WO2008121386A2 - Calcimimetic compounds for use in the treatment of bowel disorders - Google Patents
Calcimimetic compounds for use in the treatment of bowel disorders Download PDFInfo
- Publication number
- WO2008121386A2 WO2008121386A2 PCT/US2008/004166 US2008004166W WO2008121386A2 WO 2008121386 A2 WO2008121386 A2 WO 2008121386A2 US 2008004166 W US2008004166 W US 2008004166W WO 2008121386 A2 WO2008121386 A2 WO 2008121386A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- haloalkyl
- substituted
- phenyl
- compound
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/137—Arylalkylamines, e.g. amphetamine, epinephrine, salbutamol, ephedrine or methadone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/427—Thiazoles not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/428—Thiazoles condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
Definitions
- This invention relates generally to the field of medicine and, more specifically, to methods for treating or preventing bowel disorders, in particular, inflammatory bowel disease and irritable bowel syndrome.
- IBD inflammatory bowel disease
- IBD is most common in young adults, but can occur at any age.
- the clinical symptoms of IBD include intermittent rectal bleeding, fever, abdominal pain, and diarrhea, which may range from mild to severe. Additional common signs of IBD are anemia and weight loss. 10 to 15% of all IBD patients will require surgery over ten year period. Protracted IBD is a risk factor for colon cancer, and the risk begins to rise significantly after eight to ten years of IBD.
- the first line therapy that is often used for IBD is aminosalicylates, which include sulfasalazine and the brands Asacol, Pentasa, Dipentum, and Colazal.
- Chron's disease takes a stepwise approach with nutritional supplements and 5-ASA often used as chronic therapy aimed at prophylaxis against flare-up of the disease.
- 5-ASAs are not effective in CD and start with a steroid such as budesonide.
- a steroid such as budesonide.
- immunosuppressices such as aathioprine (Imuran), 6-MP, and methotrexate are used.
- the anti-TNF antibody Remicade is also used to treat Chron's disease.
- ulcerative colitis Treatment of ulcerative colitis is very similar to treatment of Chron's disease, following a similar stepwise approach with the use of 5-ASAs, short courses of steroids, other immunosuppressives and surgery. Remicade is sometimes used for severe disease not responding to steroids or traditional immune modulators. Methotrexate and antibiotics are generally not used in UC and it's believed by many physicians that methotrexate does not work in UC. Need for surgery is more prevalent in UC than in CD, with 25-40% of patients eventually requiring colectomy. Unlike in Chron's disease, surgery for ulcerative colitis is curable. There are unmet needs for IBD patients who fail on all of the currently available therapies. Approximately 20% of patients fail all therapies and need surgery in the short term, 40% of patients will require surgery in the long term.
- IBS Irritable bowel syndrome
- IBS is the most prevalent digestive disease, accounting for 12% of visits to primary care physicians and 28% of referrals to gastroenterologists.
- IBS is one of a heterogeneous family of functional gastrointestinal disorders, which are difficult to treat because no single etiology for these disorders is known and thus treatment is directed at controlling symptoms.
- IBS affects at least 10 to 20% of adults in the US, mostly women, and second only to the common cold as a cause of absenteeism from work. The majority of cases are undiagnosed because only 25-30% of patients seek medical attention (International
- IBS Intra-Fi Protected Access to Glabras.
- IBS Intra-Fi Protected Access to Glabras.
- IBS is characterized by altered bowel habits and abdominal pain, typically in the absence of detectable structural abnormalities. No clear diagnostic markers exist for IBS, and its definition is based on its clinical presentation. IBS is often confused with IBD, colitis, mucous colitis, spastic colon, or spastic bowel. Only recently physicians started considering IBS to be a brain-gut functional disorder, rather than a somatic manifestation of physiological stress.
- the Rome diagnostic criteria of IBS can be used to rule out other disorders. According to the Rome II criteria, abdominal pain or discomfort is a prerequisite clinical feature of IBS. Drossman D. et al. (eds) Rome II: the functional gastrointestinal disorders: diagnosis, pathophysiology, and treatment: a multinational consensus. McLean, VA: Degnon Associates, 2000. Most IBS patients experience several IBS symptoms such as abdominal pain, altered bowel habits, flatulence, upper GI symptoms such as dyspepsia, heartburn nausea, and vomiting. IBS patients typically fall into two broad clinical groups. Most patients belong to the first group, presenting with abdominal pain associated with altered bowel habit that include constipation diarrhea or alternating constipation and diarrhea.
- the second group of patients have painless diarrhea. It is generally believed that the central nervous system plays an important role in the pathogenesis of IBS. This is supported by the clinical association of emotional disorders and stress with IBS symptom exacerbation, and the therapeutic response to IBS therapies that act on cerebral cortical sites.
- IBS-C constipation-predominant IBS
- Second line therapy may involve Senekot and antispasmodics (e.g., Levsin) for periodic cramping and abdominal pain.
- Third line therapy may include Zelnorm. Usually, 40% of patients with IBS-C improve with treatment. In patients with diarrhea-predominant IBS, lactose intolerance and bacterial overgrowth must be ruled out first. Imodium is standard treatment for this type of IBS.
- IBS-D diarrhea- predominant IBS
- IBS-A mixed-symptom IBS
- Bowel disorders such as IBD and IBS are a medical problem, and improved methods of treatment are necessary as no satisfactory treatments are currently available.
- the present invention provides methods for treating or preventing bowel disorders comprising administering to a subject in need thereof a therapeutically acceptable dose of a pharmaceutical composition comprising a calcimimetic compound and a pharmaceutically acceptable diluents or carrier.
- the bowel disorder can be inflammatory bowel disease (IBD).
- the IBD can be ulcerative colitis (UC).
- the IBD can be Chron's disease (CD).
- IBD can be mild, moderate, or severe.
- the methods of the invention can further comprise nutritional management.
- the nutritional management can comprise administration of calcium, magnesium, zinc, iron, folate, vitamin Bj 2 , vitamin D, or vitamin K.
- the methods of the invention can further comprise administering an antidiarrheal agent.
- the antidiarrheal agent can be loperamide or diphenoxylate.
- the methods of the invention can further comprise administering an antispasmodic agent.
- methods of the invention can further comprise administering an anticholinergic agent or an analegstic agent.
- the methods of the invention can further comprise administering a 5-aminosalicylic compound.
- the methods of the invention can further comprise administering an immunomodulator.
- the methods of the invention can further comprise administering an antibiotic.
- methods of the invention can be practiced in conjunction with a surgical treatment.
- the bowel disorder can be inflammatory bowel syndrome (IBS), hi one aspect, the IBS can be a constipation predominant IBS. In another aspect, the IBS can be a diarrhea predominant IBS or a mixed symptom IBS.
- the methods of the invention can further comprise nutritional management, hi one aspect, the nutritional management can comprise limiting fat intake, restricting intake of poorly digestible sugars, limiting of foods associated with increased flatulence, increased fiber intake, or addition of probiotics.
- the methods of the invention can further comprise administering a laxative.
- the methods of the invention can further comprise administering an opiate-based agent, an anti-spasmodic agent, an anti-depressant agent or a prokinetic agent.
- the methods of the invention can further comprise administering a peripheral dopamine receptor agonist, a hormonal treatment or a tranquilizer, hi a further aspect, the methods of the invention can further comprise psychological therapy, cognitive therapy, biofeedback and stress reduction technique, or hypnosis.
- the bowel disorder can be lymphocytic colitis, collagenous colitis, diversion colitis, endometriosis, caustic enema-induced colitis, drug-induced ischemic colitis, NSAID-induced ulcer, nonspecific ulcer, stercoral ulcer, solitary rectal ulcer, typhilitis, colitis cystica profunda, pneumatosis cystoides intestinalis, or malakoplakia.
- calcimimetic compounds useful in the methods of the present invention are described in detail in Detailed Description below.
- the subject can be mammal. In one aspect, the subject can be human.
- Figure 1 illustrates the effect of the calcimimetic compound B on colonic inflammation in 5% DSS induced colitis model.
- Panel A proximal colon
- Panel B middle colon
- Panel C distal colon
- Panel D combined colon.
- Figure 2 demonstrates that the calcimimetics Compound C (Panel A) and Compound D (Panel B) reduce inflammation scores in all parts of colon compare to the vehicle. Open bars, vehicle; black bars, calcimimetics.
- Figure 3 illustrates the changes in short-circuit secretory current in the absence and presence of 2 ⁇ M TTX, before and after addition of 10 ⁇ M of a calcimimetic Compound A, before and after the addition of 100 ⁇ M bumetanide, an inhibitor of chloride secretion.
- Figure 4 illustrates the changes in short-circuit secretory current in the absence and presence of 10 ⁇ M of Compound A, before and after addition of 2 ⁇ M TTX, before and after the addition of 100 ⁇ M bumetanide.
- Figure 5 represents clinical score on the final day of study (day 38) for controls and compound B. scores are calculated by summing the stool score (0-4) and the anal inflammation score (0-4) and averaging the scores for the group (maximal score is 8). Error bars represent SEM.
- Figure 6 represents disease onset graph. Curve representing the percentage of mice showing signs of colitis at various time points throughout the study. Day of disease onset is determined by a subject having two consecutive days of positive clinical score. The 10 mg/kg Compound B group was significantly different from the vehicle control group.
- FIG. 7 illustrates histology score. Histology scores represent the sums of scores given to the three section of colon taken: proximal, middle, and distal. The scoring system for each section is described in the methods. Error bars represent SEM.
- Figure 8 demonstrates the effect of calcimimetics on serum calcium (panel A) and phosphorus (panel B). Error bars represent SEM.
- Figure 9 demonstrates the effect of calcimimetics on serum IP-10 levels. Error bars represent SEM.
- the term "subject" is intended to mean a human, or an animal, in need of a treatment. This subject can have, or be at risk of developing, a bowel disorder, for example, inflammatory bowel disorder or irritable bowel syndrome.
- Treating" or “treatment” of a disease includes: (1) preventing the disease, i.e., causing the clinical symptoms of the disease not to develop in a subject that may be or has been exposed to the disease or conditions that may cause the disease, or predisposed to the disease but does not yet experience or display symptoms of the disease, (2) inhibiting the disease, i.e., arresting or reducing the development of the disease or any of its clinical symptoms, or (3) relieving the disease, i.e., causing regression of the disease or any of its clinical symptoms.
- Administration in combination with or “together with” one or more further therapeutic agents includes simultaneous or concurrent administration and consecutive administration in any order.
- terapéuticaally effective amount is the amount of the compound of the invention that will achieve the goal of improvement in disorder severity and the frequency of incidence.
- the improvement in disorder severity includes the reversal of the disease, as well as slowing down the progression of the disease.
- CaSR calcium sensing receptor
- Activation of the CaSR produces rapid, transient increases incytosolic calcium concentration by mobilizing calcium from thapsigargin-sensitive intracellular stores and by increasing calcium influx though voltage-insensitive calcium channels in the cell membrane (Brown et al., Nature 366: 575-580, 1993; Yamaguchi et al, Adv Pharmacol 47: 209-253, 2000).
- bowel disorders includes but is not limited to IBD, IBS, diverticular disease, collagenous and lymphatic colitis, diversion colitis, endometriosis, typhlitis, colitis, cystica profunda, pneumatosis cystoides intestinalis, and malakoplakia.
- calcimimetic compound or “calcimimetic” refers to a compound that binds to calcium sensing receptors and induces a conformational change that reduces the threshold for calcium sensing receptor activation by the endogenous ligand Ca 2+ .
- calcimimetic compounds can also be considered allosteric modulators of the calcium receptors.
- a calcimimetic can have one or more of the following activities: it evokes a transient increase in internal calcium, having a duration of less that 30 seconds (for example, by mobilizing internal calcium); it evokes a rapid increase in [Ca 2+ ( ⁇ , occurring within thirty seconds; it evokes a sustained increase (greater than thirty seconds) in [Ca 2+ j] (for example, by causing an influx of external calcium); evokes an increase in inositol- 1,4,5- triphosphate or diacylglycerol levels, usually within less than 60 seconds; and inhibits dopamine- or isoproterenol-stimulated cyclic AMP formation.
- the transient increase in [Ca 2+ i] can be abolished by pretreatment of the cell for ten minutes with 10 mM sodium fluoride or with an inhibitor of phospholipase C, or the transient increase is diminished by brief pretreatment (not more than ten minutes) of the cell with an activator of protein kinase C, for example, phorbol myristate acetate (PMA), mezerein or (-) indolactam V.
- a calcimimetic compound can be a small molecule.
- a calcimimetic can be an agonistic antibody to the CaSR.
- Calcimimetic compounds useful in the present invention include those disclosed in, for example, European Patent No. 637,237, 657,029, 724,561, 787,122, 907,631, 933,354, 1,203,761, 1,235 797, 1,258,471, 1,275,635, 1,281,702, 1,284,963, 1,296,142, 1,308,436, 1,509,497, 1,509,518, 1,553,078; International Publication Nos.
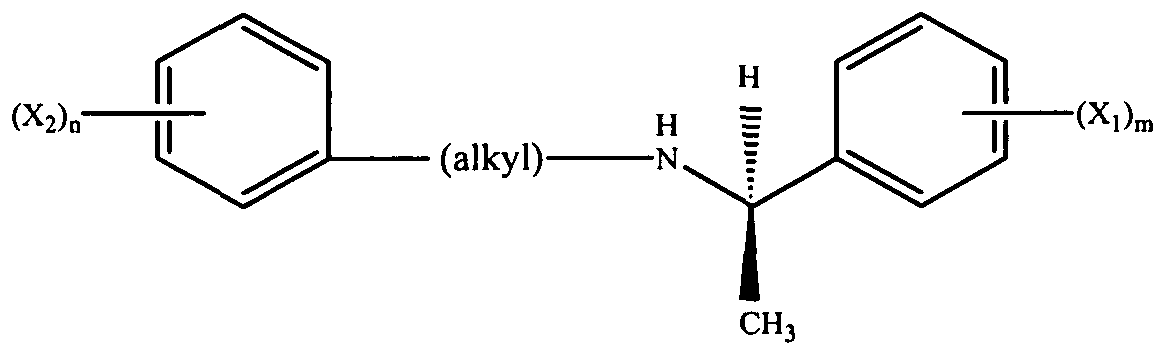
- the calcimimetic compound is chosen from compounds of Formula I and pharmaceutically acceptable salts thereof:
- Xi and X 2 which may be identical or different, are each a radical chosen from CH 3 , CH 3 O, CH 3 CH 2 O, Br, Cl, F, CF 3 , CHF 2 , CH 2 F, CF 3 O, CH 3 S, OH, CH 2 OH, CONH 2 , CN, NO 2 , CH 3 CH 2 , propyl, isopropyl, butyl, isobutyl, t-butyl, acetoxy, and acetyl radicals, or two of Xi may together form an entity chosen from fused cycloaliphatic rings, fused aromatic rings, and a methylene dioxy radical, or two of X 2 may together form an entity chosen from fused cycloaliphatic rings, fused aromatic rings, and a methylene dioxy radical; provided that X 2 is not a 3-t-butyl radical; n ranges from 0 to 5; m ranges from 1 to 5; and the alkyl radical is chosen from C
- the calcimimetic compound may also be chosen from compounds of Formula II:
- R 1 is aryl, substituted aryl, heterocyclyl, substituted heterocyclyl, cycloalkyl, or substituted cycloalkyl;
- R 2 is alkyl or haloalkyl;
- R 3 is H, alkyl, or haloalkyl
- R 6 is aryl, substituted aryl, heterocyclyl, substituted heterocyclyl, cycloalkyl, or substituted cycloalkyl; each R a is, independently, H, alkyl or haloalkyl; each R b is, independently, aryl, aralkyl, heterocyclyl, or heterocyclylalkyl, each of which may be unsubstituted or substituted by up to 3 substituents selected from the group consisting of alkyl, halogen, haloalkyl, alkoxy, cyano, and nitro; each R c is, independently, alkyl, haloalkyl, phenyl or benzyl, each of which may be substituted or unsubstituted; each R d is, independently, H, alkyl, aryl, aralkyl, heterocyclyl, or heterocyclylalkyl wherein the alkyl , aryl, aralkyl, heterocyclyl,
- the calcimimetic compound can be N-((6-(methyloxy)-4'- (trifluoromethyl)-l,r-biphenyl-3-yl)methyl)-l-phenylethanamine, or a pharmaceutically acceptable salt thereof, hi another aspect, the calcimimetic compound can be (lR)-N-((6- chloro-3'-fluoro-3-biphenylyl)methyl)-l-(3-chlorophenyl)ethanamine, or a pharmaceutically acceptable salt thereof.
- the calcimimetic compound can be (IR)-I -(6- (methyloxy)-4'-(trifluoromethyl)-3-biphenylyl)-N-((l R)- 1 -phenyl ethyl)ethanamine, or a pharmaceutically acceptable salt thereof.
- ⁇ represents a double or single bond
- R 1 is R b ;
- R 2 is Ci -8 alkyl or Ci -4 haloalkyl
- R 3 is H, C M haloalkyl or Ci -8 alkyl
- R 4 is H, Ci -4 haloalkyl or Ci -4 alkyl
- R 6 and R 7 together form a 3- to 6-atom saturated or unsaturated bridge containing 0, 1 , 2 or 3 N atoms and 0, 1 or 2 atoms selected from S and O, wherein the bridge is substituted by 0, 1 or 2 substituents selected from R 5 ; wherein when R 6 and R 7 form a benzo bridge, then the benzo bridge may be additionally substituted by a 3- or 4- atoms bridge containing 1 or 2 atoms selected from N and O, wherein the bridge is substituted by 0 or 1 substituents selected from Ci -4 alkyl;
- R a is, independently, at each instance, H, C ⁇ haloalkyl or C 1-6 alkyl
- R b is, independently, at each instance, phenyl, benzyl, naphthyl or a saturated or unsaturated 5- or 6-membered ring heterocycle containing 1, 2 or 3 atoms selected from N, O and S, with no more than 2 of the atoms selected from O and S, wherein the phenyl, benzyl or heterocycle are substituted by 0, 1, 2 or 3 substituents selected from Ci -6 alkyl, halogen, Ci ⁇ haloalkyl, -OCi -6 alkyl, cyano and nitro;
- R c is, independently, at each instance, Ci -6 alkyl, C M haloalkyl, phenyl or benzyl
- a calcimimetic compound is N-(3-[2-chlorophenyl]-propyl)-R- ⁇ -methyl-
- a calcimimetic compound is N- ((6-(methyloxy)-4'-(trifluoromethyl)- 1 , 1 '-biphenyl-3-yl)methyl)- 1 -phenylethanamine (Compound B).
- the calcimimetic compound of the invention can be chose from compounds of Formula IV
- Y is oxygen or sulphur
- Ri and R'i are the same or different, and each represents an aryl group, a heteroaryl group, or Rj and R' I , together with the carbon atom to which they are linked, form a fused ring structure of formula:
- A represents a single bond, a methylene group, a dimethyl ene group, oxygen, nitrogen or sulphur, said sulphur optionally being in the sulphoxide or sulphone forms, wherein each of Ri and R'i, or said fused ring structure formed thereby, is optionally substituted by at least one substituent selected from the group c, wherein the group c consists of: halogen atoms, hydroxyl, carboxyl, linear and branched alkyl, hydroxyalkyl, haloalkyl, alkylthio, alkenyl, and alkynyl groups; linear and branched alkoxyl groups; linear and branched thioalkyl groups; hydroxycarbonylalkyl; alkylcarbonyl; alkoxycarbonylalkyl; alkoxycarbonyl; trifluoromethyl; trifluoromethoxyl; -CN; - NO 2 ; alkyl sulphonyl groups optionally in
- R 2 and R 2 which may be the same or different, each represents: a hydrogen atom ; a linear or branched alkyl group containing from 1 to 6 carbon atoms and optionally substituted by at least one halogen atom, hydroxy or alkoxy group containing from 1 to 6 carbon atoms; an alkylaminoalkyl or dialkylaminoalkyl group wherein each alkyl group contains from 1 to 6 carbon atoms, or R 2 and R 2 , together with the nitrogen atom to which they are linked, form a saturated or unsaturated heterocycle containing 0, 1 or 2 additional heteroatoms and having 5, 6, or 7 ring atoms, said heterocycle being optionally substituted by at least one substituent selected from the group 'c' defined above, and wherein, when there is more than one substituent, said substituent is the same or different,
- R 3 represents a group of formula:
- B represents an oxygen atom or a sulphur atom
- x is 0, 1 or 2
- y and y' are the same or different, and each is 0 or 1
- Ar and Ar' are the same or different and each represents an aryl or heteroaryl group
- n and n' are the same or different, and each is 1, when the y or y' with which it is associated is 0, or is equal to the number of positions that can be substituted on the associated Ar or Ar' when the said y or y' is 1
- the fused ring containing N x is a five- or six- membered heteroaryl ring
- R and R' which may be the same or different, each represent a hydrogen atom or a substituent selected from the group ⁇ , wherein the group a consists of: halogen atoms; hydroxyl; carboxyl; aldehyde groups; linear and branched alkyl, alkenyl, alkynyl, hydroxyalkyl, hydroxyal
- the calcimimetic compound can be 3-(l,3-benzothiazol-2-yl)-l-(3,3- diphenylpropyl)-l-(2-(4-morpholinyl)ethyl)urea or pharmaceutically acceptable salt thereof.
- the calcimimetic compound can be N-(4-(2-((((3,3-diphenylpropyl)(2-(4- morpholinyl)ethyl)amino)carbonyl)amino)- 1 ,3-thiazol-4-yl)phenyl)methanesulfonamide or pharmaceutically acceptable salt thereof.
- the calcimimetic compound of the invention can be chose from compounds of Formula V
- R 1 is phenyl, benzyl, naphthyl or a saturated or unsaturated 5- or 6-membered heterocyclic ring containing 1, 2 or 3 atoms selected from N, O and S, with no more than 2 of the atoms selected from O and S, wherein the phenyl, benzyl, naphthyl or heterocyclic ring are substituted by 0, 1, 2 or 3 substituents selected from Ci -6 alkyl, halogen, -OCi -6 alkyl, cyano and nitro;
- R 2 is Ci -8 alkyl or C ⁇ haloalkyl
- R 3 is H, Ci ⁇ haloalkyl or Ci -8 alkyl
- R 4 is H, C M haloalkyl or Ci -8 alkyl
- R a is, independently, at each instance, H, Ci ⁇ alkyl, Ci -6 alkenyl, C 1-6 alkylaryl or arylCi -6 alkyl:
- R b is, independently, at each instance, Cugalkyl, C 1-4 haloalkyl, phenyl, benzyl, naphthyl or a saturated or unsaturated 5- or 6-membered heterocyclic ring containing 1 , 2 or 3 atoms selected from N, O and S, with no more than 2 of the atoms selected from O and S, wherein the phenyl, benzyl, naphthyl or heterocyclic ring are substituted by 0, 1 , 2 or 3 substituents selected from Ci -6 alkyl, halogen, -OCi -6 alkyl, cyano and nitro;
- R c is, independently, at each instance, Cj -6 alkyl, C ⁇ haloalkyl, phenyl or benzyl;
- the calcimimetic compound can be N-(2-chloro-5-(((( IR)-I- phenylethyl)amino)methyl)phenyl)-5-methyl-3-isoxazolecarboxamide or a pharmaceutically acceptable salt thereof.
- the calcimimetic compound can be N-(2-chloro-5- ((((lR)-l-phenylethyl)amino)methyl)phenyl)-2-pyridinecarboxamide or a pharmaceutically acceptable salt thereof.
- Calcimimetic compounds useful in the methods of the invention include the calcimimetic compounds described above, as well as their stereoisomers, enantiomers, polymorphs, hydrates, and pharmaceutically acceptable salts of any of the foregoing.
- compounds binding at the CaSR-activity modulating site can be identified using, for example, a labeled compound binding to the site in a competition-binding assay format.
- Calcimimetic activity of a compound can be determined using techniques such as those described in International Publications WO 93/04373, WO 94/18959 and WO 95/11211.
- HEK 293 Cell Assay HEK 293 cells engineered to express human parathyroid CaSR (HEK 293 4.0-7) have been described in detail previously (Nemeth EF et al. (1998) Proc. Natl. Acad. Sci. USA 95:4040-4045). This clonal cell line has been used extensively to screen for agonists, allosteric modulators, and antagonists of the CaSR (Nemeth EF et al. (2001) J. Pharmacol. Exp. Ther. 299:323-331).
- the cells are recovered from tissue culture flasks by brief treatment with 0.02% ethylenediaminetetraacetic acid (EDTA) in phosphate-buffered saline (PBS) and then washed and resuspended in Buffer A (126 mM NaCl, 4 mM KCl, 1 mM CaCl 2 , 1 mM MgSO 4 , 0.7 mM K 2 HPO 4 /KH 2 PO 4 , 20 mM Na-Hepes, pH 7.4) supplemented with 0.1% bovine serum albumin (BSA) and 1 mg/ml D-glucose.
- BSA bovine serum albumin
- the cells are loaded with fura-2 by incubation for 30 minutes at 37°C in Buffer A and 2 ⁇ M fura-2 acetoxymethylester.
- the cells are washed with Buffer B (Buffer B is Buffer A lacking sulfate and phosphate and containing 5 mM KCl, 1 mM MgCl 2 , 0.5 mM CaCl 2 supplemented with 0.5% BSA and 1 mg/ml D-glucose) and resuspended to a density of 4 to 5 x 10 6 cells/ml at room temperature.
- Buffer B is Buffer A lacking sulfate and phosphate and containing 5 mM KCl, 1 mM MgCl 2 , 0.5 mM CaCl 2 supplemented with 0.5% BSA and 1 mg/ml D-glucose
- Excitation and emission wavelengths are 340 and 510 ran, respectively.
- the fluorescent signal is recorded in real time using a strip- chart recorder
- HEK 293 cells are maintained in Dulbecco's modified Eagle's medium (DMEM) with 10% fetal bovine serum (FBS) and 200 ⁇ g/ml hygromycin.
- DMEM Dulbecco's modified Eagle's medium
- FBS fetal bovine serum
- the cells are trypsinized and plated in the above medium at 1.2 x 10 5 cells/well in black sided, clear-bottom, collagen 1 -coated, 96- well plates. The plates are centrifuged at 1,000 rpm for 2 minutes and incubated under 5% CO 2 at 37°C overnight. Cells are then loaded with 6 ⁇ M fluo-3 acetoxymethylester for 60 minutes at room temperature.
- All assays are performed in a buffer containing 126 mM NaCl, 5 mM KCl, 1 mM MgCl 2 , 20 mM Na-Hepes, supplemented with 1.0 mg/ml D-glucose and 1.0 mg/ml BSA fraction IV (pH 7.4).
- the EC 5 o's for the CaSR-active compounds can be determined in the presence of 1 mM Ca 2+ .
- the EC 5O for cytoplasmic calcium concentration can be determined starting at an extracellular Ca 2+ level of 0.5 mM.
- FLIPR experiments are done using a laser setting of 0.8 W and a 0.4 second CCD camera shutter speed. Cells are challenged with calcium, CaSR-active compound or vehicle (20 ⁇ l) and fluorescence monitored at 1 second intervals for 50 seconds. Then a second challenge (50 ⁇ l) of calcium, CaSR-active compound, or vehicle can be made and the fluorescent signal monitored. Fluorescent signals are measured as the peak height of the response within the sample period. Each response is then normalized to the maximum peak observed in the plate to determine a percentage maximum fluorescence.
- Bovine Parathyroid Cells are challenged with calcium, CaSR-active compound or vehicle (20 ⁇ l) and fluorescence monitored at 1 second intervals for 50 seconds. Then a second challenge (50 ⁇ l) of calcium
- Dissociated bovine parathyroid cells can be obtained by collagenase digestion, pooled, then resuspended in Percoll purification buffer and purified by centrifugation at 14,500 x g for 20 minutes at 4°C.
- the dissociated parathyroid cells are removed and washed in a 1 : 1 mixture of Ham's F-12 and DMEM (F- 12/DMEM) supplemented with 0.5% BSA, 100 U/ml penicillin, 100 ⁇ g/ml streptomycin, and 20 ⁇ g/ml gentamicin.
- the cells are finally resuspended in F-12/DMEM containing 10 U/ml penicillin, 10 ⁇ g/ml streptomycin, and 4 ⁇ g/ml gentamicin, and BSA was substituted with ITS+ (insulin, transferrin, selenous acid, BSA, and linoleic acid; Collaborative Research, Bedford, MA).
- ITS+ insulin, transferrin, selenous acid, BSA, and linoleic acid
- the cells are removed from flasks by decanting and washed with parathyroid cell buffer (126 mM NaCl, 4 mM KCl, 1 mM MgSO 4 , 0.7 mM K 2 HPO 4 ZKH 2 PO 4 , 20 mM Na-Hepes, 20; pH 7.45 and variable amounts of CaCl 2 as specified) containing 0.1% BSA and 0.5 mM CaCl 2 .
- the cells are resuspended in this same buffer and portions (0.3 ml) are added to polystyrene tubes containing appropriate controls, CaSR-active compound, and/or varying concentrations of CaCl 2 . Each experimental condition is performed in triplicate.
- Rat MTC 6-23 cells (clone 6), purchased from ATCC (Manassas, VA) are maintained in growth media (DMEM high glucose with calcium/15% HIHS) that is replaced every 3 to 4 days. The cultures are passaged weekly at a 1 :4 split ratio. Calcium concentration in the formulated growth media is calculated to be 3.2 mM. Cells are incubated in an atmosphere of 90% O 2 A 0% CO 2 , at 37°C. Prior to the experiment, cells from sub-confluent cultures are aspirated and rinsed once with trypsin solution. The flasks are aspirated again and incubated at room temperature with fresh trypsin solution for 5-10 minutes to detach the cells.
- DMEM high glucose with calcium/15% HIHS calcium/15% HIHS
- the detached cells are suspended at a density of 3.0 x 10 5 cells/mL in growth media and seeded at a density of 1.5 x 10 5 cells/well (0.5 mL cell suspension) in collagen-coated 48 well plates (Becton Dickinson Labware, Bedford, MA).
- the cells are allowed to adhere for 56 hours post- seeding, after which the growth media was aspirated and replaced with 0.5 mL of assay media (DMEM high glucose without/2% FBS).
- the cells are then incubated for 16 hours prior to determination of calcium-stimulated calcitonin release.
- the actual calcium concentration in this media is calculated to be less than 0.07 mM.
- calcitonin release 0.35 mL of test agent in assay media is added to each well and incubated for 4 hours prior to determination of calcitonin content in the media. Calcitonin levels are quantified according to the vendor's instructions using a rat calcitonin immunoradiometric assay kit (Immutopics, San Clemente, CA).
- CHO(CaSR) Chinese hamster ovarian cells transfected with an expression vector containing cloned CaSR from rat brain [CHO(CaSR)] or not [CHO(WT)] (Ruat M., Snowman AM., J. Biol. Chem 271, 1996, p 5972).
- CHO(CaSR) has been shown to stimulate tritiated inositol phosphate ([ 3 H]IP) accumulation upon activation of the CaSR by Ca 2+ and other divalent cations and by NPS 568 (Ruat et al, J. Biol. Chem 271, 1996).
- [ 3H ]IP accumulation produced by 10 ⁇ M of each CaSR-active compound in the presence of 2 mM extracellular calcium can be measured and compared to the effect produced by 10 mM extracellular calcium, a concentration eliciting maximal CaSR activation (Dauban P. et al., Bioorganic & Medicinal Chemistry Letters, 10, 2000, p 2001).
- concentration eliciting maximal CaSR activation Daban P. et al., Bioorganic & Medicinal Chemistry Letters, 10, 2000, p 2001.
- Calcimimetic compounds useful in the present invention can be used in the form of pharmaceutically acceptable salts derived from inorganic or organic acids.
- the salts include, but are not limited to, the following: acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, camphorsulfonate, digluconate, cyclopentanepropionate, dodecylsulfate, ethanesulfonate, glucoheptanoate, glycerophosphate, hemisulfate, heptanoate, hexanoate, fumarate, hydrochloride, hydrobromide, hydroiodide, 2- hydroxy-ethanesulfonate, lactate, maleate, mandelate, methansulfonate, nicotinate, 2- naphthalenesulfonate, oxalate, palm
- salts for the carboxy group are well known to those skilled in the art and include, for example, alkaline, alkaline earth, ammonium, quaternary ammonium cations and the like.
- suitable pharmaceutically acceptable salts see Berge et al. J. Pharm. Sci. 66: 1, 1977.
- salts of hydrochloride and salts of methanesulfonic acid can be used.
- the calcium-receptor active compound can be chosen from cinacalcet, i.e., N-(l-(R)-(l-naphthyl)ethyl]-3-[3-(trifluoromethyl)phenyl]-l- aminopropane, cinacalcet HCl, and cinacalcet methanesulfonate.
- the calcimimetic compound such as cinacalcet HCl and cinacalcet methanesulfonate, can be in various forms such as amorphous powders, crystalline powders, and mixtures thereof.
- the crystalline powders can be in forms including polymorphs, psuedopolymorphs, crystal habits, micromeretics, and particle morphology.
- the compounds useful in this invention are ordinarily combined with one or more adjuvants appropriate for the indicated route of administration.
- the compounds may be admixed with lactose, sucrose, starch powder, cellulose esters of alkanoic acids, stearic acid, talc, magnesium stearate, magnesium oxide, sodium and calcium salts of phosphoric and sulphuric acids, acacia, gelatin, sodium alginate, polyvinyl-pyrrolidine, and/or polyvinyl alcohol, and tableted or encapsulated for conventional administration.
- the compounds useful in this invention may be dissolved in saline, water, polyethylene glycol, propylene glycol, ethanol, corn oil, peanut oil, cottonseed oil, sesame oil, tragacanth gum, and/or various buffers.
- Other adjuvants and modes of administration are well known in the pharmaceutical art.
- the carrier or diluent may include time delay material, such as glyceryl monostearate or glyceryl distearate alone or with a wax, or other materials well known in the art.
- the pharmaceutical compositions may be made up in a solid form (including granules, powders or suppositories) or in a liquid form (e.g., solutions, suspensions, or emulsions).
- the pharmaceutical compositions may be subjected to conventional pharmaceutical operations such as sterilization and/or may contain conventional adjuvants, such as preservatives, stabilizers, wetting agents, emulsifiers, buffers etc.
- Solid dosage forms for oral administration may include capsules, tablets, pills, powders, suppositories, and granules.
- the active compound may be admixed with at least one inert diluent such as sucrose, lactose, or starch.
- Such dosage forms may also comprise, as in normal practice, additional substances other than inert diluents, e.g., lubricating agents such as magnesium stearate.
- the dosage forms may also comprise buffering agents. Tablets and pills can additionally be prepared with enteric coatings.
- Liquid dosage forms for oral administration may include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs containing inert diluents commonly used in the art, such as water. Such compositions may also comprise adjuvants, such as wetting, sweetening, flavoring, and perfuming agents.
- the therapeutically effective amount of the calcium receptor-active compound in the compositions useful in the invention can range from about 0.1 mg to about 180 mg, for example from about 5 mg to about 180 mg, or from about lmg to about 100 mg of the calcimimetic compound per subject, hi some aspects, the therapeutically effective amount of calcium receptor-active compound in the composition can be chosen from about 0.1 mg, about 1 mg, 5 mg, about 15 mg, about 20 mg, about 30 mg, about 50 mg, about 60 mg, about 75 mg, about 90 mg, about 120 mg, about 150 mg, about 180 mg.
- a pharmaceutical composition of the invention may comprise a therapeutically effective amount of at least one calcimimetic compound, or an effective dosage amount of at least one calcimimetic compound.
- an "effective dosage amount” is an amount that provides a therapeutically effective amount of the calcium receptor-active compound when provided as a single dose, in multiple doses, or as a partial dose.
- an effective dosage amount of the calcium receptor-active compound of the invention includes an amount less than, equal to or greater than an effective amount of the compound; for example, a pharmaceutical composition in which two or more unit dosages, such as in tablets, capsules and the like, are required to administer an effective amount of the compound, or alternatively, a multidose pharmaceutical composition, such as powders, liquids and the like, in which an effective amount of the calcimimetic compound is administered by administering a portion of the composition.
- a pharmaceutical composition in which two or more unit dosages, such as in tablets, capsules and the like, are required to administer an effective amount of the calcium receptor-active compound may be administered in less than an effective amount for one or more periods of time (e.g., a once-a-day administration, and a twice-a-day administration), for example to ascertain the effective dose for an individual subject, to desensitize an individual subject to potential side effects, to permit effective dosing readjustment or depletion of one or more other therapeutics administered to an individual subject, and/or the like.
- the effective dosage amount of the pharmaceutical composition useful in the invention can range from about 1 mg to about 360 mg from a unit dosage form, for example about 5 mg, about 15 mg, about 30 mg, about 50 mg, about 60 mg, about 75 mg, about 90 mg, about 120 mg, about 150 mg, about 180 mg, about 210 mg, about 240 mg, about 300 mg, or about 360 mg from a unit dosage form.
- the compositions disclosed herein comprise a therapeutically effective amount of a calcium receptor-active compound for the treatment or prevention of bowel disorders.
- the calcimimetic compound such as cinacalcet HCl can be present in an amount ranging from about 1 % to about 70%, such as from about 5% to about 40%, from about 10% to about 30%, or from about 15% to about 20%, by weight relative to the total weight of the composition.
- compositions useful in the invention may contain one or more active ingredients in addition to the calcium sensing receptor-active compound.
- the additional active ingredient may be another calcimimetic compound, or it may be an active ingredient having a different therapeutic activity.
- additional active ingredients include vitamins and their analogs, such as antibiotics, lanthanum carbonate, anti-inflammatory agents (steroidal and nonsteroidal) and inhibitors of pro-inflammatory cytokine (ENBREL®, KJNERET®).
- the therapeutic agents can be formulated as separate compositions that are given at the same time or different times, or the therapeutic agents can be given as a single composition.
- the pharmaceutical compositions useful for methods of the invention may include additional compounds as described in more detail below.
- the compounds used to practice the methods of the instant invention can be formulated for oral administration that release biologically active ingredients in the colon without substantial release into the upper gastrointestinal tract, e.g. stomach and intestine.
- Oral delivery of drugs to the colon can allow achieving high local concentration while minimizing side effects that occur because of release of drugs in the upper GI tract or unnecessary systemic absorption.
- the advantage of colonic delivery of drugs can be due to the fact that poorly absorbed drugs may have an improved bioavailability, colon is somewhat less hostile environments with less diversity and intensity of activity that the stomach and small intestine, and the colon has a longer retention time and appears highly responsive to agents that enhance the absorption of poorly absorbed drugs. Chourasia, M. et al. (2003) J. Pharm. Pharmaceut. Sci 6(1): 33-66.
- Cyclodextrin conjugates The drug is conjugated with cyclodextrin
- Glycoside conjugates The drug is conjugated with glycisode
- Glucoronate conjugates The drug is conjugated with glucoronate
- Polypetide conjugates The drug is conjugated with poly(aspartic acid)
- Redox-sensitive polymers Drug formulated with azo polymer and disulfide polymers that selectively respond to the redox potential of the colon provides colonic delivery
- Bioadhesive systems Drug coated with a bioadhesive polymer that selectively provides adhesion to the colonic mucosa may release drug in the colon
- Osmotic controlled drug delivery Drug is released through semipermeable membrane due to osmotic pressure
- Suppositories Drug is released from microscopic polymeric particles wherein drug is homogeneously incorporated within a pharmaceutically acceptable suppository base.
- compositions of the invention can be used with the drug carrier including pectin and galactomannan, polysaccaharides that are both degradable by colonic bacterial enzymes (US Pat No. 6,413,494). While pectin or galactomannan, if used alone as a drug carrier, are easily dissolved in simulated gastric fluid and simulated intestinal fluid, a mixture of these two polysaccharides prepared at a pH of about 7 or above produces a strong, elastic, and insoluble gel that is not dissolved or disintegrated in the simulated gastric and intestinal fluids, thus protecting drugs coated with the mixture from being released in the upper GI tract.
- pectin or galactomannan if used alone as a drug carrier, are easily dissolved in simulated gastric fluid and simulated intestinal fluid, a mixture of these two polysaccharides prepared at a pH of about 7 or above produces a strong, elastic, and insoluble gel that is not dissolved or disintegrated in the simulated gastric and intestinal fluids, thus protecting drugs
- compositions of the invention may be used with the pharmaceutical matrix of a complex of gelatin and an anionic polysaccharide (e.g., pectinate, pectate, alginate, chondroitin sulfate, polygalacturonic acid, tragacanth gum, arabic gum, and a mixture thereof), which is degradable by colonic enzymes (US Pat No. 6,319,518).
- an anionic polysaccharide e.g., pectinate, pectate, alginate, chondroitin sulfate, polygalacturonic acid, tragacanth gum, arabic gum, and a mixture thereof
- bowels disorders include inflammatory bowel disease
- bowel disorders include irritable bowel syndrome.
- bowel disorders include diverticular disease, collagenous and lymphatic colitis, diversion colitis, endometriosis, typhlitis, colitis, cystica profunda, pneumatosis cystoides intestinalis, or malakoplakia.
- the present invention provides method of treatment or prevention of inflammatory bowel disease.
- Inflammatory bowel disease or IBD, as used herein, is a disease characterized by inflammation or ulcerations in the small and/or large intestine with chronically recurring symptoms of abdominal pain and alteration in bowel habits.
- IBD has been classified into the broad categories of Chron's disease (CD) and ulcerative colitis (UC). Though patients with CD and UC may present with similar symptoms such as diarrhea and abdominal pain, the characteristic differences are listed in Table 2.
- the invention provides methods for treating UC using calcimimetic compounds and compositions.
- the methods of the invention can be used for treatment of CD using calcimimetic compounds and compositions.
- methods of the present invention result in prevention of onset or alleviation of one or more signs or symptoms of UC or CD.
- Table 3 further summarizes inflammatory markers in pathophysiology of IBD and signs /symptoms commonly found in ulcerative colitis and Chron's disease.
- IBD IBD-related disease
- a careful medical history is taken, including details and durations of symptoms, and whether there is a family history of IBD or cigarette smoking history.
- blood tests can help detect anemia, high white cell counts (indicating inflammation or infection), and low nutrient levels.
- Stool samples can rule out intestinal infection which can lead to similar symptoms as IBD.
- the diagnosis of IBD is established by finding characteristic changes in the lining of the intestinal tract by endoscopy and characteristic histological features on biopsy, which can be obtained via endoscopy.
- X-ray examinations such as double-contrast barium enemas or oral barium with small bowel follow-through ("upper GI series") are used to confirm the diagnosis following endoscopy and examine the extent of mucosal disease.
- CT or MRI scans are occasionally ordered to look for complications of IBD, such as obstruction.
- Current area of investigation includes genetic testing and video capsule endoscopy.
- wireless capsule endoscopy a pill is swallowed and travels through the small intestine taking pictures that are transmitted to a recorder and later viewed on a computer. This test is more sensitive for CD in the small intestine than X-rays.
- the invention provides methods for treating mild IBD. In another aspect, the invention provides methods for treating moderate IBD. In a further aspect, the invention provides methods for treating severe IBD.
- the severity of inflammatory bowel disease is characterized in Table 4.
- methods of the present invention can be practiced in combination with nutritional management. Deficiencies of specific nutrients in IBD can be managed by supplementation.
- calcimimetic compounds and compositions can be administered in combination with calcium, magnesium, zinc, iron, folate, vitamin B 12 , vitamin D, or vitamin K.
- compound and compositions of the invention can be administered concurrently with cholestyramine therapy.
- compounds and compositions of the invention can be administered with antidiarrheal agents, antispasmodics or anticholinergics, or analgesics. Examples of antidiarrheal agents include loperamide or diphenoxylate.
- Examples of anticholinergics include belladonna, clidinium, propantheline bromides, and dicyclomine hydrochloride.
- An analgesic can be chosen based on the disease severity.
- compounds and compositions of the invention can be administered together with 5-aminosalicylic acid compounds (5-ASA).
- 5-ASA preparations include mesalamine (5-ASA) suppositories, 5-ASA enemas, sulfasalazine, asacol, pentasa, dipentum and colazol.
- compounds and compositions of the invention can be administered concurrently with steroids, such as cortisone, corticosteroid enemas, budesonide, or prednisone.
- compounds and compositions of the invention can be administered together with immunomodulators (i.e., drugs that act by blocking lymphocyte proliferation, activation, or effector mechanisms).
- immunomodulators include 6- mercaptopurine (6-MP), azathioprine, methotrexate, cyclosporine, or anti-TNF antibodies such as infliximab (Remicade®) or CDP571 (humanized IgG4 antibody against TNF- ⁇ ).
- compounds and compositions of the invention can be administered concurrently with antibiotics, such as metronidazole or ciproflaxin.
- compounds and compositions of the invention can be administered concurrently with Humira®.
- compounds and compositions of the invention can be administered together with
- CNTO- 1275 (Centocor).
- compounds and compositions of the invention can be administered concurrently with Basiliximab (Simulect), FK506 or sargramostim.
- compounds and compositions of the invention can be administered in conjunction with surgical treatments.
- calcimimetic compounds and compositions of the invention can be administered concurrently with treatment involving recombinant cytokines or antibodies to cytokines.
- a combination therapy may include recombinant IL-IO.
- calcimimetic compounds and compositions of the invention can be used concurrently with probiotics.
- the calcimimetic compounds of the present invention can be administered together with compounds or treatments that block the proinflammatory properties of CpG motifs, such as CpG-ODN or
- calcimimetic compounds and compositions of the invention can be used with heparin, lidocaine, or rosiglitazone.
- the present invention provides method of treatment or prevention of irritable bowel syndrome.
- Irritable bowel syndrome or IBS, as used herein, is a gastrointestinal disorder characterized by altered bowel habits and abdominal pain, typically in the absence of detectable structural abnormalities or biochemical cause.
- the Rome II criteria can be used to diagnose IBS and rule out other disorders.
- the criteria include at least 3 months of the following continuous recurrent symptoms: abdominal pain or discomfort that is relieved by defecation or is associated with a change in the frequency or consistency of stool, and disturbed defecation involving two or more of the following characteristics at least 25% of the time: altered stool frequency, altered stool form (e.g., lumpy or hard, or loose or watery), altered stool passage (e.g., straining, urgency, or feeling of incomplete evacuation), passage of mucus, bloating or feeling of abdominal distention.
- the intensity and location of abdominal pain in IBS can be highly variable, even within an individual patient: it is localized to the hypogastrium in 25%, the right side in 20%, the left side in 20%, and the epigastrium in 10% of the patients.
- the pain can be generally crampy or achy, although sharp, dull, gas-like, or non- descript pains are also common, hi one aspect, patients with IBS may present with constipation (IBS-C, constipation predominant IBS), diarrhea (IBS-D, diarrhea-predominant IBS), or constipation alternating with diarrhea (IBS-A, mixed symptom IBS, or "alternators"). Long period of straining may be required for fecal evaluation both in constipation- and diarrhea-predominant patient. Constipation may persist for weeks to months, interrupted by brief periods of diarrhea. Feelings of incomplete fecal evacuation may lead to multiple attempts at stool passage daily. In patients with IBS-D, stools are characteristically loose and frequent but of normal daily volume.
- Mucus discharge has been reported in up to 50% of patients with IBS. Upper gut symptoms are common in IBS, with 25% to 50% of patients reporting heartburn, early satiety, nausea, and vomiting, up to 87% note intermittent dyspepsia. Agreus L. et al. (1995) Gastroenterology 109: 671.
- Extraintestinal complaints in patients with IBS include chronic pelvic pain, fibromyalgia, genitourinary dysfunctions, such as dysmenorrheal, dyspareunia, impotence, urinary frequency, nocturia, and a sensation of incomplete bladder emptying. Impaired sexual function is reported by 83% of patients with IBS. Patients with functional bowel disorders have higher incidences of hypertension, headaches, peptic ulcer disease, rashes than the general population and more commonly report fatigue, loss of concentration, insomnia, palpitations, and unpleasant tastes in the mouth.
- IBS ulcerative colitis
- gut motor and sensory activity has been associated with colonic and small intestinal motility abnormalities, as well as with motor abnormalities in other smooth muscle sites.
- the visceral sensory abnormalities which maybe responsible for sensations of pain, gas, or bloating in IBS, have been a major focus of investigation.
- Perception of abdominal symptoms is mediated by afferent neural pathways which are activated by visceral stimuli acting on chemoreceptors, mechanoreceptors, and receptors in the mesentery which may play a role in painful stimulation of the gut.
- IBS Information from these activated receptors is carried in spinal afferent nerves and thus transmitted to the brain where conscious perception occurs. It is postulated that IBS results from sensitization of afferent pathways such that normal physiological gut stimuli not perceived by healthy individuals induce pain in the patient with IBS. The sensitizing event responsible for induction of symptoms in IBS is unknown. The clinical association of emotional disorders and stress with symptom exacerbation and the therapeutic response to therapies that act on cerebral cortical sites strongly suggests the role of central nervous system factors in the pathogenesis of IBS. However, it is unclear whether IBS represents a primary gut disturbance with inappropriate input from the central nervous system or a central nervous system disorder with centrally directed changes in gut motor and sensory activity.
- the methods of the invention can be practiced concurrently with dietary modifications.
- These modifications can include, for example, limiting fat intake or restricting intake of poorly digestible sugars such as fructose and sorbitol, or exclusion or limiting of foods associated with increased flatulence, e.g., beans, onions, celery, carrots, raisins, bananas, apricots, prunes, brussel sprouts, pretzels, wheat germ, and bagels.
- the modifications may include increased fiber intake.
- Fiber supplements can include bran, Metamucil, psyllium, process flea seed husk (ispaghula), and calcium polycarbophyl.
- probiotics can be used to repopulate the gut with good bacteria.
- osmotic laxatives can be used to effect defecation.
- These laxatives include hypertonic salt solution such as milk of magnesia, poorly absorbable sugars such as lactulose and sorbitol, and isotonic electrolyte solutions containing polyethylene glycol.
- hypertonic salt solution such as milk of magnesia
- poorly absorbable sugars such as lactulose and sorbitol
- isotonic electrolyte solutions containing polyethylene glycol for diarrhea-predominant IBS
- opiate-based agents can be used, such as loperamide, Imodium, bile acid-sequestering drugs, acid-suppressing drugs in the H 2 receptor agonist and proton pump inhibitor classes.
- anti-spasmodic agents such as drugs that block cholinergic nerve function (e.g., dicyclomine, prifinium, cimetropiuim, zamifenacin), agents that prevent calcium flux (e.g., dilatiazem, pinaverium, octylonium, peppermint oil), and direct gut smooth muscle relaxants, as well as agents that act via unknown pathways.
- drugs that block cholinergic nerve function e.g., dicyclomine, prifinium, cimetropiuim, zamifenacin
- agents that prevent calcium flux e.g., dilatiazem, pinaverium, octylonium, peppermint oil
- compounds and compositions of the invention can be used in the treatment of IBS with anti-depressant agents, for example, agents in the tricyclic class, such as amitriotyline, trimipramine, desipramine, nortriotyline, fluphenazine; the selective serotonin reuptake inhibitors, e.g., paroxetine, citalopram, mianserin; or serotonin receptor antagonists, e.g., ondansertron, granisetron, alosertron, or 5HT 4 receptor antagonist SB-207266-A.
- agents in the tricyclic class such as amitriotyline, trimipramine, desipramine, nortriotyline, fluphenazine
- the selective serotonin reuptake inhibitors e.g., paroxetine, citalopram, mianserin
- serotonin receptor antagonists e.g., ondansertron, granisetron, alosertron, or 5
- the invention provides methods for treating IBS in conjunction with other medications, for example, prokinetic medications, such as tegaserod, peripheral dopamine receptor antagonists, such as domperidone; hormonal treatments (for example, gonadotropin- releasing hormone, such as leuprolide; tranquilizers, such as phenaglycodol, meprobamate, heteronium plus amobarbital, propantheline plus phenobarbital, chlordiazepoxide, diazepam, medazepam, and alprazolam.
- prokinetic medications such as tegaserod
- peripheral dopamine receptor antagonists such as domperidone
- hormonal treatments for example, gonadotropin- releasing hormone, such as leuprolide
- tranquilizers such as phenaglycodol, meprobamate, heteronium plus amobarbital, propantheline plus phenobarbital, chlordiazepoxide, diazepam, med
- the invention provides methods for treating IBS in conjunction with other medications, such as agents that blunt visceral hyperalgesia in IBS, for example, kappa-opioid compounds, ⁇ 2 -adrenoceptor agonists (e.g., yohimbine, lidamidine), neurokinin-1 (NKi) receptor antagonists, somatostatin analogs (e.g., octreotide), or oxytocin.
- methods of the invention can be practiced in conjunction with psychological therapy, cognitive therapy, biofeedback and stress reduction techniques, and hypnosis.
- compounds and compositions of the invention can be used in conjunction with itopride, saredutant, renzapride, lubiprostone, or dynogen.
- the invention further provides methods for using calcimimetic compounds and compositions of the invention for treatment of colitis, e.g., collagenous and lymphocytic colitis, disorders characterized by chronic watery diarrhea with normal endoscopic and radiographic evaluations but histological evidence of chronic mucosal inflammation.
- colitis e.g., collagenous and lymphocytic colitis
- the compounds and compositions of the invention can be used for treatment of diversion colitis, endometriosis, typhlitis, colitis, cystica profunda, pneumatosis cystoides intestinalis, and malakoplakia.
- Clinical and pathological characteristics of these and other inflammatory disorders of the colon that can be treated using the methods of the invention are summarized in Table 5.
- Example 1 is offered to more fully illustrate the invention, but are not to be construed as limiting the scope thereof.
- Example 1
- mice Female Spraque-Dawley rats (approximately 150- 300 grams) were purchased from Charles River Laboratories Inc. (Wilmington, MA). Mdrla-/- mice (FVB.129P2-Abcbla tmlBor N7), weight 20-21 g, 8 weeks at initiation of experiments, were housed and maintained (5 per cage) in the animal facility at Amgen Washington. All animals were cared for according to the standard protocols of the Amgen Institutional Animal Care and Use Committee.
- DSS Dextran sodium sulfate
- ICN Chemicals Aurora, Ohio
- the DSS solution was prepared as a 5% w/v solution in sterile water (Baxter Healthcare Corporation) and sterile filtered through a 0.45 ⁇ m filter before being added to drinking water. Water for the negative control groups was similarly filtered.
- FK506, (also known as Tacrolimus, active ingredient in Prograf®), chemical name [3S-[3R*[E(1S*, 3S*, 4S*)], 4S*, 5R*, 8S*, 9E, 12R*, 14R*, 15S*, 16R*, 18S*, 19S*, 26aR*]]-5, 6, 8, 11, 12, 13, 14, 15, 16, 17, 18, 19, 24, 25, 26, 26a-hexadecahydro-5, 19- dihydroxy-3 - [2-(4-hydroxy-3 -methoxycyclohexyl)- 1 -methyl ethenyl] - 14, 16-dimethoxy-4, 10, 12, 18-tetramethyl-8-(2-propenyl)-15, 19-epoxy-3H-pyrodi [2,1-c] [1, 4] oxaazacyclotricosine- 1, 7, 20, 21 (4H, 23H)-tetrone monohydrate, was used as a positive control for the inflammation treatment.
- Tacrolimus is a macrolide immunosuppressant produced by Streptomyces tsukubaensis.
- the solution of FK506 (Prograf®, Astellas) was prepared as follows: 0.1 mg/ml solution in phosphate buffered saline (PBS) was prepared by diluting 5 mg/ml stock solution. The animals were dosed with FK506 at 1 mg/kg in 200 ⁇ l of PBS.
- the HEPES-Ringer solution contained (in mmol/L): NaCl 125; KCl 5; MgCl 2 0.5; HEPES 22, CaCl 2 0.1 or 1.6; glucose 10, pH 7.4. The solution was bubbled with 100%O 2 .
- Tetrodotoxin TTX
- Bumetanide Bumet was obtained from Sigma Chemical (St Louis, MO, USA) and stock solutions were prepared in dimethyl sulphoxide (DMSO).
- DMSO dimethyl sulphoxide
- Formulation vehicle was 20% (w/v) Captisol ( ⁇ - Cyclodextrin Sulfobutyl Ether Sodium Salt, Research Grade from CyDex, Inc.) in water with a final pH of 2.0-2.2 at room temperature, adjusted with HCl and NaOH as required. Final concentrations of DMSO never exceeded 0.1% (v/v). Preliminary experiments indicated that the vehicle did not alter any baseline electrophysiological parameters.
- DSS colitis in rats All experiments were performed on groups of 6 male Sprague- Dawley rats, housed 2 per cage. For disease induction, rats were given 3% or 5% DSS ad libitum as drinking water for 8 days according to Okayasu, et al. (1990) Gastroenterology 98: 694-702.
- the DSS solution was prepared as a 3% w/v solution in sterile water for Irrigation, USP (Baxter Healthcare Corporation) and then sterile filtered through a 0.45 mm filter before being added to the sterile drinking bottle for the cage.
- the control water was similarly filtered and handled. Animals were administered either filtered drinking water or water containing 5% DSS for 8 days and the water in the drinking bottle was replaced from the stock solution every 3-4 days.
- mice DSS colitis in mice. All experiments were performed on groups of 8 to 10 SJL-J mice, housed in the same cage. For disease induction, mice were given a 5% DSS ad libitum as drinking water for 9 days.
- mice and rats were euthanized by CO 2 asphyxiation.
- the large intestine were removed from the carcass, evaluated for gross lesions and three individual non-adjacent segments of proximal, middle and distal large intestine 2 centimeters long were immersed in the Zinc Formalin fix for 24 hours.
- the sections were washed with tap water and put into 70% ethanol.
- Each section was then cut into three 3 mm pieces (9 pieces per animal), processed into paraffin blocks, and sections were cut in 5 ⁇ m sections and stained with hematoxylin and eosin.
- animals were anaesthetized by isofluorane inhalation and killed by cervical dislocation. Segments of distal colon between the 1 st and 2 nd distal lymph nodes were removed quickly, cut along the mesenteric border into a flat sheet and flushed with ice-cold basal HEPES-Ringer solution containing 0.1 mM Ca and Mg. Up to four of these unstripped colonic sheets were obtained from each animal. Histomorphologic Examination
- Histopathologic changes of the proximal, middle, and distal small and large intestine was scored as a global assessment of inflammation based on inflammatory infiltrate, edema, mucosal erosions or ulcerations as described in Table 6.
- Nutrient diets formulations containing an amino acid or polyamine agonist of the CaSR Spargue-Dawley rats were fed either a control "1% Ca 2+ diet" (Altromin, Germany; containing 0.95% (w/w) calcium, 0.2% (w/w) tryptophan; 0% (w/w) spermine) or a "high spermine diet" (Altromin, Germany; containing 0.95% (w/w) calcium and 0.1% (w/w) spermine) or a "high tryptophan diet” (Altromin, Germany; containing 0.95% (w/w) calcium, 1.0% (w/w) tryptophan; 0% (w/w) spermine) for 2 weeks before and also during the periods of DSS treatment.
- a control "1% Ca 2+ diet” Altromin, Germany; containing 0.95% (w/w) calcium, 0.2% (w/w) tryptophan; 0% (w/w) spermine) or a
- Unstripped sheets from either proximal or distal colon were mounted between two halves of a modified Ussing chamber and short-circuited by a voltage clamp (VCC MC6; Physiologic Instruments) with correction for solution resistance.
- the exposure area was 0.3 cm 2
- the mucosal and serosal surfaces of the tissue were bathed in reservoirs with 3-5 mL HEPES-Ringer solution, pH 7.4 (37°C), maintained at 37°C and continuously bubbled with 100% O 2 .
- Tissues were allowed a minimum of 40-minute stabilization and basal recording period before drugs were added directly to the apical or basolateral side of the epithelium. Responses were recorded continuously and data were acquired via DATAQTM instruments and were stored in a PC and processed using the program AcqualizeTM.
- the pathology scores (0,1,2,3,4 and 5) are treated as ordinal data in the analysis.
- Analyses of the comparisons of the calcimimetic (Compound A) vs Vehicle, calcimimetic (Compound A) vs FK 506, and FK 506 vs Vehicle were conducted separately in the following two fashions.
- the scores in the groups were compared at each location and across all location, treating the scores from three locations as repeated measures.
- the analysis for each location was conducted using Mantel-Haenszel Chi-square Exact test.
- the analysis of repeated measures was done using Generalize Estimating Equation (GEE). (Models for discrete longitudinal data; Geert Molenberghs and Geert Verbeke, Springer 9 2005, Chapter 18.5).
- GEE Generalize Estimating Equation
- Colitis was induced in female mdrla 7" mice by infection with Helicobacter bilis via oral gavage.
- Mdr 1 a ⁇ ' ⁇ mice (FVB.129?2-Abcbla' m ' Bor N7) were obtained from Taconic (Hudson, NY). These mice spontaneously develop colitis, but inoculation with H. bilis can induce a more robust colitis in a shorter timeframe with a higher frequency of disease.
- the protocol used in this study was modeled after the protocol described by Maggio-Price et al. in 2002 (Am J Pathol. 160(2): 739-751).
- H. bilis culture and infection A frozen stock vial of H. bilis was thawed in a 1 : 1 mixture of brain heart infusion:Z?/-Mce/7 ⁇ broth and streaked onto blood agar plates. Plates were incubated for 2 days at 37 0 C in a microaerobic chamber containing an atmosphere of 90% N 2 , 5% H 2 , and 5% CO 2 . At the end of the 2 days of static growth, bacteria were harvested from the blood agar plate and placed into Erlenmeyer flasks containing Brucella broth supplemented with 5% FBS. This liquid culture was maintained with constant shaking for 24 hours at 37°C in a microaerobic chamber.
- Bacteria were concentrated by centrifugation and resuspended in Brucella broth. Bacteria were examined by gram stain for morphology, and other tests were run to confirm the presence of peroxidase, oxidase and urease activity. Bacterial concentration was determined by measuring the optical density at 600 nm (1 OD 60 O ⁇ 10 8 CFU/mL). Bacteria were diluted to a concentration of 10 8 /mL, and 100 ⁇ L ( 10 7 bacteria) was orally gavaged to 40 mdrla "7" mice. As a control, 5 mdrla 7" mice were dosed with 100 ⁇ L of Brucella broth, but were not further treated with any experimental compounds. H. bilis infections were done at the beginning of the study (day 0) and repeated one week later (day 7). Treatment
- mice were treated PO daily with the calcimimetic compound, R- Compound B, at doses of 10 mg/kg or 3 mg/kg.
- R- Compound B the calcimimetic compound
- 100 ⁇ L of vehicle or 10 mg/kg of the less active enantiomer of Compound B, S-Compound B were used. All treatments were given in 100 ⁇ L of vehicle beginning on day 1.
- mice were treated IP once per week with 250 ⁇ g of CTLA4-Fc in 100 ⁇ L of PBS beginning on day 1. All experimental compounds were generated at Amgen.
- mice were monitored at least 3 times per week for clinical signs of colitis.
- Clinical scoring criteria consisted of two parts: fecal consistency and anal inflammation.
- mice were necropsied on day 38 post initial inoculation. Mice were euthanized by CO 2 asphyxiation. Blood was obtained by intracardiac puncture and collected in serum separator tubes. Serum was harvested and stored at -80 0 C. Colons were dissected below the cecum and just above the anus, and the contents of the lumen were removed. Sections of the proximal, middle, and distal colon regions were fixed in 10% neutral buffered formalin for histopathological analysis.
- Serum clinical chemistry analysis was performed on the Olympus AU400 using standard tests and protocols. Calcium and phosphorus measurements were performed on undiluted serum for all samples. Serum cytokines and chemokines were assayed by Lincoplex mouse cytokine/chemokine muliplex immunoassasy (Millipore; Billerica, MA) per manufacturer instructions.
- Histology Portions from the proximal, middle, and distal colon were placed into 10% neutral buffered formalin, processed, and paraffin embedded using routine histology practices. Hematoxylin and eosin stained slides were prepared using routine histology protocols. One 5 ⁇ m section per colon sample was evaluated and scored using the criteria of Maggio-Price et al.
- 0 no inflammation
- 1 mild inflammation limited to mucosa
- 2 moderate inflammation in mucosa and submucosa
- 3 severe inflammation with obliteration of normal architecture, superficial erosions and/or crypt abscesses
- 4 severe inflammation with obliteration of normal architecture, superficial erosions and/or crypt abscesses plus mucosal ulceration.
- the histology score is the total of individual section scores.
- Results for serum analytes were expressed as the mean ⁇ standard error of the mean (SEM).
- a one-way ANOVA with Tukey's post test was performed using GraphPad Prism.
- a p- value of 0.05 was used in the calculation to determine whether there were significant differences between any two groups.
- Results for disease onset were expressed as a standard survival curve.
- a Log-Rank (Mantel-Cox) Test was performed using GraphPad Prism to determine significance.
- a p-value of 0.05 was used in the calculation to determine whether there were significant differences between any two groups.
- Results for clinical score and histology score were expressed as the mean ⁇ SEM.
- a Cochran-Mantel-Haenszel mean score test was performed using SAS software.
- a p-value of 0.05 was used in the calculation to determine whether there were significant differences between any two groups.
- This experiment demonstrates the effect of calcimimetics on inflammation in the DSS- induced mice model of IBD.
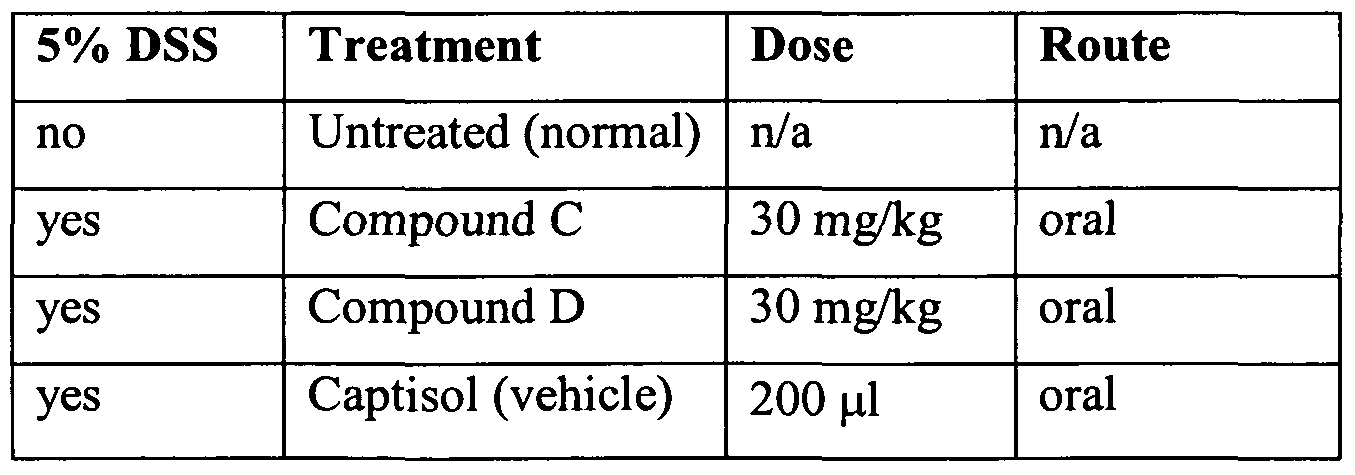
- mice were given 5% DSS in water ad libitum as drinking water for 9 days as described in Example 1 and were concurrently treated orally during that period with one of the treatments listed in Table 8.
- Figure 1 and Table 8 summarize the effect of the calcimimetic compound B on colonic inflammation in 5% DSS induced colitis.
- Animals were euthanized with carbon dioxide on day nine post treatment.
- the large intestine (colon) was removed and processed for histopathologic changes of the proximal, middle and distal segments as described in Example 1.
- the intestine was scored as a global assessment of inflammation based on inflammatory infiltrate, edema, mucosal erosions or ulcerations as indicated in Table 6. All samples were scored in a blinded fashion.
- Example 3 This example demonstrates that calcimimetics inhibit enteric nervous system activity.
- Figure 3 summarizes the changes in short-circuit secretory current in the absence and presence of 2 ⁇ M TTX, before and after addition of 10 ⁇ M of a calcimimetic Compound A, before and after the addition of 100 ⁇ M bumetanide, an inhibitor of chloride secretion. Representative traces of the temporal changes in short-circuit current are shown for Proximal (Panel A) and Distal (Panel C) colon before and after addition of the agents indicated by arrows. TTX inhibited the secretory short-circuit current over 15 minutes. Subsequent addition of Compound A or bumetanide (Bumet) had no additional effect on short-circuit current.
- Figure 4 summarizes the changes in short-circuit secretory current in the absence and presence of 10 ⁇ M of Compound A, a calcimimetic, before and after addition of 2 ⁇ M TTX, an inhibitor of ENS activity, before and after the addition of 100 ⁇ M bumetanide, an inhibitor of chloride secretion. Representative traces of the temporal changes in short-circuit current are shown for Proximal (A) and Distal (C) colon before and after addition of the agents indicated by arrows. Compound A inhibited the secretory short-circuit current over 15 minutes. Subsequent addition of TTX or bumetanide (Bumet) had no additional effect on short-circuit current. The rates of change in short-circuit current ( ⁇ A/cm 2 min) induced by addition of these agents are summarized for Proximal (B) and Distal (D) colon sheets. The number in parentheses indicates the number of observations.
- Example 1 Scores from each section of the colon were summed to generate a total large intestine (LI) score. The maximum LI score for each mouse was 12. Mouse 3-5 was found dead in its cage, so the tissue was not present (tnp). 5 In the vehicle treated group, 5 out of 9 animals (55%) had colitis, while 2 out of 10 animals (20%) in each of the R-Compound B treated groups had colon pathology ( Figure 7). The total large intestine (LI) scores of the colitic mice in the vehicle group were all > 9, while scores of the R-Compound B treated animals were 8, 8, 7, and 2. Taken together, these data indicate that treatment with R-Compound B reduced the incidence and severity of disease. In
- Serum IP-10 levels were elevated in vehicle treated animals that demonstrated clinical symptoms of disease, and they correlated with the severity of the disease as measured by clinical score. This correlation with severity was present in every treatment group. In the R- Compound B and S-Compound B groups, less elevation of IP-10 was observed, further suggesting a protective benefit.
Landscapes
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA2681582A CA2681582C (en) | 2007-03-30 | 2008-03-31 | Methods of treating bowel disorders |
| EP08742406.5A EP2139462B1 (en) | 2007-03-30 | 2008-03-31 | Calcimimetic compounds for use in the treatment of bowel disorders |
| AU2008233088A AU2008233088B2 (en) | 2007-03-30 | 2008-03-31 | Calcimimetic compounds for use in the treatment of bowel disorders |
| ES08742406.5T ES2449340T3 (en) | 2007-03-30 | 2008-03-31 | Calcimimetic compounds for use in the treatment of intestinal disorders |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US92113207P | 2007-03-30 | 2007-03-30 | |
| US60/921,132 | 2007-03-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008121386A2 true WO2008121386A2 (en) | 2008-10-09 |
| WO2008121386A3 WO2008121386A3 (en) | 2008-12-31 |
Family
ID=39628941
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/004166 WO2008121386A2 (en) | 2007-03-30 | 2008-03-31 | Calcimimetic compounds for use in the treatment of bowel disorders |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8093299B2 (en) |
| EP (1) | EP2139462B1 (en) |
| AU (1) | AU2008233088B2 (en) |
| CA (1) | CA2681582C (en) |
| ES (1) | ES2449340T3 (en) |
| WO (1) | WO2008121386A2 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009121015A2 (en) * | 2008-03-28 | 2009-10-01 | Amgen Inc. | Methods of treating epithelial injury |
| WO2010104882A1 (en) * | 2009-03-10 | 2010-09-16 | Amgen Inc. | Methods of modulating sperm motility |
| JP2011517189A (en) * | 2008-03-28 | 2011-05-26 | クゥアルコム・インコーポレイテッド | Sending signaling messages using beacon signals |
| WO2011056325A3 (en) * | 2009-09-29 | 2011-09-29 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compounds |
| US8722732B2 (en) | 2009-09-29 | 2014-05-13 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compounds |
| US8995559B2 (en) | 2008-03-28 | 2015-03-31 | Qualcomm Incorporated | Signaling message transmission in a wireless communication network |
| CN112220783A (en) * | 2020-10-26 | 2021-01-15 | 四川大学华西医院 | Medical application of cinacalcet in treating inflammatory bowel disease through NK1R target |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9770047B2 (en) * | 2008-04-10 | 2017-09-26 | U.S. Nutraceuticals, LLC | Perilla seed composition |
| WO2017003724A1 (en) | 2015-07-01 | 2017-01-05 | Crinetics Pharmaceuticals, Inc. | Somatostatin modulators and uses thereof |
| EP3658560A4 (en) | 2017-07-25 | 2021-01-06 | Crinetics Pharmaceuticals, Inc. | Somatostatin modulators and uses thereof |
Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR511940A (en) | 1916-12-08 | 1921-01-07 | Rene Emile Trottier | Device for controlling apparatus intended for classifying or separating solids of all kinds |
| WO1993004373A1 (en) | 1991-08-23 | 1993-03-04 | Nps Pharmaceuticals, Inc. | Calcium receptor active molecules |
| WO1994018959A1 (en) | 1993-02-23 | 1994-09-01 | Brigham And Women's Hospital, Inc. | Calcium receptor-active molecules |
| WO1995011221A1 (en) | 1991-08-23 | 1995-04-27 | Nps Pharmaceuticals, Inc. | Calcium receptor-active arylalkyl amines |
| WO1996012697A2 (en) | 1994-10-21 | 1996-05-02 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| WO1997041090A1 (en) | 1996-05-01 | 1997-11-06 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| US5688938A (en) | 1991-08-23 | 1997-11-18 | The Brigham & Women's Hospital, Inc. | Calcium receptor-active molecules |
| US5763569A (en) | 1991-08-23 | 1998-06-09 | The Brigham And Women's Hospital, Inc | Calcium receptor-active molecules |
| EP0933354A1 (en) | 1996-07-08 | 1999-08-04 | Kirin Beer Kabushiki Kaisha | Calcium receptor-active compounds |
| US5962314A (en) | 1993-02-23 | 1999-10-05 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6001884A (en) | 1991-08-23 | 1999-12-14 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6011068A (en) | 1991-08-23 | 2000-01-04 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6031003A (en) | 1991-08-23 | 2000-02-29 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6172091B1 (en) | 1998-10-14 | 2001-01-09 | Ortho-Mcneil Pharmaceutical, Inc. | 1,2-Disubstituted cyclopropanes |
| WO2001034562A1 (en) | 1999-11-09 | 2001-05-17 | Centre National De La Recherche Scientifique (Cnrs) | Aralkyl-1,2-diamines having calcimimetic activity and preparation mode |
| US6313146B1 (en) | 1991-08-23 | 2001-11-06 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6319518B1 (en) | 1997-07-03 | 2001-11-20 | Samyang Corporation | Colon selective drug delivery composition |
| WO2001090069A1 (en) | 2000-05-24 | 2001-11-29 | Centre National De La Recherche Scientifique (Cnrs) | Novel calcium receptor active molecules and method for preparing same |
| WO2002014259A1 (en) | 2000-08-11 | 2002-02-21 | Japan Tobacco Inc. | Calcium receptor antagonists |
| US6413494B1 (en) | 1998-07-23 | 2002-07-02 | Samyang Corporation | Composition and pharmaceutical dosage form for colonic drug delivery using polysaccharides |
| WO2002059102A2 (en) | 2001-01-26 | 2002-08-01 | Aventis Pharma S.A. | Urea derivatives, method for preparing same, use thereof as medicines, pharmaceutical compositions and use |
| US6432656B1 (en) | 1996-12-03 | 2002-08-13 | Nps Pharmaceuticals, Inc. | Calcilytic compounds |
| EP1258471A2 (en) | 1996-05-01 | 2002-11-20 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| WO2003099814A1 (en) | 2002-05-23 | 2003-12-04 | Amgen, Inc. | Calcium receptor modulating agents |
| WO2003099776A1 (en) | 2002-05-23 | 2003-12-04 | Amgen Inc. | Calcium receptor modulating arylalkylamines |
| WO2004017908A2 (en) | 2002-08-26 | 2004-03-04 | Takeda Pharmaceutical Company Limited | Calcium receptor modulating compound and use thereof |
| WO2004094362A1 (en) | 2003-04-23 | 2004-11-04 | Japan Tobacco Inc. | CaSR ANTAGONIST |
| WO2004106280A1 (en) | 2003-05-28 | 2004-12-09 | Japan Tobacco Inc. | CaSR ANTAGONIST |
| WO2006117211A2 (en) | 2005-04-29 | 2006-11-09 | Proskelia Sas | Urea derivatives methods for their manufacture and uses thereof |
| WO2006123725A1 (en) | 2005-05-19 | 2006-11-23 | Astellas Pharma Inc. | Pyrrolidine derivative or salt thereof |
| US7157498B2 (en) | 2000-08-08 | 2007-01-02 | Centre National De La Recherche Scientifique (Cnrs) | Diamines having a CaSR modulating activity |
| WO2007060026A1 (en) | 2005-11-25 | 2007-05-31 | Galapagos Sas | Urea derivatives useful as calcium receptor modulators |
| WO2008006625A2 (en) | 2006-07-10 | 2008-01-17 | Galapagos Sas | Derivatives of urea and related diamines, methods for their manufacture, and uses therefor |
| WO2008019690A1 (en) | 2006-08-18 | 2008-02-21 | Leo Pharma A/S | Substituted acetylenic compounds useful for the treatment of diseases |
Family Cites Families (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6861053B1 (en) | 1999-08-11 | 2005-03-01 | Cedars-Sinai Medical Center | Methods of diagnosing or treating irritable bowel syndrome and other disorders caused by small intestinal bacterial overgrowth |
| PE20000415A1 (en) | 1998-04-08 | 2000-05-21 | Smithkline Beecham Corp | CALCILITIC COMPOUNDS |
| US6395273B1 (en) | 1998-06-10 | 2002-05-28 | Promega Corporation | Prevention and treatment of inflammatory bowel disease |
| US6214373B1 (en) | 1999-10-07 | 2001-04-10 | Snowden-Sutton Associates, Inc. | Nutritional composition for treating inflammatory bowel diseases |
| US6358939B1 (en) | 1999-12-21 | 2002-03-19 | Northern Lights Pharmaceuticals, Llc | Use of biologically active vitamin D compounds for the prevention and treatment of inflammatory bowel disease |
| US6613751B2 (en) | 2000-02-23 | 2003-09-02 | The Regents Of The University Of California | Method for treating inflammatory bowel disease and other forms of gastrointestinal inflammation |
| PL362513A1 (en) * | 2000-12-19 | 2004-11-02 | Merck Patent Gmbh | Pharmaceutical formulation containing pyrazolo[4,3-d]pyrimidines and antithrombotic agents, calcium-antagonists, prostaglandins or prostaglandin derivatives |
| US20030092765A1 (en) * | 2001-11-15 | 2003-05-15 | John Kelly | Treatment of abnormal increases in gastrointestinal motility with (R)-verapamil |
| US20040219232A1 (en) | 2003-02-06 | 2004-11-04 | Zengen, Inc. | Methods and compounds for treating malabsorption diseases and inflammatory conditions of the gastrointestinal tract |
| US7205322B2 (en) | 2003-02-12 | 2007-04-17 | Bristol-Myers Squibb Company | Thiazolidine compounds as calcium sensing receptor modulators |
| US20050004155A1 (en) | 2003-04-08 | 2005-01-06 | Boyd Thomas A. | Use of methylnaltrexone to treat irritable bowel syndrome |
| EP1615667A2 (en) | 2003-04-11 | 2006-01-18 | Novo Nordisk A/S | Combinations of an 11-beta-hydroxysteroid dehydrogenase type 1 inhibitor and a glucocorticoid receptor agonist |
| US7265145B2 (en) | 2003-05-28 | 2007-09-04 | Bristol-Myers Squibb Company | Substituted piperidines and pyrrolidines as calcium sensing receptor modulators and method |
| JP5244597B2 (en) | 2005-09-02 | 2013-07-24 | アムジエン・インコーポレーテツド | Method for regulating intestinal fluid balance using calcilytic or calcimimetic compounds |
| JP5309014B2 (en) | 2006-03-23 | 2013-10-09 | アムジエン・インコーポレーテツド | Methods and compositions for the production and use of cinacalcet polymorphs |
| MX2008013339A (en) * | 2006-04-20 | 2009-01-26 | Amgen Inc | Stable emulsion formulations. |
| MX2009003981A (en) | 2006-10-26 | 2009-04-27 | Amgen Inc | Calcium receptor modulating agents. |
-
2008
- 2008-03-31 US US12/080,086 patent/US8093299B2/en active Active
- 2008-03-31 WO PCT/US2008/004166 patent/WO2008121386A2/en active Application Filing
- 2008-03-31 EP EP08742406.5A patent/EP2139462B1/en active Active
- 2008-03-31 AU AU2008233088A patent/AU2008233088B2/en active Active
- 2008-03-31 CA CA2681582A patent/CA2681582C/en active Active
- 2008-03-31 ES ES08742406.5T patent/ES2449340T3/en active Active
Patent Citations (67)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR511940A (en) | 1916-12-08 | 1921-01-07 | Rene Emile Trottier | Device for controlling apparatus intended for classifying or separating solids of all kinds |
| EP1281702A2 (en) | 1991-08-23 | 2003-02-05 | Nps Pharmaceuticals, Inc. | Calcium receptor active molecules |
| EP1296142A2 (en) | 1991-08-23 | 2003-03-26 | Nps Pharmaceuticals, Inc. | Calcium receptor active molecules |
| US6011068A (en) | 1991-08-23 | 2000-01-04 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| WO1995011221A1 (en) | 1991-08-23 | 1995-04-27 | Nps Pharmaceuticals, Inc. | Calcium receptor-active arylalkyl amines |
| EP0657029A1 (en) | 1991-08-23 | 1995-06-14 | Nps Pharmaceuticals, Inc. | Calcium receptor active molecules |
| WO1993004373A1 (en) | 1991-08-23 | 1993-03-04 | Nps Pharmaceuticals, Inc. | Calcium receptor active molecules |
| US6031003A (en) | 1991-08-23 | 2000-02-29 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| US6001884A (en) | 1991-08-23 | 1999-12-14 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| EP0724561A1 (en) | 1991-08-23 | 1996-08-07 | Nps Pharmaceuticals, Inc. | Calcium receptor-active arylalkyl amines |
| US5688938A (en) | 1991-08-23 | 1997-11-18 | The Brigham & Women's Hospital, Inc. | Calcium receptor-active molecules |
| US5763569A (en) | 1991-08-23 | 1998-06-09 | The Brigham And Women's Hospital, Inc | Calcium receptor-active molecules |
| US6313146B1 (en) | 1991-08-23 | 2001-11-06 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| WO1994018959A1 (en) | 1993-02-23 | 1994-09-01 | Brigham And Women's Hospital, Inc. | Calcium receptor-active molecules |
| US5962314A (en) | 1993-02-23 | 1999-10-05 | Nps Pharmaceuticals, Inc. | Calcium receptor-active molecules |
| EP0637237A1 (en) | 1993-02-23 | 1995-02-08 | Brigham And Women's Hospital, Inc. | Calcium receptor-active molecules |
| EP0787122A2 (en) | 1994-10-21 | 1997-08-06 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| EP1203761A2 (en) | 1994-10-21 | 2002-05-08 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| EP1275635A1 (en) | 1994-10-21 | 2003-01-15 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| EP1553078A1 (en) | 1994-10-21 | 2005-07-13 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| US6211244B1 (en) | 1994-10-21 | 2001-04-03 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| WO1996012697A2 (en) | 1994-10-21 | 1996-05-02 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| WO1997041090A1 (en) | 1996-05-01 | 1997-11-06 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| US6710088B2 (en) | 1996-05-01 | 2004-03-23 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| US6342532B1 (en) | 1996-05-01 | 2002-01-29 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor active compounds |
| EP0907631A1 (en) | 1996-05-01 | 1999-04-14 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| US5981599A (en) | 1996-05-01 | 1999-11-09 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor active compounds |
| US20030008876A1 (en) | 1996-05-01 | 2003-01-09 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| EP1258471A2 (en) | 1996-05-01 | 2002-11-20 | Nps Pharmaceuticals, Inc. | Inorganic ion receptor-active compounds |
| US20030176485A1 (en) | 1996-07-08 | 2003-09-18 | Nps Pharmaceuticals, Inc. | Calcium receptor active compounds |
| US20030144526A1 (en) | 1996-07-08 | 2003-07-31 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| US6362231B1 (en) | 1996-07-08 | 2002-03-26 | Nps Pharmaceuticals, Inc. | Calcium receptor active compounds |
| US20020107406A1 (en) | 1996-07-08 | 2002-08-08 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| US6750255B2 (en) | 1996-07-08 | 2004-06-15 | Nps Pharmaceuticals, Inc. | Calcium receptor-active compounds |
| EP0933354A1 (en) | 1996-07-08 | 1999-08-04 | Kirin Beer Kabushiki Kaisha | Calcium receptor-active compounds |
| US6432656B1 (en) | 1996-12-03 | 2002-08-13 | Nps Pharmaceuticals, Inc. | Calcilytic compounds |
| US6319518B1 (en) | 1997-07-03 | 2001-11-20 | Samyang Corporation | Colon selective drug delivery composition |
| US6413494B1 (en) | 1998-07-23 | 2002-07-02 | Samyang Corporation | Composition and pharmaceutical dosage form for colonic drug delivery using polysaccharides |
| US6172091B1 (en) | 1998-10-14 | 2001-01-09 | Ortho-Mcneil Pharmaceutical, Inc. | 1,2-Disubstituted cyclopropanes |
| EP1235797A1 (en) | 1999-11-09 | 2002-09-04 | Centre National De La Recherche Scientifique (Cnrs) | Aralkyl-1,2-diamines having calcimimetic activity and preparation mode |
| WO2001034562A1 (en) | 1999-11-09 | 2001-05-17 | Centre National De La Recherche Scientifique (Cnrs) | Aralkyl-1,2-diamines having calcimimetic activity and preparation mode |
| US20030199497A1 (en) | 2000-05-24 | 2003-10-23 | Martial Ruat | Novel calcium receptor active molecules and method for preparing same |
| EP1284963A1 (en) | 2000-05-24 | 2003-02-26 | Centre National De La Recherche Scientifique (Cnrs) | Novel calcium receptor active molecules and method for preparing same |
| WO2001090069A1 (en) | 2000-05-24 | 2001-11-29 | Centre National De La Recherche Scientifique (Cnrs) | Novel calcium receptor active molecules and method for preparing same |
| US7157498B2 (en) | 2000-08-08 | 2007-01-02 | Centre National De La Recherche Scientifique (Cnrs) | Diamines having a CaSR modulating activity |
| WO2002014259A1 (en) | 2000-08-11 | 2002-02-21 | Japan Tobacco Inc. | Calcium receptor antagonists |
| US20040006130A1 (en) | 2000-08-11 | 2004-01-08 | Yuko Shinagawa | Calcium receptor antagonist |
| US20050107448A1 (en) | 2000-08-11 | 2005-05-19 | Japan Tabacco Inc. | Calcium receptor antagonists |
| EP1308436A1 (en) | 2000-08-11 | 2003-05-07 | Japan Tobacco Inc. | Calcium receptor antagonists |
| WO2002059102A2 (en) | 2001-01-26 | 2002-08-01 | Aventis Pharma S.A. | Urea derivatives, method for preparing same, use thereof as medicines, pharmaceutical compositions and use |
| EP1509497A1 (en) | 2002-05-23 | 2005-03-02 | Amgen Inc. | Calcium receptor modulating arylalkylamines |
| WO2003099776A1 (en) | 2002-05-23 | 2003-12-04 | Amgen Inc. | Calcium receptor modulating arylalkylamines |
| US7176322B2 (en) | 2002-05-23 | 2007-02-13 | Amgen Inc. | Calcium receptor modulating agents |
| WO2003099814A1 (en) | 2002-05-23 | 2003-12-04 | Amgen, Inc. | Calcium receptor modulating agents |
| US20040077619A1 (en) | 2002-05-23 | 2004-04-22 | Kelly Michael G. | Calcium receptor modulating agents |
| EP1509518A1 (en) | 2002-05-23 | 2005-03-02 | Amgen, Inc. | Calcium receptor modulating agents |
| US20050143426A1 (en) | 2002-05-23 | 2005-06-30 | Amgen Inc. | Calcium receptor modulating agents |
| US6908935B2 (en) | 2002-05-23 | 2005-06-21 | Amgen Inc. | Calcium receptor modulating agents |
| WO2004017908A2 (en) | 2002-08-26 | 2004-03-04 | Takeda Pharmaceutical Company Limited | Calcium receptor modulating compound and use thereof |
| WO2004094362A1 (en) | 2003-04-23 | 2004-11-04 | Japan Tobacco Inc. | CaSR ANTAGONIST |
| US20050032796A1 (en) | 2003-04-23 | 2005-02-10 | Japan Tobacco Inc. | CaSR antagonist |
| WO2004106280A1 (en) | 2003-05-28 | 2004-12-09 | Japan Tobacco Inc. | CaSR ANTAGONIST |
| WO2006117211A2 (en) | 2005-04-29 | 2006-11-09 | Proskelia Sas | Urea derivatives methods for their manufacture and uses thereof |
| WO2006123725A1 (en) | 2005-05-19 | 2006-11-23 | Astellas Pharma Inc. | Pyrrolidine derivative or salt thereof |
| WO2007060026A1 (en) | 2005-11-25 | 2007-05-31 | Galapagos Sas | Urea derivatives useful as calcium receptor modulators |
| WO2008006625A2 (en) | 2006-07-10 | 2008-01-17 | Galapagos Sas | Derivatives of urea and related diamines, methods for their manufacture, and uses therefor |
| WO2008019690A1 (en) | 2006-08-18 | 2008-02-21 | Leo Pharma A/S | Substituted acetylenic compounds useful for the treatment of diseases |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2009228069B2 (en) * | 2008-03-28 | 2014-05-29 | Amgen Inc. | Calcimimetic compound for use in the treatment of epithelial injury |
| WO2009121015A3 (en) * | 2008-03-28 | 2009-11-26 | Amgen Inc. | Calcimimetic compound for use in the treatment of epithelial injury |
| JP2011517189A (en) * | 2008-03-28 | 2011-05-26 | クゥアルコム・インコーポレイテッド | Sending signaling messages using beacon signals |
| WO2009121015A2 (en) * | 2008-03-28 | 2009-10-01 | Amgen Inc. | Methods of treating epithelial injury |
| US9949276B2 (en) | 2008-03-28 | 2018-04-17 | Qualcomm Incorporated | Signaling message transmission in a wireless communication network |
| US9276787B2 (en) | 2008-03-28 | 2016-03-01 | Qualcomm Incorporated | Transmission of signaling messages using beacon signals |
| US8609655B2 (en) | 2008-03-28 | 2013-12-17 | Amgen Inc. | Calcimimetic compound for use in the treatment of epithelial injury |
| US8995559B2 (en) | 2008-03-28 | 2015-03-31 | Qualcomm Incorporated | Signaling message transmission in a wireless communication network |
| WO2010104882A1 (en) * | 2009-03-10 | 2010-09-16 | Amgen Inc. | Methods of modulating sperm motility |
| WO2011056325A3 (en) * | 2009-09-29 | 2011-09-29 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compounds |
| US8722732B2 (en) | 2009-09-29 | 2014-05-13 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compounds |
| AU2010315805B2 (en) * | 2009-09-29 | 2015-09-17 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compounds |
| JP2013505966A (en) * | 2009-09-29 | 2013-02-21 | ネクター セラピューティックス | Oligomer-calcimimetic conjugates and related compounds |
| US9421275B2 (en) | 2009-09-29 | 2016-08-23 | Nektar Therapeutics | Oligomer-calcimimetic conjugates and related compunds |
| KR101828613B1 (en) * | 2009-09-29 | 2018-02-12 | 넥타르 테라퓨틱스 | Oligomer-calcimimetic conjugates and related compounds |
| CN102573921A (en) * | 2009-09-29 | 2012-07-11 | 尼克塔治疗公司 | Oligomer-calcimimetic conjugates and related compounds |
| CN112220783A (en) * | 2020-10-26 | 2021-01-15 | 四川大学华西医院 | Medical application of cinacalcet in treating inflammatory bowel disease through NK1R target |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2681582C (en) | 2015-07-14 |
| CA2681582A1 (en) | 2008-10-09 |
| US20090018135A1 (en) | 2009-01-15 |
| AU2008233088B2 (en) | 2013-09-26 |
| WO2008121386A3 (en) | 2008-12-31 |
| EP2139462A2 (en) | 2010-01-06 |
| US8093299B2 (en) | 2012-01-10 |
| AU2008233088A1 (en) | 2008-10-09 |
| EP2139462B1 (en) | 2014-01-22 |
| ES2449340T3 (en) | 2014-03-19 |
| AU2008233088A8 (en) | 2009-10-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8093299B2 (en) | Methods of treating bowel disorders | |
| JP6014702B2 (en) | Milnacipran for long-term treatment of fibromyalgia syndrome | |
| JP5289765B2 (en) | Use of memantine (namenda) for the treatment of autism, obsessive-compulsive disorder, and impulsivity | |
| AU2021204465B2 (en) | Extended release pharmaceutical compositions of levetiracetam | |
| KR102654569B1 (en) | Agents for the treatment of post-traumatic stress disorder | |
| WO2005099714A1 (en) | Compositions and methods for treatment of neuropathic pain, fibromyalgia and chronic fatigue syndrome | |
| KR20120050512A (en) | Durable treatment with 4-aminopyridine in patients with demyelination | |
| CA2425812C (en) | Treatment of oppositional defiant disorder and conduct disorder with 5-amino-4,5,6,7-tetrahydro-4-oxyindolones | |
| US20230097618A1 (en) | Glycine receptor modulators and methods of use | |
| JP5109132B2 (en) | Drugs for attention deficit / hyperactivity disorder | |
| JP6084931B2 (en) | Neramexane for the treatment or prevention of tinnitus associated with stress or acute hearing loss | |
| JP2019519480A (en) | Use of Acetylcholinesterase Inhibitor and Idaropirdin to Reduce Fall in Parkinson's Disease Patients | |
| EP1778248A1 (en) | A composition containing a thiourea derivative for preventing or treating pruritic or irritant skin diseases | |
| TW202402287A (en) | Pharmaceutical compositions comprising a quinolone compound for irritable bowel syndrome | |
| CA2562078A1 (en) | Compositions and methods for treatment of neuropathic pain, fibromyalgia and chronic fatigue syndrome | |
| JP2016138144A (en) | Drug for treatment of tinnitus patients |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08742406 Country of ref document: EP Kind code of ref document: A2 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008233088 Country of ref document: AU |
|
| ENP | Entry into the national phase |
Ref document number: 2681582 Country of ref document: CA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2008233088 Country of ref document: AU Date of ref document: 20080331 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008742406 Country of ref document: EP |