WO2007145901A1 - Compositions enriched in neoplastic stem cells and methods comprising same - Google Patents
Compositions enriched in neoplastic stem cells and methods comprising same Download PDFInfo
- Publication number
- WO2007145901A1 WO2007145901A1 PCT/US2007/013167 US2007013167W WO2007145901A1 WO 2007145901 A1 WO2007145901 A1 WO 2007145901A1 US 2007013167 W US2007013167 W US 2007013167W WO 2007145901 A1 WO2007145901 A1 WO 2007145901A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- another embodiment
- oct4
- cell
- cancer
- cells
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0693—Tumour cells; Cancer cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0693—Tumour cells; Cancer cells
- C12N5/0695—Stem cells; Progenitor cells; Precursor cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/574—Immunoassay; Biospecific binding assay; Materials therefor for cancer
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/11—Epidermal growth factor [EGF]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/115—Basic fibroblast growth factor (bFGF, FGF-2)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/30—Hormones
- C12N2501/33—Insulin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2502/00—Coculture with; Conditioned medium produced by
- C12N2502/13—Coculture with; Conditioned medium produced by connective tissue cells; generic mesenchyme cells, e.g. so-called "embryonic fibroblasts"
- C12N2502/1323—Adult fibroblasts
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2503/00—Use of cells in diagnostics
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2503/00—Use of cells in diagnostics
- C12N2503/02—Drug screening
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/46—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans from vertebrates
- G01N2333/47—Assays involving proteins of known structure or function as defined in the subgroups
- G01N2333/4701—Details
- G01N2333/4703—Regulators; Modulating activity
Definitions
- Neoplastic stem cells isolated from tissue samples by various fractionation procedures consisted of mixed " cell types. Efficient isolation of neoplastic stem cells provides a means of exploring basic mechanisms in cancer cell biology and disease. Methods for specifically and efficiently isolating and propagating a cell subpopulation to provide a large neoplastic stem cell population for in-vitro and in- vivo studies are desirable.
- NSCs neoplastic stem cells
- FACS FACS
- antibiotic selection assays that do not distinguish between sub-populations of cells based on their biological activity and/or physiological function.
- the assays moreover, preclude recovery of native non-antibiotic-expressing or treated stem cells.
- Other methods of cellular identification and subsequent isolation and/or enrichment such as gel electrophoresis, fail to probe pure populations, suffer from contamination and/or compromise cell viability.
- This invention relates, in another embodiment, to a neoplastic stem cell population enriched for expression of OCT4.
- this invention provides a method of identifying neoplastic stem cells, comprising the steps of (i) contacting neoplastic cells with an agent which specifically interacts with
- OCT4 OCT4; and (ii) identifying cells with which the agent specifically interacts.
- this invention provides a method of isolating neoplastic stem cells, comprising the steps of (i) contacting neoplastic cells with an agent which specifically interacts with
- OCT4 OCT4; and (ii) isolating cells with which the agent specifically interacted.
- this invention provides a method of enriching a neoplastic cell population for neoplastic stem cells, comprising the steps of (i) contacting a mixed cell population comprising a plurality of cancerous cells with a vector comprising an antibiotic resistance gene operatively linked to an Oct4 promoter ; and (ii) culturing the mixed cell population in the presence of an antibiotic.
- this invention provides a method of inducing cancer comprising introducing a neoplastic stem cell population enriched for expression of OCT4 to a mammal.
- this invention provides a method of analyzing cancer progression and/or pathogenesis in- vivo, comprising the steps of (i) transplanting OCT4 ' neoplastic stem cells into an animal; and (ii) analyzing cancer progression and/or pathogenesis in the animal.
- this invention provides a method of abrogating, or inhibiting cancer comprising the step of: contacting neoplastic cells with an agent that inhibits OCT4 expression or function in said neoplastic cells.
- this invention provides a method of preventing, abrogating, or inhibiting tumor growth comprising the step of: contacting neoplastic cells with an agent that inhibits
- OCT4 expression or function in a tumor OCT4 expression or function in a tumor.
- this invention provides a method of preventing, abrogating, or inhibiting cell metastasis comprising the step of: contacting neoplastic cells with an agent that inhibits
- OCT4 expression or function in said neoplastic cells are examples of OCT4 expression or function in said neoplastic cells.
- Fig. 1 presents light micrographs of serial sections of solid tumors, probed with polyclonal anti-OCT4 antibodies and control sections.
- Fig. IA is a cross section of a chondrosarcoma tumor
- Fig. IB is a cross section of an osteosarcoma tumor
- Fig. 1C is a cross section of a glioblastoma multiforme (GBM) tumor
- Fig. ID is a cross section of fetal human testis used for positive control. Arrows indicate OCT4 positive nuclei.
- Fig. 2A demonstrates the results of semi-quantitive RT-PCR probing for OCT4, STAT3 and Nanog mRNA expression wherein ⁇ -tubulin and GFAP served as normalized controls in representative glioblastoma primary cultures (MT917, MT926, MT928, MTl 231) and cell lines (LN18, LN229, LN428, U251).
- Fig. 2B demonstrates results of a Western blot analysis of OCT4, STAT3 and Nanog protein expression wherein ⁇ -tubulin and GFAP served as normalized controls in cell lines (LNl 8, LN229, LN319, LN428, D247, U251, U373, T98G).
- FIG. 3 plots the results of 2-D quantitative PCR probing OCT4 and nanog gene expression in adherent cell cultures and floating osteosarcoma-derived spheres. Substrate-attached cultures showed significantly (p ⁇ 0.05) lower expression of OCT4 and Nanog. Correlation of OCT4 (X axis) and Nanog
- FIG. 4 illustrates clone-forming potential of glioblastoma-derived cells suppressed by OCT4 siRNA.
- Fig. 4A demonstrates results of a Western blot analysis wherein suppression of exogenous OCT4 protein in a transfected cell culture was achieved by treatment with specific OCT4 siRNA comprising the
- Fig. 4B plots the frequency of clone-formation by selected glioblastoma cells (MT317, LN-229, MT-917). Cells were co-transfected with eGFP (Green
- Fig. 5 A is a light micrograph image (x200) of suspended mammaspheres derived from an
- Fig. 5B is a fluorescent micrograph
- Fig.6 presents light micrographs (x200) of immunohistochemically stained breast cancer tumors probed for OCT4 expression. Dark punctuate staining are OCT4 positive nuclei.
- Fig. 6A is a cross section of ductal carcinoma tumor
- Fig. 6B is a cross section of breast cancer metastasis to brain.
- Fig. 7 is a fluorescent microscope (x200) image of an OCT4-EGFP transfected glioblastoma cell in methyl cellulose after first division (Fig. 7A).
- Fig. 7A A glioblastoma floating neurosphere (clone) of
- Fig. 8 is a fluorescent microscope image (x200) of cultured breast cancer cells (A), osteosarcoma (B), and glioblastoma multiforme cells (C) expressing EGFP through an OCT4 responsive promoter.
- Fig. 9 is a fluorescent microscope image of the cultured glioblastoma cell line Ln428 (x200)
- Fig. 10 illustrates FACS flow isolation graphs of subpopulations of tumor cells expressing
- M2 gate represents OCT4 positive cells.
- Fig. 1OA represents the initial FACS sorting for OCT4 positive cells of a mixed clonal-OCT4 cell population.
- 1OB represents isolated OCT4 positive cells after two passages (roughly 2 weeks) followed by FACS analysis (B) to determine their purity. This population was found to be 96.16 % pure for OCT4 protein expression.
- Fig. 11 is a graph illustrating the tumor forming potential of OCT4 positive and OCT4 negative
- FIG. 12 schematically depicts the procedure for obtaining OCT4 enriched tumor stem cells from any tumor tissue for cancer related studies including drug discovery studies.
- Stage A Preparation of Cells: 1) Surgical removal of tumor 2) Mincing and preparations to create a single cells suspension.
- Stage B Stable labeling of tumor stem cells with OCT4 responsive promoter: 1) Tumor stem cell culture in a single cell suspension for expantion and selection for tumor stem cells under the appropriate conditions 2) transfection with a plasmid comprising an EGFP gene under the control of an OCT4 responsive promoter (stage C).
- Stage C Creation of a highly pure tumor stem cell cultures: EGFP expressing cells are further selected via FACS and re-cultured for expansion resulting in bulk culture quantities.
- Stage D Dl, tumor stem cells are further studied using rigorous cell and molecular biology techniques. D2, tumor stem cells are exposed to a vast variety of drugs
- Stage E Isolated tumor stem cells are inoculated into immunodeficient mice to create xenograft tumor models followed by basic efficacy, safety (or lack of toxicity), and outcomes studies generating final drug lists.
- Fig. 13 is a microscope image (x200) of mammosphere cultures derived from an MDA-MB- 435 melanoma cell line, biomarked for the presence of NSC expressing Oct-3/4.
- Fig. 13A is a light micrograph of suspended tumor-derived spheres cultured in methylcellulose.
- Fig. 13B is a fluorescent micrograph of the suspended tumor-derived spheres shown in Fig. 13 A.
- Fig. 13C is a light micrograph of tumor spheres after attachment to the substratum.
- Fig. 13D is a fluorescent micrograph of the attached tumor spheres shown in Fig. 13C.
- the invention comprises a neoplastic stem cell (NSC) population enriched for expression of OCT4, Nanog, STAT3 or combinations thereof.
- NSCs represent a subpopulation of cells within a population comprising neoplastic cells, which is capable of initiating and maintaining cancer following a prolonged period of time.
- NSCs drive the formation and growth of tumors (Fig. 11).
- the term drive as used herein refers to guide, control, direct, initiate, go through, penetrate or combinations thereof.
- NSCs comprise properties such as longevity, self-renewal and quiescence. Ia another embodiment, NSCs comprise enhanced invasive capacity.
- NSCs are multipotent, self-renewing and are able to produce proliferating sarcospheres from sarcomas, neurospheres from brain tumors or mammaspheres from breast cancers (Fig. 5).
- NSCs are capable of keeping their self-renewal potential during 1-100 passages of in-vitro cultivation.
- NSCs are capable of keeping their self-renewal potential during 1-90 passages of in-vitro cultivation.
- NSCs are capable of keeping their self-renewal potential during 20-60 passages of in-vitro cultivation.
- NSCs express genes involved in the specific functions and/or in self-renewal of NSCs, such as OCT4, Nanog, STAT3 or combinations thereof.
- the present invention provides that solid cancer represents a population of cells derived from a common founder cell, or NSC.
- the present invention provides that tumors represent a population of cells derived from a common founder cell, or NSC.
- the present invention provides that NSC phenotype is similar in many ways to that of normal stem cells.
- the present invention provides that NSC phenotype is quite different to that of normal stem cells leading to the irregularities with respect to abnormal developmental profile.
- the present invention provides that NSC phenotype is quite different from that of normal stem cells leading to the irregularities with respect to lack of key proliferation controls.
- the present invention provides that NSC population comprises a mix of true or mother NSCs and the progenitors neoplastic cells derived from NSCs.
- the present invention provides that progenitors derived from NSCs are different in key ways from mother NSCs.
- the present invention provides that different in key ways comprise high proliferation kinetics.
- the present invention provides that NSCs are typically present in very low percentages relative to the total cancer cell population, correlating roughly to the "hostility" of the enviroment (i.e., a natural environment such as a breast NSC in its primary breast tissue location versus a breast NSC located in a metatstatic and/or foreign location such as the brain.
- the present invention provides that NSCs comprise about 0.001 to 1% of the parental primary cancer population. In another embodiment, the present invention provides that NSCs comprise about 0.005 to 1% of the parental primary cancer population. In another embodiment, the present invention provides that NSCs comprise about 0.01 to 0.1 % of the parental primary cancer population. In another embodiment, the present invention provides that NSCs comprise about 0.05 to 0.1% of the parental primary cancer population. In another embodiment, the present invention provides that NSCs comprise about 0.005 to 0.01 % of the parental primary cancer population. [0035] In another embodiment, the present invention provides that NSCs comprise about 1 to 80% of the cell population in permanent cancer cell lines parental.
- the present invention provides that NSCs comprise about 1 to 10% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 7 to 14% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 15 to 25% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 10 to 30% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 30 to 50% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 20 to 40% of the cell population in permanent cancer cell lines parental.
- the present invention provides that NSCs comprise about 50 to 80% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 1 to 5% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 5 to 10% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 3 to 8% of the cell population in permanent cancer cell lines parental. In another embodiment, the present invention provides that NSCs comprise about 7 to 10% of the cell population in permanent cancer cell lines parental.
- the present invention provides that NSCs comprise about 1 to 100% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 1 to 10% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 10 to 30% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 30 to 50% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 50 to 75% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 75 to 100% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 30 to 80% of the parental metastatic cancer population cell.
- the present invention provides that NSCs comprise about 20 to 90% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 10 to 100% of the parental metastatic cancer population cell. In another embodiment, the present invention provides that NSCs comprise about 20 to 40% of the parental metastatic cancer population cell. [0037] In another embodiment, the present invention provides that bulk cancer cells (BCCs) comprise the majority of the cancer cell population from a primary soiid tumor. In another embodiment, the present invention provides that BCCs comprise the majority of the cancer cell population from a permanent cultured cell lines derived from cancers. In another embodiment, the present invention provides that a
- BCC population lacks stem cell characteristics.
- the present invention provides that a BCC population lacks OCT-4 expression.
- the methods of the present invention provides isolation of NSCs from cancer tissue biopsies and permanent cancer cell lines. by selection of NSCs previously manipulated and biomarked to allow for detection.
- the methods of the present invention provide stably transfecting NSCs with DNA vectors which expresses fluorescent or luminescent proteins regulated by an Oct-4 responsive promoter ( Figure 12).
- the methods of the present invention provides separating NSCs from the total cancer cell population resulting in cultures of high purity using FACS sorting of fluorescent biomarkers.
- the methods of the present invention provides separating NSCs from the total cancer cell population resulting in cultures of high purity using FACS sorting of those cells that express GFP (Green Fluorescent Protein) driven by an
- the sequence of the Oct-4 CDNA of the present invention comprises the sequence: tcccttcgcaagccctcatttcaccaggcccccggcttggggcgccttccttccccatggcgggacacctggcttcggatttcgccttctcgccc cctccaggtggtggaggtgatgggccaggggggccggagccgggctgggttgatccctcggacctggctaagcttccaaggccctcctggagggccag gaatcgggccgggggttgggccaggctctgaggtgtgggggattccccatgccccccgccgtatgagttctgtggggggatggccgtactgtgggccc caggttggagtggggctagt
- Oct-4 CDNA sequence is a Homo sapiens Oct-4 CDNA sequence.
- the Oct-4 CDNA sequence is from a non-human species.
- Each possibility represents a separate embodiment of the present invention.
- the sequence of the Oct-4 CDNA of the present invention comprises the sequence: gtagtcctttgttacatgcatgagtcagtgaacagggaatgggtgaatgacatttgtgggtaggttatttctagaagttaggtgggcagcttgg aaggcagaggcacttctacagactattccttggggccacacgtaggttcttgaatcccgaatggaaaggggagattgataactggtgtgtttatgttcttaca agtcttctgcttttaaatccagtcccaggacatcaaagctctgctttttaaatccagtcccaggacatcaaagctctgcagaaagaactcgagcaattttgccaagctcctgaagcagaagaggatcacc
- the Oct-4 CDNA the present invention comprises a nucleic acid sequence homologous to SEQ. ID. NO: 2.
- the Oct-4 CDNA sequence is a Homo sapiens Oct-4 CDNA sequence.
- the Oct-4 CDNA sequence is from a non-human species. Each possibility represents a separate embodiment of the present invention.
- sequence of the OCT-4 protein of the present invention comprises the sequence: MAGHLASDFAFSPPPGGGGDGPGGPEPGWVDPRTWLSFQGPPGGPGIGPGVGPG SEVWGIPPCPPPYEFCGGMAYCGPQVGVGLVPQGGLETSQPEGEAGVGVESNSDGASPEPCTVT PGAVKLEKEICLEQNPEESQDIKALQKELEQFAKLLKQKRITLGYTQADVGLTLGVLFGKVFSQT ⁇ CRFEALQLSFKNMCKLRPLLQKWVEEADNNENLQEICKAETLVQARKRKRTSIENRVRGNLE NLFLQCPKPTLQQISHIAQQLGLEKDVVRVWFCNRRQKGKRSSSDYAQREDFEAAGSPFSGGPV SFPLAPGPHFGTPGYGSPHFTALYSSVPFPEGEAFPPVSVTTLGSPMHSN (SEQ.
- the OCT-4 protein of the present invention comprises an amino acid sequence homologous to SEQ. ID. NO: 3.
- the OCT-4 protein is a Homo sapiens OCT-4 protein.
- the OCT-4 protein is from a non-human species. Each possibility represents a separate embodiment of the present invention.
- sequence of the OCT-4 protein of the present invention comprises the sequence:
- the OCT-4 protein of the present invention comprises an amino acid sequence homologous to SEQ. ID. NO: 4.
- the OCT-4 protein is a Homo sapiens OCT-4 protein.
- the OCT-4 protein is from a non-human species. Each possibility represents a separate embodiment of the present invention.
- the sequence of the Oct-4 responsive promoter of the present invention comprises the cacccaggggcggggccagaggtcaaggctagagggtggg (SEQ. ID NO: 5).
- the Oct-4 responsive promoter of the present invention comprises a nucleic acid sequence homologous to SEQ. ID. NO: 5.
- the Oct-4 responsive promoter sequence is a murine Oct-4 responsive promoter sequence.
- the Oct-4 responsive promoter sequence is from a Homo- sapiens.
- the Oct-4 responsive promoter sequence is from a non-human species.
- the Oct-4 DNA sequence of the present invention is at least 60% homologous to anyone SEQ. ED NOs: 1-2. In another embodiment, the Oct-4 DNA sequence of the present invention is at least 70% homologous to anyone SEQ. ID NOs: 1-2. In another embodiment, the Oct-4 DNA sequence of the present invention is at least 80% homologous to anyone SEQ. ID NOs: 1-2. In another embodiment, the Oct-4 DNA sequence of the present invention is at least 90% homologous to anyone SEQ. ID NOs: 1-2. Ih another embodiment, the Oct-4 DNA sequence of the present invention is at least 95% homologous to anyone SEQ. ID NOs: 1-2.
- the Oct-4 responsive promoter DNA sequence of the present invention is at least 60% homologous to anyone SEQ. ID NOs: 5. In another embodiment, the Oct-4 responsive promoter DNA sequence of the present invention is at least 70% homologous to anyone SEQ. ID NOs: 5. In another embodiment, the Oct-4 responsive promoter DNA sequence of the present invention is at least 80% homologous to anyone SEQ. ID NOs: 5. In another embodiment, the Oct-4 responsive promoter DNA sequence of the present invention is at least 90% homologous to anyone SEQ. ED NOs: 5. In another embodiment, the Oct-4 responsive promoter DNA sequence of the present invention is at least 95% homologous to anyone SEQ. ID NOs: 5.
- the Oct-4 protein sequence of the present invention is at least 60% homologous to anyone SEQ. ED NOs: 3-4. In another embodiment, the Oct-4 protein sequence of the present invention is at least 70% homologous to anyone SEQ. ID NOs: 3-4. In another embodiment, the Oct-4 protein sequence of the present invention is at least 80% homologous to anyone SEQ. ID NOs: 3-4. In another embodiment, the Oct-4 protein sequence of the present invention is at least 90% homologous to anyone SEQ. ID NOs: 3-4. In another embodiment, the Oct-4 protein sequence of the present invention is at least 95% homologous to anyone SEQ. TD NOs: 3-4.
- the methods of the present invention provide a highly pure biomarked NSC population.
- the methods of the present invention provides that a highly pure biomarked NSC population is studied in numerous ways by taking advantage of their fluorescent properties ( Figures 7 - 9).
- the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 5 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 8 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 10 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 15 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 20 passages.
- the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 25 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 30 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 35 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 40 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 45 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged without loosing their NSC phenotype for at least 50 passages.
- the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 5 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 10 passages. [0049] In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 15 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 20 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 25 passages.
- the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 30 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 35 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 40 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 45 passages. In another embodiment, the methods of the present invention provide that NSCs can be passaged and retain Oct-4 expression for at least 50 passages.
- the methods of the present invention provide that NSC populations are expanded into large volume mass cultures for extended periods of time without losing their desired pure NSC phenotype. In another embodiment, the methods of the present invention provide that NSC populations are expanded into large volume mass cultures for extended periods of time without losing their Oct-4 expression.
- NSCs are enriched for a stem cell marker.
- the stem cell marker is OCT4, Nanog, STAT3 or combinations thereof.
- the stem cell marker is a transcription factor such as OCT4.
- OCT4 is differentially expressed in NSCs.
- immunological methods of enriching for OCT4 expressing cells based on their affinity to surface antigens are used.
- NSCs are enriched by an immunomagnetic based cell separation technique.
- NSCs are enriched by the electrophoretic cell separation technique based on the electrophoretic mobility reduction via incubation with antibodies specific to surface antigen.
- NSCs are further enriched through fluorescence-activated cell sorter (FACS), immunomagnetic beads, or magnetic-activated cell sorter (MACS).
- FACS fluorescence-activated cell sorter
- MCS magnetic-activated cell sorter
- mixed populations of cancerous cells are grown under nonadherent cell culture conditions, wherein NSCs form spherical clusters of cells ("spheres") from which OCT4 positive NSCs can be enriched.
- NSCs form spherical clusters of cells
- cells derived from free floating spheres express higher levels of OCT4 and Nanog mRNA than equivalent, adherent cell cultures as shown in Fig. 3.
- the cells comprising the spheres are free floating.
- in-vitro enrichment of NSCs from breast tumor specimens is carried out using a nonadherent mammasphere cell culture system.
- in-vitro enrichment of NSCs from bone sarcoma tumor cells is carried out using a nonadherent sarcosphere cell culture system.
- in-vitro enrichment of NSCs from brain tumor cells is carried out using a nonadherent neurosphere cell culture system.
- in-vitro enrichment of NSCs from brain tumor cells is carried out using free floating spheres.
- the NSC-enriched subpopulation of cancerous cells is at least 60% positive for OCT4 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 70% positive for OCT4 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 80% positive for OCT4 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 80% positive for Nanog expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 80% positive for STAT3 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 80% positive for the expression of OCT4, STAT 3, Nanog or combinations thereof.
- the NSC-enriched subpopulation of cancerous cells is at least 90% positive for OCT4 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 90% positive for Nanog expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 90% positive for STAT3 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 90% positive for the expression of OCT4, STAT 3, Nanog or combinations thereof. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 95% positive for OCT4 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 95% positive for Nanog expression.
- the NSC-enriched subpopulation of cancerous cells is at least 95% positive for STAT3 expression. In another embodiment, the NSC-enriched subpopulation of cancerous cells is at least 95% positive for the expression of OCT4, STAT 3, Nanog or combinations thereof.
- the invention provides that the level of NSC-enriched subpopulation of cancerous cells is determined by FACS analysis (Fig. 10), in-situ hybridization, inimunohistochemistry or a combination thereof, as described in the material and methods section.
- the NSC-enriched population is characterized by OCT4 hl expression.
- OCT4 hl expression is at least twice as high as ⁇ -actin expression.
- OCT4 hl expression is at least four times as high as ⁇ -actin expression.
- the NSC-enriched population is further characterized by high expression of Nanog, STAT3, or combinations thereof.
- the expression ievel of OCT4, Nanog or STAT3 is determined by the mRNA transcription level.
- the transcription levels are determined by quantitative or semi-quantitative PCR or RT-PCR methods as shown in Fig. 2A and described in the materials and methods section.
- the expression level of OCT4, Nanog or STAT3 is determined by the protein expression level.
- the protein expression level is determined by western blot analysis as shown in Fig. 2B and described in the materials and methods section.
- protein expression level is determined indirectly by using a reporter gene.
- the reporter gene comprises an EGFP construct.
- the OCT4 expression level in an OCT4-EGFP transfected glioblastoma cell culture (Figs. 7 and 8) is determined as ⁇ , described in the materials and methods section.
- the NSC subpopulation is enriched from "soft" or "hard” tumors.
- "hard” tumors include all tumors except leukemia, lymphomas, melanomas, and multiple myeloma, which, in another embodiment, are classified as "soft.”
- the NSC subpopulation is enriched from isolated metastatic cells.
- the NSC subpopulation is enriched from a tissue culture comprising cells derived from a tumor-derived cell line.
- the subject invention comprises a composition comprising a population of NSCs enriched for expression of OCT4.
- the invention comprises a population of NSCs enriched for expression of OCT4 hl .
- the composition further comprises an appropriate environment, such as those described herein, wherein, a NSC can be induced to proliferate and generate NSC progeny.
- the term environment in which NSC progeny are placed refers to the combination of external or extrinsic physical and/or chemical conditions that affect and influence the growth and development of NSCs.
- the environment can be ex-vivo or in-vivo.
- the circulatory system blood and lymphatic
- the environment is ex-vivo and comprises NSCs placed in cell culture medium in an incubator. [0059]
- the environment further comprises cell culture medium comprising DMEM/F12.
- the cell culture medium further comprises methylcellulose in a final concentration of less than 3%, more preferably, less than 1.5%.
- the medium is supplemented with 8-20% fetal bovine serum (FBS), 30-70% media derived from cultures of primary human foreskin fibroblasts, or a combination thereof.
- FBS fetal bovine serum
- the medium further comprises screening agents which bind OCT4.
- the medium further comprises screening agents which interact with an OCT4 responsive element.
- the medium is further supplemented with 5-5OnM of progesterone, 5- 500 ⁇ M putrescine, 2-lOOng/ml recombinant EGF, 20-4OnM sodium selenit, 10-40 ⁇ g/ml transferring, 5- 50 ⁇ g/ml insulin. 2-100ng/ml recombinant FGF2 or a combination thereof.
- the medium is supplemented with 8-20% fetal bovine serum (FBS), 30-70% media derived from cultures of primary human foreskin fibroblasts, or a combination thereof.
- the medium comprises nucleic acids.
- the medium comprises a plasmid DNA.
- the plasmid DNA comprises an OCT4 responsive promoter.
- the OCT4 responsive promoter is linked to a reporter gene (Fig. 8).
- the OCT4 responsive promoter is linker to an antibiotic resistance gene.
- the medium comprises siRNA.
- the siRNA antisense encodes for anti-OCT4, anti-Nanog, anti-STAT3 or combinations thereof.
- the anti-OCT4 siRNA inhibits clone formation (Fig. 4B) by inhibiting de-novo production of OCT4 protein (Fig. 4A).
- cells are plated in ultra low attachment plates.
- the cells are kept in an incubator maintaining a temperature at 36-42°C.
- the incubator further maintains 4-8% CO 2 .
- the incubator maintains 90-100% humidity.
- cells are plated in a final density of lxl0 2 -lxl0 6 cells/cm 2 .
- NSCs of the present invention are derived from a cell line.
- NSCs of the present invention are derived from a primary cell culture.
- the primary cell culture comprising NSCs is derived from a tumor or cell metastasis.
- the invention comprises tumors and cell metastasis which comprise NSCs.
- tumors and cell metastasis are derived from but not limited to: adrenocortical carcinoma, anal cancer, bladder cancer, brain tumor, brain stem glioma, brain tumor, cerebellar astrocytoma, cerebral astrocytoma, ependymoma, medulloblastoma, supratentorial primitive neuroectodermal, pineal tumors, hypothalamic glioma, breast cancer, carcinoid tumor, carcinoma, cervical cancer, colon cancer, endometrial cancer, esophageal cancer, extrahepatic bile duct cancer, ewings family of tumors (pnet), extracranial germ cell tumor, eye cancer, intraocular melanoma, gallbladder cancer, gastric cancer, germ cell tumor, extragonadal gestational trophoblastic tumor, head and neck cancer, hypopharyngeal cancer, islet cell carcinoma,
- the invention provides a method of identifying NSCs, comprising the steps of contacting neoplastic cells with an agent which specifically interacts with OCT4 through its employment to a cell culture comprising primary cell culture or a cell line culture.
- NSCs subpopulation is identified in "soft or hard” tumor.
- "Hard” tumors include all tumors except leukemia, lymphomas, melanomas, and multiple myeloma, which are classified as "soft.”
- NSCs are identified among metastatic cells.
- the invention provides a method of identifying NSCs, comprising the steps of contacting neoplastic cells with an agent which specifically interacts with OCT4 and identifying the cells with which the agent specifically interacted, as described herein.
- the agent identifying OCT4 interacts with-the cell membrane.
- the agent interacts with the POU5F1 gene encoding OCT4 or a fragment thereof.
- the agent interacts with the mRNA encoding OCT4 or a fragment thereof.
- the agent interacts with the OCT4 protein or a fragment thereof.
- the agent interacts with a specific post translational form of OCT4 such as, but not limited to, the phosphorylated OCT4 protein.
- the invention provides a method of identifying NSCs using a DNA probe that specifically interacts with OCT4 mRNA in a DNA-RNA heteroduplex.
- the method of identifying NSCs utilizes an RNA probe that specifically interacts with OCT4 mRNA in an RNA-RNA homoduplex.
- the method of identifying NSCs utilizes a peptide nucleic acid (PNA) probe that specifically interacts with OCT4 mRNA in a PNA-RNA heteroduplex.
- the nucleic acid probe or PNA further comprises a label which can be readily identified.
- the methods utilize a specific probe comprising a nucleic acid that enables selective identification of OCT4 expressing cells.
- the invention provides a method for identification of NSCs comprising a ligand that specifically interacts with OCT4 protein or a fragment thereof. In another embodiment, the invention provides a method for identification of NSCs comprising a ligand that specifically interacts with OCT4 protein or a fragment thereof. In another embodiment, a monoclonal or polyclonal anti-OCT4 antibody is utilized to detect OCT4.
- the invention provides a method of detecting OCT4 expressing cells.
- the detection method is direct, wherein a radioactive label is used, which in another embodiment comprises a radioactive compound such as 32 P or 125 I.
- direct labeling comprises a fluorescent, chemilum ⁇ nescent, or gold label.
- the detection method is indirect comprising a nucleic acid probe similar to immunohistochemical probes as known to one skilled in the art.
- probes may be labeled with hapten or biotin used to bring an enzyme which creates the detectable event (e.g., chemiluminescent, colorimetric or fluorescent) to the site of hybridization.
- a secondary labeled antibody specifically identifying the primary antibody is utilized.
- the methods utilizing a specific probe comprising an antibody enable selective identification of OCT4 expressing cells.
- a heterogeneous cell population for OCT4 expression is transfected with a plasmid comprising an OCT4 responsive promoter controlling the expression of an identifiable, reporting gene product.
- the identifiable gene product comprises green fluorescent proteins such as but not limited to: GFP, Emerald, Azami Green, or ZsGreenl; blue fluorescent proteins such as but not limited to: EBFP or Sapphire; cyan fluorescent proteins such as but not limited to: Cerulean, ECFP, AmCyanl or Midoriishi-Cyan; yellow fluorescent proteins such as but not limited to: ZsYellowl , PhiYFP, Citrine, or Venus; orange fluorescent proteins such as but not limited to: Kusabira-Orange or mOrange; red fluorescent proteins such as but not limited to:, DsRed, HcRed, mPlum, mRaspberry, mTomato, mStrawberry or green-to-red fluorescent Dendra.
- the identifiable gene product serves as
- the invention provides a method of identifying NSCs expressing OCT4, which comprises visualizing the probed NSCs.
- visualization of NSCs expressing OCT4 is carried out by exposing the labeled specimen to a film.
- visualization of NSCs expressing OCT4 can be performed with a fluorescent microscope.
- visualization of NSCs expressing OCT4 can be performed with a confocal microscope.
- visualization of NSCs expressing OCT4 can be performed with an electron microscope.
- a light microscope is used for visualization of NSCs expressing OCT4, while in another embodiment, the signal is detectable using the naked eye.
- the results of the above mentioned visualization methods can be further recorded and/or visualized on a CCD camera.
- the invention provides a method of isolating neoplastic stem cells, comprising the steps of contacting neoplastic cells with an agent which specifically interacts with OCT4.
- a cell culture comprising primary cell culture or a cell line culture is employed.
- the invention provides cell separation methods which include cell isolation methods.
- tissue dissociation techniques are utilized prior to cell separation methods.
- enzymes such as liberase, trypsin, elastase, dispase, collagenase or combinations thereof are employed for effective tissue dissociation.
- further trituration with a pipette tip to break apart the cell aggregates is needed.
- the invention provides a method of isolating neoplastic stem cells, comprising the steps of contacting neoplastic cells with an agent which specifically interacts with OCT4 and isolating the cells with which the agent specifically interacts, as described.
- the methods described previously for identification of neoplastic stem cells particularly the steps of contacting neoplastic cells with an agent which specifically interacts with OCT4 protein or mRNA, are also used for isolation of NSCs.
- the invention provides a heterogeneous cell population transfected with a plasmid comprising an Oct4 responsive promoter controlling the expression of an identifiable and/or selectable gene product (Fig. 8).
- a plasmid comprising an Oct4 responsive promoter controlling the expression of an identifiable and/or selectable gene product
- the methods described previously for identification of neoplastic stem cells comprising the use of various identifiable fluorescent protein sequences are also employed for cell separation methods.
- the identifiable gene product is used selectively to isolate OCT4 expressing cells resulting in a uniform OCT4 expressing NSCs.
- NSCs expressing OCT4 are separated in chromatography columns in which antibodies specific to OCT4 that are attached to the column bind OCT4 expressing NSCs and thereby separate them.
- an agent that is covalently bound to magnetic particles and that specifically interacts with OCT4 is employed to retain OCT4 expressing NSCs in a magnetic field.
- sorting of OCT4 expressing NSCs labeled with antibodies comprising a fluorescent label, through a FACS is used to separate NSCs from a heterogeneous population of cells as shown in Fig. 10.
- the separation methods as described herein results in an isolated population of OCT4 expressing cells.
- the invention provides methods of enriching NSCs expressing OCT4.
- a primary cell culture is enriched for OCT4 expressing cells.
- the primary cell culture for which methods for enriching OCT4 expressing NSCs is employed is derived from a soft tumor, a hard tumor, or a metastatic cell population.
- the OCT4 expressing NSC subpopulation is enriched from a tissue culture comprising cells derived from a cell line.
- the invention provides methods of enriching OCT4 expressing NSCs which comprise transfection of a heterogeneous cell population with a plasmid comprising an Oct4 responsive promoter controlling the expression of a selectable gene product (Fig. 8).
- the selectable gene encodes an antibiotic resistance protein.
- the cell enrichment methods further comprise the selecting agent.
- the selecting agent is an antibiotic which selectively eradicates non-OCT4 expressing cells resulting in an enriched OCT4 expressing NSC cell population.
- the invention provides a method of inducing cancer comprising introducing a neoplastic stem cell population enriched for expression of OCT4 to a mammal.
- the method of inducing cancer comprises promoting cell growth that leads to cancer.
- the method of inducing cancer comprises providing metastatic cells that induce cancer.
- NSCs of the invention isolated from maspheres, sarcospheres or neurospheres are used as cancer inducers.
- an animal is inoculated with NSCs.
- NSCs are injected intravenously.
- NSCs are injected into the bone.
- NSCs are injected into an animal intradermally, intramuscularly or intraperitoneally.
- NSCs are injected directly to the mammary gland of a model animal.
- inoculation comprises injection of NSCs into the fat pads of a model animal.
- the invention provides methods of inducing cancer.
- the methods of inducing cancer as described herein are performed in immunodeficient rodents.
- the immunodeficient rodent is a nude mouse or rat.
- the immunodeficient rodent is a SCID mouse.
- the immunodeficient rodent is an NIH-III mouse.
- the invention provides a method of inducing tumors or metastases comprising introducing a neoplastic stem cell population enriched for expression of OCT4 to a mammal.
- orthotopical or ectopical tumors are being induced (Fig. 1 1).
- metastases take place through the lymphatic system, through the bloodstream, by spreading through body spaces, or through implantation.
- the invention provides a method of analyzing cancer progression and/or pathogenesis in- vivo comprising transplanting OCT4 hl neoplastic stem cells into an animal; and analyzing cancer progression and/or pathogenesis in an animal.
- cancer comprises carcinoma, sarcoma, lymphoma, leukemia, or myeloma.
- NSCs of the invention are labeled by transfecting OCT4 hl neoplastic stem cells with a fluorescent protein.
- the identifiable gene product comprises various fluorescent proteins as described hereinabove.
- the identifiable gene product comprises a luminescent protein.
- the luminescent protein is luciferase.
- isotopes are used for tracking the transplanted OCT4 hl neoplastic stem cells in the animal model.
- the isotopes comprise 32 P, 125 1, 124 1, 123 I, 14 C, 109 Cd, 51 Cr, 67 Cu, 179 Ta, 111 In, 18 F, or combinations thereof.
- a magnetic label is used for cell detection.
- the transplanted labeled cells of the invention were tracked with a single-photon emission— computed tomographic (SPECT) scanner, a positron emission tomography (PET) scanner, or single photon emission commuted tomography.
- SPECT computed tomographic
- PET positron emission tomography
- MRI magnetically
- CCD charge-coupled device
- LED flashlights with excitation filter and an emission filter are used for detection of fluorescently labeled cells.
- light box with fiber-optic lighting at about 490 nm and filters, placed on top of the light box, are used to image large tumors.
- small tumors and metastases are visualized using a fluorescence dissecting microscope that incorporates a light source and filters for excitation at about 490 nm.
- color CCD cameras as well as dual-photon lasers are used for ultra-high-resolution in-vivo imaging of fluorescent protein expression.
- the invention provides a method of analyzing cancer progression and/or pathogenesis in-vivo including determining cell metastasis.
- analysis of cell metastasis comprises determination of progressive growth of cells at a site that is discontinuous from the primary tumor.
- the site of cell metastasis analysis comprises the route of neoplastic spread.
- cells can disperse via blood vasculature, lymphatics, within body cavities or combinations thereof.
- cell metastasis analysis is performed in view of cell migration, dissemination, extravasation, proliferation or combinations thereof.
- the invention provides a method of analyzing cancer progression and/or pathogenesis in-vivo.
- analysis of cancer progression and/or pathogenesis in-vivo comprises determining the extent of tumor progression.
- analysis comprises the identification of the tumor (Fig. 11 ).
- analysis of tumor progression is performed on the original tumor or "primary tumor”.
- analysis is performed over time depending on the type of cancer as known to one skilled in the art (Fig. 1 1).
- further analysis of secondary tumors originating from metastasizing cells of the primary tumor is analyzed in-vivo.
- the size and shape of secondary tumors are analyzed.
- further ex-vivo analysis is performed.
- the frequency of OCT4 expressing cells in chondrosarcoma or oteosarcoma tumors is assessed as shown in Fig. 1.
- the terms assessed, screened, evaluated and analyzed are used interchangeably.
- pathological samples of metastasis or tumors are evaluated at specific points in time, as known to one skilled in the art.
- quantitative or qualitative methods assessing tumor suppressor genes, oncogenes, apoptotic genes, signal transduction genes, receptors, transcription factors, ligands or combinations thereof comprising: PCR, western-blot, northern blot, southern blot, immunohistochemical or in situ hybridization analysis are further employed.
- tumor or metastatic cells are isolated from pathological samples for further analysis.
- tumor or metastatic cells are isolated from pathological samples and grown in culture.
- the cell proliferation potential of the primary tumor cell culture is assessed.
- OCT4 positive cells are isolated and/or enriched from the pathological sample comprising tumor or metastatic cells according to the methods described hereinabove.
- the OCT4 positive cells isolated from a tumor are further analyzed.
- various agents are further employed to the tumor or metastasis primary cell culture.
- the agent is a carcinogen.
- the agent is a pro-apoptotic agent or a differentiating agent.
- the invention provides a method of assessing the effect of a carcinogen on a primary cell culture.
- the carcinogen comprises, but is not limited to, carcinogenic substances in categories 1 through 3 of the International Agency for Research on Cancer (IARC).
- the invention provides a method of assessing the effect of a therapeutic agent on a primary cell culture derived from a tumor or a metastasis.
- therapeutic agents are screened ex-vivo, on a tumor-, or metastasis-derived primary cell culture.
- the therapeutic agents comprise interferons, interleukins, colony-stimulating, alkylating agents, nitrosoureas, antimetabolites, antitumor antibiotics, plant (vinca) alkaloids, steroid hormones or combinations thereof.
- the therapeutic agent is a chemotherapy agent.
- the chemotherapy agent is non-specific and hence may kill a cancerous cell during any phase of the cell-cycle.
- the chemotherapy agent is specific and is thus able to kill a cancerous cell during a specific phase of the cell-cycle.
- the present invention provides that heterogeneous cancer cell populations derived from clinical tumor specimens (whether primary or metastatic) or from permanent tumor cell lines can be manipulated to allow for the isolation and propagation of their respective cancer stem cell populations.
- the present invention provides methods for the identification, sorting and stable maintenance in culture subsets of NSCs based on their ability to maintain the expression of fluorescent (or luminescent) proteins driven by the promoter of the Oct3/4 transcription factor.
- the present invention provides that Oct3/4 transcription factor in concert with SOX-2, Nanog and STAT3, are the regulators of normal stem cell phenotype in the context of embryonic development including the process of self-renewal.
- the present invention provides that in the context of cancer, NSCs do not have the appropriate proliferation controls allowing the process of self-renewal to go unchecked resulting in dysplastic tissue mass at site of proliferation.
- the methods of the present invention provide the use of biomarkers that are regulated in parallel to the molecular machinery mentioned above. In another embodiment, these regulated biomarkers monitor the "sternness" of a given cancer cell.
- the present invention allows for the monitoring of the relative viability and "sternness" of the NSC population.
- the present invention provides that NSCs are responsible for metastasis to systemic organs. In another embodiment, the present invention provides that NSCs are required for metastasis to systemic organs. In another embodiment, the present invention provides that NSCs are responsible for recurrent cancer growth in the primary location after attempts at treatment (i.e., surgery, radiation, and chemotherapy). In another embodiment, the methods of the present invention provide a platform which allows for the identification of drugs that target those NSCs with metastatic potential.
- the method of the present invention is carried out using cells cultured in miniaturized format.
- cells cultured in miniaturized format of the present invention comprise multi-well plates.
- a multi-well plate of the present invention comprises 96 wells.
- a multi-well plate of the present invention comprises 384 wells (example 2).
- a multi-well plate of the present invention comprises 1536 wells.
- a multi-well plate of the present invention comprises from 2-5000 wells.
- a multi-well plate of the present invention comprises from 20-3000 wells.
- a multi-well plate of the present invention comprises from 96-2000 wells.
- the invention provides a method of evaluating the effect of photodynamic therapy (PDT) on tumor derived primary cell culture.
- PDT photodynamic therapy
- the effect of radiation therapy or radiofrequency ablation alone or in combination with any other form of a therapeutic agent on tumor primary cell culture is further assessed.
- the effect of chemoembolization on tumor derived primary cell culture is analyzed.
- the effect of local hyperthermia on tumor derived primary cell culture is analyzed.
- the in- vivo effect of various agents and conditions is desired.
- the invention provides a method wherein an agent of interest is further administered in-vivo to an animal that has been transplanted with OCT4 expressing NSCs.
- the OCT4 expressing NSCs express OCT4 hl .
- administration of an agent is according to procedures known to one skilled in the art.
- single or multiple administrations of an agent or agents are required, as known to one skilled in the art.
- the agent or agents are administered over a period of days to weeks or over a period of months to years, depending on cancer progression and/or regression, as known to one skilled in the art.
- the agent is a carcinogen which in another embodiment is a carcinogenic substance in categories 1 through 3 of the International Agency for Research on Cancer (IARC).
- the agent is a therapeutic agent.
- the invention provides a means of exploring the effects of a therapeutic agent on cancer progression (Fig. 12).
- the effects of a therapeutic agent on cell metastasis potential are evaluated.
- the effects of a therapeutic agent on a soft tumor are evaluated.
- the effects of a therapeutic agent on a hard tumor are evaluated.
- the effect of a therapeutic agent on primary and/or secondary tumor growth is evaluated.
- the therapeutic agent or agents administered in-vivo to an animal transplanted with OCT4 or OCT4 hl neoplastic stem cells comprise: interferons, interleukins, colony-stimulating, alkylating agents, nitrosoureas, antimetabolites, antitumor antibiotics, plant (vinca) alkaloids, steroid hormones or combinations thereof.
- the therapeutic agent is a chemotherapy agent.
- the chemotherapy agent is non-specific and therefore has the potential to kill a cancerous cell during any phase of the cell-cycle.
- the chemotherapy agent is specific and thus is able to kill cancerous cells during a specific cell cycle phase.
- the in-vivo effect of PDT on an animal transplanted with OCT4 hl neoplastic stem cells is evaluated.
- the in-vivo effects of radiation therapy or radiofrequency ablation alone or in combination with any other form of a therapeutic agent in-vivo are further assessed.
- the in-vivo effects of chemoembolization or local hyperthermia, on cancer progression and/or regression are evaluated.
- biological therapies comprise immunotherapy.
- immunotherapy comprises the use of a vaccine comprising immunogenic fragments derived from Nanog, STAT3, OCT4, or combinations thereof, as described hereinabove.
- the effect of nonspecific immunomodulating agent or agents is assessed.
- the nonspecific immunomodulating agent is bacillus Calmette-Gueriri (BCG) or levamisole.
- OCT4 modifiers are screened in-vivo for cancer progression or regression in an animal transplanted with OCT4 hl neoplastic stem cells (Fig. 12).
- OCT4 modifiers are screened in-vivo for cancer progression or regression in an animal transplanted with OCT4 hl neoplastic stem cells (Fig. 12).
- OCT4 monoclonal antibodies are screened in-vivo.
- OCT4 protein are screened in-vivo.
- PNAs, aptamers, or antisense siRNA are further evaluated in-vivo as shown in Fig. 4.
- the invention provides a method of preventing, treating, abrogating, or inhibiting cancer, tumor growth, cell metastasis or combinations thereof comprising the step of contacting neoplastic cells with an agent that inhibits OCT4 expression or function.
- OCT4 is inhibited transiently.
- OCT4 is inhibited constitutively.
- the invention provides a method of inhibiting OCT4 comprising targeting OCT4 expression at the DNA level and thus inhibiting or abrogating OCT4 transcription.
- inhibition of OCT4 at the DNA level is accomplished via the formation of DNA triple-stranded structures.
- the triple helix inhibition complex is designed as an
- OCT4 gene-specific oligonucleotide and thus inhibits OCT4 transcription.
- the invention provides a method of inhibiting OCT4 comprising targeting OCT4 expression at the RNA level and thus inhibiting OCT4 expression.
- RNA is mRNA.
- antisense based therapeutics are used to inhibit
- synthetic oligonucleotides are designed to be complementary in sequence to a specific OCT4 mRNA sequence and thus inhibit OCT4 expression.
- the invention provides peptide nucleic acids (PNAs) that are artificially constructed to hybridize to an OCT4 mRNA sequence and thus inhibit OCT4 expression.
- the binding agent is a specifically engineered ribozyme, which cleaves OCT4 mRNA transcripts and subsequently inhibits OCT4 expression.
- the method of inhibiting OCT4 function comprises targeting OCT4 protein.
- inhibition of OCT4 function is achieved through the specific binding of an antibody to an OCT4 protein and thus inhibiting or abrogating OCT4 protein binding to an OCT4 responsive DNA element.
- intrabody or antibodies raised subsequent to OCT4 immunotherapy inhibit or abrogate OCT4 function.
- the invention provides an intrabody specific to OCT4 protein.
- intrabodies comprise a single chain of a coupled variable domain of the heavy chain to the variable domain of the light chain through a peptide linker and are used to interfere with the binding of the OCT4 protein to an OCT4 DNA responsive element.
- intrabodies are directed to the cell nucleus where they inhibit or abrogate binding of an OCT4 protein to an OCT4
- the intrabodies target the OCT4 protein DNA binding domain on the OCT4 protein and hence, inhibit or abrogate OCT4 protein binding to an OCT4 DNA responsive element.
- the invention provides a vaccine comprising an OCT4 peptide.
- the OCT4 peptide elevates OCT4 specific antibodies.
- the peptide consists of the full length OCT4 gene.
- the peptide is a mutated form of OCT4.
- a 4-18 amino acid long OCT4 peptide is used.
- the vaccine comprises OCT4 peptides of uniform length and sequence.
- the vaccine comprises a mixture of OCT4 peptides that differ in both length and sequence.
- oligonucleotide aptamers are used to bind specific OCT4 protein sequence and thus inhibit or abrogate OCT4 protein binding to an OCT4 DNA responsive element.
- the invention provides a mutated OCT4 protein.
- the mutated OCT4 protein is used to block the transcription of downstream OCT4 responsive genes.
- the mutated OCT4 protein used to inhibit or abrogate OCT4 responsive gene expression and the wild type OCT4 protein have similar affinities to the OCT4 DNA responsive element.
- the mutated OCT4 protein used to inhibit or abrogate OCT4 responsive genes expression have a higher affinity to the OCT4 DNA responsive element compared to the wild type OCT4 protein.
- Immunohistochemical identification of OCT4, STAT3 or Nanog positive cells in tissue [00112] Immunohistochemical staining for OCT4, STAT3 or Nanog histological analyses of tumor biopsies from human and from mice were preformed as follows: Formalin fixed paraffin embedded tissue sections (5 ⁇ m) were sequentially deparaffinized, rehydrated and blocked for endogenous peroxidase activity following a 95° C degree, 25 minute antigen retrieval in Trilogy unmasking solution (Cell Marque, Hot Springs AR).
- Tissues were cut on a cryostat at 16 ⁇ m to generate sets that are in the axial plane (breast and lung) and coronal plane (brain).
- Hematoxylin-eosin (H&E) staining was performed on one set, and immunohistochemistry on a second set. The immunohistochemistry was performed using the following procedures.
- the frozen breast, lung, and brain sections were (1) incubated in 2% non-fat milk and 0.3% Triton-X in PBS for 1 hour; (2) incubated in OCT4, STAT3 or Nanog antibodies in 3% donkey serum and 0.1 % Triton-X overnight at room temperature; (3) washed with PBS for 3 times; (4) incubated with secondary antibody for 4 hours in dark at room temperature; (5) washed with PBS for 3 times; and (6) dehydrated through graded ethanol, cleared with xylene, and coverslipped with DPX mounting medium (44581 , FIuka Biochemika). Immunoreactivity was visualized with a Bio-Rad confocal microscope and images collected on a computer for later analysis.
- the OCT4-EGFP and; Nanog- EGFP constructs were engineered using strategies and techniques previously described (Gerrard et al., 2005).
- a plasmid containing the EGFP reporter (pEGFPl , BD Biosciences) and the selectable marker G418 under the control of the OCT4 and Nanog promoter was used.
- the promoter fragment of human OCT4 spans from base -3917 to base +55 of the OCT4 gene (hOCT4pr, from 67539 to 71490 in the human DNA sequence), and contains two appropriate regulatory elements which drove developmentally specific EGFP expression.
- the promoter fragment of human Nanog spans from base -132 to base +300 (from base 697969 to base 701269 in the human genomic DNA sequence of Chromosome 12).
- Oct4hP-eGFP plasmid was constructed using the human Oct-4 responsive promoter (Oct4hP, from 67539 to 71490 in human DNA sequence with accession number AP000509)_that was amplified by polymerase chain reaction with primers Oct4hP-F (5'-TT CCC ATG TCA AGT AAG TGG GGT GG-3') and Oct4hP-R (5'-CGA GAA GGC AAA ATC TGA AGC CAG G-3') using human genomic DNA (Promega G3041) as a template. The fragment was cloned into a TOPO vector (Invitrogen) and the fidelity of the DNA sequence was confirmed with bi-directional DNA sequencing.
- TOPO vector Invitrogen
- Oct4hP was then cloned into the expression vector pEGFPl (Clontech Cat # 6086-1, Genbank Accession # U55761) by insertion into the Hind ⁇ l and BamH l sites upstream of eGFP ( Figure 12A). Modification of the neurosphere culture system for isolation and analyses of NSCs from glioblastoma, bone sarcomas, and breast cancer
- the neurosphere culture system proposed by Weiss and Raynolds (1992) was modified by inhibiting the potential of cells for substrate attachment by exploiting pleiotropical growth factors - EGF, FGF2 and insulin in semi-solid methylcellulose (MC).
- MC semi-solid methylcellulose
- NSCs were transfected with EGFP reporter plasmid having the EFGP and G418 genes under the human promoter of OCT4 as shown in Fig. 7.
- the lesions and foci were dissected from their respective tissue locations using a Leica MZl 6FA dissecting microscope with a GFP3 filter (for fluorescence capability) and Q-imaging Retiga EXi monochrome digital camera with RGB filter for in-vitro studies and molecular analysis. This tumor material was further studied by using different assays.
- a Superscript II RNase H + Reverse Transcriptase first-strand synthesis system (InVitrogen Life Technologies, Carlsbad, CA) was used to synthesize the cDNA, from 1.5 ⁇ g of total RNA by priming with Oligo(dT)i 2 -i8 (Invitrogen Life Technologies, Carlsbad, CA).
- the target cDNA was amplified by using Platinum TaqDNA Polymerase (InVitrogen Life Technologies, Carlsbad, CA) and 37 cycles of PCR.
- Primers for human beta tubulin III amplified 1356-1497 bp product transcribed from the non-translated 3' UTR-region of the Hbeta4 gene as described in (Kavallaris et al., 1997).
- the primers for human OCT3/4 (Accession # Zl 1898), Nanog (NM 024865), Stat3 (NM 139276), Gata-4 (NM 002052), -6 (NM 005257), AFP (NM 001134), Runx 1 (NM 001754), were originally generated by using the Oligo5.1 program. All primers are provided in Table 1. Table 1.
- Cells were dissolved in lysing buffer containing 50 mM Tris-HCl, 150 mM NaCl, ImM EDTA, 1% NP40, 0.1% SDS, 1 %Na-deoxycholate, ImM Na-vanadate, and protease inhibitors: 5 ⁇ g/ml pepstatin, ImM phenylmethylsulphonylfluoride, 10 ⁇ g/ml leupeptin, ImM NaF ( Sigma Chemical Co., St. Louis, MO) for at least 1 hour on ice.
- protease inhibitors 5 ⁇ g/ml pepstatin, ImM phenylmethylsulphonylfluoride, 10 ⁇ g/ml leupeptin, ImM NaF ( Sigma Chemical Co., St. Louis, MO) for at least 1 hour on ice.
- the protein concentration of the supernatant was measured by BCA Protein Assay kit (Pierce, Rockford, IL) using Benchmark Microplate Reader (Bio Rad Laboratories, Hercules, CA USA). Lysates were mixed (1:1) with Laemmli Buffer (Sigma Chemical Co., St. Louis, MO). 15 ⁇ g of protein was loaded per lane of 8- 16% or 10-20% Tris-HCl Ready Gels (Bio Rad Laboratories, Hercules, CA) and separated by electrophoresis. The nitrocellulose membranes (Sigma Chemical Co., St.
- TBS Tris-Buffered Saline
- Tween-20 Bio-Rad Laboratories, Hercules, CA USA.
- the blots were incubated with secondary peroxidase- conjugated goat antibodies to mouse or rabbit IgG (Cell Signaling Technology, IL) or rabbit antibodies to goat IgG (Jackson Immuno Research Laboratories, West Grove, PA). Immunoreactive bands were detected by ECL+ Western Blotting Detection Reagents (Amersham Biosciences.UK) for 60 seconds or more and exposed to X-ray films.
- a whole suspension was plated at a density of 60,000 cells/2ml/well in DMEM/FI2 with 0.8% of MC, supplemented with progesterone (2OnM), putrescine (100 ⁇ M), sodium selenite (3OnM), transferrin (25 ⁇ g/ml), insulin (20 ⁇ g/ml) (Sigma Chemical Co., St. Louis, MO USA) and the growth factors EGF (10ng/ml) and recombinant FGF2 (lOng/ml).
- progesterone 2OnM
- putrescine 100 ⁇ M
- sodium selenite 3OnM
- transferrin 25 ⁇ g/ml
- insulin 20 ⁇ g/ml
- G418 was added after 3 days of culturing (200mg/ml). In plates with G418, only green EGFP-positive clones were generated and collected for further manipulations. The generated EGFP- positive mammasphere were used for establishing EGFP- subpopulation with stable integration of EGFP.
- the green mammasphere expressing EGFP under the control of the Oct4 promoter grew as un-attached suspended mammasphere (Fig. 7B). EGFP cells were isolated directly under fluorescent microscope (Fig. 1 ID).
- OCT4, Nanog and STAT3 are expressed in concert in glioblastomas clinical specimens and cell lines [00122]
- OCT4, Nanog and STAT3 are co-expressed in glioblastomas clinical specimens and cell lines LNl 8, LN229, LN428 and U251 semi-quantitative RT-PCR analysis followed by western blot analysis was performed.
- the results as indicated in Fig 2A show a moderate to high mRNA expression of OCT4, Nanog and STAT3.
- the protein expression levels were in correlation with the mRNA expression levels as shown in Fig. 2B, wherein, moderate to high protein expression levels of OCT4, Nanog and STAT3 are exhibited.
- OCT4 role in clone generation from glioma-derived tumor stem cells cultured in a neurosphere system To test the functional role of OCT4 gene in maintenance of self-renewal in a model culture system, siRNA silencing of POU5F1/OCT4 gene was assessed (Fig. 4). Toward this end, tumor cells derived from three representatives of glioblastoma cell types, including two cell lines and a primary tumor culture isolated from a patient, were co-transfected with EGFP and OCT4 siRNA, and plated in a neurosphere culture system. In a set of parallel control experiments, cells were co-transfected with EGFP and scrambled control siRNA.
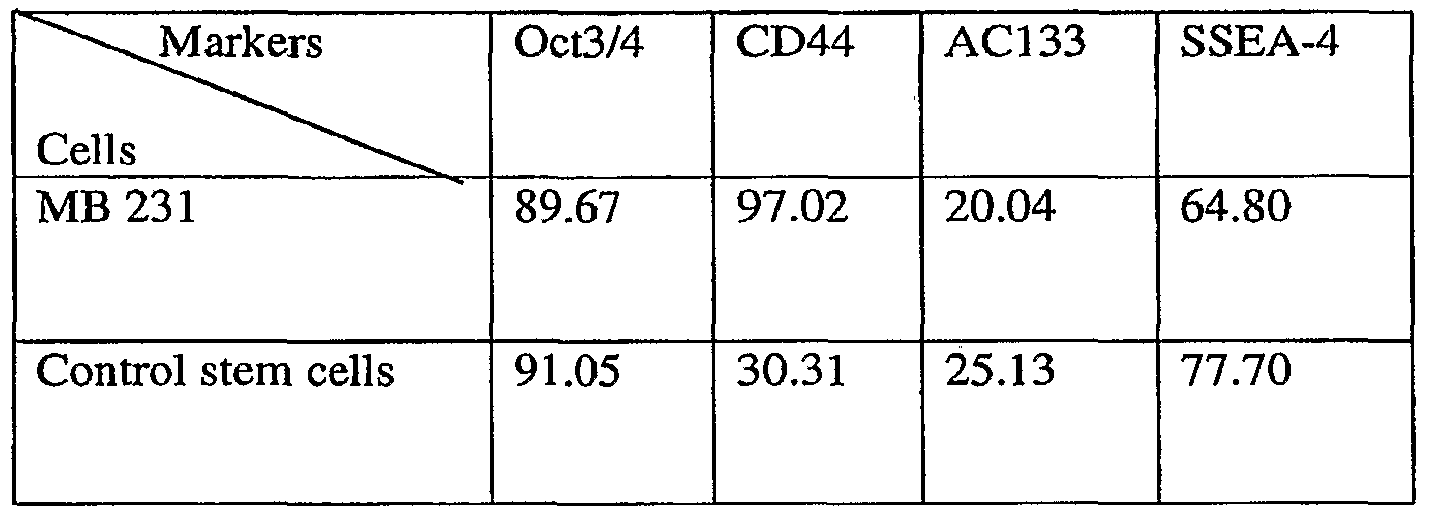
- Example 5 Tagging MDA MB 231 breast cancer cell line with EGFP to asses NSCs involvement in orthotopic tumor formation
- neoplastic stem cells derived from breast cancer cell line-MDA MB 231 stably expressing EGFP under Oct4 promoter in nude mice was assessed through NSCs involvement in orthotopic tumor formation in nude mice. This assay is based on the presumption that EGFP expression correlates with endogenous OCT4 gene expression.
- OCT4 positive and OCT4 negative MDA MB 231 breast cancer cells transfected with OCT4-EGFR construct and sorted by FACS were inoculated into fat pad of twelve nude mice which were grouped in four groups each group comprising 3 animal: Group 1 was inoculated with 5,000 OCT4 positive cells/animal, Group 2 was inoculated with 50,000 OCT4 positive cells/animal, Group 3 was inoculated with 500,000 OCT4 positive cells/animal, Group 4 was inoculated with 500,000 OCT4 negative cells/animal.
- Fluorescence biomarker driven by the Oct3/4 promoter is strong and specific for cancer stem cells
- Mammosphere cultures were derived from an MDA-MB-435 melanoma cell line and biomarked for the presence of cancer stem cells expressing Oct-3/4.
- MDA-MB-435 cells were stably transfected with Oct4hP-eGFP and CMV-mRFP. The cells were then FACS sorted for GFP expressing cells to create a highly pure cancer stem cell population for further studies.
- Fluorescent micrograph of suspended tumor-derived spheres shown in Figure 13B demonstrated that the fluorescence biomarker driven by the Oct3/4 promoter is strong and specific for cancer stem cells.
- fluorescent micrograph of the attached tumor spheres shown ( Figure 13D) exhibited a similar phenomenon.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Immunology (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- Cell Biology (AREA)
- Medicinal Chemistry (AREA)
- Urology & Nephrology (AREA)
- General Engineering & Computer Science (AREA)
- Molecular Biology (AREA)
- Hematology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- General Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Public Health (AREA)
- Food Science & Technology (AREA)
- Hospice & Palliative Care (AREA)
- Developmental Biology & Embryology (AREA)
- Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Pathology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP07777402A EP2032694A4 (en) | 2006-06-06 | 2007-06-05 | Compositions enriched in neoplastic stem cells and methods comprising same |
| AU2007258744A AU2007258744A1 (en) | 2006-06-06 | 2007-06-05 | Compositions enriched in neoplastic stem cells and methods comprising same |
| CA002658003A CA2658003A1 (en) | 2006-06-06 | 2007-06-05 | Compositions enriched in neoplastic stem cells and methods comprising same |
| JP2009514328A JP2009539374A (en) | 2006-06-06 | 2007-06-05 | Composition enriched with neoplastic stem cells and method comprising the same |
| MX2008015492A MX2008015492A (en) | 2006-06-06 | 2007-06-05 | Compositions enriched in neoplastic stem cells and methods comprising same. |
| IL195727A IL195727A0 (en) | 2006-06-06 | 2008-12-04 | Compositions enriched in neoplastic stem cells and methods comprising same |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US81109506P | 2006-06-06 | 2006-06-06 | |
| US60/811,095 | 2006-06-06 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007145901A1 true WO2007145901A1 (en) | 2007-12-21 |
Family
ID=38832061
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2007/013167 WO2007145901A1 (en) | 2006-06-06 | 2007-06-05 | Compositions enriched in neoplastic stem cells and methods comprising same |
Country Status (9)
| Country | Link |
|---|---|
| US (1) | US20070292414A1 (en) |
| EP (1) | EP2032694A4 (en) |
| JP (1) | JP2009539374A (en) |
| CN (1) | CN101501187A (en) |
| AU (1) | AU2007258744A1 (en) |
| CA (1) | CA2658003A1 (en) |
| IL (1) | IL195727A0 (en) |
| MX (1) | MX2008015492A (en) |
| WO (1) | WO2007145901A1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014028274A1 (en) * | 2012-08-15 | 2014-02-20 | California Stem Cell, Inc. | Rapid method production high purity cancer stem cells and population of high purity cancer stem cells |
| US10018630B2 (en) | 2011-09-07 | 2018-07-10 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell isolation |
| CN109705210A (en) * | 2018-12-25 | 2019-05-03 | 深圳大学 | OCT4 epitope peptide, antigen conjugates and its preparation method and application |
| US10934351B2 (en) | 2011-10-28 | 2021-03-02 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell-specific molecule |
| US11124773B2 (en) | 2010-10-06 | 2021-09-21 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell population and method for production thereof |
| US11536713B2 (en) | 2009-12-25 | 2022-12-27 | Chugai Seiyaku Kabushiki Kaisha | Method for searching and screening for target of anti-cancer agent using non-human animal model having NOG established cancer cell line transplanted therein |
Families Citing this family (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009009020A1 (en) | 2007-07-12 | 2009-01-15 | Discgenics | Human disc tissue |
| WO2009009739A2 (en) * | 2007-07-12 | 2009-01-15 | Theracrine, Inc. | Methods and compositions for facilitating cell death of cancer stem cells and for treating cancer |
| WO2009151503A2 (en) * | 2008-04-10 | 2009-12-17 | University Of Florida Research Foundation, Inc. | Compositions and methods for the treatment of a neoplasia |
| WO2009140260A2 (en) * | 2008-05-13 | 2009-11-19 | University Of Pittsburgh - Of The Commonwealth System Of Higher Education | Cancer stem cell immortalization |
| CN102344490B (en) * | 2010-07-30 | 2014-03-05 | 中国科学院上海生命科学研究院 | Anti-mouse Stk40 polyclonal antibody and preparation thereof |
| WO2012019061A2 (en) | 2010-08-05 | 2012-02-09 | Stem Centrx, Inc. | Novel effectors and methods of use |
| MX2013002255A (en) | 2010-08-27 | 2013-07-03 | Stem Centrx Inc | Notum protein modulators and methods of use. |
| WO2013119964A2 (en) | 2012-02-08 | 2013-08-15 | Stem Centrx, Inc. | Identification and enrichment of cell subpopulations |
| US9945842B2 (en) | 2010-09-03 | 2018-04-17 | Abbvie Stemcentrx Llc | Identification and enrichment of cell subpopulations |
| WO2012031273A2 (en) | 2010-09-03 | 2012-03-08 | Stem Centrx, Inc. | Novel modulators and methods of use |
| RU2016123839A (en) | 2010-12-08 | 2018-11-30 | АббВай Стемсентркс ЭлЭлСи | New modulators and methods of their application |
| SA112330278B1 (en) | 2011-02-18 | 2015-10-09 | ستيم سينتركس، انك. | Novel modulators and methods of use |
| US20130058947A1 (en) | 2011-09-02 | 2013-03-07 | Stem Centrx, Inc | Novel Modulators and Methods of Use |
| RU2014138474A (en) | 2012-02-24 | 2016-04-10 | СтемСентРкс, Инк. | NEW MODULATORS AND APPLICATION METHODS |
| MX2014010094A (en) | 2012-02-24 | 2014-09-16 | Stem Centrx Inc | Anti sez6 antibodies and methods of use. |
| SG11201506650PA (en) | 2013-02-22 | 2015-09-29 | Stemcentrx Inc | Novel antibody conjugates and uses thereof |
| CA2921187A1 (en) * | 2013-08-12 | 2015-02-19 | Minerva Biotechnologies Corporation | Method for enhancing tumor growth |
| MX2016002547A (en) | 2013-08-28 | 2016-06-17 | Stemcentrx Inc | Site-specific antibody conjugation methods and compositions. |
| JP2016538318A (en) | 2013-08-28 | 2016-12-08 | ステムセントリックス, インコーポレイテッド | New SEZ6 modulator and method of use |
| MX2016005763A (en) | 2013-11-06 | 2016-08-19 | Stemcentrx Inc | Novel anti-claudin antibodies and methods of use. |
| AU2014361856A1 (en) | 2013-12-12 | 2016-06-30 | Abbvie Stemcentrx Llc | Novel anti-DPEP3 antibodies and methods of use |
| US10308721B2 (en) | 2014-02-21 | 2019-06-04 | Abbvie Stemcentrx Llc | Anti-DLL3 antibodies and drug conjugates for use in melanoma |
| US9777070B2 (en) | 2014-04-30 | 2017-10-03 | Pfizer Inc | Anti-PTK7 antibody-drug conjugates |
| TW201617368A (en) | 2014-09-05 | 2016-05-16 | 史坦森特瑞斯公司 | Novel anti-MFI2 antibodies and methods of use |
| CN106148267A (en) * | 2015-04-04 | 2016-11-23 | 于荣敏 | The induction of the stem cell in scape cambium layer source, Changchun and separation method |
| JP2018135268A (en) * | 2015-06-05 | 2018-08-30 | 大日本住友製薬株式会社 | Novel heteroaryl amino-3-pyrazole derivative and pharmacologically acceptable salt thereof |
| AU2017254674A1 (en) | 2016-04-21 | 2018-11-01 | Abbvie Stemcentrx Llc | Novel anti-BMPR1B antibodies and methods of use |
| CN111621466B (en) * | 2020-06-09 | 2020-12-29 | 首都医科大学附属北京朝阳医院 | Preparation method and application of pulmonary artery tissue single cell suspension |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050196858A1 (en) * | 1993-04-21 | 2005-09-08 | The University Of Edinburgh | Isolation, selection and propagation of animal transgenic stem cells |
| US20060019256A1 (en) * | 2003-06-09 | 2006-01-26 | The Regents Of The University Of Michigan | Compositions and methods for treating and diagnosing cancer |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5283179A (en) * | 1990-09-10 | 1994-02-01 | Promega Corporation | Luciferase assay method |
| US6271131B1 (en) * | 1998-08-26 | 2001-08-07 | Micron Technology, Inc. | Methods for forming rhodium-containing layers such as platinum-rhodium barrier layers |
| US20020164836A1 (en) * | 2001-05-07 | 2002-11-07 | Advanced Semiconductor Engineering Inc. | Method of manufacturing printed circuit board |
| WO2005085423A1 (en) * | 2004-02-27 | 2005-09-15 | Michigan State University | Oct-4 and gjic expression as markers for adult human stem cells and cancer cell precursors |
| US7604947B2 (en) * | 2004-06-09 | 2009-10-20 | Cornell Research Foundation, Inc. | Detection and modulation of cancer stem cells |
-
2007
- 2007-06-05 JP JP2009514328A patent/JP2009539374A/en active Pending
- 2007-06-05 WO PCT/US2007/013167 patent/WO2007145901A1/en active Application Filing
- 2007-06-05 EP EP07777402A patent/EP2032694A4/en not_active Withdrawn
- 2007-06-05 US US11/806,993 patent/US20070292414A1/en not_active Abandoned
- 2007-06-05 AU AU2007258744A patent/AU2007258744A1/en not_active Abandoned
- 2007-06-05 MX MX2008015492A patent/MX2008015492A/en not_active Application Discontinuation
- 2007-06-05 CN CNA2007800292012A patent/CN101501187A/en active Pending
- 2007-06-05 CA CA002658003A patent/CA2658003A1/en not_active Abandoned
-
2008
- 2008-12-04 IL IL195727A patent/IL195727A0/en unknown
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050196858A1 (en) * | 1993-04-21 | 2005-09-08 | The University Of Edinburgh | Isolation, selection and propagation of animal transgenic stem cells |
| US20060019256A1 (en) * | 2003-06-09 | 2006-01-26 | The Regents Of The University Of Michigan | Compositions and methods for treating and diagnosing cancer |
Non-Patent Citations (3)
| Title |
|---|
| CHENG: "Establishing a germ cell origin for metastatic tumors using OCT4 immunohistochemistry", CANCER, vol. 101, August 2004 (2004-08-01), pages 2006 - 2010, XP008100647 * |
| GIBBS ET AL.: "Stem-like cells in bone sarcomas: implications for tumorigenesis", NEOPLASIA, vol. 7, November 2005 (2005-11-01), pages 967 - 976, XP008100649 * |
| See also references of EP2032694A4 * |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11536713B2 (en) | 2009-12-25 | 2022-12-27 | Chugai Seiyaku Kabushiki Kaisha | Method for searching and screening for target of anti-cancer agent using non-human animal model having NOG established cancer cell line transplanted therein |
| US11124773B2 (en) | 2010-10-06 | 2021-09-21 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell population and method for production thereof |
| US11965180B2 (en) | 2010-10-06 | 2024-04-23 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell population and method for production thereof |
| US10018630B2 (en) | 2011-09-07 | 2018-07-10 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell isolation |
| US10934351B2 (en) | 2011-10-28 | 2021-03-02 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell-specific molecule |
| US11858987B2 (en) | 2011-10-28 | 2024-01-02 | Chugai Seiyaku Kabushiki Kaisha | Cancer stem cell-specific molecule |
| WO2014028274A1 (en) * | 2012-08-15 | 2014-02-20 | California Stem Cell, Inc. | Rapid method production high purity cancer stem cells and population of high purity cancer stem cells |
| GB2519717A (en) * | 2012-08-15 | 2015-04-29 | California Stem Cell Inc | Rapid method production high purity cancer stem cells and population of high purity cancer stem cells |
| CN109705210A (en) * | 2018-12-25 | 2019-05-03 | 深圳大学 | OCT4 epitope peptide, antigen conjugates and its preparation method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| IL195727A0 (en) | 2011-08-01 |
| EP2032694A1 (en) | 2009-03-11 |
| US20070292414A1 (en) | 2007-12-20 |
| AU2007258744A1 (en) | 2007-12-21 |
| CN101501187A (en) | 2009-08-05 |
| JP2009539374A (en) | 2009-11-19 |
| MX2008015492A (en) | 2009-02-12 |
| EP2032694A4 (en) | 2010-09-15 |
| CA2658003A1 (en) | 2007-12-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20070292414A1 (en) | Compositions enriched in neoplastic stem cells and methods comprising same | |
| US20200385686A1 (en) | Cancer stem cell population and method for production thereof | |
| CA2417909C (en) | Isolation and use of solid tumor stem cells | |
| EP2529223B1 (en) | Biomarkers for circulating tumor cells | |
| US8435746B2 (en) | Aldehyde dehydrogenase 1 (ALDH1) as a cancer stem cell marker | |
| US7563619B2 (en) | Mammary stem cell marker | |
| JP2006512924A (en) | Brain endothelial cell expression pattern | |
| US8741635B2 (en) | Method of cell isolation | |
| US20110111434A1 (en) | Colon stem cells associated with colitisand colorectal cancer and methods of use | |
| KR20170041192A (en) | Identification of cancer stem cell markers and use of same for diagnosis and treatment | |
| WO2009061837A1 (en) | Neoplastic stem cell system and method | |
| CA2568806C (en) | Methods for detecting and treating cancer using podocalyxin and/or endoglycan | |
| CA2607859A1 (en) | Murine stem cells and applications thereof | |
| US20050064420A1 (en) | Methods for identifying, isolating, and controlling the growth of estrogen-responsive cells | |
| CN107208066A (en) | Δ 133P53 β and Δ 133P53 γ isotypes are the biomarkers of cancer stem cell | |
| Lee et al. | Thymic epithelial tumour progression in an SV40T transgenic mouse model: Cortical thymoma–thymic carcinoma sequence | |
| CN113249476B (en) | Key protein BLT2 influencing tumor cell dryness and application thereof | |
| CN110229896B (en) | Application of STEAP1 in diagnosis, treatment and prognosis of ovarian cancer | |
| Li et al. | Evidence for Mesenchymal2Epithelial Transition Associated with Mouse Hepatic Stem Cell |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780029201.2 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07777402 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 2658003 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 195727 Country of ref document: IL Ref document number: 2009514328 Country of ref document: JP Ref document number: MX/A/2008/015492 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007258744 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007777402 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2007258744 Country of ref document: AU Date of ref document: 20070605 Kind code of ref document: A |