WO2005092316A1 - Novel pharmaceutical compositions comprising agonists of the thyroid receptor - Google Patents
Novel pharmaceutical compositions comprising agonists of the thyroid receptor Download PDFInfo
- Publication number
- WO2005092316A1 WO2005092316A1 PCT/EP2005/003030 EP2005003030W WO2005092316A1 WO 2005092316 A1 WO2005092316 A1 WO 2005092316A1 EP 2005003030 W EP2005003030 W EP 2005003030W WO 2005092316 A1 WO2005092316 A1 WO 2005092316A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- oxy
- benzyl
- phenyl
- alkyl
- amino
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/01—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms
- C07C311/02—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
- C07C311/08—Sulfonamides having sulfur atoms of sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton having the nitrogen atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/14—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/04—Inotropic agents, i.e. stimulants of cardiac contraction; Drugs for heart failure
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C205/00—Compounds containing nitro groups bound to a carbon skeleton
- C07C205/07—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by halogen atoms
- C07C205/11—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by halogen atoms having nitro groups bound to carbon atoms of six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/01—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C233/16—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms
- C07C233/24—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by a carbon atom of a six-membered aromatic ring
- C07C233/25—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by a carbon atom of a six-membered aromatic ring having the carbon atom of the carboxamide group bound to a hydrogen atom or to a carbon atom of an acyclic saturated carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/01—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms
- C07C233/16—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms
- C07C233/24—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by a carbon atom of a six-membered aromatic ring
- C07C233/27—Carboxylic acid amides having carbon atoms of carboxamide groups bound to hydrogen atoms or to acyclic carbon atoms having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms with the substituted hydrocarbon radical bound to the nitrogen atom of the carboxamide group by a carbon atom of a six-membered aromatic ring having the carbon atom of the carboxamide group bound to a carbon atom of an acyclic unsaturated carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C233/00—Carboxylic acid amides
- C07C233/57—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of rings other than six-membered aromatic rings
- C07C233/60—Carboxylic acid amides having carbon atoms of carboxamide groups bound to carbon atoms of rings other than six-membered aromatic rings having the nitrogen atom of at least one of the carboxamide groups bound to a carbon atom of a hydrocarbon radical substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/15—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings
- C07C311/16—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the sulfonamide groups bound to hydrogen atoms or to an acyclic carbon atom
- C07C311/19—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the sulfonamide groups bound to hydrogen atoms or to an acyclic carbon atom to an acyclic carbon atom of a hydrocarbon radical substituted by carboxyl groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/15—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings
- C07C311/21—Sulfonamides having sulfur atoms of sulfonamide groups bound to carbon atoms of six-membered aromatic rings having the nitrogen atom of at least one of the sulfonamide groups bound to a carbon atom of a six-membered aromatic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C311/00—Amides of sulfonic acids, i.e. compounds having singly-bound oxygen atoms of sulfo groups replaced by nitrogen atoms, not being part of nitro or nitroso groups
- C07C311/22—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound oxygen atoms
- C07C311/23—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound oxygen atoms having the sulfur atoms of the sulfonamide groups bound to acyclic carbon atoms
- C07C311/24—Sulfonamides, the carbon skeleton of the acid part being further substituted by singly-bound oxygen atoms having the sulfur atoms of the sulfonamide groups bound to acyclic carbon atoms of an acyclic saturated carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/06—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members
- C07D261/10—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/04—Systems containing only non-condensed rings with a four-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/06—Systems containing only non-condensed rings with a five-membered ring
- C07C2601/08—Systems containing only non-condensed rings with a five-membered ring the ring being saturated
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/04—Endocrine or metabolic disorders
- G01N2800/046—Thyroid disorders
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/74—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving hormones or other non-cytokine intercellular protein regulatory factors such as growth factors, including receptors to hormones and growth factors
- G01N33/78—Thyroid gland hormones, e.g. T3, T4, TBH, TBG or their receptors
Definitions
- the present invention relates to compounds which are agonists or partial agonists of the thyroid 5 receptor and the use of such compounds for therapeutic purposes
- Thyroid hormones affect the metabolism of virtually every cell of the body. At normal levels, these 15 hormones maintain body weight, metabolic rate, body temperature and mood, and influence blood levels of serum lipoproteins. Thus, in hypothyroidism there is weight gain, high levels of LDL cholesterol, and depression. In hyperthyroidism, these hormones lead to weight loss, hypermetabolism, lowering of serum LDL cholesterol levels, cardiac arrhythmias, heart failure, muscle weakness, bone loss in postmenopausal women, and anxiety. 20 Thyroid hormones are currently used primarily as replacement therapy for patients with hypothyroidism.
- TSH thyroid-stimulating hormone
- thyroxine 3,5,3',5'-tetraiodo-L-thyronine, or T
- triiodothyronine 3,5,3'-triiodo-L-thyronine, or 25 T 3
- replacement therapy particularly in older individuals, may be restricted by certain detrimental effects from thyroid hormones.
- thyroid hormones may be therapeutically useful in non-thyroid disorders if adverse effects can be minimized or eliminated.
- These potentially useful influences include for 30 example, lowering of serum LDL levels, weight reduction, amelioration of depression and stimulation of bone formation.
- Prior attempts to utilize thyroid hormones pharmacologically to treat these disorders have been limited by manifestations of hyperthyroidism, and in particular by cardiovascular toxicity.
- useful thyroid agonist drugs should minimize the potential for undesired consequences due to locally induced hypothyroidism, i.e. sub-normal levels of thyroid hormone activity in certain tissues or organs. This can arise because increased circulating thyroid hormone agonist concentrations may cause the pituitary to suppress the secretion of thyroid stimulating hormone (TSH), thereby reducing thyroid hormone synthesis by the thyroid gland (negative feedback control). Since endogenous thyroid hormone levels are reduced, localized hypothyroidism can result wherever the administered thyroid agonist drug fails to compensate for the reduction in endogenous hormone levels in specific tissues.
- TSH thyroid stimulating hormone
- Tissue-selective thyroid hormone agonists may be obtained by selective tissue uptake or extrusion, topical or local delivery, targeting to cells through other ligands attached to the agonist and targeting receptor subtypes. Tissue selectivity can also be achieved by selective regulation of thyroid hormone responsive genes in a tissue specific manner.
- the compounds that are thyroid hormone receptor ligands, particularly selective agonists of the thyroid hormone receptor are expected to demonstrate a utility for the treatment or prevention of diseases or disorders associated with thyroid hormone activity, for example: (1) hypercholesterolemia, dyslipidemia or any other lipid disorder manifested by an unbalance of blood or tissue lipid levels ; (2) atherosclerosis; (3) replacement therapy in elderly subjects with hypothyroidism who are at risk for cardiovascular complications; (4) replacement therapy in elderly subjects with subclinical hypothyroidism who are at risk for cardiovascular complications; (5) obesity; (6) diabetes (7) depression; (8) osteoporosis (especially in combination with a bone resorption inhibitor); (9) goiter; (10) thyroid cancer; (11) cardiovascular disease or congestive heart failure; (12) glaucoma; and (13) skin disorders.
- diseases or disorders associated with thyroid hormone activity for example: (1) hypercholesterolemia, dyslipidemia or any other lipid disorder manifested by an unbalance of blood or tissue lipid levels ; (2) atherosclerosis; (3) replacement therapy in elderly
- the present invention provides a compound of formula (I) or a pharmaceutically acceptable ester, amide, solvate or salt thereof, including a salt of such an ester or amide, and a solvate of such an ester, amide or salt,
- R 1 is selected from -S0 2 R 6 , -SOR 6 and -C(0)R 6 ;
- R 6 is selected from C ⁇ . 8 alkyl, C 2 .$ alkenyl, C 2 . 8 alkynyl, C 3 . 8 cycloalkyl, C 3 . 8 cycloalkyl-C ⁇ . 3 alkyl, phenyl and C 1 .
- heterocyclyl said alkyl, alkenyl or alkynyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, hydroxy, methoxy, halomethoxy, dihalomethoxy, and trihalomethoxy; said cycloalkyl, aryl or heterocyclyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, hydroxy, C alkyl, C 2 _» alkenyl, C 2 _ t alkynyl, methoxy, halomethoxy, dihalomethoxy, trihalomethoxy, haloC ⁇ _ 4 alkyl, dihaloC M alkyl, and alkyl;
- Each R 2 is independently selected from halogen, mercapto, nitro, cyano, alkoxy, -C0 2 R c , -CONHR 0 , -CHO, -S0 2 R 6 , -S0 2 NHR 6 , C M alkyl, C 2 .
- alkenyl C M alkynyl, NHR 1 and N(R') 2 , said alkyl, alkenyl, alkynyl or alkoxy groups optionally being substituted with 1, 2 or 3 groups selected from halogen, hydroxy, methoxy, C M alkoxy, C ⁇ alkylthio, mercapto, nitro, cyano, halomethoxy, dihalomethoxy, and trihalomethoxy;
- n 0, 1, 2 or 3;
- R a is selected from hydrogen, halogen, hydroxy, mercapto, C ⁇ . alkyl, CM alkenyl, C 2 _ 4 alkynyl, C alkoxy, fluoromethyl, difluoromethyl, trifluoromethyl, fiuoromethoxy, difluoromethoxy, trifluoromethoxy, methylthio, fluoromethylthio, difluoromethylthio and thiotrifluoromethyl;
- R a is selected from hydrogen, halogen, mercapto, C M alkyl, C 2 - 4 alkenyl, C 2 _ t alkynyl, CM alkoxy, fluoromethyl, difluoromethyl, trifluoromethyl, fiuoromethoxy, difluoromethoxy, trifluoromethoxy, methylthio, fluoromethylthio, difluoromethylthio and thiotrifluoromethyl; R 3 and R 4 are independently selected from halogen, C M alkyl, C 2 - .
- W is selected from C M alkylene, C 2 . 3 alkenylene, C 2 . 3 alkynylene, N(R b )-C ⁇ . 3 alkylene, C(0)-C].
- R b is selected from hydrogen, hydroxy, C M alkyl, C 2 _ « alkenyl, C 2 _ 4 alkynyl, C alkoxy, fluoromethyl, difluoromethyl, trifluoromethyl, fiuoromethoxy, difluoromethoxy, trifluoromethoxy;
- R 5 is selected from -C0 2 R°, -PO(OR c ) 2 , -PO(OR c )NH 2 , -S0 2 OR c , -COC0 2 R°, CONROR 0 , - S0 2 NHR c , -NHS0 2 R° ' , -CONHS0 2 R c , and - S0 2 NHCOR c ;
- Each R° is independently selected from hydrogen, CM alkyl, 2 alkenyl and C 2 . 4 alkynyl;
- R° is selected from R c , C 5 . ⁇ 0 aryl and C 5 readily ⁇ o aryl substituted with 1, 2 or 3 groups independently selected from amino, hydroxy, halogen and CM alkyl.
- Compounds of the invention have surprisingly been found to be ligands of the thyroid receptor, in particular agonists or partial agonists of the thyroid receptor.
- the compounds accordingly have use in the treatment or prophylaxis of conditions associated with thyroid receptor activity.
- the compounds of formula (1) may contain chiral (asymmetric) centres or the molecule as a whole may be chiral.
- the individual stereoisomers (enantiomers and diastereoisomers) and mixtures of these are within the scope of the present invention.
- R 1 is selected from -S0 2 R 6 , and -C(0)R 6 ;
- R 6 is selected from C ⁇ ._ alkyl, C 2 _ alkenyl, C 3 . 6 cycloalkyl, C 3-5 cycloalkyl C N3 alkyl, phenyl and C 3 . 7 heterocyclyl.
- Preferred substituents for said alkyl or alkenyl include groups independently selected from halogen, methoxy or halomethoxy.
- Preferred substituents for said cycloalkyl, aryl or heterocyclyl include halogen, methyl, ethyl, halomethoxy, dihalomethoxy, trihalomethoxy, halomethyl, dihalomethyl, and trihalomethyl.
- R 6 is selected from C 5 alkyl, C 2 _ 4 alkenyl, C 3 . 6 cycloalkyl, C 3 . 6 cycloalkyl-C ⁇ . 3 alkyl, said alkyl or alkenyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, hydroxy, methoxy, halomethoxy, dihalomethoxy and trihalomethoxy.
- R 6 is selected from C M alkyl, C 2 _ t alkenyl, C 3 _j cycloalkyl, C 3 . 6 cycloalkyl C t . 3 alkyl, unsubstituted phenyl, and C 3 . 5 heterocyclyl.
- R 6 is preferably selected from phenyl, methyl, ethyl, propyl or 3,5 dimethyl isoxazole, for example methyl, ethyl and propyl.
- R 1 is C(0)R 6
- R 6 is preferably selected from methyl, ethyl, propyl, cyclcobutyl, cyclopropyl, or i-propyl.
- R 2 is preferably selected from halogen, C ⁇ _ 2 alkyl, C 2 . 3 alkenyl, C 2 . 3 alkynyl, nitro, cyano, C ⁇ . 2 alkoxy, haloC ⁇ . 2 alkyl, dihaloC ⁇ alkyl, and trihaloC ⁇ . 2 alkyl. More preferably, R 2 is selected from halogen, methyl, trifluormethyl, difluoromethyl or fluoromethyl. When R 2 is a halogen, it is preferably selected from bromine, chlorine and fluorine, especially chlorine. Preferred locations for the R 2 group or groups are in the 2- or 5- position on the phenyl ring relative to the attachment point to the Y'-Y- of the remainder of the molecule.
- n is 0, 1 or 2. More preferably, n is 0 or 1, for example 1.
- Y and Y' are independently selected from oxygen, sulphur or -CH(R a )-, with the proviso that at least one of Y and Y' is -CH(R a )- and the further proviso that when one of Y and Y' is oxygen or sulphur, then R a is hydrogen, halogen, CM alkyl, C 2 - alkenyl, C 2 _ t alkynyl, fluoromethyl, difluoromethyl, trifluoromethyl. More preferably, Y is O or S, and Y' is CH(R a ). Most preferably, Y is O and Y' is CH(R a ).
- R a is preferably selected from hydrogen, halogen, C 2 alkyl, fluoromethyl, difluoromethyl and trifluoromethyl. More preferably, R a is selected from hydrogen, halogen, and C ⁇ . 2 alkyl. Most preferably, R a is hydrogen.
- R a' is preferably selected from hydrogen, halogen, C ⁇ . 2 alkyl, fluoromethyl, difluoromethyl and trifluoromethyl. More preferably, R a is selected from hydrogen, halogen, and C ⁇ _ 2 alkyl. Most preferably, R a is hydrogen.
- R 3 and R 4 are preferably independently selected from halogen, C M alkyl, fluoromethyl, difluoromethyl, trifluoromethyl, and C alkoxy. More preferably, R 3 and R 4 are independently selected from halogen, CM alkyl, fluoromethyl, difluoromethyl and trifluoromethyl. Most preferably, R 3 and R 4 are independently selected from halogen, methyl, fluoromethyl, difluoromethyl and trifluoromethyl. Amongst the halogens, there are preferred bromine, chlorine and fluorine, especially bromine and chlorine, in particular bromine.
- R 3 and R 4 may simultaneously represent the same radical. Alternatively, R 3 and R 4 are different from each other.
- W is preferably selected from C ⁇ _ 3 alkylene, C 2 . 3 alkenylene, N(R b )-C ⁇ _ 2 alkylene, C(0)-C ⁇ . 2 alkylene, S- . ⁇ alkylene, 0-C ⁇ _ 2 alkylene, C(O)NH-C 0 . 2 alkylene or NH(CO)-C ⁇ . 2 alkylene, said alkylene or alkenylene groups or portions of groups optionally being substituted with a group selected from hydroxy, mercapto, amino, halo (preferably fluoro or chloro, particularly fluoro), . 2 alkyl, C ⁇ _ 2 alkoxy, haloC]. 2 alkyl, dihaloC ⁇ .
- W is selected from C ⁇ . 3 alkylene, C 2 . 3 alkenylene, 0-C ⁇ . 3 alkylene, C,. 3 alkylene-0-C,. 3 alkylene, C(0)NH-C ! . 2 alkylene and NH(CO)-C ⁇ . 2 alkylene.
- W is selected from C ⁇ . 3 alkylene, O-C 1 . 3 alkylene, C ⁇ . 3 alkylene-0-Ci. 3 alkylene, C(0)-C ! . 2 alkylene, C(0)NH-C,.
- W is ethylene or C(0)NH-CH 2 -.
- the alkylene group (for example the ethylene group) is substituted with one or more halo groups, for example one or more fluoro groups (for example one fluoro group).
- Monohalo C ⁇ . 3 alkylene (for example fluoro C ⁇ thread 3 alkylene) thus constitutes a preferred group W.
- R b is preferably selected from hydrogen, C ⁇ . 2 alkyl, fluoromethyl, difluoromethyl and trifluoromethyl;
- R 5 is preferably selected from -C0 2 R c , -PO(OR c ) 2 , -S0 2 OR c , -COC0 2 R°, CONR c OR° and - NHS0 2 R c' . More preferably, R 5 is -C0 2 R c , -PO(OR c ) 2 or -S0 2 OR c . Most preferably, R 5 is -C0 2 R c , particularly -C0 2 H.
- R c is preferably hydrogen or C alkyl. More preferably, R° is ethyl, methyl or hydrogen, particularly hydrogen.
- R c' is preferably selected from R c , phenyl and phenyl substituted with 1, 2 or 3 groups independently selected from amino, hydroxyl, halogen and methyl.
- one preferred group of compounds of the invention includes compounds according to formula (la) or pharmaceutically acceptable esters, amides, solvates or salts thereof, including salts of such esters or amides, and solvates of such esters, amides or salts
- R 1 is selected from -S0 2 R 6 and -C(0)R 6
- R 6 is selected from C ⁇ . 8 alkyl, C 2 _ 4 alkenyl, C 3 . 6 cycloalkyl, C 3 . ⁇ cycloalkyl-C ⁇ . 3 alkyl, C 6 aryl and C 3 .
- heterocyclyl said alkyl or alkenyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, hydroxy, methoxy, halomethoxy, dihalomethoxy and trihalomethoxy; said cycloalkyl, aryl or heterocyclyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, methyl, methoxy, halomethoxy, dihalomethoxy, and trihalomethoxy;
- Each R 2 is independently selected from halogen, C ⁇ . 2 alkyl, C 2 . 3 alkenyl, C 2 . 3 alkynyl, C ⁇ . 2 alkoxy, haloC ⁇ . 2 alkyl, dihaloC ⁇ alkyl, and trihaloC ⁇ . 2 alkyl;
- R a is selected from hydrogen, halogen, methyl, ethyl, fluoromethyl, difluoromethyl, trifluoromethyl, fluoroethyl, difluoroethyl and trifluoroethyl;
- R 3 and R 4 are independently selected from halogen, C M alkyl, fluoromethyl, difluoromethyl, trifluoromethyl, C alkoxy, fiuoromethoxy, difluoromethoxy, trifluoromethoxy, methylthio, fluoromethylthio, difluoromethylthio and trifluoromethylthio;
- W is selected from C ⁇ _ 3 alkylene, C ⁇ . 3 alkylene-0-C ⁇ . 3 alkylene, 0-C ⁇ _ 3 alkylene, C(0)-C ⁇ . 2 alkylene, C(0)NH-C ⁇ . 2 alkylene and NH(CO)-C ⁇ . 2 alkylene; the alkylene group or portion of a group optionally being substituted with one or more halo groups.
- R 5 is selected from -C0 2 R c , -PO(OR c ) 2 , -S0 2 OR c , -COC0 2 R c , CONR c OR c and NHS0 2 R c' ;
- Each R c is independently selected from hydrogen and CM alkyl
- R° is selected from R c , phenyl and phenyl substituted with amino, hydroxy, halogen and methyl.
- a further preferred group of compounds of the invention includes compounds according to formula (lb) or pharmaceutically acceptable esters, amides, solvates or salts thereof, including salts of such esters or amides, and solvates of such esters, amides or salts,
- R 1 is selected from -S0 2 R 6 and -C(0)R 6 ;
- R ⁇ is selected from C 1 . 5 alkyl, C 2 _. alkenyl, C 3 ._ cycloalkyl, C 3 . 6 cycloalkyl-C ⁇ alkyl, said alkyl or alkenyl groups or portions of groups optionally being substituted with 1, 2 or 3 groups independently selected from halogen, hydroxy, methoxy, halomethoxy, dihalomethoxy and trihalomethoxy;
- Each R 2 is independently selected from halogen, C ⁇ . 2 alkyl, C 2 . 3 alkenyl, C 2 . 3 alkynyl, C_. 2 alkoxy, haloC ⁇ . 2 alkyl, dihaloC ⁇ profession 2 alkyl, and trihaloC ⁇ . 2 alkyl;
- n 0, 1 or 2;
- R a is selected from hydrogen, halogen, and C ⁇ . 2 alkyl
- R 3 and R 4 are independently selected from halogen, C alkyl, fluoromethyl, difluoromethyl, trifluoromethyl, and C M alkoxy;
- W is selected from C ⁇ . 3 alkylene, C 2 . 3 alkenylene, 0-C ⁇ . 3 alkylene, d_ 3 alkylene-0-Ci_ 3 alkylene, C(0)NH-C ! _ 2 alkylene and NH(CO)-C t . 2 alkylene; the alkylene group or portion of a group optionally being substituted with one or more halo groups.
- R 5 is -C0 2 R c ;
- Each R° is independently selected from hydrogen and C M alkyl.
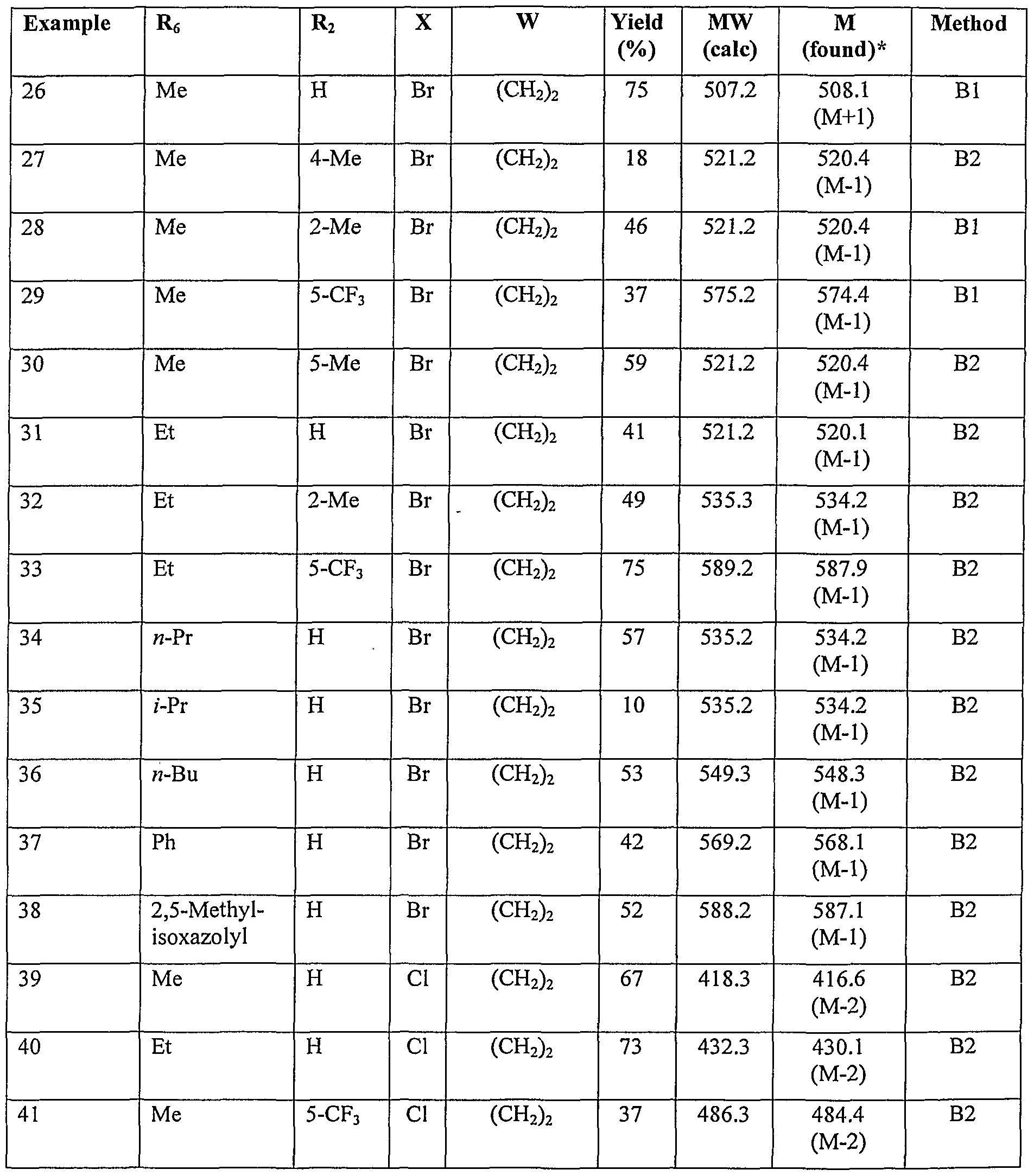
- Preferred compounds according to the invention include: 3-(4- ⁇ [3-(acetylamino)-5-(trifluoromethyl)benzyl]oxy ⁇ -3,5-dichlorophenyl) ⁇ ropanoic acid N-(4- ⁇ [3-(acetylamino)-5-(trifluoromethyl)benzyl]oxy ⁇ -3,5-dibromobenzoyl)glycine
- More preferred compounds according to the invention include:
- Most preferred compounds according to the invention include: 3-(4- ⁇ [3-(acetylamino)-5-(trifluoromethyl)benzyl]oxy ⁇ -3,5-dichlorophenyl)propanoic acid 3-(4- ⁇ [3-(acetylamino)-5-methylbenzyl]oxy ⁇ -3,5-dibromophenyl)propanoic acid 3-(4- ⁇ [3-(acetylamino)-2-chlorobenzyl]oxy ⁇ -3,5-dibromophenyl)propanoic acid 3-(4- ⁇ [3-(acetylamino)-2-fluoro-5-methylbenzyl]oxy ⁇ -3,5-dibromophenyl)propanoic acid 3-(4- ⁇ [3-(acetylamino)-5-chloro-2-fluorobenzyl]oxy ⁇ -3,5-dibromophenyl)propanoic acid 3-(4- ⁇ [3-(acet
- Salts and solvates of compounds of formula (I) which are suitable for use in medicine are those wherein a counterion or associated solvent is pharmaceutically acceptable.
- salts and solvates having non-pharmaceutically acceptable counterions or associated solvents are within the scope of the present invention, for example, for use as intermediates in the preparation of the compounds of formula (I) and their pharmaceutically acceptable salts, solvates and physiologically functional derivatives.
- physiologically functional derivatives include esters, amides, and carbamates; preferably esters and amides.
- Suitable salts according to the invention include those formed with organic or inorganic acids or bases.
- Pharmaceutically acceptable acid addition salts include those formed from hydrochloric, hydrobromic, sulphuric, nitric, citric, tartaric, acetic, phosphoric, lactic, pyruvic, acetic, trifluoroacetic, succinic, perchloric, fumaric, maleic, glycollic, lactic, salicylic, oxaloacetic, methanesulfonic, ethanesulfonic, p-toluenesulfonic, formic, benzoic, malonic, naphthalene-2- sulfonic, benzenesulfonic, and isethionic acids.
- Pharmaceutically acceptable base salts include ammonium salts, alkali metal salts, for example those of potassium and sodium, alkaline earth metal salts, for example those of calcium and magnesium, and salts with organic bases e.g. primary, secondary or tertiary organic amines, for example dicyclohexylamine, and N- methyl-D-glucomine.
- esters and amides of the compounds of formula (I) may have an appropriate group, for example an acid group, converted to a C ⁇ . 6 alkyl, C 5 . I0 aryl, C 5 . ⁇ 0 ar-C ⁇ alkyl, or amino acid ester or amide.
- Pharmaceutically acceptable amides and carbonates of the compounds of formula (I) may have an appropriate group, for example an amino group, converted to a C ⁇ . 6 alkyl, C 5 . 1 0 aryl, C5.1 0 aryl-C ⁇ . 6 alkyl, or amino acid ester or amide, or carbamate.
- a compound which, upon administration to the recipient, is capable of being converted into a compound of formula (I) as described above or an active metabolite or residue thereof, is known as a "prodrug".
- a prodrug may, for example, be converted within the body, e. g. by hydrolysis in the blood, into its active form that has medical effects.
- Pharmaceutical acceptable prodrugs are described in T. Higuchi and V. Stella, Prodrugs as Novel Delivery Systems, Vol. 14 of the A. C. S. Symposium Series (1976); and in Edward B. Roche, ed., Bioreversible Carriers in Drug Design, American Pharmaceutical Association and Pergamon Press, 1987, both of which are incorporated herein by reference.
- alkyl means both straight and branched chain saturated hydrocarbon groups.
- alkyl groups include methyl, ethyl, n-propyl, iso-propyl, n-butyl, t-butyl, i- butyl, sec-butyl pentyl, hexyl, heptyl, octyl, nonyl and decyl groups.
- unbranched alkyl groups there are preferred methyl, ethyl, n-propyl, iso-propyl, n-butyl groups.
- branched alkyl groups there may be mentioned t-butyl, i-butyl, 1-ethylpropyl, 1-ethylbutyl and 1-ethylpentyl groups.
- alkoxy means the group O-alkyl, where "alkyl” is used as described above.
- alkoxy groups include methoxy and ethoxy groups.
- Other examples include propoxy and butoxy.
- alkenyl means both straight and branched chain unsaturated hydrocarbon groups with at least one carbon carbon double bond. Up to 5 carbon carbon double bonds may, for example, be present.
- alkenyl groups include ethenyl, propenyl, butenyl, pentenyl, hexenyl, heptenyl, octenyl, nonenyl, decenyl and dodecenyl.
- Preferred alkenyl groups include ethenyl, 1- propenyl and 2- propenyl.
- alkynyl means both straight and branched chain unsaturated hydrocarbon groups with at least one carbon carbon triple bond. Up to 5 carbon carbon triple bonds may, for example, be present.
- alkynyl groups include ethynyl, propynyl, butynyl, pentynyl, hexynyl, heptynyl, octynyl, nonynyl, decynyl and dodecynyl.
- Preferred alkenyl groups include ethynyl 1- propynyl and 2- propynyl.
- cycloalkyl means a saturated group in a ring system.
- the cycloalkyl group can be monocyclic or bicyclic.
- a bicyclic group may, for example, be fused or bridged.
- monocyclic cycloalkyl groups include cyclopropyl, cyclobutyl and cyclopentyl.
- Other examples of monocyclic cycloalkyl groups are cyclohexyl, cycloheptyl and cyclooctyl.
- bicyclic cycloalkyl groups include bicyclo [2. 2.1]hept-2-yl.
- the cycloalkyl group is monocyclic.
- aryl means a monocyclic or bicyclic aromatic carbocyclic group.
- aryl groups include phenyl and naphthyl. A naphthyl group may be attached through the 1 or the 2 position.
- one of the rings may, for example, be partially saturated. Examples of such groups include indanyl and tetrahydronaphthyl.
- C 5 . 10 aryl is used herein to mean a group comprising from 5 to 10 carbon atoms in a monocyclic or bicyclic aromatic group.
- a particularly preferred C5. 10 aryl group is phenyl.
- halogen means fluorine, chlorine, bromine or iodine. Fluorine, chlorine and bromine are particularly preferred.
- heterocyclyl means an aromatic (“heteroaryl”) or a non-aromatic (“heterocycloalkyl”) cyclic group of carbon atoms wherein from one to three of the carbon atoms is/are replaced by one or more heteroatoms independently selected from nitrogen, oxygen and sulfur.
- a heterocyclyl group may, for example, be monocyclic or bicyclic. In a bicyclic heterocyclyl group there may be one or more heteroatoms in each ring, or only in one of the rings.
- a heteroatom is preferably O or N.
- Heterocyclyl groups containing a suitable nitrogen atom include the corresponding N-oxides.
- Examples of monocyclic heterocycloalkyl rings include aziridinyl, azetidinyl, pyrrolidinyl, imidazolidinyl, pyrazolidinyl, piperidinyl, piperazinyl, tetrahydrofuranyl, tetrahydropyranyl, morpholinyl, thiomorpholinyl and azepanyl.
- bicyclic heterocychc rings in which one of the rings is non-aromatic include dihydrobenzofuranyl, indanyl, indolinyl, isoindolinyl, tetrahydroisoquinolinyl, tetrahydroquinolyl and benzoazepanyl.
- Examples of monocyclic heteroaryl groups include furanyl, thienyl, pyrrolyl, oxazolyl, thiazolyl, imidazolyl, oxadiazolyl, thiadiazolyl, pyridyl, triazolyl, triazinyl, pyridazyl, pyrimidinyl, isothiazolyl, isoxazolyl, pyrazinyl, pyrazolyl and pyrimidinyl;
- examples of bicyclic heteroaryl groups include quinoxalinyl, quinazolinyl, pyridopyrazinyl, benzoxazolyl, benzothiophenyl, benzimidazolyl, naphthyridinyl, quinolinyl, benzofuranyl, indolyl, benzothiazolyl, oxazolyl[4,5- b]pyridiyl, pyridopyrimidinyl, iso
- heterocyclyl groups examples include piperidinyl, tetrahydrofuranyl, tetrahydropyranyl, pyridyl, pyrimidyl and indolyl.
- cycloalkylalkyl means a group cycloalkyl-alkyl- attached through the alkyl group, "cycloalkyl” and “alkyl” being understood to have the meanings outlined above.
- the compounds of the invention have activity as thyroid receptor ligands.
- the compounds of the invention are preferably selective agonists or partial agonists of the thyroid receptor.
- compounds of the present invention possess activity as agonists of the thyroid receptor, preferably selective agonists of the thyroid receptor-beta. They may thus be used in the treatment of diseases or disorders associated with thyroid receptor activity, particularly diseases or disorders for which selective agonists of the thyroid receptor-beta are indicated.
- compounds of the present invention may be used in the treatment of diseases or disorders associated with metabolism dysfunction or which are dependent upon the expression of a T 3 regulated gene.
- Clinical conditions for which an agonist or partial agonist is indicated include, but are not limited to, hypothyroidism; subclinical hyperthyroidism; non-toxic goiter; atherosclerosis; thyroid hormone replacement therapy (e.g., in the elderly); malignant tumor cells containing the thyroid receptor; papillary or follicular cancer; maintenance of muscle strength and function (e.g., in the elderly); reversal or prevention of frailty or age-related functional decline ("ARFD”) in the elderly (e.g., sarcopenia); treatment of catabolic side effects of glucocorticoids; prevention and/or treatment of reduced bone mass, density or growth (e.g., osteoporosis and osteopenia); treatment of chronic fatigue syndrome (CFS); accelerating healing of complicated fractures (e.g.
- distraction osteogenesis in joint replacement; eating disorders (e.g., anorexia); treatment of obesity and growth retardation associated with obesity; treatment of depression, nervousness, irritability and stress; treatment of reduced mental energy and low self-esteem (e.g., motivation/assertiveness); improvement of cognitive function (e.g., the treatment of dementia, including Alzheimer's disease and short term memory loss); treatment of catabolism in connection with pulmonary dysfunction and ventilator dependency; treatment of cardiac dysfunction (e.g., associated with valvular disease, myocardial infarction, cardiac hypertrophy or congestive heart failure); lowering blood pressure; protection against ventricular dysfunction or prevention of reperfusion events; treatment of hyperinsulinemia; stimulation of osteoblasts, bone remodeling and cartilage growth; regulation of food intake; treatment of insulin resistance, including NIDDM, in mammals (e.g., humans); treatment of insulin resistance in the heart; treatment of congestive heart failure; treatment of musculoskeletal impairment (e.g., in the elderly); improvement of the overall pulmonary function; skin disorders
- the compounds of the invention find particular application in the treatment or prophylaxis of the following: (1) hypercholesterolemia, dyslipidemia or any other lipid disorder manifested by an unbalance of blood or tissue lipid levels ; (2) atherosclerosis; (3) replacement therapy in elderly subjects with hypothyroidism who are at risk for cardiovascular complications; (4) replacement therapy in elderly subjects with subclinical hypothyroidism who are at risk for cardiovascular complications; (5) obesity; (6) diabetes (7) depression; (8) osteoporosis (especially in combination with a bone resorption inhibitor); (9) goiter; (10) thyroid cancer; (11) cardiovascular disease or congestive heart failure; (12) glaucoma; and (13) skin disorders.
- the compounds of the invention find especial application in the treatment or prophylaxis of the following: (1) hypercholesterolemia, dyslipidemia or any other lipid disorder manifested by an unbalance of blood or tissue lipid levels ; (2) atherosclerosis; (3) obesity; (4) diabetes.
- the invention also provides a method for the treatment or prophylaxis of a condition in a mammal mediated by a thyroid receptor, which comprises administering to the mammal a therapeutically effective amount of a compound of formula (I) as defined above or a pharmaceutically acceptable ester, amide, solvate or salt thereof, including a salt of such an ester or amide, and a solvate of such an ester, amide or salt.
- a thyroid receptor that may be treated by the method of the invention are those described above.
- the invention also provides the use of a compound of formula (I) as defined above or a pharmaceutically acceptable ester, amide, solvate or salt thereof, including a salt of such an ester or amide, and a solvate of such an ester, amide or salt, for the manufacture of a medicament for the treatment or prophylaxis of a condition mediated by a thyroid receptor.
- Clinical conditions mediated by a thyroid receptor that may be treated by the method of the invention are those described above.
- active ingredient means a compound of formula (I) as defined above, or a pharmaceutically acceptable ester, amide, solvate or salt thereof, including a salt of such an ester or amide, and a solvate of such an ester, amide or salt.
- the amount of active ingredient which is required to achieve a therapeutic effect will, of course, vary with the particular compound, the route of administration, the subject under treatment, and the particular disorder or disease being treated.
- the compounds of the invention may be administered orally or via injection at a dose of from 0.001 to 1500 mg/kg per day, preferably from 0.01 to 1500 mg/kg per day, more preferably from 0.1 to 1500 mg/kg per day, most preferably from 0.1 to 500 mg/kg per day.
- the dose range for adult humans is generally from 5 mg to 35 g per day and preferably 5 mg to 2 g per day. Tablets or other forms of presentation provided in discrete units may conveniently contain an amount of compound of the invention which is effective at such dosage or as a multiple of the same, for example units containing 5 mg to 500 mg, usually around 10 mg to 200 mg.
- the invention provides a pharmaceutical formulation comprising a compound of formula (I) as defined above or a pharmaceutically acceptable ester, amide, solvate or salt thereof, including a salt of such an ester or amide, and a solvate of such an ester, amide or salt, and a pharmaceutically acceptable excipient.
- the pharmaceutical formulations according to the invention include those suitable for oral, parenteral (including subcutaneous, intradermal, intramuscular, intravenous, and intraarticular), inhalation (including fine particle dusts or mists which may be generated by means of various types of metered does pressurized aerosols), nebulizers or insufflators, rectal and topical (including dermal, buccal, sublingual, and intraocular) administration, although the most suitable route may depend upon, for example, the condition and disorder of the recipient.
- parenteral including subcutaneous, intradermal, intramuscular, intravenous, and intraarticular
- inhalation including fine particle dusts or mists which may be generated by means of various types of metered does pressurized aerosols
- nebulizers or insufflators rectal and topical (including dermal, buccal, sublingual, and intraocular) administration, although the most suitable route may depend upon, for example, the condition and disorder of the recipient.
- the formulations may conveniently be presented in unit dosage form and may be prepared by any of the methods well known in the art of pharmacy. All methods include the step of bringing the active ingredient into association with the carrier which constitutes one or more accessory ingredients. In general the formulations are prepared by uniformly and intimately bringing into association the active ingredient with liquid carriers or finely divided solid carriers or both and then, if necessary, shaping the product into the desired formulation.
- Formulations of the present invention suitable for oral administration may be presented as discrete units such as capsules, cachets or tablets each containing a predetermined amount of the active ingredient; as a powder or granules; as a solution or a suspension in an aqueous liquid or a non- aqueous liquid; or as an oil-in-water liquid emulsion or a water-in-oil liquid emulsion.
- the active ingredient may also be presented as a bolus, electuary or paste.
- a tablet may be made by compression or moulding, optionally with one or more accessory ingredients.
- Compressed tablets may be prepared by compressing in a suitable machine the active ingredient in a free-flowing form such as a powder or granules, optionally mixed with a binder, lubricant, inert diluent, lubricating, surface active or dispersing agent.
- Moulded tablets may be made by moulding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
- the tablets may optionally be coated or scored and may be formulated so as to provide slow or controlled release of the active ingredient therein.
- the present compounds can, for example, be administered in a form suitable for immediate release or extended release.
- Immediate release or extended release can be achieved by the use of suitable pharmaceutical compositions comprising the present compounds, or, particularly in the case of extended release, by the use of devices such as subcutaneous implants or osmotic pumps.
- the present compounds can also be administered liposomally.
- compositions for oral administration include suspensions which can contain, for example, microcrystalline cellulose for imparting bulk, alginic acid or sodium alginate as a suspending agent, methylcellulose as a viscosity enhancer, and sweeteners or flavoring agents such as those known in the art; and immediate release tablets which can contain, for example, microcrystalline cellulose, dicalcium phosphate, starch, magnesium stearate and/or lactose and/or other excipients, binders, extenders, disintegrants, diluents and lubricants such as those known in the art.
- the compounds of formula I can also be delivered through the oral cavity by sublingual and/or buccal administration.
- Molded tablets, compressed tablets or freeze-dried tablets are exemplary forms which may be used.
- Exemplary compositions include those formulating the present compound(s) with fast dissolving diluents such as mannitol, lactose, sucrose and/or cyclodextrins. Also included in such formulations may be high molecular weight excipients such as celluloses (avicel) or polyethylene glycols (PEG).
- Such formulations can also include an excipient to aid mucosal adhesion such as hydroxy propyl cellulose (HPC), hydroxy propyl methyl cellulose (HPMC), sodium carboxy methyl cellulose (SCMC), maleic anhydride copolymer (e.g., Gantrez), and agents to control release such as polyacrylic copolymer (e.g. Carbopol 934).
- HPC hydroxy propyl cellulose
- HPMC hydroxy propyl methyl cellulose
- SCMC sodium carboxy methyl cellulose
- maleic anhydride copolymer e.g., Gantrez
- agents to control release such as polyacrylic copolymer (e.g. Carbopol 934).
- Lubricants, glidants, flavors, coloring agents and stabilizers may also be added for ease of fabrication and use.
- Formulations for parenteral administration include aqueous and non-aqueous sterile injection solutions which may contain anit-oxidants, buffers, bacteriostats and solutes which render the formulation isotonic with the blood of the intended recipient; and aqueous and non-aqueous sterile suspensions which may include suspending agents and thickening agents.
- the formulations may be presented in unit-dose or multi-dose containers, for example sealed ampoules and vials, and may be stored in a freeze-dried (lyophilised) condition requiring only the addition of the sterile liquid carrier, for example saline or water-for-injection, immediately prior to use.
- compositions for parenteral administration include injectable solutions or suspensions which can contain, for example, suitable non-toxic, parenterally acceptable diluents or solvents, such as mannitol, 1,3-butanediol, water, Ringer's solution, an isotonic sodium chloride solution, or other suitable dispersing or wetting and suspending agents, including synthetic mono- or diglycerides, and fatty acids, including oleic acid, or Cremaphor.
- suitable non-toxic, parenterally acceptable diluents or solvents such as mannitol, 1,3-butanediol, water, Ringer's solution, an isotonic sodium chloride solution, or other suitable dispersing or wetting and suspending agents, including synthetic mono- or diglycerides, and fatty acids, including oleic acid, or Cremaphor.
- compositions for nasal aerosol or inhalation administration include solutions in saline, which can contain, for example, benzyl alcohol or other suitable preservatives, absorption promoters to enhance bioavailability, and/or other solubilizing or dispersing agents such as those known in the art.
- Formulations for rectal administration may be presented as a suppository with the usual carriers such as cocoa butter, synthetic glyceride esters or polyethylene glycol. Such carriers are typically solid at ordinary temperatures, but liquify and/or dissolve in the rectal cavity to release the drug.
- Formulations for topical administration in the mouth include lozenges comprising the active ingredient in a flavoured basis such as sucrose and acacia or tragacanth, and pastilles comprising the active ingredient in a basis such as gelatin and glycerine or sucrose and acacia.
- exemplary compositions for topical administration include a topical carrier such as Plastibase (mineral oil gelled with polyethylene).
- Preferred unit dosage formulations are those containing an effective dose, as hereinbefore recited, or an appropriate fraction thereof, of the active ingredient.
- formulations of this invention may include other agents conventional in the art having regard to the type of formulation in question, for example those suitable for oral administration may include flavouring agents.
- a compound of the invention may be used as the sole active ingredient in a medicament, it is also possible for the compound to be used in combination with one or more further active agents.
- Such further active agents may be further compounds according to the invention, or they may be different therapeutic agents, for example an anti-dyslipidemic agent or other pharmaceutically active material.
- the compounds of the present invention may be employed in combination with one or more other modulators and/or ligands of the thyroid receptor or one or more other suitable therapeutic agents selected from the group consisting of cholesterol/ lipid lowering agents, hypolipidemic agents, anti- atherosclerotic agents, anti-diabetic agents, anti-osteoporosis agents, anti-obesity agents, growth promoting agents, anti-inflammatory agents, anti-anxiety agents, anti-depressants, anti-hypertensive agents, cardiac glycosides, appetite supressants, bone resorption inhibitors, thyroid mimetics, anabolic agents, anti-tumor agents and retinoids.
- suitable therapeutic agents selected from the group consisting of cholesterol/ lipid lowering agents, hypolipidemic agents, anti- atherosclerotic agents, anti-diabetic agents, anti-osteoporosis agents, anti-obesity agents, growth promoting agents, anti-inflammatory agents, anti-anxiety agents, anti-depressants,
- hypolipidemic agents for use in combination with the compounds of the present invention also include ezetimibe, simvastatin, atorvastatin, rosuvastatin, cerivastatin, fluvastatin, lovastatin, pravastatin, fenofibrate, gemfibrozil and bezafibrate.
- Suitable anti-diabetic agents for use in combination with the compounds of the present invention include biguanides (e.g., metformin or phenformin), glucosidase inhibitors (e.g,. acarbose or miglitol), insulins (including insulin secretagogues or insulin sensitizers), meglitinides (e.g., repaglinide), sulfonylureas (e.g., glimepiride, glyburide, glipyride, gliclazide, chlo ⁇ ropamide and glipizide), biguanide/glyburide combinations (e.g., Glucovance®), thiazolidinediones (e.g., troglitazone, rosiglitazone, englitazone, darglitazone and pioglitazone), PPAR-alpha agonists, PPAR-gamma agonists, PPAR alpha/gamm
- Suitable anti-osteoporosis agents for use in combination with the compounds of the present invention include alendronate, risedronate, PTH, PTH fragment, raloxifene, calcitonin, RANK ligand antagonists, calcium sensing receptor antagonists, TRAP inhibitors, selective estrogen receptor modulators (SERM) and AP-1 inhibitors.
- Suitable anti-obesity agents for use in combination with the compounds of the present invention include aP2 inhibitors, PPAR gamma antagonists, PPAR delta agonists, beta 3 adrenergic agonists, such as AJ9677 (Takeda/Dainippon), L750355 (Merck), or CP331648 (Pfizer) or other known beta 3 agonists as disclosed in U.S. Patent Nos.
- a lipase inhibitor such as orlistat or ATL-962 (Alizyme)
- a serotonin (and dopamine) reuptake inhibitor such as sibutramine, topiramate (Johnson & Johnson) or axokine (Regeneron)
- other thyroid receptor beta drugs such as a thyroid receptor ligand as disclosed in WO 97/21993 (U. Cal SF), WO 99/00353 (KaroBio) and GB98/284425 (KaroBio), CB-1 (cannabinoid receptor) antagonists (see G.
- Colombo et al "Appetite Suppression and Weight Loss After the Cannabionid Antagonist SR 141716", Life Sciences, Vol 63, PL 113-117 (1998)) and/or an anorectic agent, such as dexamphetamine, phentermine, phenylpropanolamine or mazindol.
- the compounds of the present invention may be combined with growth promoting agents, such as, but not limited to, TRH, diethylstilbesterol, theophylline, enkephalins, E series prostaglandins, compounds disclosed in U.S. Patent No. 3,239,345, e.g., zeranol, and compounds disclosed in U.S. Patent No.4,036,979, e.g., sulbenox or peptides disclosed in U.S. Patent No. 4,411,890.
- growth promoting agents such as, but not limited to, TRH, diethylstilbesterol, theophylline, enkephalins, E series prostaglandins, compounds disclosed in U.S. Patent No. 3,239,345, e.g., zeranol, and compounds disclosed in U.S. Patent No.4,036,979, e.g., sulbenox or peptides disclosed in U.S. Patent No. 4,411,890
- the compounds of the invention may also be used in combination with growth hormone secretagogues such as GHRP-6, GHRP-1 (as described in U.S. Patent No. 4,411,890 and publications WO 89/07110 and WO 89/07111), GHRP-2 (as described in WO 93/04081), NN703 (Novo Nordisk), LY444711 (Lilly), MK-677 (Merck), CP424391 (Pfizer) and B-HT920, or with growth hormone releasing factor and its analogs or growth hormone and its analogs or somatomedins including IGF-1 and IGF-2, or with alpha-adrenergic agonists, such as clonidine or serotinin 5-HT D agonists, such as sumatriptan, or agents which inhibit somatostatin or its release, such as physostigmine and pyridostigmine.
- growth hormone secretagogues such as GHRP-6, GHRP-1 (as described in U.
- Suitable anti-inflammatory agents for use in combination with the compounds of the present invention include prednisone, dexamethasone, Enbrel®, cyclooxygenase inhibitors (i.e., COX-1 and/or COX -2 inhibitors such as NSAIDs, aspirin, indomethacin, ibuprofen, piroxicam, Naproxen®, Celebrex®, Vioxx®), CTLA4-Ig agonists/antagonists, CD40 ligand antagonists, IMPDH inhibitors, such as mycophenolate (CellCept®), integrin antagonists, alpha-4 beta-7 integrin antagonists, cell adhesion inhibitors, interferon gamma antagonists, ICAM-1, tumor necrosis factor (TNF) antagonists (e.g., infliximab, OR 1384), prostaglandin synthesis inhibitors, budesonide, clofazimine, CNI-1493, CD4 antagonists (e.g., pri
- Example of suitable anti-anxiety agents for use in combination with the compounds of the present invention include diazepam, lorazepam, buspirone, oxazepam, and hydroxyzine pamoate.
- Suitable anti-depressants for use in combination with the compounds of the present invention include citalopram, fluoxetine, nefazodone, sertraline, and paroxetine.
- Suitable anti-hypertensive agents for use in combination with the compounds of the present invention include beta adrenergic blockers, calcium channel blockers (L-type and T-type; e.g. diltiazem, verapamil, nifedipine, amlodipine and mybefradil), diuretics (e.g., chlorothiazide, hydrochlorothiazide, flumethiazide, hydroflumethiazide, bendroflumethiazide, methylchlorothiazide, trichloromethiazide, polythiazide, benzthiazide, ethacrynic acid tricrynafen, chlorthalidone, furosemide, musolimine, bumetanide, triamtrenene, amiloride, spironolactone), renin inhibitors, ACE inhibitors (e.g., captopril, zofenopril, fosino
- Dual ET/AII antagonist e.g., compounds disclosed in WO 00/01389
- neutral endopeptidase (NEP) inhibitors neutral endopeptidase (NEP) inhibitors
- vasopepsidase inhibitors dual NEP-ACE inhibitors
- omapatrilat and gemopatrilat e.g., omapatrilat and gemopatrilat
- Suitable cholesterol/lipid lowering agents for use in combination with the compounds of the present invention include HMG-CoA reductase inhibitors, squalene synthetase inhibitors, fibrates, bile acid sequestrants, ACAT inhibitors, MTP inhibitors, lipooxygenase inhibitors, an ileal NaVbile acid cotransporter inhibitor, cholesterol absorption inhibitors, and cholesterol ester transfer protein inhibitors (e.g., CP-529414).
- MTP inhibitors which may be employed herein in combination with one or more compounds of formula I include MTP inhibitors as disclosed in U.S. Patent No. 5,595,872, U.S. Patent No. 5,739,135, U.S. Patent No. 5,712,279, U.S. Patent No. 5,760,246, U.S. Patent No. 5,827,875, U.S. Patent No. 5,885,983 and U.S. Patent No. 5,962,440 all inco ⁇ orated herein by reference.
- HMG CoA reductase inhibitors which may be employed in combination with one or more compounds of formula I include mevastatin and related compounds as disclosed in U.S . Patent No. 3,983,140, lovastatin (mevinolin) and related compounds as disclosed in U.S. Patent No. 4,231,938, pravastatin and related compounds such as disclosed in U.S. Patent No. 4,346,227, simvastatin and related compounds as disclosed in U.S. Patent Nos. 4,448,784 and 4,450,171.
- Further HMG CoA reductase inhibitors which may be employed herein include fluvastatin, disclosed in U.S. Patent No. 5,354,772, cerivastatin disclosed in U.S. Patent Nos.
- the squalene synthetase inhibitors which may be used in combination with the compounds of the present invention include, but are not limited to, ⁇ -phosphono-sulfonates disclosed in U.S. Patent No. 5,712,396, those disclosed by Biller et al, J. Med. Chem., 1988, Vol. 31, No. 10, pp 1869-1871, including isoprenoid (phos ⁇ hinylmethyl)phosphonates, te ⁇ enoid pyrophosphates disclosed by P. Ortiz de Montellano et al, J. Med.
- Bile acid sequestrants which may be used in combination with the compounds of the present invention include cholestyramine, colestipol and DEAE-Sephadex (Secholex®, policexide®), as well as lipostabil (Rhone-Poulenc), Eisai E-5050 (an N-substituted ethanolamine derivative), imanixil (HOE-402), tetrahydrolipstatin (THL), istigmastanylphos-phorylcholine (SPC, Roche), aminocyclodextrin (Tanabe Seiyoku), Ajinomoto AJ-814 (azulene derivative), melinamide
- ACAT inhibitors suitable for use in combination with compounds of the invention include ACAT inhibitors as described in, Drugs of the Future 24, 9-15 (1999), (Avasimibe); "The ACAT inhibitor, Cl-1011 is effective in the prevention and regression of aortic fatty streak area in hamsters", Nicolosi et al, Atherosclerosis (Shannon, Irel). (1998), 137(1), 77-85; "The pharmacological profile of FCE 27677: a novel ACAT inhibitor with potent hypolipidemic activity mediated by selective suppression of the hepatic secretion of ApoBlOO-containing lipoprotein", Ghiselli, Giancarlo, Cardiovasc. Drug Rev.
- Suitable cholesterol absorption inhibitor for use in combination with the compounds of the invention include SCH48461 (Schering-Plough), as well as those disclosed in Atherosclerosis 1 15, 45-63 (1995) and J. Med. Chem. 41, 973 (1998).
- ileal Na + / bile acid cotransporter inhibitors for use in combination with the compounds of the invention include compounds as disclosed in Drugs of the Future, 24, 425-430 (1999).
- suitable thyroid mimetics for use in combination with the compounds of the present invention include thyrotropin, polythyroid, KB-130015, and dronedarone.
- the compounds of the present invention may be used alone or optionally in combination with a retinoid, such as tretinoin, or a vitamin D analog.
- a still further use of the compounds of the invention is in combination with estrogen, testosterone, a selective estrogen receptor modulator, such as tamoxifen or raloxifene, or other androgen receptor modulators, such as those disclosed in Edwards, J. P. et al., Bio. Med. Chem. Let, 9, 1003-1008 (1999) and Hamann, L. G. et al., J. Med. Chem., 42, 210-212 (1999).
- a selective estrogen receptor modulator such as tamoxifen or raloxifene
- other androgen receptor modulators such as those disclosed in Edwards, J. P. et al., Bio. Med. Chem. Let, 9, 1003-1008 (1999) and Hamann, L. G. et al., J. Med. Chem., 42, 210-212 (1999).
- a further use of the compounds of this invention is in combination with steriodal or non-steroidal progesterone receptor agonists ("PRA”), such as levonorgestrel, medroxyprogesterone acetate (MPA).
- PRA steriodal or non-steroidal progesterone receptor agonists
- MPA medroxyprogesterone acetate
- the compounds of formula I When combined with a hypolypidemic agent, an antidepressant, a bone resorption inhibitor and/or an appetite suppressant, the compounds of formula I may be employed in a weight ratio to the additional agent within the range from about 500: 1 to about 0.005: 1 , preferably from about 300: 1 to about 0.01:1. Where the antidiabetic agent is a biguanide, the compounds of formula I may be employed in a weight ratio to biguanide within the range from about 0.01:1 to about 100:1, preferably from about 0.5:1 to about 2:1.
- the compounds of formula I may be employed in a weight ratio to a glucosidase inhibitor within the range from about 0.01:1 to about 100:1, preferably from about 0.5:1 to about 50:1.
- the compounds of formula I may be employed in a weight ratio to a sulfonylurea in the range from about 0.01:1 to about 100:1, preferably from about 0.2:1 to about 10:1.
- the compounds of formula I may be employed in a weight ratio to a thiazolidinedione in an amount within the range from about 0.01:1 to about 100:1, preferably from about 0.5:1 to about 5:1.
- the thiazolidinedione may be employed in amounts within the range from about 0.01 to about 2000 mg/day, which may optionally be administered in single or divided doses of one to four times per day.
- these additional agents may be inco ⁇ orated into a combined single tablet with a therapeutically effective amount of the compounds of formula I.
- Metformin, or salt thereof may be employed with the compounds of formula I in amounts within the range from about 500 to about 2000 mg per day, which may be administered in single or divided doses one to four times daily.
- the compounds of formula I may be employed in a weight ratio to a PPAR-alpha agonist, a PPAR- gamma agonist, a PPAR-alpha/gamma dual agonist, an SGLT2 inhibitor and/or an aP2 inhibitor within the range from about 0.01:1 to about 100:1, preferably from about 0.5:1 to about 5:1.
- An MTP inhibitor may be administered orally with the compounds of formula I in an amount within the range of from about 0.01 mg/kg to about 100 mg/kg and preferably from about 0.1 mg/kg to about 75 mg/kg, one to four times daily.
- a preferred oral dosage form such as tablets or capsules, may contain the MTP inhibitor in an amount of from about 1 to about 500 mg, preferably from about 2 to about 400 mg, and more preferably from about 5 to about 250 mg, administered on a regimen of one to four times daily.
- the MTP inhibitor may be employed in an amount within the range of from about 0.005 mg/kg to about 10 mg/kg and preferably from about 0.005 mg/kg to about 8 mg/kg, administered on a regimen of one to four times daily.
- a HMG CoA reductase inhibitor may be administered orally with the compounds of formula I within the range of from about 1 to 2000 mg, and preferably from about 4 to about 200 mg.
- a preferred oral dosage form, such as tablets or capsules, will contain the HMG CoA reductase inhibitor in an amount from about 0.1 to about 100 mg, preferably from about 5 to about 80 mg, and more preferably from about 10 to about 40 mg.
- a squalene synthetase inhibitor may be administered with the compounds of formula I within the range of from about 10 mg to about 2000 mg and preferably from about 25 mg to about 200 mg.
- a preferred oral dosage form, such as tablets or capsules, will contain the squalene synthetase inhibitor in an amount of from about 10 to about 500 mg, preferably from about 25 to about 200 mg.
- the compounds of formula (I) as described above also find use, optionally in labelled form, as a diagnostic agent for the diagnosis of conditions associated with malfunction of the thyroid receptor.
- a diagnostic agent for the diagnosis of conditions associated with malfunction of the thyroid receptor.
- such a compound may be radioactively labelled.
- the compounds of formula (I) as described above, optionally in labelled form, also find use as a reference compound in methods of discovering other antagonists or partial antagonists of the thyroid receptor.
- the invention provides a method of discovering a ligand of the thyroid receptor which comprises use of a compound of the invention or a compound of the invention in labelled form, as a reference compound.
- a method may involve a competitive binding experiment in which binding of a compound of formula (I) to the thyroid receptor is reduced by the presence of a further compound which has thyroid receptor-binding characteristics, for example stronger thyroid receptor-binding characteristics than the compound of formula (I) in question.
- the invention also provides a method for preparing a compound in of formula (I) accordance with the invention as described above comprising a step of reacting - a compound of formula (II)
- R 2 , n, Y', Y, R 3 , R 4 , W and R 5 are as defined above
- Suitable leaving groups L include halogen, OR c , -SR°, CM lkyl, C 5 . ⁇ 0 aryl or C 5 . ⁇ 0 aryl-C M lkyl sulphonate esters, for example, a bromide, a methylsulfonyl or a toluenesufonyl group.
- Particularly preferred compounds R'-L are acid chlorides (R 6 COCl) and sulphonylchlorides (R 6 S0 2 C1) ie reagents in which the leaving group L is CI.
- Suitable bases include carbonates, alkylamines and alkali metal hydroxides, for example potassium carbonate, cesium carbonate, potassium hydroxide, sodium hydroxide diisopropylamine and triethylamine. Trimethylsilanoate may also be used. Other combinations of leaving groups and bases may be employed, as is known by the person skilled in the art.
- one or more coupling reagents may be employed. The reaction mixture is stirred at room temperature, or heated until the starting materials have been consumed. The reaction may be carried out with protecting groups present and those protecting groups may be removed after the reaction. Suitable protecting groups are known to the person skilled in the art (see T. W. Greene, "Protective Groups in Organic Synthesis", 3 rd Edition, New York, 1999).
- Methyl 3-(4-hydroxy-3,5-dichIorophenyl) propionate Methyl-3-(4-hydroxyphenyl) propionate (35.6 g, 0.198 mol) was dissolved in dichloromethane (200 mL). The reaction mixture was cooled to 4°C and sulfuryl chloride (120 mL, 1.42 mol) in diethyl ether (200 mL) was added drop wise to the reaction mixture over 1 h. After 3 h at room temperature the solvent was removed. The reaction mixture was dissolved in dichloromethane and washed with water. The combined organic phases were dried over sodium sulphate, filtered and evaporated. The product was purified by flash chromatography (diethyl ether/heptane) to provide 16.6 g (34%) of the title compound.
- the resin was filtered off, and the dichloromethane / tetrahydrofuran solution was run through a short silica based amine column (Isolute, lg, 0.6 mmol/g) to remove unreacted acid chloride.
- the column was rinsed with dichloromethane (2 ml), and the combined eluates were evaporated.
- the material was dissolved in tetrahydrofuran (0.5 ml) and lithium hydroxide (1 M, 1 ml) was added. The mixture was stirred over night at room temperature.
- reaction mixture was separated by semi-preparative-HPLC (Zorbax CombiHT (SB-C8 50x21.2 mm, 5 ⁇ ) Mobile Phase: Solvent A: Water with 0.5% formic acid; Solvent B: acetonitrile. Gradient: 20-100%) acetonitrile gradient). Appropriate fractions were combined and evaporated to give the expected acid (e.g. 3-[3,5-dihalo-4-(3-acetylamino-benzyloxy)phenyl] propionic acid).
- the dichloromethane solution of the appropriate aniline (e.g. methyl 3-(3,5-dihalo-4-(3- aminobenzyloxy)phenyl) propionate) (1 eq.) was treated with the appropriate sulphonylchloride (R 6 S0 2 C1) (4 eq.) and pyridine (2.5 eq.). The mixture was stirred at 40°C for 2 h. Water was added and the mixture was acidified with hydrochloric acid (1 M) and extracted with dichloromethane (3 x 25 mL). The organic phases were combined, the solvent was removed under vacuum and the residue was purified by flash chromatography to provide the desired sulphonamide (e.g. methyl 3-[3,5- dihalo-4-(3-methanesulfonyla_nino-benzyloxy)phenyl] propionate).
- aniline e.g. methyl 3-(3,5-dihalo-4-(3- aminobenzyloxy)phenyl)
- the sulphonamide (e.g. methyl 3-[3,5-dihalo-4-(3-methanesulfonylamino-benzyloxy)phenyl] propionate) was dissolved in dioxane (7 mL/mmol ester), sodium hydroxide (lithium hydroxide has also been used) (1 N in water, 5 eq.) was added and the mixture was stirred at room temperature over night. After acidification with hydrochloric acid (1 N) the product was extracted into ethyl acetate. The solvent was evaporated under vacuum to yield the corresponding acid (e.g. 3-[3,5- dihalo-4-(3 -methanesulfonylamino-benzyloxy)phenyl] propionic acid).
- NBS N-Bromosuccinimide
- ACN acetonitrile
- DMPU 1 ,3-dimethyl-3,4,5,6-tetrahydro-2(l__)-pyrimidone
- PBr 3 phosphorus tribromide
- CC1 4 tetrachloromethane
- mice were placed on a special diet protocol (Purina chow supplemented with 1.5%) cholesterol, 15% saturated fat and 0.5%> cholic acid) for two weeks before administration of drugs.
- the animals were housed at room temperature, 12:12 light dark cycle, and free access to food and water.
- On the day of treatment all animals were weighed before drug was administrated by intraperitoneal injection or by gavage. Compounds were administrated once daily for 5-10 days, at different concentrations (nmol/kg body weight), in suitable vehicle.
- food was removed from the cages and the animals were fasted for at least 4 hours before termination of the study. Blood for serum or plasma was collected, and different organs were dissected and immediately frozen for later analyses. Blood and tissue lipid analyses were consecutively executed using commercial and readily available kits for the determination.
- mice 6-8 weeks old female ob/ob mice (i.e. leptin deficient mice) purchased from commercial supplier were used to characterize compounds binding to thyroid hormone receptors alpha (TR ⁇ ) and beta (TR ⁇ ).
- TR ⁇ thyroid hormone receptors alpha
- TR ⁇ beta
- the animals were weighed and randomly divided into different study groups, and kept for a minimum of 5 days to adapt to the new environment (animal facility). The animals were housed at room temperature, 12:12 light dark cycle, and free access to food and water. On the day of treatment all animals were weighed before drug was administrated by intraperitoneal injection or by gavage. Compounds were administrated once daily for 5-10 days, at different concentrations (nmol/kg body weight), in suitable vehicle.
- assays to determine thyroid receptor mediated activity of the test compounds include assays that demonstrate modulation of endogenous TR mediated transcription in cell culture systems; assays that demonstrate modulation of thyroid responsive tissue effects in rodents; assays for the identification of receptor surface conformation changes; and assays that demonstrate binding specificity to TR versus other nuclear receptors.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Physical Education & Sports Medicine (AREA)
- Diabetes (AREA)
- Neurology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Cardiology (AREA)
- Rheumatology (AREA)
- Heart & Thoracic Surgery (AREA)
- Psychiatry (AREA)
- Endocrinology (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Hospice & Palliative Care (AREA)
- Pain & Pain Management (AREA)
- Immunology (AREA)
- Pulmonology (AREA)
- Ophthalmology & Optometry (AREA)
- Dermatology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Child & Adolescent Psychology (AREA)
- Emergency Medicine (AREA)
- Nutrition Science (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05716286A EP1732532A1 (en) | 2004-03-22 | 2005-03-22 | Novel pharmaceutical compositions comprising agonists of the thyroid receptor |
| BRPI0509074-1A BRPI0509074A (en) | 2004-03-22 | 2005-03-22 | pharmaceutical compositions comprising thyroid receptor agonists |
| US10/593,927 US20080004251A1 (en) | 2004-03-22 | 2005-03-22 | Novel Pharmaceutical Compositions Comprising Agonists of the Thyroid Receptor |
| CA002560526A CA2560526A1 (en) | 2004-03-22 | 2005-03-22 | Novel pharmaceutical compositions comprising agonists of the thyroid receptor |
| AU2005226914A AU2005226914A1 (en) | 2004-03-22 | 2005-03-22 | Novel pharmaceutical compositions comprising agonists of the thyroid receptor |
| JP2007504341A JP2007529566A (en) | 2004-03-22 | 2005-03-22 | Novel pharmaceutical composition comprising an agonist of thyroid receptor |
| IL177665A IL177665A0 (en) | 2004-03-22 | 2006-08-23 | Novel pharmaceutical compositions comprising agonists of the thyroid receptor |
| NO20064705A NO20064705L (en) | 2004-03-22 | 2006-10-18 | New pharmaceutical preparations comprising agonists of the thyroid receptor |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB0406378.0A GB0406378D0 (en) | 2004-03-22 | 2004-03-22 | Novel pharmaceutical compositions |
| GB0406378.0 | 2004-03-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2005092316A1 true WO2005092316A1 (en) | 2005-10-06 |
Family
ID=32118142
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2005/003030 WO2005092316A1 (en) | 2004-03-22 | 2005-03-22 | Novel pharmaceutical compositions comprising agonists of the thyroid receptor |
Country Status (12)
| Country | Link |
|---|---|
| US (1) | US20080004251A1 (en) |
| EP (1) | EP1732532A1 (en) |
| JP (1) | JP2007529566A (en) |
| KR (1) | KR20060130728A (en) |
| CN (1) | CN1933826A (en) |
| AU (1) | AU2005226914A1 (en) |
| BR (1) | BRPI0509074A (en) |
| CA (1) | CA2560526A1 (en) |
| GB (1) | GB0406378D0 (en) |
| IL (1) | IL177665A0 (en) |
| NO (1) | NO20064705L (en) |
| WO (1) | WO2005092316A1 (en) |
Cited By (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1688138A1 (en) * | 2003-11-26 | 2006-08-09 | Takeda Pharmaceutical Company Limited | Receptor function regulating agent |
| WO2007039125A2 (en) * | 2005-09-21 | 2007-04-12 | Karo Bio Ab | An optically active thyroid receptor agonist and optically active key intermediates in its production |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| WO2012030165A2 (en) | 2010-08-31 | 2012-03-08 | 서울대학교산학협력단 | Use of the fetal reprogramming of a ppar δ agonist |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2017184811A1 (en) * | 2016-04-22 | 2017-10-26 | Metabasis Therapeutics, Inc. | Thyroid hormone receptor agonist and use thereof |
| CN113134086A (en) * | 2020-01-20 | 2021-07-20 | 深圳市长卿医学研究院 | Pharmaceutical composition for reducing blood fat |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2872159B1 (en) * | 2004-06-28 | 2007-10-05 | Merck Sante Soc Par Actions Si | NOVEL PHENYL CARBOXYLIC ACID DERIVATIVES AND THEIR USE IN THE TREATMENT OF DIABETES |
| CN112300133B (en) * | 2019-07-31 | 2024-07-30 | 深圳微芯生物科技股份有限公司 | Heterocyclic compound and application thereof |
| WO2023177667A1 (en) * | 2022-03-16 | 2023-09-21 | Brii Biosciences, Inc. | Thyroid hormone receptor agonists |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001036365A2 (en) * | 1999-11-17 | 2001-05-25 | Karo Bio Ab | Thyroid receptor antagonists for the treatment of cardiac and metabolic disorders |
| WO2001060784A1 (en) * | 2000-02-17 | 2001-08-23 | Bristol-Myers Squibb Co. | Aniline-derived ligands for the thyroid receptor |
| EP1148054A1 (en) * | 2000-04-21 | 2001-10-24 | Pfizer Products Inc. | Thyroid receptor ligands |
| WO2003059872A1 (en) * | 2001-12-31 | 2003-07-24 | Bayer Pharmaceuticals Corporation | Avb3 and avb5 integrin antagonists and methods of treating diseases or conditions associated with avb3 and avb5 integrins |
| WO2003094845A2 (en) * | 2002-05-08 | 2003-11-20 | Bristol-Myers Squibb Company | Pyridine-based thyroid receptor ligands |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1174051A (en) * | 1965-11-30 | 1969-12-10 | Beecham Group Ltd | Aminophenol Derivatives and Analgesic and Antipyretic Compositions |
| BE689347A (en) * | 1965-12-09 | 1967-04-14 | Madan Ag | |
| AU4154799A (en) * | 1998-06-02 | 1999-12-20 | Arthromics Plc | Compounds which interact with the thyroid hormone receptor for the treatment of fibrotic disease |
| GB9816935D0 (en) * | 1998-08-05 | 1998-09-30 | Karobio Ab | Novel glucocortoid and thyroid receptor ligands for the treatment of metabolic disorders |
| US6395784B1 (en) * | 2000-06-07 | 2002-05-28 | Bristol-Myers Squibb Company | Benzamide ligands for the thyroid receptor |
| GB0015205D0 (en) * | 2000-06-21 | 2000-08-09 | Karobio Ab | Bioisosteric thyroid receptor ligands and method |
| GB0215978D0 (en) * | 2002-07-10 | 2002-08-21 | Karobio Ab | Novel compounds |
| GB0406380D0 (en) * | 2004-03-22 | 2004-04-21 | Karobio Ab | Novel pharmaceutical compositions |
-
2004
- 2004-03-22 GB GBGB0406378.0A patent/GB0406378D0/en not_active Ceased
-
2005
- 2005-03-22 CN CNA2005800091283A patent/CN1933826A/en active Pending
- 2005-03-22 CA CA002560526A patent/CA2560526A1/en not_active Abandoned
- 2005-03-22 US US10/593,927 patent/US20080004251A1/en not_active Abandoned
- 2005-03-22 JP JP2007504341A patent/JP2007529566A/en active Pending
- 2005-03-22 EP EP05716286A patent/EP1732532A1/en not_active Withdrawn
- 2005-03-22 BR BRPI0509074-1A patent/BRPI0509074A/en not_active IP Right Cessation
- 2005-03-22 AU AU2005226914A patent/AU2005226914A1/en not_active Abandoned
- 2005-03-22 WO PCT/EP2005/003030 patent/WO2005092316A1/en active Application Filing
- 2005-03-22 KR KR1020067021050A patent/KR20060130728A/en not_active Application Discontinuation
-
2006
- 2006-08-23 IL IL177665A patent/IL177665A0/en unknown
- 2006-10-18 NO NO20064705A patent/NO20064705L/en not_active Application Discontinuation
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001036365A2 (en) * | 1999-11-17 | 2001-05-25 | Karo Bio Ab | Thyroid receptor antagonists for the treatment of cardiac and metabolic disorders |
| WO2001060784A1 (en) * | 2000-02-17 | 2001-08-23 | Bristol-Myers Squibb Co. | Aniline-derived ligands for the thyroid receptor |
| EP1148054A1 (en) * | 2000-04-21 | 2001-10-24 | Pfizer Products Inc. | Thyroid receptor ligands |
| WO2003059872A1 (en) * | 2001-12-31 | 2003-07-24 | Bayer Pharmaceuticals Corporation | Avb3 and avb5 integrin antagonists and methods of treating diseases or conditions associated with avb3 and avb5 integrins |
| WO2003094845A2 (en) * | 2002-05-08 | 2003-11-20 | Bristol-Myers Squibb Company | Pyridine-based thyroid receptor ligands |
Non-Patent Citations (1)
| Title |
|---|
| LEESON P D ET AL: "Thyroid Hormone Analogues", JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 31, no. 1, 1988, pages 37 - 54, XP002101081, ISSN: 0022-2623 * |
Cited By (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1688138A1 (en) * | 2003-11-26 | 2006-08-09 | Takeda Pharmaceutical Company Limited | Receptor function regulating agent |
| EP1688138A4 (en) * | 2003-11-26 | 2008-01-16 | Takeda Pharmaceutical | Receptor function regulating agent |
| US8008525B2 (en) | 2003-11-26 | 2011-08-30 | Takeda Pharmaceutical Company Limited | Receptor function regulating agent |
| WO2007039125A2 (en) * | 2005-09-21 | 2007-04-12 | Karo Bio Ab | An optically active thyroid receptor agonist and optically active key intermediates in its production |
| WO2007039125A3 (en) * | 2005-09-21 | 2007-06-28 | Karobio Ab | An optically active thyroid receptor agonist and optically active key intermediates in its production |
| WO2008017381A1 (en) | 2006-08-08 | 2008-02-14 | Sanofi-Aventis | Arylaminoaryl-alkyl-substituted imidazolidine-2,4-diones, processes for preparing them, medicaments comprising these compounds, and their use |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| WO2011023754A1 (en) | 2009-08-26 | 2011-03-03 | Sanofi-Aventis | Novel crystalline heteroaromatic fluoroglycoside hydrates, pharmaceuticals comprising these compounds and their use |
| WO2012030165A2 (en) | 2010-08-31 | 2012-03-08 | 서울대학교산학협력단 | Use of the fetal reprogramming of a ppar δ agonist |
| WO2012120053A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120050A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120057A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Novel substituted phenyl-oxathiazine derivatives, method for producing them, drugs containing said compounds and the use thereof |
| WO2012120054A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120058A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives which are substituted with benzyl or heteromethylene groups, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2012120051A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Benzyl-oxathiazine derivates substituted with adamantane or noradamantane, medicaments containing said compounds and use thereof |
| WO2012120055A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Di- and tri-substituted oxathiazine derivates, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120052A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Oxathiazine derivatives substituted with carbocycles or heterocycles, method for producing same, drugs containing said compounds, and use thereof |
| WO2012120056A1 (en) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituted oxathiazine derivatives, method for producing them, their use as medicine and drug containing said derivatives and the use thereof |
| WO2017184811A1 (en) * | 2016-04-22 | 2017-10-26 | Metabasis Therapeutics, Inc. | Thyroid hormone receptor agonist and use thereof |
| US10420781B2 (en) | 2016-04-22 | 2019-09-24 | Metabasis Therapeutics Inc. | Thyroid hormone receptor agonist and use thereof |
| CN113134086A (en) * | 2020-01-20 | 2021-07-20 | 深圳市长卿医学研究院 | Pharmaceutical composition for reducing blood fat |
| CN113134086B (en) * | 2020-01-20 | 2024-05-24 | 深圳市长卿医学研究院 | Pharmaceutical composition for reducing blood fat |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20060130728A (en) | 2006-12-19 |
| BRPI0509074A (en) | 2007-08-21 |
| GB0406378D0 (en) | 2004-04-21 |
| IL177665A0 (en) | 2008-03-20 |
| JP2007529566A (en) | 2007-10-25 |
| CA2560526A1 (en) | 2005-10-06 |
| AU2005226914A1 (en) | 2005-10-06 |
| CN1933826A (en) | 2007-03-21 |
| US20080004251A1 (en) | 2008-01-03 |
| NO20064705L (en) | 2006-12-19 |
| EP1732532A1 (en) | 2006-12-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20080004251A1 (en) | Novel Pharmaceutical Compositions Comprising Agonists of the Thyroid Receptor | |
| US20080221210A1 (en) | Thyroid Receptor Agonists | |
| US20100004271A1 (en) | Heterocyclic compounds as aganist for the thyroid receptor | |
| US20090318514A1 (en) | Novel Pharmaceutical Compositions | |
| US6831102B2 (en) | Phenyl naphthol ligands for thyroid hormone receptor | |
| US8263659B2 (en) | Thyroid receptor ligands | |
| US20090233979A1 (en) | Indole Derivatives and Their Use as Thyroid Receptor Ligands | |
| US6747048B2 (en) | Pyridine-based thyroid receptor ligands | |
| US6395784B1 (en) | Benzamide ligands for the thyroid receptor | |
| WO2007039125A2 (en) | An optically active thyroid receptor agonist and optically active key intermediates in its production | |
| ZA200500167B (en) | Benzamide or phenylacetamide derivatives useful as thyroid receptor ligands | |
| WO2004018421A1 (en) | Idol derivatives as new thyroid receptor agonist ligands and methods | |
| US7342127B2 (en) | Substituted anilide ligands for the thyroid receptor | |
| US20040176425A1 (en) | Cycloalkyl containing anilide ligands for the thyroid receptor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SM SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IS IT LT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2005226914 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 4729/DELNP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 177665 Country of ref document: IL |
|
| ENP | Entry into the national phase |
Ref document number: 2005226914 Country of ref document: AU Date of ref document: 20050322 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005226914 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2560526 Country of ref document: CA Ref document number: 2007504341 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200580009128.3 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005716286 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020067021050 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067021050 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2005716286 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10593927 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0509074 Country of ref document: BR |
|
| WWP | Wipo information: published in national office |
Ref document number: 10593927 Country of ref document: US |