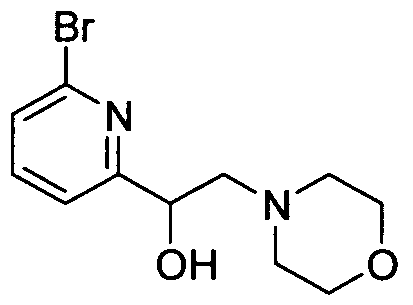
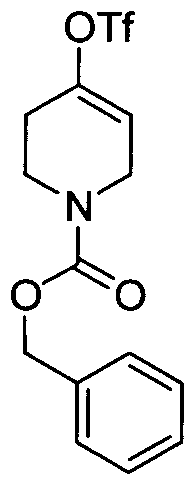
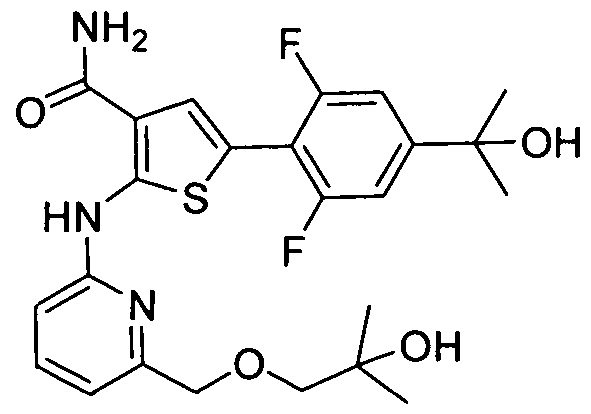
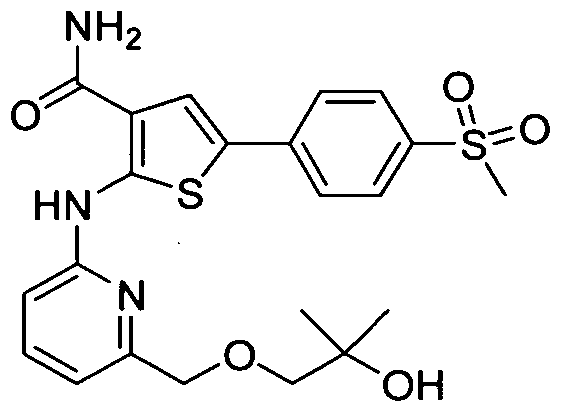
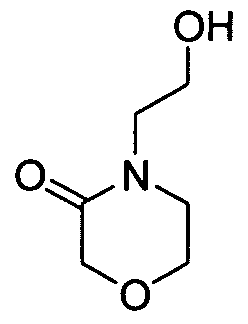
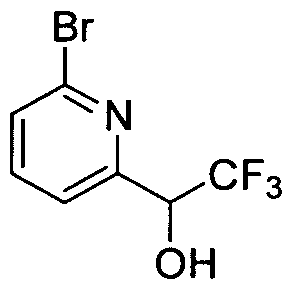
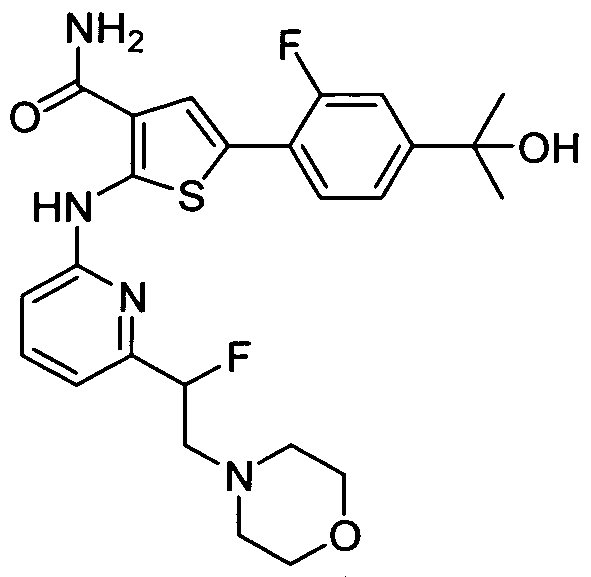
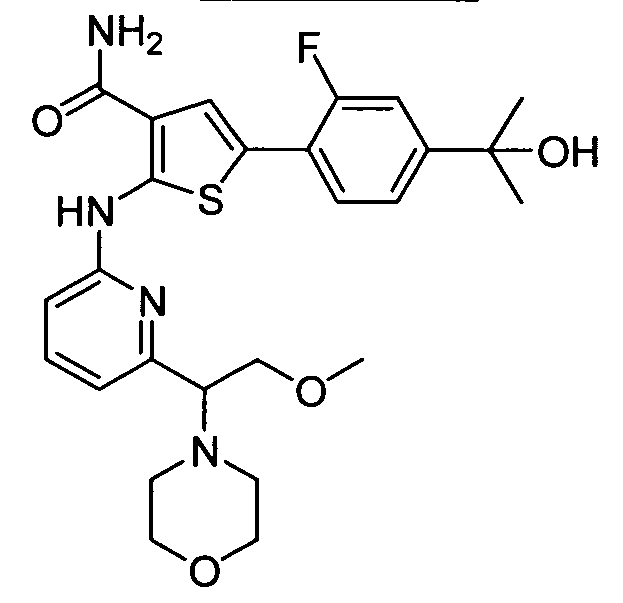
WO2008156726A1 - Inhibitors of janus kinases - Google Patents
Inhibitors of janus kinases Download PDFInfo
- Publication number
- WO2008156726A1 WO2008156726A1 PCT/US2008/007486 US2008007486W WO2008156726A1 WO 2008156726 A1 WO2008156726 A1 WO 2008156726A1 US 2008007486 W US2008007486 W US 2008007486W WO 2008156726 A1 WO2008156726 A1 WO 2008156726A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino
- carboxamide
- hydroxy
- thiophene
- phenyl
- Prior art date
Links
- 108010024121 Janus Kinases Proteins 0.000 title abstract description 8
- 102000015617 Janus Kinases Human genes 0.000 title abstract description 8
- 239000003112 inhibitor Substances 0.000 title description 63
- 150000001875 compounds Chemical class 0.000 claims abstract description 245
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 49
- 201000011510 cancer Diseases 0.000 claims abstract description 32
- 208000014767 Myeloproliferative disease Diseases 0.000 claims abstract description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 408
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 272
- DAUYIKBTMNZABP-UHFFFAOYSA-N thiophene-3-carboxamide Chemical compound NC(=O)C=1C=CSC=1 DAUYIKBTMNZABP-UHFFFAOYSA-N 0.000 claims description 222
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 claims description 192
- -1 trimethylsilylethoxy Chemical group 0.000 claims description 136
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 125
- 125000000217 alkyl group Chemical group 0.000 claims description 82
- 150000003839 salts Chemical class 0.000 claims description 45
- 125000005843 halogen group Chemical group 0.000 claims description 37
- 125000001424 substituent group Chemical group 0.000 claims description 37
- 125000003118 aryl group Chemical group 0.000 claims description 34
- 229910052739 hydrogen Inorganic materials 0.000 claims description 33
- 239000001257 hydrogen Substances 0.000 claims description 33
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 claims description 31
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 30
- 239000003814 drug Substances 0.000 claims description 27
- 229910052799 carbon Inorganic materials 0.000 claims description 25
- 125000000623 heterocyclic group Chemical group 0.000 claims description 23
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 23
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 20
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 19
- 125000001072 heteroaryl group Chemical group 0.000 claims description 19
- 125000005842 heteroatom Chemical group 0.000 claims description 17
- 239000008194 pharmaceutical composition Substances 0.000 claims description 16
- 125000003821 2-(trimethylsilyl)ethoxymethyl group Chemical group [H]C([H])([H])[Si](C([H])([H])[H])(C([H])([H])[H])C([H])([H])C(OC([H])([H])[*])([H])[H] 0.000 claims description 14
- 241000124008 Mammalia Species 0.000 claims description 14
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 14
- IBBMAWULFFBRKK-UHFFFAOYSA-N picolinamide Chemical compound NC(=O)C1=CC=CC=N1 IBBMAWULFFBRKK-UHFFFAOYSA-N 0.000 claims description 14
- 238000002360 preparation method Methods 0.000 claims description 14
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 claims description 13
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical group CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 13
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 12
- 229910052757 nitrogen Inorganic materials 0.000 claims description 11
- 125000004043 oxo group Chemical group O=* 0.000 claims description 11
- 229910052717 sulfur Inorganic materials 0.000 claims description 11
- 125000003342 alkenyl group Chemical group 0.000 claims description 9
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 9
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 claims description 8
- 230000002265 prevention Effects 0.000 claims description 8
- 125000000304 alkynyl group Chemical group 0.000 claims description 7
- 229910052760 oxygen Inorganic materials 0.000 claims description 7
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 6
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 6
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 5
- 239000003937 drug carrier Substances 0.000 claims description 5
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 5
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims description 4
- 125000002206 pyridazin-3-yl group Chemical group [H]C1=C([H])C([H])=C(*)N=N1 0.000 claims description 4
- DLKJUKMBGLLCCH-UHFFFAOYSA-N 2-[[6-[(2,5-dimethylpyrazol-3-yl)methoxymethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CN1N=C(C)C=C1COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 DLKJUKMBGLLCCH-UHFFFAOYSA-N 0.000 claims description 3
- YYGGBTVDXCDRDD-UHFFFAOYSA-N 5-(2,4-difluorophenyl)-2-[[6-(hydroxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC(F)=CC=2)F)SC=1NC1=CC=CC(CO)=N1 YYGGBTVDXCDRDD-UHFFFAOYSA-N 0.000 claims description 3
- KSLFMOQLCWQCDV-UHFFFAOYSA-N 5-(2-fluorophenyl)-2-[[6-(1-hydroxyethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)F)C(N)=O)=N1 KSLFMOQLCWQCDV-UHFFFAOYSA-N 0.000 claims description 3
- VWZIWKHACUENGU-UHFFFAOYSA-N 5-(2-fluorophenyl)-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=CC=2)F)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 VWZIWKHACUENGU-UHFFFAOYSA-N 0.000 claims description 3
- DNBXGECMOMERGE-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-(1-pyrrolidin-1-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C=CC(F)=CC=2)C(N)=O)=NC=1C(C)N1CCCC1 DNBXGECMOMERGE-UHFFFAOYSA-N 0.000 claims description 3
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 3
- 125000001153 fluoro group Chemical group F* 0.000 claims description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 3
- SHNHBCRCMYROHN-UHFFFAOYSA-N 2-[(2-cyanopyrimidin-4-yl)amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(C#N)=N1 SHNHBCRCMYROHN-UHFFFAOYSA-N 0.000 claims description 2
- UODZILSKQFMUEU-UHFFFAOYSA-N 2-[(2-cyclopropylpyrimidin-4-yl)amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(C2CC2)=N1 UODZILSKQFMUEU-UHFFFAOYSA-N 0.000 claims description 2
- AWWFHHWPYXDQPF-UHFFFAOYSA-N 2-[(2-cyclopropylpyrimidin-4-yl)amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(C2CC2)=N1 AWWFHHWPYXDQPF-UHFFFAOYSA-N 0.000 claims description 2
- WXPDYKHYNWPMOZ-UHFFFAOYSA-N 2-[(3-cyanopyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=NC=CC=C1C#N WXPDYKHYNWPMOZ-UHFFFAOYSA-N 0.000 claims description 2
- QNRDVJGUFDCZER-UHFFFAOYSA-N 2-[(3-methylpyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound CC1=CC=CN=C1NC1=C(C(N)=O)C=C(C=2C=CC=CC=2)S1 QNRDVJGUFDCZER-UHFFFAOYSA-N 0.000 claims description 2
- IACSKFARINEVLY-UHFFFAOYSA-N 2-[(4-cyanopyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC(C#N)=CC=N1 IACSKFARINEVLY-UHFFFAOYSA-N 0.000 claims description 2
- BMKQUIYXVMJNGT-UHFFFAOYSA-N 2-[(4-fluoropyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC(F)=CC=N1 BMKQUIYXVMJNGT-UHFFFAOYSA-N 0.000 claims description 2
- VTXTZOSQVPMRAU-UHFFFAOYSA-N 2-[(4-methylpyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound CC1=CC=NC(NC2=C(C=C(S2)C=2C=CC=CC=2)C(N)=O)=C1 VTXTZOSQVPMRAU-UHFFFAOYSA-N 0.000 claims description 2
- WGWROBGETVTDKT-UHFFFAOYSA-N 2-[(5-chloropyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=C(Cl)C=N1 WGWROBGETVTDKT-UHFFFAOYSA-N 0.000 claims description 2
- HKDAFZSLOWQBIO-UHFFFAOYSA-N 2-[(5-cyanopyridin-2-yl)amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C#N)C=N1 HKDAFZSLOWQBIO-UHFFFAOYSA-N 0.000 claims description 2
- LNKJVBPMSIWNLF-UHFFFAOYSA-N 2-[(5-cyanopyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=C(C#N)C=N1 LNKJVBPMSIWNLF-UHFFFAOYSA-N 0.000 claims description 2
- UHRZCQNOYZSYSW-UHFFFAOYSA-N 2-[(5-fluoropyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=C(F)C=N1 UHRZCQNOYZSYSW-UHFFFAOYSA-N 0.000 claims description 2
- GYDCIXOMZASZCK-UHFFFAOYSA-N 2-[(5-methylpyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound N1=CC(C)=CC=C1NC1=C(C(N)=O)C=C(C=2C=CC=CC=2)S1 GYDCIXOMZASZCK-UHFFFAOYSA-N 0.000 claims description 2
- ALQAFKSRAQPQEL-UHFFFAOYSA-N 2-[(5-methylsulfonylpyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound N1=CC(S(=O)(=O)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C=CC=CC=2)S1 ALQAFKSRAQPQEL-UHFFFAOYSA-N 0.000 claims description 2
- XMRBESXMJKJKER-UHFFFAOYSA-N 2-[(6-aminopyridazin-3-yl)amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(N)N=N1 XMRBESXMJKJKER-UHFFFAOYSA-N 0.000 claims description 2
- LINDYQCNJFSPNQ-UHFFFAOYSA-N 2-[(6-methylpyridin-2-yl)amino]-5-[3-(trifluoromethyl)phenyl]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=C(C=CC=2)C(F)(F)F)C(N)=O)=N1 LINDYQCNJFSPNQ-UHFFFAOYSA-N 0.000 claims description 2
- QEKRWXNCLYLCFJ-UHFFFAOYSA-N 2-[(6-methylpyridin-2-yl)amino]-5-[4-(trifluoromethyl)phenyl]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(F)(F)F)C(N)=O)=N1 QEKRWXNCLYLCFJ-UHFFFAOYSA-N 0.000 claims description 2
- KFKOZYNYWLCLCJ-UHFFFAOYSA-N 2-[(6-methylpyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC=CC=2)C(N)=O)=N1 KFKOZYNYWLCLCJ-UHFFFAOYSA-N 0.000 claims description 2
- ZDYAPPQDIHTBMT-UHFFFAOYSA-N 2-[[5-(2-hydroxypropan-2-yl)-6-methylpyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound C1=C(C(C)(C)O)C(C)=NC(NC2=C(C=C(S2)C=2C=NC(=CC=2)N2CCOCC2)C(N)=O)=C1 ZDYAPPQDIHTBMT-UHFFFAOYSA-N 0.000 claims description 2
- LBPWXQATVAGCCH-UHFFFAOYSA-N 2-[[5-(azetidin-3-yloxymethyl)-6-methylpyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C=1C=C(COC2CNC2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F LBPWXQATVAGCCH-UHFFFAOYSA-N 0.000 claims description 2
- OTJUOQPMJGCYRY-UHFFFAOYSA-N 2-[[5-(cyclopropylmethylamino)-6-methylpyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C=1C=C(NCC2CC2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F OTJUOQPMJGCYRY-UHFFFAOYSA-N 0.000 claims description 2
- SHEYPXMQXOPKDR-UHFFFAOYSA-N 2-[[5-[bis(methylsulfonyl)amino]-6-methylpyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=C(N(S(C)(=O)=O)S(C)(=O)=O)C(C)=NC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=C1 SHEYPXMQXOPKDR-UHFFFAOYSA-N 0.000 claims description 2
- HBYJQIKNNXLSIB-UHFFFAOYSA-N 2-[[6-(1,2-diamino-2-oxoethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(N)C(N)=O)=N1 HBYJQIKNNXLSIB-UHFFFAOYSA-N 0.000 claims description 2
- HLNZAPRWXJJTEO-UHFFFAOYSA-N 2-[[6-(1,2-dihydroxyethyl)pyridin-2-yl]amino]-5-(2-fluorophenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=CC=2)F)SC=1NC1=CC=CC(C(O)CO)=N1 HLNZAPRWXJJTEO-UHFFFAOYSA-N 0.000 claims description 2
- WMBQGRDXMBEUNA-UHFFFAOYSA-N 2-[[6-(1,2-dihydroxyethyl)pyridin-2-yl]amino]-5-(4-fluorophenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(F)=CC=2)SC=1NC1=CC=CC(C(O)CO)=N1 WMBQGRDXMBEUNA-UHFFFAOYSA-N 0.000 claims description 2
- WVHKGQLYBRAOKW-UHFFFAOYSA-N 2-[[6-(1-amino-2-methyl-1-oxopropan-2-yl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(C)(C)C(N)=O)=N1 WVHKGQLYBRAOKW-UHFFFAOYSA-N 0.000 claims description 2
- IXJZYQVLTCFAJL-UHFFFAOYSA-N 2-[[6-(1-cyanocyclopropyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C2(CC2)C#N)=N1 IXJZYQVLTCFAJL-UHFFFAOYSA-N 0.000 claims description 2
- ANGWDGSKVXJNDI-UHFFFAOYSA-N 2-[[6-(1-cyanoethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound N#CC(C)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 ANGWDGSKVXJNDI-UHFFFAOYSA-N 0.000 claims description 2
- HCEJSVLHVHNXDQ-UHFFFAOYSA-N 2-[[6-(2,2-difluoroethoxymethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(F)F)=N1 HCEJSVLHVHNXDQ-UHFFFAOYSA-N 0.000 claims description 2
- VUIIBIYIRUBLRM-UHFFFAOYSA-N 2-[[6-(2-amino-1-hydroxy-2-oxoethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(O)C(N)=O)=N1 VUIIBIYIRUBLRM-UHFFFAOYSA-N 0.000 claims description 2
- VWCHGRLVRAILGK-UHFFFAOYSA-N 2-[[6-(2-amino-2-oxoethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC(N)=O)=N1 VWCHGRLVRAILGK-UHFFFAOYSA-N 0.000 claims description 2
- VRXQTRWUMPQNPU-UHFFFAOYSA-N 2-[[6-(2-cyanopropan-2-yl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(C)(C)C#N)=N1 VRXQTRWUMPQNPU-UHFFFAOYSA-N 0.000 claims description 2
- QYVVXUXARYNYTL-UHFFFAOYSA-N 2-[[6-(3,3-difluoroazetidin-1-yl)pyridazin-3-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(N2CC(F)(F)C2)N=N1 QYVVXUXARYNYTL-UHFFFAOYSA-N 0.000 claims description 2
- IDFMVYAHNNXBEE-UHFFFAOYSA-N 2-[[6-(azetidin-1-ylmethyl)pyridin-2-yl]amino]-5-(2,5-dichlorophenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=C(Cl)C=2)Cl)SC=1NC(N=1)=CC=CC=1CN1CCC1 IDFMVYAHNNXBEE-UHFFFAOYSA-N 0.000 claims description 2
- MOUFQMUAXPFETF-UHFFFAOYSA-N 2-[[6-(cyanomethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC#N)=N1 MOUFQMUAXPFETF-UHFFFAOYSA-N 0.000 claims description 2
- QUKAQWXWUBOZPE-UHFFFAOYSA-N 2-[[6-(cyanomethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC#N)=N1 QUKAQWXWUBOZPE-UHFFFAOYSA-N 0.000 claims description 2
- QQSMDDFIHDZMFN-UHFFFAOYSA-N 2-[[6-(cyclobutylmethoxymethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC2CCC2)=N1 QQSMDDFIHDZMFN-UHFFFAOYSA-N 0.000 claims description 2
- SRJOSBMLAKNAJL-UHFFFAOYSA-N 2-[[6-(dimethylphosphoryloxymethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COP(C)(C)=O)=N1 SRJOSBMLAKNAJL-UHFFFAOYSA-N 0.000 claims description 2
- ORDODNYOMFSQSD-UHFFFAOYSA-N 2-[[6-(ethoxymethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 ORDODNYOMFSQSD-UHFFFAOYSA-N 0.000 claims description 2
- QIWVKCPTEPDTHV-UHFFFAOYSA-N 2-[[6-(hydroxymethyl)pyridin-2-yl]amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=CC(CO)=N1 QIWVKCPTEPDTHV-UHFFFAOYSA-N 0.000 claims description 2
- YKHHVRCRJHPQCE-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(2-piperazin-1-ylpyridin-4-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=C(N=CC=2)N2CCNCC2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 YKHHVRCRJHPQCE-UHFFFAOYSA-N 0.000 claims description 2
- FNWNUKZTPIQCKI-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-pyridin-4-ylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CN=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 FNWNUKZTPIQCKI-UHFFFAOYSA-N 0.000 claims description 2
- WIWGITIJSOXNQM-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-pyrimidin-5-ylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC=NC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 WIWGITIJSOXNQM-UHFFFAOYSA-N 0.000 claims description 2
- PIVCHZPRURUMLA-UHFFFAOYSA-N 2-[[6-(tert-butylsulfonylmethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CS(=O)(=O)C(C)(C)C)=N1 PIVCHZPRURUMLA-UHFFFAOYSA-N 0.000 claims description 2
- HAAKDVPJUMWTGY-UHFFFAOYSA-N 2-[[6-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCS(=O)(=O)CC2)=N1 HAAKDVPJUMWTGY-UHFFFAOYSA-N 0.000 claims description 2
- SFZUMXUMABPVCT-UHFFFAOYSA-N 2-[[6-[(2,4-dimethyl-1,3-thiazol-5-yl)methoxymethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound S1C(C)=NC(C)=C1COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 SFZUMXUMABPVCT-UHFFFAOYSA-N 0.000 claims description 2
- SLWVCHTVNYSTPV-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)O)C(N)=O)=N1 SLWVCHTVNYSTPV-UHFFFAOYSA-N 0.000 claims description 2
- LDLMJMPQUILFCX-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-[6-(2-hydroxypropan-2-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=NC(=CC=2)C(C)(C)O)C(N)=O)=N1 LDLMJMPQUILFCX-UHFFFAOYSA-N 0.000 claims description 2
- UIUYQPIOJYZABJ-JOCHJYFZSA-N 2-[[6-[(2s)-1,2-dihydroxypropan-2-yl]pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC([C@](C)(O)CO)=N1 UIUYQPIOJYZABJ-JOCHJYFZSA-N 0.000 claims description 2
- CETFPZNXMWMCHJ-UHFFFAOYSA-N 2-[[6-[(4-acetylpiperazin-1-yl)methyl]pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1CN(C(=O)C)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)O)C(N)=O)=N1 CETFPZNXMWMCHJ-UHFFFAOYSA-N 0.000 claims description 2
- AVKFNVJIYJAFJK-UHFFFAOYSA-N 2-[[6-[(4-diethoxyphosphorylpiperidin-1-yl)methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1CC(P(=O)(OCC)OCC)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 AVKFNVJIYJAFJK-UHFFFAOYSA-N 0.000 claims description 2
- QYGDPNBQNIDZHH-UHFFFAOYSA-N 2-[[6-[2-(2-azaspiro[3.3]heptan-2-yl)ethoxymethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCCN2CC3(CCC3)C2)=N1 QYGDPNBQNIDZHH-UHFFFAOYSA-N 0.000 claims description 2
- WQMOGEQRLYEQCS-UHFFFAOYSA-N 2-[[6-[2-(dimethylamino)-2-oxoethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CN(C)C(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 WQMOGEQRLYEQCS-UHFFFAOYSA-N 0.000 claims description 2
- XVLOEBVEBXXZCJ-UHFFFAOYSA-N 2-[[6-[2-(dimethylamino)ethoxymethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CN(C)CCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 XVLOEBVEBXXZCJ-UHFFFAOYSA-N 0.000 claims description 2
- SZTRDXFWOUQRLL-UHFFFAOYSA-N 2-[[6-[[(2-cyanoacetyl)amino]methyl]pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC(=O)CC#N)=N1 SZTRDXFWOUQRLL-UHFFFAOYSA-N 0.000 claims description 2
- FFCIFMGFBDOMKY-UHFFFAOYSA-N 2-[[6-[[1-[2-(dimethylamino)-2-oxoethyl]triazol-4-yl]-hydroxymethyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound N1=NN(CC(=O)N(C)C)C=C1C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 FFCIFMGFBDOMKY-UHFFFAOYSA-N 0.000 claims description 2
- WNVSUINBPOVZJO-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-(4-pyridin-4-ylphenyl)thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C=2C=CN=CC=2)C(N)=O)=N1 WNVSUINBPOVZJO-UHFFFAOYSA-N 0.000 claims description 2
- DSEVWACLHAAORQ-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-(6-pyridin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=NC(=CC=2)C=2C=CN=CC=2)C(N)=O)=N1 DSEVWACLHAAORQ-UHFFFAOYSA-N 0.000 claims description 2
- DEHKGGIQQAFASH-UHFFFAOYSA-N 2-[[6-[[[2-(2-dimethylphosphorylethylamino)-2-oxoethyl]amino]methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(=O)NCCP(C)(C)=O)=N1 DEHKGGIQQAFASH-UHFFFAOYSA-N 0.000 claims description 2
- IENYKESGPZXKRT-UHFFFAOYSA-N 2-[[6-[[[2-(cyclopentylamino)-2-oxoethyl]-methylamino]methyl]pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=NC=1CN(C)CC(=O)NC1CCCC1 IENYKESGPZXKRT-UHFFFAOYSA-N 0.000 claims description 2
- ORNYBEKCGBJBSL-UHFFFAOYSA-N 2-[[6-[[[2-[(1,1-dioxothiolan-3-yl)amino]-2-oxoethyl]amino]methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(=O)NC2CS(=O)(=O)CC2)=N1 ORNYBEKCGBJBSL-UHFFFAOYSA-N 0.000 claims description 2
- BPESHROBJVDGFI-UHFFFAOYSA-N 2-[[6-[[acetyl-[2-(cyclopentylamino)-2-oxoethyl]amino]methyl]pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=NC=1CN(C(=O)C)CC(=O)NC1CCCC1 BPESHROBJVDGFI-UHFFFAOYSA-N 0.000 claims description 2
- MDWKXORSXQGCNB-UHFFFAOYSA-N 2-[[6-chloro-5-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C(C)(C)O)C(Cl)=N1 MDWKXORSXQGCNB-UHFFFAOYSA-N 0.000 claims description 2
- TZVGQLGCOMURSY-UHFFFAOYSA-N 5-(2,3-difluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=C(F)C=CC=2)F)C(N)=O)=N1 TZVGQLGCOMURSY-UHFFFAOYSA-N 0.000 claims description 2
- AGPCFCFLRORQFL-UHFFFAOYSA-N 5-(2,4-difluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(F)=CC=2)F)C(N)=O)=N1 AGPCFCFLRORQFL-UHFFFAOYSA-N 0.000 claims description 2
- PVLAHHAALFBREA-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(Cl)C=2)Cl)C(N)=O)=N1 PVLAHHAALFBREA-UHFFFAOYSA-N 0.000 claims description 2
- XOQKFPQDPBTUOP-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-(1,2-dihydroxyethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=C(Cl)C=2)Cl)SC=1NC1=CC=CC(C(O)CO)=N1 XOQKFPQDPBTUOP-UHFFFAOYSA-N 0.000 claims description 2
- QVMIRVWRAQBZBR-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(Cl)C=2)Cl)C(N)=O)=N1 QVMIRVWRAQBZBR-UHFFFAOYSA-N 0.000 claims description 2
- JKPXYFCBOBZOHM-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-(methoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(Cl)C=2)Cl)C(N)=O)=N1 JKPXYFCBOBZOHM-UHFFFAOYSA-N 0.000 claims description 2
- HAOVUJXZLFQISY-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-(piperidin-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=C(Cl)C=2)Cl)SC=1NC(N=1)=CC=CC=1CN1CCCCC1 HAOVUJXZLFQISY-UHFFFAOYSA-N 0.000 claims description 2
- YGUVSIVROCIMFV-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-[(dimethylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN(C)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(Cl)C=2)Cl)C(N)=O)=N1 YGUVSIVROCIMFV-UHFFFAOYSA-N 0.000 claims description 2
- YDWVZOQYIRKCBZ-UHFFFAOYSA-N 5-(2,5-difluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(F)C=2)F)C(N)=O)=N1 YDWVZOQYIRKCBZ-UHFFFAOYSA-N 0.000 claims description 2
- AJXRGJGBKSFLRB-UHFFFAOYSA-N 5-(2-aminophenyl)-2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)N)C(N)=O)=N1 AJXRGJGBKSFLRB-UHFFFAOYSA-N 0.000 claims description 2
- WHFCMMSQBVBJNM-UHFFFAOYSA-N 5-(2-chlorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)Cl)C(N)=O)=N1 WHFCMMSQBVBJNM-UHFFFAOYSA-N 0.000 claims description 2
- LKLNOHJSWPHQHN-UHFFFAOYSA-N 5-(2-fluoro-5-methoxyphenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound COC1=CC=C(F)C(C=2SC(NC=3N=C(C)C=CC=3)=C(C(N)=O)C=2)=C1 LKLNOHJSWPHQHN-UHFFFAOYSA-N 0.000 claims description 2
- LVLRUUAOYVXFQO-UHFFFAOYSA-N 5-(2-fluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)F)C(N)=O)=N1 LVLRUUAOYVXFQO-UHFFFAOYSA-N 0.000 claims description 2
- JKNXJMVCLNSMSS-UHFFFAOYSA-N 5-(3,4-dichlorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=C(Cl)C(Cl)=CC=2)C(N)=O)=N1 JKNXJMVCLNSMSS-UHFFFAOYSA-N 0.000 claims description 2
- JKTSJFULSAPROF-UHFFFAOYSA-N 5-(3-chlorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=C(Cl)C=CC=2)C(N)=O)=N1 JKTSJFULSAPROF-UHFFFAOYSA-N 0.000 claims description 2
- SEJJQNQCXGAZTA-UHFFFAOYSA-N 5-(3-fluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=C(F)C=CC=2)C(N)=O)=N1 SEJJQNQCXGAZTA-UHFFFAOYSA-N 0.000 claims description 2
- YDJVNHPZCPYDLY-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-(1,2-dihydroxypropan-2-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound OCC(O)(C)C1=CC=CC(NC2=C(C=C(S2)C=2C=CC(Cl)=CC=2)C(N)=O)=N1 YDJVNHPZCPYDLY-UHFFFAOYSA-N 0.000 claims description 2
- HMPGNCLOTXPDBR-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-(pyrrolidin-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(Cl)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCCC1 HMPGNCLOTXPDBR-UHFFFAOYSA-N 0.000 claims description 2
- YKYMQRMNTMWWMX-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-[2-(methylamino)-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CNC(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(Cl)=CC=2)C(N)=O)=N1 YKYMQRMNTMWWMX-UHFFFAOYSA-N 0.000 claims description 2
- IUXQKKOQMORCCX-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-[2-oxo-2-(prop-2-ynylamino)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(Cl)=CC=2)SC=1NC1=CC=CC(CC(=O)NCC#C)=N1 IUXQKKOQMORCCX-UHFFFAOYSA-N 0.000 claims description 2
- ACAOHWOPNZHWAF-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(F)=CC=2)C(N)=O)=N1 ACAOHWOPNZHWAF-UHFFFAOYSA-N 0.000 claims description 2
- WLPHONPBTYYSNN-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-(methylaminomethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CNCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(F)=CC=2)C(N)=O)=N1 WLPHONPBTYYSNN-UHFFFAOYSA-N 0.000 claims description 2
- LVEUPYNQMYUHIA-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-[(3-hydroxyazetidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(F)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CC(O)C1 LVEUPYNQMYUHIA-UHFFFAOYSA-N 0.000 claims description 2
- BEJSRXWDGQRSOR-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-[(3-hydroxypyrrolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(F)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCC(O)C1 BEJSRXWDGQRSOR-UHFFFAOYSA-N 0.000 claims description 2
- KYGAEJKWNAIYOD-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-[(4-hydroxypiperidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(F)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCC(O)CC1 KYGAEJKWNAIYOD-UHFFFAOYSA-N 0.000 claims description 2
- LXBWJZBIYDGKPV-UHFFFAOYSA-N 5-(4-hydroxyphenyl)-2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(O)=CC=2)C(N)=O)=N1 LXBWJZBIYDGKPV-UHFFFAOYSA-N 0.000 claims description 2
- MHFXJRKEGJZNJL-UHFFFAOYSA-N 5-(5-chloro-2-fluorophenyl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(Cl)C=2)F)C(N)=O)=N1 MHFXJRKEGJZNJL-UHFFFAOYSA-N 0.000 claims description 2
- CUMFSPRLPKJRLW-UHFFFAOYSA-N 5-[1-(2-methylpropyl)pyrazol-4-yl]-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound C1=NN(CC(C)C)C=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C)=N1 CUMFSPRLPKJRLW-UHFFFAOYSA-N 0.000 claims description 2
- MAXVIUDAWQJNIQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-(pyridin-2-ylamino)thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC=N1 MAXVIUDAWQJNIQ-UHFFFAOYSA-N 0.000 claims description 2
- LQFBULKJOLEVCA-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(2-methylpyrimidin-4-yl)amino]thiophene-3-carboxamide Chemical compound CC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 LQFBULKJOLEVCA-UHFFFAOYSA-N 0.000 claims description 2
- YBAXKKJUDRMSNT-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(2-oxo-1h-pyrimidin-6-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=NC(=O)NC=C1 YBAXKKJUDRMSNT-UHFFFAOYSA-N 0.000 claims description 2
- HRGJOTUOOVQVEO-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(2-pyrrolidin-1-ylpyrimidin-4-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(N2CCCC2)=N1 HRGJOTUOOVQVEO-UHFFFAOYSA-N 0.000 claims description 2
- LEKLMWDYEPLRPQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(4-fluoropyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC(F)=CC=N1 LEKLMWDYEPLRPQ-UHFFFAOYSA-N 0.000 claims description 2
- KZLDMPNONBWXSX-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-fluoropyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(F)=N1 KZLDMPNONBWXSX-UHFFFAOYSA-N 0.000 claims description 2
- ZLQXNPNNCPHYNS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-methoxypyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound COC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 ZLQXNPNNCPHYNS-UHFFFAOYSA-N 0.000 claims description 2
- TYVVNOHTXHAJGL-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-oxo-1h-pyridazin-3-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=NNC(=O)C=C1 TYVVNOHTXHAJGL-UHFFFAOYSA-N 0.000 claims description 2
- UTDYNVVBGGOCDH-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-pyrrolidin-1-ylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(N2CCCC2)=N1 UTDYNVVBGGOCDH-UHFFFAOYSA-N 0.000 claims description 2
- YLRLUTLWEKUVRJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-(ethylsulfonylmethyl)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound CCS(=O)(=O)CC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 YLRLUTLWEKUVRJ-UHFFFAOYSA-N 0.000 claims description 2
- BOIRMISMSLRHBJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-(methanesulfonamido)-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=C(NS(C)(=O)=O)C(C)=NC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=C1 BOIRMISMSLRHBJ-UHFFFAOYSA-N 0.000 claims description 2
- AQZCWVSXLNCVIZ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]-6-fluoropyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC(N=C1F)=CC=C1CN1CCS(=O)(=O)CC1 AQZCWVSXLNCVIZ-UHFFFAOYSA-N 0.000 claims description 2
- DUXXVQWVZIIUIU-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(CN2CCS(=O)(=O)CC2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F DUXXVQWVZIIUIU-UHFFFAOYSA-N 0.000 claims description 2
- NSEAPDJSBRKZGC-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(2-hydroxy-2-methylpropoxy)methyl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=C(COCC(C)(C)O)C(C)=NC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=C1 NSEAPDJSBRKZGC-UHFFFAOYSA-N 0.000 claims description 2
- JZOMXSWPZISORO-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(3-hydroxyazetidin-1-yl)methyl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(CN2CC(O)C2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F JZOMXSWPZISORO-UHFFFAOYSA-N 0.000 claims description 2
- NVJHQHHFZLKYJQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(4,4-difluoropiperidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC(N=C1)=CC=C1CN1CCC(F)(F)CC1 NVJHQHHFZLKYJQ-UHFFFAOYSA-N 0.000 claims description 2
- AFZDUJBWGKXTIK-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(dimethylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=CC(CN(C)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2F)C(C)(C)O)F)S1 AFZDUJBWGKXTIK-UHFFFAOYSA-N 0.000 claims description 2
- LYVXYMQQFSYLBI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxypropylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(O)CS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 LYVXYMQQFSYLBI-UHFFFAOYSA-N 0.000 claims description 2
- LFGWSPRKMWKHHP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-methylpropylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)CS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 LFGWSPRKMWKHHP-UHFFFAOYSA-N 0.000 claims description 2
- KSODHIXBMKCCBS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-hydroxy-3-methylbut-1-ynyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)C#CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 KSODHIXBMKCCBS-UHFFFAOYSA-N 0.000 claims description 2
- FKPWIVZXCIGENY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-hydroxypyrrolidine-1-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CC(O)CC2)=N1 FKPWIVZXCIGENY-UHFFFAOYSA-N 0.000 claims description 2
- FFQJJWPRXSUTOD-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(4-hydroxybutylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CS(=O)(=O)CCCCO)=N1 FFQJJWPRXSUTOD-UHFFFAOYSA-N 0.000 claims description 2
- FBHFZEGIEVFCCF-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(4-methylpiperazine-1-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1CN(C)CCN1C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 FBHFZEGIEVFCCF-UHFFFAOYSA-N 0.000 claims description 2
- JNTYDIQSEOJDDD-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(ethylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CCS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 JNTYDIQSEOJDDD-UHFFFAOYSA-N 0.000 claims description 2
- KWCIRYVCYXWPCO-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(methylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CS(C)(=O)=O)=N1 KWCIRYVCYXWPCO-UHFFFAOYSA-N 0.000 claims description 2
- KYGBMIGZXXIWJZ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(propan-2-ylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)S(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 KYGBMIGZXXIWJZ-UHFFFAOYSA-N 0.000 claims description 2
- BBWVAAFZVFVSFI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(propylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CCCS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 BBWVAAFZVFVSFI-UHFFFAOYSA-N 0.000 claims description 2
- VVSPFNSEJMXEDA-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1,1-dioxothian-4-yl)oxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COC2CCS(=O)(=O)CC2)=N1 VVSPFNSEJMXEDA-UHFFFAOYSA-N 0.000 claims description 2
- PDWZWFNKXFJUQZ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1,3-thiazol-2-ylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC=2SC=CN=2)=N1 PDWZWFNKXFJUQZ-UHFFFAOYSA-N 0.000 claims description 2
- MWRUVXRNXOIRCT-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1-methyltetrazol-5-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1N=NN=C1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 MWRUVXRNXOIRCT-UHFFFAOYSA-N 0.000 claims description 2
- HRQQSHKLURUDMQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 HRQQSHKLURUDMQ-UHFFFAOYSA-N 0.000 claims description 2
- QLZXXXVHLDBXHK-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropyl)sulfonylmethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)CS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 QLZXXXVHLDBXHK-UHFFFAOYSA-N 0.000 claims description 2
- KXRRTOCNSMKSCV-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-methyltetrazol-5-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1N=NC(CC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 KXRRTOCNSMKSCV-UHFFFAOYSA-N 0.000 claims description 2
- IKVRXVFNZUXODH-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-oxo-1,3-oxazolidin-3-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(OCC2)=O)=N1 IKVRXVFNZUXODH-UHFFFAOYSA-N 0.000 claims description 2
- NTDPVRRWBHEKJL-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-oxoimidazolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(NCC2)=O)=N1 NTDPVRRWBHEKJL-UHFFFAOYSA-N 0.000 claims description 2
- LXPBIQPDNPSVOI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-oxopyrrolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(CCC2)=O)=N1 LXPBIQPDNPSVOI-UHFFFAOYSA-N 0.000 claims description 2
- FWMTWGGAXJGEPT-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3,3-difluoropiperidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CC(F)(F)CCC2)=N1 FWMTWGGAXJGEPT-UHFFFAOYSA-N 0.000 claims description 2
- JALWDEAULLWJPT-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-methyl-2-oxoimidazolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O=C1N(C)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 JALWDEAULLWJPT-UHFFFAOYSA-N 0.000 claims description 2
- VCRLPARXSGSNTD-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-oxomorpholin-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(COCC2)=O)=N1 VCRLPARXSGSNTD-UHFFFAOYSA-N 0.000 claims description 2
- AFKWOJDUKGTEAS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methyl-3-oxopiperazin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1C(=O)N(C)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 AFKWOJDUKGTEAS-UHFFFAOYSA-N 0.000 claims description 2
- UEUZGTKGKZMNNX-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(5-methyl-1,3,4-oxadiazol-2-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1C(C)=NN=C1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 UEUZGTKGKZMNNX-UHFFFAOYSA-N 0.000 claims description 2
- SMXGNZXYIPDLRX-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[1-(2-hydroxyethyl)pyrazol-4-yl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C2=CN(CCO)N=C2)=N1 SMXGNZXYIPDLRX-UHFFFAOYSA-N 0.000 claims description 2
- MVTKDNKJHXJNQV-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[1-(3,3-difluoropiperidin-1-yl)-2-hydroxyethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2CC(F)(F)CCC2)=N1 MVTKDNKJHXJNQV-UHFFFAOYSA-N 0.000 claims description 2
- MHJCSSCENAJWGP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(2-hydroxy-2-methylpropoxy)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 MHJCSSCENAJWGP-UHFFFAOYSA-N 0.000 claims description 2
- XRZCPQGWFQFGDP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(dimethylamino)-1-hydroxy-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN(C)C(=O)C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 XRZCPQGWFQFGDP-UHFFFAOYSA-N 0.000 claims description 2
- XFGWNHRTXPQJBQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(dimethylamino)-1-morpholin-4-yl-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=NC=1C(C(=O)N(C)C)N1CCOCC1 XFGWNHRTXPQJBQ-UHFFFAOYSA-N 0.000 claims description 2
- ZVOBBBBDPPSHKY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(1-methylpyrazol-3-yl)amino]-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1C=CC(NC(=O)CC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 ZVOBBBBDPPSHKY-UHFFFAOYSA-N 0.000 claims description 2
- BXHHEZMHALNAHF-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(5-methyl-1,2,4-oxadiazol-3-yl)methoxy]ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1C(C)=NC(COCCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 BXHHEZMHALNAHF-UHFFFAOYSA-N 0.000 claims description 2
- KXJNSVGZXUESFY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(5-methyl-1,2-oxazol-3-yl)methoxy]ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1C(C)=CC(COCCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 KXJNSVGZXUESFY-UHFFFAOYSA-N 0.000 claims description 2
- PCCYJWDCGKAJOR-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[[3-(hydroxymethyl)-1,2-thiazol-5-yl]amino]-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC(=O)NC=2SN=C(CO)C=2)=N1 PCCYJWDCGKAJOR-UHFFFAOYSA-N 0.000 claims description 2
- AOBQAEJUFAJIGS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-hydroxyethyl(methyl)amino]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound OCCN(C)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 AOBQAEJUFAJIGS-UHFFFAOYSA-N 0.000 claims description 2
- ODTQKPHDHIIUEM-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-methyl-1-[(1-methylpyrazol-4-yl)amino]-1-oxopropan-2-yl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1NC(=O)C(C)(C)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 ODTQKPHDHIIUEM-UHFFFAOYSA-N 0.000 claims description 2
- DXWAYSQKTPPKAY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3,5-dimethyl-1,2-oxazol-4-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC1=NOC(C)=C1NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 DXWAYSQKTPPKAY-UHFFFAOYSA-N 0.000 claims description 2
- QXCQUTFJJXPGSG-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3-hydroxy-3-methylbutan-2-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(O)(C)C(C)NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 QXCQUTFJJXPGSG-UHFFFAOYSA-N 0.000 claims description 2
- AMVZMYOQZAKLIL-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(2-hydroxyethylamino)-2-oxoethoxy]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(=O)NCCO)=N1 AMVZMYOQZAKLIL-UHFFFAOYSA-N 0.000 claims description 2
- JJUCLNRUSOKIRY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(3-hydroxypyrrolidin-1-yl)-2-oxoethoxy]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(=O)N2CC(O)CC2)=N1 JJUCLNRUSOKIRY-UHFFFAOYSA-N 0.000 claims description 2
- DHVXVOGHFDKEPU-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[3-(2-hydroxyethyl)-2-oxoimidazolidin-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(N(CCO)CC2)=O)=N1 DHVXVOGHFDKEPU-UHFFFAOYSA-N 0.000 claims description 2
- SWJRBBFBGPXYNT-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[[3-(methoxymethyl)-1,2,4-oxadiazol-5-yl]methylamino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COCC1=NOC(CNCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 SWJRBBFBGPXYNT-UHFFFAOYSA-N 0.000 claims description 2
- ZBSNOTQYUDBMAJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[hydroxy(oxan-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(O)C2CCOCC2)=N1 ZBSNOTQYUDBMAJ-UHFFFAOYSA-N 0.000 claims description 2
- IPXXUPYDRVLOKN-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-fluoro-5-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C(C)(C)O)C(F)=N1 IPXXUPYDRVLOKN-UHFFFAOYSA-N 0.000 claims description 2
- KEJIGOPAXKCICD-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-methyl-5-[2-(3-oxomorpholin-4-yl)ethoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(COCCN2C(COCC2)=O)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F KEJIGOPAXKCICD-UHFFFAOYSA-N 0.000 claims description 2
- NJQLDEAANCLKIS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(3-hydroxyoxetan-3-yl)phenyl]-2-[[6-(propan-2-ylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)S(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C2(O)COC2)F)C(N)=O)=N1 NJQLDEAANCLKIS-UHFFFAOYSA-N 0.000 claims description 2
- FOMBVXKNOGKRCW-UHFFFAOYSA-N 5-[2-chloro-5-(trifluoromethyl)phenyl]-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=C(C=2)C(F)(F)F)Cl)C(N)=O)=N1 FOMBVXKNOGKRCW-UHFFFAOYSA-N 0.000 claims description 2
- FPERTSLXSOJYOF-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(5-fluoropyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(F)C=N1 FPERTSLXSOJYOF-UHFFFAOYSA-N 0.000 claims description 2
- WETFEVNRSZBFMU-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-(2,2,2-trifluoro-1-hydroxyethyl)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(C(O)C(F)(F)F)=N1 WETFEVNRSZBFMU-UHFFFAOYSA-N 0.000 claims description 2
- PKGSPNWLHTWPMC-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-[[2-(2-hydroxyethyl)morpholin-4-yl]methyl]pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2CC(CCO)OCC2)=N1 PKGSPNWLHTWPMC-UHFFFAOYSA-N 0.000 claims description 2
- OVTWJZIMASERAW-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-[[3-(fluoromethyl)morpholin-4-yl]methyl]pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2C(COCC2)CF)=N1 OVTWJZIMASERAW-UHFFFAOYSA-N 0.000 claims description 2
- YOCVXTJKRRMBKF-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(1,3-oxazol-2-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2OC=CN=2)=N1 YOCVXTJKRRMBKF-UHFFFAOYSA-N 0.000 claims description 2
- RLAQFHOTNJHPMY-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-methoxyethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COCCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 RLAQFHOTNJHPMY-UHFFFAOYSA-N 0.000 claims description 2
- QBHAJJPFWOARNZ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-propan-2-yloxyethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)OCCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 QBHAJJPFWOARNZ-UHFFFAOYSA-N 0.000 claims description 2
- JILRDZPHDBXTEQ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-hydroxyazetidine-1-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CC(O)C2)=N1 JILRDZPHDBXTEQ-UHFFFAOYSA-N 0.000 claims description 2
- GXAGNZVCXMXOGG-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-hydroxypyrrolidine-1-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CC(O)CC2)=N1 GXAGNZVCXMXOGG-UHFFFAOYSA-N 0.000 claims description 2
- FQVGLVZEXICOPL-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-methoxyazetidine-1-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1C(OC)CN1C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 FQVGLVZEXICOPL-UHFFFAOYSA-N 0.000 claims description 2
- RRUOFJNRCKSZKG-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3-methylsulfonylphenyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C=2C=C(C=CC=2)S(C)(=O)=O)=N1 RRUOFJNRCKSZKG-UHFFFAOYSA-N 0.000 claims description 2
- GJHJODFZOJMHMG-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(hept-3-ynoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CCCC#CCCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 GJHJODFZOJMHMG-UHFFFAOYSA-N 0.000 claims description 2
- RNPNRQKWNULPEH-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(methylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CS(C)(=O)=O)=N1 RNPNRQKWNULPEH-UHFFFAOYSA-N 0.000 claims description 2
- QGCBVKYSVSPQIW-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(oxan-3-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC2COCCC2)=N1 QGCBVKYSVSPQIW-UHFFFAOYSA-N 0.000 claims description 2
- DZFSWJNSBGFCCO-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(pyridin-4-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2C=CN=CC=2)=N1 DZFSWJNSBGFCCO-UHFFFAOYSA-N 0.000 claims description 2
- XIRNBTZWVLSNQX-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2,2,2-trifluoroethylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(F)(F)F)=N1 XIRNBTZWVLSNQX-UHFFFAOYSA-N 0.000 claims description 2
- JVQATNQSPBFRTK-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 JVQATNQSPBFRTK-UHFFFAOYSA-N 0.000 claims description 2
- XLCBFVCVTBFKMK-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropyl)sulfonylmethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)CS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 XLCBFVCVTBFKMK-UHFFFAOYSA-N 0.000 claims description 2
- BCIAXMHCAJMRRP-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-hydroxyethylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCCO)=N1 BCIAXMHCAJMRRP-UHFFFAOYSA-N 0.000 claims description 2
- VBQYHYFKWOQMNB-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-methyltetrazol-5-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1N=NC(CC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 VBQYHYFKWOQMNB-UHFFFAOYSA-N 0.000 claims description 2
- OAPCLBPQIXQXCS-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-oxo-1-oxa-3,8-diazaspiro[4.5]decan-8-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCC3(OC(=O)NC3)CC2)=N1 OAPCLBPQIXQXCS-UHFFFAOYSA-N 0.000 claims description 2
- GOZLSOVLDQVKRN-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-methoxy-3-methylbutoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COC(C)(C)CCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 GOZLSOVLDQVKRN-UHFFFAOYSA-N 0.000 claims description 2
- GOEZUUWISGLADM-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-methylsulfonylpyrrolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CC(CC2)S(C)(=O)=O)=N1 GOEZUUWISGLADM-UHFFFAOYSA-N 0.000 claims description 2
- GNPLMWNNJINNDK-OAHLLOKOSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3r)-3-methoxypyrrolidine-1-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1[C@H](OC)CCN1C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 GNPLMWNNJINNDK-OAHLLOKOSA-N 0.000 claims description 2
- QKJJCCPBKPDCTC-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methyl-3-oxopiperazin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1C(=O)N(C)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 QKJJCCPBKPDCTC-UHFFFAOYSA-N 0.000 claims description 2
- WEQCXHBDOVHADE-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(5-methyl-1,3,4-oxadiazol-2-yl)methoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1C(C)=NN=C1COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 WEQCXHBDOVHADE-UHFFFAOYSA-N 0.000 claims description 2
- KEFCKIVTDQLREK-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(pyrrolidin-3-ylamino)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC2CNCC2)=N1 KEFCKIVTDQLREK-UHFFFAOYSA-N 0.000 claims description 2
- IDZPQAAZEZGEPA-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-hydroxy-1-(triazol-1-yl)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2N=NC=C2)=N1 IDZPQAAZEZGEPA-UHFFFAOYSA-N 0.000 claims description 2
- WLYFLBCNEBPCNH-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[3-(hydroxymethyl)morpholine-4-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2C(COCC2)CO)=N1 WLYFLBCNEBPCNH-UHFFFAOYSA-N 0.000 claims description 2
- GZZBAXFKTMVIJZ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[4-(hydroxymethyl)piperidine-1-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CCC(CO)CC2)=N1 GZZBAXFKTMVIJZ-UHFFFAOYSA-N 0.000 claims description 2
- KCCJHDYRGWZNCL-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(1-methyl-2-oxopyrrolidin-3-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O=C1N(C)CCC1NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 KCCJHDYRGWZNCL-UHFFFAOYSA-N 0.000 claims description 2
- HKUWCXBALDXZKM-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3,3,3-trifluoro-2-hydroxypropyl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(O)C(F)(F)F)=N1 HKUWCXBALDXZKM-UHFFFAOYSA-N 0.000 claims description 2
- MWKCZJASMKJSHT-HNNXBMFYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3s)-3-fluoropyrrolidin-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C[C@@H](F)CC2)=N1 MWKCZJASMKJSHT-HNNXBMFYSA-N 0.000 claims description 2
- ZPWZPXSFURZLNM-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(4-hydroxy-1,1-dioxothiolan-3-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC2C(CS(=O)(=O)C2)O)=N1 ZPWZPXSFURZLNM-UHFFFAOYSA-N 0.000 claims description 2
- OQQPJUSXFOVEHC-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[1-(2-hydroxyethyl)tetrazol-5-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC=2N(N=NN=2)CCO)=N1 OQQPJUSXFOVEHC-UHFFFAOYSA-N 0.000 claims description 2
- SQVHEYHHAZEABE-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(2-hydroxyethyl)tetrazol-5-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC2=NN(CCO)N=N2)=N1 SQVHEYHHAZEABE-UHFFFAOYSA-N 0.000 claims description 2
- XBQWUYYWCORVQH-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(2-methoxyethylamino)-2-oxoethoxy]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COCCNC(=O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 XBQWUYYWCORVQH-UHFFFAOYSA-N 0.000 claims description 2
- LQFWJESCKXZZBE-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(hydroxymethyl)morpholin-4-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CC(CO)OCC2)=N1 LQFWJESCKXZZBE-UHFFFAOYSA-N 0.000 claims description 2
- MOGQTIHHGWRJDF-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(trifluoromethyl)pyrrolidin-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(CCC2)C(F)(F)F)=N1 MOGQTIHHGWRJDF-UHFFFAOYSA-N 0.000 claims description 2
- FVDSPBSINAAENT-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-hydroxyethyl(methyl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound OCCN(C)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 FVDSPBSINAAENT-UHFFFAOYSA-N 0.000 claims description 2
- KXGCTVAYXUXXLL-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-oxo-2-(oxolan-3-ylmethylamino)ethoxy]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(=O)NCC2COCC2)=N1 KXGCTVAYXUXXLL-UHFFFAOYSA-N 0.000 claims description 2
- WUMKAGGBQRSLON-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-(2-hydroxyethyl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC(CCO)=C2)=N1 WUMKAGGBQRSLON-UHFFFAOYSA-N 0.000 claims description 2
- RQPHMRAKUQYTQJ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 RQPHMRAKUQYTQJ-UHFFFAOYSA-N 0.000 claims description 2
- BLXUSHZNFBZAKN-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-(hydroxymethyl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC(CO)=C2)=N1 BLXUSHZNFBZAKN-UHFFFAOYSA-N 0.000 claims description 2
- OIWDLNBUNOXMJV-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[[2-(2-hydroxyethylamino)-2-oxoethyl]amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(=O)NCCO)=N1 OIWDLNBUNOXMJV-UHFFFAOYSA-N 0.000 claims description 2
- DQUAHCLRZCNHJF-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[fluoro-(1-methyltriazol-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NN(C)C=C1C(F)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 DQUAHCLRZCNHJF-UHFFFAOYSA-N 0.000 claims description 2
- GZPPFYPOSANEAZ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[hydroxy(oxan-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(O)C2CCOCC2)=N1 GZPPFYPOSANEAZ-UHFFFAOYSA-N 0.000 claims description 2
- ORFPLOUQQNEHDA-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[hydroxy-(1-methyltriazol-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NN(C)C=C1C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 ORFPLOUQQNEHDA-UHFFFAOYSA-N 0.000 claims description 2
- JLZNJZKTQLNEBP-UHFFFAOYSA-N 5-[4-(1,1-dioxo-1,4-thiazinan-4-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)N2CCS(=O)(=O)CC2)C(N)=O)=N1 JLZNJZKTQLNEBP-UHFFFAOYSA-N 0.000 claims description 2
- RDGVWOOKXSLJKL-UHFFFAOYSA-N 5-[4-(2-hydroxypropan-2-yl)phenyl]-2-(pyridin-3-ylamino)thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CN=C1 RDGVWOOKXSLJKL-UHFFFAOYSA-N 0.000 claims description 2
- WXOGMYUOBRJWHG-UHFFFAOYSA-N 5-[4-(3-fluorooxetan-3-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C2(F)COC2)C(N)=O)=N1 WXOGMYUOBRJWHG-UHFFFAOYSA-N 0.000 claims description 2
- KDUCCSMULYHFMY-UHFFFAOYSA-N 5-[4-(acetamidomethyl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=CC(CNC(=O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(C)(C)O)=N1 KDUCCSMULYHFMY-UHFFFAOYSA-N 0.000 claims description 2
- VCCDHNSFGNKDHR-UHFFFAOYSA-N 5-phenyl-2-(pyridin-2-ylamino)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=CC=N1 VCCDHNSFGNKDHR-UHFFFAOYSA-N 0.000 claims description 2
- GYPULEGDSSWHQJ-UHFFFAOYSA-N 5-phenyl-2-[[5-(trifluoromethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=C(C(F)(F)F)C=N1 GYPULEGDSSWHQJ-UHFFFAOYSA-N 0.000 claims description 2
- YADIXEQCUGBDJK-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n,n-bis(2-hydroxyethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N(CCO)CCO)=N1 YADIXEQCUGBDJK-UHFFFAOYSA-N 0.000 claims description 2
- ATIIIEKNKNUTEP-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(1,3-oxazol-2-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC=2OC=CN=2)=N1 ATIIIEKNKNUTEP-UHFFFAOYSA-N 0.000 claims description 2
- HBNCMACDTQGQRZ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-hydroxypropyl)pyridine-2-carboxamide Chemical compound CC(O)CNC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 HBNCMACDTQGQRZ-UHFFFAOYSA-N 0.000 claims description 2
- SSIMTNXPLAXFDQ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-methoxyethyl)pyridine-2-carboxamide Chemical compound COCCNC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 SSIMTNXPLAXFDQ-UHFFFAOYSA-N 0.000 claims description 2
- MSCSINHPEWEVAP-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxetan-3-yl)pyridine-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C(=O)NC2COC2)C=N1 MSCSINHPEWEVAP-UHFFFAOYSA-N 0.000 claims description 2
- VHCXNGDERPVIGJ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-[(3-methyloxetan-3-yl)methyl]pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2(C)COC2)=N1 VHCXNGDERPVIGJ-UHFFFAOYSA-N 0.000 claims description 2
- BYWRMOUQCMKWJI-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-ethyl-n-methylpyridine-2-carboxamide Chemical compound CCN(C)C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 BYWRMOUQCMKWJI-UHFFFAOYSA-N 0.000 claims description 2
- KPYXSPVDAAKLQF-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-methyl-n-(pyrazin-2-ylmethyl)pyridine-2-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=NC=1C(=O)N(C)CC1=CN=CC=N1 KPYXSPVDAAKLQF-UHFFFAOYSA-N 0.000 claims description 2
- VUNDPURHPYTTPY-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-methyl-n-[2-(1-methylpyrazol-4-yl)ethyl]pyridine-2-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=NC=1C(=O)N(C)CCC=1C=NN(C)C=1 VUNDPURHPYTTPY-UHFFFAOYSA-N 0.000 claims description 2
- VPFPTDBDWPWAHT-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-methylpyridine-2-carboxamide Chemical compound CNC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 VPFPTDBDWPWAHT-UHFFFAOYSA-N 0.000 claims description 2
- SVLXPIONCLRZGS-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-methylpyridine-3-carboxamide Chemical compound N1=CC(C(=O)NC)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2)C(C)(C)O)F)S1 SVLXPIONCLRZGS-UHFFFAOYSA-N 0.000 claims description 2
- MTPFSVPMMKWSGF-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridine-2-carboxylic acid Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(O)=O)=N1 MTPFSVPMMKWSGF-UHFFFAOYSA-N 0.000 claims description 2
- 101100518501 Mus musculus Spp1 gene Proteins 0.000 claims description 2
- 229910007161 Si(CH3)3 Inorganic materials 0.000 claims description 2
- GHXYXFKOLZYUET-UHFFFAOYSA-N [6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl n-methylcarbamate Chemical compound CNC(=O)OCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 GHXYXFKOLZYUET-UHFFFAOYSA-N 0.000 claims description 2
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 claims description 2
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 claims description 2
- PXBRQCKWGAHEHS-UHFFFAOYSA-N dichlorodifluoromethane Chemical group FC(F)(Cl)Cl PXBRQCKWGAHEHS-UHFFFAOYSA-N 0.000 claims description 2
- 125000004005 formimidoyl group Chemical group [H]\N=C(/[H])* 0.000 claims description 2
- 125000001188 haloalkyl group Chemical group 0.000 claims description 2
- SLHMSMWWLDLFHH-UHFFFAOYSA-N methyl 5-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyrazine-2-carboxylate Chemical compound C1=NC(C(=O)OC)=CN=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2)C(C)(C)O)F)S1 SLHMSMWWLDLFHH-UHFFFAOYSA-N 0.000 claims description 2
- FEYQIFZPPZWAHG-UHFFFAOYSA-N methyl 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridine-2-carboxylate Chemical compound COC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 FEYQIFZPPZWAHG-UHFFFAOYSA-N 0.000 claims description 2
- IDHOQENKVRYZBZ-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-1-methylpyrazole-4-carboxamide Chemical compound C1=NN(C)C=C1C(=O)NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 IDHOQENKVRYZBZ-UHFFFAOYSA-N 0.000 claims description 2
- IJUCAELNFRKYIR-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-2-(hydroxymethyl)pyridine-4-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC(=O)C=2C=C(CO)N=CC=2)=N1 IJUCAELNFRKYIR-UHFFFAOYSA-N 0.000 claims description 2
- IHHCXNSTHZDLGF-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-5-methyl-1,3,4-oxadiazole-2-carboxamide Chemical compound O1C(C)=NN=C1C(=O)NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 IHHCXNSTHZDLGF-UHFFFAOYSA-N 0.000 claims description 2
- IKGPNFLRHBVUCD-UHFFFAOYSA-N tert-butyl 4-[[6-[[3-carbamoyl-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]-hydroxymethyl]piperidine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCC1C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)O)C(N)=O)=N1 IKGPNFLRHBVUCD-UHFFFAOYSA-N 0.000 claims description 2
- OSSPWUJREOGIAT-UHFFFAOYSA-N tert-butyl n-[2-[[6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methylamino]ethyl]carbamate Chemical compound CC(C)(C)OC(=O)NCCNCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 OSSPWUJREOGIAT-UHFFFAOYSA-N 0.000 claims description 2
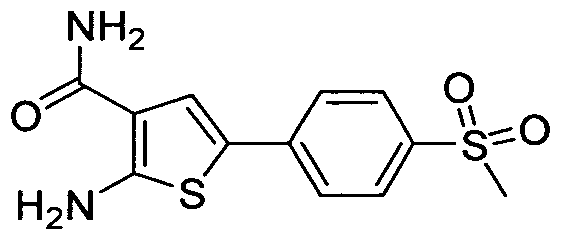
- LSXURHOVKBWUCZ-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-(4-methylsulfonylphenyl)thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)S(C)(=O)=O)C(N)=O)=N1 LSXURHOVKBWUCZ-UHFFFAOYSA-N 0.000 claims 2
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 claims 2
- VUAXHMVRKOTJKP-UHFFFAOYSA-M 2,2-dimethylbutanoate Chemical compound CCC(C)(C)C([O-])=O VUAXHMVRKOTJKP-UHFFFAOYSA-M 0.000 claims 1
- IGZTXAYDXRHINW-UHFFFAOYSA-N 2-[(3-fluoropyridin-2-yl)amino]-5-phenylthiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=NC=CC=C1F IGZTXAYDXRHINW-UHFFFAOYSA-N 0.000 claims 1
- PCXZCYZZNIAESG-UHFFFAOYSA-N 2-[(5-cyano-6-methylpyridin-2-yl)amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=C(C#N)C(C)=NC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=C1 PCXZCYZZNIAESG-UHFFFAOYSA-N 0.000 claims 1
- RWKUMUJARRQADT-UHFFFAOYSA-N 2-[(6-methylpyridin-2-yl)amino]-5-(1,3,5-trimethylpyrazol-4-yl)thiophene-3-carboxamide Chemical compound CC1=NN(C)C(C)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C)=N1 RWKUMUJARRQADT-UHFFFAOYSA-N 0.000 claims 1
- GKHCAEWTRATXDH-UHFFFAOYSA-N 2-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=C(C(=CC(=C1)C(C)(C)O)F)C=1SC=CC=1C(=O)N GKHCAEWTRATXDH-UHFFFAOYSA-N 0.000 claims 1
- LZCVAERZJGTQOR-UHFFFAOYSA-N 2-[[2-(morpholin-4-ylmethyl)pyrimidin-4-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC(=CC=2)N2CCOCC2)SC=1NC(N=1)=CC=NC=1CN1CCOCC1 LZCVAERZJGTQOR-UHFFFAOYSA-N 0.000 claims 1
- PYKMSPPGONJSEB-UHFFFAOYSA-N 2-[[5-(1-hydroxyethyl)-6-methylpyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound N1=C(C)C(C(O)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C=NC(=CC=2)N2CCOCC2)S1 PYKMSPPGONJSEB-UHFFFAOYSA-N 0.000 claims 1
- RQZRAIZHBGBECV-UHFFFAOYSA-N 2-[[5-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]-1-phenylthiophene-1-carboxamide Chemical compound N1=CC(C(C)(O)C)=CC=C1NC1=CC=CS1(C(N)=O)C1=CC=CC=C1 RQZRAIZHBGBECV-UHFFFAOYSA-N 0.000 claims 1
- YGWXZHAXTGBCIZ-UHFFFAOYSA-N 2-[[5-[(3,3-difluoroazetidin-1-yl)methyl]-6-methylpyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C=1C=C(CN2CC(F)(F)C2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F YGWXZHAXTGBCIZ-UHFFFAOYSA-N 0.000 claims 1
- PQEKFRLYGFKXQC-UHFFFAOYSA-N 2-[[6-(2-amino-1-morpholin-4-yl-2-oxoethyl)pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(N2CCOCC2)C(N)=O)=N1 PQEKFRLYGFKXQC-UHFFFAOYSA-N 0.000 claims 1
- ANSKLECZUUTFCO-UHFFFAOYSA-N 2-[[6-(2-hydroxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC(=CC=2)N2CCOCC2)SC=1NC(N=1)=CC=CC=1C(CO)N1CCOCC1 ANSKLECZUUTFCO-UHFFFAOYSA-N 0.000 claims 1
- NSEVYBGUGGWQBX-UHFFFAOYSA-N 2-[[6-(2-hydroxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]-5-(6-piperidin-1-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC(=CC=2)N2CCCCC2)SC=1NC(N=1)=CC=CC=1C(CO)N1CCOCC1 NSEVYBGUGGWQBX-UHFFFAOYSA-N 0.000 claims 1
- NOYMKGZALRPHLF-UHFFFAOYSA-N 2-[[6-(2-hydroxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]-5-[6-(4-methylpiperazin-1-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound C1CN(C)CCN1C1=CC=C(C=2SC(NC=3N=C(C=CC=3)C(CO)N3CCOCC3)=C(C(N)=O)C=2)C=N1 NOYMKGZALRPHLF-UHFFFAOYSA-N 0.000 claims 1
- KJUFAHUBLYVJCY-UHFFFAOYSA-N 2-[[6-(2-hydroxyethoxy)pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(OCCO)=N1 KJUFAHUBLYVJCY-UHFFFAOYSA-N 0.000 claims 1
- SJCXMPPVARQLPA-UHFFFAOYSA-N 2-[[6-(2-hydroxypropan-2-yl)pyridazin-3-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C=NC(=CC=2)N2CCOCC2)S1 SJCXMPPVARQLPA-UHFFFAOYSA-N 0.000 claims 1
- LENFRCNTPBYFBD-UHFFFAOYSA-N 2-[[6-(cyclopropylmethoxymethyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC2CC2)=N1 LENFRCNTPBYFBD-UHFFFAOYSA-N 0.000 claims 1
- IIQLBNYKFREGIZ-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(2,4,6-trifluorophenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC(F)=CC=2F)F)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 IIQLBNYKFREGIZ-UHFFFAOYSA-N 0.000 claims 1
- ABAQZGQISQMDSW-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(4-pyrazol-1-ylphenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(=CC=2)N2N=CC=C2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 ABAQZGQISQMDSW-UHFFFAOYSA-N 0.000 claims 1
- RATBCHTZPSYKDP-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(4-pyridin-4-ylphenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(=CC=2)C=2C=CN=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 RATBCHTZPSYKDP-UHFFFAOYSA-N 0.000 claims 1
- HDKNCUABXBFCRH-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC(=CC=2)N2CCOCC2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 HDKNCUABXBFCRH-UHFFFAOYSA-N 0.000 claims 1
- USVVPSCHHYHDEQ-UHFFFAOYSA-N 2-[[6-(pyrrolidin-1-ylmethyl)pyridin-2-yl]amino]-5-(2,4,6-trifluorophenyl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC(F)=CC=2F)F)SC=1NC(N=1)=CC=CC=1CN1CCCC1 USVVPSCHHYHDEQ-UHFFFAOYSA-N 0.000 claims 1
- NQCQWCUKGAKMGQ-UHFFFAOYSA-N 2-[[6-[(2,6-dimethylmorpholin-4-yl)methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1C(C)OC(C)CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 NQCQWCUKGAKMGQ-UHFFFAOYSA-N 0.000 claims 1
- WGXZYGVSFOEXTL-UHFFFAOYSA-N 2-[[6-[(dimethylamino)methyl]pyridin-2-yl]amino]-5-(2-fluorophenyl)thiophene-3-carboxamide Chemical compound CN(C)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)F)C(N)=O)=N1 WGXZYGVSFOEXTL-UHFFFAOYSA-N 0.000 claims 1
- JHULRTOSZFBERA-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-(4-pyrazol-1-ylphenyl)thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)N2N=CC=C2)C(N)=O)=N1 JHULRTOSZFBERA-UHFFFAOYSA-N 0.000 claims 1
- RPFXBVFGCJCSMV-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-[6-(2-methylpyrazol-3-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound CN1N=CC=C1C1=CC=C(C=2SC(NC=3N=C(CN4N=NC(=C4)C(C)(C)O)C=CC=3)=C(C(N)=O)C=2)C=N1 RPFXBVFGCJCSMV-UHFFFAOYSA-N 0.000 claims 1
- HSJOIUICDBWWQF-UHFFFAOYSA-N 2-[[6-[[[2-(cyclopentylamino)-2-oxoethyl]-methylsulfonylamino]methyl]pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN(CC(=O)NC2CCCC2)S(C)(=O)=O)=N1 HSJOIUICDBWWQF-UHFFFAOYSA-N 0.000 claims 1
- LGVWPQJMTXOITE-UHFFFAOYSA-N 2-[[6-[[[2-(cyclopentylamino)-2-oxoethyl]amino]methyl]pyridin-2-yl]amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNCC(=O)NC2CCCC2)=N1 LGVWPQJMTXOITE-UHFFFAOYSA-N 0.000 claims 1
- FLKJCTIFKOQZEN-UHFFFAOYSA-N 2-[[6-[[[2-(dimethylamino)-2-oxoethyl]-methylamino]methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound CN(C)C(=O)CN(C)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 FLKJCTIFKOQZEN-UHFFFAOYSA-N 0.000 claims 1
- UIEOTLXFSCHEBI-UHFFFAOYSA-N 2-[[6-methyl-5-(2,2,2-trifluoro-1-hydroxyethyl)pyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound C1=C(C(O)C(F)(F)F)C(C)=NC(NC2=C(C=C(S2)C=2C=NC(=CC=2)N2CCOCC2)C(N)=O)=C1 UIEOTLXFSCHEBI-UHFFFAOYSA-N 0.000 claims 1
- OHMCKNAPERNNEE-UHFFFAOYSA-N 5-(2-fluorophenyl)-2-[[6-(1-hydroxy-2-morpholin-4-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=CC=2)F)SC=1NC(N=1)=CC=CC=1C(O)CN1CCOCC1 OHMCKNAPERNNEE-UHFFFAOYSA-N 0.000 claims 1
- GJJYWLDBEBKFQB-UHFFFAOYSA-N 5-(2-fluorophenyl)-2-[[6-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC=CC=2)F)C(N)=O)=N1 GJJYWLDBEBKFQB-UHFFFAOYSA-N 0.000 claims 1
- UTEILPATDFSLTD-UHFFFAOYSA-N 5-(4-fluorophenyl)-2-[[6-(pyrrolidin-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(F)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCCC1 UTEILPATDFSLTD-UHFFFAOYSA-N 0.000 claims 1
- QTCHJENJNCTWQV-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 QTCHJENJNCTWQV-UHFFFAOYSA-N 0.000 claims 1
- OIOLDTRJLVTFHK-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-phenylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C=2C=CC=CC=2)=N1 OIOLDTRJLVTFHK-UHFFFAOYSA-N 0.000 claims 1
- BIGZXZXCMQMNLU-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-(dimethylsulfamoylamino)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound CN(C)S(=O)(=O)NC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 BIGZXZXCMQMNLU-UHFFFAOYSA-N 0.000 claims 1
- PVZISPYZJOTTCO-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-[(2-oxo-1,3-oxazolidin-3-yl)methyl]pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2C(OCC2)=O)=N1 PVZISPYZJOTTCO-UHFFFAOYSA-N 0.000 claims 1
- AJFYNGAQMVOSHO-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-[(4-trimethylsilyltriazol-1-yl)methyl]pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2N=NC(=C2)[Si](C)(C)C)=N1 AJFYNGAQMVOSHO-UHFFFAOYSA-N 0.000 claims 1
- PRAQEBZQZFOIIF-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-[[(1-methylpyrazol-4-yl)methylamino]methyl]pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1CNCC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 PRAQEBZQZFOIIF-UHFFFAOYSA-N 0.000 claims 1
- ZVLDEOVUDJHEJE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-(2-hydroxypropan-2-yl)-6-methoxypyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=C(C(C)(C)O)C(OC)=NC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=C1 ZVLDEOVUDJHEJE-UHFFFAOYSA-N 0.000 claims 1
- SEKWGFMIPNCQJE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC(N=C1)=CC=C1CN1CCOCC1 SEKWGFMIPNCQJE-UHFFFAOYSA-N 0.000 claims 1
- WZAOVRPPMBXYLY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-(triazol-2-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC(N=C1)=CC=C1CN1N=CC=N1 WZAOVRPPMBXYLY-UHFFFAOYSA-N 0.000 claims 1
- RFWWFPKAARCEKS-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC(N=C1)=CC=C1CN1CCS(=O)(=O)CC1 RFWWFPKAARCEKS-UHFFFAOYSA-N 0.000 claims 1
- LKSDQAGHORONDR-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[(3,3-difluoropyrrolidin-1-yl)methyl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(CN2CC(F)(F)CC2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F LKSDQAGHORONDR-UHFFFAOYSA-N 0.000 claims 1
- OWPFLYMRBFPENE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[5-(2-methoxyethyl)-1,3,4-oxadiazol-2-yl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1C(CCOC)=NN=C1C(C(=N1)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2F)C(C)(C)O)F)S1 OWPFLYMRBFPENE-UHFFFAOYSA-N 0.000 claims 1
- RQBVMGXZVAHIOR-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(1-methylpyrazol-4-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 RQBVMGXZVAHIOR-UHFFFAOYSA-N 0.000 claims 1
- LJKRXPKIXGVXFP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(1-methyltriazol-4-yl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NN(C)C=C1C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 LJKRXPKIXGVXFP-UHFFFAOYSA-N 0.000 claims 1
- HPWWQSIZDFEZEZ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2CCOCC2)=N1 HPWWQSIZDFEZEZ-UHFFFAOYSA-N 0.000 claims 1
- DDKYWOLXTNPWDW-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxy-2-methylpropyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 DDKYWOLXTNPWDW-UHFFFAOYSA-N 0.000 claims 1
- JSGXUEZALPLUGV-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxypropan-2-yl)pyridazin-3-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C(C)(C)O)N=N1 JSGXUEZALPLUGV-UHFFFAOYSA-N 0.000 claims 1
- MRSLPPASSSVJDM-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-methylsulfonylethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCCS(C)(=O)=O)=N1 MRSLPPASSSVJDM-UHFFFAOYSA-N 0.000 claims 1
- POFZLKWHWDWZQB-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(morpholine-4-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CCOCC2)=N1 POFZLKWHWDWZQB-UHFFFAOYSA-N 0.000 claims 1
- GXCFYVICGSIVIL-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(pyrazol-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=CC=C2)=N1 GXCFYVICGSIVIL-UHFFFAOYSA-N 0.000 claims 1
- SQEKXKZOWATCPH-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1,1-dioxothian-4-yl)-hydroxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(O)C2CCS(=O)(=O)CC2)=N1 SQEKXKZOWATCPH-UHFFFAOYSA-N 0.000 claims 1
- JSJGWPPVRNJINI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methyl-1,2,4-triazol-3-yl)sulfonylmethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1C=NN=C1S(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 JSJGWPPVRNJINI-UHFFFAOYSA-N 0.000 claims 1
- OGBFAQMEIJFQCN-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[1-(1,1-dioxo-1,4-thiazinan-4-yl)-2-hydroxyethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2CCS(=O)(=O)CC2)=N1 OGBFAQMEIJFQCN-UHFFFAOYSA-N 0.000 claims 1
- SRVQWAROKOYWAH-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(methylamino)-1-morpholin-4-yl-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=NC=1C(C(=O)NC)N1CCOCC1 SRVQWAROKOYWAH-UHFFFAOYSA-N 0.000 claims 1
- MLMUUOHTWWJYEI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(3,5-dimethyl-1,2-oxazol-4-yl)methoxy]ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC1=NOC(C)=C1COCCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 MLMUUOHTWWJYEI-UHFFFAOYSA-N 0.000 claims 1
- SUPDAYWSMNSFJL-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-hydroxy-2-(1-methylpyrazol-4-yl)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1C(O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 SUPDAYWSMNSFJL-UHFFFAOYSA-N 0.000 claims 1
- CLTRUXVANPHLFC-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(1-methylpyrazol-3-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1C=CC(NCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 CLTRUXVANPHLFC-UHFFFAOYSA-N 0.000 claims 1
- WZRUDNQOKQXTCE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(2-hydroxy-2-methylpropanoyl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)C(=O)NCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 WZRUDNQOKQXTCE-UHFFFAOYSA-N 0.000 claims 1
- FVMVBCDFJKLBEJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(methylamino)-2-oxoethyl]sulfonylmethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CNC(=O)CS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 FVMVBCDFJKLBEJ-UHFFFAOYSA-N 0.000 claims 1
- BQAGGROWKCXVBM-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-[(dimethylamino)methyl]triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NC(CN(C)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 BQAGGROWKCXVBM-UHFFFAOYSA-N 0.000 claims 1
- GXIAACJIVGCQIW-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(2-methylpyrimidin-4-yl)amino]thiophene-3-carboxamide Chemical compound CC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 GXIAACJIVGCQIW-UHFFFAOYSA-N 0.000 claims 1
- XQBTYRWMOUCEFA-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-(morpholin-4-ylmethyl)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2CCOCC2)=N1 XQBTYRWMOUCEFA-UHFFFAOYSA-N 0.000 claims 1
- HZPVZGAQJYKNKJ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxy-3-methyl-1-morpholin-4-ylbutyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=NC=1C(C(O)C(C)C)N1CCOCC1 HZPVZGAQJYKNKJ-UHFFFAOYSA-N 0.000 claims 1
- MGCMKIXGAMUSHJ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(3,3,3-trifluoropropoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCCC(F)(F)F)=N1 MGCMKIXGAMUSHJ-UHFFFAOYSA-N 0.000 claims 1
- JKCFCILIFXRDHX-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(oxolan-3-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC2COCC2)=N1 JKCFCILIFXRDHX-UHFFFAOYSA-N 0.000 claims 1
- JTDWJCNDIKJIBI-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(pyrimidin-2-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2N=CC=CN=2)=N1 JTDWJCNDIKJIBI-UHFFFAOYSA-N 0.000 claims 1
- NEKRFSYOYBIZAO-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1-methyltetrazol-5-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1N=NN=C1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 NEKRFSYOYBIZAO-UHFFFAOYSA-N 0.000 claims 1
- HQFRBAXDCWEYQY-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-oxomorpholin-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(COCC2)=O)=N1 HQFRBAXDCWEYQY-UHFFFAOYSA-N 0.000 claims 1
- LDQBCSBMWWTPGA-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methylpiperazin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1CN(C)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 LDQBCSBMWWTPGA-UHFFFAOYSA-N 0.000 claims 1
- SRGCFXHEEFVINQ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[1-hydroxy-1-(1-methyltriazol-4-yl)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NN(C)C=C1C(C)(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 SRGCFXHEEFVINQ-UHFFFAOYSA-N 0.000 claims 1
- VPLFXZAJDRGXQF-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(4-methylpiperazin-1-yl)ethoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1CN(C)CCN1CCOCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 VPLFXZAJDRGXQF-UHFFFAOYSA-N 0.000 claims 1
- YCOBDRMUWPLFHR-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[6-(hydroxymethyl)-3-azabicyclo[3.1.0]hexane-3-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CC3C(CO)C3C2)=N1 YCOBDRMUWPLFHR-UHFFFAOYSA-N 0.000 claims 1
- WJOIORMRNGPLDN-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[2-(2-hydroxyethyl)morpholin-4-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CC(CCO)OCC2)=N1 WJOIORMRNGPLDN-UHFFFAOYSA-N 0.000 claims 1
- XVLVEQKOJKWSAE-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[3-(hydroxymethyl)morpholin-4-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(COCC2)CO)=N1 XVLVEQKOJKWSAE-UHFFFAOYSA-N 0.000 claims 1
- FONRMYRECTWPOJ-UHFFFAOYSA-N 5-[4-(1,1-dioxo-1,4-thiazinan-4-yl)phenyl]-2-[[2-(morpholin-4-ylmethyl)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(=CC=2)N2CCS(=O)(=O)CC2)SC=1NC(N=1)=CC=NC=1CN1CCOCC1 FONRMYRECTWPOJ-UHFFFAOYSA-N 0.000 claims 1
- DKNIXUJEVVCIRT-UHFFFAOYSA-N 5-[4-(2-cyanopropan-2-yl)phenyl]-2-[(2-methylpyrimidin-4-yl)amino]thiophene-3-carboxamide Chemical compound CC1=NC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)C#N)C(N)=O)=N1 DKNIXUJEVVCIRT-UHFFFAOYSA-N 0.000 claims 1
- TWVMLDGNJOVFFF-UHFFFAOYSA-N 5-[4-(2-cyanopropan-2-yl)phenyl]-2-[(2-oxo-1h-pyrimidin-6-yl)amino]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(C#N)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=NC(=O)NC=C1 TWVMLDGNJOVFFF-UHFFFAOYSA-N 0.000 claims 1
- BCMDTOXGFFLULX-UHFFFAOYSA-N 5-[4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCOCC2)=N1 BCMDTOXGFFLULX-UHFFFAOYSA-N 0.000 claims 1
- BXRNOMYJRFUOHK-UHFFFAOYSA-N 5-[6-(2-cyanopropan-2-yl)pyridin-3-yl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=NC(=CC=2)C(C)(C)C#N)C(N)=O)=N1 BXRNOMYJRFUOHK-UHFFFAOYSA-N 0.000 claims 1
- ZDASXQPBOMHLLW-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-hydroxyethyl)-n-methylpyridine-2-carboxamide Chemical compound OCCN(C)C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 ZDASXQPBOMHLLW-UHFFFAOYSA-N 0.000 claims 1
- DDJZTDMTSBDDMB-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-hydroxyethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCCO)=N1 DDJZTDMTSBDDMB-UHFFFAOYSA-N 0.000 claims 1
- KGJZWVLVGCKKHQ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxan-4-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2CCOCC2)=N1 KGJZWVLVGCKKHQ-UHFFFAOYSA-N 0.000 claims 1
- GIOLHTGDTQJENQ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxolan-2-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2OCCC2)=N1 GIOLHTGDTQJENQ-UHFFFAOYSA-N 0.000 claims 1
- ZUHZFTROKCJPQJ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(1,1-dioxothiolan-3-yl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NC2CS(=O)(=O)CC2)=N1 ZUHZFTROKCJPQJ-UHFFFAOYSA-N 0.000 claims 1
- WBFUQZCJEQEURV-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(cyclopropylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2CC2)=N1 WBFUQZCJEQEURV-UHFFFAOYSA-N 0.000 claims 1
- SJDAAZUSWYRNHY-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxan-4-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2CCOCC2)=N1 SJDAAZUSWYRNHY-UHFFFAOYSA-N 0.000 claims 1
- HIUMYOWDLVRANS-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxetan-2-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2OCC2)=N1 HIUMYOWDLVRANS-UHFFFAOYSA-N 0.000 claims 1
- RPNLXXAOPPROHM-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(oxolan-2-ylmethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCC2OCCC2)=N1 RPNLXXAOPPROHM-UHFFFAOYSA-N 0.000 claims 1
- HBNCMACDTQGQRZ-LBPRGKRZSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-[(2s)-2-hydroxypropyl]pyridine-2-carboxamide Chemical compound C[C@H](O)CNC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 HBNCMACDTQGQRZ-LBPRGKRZSA-N 0.000 claims 1
- OEMXSIHIDGXSGF-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-[2-(oxan-4-yl)ethyl]pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCCC2CCOCC2)=N1 OEMXSIHIDGXSGF-UHFFFAOYSA-N 0.000 claims 1
- WRQURYHKPBRKMJ-UHFFFAOYSA-N benzyl n-[[6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-n-[2-(2-methylsulfonylethylamino)-2-oxoethyl]carbamate Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN(CC(=O)NCCS(C)(=O)=O)C(=O)OCC=2C=CC=CC=2)=N1 WRQURYHKPBRKMJ-UHFFFAOYSA-N 0.000 claims 1
- GTCAXTIRRLKXRU-UHFFFAOYSA-N carbamic acid methyl ester Natural products COC(N)=O GTCAXTIRRLKXRU-UHFFFAOYSA-N 0.000 claims 1
- SKUOSYZHLRRQPS-UHFFFAOYSA-N methyl 2-[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]-2-morpholin-4-ylacetate Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=NC=1C(C(=O)OC)N1CCOCC1 SKUOSYZHLRRQPS-UHFFFAOYSA-N 0.000 claims 1
- WGOIPJZXHXZRKL-UHFFFAOYSA-N methyl 4-[[6-[[3-carbamoyl-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC)CCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)O)C(N)=O)=N1 WGOIPJZXHXZRKL-UHFFFAOYSA-N 0.000 claims 1
- OKMXJBRJYDLDPM-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-1,3-oxazole-5-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC(=O)C=2OC=NC=2)=N1 OKMXJBRJYDLDPM-UHFFFAOYSA-N 0.000 claims 1
- VLRVJOLWCLDEAB-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-1-methylpyrazole-3-carboxamide Chemical compound CN1C=CC(C(=O)NCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 VLRVJOLWCLDEAB-UHFFFAOYSA-N 0.000 claims 1
- OTHYUBGCZSIZDL-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-1h-pyrazole-5-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC(=O)C=2NN=CC=2)=N1 OTHYUBGCZSIZDL-UHFFFAOYSA-N 0.000 claims 1
- BFQGAHZJRHMEOR-UHFFFAOYSA-N n-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]pyrimidine-5-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC(=O)C=2C=NC=NC=2)=N1 BFQGAHZJRHMEOR-UHFFFAOYSA-N 0.000 claims 1
- 229960003966 nicotinamide Drugs 0.000 claims 1
- 235000005152 nicotinamide Nutrition 0.000 claims 1
- 239000011570 nicotinamide Substances 0.000 claims 1
- BQLVTEQZYJUFDZ-UHFFFAOYSA-N tert-butyl 4-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]-hydroxymethyl]piperazine-1-carboxylate Chemical compound C1CN(C(=O)OC(C)(C)C)CCN1C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 BQLVTEQZYJUFDZ-UHFFFAOYSA-N 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 101
- 239000000203 mixture Substances 0.000 abstract description 66
- 101000997832 Homo sapiens Tyrosine-protein kinase JAK2 Proteins 0.000 abstract description 22
- 102100033444 Tyrosine-protein kinase JAK2 Human genes 0.000 abstract description 20
- 230000002401 inhibitory effect Effects 0.000 abstract description 16
- 101000934996 Homo sapiens Tyrosine-protein kinase JAK3 Proteins 0.000 abstract description 14
- 102100025387 Tyrosine-protein kinase JAK3 Human genes 0.000 abstract description 13
- 101000844245 Homo sapiens Non-receptor tyrosine-protein kinase TYK2 Proteins 0.000 abstract description 11
- 102100032028 Non-receptor tyrosine-protein kinase TYK2 Human genes 0.000 abstract description 10
- 230000000694 effects Effects 0.000 abstract description 10
- 101000997835 Homo sapiens Tyrosine-protein kinase JAK1 Proteins 0.000 abstract description 4
- 102100033438 Tyrosine-protein kinase JAK1 Human genes 0.000 abstract description 4
- 101000600756 Homo sapiens 3-phosphoinositide-dependent protein kinase 1 Proteins 0.000 abstract 2
- 101001117146 Homo sapiens [Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 1, mitochondrial Proteins 0.000 abstract 2
- 102100024148 [Pyruvate dehydrogenase (acetyl-transferring)] kinase isozyme 1, mitochondrial Human genes 0.000 abstract 2
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 169
- 239000000243 solution Substances 0.000 description 82
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 72
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 69
- 239000000543 intermediate Substances 0.000 description 69
- 235000019439 ethyl acetate Nutrition 0.000 description 67
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 62
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 59
- 239000007787 solid Substances 0.000 description 59
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 58
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 57
- 229910001868 water Inorganic materials 0.000 description 56
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 53
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 48
- 238000006243 chemical reaction Methods 0.000 description 48
- 238000005160 1H NMR spectroscopy Methods 0.000 description 46
- 239000003795 chemical substances by application Substances 0.000 description 39
- 239000012267 brine Substances 0.000 description 38
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 38
- 239000012044 organic layer Substances 0.000 description 36
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 34
- 210000004027 cell Anatomy 0.000 description 32
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 31
- 239000011541 reaction mixture Substances 0.000 description 31
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 29
- 239000007858 starting material Substances 0.000 description 28
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 26
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 24
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 23
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 22
- 239000010410 layer Substances 0.000 description 22
- 239000000725 suspension Substances 0.000 description 22
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 21
- 229960004132 diethyl ether Drugs 0.000 description 21
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 21
- 238000003818 flash chromatography Methods 0.000 description 20
- 229920006395 saturated elastomer Polymers 0.000 description 19
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Divinylene sulfide Natural products C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 18
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 18
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 17
- 238000000746 purification Methods 0.000 description 17
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 16
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 15
- 238000001914 filtration Methods 0.000 description 15
- 239000000047 product Substances 0.000 description 15
- 108091008598 receptor tyrosine kinases Proteins 0.000 description 15
- 102000027426 receptor tyrosine kinases Human genes 0.000 description 15
- 229910052938 sodium sulfate Inorganic materials 0.000 description 15
- 238000003756 stirring Methods 0.000 description 15
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 14
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 14
- 235000017557 sodium bicarbonate Nutrition 0.000 description 14
- 235000011152 sodium sulphate Nutrition 0.000 description 14
- SNTWKPAKVQFCCF-UHFFFAOYSA-N 2,3-dihydro-1h-triazole Chemical compound N1NC=CN1 SNTWKPAKVQFCCF-UHFFFAOYSA-N 0.000 description 13
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 13
- 238000010898 silica gel chromatography Methods 0.000 description 13
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 12
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 12
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 12
- 230000033115 angiogenesis Effects 0.000 description 12
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 12
- 239000003921 oil Substances 0.000 description 12
- 235000019198 oils Nutrition 0.000 description 12
- 230000009467 reduction Effects 0.000 description 12
- 239000002904 solvent Substances 0.000 description 12
- UUIXLRCZHBPZNE-UHFFFAOYSA-N 5-bromo-2-nitrothiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(Br)SC=1[N+]([O-])=O UUIXLRCZHBPZNE-UHFFFAOYSA-N 0.000 description 11
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 11
- 229910052786 argon Inorganic materials 0.000 description 11
- 229940079593 drug Drugs 0.000 description 11
- 239000000284 extract Substances 0.000 description 11
- 230000002829 reductive effect Effects 0.000 description 11
- LQZMLBORDGWNPD-UHFFFAOYSA-N N-iodosuccinimide Chemical compound IN1C(=O)CCC1=O LQZMLBORDGWNPD-UHFFFAOYSA-N 0.000 description 10
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 10
- 239000002585 base Substances 0.000 description 10
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 10
- 239000000706 filtrate Substances 0.000 description 10
- 229910052763 palladium Inorganic materials 0.000 description 10
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 10
- 229930192474 thiophene Natural products 0.000 description 10
- 206010025323 Lymphomas Diseases 0.000 description 9
- 239000000556 agonist Substances 0.000 description 9
- 102000006495 integrins Human genes 0.000 description 9
- 108010044426 integrins Proteins 0.000 description 9
- 239000000463 material Substances 0.000 description 9
- 229910000029 sodium carbonate Inorganic materials 0.000 description 9
- 229940124597 therapeutic agent Drugs 0.000 description 9
- GLQWRXYOTXRDNH-UHFFFAOYSA-N thiophen-2-amine Chemical compound NC1=CC=CS1 GLQWRXYOTXRDNH-UHFFFAOYSA-N 0.000 description 9
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 8
- 108010016731 PPAR gamma Proteins 0.000 description 8
- 102100038825 Peroxisome proliferator-activated receptor gamma Human genes 0.000 description 8
- 239000002253 acid Substances 0.000 description 8
- 239000004480 active ingredient Substances 0.000 description 8
- 208000009956 adenocarcinoma Diseases 0.000 description 8
- 239000004037 angiogenesis inhibitor Substances 0.000 description 8
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 8
- 125000004432 carbon atom Chemical group C* 0.000 description 8
- 238000001816 cooling Methods 0.000 description 8
- 201000010099 disease Diseases 0.000 description 8
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 8
- 210000004072 lung Anatomy 0.000 description 8
- 229940122361 Bisphosphonate Drugs 0.000 description 7
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 description 7
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 7
- 241000699670 Mus sp. Species 0.000 description 7
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 7
- 102000007399 Nuclear hormone receptor Human genes 0.000 description 7
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 7
- 102100038280 Prostaglandin G/H synthase 2 Human genes 0.000 description 7
- 108050003267 Prostaglandin G/H synthase 2 Proteins 0.000 description 7
- 206010039491 Sarcoma Diseases 0.000 description 7
- 125000002619 bicyclic group Chemical group 0.000 description 7
- 150000004663 bisphosphonates Chemical class 0.000 description 7
- 230000004663 cell proliferation Effects 0.000 description 7
- 239000002254 cytotoxic agent Substances 0.000 description 7
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 7
- 235000014113 dietary fatty acids Nutrition 0.000 description 7
- 150000002148 esters Chemical class 0.000 description 7
- 229930195729 fatty acid Natural products 0.000 description 7
- 239000000194 fatty acid Substances 0.000 description 7
- 125000000325 methylidene group Chemical group [H]C([H])=* 0.000 description 7
- 239000000546 pharmaceutical excipient Substances 0.000 description 7
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 7
- 239000002244 precipitate Substances 0.000 description 7
- 238000010992 reflux Methods 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- 239000003558 transferase inhibitor Substances 0.000 description 7
- SYOANZBNGDEJFH-UHFFFAOYSA-N 2,5-dihydro-1h-triazole Chemical compound C1NNN=C1 SYOANZBNGDEJFH-UHFFFAOYSA-N 0.000 description 6
- ROIMNSWDOJCBFR-UHFFFAOYSA-N 2-iodothiophene Chemical compound IC1=CC=CS1 ROIMNSWDOJCBFR-UHFFFAOYSA-N 0.000 description 6
- MSXVEPNJUHWQHW-UHFFFAOYSA-N 2-methylbutan-2-ol Chemical compound CCC(C)(C)O MSXVEPNJUHWQHW-UHFFFAOYSA-N 0.000 description 6
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 6
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 6
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 230000002378 acidificating effect Effects 0.000 description 6
- 239000003886 aromatase inhibitor Substances 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 230000012820 cell cycle checkpoint Effects 0.000 description 6
- 231100000433 cytotoxic Toxicity 0.000 description 6
- 230000001472 cytotoxic effect Effects 0.000 description 6
- UAOMVDZJSHZZME-UHFFFAOYSA-N diisopropylamine Chemical compound CC(C)NC(C)C UAOMVDZJSHZZME-UHFFFAOYSA-N 0.000 description 6
- 239000002834 estrogen receptor modulator Substances 0.000 description 6
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 6
- 150000002431 hydrogen Chemical group 0.000 description 6
- 239000005457 ice water Substances 0.000 description 6
- 235000019341 magnesium sulphate Nutrition 0.000 description 6
- 230000007246 mechanism Effects 0.000 description 6
- 230000036457 multidrug resistance Effects 0.000 description 6
- 239000000041 non-steroidal anti-inflammatory agent Substances 0.000 description 6
- 231100000252 nontoxic Toxicity 0.000 description 6
- 230000003000 nontoxic effect Effects 0.000 description 6
- 230000037361 pathway Effects 0.000 description 6
- 108091008765 peroxisome proliferator-activated receptors β/δ Proteins 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 102000027483 retinoid hormone receptors Human genes 0.000 description 6
- 108091008679 retinoid hormone receptors Proteins 0.000 description 6
- 239000000849 selective androgen receptor modulator Substances 0.000 description 6
- 229960005267 tositumomab Drugs 0.000 description 6
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 5
- YFTHTJAPODJVSL-UHFFFAOYSA-N 2-(1-benzothiophen-5-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane Chemical compound O1C(C)(C)C(C)(C)OB1C1=CC=C(SC=C2)C2=C1 YFTHTJAPODJVSL-UHFFFAOYSA-N 0.000 description 5
- DWUVPXNRZKQAHM-UHFFFAOYSA-N 2-amino-5-(2-fluorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=CC=C1F DWUVPXNRZKQAHM-UHFFFAOYSA-N 0.000 description 5
- GMHJYWCFYIVPEY-UHFFFAOYSA-N 3-thiophen-2-ylpyridin-2-amine Chemical compound NC1=NC=CC=C1C1=CC=CS1 GMHJYWCFYIVPEY-UHFFFAOYSA-N 0.000 description 5
- IKHGUXGNUITLKF-UHFFFAOYSA-N Acetaldehyde Natural products CC=O IKHGUXGNUITLKF-UHFFFAOYSA-N 0.000 description 5
- 206010006187 Breast cancer Diseases 0.000 description 5
- COVZYZSDYWQREU-UHFFFAOYSA-N Busulfan Chemical compound CS(=O)(=O)OCCCCOS(C)(=O)=O COVZYZSDYWQREU-UHFFFAOYSA-N 0.000 description 5
- 201000009030 Carcinoma Diseases 0.000 description 5
- 108010011536 PTEN Phosphohydrolase Proteins 0.000 description 5
- 102000014160 PTEN Phosphohydrolase Human genes 0.000 description 5
- 102100038824 Peroxisome proliferator-activated receptor delta Human genes 0.000 description 5
- 108091000080 Phosphotransferase Proteins 0.000 description 5
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 5
- 108010017324 STAT3 Transcription Factor Proteins 0.000 description 5
- 102100024040 Signal transducer and activator of transcription 3 Human genes 0.000 description 5
- 108020004459 Small interfering RNA Proteins 0.000 description 5
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 5
- NKANXQFJJICGDU-QPLCGJKRSA-N Tamoxifen Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN(C)C)=CC=1)/C1=CC=CC=C1 NKANXQFJJICGDU-QPLCGJKRSA-N 0.000 description 5
- NAVMQTYZDKMPEU-UHFFFAOYSA-N Targretin Chemical compound CC1=CC(C(CCC2(C)C)(C)C)=C2C=C1C(=C)C1=CC=C(C(O)=O)C=C1 NAVMQTYZDKMPEU-UHFFFAOYSA-N 0.000 description 5
- 229940123468 Transferase inhibitor Drugs 0.000 description 5
- 206010047700 Vomiting Diseases 0.000 description 5
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 5
- 230000001028 anti-proliverative effect Effects 0.000 description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 5
- 239000012455 biphasic mixture Substances 0.000 description 5
- 210000000481 breast Anatomy 0.000 description 5
- 238000006555 catalytic reaction Methods 0.000 description 5
- 239000007859 condensation product Substances 0.000 description 5
- 230000008878 coupling Effects 0.000 description 5
- 238000010168 coupling process Methods 0.000 description 5
- 238000005859 coupling reaction Methods 0.000 description 5
- DGJMPUGMZIKDRO-UHFFFAOYSA-N cyanoacetamide Chemical compound NC(=O)CC#N DGJMPUGMZIKDRO-UHFFFAOYSA-N 0.000 description 5
- 239000000824 cytostatic agent Substances 0.000 description 5
- 229960000975 daunorubicin Drugs 0.000 description 5
- 150000004665 fatty acids Chemical class 0.000 description 5
- 239000003540 gamma secretase inhibitor Substances 0.000 description 5
- MZRVEZGGRBJDDB-UHFFFAOYSA-N n-Butyllithium Substances [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 5
- 229940021182 non-steroidal anti-inflammatory drug Drugs 0.000 description 5
- 239000012074 organic phase Substances 0.000 description 5
- 239000012071 phase Substances 0.000 description 5
- 102000020233 phosphotransferase Human genes 0.000 description 5
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 239000003755 preservative agent Substances 0.000 description 5
- 210000002307 prostate Anatomy 0.000 description 5
- 108090000623 proteins and genes Proteins 0.000 description 5
- 102000005962 receptors Human genes 0.000 description 5
- 108020003175 receptors Proteins 0.000 description 5
- 108010038379 sargramostim Proteins 0.000 description 5
- 230000019491 signal transduction Effects 0.000 description 5
- 230000011664 signaling Effects 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- 206010041823 squamous cell carcinoma Diseases 0.000 description 5
- 239000011593 sulfur Substances 0.000 description 5
- 230000007755 survival signaling Effects 0.000 description 5
- 239000003765 sweetening agent Substances 0.000 description 5
- 238000003786 synthesis reaction Methods 0.000 description 5
- 239000003826 tablet Substances 0.000 description 5
- WYWHKKSPHMUBEB-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 5
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 4
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- MLDQJTXFUGDVEO-UHFFFAOYSA-N BAY-43-9006 Chemical compound C1=NC(C(=O)NC)=CC(OC=2C=CC(NC(=O)NC=3C=C(C(Cl)=CC=3)C(F)(F)F)=CC=2)=C1 MLDQJTXFUGDVEO-UHFFFAOYSA-N 0.000 description 4
- 208000026310 Breast neoplasm Diseases 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 description 4
- 102000004127 Cytokines Human genes 0.000 description 4
- 108090000695 Cytokines Proteins 0.000 description 4
- 206010012689 Diabetic retinopathy Diseases 0.000 description 4
- 208000032027 Essential Thrombocythemia Diseases 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 4
- 108010069236 Goserelin Proteins 0.000 description 4
- 102100039619 Granulocyte colony-stimulating factor Human genes 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- 206010048643 Hypereosinophilic syndrome Diseases 0.000 description 4
- 102000010638 Kinesin Human genes 0.000 description 4
- 108010063296 Kinesin Proteins 0.000 description 4
- 206010024612 Lipoma Diseases 0.000 description 4
- XOGTZOOQQBDUSI-UHFFFAOYSA-M Mesna Chemical compound [Na+].[O-]S(=O)(=O)CCS XOGTZOOQQBDUSI-UHFFFAOYSA-M 0.000 description 4
- 206010027476 Metastases Diseases 0.000 description 4
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 4
- 208000034578 Multiple myelomas Diseases 0.000 description 4
- 208000037538 Myelomonocytic Juvenile Leukemia Diseases 0.000 description 4
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 4
- 206010035226 Plasma cell myeloma Diseases 0.000 description 4
- 102000004022 Protein-Tyrosine Kinases Human genes 0.000 description 4
- 108090000412 Protein-Tyrosine Kinases Proteins 0.000 description 4
- 201000008736 Systemic mastocytosis Diseases 0.000 description 4
- 102000004357 Transferases Human genes 0.000 description 4
- 108090000992 Transferases Proteins 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical class OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 208000017733 acquired polycythemia vera Diseases 0.000 description 4
- 206010064930 age-related macular degeneration Diseases 0.000 description 4
- BHELZAPQIKSEDF-UHFFFAOYSA-N allyl bromide Chemical compound BrCC=C BHELZAPQIKSEDF-UHFFFAOYSA-N 0.000 description 4
- 208000007502 anemia Diseases 0.000 description 4
- 239000005557 antagonist Substances 0.000 description 4
- 239000002111 antiemetic agent Substances 0.000 description 4
- 229940125683 antiemetic agent Drugs 0.000 description 4
- 239000002246 antineoplastic agent Substances 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 4
- 235000006708 antioxidants Nutrition 0.000 description 4
- 239000007900 aqueous suspension Substances 0.000 description 4
- 239000012300 argon atmosphere Substances 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- 210000004204 blood vessel Anatomy 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- GXJABQQUPOEUTA-RDJZCZTQSA-N bortezomib Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)B(O)O)NC(=O)C=1N=CC=NC=1)C1=CC=CC=C1 GXJABQQUPOEUTA-RDJZCZTQSA-N 0.000 description 4
- 239000003054 catalyst Substances 0.000 description 4
- RZEKVGVHFLEQIL-UHFFFAOYSA-N celecoxib Chemical compound C1=CC(C)=CC=C1C1=CC(C(F)(F)F)=NN1C1=CC=C(S(N)(=O)=O)C=C1 RZEKVGVHFLEQIL-UHFFFAOYSA-N 0.000 description 4
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 4
- 210000001072 colon Anatomy 0.000 description 4
- 239000003086 colorant Substances 0.000 description 4
- 239000012043 crude product Substances 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 239000002270 dispersing agent Substances 0.000 description 4
- 229960004679 doxorubicin Drugs 0.000 description 4
- NOTIQUSPUUHHEH-UXOVVSIBSA-N dromostanolone propionate Chemical compound C([C@@H]1CC2)C(=O)[C@H](C)C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](OC(=O)CC)[C@@]2(C)CC1 NOTIQUSPUUHHEH-UXOVVSIBSA-N 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- 235000013355 food flavoring agent Nutrition 0.000 description 4
- 235000003599 food sweetener Nutrition 0.000 description 4
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 description 4
- 229910000041 hydrogen chloride Inorganic materials 0.000 description 4
- YLMAHDNUQAMNNX-UHFFFAOYSA-N imatinib methanesulfonate Chemical compound CS(O)(=O)=O.C1CN(C)CCN1CC1=CC=C(C(=O)NC=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)C=C1 YLMAHDNUQAMNNX-UHFFFAOYSA-N 0.000 description 4
- 238000002347 injection Methods 0.000 description 4
- 239000007924 injection Substances 0.000 description 4
- 238000001990 intravenous administration Methods 0.000 description 4
- 229960004592 isopropanol Drugs 0.000 description 4
- 201000005992 juvenile myelomonocytic leukemia Diseases 0.000 description 4
- 208000032839 leukemia Diseases 0.000 description 4
- 239000003446 ligand Substances 0.000 description 4
- KWGKDLIKAYFUFQ-UHFFFAOYSA-M lithium chloride Chemical compound [Li+].[Cl-] KWGKDLIKAYFUFQ-UHFFFAOYSA-M 0.000 description 4
- 208000002780 macular degeneration Diseases 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 201000001441 melanoma Diseases 0.000 description 4
- 230000009401 metastasis Effects 0.000 description 4
- 206010061289 metastatic neoplasm Diseases 0.000 description 4
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 4
- 230000000394 mitotic effect Effects 0.000 description 4
- 239000012452 mother liquor Substances 0.000 description 4
- 239000002742 neurokinin 1 receptor antagonist Substances 0.000 description 4
- 208000004235 neutropenia Diseases 0.000 description 4
- WRUUGTRCQOWXEG-UHFFFAOYSA-N pamidronate Chemical compound NCCC(O)(P(O)(O)=O)P(O)(O)=O WRUUGTRCQOWXEG-UHFFFAOYSA-N 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- 229940096701 plain lipid modifying drug hmg coa reductase inhibitors Drugs 0.000 description 4
- 208000037244 polycythemia vera Diseases 0.000 description 4
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 4
- 230000004044 response Effects 0.000 description 4
- 238000004007 reversed phase HPLC Methods 0.000 description 4
- 239000003419 rna directed dna polymerase inhibitor Substances 0.000 description 4
- 239000000375 suspending agent Substances 0.000 description 4
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 4
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 4
- 230000000699 topical effect Effects 0.000 description 4
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 4
- 239000000080 wetting agent Substances 0.000 description 4
- XRASPMIURGNCCH-UHFFFAOYSA-N zoledronic acid Chemical compound OP(=O)(O)C(P(O)(O)=O)(O)CN1C=CN=C1 XRASPMIURGNCCH-UHFFFAOYSA-N 0.000 description 4
- BMKDZUISNHGIBY-ZETCQYMHSA-N (+)-dexrazoxane Chemical compound C([C@H](C)N1CC(=O)NC(=O)C1)N1CC(=O)NC(=O)C1 BMKDZUISNHGIBY-ZETCQYMHSA-N 0.000 description 3
- NWZSZGALRFJKBT-KNIFDHDWSA-N (2s)-2,6-diaminohexanoic acid;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.NCCCC[C@H](N)C(O)=O NWZSZGALRFJKBT-KNIFDHDWSA-N 0.000 description 3
- VEEGZPWAAPPXRB-BJMVGYQFSA-N (3e)-3-(1h-imidazol-5-ylmethylidene)-1h-indol-2-one Chemical compound O=C1NC2=CC=CC=C2\C1=C/C1=CN=CN1 VEEGZPWAAPPXRB-BJMVGYQFSA-N 0.000 description 3
- PQQBXXXUTBOSEE-UHFFFAOYSA-N 1,4-dichloro-2-(2-methoxyethenyl)benzene Chemical compound COC=CC1=CC(Cl)=CC=C1Cl PQQBXXXUTBOSEE-UHFFFAOYSA-N 0.000 description 3
- OISVCGZHLKNMSJ-UHFFFAOYSA-N 2,6-dimethylpyridine Chemical compound CC1=CC=CC(C)=N1 OISVCGZHLKNMSJ-UHFFFAOYSA-N 0.000 description 3
- QOMHNYHIEQMZKG-UHFFFAOYSA-N 2-[[5-(2-hydroxypropan-2-yl)pyridin-2-yl]amino]-5-phenylthiophene-3-carboxamide Chemical compound N1=CC(C(C)(O)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C=CC=CC=2)S1 QOMHNYHIEQMZKG-UHFFFAOYSA-N 0.000 description 3
- FPWBGQCTDHRWHJ-UHFFFAOYSA-N 2-amino-5-(2,4-difluorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(F)C=C1F FPWBGQCTDHRWHJ-UHFFFAOYSA-N 0.000 description 3
- WHZIZZOTISTHCT-UHFFFAOYSA-N 2-aminothiophene-3-carboxamide Chemical compound NC(=O)C=1C=CSC=1N WHZIZZOTISTHCT-UHFFFAOYSA-N 0.000 description 3
- BFYIZQONLCFLEV-DAELLWKTSA-N Aromasine Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CC(=C)C2=C1 BFYIZQONLCFLEV-DAELLWKTSA-N 0.000 description 3
- 229940122815 Aromatase inhibitor Drugs 0.000 description 3
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 description 3
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 3
- PTOAARAWEBMLNO-KVQBGUIXSA-N Cladribine Chemical compound C1=NC=2C(N)=NC(Cl)=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)O1 PTOAARAWEBMLNO-KVQBGUIXSA-N 0.000 description 3
- 108010092160 Dactinomycin Proteins 0.000 description 3
- 108010074604 Epoetin Alfa Proteins 0.000 description 3
- 201000008808 Fibrosarcoma Diseases 0.000 description 3
- 108010029961 Filgrastim Proteins 0.000 description 3
- VWUXBMIQPBEWFH-WCCTWKNTSA-N Fulvestrant Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3[C@H](CCCCCCCCCS(=O)CCCC(F)(F)C(F)(F)F)CC2=C1 VWUXBMIQPBEWFH-WCCTWKNTSA-N 0.000 description 3
- 108010010803 Gelatin Proteins 0.000 description 3
- 206010018338 Glioma Diseases 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 3
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 3
- 108010078049 Interferon alpha-2 Proteins 0.000 description 3
- 102000042838 JAK family Human genes 0.000 description 3
- 108091082332 JAK family Proteins 0.000 description 3
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 3
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 3
- 208000015914 Non-Hodgkin lymphomas Diseases 0.000 description 3
- 208000022873 Ocular disease Diseases 0.000 description 3
- 108010058765 Oncogene Protein pp60(v-src) Proteins 0.000 description 3
- 102000038030 PI3Ks Human genes 0.000 description 3
- 108091007960 PI3Ks Proteins 0.000 description 3
- 229930012538 Paclitaxel Natural products 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 3
- 206010060862 Prostate cancer Diseases 0.000 description 3
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 3
- 201000004681 Psoriasis Diseases 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- BPEGJWRSRHCHSN-UHFFFAOYSA-N Temozolomide Chemical compound O=C1N(C)N=NC2=C(C(N)=O)N=CN21 BPEGJWRSRHCHSN-UHFFFAOYSA-N 0.000 description 3
- 206010043276 Teratoma Diseases 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 3
- 150000001299 aldehydes Chemical class 0.000 description 3
- 108700025316 aldesleukin Proteins 0.000 description 3
- SHGAZHPCJJPHSC-YCNIQYBTSA-N all-trans-retinoic acid Chemical compound OC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 description 3
- 235000019270 ammonium chloride Nutrition 0.000 description 3
- YBBLVLTVTVSKRW-UHFFFAOYSA-N anastrozole Chemical compound N#CC(C)(C)C1=CC(C(C)(C#N)C)=CC(CN2N=CN=C2)=C1 YBBLVLTVTVSKRW-UHFFFAOYSA-N 0.000 description 3
- 230000003078 antioxidant effect Effects 0.000 description 3
- 229940046844 aromatase inhibitors Drugs 0.000 description 3
- GOLCXWYRSKYTSP-UHFFFAOYSA-N arsenic trioxide Inorganic materials O1[As]2O[As]1O2 GOLCXWYRSKYTSP-UHFFFAOYSA-N 0.000 description 3
- 239000012298 atmosphere Substances 0.000 description 3
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 3
- 229960002938 bexarotene Drugs 0.000 description 3
- 229910052796 boron Inorganic materials 0.000 description 3
- 229960005243 carmustine Drugs 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 230000006552 constitutive activation Effects 0.000 description 3
- 239000003246 corticosteroid Substances 0.000 description 3
- 239000013058 crude material Substances 0.000 description 3
- 125000000753 cycloalkyl group Chemical group 0.000 description 3
- 229960004397 cyclophosphamide Drugs 0.000 description 3
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 3
- 229960000605 dexrazoxane Drugs 0.000 description 3
- 239000003085 diluting agent Substances 0.000 description 3
- AAKJLRGGTJKAMG-UHFFFAOYSA-N erlotinib Chemical compound C=12C=C(OCCOC)C(OCCOC)=CC2=NC=NC=1NC1=CC=CC(C#C)=C1 AAKJLRGGTJKAMG-UHFFFAOYSA-N 0.000 description 3
- SKFQJMAGPTUFEW-UHFFFAOYSA-N ethyl 4-bromobenzenecarboximidate Chemical compound CCOC(=N)C1=CC=C(Br)C=C1 SKFQJMAGPTUFEW-UHFFFAOYSA-N 0.000 description 3
- LIQODXNTTZAGID-OCBXBXKTSA-N etoposide phosphate Chemical compound COC1=C(OP(O)(O)=O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 LIQODXNTTZAGID-OCBXBXKTSA-N 0.000 description 3
- 229960000752 etoposide phosphate Drugs 0.000 description 3
- 206010016629 fibroma Diseases 0.000 description 3
- GIUYCYHIANZCFB-FJFJXFQQSA-N fludarabine phosphate Chemical compound C1=NC=2C(N)=NC(F)=NC=2N1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O GIUYCYHIANZCFB-FJFJXFQQSA-N 0.000 description 3
- 229960002949 fluorouracil Drugs 0.000 description 3
- VVIAGPKUTFNRDU-ABLWVSNPSA-N folinic acid Chemical compound C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-ABLWVSNPSA-N 0.000 description 3
- XGALLCVXEZPNRQ-UHFFFAOYSA-N gefitinib Chemical compound C=12C=C(OCCCN3CCOCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 XGALLCVXEZPNRQ-UHFFFAOYSA-N 0.000 description 3
- 239000008273 gelatin Substances 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 235000011852 gelatine desserts Nutrition 0.000 description 3
- 238000001415 gene therapy Methods 0.000 description 3
- 208000005017 glioblastoma Diseases 0.000 description 3
- 239000003102 growth factor Substances 0.000 description 3
- 201000011066 hemangioma Diseases 0.000 description 3
- UUVWYPNAQBNQJQ-UHFFFAOYSA-N hexamethylmelamine Chemical compound CN(C)C1=NC(N(C)C)=NC(N(C)C)=N1 UUVWYPNAQBNQJQ-UHFFFAOYSA-N 0.000 description 3
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 3
- 229940121372 histone deacetylase inhibitor Drugs 0.000 description 3
- 239000004030 hiv protease inhibitor Substances 0.000 description 3
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine monohydrate Substances O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 description 3
- HOMGKSMUEGBAAB-UHFFFAOYSA-N ifosfamide Chemical compound ClCCNP1(=O)OCCCN1CCCl HOMGKSMUEGBAAB-UHFFFAOYSA-N 0.000 description 3
- 239000007943 implant Substances 0.000 description 3
- 125000001041 indolyl group Chemical group 0.000 description 3
- 230000008595 infiltration Effects 0.000 description 3
- 238000001764 infiltration Methods 0.000 description 3
- 150000007529 inorganic bases Chemical class 0.000 description 3
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 description 3
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 3
- 150000002596 lactones Chemical class 0.000 description 3
- HPJKCIUCZWXJDR-UHFFFAOYSA-N letrozole Chemical compound C1=CC(C#N)=CC=C1C(N1N=CN=C1)C1=CC=C(C#N)C=C1 HPJKCIUCZWXJDR-UHFFFAOYSA-N 0.000 description 3
- 229960001691 leucovorin Drugs 0.000 description 3
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 3
- 229940057995 liquid paraffin Drugs 0.000 description 3
- 230000001404 mediated effect Effects 0.000 description 3
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 3
- 230000001394 metastastic effect Effects 0.000 description 3
- SJFNDMHZXCUXSA-UHFFFAOYSA-M methoxymethyl(triphenyl)phosphanium;chloride Chemical compound [Cl-].C=1C=CC=CC=1[P+](C=1C=CC=CC=1)(COC)C1=CC=CC=C1 SJFNDMHZXCUXSA-UHFFFAOYSA-M 0.000 description 3
- 239000004530 micro-emulsion Substances 0.000 description 3
- 231100000782 microtubule inhibitor Toxicity 0.000 description 3
- 150000007522 mineralic acids Chemical class 0.000 description 3
- CFCUWKMKBJTWLW-BKHRDMLASA-N mithramycin Chemical compound O([C@@H]1C[C@@H](O[C@H](C)[C@H]1O)OC=1C=C2C=C3C[C@H]([C@@H](C(=O)C3=C(O)C2=C(O)C=1C)O[C@@H]1O[C@H](C)[C@@H](O)[C@H](O[C@@H]2O[C@H](C)[C@H](O)[C@H](O[C@@H]3O[C@H](C)[C@@H](O)[C@@](C)(O)C3)C2)C1)[C@H](OC)C(=O)[C@@H](O)[C@@H](C)O)[C@H]1C[C@@H](O)[C@H](O)[C@@H](C)O1 CFCUWKMKBJTWLW-BKHRDMLASA-N 0.000 description 3
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- IXOXBSCIXZEQEQ-UHTZMRCNSA-N nelarabine Chemical compound C1=NC=2C(OC)=NC(N)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1O IXOXBSCIXZEQEQ-UHTZMRCNSA-N 0.000 description 3
- 239000004006 olive oil Substances 0.000 description 3
- 235000008390 olive oil Nutrition 0.000 description 3
- 150000007524 organic acids Chemical class 0.000 description 3
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 3
- 230000002611 ovarian Effects 0.000 description 3
- TWSTVYFPWIGASI-UHFFFAOYSA-N oxetan-3-ol Chemical compound OC1COC1.OC1COC1 TWSTVYFPWIGASI-UHFFFAOYSA-N 0.000 description 3
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 229960001592 paclitaxel Drugs 0.000 description 3
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 3
- 229960003171 plicamycin Drugs 0.000 description 3
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 3
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 3
- 239000011591 potassium Substances 0.000 description 3
- 229910052700 potassium Inorganic materials 0.000 description 3
- 235000011056 potassium acetate Nutrition 0.000 description 3
- 229910000027 potassium carbonate Inorganic materials 0.000 description 3
- 229940002612 prodrug Drugs 0.000 description 3
- 239000000651 prodrug Substances 0.000 description 3
- 102000004169 proteins and genes Human genes 0.000 description 3
- 125000002098 pyridazinyl group Chemical group 0.000 description 3
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 3
- 238000010791 quenching Methods 0.000 description 3
- 238000001959 radiotherapy Methods 0.000 description 3
- 229940075993 receptor modulator Drugs 0.000 description 3
- 230000004276 retinal vascularization Effects 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 229960003787 sorafenib Drugs 0.000 description 3
- 241000894007 species Species 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 150000003512 tertiary amines Chemical class 0.000 description 3
- 238000004809 thin layer chromatography Methods 0.000 description 3
- UCFGDBYHRUNTLO-QHCPKHFHSA-N topotecan Chemical compound C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 UCFGDBYHRUNTLO-QHCPKHFHSA-N 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- 229940086542 triethylamine Drugs 0.000 description 3
- ZOCKGBMQLCSHFP-KQRAQHLDSA-N valrubicin Chemical compound O([C@H]1C[C@](CC2=C(O)C=3C(=O)C4=CC=CC(OC)=C4C(=O)C=3C(O)=C21)(O)C(=O)COC(=O)CCCC)[C@H]1C[C@H](NC(=O)C(F)(F)F)[C@H](O)[C@H](C)O1 ZOCKGBMQLCSHFP-KQRAQHLDSA-N 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- NAOVNRUMPBRELH-UHFFFAOYSA-N (2,6-difluoro-4-morpholin-4-ylphenyl)boronic acid Chemical compound C1=C(F)C(B(O)O)=C(F)C=C1N1CCOCC1 NAOVNRUMPBRELH-UHFFFAOYSA-N 0.000 description 2
- ZGGHKIMDNBDHJB-NRFPMOEYSA-M (3R,5S)-fluvastatin sodium Chemical compound [Na+].C12=CC=CC=C2N(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC([O-])=O)=C1C1=CC=C(F)C=C1 ZGGHKIMDNBDHJB-NRFPMOEYSA-M 0.000 description 2
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 2
- FDKXTQMXEQVLRF-ZHACJKMWSA-N (E)-dacarbazine Chemical compound CN(C)\N=N\c1[nH]cnc1C(N)=O FDKXTQMXEQVLRF-ZHACJKMWSA-N 0.000 description 2
- BIOZEBWUZFUNJP-FARCUNLSSA-N (ne)-n-[(5-bromo-2-nitrothiophen-3-yl)methylidene]hydroxylamine Chemical compound O\N=C\C=1C=C(Br)SC=1[N+]([O-])=O BIOZEBWUZFUNJP-FARCUNLSSA-N 0.000 description 2
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 2
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 2
- LBUJPTNKIBCYBY-UHFFFAOYSA-N 1,2,3,4-tetrahydroquinoline Chemical compound C1=CC=C2CCCNC2=C1 LBUJPTNKIBCYBY-UHFFFAOYSA-N 0.000 description 2
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 2
- QFMZQPDHXULLKC-UHFFFAOYSA-N 1,2-bis(diphenylphosphino)ethane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CCP(C=1C=CC=CC=1)C1=CC=CC=C1 QFMZQPDHXULLKC-UHFFFAOYSA-N 0.000 description 2
- SWJPEBQEEAHIGZ-UHFFFAOYSA-N 1,4-dibromobenzene Chemical compound BrC1=CC=C(Br)C=C1 SWJPEBQEEAHIGZ-UHFFFAOYSA-N 0.000 description 2
- HJTAZXHBEBIQQX-UHFFFAOYSA-N 1,5-bis(chloromethyl)naphthalene Chemical compound C1=CC=C2C(CCl)=CC=CC2=C1CCl HJTAZXHBEBIQQX-UHFFFAOYSA-N 0.000 description 2
- NYFIDHXRJSCAOZ-UHFFFAOYSA-N 1-fluoro-4-prop-2-enylbenzene Chemical compound FC1=CC=C(CC=C)C=C1 NYFIDHXRJSCAOZ-UHFFFAOYSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- BUXHYMZMVMNDMG-UHFFFAOYSA-N 2,5-dichlorobenzaldehyde Chemical compound ClC1=CC=C(Cl)C(C=O)=C1 BUXHYMZMVMNDMG-UHFFFAOYSA-N 0.000 description 2
- VQLVKLHHPJTTAW-UHFFFAOYSA-N 2-(2,5-dichlorophenyl)acetaldehyde Chemical compound ClC1=CC=C(Cl)C(CC=O)=C1 VQLVKLHHPJTTAW-UHFFFAOYSA-N 0.000 description 2
- CIIRHUMJFRMXRZ-UHFFFAOYSA-N 2-(4-prop-2-enylphenyl)propan-2-ol Chemical compound CC(C)(O)C1=CC=C(CC=C)C=C1 CIIRHUMJFRMXRZ-UHFFFAOYSA-N 0.000 description 2
- LCFRTRIRICENNR-UHFFFAOYSA-N 2-(5-bromopyridin-2-yl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)C1=CC=C(Br)C=N1 LCFRTRIRICENNR-UHFFFAOYSA-N 0.000 description 2
- OXSDDDKLMCHNHF-UHFFFAOYSA-N 2-(6-bromopyridin-2-yl)propan-2-ol Chemical compound CC(C)(O)C1=CC=CC(Br)=N1 OXSDDDKLMCHNHF-UHFFFAOYSA-N 0.000 description 2
- BPXKZEMBEZGUAH-UHFFFAOYSA-N 2-(chloromethoxy)ethyl-trimethylsilane Chemical compound C[Si](C)(C)CCOCCl BPXKZEMBEZGUAH-UHFFFAOYSA-N 0.000 description 2
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 2
- RTQWWZBSTRGEAV-PKHIMPSTSA-N 2-[[(2s)-2-[bis(carboxymethyl)amino]-3-[4-(methylcarbamoylamino)phenyl]propyl]-[2-[bis(carboxymethyl)amino]propyl]amino]acetic acid Chemical compound CNC(=O)NC1=CC=C(C[C@@H](CN(CC(C)N(CC(O)=O)CC(O)=O)CC(O)=O)N(CC(O)=O)CC(O)=O)C=C1 RTQWWZBSTRGEAV-PKHIMPSTSA-N 0.000 description 2
- DHQKEVGOTKOXSU-UHFFFAOYSA-N 2-amino-5-(4-chlorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(Cl)C=C1 DHQKEVGOTKOXSU-UHFFFAOYSA-N 0.000 description 2
- VBTAQVWRVWJQFL-UHFFFAOYSA-N 2-amino-5-(4-fluorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(F)C=C1 VBTAQVWRVWJQFL-UHFFFAOYSA-N 0.000 description 2
- RJENQXUXRUWMFJ-UHFFFAOYSA-N 2-amino-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C1=CC(C(N)=O)=C(N)S1 RJENQXUXRUWMFJ-UHFFFAOYSA-N 0.000 description 2
- XVGHZFWFGXDIOU-UHFFFAOYSA-N 2-aminothiophene-3-carbonitrile Chemical compound NC=1SC=CC=1C#N XVGHZFWFGXDIOU-UHFFFAOYSA-N 0.000 description 2
- BSKHPKMHTQYZBB-UHFFFAOYSA-N 2-methylpyridine Chemical compound CC1=CC=CC=N1 BSKHPKMHTQYZBB-UHFFFAOYSA-N 0.000 description 2
- JIZRGGUCOQKGQD-UHFFFAOYSA-N 2-nitrothiophene Chemical compound [O-][N+](=O)C1=CC=CS1 JIZRGGUCOQKGQD-UHFFFAOYSA-N 0.000 description 2
- PPYFLTQEWBFPAU-UHFFFAOYSA-N 3-(oxan-4-ylmethyl)pyridine-2-carboxamide Chemical compound NC(=O)C1=NC=CC=C1CC1CCOCC1 PPYFLTQEWBFPAU-UHFFFAOYSA-N 0.000 description 2
- NXJGKRPGEJUREY-UHFFFAOYSA-N 3-(oxolan-2-ylmethyl)pyridine-2-carboxamide Chemical compound NC(=O)C1=NC=CC=C1CC1OCCC1 NXJGKRPGEJUREY-UHFFFAOYSA-N 0.000 description 2
- OVKDLPZRDQTOJW-UHFFFAOYSA-N 3-amino-2-methylbutan-2-ol Chemical compound CC(N)C(C)(C)O OVKDLPZRDQTOJW-UHFFFAOYSA-N 0.000 description 2
- ROADCYAOHVSOLQ-UHFFFAOYSA-N 3-oxetanone Chemical compound O=C1COC1 ROADCYAOHVSOLQ-UHFFFAOYSA-N 0.000 description 2
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 description 2
- RYKZCPFWGNDOQV-UHFFFAOYSA-N 4-(4-bromophenyl)-2h-triazole Chemical compound C1=CC(Br)=CC=C1C1=NNN=C1 RYKZCPFWGNDOQV-UHFFFAOYSA-N 0.000 description 2
- CZGVAISJIQNQEJ-UHFFFAOYSA-N 4-bromo-2,6-difluorobenzaldehyde Chemical compound FC1=CC(Br)=CC(F)=C1C=O CZGVAISJIQNQEJ-UHFFFAOYSA-N 0.000 description 2
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 2
- FZTHBWCYLRWYEY-UHFFFAOYSA-N 5-(2,6-difluoro-4-morpholin-4-ylphenyl)-2-nitrothiophene-3-carboxamide Chemical compound S1C([N+]([O-])=O)=C(C(=O)N)C=C1C1=C(F)C=C(N2CCOCC2)C=C1F FZTHBWCYLRWYEY-UHFFFAOYSA-N 0.000 description 2
- FXLMGCVSJAHDTJ-UHFFFAOYSA-N 5-(4-acetylphenyl)-2-nitrothiophene-3-carboxamide Chemical compound C1=CC(C(=O)C)=CC=C1C1=CC(C(N)=O)=C([N+]([O-])=O)S1 FXLMGCVSJAHDTJ-UHFFFAOYSA-N 0.000 description 2
- XJSPWQRTWATLKK-UHFFFAOYSA-N 5-(4-bromophenyl)-1h-1,2,4-triazole Chemical compound C1=CC(Br)=CC=C1C1=NN=CN1 XJSPWQRTWATLKK-UHFFFAOYSA-N 0.000 description 2
- BNFKQJANQFSCCU-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[[(3-hydroxy-3-methylbutan-2-yl)amino]methyl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=C(C)C(CNC(C)C(C)(C)O)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2F)C(C)(C)O)F)S1 BNFKQJANQFSCCU-UHFFFAOYSA-N 0.000 description 2
- UBCWOSPOCHARSI-AWEZNQCLSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3s)-3-methylmorpholin-4-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C[C@H]1COCCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 UBCWOSPOCHARSI-AWEZNQCLSA-N 0.000 description 2
- SJNLIUTWFZSIAQ-UHFFFAOYSA-N 5-[4-(3-hydroxyoxetan-3-yl)phenyl]-2-nitrothiophene-3-carboxamide Chemical compound S1C([N+]([O-])=O)=C(C(=O)N)C=C1C1=CC=C(C2(O)COC2)C=C1 SJNLIUTWFZSIAQ-UHFFFAOYSA-N 0.000 description 2
- IDPUKCWIGUEADI-UHFFFAOYSA-N 5-[bis(2-chloroethyl)amino]uracil Chemical compound ClCCN(CCCl)C1=CNC(=O)NC1=O IDPUKCWIGUEADI-UHFFFAOYSA-N 0.000 description 2
- NMUSYJAQQFHJEW-KVTDHHQDSA-N 5-azacytidine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-KVTDHHQDSA-N 0.000 description 2
- XIKMORWWTRWLAS-UHFFFAOYSA-N 5-bromo-1,3-difluoro-2-(2-methoxyethenyl)benzene Chemical compound COC=CC1=C(F)C=C(Br)C=C1F XIKMORWWTRWLAS-UHFFFAOYSA-N 0.000 description 2
- FSWWDXRRJRWFTC-UHFFFAOYSA-N 5-bromo-2-nitrothiophene-3-carbaldehyde Chemical compound [O-][N+](=O)C=1SC(Br)=CC=1C=O FSWWDXRRJRWFTC-UHFFFAOYSA-N 0.000 description 2
- QLOXTYRUKBYUTJ-UHFFFAOYSA-N 5-bromo-3-(dichloromethyl)-2-nitrothiophene Chemical compound [O-][N+](=O)C=1SC(Br)=CC=1C(Cl)Cl QLOXTYRUKBYUTJ-UHFFFAOYSA-N 0.000 description 2
- KOSBXZHFWKTMLO-UHFFFAOYSA-N 5-nitrothiophene-2-carboxamide Chemical compound NC(=O)C1=CC=C([N+]([O-])=O)S1 KOSBXZHFWKTMLO-UHFFFAOYSA-N 0.000 description 2
- QWFHFNGMCPMOCD-UHFFFAOYSA-N 6-bromopyridine-2-carbaldehyde Chemical compound BrC1=CC=CC(C=O)=N1 QWFHFNGMCPMOCD-UHFFFAOYSA-N 0.000 description 2
- VVIAGPKUTFNRDU-UHFFFAOYSA-N 6S-folinic acid Natural products C1NC=2NC(N)=NC(=O)C=2N(C=O)C1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 VVIAGPKUTFNRDU-UHFFFAOYSA-N 0.000 description 2
- SHGAZHPCJJPHSC-ZVCIMWCZSA-N 9-cis-retinoic acid Chemical compound OC(=O)/C=C(\C)/C=C/C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-ZVCIMWCZSA-N 0.000 description 2
- 102100033350 ATP-dependent translocase ABCB1 Human genes 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- 201000003076 Angiosarcoma Diseases 0.000 description 2
- 235000003911 Arachis Nutrition 0.000 description 2
- 244000105624 Arachis hypogaea Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- 108010024976 Asparaginase Proteins 0.000 description 2
- 102000015790 Asparaginase Human genes 0.000 description 2
- 201000001320 Atherosclerosis Diseases 0.000 description 2
- 208000023275 Autoimmune disease Diseases 0.000 description 2
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 description 2
- 108010006654 Bleomycin Proteins 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- 101150006084 CHKB gene Proteins 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- GAGWJHPBXLXJQN-UHFFFAOYSA-N Capecitabine Natural products C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1C1C(O)C(O)C(C)O1 GAGWJHPBXLXJQN-UHFFFAOYSA-N 0.000 description 2
- 102000003847 Carboxypeptidase B2 Human genes 0.000 description 2
- 108090000201 Carboxypeptidase B2 Proteins 0.000 description 2
- JWBOIMRXGHLCPP-UHFFFAOYSA-N Chloditan Chemical compound C=1C=CC=C(Cl)C=1C(C(Cl)Cl)C1=CC=C(Cl)C=C1 JWBOIMRXGHLCPP-UHFFFAOYSA-N 0.000 description 2
- 229920000742 Cotton Polymers 0.000 description 2
- 108010019673 Darbepoetin alfa Proteins 0.000 description 2
- WEAHRLBPCANXCN-UHFFFAOYSA-N Daunomycin Natural products CCC1(O)CC(OC2CC(N)C(O)C(C)O2)c3cc4C(=O)c5c(OC)cccc5C(=O)c4c(O)c3C1 WEAHRLBPCANXCN-UHFFFAOYSA-N 0.000 description 2
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 2
- MWWSFMDVAYGXBV-RUELKSSGSA-N Doxorubicin hydrochloride Chemical compound Cl.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 MWWSFMDVAYGXBV-RUELKSSGSA-N 0.000 description 2
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 2
- 208000032612 Glial tumor Diseases 0.000 description 2
- BLCLNMBMMGCOAS-URPVMXJPSA-N Goserelin Chemical compound C([C@@H](C(=O)N[C@H](COC(C)(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NNC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 BLCLNMBMMGCOAS-URPVMXJPSA-N 0.000 description 2
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- 229940122440 HIV protease inhibitor Drugs 0.000 description 2
- 208000002927 Hamartoma Diseases 0.000 description 2
- 208000001258 Hemangiosarcoma Diseases 0.000 description 2
- 102000004286 Hydroxymethylglutaryl CoA Reductases Human genes 0.000 description 2
- 108090000895 Hydroxymethylglutaryl CoA Reductases Proteins 0.000 description 2
- VSNHCAURESNICA-UHFFFAOYSA-N Hydroxyurea Chemical compound NC(=O)NO VSNHCAURESNICA-UHFFFAOYSA-N 0.000 description 2
- 206010021143 Hypoxia Diseases 0.000 description 2
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 2
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 2
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 2
- 108010047761 Interferon-alpha Proteins 0.000 description 2
- 102000006992 Interferon-alpha Human genes 0.000 description 2
- 108010074328 Interferon-gamma Proteins 0.000 description 2
- 108090000174 Interleukin-10 Proteins 0.000 description 2
- 108010002350 Interleukin-2 Proteins 0.000 description 2
- 102000000588 Interleukin-2 Human genes 0.000 description 2
- 108090000978 Interleukin-4 Proteins 0.000 description 2
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 2
- 230000004163 JAK-STAT signaling pathway Effects 0.000 description 2
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 2
- 239000005551 L01XE03 - Erlotinib Substances 0.000 description 2
- 239000005511 L01XE05 - Sorafenib Substances 0.000 description 2
- 208000018142 Leiomyosarcoma Diseases 0.000 description 2
- GQYIWUVLTXOXAJ-UHFFFAOYSA-N Lomustine Chemical compound ClCCN(N=O)C(=O)NC1CCCCC1 GQYIWUVLTXOXAJ-UHFFFAOYSA-N 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- QXKHYNVANLEOEG-UHFFFAOYSA-N Methoxsalen Chemical compound C1=CC(=O)OC2=C1C=C1C=COC1=C2OC QXKHYNVANLEOEG-UHFFFAOYSA-N 0.000 description 2
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 2
- 206010028561 Myeloid metaplasia Diseases 0.000 description 2
- HSHXDCVZWHOWCS-UHFFFAOYSA-N N'-hexadecylthiophene-2-carbohydrazide Chemical compound CCCCCCCCCCCCCCCCNNC(=O)c1cccs1 HSHXDCVZWHOWCS-UHFFFAOYSA-N 0.000 description 2
- 150000001204 N-oxides Chemical class 0.000 description 2
- 238000005481 NMR spectroscopy Methods 0.000 description 2
- 229940123821 Neurokinin 1 receptor antagonist Drugs 0.000 description 2
- 206010033128 Ovarian cancer Diseases 0.000 description 2
- SHGAZHPCJJPHSC-UHFFFAOYSA-N Panrexin Chemical compound OC(=O)C=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-UHFFFAOYSA-N 0.000 description 2
- 229910002666 PdCl2 Inorganic materials 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 229920000954 Polyglycolide Polymers 0.000 description 2
- IIDJRNMFWXDHID-UHFFFAOYSA-N Risedronic acid Chemical compound OP(=O)(O)C(P(O)(O)=O)(O)CC1=CC=CN=C1 IIDJRNMFWXDHID-UHFFFAOYSA-N 0.000 description 2
- YASAKCUCGLMORW-UHFFFAOYSA-N Rosiglitazone Chemical compound C=1C=CC=NC=1N(C)CCOC(C=C1)=CC=C1CC1SC(=O)NC1=O YASAKCUCGLMORW-UHFFFAOYSA-N 0.000 description 2
- YJDYDFNKCBANTM-QCWCSKBGSA-N SDZ PSC 833 Chemical compound C\C=C\C[C@@H](C)C(=O)[C@@H]1N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C(=O)[C@H](C(C)C)NC1=O YJDYDFNKCBANTM-QCWCSKBGSA-N 0.000 description 2
- 108060006706 SRC Proteins 0.000 description 2
- 102000001332 SRC Human genes 0.000 description 2
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- UIRKNQLZZXALBI-MSVGPLKSSA-N Squalamine Chemical compound C([C@@H]1C[C@H]2O)[C@@H](NCCCNCCCCN)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@H](C)CC[C@H](C(C)C)OS(O)(=O)=O)[C@@]2(C)CC1 UIRKNQLZZXALBI-MSVGPLKSSA-N 0.000 description 2
- UIRKNQLZZXALBI-UHFFFAOYSA-N Squalamine Natural products OC1CC2CC(NCCCNCCCCN)CCC2(C)C2C1C1CCC(C(C)CCC(C(C)C)OS(O)(=O)=O)C1(C)CC2 UIRKNQLZZXALBI-UHFFFAOYSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- CBPNZQVSJQDFBE-FUXHJELOSA-N Temsirolimus Chemical compound C1C[C@@H](OC(=O)C(C)(CO)CO)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 CBPNZQVSJQDFBE-FUXHJELOSA-N 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- FOCVUCIESVLUNU-UHFFFAOYSA-N Thiotepa Chemical compound C1CN1P(N1CC1)(=S)N1CC1 FOCVUCIESVLUNU-UHFFFAOYSA-N 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 102000005789 Vascular Endothelial Growth Factors Human genes 0.000 description 2
- 108010019530 Vascular Endothelial Growth Factors Proteins 0.000 description 2
- 102100033177 Vascular endothelial growth factor receptor 2 Human genes 0.000 description 2
- 208000008383 Wilms tumor Diseases 0.000 description 2
- AIWRTTMUVOZGPW-HSPKUQOVSA-N abarelix Chemical compound C([C@@H](C(=O)N[C@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)N(C)C(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 AIWRTTMUVOZGPW-HSPKUQOVSA-N 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 230000001154 acute effect Effects 0.000 description 2
- 229960005310 aldesleukin Drugs 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 229960001445 alitretinoin Drugs 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- OFCNXPDARWKPPY-UHFFFAOYSA-N allopurinol Chemical compound OC1=NC=NC2=C1C=NN2 OFCNXPDARWKPPY-UHFFFAOYSA-N 0.000 description 2
- 229960000473 altretamine Drugs 0.000 description 2
- JKOQGQFVAUAYPM-UHFFFAOYSA-N amifostine Chemical compound NCCCNCCSP(O)(O)=O JKOQGQFVAUAYPM-UHFFFAOYSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 229960002932 anastrozole Drugs 0.000 description 2
- 230000000340 anti-metabolite Effects 0.000 description 2
- 239000000427 antigen Substances 0.000 description 2
- 108091007433 antigens Proteins 0.000 description 2
- 102000036639 antigens Human genes 0.000 description 2
- 229940100197 antimetabolite Drugs 0.000 description 2
- 239000002256 antimetabolite Substances 0.000 description 2
- 206010003246 arthritis Diseases 0.000 description 2
- 238000003556 assay Methods 0.000 description 2
- 208000006673 asthma Diseases 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 239000003719 aurora kinase inhibitor Substances 0.000 description 2
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 2
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 2
- HSDAJNMJOMSNEV-UHFFFAOYSA-N benzyl chloroformate Chemical compound ClC(=O)OCC1=CC=CC=C1 HSDAJNMJOMSNEV-UHFFFAOYSA-N 0.000 description 2
- NMSSIVVNMBREJK-UHFFFAOYSA-N benzyl n-(3-carbamoyl-5-iodothiophen-2-yl)carbamate Chemical compound C1=C(I)SC(NC(=O)OCC=2C=CC=CC=2)=C1C(=O)N NMSSIVVNMBREJK-UHFFFAOYSA-N 0.000 description 2
- 230000033228 biological regulation Effects 0.000 description 2
- 239000004305 biphenyl Substances 0.000 description 2
- IPWKHHSGDUIRAH-UHFFFAOYSA-N bis(pinacolato)diboron Chemical compound O1C(C)(C)C(C)(C)OB1B1OC(C)(C)C(C)(C)O1 IPWKHHSGDUIRAH-UHFFFAOYSA-N 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000000988 bone and bone Anatomy 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 229960002092 busulfan Drugs 0.000 description 2
- RYYVLZVUVIJVGH-UHFFFAOYSA-N caffeine Chemical compound CN1C(=O)N(C)C(=O)C2=C1N=CN2C RYYVLZVUVIJVGH-UHFFFAOYSA-N 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- 235000010216 calcium carbonate Nutrition 0.000 description 2
- 239000001506 calcium phosphate Substances 0.000 description 2
- 229910000389 calcium phosphate Inorganic materials 0.000 description 2
- 235000011010 calcium phosphates Nutrition 0.000 description 2
- IVFYLRMMHVYGJH-PVPPCFLZSA-N calusterone Chemical compound C1C[C@]2(C)[C@](O)(C)CC[C@H]2[C@@H]2[C@@H](C)CC3=CC(=O)CC[C@]3(C)[C@H]21 IVFYLRMMHVYGJH-PVPPCFLZSA-N 0.000 description 2
- 229950009823 calusterone Drugs 0.000 description 2
- 229960004117 capecitabine Drugs 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 2
- 229960004562 carboplatin Drugs 0.000 description 2
- 208000002458 carcinoid tumor Diseases 0.000 description 2
- 229940047495 celebrex Drugs 0.000 description 2
- 229960000590 celecoxib Drugs 0.000 description 2
- 229960005110 cerivastatin Drugs 0.000 description 2
- SEERZIQQUAZTOL-ANMDKAQQSA-N cerivastatin Chemical compound COCC1=C(C(C)C)N=C(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)=C1C1=CC=C(F)C=C1 SEERZIQQUAZTOL-ANMDKAQQSA-N 0.000 description 2
- 230000000973 chemotherapeutic effect Effects 0.000 description 2
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 2
- 235000012000 cholesterol Nutrition 0.000 description 2
- 125000000259 cinnolinyl group Chemical group N1=NC(=CC2=CC=CC=C12)* 0.000 description 2
- 229960004316 cisplatin Drugs 0.000 description 2
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 2
- 229960002436 cladribine Drugs 0.000 description 2
- WDDPHFBMKLOVOX-AYQXTPAHSA-N clofarabine Chemical compound C1=NC=2C(N)=NC(Cl)=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@@H]1F WDDPHFBMKLOVOX-AYQXTPAHSA-N 0.000 description 2
- 238000005345 coagulation Methods 0.000 description 2
- 230000015271 coagulation Effects 0.000 description 2
- 229940110456 cocoa butter Drugs 0.000 description 2
- 235000019868 cocoa butter Nutrition 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 2
- 239000003255 cyclooxygenase 2 inhibitor Substances 0.000 description 2
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 2
- 229960000684 cytarabine Drugs 0.000 description 2
- 229940127089 cytotoxic agent Drugs 0.000 description 2
- 231100000599 cytotoxic agent Toxicity 0.000 description 2
- 229960000640 dactinomycin Drugs 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- 230000007812 deficiency Effects 0.000 description 2
- 108010017271 denileukin diftitox Proteins 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 230000002074 deregulated effect Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 229960003957 dexamethasone Drugs 0.000 description 2
- CSJLBAMHHLJAAS-UHFFFAOYSA-N diethylaminosulfur trifluoride Chemical compound CCN(CC)S(F)(F)F CSJLBAMHHLJAAS-UHFFFAOYSA-N 0.000 description 2
- 229940043279 diisopropylamine Drugs 0.000 description 2
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 2
- 208000035475 disorder Diseases 0.000 description 2
- 239000003534 dna topoisomerase inhibitor Substances 0.000 description 2
- 229960003668 docetaxel Drugs 0.000 description 2
- 229950004683 drostanolone propionate Drugs 0.000 description 2
- 230000007613 environmental effect Effects 0.000 description 2
- 229960003388 epoetin alfa Drugs 0.000 description 2
- 229930013356 epothilone Natural products 0.000 description 2
- HESCAJZNRMSMJG-KKQRBIROSA-N epothilone A Chemical class C/C([C@@H]1C[C@@H]2O[C@@H]2CCC[C@@H]([C@@H]([C@@H](C)C(=O)C(C)(C)[C@@H](O)CC(=O)O1)O)C)=C\C1=CSC(C)=N1 HESCAJZNRMSMJG-KKQRBIROSA-N 0.000 description 2
- 229960001842 estramustine Drugs 0.000 description 2
- FRPJXPJMRWBBIH-RBRWEJTLSA-N estramustine Chemical compound ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 FRPJXPJMRWBBIH-RBRWEJTLSA-N 0.000 description 2
- NHOGGUYTANYCGQ-UHFFFAOYSA-N ethenoxybenzene Chemical class C=COC1=CC=CC=C1 NHOGGUYTANYCGQ-UHFFFAOYSA-N 0.000 description 2
- SWJFEVFZCZJOOE-UHFFFAOYSA-N ethyl 4-bromobenzenecarboximidate;hydrochloride Chemical compound Cl.CCOC(=N)C1=CC=C(Br)C=C1 SWJFEVFZCZJOOE-UHFFFAOYSA-N 0.000 description 2
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 2
- 229960005420 etoposide Drugs 0.000 description 2
- 229960000255 exemestane Drugs 0.000 description 2
- 230000020764 fibrinolysis Effects 0.000 description 2
- 210000002950 fibroblast Anatomy 0.000 description 2
- 229960004177 filgrastim Drugs 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 239000012467 final product Substances 0.000 description 2
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 2
- 229960000390 fludarabine Drugs 0.000 description 2
- 235000008191 folinic acid Nutrition 0.000 description 2
- 239000011672 folinic acid Substances 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 229960002258 fulvestrant Drugs 0.000 description 2
- 125000002541 furyl group Chemical group 0.000 description 2
- 230000002496 gastric effect Effects 0.000 description 2
- 239000007903 gelatin capsule Substances 0.000 description 2
- 229960003297 gemtuzumab ozogamicin Drugs 0.000 description 2
- KWIUHFFTVRNATP-UHFFFAOYSA-N glycine betaine Chemical compound C[N+](C)(C)CC([O-])=O KWIUHFFTVRNATP-UHFFFAOYSA-N 0.000 description 2
- 229960003690 goserelin acetate Drugs 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 229910052736 halogen Inorganic materials 0.000 description 2
- 150000002367 halogens Chemical class 0.000 description 2
- 210000003128 head Anatomy 0.000 description 2
- 210000002443 helper t lymphocyte Anatomy 0.000 description 2
- 206010073071 hepatocellular carcinoma Diseases 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- HHXHVIJIIXKSOE-QILQGKCVSA-N histrelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC(N=C1)=CN1CC1=CC=CC=C1 HHXHVIJIIXKSOE-QILQGKCVSA-N 0.000 description 2
- 239000008172 hydrogenated vegetable oil Substances 0.000 description 2
- 230000007062 hydrolysis Effects 0.000 description 2
- 238000006460 hydrolysis reaction Methods 0.000 description 2
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 2
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 2
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 2
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 2
- 230000007954 hypoxia Effects 0.000 description 2
- 229960001001 ibritumomab tiuxetan Drugs 0.000 description 2
- 229960000908 idarubicin Drugs 0.000 description 2
- 229960001101 ifosfamide Drugs 0.000 description 2
- 229960002411 imatinib Drugs 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- LWRDQHOZTAOILO-UHFFFAOYSA-N incadronic acid Chemical compound OP(O)(=O)C(P(O)(O)=O)NC1CCCCCC1 LWRDQHOZTAOILO-UHFFFAOYSA-N 0.000 description 2
- 229950006971 incadronic acid Drugs 0.000 description 2
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 239000002198 insoluble material Substances 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 229960004768 irinotecan Drugs 0.000 description 2
- 125000000842 isoxazolyl group Chemical group 0.000 description 2
- 210000003734 kidney Anatomy 0.000 description 2
- 229940043355 kinase inhibitor Drugs 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 201000010260 leiomyoma Diseases 0.000 description 2
- GOTYRUGSSMKFNF-UHFFFAOYSA-N lenalidomide Chemical compound C1C=2C(N)=CC=CC=2C(=O)N1C1CCC(=O)NC1=O GOTYRUGSSMKFNF-UHFFFAOYSA-N 0.000 description 2
- 229960003881 letrozole Drugs 0.000 description 2
- GFIJNRVAKGFPGQ-LIJARHBVSA-N leuprolide Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 GFIJNRVAKGFPGQ-LIJARHBVSA-N 0.000 description 2
- 229960004338 leuprorelin Drugs 0.000 description 2
- 229960001614 levamisole Drugs 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- UBJFKNSINUCEAL-UHFFFAOYSA-N lithium;2-methylpropane Chemical compound [Li+].C[C-](C)C UBJFKNSINUCEAL-UHFFFAOYSA-N 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 230000004777 loss-of-function mutation Effects 0.000 description 2
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- NXPHGHWWQRMDIA-UHFFFAOYSA-M magnesium;carbanide;bromide Chemical compound [CH3-].[Mg+2].[Br-] NXPHGHWWQRMDIA-UHFFFAOYSA-M 0.000 description 2
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical class ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 description 2
- 239000002609 medium Substances 0.000 description 2
- RQZAXGRLVPAYTJ-GQFGMJRRSA-N megestrol acetate Chemical compound C1=C(C)C2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@@](C(C)=O)(OC(=O)C)[C@@]1(C)CC2 RQZAXGRLVPAYTJ-GQFGMJRRSA-N 0.000 description 2
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 2
- 206010027191 meningioma Diseases 0.000 description 2
- 229960000901 mepacrine Drugs 0.000 description 2
- 229960004635 mesna Drugs 0.000 description 2
- 229940101533 mesnex Drugs 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 229960000485 methotrexate Drugs 0.000 description 2
- XLZPPHWBAKACEX-UHFFFAOYSA-N methyl 4-prop-2-enylbenzoate Chemical compound COC(=O)C1=CC=C(CC=C)C=C1 XLZPPHWBAKACEX-UHFFFAOYSA-N 0.000 description 2
- 230000003228 microsomal effect Effects 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- 229960004857 mitomycin Drugs 0.000 description 2
- 230000008600 mitotic progression Effects 0.000 description 2
- 229960001156 mitoxantrone Drugs 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 206010028537 myelofibrosis Diseases 0.000 description 2
- PFPSZGPAQFBVHZ-UHFFFAOYSA-N n-(3-chlorophenyl)-2-[(4-phenyl-5-pyridin-4-yl-1,2,4-triazol-3-yl)sulfanyl]acetamide Chemical compound ClC1=CC=CC(NC(=O)CSC=2N(C(C=3C=CN=CC=3)=NN=2)C=2C=CC=CC=2)=C1 PFPSZGPAQFBVHZ-UHFFFAOYSA-N 0.000 description 2
- BLCLNMBMMGCOAS-UHFFFAOYSA-N n-[1-[[1-[[1-[[1-[[1-[[1-[[1-[2-[(carbamoylamino)carbamoyl]pyrrolidin-1-yl]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-[(2-methylpropan-2-yl)oxy]-1-oxopropan-2-yl]amino]-3-(4-hydroxyphenyl)-1-oxopropan-2-yl]amin Chemical compound C1CCC(C(=O)NNC(N)=O)N1C(=O)C(CCCN=C(N)N)NC(=O)C(CC(C)C)NC(=O)C(COC(C)(C)C)NC(=O)C(NC(=O)C(CO)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C(CC=1NC=NC=1)NC(=O)C1NC(=O)CC1)CC1=CC=C(O)C=C1 BLCLNMBMMGCOAS-UHFFFAOYSA-N 0.000 description 2
- UBWXUGDQUBIEIZ-QNTYDACNSA-N nandrolone phenpropionate Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@H]4CCC(=O)C=C4CC3)CC[C@@]21C)C(=O)CCC1=CC=CC=C1 UBWXUGDQUBIEIZ-QNTYDACNSA-N 0.000 description 2
- 229960000801 nelarabine Drugs 0.000 description 2
- 102000037979 non-receptor tyrosine kinases Human genes 0.000 description 2
- 108091008046 non-receptor tyrosine kinases Proteins 0.000 description 2
- 239000000346 nonvolatile oil Substances 0.000 description 2
- 108010046821 oprelvekin Proteins 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 239000012285 osmium tetroxide Substances 0.000 description 2
- 229910000489 osmium tetroxide Inorganic materials 0.000 description 2
- 201000008968 osteosarcoma Diseases 0.000 description 2
- 229960001756 oxaliplatin Drugs 0.000 description 2
- DWAFYCQODLXJNR-BNTLRKBRSA-L oxaliplatin Chemical compound O1C(=O)C(=O)O[Pt]11N[C@@H]2CCCC[C@H]2N1 DWAFYCQODLXJNR-BNTLRKBRSA-L 0.000 description 2
- 125000002971 oxazolyl group Chemical group 0.000 description 2
- 150000002923 oximes Chemical class 0.000 description 2
- ZRSNZINYAWTAHE-UHFFFAOYSA-N p-methoxybenzaldehyde Chemical compound COC1=CC=C(C=O)C=C1 ZRSNZINYAWTAHE-UHFFFAOYSA-N 0.000 description 2
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 2
- 229940046231 pamidronate Drugs 0.000 description 2
- 108010027841 pegademase bovine Proteins 0.000 description 2
- 108010001564 pegaspargase Proteins 0.000 description 2
- 108010044644 pegfilgrastim Proteins 0.000 description 2
- WBXPDJSOTKVWSJ-ZDUSSCGKSA-L pemetrexed(2-) Chemical compound C=1NC=2NC(N)=NC(=O)C=2C=1CCC1=CC=C(C(=O)N[C@@H](CCC([O-])=O)C([O-])=O)C=C1 WBXPDJSOTKVWSJ-ZDUSSCGKSA-L 0.000 description 2
- 229960002340 pentostatin Drugs 0.000 description 2
- 239000003208 petroleum Substances 0.000 description 2
- DTUQWGWMVIHBKE-UHFFFAOYSA-N phenylacetaldehyde Chemical class O=CCC1=CC=CC=C1 DTUQWGWMVIHBKE-UHFFFAOYSA-N 0.000 description 2
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 2
- HYAFETHFCAUJAY-UHFFFAOYSA-N pioglitazone Chemical compound N1=CC(CC)=CC=C1CCOC(C=C1)=CC=C1CC1C(=O)NC(=O)S1 HYAFETHFCAUJAY-UHFFFAOYSA-N 0.000 description 2
- NJBFOOCLYDNZJN-UHFFFAOYSA-N pipobroman Chemical compound BrCCC(=O)N1CCN(C(=O)CCBr)CC1 NJBFOOCLYDNZJN-UHFFFAOYSA-N 0.000 description 2
- 229960000952 pipobroman Drugs 0.000 description 2
- 239000004633 polyglycolic acid Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- CPTBDICYNRMXFX-UHFFFAOYSA-N procarbazine Chemical compound CNNCC1=CC=C(C(=O)NC(C)C)C=C1 CPTBDICYNRMXFX-UHFFFAOYSA-N 0.000 description 2
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 2
- 229960004063 propylene glycol Drugs 0.000 description 2
- 235000013772 propylene glycol Nutrition 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- 125000003373 pyrazinyl group Chemical group 0.000 description 2
- 125000004076 pyridyl group Chemical group 0.000 description 2
- 125000000714 pyrimidinyl group Chemical group 0.000 description 2
- 125000000168 pyrrolyl group Chemical group 0.000 description 2
- GPKJTRJOBQGKQK-UHFFFAOYSA-N quinacrine Chemical compound C1=C(OC)C=C2C(NC(C)CCCN(CC)CC)=C(C=CC(Cl)=C3)C3=NC2=C1 GPKJTRJOBQGKQK-UHFFFAOYSA-N 0.000 description 2
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 2
- 102000009929 raf Kinases Human genes 0.000 description 2
- 108010077182 raf Kinases Proteins 0.000 description 2
- 108010084837 rasburicase Proteins 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 208000037803 restenosis Diseases 0.000 description 2
- 229960004641 rituximab Drugs 0.000 description 2
- RZJQGNCSTQAWON-UHFFFAOYSA-N rofecoxib Chemical compound C1=CC(S(=O)(=O)C)=CC=C1C1=C(C=2C=CC=CC=2)C(=O)OC1 RZJQGNCSTQAWON-UHFFFAOYSA-N 0.000 description 2
- 229960002530 sargramostim Drugs 0.000 description 2
- WUWDLXZGHZSWQZ-WQLSENKSSA-N semaxanib Chemical compound N1C(C)=CC(C)=C1\C=C/1C2=CC=CC=C2NC\1=O WUWDLXZGHZSWQZ-WQLSENKSSA-N 0.000 description 2
- 229910052710 silicon Inorganic materials 0.000 description 2
- 239000010703 silicon Substances 0.000 description 2
- RYMZZMVNJRMUDD-HGQWONQESA-N simvastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)C(C)(C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 RYMZZMVNJRMUDD-HGQWONQESA-N 0.000 description 2
- 210000003491 skin Anatomy 0.000 description 2
- 229910052708 sodium Inorganic materials 0.000 description 2
- 235000015424 sodium Nutrition 0.000 description 2
- 239000012279 sodium borohydride Substances 0.000 description 2
- 229910000033 sodium borohydride Inorganic materials 0.000 description 2
- 235000011121 sodium hydroxide Nutrition 0.000 description 2
- JQWHASGSAFIOCM-UHFFFAOYSA-M sodium periodate Chemical compound [Na+].[O-]I(=O)(=O)=O JQWHASGSAFIOCM-UHFFFAOYSA-M 0.000 description 2
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 2
- 235000019345 sodium thiosulphate Nutrition 0.000 description 2
- NSFFYSQTVOCNLX-JKIHJDPOSA-M sodium;[(2r,3s,4s,5r)-5-(4-amino-2-oxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl octadecyl phosphate;hydrate Chemical compound O.[Na+].O[C@H]1[C@H](O)[C@@H](COP([O-])(=O)OCCCCCCCCCCCCCCCCCC)O[C@H]1N1C(=O)N=C(N)C=C1 NSFFYSQTVOCNLX-JKIHJDPOSA-M 0.000 description 2
- 235000011069 sorbitan monooleate Nutrition 0.000 description 2
- 239000001593 sorbitan monooleate Substances 0.000 description 2
- 229940035049 sorbitan monooleate Drugs 0.000 description 2
- 229950001248 squalamine Drugs 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000000454 talc Substances 0.000 description 2
- 235000012222 talc Nutrition 0.000 description 2
- 229910052623 talc Inorganic materials 0.000 description 2
- 229960001603 tamoxifen Drugs 0.000 description 2
- 229940099419 targretin Drugs 0.000 description 2
- 229960004964 temozolomide Drugs 0.000 description 2
- 229960000235 temsirolimus Drugs 0.000 description 2
- 229960001278 teniposide Drugs 0.000 description 2
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- BPEWUONYVDABNZ-DZBHQSCQSA-N testolactone Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)(OC(=O)CC4)[C@@H]4[C@@H]3CCC2=C1 BPEWUONYVDABNZ-DZBHQSCQSA-N 0.000 description 2
- 229960005353 testolactone Drugs 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- YAPQBXQYLJRXSA-UHFFFAOYSA-N theobromine Chemical compound CN1C(=O)NC(=O)C2=C1N=CN2C YAPQBXQYLJRXSA-UHFFFAOYSA-N 0.000 description 2
- 229960004559 theobromine Drugs 0.000 description 2
- 125000001544 thienyl group Chemical group 0.000 description 2
- 229960003087 tioguanine Drugs 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 229940044693 topoisomerase inhibitor Drugs 0.000 description 2
- 229960000303 topotecan Drugs 0.000 description 2
- XFCLJVABOIYOMF-QPLCGJKRSA-N toremifene Chemical compound C1=CC(OCCN(C)C)=CC=C1C(\C=1C=CC=CC=1)=C(\CCCl)C1=CC=CC=C1 XFCLJVABOIYOMF-QPLCGJKRSA-N 0.000 description 2
- 229960005026 toremifene Drugs 0.000 description 2
- 229960000575 trastuzumab Drugs 0.000 description 2
- 229960001727 tretinoin Drugs 0.000 description 2
- YNDXUCZADRHECN-JNQJZLCISA-N triamcinolone acetonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O YNDXUCZADRHECN-JNQJZLCISA-N 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- 230000001960 triggered effect Effects 0.000 description 2
- GXPHKUHSUJUWKP-UHFFFAOYSA-N troglitazone Chemical compound C1CC=2C(C)=C(O)C(C)=C(C)C=2OC1(C)COC(C=C1)=CC=C1CC1SC(=O)NC1=O GXPHKUHSUJUWKP-UHFFFAOYSA-N 0.000 description 2
- 229960001641 troglitazone Drugs 0.000 description 2
- GXPHKUHSUJUWKP-NTKDMRAZSA-N troglitazone Natural products C([C@@]1(OC=2C(C)=C(C(=C(C)C=2CC1)O)C)C)OC(C=C1)=CC=C1C[C@H]1SC(=O)NC1=O GXPHKUHSUJUWKP-NTKDMRAZSA-N 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 description 2
- 239000005483 tyrosine kinase inhibitor Substances 0.000 description 2
- 239000002691 unilamellar liposome Substances 0.000 description 2
- 229960001055 uracil mustard Drugs 0.000 description 2
- 210000003932 urinary bladder Anatomy 0.000 description 2
- 102000009816 urokinase plasminogen activator receptor activity proteins Human genes 0.000 description 2
- 108040001269 urokinase plasminogen activator receptor activity proteins Proteins 0.000 description 2
- 229960000653 valrubicin Drugs 0.000 description 2
- 229950010938 valspodar Drugs 0.000 description 2
- 108010082372 valspodar Proteins 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- 235000015112 vegetable and seed oil Nutrition 0.000 description 2
- 239000008158 vegetable oil Substances 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- 229940099039 velcade Drugs 0.000 description 2
- JXLYSJRDGCGARV-CFWMRBGOSA-N vinblastine Chemical compound C([C@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-CFWMRBGOSA-N 0.000 description 2
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 2
- 229960004528 vincristine Drugs 0.000 description 2
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 2
- JIAARYAFYJHUJI-UHFFFAOYSA-L zinc dichloride Chemical compound [Cl-].[Cl-].[Zn+2] JIAARYAFYJHUJI-UHFFFAOYSA-L 0.000 description 2
- 229940033942 zoladex Drugs 0.000 description 2
- 229960004276 zoledronic acid Drugs 0.000 description 2
- 229940002005 zometa Drugs 0.000 description 2
- JKFZMIQMKFWJAY-RQJQXFIZSA-N (1r,3s,5z)-5-[(2e)-2-[(3as,7as)-1-[(2r)-6-hydroxy-6-methylhept-4-yn-2-yl]-7a-methyl-3a,5,6,7-tetrahydro-3h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C1(/[C@@H]2CC=C([C@]2(CCC1)C)[C@@H](CC#CC(C)(C)O)C)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C JKFZMIQMKFWJAY-RQJQXFIZSA-N 0.000 description 1
- QQLRSCZSKQTFGY-UHFFFAOYSA-N (2,4-difluorophenyl)boronic acid Chemical compound OB(O)C1=CC=C(F)C=C1F QQLRSCZSKQTFGY-UHFFFAOYSA-N 0.000 description 1
- USYHIIHUJPBCQG-UHFFFAOYSA-N (2-chloroacetyl)-[5-methoxy-4-[2-methyl-3-(3-methylbut-2-enyl)oxiran-2-yl]-1-oxaspiro[2.5]octan-6-yl]carbamic acid Chemical compound O1C(CC=C(C)C)C1(C)C1C(OC)C(N(C(O)=O)C(=O)CCl)CCC21CO2 USYHIIHUJPBCQG-UHFFFAOYSA-N 0.000 description 1
- QCSLIRFWJPOENV-UHFFFAOYSA-N (2-fluorophenyl)boronic acid Chemical compound OB(O)C1=CC=CC=C1F QCSLIRFWJPOENV-UHFFFAOYSA-N 0.000 description 1
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- JTBVPIHWMWILJU-MHZLTWQESA-N (2s)-2-(2-acetylanilino)-3-[4-[2-(5-methyl-2-phenyl-1,3-oxazol-4-yl)ethoxy]phenyl]propanoic acid Chemical compound CC(=O)C1=CC=CC=C1N[C@H](C(O)=O)CC(C=C1)=CC=C1OCCC1=C(C)OC(C=2C=CC=CC=2)=N1 JTBVPIHWMWILJU-MHZLTWQESA-N 0.000 description 1
- VGSJXSLGVQINOL-MHZLTWQESA-N (2s)-2-[4-[2-[(2,4-difluorophenyl)carbamoyl-heptylamino]ethyl]phenoxy]-2-methylbutanoic acid Chemical compound C=1C=C(F)C=C(F)C=1NC(=O)N(CCCCCCC)CCC1=CC=C(O[C@@](C)(CC)C(O)=O)C=C1 VGSJXSLGVQINOL-MHZLTWQESA-N 0.000 description 1
- ZUQBAQVRAURMCL-DOMZBBRYSA-N (2s)-2-[[4-[2-[(6r)-2-amino-4-oxo-5,6,7,8-tetrahydro-1h-pyrido[2,3-d]pyrimidin-6-yl]ethyl]benzoyl]amino]pentanedioic acid Chemical compound C([C@@H]1CC=2C(=O)N=C(NC=2NC1)N)CC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 ZUQBAQVRAURMCL-DOMZBBRYSA-N 0.000 description 1
- WMUIIGVAWPWQAW-DEOSSOPVSA-N (2s)-2-ethoxy-3-{4-[2-(10h-phenoxazin-10-yl)ethoxy]phenyl}propanoic acid Chemical compound C1=CC(C[C@H](OCC)C(O)=O)=CC=C1OCCN1C2=CC=CC=C2OC2=CC=CC=C21 WMUIIGVAWPWQAW-DEOSSOPVSA-N 0.000 description 1
- MHFUWOIXNMZFIW-WNQIDUERSA-N (2s)-2-hydroxypropanoic acid;n-[4-[4-(4-methylpiperazin-1-yl)-6-[(5-methyl-1h-pyrazol-3-yl)amino]pyrimidin-2-yl]sulfanylphenyl]cyclopropanecarboxamide Chemical compound C[C@H](O)C(O)=O.C1CN(C)CCN1C1=CC(NC2=NNC(C)=C2)=NC(SC=2C=CC(NC(=O)C3CC3)=CC=2)=N1 MHFUWOIXNMZFIW-WNQIDUERSA-N 0.000 description 1
- IRAAJHYKQDFNFO-SFHVURJKSA-N (2s)-3-[4-[2-[1,3-benzoxazol-2-yl(methyl)amino]ethoxy]phenyl]-2-(2,2,2-trifluoroethoxy)propanoic acid Chemical compound N=1C2=CC=CC=C2OC=1N(C)CCOC1=CC=C(C[C@H](OCC(F)(F)F)C(O)=O)C=C1 IRAAJHYKQDFNFO-SFHVURJKSA-N 0.000 description 1
- XSAKVDNHFRWJKS-IIZANFQQSA-N (2s)-n-benzyl-1-[(2s)-1-[(2s)-2-[[(2s)-2-[[(2s)-2-(dimethylamino)-3-methylbutanoyl]amino]-3-methylbutanoyl]-methylamino]-3-methylbutanoyl]pyrrolidine-2-carbonyl]pyrrolidine-2-carboxamide Chemical compound CC(C)[C@H](N(C)C)C(=O)N[C@@H](C(C)C)C(=O)N(C)[C@@H](C(C)C)C(=O)N1CCC[C@H]1C(=O)N1[C@H](C(=O)NCC=2C=CC=CC=2)CCC1 XSAKVDNHFRWJKS-IIZANFQQSA-N 0.000 description 1
- PSVUJBVBCOISSP-SPFKKGSWSA-N (2s,3r,4s,5s,6r)-2-bis(2-chloroethylamino)phosphoryloxy-6-(hydroxymethyl)oxane-3,4,5-triol Chemical compound OC[C@H]1O[C@@H](OP(=O)(NCCCl)NCCCl)[C@H](O)[C@@H](O)[C@@H]1O PSVUJBVBCOISSP-SPFKKGSWSA-N 0.000 description 1
- FELGMEQIXOGIFQ-CYBMUJFWSA-N (3r)-9-methyl-3-[(2-methylimidazol-1-yl)methyl]-2,3-dihydro-1h-carbazol-4-one Chemical compound CC1=NC=CN1C[C@@H]1C(=O)C(C=2C(=CC=CC=2)N2C)=C2CC1 FELGMEQIXOGIFQ-CYBMUJFWSA-N 0.000 description 1
- OBQRODBYVNIZJU-UHFFFAOYSA-N (4-acetylphenyl)boronic acid Chemical compound CC(=O)C1=CC=C(B(O)O)C=C1 OBQRODBYVNIZJU-UHFFFAOYSA-N 0.000 description 1
- DLROLUIVVKTFPW-LVEBQJTPSA-N (5s,5as,8ar,9r)-9-(4-hydroxy-3,5-dimethoxyphenyl)-5-(4-nitroanilino)-5a,6,8a,9-tetrahydro-5h-[2]benzofuro[5,6-f][1,3]benzodioxol-8-one Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](NC=3C=CC(=CC=3)[N+]([O-])=O)[C@@H]3[C@@H]2C(OC3)=O)=C1 DLROLUIVVKTFPW-LVEBQJTPSA-N 0.000 description 1
- WTSKMKRYHATLLL-UHFFFAOYSA-N (6-benzoyloxy-3-cyanopyridin-2-yl) 3-[3-(ethoxymethyl)-5-fluoro-2,6-dioxopyrimidine-1-carbonyl]benzoate Chemical compound O=C1N(COCC)C=C(F)C(=O)N1C(=O)C1=CC=CC(C(=O)OC=2C(=CC=C(OC(=O)C=3C=CC=CC=3)N=2)C#N)=C1 WTSKMKRYHATLLL-UHFFFAOYSA-N 0.000 description 1
- XDDGKNRSCDEWBR-UHFFFAOYSA-N (6-bromopyridin-2-yl)methanol Chemical compound OCC1=CC=CC(Br)=N1 XDDGKNRSCDEWBR-UHFFFAOYSA-N 0.000 description 1
- WPAPNCXMYWRTTL-UHFFFAOYSA-N (6-chloropyridin-3-yl)boronic acid Chemical compound OB(O)C1=CC=C(Cl)N=C1 WPAPNCXMYWRTTL-UHFFFAOYSA-N 0.000 description 1
- OPBPMGYBSDKJBT-DQHLZUIQSA-N (7s,9r,10r)-9-ethyl-4,6,9,10,11-pentahydroxy-7-[(2r,4s,5s,6s)-5-hydroxy-6-methyl-4-morpholin-4-yloxan-2-yl]oxy-8,10-dihydro-7h-tetracene-5,12-dione Chemical compound N1([C@H]2C[C@@H](O[C@@H](C)[C@H]2O)O[C@H]2C[C@]([C@@H](C3=C(O)C=4C(=O)C5=CC=CC(O)=C5C(=O)C=4C(O)=C32)O)(O)CC)CCOCC1 OPBPMGYBSDKJBT-DQHLZUIQSA-N 0.000 description 1
- BSRQHWFOFMAZRL-BODGVHBXSA-N (7s,9s)-7-[(2r,4s,5s,6s)-5-[(2s,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-4-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-8,10-dihydro-7h-tetracene-5,12-dione;hydron;chloride Chemical compound Cl.C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@H]1[C@@H](O)C[C@H](O[C@@H]2C3=C(O)C=4C(=O)C5=CC=CC=C5C(=O)C=4C(O)=C3C[C@](O)(C2)C(=O)CO)O[C@H]1C BSRQHWFOFMAZRL-BODGVHBXSA-N 0.000 description 1
- VNTHYLVDGVBPOU-QQYBVWGSSA-N (7s,9s)-9-acetyl-7-[(2r,4s,5s,6s)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;2-hydroxypropane-1,2,3-tricarboxylic acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O.O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 VNTHYLVDGVBPOU-QQYBVWGSSA-N 0.000 description 1
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- GVJHHUAWPYXKBD-IEOSBIPESA-N (R)-alpha-Tocopherol Natural products OC1=C(C)C(C)=C2O[C@@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C GVJHHUAWPYXKBD-IEOSBIPESA-N 0.000 description 1
- LKJPYSCBVHEWIU-KRWDZBQOSA-N (R)-bicalutamide Chemical compound C([C@@](O)(C)C(=O)NC=1C=C(C(C#N)=CC=1)C(F)(F)F)S(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-KRWDZBQOSA-N 0.000 description 1
- BKMMTJMQCTUHRP-VKHMYHEASA-N (S)-2-aminopropan-1-ol Chemical compound C[C@H](N)CO BKMMTJMQCTUHRP-VKHMYHEASA-N 0.000 description 1
- ZGNLFUXWZJGETL-YUSKDDKASA-N (Z)-[(2S)-2-amino-2-carboxyethyl]-hydroxyimino-oxidoazanium Chemical compound N[C@@H](C\[N+]([O-])=N\O)C(O)=O ZGNLFUXWZJGETL-YUSKDDKASA-N 0.000 description 1
- QRPSQQUYPMFERG-LFYBBSHMSA-N (e)-5-[3-(benzenesulfonamido)phenyl]-n-hydroxypent-2-en-4-ynamide Chemical compound ONC(=O)\C=C\C#CC1=CC=CC(NS(=O)(=O)C=2C=CC=CC=2)=C1 QRPSQQUYPMFERG-LFYBBSHMSA-N 0.000 description 1
- XGQXULJHBWKUJY-LYIKAWCPSA-N (z)-but-2-enedioic acid;n-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-2-oxo-1h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide Chemical compound OC(=O)\C=C/C(O)=O.CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C XGQXULJHBWKUJY-LYIKAWCPSA-N 0.000 description 1
- KPZGRMZPZLOPBS-UHFFFAOYSA-N 1,3-dichloro-2,2-bis(chloromethyl)propane Chemical compound ClCC(CCl)(CCl)CCl KPZGRMZPZLOPBS-UHFFFAOYSA-N 0.000 description 1
- 125000005940 1,4-dioxanyl group Chemical group 0.000 description 1
- NDOVLWQBFFJETK-UHFFFAOYSA-N 1,4-thiazinane 1,1-dioxide Chemical compound O=S1(=O)CCNCC1 NDOVLWQBFFJETK-UHFFFAOYSA-N 0.000 description 1
- VAMFSFIPDOODFH-UHFFFAOYSA-N 1-(3,4-dichlorophenyl)-3-(2,3-dihydro-1-benzofuran-5-ylsulfonyl)urea Chemical compound C1=C(Cl)C(Cl)=CC=C1NC(=O)NS(=O)(=O)C1=CC=C(OCC2)C2=C1 VAMFSFIPDOODFH-UHFFFAOYSA-N 0.000 description 1
- WOLKOZYBCJFJCM-UHFFFAOYSA-N 1-(6-bromopyridin-2-yl)ethanol Chemical compound CC(O)C1=CC=CC(Br)=N1 WOLKOZYBCJFJCM-UHFFFAOYSA-N 0.000 description 1
- DBJNIJFYCYARLW-UHFFFAOYSA-N 1-[2-methyl-6-[[5-(6-morpholin-4-ylpyridin-3-yl)thiophen-2-yl]amino]pyridin-3-yl]ethanol Chemical compound N1=C(C)C(C(O)C)=CC=C1NC1=CC=C(C=2C=NC(=CC=2)N2CCOCC2)S1 DBJNIJFYCYARLW-UHFFFAOYSA-N 0.000 description 1
- XAGOWQUMIXRYKG-UHFFFAOYSA-N 1-[[6-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-n,n-dimethyltriazole-4-carboxamide Chemical compound N1=NC(C(=O)N(C)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 XAGOWQUMIXRYKG-UHFFFAOYSA-N 0.000 description 1
- KCSPLSJKNVHJML-UHFFFAOYSA-N 1-[[6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]-n-methyltriazole-4-carboxamide Chemical compound N1=NC(C(=O)NC)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 KCSPLSJKNVHJML-UHFFFAOYSA-N 0.000 description 1
- RRMGJFCUWRZWLW-UHFFFAOYSA-N 1-[[6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]triazole-4-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC(=C2)C(N)=O)=N1 RRMGJFCUWRZWLW-UHFFFAOYSA-N 0.000 description 1
- VGUNPRNQXXTCCL-UHFFFAOYSA-N 1-chloro-4-prop-2-enylbenzene Chemical compound ClC1=CC=C(CC=C)C=C1 VGUNPRNQXXTCCL-UHFFFAOYSA-N 0.000 description 1
- VPDNERVQTLFVLS-UHFFFAOYSA-N 1-ethenylsulfonylethene Chemical compound C=CS(=O)(=O)C=C.C=CS(=O)(=O)C=C VPDNERVQTLFVLS-UHFFFAOYSA-N 0.000 description 1
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- OQTGVUOABUAUIJ-UHFFFAOYSA-N 1-methyl-4-(pyridin-2-ylmethyl)piperazine Chemical compound C1CN(C)CCN1CC1=CC=CC=N1 OQTGVUOABUAUIJ-UHFFFAOYSA-N 0.000 description 1
- HLXOVAMYQUFLPE-UHFFFAOYSA-N 1-methyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyrazole Chemical compound CN1N=CC=C1B1OC(C)(C)C(C)(C)O1 HLXOVAMYQUFLPE-UHFFFAOYSA-N 0.000 description 1
- QPBYBLZYMNWGMO-UHFFFAOYSA-N 2,2,3-trimethyloxirane Chemical compound CC1OC1(C)C QPBYBLZYMNWGMO-UHFFFAOYSA-N 0.000 description 1
- ROZCIVXTLACYNY-UHFFFAOYSA-N 2,3,4,5,6-pentafluoro-n-(3-fluoro-4-methoxyphenyl)benzenesulfonamide Chemical compound C1=C(F)C(OC)=CC=C1NS(=O)(=O)C1=C(F)C(F)=C(F)C(F)=C1F ROZCIVXTLACYNY-UHFFFAOYSA-N 0.000 description 1
- KPJIEPBITZLHPQ-UHFFFAOYSA-N 2,4,6-trifluorobenzaldehyde Chemical compound FC1=CC(F)=C(C=O)C(F)=C1 KPJIEPBITZLHPQ-UHFFFAOYSA-N 0.000 description 1
- ZHXUWDPHUQHFOV-UHFFFAOYSA-N 2,5-dibromopyridine Chemical compound BrC1=CC=C(Br)N=C1 ZHXUWDPHUQHFOV-UHFFFAOYSA-N 0.000 description 1
- FHVNVODIGXAGOT-UHFFFAOYSA-N 2-(2,4,6-trifluorophenyl)acetaldehyde Chemical compound FC1=CC(F)=C(CC=O)C(F)=C1 FHVNVODIGXAGOT-UHFFFAOYSA-N 0.000 description 1
- UEJJHQNACJXSKW-UHFFFAOYSA-N 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione Chemical compound O=C1C2=CC=CC=C2C(=O)N1C1CCC(=O)NC1=O UEJJHQNACJXSKW-UHFFFAOYSA-N 0.000 description 1
- MWSNYBOKRSGWAN-UHFFFAOYSA-N 2-(4-chlorophenyl)acetaldehyde Chemical compound ClC1=CC=C(CC=O)C=C1 MWSNYBOKRSGWAN-UHFFFAOYSA-N 0.000 description 1
- KCXZRESSSSYYCW-UHFFFAOYSA-N 2-(4-fluorophenyl)acetaldehyde Chemical compound FC1=CC=C(CC=O)C=C1 KCXZRESSSSYYCW-UHFFFAOYSA-N 0.000 description 1
- RDQCATQYMZDJLU-UHFFFAOYSA-N 2-(5-bromopyridin-2-yl)propan-2-ol Chemical compound CC(C)(O)C1=CC=C(Br)C=N1 RDQCATQYMZDJLU-UHFFFAOYSA-N 0.000 description 1
- MFOBLZAKOUBTGW-UHFFFAOYSA-N 2-(5-bromopyridin-2-yl)propan-2-yloxy-tert-butyl-dimethylsilane Chemical compound CC(C)(C)[Si](C)(C)OC(C)(C)C1=CC=C(Br)C=N1 MFOBLZAKOUBTGW-UHFFFAOYSA-N 0.000 description 1
- QXXFENKOGOCHSU-UHFFFAOYSA-N 2-(6-bromopyridin-3-yl)propan-2-ol Chemical compound CC(C)(O)C1=CC=C(Br)N=C1 QXXFENKOGOCHSU-UHFFFAOYSA-N 0.000 description 1
- XWNJMSJGJFSGRY-UHFFFAOYSA-N 2-(benzylamino)-3,7-dihydropurin-6-one Chemical compound N1C=2N=CNC=2C(=O)N=C1NCC1=CC=CC=C1 XWNJMSJGJFSGRY-UHFFFAOYSA-N 0.000 description 1
- AGUDSFZEJYRLGH-UHFFFAOYSA-N 2-(morpholin-4-ylmethyl)-n-[5-(6-morpholin-4-ylpyridin-3-yl)thiophen-2-yl]pyrimidin-4-amine Chemical compound N=1C=CC(NC=2SC(=CC=2)C=2C=NC(=CC=2)N2CCOCC2)=NC=1CN1CCOCC1 AGUDSFZEJYRLGH-UHFFFAOYSA-N 0.000 description 1
- IMSODMZESSGVBE-UHFFFAOYSA-N 2-Oxazoline Chemical compound C1CN=CO1 IMSODMZESSGVBE-UHFFFAOYSA-N 0.000 description 1
- OQQQETZKOKCPSB-UHFFFAOYSA-N 2-[(5-acetamido-6-methylpyridin-2-yl)amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound N1=C(C)C(NC(=O)C)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2F)C(C)(C)O)F)S1 OQQQETZKOKCPSB-UHFFFAOYSA-N 0.000 description 1
- HJFHJDLHKLIKJR-UHFFFAOYSA-N 2-[(6-cyanopyridin-2-yl)amino]-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C#N)=N1 HJFHJDLHKLIKJR-UHFFFAOYSA-N 0.000 description 1
- QXLQZLBNPTZMRK-UHFFFAOYSA-N 2-[(dimethylamino)methyl]-1-(2,4-dimethylphenyl)prop-2-en-1-one Chemical compound CN(C)CC(=C)C(=O)C1=CC=C(C)C=C1C QXLQZLBNPTZMRK-UHFFFAOYSA-N 0.000 description 1
- DRKPKFOWVXKPTD-UHFFFAOYSA-N 2-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]acetaldehyde Chemical compound CC(C)(O)C1=CC(F)=C(CC=O)C(F)=C1 DRKPKFOWVXKPTD-UHFFFAOYSA-N 0.000 description 1
- QUNOQBDEVTWCTA-UHFFFAOYSA-N 2-[2-[3-[2-(1,3-dioxobenzo[de]isoquinolin-2-yl)ethylamino]propylamino]ethyl]benzo[de]isoquinoline-1,3-dione Chemical compound C1=CC(C(=O)N(CCNCCCNCCN2C(C=3C=CC=C4C=CC=C(C=34)C2=O)=O)C2=O)=C3C2=CC=CC3=C1 QUNOQBDEVTWCTA-UHFFFAOYSA-N 0.000 description 1
- STHKNLNWXGPXEN-UHFFFAOYSA-N 2-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]acetaldehyde Chemical compound CC(C)(O)C1=CC=C(CC=O)C(F)=C1 STHKNLNWXGPXEN-UHFFFAOYSA-N 0.000 description 1
- SZAGFEHWHJUDFE-UHFFFAOYSA-N 2-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=C(C=CC(=C1)C(C)(C)O)C=1SC=CC=1C(=O)N SZAGFEHWHJUDFE-UHFFFAOYSA-N 0.000 description 1
- CVXSCZYBJFXAPZ-UHFFFAOYSA-N 2-[3,5-difluoro-4-(2-methoxyethenyl)phenyl]propan-2-ol Chemical compound COC=CC1=C(F)C=C(C(C)(C)O)C=C1F CVXSCZYBJFXAPZ-UHFFFAOYSA-N 0.000 description 1
- FFBNSFODWCNIQS-UHFFFAOYSA-N 2-[3,5-difluoro-4-[5-[(6-methylpyridin-2-yl)amino]thiophen-2-yl]phenyl]propan-2-ol Chemical compound CC1=CC=CC(NC=2SC(=CC=2)C=2C(=CC(=CC=2F)C(C)(C)O)F)=N1 FFBNSFODWCNIQS-UHFFFAOYSA-N 0.000 description 1
- NDLLVIABATXOCX-UHFFFAOYSA-N 2-[3,5-difluoro-4-[5-[(6-phenylpyridin-2-yl)amino]thiophen-2-yl]phenyl]propan-2-ol Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC=C1NC1=CC=CC(C=2C=CC=CC=2)=N1 NDLLVIABATXOCX-UHFFFAOYSA-N 0.000 description 1
- KXXARYDUDQBNCF-UHFFFAOYSA-N 2-[3-fluoro-4-(2-methoxyethenyl)phenyl]propan-2-ol Chemical compound COC=CC1=CC=C(C(C)(C)O)C=C1F KXXARYDUDQBNCF-UHFFFAOYSA-N 0.000 description 1
- FUIQEVGCHFNIQY-UHFFFAOYSA-N 2-[3-fluoro-4-[5-[(2-methylpyrimidin-4-yl)amino]thiophen-2-yl]phenyl]propan-2-ol Chemical compound CC1=NC=CC(NC=2SC(=CC=2)C=2C(=CC(=CC=2)C(C)(C)O)F)=N1 FUIQEVGCHFNIQY-UHFFFAOYSA-N 0.000 description 1
- VWKWWTPHUQXXJL-UHFFFAOYSA-N 2-[4-(2-hydroxypropan-2-yl)phenyl]acetaldehyde Chemical compound CC(C)(O)C1=CC=C(CC=O)C=C1 VWKWWTPHUQXXJL-UHFFFAOYSA-N 0.000 description 1
- CJCKXIMSQYAMHG-UHFFFAOYSA-N 2-[4-[5-[[6-(2-hydroxyethoxy)pyridin-2-yl]amino]thiophen-2-yl]phenyl]propan-2-ol Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC=C1NC1=CC=CC(OCCO)=N1 CJCKXIMSQYAMHG-UHFFFAOYSA-N 0.000 description 1
- JBXHMDLKOUNNES-UHFFFAOYSA-N 2-[[2-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]pyrimidin-4-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2CCS(=O)(=O)CC2)=N1 JBXHMDLKOUNNES-UHFFFAOYSA-N 0.000 description 1
- MFUCOINADABTNC-UHFFFAOYSA-N 2-[[3-(4-bromophenyl)-1,2,4-triazol-1-yl]methoxy]ethyl-trimethylsilane Chemical compound C[Si](C)(C)CCOCN1C=NC(C=2C=CC(Br)=CC=2)=N1 MFUCOINADABTNC-UHFFFAOYSA-N 0.000 description 1
- NFXWXQLZQRTPQI-UHFFFAOYSA-N 2-[[4-(4-bromophenyl)triazol-1-yl]methoxy]ethyl-trimethylsilane Chemical compound N1=NN(COCC[Si](C)(C)C)C=C1C1=CC=C(Br)C=C1 NFXWXQLZQRTPQI-UHFFFAOYSA-N 0.000 description 1
- NHTRSESQWOKOIU-UHFFFAOYSA-N 2-[[5-(4-bromophenyl)-1,2,4-triazol-1-yl]methoxy]ethyl-trimethylsilane Chemical compound C[Si](C)(C)CCOCN1N=CN=C1C1=CC=C(Br)C=C1 NHTRSESQWOKOIU-UHFFFAOYSA-N 0.000 description 1
- PZESVFXEBXBNQY-UHFFFAOYSA-N 2-[[6-(1-cyano-3-hydroxypropyl)pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CCO)C#N)=N1 PZESVFXEBXBNQY-UHFFFAOYSA-N 0.000 description 1
- DBYNLTZUIFJFIM-UHFFFAOYSA-N 2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]-5-(2h-triazol-4-yl)thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2N=NNC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 DBYNLTZUIFJFIM-UHFFFAOYSA-N 0.000 description 1
- ISOQSEUOZSIELI-UHFFFAOYSA-N 2-[[6-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCS(=O)(=O)CC2)=N1 ISOQSEUOZSIELI-UHFFFAOYSA-N 0.000 description 1
- UPMVZHWSMMTOIA-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-(1h-1,2,4-triazol-5-yl)thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C2=NNC=N2)C(N)=O)=N1 UPMVZHWSMMTOIA-UHFFFAOYSA-N 0.000 description 1
- DPZUMPBRFHVJES-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=NC(=CC=2)N2CCOCC2)C(N)=O)=N1 DPZUMPBRFHVJES-UHFFFAOYSA-N 0.000 description 1
- YRCGUXIQVIQZFW-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-[4-(2h-triazol-4-yl)phenyl]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C=2N=NNC=2)C(N)=O)=N1 YRCGUXIQVIQZFW-UHFFFAOYSA-N 0.000 description 1
- LOFCVUGJNUEPSY-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-[4-(morpholin-4-ylmethyl)phenyl]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(CN3CCOCC3)=CC=2)C(N)=O)=N1 LOFCVUGJNUEPSY-UHFFFAOYSA-N 0.000 description 1
- UCSRKJCTHQSZNW-UHFFFAOYSA-N 2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]-5-[4-(morpholine-4-carbonyl)phenyl]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(=O)N2CCOCC2)C(N)=O)=N1 UCSRKJCTHQSZNW-UHFFFAOYSA-N 0.000 description 1
- RJSHWWUKSPVILZ-UHFFFAOYSA-N 2-[[6-[(3,3-difluoroazetidin-1-yl)methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CC(F)(F)C2)=N1 RJSHWWUKSPVILZ-UHFFFAOYSA-N 0.000 description 1
- WDCGIBZLZHMXJI-UHFFFAOYSA-N 2-[[6-[1-(1,1-dioxo-1,4-thiazinan-4-yl)-2-hydroxyethyl]pyridin-2-yl]amino]-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2CCS(=O)(=O)CC2)=N1 WDCGIBZLZHMXJI-UHFFFAOYSA-N 0.000 description 1
- JVZCDOGLUJXFLL-UHFFFAOYSA-N 2-[[6-[[(1,1-dioxothiolan-3-yl)amino]methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC2CS(=O)(=O)CC2)=N1 JVZCDOGLUJXFLL-UHFFFAOYSA-N 0.000 description 1
- IXEXYMYAVXXLCF-UHFFFAOYSA-N 2-[[6-[[3-(dimethylamino)piperidin-1-yl]methyl]pyridin-2-yl]amino]-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1C(N(C)C)CCCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 IXEXYMYAVXXLCF-UHFFFAOYSA-N 0.000 description 1
- SCNWEDMMBGWSKH-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-(4-morpholin-4-ylphenyl)thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)N2CCOCC2)C(N)=O)=N1 SCNWEDMMBGWSKH-UHFFFAOYSA-N 0.000 description 1
- PLEXNDSGQQUSMQ-UHFFFAOYSA-N 2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]-5-[6-(1-methylpyrazol-4-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1C1=CC=C(C=2SC(NC=3N=C(CN4N=NC(=C4)C(C)(C)O)C=CC=3)=C(C(N)=O)C=2)C=N1 PLEXNDSGQQUSMQ-UHFFFAOYSA-N 0.000 description 1
- 125000005273 2-acetoxybenzoic acid group Chemical group 0.000 description 1
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 1
- WCCAPKHZQRQPGJ-UHFFFAOYSA-N 2-amino-5-(2,4,6-trifluorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=C(F)C=C(F)C=C1F WCCAPKHZQRQPGJ-UHFFFAOYSA-N 0.000 description 1
- QUACYBLURPXQEA-UHFFFAOYSA-N 2-amino-5-(2,5-dichlorophenyl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC(Cl)=CC=C1Cl QUACYBLURPXQEA-UHFFFAOYSA-N 0.000 description 1
- GWTIEKVWNLIBIZ-UHFFFAOYSA-N 2-amino-5-(4-methylsulfonylphenyl)thiophene-3-carboxamide Chemical compound C1=CC(S(=O)(=O)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 GWTIEKVWNLIBIZ-UHFFFAOYSA-N 0.000 description 1
- GWYZTHLKHYWQMV-UHFFFAOYSA-N 2-amino-5-(6-morpholin-4-ylpyridin-3-yl)thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(N2CCOCC2)N=C1 GWYZTHLKHYWQMV-UHFFFAOYSA-N 0.000 description 1
- HLIMBOCCYXHLJD-UHFFFAOYSA-N 2-amino-5-[1-(2-trimethylsilylethoxymethyl)-1,2,4-triazol-3-yl]thiophene-3-carboxamide Chemical compound C[Si](C)(C)CCOCN1C=NC(C=2SC(N)=C(C(N)=O)C=2)=N1 HLIMBOCCYXHLJD-UHFFFAOYSA-N 0.000 description 1
- BLOPQIAUSFUIFG-UHFFFAOYSA-N 2-amino-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 BLOPQIAUSFUIFG-UHFFFAOYSA-N 0.000 description 1
- YDHVPMNVUYTJCI-UHFFFAOYSA-N 2-amino-5-[4-(1-hydroxyethyl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(O)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 YDHVPMNVUYTJCI-UHFFFAOYSA-N 0.000 description 1
- QUJICLBPMKSQDG-UHFFFAOYSA-N 2-amino-5-[4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound C1=CC(C(C)(O)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 QUJICLBPMKSQDG-UHFFFAOYSA-N 0.000 description 1
- RENRXEVPZMWRIU-UHFFFAOYSA-N 2-amino-5-[4-(3-fluorooxetan-3-yl)phenyl]thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(C2(F)COC2)C=C1 RENRXEVPZMWRIU-UHFFFAOYSA-N 0.000 description 1
- GCMNWTAHFMIFEF-UHFFFAOYSA-N 2-amino-5-[4-(3-hydroxyoxetan-3-yl)phenyl]thiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=C(C2(O)COC2)C=C1 GCMNWTAHFMIFEF-UHFFFAOYSA-N 0.000 description 1
- GDUAFOCWHXBSQV-UHFFFAOYSA-N 2-amino-5-[6-(2-cyanopropan-2-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound C1=NC(C(C)(C#N)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 GDUAFOCWHXBSQV-UHFFFAOYSA-N 0.000 description 1
- KWDGJCXTTWZBIZ-UHFFFAOYSA-N 2-amino-5-[6-(2-hydroxypropan-2-yl)pyridin-3-yl]thiophene-3-carboxamide Chemical compound C1=NC(C(C)(O)C)=CC=C1C1=CC(C(N)=O)=C(N)S1 KWDGJCXTTWZBIZ-UHFFFAOYSA-N 0.000 description 1
- UHEGYTDIDBFUJD-UHFFFAOYSA-N 2-amino-5-phenylthiophene-3-carboxamide Chemical compound S1C(N)=C(C(=O)N)C=C1C1=CC=CC=C1 UHEGYTDIDBFUJD-UHFFFAOYSA-N 0.000 description 1
- 125000000022 2-aminoethyl group Chemical group [H]C([*])([H])C([H])([H])N([H])[H] 0.000 description 1
- 125000004174 2-benzimidazolyl group Chemical group [H]N1C(*)=NC2=C([H])C([H])=C([H])C([H])=C12 0.000 description 1
- CBIAKDAYHRWZCU-UHFFFAOYSA-N 2-bromo-4-[(6,7-dimethoxyquinazolin-4-yl)amino]phenol Chemical compound C=12C=C(OC)C(OC)=CC2=NC=NC=1NC1=CC=C(O)C(Br)=C1 CBIAKDAYHRWZCU-UHFFFAOYSA-N 0.000 description 1
- ZPNFMDYBAQDFDY-UHFFFAOYSA-N 2-bromo-5-nitrothiophene Chemical compound [O-][N+](=O)C1=CC=C(Br)S1 ZPNFMDYBAQDFDY-UHFFFAOYSA-N 0.000 description 1
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 1
- 229940013085 2-diethylaminoethanol Drugs 0.000 description 1
- 150000005749 2-halopyridines Chemical class 0.000 description 1
- YEDUAINPPJYDJZ-UHFFFAOYSA-N 2-hydroxybenzothiazole Chemical compound C1=CC=C2SC(O)=NC2=C1 YEDUAINPPJYDJZ-UHFFFAOYSA-N 0.000 description 1
- XWKFPIODWVPXLX-UHFFFAOYSA-N 2-methyl-5-methylpyridine Natural products CC1=CC=C(C)N=C1 XWKFPIODWVPXLX-UHFFFAOYSA-N 0.000 description 1
- SJHWDQKRGQLVSH-UHFFFAOYSA-N 2-nitrothiophene-3-carboxamide Chemical compound NC(=O)C=1C=CSC=1[N+]([O-])=O SJHWDQKRGQLVSH-UHFFFAOYSA-N 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- FFRFGVHNKJYNOV-DOVUUNBWSA-N 3',4'-Anhydrovinblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C=C(C2)CC)N2CCC2=C1NC1=CC=CC=C21 FFRFGVHNKJYNOV-DOVUUNBWSA-N 0.000 description 1
- JLNPZZYATPDOHX-UHFFFAOYSA-N 3-(2-hydroxyethyl)pyridine-2-carboxamide Chemical compound NC(=O)C1=NC=CC=C1CCO JLNPZZYATPDOHX-UHFFFAOYSA-N 0.000 description 1
- SQVOYDWWROMJHE-UHFFFAOYSA-N 3-(4-bromophenyl)-3-fluorooxetane Chemical compound C=1C=C(Br)C=CC=1C1(F)COC1 SQVOYDWWROMJHE-UHFFFAOYSA-N 0.000 description 1
- KNQDDTUCGFNECR-UHFFFAOYSA-N 3-(4-bromophenyl)oxetan-3-ol Chemical compound C=1C=C(Br)C=CC=1C1(O)COC1 KNQDDTUCGFNECR-UHFFFAOYSA-N 0.000 description 1
- HRKYJXZQZROJJV-UHFFFAOYSA-N 3-(4-chlorophenyl)propane-1,2-diol Chemical compound OCC(O)CC1=CC=C(Cl)C=C1 HRKYJXZQZROJJV-UHFFFAOYSA-N 0.000 description 1
- JJYHGJHYAOCLSZ-UHFFFAOYSA-N 3-(4-fluorophenyl)propane-1,2-diol Chemical compound OCC(O)CC1=CC=C(F)C=C1 JJYHGJHYAOCLSZ-UHFFFAOYSA-N 0.000 description 1
- JAGAGRQCVBAHPT-UHFFFAOYSA-N 3-(cyclopropylmethyl)pyridine-2-carboxamide Chemical compound NC(=O)C1=NC=CC=C1CC1CC1 JAGAGRQCVBAHPT-UHFFFAOYSA-N 0.000 description 1
- QEMFQOAXARHQQI-UHFFFAOYSA-N 3-(oxetan-2-ylmethyl)pyridine-2-carboxamide Chemical compound NC(=O)C1=NC=CC=C1CC1OCC1 QEMFQOAXARHQQI-UHFFFAOYSA-N 0.000 description 1
- GDAGFEIIOHUGFY-LURJTMIESA-N 3-[(2R)-1-hydroxypropan-2-yl]pyridine-2-carboxamide Chemical compound OC[C@H](C)C=1C(=NC=CC=1)C(=O)N GDAGFEIIOHUGFY-LURJTMIESA-N 0.000 description 1
- VTEZCXWZRATCBO-LURJTMIESA-N 3-[(2S)-2-hydroxypropyl]pyridine-2-carboxamide Chemical compound O[C@H](CC=1C(=NC=CC=1)C(=O)N)C VTEZCXWZRATCBO-LURJTMIESA-N 0.000 description 1
- WUWDLXZGHZSWQZ-UHFFFAOYSA-N 3-[(3,5-dimethyl-1H-pyrrol-2-yl)methylidene]-1H-indol-2-one Chemical compound N1C(C)=CC(C)=C1C=C1C2=CC=CC=C2NC1=O WUWDLXZGHZSWQZ-UHFFFAOYSA-N 0.000 description 1
- WAXQRTJWNZIMMW-UHFFFAOYSA-N 3-[(6-bromopyridin-2-yl)methylamino]-2-methylbutan-2-ol Chemical compound CC(O)(C)C(C)NCC1=CC=CC(Br)=N1 WAXQRTJWNZIMMW-UHFFFAOYSA-N 0.000 description 1
- UZFPOOOQHWICKY-UHFFFAOYSA-N 3-[13-[1-[1-[8,12-bis(2-carboxyethyl)-17-(1-hydroxyethyl)-3,7,13,18-tetramethyl-21,24-dihydroporphyrin-2-yl]ethoxy]ethyl]-18-(2-carboxyethyl)-8-(1-hydroxyethyl)-3,7,12,17-tetramethyl-22,23-dihydroporphyrin-2-yl]propanoic acid Chemical compound N1C(C=C2C(=C(CCC(O)=O)C(C=C3C(=C(C)C(C=C4N5)=N3)CCC(O)=O)=N2)C)=C(C)C(C(C)O)=C1C=C5C(C)=C4C(C)OC(C)C1=C(N2)C=C(N3)C(C)=C(C(O)C)C3=CC(C(C)=C3CCC(O)=O)=NC3=CC(C(CCC(O)=O)=C3C)=NC3=CC2=C1C UZFPOOOQHWICKY-UHFFFAOYSA-N 0.000 description 1
- NHFDRBXTEDBWCZ-ZROIWOOFSA-N 3-[2,4-dimethyl-5-[(z)-(2-oxo-1h-indol-3-ylidene)methyl]-1h-pyrrol-3-yl]propanoic acid Chemical compound OC(=O)CCC1=C(C)NC(\C=C/2C3=CC=CC=C3NC\2=O)=C1C NHFDRBXTEDBWCZ-ZROIWOOFSA-N 0.000 description 1
- WUIABRMSWOKTOF-OYALTWQYSA-N 3-[[2-[2-[2-[[(2s,3r)-2-[[(2s,3s,4r)-4-[[(2s,3r)-2-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[(2r,3s,4s,5s,6s)-3-[(2r,3s,4s,5r,6r)-4-carbamoyloxy-3,5-dihydroxy-6-(hydroxymethyl)ox Chemical compound OS([O-])(=O)=O.N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1NC=NC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C WUIABRMSWOKTOF-OYALTWQYSA-N 0.000 description 1
- CURYRIVJTBNEGU-UHFFFAOYSA-L 3-bromo-1-[12-(3-bromopropanoyl)-3,12-diaza-6,9-diazoniadispiro[5.2.5^{9}.2^{6}]hexadecan-3-yl]propan-1-one;dichloride Chemical compound [Cl-].[Cl-].C1CN(C(=O)CCBr)CC[N+]21CC[N+]1(CCN(CC1)C(=O)CCBr)CC2 CURYRIVJTBNEGU-UHFFFAOYSA-L 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- MRWWWZLJWNIEEJ-UHFFFAOYSA-N 4,4,5,5-tetramethyl-2-propan-2-yloxy-1,3,2-dioxaborolane Chemical compound CC(C)OB1OC(C)(C)C(C)(C)O1 MRWWWZLJWNIEEJ-UHFFFAOYSA-N 0.000 description 1
- WEQPBCSPRXFQQS-UHFFFAOYSA-N 4,5-dihydro-1,2-oxazole Chemical compound C1CC=NO1 WEQPBCSPRXFQQS-UHFFFAOYSA-N 0.000 description 1
- WIYNWLBOSGNXEH-UHFFFAOYSA-N 4-(2-amino-6,7-dimethoxyquinazolin-4-yl)phenol Chemical compound C=12C=C(OC)C(OC)=CC2=NC(N)=NC=1C1=CC=C(O)C=C1 WIYNWLBOSGNXEH-UHFFFAOYSA-N 0.000 description 1
- KVISFFRCMNRVHU-UHFFFAOYSA-N 4-(3,5-difluorophenyl)morpholine Chemical compound FC1=CC(F)=CC(N2CCOCC2)=C1 KVISFFRCMNRVHU-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- YDNMNPHZKMEGDR-UHFFFAOYSA-N 4-(4-bromophenyl)-1,4-thiazinane 1,1-dioxide Chemical compound C1=CC(Br)=CC=C1N1CCS(=O)(=O)CC1 YDNMNPHZKMEGDR-UHFFFAOYSA-N 0.000 description 1
- IYLFMVWZQBQAOU-UHFFFAOYSA-N 4-[(6-bromopyridin-2-yl)methyl]morpholine Chemical compound BrC1=CC=CC(CN2CCOCC2)=N1 IYLFMVWZQBQAOU-UHFFFAOYSA-N 0.000 description 1
- JVYNJRBSXBYXQB-UHFFFAOYSA-N 4-[3-(4-carboxyphenoxy)propoxy]benzoic acid;decanedioic acid Chemical compound OC(=O)CCCCCCCCC(O)=O.C1=CC(C(=O)O)=CC=C1OCCCOC1=CC=C(C(O)=O)C=C1 JVYNJRBSXBYXQB-UHFFFAOYSA-N 0.000 description 1
- OZBUFFXESDBEHG-FXILSDISSA-N 4-[[(2e,4e,6e,8e)-3,7-dimethyl-9-(2,6,6-trimethylcyclohexen-1-yl)nona-2,4,6,8-tetraenoyl]amino]benzoic acid Chemical compound C=1C=C(C(O)=O)C=CC=1NC(=O)\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C OZBUFFXESDBEHG-FXILSDISSA-N 0.000 description 1
- QBQLYIISSRXYKL-UHFFFAOYSA-N 4-[[4-[2-(5-methyl-2-phenyl-1,3-oxazol-4-yl)ethoxy]phenyl]methyl]-1,2-oxazolidine-3,5-dione Chemical compound CC=1OC(C=2C=CC=CC=2)=NC=1CCOC(C=C1)=CC=C1CC1C(=O)NOC1=O QBQLYIISSRXYKL-UHFFFAOYSA-N 0.000 description 1
- YLDCUKJMEKGGFI-QCSRICIXSA-N 4-acetamidobenzoic acid;9-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3h-purin-6-one;1-(dimethylamino)propan-2-ol Chemical compound CC(O)CN(C)C.CC(O)CN(C)C.CC(O)CN(C)C.CC(=O)NC1=CC=C(C(O)=O)C=C1.CC(=O)NC1=CC=C(C(O)=O)C=C1.CC(=O)NC1=CC=C(C(O)=O)C=C1.O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C(NC=NC2=O)=C2N=C1 YLDCUKJMEKGGFI-QCSRICIXSA-N 0.000 description 1
- GFFXZLZWLOBBLO-BWVDBABLSA-N 4-amino-1-[(2r,4s,5r)-3-(fluoromethylidene)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidin-2-one Chemical compound O=C1N=C(N)C=CN1[C@H]1C(=CF)[C@H](O)[C@@H](CO)O1 GFFXZLZWLOBBLO-BWVDBABLSA-N 0.000 description 1
- PULHLIOPJXPGJN-BWVDBABLSA-N 4-amino-1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)-3-methylideneoxolan-2-yl]pyrimidin-2-one Chemical compound O=C1N=C(N)C=CN1[C@H]1C(=C)[C@H](O)[C@@H](CO)O1 PULHLIOPJXPGJN-BWVDBABLSA-N 0.000 description 1
- TVZGACDUOSZQKY-LBPRGKRZSA-N 4-aminofolic acid Chemical compound C1=NC2=NC(N)=NC(N)=C2N=C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 TVZGACDUOSZQKY-LBPRGKRZSA-N 0.000 description 1
- LOQLPNJGMBBLRT-UHFFFAOYSA-N 4-bromo-2-fluoro-1-(2-methoxyethenyl)benzene Chemical compound COC=CC1=CC=C(Br)C=C1F LOQLPNJGMBBLRT-UHFFFAOYSA-N 0.000 description 1
- UPCARQPLANFGQJ-UHFFFAOYSA-N 4-bromo-2-fluorobenzaldehyde Chemical compound FC1=CC(Br)=CC=C1C=O UPCARQPLANFGQJ-UHFFFAOYSA-N 0.000 description 1
- WDFQBORIUYODSI-UHFFFAOYSA-N 4-bromoaniline Chemical compound NC1=CC=C(Br)C=C1 WDFQBORIUYODSI-UHFFFAOYSA-N 0.000 description 1
- HQSCPPCMBMFJJN-UHFFFAOYSA-N 4-bromobenzonitrile Chemical compound BrC1=CC=C(C#N)C=C1 HQSCPPCMBMFJJN-UHFFFAOYSA-N 0.000 description 1
- HVCNXQOWACZAFN-UHFFFAOYSA-N 4-ethylmorpholine Chemical compound CCN1CCOCC1 HVCNXQOWACZAFN-UHFFFAOYSA-N 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- 150000005696 4-halopyrimidines Chemical class 0.000 description 1
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical compound C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 1
- 229940113081 5 Hydroxytryptamine 3 receptor antagonist Drugs 0.000 description 1
- LQOVJDXMIGMWSP-UHFFFAOYSA-N 5-(1-methylpyrazol-4-yl)-2-[(6-methylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound CC1=CC=CC(NC2=C(C=C(S2)C2=CN(C)N=C2)C(N)=O)=N1 LQOVJDXMIGMWSP-UHFFFAOYSA-N 0.000 description 1
- OIGBBOKDHJWBLK-UHFFFAOYSA-N 5-(2,5-dichlorophenyl)-2-[[6-(pyrrolidin-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC=C(Cl)C=2)Cl)SC=1NC(N=1)=CC=CC=1CN1CCCC1 OIGBBOKDHJWBLK-UHFFFAOYSA-N 0.000 description 1
- QWHAPABLCCLUNV-UHFFFAOYSA-N 5-(2,6-difluoro-4-morpholin-4-ylphenyl)-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C(=CC(=CC=2F)N2CCOCC2)F)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 QWHAPABLCCLUNV-UHFFFAOYSA-N 0.000 description 1
- VPHPMMAZSORYFP-UHFFFAOYSA-N 5-(2-aminopyrimidin-5-yl)-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=NC(N)=NC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 VPHPMMAZSORYFP-UHFFFAOYSA-N 0.000 description 1
- ZPXLEXXBDJBFAO-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(Cl)=CC=2)SC=1NC(N=1)=CC=CC=1CN1CCOCC1 ZPXLEXXBDJBFAO-UHFFFAOYSA-N 0.000 description 1
- IQNJFLZGWKJAAM-UHFFFAOYSA-N 5-(4-chlorophenyl)-2-[[6-[2-(cyanomethylamino)-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC(Cl)=CC=2)SC=1NC1=CC=CC(CC(=O)NCC#N)=N1 IQNJFLZGWKJAAM-UHFFFAOYSA-N 0.000 description 1
- GKJXBTMEUWIYLF-UHFFFAOYSA-N 5-(4-methylsulfonylphenyl)-2-nitrothiophene-3-carboxamide Chemical compound C1=CC(S(=O)(=O)C)=CC=C1C1=CC(C(N)=O)=C([N+]([O-])=O)S1 GKJXBTMEUWIYLF-UHFFFAOYSA-N 0.000 description 1
- CTXGOUGLNLCQKP-UHFFFAOYSA-N 5-(6-chloropyridin-3-yl)-2-nitrothiophene-3-carboxamide Chemical compound S1C([N+]([O-])=O)=C(C(=O)N)C=C1C1=CC=C(Cl)N=C1 CTXGOUGLNLCQKP-UHFFFAOYSA-N 0.000 description 1
- KDFBZDDSDLRPQR-UHFFFAOYSA-N 5-(6-morpholin-4-ylpyridin-3-yl)-2-nitrothiophene-3-carboxamide Chemical compound S1C([N+]([O-])=O)=C(C(=O)N)C=C1C1=CC=C(N2CCOCC2)N=C1 KDFBZDDSDLRPQR-UHFFFAOYSA-N 0.000 description 1
- 102000035037 5-HT3 receptors Human genes 0.000 description 1
- 108091005477 5-HT3 receptors Proteins 0.000 description 1
- NFFXEUUOMTXWCX-UHFFFAOYSA-N 5-[(2,4-dioxo-1,3-thiazolidin-5-yl)methyl]-2-methoxy-n-[[4-(trifluoromethyl)phenyl]methyl]benzamide Chemical compound C1=C(C(=O)NCC=2C=CC(=CC=2)C(F)(F)F)C(OC)=CC=C1CC1SC(=O)NC1=O NFFXEUUOMTXWCX-UHFFFAOYSA-N 0.000 description 1
- LGZKGOGODCLQHG-CYBMUJFWSA-N 5-[(2r)-2-hydroxy-2-(3,4,5-trimethoxyphenyl)ethyl]-2-methoxyphenol Chemical compound C1=C(O)C(OC)=CC=C1C[C@@H](O)C1=CC(OC)=C(OC)C(OC)=C1 LGZKGOGODCLQHG-CYBMUJFWSA-N 0.000 description 1
- MBSUUYBLMHXVFG-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[(6-pyridin-3-ylpyridin-2-yl)amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C=2C=NC=CC=2)=N1 MBSUUYBLMHXVFG-UHFFFAOYSA-N 0.000 description 1
- DPMXQYRWLUUBOM-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[2-(fluoromethyl)pyrimidin-4-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CF)=N1 DPMXQYRWLUUBOM-UHFFFAOYSA-N 0.000 description 1
- NEMRAXVICMFWSV-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-[3-(2-hydroxy-2-methylpropyl)triazol-4-yl]-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(C=2N(N=NC=2)CC(C)(C)O)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F NEMRAXVICMFWSV-UHFFFAOYSA-N 0.000 description 1
- URQOLCRDOGZBDU-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(1h-pyrazol-5-yl)pyridazin-3-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(C=2NN=CC=2)N=N1 URQOLCRDOGZBDU-UHFFFAOYSA-N 0.000 description 1
- OIUMVSBILBFURI-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxypropyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 OIUMVSBILBFURI-UHFFFAOYSA-N 0.000 description 1
- ATJNOJLETCSLOE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(imidazol-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C=NC=C2)=N1 ATJNOJLETCSLOE-UHFFFAOYSA-N 0.000 description 1
- PEVAUEIRXBPRLP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(methanesulfonamidomethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNS(C)(=O)=O)=N1 PEVAUEIRXBPRLP-UHFFFAOYSA-N 0.000 description 1
- PLMUVWATPNLPKE-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(triazol-1-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC=C2)=N1 PLMUVWATPNLPKE-UHFFFAOYSA-N 0.000 description 1
- QMSIZZFYVDZGII-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(1,1-dioxo-1,4-thiazinan-4-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCS(=O)(=O)CC2)=N1 QMSIZZFYVDZGII-UHFFFAOYSA-N 0.000 description 1
- GFONEFAHYPGUPJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-morpholin-4-yl-2-oxoethoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(=O)N2CCOCC2)=N1 GFONEFAHYPGUPJ-UHFFFAOYSA-N 0.000 description 1
- SDPPSIABBJVRDX-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methyl-4-oxo-1,4$l^{5}-azaphosphinan-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCP(C)(=O)CC2)=N1 SDPPSIABBJVRDX-UHFFFAOYSA-N 0.000 description 1
- MNJYHIWHDBIHJR-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[1-hydroxy-2-(methylamino)-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CNC(=O)C(O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 MNJYHIWHDBIHJR-UHFFFAOYSA-N 0.000 description 1
- LEDWFUAUOQMLPW-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(1,1-dioxothian-4-yl)-2-hydroxyethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CC(O)C2CCS(=O)(=O)CC2)=N1 LEDWFUAUOQMLPW-UHFFFAOYSA-N 0.000 description 1
- XVHVFUZXBCUCFQ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(1-methylpyrazol-4-yl)amino]-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1=NN(C)C=C1NC(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 XVHVFUZXBCUCFQ-UHFFFAOYSA-N 0.000 description 1
- WLIJFZGVUUBNET-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(2-methylpyrazol-3-yl)amino]-2-oxoethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CN1N=CC=C1NC(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 WLIJFZGVUUBNET-UHFFFAOYSA-N 0.000 description 1
- GLJGUQIZGJGXJW-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-[(3-methyl-1,2,4-oxadiazol-5-yl)methoxy]ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC1=NOC(COCCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3F)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 GLJGUQIZGJGXJW-UHFFFAOYSA-N 0.000 description 1
- KBCXJPVSQSZXMJ-LBPRGKRZSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(2s)-2-hydroxypropoxy]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C[C@H](O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 KBCXJPVSQSZXMJ-LBPRGKRZSA-N 0.000 description 1
- CWHMBXVAYWPBGY-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3-oxocyclohexen-1-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC=2CCCC(=O)C=2)=N1 CWHMBXVAYWPBGY-UHFFFAOYSA-N 0.000 description 1
- UBCWOSPOCHARSI-CQSZACIVSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(3r)-3-methylmorpholin-4-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C[C@@H]1COCCN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 UBCWOSPOCHARSI-CQSZACIVSA-N 0.000 description 1
- QOOCRVOOFCBVNJ-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-(2-hydroxyethyl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC(CCO)=C2)=N1 QOOCRVOOFCBVNJ-UHFFFAOYSA-N 0.000 description 1
- XUMORSLFLBJPKC-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[4-(hydroxymethyl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2N=NC(CO)=C2)=N1 XUMORSLFLBJPKC-UHFFFAOYSA-N 0.000 description 1
- WFRLUVMBJLSERH-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-methyl-5-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=C(CN2CCOCC2)C(C)=NC=1NC(=C(C=1)C(N)=O)SC=1C1=C(F)C=C(C(C)(C)O)C=C1F WFRLUVMBJLSERH-UHFFFAOYSA-N 0.000 description 1
- MKWAGZFXOIAEQP-UHFFFAOYSA-N 5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C1=CC(C(N)=O)=CS1 MKWAGZFXOIAEQP-UHFFFAOYSA-N 0.000 description 1
- GMTZNFMWDNQPBC-UHFFFAOYSA-N 5-[2,6-difluoro-4-(3-hydroxyoxetan-3-yl)phenyl]-2-[[6-(ethylsulfonylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CCS(=O)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C2(O)COC2)F)C(N)=O)=N1 GMTZNFMWDNQPBC-UHFFFAOYSA-N 0.000 description 1
- GQQDMAHPFXGYHP-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[5-(hydroxymethyl)-6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=C(CO)C(CN2CCOCC2)=N1 GQQDMAHPFXGYHP-UHFFFAOYSA-N 0.000 description 1
- KNDXBQFITWKRAY-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-fluoro-1-morpholin-4-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CF)N2CCOCC2)=N1 KNDXBQFITWKRAY-UHFFFAOYSA-N 0.000 description 1
- DKMVLLQKBHDQTB-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2CCOCC2)=N1 DKMVLLQKBHDQTB-UHFFFAOYSA-N 0.000 description 1
- XXDZBMUYLXGODC-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-hydroxyethoxy)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(OCCO)=N1 XXDZBMUYLXGODC-UHFFFAOYSA-N 0.000 description 1
- IGUBXGBCWMEXJQ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-methoxy-1-morpholin-4-ylethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C=1C=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=NC=1C(COC)N1CCOCC1 IGUBXGBCWMEXJQ-UHFFFAOYSA-N 0.000 description 1
- CADHFEQRLJUKDK-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(2-methyl-6,7-dihydro-4h-[1,3]oxazolo[4,5-c]pyridine-5-carbonyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1CC=2OC(C)=NC=2CN1C(=O)C(N=1)=CC=CC=1NC(=C(C=1)C(N)=O)SC=1C1=CC=C(C(C)(C)O)C=C1F CADHFEQRLJUKDK-UHFFFAOYSA-N 0.000 description 1
- RBNOUIYZJXMLLC-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(methoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 RBNOUIYZJXMLLC-UHFFFAOYSA-N 0.000 description 1
- CTMBKEGFBBTFRB-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCOCC2)=N1 CTMBKEGFBBTFRB-UHFFFAOYSA-N 0.000 description 1
- CILYWAFWXHONKL-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-(pyrazin-2-ylmethoxymethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2N=CC=NC=2)=N1 CILYWAFWXHONKL-UHFFFAOYSA-N 0.000 description 1
- LDGHYVAJXLNVAJ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-morpholin-4-yl-2-oxoethoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC(=O)N2CCOCC2)=N1 LDGHYVAJXLNVAJ-UHFFFAOYSA-N 0.000 description 1
- KZVJNVGKVGBMLQ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(2-oxopyrrolidin-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2C(CCC2)=O)=N1 KZVJNVGKVGBMLQ-UHFFFAOYSA-N 0.000 description 1
- DWJNHHZJJABADG-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-fluoropyridin-2-yl)methoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2C(=CC=CN=2)F)=N1 DWJNHHZJJABADG-UHFFFAOYSA-N 0.000 description 1
- MQAOESPDWOQNIP-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-methyl-1,2,4-oxadiazol-5-yl)methoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC1=NOC(COCC=2N=C(NC3=C(C=C(S3)C=3C(=CC(=CC=3)C(C)(C)O)F)C(N)=O)C=CC=2)=N1 MQAOESPDWOQNIP-UHFFFAOYSA-N 0.000 description 1
- QQBDANOBQVDRDS-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3-methyl-1,2-oxazol-5-yl)methoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound O1N=C(C)C=C1COCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 QQBDANOBQVDRDS-UHFFFAOYSA-N 0.000 description 1
- GNPLMWNNJINNDK-HNNXBMFYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(3s)-3-methoxypyrrolidine-1-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound C1[C@@H](OC)CCN1C(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 GNPLMWNNJINNDK-HNNXBMFYSA-N 0.000 description 1
- HXCPPKCULYWSTK-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-fluorophenyl)methoxymethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(COCC=2C=CC(F)=CC=2)=N1 HXCPPKCULYWSTK-UHFFFAOYSA-N 0.000 description 1
- IHECRQFUVOMEGM-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[(4-methyl-4-oxo-1,4$l^{5}-azaphosphinan-1-yl)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCP(C)(=O)CC2)=N1 IHECRQFUVOMEGM-UHFFFAOYSA-N 0.000 description 1
- NTJFUHMGBSKANI-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-(hydroxymethyl)morpholine-4-carbonyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)N2CC(CO)OCC2)=N1 NTJFUHMGBSKANI-UHFFFAOYSA-N 0.000 description 1
- QCQCQVRTULMGRP-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[2-hydroxy-1-(2-oxo-1,3-oxazolidin-3-yl)ethyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(CO)N2C(OCC2)=O)=N1 QCQCQVRTULMGRP-UHFFFAOYSA-N 0.000 description 1
- UCMGYNWKZVPYAH-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(2-hydroxy-2-methylpropyl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)CNCC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 UCMGYNWKZVPYAH-UHFFFAOYSA-N 0.000 description 1
- NGWMODGLXKGOBQ-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[(2-oxopyrrolidin-3-yl)amino]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CNC2C(NCC2)=O)=N1 NGWMODGLXKGOBQ-UHFFFAOYSA-N 0.000 description 1
- SWVIGYJLHVIZFI-UHFFFAOYSA-N 5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]-2-[[6-[[methoxy(methyl)phosphoryl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound COP(C)(=O)CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 SWVIGYJLHVIZFI-UHFFFAOYSA-N 0.000 description 1
- NAKPGUKOMXENAA-UHFFFAOYSA-N 5-[2-fluoro-4-(2-methoxypropan-2-yl)phenyl]-2-[[5-(1-methoxyethyl)-6-methylpyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=C(C)C(C(C)OC)=CC=C1NC1=C(C(N)=O)C=C(C=2C(=CC(=CC=2)C(C)(C)OC)F)S1 NAKPGUKOMXENAA-UHFFFAOYSA-N 0.000 description 1
- BIJVLUBPQALTAD-UHFFFAOYSA-N 5-[4-(1,1-dioxo-1,4-thiazinan-4-yl)phenyl]-2-[[6-[[4-(2-hydroxypropan-2-yl)triazol-1-yl]methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound N1=NC(C(C)(O)C)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)N2CCS(=O)(=O)CC2)C(N)=O)=N1 BIJVLUBPQALTAD-UHFFFAOYSA-N 0.000 description 1
- MTIJLDREPVBBEB-UHFFFAOYSA-N 5-[4-(1-hydroxyethyl)phenyl]-2-nitrothiophene-3-carboxamide Chemical compound C1=CC(C(O)C)=CC=C1C1=CC(C(N)=O)=C([N+]([O-])=O)S1 MTIJLDREPVBBEB-UHFFFAOYSA-N 0.000 description 1
- MCHJPUPQGBNHTP-UHFFFAOYSA-N 5-[4-(2-cyanopropan-2-yl)phenyl]-2-[[6-[(2-hydroxy-2-methylpropoxy)methyl]pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound CC(C)(O)COCC1=CC=CC(NC2=C(C=C(S2)C=2C=CC(=CC=2)C(C)(C)C#N)C(N)=O)=N1 MCHJPUPQGBNHTP-UHFFFAOYSA-N 0.000 description 1
- YCPWQRLIISVZHH-UHFFFAOYSA-N 5-[4-(3-fluorooxetan-3-yl)phenyl]-2-nitrothiophene-3-carboxamide Chemical compound S1C([N+]([O-])=O)=C(C(=O)N)C=C1C1=CC=C(C2(F)COC2)C=C1 YCPWQRLIISVZHH-UHFFFAOYSA-N 0.000 description 1
- FGPOJDQHRVAJKH-UHFFFAOYSA-N 5-[6-(2-cyanopropan-2-yl)pyridin-3-yl]-2-nitrothiophene-3-carboxamide Chemical compound C1=NC(C(C)(C#N)C)=CC=C1C1=CC(C(N)=O)=C([N+]([O-])=O)S1 FGPOJDQHRVAJKH-UHFFFAOYSA-N 0.000 description 1
- AMIXHZQKLMWDNO-UHFFFAOYSA-N 5-[6-(2-hydroxypropan-2-yl)pyridin-3-yl]-2-nitrothiophene-3-carboxamide Chemical compound C1=NC(C(C)(O)C)=CC=C1C1=CC(C(N)=O)=C([N+]([O-])=O)S1 AMIXHZQKLMWDNO-UHFFFAOYSA-N 0.000 description 1
- ZZCKGEKRGWARGR-UHFFFAOYSA-N 5-[6-(2-methylpyrazol-3-yl)pyridin-3-yl]-2-nitrothiophene-3-carboxamide Chemical compound CN1N=CC=C1C1=CC=C(C=2SC(=C(C(N)=O)C=2)[N+]([O-])=O)C=N1 ZZCKGEKRGWARGR-UHFFFAOYSA-N 0.000 description 1
- IETKPTYAGKZLKY-UHFFFAOYSA-N 5-[[4-[(3-methyl-4-oxoquinazolin-2-yl)methoxy]phenyl]methyl]-1,3-thiazolidine-2,4-dione Chemical compound N=1C2=CC=CC=C2C(=O)N(C)C=1COC(C=C1)=CC=C1CC1SC(=O)NC1=O IETKPTYAGKZLKY-UHFFFAOYSA-N 0.000 description 1
- 239000002677 5-alpha reductase inhibitor Substances 0.000 description 1
- XAUDJQYHKZQPEU-KVQBGUIXSA-N 5-aza-2'-deoxycytidine Chemical compound O=C1N=C(N)N=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 XAUDJQYHKZQPEU-KVQBGUIXSA-N 0.000 description 1
- GBOQUHPYCRYKGV-UHFFFAOYSA-N 5-nitro-2-(2-pyrrolidin-1-ylethyl)benzo[de]isoquinoline-1,3-dione Chemical compound O=C1C(C=23)=CC=CC3=CC([N+](=O)[O-])=CC=2C(=O)N1CCN1CCCC1 GBOQUHPYCRYKGV-UHFFFAOYSA-N 0.000 description 1
- FNJZWQUSMGXAFB-UHFFFAOYSA-N 5-phenyl-2-[[4-(trifluoromethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC(C(F)(F)F)=CC=N1 FNJZWQUSMGXAFB-UHFFFAOYSA-N 0.000 description 1
- KRUHDWCIWRXWRL-UHFFFAOYSA-N 5-phenyl-2-[[6-(trifluoromethyl)pyridin-2-yl]amino]thiophene-3-carboxamide Chemical compound NC(=O)C=1C=C(C=2C=CC=CC=2)SC=1NC1=CC=CC(C(F)(F)F)=N1 KRUHDWCIWRXWRL-UHFFFAOYSA-N 0.000 description 1
- JTDYUFSDZATMKU-UHFFFAOYSA-N 6-(1,3-dioxo-2-benzo[de]isoquinolinyl)-N-hydroxyhexanamide Chemical compound C1=CC(C(N(CCCCCC(=O)NO)C2=O)=O)=C3C2=CC=CC3=C1 JTDYUFSDZATMKU-UHFFFAOYSA-N 0.000 description 1
- DMEWPMJARYYCMJ-UHFFFAOYSA-N 6-(morpholin-4-ylmethyl)-n-[5-[4-(2h-triazol-4-yl)phenyl]thiophen-2-yl]pyridin-2-amine Chemical compound C=1C=CC(NC=2SC(=CC=2)C=2C=CC(=CC=2)C2=NNN=C2)=NC=1CN1CCOCC1 DMEWPMJARYYCMJ-UHFFFAOYSA-N 0.000 description 1
- ZWAVODRZPFSYHB-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-hydroxy-2-methylpropyl)pyridine-2-carboxamide Chemical compound CC(C)(O)CNC(=O)C1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 ZWAVODRZPFSYHB-UHFFFAOYSA-N 0.000 description 1
- SUWZFNNMECRVCZ-UHFFFAOYSA-N 6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]-n-(2-hydroxyethyl)pyridine-2-carboxamide Chemical compound FC1=CC(C(C)(O)C)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(C(=O)NCCO)=N1 SUWZFNNMECRVCZ-UHFFFAOYSA-N 0.000 description 1
- GVZLCDXPQFPUQU-UHFFFAOYSA-N 6-[[5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridazine-3-carbonitrile Chemical compound FC1=CC(C(C)(O)C)=CC(F)=C1C(S1)=CC=C1NC1=CC=C(C#N)N=N1 GVZLCDXPQFPUQU-UHFFFAOYSA-N 0.000 description 1
- KAEVHZSIYLATMK-UHFFFAOYSA-N 6-n-[bis(aziridin-1-yl)phosphoryl]-2-n,2-n,7-trimethylpurine-2,6-diamine Chemical compound C=12N(C)C=NC2=NC(N(C)C)=NC=1NP(=O)(N1CC1)N1CC1 KAEVHZSIYLATMK-UHFFFAOYSA-N 0.000 description 1
- UNEPVPOHGXLUIR-UHFFFAOYSA-N 6317-37-9 Chemical compound OC(=O)C1=CC=C([N+]([O-])=O)S1 UNEPVPOHGXLUIR-UHFFFAOYSA-N 0.000 description 1
- KABRXLINDSPGDF-UHFFFAOYSA-N 7-bromoisoquinoline Chemical compound C1=CN=CC2=CC(Br)=CC=C21 KABRXLINDSPGDF-UHFFFAOYSA-N 0.000 description 1
- PBCZSGKMGDDXIJ-UHFFFAOYSA-N 7beta-hydroxystaurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3C(O)NC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(C)O1 PBCZSGKMGDDXIJ-UHFFFAOYSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- ZKHQWZAMYRWXGA-KQYNXXCUSA-J ATP(4-) Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O ZKHQWZAMYRWXGA-KQYNXXCUSA-J 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 235000006491 Acacia senegal Nutrition 0.000 description 1
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 description 1
- 208000014697 Acute lymphocytic leukaemia Diseases 0.000 description 1
- 229930024421 Adenine Natural products 0.000 description 1
- GFFGJBXGBJISGV-UHFFFAOYSA-N Adenine Chemical compound NC1=NC=NC2=C1N=CN2 GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 1
- 206010001233 Adenoma benign Diseases 0.000 description 1
- ZKHQWZAMYRWXGA-UHFFFAOYSA-N Adenosine triphosphate Natural products C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O ZKHQWZAMYRWXGA-UHFFFAOYSA-N 0.000 description 1
- 208000000884 Airway Obstruction Diseases 0.000 description 1
- 208000036065 Airway Remodeling Diseases 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- KLSJWNVTNUYHDU-UHFFFAOYSA-N Amitrole Chemical compound NC1=NC=NN1 KLSJWNVTNUYHDU-UHFFFAOYSA-N 0.000 description 1
- 102400000068 Angiostatin Human genes 0.000 description 1
- 108010079709 Angiostatins Proteins 0.000 description 1
- 229940123413 Angiotensin II antagonist Drugs 0.000 description 1
- 108020004491 Antisense DNA Proteins 0.000 description 1
- 108020000948 Antisense Oligonucleotides Proteins 0.000 description 1
- 108020005544 Antisense RNA Proteins 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 108010011485 Aspartame Proteins 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 206010003571 Astrocytoma Diseases 0.000 description 1
- XUKUURHRXDUEBC-KAYWLYCHSA-N Atorvastatin Chemical compound C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-KAYWLYCHSA-N 0.000 description 1
- XUKUURHRXDUEBC-UHFFFAOYSA-N Atorvastatin Natural products C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CCC(O)CC(O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-UHFFFAOYSA-N 0.000 description 1
- 229940123877 Aurora kinase inhibitor Drugs 0.000 description 1
- 102000003989 Aurora kinases Human genes 0.000 description 1
- 108090000433 Aurora kinases Proteins 0.000 description 1
- 208000010839 B-cell chronic lymphocytic leukemia Diseases 0.000 description 1
- KPYSYYIEGFHWSV-UHFFFAOYSA-N Baclofen Chemical compound OC(=O)CC(CN)C1=CC=C(Cl)C=C1 KPYSYYIEGFHWSV-UHFFFAOYSA-N 0.000 description 1
- 206010004146 Basal cell carcinoma Diseases 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 208000010392 Bone Fractures Diseases 0.000 description 1
- 208000006386 Bone Resorption Diseases 0.000 description 1
- 206010005949 Bone cancer Diseases 0.000 description 1
- 208000020084 Bone disease Diseases 0.000 description 1
- 206010073106 Bone giant cell tumour malignant Diseases 0.000 description 1
- 206010065687 Bone loss Diseases 0.000 description 1
- 208000018084 Bone neoplasm Diseases 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- VOVIALXJUBGFJZ-KWVAZRHASA-N Budesonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(CCC)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O VOVIALXJUBGFJZ-KWVAZRHASA-N 0.000 description 1
- 108010082830 CEP 2563 Proteins 0.000 description 1
- 101100326430 Caenorhabditis elegans bub-1 gene Proteins 0.000 description 1
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- 208000009458 Carcinoma in Situ Diseases 0.000 description 1
- AOCCBINRVIKJHY-UHFFFAOYSA-N Carmofur Chemical compound CCCCCCNC(=O)N1C=C(F)C(=O)NC1=O AOCCBINRVIKJHY-UHFFFAOYSA-N 0.000 description 1
- 108010078791 Carrier Proteins Proteins 0.000 description 1
- 102000005600 Cathepsins Human genes 0.000 description 1
- 108010084457 Cathepsins Proteins 0.000 description 1
- 102000000844 Cell Surface Receptors Human genes 0.000 description 1
- 108010001857 Cell Surface Receptors Proteins 0.000 description 1
- 102100025832 Centromere-associated protein E Human genes 0.000 description 1
- 206010008263 Cervical dysplasia Diseases 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- 201000005262 Chondroma Diseases 0.000 description 1
- 208000005243 Chondrosarcoma Diseases 0.000 description 1
- 201000009047 Chordoma Diseases 0.000 description 1
- 208000006332 Choriocarcinoma Diseases 0.000 description 1
- 208000005590 Choroidal Neovascularization Diseases 0.000 description 1
- 206010060823 Choroidal neovascularisation Diseases 0.000 description 1
- 102100031162 Collagen alpha-1(XVIII) chain Human genes 0.000 description 1
- 206010048832 Colon adenoma Diseases 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 206010055114 Colon cancer metastatic Diseases 0.000 description 1
- HVXBOLULGPECHP-WAYWQWQTSA-N Combretastatin A4 Chemical compound C1=C(O)C(OC)=CC=C1\C=C/C1=CC(OC)=C(OC)C(OC)=C1 HVXBOLULGPECHP-WAYWQWQTSA-N 0.000 description 1
- 206010010356 Congenital anomaly Diseases 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 229930188224 Cryptophycin Natural products 0.000 description 1
- 102100021906 Cyclin-O Human genes 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 230000005778 DNA damage Effects 0.000 description 1
- 231100000277 DNA damage Toxicity 0.000 description 1
- 239000012623 DNA damaging agent Substances 0.000 description 1
- 101000582926 Dictyostelium discoideum Probable serine/threonine-protein kinase PLK Proteins 0.000 description 1
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 1
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 1
- LQKSHSFQQRCAFW-UHFFFAOYSA-N Dolastatin 15 Natural products COC1=CC(=O)N(C(=O)C(OC(=O)C2N(CCC2)C(=O)C2N(CCC2)C(=O)C(C(C)C)N(C)C(=O)C(NC(=O)C(C(C)C)N(C)C)C(C)C)C(C)C)C1CC1=CC=CC=C1 LQKSHSFQQRCAFW-UHFFFAOYSA-N 0.000 description 1
- CYQFCXCEBYINGO-DLBZAZTESA-N Dronabinol Natural products C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@H]21 CYQFCXCEBYINGO-DLBZAZTESA-N 0.000 description 1
- 208000007033 Dysgerminoma Diseases 0.000 description 1
- 208000000471 Dysplastic Nevus Syndrome Diseases 0.000 description 1
- 201000009051 Embryonal Carcinoma Diseases 0.000 description 1
- 206010014733 Endometrial cancer Diseases 0.000 description 1
- 206010014759 Endometrial neoplasm Diseases 0.000 description 1
- 108010079505 Endostatins Proteins 0.000 description 1
- SAMRUMKYXPVKPA-VFKOLLTISA-N Enocitabine Chemical compound O=C1N=C(NC(=O)CCCCCCCCCCCCCCCCCCCCC)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 SAMRUMKYXPVKPA-VFKOLLTISA-N 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 206010014967 Ependymoma Diseases 0.000 description 1
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 description 1
- OTMSDBZUPAUEDD-UHFFFAOYSA-N Ethane Chemical compound CC OTMSDBZUPAUEDD-UHFFFAOYSA-N 0.000 description 1
- 239000001856 Ethyl cellulose Substances 0.000 description 1
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- DBVJJBKOTRCVKF-UHFFFAOYSA-N Etidronic acid Chemical compound OP(=O)(O)C(O)(C)P(O)(O)=O DBVJJBKOTRCVKF-UHFFFAOYSA-N 0.000 description 1
- 208000006168 Ewing Sarcoma Diseases 0.000 description 1
- RSCIYYHIBVZXDI-UHFFFAOYSA-O Fagaridine Chemical compound C1=C2OCOC2=CC2=CC=C3C4=CC=C(OC)C(O)=C4C=[N+](C)C3=C21 RSCIYYHIBVZXDI-UHFFFAOYSA-O 0.000 description 1
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 208000007659 Fibroadenoma Diseases 0.000 description 1
- 102000003972 Fibroblast growth factor 7 Human genes 0.000 description 1
- 108090000385 Fibroblast growth factor 7 Proteins 0.000 description 1
- 206010053717 Fibrous histiocytoma Diseases 0.000 description 1
- PLDUPXSUYLZYBN-UHFFFAOYSA-N Fluphenazine Chemical compound C1CN(CCO)CCN1CCCN1C2=CC(C(F)(F)F)=CC=C2SC2=CC=CC=C21 PLDUPXSUYLZYBN-UHFFFAOYSA-N 0.000 description 1
- 102000003688 G-Protein-Coupled Receptors Human genes 0.000 description 1
- 108090000045 G-Protein-Coupled Receptors Proteins 0.000 description 1
- GGUVRMBIEPYOKL-WMVCGJOFSA-N GW 409544 Chemical compound C([C@H](NC(/C)=C\C(=O)C=1C=CC=CC=1)C(O)=O)C(C=C1)=CC=C1OCCC(=C(O1)C)N=C1C1=CC=CC=C1 GGUVRMBIEPYOKL-WMVCGJOFSA-N 0.000 description 1
- HEMJJKBWTPKOJG-UHFFFAOYSA-N Gemfibrozil Chemical compound CC1=CC=C(C)C(OCCCC(C)(C)C(O)=O)=C1 HEMJJKBWTPKOJG-UHFFFAOYSA-N 0.000 description 1
- 208000000527 Germinoma Diseases 0.000 description 1
- 208000007569 Giant Cell Tumors Diseases 0.000 description 1
- 201000005409 Gliomatosis cerebri Diseases 0.000 description 1
- 206010018404 Glucagonoma Diseases 0.000 description 1
- 206010018691 Granuloma Diseases 0.000 description 1
- 229920000084 Gum arabic Polymers 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- 206010019629 Hepatic adenoma Diseases 0.000 description 1
- 208000017604 Hodgkin disease Diseases 0.000 description 1
- 208000010747 Hodgkins lymphoma Diseases 0.000 description 1
- 101000897441 Homo sapiens Cyclin-O Proteins 0.000 description 1
- 101000746367 Homo sapiens Granulocyte colony-stimulating factor Proteins 0.000 description 1
- 101001059454 Homo sapiens Serine/threonine-protein kinase MARK2 Proteins 0.000 description 1
- 101000851007 Homo sapiens Vascular endothelial growth factor receptor 2 Proteins 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- HEFNNWSXXWATRW-UHFFFAOYSA-N Ibuprofen Chemical compound CC(C)CC1=CC=C(C(C)C(O)=O)C=C1 HEFNNWSXXWATRW-UHFFFAOYSA-N 0.000 description 1
- JJKOTMDDZAJTGQ-DQSJHHFOSA-N Idoxifene Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN2CCCC2)=CC=1)/C1=CC=C(I)C=C1 JJKOTMDDZAJTGQ-DQSJHHFOSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 208000005045 Interdigitating dendritic cell sarcoma Diseases 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 102000008070 Interferon-gamma Human genes 0.000 description 1
- 102000018682 Interleukin Receptor Common gamma Subunit Human genes 0.000 description 1
- 108010066719 Interleukin Receptor Common gamma Subunit Proteins 0.000 description 1
- 102100030694 Interleukin-11 Human genes 0.000 description 1
- 108010065805 Interleukin-12 Proteins 0.000 description 1
- 102000013462 Interleukin-12 Human genes 0.000 description 1
- 108090000172 Interleukin-15 Proteins 0.000 description 1
- 108010002386 Interleukin-3 Proteins 0.000 description 1
- 102100039064 Interleukin-3 Human genes 0.000 description 1
- 108010002616 Interleukin-5 Proteins 0.000 description 1
- 108090001005 Interleukin-6 Proteins 0.000 description 1
- 108010002586 Interleukin-7 Proteins 0.000 description 1
- 108010002335 Interleukin-9 Proteins 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 229910021640 Iridium dichloride Inorganic materials 0.000 description 1
- LPHGQDQBBGAPDZ-UHFFFAOYSA-N Isocaffeine Natural products CN1C(=O)N(C)C(=O)C2=C1N(C)C=N2 LPHGQDQBBGAPDZ-UHFFFAOYSA-N 0.000 description 1
- SHGAZHPCJJPHSC-NUEINMDLSA-N Isotretinoin Chemical compound OC(=O)C=C(C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-NUEINMDLSA-N 0.000 description 1
- 229940121730 Janus kinase 2 inhibitor Drugs 0.000 description 1
- 206010072206 Janus kinase 2 mutation Diseases 0.000 description 1
- 208000007766 Kaposi sarcoma Diseases 0.000 description 1
- 208000002260 Keloid Diseases 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- 102100023424 Kinesin-like protein KIF2C Human genes 0.000 description 1
- 101710134369 Kinesin-like protein KIF2C Proteins 0.000 description 1
- MLFKVJCWGUZWNV-UHFFFAOYSA-N L-alanosine Natural products OC(=O)C(N)CN(O)N=O MLFKVJCWGUZWNV-UHFFFAOYSA-N 0.000 description 1
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 1
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 1
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 1
- 108010043135 L-methionine gamma-lyase Proteins 0.000 description 1
- 239000005517 L01XE01 - Imatinib Substances 0.000 description 1
- 239000005411 L01XE02 - Gefitinib Substances 0.000 description 1
- 239000002147 L01XE04 - Sunitinib Substances 0.000 description 1
- DAQAKHDKYAWHCG-UHFFFAOYSA-N Lactacystin Natural products CC(=O)NC(C(O)=O)CSC(=O)C1(C(O)C(C)C)NC(=O)C(C)C1O DAQAKHDKYAWHCG-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 108010000817 Leuprolide Proteins 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 208000002404 Liver Cell Adenoma Diseases 0.000 description 1
- 208000031422 Lymphocytic Chronic B-Cell Leukemia Diseases 0.000 description 1
- 102000004083 Lymphotoxin-alpha Human genes 0.000 description 1
- 108090000542 Lymphotoxin-alpha Proteins 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 208000006644 Malignant Fibrous Histiocytoma Diseases 0.000 description 1
- 208000000172 Medulloblastoma Diseases 0.000 description 1
- 108010047230 Member 1 Subfamily B ATP Binding Cassette Transporter Proteins 0.000 description 1
- 206010027406 Mesothelioma Diseases 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- 208000037848 Metastatic bone disease Diseases 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 101100335081 Mus musculus Flt3 gene Proteins 0.000 description 1
- 201000003793 Myelodysplastic syndrome Diseases 0.000 description 1
- 201000007224 Myeloproliferative neoplasm Diseases 0.000 description 1
- OUSFTKFNBAZUKL-UHFFFAOYSA-N N-(5-{[(5-tert-butyl-1,3-oxazol-2-yl)methyl]sulfanyl}-1,3-thiazol-2-yl)piperidine-4-carboxamide Chemical compound O1C(C(C)(C)C)=CN=C1CSC(S1)=CN=C1NC(=O)C1CCNCC1 OUSFTKFNBAZUKL-UHFFFAOYSA-N 0.000 description 1
- UEEJHVSXFDXPFK-UHFFFAOYSA-N N-dimethylaminoethanol Chemical compound CN(C)CCO UEEJHVSXFDXPFK-UHFFFAOYSA-N 0.000 description 1
- HTLZVHNRZJPSMI-UHFFFAOYSA-N N-ethylpiperidine Chemical compound CCN1CCCCC1 HTLZVHNRZJPSMI-UHFFFAOYSA-N 0.000 description 1
- MBBZMMPHUWSWHV-BDVNFPICSA-N N-methylglucamine Chemical compound CNC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO MBBZMMPHUWSWHV-BDVNFPICSA-N 0.000 description 1
- LFTLOKWAGJYHHR-UHFFFAOYSA-N N-methylmorpholine N-oxide Chemical compound CN1(=O)CCOCC1 LFTLOKWAGJYHHR-UHFFFAOYSA-N 0.000 description 1
- FTFRZXFNZVCRSK-UHFFFAOYSA-N N4-(3-chloro-4-fluorophenyl)-N6-(1-methyl-4-piperidinyl)pyrimido[5,4-d]pyrimidine-4,6-diamine Chemical compound C1CN(C)CCC1NC1=NC=C(N=CN=C2NC=3C=C(Cl)C(F)=CC=3)C2=N1 FTFRZXFNZVCRSK-UHFFFAOYSA-N 0.000 description 1
- 239000007832 Na2SO4 Substances 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010061309 Neoplasm progression Diseases 0.000 description 1
- 206010029113 Neovascularisation Diseases 0.000 description 1
- 206010029260 Neuroblastoma Diseases 0.000 description 1
- 201000004404 Neurofibroma Diseases 0.000 description 1
- 108010070047 Notch Receptors Proteins 0.000 description 1
- 102000005650 Notch Receptors Human genes 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- QSSYUVGWWQBOBQ-YXSASFKJSA-N OC144-093 Chemical compound CCO/C=C\CC(C=C1)=CC=C1C1=NC(C(C=C2)=CC=C2NC(C)C)=C(C(C=C2)=CC=C2NC(C)C)N1 QSSYUVGWWQBOBQ-YXSASFKJSA-N 0.000 description 1
- 208000008589 Obesity Diseases 0.000 description 1
- REYJJPSVUYRZGE-UHFFFAOYSA-N Octadecylamine Chemical compound CCCCCCCCCCCCCCCCCCN REYJJPSVUYRZGE-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 201000010133 Oligodendroglioma Diseases 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- 241000906034 Orthops Species 0.000 description 1
- 208000010191 Osteitis Deformans Diseases 0.000 description 1
- 208000000035 Osteochondroma Diseases 0.000 description 1
- 208000001132 Osteoporosis Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 239000012661 PARP inhibitor Substances 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 101150023417 PPARG gene Proteins 0.000 description 1
- 208000027067 Paget disease of bone Diseases 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- 108010067035 Pancrelipase Proteins 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 241000157426 Pernis Species 0.000 description 1
- 229940122054 Peroxisome proliferator-activated receptor delta agonist Drugs 0.000 description 1
- 229940080774 Peroxisome proliferator-activated receptor gamma agonist Drugs 0.000 description 1
- 208000007641 Pinealoma Diseases 0.000 description 1
- KMSKQZKKOZQFFG-HSUXVGOQSA-N Pirarubicin Chemical compound O([C@H]1[C@@H](N)C[C@@H](O[C@H]1C)O[C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1CCCCO1 KMSKQZKKOZQFFG-HSUXVGOQSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229940121906 Poly ADP ribose polymerase inhibitor Drugs 0.000 description 1
- 229920001710 Polyorthoester Polymers 0.000 description 1
- HRHKSTOGXBBQCB-UHFFFAOYSA-N Porfiromycine Chemical compound O=C1C(N)=C(C)C(=O)C2=C1C(COC(N)=O)C1(OC)C3N(C)C3CN12 HRHKSTOGXBBQCB-UHFFFAOYSA-N 0.000 description 1
- TUZYXOIXSAXUGO-UHFFFAOYSA-N Pravastatin Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(O)C=C21 TUZYXOIXSAXUGO-UHFFFAOYSA-N 0.000 description 1
- 208000006664 Precursor Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 description 1
- HFVNWDWLWUCIHC-GUPDPFMOSA-N Prednimustine Chemical compound O=C([C@@]1(O)CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)[C@@H](O)C[C@@]21C)COC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 HFVNWDWLWUCIHC-GUPDPFMOSA-N 0.000 description 1
- 239000004146 Propane-1,2-diol Substances 0.000 description 1
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 description 1
- 102000001253 Protein Kinase Human genes 0.000 description 1
- 102000016971 Proto-Oncogene Proteins c-kit Human genes 0.000 description 1
- 108010014608 Proto-Oncogene Proteins c-kit Proteins 0.000 description 1
- AHHFEZNOXOZZQA-ZEBDFXRSSA-N Ranimustine Chemical compound CO[C@H]1O[C@H](CNC(=O)N(CCCl)N=O)[C@@H](O)[C@H](O)[C@H]1O AHHFEZNOXOZZQA-ZEBDFXRSSA-N 0.000 description 1
- 108090000873 Receptor Protein-Tyrosine Kinases Proteins 0.000 description 1
- 102000004278 Receptor Protein-Tyrosine Kinases Human genes 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 208000006265 Renal cell carcinoma Diseases 0.000 description 1
- 208000002200 Respiratory Hypersensitivity Diseases 0.000 description 1
- 208000007135 Retinal Neovascularization Diseases 0.000 description 1
- 208000017442 Retinal disease Diseases 0.000 description 1
- 201000000582 Retinoblastoma Diseases 0.000 description 1
- 206010038923 Retinopathy Diseases 0.000 description 1
- 206010038933 Retinopathy of prematurity Diseases 0.000 description 1
- 208000005678 Rhabdomyoma Diseases 0.000 description 1
- OWPCHSCAPHNHAV-UHFFFAOYSA-N Rhizoxin Natural products C1C(O)C2(C)OC2C=CC(C)C(OC(=O)C2)CC2CC2OC2C(=O)OC1C(C)C(OC)C(C)=CC=CC(C)=CC1=COC(C)=N1 OWPCHSCAPHNHAV-UHFFFAOYSA-N 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- RYMZZMVNJRMUDD-UHFFFAOYSA-N SJ000286063 Natural products C12C(OC(=O)C(C)(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 RYMZZMVNJRMUDD-UHFFFAOYSA-N 0.000 description 1
- 108010011005 STAT6 Transcription Factor Proteins 0.000 description 1
- 229940124639 Selective inhibitor Drugs 0.000 description 1
- 201000010208 Seminoma Diseases 0.000 description 1
- 102100028904 Serine/threonine-protein kinase MARK2 Human genes 0.000 description 1
- 208000000097 Sertoli-Leydig cell tumor Diseases 0.000 description 1
- 102100023980 Signal transducer and activator of transcription 6 Human genes 0.000 description 1
- OCOKWVBYZHBHLU-UHFFFAOYSA-N Sobuzoxane Chemical compound C1C(=O)N(COC(=O)OCC(C)C)C(=O)CN1CCN1CC(=O)N(COC(=O)OCC(C)C)C(=O)C1 OCOKWVBYZHBHLU-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 230000006044 T cell activation Effects 0.000 description 1
- CYQFCXCEBYINGO-UHFFFAOYSA-N THC Natural products C1=C(C)CCC2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3C21 CYQFCXCEBYINGO-UHFFFAOYSA-N 0.000 description 1
- 108010065917 TOR Serine-Threonine Kinases Proteins 0.000 description 1
- 102000013530 TOR Serine-Threonine Kinases Human genes 0.000 description 1
- LGGHDPFKSSRQNS-UHFFFAOYSA-N Tariquidar Chemical compound C1=CC=CC2=CC(C(=O)NC3=CC(OC)=C(OC)C=C3C(=O)NC3=CC=C(C=C3)CCN3CCC=4C=C(C(=CC=4C3)OC)OC)=CN=C21 LGGHDPFKSSRQNS-UHFFFAOYSA-N 0.000 description 1
- 229940123464 Thiazolidinedione Drugs 0.000 description 1
- KLBQZWRITKRQQV-UHFFFAOYSA-N Thioridazine Chemical compound C12=CC(SC)=CC=C2SC2=CC=CC=C2N1CCC1CCCCN1C KLBQZWRITKRQQV-UHFFFAOYSA-N 0.000 description 1
- 208000007536 Thrombosis Diseases 0.000 description 1
- 108010078233 Thymalfasin Proteins 0.000 description 1
- DKJJVAGXPKPDRL-UHFFFAOYSA-N Tiludronic acid Chemical compound OP(O)(=O)C(P(O)(O)=O)SC1=CC=C(Cl)C=C1 DKJJVAGXPKPDRL-UHFFFAOYSA-N 0.000 description 1
- IVTVGDXNLFLDRM-HNNXBMFYSA-N Tomudex Chemical compound C=1C=C2NC(C)=NC(=O)C2=CC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)S1 IVTVGDXNLFLDRM-HNNXBMFYSA-N 0.000 description 1
- IWEQQRMGNVVKQW-OQKDUQJOSA-N Toremifene citrate Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O.C1=CC(OCCN(C)C)=CC=C1C(\C=1C=CC=CC=1)=C(\CCCl)C1=CC=CC=C1 IWEQQRMGNVVKQW-OQKDUQJOSA-N 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 1
- 102000044209 Tumor Suppressor Genes Human genes 0.000 description 1
- 108700025716 Tumor Suppressor Genes Proteins 0.000 description 1
- 102000001742 Tumor Suppressor Proteins Human genes 0.000 description 1
- 108010040002 Tumor Suppressor Proteins Proteins 0.000 description 1
- 229940127507 Ubiquitin Ligase Inhibitors Drugs 0.000 description 1
- 208000015778 Undifferentiated pleomorphic sarcoma Diseases 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 208000009311 VIPoma Diseases 0.000 description 1
- 108010053099 Vascular Endothelial Growth Factor Receptor-2 Proteins 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 206010048214 Xanthoma Diseases 0.000 description 1
- 206010048215 Xanthomatosis Diseases 0.000 description 1
- PCWZKQSKUXXDDJ-UHFFFAOYSA-N Xanthotoxin Natural products COCc1c2OC(=O)C=Cc2cc3ccoc13 PCWZKQSKUXXDDJ-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- WERKSKAQRVDLDW-ANOHMWSOSA-N [(2s,3r,4r,5r)-2,3,4,5,6-pentahydroxyhexyl] (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO WERKSKAQRVDLDW-ANOHMWSOSA-N 0.000 description 1
- XMYKNCNAZKMVQN-NYYWCZLTSA-N [(e)-(3-aminopyridin-2-yl)methylideneamino]thiourea Chemical compound NC(=S)N\N=C\C1=NC=CC=C1N XMYKNCNAZKMVQN-NYYWCZLTSA-N 0.000 description 1
- XSMVECZRZBFTIZ-UHFFFAOYSA-M [2-(aminomethyl)cyclobutyl]methanamine;2-oxidopropanoate;platinum(4+) Chemical compound [Pt+4].CC([O-])C([O-])=O.NCC1CCC1CN XSMVECZRZBFTIZ-UHFFFAOYSA-M 0.000 description 1
- CKXIPXAIFMTQCS-LRDUUELOSA-N [2-[(2s,4s)-4-[(2r,3r,4r,5s,6s)-3-fluoro-4,5-dihydroxy-6-methyloxan-2-yl]oxy-2,5,12-trihydroxy-7-methoxy-6,11-dioxo-3,4-dihydro-1h-tetracen-2-yl]-2-oxoethyl] 3-aminopropanoate Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)COC(=O)CCN)[C@@H]1O[C@@H](C)[C@@H](O)[C@@H](O)[C@H]1F CKXIPXAIFMTQCS-LRDUUELOSA-N 0.000 description 1
- JLCPHMBAVCMARE-UHFFFAOYSA-N [3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[3-[[3-[[3-[[3-[[3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-[[5-(2-amino-6-oxo-1H-purin-9-yl)-3-hydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(5-methyl-2,4-dioxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(6-aminopurin-9-yl)oxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-5-(4-amino-2-oxopyrimidin-1-yl)oxolan-2-yl]methyl [5-(6-aminopurin-9-yl)-2-(hydroxymethyl)oxolan-3-yl] hydrogen phosphate Polymers Cc1cn(C2CC(OP(O)(=O)OCC3OC(CC3OP(O)(=O)OCC3OC(CC3O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c3nc(N)[nH]c4=O)C(COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3COP(O)(=O)OC3CC(OC3CO)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3ccc(N)nc3=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cc(C)c(=O)[nH]c3=O)n3cc(C)c(=O)[nH]c3=O)n3ccc(N)nc3=O)n3cc(C)c(=O)[nH]c3=O)n3cnc4c3nc(N)[nH]c4=O)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)n3cnc4c(N)ncnc34)O2)c(=O)[nH]c1=O JLCPHMBAVCMARE-UHFFFAOYSA-N 0.000 description 1
- DJSLOQFVVUBAKG-UHFFFAOYSA-N [4-(3-chloroanilino)-5,6-dimethyl-7H-pyrrolo[2,3-d]pyrimidin-2-yl]methanesulfonic acid Chemical compound C=12C(C)=C(C)NC2=NC(CS(O)(=O)=O)=NC=1NC1=CC=CC(Cl)=C1 DJSLOQFVVUBAKG-UHFFFAOYSA-N 0.000 description 1
- JBNBEVRUHBIRLD-UHFFFAOYSA-N [4-[4-carbamoyl-5-[[2-(morpholin-4-ylmethyl)pyrimidin-4-yl]amino]thiophen-2-yl]triazol-1-yl]methyl 2,2-dimethylpropanoate Chemical compound N1=NN(COC(=O)C(C)(C)C)C=C1C(S1)=CC(C(N)=O)=C1NC1=CC=NC(CN2CCOCC2)=N1 JBNBEVRUHBIRLD-UHFFFAOYSA-N 0.000 description 1
- ZMOIZCNXIOSNKX-UHFFFAOYSA-N [4-[[3-carbamoyl-5-[2,6-difluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyrimidin-2-yl]methyl n-methylcarbamate Chemical compound CNC(=O)OCC1=NC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2F)C(C)(C)O)F)C(N)=O)=N1 ZMOIZCNXIOSNKX-UHFFFAOYSA-N 0.000 description 1
- JNYQEKQLWIEOSI-UHFFFAOYSA-N [6-(2-cyanopropan-2-yl)pyridin-3-yl]boronic acid Chemical compound N#CC(C)(C)C1=CC=C(B(O)O)C=N1 JNYQEKQLWIEOSI-UHFFFAOYSA-N 0.000 description 1
- SZPWXAOBLNYOHY-UHFFFAOYSA-N [C]1=CC=NC2=CC=CC=C12 Chemical group [C]1=CC=NC2=CC=CC=C12 SZPWXAOBLNYOHY-UHFFFAOYSA-N 0.000 description 1
- WXGSWQWQLHAEKI-UHFFFAOYSA-M [Cl-].[Li+].[Cu](C#N)C#N Chemical compound [Cl-].[Li+].[Cu](C#N)C#N WXGSWQWQLHAEKI-UHFFFAOYSA-M 0.000 description 1
- WLLIXJBWWFGEHT-UHFFFAOYSA-N [tert-butyl(dimethyl)silyl] trifluoromethanesulfonate Chemical compound CC(C)(C)[Si](C)(C)OS(=O)(=O)C(F)(F)F WLLIXJBWWFGEHT-UHFFFAOYSA-N 0.000 description 1
- 108010023617 abarelix Proteins 0.000 description 1
- 229960002184 abarelix Drugs 0.000 description 1
- 230000001594 aberrant effect Effects 0.000 description 1
- UVIQSJCZCSLXRZ-UBUQANBQSA-N abiraterone acetate Chemical compound C([C@@H]1[C@]2(C)CC[C@@H]3[C@@]4(C)CC[C@@H](CC4=CC[C@H]31)OC(=O)C)C=C2C1=CC=CN=C1 UVIQSJCZCSLXRZ-UBUQANBQSA-N 0.000 description 1
- 229960004103 abiraterone acetate Drugs 0.000 description 1
- 229940028652 abraxane Drugs 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 235000010489 acacia gum Nutrition 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 229960001138 acetylsalicylic acid Drugs 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000013543 active substance Substances 0.000 description 1
- 229940037127 actonel Drugs 0.000 description 1
- 229940060205 adagen Drugs 0.000 description 1
- 229960000643 adenine Drugs 0.000 description 1
- 208000002718 adenomatoid tumor Diseases 0.000 description 1
- 210000004100 adrenal gland Anatomy 0.000 description 1
- 229940009456 adriamycin Drugs 0.000 description 1
- 229940064305 adrucil Drugs 0.000 description 1
- 230000010085 airway hyperresponsiveness Effects 0.000 description 1
- 208000037883 airway inflammation Diseases 0.000 description 1
- 229950005033 alanosine Drugs 0.000 description 1
- DCSBSVSZJRSITC-UHFFFAOYSA-M alendronate sodium trihydrate Chemical compound O.O.O.[Na+].NCCCC(O)(P(O)(O)=O)P(O)([O-])=O DCSBSVSZJRSITC-UHFFFAOYSA-M 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 229940110282 alimta Drugs 0.000 description 1
- 229940098174 alkeran Drugs 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 230000007815 allergy Effects 0.000 description 1
- 229960003459 allopurinol Drugs 0.000 description 1
- 229940087168 alpha tocopherol Drugs 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 229950010817 alvocidib Drugs 0.000 description 1
- BIIVYFLTOXDAOV-YVEFUNNKSA-N alvocidib Chemical compound O[C@@H]1CN(C)CC[C@@H]1C1=C(O)C=C(O)C2=C1OC(C=1C(=CC=CC=1)Cl)=CC2=O BIIVYFLTOXDAOV-YVEFUNNKSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 229960001097 amifostine Drugs 0.000 description 1
- 229960003896 aminopterin Drugs 0.000 description 1
- 229960002550 amrubicin Drugs 0.000 description 1
- VJZITPJGSQKZMX-XDPRQOKASA-N amrubicin Chemical compound O([C@H]1C[C@](CC2=C(O)C=3C(=O)C4=CC=CC=C4C(=O)C=3C(O)=C21)(N)C(=O)C)[C@H]1C[C@H](O)[C@H](O)CO1 VJZITPJGSQKZMX-XDPRQOKASA-N 0.000 description 1
- 239000003098 androgen Substances 0.000 description 1
- 229940030486 androgens Drugs 0.000 description 1
- 230000006427 angiogenic response Effects 0.000 description 1
- 239000002333 angiotensin II receptor antagonist Substances 0.000 description 1
- 229950001104 anhydrovinblastine Drugs 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- CIDNKDMVSINJCG-GKXONYSUSA-N annamycin Chemical compound I[C@@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(=O)CO)C1 CIDNKDMVSINJCG-GKXONYSUSA-N 0.000 description 1
- 230000001093 anti-cancer Effects 0.000 description 1
- 230000000454 anti-cipatory effect Effects 0.000 description 1
- 230000001090 anti-dopaminergic effect Effects 0.000 description 1
- 230000003388 anti-hormonal effect Effects 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 239000003816 antisense DNA Substances 0.000 description 1
- 239000000074 antisense oligonucleotide Substances 0.000 description 1
- 238000012230 antisense oligonucleotides Methods 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 229940115115 aranesp Drugs 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- 229940078010 arimidex Drugs 0.000 description 1
- 229940087620 aromasin Drugs 0.000 description 1
- 229940014583 arranon Drugs 0.000 description 1
- MCGDSOGUHLTADD-UHFFFAOYSA-N arzoxifene Chemical compound C1=CC(OC)=CC=C1C1=C(OC=2C=CC(OCCN3CCCCC3)=CC=2)C2=CC=C(O)C=C2S1 MCGDSOGUHLTADD-UHFFFAOYSA-N 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229960003272 asparaginase Drugs 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-M asparaginate Chemical compound [O-]C(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-M 0.000 description 1
- 239000000605 aspartame Substances 0.000 description 1
- IAOZJIPTCAWIRG-QWRGUYRKSA-N aspartame Chemical compound OC(=O)C[C@H](N)C(=O)N[C@H](C(=O)OC)CC1=CC=CC=C1 IAOZJIPTCAWIRG-QWRGUYRKSA-N 0.000 description 1
- 235000010357 aspartame Nutrition 0.000 description 1
- 229960003438 aspartame Drugs 0.000 description 1
- FZCSTZYAHCUGEM-UHFFFAOYSA-N aspergillomarasmine B Natural products OC(=O)CNC(C(O)=O)CNC(C(O)=O)CC(O)=O FZCSTZYAHCUGEM-UHFFFAOYSA-N 0.000 description 1
- TWHSQQYCDVSBRK-UHFFFAOYSA-N asulacrine Chemical compound C12=CC=CC(C)=C2N=C2C(C(=O)NC)=CC=CC2=C1NC1=CC=C(NS(C)(=O)=O)C=C1OC TWHSQQYCDVSBRK-UHFFFAOYSA-N 0.000 description 1
- 229950011088 asulacrine Drugs 0.000 description 1
- 229960005370 atorvastatin Drugs 0.000 description 1
- SHZPNDRIDUBNMH-NIJVSVLQSA-L atorvastatin calcium trihydrate Chemical compound O.O.O.[Ca+2].C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC([O-])=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1.C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC([O-])=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 SHZPNDRIDUBNMH-NIJVSVLQSA-L 0.000 description 1
- 108010044540 auristatin Proteins 0.000 description 1
- 230000035578 autophosphorylation Effects 0.000 description 1
- 229940120638 avastin Drugs 0.000 description 1
- 229960002756 azacitidine Drugs 0.000 description 1
- KLNFSAOEKUDMFA-UHFFFAOYSA-N azanide;2-hydroxyacetic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OCC(O)=O KLNFSAOEKUDMFA-UHFFFAOYSA-N 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 229960000794 baclofen Drugs 0.000 description 1
- 150000007514 bases Chemical class 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 208000001119 benign fibrous histiocytoma Diseases 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 125000004601 benzofurazanyl group Chemical group N1=C2C(=NO1)C(=CC=C2)* 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid group Chemical group C(C1=CC=CC=C1)(=O)O WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- PKCJANJLZJAAEE-UHFFFAOYSA-N benzyl n-(3-carbamoylthiophen-2-yl)carbamate Chemical compound C1=CSC(NC(=O)OCC=2C=CC=CC=2)=C1C(=O)N PKCJANJLZJAAEE-UHFFFAOYSA-N 0.000 description 1
- JOSYPSQWYUALQM-UHFFFAOYSA-N benzyl n-[3-carbamoyl-5-(2-fluorophenyl)thiophen-2-yl]carbamate Chemical compound NC(=O)C=1C=C(C=2C(=CC=CC=2)F)SC=1NC(=O)OCC1=CC=CC=C1 JOSYPSQWYUALQM-UHFFFAOYSA-N 0.000 description 1
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 1
- 229960003237 betaine Drugs 0.000 description 1
- 229960002537 betamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-DVTGEIKXSA-N betamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-DVTGEIKXSA-N 0.000 description 1
- 229940110331 bextra Drugs 0.000 description 1
- 229960000997 bicalutamide Drugs 0.000 description 1
- 229940108502 bicnu Drugs 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 229920002988 biodegradable polymer Polymers 0.000 description 1
- 239000004621 biodegradable polymer Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- YNHIGQDRGKUECZ-UHFFFAOYSA-L bis(triphenylphosphine)palladium(ii) dichloride Chemical compound [Cl-].[Cl-].[Pd+2].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 YNHIGQDRGKUECZ-UHFFFAOYSA-L 0.000 description 1
- 229950008548 bisantrene Drugs 0.000 description 1
- 201000001531 bladder carcinoma Diseases 0.000 description 1
- 229960001561 bleomycin Drugs 0.000 description 1
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- UBJAHGAUPNGZFF-XOVTVWCYSA-N bms-184476 Chemical compound O([C@H]1[C@@H]2[C@]3(OC(C)=O)CO[C@@H]3C[C@@H]([C@]2(C(=O)[C@H](OC(C)=O)C2=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)C=3C=CC=CC=3)C=3C=CC=CC=3)C[C@]1(O)C2(C)C)C)OCSC)C(=O)C1=CC=CC=C1 UBJAHGAUPNGZFF-XOVTVWCYSA-N 0.000 description 1
- GMJWGJSDPOAZTP-MIDYMNAOSA-N bms-188797 Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](OC(C)=O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)C=4C=CC=CC=4)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)OC)C(=O)C1=CC=CC=C1 GMJWGJSDPOAZTP-MIDYMNAOSA-N 0.000 description 1
- 208000016738 bone Paget disease Diseases 0.000 description 1
- 230000024279 bone resorption Effects 0.000 description 1
- 229940028101 boniva Drugs 0.000 description 1
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 1
- 229960001467 bortezomib Drugs 0.000 description 1
- 201000009480 botryoid rhabdomyosarcoma Diseases 0.000 description 1
- 201000008274 breast adenocarcinoma Diseases 0.000 description 1
- 201000008275 breast carcinoma Diseases 0.000 description 1
- 201000003149 breast fibroadenoma Diseases 0.000 description 1
- 125000001246 bromo group Chemical group Br* 0.000 description 1
- 208000003362 bronchogenic carcinoma Diseases 0.000 description 1
- 201000002143 bronchus adenoma Diseases 0.000 description 1
- 229940112133 busulfex Drugs 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000000480 butynyl group Chemical group [*]C#CC([H])([H])C([H])([H])[H] 0.000 description 1
- 229960001948 caffeine Drugs 0.000 description 1
- VJEONQKOZGKCAK-UHFFFAOYSA-N caffeine Natural products CN1C(=O)N(C)C(=O)C2=C1C=CN2C VJEONQKOZGKCAK-UHFFFAOYSA-N 0.000 description 1
- 229940112129 campath Drugs 0.000 description 1
- 229940088954 camptosar Drugs 0.000 description 1
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical compound C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 description 1
- 229940127093 camptothecin Drugs 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- 210000001043 capillary endothelial cell Anatomy 0.000 description 1
- 125000004623 carbolinyl group Chemical group 0.000 description 1
- 150000001721 carbon Chemical group 0.000 description 1
- 150000001722 carbon compounds Chemical class 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- WNRZHQBJSXRYJK-UHFFFAOYSA-N carboxyamidotriazole Chemical compound NC1=C(C(=O)N)N=NN1CC(C=C1Cl)=CC(Cl)=C1C(=O)C1=CC=C(Cl)C=C1 WNRZHQBJSXRYJK-UHFFFAOYSA-N 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 230000000747 cardiac effect Effects 0.000 description 1
- 229960003261 carmofur Drugs 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 108010046713 cemadotin Proteins 0.000 description 1
- 229950009017 cemadotin Drugs 0.000 description 1
- 108010031379 centromere protein E Proteins 0.000 description 1
- GPUADMRJQVPIAS-QCVDVZFFSA-M cerivastatin sodium Chemical compound [Na+].COCC1=C(C(C)C)N=C(C(C)C)C(\C=C\[C@@H](O)C[C@@H](O)CC([O-])=O)=C1C1=CC=C(F)C=C1 GPUADMRJQVPIAS-QCVDVZFFSA-M 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 208000019065 cervical carcinoma Diseases 0.000 description 1
- 210000003679 cervix uteri Anatomy 0.000 description 1
- 229960005395 cetuximab Drugs 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- IQCIQDNWBGEGRL-UHFFFAOYSA-N chembl1614651 Chemical compound O=C1C2=C(O)C=CC(O)=C2N2N=C(CNCCO)C3=CC=C(NCCCN)C1=C32 IQCIQDNWBGEGRL-UHFFFAOYSA-N 0.000 description 1
- YOQPCWIXYUNEET-UHFFFAOYSA-N chembl307697 Chemical compound C1=CC(O)=CC=C1C(C=1C=CC(O)=CC=1)=NNC1=CC=C([N+]([O-])=O)C=C1[N+]([O-])=O YOQPCWIXYUNEET-UHFFFAOYSA-N 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229960004630 chlorambucil Drugs 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 208000006990 cholangiocarcinoma Diseases 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 201000005217 chondroblastoma Diseases 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 208000032852 chronic lymphocytic leukemia Diseases 0.000 description 1
- 201000010902 chronic myelomonocytic leukemia Diseases 0.000 description 1
- 208000009060 clear cell adenocarcinoma Diseases 0.000 description 1
- ACSIXWWBWUQEHA-UHFFFAOYSA-N clodronic acid Chemical compound OP(O)(=O)C(Cl)(Cl)P(O)(O)=O ACSIXWWBWUQEHA-UHFFFAOYSA-N 0.000 description 1
- 229960002286 clodronic acid Drugs 0.000 description 1
- 229960000928 clofarabine Drugs 0.000 description 1
- 229960001214 clofibrate Drugs 0.000 description 1
- KNHUKKLJHYUCFP-UHFFFAOYSA-N clofibrate Chemical compound CCOC(=O)C(C)(C)OC1=CC=C(Cl)C=C1 KNHUKKLJHYUCFP-UHFFFAOYSA-N 0.000 description 1
- 229940103380 clolar Drugs 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- LGZKGOGODCLQHG-UHFFFAOYSA-N combretastatin Natural products C1=C(O)C(OC)=CC=C1CC(O)C1=CC(OC)=C(OC)C(OC)=C1 LGZKGOGODCLQHG-UHFFFAOYSA-N 0.000 description 1
- 229960005537 combretastatin A-4 Drugs 0.000 description 1
- HVXBOLULGPECHP-UHFFFAOYSA-N combretastatin A4 Natural products C1=C(O)C(OC)=CC=C1C=CC1=CC(OC)=C(OC)C(OC)=C1 HVXBOLULGPECHP-UHFFFAOYSA-N 0.000 description 1
- 239000003184 complementary RNA Substances 0.000 description 1
- 230000001010 compromised effect Effects 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910000366 copper(II) sulfate Inorganic materials 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 230000000875 corresponding effect Effects 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- POADTFBBIXOWFJ-VWLOTQADSA-N cositecan Chemical compound C1=CC=C2C(CC[Si](C)(C)C)=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 POADTFBBIXOWFJ-VWLOTQADSA-N 0.000 description 1
- 229940088547 cosmegen Drugs 0.000 description 1
- 229940111134 coxibs Drugs 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 239000010779 crude oil Substances 0.000 description 1
- 108010006226 cryptophycin Proteins 0.000 description 1
- PSNOPSMXOBPNNV-VVCTWANISA-N cryptophycin 1 Chemical compound C1=C(Cl)C(OC)=CC=C1C[C@@H]1C(=O)NC[C@@H](C)C(=O)O[C@@H](CC(C)C)C(=O)O[C@H]([C@H](C)[C@@H]2[C@H](O2)C=2C=CC=CC=2)C/C=C/C(=O)N1 PSNOPSMXOBPNNV-VVCTWANISA-N 0.000 description 1
- PSNOPSMXOBPNNV-UHFFFAOYSA-N cryptophycin-327 Natural products C1=C(Cl)C(OC)=CC=C1CC1C(=O)NCC(C)C(=O)OC(CC(C)C)C(=O)OC(C(C)C2C(O2)C=2C=CC=CC=2)CC=CC(=O)N1 PSNOPSMXOBPNNV-UHFFFAOYSA-N 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 201000010305 cutaneous fibrous histiocytoma Diseases 0.000 description 1
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- SNRCKKQHDUIRIY-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloromethane;dichloropalladium;iron(2+) Chemical compound [Fe+2].ClCCl.Cl[Pd]Cl.C1=C[CH-]C(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.C1=C[CH-]C(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 SNRCKKQHDUIRIY-UHFFFAOYSA-L 0.000 description 1
- 229950006614 cytarabine ocfosfate Drugs 0.000 description 1
- 102000003675 cytokine receptors Human genes 0.000 description 1
- 108010057085 cytokine receptors Proteins 0.000 description 1
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Natural products NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 230000001086 cytosolic effect Effects 0.000 description 1
- 229960003901 dacarbazine Drugs 0.000 description 1
- 229960005029 darbepoetin alfa Drugs 0.000 description 1
- 229940041983 daunorubicin liposomal Drugs 0.000 description 1
- 229940026692 decadron Drugs 0.000 description 1
- 229960003603 decitabine Drugs 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- CYQFCXCEBYINGO-IAGOWNOFSA-N delta1-THC Chemical compound C1=C(C)CC[C@H]2C(C)(C)OC3=CC(CCCCC)=CC(O)=C3[C@@H]21 CYQFCXCEBYINGO-IAGOWNOFSA-N 0.000 description 1
- 229960002923 denileukin diftitox Drugs 0.000 description 1
- CFCUWKMKBJTWLW-UHFFFAOYSA-N deoliosyl-3C-alpha-L-digitoxosyl-MTM Natural products CC=1C(O)=C2C(O)=C3C(=O)C(OC4OC(C)C(O)C(OC5OC(C)C(O)C(OC6OC(C)C(O)C(C)(O)C6)C5)C4)C(C(OC)C(=O)C(O)C(C)O)CC3=CC2=CC=1OC(OC(C)C1O)CC1OC1CC(O)C(O)C(C)O1 CFCUWKMKBJTWLW-UHFFFAOYSA-N 0.000 description 1
- 229940070968 depocyt Drugs 0.000 description 1
- 239000007933 dermal patch Substances 0.000 description 1
- 150000004985 diamines Chemical class 0.000 description 1
- 229950007457 dibrospidium chloride Drugs 0.000 description 1
- 125000006003 dichloroethyl group Chemical group 0.000 description 1
- HPNMFZURTQLUMO-UHFFFAOYSA-N diethylamine Chemical compound CCNCC HPNMFZURTQLUMO-UHFFFAOYSA-N 0.000 description 1
- LFQCJSBXBZRMTN-OAQYLSRUSA-N diflomotecan Chemical compound CC[C@@]1(O)CC(=O)OCC(C2=O)=C1C=C1N2CC2=CC3=CC(F)=C(F)C=C3N=C21 LFQCJSBXBZRMTN-OAQYLSRUSA-N 0.000 description 1
- 125000000723 dihydrobenzofuranyl group Chemical group O1C(CC2=C1C=CC=C2)* 0.000 description 1
- 125000005436 dihydrobenzothiophenyl group Chemical group S1C(CC2=C1C=CC=C2)* 0.000 description 1
- 125000005435 dihydrobenzoxazolyl group Chemical group O1C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000004852 dihydrofuranyl group Chemical group O1C(CC=C1)* 0.000 description 1
- 125000005047 dihydroimidazolyl group Chemical group N1(CNC=C1)* 0.000 description 1
- 125000001070 dihydroindolyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000005049 dihydrooxadiazolyl group Chemical group O1N(NC=C1)* 0.000 description 1
- 125000005050 dihydrooxazolyl group Chemical group O1C(NC=C1)* 0.000 description 1
- 125000005051 dihydropyrazinyl group Chemical group N1(CC=NC=C1)* 0.000 description 1
- 125000005052 dihydropyrazolyl group Chemical group N1(NCC=C1)* 0.000 description 1
- 125000004655 dihydropyridinyl group Chemical group N1(CC=CC=C1)* 0.000 description 1
- 125000005053 dihydropyrimidinyl group Chemical group N1(CN=CC=C1)* 0.000 description 1
- 125000005054 dihydropyrrolyl group Chemical group [H]C1=C([H])C([H])([H])C([H])([H])N1* 0.000 description 1
- 125000005044 dihydroquinolinyl group Chemical group N1(CC=CC2=CC=CC=C12)* 0.000 description 1
- 125000005056 dihydrothiazolyl group Chemical group S1C(NC=C1)* 0.000 description 1
- 125000005057 dihydrothienyl group Chemical group S1C(CC=C1)* 0.000 description 1
- 125000005058 dihydrotriazolyl group Chemical group N1(NNC=C1)* 0.000 description 1
- 229950009278 dimesna Drugs 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 150000002009 diols Chemical class 0.000 description 1
- YFMGHVQBAINRBB-UHFFFAOYSA-L disodium hydrogen carbonate chloride hydrate Chemical compound C([O-])(O)=O.[Na+].Cl.[OH-].[Na+] YFMGHVQBAINRBB-UHFFFAOYSA-L 0.000 description 1
- KQYGMURBTJPBPQ-UHFFFAOYSA-L disodium;2-(2-sulfonatoethyldisulfanyl)ethanesulfonate Chemical compound [Na+].[Na+].[O-]S(=O)(=O)CCSSCCS([O-])(=O)=O KQYGMURBTJPBPQ-UHFFFAOYSA-L 0.000 description 1
- NYDXNILOWQXUOF-UHFFFAOYSA-L disodium;2-[[4-[2-(2-amino-4-oxo-1,7-dihydropyrrolo[2,3-d]pyrimidin-5-yl)ethyl]benzoyl]amino]pentanedioate Chemical compound [Na+].[Na+].C=1NC=2NC(N)=NC(=O)C=2C=1CCC1=CC=C(C(=O)NC(CCC([O-])=O)C([O-])=O)C=C1 NYDXNILOWQXUOF-UHFFFAOYSA-L 0.000 description 1
- GRWZHXKQBITJKP-UHFFFAOYSA-L dithionite(2-) Chemical compound [O-]S(=O)S([O-])=O GRWZHXKQBITJKP-UHFFFAOYSA-L 0.000 description 1
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 description 1
- AMRJKAQTDDKMCE-UHFFFAOYSA-N dolastatin Chemical compound CC(C)C(N(C)C)C(=O)NC(C(C)C)C(=O)N(C)C(C(C)C)C(OC)CC(=O)N1CCCC1C(OC)C(C)C(=O)NC(C=1SC=CN=1)CC1=CC=CC=C1 AMRJKAQTDDKMCE-UHFFFAOYSA-N 0.000 description 1
- 229930188854 dolastatin Natural products 0.000 description 1
- ZWAOHEXOSAUJHY-ZIYNGMLESA-N doxifluridine Chemical compound O[C@@H]1[C@H](O)[C@@H](C)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ZWAOHEXOSAUJHY-ZIYNGMLESA-N 0.000 description 1
- 229950005454 doxifluridine Drugs 0.000 description 1
- 229940115080 doxil Drugs 0.000 description 1
- 229940017825 dromostanolone Drugs 0.000 description 1
- 229960004242 dronabinol Drugs 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 239000012636 effector Substances 0.000 description 1
- 229950004438 elinafide Drugs 0.000 description 1
- 229940053603 elitek Drugs 0.000 description 1
- 229940087477 ellence Drugs 0.000 description 1
- 229940120655 eloxatin Drugs 0.000 description 1
- 229940073038 elspar Drugs 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 201000009409 embryonal rhabdomyosarcoma Diseases 0.000 description 1
- 229940000733 emcyt Drugs 0.000 description 1
- 229950005450 emitefur Drugs 0.000 description 1
- 239000003974 emollient agent Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 201000003914 endometrial carcinoma Diseases 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 229950011487 enocitabine Drugs 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 229960001904 epirubicin Drugs 0.000 description 1
- 229940089118 epogen Drugs 0.000 description 1
- 229940082789 erbitux Drugs 0.000 description 1
- 229960001433 erlotinib Drugs 0.000 description 1
- 230000000925 erythroid effect Effects 0.000 description 1
- 210000003238 esophagus Anatomy 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 150000002168 ethanoic acid esters Chemical class 0.000 description 1
- UREBWPXBXRYXRJ-UHFFFAOYSA-N ethyl acetate;methanol Chemical compound OC.CCOC(C)=O UREBWPXBXRYXRJ-UHFFFAOYSA-N 0.000 description 1
- 235000019325 ethyl cellulose Nutrition 0.000 description 1
- 229920001249 ethyl cellulose Polymers 0.000 description 1
- JEFPWOBULVSOTM-PPHPATTJSA-N ethyl n-[(2s)-5-amino-2-methyl-3-phenyl-1,2-dihydropyrido[3,4-b]pyrazin-7-yl]carbamate;2-hydroxyethanesulfonic acid Chemical compound OCCS(O)(=O)=O.C=1([C@H](C)NC=2C=C(N=C(N)C=2N=1)NC(=O)OCC)C1=CC=CC=C1 JEFPWOBULVSOTM-PPHPATTJSA-N 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 229940098617 ethyol Drugs 0.000 description 1
- 229940043168 fareston Drugs 0.000 description 1
- 229940087861 faslodex Drugs 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 229940087476 femara Drugs 0.000 description 1
- 229960002297 fenofibrate Drugs 0.000 description 1
- YMTINGFKWWXKFG-UHFFFAOYSA-N fenofibrate Chemical compound C1=CC(OC(C)(C)C(=O)OC(C)C)=CC=C1C(=O)C1=CC=C(Cl)C=C1 YMTINGFKWWXKFG-UHFFFAOYSA-N 0.000 description 1
- DBEPLOCGEIEOCV-WSBQPABSSA-N finasteride Chemical compound N([C@@H]1CC2)C(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H](C(=O)NC(C)(C)C)[C@@]2(C)CC1 DBEPLOCGEIEOCV-WSBQPABSSA-N 0.000 description 1
- 229960004039 finasteride Drugs 0.000 description 1
- 229960000961 floxuridine Drugs 0.000 description 1
- 229960005304 fludarabine phosphate Drugs 0.000 description 1
- XSFJVAJPIHIPKU-XWCQMRHXSA-N flunisolide Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O XSFJVAJPIHIPKU-XWCQMRHXSA-N 0.000 description 1
- 229960000676 flunisolide Drugs 0.000 description 1
- 229960002690 fluphenazine Drugs 0.000 description 1
- 229960002074 flutamide Drugs 0.000 description 1
- MKXKFYHWDHIYRV-UHFFFAOYSA-N flutamide Chemical compound CC(C)C(=O)NC1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 MKXKFYHWDHIYRV-UHFFFAOYSA-N 0.000 description 1
- 229960003765 fluvastatin Drugs 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- XZBIXDPGRMLSTC-UHFFFAOYSA-N formohydrazide Chemical compound NNC=O XZBIXDPGRMLSTC-UHFFFAOYSA-N 0.000 description 1
- FATAVLOOLIRUNA-UHFFFAOYSA-N formylmethyl Chemical group [CH2]C=O FATAVLOOLIRUNA-UHFFFAOYSA-N 0.000 description 1
- 229960004783 fotemustine Drugs 0.000 description 1
- YAKWPXVTIGTRJH-UHFFFAOYSA-N fotemustine Chemical compound CCOP(=O)(OCC)C(C)NC(=O)N(CCCl)N=O YAKWPXVTIGTRJH-UHFFFAOYSA-N 0.000 description 1
- 239000012458 free base Substances 0.000 description 1
- 229950011325 galarubicin Drugs 0.000 description 1
- 229950004410 galocitabine Drugs 0.000 description 1
- 229940083124 ganglion-blocking antiadrenergic secondary and tertiary amines Drugs 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 208000015419 gastrin-producing neuroendocrine tumor Diseases 0.000 description 1
- 201000000052 gastrinoma Diseases 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 229960002584 gefitinib Drugs 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 229960005277 gemcitabine Drugs 0.000 description 1
- 229960003627 gemfibrozil Drugs 0.000 description 1
- 229940020967 gemzar Drugs 0.000 description 1
- 230000002068 genetic effect Effects 0.000 description 1
- 229940045109 genistein Drugs 0.000 description 1
- TZBJGXHYKVUXJN-UHFFFAOYSA-N genistein Natural products C1=CC(O)=CC=C1C1=COC2=CC(O)=CC(O)=C2C1=O TZBJGXHYKVUXJN-UHFFFAOYSA-N 0.000 description 1
- 235000006539 genistein Nutrition 0.000 description 1
- ZCOLJUOHXJRHDI-CMWLGVBASA-N genistein 7-O-beta-D-glucoside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=C2C(=O)C(C=3C=CC(O)=CC=3)=COC2=C1 ZCOLJUOHXJRHDI-CMWLGVBASA-N 0.000 description 1
- 201000003115 germ cell cancer Diseases 0.000 description 1
- 229940080856 gleevec Drugs 0.000 description 1
- 229940084910 gliadel Drugs 0.000 description 1
- 229960002442 glucosamine Drugs 0.000 description 1
- 229950011595 glufosfamide Drugs 0.000 description 1
- 229960002989 glutamic acid Drugs 0.000 description 1
- 208000026436 grade III glioma Diseases 0.000 description 1
- MFWNKCLOYSRHCJ-BTTYYORXSA-N granisetron Chemical compound C1=CC=C2C(C(=O)N[C@H]3C[C@H]4CCC[C@@H](C3)N4C)=NN(C)C2=C1 MFWNKCLOYSRHCJ-BTTYYORXSA-N 0.000 description 1
- 229960003727 granisetron Drugs 0.000 description 1
- 210000003714 granulocyte Anatomy 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- LHGVFZTZFXWLCP-UHFFFAOYSA-N guaiacol Chemical class COC1=CC=CC=C1O LHGVFZTZFXWLCP-UHFFFAOYSA-N 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 230000003394 haemopoietic effect Effects 0.000 description 1
- 201000009277 hairy cell leukemia Diseases 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000002489 hematologic effect Effects 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 208000006359 hepatoblastoma Diseases 0.000 description 1
- 201000002735 hepatocellular adenoma Diseases 0.000 description 1
- 231100000844 hepatocellular carcinoma Toxicity 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940022353 herceptin Drugs 0.000 description 1
- 125000004634 hexahydroazepinyl group Chemical group N1(CCCCCC1)* 0.000 description 1
- 229940003183 hexalen Drugs 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 208000029824 high grade glioma Diseases 0.000 description 1
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 1
- 229960002193 histrelin Drugs 0.000 description 1
- 108700020746 histrelin Proteins 0.000 description 1
- 229960003911 histrelin acetate Drugs 0.000 description 1
- 238000011905 homologation Methods 0.000 description 1
- 230000003054 hormonal effect Effects 0.000 description 1
- 229940088013 hycamtin Drugs 0.000 description 1
- XGIHQYAWBCFNPY-AZOCGYLKSA-N hydrabamine Chemical compound C([C@@H]12)CC3=CC(C(C)C)=CC=C3[C@@]2(C)CCC[C@@]1(C)CNCCNC[C@@]1(C)[C@@H]2CCC3=CC(C(C)C)=CC=C3[C@@]2(C)CCC1 XGIHQYAWBCFNPY-AZOCGYLKSA-N 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 229940096120 hydrea Drugs 0.000 description 1
- 239000000017 hydrogel Substances 0.000 description 1
- 238000007327 hydrogenolysis reaction Methods 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 229960001330 hydroxycarbamide Drugs 0.000 description 1
- UWYVPFMHMJIBHE-OWOJBTEDSA-N hydroxymaleic acid group Chemical group O/C(/C(=O)O)=C/C(=O)O UWYVPFMHMJIBHE-OWOJBTEDSA-N 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 230000037417 hyperactivation Effects 0.000 description 1
- 230000003463 hyperproliferative effect Effects 0.000 description 1
- 229940015872 ibandronate Drugs 0.000 description 1
- 229960001680 ibuprofen Drugs 0.000 description 1
- 229940099279 idamycin Drugs 0.000 description 1
- 229950002248 idoxifene Drugs 0.000 description 1
- 229940090411 ifex Drugs 0.000 description 1
- 229960003685 imatinib mesylate Drugs 0.000 description 1
- 150000002460 imidazoles Chemical class 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 239000000367 immunologic factor Substances 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 229950008097 improsulfan Drugs 0.000 description 1
- DBIGHPPNXATHOF-UHFFFAOYSA-N improsulfan Chemical compound CS(=O)(=O)OCCCNCCCOS(C)(=O)=O DBIGHPPNXATHOF-UHFFFAOYSA-N 0.000 description 1
- 201000004933 in situ carcinoma Diseases 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- 125000003387 indolinyl group Chemical group N1(CCC2=CC=CC=C12)* 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 229940102223 injectable solution Drugs 0.000 description 1
- 229940102213 injectable suspension Drugs 0.000 description 1
- 206010022498 insulinoma Diseases 0.000 description 1
- 230000002452 interceptive effect Effects 0.000 description 1
- 229960003521 interferon alfa-2a Drugs 0.000 description 1
- 229960003507 interferon alfa-2b Drugs 0.000 description 1
- 229960003130 interferon gamma Drugs 0.000 description 1
- 229940117681 interleukin-12 Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 210000002570 interstitial cell Anatomy 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 229940065638 intron a Drugs 0.000 description 1
- 201000010985 invasive ductal carcinoma Diseases 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 150000008424 iodobenzenes Chemical class 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 229940084651 iressa Drugs 0.000 description 1
- 229950005254 irofulven Drugs 0.000 description 1
- NICJCIQSJJKZAH-AWEZNQCLSA-N irofulven Chemical compound O=C([C@@]1(O)C)C2=CC(C)=C(CO)C2=C(C)C21CC2 NICJCIQSJJKZAH-AWEZNQCLSA-N 0.000 description 1
- 230000000302 ischemic effect Effects 0.000 description 1
- 125000001977 isobenzofuranyl group Chemical group C=1(OC=C2C=CC=CC12)* 0.000 description 1
- LRDFRRGEGBBSRN-UHFFFAOYSA-N isobutyronitrile Chemical compound CC(C)C#N LRDFRRGEGBBSRN-UHFFFAOYSA-N 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- JJWLVOIRVHMVIS-UHFFFAOYSA-N isopropylamine Chemical compound CC(C)N JJWLVOIRVHMVIS-UHFFFAOYSA-N 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 125000005956 isoquinolyl group Chemical group 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 229960005280 isotretinoin Drugs 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 210000001117 keloid Anatomy 0.000 description 1
- 229940063199 kenalog Drugs 0.000 description 1
- 229940065223 kepivance Drugs 0.000 description 1
- 208000022013 kidney Wilms tumor Diseases 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- DAQAKHDKYAWHCG-RWTHQLGUSA-N lactacystin Chemical compound CC(=O)N[C@H](C(O)=O)CSC(=O)[C@]1([C@@H](O)C(C)C)NC(=O)[C@H](C)[C@@H]1O DAQAKHDKYAWHCG-RWTHQLGUSA-N 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 210000002429 large intestine Anatomy 0.000 description 1
- 229960004942 lenalidomide Drugs 0.000 description 1
- 229940095570 lescol Drugs 0.000 description 1
- 229940063725 leukeran Drugs 0.000 description 1
- 229940087875 leukine Drugs 0.000 description 1
- UGFHIPBXIWJXNA-UHFFFAOYSA-N liarozole Chemical compound ClC1=CC=CC(C(C=2C=C3NC=NC3=CC=2)N2C=NC=C2)=C1 UGFHIPBXIWJXNA-UHFFFAOYSA-N 0.000 description 1
- 229950007056 liarozole Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229940002661 lipitor Drugs 0.000 description 1
- 206010024627 liposarcoma Diseases 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 1
- DBTNVRCCIDISMV-UHFFFAOYSA-L lithium;magnesium;propane;dichloride Chemical compound [Li+].[Mg+2].[Cl-].[Cl-].C[CH-]C DBTNVRCCIDISMV-UHFFFAOYSA-L 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 229950008991 lobaplatin Drugs 0.000 description 1
- 229950000909 lometrexol Drugs 0.000 description 1
- 229960002247 lomustine Drugs 0.000 description 1
- 229960003538 lonidamine Drugs 0.000 description 1
- WDRYRZXSPDWGEB-UHFFFAOYSA-N lonidamine Chemical compound C12=CC=CC=C2C(C(=O)O)=NN1CC1=CC=C(Cl)C=C1Cl WDRYRZXSPDWGEB-UHFFFAOYSA-N 0.000 description 1
- 229960004844 lovastatin Drugs 0.000 description 1
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 1
- 229940127215 low-molecular weight heparin Drugs 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 108010078259 luprolide acetate gel depot Proteins 0.000 description 1
- RVFGKBWWUQOIOU-NDEPHWFRSA-N lurtotecan Chemical compound O=C([C@]1(O)CC)OCC(C(N2CC3=4)=O)=C1C=C2C3=NC1=CC=2OCCOC=2C=C1C=4CN1CCN(C)CC1 RVFGKBWWUQOIOU-NDEPHWFRSA-N 0.000 description 1
- 229950002654 lurtotecan Drugs 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 229940100029 lysodren Drugs 0.000 description 1
- 229940124302 mTOR inhibitor Drugs 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- BRKADVNLTRCLOW-UHFFFAOYSA-M magnesium;fluorobenzene;bromide Chemical compound [Mg+2].[Br-].FC1=CC=[C-]C=C1 BRKADVNLTRCLOW-UHFFFAOYSA-M 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 201000004593 malignant giant cell tumor Diseases 0.000 description 1
- 201000011614 malignant glioma Diseases 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 201000000289 malignant teratoma Diseases 0.000 description 1
- 210000001161 mammalian embryo Anatomy 0.000 description 1
- 239000003628 mammalian target of rapamycin inhibitor Substances 0.000 description 1
- 230000000873 masking effect Effects 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 229940087732 matulane Drugs 0.000 description 1
- 229960004961 mechlorethamine Drugs 0.000 description 1
- 229940090004 megace Drugs 0.000 description 1
- 210000003593 megakaryocyte Anatomy 0.000 description 1
- 229960004296 megestrol acetate Drugs 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 210000002418 meninge Anatomy 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- SLVMESMUVMCQIY-UHFFFAOYSA-N mesoridazine Chemical compound CN1CCCCC1CCN1C2=CC(S(C)=O)=CC=C2SC2=CC=CC=C21 SLVMESMUVMCQIY-UHFFFAOYSA-N 0.000 description 1
- 229960000300 mesoridazine Drugs 0.000 description 1
- 229960004469 methoxsalen Drugs 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- JKPADTOZPCRWBO-UHFFFAOYSA-N methyl 1-[[6-[[3-carbamoyl-5-[2-fluoro-4-(2-hydroxypropan-2-yl)phenyl]thiophen-2-yl]amino]pyridin-2-yl]methyl]triazole-4-carboxylate Chemical compound N1=NC(C(=O)OC)=CN1CC1=CC=CC(NC2=C(C=C(S2)C=2C(=CC(=CC=2)C(C)(C)O)F)C(N)=O)=N1 JKPADTOZPCRWBO-UHFFFAOYSA-N 0.000 description 1
- DYUWQWMXZHDZOR-UHFFFAOYSA-N methyl 4-iodobenzoate Chemical compound COC(=O)C1=CC=C(I)C=C1 DYUWQWMXZHDZOR-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 125000006431 methyl cyclopropyl group Chemical group 0.000 description 1
- KBDQRBDBWLCMKG-UHFFFAOYSA-N methyl n-[4-[4-carbamoyl-5-[[6-(morpholin-4-ylmethyl)pyridin-2-yl]amino]thiophen-2-yl]phenyl]carbamate Chemical compound C1=CC(NC(=O)OC)=CC=C1C(S1)=CC(C(N)=O)=C1NC1=CC=CC(CN2CCOCC2)=N1 KBDQRBDBWLCMKG-UHFFFAOYSA-N 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- TTWJBBZEZQICBI-UHFFFAOYSA-N metoclopramide Chemical compound CCN(CC)CCNC(=O)C1=CC(Cl)=C(N)C=C1OC TTWJBBZEZQICBI-UHFFFAOYSA-N 0.000 description 1
- 229960004503 metoclopramide Drugs 0.000 description 1
- 229940099246 mevacor Drugs 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 239000002395 mineralocorticoid Substances 0.000 description 1
- VMMKGHQPQIEGSQ-UHFFFAOYSA-N minodronic acid Chemical compound C1=CC=CN2C(CC(O)(P(O)(O)=O)P(O)(O)=O)=CN=C21 VMMKGHQPQIEGSQ-UHFFFAOYSA-N 0.000 description 1
- 229950011129 minodronic acid Drugs 0.000 description 1
- 229950010913 mitolactol Drugs 0.000 description 1
- VFKZTMPDYBFSTM-GUCUJZIJSA-N mitolactol Chemical compound BrC[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)CBr VFKZTMPDYBFSTM-GUCUJZIJSA-N 0.000 description 1
- 229960000350 mitotane Drugs 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 239000002324 mouth wash Substances 0.000 description 1
- 208000010492 mucinous cystadenocarcinoma Diseases 0.000 description 1
- 229940087004 mustargen Drugs 0.000 description 1
- 208000025113 myeloid leukemia Diseases 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 229940090009 myleran Drugs 0.000 description 1
- 208000009091 myxoma Diseases 0.000 description 1
- UDGSVBYJWHOHNN-UHFFFAOYSA-N n',n'-diethylethane-1,2-diamine Chemical group CCN(CC)CCN UDGSVBYJWHOHNN-UHFFFAOYSA-N 0.000 description 1
- BGTBRDJUHRMBQB-UHFFFAOYSA-N n,n-dimethylmethanamine;n,n-dipropylpropan-1-amine Chemical compound CN(C)C.CCCN(CCC)CCC BGTBRDJUHRMBQB-UHFFFAOYSA-N 0.000 description 1
- DXASQZJWWGZNSF-UHFFFAOYSA-N n,n-dimethylmethanamine;sulfur trioxide Chemical group CN(C)C.O=S(=O)=O DXASQZJWWGZNSF-UHFFFAOYSA-N 0.000 description 1
- NJSMWLQOCQIOPE-OCHFTUDZSA-N n-[(e)-[10-[(e)-(4,5-dihydro-1h-imidazol-2-ylhydrazinylidene)methyl]anthracen-9-yl]methylideneamino]-4,5-dihydro-1h-imidazol-2-amine Chemical compound N1CCN=C1N\N=C\C(C1=CC=CC=C11)=C(C=CC=C2)C2=C1\C=N\NC1=NCCN1 NJSMWLQOCQIOPE-OCHFTUDZSA-N 0.000 description 1
- TVYPSLDUBVTDIS-FUOMVGGVSA-N n-[1-[(2r,3r,4s,5r)-3,4-dihydroxy-5-methyloxolan-2-yl]-5-fluoro-2-oxopyrimidin-4-yl]-3,4,5-trimethoxybenzamide Chemical compound COC1=C(OC)C(OC)=CC(C(=O)NC=2C(=CN(C(=O)N=2)[C@H]2[C@@H]([C@H](O)[C@@H](C)O2)O)F)=C1 TVYPSLDUBVTDIS-FUOMVGGVSA-N 0.000 description 1
- LBWFXVZLPYTWQI-IPOVEDGCSA-N n-[2-(diethylamino)ethyl]-5-[(z)-(5-fluoro-2-oxo-1h-indol-3-ylidene)methyl]-2,4-dimethyl-1h-pyrrole-3-carboxamide;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C LBWFXVZLPYTWQI-IPOVEDGCSA-N 0.000 description 1
- ZDUZYDDAHVZGCI-UHFFFAOYSA-N n-[2-(dimethylamino)ethyl]-9-hydroxy-5,6-dimethylpyrido[4,3-b]carbazole-1-carboxamide Chemical compound CN1C2=CC=C(O)C=C2C2=C1C(C)=C1C=CN=C(C(=O)NCCN(C)C)C1=C2 ZDUZYDDAHVZGCI-UHFFFAOYSA-N 0.000 description 1
- XBGNERSKEKDZDS-UHFFFAOYSA-N n-[2-(dimethylamino)ethyl]acridine-4-carboxamide Chemical compound C1=CC=C2N=C3C(C(=O)NCCN(C)C)=CC=CC3=CC2=C1 XBGNERSKEKDZDS-UHFFFAOYSA-N 0.000 description 1
- LZGSVLJFDBYFMR-UHFFFAOYSA-N n-[5-(1-methyltriazol-4-yl)thiophen-2-yl]-6-(morpholin-4-ylmethyl)pyridin-2-amine Chemical compound N1=NN(C)C=C1C(S1)=CC=C1NC1=CC=CC(CN2CCOCC2)=N1 LZGSVLJFDBYFMR-UHFFFAOYSA-N 0.000 description 1
- LCQYPDGCGFIGOE-UHFFFAOYSA-N n-[[2-nitro-5-[1-(2-trimethylsilylethoxymethyl)-1,2,4-triazol-3-yl]thiophen-3-yl]methylidene]hydroxylamine Chemical compound C[Si](C)(C)CCOCN1C=NC(C=2SC(=C(C=NO)C=2)[N+]([O-])=O)=N1 LCQYPDGCGFIGOE-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- OHDXDNUPVVYWOV-UHFFFAOYSA-N n-methyl-1-(2-naphthalen-1-ylsulfanylphenyl)methanamine Chemical compound CNCC1=CC=CC=C1SC1=CC=CC2=CC=CC=C12 OHDXDNUPVVYWOV-UHFFFAOYSA-N 0.000 description 1
- 229960001133 nandrolone phenpropionate Drugs 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 229940086322 navelbine Drugs 0.000 description 1
- 210000003739 neck Anatomy 0.000 description 1
- 229950007221 nedaplatin Drugs 0.000 description 1
- 230000006654 negative regulation of apoptotic process Effects 0.000 description 1
- 230000017095 negative regulation of cell growth Effects 0.000 description 1
- 230000035407 negative regulation of cell proliferation Effects 0.000 description 1
- 230000017066 negative regulation of growth Effects 0.000 description 1
- 201000008026 nephroblastoma Diseases 0.000 description 1
- PUUSSSIBPPTKTP-UHFFFAOYSA-N neridronic acid Chemical compound NCCCCCC(O)(P(O)(O)=O)P(O)(O)=O PUUSSSIBPPTKTP-UHFFFAOYSA-N 0.000 description 1
- 229950010733 neridronic acid Drugs 0.000 description 1
- 210000000653 nervous system Anatomy 0.000 description 1
- PKWDZWYVIHVNKS-UHFFFAOYSA-N netoglitazone Chemical compound FC1=CC=CC=C1COC1=CC=C(C=C(CC2C(NC(=O)S2)=O)C=C2)C2=C1 PKWDZWYVIHVNKS-UHFFFAOYSA-N 0.000 description 1
- 229940071846 neulasta Drugs 0.000 description 1
- 229940082926 neumega Drugs 0.000 description 1
- 229940029345 neupogen Drugs 0.000 description 1
- 208000007538 neurilemmoma Diseases 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 201000004662 neurofibroma of spinal cord Diseases 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 208000004649 neutrophil actin dysfunction Diseases 0.000 description 1
- 229940080607 nexavar Drugs 0.000 description 1
- XWXYUMMDTVBTOU-UHFFFAOYSA-N nilutamide Chemical compound O=C1C(C)(C)NC(=O)N1C1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 XWXYUMMDTVBTOU-UHFFFAOYSA-N 0.000 description 1
- 229960002653 nilutamide Drugs 0.000 description 1
- 229960001420 nimustine Drugs 0.000 description 1
- VFEDRRNHLBGPNN-UHFFFAOYSA-N nimustine Chemical compound CC1=NC=C(CNC(=O)N(CCCl)N=O)C(N)=N1 VFEDRRNHLBGPNN-UHFFFAOYSA-N 0.000 description 1
- 229940109551 nipent Drugs 0.000 description 1
- XHWRWCSCBDLOLM-UHFFFAOYSA-N nolatrexed Chemical compound CC1=CC=C2NC(N)=NC(=O)C2=C1SC1=CC=NC=C1 XHWRWCSCBDLOLM-UHFFFAOYSA-N 0.000 description 1
- 229950000891 nolatrexed Drugs 0.000 description 1
- 229940085033 nolvadex Drugs 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- GYCKQBWUSACYIF-UHFFFAOYSA-N o-hydroxybenzoic acid ethyl ester Natural products CCOC(=O)C1=CC=CC=C1O GYCKQBWUSACYIF-UHFFFAOYSA-N 0.000 description 1
- 239000007764 o/w emulsion Substances 0.000 description 1
- 235000020824 obesity Nutrition 0.000 description 1
- 229960000435 oblimersen Drugs 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000009437 off-target effect Effects 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 229940099216 oncaspar Drugs 0.000 description 1
- 229960005343 ondansetron Drugs 0.000 description 1
- 229940100027 ontak Drugs 0.000 description 1
- 229960001840 oprelvekin Drugs 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 208000023983 oral cavity neoplasm Diseases 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 125000002524 organometallic group Chemical group 0.000 description 1
- 201000008482 osteoarthritis Diseases 0.000 description 1
- 208000003388 osteoid osteoma Diseases 0.000 description 1
- 208000008798 osteoma Diseases 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- 210000003101 oviduct Anatomy 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000003566 oxetanyl group Chemical group 0.000 description 1
- 238000004806 packaging method and process Methods 0.000 description 1
- 229960002502 paclitaxel protein-bound Drugs 0.000 description 1
- 229960002404 palifermin Drugs 0.000 description 1
- 238000010651 palladium-catalyzed cross coupling reaction Methods 0.000 description 1
- 125000001312 palmitoyl group Chemical group O=C([*])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- 208000021255 pancreatic insulinoma Diseases 0.000 description 1
- 229940096763 panretin Drugs 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 229960004662 parecoxib Drugs 0.000 description 1
- TZRHLKRLEZJVIJ-UHFFFAOYSA-N parecoxib Chemical compound C1=CC(S(=O)(=O)NC(=O)CC)=CC=C1C1=C(C)ON=C1C1=CC=CC=C1 TZRHLKRLEZJVIJ-UHFFFAOYSA-N 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- HQQSBEDKMRHYME-UHFFFAOYSA-N pefloxacin mesylate Chemical compound [H+].CS([O-])(=O)=O.C1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC(F)=C1N1CCN(C)CC1 HQQSBEDKMRHYME-UHFFFAOYSA-N 0.000 description 1
- 229960001218 pegademase Drugs 0.000 description 1
- 229940048111 pegademase bovine Drugs 0.000 description 1
- 229960001744 pegaspargase Drugs 0.000 description 1
- 229960001373 pegfilgrastim Drugs 0.000 description 1
- 229960005079 pemetrexed Drugs 0.000 description 1
- 229960003349 pemetrexed disodium Drugs 0.000 description 1
- 229940043138 pentosan polysulfate Drugs 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- 239000008177 pharmaceutical agent Substances 0.000 description 1
- 210000003800 pharynx Anatomy 0.000 description 1
- 150000002990 phenothiazines Chemical class 0.000 description 1
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 1
- HFNGWVXNSWWIGC-UHFFFAOYSA-N phenylmethoxy carbonochloridate Chemical compound ClC(=O)OOCC1=CC=CC=C1 HFNGWVXNSWWIGC-UHFFFAOYSA-N 0.000 description 1
- 229930015698 phenylpropene Natural products 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 150000008105 phosphatidylcholines Chemical class 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 229940109328 photofrin Drugs 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 230000004962 physiological condition Effects 0.000 description 1
- 229950004317 pinafide Drugs 0.000 description 1
- 208000024724 pineal body neoplasm Diseases 0.000 description 1
- 201000004123 pineal gland cancer Diseases 0.000 description 1
- 229960005095 pioglitazone Drugs 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229960005235 piperonyl butoxide Drugs 0.000 description 1
- 229960001221 pirarubicin Drugs 0.000 description 1
- 229940037129 plain mineralocorticoids for systemic use Drugs 0.000 description 1
- 229940063179 platinol Drugs 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- HRGDZIGMBDGFTC-UHFFFAOYSA-N platinum(2+) Chemical compound [Pt+2] HRGDZIGMBDGFTC-UHFFFAOYSA-N 0.000 description 1
- 229950008499 plitidepsin Drugs 0.000 description 1
- UUSZLLQJYRSZIS-LXNNNBEUSA-N plitidepsin Chemical compound CN([C@H](CC(C)C)C(=O)N[C@@H]1C(=O)N[C@@H]([C@H](CC(=O)O[C@H](C(=O)[C@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N2CCC[C@H]2C(=O)N(C)[C@@H](CC=2C=CC(OC)=CC=2)C(=O)O[C@@H]1C)C(C)C)O)[C@@H](C)CC)C(=O)[C@@H]1CCCN1C(=O)C(C)=O UUSZLLQJYRSZIS-LXNNNBEUSA-N 0.000 description 1
- 108010049948 plitidepsin Proteins 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229940098901 polifeprosan 20 Drugs 0.000 description 1
- 229920000747 poly(lactic acid) Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 229920001610 polycaprolactone Polymers 0.000 description 1
- 229920002721 polycyanoacrylate Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000004626 polylactic acid Substances 0.000 description 1
- 229920000656 polylysine Polymers 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 229920006324 polyoxymethylene Polymers 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 229960004293 porfimer sodium Drugs 0.000 description 1
- 229940089484 pravachol Drugs 0.000 description 1
- 229960002965 pravastatin Drugs 0.000 description 1
- TUZYXOIXSAXUGO-PZAWKZKUSA-N pravastatin Chemical compound C1=C[C@H](C)[C@H](CC[C@@H](O)C[C@@H](O)CC(O)=O)[C@H]2[C@@H](OC(=O)[C@@H](C)CC)C[C@H](O)C=C21 TUZYXOIXSAXUGO-PZAWKZKUSA-N 0.000 description 1
- VWBQYTRBTXKKOG-IYNICTALSA-M pravastatin sodium Chemical compound [Na+].C1=C[C@H](C)[C@H](CC[C@@H](O)C[C@@H](O)CC([O-])=O)[C@H]2[C@@H](OC(=O)[C@@H](C)CC)C[C@H](O)C=C21 VWBQYTRBTXKKOG-IYNICTALSA-M 0.000 description 1
- 229960004694 prednimustine Drugs 0.000 description 1
- 229960005205 prednisolone Drugs 0.000 description 1
- OIGNJSKKLXVSLS-VWUMJDOOSA-N prednisolone Chemical compound O=C1C=C[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 OIGNJSKKLXVSLS-VWUMJDOOSA-N 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 150000003141 primary amines Chemical class 0.000 description 1
- 210000001948 pro-b lymphocyte Anatomy 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- 229960000624 procarbazine Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- WIKYUJGCLQQFNW-UHFFFAOYSA-N prochlorperazine Chemical compound C1CN(C)CCN1CCCN1C2=CC(Cl)=CC=C2SC2=CC=CC=C21 WIKYUJGCLQQFNW-UHFFFAOYSA-N 0.000 description 1
- 229960003111 prochlorperazine Drugs 0.000 description 1
- 229940087463 proleukin Drugs 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- VQGISNOMGHCEPX-UHFFFAOYSA-N propanenitrile Chemical compound C[CH]C#N VQGISNOMGHCEPX-UHFFFAOYSA-N 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- WGYKZJWCGVVSQN-UHFFFAOYSA-N propylamine Chemical compound CCCN WGYKZJWCGVVSQN-UHFFFAOYSA-N 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 239000002599 prostaglandin synthase inhibitor Substances 0.000 description 1
- 108060006633 protein kinase Proteins 0.000 description 1
- 229950007401 pumitepa Drugs 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 229940117820 purinethol Drugs 0.000 description 1
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- XCRPPAPDRUBKRJ-UHFFFAOYSA-N quinolin-7-ol Chemical compound C1=CC=NC2=CC(O)=CC=C21 XCRPPAPDRUBKRJ-UHFFFAOYSA-N 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000005493 quinolyl group Chemical group 0.000 description 1
- 229960004622 raloxifene Drugs 0.000 description 1
- GZUITABIAKMVPG-UHFFFAOYSA-N raloxifene Chemical compound C1=CC(O)=CC=C1C1=C(C(=O)C=2C=CC(OCCN3CCCCC3)=CC=2)C2=CC=C(O)C=C2S1 GZUITABIAKMVPG-UHFFFAOYSA-N 0.000 description 1
- 229960004432 raltitrexed Drugs 0.000 description 1
- 229960002185 ranimustine Drugs 0.000 description 1
- 229950007649 ranpirnase Drugs 0.000 description 1
- 108010061338 ranpirnase Proteins 0.000 description 1
- 229960000424 rasburicase Drugs 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 230000008707 rearrangement Effects 0.000 description 1
- 108091006084 receptor activators Proteins 0.000 description 1
- 229940044601 receptor agonist Drugs 0.000 description 1
- 239000000018 receptor agonist Substances 0.000 description 1
- 229940044551 receptor antagonist Drugs 0.000 description 1
- 239000002464 receptor antagonist Substances 0.000 description 1
- 238000001953 recrystallisation Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000000306 recurrent effect Effects 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 239000011347 resin Substances 0.000 description 1
- 229920005989 resin Polymers 0.000 description 1
- 208000029922 reticulum cell sarcoma Diseases 0.000 description 1
- 208000004644 retinal vein occlusion Diseases 0.000 description 1
- 229940100552 retinamide Drugs 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 229940120975 revlimid Drugs 0.000 description 1
- 201000009410 rhabdomyosarcoma Diseases 0.000 description 1
- OWPCHSCAPHNHAV-LMONGJCWSA-N rhizoxin Chemical compound C/C([C@H](OC)[C@@H](C)[C@@H]1C[C@H](O)[C@]2(C)O[C@@H]2/C=C/[C@@H](C)[C@]2([H])OC(=O)C[C@@](C2)(C[C@@H]2O[C@H]2C(=O)O1)[H])=C\C=C\C(\C)=C\C1=COC(C)=N1 OWPCHSCAPHNHAV-LMONGJCWSA-N 0.000 description 1
- 125000006413 ring segment Chemical group 0.000 description 1
- 229940089617 risedronate Drugs 0.000 description 1
- 229960000371 rofecoxib Drugs 0.000 description 1
- 229960004586 rosiglitazone Drugs 0.000 description 1
- SUFUKZSWUHZXAV-BTJKTKAUSA-N rosiglitazone maleate Chemical compound [H+].[H+].[O-]C(=O)\C=C/C([O-])=O.C=1C=CC=NC=1N(C)CCOC(C=C1)=CC=C1CC1SC(=O)NC1=O SUFUKZSWUHZXAV-BTJKTKAUSA-N 0.000 description 1
- 229960003271 rosiglitazone maleate Drugs 0.000 description 1
- 238000002390 rotary evaporation Methods 0.000 description 1
- 102200087780 rs77375493 Human genes 0.000 description 1
- VHXNKPBCCMUMSW-FQEVSTJZSA-N rubitecan Chemical compound C1=CC([N+]([O-])=O)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VHXNKPBCCMUMSW-FQEVSTJZSA-N 0.000 description 1
- 229950009213 rubitecan Drugs 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 229960005399 satraplatin Drugs 0.000 description 1
- 190014017285 satraplatin Chemical compound 0.000 description 1
- 206010039667 schwannoma Diseases 0.000 description 1
- 229940053186 sclerosol Drugs 0.000 description 1
- BTIHMVBBUGXLCJ-OAHLLOKOSA-N seliciclib Chemical compound C=12N=CN(C(C)C)C2=NC(N[C@@H](CO)CC)=NC=1NCC1=CC=CC=C1 BTIHMVBBUGXLCJ-OAHLLOKOSA-N 0.000 description 1
- 229950003647 semaxanib Drugs 0.000 description 1
- 230000001235 sensitizing effect Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000003369 serotonin 5-HT3 receptor antagonist Substances 0.000 description 1
- 208000004548 serous cystadenocarcinoma Diseases 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 229960002855 simvastatin Drugs 0.000 description 1
- 210000003625 skull Anatomy 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 229950010372 sobuzoxane Drugs 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- PPASLZSBLFJQEF-RKJRWTFHSA-M sodium ascorbate Substances [Na+].OC[C@@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RKJRWTFHSA-M 0.000 description 1
- 235000010378 sodium ascorbate Nutrition 0.000 description 1
- 229960005055 sodium ascorbate Drugs 0.000 description 1
- WRIKHQLVHPKCJU-UHFFFAOYSA-N sodium bis(trimethylsilyl)amide Chemical compound C[Si](C)(C)N([Na])[Si](C)(C)C WRIKHQLVHPKCJU-UHFFFAOYSA-N 0.000 description 1
- 229910001467 sodium calcium phosphate Inorganic materials 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 1
- PPASLZSBLFJQEF-RXSVEWSESA-M sodium-L-ascorbate Chemical compound [Na+].OC[C@H](O)[C@H]1OC(=O)C(O)=C1[O-] PPASLZSBLFJQEF-RXSVEWSESA-M 0.000 description 1
- MIXCUJKCXRNYFM-UHFFFAOYSA-M sodium;diiodomethanesulfonate;n-propyl-n-[2-(2,4,6-trichlorophenoxy)ethyl]imidazole-1-carboxamide Chemical compound [Na+].[O-]S(=O)(=O)C(I)I.C1=CN=CN1C(=O)N(CCC)CCOC1=C(Cl)C=C(Cl)C=C1Cl MIXCUJKCXRNYFM-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 238000000527 sonication Methods 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 229940083466 soybean lecithin Drugs 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 230000003637 steroidlike Effects 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000005017 substituted alkenyl group Chemical group 0.000 description 1
- 125000004426 substituted alkynyl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 229940034785 sutent Drugs 0.000 description 1
- 229960005566 swainsonine Drugs 0.000 description 1
- FXUAIOOAOAVCGD-FKSUSPILSA-N swainsonine Chemical compound C1CC[C@H](O)[C@H]2[C@H](O)[C@H](O)CN21 FXUAIOOAOAVCGD-FKSUSPILSA-N 0.000 description 1
- FXUAIOOAOAVCGD-UHFFFAOYSA-N swainsonine Natural products C1CCC(O)C2C(O)C(O)CN21 FXUAIOOAOAVCGD-UHFFFAOYSA-N 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- FKHIFSZMMVMEQY-UHFFFAOYSA-N talc Chemical compound [Mg+2].[O-][Si]([O-])=O FKHIFSZMMVMEQY-UHFFFAOYSA-N 0.000 description 1
- AYUNIORJHRXIBJ-TXHRRWQRSA-N tanespimycin Chemical compound N1C(=O)\C(C)=C\C=C/[C@H](OC)[C@@H](OC(N)=O)\C(C)=C\[C@H](C)[C@@H](O)[C@@H](OC)C[C@H](C)CC2=C(NCC=C)C(=O)C=C1C2=O AYUNIORJHRXIBJ-TXHRRWQRSA-N 0.000 description 1
- 229940120982 tarceva Drugs 0.000 description 1
- 229960003102 tasonermin Drugs 0.000 description 1
- RCINICONZNJXQF-XAZOAEDWSA-N taxol® Chemical compound O([C@@H]1[C@@]2(CC(C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3(C21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-XAZOAEDWSA-N 0.000 description 1
- 229940063683 taxotere Drugs 0.000 description 1
- 229960001674 tegafur Drugs 0.000 description 1
- WFWLQNSHRPWKFK-ZCFIWIBFSA-N tegafur Chemical compound O=C1NC(=O)C(F)=CN1[C@@H]1OCCC1 WFWLQNSHRPWKFK-ZCFIWIBFSA-N 0.000 description 1
- 229940061353 temodar Drugs 0.000 description 1
- 208000001608 teratocarcinoma Diseases 0.000 description 1
- 230000002381 testicular Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 210000001550 testis Anatomy 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- 125000001712 tetrahydronaphthyl group Chemical group C1(CCCC2=CC=CC=C12)* 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 229960003433 thalidomide Drugs 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 150000001467 thiazolidinediones Chemical class 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 229960002784 thioridazine Drugs 0.000 description 1
- 229960001196 thiotepa Drugs 0.000 description 1
- NZVYCXVTEHPMHE-ZSUJOUNUSA-N thymalfasin Chemical compound CC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O NZVYCXVTEHPMHE-ZSUJOUNUSA-N 0.000 description 1
- 229960004231 thymalfasin Drugs 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 229960003723 tiazofurine Drugs 0.000 description 1
- FVRDYQYEVDDKCR-DBRKOABJSA-N tiazofurine Chemical compound NC(=O)C1=CSC([C@H]2[C@@H]([C@H](O)[C@@H](CO)O2)O)=N1 FVRDYQYEVDDKCR-DBRKOABJSA-N 0.000 description 1
- 229940019375 tiludronate Drugs 0.000 description 1
- 229950002376 tirapazamine Drugs 0.000 description 1
- ORYDPOVDJJZGHQ-UHFFFAOYSA-N tirapazamine Chemical compound C1=CC=CC2=[N+]([O-])C(N)=N[N+]([O-])=C21 ORYDPOVDJJZGHQ-UHFFFAOYSA-N 0.000 description 1
- 230000000451 tissue damage Effects 0.000 description 1
- 231100000827 tissue damage Toxicity 0.000 description 1
- AOBORMOPSGHCAX-DGHZZKTQSA-N tocofersolan Chemical compound OCCOC(=O)CCC(=O)OC1=C(C)C(C)=C2O[C@](CCC[C@H](C)CCC[C@H](C)CCCC(C)C)(C)CCC2=C1C AOBORMOPSGHCAX-DGHZZKTQSA-N 0.000 description 1
- 229960000984 tocofersolan Drugs 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- PKVRCIRHQMSYJX-AIFWHQITSA-N trabectedin Chemical compound C([C@@]1(C(OC2)=O)NCCC3=C1C=C(C(=C3)O)OC)S[C@@H]1C3=C(OC(C)=O)C(C)=C4OCOC4=C3[C@H]2N2[C@@H](O)[C@H](CC=3C4=C(O)C(OC)=C(C)C=3)N(C)[C@H]4[C@@H]21 PKVRCIRHQMSYJX-AIFWHQITSA-N 0.000 description 1
- 229960000977 trabectedin Drugs 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000037317 transdermal delivery Effects 0.000 description 1
- 230000001131 transforming effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 206010044412 transitional cell carcinoma Diseases 0.000 description 1
- 229960005526 triapine Drugs 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 1
- WRECIMRULFAWHA-UHFFFAOYSA-N trimethyl borate Chemical compound COB(OC)OC WRECIMRULFAWHA-UHFFFAOYSA-N 0.000 description 1
- YHYUJZFXCDRHHZ-UHFFFAOYSA-N trimethyl-[2-[[5-(5-nitrothiophen-2-yl)-1,2,4-triazol-1-yl]methoxy]ethyl]silane Chemical compound C[Si](C)(C)CCOCN1N=CN=C1C1=CC=C([N+]([O-])=O)S1 YHYUJZFXCDRHHZ-UHFFFAOYSA-N 0.000 description 1
- SEDZOYHHAIAQIW-UHFFFAOYSA-N trimethylsilyl azide Chemical compound C[Si](C)(C)N=[N+]=[N-] SEDZOYHHAIAQIW-UHFFFAOYSA-N 0.000 description 1
- 229960001099 trimetrexate Drugs 0.000 description 1
- NOYPYLRCIDNJJB-UHFFFAOYSA-N trimetrexate Chemical compound COC1=C(OC)C(OC)=CC(NCC=2C(=C3C(N)=NC(N)=NC3=CC=2)C)=C1 NOYPYLRCIDNJJB-UHFFFAOYSA-N 0.000 description 1
- AVCVDUDESCZFHJ-UHFFFAOYSA-N triphenylphosphane;hydrochloride Chemical compound [Cl-].C1=CC=CC=C1[PH+](C=1C=CC=CC=1)C1=CC=CC=C1 AVCVDUDESCZFHJ-UHFFFAOYSA-N 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229940086984 trisenox Drugs 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 238000001665 trituration Methods 0.000 description 1
- 229960000875 trofosfamide Drugs 0.000 description 1
- UMKFEPPTGMDVMI-UHFFFAOYSA-N trofosfamide Chemical compound ClCCN(CCCl)P1(=O)OCCCN1CCCl UMKFEPPTGMDVMI-UHFFFAOYSA-N 0.000 description 1
- 229960000281 trometamol Drugs 0.000 description 1
- ZNRGQMMCGHDTEI-ITGUQSILSA-N tropisetron Chemical compound C1=CC=C2C(C(=O)O[C@H]3C[C@H]4CC[C@@H](C3)N4C)=CNC2=C1 ZNRGQMMCGHDTEI-ITGUQSILSA-N 0.000 description 1
- 229960003688 tropisetron Drugs 0.000 description 1
- 229950010147 troxacitabine Drugs 0.000 description 1
- RXRGZNYSEHTMHC-BQBZGAKWSA-N troxacitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1O[C@@H](CO)OC1 RXRGZNYSEHTMHC-BQBZGAKWSA-N 0.000 description 1
- 208000022271 tubular adenoma Diseases 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 102000003390 tumor necrosis factor Human genes 0.000 description 1
- 230000005751 tumor progression Effects 0.000 description 1
- 208000027930 type IV hypersensitivity disease Diseases 0.000 description 1
- 125000001493 tyrosinyl group Chemical group [H]OC1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 1
- 241000701161 unidentified adenovirus Species 0.000 description 1
- 210000003708 urethra Anatomy 0.000 description 1
- 208000010570 urinary bladder carcinoma Diseases 0.000 description 1
- 210000004291 uterus Anatomy 0.000 description 1
- 229940001814 uvadex Drugs 0.000 description 1
- 210000001215 vagina Anatomy 0.000 description 1
- LNPDTQAFDNKSHK-UHFFFAOYSA-N valdecoxib Chemical compound CC=1ON=C(C=2C=CC=CC=2)C=1C1=CC=C(S(N)(=O)=O)C=C1 LNPDTQAFDNKSHK-UHFFFAOYSA-N 0.000 description 1
- 229940054937 valstar Drugs 0.000 description 1
- YCOYDOIWSSHVCK-UHFFFAOYSA-N vatalanib Chemical compound C1=CC(Cl)=CC=C1NC(C1=CC=CC=C11)=NN=C1CC1=CC=NC=C1 YCOYDOIWSSHVCK-UHFFFAOYSA-N 0.000 description 1
- 229940065658 vidaza Drugs 0.000 description 1
- 208000009540 villous adenoma Diseases 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- 229960005212 vindesine sulfate Drugs 0.000 description 1
- NMDYYWFGPIMTKO-HBVLKOHWSA-N vinflunine Chemical compound C([C@@](C1=C(C2=CC=CC=C2N1)C1)(C2=C(OC)C=C3N(C)[C@@H]4[C@@]5(C3=C2)CCN2CC=C[C@]([C@@H]52)([C@H]([C@]4(O)C(=O)OC)OC(C)=O)CC)C(=O)OC)[C@H]2C[C@@H](C(C)(F)F)CN1C2 NMDYYWFGPIMTKO-HBVLKOHWSA-N 0.000 description 1
- 229960000922 vinflunine Drugs 0.000 description 1
- GBABOYUKABKIAF-IELIFDKJSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-IELIFDKJSA-N 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- CILBMBUYJCWATM-PYGJLNRPSA-N vinorelbine ditartrate Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O.OC(=O)[C@H](O)[C@@H](O)C(O)=O.C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC CILBMBUYJCWATM-PYGJLNRPSA-N 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 210000003905 vulva Anatomy 0.000 description 1
- 229940053867 xeloda Drugs 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 229940053890 zanosar Drugs 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011592 zinc chloride Substances 0.000 description 1
- 235000005074 zinc chloride Nutrition 0.000 description 1
- 229940072168 zocor Drugs 0.000 description 1
- FBTUMDXHSRTGRV-ALTNURHMSA-N zorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(\C)=N\NC(=O)C=1C=CC=CC=1)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 FBTUMDXHSRTGRV-ALTNURHMSA-N 0.000 description 1
- 229960000641 zorubicin Drugs 0.000 description 1
- 229950005752 zosuquidar Drugs 0.000 description 1
- IHOVFYSQUDPMCN-QKUIIBHLSA-N zosuquidar Chemical compound C([C@H](COC=1C2=CC=CN=C2C=CC=1)O)N(CC1)CCN1C1C2=CC=CC=C2C2C(F)(F)C2C2=CC=CC=C12 IHOVFYSQUDPMCN-QKUIIBHLSA-N 0.000 description 1
- 229940088909 zyloprim Drugs 0.000 description 1
- 239000002076 α-tocopherol Substances 0.000 description 1
- 235000004835 α-tocopherol Nutrition 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6558—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system
- C07F9/65586—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system at least one of the hetero rings does not contain nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6564—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms
- C07F9/6568—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms having phosphorus atoms as the only ring hetero atoms
- C07F9/65685—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms having phosphorus atoms as the only ring hetero atoms the ring phosphorus atom being part of a phosphine oxide or thioxide
Definitions
- JAK Janus kinase
- JAK family members are non-receptor tyrosine kinases that associate with many hematopoietin cytokines, receptor tyrosine kinases and GPCR' s.
- JAKl- /- mice were found to be developmentally similar to the JAK1+/+ although they weighed 40% less than the wild-type and failed to nurse at birth. These pups were not viable and died within 24 hours of birth (Meraz et al Cell, 1998, 373-383). JAKl deficiency led to reduced number of thymocytes, pre-B cells and mature T and B lymphocytes.
- TYK2(-/-) mice are viable, demonstrating subtle defects in their response to IFN- ⁇ / ⁇ and IL-10 and profound defects to the response of IL- 12 and LPS.
- the breast cancer susceptibility protein acts as a tumor suppressor and contributes to cell proliferation, cycle regulation, as well as DNA damage and repair.
- BRCAl (- /-) mice develop normally but die by 7.5 days post embryo suggesting a key role of BRCAl for development. Mice in which the BRCAl protein was overexpressed led to inhibition of cell growth and sensitized cells to cytotoxic reagents.
- Du- 145 Gao FEBS Letters 2001, 488, 179-184
- enhanced expression of BRCAl was found to correlate with constitutive activation of STAT3 as well as activation of JAKl and JAK2.
- antisense oligonucleotides selective for STAT3 led to significant inhibition of cell proliferation and apoptosis in Du- 145 cells. This data supports the potential utility of JAKl and JAK2 inhibitors in the treatment of prostate cancer.
- ThI cells are thought to be involved with the "delayed-type hypersensitivity responses" which secrete IL-2, IFN- ⁇ , and TNF- ⁇ and signal through the JAK2/TYK2-STAT4 pathway.
- STAT6 (-/-) mice were protected from AHR when challenged with environmental antigens and showed no increase in IgE levels or the quantity of mucous containing cells.
- JAK2 is a cytoplasmic protein-tyrosine kinase that catalyzes the transfer of the gamma-phosphate group of adenosine triphosphate to the hydroxyl groups of specific tyrosine residues in signal transduction molecules. JAK2 mediates signaling downstream of cytokine receptors after ligand-induced autophosphorylation of both receptor and enzyme.
- the main downstream effectors of JAK2 are a family of transcription factors known as signal transducers and activators of transcription (STAT) proteins. Studies have disclosed an association between an activating JAK2 mutation (JAK2V617F) and myleoproliferative disorders.
- the myeloproliferative disorders are clonal stem cell diseases characterized by an expansion of morphologically mature granulocyte, erythroid, megakaryocyte, or monocyte lineage cells.
- Myeloproliferative disorders include polycythemia vera (PV), essential thrombocythemia (ET), myeloid metaplasia with myelofibrosis (MMM), chronic myelogenous leukemia (CML), chronic myelomonocytic leukemia (CMML), hypereosinophilic syndrome (HES), juvenile myelomonocytic leukemia (JMML) and systemic mast cell disease (SMCD). It has been suggested that abnormalties in signal transduction mechanisms, including constitutive activation of protein tyrosine kinases, initiate MPD.
- JAK3 associates with the common gamma chain of the extracellular receptors for the following interleukins: IL-2, IL-4, IL-7, IL-9 and IL-15.
- a JAK3 deficiency is associated with an immune compromised (SCID) phenotype in both rodents and humans.
- SCID immune compromised
- the SCID phenotype of JAK3 -/- mammals and the lymphoid cell specific expression of JAK3 are two favorable attributes of a target for an immune suppressant. Data suggests that inhibitors of JAK3 could impede T-cell activation and prevent rejection of grafts following transplant surgery, or to provide therapeutic benefit to patients suffering autoimmune disorders.
- PDKl signalling regulates multiple critical steps in angiogenesis.
- Inhibitors of the activity of PDKl are thus useful in the treatment of cancer, in particular cancers associated with deregulated activity of the PTEN/PI3K pathway including, but not limited to PTEN loss of function mutations and receptor tyrosine kinase gain of function mutations.
- the instant invention provides for compounds that inhibit mammalian JAK kinases (such as JAKl, JAK2, JAK3 and TYK2) and PDKl.
- the invention also provides for compositions comprising such inhibitory compounds and methods of inhibiting the activity of JAKl, JAK2, JAK3 TYK2 and PDKl by administering the compound to a patient in need of treatment for myeloproliferative disorders or cancer.
- One embodiment of the invention is illustrated by a compound of formula I, and the pharmaceutically acceptable salts and stereoisomers thereof:
- the instant invention provides for compounds that inhibit the four known mammalian JAK kinases (JAKl , JAK2, JAK3 and TYK2) and PDKl .
- the invention also provides for compositions comprising such inhibitory compounds and methods of inhibiting the activity of JAKl, JAK2, JAK3, TYK2 and PDKl by administering the compound to a patient in need of treatment for myeloproliferative disorders or cancer.
- One embodiment of the invention is illustrated by a compound of formula I:
- W is N or CR 4 ;
- Y is N or CR 3 ;
- Z is N or CR 2 ;
- R 1 is hydrogen, halo, cyano or Cl -3 alkyl, wherein said alkyl group is optionally substituted with one to three halo;
- R 2 is hydrogen, halo, cyano or C 1-3 alkyl, wherein said alkyl group is optionally substituted with one to three halo;
- R 6 is hydrogen or C 1-6 alkyl, wherein said alkyl group is optionally substituted with SO m , - NR 8 R 9 , hydroxyl or -OR 8 ;
- R 7 is hydrogen or C 1-6 alkyl, wherein said alkyl group is optionally substituted with SO m , -
- NR 8 R 9 Or -OR 8 ; or R 6 and R 7 can be taken together with the nitrogen atom to which they are attached to form a 4 to 8 memebered heterocyclic or heteroaryl ring (wherein the heterocyclic or heteroaryl ring may further incorporate another heteroatom selected from the group consisting of O, SO m and NR ), wherein said heterocyclic or heteroaryl ring is optionally substituted on either the carbon or heteroatom with one to three substituents independently selected from the group consisting of C 1-6 alkyl, -C(O)Ci -6 alkyl, hydroxyl and halo; R is hydrogen or C 1-6 alkyl;
- R 9 is hydrogen or Ci -6 alkyl
- R 1 ' is hydrogen, C 3-6 cycloalkyl (which is optionally substituted with one to three substituents independently selected from the group consisting of fluoro, hydroxyl and(Ci -6 alkyl)OR 12 ), heterocyclyl (which is optionally substituted on either the carbon or heteroatom with one to three substituents independently selected from the group consisting of halo, oxo, Ci-6 alkyl, OR 12 ,
- heteroaryl which is optionally substituted on either the carbon or heteroatom with one to three substituents independently selected from the group consisting of C 3-6 cycloalkyl,
- R 12 is hydrogen or Ci -6 alkyl, wherein said alkyl group is optionally substituted with one to four substituents independently selected from the group consisting of halo, hydroxyl, cyano, SO m R ,
- R 1 is hydrogen or Ci_3 alkyl. In a class of the invention, R 1 is hydrogen,
- R 2 is hydrogen or Ci_3 alkyl. In a class of the invention, R 2 is hydrogen.
- R3 is SO m (Ci-3 alkyl).
- R 4 is C ⁇ . ⁇ alkyl, CH 2 OR 12 ,
- R 4 is CH 2 OR 12
- R 12 is C 1-6 alkyl, wherein said alkyl group is optionally substituted with hydroxyl.
- R 5 is aryl, wherein said aryl group is substituted with one to three substituents independently selected from the group consisting of halo, R 1 , R 11 and SO m R .
- R 5 is aryl, wherein said aryl group is substituted with one to three substituents independently selected from the group consisting of halo and R 1 .
- R 5 is phenyl, wherein said phenyl group is substituted with one to three substituents independently selected from the group consisting of halo and R 12
- R 1 is hydrogen or Ci -6 alkyl, wherein said alkyl group is optionally substituted with one to two hydroxyl.
- R 11 is heterocyclyl (which is optionally substituted on either the carbon or heteroatom with one to three substituents independently selected from the group consisting of oxo and OR 12 ).
- R 11 is heterocyclyl (which is optionally substituted with one to two oxo or OR 12 ), and R 12 is hydrogen.
- Reference to the preferred embodiments set forth above is meant to include all combinations of particular and preferred groups unless stated otherwise.
- compositions which is comprised of a compound of Formula I as described above and a pharmaceutically acceptable carrier.
- the invention is also contemplated to encompass a pharmaceutical composition which is comprised of a pharmaceutically acceptable carrier and any of the compounds specifically disclosed in the present application.
- the compounds of the present invention may have asymmetric centers, chiral axes, and chiral planes (as described in: E.L. Eliel and S.H. Wilen, Stereochemistry of Carbon Compounds, John Wiley & Sons, New York, 1994, pages 1119-1190), and occur as racemates, racemic mixtures, and as individual diastereomers, with all possible isomers and mixtures thereof, including optical isomers, all such stereoisomers being included in the present invention.
- heteroaryl groups such as imidazoles, exist as a mixture of 1H/2H tautomers.
- the tautomeric forms of these heteroaryl moieties are also within the scope of the instant invention.
- any variable e.g. Rl 1, etc.
- Rl 1, etc. its definition on each occurrence is independent at every other occurrence.
- combinations of substituents and variables are permissible only if such combinations result in stable compounds.
- Lines drawn into the ring systems from substituents represent that the indicated bond may be attached to any of the substitutable ring atoms. If the ring system is bicyclic, it is intended that the bond be attached to any of the suitable atoms on either ring of the bicyclic moiety.
- one or more silicon (Si) atoms can be incorporated into the compounds of the instant invention in place of one or more carbon atoms by one of ordinary skill in the art to provide compounds that are chemically stable and that can be readily synthesized by techniques known in the art from readily available starting materials.
- Carbon and silicon differ in their covalent radius leading to differences in bond distance and the steric arrangement when comparing analogous C-element and Si-element bonds. These differences lead to subtle changes in the size and shape of silicon-containing compounds when compared to carbon.
- size and shape differences can lead to subtle or dramatic changes in potency, solubility, lack of off target activity, packaging properties, and so on.
- substituents and substitution patterns on the compounds of the instant invention can be selected by one of ordinary skill in the art to provide compounds that are chemically stable and that can be readily synthesized by techniques known in the art, as well as those methods set forth below, from readily available starting materials. If a substituent is itself substituted with more than one group, it is understood that these multiple groups may be on the same carbon or on different carbons, so long as a stable structure results.
- the phrase "optionally substituted with one or more substituents” should be taken to be equivalent to the phrase “optionally substituted with at least one substituent” and in such cases the preferred embodiment will have from zero to four substituents, and the more preferred embodiment will have from zero to three substituents.
- alkyl is intended to include both branched and straight-chain saturated aliphatic hydrocarbon groups having the specified number of carbon atoms.
- Ci-CiO as in “(Ci-Cio)alkyl” is defined to include groups having 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10 carbons in a linear or branched arrange-ment.
- (Ci-Ci ⁇ )alkyl specifically includes methyl, ethyl, rc-propyl, /-propyl, n-butyl, r-butyl, /-butyl, pentyl, hexyl, heptyl, octyl, nonyl, decyl, and so on.
- haloalkyl means an alkyl radical as defined above, unless otherwise specified, that is substituted with one to five, preferably one to three halogen. Representative examples include, but are not limited to trifluoromethyl, dichloroethyl, and the like.
- alkenyl refers to a non-aromatic hydrocarbon radical, straight or branched, containing from 2 to 10 carbon atoms and at least 1 carbon to carbon double bond. Preferably 1 carbon to carbon double bond is present, and up to 4 non-aromatic carbon- carbon double bonds may be present.
- C2-C6 alkenyl means an alkenyl radical having from 2 to 6 carbon atoms.
- Alkenyl groups include ethenyl, propenyl, butenyl and cyclohexenyl.
- alkynyl refers to a non-aromatic hydrocarbon radical, straight or branched, containing from 2 to 10 carbon atoms and at least 1 carbon to carbon triple bond. Preferably 1 carbon to carbon triple bond is present.
- C2-C6 alkynyl means an alkynyl radical having from 2 to 6 carbon atoms.
- Alkynyl groups include ethynyl, propynyl, butynyl, and the like. As described above with respect to alkyl, the straight or branched portion of the alkynyl group may be substituted if a substituted alkynyl group is indicated.
- cycloalkyl means a monocyclic saturated aliphatic hydrocarbon group having the specified number of carbon atoms.
- cycloalkyl includes cyclopropyl, methyl-cyclopropyl, 2,2-dimethyl-cyclobutyl, 2-ethyl-cyclopentyl, cyclohexyl, and so on.
- Alkoxy represents either a cyclic or non-cyclic alkyl group of indicated number of carbon atoms attached through an oxygen bridge. "Alkoxy” therefore encompasses the definitions of alkyl and cycloalkyl above.
- aryl is intended to mean any stable monocyclic or bicyclic carbon ring of up to 7 atoms in each ring, wherein at least one ring is aromatic.
- aryl elements include phenyl, naphthyl, tetrahydro-naphthyl, indanyl and biphenyl.
- the aryl substituent is bicyclic and one ring is non-aromatic, it is understood that attachment is via the aromatic ring.
- heteroaryl represents a stable monocyclic or bicyclic ring of up to 7 atoms in each ring, wherein at least one ring is aromatic and contains from 1 to 4 heteroatoms selected from the group consisting of O, N and S.
- Heteroaryl groups within the scope of this definition include but are not limited to: acridinyl, carbazolyl, cinnolinyl, quinoxalinyl, pyrrazolyl, indolyl, benzotriazolyl, furanyl, thienyl, benzothienyl, benzofuranyl, quinolinyl, isoquinolinyl, oxazolyl, isoxazolyl, indolyl, pyrazinyl, pyridazinyl, pyridinyl, pyrimidinyl, pyrrolyl, tetrahydroquinoline.
- heterocycle As with the definition of heterocycle below,
- heteroaryl is also understood to include the N-oxide derivative of any nitrogen-containing heteroaryl. hi cases where the heteroaryl substituent is bicyclic and one ring is non-aromatic or contains no heteroatoms, it is understood that attachment is via the aromatic ring or via the heteroatom containing ring, respectively.
- Such heteraoaryl moieties include but are not limited to: 2-benzimidazolyl, 2-quinolinyl, 3-quinolinyl, 4-quinolinyl, 1 -isoquinolinyl, 3 -isoquinolinyl, 4-isoquinolinyl, dihydroimidazopyrazinyl and dihydrooxozolopyridinyl.
- heterocycle or “heterocyclyl” as used herein is intended to mean a 3- to 10-membered aromatic or nonaromatic heterocycle containing from 1 to 4 heteroatoms selected from the group consisting of O, N and S, and includes bicyclic groups.
- Heterocyclyl therefore includes the above mentioned heteroaryls, as well as dihydro and tetrathydro analogs thereof.
- heterocyclyl include, but are not limited to the following: azabicyclohexyl, azaphosphinyl, azaspiroheptyl, benzoimidazolyl, benzoimidazolonyl, benzofuranyl, benzofurazanyl, benzopyrazolyl, benzotriazolyl, benzothiophenyl, benzoxazolyl, carbazolyl, carbolinyl, cinnolinyl, dioxidothiomorpholinyl, furanyl, imidazolyl, indolinyl, indolyl, indolazinyl, indazolyl, isobenzofuranyl, isoindolyl, isoquinolyl, isothiazolyl, isoxazolyl, naphthpyridinyl, oxadiazolyl, oxazolyl, oxazoline, isoxazoline, oxetanyl, oxetanyl
- halo or halogen as used herein is intended to include chloro (Cl), fluoro (F), bromo (Br) and iodo (I).
- the instant invention is the free form of compounds of the instant invention, as well as the pharmaceutically acceptable salts and stereoisomers thereof.
- Some of the isolated specific compounds exemplified herein are the protonated salts of amine compounds.
- the term "free form” refers to the amine compounds in non-salt form.
- the encompassed pharmaceutically acceptable salts not only include the isolated salts exemplified for the specific compounds described herein, but also all the typical pharmaceutically acceptable salts of the free form of compounds of the instant invention.
- the free form of the specific salt compounds described may be isolated using techniques known in the art.
- the free form may be regenerated by treating the salt with a suitable dilute aqueous base solution such as dilute aqueous NaOH, potassium carbonate, ammonia and sodium bicarbonate.
- a suitable dilute aqueous base solution such as dilute aqueous NaOH, potassium carbonate, ammonia and sodium bicarbonate.
- the free forms may differ from their respective salt forms somewhat in certain physical properties, such as solubility in polar solvents, but the acid and base salts are otherwise pharmaceutically equivalent to their respective free forms for purposes of the invention.
- the pharmaceutically acceptable salts of the instant compounds can be synthesized from the compounds of this invention which contain a basic or acidic moiety by conventional chemical methods.
- the salts of the basic compounds are prepared either by ion exchange chromatography or by reacting the free base with stoichiometric amounts or with an excess of the desired salt-forming inorganic or organic acid in a suitable solvent or various combinations of solvents.
- the salts of the acidic compounds are formed by reactions with the appropriate inorganic or organic base.
- pharmaceutically acceptable salts of the compounds of this invention include the conventional non-toxic salts of the compounds of this invention as formed by reacting a basic instant compound with an inorganic or organic acid.
- conventional non-toxic salts include those derived from inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, nitric and the like, as well as salts prepared from organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic, tartaric, citric, ascorbic, pamoic, maleic, hydroxymaleic, phenylacetic, glutamic, benzoic, salicylic, sulfanilic, 2-acetoxy- benzoic, fumaric, toluenesulfonic, methanesulfonic, ethane disulfonic, oxalic, isethionic, trifluoroacetic (TFA) and the like.
- inorganic acids such as hydroch
- suitable “pharmaceutically acceptable salts” refers to salts prepared form pharmaceutically acceptable non-toxic bases including inorganic bases and organic bases.
- Salts derived from inorganic bases include aluminum, ammonium, calcium, copper, ferric, ferrous, lithium, magnesium, manganic salts, manganous, potassium, sodium, zinc and the like. Particularly preferred are the ammonium, calcium, magnesium, potassium and sodium salts.
- Salts derived from pharmaceutically acceptable organic non-toxic bases include salts of primary, secondary and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines and basic ion exchange resins, such as arginine, betaine caffeine, choline, N ⁇ -dibenzylethylenediamine, diethylamin, 2-diethylaminoethanol, 2-dimethylaminoethanol, ethanolamine, ethylenediamine, N-ethylmorpholine, N-ethylpiperidine, glucamine, glucosamine, histidine, hydrabamine, isopropylamine, lysine, methylglucamine, morpholine, piperazine, piperidine, polyamine resins, procaine, purines, theobromine, triethylamine, trimethylamine tripropylamine, tromethamine and the like.
- basic ion exchange resins such as arginine, betaine caffeine
- the compounds of the present invention are inhibitors of JAK 1, JAK2, JAK 3, TYK2 and PDKl, and are therefore useful to treat or prevent myeloproliferative disorders or cancer in mammals, preferably humans.
- An embodiment of the invention provides a method for inhibiting JAKl tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- An embodiment of the invention provides a method for inhibiting JAK2 tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- An embodiment of the invention provides a method for inhibiting wild type or mutant JAK2 tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- An embodiment of the invention provides a method for inhibiting JAK2 V617F tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- the compounds, compositions and methods provided herein are particularly deemed useful for the treatment of myeloproliferative disorder(s).
- Myeloproliferative disorders that may be treated include polycythemia vera (PV), essential thrombocythemia (ET), myeloid metaplasia with myelofibrosis (MMM), chronic myelogenous leukemia (CML), myelomonocytic leukemia (CMML), hypereosinophilic syndrome (HES), juvenile myelomonocytic leukemia (JMML), and systemic mast cell disease (SMCD).
- PV polycythemia vera
- E essential thrombocythemia
- MMM myeloid metaplasia with myelofibrosis
- CML chronic myelogenous leukemia
- CMML myelomonocytic leukemia
- HES hypereosinophilic syndrome
- JMML juvenile myelomonocytic leukemia
- SMCD systemic mast cell disease
- JAK2 JAK2-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl-N-phenyl
- Cancers that may be treated by the compounds, compositions and methods of the invention include, but are not limited to: Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma; Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma; Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinom
- the compounds, compositions and methods of the invention may also be useful in treating the following disease states: keloids and psoriasis.
- Cancers that may be treated by the compounds, compositions and methods of the invention include, but are not limited to: breast, prostate, colon, colorectal, lung, brain, testicular, stomach, pancrease, skin, small intestine, large intestine, throat, head and neck, oral, bone, liver, bladder, kidney, thyroid and blood. Cancers that may be treated by the compounds, compositions and methods of the invention include: breast, prostate, colon, ovarian, colorectal and lung (non-small cell lung).
- Cancers that may be treated by the compounds, compositions and methods of the invention include: breast, colon, colorectal and lung.
- Cancers that may be treated by the compounds, compositions and methods of the invention include: lymphoma and leukemia.
- the compounds of the instant invention are also inhibitors of the activity of PDKl and are thus useful in the treatment of cancer, in particular cancers associated with deregulated activity of the PTEN/PI3K pathway including, but not limited to PTEN loss of function mutations and receptor tyrosine kinase gain of function mutations.
- cancers include, but are not limited to, ovarian, pancreatic, breast and prostate cancer, as well as cancers (including glioblastoma) where the tumor suppressor PTEN is mutated. See, Feldman, Richard I., et al., "Novel Small Molecule Inhibitors of 3-Phosphoinositide-dependent Kinase-1," The Journal of Biological Chemistry, Vol. 280, No. 20, Issue of May 20, pp. 19867-19874, 2005.
- PDKl signaling regulates multiple critical steps in angiogenesis. See, Mora, Alfonso et al., "PDKl, the master regulator of AGC kinase signal transduction," Seminars in Cell & Developmental Biology 15 (2004) 161-170.
- the utility of angiogenesis inhibitors in the treatment of cancer is known in the literature, see J. Rak et al. Cancer Research, 55:4575-4580, 1995 and Dredge et al., Expert Opin. Biol. Ther. (2002) 2(8):953-966, for example.
- the role of angiogenesis in cancer has been shown in numerous types of cancer and tissues: breast carcinoma (G. Gasparini and A.L. Harris, J Clin. Oncol, 1995, 13:765-782; M.
- bladder carcinomas AJ. Dickinson et al., Br. J. Urol., 1994, 74:762-766
- colon carcinomas L.M. Ellis et al., Surgery, 1996, 120(5):871-878
- oral cavity tumors J.K. Williams et al., Am. J. Surg., 1994, 168:373-380.
- cancers include, advanced tumors, hairy cell leukemia, melanoma, advanced head and neck, metastatic renal cell, non-Hodgkin's lymphoma, metastatic breast, breast adenocarcinoma, advanced melanoma, pancreatic, gastric, glioblastoma, lung, ovarian, non-small cell lung, prostate, small cell lung, renal cell carcinoma, various solid tumors, multiple myeloma, metastatic prostate, malignant glioma, renal cancer, lymphoma, refractory metastatic disease, refractory multiple myeloma, cervical cancer, Kaposi's sarcoma, recurrent anaplastic glioma, and metastatic colon cancer (Dredge et al., Expert Opin.
- the PDKl inhibitors disclosed in the instant application are also useful in the treatment of these angiogenesis related cancers.
- Tumors which have undergone neovascularization show an increased potential for metastasis, hi fact, angiogenesis is essential for tumor growth and metastasis.
- Tljie PDKl inhibitors disclosed in the present application are therefore also useful to prevent or decrease tumor cell metastasis.
- a method of treating or preventing a disease in which angiogenesis is implicated which is comprised of administering to a mammal in need of such treatment a therapeutically effective amount of a compound of the present invention.
- Ocular neovascular diseases are an example of conditions where much of the resulting tissue damage can be attributed to aberrant infiltration of blood vessels in the eye (see WO 00/30651, published 2 June 2000).
- the undesireable infiltration can be triggered by ischemic retinopathy, such as that resulting from diabetic retinopathy, retinopathy of prematurity, retinal vein occlusions, etc., or by degenerative diseases, such as the choroidal neovascularization observed in age-related macular degeneration.
- ischemic retinopathy such as that resulting from diabetic retinopathy, retinopathy of prematurity, retinal vein occlusions, etc.
- degenerative diseases such as the choroidal neovascularization observed in age-related macular degeneration.
- Inhibiting the growth of blood vessels by administration of the present compounds would therefore prevent the infiltration of blood vessels and prevent or treat diseases where angiogenesis is implicated, such as ocular diseases like retinal vascularization, diabetic retinopathy, age-related macular degeneration, and the like.
- a method of treating or preventing a non-malignant disease in which angiogenesis is implicated including but not limited to: ocular diseases (such as, retinal vascularization, diabetic retinopathy and age-related macular degeneration), atherosclerosis, arthritis, psoriasis, obesity and Alzheimer's disease (Dredge et al., Expert Opin. Biol. Ther. (2002) 2(8):953-966).
- a method of treating or preventing a disease in which angiogenesis is implicated includes: ocular diseases (such as, retinal vascularization, diabetic retinopathy and age-related macular degeneration), atherosclerosis, arthritis and psoriasis.
- hyperproliferative disorders such as restenosis, inflammation, autoimmune diseases and allergy/asthma.
- hypoinsulinism is a method of treating hypoinsulinism.
- An embodiment of the invention provides a method for inhibiting JAK3 tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- An embodiment of the invention provides a method for inhibiting TYK2 tyrosine kinase, comprising administering to the mammal a therapeutically effective amount of any of the compounds or any of the pharmaceutical compositions described above.
- Exemplifying the invention is the use of any of the compounds described above in the preparation of a medicament for the treatment and/or prevention of osteoporosis in a mammal in need thereof. Still further exemplifying the invention is the use of any of the compounds described above in the preparation of a medicament for the treatment and/or prevention of: bone loss, bone resorption, bone fractures, metastatic bone disease and/or disorders related to cathepsin functioning.
- the compounds of this invention may be administered to mammals, including humans, either alone or, in combination with pharmaceutically acceptable carriers, excipients or diluents, in a pharmaceutical composition, according to standard pharmaceutical practice.
- the compounds can be administered orally or parenterally, including the intravenous, intramuscular, intraperitoneal, subcutaneous, rectal and topical routes of administration.
- compositions containing the active ingredient may be in a form suitable for oral use, for example, as tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsions, hard or soft capsules, or syrups or elixirs.
- compositions intended for oral use may be prepared according to any method known to the art for the manufacture of pharmaceutical compositions and such compositions may contain one or more agents selected from the group consisting of sweetening agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations.
- Tablets contain the active ingredient in admixture with non-toxic pharmaceutically acceptable excipients which are suitable for the manufacture of tablets.
- excipients may be for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, for example, microcrystalline cellulose, sodium crosscarmellose, corn starch, or alginic acid; binding agents, for example starch, gelatin, polyvinyl-pyrrolidone or acacia, and lubricating agents, for example, magnesium stearate, stearic acid or talc.
- the tablets may be uncoated or they may be coated by known techniques to mask the unpleasant taste of the drug or delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period.
- a water soluble taste masking material such as hydroxypropylmethyl-cellulose or hydroxypropylcellulose, or a time delay material such as ethyl cellulose, cellulose acetate buryrate may be employed.
- Formulations for oral use may also be presented as hard gelatin capsules wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules wherein the active ingredient is mixed with water soluble carrier such as polyethyleneglycol or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- an inert solid diluent for example, calcium carbonate, calcium phosphate or kaolin
- water soluble carrier such as polyethyleneglycol or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- Aqueous suspensions contain the active material in admixture with excipients suitable for the manufacture of aqueous suspensions.
- excipients are suspending agents, for example sodium carboxymethylcellulose, methylcellulose, hydroxypropylmethyl-cellulose, sodium alginate, polyvinyl-pyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents may be a naturally-occurring phosphatide, for example lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example heptadecaethylene- oxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan
- the aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents, such as sucrose, saccharin or aspartame.
- preservatives for example ethyl, or n-propyl p-hydroxybenzoate
- coloring agents for example ethyl, or n-propyl p-hydroxybenzoate
- flavoring agents such as sucrose, saccharin or aspartame.
- sweetening agents such as sucrose, saccharin or aspartame.
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example arachis oil, olive oil, sesame oil or coconut oil, or in mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent, for example beeswax, hard paraffin or cetyl alcohol.
- Sweetening agents such as those set forth above, and flavoring agents may be added to provide a palatable oral preparation.
- These compositions may be preserved by the addition of an anti-oxidant such as butylated hydroxyanisol or alpha-tocopherol.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives.
- Suitable dispersing or wetting agents and suspending agents are exemplified by those already mentioned above. Additional excipients, for example sweetening, flavoring and coloring agents, may also be present. These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid.
- the pharmaceutical compositions of the invention may also be in the form of an oil-in-water emulsion.
- the oily phase may be a vegetable oil, for example olive oil or arachis oil, or a mineral oil, for example liquid paraffin or mixtures of these.
- Suitable emulsifying agents may be naturally-occurring phosphatides, for example soy bean lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example sorbitan monooleate, and condensation products of the said partial esters with ethylene oxide, for example polyoxyethylene sorbitan monooleate.
- the emulsions may also contain sweetening, flavouring agents, preservatives and antioxidants.
- Syrups and elixirs may be formulated with sweetening agents, for example glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative, flavoring and coloring agents and antioxidant.
- sweetening agents for example glycerol, propylene glycol, sorbitol or sucrose.
- Such formulations may also contain a demulcent, a preservative, flavoring and coloring agents and antioxidant.
- compositions may be in the form of sterile injectable aqueous solutions.
- acceptable vehicles and solvents that may be employed are water, Ringer's solution and isotonic sodium chloride solution.
- the sterile injectable preparation may also be a sterile injectable oil-in-water microemulsion where the active ingredient is dissolved in the oily phase.
- the active ingredient may be first dissolved in a mixture of soybean oil and lecithin. The oil solution then introduced into a water and glycerol mixture and processed to form a microemulation.
- the injectable solutions or microemulsions may be introduced into a patient's blood-stream by local bolus injection.
- a continuous intravenous delivery device may be utilized.
- An example of such a device is the Deltec CADD- PLUSTM model 5400 intravenous pump.
- the pharmaceutical compositions may be in the form of a sterile injectable aqueous or oleagenous suspension for intramuscular and subcutaneous administration.
- This suspension may be formulated according to the known art using those suitable dispersing or wetting agents and suspending agents which have been mentioned above.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally- acceptable diluent or solvent, for example as a solution in 1,3-butane diol.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- compositions can be prepared by mixing the drug with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at the rectal temperature and will therefore melt in the rectum to release the drug.
- suitable non-irritating excipient include cocoa butter, glycerinated gelatin, hydrogenated vegetable oils, mixtures of polyethylene glycols of various molecular weights and fatty acid esters of polyethylene glycol.
- topical application For topical use, creams, ointments, jellies, solutions or suspensions, etc., containing the compounds of the instant invention are employed. (For purposes of this application, topical application shall include mouth washes and gargles.)
- the compounds for the present invention can be administered in intranasal form via topical use of suitable intranasal vehicles and delivery devices, or via transdermal routes, using those forms of transdermal skin patches well known to those of ordinary skill in the art.
- the dosage administration will, of course, be continuous rather than intermittent throughout the dosage regimen.
- Compounds of the present invention may also be delivered as a suppository employing bases such as cocoa butter, glycerinated gelatin, hydrogenated vegetable oils, mixtures of polyethylene glycols of various molecular weights and fatty acid esters of polyethylene glycol.
- the compounds of the present invention can also be administered in the form of liposome delivery systems, such as small unilamellar vesicles, large unilamellar vesicles and multilamellar vesicles.
- Liposomes can be formed from a variety of phospholipids, such as cholesterol, stearylamine or phosphatidylcholines.
- Compounds of the present invention may also be delivered by the use of monoclonal antibodies as individual carriers to which the compound molecules are coupled.
- the compounds of the present invention may also be coupled with soluble polymers as targetable drug carriers.
- Such polymers can include polyvinylpyrrolidone, pyran copolymer, polyhydroxypropylmethacrylamide-phenol, polyhydroxy-ethylaspartamide-phenol, or polyethyleneoxide-polylysine substituted with palmitoyl residues.
- the compounds of the present invention may be coupled to a class of biodegradable polymers useful in achieving controlled release of a drug, for example, polylactic acid, polyglycolic acid, copolymers of polyactic and polyglycolic acid, polyepsilon caprolactone, polyhydroxy butyric acid, polyorthoesters, polyacetals, polydihydropyrans, polycyanoacrylates and crosslinked or amphipathic block copolymers of hydrogels.
- a composition according to this invention is administered into a human subject, the daily dosage will normally be determined by the prescribing physician with the dosage generally varying according to the age, weight, and response of the individual patient, as well as the severity of the patient's symptoms.
- a suitable amount of an inhibitor of JAK2 is administered to a mammal undergoing treatment for cancer.
- Administration occurs in an amount of inhibitor of between about 0.1 mg/kg of body weight to about 60 mg/kg of body weight per day, or between 0.5 mg/kg of body weight to about 40 mg/kg of body weight per day.
- Another therapeutic dosage that comprises the instant composition includes from about 0.01 mg to about 1000 mg of inhibitor of JAK2. In another embodiment, the dosage comprises from about 1 mg to about 1000 mg of inhibitor of JAK2.
- the instant compounds are also useful in combination with therapeutic, chemotherapeutic and anti-cancer agents. Combinations of the presently disclosed compounds with therapeutic, chemotherapeutic and anti-cancer agents are within the scope of the invention.
- agents include the following: estrogen receptor modulators, androgen receptor modulators, retinoid receptor modulators, cytotoxic/cytostatic agents, antiproliferative agents, prenyl-protein transferase inhibitors, HMG-CoA reductase inhibitors and other angiogenesis inhibitors, HIV protease inhibitors, reverse transcriptase inhibitors, inhibitors of cell proliferation and survival signaling, bisphosphonates, aromatase inhibitors, siRNA therapeutics, ⁇ -secretase inhibitors, agents that interfere with receptor tyrosine kinases (RTKs) and agents that interfere with cell cycle checkpoints.
- RTKs receptor tyrosine kinases
- Estrogen receptor modulators refers to compounds that interfere with or inhibit the binding of estrogen to the receptor, regardless of mechanism. Examples of estrogen receptor modulators include, but are not limited to, tamoxifen, raloxifene, idoxifene, LY353381,
- Androgen receptor modulators refers to compounds which interfere or inhibit the binding of androgens to the receptor, regardless of mechanism.
- Examples of androgen receptor modulators include finasteride and other 5 ⁇ -reductase inhibitors, nilutamide, flutamide, bicalutamide, liarozole, and abiraterone acetate.
- Retinoid receptor modulators refers to compounds which interfere or inhibit the binding of retinoids to the receptor, regardless of mechanism.
- retinoid receptor modulators include bexarotene, tretinoin, 13-cis-retinoic acid, 9-cis-retinoic acid, ⁇ - difiuoromethylornithine, ILX23-7553, trans-N-(4'-hydroxyphenyl) retinamide, and N-4- carboxyphenyl retinamide.
- Cytotoxic/cytostatic agents refer to compounds which cause cell death or inhibit cell proliferation primarily by interfering directly with the cell's functioning or inhibit or interfere with cell myosis, including alkylating agents, tumor necrosis factors, intercalators, hypoxia activatable compounds, microtubule inhibitors/microtubule-stabilizing agents, inhibitors of mitotic kinesins, histone deacetylase inhibitors, inhibitors of kinases involved in mitotic progression, inhibitors of kinases involved in growth factor and cytokine signal transduction pathways, antimetabolites, biological response modifiers, hormonal/anti-hormonal therapeutic agents, haematopoietic growth factors, monoclonal antibody targeted therapeutic agents, topoisomerase inhibitors, proteosome inhibitors, ubiquitin ligase inhibitors, and aurora kinase inhibitors.
- cytotoxic/cytostatic agents include, but are not limited to, sertenef, cachectin, ifosfamide, tasonermin, lonidamine, carboplatin, altretamine, prednimustine, dibromodulcitol, ranimustine, fotemustine, nedaplatin, oxaliplatin, temozolomide, heptaplatin, estramustine, improsulfan tosilate, trofosfamide, nimustine, dibrospidium chloride, pumitepa, lobaplatin, satraplatin, profiromycin, cisplatin, irofulven, dexifosfamide, cis-aminedichloro(2- methyl-pyridine)platinum, benzylguanine, glufosfamide, GPXlOO, (trans, trans, trans)-bis-mu- (hexane-l,6-
- hypoxia activatable compound is tirapazamine.
- proteosome inhibitors include but are not limited to lactacystin and MLN-341 (Velcade).
- microtubule inhibitors/microtubule-stabilising agents include paclitaxel, vindesine sulfate, 3',4'-didehydro-4'-deoxy-8'-norvincaleukoblastine, docetaxol, rhizoxin, dolastatin, mivobulin isethionate, auristatin, cemadotin, RPRl 09881, BMS 184476, vinflunine, cryptophycin, 2,3,4,5,6-pentafluoro-N-(3-fluoro-4-methoxyphenyl) benzene sulfonamide, anhydrovinblastine, N,N-dimethyl-L-valyl-L-valyl-N-methyl-L-valyl-L-prolyl-L- proline-t-butylamide, TDX258, the epothilones (see for example U.S. Pat. Nos. 6,284,781 and 6,288,23
- topoisomerase inhibitors are topotecan, hycaptamine, irinotecan, rubitecan, 6-ethoxypropionyl-3',4'-O-exo-benzylidene-chartreusin, 9-methoxy-N,N- dimethyl-5-nitropyrazolo[3,4,5-kl]acridine-2-(6H) propanamine, l-amino-9-ethyl-5-fluoro-2,3- dihydro-9-hydroxy-4-methyl- 1 H, 12H-benzo[de]pyrano[3 ',4' :b,7]-indolizino[l ,2b]quinoline- 10,13(9H,15H)dione, lurtotecan, 7-[2-(N-isopropylamino)ethyl]-(20S)camptothecin, BNP1350, BNPII lOO, BN80915, BN80942, e
- inhibitors of mitotic kinesins are described in Publications WO03/039460, WO03/050064, WO03/050122, WO03/049527, WO03/049679, WO03/049678, WO04/039774, WO03/079973, WO03/099211, WO03/105855, WO03/106417, WO04/037171, WO04/058148, WO04/058700, WO04/126699, WO05/018638, WO05/019206, WO05/019205, WO05/018547, WO05/017190, US2005/0176776.
- inhibitors of mitotic kinesins include, but are not limited to inhibitors of KSP, inhibitors of MKLPl, inhibitors of CENP-E, inhibitors of MCAK and inhibitors of Rab6-KIFL.
- histone deacetylase inhibitors include, but are not limited to,
- “Inhibitors of kinases involved in mitotic progression” include, but are not limited to, inhibitors of aurora kinase, inhibitors of Polo-like kinases (PLK; in particular inhibitors of PLK-I), inhibitors of bub- 1 and inhibitors of bub-Rl.
- PLK Polo-like kinases
- An example of an "aurora kinase inhibitor” is VX-680.
- Antiproliferative agents includes antisense RNA and DNA oligonucleotides such as G3139, ODN698, RVASKRAS, GEM231, and INX3001, and antimetabolites such as enocitabine, carmofur, tegafur, pentostatin, doxifluridine, trimetrexate, fludarabine, capecitabine, galocitabine, cytarabine ocfosfate, fosteabine sodium hydrate, raltitrexed, paltitrexid, emitefur, tiazofurin, decitabine, nolatrexed, pemetrexed, nelzarabine, 2'-deoxy-2'-methylidenecytidine, 2'- fluoromethylene-2 ' -deoxycytidine, N- [5-(2,3 -dihydro-benzofuryl)sulfonyl] -N' -(3 ,4- dichlor
- HMG-CoA reductase inhibitors refers to inhibitors of 3-hydroxy-3- methylglutaryl-CoA reductase.
- HMG-CoA reductase inhibitors include but are not limited to lovastatin (MEV ACOR®; see U.S. Patent Nos. 4,231,938, 4,294,926 and 4,319,039), simvastatin (ZOCOR®; see U.S. Patent Nos. 4,444,784, 4,820,850 and 4,916,239), pravastatin (PRAVACHOL®; see U.S. Patent Nos.
- HMG-CoA reductase inhibitor as used herein includes all pharmaceutically acceptable lactone and open-acid forms (i.e., where the lactone ring is opened to form the free acid) as well as salt and ester forms of compounds which have HMG-CoA reductase inhibitory activity, and therefor the use of such salts, esters, open-acid and lactone forms is included within the scope of this invention.
- Prenyl-protein transferase inhibitor refers to a compound which inhibits any one or any combination of the prenyl-protein transferase enzymes, including farnesyl-protein transferase (FPTase), geranylgeranyl-protein transferase type I (GGPTase-I), and geranylgeranyl- protein transferase type-II (GGPTase-II, also called Rab GGPTase).
- FPTase farnesyl-protein transferase
- GGPTase-I geranylgeranyl-protein transferase type I
- GGPTase-II geranylgeranyl- protein transferase type-II
- prenyl-protein transferase inhibitors can be found in the following publications and patents: WO 96/30343, WO 97/18813, WO 97/21701, WO 97/23478, WO
- Angiogenesis inhibitors refers to compounds that inhibit the formation of new blood vessels, regardless of mechanism.
- angiogenesis inhibitors include, but are not limited to, tyrosine kinase inhibitors, such as inhibitors of the tyrosine kinase receptors FIt-I (VEGFRl) and FIk-I /KDR (VEGFR2), inhibitors of epidermal-derived, fibroblast-derived, or platelet derived growth factors, MMP (matrix metalloprotease) inhibitors, integrin blockers, interferon- ⁇ , interleukin-12, pentosan polysulfate, cyclooxygenase inhibitors, including nonsteroidal antiinflammatories (NSAIDs) like aspirin and ibuprofen as well as selective cyclooxy-genase-2 inhibitors like celecoxib and rofecoxib (PNAS, Vol.
- NSAIDs nonsteroidal antiinflammatories
- NSAIDs non
- steroidal anti-inflammatories such as corticosteroids, mineralocorticoids, dexamethasone, prednisone, prednisolone, methylpred, betamethasone), carboxyamidotriazole, combretastatin A-4, squalamine, 6-O-chloroacetyl-carbonyl)-fumagillol, thalidomide, angiostatin, troponin- 1, angiotensin II antagonists (see Fernandez et al., J. Lab. Clin. Med.
- agents that modulate or inhibit angiogenesis and may also be used in combination with the compounds of the instant invention include agents that modulate or inhibit the coagulation and fibrinolysis systems (see review in Clin. Chem. La. Med. 38:679-692 (2000)).
- agents that modulate or inhibit the coagulation and fibrinolysis pathways include, but are not limited to, heparin (see Thromb. Haemost. 80:10-23 (1998)), low molecular weight heparins and carboxypeptidase U inhibitors (also known as inhibitors of active thrombin activatable fibrinolysis inhibitor [TAFIa]) (see Thrombosis Res. 101 :329-354 (2001)).
- TAFIa inhibitors have been described in U.S. Ser. Nos. 60/310,927 (filed August 8, 2001) and 60/349,925 (filed January 18, 2002).
- Agents that interfere with cell cycle checkpoints refer to compounds that inhibit protein kinases that transduce cell cycle checkpoint signals, thereby sensitizing the cancer cell to DNA damaging agents.
- agents include inhibitors of ATR, ATM, the CHKl 1 and CHKl 2 kinases and cdk and cdc kinase inhibitors and are specifically exemplified by 7- hydroxystaurosporin, flavopiridol, CYC202 (Cyclacel) and BMS-387032.
- agents that interfere with receptor tyrosine kinases refer to compounds that inhibit RTKs and therefore mechanisms involved in oncogenesis and tumor progression.
- agents include inhibitors of c-Kit, Eph, PDGF, Flt3 and c-Met.
- Further agents include inhibitors of RTKs as described by Bume- Jensen and Hunter, Nature, 411 :355-365, 2001.
- “Inhibitors of cell proliferation and survival signalling pathway” refer to compounds that inhibit signal transduction cascades downstream of cell surface receptors. Such agents include inhibitors of serine/threonine kinases (including but not limited to inhibitors of Akt such as described in WO 02/083064, WO 02/083139, WO 02/083140, US 2004-0116432, WO 02/083138, US 2004-0102360, WO 03/086404, WO 03/086279, WO 03/086394, WO 03/084473, WO 03/086403, WO 2004/041162, WO 2004/096131, WO 2004/096129, WO 2004/096135, WO 2004/096130, WO 2005/100356, WO 2005/100344, US 2005/029941, US 2005/44294, US 2005/43361, 60/734188, 60/652737, 60/670469), inhibitors of Raf kinase (for example BAY-43-9006 ),
- NSAID As described above, the combinations with NSAID 's are directed to the use of NS AID's which are potent COX-2 inhibiting agents.
- an NSAID is potent if it possesses an IC 50 for the inhibition of COX-2 of 1 ⁇ M or less as measured by cell or microsomal assays.
- NSAID' s which are selective COX-2 inhibitors are defined as those which possess a specificity for inhibiting COX-2 over COX-I of at least 100 fold as measured by the ratio of IC50 for COX-2 over IC50 for COX-I evaluated by cell or microsomal assays.
- Such compounds include, but are not limited to those disclosed in U.S. Patent 5,474,995, U.S. Patent 5,861,419, U.S. Patent 6,001,843, U.S. Patent 6,020,343, U.S. Patent 5,409,944, U.S. Patent 5,436,265, U.S. Patent 5,536,752, U.S.
- Inhibitors of COX-2 that are particularly useful in the instant method of treatment are: 3-phenyl-4-(4-(methylsulfonyl)phenyl)-2-(5H)-furanone; and 5-chloro-3-(4-methylsulfonyl)phenyl-2-(2-methyl-5-pyridinyl)pyridine; or a pharmaceutically acceptable salt thereof.
- angiogenesis inhibitors include, but are not limited to, endostatin, ukrain, ranpirnase, IM862, 5-methoxy-4-[2-methyl-3-(3-methyl-2-butenyl)oxiranyl]- 1 -oxaspiro [2,5] oct-6-yl(chloroacetyl)carbamate, acety ldinanaline, 5 -amino- 1 - [ [3 , 5 -dichloro-4-(4- chlorobenzoyl)phenyl]methyl]- IH-1 ,2,3-triazole-4-carboxamide,CM 101, squalamine, combretastatin, RPI4610, NX31838, sulfated mannopentaose phosphate, 7,7-(carbonyl- bis[imino-N-methyl-4,2-pyi ⁇ olocarbonylimino[N-methyl-4,2-pyrrole]-carbon
- integrated circuit blockers refers to compounds which selectively antagonize, inhibit or counteract binding of a physiological ligand to the ⁇ y ⁇ 3 integrin, to compounds which selectively antagonize, inhibit or counteract binding of a physiological ligand to the ⁇ v ⁇ 5 integrin, to compounds which antagonize, inhibit or counteract binding of a physiological ligand to both the ⁇ v ⁇ 3 integrin and the ⁇ v ⁇ 5 integrin, and to compounds which antagonize, inhibit or counteract the activity of the particular integrin(s) expressed on capillary endothelial cells.
- the term also refers to antagonists of the ⁇ v ⁇ 6 > ⁇ *v ⁇ 8 > ⁇ i ⁇ i, ⁇ 2 ⁇ L «5 ⁇ L ⁇ 6 ⁇ l and ⁇ 6 ⁇ 4 integrins.
- the term also refers to antagonists of any combination of ⁇ v ⁇ 3, ⁇ v ⁇ 5, ⁇ v ⁇ 6> ⁇ v ⁇ 8, oq ⁇ l, «2 ⁇ l, ⁇ 5 ⁇ i, ⁇ l and ⁇ 6 ⁇ 4 integrins.
- tyrosine kinase inhibitors include N- (trifluoromethylphenyl)-5-methylisoxazol-4-carboxamide, 3-[(2,4-dimethylpyrrol-5- yl)methylidenyl)indolin-2-one, 17-(allylamino)- 17-demethoxygeldanamycin, 4-(3-chloro-4- fluorophenylamino)-7-methoxy-6-[3-(4-morpholinyl)propoxyl]quinazoline, N-(3-ethynylphenyl)- 6,7-bis(2-methoxyethoxy)-4-quinazolinamine, BIBX1382, 2,3,9,10,11,12-hexahydro- 10- (hydroxymethyl)- 10-hydroxy-9-methyl-9, 12-epoxy- lH-diindolo[l ,2,3-fg:3 ⁇ 2 ⁇ 1 '-kl]pyrrolo[3,4
- Combinations with compounds other than anti-cancer compounds are also encompassed in the instant methods.
- combinations of the instantly claimed compounds with PPAR- ⁇ (i.e., PPAR-gamma) agonists and PPAR- ⁇ (i.e., PPAR-delta) agonists are useful in the treatment of certain malingnancies.
- PPAR- ⁇ and PPAR- ⁇ are the nuclear peroxisome proliferator-activated receptors ⁇ and ⁇ .
- the expression of PPAR- ⁇ on endothelial cells and its involvement in angiogenesis has been reported in the literature (see J Cardiovasc. Pharmacol 1998; 31 :909-913; J Biol. Chem. 1999;274:9116-9121; Invest. Ophthalmol Vis.
- PPAR- ⁇ agonists and PPAR- ⁇ / ⁇ agonists include, but are not limited to, thiazolidinediones (such as DRF2725, CS-Ol 1, troglitazone, rosiglitazone, and pioglitazone), fenofibrate, gemfibrozil, clofibrate, GW2570, SB219994, AR-H039242, JTT-501, MCC-555, GW2331, GW409544, NN2344, KRP297, NP0110, DRF4158, NN622, GI262570, PNU182716, DRF552926, 2-[(5,7-dipropyl-3-trifluoromethyl-l,2-benzisoxazol-6-yl)oxy]-2-methylpropionic acid (disclosed in USSN 09/782,856), and 2(R)-7-(3-(2-chloro-4-(4-fluorophenoxy)
- Another embodiment of the instant invention is the use of the presently disclosed compounds in combination with gene therapy for the treatment of cancer.
- Gene therapy can be used to deliver any tumor suppressing gene. Examples of such genes include, but are not limited to, p53, which can be delivered via recombinant virus-mediated gene transfer (see U.S. Patent No.
- a uPA/uPAR antagonist (Adenovirus-Mediated Delivery of a uPA/uPAR Antagonist Suppresses Angiogenesis-Dependent Tumor Growth and Dissemination in Mice," Gene Therapy, August 1998;5(8):1105-13), and interferon gamma (J. Immunol. 2000; 164:217-222).
- the compounds of the instant invention may also be administered in combination with an inhibitor of inherent multidrug resistance (MDR), in particular MDR associated with high levels of expression of transporter proteins.
- MDR inhibitors include inhibitors of p- glycoprotein (P-gp), such as LY335979, XR9576, OC144-093, R101922, VX853 and PSC833 (valspodar).
- a compound of the present invention may be employed in conjunction with antiemetic agents to treat nausea or emesis, including acute, delayed, late-phase, and anticipatory emesis, which may result from the use of a compound of the present invention, alone or with radiation therapy.
- a compound of the present invention may be used in conjunction with other anti-emetic agents, especially neurokinin- 1 receptor antagonists, 5HT3 receptor antagonists, such as ondansetron, granisetron, tropisetron, and zatisetron, GABAB receptor agonists, such as baclofen, a corticosteroid such as Decadron (dexamethasone), Kenalog, Aristocort, Nasalide, Preferid, Benecorten or others such as disclosed in U.S.Patent Nos.
- neurokinin- 1 receptor antagonists especially 5HT3 receptor antagonists, such as ondansetron, granisetron, tropisetron, and zatisetron, GABAB receptor agonists, such as baclofen, a corticosteroid such as Decadron (dexamethasone), Kenalog, Aristocort, Nasalide, Preferid, Benecorten or others such as disclosed in U.S.Patent No
- an antidopaminergic such as the phenothiazines (for example prochlorperazine, fluphenazine, thioridazine and mesoridazine), metoclopramide or dronabinol.
- phenothiazines for example prochlorperazine, fluphenazine, thioridazine and mesoridazine
- metoclopramide metoclopramide or dronabinol.
- conjunctive therapy with an anti-emesis agent selected from a neurokinin- 1 receptor antagonist, a 5HT3 receptor antagonist and a corticosteroid is disclosed for the treatment or prevention of emesis that may result upon administration of the instant compounds.
- Neurokinin- 1 receptor antagonists of use in conjunction with the compounds of the present invention are fully described, for example, in U.S. Patent Nos. 5,162,339, 5,232,929, 5,242,930, 5,373,003, 5,387,595, 5,459,270, 5,494,926, 5,496,833, 5,637,699, 5,719,147; European Patent Publication Nos.
- the neurokinin- 1 receptor antagonist for use in conjunction with the compounds of the present invention is selected from: 2-(R)-(l-(R)-(3,5- bis(trifluoromethyl)phenyl)ethoxy)-3-(S)-(4-fluorophenyl)-4-(3-(5-oxo-lH,4H-l,2,4- triazolo)methyl)morpholine, or a pharmaceutically acceptable salt thereof, which is described in U.S. Patent No. 5,719,147.
- a compound of the instant invention may also be administered with an agent useful in the treatment of anemia.
- an anemia treatment agent is, for example, a continuous eythropoiesis receptor activator (such as epoetin alfa).
- a compound of the instant invention may also be administered with an agent useful in the treatment of neutropenia.
- a neutropenia treatment agent is, for example, a hematopoietic growth factor which regulates the production and function of neutrophils such as a human granulocyte colony stimulating factor, (G-CSF).
- G-CSF human granulocyte colony stimulating factor
- Examples of a G-CSF include filgrastim.
- a compound of the instant invention may also be administered with an immunologic-enhancing drug, such as levamisole, isoprinosine and Zadaxin.
- an immunologic-enhancing drug such as levamisole, isoprinosine and Zadaxin.
- a compound of the instant invention may also be useful for treating or preventing cancer, including bone cancer, in combination with bisphosphonates (understood to include bisphosphonates, diphosphonates, bisphosphonic acids and diphosphonic acids).
- bisphosphonates include but are not limited to: etidronate (Didronel), pamidronate (Aredia), alendronate (Fosamax), risedronate (Actonel), zoledronate (Zometa), ibandronate (Boniva), incadronate or cimadronate, clodronate, EB- 1053, minodronate, neridronate, piridronate and tiludronate including any and all pharmaceutically acceptable salts, derivatives, hydrates and mixtures thereof.
- a compound of the instant invention may also be useful for treating or preventing breast cancer in combination with aromatase inhibitors.
- aromatase inhibitors include but are not limited to: anastrozole, letrozole and exemestane.
- a compound of the instant invention may also be useful for treating or preventing cancer in combination with siRNA therapeutics.
- the compounds of the instant invention may also be administered in combination with ⁇ -secretase inhibitors and/or inhibitors of NOTCH signaling.
- Such inhibitors include compounds described in WO 01/90084, WO 02/30912, WO 01/70677, WO 03/013506, WO 02/36555, WO 03/093252, WO 03/093264, WO 03/093251, WO 03/093253, WO 2004/039800, WO 2004/039370, WO 2005/030731, WO 2005/014553, USSN 10/957,251, WO 2004/089911, WO 02/081435, WO 02/081433, WO 03/018543, WO 2004/031137, WO 2004/031139, WO 2004/031138, WO 2004/101538, WO 2004/101539 and WO 02/47671 (including LY-450139).
- a compound of the instant invention may also be useful for treating or preventing cancer in combination with inhibitors of Akt.
- inhibitors include compounds described in, but not limited to, the following publications: WO 02/083064, WO 02/083139, WO 02/083140, US 2004-0116432, WO 02/083138, US 2004-0102360, WO 03/086404, WO 03/086279, WO 03/086394, WO 03/084473, WO 03/086403, WO 2004/041162, WO 2004/096131, WO
- a compound of the instant invention may also be useful for treating or preventing cancer in combination with PARP inhibitors.
- a compound of the instant invention may also be useful for treating cancer in combination with the following therapeutic agents: abarelix (Plenaxis depot®); aldesleukin (Prokine®); Aldesleukin (Proleukin®); Alemtuzumabb (Campath®); alitretinoin (Panretin®); allopurinol (Zyloprim®); altretamine (Hexalen®); amifostine (Ethyol®); anastrozole (Arimidex®); arsenic trioxide (Trisenox®); asparaginase (Elspar®); azacitidine (Vidaza®); bevacuzimab (Avastin®); bexarotene capsules (Targretin®); bexarotene gel (Targretin®); bleomycin (Blenoxane®); bortezomib
- the scope of the instant invention encompasses the use of the instantly claimed compounds in combination with a second compound selected from: an estrogen receptor modulator, an androgen receptor modulator, a retinoid receptor modulator, a cytotoxic/cytostatic agent, an antiproliferative agent, a prenyl-protein transferase inhibitor, an HMG-CoA reductase inhibitor, an HIV protease inhibitor, a reverse transcriptase inhibitor, an angiogenesis inhibitor, PPAR- ⁇ agonists, PPAR- ⁇ agonists, an inhibitor of inherent multidrug resistance, an anti-emetic agent, an agent useful in the treatment of anemia, an agent useful in the treatment of neutropenia, an immunologic-enhancing drug, an inhibitor of cell proliferation and survival signaling, a bisphosphonate, an aromatase inhibitor, an siRNA therapeutic, ⁇ -secretase inhibitors, agents that interfere with receptor tyrosine kinases (RTKs), an agent that interferes with a cell cycle checkpoint
- administration means introducing the compound or a prodrug of the compound into the system of the animal in need of treatment.
- a compound of the invention or prodrug thereof is provided in combination with one or more other active agents (e.g., a cytotoxic agent, etc.)
- administration and its variants are each understood to include concurrent and sequential introduction of the compound or prodrug thereof and other agents.
- composition is intended to encompass a product comprising the specified ingredients in the specified amounts, as well as any product which results, directly or indirectly, from combination of the specified ingredients in the specified amounts.
- terapéuticaally effective amount means that amount of active compound or pharmaceutical agent that elicits the biological or medicinal response in a tissue, system, animal or human that is being sought by a researcher, veterinarian, medical doctor or other clinician.
- treating cancer refers to administration to a mammal afflicted with a cancerous condition and refers to an effect that alleviates the cancerous condition by killing the cancerous cells, but also to an effect that results in the inhibition of growth and/or metastasis of the cancer.
- a method of treating cancer comprises administering a therapeutically effective amount of a compound of the instant invention in combination with radiation therapy and/or in combination with a second compound selected from: an estrogen receptor modulator, an androgen receptor modulator, a retinoid receptor modulator, a cytotoxiccytostatic agent, an antiproliferative agent, a prenyl-protein transferase inhibitor, an HMG-CoA reductase inhibitor, an HIV protease inhibitor, a reverse transcriptase inhibitor, an angiogenesis inhibitor, PPAR- ⁇ agonists, PPAR- ⁇ agonists, an inhibitor of inherent multidrug resistance, an anti-emetic agent, an agent useful in the treatment of anemia, an agent useful in the treatment of neutropenia, an immunologic-enhancing drug, an inhibitor of cell proliferation and survival signaling, a bisphosphonate, an aromatase inhibitor, an siRNA therapeutic, ⁇ -secretase inhibitors, agents that interfere with receptor tyros
- the instant invention also includes a pharmaceutical composition useful for treating or preventing cancer that comprises a therapeutically effective amount of a compound of the instant invention and a second compound selected from: an estrogen receptor modulator, an androgen receptor modulator, a retinoid receptor modulator, a cytotoxic/cytostatic agent, an antiproliferative agent, a prenyl-protein transferase inhibitor, an HMG-CoA reductase inhibitor, an HFV protease inhibitor, a reverse transcriptase inhibitor, an angiogenesis inhibitor, a PPAR- ⁇ agonist, a PPAR- ⁇ agonist, an inhibitor of cell proliferation and survival signaling, a bisphosphonate, an aromatase inhibitor, an siRNA therapeutic, ⁇ -secretase inhibitors, agents that interfere with receptor tyrosine kinases (RTKs), an agent that interferes with a cell cycle checkpoint and any of the therapeutic agents listed above.
- a pharmaceutical composition useful for treating or preventing cancer that comprises a therapeutically effective
- the compounds of the present invention can be prepared according to the following general schemes, using appropriate materials, and are further exemplified by the subsequent specific examples.
- the compounds illustrated in the examples are not, however, to be construed as forming the only genus that is considered as the invention.
- the illustrative Examples below, therefore, are not limited by the compounds listed or by any particular substituents employed for illustrative purposes. Those skilled in the art will readily understand that known variations of the conditions and processes of the following preparative procedures can be used to prepare these compounds. All temperatures are degrees Celsius unless otherwise noted.
- Aryl or heteroaryl aldehyde IX can be homologated by one carbon using the appropriate Wittig agent and base, and the corresponding methyl enol ether X can be hydrolyzed under acidic conditions to give the aryl or heteroaryl acetaldehyde XI.
- the aldehyde can be condensed with 2-cyanoacetamide and sulfur in the presence of a tertiary amine base to afford thiophene XII, which can be coupled with an appropriate six-memered halogenated heterocycle optionally substituted (R 2 ) using palladium catalysis to give the 2-aminopyridyl thiophene VI.
- An appropriate aryl or heteroaryl iodide XV can be coupled to allyl bromide by first performing an iodo-magensium exchange and then introducing allyl bromide in the presence of a copper cyanide-lithium chloride complex.
- the resulting allylated aromatic XVI can be dihydroxylated using a catalytic amount of osmium tetroxide in the presence of a stoichiometric reoxidant, giving the diol XV that can be cleaved to give the aryl or heteroaryl acetylaldehyde XI.
- the aldehyde can be condensed with 2-cyanoacetamide and sulfur to afford thiophene XII, which can be coupled with an appropriate six-memered halogenated heterocycle optionally substituted (R 2 ) using palladium catalysis to give the 2-aminopyridyl thiophene VI.
- Nitrothiophene XVI can be alkylated with chloroform using a variety of strongly basic conditions to afford the dihchloromethyl adduct XVII.
- Hydrolysis to the aldehyde XVIII under acidic or basic conditions followed by condensation with hydroxylamine affords oxime XTV.
- Transition metal-catalyzed rearrangement to the amide XV followed by palladium catalyzed cross coupling with a variety of aryl and heteroaryl boronates affords the functionalized nitro thiophene intermediate XVI.
- Reduction of the nitro group and coupling with an appropriate six- memered halogenated heterocycle optionally substituted (R 2 ) using palladium catalysis gives the 2-aminopyridyl thiophene VI.
- the 2-amino thiophene XXI is then elaborated to the 2-aminopyridyl thiophene VII through palladium catalyzed coupling with an appropriate six-memered halogenated heterocycle optionally substituted (R 2 ) using palladium catalysis. Finally, the 2-aminopyridyl thiophene XXII is hydrolyzed with base to give the final 3 -amide thiophene product VI.
- Step 3 Benzyl [3 -(aminocarbonyl)- 5 -( " 2 ,4-difluorophenyl)-2-thienyll carbamate A suspension of benzyl [3-(aminocarbonyl)-5-iodo-2-thienyl]carbamate (250 mg,
- Step 1 Benzyl [3 -(aminocarbonyl>5 -(2-fluorophenyl)-2-thienyll carbamate
- the crude oil was diluted with ethyl acetate (140 mL), isopropyl alcohol (10 mL), and aqueous citric acid (1 M, 25 mL). The layers were separated and the organic layer was washed with water (25 mL), 5:1 saturated aqueous sodium bicarbonate brine (30 mL), and brine (25 mL), dried over sodium sulfate, filtered, and concentrated. The crude yellow solid was triturated with dichloromethane to give the title compound.
- Step l ' 13.5-Trifluoro-2-IT£, Z)-2-methoxyvinyl1benzene
- the title compound was prepared from 2,4,6-trifluorobenzaldehyde (5 g, 31.2 mmol) as the starting material according to the general procedure described in Intermediate 3 Step 1.
- Step 3 2- Amino-5 -(2 A6-trifluorophenvPthiophene-3 -carboxamide
- the title compound was prepared from (2,4,6-trifluorophenyl)acetaldehyde (3.64 g, 20.9 mmol) as the starting material according to the general procedure described in Intermediate 3 Step 3. Calc'd for C 11 H 8 F 3 N 2 OS [M+H]: 273, Found: 273.
- Step 1 General Procedure for the Preparation of Allylbenzenes from Iodobenzenes
- a 100 mL flask containing copper(I) cyanide (0.854 g, 9.54 mmol) and lithium chloride (0.809 g, 19.08 mmol) was placed under vacuum and heated to 150°C for 90 minutes, and then cooled to room temperature.
- a 500 mL flask containing a solution of methyl 4- iodobenzoate (12.5 g, 47.7 mmol) in tetrahydrofuran (75 mL) under argon was cooled to -25°C and isopropylmagnesium chloride - lithium chloride complex (49.1 mL, 1 M, 49.1 mmol) was added over 16 minutes while the internal temperature was maintained at or below -20°C.
- Step 2 2-(4-Allylphenyl)propan-2-ol
- methyl 4-allylbenzoate 5.66 g, 32.1 mmol
- tetrahydrofuran 37 mL
- diethyl ether 3 M, 26.8 mL, 80 mmol
- the reaction mixture was allowed to warm to room temperature and then it was cooled to 0°C after an additional four hours, at which time a saturated aqueous ammonium chloride solution (50 mL) was added slowly followed by diethyl ether (100 mL).
- the layers were separated, and the aqueous layer was extracted with diethyl ether (2 x 50 mL).
- the combined organic layers were dried over magnesium sulfate, filtered, and concentrated to afford the title compound.
- Step 3 3 - ⁇ 4-( 1 -Hydroxy- 1 -methylethyl)phenyl]propane- 1 ,2-diol
- the filter was rinsed with acetone (2 x 5 mL, then 2 x 10 mL), and filtrate was concentrated by rotary evaporation to remove the acetone.
- the combined organic layers were washed with 3:1 water :brine (12 mL), saturated aqueous sodium bicarbonate (10 mL), and brine (10 mL).
- Step 4 ⁇ 4-( 1 -Hydroxy- 1 -methylethyDphenyliacetaldehyde
- the aqueous layer was extracted with diethyl ether (2 x 100 mL), and the combined organic layers were washed with brine (25 mL), dried over sodium sulfate, filtered, and concentrated. To remove water left over, the concentrated material was dissolved in dichloromethane (70 mL), washed with brine (20 mL), dried over sodium sulfate, filtered, and concentrated to yield the title compound.
- Step 2 3-(4-Fluorophenyl)propane- 1 ,2-diol
- Step 1 3 -(4-Chlorophenyl)propane- 1 ,2-diol
- Methoxymethyl)triphenylphosphonium chloride (147 g, 0.48 mol) was suspended in tetrahydrofuran (1.5 L) under an argon atmosphere, and cooled to 0°C t-BuOK (51.6 g, 0.46 mol) was added in portions.
- a solution of 4-bromo-2-fluorobenzaldehyde (40.6 g, 0.2 mol) in tetrahydrofuran (500 mL) was then added to the reaction mixture. The solution was stirred at room temperature for 1 hour. The solution was poured into ice-water and extracted with ethyl acetate (2x). The combined organic phases were dried and concentrated in vacuo. Purification via flash chromatography afforded the title compound.
- Step 1 5 -Bromo-3 -(dichloromethyl)-2-nitrothiophene
- 2-bromo-5-nitrothiophene 29 g, 139 mmol
- chloroform 12.37 mL, 153 mmol
- DMF 110 mL
- the internal temperature was monitored and maintained at ⁇ -60°C during the addition.
- the reaction was stirred at -78 0 C for 30 minutes. 2 N HCl was added and the products were extracted into EtOAc (3x).
- Step 1 2-(5-Bromopyridin-2-yl)-2-methylpropanenitrile
- 2,5-Dibromopyridine (2g, 8.44 mmol) was placed in a vial that was evacuated and backfilled with argon three times. Anhydrous dioxane (8.4 ml) was then added and the suspension was stirred. In a separate vial, 2-methylpropanenitrile (0.76 ml, 8.44 mmol) was added to a solution of sodium 1,1, 1,3,3, 3-hexamethyldisilazan-2-ide (28.1 ml of 0.6 M in toluene, 16.9 mmol). This was stirred for 10 minutes then added to the suspension. The reaction was heated to 70°C for 1.5 hours. It was then cooled to room temperature, quenched with water, and extracted with ethyl acetate.
- Step 2 2-Metfayl-2-r5-(4,4,5J-tetrametfayl-1.3.2-dioxaborolan-2-vnpyridin-2- yllpropanenitrile and [6-(l-cvano-l-methylethyr)pyridin-3-yl1boronic acid 2-(5-Bromopyridin-2-yl)-2-methylpropanenitrile (300 mg, 1.33 mmol),
- Step 3 5-
- Step 4 Z-Amino-S-r ⁇ -d-cvano-l-methylethyDpyridin-S-ylithiophene-S-carboxainide Nitro reduction, Method B: 5-[6-(l-Cyano-l-methylethyl)pyridin-3-yl]-2- nitrothiophene-3-carboxamide (95 mg, 0.29 mmol) and iron (III) chloride (2.33 mg, 0.014 mmol) were put in a vial. The vial was evacuated and backfilled with argon three times. Degassed methanol (4.1 mL) was added and the solution was heated to 65°C for 10 minutes.
- Step 2. 5 - 1 4-(3 -Hydroxyoxetan-3 -vOphenyl] -2-nitrothiophene-3 -carboxamide
- Step 1 5-(6-Mo ⁇ holin-4-ylpyridin-3-yl)-2-nitrothiophene-3-carboxamide
- the title compound was prepared from 5-bromo-2-nitrothiophene-3-carboxamide
- Step 1 4-(4-Bromo-phenyl ' )-thiomorpholine 1 , 1 -dioxide 1,1 '-Sulfonyldiethylene divinyl sulfone (25 g, 21.18 mmol; 0.02 M in proan-2-ol) was added dropwise to a stirred refluxing solution of 4-bromoaniline (36.44 g, 21.18 mmol; 0.01 M in propan-2-o I/water, 1 :1). The reaction mixture was refluxed for 48 h.
- the reaction mixture was cooled to room temperature and the precipitate was collected by filtration and washed with water followed by hexane/diethylether (1 :1) to afford the title compound as a white solid.
- the mother liquor was concentrated to half volume under reduced pressure, cooled to room temperature, and the precipitate was collected and washed as above.
- Step 2 4-r4-(4.4.5.5-Tetramethyl- ⁇ 1.3.21 dioxaborolan-2-ylVphenyll-thiomorpholine 1.1- dioxide
- Step 3 5-[4-(1 , 1 -Dioxidothiomo ⁇ holin-4-yl)phenyll-2-nitrothiophene-3-carboxamide
- a sealed tube was charged with l-bromo-3,5-difluorobenzene (1.79 ml, 15.5 mmol), Pd 2 (dba) 3 (0.85 g, 0.93 mmol), X-PHOS (2.22 g, 4.66 mmol), and potassium carbonate (4.73 g, 34.2 mmol).
- the tube was evacuated and backfilled with argon 3x.
- Fully degassed tert- amyl alcohol (51 mL) was added followed immediately by the addition of morpholine (2.71 ml, 31.1 mmol).
- the tube was then sealed and placed in an oil bath at 100°C and stirred overnight. The reaction mixture was taken up in diethyl ether and water.
- Step 2 (2,6-Difluoro-4-morpholin-4-ylphenyl)boronic acid (TFA salt)
- TFA salt (2,6-Difluoro-4-morpholin-4-ylphenyl)boronic acid
- trimethyl borate (4.4 ml, 39.4 mmol) was added dropwise. The reaction was allowed to warm to room temperature overnight. The reaction was then quenched with 2 N aqueous hydrochloric acid and stirred for 10 minutes at room temperature.
- Step 3 5-(2,6-Difluoro-4-morpholin-4-ylphenyl)-2-nitrothiophene-3-carboxamide
- the title compound was prepared as described in Intermediate 10 Step 5 using 5- bromo-2-nitrothiophene-3-carboxamide (Intermediate 10 Step 4) (582 mg, 2.32 mmol) and (2,6- difluoro-4-morpholin-4-ylphenyl)boronic acid (TFA salt) (830 mg, 2.33 mmol) as starting materials.
- Step 1 5-Bromo-2-( 1 - ⁇
- 2-(5-bromopyridin-2-yl)propan-2-ol (1.20 g, 5.55 mmol, prepared according to the method in Tetrahedron Lett. 2000, 41, 4335) and 2,6-lutidine (1.29 mL, 11.11 mmol) in CH 2 Cl 2 (12 mL) was added TBSOTf (1.91 mL, 8.33 mmol). After stirring at room temperature for 2 h, the reaction was diluted with water and extracted with CH 2 Cl 2 (2x).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2690141A CA2690141A1 (en) | 2007-06-20 | 2008-06-16 | Inhibitors of janus kinases |
| AU2008266883A AU2008266883B2 (en) | 2007-06-20 | 2008-06-16 | Inhibitors of janus kinases |
| JP2010513207A JP5478488B2 (en) | 2007-06-20 | 2008-06-16 | JANUS kinase inhibitors |
| EP08768504A EP2166846B1 (en) | 2007-06-20 | 2008-06-16 | Inhibitors of janus kinases |
| US12/665,045 US8367706B2 (en) | 2007-06-20 | 2008-06-16 | Inhibitors of janus kinases |
| ES08768504T ES2395581T3 (en) | 2007-06-20 | 2008-06-16 | Janus kinase inhibitors |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US93657207P | 2007-06-20 | 2007-06-20 | |
| US60/936,572 | 2007-06-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008156726A1 true WO2008156726A1 (en) | 2008-12-24 |
Family
ID=40156543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/007486 WO2008156726A1 (en) | 2007-06-20 | 2008-06-16 | Inhibitors of janus kinases |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8367706B2 (en) |
| EP (1) | EP2166846B1 (en) |
| JP (1) | JP5478488B2 (en) |
| AU (1) | AU2008266883B2 (en) |
| CA (1) | CA2690141A1 (en) |
| ES (1) | ES2395581T3 (en) |
| WO (1) | WO2008156726A1 (en) |
Cited By (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011044157A1 (en) * | 2009-10-06 | 2011-04-14 | Biogen Idec Ma Inc. | Heterocyclic compounds useful as pdk1 inhibitors |
| EP2310384A1 (en) * | 2008-07-09 | 2011-04-20 | Merck Sharp & Dohme Corp. | Inhibitors of janus kinases |
| WO2011074560A1 (en) * | 2009-12-15 | 2011-06-23 | 塩野義製薬株式会社 | Oxadiazole derivative having endothelial lipase inhibitory activity |
| DE102010049877A1 (en) | 2010-11-01 | 2012-05-03 | Merck Patent Gmbh | 7 - ((1,2,3) triazol-4-yl) -pyrrolo (2,3) pyrazine derivatives |
| DE102011008352A1 (en) | 2011-01-12 | 2012-07-12 | Merck Patent Gmbh | 5 - ([1,2,3] triazol-4-yl) -7H-pyrrolo [2,3-d] pyrimidine derivatives |
| DE102011009961A1 (en) | 2011-02-01 | 2012-08-02 | Merck Patent Gmbh | 7-azaindole derivatives |
| WO2013098229A2 (en) | 2011-12-27 | 2013-07-04 | Bayer Intellectual Property Gmbh | Heteroarylpiperidine and piperazine derivatives as fungicides |
| WO2013113791A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113782A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113720A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113716A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113776A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113781A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds i |
| WO2013113778A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113773A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113863A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113719A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds ii |
| WO2013113715A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| EP2634185A1 (en) | 2012-03-02 | 2013-09-04 | Sareum Limited | TYK2 kinase inhibitors |
| JP2013536193A (en) * | 2010-08-11 | 2013-09-19 | ミレニアム ファーマシューティカルズ, インコーポレイテッド | Heteroaryls and uses thereof |
| WO2013135672A1 (en) | 2012-03-13 | 2013-09-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013135671A1 (en) | 2012-03-13 | 2013-09-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2014013014A1 (en) | 2012-07-18 | 2014-01-23 | Fundació Privada Centre De Regulació Genòmica (Crg) | Jak inhibitors for activation of epidermal stem cell populations |
| DE102012019369A1 (en) | 2012-10-02 | 2014-04-03 | Merck Patent Gmbh | 7-Azaindolderivat |
| WO2015032423A1 (en) | 2013-09-03 | 2015-03-12 | Sareum Limited | Pharmaceutical compounds |
| WO2015036058A1 (en) | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2015036059A1 (en) | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2015186056A1 (en) | 2014-06-03 | 2015-12-10 | Actelion Pharmaceuticals Ltd | Pyrazole compounds and their use as t-type calcium channel blockers |
| WO2016041892A1 (en) | 2014-09-15 | 2016-03-24 | Actelion Pharmaceuticals Ltd | Triazole compounds as t-type calcium channel blockers |
| WO2018041989A1 (en) | 2016-09-02 | 2018-03-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing and treating refractory celiac disease type 2 |
| KR101839137B1 (en) | 2015-10-12 | 2018-03-26 | 주식회사 종근당 | Oxadiazole Amine Derivative Compounds as Histone Deacetylase 6 Inhibitor, and the Pharmaceutical Composition Comprising the same |
| WO2018073438A1 (en) | 2016-10-21 | 2018-04-26 | Sareum Limited | Pharmaceutical compounds |
| WO2020074461A1 (en) | 2018-10-08 | 2020-04-16 | Sareum Limited | Tyk2 kinase inhibitors |
| WO2020139992A1 (en) | 2018-12-27 | 2020-07-02 | Agios Pharmaceuticals, Inc. | Aza-heterobicyclic inhibitors of mat2a and methods of use for treating cancer |
| WO2020201362A2 (en) | 2019-04-02 | 2020-10-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods of predicting and preventing cancer in patients having premalignant lesions |
| WO2020212395A1 (en) | 2019-04-16 | 2020-10-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of jak inhibitors for the treatment of painful conditions involving nav1.7 channels |
| WO2020243376A1 (en) | 2019-05-31 | 2020-12-03 | Agios Pharmaceuticals, Inc. | Heterobicyclic inhibitors of mat2a and methods of use for treating cancer |
| US10899695B2 (en) | 2017-02-06 | 2021-01-26 | Idorsia Pharmaceuticals Ltd | Process for the synthesis of 1-aryl-1-trifluoromethylcyclopropanes |
| GB2589910A (en) * | 2019-12-12 | 2021-06-16 | Henkel IP & Holding GmbH | Process for preparing cyanoacetates |
| WO2021204762A1 (en) | 2020-04-07 | 2021-10-14 | Sareum Limited | Crystalline forms of a tyk2 inhibitor |
| US11213517B2 (en) | 2016-12-16 | 2022-01-04 | Idorsia Pharmaceuticals Ltd | Pharmaceutical combination comprising a T-type calcium channel blocker |
| CN115215793A (en) * | 2022-08-11 | 2022-10-21 | 南开大学 | Synthesis method of fluopyram |
| WO2023222565A1 (en) | 2022-05-16 | 2023-11-23 | Institut National de la Santé et de la Recherche Médicale | Methods for assessing the exhaustion of hematopoietic stems cells induced by chronic inflammation |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2181110A2 (en) * | 2007-07-13 | 2010-05-05 | ADDEX Pharma S.A. | Novel heteroaromatic derivatives and their use as positive allosteric modulators of metabotropic glutamate receptors |
| JP2011518221A (en) * | 2008-04-21 | 2011-06-23 | メルク・シャープ・エンド・ドーム・コーポレイション | JANUS kinase inhibitors |
| AU2009260389A1 (en) * | 2008-06-18 | 2009-12-23 | Merck Sharp & Dohme Corp. | Inhibitors of Janus kinases |
| UY31984A (en) * | 2008-07-16 | 2010-02-26 | Boehringer Ingelheim Int | DERIVATIVES OF N-substituted 1- (3,4-difluorobenzyl) -6-oxo-1,6-dihydropyrimidin-5-carboxamides and 2- (3,4-difluorobenzyl) -3-oxo-2,3-dihydro- N-substituted 1H-pyrazol-4-carboxamides. |
| EP2736899A1 (en) | 2011-07-26 | 2014-06-04 | Grünenthal GmbH | Substituted bicyclic aromatic carboxamide and urea derivatives as vanilloid receptor ligands |
| FR3046793B1 (en) * | 2016-01-14 | 2018-01-05 | Les Laboratoires Servier | NOVEL PHOSPHINAN DERIVATIVES AND AZAPHOSPHINANES, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| US10696638B2 (en) | 2017-12-26 | 2020-06-30 | Industrial Technology Research Institute | Compounds for inhibiting AGC kinase and pharmaceutical compositions comprising the same |
| US20230183234A1 (en) * | 2020-05-04 | 2023-06-15 | Otsuka Pharmaceutical Co., Ltd. | Iap antagonist compounds and intermediates and methods for synthesizing the same |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050154014A1 (en) * | 2003-01-06 | 2005-07-14 | Jason Bloxham | (2-carboxamido)(3-amino) thiophene compounds |
| US7112594B2 (en) * | 2000-08-09 | 2006-09-26 | Mitsubishi Pharma Corporation | Fused bicyclic amide compounds and medicinal use thereof |
| US7179836B2 (en) * | 2002-09-20 | 2007-02-20 | Smithkline Beecham Corporation | Chemical compounds |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT853083E (en) * | 1997-01-06 | 2001-12-28 | Pfizer | COMPOSITION OF PYRIDILFURANE AND PYRIDYLTHOPHENE AND ITS PHARMACEUTICAL UTILIZATION |
| US6414013B1 (en) | 2000-06-19 | 2002-07-02 | Pharmacia & Upjohn S.P.A. | Thiophene compounds, process for preparing the same, and pharmaceutical compositions containing the same background of the invention |
| PL360439A1 (en) * | 2000-06-28 | 2004-09-06 | Astrazeneca Ab | Substituted quinazoline derivatives and their use as inhibitors |
| EP1324759A4 (en) | 2000-10-12 | 2004-05-12 | Smithkline Beecham Corp | Nf-g(k)b inhibitors |
| WO2003027093A1 (en) * | 2001-09-21 | 2003-04-03 | Smithkline Beecham Corporation | Chemical compounds |
| SE0300092D0 (en) * | 2003-01-15 | 2003-01-15 | Astrazeneca Ab | Novel compounds |
| US20050085531A1 (en) | 2003-10-03 | 2005-04-21 | Hodge Carl N. | Thiophene-based compounds exhibiting ATP-utilizing enzyme inhibitory activity, and compositions, and uses thereof |
| JP2009533378A (en) | 2006-04-10 | 2009-09-17 | ベーリンガー インゲルハイム インターナショナル ゲゼルシャフト ミット ベシュレンクテル ハフツング | 2,4-Diaminopyrimidine derivatives and their use for the treatment of cancer |
-
2008
- 2008-06-16 AU AU2008266883A patent/AU2008266883B2/en not_active Ceased
- 2008-06-16 ES ES08768504T patent/ES2395581T3/en active Active
- 2008-06-16 US US12/665,045 patent/US8367706B2/en active Active
- 2008-06-16 EP EP08768504A patent/EP2166846B1/en active Active
- 2008-06-16 JP JP2010513207A patent/JP5478488B2/en not_active Expired - Fee Related
- 2008-06-16 CA CA2690141A patent/CA2690141A1/en not_active Abandoned
- 2008-06-16 WO PCT/US2008/007486 patent/WO2008156726A1/en active Application Filing
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7112594B2 (en) * | 2000-08-09 | 2006-09-26 | Mitsubishi Pharma Corporation | Fused bicyclic amide compounds and medicinal use thereof |
| US7179836B2 (en) * | 2002-09-20 | 2007-02-20 | Smithkline Beecham Corporation | Chemical compounds |
| US20050154014A1 (en) * | 2003-01-06 | 2005-07-14 | Jason Bloxham | (2-carboxamido)(3-amino) thiophene compounds |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP2166846A4 * |
Cited By (80)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2310384A1 (en) * | 2008-07-09 | 2011-04-20 | Merck Sharp & Dohme Corp. | Inhibitors of janus kinases |
| EP2310384A4 (en) * | 2008-07-09 | 2012-02-22 | Merck Sharp & Dohme | Inhibitors of janus kinases |
| US8420695B2 (en) | 2008-07-09 | 2013-04-16 | Merck Sharp & Dohme Corp. | Inhibitors of janus kinases |
| CN102665718A (en) * | 2009-10-06 | 2012-09-12 | 米伦纽姆医药公司 | Heterocyclic compounds useful as pdk1 inhibitors |
| WO2011044157A1 (en) * | 2009-10-06 | 2011-04-14 | Biogen Idec Ma Inc. | Heterocyclic compounds useful as pdk1 inhibitors |
| CN102665718B (en) * | 2009-10-06 | 2016-03-09 | 米伦纽姆医药公司 | Can be used as the heterocyclic compound of PDK1 inhibitor |
| US9546165B2 (en) | 2009-10-06 | 2017-01-17 | Sunesis Pharmaceuticals, Inc. | Heterocyclic compounds useful as PDK1 inhibitors |
| RU2615130C2 (en) * | 2009-10-06 | 2017-04-04 | Милленниум Фармасьютикалз, Инк | Heterocyclic compounds useful as pdk1 inhibitors |
| US10030016B2 (en) | 2009-10-06 | 2018-07-24 | Sunesis Pharmaceuticals, Inc. | Heterocyclic compounds useful as PDK1 inhibitors |
| AU2018222943B2 (en) * | 2009-10-06 | 2019-09-26 | Millennium Pharmaceuticals, Inc. | Heterocyclic compounds useful as PDK1 inhibitors |
| JP5791150B2 (en) * | 2009-12-15 | 2015-10-07 | 塩野義製薬株式会社 | Oxadiazole derivatives having vascular endothelial lipase inhibitory activity |
| US8754113B2 (en) | 2009-12-15 | 2014-06-17 | Shionogi & Co., Ltd. | Oxadiazole derivative having endothelial lipase inhibitory activity |
| WO2011074560A1 (en) * | 2009-12-15 | 2011-06-23 | 塩野義製薬株式会社 | Oxadiazole derivative having endothelial lipase inhibitory activity |
| CN102753545A (en) * | 2009-12-15 | 2012-10-24 | 盐野义制药株式会社 | Oxadiazole derivative having endothelial lipase inhibitory activity |
| JP2013536193A (en) * | 2010-08-11 | 2013-09-19 | ミレニアム ファーマシューティカルズ, インコーポレイテッド | Heteroaryls and uses thereof |
| WO2012059171A1 (en) | 2010-11-01 | 2012-05-10 | Merck Patent Gmbh | 7-([1,2,3]triazol-4-yl)-pyrrolo[2,3-b]pyrazine derivatives |
| DE102010049877A1 (en) | 2010-11-01 | 2012-05-03 | Merck Patent Gmbh | 7 - ((1,2,3) triazol-4-yl) -pyrrolo (2,3) pyrazine derivatives |
| DE102011008352A1 (en) | 2011-01-12 | 2012-07-12 | Merck Patent Gmbh | 5 - ([1,2,3] triazol-4-yl) -7H-pyrrolo [2,3-d] pyrimidine derivatives |
| WO2012095142A1 (en) | 2011-01-12 | 2012-07-19 | Merck Patent Gmbh | 5-([1,2,3]triazole-4-yl)-7h-pyrrolo[2,3-d]pyrimidine derivatives |
| EP3133074A2 (en) | 2011-02-01 | 2017-02-22 | Merck Patent GmbH | 7-azaindole derivatives |
| DE102011009961A1 (en) | 2011-02-01 | 2012-08-02 | Merck Patent Gmbh | 7-azaindole derivatives |
| WO2012104007A2 (en) | 2011-02-01 | 2012-08-09 | Merck Patent Gmbh | 7-azaindole derivatives |
| EP2921495A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Heteroarylpiperidine and heteroarylpiperazine derivatives as fungicides |
| EP2921484A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Oxazole derivatives |
| EP2921494A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Heteroarylpiperidine and heteroarylpiperazine derivatives |
| EP2921491A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Intermediates for the production of heteroarylpiperidine and heteroarylpiperazine derivatives as fungicides |
| EP2921485A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Isoxazole derivatives |
| EP2921492A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Heteroarylpiperidine and heteroarylpiperazine derivatives |
| WO2013098229A2 (en) | 2011-12-27 | 2013-07-04 | Bayer Intellectual Property Gmbh | Heteroarylpiperidine and piperazine derivatives as fungicides |
| EP2921493A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | Heteroarylpiperidine and heteroarylpiperazine derivatives |
| EP2921481A1 (en) | 2011-12-27 | 2015-09-23 | Bayer Intellectual Property GmbH | 4-piperidine carboxylic acid derivatives |
| WO2013113776A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113773A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113782A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113791A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113720A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113787A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113716A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113788A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113781A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds i |
| WO2013113778A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113715A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013113719A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds ii |
| WO2013113863A1 (en) | 2012-02-03 | 2013-08-08 | Basf Se | Fungicidal pyrimidine compounds |
| EP3040336A1 (en) | 2012-03-02 | 2016-07-06 | Sareum Limited | Tyk2 kinase inhibitors |
| EP2634185A1 (en) | 2012-03-02 | 2013-09-04 | Sareum Limited | TYK2 kinase inhibitors |
| US11673870B2 (en) | 2012-03-02 | 2023-06-13 | Sareum Limited | Pharmaceutical compounds |
| US10882829B2 (en) | 2012-03-02 | 2021-01-05 | Sareum Limited | Pharmaceutical compounds |
| WO2013135672A1 (en) | 2012-03-13 | 2013-09-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2013135671A1 (en) | 2012-03-13 | 2013-09-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2014013014A1 (en) | 2012-07-18 | 2014-01-23 | Fundació Privada Centre De Regulació Genòmica (Crg) | Jak inhibitors for activation of epidermal stem cell populations |
| DE102012019369A1 (en) | 2012-10-02 | 2014-04-03 | Merck Patent Gmbh | 7-Azaindolderivat |
| WO2014053208A1 (en) | 2012-10-02 | 2014-04-10 | Merck Patent Gmbh | 7-azaindol-2,7-naphthyridine derivative for the treatment of tumors |
| WO2015032423A1 (en) | 2013-09-03 | 2015-03-12 | Sareum Limited | Pharmaceutical compounds |
| WO2015036059A1 (en) | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| WO2015036058A1 (en) | 2013-09-16 | 2015-03-19 | Basf Se | Fungicidal pyrimidine compounds |
| US9932314B2 (en) | 2014-06-03 | 2018-04-03 | Idorsia Pharmaceuticals Ltd | Pyrazole compounds and their use as T-type calcium channel blockers |
| US10065929B2 (en) | 2014-06-03 | 2018-09-04 | Idorsia Pharmaceuticals Ltd | Pyrazole compounds and their use as T-type calcium channel blockers |
| WO2015186056A1 (en) | 2014-06-03 | 2015-12-10 | Actelion Pharmaceuticals Ltd | Pyrazole compounds and their use as t-type calcium channel blockers |
| WO2016041892A1 (en) | 2014-09-15 | 2016-03-24 | Actelion Pharmaceuticals Ltd | Triazole compounds as t-type calcium channel blockers |
| US10246426B2 (en) | 2014-09-15 | 2019-04-02 | Idorsia Pharmaceuticals Ltd | Triazole compounds as T-type calcium channel blockers |
| KR101839137B1 (en) | 2015-10-12 | 2018-03-26 | 주식회사 종근당 | Oxadiazole Amine Derivative Compounds as Histone Deacetylase 6 Inhibitor, and the Pharmaceutical Composition Comprising the same |
| WO2018041989A1 (en) | 2016-09-02 | 2018-03-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods for diagnosing and treating refractory celiac disease type 2 |
| US11154539B2 (en) | 2016-10-21 | 2021-10-26 | Sareum Limited | Pharmaceutical compounds |
| WO2018073438A1 (en) | 2016-10-21 | 2018-04-26 | Sareum Limited | Pharmaceutical compounds |
| EP4066831A1 (en) | 2016-10-21 | 2022-10-05 | Sareum Limited | Pharmaceutical compounds |
| US11213517B2 (en) | 2016-12-16 | 2022-01-04 | Idorsia Pharmaceuticals Ltd | Pharmaceutical combination comprising a T-type calcium channel blocker |
| US10899695B2 (en) | 2017-02-06 | 2021-01-26 | Idorsia Pharmaceuticals Ltd | Process for the synthesis of 1-aryl-1-trifluoromethylcyclopropanes |
| WO2020074461A1 (en) | 2018-10-08 | 2020-04-16 | Sareum Limited | Tyk2 kinase inhibitors |
| WO2020139992A1 (en) | 2018-12-27 | 2020-07-02 | Agios Pharmaceuticals, Inc. | Aza-heterobicyclic inhibitors of mat2a and methods of use for treating cancer |
| WO2020201362A2 (en) | 2019-04-02 | 2020-10-08 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Methods of predicting and preventing cancer in patients having premalignant lesions |
| WO2020212395A1 (en) | 2019-04-16 | 2020-10-22 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of jak inhibitors for the treatment of painful conditions involving nav1.7 channels |
| WO2020243376A1 (en) | 2019-05-31 | 2020-12-03 | Agios Pharmaceuticals, Inc. | Heterobicyclic inhibitors of mat2a and methods of use for treating cancer |
| GB2589910A (en) * | 2019-12-12 | 2021-06-16 | Henkel IP & Holding GmbH | Process for preparing cyanoacetates |
| WO2021116128A1 (en) * | 2019-12-12 | 2021-06-17 | Henkel IP & Holding GmbH | Process for preparing cyanoacetates |
| GB2589910B (en) * | 2019-12-12 | 2021-12-01 | Henkel IP & Holding GmbH | Process for preparing cyanoacetates |
| US11976024B2 (en) | 2019-12-12 | 2024-05-07 | Henkel Ag & Co. Kgaa | Process for preparing cyanoacetates |
| WO2021204762A1 (en) | 2020-04-07 | 2021-10-14 | Sareum Limited | Crystalline forms of a tyk2 inhibitor |
| WO2023222565A1 (en) | 2022-05-16 | 2023-11-23 | Institut National de la Santé et de la Recherche Médicale | Methods for assessing the exhaustion of hematopoietic stems cells induced by chronic inflammation |
| CN115215793A (en) * | 2022-08-11 | 2022-10-21 | 南开大学 | Synthesis method of fluopyram |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5478488B2 (en) | 2014-04-23 |
| US8367706B2 (en) | 2013-02-05 |
| AU2008266883B2 (en) | 2014-01-30 |
| CA2690141A1 (en) | 2008-12-24 |
| ES2395581T3 (en) | 2013-02-13 |
| AU2008266883A1 (en) | 2008-12-24 |
| EP2166846A4 (en) | 2011-03-23 |
| EP2166846A1 (en) | 2010-03-31 |
| JP2010530420A (en) | 2010-09-09 |
| US20100256097A1 (en) | 2010-10-07 |
| EP2166846B1 (en) | 2012-10-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2166846B1 (en) | Inhibitors of janus kinases | |
| EP2278969B1 (en) | Inhibitors of Janus kinases | |
| AU2008335761B2 (en) | Inhibitors of janus kinases | |
| CA2703125C (en) | Pyrazinyl-substituted pyrrolo[2,3-b]pyridines, compositions thereof, and their use in the treatment of cancer | |
| US8329722B2 (en) | Inhibitors of janus kinases and/or 3-phosphoinositide-dependent protein kinase-1 | |
| EP2200612B1 (en) | Inhibitors of janus kinases | |
| EP2341775B1 (en) | Inhibitors of janus kinases | |
| CA2610884A1 (en) | Inhibitors of checkpoint kinases | |
| EP2032141B1 (en) | Inhibitors of janus kinases | |
| EP2611450A1 (en) | Tyrosine kinase inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08768504 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008266883 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2690141 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12665045 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2010513207 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008768504 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2008266883 Country of ref document: AU Date of ref document: 20080616 Kind code of ref document: A |