WO2004105722A1 - A formulation for chemical peeling - Google Patents
A formulation for chemical peeling Download PDFInfo
- Publication number
- WO2004105722A1 WO2004105722A1 PCT/IT2003/000339 IT0300339W WO2004105722A1 WO 2004105722 A1 WO2004105722 A1 WO 2004105722A1 IT 0300339 W IT0300339 W IT 0300339W WO 2004105722 A1 WO2004105722 A1 WO 2004105722A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- keratolytic agent
- acid
- composition according
- keratolytic
- agent
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/347—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/361—Carboxylic acids having more than seven carbon atoms in an unbroken chain; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/362—Polycarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/365—Hydroxycarboxylic acids; Ketocarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/368—Carboxylic acids; Salts or anhydrides thereof with carboxyl groups directly bound to carbon atoms of aromatic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/46—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing sulfur
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/494—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom
- A61K8/4953—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with more than one nitrogen as the only hetero atom containing pyrimidine ring derivatives, e.g. minoxidil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/49—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds
- A61K8/4973—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing heterocyclic compounds with oxygen as the only hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/60—Sugars; Derivatives thereof
- A61K8/602—Glycosides, e.g. rutin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/12—Keratolytics, e.g. wart or anti-corn preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/20—Chemical, physico-chemical or functional or structural properties of the composition as a whole
- A61K2800/28—Rubbing or scrubbing compositions; Peeling or abrasive compositions; Containing exfoliants
Definitions
- the present invention relates to a new formulation able to increase the efficacy and the tolerability of preparations based on the use of compounds used to carry out chemical peeling.
- a very superficial peeling accelerates the natural exfoliation of the corneous layer, whilst a peeling which acts at a much deeper level causes necrosis and inflammation of the epidermis, the papillary dermis or of the reticular dermis.
- Chemical peeling is particularly recommended in the following cases: a) Keratosis and cutaneous ageing b) Dischromia c) Post - acne scarring d) Common acne and rosacea e) Radiodermatitis f) Stretch marks g) Seborrheic dermatitis
- the various types of chemical peeling can be classified thus: - Very superficial peeling: this type of peeling only removes the superficial corneous layer; - Superficial peeling: this type of peeling causes necrosis of a part or in all of the epidermal layer reaching the basal layer of the epidermis; - Average depth peeling: this type of peeling causes necrosis of the epidermis and part of the papillary dermis;
- this type of peeling causes necrosis of the epidermis, the papillary dermis and can extend to the reticular dermis.
- the depth of peeling depends on numerous factors, such as: i) the type of substance used; ii) the concentration of the substance used, iii) the number of steps with the chosen substance on the same part of the skin, iv) the application technique, v) the preparation of the skin in the pre-treatment phase, vi) the type of cutaneous treatment in the period preceding the peeling, vii) the patients skin type. viii) the area of the cuteous treated, and ix) the exposure time to the selected chemical agent on the skin.
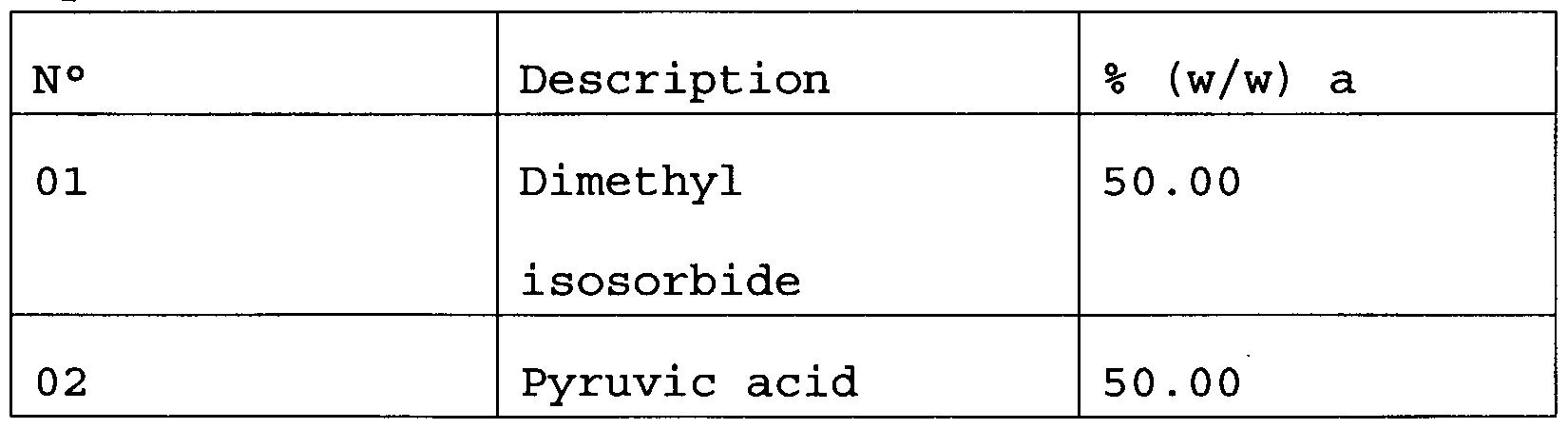
- the present invention refers more specifically to a formulation for chemical peeling comprising a keratolytic agent in combination with dimethyl isosorbide.
- the dimethyl isosorbide will be present in such a quantity as to obtain an increase in the absorption kinetics of said keratolytic agent, if compared with the use of the keratolytic agent by itself.
- the increase in absorption kinetics is evaluated as shown below, i.e. by determining the quantity of keratolytic agent able to permeate through an SCE membrane by HPLC.
- Preferred keratolytic compounds are selected from the chemical group of saturated and unsaturated monocarboxylic acids, saturated and unsaturated bicarboxylic acids, tricarboxylic acids, alpha hydroxyacids and beta hydroxyacids of monocarboxylic acids, alpha hydroxyacids and beta hydroxyacids of bicarboxylic acids, alpha hydroxyacids and beta hydroxyacids of tricarboxylic acids, ketoacids, alpha ketoacids, beta ketoacids, of the polycarboxylic acids, of the polyhydroxy monocarboxylic acids, of the polyhydroxy bicarboxylic acids, of the polyhydroxy tricarboxylic acids.
- keratolytic agents are selected from the group comprising glycolic acid, tartaric acid, salicylic acid, citric acid, lactic acid, pyruvic acid, gluconic acid, glucuron ⁇ c acid, malic acid, oxalic acid, malonic acid, succinic acid, acetic acid, phenol, resorcine, retinoic acid, adapalene, trichloroacetic acid, 5-fluoro uracil, azelaic acid.
- Keratolytic agents comprised within the scope of the present invention are also the salts, esters, possible cis or trans forms, racemic mixtures and/or the relative dextrorotatory or levorotatory forms of the above listed compounds.
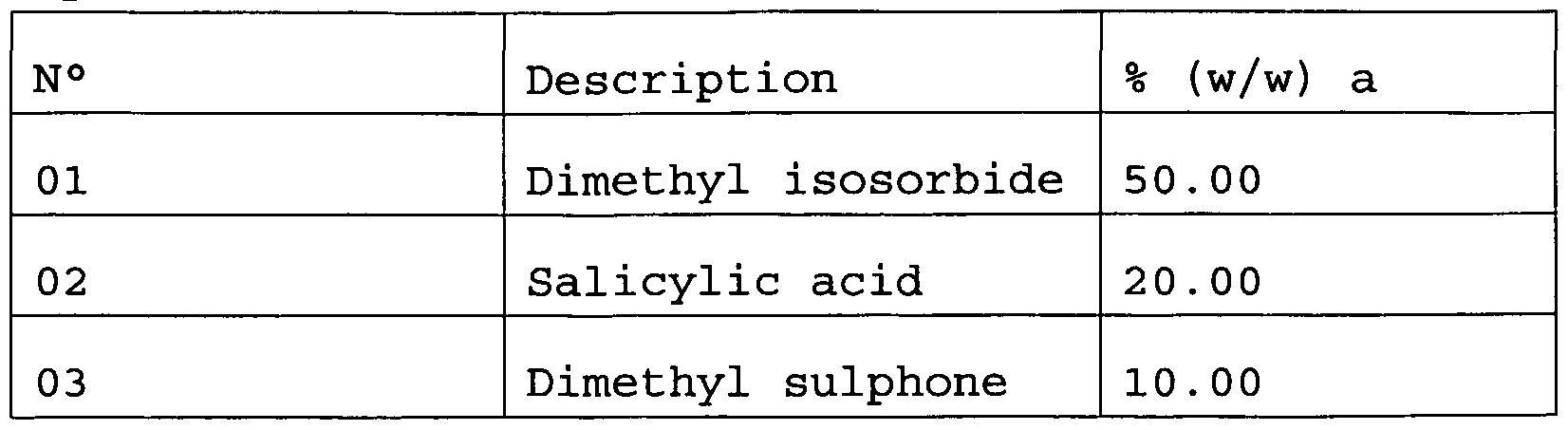
- the pharmaceutical and/or cosmetic composition of dimethyl isosorbide with one or more keratolytic agents comprises additionally dimethyl sulphone .
- the dimethyl sulphone combined with keratolytic agents is capable of reducing the erythema induced by the agents themselves.
- the reduction of inflammation, irritation and erythema is obtained through the combination of the activity of the dimethyl sulphone with the fact that the quantity of keratolytic agent used to obtain the "peeling" effect is reduced thanks to the action of the dimethyl isosorbide.
- This latter component increases the kinetics of percutaneous absorption of the keratolytic agent, rendering it more available for the action intended.
- a third particularly preferred embodiment is that in which the keratolytic agent and/or the mixed keratolytic agents, combined with dimethyl isosorbide and dimethyl sulphone, are associated with the ester of an acid with keratolytic activity.
- the dimethyl isosorbide and the keratolytic agent and/or a mixture of keratolytic agents can each be contained in the composition in a quantity by weight of from 1 to 99%, preferably each in a quantity comprised of between 5 and 40%. More preferably, dimethyl isosorbide and the keratolytic agents will be present in the composition in weight ratios comprised of between 1 : 4 and 4 : 1.
- the dimethyl isosorbide and the keratolytic agent and/or a mixture of keratolytic agents, associated with dimethyl sulphone can be contained in the compositions in a quantity by weight of from 1 to 99% each, preferably in a quantity comprised of between 5 and 70%.
- dimethyl isosorbide and the keratolytic agent will be present in the composition in a ratio comprised of between 1 : 4 and 4 : 1.
- the dimethyl sulphone will preferably be present in a quantity comprised of between 2% and 70% by weight, more preferably between 10% and 65%, with respect to the keratolytic agent.
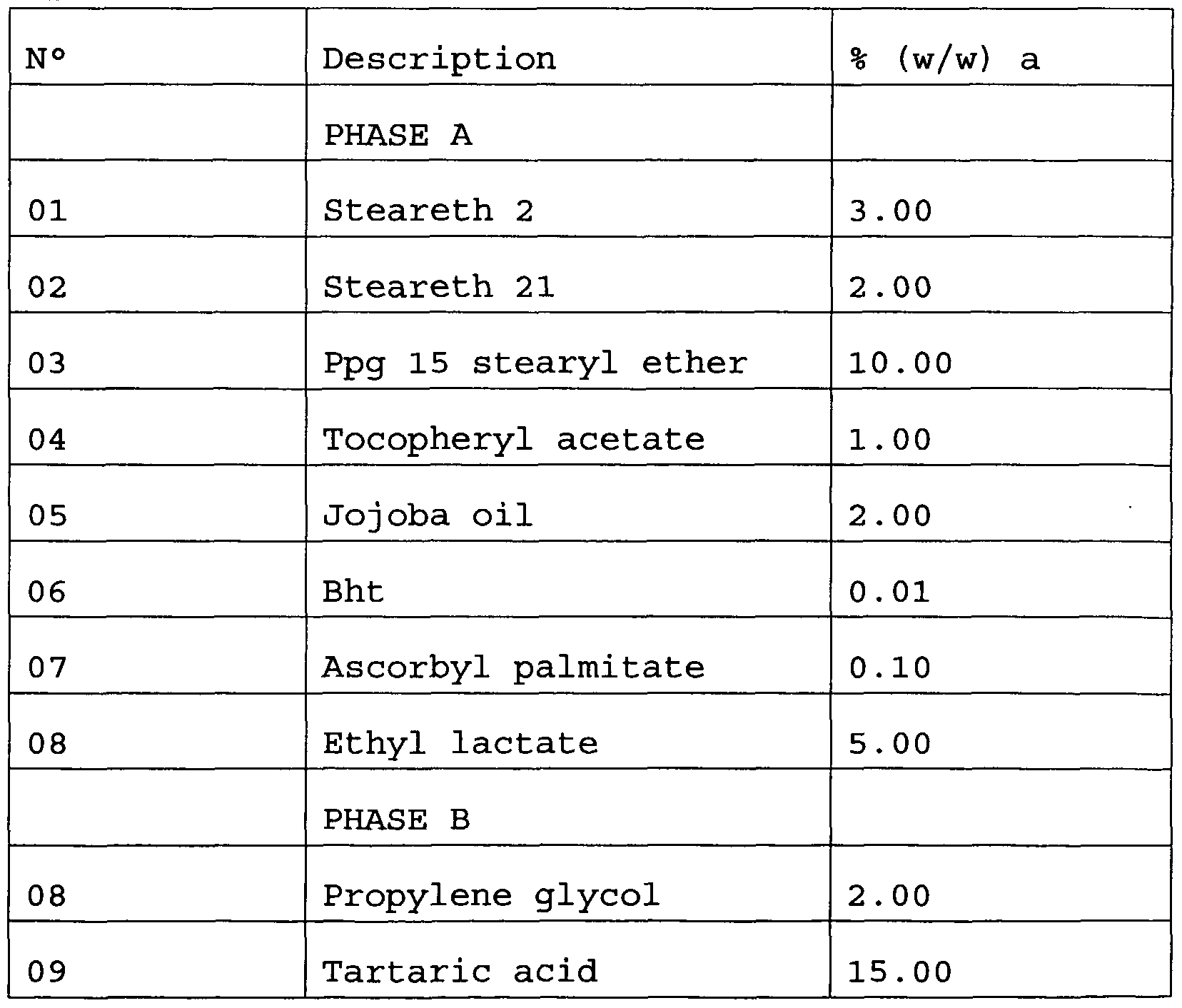
- solvents such as water (in particular demineralised water) , alcohols (such as ethyl alcohol) or glycols
- excipients for example, ethylene glycol or propylene glycol
- excipients such as emulsifiers, antioxidants, lipid excipients, sequestrants, preservatives.
- excipients used in particular for the preparation of emulsions, gels, creams, ointments, etc., are widely known to the expert in the field and will therefore not be described in any further detail .
- Experiments relating to the evaluation of the favourable effect on percutaneous absorption of the keratolytic agent in the presence of dimethyl isosorbide is reported in corroboration of the present invention.
- the aim of these experiments has been that of evaluating the in vi tro percutaneous absorption across isolated human skin, of glycolic acid comprised in a formulation in which the keratolytic agent has been dissolved in water and propylene glycol (solution GCl) and another in which the keratolytic agent has been vehicularised using dimethyl isosorbide (solution GC2) .
- the experiment has been carried out using a system of Franz cells with a corneous-epidermis membrane layer (SCE membrane) , the experimental protocol of which has already been widely described in the literature.
- SCE membrane corneous-epidermis membrane layer
- SCE corneous-epidermis membrane layer
- the SCE membranes thus prepared, have been dried and then placed in an appropriate desiccator. These membranes have then been conserved in sheets of aluminium at a temperature of around 4°C and rehydrated at the time of use, by immersion in distilled water, one hour prior to the start of the permeation experiments.
- the coefficient of permeability (Kp) of tritiated water Prior to proceeding to the cutaneous permeation experiments and with the aim of evaluating the integrity of the SCE membranes used, the coefficient of permeability (Kp) of tritiated water has been determined for each sample of SCE membranes, the value of which is a sufficiently indicative parameter of the integrity of said membranes .
- the "receptor" compartment stirred and thermostated at a temperature of 35-36 °C, has been fed with an aqueous saline solution of 0.9% (w/v) NaCl .
- 200 mg/cm 2 of each formulation GCl and GC2 , containing the glycolic acid have been initially deposited onto each SCE membrane.
- the monitoring of the permeation process has then been carried out by determining, by a suitable HPLC method, the quantity of glycolic acid that has permeated through the corneous-epidermis (SCE) membrane layer over the 24 hour period following the application of the product into the "donor” .
- SCE corneous-epidermis
- Tab. 1 The quantity of glycolic acid (expressed in ⁇ g/cm 2 ) permeated from the formulations GCl and GC2 across the human skin (SCE) membranes originating from six different subjects (A-F) in 24 hours.
- the mixture of dimethyl isosorbide, associated with a keratolytic agent and/or a mixture of keratolytic agents, together with dimethyl sulphone can be combined with esters of the keratolytic agents, preferably the ethyl esters.
- esters of the keratolytic agents preferably the ethyl esters.
- esters of the keratolytic agents are ethyl pyruvate, ethyl glycolate, triethyl citrate, ethyl resorcinate, the ethyl ester of retinoic acid, ethyl salicylate, methyl salicylate, ethyl malnate, ethyl acetate, ethyl tartrate.
- a further subject of the present invention is a formulation for chemical peeling comprising of one or more keratolytic agents, preferably selected from the above described group, together with a keratolytic agent ester.
- the ester of the keratolytic agent will preferably be selected from the above listed group and can be the ester of the same keratolytic agent used in acid form or the ester of a different keratolytic agent.
- Such keratolytic agent esters will be present in the composition in a quantity preferably comprised of between 3% and 60% by weight, more preferably in a quantity comprised of between 15% and 50% by weight, with respect to the keratolytic agent (or to the mixture of keratolytic agents) .
- keratolytic agent esters In place of the keratolytic agent esters, another derivative or pro-drug can however be used, in the same proportions indicated above, which is capable of liberating after administration under biological conditions, the keratolytic agent in the treatment site. It is evident that the combination of one or more keratolytic agents and their derivative or pro-drug as defined above can also be applied to compositions in which the dimethyl isosorbide or the dimethyl sulphone are not present. One will in fact obtain, in any case, the desired effect of achieving a more prolonged over time and at the same time less acute chemical peeling, with the consequent reduction of the irritant phenomena caused by intense and acute treatment .
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Dermatology (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Cosmetics (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Detergent Compositions (AREA)
- Paints Or Removers (AREA)
Abstract
Description
Claims
Priority Applications (14)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/557,923 US20070010580A1 (en) | 2003-05-30 | 2003-05-30 | Formulation for chemical peeling |
| CN038265567A CN1771021B (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical skin-changing |
| TR2018/07182T TR201807182T4 (en) | 2003-05-30 | 2003-05-30 | The use of dimethyl sulfone in formulations for chemical skin / facial peels. |
| EP10181232.9A EP2269615B1 (en) | 2003-05-30 | 2003-05-30 | Use of dimethyl sulphone in formulations for chemical peeling |
| JP2005500189A JP4260163B2 (en) | 2003-05-30 | 2003-05-30 | Formula for chemical peeling |
| AU2003238685A AU2003238685B2 (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
| PCT/IT2003/000339 WO2004105722A1 (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
| CA2525691A CA2525691C (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
| ES10181232.9T ES2672202T3 (en) | 2003-05-30 | 2003-05-30 | Use of dimethyl sulfone in chemical release formulations |
| EP03733016A EP1631248A1 (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
| TW093114247A TWI333854B (en) | 2003-05-30 | 2004-05-20 | A formulation for chemical peeling |
| HRP20050972AA HRP20050972B1 (en) | 2003-05-30 | 2005-11-29 | A formulation for chemical peeling |
| US13/089,826 US20120094965A1 (en) | 2003-05-30 | 2011-04-19 | Method and Formulation for Chemical Peeling |
| US14/188,496 US20140221474A1 (en) | 2003-05-30 | 2014-02-24 | Method and Formulation for Chemical Peeling |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/IT2003/000339 WO2004105722A1 (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/557,923 A-371-Of-International US20070010580A1 (en) | 2003-05-30 | 2003-05-30 | Formulation for chemical peeling |
| US13/089,826 Continuation-In-Part US20120094965A1 (en) | 2003-05-30 | 2011-04-19 | Method and Formulation for Chemical Peeling |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2004105722A1 true WO2004105722A1 (en) | 2004-12-09 |
| WO2004105722A8 WO2004105722A8 (en) | 2005-02-10 |
Family
ID=33485484
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IT2003/000339 WO2004105722A1 (en) | 2003-05-30 | 2003-05-30 | A formulation for chemical peeling |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US20070010580A1 (en) |
| EP (2) | EP1631248A1 (en) |
| JP (1) | JP4260163B2 (en) |
| CN (1) | CN1771021B (en) |
| AU (1) | AU2003238685B2 (en) |
| CA (1) | CA2525691C (en) |
| ES (1) | ES2672202T3 (en) |
| HR (1) | HRP20050972B1 (en) |
| TR (1) | TR201807182T4 (en) |
| TW (1) | TWI333854B (en) |
| WO (1) | WO2004105722A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006120342A2 (en) * | 2005-05-12 | 2006-11-16 | Roquette Freres | Method for the treatment of a material other than the human body |
| EP1867318A1 (en) * | 2006-06-13 | 2007-12-19 | Wella Aktiengesellschaft | Body Care composition with water-soluble abrasive particles |
| EP2269615A1 (en) * | 2003-05-30 | 2011-01-05 | Gianfranco De Paoli Ambrosi | Use of dimethyl sulphone in formulations for chemical peeling |
| GB2507639A (en) * | 2012-09-10 | 2014-05-07 | Ad Lunam Labs Inc | Pharmaceutical serum comprising an alkyl lactate and Simmondsia chinensis seed oil |
| EP2537512A3 (en) * | 2011-04-19 | 2015-12-02 | Gianfranco De Paoli Ambrosi | Method and formulation for chemical peeling |
Families Citing this family (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL152486A0 (en) | 2002-10-25 | 2003-05-29 | Meir Eini | Alcohol-free cosmetic and pharmaceutical foam carrier |
| US7704518B2 (en) | 2003-08-04 | 2010-04-27 | Foamix, Ltd. | Foamable vehicle and pharmaceutical compositions thereof |
| US20080138296A1 (en) * | 2002-10-25 | 2008-06-12 | Foamix Ltd. | Foam prepared from nanoemulsions and uses |
| US7700076B2 (en) | 2002-10-25 | 2010-04-20 | Foamix, Ltd. | Penetrating pharmaceutical foam |
| US20050205086A1 (en) * | 2002-10-25 | 2005-09-22 | Foamix Ltd. | Retinoid immunomodulating kit and composition and uses thereof |
| US20070292359A1 (en) * | 2002-10-25 | 2007-12-20 | Foamix Ltd. | Polypropylene glycol foamable vehicle and pharmaceutical compositions thereof |
| US20050069566A1 (en) * | 2003-08-04 | 2005-03-31 | Foamix Ltd. | Foam carrier containing amphiphilic copolymeric gelling agent |
| US9211259B2 (en) | 2002-11-29 | 2015-12-15 | Foamix Pharmaceuticals Ltd. | Antibiotic kit and composition and uses thereof |
| US8900554B2 (en) | 2002-10-25 | 2014-12-02 | Foamix Pharmaceuticals Ltd. | Foamable composition and uses thereof |
| US10117812B2 (en) * | 2002-10-25 | 2018-11-06 | Foamix Pharmaceuticals Ltd. | Foamable composition combining a polar solvent and a hydrophobic carrier |
| US20060233721A1 (en) * | 2002-10-25 | 2006-10-19 | Foamix Ltd. | Foam containing unique oil globules |
| US20060018937A1 (en) * | 2002-10-25 | 2006-01-26 | Foamix Ltd. | Steroid kit and foamable composition and uses thereof |
| US7820145B2 (en) | 2003-08-04 | 2010-10-26 | Foamix Ltd. | Oleaginous pharmaceutical and cosmetic foam |
| US20050271596A1 (en) * | 2002-10-25 | 2005-12-08 | Foamix Ltd. | Vasoactive kit and composition and uses thereof |
| US9265725B2 (en) * | 2002-10-25 | 2016-02-23 | Foamix Pharmaceuticals Ltd. | Dicarboxylic acid foamable vehicle and pharmaceutical compositions thereof |
| US9668972B2 (en) * | 2002-10-25 | 2017-06-06 | Foamix Pharmaceuticals Ltd. | Nonsteroidal immunomodulating kit and composition and uses thereof |
| MXPA05004278A (en) | 2002-10-25 | 2005-10-05 | Foamix Ltd | Cosmetic and pharmaceutical foam. |
| US20080031907A1 (en) * | 2002-10-25 | 2008-02-07 | Foamix Ltd. | Cosmetic and pharmaceutical foam |
| US20120094965A1 (en) * | 2003-05-30 | 2012-04-19 | Gianfranco De Paoli Ambrosi | Method and Formulation for Chemical Peeling |
| US8795693B2 (en) | 2003-08-04 | 2014-08-05 | Foamix Ltd. | Compositions with modulating agents |
| JP2005310310A (en) * | 2004-04-23 | 2005-11-04 | Sanyo Electric Co Ltd | Tracking balance adjustment device |
| JP4565639B2 (en) * | 2005-04-04 | 2010-10-20 | 株式会社アンプリー | Peeling agent composition |
| US20080152596A1 (en) * | 2005-07-19 | 2008-06-26 | Foamix Ltd. | Polypropylene glycol foamable vehicle and pharmaceutical compositions thereof |
| US7709014B2 (en) * | 2005-10-17 | 2010-05-04 | Yu Ruey J | Hydroxy-oligocarboxylic esters: effects on nerve and use for cutaneous and mucocutaneous organs or sites |
| MX2009002536A (en) * | 2006-09-08 | 2009-04-14 | Foamix Ltd | Colored or colorable foamable composition and foam. |
| US20080260655A1 (en) * | 2006-11-14 | 2008-10-23 | Dov Tamarkin | Substantially non-aqueous foamable petrolatum based pharmaceutical and cosmetic compositions and their uses |
| US20080292560A1 (en) * | 2007-01-12 | 2008-11-27 | Dov Tamarkin | Silicone in glycol pharmaceutical and cosmetic compositions with accommodating agent |
| FR2918876B1 (en) * | 2007-07-16 | 2012-10-05 | Oreal | USE OF GREEN LIGHT TO ACTIVATE L-AMINO ACID OXIDASE |
| US8636982B2 (en) | 2007-08-07 | 2014-01-28 | Foamix Ltd. | Wax foamable vehicle and pharmaceutical compositions thereof |
| WO2009069006A2 (en) * | 2007-11-30 | 2009-06-04 | Foamix Ltd. | Foam containing benzoyl peroxide |
| WO2009072007A2 (en) | 2007-12-07 | 2009-06-11 | Foamix Ltd. | Carriers, formulations, methods for formulating unstable active agents for external application and uses thereof |
| FR2927800A1 (en) * | 2008-02-25 | 2009-08-28 | Oreal | COMBINATION OF LUMINOUS RADIATION AND A BIOCONVERTIBLE COMPOUND BY LIPASE TO IMPROVE THE APPEARANCE OF THE SKIN AND / OR HAIR. |
| FR2941377B1 (en) * | 2009-01-29 | 2013-02-15 | Soliance | COSMETIC COMPOSITIONS COMPRISING CETOGLUCONIC ACID DERIVATIVES |
| US20120087872A1 (en) | 2009-04-28 | 2012-04-12 | Foamix Ltd. | Foamable Vehicles and Pharmaceutical Compositions Comprising Aprotic Polar Solvents and Uses Thereof |
| CA2769677A1 (en) | 2009-07-29 | 2011-02-03 | Foamix Ltd. | Non surface active agent non polymeric agent hydro-alcoholic foamable compositions, breakable foams and their uses |
| WO2011013009A2 (en) | 2009-07-29 | 2011-02-03 | Foamix Ltd. | Non surfactant hydro-alcoholic foamable compositions, breakable foams and their uses |
| NL2003419C2 (en) * | 2009-09-01 | 2011-03-02 | Shieldmark Zacco | Composition for tropical application, uses thereof, applicator device and kit of parts. |
| US9849142B2 (en) | 2009-10-02 | 2017-12-26 | Foamix Pharmaceuticals Ltd. | Methods for accelerated return of skin integrity and for the treatment of impetigo |
| BR112012007473A2 (en) | 2009-10-02 | 2019-05-07 | Foamix Ltd | tetracycline topical compositions and methods of use |
| CA2920859A1 (en) * | 2013-08-09 | 2015-02-12 | The Chemours Company Fc, Llc | Skin care compositions having cyclic diesters and methods thereof |
| US10398641B2 (en) | 2016-09-08 | 2019-09-03 | Foamix Pharmaceuticals Ltd. | Compositions and methods for treating rosacea and acne |
| GEP20227384B (en) * | 2018-04-09 | 2022-05-25 | Noon Aesthetics M R Ltd | Topical formulations comprising strontium and methylsulfonylmethane (msm) and methods of treatment |
| CN116492240B (en) * | 2023-06-30 | 2023-09-19 | 北京大学第三医院(北京大学第三临床医学院) | Compound chemical stripping skin-changing reagent containing baratinib and application thereof |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997009027A1 (en) * | 1995-09-01 | 1997-03-13 | Isp Investments Inc. | Cosmetic composition of an alpha or beta-hydroxy acid and a polyvinylpyrrolidone complexing agent |
| WO2002022103A1 (en) * | 2000-09-13 | 2002-03-21 | The Procter & Gamble Company | Cosmetic compositions |
| WO2002038121A1 (en) * | 2000-11-09 | 2002-05-16 | Henkel Kommanditgesellschaft Auf Aktien | Cosmetic skin treatment, care or protection composition |
| WO2003002674A1 (en) * | 2001-06-27 | 2003-01-09 | Pennzoil-Quaker State Company | Coating composition |
| EP1304101A1 (en) * | 2001-10-19 | 2003-04-23 | Torrent Pharmaceuticals Ltd | Composition and method for use of pyridinium derivatives in cosmetic and therapeutic applications |
| US20030194385A1 (en) * | 2002-01-29 | 2003-10-16 | Lila Gruber Research Foundation | Pharmaceutical formulations containing solubilized hydroquinone, salicylic acid and hydrocortisone for the treatment of melasma and related dermatological problems |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2452119A1 (en) * | 1974-11-02 | 1976-05-13 | Henkel & Cie Gmbh | Cosmetic preparations against acne - contg. neutral esters of hydroxycarboxylic acids and alcohols |
| US4082881A (en) * | 1976-12-23 | 1978-04-04 | E. R. Squibb & Sons, Inc. | Topical and other type pharmaceutical formulations containing isosorbide carrier |
| US5166176A (en) * | 1986-12-29 | 1992-11-24 | Obagi Zein E | Composition for healing damaged skin |
| GB9218701D0 (en) * | 1992-09-04 | 1992-10-21 | Salim Aws S M | Housewife dermatitis treatment |
| US5824326A (en) * | 1997-06-27 | 1998-10-20 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Activity enhancement of ferulic acid with dimethyl isosorbride in cosmetic compositions |
| US7074747B1 (en) * | 1999-07-01 | 2006-07-11 | Johnson & Johnson Consumer Companies, Inc. | Cleansing compositions |
| FR2813528B1 (en) * | 2000-09-07 | 2004-02-27 | Oreal | COSMETIC COMPOSITION, AND ITS USE AS A SKIN CLEANSING MASK |
| AU2001224380A1 (en) * | 2000-12-18 | 2002-07-01 | The Procter And Gamble Company | Wet wipe |
| AU2003238687B8 (en) * | 2003-05-30 | 2009-08-06 | Gianfranco De Paoli Ambrosi | Cosmetic and/or pharmaceutical compositions comprising dimthylsulphone for the cure and prevention of irritation, inflammation and cutaneous erythema |
| TR201807182T4 (en) * | 2003-05-30 | 2018-06-21 | De Paoli Ambrosi Gianfranco | The use of dimethyl sulfone in formulations for chemical skin / facial peels. |
-
2003
- 2003-05-30 TR TR2018/07182T patent/TR201807182T4/en unknown
- 2003-05-30 ES ES10181232.9T patent/ES2672202T3/en not_active Expired - Lifetime
- 2003-05-30 WO PCT/IT2003/000339 patent/WO2004105722A1/en active Application Filing
- 2003-05-30 AU AU2003238685A patent/AU2003238685B2/en not_active Ceased
- 2003-05-30 US US10/557,923 patent/US20070010580A1/en not_active Abandoned
- 2003-05-30 EP EP03733016A patent/EP1631248A1/en not_active Ceased
- 2003-05-30 CN CN038265567A patent/CN1771021B/en not_active Expired - Fee Related
- 2003-05-30 CA CA2525691A patent/CA2525691C/en not_active Expired - Fee Related
- 2003-05-30 EP EP10181232.9A patent/EP2269615B1/en not_active Expired - Lifetime
- 2003-05-30 JP JP2005500189A patent/JP4260163B2/en not_active Expired - Fee Related
-
2004
- 2004-05-20 TW TW093114247A patent/TWI333854B/en not_active IP Right Cessation
-
2005
- 2005-11-29 HR HRP20050972AA patent/HRP20050972B1/en not_active IP Right Cessation
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997009027A1 (en) * | 1995-09-01 | 1997-03-13 | Isp Investments Inc. | Cosmetic composition of an alpha or beta-hydroxy acid and a polyvinylpyrrolidone complexing agent |
| WO2002022103A1 (en) * | 2000-09-13 | 2002-03-21 | The Procter & Gamble Company | Cosmetic compositions |
| WO2002038121A1 (en) * | 2000-11-09 | 2002-05-16 | Henkel Kommanditgesellschaft Auf Aktien | Cosmetic skin treatment, care or protection composition |
| WO2003002674A1 (en) * | 2001-06-27 | 2003-01-09 | Pennzoil-Quaker State Company | Coating composition |
| EP1304101A1 (en) * | 2001-10-19 | 2003-04-23 | Torrent Pharmaceuticals Ltd | Composition and method for use of pyridinium derivatives in cosmetic and therapeutic applications |
| US20030194385A1 (en) * | 2002-01-29 | 2003-10-16 | Lila Gruber Research Foundation | Pharmaceutical formulations containing solubilized hydroquinone, salicylic acid and hydrocortisone for the treatment of melasma and related dermatological problems |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2269615A1 (en) * | 2003-05-30 | 2011-01-05 | Gianfranco De Paoli Ambrosi | Use of dimethyl sulphone in formulations for chemical peeling |
| EP2003163A1 (en) | 2005-05-12 | 2008-12-17 | Roquette Frˬres | Method for surface treatment of a metal or fibrous material |
| US8080115B2 (en) | 2005-05-12 | 2011-12-20 | Roquette Freres | Method for the surface treatment of a metallic or fibrous material |
| WO2006120342A3 (en) * | 2005-05-12 | 2007-02-01 | Roquette Freres | Method for the treatment of a material other than the human body |
| WO2006120343A3 (en) * | 2005-05-12 | 2007-02-08 | Roquette Freres | Method for the surface treatment of a metallic or fibrous material |
| FR2885536A1 (en) * | 2005-05-12 | 2006-11-17 | Roquette Freres | COMPOSITION BASED ON DIANHYDROHEXITOL ETHERS FOR THE TREATMENT OF MATTER OTHER THAN THE HUMAN BODY |
| WO2006120343A2 (en) * | 2005-05-12 | 2006-11-16 | Roquette Freres | Method for the surface treatment of a metallic or fibrous material |
| WO2006120342A2 (en) * | 2005-05-12 | 2006-11-16 | Roquette Freres | Method for the treatment of a material other than the human body |
| JP2008540097A (en) * | 2005-05-12 | 2008-11-20 | ロケット・フルーレ | Method of processing materials other than human body |
| WO2007144839A3 (en) * | 2006-06-13 | 2008-05-02 | Procter & Gamble | Body care composition with water-soluble abrasive particles |
| WO2007144839A2 (en) * | 2006-06-13 | 2007-12-21 | The Procter & Gamble Company | Body care composition with water-soluble abrasive particles |
| EP1867318A1 (en) * | 2006-06-13 | 2007-12-19 | Wella Aktiengesellschaft | Body Care composition with water-soluble abrasive particles |
| EP2537512A3 (en) * | 2011-04-19 | 2015-12-02 | Gianfranco De Paoli Ambrosi | Method and formulation for chemical peeling |
| GB2507639A (en) * | 2012-09-10 | 2014-05-07 | Ad Lunam Labs Inc | Pharmaceutical serum comprising an alkyl lactate and Simmondsia chinensis seed oil |
| GB2507639B (en) * | 2012-09-10 | 2019-02-13 | Ad Lunam Labs Inc | Antifungal serum comprising alkyl lactate and simmondsia chinesis seed oil |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2672202T3 (en) | 2018-06-13 |
| AU2003238685A1 (en) | 2005-01-21 |
| EP1631248A1 (en) | 2006-03-08 |
| US20070010580A1 (en) | 2007-01-11 |
| JP4260163B2 (en) | 2009-04-30 |
| CN1771021A (en) | 2006-05-10 |
| EP2269615B1 (en) | 2018-03-07 |
| CN1771021B (en) | 2011-11-16 |
| TWI333854B (en) | 2010-12-01 |
| JP2006525950A (en) | 2006-11-16 |
| WO2004105722A8 (en) | 2005-02-10 |
| HRP20050972A2 (en) | 2006-09-30 |
| AU2003238685B2 (en) | 2009-10-08 |
| TW200505411A (en) | 2005-02-16 |
| CA2525691C (en) | 2012-01-24 |
| HRP20050972B1 (en) | 2013-09-30 |
| TR201807182T4 (en) | 2018-06-21 |
| EP2269615A1 (en) | 2011-01-05 |
| CA2525691A1 (en) | 2004-12-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA2525691C (en) | A formulation for chemical peeling | |
| WO2021073620A1 (en) | Use of acellular adipose tissue extract in promoting hair growth and retention | |
| JP2003530330A (en) | Treatment of skin damage using polyenyl phosphatidylcholine | |
| EP0661036A1 (en) | Antiacne composition for the simultaneous treatment of superficial and lower layers of the skin | |
| JP2000504689A (en) | Compositions and methods for performing superficial chemical exfoliation | |
| JP2008533037A (en) | Benzoyl peroxide composition and method of use | |
| JP5013648B2 (en) | Composition for enhancing skin absorption of active ingredient in external preparation for skin | |
| CN115137667A (en) | Cosmetic composition with permeation promoting effect and application thereof | |
| KR20070001045A (en) | Nanoliposome particles containing taxoxyline and Cudrania extract and cosmetic and soap compositions containing them | |
| JP2015518033A (en) | O / W-emulsion type topical pharmaceutical composition containing retinoid | |
| JPH09301880A (en) | Preparation for external use for skin | |
| CN115531226A (en) | Preparation method and whitening application of glycyrrhizic acid-licochalcone A micelle emulsion | |
| JPWO2019171843A1 (en) | Cosmetics or medical materials | |
| RU2313334C2 (en) | Chemical peeling composition | |
| ZA200509283B (en) | A formulation for chemical peeling | |
| EP3548017B1 (en) | Composition comprising glutathion tioester and secoiridoids of the plant olea europea for treating hair loss | |
| FR2686251A1 (en) | COMPOSITION FOR THE HEALING OF A WOUND. | |
| JP2009137939A (en) | Formulation for chemical peeling | |
| JPH1160481A (en) | Iontophoretic liquid for skin preparation for external use | |
| US20140221474A1 (en) | Method and Formulation for Chemical Peeling | |
| JP2008291032A (en) | Prescription for chemical peeling | |
| KR102691624B1 (en) | A cosmetic composition and pharmaceutical composition comprising carnitine, arginine, creatine, and vitamin C as active ingredients | |
| RU2826643C2 (en) | Compositions for odontological and dermatological use, containing trichloroacetate salts and hydroxyacids | |
| FR2646080A1 (en) | COSMETIC COMPOSITION COMPRISING COMPOUNDS OBTAINED BY CULTURE OF CELLS, IN PARTICULAR FIBROBLASTS OR KERATINOCYTES | |
| SG185225A1 (en) | Method and formulation for chemical peeling |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NI NO NZ OM PH PL PT RO RU SC SD SE SG SK SL TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| CFP | Corrected version of a pamphlet front page | ||
| CR1 | Correction of entry in section i |
Free format text: IN PCT GAZETTE 50/2004 UNDER (72) REPLACE "DE PAOLI AMBROSIO, GIANFRANCO" BY "DE PAOLI AMBROSI, GIANFRANCO" |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003733016 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005/09283 Country of ref document: ZA Ref document number: 200509283 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2525691 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2003238685 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: P20050972A Country of ref document: HR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005500189 Country of ref document: JP Ref document number: 20038265567 Country of ref document: CN Ref document number: 3211/CHENP/2005 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005141489 Country of ref document: RU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2003733016 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007010580 Country of ref document: US Ref document number: 10557923 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 10557923 Country of ref document: US |