WO2005021732A2 - Organic cooling medium and its uses - Google Patents
Organic cooling medium and its uses Download PDFInfo
- Publication number
- WO2005021732A2 WO2005021732A2 PCT/US2004/028597 US2004028597W WO2005021732A2 WO 2005021732 A2 WO2005021732 A2 WO 2005021732A2 US 2004028597 W US2004028597 W US 2004028597W WO 2005021732 A2 WO2005021732 A2 WO 2005021732A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cooling medium
- mixtures
- cooling
- group
- sugar
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K5/00—Heat-transfer, heat-exchange or heat-storage materials, e.g. refrigerants; Materials for the production of heat or cold by chemical reactions other than by combustion
- C09K5/08—Materials not undergoing a change of physical state when used
- C09K5/10—Liquid materials
Definitions
- the present invention relates to cooling mediums for use in various applications including, without limitation, the cooling of ice-skating rinks and in building sprinkler systems, cold storage systems, plate freezers, blast freezers, brine freezers, cooling towers, air conditioners and condensers, radiator cooling applications, and heat exchangers.
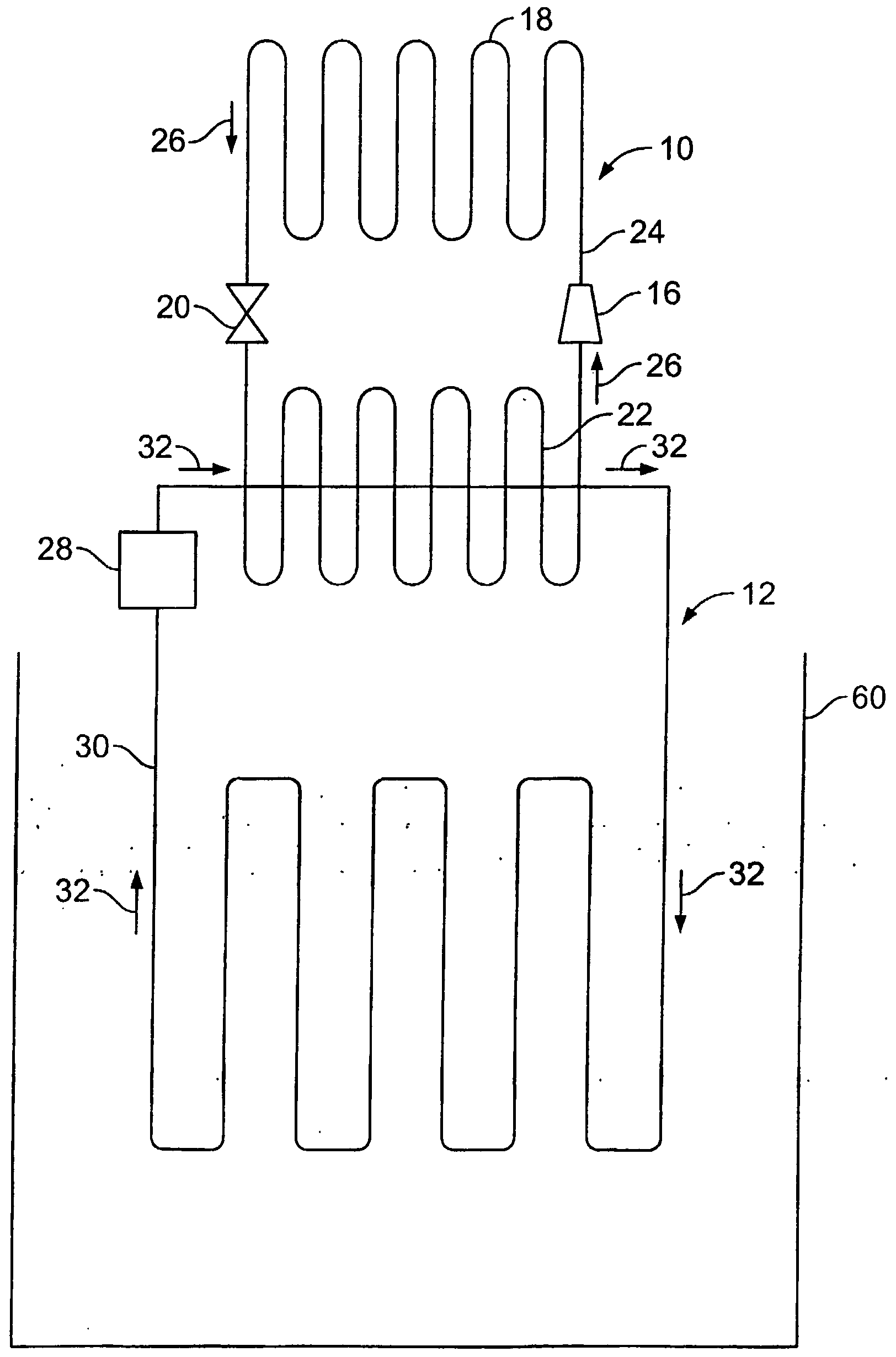
- FIG. 1 An exemplary use of the invention is depicted generally in Figure 1 wherein there is depicted a primary refrigeration system 10 opera tively connected to a secondary refrigeration system 12, which may be used in numerous applications including the cooling of an ice skating rink 14.
- the primary refrigeration system 10 is a closed loop system that circulates refrigerant through the system 10 and includes the use of a compressor 16, a condenser 18, an expansion valve 20, an evaporator 22, and tube 24.
- the refrigerant flows through tube 24, as indicated by direction arrow 26, through the compressor 16, which raises the pressure of the refrigerant.
- the refrigerant then flows through the condenser 18, where the refrigerant condenses from vapor form to liquid form, giving off heat in the process.
- the secondary refrigeration system 12 includes a pump 28 and a length of tube 30 that is positioned underneath the surface of the ice-skating rink 14.
- the pump 28 circulates the cooling medium of the invention, as described below, through the length of tube 30 as indicated by direction arrow 32 to cool the ice-skating rink and maintain the proper ice temperature.
- FIG. 2 Another exemplary use of the invention is depicted generally in Figure 2 wherein there is depicted generally a sprinkler system 48 that may be installed in a building 50.
- the sprinkler system 48 operates when there is a fire situation and provides a fluid to the fire to extinguish the fire.
- the building 50 is generally depicted but may include all types of buildings that use or have installed a sprinkler system.
- the sprinkler system 48 includes generally a fluid line or tube 52 operatively connected to a fluid source 54.
- the fluid source 54 provides the fluid, as indicated by direction arrow 56 to a plurality of sprinklers 58 positioned, through the building.
- the fluid In the event of a fire, the fluid is dispersed through the sprinklers 58 and onto the fire, thereby extinguishing the fire. It is known to use a salt-water solution as a fire extinguishing fluid.
- the known saltwater solution has a lower freezing point to prevent the fluid from freezing within the supply tubes in cold environments.
- the invention contemplates the use of the cooling medium, as described below, as the fluid used within the sprinkler system 48.
- the ice-rink 14 may be replaced with an open tank 60. Materials may be placed directly in the tank 60 for freezing.
- the cooling medium of the present invention provides a non-toxic alternative to conventional cooling mediums. Since the present cooling medium is non-toxic it is safer to use, especially in cooling systems which are used in the processing of foods.
- the cooling medium is further described in U.S. Serial No. 10/654,590, filed September 2, 2003, which is incorporated herein by reference.
- the invention contemplates the use of a cooling medium that includes an organic cooling agent, preferably in combination with a chloride salt.
- the chloride salt is preferably at least one selected from the group consisting of sodium chloride, magnesium chloride and calcium chloride.
- the chloride salt is preferably sodium chloride.
- Calcium chloride is believed to provide solutions that yield the lowest freezing point and is thus more generally preferred.
- the organic cooling agent may be any suitable organic cooling agent.
- Preferred cooling agents include carbohydrates having a molecular weight ranging from 180-1500, sugar alcohols having a molecular weight ranging from 180-1500, glycosides having a molecular weight ranging from 180-1500, maltodextrins, hydrogenated maltodextrins, starch hydrolyzates, hydrogenated starch hydrolyzates, and non-toxic oils, and any mixture of the foregoing and of other organic cooling agents.
- carbohydrates having a molecular weight ranging from 180-1500 sugar alcohols having a molecular weight ranging from 180-1500
- glycosides having a molecular weight ranging from 180-1500
- maltodextrins maltodextrins
- hydrogenated maltodextrins starch hydrolyzates
- non-toxic oils any mixture of the foregoing and of other organic cooling agents.
- an oil is used, it is not used in conjunction with a chloride salt or aqueous medium.
- At least some of the foregoing organic cooling agents are believed to provide beneficial effects when used in conjunction with the freezing of edible marine animals and other food products, in that preservation of the mucosal layer is believed to be enhanced, although the invention should not be deemed limited to any such enhanced effect
- certain of the foregoing cooling agents are believed to contribute to a lowering of freezing point in addition to the colligative lowering contributed by the presence of the dissolved material in solution.
- the carbohydrate may be any suitable carbohydrate, and may include, for instance, glucose, maltose, maltotriose, lactose, fructose, sucrose, and mixtures thereof.
- the sugar may be selected from monosaccharides, disaccharides, trisaccharides, tetrasaccharides, pentasaccharides, hexasaccharides, and mixtures thereof.
- Sugar alcohols of the foregoing, such as sorbitol and maltitol, or other sugar alcohols in the weight range provided may be used in conjunction with the invention.
- the carbohydrate may be provided as a pure solution, but ordinarily is provided in admixture with other materials, in that an industrial waste stream that includes carbohydrates may be used as a source of such carbohydrates.
- Carbohydrates can be obtained from a wide range of agricultural based products such as those derived from corn, wheat, barley, oats, sugar cane, sugar beets etc.
- Suitable sugars include, but are not limited to, corn sugar, cane sugar, beet sugar, sorghum sugar, maple sugar, wheat sugar, tapioca sugar, potato sugar, cassava sugar, and manioca sugar.
- the cooling medium has a freezing point of at least -5F or less, in another aspect -10 F or less, in another aspect -20F or less, in another aspect - 25F or less, in another aspect -30F or less, in another aspect -35F or less, in another aspect -40F or less, in another aspect -45F or less, in another aspect -50F or less, in another aspect -55F or less, in another aspect -60F or less, in another aspect -65F or less, in another aspect -70F or less, in another aspect -75F or less, in another aspect -80F or less, in another aspect -85F or less, in another aspect -90F or less, and in another aspect -95F or less.
- the cooling medium includes from about 10 to about 80 weight percent, in another aspect, 15 to 75 weight percent, in another aspect, 20 to 70 weight percent, , in another aspect, 25 to 65 weight percent, and in another aspect, 30 to 60 weight percent, cooling agent, based on the weight of the cooling medium.
- the cooling medium may include the above indicated ranges of cooling agent. Further, the cooling medium may include from about 1 to about 94 weight percent chloride salt, in another aspect, 1 to about 40 weight percent chloride sale, in another aspect, 5 to 30 weight percent, and in another aspect 20 to 30 weight percent chloride salt, based on the weight of the cooling medium.
- Preferred embodiments of the invention employ an aqueous solution of molasses Solids.
- Molasses is the mother liquor left over after crystallization of sugar from materials such as sugar beets and sugar cane.
- Many grades of molasses are available commercially; one suitable grade is desugared sugar beet molasses, which is molasses from which a second sugar fraction has been taken.
- this product contains 60- 75% solids, the solids including carbohydrate, protein, ash, and other components. Further details concerning molasses solids can be found in U.S. Patent 6,080,330 (Bloomer). Most preferably, the molasses solids are used in conjunction with a chloride salt.
- the cooling medium includes from about 1 to about 25 weight percent, in another aspect 10 to 20 weight percent, an in another aspect 12 to 16 weight percent molasses.
- the cooling medium may further includes from about 16 to 40 weight percent, in another aspect 20 to 30 weight percent, and in another aspect 22 to 26 weight percent chloride salt.
- a highly preferred solution includes 14% desugared sugar beet molasses solids and 25.6% calcium chloride.
- the cooling agent also may be a glycoside, in particular a hydrocarbyl aldoside.
- Suitable hydrocarbyl aldosides may be glucosides, maltosides, rnaltotriosides, and mixtures thereof.
- the hydrocarbyl aldoside may be an alkyl aldoside such as alpha-methyl glucoside, beta-methyl glucoside, methyl furanosides, methyl maltosides, methyl rnaltotriosides, and mixtures thereof.
- the hydrocarbyl aldoside is methyl glucoside.
- U.S. Patents 6,582,622; 6,440,325; 6,436,310; and 6,299,793 purport to describe de-icing and anti-icing compositions containing carbohydrates of less than about 1,500 molecular weight.
- the carbohydrates include glucose/fructose, disaccharides, trisaccharides, tetrasaccharides, pentasaccharides, hexasaccharides, and mixtures thereof.
- the carbohydrate molecular weight is from about 180 to 1,500, preferably about 180 to 1,000.
- the carbohydrates can be obtained from a wide range of agricultural based products such as those derived from corn, wheat, barley, oats, sugar cane, sugar beets etc.
- U.S. Patent 6,468,442 describes a de-icing or anti-icing composition using a sugar- water mixture having approximately 15 to 80 percent by weight of a sugar solid, wherein the sugar solid contains approximately 2-60 percent by weight of a monosaccharide.
- a variety of sugars are disclosed, including corn sugar, cane sugar, beet sugar, sorghum sugar, maple sugar, wheat sugar, tapioca sugar, potato sugar, cassava sugar, and manioca sugar.
- al.S. 6,544,434 and 6,315,919 purport to describe de-icing compositions containing hydrocarbyl aldosides including alkyl aldosides, furanosides, maltosides, rnaltotriosides, glucopyranosides and mixtures thereof.
- Alkyl aldosides disclosed are alpha-methyl glucoside, beta-methyl glucoside, methyl furanosides, methyl maltosides, methyl rnaltotriosides, and mixtures thereof.
- de-icing compositions containing a hydroxyl- containing organic compound selected from the group consisting of hydrocarbyl aldosides including glucosides, furanosides, maltosides, rnaltotriosides, and glucopyranosides, sorbitol and other hydrogenation products of sugars, monosaccharides, maltodextrins and sucrose; maltitol; glycols; monosaccharides; glycerol; and mixtures thereof.
- hydrocarbyl aldosides including glucosides, furanosides, maltosides, rnaltotriosides, and glucopyranosides
- sorbitol and other hydrogenation products of sugars monosaccharides, maltodextrins and sucrose
- maltitol glycols
- monosaccharides glycerol
- Suitable hydrocarbyl aldoside include glucopyranoside sucrose arid alkyl aldosides such as alkyl glucosides, alkyl furanosides, alkyl maltosides, alkyl rnaltotriosides, alkylglucopyranosides, and mixtures thereof.
- Other hydrogenation products of sugars, monosaccharides, maltodextrins and sucrose include maltitol, xylitol and mannitol.
- U.S. Patent 6,398,979 describes a liquid deicer composition containing molasses solids.
- molasses solids refers to the components of molasses that are not water such as various carbohydrates (e.g.
- Suitable molasses includes cane molasses, citrus molasses, wood molasses, grain molasses, and combinations thereof.
- U.S. 6,416,648 is directed to a composition useful for preventing the formation of ice or snow on surfaces or for deicing surfaces having ice or snow.
- the composition is formed from a waste product of the process of removing sugar from molasses, also known as desugared molasses.
- the sugar may be removed from sugar beet or cane molasses or other types of molasses such as sorghum or citrus.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Combustion & Propulsion (AREA)
- Thermal Sciences (AREA)
- Physics & Mathematics (AREA)
- Organic Chemistry (AREA)
- Materials Applied To Surfaces To Minimize Adherence Of Mist Or Water (AREA)
- Emulsifying, Dispersing, Foam-Producing Or Wetting Agents (AREA)
- Polysaccharides And Polysaccharide Derivatives (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Seasonings (AREA)
- Confectionery (AREA)
- Saccharide Compounds (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AP2006003560A AP2006003560A0 (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and its uses |
| EP04782984A EP1670873A2 (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and its uses |
| JP2006525443A JP2007504326A (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and use thereof |
| BRPI0414015-0A BRPI0414015A (en) | 2003-09-02 | 2004-09-02 | cooling medium, method for cooling, and method for transferring heat |
| AU2004269423A AU2004269423A1 (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and its uses |
| CA002537528A CA2537528A1 (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and its uses |
| NO20061467A NO20061467L (en) | 2003-09-02 | 2006-03-31 | Organic dressing medium and its use |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US49980303P | 2003-09-02 | 2003-09-02 | |
| US60/499,803 | 2003-09-02 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005021732A2 true WO2005021732A2 (en) | 2005-03-10 |
| WO2005021732A3 WO2005021732A3 (en) | 2005-08-18 |
Family
ID=34272872
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2004/028597 WO2005021732A2 (en) | 2003-09-02 | 2004-09-02 | Organic cooling medium and its uses |
Country Status (14)
| Country | Link |
|---|---|
| US (2) | US20050184272A1 (en) |
| EP (1) | EP1670873A2 (en) |
| JP (1) | JP2007504326A (en) |
| CN (1) | CN1845978A (en) |
| AP (1) | AP2006003560A0 (en) |
| AU (1) | AU2004269423A1 (en) |
| BR (1) | BRPI0414015A (en) |
| CA (1) | CA2537528A1 (en) |
| EC (1) | ECSP066470A (en) |
| MA (1) | MA28324A1 (en) |
| NO (1) | NO20061467L (en) |
| OA (1) | OA13249A (en) |
| RU (1) | RU2006110619A (en) |
| WO (1) | WO2005021732A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7272940B2 (en) | 2004-06-14 | 2007-09-25 | Vinberg Donald J | Submersion tank for on-board fish freezing |
Families Citing this family (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005006476A1 (en) | 2003-07-11 | 2005-01-20 | Shishiai-Kabushikigaisha | Cooling fluid composition for fuel battery |
| JPWO2005091413A1 (en) * | 2004-03-24 | 2007-08-30 | シーシーアイ株式会社 | Coolant composition for fuel cell |
| CN104927789B (en) * | 2015-06-26 | 2016-04-20 | 中南大学 | A kind of secondary refrigerant for the energy storage of characteristics of dynamic ice slurry ice-reserving |
| GB2564063B (en) | 2016-03-24 | 2022-04-06 | Tetra Tech | High density, low TCT divalent brines and uses thereof |
| US10913884B2 (en) | 2016-03-24 | 2021-02-09 | Tetra Technologies, Inc | Temperature stability of polyols and sugar alcohols in brines |
| GB2564359B (en) | 2016-03-24 | 2022-01-05 | Tetra Tech | High density, low TCT monovalent brines and uses thereof |
| US11021645B2 (en) | 2017-10-24 | 2021-06-01 | Tetra Technologies, Inc | Stabilization and reduction of TCT of divalent iodide-containing brines |
| US11453817B2 (en) | 2017-10-24 | 2022-09-27 | Tetra Technologies, Inc. | Stabilization of iodide-containing brines and brine mixtures |
| US10851278B2 (en) | 2017-10-24 | 2020-12-01 | Tetra Technologies, Inc. | Stabilization and reduction of TCT of brines containing monovalent iodides |
| US11166577B2 (en) * | 2017-11-10 | 2021-11-09 | Forgotten Kingdom LLC | Beverage cooling apparatus |
| PT3834893T (en) * | 2018-08-06 | 2024-03-04 | Success World S L | Fireproof composition |
| CA3210867A1 (en) * | 2021-02-08 | 2022-08-11 | Uniquem Inc. | Environmentally friendly additives |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20010011716A1 (en) * | 1997-02-27 | 2001-08-09 | Idemitsu Kosan Co., Ltd. | Refrigerating oil composition |
| US6398979B2 (en) * | 2000-02-28 | 2002-06-04 | Cargill, Incorporated | Deicer and pre-wetting agent |
| US6440325B1 (en) * | 1998-01-07 | 2002-08-27 | Sears Petroleum & Transport Corporation | De-icing solution |
| US20030198847A1 (en) * | 2002-02-19 | 2003-10-23 | Jeffcoate Carol S. | Heat transfer compositions with high electrical resistance for fuel cell assemblies |
Family Cites Families (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS61219364A (en) * | 1985-03-27 | 1986-09-29 | Shigeko Ueno | Heat storage material or the like having freshness preserving temperature range |
| JPS62285978A (en) * | 1986-06-05 | 1987-12-11 | Nippon Light Metal Co Ltd | Cooling energy storing agent composition |
| JPH0657242A (en) * | 1992-08-05 | 1994-03-01 | Mitsui Eng & Shipbuild Co Ltd | Heat-transport medium for ice heat storage |
| JPH0654645A (en) * | 1992-08-06 | 1994-03-01 | Numata Masao | Immersion freezing |
| JPH0719682A (en) * | 1993-06-29 | 1995-01-20 | Mitsui Eng & Shipbuild Co Ltd | Manufacture of ice slurry by ice cold accumulator |
| US5876621A (en) * | 1997-09-30 | 1999-03-02 | Sapienza; Richard | Environmentally benign anti-icing or deicing fluids |
| US6596188B1 (en) * | 1998-01-07 | 2003-07-22 | Sears Petroleum & Transport Corp. | Deicing solution |
| JPH11335660A (en) * | 1998-05-29 | 1999-12-07 | Hitachi Zosen Corp | Cold-reserving material |
| US6080330A (en) * | 1999-06-14 | 2000-06-27 | Bloomer; Todd A. | Anti-freezing and deicing composition and method |
| CN1370212A (en) * | 1999-07-26 | 2002-09-18 | 明尼苏达粮食加工公司 | De-icing compsn. and method |
| JP4842420B2 (en) * | 1999-09-28 | 2011-12-21 | トヨタ自動車株式会社 | Cooling liquid, cooling liquid sealing method and cooling system |
| JP2001271060A (en) * | 2000-03-28 | 2001-10-02 | Taki Chem Co Ltd | Snow-melting agent |
| CA2534715A1 (en) * | 2003-08-07 | 2005-02-24 | Glendon C. Daly | Heat transfer fluid |
-
2004
- 2004-09-02 BR BRPI0414015-0A patent/BRPI0414015A/en not_active IP Right Cessation
- 2004-09-02 AU AU2004269423A patent/AU2004269423A1/en not_active Abandoned
- 2004-09-02 CA CA002537528A patent/CA2537528A1/en not_active Abandoned
- 2004-09-02 CN CNA200480025124XA patent/CN1845978A/en active Pending
- 2004-09-02 WO PCT/US2004/028597 patent/WO2005021732A2/en active Application Filing
- 2004-09-02 OA OA1200600075A patent/OA13249A/en unknown
- 2004-09-02 JP JP2006525443A patent/JP2007504326A/en active Pending
- 2004-09-02 US US10/932,927 patent/US20050184272A1/en not_active Abandoned
- 2004-09-02 EP EP04782984A patent/EP1670873A2/en not_active Withdrawn
- 2004-09-02 AP AP2006003560A patent/AP2006003560A0/en unknown
- 2004-09-02 RU RU2006110619/04A patent/RU2006110619A/en not_active Application Discontinuation
-
2006
- 2006-03-31 EC EC2006006470A patent/ECSP066470A/en unknown
- 2006-03-31 MA MA28902A patent/MA28324A1/en unknown
- 2006-03-31 NO NO20061467A patent/NO20061467L/en not_active Application Discontinuation
-
2007
- 2007-08-30 US US11/847,837 patent/US20080093579A1/en not_active Abandoned
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20010011716A1 (en) * | 1997-02-27 | 2001-08-09 | Idemitsu Kosan Co., Ltd. | Refrigerating oil composition |
| US6440325B1 (en) * | 1998-01-07 | 2002-08-27 | Sears Petroleum & Transport Corporation | De-icing solution |
| US6398979B2 (en) * | 2000-02-28 | 2002-06-04 | Cargill, Incorporated | Deicer and pre-wetting agent |
| US20030198847A1 (en) * | 2002-02-19 | 2003-10-23 | Jeffcoate Carol S. | Heat transfer compositions with high electrical resistance for fuel cell assemblies |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7272940B2 (en) | 2004-06-14 | 2007-09-25 | Vinberg Donald J | Submersion tank for on-board fish freezing |
Also Published As
| Publication number | Publication date |
|---|---|
| US20080093579A1 (en) | 2008-04-24 |
| US20050184272A1 (en) | 2005-08-25 |
| CN1845978A (en) | 2006-10-11 |
| AP2006003560A0 (en) | 2006-04-30 |
| EP1670873A2 (en) | 2006-06-21 |
| CA2537528A1 (en) | 2005-03-10 |
| OA13249A (en) | 2007-01-31 |
| JP2007504326A (en) | 2007-03-01 |
| WO2005021732A3 (en) | 2005-08-18 |
| RU2006110619A (en) | 2007-10-10 |
| MA28324A1 (en) | 2006-12-01 |
| BRPI0414015A (en) | 2006-10-24 |
| AU2004269423A1 (en) | 2005-03-10 |
| ECSP066470A (en) | 2006-09-18 |
| NO20061467L (en) | 2006-04-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20080093579A1 (en) | Organic Cooling Medium And Its Uses | |
| US7658861B2 (en) | Corrosion-inhibiting deicer composition | |
| US6506318B1 (en) | Environmentally benign anti-icing or deicing fluids | |
| KR960006073B1 (en) | Method of production of ice by using fluorinated pentane | |
| EP0611388B1 (en) | Vapour absorbent compositions | |
| US6544434B2 (en) | Environmentally benign anti-icing or deicing fluids | |
| US20090314983A1 (en) | Environmentally benign anti-icing or deicing fluids | |
| NO340830B1 (en) | Runway de-icing fluid | |
| EP1449903B1 (en) | Synergistic combinations of carboxylates for use as freezing point depressants and corrosion inhibitors in heat transfer fluids | |
| CA2431235A1 (en) | Deicing solution | |
| EP2789669A2 (en) | Combinations of carboxylates for use as freezing point depressants and corrosion protection in heat-transfer fluids | |
| EP1664232A2 (en) | Heat transfer fluid | |
| KR20070020368A (en) | Organic cooling medium and its uses | |
| RU2277568C2 (en) | Agent for elimination of the highways and roads icing-up and the method of the ice and snow thawing | |
| CA2246793A1 (en) | Snow inducing agents for artificial snow manufacturing | |
| US6478984B1 (en) | Liquid for evaporative cooling apparatus | |
| JP4197161B2 (en) | Deicing agent and method for melting snow and ice | |
| URAJI et al. | Freezing point depression of polyol-aqueous solutions in the high concentration range | |
| JP2740554B2 (en) | Brine composition | |
| Ferguson | The Melting Interval of Certain Under-cooled Liquids with a Note on the Use of Liquid Air as a Refrigerant | |
| CA2210902A1 (en) | Use as cold transfer media of aqueous solutions of lower alcohols | |
| EP3088820A1 (en) | Household refrigerating and/or deep-freezing apparatus | |
| Briley | Moisture loss during freezing |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 200480025124.X Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 12006500430 Country of ref document: PH Ref document number: 2537528 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006525443 Country of ref document: JP Ref document number: 1020067004321 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 546039 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004782984 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AP/P/2006/003560 Country of ref document: AP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004269423 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1125/CHENP/2006 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006110619 Country of ref document: RU Ref document number: 1200600518 Country of ref document: VN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200602678 Country of ref document: ZA |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004269423 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004782984 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0414015 Country of ref document: BR |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020067004321 Country of ref document: KR |