WO2007137333A1 - Impedance measurements - Google Patents
Impedance measurements Download PDFInfo
- Publication number
- WO2007137333A1 WO2007137333A1 PCT/AU2007/000726 AU2007000726W WO2007137333A1 WO 2007137333 A1 WO2007137333 A1 WO 2007137333A1 AU 2007000726 W AU2007000726 W AU 2007000726W WO 2007137333 A1 WO2007137333 A1 WO 2007137333A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- khz
- frequencies
- impedance
- determining
- design
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/05—Detecting, measuring or recording for diagnosis by means of electric currents or magnetic fields; Measuring using microwaves or radio waves
- A61B5/053—Measuring electrical impedance or conductance of a portion of the body
- A61B5/0537—Measuring body composition by impedance, e.g. tissue hydration or fat content
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/05—Detecting, measuring or recording for diagnosis by means of electric currents or magnetic fields; Measuring using microwaves or radio waves
- A61B5/053—Measuring electrical impedance or conductance of a portion of the body
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B5/00—Measuring for diagnostic purposes; Identification of persons
- A61B5/48—Other medical applications
- A61B5/4869—Determining body composition
Definitions
- the present invention relates to a method and apparatus for determining frequencies for use in performing impedance measurements on a subject, as well as to a method and apparatus for performing impedance measurements.
- Bioelectrical Impedance Analysis measures the impedance to flow of an alternating electrical current passed through biological tissue. Such impedance measurements are typically performed at a number of different frequencies, allowing a subject's impedance response to be modelled, using a mathematic relationship, such as the Cole model. This in turn enables the estimation of various parameters, which can in turn be used to derive information regarding a subject's health.
- the frequencies used are typically selected randomly, or based on the ability of the measuring device used to perform the measurements. Consequently the effectiveness and accuracy of the measurements varies greatly.
- the present invention provides a method of determining frequencies for use in performing impedance measurements, the method including: a) determining estimates for parameter values representing an impedance response for at least one subject; b) using the estimated parameter values to determine a design; and, c) using the design to determine frequencies for use in impedance measurements.
- the method includes, determining the estimates for the parameter values at least in part using a model of the subject's impedance response.
- the method includes: a) determining a residual variance model having a predetermined distribution; and, b) determining the estimates for the parameter values at least in part using the selecting variance model.
- the method of determining the residual variance model includes using an expectation maximization algorithm.
- the method includes determining a range of parameter values representing the impedance response of a number of subjects.
- the method includes: a) determining a Fisher information matrix for a model of the impedance response of the at least one subject; and, b) determining a design using the Fisher information matrix.
- the model includes at least one of: a) A Cole model; and, b) A residual variance model.
- the model includes determining the design by optimising the determinant of the Fisher information matrix.
- the design is a D-optimal design.
- the method includes determining a design space to take into account practical limitations.
- the method includes, modifying the design by at least one of: a) restricting the frequencies; and, b) providing an error structure as a function of frequency.
- the method is performed at least in part using a processing system.
- the method includes, in the processing system: a) determining impedance data representing the impedance response of the at least one subject; and, b) using the impedance data to determine the estimates for the parameter values.
- the method includes, in the processing system: a) determining an impedance model; and, b) using the impedance model and the estimated parameter values to determine the design.
- the method includes, in the processing system: a) calculating a Fisher information matrix for the model; and, b) using the Fisher information matrix to determine the design.
- the method includes, in the processing system, optimising a determinant of the Fisher information matrix for the model.
- the method includes, in the processing system: a) receiving limits on applicable frequencies; and, b) using the limits and the design to determine the frequencies.
- the method includes, in the processing system, determining frequencies for use in impedance measurements within the range : a) 0-5O kHz; b) 2-20O kHz; c) 50-500 kHz; and, d) 200-100O kHz.
- the frequencies are within the ranges: a) 0-24 IcHz; b) 5-74 kHz; c) 77-20O kHz; - A -
- the frequencies are: a) 14 kHz b) 57 kHz c) 188 kHz d) 679 kHz
- the frequencies are: a) 14.1844 kHz b) 56.920I kHz c) 187.5397 kHz d) 679.114I kHz
- the present invention provides apparatus for determining frequencies for use in performing impedance measurements, the apparatus including, a processing system for: a) determining estimates for parameter values representing an impedance response for at least one subject; b) using the estimated parameter values to determine a design; and, c) using the design to determine frequencies for use in impedance measurements.
- the apparatus is for performing the first broad form of the invention.
- the present invention provides a method of measuring the impedance of a subject the method including, the method including, in a measuring device: a) causing one or more electrical signals to be applied to the subject using a first set of electrodes, the one or more electrical signals having four frequencies; b) measuring electrical signals across a second set of electrodes applied to the subject in response to the applied one or more signals; c) determining from the applied signals and the measured signals at least one measured impedance value at each of the four frequencies, wherein the four frequencies are in the ranges: i) 0-5O kHz; ii) 2-20O kHz; iii) 50-500 kHz; and, iv) 200-1000 kHz.
- a restricted range of frequencies can be defined by the ranges: a) 0-24 kHz; b) 5-74 kHz; c) 77-20O kHz; d) 530-100O kHz.
- the frequencies are approximately: a) 14 kHz b) 57 kHz c) 188 kHz d) 679 kHz
- the frequencies are: a) 14.1844 kHz b) 56.920I kHz c) 187.5397 kHz d) 679.114I kHz
- the method includes determining one or more impedance parameter values based on the plurality of measured impedance values.
- the present invention provides apparatus for measuring the impedance of a subject the apparatus including a measuring device for: a) causing one or more electrical signals to be applied to the subject using a first set of electrodes, the one or more electrical signals having four frequencies; b) measuring electrical signals across a second set of electrodes applied to the subject in response to the applied one or more signals; c) determining from the applied signals and the measured signals at least one measured impedance value at each of the four frequencies, wherein the four frequencies are in the ranges: i) 0-5O kHz; ii) 2-20O kHz; iii) 50-500 kHz; and, iv) 200-100O kHz.
- the measuring device includes: a) a current source for applying current signals to the subject; b) a voltage sensor for measuring voltages signals across the subject; and, c) a processing system could to the current source and the voltage sensor for: i) causing the current signals to be applied to the subject; and, ii) determining the impedance using the applied current signals and measured voltage signals.
- the apparatus is for performing the method of the third broad form of the invention.
- Figure 1 is a flow chart of an example of a process for determining frequencies for use in performing impedance measurements
- Figure 2 is a schematic diagram of an example of a processing system for use in determining frequencies
- Figure 3 is a flow chart of a specific example of a process for determining frequencies for use in performing impedance measurements
- Figure 4 is a schematic diagram of an example of an "idealised" equivalent circuit for the
- Figure 5 is a schematic diagram of an example of apparatus for measuring impedance
- Figure 6 is an example of a residual plot for determining an initial model fit
- Figure 7 is an example of a resistance versus reactance plot for example impedance measurements
- Figure 8 is an example of plots of a variance function for various values of ⁇ 2 ; and, Figure 9 is an example of a plot of residuals from a model fit using an example extracted data set.
- parameter values are determined representing the impedance response of one or more subjects.
- the parameter values may be determined in any one of a number of manners, such by modelling impedance data collected from a number of subjects from a prior study, as will be described in more detail below.
- the parameter values are used to determine an optimal design for studying the impedance response of the one or more subjects. This can be achieved, for example, by optimising designs representing frequencies that may be used in performing impedance measurements according to optimality criteria.
- the optimal designs may optionally be modified to take into account practical effects, such as the variants of subjects within a population to impedance measurement.
- the optimal designs are used to determine frequencies that may be used for performing impedance measurements.
- the process may be performed manually, but typically requires advanced computation and therefore typically requires the use of a processing system.
- the processing system 200 is formed from a processor 210, a memory 211, an input/output device 212 and an optional external interface 213 interconnected via a bus 214.
- the external interface 213 may be used to couple the processing system 200 to a database 220.
- the processing system 200 executes applications software stored in the memory 211, to allow parts of the process to be performed, as will be described in more detail below.
- the processing system 200 may be any suitable form of processing system 200, such as a personal computer, desktop, laptop, super computer, Sparc station, or the like.
- impedance data is collected from a sample population of subjects.
- the data is collected using a suitable measuring device that applies alternating electrical signals to a subject, and measures the electrical response of the subject. This is typically achieved by applying alternating currents to the subject at a number of different frequencies, and then measures voltage signals across the subject, to allow the impedance to be determined at each frequency.
- a suitable measuring device that applies alternating electrical signals to a subject, and measures the electrical response of the subject. This is typically achieved by applying alternating currents to the subject at a number of different frequencies, and then measures voltage signals across the subject, to allow the impedance to be determined at each frequency.
- An example measuring device will be described in more detail below.
- the impedance response of the subject can be modelled using a suitable model, such as the Cole model, which is based on an equivalent circuit that effectively models the electrical behaviour of biological tissue, an example of which is shown in Figure 4.
- a suitable model such as the Cole model, which is based on an equivalent circuit that effectively models the electrical behaviour of biological tissue, an example of which is shown in Figure 4.
- the equivalent circuit includes two branches representing current flow through extracellular fluid and intracellular fluid.
- the extracellular component of biological impedance is represented by a resistance R e
- the intracellular component is represented by a resistance Rj and a capacitance C.
- equation (A) can be modified to include an exponent ⁇ to account for the distribution of time constants observed in biological tissues (or systems), as follows:
- R ⁇ ,R e ,C and a are parameters in the model
- Z is the predicted impedance
- R 1 and R e are the resistances due to the intracellular and extracellular fluids in the body, respectively.
- a residual variance model is selected to allow variations from the predicted response to be taken into account.
- the residual variance model may be selected in any one of ways as will be described in more detail below.
- parameter estimates are determined that represent the measured responses of the subjects.
- the initial parameters are parameters which when inserted into the models provide an estimate of the response of the sample population.
- a number of Cole models are constructed across the parameter space, using the range of parameter values. This effectively models the range of different impedance responses of the subjects within the population.
- a Fisher information matrix is determined for the residual and Cole models. This can be achieved in any one of a number of ways, but typically involves using suitable applications software, such as the software package POPT by S. B. Duffull, implemented by the processing system 200.
- the product determinant of the Fisher information matrices is optimised. Again, this may be achieved in any one of a number of manners, such as by using a simulated annealing approach to find maxima for the expression. This provides an optimised design which represents the theoretical preferred frequencies at which impedance measurements may be made.
- the optimal design can be used to determine preferred practical frequencies at which impedance measurements may be performed, together with an indication of the relative efficiency of the measurement procedure.
- the above-described process operates by utilising impedance measurements for a sample population to determine optimal design, which can in turn be used to determine preferred frequencies for performing impedance measurements.
- the process is used to determine that in general at least four frequencies should be used for performing impedance measurements. This can also be used to determine preferred frequency ranges for the four frequencies.
- the preferred frequency ranges are as follows: • 0-50 kHz;
- the range of frequencies can be further limited to specific values as follows:
- Determination of the frequencies allows an impedance measuring device to be provided, which is adapted to utilise the preferred frequencies, as shown for example in Figure 5.
- the impedance measuring device typically includes a processing system 1 coupled to a current source 11 and a voltage sensor 12.
- the current source 11 is coupled via electrodes 13, 14 to a subject S with the voltage sensor being coupled to the subject S via electrodes 15, 16.
- the processing system 1 causes the current source 11 to apply alternating current signals to the subject S via the electrodes 13, 14, at each of the preferred frequencies determined using the above described process.
- the response of subject S is then measured via the electrodes 15, 16 using the voltage sensor 12.
- the processing system 1 can then use details of the applied current and the measured voltage signals to determine an impedance value at each of the preferred frequencies.
- parameters of the Cole model By utilising the four measured frequencies, this allows parameters of the Cole model to be determined, such as the subject's impedance at a characteristic frequency Z 0 , as well as values for the intracellular and extracellular impedance. This in turn allows information such as indicators of the subject's of intracellular or extracellular fluid, or the ratio there between, to be determined.
- the Biology 8 110-122 is used to model bioimpedance.
- the model includes nonlinear parameters, which are known to vary between individuals. Accordingly the process uses the theory of experimental design to find the frequencies at which measurements of bioimpedance are optimally made. This can also be used to determine how many frequencies are needed in each patient in order to obtain good estimates of the parameters.
- a Design ⁇ is defined by: X 1 ⁇ — X 2 W 2
- W 1 is the experimental effort given to the i -th vector of covariates X 1 .
- the expected Fisher information matrix can be defined as:
- a design ⁇ is D-optimal if it maximizes the determinant of the expected fisher information matrix. That is, arg max ⁇ ⁇ which,
- the D-efficiency of the design ⁇ compared to the D-optimal design ⁇ * for a particular model is:
- a D-efficiency of 0.5 for ⁇ compared with ⁇ * means that twice as many samples need to be taken using ⁇ in order to obtain as accurate parameter estimates as given by ⁇ *.
- M x ⁇ ⁇ , ⁇ ) is the expected Fisher information matrix for model 1 with parameters ⁇ x and P 1 is the number of model parameters. Similarly for model 2.
- Nonlinear Regression A nonlinear model is specified by two main parts; a function expressing the predicted response and residual response variance structure. This, for the jth observation, can be written as follows:
- X j are the covariates
- ⁇ refers to the model parameters
- S j is error or uncertainty in the response for a given individual.
- One relaxation is a generalization of the assumption of constant variance. Such a relaxation will allow flexibility in specifying intra-individual variance. This involves specifying a variance function h which can depend upon the predicted response EIyJ, covariates X j and/or additional parameters ⁇ . The model is then specified by:
- W is a diagonalized nx 1 vector of weights and is a row vector of derivatives of d ⁇ ,. the model with respect to the /th parameter evaluated at the covariates in ⁇ .
- Q 1 is the p- vector of individual parameters and ⁇ , is the n t -vector of random errors.
- ⁇ is normally distributed with zero mean and a diagonal variance matrix characterized by parameters s and t so that s relates to the additive error component while t relates to the multiplicative error component.
- ⁇ is defined by ⁇ +b t
- ⁇ is the vector of fixed effects
- b is the vector of random effects for individual /.
- b t is normally distributed with mean zero and variance ⁇ .
- the covariates for individual / is denoted by the vector X 1 , and Q 1 is defined by g( ⁇ , B ⁇ x 1 ), where g is a known function.
- l( ⁇ ;y) is the log-likelihood of observations y for the population parameters ⁇ . Due to the nonlinearity of/ with respect to ⁇ , there is no analytical expression for l ⁇ ;y). That is, in general, for say N individuals where Y is a matrix of responses for the whole population and y t is the vector of responses for the /th individual, the likelihood can be expressed as:
- the Fisher information matrices are determined using the software package POPT written by S. B. Duffull.
- the search routine uses an adapted version of a simulated annealing algorithm for continuous variables. Together these techniques provide a means of finding D-optimal designs for nonlinear mixed and fixed effects models across a continuous search space.
- Modelling was performed using the software package MONOLIX, which is a MATLAB based package that uses stochastic approximation to the expectation maximization (SAEM) algorithm, in parametric maximum likelihood estimation for nonlinear mixed effects models.
- SAEM expectation maximization
- the Expectation-Maximization (EM) algorithm is an iterative procedure used for likelihood function maximization given incomplete data sets formed from observable and non observable data.
- the E-step in this algorithm cannot be performed in closed formed due to the nonlinearity in/of the random effects, as described above.
- the SAEM algorithm replaces this step with a stochastic procedure.
- the SAEM replaces this step by drawing ⁇ , w from the conditional distribution p(. ⁇ y, ⁇ k ) and updates ⁇ 2 A ( ⁇ ) as follows:
- ⁇ k is a decreasing sequence of positive numbers.
- initial parameter estimates are also needed.
- initial estimates of fixed effects were taken from previous studies, and estimates of variance were found by what is called a 'Standard Two Stage Approach'. This involves estimating fixed effects for all data specific to each individual. Then, the variance of these estimates is calculated and used as an estimate for ⁇ .
- the first stage of the modeling process was to find a suitable residual variance model such that the residuals are normally distributed around zero so that tests, such as the likelihood ratio test, are valid.
- the first run uses a diagonal variance- covariance matrix with all four parameters having a normally distributed random component and the model having additive residuals.
- the residual plot is shown in Figure 6. This shows that the assumption about additive variance holds reasonably well and the residuals are centered around zero. Whilst the plot is not completely random at low frequencies, due for example to technical errors of measurements and inaccuracies associated with using the Cole equation to model bioelectrical impedance, this is considered acceptable.
- This additive variance model can then be compared with the multiplicative and additive plus multiplicative error models.
- the residual plot was best when the additive error model was used.
- the initial model can then be compared with different models having various fixed effects parameters. For example, the initial model can be compared with a model setting the parameter c to have no random component to it, that is having a fixed effect only.
- the likelihood ratio test for nested models can then be used to compare these models with the initial run. In this instance it was found that the initial run significantly increased the log- likelihood to justify having all parameters in the model.
- the final step of the modeling process is to allow some or all of the random effects to have a log-normal distribution. This shows that found parameter estimates were not biologically plausible and can therefore be disregarded.
- the initial model chosen to start the modeling process was the one chosen based on the above reasoning. It was also the model that provided the maximum value of the log- likelihood of (y; ⁇ ) .
- CV% coefficient of variation
- C(E, V) is set to a block matrix of zeros. Then, under this assumption, the fixed and random effects of the model are independent.
- the D-efficiency of the fixed effect design under the mixed effects model is 0.9692.
- Such a high efficiency of the fixed effects D-optimal design suggests that, in this case, accounting for between subject variance only marginally improves the ability to estimate the parameters of the Cole equation.
- An assessment of the preferred model can be made by adding a variance function into the model that mimics the variance of bioimpedance in real life studies.
- the D-optimal design will provide the best estimates of the parameters. Inherent in this design will be the ideal range to restrict our frequencies, based on the variance function used.
- Bioimpedance is made up of two parts, resistance and reactance, relating to the real and imaginary parts of the Cole equation, respectively.
- An example plot of resistance vs reactance is shown in Figure 7. This plot is of real data and as such shows the real life deviations from the theoretical semi-circle that would be produced from the pure circuit seen in Figure 4.
- the peak of the locus of the semi-circle identifies the characteristic frequency f c .
- This frequency yields the smallest variance due to the variance function and consequently, it is assumed that at this frequency the constant additive variance dominates.
- the variance ftinction needs to be such that there is relatively small variance between the chosen interval, for example [4, 750], with rapidly increasing variance for frequencies outside these bounds.
- a double exponential model centred around f c , can be used to adequately model the variance of such a study.
- frequencies between f c and 1000 are rescaled to be between 0 and 1.
- frequencies between 0 and f c are rescaled so that they are between 1 and 0.
- the new vector of rescaled frequencies is then exponentiated twice, and scaled such that the vector has a minimum and a maximum at 0 and 1, respectively.
- slightly extra variability is added to frequencies greater than f c by multiplying the rescaled numbers which relate to these frequencies by 2. This whole vector is then multiplied by ⁇ e extra
- Example residual plots of this variance function h( ⁇ ) for various ⁇ alra 2 values are shown in Figure 8.
- the log of frequency is shown in all four plots.
- Table 3 shows D-optimal designs found for various values of ⁇ a(ra 2 .
- the D-efficiencies shown refer to the efficiency of each design compared with the D-optimal design described above.
- the model considered is from equation 16.
- a broad set of preferred frequency ranges can be defined as follows:
- a product design is generally more robust to changes in initial parameter estimates than local D-optimal designs for each parameter set. Thus, a product design is preferred if uncertainty exists in the initial parameter estimates.
- the product design is formed by optimizing over the product of the determinants of the respective variance-covariance matrices of the eight models as a single function.
- the idea behind forming product designs in this way is that optimizing across all eight models will provide a design which is efficient across a wide range of parameter estimates.
- the efficiency of the product design can be calculated by comparing its D-value under each model compared to the D-optimal value under the respective model.
- Table 5 shows the efficiency of the product design compared to each D-optimal design for each parameter set. Relatively high efficiencies across all models suggest that the product design is a good means for allowing for uncertainty in initial parameter estimates. Further, given the range of parameter values considered, it is believed that these designs will efficiently estimate model parameters for a variety of individuals. Hence, this approach and the frequencies found should benefit bioimpedance analysis.
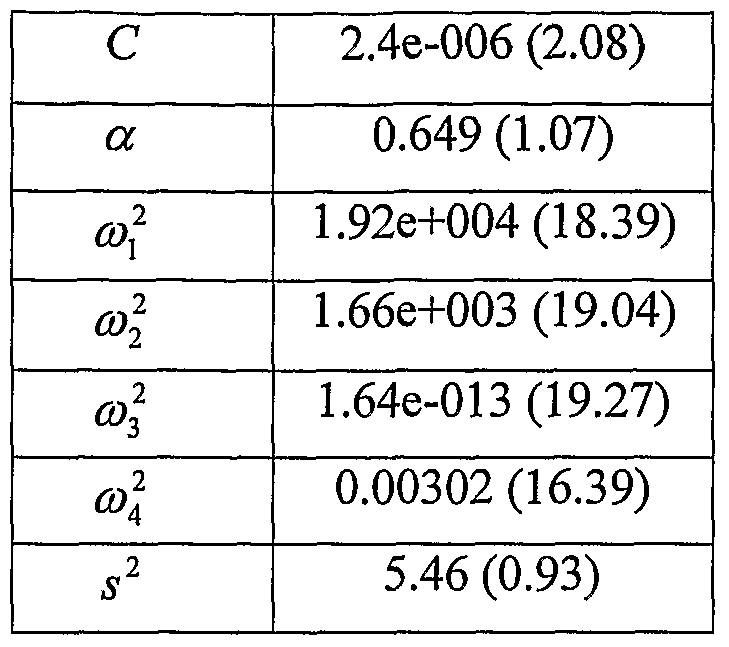
- MONOLIX provides estimates for the parameter values as shown in table 6 and estimates of coefficients of variation found in table 7.
- Figure 9 shows the residual plot for this fit, indicating that the nonlinear regression assumptions discussed above hold.
- Table 7 also shows the expected coefficients of variation calculated by using the expected Fisher information matrix to form the expected standard errors of estimates.
- D-optimal designs can also be applied to real data where it was shown to perform well against the full data set. It is hoped that the D-optimal design approach used here and the designs found will aid BIA researchers in the design of optimised multiple frequency BIA instrumentation. This will mitigate the acknowledged inadequacy of some currently used instrumentation and provide for better clinical utility, for example, the accurate prediction of lean body mass for drug dosing in the obese.
- Persons skilled in the art will appreciate that numerous variations and modifications will become apparent. All such variations and modifications which become apparent to persons skilled in the art, should be considered to fall within the spirit and scope that the invention broadly appearing before described.
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009512367A JP5431147B2 (en) | 2006-05-30 | 2007-05-25 | Impedance measurement |
| EP20070718972 EP2020918B1 (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
| US12/302,914 US8761870B2 (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
| ES07718972.8T ES2545730T3 (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
| CA2653406A CA2653406C (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
| AU2007266311A AU2007266311B2 (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2006902907A AU2006902907A0 (en) | 2006-05-30 | Impedance Measurements | |
| AU2006902907 | 2006-05-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007137333A1 true WO2007137333A1 (en) | 2007-12-06 |
Family
ID=38777994
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/AU2007/000726 WO2007137333A1 (en) | 2006-05-30 | 2007-05-25 | Impedance measurements |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US8761870B2 (en) |
| EP (1) | EP2020918B1 (en) |
| JP (1) | JP5431147B2 (en) |
| AU (1) | AU2007266311B2 (en) |
| CA (1) | CA2653406C (en) |
| ES (1) | ES2545730T3 (en) |
| WO (1) | WO2007137333A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105184062A (en) * | 2015-08-25 | 2015-12-23 | 中国人民解放军后勤工程学院 | User perception quality evaluation method based on confidence interval in crowd-sourcing perception network |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105534524A (en) * | 2016-02-05 | 2016-05-04 | 思澜科技(成都)有限公司 | Device and method for quickly recognizing parathyroid gland in thyroid surgery |
| JP2019509153A (en) * | 2016-02-16 | 2019-04-04 | インペディメッド・リミテッド | Heart failure indicator |
| CN108852352B (en) * | 2018-03-26 | 2021-07-30 | 深圳市麦德安医疗设备有限公司 | Parameter estimation method of bio-impedance Cole equation, intelligent device and storage medium |
| EP3856024A4 (en) * | 2018-09-27 | 2022-06-22 | Impedimed Limited | Evaluating impedance measurements |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5280429A (en) * | 1991-04-30 | 1994-01-18 | Xitron Technologies | Method and apparatus for displaying multi-frequency bio-impedance |
| WO2000040955A1 (en) * | 1999-01-05 | 2000-07-13 | Kaiku Limited | Impedance measurements of bodily matter |
| US6125297A (en) * | 1998-02-06 | 2000-09-26 | The United States Of America As Represented By The United States National Aeronautics And Space Administration | Body fluids monitor |
| WO2001067098A1 (en) | 2000-03-03 | 2001-09-13 | Btg International Limited | Electrical impedance measuring method for differentiating tissue types |
| US20040015095A1 (en) * | 2002-07-16 | 2004-01-22 | Jianhua Li | Method and device for measuring signals for electrical impedance tomography by using correlation techinique |
| WO2004047635A1 (en) * | 2002-11-22 | 2004-06-10 | Impedimed Pty Ltd | Multifrequency bioimpedance determination |
| WO2005027717A2 (en) * | 2003-09-12 | 2005-03-31 | Renal Research Institute, Llc | Bioimpedance methods and apparatus |
| WO2007014417A1 (en) * | 2005-08-02 | 2007-02-08 | Impedimed Limited | Impedance parameter values |
Family Cites Families (324)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR1344459A (en) | 1962-10-18 | 1963-11-29 | Method and apparatus for the electrical study of living organisms | |
| USRE30101E (en) | 1964-08-19 | 1979-09-25 | Regents Of The University Of Minnesota | Impedance plethysmograph |
| US4314563A (en) | 1970-09-24 | 1982-02-09 | The United States Of America As Represented By The Administrator Of The Veterans Administration | Apparatus for measuring relative changes in blood volume in a portion of an animal body to detect a venous occlusion |
| US3834374A (en) | 1972-04-06 | 1974-09-10 | M Ensanian | Diagnostic electrical scanning |
| US3871359A (en) | 1973-06-25 | 1975-03-18 | Interscience Technology Corp | Impedance measuring system |
| US3851641A (en) | 1973-11-29 | 1974-12-03 | J Toole | Method and apparatus for determining internal impedance of animal body part |
| US3996924A (en) | 1974-06-19 | 1976-12-14 | Wheeler H Brownell | Occlusive impedance phlebograph and method therefor |
| US4008712A (en) | 1975-11-14 | 1977-02-22 | J. M. Richards Laboratories | Method for monitoring body characteristics |
| US4034854A (en) | 1976-07-16 | 1977-07-12 | M I Systems, Inc. | Electrode package |
| US4184486A (en) | 1977-08-11 | 1980-01-22 | Radelkis Elektrokemiai Muszergyarto Szovetkezet | Diagnostic method and sensor device for detecting lesions in body tissues |
| US4458694A (en) | 1977-11-02 | 1984-07-10 | Yeda Research & Development Co., Ltd. | Apparatus and method for detection of tumors in tissue |
| IL53286A (en) | 1977-11-02 | 1980-01-31 | Yeda Res & Dev | Apparatus and method for detection of tumors in tissue |
| DE2912349A1 (en) | 1979-03-29 | 1980-10-16 | Liebisch Geb | Detector system for human skin moisture content - has scanning head with two contact electrodes attached to skin under specified pressure |
| US4365634A (en) | 1979-12-06 | 1982-12-28 | C. R. Bard, Inc. | Medical electrode construction |
| US4353372A (en) | 1980-02-11 | 1982-10-12 | Bunker Ramo Corporation | Medical cable set and electrode therefor |
| FR2486386A1 (en) | 1980-07-09 | 1982-01-15 | Argamakoff Alexis | Thermographic and impedance measurer for cancer scanning - has single electrode or matrix supplying temp. and impedance signals |
| US4407300A (en) | 1980-07-14 | 1983-10-04 | Davis Robert E | Potentiometric diagnosis of cancer in vivo |
| JPS5772627A (en) | 1980-10-21 | 1982-05-07 | Tokyo Shibaura Electric Co | Apparatus for detecting abnormal cell |
| US4401356A (en) | 1980-11-24 | 1983-08-30 | C. R. Bard, Inc. | Electrical terminal |
| US4942880A (en) | 1981-01-28 | 1990-07-24 | Ceske Vysoke Uceni Technicke V Praze | Method for non-invasive electric diagnosis and therapy in hemodialysis and general medicine |
| US4407288B1 (en) | 1981-02-18 | 2000-09-19 | Mieczyslaw Mirowski | Implantable heart stimulator and stimulation method |
| IL62861A (en) | 1981-05-13 | 1988-01-31 | Yeda Res & Dev | Method and apparatus for carrying out electric tomography |
| CA1196691A (en) | 1982-01-12 | 1985-11-12 | Bradley Fry | Reconstruction system and methods for impedance imaging |
| SE455043B (en) | 1982-04-22 | 1988-06-20 | Karolinska Inst | DEVICE FOR MONITORING THE LIQUID BALANCE OF THE HUMAN BODY BY MEASURING THE IMPEDANCE OF THE BODY |
| US4617939A (en) | 1982-04-30 | 1986-10-21 | The University Of Sheffield | Tomography |
| US4450527A (en) | 1982-06-29 | 1984-05-22 | Bomed Medical Mfg. Ltd. | Noninvasive continuous cardiac output monitor |
| GB2126732B (en) | 1982-09-02 | 1986-01-15 | British Telecomm | Impedance measurement in 4-wire to 2-wire converters |
| GB2131558B (en) | 1982-11-05 | 1986-03-05 | Walter Farrer | Measuring potential difference |
| SU1132911A1 (en) | 1983-02-18 | 1985-01-07 | Muzhikov Valerij G | Method of diagnosis of vascular insufficiency of lower extremities |
| US4557271A (en) | 1983-05-11 | 1985-12-10 | Stoller Kenneth P | Method and apparatus for detecting body illness, dysfunction, disease and/or pathology |
| FR2557318A1 (en) | 1983-12-26 | 1985-06-28 | A2F | ELECTRONIC DEVICE PROVIDING A UNIVERSAL INTERFACE BETWEEN SENSORS AND AN APPARATUS FOR INPUT AND SIGNAL PROCESSING FROM SUCH SENSORS |
| US4583549A (en) | 1984-05-30 | 1986-04-22 | Samir Manoli | ECG electrode pad |
| US4646754A (en) | 1985-02-19 | 1987-03-03 | Seale Joseph B | Non-invasive determination of mechanical characteristics in the body |
| US4688580A (en) | 1985-07-11 | 1987-08-25 | The Johns Hopkins University | Non-invasive electromagnetic technique for monitoring bone healing and bone fracture localization |
| US4686477A (en) | 1985-09-30 | 1987-08-11 | Mobil Oil Corporation | Multiple frequency electric excitation method and identifying complex lithologies of subsurface formations |
| US4763660A (en) | 1985-12-10 | 1988-08-16 | Cherne Industries, Inc. | Flexible and disposable electrode belt device |
| WO1988007392A1 (en) | 1987-03-26 | 1988-10-06 | Intrinsic Ag | Signal control process, electric treatment device and electrode system |
| US4832608A (en) | 1987-05-22 | 1989-05-23 | Cherne Medical, Inc. | Electrode belt adapter |
| CN1024161C (en) | 1987-09-05 | 1994-04-13 | 哈尔滨工业大学 | Intellectual faculties type free respiration resistant tachogram picturing instrument |
| US4911175A (en) | 1987-09-17 | 1990-03-27 | Diana Twyman | Method for measuring total body cell mass and total extracellular mass by bioelectrical resistance and reactance |
| US4924875A (en) | 1987-10-09 | 1990-05-15 | Biometrak Corporation | Cardiac biopotential analysis system and method |
| US5078134A (en) | 1988-04-25 | 1992-01-07 | Lifecor, Inc. | Portable device for sensing cardiac function and automatically delivering electrical therapy |
| US4895163A (en) | 1988-05-24 | 1990-01-23 | Bio Analogics, Inc. | System for body impedance data acquisition |
| US4951682A (en) | 1988-06-22 | 1990-08-28 | The Cleveland Clinic Foundation | Continuous cardiac output by impedance measurements in the heart |
| US4890630A (en) | 1989-01-23 | 1990-01-02 | Cherne Medical, Inc. | Bio-electric noise cancellation system |
| US5511553A (en) | 1989-02-15 | 1996-04-30 | Segalowitz; Jacob | Device-system and method for monitoring multiple physiological parameters (MMPP) continuously and simultaneously |
| US4905705A (en) | 1989-03-03 | 1990-03-06 | Research Triangle Institute | Impedance cardiometer |
| IL91193A (en) | 1989-08-02 | 1996-01-19 | Yeda Res & Dev | Tumor detection system |
| US5086781A (en) | 1989-11-14 | 1992-02-11 | Bookspan Mark A | Bioelectric apparatus for monitoring body fluid compartments |
| GB9013177D0 (en) | 1990-06-13 | 1990-08-01 | Brown Brian H | Real-time imaging, etc. |
| US5063937A (en) | 1990-09-12 | 1991-11-12 | Wright State University | Multiple frequency bio-impedance measurement system |
| US5526808A (en) | 1990-10-04 | 1996-06-18 | Microcor, Inc. | Method and apparatus for noninvasively determining hematocrit |
| US5199432A (en) | 1990-10-30 | 1993-04-06 | American Home Products Corporation | Fetal electrode product for use in monitoring fetal heart rate |
| DE4100568A1 (en) | 1991-01-11 | 1992-07-16 | Fehling Guido | DEVICE FOR MONITORING A PATIENT FOR REPELLATION REACTIONS OF AN IMPLANTED ORGAN |
| US5101828A (en) | 1991-04-11 | 1992-04-07 | Rutgers, The State University Of Nj | Methods and apparatus for nonivasive monitoring of dynamic cardiac performance |
| US5197479A (en) | 1991-05-13 | 1993-03-30 | Mortara Instrument | Automatic electrode channel impedance measurement system for egg monitor |
| JPH05196681A (en) | 1991-06-26 | 1993-08-06 | Digital Equip Corp <Dec> | Method and apparatus for testing interconnection of continuously moving electric circuit |
| US5588429A (en) | 1991-07-09 | 1996-12-31 | Rensselaer Polytechnic Institute | Process for producing optimal current patterns for electrical impedance tomography |
| US5544662A (en) | 1991-07-09 | 1996-08-13 | Rensselaer Polytechnic Institute | High-speed electric tomography |
| GB9116215D0 (en) | 1991-07-26 | 1991-09-11 | Nat Res Dev | Electrical impedance tomography |
| US5423326A (en) | 1991-09-12 | 1995-06-13 | Drexel University | Apparatus and method for measuring cardiac output |
| US5309917A (en) | 1991-09-12 | 1994-05-10 | Drexel University | System and method of impedance cardiography and heartbeat determination |
| GB2260416B (en) | 1991-10-10 | 1995-07-26 | Smiths Industries Plc | Resistance monitors |
| DE4134960A1 (en) | 1991-10-23 | 1993-04-29 | Popp Fritz Albert Dr | METHOD FOR A HOLISTIC ANALYSIS OF THE HEALTH CONDITION |
| US5305192A (en) | 1991-11-01 | 1994-04-19 | Linear Technology Corporation | Switching regulator circuit using magnetic flux-sensing |
| US5415164A (en) | 1991-11-04 | 1995-05-16 | Biofield Corp. | Apparatus and method for screening and diagnosing trauma or disease in body tissues |
| US5906614A (en) | 1991-11-08 | 1999-05-25 | Ep Technologies, Inc. | Tissue heating and ablation systems and methods using predicted temperature for monitoring and control |
| US5282840A (en) | 1992-03-26 | 1994-02-01 | Medtronic, Inc. | Multiple frequency impedance measurement system |
| IL102300A (en) | 1992-06-24 | 1996-07-23 | N I Medical Ltd | Non-invasive system for determining of the main cardiorespiratory parameters of the human body |
| US5735284A (en) | 1992-06-24 | 1998-04-07 | N.I. Medical Ltd. | Method and system for non-invasive determination of the main cardiorespiratory parameters of the human body |
| US5372141A (en) | 1992-07-01 | 1994-12-13 | Body Composition Analyzers, Inc. | Body composition analyzer |
| GB9222888D0 (en) | 1992-10-30 | 1992-12-16 | British Tech Group | Tomography |
| WO1994010922A1 (en) | 1992-11-13 | 1994-05-26 | Ep Technologies, Inc. | Cardial ablation systems using temperature monitoring |
| US5557210A (en) | 1992-11-20 | 1996-09-17 | Pacesetter, Inc. | Universal cable connector for temporarily connecting implantable stimulation leads and implantable stimulation devices with a non-implantable system analyzer |
| GB9226376D0 (en) | 1992-12-18 | 1993-02-10 | British Tech Group | Tomography |
| GB9312327D0 (en) | 1993-06-15 | 1993-07-28 | British Tech Group | Laser ultrasound probe and ablator |
| US5454377A (en) | 1993-10-08 | 1995-10-03 | The Ohio State University | Method for measuring the myocardial electrical impedance spectrum |
| ZA948393B (en) | 1993-11-01 | 1995-06-26 | Polartechnics Ltd | Method and apparatus for tissue type recognition |
| WO1995024155A1 (en) | 1994-03-11 | 1995-09-14 | British Technology Group Limited | Electrical impedance tomography |
| RU2112416C1 (en) | 1994-05-10 | 1998-06-10 | Научно-исследовательский институт вычислительной техники | Method for checking of tissue or organ condition after operation and device for its realization |
| US5807270A (en) | 1994-06-20 | 1998-09-15 | Williams; Christopher Edward | Brain damage monitor |
| US5704355A (en) | 1994-07-01 | 1998-01-06 | Bridges; Jack E. | Non-invasive system for breast cancer detection |
| US5505209A (en) | 1994-07-07 | 1996-04-09 | Reining International, Ltd. | Impedance cardiograph apparatus and method |
| US5810742A (en) | 1994-10-24 | 1998-09-22 | Transcan Research & Development Co., Ltd. | Tissue characterization based on impedance images and on impedance measurements |
| WO1996012439A1 (en) | 1994-10-24 | 1996-05-02 | Transscan Research & Development Co. Ltd. | Impedance imaging devices and multi-element probe |
| US6560480B1 (en) | 1994-10-24 | 2003-05-06 | Transscan Medical Ltd. | Localization of anomalies in tissue and guidance of invasive tools based on impedance imaging |
| US5562607A (en) | 1995-01-18 | 1996-10-08 | Alza Corporation | Electrotransport device having reusable controller power saver |
| US5503157A (en) | 1995-03-17 | 1996-04-02 | Sramek; Bohumir | System for detection of electrical bioimpedance signals |
| DE19514698C1 (en) | 1995-04-13 | 1996-12-12 | Siemens Ag | Procedure for taking a distance measurement |
| US5557242A (en) | 1995-05-22 | 1996-09-17 | Motorola, Inc. | Method and apparatus for dielectric absorption compensation |
| US5919142A (en) | 1995-06-22 | 1999-07-06 | Btg International Limited | Electrical impedance tomography method and apparatus |
| NL1001282C2 (en) * | 1995-09-26 | 1997-03-28 | A J Van Liebergen Holding B V | Stroke volume determination device for a human heart. |
| US5813404A (en) | 1995-10-20 | 1998-09-29 | Aspect Medical Systems, Inc. | Electrode connector system |
| US5807272A (en) | 1995-10-31 | 1998-09-15 | Worcester Polytechnic Institute | Impedance spectroscopy system for ischemia monitoring and detection |
| GB9524968D0 (en) | 1995-12-06 | 1996-02-07 | Brown Brian H | Impedance pneumography |
| US5685316A (en) | 1996-04-08 | 1997-11-11 | Rheo-Graphic Pte Ltd. | Non-invasive monitoring of hemodynamic parameters using impedance cardiography |
| US6011992A (en) | 1996-05-09 | 2000-01-04 | Church Of Spirtual Technology | System for measuring and indicating changes in the resistance of a living body |
| EP0956089A4 (en) | 1996-05-10 | 2000-11-08 | Survivalink Corp | Defibrillator electrode circuitry |
| FR2748928A1 (en) | 1996-05-23 | 1997-11-28 | Jabourian Artin Pascal | Portable electronic cardiac rhythm detector |
| RU2138193C1 (en) | 1996-06-26 | 1999-09-27 | Государственное предприятие конструкторское бюро "СПЕЦВУЗАВТОМАТИКА" | Method of separation of electrical component of cardiac activity |
| US5732710A (en) | 1996-08-09 | 1998-03-31 | R.S. Medical Monitoring Ltd. | Method and device for stable impedance plethysmography |
| US5749369A (en) | 1996-08-09 | 1998-05-12 | R.S. Medical Monitoring Ltd. | Method and device for stable impedance plethysmography |
| US5759159A (en) | 1996-09-25 | 1998-06-02 | Ormco Corporation | Method and apparatus for apical detection with complex impedance measurement |
| CN1180513A (en) | 1996-10-23 | 1998-05-06 | 黄莹 | Cardiac function recording transmitter |
| RU2127075C1 (en) | 1996-12-11 | 1999-03-10 | Корженевский Александр Владимирович | Method for producing tomographic image of body and electrical-impedance tomographic scanner |
| US5876353A (en) | 1997-01-31 | 1999-03-02 | Medtronic, Inc. | Impedance monitor for discerning edema through evaluation of respiratory rate |
| US5957861A (en) | 1997-01-31 | 1999-09-28 | Medtronic, Inc. | Impedance monitor for discerning edema through evaluation of respiratory rate |
| ES2151774B1 (en) | 1997-03-06 | 2001-07-01 | Nte Sa | APPARATUS AND PROCEDURE FOR THE MEASUREMENT OF GLOBAL AND SEGMENTAL BODY VOLUMES AND COMPOSITION IN HUMAN BEINGS. |
| US6026323A (en) | 1997-03-20 | 2000-02-15 | Polartechnics Limited | Tissue diagnostic system |
| US6248083B1 (en) | 1997-03-25 | 2001-06-19 | Radi Medical Systems Ab | Device for pressure measurements |
| US5788643A (en) | 1997-04-22 | 1998-08-04 | Zymed Medical Instrumentation, Inc. | Process for monitoring patients with chronic congestive heart failure |
| FI972067A0 (en) | 1997-05-14 | 1997-05-14 | Tiit Koeoebi | Apparaturer ocffaranden Foer utvaendig maetning av physiologiska parametar |
| US5895298A (en) | 1997-05-29 | 1999-04-20 | Biofield Corp. | DC biopotential electrode connector and connector condition sensor |
| US6745070B2 (en) | 1997-10-03 | 2004-06-01 | Tasc Ltd. | High definition electrical impedance tomography |
| US6018677A (en) | 1997-11-25 | 2000-01-25 | Tectrix Fitness Equipment, Inc. | Heart rate monitor and method |
| US6006125A (en) | 1998-02-12 | 1999-12-21 | Unilead International Inc. | Universal electrocardiogram sensor positioning device and method |
| FR2775581B1 (en) | 1998-03-03 | 2000-05-05 | Seb Sa | APPARATUS AND METHOD FOR MEASURING BODY COMPOSITION |
| US6173003B1 (en) | 1998-03-26 | 2001-01-09 | Visteon Global Technologies, Inc. | Dither noise source with notched frequency spectrum |
| US6354996B1 (en) | 1998-04-15 | 2002-03-12 | Braun Gmbh | Body composition analyzer with trend display |
| US6122544A (en) | 1998-05-01 | 2000-09-19 | Organ; Leslie William | Electrical impedance method and apparatus for detecting and diagnosing diseases |
| CA2231038C (en) | 1998-05-05 | 2005-12-13 | Leslie W. Organ | Electrical impedance method and apparatus for detecting and diagnosing diseases |
| BR9911866A (en) | 1998-07-06 | 2002-01-29 | Aleksander Pastor | Apparatus for evaluating skin impedance variations |
| JP3778330B2 (en) | 1998-10-01 | 2006-05-24 | 株式会社デンソー | Health care equipment |
| US6845264B1 (en) | 1998-10-08 | 2005-01-18 | Victor Skladnev | Apparatus for recognizing tissue types |
| US6469732B1 (en) | 1998-11-06 | 2002-10-22 | Vtel Corporation | Acoustic source location using a microphone array |
| JP4025438B2 (en) | 1998-11-10 | 2007-12-19 | 積水化学工業株式会社 | Body composition estimation device |
| US6142949A (en) | 1998-11-24 | 2000-11-07 | Ortivus Ab | Lead protection and identification system |
| DE19857090A1 (en) | 1998-12-10 | 2000-06-29 | Stephan Boehm | Procedure for the regional determination of alveolar opening and closing of the lungs |
| US6317628B1 (en) | 1999-01-25 | 2001-11-13 | Cardiac Pacemakers, Inc. | Cardiac rhythm management system with painless defribillation lead impedance measurement |
| US6151520A (en) | 1999-01-26 | 2000-11-21 | Ge Medical Systems Information Technologies, Inc. | Connector for fetal probe |
| US6233473B1 (en) | 1999-02-16 | 2001-05-15 | Hologic, Inc. | Determining body composition using fan beam dual-energy x-ray absorptiometry |
| CN1236597A (en) | 1999-03-16 | 1999-12-01 | 秦大明 | Automatic analysis system for remote-measuring dynamic cardiac function and method for measuring dynamic cardiac kinetic energy. |
| US6497659B1 (en) | 1999-04-09 | 2002-12-24 | Spacelabs Medical, Inc. | System for identifying a cable transmitting a signal from a sensor to an electronic instrument |
| AUPQ113799A0 (en) | 1999-06-22 | 1999-07-15 | University Of Queensland, The | A method and device for measuring lymphoedema |
| US6512949B1 (en) | 1999-07-12 | 2003-01-28 | Medtronic, Inc. | Implantable medical device for measuring time varying physiologic conditions especially edema and for responding thereto |
| JP2001037735A (en) | 1999-07-27 | 2001-02-13 | Matsushita Electric Ind Co Ltd | Biological impedance measuring instrument |
| US6408204B1 (en) | 1999-07-28 | 2002-06-18 | Medrad, Inc. | Apparatuses and methods for extravasation detection |
| KR100333166B1 (en) | 1999-07-29 | 2002-04-18 | 차기철 | Useful Apparatus and Method for Analyzing Body Composition Based on Bioelectrical Impedance |
| JP3907353B2 (en) | 1999-08-26 | 2007-04-18 | 株式会社タニタ | Bioimpedance measurement device |
| JP2001070273A (en) | 1999-09-03 | 2001-03-21 | Tanita Corp | Method for measuring of biological electric impedance and body composition measuring device |
| EP1217942A1 (en) | 1999-09-24 | 2002-07-03 | Healthetech, Inc. | Physiological monitor and associated computation, display and communication unit |
| EP1092387B1 (en) | 1999-10-12 | 2004-07-21 | Tanita Corporation | Living body measuring apparatus |
| AU777114B2 (en) | 1999-10-12 | 2004-09-30 | Erich Sackmann | Highly time resolved impedance spectroscopy |
| JP2001187035A (en) | 1999-12-28 | 2001-07-10 | Tanita Corp | Lesion recovery degree judging device |
| JP4064028B2 (en) | 2000-01-05 | 2008-03-19 | 株式会社タニタ | Physical fatigue assessment device |
| US6292690B1 (en) | 2000-01-12 | 2001-09-18 | Measurement Specialities Inc. | Apparatus and method for measuring bioelectric impedance |
| JP2001198098A (en) | 2000-01-21 | 2001-07-24 | Tanita Corp | Dropsy measurement method and dropsy measurement device |
| JP2001212098A (en) | 2000-01-31 | 2001-08-07 | Tanita Corp | Equipment for measuring bioelectric impedance whose circuit is integrated into one chip |
| WO2001054578A1 (en) | 2000-01-31 | 2001-08-02 | Pearlman Justin D | Multivariate cardiac monitor |
| JP4454092B2 (en) | 2000-02-15 | 2010-04-21 | 大和製衡株式会社 | Body fat mass measuring device |
| US7499745B2 (en) | 2000-02-28 | 2009-03-03 | Barbara Ann Karmanos Cancer Institute | Multidimensional bioelectrical tissue analyzer |
| US6714814B2 (en) | 2000-03-30 | 2004-03-30 | Tanita Corporation | Bioelectrical impedance measuring apparatus |
| JP4401529B2 (en) | 2000-04-10 | 2010-01-20 | パナソニック株式会社 | Laminate voltage measuring device |
| AU2001251539A1 (en) | 2000-04-11 | 2001-10-23 | Cornell Research Foundation Inc. | System and method for three-dimensional image rendering and analysis |
| WO2001078577A2 (en) | 2000-04-17 | 2001-10-25 | Vivometrics, Inc. | Systems and methods for ambulatory monitoring of physiological signs |
| AU5202501A (en) | 2000-04-20 | 2001-11-07 | Univ Monash | Method and apparatus for determining conditions of biological tissues |
| JP3918559B2 (en) | 2000-04-21 | 2007-05-23 | オムロン株式会社 | Electrostatic relay and communication equipment using the relay |
| US6496721B1 (en) | 2000-04-28 | 2002-12-17 | Cardiac Pacemakers, Inc. | Automatic input impedance balancing for electrocardiogram (ECG) sensing applications |
| JP2001321352A (en) | 2000-05-16 | 2001-11-20 | Sekisui Chem Co Ltd | Electric characteristic measuring device |
| AU2001263239A1 (en) | 2000-05-18 | 2001-11-26 | Nuvasive, Inc. | Tissue discrimination and applications in medical procedures |
| US6928315B1 (en) | 2000-05-21 | 2005-08-09 | Mirabel Medical Systems Ltd. | Apparatus for impedance imaging coupled with another modality |
| IL163684A0 (en) | 2000-05-31 | 2005-12-18 | Given Imaging Ltd | Measurement of electrical characteristics of tissue |
| CA2411589C (en) | 2000-06-09 | 2009-06-02 | Bohm, Stephan | Method and apparatus for displaying information obtained by electrical impedance tomography data |
| JP3792489B2 (en) | 2000-06-30 | 2006-07-05 | 株式会社タニタ | Bioimpedance measurement device |
| US6569160B1 (en) | 2000-07-07 | 2003-05-27 | Biosense, Inc. | System and method for detecting electrode-tissue contact |
| US7149576B1 (en) | 2000-07-10 | 2006-12-12 | Cardiodynamics International Corporation | Apparatus and method for defibrillation of a living subject |
| US6602201B1 (en) | 2000-07-10 | 2003-08-05 | Cardiodynamics International Corporation | Apparatus and method for determining cardiac output in a living subject |
| US6636754B1 (en) | 2000-07-10 | 2003-10-21 | Cardiodynamics International Corporation | Apparatus and method for determining cardiac output in a living subject |
| JP2004512864A (en) | 2000-07-18 | 2004-04-30 | モトローラ・インコーポレイテッド | Wireless electrocardiograph system and method |
| JP3699640B2 (en) | 2000-08-01 | 2005-09-28 | 株式会社タニタ | Body water content state determination device by multi-frequency bioimpedance measurement |
| US7228170B2 (en) | 2000-08-14 | 2007-06-05 | Renal Research Institute, Llc | Device and method for monitoring and controlling physiologic parameters of a dialysis patient using segmental bioimpedance |
| US6615077B1 (en) | 2000-08-14 | 2003-09-02 | Renal Research Institute, Llc | Device and method for monitoring and controlling physiologic parameters of a dialysis patient using segmental bioimpedence |
| US7801598B2 (en) | 2000-08-14 | 2010-09-21 | Fresenius Medical Care Holdings, Inc. | Device and method for the determination of dry weight by continuous measurement of resistance and calculation of circumference in a body segment using segmental bioimpedance analysis |
| US6505079B1 (en) | 2000-09-13 | 2003-01-07 | Foster Bio Technology Corp. | Electrical stimulation of tissue for therapeutic and diagnostic purposes |
| CN100403979C (en) | 2000-11-29 | 2008-07-23 | 株式会社弗森 | Method and device for measuring body composition |
| AU2002221109A1 (en) | 2000-12-14 | 2002-06-24 | Art Haven 9 Co., Ltd. | Body impedance measuring instrument |
| JP3947651B2 (en) | 2000-12-28 | 2007-07-25 | 株式会社タニタ | Postpartum support device |
| US6768921B2 (en) | 2000-12-28 | 2004-07-27 | Z-Tech (Canada) Inc. | Electrical impedance method and apparatus for detecting and diagnosing diseases |
| DE10100569A1 (en) | 2001-01-09 | 2002-07-11 | Koninkl Philips Electronics Nv | Driver circuit for display device |
| US6561986B2 (en) | 2001-01-17 | 2003-05-13 | Cardiodynamics International Corporation | Method and apparatus for hemodynamic assessment including fiducial point detection |
| WO2002062214A1 (en) | 2001-02-05 | 2002-08-15 | Glucosens, Inc. | Methods of determining concentration of glucose in blood |
| ITBO20010110A1 (en) | 2001-03-01 | 2002-09-01 | Tre Esse Progettazione Biomedi | PROCEDURE AND IMPLANTABLE DEVICE FOR THE INTRA-PULMONARY MEASUREMENT OF PHYSICAL PROPERTIES OF THE PULMONARY FABRIC DEPENDENT ON ITS DENSIT |
| US6631292B1 (en) | 2001-03-23 | 2003-10-07 | Rjl Systems, Inc. | Bio-electrical impedance analyzer |
| US6931605B2 (en) | 2001-03-28 | 2005-08-16 | Council Of Scientific & Industrial Research | Simulated circuit layout for low voltage, low paper and high performance type II current conveyor |
| US6511438B2 (en) | 2001-04-03 | 2003-01-28 | Osypka Medical Gmbh | Apparatus and method for determining an approximation of the stroke volume and the cardiac output of the heart |
| US6807443B2 (en) | 2001-05-01 | 2004-10-19 | Cheetah Medical Inc. | High-resolution medical monitoring apparatus particularly useful for electrocardiographs |
| JP2002330938A (en) | 2001-05-10 | 2002-11-19 | Inax Corp | Toilet seat cover device with body fat meter |
| JPWO2002094096A1 (en) | 2001-05-22 | 2004-09-02 | バンブーカンパニー有限会社 | Diagnostic device for neuromusculoskeletal system and method of using the same |
| AUPR571801A0 (en) | 2001-06-15 | 2001-07-12 | Polartechnics Limited | Apparatus for tissue type recognition using multiple measurement techniques |
| US7044911B2 (en) | 2001-06-29 | 2006-05-16 | Philometron, Inc. | Gateway platform for biological monitoring and delivery of therapeutic compounds |
| US6870109B1 (en) | 2001-06-29 | 2005-03-22 | Cadwell Industries, Inc. | System and device for reducing signal interference in patient monitoring systems |
| US7783344B2 (en) | 2003-08-20 | 2010-08-24 | Philometron, Inc. | Hydration monitoring |
| US7933642B2 (en) | 2001-07-17 | 2011-04-26 | Rud Istvan | Wireless ECG system |
| US6625487B2 (en) | 2001-07-17 | 2003-09-23 | Koninklijke Philips Electronics N.V. | Bioelectrical impedance ECG measurement and defibrillator implementing same |
| JP3792547B2 (en) | 2001-07-19 | 2006-07-05 | 株式会社タニタ | Biometric device |
| US6595927B2 (en) | 2001-07-23 | 2003-07-22 | Medtronic, Inc. | Method and system for diagnosing and administering therapy of pulmonary congestion |
| WO2003009753A2 (en) | 2001-07-26 | 2003-02-06 | Chad Bouton | Detection of fluids in tissue |
| US7191000B2 (en) | 2001-07-31 | 2007-03-13 | Cardiac Pacemakers, Inc. | Cardiac rhythm management system for edema |
| US8777851B2 (en) | 2001-10-01 | 2014-07-15 | Medtronic, Inc. | Congestive heart failure monitor and ventilation measuring implant |
| US20050137480A1 (en) | 2001-10-01 | 2005-06-23 | Eckhard Alt | Remote control of implantable device through medical implant communication service band |
| DE10148440A1 (en) | 2001-10-01 | 2003-04-17 | Inflow Dynamics Inc | Implantable medical device for monitoring congestive heart failure comprises electrodes for measuring lung and heart tissue impedance, with an increase in impedance above a threshold value triggering an alarm |
| US6623312B2 (en) | 2001-10-04 | 2003-09-23 | Unilead International | Precordial electrocardiogram electrode connector |
| US20050101875A1 (en) | 2001-10-04 | 2005-05-12 | Right Corporation | Non-invasive body composition monitor, system and method |
| FR2830740B1 (en) | 2001-10-12 | 2004-07-23 | Seb Sa | BODY COMPOSITION MEASURING APPARATUS |
| JP3947379B2 (en) | 2001-10-12 | 2007-07-18 | 積水化学工業株式会社 | Electrical property measuring device |
| AU2002235772A1 (en) | 2001-12-12 | 2003-07-09 | Fresenius Medical Care Deutschland Gmbh | Determining the hydration status of a patient |
| US6829501B2 (en) | 2001-12-20 | 2004-12-07 | Ge Medical Systems Information Technologies, Inc. | Patient monitor and method with non-invasive cardiac output monitoring |
| US7184820B2 (en) | 2002-01-25 | 2007-02-27 | Subqiview, Inc. | Tissue monitoring system for intravascular infusion |
| JP3943955B2 (en) | 2002-02-25 | 2007-07-11 | 株式会社タニタ | Deep vein thrombosis determination device |
| JP3089347U (en) | 2002-04-17 | 2002-10-25 | 船井電機株式会社 | Remote control for TV with body fat measurement function |
| US6922586B2 (en) | 2002-05-20 | 2005-07-26 | Richard J. Davies | Method and system for detecting electrophysiological changes in pre-cancerous and cancerous tissue |
| US6780182B2 (en) | 2002-05-23 | 2004-08-24 | Adiana, Inc. | Catheter placement detection system and operator interface |
| AU2003238754A1 (en) | 2002-06-19 | 2004-01-06 | Brainz Instruments Limited | Artefact removal during eeg recordings |
| US7907998B2 (en) | 2002-07-03 | 2011-03-15 | Tel Aviv University Future Technology Development L.P. | Bio-impedance apparatus and method |
| US7096061B2 (en) | 2002-07-03 | 2006-08-22 | Tel-Aviv University Future Technology Development L.P. | Apparatus for monitoring CHF patients using bio-impedance technique |
| US20040019292A1 (en) | 2002-07-29 | 2004-01-29 | Drinan Darrel Dean | Method and apparatus for bioelectric impedance based identification of subjects |
| US7085598B2 (en) | 2002-08-23 | 2006-08-01 | Nihon Kohden Corporation | Biological electrode and connector for the same |
| WO2004026136A1 (en) | 2002-09-17 | 2004-04-01 | Beth Israel Deaconess Medical Center, Inc. | Radio frequency impedance mapping |
| AT413189B (en) * | 2002-10-07 | 2005-12-15 | Cnsystems Medizintechnik Gmbh | MEDICAL ELECTRODE ELEMENT |
| US7783345B2 (en) | 2002-10-07 | 2010-08-24 | Cnsystems Medizintechnik Gmbh | Impedance-based measuring method for hemodynamic parameters |
| AU2002951925A0 (en) | 2002-10-09 | 2002-10-24 | Queensland University Of Technology | An Impedence Cardiography Device |
| WO2004036379A2 (en) | 2002-10-17 | 2004-04-29 | The General Hospital Corporation | Arrangement and method for detecting abnormalities and inconsistencies in a body |
| US20080064981A1 (en) | 2002-11-07 | 2008-03-13 | Christopher Gregory | Method and apparatus for determining electrical properties of objects containing inhomogeneities |
| US7313434B2 (en) | 2002-11-25 | 2007-12-25 | Regents Of The University Of Minnesota | Impedance monitoring for detecting pulmonary edema and thoracic congestion |
| CA2451054A1 (en) | 2002-11-27 | 2004-05-27 | Z-Tech (Canada) Inc. | Improved apparatus and method for performing impedance measurements |
| AU2003286054A1 (en) | 2002-11-29 | 2004-06-23 | Z-Tech (Canada) Inc. | Improved breast electrode array and method of analysis for detecting and diagnosing diseases |
| NO20025803D0 (en) * | 2002-12-03 | 2002-12-03 | Idex Asa | Live finger |
| GB0228375D0 (en) | 2002-12-05 | 2003-01-08 | Innovation And Entpr Off Of | Wound mapping |
| EE04767B1 (en) | 2002-12-06 | 2007-02-15 | Tallinna Tehnika�likool | Method and apparatus for measuring electrical bio-impedance |
| EP1329190B1 (en) | 2002-12-14 | 2010-10-27 | Research Institute of Tsinghua University in Shenzhen | Apparatus and method for monitoring body composition by measuring body dielectric constant and body impedance based on digital frequency sampling |
| US20040167423A1 (en) | 2002-12-20 | 2004-08-26 | Luana Pillon | RXc graph and RXc Z-score graph methods |
| US6790185B1 (en) | 2002-12-31 | 2004-09-14 | Biopsy Sciences, Llc | Sealant plug delivery methods |
| ATE344637T1 (en) | 2003-01-09 | 2006-11-15 | Ge Healthcare Finland Oy | SHIELDING ARRANGEMENT FOR ECG CONNECTION WIRES |
| US7257244B2 (en) | 2003-02-24 | 2007-08-14 | Vanderbilt University | Elastography imaging modalities for characterizing properties of tissue |
| JP3907595B2 (en) | 2003-02-25 | 2007-04-18 | 株式会社タニタ | Vein extensibility evaluation index measuring device |
| JP2004255120A (en) | 2003-02-28 | 2004-09-16 | Tanita Corp | Estimating method and measuring device for body composition |
| WO2004082460A2 (en) | 2003-03-14 | 2004-09-30 | Shock, Llc | Methods of and apparatus for determining fluid volume presence in mammalian tissue |
| EP1517140A3 (en) | 2003-03-19 | 2005-04-06 | TF Instruments GmbH | Method and device for diagnostic investigation of biological samples |
| US7945318B2 (en) | 2003-03-20 | 2011-05-17 | Smithmarks, Inc. | Peripheral impedance plethysmography electrode and system with detection of electrode spacing |
| US8045770B2 (en) | 2003-03-24 | 2011-10-25 | Cornell Research Foundation, Inc. | System and method for three-dimensional image rendering and analysis |
| EP1613212B1 (en) | 2003-03-25 | 2016-08-24 | Fresenius Medical Care Holdings, Inc. | Device and method for performing electrical impedance tomography |
| DE10315863B4 (en) | 2003-04-08 | 2013-03-14 | Dräger Medical GmbH | electrode belt |
| US20040204658A1 (en) | 2003-04-10 | 2004-10-14 | Dietz Phillip W. | Systems and methods for providing an enhanced bioelectric sensing surface |
| US7149573B2 (en) | 2003-04-25 | 2006-12-12 | Medtronic, Inc. | Method and apparatus for impedance signal localizations from implanted devices |
| JP2004329412A (en) | 2003-05-02 | 2004-11-25 | Tanita Corp | Body composition measuring instrument |
| AU2004236588B2 (en) | 2003-05-12 | 2009-07-09 | Cheetah Medical, Inc. | System, method and apparatus for measuring blood flow and blood volume |
| US20040236202A1 (en) | 2003-05-22 | 2004-11-25 | Burton Steven Angell | Expandable strap for use in electrical impedance tomography |
| US7186220B2 (en) | 2003-07-02 | 2007-03-06 | Cardiac Pacemakers, Inc. | Implantable devices and methods using frequency-domain analysis of thoracic signal |
| US8682424B2 (en) | 2003-07-31 | 2014-03-25 | Dst Delta Segments Technology, Inc. | Noninvasive multi-channel monitoring of hemodynamic parameters |
| JP4406549B2 (en) | 2003-09-22 | 2010-01-27 | 富士フイルム株式会社 | Light modulation element, light modulation array element, and exposure apparatus using the same |
| US7390303B2 (en) | 2003-09-30 | 2008-06-24 | Ehud Dafni | Assessment of vascular dilatation |
| US8428717B2 (en) | 2003-10-14 | 2013-04-23 | Medtronic, Inc. | Method and apparatus for monitoring tissue fluid content for use in an implantable cardiac device |
| JP4600916B2 (en) | 2003-11-07 | 2010-12-22 | 株式会社タニタ | Shielded cable and bioelectrical impedance value or biological composition information acquisition device using shielded cable |
| JP2005143786A (en) | 2003-11-14 | 2005-06-09 | Tanita Corp | Organism measurement instrument |
| US20050113704A1 (en) | 2003-11-26 | 2005-05-26 | Lawson Corey J. | Patient monitoring system that incorporates memory into patient parameter cables |
| US7184821B2 (en) | 2003-12-03 | 2007-02-27 | Regents Of The University Of Minnesota | Monitoring thoracic fluid changes |
| KR20050072990A (en) | 2004-01-08 | 2005-07-13 | 황인덕 | Electrical impedance measuring apparatus |
| EP1723570A4 (en) | 2004-02-09 | 2010-06-02 | Inst Cardiologie De Montreal M | Computation of a geometric parameter of a cardiac chamber from a cardiac tomography data set |
| CA2555807A1 (en) | 2004-02-12 | 2005-08-25 | Biopeak Corporation | Non-invasive method and apparatus for determining a physiological parameter |
| JP4646614B2 (en) | 2004-03-01 | 2011-03-09 | 株式会社タニタ | Body composition measuring device |
| JP2005253840A (en) | 2004-03-15 | 2005-09-22 | Tanita Corp | Skin condition estimating device |
| WO2005094498A2 (en) | 2004-03-24 | 2005-10-13 | Noninvasive Medical Technologies, Llc | Thoracic impedance monitor and electrode array and method of use |
| US20050261743A1 (en) | 2004-05-19 | 2005-11-24 | Kroll Mark W | System and method for automated fluid monitoring |
| US7970461B2 (en) | 2004-06-18 | 2011-06-28 | Andres Kink | Method and apparatus for determining conditions of a biological tissue |
| EP1765161B1 (en) | 2004-06-18 | 2019-09-25 | Impedimed Limited | Oedema detection |
| US8068906B2 (en) | 2004-06-21 | 2011-11-29 | Aorora Technologies Pty Ltd | Cardiac monitoring system |
| WO2005122881A1 (en) | 2004-06-21 | 2005-12-29 | Aorora Technologies Pty Ltd | Cardiac monitoring system |
| US7387610B2 (en) | 2004-08-19 | 2008-06-17 | Cardiac Pacemakers, Inc. | Thoracic impedance detection with blood resistivity compensation |
| JP4578187B2 (en) | 2004-08-31 | 2010-11-10 | 株式会社タニタ | Body composition meter with judgment function for children |
| US9820658B2 (en) | 2006-06-30 | 2017-11-21 | Bao Q. Tran | Systems and methods for providing interoperability among healthcare devices |
| US7865236B2 (en) | 2004-10-20 | 2011-01-04 | Nervonix, Inc. | Active electrode, bio-impedance based, tissue discrimination system and methods of use |
| US20060085048A1 (en) | 2004-10-20 | 2006-04-20 | Nervonix, Inc. | Algorithms for an active electrode, bioimpedance-based tissue discrimination system |
| US7660617B2 (en) | 2004-11-13 | 2010-02-09 | The Boeing Company | Electrical impedance tomography using a virtual short measurement technique |
| US7270580B2 (en) | 2004-11-22 | 2007-09-18 | Cardio Dynamics International Corporation | Methods and apparatus for conducting electrical current |
| US20060111652A1 (en) | 2004-11-22 | 2006-05-25 | Mcleod Kenneth J | Method for enhancing blood and lymph flow in the extremities |
| US7214107B2 (en) | 2004-11-22 | 2007-05-08 | Cardiodynamics International Corporation | Electrical connector apparatus and methods |
| WO2006056074A1 (en) | 2004-11-26 | 2006-06-01 | Z-Tech (Canada) Inc. | Weighted gradient method and system for diagnosing disease |
| DE102004059082A1 (en) | 2004-12-02 | 2006-06-08 | Biotronik Crm Patent Ag | Device for determining the thorax impedance |
| WO2006074092A1 (en) | 2005-01-05 | 2006-07-13 | Rensselaer Polytechnic Institute | High precision voltage source for electrical impedance tomography |
| US7242169B2 (en) | 2005-03-01 | 2007-07-10 | Apple Inc. | Method and apparatus for voltage compensation for parasitic impedance |
| JP4645266B2 (en) | 2005-03-30 | 2011-03-09 | オムロンヘルスケア株式会社 | Body fat measuring device |
| US20060224079A1 (en) | 2005-03-31 | 2006-10-05 | Washchuk Bohdan O | Edema monitoring system and method utilizing an implantable medical device |
| US7603170B2 (en) | 2005-04-26 | 2009-10-13 | Cardiac Pacemakers, Inc. | Calibration of impedance monitoring of respiratory volumes using thoracic D.C. impedance |
| US7389147B2 (en) | 2005-04-29 | 2008-06-17 | Medtronic, Inc. | Therapy delivery mode selection |
| US9089275B2 (en) | 2005-05-11 | 2015-07-28 | Cardiac Pacemakers, Inc. | Sensitivity and specificity of pulmonary edema detection when using transthoracic impedance |
| US7907997B2 (en) | 2005-05-11 | 2011-03-15 | Cardiac Pacemakers, Inc. | Enhancements to the detection of pulmonary edema when using transthoracic impedance |
| US7340296B2 (en) | 2005-05-18 | 2008-03-04 | Cardiac Pacemakers, Inc. | Detection of pleural effusion using transthoracic impedance |
| GB2426824A (en) | 2005-06-03 | 2006-12-06 | Sheffield Teaching Hospitals | Body tissue impedance measuring probe with wireless transmitter |
| GB0511323D0 (en) | 2005-06-03 | 2005-07-13 | Sheffield Teaching Hospitals | Apparatus for measuring tissue sample electrical impedance |
| US8781551B2 (en) | 2005-07-01 | 2014-07-15 | Impedimed Limited | Apparatus for connecting impedance measurement apparatus to an electrode |
| WO2007002991A1 (en) | 2005-07-01 | 2007-01-11 | Impedimed Limited | Monitoring system |
| DE102005031752B4 (en) | 2005-07-07 | 2017-11-02 | Drägerwerk AG & Co. KGaA | Electroimpedance tomography device with common-mode signal suppression |
| US7205782B2 (en) | 2005-07-11 | 2007-04-17 | Brigham Young University | Scanned impedance imaging system method and apparatus |
| WO2007009183A1 (en) | 2005-07-20 | 2007-01-25 | Impedance Cardiology Systems, Inc. | Index determination |
| DE602005004282T2 (en) | 2005-08-17 | 2008-11-27 | Osypka Medical Gmbh | Digital demodulation apparatus and method for measuring electrical bioimpedance or bioadmittance |
| DE102005041385B4 (en) | 2005-09-01 | 2018-10-04 | Drägerwerk AG & Co. KGaA | Device for protecting an electrical impedance tomograph against overvoltage pulses |
| WO2007041783A1 (en) | 2005-10-11 | 2007-04-19 | Impedance Cardiology Systems, Inc. | Hydration status monitoring |
| US7733224B2 (en) | 2006-06-30 | 2010-06-08 | Bao Tran | Mesh network personal emergency response appliance |
| CN100423688C (en) | 2005-10-19 | 2008-10-08 | 深圳迈瑞生物医疗电子股份有限公司 | Method and apparatus for inhibiting power frequency common-mode interference |
| US8108047B2 (en) | 2005-11-08 | 2012-01-31 | Newlife Sciences Llc | Device and method for the treatment of pain with electrical energy |
| US7353058B2 (en) | 2005-12-07 | 2008-04-01 | Healthy Biotech Corp. Ltd. | Bio-impedance sensing device for homecare and eHealth |
| JP5124881B2 (en) | 2005-12-20 | 2013-01-23 | アウトポイエジ・パルテイシパソエス・リミタダ | Electrode assembly for electrical impedance tomography |
| US8442627B2 (en) | 2005-12-30 | 2013-05-14 | Medtronic, Inc. | Advanced thoracic fluid monitoring capability with impedance |
| KR100700112B1 (en) | 2006-02-03 | 2007-03-28 | 경희대학교 산학협력단 | System and method for Electrical Impedance Tomography |
| JP5069878B2 (en) | 2006-07-19 | 2012-11-07 | フクダ電子株式会社 | Vein inspection device |
| EP2091425A4 (en) | 2006-11-30 | 2012-07-25 | Impedimed Ltd | Measurement apparatus |
| EP2106241B1 (en) | 2007-01-15 | 2015-05-06 | Impedimed Limited | Method for performing impedance measurements on a subject |
| WO2008119166A1 (en) | 2007-03-30 | 2008-10-09 | Z-Tech (Canada) Inc. | Active guarding for reduction of resistive and capactive signal loading with adjustable control of compensation level |
| EP2155042A1 (en) | 2007-04-30 | 2010-02-24 | Clifford C. Dacso | Non-invasive monitoring of physiological measurements in a distributed health care environment |
| US20090318778A1 (en) | 2007-04-30 | 2009-12-24 | Clifford Dacso | Non-invasive monitoring of physiological measurements in a distributed health care environment |
| US20110251513A1 (en) | 2007-05-14 | 2011-10-13 | Impedimed Limited | Indicator |
| US8467880B2 (en) | 2007-08-23 | 2013-06-18 | Bioness Inc. | System for transmitting electrical current to a bodily tissue |
| KR101629974B1 (en) | 2007-09-07 | 2016-06-14 | 플로레, 잉고 | Medical measurement device for bioelectrical impedance measurement |
| WO2009036313A1 (en) | 2007-09-14 | 2009-03-19 | Corventis, Inc. | Adherent device with multiple physiological sensors |
| US8790257B2 (en) | 2007-09-14 | 2014-07-29 | Corventis, Inc. | Multi-sensor patient monitor to detect impending cardiac decompensation |
| US9411936B2 (en) | 2007-09-14 | 2016-08-09 | Medtronic Monitoring, Inc. | Dynamic pairing of patients to data collection gateways |
| US20090076349A1 (en) | 2007-09-14 | 2009-03-19 | Corventis, Inc. | Adherent Multi-Sensor Device with Implantable Device Communication Capabilities |
| US9204449B2 (en) | 2008-01-22 | 2015-12-01 | Alcatel Lucent | Method of assigning an idle state access terminal to a carrier in a multiple carrier wireless communication system based on load on control channel resources |
| AU2008207672B2 (en) | 2008-02-15 | 2013-10-31 | Impedimed Limited | Impedance Analysis |
| US20090264776A1 (en) | 2008-04-17 | 2009-10-22 | Terence Vardy | Measurement of physiological characteristics |
| GB0822237D0 (en) | 2008-12-05 | 2009-01-14 | Howell Steven | Remote health and security monitoring |
| US9974463B2 (en) | 2009-08-21 | 2018-05-22 | Beth Israel Deaconess Medical Center, Inc. | Hand-held device for electrical impedance myography |
| US9615767B2 (en) | 2009-10-26 | 2017-04-11 | Impedimed Limited | Fluid level indicator determination |
| JP2013514845A (en) | 2009-12-21 | 2013-05-02 | インぺディメッド リミテッド | Impedance measurement analysis |
-
2007
- 2007-05-25 WO PCT/AU2007/000726 patent/WO2007137333A1/en active Application Filing
- 2007-05-25 ES ES07718972.8T patent/ES2545730T3/en active Active
- 2007-05-25 CA CA2653406A patent/CA2653406C/en not_active Expired - Fee Related
- 2007-05-25 EP EP20070718972 patent/EP2020918B1/en not_active Not-in-force
- 2007-05-25 US US12/302,914 patent/US8761870B2/en not_active Expired - Fee Related
- 2007-05-25 AU AU2007266311A patent/AU2007266311B2/en not_active Ceased
- 2007-05-25 JP JP2009512367A patent/JP5431147B2/en not_active Expired - Fee Related
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5280429A (en) * | 1991-04-30 | 1994-01-18 | Xitron Technologies | Method and apparatus for displaying multi-frequency bio-impedance |
| US6125297A (en) * | 1998-02-06 | 2000-09-26 | The United States Of America As Represented By The United States National Aeronautics And Space Administration | Body fluids monitor |
| WO2000040955A1 (en) * | 1999-01-05 | 2000-07-13 | Kaiku Limited | Impedance measurements of bodily matter |
| WO2001067098A1 (en) | 2000-03-03 | 2001-09-13 | Btg International Limited | Electrical impedance measuring method for differentiating tissue types |
| US20030105411A1 (en) * | 2000-03-03 | 2003-06-05 | Smallwood Rodney Harris | Electrical impedance measuring method for differentiating tissue types |
| US20040015095A1 (en) * | 2002-07-16 | 2004-01-22 | Jianhua Li | Method and device for measuring signals for electrical impedance tomography by using correlation techinique |
| WO2004047635A1 (en) * | 2002-11-22 | 2004-06-10 | Impedimed Pty Ltd | Multifrequency bioimpedance determination |
| WO2005027717A2 (en) * | 2003-09-12 | 2005-03-31 | Renal Research Institute, Llc | Bioimpedance methods and apparatus |
| WO2007014417A1 (en) * | 2005-08-02 | 2007-02-08 | Impedimed Limited | Impedance parameter values |
Non-Patent Citations (5)
| Title |
|---|
| B. J. THOMAS*L; L. C. W ARD2; B. H. CORNISH: "Bioimpedance Spectrometry in the Determination of Body Water Compartments: Accuracy and Clinical Significance", APPL. RADIAT. ISOT., vol. 49, no. 5/6, 1998, pages 447 - 455 |
| GUDIVAKA R. ET AL.: "Single- and multifrequency models for bioelectrical impedance analysis of body water compartments", APPL. PHYSIOL., vol. 87, no. 3, 1999, pages 1087 - 1096, XP008090490 * |
| H. CRAMER: "Mathematical methods of statistics.", 1946, PRINCETON UNIVERSITY PRESS, pages: 474 - 477 |
| LIU R. ET AL.: "Primary Multi-frequency Data Analyze in Electrical Impedance Scanning", PROCEEDINGS OF THE IEEE-EMBS 2005, 27TH ANNUAL INTERNATIONAL CONFERENCE OF THE ENGINEERING IN MEDICINE AND BIOLOGY SOCIETY, SHANGHAI, CHINA, 1 September 2005 (2005-09-01) - 4 September 2005 (2005-09-04), pages 1504 - 1507, XP010908064 * |
| See also references of EP2020918A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105184062A (en) * | 2015-08-25 | 2015-12-23 | 中国人民解放军后勤工程学院 | User perception quality evaluation method based on confidence interval in crowd-sourcing perception network |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2007266311A1 (en) | 2007-12-06 |
| EP2020918A4 (en) | 2010-03-31 |
| CA2653406C (en) | 2015-03-17 |
| AU2007266311B2 (en) | 2014-01-30 |
| US8761870B2 (en) | 2014-06-24 |
| JP5431147B2 (en) | 2014-03-05 |
| EP2020918B1 (en) | 2015-05-20 |
| CA2653406A1 (en) | 2007-12-06 |
| ES2545730T3 (en) | 2015-09-15 |
| US20100087750A1 (en) | 2010-04-08 |
| JP2009538642A (en) | 2009-11-12 |
| EP2020918A1 (en) | 2009-02-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Li et al. | Deconvolving distribution of relaxation times, resistances and inductance from electrochemical impedance spectroscopy via statistical model selection: Exploiting structural-sparsity regularization and data-driven parameter tuning | |
| Saccoccio et al. | Optimal regularization in distribution of relaxation times applied to electrochemical impedance spectroscopy: ridge and lasso regression methods-a theoretical and experimental study | |
| Bondarenko | Analysis of large experimental datasets in electrochemical impedance spectroscopy | |
| US8099250B2 (en) | Impedance parameter values | |
| Copot et al. | Fractional order impedance model to estimate glucose concentration: in vitro analysis | |
| AU2007266311B2 (en) | Impedance measurements | |
| Pechlivanidis et al. | Robust informational entropy-based descriptors of flow in catchment hydrology | |
| EP3612826A1 (en) | Analyte measurement system and method | |
| US11666253B2 (en) | Methods and apparatus for analyte concentration monitoring using harmonic relationships | |
| Staudenmayer et al. | Robustness for general design mixed models using the t-distribution | |
| Hakula et al. | On the hp-adaptive solution of complete electrode model forward problems of electrical impedance tomography | |
| Ditlevsen et al. | Parameter estimation from observations of first-passage times of the Ornstein–Uhlenbeck process and the Feller process | |
| Renaut et al. | Stability and error analysis of the polarization estimation inverse problem for microbial fuel cells | |
| Contoyiannis et al. | Criticality in the relaxation phase of a spontaneously contracting atria isolated from a frog's heart | |
| Sorrentino et al. | Determination of the distribution of relaxation times through Loewner framework: A direct and versatile approach | |
| Xiong et al. | Time-singularity multifractal spectrum distribution based on detrended fluctuation analysis | |
| Olarte et al. | Measurement and characterization of glucose in NaCl aqueous solutions by electrochemical impedance spectroscopy | |
| Ibrahim et al. | Skin layer classification by feedforward neural network in bioelectrical impedance spectroscopy | |
| Olarte et al. | Using the best linear approximation as a first step to a new non-invasive glucose measurement | |
| Banks et al. | Existence and consistency of a nonparametric estimator of probability measures in the prohorov metric framework | |
| Olarte et al. | Fractional models in electrical impedance spectroscopy data for glucose detection | |
| McGree et al. | Optimal designs for studying bioimpedance | |
| Akhtar et al. | Digital signal processing techniques for gene finding in eukaryotes | |
| Dominicus et al. | Modelling variability in longitudinal data using random change point models | |
| Islam et al. | Effectivity of modified maximum likelihood estimators using selected ranked set sampling Data |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07718972 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007266311 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007718972 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2653406 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2009512367 Country of ref document: JP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2007266311 Country of ref document: AU Date of ref document: 20070525 Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12302914 Country of ref document: US |