn or citrulline for position 20.
- the Class 3 glucagon related analog peptide comprises an analog peptide sequence of SEQ ID NO: 34 which differs from SEQ ID NO: 34 by having an amino acid other than serine at position 2.
- the serine residue is substituted with aminoisobutyric acid, D-alanine, and in one embodiment the serine residue is substituted with aminoisobutyric acid.
- Such modifications suppresses cleavage by dipeptidyl peptidase IV while retaining the inherent potency of the parent compound (e.g. at least 75, 80, 85, 90, 95% or more of the potentcy of the parent compound).
- the solubility of the analog is increased, for example, by introducing one, two, three or more charged amino acid(s) to the C-terminal portion of native glucagon, preferably at a position C-terminal to position 27.
- one, two, three or all of the charged amino acids are negatively charged.
- the Class 3 glucagon related analog peptide further comprises an acidic amino acid substituted for the native amino acid at position 28 or 29 or an acidic amino acid added to the carboxy terminus of the peptide of SEQ ID NO: 34.
- the Class 3 glucagon related analog peptides disclosed herein are further modified at position 1 or 2 to reduce susceptibility to cleavage by dipeptidyl peptidase IV.
- the Class 3 glucagon related analog peptide comprises a sequence of SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14 or SEQ ID NO: 15 which differs from the parent molecule by a substitution at position 2 and exhibits reduced susceptibility (i.e., resistance) to cleavage by dipeptidyl peptidase IV.
- position 2 of the Class 3 glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-serine, D-alanine, valine, ⁇ -amino isobutyric acid, glycine, N-methyl serine and ⁇ -amino butyric acid.
- position 2 of the Class 3 glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-serine, D-alanine, glycine, N-methyl serine and aminoisobutyric acid.
- position 2 of the Class 3 glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-serine, glycine, N-methyl serine and ⁇ -amino butyric acid.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 21 or SEQ ID NO: 22.
- the Class 3 glucagon related analog peptide comprises a vanant sequence of SEQ ID NO: 9, SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14 or SEQ ID NO: 15 which differs from the parent molecule by a substitution at position 1 and exhibits reduced susceptibility (i.e., resistance) to cleavage by dipeptidyl peptidase IV.
- position 1 of the Class 3 glucagon related analog peptide is substituted with an amino acid selected from the group consisting of D-histidine, alpha, alpha-dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha-methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo-histidine.
- the Class 3 glucagon related analog peptide comprises an analog peptide of SEQ ID NO: 34 wherein the analog differs from SEQ ID NO: 34 by having an amino acid other than histidine at position 1.
- the solubility of the analog is increased, for example, by introducing one, two, three or more charged amino acid(s) to the C-terminal portion of native glucagon, preferably at a position C-terminal to position 27.
- one, two, three or all of the charged amino acids are negatively charged.
- the analog further comprises an acidic amino acid substituted for the native amino acid at position 28 or 29 or an acidic amino acid added to the carboxy terminus of the peptide of SEQ ID NO: 34.
- the acidic amino acid is aspartic acid or glutamic acid.
- the Class 3 glucagon related analog peptide comprises a sequence of SEQ ID NO: 20 further comprising an additional carboxy terminal extension of one amino acid or a peptide selected from the group consisting of SEQ ID NO: 26, SEQ ID NO: 27 and SEQ ID NO: 28.
- the amino acid is typically selected from one of the 20 common amino acids, and, in one embodiment, the additional carboxy terminus amino acid has an amide group in place of the carboxylic acid of the native amino acid.
- the additional amino acid is selected from the group consisting of glutamic acid, aspartic acid and glycine.
- the Class 3 glucagon related analog peptide comprises at least one lactam ring formed between the side chain of a glutamic acid residue and a lysine residue, wherein the glutamic acid residue and a lysine residue are separated by three amino acids.
- the carboxy terminal amino acid of the lactam bearing Class 3 glucagon related analog peptide has an amide group in place of the carboxylic acid of the native amino acid. More particularly, in one embodiment, the Class 3 glucagon related analog peptide comprises a modified glucagon peptide selected from the group consisting of:
- Xaa at position 28 Asp, or Asn
- the Xaa at position 29 is Thr or GIy
- R is selected from the group consisting of COOH, CONH 2 , glutamic acid, aspartic acid, glycine, SEQ ID NO: 26, SEQ ID NO: 27 and SEQ ID NO: 28, and a lactam bridge is formed between Lys at position 12 and GIu at position 16 for SEQ ID NO: 66, between GIu at position 16 and Lys at position 20 for SEQ ID NO: 67, between Lys at position 20 and GIu at position 24 for SEQ ID NO: 68, between GIu at position 24 and Lys at position 28 for SEQ ID NO: 69, between Lys at position 12 and GIu at position 16 and between Lys at position 20 and GIu at position 24 for S
- R is selected from the group consisting of COOH, CONH 2 , glutamic acid, aspartic acid, glycine, the amino acid at position 28 is Asn, and the amino acid at position 29 is threonine.
- R is CONH 2 , the amino acid at position 28 is Asn and the amino acid at position 29 is threonine.
- R is selected from the group consisting of SEQ ID NO: 26, SEQ ID NO: 29 and SEQ ID NO: 65 and the amino acid at position 29 is glycine.
- the Class 3 glucagon related analog peptide is selected from the group consisting of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17 and SEQ ID NO: 18, wherein the peptide further comprises an additional carboxy terminal extension of one amino acid or a peptide selected from the group consisting of SEQ ID NO: 26, SEQ ID NO: 27 and SEQ ID NO: 28.
- the terminal extension comprises the sequence of SEQ ID NO: 26, SEQ ID NO: 29 or SEQ ID NO: 65 and the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 55.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 33 wherein the amino acid at position 16 is glutamic acid, the amino acid at position 20 is lysine, the amino acid at position 28 is asparagine and the amino acid sequence of SEQ ID No: 26 or SEQ ID NO: 29 is linked to the carboxy terminus of SEQ ID NO: 33.
- the amino acid at position 16 is glutamic acid
- the amino acid at position 20 is lysine
- the amino acid at position 28 is asparagine
- the amino acid sequence of SEQ ID No: 26 or SEQ ID NO: 29 is linked to the carboxy terminus of SEQ ID NO: 33.
- the amino acid is typically selected from one of the 20 common amino acids, and in one embodiment the amino acid has an amide group in place of the carboxy lie acid of the native amino acid.
- the additional amino acid is selected from the group consisting of glutamic acid and aspartic acid and glycine.
- the carboxy terminal amino acid of the extension in one embodiment, ends in an amide group or an ester group rather than a carboxylic acid.
- the Class 3 glucagon related analog peptide comprises the sequence: NH 2 -His-Ser-Gln-Gly-Thr-Phe- Thr-Ser-Asp-Tyr-Ser-Lys-Tyr-Leu-Asp-Glu-Arg- Arg-Ala-Gln-Asp-Phe-Val-Gln-Trp-Leu-Met-Asn-Thr-Xaa-CONHz (SEQ ID NO: 19), wherein the Xaa at position 30 represents any amino acid.
- Xaa is selected from one of the 20 common amino acids, and, in one embodiment, the amino acid is glutamic acid, aspartic acid or glycine.
- the solubility of this peptide can be further improved by covalently linking a PEG chain to the side chain of amino acid at position 17, 21, 24 or 30 of SEQ ID NO: 19.
- the Class 3 glucagon related analog peptide comprises an additional carboxy terminal extension of a peptide selected from the group consisting of SEQ ID NO: 26, SEQ ID NO: 27 and SEQ ID NO: 28.
- the glucagon/GLP-1 receptor co-agonist comprises the sequence of SEQ ID NO: 30, SEQ ID NO: 31 and SEQ ID NO: 32.
- SEQ ID NO: 11 SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, SEQ ID NO: 19 and SEQ ID NO: 64 can be made to yield a set of Class 3 glucagon related analog peptides that possess variable degrees of GLP-I agonism. Accordingly, peptides that possess virtually identical in vitro potency at each receptor have been prepared and characterized. Similarly, peptides with tenfold selectively enhanced potency at each of the two receptors have been identified and characterized.
- substitution of the serine residue at position 16 with glutamic acid enhances the potency of native glucagon at both the glucagon and GLP-I receptors, but maintains approximately a tenfold selectivity for the glucagon receptor.
- substitution of the native glutamine at position 3 with glutamic acid (SEQ ID NO: 22) generates a glucagon analog that exhibits approximately a tenfold selectivity for the GLP- 1 receptor.
- the solubility of the Class 3 glucagon related analog peptides can be further enhanced in aqueous solutions at physiological pH, while retaining the high biological activity relative to native glucagon by the introduction of hydrophilic groups at positions 16, 17, 21, and 24 of the peptide, or by the addition of a single modified amino acid (i.e., an amino acid modified to comprise a hydrophilic group) at the carboxy terminus of the Class 3 glucagon related analog peptide.
- the hydrophilic group comprises a polyethylene (PEG) chain.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 10, SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17 or SEQ ID NO: 18 wherein a PEG chain is covalently linked to the side chain of an amino acids at position 16, 17, 21, 24, 29 or the C-terminal amino acid of the Class 3 glucagon related analog peptide, with the proviso that, when the peptide comprises SEQ ID NO: 10, SEQ ID NO: 11, SEQ ID NO: 12 or SEQ ID NO: 13, the polyethylene glycol chain is covalently bound to an amino acid residue at position 17, 21 or 24, when the peptide comprises SEQ ID NO: 14 or SEQ ID NO: 15 the polyethylene glycol chain is covalently bound to an amino acid residue at position 16, 17 or 21, and when the peptide comprises SEQ ID NO: 16, SEQ ID NO: 17 or SEQ ID NO: 18, the sequence of SEQ ID NO
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 11, SEQ ID NO: 12 or SEQ ID NO: 13, wherein a PEG chain is covalently linked to the side chain of an amino acids at position 17, 21, 24, or the C-terminal amino acid of the Class 3 glucagon related analog peptide, and the carboxy terminal amino acid of the peptide has an amide group in place of the carboxylic acid group of the native amino acid.
- the Class 3 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18 and SEQ ID NO: 19, wherein a PEG chain is covalently linked to the side chain of an amino acid at position 17, 21 or 24 of SEQ ID NO: 12, SEQ ID NO: 13 and SEQ ID NO: 19, or at position 16, 17 or 21 of SEQ ID NO: 14 and SEQ ID NO: 15 or at position 17 or 21 of SEQ ID NO: 16, SEQ ID NO: 17 and SEQ ID NO: 18 of the Class 3 glucagon related analog peptide.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 11 or SEQ ID NO: 19, wherein a PEG chain is covalently linked to the side chain of an amino acids at position 17, 21 or 24 or the C-terminal amino acid of the Class 3 glucagon related analog peptide.
- the Class 3 glucagon related analog peptide is modified to contain one or more amino acid substitution at positions 16, 17, 21, 24, or 29 or the C- terminal amino acid, wherein the native amino acid is substituted with an amino acid having a side chain suitable for crosslinking with hydrophilic moieties, including for example, PEG.
- the native peptide can be substituted with a naturally occurring amino acid or a synthetic (non-naturally occurring) amino acid.
- Synthetic or non-naturally occurring amino acids refer to amino acids that do not naturally occur in vivo but which, nevertheless, can be incorporated into the peptide structures described herein.
- the amino acid having a side chain suitable for crosslinking with hydrophilic moieties can be added to the carboxy terminus of any of the Class 3 glucagon related analog peptides disclosed herein.
- an amino acid substitution is made in the Class 3 glucagon related analog peptide at a position selected from the group consisting of 16, 17, 21, 24, or 29 replacing the native amino acid with an amino acid selected from the group consisting of lysine, cysteine, ornithine, homocysteine and acetyl phenylalanine, wherein the substituting amino acid further comprises a PEG chain covalently bound to the side chain of the amino acid.
- the Class 3 glucagon related analog peptide selected form the group consisting of SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17, SEQ ID NO: 18, and SEQ ID NO: 19 is further modified to comprise a PEG chain is covalently linked to the side chain of an amino acid at position 17 or 21 of the glucagon peptide.
- the pegylated Class 3 glucagon related analog peptide further comprises the sequence of SEQ ID NO: 26, SEQ ID NO: 27 or SEQ ID NO: 29.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 55 or SEQ ID NO: 56, further comprising a C-terminal extension of SEQ ID NO: 26, SEQ ID NO: 29 or SEQ ID NO: 65 linked to the C-terminal amino acid of SEQ ID NO: 55 or SEQ ID NO: 56, and optionally further comprising a PEG chain covalently linked to the side chain of an amino acids at position 17, 18, 21, 24 or 29 or the C- terminal amino acid of the peptide.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 55 or SEQ ID NO: 56, wherein a PEG chain is covalently linked to the side chain of an amino acids at position 21 or 24 of the Class 3 glucagon related analog peptide and the Class 3 glucagon related analog peptide further comprises a C-terminal extension of SEQ ID NO: 26, or SEQ ID NO: 29.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 55, or SEQ ID NO: 33 or SEQ ID NO: 34, wherein an additional amino acid is added to the carboxy terminus of SEQ ID NO: 33 or SEQ ID NO: 34, and a PEG chain is covalently linked to the side chain of the added amino acid.
- the pegylated Class 3 glucagon related analog peptide further comprises a C- terminal extension of SEQ ID NO: 26 or SEQ ID NO: 29 linked to the C-terminal amino acid of SEQ ID NO: 33 or SEQ ID NO: 34.
- the Class 3 glucagon related analog peptide comprises the sequence of SEQ ID NO: 19, wherein a PEG chain is covalently linked to the side chain of the amino acid at position 30 of the Class 3 glucagon related analog peptide and the Class 3 glucagon related analog peptide further comprises a C- terminal extension of SEQ ID NO: 26 or SEQ ID NO: 29 linked to the C-terminal amino acid of SEQ ID NO: 19.
- the Class 3 glucagon related analog peptide comprises a sequence of SEQ ID NO: 20, wherein a lactam ring is formed between the side chains of a lysine residue, located at position 12, 20 or 28, and a glutamic acid residue, located at position 16 or 24, wherein the two amino acids of the glucagon peptide whose side chains participate in forming the lactam ring are spaced from one another by three intervening amino acids.
- the lactam bearing Class 3 glucagon related analog peptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17 and SEQ ID NO: 18.
- the carboxy terminal amino acid of the lactam bearing peptide comprises an amide group or an ester group in place of the terminal carboxylic acid.
- the Class 3 glucagon related analog peptide comprises SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, and SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17 and SEQ ID NO: 18 further comprising an additional amino acid covalently bound to the carboxy terminus of SEQ ID NO: 11, SEQ ID NO: 12, SEQ ID NO: 13, SEQ ID NO: 14, SEQ ID NO: 15, SEQ ID NO: 16, SEQ ID NO: 17 or SEQ ID NO: 18.
- the Class 3 glucagon related analog peptide comprises a sequence selected from the group consisting of SEQ ID NO: 66, SEQ ID NO: 67, SEQ ID NO: 68 and SEQ ID NO: 69 further comprising an additional amino acid covalently bound to the carboxy terminus of SEQ ID NO: 66, SEQ ID NO: 67, SEQ ID NO: 68 and SEQ ID NO: 69.
- the amino acid at position 28 is asparagine or lysine and the amino acid at position 29 is threonine.
- the Class 3 glucagon related analog peptide comprises the amino acid sequence of native glucagon (SEQ ID NO: 1) comprising the following modifications: AIB at position 2, GIu at position 3, Lys at position 10, GIu at position 16, GIn at position 17, Ala at position 18, Lys at position 20, GIu at position 21, He at position 23, Ala at position 24; wherein Lys at position 10 is acylated with a C14 or C16 fatty acid, and wherein the C-terminal carboxylate is replaced with an amide.
- this Class 3 glucagon related analog peptide is attached via its N-terminal amino acid to the dipeptide D-Lys-Sarcosine.
- the Class 3 glucagon related analog peptide comprises, consists essentially of, or consists of an amino acid sequence of any of SEQ ID NOs: 70-514, 517-534, or 554, optionally with up to 1, 2, 3, 4, or 5 further modifications that retain GLP-I agonist and/or glucagon agonist activity.
- a glucagon related analog peptide is a Class 4 glucagon related analog peptide ⁇ see, e.g., International (PCT) Patent Application No. PCT/US2008/080973, incorporated herein by reference in its entirety).
- Activity e.g., International (PCT) Patent Application No. PCT/US2008/080973, incorporated herein by reference in its entirety.
- Class 4 glucagon related analog peptides are provided (hereafter refered to as "Class 4 peptides").
- a Class 4 peptide which has glucagon antagonist activity.
- a glucagon antagonists would be used in any setting where the suppression of glucagon agonism is desired. The most immediate and obvious use would be in the treatment of diabetes where glucagon antagonism has been demonstrated in pre-clinical models of hyperglycemia to yield a lowering of blood glucose.
- Glucagon antagonists can be further modified to improve the biophysical stability and/or aqueous solubility of the compounds while maintaining the antagonist activity of the parent compound.
- a Class 4 peptide is defined as a pure glucagon antagonist.
- the term "glucagon antagonist" refers to a compound that counteracts glucagon activity or prevents glucagon function.
- a glucagon antagonist exhibits at least 60% inhibition (e.g., at least 70% inhibition) and preferably, at least 80% inhibition, of the maximum response achieved by glucagon at the glucagon receptor.
- the glucagon antagonist exhibits at least 90% inhibition of the maximum response achieved by glucagon at the glucagon receptor.
- the glucagon antagonist exhibits 100% inhibition of the maximum response achieved by glucagon at the glucagon receptor.
- a glucagon antagonist at a concentration of about 1 ⁇ M exhibits less than about 20% of the maximum agonist activity achieved by glucagon at the glucagon receptor. In one embodiment, the glucagon antagonist exhibits less than about 10% of the maximum agonist activity achieved by glucagon at the glucagon receptor. In a specific embodiment, the glucagon antagonist exhibits less than about 5% of the maximum agonist activity achieved by glucagon at the glucagon receptor. In yet another specific embodiment, the glucagon antagonist exhibits 0% of the maximum agonist activity achieved by glucagon at the glucagon receptor.
- a "pure glucagon antagonist” is a glucagon antagonist that does not produce any detected stimulation of glucagon or GLP-I receptor activity, as measured by cAMP production using a validated in vitro model assay ⁇ see, e.g., PCT/US2008/080973).
- a pure glucagon antagonist exhibits less than about 5% (e.g., less than about 4%, less than about 3%, less than about 2%, less than about 1%, about 0%) of the maximum agonist activity achieved by glucagon at the glucagon receptor and exhibits less than about 5% (e.g., less than about 4%, less than about 3%, less than about 2%, less than about 1%, and about 0%) of the maximum agonist activity achieved by GLP-I at the GLP-I receptor.
- Class 4 peptides that exhibit pure glucagon antagonist activity.
- the glucagon antagonist exhibits activity that reduces glucagon receptor glucagon-induced cAMP production by a maximum of at least 50% when the glucagon receptor is contacted simultaneously with 0.8nM of glucagon and the glucagon antagonist, as measured by cAMP production in an in vitro assay.
- the glucagon antagonist reduces glucagon receptor glucagon-induced cAMP production by a maximum amount of at least 80%.
- Class 4 peptides are believed to be suitable for any use that has previously been described for glucagon antagonists. Accordingly, the Class 4 peptides described herein can be used to treat hyperglycemia, or treat other metabolic diseases that result from high blood levels of glucagon or high blood glucose levels.
- the patient to be treated using the Class 4 peptides disclosed herein is a domesticated animal, and in another embodiment the patient to be treated is a human. Studies suggest that lack of glucagon suppression in diabetic patients contributes to postprandial hyperglycemia in part via accelerated glycogenosis.
- the Class 4 peptides of the present invention can be used to treat hyperglycemia, and are expected to be useful for treating a variety of types of diabetes including diabetes mellitus type I, diabetes mellitus type II, or gestational diabetes, either insulin-dependent or non-insulin-dependent, and reducing complications of diabetes including nephropathy, retinopathy and vascular disease.
- the terminal ten amino acids of Exendin-4 i.e. the sequence of SEQ ID NO: 919 (GPSSGAPPPS)
- GPS sequence of SEQ ID NO: 919
- these fusion proteins are anticipated to have pharmacological activity for suppressing appetite and inducing weight loss/weight maintenance.
- the Class 4 peptides disclosed herein can be further modified to include the amino acid sequence of SEQ ID NO: 919 (GPSSGAPPPS) linked to amino acid 24 of the Class 4 peptide of SEQ ID NO: 942 and administered to individuals to induce weight loss or assist in weight maintenance.
- the Class 4 peptide comprises a sequence selected from the group consisting of SEQ ID NO: 902, SEQ ID NO: 903, SEQ ID NO: 904 SEQ ID NO: 905, SEQ ID NO: 906, SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 936, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941, SEQ ID NO: 942, SEQ ID NO: 943 and SEQ ID NO: 944 and further comprising the amino acid sequence of SEQ ID NO: 919 (GPSSGAPPPS) linked to amino acid 24 of the Class 4 peptide is used to suppress appetite and inducing weight loss/weight maintenance.
- GPS amino acid sequence of SEQ ID NO: 919
- the administered Class 4 peptide comprises the sequence of SEQ ID NO: 946 or SEQ ID NO: 947.
- Such methods for reducing appetite or promoting loss of body weight are expected to be useful in reducing body weight, preventing weight gain, or treating obesity of various causes, including drug-induced obesity, and reducing complications associated with obesity including vascular disease (coronary artery disease, stroke, peripheral vascular disease, ischemia reperfusion, etc.), hypertension, onset of diabetes type II, hyperlipidemia and musculoskeletal diseases.
- the Class 4 peptides of the invention may be administered alone or in combination with other anti-diabetic or anti-obesity agents.
- Anti-diabetic agents known in the art or under investigation include insulin, sulfonylureas, such as tolbutamide (Orinase), acetohexamide (Dymelor), tolazamide (Tolinase), chlorpropamide (Diabinese), glipizide (Glucotrol), glyburide (Diabeta, Micronase, Glynase), glimepiride (Amaryl), or gliclazide (Diamicron); meglitinides, such as repaglinide (Prandin) or nateglinide (Starlix); biguanides such as metformin (Glucophage) or phenformin; thiazolidinediones such as rosiglitazone (Avandia), pioglitazone (Actos), or trogli
- Anti-obesity agents known in the art or under investigation include appetite suppressants, including phenethylamine type stimulants, phentermine (optionally with fenfluramine or dexfenfluramine), diethylpropion (Tenuate®), phendimetrazine (Prelu-2®, Bontril®), benzphetamine (Didrex®), sibutramine (Meridia®, Reductil®); rimonabant (Acomplia®), other cannabinoid receptor antagonists; oxyntomodulin; fluoxetine hydrochloride (Prozac); Qnexa (topiramate and phentermine), Excalia (bupropion and zonisamide) or Contrave (bupropion and naltrexone); or lipase inhibitors, similar to xenical (Orlistat) or Cetilistat (also known as ATL-962), or GT 389-255.
- appetite suppressants including phenethylamine type stimulants
- the Class 4 peptides of the present invention can also be administered to patients suffering from catabolic wasting. It is estimated that over half of cancer patients experience catabolic wasting which is characterized by unintended and progressive weight loss, weakness, and low body fat and muscle. The syndrome is equally common in AIDS patients and can also be present in bacterial and parasitic diseases, rheumatoid arthritis, and chronic diseases of the bowel, liver, lungs, and heart. It is usually associated with anorexia and can manifest as a condition in aging or as a result of physical trauma. Catabolic wasting is a symptom that diminishes the quality of life, worsens the underlying condition, and is a major cause of death. Applicants anticipate that the Class 4 peptides disclosed herein can be administered to patients to treat catabolic wasting.
- compositions comprising the Class 4 peptides disclosed herein can be formulated and administered to patients to using standard pharmaeuctically acceptable carriers and routes of administration known to those skilled in the art. Accordingly the present disclosure also encompasses pharmaceutical compositions comprising one or more of the Class 4 peptides disclosed herein in combination with a pharmaceutically acceptable carrier.
- the pharmaceutical compositions may comprise the Class 4 peptides as the sole pharmaceutically active component, or the Class 4 peptides can be combined with one or more additional active agents.
- a composition comprising a Class 4 peptide of the present invention and a compound that activates the GLP-I receptor (such as GLP-I, a GLP-I analog, an exendin-4 analog, or derivatives thereof).
- a composition comprising a Class 4 peptide of the present invention and insulin or an insulin analog.
- a composition provided for inducing weight loss or preventing weight gain can be provided that comprises the sequence of SEQ ID NO: 942 further comprising the amino acid sequence of SEQ ID NO: 919 (GPSSGAPPPS) linked to amino acid 24 of SEQ ID NO: 942, and an anti-obesity peptide.
- Suitable anti-obesity peptides include those disclosed in US patents 5,691,309, 6,436,435 or US Patent application 20050176643, and including, but not limited to GLP-I, GIP (Gastric Inhibitory Polypeptide), MPl , PYY, MC-4, Leptin.
- Class 4 glucagon related analog peptides are provided wherein the normally occurring aspartic acid at position nine (of glucagon, SEQ ID NO: 901) has been substituted with glutamic acid or a cysteic acid-based derivative. More particularly, deletion of the first amino acid (des-His) and substitution of the aspartic acid at position 9 with glutamic acid, in some aspects, produces a Class 4 peptide.
- Class 4 glucagon related analog peptides having sulfonic acid substituents substituted at amino acid position nine of glucagon perform similarly to the carboxylic acid-based amino acids but with a few critical differences in relation to physical properties such as solubility. Homocysteic acid (hCysSO 3 ) when substituted for the isosteric glutamic acid at position nine in the conventional des-His, Glu9 Class 4 peptide retains a partial antagonist and weak agonist.
- a Class 4 peptide wherein the first two to five amino acids are removed, and position 9 (according to the numbering of SEQ ID NO: 901) is replaced with hCys(SO 3 ), homoglutamic acid, ⁇ -homoglutamic acid, or an alkylcarboxylate derivative of cysteine having the structure of:
- OOH wherein X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl, provides a compound that performs as a hormonal antagonist that is highly specific, potent and without contaminating agonist properties.
- a Class 4 peptide comprises a glucagon peptide modified, relative to the wild type sequence of SEQ ID NO: 901, by the deletion of two to five amino acid residues from the N-terminus and substitution of the aspartic acid residue at position nine of the native protein with a glutamic acid, homoglutamic acid, ⁇ -homoglutamic acid, a sulfonic acid derivative of cysteine, or an alkylcarboxylate derivative of cysteine having the structure of:
- X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl.
- the Class 4 peptide comprising the deletion of two to five amino acid residues from the N-terminus and substitution of the Asp at position 9 of the native glucagon, is further modified by up to three amino acid modifications.
- the Class 4 peptide may comprise one, two, or three conservative amino acid modifications.
- the Class 4 peptide may comprise one or more amino acid modifications selected from the group consisting of:
- A substitution of one or two amino acids at positions 10, 20, and 24, (according to the amino acid numbering of SEQ ID NO: 901), or the N- or C-terminal amino acid of the Class 4 peptide with an amino acid covalently attached to an acyl group or alkyl group via an ester, ether, thioether, amide, or alkyl amine linkage;
- N- or C-terminal amino acid of the Class 4 peptide with an amino acid selected from the group consisting of: Cys, Lys, ornithine, homocysteine, and acetyl-phenylalanine (Ac-Phe), wherein the amino acid of the group is covalently bonded to a hydrophilic moiety;
- SEQ ID NO: 901 with charged amino acids; and/or addition of one to two charged amino acids at the C-terminus of SEQ ID NO: 901;
- the Class 4 peptide comprises an amino acid modification of A, B, or C, as described above, or a combination thereof. In yet another specific embodiment, the Class 4 peptide further comprises an amino acid modification of any of D to K as described above, or a combination thereof, in addition to the amino acid modification(s) of A, B, and/or C.
- the Class 4 peptide comprises a glucagon peptide, wherein the first 5 amino acids have been removed from the N-terminus, and the remaining N-terminal amino group has been replaced with a hydroxyl group (the "PLA6 analog"), producing the peptide of SEQ ID NO: 939.
- PVA6 analog a hydroxyl group
- substitution of phenyl-lactic acid for phenylalanine in Class 4 peptide analogs that have the first five amino acids deleted and substitution of a glutamic acid at position 9 (relative to native glucagon) further enhances the potency of those Class 4 peptide analogs.
- Class 4 peptide peptide of SEQ ID NO: 939 is further modified by substituting the aspartic acid residue at position four (position 9 of the native glucagon) with an amino acid of the general structure: wherein X 6 is C 1 -C 3 alkyl, C 2 -C3 alkene or C 2 -C3 alkynyl, and in one embodiment X 6 is C 1 -C 3 alkyl, and in another embodiment X 6 is C 2 alkyl.
- the Class 4 peptide comprises a glucagon peptide, wherein the first 5 amino acids have been removed from the N-terminus, and the aspartic acid residue at position four (position 9 of the native glucagon) has been substituted with cysteic acid or homocysteic acid.
- the Class 4 peptide comprises a glucagon peptide comprising an amino acid sequence selected from the group consisting of SEQ ID NO: 939, SEQ ID NO: 907 and SEQ ID NO: 908.
- the Class 4 peptide comprises an amino acid sequence selected from the group consisting of SEQ ID NO: 908, wherein the amino acid at position four is homocysteic acid.
- Class 4 peptide of SEQ ID NO: 939 is further modified by substituting the aspartic acid residue at position four (position 9 of the native glucagon) with glutamic acid, homoglutamic acid, ⁇ -homoglutamic acid, or an alkylcarboxylate derivative of cysteine having the structure of:
- Xs ⁇ COOH wherein X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl. In a specific embodiment, X 5 is C 1 or C 2 alkyl.
- Substitution of the phenylalanine residue with other phenylalanine analogs including 3,4-2F-phenylalnine (3,4-2F-Phe), 2-naphthyalanine (2-Nal), N-acyl-phenylalanine (Ac-Phe), alpha-methylhydrocinnamic acid (MCA) and benzylmalonic acid (BMA) did not perform as potently as the PLA substitution.
- Substituting PLA at sites other than at position six (according to the amino acid numbering of native glucagon), including at positions 4 and 5 reveals that the PLA6 analog is an appreciably more potent antagonist than glucagon analogs having a slightly extended N- terminus.
- the present invention also includes analogs wherein the N-terminal amino group is substituted with an acylated and alkylated "O-terminal" peptides.
- the PLA6 substitution not only increases the potency of the antagonist but also serves a critical role in pegylation.
- the PLA6 analogs can be selectively pegylated without restoration of glucagon agonism. In the absence of the PLA substitution, pegylation of the analog surprisingly induces glucagon agonism. This glucagon agonism is not seen in the pegylated PLA6 analogs.
- Several sites for pegylation were investigated including positions 3, 6 and 19 (positions 8, 11 and 19 of native glucagon) and at the N-terminal amino acid residue. In one embodiment the pegylation is at position 19 (position 24 of native glucagon) as that site exhibits the most potent and selective glucagon antagonism.
- the Class 4 peptide comprises the general structure of A-B-C, wherein A is selected from the group consisting of: (i) phenyl lactic acid (PLA);
- B represents amino acids i to 26 of SEQ ID NO: 901 , wherein i is 3, 4, 5, 6, or 7, optionally comprising one or more amino acid modifications selected from the group consisting of:
- Asp at position 9 (according to the amino acid numbering of SEQ ID NO: 901) is substituted with a GIu, a sulfonic acid derivative of Cys, homoglutamic acid, ⁇ -homoglutamic acid, or an alkylcarboxylate derivative of cysteine having the structure of: OOH , wherein X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl.
- substitution of one or two amino acids at positions 16, 17, 20, 21, and 24 (according to the amino acid numbering of SEQ ID NO: 901) with an amino acid selected from the group consisting of: Cys, Lys, ornithine, homocysteine, and acetyl- phenylalanine (Ac-Phe), wherein the amino acid of the group is covalently attached to a hydrophilic moiety;
- Asp at position 15 (according to the numbering of SEQ ID NO: 901) is substituted with cysteic acid, glutamic acid, homoglutamic acid, and homocysteic acid;
- Ser at position 16 is substituted with cysteic acid, glutamic acid, homoglutamic acid, and homocysteic acid; (ix) substitution with AIB at one or more of positions 16, 20, 21, and 24 according to the amino acid numbering of SEQ ID NO: 901; and C is selected from the group consisting of:
- the Class 4 peptide comprises an oxy derivative of PLA.
- oxy derivative of PLA refers to a compound comprising a modified structure of PLA in which the hydroxyl group has been replaced with O-R ⁇ , wherein Rn is a chemical moiety.
- the oxy derivative of PLA can be, for example, an ester of PLA or an ether of PLA.
- the ester may be formed by upon reaction of the hydroxyl of PLA with a carbonyl bearing a nucleophile.
- the nucleophile can be any suitable nucleophile, including, but not limited to an amine or hydroxyl. Accordingly, the ester of PLA can comprise the structure of Formula XI:
- the carbonyl bearing a nucleophile (which reacts with the hydroxyl of PLA to form an ester) can be, for example, a carboxylic acid, a carboxylic acid derivative, or an activated ester of a carboxylic acid.
- the carboxylic acid derivative can be, but is not limited to, an acyl chloride, an acid anhydride, an amide, an ester, or a nitrile.
- the activated ester of a carboxylic acid can be, for example, N-hydroxysuccinimide (NHS), tosylate (Tos), a carbodiimide, or a hexafluorophosphate.
- the carbodiimide is 1,3- dicyclohexylcarbodiimide (DCC), l,l'-carbonyldiimidazole (CDI), l-ethyl-3-(3- dimethylaminopropyl)carbodiimide hydrochloride (EDC), or 1,3-diisopropylcarbodiimide (DICD).
- DCC 1,3- dicyclohexylcarbodiimide
- CDI l,l'-carbonyldiimidazole
- EDC l-ethyl-3-(3- dimethylaminopropyl)carbodiimide hydrochloride
- DICD 1,3-diisopropylcarbodiimide
- the hexafluorophosphate is selected from a group consisting of hexafluorophosphate benzotriazol- 1 -yl-oxy-tris(dimethylamino)phosphonium hexafluorophosphate (BOP), benzotriazol- 1-yl- oxytripyrrolidinophosphonium hexafluorophosphate (PyBOP), 2-(lH-7-azabenzotriazol-l-yl)-l,l,3,3-tetramethyl uronium hexafluorophosphate (HATU), and o-benzotriazole-N,N,N',N'-tetramethyl-uronium- hexafluoro-phosphate (HBTU).
- BOP hexafluorophosphate benzotriazol- 1 -yl-oxy-tris(dimethylamino)phosphonium hexafluorophosphate
- PyBOP benzotriazol
- ethers from reaction with a hydroxyl group e.g., the hydroxyl of PLA
- a hydroxyl group of PLA may be reacted with a halogenated alkyl or tosylated alkyl alcohol to form an ether bond.
- the chemical moiety of Rn is one which does not decrease the activity of the Class 4 peptide. In some embodiments, the chemical moiety enhances the activity, stability, and/or solubility of the Class 4 peptide.
- the chemical moiety bound to PLA via an oxygen- containing bond is a polymer (e.g., a polyalkylene glycol), a carbohydrate, an amino acid, a peptide, or a lipid, e.g., a fatty acid or a steroid.
- a polymer e.g., a polyalkylene glycol
- carbohydrate e.g., an amino acid, a peptide, or a lipid, e.g., a fatty acid or a steroid.
- the chemical moiety is an amino acid, which, optionally, is a part of a peptide, such that Formula XI is a depsipeptide.
- PLA may be at a position other than the N-terminal amino acid residue of the Class 4 peptide, such that the Class 4 peptide comprises one or more (e.g., 1, 2, 3, 4, 5, 6, or more) amino acids N-terminal to the PLA residue.
- the Class 4 peptide can comprise PLA at position n, wherein n is 2, 3, 4, 5, or 6 of the Class 4 peptide.
- the amino acids N-terminal to the PLA residue may be synthetic or naturally- occurring.
- the amino acids which are N-terminal PLA are naturally-occurring amino acids.
- the amino acids which are N-terminal to PLA are the N-terminal amino acids of native glucagon.
- the Class 4 peptide can comprise at the N-terminus the amino acid sequence of any of SEQ ID NOs: 954-958, wherein PLA is linked to threonine via an ester bond:
- one or more of the N-terminal amino acids may be substituted with an amino acid other than the amino acid of native glucagon.
- the Class 4 peptide comprises PLA as the amino acid at position 5 or 6
- the amino acid at position 1 and/or position 2 may be an amino acid which reduces susceptibility to cleavage by dipeptidyl peptidase IV.
- position 1 of the Class 4 peptide is an amino acid selected from the group consisting of D-histidine, alpha, alpha- dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha- methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo- histidine.
- position 2 of the antagonist peptide is an amino acid selected from the group consisting of D-serine, D-alanine, valine, glycine, N- methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB).
- the Class 4 peptide comprises PLA as the amino acid at position 4, 5, or 6, the amino acid at position 3 of the Class 4 peptide may be glutamic acid, as opposed to the native glutamine residue of native glucagon.
- the Class 4 peptide comprises at the N-terminus the amino acid sequence of any of SEQ ID NOs: 959- 961.
- the polymer may be any polymer, provided that it can react with the hydroxyl group of PLA.
- the polymer may be one that naturally or normally comprises a carbonyl bearing a nucleophile.
- the polymer may be one which was derivatized to comprise the carbonyl bearing the carbonyl.
- the polymer may be a derivatized polymer of any of: polyamides, polycarbonates, polyalkylenes and derivatives thereof including, polyalkylene glycols, polyalkylene oxides, polyalkylene terepthalates, polymers of acrylic and methacrylic esters, including poly(methyl methacrylate), poly(ethyl methacrylate), poly(butylmethacrylate), poly(isobutyl methacrylate), poly(hexylmethacrylate), poly(isodecyl methacrylate), poly(lauryl methacrylate), poly (phenyl methacrylate), poly (methyl acrylate), poly(isopropyl acrylate), poly(isobutyl acrylate), and poly(octadecyl acrylate), polyvinyl polymers including polyvinyl alcohols, polyvinyl ethers, polyvinyl esters, polyvinyl halides, poly(vinyl acetate), and polyvinyl
- the polymer can be a biodegradable polymer, including a synthetic biodegradable polymer (e.g., polymers of lactic acid and glycolic acid, poly anhydrides, poly(ortho)esters, polyurethanes, poly(butic acid), poly(valeric acid), and poly(lactide-cocaprolactone)), and a natural biodegradable polymer (e.g., alginate and other polysaccharides including dextran and cellulose, collagen, chemical derivatives thereof (substitutions, additions of chemical groups, for example, alkyl, alkylene, hydroxylations, oxidations, and other modifications routinely made by those skilled in the art), albumin and other hydrophilic proteins (e.g., zein and other prolamines and hydrophobic proteins)), as well as any copolymer or mixture thereof.
- these materials degrade either by enzymatic hydrolysis or exposure to water in vivo, by surface or bulk erosion.
- the polymer can be a bioadhesive polymer, such as a bioerodible hydrogel described by H. S. Sawhney, C. P. Pathak and J. A. Hubbell in Macromolecules, 1993, 26, 581-587, the teachings of which are incorporated herein, polyhyaluronic acids, casein, gelatin, glutin, poly anhydrides, polyacrylic acid, alginate, chitosan, poly(methyl methacrylates), poly(ethyl methacrylates), poly(butylmethacrylate), poly(isobutyl methacrylate), poly(hexylmethacrylate), poly(isodecyl methacrylate), poly(lauryl methacrylate), poly(phenyl methacrylate), poly(methyl acrylate), poly(isopropyl acrylate), poly(isobutyl acrylate), and poly(octadecyl acrylate).
- a bioadhesive polymer such as
- the polymer is a water-soluble polymer.
- Suitable water-soluble polymers include, for example, polyvinylpyrrolidone, hydroxypropyl cellulose (HPC; Klucel), hydroxypropyl methylcellulose (HPMC; Methocel), nitrocellulose, hydroxypropyl ethylcellulose, hydroxypropyl butylcellulose, hydroxypropyl pentylcellulose, methyl cellulose, ethylcellulose (Ethocel), hydroxyethyl cellulose, various alkyl celluloses and hydroxyalkyl celluloses, various cellulose ethers, cellulose acetate, carboxymethyl cellulose, sodium carboxymethyl cellulose, calcium carboxymethyl cellulose, vinyl acetate/crotonic acid copolymers, poly-hydroxyalkyl methacrylate, hydroxymethyl methacrylate, methacrylic acid copolymers, polymethacrylic acid, polymethylmethacrylate, maleic anhydride/methyl vinyl ether
- the polymer is a polyalkylene glycol, including, for example, polyethylene glycol (PEG).
- PEG polyethylene glycol
- the carbohydrate may be any carbohydrate provided that it comprises or is made to comprise a carbonyl with an alpha leaving group.
- the carbohydrate for example, may be one which has been derivatized to comprise a carbonyl with an alpha leaving group.
- the carbohydrate may be a derivatized form of a monosaccharide (e.g., glucose, galactose, fructose), a disaccharide (e.g., sucrose, lactose, maltose), an oligosaccharide (e.g., raffinose, stachyose), a polysaccharide (a starch, amylase, amylopectin, cellulose, chitin, callose, laminarin, xylan, mannan, fucoidan, galactomannan.
- a monosaccharide e.g., glucose, galactose, fructose
- a disaccharide
- the lipid may be any lipid comprising a carbonyl with an alpha leaving group.
- the lipid for example, may be one which is derivatized to comprise the carbonyl.
- the lipid may be a derivative of a fatty acid (e.g., a C4-C30 fatty acid, eicosanoid, prostaglandin, leukotriene, thromboxane, N-acyl ethanolamine), glycerolipid (e.g., mono-, di-, tri-substituted glycerols), glycerophospholipid (e.g., phosphatidylcholine, phosphatidylinositol, phosphatidylethanolamine, phosphatidylserine), sphingolipid (e.g., sphingosine, ceramide), sterol lipid (e.g., steroid, cholesterol), prenol lipid, saccharolipid, or a polyketide. oil, wax, cholesterol, sterol, fat-soluble vitamin, monoglyceride, diglyceride, triglyceride, a phospholipid.
- a fatty acid
- R7 has a molecular weight of about 100 kDa or less, e.g., about 90 kDa or less, about 80 kDa or less, about 70 kDa or less, about 60 kDa or less, about 50 kDa or less, about 40 kDa or less. Accordingly, R7 can have a molecular weight of about 35 kDa or less, about 30 kDa or less, about 25 kDa or less, about 20 kDa or less, about 15 kDa or less, about 10 kDa or less, about 5 kDa or less, or about 1 kDa.
- the Class 4 peptide comprises as A, a peptide of 2 to 6 amino acids in which two consecutive amino acids of the peptide are linked via an ester or ether bond.
- the ester or ether bond may be, e.g., between amino acids 2 and 3, 3 and 4, 4 and 5, or 5 and 6.
- the peptide may be further modified by covalent linkage to another chemical moiety including linkage to a polymer (e.g. a hydrophilic polymer), alkylation, or acylation.
- B represents amino acids of native glucagon, e.g., i to 26 of SEQ ID NO: 901, wherein i is 3, 4, 5, 6, or 7, optionally comprising one or more amino acid modifications.
- B represents amino acids 7 to 26 of SEQ ID NO: 901, optionally further modified.
- B is modified by up to three amino acid modifications.
- B which represents native amino acid sequence of SEQ ID NO: 901 is modified by one or more conservative amino acid modifications.
- B comprises one or more amino acid modifications selected from the group consisting of (iv) to (ix), as described herein.
- B comprises one or both of the amino acid modifications (v) and (vi).
- B comprises one or a combination of amino acid modifications selected from the group consisting of (iv), (vii), (viii), and (ix), in addition to (v) and (vi).
- the Class 4 peptide comprises one or more charged amino acids at the C-terminus.
- Y and/or Z can be a charged amino acid, e.g., Lys, Arg, His, Asp, and GIu.
- the Class 4 peptide comprises one to two charged amino acids (e.g., Lys, Arg, His, Asp, and GIu) C-terminal to Z.
- Z followed by one to two charged amino acids does not comprise RlO.
- the Class 4 peptide in one embodiment comprises a hydrophilic moiety covalently bound to an amino acid residue of the Class 4 peptide, as described herein.
- the Class 4 peptide can comprise a hydrophilic moiety covalently attached to an amino acid at position 1, 16, 20, 21, or 24 according to the numbering of SEQ ID NO: 901.
- the hydrophilic moiety is attached to the C-terminal amino acid of the Class 4 peptide, which in some cases, is 1 or 11 amino acids C-terminal to Z.
- the hydrophilic moiety is attached to PLA, when A is PLA, PLA-Phe, or PLA- Thr-Phe, wherein PLA is modified to comprise the hydrophilic moiety.
- an amino acid comprising a hydrophilic moiety is added to the N- or C- terminus of the Class 4 peptide.
- the Class 4 peptide comprises an acyl group or alkyl group as described herein.
- the acylation or alkylation can occur off the side chain of the amino acid at position 10, 20, or 24, according to the numbering of SEQ ID NO: 901.
- the acylation or alkylation occurs off the side chain of the C-terminal amino acid of the Class 4 peptide, which in some cases, is 1 or 11 amino acids C-terminal to Z.
- A is PLA, PLA-Phe, or PLA-Thr-Phe
- the PLA is modified to comprise an acyl or alkyl group.
- the Class 4 peptide may comprise any amino acids, synthetic or naturally occurring, provided that at least two consecutive amino acids of the peptide are linked via an ester or ether bond.
- the peptide comprises amino acids of native glucagon.
- the peptide can comprise j to 6 of native glucagon (SEQ ID NO: 901), wherein j is 1, 2, 3, 4, or 5.
- the peptide can comprise an amino acid sequence based on the N-terminus of SEQ ID NO: 901 with one or more amino acid modifications.
- the amino acid at position 1 and/or position 2 may be an amino acid which reduces susceptibility to cleavage by dipeptidyl peptidase IV.
- the peptide can comprise at position 1 of the Class 4 peptide an amino acid selected from the group consisting of D-histidine, alpha, alpha-dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha-methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo- histidine.
- position 2 of the antagonist peptide is an amino acid selected from the group consisting of D-serine, D-alanine, valine, glycine, N- methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB).
- the amino acid at position 3 of the Class 4 peptide may be glutamic acid, as opposed to the native glutamine residue of native glucagon.
- the Class 4 peptide can comprise an amino acid sequence of:
- Xaa 1 -Xaa2-Xaa 3 -Thr-Gly-Phe SEQ ID NO: 968); Xaa 2 -Xaa 3 -Thr-Gly-Phe (SEQ ID NO: 969); or Xaa 3 -Thr-Gly-Phe (SEQ ID NO: 970); wherein Xaai is selected from a group consisting of: His, D-histidine, alpha, alpha- dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha- methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo- histidine; Xaa 2 is selected from a group consisting of: Ser, D-serine, D-alanine, valine, glycine, N-methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB); and X
- the present invention also encompasses embodiments wherein the C-terminal amino acid of the Class 4 peptides have an amide group substituting for the carboxylic acid group that is present on the native amino acid.
- the Class 4 peptide is PEGylated
- the Class 4 peptide comprises the shortened glucagon peptides, specifically 6-29 where the "N-terminal" amino acid is PLA (phenyl-lactic acid).
- PLA phenyl-lactic acid
- an amino acid of the Class 4 peptide is substituted with at least one cysteine residue, wherein the side chain of the cysteine residue is further modified with a thiol reactive reagent, including for example, maleimido, vinyl sulfone, 2-pyridylthio, haloalkyl, and haloacyl.
- thiol reactive reagents may contain carboxy, keto, hydroxyl, and ether groups as well as other hydrophilic moieties such as polyethylene glycol units.
- an amino acid of the Class 4 peptide is substituted with lysine, and the side chain of the substituting lysine residue is further modified using amine reactive reagents such as active esters (succinimido, anhydride, etc) of carboxylic acids or aldehydes of hydrophilic moieties such as polyethylene glycol.
- the lysine residue corresponding to position 12 of the native peptide is substituted with arginine and a single lysine substitution is inserted for one of the amino acids corresponding to position 1, 16, 17, 20, 21, 24 or 29 of the native peptide, or a lysine is added to the N- or C- terminus of the Class 4 peptide.
- the methionine residue corresponding to position 27 of the native peptide is changed to leucine or norleucine to prevent oxidative degradation of the peptide.
- the Class 4 peptides described herein are further modified by truncation or deletion of one or two amino acids of the C-terminus of the glucagon peptide (i.e., truncation of the amino acid at position 29 or at positions 28 and 29 of native glucagon) without affecting activity and/or potency at the glucagon receptor.
- the Class 4 peptide described herein can, for example, consist essentially of or consist of amino acids 1- 27, 1-28, 2-27, 2-28, 3-27, 3-28, 4-27, 4-28, 5-27, 5-28, 6-27, or 6-28 of the native glucagon peptide (SEQ ID NO: 901) with one or more modifications resulting in Class 4 peptideic activity as described herein.
- the presently disclosed Class 4 peptides also encompass amino acid substitutions at positions that are known not to be critical to the function of the glucagon peptide. In one embodiment the substitutions are conservative amino acid substitutions at one, two or three positions selected from the group consisting of 2, 5, 6, 7, 8, 9, 12, 13, 14, 15, 16, 19, 22, 23 or 24 of SEQ ID NO: 939.
- the Class 4 peptide comprises a derivative peptide of SEQ ID NO: 942 wherein the glucagon peptide comprises a further amino acid substitution relative to SEQ ID NO: 942 at one to three amino acid positions selected from positions 2, 5, 6, 8, 9, 12, 13 and 14.
- the substitutions at positions 2, 5, 6, 8, 9, 12, 13 and 14 of SEQ ID NO: 942 are conservative amino acid substitutions.
- the amino acids corresponding to positions 16, 17, 20, 21, 24 or 29 of the native peptide, and more particularly at position 21 and/or 24 are substituted with cysteine or lysine, wherein a PEG chain is covalently attached to the substituted cysteine or lysine residue.
- the modified Class 4 peptide comprises two or more polyethylene chains covalently bound to the peptide wherein the total molecular weight of the glucagon chains is about 1 ,000 to about 5,000 Daltons.
- the pegylated Class 4 peptide comprises a peptide selected from the group consisting of SEQ ID NO: 912, and SEQ ID NO: 922, wherein said peptide comprise a polyethylene glycol chain linked to the amino acid at positions 11 and 19 and the combined molecular weight of the two PEG chains is about 1,000 to about 5,000 Daltons.
- a Class 4 peptide is provided comprising a modified glucagon peptide selected from the group consisting of:
- Xaa at position 7 Lys or Arg
- Xaa at position 10 is aspartic acid, cysteic acid, glutamic acid, homoglutamic acid and homocysteic acid
- Xaa at position 11 is Ser, Lys, Cys, Orn, homocysteine or acetyl phenylalanine
- Xaa at position 16 is Asp, Lys, Cys, Orn, homocysteine or acetyl phenylalanine a
- Xaa at position 19 is GIn, Lys, Cys, Orn, homocysteine and acetyl phenylalanine
- Xaa at position 22 Met, Leu or NIe
- Ri is OH or NH 2
- R 2 is COOH or CONH 2 , wherein the peptide is pegylated at position 11 for SEQ ID NO: 909, at position 16 for SEQ ID NO: 910, position 19 for SEQ ID NO: 911 and at
- the peptide comprises the sequence of SEQ ID NO: 909, SEQ ID NO: 910 or SEQ ID NO: 911, wherein Ri is OH and R 2 is CONH 2 .
- the peptide comprises the sequence of SEQ ID NO: 909, SEQ ID NO: 910 or SEQ ID NO: 911, wherein Ri is OH, R 2 is CONH 2 and the amino acid at position 4 is aspartic acid, and in a further embodiment such peptides comprise a carboxy terminal extension comprising the sequence of SEQ ID NO: 919.
- the peptide comprises a sequence selected from the group consisting of SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 913, SEQ ID NO: 914, and SEQ ID NO: 916, wherein the peptide is pegylated at position 11 for SEQ ID NO: 909 and SEQ ID NO: 913, pegylated at position 16 for SEQ ID NO: 910, and pegylated at position 19 for SEQ ID NO: 910 and SEQ ID NO: 914.
- the glucagon agonist comprises the peptide of SEQ ID NO: 913 or SEQ ID NO: 914.
- the C-terminal amino acid of the Class 4 peptides disclosed herein have an amide group in place of the carboxylic acid group that is present on the native amino acid.
- the Class 4 peptide comprises the sequence of SEQ ID NO: 918.
- a Class 4 peptide wherein a plasma protein has been covalently linked to an amino acid side chain of the peptide to improve the solubility, stability and/or pharmacokinetics of the glucagon peptide.
- serum albumin can be covalently bound to the Class 4 peptides presented herein.
- the plasma protein is covalently bound to an amino acid corresponding to position 16, 17, 20, 21, 24 or 29 of the native glucagon peptide.
- the plasma protein is bound to an amino acid corresponding to position 16 or 24 of the native glucagon peptide
- the Class 4 peptide comprises the sequence of SEQ ID NO: 903, SEQ ID NO: 904, SEQ ID NO: 905, SEQ ID NO: 906, SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 909, SEQ ID NO: 911, SEQ ID NO: 912, SEQ ID NO: 922, SEQ ID NO: 923, SEQ ID NO: 924, SEQ ID NO: 925, SEQ ID NO: 926, SEQ ID NO: 927, SEQ ID NO: 928, SEQ ID NO: 936 and SEQ ID NO: 939.
- the Class 4 peptide comprises a peptide selected from the group consisting of SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911 and SEQ ID NO: 912.
- a Class 4 peptide wherein a linear amino acid sequence representing the Fc portion of an immunoglobin molecule has been covalently linked to an amino acid side chain of a Class 4 peptide disclosed herein to improve the solubility, stability and/or pharmacokinetics of the glucagon peptide.
- the amino acid sequence representing the Fc portion of an immunoglobin molecule can be covalently bound to position 11, 12, 15, 16, 19, 21 or 24 of the glucagon peptide of SEQ ID NO: 907, SEQ ID NO: 939, or a glucagon analog thereof.
- the Fc peptide is covalently bound to position 11 or 19 of the Class 4 peptide of SEQ ID NO: 906, SEQ ID NO: 907, SEQ ID NO: 908 or SEQ ID NO: 936.
- the Fc portion is usually isolated from IgG, but the Fc peptide fragment from any immunoglobin should function equivalently.
- the glucagon peptide is selected from the group consisting of SEQ ID NO: 903, SEQ ID NO: 904, SEQ ID NO: 905, SEQ ID NO: 907 SEQ ID NO: 908, and SEQ ID NO: 939, wherein the Fc portion is linked to the corresponding position of 16, 17, 20, 21, 24 or 29 of the native glucagon peptide.
- the Class 4 peptide comprises a glucagon peptide selected from the group consisting of SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911 and SEQ ID NO: 912, wherein the Fc peptide is bound to the side chain of the amino acid located at position 11, 16 or 19 of SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911, respectively, and at both positions 11 and 19 for SEQ ID NO: 912.
- the Class 4 peptide comprises the amino acid sequence of any of SEQ ID NOs: 962, 964-967, and 971.
- the Class 4 peptides can be further modified to improve the peptide's solubility in aqueous solutions at physiological pH, while, in some aspects retaining a glucagon antagonist activity.
- Introduction of hydrophilic groups at positions corresponding to positions 1, 16, 17, 20, 21, 24 and 29 of the native peptide, or at the C-terminus, can improve the solubility of the resulting Class 4 peptide in solutions having a physiological pH, while retaining the parent compounds antagonist activity.
- the presently disclosed Class 4 peptides are further modified to comprise one or more hydrophilic groups covalently linked to the side chains of amino acids corresponding to amino acid positions 1, 16, 17, 20, 21, 24 and 29 of the native glucagon peptide or of the N- or C-terminal amino acid.
- the side chains of amino acids corresponding to amino acid positions 16 and 24 of the native glucagon peptide are covalently bound to hydrophilic groups, and in one embodiment the hydrophilic group is polyethylene glycol (PEG).
- native glucagon can be modified by introducing charge at its carboxy terminus to enhance the solubility of the peptide while retaining the agonist properties of the peptide.
- the enhanced solubility allows for the preparation and storage of glucagon solutions at near neutral pH.
- Formulating glucagon solutions at relatively neutral pHs e.g. pH of about 6.0 to about 8.0 improves the long term stability of the Class 4 peptides.
- Class 4 peptides disclosed herein can be similarly modified to enhance their solubility in aqueous solutions at relatively neutral pH (e.g. pH of about 6.0 to about 8.0) while retaining the antagonist properties of the parent protein.
- one embodiment of the present invention is directed to a Class 4 peptide of SEQ ID NO: 939 that has been further modified relative to the native amino acids present at positions 6-29 of the wild type glucagon (SEQ ID NO: 901) to add charge to the peptide by the substitution of native non-charged amino acids with charged amino acids, or the addition of charged amino acids to the carboxy terminus.
- one to three of the non-charged native amino acids of the Class 4 peptide of SEQ ID NO: 939 are replaced with a charged amino acid.
- the charged amino acid is selected from the group consisting of lysine, arginine, histidine, aspartic acid and glutamic acid. More particularly, applicants have discovered that substituting the normally occurring amino acid at corresponding position 28 and/or 29 relative to native glucagon with charged amino acids, and/or the addition of one to two charged amino acids at the carboxy terminus of the Class 4 peptide, enhances the solubility and stability of the Class 4 peptides in aqueous solutions at physiologically relevant pHs (i.e., a pH of about 6.5 to about 7.5). Accordingly, such modifications of the Class 4 peptide disclosed herein are anticipated to have a similar effect on the solubility in aqueous solutions, particularly at a pH ranging from about 5.5 to about 8.0, while retaining the parent peptide's biological activity
- the Class 4 peptide of SEQ ID NO: 939 is modified by the substitution of the native amino acid at corresponding position 28 and/or 29 relative to native glucagon with a negatively charged amino acid (e.g., aspartic acid or glutamic acid) and optionally the addition of a negatively charged amino acid (e.g., aspartic acid or glutamic acid) to the carboxy terminus of the peptide.
- a negatively charged amino acid e.g., aspartic acid or glutamic acid
- a negatively charged amino acid e.g., aspartic acid or glutamic acid
- the Class 4 peptide of SEQ ID NO: 939 is modified by the substitution of the native amino acid at corresponding position 29 relative to native glucagon with a positively charged amino acid (e.g., lysine, arginine or histidine) and optionally the addition of one or two positively charged amino acid (e.g., lysine, arginine or histidine) on the carboxy terminus of the peptide.
- a positively charged amino acid e.g., lysine, arginine or histidine
- one or two positively charged amino acid e.g., lysine, arginine or histidine
- a Class 4 peptide having improved solubility and stability wherein the peptide comprises the amino acid sequence of SEQ ID NO: 941 with the proviso that at least one amino acids at position, 23, or 24 of SEQ ID NO: 941 is substituted with an acidic amino acid, and/or an additional acidic amino acid is added at the carboxy terminus of SEQ ID NO: 941.
- the acidic amino acids are independently selected from the group consisting of Asp, GIu, cysteic acid and homocysteic acid.
- a Class 4 peptide having improved solubility and stability wherein the antagonist comprises the amino acid sequence of SEQ ID NO: 941, SEQ ID NO: 942, SEQ ID NO: 943 or SEQ ID NO: 944, wherein at least one of the amino acids at positions 23 or 24 is substituted with a non-native amino acid residue (i.e. at least one amino acid present at position 23 or 24 of the analog is an acidic amino acid different from the amino acid present at the corresponding position in SEQ ID NO: 907).
- a glucagon agonist comprising the sequence of SEQ ID NO: 941 or 942 with the proviso that when the amino acid at position 23 is asparagine and the amino acid at position 24 is threonine, the peptide further comprises one to two amino acids, independently selected from the group consisting of Lys, Arg, His, Asp or GIu, added to the carboxy terminus of the Class 4 peptide.
- the solubility of the Class 4 peptide of SEQ ID NO: 942 can be improved by covalently linking a hydrophilic moiety to an amino acid residue at position 11, 12, 15, 16, 19 or 24, and in one embodiment the hydrophilic moiety is linked to an amino acid at position 11, 16 or 19, and in a further embodiment the hydrophilic moiety is linked to amino acid 19.
- the hydrophilic moiety is a plasma protein or the Fc portion of an immunoglobin, and in an alternative embodiment the hydrophilic moiety is a hydrophilic hydrocarbon chain.
- the hydrophilic moiety is polyethylene glycol, having a molecular weight selected from the range of about 1,000 to about 5,000 Dal tons.
- the hydrophilic moiety is polyethylene glycol, having a molecular weight of at least about 20,000 Daltons.
- polyethylene modified Class 4 peptide comprises the amino acids sequence of SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911, SEQ ID NO: 912, SEQ ID NO: 943, SEQ ID NO: 944 or SEQ ID NO: 945.
- the Asp-Ser sequence at position 15-16 of native glucagon has been identified as a uniquely unstable dipeptide that leads to premature chemical cleavage of the native hormone in aqueous buffers. For example, when maintained at 0.01N HCl at 37 0 C for 2 weeks, more than 50% of the native glucagon may be cleaved into fragments.
- the two liberated cleavage peptides 1-15 and 16-29 are devoid of glucagon- like biological activity and thus represent a limitation on the aqueous pre-formulation of glucagon and its related analogs.
- the selective chemical substitution of the Asp at position 15 of the native glucagon peptide with GIu has been observed to virtually eliminate chemical cleavage of the 15-16 peptide bond.
- the Class 4 peptides of the present invention can be similarly modified to decrease their susceptibility to premature chemical cleavage in aqueous buffers.
- the Class 4 peptides described herein can be further modified to enhance their stability in aqueous solutions by replacing the native aspartic amino acid, located at position 15 of the native glucagon peptide, with an amino acid selected from the group consisting of cysteic acid, glutamic acid, homoglutamic acid and homocysteic acid.
- the aspartic acid residue at position 10 of the Class 4 peptide of SEQ ID NO: 939 can be substituted with an amino acid selected from the group consisting of cysteic acid, glutamic acid, homoglutamic acid and homocysteic acid, and in one embodiment the native aspartic acid at position 10 of SEQ ID NO: 939 is replaced with glutamic acid.
- a Class 4 peptide having improved stability in aqueous solutions wherein the antagonist comprises a sequence selected from the group consisting of SEQ ID NO: 936, SEQ ID NO: 940 and SEQ ID NO: 942.
- the Class 4 peptide is amidated.
- increased stability by way of reduced degradation of the Class 4 peptide described herein may also be achieved by substitution of the serine at position 16 (according to the numbering of native glucagon) with glutamic acid, cysteic acid, homo-glutamic acid,or homo-cysteic acid.
- the serine at position 16 (according to the native glucagon sequence numbering) is replaced with glutamic acid.
- the Class 4 peptide comprising such a modification comprises a C-terminal carboxylate and is not amidated.
- a Class 4 peptide comprising a glucagon peptide selected from the group consisting of SEQ ID NO: 907, SEQ ID NO: 936, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941, SEQ ID NO: 942, SEQ ID NO: 943 and SEQ ID NO: 944, further modified by one or more additional amino acid substitutions at positions corresponding to positions 11, 12, 15, 16, 19 and/or 24 of the native glucagon peptide, wherein the amino acid substitutions comprise a substitution with an amino acid having a side chain suitable for crosslinking with hydrophilic moieties, including for example, PEG.
- the peptide can be substituted with a naturally occurring amino acid or a synthetic (non-naturally occurring) amino acid.
- Synthetic or non-naturally occurring amino acids refer to amino acids that do not naturally occur in vivo but which, nevertheless, can be incorporated into the peptide structures described herein.
- a Class 4 peptide is provided wherein the peptide comprises the sequence of SEQ ID NO: 907, SEQ ID NO: 936, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941, SEQ ID NO: 942, SEQ ID NO: 943 and SEQ ID NO: 944, and further comprises a polyethylene chain bound to corresponding position 21 or 24 of the native glucagon peptide.
- the C-terminus of the Class 4 peptide is modified to replace the carboxylic acid group with an amide group.
- Fusion peptides and conjugates also encompasses Class 4 peptide fusion peptides wherein a second peptide has been fused to the C-terminus of the Class 4 peptide.
- the fusion peptide may comprise a Class 4 peptide peptide of SEQ ID NO: 944 that further comprises an amino acid sequence of SEQ ID NO: 919 (GPSSGAPPPS), SEQ ID NO: 920 (Lys Arg Asn Arg Asn Asn lie Ala ) or SEQ ID NO: 921 (Lys Arg Asn Arg) linked to the c- terminal amino acid of the Class 4 peptide.
- the amino acid sequence of SEQ ID NO: 919 is bound to amino acid 24 of the Class 4 peptide of SEQ ID NO: 942 through a peptide bond.
- the fusion peptide comprises a Class 4 peptide peptide of SEQ ID NO: 907, SEQ ID NO: 936, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941 or SEQ ID NO: 943 that further comprises an amino acid sequence of SEQ ID NO: 919 (GPSSGAPPPS) linked to amino acid 24 of the Class 4 peptide.
- the fusion peptide comprises a Class 4 peptide peptide of SEQ ID NO: 907, SEQ ID NO: 936, SEQ ID NO: 937, SEQ ID NO: 938, SEQ ID NO: 939, SEQ ID NO: 941 or SEQ ID NO: 943 that further comprises an amino acid sequence of SEQ ID NO: 920, SEQ ID NO: 921 or SEQ ID NO: 953 linked to amino acid 24 of the Class 4 peptide.
- the Class 4 peptide fusion peptide comprises a sequence selected from the group consisting of SEQ ID NO: 946 and SEQ ID NO: 947.
- the C-terminus of the fusion peptide is modified to replace the carboxylic acid group with an amide group.
- a Class 4 peptide fusion peptide is provided wherein the Class 4 peptide portion of the fusion peptide is selected from the group consisting of SEQ ID NO: 903, SEQ ID NO: 904, SEQ ID NO: 905, SEQ ID NO: 906, SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 909, SEQ ID NO: 911, SEQ ID NO: 912, SEQ ID NO: 913, SEQ ID NO: 914, SEQ ID NO: 915, SEQ ID NO: 910, SEQ ID NO: 916, SEQ ID NO: 917, SEQ ID NO: 918 and SEQ ID NO: 939 and the sequence of SEQ ID NO: 919 is fused to the carboxy terminus of the Class 4 peptide portion, and wherein the PEG chain, when present, is selected from the range of 500 to 40,000 Daltons
- the Class 4 peptide segment is selected from the group consisting of SEQ ID NO: 913, SEQ ID NO: 914, SEQ ID NO: 915, SEQ ID NO: 916, SEQ ID NO: 946 and SEQ ID NO: 947 wherein the PEG chain is selected from the range of about 500 to about 5,000 Daltons, and more particularly, in one embodiment the PEG chain is about 1,000 Daltons.
- the C-terminus is modified to replace the carboxylic acid group with an amide group.
- the Class 4 peptide may further comprise one to two charged amino acids added to the carboxy terminus.
- the amino acids are negatively charged amino acids, including for example glutamic acid and aspartic acid.
- the Class 4 peptide comprises the sequence of SEQ ID NO: 942 wherein at least one of corresponding positions 27 and 28 relative to the native glucagon peptide comprises an amino acid selected from the group consisting of aspartic acid and glutamic acid and wherein SEQ ID NO: 942 is optionally modified to include an addition one to two negatively charged amino acids added to the carboxy terminus.
- the negatively charged amino acids are glutamic acid or aspartic acid.
- the Class 4 peptides disclosed herein can be combined with other active agents, including for example, insulin, to treat diseases or conditions that are characterized by excessive glucagon activity.
- Class 4 peptides that have been modified to be covalently bound to a PEG chain having a molecular weight of greater than 10,000 Daltons can be administered in conjunction with insulin to help to maintain stable blood glucose levels in diabetics.
- the Class 4 peptides of the present disclosure can be coadministered with insulin as a single composition, simultaneously administered as separate solutions, or alternatively, the insulin and the Class 4 peptide can be administered at different times relative to one another.
- the composition comprising insulin and the composition comprising the Class 4 peptide are administered within 12 hours of one another. The exact ratio of the Class 4 peptide relative to the administered insulin will be dependent in part on determining the glucagon levels of the patient, and can be determined through routine experimentation.
- Dimer peptides The present disclosure also encompasses multimers of the modified Class 4 peptides disclosed herein. Two or more of the modified Class 4 peptides can be linked together using standard linking agents and procedures known to those skilled in the art. For example, dimers can be formed between two modified Class 4 peptides through the use of bifunctional thiol crosslinkers and bi-functional amine crosslinkers, particularly for Class 4 peptides that have been substituted (at positions 11, 16 or 19, for example) with cysteine, lysine ornithine, homocysteine or acetyl phenylalanine residues (e.g. SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911 and SEQ ID NO: 912).
- bifunctional thiol crosslinkers and bi-functional amine crosslinkers particularly for Class 4 peptides that have been substituted (at positions 11, 16 or 19, for example) with cysteine, lysine ornithine, homocysteine
- the dimer can be a homodimer or alternatively can be a heterodimer.
- the dimer is formed between two Class 4 peptides independently selected from the group consisting of SEQ ID NO: 908, SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911, SEQ ID NO: 912, SEQ ID NO: 945, SEQ ID NO: 946, or SEQ ID NO: 947, wherein the two peptides are linked to one another via a linker attached to position 11 of each peptide, 16 of each peptide, or position 19 of each peptide or any combination thereof.
- the linkage is a disulfide linkage between a Cysl 1 to Cysl lor a Cysl9 to Cysl9 or a Cysl 1 to Cysl9 residue of the respective Class 4 peptide peptides.
- a dimer can be formed between two Class 4 peptide peptides independently selected form the group consisting of SEQ ID NO: 903, SEQ ID NO: 904, SEQ ID NO: 905, SEQ ID NO: 906, SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 909, SEQ ID NO: 910, SEQ ID NO: 911, SEQ ID NO: 912, SEQ ID NO: 936, SEQ ID NO: 937, SEQ ID NO: 938, SEQ ID NO: 939 and SEQ ID NO: 942 wherein the linkage is formed between amino acid positions independently selected from positions 16, 21 and 24 with respect to the native glucagon peptide.
- a Class 4 peptide dimer comprising two Class 4 peptides, each comprising the sequence of SEQ ID NO: 946, wherein the two antagonists are linked to one another by a disulfide bond through amino acid position 25.
- a Class 4 peptide dimer is provided comprising two Class 4 peptides, each comprising the sequence of SEQ ID NO: 947, wherein the two antagonists are linked to one another by a disulfide bond through amino acid position 35.
- the dimer is formed from Class 4 peptides of SEQ ID NO: 946 and SEQ ID NO: 947 wherein the amino acid at position 10 is glutamic acid.
- the dimer comprises a homodimer of a Class 4 peptide fusion peptide selected from the group consisting of SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 936, SEQ ID NO: 937, SEQID NO: 940, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941, SEQ ID NO: 942 and pharmaceutically acceptable salts of said Class 4 peptides.
- a dimer comprising a first Class 4 peptide bound to a second Class 4 peptide via a linker, wherein the first and second peptides of the dimer are independently selected from the group consisting of SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 936, SEQ ID NO: 937, SEQID NO: 940, SEQ ID NO: 939, SEQ ID NO: 940, SEQ ID NO: 941, SEQ ID NO: 942 and pharmaceutically acceptable salts of said Class 4 peptides
- the first and second Class 4 peptides of the dimer are independently selected from the group consisting of SEQ ID NO: 907, SEQ ID NO: 908, SEQ ID NO: 936 and SEQ ID NO: 939.
- the dimer comprises a homodimer of a Class 4 peptide selected from the group consisting of SEQ ID NO: 923, SEQ ID NO: 924, SEQ ID NO: 925, SEQ ID NO: 926, SEQ ID NO: 927, SEQ ID NO: 928, SEQ ID NO: 929, SEQ ID NO: 930, SEQ ID NO: 931.
- a Class 4 peptide dimer is provided wherein the first and second peptides of the dimer comprise an amino acid sequence independently selected from the group consisting of SEQ ID NO: 923, SEQ ID NO: 924, SEQ ID NO: 925, SEQ ID NO: 926, SEQ ID NO: 927 and SEQ ID NO: 928.
- the dimer comprises a homodimer of a Class 4 peptide selected from the group consisting of SEQ ID NO: 909, SEQ ID NO: 911 and SEQ ID NO: 912, wherein the peptide further comprises a polyethylene glycol chain covalently bound to position 11 or 19 of the glucagon peptide.
- the Class 4 glucagon related analog peptide may comprise the amino acid sequence of any of the amino acid sequences of SEQ ID NOs: 901-971, optionally with up to 1, 2, 3, 4, or 5 further modifications that retain glucagon antagonist activity.
- a glucagon related analog peptide is a class 5 glucagon related analog peptide ⁇ see, e.g., International (PCT) Patent Application No. PCT/US2008/081333, incorporated herein by reference in its entirety).
- a class 5 glucagon related analog peptide may be a glucagon antagonists/GLP-1 agonists are utilized in any setting where the suppression of glucagon agonism is desired while simultaneous stimulation of GLP-I activity is also desired.
- glucagon antagonist activity in conjunction with GLP-I stimulation can be used in the treatment of diabetes where glucagon antagonism has been demonstrated in pre-clinical models of hyperglycemia to yield a lowering of blood glucose and GLP-I activity is associated with insulin production.
- Compounds demonstrating GLP- 1 activity have also been known to be useful for treating obesity and preventing weight gain.
- class 5 peptides are believed to be suitable for any use that has previously been described for other glucagon antagonist/GLP- 1 agonists. These two activities have separately been shown to be highly desirable properties for the treatment of the metabolic syndrome, specifically diabetes and obesity.
- the glucagon antagonist activity is useful in any setting where the suppression of glucagon agonism is desired.
- the presence of GLP-I agonism further suppresses the endogenous secretion of glucagon from the pancreas while stimulating insulin synthesis and secretion.
- the two pharmacological actions serve in a synergistic fashion to normalize metabolic abnormalities.
- the Class 5 peptides can be used to treat hyperglycemia, or treat other metabolic diseases that result from high blood levels of glucagon or high blood glucose levels.
- the patient to be treated using the glucagon antagonist/GLP- 1 agonists such as class 5 peptides disclosed herein is a domesticated animal, and in another embodiment the patient to be treated is a human. Studies suggest that lack of glucagon suppression in diabetic patients contributes to postprandial hyperglycemia in part via accelerated glycogenosis.
- a glucagon antagonist/GLP- 1 agonists or Class 5 peptides described herein can be used to treating hyperglycemia, and are expected to be useful for treating a variety of types of diabetes including diabetes mellitus type I, diabetes mellitus type II, or gestational diabetes, either insulin-dependent or non-insulin-dependent, and reducing complications of diabetes including nephropathy, retinopathy and vascular disease.
- Such methods for reducing appetite or promoting loss of body weight are expected to be useful in reducing body weight, preventing weight gain, or treating obesity of various causes, including drug-induced obesity, and reducing complications associated with obesity including vascular disease (coronary artery disease, stroke, peripheral vascular disease, ischemia reperfusion, etc.), hypertension, onset of diabetes type II, hyperlipidemia and musculoskeletal diseases.
- vascular disease coronary artery disease, stroke, peripheral vascular disease, ischemia reperfusion, etc.
- hypertension onset of diabetes type II
- hyperlipidemia hyperlipidemia and musculoskeletal diseases.
- compositions comprising class 5 peptides can be formulated and administered to patients using standard pharmaeuctically acceptable carriers and routes of administration known to those skilled in the art. Accordingly, the present disclosure also encompasses pharmaceutical compositions comprising one or more class 5 peptides disclosed herein in combination with a pharmaceutically acceptable carrier.
- the pharmaceutical compositions may comprise the class 5 peptides as the sole pharmaceutically active component, or the class 5 peptides can be combined with one or more additional active agents.
- a composition is provided comprising a Class 5 peptide and insulin or an insulin analog.
- compositions for inducing weight loss or preventing weight gain can be provided that comprises the sequence of SEQ ID NO: 1015 or SEQ ID NO: 1051 further comprising the amino acid sequence of SEQ ID NO: 1021 (GPSSGAPPPS) or SEQ ID NO: 1050 linked to amino acid 24 of SEQ ID NO: 1015 or SEQ ID NO: 1051, and an anti-obesity peptide.
- Suitable anti-obesity peptides include those disclosed in US patents 5,691,309, 6,436,435 or US Patent application 20050176643.
- a Class 5 peptide comprising a glucagon peptide that has been modified by the deletion of the first 1 to 5 amino acids residues ⁇ e.g., first amino acid, first two amino acids, first three amino acids, first four amino acids, first five amino acids) from the N-terminus, and stabilization of the alpha-helix structure in the C-terminal portion of the compound (around amino acid positions 12-29 according to the amino acid numbering of wild type glucagon, SEQ ID NO: 1001), e.g., by the linkage of the side chains of amino acid pairs, selected from positions 12 and 16, 16 and 20, 20 and 24, and 24 and 28 (relative to the native glucagon peptide sequence), to one another through hydrogen-bonding or ionic interactions, such as the formation of salt bridges, or by covalent bonds.
- first 1 to 5 amino acids residues e.g., first amino acid, first two amino acids, first three amino acids, first four amino acids, first five amino acids
- stabilization of the alpha-helix around residues 12-29 is achieved through introduction of one or more ⁇ , ⁇ -disubstituted amino acids at positions that retain the desired activity.
- one, two, three, four or more of positions 16, 17, 18, 19, 20, 21, 24 or 29 (according to the amino acid numbering of wild type glucagon) of the Class 5 peptide or analog thereof is substituted with an ⁇ , ⁇ -disubstituted amino acid.
- substitution of position 16 (according to the amino acid numbering of wild type glucagon) of a Class 5 peptide or analog thereof with amino iso-butyric acid (AIB) provides a stabilized alpha helix in the absence of a salt bridge or lactam.
- one, two, three or more of positions 16, 20, 21 or 24 (according to the amino acid numbering of wild type glucagon) are substituted with AIB
- a class 5 peptide wherein the peptide exhibits at least 80% of the maximum agonism achieved by native GLP-I at the GLP-I receptor, and exhibits glucagon antagonist activity that reduces the maximum glucagon- induced cAMP production at the glucagon receptor by at least about 50%, as measured by cAMP production in an in vitro assay.
- the class 5 peptide exhibits at least 90% of the activity of native GLP-I at the GLP-I receptor, and exhibits glucagon antagonist activity, that reduces the maximum glucagon-induced cAMP production at the glucagon receptor by at least about 80%.
- the class 5 peptide comprises a derivative peptide of SEQ ID NO: 1002 wherein the peptide comprises further amino acid substitutions relative to SEQ ID NO: 1002 at one to three amino acid positions selected from positions 1, 2, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 19, 22 and 24, and exhibits at least 90% of the activity of native GLP-I at the GLP-I receptor, and exhibits glucagon antagonist activity, that reduces the maximum glucagon-induced cAMP production at the glucagon receptor by at least about 80%.
- the alpha-helix structure in the C-terminal portion of the Class 5 peptide (around amino acids 12-29 according to the amino acid numbering of wild type glucagon) is stabilized by, e.g., formation of a covalent or non-covalent intramolecular bridge, or substitution and/or insertion of amino acids around positions 12-29 with an alpha helix- stabilizing amino acid (e.g., an ⁇ , ⁇ -disubstituted amino acid).
- an alpha helix- stabilizing amino acid e.g., an ⁇ , ⁇ -disubstituted amino acid
- one, two, three, four or more of positions 16, 17, 18, 19, 20, 21, 24 or 29 (according to the amino acid numbering of wild type glucagon) of the Class 5 peptide or analog thereof is substituted with an ⁇ , ⁇ -disubstituted amino acid e.g., amino isobutyric acid (AIB).
- amino isobutyric acid amino isobutyric acid
- substitution of position 16 (according to the amino acid numbering of wild type glucagon) of a Class 5 peptide or analog thereof with amino iso-butyric acid (AIB) provides a stabilized alpha helix in the absence of a salt bridge or lactam.
- the class 5 peptide comprises SEQ ID NO: 1015 or SEQ ID NO: 1051, and more particularly, a sequence selected from the group consisting of SEQ ID NO: 1005, SEQ ID NO: 1006, SEQ ID NO: 1007, SEQ ID NO: 1008, SEQ ID NO: 1009, SEQ ID NO: 1016, SEQ ID NO: 1017, SEQ ID NO: 1018, SEQ ID NO: 1019, SEQ ID NO: 1022, SEQ ID NO: 1023, SEQ ID NO: 1024 and SEQ ID NO: 1025.
- the class 5 peptide comprises a derivative peptide of SEQ ID NO: 1015 or SEQ ID NO: 1051 wherein the peptide comprises a further amino acid substitution relative to SEQ ID NO: 1015 or SEQ ID NO: 1051 at one to three amino acid positions selected from positions 1, 2, 5, 6, 8, 9, 12, 13 and 14.
- the substitutions at positions 1, 2, 5, 6, 8, 9, 12, 13 and 14 are conservative amino acid substitutions.
- the threonine at position 24 of SEQ ID NO: 1005 or SEQ ID NO: 1006 is substituted with glycine.
- the class 5 peptide represents a further modification of the peptide wherein in addition to the N-terminal deletion, the phenylalanine at position 6 of the native glucagon peptide is modified, e.g., to comprise a hydroxyl group in place of the N-terminus amino group.
- the natural carboxylic acid of the C-terminal amino acid is replaced with a charge-neutral group, such as an amide or ester.
- Class 5 peptides have been prepared wherein the first three to five amino acids of native glucagon have been deleted, the amino acid at position 9, relative to the native glucagon peptide, has been substituted with an amino acid selected from the group consisting of glutamic acid, homoglutamic acid, ⁇ -homoglutamic acid, a sulfonic acid derivative of cysteine, or an alkylcarboxylate derivative of cysteine having the structure of: OOH , wherein X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl, and the alpha-helix structure in the C-terminal portion of glucagon (around amino acids 12-29 according to the amino acid numbering of wild type glucagon) is stabilized, e.g., via a lactam bridge is formed between the side chains of amino acids 12 and 16 or between amino acids 16 and 20, relative to the native glucagon peptide.
- the sulfonic acid derivative of cysteine is cysteic acid or homocysteic acid.
- a class 5 is provided comprising an amino acid sequence selected from the group consisting of SEQ ID NO: 1005, SEQ ID NO: SEQ ID NO: 1006, SEQ ID NO: 1007, or SEQ ID NO: 1008, wherein said peptide comprises a lactam ring formed between the side chains of amino acids 7 and 11 for SEQ ID NO: 1005, between 11 and 15 for SEQ ID NO: 1006, between positions 15 and 19 for SEQ ID NO: 1007 and between positions 19 and 24 for SEQ ID NO: 1008, each of said sequences being further modified to comprise a hydrophilic moiety covalently bound to the peptide.
- each of the lactam bearing class 5 peptide are modified by covalent attachment of a polyethylene glycol chain.
- a polyethylene glycol chain For example, for a class 5 peptide comprising SEQ ID NO: 1005, the peptide is pegylated at a position selected from the group consisting of 12, 15, 16, 19 and 24; for a class 5 peptide comprising SEQ ID NO: 1006, the peptide is pegylated at a position selected from the group consisting of 12, 16, 19 and 24; for a class 5 peptide comprising SEQ ID NO: 1007, the peptide is pegylated at a position selected from the group consisting of 11, 12, 16 and 24; for class 5 peptide comprising SEQ ID NO: 1008, the peptide is pegylated at a position selected from the group consisting of 11, 12, 15 and 16.
- a class 5 peptide comprising SEQ ID NO: 1047 or SEQ ID NO: 1048 wherein the petide is pegylated at a position selected from the group consisting of 12, 16, 19 and 24, relative to the SEQ ID NO: 1047 or SEQ ID NO: 1048 sequence.
- the peptide of SEQ ID NO: 1047 or SEQ ID NO: 1048 is further modified by the addition of the sequence of SEQ ID NO: 1021 to the carboxy terminus of the peptide.
- Class 5 peptides are provided wherein the first five amino acids of native glucagon have been deleted, the amino group of the N-terminal amino acid (phenylalanine) has been replaced with a hydroxyl group (i.e., the first amino acid is phenyl-lactic acid) and the side chains of one or more amino acid pairs selected from positions 12 and 16, 16 and 20, 20 and 24, and 24 and 28 are linked to one another, thus stabilizing the Class 5 peptide alpha helix.
- the amino group of the N-terminal amino acid phenylalanine
- the side chains of one or more amino acid pairs selected from positions 12 and 16, 16 and 20, 20 and 24, and 24 and 28 are linked to one another, thus stabilizing the Class 5 peptide alpha helix.
- a class 5 peptide comprising the sequence of SEQ ID NO: 1002 that is modified by a substitution of the serine residue at position 11 of SEQ ID NO: 1002 (position 16 according to the amino acid numbering of native glucagon) with an amino acid selected from the group consisting of glutamic acid, glutamine, homoglutamic acid, homocysteic acid, threonine or glycine.
- the serine residue at position 11 of SEQ ID NO: 1002 is substituted with an amino acid selected from the group consisting of glutamic acid, glutamine, homoglutamic acid and homocysteic acid, and in one embodiment the serine residue is substituted with glutamic acid.
- the class 5 peptide comprises the sequence of SEQ ID NO: 1038.
- a class 5 peptide wherein an intramolecular bridge is formed between two amino acid side chains to stabilize the three dimensional structure of the carboxy terminus of the peptide of SEQ ID NO: 1002. More particularly, the side chains of one or more amino acids selected from amino acid pairs 7 and 11, 11 and 15, 15 and 19 or 19 and 23 of SEQ ID NO: 1002 are linked to one another, thus stabilizing the alpha helix in the C-terminal portion.
- the two side chains can be linked to one another through hydrogen- bonding, ionic interactions (such as the formation of salt bridges), or by covalent bonds.
- the size of the linker is 7-9 atoms, and in one embodiment the size of the linker is 8 atoms.
- the class 5 peptide is selected from the group consisting of SEQ ID NO: 1005, SEQ ID NO: 1006, SEQ ID NO: 1007 and SEQ ID NO: 1008.
- the C-terminal amino acid of the class 5 peptide have an amide group substituting for the carboxylic acid group that is present on the native amino acid.
- class 5 peptide wherein the analog comprises an amino acid sequence of SEQ ID NO: 1009.
- the three dimensional structure of the carboxy terminus of the peptide of SEQ ID NO: 1009 is stabilized by the formation of covalent bonds between the side chains of the peptide.
- two amino acid side chains are bound to one another to form a lactam ring.
- the size of the lactam ring can vary depending on the length of the amino acid side chains, and in one embodiment the lactam is formed by linking the side chains of a lysine amino acid to a glutamic acid side chain.
- the C-terminal amino acid of the class 5 peptides have an amide group substituting for the carboxylic acid group that is present on the native amino acid.
- a lactam ring can be formed between the side chains of a Lysl2 and a GIu 16 or alternatively between a GIu 12 and a Lysl ⁇ ).
- a glucagon analog of SEQ ID NO: 1009 is provided wherein at least one lactam ring is formed between the side chains of an amino acid pair selected from the group consisting of amino acid pairs 7 and 11, 11 and 15 , 15 and 19 or 19 and 23 of SEQ ID NO: 1009.
- a class 5 peptide wherein the peptide comprises the sequence of SEQ ID NO: 1010, said sequence further comprising an intramolecular lactam bridge formed between amino acid positions 7 and 11 , or between amino acid positions 11 and 15, or between amino acid positions 15 and 19 of SEQ ID NO: 1010.
- a class 5 peptide is provided wherein the peptide comprises the sequence of SEQ ID NO: 1011, said sequence further comprising an intramolecular lactam bridge formed between amino acid positions 7 and 11, or between amino acid positions 11 and 15 of SEQ ID NO: 1011.
- the class 5 peptide comprises the sequence of SEQ ID NO: 17.
- Additional class 5 peptide comprising derivatives of SEQ ID NO: 1005, wherein the aspartic acid at position 10 of SEQ ID NO: 1005 (position 15 of native glucagon) has been substituted with glutamic acid, an amino acid of the general structure:
- X 6 is C 1 -C 3 alkyl, C 2 -C 3 alkene or C 2 -C 3 alkynyl, and in one embodiment X 6 is C 1 -C 3 alkyl, and in another embodiment X 6 is C 2 alkyl.
- a Class 5 peptide derivative of SEQ ID NO: 1009 is provided wherein position 10 of SEQ ID NO: 1009 (position 15 of native glucagon) is substituted with an amino acid selected from the group consisting of glutamic acid, cysteic acid, homocysteic acid and homoglutamic acid. In a further embodiment position 10 of SEQ ID NO: 1009 is substituted with an amino acid selected from the group consisting of cysteic acid or homocysteic acid.
- a Class 5 peptide derivative of SEQ ID NO: 1006, SEQ ID NO: 1007 or SEQ ID NO: 1008 is provided wherein position 10 of SEQ ID NO: 1006, SEQ ID NO: 1007 or SEQ ID NO: 1008 is substituted with an amino acid selected from the group consisting of glutamic acid, cysteic acid, homocysteic acid and homoglutamic acid.
- an amino acid selected from the group consisting of glutamic acid, cysteic acid, homocysteic acid and homoglutamic acid.
- the C-terminal amino acid of a class 5 peptide have an amide group substituting for the carboxylic acid group that is present on the native amino acid.
- an amino acid of class 5 peptide is substituted with at least one cysteine residue, wherein the side chain of the cysteine residue is further modified with a thiol reactive reagent, including for example, maleimido, vinyl sulfone, 2-pyridylthio, haloalkyl, and haloacyl.
- thiol reactive reagents may contain carboxy, keto, hydroxyl, and ether groups as well as other hydrophilic moieties such as polyethylene glycol units.
- an amino acid of a class 5 peptide is substituted with lysine, and the side chain of the substituting lysine residue is further modified using amine reactive reagents such as active esters (succinimido, anhydride, etc) of carboxylic acids or aldehydes of hydrophilic moieties such as polyethylene glycol.
- amine reactive reagents such as active esters (succinimido, anhydride, etc) of carboxylic acids or aldehydes of hydrophilic moieties such as polyethylene glycol.
- the lysine residue corresponding to position 7 of the peptide of SEQ ID NO: 1005 is substituted with arginine and a single lysine substitution is inserted for one of the amino acids corresponding to position 12, 15, 16, 19 and 24 of SEQ ID NO: 1005.
- the methionine residue corresponding to position 22 of the class 5 peptides disclosed herein is changed to leucine or norleucine to prevent oxidative degradation of the peptide.
- class 5 peptides in some aspects, also encompass amino acid substitutions at positions that are known not to be critical to the function of the glucagon analog. In one embodiment the substitutions are conservative amino acid substitutions at one, two or three positions selected from the group consisting of 2, 5, 6, 7, 8, 9, 12, 13, 14, 15, 16, 19, 22, 23 or 24.
- amino acids corresponding to positions 16, 17, 20, 21, 24 or 29 of the native glucagon peptide, and more particularly at position 21 and/or 24 relative to native glucagon are substituted with cysteine or lysine, wherein a PEG chain is covalently attached to the substituted cysteine or lysine residue.
- a class 5 peptide is provided comprising a sequence consisting of SEQ ID NO: 1009, further modified by one or more additional amino acid substitutions at positions corresponding to positions 11, 12, 15, 16, 19 and/or 24 of the peptide (including for example substitution with cysteine), wherein the amino acid substitution comprises an amino acid having a side chain suitable for crosslinking with hydrophilic moieties, including for example, PEG.
- Native glucagon can be substituted with a naturally occurring amino acid or a synthetic (non-naturally occurring) amino acid.
- Synthetic or non-naturally occurring amino acids refer to amino acids that do not naturally occur in vivo but which, nevertheless, can be incorporated into the peptide structures described herein.
- a Class 5 peptide is provided wherein the peptide comprises the sequence of SEQ ID NO: 1009 and further comprises a polyethylene chain bound to position 16 or 19 of the peptide.
- the C-terminus of the glucagon analog is modified to replace the carboxylic acid group with an amide group.
- a class 5 peptide comprising a glucagon analog selected from the group consisting of:
- Xaa at position 4 aspartic acid, glutamic acid, cysteic acid or homocysteic acid
- Xaa at position 10 Asp, GIu, cysteic acid, homoglutamic acid and homocysteic acid
- Xaa at position 16 is Asp
- the Xaa at position 19 is GIn, Cys, Orn, homocysteine and acetyl phenylalanine
- Xaa at position 22 Met, Leu or NIe
- R 1 is OH or
- the Thr at position 24 of SEQ ID NOs: 1012-1014 and 1039 is substituted with GIy.
- the peptide comprises the sequence of SEQ ID NO: 1013 or SEQ ID NO: 1014, wherein Ri is OH.
- the peptide comprises the sequence of SEQ ID NO: 1013 or SEQ ID NO: 1014, wherein R 1 is OH and R 2 is CONH 2 .
- the peptide comprises the sequence of SEQ ID NO: 1013 or SEQ ID NO: 1014, wherein R 1 is OH, R 2 is CONH 2 and the threonine at position 24 is substituted with glycine.
- a class 5 peptide is further modified to comprise one or more amino acids of native GLP-I by substitution of the native glucagon residue(s) at corresponding amino acid positions.
- the class 5 peptide may comprise one or more amino acid substitutions at any of positions 2, 3, 17, 18, 21, 23, and 24 (according to the amino acid numbering of native glucagon).
- the class 5 peptide is modified by one or more of the following amino acid substitutions: Ser2 is replaced with Ala, Gln3 is replaced with GIu, Argl7 is replaced with GIn, Arg at position 18 is replaced with Ala, Asp at position 21 is replaced with GIu, VaI at position 23 is replaced with lie, and GIn at position 24 is replaced with Ala (amino acid positions are in accordance with the native glucagon sequence).
- the class 5 peptide is modified by replacing Ser2 with Ala and Gln3 with GIu (according to the amino acid numbering of native glucagon).
- the class 5 peptide is modified with all of the following amino acid substitutions: Arg 17 is replaced with GIn, Arg at position 18 is replaced with Ala, Asp at position 21 is replaced with GIu, VaI at position 23 is replaced with lie, and GIn at position 24 is replaced with Ala (amino acid numbering according to native glucagon).
- the class 5 peptide is modified to comprise just GIu at position 21 (according to the numbering of SEQ ID NO: 1001). Accordingly, the class 5 peptide can comprise the amino acid sequence of any of SEQ ID NOs: 1060-1070, 1073- 1078, 1080-1088, 1090-1096, 1103, 1104, 1106, and 1114-1118.
- a class 5 peptide or conjugate thereof comprising (1) a stabilized alpha helix through means described herein (e.g., through an intramolecular bridge, or incorporation of one or more alpha, alpha-di-substituted amino acids, or an acidic amino acid at position 16 (according to the numbering of SEQ ID NO : 1001), or a combination thereof; (2) a C-terminal amide or ester in place of a C-terminal carboxylate, and (3) a general structure of A-B-C, wherein A is selected from the group consisting of (i) phenyl lactic acid (PLA); (ii) an oxy derivative of PLA; and
- the one or more modification may be selected from the group consisting of:
- Asp at position 9 (according to the amino acid numbering of SEQ ID NO: 1001) is substituted with a GIu, a sulfonic acid derivative of Cys, homoglutamic acid, ⁇ -homoglutamic acid, or an alkylcarboxylate derivative of cysteine having the structure of:
- Arg at position 17 is replaced with GIn
- Arg at position 18 is replaced with Ala
- Asp at position 21 is replaced with GIu
- VaI at position 23 is replaced with He
- GIn at position 24 is replaced with Ala (according to amino acid numbering of SEQ ID NO: 1001 );
- Ser at position 16 is replaced with GIu
- GIn at position 20 is replaced with GIu
- GIn at position 24 is replaced with Glu(according to the amino acid numbering of SEQ ID NO: 1001); wherein C (of the general structure of A-B-C) is selected from the group consisting of:
- (x) X-Y-Z-RlO wherein X is Met, Leu, or NIe; Y is Asn or a charged amino acid; Z is Thr, GIy, Cys, Lys, ornithine (Orn), homocysteine, acetyl phenylalanine (Ac-Phe), or a charged amino acid; wherein RlO is selected from a group consisting of SEQ ID NOs: 1021, 1026, 1027, and 1050.
- the peptide comprises an oxy derivative of PLA.
- oxy derivative of PLA refers to a compound comprising a modified structure of PLA in which the hydroxyl group has been replaced with O-R ⁇ , wherein Rn is a chemical moiety.
- the oxy derivative of PLA can be, for example, an ester of PLA or an ether of PLA.
- the ester may be formed by upon reaction of the hydroxyl of PLA with a carbonyl bearing a nucleophile.
- the nucleophile can be any suitable nucleophile, including, but not limited to an amine or hydroxyl. Accordingly, the ester of PLA can comprise the structure of Formula XI:
- R7 is an ester formed upon reaction of the hydroxyl of PLA with a carbonyl bearing a nucleophile.
- the carbonyl bearing a nucleophile (which reacts with the hydroxyl of PLA to form an ester) can be, for example, a carboxylic acid, a carboxylic acid derivative, or an activated ester of a carboxylic acid.
- the carboxylic acid derivative can be, but is not limited to, an acyl chloride, an acid anhydride, an amide, an ester, or a nitrile.
- the activated ester of a carboxylic acid can be, for example, N-hydroxysuccinimide (NHS), tosylate (Tos), a carbodiimide, or a hexafluorophosphate.
- the carbodiimide is 1,3- dicyclohexylcarbodiimide (DCC), l,l'-carbonyldiimidazole (CDI), l-ethyl-3-(3- dimethylaminopropyl)carbodiimide hydrochloride (EDC), or 1,3-diisopropylcarbodiimide (DICD).
- the hexafluorophosphate is selected from a group consisting of hexafluorophosphate benzotriazol- 1 -yl-oxy-tris(dimethylamino)phosphonium hexafluorophosphate (BOP), benzotriazol- 1-yl- oxytripyrrolidinophosphonium hexafluorophosphate (PyBOP), 2-(lH-7-azabenzotriazol-l-yl)-l,l,3,3-tetramethyl uronium hexafluorophosphate (HATU), and o-benzotriazole-N,N,N',N'-tetramethyl-uronium- hexafluoro-phosphate (HBTU).
- BOP hexafluorophosphate benzotriazol- 1 -yl-oxy-tris(dimethylamino)phosphonium hexafluorophosphate
- PyBOP benzotriazol
- ethers from reaction with a hydroxyl group e.g., the hydroxyl of PLA
- a hydroxyl group of PLA may be reacted with a halogenated alkyl or tosylated alkyl alcohol to form an ether bond.
- the chemical moiety bound to PLA via an oxygen- containing bond is a polymer (e.g., a polyalkylene glycol), a carbohydrate, an amino acid, a peptide, or a lipid, e.g., a fatty acid or a steroid.
- the chemical moiety is an amino acid, which, optionally, is a part of a peptide, such that Formula XI is a depsipeptide.
- PLA may be at a position other than the N-terminal amino acid residue of the peptide, such that the peptide comprises one or more (e.g., 1, 2, 3, 4, 5, 6, or more) amino acids N-terminal to the PLA residue.
- the peptide can comprise PLA at position n, wherein n is 2, 3, 4, 5, or 6 of the peptide.
- the amino acids N-terminal to the PLA residue may be synthetic or naturally- occurring.
- the amino acids which are N-terminal PLA are naturally-occurring amino acids.
- the amino acids which are N-terminal to PLA are the N-terminal amino acids of native glucagon.
- the peptide can comprise at the N-terminus the amino acid sequence of any of SEQ ID NOs: 1052-1056, wherein PLA is linked to threonine via an ester bond: SEQ ID NO: 1052 His-Ser-Gln-Gly-Thr-PLA
- one or more of the N-terminal amino acids may be substituted with an amino acid other than the amino acid of native glucagon.
- the amino acid at position 1 and/or position 2 may be an amino acid which reduces susceptibility to cleavage by dipeptidyl peptidase IV.
- position 1 of the peptide is an amino acid selected from the group consisting of D-histidine, alpha, alpha- dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha- methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo- histidine.
- position 2 of the antagonist/agonist peptide is an amino acid selected from the group consisting of D-serine, D-alanine, valine, glycine, N-methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB).
- the amino acid at position 3 of the peptide may be glutamic acid, as opposed to the native glutamine residue of native glucagon.
- the peptide comprises at the N-terminus the amino acid sequence of any of SEQ ID NOs: 1057-1059.
- the polymer may be any polymer, provided that it can react with the hydroxyl group of PLA.
- the polymer may be one that naturally or normally comprises a carbonyl bearing a nucleophile. Alternatively, the polymer may be one which was derivatized to comprise the carbonyl bearing the carbonyl.
- the polymer may be a derivatized polymer of any of: polyamides, polycarbonates, polyalkylenes and derivatives thereof including, polyalkylene glycols, poly alky lene oxides, polyalkylene terepthalates, polymers of acrylic and methacrylic esters, including poly(methyl methacrylate), poly(ethyl methacrylate), poly(butylmethacrylate), poly(isobutyl methacrylate), poly(hexylmethacrylate), poly(isodecyl methacrylate), poly(lauryl methacrylate), poly(phenyl methacrylate), poly(methyl acrylate), poly(isopropyl acrylate), poly(isobutyl acrylate), and poly(octadecyl acrylate), polyvinyl polymers including polyvinyl alcohols, polyvinyl ethers, polyvinyl esters, polyvinyl halides, poly(vinyl acetate), and polyviny
- the polymer can be a biodegradable polymer, including a synthetic biodegradable polymer (e.g., polymers of lactic acid and glycolic acid, poly anhydrides, poly(ortho)esters, polyurethanes, poly(butic acid), poly(valeric acid), and poly(lactide-cocaprolactone)), and a natural biodegradable polymer (e.g., alginate and other polysaccharides including dextran and cellulose, collagen, chemical derivatives thereof (substitutions, additions of chemical groups, for example, alkyl, alkylene, hydroxylations, oxidations, and other modifications routinely made by those skilled in the art), albumin and other hydrophilic proteins (e.g., zein and other prolamines and hydrophobic proteins)), as well as any copolymer or mixture thereof.
- these materials degrade either by enzymatic hydrolysis or exposure to water in vivo, by surface or bulk erosion.
- the polymer can be a bioadhesive polymer, such as a bioerodible hydrogel described by H. S. Sawhney, C. P. Pathak and J. A. Hubbell in Macromolecules, 1993, 26, 581-587, the teachings of which are incorporated herein, polyhyaluronic acids, casein, gelatin, glutin, poly anhydrides, polyacrylic acid, alginate, chitosan, poly(methyl methacrylates), poly(ethyl methacrylates), poly(butylmethacrylate), poly(isobutyl methacrylate), poly(hexylmethacrylate), poly(isodecyl methacrylate), poly(lauryl methacrylate), poly(phenyl methacrylate), poly(methyl acrylate), poly(isopropyl acrylate), poly(isobutyl acrylate), and poly(octadecyl acrylate).
- a bioadhesive polymer such as
- the polymer is a water-soluble polymer.
- Suitable water-soluble polymers include, for example, polyvinylpyrrolidone, hydroxypropyl cellulose (HPC; Klucel), hydroxypropyl methylcellulose (HPMC; Methocel), nitrocellulose, hydroxypropyl ethylcellulose, hydroxypropyl butylcellulose, hydroxypropyl pentylcellulose, methyl cellulose, ethylcellulose (Ethocel), hydroxyethyl cellulose, various alkyl celluloses and hydroxyalkyl celluloses, various cellulose ethers, cellulose acetate, carboxymethyl cellulose, sodium carboxymethyl cellulose, calcium carboxymethyl cellulose, vinyl acetate/crotonic acid copolymers, poly-hydroxyalkyl methacrylate, hydroxymethyl methacrylate, methacrylic acid copolymers, polymethacrylic acid, polymethylmethacrylate, maleic anhydride/methyl vinyl ether
- the polymer is a polyalkylene glycol, including, for example, polyethylene glycol (PEG).
- the carbohydrate may be any carbohydrate provided that it comprises or is made to comprise a carbonyl with an alpha leaving group.
- the carbohydrate for example, may be one which has been derivatized to comprise a carbonyl with an alpha leaving group.
- the carbohydrate may be a derivatized form of a monosaccharide (e.g., glucose, galactose, fructose), a disaccharide (e.g., sucrose, lactose, maltose), an oligosaccharide (e.g., raffinose, stachyose), a polysaccharide (a starch, amylase, amylopectin, cellulose, chitin, callose, laminarin, xylan, mannan, fucoidan, galactomannan.
- a monosaccharide e.g., glucose, galactose, fructose
- a disaccharide e.g., sucrose, lactose, maltose
- an oligosaccharide e.g., raffinose, stachyose
- a polysaccharide a starch, amylase, amy
- the lipid may be any lipid comprising a carbonyl with an alpha leaving group.
- the lipid for example, may be one which is derivatized to comprise the carbonyl.
- the lipid may be a derivative of a fatty acid (e.g., a C4-C30 fatty acid, eicosanoid, prostaglandin, leukotriene, thromboxane, N-acyl ethanolamine), glycerolipid (e.g., mono-, di- , tri-substituted glycerols), glycerophospholipid (e.g., phosphatidylcholine, phosphatidylinositol, phosphatidylethanolamine, phosphatidylserine), sphingolipid (e.g., sphingosine, ceramide), sterol lipid (e.g., steroid, cholesterol), prenol lipid, saccharolipid, or a
- R7 has a molecular weight of about 100 kDa or less, e.g., about
- R7 can have a molecular weight of about 35 kDa or less, about 30 kDa or less, about 25 kDa or less, about 20 kDa or less, about 15 kDa or less, about 10 kDa or less, about 5 kDa or less, or about 1 kDa.
- the peptide comprising the general structure of A-B-C comprises, as A, a peptide of 2 to 6 amino acids in which two consecutive amino acids of the peptide of A are linked via an ester or ether bond.
- the ester or ether bond may be, e.g., between amino acids 2 and 3, 3 and 4, 4 and 5, or 5 and 6.
- the peptide of A may be further modified by covalent linkage to another chemical moiety including linkage to a polymer (e.g. a hydrophilic polymer), alkylation, or acylation.
- the above-described class 5 peptide comprising PLA is modified to comprise an oxy derivative of PLA, such as, for instance, an ester of PLA or an ether of PLA.
- the class 5 peptide can comprise the amino acid sequence of any of SEQ ID NOs: 1002, 1005-1020, 1022-1025, 1032-1036, 1038, 1039, 1045, 1046, and 1051 , wherein the PLA is linked via an ester or ether bond to an amino acid, peptide, polymer, acyl group, or alkyl group.
- the amino acid, peptide, polymer, acyl group, or alkyl group may be any of those described herein.
- the class 5 peptide may be considered as a depsipeptide.
- the above-described class 5 peptide which lacks PLA is modified to comprise at least one ester bond or ether bond between two consecutive amino acids which are N-terminal to the amino acid at position 7 (according to the numbering of native glucagon).
- the class 5 peptide comprises at least one ester or ether bond between the two consecutive amino acids.
- the Class 5 peptide comprises the N-terminal 6 amino acids of SEQ ID NO: 1001 and two consecutive amino acids of the N-terminal 6 amino acids are linked via an ester or ether bond.
- the peptide of A may comprise any amino acids, synthetic or naturally occurring, provided that at least two consecutive amino acids are linked via an ester or ether bond.
- the peptide of A comprises amino acids of native glucagon.
- the amino acid at position 1 and/or position 2 may be an amino acid which reduces susceptibility to cleavage by dipeptidyl peptidase IV.
- the peptide of A can comprise at position 1 an amino acid selected from the group consisting of D-histidine, alpha, alpha-dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha-methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo-histidine. More particularly, in some embodiments, position 2 of the peptide of A is an amino acid selected from the group consisting of D-serine, D-alanine, valine, glycine, N-methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB).
- DIA alpha
- N-methyl histidine alpha-methyl histidine
- imidazole acetic acid desaminohistidine
- hydroxyl-histidine hydroxyl-histidine
- acetyl-histidine acetyl-histidine and
- amino acid at position 3 of the peptide of A may be glutamic acid, as opposed to the native glutamine residue of native glucagon.
- the peptide of general structure of A-B-C can comprise an amino acid sequence of:
- Xaa 1 -Xaa 2 -Xaa 3 -Thr-Gly-Phe SEQ ID NO: 1107
- Xaa 2 -Xaa 3 -Thr-Gly-Phe SEQ ID NO: 1108
- Xaa 3 -Thr-Gly-Phe SEQ ID NO: 1109
- Xaaj is selected from a group consisting of: His, D-histidine, alpha, alpha- dimethyl imidiazole acetic acid (DMIA), N-methyl histidine, alpha- methyl histidine, imidazole acetic acid, desaminohistidine, hydroxyl-histidine, acetyl-histidine and homo- histidine
- Xaa 2 is selected from a group consisting of: Ser, D-serine, D-alanine, valine, glycine, N-methyl serine, N-methyl alanine, and aminoisobutyric acid (AIB); and
- B is modified by up to three amino acid modifications.
- B which represents native amino acid sequence of SEQ ID NO: 1001 is modified by one or more conservative amino acid modifications.
- B comprises one or more amino acid modifications selected from the group consisting of (iv) to (x), as described herein.
- B comprises one or both of the amino acid modifications (v) and (vi).
- B comprises one or a combination of amino acid modifications selected from the group consisting of (iv), (vii), (viii), (ix), and (x), in addition to (v) and (vi).
- the peptide comprising the general structure A-B-C may comprise one or more charged amino acids at the C-terminus, e.g., as Y and/or Z, as described herein.
- the peptide comprising the general structure A-B-C may further comprise one to two charged amino acids C-terminal to Z, when C comprises X-Y-Z.
- the charged amino acids can be, for example, one of Lys, Arg, His, Asp, and GIu.
- Y is Asp.
- the peptide comprising the general structure A-B-C comprises a hydrophilic moiety covalently bound to an amino acid residue at position 1, 16, 20, 21, or 24 (according to the amino acid numbering of SEQ ID NO: 1001), or at the N- or C-terminal residue of the peptide comprising the general structure A-B-C.
- the hydrophilic moiety is attached to a Cys residue of the peptide comprising the general structure A-B-C.
- the amino acid at position 16, 21, 24, or 29 of native glucagon (SEQ ID NO: 1001) may be substituted with a Cys residue.
- a Cys residue comprising a hydrophilic moiety may be added to the C-terminus of the peptide comprising the general structure A-B-C as position 30 or as position 40, e.g., when the peptide comprising the general structure A-B-C comprises a C-terminal extension (positions according to the amino acid numbering of SEQ ID NO: 1001).
- the hydrophilic moiety may be attached to the PLA of the peptide comprising the general structure A-B-C via the hydroxyl moiety of PLA.
- the hydrophilic moiety can be any of those described herein, including, for example, polyethylene glycol.
- the peptide comprising the general structure A-B-C comprises a stabilized alpha helix by virtue of incorporation of an intramolecular bridge.
- the intramolecular bridge is a lactam bridge.
- the lactam bridge may be between the amino acids at positions 9 and 12, the amino acids at positions 12 and 16, the amino acids at positions 16 and 20, the amino acids at positions 20 and 24, or the amino acids at positions 24 and 28 (according to the amino acid numbering of SEQ ID NO: 1001).
- the amino acids at positions 12 and 16 or at positions 16 and 20 are linked via a lactam bridge. Other positions of the lactam bridge are contemplated.
- the peptide comprising the general structure A-B-C can comprise an alpha, alpha di-substituted amino acid at, for example, any of positions 16, 20, 21, or 24 (according to the amino acid numbering of SEQ ID NO: 1001).
- the alpha, alpha di-substituted amino acid is AIB.
- the AIB is located at position 16 (according to the numbering of SEQ ID NO: 1001).
- the peptide comprising the general structure A-B-C may be modified to comprise an acidic amino acid at position 16 (according to the numbering of SEQ ID NO: 1001), which modification enhances the stability of the alpha helix.
- the acidic amino acid in one embodiment, is an amino acid comprising a side chain sulfonic acid or a side chain carboxylic acid.
- the acidic amino acid is selected from the group consisting of GIu, Asp, homoglutamic acid, a sulfonic acid derivative of Cys, cysteic acid, homocysteic acid, Asp, and an alkylated derivative of Cys having the structure of
- X 5 is C 1 -C 4 alkyl, C 2 -C 4 alkenyl, or C 2 -C 4 alkynyl.
- the Class 5 peptide may comprise the amino acid sequence of any of SEQ ID NOs: 1060-1070, 1073-1078, 1080-1088, 1090-1096, 1103, 1104, 1106, and 1114-1118, or comprising the amino acid sequence of any of Peptides 2-6 of Table 9, Peptides 1-8 of Table 10, and Peptides 2-6, 8, and 9 of Table 11.
- the peptide comprising the general structure A-B-C is a Class 5 peptide.
- the peptide exhibits exhibits at least about 50% of the maximum agonism achieved by native GLP-I at the GLP-I receptor and at least about 50% inhibition of the maximum response achieved by native glucagon at the glucagon receptor.
- the peptide exhibits at least about 55%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 95%, or about 100% of the maximum agonism achieved by native GLP-I at the GLP-I receptor.
- the peptide may exhibit at least about 55%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 95%, or about 100% inhibition of the maximum response achieved by native glucagon at the glucagon receptor.
- a peptide with Class 5 peptide or conjugate thereof comprising:
- Arg at position 18 is replaced with Ala
- Asp at position 21 is replaced with GIu
- VaI at position 23 is replaced with He
- GIn at position 24 is replaced with Ala; or (d) other modifications that stabilize the alpha-helix structure around amino acid positions 12-29 according to the amino acid numbering of wild type glucagon; and
- hydrophilic moiety such as polyethylene glycol, e.g. at the N-terminus, or at position 6, 16, 17, 20, 21, 24, 29, 40 or at the C-terminal amino acid; and/or
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2713348A CA2713348C (en) | 2008-01-30 | 2009-01-21 | Ester-based peptide prodrugs |
| CN200980112047.4A CN101983066B (en) | 2008-01-30 | 2009-01-21 | Insulin prodrug based on ester |
| US12/865,058 US20110065633A1 (en) | 2008-01-30 | 2009-01-21 | Ester-based peptide prodrugs |
| EP09708686A EP2249853A4 (en) | 2008-01-30 | 2009-01-21 | Ester-based peptide prodrugs |
| JP2010545058A JP2011511778A (en) | 2008-01-30 | 2009-01-21 | Ester-based peptide prodrugs |
| AU2009210570A AU2009210570B2 (en) | 2008-01-30 | 2009-01-21 | Ester-based insulin prodrugs |
| US12/845,455 US8697838B2 (en) | 2008-01-30 | 2010-07-28 | Ester-based insulin prodrugs |
| US14/182,706 US9089539B2 (en) | 2008-01-30 | 2014-02-18 | Ester-based insulin prodrugs |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US2467208P | 2008-01-30 | 2008-01-30 | |
| US61/024,672 | 2008-01-30 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/845,455 Continuation US8697838B2 (en) | 2008-01-30 | 2010-07-28 | Ester-based insulin prodrugs |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2009099763A1 true WO2009099763A1 (en) | 2009-08-13 |
Family
ID=40952419
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2009/031593 WO2009099763A1 (en) | 2008-01-30 | 2009-01-21 | Ester-based peptide prodrugs |
Country Status (12)
| Country | Link |
|---|---|
| US (3) | US20110065633A1 (en) |
| EP (1) | EP2249853A4 (en) |
| JP (1) | JP2011511778A (en) |
| CN (1) | CN101983066B (en) |
| AR (1) | AR073335A1 (en) |
| AU (1) | AU2009210570B2 (en) |
| CA (1) | CA2713348C (en) |
| CL (1) | CL2009000202A1 (en) |
| PA (1) | PA8814001A1 (en) |
| PE (1) | PE20091440A1 (en) |
| TW (1) | TW200938225A (en) |
| WO (1) | WO2009099763A1 (en) |
Cited By (93)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010080609A1 (en) | 2008-12-19 | 2010-07-15 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| WO2011089215A1 (en) | 2010-01-22 | 2011-07-28 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aromatic amine-containing drugs |
| WO2011089216A1 (en) | 2010-01-22 | 2011-07-28 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aliphatic amine-containing drugs |
| WO2011143209A1 (en) | 2010-05-13 | 2011-11-17 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| WO2011159895A2 (en) | 2010-06-16 | 2011-12-22 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| WO2011162968A1 (en) * | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Dipeptide linked medicinal agents |
| WO2011163462A2 (en) | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| WO2011163460A1 (en) * | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Yl-based insulin-like growth factors exhibiting high activity at the insulin receptor |
| WO2012059762A1 (en) | 2010-11-03 | 2012-05-10 | Arecor Limited | Novel composition comprising glucagon |
| US8338368B2 (en) | 2005-11-07 | 2012-12-25 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting physiological solubility and stability |
| WO2013074910A1 (en) | 2011-11-17 | 2013-05-23 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting glucocorticoid receptor activity |
| US8450270B2 (en) | 2008-06-17 | 2013-05-28 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility and stability in physiological pH buffers |
| US8454971B2 (en) | 2007-02-15 | 2013-06-04 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| CN103179979A (en) * | 2010-06-24 | 2013-06-26 | 印第安纳大学研究及科技有限公司 | Amide based glucagon superfamily peptide prodrugs |
| WO2013096386A1 (en) | 2011-12-20 | 2013-06-27 | Indiana University Research And Technology Corporation | Ctp-based insulin analogs for treatment of diabetes |
| US8481485B2 (en) | 2008-12-19 | 2013-07-09 | Indiana University Research And Technology Corporation | Insulin analogs |
| US8507428B2 (en) | 2010-12-22 | 2013-08-13 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| WO2013127779A1 (en) * | 2012-03-01 | 2013-09-06 | Novo Nordisk A/S | Glp-1 prodrugs |
| US8541368B2 (en) | 2011-09-23 | 2013-09-24 | Novo Nordisk A/S | Glucagon analogues |
| US8546327B2 (en) | 2008-06-17 | 2013-10-01 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8669228B2 (en) | 2007-01-05 | 2014-03-11 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility in physiological pH buffers |
| WO2014052451A2 (en) | 2012-09-26 | 2014-04-03 | Indiana University Research And Technology Corporation | Insulin analog dimers |
| US8697838B2 (en) | 2008-01-30 | 2014-04-15 | Indiana University Research And Technology Corporation | Ester-based insulin prodrugs |
| US8703701B2 (en) | 2009-12-18 | 2014-04-22 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8729017B2 (en) | 2011-06-22 | 2014-05-20 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| WO2014088836A1 (en) | 2012-12-03 | 2014-06-12 | Merck Sharp & Dohme Corp. | O-glycosylated carboxy terminal portion (ctp) peptide-based insulin and insulin analogues |
| EP2741744A1 (en) * | 2011-08-12 | 2014-06-18 | Ascendis Pharma A/S | Carrier-linked prodrugs having reversible carboxylic ester linkages |
| WO2014099577A1 (en) | 2012-12-17 | 2014-06-26 | Merck Sharp & Dohme Corp. | Process for purifying insulin and analogues thereof |
| US8969288B2 (en) | 2008-12-19 | 2015-03-03 | Indiana University Research And Technology Corporation | Amide based glucagon and superfamily peptide prodrugs |
| US8975223B2 (en) | 2010-12-22 | 2015-03-10 | Marcadia Biotech, Inc. | Methods for treating metabolic disorders and obesity with a peptide comprising the amino acid sequence of SEQ ID No. 146 |
| US8981047B2 (en) | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Glucagon antagonists |
| US8980830B2 (en) | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Peptide compounds exhibiting glucagon antagonist and GLP-1 agonist activity |
| WO2015051052A2 (en) | 2013-10-04 | 2015-04-09 | Merck Sharp & Dohme Corp. | Glucose-responsive insulin conjugates |
| US9023986B2 (en) | 2010-10-25 | 2015-05-05 | Hoffmann-La Roche Inc. | Glucose-dependent insulinotropic peptide analogs |
| EP2528618A4 (en) * | 2010-01-27 | 2015-05-27 | Univ Indiana Res & Tech Corp | Glucagon antagonist - gip agonist conjugates and compositions for the treatment of metabolic disorders and obesity |
| WO2015084694A2 (en) | 2013-12-04 | 2015-06-11 | Merck Sharp & Dohme Corp. | Method for preparing crystalline insulin |
| US9062124B2 (en) | 2008-06-17 | 2015-06-23 | Indiana University Research And Technology Corporation | GIP-based mixed agonists for treatment of metabolic disorders and obesity |
| WO2015094876A1 (en) * | 2013-12-18 | 2015-06-25 | Eli Lilly And Company | Novel compound for treatment of severe hypoglycemia |
| WO2015138548A1 (en) | 2014-03-14 | 2015-09-17 | Merck Sharp & Dohme Corp. | Purifying insulin using cation exchange and reverse phase chromatography in the presence of an organic modifier and elevated temperature |
| US9145451B2 (en) | 2010-05-13 | 2015-09-29 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhbiting G protein coupled receptor activity |
| US9150632B2 (en) | 2009-06-16 | 2015-10-06 | Indiana University Research And Technology Corporation | GIP receptor-active glucagon compounds |
| US9156902B2 (en) | 2011-06-22 | 2015-10-13 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| WO2016014325A1 (en) | 2014-07-21 | 2016-01-28 | Merck Sharp & Dohme Corp. | Chromatography process for purification of inlsulin and insulin analogs |
| RU2578591C2 (en) * | 2008-12-19 | 2016-03-27 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Dipeptide linked medicinal agents |
| US9340600B2 (en) | 2012-06-21 | 2016-05-17 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| WO2016081670A2 (en) | 2014-11-21 | 2016-05-26 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| US9365632B2 (en) | 2012-10-09 | 2016-06-14 | Sanofi | Exendin-4 derivatives as dual GLP1/glucagon agonists |
| WO2016144658A1 (en) | 2015-03-10 | 2016-09-15 | Merck Sharp & Dohme Corp. | Process for preparing recombinant insulin using microfiltration |
| US9474790B2 (en) | 2013-04-18 | 2016-10-25 | Novo Nordisk A/S | Stable, protracted GLP-1/glucagon receptor co-agonists for medical use |
| US9522946B2 (en) | 2011-06-10 | 2016-12-20 | Hanmi Science Co., Ltd. | Oxyntomodulin derivatives and pharmaceutical composition for treating obesity comprising the same |
| WO2017040363A1 (en) | 2015-09-02 | 2017-03-09 | Merck Sharp & Dohme Corp. | A process for obtaining insulin with correctly formed disulfide bonds |
| US9670261B2 (en) | 2012-12-21 | 2017-06-06 | Sanofi | Functionalized exendin-4 derivatives |
| US9688736B2 (en) | 2013-12-18 | 2017-06-27 | Eli Lilly And Company | Compound for treatment of severe hypoglycemia |
| US9694053B2 (en) | 2013-12-13 | 2017-07-04 | Sanofi | Dual GLP-1/glucagon receptor agonists |
| US9724420B2 (en) | 2012-11-06 | 2017-08-08 | Hanmi Pharm. Co., Ltd. | Liquid formulation of protein conjugate comprising an oxyntomodulin derivative covalently linked to a non-peptidyl polymer to an immunoglobulin FC region |
| US9731031B2 (en) | 2011-06-17 | 2017-08-15 | Hanmi Science Co., Ltd. | Conjugate comprising oxyntomodulin and an immunoglobulin fragment, and use thereof |
| US9751926B2 (en) | 2013-12-13 | 2017-09-05 | Sanofi | Dual GLP-1/GIP receptor agonists |
| US9750788B2 (en) | 2013-12-13 | 2017-09-05 | Sanofi | Non-acylated exendin-4 peptide analogues |
| US9758561B2 (en) | 2014-04-07 | 2017-09-12 | Sanofi | Dual GLP-1/glucagon receptor agonists derived from exendin-4 |
| US9764004B2 (en) | 2015-10-26 | 2017-09-19 | Eli Lilly And Company | Glucagon receptor agonists |
| US9771406B2 (en) | 2014-04-07 | 2017-09-26 | Sanofi | Peptidic dual GLP-1/glucagon receptor agonists derived from exendin-4 |
| US9775904B2 (en) | 2014-04-07 | 2017-10-03 | Sanofi | Exendin-4 derivatives as peptidic dual GLP-1/glucagon receptor agonists |
| US9789165B2 (en) | 2013-12-13 | 2017-10-17 | Sanofi | Exendin-4 peptide analogues as dual GLP-1/GIP receptor agonists |
| WO2017189342A1 (en) | 2016-04-26 | 2017-11-02 | Merck Sharp & Dohme Corp. | Insulin dimer-incretin conjugates |
| WO2017192449A1 (en) * | 2016-05-06 | 2017-11-09 | Phasebio Pharmaceuticals, Inc. | Elp fusion proteins for controlled and sustained release |
| WO2017221026A1 (en) | 2016-06-24 | 2017-12-28 | Arecor Limited | Novel composition |
| EP3272877A1 (en) | 2016-07-18 | 2018-01-24 | ETH Zurich | B-cell-mimetic cells |
| WO2018015330A1 (en) | 2016-07-18 | 2018-01-25 | Eth Zurich | B-cell-mimetic cells |
| US9901621B2 (en) | 2012-07-25 | 2018-02-27 | Hanmi Pharm. Co., Ltd. | Composition for treating hyperlipidemia comprising oxyntomodulin derivative |
| US9932381B2 (en) | 2014-06-18 | 2018-04-03 | Sanofi | Exendin-4 derivatives as selective glucagon receptor agonists |
| US9944687B2 (en) | 2011-07-04 | 2018-04-17 | Imperial Innovations Limited | Compounds and their effects on feeding behaviour |
| US9982029B2 (en) | 2015-07-10 | 2018-05-29 | Sanofi | Exendin-4 derivatives as selective peptidic dual GLP-1/glucagon receptor agonists |
| WO2018213377A1 (en) | 2017-05-17 | 2018-11-22 | Arcus Biosciences, Inc. | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
| US10233230B2 (en) | 2014-09-16 | 2019-03-19 | Hanmi Pharm. Co., Ltd. | Use of a long acting GLP-1/glucagon receptor dual agonist for the treatment of non-alcoholic fatty liver disease |
| US10232020B2 (en) | 2014-09-24 | 2019-03-19 | Indiana University Research And Technology Corporation | Incretin-insulin conjugates |
| US10385107B2 (en) | 2014-09-24 | 2019-08-20 | Indiana Univeresity Researc and Technology Corporation | Lipidated amide-based insulin prodrugs |
| US10413593B2 (en) | 2014-10-24 | 2019-09-17 | Merck Sharp & Dohme Corp. | Co-agonists of the glucagon and GLP-1 receptors |
| US10550168B2 (en) | 2012-11-06 | 2020-02-04 | Hanmi Pharm. Co., Ltd. | Composition for treating diabetes or diabesity comprising oxyntomodulin analog |
| US10570184B2 (en) | 2014-06-04 | 2020-02-25 | Novo Nordisk A/S | GLP-1/glucagon receptor co-agonists for medical use |
| US10689430B2 (en) | 2016-05-25 | 2020-06-23 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| US10696725B2 (en) | 2015-06-30 | 2020-06-30 | Hanmi Pharm. Co., Ltd. | Glucagon derivative and a composition comprising a long acting conjugate of the same |
| US10696726B2 (en) | 2013-03-14 | 2020-06-30 | Indiana University Research And Technology Corporation | Insulin-incretin conjugates |
| US10806797B2 (en) | 2015-06-05 | 2020-10-20 | Sanofi | Prodrugs comprising an GLP-1/glucagon dual agonist linker hyaluronic acid conjugate |
| US10953076B2 (en) | 2016-05-24 | 2021-03-23 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists and GLP-1 analogues |
| US10981967B2 (en) | 2015-12-31 | 2021-04-20 | Hanmi Pharm. Co., Ltd. | Long-acting conjugate of triple glucagon/GLP-1/GIP receptor agonist |
| US11041009B2 (en) | 2017-03-23 | 2021-06-22 | Merck Sharp & Dohme Corp. | Glucose responsive insulin comprising a tri-valent sugar cluster for treatment of diabetes |
| WO2021188769A1 (en) | 2020-03-19 | 2021-09-23 | Arcus Biosciences, Inc. | Tetralin and tetrahydroquinoline compounds as inhibitors of hif-2alpha |
| US11135271B2 (en) | 2014-12-30 | 2021-10-05 | Hanmi Pharm. Co., Ltd. | Glucagon derivatives with improved stability |
| US11142559B2 (en) | 2016-06-29 | 2021-10-12 | Hanmi Pharm. Co., Ltd. | Glucagon derivative, conjugate thereof, composition comprising same, and therapeutic use thereof |
| EP3889185A3 (en) * | 2014-03-31 | 2022-01-26 | Hanmi Pharm. Co., Ltd. | Composition for improving the solubility of a protein or peptide by using immunoglobulin fc fragment linkage |
| US11254724B2 (en) | 2014-12-30 | 2022-02-22 | Hanmi Pharm. Co., Ltd. | Glucagon derivatives |
| US11434271B2 (en) | 2011-11-04 | 2022-09-06 | Hanmi Science Co., Ltd. | Method for preparing physiologically active polypeptide complex |
| EP4310082A2 (en) | 2017-01-20 | 2024-01-24 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
Families Citing this family (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2932477C (en) * | 2013-12-06 | 2023-10-10 | Baikang (Suzhou) Co., Ltd | Bioreversable promoieties for nitrogen-containing and hydroxyl-containing drugs |
| US10918730B2 (en) | 2016-04-19 | 2021-02-16 | Griffon Pharmaceuticals International Sa | Pegylated bioactive peptides and uses thereof |
| EP3661543A4 (en) * | 2017-07-31 | 2021-09-22 | The Trustees of Indiana University | Modified ddah polypeptides comprising a pharmacokinetic enhancing moiety, improved pharmacology and their uses |
| CN111621014B (en) * | 2019-02-28 | 2022-09-30 | 北京大学 | Hydroxyproline-based polymer, and preparation method and depolymerization method thereof |
| CN109721653B (en) * | 2019-03-05 | 2023-02-03 | 嘉兴学院 | Glucagon-like peptide-1 fragment analogue and application thereof |
| WO2022066827A1 (en) * | 2020-09-25 | 2022-03-31 | University Of Washington | De novo designed mixed chirality peptide macrocycles with internal symmetry |
| CN114437181B (en) * | 2022-01-25 | 2023-03-24 | 深圳深创生物药业有限公司 | GLP-1R/GCGR/GIPR triple receptor agonist and application thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060171920A1 (en) * | 2003-04-08 | 2006-08-03 | Yeda Research And Development Co., Ltd. | Reversible pegylated drugs |
| US20070203058A1 (en) * | 2003-09-19 | 2007-08-30 | Novo Nordisk A/S | Novel Glp-1 Derivatives |
Family Cites Families (114)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3740385A (en) | 1970-05-07 | 1973-06-19 | M Ondetti | N-terminal derivatives of secretin |
| US4275152A (en) * | 1977-02-03 | 1981-06-23 | Eastman Kodak Company | Hydrolysis of protein-bound cholesterol esters |
| DK119785D0 (en) * | 1985-03-15 | 1985-03-15 | Nordisk Gentofte | INSULIN PREPARATION |
| PT83613B (en) | 1985-10-28 | 1988-11-21 | Lilly Co Eli | Process for the selective chemical removal of a protein amino-terminal residue |
| US4741897A (en) | 1986-07-08 | 1988-05-03 | Baxter Travenol | Thyroxine analogs and reagents for thyroid hormone assays |
| US4876242A (en) | 1987-09-21 | 1989-10-24 | Merck & Co., Inc. | Human insulin-like growth factor analoges with reduced binding to serum carrier proteins and their production in yeast |
| GB8728561D0 (en) | 1987-12-07 | 1988-01-13 | Glaxo Group Ltd | Chemical compounds |
| US5514646A (en) | 1989-02-09 | 1996-05-07 | Chance; Ronald E. | Insulin analogs modified at position 29 of the B chain |
| US5539084A (en) * | 1989-02-17 | 1996-07-23 | Coselco Mimotopes Pty. Ltd. | Method for the use and synthesis of peptides |
| US6225449B1 (en) | 1991-10-04 | 2001-05-01 | Washington University | Hormone analogs with multiple CTP extensions |
| HU210142B (en) | 1989-04-20 | 1995-02-28 | Sinai School Medicine | Process for producing hepatospecific insulin-analogues and pharmaceutical compositions containing them |
| US5545618A (en) | 1990-01-24 | 1996-08-13 | Buckley; Douglas I. | GLP-1 analogs useful for diabetes treatment |
| DK0512042T3 (en) | 1990-01-24 | 1998-05-11 | Douglas I Buckley | GLP-1 analogues useful in diabetes treatment |
| US5510459A (en) * | 1991-01-17 | 1996-04-23 | Zymogenetics, Inc. | Glucagon antagonists |
| US5268453A (en) | 1991-08-08 | 1993-12-07 | Scios Inc. | Tissue-selective insulin analogs |
| EP0688364A4 (en) | 1993-03-09 | 1997-10-15 | Abbott Lab | Genetically engineered enzymes and their conjugates for diagnostic assays |
| US5359030A (en) * | 1993-05-10 | 1994-10-25 | Protein Delivery, Inc. | Conjugation-stabilized polypeptide compositions, therapeutic delivery and diagnostic formulations comprising same, and method of making and using the same |
| US5480867A (en) * | 1993-12-29 | 1996-01-02 | The Rockefeller University | Glucagon analogs with serine replacements |
| US5512549A (en) * | 1994-10-18 | 1996-04-30 | Eli Lilly And Company | Glucagon-like insulinotropic peptide analogs, compositions, and methods of use |
| US5869602A (en) * | 1995-03-17 | 1999-02-09 | Novo Nordisk A/S | Peptide derivatives |
| US6476290B1 (en) | 1995-04-19 | 2002-11-05 | Dalhousie University | Transgenic tilapia comprising a humanized insulin gene |
| AU5724996A (en) | 1995-05-05 | 1996-11-21 | Eli Lilly And Company | Single chain insulin with high bioactivity |
| US6180767B1 (en) * | 1996-01-11 | 2001-01-30 | Thomas Jefferson University | Peptide nucleic acid conjugates |
| JP2001505872A (en) | 1996-09-09 | 2001-05-08 | ジーランド ファーマシューティカルズ アクティーゼルスカブ | Peptide prodrug containing α-hydroxy acid linker |
| UA65549C2 (en) * | 1996-11-05 | 2004-04-15 | Елі Ліллі Енд Компані | Use of glucagon-like peptides such as glp-1, glp-1 analog, or glp-1 derivative in methods and compositions for reducing body weight |
| HUP0100928A3 (en) | 1998-02-23 | 2001-11-28 | Neurocrine Biosciences Inc San | Methods for treatment of diabetes using peptide analogues of insulin |
| EP1950223A3 (en) | 1998-03-09 | 2009-05-13 | Zealand Pharma A/S | Pharmacologically active peptide conjugates having a reduced tendency towards enzymatic hydrolysis |
| DE19828113A1 (en) | 1998-06-24 | 2000-01-05 | Probiodrug Ges Fuer Arzneim | Prodrugs of Dipeptidyl Peptidase IV Inhibitors |
| US6875741B2 (en) | 1998-09-02 | 2005-04-05 | Renuka Pillutla | Insulin and IGF-1 receptor agonists and antagonists |
| US20030236190A1 (en) * | 1998-09-02 | 2003-12-25 | Renuka Pillutla | Isulin and IGF-1 receptor agonists and antagonists |
| DE19908041A1 (en) | 1999-02-24 | 2000-08-31 | Hoecker Hartwig | Covalently bridged insulin dimers |
| MXPA01009805A (en) | 1999-03-29 | 2004-07-30 | Univ Ulster | Peptide. |
| US6746853B1 (en) | 1999-05-19 | 2004-06-08 | Xencor, Inc. | Proteins with insulin-like activity useful in the treatment of diabetes |
| AUPQ661800A0 (en) * | 2000-03-31 | 2000-05-04 | Metabolic Pharmaceuticals Limited | Insulin-potentiating compounds |
| US6677136B2 (en) * | 2000-05-03 | 2004-01-13 | Amgen Inc. | Glucagon antagonists |
| EP1290019A2 (en) * | 2000-06-14 | 2003-03-12 | Cytovax Biotechnologies Inc. | Use of coiled-coil structural scaffold to generate structure-specific peptides |
| CN1454214A (en) | 2000-08-02 | 2003-11-05 | 赛莱技术公司 | Modified peptides with increased potency |
| WO2002012201A1 (en) * | 2000-08-04 | 2002-02-14 | Dmi Biosciences, Inc. | Method of synthesizing diketopiperazines |
| ATE516820T1 (en) * | 2000-09-29 | 2011-08-15 | Schering Corp | PEGYLATED INTERLEUKIN 10 |
| KR100449454B1 (en) | 2000-10-02 | 2004-09-21 | 이현철 | Vector for Curing Diabetes Mellitus Containing Gene of Single-chain Insulin Analog |
| KR101229995B1 (en) | 2000-12-11 | 2013-02-06 | 씨제이 주식회사 | Fusion protein having the enhanced in vivo activity of erythropoietin |
| EP1243276A1 (en) | 2001-03-23 | 2002-09-25 | Franciscus Marinus Hendrikus De Groot | Elongated and multiple spacers containing activatible prodrugs |
| EP2322631B1 (en) * | 2001-04-19 | 2014-11-12 | The Scripps Research Institute | Methods and compositions for the prodcution of orthogonal tRNA-aminoacyl-tRNA synthetase pairs |
| US7085923B2 (en) | 2001-06-05 | 2006-08-01 | International Business Machines Corporation | High volume secure internet server |
| US20040002468A1 (en) * | 2001-08-08 | 2004-01-01 | Genzyme Corporation | Methods of treating diabetes and other blood sugar disorders |
| WO2003020201A2 (en) * | 2001-08-28 | 2003-03-13 | Eli Lilly And Company | Pre-mixes of glp-1 and basal insulin |
| GB0121709D0 (en) * | 2001-09-07 | 2001-10-31 | Imp College Innovations Ltd | Food inhibition agent |
| US7041646B2 (en) * | 2001-10-05 | 2006-05-09 | Bayer Pharmaceuticals Corporation | Methods of treating type 2 diabetes with peptides acting as both GLP-1 receptor agonists and glucagon receptor antagonists |
| AR036711A1 (en) * | 2001-10-05 | 2004-09-29 | Bayer Corp | PEPTIDES THAT ACT AS GLON-RECEPTOR AGONISTS AND AS GLUCAGON RECEPTOR ANTAGONISTS AND THEIR PHARMACOLOGICAL USE METHODS |
| CA2463908A1 (en) * | 2001-10-18 | 2003-04-24 | Bristol-Myers Squibb Company | Human glucagon-like-peptide-1 mimics and their use in the treatment of diabetes and related conditions |
| MXPA04006084A (en) | 2001-12-20 | 2005-03-31 | Lilly Co Eli | Insulin molecule having protracted time action. |
| WO2003059378A2 (en) * | 2001-12-29 | 2003-07-24 | Novo Nordisk A/S | Combined use of a glp-1 compound and another drug for treating dyslipidemia |
| EP1490090A4 (en) * | 2002-02-22 | 2006-09-20 | New River Pharmaceuticals Inc | Active agent delivery systems and methods for protecting and administering active agents |
| AU2003239478A1 (en) * | 2002-06-04 | 2003-12-22 | Eli Lilly And Company | Modified glucagon-like peptide-1 analogs |
| FR2842209B1 (en) | 2002-07-09 | 2007-11-23 | NOVEL ASPARTIC PROTEASE DITE SASPASE AND ITS USE IN THE COSMETIC AND THERAPEUTIC FIELD | |
| US7192922B2 (en) * | 2002-11-19 | 2007-03-20 | Allegheny-Singer Research Institute | Method of treating left ventricular dysfunction |
| WO2004067548A2 (en) | 2003-01-31 | 2004-08-12 | Theratechnologies Inc. | Chemically modified metabolites of regulatory peptides and methods of producing and using same |
| EP1626981A4 (en) | 2003-03-04 | 2006-11-22 | Biorexis Pharmaceutical Corp | Dipeptidyl-peptidase protected proteins |
| EP1605897B1 (en) * | 2003-03-19 | 2012-07-25 | Eli Lilly And Company | Polyethelene glycol link glp-1 compounds |
| GB0310593D0 (en) | 2003-05-08 | 2003-06-11 | Leuven K U Res & Dev | Peptidic prodrugs |
| EA008831B1 (en) * | 2003-06-12 | 2007-08-31 | Эли Лилли Энд Компани | Glp-1 analog fusion proteins |
| WO2004111078A2 (en) * | 2003-06-18 | 2004-12-23 | Theratechnologies Inc. | Compounds that modulate the glucagon response and uses thereof |
| WO2005025554A2 (en) | 2003-09-09 | 2005-03-24 | Japan Tobacco Inc. | Dipeptidyl peptidase iv inhibitor |
| CN101380476A (en) * | 2003-09-19 | 2009-03-11 | 诺沃挪第克公司 | Albumin-binding derivatives of therapeutic peptides |
| WO2005035003A2 (en) | 2003-09-22 | 2005-04-21 | Dihedron Corporation | Compositions and methods for increasing drug efficiency |
| US7364875B2 (en) * | 2003-10-30 | 2008-04-29 | Cresent Innovations, Inc. | Method for producing medical and commercial grade poly-gamma-glutamic acid of high molecular weight |
| PT1680443E (en) | 2003-11-05 | 2013-12-11 | Harvard College | Stabilized alpha helical peptides and uses thereof |
| CN102816228A (en) | 2003-12-03 | 2012-12-12 | 诺和诺德公司 | Single-chain insulin |
| US7028106B2 (en) | 2003-12-05 | 2006-04-11 | Hewlett-Packard Development Company, L.P. | Remapping routing information entries in an expander |
| MXPA06006745A (en) * | 2003-12-18 | 2006-08-18 | Novo Nordisk As | Novel glp-1 compounds. |
| US20060286129A1 (en) * | 2003-12-19 | 2006-12-21 | Emisphere Technologies, Inc. | Oral GLP-1 formulations |
| US20080318837A1 (en) * | 2003-12-26 | 2008-12-25 | Nastech Pharmaceutical Company Inc. | Pharmaceutical Formation For Increased Epithelial Permeability of Glucose-Regulating Peptide |
| JP2008515768A (en) * | 2004-06-17 | 2008-05-15 | エム ユー エス シー ファンデーション フォー リサーチ ディベロップメント | Unnatural amino acid |
| WO2006047214A2 (en) | 2004-10-21 | 2006-05-04 | Igf Oncology, Llc | Toxins and radionuclides coupled to igf-1 receptor ligands for treatment of cancer |
| US8263545B2 (en) | 2005-02-11 | 2012-09-11 | Amylin Pharmaceuticals, Inc. | GIP analog and hybrid polypeptides with selectable properties |
| CA2597649A1 (en) * | 2005-02-11 | 2006-08-17 | Amylin Pharmaceuticals, Inc. | Gip analog and hybrid polypeptides with selectable properties |
| WO2006097521A1 (en) | 2005-03-18 | 2006-09-21 | Novo Nordisk A/S | Pegylated single-chain insulin |
| WO2006097536A2 (en) * | 2005-03-18 | 2006-09-21 | Novo Nordisk A/S | Dimeric peptide agonists of the glp-1 receptor |
| US7521211B2 (en) | 2005-04-05 | 2009-04-21 | Regeneron Pharmaceuticals, Inc | IGF-1 and IGF-2 chimeric polypeptides and therapeutic uses thereof |
| US20090209453A1 (en) | 2005-04-13 | 2009-08-20 | University Of Medicine And Dentistry Of New Jersey | Glycoprotein Hormone Analogs |
| US20070099841A1 (en) | 2005-09-08 | 2007-05-03 | New River Pharmaceuticals Inc. | Prodrugs of T3 and T4 with enhanced bioavailability |
| ES2507098T3 (en) * | 2005-11-07 | 2014-10-14 | Indiana University Research And Technology Corporation | Glucagon analogs showing physiological solubility and stability |
| US8304386B2 (en) | 2006-02-03 | 2012-11-06 | Prolor Biotech, Inc. | Long-acting growth hormone and methods of producing same |
| US20090069216A1 (en) | 2006-02-21 | 2009-03-12 | Novo Nordisk A/S | Single-Chain Insulin Analogues and Pharmaceutical Formulations Thereof |
| WO2007104786A1 (en) * | 2006-03-15 | 2007-09-20 | Novo Nordisk A/S | Mixtures of amylin and insulin |
| AU2007227202B2 (en) | 2006-03-21 | 2013-08-22 | Amylin Pharmaceuticals, Llc | Peptide-peptidase inhibitor conjugates and methods of using same |
| US9505823B2 (en) | 2006-08-07 | 2016-11-29 | TEV A Biopharmaceuticals USA, Inc. | Albumin-insulin fusion proteins |
| JP5399244B2 (en) | 2006-08-17 | 2014-01-29 | アミリン・ファーマシューティカルズ,リミテッド・ライアビリティ・カンパニー | DPP-IV resistant GIP hybrid polypeptide with selectable properties |
| CL2007002502A1 (en) | 2006-08-31 | 2008-05-30 | Hoffmann La Roche | VARIANTS OF THE SIMILAR GROWTH FACTOR TO HUMAN INSULIN-1 (IGF-1) PEGILATED IN LISIN; METHOD OF PRODUCTION; FUSION PROTEIN THAT UNDERSTANDS IT; AND ITS USE TO TREAT ALZHEIMER'S DISEASE. |
| AU2007292903B2 (en) * | 2006-09-08 | 2012-03-29 | Ambrx, Inc. | Modified human plasma polypeptide or Fc scaffolds and their uses |
| DE102006052755A1 (en) | 2006-11-08 | 2008-05-15 | N-Zyme Biotec Gmbh | New peptide derivatives and peptidomimetics are transglutaminase inhibitors useful for the treatment or prophylaxis of e.g. celiac disease, fibrosis, thrombosis, neurodegenerative disorders, Huntington's chorea and Parkinson disease |
| CN101578102B (en) * | 2007-01-05 | 2013-07-17 | 印第安纳大学研究及科技有限公司 | Glucagon analogs exhibiting enhanced solubility in physiological pH buffers |
| US20090098130A1 (en) | 2007-01-05 | 2009-04-16 | Bradshaw Curt W | Glucagon-like protein-1 receptor (glp-1r) agonist compounds |
| CA2677932A1 (en) * | 2007-02-15 | 2008-08-21 | Indiana University Research And Technology Corporation | Glucagon/glp-1 receptor co-agonists |
| US7859468B2 (en) * | 2007-08-30 | 2010-12-28 | Research In Motion Limited | Mobile wireless communications device including a folded monopole multi-band antenna and related methods |
| EP2036923A1 (en) | 2007-09-11 | 2009-03-18 | Novo Nordisk A/S | Improved derivates of amylin |
| EP2036539A1 (en) | 2007-09-11 | 2009-03-18 | Novo Nordisk A/S | Stable formulations of amylin and its analogues |
| MX2010004298A (en) * | 2007-10-30 | 2010-05-03 | Univ Indiana Res & Tech Corp | Compounds exhibiting glucagon antagonist and glp-1 agonist activity. |
| US8946148B2 (en) | 2007-11-20 | 2015-02-03 | Ambrx, Inc. | Modified insulin polypeptides and their uses |
| CN101983066B (en) | 2008-01-30 | 2016-06-29 | 印第安那大学科技研究公司 | Insulin prodrug based on ester |
| AU2009209565B2 (en) | 2008-02-01 | 2013-09-19 | Ascendis Pharma As | Prodrug comprising a self-cleavable linker |
| AU2009260301B2 (en) * | 2008-06-17 | 2015-09-03 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility and stability in physiological pH buffers |
| AU2009260302B2 (en) * | 2008-06-17 | 2014-10-23 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| CA2729296A1 (en) * | 2008-06-17 | 2010-01-28 | Richard D. Dimarchi | Gip-based mixed agonists for treatment of metabolic disorders and obesity |
| EP2356139A4 (en) | 2008-07-23 | 2013-01-09 | Harvard College | Ligation of stapled polypeptides |
| MX2011006527A (en) | 2008-12-19 | 2011-08-17 | Univ Indiana Res & Tech Corp | Dipeptide linked medicinal agents. |
| EP2376520B1 (en) | 2008-12-19 | 2014-02-12 | Indiana University Research&Technology Corporation | Insulin analogs |
| EP2376521B1 (en) | 2008-12-19 | 2016-04-13 | Indiana University Research and Technology Corporation | Amide-based insulin prodrugs |
| US8969288B2 (en) * | 2008-12-19 | 2015-03-03 | Indiana University Research And Technology Corporation | Amide based glucagon and superfamily peptide prodrugs |
| US8569231B2 (en) | 2009-03-20 | 2013-10-29 | Smartcells, Inc. | Soluble non-depot insulin conjugates and uses thereof |
| WO2011159895A2 (en) | 2010-06-16 | 2011-12-22 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| AU2011270832B2 (en) | 2010-06-24 | 2016-05-12 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| WO2011163460A1 (en) | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Yl-based insulin-like growth factors exhibiting high activity at the insulin receptor |
| MX2012014576A (en) | 2010-06-24 | 2013-02-21 | Univ Indiana Res & Tech Corp | Amide based glucagon superfamily peptide prodrugs. |
-
2009
- 2009-01-21 CN CN200980112047.4A patent/CN101983066B/en not_active Expired - Fee Related
- 2009-01-21 EP EP09708686A patent/EP2249853A4/en not_active Withdrawn
- 2009-01-21 JP JP2010545058A patent/JP2011511778A/en active Pending
- 2009-01-21 WO PCT/US2009/031593 patent/WO2009099763A1/en active Application Filing
- 2009-01-21 US US12/865,058 patent/US20110065633A1/en not_active Abandoned
- 2009-01-21 CA CA2713348A patent/CA2713348C/en not_active Expired - Fee Related
- 2009-01-21 AU AU2009210570A patent/AU2009210570B2/en not_active Ceased
- 2009-01-23 TW TW098103051A patent/TW200938225A/en unknown
- 2009-01-29 PE PE2009000119A patent/PE20091440A1/en not_active Application Discontinuation
- 2009-01-29 PA PA20098814001A patent/PA8814001A1/en unknown
- 2009-01-30 CL CL2009000202A patent/CL2009000202A1/en unknown
- 2009-01-30 AR ARP090100301A patent/AR073335A1/en unknown
-
2010
- 2010-07-28 US US12/845,455 patent/US8697838B2/en not_active Expired - Fee Related
-
2014
- 2014-02-18 US US14/182,706 patent/US9089539B2/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060171920A1 (en) * | 2003-04-08 | 2006-08-03 | Yeda Research And Development Co., Ltd. | Reversible pegylated drugs |
| US20070203058A1 (en) * | 2003-09-19 | 2007-08-30 | Novo Nordisk A/S | Novel Glp-1 Derivatives |
Non-Patent Citations (3)
| Title |
|---|
| DATABASE GENBANK [online] 15 July 2006 (2006-07-15), XP008140416, Database accession no. AAH05278 * |
| DE.: "Design of peptide-based prodrug chemistry and its application to glucagon-like peptdie I.", MASTERS THESIS, August 2007 (2007-08-01), XP008143583, Retrieved from the Internet <URL:scholarworks.iu.edu/dspace/browse?value=De%2C+ArnabBtype=author> [retrieved on 20090618] * |
| See also references of EP2249853A4 * |
Cited By (165)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9018164B2 (en) | 2005-11-07 | 2015-04-28 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting physiological solubility and stability |
| US8338368B2 (en) | 2005-11-07 | 2012-12-25 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting physiological solubility and stability |
| US8669228B2 (en) | 2007-01-05 | 2014-03-11 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility in physiological pH buffers |
| US8900593B2 (en) | 2007-02-15 | 2014-12-02 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8454971B2 (en) | 2007-02-15 | 2013-06-04 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US9447162B2 (en) | 2007-02-15 | 2016-09-20 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8980830B2 (en) | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Peptide compounds exhibiting glucagon antagonist and GLP-1 agonist activity |
| US8981047B2 (en) | 2007-10-30 | 2015-03-17 | Indiana University Research And Technology Corporation | Glucagon antagonists |
| US9089539B2 (en) | 2008-01-30 | 2015-07-28 | Indiana University Research And Technology Corporation | Ester-based insulin prodrugs |
| US8697838B2 (en) | 2008-01-30 | 2014-04-15 | Indiana University Research And Technology Corporation | Ester-based insulin prodrugs |
| US8546327B2 (en) | 2008-06-17 | 2013-10-01 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8969294B2 (en) | 2008-06-17 | 2015-03-03 | Istituto Di Recerche Di Biologia Molecolare P. Angeletti S.R.L. | Glucagon/GLP-1 receptor co-agonists |
| US9062124B2 (en) | 2008-06-17 | 2015-06-23 | Indiana University Research And Technology Corporation | GIP-based mixed agonists for treatment of metabolic disorders and obesity |
| US8450270B2 (en) | 2008-06-17 | 2013-05-28 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting enhanced solubility and stability in physiological pH buffers |
| EP2376521A4 (en) * | 2008-12-19 | 2014-06-25 | Univ Indiana Res & Tech Corp | Amide-based insulin prodrugs |
| US8697632B2 (en) | 2008-12-19 | 2014-04-15 | Indiana University Research And Technology Corporation | Amide based insulin prodrugs |
| RU2578591C2 (en) * | 2008-12-19 | 2016-03-27 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Dipeptide linked medicinal agents |
| WO2010080609A1 (en) | 2008-12-19 | 2010-07-15 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| US8481485B2 (en) | 2008-12-19 | 2013-07-09 | Indiana University Research And Technology Corporation | Insulin analogs |
| EP2376521A1 (en) * | 2008-12-19 | 2011-10-19 | Indiana University Research and Technology Corporation | Amide-based insulin prodrugs |
| RU2550696C2 (en) * | 2008-12-19 | 2015-05-10 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Amide-based prodrugs of glucagon superfamily peptides |
| US8969288B2 (en) | 2008-12-19 | 2015-03-03 | Indiana University Research And Technology Corporation | Amide based glucagon and superfamily peptide prodrugs |
| US9150632B2 (en) | 2009-06-16 | 2015-10-06 | Indiana University Research And Technology Corporation | GIP receptor-active glucagon compounds |
| US9790263B2 (en) | 2009-06-16 | 2017-10-17 | Indiana University Research And Technology Corporation | GIP receptor-active glucagon compounds |
| US8703701B2 (en) | 2009-12-18 | 2014-04-22 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US9062094B2 (en) | 2010-01-22 | 2015-06-23 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aliphatic amine-containing drugs |
| WO2011089216A1 (en) | 2010-01-22 | 2011-07-28 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aliphatic amine-containing drugs |
| WO2011089215A1 (en) | 2010-01-22 | 2011-07-28 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aromatic amine-containing drugs |
| US9533056B2 (en) | 2010-01-22 | 2017-01-03 | Ascendis Pharma As | Dipeptide-based prodrug linkers for aliphatic amine-containing drugs |
| US9487571B2 (en) | 2010-01-27 | 2016-11-08 | Indiana University Research And Technology Corporation | Glucagon antagonist-GIP agonist conjugates and compositions for the treatment of metabolic disorders and obesity |
| EP2528618A4 (en) * | 2010-01-27 | 2015-05-27 | Univ Indiana Res & Tech Corp | Glucagon antagonist - gip agonist conjugates and compositions for the treatment of metabolic disorders and obesity |
| WO2011143209A1 (en) | 2010-05-13 | 2011-11-17 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| US9783592B2 (en) | 2010-05-13 | 2017-10-10 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| US9127088B2 (en) | 2010-05-13 | 2015-09-08 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting nuclear hormone receptor activity |
| US9145451B2 (en) | 2010-05-13 | 2015-09-29 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhbiting G protein coupled receptor activity |
| WO2011159895A3 (en) * | 2010-06-16 | 2012-05-18 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| EP2582719A2 (en) * | 2010-06-16 | 2013-04-24 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| US9458220B2 (en) | 2010-06-16 | 2016-10-04 | Indiana University Research And Technology Corporation | Single-chain insulin agonists exhibiting high activity at the insulin receptor |
| JP2013533864A (en) * | 2010-06-16 | 2013-08-29 | インディアナ ユニバーシティー リサーチ アンド テクノロジー コーポレーション | Single-chain insulin agonist with high activity for insulin receptor |
| US10233225B2 (en) | 2010-06-16 | 2019-03-19 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| WO2011159895A2 (en) | 2010-06-16 | 2011-12-22 | Indiana University Research And Technology Corporation | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| US8940860B2 (en) | 2010-06-16 | 2015-01-27 | Indiana University Research And Technology Corporation | Single-chain insulin agonists exhibiting high activity at the insulin receptor |
| EP2582719A4 (en) * | 2010-06-16 | 2013-12-04 | Univ Indiana Res & Tech Corp | Single chain insulin agonists exhibiting high activity at the insulin receptor |
| US8778872B2 (en) | 2010-06-24 | 2014-07-15 | Indiana University Research And Technology Corporation | Amide based glucagon superfamily peptide prodrugs |
| CN103179979A (en) * | 2010-06-24 | 2013-06-26 | 印第安纳大学研究及科技有限公司 | Amide based glucagon superfamily peptide prodrugs |
| RU2580317C2 (en) * | 2010-06-24 | 2016-04-10 | Индиана Юниверсити Рисерч Энд Текнолоджи Корпорейшн | Peptide pro-drugs of amide glucagon superfamily |
| US8946147B2 (en) | 2010-06-24 | 2015-02-03 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| CN103119057A (en) * | 2010-06-24 | 2013-05-22 | 印第安纳大学研究及科技有限公司 | Amide-based insulin prodrugs |
| CN103119057B (en) * | 2010-06-24 | 2016-06-15 | 印第安纳大学研究及科技有限公司 | Insulin prodrug based on amide |
| CN102958533B (en) * | 2010-06-24 | 2016-01-20 | 印第安纳大学研究及科技有限公司 | The medicament that dipeptides connects |
| WO2011162968A1 (en) * | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Dipeptide linked medicinal agents |
| WO2011163462A2 (en) | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| WO2011163460A1 (en) * | 2010-06-24 | 2011-12-29 | Indiana University Research And Technology Corporation | Yl-based insulin-like growth factors exhibiting high activity at the insulin receptor |
| JP2013540102A (en) * | 2010-06-24 | 2013-10-31 | インディアナ ユニバーシティー リサーチ アンド テクノロジー コーポレーション | Peptide prodrugs of the glucagon superfamily modified via amide bonds |
| CN102958533A (en) * | 2010-06-24 | 2013-03-06 | 印第安纳大学研究及科技有限公司 | Dipeptide linked medicinal agents |
| AU2011270832B2 (en) * | 2010-06-24 | 2016-05-12 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| WO2011163012A3 (en) * | 2010-06-24 | 2013-07-11 | Indiana University Research And Technology Corporation | Amide based glucagon superfamily peptide prodrugs |
| WO2011163462A3 (en) * | 2010-06-24 | 2012-04-05 | Indiana University Research And Technology Corporation | Amide-based insulin prodrugs |
| EP2588126A4 (en) * | 2010-06-24 | 2015-07-08 | Univ Indiana Res & Tech Corp | Amide based glucagon superfamily peptide prodrugs |
| JP2013531660A (en) * | 2010-06-24 | 2013-08-08 | インディアナ ユニバーシティー リサーチ アンド テクノロジー コーポレーション | Amide insulin prodrug |
| US9023986B2 (en) | 2010-10-25 | 2015-05-05 | Hoffmann-La Roche Inc. | Glucose-dependent insulinotropic peptide analogs |
| WO2012059764A1 (en) | 2010-11-03 | 2012-05-10 | Arecor Limited | Novel composition comprising glucagon |
| WO2012059762A1 (en) | 2010-11-03 | 2012-05-10 | Arecor Limited | Novel composition comprising glucagon |
| US8507428B2 (en) | 2010-12-22 | 2013-08-13 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| US9249206B2 (en) | 2010-12-22 | 2016-02-02 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| US8975223B2 (en) | 2010-12-22 | 2015-03-10 | Marcadia Biotech, Inc. | Methods for treating metabolic disorders and obesity with a peptide comprising the amino acid sequence of SEQ ID No. 146 |
| US9522946B2 (en) | 2011-06-10 | 2016-12-20 | Hanmi Science Co., Ltd. | Oxyntomodulin derivatives and pharmaceutical composition for treating obesity comprising the same |
| US9765131B2 (en) | 2011-06-10 | 2017-09-19 | Hanmi Science Co., Ltd. | Oxyntomodulin derivatives and pharmaceutical composition for treating obesity comprising the same |
| US9527898B2 (en) | 2011-06-10 | 2016-12-27 | Hanmi Science Co., Ltd. | Oxyntomodulin derivatives and pharmaceutical composition for treating obesity comprising the same |
| US10442848B2 (en) | 2011-06-10 | 2019-10-15 | Hanmi Science Co., Ltd. | Oxyntomodulin derivatives and pharmaceutical composition for treating obesity comprising the same |
| US10363320B2 (en) | 2011-06-17 | 2019-07-30 | Hanmi Science Co., Ltd. | Conjugate comprising oxyntomodulin and an immunoglobulin fragment, and use thereof |
| US9731031B2 (en) | 2011-06-17 | 2017-08-15 | Hanmi Science Co., Ltd. | Conjugate comprising oxyntomodulin and an immunoglobulin fragment, and use thereof |
| US11872283B2 (en) | 2011-06-17 | 2024-01-16 | Hanmi Science Co., Ltd | Conjugate comprising oxyntomodulin and an immunoglobulin fragment, and use thereof |
| US9758562B2 (en) | 2011-06-22 | 2017-09-12 | Indiana University and Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US9309301B2 (en) | 2011-06-22 | 2016-04-12 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US8729017B2 (en) | 2011-06-22 | 2014-05-20 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US9156902B2 (en) | 2011-06-22 | 2015-10-13 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US10730923B2 (en) | 2011-06-22 | 2020-08-04 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US10174093B2 (en) | 2011-06-22 | 2019-01-08 | Indiana University Research And Technology Corporation | Glucagon/GLP-1 receptor co-agonists |
| US9944687B2 (en) | 2011-07-04 | 2018-04-17 | Imperial Innovations Limited | Compounds and their effects on feeding behaviour |
| EP3643306A3 (en) * | 2011-08-12 | 2020-08-26 | Ascendis Pharma A/S | Carrier-linked prodrugs having reversible carboxylic ester linkages |
| EP2741744A1 (en) * | 2011-08-12 | 2014-06-18 | Ascendis Pharma A/S | Carrier-linked prodrugs having reversible carboxylic ester linkages |
| US9486505B2 (en) | 2011-09-23 | 2016-11-08 | Novo Nordisk A/S | Glucagon analogues |
| US8541368B2 (en) | 2011-09-23 | 2013-09-24 | Novo Nordisk A/S | Glucagon analogues |
| US11434271B2 (en) | 2011-11-04 | 2022-09-06 | Hanmi Science Co., Ltd. | Method for preparing physiologically active polypeptide complex |
| WO2013074910A1 (en) | 2011-11-17 | 2013-05-23 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting glucocorticoid receptor activity |
| US8859491B2 (en) | 2011-11-17 | 2014-10-14 | Indiana University Research And Technology Corporation | Glucagon superfamily peptides exhibiting glucocorticoid receptor activity |
| US9573987B2 (en) | 2011-12-20 | 2017-02-21 | Indiana University Research And Technology Corporation | CTP-based insulin analogs for treatment of diabetes |
| WO2013096386A1 (en) | 2011-12-20 | 2013-06-27 | Indiana University Research And Technology Corporation | Ctp-based insulin analogs for treatment of diabetes |
| WO2013127779A1 (en) * | 2012-03-01 | 2013-09-06 | Novo Nordisk A/S | Glp-1 prodrugs |
| US9452225B2 (en) | 2012-03-01 | 2016-09-27 | Novo Nordisk A/S | GLP-1 prodrugs |
| US9340600B2 (en) | 2012-06-21 | 2016-05-17 | Indiana University Research And Technology Corporation | Glucagon analogs exhibiting GIP receptor activity |
| US10493132B2 (en) | 2012-07-25 | 2019-12-03 | Hanmi Pharm. Co., Ltd. | Composition for treating hyperlipidemia comprising oxyntomodulin derivative |
| US9901621B2 (en) | 2012-07-25 | 2018-02-27 | Hanmi Pharm. Co., Ltd. | Composition for treating hyperlipidemia comprising oxyntomodulin derivative |
| EP3395358A1 (en) | 2012-09-26 | 2018-10-31 | Indiana University Research and Technology Corporation | Insulin analog dimers |
| US9593156B2 (en) | 2012-09-26 | 2017-03-14 | Indiana University Research And Technology Corporation | Insulin analog dimers |
| WO2014052451A2 (en) | 2012-09-26 | 2014-04-03 | Indiana University Research And Technology Corporation | Insulin analog dimers |
| US10758592B2 (en) | 2012-10-09 | 2020-09-01 | Sanofi | Exendin-4 derivatives as dual GLP1/glucagon agonists |
| US9365632B2 (en) | 2012-10-09 | 2016-06-14 | Sanofi | Exendin-4 derivatives as dual GLP1/glucagon agonists |
| US10279041B2 (en) | 2012-11-06 | 2019-05-07 | Hanmi Pharm Co. Ltd. | Liquid formulation of long-lasting protein conjugate comprising the oxyntomodulin and an immunoglobulin fragment |
| US10550168B2 (en) | 2012-11-06 | 2020-02-04 | Hanmi Pharm. Co., Ltd. | Composition for treating diabetes or diabesity comprising oxyntomodulin analog |
| US9724420B2 (en) | 2012-11-06 | 2017-08-08 | Hanmi Pharm. Co., Ltd. | Liquid formulation of protein conjugate comprising an oxyntomodulin derivative covalently linked to a non-peptidyl polymer to an immunoglobulin FC region |
| US11071785B2 (en) | 2012-11-06 | 2021-07-27 | Hanmi Pharm. Co., Ltd. | Liquid formulation of long-lasting protein conjugate comprising the oxyntomodulin and an immunoglobulin Fc region |
| WO2014088836A1 (en) | 2012-12-03 | 2014-06-12 | Merck Sharp & Dohme Corp. | O-glycosylated carboxy terminal portion (ctp) peptide-based insulin and insulin analogues |
| WO2014099577A1 (en) | 2012-12-17 | 2014-06-26 | Merck Sharp & Dohme Corp. | Process for purifying insulin and analogues thereof |
| US10253079B2 (en) | 2012-12-21 | 2019-04-09 | Sanofi | Functionalized Exendin-4 derivatives |
| US9745360B2 (en) | 2012-12-21 | 2017-08-29 | Sanofi | Dual GLP1/GIP or trigonal GLP1/GIP/glucagon agonists |
| US9670261B2 (en) | 2012-12-21 | 2017-06-06 | Sanofi | Functionalized exendin-4 derivatives |
| US10696726B2 (en) | 2013-03-14 | 2020-06-30 | Indiana University Research And Technology Corporation | Insulin-incretin conjugates |
| US9751927B2 (en) | 2013-04-18 | 2017-09-05 | Novo Nordisk A/S | Stable, protracted GLP-1/glucagon receptor co-agonists for medical use |
| US9474790B2 (en) | 2013-04-18 | 2016-10-25 | Novo Nordisk A/S | Stable, protracted GLP-1/glucagon receptor co-agonists for medical use |
| WO2015051052A2 (en) | 2013-10-04 | 2015-04-09 | Merck Sharp & Dohme Corp. | Glucose-responsive insulin conjugates |
| WO2015084694A2 (en) | 2013-12-04 | 2015-06-11 | Merck Sharp & Dohme Corp. | Method for preparing crystalline insulin |
| US9751926B2 (en) | 2013-12-13 | 2017-09-05 | Sanofi | Dual GLP-1/GIP receptor agonists |
| US9750788B2 (en) | 2013-12-13 | 2017-09-05 | Sanofi | Non-acylated exendin-4 peptide analogues |
| US9789165B2 (en) | 2013-12-13 | 2017-10-17 | Sanofi | Exendin-4 peptide analogues as dual GLP-1/GIP receptor agonists |
| US9694053B2 (en) | 2013-12-13 | 2017-07-04 | Sanofi | Dual GLP-1/glucagon receptor agonists |
| AU2014366425B2 (en) * | 2013-12-18 | 2016-12-08 | Eli Lilly And Company | Novel compound for treatment of severe hypoglycemia |
| US9688736B2 (en) | 2013-12-18 | 2017-06-27 | Eli Lilly And Company | Compound for treatment of severe hypoglycemia |
| WO2015094876A1 (en) * | 2013-12-18 | 2015-06-25 | Eli Lilly And Company | Novel compound for treatment of severe hypoglycemia |
| CN105829340A (en) * | 2013-12-18 | 2016-08-03 | 伊莱利利公司 | Novel compound for treatment of severe hypoglycemia |
| WO2015138548A1 (en) | 2014-03-14 | 2015-09-17 | Merck Sharp & Dohme Corp. | Purifying insulin using cation exchange and reverse phase chromatography in the presence of an organic modifier and elevated temperature |
| EP3889185A3 (en) * | 2014-03-31 | 2022-01-26 | Hanmi Pharm. Co., Ltd. | Composition for improving the solubility of a protein or peptide by using immunoglobulin fc fragment linkage |
| US9758561B2 (en) | 2014-04-07 | 2017-09-12 | Sanofi | Dual GLP-1/glucagon receptor agonists derived from exendin-4 |
| US9771406B2 (en) | 2014-04-07 | 2017-09-26 | Sanofi | Peptidic dual GLP-1/glucagon receptor agonists derived from exendin-4 |
| US9775904B2 (en) | 2014-04-07 | 2017-10-03 | Sanofi | Exendin-4 derivatives as peptidic dual GLP-1/glucagon receptor agonists |
| US10570184B2 (en) | 2014-06-04 | 2020-02-25 | Novo Nordisk A/S | GLP-1/glucagon receptor co-agonists for medical use |
| US9932381B2 (en) | 2014-06-18 | 2018-04-03 | Sanofi | Exendin-4 derivatives as selective glucagon receptor agonists |
| WO2016014325A1 (en) | 2014-07-21 | 2016-01-28 | Merck Sharp & Dohme Corp. | Chromatography process for purification of inlsulin and insulin analogs |
| US10233230B2 (en) | 2014-09-16 | 2019-03-19 | Hanmi Pharm. Co., Ltd. | Use of a long acting GLP-1/glucagon receptor dual agonist for the treatment of non-alcoholic fatty liver disease |
| US10435459B2 (en) | 2014-09-16 | 2019-10-08 | Hanmi Pharm. Co., Ltd. | Use of a long acting GLP-1/glucagon receptor dual agonist for the treatment of non-alcoholic fatty liver disease |
| US10232020B2 (en) | 2014-09-24 | 2019-03-19 | Indiana University Research And Technology Corporation | Incretin-insulin conjugates |
| US10385107B2 (en) | 2014-09-24 | 2019-08-20 | Indiana Univeresity Researc and Technology Corporation | Lipidated amide-based insulin prodrugs |
| US10413593B2 (en) | 2014-10-24 | 2019-09-17 | Merck Sharp & Dohme Corp. | Co-agonists of the glucagon and GLP-1 receptors |
| EP3666792A2 (en) | 2014-11-21 | 2020-06-17 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| EP3660040A2 (en) | 2014-11-21 | 2020-06-03 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| EP3660041A1 (en) | 2014-11-21 | 2020-06-03 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| WO2016081670A2 (en) | 2014-11-21 | 2016-05-26 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| US11254724B2 (en) | 2014-12-30 | 2022-02-22 | Hanmi Pharm. Co., Ltd. | Glucagon derivatives |
| US11135271B2 (en) | 2014-12-30 | 2021-10-05 | Hanmi Pharm. Co., Ltd. | Glucagon derivatives with improved stability |
| WO2016144658A1 (en) | 2015-03-10 | 2016-09-15 | Merck Sharp & Dohme Corp. | Process for preparing recombinant insulin using microfiltration |
| US10806797B2 (en) | 2015-06-05 | 2020-10-20 | Sanofi | Prodrugs comprising an GLP-1/glucagon dual agonist linker hyaluronic acid conjugate |
| US11667688B2 (en) | 2015-06-30 | 2023-06-06 | Hanmi Pharm. Co., Ltd. | Glucagon derivative and a composition comprising a long acting conjugate of the same |
| US10696725B2 (en) | 2015-06-30 | 2020-06-30 | Hanmi Pharm. Co., Ltd. | Glucagon derivative and a composition comprising a long acting conjugate of the same |
| US11261227B2 (en) | 2015-06-30 | 2022-03-01 | Hanmi Pharm. Co., Ltd. | Glucagon derivative and a composition comprising a long acting conjugate of the same |
| US9982029B2 (en) | 2015-07-10 | 2018-05-29 | Sanofi | Exendin-4 derivatives as selective peptidic dual GLP-1/glucagon receptor agonists |
| WO2017040363A1 (en) | 2015-09-02 | 2017-03-09 | Merck Sharp & Dohme Corp. | A process for obtaining insulin with correctly formed disulfide bonds |
| US9884093B2 (en) | 2015-10-26 | 2018-02-06 | Eli Lilly And Company | Glucagon receptor agonists |
| US9764004B2 (en) | 2015-10-26 | 2017-09-19 | Eli Lilly And Company | Glucagon receptor agonists |
| US10981967B2 (en) | 2015-12-31 | 2021-04-20 | Hanmi Pharm. Co., Ltd. | Long-acting conjugate of triple glucagon/GLP-1/GIP receptor agonist |
| US11332508B2 (en) | 2015-12-31 | 2022-05-17 | Hanmi Pharm. Co., Ltd. | Triple glucagon/GLP-1/GIP receptor agonist |
| US11058775B2 (en) | 2016-04-26 | 2021-07-13 | Merck Sharp & Dohme Corp. | Insulin dimer-incretin conjugates |
| WO2017189342A1 (en) | 2016-04-26 | 2017-11-02 | Merck Sharp & Dohme Corp. | Insulin dimer-incretin conjugates |
| WO2017192449A1 (en) * | 2016-05-06 | 2017-11-09 | Phasebio Pharmaceuticals, Inc. | Elp fusion proteins for controlled and sustained release |
| US10953076B2 (en) | 2016-05-24 | 2021-03-23 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists and GLP-1 analogues |
| EP3922260A2 (en) | 2016-05-24 | 2021-12-15 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists and glp-1 analogues |
| US10689430B2 (en) | 2016-05-25 | 2020-06-23 | Merck Sharp & Dohme Corp. | Insulin receptor partial agonists |
| WO2017221026A1 (en) | 2016-06-24 | 2017-12-28 | Arecor Limited | Novel composition |
| US11142559B2 (en) | 2016-06-29 | 2021-10-12 | Hanmi Pharm. Co., Ltd. | Glucagon derivative, conjugate thereof, composition comprising same, and therapeutic use thereof |
| EP3272877A1 (en) | 2016-07-18 | 2018-01-24 | ETH Zurich | B-cell-mimetic cells |
| WO2018015330A1 (en) | 2016-07-18 | 2018-01-25 | Eth Zurich | B-cell-mimetic cells |
| EP4310082A2 (en) | 2017-01-20 | 2024-01-24 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| US11041009B2 (en) | 2017-03-23 | 2021-06-22 | Merck Sharp & Dohme Corp. | Glucose responsive insulin comprising a tri-valent sugar cluster for treatment of diabetes |
| WO2018213377A1 (en) | 2017-05-17 | 2018-11-22 | Arcus Biosciences, Inc. | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
| WO2021188769A1 (en) | 2020-03-19 | 2021-09-23 | Arcus Biosciences, Inc. | Tetralin and tetrahydroquinoline compounds as inhibitors of hif-2alpha |
Also Published As
| Publication number | Publication date |
|---|---|
| PE20091440A1 (en) | 2009-09-20 |
| CA2713348C (en) | 2018-10-09 |
| US9089539B2 (en) | 2015-07-28 |
| CN101983066A (en) | 2011-03-02 |
| CN101983066B (en) | 2016-06-29 |
| PA8814001A1 (en) | 2009-09-17 |
| EP2249853A4 (en) | 2012-12-26 |
| JP2011511778A (en) | 2011-04-14 |
| AU2009210570A1 (en) | 2009-08-13 |
| EP2249853A1 (en) | 2010-11-17 |
| US20110065633A1 (en) | 2011-03-17 |
| US8697838B2 (en) | 2014-04-15 |
| US20100331246A1 (en) | 2010-12-30 |
| CA2713348A1 (en) | 2009-08-13 |
| CL2009000202A1 (en) | 2009-05-22 |
| AU2009210570B2 (en) | 2014-11-20 |
| TW200938225A (en) | 2009-09-16 |
| US20140212419A1 (en) | 2014-07-31 |
| AR073335A1 (en) | 2010-11-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9089539B2 (en) | Ester-based insulin prodrugs | |
| US8969288B2 (en) | Amide based glucagon and superfamily peptide prodrugs | |
| US8778872B2 (en) | Amide based glucagon superfamily peptide prodrugs | |
| EP3206710B1 (en) | Incretin-insulin conjugates | |
| US8980830B2 (en) | Peptide compounds exhibiting glucagon antagonist and GLP-1 agonist activity | |
| EP2217701B1 (en) | Glucagon antagonists | |
| US9487571B2 (en) | Glucagon antagonist-GIP agonist conjugates and compositions for the treatment of metabolic disorders and obesity | |
| CA2802485C (en) | Single chain insulin agonists exhibiting high activity at the insulin receptor | |
| US20160058881A1 (en) | Prodrugs with prolonged action |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200980112047.4 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 09708686 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009210570 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2713348 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2009210570 Country of ref document: AU Date of ref document: 20090121 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12865058 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010545058 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 5486/DELNP/2010 Country of ref document: IN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REEP | Request for entry into the european phase |
Ref document number: 2009708686 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009708686 Country of ref document: EP |