Antibacterial Cyano-(Substituted)-Methylenepiperidinophenyl Oxazolidinones Targeting Multiple
Ribonucleoprotein Sites
Field of the Invention
The present invention relates to the field of cyano-(substituted) methylenepiperidinophenyl oxazolidinones having antibacterial activity against Gram-positive and Gram-negative bacteria. While not being bound to any theory, it is thought that the antibacterial activity is based on the their ability to inhibit bacterial ribonucleoprotein through differential binding at single/multiple ribonucleoprotein sites. The invention also relates to processes for making the compounds, to pharmaceutical compositions containing the compounds and to methods of using the compounds including treating bacterial infections with the compounds.
Background of the Invention
Oxazolidinones represent a novel chemical class of synthetic antimicrobial agents. Following a chequered historical development since about the early-1980s, a watershed event took place with the clinical 'development and release for medical use in the late 2000s of the first representative, Linezolid, of this class1'2 This advance enabled the profiling of the unique properties of the members of this class, which is that they display activity against important Gram-positive human and veterinary pathogens including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin resistant enterococci (VRE) and β-lactam resistant Streptococcus pneumoniae (PRSP). The oxazolidinones also show activity against Gram-negative aerobic bacteria and Gram- positive and Gram-negative anaerobes .
The deficiencies of this class of oxazolidinones have also surfaced. They are inactive against Enterobacteriaceae4. They are generally bacteriostatic and do not display activity at a useful level against aerobic fastidious Gram-negative pathogens, as well as Gram-negative anaerobes. Moreover their potency for atypical respiratory pathogens such as Mycoplasma pneumoniae, M. hominis, Ureaplasma Urealyticium and Chlamydia species is of a borderline range which could result into unacceptable clinical efficacy for the treatment of respiratory tract infections3.
Other limitations that have appeared through the clinical development studies and use of Linezolid and its potential successors in development are that the class has a propensity to induce myelosuppression with consequent thrombocytopenia5. Inhibition of monoa ine oxidase by oxazolidinones has prompted a recommendation made to clinicians that clinical use of members of this class be done with caution during concomitant usage of adrenergic or serotonergic agents and selective serotonin reuptake inhibitors6.
Linezolid is shown to have two targets in cells for its inhibitory effects. It binds to the 50S subunit within domain V of the 23S or RNA peptidyl fransferase center near the interface with the 30S subunit, thereby blocking the formation of the tMet-tRNA-ribosome-mRNA ternary complex. In addition, linezolid associates with the nascent 50S particle and stops the assembly process7.
Considering that the oxazolidinones are bacteriostatic, as indeed are most other agents that inhibit bacterial protein synthesis, there is a strong likelihood that resistance can emerge under selective pressure during therapy, specially for infections which require a bactericidal therapy to be used. The significant concern related to this class of antibacterials is attributed to this essentially bacteriostatic effect against their prime target pathogens such as staphylococci, enterococci and pneumococci. It is pertinent to quote from the Adis R&D insight report (Document 013296 dated December 27, 2001) that an oxazolidinone AZD 2563 under clinical development is described to be "ineffective against linezolid-resistant S. pneumoniae". This concern is further aggravated due to the recent reports of emergence of Linezolid-resistant strains of enterococci and staphylococci in clinics, h fact the first clinical isolates of E. faecium, E.faecalis and S. aureus resistant to linezolid have recently been described8. Also, resistant strains have been generated by serial passage techniques, the resistance being associated with specific mutations in the 23 S rRNA gene9.
Our own studies have also led to the identification of novel Linezolid-resistant strains, an embodiment of this invention. It has been reported that in- vitro staphylococci and enterococci resistant to linezolid can be selected only with difficulty8, which through genome characterization studies have shown the resistance to be associated with specific mutations in the 23 S rRNA nucleotide sequence. The linezolid-resistant strain of S. pneumoniae ATCC 6303 LR has guanine replacing adenine at the nucleotide position 2160 of 23 S rRNA. Similarly, our Linezolid- resistant
strains of S. aureus Smith & MRSA 032 have uracil replacing guanine at nucleotide position 2447. These three resistant mutants, harbouring changes in the molecular targets of linezolid, showed significant elevation of MIC values for Linezolid indicating the loss in affinity of the drug to its ribosomal targets. "Fine tuning" of this class of agents to improve the affinity of its members for the ribosome at existing or altered single or multiple target sites is conceivable, resulting thereby in significantly increasing their potency, and in incorporating bactericidal activity against Linezolid-sensitive/- resistant strains.
The present invention describes a novel series of oxazolidinones which display increased potency, and incorporate bactericidal activity, in contrast to the earlier-described bacteriostatic activity, against Linezolid-sensitive/-resistant strains, thus indicating a differential binding at the conventional site/s of the ribonucleoprotein and/or targeting multiple such receptor sites. In addition, using comparative molecular field analysis10, a study of literature-described oxazolidinones and the novel compounds of the present invention has enabled the identification of newer / additional structural motifs of the oxazolidinone class, novel and non-obvious from the prior art, which support the activity against the Linezolid-sensitive/-resistant pathogens. There is no prior description of oxazolidinones displaying such bactericidal activity or useful activity against Linezolid-sensitive/-resistant or other oxazolidinone-resistant microbial pathogens.
The following publications may be referred to with respect to the statements made in the above- described background information.
1 Slee AM, et al, Antimicrob. Agents Chemother (1987) 31:1791 -1797;
2 2nd European Congress of Chemotherapy and 7th Biennial Conference on Antiinfective Agents and Chemotherapy (Final Program), (1998): 93;
3Diekema D J et al, Lancet 2001; 358: 1975-82;
4Zhanel GG et al., Canadian Journal of Infectious Diseases, 2001, 12: 379-390; 5Kuter D J et al., Pharmacotherapy, 2001: 21: 1010-1030; 6 Ament P W et al, Am Fam Physician 2002, 65: 663-70; 7 Shinabarger D, Exp. Opin. Invest. Drugs (1999) 8: 1195-1202; Champrey W S et al., Curr. Microb. 2002, 44: 350-356;
8Zurenko GE et al, hi 39th Interscience Conference on Antimicrobial Agents and Chemotherapy, Washington DC, (1999) abstr. 848; Gonzales RD et al., Lancet 2001; 357:1179; Tsiodras S, et al, Lancet 2001; 358: 207-08;
9 Swaney SM et al., hi 38th Interscience Conference on Antimicrobial Agents and Chemotherapy, Washington DC, (1998) abstr. C-l 04;
10 Pae, A. N. et al, Bioorg. & Med. Chem. Lett., 1999, 9:2685-90.
After filing of our pending provisional US application No. 60/395,164 methylenepiperidinyl and methylenepyrrolidinyl oxazolidinone antibacterial agents were described in π Kim H Y et al., Bioorg. & Med. Chem. Lett., (2003), 13:2227-2230.
Information Disclosure
There are several patents cited in the literature, which refer to oxazolidinones having antibacterial activity.
WO95/25106 dated September 21, 1995 discloses substituted piperidino phenyloxazolidinones. This corresponds to US Patent 5,668,286 and EP 0 750 618.
WO96/13502 dated May 9, 1996 discloses phenyloxazolidinones having a multisubstituted azetidinyl or pyrrolidinyl moiety. US 5,574,055 dated November 12, 1996 discloses oxazolidinone derivatives that can be used for prevention or control of depressive, panic and anxiety states.
Other publications are as follows:
WO 99/24428 dated May 20, 1999 discloses diazepenophenyloxazolidinone derivatives. WOO 1/44212 dated June 21, 2001 discloses benzoic acid esters of oxazolidinones having a hydroxyacetylpiperazine substituent.
WO 02/06278 dated Jan 24, 2002 discloses substituted aminopiperidino phenyloxazolidinone derivatives.
U.S. Patent 6,358,942 dated March 19, 2002 discloses phenyloxazolidinones having a C-C bond to 4-8 membered heterocyclic rings.
WO 00/21960 dated April 20, 2000 discloses heterocyclicphenyl oxazolidinones having the heterocycle linked through a carbon atom to the phenyl moiety.
WO 95/07271 (U.S. Patent 5,688,792 dated Nov. 18, 1997) discloses oxazolidinones containing moφholine and tliiomorpholine.
The following references disclose various oxazolidinones, which have a thiocarbonyl functionality.
U.S. 6,387,896 dated May 14, 2002.
WO 98/54161 dated Dec.3, 1998 (U.S. 2002/0016323A1 Feb 7, 2002);
WO 00/27830 dated May 18, 2000.
WO 01/09107 dated Feb. 8, 2001.
The following citations pertain to oxazolidinones some of which have a cyano substituent and others of which have heterocyclic moieties incorporated in the described molecules.
U.S. Patent 5,977,373 dated Nov. 2, 1999 (WO 99/02525 dated July 8, 1998) discloses thiadiazolyl and oxadiazolyl phenyloxazolidinones. U.S. Patent 5,910,504 dated June 8, 1999 (WO 96/23788 dated Aug 8, 1996) discloses heteroaromatic ring substituted phenyloxazolidinones.
U.S. Patent 5,547,950 dated Aug. 20, 1996 and U.S. Patent 5,700,799 dated Dec 23, 1997 [WO 93/23384] disclose oxazolidinone antimicrobials containing substituted diazine moieties.
Genin M. J. et al J.Med.Chem 2000, 43,953-970 Weidner- Wells et al. Biorganic and Medicinal Chemistry Letters 2001, 11, 1829-1832.
WO 01/58885A1 dated August 16, 2001 discloses oxazolidinone thioamides with piperazine amide substituents.
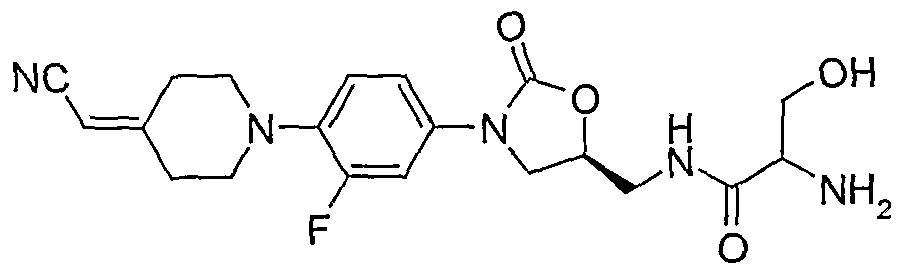
Ryan B. et al., Exp. Opin. Invest. Drugs (2000) 9: 2959 - 60 discloses an oxazolidinone that is active against linezolid-resistant S.aureus.
The compounds of the present invention are novel, none of them having being previously reported in the literature. They are non-obvious over the compounds in the prior art by virtue of their being bactericidal, in contrast to the compounds of the prior art being generally bacteriostatic. They are active against linezolid-resistant strains, in particular against novel linezolid-resistant strains of this invention, in further particular against linezolid-resistant Streptococcus pneumoniae and against resistant enterococci, such activity features being disclosed here for the first time. There is no previous report of oxazolidinones of the structure presented in this invention which display
activity against difficult-to-obtain linezolid-resistant strains. While not being bound by any theory, it is surmised by displaying such activity against linezolid-resistant sfrains, the compounds of the invention for the first time thus establish their ability to inhibit bacterial ribonucleoprotein through differential binding at single/ multiple sites.
In addition, a Comparative Molecular Field Analysis (CoMFA), 3 -dimensional quantitative structure activity relationship study, as described in more detail later in this specification, shows that in contrast to the prior art, which teaches that the elecfrostatic contributions play a more predominant role than the steric contributions, the compounds of the present invention require a comparatively higher steric contribution, more than one and a half times over the electrostatic contributions.
Summary of the Invention
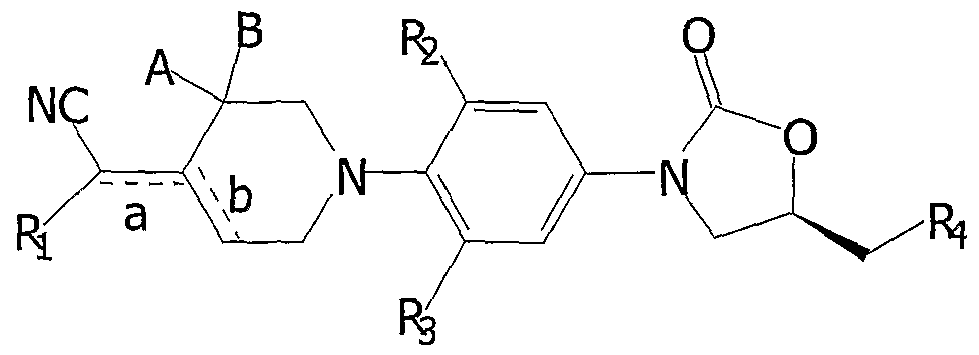
The present invention provides new compounds of the Formula I.
Formula I
or pharmaceutical acceptable salts thereof, wherein
"a" represents a single bond or a double bond "b" represents a single bond or a double bond "a" and "b" cannot both be double bonds at the same time.
"A" and "B' are each and independently selected from H, Cι-C6 alkyl, CO2Et, or halogen.
When "a" is a double bond,
H, alkyl, substituted alkyl, alkenyl, alkynyl, cycloalkyl, substituted cycloalkyl, alkanoyl, substituted alkanoyl, aralkanoyl, substituted aralkanoyl, alkoxycarbonyl, substituted alkoxycarbonyl, thioacyl, substituted thioacyl, aroyl, substituted aroyl, alkylmercapto, arylmercapto, heterocyclylcarbonyl, heterocyclylthiocarbonyl, aralkyl, aryl, substituted aryl, heterocyclyl, substituted herocyclyl, heteroaryl, substituted heteroaryl, cyano, carboxylic acid, carboxamido, amino, substituted arnino, or halogen.
When "a" is a single bond and "A" is H, CH3,CO2Et, F, then; Ri is alkenyl, alkynyl, cycloalkyl, substituted cycloalkyl, alkanoyl, substituted alkanoyl, aralkanoyl, substituted aralkanoyl, alkoxycarbonyl, substituted alkoxycarbonyl, thioacyl, substituted thioacyl, aroyl, substituted aroyl, alkylmercapto, arylmercapto, heterocyclylcarbonyl, heterocyclylthiocarbonyl, aralkyl, aryl, substituted aryl, heterocyclyl, substituted herocyclyl, heteroaryl, substituted heteroaryl or carboxamido.
When "a" is a single bond and "A" is not H, CH3, CO Et, or F, then, Ri is defined as when "a" is double bond.
R and R3 are the same or different and are hydrogen or halo; R-t is,
Cι-C6 alkylsulphonyloxy, arylsulphonyloxy, amino, mono or di substituted amino, azido, nitrilo, substituted nitrilo, aminonitrilo, isocynato, formamido, Cι-C6 alkyl amido, substituted Cι-C6 alkyl amido, Cι-C6 alkyl thiocarbonylamino, substituted Cι-C6 alkyl thiocarbonylarnino, sulphonamido, substituted sulphonamido, pthalamido, carbamato, substituted carbamato, ureido, substituited ureido, five to six membered heterocyclyl, or substituted five to six membered heterocyclyl. In another aspect, the present invention also provides: a pharmaceutical composition comprising a compound of Formula I or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable carrier, a method for treating Gram-positive microbial infections in human or other warm-blooded animals by administering to the subject in need thereof a therapeutically effective amount of a compound of Formula I or a pharmaceutically acceptable salt thereof, a method for treating Gram-negative microbial infections in human or other warm-blooded animals by administering to the subject in need a therapeutically effective amount of a compound of Formula I or a pharmaceutically acceptable salt thereof,
The invention also includes novel intermediates and processes that are used to prepare compounds of Formula I.
Detailed Description of the Invention
The present invention provides new compounds of the Formula I.
Formula I
or pharmaceutical acceptable salts thereof, wherein
"a" represents a single bond or a double bond
"b" represents a single bond or a double bond a" and "b" cannot both be double bonds at the same time.
"A" and "B' are each and independently selected from H, Cι-C6 alkyl, CO2Et, or halogen.
When "a" is a double bond,
H, alkyl, substituted alkyl, alkenyl, alkynyl, cycloalkyl, substituted cycloalkyl, alkanoyl, substituted alkanoyl, aralkanoyl, substituted aralkanoyl, alkoxycarbonyl, substituted alkoxycarbonyl, thioacyl, substituted thioacyl, aroyl, substituted aroyl, alkylmercapto, arylmercapto, heterocyclylcarbonyl, heterocyclylthiocarbonyl, aralkyl, aryl, substituted aryl, heterocyclyl, substituted herocyclyl, heteroaryl, substituted heteroaryl, cyano, carboxylic acid, carboxamido, amino, substituted amino, or halogen.
When "a" is a single bond and "A" is H, CH3,CO2Et, F, then; Ri is alkenyl, alkynyl, cycloalkyl, substituted cycloalkyl, alkanoyl, substituted alkanoyl, aralkanoyl, substituted aralkanoyl, alkoxycarbonyl, substituted alkoxycarbonyl, thioacyl, substituted thioacyl, aroyl, substituted
aroyl, alkylmercapto, arylmercapto, heterocyclylcarbonyl, heterocyclylthiocarbonyl, aralkyl, aryl, substituted aryl, heterocyclyl, substituted herocyclyl, heteroaryl, substituted heteroaryl or carboxamido.
When "a" is a single bond and "A" is not H, CH3, CO2Et, or F, then, Ri is defined as when "a" is double bond.
R and R3 are the same or different and are hydrogen or halo;
Rt is,
Ci-Cδ alkylsulphonyloxy, arylsulphonyloxy, amino, mono or di substituted amino, azido, nitrilo, substituted nitrilo, aminonitrilo, isocynato, formamido, Cι-C6 alkyl amido, substituted Cι-C6 alkyl amido, Cι-C6 alkyl thiocarbonylamino, substituted Cι-C6 alkyl thiocarbonylamino, sulphonamido, substituted sulphonamido, pthalamido, carbamato, substituted carbamato, ureido, substituited ureido, five to six membered heterocyclyl, or substituted five to six membered heterocyclyl.
"Alkyl" means carbon atom chains having Cι-C6 number of carbon atoms which can be either sfraight chain or branched such as methyl, ethyl, propyl, butyl, pentyl, or hexyl.
"Substituted alkyl" means Cι-C6 alkyl, sfraight chain or branched, bearing substituents like one or more aryl, hydroxy, substituted hydroxy for example methanesulphonyloxy, heterocyclyl, substituted heterocycyl, cyano, halo, for example fluorine or chlorine, amino, substituted amino;
"Alkenyl" means carbon atom chains having C2-C6 number of carbon atoms which can be either straight chain or branched such as ethene, propene, butene, pentene, hexene, butadiene, or hexadiene.
"Alkynyl" means carbon atom chains having C2-C6 number of carbon atoms which can be either sfraight chain or branched such as ethyne, propyne, butyne, pentyne, hexyne, butadiyne, or hexadiyne.
"Cycloalkyl" means C3-C6 carbocycles such as cyclopropyl, cyclobutyl, cyclopentyl, or cyclohexyl.
" Substituted cycloalkyl" means cycloalkyl substituted with a groups such as alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano or halogen;
"Alkanoyl" means Cι-C6 number of carbon atoms to form an organic acid where the OH group has been deleted, such as formyl, HCO-; acetyl, or CH3CO-.
"Substituted alkanoyl" means alkanoyl bearing substitutents like one or more alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano, or halogen.
"Aralkanoyl" means Cι-C6 number of carbon atoms to form an aralkyl organic acid where the OH group has been deleted, such as phenylacetyl, C6H5CH2CO-; "Substituted aralkanoyl" means aralkanoyl bearing substitutents like one or more alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano, or halogen.
"Alkoxycarbonyl" means alkanoyl group substituted with alkyl ether such as methoxy, ethoxy, propyloxy so on;
"Substituted alkoxycarbonyl" means alkoxycarbonyl bearing substitutents like one or more alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano,or halogen;
"thioacyl" means Cι-C6 number of carbon atoms to form an thioorganic acid where the OH group has been deleted, such as thioformyl, HCS-; thioacetyl, CH3CS-;
"substituted thioacyl" means thioacyl bearing substitutents like one or more alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano, halogen; "aroyl" means Cι-C6 number of carbon atoms to form an aryl organic acid where the OH group has been deleted, such as benzoyl, C6H5CO-;
"substituted aroyl" means alkanoyl bearing substitutents like one or more alkyl, hydroxyl, amino, substituted amino, alkoxycarbonyl, carboxamido, cyano, halogen;
"alkylmercapto" means alkylthiol in which H group is deleted such as CH3S-, C2H5S-SO on; "arylmercapto" means arylthiol in which H group is deleted such as C6H5S- so on;
"heterocyclylcarbonyl" means groups such as carbonyl bearing heterocycles like morpholine, piperidine, piperazine and so on;
"heterocyclylthiocarbonyl" means groups such as thiocarbonyl bearing heterocycles like morpholine, piperidine, piperazine and so on; "aralkyl" are groups such as benzyl, benzhydryl, trityl and so on;
"aryl" stands for phenyl, naphthyl, so on;
"substituted aryl" stands for aryl which may optionally be substituted with groups such as like one or more alkyl, hydroxyl, a ino, substituted amino, alkoxycarbonyl, carboxamido, cyano, halogen;
"heterocyclyl" means groups such as heterocycles like morpholine, piperidine, piperazine and so on;
"substituted heterocyclyl" stands for herterocyclyl which may optionally be substituted with groups such as like one or more alkyl, alkoxycarbonyl, carboxamido, cyano, halogen;
"heteroaryl" means groups such as heterocycles like pyrrole, fiirane, thiophene, pyrazole, imidazole, trizole, tefrazole, thiazole, pyridine, pyrimidine, and so on;
"substituted heteroaryl" stands for herteroaryl which may optionally be substituted with groups such as like one or more alkyl, alkoxycarbonyl, carboxamido, cyano, halogen; "cyano" is -CN; carboxamido" is -CONH2;
"substituted amino" stands for NH2, in which one or more hydrogen atoms may be optionally substituted by Cι-C3 alkyl groups also unsubstituted or optionally substituted by substituents as defined earlier in the specification under "substituted alkyl"; "halogen" means atoms such as fluorine, chlorine, bromine, iodine.
and pharmaceutically acceptable salts thereof including isomers, polymorphs or pharmaceutical acceptable salts thereof.
Preferred salts are those of hydrochloride, hydrobronide, hydroiodide, sulphate, phosphate and salts of organic acids such as acetate, lactate, succmate, oxalate, maleate, fumarate, malate, tatrate, citrate, ascorbate, cinnamate, gluconate, benzoate, methane sulfonate and p-toluene sulfonate; lithium, sodium, magnesium, calcium and potassium salts, and amino acids salts such as alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, serine, threonine, tryptopham tyrosine or valine.
More particularly, the present invention currently provides compounds of Formula I, which can be represented as Formulae II, III and IV.
Formula II
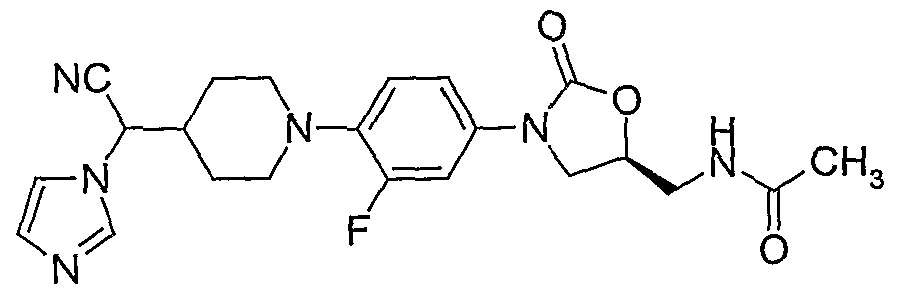
Formula IV
wherein, preferably A, B, Rj, and R2, R3 and are as defined above. 1
Preferred Compounds
Some preferred examples of the oxazolidinone derivatives represented by the general Formula I and belonging to the subclass Formula II are as follows:
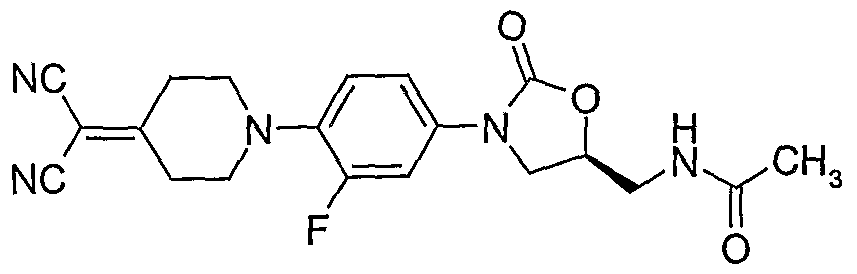
1. (S)-{3-[4-(4-cy-momethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide;
2. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- formamide;
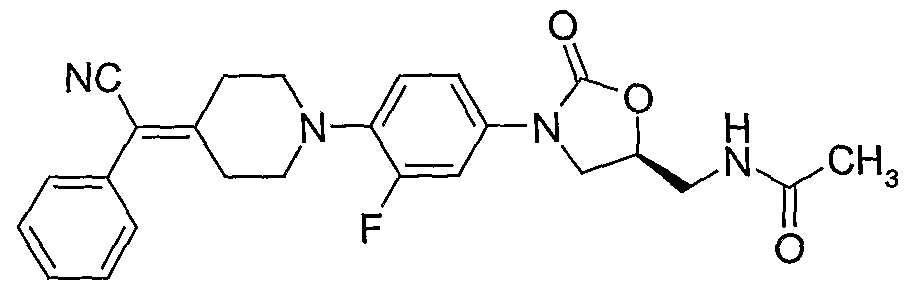
3. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide;
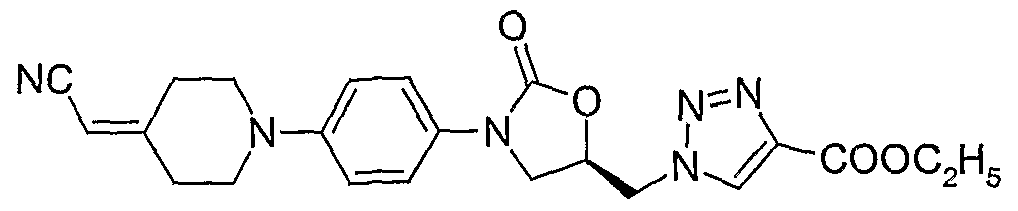
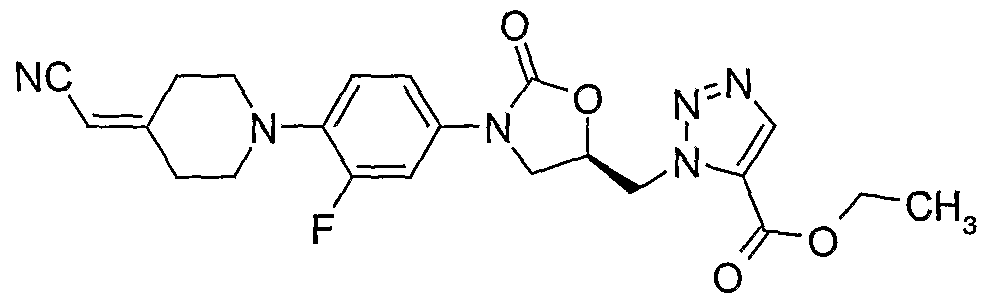
4. (S)-l-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-4- carboxyethyl- 1,2,3 -triazole;
5. (S)-l - {3-[4-(4-cyanomethylidene-piperidin-l -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} -4- cyano-1 ,2,3-triazole; 6. (R)- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - methanesulphonate;
7. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -isocynate;
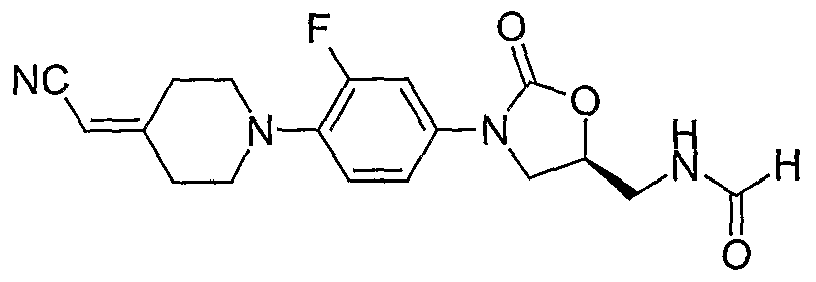
8. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl}-formamide;
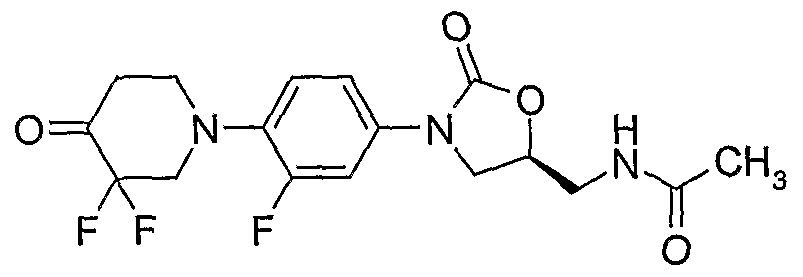
9. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -acetamide;
10. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -propionamide; 11. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl}-2-dimethylpropionamide;
12. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl}-3-dimethylbutanamide;
13. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -2-hydroxyacetamide;
14. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl}-2-pivolyloxyacetamide;
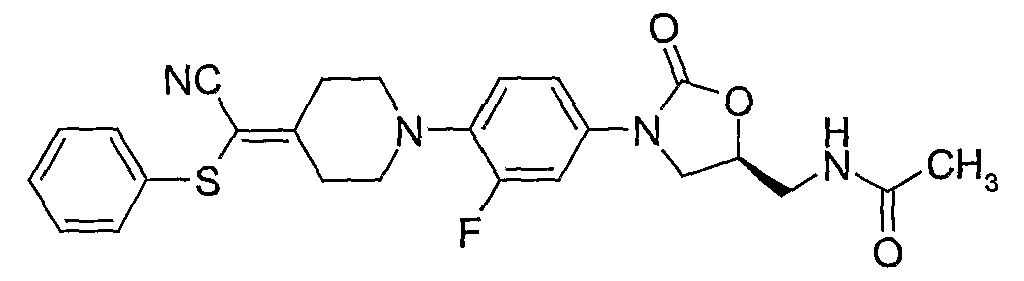
15. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl } -N-methylacetamide; 16. (S)-{3- [4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -azide;
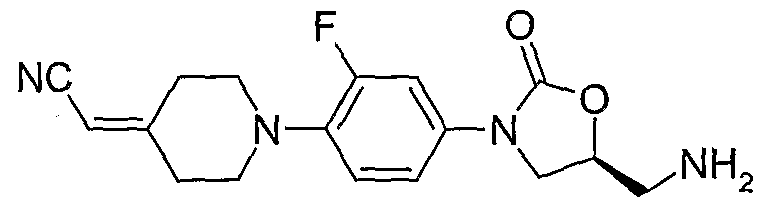
17. (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine;
18. (S)- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylarnino}-prop-2-ene;
19. (S)-{3-[4-(4-cyanomethylidene-ρiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -nitrile;
20. (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -acetonitrile;
21. (S)- {3-[4-(4-cy- omethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -methylamine; 22. (S)- {N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino}N-cyano}-prop-2-ene;
23. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-cyanoacetamide;
24. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-oxo-oxazolidin-4-yl-carboxamide;
25. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -pyrrolidin-2-carboxamide;
26. (S)-l - {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- yimethyl}-4-carboethoxy-l,2,3-triazole; 27. (S)-1- {3 -[4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -5-carboethoxy-l ,2,3-triazole; 28. (S)-l-{ 3 -[4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -4-cyano- 1 ,2,3-triazole;
29. (S)-N- { 3 - [4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -difluoroacetamide;
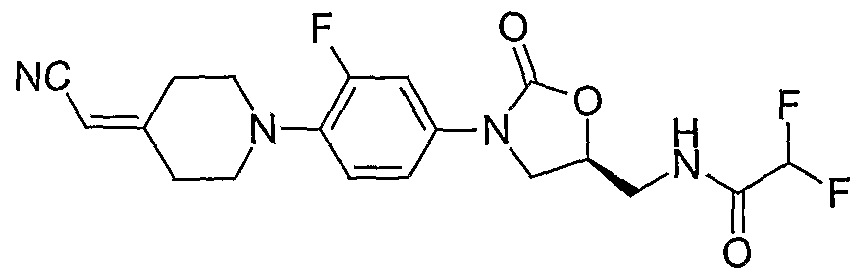
30. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -trifluoroacetamide;
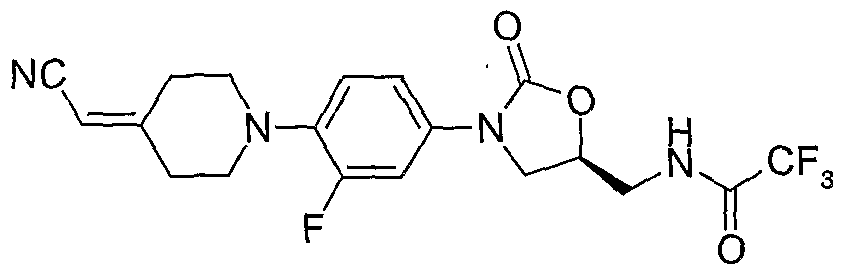
31. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -chloroacetamide; 32. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl] -dichloroacetamide;
33. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -trichloroacetamide;
34. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -bromoacetamide;
35. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -dibromoacetamide ;
36. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -iodoacetamide;
37. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluoropheiiyl]-2-oxo-oxazolidin-5- ylmethyl}-4-methylphenylsulphonamide; 38. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methylcarbamate;
39. (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluoroplιenyl]-2-oxo-oxazolidin-5- ylmethyl} -ethylcarbamate;
40. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -isopropylcarbamate;
41. (2S,5S)-{N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -propionamid-2-yl} -amine;
42. (2S, 5S)-{N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -3-hydroxypropionamid-2-yl} -amine; 43. (2S,5S)-{N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -3-(imidazol-4-yl)-propionanιid-2-yl} -amine;
44. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethy 1 } - 1 -pthalamide ;
45. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
46. (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methylthiocarbamate;
47. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-hydroxythioacetamide; 48. (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl}-2-hydroxyethylthiocarbamide;
49. (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -thiocarbonylmethylamine;
50. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-dimethylaminoethylthiocarbarnide;
51. (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thiocarbamide;
52. (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methylthiocarbamide;
53. (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -methanesulphonamide; 54. (R)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulphonate;
55. (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl } -acetamide;
56. (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
57. (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -difluoroacetamide;
58. (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -isobutylcarbamate; 59. (R)-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulphonate;
60. (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
61. (S)-N- {3-[4-(4-cyanomethylidene-3,3-difluoropiperidin-l -yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
62. (S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -formamide;
63. (S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide; 64. (S)-N-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -trifluoroacetamide;
65. (S)-N-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -2-cyanoacetamide;
66. (S)-2-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethylamino}-l,3-thiazole;
67. (S)-N-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl}-methylthiocarbamate;
68. (S)-N-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -thiocarbamide;
69. (S)-N-{3-[4-(4-cyanomethylidene-3-methylpiρeridin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -methyltl iocarbamide; 70. (S)-N-{3-[4-(4-cyanomethylidene-3,3-dimethylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
71. (R)-{3-[4-(4-cyanomethylidene-3,3-dimethylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulphonate;
72. (S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - acetamide;
73. (S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -formamide;
74. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide; 75. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-cyanoacetamide;
76. (S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyla ino } -carboxymethylamine;
77. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -difluoroacetamide;
78. (S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -chloroacetamide;
79. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichlordacetamide; 80. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -trichloroacetamide;
81. (S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -isobutylcarbamate;
82. (R)- {3-[4-(4-(l-cyanoethylidene)-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol;
83. (R)-3-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyloxy} -iso-oxazole;
84. (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
85. E/Z mixture of (S)-N-{3-[4-(4-(l-cyanoethylidene)-3-fluoropiperidin-l-yl)-3-fluoiOphenyl]-2- oxo-oχazolidin-5-ylmethyl}-acetamide; 86. E-(S)-N-{3-[4-(4-(l-cyanoethylidene)-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
87. Z-(S)-N-{3-[4-(4-(l-cyanoethylidene)-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
88. (S)-N- {3-[4-(4-( 1 -cyanopropylidene)-piperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - acetamide;
89. (S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
90. (S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichloroacetamide; 91. (S)-N-{3-[4-(4-(l-cyanopiOpylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -trichloroacetamide;
92. (S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -bromoacetamide;
93. (S)-N- {3-[4-(4-(l -cyanopropylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
94. (S)-N-{3-[4-(4-(l-cyano-cyclopropylmethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
95. (S)-N-{3-[4-(4-(l-cyano-3-ene-butylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide; 96. (S)-N-{3-[4-(4-(l-cyano-3-yne-butylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide;
97. (S)-N- { 3 - [4-(4-( 1 -cyano-2-phenyl-ethylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-acetamide;
98. (S)-N-{3-[4-(4-(l-cyano-l-phenyl-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
99. (S)-N-{3-[4-(4-(l-cyano-l-(3,4-difluorophenyl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
100. (S)-N-{3-[4-(4-(l-cy- o-l-( yridin-2-yl)-methylidene)-piρeridin-l-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide;
101. (S)-N- {3-[4-(4-( 1 -cyano-2-(morpholin- 1 -yl)-ethylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide; 102. (S)-N-{3-[4-(4-(l-cy-mo-l-(irnidazol-l-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
103. (S)-N- {3-[4-(4-( 1 -cyano- 1 -(2-methyl-imidazol- 1 -yl)-methylidene)-piperidin- 1 -yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
104. (S)-N-{3-[4-(4-(l-cyano-l-(l,2,4-triazol-l-yl)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
105. (S)-N-{3-[4-(4-(l-cyano-l-(thiophen-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
106. (S)-N- {3-[4-(4-(l , 1 -dicyano-methylidene)-piperidin- l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide; 107. (S)-N-{3-[4-(4-(l-cyano-l-carboxamido-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide;
108. (S)-N- {3-[4-(4-( 1 -cyano- 1 -(N-prop-2-ene-carboxamido)-methylidene)-piperidin- 1 -yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
109. (S)-N- {3-[4-(4-(l -cyano-1 -(N-cyclopropyl-carboxamido)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide;
110. (S)-N-{3-[4-(4-(l-cy- o-l-(N-cyclohexyl-carboxamido)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
111. (S) -N- { 3 -[4-(4-( 1 -cyano- 1 -(pyrrolidin- 1 -yl-carbonyl)-methylidene)-piperidin- 1 -yl)-3 - fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide; 112. (S)-N-{3-[4-(4-(l-cyano-l-(morpholin-l-yl-carbonyl)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
113. (S)-N-{3-[4-(4-(l-cyano-3-hydroxy-propylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
114. (S)-N- { 3 -[4-(4-( 1 -cyano- 1 -ethoxycarbonyl-methylidene)-piperidin- 1 -yl)-3 -fluorophenyl] - 2-oxo-oxazolidin-5-ylmethyl} -acetamide;
115. (S)-N-{3-[4-(4-(l-cyano-l-methylmercapto-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
116. (S)-N- {3-[4-(4-(l -cyano- 1 -phenylmercapto-methylidene)-piperidin- 1 -yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
117. (S)-N-{3-[4-(4-(l-cyano-l-bromo-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-yhnethyl}-acetamide; 118. (S)-N-{3-[4-(4-(l-cyano-l-(ρyridin-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-thioacetamide;
119. (S)-N-{3-[4-(4-(l,l-dicyano-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide;
120. (S)-N-{3-[4-(4-(l-cyano-l-ethoxycarbonyl-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylrnethyl} -thioacetamide;
121. (S)-N-{3-[4-(4-(l-cyano-l-(morpholin-l-yl-thiocarbonyl)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-thioacetamide;
Some preferred examples of the oxazolidinone derivatives represented by the general Formula I and belonging to the subclass Formula III are as follows:
122. (S)-{3-[4-(4-cyanometlιyl-piperidm-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide; 123. (S)-l-{3-[4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-4- carboethoxy- 1 ,2,3-triazole;
124. (R)-{3-[4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol;
125. (R)-{3-[4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- methanesulphonate; 126. (S)-N- {3-[4-(4-cyanomethyl-piperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - thioacetamide; 127. (S)- 1 - {3 -[4-(4-cyanomethyl-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl}-4-carboxamido-l,2,3-friazole;
128. (S)-l-{3-[4-(4-cyanomethyl-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl}-4-cyano-l,2,3-triazole;
129. (S)-N-{3-[4-(4-cyanomethyl-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide; 130. (S)-N- { 3-[4-(4-cyanomethyl-3 -fluoropiperidin- 1 -yl)-phenyl] -2-oxo-oxazolidin- 5- ylmethyl} -thioacetamide;
131. (R)-{3-[4-(4-cyanomethyl-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- alcohol;
132. (R)- {3-[4-(4-cyanomethyl-3-fluoropiperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - methanesulphonate;
133. (S)-N-{3-[4-(4-(l-cyano-l-benzyl)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
134. (S)-N-{3-[4-(4-(l-cyano-2-methanesulphonyloxy)-ethylpiperidin-l-yl)-3-fluoiOphenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide; 135. (S)-N-{3-[4-(4-(l-cyano-l-(3,4-difluorophenyl))-methylpiρeridin-l-yl)-3-fiuorophenyl]-2- oxo-oxazolidin-5-ylnιethyl} -acetamide;
136. (S)-N-{3-[4-(4-(l-cyano-l-(irnidazol-l-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
137. (S)-N-{3-[4-(4-(l-cyano-l-(thiophen-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
138. (S)-N-{3-[4-(4-(l-cyano-l-(pyridin-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmetlιyl}-acetamide;
139. (S)-N- { 3 -[4-(4-( 1 -cyano- 1 -carboxamido)-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-acetamide; 140. (S)-N- {3-[4-(4-(l -cyano-1 -cyclohexylaminocarbonyl)-methylρiperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-yhnethyl}-acetamide;
141. (S)-N- {3-[4-(4-( 1 -cyano- 1 -(pyrrolidin-1 -yl-carbonyl))-methylpiperidin- 1 -yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
142. (S)-N-{3-[4-(4-(l-cyano-l-(morpholin-l-yl-carbonyl))-methylpiperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide;
143. (S)-N-{3-[4-(4-(l-cyano-l-ethoxycarbonyl)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
144. (S)-N- { 3 - [4-(4-( 1 -cyano- 1 -(phenylmercapto))-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2- oxo-oxazolidin-5-ylmethyl}-acetamide;
145. (S)-N-{3-[4-(4-(l-cyano-l-(pyridin-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide; 146. (S)-N- { 3 -[4-(4-( 1 -cyano- 1 -(morpholin- 1 -yl-carbonyl))-methylpiperidin- 1 -yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-thioacetamide;
147. (S)-N-{3-[4-(4-(l-cyano-l-ethoxycarbonyl)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide;
148. (S)-N- {3-[4-(4-(l -cyano-1 -carboxamido)-methylpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide;
149. (S)-N- {3-[4-(4-( 1 -cyano- 1 -thiocarboxamido)-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2- oxo-oxazolidin-5-ylmethyl}-thioacetamide;
150. (S)-l-{3-[4-(4-(l-cyano-2-hydroxy)-ethylpiperidin-l-yl)-3-fluoiOphenyl]-2-oxo- oxazolidin-5-ylmethyl}-4-methoxycarbonyl-l,2,3-friazole; 151. (S)-l-{3-[4-(4-(l-cyano-2-hydroxy)-ethylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -4-ethoxycarbonyl-l ,2,3-triazole;
152. (S)- 1 - {3 -[4-(4-( 1 -cyano-2-hydroxy)-ethylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-5-ethoxycarbonyl-l,2,3-triazole;
153. (R)-3-{3-[4-(4-(l-cyano-2-hydroxy)-ethylpiperidin-l-yl)-3-fluoropheιιyl]-2-oxo- oxazolidin-5-ylmethyloxy} -iso-oxazole;
154. (R)- {3 -[4-(4-( 1 -cyano-2-hydroxy)-ethylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin- 5-ylmethyl} -methanesulphonate;
155. (R)- {3-[4-(4-(l -cyano-1 -hydroxycarbonyl)-methylpiperidin-l -yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulphonate; 156. (R)-{3-[4-(4-(l-cyano-l-ethoxycarbonyl)-methylpiperidin-l-yl)-3-fluoiOphenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulphonate;
157. (R)-{3-[4-(4-(l-cyano-l-(l,3-thiazol-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulphonate;
158. (R)- { 3 -[4-(4-( 1 -cyano- 1 -carboxamido)-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -methanesulphonate;
Some preferred examples of the oxazolidinone derivatives represented by the general Formula I and belonging to the subclass Formula IV are as follows:
159. (S)-N- { 3 - [4-(4-cyanomethyl-3 ,4-dehydropiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5 -ylmethyl } -acetamide;
160. (S)-N-{3-[4-(4-cyanomethyl-3,4-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -thioacetamide;
161. (S)-N- { 3 - [4-(4-cyanome thyl-3 -methyl-4,5-dehydropiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-acetamide;
162. (S)-N-{3-[4-(4-cyanomethyl-3-fluoro-4,5-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide; 163. (S)-N- { 3 - [4-(4-cyanomethyl-3 -fluoro-4, 5-dehydropiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-isobutylcarbamate;
More particularly preferred compounds of the invention of the Formula 1 are:
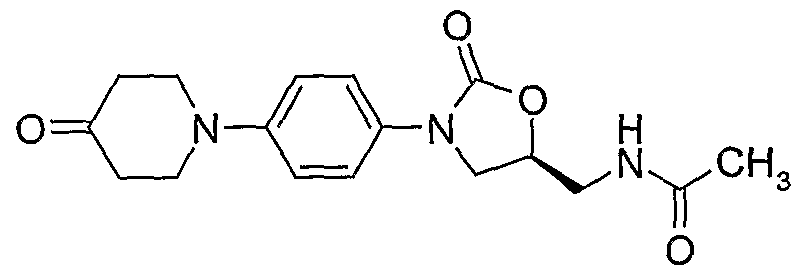
(S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} - acetamide;
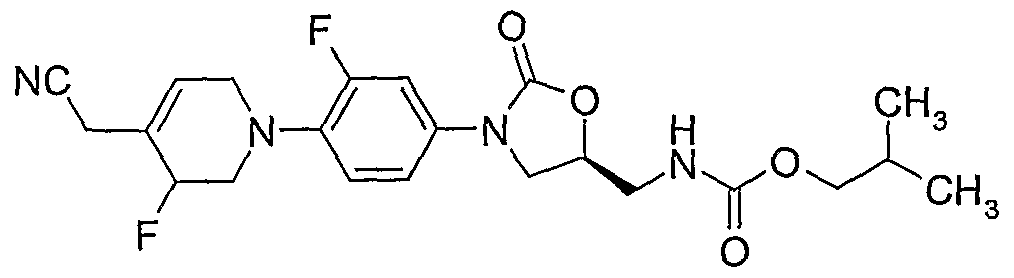
(S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} - acetamide;
(S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-
2-cyanoacetamide; (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- trifluoroacetamide;
(S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- chloroacetamide;
(S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- dichloroacetamide;
(S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-yhnethyl} - thioacetamide; (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- methylthiocarbamate;
(S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -
2-hydroxythio acetamide;
(S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- thiocarbamide;
E-(S)-N-{3-[4-(4-cyanomethylidene-3-fluoiOpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
Z-(S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide; (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -difluoroacetamide;
(S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
(S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thiocarbamide;
(S)-N-{3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methylthiocarbamide;
(S)-N- {3-[4-(4-(l -cyanoethylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -acetamide; (S) -N- { 3 -[4-(4-( 1 -cyanoethylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -difluoroacetamide;
(S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichloroacetamide;
(S)-N- { 3 -[4-(4-( 1 -cyanoethylidene)-piperidin- 1 -yl)-3 -fluorophenyl] ~2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
(S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
(S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichloroacetamide;
(S)-N-{3-[4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide; (S)-N-{3-[4-(4-(l-cyano-3-yne-butylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
(S)-N-{3-[4-(4-(l-cyano-l-(thiophen-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
(S)-N-{3-[4-(4-(l-cyano-l-methylmercapto-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
(S)-N- {3-[4-(4-(l -cyano-1 -bromo-methylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-
5-ylmethyl} -acetamide;
(S)-N- {3 -[4-(4-( 1 -cyano- 1 -(pyridin-2-yl)-methylidene)-piperidin- 1 -yl)-3-fluorophenyι]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide; (S)-N-{3-[4-(4-(l,l-dicyano-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
(S)-N-{3-[4-(4-(l-cyano-l-ethoxycarbonyl-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide;
(S)-N- { 3 -[4-(4-( 1 -cyano- 1 -(morpholin- 1 -yl-thiocarbonyl)-methylidene)-piperidin- 1 -yl)-3 - fluorophenyl]-2-oxo-oxazolidrn-5-ylmethyl} -thioacetamide;
(S)-N-{3-[4-(4-cyanomethyl-3,4-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
(S)-N-{3-[4-(4-cyanomethyl-3,4-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide; (S)-N-{3-[4-(4-cyanomethyl-3-fluoro-4,5-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
A further embodiment of the invention is to provide methods of preparation of the compound of the invention.
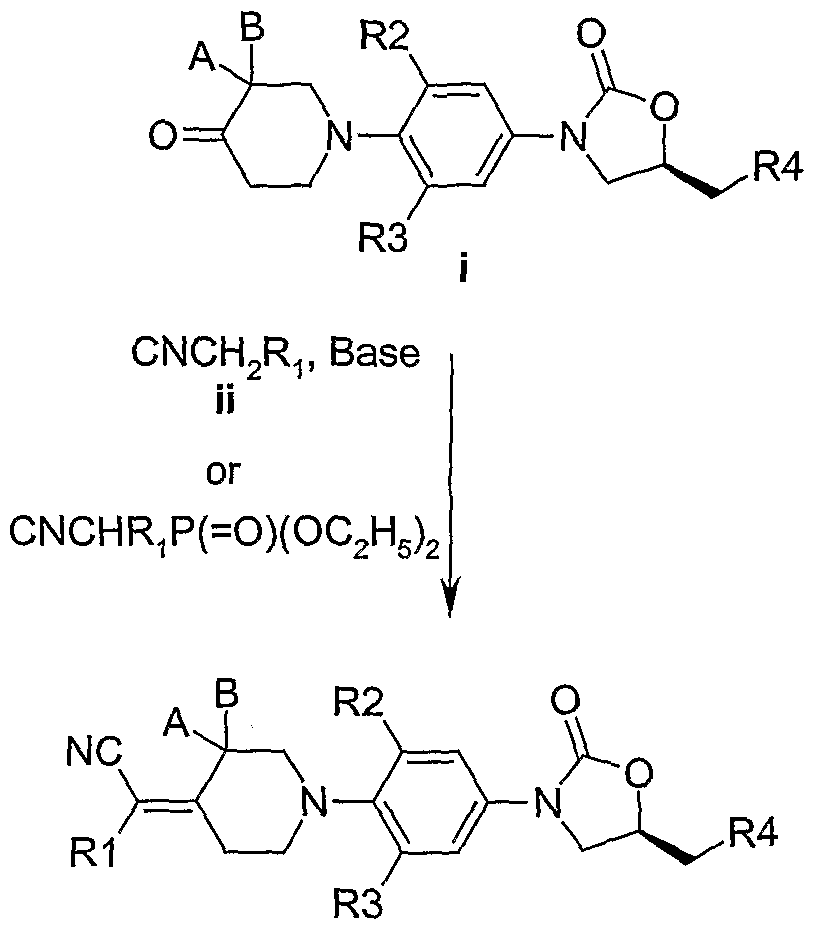
Scheme I describes the preparation of compounds of Formulae II, HI and IV of the present invention. All of the starting materials are prepared by procedures described in this scheme or by
procedures that would be well known to one of ordinary skill in organic chemistry. The variables used in Scheme 1 are as defined above. Optically pure material could be obtained either by one of a number of asymmetric synthesis or alternatively by resolution from a race ic mixture.
In accordance with the Scheme-I piperidone (i) (for example, the preparation of one such piperidone is described in US Patent 5,668,286) is reacted with cyano substituted active methylene compounds ii (Ri as defined) in the presence of a base such as ammonium acetate, sodium methoxide, pyridine and piperidine acetate, preferably ammonium acetate and pyridine and in a solvent such as toluene, THF and methanol at 30 - 110 °C for 2 - 48 hrs. to provide compounds of formula II. Alternatively, i is reacted with a Wittig reagent optionally in the presence of a base such as triethylamine, sodium hydride or n-butyl lithium in a solvent such as ether, tefrahycfrofuran or benzene at 10 - 80 °C to provide compounds of formula II.
The resultant unsaturated cyano derivatives are reduced by hydrogenation in the presence of catalysts such as 5% palladium on carbon, 10% palladium on carbon, palladium hydroxide at atmospheric pressure of hydrogen gas, alternatively by using hydrogen sources such as ammonium formate, cyclohexene in a solvent such as ethyl acetate, methanol, tefrahydrofuran, dichloromethane or chloroform or a mixture thereof at 20 - 60 °C for 1 to 24 hrs. to provide compounds of formula III.
In accordance with the scheme-I, i is reacted with unsubstituted/substituted cyanoacetic acid in the presence of a base such as pyridine, piperidine and ammonium acetate in a solvent such as benzene, toluene at a temperature of 80 to 120 °C for 3 to 24 hrs. to provide compounds of formula IV.
FORMULA-IV
CNCH^COOH pyridine, NH4OAc toluene
10% Pd on carbon
Thioacetamides can conveniently be prepared by allowing the acetamide derivatives to react with Lawesson's reagent in 1 ,4 dioxane, benzene, toluene or tetrahydrofuran at 60 to 110 °C.
The oxazolidinone antibacterial agents of this invention have potential for treatment of specially Gram-positive infections including multi-resistant sfrains. In confrast to compounds of the prior art, they demonstrate bactericidal activity against different resistant microorganisms and in
particular different strains of Enterococcus faecalis. hi addition they display activity against linezolid-resistant S. aureus strains, linezolid-resistant E. faecalis strains and in particular linezolid-resistant S. pneumoniae strains. These compounds are useful for the treatment of Gram- positive or Gram-negative microbial infections in humans and other warm blooded animals by either parenteral, oral or topical administration. The infection in human and other warm blooded animals can be systemic or topical.
The compounds of this invention may be used to prevent infections caused by Gram-positive and Gram-negative bacteria by administering the compound to a subject that is at risk for developing an infection caused by Gram-positive or Gram-negative bacteria. A subject at risk for developing an infection may be a health care worker, surgical patient and the like.
The present invention encompasses certain compounds, dosage forms, and methods of administering the compounds to a human or other animal subject. Specific compounds and compositions to be used in the invention must, accordingly, be pharmaceutically acceptable. As used herein, such a "pharmaceutically acceptable" component is one that is suitable for use with humans and/or animals without undue adverse side effects (such as toxicity, irritation, and allergic response) commensurate with a reasonable benefit/risk ratio.
The pharmaceutical compositions are prepared according to conventional procedures used by persons skilled in the art to make stable and effective compositions. In the solid, liquid, parenteral and topical dosage forms, an effective amount of the active compound or the active ingredient is any amount, which produces the desired results.
For the purpose of this invention the pharmaceutical compositions may contain the active compounds of the invention, their derivatives, salts and hydrates thereof, in a form to be administered alone, but generally in a form to be administered in admixture with a pharmaceutical carrier selected with regard to the intended route of adminisfration and standard pharmaceutical practice. Suitable carriers which can be used are, for example, diluents or excipients such as fillers, extenders, binders, emollients, wetting agents, disintegrants, surface active agents and lubricants which are usually employed to prepare such drugs depending on the type of dosage form.
Any suitable route of administration may be employed for providing the patient with an effective dosage of the compound of the invention their derivatives, salts and hydrates thereof. For example, oral, rectal, viginal, parenteral (subcutaneous, intramuscular, intravenous), transdermal, topical and like forms of administration maybe employed. Dosage forms include (solutions, suspensions, etc) tablets, pills, powders, troches, dispersions, suspensions, emulsions, solutions, capsules, injectable preparations, patches, ointments, creams, lotions, shampoos and the like.
The prophylactic or therapeutic dose of the compounds of the invention, their derivatives, salts or hydrates thereof, in the acute or chronic management of disease will vary with the severity of condition to be freated, and the route of administration, h addition, the dose, and perhaps the dose frequency, will also vary according to the age, body weight and response of the individual patient, h general, the total daily dose range, for the compounds of the invention, the derivatives, salts or hydrates thereof, for the conditions described herein, is from about 200 mg to about 1500 mg, in single or divided doses. Preferably, a daily dose range should be between about 400 mg to 1200 mg, in single or divided dosage, while most preferably a daily dose range should be between about 500 mg to about 1000 mg in divided dosage. While intramuscular administration may be a single dose or up to 3 divided doses, intravenous adminisfration can include a continuous drip. It may be necessary to use dosages outside these ranges in some cases as will be apparent to those skilled in
/ the art. Further, it is noted that the clinician or treating physician will know how and when to interrupt, adjust, or terminate therapy in conjunction with individual patient's response. The term "an amount sufficient to eradicate such infections but insufficient to cause undue side effects" is encompassed by the above - described dosage amount and dose frequency schedule.
A specific embodiment of the invention is that the pharmacokinetic profile of a compound of the invention is such that it permits administration of a dosage schedule which is a much desired once- a-day dosing, a schedule not so far advocated for the only currently available oxazolidinone drug in the market.
A further specific embodiment of the invention is that a compound of the invention has favourable safety advantages in particular no or lower potential to cause myelosuppression. Myelosuppression is known to be a typical class-specific toxicological feature of the oxazolidinone class of antibacterial agents.
Pharmaceutical compositions of the present invention suitable for oral administration maybe presented as discrete units such as capsules, cachets, or tablets, or aerosol sprays, each containing a predetermined amount of the active ingredient, as a powder or granules, or as a solution or a suspension in an aqueous liquid, a non-aqueous liquid, an oil-in-water emulsion, or a water-in-oil liquid emulsion. Such compositions may be prepared by any of the methods of pharmacy, but all methods include the step of bringing into association the active ingredient with the carrier, which constitutes one or more necessary ingredients. In general, the compositions are prepared by uniformly and intimately admixing the active ingredient with liquid carriers or finely divided solid carriers or both, and then, if necessary, shaping the product into the desired presentation.
The compositions of the present invention include compositions such as suspensions, solutions, elixirs, aerosols, and solid dosage forms. Carriers as described in general above are commonly used in the case of oral solid preparations (such as powders, capsules and tablets), with the oral solid preparations being preferred over the oral liquid preparations. The most preferred oral solid preparation is tablets .
Because of their ease of administration, tablets and capsules represent the most advantageous oral dosage unit form, in which case solid pharmaceutical carriers are employed. Examples of suitable carriers include excipients such as lactose, white sugar, sodium chloride, glucose solution, urea, starch, calcium carbonate, kaolin, crystalline cellulose and silicic acid, binders such as water, ethanol, propanol, simple syrup, glucose, starch solution, gelatin solution, carboxymethyl cellulose, shellac, methyl cellulose, potassium phosphate and polyvinyl pyrrolidone, disintegrants such as dried starch, sodium alginate, agar powder, la inaria powder, sodium hydrogen carbonate, calcium carbonate, Tween (fatty acid ester of polyoxyethylenesorbitan), sodium lauryl sulfate, stearic acid monoglyceride, starch, and lactose, disintegration inhibitors such as white sugar, stearic acid glyceryl ester, cacao butter and hydrogenated oils, absorption promoters such as quaternary ammonium bases and sodium lauryl sulfate, humectants such as glycerol and starch, absorbents such as starch, lactose, kaolin, bentonite and colloidal silicic acid, and lubricants such as purified talc, stearic acid salts, boric acid powder, polyethylene glycol and solid polyethylene glycol.
The tablet, if desired, can be coated, and made into sugar-coated tablets, gelatin-coated tablets, enteric-coated tablets, film-coated tablets, or tablets comprising two or more layers.
If desired, tablets may be coated by standard aqueous or non-aqueous techniques. In molding the pharmaceutical composition into pills, a wide variety of conventional carriers known in the art can be used. Examples of suitable carriers are excipients such as glucose, lactose, starch, cacao butter, hardened vegetable oils, kaolin and talc, binders such as gum arabic powder, fragacanth powder, gelatin, and ethanol, and disintegrants such as laminaria and agar.
fri molding the pharmaceutical composition into a suppository form, a wide variety of carriers known in the art can be used. Examples of suitable carriers include polyethylene glycol, cacao butter, higher alcohols, gelatin, and semi-synthetic glycerides.
A second preferred method is parenterally for intramuscular, intravenous or subcutaneous administration.
A third preferred route of adminisfration is topically, for which creams, ointments, shampoos, lotions, dusting powders and the like are well suited. Generally, an effective amount of the compound according to this invention in a topical form is from about 0.1% w/w to about 10% w/w of the total composition. Preferably, the effective amount of the compound of the invention is 1% w/w of the total composition.
In addition to the common dosage forms set out above, the compounds of the present invention may also be administered by controlled release means and/or delivery devices such as those described in U.S. Patent Nos. 3,845,770; 3,916,899; 3,536,809; 3,598,123 and 4,008,719; the disclosures of which are hereby incorporated by reference.
Desirably, each tablet contains from about 200 mg to about 1500 mg of the active ingredient. Most preferably, the tablet, cachet or capsule contains either one of three dosages, about 200 mg, about 400 mg, or about 600 mg of the active ingredient.
When the pharmaceutical composition is formulated into an injectable preparation, in formulating the pharmaceutical composition into the form of a solution or suspension, all diluents customarily used in the art can be used. Examples of suitable diluents are water, ethyl alcohol, polypropylene glycol, ethoxylated isostearyl alcohol, polyoxyethylene sorbitol, and sorbitan esters. Sodium chloride, glucose or glycerol may be incorporated into a therapeutic agent.
The antimicrobial pharmaceutical composition may further contain ordinary dissolving aids, buffers, pain-alleviating agents, and preservatives, and optionally coloring agents, perfumes, flavors, sweeteners, and other drugs.
For topical application, there are employed as non-sprayable forms, viscous to semi-solid or solid forms comprising a carrier compatible with topical application and having a dynamic viscosity preferably greater than water. Suitable formulations include but are not limited to solutions, suspensions, emulsions, creams, ointments, powders, liniments, salves, aerosols, etc., which are, if desired, sterilized or mixed with auxiliary agents, e.g. preservatives, antioxidants, stabilizers, wetting agents, buffers or salts for influencing osmotic pressure, etc. For topical application, also suitable are sprayable aerosol preparations wherein the active ingredient preferably in combination with a solid or liquid inert carrier material.
A specific embodiment of the invention is the preparation of storage stable compositions of the compounds of the invention of formula I. Such stable compositions can be advantageously made through the use of selective stabilizers. Different stabilizers are known to those skilled in the art of making pharmaceutical compositions. Of special utility for making storage stable compositions of the compound of the invention of formula I, stabilizers such as disodium ethylenediaminetetraacetic acid (EDTA), tromethamine, cyclodextrins such as gamma- cyclodextrin, hydiOxy-propyl-gamma-cyclodextrin have been found to be useful.
In a specific embodiment of the invention, the pharmaceutical compositions contain an effective amount of the active compounds of the invention, its derivatives, salts or hydrates thereof described in this specification as hereinbefore described in admixture with a pharmaceutically acceptable carrier, diluent or excipients, and optionally other therapeutic ingredients.
The invention is further defined by reference to the following examples describing in detail the preparation of the composition of the present invention as well as their utility. It will be apparent to those skilled in the art that many modifications, both to materials and methods may be practiced without departing from the purpose and scope of this invention.
The compounds of this invention are useful antimicrobial agents effective against various humans and veterinary pathogens specially including Linezolid-resistant strains.
Further embodiments of the invention are the linezolid-resistant strains of the invention and methods for producing them. Linezolid-resistant mutants S. pneumoniae ATCC 6303 LR, S. aureus Smith LR & MRSA 032 LR were selected from corresponding sensitive strains S. pneumoniae ATCC 6303, S. aureus Smith & MRSA 032 respectively under in-vivo conditions from mice infected with respective parent strains and freated with various dosages of linezolid . Selected mutants and parent strains were analyzed for the presence of mutation in 23 S rRNA by sequencing. The methodology involved amplication of genes coding 23 S rRNA from linezolid- resistant mutants employing a PCR based DNA amplification method. The mutations in 23 S rRNA gene were identified by sequencing of amplified DNA following elecfrophoretic separation.
Preparations
Preparation- 1 Preparation of 4-[4-oxo-piperidin- 1 -yl]-nitrobenzene
0=<^ -\^>- 02
The mixture of 4-piperidone hydrochloride (0.851 mol), triethylamine (1.70 mol), 4- fluoronifrobenzene (0.851 mol) in 800 ml chloroform was heated under reflux for 16 hours. The solvent was removed under vacuum and to the residue water (1 liter) was added and the precipitate was filtered to afford 4-[4-oxo-piperidin-l-yl]-nifrobenzene in 80% yield. MS (M+l) = 221 (MH+, 100%), M.F = CnHι2N2O3
Preparation-2 Preparation of 4-[4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)]-nitrobenzene
The mixture of 4-[4-oxo-piperidin-l-yl]-nitrobenzene from step-1 (0.596 mol), ethylene glycol (1.09 mol) and p-toluenesulphonic acid monohydrate (0.147 mol) in toluene was heated to reflux for 5 hours. The reaction mixture was washed with water. The organic layer was evaporated to afford 4-[4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)]-nitrobenzene as a solid in 98% yield. MS (M+l) = 266 (MH+, 100%), M.F .= Cι3Hι6N2O4
Preparation-3
Preparation of [4-(l ,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)]-phenyl-4-yl]-arninocarbonyloxymethyl]- benzene
The suspension of 4-[4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)]-nitrobenzene (0.377 mol), 10% palladium on carbon (10 g) in tefrahydrofuran (800 ml) was stirred at room temperature under hydrogen atmosphere (400 psi) overnight.
The reaction mixture filtered to remove the catalyst. To the filtrate, sodium bicarbonate (0.56 mol) and benzyl chloroformate (0.41 mol) was added at 0-5 °C and stirred at room temperature for 30 minutes. The solvent was evaporated under vacuum and the residue stirred with hexane. The precipitate was filtered to give the title compound in 97% yield. MS (M+l) = 369 (MH+, 100%), M.F.= C2ιH24N2O4
Preparation-4 Preparation of (R)-3- {4-(l ,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)]-phenyl)--2-oxo-oxazolidin-5- ylmethyl} -alcohol
Butyl lithium (1.6 M in hexane, 180 ml) was added to the solution [4-(l,4-dioxa-8-aza-spiro[4.5]- dec-8-yl)]-phenyl-4-yl]-aminocarbonyloxymethyl]-benzene
(0.313 mol) in tefrahydrofuran (1000 ml) at-78°C. (R)-(-)-Glycidyl butyrate (0.32 mol) was added to the reaction mixture and it was stirred overnight. The reaction mixture was extracted with the ethyl acetate after quenching with saturated aqueous ammonium chloride solution. The evaporation of solvent afforded title compound in 80% yield. MS (M+l) = 335 (MH+, 100%), M.F.= Cι7H22N2O5
Preparation-5 Preparation of (R)-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol
The mixture of (R)- {3-[4-(4-(l ,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol (0.016 mol), j_>-toluene sulfonic acid (0.032 mol) in acetone water (300 ml, 40: 60) mixture was refluxed for 6 hours. The reaction mixture was concentrated under vacuum and treated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford title compound 78% yield. MS (M+l) = 291 (MH+, 100%), M.F.= Cι5Hι8N2O4
Preparation-6 Preparation of (R)-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- methanesulphonate
The mixture of (R)-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- alcohol(0.194 mol), friethylamine (0.213 mol), and methanesulphonyl chloride (0.232 mol) in 700 ml of dichloromethane was stirred for 1 hour. The reaction mixture was washed with 1 liter water. The organic layer was dried and evaporated under vacuum to afford title compound in 87% yield. MS (M+l) = 369 (MH+, 100%), M.F.= Cι
6H
2oN
2O
6S
Preparation-7 Preparation of (S)-{3-[4-(4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- yimethyl} -azide
The mixture of (R)- {3-[4-(4-(l ,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulfonate (0.16 mol), sodium azide (0.46 mol) in dimethylformamide (200 ml) was heated at 70°C for 14 hours. The reaction mixture was cooled and poured in ice cold water. The precipitate was filtered to provide title compound in 85% yield. MS (M+l) = 360 (MH+, 100%), M.F.= Cι7H2ιN5O4
Preparation-8 Preparation of (S)- {3-[4-(4-oxo-piperidm-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} -azide
The mixture of (S)-{3-[4-(4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide (0.014 mol), />-toluene sulfonic acid (0.026 mol) in acetone water (150 ml, 40: 60) mixture was refluxed for 6 hours. The reaction mixture was concentrated under vacuum and freated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford keto oxazolidinone azide compound 50% yield. MS (M+l) = 316 (MH+, 100%), M.F.= Cι
5Hι
7N
5O
3
Preparation-9 Preparation of (S)-N- {3-[4-(4-(l ,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide
The suspension of (S)-{3-[4-(4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide (0.153 mol), 10% palladium on carbon (7 g), pyridine (0.45 mol), acetic anhydride (0.18 mol) in 700 ml ethyl acetate was stirred at 400 psi hydrogen gas pressure overnight. The suspension was filtered. Filtrate was purified to provide title compound in 70% yield.
MS (M+l) = 376 (MH+, 100%), M.F.= Cι9H25N3O5
Preparation- 10 Preparation of (S)-N-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyI}- acetamide
The (S)-N-{3-[4-(4-(l,4-dioxa-8-aza-spiro[4.5]-dec-8-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide (0.040 mol), -toluene sulfonic acid (0.080 mol) in acetone water (350 ml, 40: 60) mixture was refluxed for 5 hours. The reaction mixture was concenfrated under vacuum and treated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford keto oxazolidinone acetamide compound 76% yield. MS (M+l) = 332 (MH+, 100%), M.F.= Cι
7H
2ιN
3O=
Preparation- 11 Preparation of 4-(4-trimethylsilyloxy-3 ,4-dehydropiperidin- 1 -yl)-nitrobenzene
The mixture of 4-(4-oxo-piperidin-l-yl)-nitrobenzene (126 mmol), triethylamine (630 mmol), trimethylsilylchloride (375.0 mmol) in dimethylformamide was heated at 70°C for 24h. The solvent was removed under vacuum and to the residual mass was extracted with the ethyl acetate water mixture. The combined organic layer was dried and after removal of the solvent afforded title compound as a solid in 90% yield. MS (M+l) = 293 (MH+, 100%), M.F.= Cι4H20N2O3Si
Preparation- 12 Preparation of 4-(3-fluoro-4-oxo-piperidin-l-yl)-nitrobenzene
The mixture of 4-(4-trimethylsilyloxy-3 ,4-dehydropiperidin- 1 -yl)-nitrobenzene
(101 mmol), selectfluor (101 mmol) in acetonitrile (100 ml) was stirred for 4 hours. The solvent was removed under reduced pressure and to the residual mass was extracted into ethyl acetate water mixture. The combined organic layer was dried and removal of the solvent afforded title compound in 95% yield. MS (M+l) = 239 (MH+, 100%), M.F.= CπHπFN2O3
Preparation- 13 Preparation of 4-(4,4-dimethoxy — 3 -fluoropiperidin- 1 -yl)-nitrobenzene
The mixture of 4-(3-fluoro-4-oxo-piperidin-l-yl)-nifroberιzene (51 mmol), trimethylorthoformate (103 mmol), p-toluene-sulphonic acid monohydrate (51 mmol) in methanol (100 ml) was heated at 45°C for 24 hours. Solvent was removed and residual mass was taken into ethyl acetate and saturated sodium bicarbonate solution mixture. The organic layer was dried and removal of the solvent afforded title compound as a solid in 91% yield.
MS (M+l) = 285 (MH+, 100%), M.F.= d3Hι7FN2O4
Preparation- 14 Preparation of [4-(4,4-dimethoxy-3-fluoropiperiά n-yl)-aminocarbonyloxymethyl]-benzene
The suspension of 4-(4,4-dimethoxy — 3-fluoropiperidin-yl)-nitrobenzene (38 mmol), and 10% palladium on carbon (1 g) in tefrahydrofuran (500 ml) was stirred at room temperature under hydrogen atmosphere (200 psi) for 6 hour. The suspension was filtered. To the filtrate sodium bicarbonate (57 mmol) and benzyl chloroformate (46 mmol) was added and the reaction mixture was stirred at room temperature for 30 min. The solvent was removed and the residue was extracted with ethyl acetate and water mixture. The organic layer was dried and the residue was recrystallized from hexane:ehtyl acetate to give the title compound in 93% yield. MS (M+l) = 389 (MH+, 100%), M.F = C
2!H
25FN
2O
4
Preparation- 15
Preparation of (R)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl } -alcohol
Butyl lithium (1.6 M in hexane, 27 ml) was added to the solution of [4-(4,4-dimethoxy — 3- fluoropiperidin-yl)-aminocarbonyloxymethyl]-benzene (35.7mmol) in tefrahydrofuran (250 ml) at -78°C under an inert atmosphere. (R)-(-)-Glycidyl butyrate (37.5 mmol) was added to the reaction mixture and was stirred for 15 hours. The reaction mixture was extracted with the ethyl acetate water mixture. The combined organic layer was dried and removal of the solvent afforded a residue which was recrystallized from dichloromethane:hexane mixture to give title product in 89% yield.
MS (M+l) = 355 (MH+, 100%), M.F.= Cι7H23FN2O5
Preparation- 16 Preparation of (R)- { 3 - [4-(4-oxo-3 -fluoropiperidin- 1 -yl)-phenyl] -2-oxo-oxazolidin-5 -ylmethyl } - alcohol
To the mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol (0.726 mmol), freshly fused zinc chloride (2.17mmol), dimethyl sulphide (3.2mmol), acetyl chloride (2.17mmol) in tetrahydrofuaran (50 ml) was stirred at 40°C for 4 days. To this reaction mixture extracted with the ethyl acetate water mixture and organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel afforded title compound in 49% yield. MS (M+l) = 309 (MH+, 100%), M.F.= C
15H
17FN
2O
4
Preparation- 17 Preparration of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl } -methanesulfonate
The mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol (29.6mmol), triethylamine (65 mmol), methanesulphonyl chloride (41.5mmol) in dichloromethane (100 ml) was stirred for 1 hour at room temperature. Reaction mixture was extracted with the dichloromethane water mixture. The combined organic layer was dried over sodium sulfate and removal of solvent afforded title compound in 98% yield. MS (M+l) = 433 (MH+, 100%), M.F.= C18H25FN2O7S
Preparation- 18
Preparation of (R)-{3-[4-(4-oxo-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- methanesulfonate
To the mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulfonate (0.726 mmol), freshly fused zinc chloride (2.17mmol), dimethyl sulphide (3.2mmol), acetyl chloride (2.17mmol) in tetrahydrofuaran (50 ml) was stirred at 40°C for 4 days. To this reaction mixture extracted with the ethyl acetate water mixture and organic
layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel afforded title compound in 57% yield. MS (M+l) = 387 (MH+, 100%), M.F ~ Cι6Hι9FN2O6S
Preparation- 19 Preparation of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide
The mixture of sodium azide (88.5 mmol) and (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)- phenyl]-2-oxo-oxazolidin-5-ylmethyl}-methanesulfonate
(29.6mmol) in dimethylformamide (75 ml) was heated at 70°C for 14 hours. The reaction mixture was poured on ice cold water, and the solid was filtered to afford title compound in 78% yield.
MS (M+l) = 380 (MH+, 100%), M.F.= Cι7H22FN5O4
Preparation-20
Preparation of (S)- { 3 -[4-(4-oxo-3 -fluoropiperidin- 1 -yl)-phenyl] -2-oxo-oxazolidin-5 -ylmethyl} - azide
To the mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide (0.5 mmol), freshly fused zinc chloride (1.5 mmol), dimethyl sulphide (2.5 mmol), acetyl chloride (1.5 mmol) in tefrahydrofuran (50 ml) was stirred at 40°C for 4 days. To this reaction mixture extracted with the ethyl acetate water mixture and organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel afforded title compound in 67% yield. MS (M+l) = 334 (MH+, 100%), M.F.= 5H16FN5O3
Preparation-21 Preparation of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine
The suspension of (S)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-ox-ιzolidin-5- ylmethyl} -azide (25.2 mmol) and 10% palladium on carbon (1.0 g) was in ethyl acetate (150 ml) was stirred at a room temperature under hydrogen atmosphere for 10 hours. The reaction mixture was filtered and the filtrate was concentrated to give a residue, which was purified on silica gel column chromatography to provide title compound in 89% yield. MS (M+l) = 308 (MH+, 100%), M.F .= Cι5Hι8FN3O3
Preparation-22 Preparation of (S)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine (6.73 mmol), pyridine (26.9 mmol), acetic anhydride (9.43 mmol) in ethyl acetate (25 ml) was stirred for 5 hours at room temperature. The reaction mixture was extracted with the ethyl acetate water mixture and combined organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound in 49% yield. MS (M+l) = 396 (MH+, 100%), M.F.= C19H26FN3O5
Prgparation-23
Preparation of (S)-{3-[4-(4-oxo-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide (1.0 mmol), freshly fused zinc chloride (3.1 mmol), dimethyl sulphide (5.1 mmol), acetyl cliloride (3.1 mmol) in tefrahydrofuaran (50 ml) was stirred at 40°C for 4 days. The reaction mixture was extracted with ethyl acetate water mixture and the organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound in 61% yield. MS (M+l) = 350 (MH+, 100%), M.F = Cι7H20FN3O
Preparation-24 Preparation of 4-(4-tiimethylsilyloxy-3,4-dehydropiperidin-l-yl)-3-fluoronitrobenzene
The mixture of 4-(4-oxo-piperidin-l-yl)-3-fluoronifrobenzene (0.250 mol), triethylamine (1.250 mol), trimethylsilylchloride (0.750 mol) in dimethylformamide was heated at 70°C for 24h. The solvent was removed under vacuum and to the residual mass was extracted with the ethyl acetate water mixture. The combined organic layer was dried and after removal of the solvent afforded title compound as a solid in 86% yield. MS (M+l) = 311 (MH+, 100%), M.F.= Cι
4Hι
9FN
2O
3Si
Preparation-25
Preparation of 4-(3-fluoro-4-oxo-piperidin-l-yl)-3-fluoronitrobenzene
The mixture of 4-(4-trimethylsilyloxy-3 ,4-dehydropiperidin- 1 -yl)-3 -fluoronifrobenzene (151 mmol), selectfluor (151 mmol) in acetonitrile was stirred for 4 hours. The solvent was removed under reduced pressure and to the residual mass was exfracted into ethyl acetate water mixture. The combined organic layer was dried and removal of the solvent afforded title compound in 88% yield. MS (M+l) = 257 (MH+, 100%), M.F.= CπH10F2N2O3
Preparation-26
Preparation of 4-(4,4-dimethoxy — 3 -fluoropiperidin-l-yι)-3 -fluoronifrobenzene
The mixture of 4-(3-fluoro-4-oxo-piperidin-l-yl)-3 -fluoronifrobenzene (65 mmol), trimethylorthoformate (130 mmol), p-toluene-sulphonic acid monohydrate (67mmol) in methanol was heated at 45°C for 24 hours. Solvent was removed and residual mass was taken into ethyl acetate and saturated sodium bicarbonate solution mixture. The organic layer was dried and removal of the solvent afforded title compound as a solid in 78% yield.
MS (M+l) = 303 (MH+, 100%), M.F.= Cι3Hι6F2N2O4
Preparation-27 Preparation of [4-(4,4-dimethoxy-3-fluoropiperidin-yl)-arninocarbonyloxyιτιethyl]-3- fluorobenzene
The suspension of 4-(4,4-dimethoxy — 3-fluoropiperidin-yl)-3-fluoromfrobenzene (48 mmol), and 10% palladium on carbon (1 g) in tefrahydrofuran was stirred at room temperature under hydrogen atmosphere (200 psi) for 6 hour. The suspension was filtered. To the filtrate sodium bicarbonate
(72 mmol) and benzyl chloroformate (58 mmol) was added and the reaction mixture was stirred at room temperature for 30 min. The solvent was removed and the residue was exfracted with ethyl acetate and water mixture. The organic layer was dried and the residue was recrystallized from hexane:ehtyl acetate to give the title compound in 80% yield. MS (M+l) = 407 (MH+, 100%), M.F.= C2ιH24F2N2O4
Preparation-28 Preparation of (R)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin-l -yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -alcohol
Butyl lithium (1.6 M in hexane, 27 ml) was added to the solution of [4-(4,4-dimethoxy — 3- fluoropiperidin-yl)--ιrrιinocarbonyloxymethyl]- 3-fluorobenzene (35.0 mmol) in tefrahydrofuran (250 ml) at -78°C under an inert atmosphere. (R)-(-)-Glycidyl butyrate (37.1 mmol) was added to the reaction mixture and was stirred for 15 hours. The reaction mixture was extracted with the ethyl acetate water mixture. The combined organic layer was dried and removal of the solvent afforded a residue which was recrystallized from dichloromethane-.hexane mixture to give title product in 78% yield. MS (M+l) - 373 (MH+, 100%), M.F.= Cι7H22F2N2O5
Preparation-29
Preparation of (R)-{3-[4-(4-oxo-3-fluoropiperidin-l-yl)- 3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol
To the mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)- 3-fluoro phenyl]-2-oxo- oxazolidin-5-ylmethyl} -alcohol (0.726 mmol), freshly fused zinc chloride (2.17mmol), dimethyl sulphide (3.2mmol), acetyl chloride (2.17mmol) in tetrahydrofuaran (50 ml) was stirred at 40°C for 4 days. To this reaction mixture extracted with the ethyl acetate water mixture and organic
layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel afforded title compound in 54% yield. MS (M+l) = 327 (MH+, 100%), M.F.= Cι5H16F2N2O=
Preparation- 30
Preparration of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl } -methanesulfonate
The mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -alcohol (29.6 mmol), triethylamine (65 mmol), methanesulphonyl chloride (41.5 mmol) in dichloromethane (100 ml) was stirred for 1 hour at room temperature. Reaction mixture was extracted with the dichloromethane water mixture. The combined organic layer was dried over sodium sulfate and removal of solvent afforded title compound in 86% yield. MS (M+l) = 451 (MH+, 100%), M.F.= Cι8H24F2N2O7S
Preparation-31 Preparation of (R)-{3-[4-(4-oxo-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulfonate
To the mixture of (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulfonate (0.5 mmol), freshly fused zinc chloride (1.5 mmol), dimethyl sulphide (2.5 mmol), acetyl chloride (0.5 mmol) in tetrahydrofuaran was stirred at 40°C for 5 days. To this reaction mixture extracted with the ethyl acetate water mixture and organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue which was chromatographed over silica gel afforded title compound in 55% yield. MS (M+l) = 405 (MH+, 100%), M.F .= d
6Hι
8F
2N
2O
6S
Preparation-32 Preparation of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -azide
The mixture of sodium azide (67.0 mmol) and (R)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yι)- 3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-methanesulfonate
(27.0 mmol) in dimethylformamide was heated at 70°C for 14 hours. The reaction mixture was poured on ice cold water, and the solid was filtered to afford title compound in 78% yield. MS (M+l) = 398 (MH+, 100%), M.F.= Cι7H21F2N5O4
Preparation-33 Preparation of (S)- {3-[4-(4-oxo-3-fluoropiperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -azide
To the mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluoiOphenyl]-2-oxo- oxazolidin-5 -ylmethyl} -azide (0.5 mmol), freshly fused zinc chloride (1.5 mmol), dimethyl sulphide (2.5 mmol), acetyl chloride (1.5 mmol) in tetrahydrofuaran (50 ml) was stirred at 40°C for 4 days. To this reaction mixture exfracted with the ethyl acetate water mixture and organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel afforded title compound in 69% yield. MS (M+l) = 352 (MH+, 100%), M.F.= Cι
5Hι
5F
2N
5O
3
Preparation-34
Preparation of (S)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine
The suspension of (S)-{3-[4-(4,4-dimethoxy-3-fluoroρiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -azide (25.2 mmol) and 10% palladium on carbon (1.0 g) was in ethyl acetate (150 ml) was stirred at a room temperature under hydrogen atmosphere for 10 hours. The reaction mixture was filtered and the filtrate was concentrated to give a residue, which was purified on silica gel column chromatography to provide title compound in 76% yield. MS (M+l) = 372 (MH+, 100%), M.F = CπH23F2N3O4
Pre aration-35 Preparation of (S)- {3-[4-(4,4-dimethoxy-3-fluoropiperidin- l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidm-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine (6.73 mmol), pyridine (26.9 mmol), acetic anhydride (9.43 mmol) in ethyl acetate (25 ml) was stirred for 5 hours at room temperature. The reaction mixture was extracted with the ethyl acetate water mixture and combined organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound in 58% yield.
MS (M+l) = 414 (MH+, 100%), M.F - Ci9H25F2N3O5
Preparation-36 Preparation of (S)- {3-[4-(4-oxo-3-fluoropiperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide (1.0 mmol), freshly fused zinc chloride (3.1 mmol), dimethyl sulphide (5.1 mmol), acetyl chloride (3.1 mmol) in tefrahydrofuaran (50 ml) was stirred at 40° C for 4 days. The reaction mixture was extracted with ethyl acetate water mixture and the organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound in 61% yield. MS (M+l) = 368 (MH+, 100%), M.F.= Cι
7Hι
9F
2N
3O
4
Preparation-37 Preparation of 4-(4,4-dimethoxy — 3,3-difluoropiperidin-l-yl)-nitrobenzene
The mixture of 4-(3,3-difluoro-4-oxo-piperidin-l-yl)-nifrobenzene (25 mmol), trimethylorthoformate (51 mmol), p-toluene-sulphonic acid monohydrate (27mmol) in methanol was heated at 45 °C for 24 hours. Solvent was removed and residual mass was taken into ethyl acetate and saturated sodium bicarbonate solution mixture. The organic layer was dried and removal of the solvent afforded title compound as a solid in 84% yield. MS (M+l) = 321 (MH+, 100%), M.F.= Cι3Hι5F3N2O4
Preparation-38 Preparation of [4-(4,4-dimethoxy-3,3-difluoropiperidin-yl)-aminocarbonyloxymethyl]-benzene
The suspension of 4-(4,4-dimethoxy — 3,3-difluoropiperidin-yl)-nitrobenzene (20 mmol), and 10% palladium on carbon (1 g) in tetrahydrofuran was stirred at room temperature under hydrogen atmosphere (200 psi) for 6 hour. The suspension was filtered. To the filtrate sodium bicarbonate (40 mmol) and benzyl chloroformate (25 mmol) was added and the reaction mixture was stirred at room temperature for 30 min. The solvent was removed and the residue was exfracted with ethyl acetate and water mixture. The organic layer was dried and the residue was recrystallized from hexane:ehtyl acetate to give the title compound in 75% yield. MS (M+l) = 425 (MH+, 100%), M.F.= C
2ιH
3F
3N
2O
4
Preparation-39
Preparation of (R)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl } -alcohol
Butyl lithium (1.6 M in hexane, 27 ml) was added to the solution of [4-(4,4-dimethoxy — 3,3- difluoropiperidin-yl)-aminocarbonyloxymethyl]-beιιzene (35.0 mmol) in tefrahydrofuran (250 ml) at -78°C under an inert atmosphere. (R)-(-)-Glycidyl butyrate (37.1 mmol) was added to the reaction mixture and was stirred for 15 hours. The reaction mixture was extracted with the ethyl acetate water mixture. The combined organic layer was dried and removal of the solvent afforded a residue which was recrystallized from dichloromethane:hexane mixture to give title product in 80% yield.
MS (M+l) = 391 (MH+, 100%), M.F.= Cι7H2ιF3N2O5
Preparation- 40 Preparration of (R)- {3-[4-(4,4-dimethoxy-3,3-difluoropiperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulfonate
The mixture of (R)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol (15 mmol), triethylamine (32 mmol), methanesulphonyl chloride (21 mmol) in dichloromethane was stirred for 1 hour at room temperature. Reaction mixture was extracted with the dichloromethane water mixture. The combined organic layer was dried over sodium sulfate and removal of solvent afforded title compound in 90% yield. MS (M+l) = 469 (MH+, 100%), M.F.= Cι
8H
23F
3N
2O
7S
Preparation-41 Preparation of (S)- {3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide
The mixture of sodium azide (26.0 mmol) and (R)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l- yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-methanesulfonate
(12.0 mmol) in dimethylformamide was heated at 70°C for 14 hours. The reaction mixture was poured on ice cold water, and the solid was filtered to afford title compound in 88% yield. MS (M+l) = 416 (MH+, 100%), M.F.= Cι7H20F3N5O4
Preparation-42 Preparation of (S)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine
The suspension of (S)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo- oχazolidin-5-ylmethyl} -azide (10 mmol) and 10% palladium on carbon (0.15 g) was in ethyl acetate was stirred at a room temperature under hydrogen atmosphere for 10 hours. The reaction mixture was filtered and the filtrate was concentrated to give a residue, which was purified on silica gel column chromatography to provide title compound in 88% yield. MS (M+l) = 390 (MH+, 100%), M.F .= Cι7H22F3N3O4
Preparation-43 Preparation of (S)- {3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-3-phenyl]-2-oxo-oxazolidin- 5-ylmethyl} -amine (8 mmol), pyridine (32 mmol), acetic anhydride (16 mmol) in ethyl acetate was stirred for 5 hours at room temperature. The reaction mixture was exfracted with the ethyl acetate water mixture and combined organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound in 80% yield. MS (M+l) = 432 (MH+, 100%), M.F.= Cι9H24F3N3O5
Preparation-44 Preparation of (S)-{3-[4-(4-oxo-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl } -acetamide
The mixture of (S)-{3-[4-(4,4-dimethoxy-3,3-difluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide (1.0 mmol), freshly fused zinc chloride (3.1 mmol), dimethyl sulphide (5.1 mmol), acetyl chloride (3.1 mmol) in tetrahydrofuaran (50 ml) was stirred at 40°C for 4 days. The reaction mixture was extracted with ethyl acetate water mixture and the organic layer was dried over sodium sulfate. The removal of the solvent afforded a residue, which was chromatographed over silica gel to give title compound hi 71% yield. MS (M+l) = 386 (MH+, 100%), M.F .= Cι7H)8F3N3O4
Preparation-45 Preparation of 4-r3-methyl-4-oxo-piperidin- 1 -yl]-3-fluoronitrobenzene
The mixture of 3-methyl-4-piperidone hydrochloride (0.085 mol), triethylamine (0.170 mol), 4- fluoronifrobenzene (0.085 mol) in chloroform was heated under reflux for 16 hours. The solvent was removed under vacuum and to the residue water was added and the precipitate was filtered to afford 4-[3-methyl-4-oxo-piperidin-l-yl]-3-fluoronitrobenzene in 76% yield. MS (M+l) = 253 (MH+, 100%), M.F.= Cι2Hι3FN2O3
Preparation-46
Preparation of 4-[4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluoronitrobenzene
The mixture of 4-[3-methyl-4-oxo-piperidin-l-yl]-3-fluoronitrobenzene from step-1 (0.059 mol), ethylene glycol (1.09 mol) and p-toluenesulphonic acid monohydrate (0.014 mol) in toluene was heated to reflux for 5 hours. The reaction mixture was washed with water. The organic layer was evaporated to afford 4-[4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluoronifrobenzene as a solid in 78% yield. MS (M+l) = 297 (MH+, 100%), M.F.= Cι4Hι7FN2O4
Preparation-47
Preparation of [4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluorophenyl-4-yl]- aminocarbonyloxymethyl]-benzene
The suspension of 4-[4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluoronitrobenzene (0.038 mol), 10% palladium on carbon (0.5 g) in tefrahydrofuran was stirred at room temperature under hydrogen atmosphere (400 psi) overnight.
The reaction mixture filtered to remove the catalyst. To the filtrate, sodiumbicarbonate (0.056 mol) and benzyl chloroformate (0.041 mol) was added at 0-5 °C and stirred at room temperature for 30 minutes. The solvent was evaporated under vacuum and the residue stirred with hexane. The precipitate was filtered to give the title compound in 89% yield. MS (M+l) = 401 (MH+, 100%), M.F.= C22H25FN2O4
Preparation-48 Preparation of (R)-3-{4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluorophenyl)~2-oxo- oxazolidin-5-ylmethyl} -alcohol
Butyl lithium (1.6 M in hexane, 180 ml) was added to the solution [4-(l,4-dioxa-3-methyl-8-aza- spiro[4.5]-dec-8-yl)]-3-fluorophenyl-4-yl]-aminocarbonyloxymethyl]-benzene (0.031 mol) in tefrahydrofuran at -78°C. (R)-(-)-Glycidyl butyrate (0.032 mol) was added to the reaction mixture and it was stirred overnight. The reaction mixture was extracted with the ethyl acetate after quenching with saturated aqueous ammonium chloride solution. The evaporation of solvent afforded title compound in 88% yield. MS (M+l) = 367 (MH+, 100%), M.F.= Cι8H23FN2O5
Preparation-49 Preparation of R)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-methanesulfonate
The mixture of (R)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-phenyl]-2-oxo- oxazolidin-5-ylmethyl} -alcohol (0.194 mol), triethylamine (0.213 mmol), and methanesulphonyl chloride (0.232 mol) in 700 ml of dicliloromethane was stirred for 1 hour. The reaction mixture was washed with 1 liter water. The organic layer was dried and evaporated under vacuum to afford title compound in 87% yield.
MS (M+l) = 445 (MH+, 100%), M.F.= d9H25FN2O7S
Preparation-50 Preparation of (S)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-azide
The mixture of (R)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-methanesulfonate (0.16 mol), sodium azide (0.46 mol) in dimethylformamide (200 ml) was heated at 70°C for 14 hours. The reaction mixture was cooled and poured in ice cold water. The precipitate was filtered to provide title compound in 85% yield. MS (M+l) = 392 (MH+, 100%), M.F.= Cι8H22FN5O4
Preparation-51 Preparation of (S)- {3-[4-(4-(l ,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine
The suspension of (S)-{3-[4-(4,4-dimethoxy-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide (25.2 mmol) and 10% palladium on carbon (1.0 g) was in ethyl acetate was stirred at a room temperature under hydrogen atmosphere for 10 hours. The reaction mixture was filtered and the filtrate was concentrated to give a residue, which was purified on silica gel column chromatography to provide title compound in 85% yield. MS (M+l) = 366 (MH+, 100%), M.F.= Cι8H24FN3O4
Preparation-52 Preparation of (S)-{3-[4-(4-(3-methyl-4-oxo-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine
The mixture of (S)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-amine (0.016 mol), j->-toluene sulfonic acid (0.032 mol) in acetone water (300 ml, 40: 60) mixture was refluxed for 6 hours. The reaction mixture was concentrated under vacuum and treated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford title compound 78% yield. MS (M+l) = 322 (MH+, 100%), M.F.= Cι6H20FN3θ3
Preparation-53 Preparation of (S)-N-{3-[4-(4-(l, 4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide
The suspension of (S)-{3-[4-(4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-azide (0.153 mol), 10% palladium on carbon (7 g), pyridine (0.45 mol), acetic anliydride (0.18 mol) in 700 ml ethyl acetate was stirred at 400 psi hydrogen gas pressure overnight. The suspension was filtered. Filfrate was purified to provide title compound in
70% yield.
MS (M+l) = 408 (MH+, 100%), M.F .= C2oH26FN3O5
Preparation-54
Preparation of (S)-N-{3-[4-(3-methyl-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide
The (S)-N- {3-[4-(4-(l ,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide (0.040 mol), j_>-toluene sulfonic acid (0.080 mol) in acetone water (350 ml, 40: 60) mixture was refluxed for 5 hours. The reaction mixture was concenfrated under vacuum and treated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford keto oxazolidinone acetamide compound 76% yield. MS (M+l) = 364 (MH+, 100%), M.F.= Cι
8H
22FN
3O
4
Preparation-55 Preparation of 4-[3 ,3 -dimethyl-4-oxo-piperidin- 1 -yl]-3-fluoronifrobenzene
The mixture of 3,3-dimethyl-4-piperidone hydrochloride (0.085 mol), triethylamine (0.170 mol), 4-fluoronitrobenzene (0.085 mol) in chloroform was heated under reflux for 16 hours. The solvent was removed under vacuum and to the residue water was added and the precipitate was filtered to afford 4-[3-methyl-4-oxo-piperidin-l-yl]-3-fluoronitrobenzene in 76% yield. MS (M+l) = 267 (MH+, 100%), M.F.= Cι3Hι5FN2O3
Preparation-56 Preparation of 4-[4-(l ,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluoronifrobenzene
The mixture of 4-[3,3-dimethyl-4-oxo-piperidin-l-yl]-3-fluoronifrobenzene from step-1 (0.059 mol), ethylene glycol (0.109 mol) and p-toluenesulphonic acid monohydrate (0.014 mol) in toluene was heated to reflux for 5 hours. The reaction mixture was washed with water. The organic layer was evaporated to afford 4-[4-(l,4-dioxa-3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3- fluoronifrobenzene as a solid in 78% yield.
MS (M+l) = 311 (MH+, 100%), M.F.= C15Hι9FN2O4
Preparation-57 Preparation of [4-(l,4-dioxa-3,3-methyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluorophenyl-4-yl]- aminocarbonyloxymethylj-benzene
The suspension of 4-[4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluoronitrobenzene (0.038 mol), 10% palladium on carbon (0.5 g) in tetrahydrofuran was stirred at room temperature under hydrogen atmosphere (400 psi) overnight.
The reaction mixture filtered to remove the catalyst. To the filtrate, sodiumbicarbonate (0.056 mol) and benzyl chloroformate (0.041 mol) was added at 0-5 °C and stirred at room temperature for 30 minutes. The solvent was evaporated under vacuum and the residue stirred with hexane. The precipitate was filtered to give the title compound in 89% yield. MS (M+l) = 415 (MH+, 100%), M.F.= C23H27FN2O4
Preparation-58 Preparation of (R)-3-{4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)]-3-fluorophenyl)— 2- oxo-oxazolidin-5-ylmethyl}-alcohol
Butyl lithium (1.6 M in hexane, 180 ml) was added to the solution [4-(l,4-dioxa-3,3-dimethyl-8- aza-spiro[4.5]-dec-8-yl)]-3-fluorophenyl-4-yl]-aminocarbonyloxymethyl]-benzene (0.031 mol) in tetrahydrofuran at -78°C. (R)-(-)-Glycidyl butyrate (0.032 mol) was added to the reaction mixture and it was stirred overnight. The reaction mixture was exfracted with the ethyl acetate after quenching with saturated aqueous ammonium chloride solution.
The evaporation of solvent afforded title compound in 88% yield.
MS (M+l) = 381 (MH+, 100%), M.F.= Cι9H25FN2O5
Preparation-59 Preparation of R)- {3-[4-(4-(l ,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-methanesulfonate
The mixture of (R)-{3-[4-(4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-phenyl]-2-oxo- oxazolidin-5-ylmethyl} -alcohol (0.194 mol), triethylamine (0.213 mmol), and methanesulphonyl chloride (0.232 mol) in 700 ml of dichloromethane was stirred for 1 hour. The reaction mixture was washed with 1 liter water. The organic layer was dried and evaporated under vacuum to afford title compound in 87% yield. MS (M+l) = 459 (MH+, 100%), M.F.= C
20H
27F
2O
7S
Preparation-60 Preparation of (S)-{3-[4-(4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-azide
The rmxture of(R)-{3-[4-(4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl} -methanesulfonate (0.16 mol), sodium azide (0.46 mol) in dimethylformamide (200 ml) was heated at 70°C for 14 hours. The reaction mixture was cooled and poured in ice cold water. The precipitate was filtered to provide title compound in 85% yield. MS (M+l) = 406 (MH+, 100%), M.F.= Cι9H24FN5O4
Preparation-61
Preparation of (S)-N-{3-[4-(4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide
The suspension of (S)- {3-[4-(4-(l,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide (0.153 mol), 10% palladium on carbon (7 g), pyridine (0.45 mol), acetic anhydride (0.18 mol) in 700 ml ethyl acetate was stirred at 400 psi hydrogen gas pressure overnight. The suspension was filtered. Filtrate was purified to provide title compound in 70% yield. MS (M+l) = 422 (MH+, 100%), M.F.= C
2ιH
28FN
3O
5
Preparation-62 Preparation of (S)-N-{3-[4-(3,3-dimethyl-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide
The (S)-N- {3-[4-(4-(l ,4-dioxa-3,3-dimethyl-8-aza-spiro[4.5]-dec-8-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide (0.040 mol), ^-toluene sulfonic acid (0.080 mol) in acetone water (350 ml, 40: 60) mixture was refluxed for 5 hours. The reaction mixture was concenfrated under vacuum and treated with saturated aqueous sodiumbicarbonate solution. The precipitate was filtered to afford keto oxazolidinone acetamide compound 76% yield.
MS (M+l) = 378 (MH+, 100%), M.F.= Cι9H24FN3O4
The Table 1 below shows the linezolid MIC values for the sensitive and the corresponding resistant sfrains as well as the site of mutation in their ribosomal RNA.
Table 1
A further embodiment in support of the invention is the result derived from a CoMFA 3D-QSAR study carried out on the compounds of the invention. The study and the results obtained are briefed described below.
Three-dimensional quantitative structure activity relationship of (3D-OSAR) of oxazolidinone antibacterials
Comparative molecular field analysis (CoMFA) a three-dimensional quantitative structure activity relationship technique was applied to series of oxazolidinone antibacterials to understand pharmacophoric factors necessary for optimal activity. CoMFA technique derives the relationship between steric and electrostatic fields of the molecules and their biological activity. Minimum inhibitory concentration against Staphylococcus aureus (MRSA 032 strain) was used as biological activity.
Computational Methods
Study was performed using Sybyl 6.6 (Tripos h e. St. Louis MO) molecular modeling software. All the molecules were constructed and assigned Gasteiger-Huckel charges. All the molecules were minimized using Conjugate Gradient method. Molecules were aligned using rigid fit method with oxazolidinone ring as a template. Biological activity is expressed as log (1/MIC) in molar units. CoMFA fields were calculated for all the molecules and CoMFA field values were
correlated with biological activity using partial-least squares (PLS) method. PLS was used to determine optimum number of components and these were used to calculate r and F - value.
Statistical Results From CoMFA Analysis Of Oxazolidinones.
The results clearly show that in the case of the prior art literature molecules the steric contributions are less than the electrostatic confributions. hi contrast, for the compounds of the present invention as opposed to the prior art, and thus non-obvious, the steric confributions are more than one and half times more than the elecfrostatic contributions.
EXAMPLES
The following examples illustrate the methods of selection of resistant mutant sfrains and are provided only as examples, but not to limit the scope.
Example A
Selection of Linezolid (LNZ) resistant mutants of Methicillin Resistant Staphylococcus aureus-32
(MRSA-32) and Streptococcus pneumoniae 6303 fSPN 6303) in murine infections.
Method
Linezolid resistant mutants of MRSA-32 and SPN 6303 were recovered while studying in vivo efficacy of linezolid in immunocompetent Swiss mice. Infecting doses of organisms given by intraperitoneal route in 5% hog gastric mucin were 103 - 104CFU (colony forming units)/animal for SPN 6303 and 108 CFU/animal for MRSA-32. In case of MRSA-32, Linezolid was administered by oral route, 1 and 4 h post infection, BID (twice a day) for 1 day and for SPN 6303 BID for 2 days. Mice dying at the highest dose of Linezolid were dissected to recover organisms from heart and liver for MRSA-32 and from lung for SPN 6303 by plating on blood agar containing Linezolid at a concentration of 4X MIC ( Minimum Inhibitory Concentration). MICs of Methicillin Resistant Staphylococcus aureus- 32 Linezolid resistant (MRSA-32 LNZR), S. pneumoniae 744 Linezolid resistant ( SPN 744 LNZR) and parent Linezolid sensitive strains MRSA-32 and SPN 6303 were determined for Linezolid by NCCLS agar dilution method. In vivo expression of Linezolid resistance by mutants was further confirmed by determining Linezolid ED50 ( Efficacy dose at which 50% of animals show mortality) values for mutants and parent strains in mouse systemic infection model.
Results
MICs of Linezolid for parent Linezolid sensitive strains of MRSA -32 and SPN 6303 and were
1.56 and 0.8 mcg/ml respectively. However, mutant sfrains MRSA-32 LNZR and SPN 744 LNZR recovered from mice had higher MICs of 50 and 25 mcg/ml respectively. ED50 values of
Linezolid for MRSA -32 and SPN 6303 were raised from 5 and 75 mg/kg to 100 and >200 mg/kg for the corresponding mutant strains.
Example B
Method used for selecting LNZ resistant mutant oi Enter ococcus faecium ATCC 19434
E. faecium ATCC 19434 strain at a cell density of 10^ / ml was inoculated in Mueller Hinton broth medium containing Linezolid at a concentration of 5 and 7.5 meg / ml. The stationary culture was incubated at 37°C and inspected at every 24 hours to assess the formation of turbidity due to the onset of bacterial growth. Following an extended incubation for 96 - 120 hrs turbidity development was noticed in a flask containing medium incorporated with linezolid at 5 & 7.5 meg / ml. A 50 microliter sample of turbid flask was plated on Mueller Hinton agar medium
incorporated with linezolid at 7.5 meg / ml. The agar plates were incubated for 48 hours at 37°C for the formation of discrete colonies of linezolid-resistant strain of E. faecium (E. faecium 361 LNZR ). MICs of resistant mutant E. faecium 361 LNZR and parent strain E. faecium ATCC 19434 were determined for linezolid by NCCLS method.
Results
MIC value of Linezolid sensitive strain E. faecium ATCC 19434 increased from
1.56 mcg/ml to 25 mcg/ml for E. faecium 367 LNZR, on becoming resistant to Linezolid.
Antibacterial activity
The compounds of the invention have distinctive antibacterial activities over the compounds of the prior art. Examples of such activity are provided below. The methods for subjecting the compounds of the invention to various antimicrobial activity tests, in which they exhibited antimicrobial activity are also described.
TEST EXAMPLES Test Example 1 Minimum Inhibitory Concentration (MIC) Determination
Overnight grown cultures of S. aureus organisms in Tryptic Soya broth were diluted in Mueller Hinton Broth to give optical density matching with MacFarland tube 0.5 standard. Cultures were further diluted 1 : 10 in Mueller Hinton broth. Using Denley's mutipoint inoculator, 104 cells were deposited on Mueller Hinton agar ( Difco) containing range of 2 fold dilutions of test compounds. These plates were incubated for 24 hrs at 35°C and MIC results recorded. MIC is defined as minimum drug concenfration that inhibits test organisms. For determining MIC of test compounds against Streptococcus pneumoniae, Mueller Hinton agar containing 5% sheep blood was employed.
The MIC values for representative compounds of the invention against linezolid resistant (LNZR) sfrains S. aureus MRSA-32 LNZR, SPN 744 LNZR and E.faecium 367 LNZR are shown in Table-2
Table - 2 : MICs against linezolid resistant strains (MIC μg/ml)
: not done
Test Example 2
The MIC values for representative compounds of the invention against Linezolid sensitive MRSA- 32, Linezolid sensitive SPN 49619 are shown in Table - 3
Table - 3: MICs against linezolid sensitive sfrains (MIC μg/ml)
Test Example 3 Bactericidal activity Vs Bacteriostatic activity
Killing effect study of linezolid and test compound was carried out against Enterococcus faecalis 416 strain. Test organism was appropriately diluted in 20 ml Muller Hinton broth containing 5 meg /ml of linezolid and test compound in 50 ml conical flasks. The initial inoculum was adjusted to 106 CFU (colony forming units) per ml. The flasks were kept on shaker in close cabinet at 35°C. Aliquots were drawn at 112 hours and cell count was determined to assess the extent of loss in viability.
Results
In case of Linezolid, the initial count of 106 CFU/ml was increased to 109 CFU/ml, indicating that there was no static or cidal action exerted by linezolid. However, flasks containing a representative compound of the invention showed excellent cidal potential by reducing initial count of 106 CFU/ml to 103 CFU/ml. This 3 log kill amounts to 99.9 % kill of test organism.
Table - 4: Bactericidal potential of Compounds of the invention
Low propensity to resistance development
Method
Enterococcus faecalis 416 strain at 108 CFU density level was spirally plated on Mueller Hinton agar containing 5 mcg/ml each of linezolid and representative compound of the invention. Plates were incubated at 35 °C for 48 hours and number of resistant colonies developed in presence of respective compounds were counted by automated colony counter.
Results
Table 5: Low propensity of compounds of invention towards resistance development.
Compounds of the invention show absence of or very low frequency of emergence of mutant resistant colonies, in comparison to linezolid. This result is indicative of the superior curing effect
of the compounds of the invention compared to linezolid for the treatment of enterococcal infections.
Test Example 5
Mutant Prevention Concentration ( MPC) determination Method
Clinical isolate Enterococcus faecalis 416 was spirally plated on Tryptic Soya agar (TSA) plate containing various 2 fold dilutions each of a representative compound of the invention and Linezolid so as to give 109 CFU/plate. After incubation at 35°C for 48 hours, the CFU on each plate was determined. MPC is defined as minimum concentration of drug that prevents mutant colonies on respective antibiotic containing plate. Results
Table 6 : MPC of compounds of invention for Enterococcus faecalis 416 strain
Conclusion The lower MPC values are indicative of superior curing effect of compounds of the invention compared to linezolid in the treatment of enterococcal infections.
The following examples illustrate the methods of preparation of the compounds of the invention and are provided only as examples, but not to limit the scope of the compounds of the invention.
Examples
Example 1 Preparation of (S)-l3-r4-(4-cyanomethylidene-piρeridin-l-yl)-phenyl]-2 -oxo-oxazolidin-5- ylmethyl} -azide
The mixture of triethylamine (13 N.K8 mmol)>, lithθium bromiide (8.2 mmol) and diethylcyanomethylphosphonate (7.2 mmol) in 25 ml tefrahydrofuran was stirred for 20 minutes at room temperature. To the suspension, (S)-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-
5-ylmethyl} -azide
(6.9 mmol) was added. The reaction mixture stirred for 5 hours.
The suspension was filtered and the filtrate was treated with water and exfracted with ethyl acetate.
The combined organic layer was dried and evaporated to give a residue which purified by silica gel column chromatography to provide the titled compound in 91 % yield.
M.P. 80-82°C and MS (M+l) = 339 (MH+, 100%), M.F. = Cι7Hι8N6O2.
Example 2 Preparation of (S)-N-{3-|"4-(4-cvanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -formamide;
Step-1 To a mixture of (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -azide (2.94 mmol), triphenylphosphine (3.81 mmol) was stirred for 3 hours. It was refluxed byadding water overnight. Removal of solvent and purification of the product on silica gel column chromatography provided (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo- oxazolidin-5 -ylmethyl} -amine in 71% yield.
Step-2
The solution of (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- yhnethyl} -amine (2.3 mmol) in ethyl formate (10 ml) was heated at 80 °C over night. The solvent was evaporated under vacuum and the residue obtained was chromatographed over silica gel to afford the titled compound in 68% yield.
MS (M+l) = 341 (MH+, 100%), M.F. = Cι8H20N4O3.
Example 3 Preparation of (S)-N- {3- 4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The titled compound was obtained as per Example-1 by using (S)-{3-[4-(4-oxo-piperidin-l-yl)- phenyl] -2-oxo-oxazolidm-5-ylmethyl} -acetamide in 80% yield. M.P. 168-170°C and MS (M+l) = 355 (MH+, 100%), M.F. = Cι9H22N4O3.
Example 4 Preparation of (S)-l-i3-["4-(4-cvanometfaylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl}-4-carboxyethyl-1.2.3-triazole;
The mixture of compovrnd of Example 1 (2.05 mmol), ethyl propiolate (4.21 mmol), in toluene (10 ml) was heated under reflux for 4 hours. The solvent was removed under vacuum. The residue was triturated with the hexane and filtered to provide a isomeric mixture of two compounds. The column chromatographic purification on silica gel afforded the titled compound in 70% yield. M.P. 120-122°C and MS (M+l) = 437 (MH+, 100%), M.F. = C22H24N6O4.
Example 5 Preparation of (S)-l-{3-["4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyll -4-cyano- 1 ,2,3-triazole;
Step-1
The mixture of (S)- 1 - {3-[4-(4-cyanomethylidene-piperidin-l -yl)-phenyl]-2-oxo-oxazolidin-5- yimethyl}-4-carboxyethyl-l,2,3-triazole (1.14 mmol), ammonium hydroxide (60 ml) in acetonitrile (10 ml) was heated at 50°C for 3 hours. The solvent was removed under reduced pressure. The solid compound obtained was dried under vacuum to afford 4-carboxamide 1 ,2,3-triazole compound in 58% yield. Step-2
To a suspension of the 4-carboxamide 1,2,3-triazole compound (0.82 mmol), pyridine (2.06 mmol), trifluoroacetic anhydride (0.23 ml, 1.65 mmol) in dichloromethane (10 ml) was stirred at room temperature for 15 hours. The reaction mixture treated with saturated aqueous solution of sodium bicarbonate. Organic layer was dried and removal of solvent and purification of residue over a silica gel column chromatography furnished the titled compound in 51% yield.
M.P. 186-188°C and MS (M+l) = 390 (MH+, 100%), M.F. = C20H19N7O2.
Example 6
Preparation of (RV (3-|'4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulphonate;
The (R)-{3-[4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-methanesulphonate was treated with diethylcyanoniethyl phosphonate as per Example 1 to provide titled compound in 42% yield. M.P. 120-122°C and MS (M+l) = 392 (MH+, 100%), M.F. = Cι8H2ιN3O5S.
Example 7
Preparation (S)-N-{3-r4-(4-cyanomethylidene-piperidin-l-yl -3-fluorophenyl1-2-oxo-oχazolidin-5- ylmethyl } -isocyanate;
The (S)-{3-[4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine was was treated with diethylphosphonate as per Example-1 to provide (S)-N-{3-[4-(4- cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -amine in 88% yield. Step-2
Triphosgene (0.580, 1.81 mmol) was added to a solution of compound of step-1 (0.50 g, 1.5 mmol) in 25 ml dichloromethane followed by triethyl amine (3.5 ml) at 0- 5 °C under nitrogen. The reaction mixture was stirred at room temperature for 1 hour. The solvent was evaporated and the residue obtained was chromatographed on the silica gel to afford the titled compound. M.P. 172-174°C and MS (M+l) = 357 (MH+, 100%), M.F. = Cι8Hι7FN4O3.
Example 8 Preparation of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -formamide;
The (S)-N- { 3 -[4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -amine of Example-7, step-1 was subjected to the procedure described in Example -2 step-2 , the title compound was isolated in 48% yield.
M.P. 183-184°C and MS (M+l) = 359 (MH+, 100%), M.F. = Cι8Hι9FN4O3.
Example 9 Preparation of (S)-N- { 3 - f 4-(4-c yanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The intermediate i was treated with diethylcyanomethyl phosphonate as per Example 1 to provide titled compound in 91% yield.
Mp. 159-160 °C 1H-NMR (CDC13, 200 MHz): δ 2.02 (3H, s), 2.50-2.61(2H, m), 2.71-2.82 (2H,m), 3.05-3.29 (4H, m), 3.52-3.81 (3H,m), 3.95-4.11 (lH,m), 4.69-4.85 (IH, m), 5.21(lH,s), 6.19 (lH,t, J= 5.9 Hz),
6.95 (lH,dd, J= 9.2, 9.2 Hz), 7.10 (lH,dd, J= 2.2, 2.2 Hz), 7.45 (lH,dd, J= 2.2, 14.0 Hz).
ESMS m/z 373 (MH+, 100%).
Example 10 Preparation of (S)-N- 13- 4-( 4-cyanomethylidene-piperidin- 1 -yP-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl } -propionamide ;
The mixture of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine ( 1.5 mmol), propionyl chloride (1.79 mmol) in 10 ml pyridine was stirred at room temperature for 3 hours. Evaporation of the solvent under vacuum and silica gel column chromatography afforded the titled compound in 52% yield. Mp. 200-202 °C
1H-NMR (CDC13, 200 MHz): δ 1.10 (3H,t, J = 7.2Hz), 2.23 (2H,q, J= 4.8Hz), 2.50-2.60 (2H,m), 2.70-2.85 (2H,m), 310-3.21(4H,m), 3.59-3.82 (3H,m), 3.95-4.10 (lH,m), 4.70-4.85 (lH,m), 5.20 (lH,s), 6.09 (lH,t,J=5.9Hz), 6.90 (IH, dd, J= 9.2, 9.2 Hz), 7.10 (IH, dd, J= 2.2, 2.2 Hz), 7.44 (lH,dd, J= 2.2, 14.0 Hz). ESMS m z 387 (MH+, 100%).
Example 11 Preparation of (S)-N-(3- 4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-2-dimethylpropionamide;
Following the procedure described in Example 10 and using pivaloyl chloride in the place of propionyl chloride the title compound was isolated in 65% yield. Mp. 198-200 °C 'H-NMR (CDCh, 200 MHz): δ 1.18 (9H,s), 2.45-2.61(2H,m), 2.70-2.85 (2H,m), 3.10-3.25 (4H,m), 3.60-3.85 (3H,m), 3.90-4.10 (lH,m), 4.70-4.85 (lH,m), 5.20 (lH,s), 6.10 (lH,t,J=5.9Hz), 6.90 (IH, dd, J= 9.2, 9.2 Hz), 7.05 (IH, dd, J= 2.2, 2.2 Hz), 7.45 (lH,dd, J= 2.2, 14.0 Hz). ESMS m/z 415 (MH+, 100%).
Example 12 Preparation of (S)-N-{3-r4-(4-cvanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -3 ,3-dimethylbutanamide;
Following the procedure described in Example 10 and using 3, 3 -dimethyl butyryl chloride in the place of propionyl chloride the title compound was isolated in 54% yield. M.P. 210-212°C and MS (M+l) = 431 (MH+, 100%), M.F. = C22H27FN4O .
Example 13
Preparation of (S)-N- {3-[4-(4-cvanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-2-hydroxyacetamide;
The mixture of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine (2.87 mmol), glycolic acid (5.74 mmol), dicyclohexylcarbodiimide (7.17 mmol), 4-dimethylaminopyridine (100 mg) in dichloromethane (50 ml) was stirred for 3
hours. The reaction mixture was filtered. The filfrate was evaporated and the residue was purified by column chromatography over a silica gel to give title compound in 70% yield. M.P. 68-70°C and MS (M+l) = 389 (MH+, 100%), M.F. = Cι9H2ιFN4O4.
Example 14 , Preparation of (S)-N- (3-['4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenvn-2-oχo- oxazolidin-5-ylmethyl}-2-pivolyloxyacetamide;
Following the procedure described in Example 10 and using t-butylcarbonyloxy-methyl chlorofonriate in the place of propionyl chloride the title compound was isolated in 54% yield.
M.P. 91-93°C and MS (M+l) = 473 (MH+, 100%), M.F. = C2 H29FN4O5.
Example 15 Preparation of (S)-N- {3-f4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyll-N-methylacetamide;
The compound obtained in Example -9 (1 mmol) was treated with n-butyl lithium (1.6 M in hexane, 1.6 mmol), methyl iodide (2 mmol) in 10 ml tefrahydrofuran at -78 °C temperature. The reaction mixture was with ethyl acetate water mixture. The organic layer was evaporated to give a crude compound, which was chromatographed on a silica gel to give the titled compound in 44% yield.
Mp. 128-132 °C
1H-NMR (CDC13, 200 MHz): δ 2.15 (3H,s), 2.45-2.65 (2H,m), 2.70-2.90 (2H,m), 3.10-3.30 (7H,m), 3.41-3.60 (lH,m), 3.61-3.80 (lH,m), 3.82-4.10 (2H,m), 4.80-5.00 (lH,m), 5.20 (lH,s), 6.90-7.19 (2H,m), 7.40-7.60(lH,m). ESMS m/z 387 (MH+, 100%).
Example 16
Prer^ration of(S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl} -azide;
The (S)-{3-[4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide was converted to the title compound by using the procedure described in Example- 1 in 40% yield .
M.P. 112-114°C and MS (M+l) = 357 (MH+ 100%), M.F. = Cι7H FN4O .
Example 17 Preparation of (S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine;
The procedure to prepare title compound is described in Example 7. M.P. 162-164°C and MS (M+l) = 331 (MH+, 100%), M.F. = C17Hι9FN4O2.
Example 18 Preparation of (S)-{3-r4-(4-cvanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylaminol -prop-2-ene;
The mixture of (S)-N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine (0.12 mmol), allyl bromide (0.18 mmol), and potassium carbonate (0.25 mmol) in tetrahydrofuran was heated at reflux temperature for 12 hours. Solvent evaporation and purification to provided the title compound in 35% yield. M.P. 102-104°C and MS (M+l) = 371 (MH+, 100%), M.F. = C20H23FN4O .
Example 19 Preparation of (S)-l3-[4-(4-cvanomethylidene-piρeridin-l-yl)-3-fluorophenyl]-2-oxo-oχazolidin-5- ylmethylamino } -nitrile;
Following the procedure described in Example 10 and using cyanogen bromide in the place of propionyl cliloride the title compound was isolated in 66% yield.
M.P. 121-122°C and MS (M+l) = 356 (MH+, 100%), M.F. = Cι8Hι8FN5O2.
Example 20 (S)-{3- 4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidm-5- ylmethylamino } -acetonitrile;
Following the procedure described in Example 10 and using bromoacetotrile in the place of propionyl chloride the title compound was isolated in 55% yield. M.P. 129-131°C and MS (M+l) = 370 (MH
+, 100%), M.F. = Cι
9H
20FN
5O
2.
Example 21 (S)- |3-r4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -methylamine;
The mixture of friethylainine (2.65 mmol), Fmoc L-glycine (2.54 mmol), isobutylchloroformate (2.54 mmol), (S)-N- { 3- [4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin- 5-ylmethyl} -amine (2.12 mmol) in dichloromethane (10 ml) was stirred for 3-5 h at room temperature. The reaction mixture was extracted with dichloromethane and water mixture. The
combined organic layer was dried and evaporation of the solvent afforded a crude compound. It was further purified by column chromatography over a silica gel to give a solid.
The solid was stirred with 40% piperidine in tetrahydrofurane for 2-3 hours. The solvent was removed under reduced pressure and the residue was crystallized to give the title compound in 70% yield.
M.P. 171-172°C and MS (M+l) = 388 (MH+, 100%), M.F. = Cι9H22FN5O3.
Example 22 (S)- {N- {3-[4-(4-cyanomethylidene-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethylamino } N-cyano I -prop-2-ene ;
The compound of Example 18 was treated with cyanogen bromide as per procedure in Example- 10 to give titled compound in 32% yield. M.P. 111-113°C and MS (M+l) = 396 (MH+, 100%), M.F. = C2ιH22FN5O2.
Example 23 S)-N-{3-r4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5-ylmethyl}- 2-cyanoacetamide;
By following the procedure as per Example 13, and by using 2-cyanoacetic acid in the place of glycolic acid, the title compound was isolated in 42% yield.
M.P. 150-152°C and MS (M+l) = 398 (MH+, 100%), M.F. = C20H20FN5O3.
Example 24
(S)-N-{3-[4-r4-cvanometlιylidene-piperidin-l-yl)-3-fluoiOphenvn-2-oxo-oxazolidin-5-ylmethyll- 2-oxo-oxazolidin-4-yl-carboxamide.-
Step-1 By following the procedure as per Example 21, and by using L-serine in the place of L-glycine the 2-amino-3-hydroxypropionaιnide compound was isolated in 42% yield. Step-2
The solution of above compound (0.25 mmol), carbonyldiimidazole (0.095 g, 0.58 mmol) in dry tefrahydrofuraii (10 ml) was stirred for 18 hours. Evaporation of the solvent gave crude solid, which was purified by column chromatography over a silica gel to give the title compound in 84% yield.
M.P. 145°C and MS (M+l) = 444 (MH+, 100%), M.F. = C2ιH22FN5O5.
Example 25 (S)-N-{3-[4-(4-cvanomethylidene-piperidin-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5-ylmethyl}- pyrrolidin-2-carboxamide;
By following the procedure of Example 21, and by using L-proline in the place of L-glycine the title compound was isolated in 42% yield. M.P. 155°C and MS (M+l) = 428 (MH+, 100%), M.F. = C22H26FN5O3.
Example 26 (S)- 1 - { 3 -[4-(4-cyanomethylidene-piperidin- 1 -vD-3 -fluorophenyl] -2-oxo-oxazolidin-5-ylmethyl I -4- carboethoxy-1 ,2,3-triazole;
The title compound was prepared by using compound of Example 16 and following the procedure of Example -4 in 64% yield.
M.P. 164-166°C and MS (M+l) = 455 (MH+, 100%), M.F. = C22H23FN6O4.
Example 27 (S)-l-{3-[4-(4-cvanomethylidene-piperidm-l-yl)-3-fluoiOphenyl]-2-oxo-oxazolidin-5-ylmethyll-5- carboethoxy-1 ,2,3-triazole;
The title compoxmd was prepared by using the compound of Example 16 and following the procedure of Example -4 in 26% yield. M.P. 70-72°C and MS (M+l) = 455 (MH+, 100%), M.F. = C
22H
23FN
6O
4.
Example 28 (S)- 1 - {3 -[4-(4-cvanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl| -4- cyano-1 ,2,3-triazole;
The title compound was prepared by using compound of Example 26 and following the procedure of Example -5 in 45% yield. M.P. 78-80°C and MS (M+l) = 408 (MH+, 100%), M.F. = C
0Hι
8FN
7O
2.
Example 29 (S)-N- {3-[4-(4-cvanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll - difluoroacetamide;
The mixture of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine (0.5 g, 1.45 mmol), oxalyl chloride (0.150 ml, 1.74 mmol), difluoroacetic acid (0.1 ml, 1.74 mmol) and triethyl-imine (0.242 ml, 1.74 mmol) in dicliloromethane (20 ml) was stirred for lhour at room temperature. The reaction mixture was exfracted with the dichloromethane water mixture. The combined organic layer was dried and removal of the solvent afforded a residue which was chromatographed over silica gel to give title compound in 48% yield.
M.P. 162-164°C and MS (M+l) = 409 (MH+, 100%), M.F. = Cι9Hι9F3N4O3.
Example 30
(S)-N-(3-["4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- trifluoroacetamide;
The title compound was prepared by following the procedure of Example -29 and by using trifluoroacetic acid in the place of difluoroacetic acid in 45% yield.
Mp. 181-183 °C
1H-NMR (CDC13, 200 MHz): δ 2.50-2.60 (2H,m), 2.70-2.85 (2H,m), 310-3.21 (4H,m), 3.59-3.80
(2H,m) 3.82-4.00 (lH,m), 4.10-4.20 (lH,m), 4.75-4.95 (lH,m), 5.20 (lH,s), 6.92 (IH, dd, J= 9.2, 9.2 Hz), 7.05 (IH, dd, J= 2.2, 2.2 Hz),7.20-7.30 (lH,m), 7.42 (lH,dd, J= 2.2, 14.0 Hz).
ESMS m/z 427 (Mf^, 100%).
Example 31 (S)-N- {3- 4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll - chloroacetamide:
The title compound was prepared by following the procedure of Example -29 and by using chloroacetic acid in the place of difluoroacetic acid in 77% yield.
MS (M+l) = 407 (MH+, 100%), M.F. = C19H20FN4O3C1.
Example 32 (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- dichloroacetamide:
The title compound was prepared by following the procedure of Example -29 and by using dichloroacetic acid in the place of difluoroacetic acid in 39% yield.
M.P. 159-16PC and MS (M+l) = 442 (MH+, 100%), M.F. = Cι9H19FN4O3Cl2.
Example 33 fS)-N-{3-r4-f4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll- trichloroacetamide;
The title compound was prepared by following the procedure of Example -29 and by using trichloroacetic acid in the place of difluoroacetic acid in 42% yield. M.P. 142-144°C and MS (M+l) = 476 (MH+, 100%), M.F. = Cι9Hι8FN4O3Cl3.
Example 34 (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll- bromoacetamide;
The title compound was prepared by following the procedure of Example -10 and by using bromoacetylbromide in the place of propionyl chloride in 77% yield.
M.P. 160-162°C and MS (M+l) = 452 (MH+, 100%), M.F. = Cι9H20FN4O3Br.
Example 35 (S)-N-{3-r4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- dibromoacetamide;
The title compound was prepared by following the procedure of Example -29 and by using dibromoacetic acid in the place of difluoroacetic acid in 57% yield.
MS (M+l) = 531 (MH+, 100%), M.F. = Cι9Hι9FN4O3Br2.
Example 36 (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- iodoacetamide;
The title compound was prepared by following the procedure of Example -10 and by using iodoacetyliodide in the place of propionyl chloride in 29% yield. M.P. 178-180°C and MS (M+l) = 499 (MH+, 100%), M.F. = C,9H20FN4O3I.
Example 37 (S)-N-|3-[
"4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- 4-methylphenylsulphonamide;
The title compound was prepared by following the procedure of Example -10 and by using p- toluene sulfonylchloride in the place of propionyl chloride in 69% yield.
Mp. 180-182 °C 1H-NMR (CDC13, 200 MHz): δ 2.45 (3H,s), 2.50-2.60 (2H,m), 2.70-2.85 (2H,m), 3.09-3.40 (64,m),
3.85-4.10 (2H,m), 4.65-4.80 (lH,m), 5.20 (lH,s), 6.90 (IH, dd, J= 9.2, 9.2 Hz), 7.05 (IH, dd, J=
2.2, 2.2 Hz),
7.25-7.39 (4H,m), 7.40 (lH,d, J=2.2,9.2Hz).
ESMS m/z 485 (MFT, 100%). Example 38
(S)-N-{3-|"4-(4-cyanomethylidene-piperidin-l-yl -3-fluorophenyn-2-oxo-oxazolidin-5-ylmethyl}- methylcarbamate;
The mixture of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-isocyanate (1.0 mmol), sodium methoxide (1.2 mmol) in methanol (10 ml) was stirred for 2 hours at room temperature. The reaction mixture was exfracted with the ethyl acetate water mixture. The organic extract was dried and removal of solvent afforded title compound in 75% yield.
MS (M+l) = 389 (MH+, 100%), M.F. = Cι9H2ιFN4O4.
Example 39 (S)-N- (3-|"4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll - ethylcarbamate;
The title compound was prepared by following the procedure of Example -38 and by using sodium ethoxide in the place of sodium methoxide in 54% yield.
MS (M+l) = 403 (MH+, 100%), M.F. = C20H23FN4O4.
Example 40
(S)-N-{3-f4-f4-cyanomethylidene-piperichn-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- isopropylcarbamate;
The title compound was prepared by following the procedure of Example -10 and by using isopropylchloroformate in the place of propionyl chloride in 48% yield.
202-204°C and MS (M+l) = 431 (MH+, 100%), M.F. = C22H27FN4O4.
Example 41 (2S,5S)-{N-(3-[4- 4-cvanomethylidene-piperidm-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -propionamid-2-yl } -amine;
The title compound was prepared by following the procedure of Example -21 and by using L- alanine in the place of L-glycine in 67% yield.
M.P. 103-105 °C and MS (M+l) = 402 (MH+, 100%), M.F. = C20H24FN5O3.
Example 42 (2S, 5S)- {N- {3-f4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl}-3-hydroxypropionamid-2-yl}-amine;
The procedure to prepare the title compound is described in Example 24. M.P. 88-90°C and MS (M+l) = 418 (MH+, 100%), M.F. = C
20H
24FN
5O
4.
Example 43 (2S,5S)- {N- {3-r4-(4-cvanomethylidene-piperidin- 1 -yl)-3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl} -3 -(imidazol-4-yl)-propionamid-2-yl} -amine;
The title compound was prepared by following the procedure of Example -21 and by using L- Mstidine in the place of L-glycine in 63% yield. M.P. 93-96°C and MS (M+l) = 468 (MH+, 100%), M.F. = C23H26FN7O3.
Example 44 (S)-N- {3- 4-(4-cyanomethylidene-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl| - 1-pthalamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)-{3- [4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-pthalamide in 67% yield.
M.P. 183-185°C and MS (M+l) = 461 (MH+, 100%), M.F. = C25H2ιFN4O4.
Example 45
(S)-N- (3-r4-(4-cvanomethylidene-ρiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidm-5-ylmethyl j - thioacetamide;
A mixture of the compound of Example- 9 (0.26 mmol), Lawesson's reagent (0.40 mmol) in dioxane (10 ml) was stirred at 100 °C for one hour. The reaction mixture was concentrated in vacuo to give a residue. The residue was purified by silica gel column chromatography to afford title compound in 82% yield. Mp. 190-192 °C
1H-NMR (CDC13, 200 MHz): δ 2.50-2.70 (5H, m), 2.76-2.90 (2H, m),3.10-3.3.0 (4H, m), 3.75-3.90
(IH, m), 4.00-4.20 (2H, m), 4.25-4.40 (IH, m), 4.90-5.10 (lH,m), 5.21(lH,s), 6.90-7.10 (2H,m),
7.45 (lH,dd, J= 2.2, 14.0 Hz), 8.10-8.30 (IH, s).
ESMS m/z 389 (MH+, 100%). Example 46
(S)-N-l3- 4-r4-cvanomethylidene-piperidin-l-yl)-3-fluorophenyl1-2-oxo-oxazolidin-5-ylmethyll- methylthiocarbamate;
Stepl The mixture of (S)-N-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-amine (10 mmol), triethylamine (10 mmol) and carbon disulphide (20 mmol) in tetrahydrofuran (50 ml) was stirred for 4 hours at 0°C.
To the solution ethyl chloroformate (5.8 mmol) was added and stirred for 1 hour. The reaction mixture washed with water followed by brine and dried over sodium sulfate. The evaporation of solvent gave (S)-N- {3-[4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-
5-ylmethyl}-thioisocyanate in 69% yield.
Step-2
Thioisocyanate compound was treated with sodium methoxide as per procedure described in Example-38 to afford the title compound in 77% yield.
M.P. 147-148°C and MS (M+l) = 405 (MH+, 100%), M.F. = Cι9H2,FN4O3S.
Example 47
(S -N-{3-|"4-(4-cyanomethylidene-piperidin-l-ylV3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- 2-hydroxythioacetamide;
The title compound was prepared by following the procedure of Example -45 and by using compound of Example- 13 in 42% yield.
M.P. 102-105°C and MS (M+l) = 405 (MH+, 100%), M.F. = Cι9H2ιFN4O3S.
Example 48 (S)-N-(3- 4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- 2-hydroxyethylthiocarbamide;
The title compound was prepared by following the procedure of Example -46, step-2 by using aminoethanol in place of sodium methoxide in 36% yield.
M.P. 168-170°C and MS (M+l) = 434 (MH+, 100%), M.F. = C20H24FN5O3S.
Example 49 (S)-N-{3-["4-(4-cyanomethylidene-piperidin-l-yl -3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -thiocarbonylmethylamine;
The title compound was prepared by following the procedure of Example -45 and by using S)-N- {3-[4-(4-cymome1frylidene-piperi(hn-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethylamino}- c-trbonylmethylamine in 38% yield.
M.P. 157-158°C and MS (M+l) = 404 (MH+ 100%), M.F. = Cι9H22FN5O2S.
Example 50 (S)-N- {3-[4-(4-cyanomethylidene-piρeridin- 1 -ylV3-fluorophenyl1-2-oxo-oxazolidin-5-yhnethyl} - 2-dimethylaminoethylthiocarbamide;
The title compound was prepared by following the procedure of Example -46, step-2 by using dimethylaminoethylamine in place of sodium methoxide in 40% yield.
M.P. 153-155°C and MS (M+l) = 461 (MH+, 100%), M.F. = C22H29FN6O3.
Example 51 (S)-N- {3-[4-(4-cyanomethylidene-piρeridin- 1 -ylV 3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl) - thiocarbamide;
The title compound was prepared by following the procedure of Example —46, step-2 by using sodamide in place of sodium methoxide in 80% yield. M.P. 190-191°C and MS (M+l) = 390 (MH+ 100%), M.F. = Cι8H20FN5O2S.
Example 52
(S)-N-{3-|"4-(4-cyanom neetthhyylliiddeennee--ppiippeerriiddimr-l-yl)-3-fluoiOphenyl1-2-oχo-oxazolidm-5-ylmethyl}- methylthiocarbamide;
The title compound was prepared by following the procedure of Example -46, step-2 by using methylamine in place of sodium methoxide in 65% yield.
M.P. 191-192°C and MS (M+l) = 404 (MH+, 100%), M.F. = Cι9H22FN5O2S.
Example 53 (S)-N- |3-r4-(4-cvanomethylidene-piperidin- 1 -ylV 3-fluorophenyl]-2-oxo-oxazolidm-5-ylmethyll - methanesulphonamide:
The title compound was prepared by following the procedure of Example -10 and by using methanesulfonylchloride in the place of propionyl chloride in 88% yield.
Mp. 235-237 °C
1H-NMR (CDC13, 200 MHz): δ 2.45-2.60 (2H,m), 2.70-2.85 (2H,m), 3.09 (3H,s), 3.10-3.30 (4H,m), 3.35-3.65 (2H,m), 3.85-4.15 (2H,m), 4.70-5.00 (lH,m), 5.20 (lH,s), 6.90 (IH, dd, J= 9.2,
9.2 Hz), 7.10 (IH, dd, J= 2.2, 2.2 Hz), 7.42 (lH,dd, J= 2.2, 14.0 Hz).
ESMS m/z 409 (MH+, 100%).
Example 54
(R)- (3 - |"4-(4-cyanomethylidene-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5-ylmethyl I - methanesulphonate;
The title compound was prepared by following the procedure of Example -1 and by using (R)-{3- [4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-methanesulphonate in 88% yield. M.P. 126-128°C and MS (M+l) = 410 (MH+ 100%), M.F. = Cι8H20FN3O5S.
Example 55 (S)-N-{3-r4-(4-cyanomethylidene-3-fluoroρiρeridin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)-{3- [4-(3-fluoro-4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 76% yield. MS (M+l) = 373 (MH
+, 100%), M.F. = Cι
9H
2ιFN
4O
3.
Example 56 E/Z mixture/E and Z isomer of (S)-N-{3-["4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3- fluorophenyl1-2-oxo-oxazolidin-5-ylmethyl|-acetamide;
The title compound was prepared by following the procedure of Example -1 and by using S)-{3- [4-(3-fluoro-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 71% yield as a mixture of isomers.
M.P. 100-102°C and MS (M+l) = 391 (MH+, 100%), M.F. = Cι9H20F2N4O3.
The mixture of compound was separated on preparative HPLC to provide E isomer in 56% yield as a white solid.
M.P. 138-140°C and MS (M+l) = 391 (MH+, 100%), M.F. = Cι9H20F2N4O3.
The mixture of compound was separated on preparative HPLC to provide Z isomer in 18% yield as a white solid. M.P. 170-172°C and MS (M+l) = 391 (MH+, 100%), M.F. = Cι9H20F2N4O3.
Example 57 SVN-(3-r4-(4-cvanomethylidene-3-fluoropiρeridin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl! -difluoroacetamide;
Step-1
Preparation of (S)-N-{3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine: The compound was prepared by following the procedure of Example -1 and by using (S)- {3-[4-
(3,3--hfluoro-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-aιιιine in 77% yield.
Step-2
The title compound was prepared by following the procedure of Example -29 and by using (S)-N- {3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- amine in 80% yield.
M.P. 136-138°C and MS (M+l) = 427 (MH+, 100%), M.F. = C19H18F4N4O3.
Example 58 (S)-N-{3-r4-(4-cvanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -isobutylcarbamate;
The title compound was prepared by following the procedure of Example -10 and by using (S)-N- {3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- amine and isobutylchloroformate in 69% yield.
M.P. 162-164°C and MS (M+l) = 448 (MH+, 100%), M.F. = C22H26F2N4O4.
Example 59 (R)-|3-[4-(4-cvanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulphonate:
The title compound was prepared by following the procedure of Example -1 and by using (R)- {3- [4-(3-fluoro-4-oxo-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} - methanesulphonate in 59% yield. M.P. 116-118°C and MS (M+l) = 428 (MH+, 100%), M.F. = Cι8Hι9F2N3O5S.
Example 60 (S)-N-|3-("4-(4-cyanomethylidene-3-fluoropiperidi-ι-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
The title compound was prepared by following the procedure of Example -45 and by using (S)-N- {3-[4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 56% yield. M.P. 137-138°C and MS (M+l) = 405 (MH+, 100%), M.F. = Cι9H2oF2N4θ2S.
Example 61 (S)-N-|3-r4-(4-cvanomethylidene-3,3-difluoropiperidin-l-yl)-phenyll-2-oxo-oxazolidin-5- yimethyl} -acetamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)- {3 - [4-(3,3-difluoro-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 88% yield. MS (M+l) = 409 (MH+ 100%), M.F. = Cι
9Hι
9F
3N
4O
3.
Example 62 S
)-N-{3-[4-
(4-cyanomethylidene-3-methylpiperidin-l-ylV3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl } -formamide;
Preparation of (S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine:
The compound was prepared by following the procedure of Example -1 and by using (S)-{3-[4- (3,3-mΕuoro-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidm-5-ylmethyl}-amine in 80% yield.
Step-2
The title compound was prepared by following the procedure of Example -2 and by using (S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine in 59% yield.
M.P. 98-100°C and MS (M+l) = 373 (MH+, 100%), M.F. = Cι9H2ιFN4O3.
Example 63 (S)-N-{3- 4-(4-cvanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylj -acetamide;
The title compoxmd was prepared by following the procedure of Example -1 and by using (S)-{3- [4-(3,3-difluoro-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 62% yield.
M.P. 152-153°C and MS (M+l) = 387 (MH+, 100%), M.F. = C20H23FN4O3.
Example 64
(S)-N-i3-r4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl1-2-oxo-oxazolidin-5- ylmethyl} -trifluoroaceta ide;
The title compound was prepared as per Example -29 by using (S)-N- {3-[4-(4-cyanomethylidene- 3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -amine and trifluoroacetic acid in 51% yield.
MS (M+l) = 441 (MH+, 100%), M.F. = C20H2oF4N4O3.
Example 65
(S)-N-{3-r4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-cyanoacetamide;
The title compound was prepared as per Example -29 by using (S)-N-{3-[4-(4-cyanomethylidene- 3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and cyanoacetic acid in 81% yield. M.P. 102-104°C and MS (M+l) = 412 (MH+, 100%), M.F. = C2ιH22FN5O3.
Example 66 (S)-2-{3-("4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } - 1 , 3-thiazole;
The mixture of (S)-N- {3-[4-(4-cyanomethylidene-3-methylpiperidin-l -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -thiocarbamide (0.5 mmol), acetaldehyde diethylacetal (0.6 mmol) and p-
toluene-sulphonic acid (0.05mmol) in acetic acid (5 ml) was heated at 70°C for 1 hour. The reaction mixture extracted with ethyl acetate water mixture. The organic layer treated with sodium bicarbonate dried. The evaporation of the solvent silica gel column chromatographic purification afforded title compound in 60% yield. M.P. 75-77°C and MS (M+l) = 428 (MH+, 100%), M.F. = C2ιH22FN5O2S.
Example 67 (S)-N-{3-[4-(4-cvanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyll -methylthiocarbamate;
The title compound was prepared as per procedure described in Example -46 in by using (S)-N-{3-
[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluoiOphenyl]-2-oxo-oxazolidin-5-ylmethyl}- methylthiocarbamate in 46% yield.
M.P. 78-80°C and MS (M+l) = 419 (MH+ 100%), M.F. = C20H23FN4O3S.
Example 68 (S)-N-{3-r4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thiocarbamide;
The title compound was prepared as per procedure described in Example -51 in by using (S)-N-{3-
[4-(4-cyanomethylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- thioisocyanate in 79% yield.
M.P. 142-144°C and MS (M+l) = 404 (MH+, 100%), M.F. = C]9H22FN5O2S.
Example 69
(S)-Nr(3-[4-(4-cyanome1frylidene-3-methylpiperidin-l-yl)-3-fluorophenyl]-2-oχo-oχazolidin-5- ylmethyl} -methylthiocarbamide;
Nc _ _ H H
H3C F S
The title compound was prepared as per procedure described in Example -52 in by using (S)-N-{3- [4-(4-cyanomethylidene-3 -methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5-ylmethyl} - thioisocyanate in 45% yield.
M.P. 180-182°C and MS (M+l) = 418 (MH+, 100%), M.F. = C 0H24FN5O2S.
Example 70 (S)-N-{3-[4-(4-cyanomethylidene-3,3-dimethylpiρeridin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)-{3- [4-(3,3-dimethyl-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 78% yield.
M.P. 150-152°C and MS (M+l) = 401 (MH+, 100%), M.F. = C2ιH25FN4O3.
Example 71 rR)-i3-r4-(4-cyanomethylidene-3,3-dimethylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -methanesulphonate;
The title compound was prepared by following the procedure of Example -1 and by using (S)-{3- [4-(3,3-dimethyl-4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- methanesulfonate in 69% yield. MS (M+l) = 438 (MH+, 100%), M.F. = C20H24FN3O5S.
Example 72 (S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl -phenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)- {3- [4-(4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide and diethyl-(l- cyanoethyl)-phosphonate in 65% yield.
M.P. 94-96°C and MS (M+l) = 369 (MH+ 100%), M.F. = C20H24N4O3.
Example 73
(S)-N- 13-[4-(4-( 1 -cyanoethylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -formamide;
Step-1 Prepartion of(S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -amine:
The compound was prepared by following the procedure of Example -1 and by using (S)-{3-[4-(4- oxo-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -amine and diethyl-(l - cyanoethyl)-phosphonate in 74% yield. Step-2
The title compound was prepared by following the procedure of Example -2 and by using (S)-N-
{3_.[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 66% yield.
M.P. 188-190°C and MS (M+l) = 373 (MH+, 100%), M.F. = C19H2ιFN4O3.
Example 74
(S)-N-|3-r4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluoroρhenyll-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The title compound was prepared by following the procedure of Example -1 and by using (S)- {3- [4-(4-oxo-piperidin- 1 -yl)-3 -fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide and diethyl-( 1 - cyanoethyl)-phosphonate in 80% yield.
M.P. 181-182°C and MS (M+l) = 387 (MH+, 100%) M.F. = C 0H23FN4O3.
Example 75 (S)-N-{3- 4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -2-cyanoacetaιτιide;
The title compoxmd was prepared by following the procedure of Example -23 and by using (S)-N- {3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 80% yield.
M.P. 194-196°C and MS (M+l) = 412 (MH+ 100%), M.F. = C21H22FN5O3.
Example 76
(S)-N-{3-("4-(4-(l-cy-vnoethylidene -piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylamino } -carboxymethylamine;
The title compound was prepared by following the procedure of Example -21 and by using (S)-N- f3-.[4-.(4.(l-.cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 63% yield.
M.P. 141-142°C and MS (M+l) = 402 (MH+, 100%), M.F. = C20H24FN5O3.
Example 77 (S)-N-{3-r4-(4-(l-cvanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -difluoroacetamide;
The title compoimd was prepared by following the procedure of Example -29 and by using (S)-N-
{3-[4-(4-(l-cyanoemylidene)-piperidin-l-yl)-3-fluoiOphenyl]-2-oxo-oxazolidin-5-yhnethyl}-amine in 75% yield. M.P. 172-174°C and MS (M+l) = 423 (MH+, 100%), M.F. = C20H21F3N4O3.
Example 78 (S)-N-l3-r4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -chloroacetamide;
The title compound was prepared by following the procedure of Example -31 and by using (S)-N-
{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 57% yield. .
MS (M+l) = 421 (MH+, 100%), M.F. = C20H22F 4O3CI.
Example 79 fS)-N-{3-|"4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichloroacetamide;
The title compound was prepared by following the procedure of Example -29 and by using (S)-N-
{3-[4-(4-(l-cymoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and dichloroacetic acid in 63% yield.
M.P. 202-204°C and MS (M+l) = 455 (MH+, 100%), M.F. = C20H2ιFN4O3Cl2.
Example 80 (S)-N-{3-r4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl) -trichloroacetamide:
The title compound was prepared by following the procedure of Example -29 and by using (S)-N- {3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and trichloroacetic acid in 45% yield.
M.P. 166-168°C and MS (M+l) = 489 (MH+, 100%), M.F. = C20H20FN4O3C13.
Example 81
(S)-N-{3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethylj -isobutylcarbamate;
The title compound was prepared by following the procedure of Example -58 and by using (S)-N- {3-[4-(4-(l-cyanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 63% yield. M.P. 158-160°C and MS (M+l) = 445 (MH+, 100%), M.F. = C23H29FN4O4.
Example 82 (R)-{3-f4-(4-(l-cvanoethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyll- alcohol;
The title compound was prepared by following the procedure of Example -1 and by using (R)-{3- [4-(4-oxo-piperidin- 1 -yl)-3 -fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -alcohol and diethyl( 1 - cyanoethyl)-phosphonate in 63% yield.
M.P. 100-102°C and MS (M+l) = 346 (MH+, 100%), M.F. = Cι8H20FN3O3.
Example 83 (R -3-{3-r4-(4-(l-cvanoethylidene)-piperidin-l-yl)-3-fluoiOphenyl]-2-oxo-oxazolidin-5- ylmethyloxy}-iso-oxazole;
The mixture of diisopropyldiazodicarboxylate (3.75 mmol), (R)-{3-[4-(4-(l-cyanoethylidene)- piperidin- 1 -yl)-3 -fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -alcohol (3.13mmol), triphenylphosphine (3.44 mmol) and 3-hydroxy-isoxazole (3.44 mmol) in tefrahydrofuran (20 ml) was stirred for 2 hours. The reaction mixture was extracted with the ethyl acetate water mixture and the combine organic layer was dried. Evaporation of the solvent afforded a sticky solid, which was upon silica gel colmn chromatography afforded the title compound in 70% yield.
M.P. 137-138°C and MS (M+l) = 413 (MH+, 100%), M.F. = C2ιH2ιFN4O4.
Example 84 (S)-N- |3-[4-(4-f 1 -cyanoethylideneVpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -thioacetamide;
The title compound was prepared as per procedure described in Example —45 by using (S)-N-{3- [4-(4-( 1 -cyanoethylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5-ylmethyl } - acetamide in 56% yield.
M.P. 171-172°C and MS (M+l) = 403 (MH+, 100%). M.F. = C2oH23FN4O2S
Example 85 E/Z mixture of (S)-N-(3-r4-(4-fl-cyanoethylideneV3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide:
The title compound was prepared as per procedure described in Example -1 by using (S)-{3-[4-(3- fluoro-4-oxo-piperidin- 1 -yl)-3-fluoroρhenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide and diethyl- (l-cyanoethyl)-phosphonate in 75% yield as a isomeric mixture. M.P. 148-150°C and MS (M+l) = 405 (MH+, 100%), M.F. = C20H22F2N4O3.
Example 86 E-(S)-N-{3-l4-(4-(l-cvaιιoethylidene)-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
Isomeric mixture obtained as per Example-85 was separated on preparative HPLC to provide title compound in 42% yield.
M.P. 148-150°C and MS (M+l) = 405 (MH+, 100%), M.F. = C20H22F2N4O3.
Example 87
Z-(S)-N-{3-("4-(4-(l-cyanoethylidene)-3-fluoropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl } -acetamide;
Isomeric mixture obtained as per Example- 85 was separated on preparative HPLC to provide title compound in 22% yield.
M.P. 148-150°C and MS (M+l) = 405 (MH+, 100%), M.F. = C20H22F N4O3.
Example 88 SVN-(3-r4-(4-(l-cyanopropylidene)-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyll- acetamide;
The title compoimd was prepared as per procedure described in Example -1 by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazoliolin-5-ylmethyl}-acetamide and diethyl-(l-cyanopropyl)- phosphonate in 78% yield.
MS (M+l) = 383 (MH+, 100%), M.F. = C21H26N4O3.
Example 89 (S)-N-{3-r4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example -1 by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide and diethyl-(l- cyanopropyl)-phosphonate in 61% yield. Mp. 185-186 °C
1H-NMR (CDC13, 200 MHz): δ 1.20 (3H, t, J= 5.00 Hz), 2.05 (3H,s), 2.30 (2H, q, J = 6.5 Hz), 2.50-2.65 (2H,m), 2.75-2.85 (2H, m), 3.00-3.25 (3H, m), 3.95-4.15 (lH,m), 4.65-4.85 (lH,m), 6.05-6.10 (lH,m), 6.90 (IH, dd, J= 9.2, 9.2 Hz), 7.05 (IH, dd, J= 2.2, 2.2 Hz), 7.41 (lH,dd, J= 2.2, 14.0 Hz).
ESMS m/z 401 (MH+, 100%).
Example 90
(S)-N-(3-r4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -dichloroacetamide;
Step-1 Preparation of (S)-N-{3-[4-(4-cyanopropylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-
5-ylmethyl}-amine:
The compound was prepared by following the procedure of Example -1 and by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 75% yield.
Step-2 The title compound was prepared by following the procedure of Example -29 and by using (S)-N-
{3-[4-(4-cyanopropylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and dichloroacetic acid in 50% yield.
M.P. 214-216°C and MS (M+l) = 469 (MH+, 100%), M.F. = C2ιH23FN4O3Cl2.
- Example 91
(S)-N-{3-r4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl) -trichloiOacetamide;
The title compound was prepared by following the procedure of Example -29 and by using (S)-N- {3-[4-(4-cyanopropylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and trichloroacetic acid in 63% yield.
M.P. 150-152°C and MS (M+l) = 503 (MH+, 100%), M.F. = C2ιH22FN4O3Cl3.
Example 92
(
rS)-N-{3-r4-(4-(l-cyanopropylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyll -bromoacetamide;
The title compoxmd was prepared by following the procedure of Example -10 and by using (S)-N-
{3-[4-(4-cyanopropylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine and bromoacetyl bromide in 65% yield. M.P. 200-203°C and MS (M+l) = 480 (MH+, 100%), M.F. = C2ιH24FN4O3Br.
Example 93 (S)-N- (3-[4-(4-(l -cyanopropylidene)-piperidin-l -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl) -thioacetamide;
The title compound was prepared by following the procedure of Example -45 and by using (S)-N- {3-[4-(4-(l-cyanopiOpylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 55% yield.
M.P. 174-176°C and MS (M+l) = 417 (MH+, 100%), M.F. = C2ιH25FN4O S
Example 94 (S)-N-{3-[4-(4-(l-cyano-cyclopropylmethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The mixture of (S)-N-{3-[4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide (2.86 mmol), cyclopropylacetonitrile (5.73 mmol), ammonium acetate (catalytic) in 100 ml of toluene was heated at reflux temperature for 5 to 6 hours. The reaction mixture was cooled to room temperature and exfracted with ethyl acetate water mixture, dried and evaporated to give
crude product. The crude product was recrystallized from ethyl acetate to furnish title compound in
69% yield.
Mp: 175-79 °C
1H-NMR (CDC13, 200 MHz): δ 0.70-0.80 (2H, m), 0.81-1.00 (2H, m), 1.60-1.70 (IH, m), 2.05 (3H, m), 2.65-2.80 (4H, m), 3.01-3.30 (4H, m), 3.60-3.81 (IH, m), 3.85-4.15 (3H, m), 4.71-4.85 (IH, m), 6.97 (IH, m), 7.10 (IH, m), 7.45 (IH, m).
ESMS m/z 413 (MH+, 100%).
Example 95
(S)-N-{3-[4-(4-(l-cvano-3-ene-butylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example -1 by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide and diethyl-(l- cyano-3-ene-butyl)-phosphonate in 67% yield. Mp. 189-191 °C
1H-NMR (CDC13, 200 MHz): δ 2.01(3H, s), 2.51-2.61 (2H, m), 2.78-2.90 (2H, m), 3.00-3.20 (6H, m), 3.55-3.82 (3H, m), 3.95-4.10 (IH, m), 4.70-4.85 (IH, m), 5.10-5.22 (2H, m), 5.70-5.90 (lH,m),
6.00-6.10 (IH, m), 6.90 (IH, m), 7.05 (IH, m), 7.44 (IH, m).
ESMS m/z 413 (MH+, 100%). Example 96
(S)-N- 13 - [4-(4-( 1 -cyano-3- we-butylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylrnethyl} -acetamide;
The title compound was prepared as per procedure described in Example -1 by using (S)-{3-[4-(4- oxo-piperidin-1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide and diethyl-(l - cyano-3-yne-butyl)-phosphonate in 54% yield.
M.P. 171-172°C and MS (M+l) = 411 (MH+, 100%), M.F. = C22H23FN4O3.
Example 97 (S)-N- {3-[4-(4-(l -cyano-2-phenyl-ethylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oχazolidin-5- ylmethyl j -acetamide;
The title compound was prepared as per procedure described in Example -1 by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide and diethyl-(l- cyano- l-benzylmethyl)-phosphonate in 40% yield.
Mp. 98-100 °C
1H-NMR (CDC13, 200 MHz): δ 2.05 (3H, s), 2.60-2.79 (2H, m), 2.80-2.90 (2H, m), 3.05-3.21 (4H, m), 3.50-3.80 (5H, m), 3.95-4.10 (IH, m), 4.70-4.85 (IH, m), 6.00 (IH, t), 6.90 (IH, m), 7.05 (IH, m),7.20-7.39 (6H, m), 7.41 (IH, m).
Example 98
(S)-N- {3-r4-(4-(l -cyano-1 -phenyl-methylidene)-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-
5-ylmethylj -acetamide;
The title compound was prepared as per procedure described in Example -94 by using phenylacetonitrile in the place of cyclopropylacetonitrile in 62% yield. Mp.200-205 °C 1H-NMR (CDC13, 200 MHz): δ 2.01 (3H, s), 2.60-2.70 (2H, m), 2.90-3.10 (4H, m), 3.21-3.35 (2H, m), 3.55-3.85 (3H, m), 3.97-4.10 (IH, m), 4.70-4.90 (IH, m), 6.00 (IH, t), 6.85-7.19 (2H, m), 7.30- 7.59 (6H, ). ESMS m/z 449 (MH+, 100%).
Example 99
(S)-N- (3-[4-(4-( 1 -cyano-1 -(3 ,4-difluorophenyl)-methylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2- oxo-oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example -94 by using 3,4- difluorophenylacetonitrile in the place of cyclopropylacetonitrile in 54% yield. Mp: 180-183 °C 1H-NMR (CDC1
3, 200 MHz): δ 2.01(3H, s), 2.57-2.69 (2H, m), 2.90-3.10 (4H, m), 3.19-3.30 (2H, m), 3.65-3.80 (3H, m), 3.90-4.10 (IH, m), 4.69-4.85 (IH, m), 6.10 (IH, m), 6.95 (IH, m), 7.00- 7.30 (4H, m), 7.45 (IH, m). ESMS m/z 485 (M , 100%).
Example 100 (S)-N-{3-[4-(4-(l-cyano-l-(pyridin-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example -94 by using pyridin-2- ylacetonitrile in the place of cyclopropylacetonitrile in 62% yield. Mp. 265-267 °C
1H-NMR (CDCI3, 200 MHz): δ 2.00 (3H, s), 2.79-3.02 (4H, m), 3.10-3.20 (2H,m),3.30-3.40 (2H, m), 3.50-3.85 (3H, m), 3.90-4.10 (IH, m), 4.61-4.90 (IH, ), 6.10-6.30 (IH, m), 6.95-7.10 (2H, m), 7.30 (IH, m), 7.40 (2H, m),7.70-7.90 (IH, m), 8.69 (IH, m). ESMS m z 450 (M^, 100%).
Example 101 (S)-N-{3-[4-(4-(l-cyano-2-(morpholin-l-yl)-ethylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example 1 by using (S)-{3-[4-(4- oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide and diethyl [1- cyano(2-morpholin-l-yl)-ethyl]- in 43% yield. MS (M+l) = 472 (MH+, 100%), M.F. = C24H30FN5O4.
Example 102 (S)-N-{3-[4-(4-(l-cyano-l-(imidazol-l-yl -methylidene)-piperidin-l-yl)-3-fluoroρhenyll-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example 94 by using 1- imidazolylacetonitrile in the place of cyclopropylacetonitrile in 33% yield. M.P. 196-198°C and MS (M+l) = 439 (MH+, 100%), M.F. = C22H23FN6O3.
Example 103
(S)-N-{3-|"4-(4-(l-cyano-l-(2-methyl-imidazol-l-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]- 2-oxo-oxazolidin-5-ylmethyl}-acetamide;
The title compoxmd was prepared as per procedure described in Example 94 by using l-(2-methyl- imidazoyl)acetonitrile in the place of cyclopropylacetonitrile in 27% yield.
M.P. 204-206°C and MS (M+l) = 453 (MH+, 100%), M.F. = C23H25FN6O3.
I l l
Example 104 rS)-N- I3-Γ4-(4-( 1 -cvano- 1 -( 1 ,2.4-triazol- 1 -yl)-methylidene)-piperidin- 1 -yl)-3-fluorophenvH-2- oxo^oxazolidin-5-ylmethyll-acetamide;
The title compound was prepared as per procedure described in Example 94 by using 1-(1 ,2,4- triazolyl)acetonitrile in the place of cyclopropylacetonitrile in 39% yield.
M.P. 194-196°C and MS (M+l) = 440 (MH+, 100%), M.F. = C2ιH22FN7O3.
Example 105 (S)-N-{3- 4-(4-(l-cyano-l-(thiophen-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethv -acetamide;
The title compound was prepared as per procedure described in Example 94 by using (thiophen-2- yl)acetonitrile in the place of cyclopropylacetonitrile in 56% yield. Mp. 260-63 °C
1H-NMR (CDC13, 200 MHz): δ 2.01 (3H, s), 2.80-2.85 (2H, m), 2.90-3.01 (2H, m), 3.05-3.19 (2H, m), 3.20-3.30 (2H, m), 3.59-3.80 (3H, m), 3.95-4.10 (IH, m), 4.70-4.81(lH, m), 6.01 (lH,t), 6.90 (IH, m), 7.00-7.19 (4H, m), 7.35-7.50 (IH, m). ESMS m/z 455 (MH+, 100%).
Example 106 (S)-N-{3- 4-(4-(l,l-dicyano-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example 94 by using malanonitrile in the place of cyclopropylacetonitrile in 79% yield. Mp. 158-160 °C,
1H-NMR (CDC13, 200 MHz): δ 2.01(3H, s), 3.00-3.15 (4H, m), 3.21-3.41(4H, m), 3.50-3.81(3H, m), 3.92-4.10 (IH, m), 4.70-4.90 (IH, m), 5.90-6.05 (IH, m), 7.10 (IH, m),7.20 (IH, m),7.60 (IH, m) ESMS m/z 398 (MH+, 100%).
Example 107 (S)-N-{3-r4-(4'-(l-cyano-l-carboxamido-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title compoxmd was prepared as per procedure described in Example 94 by using carboxamidoacetonitrile in the place of cyclopropylacetonitrile in 68% yield. Mp. 157-159 °C 1H-NMR (CDC13, 200 MHz): δ 2.05 (3H, s), 2.81-3.00 (4H, m), 3.15-3.33 (4H, m),3.52-3.70 (3H, m), 3.70-3.90 (IH, m), 4.75-5.01 (IH, m), 7.01-7.20 (2H, m), 7.50 (IH, m). ESMS m/z 416 (MH+, 100%).
Example 108 (S)-N- { 3 - (4-(4-( 1 -cyano-1 -(N-prop-2-ene-carboxamido)-methylidene)-piperidin- 1 -yl)-3 - fluorophenyl1-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
The title compoimd was prepared as per procedure described in Example 94 by using Nprop-2-ene- aminocarbonylacetonitrile in the place of cyclopropylacetonitrile in 59% yield. Mp: 168-170 °C
1H-NMR (CDCI3, 200 MHz): δ 2.01 (3H, s), 2.81-2.95 (2H, m), 3.15-3.40 (6H, m),3.60-3.81 (3H, m) 3.92-4.10 (3H, m), 4.70-4.85 (IH, m), 5.18-5.38 (2H, m), 5.75-5.95 (IH, m), 6.05 (IH, t), 6.30- 6.40 (IH, m), 6.90 (IH, m), 7.10 (IH, m), 7.50 (IH, m) ESMS m/z 456 (MH+, 100%). Example 109
(S)-N- {3-f4-(4-(l -cyano- 1 -(N-cvclopropyl-carboxamido)-methylidene)-piperidin- 1-yl V3- fluorophenyl] -2-oxo-oxazolidin-5 -ylmethyl } -acetamide:
The title compound was prepared as per procedure described in Example 94 by using N- cyclopropylaminocarbonylacetonitrile in the place of cyclopropylacetonitrile in 44% yield. Mp: 210-212 °C
1H-NMR (CDCI3, 200 MHz): δ 0.58-0.65 (2H,m), 0.80-0.92 (2H, m), 2.05 (3H, s), 2.75-2.95 (3H, m), 3.15-3.25 (4H, m), 3.30-3.41 (2H, m), 3.60-3.81 (3H, m), 3.90-3.41 (IH, m), 4.70-4.85 (IH, m), 5.95-6.10 (IH, m), 6.30-6.40 (IH, m), 6.95 (IH, m), 7.10 (IH, m), 7.45 (IH, m) ESMS m/z 456 (MH+, 100%).
Example 110 (S)-N- { 3 - f4-(4-( 1 -cyano- 1 -(N-c vclohexyl-carboxamido)-methylidene)-piperidin- 1 - yl)-3 - fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide:
The title compound was prepared as per procedure described in Example 94 by using N- cyclohexylaminocarbonylacetonitrile in the place of cyclopropylacetonitrile in 54% yield. Mp. 195-198 °C 1H-NMR (CDCI3, 200 MHz): δ 1.10-155 (m, 6H), 1.58-1.82(4H, m), 1.90-2..05 (IH, m) 2.05 (3H, s), 2.80-2.90 (2H, m), 3.10-3.42 (6H, m), 3.52-3.81 (4H, m), 3.97-4.10 (IH, m),4.70-4.90 (IH, m), 5.99-6.20 (2H, m), 6.95 (IH, m),7.10 (IH, m),7.49 (IH, m)
ESMS m/z 498 (MIT", 100%).
Example 111 (S)-N-{3-[4-(4-(l-cvano-l-(pyrrolidin-l-yl-carbonyl)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidm-5-ylmethyli-acetamide:
The title compound was prepared as per procedure described in Example 94 by using N- pyrrolidinylcarbonylacetonitrile in the place of cyclopropylacetonitrile in 38% yield. Mp: 125-128 °C
1H-NMR (CDC13, 200 MHz): δ 1.90-2.10 (4H, m), 2.00 (3H, s), 2.65-2.79 (2H, m), 2.83-2.90 (2H, m), 3.10-3.30 (4H, m), 3.48-3.60 (4H, m), 3.61-3.80 (3H, m), 3.95-4.10 (IH, m), 4.70-4.85 (IH, m), 6.05 (IH, t), 6.90 (IH, m), 7.10 (IH, m), 7.45 (IH, m) ESMS m/z 470 (MIT", 100%).
Example 112 (S)-N-|3-r4-(4-(l-cvano-l-(morpholin-l-yl-carbonyl)-methylidene)-piperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example 94 by using N- morpholinylcai-bonylacetonifrile in the place of cyclopropylacetonitrile in 62% yield.
Mp.l45-146 °C
!H-NMR (CDC13, 200 MHz): δ 2.05 (3H, s), 2.60-2.70 (2H, m), 2.80-2.90 (2H, m),3.10-3.30 (4H, m), 3.50-3.85 (11H, m), 3.95-4.10 (IH, m), 4.70-4.82 (IH, m), 6.05-6.20 (IH, m), 6.95 (IH, m),7.10 (lH, m),7.45 (lH, m) ESMS m/z 486 (MH+, 100%).
Example 113
(S)-N-{3-f4-(4-(l-cyano-3-hvdroxy-propylidene)-piperidin-l-yl)-3-fluorophenyll-2-oxo- oxazolidin-5-ylmethyl} -acetamide:
The title compound was prepared as per procedure described in Example 1 by using
S)- {3-[4-(4-oxo-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide and diethyl (l-cyano-3-hydiOxypropyl)phosphonate in 45% yield.
M.P. 192-194°C and MS (M+l) = 417 (MH+, 100%), M.F. = C2ιH25FN4O4.
Example 114
(S)-N-|3-r4-(4-(l-cyano-l-ethoxycarbonyl-methylidene)-piperidin-l-yl)-3-fluorophenyl1-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title compound was prepared as per procedure described in Example 94 by using ethoxycarbonylacetonitrile in the place of cyclopropylacetonitrile in 79% yield.
Mp. 165-169 °C
1H-NMR (CDC1 , 200 MHz): δ 1.40 (3H, t), 2.01 (3H, s), 2.90-3.01 (2H, m), 3.18-3.40 (6H, m),
3.50-3.80 (3H, m), 3.95-4.10 (IH, m), 4.21-4.40 (2H, m), 4.70-4.90 (IH, m), 5.95 (IH, t), 6.95
(IH, m),7.10 (IH, m),7.45 (IH, m) ESMS m/z 445 (MH"1", 100%).
Example 115 fS)-N-{3-r4-(4-(l-cyano-l-methylmercapto-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example 94 by using methylmercaptoacetonitrile in the place of cyclopropylacetonitrile in 56% yield.
Mp: 205-207 °C ' tø-NMR (CDC13, 200 MHz): δ 2.01 (3H, s), 2.41 (3H, s), 2.70-2.90 (4H, m), 3.10-3.21 (4H, m),
3.60-3.81 (3H, m), 3.90-4.11 (IH, m), 4.70-4.90 (IH, m), 6.10-6.21 (IH, m), 6.95 (IH, m), 7.10
(IH, m), 7.45 (IH, m).
ESMS m z 419 (MH+, 100%).
Example 116 (S)-N-{3-l4-(4-(l-cyano-l-phenylmercapto-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The title compoxmd was prepared as per procedure described in Example 94 by using phenylmercaptoacetonitrile in the place of cyclopropylacetonitrile in 61% yield. Mp.148-150 °C
ESMS m/z 481(MH+, 100%).
Example 117 (S)-N-{3-[4-(4-(l-cyano-l-bromo-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl} -acetamide;
The mixture of S)-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide (0.12 mmol) and bromine ( 0.72 mmol) in 25 ml chloroform was stirred at reflux for 2 hours.
Chloroform was evaporated under vacuum and the residue was chromatographed on the silica gel to afford the title compound in 39% yield.
M.P. 148-150°C and MS (M+l) = 452 (MH+, 100%), M.F. = C19H20BrFN4O3.
Example 118
(S)-N-{3-r4-(4-(l-cyano-l-(pyridin-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethvU-thioacetamide;
The title compound was prepared as per procedure described in Example 45 by using (S)-N-{3-[4- (4-(l-cyano-l-(pyridin-2-yl)-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 39% yield.
M.P. 110-115°C and MS (M+l) = 466 (MH+, 100%), M.F. = C24H24FN5O2S.
Example 119 (S)-N- {3-[4-(4-(l , 1 -dicvano-methylideneVpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethv -thioacetamide;
The title compound was prepared as per procedure described in Example 45 by using (S)-N-{3-[4- (4-(l , 1 -dicyano-methylidene)-piperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-yhnethyl} - acetamide in 87% yield. Mp. 210-212 °C
1H-NMR (CDC13, 200 MHz): δ 2.60 (3H, m), 2.90-3.05 (4H, m), 3.20-3.40 (4H, m), 3.70-3.90 (IH, m), 4.00-4.20 (IH, m), 4.21-4.40 (IH, m), 4.90-5.10 (IH, m), 6.91-7.19 (2H, m), 7.50 (IH, m), 8.02-8.12 (lH, m). ESMS m/z 414 (MH+, 100%).
Example 120 (S)-N- 13 -f4-(4-(l -cyano- 1 -ethoxycarbonyl-methylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-vhnethyl}-thioacetamide:
The title compound was prepared as per procedure described in Example 45 by using (S)-N-{3-[4-
(4-(l-cyano-l-ethoxycarbonyl-methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 75% yield.
Mp. 182-85 °C
ESMS m/z 432 (MH+, 100%) ■ Example 121
(S)-N- { 3 -r4-(4-( 1 -cyano- 1 -(morpholin- 1 -yl-thiocarbonyl)-methylidene)-piperidin- 1 -yl)-3 - fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl)-thioacetamide;
The lriixture of (S)-N-{3-[4-(4-(l-cyano-l-(morpholin-l-yl-thiocarbonyl)-methylidene)-piperidin- l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide (0.26 mmol), Lowesson's reagent
(0.80 mmol) in dioxane (10 ml) was heated at 100 °C. The reaction mixture was concentrated and the obtained residue was purified by silica gel column chromatography in 76% yield.
Mp.180-182 °C
1H-NMR (CDC13, 200 MHz): δ 2.40-2.58 (2H, m), 2.60 (3H, s), 2.62-2.95 (2H, m), 3.60-3.99 (8H, m), 4.00-4.42 (4H, m), 4.40-4.58 (IH, m), 4.85-5.10 (IH, m), 6.95 (IH, m), 7.10 (IH, m),7.45 (IH, m).
ESMS m/z 518 (MH+, 100%).
Example 122 fS -f3-r4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide;
Preparation of (R)-{3-[4-(4-cyanomethylidenepiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol:
The compound was prepared as per procedure described in Example 1 by using (R)- {3-[4-(4-oxo- piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5 -ylmethyl} -methanesulphonate in 87% yield.
Step-2
Preparation of (R)-{3-[4-(4-cyanomethypiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- alcohol:
The suspension of (R)-{3-[4-(4-cyanomethylidenepiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- yhnethyl} -alcohol (0.11 mmol), 10% palladium on carbon (0.1 g) tetrahydrofuran was stirred under atmospheric hydrogen pressure at 30 C for overnight.
The suspension was filtered and the filtrate was concentrated to the dryness. The residue obtained was chromatographed on the silica gel to provide desired compound in 94% yield.
Step-3 The title compound was prepared as per procedure described in Preparation- 6 followed by
Preparation- 7 by using (R)-{3-[4-(4-cyanomethypiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol in 68% yield.
M.P. 85-86°C and MS (M+l) = 341 (MH+, 100%), M.F. = Cι7H20F2N6O2.
Example 123
(S 1 - (3- 4-(4-cyanomethyl-piperidin- 1 -yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} -4-carboethoxy- 1,2,3-friazole;
The suspension of (S)-l-{3-[4-(4-cyanomethylidene-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5- ylmethyl}-4-carboethoxy-l,2,3-triazole (0.14 mmol), 10% palladium on carbon (0.1 g) tetrahydrofuran was stirred under atmospheric hydrogen pressure at 30°C for overnight.
The suspension was filtered and the filfrate was concentrated to the dryness. The residue obtained was chromatographed on the silica gel to provide desired compound in 42% yield.
M.P. 198-200°C and MS (M+l) = 439 (MH+, 100%), M.F. = C22H26N6O4.
Example 124 (R)-{3-[4-(4-cyanomethyl-ρiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol:
The preparation of the title compound is mentioned in Example- 122. M.P. 110-112°C and MS (M+l) = 316 (MH+, 100%), M.F. = Cι7H21N3O3.
Example 125 (R)-{3-[4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyli- methanesulphonate;
The preparation of the title compound is mentioned in Example- 122.
M.P. 114-116°C and MS (M+l) = 394 (MH+, 100%), M.F. = C18H23N3O5S.
Example 126 (S)-N-{3-[4-(4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-thioacetamide:
The title compound was prepared as per procedure described in Example- 45 by using (S)-N-{3-[4- (4-cyanomethyl-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 65% yield.
MS (M+l) = 373 (MH+, 100%), M.F. = Cι9H24N4O2S.
Example 127 (S)- 1 - {3 - [4-(4-cyanomethyl-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 -ylmethyl } -4- carboxamido- 1 ,2,3-triazole;
The title compoxmd was prepared as per procedure described in Example- 123 by using (S)-l-{3- [4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-4- carboxamido- 1,2,3-triazole in 42% yield. M.P. 248-250°C and MS (M+l) = 428 (MH+, 100%), M.F. = C
20H
2FN
7O
3.
Example 128 (S)-l-{3- [4-(4-cyanomethyl-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5-ylmethyl } -4- cyano-1.2.3-triazole;
The title compound was prepared as per procedure described in Example- 123 by using (S)-l-{3- [4-(4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-4-cyano- 1 ,2,3-triazole in 42% yield.
M.P. 200-202°C and MS (M+l) = 410 (MH+, 100%), M.F. = C^oFN^.
Example 129 (S)-N- { 3 -[4-(4-cvanomethyl-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 -ylmethyl} - thioacetamide;
The title compound was prepared as per procedure described in Example- 45 by using (S)-l-{3-[4- (4-cyanomethylidene-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 87% yield. Mp.195- 197 °C
Η-NMR (CDCI3, 200 MHz): δ 1.50-1.82 (2H, ), 1.85-2.00 (2H, m), 2.40-2.50 (2H, m), 2.59- 2.81 (5H, m), 3.30-3.49 (2H, m), 3.75-3.90 (IH, m), 4.00-4.39 (3H, m), 4.90-5.10 (IH, m), 6.90- 7.10 (2H, m), 7.41 (IH, m), 8.19-8.38 (IH, m) ESMS m/z 391(MH
+, 100%).
Example 130 (S)-N-{3-[4-(4-cvanomethyl-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}- thioacetamide;
The title compoxmd was prepared as per procedure described in Example- 123 by using (S)-l-{3- [4-(4-cyanomethylidene-3-fluoropiperidin-l-yl)-phenyl]-2-oxo-oxazolidm-5-yhuethyl}-acetamide in 56% yield.
MS (M+l) = 331 (MH+ 100%), M.F. = Cι9H23FN4O2S.
Example 131 (R)- { 3 - [4-(4-c vanomethyl-3 -fluoropiperidin- 1 -yl)-phenyl] -2-oxo-oxazolidin-5- ylmethyl } -alcohol;
The title compound was prepared as per procedure described in Example- 123 by using (S)-l-{3- [4-(4-cyanomethylidene-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -alcohol in 89% yield. MS (M+l) = 334 (MH+, 100%), M.F. = Cι7H20FN3O3.
Example 132 (R)- 13 - f4-(4-c vanomethyl-3 -fluoropiperidin- 1 -yl)-phenyl] -2-oxo-oxazolidin-5- ylmethyl } - methanesulphonate:
The title compound was prepared as per procedure described in Preparation-6 by using (R)-{3-[4- (4-cyanomethyl-3-fluoropiperidin- 1 -yι)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} -alcohol in 78% yield.
M.P. 146-148°C and MS (M+l) = 449 (MH+, 100%), M.F. = C22H26F2N4O4.
Example 133 (S)-N-|3-|"4-(4-(l-cvano-l-benzyl)-methylpiperidin-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example- 123 by using (S)-N- {3-
[4-(4-(l-cy-mo-l-benzyl)-methylidenepiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- yhnethyl} -acetamide in 76% yield.
Mp. 210-211 °C
JH-NMR (CDC13, 200 MHz): δ 1.45-1.95 (3H, m), 2.01(3H, s), 2.55-2.80 (3H, m), 2.90-3.00 (2H, m), 3.40-3.59 (2H, m), 3.60-3.80 (3H, m), 3.90-4.10 (IH, m), 4.70-4.85 (IH, m), 5.90 (IH, m),
6.90 (IH, m), 7.05 (IH, m), 7.20-7.39 (6H, m), 7.41 (IH, m).
ESMS m z 465 (Mϊt, 100%).
Example 134 (S)-N- { 3 - [4-(4-( 1 -cvano-2-methanesulphonyloxy -ethylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -acetamide;
Step-1
Preparation of (S)-N-{3-[4-(4-(l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide:
The mixture of sodium borohydride (5.26 mmol), (S)-N-{3-[4-(4-(l-cyano-l-ethoxycarbonyl- methylidene)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide (0.34 mmol) in methanol (15 ml) was stirred for 0.5 hours at room temperature. The reaction mixture was neutralized with dilute hydrochloric acid. The solid was filtered and purified on silica gel column chromatography recrystallized from chloroform:methanol to provide tile compound in 76% yield. Step-2
The title compound was prepared as per procedure described in Preparation-6 by using (S)-N-{3- [4-(4-(l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 89% yield.
M.P. 136-138°C and MS (M+l) = 483 (MH+, 100%), M.F. = C21H27FN4O6S.
Example 135 (S)-N-(3-r4-(4-(l-cvano-l-(3,4-difluorophenyl )-methyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example- 123 by using (S)-N-{3- [4-(4-( 1 -cyano- 1 -(3 ,4-difluorophenyl))-methylidene)-piperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -acetamide in 88% yield. MS (M+l) = 487 (MH+, 100%), M.F. = C
25H
25F
3N
4O
3.
Example 136 (S)-N- ( 3 -f4-(4-( 1 -cyano- 1 -(imidazol- 1 -yl) -methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N-{3-[4- (4-( 1 -cyano- 1 -(imidazol- 1 -yι)-methylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 80% yield.
M.P. 210-212°C and MS (M+l) = 441 (MH+ 100%), M.F. = C22H25FN6O3.
Example 137 (S)-N-(3-r4-(4-(l-cvano-l-(thioplιen-2-yl))-methylpiperidin-l-yl)-3-luorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using .(S)-N-{3-[4- (4-( 1 -cyano- 1 -(thiophen-2-yl))-methylidenepiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 92% yield.
Mp. 91-94 °C
1H-NMR (CDC13, 200 MHz): δ 0.80-1.05 (2H, m), 1.58-1.82 (2H, m), 1.85-2.05 (IH, m), 2.10 (3H, s), 2.50-2.79 (2H, m), 3.39-3.60 (2H, m), 3.60-3.81 (3H, ), 3.90-4.10 (2H, m), 4.65-4.81 (IH, m), 6.10 (IH, t), 6.81-7.15 (4H, m), 7.21-7.45 (2H, m).
ESMS m/z 457 (MH+, 100%).
Example 138
(S)-N-(3-[4-(4-(l-cyano-l-(pyridin-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyll-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N- {3-[4- (4-( 1 -cyano- 1 -(pyridin-2-yl))-methylidenepiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5 - ylmethyl} -acetamide in 78% yield. Mp.160- 162 °C
1H-NMR (CDCI3, 200 MHz): δ 1.61-1.91 (3H, m), 2.01 (3H, s), 2.60-2.80 (2H, m), 3.30-3.50 (4H, m), 3.55-3.80 (3H, m), 3.90-4.10 (2H, m), 4.70-4.85 (IH, m), 6.15 (IH, t), 6.95-7.19 (2H, m), 7.21- 7.39 (lH,m), 7.39-7.49 (2H,m) 7.70-7.85 (lH,m), 8.60 (IH, m). ESMS m z 452 (MH+, 100%).
Example 139 (S)-N-{3-f4-(4-(l-cvano-l-carboxamido)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N-{3-[4- (4-(l-cyano-l-carboxamido)-methylidenepiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 89% yield. Mp. 180-182°C ESMS m/z 418 (MH+, 100%).
Example 140 (S)-N-{3-[4-(4-(l-cyano-l-cyclohexylaminocarbonyl)-methylpiperidin-l-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N-{3-[4- (4-(l-cyano-l-cyclohexylaminocarbonyl)-methylidenepiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide in 90% yield. Mp: 210-213 °C
1H-NMR (CDCI3, 200 MHz): δ 1.10-155 (m, 6H), 1.62-1.83 (5H,m), 1.85-2.00 (2H, m), 2.05 (3H, s), 2.19-2.40 (IH, m), 2.60-2.80 (2H, m), 3.25-3.59 (4H, m), 3.61-3.85 (3H, m), 3.90-4.10 (IH, m), 4.65-4.82 (IH, m), 5.92-6.19 (2H, m), 6.95 (IH, m), 7.10 (IH, m), 7.49 (IH, m) ESMS m/z 500 (MH+, 100%).
Example 141 (S)-N- {3-f 4-(4-( 1 -cyano- 1 -(pyrrolidin- 1 -yl-carbonyl))-methylpiperidin- 1 -yl)-3-fluorophenyl]-2- oxo-oxazolidm-5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N- {3-[4-
(4-( 1 -cyano- 1 -(pyrrolidin- 1 -yl-carbonyl))-methylidenepiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -acetamide in 78% yield.
Mp: 100-102 °C
ESMS m/z 472 (MH+, 100%). Example 142
(S)-N- {3-f4-(4-(l -cyano- 1 -(morpholin- 1 -yl-carbonyl))-methylpiperidin-l -yl)-3 -fluorophenyl] -2- oxo-oxazolidin-5 -ylmethyl} -acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N-{3-[4- (4-( 1 -cyano- 1 -(morpholin- 1 -yl-carbonyl))-methylidenepiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl} -acetamide in 79% yield.
Mp.240-241°C
1H-NMR (CDC13, 200 MHz): δ 2.00 (3H, s), 2.10-2.35 (2H, m), 2.55-2.81 (2H, m), 3.25-3.85
(15H, m), 3.95-4.10 (IH, m), 4.62-4.83 (IH, m), 6.20-6.40 (IH, m), 6.90 (IH, m), 7.05 (IH, m), 7.41 (IH, m)
ESMS m/z 488 (MH+, 100%).
Example 143 f S)-N- (3-[4-(4-(l -cyano- 1 -ethoxycarbonvD-methylpiperidin- 1 -yl)-3-fluorophenyl] -2-oxo- oxazolidin-5-ylmethyll-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N-{3-[4-
(4-(l-cyano-l-ethoxycarbonyl)-methylienepiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 70% yield.
Mp. 98-100 °C
1H-NMR (CDC13, 200 MHz): δ 1.30 (3H, t), 1.75-1.95 (2H, m), 2.10 (3H, s), 2.15-2.20 (IH, m),
2.60-2.81 (2H, ), 3.35-3.59 (4H, m), 3.60-3.80 (3H, m), 3.90-4.10 (IH, m), 4.30 (2H, q), 4.70-
4.85 (IH, m), 6.10 (IH, t), 6.90 (IH, m), 7.05 (IH, m), 7.41 (IH, m)
ESMS m/z 447 (MH+, 100%).
Example 144 (S)-N- ( 3 -f 4-(4-( 1 -cyano- 1 -(phenylmercapto))-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-acetamide;
The title product was prepared as per procedure described in Example 123 by using (S)-N- {3-[4-
(4-(l-cyano-l-(phenylmercapto))-methylidenepiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 78% yield.
Mp.148- 150 °C
Η-NMR (CDCI3, 200 MHz): δ 1.61-1.90 (2H, m), 2.00 (3H, s), 2.05-2.25 (2H, m), 2.60-2.79 (2H, m), 3.40-3.59 (2H, m), 3.60-3.81(3H, m), 3.91-4.10 (IH, m), 4.70-4.90 (IH, m), 6.25 (IH, t), 6.95
(IH, dd),7.10 (IH, m), 7.35-7.45 (4H, m), 7.59-7.65 (2H, m).
ESMS m/z 483 (MH+, 100%).
Example 145 (S)-N-{3-r4-(4-(l-cyano-l-(
"ρyridin-2-yl))-methylpiperidm-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyll-thioacetamide;
The title product was prepared as per procedure described in Example 45 by using (S)-N-{3-[4-(4-
(l-cyano-l-(pyridin-2-yl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 77% yield. Mp.102- 103 °C
1H-NMR (CDC13, 200 MHz): δ 1.60-1.95 (3H, m), 2.01-2.30 (2H, m), 2.40 (3H, s), 3.20-3.90 (6H, m), 3.95-4.39 (3H, m), 4.81-5.10 (IH, ), 5.20-5.40 (IH, m), 6.70-7.10 (2H, m), 7.20-7.49 (2H, m), 7.65-8.10 (2H, m), 8.20-8.40 (IH, m), 8.60-8.80(lH, m).
ESMS m z 468 (MH+, 100%). Example 146
(S)-N- |3-[4-(4-(l -cyano- 1 -(morpholin-1 -yl-carbonyl))-methylpiperidin- 1 -yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl)-thioacetamide;
The title product was prepared as per procedure described in Example 45 by using (S)-N-{3-[4-(4- (l-cyano-l-(morpholin-l-yl-carbonyl))-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide in 75% yield. Mp. 220-222 °C ESMS m/z 504 (MH+, 100%).
Example 147
(S)-N- {3-f4-(4-( 1 -cyano- 1 -ethoxycarbonyl)-methylpiperidin- 1 -yl)-3 -fluorophenyl]-2-oxo- oxazolidin-5-ylmethyll-thioacetamide;
The title product was prepared as per procedure described in Example 45 by using (S)-N-{3-[4-(4-
(l-cyano-l-ethoxycarbonyl)-methylpiperidin-l-yl)-3-fluoroρhenyl]-2-oxo-oxazolidin-5-ylmethyl}- thioacetamide in 82% yield.
Mp. 93-95 °C
ESMS m/z 463 (MH+, 100%).
Example 148 (S)-N-{3- 4-(4-(l-cvano-l-carboxamido)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin- 5-ylmethyli -thioacetamide;
The title product was prepared as per procedure described in Example 45 by using (S)-N- {3-[4-(4- (1 -cyano- 1 -carboxamido)-methylpiperidin-l -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} - acetamide in 68% yield.
MS (M+l) = 434 (MH+, 100%), M.F. = Czo^FNsOsS.
Example 149
(S)-N-{3-[4-(4-(l-cyano-l-thiocarboxamido)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-thioacetamide;
The title product was prepared as per procedure described in Example 121 by using (S)-N-{3-[4- (4-(l-cyano-l-carboxamido)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 38% yield. Mp. 178-180°C ESMS m/z 449(MH+, 100%).
Example 150
(S)- 1 - (3 -[
~4-(4-( 1 -cvano-2-hydroxy)-ethylpiperidin- 1 -yl)-3 -fluorophenyl]-2-oxo-oxazolidin-5- ylmethyll -4-methoxycarbonyl- 1 ,2,3-triazole;
S ej l
Preparation of (S)-N-{3-[4-(4-(l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -azide: The compound was prepared by using the procedure described in Example- 134 from (S)-N- {3 -[4-
(4-(l-cyano- 1 -ethoxycarbonyl-methylidene)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol.
This compound was converted to the desired compound by using procedures of Preparation-6 followed by Preparation-7 in overall 64% yield. Step-2
The title product was prepared as per procedure described in Example 4 by using (S)-N- {3-[4-(4-
( 1 -cyano-2-hydroxy-ethyl)-piperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl} -azide and methyl propiolate in the place of ethyl propiolate in 68% yield.
M.P. 204-206°C and MS (M+l) = 474 (MH+, 100%), M.F. = C22H25FN6O5.
Example 151 (S)~ 1 - |3-|~4-(4-(l -cyano-2-hydroxy)-ethylpiperidin- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl}-4-ethoxycarbonyl-l,2,3-triazole;
The isomeric mixture was prepared as per procedure described in Example 4 by using (S)-N-{3-[4- (4-(l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-azide in 59% yield as a isomeric mixture. The title compound isomer was separated on preparative HPLC in 44% yield. M.P. 170-172°C and MS (M+l) = 487 (MH+, 100%), M.F. = C23H27FN6O5.
Example 152 (S)- 1 - { 3 - [4-(4-( 1 -c vano-2-hydroxy)-ethylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -5-ethoxycarbonyl-l ,2,3-triazole;
The other isomer obtained in Example- 151 was separated by using preparative HPLC in 12% yield.
M.P. 76-78°C and MS (M+l) = 487 (MH+ 100%), M.F. = C23H27FN6O5.
Example 153 (R)-3-{3-[4-(4-(l-cyano-2-hydroxy)-ethylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyloxy} -iso-oxazole;
The title product was prepared as per procedure described in Example 83 by using (S)-N-{3-[4-(4- (l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol in 67% yield.
M.P. 146-148°C and MS (M+l) = 431 (MH+, 100%), M.F. = C2ιH23FN4O5.
Example 154 (R)-{3- 4-f4-(l-cyano-2-hydroxy)-ethylpiperidin-l-yl)-3-fluorophenyll-2-oxo-oxazolidin-5- ylmethyl I -methanesulphonate;
The title compound was prepared by using the procedure described in Preparation-6 from (S)-N- {3-.[4-.(4-(l-cyano-2-hydroxy-ethyl)-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol in overall 74% yield.
M.P. 144-146°C and MS (M+l) = 442 (MH+, 100%), M.F. = Cι9H24FN3O6S.
Example 155 (R)- ( 3 -|"4-(4-(l -cyano- 1 -hydroxycarbonylVmethylpiperidin- 1 -yl)-3-fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl}-methanesulphonate;
The title compound was prepared by using the procedure described in Preparation-6 from (S)-N- {3-[4-(4-(l-cyano-l-hydroxycarbonyl)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol in overall 80% yield. M.P. 108-110°C and MS (M+l) = 456 (MH+, 100%), M.F. = Ci9H22FN3O7S.
Example 156 (R)- 13 -|"4-(4-( 1 -cyano- 1 -ethoxycarbonvD-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin- 5-ylmethyl} -methanesulphonate;
The title compound was prepared by using the procedure described in Preparation-6 from (S)-N- {3-[4-(4-(l -cyano- 1 -ethoxycarboιιyl)-methylpiperidm- 1 -yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol in overall 85% yield. M.P. 102-104°C and MS (M+l) = 484 (MH+ 100%), M.F. = C2ιH26FN3O7S.
Example 157 (R - {3 - f4-(4-( 1 -cyano- 1 -( 1 ,3 -thiazol-2-yl))-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo- oxazolidin-5-ylmethyl)-methanesulphonate;
Step-1
Preparation of (S)-N-{3-[4-(4-(l-cyano-l-(l,3-thiazol-2-yl))-methylidenepiperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol: The desired compound was prepared by following the procedure described in Example 94 and using (l,3-thiathiazol-2-yl)-acetonitrile in the place of cyclopropylacetonitrile in 66% yield.
Step-2
Preparation of (S)-N-{3-[4-(4-(l-cyano-l-(l,3-thiazol-2-yl))-methylpiperidin-l-yl)-3- fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}-alcohol:
The desired compound was prepared by following the procedure described in Example 122 and using (S)-N- { 3 - [4-(4-( 1 -cyano- 1 -( 1 ,3 -thiazol-2-yl))-methylidenepiperidin- 1 -yl)-3 -fluorophenyl] -2- oxo-oxazolidin-5-ylmethyl} -alcohol in 63%> yield.
Step-3
The title compound was prepared by using the procedure described in Preparation-6 from (S)-N- {3-[4-(4-(l -cyano-1 -(1 , 3-thiazol-2-yl))-memylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -alcohol in 66% yield.
M.P. 64-66°C and MS (M+l) = 495 (MH+, 100%), M.F. = C2ιH23FN4O5S2.
Example 158 (R)-{3- 4-(4-(l-cyano-l-carboxamido)-methylpiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl) -methanesulphonate;
The title compound was prepared by using the procedure described in Preparation-6 from (S)-N- {3-[4-(4-(l -cyano- 1 -carboxamide)-methylpiperidin- 1 -yl)-3 -fluorophenyl] -2-oxo-oxazolidin-5- ylmethyl} -alcohol in 80% yield.
M.P.220-222°C and MS (M+l) = 455 (MH+, 100%), M.F. = Cι
9H
23FN
4O
6S.
Example 159 rS)-N-{3-r4-(4-cyanomethyl-3,4-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -acetamide;
A mixture of (S)-N-{3-[4-(4-oxo-piperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide (28.64 mmol), pyridine (6.32 mmol), cyanoacetic acid (30.00 mmol) and ammonium acetate (6.40 mmol) in toluene was refluxed with azeotropic removal of water for 5 to 6 hours. The reaction mixture was cooled at room temperature, diluted with ethyl acetate and washed with water. The organic layer was dried and concentrated in vacuo to afford crude product. The crude product was recrystallized from ethyl acetate to give the title compound in 67% yield. Mp. 132 °C and MS (M+l) = 373.
Example 160
(S)-N-(3-r4-(4-cyanomethyl-3,4-dehvdroρiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -thioacetamide;
The title compound was prepared as per procedure described in Example- 45 and by using (S)-N- {3-[4-(4-cyanomethyl-3,4-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5-ylmethyl}- acetamide in 87% yield as a solid. Mp.l 10-112°C and MS (M+l) = = 389.
Example 161 (S)~N-{3-|'4-f4-cvanomethyl-3-methyl-4.5-dehydropiρeridin-l-yl)-3-fluorophenyll-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example- 159 and by using (S)-{3- [4-(3-methyl-4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-acetamide in 65% yield.
M.P. 149-150°C and MS (M+l) = 387 (MH+, 100%), M.F. = C20H23FN4O3.
Example 162 (S)-N-{3-[4-(4-cvanomethyl-3-fluoro-4,5-dehydiOpiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl} -acetamide;
The title compound was prepared as per procedure described in Example- 159 and by using (S)-{3- [4-(3-fluoro-4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl} -acetamide in 71% yield. M.P. 128-130°C and MS (M+l) = 391 (MH+, 100%), M.F. = Cι9H20F2N4O3.
Example 163 (S)-N-{3-[4-(4-cyanomethyl-3-fluoro-4,5-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo- oxazolidin-5-ylmethyl}-isobutylcarbamate;
Step-1
Preparation of (S)-N-{3-[4-(4-cyanomethyl-3-fluoro-4,5-dehydropiperidin-l-yl)-3-fluorophenyl]-2- oxo-oxazolidin-5-ylmethyl}-amine:
The title compoxmd was prepared as per procedure described in Example- 159 and by using (S)-{3- [4-{3-fluoro-4-oxo-piperidin-l-yl)-phenyl]-2-oxo-oxazolidin-5-ylmethyl}-amine in 48% yield.
Step-2
The title compoxmd was prepared by following the procedure of Example —10 and by using (S)-N- {3-[4-(4-cyanomethyl-3-fluoro-4,5-dehydropiperidin-l-yl)-3-fluorophenyl]-2-oxo-oxazolidin-5- ylmethyl} -amine and isobutylchloroformate in 77% yield.
M.P. 146-148°C and MS (M+l) = 449 (MH+, 100%), M.F. = C22H26F2N4O4.