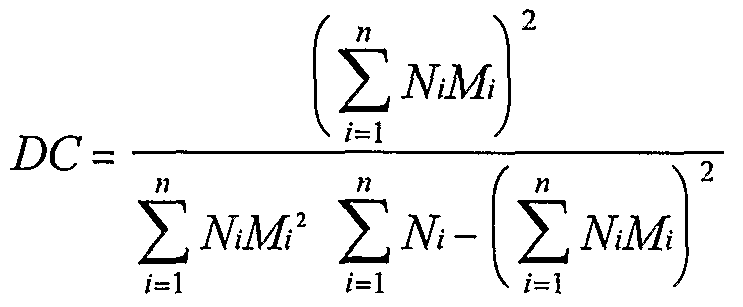WO2005004792A2 - Mixtures of calcitonin drug-oligomer conjugates and methods of use in pain treatment - Google Patents
Mixtures of calcitonin drug-oligomer conjugates and methods of use in pain treatment Download PDFInfo
- Publication number
- WO2005004792A2 WO2005004792A2 PCT/US2004/016784 US2004016784W WO2005004792A2 WO 2005004792 A2 WO2005004792 A2 WO 2005004792A2 US 2004016784 W US2004016784 W US 2004016784W WO 2005004792 A2 WO2005004792 A2 WO 2005004792A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- oligomer
- mixture
- calcitonin
- moiety
- conjugates
- Prior art date
Links
- YWIMSRRHADZIMX-UHFFFAOYSA-N CC(C)(CCOC(C)(C)CCCCCC(ON(C)C1OC1CCC=O)=O)OC Chemical compound CC(C)(CCOC(C)(C)CCCCCC(ON(C)C1OC1CCC=O)=O)OC YWIMSRRHADZIMX-UHFFFAOYSA-N 0.000 description 1
- 0 CC*C(CCCCCC(C)(C*)OCCC(C)OCc1ccccc1)=O Chemical compound CC*C(CCCCCC(C)(C*)OCCC(C)OCc1ccccc1)=O 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/22—Hormones
- A61K38/23—Calcitonins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/02—Drugs for disorders of the nervous system for peripheral neuropathies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
- A61P29/02—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID] without antiinflammatory effect
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/12—Drugs for disorders of the metabolism for electrolyte homeostasis
- A61P3/14—Drugs for disorders of the metabolism for electrolyte homeostasis for calcium homeostasis
Definitions
- the present invention relates to drug-oligomer conjugates, and, more particularly, to calcitonin drug-oligomer conjugates and methods of using these conjugates to treat various disorders.
- Calcitonin is a naturally occurring hormone with a short half-life that is believed to act directly on osteoclasts (via receptors on the cell surface for calcitonin). This action may directly inhibit osteoclastic bone resorption, which may lead to hypocalcemic and/or hypophosphatemic serum effects. Calcitonin may be useful in treating various bone disorders including, but not limited to, osteoporosis and Paget's disease.
- Osteoporosis is a bone disease in which bone tissue is normally mineralized, but the amount of bone is decreased and the structural integrity of trabecular bone is impaired. Cortical bone becomes more porous and thinner. This makes the bone weaker and more likely to fracture.
- about 21% of postmenopausal women have osteoporosis (low bone density), and about 16% have had a fracture.
- women older than 80 about 40% have experienced a fracture of the hip, vertebra, arm, or pelvis. The population of older men and women has been increasing, and therefore the number of people with osteoporosis is increasing.
- Calcitonin given as a subcutaneous injection has shown significant improvements in bone density; however, a high incidence of side effects, including pain at the injection site, flushing and nausea, have been reported which may limit the use of the drug.
- Paget's disease of bone is a metabolic bone disorder of unknown origin which normally affects older people. The disease causes an increased and irregular formation of bone as the bone cells, which are responsible for dissolving the body's old bone and replacing it with new, become out of control. Over a period of time the deformed new bone becomes larger, weaker and has more blood vessels than normal bone. Unlike normal bone, the structure is irregular and consequently weaker, which makes it prone to fracture even after a minor injury. In its mildest form the disease has no symptoms. In more severe cases the pain can be intense.
- Calcitonin may be effective in treating disorders of increased skeletal remodeling, such as Paget's disease.
- Paget's disease chronic use of calcitonin may produce long-term reduction in symptoms; however, side effects of calcitonin administration may include nausea, hand swelling, urticaria, and intestinal cramping.
- PEG is typically produced by base-catalyzed ring-opening polymerization of ethylene oxide. The reaction is initiated by adding ethylene oxide to ethylene glycol, with potassium hydroxide as catalyst. This process results in a polydispersed mixture of polyethylene glycol polymers having a number average molecular weight within a given range of molecular weights.
- PEG products offered by Sigma- Aldrich of Milwaukee, Wisconsin are provided in polydispersed mixtures such as PEG 400 (M n 380-420); PEG 1,000 (M span 950-1,050); PEG 1,500 (M n 1,400-1,600); and PEG 2,000 (M n 1,900-2,200).
- a mixture of calcitonin-oligomer conjugates comprising polyethylene glycol according to embodiments of the present invention may lower serum calcium levels by 10, 15 or even 20 percent or more.
- a mixture of calcitonin-oligomer conjugates comprising polyethylene glycol according to embodiments of the present invention may be more effective at surviving an in vitro model of intestinal digestion than non-conjugated calcitonin.
- mixtures of calcitonin-oligomer conjugates comprising polyethylene glycol may exhibit a higher bioavailability than non-conjugated calcitonin.
- a substantially monodispersed mixture of conjugates each comprising a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety is provided.
- the polyethylene glycol moiety preferably has at least 2, 3, or 4 polyethylene glycol subunits and, most preferably, has at least 7 polyethylene glycol subunits.
- the oligomer preferably further comprises a lipophilic moiety.
- the calcitonin drug is preferably salmon calcitonin.
- Oligomers are preferably coupled at Lys 11 and Lys 18 of the salmon calcitonin.
- the conjugate is preferably amphiphilically balanced such that the conjugate is aqueously soluble and able to penetrate biological membranes.
- a substantially monodispersed mixture of conjugates is provided where each conjugate includes salmon calcitonin covalently coupled at Lys 1 of the salmon calcitonin to a carboxylic acid moiety of a first oligomer that comprises octanoic acid covalently coupled at the end distal to the carboxylic acid moiety to a methyl terminated polyethylene glycol moiety having at least 7 polyethylene glycol subunits, and covalently coupled at Lys 18 of the salmon calcitonin to a carboxylic acid moiety of a second oligomer that comprises octanoic acid covalently coupled at the end distal to the carboxylic acid moiety to a methyl terminated polyethylene glycol moiety having at least 7 polyethylene
- a substantially monodispersed mixture of conjugates where each conjugate comprises a calcitonin drug coupled to an oligomer comprising a polyethylene glycol moiety, and the mixture is capable of lowering serum calcium levels in a subject by at least 5 percent.
- a substantially monodispersed mixture of conjugates where each conjugate comprises a calcitonin drug coupled to an oligomer comprising a polyethylene glycol moiety, and the mixture has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of the calcitonin drug which is not coupled to the oligomer.
- a substantially monodispersed mixture of conjugates where each conjugate comprises a calcitonin drug coupled to an oligomer comprising a polyethylene glycol moiety, and the mixture has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- a mixture of conjugates is provided where each conjugate includes a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety, and the mixture has a molecular weight distribution with a standard deviation of less than about 22 Daltons.
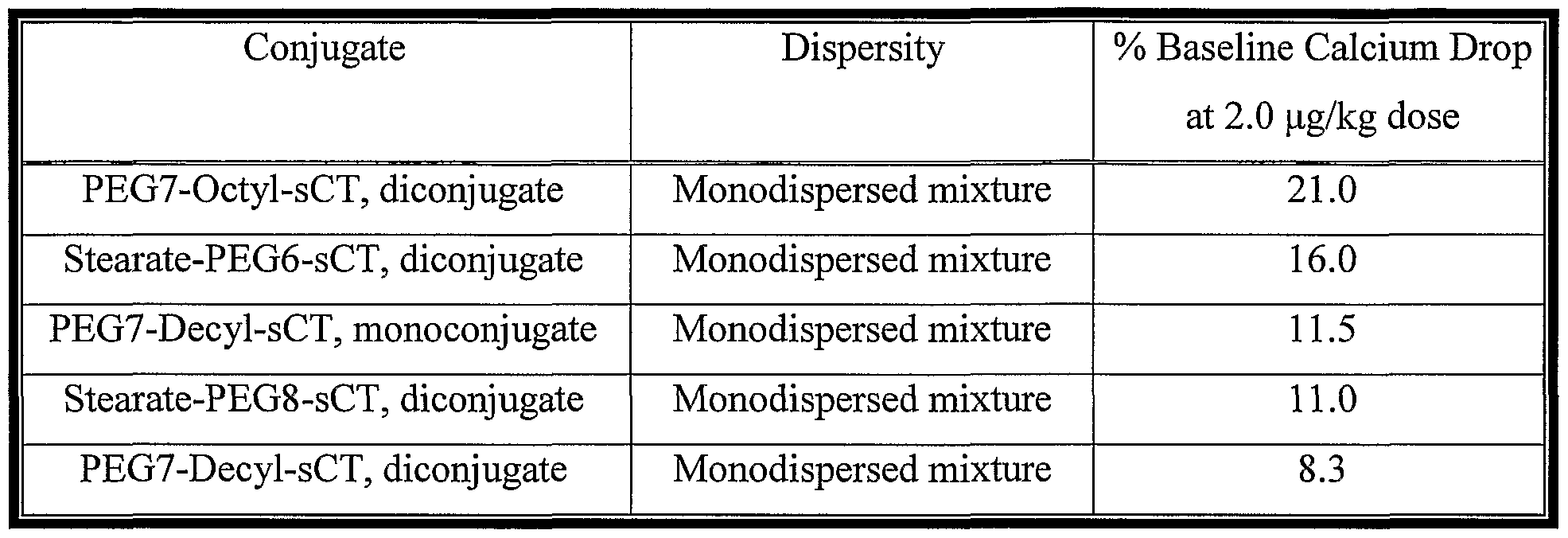
- each conjugate includes a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety, and the mixture has a dispersity coefficient (DC) greater than 10,000 where
- a mixture of conjugates is provided in which each conjugate includes a calcitonin drug coupled to an oligomer and has the same number of polyethylene glycol subunits.
- a mixture of conjugates is provided in which each conjugate has the same molecular weight and has the formula: Calcitonin Drug- B-L j -G k - • R-G' — R*— G" n — T (A)
- B is a bonding moiety
- L is a linking group
- G, G and G" are individually selected spacer groups
- R is a lipophilic group and R' is a polyalkylene glycol group, or R' is the lipophilic group and R is the polyalkylene oxide group
- T is a terminating group
- j, k, m and n are individually 0 or 1
- p is an integer from 1 to the number of nucleophilic residues on the calcitonin drug.
- Calcitonin-oligomer conjugate mixtures may lower serum calcium levels by 20 percent or more. Moreover, such conjugates may provide decreased degradation by intestinal enzymes and/or provide increased bioavailability when compared to non-conjugated calcitonin.
- the present invention further provides methods of treating pain (e.g., peripheral pain; central pain) in a subject in need thereof comprising administering to the subject an effective amount of a composition of this invention, which can include one or more of the following. 1.
- a substantially monodispersed mixture of conjugates wherein the conjugate comprises a first oligomer and a second oligomer, wherein each oligomer is coupled to salmon calcitonin and wherein the first oligomer is covalently coupled to an amine function of Lys 11 of the salmon calcitonin and the second oligomer is covalently coupled to an amine function of Lys 18 of the salmon calcitonin; 2.
- a substantially monodispersed mixture of conjugates each conjugate comprising a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety, wherein the oligomer comprises a first polyethylene glycol moiety covalently coupled to the calcitonin drug by a non-hydrolyzable bond and a second polyethylene glycol moiety covalently coupled to the first polyethylene glycol moiety by a hydrolyzable bond;
- a method of treating peripheral pain in a subject in need thereof comprising administering to the subject an effective amount of a mixture of conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons, wherein each conjugate in the mixture comprises salmon calcitonin coupled at Lys 11 to a first oligomer and coupled at Lys 18 to a second oligomer, and wherein the first oligomer and the
- second oligomer each have the formula: O O I I I I — C— (OCH 2 CH 2 ) 6 -OC(CH 2 ) 16 CH 3 .
- n is the number of different molecules in the sample; ⁇ ; is the number of i* molecules in the sample; and th Mj is the mass of the i molecule, and wherein each conjugate in the mixture comprises salmon calcitonin coupled at
- Lys 11 to a first oligomer and coupled at Lys 18 to a second oligomer, and wherein the first oligomer and the second oligomer each have the formula: O O I I II — C-(OCH 2 CH 2 ) 6 -OC(CH 2 ) 16 CH 3 .
- n is the number of different molecules in the sample; ⁇ ; is the number of i th molecules in the sample; and Mj is the mass of the i molecule, and wherein each conjugate in the mixture comprises salmon calcitonin coupled at Lys or Lys to an oligomer having the formula: O II — C— (CH 2 ) 9 — (OC 2 H 4 ) 7 - OCH 3 . . 5
- the calcitonin drug is a salmon calcitonin coupled to the oligomer at Lys n and Lys 18 ;
- B is a bonding moiety;
- L is a linker moiety;
- G, G' and G" are individually selected spacer moieties;
- R is a lipophilic moiety and R' is a polyalkylene glycol moiety, or R' is the lipophilic moiety and R is the polyalkylene glycol moiety;
- T is methoxy; j, k, m and n are individually 0 or 1 ; and
- p is an integer from 1 to the number of nucleophilic residues on the calcitonin drug.
- Figure 1 illustrates a generic scheme for synthesizing a mixture of activated polymers comprising a polyethylene glycol moiety and a fatty acid moiety according to embodiments of the present invention.
- Figure 2 illustrates a scheme for synthesizing a mixture of mPEG according to embodiments of the present invention.
- Figure 3 illustrates a scheme for synthesizing a mixture of activated mPEG7-hexyl oligomers according to embodiments of the present invention.
- Figure 4 illustrates a scheme for synthesizing a mixture of activated mPEG7-octyl oligomers according to embodiments of the present invention.
- Figure 5 illustrates a scheme for synthesizing a mixture of activated mPEG-decyl oligomers according to embodiments of the present invention.
- Figure 6 illustrates a scheme for synthesizing a mixture of activated stearate-PEG6 oligomers according to embodiments of the present invention.
- Figure 7 illustrates a scheme for synthesizing a mixture of activated stearate-PEG8 oligomers according to embodiments of the present invention.
- Figure 8 illustrates a scheme for synthesizing a mixture of activated PEG3 oligomers according to embodiments of the present invention.
- Figure 9 illustrates a scheme for synthesizing a mixture of activated palmitate-PEG3 oligomers according to embodiments of the present invention.
- Figure 10 illustrates a scheme for synthesizing a mixture of activated PEG6 oligomers according to embodiments of the present invention.
- Figure 11 illustrates a scheme for synthesizing various propylene glycol monomers according to embodiments of the present invention.
- Figure 12 illustrates a scheme for synthesizing various propylene glycol polymers according to embodiments of the present invention.
- Figure 13 illustrates a scheme for synthesizing various propylene glycol polymers according to embodiments of the present invention.
- Figure 14 illustrates a comparison of the average AUCs for various mixtures of calcitonin-oligomer conjugates according to embodiments of the present invention with non- conjugated calcitonin, which is provided for comparison purposes only and does not form part of the invention.
- Figure 15 illustrates a dose-response curve for a mixture of mPEG7-octyl-calcitonin diconjugates according to embodiments of the present invention compared with a dose- response curve for calcitonin, which is provided for comparison purposes and is not a part of the present invention.
- Figure 16 illustrates a dose-response curve after oral administration of a mixture of mPEG7-octyl-calcitonin diconjugates according to embodiments of the present invention.
- Figure 17 illustrates a dose-response curve after subcutaneous administration of a mixture of mPEG7-octyl-calcitonin diconjugates according to embodiments of the present invention.
- Figure 18 illustrates a dose-response curve after subcutaneous administration of salmon calcitonin, which is provided for comparison purposes and is not part of the present invention.
- Figure 19 shows latency results obtained after oral administration of CT-025 and drug vehicle in the tail flick and hot-plate assays.
- Figure 20 shows the number of stretches as a measure of the analgesic response during the acetic acid writhing test.
- non-polydispersed is used to describe a mixture of compounds having a dispersity that is in contrast to the polydispersed mixtures described in U.S. Patent No. 5,359,030 to Ekwuribe.
- substantially monodispersed is used to describe a mixture of compounds wherein at least about 95 percent of the compounds in the mixture have the same molecular weight.
- the term "monodispersed” is used to describe a mixture of compounds wherein about 100 percent of the compounds in the mixture have the same molecular weight.
- the term “substantially purely monodispersed” is used to describe a mixture of compounds wherein at least about 95 percent of the compounds in the mixture have the same molecular weight and have the same molecular structure.
- a substantially purely monodispersed mixture is a substantially monodispersed mixture, but a substantially monodispersed mixture is not necessarily a substantially purely monodispersed mixture.
- the term "purely monodispersed” is used to describe a mixture of compounds wherein about 100 percent of the compounds in the mixture have the same molecular weight and have the same molecular structure. Thus, a purely monodispersed mixture is a monodispersed mixture, but a monodispersed mixture is not necessarily a purely monodispersed mixture.
- the term "weight average molecular weight” is defined as the sum of the products of the weight fraction for a given molecule in the mixture times the mass of the molecule for each molecule in the mixture. The "weight average molecular weight” is represented by the symbol M w .
- the term "number average molecular weight” is defined as the total weight of a mixture divided by the number of molecules in the mixture and is represented by the symbol M n .
- the term “dispersity coefficient” (DC) is defined by the formula:
- n is the number of different molecules in the sample; Ni is the number of i ⁇ molecules in the sample; and th Mj is the mass of the i ⁇ molecule.
- intra-subject variability means the variability in activity occurring within the same subject when the subject is administered the same dose of a drug or pharmaceutical composition at different times.
- inter-subject variability means the variability in activity between two or more subjects when each subject is administered the same dose of a given drug or pharmaceutical formulation.
- bioefficacy means the ability of a drug or drug conjugate to interact with one or more desired receptors in vivo.
- the term "calcitonin drug” means a drug possessing all or some of the biological activity of calcitonin.
- the term “calcitonin” means chicken calcitonin, eel calcitonin, human calcitonin, porcine calcitonin, rat calcitonin or salmon calcitonin provided by natural, synthetic, or genetically engineered sources.
- the term “calcitonin analog” means calcitonin wherein one or more of the amino acids have been replaced while retaining some or all of the activity of the calcitonin. The analog is described by noting the replacement amino acids with the position of the replacement as a superscript followed by a description of the calcitonin.
- Pro 2 calcitonin, human means that the glycine typically found at the 2 position of a human calcitonin molecule has been replaced with proline.
- Calcitonin analogs may be obtained by various means, as will be understood by those skilled in the art.
- certain amino acids may be substituted for other amino acids in the calcitonin structure without appreciable loss of interactive binding capacity with structures such as, for example, antigen-binding regions of antibodies or binding sites on substrate molecules.
- the interactive capacity and nature of calcitonin defines its biological functional activity, certain amino acid sequence substitutions can be made in the amino acid sequence and nevertheless remain a polypeptide with like properties. In making such substitutions, the hydropathic index of amino acids may be considered.
- hydropathic amino acid index in conferring interactive biologic function on a polypeptide is generally understood in the art. It is accepted that the relative hydropathic character of the amino acid contributes to the secondary structure of the resultant polypeptide, which in turn defines the interaction of the polypeptide with other molecules, for example, enzymes, substrates, receptors, DNA, antibodies, antigens, and the like.
- Each amino acid has been assigned a hydropathic index on the basis of its hydrophobicity and charge characteristics as follows: isoleucine (+4.5); valine (+4.2); leucine (+3.8); phenylalanine (+2.8); cysteine/cystine (+2.5); methionine (+1.9); alanine (+1.8); glycine (-0.4); threonine (-0.7); serine (-0.8); tryptophan (-0.9); tyrosine (-1.3); proline (-1.6); histidine (-3.2); glutamate (-3.5); glutamine (-3.5); aspartate (-3.5); asparagine (-3.5); lysine (-3.9); and arginine (-4.5).
- hydrophilicity values have been assigned to amino acid residues: arginine (+3.0); lysine ( ⁇ 3.0); aspartate (+3.0 ⁇ 1); glutamate (+3.0 ⁇ 1); seine (+0.3); asparagine (+0.2); glutamine (+0.2); glycine (0); threonine (-0.4); proline (-0.5 ⁇ 1); alanine (-0.5); histidine (-0.5); cysteine (-1.0); methionine (-1.3); valine (-1.5); leucine (-1.8); isoleucine (-1.8); tyrosine (-2.3); phenylalanine (-2.5); tryptophan (-3.4).
- amino acid can be substituted for another having a similar hydrophilicity value and still obtain a biologically equivalent, and in particular, an immunologically equivalent polypeptide.
- substitution of amino acids whose hydrophilicity values are within ⁇ 2 of each other is preferred, those which are within ⁇ 1 of each other are particularly preferred, and those within ⁇ 0.5 of each other are even more particularly preferred.
- amino acid substitutions are generally therefore based on the relative similarity of the amino acid side-chain substituents, for example, their hydrophobicity, hydrophilicity, charge, size, and the like.
- substitutions i.e., amino acids that may be interchanged without significantly altering the biological activity of the polypeptide
- calcitonin fragment means a segment of the amino acid sequence found in the calcitonin that retains some or all of the activity of the calcitonin.
- the term "calcitonin fragment analog” means a segment of the amino acid sequence found in the calcitonin molecule wherein one or more of the amino acids in the segment have been replace while retaining some or all of the activity of the calcitonin.
- PEG refers to straight or branched polyethylene glycol polymers, and includes the monomethylether of polyethylene glycol (mPEG).
- PEG subunit and polyethylene glycol subunit refer to a single polyethylene glycol unit, i.e., — (CH 2 CH 2 O)— .
- lipophilic means the ability to dissolve in lipids and/or the ability to penetrate, interact with and/or traverse biological membranes
- lipophilic moiety or “lipophile” means a moiety which is lipophilic and/or which, when attached to another chemical entity, increases the lipophilicity of such chemical entity.
- lipophilic moieties include, but are not limited to, alkyls, fatty acids, esters of fatty acids, cholesteryl, adamantyl and the like.
- lower alkyl refers to substituted or unsubstituted alkyl moieties having from 1 to 5 carbon atoms.
- higher alkyl refers to substituted or unsubstituted alkyl moieties having 6 or more carbon atoms.
- a substantially monodispersed mixture of calcitonin drug-oligomer conjugates is provided.
- Each calcitonin drug-oligomer conjugate in the monodispersed mixture includes a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety.
- a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety.
- at least about 96, 97, 98 or 99 percent of the conjugates in the mixture have the same molecular weight.
- the mixture is a monodispersed mixture. Even more preferably, the mixture is a substantially purely monodispersed mixture. Still more preferably, at least about 96, 97, 98 or 99 percent of the conjugates in the mixture have the same molecular weight and have the same molecular structure. Most preferably, the mixture is a purely monodispersed mixture.
- the calcitonin drug is preferably calcitonin.
- the calcitonin drag is salmon calcitonin.
- the calcitonin drag may be selected from various calcitonin drugs known to those skilled in the art including, for example, calcitonin precursor peptides, calcitonin, calcitonin analogs, calcitonin fragments, and calcitonin fragment analogs.
- Calcitonin precursor peptides include, but are not limited to, katacalcin (PDN-21) (C-procalcitonin), and N-proCT (amino-terminal procalcitonin cleavage peptide), human.
- Calcitonin analogs may be provided by substitution of one or more amino acids in calcitonin as described above.
- Calcitonin fragments include, but are not limited to, calcitonin 1-7, human; and calcitonin 8-32, salmon. Calcitonin fragment analogs may be provided by substitution of one or more of the amino acids in a calcitonin fragment as described above.
- the oligomer may be various oligomers comprising a polyethylene glycol moiety as will be understood by those skilled in the art.
- the polyethylene glycol moiety of the oligomer has at least 2, 3 or 4 polyethylene glycol subunits. More preferably, the polyethylene glycol moiety has at least 5 or 6 polyethylene glycol subunits and, most preferably, the polyethylene glycol moiety has at least 7 polyethylene glycol subunits.
- the oligomer may comprise one or more other moieties as will be understood by those skilled in the art including, but not limited to, additional hydrophilic moieties, lipophilic moieties, spacer moieties, linker moieties, and terminating moieties.
- the various moieties in the oligomer are covalently coupled to one another by either hydrolyzable or non- hydrolyzable bonds.
- the oligomer may further comprise one or more additional hydrophilic moieties (i.e., moieties in addition to the polyethylene glycol moiety) including, but not limited to, sugars, polyalkylene oxides, and polyamine/PEG copolymers.
- the additional hydrophilic moiety may be a polyethylene glycol moiety. Adjacent polyethylene glycol moieties will be considered to be the same moiety if they are coupled by an ether bond.
- the moiety — O-C 2 H 4 — O-C 2 H 4 — O- H 4 — O-C 2 H 4 — O-C 2 H 4 — O-C 2 H 4 — is a single polyethylene glycol moiety having six polyethylene glycol subunits. If this moiety were the only hydrophilic moiety in the oligomer, the oligomer would not contain an additional hydrophilic moiety.
- Adjacent polyethylene glycol moieties will be considered to be different moieties if they are coupled by a bond other than an ether bond.
- the moiety O II —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —C-O—C 2 H 4 —O—C 2 H 4 is a polyethylene glycol moiety having four polyethylene glycol subunits and an additional hydrophilic moiety having two polyethylene glycol subunits.
- oligomers according to embodiments of the present invention comprise a polyethylene glycol moiety and no additional hydrophilic moieties.
- the oligomer may further comprise one or more lipophilic moieties as will be understood by those skilled in the art.
- the lipophilic moiety is preferably a saturated or unsaturated, linear or branched alkyl moiety or a saturated or unsaturated, linear or branched fatty acid moiety.
- the lipophilic moiety is an alkyl moiety, it is preferably a linear, saturated or unsaturated alkyl moiety having 1 to 28 carbon atoms. More preferably, the alkyl moiety has 2 to 12 carbon atoms.
- the lipophilic moiety is a fatty acid moiety, it is preferably a natural fatty acid moiety that is linear, saturated or unsaturated, having 2 to 18 carbon atoms. More preferably, the fatty acid moiety has 3 to 14 carbon atoms.
- the fatty acid moiety has at least 4, 5 or 6 carbon atoms.
- the oligomer may further comprise one or more spacer moieties as will be understood by those skilled in the art. Spacer moieties may, for example, be used to separate a hydrophilic moiety from a lipophilic moiety, to separate a lipophilic moiety or hydrophilic moiety from the calcitonin drug, to separate a first hydrophilic or lipophilic moiety from a second hydrophilic or lipophilic moiety, or to separate a hydrophilic moiety or lipophilic moiety from a linker moiety.
- Spacer moieties are preferably selected from the group consisting of sugar, cholesterol and glycerine moieties.
- the oligomer may further comprise one or more linker moieties that are used to couple the oligomer with the calcitonin drag as will be understood by those skilled in the art.
- Linker moieties are preferably selected from the group consisting of alkyl and fatty acid moieties.
- the oligomer may further comprise one or more terminating moieties at the one or more ends of the oligomer which are not coupled to the calcitonin drug.
- the terminating moiety is preferably an alkyl or alkoxy moiety, and is more preferably a lower alkyl or lower alkoxy moiety. Most preferably, the terminating moiety is methyl or methoxy.
- the terminating moiety is preferably an alkyl or alkoxy moiety, it is to be understood that the terminating moiety may be various moieties as will be understood by those skilled in the art including, but not limited to, sugars, cholesterol, alcohols, and fatty acids.
- the oligomer is preferably covalently coupled to the calcitonin drag.
- the calcitonin drug is coupled to the oligomer utilizing a hydrolyzable bond (e.g., an ester or carbonate bond).
- a hydrolyzable coupling may provide a calcitonin drag- oligomer conjugate that acts as a prodrug.
- a hydrolyzable coupling may provide for a time-release or controlled-release effect, administering the calcitonin drug over a given time period as one or more oligomers are cleaved from their respective calcitonin drug-oligomer conjugates to provide the active drug.
- the calcitonin drug is coupled to the oligomer utilizing a non-hydrolyzable bond (e.g., a carbamate, amide, or ether bond).
- the oligomer When the oligomer is covalently coupled to the calcitonin drug, the oligomer further comprises one or more bonding moieties that are used to covalently couple the oligomer with the calcitonin drag as will be understood by those skilled in the art. Bonding moieties are preferably selected from the group consisting of covalent bond(s), ester moieties, carbonate moieties, carbamate moieties, amide moieties and secondary amine moieties.
- More than one moiety on the oligomer may be covalently coupled to the calcitonin drag. While the oligomer is preferably covalently coupled to the calcitonin drag, it is to be understood that the oligomer may be non-covalently coupled to the calcitonin drag to form a non-covalently conjugated calcitonin drug-oligomer complex. As will be understood by those skilled in the art, non-covalent couplings include, but are not limited to, hydrogen bonding, ionic bonding, Van der Waals bonding, and micellular or liposomal encapsulation.
- oligomers may be suitably constructed, modified and/or appropriately functionalized to impart the ability for non-covalent conjugation in a selected manner (e.g., to impart hydrogen bonding capability), as will be understood by those skilled in the art.
- oligomers may be derivatized with various compounds including, but not limited to, amino acids, oligopeptides, peptides, bile acids, bile acid derivatives, fatty acids, fatty acid derivatives, salicylic acids, salicylic acid derivatives, aminosalicylic acids, and aminosalicylic acid derivatives.
- the resulting oligomers can non-covalently couple (complex) with drug molecules, pharmaceutical products, and/or pharmaceutical excipients.
- the resulting complexes preferably have balanced lipophilic and hydrophilic properties.
- oligomers may be derivatized with amine and/or alkyl amines. Under suitable acidic conditions, the resulting oligomers can form non- covalently conjugated complexes with drag molecules, pharmaceutical products and/or pharmaceutical excipients.
- the products resulting from such complexation preferably have balanced lipophilic and hydrophilic properties. More than one oligomer (i.e., a plurality of oligomers) may be coupled to the calcitonin drug.
- the oligomers in the plurality are preferably the same. However, it is to be understood that the oligomers in the plurality may be different from one another, or, alternatively, some of the oligomers in the plurality may be the same and some may be different.
- a plurality of oligomers are coupled to the calcitonin drug, it may be preferable to couple one or more of the oligomers to the calcitonin drag with hydrolyzable bonds and couple one or more of the oligomers to the calcitonin drag with non-hydrolyzable bonds.
- all of the bonds coupling the plurality of oligomers to the calcitonin drag may be hydrolyzable, but have varying degrees of hydrolyzability such that, for example, one or more of the oligomers is rapidly removed from the calcitonin drag by hydrolysis in the body and one or more of the oligomers is slowly removed from the calcitonin drag by hydrolysis in the body.
- the oligomer may be coupled to the calcitonin drug at various nucleophilic residues of the calcitonin drug including, but not limited to, nucleophilic hydroxyl functions and/or amino functions.
- a nucleophilic hydroxyl function may be found, for example, at serine and/or tyrosine residues, and a nucleophilic amino function may be found, for example, at histidine and/or lysine residues, and/or at the one or more N-termini of the polypeptide.
- the coupling preferably forms a secondary amine.
- the oligomer may be coupled to an amino functionality of the salmon calcitonin, including the amino functionality of Lys 11 , Lys 18 and/or the N-terminus. While one or more oligomers may be coupled to the salmon calcitonin, a higher bioefficacy, such as improved serum calcium lowering ability, is observed for the di-conjugated salmon calcitonin where an oligomer is coupled to the amino functionalities of Lys 11 and the Lys 18 . Substantially monodispersed mixtures of calcitonin drug-oligomer conjugates of the present invention may be synthesized by various methods.
- a substantially monodispersed mixture of oligomers consisting of carboxylic acid and polyethylene glycol is synthesized by contacting a substantially monodispersed mixture of carboxylic acid with a substantially monodispersed mixture of polyethylene glycol under conditions sufficient to provide a substantially monodispersed mixture of oligomers.
- the oligomers of the substantially monodispersed mixture are then activated so that they are capable of reacting with a calcitonin drag to provide a calcitonin drug-oligomer conjugate.
- a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 3 and described in Examples 11-18 hereinbelow.
- FIG. 4 Another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 4 and described in Examples 19-24 hereinbelow. Still another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 5 and described in Examples 25-29 hereinbelow. Yet another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 6 and described in Examples 30-31 hereinbelow. Another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 7 and described in Examples 32-37 hereinbelow.
- Still another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 8 and described in Example 38 hereinbelow. Yet another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 9 and described in Example 39 hereinbelow. Another embodiment of a synthesis route for providing a substantially monodispersed mixture of oligomers is illustrated in Figure 10 and described in Example 40 hereinbelow.
- the substantially monodispersed mixture of activated oligomers may be reacted with a substantially monodispersed mixture of calcitonin drugs under conditions sufficient to provide a mixture of calcitonin drug-oligomer conjugates.
- a preferred synthesis is described in Example 41 hereinbelow.
- reaction conditions e.g., selected molar ratios, solvent mixtures and/or pH
- the reaction conditions may be controlled such that the mixture of calcitonin drug-oligomer conjugates resulting from the reaction of the substantially monodispersed mixture of activated oligomers and the substantially monodispersed mixture of calcitonin drugs is a substantially monodispersed mixture.
- conjugation at the amino functionality of lysine may be suppressed by maintaining the pH of the reaction solution below the pK a of lysine.
- the mixture of calcitonin drug-oligomer conjugates may be separated and isolated utilizing, for example, HPLC to provide a substantially monodispersed mixture of calcitonin drug-oligomer conjugates, for example mono-, di-, or tri-conjugates.
- the degree of conjugation e.g., whether the isolated molecule is a mono-, di-, or tri-conjugate
- a particular isolated conjugate may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, mass spectroscopy.
- the 1 1 I S particular conjugate stracture (e.g., whether the oligomer is at Lys , Lys or the N-terminus of a salmon calcitonin monoconjugate) may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, sequence analysis, peptide mapping, selective enzymatic cleavage, and/or endopeptidase cleavage.
- reaction sites on the calcitonin drag may be blocked by, for example, reacting the calcitonin drug with a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- t-BOC N-tert-butoxycarbonyl
- N-FMOC N-(9- fluorenylmethoxycarbonyl)
- the substantially monodispersed mixture of blocked calcitonin drugs may be reacted with the substantially monodispersed mixture of activated oligomers to provide a mixture of calcitonin drug-oligomer conjugates having oligomer(s) coupled to one or more nucleophilic residues and having blocking moieties coupled to other nucleophilic residues.
- the calcitonin drug-oligomer conjugates may be de-blocked as will be understood by those skilled in the art. If necessary, the mixture of calcitonin drug-oligomer conjugates may then be separated as described above to provide a substantially monodispersed mixture of calcitonin drug- oligomer conjugates.
- the mixture of calcitonin drug-oligomer conjugates may be separated prior to de-blocking.
- Substantially monodispersed mixtures of calcitonin drug-oligomer conjugates according to embodiments of the present invention preferably have improved properties when compared with those of conventional mixtures.
- a substantially monodispersed mixture of calcitonin-oligomer conjugates preferably is capable of lowering serum calcium levels by at least 5 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least 10, 11, 12, 13 or 14 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least 15, 16, 17, 18 or 19 percent, and, most preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 20 percent.
- a substantially monodispersed mixture of calcitonin-oligomer conjugates preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin, respectively, of the calcitonin drug which is not coupled to the oligomer. Resistance to chymotrypsin or trypsin corresponds to the percent remaining when the molecule to be tested is digested in the applicable enzyme using the procedure outlined in Example 51 below.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug- oligomer conjugates is about 10 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drugs that is not conjugated with the oligomer.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 15 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 30 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- a substantially monodispersed mixture of calcitonin- oligomer conjugates preferably has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- the bioefficacy of a particular compound corresponds to its area-under-the-curve (AUC) value.
- AUC area-under-the-curve
- the bioefficacy of the mixture is about 5 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer. More preferably, the bioefficacy of the mixture is about 10 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- a substantially monodispersed mixture of calcitonin-oligomer conjugates preferably has an in vivo activity that is greater than the in vivo activity of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the substantially monodispersed mixture.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography such as gel permeation chromatography as described, for example, in H.R. Allcock & F.W. Lampe, CONTEMPORARY POLYMER CHEMISTRY 394-402 (2d. ed., 1991).
- a substantially monodispersed mixture of calcitonin-oligomer conjugates preferably has an in vitro activity that is greater than the in vitro activity of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the substantially monodispersed mixture.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a substantially monodispersed mixture of calcitonin- oligomer conjugates preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the substantially monodispersed mixture.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a substantially monodispersed mixture of calcitonin-oligomer conjugates preferably has an inter-subject variability that is less than the inter-subject variability of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the substantially monodispersed mixture.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- the inter-subject variability may be measured by various methods, as will be understood by those skilled in the art.
- the inter-subject variability is preferably calculated as follows.
- the area under a dose response curve (i.e., the area between the dose- response curve and a baseline value) is determined for each subject.
- the average AUC for all subjects is determined by summing the AUCs of each subject and dividing the sum by the number of subjects.
- the absolute value of the difference between the subject's AUC and the average AUC is then determined for each subject.
- the absolute values of the differences obtained are then summed to give a value that represents the inter-subject variability. Lower values represent lower inter-subject variabilities and higher values represent higher inter- subject variabilities.
- Substantially monodispersed mixtures of calcitonin drug-oligomer conjugates according to embodiments of the present invention preferably have two or more of the above- described improved properties.
- substantially monodispersed mixtures of calcitonin drug-oligomer conjugates according to embodiments of the present invention have three or more of the above-described improved properties. Most preferably, substantially monodispersed mixtures of calcitonin drug-oligomer conjugates according to embodiments of the present invention have four or more of the above-described improved properties.
- a mixture of conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons is provided. Each conjugate in the mixture includes a calcitonin drug coupled to an oligomer that comprises a polyethylene glycol moiety. The standard deviation is preferably less than about 14 Daltons and is more preferably less than about 11 Daltons.
- the molecular weight distribution may be determined by methods known to those skilled in the art including, but not limited to, size exclusion chromatography such as gel permeation chromatography as described, for example, in H.R. Allcock & F.W. Lampe, CONTEMPORARY POLYMER CHEMISTRY 394-402 (2d. ed., 1991). The standard deviation of the molecular weight distribution may then be determined by statistical methods as will be understood by those skilled in the art.
- the calcitonin drug is preferably calcitonin. More preferably, the calcitonin drag is salmon calcitonin.
- the calcitonin drug may be selected from various calcitonin drugs known to those skilled in the art including, for example, calcitonin precursor peptides, calcitonin, calcitonin analogs, calcitonin fragments, and calcitonin fragment analogs.
- Calcitonin precursor peptides include, but are not limited to, katacalcin (PDN-21) (C-procalcitonin), and N-proCT (amino-terminal procalcitonin cleavage peptide), human.
- Calcitonin analogs may be provided by substitution of one or more amino acids in calcitonin as described above.
- Calcitonin fragments include, but are not limited to, calcitonin 1-7, human; and calcitonin 8-32, salmon. Calcitonin fragment analogs may be provided by substitution of one or more of the amino acids in a calcitonin fragment as described above.
- the oligomer may be various oligomers comprising a polyethylene glycol moiety as will be understood by those skilled in the art.
- the polyethylene glycol moiety of the oligomer has at least 2, 3 or 4 polyethylene glycol subunits. More preferably, the polyethylene glycol moiety has at least 5 or 6 polyethylene glycol subunits and, most preferably, the polyethylene glycol moiety has at least 7 polyethylene glycol subunits.
- the oligomer may comprise one or more other moieties as will be understood by those skilled in the art including, but not limited to, additional hydrophilic moieties, lipophilic moieties, spacer moieties, linker moieties, and terminating moieties.
- the various moieties in the oligomer are covalently coupled to one another by either hydrolyzable or non- hydrolyzable bonds.
- the oligomer may further comprise one or more additional hydrophilic moieties (i.e., moieties in addition to the polyethylene glycol moiety) including, but not limited to, sugars, polyalkylene oxides, and polyamine/PEG copolymers.
- the additional hydrophilic moiety may be a polyethylene glycol moiety.
- Adjacent polyethylene glycol moieties will be considered to be the same moiety if they are coupled by an ether bond.
- the moiety — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — is a single polyethylene glycol moiety having six polyethylene glycol subunits. If this moiety were the only hydrophilic moiety in the oligomer, the oligomer would not contain an additional hydrophilic moiety.
- Adjacent polyethylene glycol moieties will be considered to be different moieties if they are coupled by a bond other than an ether bond.
- the moiety — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — is a single polyethylene glycol moiety having six polyethylene glycol subunits. If this moiety were the
- oligomers according to embodiments of the present invention comprise a polyethylene glycol moiety and no additional hydrophilic moieties.
- the oligomer may further comprise one or more lipophilic moieties as will be understood by those skilled in the art.
- the lipophilic moiety is preferably a saturated or unsaturated, linear or branched alkyl moiety or a saturated or unsaturated, linear or branched fatty acid moiety.
- the lipophilic moiety is an alkyl moiety, it is preferably a linear, saturated or unsaturated alkyl moiety having 1 to 28 carbon atoms.
- the alkyl moiety has 2 to 12 carbon atoms.
- the lipophilic moiety is a fatty acid moiety, it is preferably a natural fatty acid moiety that is linear, saturated or unsaturated, having 2 to 18 carbon atoms. More preferably, the fatty acid moiety has 3 to 14 carbon atoms. Most preferably, the fatty acid moiety has at least 4, 5 or 6 carbon atoms.
- the oligomer may further comprise one or more spacer moieties as will be understood by those skilled in the art.
- Spacer moieties may, for example, be used to separate a hydrophilic moiety from a lipophilic moiety, to separate a lipophilic moiety or hydrophilic moiety from the calcitonin drug, to separate a first hydrophilic or lipophilic moiety from a second hydrophilic or lipophilic moiety, or to separate a hydrophilic moiety or lipophilic moiety from a linker moiety.
- Spacer moieties are preferably selected from the group consisting of sugar, cholesterol and glycerine moieties.
- the oligomer may further comprise one or more linker moieties that are used to couple the oligomer with the calcitonin drug as will be understood by those skilled in the art.
- Linker moieties are preferably selected from the group consisting of alkyl and fatty acid moieties.
- the oligomer may further comprise one or more terminating moieties at the one or more ends of the oligomer which are not coupled to the calcitonin drug.
- the terminating moiety is preferably an alkyl or alkoxy moiety, and is more preferably a lower alkyl or lower alkoxy moiety. Most preferably, the terminating moiety is methyl or methoxy. While the terminating moiety is preferably an alkyl or alkoxy moiety, it is to be understood that the terminating moiety may be various moieties as will be understood by those skilled in the art including, but not limited to, sugars, cholesterol, alcohols, and fatty acids.
- the oligomer is preferably covalently coupled to the calcitonin drug.
- the calcitonin drug is coupled to the oligomer utilizing a hydrolyzable bond (e.g., an ester or carbonate bond).
- a hydrolyzable coupling may provide a calcitonin drag- oligomer conjugate that acts as a prodrug.
- a hydrolyzable coupling may provide for a time-release or controlled-release effect, administering the calcitonin drag over a given time period as one or more oligomers are cleaved from their respective calcitonin drug-oligomer conjugates to provide the active drug.
- the calcitonin drug is coupled to the oligomer utilizing a non-hydrolyzable bond (e.g., a carbamate, amide, or ether bond).
- the oligomer When the oligomer is covalently coupled to the calcitonin drug, the oligomer further comprises one or more bonding moieties that are used to covalently couple the oligomer with the calcitonin drag as will be understood by those skilled in the art. Bonding moieties are preferably selected from the group consisting of covalent bond(s), ester moieties, carbonate moieties, carbamate moieties, amide moieties and secondary amine moieties.
- More than one moiety on the oligomer may be covalently coupled to the calcitonin drug. While the oligomer is preferably covalently coupled to the calcitonin drug, it is to be understood that the oligomer may be non-covalently coupled to the calcitonin drug to form a non-covalently conjugated calcitonin drug-oligomer complex. As will be understood by those skilled in the art, non-covalent couplings include, but are not limited to, hydrogen bonding, ionic bonding, Nan der Waals bonding, and micellular or liposomal encapsulation.
- oligomers may be suitably constructed, modified and/or appropriately functionalized to impart the ability for non-covalent conjugation in a selected manner (e.g., to impart hydrogen bonding capability), as will be understood by those skilled in the art.
- oligomers may be derivatized with various compounds including, but not limited to, amino acids, oligopeptides, peptides, bile acids, bile acid derivatives, fatty acids, fatty acid derivatives, salicylic acids, salicylic acid derivatives, aminosalicylic acids, and aminosalicylic acid derivatives.
- the resulting oligomers can non-covalently couple (complex) with drug molecules, pharmaceutical products, and/or pharmaceutical excipients.
- the resulting complexes preferably have balanced lipophilic and hydrophilic properties.
- oligomers may be derivatized with amine and/or alkyl amines. Under suitable acidic conditions, the resulting oligomers can form non- covalently conjugated complexes with drug molecules, pharmaceutical products and/or pharmaceutical excipients.
- the products resulting from such complexation preferably have balanced lipophilic and hydrophilic properties. More than one oligomer (i.e., a plurality of oligomers) may be coupled to the calcitonin drug.
- the oligomers in the plurality are preferably the same. However, it is to be understood that the oligomers in the plurality may be different from one another, or, alternatively, some of the oligomers in the plurality may be the same and some may be different.
- a plurality of oligomers are coupled to the calcitonin drug, it may be preferable to couple one or more of the oligomers to the calcitonin drug with hydrolyzable bonds and couple one or more of the oligomers to the calcitonin drag with non-hydrolyzable bonds.
- all of the bonds coupling the plurality of oligomers to the calcitonin drug may be hydrolyzable, but have varying degrees of hydrolyzability such that, for example, one or more of the oligomers is rapidly removed from the calcitonin drug by hydrolysis in the body and one or more of the oligomers is slowly removed from the calcitonin drag by hydrolysis in the body.
- the oligomer may be coupled to the calcitonin drug at various nucleophilic residues of the calcitonin drag including, but not limited to, nucleophilic hydroxyl functions and/or amino functions.
- a nucleophilic hydroxyl function may be found, for example, at serine and/or tyrosine residues, and a nucleophilic amino function may be found, for example, at histidine and/or lysine residues, and/or at the one or more N-termini of the polypeptide.
- the coupling preferably forms a secondary amine.
- the oligomer may be coupled to an amino functionality of the salmon calcitonin, including the amino functionality of Lys 11 , Lys and/or the N-terminus. While one or more oligomers may be coupled to the salmon calcitonin, a higher bioefficacy, such as improved serum calcium lowering ability, is observed for the di-conjugated salmon calcitonin where an oligomer is coupled to the amino functionalities of Lys 11 and the Lys 18 . Mixtures of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons may be synthesized by various methods.
- a mixture of oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons consisting of carboxylic acid and polyethylene glycol is synthesized by contacting a mixture of carboxylic acid having a molecular weight distribution with a standard deviation of less than about 22 Daltons with a mixture of polyethylene glycol having a molecular weight distribution with a standard deviation of less than about 22 Daltons under conditions sufficient to provide a mixture of oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the oligomers of the mixture having a molecular weight distribution with a standard deviation of less than about 22 Daltons are then activated so that they are capable of reacting with a calcitonin drug to provide a calcitonin drug-oligomer conjugate.
- a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 3 and described in Examples 11-18 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 4 and described in Examples 19-24 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 5 and described in Examples 25-29 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 6 and described in Examples 30-31 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 7 and described in Examples 32-37 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 8 and described in Example 38 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 9 and described in Example 39 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is illustrated in Figure 10 and described in Example 40 hereinbelow.
- the mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons is reacted with a mixture of calcitonin drugs having a molecular weight distribution with a standard deviation of less than about 22 Daltons under conditions sufficient to provide a mixture of calcitonin drug-oligomer conjugates.
- a preferred synthesis is described in Example 41 hereinbelow.
- the reaction conditions e.g., selected molar ratios, solvent mixtures and/or pH
- the mixture of calcitonin drug- oligomer conjugates resulting from the reaction of the mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons and the mixture of calcitonin drags having a molecular weight distribution with a standard deviation of less than about 22 Daltons is a mixture having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- conjugation at the amino functionality of lysine may be suppressed by maintaining the pH of the reaction solution below the pK a of lysine.
- the mixture of calcitonin drug-oligomer conjugates may be separated and isolated utilizing, for example, HPLC to provide a mixture of calcitonin drug-oligomer conjugates, for example mono-, di-, or tri-conjugates, having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the degree of conjugation e.g., whether the isolated molecule is a mono-, di-, or tri-conjugate
- a particular isolated conjugate may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, mass spectroscopy.
- the particular conjugate stracture (e.g., whether the oligomer is at Lys , Lys or the N-terminus of a salmon calcitonin monoconjugate) may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, sequence analysis, peptide mapping, selective enzymatic cleavage, and/or endopeptidase cleavage.
- one or more of the reaction sites on the calcitonin drug may be blocked by, for example, reacting the calcitonin drug with a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- t-BOC N-tert-butoxycarbonyl
- N-FMOC N-(9- fluorenylmethoxycarbonyl)
- the mixture of blocked calcitonin drugs having a molecular weight distribution with a standard deviation of less than about 22 Daltons may be reacted with the mixture of activated oligomers having a molecular weight distribution with a standard deviation of less than about 22 Daltons to provide a mixture of calcitonin drug-oligomer conjugates having oligomer(s) coupled to one or more nucleophilic residues and having blocking moieties coupled to other nucleophilic residues.
- the calcitonin drug-oligomer conjugates may be de-blocked as will be understood by those skilled in the art.
- the mixture of calcitonin drug-oligomer conjugates may then be separated as described above to provide a mixture of calcitonin drug- oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the mixture of calcitonin drug-oligomer conjugates may be separated prior to de-blocking.
- Mixtures of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons according to embodiments of the present invention preferably have improved properties when compared with those of conventional mixtures.
- a mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably is capable of lowering serum calcium levels by at least 5 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least 10, 11, 12, 13 or 14 percent. More preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 15, 16, 17, 18 or 19 percent, and, most preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 20 percent.
- a mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin, respectively, of the calcitonin drug which is not coupled to the oligomer.
- Resistance to chymotrypsin or trypsin corresponds to the percent remaining when the molecule to be tested is digested in the applicable enzyme using a procedure similar to the one outlined in Example 51 below.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug- oligomer conjugates is about 10 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drags that is not conjugated with the oligomer.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 15 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drag that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 30 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- a mixture of calcitonin drag-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- the bioefficacy of a particular compound corresponds to its area- under-the-curve (AUC) value.
- AUC area- under-the-curve
- the bioefficacy of the mixture is about 5 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer. More preferably, the bioefficacy of the mixture is about 10 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- a mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has an in vivo activity that is greater than the in vivo activity of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography such as gel permeation chromatography as described, for example, in H.R.
- a mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has an in vitro activity that is greater than the in vitro activity of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drag-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons preferably has an inter-subject variability that is less than the inter-subject variability of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- the inter-subject variability may be measured by various methods, as will be understood by those skilled in the art.
- the inter-subject variability is preferably calculated as follows.
- the area under a dose response curve (AUC) i.e., the area between the dose- response curve and a baseline value
- the average AUC for all subjects is determined by summing the AUCs of each subject and dividing the sum by the number of subjects.
- the absolute value of the difference between the subject's AUC and the average AUC is then determined for each subject.
- mixtures of calcitonin drug-oligomer conjugates having a molecular weight distribution with a standard deviation of less than about 22 Daltons have four or more of the above-described improved properties.
- a mixture of conjugates is provided where each conjugate includes a calcitonin drag coupled to an oligomer comprising a polyethylene glycol moiety, and the mixture has a dispersity coefficient (DC) greater than 10,000 where
- the mixture of conjugates preferably has a dispersity coefficient greater than 100,000. More preferably, the dispersity coefficient of the conjugate mixture is greater than 500,000 and, most preferably, the dispersity coefficient is greater than 10,000,000.
- the variables n, Nj, and Mi may be determined by various methods as will be understood by those skilled in the art, including, but not limited to, methods described below in Example 49.
- the calcitonin drug is preferably calcitonin. More preferably, the calcitonin drug is salmon calcitonin.
- the calcitonin drug may be selected from various calcitonin drugs known to those skilled in the art including, for example, calcitonin precursor peptides, calcitonin, calcitonin analogs, calcitonin fragments, and calcitonin fragment analogs.
- Calcitonin precursor peptides include, but are not limited to, katacalcin (PDN-21) (C-procalcitonin), and N-proCT (amino-terminal procalcitonin cleavage peptide), human.
- Calcitonin analogs may be provided by substitution of one or more amino acids in calcitonin as described above.
- Calcitonin fragments include, but are not limited to, calcitonin 1-7, human; and calcitonin 8-32, salmon. Calcitonin fragment analogs may be provided by substitution of one or more of the amino acids in a calcitonin fragment as described above.
- the oligomer may be various oligomers comprising a polyethylene glycol moiety as will be understood by those skilled in the art.
- the polyethylene glycol moiety of the oligomer has at least 2, 3 or 4 polyethylene glycol subunits. More preferably, the polyethylene glycol moiety has at least 5 or 6 polyethylene glycol subunits and, most preferably, the polyethylene glycol moiety has at least 7 polyethylene glycol subunits.
- the oligomer may comprise one or more other moieties as will be understood by those skilled in the art including, but not limited to, additional hydrophilic moieties, lipophilic moieties, spacer moieties, linker moieties, and terminating moieties.
- the various moieties in the oligomer are covalently coupled to one another by either hydrolyzable or non- hydrolyzable bonds.
- the oligomer may further comprise one or more additional hydrophilic moieties (i.e., moieties in addition to the polyethylene glycol moiety) including, but not limited to, sugars, polyalkylene oxides, and polyamine/PEG copolymers.
- the additional hydrophilic moiety may be a polyethylene glycol moiety. Adjacent polyethylene glycol moieties will be considered to be the same moiety if they are coupled by an ether bond.
- the moiety — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — is a single polyethylene glycol moiety having six polyethylene glycol subunits. If this moiety were the only hydrophilic moiety in the oligomer, the oligomer would not contain an additional hydrophilic moiety.
- Adjacent polyethylene glycol moieties will be considered to be different moieties if they are coupled by a bond other than an ether bond.
- the moiety O II —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —C-O—C 2 H 4 —O—C 2 H 4 is a polyethylene glycol moiety having four polyethylene glycol subunits and an additional hydrophilic moiety having two polyethylene glycol subunits.
- oligomers according to embodiments of the present invention comprise a polyethylene glycol moiety and no additional hydrophilic moieties.
- the oligomer may further comprise one or more lipophilic moieties as will be understood by those skilled in the art.
- the lipophilic moiety is preferably a saturated or unsaturated, linear or branched alkyl moiety or a saturated or unsaturated, linear or branched fatty acid moiety.
- the lipophilic moiety is an alkyl moiety, it is preferably a linear, saturated or unsaturated alkyl moiety having 1 to 28 carbon atoms. More preferably, the alkyl moiety has 2 to 12 carbon atoms.
- the lipophilic moiety is a fatty acid moiety, it is preferably a natural fatty acid moiety that is linear, saturated or unsaturated, having 2 to 18 carbon atoms. More preferably, the fatty acid moiety has 3 to 14 carbon atoms.
- the fatty acid moiety has at least 4, 5 or 6 carbon atoms.
- the oligomer may further comprise one or more spacer moieties as will be understood by those skilled in the art. Spacer moieties may, for example, be used to separate a hydrophilic moiety from a lipophilic moiety, to separate a lipophilic moiety or hydrophilic moiety from the calcitonin drag, to separate a first hydrophilic or lipophilic moiety from a second hydrophilic or lipophilic moiety, or to separate a hydrophilic moiety or lipophilic moiety from a linker moiety.
- Spacer moieties are preferably selected from the group consisting of sugar, cholesterol and glycerine moieties.
- the oligomer may further comprise one or more linker moieties that are used to couple the oligomer with the calcitonin drug as will be understood by those skilled in the art.
- Linker moieties are preferably selected from the group consisting of alkyl and fatty acid moieties.
- the oligomer may further comprise one or more terminating moieties at the one or more ends of the oligomer which are not coupled to the calcitonin drag.
- the terminating moiety is preferably an alkyl or alkoxy moiety, and is more preferably a lower alkyl or lower alkoxy moiety. Most preferably, the terminating moiety is methyl or methoxy.
- the terminating moiety is preferably an alkyl or alkoxy moiety, it is to be understood that the terminating moiety may be various moieties as will be understood by those skilled in the art including, but not limited to, sugars, cholesterol, alcohols, and fatty acids.
- the oligomer is preferably covalently coupled to the calcitonin drug.
- the calcitonin drug is coupled to the oligomer utilizing a hydrolyzable bond (e.g., an ester or carbonate bond).
- a hydrolyzable coupling may provide a calcitonin drug- oligomer conjugate that acts as a prodrug.
- a hydrolyzable coupling may provide for a time-release or controlled-release effect, administering the calcitonin drug over a given time period as one or more oligomers are cleaved from their respective calcitonin drug-oligomer conjugates to provide the active drug.
- the calcitonin drag is coupled to the oligomer utilizing a non-hydrolyzable bond (e.g., a carbamate, amide, or ether bond).
- the oligomer When the oligomer is covalently coupled to the calcitonin drug, the oligomer further comprises one or more bonding moieties that are used to covalently couple the oligomer with the calcitonin drug as will be understood by those skilled in the art. Bonding moieties are preferably selected from the group consisting of covalent bond(s), ester moieties, carbonate moieties, carbamate moieties, amide moieties and secondary amine moieties.
- More than one moiety on the oligomer may be covalently coupled to the calcitonin drug. While the oligomer is preferably covalently coupled to the calcitonin drug, it is to be understood that the oligomer may be non-covalently coupled to the calcitonin drag to form a non-covalently conjugated calcitonin drug-oligomer complex. As will be understood by those skilled in the art, non-covalent couplings include, but are not limited to, hydrogen bonding, ionic bonding, Nan der Waals bonding, and micellular or liposomal encapsulation.
- oligomers may be suitably constracted, modified and/or appropriately functionalized to impart the ability for non-covalent * conjugation in a selected manner (e.g., to impart hydrogen bonding capability), as will be understood by those skilled in the art.
- oligomers may be derivatized with various compounds including, but not limited to, amino acids, oligopeptides, peptides, bile acids, bile acid derivatives, fatty acids, fatty acid derivatives, salicylic acids, salicylic acid derivatives, aminosalicylic acids, and aminosalicylic acid derivatives.
- the resulting oligomers can non-covalently couple (complex) with drag molecules, pharmaceutical products, and/or pharmaceutical excipients.
- the resulting complexes preferably have balanced lipophilic and hydrophilic properties.
- oligomers may be derivatized with amine and/or alkyl amines. Under suitable acidic conditions, the resulting oligomers can form non- covalently conjugated complexes with drug molecules, pharmaceutical products and/or pharmaceutical excipients.
- the products resulting from such complexation preferably have balanced lipophilic and hydrophilic properties. More than one oligomer (i.e., a plurality of oligomers) may be coupled to the calcitonin drug.
- the oligomers in the plurality are preferably the same. However, it is to be understood that the oligomers in the plurality may be different from one another, or, alternatively, some of the oligomers in the plurality may be the same and some may be different.
- a plurality of oligomers are coupled to the calcitonin drag, it may be preferable to couple one or more of the oligomers to the calcitonin drag with hydrolyzable bonds and couple one or more of the oligomers to the calcitonin drag with non-hydrolyzable bonds.
- all of the bonds coupling the plurality of oligomers to the calcitonin drug may be hydrolyzable, but have varying degrees of hydrolyzability such that, for example, one or more of the oligomers is rapidly removed from the calcitonin drug by hydrolysis in the body and one or more of the oligomers is slowly removed from the calcitonin drug by hydrolysis in the body.
- the oligomer may be coupled to the calcitonin drug at various nucleophilic residues of the calcitonin drug including, but not limited to, nucleophilic hydroxyl functions and/or amino functions.
- a nucleophilic hydroxyl function may be found, for example, at serine and/or tyrosine residues, and a nucleophilic amino function may be found, for example, at histidine and/or lysine residues, and/or at the one or more N-termini of the polypeptide.
- the coupling preferably forms a secondary amine.
- the oligomer may be coupled to an amino functionality of the salmon calcitonin, including the amino functionality of Lys 11 , Lys 18 and/or the N-terminus. While one or more oligomers may be coupled to the salmon calcitonin, a higher bioefficacy, such as improved serum calcium lowering ability, is observed for the di-conjugated salmon calcitonin where an oligomer is coupled to the amino functionalities of Lys 11 and the Lys 18 . Mixtures of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 may be synthesized by various methods.
- a mixture of oligomers having a dispersity coefficient greater than 10,000 consisting of carboxylic acid and polyethylene glycol is synthesized by contacting a mixture of carboxylic acid having a dispersity coefficient greater than 10,000 with a mixture of polyethylene glycol having a dispersity coefficient greater than 10,000 under conditions sufficient to provide a mixture of oligomers having a dispersity coefficient greater than 10,000.
- the oligomers of the mixture having a dispersity coefficient greater than 10,000 are then activated so that they are capable of reacting with a calcitonin drug to provide a calcitonin drag-oligomer conjugate.
- a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 3 and described in Examples 11-18 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 4 and described in Examples 19-24 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 5 and described in Examples 25-29 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 6 and described in Examples 30-31 hereinbelow.
- FIG. 7 Another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 7 and described in Examples 32-37 hereinbelow. Still another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 8 and described in Example 38 hereinbelow. Yet another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 9 and described in Example 39 hereinbelow. Another embodiment of a synthesis route for providing a mixture of activated oligomers having a dispersity coefficient greater than 10,000 is illustrated in Figure 10 and described in Example 40 hereinbelow.
- the mixture of activated oligomers having a dispersity coefficient greater than 10,000 is reacted with a mixture of calcitonin drugs having a dispersity coefficient greater than 10,000 under conditions sufficient to provide a mixture of calcitonin drug-oligomer conjugates.
- a preferred synthesis is described in Example 41 hereinbelow.
- reaction conditions e.g., selected molar ratios, solvent mixtures and/or pH
- the mixture of calcitonin drug- oligomer conjugates resulting from the reaction of the mixture of activated oligomers having a dispersity coefficient greater than 10,000 and the mixture of calcitonin drugs having a dispersity coefficient greater than 10,000 is a mixture having a dispersity coefficient greater than 10,000.
- conjugation at the amino functionality of lysine may be suppressed by maintaining the pH of the reaction solution below the pK a of lysine.
- the mixture of calcitonin drag-oligomer conjugates may be separated and isolated utilizing, for example, HPLC to provide a mixture of calcitonin drag-oligomer conjugates, for example mono-, di-, or tri-conjugates, having a dispersity coefficient greater than 10,000.
- the degree of conjugation e.g., whether the isolated molecule is a mono-, di-, or tri-conjugate
- a particular isolated conjugate may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, mass spectroscopy.
- the particular conjugate structure (e.g., whether the oligomer is at Lys 11 , Lys 18 or the N-terminus of a salmon calcitonin monoconjugate) may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, sequence analysis, peptide mapping, selective enzymatic cleavage, and/or endopeptidase cleavage.
- one or more of the reaction sites on the calcitonin drug may be blocked by, for example, reacting the calcitonin drug with a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- t-BOC N-tert-butoxycarbonyl
- N-FMOC N-(9- fluorenylmethoxycarbonyl)
- the mixture of blocked calcitonin drugs having a dispersity coefficient greater than 10,000 may be reacted with the mixture of activated oligomers having a dispersity coefficient greater than 10,000 to provide a mixture of calcitonin drug-oligomer conjugates having oligomer(s) coupled to one or more nucleophilic residues and having blocking moieties coupled to other nucleophilic residues.
- the calcitonin drug-oligomer conjugates may be de-blocked as will be understood by those skilled in the art.
- the mixture of calcitonin drug- oligomer conjugates may then be separated as described above to provide a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000.
- the mixture of calcitonin drug-oligomer conjugates may be separated prior to de-blocking.
- Mixtures of calcitonin drag-oligomer conjugates having a dispersity coefficient greater than 10,000 according to embodiments of the present invention preferably have improved properties when compared with those of conventional mixtures.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably is capable of lowering serum calcium levels by at least 5 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least
- the mixture of conjugates is capable of lowering serum calcium levels by at least 15, 16, 17, 18 or 19 percent, and, most preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 20 percent.
- a mixture of calcitonin drag-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin, respectively, of the calcitonin drug which is not coupled to the oligomer.
- Resistance to chymotrypsin or trypsin corresponds to the percent remaining when the molecule to be tested is digested in the applicable enzyme using a procedure similar to the one outlined in Example 51 below.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drugs that is not conjugated with the oligomer.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drag-oligomer conjugates is about 15 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 30 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- the bioefficacy of a particular compound corresponds to its area-under-the-curve (AUC) value.
- AUC area-under-the-curve
- the bioefficacy of the mixture is about 5 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer. More preferably, the bioefficacy of the mixture is about 10 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has an in vivo activity that is greater than the in vivo activity of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates having a dispersity coefficient greater than 10,000.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion cliromatography such as gel permeation chromatography as described, for example, in H.R. Allcock & F.W.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has an in vitro activity that is greater than the in vitro activity of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion cliromatography.
- a mixture of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 preferably has an inter-subject variability that is less than the inter-subject variability of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates having a dispersity coefficient greater than 10,000.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- the inter-subject variability may be measured by various methods, as will be understood by those skilled in the art.
- the inter-subject variability is preferably calculated as follows.
- the area under a dose response curve (i.e., the area between the dose- response curve and a baseline value) is determined for each subject.
- the average AUC for all subjects is determined by summing the AUCs of each subject and dividing the sum by the number of subjects.
- the absolute value of the difference between the subject's AUC and the average AUC is then determined for each subject.
- the absolute values of the differences obtained are then summed to give a value that represents the inter-subject variability. Lower values represent lower inter-subject variabilities and higher values represent higher inter- subject variabilities.
- Mixtures of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 according to embodiments of the present invention preferably have two or more of the above-described improved properties.
- mixtures of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 according to embodiments of the present invention have three or more of the above-described improved properties. Most preferably, mixtures of calcitonin drug-oligomer conjugates having a dispersity coefficient greater than 10,000 according to embodiments of the present invention have four or more of the above-described improved properties.
- a mixture of conjugates in which each conjugate includes a calcitonin drag coupled to an oligomer and has the same number of polyethylene glycol subunits is provided.
- the calcitonin drag is preferably calcitonin. More preferably, the calcitonin drug is salmon calcitonin.
- the calcitonin drug may be selected from various calcitonin drugs known to those skilled in the art including, for example, calcitonin precursor peptides, calcitonin, calcitonin analogs, calcitonin fragments, and calcitonin fragment analogs.
- Calcitonin precursor peptides include, but are not limited to, katacalcin (PDN-21) (C-procalcitonin), and N-proCT (amino-terminal procalcitonin cleavage peptide), human.
- Calcitonin analogs may be provided by substitution of one or more amino acids in calcitonin as described above.
- Calcitonin fragments include, but are not limited to, calcitonin 1-7, human; and calcitonin 8-32, salmon. Calcitonin fragment analogs may be provided by substitution of one or more of the amino acids in a calcitonin fragment as described above.
- the oligomer may be various oligomers comprising a polyethylene glycol moiety as will be understood by those skilled in the art.
- the polyethylene glycol moiety of the oligomer has at least 2, 3 or 4 polyethylene glycol subunits. More preferably, the polyethylene glycol moiety has at least 5 or 6 polyethylene glycol subunits and, most preferably, the polyethylene glycol moiety has at least 7 polyethylene glycol subunits.
- the oligomer may comprise one or more other moieties as will be understood by those skilled in the art including, but not limited to, additional hydrophilic moieties, lipophilic moieties, spacer moieties, linker moieties, and terminating moieties " .
- the various moieties in the oligomer are covalently coupled to one another by either hydrolyzable or non- hydrolyzable bonds.
- the oligomer may further comprise one or more additional hydrophilic moieties (i.e., moieties in addition to the polyethylene glycol moiety) including, but not limited to, sugars, polyalkylene oxides, and polyamine/PEG copolymers.
- the additional hydrophilic moiety may be a polyethylene glycol moiety. Adjacent polyethylene glycol moieties will be considered to be the same moiety if they are coupled by an ether bond.
- the moiety — O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 -O-C 2 H 4 — is a single polyethylene glycol moiety having six polyethylene glycol subunits. If this moiety were the only hydrophilic moiety in the oligomer, the oligomer would not contain an additional hydrophilic moiety.
- Adjacent polyethylene glycol moieties will be considered to be different moieties if they are coupled by a bond other than an ether bond.
- the moiety O II —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —O—C 2 H 4 —C-O—C 2 H 4 —O—C 2 H 4 — is a polyethylene glycol moiety having four polyethylene glycol subunits and an additional hydrophilic moiety having two polyethylene glycol subunits.
- oligomers according to embodiments of the present invention comprise a polyethylene glycol moiety and no additional hydrophilic moieties.
- the oligomer may further comprise one or more lipophilic moieties as will be understood by those skilled in the art.
- the lipophilic moiety is preferably a saturated or unsaturated, linear or branched alkyl moiety or a saturated or unsaturated, linear or branched fatty acid moiety.
- the lipophilic moiety is an alkyl moiety, it is preferably a linear, saturated or unsaturated alkyl moiety having 1 to 28 carbon atoms. More preferably, the alkyl moiety has 2 to 12 carbon atoms.
- the lipophilic moiety is a fatty acid moiety, it is preferably a natural fatty acid moiety that is linear, saturated or unsaturated, having 2 to 18 carbon atoms.
- the fatty acid moiety has 3 to 14 carbon atoms. Most preferably, the fatty acid moiety has at least 4, 5 or 6 carbon atoms.
- the oligomer may further comprise one or more spacer moieties as will be understood by those skilled in the art. Spacer moieties may, for example, be used to separate a hydrophilic moiety from a lipophilic moiety, to separate a lipophilic moiety or hydrophilic moiety from the calcitonin drug, to separate a first hydrophilic or lipophilic moiety from a second hydrophilic or lipophilic moiety, or to separate a hydrophilic moiety or lipophilic moiety from a linker moiety.
- Spacer moieties are preferably selected from the group consisting of sugar, cholesterol and glycerine moieties.
- the oligomer may further comprise one or more linker moieties that are used to couple the oligomer with the calcitonin drag as will be understood by those skilled in the art. Linker moieties are preferably selected from the group consisting of alkyl and fatty acid moieties.
- the oligomer may further comprise one or more terminating moieties at the one or more ends of the oligomer which are not coupled to the calcitonin drag.
- the terminating moiety is preferably an alkyl or alkoxy moiety, and is more preferably a lower alkyl or lower alkoxy moiety.
- the terminating moiety is methyl or methoxy. While the terminating moiety is preferably an alkyl or alkoxy moiety, it is to be understood that the terminating moiety may be various moieties as will be understood by those skilled in the art including, but not limited to, sugars, cholesterol, alcohols, and fatty acids.
- the oligomer is preferably covalently coupled to the calcitonin drag.
- the calcitonin drug is coupled to the oligomer utilizing a hydrolyzable bond (e.g., an ester or carbonate bond).
- a hydrolyzable coupling may provide a calcitonin drag- oligomer conjugate that acts as a prodrug.
- a hydrolyzable coupling may provide for a time-release or controlled-release effect, administering the calcitonin drug over a given time period as one or more oligomers are cleaved from their respective calcitonin drag-oligomer conjugates to provide the active drug.
- the calcitonin drag is coupled to the oligomer utilizing a non-hydrolyzable bond (e.g., a carbamate, amide, or ether bond).
- the oligomer When the oligomer is covalently coupled to the calcitonin drug, the oligomer further comprises one or more bonding moieties that are used to covalently couple the oligomer with the calcitonin drug as will be understood by those skilled in the art. Bonding moieties are preferably selected from the group consisting of covalent bond(s), ester moieties, carbonate moieties, carbamate moieties, amide moieties and secondary amine moieties.
- More than one moiety on the oligomer may be covalently coupled to the calcitonin drag. While the oligomer is preferably covalently coupled to the calcitonin drag, it is to be understood that the oligomer may be non-covalently coupled to the calcitonin drug to form a non-covalently conjugated calcitonin drag-oligomer complex. As will be understood by those skilled in the art, non-covalent couplings include, but are not limited to, hydrogen bonding, ionic bonding, Van der Waals bonding, and micellular or liposomal encapsulation.
- oligomers may be suitably constructed, modified and/or appropriately functionalized to impart the ability for non-covalent conjugation in a selected manner (e.g., to impart hydrogen bonding capability), as will be understood by those skilled in the art.
- oligomers may be derivatized with various compounds including, but not limited to, amino acids, oligopeptides, peptides, bile acids, bile acid derivatives, fatty acids, fatty acid derivatives, salicylic acids, salicylic acid derivatives, aminosalicylic acids, and aminosalicylic acid derivatives.
- the resulting oligomers can non-covalently couple (complex) with drag molecules, pharmaceutical products, and/or pharmaceutical excipients.
- the resulting complexes preferably have balanced lipophilic and hydrophilic properties.
- oligomers may be derivatized with amine and/or alkyl amines. Under suitable acidic conditions, the resulting oligomers can form non- covalently conjugated complexes with drag molecules, pharmaceutical products and/or pharmaceutical excipients.
- the products resulting from such complexation preferably have balanced lipophilic and hydrophilic properties. More than one oligomer (i.e., a plurality of oligomers) may be coupled to the calcitonin drug.
- the oligomers in the plurality are preferably the same. However, it is to be understood that the oligomers in the plurality may be different from one another, or, alternatively, some of the oligomers in the plurality may be the same and some may be different.
- a plurality of oligomers are coupled to the calcitonin drug, it may be preferable to couple one or more of the oligomers to the calcitonin drug with hydrolyzable bonds and couple one or more of the oligomers to the calcitonin drag with non-hydrolyzable bonds.
- all of the bonds coupling the plurality of oligomers to the calcitonin drug may be hydrolyzable, but have varying degrees of hydrolyzability such that, for example, one or more of the oligomers is rapidly removed from the calcitonin drug by hydrolysis in the body and one or more of the oligomers is slowly removed from the calcitonin drug by hydrolysis in the body.
- the oligomer may be coupled to the calcitonin drug at various nucleophilic residues of the calcitonin drug including, but not limited to, nucleophilic hydroxyl functions and/or amino functions.
- a nucleophilic hydroxyl function may be found, for example, at serine and/or tyrosine residues, and a nucleophilic amino function may be found, for example, at histidine and/or lysine residues, and/or at the one or more N-termini of the polypeptide.
- the coupling preferably forms a secondary amine.
- the oligomer may be coupled to an amino functionality of the salmon calcitonin, including the amino functionality of Lys , Lys 18 and/or the N-terminus. While one or more oligomers may be coupled to the salmon calcitonin, a higher bioefficacy, such as improved serum calcium lowering ability, is observed for the di-conjugated salmon calcitonin where an oligomer is coupled to the amino functionalities of Lys 11 and the Lys 18 . Mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits may be synthesized by various methods.
- a mixture of oligomers consisting of carboxylic acid and polyethylene glycol where each oligomer in the mixture has the same number of polyethylene glycol subunits is synthesized by contacting a mixture of carboxylic acid with a mixture of polyethylene glycol where each polyethylene glycol molecule in the mixture has the same number of polyethylene glycol subunits under conditions sufficient to provide a mixture of oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits.
- the oligomers of the mixture where each oligomer in the mixture has the same number of polyethylene glycol subunits are then activated so that they are capable of reacting with a calcitonin drug to provide a calcitonin drag-oligomer conjugate.
- a , synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 3 and described in Examples 11-18 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 4 and described in Examples 19-24 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 5 and described in Examples 25-29 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 6 and described in Examples 30-31 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 7 and described in Examples 32-37 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 8 and described in Example 38 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 9 and described in Example 39 hereinbelow.
- Another embodiment of a synthesis route for providing a mixture of activated oligomers having a mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits is illustrated in Figure 10 and described in Example 40 hereinbelow.
- reaction conditions e.g., selected molar ratios, solvent mixtures and/or pH
- the mixture of calcitonin drag-oligomer conjugates resulting from the reaction of the mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits and the mixture of calcitonin drags is a mixture of conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- conjugation at the amino functionality of lysine may be suppressed by maintaining the pH of the reaction solution below the pK a of lysine.
- the mixture of calcitonin drag-oligomer conjugates may be separated and isolated utilizing, for example, HPLC to provide a mixture of calcitonin drag-oligomer conjugates, for example mono-, di-, or tri-conjugates, where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the degree of conjugation e.g., whether the isolated molecule is a mono-, di-, or tri-conjugate
- a particular isolated conjugate may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, mass spectroscopy.
- the particular conjugate structure (e.g., whether the oligomer is at Lys , Lys or the N-terminus of a salmon calcitonin monoconjugate) may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, sequence analysis, peptide mapping, selective enzymatic cleavage, and/or endopeptidase cleavage.
- reaction sites on the calcitonin drag may be blocked by, for example, reacting the calcitonin drug with a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- t-BOC N-tert-butoxycarbonyl
- N-FMOC N-(9- fluorenylmethoxycarbonyl)
- the mixture of blocked calcitonin drugs may be reacted with the mixture of activated oligomers where each oligomer in the mixture has the same number of polyethylene glycol subunits to provide a mixture of calcitonin drug-oligomer conjugates having oligomer(s) coupled to one or more nucleophilic residues and having blocking moieties coupled to other nucleophilic residues.
- the calcitonin drag-oligomer conjugates may be de-blocked as will be understood by those skilled in the art.
- the mixture of calcitonin drug-oligomer conjugates may then be separated as described above to provide a mixture of calcitonin drug- oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the mixture of calcitonin drug-oligomer conjugates may be separated prior to de-blocking.
- Mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits according to embodiments of the present invention preferably have improved properties when compared with those of conventional mixtures.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably is capable of lowering serum calcium levels by at least 5 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least 10, 11, 12, 13 or 14 percent. More preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 15, 16, 17, 18 or 19 percent, and, most preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 20 percent.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin, respectively, of the calcitonin drug which is not coupled to the oligomer.
- Resistance to chymotrypsin or trypsin corresponds to the percent remaining when the molecule to be tested is digested in the applicable enzyme using a procedure similar to the one outlined in Example 51 below.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drugs that is not conjugated with the oligomer.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drag-oligomer conjugates is about 15 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 10 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by trypsin of the mixture of calcitonin drag-oligomer conjugates is about 30 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- the bioefficacy of a particular compound corresponds to its area- under-the-curve (AUC) value.
- AUC area- under-the-curve
- the bioefficacy of the mixture is about 5 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer. More preferably, the bioefficacy of the mixture is about 10 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has an in vivo activity that is greater than the in vivo activity of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography such as gel permeation chromatography as described, for example, in H.R.
- a mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has an in vitro activity that is greater than the in vitro activity of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits preferably has an inter-subject variability that is less than the inter-subject variability of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- the inter- subject variability may be measured by various methods, as will be understood by those skilled in the art.
- the inter-subject variability is preferably calculated as follows.
- the area under a dose response curve (AUC) i.e., the area between the dose-response curve and a baseline value
- the average AUC for all subjects is determined by summing the AUCs of each subject and dividing the sum by the number of subjects.
- AUC is then determined for each subject.
- the absolute values of the differences obtained are then summed to give a value that represents the inter-subject variability.
- Lower values represent lower inter-subject variabilities and higher values represent higher inter-subject variabilities.
- Mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits according to embodiments of the present invention preferably have two or more of the above-described improved properties. More preferably, mixtures of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits according to embodiments of the present invention have three or more of the above-described improved properties.
- mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number of polyethylene glycol subunits according to embodiments of the present invention have four or more of the above-described improved properties.
- a mixture of conjugates is provided in which each conjugate has the same molecular weight and has the stracture of Formula A:
- B is a bonding moiety; L is a linker moiety; G, G' and G" are individually selected spacer moieties; R is a lipophilic moiety and R' is a polyalkylene glycol moiety, or R' is the lipophilic moiety and R is the polyalkylene glycol moiety; T is a terminating moiety; j, k, m and n are individually 0 or 1; and p is an integer from 1 to the number of nucleophilic residues on the calcitonin drug.
- the calcitonin drug is preferably calcitonin. More preferably, the calcitonin drug is salmon calcitonin.
- the calcitonin drag may be selected from various calcitonin drugs known to those skilled in the art including, for example, calcitonin precursor peptides, calcitonin, calcitonin analogs, calcitonin fragments, and calcitonin fragment analogs.
- Calcitonin precursor peptides include, but are not limited to, katacalcin (PDN-21) (C-procalcitonin), and N-proCT (amino-terminal procalcitonin cleavage peptide), human.
- Calcitonin analogs may be provided by substitution of one or more amino acids in calcitonin as described above.
- Calcitonin fragments include, but are not limited to, calcitonin 1-7, human; and calcitonin 8-32, salmon. Calcitonin fragment analogs may be provided by substitution of one or more of the amino acids in a calcitonin fragment as described above.
- the polyalkylene glycol moiety of the oligomer preferably has at least 2, 3 or 4 polyalkylene glycol subunits. More preferably, the polyalkylene glycol moiety has at least 5 or 6 polyalkylene glycol subunits and, most preferably, the polyethylene glycol moiety has at least 7 polyalkylene glycol subunits.
- the polyalkylene glycol moiety is preferably a lower polyalkylene glycol moiety such as a polyethylene glycol moiety, a polypropylene glycol moiety, or a polybutylene glycol moiety. More preferably, the polyalkylene glycol moiety is a polyethylene glycol moiety or a polypropylene glycol moiety. Most preferably, the polyalkylene glycol moiety is a polyethylene glycol moiety. When the polyalkylene glycol moiety is a polypropylene glycol moiety, the moiety preferably has a uniform (i.e., not random) structure.
- An exemplary polypropylene glycol moiety having a uniform structure is as follows: — O-CH-CH 2 — O-CH-CH 2 — O-CH-CH 2 -O-CH-CH 2 — O — CH 3 CH 3 CH 3 CH 3
- This uniform polypropylene glycol structure may be described as having only one methyl substituted carbon atom adjacent each oxygen atom in the polypropylene glycol chain.
- Such uniform polypropylene glycol moieties may exhibit both lipophilic and hydrophilic characteristics and thus be useful in providing amphiphilic calcitonin drug-oligomer conjugates without the use of lipophilic polymer moieties.
- coupling the secondary alcohol moiety of the polypropylene glycol moiety with a calcitonin drag may provide the calcitonin drug (e.g., salmon calcitonin) with improved resistance to degradation caused by enzymes such as trypsin and chymotrypsin found, for example, in the gut.
- Uniform polypropylene glycol according to embodiments of the present invention is preferably synthesized as illustrated in Figures 11 through 13, which will now be described.
- 1 2-propanediol 53 is reacted with a primary alcohol blocking reagent to provide a secondary alcohol extension monomer 54.
- the primary alcohol blocking reagent may be various primary alcohol blocking reagents as will be understood by those skilled in the art including, but not limited to, silylchloride compounds such as t- butyldiphenylsilylchloride and t-butyldimethylsilylchloride, and esterification reagents such as Ac 2 O.
- the primary alcohol blocking reagent is a primary alcohol blocking reagent that is substantially non-reactive with secondary alcohols, such as t- butyldiphenylsilylchloride or t-butyldimethylsilylchloride.
- the secondary alcohol extension monomer (54) may be reacted with methanesulfonyl chloride (MeSO 2 Cl) to provide a primary extension alcohol monomer mesylate 55.
- the secondary alcohol extension monomer 54 may be reacted with a secondary alcohol blocking reagent to provide compound 56.
- the secondary alcohol blocking reagent may be various secondary alcohol blocking reagents as will be understood by those skilled in the art including, but not limited to, benzyl chloride.
- the compound 56 may be reacted with a B t de-blocking reagent to remove the blocking moiety B 1 and provide a primary alcohol extension monomer 57.
- the B ⁇ de-blocking reagent may be selected from various de-blocking reagents as will be understood by one skilled in the art.
- the Bi de-blocking reagent is a de- esterification reagent, such as a base (e.g., potassium carbonate).
- the Bi de-blocking reagent is preferably tetrabutylammonium fluoride (TBAF).
- the primary alcohol extension monomer 57 may be reacted with methane sulfonyl chloride to provide a secondary alcohol extension monomer mesylate 58.
- the primary alcohol extension monomer 54 and the secondary alcohol extension monomer 57 may be capped as follows.
- the secondary alcohol extension monomer 54 may be reacted with a capping reagent to provide a compound 59.
- the capping reagent may be various capping reagents as will be understood by those skilled in the art including, but not limited to, alkyl halides such as methyl chloride.
- the compound 59 may be reacted with a Bi de-blocking agent as described above to provide a primary alcohol capping monomer 60.
- the primary alcohol capping monomer 60 may be reacted with methane sulfonyl chloride to provide the secondary alcohol capping monomer mesylate 61.
- the primary alcohol extension monomer 57 may be reacted with a capping reagent to provide a compound 62.
- the capping reagent may be various capping reagents as described above.
- the compound 62 may be reacted with a B de-blocking reagent to remove the blocking moiety B 2 and provide a secondary alcohol capping monomer 63.
- the B 2 de-blocking reagent may be various deblocking agents as will be understood by those skilled in the art including, but not limited to, H 2 in the presence of a palladium/activated carbon catalyst.
- the secondary alcohol capping monomer may be reacted with methanesulfonyl chloride to provide a primary alcohol capping monomer mesylate 64. While the embodiments illustrated in Figure 11 show the synthesis of capping monomers, it is to be understood that similar reactions may be performed to provide capping polymers.
- chain extensions may be effected by reacting a primary alcohol extension mono- or poly-mer such as the primary alcohol extension monomer 57 with a primary alcohol extension mono- or poly-mer mesylate such as the primary alcohol extension monomer mesylate 55 to provide various uniform polypropylene chains or by reacting a secondary alcohol extension mono- or poly-mer such as the secondary alcohol extension monomer 54 with a secondary alcohol extension mono-or poly-mer mesylate such as the secondary alcohol extension monomer mesylate 58.
- the primary alcohol extension monomer mesylate 55 is reacted with the primary alcohol extension monomer 57 to provide a dimer compound 65.
- the secondary alcohol extension monomer mesylate 58 may be reacted with the secondary alcohol extension monomer 54 to provide the dimer compound 65.
- the B 1 blocking moiety on the dimer compound 65 may be removed using a Bi de-blocking reagent as described above to provide a primary alcohol extension dimer 66.
- the primary alcohol extension dimer 66 may be reacted with methane sulfonyl chloride to provide a secondary alcohol extension dimer mesylate 67.
- the B blocking moiety on the dimer compound 65 may be removed using the B de-blocking reagent as described above to provide a secondary alcohol extension dimer 69.
- the secondary alcohol extension dimer 69 may be reacted with methane sulfonyl chloride to provide a primary alcohol extension dimer mesylate 70.
- the chain extension process may be repeated to achieve various other chain lengths.
- the primary alcohol extension dimer 66 may be reacted with the primary alcohol extension dimer mesylate 70 to provide a tetramer compound 72.
- a generic chain extension reaction scheme involves reacting the primary alcohol extension mono- or poly-mer 73 with the primary alcohol extension mono- or poly-mer mesylate 74 to provide the uniform polypropylene polymer 75.
- the values of m and n may each range from 0 to 1000 or more.
- m and n are each from 0 to 50. While the embodiments illustrated in Figure 13 show primary alcohol extension mono- and/or poly-mers being reacted with primary alcohol extension mono- and/or poly-mer mesylates. it is to be understood that similar reactions may be carried out using secondary alcohol extension mono- and/or poly-mers and secondary alcohol extension mono- and/or poly-mer mesylates. An end of a primary alcohol extension mono- or poly-mer or an end of a primary alcohol extension mono- or poly-mer mesylate may be reacted with a primary alcohol capping mono- or poly-mer mesylate or a primary alcohol capping mono- or poly-mer, respectively, to provide a capped uniform polypropylene chain.
- the primary alcohol extension dimer mesylate 70 is reacted with the primary alcohol capping monomer 60 to provide the capped/blocked primary alcohol extension trimer 71.
- the Bi blocking moiety may be removed and the resulting capped primary alcohol extension trimer may be reacted with a primary alcohol extension mono- or poly-mer mesylate to extend the chain of the capped trimer 71.
- An end of a secondary alcohol extension mono-or poly-mer or an end of a secondary alcohol extension mono-or poly-mer mesylate may be reacted with a secondary alcohol capping mono-or poly-mer mesylate or a secondary alcohol capping mono- or poly-mer, respectively, to provide a capped uniform polypropylene chain.
- the secondary alcohol extension dimer mesylate 67 is reacted with the secondary alcohol capping monomer 63 to provide the capped/blocked primary alcohol extension trimer 68.
- the B blocking moiety may be removed as described above and the resulting capped secondary alcohol extension trimer may be reacted with a secondary alcohol extension mer mesylate to extend the chain of the capped trimer 68. While the syntheses illustrated in
- Figure 12 show the reaction of a dimer with a capping monomer to provide a trimer, it is to be understood that the capping process may be performed at any point in the synthesis of a uniform polypropylene glycol moiety, or, alternatively, uniform polypropylene glycol moieties may be provided that are not capped. While the embodiments illustrated in Figure 12 show the capping of a polybutylene oligomer by synthesis with a capping monomer, it is to be understood that polybutylene oligomers of the present invention may be capped directly (i.e., without the addition of a capping monomer) using a capping reagent as described above in Figure 11.
- Uniform polypropylene glycol moieties may be coupled to a calcitonin drug, a lipophilic moiety such as a carboxylic acid, and/or various other moieties by various methods as will be understood by those skilled in the art including, but not limited to, those described herein with respect to polyethylene glycol moieties.
- the lipophilic moiety is a lipophilic moiety as will be understood by those skilled in the art.
- the lipophilic moiety is preferably a saturated or unsaturated, linear or branched alkyl moiety or a saturated or unsaturated, linear or branched fatty acid moiety.
- the lipophilic moiety is an alkyl moiety, it is preferably a linear, saturated or unsaturated alkyl moiety having 1 to 28 carbon atoms. More preferably, the alkyl moiety has 2 to 12 carbon atoms.
- the lipophilic moiety is a fatty acid moiety, it is preferably a natural fatty acid moiety that is linear, saturated or unsaturated, having 2 to 18 carbon atoms. More preferably, the fatty acid moiety has 3 to 14 carbon atoms. Most preferably, the fatty acid moiety has at least 4, 5 or 6 carbon atoms.
- the spacer moieties, G, G' and G are spacer moieties as will be understood by those skilled in the art. Spacer moieties are preferably selected from the group consisting of sugar, cholesterol and glycerine moieties.
- oligomers of these embodiments do not include spacer moieties (i.e., k, m and n are preferably 0).
- the linker moiety, L may be used to couple the oligomer with the drug as will be understood by those skilled in the art.
- Linker moieties are preferably selected from the group consisting of alkyl and fatty acid moieties.
- the terminating moiety is preferably an alkyl or alkoxy moiety, and is more preferably a lower alkyl or lower alkoxy moiety. Most preferably, the terminating moiety is methyl or methoxy.
- the terminating moiety is preferably an alkyl or alkoxy moiety, it is to be understood that the terminating moiety may be various moieties as will be understood by those skilled in the art including, but not limited to, sugars, cholesterol, alcohols, and fatty acids.
- the oligomer which is represented by the bracketed portion of the stracture of Formula A, is covalently coupled to the calcitonin drug.
- the calcitonin drag is coupled to the oligomer utilizing a hydrolyzable bond (e.g., an ester or carbonate bond).
- a hydrolyzable coupling may provide a calcitonin drag-oligomer conjugate that acts as a prodrug.
- a hydrolyzable coupling may provide for a time-release or controlled-release effect, administering the calcitonin drug over a given time period as one or more oligomers are cleaved from their respective calcitonin drug-oligomer conjugates to provide the active drug.
- the calcitonin drug is coupled to the oligomer utilizing a non- hydrolyzable bond (e.g., a carbamate, amide, or ether bond).
- the bonding moiety, B may be various bonding moieties that may be used to covalently couple the oligomer with the calcitonin drag as will be understood by those skilled in the art. Bonding moieties are preferably selected from the group consisting of covalent bond(s), ester moieties, carbonate moieties, carbamate moieties, amide moieties and secondary amine moieties.
- the variable p is an integer from 1 to the number of nucleophilic residues on the calcitonin drag.
- oligomers in the plurality are the same.
- all of the bonds coupling the plurality of oligomers to the drug may be hydrolyzable, but have varying degrees of hydrolyzability such that, for example, one or more of the oligomers is rapidly removed from the drug by hydrolysis in the body and one or more of the oligomers is slowly removed from the drug by hydrolysis in the body.
- p is preferably 1 or 2, and is more preferably 2.
- the oligomer may be coupled to the calcitonin drug at various nucleophilic residues of the calcitonin drag including, but not limited to, nucleophilic hydroxyl functions and/or amino functions.
- a nucleophilic hydroxyl function may be found, for example, at serine and/or tyrosine residues, and a nucleophilic amino function may be found, for example, at histidine and/or lysine residues, and/or at the one or more N-termini of the polypeptide.
- the coupling preferably forms a secondary amine.
- the oligomer may be coupled to an amino functionality of the salmon calcitonin, including the amino functionality of Lys 11 , 1 S
- oligomers may be coupled to the salmon calcitonin, a higher bioefficacy, such as improved serum calcium lowering ability, is observed for the di-conjugated salmon calcitonin where an oligomer is coupled to the amino functionalities of Lys 11 and the Lys 18 .
- Mixtures of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A may be synthesized by various methods.
- a mixture of oligomers consisting of carboxylic acid and polyethylene glycol is synthesized by contacting a mixture of carboxylic acid with a mixture of polyethylene glycol under conditions sufficient to provide a mixture of oligomers.
- the oligomers of the mixture are then activated so that they are capable of reacting with a calcitonin drug to provide a calcitonin drag-oligomer conjugate.
- a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a structure of the oligomer of Formula A is illustrated in Figure 3 and described in Examples 11-18 hereinbelow.
- FIG. 4 Another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a structure of the oligomer of Formula A is illustrated in Figure 4 and described in Examples 19-24 hereinbelow. Still another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a stracture of the oligomer of Formula A is illustrated in Figure 5 and described in Examples 25-29 hereinbelow.
- Still another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a stracture of the oligomer of Formula A is illustrated in Figure 8 and described in Example 38 hereinbelow.
- Yet another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a structure of the oligomer of Formula A is illustrated in Figure 9 and described in Example 39 hereinbelow.
- FIG. 10 Another embodiment of a synthesis route for providing a mixture of activated oligomers where each oligomer has the same molecular weight and has a stracture of the oligomer of Formula A is illustrated in Figure 10 and described in Example 40 hereinbelow.
- the mixture of activated oligomers where each oligomer has the same molecular weight and has a structure of the oligomer of Formula A is reacted with a mixture of calcitonin drags where each drag in the mixture has the same molecular weight under conditions sufficient to provide a mixture of calcitonin drug-oligomer conjugates.
- a preferred synthesis is described in Example 41 hereinbelow.
- reaction conditions e.g., selected molar ratios, solvent mixtures and/or pH
- the mixture of calcitonin drag-oligomer conjugates resulting from the reaction of the mixture of activated oligomers where each oligomer has the same molecular weight and has a stracture of the oligomer of Formula A and the mixture of calcitonin drugs is a mixture of conjugates where each conjugate has the same molecular weight and has the stracture Formula A.
- conjugation at the amino functionality of lysine may be suppressed by maintaining the pH of the reaction solution below the pK a of lysine.
- the mixture of calcitonin drag-oligomer conjugates may be separated and isolated utilizing, for example, HPLC to provide a mixture of calcitonin drug-oligomer conjugates, for example mono-, di-, or tri-conjugates, where each conjugate in the 'mixture has the same number molecular weight and has the stracture of Formula A.
- the degree of conjugation e.g., whether the isolated molecule is a mono-, di-, or tri-co ⁇ jugate
- a particular isolated conjugate may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, mass spectroscopy.
- the particular conjugate stracture (e.g., whether the oligomer is at Lys 11 , Lys 18 or the N-terminus of a salmon calcitonin monoconjugate) may be determined and/or verified utilizing various techniques as will be understood by those skilled in the art including, but not limited to, sequence analysis, peptide mapping, selective enzymatic cleavage, and/or endopeptidase cleavage.
- one or more of the reaction sites on the calcitonin drag may be blocked by, for example, reacting the calcitonin drug with a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- a suitable blocking reagent such as N-tert-butoxycarbonyl (t-BOC), or N-(9- fluorenylmethoxycarbonyl) (N-FMOC).
- t-BOC N-tert-butoxycarbonyl
- N-FMOC N-(9- fluorenylmethoxycarbonyl)
- the mixture of blocked calcitonin drags may be reacted with the mixture of activated oligomers where each oligomer in the mixture has the same molecular weight and has a stracture of the oligomer of Formula A to provide a mixture of calcitonin drug-oligomer conjugates having oligomer(s) coupled to one or more nucleophilic residues and having blocking moieties coupled to other nucleophilic residues.
- the calcitonin drag-oligomer conjugates may be deblocked as will be understood by those skilled in the art.
- the mixture of calcitonin drag-oligomer conjugates may then be separated as described above to provide a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same number molecular weight and has the structure of Formula A.
- the mixture of calcitonin drug-oligomer conjugates may be separated prior to de-blocking.
- Mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A according to embodiments of the present invention preferably have improved properties when compared with those of conventional mixtures.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A preferably is capable of lowering serum calcium levels by at least 5 percent.
- the mixture of conjugates is capable of lowering serum calcium levels by at least 10, 11, 12, 13 or 14 percent. More preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 15, 16, 17, 18 or 19 percent, and, most preferably, the mixture of conjugates is capable of lowering serum calcium levels by at least 20 percent.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin, respectively, of the calcitonin drug which is not coupled to the oligomer.
- Resistance to chymotrypsin or trypsin corresponds to the percent remaining when the molecule to be tested is digested in the applicable enzyme using a procedure similar to the one outlined in Example 51 below.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drug- oligomer conjugates is about 10 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drags that is not conjugated with the oligomer.
- the resistance to degradation by chymotrypsin of the mixture of calcitonin drag-oligomer conjugates is about 15 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by chymotrypsin of the mixture of calcitonin drug-oligomer conjugates is about 20 percent greater than the resistance to degradation by chymotrypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drag-oligomer conjugates is about 10 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- the resistance to degradation by trypsin of the mixture of calcitonin drag-oligomer conjugates is about 20 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drag that is not conjugated with the oligomer, and, most preferably, the resistance to degradation by trypsin of the mixture of calcitonin drag-oligomer conjugates is about 30 percent greater than the resistance to degradation by trypsin of the mixture of calcitonin drug that is not conjugated with the oligomer.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of
- Formula A preferably has a higher bioefficacy than the bioefficacy of the calcitonin drug which is not coupled to the oligomer.
- the bioefficacy of a particular compound corresponds to its area-under-the-curve (AUC) value.
- AUC area-under-the-curve
- the bioefficacy of the mixture is about 5 percent greater than the bioefficacy of the calcitonin drug which is not coupled to the oligomer. More preferably, the bioefficacy of the mixture is about 10 percent greater than the bioefficacy of the calcitonin drag which is not coupled to the oligomer.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A preferably has an in vivo activity that is greater than the in vivo activity of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography such as gel permeation chromatography as described, for example, in H.R. Allcock & F.W. Lampe, CONTEMPORARY POLYMER CHEMISTRY 394-402 (2d. ed., 1991).
- a mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A preferably has an in vitro activity that is greater than the in vitro activity of a polydispersed mixture of calcitonin drag-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A preferably has an increased resistance to degradation by chymotrypsin and/or trypsin when compared to the resistance to degradation by chymotrypsin and/or trypsin of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- a mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A preferably has an inter-subject variability that is less than the inter-subject variability of a polydispersed mixture of calcitonin drug-oligomer conjugates having the same number average molecular weight as the mixture of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A.
- the number average molecular weight of a mixture may be measured by various methods including, but not limited to, size exclusion chromatography.
- the inter-subject variability may be measured by various methods, as will be understood by those skilled in the art.
- the inter-subject variability is preferably calculated as follows.
- the area under a dose response curve (AUC) i.e., the area between the dose- response curve and a baseline value
- the average AUC for all subjects is determined by summing the AUCs of each subject and dividing the sum by the number of subjects.
- the absolute value of the difference between the subject's AUC and the average AUC is then determined for each subject.
- Mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the structure of Formula A according to embodiments of the present invention preferably have two or more of the above-described improved properties. More preferably, mixtures of calcitonin drug-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A according to embodiments of the present invention have three or more of the above-described improved properties.
- mixtures of calcitonin drag-oligomer conjugates where each conjugate in the mixture has the same molecular weight and has the stracture of Formula A according to embodiments of the present invention have four or more of the above-described improved properties.
- Pharmaceutical compositions comprising a conjugate mixture according to embodiments of the present invention are also provided.
- the mixtures of calcitonin drug- oligomer conjugates described above may be formulated for administration in a pharmaceutical carrier in accordance with known techniques. See, e.g., Remington, The Science And Practice of Pharmacy (9 th Ed. 1995).
- the mixture of calcitonin drug-oligomer conjugates is typically admixed with, ter alia, a pharmaceutically acceptable carrier.
- the carrier must, of course, be acceptable in the sense of being compatible with any other ingredients in the pharmaceutical composition and should not be deleterious to the patient.
- the carrier may be a solid or a liquid, or both, and is preferably formulated with the mixture of calcitonin drug-oligomer conjugates as a unit-dose formulation, for example, a tablet, which may contain from about 0.01 or 0.5% to about 95% or 99% by weight of the mixture of calcitonin drug-oligomer conjugates.
- compositions may be prepared by any of the well known techniques of pharmacy including, but not limited to, admixing the components, optionally including one or more accessory ingredients.
- the pharmaceutical compositions according to embodiments of the present invention include those suitable for oral, rectal, topical, inhalation (e.g., via an aerosol) buccal (e.g., sub-lingual), vaginal, parenteral (e.g., subcutaneous, intramuscular, intradermal, intraarticular, intrapleural, intraperitoneal, inracerebral, intraarterial, or intravenous), topical (i.e., both skin and mucosal surfaces, including airway surfaces) and transdermal administration, although the most suitable route in any given case will depend on the nature and severity of the condition being treated and on the nature of the particular mixture of calcitonin drag-oligomer conjugates which is being used.
- compositions suitable for oral administration may be presented in discrete units, such as capsules, cachets, lozenges, or tables, each containing a predetermined amount of the mixture of calcitonin drag-oligomer conjugates; as a powder or granules; as a solution or a suspension in an aqueous or non-aqueous liquid; or as an oil-in- water or water- in-oil emulsion.
- Such formulations may be prepared by any suitable method of pharmacy which includes the step of bringing into association the mixture of calcitonin drug-oligomer conjugates and a suitable carrier (which may contain one or more accessory ingredients as noted above).
- the pharmaceutical composition according to embodiments of the present invention are prepared by uniformly and intimately admixing the mixture of calcitonin drug-oligomer conjugates with a liquid or finely divided solid carrier, or both, and then, if necessary, shaping the resulting mixture.
- a tablet may be prepared by compressing or molding a powder or granules containing the mixture of calcitonin drug- oligomer conjugates, optionally with one or more accessory ingredients.
- Compressed tablets may be prepared by compressing, in a suitable machine, the mixture in a free-flowing form, such as a powder or granules optionally mixed with a binder, lubricant, inert diluent, and/or surface active/dispersing agent(s). Molded tablets may be made by molding, in a suitable machine, the powdered compound moistened with an inert liquid binder.
- compositions suitable for buccal (sub-lingual) administration include lozenges comprising the mixture of calcitonin drag-oligomer conjugates in a flavoured base, usually sucrose and acacia or tragacanth; and pastilles comprising the mixture of calcitonin drug-oligomer conjugates in an inert base such as gelatin and glycerin or sucrose and acacia.
- Pharmaceutical compositions according to embodiments of the present invention suitable for parenteral administration comprise sterile aqueous and non-aqueous injection solutions of the mixture of calcitonin drag-oligomer conjugates, which preparations are preferably isotonic with the blood of the intended recipient.
- compositions may contain anti-oxidants, buffers, bacteriostats and solutes which render the composition isotonic with the blood of the intended recipient.
- Aqueous and non-aqueous sterile suspensions may include suspending agents and thickening agents.
- the compositions may be presented in unit ⁇ dose or multi-dose containers, for example sealed ampoules and vials, and may be stored in a freeze-dried (lyophilized) condition requiring only the addition of the sterile liquid carrier, for example, saline or water-for-injection immediately prior to use.
- sterile liquid carrier for example, saline or water-for-injection immediately prior to use.
- Extemporaneous injection solutions and suspensions may be prepared from sterile powders, granules and tablets of the kind previously described.
- an injectable, stable, sterile composition comprising a mixture of calcitonin drug-oligomer conjugates in a unit dosage form in a sealed container.
- the mixture of calcitonin drug-oligomer conjugates is provided in the form of a lyophilizate which is capable of being reconstituted with a suitable pharmaceutically acceptable carrier to form a liquid composition suitable for injection thereof into a subject.
- the unit dosage form typically comprises from about 10 mg to about 10 grams of the mixture of calcitonin drag-oligomer conjugates.
- a sufficient amount of emulsifying agent which is physiologically acceptable may be employed in sufficient quantity to emulsify the mixture of calcitonin drug-oligomer conjugates in an aqueous carrier.
- emulsifying agent is phosphatidyl choline.
- Pharmaceutical compositions suitable for rectal administration are preferably presented as unit dose suppositories. These may be prepared by admixing the mixture of calcitonin drug-oligomer conjugates with one or more conventional solid carriers, for example, cocoa butter, and then shaping the resulting mixture.
- compositions suitable for topical application to the skin preferably take the form of an ointment, cream, lotion, paste, gel, spray, aerosol, or oil.
- Carriers which may be used include petroleum jelly, lanoline, polyethylene glycols, alcohols, transdermal enhancers, and combinations of two or more thereof.
- Pharmaceutical compositions suitable for transdermal administration may be presented as discrete patches adapted to remain in intimate contact with the epidermis of the recipient for a prolonged period of time.
- Compositions suitable for transdermal administration may also be delivered by iontophoresis (see, for example, Pharmaceutical Research 3 (6):318 (1986)) and typically take the form of an optionally buffered aqueous solution of the mixture of calcitonin drag-oligomer conjugates.
- Suitable formulations comprise citrate or bis ⁇ tris buffer (pH 6) or ethanol/water and contain from 0.1 to 0.2M active ingredient.
- Methods of treating a bone disorder in a subject in need of such treatment by administering an effective amount of such pharmaceutical compositions are also provided.
- the bone disorder is preferably characterized by excessive osteoclastic bone resorption and/or hypercalcemic serum effects.
- Bone disorders that may be treated and/or prevented by methods of the present invention include, but are not limited to, osteoporosis, Paget's disease, and hypercalcemia.
- the present invention further provides methods of treating pain in a s subject in need of such treatment by administering an effective amount of a calcitonin drug-oligomer conjugate of this invention.
- treating pain refers to any type of action or mechanism that imparts a pain-relieving effect upon a subject afflicted with or experiencing the sensation of pain or at risk of experiencing pain or the sensation of pain, including reducing the sensation of pain or the report of pain or delaying the development of the sensation of pain or the report of pain. That pain is treated (e.g., by complete or partial abolition of pain symptoms) by administering the compositions of this invention according to the methods provided herein can be determined by art-known assays designed to measure, either quantitatively or qualitatively, the sensation of pain or the report or perception of pain. The sensation of pain or the report of pain can be evaluated by protocols understood by those of ordinary skill in the art to which this invention pertains.
- pain can be quantitatively assessed using a visual analog scale (VAS), which comprises a 10 cm line with "No Pain” above one end and “Worst Pain Imaginable” on the other end.
- VAS visual analog scale
- a mechanical VAS device a can be used to assess pain. Pain after surgery can be assessed using either the line or mechanical VAS.
- the phrase "treating pain” further includes prophylactic treatment of the subject to prevent the onset of the sensation of pain or the report of pain.
- treatment of pain can include a complete and/or partial abolition of the sensation of pain or the report of pain.
- treatment can include any reduction in the sensation and/or symptoms of pain including reducing the intensity and/or unpleasantness of the perceived pain.
- Pain refers to all types of pain and the methods and compositions of this invention are directed to treating a subject to relieve and/or diminish the sensation and/or report of a specific type of pain or more than one type of pain as described herein. Pain can be acute or chronic pain. Pain as described herein can include sensations such as discomfort, sensitivity, burning, pinching, stinging, etc.
- Examples of types of pain that can be treated according to the present invention include, but are not limited to, inflammation, visceral pain, neuropathic pain, lower back pain, incisional pain (pain due to or caused by an incision), post-surgical pain, peripheral pain (i.e., pain originating in muscles, tendons, etc., or in the peripheral nerves themselves), central and spinal pain (i.e., pain arising from central nervous system pathology), and post-surgical incisional pain, as well as other types of pain now known or later identified as described in the literature.
- the term “pain” also refers to nociceptive pain or nociception.
- An "effective amount” as used herein refers to an amount of a compound or composition of this invention that is sufficient to produce the desired therapeutic effect.
- the effective amount will vary with the age and physical condition of the subject, the severity of the disorder, the duration of the treatment, the nature of any concurrent treatment, the pharmaceutically acceptable carrier used, and like factors within the knowledge and expertise of those skilled in the art.
- An appropriate "effective amount" in any individual case can be determined by one of ordinary skill in the art by reference to the pertinent texts and literature and/or by using routine experimentation. (See, for example, Remington, J/ze Science And Practice of Pharmacy (9 th Ed. 1995).
- the effective amount of any mixture of calcitonin drug-oligomer conjugates, the use of which is in the scope of present invention, will vary somewhat from mixture to mixture, and patient to patient, and will depend upon factors such as the age and condition of the patient and the route of delivery.
- Such dosages can be determined in accordance with routine pharmacological procedures known to those skilled in the art. As a general proposition, a dosage from about 0.1 to about 50 mg/kg will have therapeutic efficacy, with all weights being calculated based upon the weight of the mixture of calcitonin drug-oligomer conjugates. Toxicity concerns at the higher level may restrict intravenous dosages to a lower level such as up to about 10 mg/kg, with all weights being calculated based upon the weight of the active base. A dosage from about 10 mg/kg to about 50 mg/kg may be employed for oral administration. Typically, a dosage from about 0.5 mg/kg to 5 mg/kg may be employed for intramuscular injection.
- the frequency of administration is usually one, two, or three times per day or as necessary to control the condition.
- the drug-oligomer conjugates may be administered by continuous infusion.
- the duration of treatment depends on the type of bone disorder being treated and may be for as long as the life of the patient.
- R is H or a lipophilic moiety.
- R 1 is preferably H, alkyl, aryl alkyl, an aromatic moiety, a fatty acid moiety, an ester of a fatty acid moiety, cholesteryl, or adamantyl.
- R 1 is more preferably H, lower alkyl, or an aromatic moiety.
- R 1 is most preferably H, methyl, or benzyl.
- n is from 1 to 25.
- n is from 1 to 6.
- X is a positive ion.
- X + is any positive ion in a compound, such as a strong base, that is capable of ionizing a hydroxyl moiety on PEG.
- positive ions include, but are not limited to, sodium ions, potassium ions, lithium ions, cesium ions, and thallium ions.
- R is H or a lipophilic moiety.
- R is preferably linear or branched alkyl, aryl alkyl, an aromatic moiety, a fatty acid moiety, or an ester of a fatty acid moiety.
- R 2 is more preferably lower alkyl, benzyl, a fatty acid moiety having 1 to 24 carbon atoms, or an ester of a fatty acid moiety having 1 to 24 carbon atoms.
- R 2 is most preferably methyl, a fatty acid moiety having 1 to 18 carbon atoms or an ethyl ester of a fatty acid moiety having 1 to 18 carbon atoms.
- m is from 1 to 25.
- m is from 1 to 6.
- Ms is a mesylate moiety (i.e., CH 3 S(O 2 )-).
- a mixture of compounds having the structure of Formula I is reacted with a mixture of compounds having the structure of Formula II to provide a mixture of polymers comprising polyethylene glycol moieties and having the stracture of Formula III.
- the mixture of compounds having the structure of Formula I is a substantially monodispersed mixture.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula I have the same molecular weight, and, more preferably, the mixture of compounds of Formula I is a monodispersed mixture.
- the mixture of compounds of Formula II is a substantially monodispersed mixture.
- At least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula II have the same molecular weight, and, more preferably, the mixture of compounds of Formula II is a monodispersed mixture.
- the mixture of compounds of Formula III is a substantially monodispersed mixture.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compound of Formula III have the same molecular weight. More preferably, the mixture of compounds of Formula III is a monodispersed mixture.
- Reaction 1 is preferably performed between about 0°C and about 40°C, is more preferably performed between about 15°C and about 35°C, and is most preferably performed at room temperature (approximately 25 °C).
- Reaction 1 may be performed for various periods of time as will be understood by those skilled in the art. Reaction 1 is preferably performed for a period of time between about 0.25, 0.5 or 0.75 hours and about 2, 4 or 8 hours. Reaction 1 is preferably carried out in an aprotic solvent such as, but not limited to, N,N-dimethylacetamide (DMA), N,N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), hexamethylphosphoric triamide, tetrahydrofuran (THF), dioxane, diethyl ether, methyl t-butyl ether (MTBE), toluene, benzene, hexane, pentane, N-methylpyrollidinone, tetrahydronaphthalene, decahydronaphthalene, 1,2-dichlorobenzene, l,3-dimethyl-2- imidazolidinone, or a mixture thereof.
- DMA N,N
- the solvent is DMF, DMA or toluene.
- the molar ratio of the compound of Formula I to the compound of Formula II is preferably greater than about 1:1. More preferably, the molar ratio is at least about 2:1.
- the compound capable of ionizing a hydroxyl moiety is preferably a strong base. More preferably, the compound capable of ionizing a hydroxyl moiety is selected from the group consisting of sodium hydride, potassium hydride, sodium t-butoxide, potassium t-butoxide, butyl lithium (BuLi), and lithium diisopropylamine.
- the compound capable of ionizing a hydroxyl moiety is more preferably sodium hydride.
- the molar ratio of the compound capable of ionizing a hydroxyl moiety on the PEG moiety of the compound of Formula IV to the compound of Formula IV is preferably at least about 1 :1, and is more preferably at least about 2:1.
- Reaction 2 is preferably performed between about 0°C and about 40°C, is more preferably performed between about 0°C and about 35°C, and is most preferably performed between about 0°C and room temperature (approximately 25°C). Reaction 2 may be performed for various periods of time as will be understood by those skilled in the art. Reaction 2 is preferably performed for a period of time between about 0.25, 0.5 or 0.75 hours and about 2, 4 or 8 hours.
- Reaction 2 is preferably carried out in an aprotic solvent such as, but not limited to, N,N-dimethylacetamide (DMA), N,N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), hexamethylphosphoric triamide, tetrahydrofuran (THF), dioxane, diethyl ether, methyl t-butyl ether (MTBE), toluene, benzene, hexane, pentane, N-methylpyrollidinone, dichloromethane, chloroform, tetrahydronaphthalene, decahydronaphthalene, 1,2- dichlorobenzene, l,3-dimethyl-2-imidazolidinone, or a mixture thereof.
- an aprotic solvent such as, but not limited to, N,N-dimethylacetamide (DMA), N,N-dimethylformamide (DMF), dimethyl sulfoxide (DM
- the solvent is DMF, dichloromethane or toluene.
- Compounds of Formula II are preferably prepared as illustrated in reaction 3: O I I 2 R 2 (OC 2 H 4 ) m OH + CH 3 SQ > ⁇ R (OC 2 H4) m OMs 3 (V) ° (II) R and Ms are as described above and the compound of Formula V is present as a substantially monodispersed mixture of compounds of Formula V; preferably at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula V have the same molecular weight; and, more preferably, the mixture of compounds of Formula V is a monodispersed mixture.
- Q is a halide, preferably chloride or fluoride.
- CH 3 S(O 2 )Q is methanesulfonyl halide.
- the methanesulfonyl halide is preferably methanesulfonyl chloride or methanesulfonyl fluoride. More preferably, the methanesulfonyl halide is methanesulfonyl chloride.
- the molar ratio of the methane sulfonyl halide to the compound of Formula V is preferably greater than about 1:1, and is more preferably at least about 2:1.
- Reaction 3 is preferably performed between about -10°C and about 40°C, is more preferably performed between about 0°C and about 35°C, and is most preferably performed between about 0°C and room temperature (approximately 25°C). Reaction 3 may be performed for various periods of time as will be understood by those skilled in the art. Reaction 3 is preferably performed for a period of time between about 0.25, 0.5 or 0J5 hours and about 2, 4 or 8 hours.
- Reaction 3 is preferably carried out in the presence of an aliphatic amine including, but not limited to, monomethylamine, dimethylamine, trimethylamine, monoethylamine, diethylamine, triethylamine, monoisopropylamine, diisopropylamine, mono-n-butylamine, di- n-butylamine, tri-n-butylamine, monocyclohexylamine, dicyclohexylamine, or mixtures thereof. More preferably, the aliphatic amine is a tertiary amine such as triethylamine. As will be understood by those skilled in the art, various substantially monodispersed mixtures of compounds of Formula V are commercially available.
- the compounds of Formula V are PEG or mPEG compounds, respectively, which are commercially available from Aldrich of Milwaukee, Wisconsin; Fluka of Switzerland, and/or TCI America of Portland, Oregon.
- R is a lipophilic moiety such as, for example, higher alkyl, fatty acid, an ester of a fatty acid, cholesteryl, or adamantyl
- the compounds of Formula V may be provided by various methods as will be understood by those skilled in the art.
- the compounds of Formula V are preferably provided as follows:
- R is a lipophilic moiety, preferably higher alkyl, fatty acid ester, cholesteryl, or adamantyl, more preferably a lower alkyl ester of a fatty acid, and most preferably an ethyl ester of a fatty acid having from 1 to 18 carbon atoms.
- R is H, benzyl, trityl, tetrahydropyran, or other alcohol protecting groups as will be understood by those skilled in the art.
- X 2 + is a positive ion as described above with respect to X + .
- m is as described above.
- a mixture of compounds of Formula VI is reacted with a mixture of compounds of Formula VII under reaction conditions similar to those described above with reference to reaction 1.
- the mixture of compounds of Formula VI is a substantially monodispersed mixture.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula VI have the same molecular weight.
- the mixture of compounds of Formula VI is a monodispersed mixture.
- the mixture of compounds of Formula VII is a substantially monodispersed mixture.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula VII have the same molecular weight.
- the mixture of compounds of Formula VII is a monodispersed mixture.
- the compound of Formula VIII may be hydrolyzed to convert the R 3 moiety into an alcohol by various methods as will be understood by those skilled in the art.
- R 3 is benzyl or trityl
- the hydrolysis is preferably performed utilizing H 2 in the presence of a palladium-charcoal catalyst as is known by those skilled in the art.
- reaction 5 is unnecessary.
- the compound of Formula VI may be commercially available or be provided as described above with reference to reaction 3.
- the compound of Formula VII may be provided as described above with reference to reaction 2.
- Substantially monodispersed mixtures of polymers comprising PEG moieties and having the stracture of Formula III above can further be reacted with other substantially monodispersed polymers comprising PEG moieties in order to extend the PEG chain.
- the following scheme may be employed:
- Ms, m and n are as described above with reference to reaction 1; p is similar to n and m, and X 2 + is similar to X + as described above with reference to reaction 1.
- Q is as described above with reference to reaction 3.
- R is as described above with reference to reaction 1 and is preferably lower alkyl.
- R 1 is H.
- Reaction 6 is preferably performed in a manner similar to that described above with reference to reaction 3.
- Reaction 7 is preferably performed in a manner similar to that described above with reference to reaction 1.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula III have the same molecular weight, and, more preferably, the mixture of compounds of Formula III is a monodispersed mixture.
- the mixture of compounds of Formula X is a substantially monodispersed mixture.
- at least about 96, 97, 98 or 99 percent of the compounds in the mixture of compounds of Formula X have the same molecular weight, and, more preferably, the mixture of compounds of Formula X is a monodispersed mixture.
- a process according to embodiments of the present invention is illustrated by the scheme shown in Figure 1, which will now be described.
- the synthesis of substantially monodispersed polyethylene glycol-containing oligomers begins by the preparation of the monobenzyl ether (1) of a substantially monodispersed polyethylene glycol.
- the mesylate (5) of this alcohol may be prepared by addition of methanesulfonyl chloride and used as the electrophile in the reaction with the sodium salt of the monomethyl ether of a substantially monodispersed polyethylene glycol derivative, thereby extending the polyethylene glycol portion of the oligomer to the desired length, obtaining the elongated ester (6).
- the ester may be hydrolyzed to the acid (7) in aqueous base and transformed into the activated ester (8) by reaction with a carbodiimide and N-hydroxysuccinimide.
- oligomer illustrated in Figure 1 is activated using N-hydroxysuccinimide
- various other reagents may be used to activate oligomers of the present invention including, but not limited to, active phenyl chloroformates such as j-? ⁇ r ⁇ -nitrophenyl chloroformate, phenyl chloroformate, 3,4-phenyldichloroformate, and 3,4-phenyldichloroformate; tresylation; and acetal formation.
- q is from 1 to 24.
- q is from 1 to 18, and q is more preferably from 4 to 16.
- R 4 is a moiety capable of undergoing hydrolysis to provide the carboxylic acid.
- R 4 is preferably lower alkyl and is more preferably ethyl.
- the variables n and m are as described above with reference to reaction 1. All starting materials used in the procedures described herein are either commercially available or can be prepared by methods known in the art using commercially available starting materials. The present invention will now be described with reference to the following examples. It should be appreciated that these examples are for the purposes of illustrating aspects of the present invention, and do not limit the scope of the invention as defined by the claims.
- Examples 1 through 10 Reactions in Examples 1 through 10 were carried out under nitrogen with magnetic stirring, unless otherwise specified. "Work-up” denotes extraction with an organic solvent, washing of the organic phase with saturated NaCl solution, drying (MgSO 4 ), and evaporation (rotary evaporator). Thin layer chromatography was conducted with Merck glass plates precoated with silica gel 60°F - 254 and spots were visualized by iodine vapor. All mass spectra were determined by Macromolecular Resources Colorado State University, CO and are reported in the order m/z, (relative intensity). Elemental analyses and melting points were performed by Galbraith Laboratories, Inc., Knoxville, TN. Examples 1-10 refer to the scheme illustrated in Figure 2.
- Example 1 8-Methoxy-l-(methylsulfonyl)oxy-3,6-dioxaoctane (9)
- a solution of non-polydispersed triethylene glycol monomethyl ether molecules (4.00 mL, 4.19 g, 25.5 mmol) and triethylamine (4.26 mL, 3.09 g, 30.6 mmol) in dry dichloromethane (50 mL) was chilled in an ice bath and place under a nitrogen atmosphere.
- a solution of methanesulfonyl chloride (2.37 mL, 3.51 g, 30.6 mmol) in dry dichloromethane (20 mL) was added dropwise from an addition funnel. Ten minutes after the completion of the chloride addition, the reaction mixture was removed from the ice bath and allowed to come to room temperature. The mixture was stirred for an additional hour, at which time
- the crude product mixture was purified via flash chromatography (silica gel, gradient elution: ethyl acetate to 9/1 ethyl acetate/methanol) to yield 8.099 g (70 %) of non-polydispersed 16 as a yellow oil.
- Example 12 Ethyl 6-methylsulfonyloxyhexanoate (17) A solution of non-polydispersed ethyl 6-hydroxyhexanoate (50.76 ml, 50.41 g, 227 mmol) in dry dichloromethane (75 ml) was chilled in a ice bath and placed under a nitrogen atmosphere. Triethylamine (34.43 ml, 24.99 g, 247 mmol) was added. A solution of methanesulfonyl chloride (19.15 ml, 28.3 g, 247 mmol) in dry dichloromethane (75 ml) was added dropwise from an addition funnel.
- Example 13 6- ⁇ 2-[2-(2- ⁇ 2-[2-(2-Benzyloxyethoxy)ethoxy]ethoxy ⁇ -ethoxy)- ethoxy]-ethoxy ⁇ -hexanoic acid ethyl ester (18)
- Sodium hydride (3.225 g or a 60 % oil dispersion, 80.6 mmol) was suspended in 80 ml of anhydrous toluene, placed under a nitrogen atmosphere and cooled in an ice bath.
- a solution of the non-polydispersed alcohol 16 (27.3 g, 73.3 mmol) in 80 ml dry toluene was added to the NaH suspension.
- the mixture was stirred at 0°C for thirty minutes, allowed to come to room temperature and stirred for another five hours, during which time the mixture became a clear brown solution.
- the non-polydispersed mesylate 17 (19.21 g, 80.6 mmol) in 80 ml dry toluene was added to the NaH/alcohol mixture, and the combined solutions were stirred at room temperature for three days.
- the reaction mixture was quenched with 50 ml methanol and filtered through basic alumina.
- Example 14 6- ⁇ 2-[2-(2- ⁇ 2-[2-(2-hydroxyethoxy)ethoxy]ethoxy ⁇ -ethoxy)- ethoxy]-ethoxy ⁇ -hexanoic acid ethyl ester (19)
- Non-polydispersed benzyl ether 18 (1.03 g, 2.0 mmol) was dissolved in 25 ml ethanol.
- To this solution was added 270 mg 10 % Pd/C, and the mixture was placed under a hydrogen atmosphere and stirred for four hours, at which time TLC showed the complete disappearance of the starting material.
- Example 15 6- ⁇ 2-[2-(2- ⁇ 2-[2-(2-methylsuIfonyIethoxy)ethoxy]ethoxy ⁇ - ethoxy)-ethoxy]-ethoxy ⁇ -hexanoic acid ethyl ester (20)
- the non-polydispersed alcohol 19 (0.835 g, 1.97 mmol) was dissolved in 3.5 ml dry dichloromethane and placed under a nitrogen atmosphere.
- Triethylamine (0.301 ml, 0.219 g, 2.16 mmol) was added and the mixture was chilled in an ice bath. After two minutes, the methanesulfonyl chloride (0.16 ml, 0.248 g, 2.16 mmol) was added.
- Example 16 6-(2- ⁇ 2-[2-(2- ⁇ 2-[2-(2-methoxyethoxy)ethoxy]-ethoxy ⁇ -ethoxy)- ethoxy]-ethoxy ⁇ -ethoxy)-hexanoic acid ethyl ester (21) NaH (88 mg of a 60 % dispersion in oil, 2.2 mmol) was suspended in anhydrous toluene (3 ml) under N 2 and chilled to 0 C. Non-polydispersed diethylene glycol monomethyl ether (0.26 ml, 0.26 g, 2.2 mmol) that had been dried via azeotropic distillation with toluene was added.
- Example 17 6-(2- ⁇ 2-[2-(2- ⁇ 2-[2-(2-methoxyethoxy)ethoxy]-ethoxy ⁇ - ethoxy)-ethoxy]-ethoxy ⁇ -ethoxy)-hexanoic acid (22)
- Non-polydispersed ester 21 (0.25 g, 0.46 mmol) was stirred for 18 hours in 0.71 ml of 1 N NaOH. After 18 hours, the mixture was concentrated in vacuo to remove the alcohol and the residue dissolved in a further 10 ml of water. The aqueous solution was acidified to pH 2 with 2 N HCl and the product was extracted into dichloromethane (30 ml x 2).
- Example 18 6-(2- ⁇ 2-[2-(2- ⁇ 2-[2-(2-methoxyethoxy)ethoxy]-ethoxy ⁇ -ethoxy)-ethoxy]-ethoxy ⁇ -ethoxy)- hexanoic acid 2,5-dioxo-pyrrolidin-l-yl ester (23)
- Non-polydispersed acid 22 (0.209 g, 0.42 mmol) were dissolved in 4 ml of dry dichloromethane and added to a dry flask already containing NHS (N-hydroxysuccinimide) (57.8 mg, 0.502 mmol) and EDC (l-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride) (98.0 mg, 0.502 mmol) under a N 2 atmosphere.
- NHS N-hydroxysuccinimide
- EDC l-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydroch
- Example 19 Mesylate of triethylene glycol monomethyl ether (24) To a solution of CH 2 C1 (100 mL) cooled to 0°C in an ice bath was added non- polydispersed triethylene glycol monomethyl ether (25 g, 0.15 mol). Then triethylamine (29.5 mL, 0.22 mol) was added and the solution was stirred for 15 min at 0°C, which was followed by dropwise addition of methanesulfonyl chloride (13.8 mL, 0.18 mol, dissolved in 20 mL CH 2 C1 ). The reaction mixture was stirred for 30 min at 0°C, allowed to warm to room temperature, and then stirred for 2 h.
- the crude reaction mixture was filtered through Celite (washed CH 2 C1 2 -200 mL), then washed with H 2 O (300 mL), 5% NaHCO 3 (300 mL), H 2 O (300 mL), sat. NaCl (300 mL), dried MgSO , and evaporated to dryness. The oil was then placed on a vacuum line for ⁇ 2h to ensure dryness and afforded the non-polydispersed title compound as a yellow oil (29.15 g, 80% yield).
- Example 20 Heptaethylene glycol monomethyl ether (25) To a solution of non-polydispersed tetraethylene glycol (51.5 g, 0.27 mol) in THF (IL) was added potassium t-butoxide (14.8 g, 0.13 mol, small portions over -30 min). The reaction mixture was then stirred for lh and then 24 (29.15 g, 0.12 mol) dissolved in THF (90 mL) was added dropwise and the reaction mixture was stirred overnight. The crude reaction mixture was filtered through Celite (washed CH C1 2 , -200 mL) and evaporated to dryness.
- the oil was then dissolved in HCl (250 mL, 1 N) and washed with ethyl acetate (250 mL) to remove excess 24. Additional washings of ethyl acetate (125 mL) may be required to remove remaining 24.
- the aqueous phase was washed repetitively with CH 2 C1 2 (125 mL volumes) until most of the 25 has been removed from the aqueous phase.
- the first extraction will contain 24, 25, and dicoupled side product and should be back extracted with HCl (125 mL, IN).
- the organic layers were combined and evaporated to dryness.
- the resultant oil was then dissolved in CH 2 C1 2 (100 mL) and washed repetitively with H 2 O (50 mL volumes) until 25 was removed.
- the aqueous fractions were combined, total volume 500 mL, and NaCl was added until the solution became cloudy and then was washed with CH 2 C1 2 (2 x 500 mL).
- the organic layers were combined, dried MgSO 4 , and evaporated to dryness to afford a the non- polydispersed title compound as an oil (16.9 g, 41% yield). It may be desirable to repeat one or more steps of the purification procedure to ensure high purity.
- Example 21 8-Bromooctoanate (26) To a solution of 8-bromooctanoic acid (5.0 g, 22 mmol) in ethanol (100 mL) was added H 2 SO (0.36 mL, 7.5 mmol) and the reaction was heated to reflux with stirring for 3 h. The crude reaction mixture was cooled to room temperature and washed H 2 O (100 mL), sat. NaHCO 3 (2 x 100 mL), H 2 O (100 mL), dried MgSO , and evaporated to dryness to afford a clear oil (5.5 g, 98% yield).
- Example 22 Synthesis of MPEG7-C8 ester (27) To a solution of the non-polydispersed compound 25 (3.0 g, 8.8 mmol) in ether (90 mL) was added potassium t-butoxide (1.2 g, 9.6 mmol) and the reaction mixture was stirred for 1 h. Then dropwise addition of the non-polydispersed compound 26 (2.4 g, 9.6 mmol), dissolved in ether (10 mL), was added and the reaction mixture was stirred overnight. The crude reaction mixture was filtered through Celite (washed CH 2 C1 2 , -200 mL) and evaporated to dryness.
- Example 23 MPEG7-C8 acid (28) To the oil of the non-polydispersed compound 27 (0J0 g, 1.4 mmol) was added IN NaOH (2.0 mL) and the reaction mixture was stirred for 4h. The crude reaction mixture was concentrated, acidified (pH ⁇ 2), saturated with NaCl, and washed CH 2 C1 2 (2 x 50 mL). The organic layers were combined, washed sat. NaCl, dried MgSO 4 , and evaporated to dryness to afford the non-polydispersed title compound as a clear oil (0.35 g, 53% yield).
- Non-polydispersed mPEG7-C8-acid 28 (0.3 lg, 0.64 mmol) was dissolved in 3 ml of anhydrous methylene chloride and then solution of N-hydroxysuccinimide (0.079g, 0.69 mmol) and EDCI ⁇ C1 (135.6 mg, 0J1 mmol) in anhydrous methylene chloride added. Reaction was stirred for several hours, then washed with IN HCl, water, dried over MgSO 4 , filtered and concentrated. Crude material was purified by column chromatography, concentrated to afford the non-polydispersed title compound as a clear oil and dried via vacuum. Examples 25 through 29 refer to the scheme illustrated in Figure 5.
- Example 25 10-hydroxydecanoate (30) To a solution of non-polydispersed 10-hydroxydecanoic acid (5.0 g, 26.5 mmol) in ethanol (100 mL) was added H 2 S0 4 (0.43 mL, 8.8 mmol) and the reaction was heated to reflux with stirring for 3 h. The crude reaction mixture was cooled to room temperature and washed H 2 O (100 mL), sat. NaHCO 3 (2 x 100 mL), H 2 0 (100 mL), dried MgSO 4 , and evaporated to dryness to afford the non-polydispersed title compound as a clear oil (6.9 g, 98% yield).
- the crade reaction mixture was filtered through Celite (washed CH 2 C1 2 , 80 mL) and the filtrate was washed H 2 O (100 mL), 5% NaHCO 3 (2 x 100 mL), H 2 O (100 mL), sat. NaCl (100 mL), dried MgSO , and evaporated to dryness to afford the non-polydispersed title compound as a yellowish oil (7.42 g, 97% yield).
- Example 27 MPEG 7 -C 10 Ester (32) To a solution of non-polydispersed heptaethylene glycol monomethyl ether 25 (2.5 g, 7.3 mmol) in tetrahydrofuran (100 mL) was added sodium hydride (0.194 g, 8.1 mmol) and the reaction mixture was stirred for 1 h. Then dropwise addition of mesylate of non- polydispersed 10-hydroxydecanoate 31 (2.4 g, 8.1 mmol), dissolved in tetrahydrofuran (10 mL), was added and the reaction mixture was stirred overnight. The crude reaction mixture was filtered through Celite (washed CH 2 C1 2 , -200 mL) and evaporated to dryness.
- Example 28 MPEG 7 -C ⁇ o Acid (33) To the oil of non-polydispersed mPEG -C 1 o ester 32 (0.570 g, 1.1 mmol) was added IN NaOH (1.6 mL) and the reaction mixture was stirred overnight. The crude reaction mixture was concentrated, acidified (pH ⁇ 2), saturated with NaCl, and washed CH 2 C1 2 (2 x 50 mL). The organic layers were combined, washed sat. NaCl (2 x 50 mL), dried MgSO 4 , and evaporated to dryness to afford the non-polydispersed title compound as a clear oil (0.340 g, 62% yield).
- Example 29 Activation of MPEG 7 -C ⁇ 0 Acid (34) The non-polydispersed acid 33 was activated using procedures similar to those described above in Example 24. Examples 30 and 31 refer to the scheme illustrated in Figure 6.
- Non-polydispersed stearoyl chloride 35 (0.7g, 2.31 mmol) was added slowly to a mixture of PEG6 (5 g, 17J mmol) and pyridine (0.97g, 12.4 mmol) in benzene. The reaction mixture was stirred for several hours (-5). The reaction was followed by TLC using ethylacetate/methanol as a developing solvent. Then the. reaction mixture was washed with water, dried over MgSO 4 , concentrated and dried via vacuum. Purified non-polydispersed compound 36 was analyzed by FABMS: m/e 549/ M + H.
- Example 31 Activation of C18(PEG6) Oligomer Activation of non-polydispersed C18(PEG6) oligomer was accomplished in two steps: 1) Non-polydispersed stearoyl-PEG6 36 ( 0.8 g, 1.46 mmol ) was dissolved in toluene and added to a phosgene solution (10 ml, 20 % in toluene) which was cooled with an ice bath. The reaction mixture was stirred for 1 h at 0 C and then for 3 h at room temperature. Then phosgene and toluene were distilled off and the remaining non-polydispersed stearoyl PEG6 chloroformate 37 was dried over P O 5 overnight.
- Example 32 Tetraethylene glycol monobenzylether (39) To the oil of non-polydispersed tetraethylene glycol (19.4 g, 0.10 mol) was added a solution of NaOH (4.0 g in 4.0 mL) and the reaction was stirred for 15 mm. Then benzyl chloride (3.54 mL, 30.8 mmol) was added and the reaction mixture was heated to 100°C and stirred overnight. The reaction mixture was cooled to room temperature, diluted with sat. NaCl (250 mL), and washed CH 2 C1 2 (2 x 200 mL). The organic layers were combined, washed sat. NaCl, dried MgSO , and chromatographed (silica, ethyl acetate) to afford the non-polydispersed title compound as a yellow oil (6.21 g, 71% yield).
- Example 33 Mesylate of tetraethylene glycol monobenzylether (40) To a solution of CH 2 CI 2 (20 mL) was added non-polydispersed tetraethylene glycol monobenzylether 39 (6.21 g, 22 mmol) and cooled to 0°C in an ice bath. Then triethylamine
- the crude reaction mixture was filtered through Celite (washed, CH C1 2 , 250 mL) arid the filtrate was washed H 2 O, dried MgSO 4 , and evaporated to dryness.
- the resultant oil was chromatographed (silica, ethyl acetate/methanol, 10:1) and chromatographed (silica, chloroform/methanol, 25:1) to afford the non-polydispersed title compound as a clear oil (2.62 g, 34% yield).
- Example 37 Activation of C18(PEG8) Oligomer Two step activation of non-polydispersed stearate-PEG8 oligomer was performed as described for stearate-PEG6 in Example 31 above to provide the non-polydispersed activated
- Example 38 Synthesis of Activated Triethylene Glycol Monomethyl Oligomers The following description refers to the scheme illustrated in Figure 8.
- a solution of toluene containing 20% phosgene (100 ml, approximately 18J g, 189 mmol phosgene) was chilled to 0°C under a N 2 atmosphere.
- Non-polydispersed mTEG triethylene glycol, monomethyl ether, 7.8 g, 47.5 mmol
- the remaining phosgene, ethyl acetate and toluene were removed via vacuum distillation to leave the non-polydispersed mTEG chloroformate 46 as a clear oily residue.
- the non-polydispersed residue 46 was dissolved in 50 mL of dry dichloromethane to which was added TEA (triethyleamine, 6.62 mL, 47.5 mmol) and NHS
- Example 39 Synthesis of Activated Palmitate-TEG Oligomers The following description refers to the scheme illustrated in Figure 9.
- Non- polydispersed palmitic anhydride (5 g; 10 mmol) was dissolved in dry THF (20 mL) and stirred at room temperature.
- 3 mol excess of pyridine was added followed by non-polydispersed triethylene glycol (1.4 mL).
- the reaction mixture was stirred for 1 hour (progress of the reaction was monitored by TLC; ethyl acetate-chloroform; 3:7).
- THF was removed and the product was mixed with 10% H 2 SO acid and extracted ethyl acetate (3 x 30 mL).
- non-polydispersed product 48 was washed sequentially with water, brine, dried over MgSO , and evaporated to give non-polydispersed product 48.
- a solution of N,N'-disuccinimidyl carbonate (3 mmol) in DMF (-10 mL) is added to a solution of the non-polydispersed product 48 (1 mmol) in 10 mL of anydrous DMF while stirring.
- Sodium hydride (3 mmol) is added slowly to the reaction mixture. The reaction mixture is stirred for several hours (e.g., 5 hours). Diethyl ether is added to precipitate the activated oligomer. This process is repeated 3 times and the product is finally dried.
- Example 40 Synthesis of Activated Hexaethylene Glycol Monomethyl Oligomers The following description refers to the scheme illustrated in Figure 10.
- Non- polydispersed activated hexaethylene glycol monomethyl ether was prepared analogously to that of non-polydispersed triethylene glycol in Example 39 above.
- a 20%> phosgene in toluene solution 35 mL, 6.66 g, 67.4 mmol phosgene was chilled under a N atmosphere in an ice/salt water bath.
- Non-polydispersed hexaethylene glycol 50 (1.85 mL, 2.0 g, 6.74 mmol) was dissolved in 5 mL anhydrous EtOAc and added to the phosgene solution via syringe. The reaction mixture was kept stirring in the ice bath for one hour, removed and stirred a further 2.5 hours at room temperature. The phosgene, EtOAc, and toluene were removed by vacuum distillation, leaving non-polydispersed compound 51 as a clear, oily residue. The non-polydispersed residue 51 was dissolved in 20 mL dry dichloromethane and placed under a dry, inert atmosphere.
- Example 41 150 mg of salmon calcitonin (MW 3432, 0.043 mmol) was dissolved in 30 ml of anhydrous DMF. Then TEA (35 ⁇ L) and the activated oligomer of Example 24 (42 mg, 0.067 mmol) in anhydrous THF (2 mL) was added. The reaction was stirred for 1 hour, then quenched with 2 mL of 0.1 % TFA in water. The reaction was followed by HPLC. Then the reaction mixture was concentrated and purified by prep. HPLC (RC Vydac C18 Protein and peptide, 1x25 column, water/acetonitrile with 0.1 % TFA, detection at 280 nm).
- Example 42 The procedure of Example 41 was used to conjugate salmon calcitonin with the activated oligomer of Example 29. MS for PEG7-decyl-sCT, mono-conjugate: 3926. MS for PEG7-decyl-sCT, di-conjugate: 4420.
- Example 43 The procedure of Example 41 was used to conjugate salmon calcitonin with the activated oligomer of Example 31. MS for stearate-PEG6-sCT, mono-conjugate: 4006. MS for stearate-PEG6-sCT, di-conjugate: 4582.
- Example 44 The procedure of Example 41 was used to conjugate salmon calcitonin with the activated oligomer of Example 37. MS for stearate-PEG8-sCT, mono-conjugate: 4095.
- Example 45 The procedure of Example 41 is used to conjugate salmon calcitonin with the activated oligomer of Example 18.
- Example 46 The procedure of Example 41 is used to ' conjugate salmon calcitonin with the activated oligomer of Example 38.
- Example 47 The procedure of Example 41 is used to conjugate salmon calcitonin with the activated oligomer of Example 39.
- Example 48 The procedure of Example 41 is used to conjugate salmon calcitonin with the activated oligomer of Example 40.
- Example 49 Determination of the Dispersity Coefficient for a Mixture of Salmon Calcitonin-Oligomer Conjugates
- the dispersity coefficient of a mixture of salmon calcitonin-oligomer conjugates is determined as follows. A mixture of salmon calcitonin-oligomer conjugates is provided, for example as described above in Example 41. A first sample of the mixture is purified via
- the mixture may include one or more of the following conjugates, which are described by stating the conjugation position followed by the degree of conjugation: Lys 11 monoconjugate; Lys 18 monoconjugate; N- terminus monoconjugate; Lys 11 ' 18 diconjugate; Lys 11 , N-terminus diconjugate; Lys 18 , N- terminus diconjugate; and/or Lys 11,18 , N-terminus triconjugate.
- Each isolated fraction of the mixture is analyzed via mass spectroscopy to determine the mass of the fraction, which allows each isolated fraction to be categorized as a mono-, di-, or tri-conjugate and provides a value for the variable "Mj" for each conjugate in the sample.
- a second sample of the mixture is analyzed via HPLC to provide an HPLC trace.
- the weight percent of a particular conjugate in the mixture is provided by the area under the peak of the HPLC trace corresponding to the particular conjugate as a percentage of the total area under all peaks of the HPLC trace.
- the sample is collected and lyophilized to dryness to determine the anhydrous gram weight of the sample.
- the gram weight of the sample is multiplied by the weight percent of each component in the sample to determine the gram weight of each conjugate in the sample.
- the variable "Nj" is determined for a particular conjugate (the i* 1 conjugate) by dividing the gram weight of the particular conjugate in the sample by the mass of the particular conjugate and multiplying the quotient by Avagadro's T 1 number (6.02205 x 10 mole " ), Mj, determined above, to give the number of molecules of the particular conjugate, Nj, in the sample.
- the dispersity coefficient is then calculated using n, Mj as determined for each conjugate, and Nj as determined for each conjugate.
- Example 50 Cytosensor Studies T-47D cells (mammary ductal carcinoma cell line, obtained from American Type Culture Collection were suspended at a density of 1 x 10 7 cells/mL in running buffer (low- buffered, serum- free, bicarbonate-free RPMI 1640 medium from Molecular Devices of Sunnyvale, California. Approximately 100,000 cells were then immobilized in an agarose cell entrapment medium in a 10 ⁇ L droplet and sandwiched between two 3- ⁇ m polycarbonate membranes in a cytosensor capsule cup. Cytosensor capsule cups placed in sensor chambers on the Cytosensor ® Microphysiometer were then held in very close proximity to pH-sensitive detectors. Running buffer was then pumped across the cells at a rate of 100 ⁇ L/min except during 30-second intervals when the flow was stopped, and acidification of the running buffer in the sensor chamber was measured. Acidification rates were determined every 2 minutes.
- the temperature of the sensor chambers was 37°C.
- Cells were allowed to equilibrate in the sensor chambers for 2-3 hours prior to the start of the experiment during which time basal acidification rates were monitored.
- Cells were then exposed to test compounds (Salmon Calcitonin or Octyl-Di-Calcitonin) diluted in running buffer at various nM concentration. Exposure of cells to test compounds occurred for the first 40 seconds of each 2 minute pump cycle in a repeating pattern for a total of 20 minutes. This allowed sufficient exposure of the cells to the test compounds to elicit a receptor-mediated response in cellular metabolism followed by approximately 50 seconds of flow of the running buffer containing no compounds.
- Example 51 Enzymatic Stability Compounds, supplied as lyophilized powders, are resuspended in 10 mM phosphate buffer pH 7.4 and then submitted for concentration determination by HPLC.
- the phosphate buffer is used to create a solution with a pH that is optimum for activity of each particular gut enzyme. Aliquots of the compound thus prepared are transferred to 1.7 mL microcentrifuge tubes and shaken in a 37°C water bath for 15 minutes to allow compounds to equilibrate to temperature. After 15 minutes, 2 ⁇ L of the appropriate concentrated gut enzyme is added to each tube to achieve the final concentration desired. Chymotrypsin and trypsin are resuspended in 1 mM HCl.
- a control compounds are treated with 2 ⁇ L of 1 mM HCl.
- a sampling procedure is repeated at various time intervals depending on the gut enzyme used. Chymotrypsin has 15, 30 and 60 minute samples. Trypsin has 30, 60, 120 and 180 minute samples. Once all points have been acquired, a final sample is removed from the control tube to make sure that observed degradation is not temperature or buffer related.
- the chymotrypsin and trypsin samples may be collected directly into HPLC vials.
- RP-HPLC acetonitrile gradient
- AUC % degradation
- Example 52 Activity and Inter-Subject Variability
- Male CF-1 mice (Charles River, Raleigh, NC) weighing 20-25 g were housed in the Nobex vivarium in a light- (L:D cycle of 12:12, lights on at 0600 h), temperature- (21-23°C), and humidity- (40-60 % relative humidity) controlled room. Animals were permitted free access to laboratory chow (PMI Nutrition) and tap water. Mice were allowed to acclimate to housing conditions for 48-72 hours prior to the day of experiment. Prior to dosing, mice were fasted overnight and water was provided ad libitum.
- mice were randomly distributed into groups of five animals per time point and were administered a single oral dose of a PEG7-octyl-sCT, diconjugate (Octyl Di) according to the present invention or salmon calcitonin (sCT or Calcitonin) for comparison purposes.
- Oral doses were administered using a gavaging needle (Popper #18, 5 cm from hub to bevel) at 10 mL/kg in the following 0.2 ⁇ g/mL phosphate-buffered PEG7-octyl-sCT, diconjugate, formulation:
- the buffered formulation was prepared by adding 80 mL of phosphate buffer in a clean tared glass beaker. The sodium cholate was slowly added to the phosphate buffer with stirring until dissolved. The deoxy cholate was then added and stirring was continued until dissolved. The PEG7-octyl-sCT, diconjugate, solution equivalent to 20 ⁇ g was added. Finally, the remaining phosphate buffer was added to achieve a final weight of 100 g. Vehicle-control mice were used in all experiments. Dose-response curves were constracted using a single time point 60 minutes after drag administration. These curves are illustrated in Figures 15- 18.
- mice were ether-anesthetized, the vena cavae exteriorized, and blood samples were obtained via a syringe fitted with a 25-gauge needle. Blood aliquots were allowed to clot at 22°C for 1 hour, and the sera removed and pipetted into a clean receptacle. Total serum calcium was determined for each animal using a calibrated Vitros DT60 II analyzer. Serum calcium data were plotted and pharmacokinetic parameters determined via curve-fitting techniques using SigmaPlot software (Version 4.1). Means and standard deviations (or standard errors) were calculated and plotted to determine effect differences among dosing groups. Average serum calcium data for various conjugates are provided in Table 5 below.
- Example 50 Despite an in vitro activity as determined in Example 50 above that may not be comparable with the in vitro activity of PEG7-octyl-sCT and PEG7-decyl-sCT mono- and di- conjugates, the stearate-PEG6-sCT, diconjugate, and stearate-PEG8-sCT, diconjugate, appear to have in vivo activity (as evidenced by the drops in % baseline calcium from Table 5 above) that are comparable with the in vivo activity observed for the PEG7-octyl-sCT and PEG7-decyl-sCT, mono- and di-conjugates.
- the improved in vivo activity of the stearate containing conjugates may indicate that these conjugates are undergoing hydrolysis in vivo to provide an active salmon calcitonin or active salmon calcitonin-PEG conjugate.
- the objective of this study was to assess central, spinal and peripheral analgesia in rodents after repeated oral administration of CT-025.
- Adult, male Sprague-Dawley (Charles River, Raleigh, NC) rats weighingl 75-200 g were used in a study of central and spinal analgesia. There were a total of 23 animals in the study and food and water were provided ad libitum. Animals were housed in individual cages and maintained in a room with a 12:12 L:D cycle (lights on at 6:00 a.m.). Prior to dosing, rats were weighed and distributed throughout the dosing groups by body weight so that each dosing group weighed approximately the same. Two groups (10 animals per group) received oral CT-025 and drag vehicle.
- mice received morphine sulfate (2.5 mg/kg; i.m.).
- Male CF-1 (-30 g) mice were used for the peripheral analgesia study. Animals were housed in cages (5/cage) and kept in a room with a 12:12 L:D cycle (lights on at 6:00 a.m.). Food and water were provided ad libitum. A total of 45 mice were used in the study. Rats received CT-025 by oral administration at a dose of 100 ⁇ g/kg twice a day (morning and afternoon) for 9 days (excluding weekends) at a dosing volume of 2.0 mL/kg. The total dose animals that received was 1800 ⁇ g/kg.
- Compositions of rat and mouse dosing formulations are listed in Tables 6 and 7.
- a control group (3 rats) received a single intramuscular (i.m.) injection of morphine sulfate in saline at a dose of 2.5 mg/kg on the last day of the study.
- mice were given either oral CT-025 or drug vehicle using a gavaging needle (Popper gavage needle #20) using a dosing volume of 10.0 mL/kg twice a day (morning and afternoon) for 10 days (except weekend).
- the daily dose of oral CT-025 was 40 ⁇ g/kg.
- the total dose that animals received during the experiment was 400 ⁇ g/kg.
- Central and spinal analgesia were evaluated by measuring latencies in the rat paw-hot plate and rat tail-flick assays, respectively. Peripheral analgesia was evaluated using a mouse acetic acid writhing test.
- Central and Spinal Analgesia Central and Spinal Analgesia:
- Rats were placed in a cylindrical restrainer to measure tail flick latency with the tail extended through an opening in the restrainer.
- the ventral surface of the tail (4-5 cm from the tip) was placed on the testing apparatus measuring surface over a 0.5-cm diameter hole, beneath which a halogen projector bulb was fixed.
- Bulb intensity was calibrated to produce a baseline latency of approximately 3-4 seconds.
- the light beam was automatically cancelled at 8 seconds to prevent damage to the tail.
- Prior to tail flick data collection all rats were habituated to animal restrainers in three daily sessions and then tested every other day. On each test day, the rats were given oral doses of either drug vehicle or CT-025 and, 4 hours later, tested sequentially in two behavioral tests using the following order: tail flick and hot-plate assay.
- Hot-plate test was conducted secondly to avoid potentially confounding stress-induced effects. Formal data collection was performed for 9 days. Hot-plate latency was measured by placing the rat onto the testing apparatus surface which was heated to 52 °C. Latency to the heating stimulus was determined to be the time from placement until the animal either jumped or licked a hindpaw.
- mice received intraperitoneal injections of a 2%> acetic acid solution to produce the typical writhing reaction characterized by a wave of contraction of the abdominal musculature, followed by extension of the hindlimbs.
- the writhing test was conducted at the start of the experiment (baseline) and after 5 and 10 days of treatment. On each day of the writhing test, mice were given oral doses of either drag vehicle or CT-025 at 90 minutes prior to acetic acid administration. After acetic acid administration, mice were placed in individual transparent containers and, five minutes later, the number of writhes was counted during a 10-minute period. Each mouse was used only once during the study. Means and standard errors were calculated to determine inter-animal variability using
- Figure 19 illustrates latency results obtained after oral administration of CT-025 and drag vehicle in the tail flick and hot-plate assays. Average baseline latencies of drug vehicle and CT-025 groups in tail flick and hot-plate tests were ⁇ 3.7 and 1.1 s, respectively. Repeated oral administration of CT-025 did not elicit analgesic responses using either the tail flick or hot-plate assays. The latencies produced after oral administration of CT-025 were similar to those affected by the drug vehicle group for both tests and remained near baseline values.
- Figure 20 illustrates the number of stretches as a measure of the analgesic response during the acetic acid writhing test.
- the numbers of stretches elicited by acetic acid injections in the drag vehicle group at the beginning of the study, and after 1 and 2 weeks of treatment were 31.0 ⁇ 1.2, 31.0 ⁇ 1 J, and 30.0 ⁇ 1.4, respectively.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Rheumatology (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Epidemiology (AREA)
- Pain & Pain Management (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Endocrinology (AREA)
- Hematology (AREA)
- Zoology (AREA)
- Gastroenterology & Hepatology (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Psychiatry (AREA)
- Obesity (AREA)
- Diabetes (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Medicinal Preparation (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP04753588A EP1643959A4 (en) | 2003-06-24 | 2004-05-27 | Mixtures of calcitonin drug-oligomer conjugates and methods of use in pain treatment |
| US10/562,478 US20090281023A9 (en) | 2001-06-04 | 2004-05-27 | Mixtures Of Calcitonin Drug-Oligomer Conjugates And Methods Of Use In Pain Treatment |
| JP2006517158A JP4829783B2 (en) | 2003-06-24 | 2004-05-27 | Mixtures of calcitonin drug-oligomer conjugates and methods of use in treating pain |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US48213003P | 2003-06-24 | 2003-06-24 | |
| US60/482,130 | 2003-06-24 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2005004792A2 true WO2005004792A2 (en) | 2005-01-20 |
| WO2005004792A3 WO2005004792A3 (en) | 2005-09-01 |
Family
ID=34061937
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2004/016784 WO2005004792A2 (en) | 2001-06-04 | 2004-05-27 | Mixtures of calcitonin drug-oligomer conjugates and methods of use in pain treatment |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP1643959A4 (en) |
| JP (1) | JP4829783B2 (en) |
| WO (1) | WO2005004792A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9308263B2 (en) | 2011-10-21 | 2016-04-12 | Seachaid Pharmaceuticals, Inc. | Pharmaceutical compositions and uses thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002098451A1 (en) | 2001-06-04 | 2002-12-12 | Nobex Corporation | Mixtures of calcitonin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IE912365A1 (en) * | 1990-07-23 | 1992-01-29 | Zeneca Ltd | Continuous release pharmaceutical compositions |
| US6495514B1 (en) * | 1998-01-21 | 2002-12-17 | Mercer University | Method for reducing inflammation and inducing an analgesic effect and compounds thereof |
| US6703381B1 (en) * | 1998-08-14 | 2004-03-09 | Nobex Corporation | Methods for delivery therapeutic compounds across the blood-brain barrier |
| KR100345214B1 (en) * | 1999-08-17 | 2002-07-25 | 이강춘 | The nasal transmucosal delivery of peptides conjugated with biocompatible polymers |
| US6858580B2 (en) * | 2001-06-04 | 2005-02-22 | Nobex Corporation | Mixtures of drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same |
-
2004
- 2004-05-27 WO PCT/US2004/016784 patent/WO2005004792A2/en active Application Filing
- 2004-05-27 JP JP2006517158A patent/JP4829783B2/en not_active Expired - Fee Related
- 2004-05-27 EP EP04753588A patent/EP1643959A4/en not_active Withdrawn
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002098451A1 (en) | 2001-06-04 | 2002-12-12 | Nobex Corporation | Mixtures of calcitonin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same |
Non-Patent Citations (3)
| Title |
|---|
| ANTONIO QUATRARO: "Calcitonin in Painful Diabetic Neuropathy The Lancet", LANCET LIMITED, vol. 339, no. 8795, 1 March 1992 (1992-03-01), pages 746 - 747 |
| C. SIMANSKI; DER CHIRURG ET AL.: "Therapy of phantom pain with salmon calcitonin and effect on postoperative patient satisfaction", ZEITSCHRIFT FUR ALLE GEBIETE DER OPERATIVEN MEDIZEN, vol. 70, no. 6, June 1999 (1999-06-01), pages 674 - 681 |
| See also references of EP1643959A4 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9308263B2 (en) | 2011-10-21 | 2016-04-12 | Seachaid Pharmaceuticals, Inc. | Pharmaceutical compositions and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| JP4829783B2 (en) | 2011-12-07 |
| WO2005004792A3 (en) | 2005-09-01 |
| EP1643959A2 (en) | 2006-04-12 |
| EP1643959A4 (en) | 2010-06-09 |
| JP2007521268A (en) | 2007-08-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6713452B2 (en) | Mixtures of calcitonin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same | |
| US6828297B2 (en) | Mixtures of insulin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same | |
| US6828305B2 (en) | Mixtures of growth hormone drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same | |
| AU2002310291A1 (en) | Mixtures of insulin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same | |
| US8030269B2 (en) | Calcitonin drug-oligomer conjugates, and uses thereof | |
| EP1643959A2 (en) | Mixtures of calcitonin drug-oligomer conjugates and methods of use in pain treatment | |
| US20090281023A9 (en) | Mixtures Of Calcitonin Drug-Oligomer Conjugates And Methods Of Use In Pain Treatment | |
| AU2002303961A1 (en) | Mixtures of calcitonin drug-oligomer conjugates comprising polyalkylene glycol, uses thereof, and methods of making same | |
| AU2002310278A1 (en) | Mixtures of growth hormone drug-oligomer conjugates compromising polyalkylene glycol, uses thereof, and methods of making same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ NA SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 3479/CHENP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006517158 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004753588 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004753588 Country of ref document: EP |
|
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 10562478 Country of ref document: US Ref document number: 2007213262 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10562478 Country of ref document: US |