WO2008075118A1 - Novel cb1 antagonists and their preparation - Google Patents
Novel cb1 antagonists and their preparation Download PDFInfo
- Publication number
- WO2008075118A1 WO2008075118A1 PCT/HU2007/000126 HU2007000126W WO2008075118A1 WO 2008075118 A1 WO2008075118 A1 WO 2008075118A1 HU 2007000126 W HU2007000126 W HU 2007000126W WO 2008075118 A1 WO2008075118 A1 WO 2008075118A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- pyrazole
- methyl
- compounds
- Prior art date
Links
- 238000002360 preparation method Methods 0.000 title abstract description 6
- 229940124802 CB1 antagonist Drugs 0.000 title abstract 2
- 150000001875 compounds Chemical class 0.000 claims abstract description 100
- 238000000034 method Methods 0.000 claims description 47
- 150000004677 hydrates Chemical class 0.000 claims description 16
- 150000003839 salts Chemical class 0.000 claims description 16
- 239000012453 solvate Substances 0.000 claims description 16
- 239000001257 hydrogen Substances 0.000 claims description 12
- 229910052739 hydrogen Inorganic materials 0.000 claims description 12
- 229910052736 halogen Inorganic materials 0.000 claims description 10
- 150000002367 halogens Chemical class 0.000 claims description 10
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 7
- 230000015572 biosynthetic process Effects 0.000 claims description 7
- 239000008194 pharmaceutical composition Substances 0.000 claims description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical class [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 6
- 229930003827 cannabinoid Natural products 0.000 claims description 6
- 239000003557 cannabinoid Substances 0.000 claims description 6
- 150000002431 hydrogen Chemical group 0.000 claims description 6
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 6
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 claims description 6
- 238000003786 synthesis reaction Methods 0.000 claims description 6
- 125000000217 alkyl group Chemical group 0.000 claims description 4
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 4
- 230000008569 process Effects 0.000 claims description 4
- 125000006527 (C1-C5) alkyl group Chemical group 0.000 claims description 3
- 125000004414 alkyl thio group Chemical group 0.000 claims description 3
- CAAIBUKGGKFUKU-UHFFFAOYSA-N ethyl 2-imidazol-1-yl-2-oxoacetate Chemical compound CCOC(=O)C(=O)N1C=CN=C1 CAAIBUKGGKFUKU-UHFFFAOYSA-N 0.000 claims description 3
- YNESATAKKCNGOF-UHFFFAOYSA-N lithium bis(trimethylsilyl)amide Chemical compound [Li+].C[Si](C)(C)[N-][Si](C)(C)C YNESATAKKCNGOF-UHFFFAOYSA-N 0.000 claims description 3
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 claims description 3
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 3
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 2
- 125000004185 ester group Chemical group 0.000 claims description 2
- 150000002148 esters Chemical class 0.000 claims description 2
- 230000003301 hydrolyzing effect Effects 0.000 claims description 2
- JOVOSQBPPZZESK-UHFFFAOYSA-N phenylhydrazine hydrochloride Chemical class Cl.NNC1=CC=CC=C1 JOVOSQBPPZZESK-UHFFFAOYSA-N 0.000 claims description 2
- 229940038531 phenylhydrazine hydrochloride Drugs 0.000 claims description 2
- 102000018208 Cannabinoid Receptor Human genes 0.000 claims 4
- 108050007331 Cannabinoid receptor Proteins 0.000 claims 4
- 102000005962 receptors Human genes 0.000 claims 2
- 108020003175 receptors Proteins 0.000 claims 2
- CUBIHDCARJLZRL-UHFFFAOYSA-N 5-(4-cyclohexylphenyl)-1-(2,4-dichlorophenyl)-4-methyl-n-piperidin-1-ylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC(Cl)=CC=2)Cl)C=1C(C=C1)=CC=C1C1CCCCC1 CUBIHDCARJLZRL-UHFFFAOYSA-N 0.000 claims 1
- AZWLMRQUHSNCQJ-UHFFFAOYSA-N 5-(4-cyclopropylphenyl)-1-(2,4-dichlorophenyl)-4-methyl-n-piperidin-1-ylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC(Cl)=CC=2)Cl)C=1C(C=C1)=CC=C1C1CC1 AZWLMRQUHSNCQJ-UHFFFAOYSA-N 0.000 claims 1
- 241000124008 Mammalia Species 0.000 claims 1
- 239000002671 adjuvant Substances 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 239000000463 material Substances 0.000 claims 1
- 208000008589 Obesity Diseases 0.000 abstract description 6
- 235000020824 obesity Nutrition 0.000 abstract description 6
- 208000020016 psychiatric disease Diseases 0.000 abstract description 4
- 206010012335 Dependence Diseases 0.000 abstract description 2
- 230000003542 behavioural effect Effects 0.000 abstract description 2
- 208000030159 metabolic disease Diseases 0.000 abstract description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 84
- 238000005160 1H NMR spectroscopy Methods 0.000 description 34
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 18
- 230000002503 metabolic effect Effects 0.000 description 17
- 239000000203 mixture Substances 0.000 description 17
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 12
- JZCPYUJPEARBJL-UHFFFAOYSA-N rimonabant Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC(Cl)=CC=2)Cl)C=1C1=CC=C(Cl)C=C1 JZCPYUJPEARBJL-UHFFFAOYSA-N 0.000 description 12
- 238000012360 testing method Methods 0.000 description 12
- 239000005557 antagonist Substances 0.000 description 11
- 239000000725 suspension Substances 0.000 description 11
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 10
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 10
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- 239000000556 agonist Substances 0.000 description 8
- 229960003015 rimonabant Drugs 0.000 description 8
- 239000000243 solution Substances 0.000 description 8
- 208000035475 disorder Diseases 0.000 description 7
- 238000000338 in vitro Methods 0.000 description 7
- 210000001853 liver microsome Anatomy 0.000 description 7
- 230000008406 drug-drug interaction Effects 0.000 description 6
- 238000001727 in vivo Methods 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 230000036760 body temperature Effects 0.000 description 5
- 210000004556 brain Anatomy 0.000 description 5
- 239000002775 capsule Substances 0.000 description 5
- 201000010099 disease Diseases 0.000 description 5
- SBMSLRMNBSMKQC-UHFFFAOYSA-N pyrrolidin-1-amine Chemical compound NN1CCCC1 SBMSLRMNBSMKQC-UHFFFAOYSA-N 0.000 description 5
- FJYWNYLUZBMVKI-UHFFFAOYSA-N 3,3a,4,5,6,6a-hexahydro-1h-cyclopenta[c]pyrrol-2-amine Chemical compound C1CCC2CN(N)CC21 FJYWNYLUZBMVKI-UHFFFAOYSA-N 0.000 description 4
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 4
- 241000699666 Mus <mouse, genus> Species 0.000 description 4
- VSCWAEJMTAWNJL-UHFFFAOYSA-K aluminium trichloride Chemical compound Cl[Al](Cl)Cl VSCWAEJMTAWNJL-UHFFFAOYSA-K 0.000 description 4
- ZPUCINDJVBIVPJ-LJISPDSOSA-N cocaine Chemical compound O([C@H]1C[C@@H]2CC[C@@H](N2C)[C@H]1C(=O)OC)C(=O)C1=CC=CC=C1 ZPUCINDJVBIVPJ-LJISPDSOSA-N 0.000 description 4
- MKQLBNJQQZRQJU-UHFFFAOYSA-N morpholin-4-amine Chemical compound NN1CCOCC1 MKQLBNJQQZRQJU-UHFFFAOYSA-N 0.000 description 4
- 239000003921 oil Substances 0.000 description 4
- 235000019198 oils Nutrition 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- SNICXCGAKADSCV-JTQLQIEISA-N (-)-Nicotine Chemical compound CN1CCC[C@H]1C1=CC=CN=C1 SNICXCGAKADSCV-JTQLQIEISA-N 0.000 description 3
- 208000019901 Anxiety disease Diseases 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 208000032928 Dyslipidaemia Diseases 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- 239000001828 Gelatine Substances 0.000 description 3
- 208000017170 Lipid metabolism disease Diseases 0.000 description 3
- 208000001145 Metabolic Syndrome Diseases 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 239000000443 aerosol Substances 0.000 description 3
- 230000036506 anxiety Effects 0.000 description 3
- 239000003125 aqueous solvent Substances 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 238000004440 column chromatography Methods 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 239000003814 drug Substances 0.000 description 3
- 230000036267 drug metabolism Effects 0.000 description 3
- 239000003480 eluent Substances 0.000 description 3
- 238000006911 enzymatic reaction Methods 0.000 description 3
- 229920000159 gelatin Polymers 0.000 description 3
- 235000019322 gelatine Nutrition 0.000 description 3
- 230000002440 hepatic effect Effects 0.000 description 3
- 230000002631 hypothermal effect Effects 0.000 description 3
- 230000005764 inhibitory process Effects 0.000 description 3
- 230000004060 metabolic process Effects 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 229960002715 nicotine Drugs 0.000 description 3
- SNICXCGAKADSCV-UHFFFAOYSA-N nicotine Natural products CN1CCCC1C1=CC=CN=C1 SNICXCGAKADSCV-UHFFFAOYSA-N 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 238000003653 radioligand binding assay Methods 0.000 description 3
- 201000000980 schizophrenia Diseases 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000000758 substrate Substances 0.000 description 3
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 3
- 239000003981 vehicle Substances 0.000 description 3
- 208000024827 Alzheimer disease Diseases 0.000 description 2
- 201000001320 Atherosclerosis Diseases 0.000 description 2
- 206010013710 Drug interaction Diseases 0.000 description 2
- 206010016654 Fibrosis Diseases 0.000 description 2
- 208000001613 Gambling Diseases 0.000 description 2
- 208000023105 Huntington disease Diseases 0.000 description 2
- 150000001200 N-acyl ethanolamides Chemical class 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- 206010033307 Overweight Diseases 0.000 description 2
- 206010034158 Pathological gambling Diseases 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 101100082060 Xenopus laevis pou5f1.1 gene Proteins 0.000 description 2
- 239000004479 aerosol dispenser Substances 0.000 description 2
- 125000003545 alkoxy group Chemical group 0.000 description 2
- 230000027455 binding Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 235000010980 cellulose Nutrition 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 230000007882 cirrhosis Effects 0.000 description 2
- 208000019425 cirrhosis of liver Diseases 0.000 description 2
- 229960003920 cocaine Drugs 0.000 description 2
- PAFZNILMFXTMIY-UHFFFAOYSA-N cyclohexylamine Chemical compound NC1CCCCC1 PAFZNILMFXTMIY-UHFFFAOYSA-N 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000002621 endocannabinoid Substances 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 235000011187 glycerol Nutrition 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 239000007937 lozenge Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 2
- 229960001252 methamphetamine Drugs 0.000 description 2
- -1 opioid Chemical compound 0.000 description 2
- 239000012044 organic layer Substances 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 239000003380 propellant Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- WLWFQGXZIDYWQF-UHFFFAOYSA-N (2,2-dichlorocyclopropyl)benzene Chemical compound ClC1(Cl)CC1C1=CC=CC=C1 WLWFQGXZIDYWQF-UHFFFAOYSA-N 0.000 description 1
- WDOPWTAUVPJUEF-UHFFFAOYSA-N (2-bromo-4-chlorophenyl)hydrazine;hydrochloride Chemical compound Cl.NNC1=CC=C(Cl)C=C1Br WDOPWTAUVPJUEF-UHFFFAOYSA-N 0.000 description 1
- QELCWIMDZKOJBU-UHFFFAOYSA-N (2-chloro-4-fluorophenyl)hydrazine;hydrochloride Chemical compound [Cl-].[NH3+]NC1=CC=C(F)C=C1Cl QELCWIMDZKOJBU-UHFFFAOYSA-N 0.000 description 1
- AAGDJXZCBGGXTI-UHFFFAOYSA-N (4-bromo-2-chlorophenyl)hydrazine;hydrochloride Chemical compound Cl.NNC1=CC=C(Br)C=C1Cl AAGDJXZCBGGXTI-UHFFFAOYSA-N 0.000 description 1
- HPKBNVJFGMBBFK-UHFFFAOYSA-N (4-chloro-2-fluorophenyl)hydrazine;hydrochloride Chemical compound Cl.NNC1=CC=C(Cl)C=C1F HPKBNVJFGMBBFK-UHFFFAOYSA-N 0.000 description 1
- FTPCTNNAMFNKCU-UHFFFAOYSA-N 1-(2-bromo-4-chlorophenyl)-5-(4-cyclopropylphenyl)-4-methyl-n-piperidin-1-ylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC(Cl)=CC=2)Br)C=1C(C=C1)=CC=C1C1CC1 FTPCTNNAMFNKCU-UHFFFAOYSA-N 0.000 description 1
- JUYLDKHOYZAAGS-UHFFFAOYSA-N 1-(2-chlorophenyl)-5-(4-cyclopropylphenyl)-4-methyl-n-piperidin-1-ylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC=CC=2)Cl)C=1C(C=C1)=CC=C1C1CC1 JUYLDKHOYZAAGS-UHFFFAOYSA-N 0.000 description 1
- GXXJXKLHOSSYDJ-UHFFFAOYSA-N 1-(4-cyclopropylphenyl)-2-methoxyethanone Chemical compound C1=CC(C(=O)COC)=CC=C1C1CC1 GXXJXKLHOSSYDJ-UHFFFAOYSA-N 0.000 description 1
- WHBZLGZWRBQYGB-UHFFFAOYSA-N 1-(4-cyclopropylphenyl)propan-1-one Chemical compound C1=CC(C(=O)CC)=CC=C1C1CC1 WHBZLGZWRBQYGB-UHFFFAOYSA-N 0.000 description 1
- HEZVCCWYHSHJPF-UHFFFAOYSA-N 1-aminopiperidin-3-ol Chemical compound NN1CCCC(O)C1 HEZVCCWYHSHJPF-UHFFFAOYSA-N 0.000 description 1
- KEHRVXCBFSPWOH-UHFFFAOYSA-N 1-aminopiperidin-4-ol Chemical compound NN1CCC(O)CC1 KEHRVXCBFSPWOH-UHFFFAOYSA-N 0.000 description 1
- RUSPCRNKBONOHH-UHFFFAOYSA-N 1-cyclopropyl-2-methylbenzene Chemical compound CC1=CC=CC=C1C1CC1 RUSPCRNKBONOHH-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- ADODRSVGNHNKAT-UHFFFAOYSA-N 2-Chlorophenylhydrazine hydrochloride Chemical compound Cl.NNC1=CC=CC=C1Cl ADODRSVGNHNKAT-UHFFFAOYSA-N 0.000 description 1
- UTWMSGSDWZDPOM-UHFFFAOYSA-N 2-methyl-3-phenylcyclobutan-1-one Chemical compound C1C(=O)C(C)C1C1=CC=CC=C1 UTWMSGSDWZDPOM-UHFFFAOYSA-N 0.000 description 1
- JLBJTVDPSNHSKJ-UHFFFAOYSA-N 4-Methylstyrene Chemical compound CC1=CC=C(C=C)C=C1 JLBJTVDPSNHSKJ-UHFFFAOYSA-N 0.000 description 1
- SDHNLNXASWUTHB-UHFFFAOYSA-N 5-(4-cyclopropylphenyl)-1-(2,4-difluorophenyl)-4-methyl-n-piperidin-1-ylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NN2CCCCC2)=NN(C=2C(=CC(F)=CC=2)F)C=1C(C=C1)=CC=C1C1CC1 SDHNLNXASWUTHB-UHFFFAOYSA-N 0.000 description 1
- 208000007848 Alcoholism Diseases 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 235000003911 Arachis Nutrition 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- AYFYSCDQBMILFR-UHFFFAOYSA-N CCc1c(-c2ccc(C3CC3)cc2)[n](-c(ccc(Cl)c2)c2Cl)nc1C(NN1CC(CCC2)C2C1)=O Chemical compound CCc1c(-c2ccc(C3CC3)cc2)[n](-c(ccc(Cl)c2)c2Cl)nc1C(NN1CC(CCC2)C2C1)=O AYFYSCDQBMILFR-UHFFFAOYSA-N 0.000 description 1
- 244000025254 Cannabis sativa Species 0.000 description 1
- 235000008697 Cannabis sativa Nutrition 0.000 description 1
- GSZYFYMIUWFNMQ-UHFFFAOYSA-N Cc1c(-c2ccc(C3CC3)cc2)[n](-c(c(Cl)c2)ccc2Cl)nc1C(NC1CC1)=O Chemical compound Cc1c(-c2ccc(C3CC3)cc2)[n](-c(c(Cl)c2)ccc2Cl)nc1C(NC1CC1)=O GSZYFYMIUWFNMQ-UHFFFAOYSA-N 0.000 description 1
- VJBGYBDKYNBBPL-UHFFFAOYSA-N Cc1c(-c2ccc(C3CCCC3)cc2)[n](-c(c(Cl)c2)ccc2Cl)nc1C(NN1CCOCC1)=O Chemical compound Cc1c(-c2ccc(C3CCCC3)cc2)[n](-c(c(Cl)c2)ccc2Cl)nc1C(NN1CCOCC1)=O VJBGYBDKYNBBPL-UHFFFAOYSA-N 0.000 description 1
- VVODBTYKBNTQLM-UHFFFAOYSA-O Cc1c(-c2ccc(C3CCCC3)cc2)[n](-c2ccc(C)cc2Cl)nc1C([NH2+]N1CCCC1)=O Chemical compound Cc1c(-c2ccc(C3CCCC3)cc2)[n](-c2ccc(C)cc2Cl)nc1C([NH2+]N1CCCC1)=O VVODBTYKBNTQLM-UHFFFAOYSA-O 0.000 description 1
- 208000022497 Cocaine-Related disease Diseases 0.000 description 1
- HTJDQJBWANPRPF-UHFFFAOYSA-N Cyclopropylamine Chemical compound NC1CC1 HTJDQJBWANPRPF-UHFFFAOYSA-N 0.000 description 1
- 208000020401 Depressive disease Diseases 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 206010057852 Nicotine dependence Diseases 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 208000025569 Tobacco Use disease Diseases 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- XSTXAVWGXDQKEL-UHFFFAOYSA-N Trichloroethylene Chemical group ClC=C(Cl)Cl XSTXAVWGXDQKEL-UHFFFAOYSA-N 0.000 description 1
- HQVHOQAKMCMIIM-HXUWFJFHSA-N WIN 55212-2 Chemical compound C([C@@H]1COC=2C=CC=C3C(C(=O)C=4C5=CC=CC=C5C=CC=4)=C(N1C3=2)C)N1CCOCC1 HQVHOQAKMCMIIM-HXUWFJFHSA-N 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- WETWJCDKMRHUPV-UHFFFAOYSA-N acetyl chloride Chemical compound CC(Cl)=O WETWJCDKMRHUPV-UHFFFAOYSA-N 0.000 description 1
- 239000012346 acetyl chloride Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000003708 ampul Substances 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- UGBKOURNNQREPE-UHFFFAOYSA-N azepan-1-amine Chemical compound NN1CCCCCC1 UGBKOURNNQREPE-UHFFFAOYSA-N 0.000 description 1
- 238000012925 biological evaluation Methods 0.000 description 1
- DVECBJCOGJRVPX-UHFFFAOYSA-N butyryl chloride Chemical compound CCCC(Cl)=O DVECBJCOGJRVPX-UHFFFAOYSA-N 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 210000001638 cerebellum Anatomy 0.000 description 1
- 230000004087 circulation Effects 0.000 description 1
- 201000006145 cocaine dependence Diseases 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- UJMBCHVRTIOTKC-UHFFFAOYSA-N cyclobutylbenzene Chemical compound C1CCC1C1=CC=CC=C1 UJMBCHVRTIOTKC-UHFFFAOYSA-N 0.000 description 1
- IGARGHRYKHJQSM-UHFFFAOYSA-N cyclohexylbenzene Chemical compound C1CCCCC1C1=CC=CC=C1 IGARGHRYKHJQSM-UHFFFAOYSA-N 0.000 description 1
- XCIXKGXIYUWCLL-UHFFFAOYSA-N cyclopentanol Chemical compound OC1CCCC1 XCIXKGXIYUWCLL-UHFFFAOYSA-N 0.000 description 1
- VDIHFNQRHGYISG-UHFFFAOYSA-N cyclopentylbenzene Chemical compound C1CCCC1C1=CC=CC=C1 VDIHFNQRHGYISG-UHFFFAOYSA-N 0.000 description 1
- VFSFCYAQBIPUSL-UHFFFAOYSA-N cyclopropylbenzene Chemical compound C1CC1C1=CC=CC=C1 VFSFCYAQBIPUSL-UHFFFAOYSA-N 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 235000013681 dietary sucrose Nutrition 0.000 description 1
- 239000002552 dosage form Substances 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 206010013663 drug dependence Diseases 0.000 description 1
- 238000002651 drug therapy Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 239000002532 enzyme inhibitor Substances 0.000 description 1
- 229940125532 enzyme inhibitor Drugs 0.000 description 1
- VGKUVUKESUYSQO-UHFFFAOYSA-N ethyl 4-(4-cyclopropylphenyl)-3-methyl-2,4-dioxobutanoate Chemical compound C1=CC(C(=O)C(C)C(=O)C(=O)OCC)=CC=C1C1CC1 VGKUVUKESUYSQO-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 238000010579 first pass effect Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 229940124600 folk medicine Drugs 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 230000010224 hepatic metabolism Effects 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000010874 in vitro model Methods 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 239000012669 liquid formulation Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 210000001589 microsome Anatomy 0.000 description 1
- HCOVEUUIZWEZBK-UHFFFAOYSA-N n-(2,2-diethoxyethyl)formamide Chemical compound CCOC(OCC)CNC=O HCOVEUUIZWEZBK-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- DBRLNNNHHCPCSW-UHFFFAOYSA-N n-cyclopentyl-5-(4-cyclopropylphenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxamide Chemical compound CC=1C(C(=O)NC2CCCC2)=NN(C=2C(=CC(Cl)=CC=2)Cl)C=1C(C=C1)=CC=C1C1CC1 DBRLNNNHHCPCSW-UHFFFAOYSA-N 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 235000010603 pastilles Nutrition 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- LWMPFIOTEAXAGV-UHFFFAOYSA-N piperidin-1-amine Chemical compound NN1CCCCC1 LWMPFIOTEAXAGV-UHFFFAOYSA-N 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- RZWZRACFZGVKFM-UHFFFAOYSA-N propanoyl chloride Chemical compound CCC(Cl)=O RZWZRACFZGVKFM-UHFFFAOYSA-N 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- KOPFEFZSAMLEHK-UHFFFAOYSA-N pyrazolecarboxylic acid Natural products OC(=O)C=1C=CNN=1 KOPFEFZSAMLEHK-UHFFFAOYSA-N 0.000 description 1
- 239000002287 radioligand Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- 238000013112 stability test Methods 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 239000012258 stirred mixture Substances 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000002511 suppository base Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 230000001839 systemic circulation Effects 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 231100000816 toxic dose Toxicity 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- UBOXGVDOUJQMTN-UHFFFAOYSA-N trichloroethylene Natural products ClCC(Cl)Cl UBOXGVDOUJQMTN-UHFFFAOYSA-N 0.000 description 1
- 229940086542 triethylamine Drugs 0.000 description 1
- 238000003828 vacuum filtration Methods 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 239000002676 xenobiotic agent Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to new compounds of general formula (I) acting as CBl antagonists and to their preparation.
- the compounds according to this invention can be used for the treatment of obesity, behavioral addictions, psychiatric and metabolic disorders.
- Rimonabant a diaryl-pyrazole derivative
- EP 576 357 the first potent and selective CBl antagonist.
- the CBl antagonists and inverse agonists are effective in the treatment of depression, anxiety, schizophrenia, nicotine, alcohol- and cocaine addiction, obesity, metabolic syndrome and related disorders, such as dyslipidemia and type II diabetes etc.
- diaryl- pyrazole analogues may bring remarkable improvement in the effectiveness of therapies recently used.
- the present invention relates to new compounds of formula (I),
- n is an integer from 0 to 3
- X and Y are each independently hydrogen, halogen, or C 1 -C 5 alkyl group,
- R 1 and R 2 are each independently hydrogen, halogen, C 1 -C4 alkyl group or C 1 - C 4 alkylmercapto group,
- R 3 is hydrogen, C 1 -C 4 alkyl group or C 1 -C 4 alkoxy group
- R4 is a cycloalkyl ring having about 3 to about 6 members ring, optionally substituted with a hydroxy group or a group having formula a.), b.) and c.) where
- • m is an integer from 4 to 6
- • Z is hydrogen, halogen, C 1 -C 4 alkyl, hydroxy, trifluoromethyl or methoxymethyl group, and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof.
- the invention also relates to the process of their synthesis as well as the pharmaceutical composition containing the same and the use for diseases or disorders associated with cannabinoid system e.g. psychiatric disorders such as schizophrenia (Weiser and Noy; Dialogues in Clinical Neuroscience Vol.7 No.l. 2005), depression (Witkin; Trends Pharmacol Sci. 2005 Dec 26(12):609-17) anxiety, (Griebel; Biol Psychiatry. 2005 Feb 1 57(3):261-7.), Huntington's chorea (Lastres-Becket; Neuroreport. 2003 May 6 14(6):813-6.), Alzheimer's disease (Mazzola Eur J Pharmacol.
- the present invention relates to new compounds of formula (I),
- n is an integer from 0 to 3
- X and Y are each independently hydrogen, halogen, or C 1 -C 5 alkyl group,
- R 1 and R 2 are each independently hydrogen, halogen, C 1 -C 4 alkyl group or C 1 - C 4 alkylmercapto group,
- R 3 is hydrogen, CpC 4 alkyl group or Ci -C 4 alkoxy group
- R 4 is a cycloalkyl ring having about 3 to about 6 members ring, optionally substituted with a hydroxy group or a group having formula a.), b.) and c.) where
- • m is an integer from 4 to 6
- • Z is hydrogen, halogen, C 1 -C 4 alkyl, hydroxy, trifluoromethyl or methoxymethyl group, and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof.
- the invention also relates to the process of the synthesis of compounds of formula (I).
- the meaning of halogen is fluoro, chloro, bromo and iodo atom, in the meaning OfR 1 and R 2 preferably fluoro, chloro and bromo atom.
- the meaning of the alkyl group or the alkyl group at the alkoxy group is methyl, ethyl, n-propyl, n-butyl or its branched isomers such as isopropyl, tert-butyl, sec-butyl etc.
- the invention also relates to the process of their synthesis as well as the pharmaceutical composition containing the same and the use for diseases or disorders associated with cannabinoid system e.g. psychiatric disorders such as schizophrenia (Weiser and Noy; Dialogues in Clinical Neuroscience Vol.7 No.l. 2005), depression (Witkin; Trends Pharmacol Sci. 2005 Dec 26(12):609-17) anxiety, (Griebel; Biol Psychiatry. 2005 Feb 1 57(3):261-7.), Huntington's chorea (Lastres-Becket; Neuroreport. 2003 May 6 14(6):813-6.), Alzheimer's disease (Mazzola Eur J Pharmacol.
- Compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof may be administered by any convenient method, for example by oral, parenteral buccal, sublingual, nasal, rectal or transdermal administration and the pharmaceutical compositions adapted accordingly.
- compositions containing compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof when given orally can be formulated as liquids or solids, for example syrups, suspensions or emulsions, tablets, capsules and lozenges.
- Liquid formulations of compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof generally consist of a suspension or solution of a compound of formula (I) in an appropriate liquid carrier(s), for example an aqueous solvent, such as water, ethanol or glycerin or a non-aqueous solvent, such as polyethylene glycol or an oil.
- the pharmaceutical composition can also contain a suspending agent, preservative, flavoring and coloring agents.
- a composition in the solid form of tablet can be prepared using any suitable pharmaceutical carrier(s), which are commonly used in practice, such as magnesium stearate, starch, lactose, saccharose, cellulose etc.
- a composition in the solid form of capsule can be prepared using routine encapsulation procedures.
- pellets containing the active ingredient can be prepared using standard carriers and then filled into a hard gelatine capsule; alternatively, a dispersion or suspension can be prepared using any suitable pharmaceutical carrier(s), for example aqueous gums, celluloses, silicates or oils and the dispersion or suspension then filled into a soft gelatine capsule.
- Typical parenteral compositions consist of a solution or suspension of the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof in a sterile aqueous carrier or parenterally acceptable oil, for example polyethylene glycol, polyvinyl pyrrolidone, lecithin, arachis oil or sesame oil.
- the solution can be lyophilized and then reconstituted with a suitable solvent just prior to administration.
- compositions of the present invention for nasal administration containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof may conveniently be formulated as aerosols, drops, gels and powders.
- Aerosol formulations of the present invention typically comprise a solution or fine suspension of the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof in a physiologically acceptable aqueous or non-aqueous solvent and are usually presented in a single or multidose quantities in sterile form is a sealed container, which can take the form of a cartridge or refill for use with an atomizing device.
- the sealed container may be a unitary dispensing device, such as a single dose nasal inhaler or an aerosol dispenser fitted with a metering valve which is intended for disposal once the contents of the container have been exhausted.
- the dosage form comprises an aerosol dispenser
- a propellant which can be a compressed gas, such as compressed air or an organic propellant, such as a fluorochlorohydrocarbon.
- the aerosol dosages form can also take the form of a pump- atomizer.
- Compositions containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof suitable for buccal or sublingual administration include tablets, lozenges and pastilles, wherein the active ingredient is formulated with a carrier, such as sugar and acacia, tragacanth or gelatine and glycerine etc.
- composition of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof for rectal administration are conveniently in the form of suppositories containing a conventional suppository base, such as cocoa butter.
- Composition of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof for transdermal administration include ointments, gels and patches.
- compositions of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof are preferably in the unit dose form, such as tablet, capsule or ampoule.
- the compounds of formula (I) of our invention are in vitro and in vivo effective CBl antagonists and their metabolic stability better than the common diaryl-pyrazoles stability, so the compositions containing the compounds of formula (I) as active ingredient are effective in lower dose and has lower capability for drug-drug interactions because of their higher metabolic stability than the known CBl antagonists till now.
- Microsomes Rat liver microsomes (Gedeon Richter, pool of 10); human liver microsomes (Xenotech, USA; pool of 50), CD-I mouse liver microsomes (Gedeon Richter, pool of 10).
- Metabolic stabilities were determined by incubation of test compounds with liver microsomes under first order reaction conditions. Consumption of test compound was monitored in time and unchanged test compound was quantified using high performance liquid chromatography. Intrinsic clearance (CLj n O was calculated from the consumption-time curves by determination of the actual rate of enzyme reaction under first order conditions.
- Metabolic bioavailability (F M ) was calculated comparing the intrinsic clearance to the hepatic blood flow. The metabolic bioavailability obtained is the highest possible bioavailability of the test compounds that is limited by the rate of liver metabolism.
- results of the hypothermia test show that compounds of formula (I) are in vivo potent CBl antagonist compounds.
- Example 1 The invention is illustrated by the following not limiting examples.
- Example 1 The invention is illustrated by the following not limiting examples.
- Example 1 The invention is illustrated by the following not limiting examples.
- step ,,a This compound was synthesized according to the method described in example 1 using 4- cyclopentylbenzene in step ,,a", which can be prepared by the reaction of cyclopentanol and benzene (W. F. BAILEY, P. R. GAGNIER, and J. J. PATRICIA., Journal of Organic Chemistry 49, (12) 2098-2107 (1984).
- This compound was synthesized according to the method described in example 14 using commercially available l-aminopyrrolidine in step ,,e".
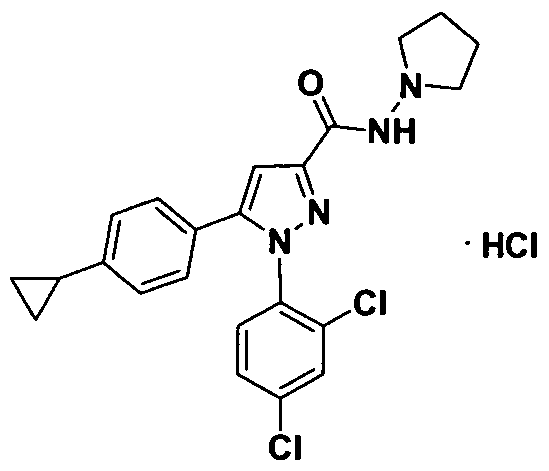
- This compound was synthesized according to the method described in example 16 using commercially available 1-aminopyrrolidine in step ,,e".
- This compound was synthesized according to the method described in example 1 starting from 2-methoxy-l-(4-cyclopropylphenyl)-ethanone (step "b").
- This compound was synthesized according to the method described in example 1 using commercially available 1-aminohomopiperidine in step ,,e".
- This compound was synthesized according to the method described in example 1 using methyl-cyclopropylbenzene in step ,,a", which can be prepared by a known method starting from commercially available methylstyrene (S. E. DENMARK and J. P. EDWARDS, Journal of Organic Chemistry 56, (25) 6974-6981 (1991).
- This compound was synthesized according to the method described in example 35 using commercially available 1 -amino-pyrrolidine in step ,,e".
Abstract
The invention relates to new compounds of general formula (I) acting as CB1 antagonists and to their preparation. The compounds according to this invention can be used for the treatment of obesity, behavioral addictions, psychiatric and metabolic disorders.
Description
Novel CBl antagonists and their preparation
Introduction
The present invention relates to new compounds of general formula (I) acting as CBl antagonists and to their preparation. The compounds according to this invention can be used for the treatment of obesity, behavioral addictions, psychiatric and metabolic disorders.
Background of the invention
Terpenoids of Cannabis sativa have been used for several thousand years as folk medicines, but their use has been banned since 1930s because of their addictive potential. A new era started in 1990s in the pharmacology of cannabionoids when the endocannabinoid system has been discovered. Identification of the receptors CBl (1990) and CB2 (1992) made it possible to develop selective agonists and antagonists and to study the endocannabinoid system (Howlett et al., 2002).
Rimonabant, a diaryl-pyrazole derivative, has been published in the patent EP 576 357 as the first potent and selective CBl antagonist. In animal models the CBl antagonists and inverse agonists are effective in the treatment of depression, anxiety, schizophrenia, nicotine, alcohol- and cocaine addiction, obesity, metabolic syndrome and related disorders, such as dyslipidemia and type II diabetes etc.
The molecules of diaryl-pyrazole structure are important among the CBl antagonists under development (Muccioli et al.: Current Medicinal Chemistry (2005), 12, 1361-94). The majority of the published CBl antagonists belong to this structural group. Their activity and medicinal application have been proven in clinical tests. The first and best known representative of this class, the above mentioned rimonabant (Acomplia®) was active and well tolerated in clinical trials of obese patients and average weight loss of 3.4 kg and 6.6 kg have observed at one year treatment in doses of 5 mg and 20 mg, resp. (LF Van Gaal et al, RIO- Europe Study (2005), Lancet, 365, 1389-1397).
Treatment of diseases e.g. obesity, connected to potential field of application of CBl antagonists often needs lifelong drug therapy. Because of high comorbidity in this condition application of several drugs at the same time is very common in clinical practice therefore there is a high risk of drug interaction incidents.
Therefore obvious need emerged for introduction of medication having good pharmacokinetic parameters resulting in less drug-drug interactions.
Compounds metabolized intensively before entering systemic circulation (first-pass effect) are highly prone to drug-drug interactions. A compound that metabolized in 80% can evoke pharmacological effect by only the remaining 20% that reaches the circulation. A coadministered enzyme inhibitor can multiply the plasma level of an enzyme substrate so much that it can reach toxic concentration. Therefore, higher metabolic stability means less capability for drug-drug interactions. High metabolic stability not only results in less capability to drug-drug interactions but it has additional advantages considering drug safety. (See: Venkatakrishnan K. et.al.: Drug metabolism and drug interactions: application and clinical value of in vitro models. Current Drug Metabolism 4, 423-459, 2003). Lower dose of a metabolically stable compound is needed to evoke the desired pharmacological effects; besides, less accumulation of metabolites occurs as well. Metabolisms of several xenobiotics results in formation of toxic metabolites.
Tremendous information is available on the mechanisms of action of diaryl-pyrazoles but their pharmacokinetic is under investigation. Only one reference has been published on the metabolism of diaryl-pyrazoles (Zhang et al., 2001); therefore, it was reasonable to study the in vitro metabolism of rimonabant. Our results showed, that in human, rat and mouse liver microsomes the rimonabant has weak/moderate metabolic stability (mouse=35%; rat=37%; human=52%).
Accordingly, potent and in vivo effective but higher metabolic stability diaryl- pyrazole analogues may bring remarkable improvement in the effectiveness of therapies recently used.
Summary of the invention
The present invention relates to new compounds of formula (I),
a.)
-N (CH2), m
b.)
— N O
c.)
wherein
• n is an integer from 0 to 3,
• X and Y are each independently hydrogen, halogen, or C1-C5 alkyl group,
• R1 and R2 are each independently hydrogen, halogen, C1-C4 alkyl group or C1- C4 alkylmercapto group,
• R3 is hydrogen, C1-C4 alkyl group or C1-C4 alkoxy group,
• R4 is a cycloalkyl ring having about 3 to about 6 members ring, optionally substituted with a hydroxy group or a group having formula a.), b.) and c.) where
• m is an integer from 4 to 6, • Z is hydrogen, halogen, C1-C4 alkyl, hydroxy, trifluoromethyl or methoxymethyl group, and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof.
The invention also relates to the process of their synthesis as well as the pharmaceutical composition containing the same and the use for diseases or disorders associated with cannabinoid system e.g. psychiatric disorders such as schizophrenia (Weiser and Noy; Dialogues in Clinical Neuroscience Vol.7 No.l. 2005), depression (Witkin; Trends Pharmacol Sci. 2005 Dec 26(12):609-17) anxiety, (Griebel; Biol Psychiatry. 2005 Feb 1 57(3):261-7.), Huntington's chorea (Lastres-Becket; Neuroreport. 2003 May 6 14(6):813-6.), Alzheimer's disease (Mazzola Eur J Pharmacol. 2003 Sep 23 477(3):219-25.), metabolic syndrome and related disorders such as obesity and being overweight (van Gaal; Lancet. 2005 Apr 16-22 365(9468): 1389-97), dyslipidemia (Despres; N Engl J Med. 2005 Nov 17 353(20):2121-34.), type II diabetes (Despres; N Engl J Med. 2005 Nov 17 353(20):2121-34.) or atherosclerosis (Gomaraschi; IDrugs. 2005 JuI 8(7):555-9.), dependence and addictive disorders such as nicotine, cocaine, opioid, alcohol, methamphetamine dependence (Fattore; Brain Res Brain Res Rev. 2006 JuI 11), social dependence, such as pathological gambling or every other diseases in which cannabinoid system plays a special role e.g. different type of cancers (Sarnataro; MoI Pharmacol. 2006 Oct 70(4): 1298-306. Epub 2006 JuI 5.), cirrhosis (Gabbay; Liver Int. 2005 Oct 25 (5):921-6.) or hemorrhagia (Cainazzo; Eur J Pharmacol. 2002 Apr 19 441(l-2):91-7).
Detailed description of the invention
The present invention relates to new compounds of formula (I),
a.)
-N (CH2) r,m
b.)
— N O
c.)
wherein
• n is an integer from 0 to 3,
• X and Y are each independently hydrogen, halogen, or C1-C5 alkyl group,
• R1 and R2 are each independently hydrogen, halogen, C1-C4 alkyl group or C1- C4 alkylmercapto group,
• R3 is hydrogen, CpC4 alkyl group or Ci -C4 alkoxy group,
• R4 is a cycloalkyl ring having about 3 to about 6 members ring, optionally substituted with a hydroxy group or a group having formula a.), b.) and c.) where
• m is an integer from 4 to 6, • Z is hydrogen, halogen, C1-C4 alkyl, hydroxy, trifluoromethyl or methoxymethyl group, and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof. .The invention also relates to the process of the synthesis of compounds of formula (I). In compounds of formula (I) the meaning of halogen is fluoro, chloro, bromo and iodo atom, in the meaning OfR1 and R2 preferably fluoro, chloro and bromo atom.
In compounds of formula (I) the meaning of the alkyl group or the alkyl group at the alkoxy group is methyl, ethyl, n-propyl, n-butyl or its branched isomers such as isopropyl, tert-butyl, sec-butyl etc.
Synthesis of compounds of formula (I)
The synthesis of compounds of formula (I) can be shown on Scheme 1.
The reaction of compound of formula (II), wherein the meaning of X, Y and n is the same as mentioned before and compound of formula (III), wherein the meaning of R3 is the same as mentioned before in the presence of AlCl3 give compound of formula (IV), wherein X, Y, n and R3 is the same as mentioned before, which is reacted with imidazol-1-yl-oxo- acetic acid ethyl ester in the presence of lithium bis(trimethylsilyl)amide to yield the diketon esters (V), wherein the meaning of X, Y, n and R3 is the same as mentioned before, then reacting the obtained compound of formula (V) with the appropriate substituted phenyl hydrazine hydrochloride (VI), wherein the meaning of R1 and R2 is the same as mentioned before to give compounds of formula (VII), wherein the meaning of X, Y, n, R1, R2 and R3 is the same as mentioned before, then hydrolyzing the ester groups of compounds of formula (VII) with KOH to give compounds having formula (VIII) wherein the meaning of X, Y, n, R1, R2 and R3 is the same as mentioned before, which is reacted with oxalyl chloride, then
reacted with compounds of formula (IX), wherein the meaning of R4 is the same as above mentioned to yield compounds of formula (I).
The invention also relates to the process of their synthesis as well as the pharmaceutical composition containing the same and the use for diseases or disorders associated with cannabinoid system e.g. psychiatric disorders such as schizophrenia (Weiser and Noy; Dialogues in Clinical Neuroscience Vol.7 No.l. 2005), depression (Witkin; Trends Pharmacol Sci. 2005 Dec 26(12):609-17) anxiety, (Griebel; Biol Psychiatry. 2005 Feb 1 57(3):261-7.), Huntington's chorea (Lastres-Becket; Neuroreport. 2003 May 6 14(6):813-6.), Alzheimer's disease (Mazzola Eur J Pharmacol. 2003 Sep 23 477(3):219-25.), metabolic syndrome and related disorders such as obesity and being overweight (van Gaal; Lancet. 2005 Apr 16-22 365(9468): 1389-97), dyslipidemia (Despres; N Engl J Med. 2005 Nov 17 353(20):2121-34.), type II diabetes (Despres; N Engl J Med. 2005 Nov 17 353(20):2121-34.) or atherosclerosis (Gomaraschi; IDrugs. 2005 JuI 8(7):555-9.), dependence and addictive disorders such as nicotine, cocaine, opioid, alcohol, methamphetamine dependence (Fattore; Brain Res Brain Res Rev. 2006 JuI 11), social dependence, such as pathological gambling or every other diseases in which cannabinoid system plays a special role e.g. different type of cancers (Samataro; MoI Pharmacol. 2006 Oct 70(4): 1298-306. Epub 2006 JuI 5.), cirrhosis (Gabbay; Liver Int. 2005 Oct 25(5):921-6.) or hemorrhagia (Cainazzo; Eur J Pharmacol. 2002 Apr l9 441(l-2):91-7).
Compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof may be administered by any convenient method, for example by oral, parenteral buccal, sublingual, nasal, rectal or transdermal administration and the pharmaceutical compositions adapted accordingly.
Pharmaceutical compositions containing compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof when given orally can be formulated as liquids or solids, for example syrups, suspensions or emulsions, tablets, capsules and lozenges.
Liquid formulations of compounds of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates
and/or solvates thereof generally consist of a suspension or solution of a compound of formula (I) in an appropriate liquid carrier(s), for example an aqueous solvent, such as water, ethanol or glycerin or a non-aqueous solvent, such as polyethylene glycol or an oil. The pharmaceutical composition can also contain a suspending agent, preservative, flavoring and coloring agents.
A composition in the solid form of tablet can be prepared using any suitable pharmaceutical carrier(s), which are commonly used in practice, such as magnesium stearate, starch, lactose, saccharose, cellulose etc.
A composition in the solid form of capsule can be prepared using routine encapsulation procedures. For example, pellets containing the active ingredient can be prepared using standard carriers and then filled into a hard gelatine capsule; alternatively, a dispersion or suspension can be prepared using any suitable pharmaceutical carrier(s), for example aqueous gums, celluloses, silicates or oils and the dispersion or suspension then filled into a soft gelatine capsule. Typical parenteral compositions consist of a solution or suspension of the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof in a sterile aqueous carrier or parenterally acceptable oil, for example polyethylene glycol, polyvinyl pyrrolidone, lecithin, arachis oil or sesame oil. Alternatively, the solution can be lyophilized and then reconstituted with a suitable solvent just prior to administration.
Compositions of the present invention for nasal administration containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof may conveniently be formulated as aerosols, drops, gels and powders. Aerosol formulations of the present invention typically comprise a solution or fine suspension of the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof in a physiologically acceptable aqueous or non-aqueous solvent and are usually presented in a single or multidose quantities in sterile form is a sealed container, which can take the form of a cartridge or refill for use with an atomizing device. Alternatively, the sealed container may be a unitary dispensing device, such as a single dose nasal inhaler or an aerosol dispenser fitted with a metering valve which is intended for disposal once the contents of the container have been
exhausted. Where the dosage form comprises an aerosol dispenser, it will contain a propellant which can be a compressed gas, such as compressed air or an organic propellant, such as a fluorochlorohydrocarbon. The aerosol dosages form can also take the form of a pump- atomizer. Compositions containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof suitable for buccal or sublingual administration include tablets, lozenges and pastilles, wherein the active ingredient is formulated with a carrier, such as sugar and acacia, tragacanth or gelatine and glycerine etc. Composition of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof for rectal administration are conveniently in the form of suppositories containing a conventional suppository base, such as cocoa butter. Composition of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof for transdermal administration include ointments, gels and patches.
The compositions of the present invention containing the compound of formula (I) and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof are preferably in the unit dose form, such as tablet, capsule or ampoule.
The compounds of formula (I) of our invention are in vitro and in vivo effective CBl antagonists and their metabolic stability better than the common diaryl-pyrazoles stability, so the compositions containing the compounds of formula (I) as active ingredient are effective in lower dose and has lower capability for drug-drug interactions because of their higher metabolic stability than the known CBl antagonists till now.
Biological evaluation
The in vitro CBl affinity of compounds of formula (I) was measured on [3H]SR- 141716A radioligand binding assay, their metabolic stability assessed using human, rat and mouse liver microsomes and their in vivo efficacy was determined by CBl agonist induced hypothermia test.
1. In vitro [3H]SR-Hl 716A radioligand binding at CBl receptors
For the evaluation of in vitro CBl affinities of compounds radioligand binding assay using [3H]SR- 141716A binding to rat cerebellum membrane preparation was employed. The assay was set up according to the methods of Thomas et al. (1998) and Devane et al. (1988). Aliquots of 200 μg rat brain membrane preparation were incubated at 30°C for 60 min in the presence of 0.04 nM [3H]SR-141716A in a thermostated shaker. Incubation was stopped by rapid vacuum filtration, filters were washed then radioactivity remaining on the filters was detected by scintillation spectrometry. Nonspecific binding was determined in the presence of 1 μM unlabelled SR-141716A. Data were analyzed and IC5O values calculated using sigmoid fitting with the software Origin 6.0 then equilibrium inhibition coefficients (Kj) calculated using the Cheng-Prusoff equation (Cheng and Prusoff, 1973).
References:
Cheng Y. and Prusoff W.H. Biochem Pharmacol 22(23): 3099-3108, 1973 Devane W.A., Dysarz F.A., Johnson M.R., Melvin L.S. and Howlett M.C. MoI Pharmacol 34 (5): 605-613, 1988
Thomas B.F., Gilliam A.F., Burch D.F., Roche MJ. and Seltzmand H.H. JPET 285 (1): 285- 292, 1998)
10
2. In vitro metabolic stability in rat, human and mouse liver microsomes
Microsomes: Rat liver microsomes (Gedeon Richter, pool of 10); human liver microsomes (Xenotech, USA; pool of 50), CD-I mouse liver microsomes (Gedeon Richter, pool of 10).
Method: Metabolic stability of the test compounds was assessed using liver microsomes (see: Houston J.B.: Utility of in vitro drug metabolism data in predicting in vivo metabolic clearance Biochemical Pharmacology 47, 1469-1479, 1994).
Metabolic stabilities were determined by incubation of test compounds with liver microsomes under first order reaction conditions. Consumption of test compound was monitored in time and unchanged test compound was quantified using high performance liquid chromatography. Intrinsic clearance (CLjnO was calculated from the consumption-time curves by determination of the actual rate of enzyme reaction under first order conditions.
Metabolic bioavailability (FM) was calculated comparing the intrinsic clearance to the hepatic blood flow. The metabolic bioavailability obtained is the highest possible bioavailability of the test compounds that is limited by the rate of liver metabolism.
Calculation of Metabolic Bioavailability: FM = (1 - EH) x 100; EH = CLint / (CLint + HBF) where CLint = Vmax / Kn, or if [S] « Kn, than CLint = v / [S] CLint = intrinsic clearance; FM = metabolic bioavailability; EH = hepatic extraction ratio; Vmax = maximal rate of enzyme reaction; Kn, = affinity constant of a substrate; [S] = substrate concentration; v = actual rate of enzyme reaction under first order conditions; HBF = hepatic blood flow
3. In vivo CBl agonist induced hypothermia
CDl mice weighted ~30g was treated orally with various doses of the test compounds (pretreatment time = 60 min, n=8/group). 15 minutes hence, CBl agonist (WIN 55,212-2, 3 mg/kg, pretreatment time = 45 min) was given
and after 45 minutes, body temperatures was measured rectally. From the mean body temperatures of the different groups, inhibition percentage" was calculated (see below), and the results were plotted as the
11
dependent variable of the logarithm of the dose. ED50 values were calculated using linear regression (Microcal Origin 6.0, linear plot).
Calculation of the inhibition percentages: inh%=100x(Mx-Ma)/(Mc-Ma); where Mx = mean body temperature of a group treated with a dose of the test compound and later with the agonist; Ma = mean body temperature of the group treated with the vehicle of the test compound and later with the agonist; Mc = mean body temperature of the group treated with vehicle of the test compound and later with the vehicle of the agonist.
Biological results
The CBl affinity of compounds of formula (I) on radioligand binding assay were the same (Ki=5.6 nM) or better (2.7 nM < Ki < 62 nM) as compared to rimonabant as main reference.
The results of in vitro metabolic stability tests are summarized in Table 1.
Table 1
12
The results show, that rimonabant has weak/moderate metabolic stability. The metabolic stability of examples (1-4) covered by general structure (I) improved in parallel to the increasing size of cycloalkyl rings and reached the highest possible value of 99% in example 4.
Results of the hypothermia test show that compounds of formula (I) are in vivo potent CBl antagonist compounds. The obtained results show that oral efficacy of compounds of formula (I) are the same (ED50=0.41mg/kg) or better (3 mg/kg > ED50 > 0.1 mg/kg) as compared to rimonabant.
Examples
The invention is illustrated by the following not limiting examples. Example 1.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-piperidin-l-yl-l-fr-pyrazole- 3-carboxamide
1 -(4-cyclopropylphenyl)propan- 1 -one
To a stirred suspension of AlCl3 (33.3 g, 0.25 mol) in trichloroethylene (50 ml) propionyl chloride (0.25 mol, 19.4 ml) was added dropwise at 0-5 °C. After stirring for 0.5 h the reaction mixture was cooled to -50 °C and cyclopropylbenzene (25 g, 0.21 mol) was added dropwise. The obtained mixture was stirred for 1 h at -50 °C then poured into a mixture of ice (250 g) and cc. HCl (30 ml). The organic layer was washed with NaHCO3 (10% solution in water), dried over anh. MgSO4 and evaporated in vacuo. The residue was purified by column chromatography using dichloromethane/hexane as eluent. Yield: 14 g (38%)
13
b.:
4-(4-cvclopropylphenyl)-3-methyl-2,4-dioxo-butyric acid ethyl ester
In a -70 0C solution of lithium bis(trimethylsilyl)amide (91 ml, 91 mmol) in methyl t-butyl ether (223 ml) under argon atmosphere l-(4-cyclopropylphenyl)propan-l-one (14 g, 0.08 mol) dissolved in methyl t-butyl ether (116 ml) was added. The reaction mixture was allowed to warm to -10 °C then imidazol-1-yl-oxo-acetic acid ethyl ester (13.5 g, 0.08 mol) dissolved in methyl t-butyl ether (70 ml) was added. The so obtained mixture was stirred for 16 h at ambient temperature then a mixture of cc. HCl (50 ml) and water (50 ml) was added. The organic layer was separated and dried over anh. MgSO4. The obtained crystalline product was used in the next step without further purification. Yield: 19.7 g (90%) c:
5-(4-cvclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-l-H-pyrazole-3-carboxylic acid ethyl ester To a stirred solution of 4-(4-cyclopropylphenyl)-3-methyl-2,4-dioxo-butyric acid ethyl ester (19.7 g, 0.072 mol) in ethanol (230 ml) 2,4-dichlorophenylhydrazine hydrochloride (15.4 g, 0.072 mol) was added at ambient temperature, followed by addition of a mixture of cc. HCl (10 ml) and water (10 ml). The obtained suspension was refluxed for 3 h, then concentrated in vacuo. The residue was purified by column chromatography using dichloromethane as eluent. Yield: 9.3 g (31%)
d.:
5-(4-cyclopropylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl- 1 -H-pyrazole-S-carboxylic acid To a stirred suspension of 5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-l-H- pyrazole-3-carboxylic acid ethyl ester (9.3 g, 0.022 mol) in methanol (28 ml) KOΗ (2.9 g, 0.051 mol) dissolved in water (28 ml) was added. The obtained suspension was refluxed for 2 h, then acidified with 3 M HCl. The precipitated product was collected by filtration and washed with water. Yield: 8.9 g (96%)
14
e.:
5-(4-cyclopropylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-iV-piperidin- 1 - yl- 1 -H-p yrazole-3 - carboxamide
To a stirred suspension of 5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-l-H- pyrazole-3-carboxylic acid (8.9 g, 0.021 mol) and DMF (0.36 ml) in dichloromethane (278 ml) oxalyl chloride (3.8 ml, 0.043 mol) was added dropwise with cooling. The mixture was stirred for 2 h at room temperature then evaporated in vacuo and dissolved in dichloromethane (80 ml). The so obtained solution was added dropwise to a stirred mixture of N-amino-piperidine (3.4 ml, 0.032 mol) and triethyl amine (4.5 ml, 0.032 mol) in dichloromethane (150 ml) at 0-5°C. The resulting mixture was allowed to warm to ambient temperature and stirred for 14-16 h then evaporated in vacuo. The residue was purified by column chromatography using dichloromethane/ethyl acetate as eluent. Yield: 8.6 g (80%)
Example 2.
5-(4-cyclobutylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-piperidin-l-yl-l-fl-pyrazole- 3-carboxamide hydrochloride
This compound was synthesized according to the method described in example 1 using 4- cyclobutylbenzene in step ,,a", which can be prepared by a known method starting from commercially available 2-phenyl-l,4-butanediol (W. F. BAILEY, P. R. GAGNIER, and J. J. PATRICIA., Journal of Organic Chemistry 49, (12) 2098-2107 (1984). 1U NMR(DMSO): 1.90-2.01 m (1Η); 1.99-2.11 m (2Η); 2.21-2.29 m (IH); 2.27 s (3H); 3.28- 3.41 m (4H); 3.44-3.55 m (IH); 7.13-7.18 m (2H); 7.23-7.27 m (2H); 7.59 d (IH); 7.77 d (IH); 7.79 d (IH)
15
Example 3.
5-(4-cyclopentylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-piperidin-l-yl-l-H-pyrazole-
3-carboxamide
This compound was synthesized according to the method described in example 1 using 4- cyclopentylbenzene in step ,,a", which can be prepared by the reaction of cyclopentanol and benzene (W. F. BAILEY, P. R. GAGNIER, and J. J. PATRICIA., Journal of Organic Chemistry 49, (12) 2098-2107 (1984).
1H NMR(DMSO): 1.44-1.56 m (4H); 1.57-1.67 m (2H); 1.70-1.79 m (2H); 1.84-1.92 m (4H); 1.95-2.04 m (2H); 2.30 s (3H); 2.89-3.01 m (IH); 3.39-3.51 m (4H); 7.13-7.18 m (2H); 7.26- 7.30 m (2H); 7.59 dd (IH); 7.78 d (IH); 7.79 d (IH); 11.60 br s (IH)
Example 4. 5-(4-cyclohexylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-piperidin-l-yl-l-J9r-pyrazole- 3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 4-cyclohexylbenzene in step ,,a".
1H NMR(DMSO): 1.16-1.26 m (IH); 1.29-1.41 m (4H); 1.46-1.56 m (2H); 1.64-1.73 m (IH); 1.73-1.81 m (4H); 1.82-1.88 m (4H); 2.28 s (3H); 2.43-2.50 m (IH); 3.34-3.48 m (4H); 7.12- 7.17 m (2H); 7.21-7.28 m (2H); 7.58 dd (IH); 7.76 d (IH); 7.79 d (IH); 11.38 br s (IH)
16
Example 5.
5-(4-cyclohexylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-iV-morpholin-l-yl-l-Hr-pyrazole-
3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 4-aminomorpholine in step ,,e".
1H NMR(DMSO): 0.63-0.70 m (2H); 0.90-0.98 m (2H); 1.81-1.92m (IH); 2.21 s (3H); 2.81-
2.87 m (4H); 3.61-3.68 m (4H); 7.06 m (4H); 7.56 dd (IH); 7.71 d (IH); 7.75 d (IH); 9.25 s
(IH)
Example 6.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-pyrrolidine-l-yl-l-/f- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 1-aminopyrrolidine in step ,,e".
1H NMR(DMSO): 0.63-0.71 m (2H); 0.90-0.99 m (2H); 1.68-1.78 m (2H); 1.81-1.93m (IH);
2.21 s (3H); 2.83-2.95 m (4H); 7.05 m (4H); 7.55 dd (IH); 7.71 d (IH); 7.74 d (IH); 9.03 s
(IH)
17
Example 7.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyI-Λ'-hexahydro- cyclopenta[c]pyrrole-2-yl-l-//-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 3-amino-3-azabicyclo[3.3.0]octane in step ,,e". 1H NMR(DMSO): 0.63-0.71 m (2H); 0.90-0.97 m (2H); 1.36-1.51 m (3H); 1.54-1.74 m (3H); 1.81-1.93 m (IH); 2.20 s (3H); 2.45-2.54 m (2H); 2.54-2.60 m (2H); 3.01-3.09 m (2H); 7.05 m (4H); 7.55 dd (IH); 7.69 d (IH); 7.74 d (IH); 8.88 s (IH)
Example 8.
5-(4-cyclopentylphenyl)-l-(2,4-dichIorophenyl)-4-methyl-N-morpholin-l-yl-l-H- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 3 using commercially available 4-aminomorpholine in step ,,e".
1H NMR(DMSO): 1.47-1.57 m (2H); 1.60-1.70 m (2H); 1.71-1.82 m (2H); 1.98-2.06 m (2H); 2.29 s (3H); 2.91-3.02 m (IH); 3.08-3.18 m (4H); 3.78-3.85 m (4H); 7.07-7.11 m (2H); 7.20- 7.23 m (2H); 7.49 dd (IH); 7.61 d (IH); 7.62 d (IH); 10.14 br s (IH)
18
Example 9.
5-(4-cyclopentylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-7V-hexahydro- cyclopenta [c] py rrole-2-yl- l-//-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 3 using commercially available 3-amino-3-azabicyclo[3.3.0]octane in step ,,e". 1H NMR(DMSO): 1.42-1.80 m (12H); 1.95-2.05 m (2H); 2.28 s (3H); 2.80-2.89 m (2H); 2.89-3.01 m (IH); 3.17-3.28 m (2H); 3.96-4.04 m (2H); 7.13-7.17 m (2H); 7.25-7.30 m (2H); 7.61 dd (IH); 7.78 d (IH); 7.81 d (IH)
Example 10.
5-(4-cyclopentylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-Λ'-pyrrolidine-l-yl-l-H- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 3 using commercially available 1-aminopyrrolidine in step ,,e".
1H NMR(DMSO): 1.42-1.53 m (2H); 1.57-1.67 m (2H); 1.68-1.78 m (2H); 1.94-2.02 m (2H); 2.04-2.13 m (4H); 2.28 s (3H); 2.91-3.00 m (IH); 3.69-3.76 m (4H); 7.13-7.19 m (2H); 7.25- 7.29 m (2H); 7.62 dd (IH); 7.77 d (IH); 7.78 d (IH)
19
Example 11.
5-(4-cyclophexylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-morpholin-l-yl-l-ZT- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 4 using commercially available 4-aminomorpholine in step ,,e".
1H NMR(DMSO): 1.17-1.27 m (IH); 1.33-1.41 m (4H); 1.68-1.75 m (IH); 1.77-1.85 m (4H); 2.27 s (3H); 2.42-2.51 m (IH); 2.88-2.95 m (4H); 3.69-3.75 m (4H); 7.05-7.08 m (2H); 7.13- 7.18 m (2H); 7.44 dd (IH); 7.54-7.57 m (2H); 9.02 s (IH)
Example 12.
5-(4-cyclohexylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-iV-hexahydro- cyclopenta[c]pyrrole-2-yl-l-/?-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 4 using commercially available 3-amino-3-azabicyclo[3.3.0]octane in step ,,e". 1H NMR(DMSO): 1.14-1.26 m (IH); 1.26-1.40 m (4H); 1.53-1.81 m (HH); 2.27 s (3H); 2.44-2.52 m (IH); 2.80-2.93 m (2H); 3.17-3.29 m (2H); 3.96-4.06 m (2H); 7.12-7.18 m (2H); 7.22-7.28 m (2H); 7.60 dd (IH); 7.77 d (IH); 7.80 d (IH)
20
Example 13.
5-[4-(2,2-dichlorocyclopropyl)-phenyl]-l-(2,4-dichlorophenyl)-4-methyl-N-pyperidine-l- yl-l-//-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 2,2-dichloro-cyclopropylbenzene in step ,,a".
1H NMR(DMSO): 1.27-1.37 m (2H); 1.52-1.65 m (4H); 2.00-2.20 m (2H); 2.28 s (3H); 2.75- 2.82 m (4H); 3.96-4.04 m (2H); 7.16-7.22 m (2H); 7.28-7.34 m (2H); 7.55 dd (IH); 7.72 d (IH); 7.73 d (IH); 9.02 s (IH)
Example 14.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-iV-piperidiii-l-yl-l-Z-r-pyrazole-3- carboxamide hydrochloride
This compound was synthesized according to the method described in example 1 using acetyl chloride in step ,,a".
1H NMR(DMSO): 0.64-0.69 m (2H); 0.91-0.98 m (2H); 1.42-1.51 m (2H); 1.76-1.83 m (4H); 1.84-1.92 m (IH); 3.25-3.35 m (4H); 7.03-7.08 m (2H); 7.10-7.15 m (2H); 7.24 s (IH); 7.65 dd (IH); 7.81 d (IH); 7.87 d (IH); 11.21 s (IH)
21
Example 15.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-Λ'-pyrrolidine-l-yl-l-H-pyrazole-3- carboxaniide hydrochloride
This compound was synthesized according to the method described in example 14 using commercially available l-aminopyrrolidine in step ,,e".
1H NMR(DMSO): 0.63-0.70 m (2H); 0.92-0.98 m (2H); 1.85-1.93 m (IH); 1.95-2.05 m (4H); 3.47-3.63 m (4H); 7.01-7.08 m (2H); 7.11-7.15 m (2H); 7.28 br s (IH); 7.66 dd (IH); 7.81 d (IH); 7.88 d (IH); 11.8O s (IH)
Example 16.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-ethyl-iV-piperidin-l-yl-l-JEr-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using butyryl chloride in step ,,a".
1H NMR(DMSO): 0.64-0.69 m (2H); 0.91-0.97 m (2H); 1.07 t (3H); 1.30-1.38 m (2H); 1.52- 1.65 m (4H); 1.82-1.92 m (IH); 2.62 q (2H); 2.74-2.81 m (4H); 7.03-7.09 m (4H); 7.51 dd (IH); 7.71 d (IH); 7.73 d (IH); 9.01 s (IH)
22
Example 17.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-ethyl-ALmorpholin-l-yl-l-ZT-pyrazole-
3-carboxamide
This compound was synthesized according to the method described in example 16 using commercially available 4-aminomorpholine in step ,,e".
1H NMR(DMSO): 0.64-0.67 m (2H); 0.91-0.98 m (2H); 1.07 t (3H); 1.82-1.92 m (IH); 2.62 q (2H); 2.80-2.88 m (4H); 3.60-3.69 m (4H); 7.04-7.10 m (4H); 7.54 dd (IH); 7.72 d (IH); 7.74 d (IH); 9.28 s (IH)
Example 18.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-ethyl-iV-pyrrolidin-l-yl-l-H-pyrazole-
3-carboxamide
This compound was synthesized according to the method described in example 16 using commercially available 1-aminopyrrolidine in step ,,e".
1H NMR(DMSO): 0.64-0.67 m (2H); 0.91-0.97 m (2H); 1.07 t (3H); 1.71-1.79 m (4H); 1.82- 1.92 m (IH); 2.62 q (2H); 2.85-2.94 m (4H); 7.03-7.09 m (4H); 7.53 dd (IH); 7.70 d (IH); 7.73 d (IH); 9.03 s (IH)
23
Example 19.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-ethyl-iV-hexahydro- cyclopenta[c]pyrrole-2-yl-l-17-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 16 using commercially available 3-amino-3-azabicyclo[3.3.0]octane in step ,,e". 1H NMR(DMSO): 0.63-0.69 m (2H); 0.91-0.97 m (2H); 1.06 t (3H); 1.36-1.52 m (3H); 1.54- 1.74 m (3H); 1.82-1.92 m (IH); 2.54-2.64 m (4H); 3.01-3.07 m (2H); 7.02-7.09 m (4H); 7.53 dd (IH); 7.70 d (IH); 7.74 d (IH); 8.90 s (IH)
Example 20.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methoxy-iV-piperidin-l-yl-l-H- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 starting from 2-methoxy-l-(4-cyclopropylphenyl)-ethanone (step "b").
1H NMR (500 MHz, DMSO-J6, 25 0C) δ 9.03 (s, IH); 7.78 (d, J - 2.3 Hz, IH); 7.75 (d, J = 8.5 Hz, IH); 7.59 (dd, J = 8.5, 2.3 Hz, IH); 7.13-7.02 (m, 4H); 3.75 (s, 3H); 2.84-2.73 (m, 4H); 1.91-1.82 (m, IH); 1.63-1.52 (m, 4H); 1.39-1.29 (m, 2H); 0.97-0.90 (m, 2H); 0.70-0.63 (m, 2H) ppm.
24
Example 21.
5-(4-cyclopropylphenyl)-l-(2-chlorophenyl)-4-methyl-N-piperidin-l-yl-lH-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 2-chloro-phenylhydrazine hydrochloride in step ,,c". 1H NMR(CDCB): 0.64-0.69 m (2H); 0.92-0.99 m (2H); 1.38-1.47 m (2H); 1.71-1.79 m (4H); 1.79-1.87 m (IH); 2.37 s (3H); 2.83-2.89 m (4H); 6.93-7.02 m (4H); 7.25-7.35 m (3H); 7.37- 7.7.43 m (IH); 7.68 s (IH)
Example 22.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-((lR,2R)-2-hydroxy- cyclohexyl)-l//-pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available tran.s-2-amino-cyclohexanol in step ,,e".
25
1H NMR(DMSO): 0.63-0.68 m (2H); 0.90-0.96 m (2H); 1.13-1.30 m (4H); 1.56-1.65 m (2H); 1.82-1.92 m (3H); 2.22 s (3H); 3.35-3.43 m (IH); 3.52-3.61 m (IH); 4.58 d (IH); 7.05 m (4H); 7.54 dd (IH); 7.68 d (IH); 7.68 s (IH); 7.74 d (IH)
Example 23.
5-(4-cyclopropylphenyl)-l-(2-chlorophenyl)-4-methyl-N-pyrrolidin-l-yl-lJ9r-pyrazole-3- carboxamide hydrochloride
This compound was synthesized according to the method described in example 22 using commercially available 1-aminopyrrolidine in step ,,e".
1H NMR(CDCl3): 0.64-0.69 m (2H); 0.93-0.99 m (2H); 1.79-1.87 m (IH); 2.23-2.31 m (4H); 2.33 s (3H); 3.86-3.93 m (4H); 6.94-7.03 m (4H); 7.29-7.41 m (4H); 9.50 br s (IH)
Example 24.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-(3-hydroxy-piperidin)-l-yl- l//-pyrazole-3-carboxamide hydrochloride
This compound was synthesized according to the method described in example 1 using 3- hydroxy-1-aminopiperidine in step ,,e".
26
1H NMR(CDCl3): 0.64-0.72 m (2H); 0.93-1.01 m (2H); 1.65-1.96 m (4H); 2.30 s (3H); 2.33- 2.44 m (IH); 3.61-4.12 m (4H); 4.34-4.43 m (IH); 6.92-7.01 m (4H); 7.24-7.38 m (4H); 10.29 br s (IH)
Example 25.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-(4-hydroxy-piperidin)-l-yl- l//-pyrazole-3-carboxamide hydrochloride
This compound was synthesized according to the method described in example 1 using 4- hydroxy-1-aminopiperidine in step ,,e".
1H NMR(DMSO): 0.65-0.69 m (2H); 0.91-0.97 m (2H); 1.44-1.55 m (2H); 1.70-1.78 m (2H); 1.83-1.91 m (IH); 2.20 s (3H); 2.66-2.74 m (2H); 2.88-2.91 m (2H); 3.44-3.51 m (2H); 4.59 d (IH); 7.04 m (4H); 7.55 dd (IH); 7.70 d (IH); 7.74 d (IH); 8.99 br s (IH)
Example 26.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-cyclohexyl-l/-r-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available cyclohexylamine in step ,,e".
27
1H NMR(DMSO): 0.64-0.70 m (2H); 0.90-0.98 m (2H); 1.20-1.41 m (6H); 1.54-1.81 m (4H); 1.83-1.91 m (IH); 2.21 s (3H); 3.70-3.81 m (IH); 7.06 m (4H); 7.55 dd (IH); 7.71 d (IH); 7.75 d (IH); 7.84 d (IH)
Example 27.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-cyclopentyl-lH-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available cyclopenylamine in step ,,e".
1H NMR(DMSO): 0.64-0.69 m (2H); 0.91-0.97 m (2H); 1.46-1.72 m (7H); 1.76-1.92 m (3H);
2.22 s (3H); 4.17-4.28 m (IH); 7.06 m (4H); 7.56 dd (IH); 7.70 d (IH); 7.75 d (IH); 7.94 d
(IH)
Example 28.
5-(4-cyclopropylphenyl)-l-(2-chloro-4-bromophenyl)-4-methyl-N-piperidin-l-yl-lZT- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 4-bromo-2-chloro-phenylhydrazine hydrochloride in step ,,c".
28
1H NMR(CDCl3): 0.65-0.73 m (2H); 0.93-1.01 m (2H); 1.39-1.46 m (2H); 1.70-1.78 m (4H); 1.79-1.87 m (IH); 2.35 s (3H); 2.81-2.91 m (4H); 6.99 m (4H); 7.18 d (IH); 7.41 dd (IH); 7.57 d (IH); 7.63 s (IH)
Example 29.
5-(4-cyclopropylphenyl)-l-(2-bromo-4-chlorophenyl)-4-methyl-N-piperidin-l-yl-lH- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 2-bromo-4-chloro-phenylhydrazine hydrochloride in step ,,c". 1H NMR(CDCl3): 0.66-0.71 m (2H); 0.94-1.01 m (2H); 1.37-1.46 m (2H); 1.71-1.79 m (4H); 1.80-1.88 m (IH); 2.36 s (3H); 2.82-2.89 m (4H); 6.96-7.02 m (4H); 7.22 d (IH); 7.30 dd (IH); 7.6O d (IH); 7.65 s (IH)
Example 30.
5-(4-cyclopropylphenyl)-l-(2-chloro-4-fluorophenyl)-4-methyl-N-piperidin-l-yl-l/f- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 4-fluoro-2-chloro-phenylhydrazine hydrochloride in step ,,c".
29
1H NMR(DMSO): 0.64-0.72 m (2H); 0.90-0.98 m (2H); 1.29-1.38 m (2H); 1.52-1.62 m (4H); 1.84-1.90 m (IH); 2.20 s (3H); 2.74-2.81 m (4H); 7.03-7.09 m (4H); 7.35 ddd (IH); 7.57 dd (IH); 7.75 dd (IH); 8.99 s (IH)
Example 31.
5-(4-cyclopropylphenyl)-l-(2-fluoro-4-chlorophenyl)-4-methyl-N-piperidin-l-yl-liϊ- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 2-fluoro-4-chloro-phenylhydrazine hydrochloride in step ,,c". 1H NMR(CDCl3): 0.63-0.73 m (2H); 0.95-1.03 m (2H); 1.39-1.46 m (2H); 1.72-1.79 m (4H); 1.80-1.88 m (IH); 2.35 s (3H); 2.82-2.91 m (4H); 6.96-7.03 m (4H); 7.08 dd (IH); 7.17 dd (IH); 7.31 dd (IH); 7.65 s (IH)
Example 32.
5-(4-cyclopropylphenyl)-l-(2,4-difluorophenyl)-4-methyl-N-piperidin-l-yl-lH-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 2,4-difluoro-phenylhydrazine hydrochloride in step ,,c".
30
1H NMR(DMSO): 0.65-0.70 m (2H); 0.91-0.98 m (2H); 1.28-1.37 m (2H); 1.54-1.63 m (4H); 1.84-1.92 m (IH); 2.19 s (3H); 2.72-2.82 m (4H); 7.03-7.08 m (4H); 7.19-7.25 m (IH); 7.35- 7.42 m (IH); 1.66-1.11 m (IH); 9.02 s (IH)
Example 33.
5-(4-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-azepan-l-yl-l-/T-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available 1-aminohomopiperidine in step ,,e".
1H NMR(DMSO): 0.64-0.72 m (2H); 0.91-0.98 m (2H); 1.53-1.59 m (4H); 1.60-1.69 m (4H); 1.84-1.92 m (IH); 2.20 s (3H); 2.95-3.03 m (4H); 7.05 m (4H); 7.55 dd (IH); 7.69 d (IH); 7.75 d (IH); 9.26 s (IH)
Example 34.
5-(4-cydopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-N-cyclopropyl-lH-pyrazole-3- carboxamide
This compound was synthesized according to the method described in example 1 using commercially available cyclopropylamine in step ,,e".
31
1H NMR(DMSO): 0.58-0.70 m (6H); 0.90-0.98 m (2H); 1.83-1.92 m (IH); 2.22 s (3H); 2.79- 2.87 m (IH); 7.06 m (4H); 7.54 dd (IH); 7.68 d (IH); 7.75 d (IH); 8.18 d (IH)
Example 35.
5-(4-methyl-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-iV-piperidin-l-yl-l-iy- pyrazole-3-carboxamide
This compound was synthesized according to the method described in example 1 using methyl-cyclopropylbenzene in step ,,a", which can be prepared by a known method starting from commercially available methylstyrene (S. E. DENMARK and J. P. EDWARDS, Journal of Organic Chemistry 56, (25) 6974-6981 (1991).
1H NMR(DMSO): 0.70-0.78m (IH); 0.83-0.89 m (IH); 1.00-1.08 m (IH); 1.12 s (3H); 1.30-
1.39 m (2H); 1.52-1.63 m (5H); 2.19 s (3H); 2.74-2.82 m (4H); 6.99-7.07 m (4H); 7.55 dd (IH); 7.71 d (IH); 7.74 d (IH); 9.00 s (IH)
Example 36.
5-(4-methyl-cyclopropylphenyl)-l-(2,4-dichlorophenyl)-4-methyl-iV-pyrrolidin-l-yl-l-/-r- pyrazoIe-3-carboxamide
This compound was synthesized according to the method described in example 35 using commercially available 1 -amino-pyrrolidine in step ,,e".
32
1H NMR(DMSO): 0.70-0.78m (IH); 0.83-0.89 m (IH); 1.00-1.09 m (IH); 1.12 s (3H); 1.53- 1.62 m (IH); 1.69-1.79 m (4H); 2.20 s (3H); 2.84-2.93 m (4H); 6.99-7.07 m (4H); 7.55 dd (IH); 7.70 d (IH); 7.75 d (IH); 9.03 s (IH)
33
Claims
1. New compounds of formula (I),
a.)
-N (CH2) 'm
b.)
— N O
c.)
• n is an integer from 0 to 3,
• X and Y are each independently hydrogen, halogen, or C1-C5 alkyl group,
• R1 and R2 are each independently hydrogen, halogen, C1-C4 alkyl group or C1- C4 alkylmercapto group,
• R3 is hydrogen, Cj-C4 alkyl group or C1-C4 alkoxy group,
34 • R4 is a cycloalkyl ring having about 3 to about 6 members ring, optionally substituted with a hydroxy group or a group having formula a.), b.) and c.) where
• m is an integer from 4 to 6, • Z is hydrogen, halogen, C1-C4 alkyl, hydroxy, trifluoromethyl or methoxymethyl group, and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof.
2. 5-(4-cyclopropylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-iV-pyrrolidine- 1 -yl- 1 -H- pyrazole-3 -carboxamide,
5-(4-cyclopropylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-N-piperidin- 1 -yl- 1 -H-pyrazole-3 - carboxamide
5-(4-cyclobutylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-iV-piperidin- 1 -yl- 1 -H-pyrazole-3 - carboxamide
5-(4-cyclopentylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-7V-piperidin- 1 -yl- 1 -H-pyrazole-3 - carboxamide
5-(4-cyclohexylphenyl)- 1 -(2,4-dichlorophenyl)-4-methyl-N-piperidin- 1 -yl- 1 -H-pyrazole-3 - carboxamide
3. Process for the synthesis of compounds of claims 1-2, characterized by a) reacting compound of formula (II), wherein the meaning of X, Y and n is the same as mentioned before with compound of formula (III), wherein the meaning of R3 is the same as mentioned before to give compound of formula (IV), wherein X, Y, n and R3 is the same as mentioned before, b) which is reacted with imidazol-1-yl-oxo-acetic acid ethyl ester in the presence of lithium bis(trimethylsilyl)amide to yield the diketon esters (V), wherein the meaning of X, Y, n and R3 is the same as mentioned before, c) then reacting the obtained compound of formula (V) with the appropriate substituted phenyl hydrazine hydrochloride (VI), wherein the meaning of Ri and
R2 is the same as mentioned before to give compounds of formula(VIi), wherein the meaning of X, Y, n, Ri, R2 and R3 is the same as mentioned before,
35 d) then hydrolyzing the ester groups of compounds of formula (VII) with KOH to give compounds having formula (VIII) as defined in claim 1 , e) which is reacted with oxalyl chloride, then reacted with compounds of formula (IX), wherein the meaning of R4 is the same as above mentioned to yield compounds of formula (I).
4. Pharmaceutical composition containing a compound of claims 1-2 and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof as well as one or more pharmaceutically acceptable adjuvant and auxiliary material.
5. Use of a compound of claims 1-2 and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof for manufacturing a pharmaceutical composition for treating and/or preventing condition requiring influence on cannabinoid receptor.
6. A use according to claim 5, wherein the cannabinoid receptor is cannabinoid CBl receptor.
7. Method of treating and/or preventing condition requiring influence on cannabinoid receptor, characterized by administering to a mammal to be treated - including human - effective amount/amounts of a compound of claims 1-2 and/or geometric isomers and/or stereoisomers and/or diastereomers and/or pharmaceutically acceptable salts and/or hydrates and/or solvates thereof.
8. A method according to claim 7, wherein the cannabinoid receptor is cannabinoid CBl receptor.
36
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| HUP0600925 | 2006-12-19 | ||
| HU0600925A HUP0600925A3 (en) | 2006-12-19 | 2006-12-19 | Dyaril-pyrazoles as cb1 antagonists, their use and pharmaceutical compositions containine them |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008075118A1 true WO2008075118A1 (en) | 2008-06-26 |
Family
ID=89987208
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/HU2007/000126 WO2008075118A1 (en) | 2006-12-19 | 2007-12-19 | Novel cb1 antagonists and their preparation |
Country Status (2)
| Country | Link |
|---|---|
| HU (1) | HUP0600925A3 (en) |
| WO (1) | WO2008075118A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102603713A (en) * | 2011-01-25 | 2012-07-25 | 范如霖 | Chiral CB1 (cannabinoid) receptor inhibitor, and preparation method and medical application thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004058744A1 (en) * | 2002-12-23 | 2004-07-15 | Sanofi-Aventis | Derivative of 5-(4-bromophenyl)-1-(2,4-dichlorophenly)-4-ethyl-n-(piperidine-1-yl)-1h-pyrazol-3-carboxamide, the preparation and therapeutic use thereof |
-
2006
- 2006-12-19 HU HU0600925A patent/HUP0600925A3/en unknown
-
2007
- 2007-12-19 WO PCT/HU2007/000126 patent/WO2008075118A1/en active Application Filing
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004058744A1 (en) * | 2002-12-23 | 2004-07-15 | Sanofi-Aventis | Derivative of 5-(4-bromophenyl)-1-(2,4-dichlorophenly)-4-ethyl-n-(piperidine-1-yl)-1h-pyrazol-3-carboxamide, the preparation and therapeutic use thereof |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102603713A (en) * | 2011-01-25 | 2012-07-25 | 范如霖 | Chiral CB1 (cannabinoid) receptor inhibitor, and preparation method and medical application thereof |
| WO2012100704A1 (en) * | 2011-01-25 | 2012-08-02 | Fan Rulin | Chiral low-toxicity cb1 receptor inhibitor, preparation method therefor, and use thereof |
| CN102603713B (en) * | 2011-01-25 | 2014-05-14 | 范如霖 | Chiral CB1 (cannabinoid) receptor inhibitor, and preparation method and medical application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| HU0600925D0 (en) | 2007-02-28 |
| HUP0600925A2 (en) | 2008-10-28 |
| HUP0600925A3 (en) | 2009-03-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1641763B1 (en) | 4-cyanopyrazole-3-carboxamide derivatives preparation and therapeutic application thereof | |
| RU2141479C1 (en) | Substituted n-piperidino-5-(4-chlorophenyl)-1-(2,4-dichloro-phenyl)-4-methylpyrazole-3-carboxamide or its salts, method of its synthesis and pharmaceutical composition based on said | |
| EP1150961B1 (en) | Pyrazolecarboxylic acid derivatives, their preparation, pharmaceutical compositions containing them | |
| EP1720837B1 (en) | Derivatives of n-' (1,5-diphenyl-1h-pyrazol-3-yl) sulphonamide with cb1 receptor affinity | |
| FR2849032A1 (en) | New 1,5-diphenyl-N-(piperidin-1-yl)-1H-pyrazole-3-carboxamide derivative, is a cannabinoid CB1 receptor antagonist useful e.g. for treating obesity, cognitive disorders or alcohol or nicotine dependence | |
| WO2006030124A1 (en) | Condensed pyrazole derivatives, preparation method thereof and therapeutic use of same | |
| WO2003084930A1 (en) | Diphenylpyridine derivatives, the preparation thereof, and pharmaceutical compositions containing said derivatives | |
| CA2554610A1 (en) | Aryl and heteroaryl-piperidinecarboxylate derivatives, the preparation and the use thereof in the form of faah enzyme inhibitors | |
| EP1230222A1 (en) | 1-benzylpyrazoles-3-carboxylic acid tricyclic derivatives as cannabinoid receptor antagonists | |
| AU2007260067A1 (en) | Phenyl-pyrazole derivatives as non-steroidal glucocoricoid receptor ligands | |
| WO2006070106A1 (en) | N-[(4, 5-diphenyl-3-alkyl-2-thienyl) methyl] amine (amide, sulfonamide, carbamate and urea) derivatives as cannabinoid cb1 receptor antagonists | |
| EP2278878A1 (en) | 2-aryl glycinamide derivatives | |
| AU2009229026A1 (en) | Azetidine-derived compounds, preparation method therefor and therapeutic use of same | |
| CA2733397A1 (en) | Azetidine polysubstituted compounds, preparation thereof, and therapeutic application thereof | |
| EP0540400A1 (en) | Quinoline derivatives, useful as angiotensin II antagonists | |
| WO2007068814A1 (en) | Diaryltriazolmethylamine derivatives, preparation and therapeutic use thereof | |
| JP2023503601A (en) | Cannabigerol derivatives and their use as cannabinoid receptor modulators | |
| WO2005035488A2 (en) | Thiophene-2-carboxamide derivatives and use thereof as cannabinoid cb-1 receptor antagonists | |
| JP2022540586A (en) | cannabinoid derivatives | |
| WO2008075118A1 (en) | Novel cb1 antagonists and their preparation | |
| JP5106410B2 (en) | 3-Acylindole derivatives, their preparation and therapeutic use | |
| FR2731223A1 (en) | NOVEL BI-TRYPTAMINE DERIVATIVES, PROCESS FOR THEIR PREPARATION AND THEIR USE AS MEDICAMENTS | |
| FR2888236A1 (en) | New N-(1,5-diphenyl-1H-pyrazol-3-yl)methylsulfonamide derivatives, useful to treat/prevent e.g. appetite disorders, metabolic disorders, gastrointestinal disorders and inflammatory disorders, are cannabinoids receptor antagonists | |
| FR2876691A1 (en) | PYRIDINE DERIVATIVES, THEIR PREPARATION, THEIR THERAPEUTIC APPLICATION | |
| EP2313392A1 (en) | Azetidine polysubstituted compounds, preparation thereof, and therapeutic application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07858826 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07858826 Country of ref document: EP Kind code of ref document: A1 |