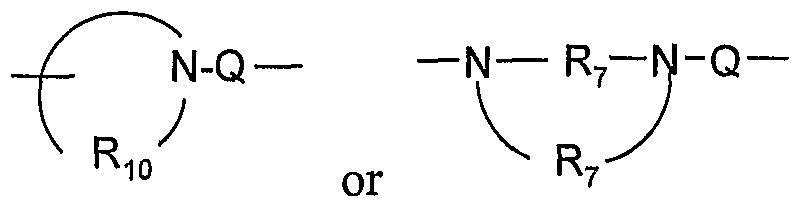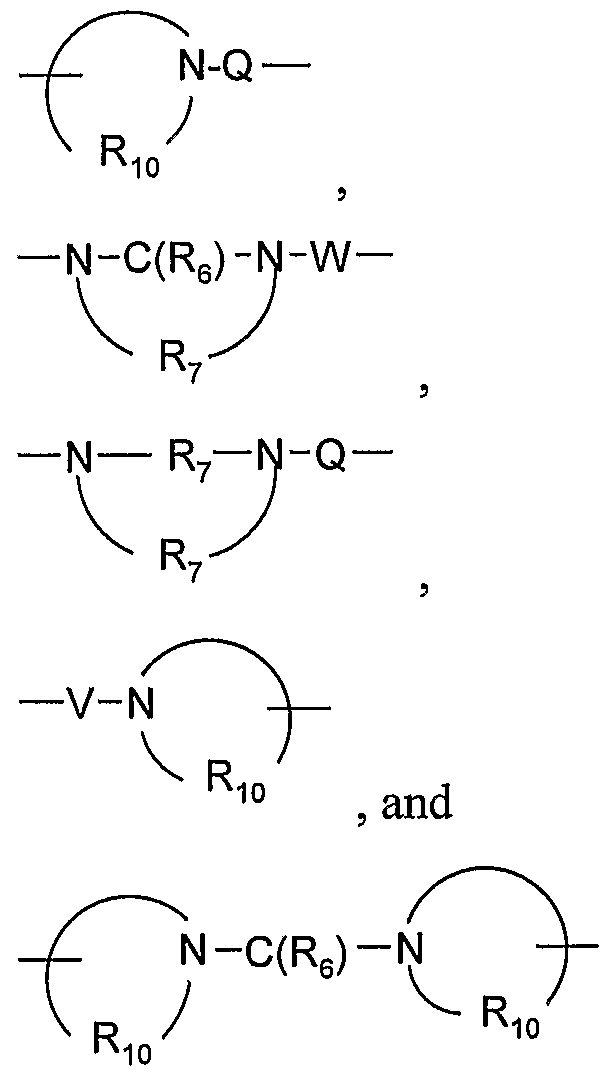WO2006107771A2 - PYRAZOLO[3,4-c]QUINOLINES, PYRAZOLO[3,4-c]NAPHTHYRIDINES, ANALOGS THEREOF, AND METHODS - Google Patents
PYRAZOLO[3,4-c]QUINOLINES, PYRAZOLO[3,4-c]NAPHTHYRIDINES, ANALOGS THEREOF, AND METHODS Download PDFInfo
- Publication number
- WO2006107771A2 WO2006107771A2 PCT/US2006/012031 US2006012031W WO2006107771A2 WO 2006107771 A2 WO2006107771 A2 WO 2006107771A2 US 2006012031 W US2006012031 W US 2006012031W WO 2006107771 A2 WO2006107771 A2 WO 2006107771A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- alkyl
- heteroaryl
- aryl
- hydroxy
- Prior art date
Links
- APODUKYURAYCBM-MRCIQYOPSA-N N=C(/C(/c1ccccc1N1)=C(/C(CC2CCNCC2)O)\Nc2ccccc2)C1=O Chemical compound N=C(/C(/c1ccccc1N1)=C(/C(CC2CCNCC2)O)\Nc2ccccc2)C1=O APODUKYURAYCBM-MRCIQYOPSA-N 0.000 description 1
- QONQNICYFZQXQV-UHFFFAOYSA-N O=C1Nc(cccc2)c2-c2c(CCN3CCN(Cc4ccccc4)CC3)[n](-c3ccccc3)nc12 Chemical compound O=C1Nc(cccc2)c2-c2c(CCN3CCN(Cc4ccccc4)CC3)[n](-c3ccccc3)nc12 QONQNICYFZQXQV-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
- A61P21/04—Drugs for disorders of the muscular or neuromuscular system for myasthenia gravis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
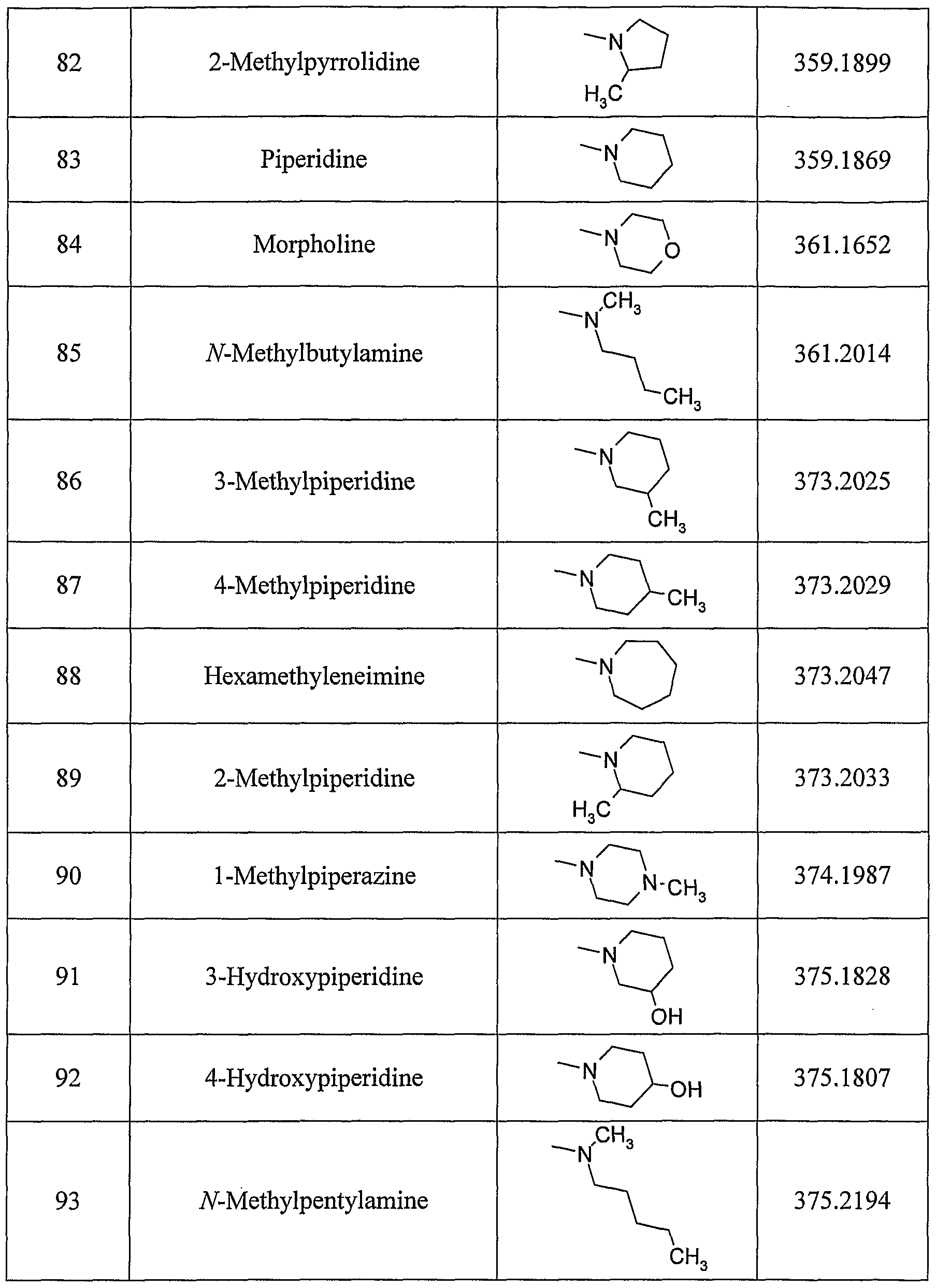
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/04—Antihaemorrhagics; Procoagulants; Haemostatic agents; Antifibrinolytic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/06—Antianaemics
Definitions
- IRMs immune response modifiers
- RA, RB, R C , R-Ah RBU RA2, RB2, RI, R 2 , R 3 , R, n, m, and p are as defined below; and pharmaceutically acceptable salts thereof.
- the compounds of Formulas I, Ia, II, III, IV, V, VI, VII, and VIII are useful as immune response modifiers (IRMs) due to their ability to modulate cytokine biosynthesis (e.g., inhibit the biosynthesis or production of one or more cytokines) and otherwise modulate the immune response when administered to animals.
- IRMs immune response modifiers
- the ability to modulate cytokine biosynthesis for example, inhibit the biosynthesis of tumor necrosis factor ( ⁇ ) (TNF- ⁇ ) or interleukin-1 (IL-I) makes the compounds useful for preventing or treating various conditions that are responsive to such changes in the immune response, for example, autoimmune diseases.
- the present invention provides pharmaceutical compositions that contain the immune response modifier compounds, and methods of modulating (e.g., inhibiting) cytokine biosynthesis in an animal, prophylactic treatment of a disease by inhibiting cytokine biosynthesis, and therapeutic treatment of a disease by inhibiting cytokine biosynthesis in an animal, by administering an effective amount of one or more compounds of the Formulas I, Ia, II, III, IV, V, VI, VII, and/or VIII, and/or pharmaceutically acceptable salts thereof to the animal.
- the invention provides methods of synthesizing compounds of the Formulas I, Ia, II, III, IV, V, VI, VII, and VIII and intermediates useful in the synthesis of these compounds.
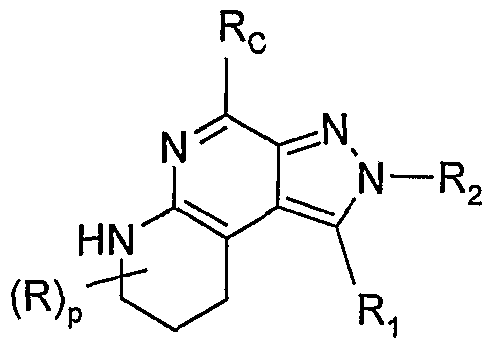
- the present invention provides compounds of the Formula I:
- RA and RB when taken together, RA and RB form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R' groups; or when taken together, RA and RB form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups;
- Rc is selected from the group consisting of hydrogen, alkyl, alkoxy, phenyl, phenoxy, heteroaryl, heteroaryloxy, halogen, haloalkyl, hydroxy, cyano, mercapto, nitro, carboxy, alkylamino, dialkylamino, benzylamino, heteroarylmethylamino, and cyclic amino; wherein phenyl, phenoxy, heteroaryl, heteroaryloxy, the phenyl ring of the benzyl group, and heteroaryl in the heteroarylmethylamino group are unsubstituted or substituted by one or more substituents independently selected from the group consisting of methyl, methoxy, bromo, chloro, fluoro, trifiuoromethyl, trifluoromethoxy, cyano, nitro, and hydroxy;
- R 1 is a non-interfering substituent
- R is selected from the group consisting of halogen, hydroxy, alkyl, alkenyl, haloalkyl, alkoxy, alkylthio, and -N(Rp) 2 ;
- R 1 is selected from the group consisting of: -R 4 ,
- R 2 is selected from the group consisting of hydrogen, halogen, hydroxy, -N(R 9 ), nitro, alkyl, aryl, and heteroaryl wherein the alkyl, aryl, or heteroaryl group is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy;
- X is selected from the group consisting of alkylene, alkenylene, alkynylene, arylene, heteroarylene, and heterocyclylene wherein the alkylene, alkenylene, and alkynylene groups can be optionally interrupted or terminated by arylene, heteroarylene or heterocyclylene and optionally interrupted by one or more -O- groups;
- Y is selected from the group consisting of: -O-,
- R 4 is selected from the group consisting of hydrogen, alkyl, alkenyl, alkynyl, aryl, arylalkylenyl, aryloxyalkylenyl, alkylarylenyl, heteroaryl, heteroarylalkylenyl, heteroaryloxyalkylenyl, alkylheteroarylenyl, and heterocyclyl wherein the alkyl, alkenyl, alkynyl, aryl, arylalkylenyl, aryloxyalkylenyl, alkylarylenyl, heteroaryl, heteroarylalkylenyl, heteroaryloxyalkylenyl, alkylheteroarylenyl, and heterocyclyl groups can be unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, haloalkyl, haloalkoxy, halogen,
- R 7 is C 2-7 alkylene;
- R 8 is selected from the group consisting of hydrogen, alkyl, alkoxyalkylenyl, hydroxyalkylenyl, arylalkylenyl, and heteroarylalkylenyl;
- R9 is selected from the group consisting of hydrogen and alkyl; Rio is C 3-8 alkylene; A is selected from the group consisting of -O-, -C(O)-, -S(O) 0-2 -, and -N(R 4 )-;
- a 1 is selected from the group consisting of -0-, -S(0)o- 2 - 3 -N ⁇ -Q-R 4 )-, and -CH 2 -;
- Q is selected from the group consisting of a bond, -C(R 6 )-, -C(R O )-C(R 6 )-, -S(O) 2 -, -C(R 6 )-N(R 8 )-W-, -S(O) 2 -N(R 8 )-, -C(Re)-O-, -C(Re)-S-, and -C(Re)-N(OR 9 )-;
- V is selected from the group consisting of -C(R 6 )-, -0-C(R 6 )-, -N(Rs)-C(R 6 )-, and
- W is selected from the group consisting of a bond, -C(O)-, and -S(O) 2 -; and a and b are independently integers from 1 to 6 with the proviso that a + b is ⁇ 7; with the proviso that R 1 is other than hydrogen, methyl, or phenyl; and with the further proviso that the compound is other than N-[l,l-dimethyl-2-(2-methyl-2H-pyrazolo[3,4- c]quinolin- 1 -yl)ethyl]benzamide; or a pharmaceutically acceptable salt thereof.
- the present invention provides compounds of the Formula II:
- RAI and RBI when taken together, form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R groups, or substituted by one R 3 group, or substituted by one R 3 group and one R group; or when taken together, RAI and RBI form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups;
- Rc, R, R 1 , and R 2 are as defined in Formula I;
- R 3 is selected from the group consisting of:
- X, Y, R 4 , and R 5 are as defined in Formula I;
- Z is a bond or -O-; with the proviso that R 1 is other than hydrogen, methyl, or phenyl; and with the further proviso that the compound is other than iV-[l,l-dimethyl-2-(2-methyl-2H-pyrazolo[3,4- c]quinolin- 1 -yl)ethyl]benzamide; or a pharmaceutically acceptable salt thereof.
- the present invention provides compounds of the Formula III:
- Rc, R 1 , R 2 , R, and R 3 are as defined in Formula II; n is an integer from 0 to 4; m is 0 or 1 , with the proviso that when m is 1 then n is 0 or 1 ; with the proviso that R 1 is other than hydrogen, methyl, or phenyl; and with the further proviso that the compound is other than N-[l,l-dimethyl-2-(2-methyl-2i7-pyrazolo[3,4- cjquinolin- 1 -yl)ethyl]benzamide; or a pharmaceutically acceptable salt thereof.
- the present invention provides compounds of the Formula IV:
- Rc, Ri, R 2 , and R are as defined in Formula I; n is an integer from 0 to 4; with the proviso that R 1 is other than hydrogen, methyl, or phenyl; or a pharmaceutically acceptable salt thereof.
- the present invention provides compounds of the Formulas V, VI, VII, and VIII:
- Rc, Ri, R 2 , R, and R 3 are as defined in Formula II; p is an integer from 0 to 3; m is 0 or 1, with the proviso that when m is 1 then p is 0 or 1 ; with the proviso that R 1 is other than hydrogen; or a pharmaceutically acceptable salt thereof.
- the present invention provides compounds of the Formulas IX, X, XI, and XII:
- Rc, Ri, R 2 , and R are as defined in Formula I; and p is an integer from O to 3; with the proviso that Ri is other than hydrogen; or a pharmaceutically acceptable salt thereof.
- the present invention provides a method of inhibiting cytokine biosynthesis comprising administering an effective amount of a compound of the Formula Ia:
- RA 2 and R ⁇ 2 are each independently selected from the group consisting of: hydrogen, halogen, alkyl, alkenyl, alkoxy, alkylthio, and or when taken together, RA 2 and RB 2 form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R 1 groups; or when taken together, RA 2 and RB 2 form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups; and
- Rc, R, R', Ri, and R 2 are as defined in Formula I; or a pharmaceutically acceptable salt thereof to an animal.
- non-interfering means that the ability of the compound or salt, which contains a non-interfering substituent, to modulate (e.g., inhibit) the biosynthesis of one or more cytokines is not destroyed by the non-interfering substitutent.
- Illustrative non- interfering R' groups include those described herein for R and R 3 .
- the terms “alkyl”, “alkenyl”, “alkynyl” and the prefix “alk-” are inclusive of both straight chain and branched chain groups and of cyclic groups, e.g., cycloalkyl and cycloalkenyl.
- these groups contain from 1 to 20 carbon atoms, with alkenyl groups containing from 2 to 20 carbon atoms, and alkynyl groups containing from 2 to 20 carbon atoms. In some embodiments, these groups have a total of up to 10 carbon atoms, up to 8 carbon atoms, up to 6 carbon atoms, or up to 4 carbon atoms. Cyclic groups can be monocyclic or polycyclic and preferably have from 3 to 10 ring carbon atoms.
- Exemplary cyclic groups include cyclopropyl, cyclopropylmethyl, cyclopentyl, cyclohexyl, adamantyl, and substituted and unsubstituted bornyl, norbornyl, and norbornenyl.
- alkylene, alkenylene, and alkynylene are the divalent forms of the “alkyl”, “alkenyl”, and “alkynyl” groups defined above.
- alkylenyl”, “alkenylenyl”, and “alkynylenyl” are used when “alkylene”, “alkenylene”, and “alkynylene”, respectively, are substituted.
- an arylalkylenyl group comprises an alkylene moiety to which an aryl group is attached.
- haloalkyl is inclusive of groups that are substituted by one or more halogen atoms, including perfluorinated groups. This is also true of other groups that include the prefix "halo-”. Examples of suitable haloalkyl groups are chloromethyl, trifluoromethyl, and the like.
- aryl as used herein includes carbocyclic aromatic rings or ring systems. Examples of aryl groups include phenyl, naphthyl, biphenyl, fluorenyl and indenyl.
- heteroatom refers to the atoms O, S, or N.
- heteroaryl includes aromatic rings or ring systems that contain at least one ring heteroatom (e.g., O 5 S, N).
- heteroaryl includes a ring or ring system that contains 2-12 carbon atoms, 1-3 rings, 1-4 heteroatoms, and O, S, and N as the heteroatoms.
- Suitable heteroaryl groups include furyl, thienyl, pyridyl, quinolinyl, isoquinolinyl, indolyl, isoindolyl, triazolyl, pyrrolyl, tetrazolyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, benzofuranyl, benzothiophenyl, carbazolyl, benzoxazolyl, pyrimidinyl, benzimidazolyl, quinoxalinyl, benzothiazolyl, naphthyridinyl, isoxazolyl, isothiazolyl, purinyl, quinazolinyl, pyrazinyl, 1-oxidopyridyl, pyridazinyl, triazinyl, tetrazinyl, oxadiazolyl, thiadiazolyl, and so on.
- heterocyclyl includes non-aromatic rings or ring systems that contain at least one ring heteroatom (e.g., O, S 5 N) and includes all of the folly saturated and partially unsaturated derivatives of the above mentioned heteroaryl groups.
- heterocyclyl includes a ring or ring system that contains 2-12 carbon atoms, 1-3 rings, 1-4 heteroatoms, and O, S, and N as the heteroatoms.
- heterocyclyl groups include pyrrolidinyl, tetrahydrofuranyl, morpholinyl, thiomorpholinyl, 1,1- dioxothiomorpholinyl, piperidinyl, piperazinyl, thiazolidinyl, imidazolidinyl, isothiazolidinyl, tetrahydropyranyl, quinuclidinyl, homopiperidinyl (azepanyl), 1 ,4- oxazepanyl, homopiperazinyl (diazepanyl), 1,3-dioxolanyl, aziridinyl, azetidinyl, dihydroisoquinolin-( lH)-yl, octahydroisoquinolin-(l H)-yl, dihydroquinolin-(2H)-yl, octahydroquinolin-(2H)-yl, dihydro--
- heterocyclyl includes bicyclic and tricyclic heterocyclic ring systems. Such ring systems include fused and/or bridged rings and spiro rings. Fused rings can include, in addition to a saturated or partially saturated ring, an aromatic ring, for example, a benzene ring. Spiro rings include two rings joined by one spiro atom and three rings joined by two spiro atoms.
- cyclic amino includes groups of the formula
- arylene is the divalent forms of the "aryl”, “heteroaryl”, and “heterocyclyl” groups defined above.
- arylenyl is used when “arylene”, “heteroarylene”, and “heterocyclylene”, respectively, are substituted.
- an alkylarylenyl group comprises an arylene moiety to which an alkyl group is attached.
- fused aryl ring includes fused carbocyclic aromatic rings or ring systems.
- offused aryl rings include benzo, naphtho, fluoreno, and indeno.
- the fused aryl ring is benzo.
- fused heteroaryl ring includes the fused forms of 5 or 6 membered aromatic rings that contain one heteroatom selected from S and N.
- the fused heteroaryl ring is pyrido or thieno.
- the fused heteroaryl ring is pyrido. In certain of these embodiments, the pyrido ring is
- fused 5 to 7 membered saturated ring includes rings which are fully saturated except for the bond where the ring is fused.
- the ring is a cyclohexene ring.
- the ring is tetrahydropyrido or dihydrothieno.
- the ring is
- the ring is or wherein the highlighted bond indicates the position where the ring is fused.
- each group is independently selected, whether explicitly stated or not.
- each Rg group is independently selected for the formula -N(R S )-C(R O )-N(R 8 )- each Rg group is independently selected.
- each R 4 group is independently selected when more than one Y group is present (i.e., R 1 and R 3 both contain a Y group) and each Y group contains one or more R 8 groups, then each Y group is independently selected, and each R 8 group is independently selected.
- the invention is inclusive of the compounds described herein and salts thereof, in any of their pharmaceutically acceptable forms, including isomers (e.g., diastereomers and enantiomers), solvates, polymorphs, prodrugs, and the like.
- the invention specifically includes each of the compound's enantiomers as well as racemic and scalemic mixtures of the enantiomers.
- the term "compound” or the term “compounds” includes any or all of such forms, whether explicitly stated or not (although at times, “salts" are explicitly stated).
- prodrug means a compound that can be transformed in vivo to yield an immune response modifying compound, including any of the salt, solvated, polymorphic, or isomeric forms described above.
- the prodrug itself, may be an immune response modifying compound, including any of the salt, solvated, polymorphic, or isomeric forms described above.
- the transformation may occur by various mechanisms, such as through a chemical (e.g., solvolysis or hydrolysis, for example, in the blood) or enzymatic biotransformation.
- a prodrug can comprise an ester formed by the replacement of the hydrogen atom of the acid group with a group such as Ci -8 alkyl, C 2-12 alkanoyloxymethyl, l-(alkanoyloxy)ethyl having from 4 to 9 carbon atoms, 1 -methyl- l-(alkanoyloxy)ethyl having from 5 to 10 carbon atoms, alkoxycarbonyloxymethyl having from 3 to 6 carbon atoms, l-(alkoxycarbonyloxy)ethyl having from 4 to 7 carbon atoms, 1 -methyl- 1- (alkoxycarbonyloxy)ethyl having from 5 to 8 carbon atoms, N- (alkoxycarbonyl)aminomethyl having from 3 to 9 carbon atoms
- alkylaminoC 2-3 alkyl such as ⁇ - dimethylaminoethyl
- carbamoyl-C ⁇ alkyl N,N-diC 1-2 alkylcarbamoyl-Ci -2 alkyl and piperidino-, pyrolidino-, or morpholinoC 2-3 alkyl.
- a prodrug can be formed by the replacement of the hydrogen atom of the alcohol group with a group such as C 1-6 alkanoyloxymethyl, 1-(C 1-6 alkanoyloxy)ethyl 5 1 -methyl- 1 -(C i -6 alkanoyloxy)ethyl, C i .6 alkoxycarbonyloxymethyl, N-(Ci -6 alkoxycarbonyl)aminomethyl, succinoyl, Ci -6 alkanoyl, ⁇ -aminoC 1-4 alkanoyl, arylacyl and ⁇ -aminoacyl, or ⁇ -aminoacyl- ⁇ -aminoacyl, where each ⁇ -aminoacyl group is independently selected from the naturally occurring L-amino acids, P(O)(OH) 2 , P(O)(O-Cj -6 alkyl) 2 or glycosyl (the radical resulting from the removal of
- a prodrug can be formed by the replacement of a hydrogen atom in the amine group with a group such as R"-carbonyl, R"-O-carbonyl, N(R")(R'")-carbonyl where R" and R'" are each independently Ci -I0 alkyl, C 3-7 cycloalkyl, benzyl, or R"-carbonyl is a natural ⁇ -aminoacyl or natural ⁇ -aminoacyl-natural ⁇ -aminoacyl, -C(OH)C(O)OY' wherein Y' is H, Ci -6 alkyl or benzyl, -C(OY 0 )Yi wherein Y 0 is Ci -4 alkyl and Y 1 is C 1-6 alkyl, carboxyCi- 6 alkyl, aminoCi- 4 alkyl or mono-N- or di-N,N-Ci_ 6 alkylaminoal
- tautomer or tautomeric form
- proton tautomers include interconversions via migration of a proton, such as keto-enol and imine-enamine isomerizations.
- proton migration between the 2- and 3- positions may occur.
- the compounds of the present invention may exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the present invention embraces both solvated and unsolvated forms.
- each one of the following variables e.g., RA, RB, RC, R, R', RI, R-2, R3, n, m, p, A, X, Y, Z 5 and so on
- each R' is independently a non- interfering substituent.
- RA and RB when taken together, RA and RB form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R' groups; or when taken together, R A and RB form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups;
- RA and R B form a fused aryl or heteroaryl ring.
- RA and R B form a fused aryl ring.
- RA and R B form a fused heteroaryl ring.
- RA and RB form a fused 5 to 7 membered saturated ring.
- RA and RB form a fused 5 to 7 membered saturated ring containing one heteroatom selected from the group consisting of N and S.
- the heteroatom is N.
- RA 2 and RB 2 are each independently selected from the group consisting of hydrogen, halogen, alkyl, alkenyl, alkoxy, alkylthio, and -N(R 9 ) 2 ; or when taken together, RA 2 and R B2 form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R' groups; or when taken together, RA 2 and RB 2 form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups.
- RA 2 and R B2 are each independently selected from the group consisting of hydrogen, halogen, alkyl, alkenyl, alkoxy, alkylthio, and -N(Rp) 2 ; or when taken together, RA 2 and RB 2 form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R groups, or substituted by one R 3 group, or substituted by one R 3 group and one R group; or when taken together, RA 2 and RB 2 form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups.
- R A2 and RB 2 are each independently selected from the group consisting of hydrogen, halogen, alkyl, alkenyl, alkoxy, alkylthio, and -N(R 9 ) 2 .
- RA 2 and R B2 form a fused aryl or heteroaryl ring.
- RA 2 and RB 2 form a fused aryl ring.
- RA 2 and R B2 form a fused heteroaryl ring.
- RA 2 and R B2 form a fused 5 to 7 membered saturated ring.
- RA 2 and RB 2 form a fused 5 to 7 membered saturated ring containing one heteroatom selected from the group consisting of N and S.
- the heteroatom is N.
- RA I and RBI when taken together, form a fused aryl ring or heteroaryl ring containing one heteroatom selected from the group consisting of N and S, wherein the aryl or heteroaryl ring is unsubstituted or substituted by one or more R groups, or substituted by one R 3 group, or substituted by one R 3 group and one R group; or when taken together, RAI and RBI form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, and unsubstituted or substituted by one or more R groups; wherein R is selected from the group consisting of halogen, hydroxy, alkyl, alkenyl, haloalkyl, alkoxy, alkylthio, and -N(R 9 ) 2 ; and R 3 is selected from the group consisting of -Z-R 4 , -Z-X-R 4 , -Z-X-Y-R 4
- RAI and R B I form a fused aryl ring.
- R A i and R B i form a fused benzene ring which is unsubstituted.
- R A i and R B i form a fused heteroaryl ring.
- RAI and RBI form a fused pyridine ring which is unsubstituted.
- RA 1 and RBI form a fused 5 to 7 membered saturated ring, optionally containing one heteroatom selected from the group consisting of N and S, wherein the ring is unsubstituted.
- RAI and RBI form a fused 5 to 7 membered saturated ring.
- RAI and RBI form a fused 5 to 7 membered saturated ring containing one heteroatom selected from the group consisting of N and S.
- the heteroatom is N.
- R is selected from the group consisting of alkyl and haloalkyl.
- R is selected from the group consisting of alkyl, alkoxy, halogen, and hydroxy.
- n 0.
- n 0.
- p is 0.
- m is 1 and p is 0.
- R 3 is selected from the group consisting of aryl, arylalkyleneoxy, heteroarylalkyleneoxy, and heteroaryl, wherein aryl, arylalkyleneoxy, and heteroaryl are unsubstituted or substituted with one or more substituents selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, and halogen.
- R 3 is selected from the group consisting of phenyl, 3- (hydroxymethyl)phenyl, benzyloxy, 3-furyl, pyridin-3-yl, 5-(hydroxymethyl)pyridin-3-yl, 6-chloropyridin-3-yl 5 6-fluoropyridin-3-yl 5 6-methylpyridin-3-yl, 3-quinolin-3-yl, thiazol- 4-ylmethoxy,/?-tolyl, (4-chlorobenzyl)oxy, and (4-methylbenzyl)oxy.
- R 3 is at the 7-position of the pyrazoloquinoline or pyrazolonaphthyridine.
- R 3 is selected from the group consisting of alkylsulfonylalkyleneoxy, alkylsulfonylaminoalkyleneoxy, alkylcarbonylaminoalkyleneoxy, aryl, arylalkyleneoxy, heteroaryl, heteroarylalkyleneoxy, heterocyclyl, heterocyclyloxy, heterocyclylalkyleneoxy, and heterocyclylcarbonylalkyleneoxy; wherein aryl and heteroaryl are unsubstituted or substituted by one or more substituents selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, halogen, heterocyclylcarbonyl, and dialkylaminocarbonyl; and wherein heterocyclyl is unsubstituted or substituted by one or more substitutents selected from the group consisting of alkylsulfonyl, alkylcarbony
- R 3 is pyridin-3-yl, pyridin-4-yl, 6-fluoropyridin-3-yl, 5- (hydroxymethyl)pyridin-3-yl, quinolin-3-yl, 2-ethoxyphenyl, or 3-(morpholine-4- carbonyl)phenyl, except where this definition is excluded.
- R 3 is 2-oxo-l,3-oxazolidin-3-yl, except where this definition is excluded.
- R 3 is l,3-thiazol-4-ylmethoxy, (1 -methyl- lH-imidazol- 2-yl)methoxy, or pyridin-3-ylmethoxy, except where this definition is excluded.
- R 3 is 2-pyrrolidin-l-ylethoxy, 2-(2-oxopyrrolidin-l- yl)ethoxy, 2-(l,l-dioxidoisothiazolidin-2-yl)ethoxy, 2-morpholin-4-ylethoxy, 3-pyrrolidin- 1 -ylpropoxy, 3 -(2-oxopyrrolidin- 1 -yl)propoxy, 3 -( 1 , 1 -dioxidoisothiazolidin-2-yl)propoxy, 3-morpholin-4-ylpropoxy, 2-morpholin-4-yl-2-oxoethoxy, and , wherein R is alkylsulfonyl or alkylcarbonyl, except where this definition is excluded.
- R 3 is alkyl-S(O) 2 -NH-(CH 2 ) 2 - 3 -O-, alkyl-S(O) 2 -(CH 2 ) 2-3 -O- 5 or alkyl-C(O)-NH-(CH 2 ) 2-3 -O-, except where this definition is excluded.
- m is 0, except where this definition of m is excluded.
- m is 0, except where this definition of m is excluded.
- Rc is selected from the group consisting of hydroxy, halogen, alkyl, haloalkyl, phenyl, phenoxy, heteroaryl, and cyclic amino; wherein phenyl, phenoxy, and heteroaryl are unsubstituted or substituted by one or more substituents independently selected from the group consisting of methyl, methoxy, bromo, chloro, fluoro, trifluoromethyl, trifluoromethoxy, cyano, nitro, and hydroxy.
- Rc is selected from the group consisting of hydroxy, chloro, methyl, trifluoromethyl, phenyl, phenoxy, 1-morpholino, 1-piperidino, 4- methylpiperazin-1-yl, and heteroaryl wherein heteroaryl is a 5 or 6 membered monocyclic ring containing one or two heteroatoms.
- R 2 is selected from the group consisting of hydrogen, alkyl, aryl, and heteroaryl wherein the alkyl, aryl, or heteroaryl group is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy.
- R 2 is selected from the group consisting of hydrogen, alkyl, aryl, and heteroaryl, wherein aryl and heteroaryl are unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy.
- R 2 is Q -4 alkyl.
- R 2 is aryl or heteroaryl, each of which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy, except where this definition of R 2 is excluded.
- R 2 is heteroaryl which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy, and wherein heteroaryl is a monocyclic, 5 or 6 membered ring containing 1 or two heteroatoms, except where this definition of R 2 is excluded.
- R 2 is selected from the group consisting of furan-2-yl, pyrrol-2-yl, 1 -methylpyrrol-2-yl, thiophen-2-yl, 3-methylthiophen-2-yl, 5- methylthiophen-2-yl, imidazol-2-yl, thiazol-2-yl, pyridin-2-yl, pyridin-3-yl, and pyridin-4- yl, each of which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of methyl, methoxy, bromo, chloro, fluoro, trifluoromethyl, trifluoromethoxy, cyano, nitro, hydroxy, except where this definition of R 2 is excluded.
- R 2 is phenyl which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, alkylthio, aryl, halogen, haloalkyl, haloalkoxy, cyano, nitro, amino, hydroxy, hydroxyalkyl, hydroxyalkoxy, acetyloxy, and alkoxycarbonylalkoxy, except where this definition of R 2 is excluded.
- phenyl is unsubstituted or substituted by one or more substituents independently selected from the group consisting of methyl, methoxy, bromo, chloro, fluoro, trifluoromethyl, trifluoromethoxy, cyano, nitro, and hydroxy, except where this definition of R 2 is excluded.
- R 1 is -X-R 4 .
- R 1 is other than hydrogen.
- Ri is other than hydrogen, methyl, or phenyl.
- Ri is other than hydrogen, methyl, or phenyl; and the compound is other than N-[l,l-dimethyl-2-(2-methyl-2//-pyrazolo[3,4-c]quinolin-l- yl)ethyl]benzamide.
- X is -(CH 2 )i_ 3 -
- R 4 is aryl, heteroaryl, or heterocyclyl, each of which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, haloalkyl, haloalkoxy, halogen, nitro, hydroxy, mercapto, cyano, aryl, aryloxy, arylalkyleneoxy, heteroaryl, heteroaryloxy, heteroarylalkyleneoxy, heterocyclyl, amino, alkylamino, dialkylamino, (dialkylamino)alkyleneoxy, and in the case of heterocyclyl, oxo, amidino, and oximido.
- R 4 is a saturated, nitrogen-containing heterocyclyl group which is unsubstituted or substituted by oxo, amidino, or oximido.
- R 4 is selected from the group consisting of piperidin-1-yl, piperidin-2-yl, piperidin-3-yl, piperidin-4-yl, piperazin- 1-yl, morpholin-4-yl, morpholin-2-yl, pyrrolidin-1-yl, and pyrrolidin-2-yl.
- R 4 is piperidin-4-yl or piperazin-1-yl.
- X is -CH 2 - or -CH 2 CH 2 -.
- Ri is -X-R 4
- Ri is -X-Y-R 4
- X is -(CH 2 ) J-3 -
- Y is
- R 7 is ethylene
- R 1 O is -(CHa) 4-S -
- R 4 is Ci -4 alkyl, phenyl, 4-chlorophenyl, 4- fluorophenyl, benzyl, 4-chlorobenzyl, or 4-fluorobenzyl.
- Rio is -(CH 2 ) 5 -.
- X is -CH 2 CH 2 -.
- R 1 is -X-Y-R 4
- X is -CH 2 -
- Y is -O- or -N(R 8 )-
- R 4 is aryl, heteroaryl, or heterocyclyl.
- Ri is -X-R 4 or -X-Y-R 4
- Ri is -X-Y-X-Y-R 4 .
- R 1 is
- Y is -C(O)-, -S(O) 2 -,
- R 4 is C 1-5 alkyl, phenyl, or pyridyl.
- R 1 is -X-R 4 , -X-Y-R 4 , or -X-Y-X-Y-R 4
- R 1 is -X-R 5 .
- -X- is -(CH 2 ) M -.
- R 5 is For certain of these embodiments.
- V is -C(O)-
- A is -N(R 4 )-
- a and b are both 2.
- R 3 is at the 7-position of the pyrazoloquinoline or pyrazolonaphthyridine.
- m is 1
- n is O.
- R 3 is at the 8-position of the pyrazoloquinoline or pyrazolonaphthyridine.
- m is 1, and n is O.
- R 4 is selected from the group consisting of hydrogen, alkyl, alkenyl, alkynyl, aryl, arylalkylenyl, aryloxyalkylenyl, alkylarylenyl, heteroaryl, heteroarylalkylenyl, heteroaryloxyalkylenyl, alkylheteroarylenyl, and heterocyclyl wherein the alkyl, alkenyl, alkynyl, aryl, arylalkylenyl, aryloxyalkylenyl, alkylarylenyl, heteroaryl, heteroarylalkylenyl, heteroaryloxyalkylenyl, alkylheteroarylenyl, and heterocyclyl groups can be unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, haloalkyl, haloalkoxy,
- R 4 is aryl, heteroaryl, or heterocyclyl, each of which is unsubstituted or substituted by one or more substituents independently selected from the group consisting of alkyl, alkoxy, hydroxyalkyl, haloalkyl, haloalkoxy, halogen, nitro, hydroxy, mercapto, cyano, aryl, aryloxy, arylalkyleneoxy, heteroaryl, heteroaryloxy, heteroarylalkyleneoxy, heterocyclyl, amino, alkylamino, dialkylamino,
- dialkylamino alkyleneoxy, and in the case of heterocyclyl, oxo, amidino, and oximido.
- R 4 is a saturated, nitrogen-containing heterocyclyl group which is unsubstituted or substituted by oxo, amidino, or oximido.
- R 4 is selected from the group consisting of piperidin-1- yl, piperidin-2-yl, piperidin-3-yl, piperidin-4-yl, piperazin-1-yl, morpholin-4-yl, morpholin-2-yl, pyrrolidin-1-yl, and pyrrolidin-2-yl.
- R 4 is piperidin-4-yl or piperazin-1-yl.
- R 4 is Ci -4 alkyl, phenyl, 4-chlorophenyl, 4-fluorophenyl, benzyl, 4-chlorobenzyl, or 4-fluorobenzyl.
- R 4 is aryl, heteroaryl, or heterocyclyl.
- R 4 is alkyl, aryl, or heteroaryl.
- R 4 is C 1-5 alkyl, phenyl, or pyridyl.
- R 4 is alkyl.
- R 4 is aryl.
- R 4 is heteroaryl.
- R 5 is selected from the group consisting of
- R 5 is selected from the group consisting of
- R 5 is selected from the group consisting of
- R 5 is .
- V is -C(O)-.
- A is -N(R 4 )-.
- a and b are both 2.
- R 6 0.
- R 6 S.
- R 7 is C 2-7 alkylene. In certain embodiments, R 7 is C 3-4 alkylene. In certain embodiments, R 7 is ethylene.
- R 8 is selected from the group consisting of hydrogen, alkyl, alkoxyalkylenyl, hydroxyalkylenyl, arylalkylenyl, and heteroarylalkylenyl. In certain embodiments, R 8 is hydrogen. In certain embodiments, R 8 is alkyl.
- R 9 is selected from the group consisting of hydrogen and alkyl. In certain embodiments, R 9 is alkyl. In certain embodiments, R 9 is hydrogen.
- R 10 is C 3-8 alkylene.
- Rj o is C 4-5 alkylene. In certain embodiments, Ri 0 is -(CH 2 ) 4-5 -. In certain embodiments, Rj 0 is
- A is selected from the group consisting of -0-, -C(O)-, -S(O) 0-2 -, and -N(R 4 )-. In certain embodiments, A is selected from the group consisting of -0-, -C(O)-, and -N(R 4 )-. In certain embodiments, A is -0-. In certain embodiments, A is -N(R 4 )-.
- A' is selected from the group consisting of -0-, -S(O) 0-2 -, -N(-Q-R 4 )-, and -CH 2 -.
- a 1 is -0-.
- A' is -CH 2 -.
- a 1 is -N ⁇ Q-R 4 )-.
- Q is a bond and R 4 is alkyl.
- Q is selected from the group consisting of a bond,
- Q is a bond, -C(R 6 )-, -S(O) 2 -, or -C(Re)-O-.
- Q is a bond, -C(O)-, -S(O) 2 -, -C(Re)-N(Re)-, or -S(O) 2 -N(R 8 )-.
- Q is a bond, -C(RO)-, -S(O) 2 -, or -C(Ro)-N(Rg)-W-.
- Q is a bond.
- V is selected from the group consisting Of-C(R 6 )-, -0-C(R 6 )-, -N(Rs)-C(R 6 )-, and -S(O) 2 -. In certain embodiments, V is -C(R 6 )-. In certain embodiments, V is -C(O)-. In certain embodiments, V is -N(Rs)-C(R 6 )-.
- W is selected from the group consisting of a bond,
- W is selected from the group consisting of a bond and -C(O)-. In certain embodiments, W is a bond.
- X is selected from the group consisting of alkylene, alkenylene, alkynylene, arylene, heteroarylene, and heterocyclylene wherein the alkylene, alkenylene, and alkynylene groups can be optionally interrupted or terminated by arylene, heteroarylene or heterocyclylene and optionally interrupted by one or more -O- groups.
- X is alkylene. In certain embodiments, X is C 1-4 alkylene. In certain embodiments, -X- is -(CH 2 ) 1-4 -. In certain embodiments, X is -(CH 2 ) J-3 -. In certain embodiments, X is -CH 2 CH 2 -. In certain embodiments, X is -CH 2 -. For certain embodiments, Y is selected from the group consisting of -0-, -S(O) 0-2 -,
- Y is
- R 7 is ethylene
- Ri 0 is -(CH 2 Vs-.
- Y is -C(O)-, -S(O) 2 -, -S(O) 2 -N(R 8 )-, or -C(O)-N(R 8 )-.
- Y is -O- or -N(R 8 )-.
- Z is a bond or -0-. In certain embodiments, Z is a bond.
- Z is -0-.
- a and b are independently integers from 1 to 6 with the proviso that a + b is ⁇ 7.
- a and b are each the integer 2.
- n 0, or m is 0.
- n is 1.
- n 0.
- p is 0, or m is 0.
- m and p are 0.
- n is 0, and p is 1.
- m is 1
- p is 0.
- n 0.
- the present invention provides a pharmaceutical composition
- a pharmaceutical composition comprising a therapeutically effective amount of a compound or salt of any one of Formulas I, Ia, II, III, IV, V, VI, VII, VIII, IX, X, XI, XII, or any one of the above embodiments in combination with a pharmaceutically acceptable carrier.
- the present invention provides a method of inhibiting cytokine biosynthesis comprising administering an effective amount of a compound or salt of any one of Formulas I, Ia, II, III, IV, V, VI, VII, VIII, IX, X, XI, XII, or any one of the above embodiments or administering any one of the above pharmaceutical compositions to the animal.
- the cytokine is TNF- ⁇ or IL-I .
- the present invention provides a method of therapeutic treatment of a disease by inhibiting cytokine biosynthesis, comprising administering a therapeutically effective amount of a compound or salt of any one of Formulas I, Ia, II, III, IV, V, VI, VII, VIII, IX, X, XI, XII, or any one of the above embodiments or administering any one of the above pharmaceutical compositions to the animal.
- the cytokine is TNF- ⁇ or IL-I.
- the present invention provides a method of prophylactic treatment of a disease by inhibiting cytokine biosynthesis, comprising administering a therapeutically effective amount of a compound or salt of any one of Formulas I, Ia, II, III, IV, V, VI, VII, VIII, IX, X, XI, XII, or any one of the above embodiments or administering any one of the above pharmaceutical compositions to the animal.
- the cytokine is TNF- ⁇ or IL-I.
- Compounds of the invention may be synthesized by synthetic routes that include processes analogous to those well known in the chemical arts, particularly in light of the description contained herein.
- the starting materials are generally available from commercial sources such as Aldrich Chemicals (Milwaukee, Wisconsin, USA) or are readily prepared using methods well known to those skilled in the art (e.g. prepared by methods generally described in Louis F. Fieser and Mary Fieser, Reagents for Organic Synthesis, v. 1-19, Wiley, New York, (1967-1999 ed.); Alan R. Katritsky, Otto Meth- Cohn, Charles W. Rees, Comprehensive Organic Functional Group Transformations, v 1- 6, Pergamon Press, Oxford, England, (1995); Barry M.
- an indole of Formula XX is acylated to provide an oxalated indole of Formula XXI.
- the reaction can be carried out by adding ethyl chlorooxoacetate to a solution of an indole of Formula XX in a suitable solvent such as diethyl ether in the presence of pyridine.
- the reaction can be carried out at a sub-ambient temperature such as 0 °C.
- Many indoles of Formula XX are known. Some are commercially available and others can be readily prepared using known synthetic methods.
- step (2) of Reaction Scheme I an oxalated indole of Formula XXI is rearranged to a pyrazolo[3,4-c]quinolin-4-one of Formula XXII.
- the reaction can be carried out by adding a hydrazine of Formula R 2 NHNH 2 to a solution of an oxalated indole of Formula XXI in a solvent or solvent mix such as ethanol/acetic acid in the presence of hydrochloric acid.
- the reaction can be carried out at an elevated temperature such as at reflux.
- step (2) is carried out using hydrazine
- the resulting pyrazolo[3,4-c]quinolin-4- one of Formula XXII where R 2 is hydrogen can be further elaborated using known synthetic methods.
- a pyrazolo[3,4-c]quinolin-4-one of Formula XXII where R 2 is hydrogen can be alkylated.
- the alkylation is conveniently carried out by treating a solution of a pyrazolo[3,4-c]quinolin-4-one of Formula XXII, where R 2 is hydrogen, with a base such as sodium ethoxide followed by the addition of an alkyl halide.
- the reaction can be run in a suitable solvent such as ethanol and can be carried out at an elevated temperature, for example, the reflux temperature of the solvent, or at ambient temperature.
- a pyrazolo[3,4-c]quinolin-4-one of Formula XXII where R 2 is hydrogen can undergo a Buchwald animation with an aryl halide or heteroaryl halide.
- Numerous alkyl halides, aryl halides, and heteroaryl halides are commercially available; others can be prepared using known synthetic methods.
- Step (2) can also be carried out using a hydrazine that will install a removable group at R 2 .
- hydrazines include benzylhydrazine and tert- butylhydrazine.
- the group can be removed using conventional methods to provide a compound in which R 2 is hydrogen. The compound may then be further elaborated using the methods described above.
- step (3) of Reaction Scheme I an aldehyde group is installed on a pyrazolo[3,4- c]quinolin-4-one of Formula XXII to provide apyrazolo[3,4-c]quinolin-4-one of Formula Ilia, which is a subgenus of Formula III.
- the reaction can be carried out by deprotonating a pyrazolo[3,4-c]quinolin-4-one of Formula XXII with 2 equivalents of n-butyl lithium followed by treatment with N,iV-dimethylformarnide (DMF) and quenching with hydrochloric acid.
- the reaction can be carried out at an elevated temperature such as 50 0 C in a suitable solvent such as tetrahydrofuran.
- step (4) of Reaction Scheme I apyrazolo[3,4-c]quinolin-4-one of Formula HIa undergoes further elaboration using conventional synthetic methods to provide a pyrazolo[3,4-c]quinolin-4-one of Formula IHb, which is a subgenus of Formula III.
- the aldehyde can be reacted with several different classes of nucleophiles such as phosphonium ylides (Wittig olefination) or phosphonates (Horner Wadsworth olefmation) to provide alkenes; amines using reductive amination to provide secondary or tertiary amines; and Grignard reagents or lithiated alkynes or alkenes to provide alcohols which may then be oxidized to provide ketones.
- nucleophiles such as phosphonium ylides (Wittig olefination) or phosphonates (Horner Wadsworth olefmation) to provide alkenes
- amines using reductive amination to provide secondary or tertiary amines
- Grignard reagents or lithiated alkynes or alkenes to provide alcohols which may then be oxidized to provide ketones.
- the olefin may
- the aldehyde can be reduced to the alcohol using known methods, for example, treating a solution of the aldehyde with sodium borohydride.
- the alcohol can then be converted to a halide or an oxygen based leaving group such as a triflate, mesylate, or tosylate using conventional methods.
- the halide or oxygen based leaving group can then be reacted with a variety of nucleophiles.
- a pyrazolo[3,4-c]quinolin-4-one of Formula IHb is chlorinated to provide to provide a 4-chloropyrazolo[3,4-c]quinoline of Formula IHc, which is a subgenus of Formula III.
- the reaction can be carried out by combining a pyrazolo[3,4-c]quinolin-4-one of Formula IHb with phosphorous oxychloride and heating.
- a 4-chloropyrazolo[3,4-c]quinoline of Formula IHc undergoes further elaboration using conventional synthetic methods to provide a pyrazolo[3,4-c]quinoline of Formula IHd 5 which is a subgenus of Formula III.
- the chloro group can be displaced with nucleophiles such as alkoxides, phenoxides, or amines to provide compounds of Formula IHd wherein Rc i is alkoxy, unsubstituted or substituted phenoxy, amino, alkylamino, dialkylamino, benzylamino, or cyclic amino.
- a 4-chloropyrazolo[3,4-c]qui ⁇ oline of Formula IHc can undergo a palladium-catalyzed coupling reaction such as a Suzuki coupling or Sonogoshira coupling with a boronic acid of Formula RD-B(OH) 2 , an anhydride thereof, or a boronic ester of Formula Ro-B(O-alkyl)2 where R D is phenyl, substituted phenyl, or heteroaryl to provide compounds of Formula IHd wherein Rc i is phenyl, substituted phenyl, or heteroaryl.
- a palladium-catalyzed coupling reaction such as a Suzuki coupling or Sonogoshira coupling with a boronic acid of Formula RD-B(OH) 2 , an anhydride thereof, or a boronic ester of Formula Ro-B(O-alkyl)2 where R D is phenyl, substituted phenyl, or heteroaryl to
- compounds of the invention can be prepared according to Reaction Scheme II, where R, Ri, R 2 , R 3 , n and m are as defined above and Ra is a subset of Rc.
- step (1) of Reaction Scheme II an ethyl indole carboxylate of Formula XXIII is converted to an aldehyde substituted indole of Formula XXIV.
- the carboxylate group is reduced to the alcohol using conventional methods, for example, by treating a solution of an indole of Formula XXIII with lithium aluminum hydride.
- the alcohol group is then oxidized to the aldehyde using conventional methods, for example, by treating a solution of the alcohol substituted indole with sulfur trioxide pyridine, dimethyl sulfoxide (DMSO), and triethylamine.
- DMSO dimethyl sulfoxide
- step (2) of Reaction Scheme II an aldehyde substituted indole of Formula XXIV is further elaborated using the methods described in step (4) of Reaction Scheme I to provide a substituted indole of Formula XXV.
- step (3) of Reaction Scheme H 5 a substituted indole of Formula XXV is acylated using the method described in step (1) of Reaction Scheme I to provide an oxalated indole of Formula XXVI.
- step (4) of Reaction Scheme II an oxalated indole of Formula XXVI is rearranged to a pyrazolo[3,4-c]quinolin-4-one of Formula IHb using the method described in step (2) of Reaction Scheme I.
- Steps (5) and (6) of Reaction Scheme II are carried out using the methods of steps (5) and (6) of Reaction Scheme I.
- compounds of the invention can also be prepared according to Reaction Scheme III, where n is defined as above and R 3 , Ri a , R 2a , and R 3a are subsets of R, Ri, R 2 , and R 3 as defined above that do not include those substituents which one skilled in the art would recognize as being susceptible to oxidation in step (5). These susceptible substituents include -S- or heteroaryl groups. Acetals of Formula XXVII are reported in the literature and can be prepared using known synthetic methods, Royals, E. E., Robinson, A. G. Ill, J Am. Chem. Soc, 78, 4161 (1956).
- a ketone of Formula CH 3 C(O)Ri 3 can be condensed with ethyl diethoxyacetate under Claisen condensation conditions to provide an acetal of Formula XXVII.
- the reaction can be carried out by adding sodium tert-butoxide to a solution of ethyl diethoxyacetate and the ketone of Formula CH 3 C(O)Ri 8 in ethanol and heating the reaction at reflux.
- Numerous ketones of Formula CH 3 C(O)R 13 are commercially available. Others can be readily prepared using known synthetic methods.
- step (1) of Reaction Scheme III an acetal of Formula XXVII is reacted with a hydrazine of Formula R 23 -NH-NH 2 to provide a pyrazole of Formula XXVIII.
- the reaction can be carried out by slowly adding the hydrazine to a solution of an acetal of Formula XXVII in a suitable solvent such as ethanol. The reaction can be run at ambient temperature.
- step (2) of Reaction Scheme III the acetal in the pyrazole of Formula XXVIII is converted to an aldehyde under acidic conditions.
- the reaction can be carried out by treating the acetal-substituted pyrazole of Formula XXVIII with hydrochloric acid in a suitable solvent such as tetrahydrofuran.
- the reaction can be carried out at ambient temperature to provide an aldehyde-substituted pyrazole of Formula XXIX.
- a pyrazole of Formula XXIX is brominated to provide a bromo-substituted pyrazole of Formula XXX.
- the bromination can be carried out by adding bromine to a solution of the aldehyde-substituted pyrazole of Formula XXIX and potassium acetate in acetic acid. The reaction can be carried out at ambient temperature.
- a bromo-substituted pyrazole of Formula XXX undergoes a transition-metal catalyzed cross coupling reaction with a reagent of Formula XXXI.
- Reagents of Formula XXXI where M is, for example, -B(OH) 2 , -B(O-alkyl) 2 , -Sn(alkyl) 3 , and -Zn-Halide, are known to undergo coupling reactions.
- Several reagents of Formula XXXI are commercially available; others can be prepared using known synthetic methods.
- tert-butoxycarbonyl (Boc)-protected anilines undergo directed ortho metalation in the presence of butyllithium reagents.
- the resulting organolithium intermediate reacts with electrophiles such as B(O-alkyl) 3 and ClSn(alkyl) 3 to provide compounds of Formula XXXI, where M is -B(O-alkyl) 2 or -B(OH) 2 and -Sn(alkyl) 3 , respectively, after removal of the Boc protecting group.
- a Suzuki coupling reaction is conveniently carried out by heating a mixture of the bromo-substituted pyrazole of Formula XXX, palladium (II) acetate, triphenylphosphine, and a boron reagent of Formula XXXI, where M is -B(OH) 2 or -B(O-alkyl) 2 , in the presence of a base such as sodium carbonate.
- the reaction is carried out in a suitable solvent or solvent mixture such as n-propanol: water and can be heated at an elevated temperature such as 100 °C. Under these reaction conditions, intramolecular condensation of the amine with the aldehyde group takes place to form a pyrazolo[3,4- c]quinoline of Formula IHe 5 which is a subgenus of Formula III.
- a pyrazolo[3,4-c]quinoline of Formula IHe is oxidized to provide a pyrazolo[3,4-c]quinoline-5iV-oxide of Formula XXXII using a conventional oxidizing agent capable of forming TV-oxides.
- the reaction can be carried out by adding 3-chloroperoxybenzoic acid to a solution of a compound of Formula IHe in a solvent such as dichloromethane or chloroform. The reaction can be carried out at ambient temperature.
- step (6) of Reaction Scheme III a pyrazolo[3,4-c]quinoline-5iV-oxide of Formula XXXII is chlorinated to provide to provide a 4-chloropyrazolo[3,4-c]quinoline of Formula HIf, which is a subgenus of Formula III.
- the reaction can be carried out as described in step (5) of Reaction Scheme I.
- step (7) of Reaction Scheme III a 4-chloropyrazolo[3,4-c]quinoline of Formula IHf undergoes further elaboration using conventional synthetic methods, as described in step (6) of Reaction Scheme I, to provide a pyrazolo[3,4-c]quinoline of Formula IHg, which is a subgenus of Formula III.
- compounds of the invention can be prepared according to Reaction Scheme IV, wherein Rj 3 R 2 , and Rci, are as defined above; R is selected from the group consisting of hydrogen, halogen, alkyl, alkenyl, haloalkyl, and dialkylamino; R 3b and R 3c are defined below, and R 43 is methyl or benzyl.
- Steps (1) through (6) of Reaction Scheme IV can be carried out as described for steps (1) through (6) of Reaction Scheme I or as described for steps (1) through (6) of Reaction Scheme II.
- Some benzyloxy-substituted indoles and methoxy-substituted indoles of Formula XXa are known; others can be prepared using known synthetic methods.
- step (7) of Reaction Scheme IV the benzyl or methyl group of pyrazolo[3,4- c]quinoline of Formula HIh, which is a subgenus of Formula III, is cleaved using conventional methods to provide a pyrazolo[3,4-c]quinolinol of Formula IHi, which is a subgenus of Formula III.
- Cleavage of the benzyl group can be carried out on a Parr apparatus under hydrogenolysis conditions using a suitable heterogeneous catalyst such as palladium on carbon in a solvent such as ethanol.
- the reaction can be carried out by transfer hydrogenation in the presence of a suitable hydrogenation catalyst.
- the transfer hydrogenation can be carried out by adding ammonium formate to a solution of a pyrazolo[3,4-c]quinoline of Formula IHh in a suitable solvent such as ethanol in the presence of a catalyst such as palladium on carbon.
- the reaction can be carried out at an elevated temperature, for example, the reflux temperature of the solvent.
- Demethylation can be carried out by treating a pyrazolo[3,4-c]quinoline of Formula IHh with a solution of boron tribromide in a suitable solvent such as dichloromethane.
- the reaction can be carried out at a sub-ambient temperature such as 0 °C.
- the demethylation can be carried out by heating a pyrazolo[3,4-c]quinoline of Formula IHh with anhydrous pyridinium chloride at an elevated temperature, such as 210 0 C.
- step (8a) of Reaction Scheme IV the hydroxy group of a pyrazolo[3,4- c]quinolinol of Formula IHi is activated by conversion to a trifluromethanesulfonate (triflate) group.
- the reaction can be carried out by treating a pyrazolo[3,4-c]quinolinol of Formula HIi with iV-phenyl-bis(trifluoromethanesulfonimide) in the presence of a tertiary amine such as triethylamine.
- the reaction can be carried out at ambient temperature in a suitable solvent such as DMF.
- the activation in step (8a) may also be accomplished by converting the hydroxy group to another good leaving group.
- Step (9) of Reaction Scheme IV can be carried out using known palladium- catalyzed coupling reactions such as the Suzuki coupling, Heck reaction, the Stille coupling, and the Sonogashira coupling.
- a triflate-substituted pyrazolo[3,4- c]quinoline of Formula XXXIII undergoes Suzuki coupling with a boronic acid of Formula R 3b -B(OH) 2 , an anhydride thereof, or a boronic acid ester of Formula R 3 b-B(O-alkyl) 2 ; wherein R 3 b is -R 4 ⁇ -X e -R4, -Xf-Y-R 4 , or -Xf-R 5 ; where X e is alkenylene; X f is arylene, heteroarylene, and alkenylene interrupted or terminated by arylene or heteroarylene; R ⁇ is aryl or heteroaryl where the aryl or heteroaryl groups can be un
- the coupling can be carried out by combining a triflate-substituted pyrazolo[3,4- cjquinoline of Formula XXXIII with a boronic acid or an ester or anhydride thereof in the presence of palladium (II) acetate, triphenylphosphine, and a base such as aqueous sodium carbonate in a suitable solvent such as n-propanol.
- the reaction can be carried out at an elevated temperature, for example, at the reflux temperature.
- Numerous boronic acids of Formula R 3b -B(OH)2, anhydrides thereof, and boronic acid esters of Formula R 3b -B(O- alkyl) 2 are commercially available; others can be readily prepared using known synthetic methods.
- the Heck reaction can be used in step (9) of Reaction Scheme IV to provide compounds of Formula IHk, wherein R 3b is -X e -R tb or -X e -Y-R 4 , wherein X e , Y, R 4 , and R 4b are as defined above.
- Several of these vinyl-substituted compounds are commercially available; others can be prepared by known methods.
- the reaction can be carried out by combining a triflate-substituted pyrazolo[3,4-c]quinoline of Formula XXXIII and the vinyl-substituted compound in the presence of palladium (II) acetate, triphenylphosphine or tri- ⁇ rt/zo-tolylphosphine, and a base such as triethylamine in a suitable solvent such as acetonitrile or toluene.
- the reaction can be carried out at an elevated temperature such as 100-120 0 C under an inert atmosphere.
- step (8b) of Reaction Scheme IV a pyrazolo[3,4-c]quinolinol of Formula IHi is converted to apyrazolo[3,4-c]quinoline of Formula IHj, wherein R 30 is -0-R 4 , -0-X-R 4 , -0-X-Y-R 4 , or -O-X-R5, and X, Y 5 R 4 , and R 5 are as defined above, using a Williamson- type ether synthesis.
- the reaction can be effected by treating a pyrazolo[3,4-c]quinolinol of Formula IHi with an aryl, alkyl, or arylalkylenyl halide of Formula Halide-R ⁇ Halide- alkylene-R ⁇ Halide-alkylene-Y-R 4 , or Halide-alkylene-R 5 in the presence of a base.
- alkyl, arylalkylenyl, and aryl halides of these formulas are commercially available, including substituted benzyl bromides and chlorides, substituted or unsubstituted alkyl or arylalkylenyl bromides and chlorides, bromo-substituted ketones, esters, and heterocycles, and substituted fluorobenzenes.
- Other halides of these formulas can be prepared using conventional synthetic methods.
- the reaction can be carried out by combining an alkyl, arylalkylenyl, or aryl halide with a pyrazolo[3,4-c]quinolinol of Formula IHi in a solvent such as DMF or ⁇ iV-dimethylacetaniide in the presence of a suitable base such as cesium carbonate.
- a suitable base such as cesium carbonate.
- catalytic tetrabutylammonium bromide can be added.
- the reaction can be carried out at ambient temperature or at an elevated temperature, for example 50 °C or 85 °C, depending on the reactivity of the halide reagent.
- step (8b) may be carried out using the Ullmann ether synthesis, in which an alkali metal aryloxide prepared from a pyrazolo[3,4-c]quinolinol of Formula IHi reacts with an aryl halide in the presence of copper salts, to provide a pyrazolo[3,4- cjquinoline of Formula IHj, where R 3c is -0-R 4I3 , -0-Xj-R 4 , or -0-X j -Y-R 4 , wherein X j is an arylene or heteroarylene and R 4b is as defined above.
- Numerous substituted and unsubstituted aryl halides are commercially available; others can be prepared using conventional methods.
- steps (7) through (9) and (7) through (8b) can also be used to install R 3 b or R 3c groups at an earlier stage in the synthetic pathway.
- compounds of the invention can be prepared according to Reaction Scheme V, where Rb, Rib, R 2b , Rc 2 are subsets of R, Ri, R 2 , and Rc as defined above that do not include those substituents which would be susceptible to reduction under the acidic hydrogenation conditions of the reaction and n is as defined above.
- a pyrazolo[3,4-c]quinoline of Formula Him is reduced to provide a 6,7,8,9-tetrahydro-2H-pyrazolo[3,4-c]quinoline of Formula IVa, which is a subgenus of Formula IV.
- the reaction may be carried out under heterogeneous hydrogenation conditions by adding platinum (IV) oxide to a solution or suspension of a pyrazolo[3,4-c]quinoline of Formula Him in a suitable solvent such as trifluoroacetic acid and placing the reaction under hydrogen pressure.
- Pyrazolo[3,4-c]naphthyridines of the invention can be prepared by using an azaindole as the starting material in Reaction Schemes I, II, and IV. Azaindoles are known compounds. Some are commercially available and others can be prepared using known synthetic methods. Alternatively, pyrazolo[3,4-c]naphthyridines of the invention can be prepared by using an aminopyridine boronic acid in Reaction Scheme III. Aminopyridine boronic acids can be prepared using known methods, for example, by directed ortho metalation of Boc-protected aminopyridines and subsequent electrophilic substitution. Alternatively, for some isomers, halogen-lithium exchange and subsequent electrophilic substitution can be used.
- halogen-lithium exchange can be carried out on a 2-bromopyridine that has a protected amino group in the 3 -position; subsequent electrophilic substitution with tributyltin chloride and deprotection of the amino group provides 3-amino-2-tri-n-butylstannylpyridine.
- 6,7,8,9-Tetrahydro-2H-pyrazolo[3,4-c]naphthyridines can be prepared by reducing pyrazolo[3,4-c]naphthyridines using the method of Reaction Scheme V.
- compounds of the invention can be prepared according to Reaction Scheme VI where R 2 and R 4 are as described above, Xj is -(CH 2 )J -4 -, Yi is selected from -C(O)-, -S(O) 2 -, -S(O) 2 -N(R 8 )-, and -C(O)-NH- where R 8 is as defined above, and Boc is f ⁇ rt-butoxycarbonyl.
- a pyrazolo[3,4-c]quinoline of Formula XXIIa is reacted with a Boc protected 4-(iodoalkyl)piperidine of Formula XXXIV to provide a pyrazolo[3,4-c]quinoline of Formula HIn 5 a subgenus of Formula III.
- the reaction can be carried out by first deprotonating a pyrazolo[3,4-c]quinoline of Formula XXIIa by treating a solution of a compound of Formula XXIIa in a suitable solvent such as tetrahydrofuran with «-butyllithium optionally in the presence of N. ⁇ N'iV'-tetramethylethylenediamine.
- the reaction can be run at a sub-ambient temperature such as 0 0 C.
- the resulting anion is then treated with a Boc protected 4-(iodoalkyl)piperidine of Formula XXXIV.
- the reaction can be run at a sub-ambient temperature such as -78 °C.
- Pyrazolo[3,4- c]quinolines of Formula XXIIa can be prepared as described in Reaction Scheme I.
- Boc protected 4-(iodoalkyl)piperidines of Formula XXXIV can be prepared using conventional synthetic methods.
- step (2) of Reaction Scheme VI the Boc group on a pyrazolo[3,4-c]quinoline of Formula IHn is removed under acidic conditions to provide a pyrazolo[3,4-c]quinoline of Formula HIo, a subgenus of Formula III.
- the reaction can be carried out by combining a compound of Formula IHn with concentrated hydrochloric acid and stirring the resulting mixture at ambient temperature .
- step (3a) of Reaction Scheme VI a pyrazolo[3,4-c]quinoline of Formula HIo is converted into an amide, sulfonamide, sulfamide, or urea of Formula HIp, a subgenus of Formula III using conventional methods.
- a compound of Formula UIo or a salt thereof can react with an acid chloride of Formula R 4 C(O)Cl to provide a compound of Formula IHp in which Y 1 is - C(O)-.
- a compound of Formula IHo can react with sulfonyl chloride of Formula R 4 S(O) 2 Cl or a sulfonic anhydride of Formula (R 4 S(O) 2 ⁇ O to provide a compound of Formula Hip in which Yj is -S(O) 2 -.
- the reaction can be carried out by adding the acid chloride of Formula R 4 C(O)Cl, sulfonyl chloride of Formula R 4 S(O) 2 Cl, or sulfonic anhydride of Formula (R 4 S(O) ⁇ 2 O to a solution or suspension of a compound of Formula IHo in a suitable solvent such as chloroform, dichloromethane, TV, iV-dimethylacetamide (DMA), or N, N- dimethylformamide (DMF).
- a base such as triethylamine or N 1 N- diisopropylethylamine can be added.
- the reaction can be carried out at ambient temperature or at a sub-ambient temperature such as O 0 C.
- a base such as triethylamine or N, JV-diisopropylethylamine can be added.
- the reaction can be carried out at ambient temperature or a sub-ambient temperature such as 0 0 C.
- a compound of Formula IHo can be treated with a carbamoyl chloride of Formula Cl-C(O)-heterocyclyl, wherein heterocyclyl is attached at a nitrogen atom, to provide a compound of Formula IHp, wherein Yi is -C(O)- and R 4 is heterocyclyl attached at a nitrogen atom.
- Sulfamides of Formula IHp where Y 1 is -S(O) 2 -N(R 8 )-, can be prepared by reacting a compound or salt of Formula IHo with sulfuryl chloride to generate a sulfamoyl chloride in situ, and then reacting the sulfamoyl chloride with an amine of Formula HN(R 8 )R 4 .
- sulfamides of Formula Hip can be prepared by reacting a compound of Formula IHo with a sulfamoyl chloride of Formula R 4 (Rg)N-S(O) 2 Cl.
- a pyrazolo[3,4-c]quinoline of Formula HIo undergoes reductive alkylation to provide a pyrazolo[3,4-c]quinoline of Formula HIq. a subgenus of Formula III.
- the alkylation can be carried out in two parts by (i) adding an aldehyde or ketone to a solution of a compound of Formula IHo or a salt thereof in a suitable solvent such as DMF, THF, or methanol in the presence of a base such as N,N- diisopropylethylamine.
- a suitable reducing agent such as the borane-pyridine complex. Both part (i) and part (ii) can be carried out at ambient temperature.
- compounds of the invention can be prepared according to Reaction Scheme VII where R 2 , R 4 , Y 1 , and Boc are as defined above.
- step (1) of Reaction Scheme VII ethyl lH-indol-2-ylacetate is reacted with ethyl chlorooxoacetate to provide ethyl (2-ethoxycarbonylmethyl-l/f-indol-3- yl)oxoacetate.
- the reaction can be carried out using the general method described in step (1) of Reaction Scheme I. Ethyl lH-indol-2-ylacetate is known.
- step (2) of Reaction Scheme VII ethyl (2-ethoxycarbonylmethyl-lH-indol-3- yl)oxoacetate is rearranged to provide a pyrazolo[3,4-c]quinoline of Formula IUr, a subgenus of Formula III.
- the reaction can be carried out using the general method described in step (2) of Reaction Scheme I.
- step (3) of Reaction Scheme VII the ester group on a pyrazolo[3,4-c]quinoline of Formula IHr is hydrolyzed to provide a pyrazolo[3,4-c]quinoline of Formula Ills, a subgenus of Formula III.
- the reaction can be carried out by treating a solution of a compound of Formula IHr in ethanol with a base such as aqueous sodium hydroxide. The reaction can be carried out at ambient temperature,
- a pyrazolo[3,4-c]quinoline of Formula Ills is coupled with N-Boc piperazine to provide a pyrazolo[3,4-c]quinoline of Formula Hit, a subgenus of Formula III.
- the reaction can be carried out by treating a mixture of a compound of Formula Ills and N-Boc piperazine in a suitable solvent such as DMF with N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride and 1- hydroxybenzotriazole.
- the reaction can be carried out at ambient temperature.
- step (5) of Reaction Scheme VII the Boc group on a pyrazolo[3,4-c]quinoline of Formula lilt is removed to provide a pyrazolo[3,4-c]quinoline of Formula IHu 5 a subgenus of Formula III.
- the reaction can be carried out by combining a compound of Formula Hit with ethanol and hydrochloric acid and heating the resulting mixture at an elevated temperature such as 60 °C.
- step (6a) of Reaction Scheme VII a pyrazolo[3,4-c]quinoline of Formula HIu is converted into an amide, sulfonamide, sulfamide, or urea of Formula IHv, a subgenus of Formula III using conventional methods as described in step (3a) of Reaction Scheme VI.
- step (6b) of Reaction Scheme VII a pyrazolo[3,4-c]quinoline of Formula IHu undergoes reductive alkylation to provide a pyrazolo[3,4-c]quinoline of Formula HIw, a subgenus of Formula III.
- the alkylation can be carried out as described in step (3b) of Reaction Scheme VI.
- compounds of the invention can be prepared using the synthetic methods described in the EXAMPLES below.
- Pharmaceutical Compositions and Biological Activity are described in the EXAMPLES below.
- compositions of the invention contain a therapeutically effective amount of a compound or salt of the invention as described above in combination with a pharmaceutically acceptable carrier.
- a therapeutically effective amount and “effective amount” mean an amount of the compound or salt sufficient to induce a therapeutic or prophylactic effect, such as cytokine inhibition and immunomodulation.
- the exact amount of active compound or salt used in a pharmaceutical composition of the invention will vary according to factors known to those of skill in the art, such as the physical and chemical nature of the compound or salt, the nature of the carrier, and the intended dosing regimen.
- compositions of the invention will contain sufficient active ingredient or prodrug to provide a dose of about 100 nanograms per kilogram (ng/kg) to about 50 milligrams per kilogram (mg/kg), preferably about 10 micrograms per kilogram ( ⁇ g/kg) to about 5 mg/kg, of the compound or salt to the subject.
- the method includes administering sufficient compound to provide a dose of from about 0.1 mg/m 2 to about 2.0 mg/ m 2 to the subject, for example, a dose of from about 0.4 mg/m 2 to about 1.2 mg/m 2 .
- dosage forms may be used, such as oral preparations in the forms of tablets, lozenges, capsules, fine granules, granules, powders, syrups, dry syrups, or parenteral preparations or formulations, for example in the forms of injections, suppositories, eye drops, eye ointments, ear drops, nasal drops, and inhalations such as aerosol formulations, or dermal preparations, for example, creams, ointments, transdermal patches, transmucosal patches and the like.
- These dosage forms can be prepared with conventional pharmaceutically acceptable carriers and additives using conventional methods, which generally include the step of bringing the active ingredient into association with the carrier.
- the compounds or salts of the invention can be administered as the single therapeutic agent in the treatment regimen, or the compounds or salts of the invention may be administered in combination with one another or with other active agents, including additional immune response modifiers, antivirals, antibiotics, antibodies, proteins, peptides, oligonucleotides, etc.
- Compounds or salts of the invention can inhibit the production of certain cytokines in experiments performed according to the tests set forth below. These results indicate that the compounds or salts are useful as immune response modifiers that can modulate the immune response in a number of different ways, rendering them useful in the treatment of a variety of disorders .
- Cytokines whose production can be inhibited by the administration of compounds or salts of the invention include tumor necrosis factor- ⁇ (TNF- ⁇ ) and IL-I .
- TNF- ⁇ tumor necrosis factor- ⁇
- IL-I IL-I .
- the invention provides a method of inhibiting TNF- ⁇ biosynthesis in an animal comprising administering an effective amount of a compound or salt or composition of the invention to the animal.
- the animal to which the compound or salt or composition is administered for inhibition of TNF- ⁇ biosynthesis may have a disease as described infra, for example an autoimmune disease, and administration of the compound or salt may provide therapeutic treatment.
- the compound or salt or composition may be administered to the animal prior to the animal acquiring the disease so that administration of the compound or salt may provide a prophylactic treatment.
- the compound or salt or composition may be administered alone or in combination with one or more active components as in, for example, a vaccine adjuvant.
- the compound or salt and other component or components may be administered separately; together but independently such as in a solution; or together and associated with one another such as (a) covalently linked or (b) non-covalently associated, e.g., in a colloidal suspension.
- Conditions for which IRMs identified herein may be used as treatments include, but are not limited to: (a) chronic inflammatory diseases such as, for example, rheumatic arthritis and osteoarthritis;
- autoimmune diseases such as, for example, autoimmune hemic diseases (e.g., hemolytic anemia, anaplastic anemia, idiopathic thrombocythemis), autoimmune intestinal diseases (e.g., ulcerative colitis, Crohn's disease), autoimmune corneitis (e.g., keratoconjunctivitis sicca, spring catarrh), endocrine ophthalmopathy, Graves disease, sarcoid granuloma, multiple sclerosis, systemic erythematodes, multiple chondritis, pachydermia, active chronic hepatitis, myasthena gravis, psoriasis, interstitial pulmonary fibrosis, and the like; and (c) various conditions including allergic rhinitis, atopic dermatitis, contact dermatitis, asthma, sepsis, septic shock, diabetes, cancerous cachexia, HIV-infectious cachexia, and the like.
- therapeutic or prophylactic treatment of one or more of the above diseases or types of diseases by inhibiting cytokine biosynthesis in an animal can be provided by administering a therapeutically effective amount of a compound or salt of the invention to the animal.
- An amount of a compound or salt effective to inhibit cytokine biosynthesis is an amount sufficient to cause one or more cell types, such as monocytes, macrophages, dendritic cells and B-cells to produce an amount of one or more cytokines such as, for example, TNF- ⁇ or IL-I that is decreased (inhibited) over a level of such cytokines in the absence of the compound or salt.
- the precise amount will vary according to factors known in the art but is expected to be a dose of about 100 ng/kg to about 50 mg/kg, preferably about 10 ⁇ g/kg to about 5 mg/kg.
- the amount is expected to be a dose of, for example, from about 0.01 mg/m 2 to about 5.0 mg/m 2 , (computed according to the Dubois method as described above) although in some embodiments the inhibition of cytokine biosynthesis may be performed by administering a compound or salt in a dose outside this range.
- the solvent mixture 80/18/2 v/v/v chloroform/methanol/concentrated ammonium hydroxide (CMA) was used as the polar component of the eluent.
- CMA was mixed with chloroform in the indicated ratio.
- Acetyl chloride (0.7 mL, 9.8 mmol), acetic acid (1 mL), and ethylhydrazine (1.5 g, 9.8 mmol) were added sequentially to a suspension of methyl (l/f-indol-3-yl)gly oxalate (1.0 g, 4.9 mmol) in ethanol (25 mL).
- the reaction mixture was heated at reflux for 18 hours.
- the reaction mixture was cooled to ambient temperature and then concentrated under reduced pressure.
- the residue was combined with acetonitrile.
- a pink solid was isolated by filtration and then purified by automated flash chromatography (Biotage, eluted with a gradient of 0 - 30% CMA in chloroform) to provide a white solid.
- reaction mixture was warmed to ambient temperature and stirred for 1 hour. 1 ⁇ hydrochloric acid was added and the reaction mixture was stirred for 1 hour.
- the reaction mixture was concentrated under reduced pressure to about half the original volume and then diluted with water and extracted with dichloromethane. The combined extracts were dried over sodium sulfate, filtered, and then concentrated under reduced pressure to provide a dark yellow oil. The oil was combined with acetonitrile and stirred for 20 minutes. A bright yellow solid was isolated by filtration to provide 4 g of 2-ethyl- 4-oxo-4,5-dihydro-2H-pyrazolo[3,4-c]quinoline-l-carbaldehyde as a yellow powder.
- the material from Part A was combined in a pressure vessel with a solution of ammonia in methanol (100 mL of 7 N). The vessel was sealed and heated at 150 0 C for 20 hours. The reaction mixture was absorbed onto silica and then purified by automated flash chromatography (Analogix, eluted with a gradient of 0 - 30% CMA in chloroform) to provide a white solid. This material was recrystallized from acetonitrile to provide 170 mg of (2-ethyl-4-methoxy-2H-pyrazolo[3,4-c]quinolin-l-yl)methanol as white crystals, mp 219-220 °C. Anal, calcd for C 14 H 15 N 3 O 2 : C, 65.36; H 5 5.88; N 5 16.33. Found: C, 65.22; H, 5.85; N 5 16.57.
- the resulting reaction mixture was poured into a 1 : 1 mixture of saturated aqueous ammonium hydroxide and ice (200 mL). A solid formed that was isolated by filtration. The filtrate was extracted with dichloromethane. The isolated solid was dissolved in dichloromethane and methanol, combined with the organic extracts, dried over sodium sulfate, filtered, and concentrated under reduced pressure to afford a yellow oily solid.
- the yellow oily solid was purified by automated flash chromatography (AnaLogix, eluted with a gradient of 0 - 50% CMA in chloroform) to provide material that was stirred in acetonitrile and filtered to provide 130 mg of2-phenyl-l-(2-piperidin-4-ylethyl)-2,5-dihydro-4H-pyrazolo[3,4-c]quinolin-4-one as a white powder, mp 250-252 0 C.
- the resulting material was purified by automated flash chromatography (AnaLogix, eluted with a gradient of 0 - 60% CMA in chloroform) to provide an oily solid.
- the oily solid was stirred in acetonitrile.
- a solid was isolated by filtration to provide 40 mg of l-(l-hydroxy-2-piperidin-4-ylethyl)-2-phenyl-2,5-dihydro-4H- pyrazolo[3,4-c]quinolin-4-one as a tan powder, mp 258-261 °C.
- the reaction mixture was warmed to ambient temperature and stirred for 30 minutes before quenching with saturated aqueous ammonium chloride.
- the aqueous layer was extracted with chloroform.
- the combined organic extracts were dried over sodium sulfate, filtered, and concentrated under reduced pressure to afford a yellow oily solid.
- the oily solid was purified by automated flash chromatography (AnaLogix, eluted with a gradient of 0 - 30% CMA in chloroform) to provide a yellow solid.
- the solid was boiled in acetonitrile and filtered.
- the filtrate was adsorbed onto silica gel and purified by automated flash chromatography (AnaLogix, eluted with a gradient of 0 - 20% CMA in chloroform) to provide a white foam.
- the foam was stirred in acetonitrile and filtered to provide 213 mg of 1 -(2-cyclohexyl-l -hydroxyethyl)-2-phenyl-2,5-dihydro-4H-pyrazolo[3,4-c]quinolin-4- one as a white powder, mp 254-259 °C.
- Acetyl chloride (11.0 mL, 152 mmol), acetic acid (13 mL), and phenylhydrazine (15 mL, 152 mmol) were added sequentially to a suspension of ethyl (2-methyl-l//-indol- 3-yl)(oxo)acetate (17 g, 76 mmol) in ethanol (380 mL).
- the reaction mixture was heated at reflux for 18 hours. After cooling to ambient temperature a red solid was isolated by filtration.
- Part D The material from Part B was dissolved in ethanol (250 mL) and heated at reflux for 3 hours. The mixture was allowed to cool to ambient temperature and concentrated under reduced pressure. The red oil was passed through a layer of silica gel, eluting with chloroform. Concentration under reduced pressure afforded an orange oil that was crystallized from ethanol to afford 17.04 g of ethyl 4-(2-nitrophenyl)-3-oxobutanoate. Part D
- the material from Part D was dissolved in dichloromethane (280 mL). and cooled to 0 °C. Pyridine (6.9 mL, 85.7 mmol) and chloroethyloxalate (8.75 mL, 78.6 mmol) were added, and the reaction mixture was allowed to stir overnight, slowly warming to ambient temperature. 200 mL of water was added, and the aqueous layer was extracted with dichloromethane. The combined organic layers were dried over magnesium sulfate and then concentrated under reduced pressure.
- the reaction mixture was cooled to -78°C in an acetone-dry ice bath and tert-butyl 4-(2-iodoethyl)piperidine-l-carboxylate (2.3 g, 6.84 mmol) was added to the reaction.
- the reaction was warmed to ambient temperature over 30 minutes and stirred at that temperature for another 2 hours. Methanol was carefully added to quench the reaction and the solvent was evaporated to afford a dark oil.
- the oil was taken up in ethyl acetate and the organic layer was washed with aqueous saturated sodium bicarbonate. The organic layer was separated, dried (magnesium sulfate), filtered, and evaporated to afford a dark oil.
- the product was purified twice by automated flash chromatography (Combiflash Separation System, eluted with a gradient of 0-5% methanol in dichloromethane with 1% ammonium hydroxide) to provide tert-butyl 4-[2-(2-tert- butyl-4-oxo-4,5-dihydro-2H " -pyrazolo[3,4-c]quinolin-l-yl)-ethyl]-piperidine-l-carboxylate as a gray solid (0.17 g), mp 223-225°C; MS (ESI) m/z 453 (M+H).
- a pressure tube was charged with material from Part D (2.00 g, 4.38 mmol) and anhydrous DMF (30 mL). Nitrogen was bubbled through the resultant solution for several minutes, and palladium acetate (294 mg, 1.31 mmol) and sodium bicarbonate (924 mg, 11.0 mmol) were then added. The pressure tube was sealed and heated in a 150 °C oil bath for 22 hours. The pressure tube was then cooled to ambient temperature and opened, and the reaction mixture was diluted with ethyl acetate (100 mL) and filtered through CELITE filter aid. The filtrate was washed with water (2x50 mL) and brine, then dried over magnesium sulfate, filtered, and concentrated to afford a yellow oil.
- a pressure tube was charged with material from Part E (730 mg, 1.94 mmol) and trifluoroacetic acid (10 mL). The pressure tube was sealed and placed in a 70 °C oil bath for 72 hours. After cooling to ambient temperature, the pressure tube was opened, and the reaction mixture was diluted with water. The pH was adjusted to 7 by addition of saturated aqueous sodium carbonate, and the resultant aqueous phase was extracted with dichloromethane (3x75 mL). The combined organic layers were washed with brine, dried over magnesium sulfate, filtered, and concentrated to a tan solid.
- the material obtained in Part C (1.33 g, 4.00 mmol) was dissolved in 1,2- dimethoxyethane (15 mL) and water (7.5 mL). To the resulting solution was added potassium carbonate (1.82 g, 13.1 mmol), 2-aminophenylboronic acid hydrochloride (1.39 g, 8.0 mmol), and dichlorobis(triphenylphosphine)palladium(0) (140 mg, 0.2 mmol). The flask was evacuated and backfilled with nitrogen before stirring at reflux for 24 hours. The reaction was cooled to ambient temperature. The reaction was partitioned between water and methyl /-butyl ether. The layers were separated.
- the compounds were purified by preparative high performance liquid chromatography (prep HPLC) using a Waters FractionLynx automated purification system.
- the prep HPLC fractions were analyzed using a Waters LC/TOF-MS, and the appropriate fractions were centrifuge evaporated to provide the trifluoroacetate salt of the desired compound.
- Reversed phase preparative liquid chromatography was performed with non-linear gradient elution from 5-95% B where A is 0.05% trifluoroacetic acid/water and B is 0.05% trifluoroacetic acid/acetonitrile. Fractions were collected by mass-selective triggering.
- the table below shows the aldehyde used for each example, the structure of the resulting compound, and the observed accurate mass for the isolated trifluoroacetate salt.
- a reagent (1.1 eq.) from the table below was added to a test tube containing a solution of 2-phenyl- 1 -(2-piperidin-4-ylethyl)-2,5-dihydro-4H-pyrazolo[3 ,4-c]quinolin-4- one (37 mg, 0.10 mmol, 1.0 eq) and N,N-diisopropylethylamine (34 ⁇ L, 2 eq) in ⁇ iV- dimethylacetamide (1 mL). The reaction mixture was stirred overnight and then quenched with water (2 drops). The solvent was removed by vacuum centrifugation. The compounds were purified using the method described in Examples 30 - 36.
- the table below shows the reagent used for each example, the structure of the resulting compound, and the observed accurate mass for the isolated trifluoroacetate salt.
- Part B l-(Chloromethyl)-2,5-dihydro-2-phenyl-4H-pyrazolo[3,4-c]quinolin-4-one hydrochloride was prepared according to the general method of Part A of Examples 44 - 79 using l-(hydroxymethyl)-2,5-dihydro-2-phenyl-4//-pyrazolo[3 5 4-c]quinolin-4-one in lieu of 2-tert-bnty ⁇ - 1 -(hydroxymethyl)-2,5-dihydro-4/i-pyrazolo[3 ,4-c]quinolin-4-one.
- Part C l-(Chloromethyl)-2,5-dihydro-2-phenyl-4H-pyrazolo[3,4-c]quinolin-4-one hydrochloride was prepared according to the general method of Part A of Examples 44 - 79 using l-(hydroxymethyl)-2,5-dihydro-2-phenyl-4//-pyrazolo[3 5
- a test tube was charged sequentially with copper iodide (4 mg, 0.2 eq), L-proline (5 mg, 0.4 eq), a reagent (1.5 eq) from the table below, potassium carbonate (55 mg, 4 eq), and a solution of l-ethyl-2,5-dihydro-pyrazolo[3,4-c]quinolin-4-one (21 mg, 0.10 mmol, 1.0 eq) in dimethylsulfoxide (2 mL).
- the reaction mixture was purged with nitrogen and then heated at 100 0 C overnight.
- the reaction mixture was filtered and the filtrate was concentrated by vacuum centrifugation.
- the compounds were purified using the method described in Examples 30 - 36.
- the table below shows the reagent used for each example, the structure of the resulting compound, and the observed accurate mass for the isolated trifluoroacetate salt.
- the reaction was quenched with water (100 ⁇ L). The solvent was removed by vacuum centrifugation. The compounds were purified as described in Examples 30 - 36.
- the table below shows the aldehyde used for each example, the structure of the resulting compound, and the observed accurate mass for the isolated trifluoroacetate salt.
- TNF- ⁇ tumor necrosis factor ⁇
- PBMC peripheral blood mononuclear cells
- LPS bacterial lipopolysaccharide
- PBMCs peripheral blood mononuclear cells
- PBMCs peripheral blood mononuclear cells
- PBMCs are separated from whole blood by density gradient centrifugation using Ficoll-Paque Plus (Amersham Biosciences Piscataway, NJ).
- Whole blood is placed in Accuspin (Sigma) or LeucoSep (Greiner Bio- One, Inc., Longwood, FL) centrifuge frit tubes containing density gradient medium.
- the PBMC layer is collected and washed twice with DPBS or HBSS and re-suspended at 2.5 x 10 6 cells/mL in RPMI complete.
- the PBMC suspension is added to 96-well flat bottom sterile tissue culture plates.
- the compounds are solubilized in dimethyl sulfoxide (DMSO).
- DMSO dimethyl sulfoxide
- Controls include cell samples with DMSO only (no compound or LPS), cell samples stimulated with LPS (Fluorescein isothiocyanate-lipopolysaccharide from Escherichia coli 0111 :B4; Sigma,), cell samples with reference compounds 4-chloro-2-phenyl-l-(2-piperidin-4-ylethyl)-lH- imidazo[4,5-c]quinoline fumarate hydrate (Izumi, et al., "lH-Imidazo[4,5-c]qumoline Derivatives as Novel Potent TNF- ⁇ Suppressors: Synthesis and Structure- Activity Relationship of 1-, 2-, and 4-Substituted l/f-Imidazo[4,5-c]quinolines or 1H-Imidazo[4,5- ⁇ pyridines", Bioorganic
- test compound is added at 1.0 mM to the first well of a 384 well dosing plate and serial 3 fold dilutions are made for the 7 subsequent concentrations in 10 % DMSO (diluted in water). Each compound is in quadruplicate on each 384 well dosing plate in an 8-point dose response.
- Incubation 80 ⁇ L of the PBMC suspension is added to each well on a 384 well sterile tissue culture treated plate. Each compound solution (0.9 ⁇ l) is transferred from the dosing plate and is added to the 384 well plate containing PBMCs. After 20 minutes, LPS (0.01 ⁇ g/mL final concentration) is added. Final compound concentrations are 10 ⁇ M - 0.005 ⁇ M and the DMSO concentration is 0.1 %. The plates are covered with sterile plastic lids and incubated for 18 to 20 hours at 37°C in a 5% carbon dioxide atmosphere.
- MSD Multi-Spot® plates contain within each well capture antibodies for human TNF- ⁇ and human IL-10 that have been pre-coated on specific spots. Each well contains four spots: one human TNF- ⁇ capture antibody (MSD) spot, one human IL-10 capture antibody (MSD) spot, and two inactive bovine serum albumin spots.
- the human TNF- ⁇ and IL-10 capture and detection antibody pairs are from MesoScale Discovery. Standards consist of recombinant human TNF- ⁇ (MSD) and recombinant human IL-10 (MSD). Samples and separate standards are added at the time of analysis to each MSD plate.
- the cytokine-specific detection antibodies are labeled with the SULFO-TAG reagent (MSD) and the cytokine standards are added to the wells of the MSD plates.
- MSD SULFO-TAG reagent
- Supernatants from each tissue culture plate are transferred to the MSD plates and incubated for 4 hours at ambient temperature on a shaker, wells are washed with PBS, and MSD Read buffer is added to each well.
- Electrochemoluminescent levels are read using MSD' s SECTOR HTS READER. Results are expressed in pg/mL upon calculation with known cytokine standards on each plate.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Diabetes (AREA)
- Physical Education & Sports Medicine (AREA)
- Hematology (AREA)
- Immunology (AREA)
- Rheumatology (AREA)
- Pulmonology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Dermatology (AREA)
- Neurology (AREA)
- Otolaryngology (AREA)
- Ophthalmology & Optometry (AREA)
- Pain & Pain Management (AREA)
- Neurosurgery (AREA)
- Biomedical Technology (AREA)
- Obesity (AREA)
- Gastroenterology & Hepatology (AREA)
- Endocrinology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Emergency Medicine (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP06740248A EP1863813A2 (en) | 2005-04-01 | 2006-03-31 | Pyrazolo[3,4-c]quinolines, pyrazolo[3,4-c]naphthyridines, analogs thereof, and methods |
| US11/887,526 US8138173B2 (en) | 2005-04-01 | 2006-03-31 | Pyrazolo[3,4-C]quinolines, pyrazolo[3,4-C]naphthyridines, analogs thereof, and methods |
| JP2008504437A JP2008538119A (en) | 2005-04-01 | 2006-03-31 | Pyrazolo [3,4-c] quinolines, pyrazolo [3,4-c] naphthyridines, analogs thereof, and methods |
| AU2006232294A AU2006232294A1 (en) | 2005-04-01 | 2006-03-31 | Pyrazolo[3,4-c]quinolines, pyrazolo[3,4-c]naphthyridines, analogs thereof, and methods |
| CA002603063A CA2603063A1 (en) | 2005-04-01 | 2006-03-31 | Pyrazolo[3,4-c]quinolines, pyrazolo[3,4-c]naphthyridines, analogs thereof, and methods |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US66787905P | 2005-04-01 | 2005-04-01 | |
| US60/667,879 | 2005-04-01 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2006107771A2 true WO2006107771A2 (en) | 2006-10-12 |
| WO2006107771A3 WO2006107771A3 (en) | 2007-10-04 |
Family
ID=37073978
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2006/012031 WO2006107771A2 (en) | 2005-04-01 | 2006-03-31 | PYRAZOLO[3,4-c]QUINOLINES, PYRAZOLO[3,4-c]NAPHTHYRIDINES, ANALOGS THEREOF, AND METHODS |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US8138173B2 (en) |
| EP (1) | EP1863813A2 (en) |
| JP (1) | JP2008538119A (en) |
| AU (1) | AU2006232294A1 (en) |
| CA (1) | CA2603063A1 (en) |
| WO (1) | WO2006107771A2 (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7879849B2 (en) | 2003-10-03 | 2011-02-01 | 3M Innovative Properties Company | Pyrazolopyridines and analogs thereof |
| US7897597B2 (en) | 2003-08-27 | 2011-03-01 | 3M Innovative Properties Company | Aryloxy and arylalkyleneoxy substituted imidazoquinolines |
| US7897767B2 (en) | 2003-11-14 | 2011-03-01 | 3M Innovative Properties Company | Oxime substituted imidazoquinolines |
| US7906506B2 (en) | 2006-07-12 | 2011-03-15 | 3M Innovative Properties Company | Substituted chiral fused [1,2] imidazo [4,5-c] ring compounds and methods |
| US7915281B2 (en) | 2004-06-18 | 2011-03-29 | 3M Innovative Properties Company | Isoxazole, dihydroisoxazole, and oxadiazole substituted imidazo ring compounds and method |
| US7923429B2 (en) | 2003-09-05 | 2011-04-12 | 3M Innovative Properties Company | Treatment for CD5+ B cell lymphoma |
| US7943609B2 (en) | 2004-12-30 | 2011-05-17 | 3M Innovative Proprerties Company | Chiral fused [1,2]imidazo[4,5-C] ring compounds |
| US7943636B2 (en) | 2005-04-01 | 2011-05-17 | 3M Innovative Properties Company | 1-substituted pyrazolo (3,4-C) ring compounds as modulators of cytokine biosynthesis for the treatment of viral infections and neoplastic diseases |
| US7943610B2 (en) | 2005-04-01 | 2011-05-17 | 3M Innovative Properties Company | Pyrazolopyridine-1,4-diamines and analogs thereof |
| US7968563B2 (en) | 2005-02-11 | 2011-06-28 | 3M Innovative Properties Company | Oxime and hydroxylamine substituted imidazo[4,5-c] ring compounds and methods |
| CN102190622A (en) * | 2010-03-15 | 2011-09-21 | 李勤耕 | Water-soluble derivatives of edaravone, preparation method and application thereof |
| US8034938B2 (en) | 2004-12-30 | 2011-10-11 | 3M Innovative Properties Company | Substituted chiral fused [1,2]imidazo[4,5-c] ring compounds |
| US8546368B2 (en) | 2006-02-15 | 2013-10-01 | Abbvie Inc. | Pyrazoloquinolones are potent PARP inhibitors |
| US8598192B2 (en) | 2003-11-14 | 2013-12-03 | 3M Innovative Properties Company | Hydroxylamine substituted imidazoquinolines |
| US8673932B2 (en) | 2003-08-12 | 2014-03-18 | 3M Innovative Properties Company | Oxime substituted imidazo-containing compounds |
| US8691837B2 (en) | 2003-11-25 | 2014-04-08 | 3M Innovative Properties Company | Substituted imidazo ring systems and methods |
| CN103717597A (en) * | 2011-04-21 | 2014-04-09 | 原真股份有限公司 | Heterocyclic compounds as kinase inhibitors |
| US8735421B2 (en) | 2003-12-30 | 2014-05-27 | 3M Innovative Properties Company | Imidazoquinolinyl sulfonamides |
| US8802853B2 (en) | 2003-12-29 | 2014-08-12 | 3M Innovative Properties Company | Arylalkenyl and arylalkynyl substituted imidazoquinolines |
| US8871782B2 (en) | 2003-10-03 | 2014-10-28 | 3M Innovative Properties Company | Alkoxy substituted imidazoquinolines |
| US8962629B2 (en) | 2008-06-10 | 2015-02-24 | Abbvie Inc. | Tricyclic compounds |
| US9248127B2 (en) | 2005-02-04 | 2016-02-02 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| EP3085373A1 (en) | 2006-02-22 | 2016-10-26 | 3M Innovative Properties Company | Immune response modifier conjugates |
| EP3153180A1 (en) | 2011-06-03 | 2017-04-12 | 3M Innovative Properties Company | Heterobifunctional linkers with polyethylene glycol segments and immune response modifier conjugates made therefrom |
| WO2017134066A1 (en) | 2016-02-05 | 2017-08-10 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| US9879019B2 (en) | 2015-10-16 | 2018-01-30 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10000482B2 (en) | 2012-10-19 | 2018-06-19 | Origenis Gmbh | Kinase inhibitors |
| US10005772B2 (en) | 2006-12-22 | 2018-06-26 | 3M Innovative Properties Company | Immune response modifier compositions and methods |
| CN108774237A (en) * | 2009-12-01 | 2018-11-09 | Abbvie 公司 | New tricyclic compound |
| US10550126B2 (en) | 2015-10-16 | 2020-02-04 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-A]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11365198B2 (en) | 2015-10-16 | 2022-06-21 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11512092B2 (en) | 2015-10-16 | 2022-11-29 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11524964B2 (en) | 2015-10-16 | 2022-12-13 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| WO2023143356A1 (en) * | 2022-01-26 | 2023-08-03 | 勤浩医药(苏州)有限公司 | Methionine adenosyltransferase 2a inhibitor for treating mtap deletion-type cancer |
| US11773106B2 (en) | 2015-10-16 | 2023-10-03 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| WO2024080788A1 (en) * | 2022-10-13 | 2024-04-18 | 한미약품 주식회사 | Novel tricyclic derivative compound and uses thereof |
Families Citing this family (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6677347B2 (en) * | 2000-12-08 | 2004-01-13 | 3M Innovative Properties Company | Sulfonamido ether substituted imidazoquinolines |
| US20040265351A1 (en) | 2003-04-10 | 2004-12-30 | Miller Richard L. | Methods and compositions for enhancing immune response |
| WO2004096144A2 (en) * | 2003-04-28 | 2004-11-11 | 3M Innovative Properties Company | Compositions and methods for induction of opioid receptors |
| US20090075980A1 (en) * | 2003-10-03 | 2009-03-19 | Coley Pharmaceutical Group, Inc. | Pyrazolopyridines and Analogs Thereof |
| BRPI0414867A (en) | 2003-10-03 | 2006-11-21 | 3M Innovative Properties Co | pyrazolopyridines and their analogues |
| AU2005228150A1 (en) * | 2004-03-24 | 2005-10-13 | 3M Innovative Properties Company | Amide substituted imidazopyridines, imidazoquinolines, and imidazonaphthyridines |
| US8017779B2 (en) * | 2004-06-15 | 2011-09-13 | 3M Innovative Properties Company | Nitrogen containing heterocyclyl substituted imidazoquinolines and imidazonaphthyridines |
| US8026366B2 (en) * | 2004-06-18 | 2011-09-27 | 3M Innovative Properties Company | Aryloxy and arylalkyleneoxy substituted thiazoloquinolines and thiazolonaphthyridines |
| US8541438B2 (en) | 2004-06-18 | 2013-09-24 | 3M Innovative Properties Company | Substituted imidazoquinolines, imidazopyridines, and imidazonaphthyridines |
| US7897609B2 (en) * | 2004-06-18 | 2011-03-01 | 3M Innovative Properties Company | Aryl substituted imidazonaphthyridines |
| WO2006026760A2 (en) * | 2004-09-02 | 2006-03-09 | 3M Innovative Properties Company | 1-amino imidazo-containing compounds and methods |
| WO2006063072A2 (en) * | 2004-12-08 | 2006-06-15 | 3M Innovative Properties Company | Immunomodulatory compositions, combinations and methods |
| AU2006338521A1 (en) | 2005-02-09 | 2007-10-11 | Coley Pharmaceutical Group, Inc. | Oxime and hydroxylamine substituted thiazolo(4,5-c) ring compounds and methods |
| ES2475728T3 (en) | 2005-02-09 | 2014-07-11 | 3M Innovative Properties Company | Thiazoloquinolines and alkoxy substituted thiazolonaphthyridines |
| AU2006216997A1 (en) * | 2005-02-11 | 2006-08-31 | Coley Pharmaceutical Group, Inc. | Substituted imidazoquinolines and imidazonaphthyridines |
| EP1850849A2 (en) * | 2005-02-23 | 2007-11-07 | Coley Pharmaceutical Group, Inc. | Method of preferentially inducing the biosynthesis of interferon |
| JP2008531567A (en) * | 2005-02-23 | 2008-08-14 | コーリー ファーマシューティカル グループ,インコーポレイテッド | Hydroxyalkyl-substituted imidazoquinoline compounds and methods |
| JP2008543725A (en) * | 2005-02-23 | 2008-12-04 | コーリー ファーマシューティカル グループ,インコーポレイテッド | Hydroxyalkyl substituted imidazoquinolines |
| EA200800782A1 (en) * | 2005-09-09 | 2008-08-29 | Коли Фармасьютикал Груп, Инк. | AMIDA AND CARBAMATE DERIVATIVES N- {2- [4-AMINO-2- (ETOXIMETHYL) -1H-IMIDAZOLO [4,5-c] QUINOLIN-1-IL] -1,1-DIMETHYLETHYL} METHANE SULFONAMIDE AND METHODS |
| ZA200803029B (en) | 2005-09-09 | 2009-02-25 | Coley Pharm Group Inc | Amide and carbamate derivatives of alkyl substituted /V-[4-(4-amino-1H-imidazo[4,5-c] quinolin-1-yl)butyl] methane-sulfonamides and methods |
| ES2429170T3 (en) | 2005-11-04 | 2013-11-13 | 3M Innovative Properties Company | 1H-Imidazoquinolines substituted with hydroxyl and alkoxy and methods |
| WO2007106854A2 (en) * | 2006-03-15 | 2007-09-20 | Coley Pharmaceutical Group, Inc. | Hydroxy and alkoxy substituted 1h-imidazonaphthyridines and methods |
| WO2008030511A2 (en) * | 2006-09-06 | 2008-03-13 | Coley Pharmaceuticial Group, Inc. | Substituted 3,4,6,7-tetrahydro-5h, 1,2a,4a,8-tetraazacyclopenta[cd]phenalenes |
| MY150481A (en) * | 2008-03-03 | 2014-01-30 | Irm Llc | Compounds and compositions as tlr activity modulators |
| CA3033133C (en) | 2009-03-25 | 2021-11-09 | The Board Of Regents Of The University Of Texas System | Compositions for stimulation of mammalian innate immune resistance to pathogens |
| PT2606047T (en) | 2010-08-17 | 2017-04-07 | 3M Innovative Properties Co | Lipidated immune response modifier compound compositions, formulations, and methods |
| CA2838158C (en) | 2011-06-03 | 2019-07-16 | 3M Innovative Properties Company | Heterobifunctional linkers with polyethylene glycol segments and immune response modifier conjugates made therefrom |
| WO2016044839A2 (en) | 2014-09-19 | 2016-03-24 | The Board Of Regents Of The University Of Texas System | Compositions and methods for treating viral infections through stimulated innate immunity in combination with antiviral compounds |
| US10654860B2 (en) * | 2016-11-30 | 2020-05-19 | Bristol-Myers Squibb Company | Tricyclic rho kinase inhibitors |
| CA3086439A1 (en) | 2017-12-20 | 2019-06-27 | 3M Innovative Properties Company | Amide substitued imidazo[4,5-c]quinoline compounds with a branched chain linking group for use as an immune response modifier |
| CN116532109B (en) * | 2023-05-04 | 2024-05-28 | 济南大学 | Preparation method of supported catalyst, obtained product and application |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7544697B2 (en) * | 2003-10-03 | 2009-06-09 | Coley Pharmaceutical Group, Inc. | Pyrazolopyridines and analogs thereof |
-
2006
- 2006-03-31 JP JP2008504437A patent/JP2008538119A/en active Pending
- 2006-03-31 AU AU2006232294A patent/AU2006232294A1/en not_active Abandoned
- 2006-03-31 CA CA002603063A patent/CA2603063A1/en not_active Abandoned
- 2006-03-31 EP EP06740248A patent/EP1863813A2/en not_active Withdrawn
- 2006-03-31 US US11/887,526 patent/US8138173B2/en not_active Expired - Fee Related
- 2006-03-31 WO PCT/US2006/012031 patent/WO2006107771A2/en active Application Filing
Non-Patent Citations (1)
| Title |
|---|
| DATABASE CAPLUS [Online] MATAKA S., TAKAHASHI K., TASHIRO M.: 'Condensation reaction of 3,4-dibenzoyl-1-methyl-2,5-diphenylpyrrole and -1-phenylpyrazole with methylamine derivatives affording pyrrolo[3,4-c]pyridine and 2H-pyrazolo-[3,4-c]- and [4,3-c]pyridines', XP003017992 Database accession no. (1982:52223) & JOURNAL OF HETEROCYCLIC CHEMISTRY vol. 18, no. 6, 1981, pages 1073 - 1075 * |
Cited By (78)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8673932B2 (en) | 2003-08-12 | 2014-03-18 | 3M Innovative Properties Company | Oxime substituted imidazo-containing compounds |
| US7897597B2 (en) | 2003-08-27 | 2011-03-01 | 3M Innovative Properties Company | Aryloxy and arylalkyleneoxy substituted imidazoquinolines |
| US7923429B2 (en) | 2003-09-05 | 2011-04-12 | 3M Innovative Properties Company | Treatment for CD5+ B cell lymphoma |
| US8871782B2 (en) | 2003-10-03 | 2014-10-28 | 3M Innovative Properties Company | Alkoxy substituted imidazoquinolines |
| US7879849B2 (en) | 2003-10-03 | 2011-02-01 | 3M Innovative Properties Company | Pyrazolopyridines and analogs thereof |
| US8598192B2 (en) | 2003-11-14 | 2013-12-03 | 3M Innovative Properties Company | Hydroxylamine substituted imidazoquinolines |
| US7897767B2 (en) | 2003-11-14 | 2011-03-01 | 3M Innovative Properties Company | Oxime substituted imidazoquinolines |
| US8691837B2 (en) | 2003-11-25 | 2014-04-08 | 3M Innovative Properties Company | Substituted imidazo ring systems and methods |
| US8802853B2 (en) | 2003-12-29 | 2014-08-12 | 3M Innovative Properties Company | Arylalkenyl and arylalkynyl substituted imidazoquinolines |
| US8735421B2 (en) | 2003-12-30 | 2014-05-27 | 3M Innovative Properties Company | Imidazoquinolinyl sulfonamides |
| US7915281B2 (en) | 2004-06-18 | 2011-03-29 | 3M Innovative Properties Company | Isoxazole, dihydroisoxazole, and oxadiazole substituted imidazo ring compounds and method |
| US7943609B2 (en) | 2004-12-30 | 2011-05-17 | 3M Innovative Proprerties Company | Chiral fused [1,2]imidazo[4,5-C] ring compounds |
| US8034938B2 (en) | 2004-12-30 | 2011-10-11 | 3M Innovative Properties Company | Substituted chiral fused [1,2]imidazo[4,5-c] ring compounds |
| US9248127B2 (en) | 2005-02-04 | 2016-02-02 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| US10071156B2 (en) | 2005-02-04 | 2018-09-11 | 3M Innovative Properties Company | Aqueous gel formulations containing immune response modifiers |
| US7968563B2 (en) | 2005-02-11 | 2011-06-28 | 3M Innovative Properties Company | Oxime and hydroxylamine substituted imidazo[4,5-c] ring compounds and methods |
| US7943610B2 (en) | 2005-04-01 | 2011-05-17 | 3M Innovative Properties Company | Pyrazolopyridine-1,4-diamines and analogs thereof |
| US7943636B2 (en) | 2005-04-01 | 2011-05-17 | 3M Innovative Properties Company | 1-substituted pyrazolo (3,4-C) ring compounds as modulators of cytokine biosynthesis for the treatment of viral infections and neoplastic diseases |
| US8546368B2 (en) | 2006-02-15 | 2013-10-01 | Abbvie Inc. | Pyrazoloquinolones are potent PARP inhibitors |
| EP3085373A1 (en) | 2006-02-22 | 2016-10-26 | 3M Innovative Properties Company | Immune response modifier conjugates |
| US7906506B2 (en) | 2006-07-12 | 2011-03-15 | 3M Innovative Properties Company | Substituted chiral fused [1,2] imidazo [4,5-c] ring compounds and methods |
| US10005772B2 (en) | 2006-12-22 | 2018-06-26 | 3M Innovative Properties Company | Immune response modifier compositions and methods |
| US10144735B2 (en) | 2006-12-22 | 2018-12-04 | 3M Innovative Properties Company | Immune response modifier compositions and methods |
| RU2545023C2 (en) * | 2008-06-10 | 2015-03-27 | Эббви Инк. | Tricyclic compounds, pharmaceutical composition containing them and using them for treating immunological and oncological compounds |
| US8962629B2 (en) | 2008-06-10 | 2015-02-24 | Abbvie Inc. | Tricyclic compounds |
| US9365579B2 (en) | 2008-06-10 | 2016-06-14 | Abbvie Inc. | Tricyclic compounds |
| RU2545023C9 (en) * | 2008-06-10 | 2016-06-20 | Эббви Инк. | Tricyclic compounds, pharmaceutical composition containing them and using them for treating immunological and oncological compounds |
| CN108774237A (en) * | 2009-12-01 | 2018-11-09 | Abbvie 公司 | New tricyclic compound |
| USRE47221E1 (en) | 2009-12-01 | 2019-02-05 | Abbvie Inc. | Tricyclic compounds |
| CN102190622B (en) * | 2010-03-15 | 2015-01-07 | 李勤耕 | Water-soluble derivatives of edaravone, preparation method and application thereof |
| CN102190622A (en) * | 2010-03-15 | 2011-09-21 | 李勤耕 | Water-soluble derivatives of edaravone, preparation method and application thereof |
| US9499535B2 (en) | 2011-04-21 | 2016-11-22 | Origenis Gmbh | Kinase inhibitors |
| CN103717597A (en) * | 2011-04-21 | 2014-04-09 | 原真股份有限公司 | Heterocyclic compounds as kinase inhibitors |
| TWI557121B (en) * | 2011-04-21 | 2016-11-11 | 原真股份有限公司 | Novel kinase inhibitors |
| EP3153180A1 (en) | 2011-06-03 | 2017-04-12 | 3M Innovative Properties Company | Heterobifunctional linkers with polyethylene glycol segments and immune response modifier conjugates made therefrom |
| US10752624B2 (en) | 2012-10-19 | 2020-08-25 | Origenis Gmbh | Kinase inhibitors |
| US10000482B2 (en) | 2012-10-19 | 2018-06-19 | Origenis Gmbh | Kinase inhibitors |
| US10995095B2 (en) | 2015-10-16 | 2021-05-04 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carb oxamide and solid state forms thereof |
| US11535624B2 (en) | 2015-10-16 | 2022-12-27 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US9963459B1 (en) | 2015-10-16 | 2018-05-08 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-alpla]pyrrolo[2,3-e]-pyrazin-8-YL)-N-(2,2,2-Trifluoroethyl)pyrrol and solid state forms thereof |
| US9951080B2 (en) | 2015-10-16 | 2018-04-24 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-alpha]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US9879018B2 (en) | 2015-10-16 | 2018-01-30 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl and solid state forms thereof |
| US10202393B2 (en) | 2015-10-16 | 2019-02-12 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10202394B2 (en) | 2015-10-16 | 2019-02-12 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10344036B2 (en) | 2015-10-16 | 2019-07-09 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-#a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-#carboxamide and solid state forms thereof |
| US10519164B2 (en) | 2015-10-16 | 2019-12-31 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3,ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10550126B2 (en) | 2015-10-16 | 2020-02-04 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-A]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10597400B2 (en) | 2015-10-16 | 2020-03-24 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carb oxamide and solid state forms thereof |
| US10730883B2 (en) | 2015-10-16 | 2020-08-04 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US9879019B2 (en) | 2015-10-16 | 2018-01-30 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10981923B2 (en) | 2015-10-16 | 2021-04-20 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[l,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10981924B2 (en) | 2015-10-16 | 2021-04-20 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US12091415B2 (en) | 2015-10-16 | 2024-09-17 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11186584B2 (en) | 2015-10-16 | 2021-11-30 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11198697B1 (en) | 2015-10-16 | 2021-12-14 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11365198B2 (en) | 2015-10-16 | 2022-06-21 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11512092B2 (en) | 2015-10-16 | 2022-11-29 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11524964B2 (en) | 2015-10-16 | 2022-12-13 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US10017517B2 (en) | 2015-10-16 | 2018-07-10 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluorethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11535626B2 (en) | 2015-10-16 | 2022-12-27 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1 carboxamide and solid state forms thereof |
| US11535625B2 (en) | 2015-10-16 | 2022-12-27 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11661425B2 (en) | 2015-10-16 | 2023-05-30 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11680069B2 (en) | 2015-10-16 | 2023-06-20 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US12077545B2 (en) | 2015-10-16 | 2024-09-03 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11718627B2 (en) | 2015-10-16 | 2023-08-08 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11767326B2 (en) | 2015-10-16 | 2023-09-26 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11773105B2 (en) | 2015-10-16 | 2023-10-03 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]- pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11773106B2 (en) | 2015-10-16 | 2023-10-03 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11780848B2 (en) | 2015-10-16 | 2023-10-10 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1- carboxamide and solid state forms thereof |
| US11780847B1 (en) | 2015-10-16 | 2023-10-10 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1- carboxamide and solid state forms thereof |
| US11787815B1 (en) | 2015-10-16 | 2023-10-17 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-N-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11795175B2 (en) | 2015-10-16 | 2023-10-24 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11993606B2 (en) | 2015-10-16 | 2024-05-28 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| US11976077B2 (en) | 2015-10-16 | 2024-05-07 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-α]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms therof |
| US11993605B2 (en) | 2015-10-16 | 2024-05-28 | Abbvie Inc. | Processes for the preparation of (3S,4R)-3-ethyl-4-(3H-imidazo[1,2-a]pyrrolo[2,3-e]-pyrazin-8-yl)-n-(2,2,2-trifluoroethyl)pyrrolidine-1-carboxamide and solid state forms thereof |
| WO2017134066A1 (en) | 2016-02-05 | 2017-08-10 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| WO2023143356A1 (en) * | 2022-01-26 | 2023-08-03 | 勤浩医药(苏州)有限公司 | Methionine adenosyltransferase 2a inhibitor for treating mtap deletion-type cancer |
| WO2024080788A1 (en) * | 2022-10-13 | 2024-04-18 | 한미약품 주식회사 | Novel tricyclic derivative compound and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US8138173B2 (en) | 2012-03-20 |
| WO2006107771A3 (en) | 2007-10-04 |
| US20090069299A1 (en) | 2009-03-12 |
| AU2006232294A1 (en) | 2006-10-12 |
| CA2603063A1 (en) | 2006-10-12 |
| JP2008538119A (en) | 2008-10-09 |
| EP1863813A2 (en) | 2007-12-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1863813A2 (en) | Pyrazolo[3,4-c]quinolines, pyrazolo[3,4-c]naphthyridines, analogs thereof, and methods | |
| US7943610B2 (en) | Pyrazolopyridine-1,4-diamines and analogs thereof | |
| JP5247458B2 (en) | Hydroxy and alkoxy substituted 1H-imidazoquinolines and methods | |
| US7943636B2 (en) | 1-substituted pyrazolo (3,4-C) ring compounds as modulators of cytokine biosynthesis for the treatment of viral infections and neoplastic diseases | |
| JP5296526B2 (en) | Pyrazolopyridines and their analogs | |
| TWI428339B (en) | Azabicyclic carboxamide derivatives, preparation and therapeutic use thereof | |
| WO2001042247A1 (en) | Fused heterotricyclic compounds, process for preparing the compounds and drugs containing the same | |
| KR20080031496A (en) | Hydroxy substituted 1h-imidazopyridines and methods | |
| WO2007143526A2 (en) | Substituted tetrahydroimidazonaphthyridines and methods | |
| AU2007248603A1 (en) | Pyrido [2, 3-b] pyrazine and [1, 8] -naphthyridine derivatives as ALK and c-Met inhibitors | |
| EP1784180A2 (en) | 2-amino 1h imidazo ring systems and methods | |
| EP1831226A2 (en) | CHIRAL FUSED 1,2 IMIDAZO 4,5-c RING COMPOUNDS | |
| WO2006009826A1 (en) | Aryloxy and arylalkyleneoxy substituted thiazoloquinolines and thiazolonaphthyridines | |
| WO2007079086A1 (en) | Pyrazoloalkyl substituted imidazo ring compounds and methods | |
| TW201127385A (en) | N-containing heteroaryl derivatives as JAK3 kinase inhibitors | |
| TW201035095A (en) | Pyrazolo[1,5-a]pyridine derivatives | |
| EP3027613A1 (en) | Pyrroloquinoline derivatives as 5-ht6 antagonists, preparation method and use thereof | |
| JP4100865B2 (en) | Tricyclic fused heterocyclic compound, process for producing the same and medicament |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| ENP | Entry into the national phase |
Ref document number: 2603063 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2008504437 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006232294 Country of ref document: AU |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006740248 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2006232294 Country of ref document: AU Date of ref document: 20060331 Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 11887526 Country of ref document: US |