WO2004081190A2 - Uses of il-23 agonists and antagonists; related reagents - Google Patents
Uses of il-23 agonists and antagonists; related reagents Download PDFInfo
- Publication number
- WO2004081190A2 WO2004081190A2 PCT/US2004/007198 US2004007198W WO2004081190A2 WO 2004081190 A2 WO2004081190 A2 WO 2004081190A2 US 2004007198 W US2004007198 W US 2004007198W WO 2004081190 A2 WO2004081190 A2 WO 2004081190A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer
- tumor
- antibody
- cell
- seq
- Prior art date
Links
- 239000005557 antagonist Substances 0.000 title claims description 39
- 239000000556 agonist Substances 0.000 title claims description 31
- 239000003153 chemical reaction reagent Substances 0.000 title description 15
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 110
- 238000000034 method Methods 0.000 claims abstract description 68
- 108010065637 Interleukin-23 Proteins 0.000 claims description 76
- 102000013264 Interleukin-23 Human genes 0.000 claims description 76
- 210000004027 cell Anatomy 0.000 claims description 62
- 238000009739 binding Methods 0.000 claims description 53
- 230000027455 binding Effects 0.000 claims description 52
- 239000000203 mixture Substances 0.000 claims description 48
- 201000011510 cancer Diseases 0.000 claims description 45
- 150000007523 nucleic acids Chemical class 0.000 claims description 38
- 102100036672 Interleukin-23 receptor Human genes 0.000 claims description 34
- 108020004707 nucleic acids Proteins 0.000 claims description 32
- 102000039446 nucleic acids Human genes 0.000 claims description 32
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 31
- 108040001844 interleukin-23 receptor activity proteins Proteins 0.000 claims description 29
- 206010006895 Cachexia Diseases 0.000 claims description 22
- 239000012634 fragment Substances 0.000 claims description 20
- 229920001184 polypeptide Polymers 0.000 claims description 17
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 17
- 150000003384 small molecules Chemical class 0.000 claims description 16
- 239000000427 antigen Substances 0.000 claims description 14
- 108091007433 antigens Proteins 0.000 claims description 14
- 102000036639 antigens Human genes 0.000 claims description 14
- 238000003745 diagnosis Methods 0.000 claims description 13
- 210000004881 tumor cell Anatomy 0.000 claims description 10
- 206010006187 Breast cancer Diseases 0.000 claims description 8
- 208000026310 Breast neoplasm Diseases 0.000 claims description 8
- 230000033115 angiogenesis Effects 0.000 claims description 8
- 239000000523 sample Substances 0.000 claims description 8
- 230000004614 tumor growth Effects 0.000 claims description 8
- 208000022531 anorexia Diseases 0.000 claims description 7
- 206010061428 decreased appetite Diseases 0.000 claims description 7
- 201000001441 melanoma Diseases 0.000 claims description 7
- 230000000692 anti-sense effect Effects 0.000 claims description 6
- 108020004711 Nucleic Acid Probes Proteins 0.000 claims description 5
- 210000001035 gastrointestinal tract Anatomy 0.000 claims description 5
- 239000002853 nucleic acid probe Substances 0.000 claims description 5
- 206010009944 Colon cancer Diseases 0.000 claims description 4
- 206010033128 Ovarian cancer Diseases 0.000 claims description 4
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 4
- 208000029742 colonic neoplasm Diseases 0.000 claims description 4
- 210000000750 endocrine system Anatomy 0.000 claims description 3
- 230000012010 growth Effects 0.000 claims description 3
- 210000004994 reproductive system Anatomy 0.000 claims description 3
- 210000002345 respiratory system Anatomy 0.000 claims description 3
- 108091033319 polynucleotide Proteins 0.000 claims description 2
- 102000040430 polynucleotide Human genes 0.000 claims description 2
- 239000002157 polynucleotide Substances 0.000 claims description 2
- 238000011282 treatment Methods 0.000 abstract description 43
- 102000004127 Cytokines Human genes 0.000 abstract description 26
- 108090000695 Cytokines Proteins 0.000 abstract description 26
- 230000000694 effects Effects 0.000 abstract description 26
- 229940124829 interleukin-23 Drugs 0.000 description 69
- 102000005962 receptors Human genes 0.000 description 34
- 108020003175 receptors Proteins 0.000 description 34
- 241000699670 Mus sp. Species 0.000 description 32
- 108090000623 proteins and genes Proteins 0.000 description 29
- 239000003446 ligand Substances 0.000 description 26
- 230000014509 gene expression Effects 0.000 description 22
- 102000004169 proteins and genes Human genes 0.000 description 20
- 241000699666 Mus <mouse, genus> Species 0.000 description 16
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 16
- 230000004913 activation Effects 0.000 description 14
- 210000001519 tissue Anatomy 0.000 description 14
- 230000001225 therapeutic effect Effects 0.000 description 13
- 239000003814 drug Substances 0.000 description 12
- 239000000126 substance Substances 0.000 description 12
- 150000001413 amino acids Chemical class 0.000 description 11
- ARSRBNBHOADGJU-UHFFFAOYSA-N 7,12-dimethyltetraphene Chemical compound C1=CC2=CC=CC=C2C2=C1C(C)=C(C=CC=C1)C1=C2C ARSRBNBHOADGJU-UHFFFAOYSA-N 0.000 description 10
- 210000001744 T-lymphocyte Anatomy 0.000 description 10
- 150000001875 compounds Chemical class 0.000 description 10
- 208000035475 disorder Diseases 0.000 description 10
- 241001465754 Metazoa Species 0.000 description 9
- 125000003275 alpha amino acid group Chemical group 0.000 description 9
- 230000008859 change Effects 0.000 description 9
- 239000012530 fluid Substances 0.000 description 9
- 210000000987 immune system Anatomy 0.000 description 9
- 238000012360 testing method Methods 0.000 description 9
- 102000008070 Interferon-gamma Human genes 0.000 description 8
- 108010074328 Interferon-gamma Proteins 0.000 description 8
- 230000034994 death Effects 0.000 description 8
- 231100000517 death Toxicity 0.000 description 8
- 229960003130 interferon gamma Drugs 0.000 description 8
- 230000004044 response Effects 0.000 description 8
- 206010027476 Metastases Diseases 0.000 description 7
- 230000028993 immune response Effects 0.000 description 7
- 230000009401 metastasis Effects 0.000 description 7
- 230000002062 proliferating effect Effects 0.000 description 7
- 206010058314 Dysplasia Diseases 0.000 description 6
- 206010061218 Inflammation Diseases 0.000 description 6
- 230000000890 antigenic effect Effects 0.000 description 6
- 201000010099 disease Diseases 0.000 description 6
- 230000004054 inflammatory process Effects 0.000 description 6
- 239000003112 inhibitor Substances 0.000 description 6
- 210000000056 organ Anatomy 0.000 description 6
- 238000011160 research Methods 0.000 description 6
- 238000010561 standard procedure Methods 0.000 description 6
- 101710195550 Interleukin-23 receptor Proteins 0.000 description 5
- 210000000612 antigen-presenting cell Anatomy 0.000 description 5
- 230000037396 body weight Effects 0.000 description 5
- 208000015181 infectious disease Diseases 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 210000004072 lung Anatomy 0.000 description 5
- 239000012528 membrane Substances 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 229940124597 therapeutic agent Drugs 0.000 description 5
- 208000023275 Autoimmune disease Diseases 0.000 description 4
- 230000008045 co-localization Effects 0.000 description 4
- 230000007423 decrease Effects 0.000 description 4
- 210000004443 dendritic cell Anatomy 0.000 description 4
- 239000000032 diagnostic agent Substances 0.000 description 4
- 229940039227 diagnostic agent Drugs 0.000 description 4
- 239000002158 endotoxin Substances 0.000 description 4
- 210000002919 epithelial cell Anatomy 0.000 description 4
- 238000000684 flow cytometry Methods 0.000 description 4
- 108020001507 fusion proteins Proteins 0.000 description 4
- 102000037865 fusion proteins Human genes 0.000 description 4
- 230000002068 genetic effect Effects 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 230000036039 immunity Effects 0.000 description 4
- 230000003053 immunization Effects 0.000 description 4
- 238000002649 immunization Methods 0.000 description 4
- 230000003834 intracellular effect Effects 0.000 description 4
- 229920006008 lipopolysaccharide Polymers 0.000 description 4
- 210000002540 macrophage Anatomy 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 210000000822 natural killer cell Anatomy 0.000 description 4
- 210000000440 neutrophil Anatomy 0.000 description 4
- 230000001575 pathological effect Effects 0.000 description 4
- 239000000546 pharmaceutical excipient Substances 0.000 description 4
- 210000003491 skin Anatomy 0.000 description 4
- 239000013598 vector Substances 0.000 description 4
- 208000016261 weight loss Diseases 0.000 description 4
- 230000004580 weight loss Effects 0.000 description 4
- 208000005623 Carcinogenesis Diseases 0.000 description 3
- 238000002965 ELISA Methods 0.000 description 3
- 101000853012 Homo sapiens Interleukin-23 receptor Proteins 0.000 description 3
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 3
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 3
- 108091028043 Nucleic acid sequence Proteins 0.000 description 3
- 208000006994 Precancerous Conditions Diseases 0.000 description 3
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 3
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 3
- 239000012190 activator Substances 0.000 description 3
- 238000004458 analytical method Methods 0.000 description 3
- 238000003556 assay Methods 0.000 description 3
- 210000003719 b-lymphocyte Anatomy 0.000 description 3
- 210000001185 bone marrow Anatomy 0.000 description 3
- 230000036952 cancer formation Effects 0.000 description 3
- 231100000504 carcinogenesis Toxicity 0.000 description 3
- 230000015556 catabolic process Effects 0.000 description 3
- 230000024245 cell differentiation Effects 0.000 description 3
- 210000003169 central nervous system Anatomy 0.000 description 3
- 210000001072 colon Anatomy 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 239000013068 control sample Substances 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 239000002552 dosage form Substances 0.000 description 3
- 229940079593 drug Drugs 0.000 description 3
- 230000011132 hemopoiesis Effects 0.000 description 3
- 239000000833 heterodimer Substances 0.000 description 3
- 210000002865 immune cell Anatomy 0.000 description 3
- 208000027866 inflammatory disease Diseases 0.000 description 3
- 238000002347 injection Methods 0.000 description 3
- 239000007924 injection Substances 0.000 description 3
- 210000002510 keratinocyte Anatomy 0.000 description 3
- 230000007246 mechanism Effects 0.000 description 3
- 238000013508 migration Methods 0.000 description 3
- -1 radiotherapy Substances 0.000 description 3
- 230000019491 signal transduction Effects 0.000 description 3
- 230000011664 signaling Effects 0.000 description 3
- 238000010186 staining Methods 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 231100000331 toxic Toxicity 0.000 description 3
- 230000002588 toxic effect Effects 0.000 description 3
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 description 2
- 108020004414 DNA Proteins 0.000 description 2
- 238000009007 Diagnostic Kit Methods 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 2
- 102100039620 Granulocyte-macrophage colony-stimulating factor Human genes 0.000 description 2
- 102000003814 Interleukin-10 Human genes 0.000 description 2
- 108090000174 Interleukin-10 Proteins 0.000 description 2
- 108090001005 Interleukin-6 Proteins 0.000 description 2
- 101150009057 JAK2 gene Proteins 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- 206010028289 Muscle atrophy Diseases 0.000 description 2
- 108700020796 Oncogene Proteins 0.000 description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 208000000453 Skin Neoplasms Diseases 0.000 description 2
- NKANXQFJJICGDU-QPLCGJKRSA-N Tamoxifen Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN(C)C)=CC=1)/C1=CC=CC=C1 NKANXQFJJICGDU-QPLCGJKRSA-N 0.000 description 2
- 102000009618 Transforming Growth Factors Human genes 0.000 description 2
- 108010009583 Transforming Growth Factors Proteins 0.000 description 2
- 208000010399 Wasting Syndrome Diseases 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 210000004100 adrenal gland Anatomy 0.000 description 2
- 230000003092 anti-cytokine Effects 0.000 description 2
- 239000002246 antineoplastic agent Substances 0.000 description 2
- 230000006907 apoptotic process Effects 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 239000013060 biological fluid Substances 0.000 description 2
- 238000001815 biotherapy Methods 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 210000000481 breast Anatomy 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 230000012292 cell migration Effects 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 230000001413 cellular effect Effects 0.000 description 2
- 230000005754 cellular signaling Effects 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 230000037011 constitutive activity Effects 0.000 description 2
- 230000000875 corresponding effect Effects 0.000 description 2
- 230000002950 deficient Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 210000002889 endothelial cell Anatomy 0.000 description 2
- 210000003979 eosinophil Anatomy 0.000 description 2
- 210000003238 esophagus Anatomy 0.000 description 2
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 2
- 229960005420 etoposide Drugs 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 2
- 230000013595 glycosylation Effects 0.000 description 2
- 238000006206 glycosylation reaction Methods 0.000 description 2
- 102000057111 human IL23R Human genes 0.000 description 2
- 210000004408 hybridoma Anatomy 0.000 description 2
- 208000026278 immune system disease Diseases 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 230000006698 induction Effects 0.000 description 2
- 230000002757 inflammatory effect Effects 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 230000015788 innate immune response Effects 0.000 description 2
- 229940076144 interleukin-10 Drugs 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 238000011813 knockout mouse model Methods 0.000 description 2
- 210000002429 large intestine Anatomy 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 208000020816 lung neoplasm Diseases 0.000 description 2
- 210000004698 lymphocyte Anatomy 0.000 description 2
- 229920002521 macromolecule Polymers 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 210000003071 memory t lymphocyte Anatomy 0.000 description 2
- 239000002207 metabolite Substances 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 238000010369 molecular cloning Methods 0.000 description 2
- 239000003068 molecular probe Substances 0.000 description 2
- 210000001616 monocyte Anatomy 0.000 description 2
- 201000000585 muscular atrophy Diseases 0.000 description 2
- 238000002703 mutagenesis Methods 0.000 description 2
- 231100000350 mutagenesis Toxicity 0.000 description 2
- 210000000066 myeloid cell Anatomy 0.000 description 2
- 201000000050 myeloid neoplasm Diseases 0.000 description 2
- 210000001672 ovary Anatomy 0.000 description 2
- 230000007170 pathology Effects 0.000 description 2
- 210000003899 penis Anatomy 0.000 description 2
- 210000001428 peripheral nervous system Anatomy 0.000 description 2
- 230000000750 progressive effect Effects 0.000 description 2
- 230000002685 pulmonary effect Effects 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 238000003127 radioimmunoassay Methods 0.000 description 2
- 238000001959 radiotherapy Methods 0.000 description 2
- 210000000664 rectum Anatomy 0.000 description 2
- 230000001105 regulatory effect Effects 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 210000000813 small intestine Anatomy 0.000 description 2
- 241000894007 species Species 0.000 description 2
- 230000009870 specific binding Effects 0.000 description 2
- 210000004988 splenocyte Anatomy 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 210000002784 stomach Anatomy 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 210000001550 testis Anatomy 0.000 description 2
- 210000001685 thyroid gland Anatomy 0.000 description 2
- 239000003053 toxin Substances 0.000 description 2
- 231100000765 toxin Toxicity 0.000 description 2
- 108700012359 toxins Proteins 0.000 description 2
- 230000005740 tumor formation Effects 0.000 description 2
- VEEGZPWAAPPXRB-BJMVGYQFSA-N (3e)-3-(1h-imidazol-5-ylmethylidene)-1h-indol-2-one Chemical compound O=C1NC2=CC=CC=C2\C1=C/C1=CN=CN1 VEEGZPWAAPPXRB-BJMVGYQFSA-N 0.000 description 1
- SNKDCTFPQUHAPR-UHFFFAOYSA-N 1-fluoropyrimidine-2,4-dione Chemical compound FN1C=CC(=O)NC1=O SNKDCTFPQUHAPR-UHFFFAOYSA-N 0.000 description 1
- AYRABHFHMLXKBT-UHFFFAOYSA-N 2,6-Dimethyl-anthracen Natural products C1=C(C)C=CC2=CC3=CC(C)=CC=C3C=C21 AYRABHFHMLXKBT-UHFFFAOYSA-N 0.000 description 1
- NDMPLJNOPCLANR-UHFFFAOYSA-N 3,4-dihydroxy-15-(4-hydroxy-18-methoxycarbonyl-5,18-seco-ibogamin-18-yl)-16-methoxy-1-methyl-6,7-didehydro-aspidospermidine-3-carboxylic acid methyl ester Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 NDMPLJNOPCLANR-UHFFFAOYSA-N 0.000 description 1
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 description 1
- IJJWOSAXNHWBPR-HUBLWGQQSA-N 5-[(3as,4s,6ar)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]-n-(6-hydrazinyl-6-oxohexyl)pentanamide Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)NCCCCCC(=O)NN)SC[C@@H]21 IJJWOSAXNHWBPR-HUBLWGQQSA-N 0.000 description 1
- 208000030507 AIDS Diseases 0.000 description 1
- 101100366892 Anopheles gambiae Stat gene Proteins 0.000 description 1
- 206010002942 Apathy Diseases 0.000 description 1
- 241000212384 Bifora Species 0.000 description 1
- 230000005653 Brownian motion process Effects 0.000 description 1
- 229940126074 CDK kinase inhibitor Drugs 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 201000009030 Carcinoma Diseases 0.000 description 1
- 241000700198 Cavia Species 0.000 description 1
- 229940123587 Cell cycle inhibitor Drugs 0.000 description 1
- 241000282693 Cercopithecidae Species 0.000 description 1
- 229920001076 Cutan Polymers 0.000 description 1
- 102100034770 Cyclin-dependent kinase inhibitor 3 Human genes 0.000 description 1
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 1
- 101100366894 Drosophila melanogaster Stat92E gene Proteins 0.000 description 1
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 108700039887 Essential Genes Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 206010049238 Food aversion Diseases 0.000 description 1
- 206010017993 Gastrointestinal neoplasms Diseases 0.000 description 1
- 102000009465 Growth Factor Receptors Human genes 0.000 description 1
- 108010009202 Growth Factor Receptors Proteins 0.000 description 1
- 229940122236 Histamine receptor antagonist Drugs 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000945639 Homo sapiens Cyclin-dependent kinase inhibitor 3 Proteins 0.000 description 1
- 101001078133 Homo sapiens Integrin alpha-2 Proteins 0.000 description 1
- 102100025305 Integrin alpha-2 Human genes 0.000 description 1
- 102000006992 Interferon-alpha Human genes 0.000 description 1
- 108010047761 Interferon-alpha Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000004560 Interleukin-12 Receptors Human genes 0.000 description 1
- 108010017515 Interleukin-12 Receptors Proteins 0.000 description 1
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 description 1
- LRQKBLKVPFOOQJ-YFKPBYRVSA-N L-norleucine Chemical compound CCCC[C@H]([NH3+])C([O-])=O LRQKBLKVPFOOQJ-YFKPBYRVSA-N 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 208000036696 Microcytic anaemia Diseases 0.000 description 1
- 108010085220 Multiprotein Complexes Proteins 0.000 description 1
- 102000007474 Multiprotein Complexes Human genes 0.000 description 1
- 241000699660 Mus musculus Species 0.000 description 1
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 1
- 206010029098 Neoplasm skin Diseases 0.000 description 1
- 206010053159 Organ failure Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 229930012538 Paclitaxel Natural products 0.000 description 1
- 241001631646 Papillomaviridae Species 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 208000037581 Persistent Infection Diseases 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 238000011529 RT qPCR Methods 0.000 description 1
- 230000010799 Receptor Interactions Effects 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 101150099493 STAT3 gene Proteins 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- 229940123237 Taxane Drugs 0.000 description 1
- 102000002689 Toll-like receptor Human genes 0.000 description 1
- 108020000411 Toll-like receptor Proteins 0.000 description 1
- 102000007537 Type II DNA Topoisomerases Human genes 0.000 description 1
- 108010046308 Type II DNA Topoisomerases Proteins 0.000 description 1
- 108090000848 Ubiquitin Proteins 0.000 description 1
- 102000044159 Ubiquitin Human genes 0.000 description 1
- 208000002495 Uterine Neoplasms Diseases 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- 229940122803 Vinca alkaloid Drugs 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 238000002679 ablation Methods 0.000 description 1
- 230000035508 accumulation Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 230000010933 acylation Effects 0.000 description 1
- 238000005917 acylation reaction Methods 0.000 description 1
- 230000004721 adaptive immunity Effects 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 238000001261 affinity purification Methods 0.000 description 1
- 230000001270 agonistic effect Effects 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- VREFGVBLTWBCJP-UHFFFAOYSA-N alprazolam Chemical compound C12=CC(Cl)=CC=C2N2C(C)=NN=C2CN=C1C1=CC=CC=C1 VREFGVBLTWBCJP-UHFFFAOYSA-N 0.000 description 1
- 239000012491 analyte Substances 0.000 description 1
- 239000003098 androgen Substances 0.000 description 1
- 208000007502 anemia Diseases 0.000 description 1
- 239000004037 angiogenesis inhibitor Substances 0.000 description 1
- 229940121369 angiogenesis inhibitor Drugs 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 229940046836 anti-estrogen Drugs 0.000 description 1
- 230000001833 anti-estrogenic effect Effects 0.000 description 1
- 230000003432 anti-folate effect Effects 0.000 description 1
- 230000006023 anti-tumor response Effects 0.000 description 1
- 238000009175 antibody therapy Methods 0.000 description 1
- 229940127074 antifolate Drugs 0.000 description 1
- 239000000739 antihistaminic agent Substances 0.000 description 1
- 238000009166 antihormone therapy Methods 0.000 description 1
- 210000000436 anus Anatomy 0.000 description 1
- 230000004596 appetite loss Effects 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 238000002820 assay format Methods 0.000 description 1
- 206010003549 asthenia Diseases 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 239000012472 biological sample Substances 0.000 description 1
- 229920001222 biopolymer Polymers 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000001124 body fluid Anatomy 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 210000000621 bronchi Anatomy 0.000 description 1
- 210000003123 bronchiole Anatomy 0.000 description 1
- 238000005537 brownian motion Methods 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 230000000711 cancerogenic effect Effects 0.000 description 1
- 231100000357 carcinogen Toxicity 0.000 description 1
- 239000003183 carcinogenic agent Substances 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000020411 cell activation Effects 0.000 description 1
- 230000021164 cell adhesion Effects 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000011712 cell development Effects 0.000 description 1
- 230000032823 cell division Effects 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 238000002659 cell therapy Methods 0.000 description 1
- 230000010307 cell transformation Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 210000003679 cervix uteri Anatomy 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- 238000003776 cleavage reaction Methods 0.000 description 1
- 238000011260 co-administration Methods 0.000 description 1
- 230000004186 co-expression Effects 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 239000002875 cyclin dependent kinase inhibitor Substances 0.000 description 1
- 229940043378 cyclin-dependent kinase inhibitor Drugs 0.000 description 1
- 229960004397 cyclophosphamide Drugs 0.000 description 1
- 239000000409 cytokine receptor agonist Substances 0.000 description 1
- 239000000430 cytokine receptor antagonist Substances 0.000 description 1
- 210000004292 cytoskeleton Anatomy 0.000 description 1
- 210000000172 cytosol Anatomy 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 230000007812 deficiency Effects 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- RGLYKWWBQGJZGM-ISLYRVAYSA-N diethylstilbestrol Chemical compound C=1C=C(O)C=CC=1C(/CC)=C(\CC)C1=CC=C(O)C=C1 RGLYKWWBQGJZGM-ISLYRVAYSA-N 0.000 description 1
- 229960000452 diethylstilbestrol Drugs 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 229960003668 docetaxel Drugs 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 239000003596 drug target Substances 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 229960001904 epirubicin Drugs 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- 239000000328 estrogen antagonist Substances 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- MKXKFYHWDHIYRV-UHFFFAOYSA-N flutamide Chemical compound CC(C)C(=O)NC1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 MKXKFYHWDHIYRV-UHFFFAOYSA-N 0.000 description 1
- 229960002074 flutamide Drugs 0.000 description 1
- 230000004907 flux Effects 0.000 description 1
- 239000004052 folic acid antagonist Substances 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 238000013467 fragmentation Methods 0.000 description 1
- 238000006062 fragmentation reaction Methods 0.000 description 1
- 238000002825 functional assay Methods 0.000 description 1
- 210000000232 gallbladder Anatomy 0.000 description 1
- 210000000609 ganglia Anatomy 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 210000002149 gonad Anatomy 0.000 description 1
- 208000037824 growth disorder Diseases 0.000 description 1
- 210000003128 head Anatomy 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 238000001794 hormone therapy Methods 0.000 description 1
- 230000008348 humoral response Effects 0.000 description 1
- 238000009396 hybridization Methods 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 210000003016 hypothalamus Anatomy 0.000 description 1
- 229960001101 ifosfamide Drugs 0.000 description 1
- HOMGKSMUEGBAAB-UHFFFAOYSA-N ifosfamide Chemical compound ClCCNP1(=O)OCCCN1CCCl HOMGKSMUEGBAAB-UHFFFAOYSA-N 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000000984 immunochemical effect Effects 0.000 description 1
- 230000002163 immunogen Effects 0.000 description 1
- 230000002998 immunogenetic effect Effects 0.000 description 1
- 230000005847 immunogenicity Effects 0.000 description 1
- 238000001114 immunoprecipitation Methods 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 238000000338 in vitro Methods 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 230000001524 infective effect Effects 0.000 description 1
- 230000036512 infertility Effects 0.000 description 1
- 208000000509 infertility Diseases 0.000 description 1
- 231100000535 infertility Toxicity 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 229960004768 irinotecan Drugs 0.000 description 1
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 description 1
- 210000004153 islets of langerhan Anatomy 0.000 description 1
- 230000000155 isotopic effect Effects 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 238000002032 lab-on-a-chip Methods 0.000 description 1
- 150000002605 large molecules Chemical class 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 210000000088 lip Anatomy 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- 208000019017 loss of appetite Diseases 0.000 description 1
- 235000021266 loss of appetite Nutrition 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- 210000004216 mammary stem cell Anatomy 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- SGDBTWWWUNNDEQ-LBPRGKRZSA-N melphalan Chemical compound OC(=O)[C@@H](N)CC1=CC=C(N(CCCl)CCCl)C=C1 SGDBTWWWUNNDEQ-LBPRGKRZSA-N 0.000 description 1
- 229960001924 melphalan Drugs 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 1
- 229960000485 methotrexate Drugs 0.000 description 1
- 210000000274 microglia Anatomy 0.000 description 1
- VFKZTMPDYBFSTM-GUCUJZIJSA-N mitolactol Chemical compound BrC[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)CBr VFKZTMPDYBFSTM-GUCUJZIJSA-N 0.000 description 1
- 229950010913 mitolactol Drugs 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 210000000214 mouth Anatomy 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 210000000581 natural killer T-cell Anatomy 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 210000003739 neck Anatomy 0.000 description 1
- 230000001338 necrotic effect Effects 0.000 description 1
- 230000001613 neoplastic effect Effects 0.000 description 1
- 210000004498 neuroglial cell Anatomy 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 238000011580 nude mouse model Methods 0.000 description 1
- 230000000414 obstructive effect Effects 0.000 description 1
- 210000003300 oropharynx Anatomy 0.000 description 1
- 210000003101 oviduct Anatomy 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229960001592 paclitaxel Drugs 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 230000000849 parathyroid Effects 0.000 description 1
- 230000006320 pegylation Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000002093 peripheral effect Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 230000000079 pharmacotherapeutic effect Effects 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 230000008560 physiological behavior Effects 0.000 description 1
- 230000035479 physiological effects, processes and functions Effects 0.000 description 1
- 210000004560 pineal gland Anatomy 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 239000000902 placebo Substances 0.000 description 1
- 229940068196 placebo Drugs 0.000 description 1
- 230000004481 post-translational protein modification Effects 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 208000026446 profound weakness Diseases 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 210000002307 prostate Anatomy 0.000 description 1
- 230000012846 protein folding Effects 0.000 description 1
- 238000001742 protein purification Methods 0.000 description 1
- 239000002534 radiation-sensitizing agent Substances 0.000 description 1
- 230000002285 radioactive effect Effects 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 210000003289 regulatory T cell Anatomy 0.000 description 1
- 210000005000 reproductive tract Anatomy 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 210000003079 salivary gland Anatomy 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000013391 scatchard analysis Methods 0.000 description 1
- 230000007017 scission Effects 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000002864 sequence alignment Methods 0.000 description 1
- 108091006024 signal transducing proteins Proteins 0.000 description 1
- 102000034285 signal transducing proteins Human genes 0.000 description 1
- 201000000849 skin cancer Diseases 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 206010041823 squamous cell carcinoma Diseases 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 230000036435 stunted growth Effects 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 1
- 238000001356 surgical procedure Methods 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229960001603 tamoxifen Drugs 0.000 description 1
- DKPFODGZWDEEBT-QFIAKTPHSA-N taxane Chemical class C([C@]1(C)CCC[C@@H](C)[C@H]1C1)C[C@H]2[C@H](C)CC[C@@H]1C2(C)C DKPFODGZWDEEBT-QFIAKTPHSA-N 0.000 description 1
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 1
- 208000001608 teratocarcinoma Diseases 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 210000001541 thymus gland Anatomy 0.000 description 1
- 230000008427 tissue turnover Effects 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 210000003437 trachea Anatomy 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 210000002438 upper gastrointestinal tract Anatomy 0.000 description 1
- 238000011144 upstream manufacturing Methods 0.000 description 1
- 210000003932 urinary bladder Anatomy 0.000 description 1
- 206010046766 uterine cancer Diseases 0.000 description 1
- 210000004291 uterus Anatomy 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 108700026220 vif Genes Proteins 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- 229960004355 vindesine Drugs 0.000 description 1
- UGGWPQSBPIFKDZ-KOTLKJBCSA-N vindesine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(N)=O)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1N=C1[C]2C=CC=C1 UGGWPQSBPIFKDZ-KOTLKJBCSA-N 0.000 description 1
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 230000004584 weight gain Effects 0.000 description 1
- 235000019786 weight gain Nutrition 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/244—Interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/52—Cytokines; Lymphokines; Interferons
- C07K14/54—Interleukins [IL]
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/715—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons
- C07K14/7155—Receptors; Cell surface antigens; Cell surface determinants for cytokines; for lymphokines; for interferons for interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
Definitions
- the present invention concerns uses of mammalian cytokine molecules and related reagents. More specifically, the invention relates to identification of mammalian cytokine-like proteins and inhibitors thereof that can be used in the treatment of proliferative disorders.
- the immune system includes several types of lymphoid and myeloid cells, e.g., monocytes, macrophages, dendritic cells (DCs), eosinophils, T cells, B cells, and neutrophils. These lymphoid and myeloid cells produce secreted signaling proteins known as cytokines.
- the cytokines include, e.g., interleukin-10 (IL-10), interferon-gamma (IFNgamma), IL-12, and IL- 23.
- Immune response includes inflammation, i.e., the accumulation of immune cells systemically or in a particular location of the body.
- immune cells secrete cytokines which, in turn, modulate immune cell proliferation, development, differentiation, or migration.
- Immune response can produce pathological consequences, e.g., when it involves excessive inflammation, as in the autoimmune disorders, whereas impaired immune response may result in cancer.
- Anti-tumor response by the immune system includes innate immunity, e.g., as mediated by macrophages, NK cells, and neutrophils, and adaptive immunity, e.g., as mediated by antigen presenting cells (APCs), T cells, and B cells (see, e.g., Abbas, et al. (eds.) (2000) Cellular and Molecular Immunology, W.B. Saunders Co., Philadelphia, PA; Oppenheim and Feldmann (eds.) (2001) Cytokine Reference, Academic Press, San Diego, CA; von Andrian and Mackay
- Methods of modulating immune response have been used in the treatment of cancers, e.g., melanoma. These methods include treatment with cytokines or anti-cytokine antibodies, such as E -2, IL-12, tumor necrosis factor-alpha (TNFalpha), IFNgamma, granulocyte macrophage-colony stimulating factor (GM-CSF), and transforming growth factor (TGF).
- cytokines or anti-cytokine antibodies such as E -2, IL-12, tumor necrosis factor-alpha (TNFalpha), IFNgamma, granulocyte macrophage-colony stimulating factor (GM-CSF), and transforming growth factor (TGF).
- Interleukin-23 is a heterodimeric cytokine comprised of two subunits, i.e., pi 9 and p40.
- the pi 9 subunit is structurally related to IL-6, g ⁇ anulocyte-colony stimulating factor (G-CSF), and the p35 subunit of IL-12.
- the p40 subunit of IL-23 is also part of IL-12, a heterodimeric cytokine comprising p35 and p40.
- IL-23 mediates signaling by binding to a heterodimeric receptor, comprised of IL-23R and IL-12betal .
- the IL-12betal subunit is shared by the IL-12 receptor, which is composed of IL-12betal and IL-12beta2.
- cytokines such as IL-12 or IFNgamma produce toxic side effects (see, e.g., Naylor and Hadden (2003) Int. Immunopharmacol. 3: 1205-1215; Fernandez, et al. (1999) J. Immunol. 162:609-617).
- the present invention addresses these problems by providing methods of using agonists and antagonists of IL-23.
- SUMMARY OF THE INVENTION [0006] The present invention is based upon the discovery that an agonist or antagonist of IL-23 can modulate tumor growth.
- the present invention provides a method of modulating tumor growth comprising contacting a tumor cell with an effective amount of an agonist or antagonist of IL-
- the present invention provides the above method wherein the agonist or antagonist of IL-23 comprises a binding composition that specifically binds a polypeptide or nucleic acid of pl9 (SEQ ID NOs: l, 2, 3, or 4); or IL-23R (SEQ ID NOs:5 or 6); or the above method wherein the binding composition comprises: an antigen-binding site of an antibody; an extracellular region of IL-23R (SEQ ID NOs:5 or 6); a small molecule; an anti-sense nucleic acid or small interference RNA (siRNA); or a detectable label; and the above method wherein the binding composition comprises: a polyclonal antibody; a monoclonal antibody; a humanized antibody, or a fragment thereof; an Fab, Fv, or F(ab') 2 fragment; or
- Yet another embodiment of the present invention provides a method of modulating tumor growth comprising contacting a tumor cell with an effective amount of an agonist or antagonist of IL-23; wherein the tumor cell is:a colon cancer cell; an ovarian cancer cell; a breast cancer cell; or a melanoma cell.
- the invention provides a method of treating a subject suffering from a cancer or tumor comprising administering to the subject an effective amount of an agonist or antagonist of IL-23; and the above method wherein the antagonist of IL-23 inhibits: growth of the cancer or tumor; cachexia; anorexia; or angiogenesis. Also provided is the above method wherein the antagonist of IL-23 comprises a binding composition that specifically binds a polypeptide or nucleic acid of: pi 9 (SEQ ID NOs: l , 2, 3, or 4) or IL-23 R (SEQ ID NOs:5 or 6).
- the binding composition comprises: an antigen-binding site of an antibody; an extracellular region of IL-23R (SEQ ID NOs:5 or 6); an anti-sense nucleic acid or small interference RNA (siRNA); a small molecule; or a detectable label; and the above method wherein the binding composition comprises: a polyclonal antibody; a monoclonal antibody; a humanized antibody, or a fragment thereof; an Fab, Fv, or F(ab') 2 fragment; or a peptide mimetic of an antibody.
- the present invention provides a method of diagnosis of a cancer or tumor comprising contacting a sample from a subject with the binding compositions of the above method, as well as the above method of diagnosis, wherein the binding composition comprises a nucleic acid probe or primer that specifically binds or hybridizes to the polynucleotide of SEQ ID NOs: 1 , 2, or 5.
- kits for the diagnosis of a cancer or tumor comprising the binding composition of the above method and a compartment or instructions for use or disposal. Also provided is the above kit wherein the binding composition comprises an antibody that specifically binds to p 19 (SEQ ID NOs: 1 , 2,
- Activation may have the same meaning, e.g., activation, stimulation, or treatment of a cell or receptor with a ligand, unless indicated otherwise by the context or explicitly.
- Ligand encompasses natural and synthetic ligands, e.g., cytokines, cytokine variants, analogues, muteins, and binding compositions derived from antibodies.
- Ligand also encompasses small molecules, e.g., peptide mimetics of cytokines and peptide mimetics of antibodies.
- Activity of a molecule may describe or refer to the binding of the molecule to a ligand or to a receptor, to catalytic activity; to the ability to stimulate gene expression or cell signaling, differentiation, or maturation; to antigenic activity, to the modulation of activities of other molecules, and the like. "Activity” of a molecule may also refer to activity in modulating or maintaining cell-to-cell interactions, e.g., adhesion, or activity in maintaining a structure of a cell, e.g., cell membranes or cytoskeleton.
- Activity can also mean specific activity, e.g., [catalytic activity]/[mg protein], or [immunological activity]/[mg protein], concentration in a biological compartment, or the like.
- Proliferative activity encompasses an activity that promotes, that is necessary for, or that is specifically associated with, e.g., normal cell division, as well as cancer, tumors, dysplasia, cell transformation, metastasis, and angiogenesis.
- administering and “treatment,” as it applies to an animal, human, experimental subject, cell, tissue, organ, or biological fluid, refers to contact of an exogenous pharmaceutical, therapeutic, diagnostic agent, compound, or composition to the animal, human, subject, cell, tissue, organ, or biological fluid.
- administering can refer, e.g., to therapeutic, placebo, pharmacokinetic, diagnostic, research, and experimental methods.
- Treatment of a cell encompasses contact of a reagent to the cell, as well as contact of a reagent to a fluid, where the fluid is in contact with the cell.
- administering and “treatment” also means in vitro and ex vivo treatments, e.g., of a cell, by a reagent, diagnostic, binding composition, or by another cell.
- Treatment refers to therapeutic treatment, prophylactic or preventative measures, to research and diagnostic applications.
- “Treatment of a cell” also encompasses situations where the IL-23 agonist or IL-23 antagonist contacts IL-23 receptor (heterodimer of IL-23R and B -12Rbetal), e.g., in the fluid phase or colloidal phase, as well as situations where the agonist or antagonist contacts a fluid, e.g., where the fluid is in contact with a cell or receptor, but where it has not been demonstrated that the agonist or antagonist contacts the cell or receptor.
- IL-23 receptor heterodimer of IL-23R and B -12Rbetal
- Binding may be defined as an association of the binding composition with a target where the association results in reduction in the normal Brownian motion of the binding composition, in cases where the binding composition can be dissolved or suspended in solution.
- Cachexia is a wasting syndrome involving loss of muscle (muscle wasting) and fat, resulting from a disorder in metabolism. Cachexia occurs in various cancers, chronic pulmonary obstructive disorder (COPD), advanced organ failure, and AIDS.
- COPD chronic pulmonary obstructive disorder
- Cancer cachexia is the cachexia that occurs with cancer. Cancer cachexia is characterized by, e.g., marked weight loss, anorexia, asthenia, and anemia.
- amino acid sequences one of skill will recognize that an individual substitution to a nucleic acid, peptide, polypeptide, or protein sequence which substitutes an amino acid or a small percentage of amino acids in the encoded sequence for a conserved amino acid is a "conservatively modified variant.” Conservative substitution tables providing functionally similar amino acids are well known in the art. An example of a conservative substitution is the exchange of an amino acid in one of the following groups for another amino acid of the same group (U.S. Pat. No. 5,767,063 issued to Lee, et al.; Kyte and
- Effective amount encompasses an amount sufficient to ameliorate or prevent a symptom or sign of the medical condition. Effective amount also means an amount sufficient to allow or facilitate diagnosis.
- An effective amount for a particular patient or veterinary subject may vary depending on factors such as the condition being treated, the overall health of the patient, the method route and dose of administration and the severity of side affects (see, e.g., U.S. Pat. No. 5,888,530 issued to Netti, et al).
- An effective amount can be the maximal dose or dosing protocol that avoids significant side effects or toxic effects.
- the effect will result in an improvement of a diagnostic measure or parameter by at least 5%, usually by at least 10%>, more usually at least 20%, most usually at least 30%, preferably at least 40%>, more preferably at least 50%, most preferably at least 60%), ideally at least 70%, more ideally at least 80%, and most ideally at least 90%, where 100% is defined as the diagnostic parameter shown by a normal subject (see, e.g., Maynard, et al (1996) A Handbook of SOPs for Good Clinical Practice, Interpharm Press, Boca Raton, FL; Dent (2001) Good Laboratory and Good Clinical Practice, Urch Publ, London, UK).
- Immunode condition or “immune disorder” encompasses, e.g., pathological inflammation, an inflammatory disorder, and an autoimmune disorder or disease.
- Immuno condition also refers to infections, persistent infections, and proliferative conditions, such as cancer, tumors, and angiogenesis, including infections, tumors, and cancers that resist irradication by the immune system.
- Inhibitors and “antagonists” or “activators” and “agonists” refer to inhibitory or activating molecules, respectively, e.g., for the activation of, e.g., a ligand, receptor, cofactor, gene, cell, tissue, or organ.
- a modulator of, e.g., a gene, a receptor, a ligand, or a cell is a molecule that alters an activity of the gene, receptor, ligand, or cell, where activity can be activated, inhibited, or altered in its regulatory properties.
- the modulator may act alone, or it may use a cofactor, e.g., a protein, metal ion, or small molecule.
- Inhibitors are compounds that decrease, block, prevent, delay activation, inactivate, desensitize, or down regulate, e.g., a gene, protein, ligand, receptor, or cell.
- Activators are compounds that increase, activate, facilitate, enhance activation, sensitize, or up regulate, e.g., a gene, protein, ligand, receptor, or cell.
- An inhibitor may also be defined as a composition that reduces, blocks, or inactivates a constitutive activity.
- An "agonist” is a compound that interacts with a target to cause or promote an increase in the activation of the target.
- An "antagonist” is a compound that opposes the actions of an agonist.
- An antagonist prevents, reduces, inhibits, or neutralizes the activity of an agonist.
- An antagonist can also prevent, inhibit, or reduce constitutive activity of a target, e.g., a target receptor, even where there is no identified agonist.
- Endpoints in activation or inhibition can be monitored as follows. Activation, inhibition, and response to treatment, e.g., of a cell, physiological fluid, tissue, organ, and animal or human subject, can be monitored by an endpoint.
- An endpoint of inhibition is generally 75% of the control or less, preferably
- an endpoint of activation is at least 150%) the control, preferably at least two times the control, more preferably at least four times the control, and most preferably at least 10 times the control.
- Ligand refers, e.g., to a small molecule, peptide, polypeptide, and membrane associated or membrane-bound molecule, or complex thereof, that can act as an agonist or antagonist of a receptor.
- Ligand also encompasses an agent that is not an agonist or antagonist, but that can bind to the receptor without significantly influencing its biological properties, e.g., signaling or adhesion.
- ligand includes a membrane-bound ligand that has been changed, e.g., by chemical or recombinant methods, to a soluble version of the membrane-bound ligand. By convention, where a ligand is membrane-bound on a first cell, the receptor usually occurs on a second cell.
- the second cell may have the same or a different identity as the first cell.
- a ligand or receptor may be entirely intracellular, that is, it may reside in the cytosol, nucleus, or some other intracellular compartment. The ligand or receptor may change its location, e.g., from an intracellular compartment to the outer face of the plasma membrane.
- the complex of a ligand and receptor is termed a "ligand receptor complex.” Where a ligand and receptor are involved in a signaling pathway, the ligand occurs at an upstream position and the receptor occurs at a downstream position of the signaling pathway.
- Small molecules are provided for the treatment of physiology and disorders of tumors and cancers.
- “Small molecule” is defined as a molecule with a molecular weight that is less than 10 kD, typically less than 2 kD, and preferably less than 1 kD.
- Small molecules include, but are not limited to, inorganic molecules, organic molecules, organic molecules containing an inorganic component, molecules comprising a radioactive atom, synthetic molecules, peptide mimetics, and antibody mimetics. As a therapeutic, a small molecule may be more permeable to cells, less susceptible to degradation, and less apt to elicit an immune response than large molecules.
- Small molecules such as peptide mimetics of antibodies and cytokines, as well as small molecule toxins are described (see, e.g., Casset, et al. (2003) Biochem. Biophys. Res. Commun. 307:198-205; Muyldermans (2001) J. Biotechnol. 74:277-302; Li (2000) Nat. Biotechnol. 18: 1251-1256; protestopoulos, et al. (2002) Curr. Med. Chem. 9:41 1 -420; Monfardini, et al. (2002) Curr. Phann. Des. 8:2185-
- a specified ligand binds to a particular receptor and does not bind in a significant amount to other proteins present in the sample.
- the antibody, or binding composition derived from the antigen-binding site of an antibody, of the contemplated method binds to its antigen, or a variant or mutein thereof, with an affinity that is at least two fold greater, preferably at least ten times greater, more preferably at least 20- times greater, and most preferably at least 100-times greater than the affinity with any other antibody, or binding composition derived thereof.
- the antibody will have an affinity that is greater than about 10 9 liters/mol, as determined, e.g., by Scatchard analysis (Munsen, et al. ( 1980) Analyt. Biochem. 107:220-239).
- the present invention provides methods of using polypeptides, nucleic acids, variants, muteins, and mimetics of the IL-23 heterodimer, pi 9 subunit, p40 subunit, the IL-23 receptor heterodimer, IL-23R subunit, or IL-12Rbetal subunit.
- a hyperkine i.e., a fusion protein comprising, e.g., the pi 9 subunit linked to the p40 subunit, as well as nucleic acids encoding the hyperkine (see, e.g., SEQ ID NOs: 10 or 1 1) (Oppmann, et al, supra; Fischer, et al. (1997) Nature Biotechnol. 15: 142-145; Rakemann, et al. (1999) J. Biol. Chem. 274:1257-1266; and Peters, et ⁇ /.(1998) J. Immunol. 161 :3575- 3581).
- Interleukin-23 (IL-23; a.k.a. IL-B30) is a heterodimeric cytokine composed of a novel pi 9 subunit (SEQ ID NOs: 2 or 4) and the p40 subunit (SEQ ID NOs: 8 or 9) of IL- 12 (Oppmann, et al, supra). Like p35, pl9 requires co-expression of p40 for biological activity (Wiekowski, et al, supra).
- the IL-23 receptor comprises a novel receptor subunit (IL-23R; SEQ ID NO: 6) that binds pl9 and EL-12Rbetal (SEQ ID NO: 7) that binds p40 (see, e,g., Parham, et al. (2002) J. Immunol. 168:5699-5708).
- IL-23R novel receptor subunit
- SEQ ID NO: 6 binds pl9
- EL-12Rbetal SEQ ID NO: 7
- p40 see, e,g., Parham, et al. (2002) J. Immunol. 168:5699-5708).
- These two receptor subunits form the functional signaling complex and are expressed on CD4 + CD45Rb'° memory T cells as well as IFNgamma activated bone marrow macrophages (Parham, et al, supra).
- Antibodies can be raised to various cytokine proteins, including individual, polymorphic, allelic, strain, or species variants, and fragments thereof, both in their naturally occurring (full-length) forms or in their recombinant forms (see, e.g., SEQ ID NO: 2, 4, 10, or 1 1). Additionally, antibodies can be raised to receptor proteins (see, e.g., SEQ ID NO: 6) in both their native (or active) forms or in their inactive, e.g., denatured, forms. Anti-idiotypic antibodies may also be used.
- an IL-23 agonist i.e., IL-23 or IL-23 hyperkine
- IFNgamma interferon-gamma
- IL-23 preferentially stimulates memory as opposed to na ⁇ ve T cell populations in both human and mouse.
- IL-23 activates a number of intracellular cell- signaling molecules, e.g., Jak2, Tyk2, Statl , Stat2, Stat3, and Stat4.
- IL-12 activates this same group of molecules, but Stat4 response to IL-23 is relatively weak, while Stat4 response to IL-12 is strong (Oppmann, et al, supra; Parham, et al. (2002) J. Immunol. 168:5699-5708).
- IL-12 and IL-23 engage similar signal transduction mechanisms. IL-23 engaging its receptor complex, activates Jak2, Tyk2, and Stat-1 , -3, -4, and -5, as does IL-12. However Stat-4 activation is significantly weaker in response to IL-23 than IL-12. Also, in contrast to IL-12, the most prominent Stat induced by IL-23 is Stat-3 (see, e.g., Parham, et al, supra).
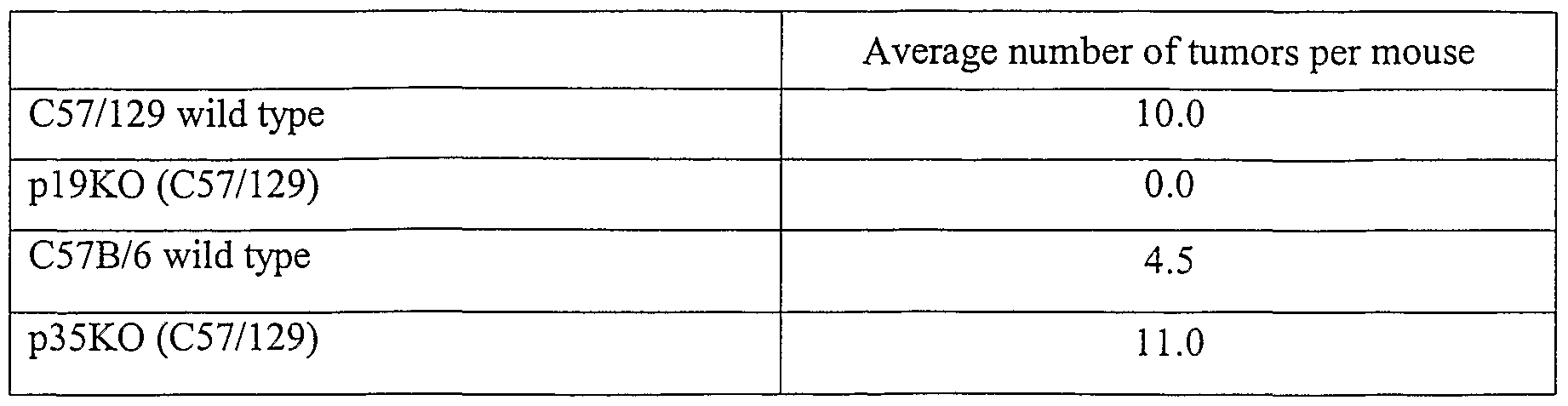
- IL-23 Administration of the pl9 subunit of IL-23 can result in, e.g., stunted growth, infertility, and death of animals, as well as inflammatory infiltrates, e.g., in the gastrointestinal tract, lungs, skin, and liver, and epithelial cell hyperplasia, microcytic anemia, increased neutrophil count, increased serum TNFalpha; and increased expression of acute phase genes in liver; (Wiekowski, et al, supra).
- Enhanced IL-23 expression occured in immortalized not transformed epithelial cell lines.
- IL-23 may provide an early signal of tumor potential in vivo.
- Gene expression data is useful tool in the diagnosis and treatment of diseases and pathological conditions (see, e.g., Li and Wong (2001) Genome Informatics 12:3-13; Lockhart, et al. (1996) Nature Biotechnol 14: 1675-1680; Homey, et al. (2000) J. Immunol. 164:3465-3470; Debets, et al. (2000) J. Immunol. 165:4950-4956).
- the present invention provides methods of using agonists and antagonist of
- IL-23 An agonist of IL-23 encompasses, e.g., IL-23, an IL-23 variant, mutein, hyperkine, or peptide mimetic, agonistic antibodies to IL-23 R, and nucleic acids encoding these agonists.
- Antagonists of IL-23 include, e.g., antibodies to IL-23, blocking antibodies to IL-23R, a soluble receptor based on the extracellular region of a subunit of the IL-23R, peptide mimetics thereto, and nucleic acids encoding these antagonists.
- the present invention provides methods of using agonists and antagonists of pl9, the complex of pl9 and p40, IL-23R, and the complex of IL-23R and IL-12Rbetal , including binding compositions that specifically bind to proteins and protein complexes of pl9, the complex of pl9 and p40, IL-23R, and the complex of IL-23R and IL-12Rbetal .
- An IL-23 hyperkine encompasses, e.g., a fusion protein comprising the polypeptide sequence of pi 9 and p40, where pi 9 and p40 occur in one continous polypeptide chain.
- the sequences of pl9 and p40 may be in either order.
- the fusion protein may contain a linker sequence, residing in between the sequences of pi 9 and p40, in one continuous polypeptide chain.
- Regions of increased antigenicity can be used for antibody generation.
- Regions of increased antigenicity of human pl9 occur, e.g., at amino acids 16-28; 57-87; 1 10- 114; 136-154; and 182-186 of GenBank AAQ89442 (gi:37183284).
- Regions of increased antigenicity of human IL-23R occur, e.g., at amino acids 22-33; 57-63; 68-74; 101-1 12; 117- 133; 164-177; 244-264; 294-302; 315-326; 347-354; 444-473; 510-530; and 554-558 of
- GenBank AAM44229 (gi: 21239252). Analysis was by a Parker plot using Vector NTI® Suite (Informax, Inc, Bethesda, MD).
- the present invention also provides an IL-23 antagonist that is a soluble receptor, i.e., comprising an extracellular region of EL-23R, e.g., amino acids 1-353 of GenBankAAM44229, or a fragment thereof, where the extracellular region or fragment thereof specifically binds to IL-23.
- Mouse EL-23R is GenBank
- NP_653131 (gi:21362353). Muteins and variants are contemplated, e.g., pegylation or mutagenesis to remove or replace deamidating Asn residues.
- Monoclonal, polyclonal, and humanized antibodies can be prepared (see, e.g.,
- Immunization can be performed by DNA vector immunization, see, e.g., Wang, et al. (1997) Virology 228:278-284.
- animals can be immunized with cells bearing the antigen of interest.
- Splenocytes can then be isolated from the immunized animals, and the splenocytes can fused with a myeloma cell line to produce a hybridoma (Meyaard, et al. (1997) Immunity 7:283-290; Wright, et al. (2000) Immunity 13:233-242; Preston, et al. (1997) Eur. J. Immunol. 27: 191 1-1918).
- Resultant hybridomas can be screened for production of the desired antibody by functional assays or biological assays, that is, assays not dependent on possession of the purified antigen. Immunization with cells may prove superior for antibody generation than immunization with purified antigen (Kaithamana, et al. (1999) J. Immunol. 163:5157-5164).
- Antibody to antigen and ligand to receptor binding properties can be measured, e.g., by surface plasmon resonance (Karlsson, et al. (1991) J. Immunol. Methods 145:229-240; Neri, et al. (1997) Nat. Biotechnol.
- Antibodies can be used for affinity purification to isolate the antibody's target antigen and associated bound proteins, see, e.g., Wilchek, et al. (1984) Meth. Enzymol. 104:3-55.
- Antibodies will usually bind with at least a KQ of about 10 "3 M, more usually at least 10 "6 M, typically at least 10 "7 M, more typically at least 10 "8 M, preferably at least about 10 "9 M, and more preferably at least 10 "10 M, and most preferably at least 10 " " M (see, e.g., Presta, et al. (2001) Thromb. Haemost. 85:379-389; Yang, et al. (2001) Crit. Rev. Oncol. Hematol. 38:17-23; Carnahan, et al. (2003) Clin. Cancer Res. (Suppl.) 9:3982s-3990s).
- Soluble receptors comprising the extracellular domains of IL-23R or IL-
- Soluble receptors can be prepared and used according to standard methods (see, e.g., Jones, et al. (2002) Biochim. Biophys. Acta
- the present invention provides IL-23 and anti-IL-23R for use, e.g., in the treatment of proliferative conditions and disorders, including cancer, tumors, angiogenesis, cachexia, cancer cachexia, anorexia, and pre-cancerous disorders, e.g., dysplasia.
- Nucleic acids are also provided for these therapeutic uses, e.g., nucleic acids encoding IL-23 or IL- 23R, or an antigenic fragment thereof, the corresponding anti-sense nucleic acids, and hybridization products thereof.
- the invention also provides compositions for siRNA interference (see, e.g., Arenz and Schepers (2003) Naturwissenschaften 90:345-359; Sazani and Kole (2003) J. Clin. Invest. 1 12:481-486; Pirollo, et al. (2003) Pharmacol. Therapeutics 99:55-77; Wang, et al. (2003) Antisense Nucl. Acid Drug Devel. 13: 169-189).
- compositions including an agonist or antagonist of IL-23, the cytokine analogue or mutein, antibody thereto, or nucleic acid thereof, is admixed with a pharmaceutically acceptable carrier or excipient, see, e.g., Remington's Pharmaceutical Sciences and U.S. Pharmacopeia: National Formulary, Mack Publishing Company, Easton, PA (1984).
- a pharmaceutically acceptable carrier or excipient see, e.g., Remington's Pharmaceutical Sciences and U.S. Pharmacopeia: National Formulary, Mack Publishing Company, Easton, PA (1984).
- Formulations of therapeutic and diagnostic agents may be prepared by mixing with physiologically acceptable carriers, excipients, or stabilizers in the form of, e.g., lyophilized powders, slurries, aqueous solutions or suspensions (see, e.g., Hardman, et al. (2001) Goodman and Gilman 's The Pharmacological Basis of Therapeutics , McGraw-Hill, New
- the route of administration is by, e.g., topical or cutaneous application, subcutaneous injection, injection or infusion by intravenous, intraperitoneal, intracerebral, intramuscular, intraocular, intraarterial, intracerebrospinal, intralesional, or pulmonary routes, or by sustained release systems or an implant.
- Gene transfer vectors e.g., for the central nervous system, have been described (see, e.g., Cua, et al. (2001) J. Immunol. 166:602-608; Sidman et al. (1983) Biopolymers 22:547-556; Langer, et al. (1981) J. Biomed. Mater. Res.
- an administration regimen for a therapeutic depends on several factors, including the serum or tissue turnover rate of the entity, the level of symptoms, the immunogenicity of the entity, and the accessibility of the target cells in the biological matrix.
- an administration regimen maximizes the amount of therapeutic delivered to the patient consistent with an acceptable level of side effects.
- the amount of biologic delivered depends in part on the particular entity and the severity of the condition being treated. Guidance in selecting appropriate doses of antibodies, cytokines, and small molecules are available (see, e.g., Wawrzynczak (1996) Antibody Therapy, Bios Scientific Pub.
- Antibodies, antibody fragments, and cytokines can be provided by continuous infusion, or by doses at intervals of, e.g., one day, one week, or 1-7 times per week. Doses may be provided intravenously, subcutaneously, topically, orally, nasally, rectally, intramuscular, intracerebrally, intraspinally, or by inhalation.
- a preferred dose protocol is one involving the maximal dose or dose frequency that avoids significant undesirable side effects.
- a total weekly dose is generally at least 0.05 ⁇ g/kg body weight, more generally at least 0.2 ⁇ g/kg, most generally at least 0.5 ⁇ g/kg, typically at least 1 ⁇ g/kg, more typically at least 10 ⁇ g/kg, most typically at least 100 ⁇ g/kg, preferably at least 0.2 mg/kg, more preferably at least 1.0 mg/kg, most preferably at least 2.0 mg/kg, optimally at least 10 mg/kg, more optimally at least 25 mg/kg, and most optimally at least 50 mg/kg (see, e.g., Yang, et al. (2003) New Engl. J. Med. 349:427-434; Herold, et al. (2002) New Engl. J. Med. 346: 1692- 1698; Liu, et al. (1999) J. Neurol. Neurosurg. Psych. 67:451-456; Portielji, et al. (20003)
- a small molecule therapeutic e.g., a peptide mimetic, natural product, or organic chemical
- the desired dose of a small molecule therapeutic is about the same as for an antibody or polypeptide, on a moles/kg basis.
- An effective amount for a particular patient may vary depending on factors such as the condition being treated, the overall health of the patient, the method route and dose of administration and the severity of side affects (see, e.g., Maynard, et al. (1996) A Handbook ofSOPsfor Good Clinical Practice, Interpharm Press, Boca Raton, FL; Dent (2001) Good Laboratory and Good Clinical Practice, Urch Publ., London, UK).
- Typical veterinary, experimental, or research subjects include monkeys, dogs, cats, rats, mice, rabbits, guinea pigs, horses, and humans.
- Determination of the appropriate dose is made by the clinician, e.g., using parameters or factors known or suspected in the art to affect treatment or predicted to affect treatment. Generally, the dose begins with an amount somewhat less than the optimum dose and it is increased by small increments thereafter until the desired or optimum effect is achieved relative to any negative side effects.
- Important diagnostic measures include those of symptoms of, e.g., the inflammation or level of inflammatory cytokines produced.
- a biologic that will be used is derived from the same species as the animal targeted for treatment, thereby minimizing a humoral response to the reagent.
- a second therapeutic agent e.g., a cytokine, steroid, chemotherapeutic agent, antibiotic, or radiation
- a second therapeutic agent e.g., a cytokine, steroid, chemotherapeutic agent, antibiotic, or radiation
- An effective amount of therapeutic will decrease the symptoms typically by at least 10%; usually by at least 20%; preferably at least about 30%; more preferably at least 40%, and most preferably by at least 50%.
- This invention provides IL-23 proteins, fragments thereof, nucleic acids, and fragments thereof, in a diagnostic kit. Also provided are binding compositions, including antibodies or antibody fragments, for the detection of IL-23 and IL-23 receptor, and metabolites and breakdown products thereof.
- the kit will have a compartment containing either a pi 9 polypeptide, or an antigenic fragment thereof, a binding composition thereto, or a nucleic acid, e.g., a nucleic acid probe or primer.
- the nucleic acid probe or primer specifically hybridizes under stringent conditions to a nucleic acid encoding pi 9 or IL-
- the kit can comprise, e.g., a reagent and a compartment, a reagent and instructions for use, or a reagent with a compartment and instructions for use.
- the reagent can comprise pi 9, the complex of p i 9 and p40, IL-23 R, the complex of IL-23R and
- kits for determining the binding of a test compound can comprise a control compound, a labeled compound, and a method for separating free labeled compound from bound labeled compound.
- Diagnostic assays can be used with biological matrices such as live cells, cell extracts, cell lysates, fixed cells, cell cultures, bodily fluids, or forensic samples.
- Conjugated antibodies useful for diagnostic or kit purposes include antibodies coupled to dyes, isotopes, enzymes, and metals (see, e.g., Le Doussal, et al.
- This invention provides polypeptides and nucleic acids of EL-23 and IL-23R, fragments thereof, in a diagnostic kit, e.g., for the diagnosis of proliferative conditions, cancer, tumors, and precancerous disorders, e.g., dysplasia.
- a diagnostic kit e.g., for the diagnosis of proliferative conditions, cancer, tumors, and precancerous disorders, e.g., dysplasia.
- binding compositions including antibodies or antibody fragments, for the detection of pl9, the complex of pl9 and p40, IL-23R, the complex of IL-
- the kit will have a compartment containing either a IL-23 or IL-23R polypeptide, or an antigenic fragment thereof, a binding composition thereto, or a nucleic acid, such as a nucleic acid probe, primer, or molecular beacon (see, e.g., Rajendran, et al. (2003) Nucleic Acids Res. 31 :5700-5713; Cockerill (2003) Arch. Pathol. Lab. Med. 127: 1 1 12-1 120; Zammatteo, et al.
- a method of diagnosis can comprise contacting a sample from a subject, e.g., a test subject, with a binding composition that specifically binds to a polypeptide or nucleic acid of pl 9, the complex of pl 9 and p40, IL-23R, and the complex of IL-23R and IL-12Rbetal .
- the method can further comprise contacting a sample from a control subject, normal subject, or normal tissue or fluid from the test subject, with the binding composition.
- the method can additionally comprise comparing the specific binding of the composition to the test subject with the specific binding of the composition to the normal subject, control subject, or normal tissue or fluid from the test subject.
- Expression or activity of a test sample or test subject can be compared with that from a control sample or control subject.
- a control sample can comprise, e.g., a sample of non-affected or non-inflamed tissue in a patient suffering from an immune disorder.
- Expression or activity from a control subject or control sample can be provided as a predetermined value, e.g., acquired from a statistically appropriate group of control subjects.
- the present invention provides methods for using agonists and antagonists of
- IL-23 for the treatment and diagnosis of inflammatory disorders and conditions, e.g., neoplastic diseases, cancers, tumors, angiogenesis, precancerous conditions such as dysplasias, anorexia, cachexia, and cancer cachexia, by modulating immune response.
- inflammatory disorders and conditions e.g., neoplastic diseases, cancers, tumors, angiogenesis, precancerous conditions such as dysplasias, anorexia, cachexia, and cancer cachexia, by modulating immune response.
- the present invention provides methods of treating or diagnosing a proliferative condition or disorder, e.g., cancer of the uterus, cervix, breast, prostate, testes, penis, gastrointestinal tract, e.g., esophagus, oropharynx, stomach, small or large intestines, colon, or rectum, kidney, renal cell, bladder, bone, bone marrow, skin, head or neck, skin, liver, gall bladder, heart, lung, pancreas, salivary gland, adrenal gland, thyroid, brain, ganglia, central nervous system (CNS) and peripheral nervous system (PNS), and immune system, e.g., spleen or thymus.
- a proliferative condition or disorder e.g., cancer of the uterus, cervix, breast, prostate, testes, penis, gastrointestinal tract, e.g., esophagus, oropharynx, stomach, small or large intestines, colon, or rect
- the present invention provides methods of treating, e.g., immunogenic tumors, non-immunogenetic tumors, dormant tumors, virus-induced cancers, e.g., epithelial cell cancers, endothelial cell cancers, squamous cell carcinomas, papillomavirus, adenocarcinomas, lymphomas, carcinomas, melanomas, leukemias, myelomas, sarcomas, teratocarcinomas, chemically-induced cancers, metastasis, and angiogenesis.
- immunogenic tumors e.g., immunogenic tumors, non-immunogenetic tumors, dormant tumors, virus-induced cancers, e.g., epithelial cell cancers, endothelial cell cancers, squamous cell carcinomas, papillomavirus, adenocarcinomas, lymphomas, carcinomas, melanomas, leukemias, myelo
- the invention also contemplates reducing tolerance to a tumor cell or cancer cell antigen, e.g., by modulating activity of a regulatory T cell (Treg) (see, e.g., Ramirez- Montagut, et al. (2003) Oncogene 22:3180-3187; Sawaya, et al. (2003) New Engl. J. Med. 349: 1501-1509; Farrar, et al. (1999) J. Immunol. 162:2842-2849; Le, et al. (2001) J.
- Treg regulatory T cell
- the present invention provides methods for treating a proliferative condition, cancer, tumor, or precancerous condition such as a dysplasia, with an agonist or antagonist of IL-23, with at least one additional therapeutic or diagnostic agent.
- the at least one additional therapeutic or diagnostic agent can be, e.g., a cytokine or cytokine antagonist, such as IL-12, interferon-alpha, or anti-epidermal growth factor receptor, doxorubicin, epirubicin, an anti- folate, e.g., methotrexate or fluoruracil, irinotecan, cyclophosphamide, radiotherapy, hormone or anti-hormone therapy, e.g., androgen, estrogen, anti-estrogen, flutamide, or diethylstilbestrol, surgery, tamoxifen, ifosfamide, mitolactol, an alkylating agent, e.g., melphalan or cis-platin
- Vaccines can be provided, e.g., as a soluble protein or as a nucleic acid encoding the protein (see, e.g., Le, et al, supra; Greco and Zellefsky (eds.) (2000) Radiotherapy of Prostate Cancer, Harwood Academic, Amsterdam; Shapiro and Junior
- the present invention provides methods for the treatment and diagnosis of anorexia and cachexia, including cancer cachexia.
- Cachexia is a wasting syndrome that occurs in a number of diseases, including cancer, e.g., cancer of the lung and upper gastrointestinal tract. Cachexia occurs in about half of all cancer patients. Diagnosis of cachexia is by a history of substantial weight loss, loss of appetite, and profound weakness, in the context of advanced disease, and muscle wasting (loss of lean body mass).
- Cytokines e.g., IL-6, DL-1 , T ⁇ Falpha, and IFNgamma
- cachexia see, e.g., MacDonald, et al, supra; Rubin, supra; Tisdale, supra; Lelli, et al, supra; Argiles, et al, supra).
- EMH extramedullary hematopoiesis
- the gastrointestinal tract comprises, e.g., the lips, mouth, esophagus, stomach, small intestines, appendix, large intestines, colon, anus, and rectum.
- the respiratory tract comprises, e.g., the trachea, bronchioles, bronchi, lungs, alveoli.
- the reproductive system includes, e.g., the testes, penis, ovaries, uterus, fallopian tubes.
- the endocrine system includes, e.g., the pituitary, hypothalamus, pineal gland, thyroid gland, parathyroid, endocrine pancreas, islets, gonads, and adrenal gland.
- FACS Fluorescence Activated Cell Sorting
- Fluorescent reagents suitable for modifying nucleic acids including nucleic acid primers and probes, polypeptides, and antibodies, for use, e.g., as diagnostic reagents, are available (Molecular Probes (2003) Catalogue, Molecular Probes, Inc., Eugene, OR; Sigma-Aldrich (2003) Catalogue, St. Louis, MO).
- IL-23 pi 9 deficient mice were generated as described in Cua, et al, supra.
- mice specifically lacking in IL-23 had a B6/129 F2 background.
- Skin tumors were chemically induced in either wild-type (wt) or IL-23 deficient mice (pl9KO mice). Tumors were initiated using 50 micrograms of 7,12- dimethylbenz[a]anthracene (DMBA) followed by a promotion steps consisting of two treatments of 30 micrograms each of TPA per week (see, e.g., Oft, et al. (2002) Nat. Cell.
- DMBA 7,12- dimethylbenz[a]anthracene
- Expression of the pi 9 subunit of IL-23 and the IL-23 R subunit of IL-23 receptor was elevated in a number of cancers, tumors, and cell lines, e.g., cancer of the gastrointestinal tract, reproductive tract, skin, and breast (Table 1 ).
- RNA from tissues or cell pellets was extracted using RNeasy® columns
- cDNA were prepared and used as templates for quantitative real time PCR. cDNA (25 ng) was analysed for expression of a range of genes using GeneAmp® 5700 Sequence Detection System (Applied Biosystems, Foster City, CA). Analysis of cDNA samples from normal and tumor colon and ovary tissue was normalized to expression of the housekeeping gene, ubiquitin.
- mice treated antagonists to IL-23 e.g., by treatment with anti-pl9 antibody, or by genetic ablation of the pi 9 subunit (pl9KO).
- pi 9 is a subunit of EL-23 only, while p40 is a subunit of both IL-23 and IL-12.
- treatment with an IL-12 under some conditions, exacerbated tumors, i.e., resulted in an increase in tumor volume, relative to control mice.
- Tumors in mice resulted in cancer, cancer cachexia, extramedullary hematopoiesis, and death.
- Treatment of tumor-bearing Balb/c mice with anti-pl 9 antibody resulted in a halt to increases in tumor volume, while treatment with anti-p40 antibody provoked weight gain of the animal, likely a reversal of cachexia, but an increase in tumor volume (Table 2).
- mice were injected with 1 x 10 6 EpXT tumor cells (s.c).
- Tumor bearing nude mice (Ep2XBl nu/nu) died from lethal lung metastasis, with deaths occurring at from days 22-42 after the injection.
- Tumor bearing Exp2XBl Balb/c mice died at about days 22-49 after the injection, where the BalbC/c mice died in absence of lung metastasis.
- Cachexia was indicated by the decrease in body weight occurring (prior to death). Progressive weight loss occurred, starting at about day 16. The initial weight, at day 1 was 22-23 grams, while the weight at death was in the range of 16-18 grams.
- Antibody treatment was with C17.8 rat anti-p40 antibody (1 mg/week). With antibody treatment, the Ep2XBl-Balb/C mice (immunocompetant mice), survived until about day 64, after which deaths occurred until day 85. Anti-p40 antibody treatment also resulted in a maintenance of body weight (at about 17 grams) in half of the mice, with a progressive increase in body weight of the remaining mice, to a maximum, within the time frame of the experiment, of 22-23 grams. Thus, anti-p40 antibody resulted in improvement in health, according to survival time and regain of body weight, though anti-p40 could also result in a decline in health, as shown by an increase in tumor size (Table 2).
- Cancer was chemically induced by treatment with DMBA (50 micrograms) and 2 x 30 micrograms tetradecanoylphorbol- 13 -acetate (TPA) per week (Gschwendt, et ⁇ /.(1991) Trends Biochem Sci. 16: 167-169).
- Chemical carcinogenesis treatments were applied to B6/129 wild type mice and to pl 9KO mice. Wild type mice readily developed tumors but the pl9KO mice did not acquire tumors (Table 3).
- Tissue and cell expression of the subunits of IL-23 and subunits of IL-12 was determined, after carcinogen treatment.
- DMBA alone, TPA alone, and DMBA with TPA, induced expression of the pl9 subunit of IL-23, these chemicals was applied to the mouse's back.
- TPA Five hours after treatment with TPA resulted in an increase in pi 9 expression (2.5 control; 15.5 with TPA treatment), but relatively little change in p40 expression (2.0 control; 3.5 with TPA treatment). Five hours after treatment with DMBA plus TPA resulted in large increases in pi 9 expression (6.0 control; 32.0 DMBA + TPA), but moderate levels of p40 expression (2.0 control; 4.0 DMBA + TPA).
- Response of human keratinocytes to, e.g., DMBA, TPA, and lipopolysaccharide (LPS) was also determined (Table 5). TPA specifically induced pi 9, with little or no induction of p40, the common subunit of IL-23 and IL-12.
- Toll-like receptors that bind LPS occur on keratinocytes (see, e.g., Song, et al. (2002) J. Invest. Dermatol. 1 19:424-432).
- Etoposide is an anti-cancer agent that inhibits topoisomerase II and induces apoptosis (see, e.g., Robertson, et al. (2000) J. Biol. Chem. 275:32438-32443; Karpinich, et al. (2000) J. Biol.
- Anti-pl9 antibodies were tested for their effect on the 4T1 mouse breast cancer cell model. Mice were treated with control mlgGl (27F1 1) antibody or with anti-pl9 antibody (29A2). Tumor growth was monitored on days 1 , 3, 4, 5, 6, 7, 8, 9, 10, and 1 1.
- Antibodies (1 mg/dose) were administered on days 2, 5, 8, and 10. On day 4, the tumor size of the control antibody treated mouse was about 175 mm 3 , while tumor size of the anti-pl9 antibody treated mouse was about 135 mm 3 . Thus, anti-pl9 antibody is effective in treating a model of breast cancer. After day 4, tumors in both groups grew at about the same rate, indicating that the antibody dose was not sufficient to counteract the IL-23 expressed by the tumor at later periods in time.
- Histology of the Ep2 mouse breast cancer model demonstrated co-localization of IL-23R and NK cells, as determined by staining for pi 9, which resides bound to IL-23R, and by staining for CD49B, a marker for NK cells. This co-localization occurred in the central part of the tumor, i.e., in the necrotic region. Histology of the Ep2 mouse breast cancer also demonstrated co-localization of pi 9 and T cells. T cell location was determined by staining for CD3. This co-localization occurred at the peripheral part of the tumor.
- SEQ ID NO: 1 is human IL-23pl 9 nucleic acid sequence.
- SEQ ID NO: 2 is human IL-23pl9 amino acid sequence.
- SEQ ID NO: 3 is mouse IL-23pl9 nucleic acid sequence.
- SEQ ID NO: 4 is mouse IL-23pl9 amino acid sequence.
- SEQ ID NO: 5 is human IL-23 receptor nucleic acid sequence.
- SEQ ID NO 6 is human IL-23 receptor amino acid sequence.
- SEQ ID NO 7 is human IL-12Rbetal amino acid sequence.
- SEQ ID NO 8 is human IL-12 p40 amino acid sequence.
- SEQ ID NO 9 is mouse EL-12 p40 amino acid sequence.
- SEQ ID NO 10 is mouse IL-23 hyperkine
- SEQ ID NO 1 1 is human IL-23 hyperkine.
Abstract
Description
Claims
Priority Applications (16)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PL04718824T PL1601694T3 (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 antagonists; related reagents |
| NZ541898A NZ541898A (en) | 2003-03-10 | 2004-03-09 | Uses of IL-23 antagonists for the manufacture of a medicament for the treatment of tumors |
| JP2006507006A JP4605798B2 (en) | 2003-03-10 | 2004-03-09 | IL-23 agonists and IL-23 antagonists; use of related reagents |
| EP04718824A EP1601694B1 (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 antagonists; related reagents |
| CA2518262A CA2518262C (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 agonists and antagonists; related reagents |
| AT04718824T ATE440864T1 (en) | 2003-03-10 | 2004-03-09 | USE OF IL-23 ANTAGONISTS; RELATED REAGENTS |
| MXPA05009717A MXPA05009717A (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 agonists and antagonists; related reagents. |
| SI200431281T SI1601694T1 (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 antagonists; related reagents |
| DE602004022781T DE602004022781D1 (en) | 2003-03-10 | 2004-03-09 | USE OF IL-23 ANTAGONISTS; RELATED REAGENTS |
| DK04718824T DK1601694T3 (en) | 2003-03-10 | 2004-03-09 | Uses of IL23 antagonists, related reagents |
| AU2004219625A AU2004219625B9 (en) | 2003-03-10 | 2004-03-09 | Uses of IL-23 agonists and antagonists; related reagents |
| BRPI0408247-8A BRPI0408247A (en) | 2003-03-10 | 2004-03-09 | uses of il-23 antagonists and agonists and related reagents |
| CN2004800067148A CN1759123B (en) | 2003-03-10 | 2004-03-09 | Uses of IL-23 agonists and antagonists, related reagents |
| NO20054630A NO20054630L (en) | 2003-03-10 | 2005-10-07 | Applications of IL-23 agonists and antagonists, related reagents |
| HK05111191.9A HK1076477A1 (en) | 2003-03-10 | 2005-12-07 | Uses of il-23 antagonists; related reagents il-23 |
| AU2010212372A AU2010212372B2 (en) | 2003-03-10 | 2010-08-16 | Uses of IL-23 agonists and antagonists; related reagents |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US45367203P | 2003-03-10 | 2003-03-10 | |
| US60/453,672 | 2003-03-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2004081190A2 true WO2004081190A2 (en) | 2004-09-23 |
| WO2004081190A3 WO2004081190A3 (en) | 2005-02-03 |
Family
ID=32990805
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2004/007198 WO2004081190A2 (en) | 2003-03-10 | 2004-03-09 | Uses of il-23 agonists and antagonists; related reagents |
Country Status (23)
| Country | Link |
|---|---|
| US (4) | US7282204B2 (en) |
| EP (2) | EP2108658B1 (en) |
| JP (4) | JP4605798B2 (en) |
| CN (1) | CN1759123B (en) |
| AT (1) | ATE440864T1 (en) |
| AU (2) | AU2004219625B9 (en) |
| BR (1) | BRPI0408247A (en) |
| CA (1) | CA2518262C (en) |
| CL (1) | CL2004000467A1 (en) |
| CY (1) | CY1109468T1 (en) |
| DE (1) | DE602004022781D1 (en) |
| DK (1) | DK1601694T3 (en) |
| ES (1) | ES2330220T3 (en) |
| HK (1) | HK1076477A1 (en) |
| MX (1) | MXPA05009717A (en) |
| NO (1) | NO20054630L (en) |
| NZ (2) | NZ567860A (en) |
| PL (1) | PL1601694T3 (en) |
| PT (1) | PT1601694E (en) |
| SI (1) | SI1601694T1 (en) |
| TW (2) | TWI439285B (en) |
| WO (1) | WO2004081190A2 (en) |
| ZA (1) | ZA200506879B (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005079837A1 (en) * | 2004-02-17 | 2005-09-01 | Schering Corporation | Methods of modulating il-23 activity; related reagents |
| WO2006068987A2 (en) * | 2004-12-20 | 2006-06-29 | Schering Corporation | Uses of il-23 antagonists in the treatment of diabetes mellitus |
| WO2007005955A2 (en) | 2005-06-30 | 2007-01-11 | Centocor, Inc. | Anti-il-23 antibodies, compositions, methods and uses |
| WO2007024846A2 (en) | 2005-08-25 | 2007-03-01 | Eli Lilly And Company | Anit-il-23 antibiodies |
| WO2007027714A2 (en) | 2005-08-31 | 2007-03-08 | Schering Corporation | Engineered anti-il-23 antibodies |
| US7247711B2 (en) | 2003-05-09 | 2007-07-24 | Centocor, Inc. | IL-23p40 specific antibody |
| US7252971B2 (en) | 2004-09-24 | 2007-08-07 | Centocor, Inc. | IL-23p40 specific immunoglobulin derived proteins |
| EP1882699A1 (en) * | 2006-07-27 | 2008-01-30 | Heinfried Radeke | Regulation of interleukin-23 |
| EP1910414A2 (en) * | 2005-06-30 | 2008-04-16 | Archemix Corp. | Polynucleotides and polypeptides of the il-12 family of cytokines |
| WO2008106131A2 (en) | 2007-02-28 | 2008-09-04 | Schering Corporation | Combination therapy for treatment of immune disorders |
| WO2008106134A2 (en) | 2007-02-28 | 2008-09-04 | Schering Corporation | Engineered anti-il-23r antibodies |
| WO2010027767A1 (en) * | 2008-08-27 | 2010-03-11 | Schering Corporation | Engineered anti-il-23r antibodies |
| WO2010046530A1 (en) * | 2008-10-20 | 2010-04-29 | Helsingin Yliopiston Rahastot | Methods and uses involving genetic aberrations of nav3 and aberrant expression of multiple genes |
| US7790862B2 (en) | 2006-06-13 | 2010-09-07 | Zymogenetics, Inc. | IL-17 and IL-23 antagonists and methods of using the same |
| WO2010115786A1 (en) * | 2009-04-01 | 2010-10-14 | Glaxo Group Limited | Anti-il-23 immunoglobulins |
| WO2009007849A3 (en) * | 2007-07-06 | 2011-01-06 | Valorisation Hsj, Societe En Commandite | Il-23 receptor antagonists and uses thereof |
| US7910703B2 (en) | 2006-03-10 | 2011-03-22 | Zymogenetics, Inc. | Antagonists to IL-17A, IL-17F, and IL-23P19 and methods of use |
| US7935344B2 (en) | 2005-12-29 | 2011-05-03 | Centocor Ortho Biotech Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| EP2426145A1 (en) | 2007-02-23 | 2012-03-07 | Schering Corporation | Engineered anti-IL-23p19 antibodies |
| EP2426144A1 (en) | 2007-02-23 | 2012-03-07 | Schering Corporation | Engineered anti-IL-23p19 antibodies |
| EP2456787A2 (en) * | 2009-07-24 | 2012-05-30 | The Board of Trustees of The Leland Stanford Junior University | Cytokine compositions and methods of use thereof |
| US8524230B2 (en) | 2005-09-01 | 2013-09-03 | Merck Sharp & Dohme Corp. | Use of IL-23 and IL-17 antagonists to treat autoimmune ocular inflammatory disease |
| WO2014004436A2 (en) | 2012-06-27 | 2014-01-03 | Merck Sharp & Dohme Corp. | Crystalline anti-human il-23 antibodies |
| US8722033B2 (en) | 2009-10-26 | 2014-05-13 | Amgen Inc. | Human IL-23 antigen binding proteins |
| WO2014093203A1 (en) | 2012-12-13 | 2014-06-19 | Merck Sharp & Dohme Corp. | SOLUTION FORMULATIONS OF ENGINEERED ANTI-IL-23p19 ANTIBODIES |
| US8778346B2 (en) | 2010-11-04 | 2014-07-15 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| US9023358B2 (en) | 2013-03-08 | 2015-05-05 | Eli Lilly And Company | Antibodies that bind to IL-23 |
| AU2012258409B2 (en) * | 2005-06-30 | 2016-01-07 | Centocor, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| US10059763B2 (en) | 2014-09-03 | 2018-08-28 | Boehringer Ingelheim International Gmbh | Compound targeting IL-23A and TNF-alpha and uses thereof |
| EP3268028A4 (en) * | 2015-03-11 | 2018-09-05 | Rush University Medical Center | Compositions and methods for treating cancer |
| US10507241B2 (en) | 2014-07-24 | 2019-12-17 | Boehringer Ingelheim International Gmbh | Biomarkers useful in the treatment of IL-23A related diseases |
| US11078265B2 (en) | 2012-05-03 | 2021-08-03 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| US11548941B2 (en) | 2018-11-20 | 2023-01-10 | Janssen Biotech, Inc. | Safe and effective method of treating psoriasis with anti-IL-23 specific antibody |
| US11633476B2 (en) | 2017-05-02 | 2023-04-25 | Merck Sharp & Dohme Llc | Stable formulations of programmed death receptor 1 (PD-1) antibodies and methods of use thereof |
| US11780911B2 (en) | 2019-05-23 | 2023-10-10 | Janssen Biotech, Inc. | Method of treating inflammatory bowel disease with a combination therapy of antibodies to IL-23 and TNF alpha |
| US11845798B2 (en) | 2017-05-02 | 2023-12-19 | Merck Sharp & Dohme Llc | Formulations of anti-LAG3 antibodies and co-formulations of anti-LAG3 antibodies and anti-PD-1 antibodies |
Families Citing this family (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020137890A1 (en) * | 1997-03-31 | 2002-09-26 | Genentech, Inc. | Secreted and transmembrane polypeptides and nucleic acids encoding the same |
| WO2004042009A2 (en) * | 2002-10-30 | 2004-05-21 | Genentech, Inc. | Inhibition of il-17 production |
| DK1601694T3 (en) * | 2003-03-10 | 2009-12-14 | Schering Corp | Uses of IL23 antagonists, related reagents |
| US20060193821A1 (en) * | 2004-03-05 | 2006-08-31 | Diener John L | Aptamers to the human IL-12 cytokine family and their use as autoimmune disease therapeutics |
| US20070066550A1 (en) * | 2004-03-05 | 2007-03-22 | Diener John L | Aptamers to the human IL-12 cytokine family and their use as autoimmune disease therapeutics |
| TWI426918B (en) * | 2007-02-12 | 2014-02-21 | Merck Sharp & Dohme | Use of il-23 antagonists for treatment of infection |
| AR068723A1 (en) * | 2007-10-05 | 2009-12-02 | Glaxo Group Ltd | PROTEIN THAT JOINS ANTIGENS THAT JOINS HUMAN IL-23 AND ITS USES |
| US20100331545A1 (en) | 2007-10-24 | 2010-12-30 | Nippon Chemiphar Co., Ltd. | Regulator for signaling toll-like receptor, which comprises cathepsin inhibitor as active ingredient |
| WO2009136286A2 (en) * | 2008-05-05 | 2009-11-12 | Novimmune Sa | Anti-il-17a/il-17f cross-reactive antibodies and methods of use thereof |
| WO2010036924A2 (en) * | 2008-09-25 | 2010-04-01 | The United States Of America, As Represented By The Secretary, Department Of Healthe And Human Serv. | Inflammatory genes and microrna-21 as biomarkers for colon cancer prognosis |
| WO2010128407A2 (en) | 2009-05-05 | 2010-11-11 | Novimmune S.A. | Anti-il-17f antibodies and methods of use thereof |
| US20110086806A1 (en) * | 2009-10-09 | 2011-04-14 | Anaphore, Inc. | Polypeptides that Bind IL-23R |
| US20110311527A1 (en) | 2010-06-16 | 2011-12-22 | Allergan, Inc. | IL23p19 ANTIBODY INHIBITOR FOR TREATING OCULAR AND OTHER CONDITIONS |
| USRE49026E1 (en) | 2011-06-14 | 2022-04-12 | Medical Diagnostic Laboratories, Llc | Polypeptides that bound to IL-23 receptor and inhibit binding of IL-23 and cell signaling thereof |
| US8946150B2 (en) | 2011-06-14 | 2015-02-03 | Medical Diagnostic Laboratories, LLC. | Polypeptides that bound to IL-23 receptor and inhibit binding of IL-23 and cell signaling thereof |
| AR102417A1 (en) | 2014-11-05 | 2017-03-01 | Lilly Co Eli | ANTI-TNF- / ANTI-IL-23 BIESPECTIFIC ANTIBODIES |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001018051A2 (en) * | 1999-09-09 | 2001-03-15 | Schering Corporation | Mammalian interleukin-12 p40 and interleukin b30, combinations thereof, antibodies, uses in pharmaceutical compositions |
| WO2001085790A2 (en) * | 2000-05-10 | 2001-11-15 | Schering Corporation | Mammalian cytokine receptor subunit proteins, related reagents and methods |
| WO2002029060A2 (en) * | 2000-10-06 | 2002-04-11 | Immunex Corporation | Hematopoietin receptors hpr1 and hpr2 |
| WO2002097048A2 (en) * | 2001-05-30 | 2002-12-05 | Centocor, Inc. | ANTI-p40 IMMUNOGLOBULIN DERIVED PROTEINS, COMPOSITIONS, METHODS AND USES |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6176962B1 (en) | 1990-02-28 | 2001-01-23 | Aclara Biosciences, Inc. | Methods for fabricating enclosed microchannel structures |
| US5767063A (en) | 1991-03-29 | 1998-06-16 | Genentech, Inc. | Human IL-8 receptor and antibodies thereto |
| US6132764A (en) | 1994-08-05 | 2000-10-17 | Targesome, Inc. | Targeted polymerized liposome diagnostic and treatment agents |
| WO1997003715A1 (en) | 1995-07-21 | 1997-02-06 | The General Hospital Corporation | Method and apparatus of enhancing the delivery of a pharmaceutical formulation |
| US6056973A (en) | 1996-10-11 | 2000-05-02 | Sequus Pharmaceuticals, Inc. | Therapeutic liposome composition and method of preparation |
| US6060284A (en) * | 1997-07-25 | 2000-05-09 | Schering Corporation | DNA encoding interleukin-B30 |
| US5842787A (en) | 1997-10-09 | 1998-12-01 | Caliper Technologies Corporation | Microfluidic systems incorporating varied channel dimensions |
| US6326482B1 (en) | 1999-04-23 | 2001-12-04 | Genentech, Inc. | SH2 domain-containing peptides |
| DE69942671D1 (en) | 1998-12-01 | 2010-09-23 | Facet Biotech Corp | HUMANIZED ANTIKOERPER AGAINST GAMMA INTERFERON |
| DK1601694T3 (en) * | 2003-03-10 | 2009-12-14 | Schering Corp | Uses of IL23 antagonists, related reagents |
-
2004
- 2004-03-09 DK DK04718824T patent/DK1601694T3/en active
- 2004-03-09 CN CN2004800067148A patent/CN1759123B/en not_active Expired - Fee Related
- 2004-03-09 EP EP09163834.6A patent/EP2108658B1/en not_active Expired - Lifetime
- 2004-03-09 DE DE602004022781T patent/DE602004022781D1/en not_active Expired - Lifetime
- 2004-03-09 TW TW100126443A patent/TWI439285B/en not_active IP Right Cessation
- 2004-03-09 WO PCT/US2004/007198 patent/WO2004081190A2/en active Application Filing
- 2004-03-09 JP JP2006507006A patent/JP4605798B2/en not_active Expired - Fee Related
- 2004-03-09 NZ NZ567860A patent/NZ567860A/en not_active IP Right Cessation
- 2004-03-09 PL PL04718824T patent/PL1601694T3/en unknown
- 2004-03-09 CA CA2518262A patent/CA2518262C/en not_active Expired - Fee Related
- 2004-03-09 AU AU2004219625A patent/AU2004219625B9/en not_active Ceased
- 2004-03-09 BR BRPI0408247-8A patent/BRPI0408247A/en not_active IP Right Cessation
- 2004-03-09 PT PT04718824T patent/PT1601694E/en unknown
- 2004-03-09 ES ES04718824T patent/ES2330220T3/en not_active Expired - Lifetime
- 2004-03-09 MX MXPA05009717A patent/MXPA05009717A/en active IP Right Grant
- 2004-03-09 NZ NZ541898A patent/NZ541898A/en not_active IP Right Cessation
- 2004-03-09 US US10/797,157 patent/US7282204B2/en active Active
- 2004-03-09 TW TW093106213A patent/TWI357336B/en not_active IP Right Cessation
- 2004-03-09 SI SI200431281T patent/SI1601694T1/en unknown
- 2004-03-09 AT AT04718824T patent/ATE440864T1/en active
- 2004-03-09 CL CL200400467A patent/CL2004000467A1/en unknown
- 2004-03-09 EP EP04718824A patent/EP1601694B1/en not_active Expired - Lifetime
-
2005
- 2005-08-26 ZA ZA200506879A patent/ZA200506879B/en unknown
- 2005-10-07 NO NO20054630A patent/NO20054630L/en not_active Application Discontinuation
- 2005-12-07 HK HK05111191.9A patent/HK1076477A1/en not_active IP Right Cessation
-
2007
- 2007-02-26 JP JP2007046287A patent/JP2007197453A/en active Pending
- 2007-08-31 US US11/848,462 patent/US20080057058A1/en not_active Abandoned
-
2009
- 2009-08-24 US US12/546,230 patent/US8034341B2/en not_active Expired - Lifetime
- 2009-10-06 CY CY20091101030T patent/CY1109468T1/en unknown
-
2010
- 2010-08-16 AU AU2010212372A patent/AU2010212372B2/en not_active Ceased
- 2010-09-07 JP JP2010200382A patent/JP2011032275A/en not_active Withdrawn
-
2011
- 2011-10-10 US US13/270,077 patent/US20120034219A1/en not_active Abandoned
-
2013
- 2013-08-15 JP JP2013168870A patent/JP2014005286A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001018051A2 (en) * | 1999-09-09 | 2001-03-15 | Schering Corporation | Mammalian interleukin-12 p40 and interleukin b30, combinations thereof, antibodies, uses in pharmaceutical compositions |
| WO2001085790A2 (en) * | 2000-05-10 | 2001-11-15 | Schering Corporation | Mammalian cytokine receptor subunit proteins, related reagents and methods |
| WO2002029060A2 (en) * | 2000-10-06 | 2002-04-11 | Immunex Corporation | Hematopoietin receptors hpr1 and hpr2 |
| WO2002097048A2 (en) * | 2001-05-30 | 2002-12-05 | Centocor, Inc. | ANTI-p40 IMMUNOGLOBULIN DERIVED PROTEINS, COMPOSITIONS, METHODS AND USES |
Non-Patent Citations (2)
| Title |
|---|
| LO CHIA-HUI ET AL: "Antitumor and antimetastatic activity of IL-23." JOURNAL OF IMMUNOLOGY, vol. 171, no. 2, 15 July 2003 (2003-07-15), pages 600-607, XP002297119 ISSN: 0022-1767 * |
| UGAI SHIN-ICHI ET AL: "Transduction of the IL-21 and IL-23 genes in human pancreatic carcinoma cells produces natural killer cell-dependent and independent antitumor effects." CANCER GENE THERAPY, vol. 10, no. 10, October 2003 (2003-10), pages 771-778, XP002297118 ISSN: 0929-1903 * |
Cited By (106)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7247711B2 (en) | 2003-05-09 | 2007-07-24 | Centocor, Inc. | IL-23p40 specific antibody |
| WO2005079837A1 (en) * | 2004-02-17 | 2005-09-01 | Schering Corporation | Methods of modulating il-23 activity; related reagents |
| US7252971B2 (en) | 2004-09-24 | 2007-08-07 | Centocor, Inc. | IL-23p40 specific immunoglobulin derived proteins |
| US7807471B2 (en) | 2004-09-24 | 2010-10-05 | Centocor Ortho Biotech, Inc. | IL-23p40 specific immunoglobulin derived proteins, compositions, epitopes, methods and uses |
| WO2006068987A2 (en) * | 2004-12-20 | 2006-06-29 | Schering Corporation | Uses of il-23 antagonists in the treatment of diabetes mellitus |
| WO2006068987A3 (en) * | 2004-12-20 | 2006-09-14 | Schering Corp | Uses of il-23 antagonists in the treatment of diabetes mellitus |
| US7820168B2 (en) | 2004-12-20 | 2010-10-26 | Schering Corporation | Treatment of diabetes using antibodies to IL-23, IL-23 receptor and IL-17 |
| EP2292758A3 (en) * | 2004-12-20 | 2013-12-25 | Merck Sharp & Dohme Corp. | Uses of mammalian cytokine; related reagents |
| JP2009501006A (en) * | 2005-06-30 | 2009-01-15 | セントカー・インコーポレーテツド | Anti-IL-23 antibodies, compositions, methods and uses |
| EP1896073A4 (en) * | 2005-06-30 | 2009-08-19 | Centocor Ortho Biotech Inc | Anti-il-23 antibodies, compositions, methods and uses |
| US10272152B2 (en) | 2005-06-30 | 2019-04-30 | Janssen Biotech, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| EP1896073A2 (en) | 2005-06-30 | 2008-03-12 | Centocor, Inc. | Anti-il-23 antibodies, compositions, methods and uses |
| US9714287B2 (en) | 2005-06-30 | 2017-07-25 | Janssen Biotech, Inc. | Anti-IL-23 antibody compositions |
| EP1910414A2 (en) * | 2005-06-30 | 2008-04-16 | Archemix Corp. | Polynucleotides and polypeptides of the il-12 family of cytokines |
| US10576150B2 (en) | 2005-06-30 | 2020-03-03 | Janssen Biotech, Inc. | Anti-IL-23 antibodies |
| US9505837B2 (en) | 2005-06-30 | 2016-11-29 | Janssen Biotech, Inc. | Anti-IL-23 antibodies |
| CN101252951B (en) * | 2005-06-30 | 2011-12-21 | 森托科尔公司 | Anti-IL-23 antibodies, compositions, methods and uses |
| EP1910414A4 (en) * | 2005-06-30 | 2009-02-11 | Archemix Corp | Polynucleotides and polypeptides of the il-12 family of cytokines |
| EP2452694A1 (en) * | 2005-06-30 | 2012-05-16 | Janssen Biotech, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| AU2012258409B2 (en) * | 2005-06-30 | 2016-01-07 | Centocor, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| US7491391B2 (en) | 2005-06-30 | 2009-02-17 | Centocor, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| EP3501537A1 (en) * | 2005-06-30 | 2019-06-26 | Janssen Biotech, Inc. | Anti-il23 antibodies, compositions, methods and uses |
| US9127058B2 (en) | 2005-06-30 | 2015-09-08 | Janssen Biotech, Inc. | Anti-IL-23 Antibodies |
| US9096667B2 (en) | 2005-06-30 | 2015-08-04 | Janssen Biotech, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| WO2007005955A2 (en) | 2005-06-30 | 2007-01-11 | Centocor, Inc. | Anti-il-23 antibodies, compositions, methods and uses |
| US11185584B2 (en) | 2005-06-30 | 2021-11-30 | Janssen Biotech, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| US7807414B2 (en) | 2005-06-30 | 2010-10-05 | Centocor Ortho Biotech Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| AU2006265002B2 (en) * | 2005-06-30 | 2012-09-20 | Centocor, Inc. | Anti-IL-23 antibodies, compositions, methods and uses |
| US8574579B2 (en) | 2005-06-30 | 2013-11-05 | Janssen Biotech, Inc. | Anti-IL-23 antibodies |
| WO2007024846A2 (en) | 2005-08-25 | 2007-03-01 | Eli Lilly And Company | Anit-il-23 antibiodies |
| AU2006283194B2 (en) * | 2005-08-25 | 2010-10-21 | Eli Lilly And Company | Anti-IL-23 Antibodies |
| EP1937721B1 (en) * | 2005-08-25 | 2010-07-28 | Eli Lilly And Company | Anti-il-23 antibodies |
| AU2006283194B8 (en) * | 2005-08-25 | 2010-10-28 | Eli Lilly And Company | Anti-IL-23 Antibodies |
| JP2009506041A (en) * | 2005-08-25 | 2009-02-12 | イーライ リリー アンド カンパニー | Anti-IL-23 antibody |
| AU2006283194B9 (en) * | 2005-08-25 | 2011-02-03 | Eli Lilly And Company | Anti-IL-23 Antibodies |
| EP3190125A1 (en) | 2005-08-31 | 2017-07-12 | Merck Sharp & Dohme Corp. | Engineered anti-il-23 antibodies |
| JP2009505677A (en) * | 2005-08-31 | 2009-02-12 | シェーリング コーポレイション | Modified anti-IL-23 antibody |
| WO2007027714A3 (en) * | 2005-08-31 | 2007-06-21 | Schering Corp | Engineered anti-il-23 antibodies |
| EP2354160A1 (en) | 2005-08-31 | 2011-08-10 | Schering Corporation | Engineered anti-IL-23-antibodies |
| US7807160B2 (en) | 2005-08-31 | 2010-10-05 | Schering Corporation | Engineered anti-IL-23 antibodies |
| US8362212B2 (en) | 2005-08-31 | 2013-01-29 | Merck Sharp & Dohme Corp. | Engineered anti-IL-23 antibodies |
| EP1931710B1 (en) | 2005-08-31 | 2017-01-18 | Merck Sharp & Dohme Corp. | Engineered anti-il-23 antibodies |
| WO2007027714A2 (en) | 2005-08-31 | 2007-03-08 | Schering Corporation | Engineered anti-il-23 antibodies |
| US8524230B2 (en) | 2005-09-01 | 2013-09-03 | Merck Sharp & Dohme Corp. | Use of IL-23 and IL-17 antagonists to treat autoimmune ocular inflammatory disease |
| US10954297B2 (en) | 2005-12-29 | 2021-03-23 | Janssen Biotech, Inc. | Methods of treatment using human anti-IL-23 antibodies |
| US8106177B2 (en) | 2005-12-29 | 2012-01-31 | Janssen Biotech, Inc. | Nucleic acids encoding human anti-IL-23 antibodies |
| US7993645B2 (en) | 2005-12-29 | 2011-08-09 | Janssen Biotech, Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| US9783607B2 (en) | 2005-12-29 | 2017-10-10 | Janssen Biotech, Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| US8221760B2 (en) | 2005-12-29 | 2012-07-17 | Janssen Biotech, Inc. | Methods of treatment using human anti-IL-23 antibodies |
| US10030070B2 (en) | 2005-12-29 | 2018-07-24 | Janssen Biotech, Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| US9353181B2 (en) | 2005-12-29 | 2016-05-31 | Janssen Biotech, Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| US7935344B2 (en) | 2005-12-29 | 2011-05-03 | Centocor Ortho Biotech Inc. | Human anti-IL-23 antibodies, compositions, methods and uses |
| EP3219328B1 (en) | 2005-12-29 | 2020-06-17 | Janssen Biotech, Inc. | Human anti-il-23 antibodies, compositions, method and uses |
| US8496936B2 (en) | 2006-03-10 | 2013-07-30 | Zymogenetics, Inc. | Antagonists of IL-17A, IL-17F, and IL-23P19 |
| US8333968B2 (en) | 2006-03-10 | 2012-12-18 | Zymogenetics, Inc. | Methods of inhibiting inflammation with antagonists to IL-17A, IL-17F, and IL-23P19 |
| US9994634B2 (en) | 2006-03-10 | 2018-06-12 | Zymogenetics, Inc. | Use of an IL-17/IL-23 bispecific antibody for treating inflammation |
| US9464134B2 (en) | 2006-03-10 | 2016-10-11 | Zymogenetics, Inc. | Polynucleotides encoding antagonists of IL-17A, IL-17F, and IL-23p19 |
| US8992922B2 (en) | 2006-03-10 | 2015-03-31 | Zymogenetics, Inc. | Antagonists of IL-17A, IL-17F, and IL-23P19 |
| US7910703B2 (en) | 2006-03-10 | 2011-03-22 | Zymogenetics, Inc. | Antagonists to IL-17A, IL-17F, and IL-23P19 and methods of use |
| US10562967B2 (en) | 2006-03-10 | 2020-02-18 | Zymogenetics, Inc. | Treating inflammation with IL-17/IL-23 bispecific antibodies |
| US8227579B2 (en) | 2006-06-13 | 2012-07-24 | Zymogenetics, Inc. | IL-23 antagonists |
| US7790862B2 (en) | 2006-06-13 | 2010-09-07 | Zymogenetics, Inc. | IL-17 and IL-23 antagonists and methods of using the same |
| EP1882699A1 (en) * | 2006-07-27 | 2008-01-30 | Heinfried Radeke | Regulation of interleukin-23 |
| WO2008012045A2 (en) * | 2006-07-27 | 2008-01-31 | Schleussner, Hans | Regulation of interleukin-23 |
| WO2008012045A3 (en) * | 2006-07-27 | 2008-04-03 | Heinfried Radeke | Regulation of interleukin-23 |
| US8404813B2 (en) | 2007-02-23 | 2013-03-26 | Merck Sharp & Dohme Corp. | Engineered anti-IL-23P19 antibodies |
| EP2426144A1 (en) | 2007-02-23 | 2012-03-07 | Schering Corporation | Engineered anti-IL-23p19 antibodies |
| US8513389B2 (en) | 2007-02-23 | 2013-08-20 | Merck Sharp & Dohme Corp. | Engineered anti-IL-23p19 antibodies |
| EP2426145A1 (en) | 2007-02-23 | 2012-03-07 | Schering Corporation | Engineered anti-IL-23p19 antibodies |
| US9809648B2 (en) | 2007-02-23 | 2017-11-07 | Merck Sharp & Dohme Corp. | Engineered anti-IL-23p19 antibodies |
| US8293883B2 (en) | 2007-02-23 | 2012-10-23 | Schering Corporation | Engineered anti-IL-23P19 antibodies |
| US8309085B2 (en) | 2007-02-28 | 2012-11-13 | Merck Sharp & Dohme Corp. | Engineered anti-IL-23R antibodies |
| US8691532B2 (en) | 2007-02-28 | 2014-04-08 | Merck Sharp & Dohme Corp. | Nucleic acids encoding engineered anti-IL-23R antibodies |
| WO2008106131A2 (en) | 2007-02-28 | 2008-09-04 | Schering Corporation | Combination therapy for treatment of immune disorders |
| US8119133B2 (en) | 2007-02-28 | 2012-02-21 | Schering Corporation | Engineered anti-IL-23R antibodies |
| WO2008106134A2 (en) | 2007-02-28 | 2008-09-04 | Schering Corporation | Engineered anti-il-23r antibodies |
| EP2417974A1 (en) | 2007-02-28 | 2012-02-15 | Schering Corporation | Combination therapy for treatment of immune disorders |
| EP2395025A1 (en) | 2007-02-28 | 2011-12-14 | Schering Corporation | Engineered Anti-IL-23R Antibodies |
| WO2009007849A3 (en) * | 2007-07-06 | 2011-01-06 | Valorisation Hsj, Societe En Commandite | Il-23 receptor antagonists and uses thereof |
| WO2010027767A1 (en) * | 2008-08-27 | 2010-03-11 | Schering Corporation | Engineered anti-il-23r antibodies |
| WO2010046530A1 (en) * | 2008-10-20 | 2010-04-29 | Helsingin Yliopiston Rahastot | Methods and uses involving genetic aberrations of nav3 and aberrant expression of multiple genes |
| WO2010115786A1 (en) * | 2009-04-01 | 2010-10-14 | Glaxo Group Limited | Anti-il-23 immunoglobulins |
| EP2456787A4 (en) * | 2009-07-24 | 2013-01-30 | Univ Leland Stanford Junior | Cytokine compositions and methods of use thereof |
| EP2456787A2 (en) * | 2009-07-24 | 2012-05-30 | The Board of Trustees of The Leland Stanford Junior University | Cytokine compositions and methods of use thereof |
| US9487580B2 (en) | 2009-10-26 | 2016-11-08 | Amgen Inc. | Human IL-23 antigen binding proteins |
| US8722033B2 (en) | 2009-10-26 | 2014-05-13 | Amgen Inc. | Human IL-23 antigen binding proteins |
| US9951129B2 (en) | 2009-10-26 | 2018-04-24 | Amgen Inc. | Human IL-23 antigen binding proteins |
| US9441036B2 (en) | 2010-11-04 | 2016-09-13 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| US10202448B2 (en) | 2010-11-04 | 2019-02-12 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| US8778346B2 (en) | 2010-11-04 | 2014-07-15 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| US11078265B2 (en) | 2012-05-03 | 2021-08-03 | Boehringer Ingelheim International Gmbh | Anti-IL-23 antibodies |
| WO2014004436A2 (en) | 2012-06-27 | 2014-01-03 | Merck Sharp & Dohme Corp. | Crystalline anti-human il-23 antibodies |
| US9803010B2 (en) | 2012-06-27 | 2017-10-31 | Merck Sharp & Dohme Corp. | Crystalline anti-human IL-23p19 antibodies |
| WO2014093203A1 (en) | 2012-12-13 | 2014-06-19 | Merck Sharp & Dohme Corp. | SOLUTION FORMULATIONS OF ENGINEERED ANTI-IL-23p19 ANTIBODIES |
| US9023358B2 (en) | 2013-03-08 | 2015-05-05 | Eli Lilly And Company | Antibodies that bind to IL-23 |
| US9688753B2 (en) | 2013-03-08 | 2017-06-27 | Eli Lilly And Company | Nucleic acids encoding antibodies that bind IL-23 |
| US10507241B2 (en) | 2014-07-24 | 2019-12-17 | Boehringer Ingelheim International Gmbh | Biomarkers useful in the treatment of IL-23A related diseases |
| US10793629B2 (en) | 2014-09-03 | 2020-10-06 | Boehringer Ingelheim International Gmbh | Compound targeting IL-23A and TNF-alpha and uses thereof |
| US10059763B2 (en) | 2014-09-03 | 2018-08-28 | Boehringer Ingelheim International Gmbh | Compound targeting IL-23A and TNF-alpha and uses thereof |
| US11680096B2 (en) | 2014-09-03 | 2023-06-20 | Boehringer Ingelheim International Gmbh | Compound targeting IL-23A and TNF-alpha and uses thereof |
| EP3268028A4 (en) * | 2015-03-11 | 2018-09-05 | Rush University Medical Center | Compositions and methods for treating cancer |
| US11279754B2 (en) | 2015-03-11 | 2022-03-22 | Rush University Medical Center | Compositions and methods for treating cancer |
| US11633476B2 (en) | 2017-05-02 | 2023-04-25 | Merck Sharp & Dohme Llc | Stable formulations of programmed death receptor 1 (PD-1) antibodies and methods of use thereof |
| US11845798B2 (en) | 2017-05-02 | 2023-12-19 | Merck Sharp & Dohme Llc | Formulations of anti-LAG3 antibodies and co-formulations of anti-LAG3 antibodies and anti-PD-1 antibodies |
| US11548941B2 (en) | 2018-11-20 | 2023-01-10 | Janssen Biotech, Inc. | Safe and effective method of treating psoriasis with anti-IL-23 specific antibody |
| US11780911B2 (en) | 2019-05-23 | 2023-10-10 | Janssen Biotech, Inc. | Method of treating inflammatory bowel disease with a combination therapy of antibodies to IL-23 and TNF alpha |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7282204B2 (en) | Uses of IL-23 agonists and antagonists; related reagents | |
| US20100143357A1 (en) | Uses of Mammalian Cytokine; Related Reagents | |
| US9970944B2 (en) | Methods of modulating cytokine activity; related reagents | |
| US7820168B2 (en) | Treatment of diabetes using antibodies to IL-23, IL-23 receptor and IL-17 | |
| US20050214296A1 (en) | Methods of modulating cytokine activity; related reagents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BW BY BZ CA CH CN CO CR CU CZ DE DK DM DZ EC EE EG ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NA NI NO NZ OM PG PH PL PT RO RU SC SD SE SG SK SL SY TJ TM TN TR TT TZ UA UG US UZ VC VN YU ZA ZM ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): BW GH GM KE LS MW MZ SD SL SZ TZ UG ZM ZW AM AZ BY KG KZ MD RU TJ TM AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HU IE IT LU MC NL PL PT RO SE SI SK TR BF BJ CF CG CI CM GA GN GQ GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DPEN | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed from 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 541898 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1-2005-501535 Country of ref document: PH |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2005/06879 Country of ref document: ZA Ref document number: 200506879 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2006507006 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2518262 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004219625 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2005/009717 Country of ref document: MX Ref document number: 20048067148 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2004718824 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2004219625 Country of ref document: AU Date of ref document: 20040309 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004219625 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2004718824 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: PI0408247 Country of ref document: BR |