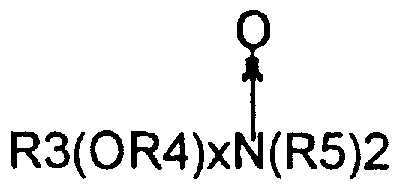WO1998013481A1 - An enzyme with amylase activity - Google Patents
An enzyme with amylase activity Download PDFInfo
- Publication number
- WO1998013481A1 WO1998013481A1 PCT/DK1997/000410 DK9700410W WO9813481A1 WO 1998013481 A1 WO1998013481 A1 WO 1998013481A1 DK 9700410 W DK9700410 W DK 9700410W WO 9813481 A1 WO9813481 A1 WO 9813481A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amylase
- enzyme
- alkyl
- thermoalcalibacter
- strain
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
- C12N9/24—Hydrolases (3) acting on glycosyl compounds (3.2)
- C12N9/2402—Hydrolases (3) acting on glycosyl compounds (3.2) hydrolysing O- and S- glycosyl compounds (3.2.1)
- C12N9/2405—Glucanases
- C12N9/2408—Glucanases acting on alpha -1,4-glucosidic bonds
- C12N9/2411—Amylases
- C12N9/2414—Alpha-amylase (3.2.1.1.)
- C12N9/2417—Alpha-amylase (3.2.1.1.) from microbiological source
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38609—Protease or amylase in solid compositions only
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/38—Products with no well-defined composition, e.g. natural products
- C11D3/386—Preparations containing enzymes, e.g. protease or amylase
- C11D3/38618—Protease or amylase in liquid compositions only
Definitions
- TITLE An enzyme with amylase activity
- the present invention relates to a novel alkali stable amylase, an enzyme composition comprising said amylase, and the use of said enzyme and enzyme composition for a number of industrial applications.
- ⁇ -Amylase enzymes have been used industrially for a number of years and for a variety of different purposes, the most important of which are starch liquefaction, textile desizing, starch modification in the paper and pulp industry, and for brewing and baking. A further use of ⁇ -amylases which is becoming increasingly important is the removal of starchy stains during washing or dishwashing.
- Ca 2+ ions may disturb in industrial applications, Ca 2+ ions are undesired in e.g. laundry and dishwashing detergents, it is the object of the present invention to provide an alkali stable amylase which is stable within a broad range of the metal ion (e.g. Ca 2+ ) concentrations.
- the metal ion e.g. Ca 2+
- the present inventors have now surprisingly identified an ⁇ -amylase from a moderately thermo alkaliphile anaerobe strain Thermoalcalibacter bogoriae , which belong to the Clostridium/ Bacillus subphyllum.
- the ⁇ -amylase of the invention has been thoroughly characterized and shown that the activity of the enzyme is rela- tively independent of the specific metal ion (e.g. Ca 2+ ) con- centrati •on, i.e. i ⁇ t i•s not acti.vated by e.g. Ca2+.
- specific metal ion e.g. Ca 2+
- con- centrati •on i.e. i ⁇ t i•s not acti.vated by e.g. Ca2+.
- the invention relates to an isolated ⁇ -amylase characterized by having at least 65 % of residual ⁇ -amylase activity, after 30 in incubation at 65°C, pH 8.0 in a substrate solution containing (wt/vol) soluble starch (0.5%), where the substrate solution is having a Ca 2+ concentration between 0.5 mM and 6 iriM Ca 2+ .
- the relatively high independence of the specific metal ion (e.g. Ca 2+ ) concentration of the ⁇ -amylase of the present in- vention is highly advantageous for a number of industrial applications as e.g. Ca 2t i•ons may di•sturb in a number of industrial applications such as in laundry or dishwash detergents.
- specific metal ion e.g. Ca 2+
- concentration of the ⁇ -amylase of the present in- vention is highly advantageous for a number of industrial applications as e.g. Ca 2t i•ons may di•sturb in a number of industrial applications such as in laundry or dishwash detergents.
- ⁇ -amylase of the invention is presently believed to be the first description and characterization of extracellular amylolytic enzymes from an anaerobe thermo alkaliphile.
- the present invention relates to an isolated extracellular ⁇ -amylase obtained from a strain of Thermoalcalibacter sp.
- the invention relates to a method of producing an ⁇ -amylase of the invention, the method comprising culturing a strain of Thermoalcalibacter sp . under conditions permitting the production of the enzyme, and recovering the enzyme from the culture.
- the invention relates to an enzyme or an enzyme composition and the use of such an enzyme or enzyme composition for various industrial applications.
- Figure 1 SDS-PAGE of various purification steps.
- Figure l shows electrophoretic separation of the proteins of Thermoalcalibacter bogoriae grown on starch. Concentrated supernatant after PD-10 (lane 1+2), ⁇ -amylase (lane 3,4,5), purified ⁇ -amylase (lane 5) , CGTase (lane 6+7) , silver staining (lane 1,3,5,6), activity staining (lane 2,4,7).
- Figure 2 shows the pH optimum of the prepurified ⁇ -amylase from Thermoalcalibacter bogoriae .
- pH optimum universal buffer (Britton & Robinson) pH 4.0-11.0 containing 0.5% (wt/vol) soluble starch was used at 65°C and 30 minutes incubation time. 100% of residual acitivty correspond to 0.28 U/ml.
- Figure 3 shows the temperature optimum of ⁇ -amylase from Thermoalcalibacter bogoriae . Incubation was done for 30 minutes in 100 itiM sodium phosphate buffer pH 9.0 containing 0.5% (wt/vol) soluble starch. 100% of residual activity correspond to 0.30 U/ml.
- Figure 4 shows the influence of metal ions and chemical reagents on amylase activity.
- a cloned DNA sequence refers to a DNA sequence cloned by a standard cloning procedure conventionally used in genetic engineering to relocate a segment of DNA from its natural location to a different site where it will be reproduced. The cloning process involves excision and isolation of the desired DNA segment, insertion of the piece of DNA into the vector molecule and incorporation of the recombinant vector into a cell where multiple copies or clones of the DNA segment will be replicated.
- the term “cloned DNA sequence” of the invention may alternatively be termed "DNA construct” or "isolated DNA sequence".
- the term “obtained from” as used herein in connection with a specific microbial source means that the enzyme is produced by the specific source, or by a cell in which a gene from the source have been inserted.
- an isolated polypeptide or "isolated ⁇ -amylase as used about the ⁇ -amylase of the invention is a ⁇ -amylase or a ⁇ -amylase part which is essentially free of other non ⁇ -amylase polypeptides, e.g., at least about 20% pure, preferably at least about 40% pure, more preferably about 60% pure, even more preferably about 80% pure, most preferably about 90% pure, and even most preferably about 95% pure, as determined by SDS-PAGE.
- isolated polypeptide may alternatively be termed “purified polypeptide” .
- homologous impurities means any impurity (e.g. another polypeptide than the enzyme of the invention) which originate from the homologous cell where the enzyme of the invention is originally obtained from.
- the homologous cell may e.g. be a strain of Thermoalcalibacter bogoriae.
- ⁇ -amylase In the present context ⁇ -amylase is defined according to the IUB enzyme nomenclature as EC 3.2.1.1. Alternative Name(s) : 1, 4-ALPHA-D-GLUCAN GLUCANOHYDROLASE, GLYCOGENASE. Reaction catalysed:
- amylolytic In the present context, the term “amylolytic” or “amylolytic activity” is intended to indicate that the enzyme in question has a starch-degrading capability.
- Specific examples of enzymes having amylolytic activity i.e. amylolytic enzymes, includes a-amylases, pullulanases, neo-pullulanases, iso- a ylases, beta-amylases, CTGases, maltogenases as well as G-4 and G-6 amylases.
- Moderate thermo alkaliphile relates to a cell which is capable of surviving at relatively high temperatures, i.e. at a temperature above 55°C such as above 60°C or 65°C, and at relatively high pH levels, above 8.5 such as above 9 or 10.
- extracellular refers to an enzyme which is exported out of the cell producing the enzyme, i.e. it is secreted by or diffused out of the cell. Such an enzyme will generally comprise a signal-peptide to guide the secretion (i.e. exporting out of the cell) of the enzyme.
- Amylase obtained from Thermoalcalibacter bogoriae The effect of metal ions on the activity of the amylase of the present invention was examined by pre-incubating the enzyme together with metal ions for 30 in. at 65°C, pH 8.0 in a substrate solution containing (wt/vol) soluble starch (0.5%).
- the amylase of the present invention is preferably having at least 65% of residual amylase activity with a Ca concentration between 0.5 mM to 6 mM Ca , more preferably having at least 65% of residual activity with a Ca 2+ concentration between 1.0 mM to 5 mM Ca , and even more preferably having at least 65% of residual amylase activity with a Ca concentration between 2 mM to 4 mM Ca + .
- the ⁇ -amylase of the invention is preferably one which has a molecular mass of 57 ⁇ 10 kDA, more preferably a molecular mass of 57 + 5 kDA, even more preferably a molecular mass of 57 ⁇ 3 kDA, and most preferably a molecular mass of 57 ⁇ 2 kDA.
- the molecular mass is measured by SDS-PAGE electrophoresis as further described in the "Materials and Methods" section (vide infra) .
- the ⁇ -amylase of the invention is preferably one which has a temperature optimum of 65 ⁇ 10 °C, more preferably a temperature optimum of 65 ⁇ 5 °C, and even more preferably a temperature optimum of 65 ⁇ 2 °C.
- the temperature optimum is measured by incubating the enzyme with a 0.5% (wt/vol) substrate solution (soluble starch; Merck) in 100 mM sodium phosphate buffer at pH 9.0. Incubation was done for 30 min. at temperatures between 30-80°C.
- substrate solution soluble starch; Merck
- the ⁇ -amylase of the invention is preferably one which has a pH optimum in the range between pH 8 to pH 10, more preferably in the range between pH 8.5 to pH 9.5.
- the pH optimum is measured by incubating the enzyme with a 0.5% (wt/vol) substrate solution (soluble starch; Merck) in a 120 mM universal Britton Robinsson buffer.
- a 0.5% (wt/vol) substrate solution soluble starch; Merck
- a 120 mM universal Britton Robinsson buffer for further details see working example herein (vide infra) .
- an DNA sequence encoding an amylase of the present invention can be cloned from a strain of Thermoalcalibacter bogoriae .
- the DNA sequence may be cloned by purifying the enzyme (e.g. as described in a working example herein (vide infra) ) , a ino acid sequencing, and preparing a suitable probe or PCR primer based on this amino acid sequence.
- the DNA sequence the invention can also be cloned by any general method involving cloning, in suitable vectors, a DNA library from any organism expected to produce the amylase of interest, transforming suitable host cells with said vectors, - culturing the host cells under suitable conditions to express any enzyme of interest encoded by a clone in the DNA library, screening for positive clones by determining any amylase activity of the enzyme produced by such clones, and
- the CGTase of the invention may be obtained from bacteria corresponding to the Thermoalcalibacter line within the Clostridium/ 'Bacillus subphyllu in particular a strain of Thermoalcalibacter bogoriae as described below:
- Cells are rod-shaped, 0.3-0.5 mm thick and 3-5 mm long. Colonies are 3-5 mm in diameter, pale-whitish, lense-shaped. Obligately anaerobic. Temperature range for growth from 30°C to 65 C, optimum around 50 C to 55 °C. Range of growth from pH 6 to 10.5, optimum at pH 9.5; growth from 0 to 4 % NaCl with an optimum around 1% NaCl, represents an optimum Na + concentration of 230 mM. Grows heterotrophically with peptone. Growth in presence of sulfate, thiosulfate or sulfur. Thiosulfate enhances growth on a fermentable substrate such as glucose and starch, resulting in the formation of H 2 S.
- Fermentation products on starch with thiosulfate are acetate and ethanol.
- Cell wall type is Gram-positive, but cell wall is atypically thin. Sheat like structures at the cell separation area. Branched cells were regularly present. Parts of an outer surface layer were observed.
- 16S rRNA analysis shows that Thermoalcalibacter bogoriae represents a new line within the Clostridium/Bacillus subphyllum.
- the 16 rRNA sequencing analysis was done at Deutche Sammlung von Mikroorganismen und Zellkulturen (DSMZ) .
- the amylase of the present invention may be produced by cultivation of a homologous strain e.g. the above mentioned deposited strain in a suitable medium resulting in conditions permitting the production of the enzyme.
- the medium used to culture the strain may be any conventional medium suitable for growing the cells in question.
- the secreted, into the culture medium, amylase may be recovered therefrom by well-known procedures including separating the cells from the medium by centrifugation or filtration, precipitating proteinaceous components of the medium by means of a salt such as ammonium sulphate, followed by chro- matographic procedures such as ion exchange chromatography, affinity chromatography, or the like.
- the present invention relates to an enzyme composition, which comprises an amylase as described above.
- the enzyme composition of the invention may contain one or more other enzymes, for instance proteases, lipases, amylolytic enzymes, oxidases (including peroxidases) , or cellulases (mentioned examples are examples preferably for use in detergents) , e.g. Savinase®, Durazyme®, Esperase®, Alcala- se®, Termamyl® or CelluzymeTM (all available from Novo Nordisk A/S)").
- enzymes for instance proteases, lipases, amylolytic enzymes, oxidases (including peroxidases) , or cellulases (mentioned examples are examples preferably for use in detergents) , e.g. Savinase®, Durazyme®, Esperase®, Alcala- se®, Termamyl® or CelluzymeTM (all available from Novo Nordisk A/S)").
- the enzyme composition may be prepared in accordance with methods known in the art and may be in the form of a liquid or a dry composition.
- the enzyme composition may be in the form of a granulate or a microgranu- late (US 4106991, US 5324649) .
- the enzyme to be included in the composition may be stabilized in accordance with methods known in the art. Examples are given below of preferred uses of the enzyme composition of the invention.
- the dosage of the enzyme composition of the invention and other conditions under which the composition is used may be determined on the basis of methods known in the art.
- the enzyme and/or the enzyme composition according to the invention may be useful for at least one of the following purposes .
- amylase enzyme of the invention may be used in conventional applications of amylase enzyme, particularly at conditions where only low concentration of Ca 2+ are present, e.g. in laundry and dishwash detergents, in institutional and industrial cleaning.
- amylase enzymes of the invention can also be used for starch liquefaction, textile desizing, starch modification in the paper and pulp industry, and for brewing and baking.
- the detergent compositions according to the present invention comprise a surfactant system, wherein the surfactant can be selected from nonionic and/or anionic and/or cationic and/or ampholytic and/or zwitterionic and/or semi-polar surfactants.
- the surfactant is typically present at a level from
- the surfactant is preferably formulated to be compatible with enzyme components present in the composition.
- the surfactant is most preferably formulated in such a way that it promotes, or at least does not degrade, the stability of any enzyme in these compositions.
- Preferred systems to be used according to the present invention comprise as a surfactant one or more of the nonionic and/or anionic surfactants described herein.
- Polyethylene, polypropylene, and polybutylene oxide condensates of alkyl phenols are suitable for use as the nonionic surfactant of the surfactant systems of the present invention, with the polyethylene oxide condensates being preferred.
- These compounds include the condensation products of alkyl phenols having an alkyl group containing from about 6 to about 14 carbon atoms, preferably from about 8 to about 14 carbon atoms, in either a straight chain or branched-chain configuration with the alkylene oxide.
- the ethylene oxide is present in an amount equal to from about 2 to about 25 moles, more preferably from about 3 to about 15 moles, of ethylene oxide per mole of alkyl phenol.
- nonionic surfactants of this type include IgepalTM CO-630, marketed by the GAF Corporation; and TritonTM X-45, X- 114, X-100 and X-102 , all marketed by the Rohm & Haas Company. These surfactants are commonly referred to as alkylphenol alkoxylates (e.g., alkyl phenol ethoxylates) .
- the condensation products of primary and secondary aliphatic alcohols with about 1 to about 25 moles of ethylene oxide are suitable for use as the nonionic surfactant of the nonionic surfactant systems of the present invention.
- the alkyl chain of the aliphatic alcohol can either be straight or branched, primary or secondary, and generally contains from about 8 to about 22 carbon atoms.
- About 2 to about 7 moles of ethylene oxide and most preferably from 2 to 5 moles of ethylene oxide per mole of alcohol are present in said condensation products.
- nonionic surfactants of this type include TergitolTM 15-S-9 (The condensation product of Cn-C 15 linear alcohol with 9 moles ethylene oxide) , TergitolTM 24-L-6 NMW (the condensation product of C 12 -C 14 primary alcohol with 6 moles ethylene oxide with a narrow molecular weight distribution) , both marketed by Union Carbide Corporation; NeodolTM 45-9 (the condensation product of C-L4-C-L 5 linear alcohol with 9 moles of ethylene oxide) , NeodolTM 23-3 (the condensation product of C 12 -C 13 linear alcohol with 3.0 moles of ethylene oxide), NeodolTM 45-7 (the condensation product of C 14 -C 15 linear alcohol with 7 moles of ethylene oxide) , NeodolTM 45-5 (the condensation product of C 14 -C 15 linear alcohol with 5 moles of ethylene oxide) marketed by Shell Chemical Company, Kyro EOB (the con- densation product of C- L3 -C 15 alcohol with 9 mo
- alkylpolysac- charides disclosed in US 4,565,647, having a hydrophobic group containing from about 6 to about 30 carbon atoms, preferably from about 10 to about 16 carbon atoms and a polysaccharide, e.g. a polyglycoside, hydrophilic group containing from about 1.3 to about 10, preferably from about 1.3 to about 3, most preferably from about 1.3 to about 2.7 saccharide units.
- Any reducing saccharide containing 5 or 6 carbon atoms can be used, e.g.
- glucose, galactose and galactosyl moieties can be substituted for the glucosyl moieties (optionally the hydrophobic group is attached at the 2-, 3-, 4-, etc. positions thus giving a glucose or galactose as opposed to a glucoside or galacto- side) .
- the intersaccharide bonds can be, e.g., between the one position of the additional saccharide units and the 2-, 3-, 4-, and/or 6- positions on the preceding saccharide units.
- the preferred alkylpolyglycosides have the formula
- R 2 is selected from the group consisting of alkyl, alkylphenyl, hydroxyalkyl, hydroxyalkylphenyl, and mixtures thereof in which the alkyl groups contain from about 10 to about 18, preferably from about 12 to about 14, carbon atoms; n is 2 or 3, preferably 2; t is from 0 to about 10, pre-ferably 0; and x is from about 1.3 to about 10, preferably from about 1.3 to about 3 , most preferably from about 1.3 to about 2.7.
- the glycosyl is preferably derived from glucose.
- the alcohol or alkylpolyethoxy alcohol is formed first and then reacted with glucose, or a source of glucose, to form the glucoside (attachment at the l-position) .
- the additional glycosyl units can then be attached between their l-position and the preceding glycosyl units 2-, 3-, 4-, and/or 6-position, preferably predominantly the 2-position.
- the condensation products of ethylene oxide with a hy- drophobic base formed by the condensation of propylene oxide with propylene glycol are also suitable for use as the additional nonionic surfactant systems of the present invention.
- the hydrophobic portion of these compounds will preferably have a molecular weight from about 1500 to about 1800 and will exhibit water insolubility.
- the addition of polyoxyethylene moieties to this hydrophobic portion tends to increase the water solubility of the molecule as a whole, and the liquid character of the product is retained up to the point where the polyoxyethylene content is about 50% of the total weight of the condensation product, which corresponds to condensation with up to about 40 moles of ethylene oxide.
- Examples of compounds of this type include certain of the commercially available PluronicTM surfactants, marketed by BASF.
- nonionic surfactant of the nonionic surfactant system of the present invention are the condensation products of ethylene oxide with the product resulting from the reaction of propylene oxide and ethylenediamine.
- the hydrophobic moiety of these products consists of the reaction product of ethylenediamine and excess propylene oxide, and generally has a molecular weight of from about 2500 to about 3000. This hydrophobic moiety is condensed with ethylene oxide to the extent that the condensation product contains from about 40% to about 80% by weight of polyoxyethylene and has a molecular weight of from about 5,000 to about 11.000.
- this type of nonionic surfactant include certain of the commercially available TetronicTM compounds, marketed by BASF.
- nonionic surfactant of the surfactant systems of the present invention are polyethylene oxide condensates of alkyl phenols, condensation products of primary and secondary aliphatic alcohols with from about 1 to about 25 moles of ethyleneoxide, alkylpolysaccharides, and mixtures hereof. Most preferred are C 8 -C 14 alkyl phenol ethoxylates having from 3 to 15 ethoxy groups and C-J-C-LS alcohol ethoxylates (preferably Cio avg.) having from 2 to 10 ethoxy groups, and mixtures thereof.
- Highly preferred nonionic surfactants are polyhydroxy fatty acid amide surfactants of the formula
- R 1 is methyl
- R 2 is straight Cn- ⁇ 5 alkyl or Cig-ig alkyl or alkenyl chain such as coconut alkyl or mixtures thereof
- Z is derived from a reducing sugar such as glucose, fructose, maltose or lactose, in a reductive amination reaction.
- Highly preferred anionic surfactants include alkyl alkoxylated sulfate surfactants.
- Examples hereof are water soluble salts or acids of the formula R0(A) m S03M wherein R is an unsubstituted C 10 -C- 24 alkyl or hydroxyalkyl group having a c io ⁇ c 24 alkyl component, preferably a C 12 ⁇ c 2 ⁇ alkyl or hydroxyalkyl, more preferably C 12 -C 18 alkyl or hydroxyalkyl, A is an ethoxy or propoxy unit, m is greater than zero, typically between about 0.5 and about 6 , more preferably between about 0.5 and about 3, and M is H or a cation which can be, for example, a metal cation (e.g.
- alkyl ethoxylated sulfates as well as alkyl propoxylated sulfates are contemplated herein.
- Specific examples of substituted ammonium cations include methyl-, dimethyl, trimethyl-ammonium cations and quaternary ammonium cations such as tetramethyl-ammonium and dimethyl piperdinium cations and those derived from alkylamines such as ethylamine, diethylamine, triethylamine, mixtures thereof, and the like.
- Exemplary surfactants are C 12 -C 18 alkyl polyethoxylate (1.0) sulfate (C 12 -C 18 E(1.0)M) , C 12 -C 18 alkyl polyethoxylate (2.25) sulfate (C 12 -C 18 (2.25)M, and C 12 -C ⁇ 8 alkyl polyethoxylate (3.0) sulfate (C 12 -C 18 E(3.0)M) , and C 12 -C 18 alkyl polyethoxylate (4.0) sulfate (Ci 2 _ C ⁇ 8 E(4.0)M) , wherein M is conveniently selected from sodium and potassium.
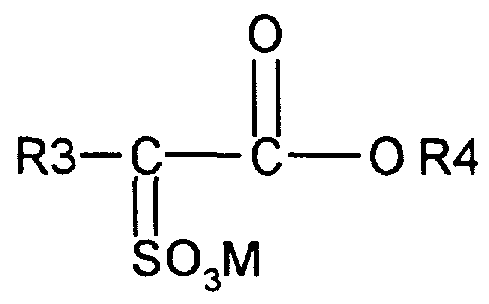
- Suitable anionic surfactants to be used are alkyl ester sulfonate surfactants including linear esters of C 3 -C 20 carboxylic acids (i.e., fatty acids) which are sulfonated with gaseous SO 3 according to "The Journal of the American Oil Chemists Society", 52 (1975), pp. 323-329.
- Suitable starting materials would include natural fatty substances as derived from tallow, palm oil, etc.
- alkyl ester sulfonate surfactant especially for laundry applications, comprise alkyl ester sulfonate surfactants of the structural formula:
- R 3 is a C 8 -C 2 o hydrocarbyl, preferably an alkyl, or combination thereof
- R 4 is a C--C 6 hydrocarbyl, preferably an alkyl, or combination thereof
- M is a cation which forms a water soluble salt with the alkyl ester sulfonate.
- Suitable salt-forming cations include metals such as sodium, potassium, and lithium, and substituted or unsubstituted ammonium cations, such as monoethanolamine, diethonolamine, and triethanolamine.
- R 3 is C 10 -C 16 alkyl
- R 4 is methyl, ethyl or isopropyl.
- methyl ester sulfonates wherein R 3 is C H3 -C 16 alkyl.
- suitable anionic surfactants include the alkyl sulfate surfactants which are water soluble salts or acids of the formula ROSO 3 M wherein R preferably is a C 10 -C 24 hydrocarbyl, preferably an alkyl or hydroxyalkyl having a C ⁇ o ⁇ C 20 alkyl component, more preferably a Ci 2 ⁇ C 18 alkyl or hydroxyalkyl, and M is H or a cation, e.g., an alkali metal cation (e.g. sodium, potassium, lithium) , or ammonium or substituted ammonium (e.g.
- alkylamines such as ethylamine, diethylamine, triethylamine, and mixtures thereof, and the like.
- alkyl chains of C 12 ⁇ C 16 are preferred for lower wash temperatures (e.g. below about 50°C) and Ci 6 ⁇ c i 8 alkyl chains are preferred for higher wash temperatures (e.g. above about 50°C) .
- anionic surfactants useful for detersive purposes can also be included in the laundry detergent compositions of the present invention. Theses can include salts (including, for example, sodium, potassium, ammonium, and substituted ammonium salts such as mono- di- and triethanolamine salts) of soap, C 8 - C 22 primary or secondary alkanesulfonates, C 8 -C 24 olefinsulfo- nates, sulfonated polycarboxylic acids prepared by sulfonation of the pyrolyzed product of alkaline earth metal citrates, e.g., as described in British patent specification No.
- salts including, for example, sodium, potassium, ammonium, and substituted ammonium salts such as mono- di- and triethanolamine salts
- C 8 - C 22 primary or secondary alkanesulfonates C 8 -C 24 olefinsulfo- nates
- sulfonated polycarboxylic acids prepared by sulfonation
- alkylpolyglycolethersulfates (containing up to 10 moles of ethylene oxide) ; alkyl glycerol sulfonates, fatty acyl glycerol sulfonates, fatty oleyl glycerol sulfates, alkyl phenol ethylene oxide ether sulfates, paraffin sulfonates, alkyl phosphates, isethionates such as the acyl isethionates, N-acyl taurates, alkyl succina ates and sulfosuccinates, monoesters of sulfosuccinates (especially saturated and unsaturated C 1 2-C 18 monoesters) and diesters of sulfosuccinates (especially saturated and unsaturated C & -Ci 2 diesters) , acyl sarcosinates, sulfates of alkyIpolysaccharide
- Alkylbenzene sulfonates are highly preferred. Especially preferred are linear (straight-chain) alkyl benzene sulfonates (LAS) wherein the alkyl group preferably contains from 10 to 18 carbon atoms.
- LAS linear (straight-chain) alkyl benzene sulfonates
- the laundry detergent compositions of the present invention typically comprise from about 1% to about 40%, preferably from about 3% to about 20% by weight of such anionic surfactants.
- the laundry detergent compositions of the present invention may also contain cationic, ampholytic, zwitterionic, and semi-polar surfactants, as well as the nonionic and/or anionic surfactants other than those already described herein.
- Cationic detersive surfactants suitable for use in the laundry detergent compositions of the present invention are those having one long-chain hydrocarbyl group. Examples of such cationic surfactants include the ammonium surfactants such as aIkyltrimethylammonium halogenides, and those surfactants having the formula:
- R 2 is an alkyl or alkyl benzyl group having from about 8 to about 18 carbon atoms in the alkyl chain
- each R 3 is selected form the group consisting of -CH 2 CH 2 -, -CH 2 CH(CH 3 ) -, - CH 2 CH(CH 2 OH)-, -CH 2 CH 2 CH 2 -, and mixtures thereof
- each R 4 is selected from the group consisting of C ⁇ ⁇ alkyl, C 1 -C 4 hydroxyalkyl, benzyl ring structures formed by joining the two R 4 groups, -CH 2 CHOHCHOHCOR 6 CHOHCH 2 OH, wherein R 6 is any hexose or hexose polymer having a molecular weight less than about 1000, and hydrogen when y is not 0
- R is the same as R or is an alkyl chain,wherein the total number
- Highly preferred cationic surfactants are the water soluble quaternary ammonium compounds useful in the present composition having the formula:
- Ri is C 8 -C 16 alkyl
- each of R 2 , R 3 and R is independently C 1 -C alkyl, C 1 -C 4 hydroxy alkyl, benzyl, and -(C 2 H 4 o) x H where x has a value from 2 to 5, and X is an anion.
- R 2 , R 3 or R 4 should be benzyl.
- the preferred alkyl chain length for Ri is C 12 ⁇ C 15 particularly where the alkyl group is a mixture of chain lengths derived from coconut or palm kernel fat or is derived synthetically by olefin build up or OXO alcohols synthesis.
- R 2 R 3 and R are methyl and hydroxyethyl groups and the anion X may be selected from halide, ethosulphate, acetate and phosphate ions.
- Suitable quaternary ammonium compounds of formulae (i) for use herein are: coconut trimethyl ammonium chloride or bromide; coconut methyl dihydroxyethyl ammonium chloride or bromide; decyl triethyl ammonium chloride; decyl dimethyl hydroxyethyl ammonium chloride or bromide; i 2 -i 5 dimethyl hydroxyethyl ammonium chloride or bromide; coconut dimethyl hydroxyethyl ammonium chloride or bromide; myristyl trimethyl ammonium methyl sulphate; lauryl dimethyl benzyl ammonium chloride or bromide; lauryl dimethyl (ethenoxy) 4 ammonium chloride or bromide; choline esters (compounds of formula (i) wherein Ri is
- the laundry detergent compositions of the present invention typically comprise from 0.2% to about 25%, preferably from about 1% to about 8% by weight of such cationic surfactants.
- Ampholytic surfactants are also suitable for use in the laundry detergent compositions of the present invention. These surfactants can be broadly described as aliphatic derivatives of secondary or tertiary amines, or aliphatic derivatives of heterocyclic secondary and tertiary amines in which the aliphatic radical can be straight- or branched-chain.
- One of the aliphatic substituents contains at least about 8 carbon atoms, typically from about 8 to about 18 carbon atoms, and at least one contains an anionic water-solubilizing group, e.g. carboxy, sulfonate, sulfate. See US 3,929,678 (column 19, lines 18-35) for examples of ampholytic surfactants.
- the laundry detergent compositions of the present invention typically comprise from 0.2% to about 15%, preferably from about 1% to about 10% by weight of such ampholytic surfactants.
- Zwitterionic surfactants are also suitable for use in laundry detergent compositions. These surfactants can be broadly described as derivatives of secondary and tertiary amines, derivatives of heterocyclic secondary and tertiary amines, or derivatives of quaternary ammonium, quaternary phos- phonium or tertiary sulfonium compounds. See US 3,929,678 (column 19, line 38 through column 22, line 48) for examples of zwitterionic surfactants.
- the laundry detergent composi- tions of the present invention typically comprise from 0.2% to about 15%, preferably from about 1% to about 10% by weight of such zwitterionic surfactants.
- Semi-polar nonionic surfactants are a special category of nonionic surfactants which include water-soluble amine oxides containing one alkyl moiety of from about 10 to about 18 carbon atoms and 2 moieties selected from the group consisting of alkyl groups and hydroxyalkyl groups containing from about 1 to about 3 carbon atoms; watersoluble phosphine oxides containing one alkyl moiety of form about 10 to about 18 carbon atoms and 2 moieties selected from the group consisting of alkyl groups and hydroxyalkyl groups containing from about 1 to about 3 carbon atoms; and water-soluble sulfoxides containing one alkyl moiety from about 10 to about 18 carbon atoms and a moiety selected from the group consisting of alkyl and hydroxyalkyl moieties of from about 1 to about 3 carbon atoms.
- Semi-polar nonionic detergent surfactants include the amine oxide surfactants having the formula:
- R is an alkyl, hydroxyalkyl, or alkyl phenyl group or mixtures thereof containing from about 8 to about 22 carbon atoms;
- R is an alkylene or hydroxyalkylene group containing from about 2 to about 3 carbon atoms or mixtures thereof;
- x is from 0 to about 3 : and each R 5 is an alkyl or hydroxyalkyl group containing from about 1 to about 3 carbon atoms or a polyethylene oxide group containing from about 1 to about 3 ethylene oxide groups.
- the R 5 groups can be attached to each other, e.g., through an oxygen or nitrogen atom, to form a ring structure.
- These amine oxide surfactants in particular include C 10 ⁇ C 18 alkyl dimethyl amine oxides and C 8 -C 12 alkoxy ethyl dihydroxy ethyl amine oxides.
- the laundry detergent compositions of the present invention typically comprise from 0.2% to about 15%, preferably from about 1% to about 10% by weight of such semi-polar nonionic surfactants.
- compositions according to the present invention may further comprise a builder system.
- a builder system Any conventional builder system is suitable for use herein including aluminosilicate materials, silicates, polycarboxylates and fatty acids, materials such as ethylenediamine tetraacetate, metal ion sequestrants such as aminopolyphosphonates, particularly ethylenediamine tetramethylene phosphonic acid and diethylene triamine pentamethylenephosphonic acid.
- phosphate builders can also be used herein.
- Suitable builders can be an inorganic ion exchange material, commonly an inorganic hydrated aluminosilicate material, more particularly a hydrated synthetic zeolite such as hydrated zeolite A, X, B, HS or MAP.
- SKS-6 is a crystalline layered silicate consisting of sodium silicate (Na 2 Si 2 0 5 ) .
- Suitable polycarboxylates containing one carboxy group include lactic acid, glycolic acid and ether derivatives thereof as disclosed in Belgian Patent Nos. 831,368, 821,369 and 821,370.
- Polycarboxylates containing two carboxy groups include the water-soluble salts of succinic acid, malonic acid, (ethylenedioxy) diacetic acid, maleic acid, diglycollic acid, tartaric acid, tartronic acid and fumaric acid, as well as the ether carboxylates described in German Offenle-enschrift 2,446,686, and 2,446,487, US 3,935,257 and the sulfinyl carboxylates described in Belgian Patent No. 840,623.
- Polycarboxylates containing three carboxy groups include, in particular, water-soluble citrates, aconitrates and citraconates as well as succinate derivatives such as the carboxymethyloxysuccinates described in British Patent No. 1,379,241, lactoxysuccinates described in Netherlands Application 7205873, and the oxypolycarboxylate materials such as 2-oxa-l, 1, 3-propane tricarboxylate ⁇ described in British Patent No. 1,387,447.
- Polycarboxylates containing four carboxy groups include oxydisuccinates disclosed in British Patent No. 1,261,829, 1,1,2,2, -ethane tetracarboxylates, 1,1,3,3-propane tetracarbo- xylates containing sulfo substituents include the sulfosuc- cinate derivatives disclosed in British Patent Nos. 1,398,421 and 1,398,422 and in US 3,936,448, and the sulfonated pyrolysed citrates described in British Patent No. 1,082,179, while polycarboxylates containing phosphone substituents are disclosed in British Patent No. 1,439,000.
- Alicyclic and heterocyclic polycarboxylates include cyclopentane-cis , cis-cis-tetracarboxylates, cyclopentadienide pentacarboxylates , 2 , 3 , 4 , 5-tetrahydro-furan - cis, cis, cis- tetracarboxylates, 2 , 5-tetrahydro-furan-cis, discarboxylates, 2 , 2 , 5,5, -tetrahydrofuran - tetracarboxylates, 1,2,3,4,5,6- hexane - hexacarboxylates and carboxymethyl derivatives of polyhydric alcohols such as sorbitol, annitol and xylitol.
- Aromatic polycarboxylates include mellitic acid, pyromellitic acid and the phthalic acid derivatives disclosed in British Patent No. 1,425,343.
- the preferred polycarboxylates are hydroxy-carboxylates containing up to three carboxy groups per molecule, more particularly citrates.
- Preferred builder systems for use in the present compositions include a mixture of a water-insoluble aluminosilicate builder such as zeolite A or of a layered silicate (SKS-6) , and a water-soluble carboxylate chelating agent such as citric acid.
- a suitable chelant for inclusion in the detergent composi-ions in accordance with the invention is ethylenediamine-N,N'-disuccinic acid (EDDS) or the alkali metal, alkaline earth metal, ammonium, or substituted ammonium salts thereof, or mixtures thereof.
- Preferred EDDS compounds are the free acid form and the sodium or magnesium salt thereof.
- Examples of such preferred sodium salts of EDDS include Na EDDS and Na 4 EDDS.
- Examples of such preferred magnesium salts of EDDS include MgEDDS and Mg 2 EDDS.
- the magnesium salts are the most preferred for inclusion in compositions in accordance with the invention.
- Preferred builder systems include a mixture of a water- insoluble aluminosilicate builder such as zeolite A, and a water soluble carboxylate chelating agent such as citric acid.
- builder materials that can form part of the builder system for use in granular compositions include inorganic materials such as alkali metal carbonates, bicarbonates, silicates, and organic materials such as the organic phosphonates, amino polyalkylene phosphonates and amino polycarboxylates .
- Suitable water-soluble organic salts are the homo- or co-polymeric acids or their salts, in which the polycarboxylie acid comprises at least two carboxyl radicals separated form each other by not more than two carbon atoms.
- salts are polyacrylates of MW 2000-5000 and their copolymers with maleic anhydride, such copolymers having a molecular weight of from 20,000 to 70,000, especially about
- Detergency builder salts are normally included in amounts of from 5% to 80% by weight of the composition. Preferred levels of builder for liquid detergents are from 5% to 30%.
- Preferred detergent compositions in addition to the enzyme preparation of the invention, comprise other enzyme (s) which provides cleaning performance and/or fabric care benefits.
- enzymes include proteases, other Upases, cutinases, amylases, cellulases, peroxidases, oxidases (e.g. laccases) .
- protease suitable for use in alkaline solutions can be used.
- Suitable proteases include those of animal, vegetable or microbial origin. Microbial origin is preferred. Chemically or genetically modified mutants are included.
- the protease may be a serine protease, preferably an alkaline microbial protease or a trypsin-like protease.
- alkaline proteases are subtilisins, especially those derived from Bacillus, e.g., subtilisin Novo, subtili ⁇ in Carlsberg, subtilisin 309, subtilisin 147 and subtilisin 168 (described in WO 89/06279) .
- trypsin-like proteases are trypsin (e.g. of porcine or bovine origin) and the Fusariu protease described in WO 89/06270.
- Preferred commercially available protease enzymes include those sold under the trade names Alcalase, Savinase, Primase, Durazym, and Esperase by Novo Nordisk A/S (Denmark) , those sold under the tradename Maxatase, Maxacal, Maxapem and Properase by Gist-Brocades, those sold under the tradename Purafect and Purafect OXP by Genencor International, and those sold under the tradename Opticlean and Optimase by Solvay Enzymes.
- Protease enzymes may be incorporated into the composi- tions in accordance with the invention at a level of from 0.00001% to 2% of enzyme protein by weight of the composition, preferably at a level of from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level of from 0.001% to 0.5% of enzyme protein by weight of the composition, even more preferably at a level of from 0.01% to 0.2% of enzyme protein by weight of the composition.
- Lipases Any lipase suitable for use in alkaline solutions can be used. Suitable lipases include those of bacterial or fungal origin. Chemically or genetically modified mutants are included. Examples of useful lipases include a Humicola lanu i- nosa lipase, e.g., as described in EP 258 068 and EP 305 216, a Rhizomucor miehei lipase, e.g., as described in EP 238 023, a Candida lipase, such as a C.
- Suitable lipases include those of bacterial or fungal origin. Chemically or genetically modified mutants are included. Examples of useful lipases include a Humicola lanu i- nosa lipase, e.g., as described in EP 258 068 and EP 305 216, a Rhizomucor miehei lipase, e.g., as described in EP 238 023, a Candida lipase, such as a C.
- antarctica lipase e.g., the c_j_ antarctica lipase A or B described in EP 214 761
- a Pseudomonas lipase such as a P. alcaliqenes and P. pseudoalcaligenes lipase, e.g., as described in EP 218 272
- a P. cepacia lipase e.g., as described in EP 331 376
- a P. stutzeri lipase e.g., as disclosed in GB 1,372,034
- a P. fluorescens lipase a Bacil- lus lipase, e.g., a B.
- subtilis lipase (Dartois et al. , (1993), Biochemica et Biophysica acta 1131, 253-260) , a B. stearo- thermophilus lipase (JP 64/744992) and a B. pumilus lipase (WO 91/16422) .
- cloned lipases may be useful, including the PeniciIlium camembertii lipase described by Ya aguchi et al., (1991), Gene 103, 61-67), the Geotricum candidum lipase (Schimada, Y. et al., (1989), J. Biochem., 106, 383-388) , and various Rhizopus lipases such as a R. delemar lipase (Hass, M.J et al., (1991), Gene 109, 117-113), a R ⁇ niveus lipase (Kugimiya et al., (1992), Biosci. Biotech. Biochem. 56, 716-719) and a R. oryzae lipase.
- R. delemar lipase Hass, M.J et al., (1991), Gene 109, 117-113
- R ⁇ niveus lipase K
- cutinases may also be useful, e.g., a cutinase derived from Pseudomonas mendocina as described in WO 88/09367, or a cutinase derived from Fusarium solani pisi (e.g. described in WO 90/09446) .
- lipases such as Ml LipaseTM, TM TM •
- the lipases are normally incorporated in the detergent composition at a level of from 0.00001% to 2% of enzyme protein by weight of the composition, preferably at a level of from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level of from 0.001% to 0.5% of enzyme protein by weight of the composition, even more preferably at a level of from 0.01% to 0.2% of enzyme protein by weight of the composition.
- Amylases Any amylase ( ⁇ and/or ⁇ ) suitable for use in alkaline solutions can be used.
- Suitable amylases include those of bacterial or fungal origin. Chemically or genetically modified mutants are included. Amylases include, for example, ⁇ - amylases obtained from a special strain of B. licheniformis, described in more detail in GB 1,296,839. Commercially available amylases are DuramylTM, TermamylTM, FungamylTM and BANTM (available from Novo Nordisk A/S) and RapidaseTM and Maxamyl PTM (available from Genencor) .
- amylases are normally incorporated in the detergent composition at a level of from 0.00001% to 2% of enzyme protein by weight of the composition, preferably at a level of from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level of from 0.001% to 0.5% of enzyme protein by weight of the composition, even more preferably at a level of from 0.01% to 0.2% of enzyme protein by weight of the composition.
- Cellulases Any cellulase suitable for use in alkaline solutions can be used. Suitable cellulases include those of bacterial or fungal origin. Chemically or genetically modified mutants are included. Suitable cellulases are disclosed in US 4,435,307, which discloses fungal cellulases produced from Humicola insolens. Especially suitable cellulases are the cellulases having colour care benefits. Examples of such cellulases are cellulases described in European patent application No. 0 495 257.
- cellulases include CelluzymeTM produced by a strain of Humicola insolens. (Novo Nordisk A/S) . and KAC- 500(B)TM (Kao Corporation).
- Cellulases are normally incorporated in the detergent composition at a level of from 0.00001% to 2% of enzyme protein by weight of the composition, preferably at a level of from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level of from 0.001% to 0.5% of enzyme protein by weight of the composition, even more preferably at a level of from 0.01% to 0.2% of enzyme protein by weight of the 5 composition.
- Peroxidases/Oxidases Peroxidases/Oxidases : Peroxidase enzymes are used in combination with hydrogen peroxide or a source thereof (e.g. a percarbonate, perborate or persulfate) . Oxidase enzymes are
- Both types of enzymes are used for "solution bleaching", i.e. to prevent transfer of a textile dye from a dyed fabric to another fabric when said fabrics are washed together in a wash liquor, preferably together with an enhancing agent as described in e.g. WO 94/12621 and WO
- Suitable peroxidase ⁇ /oxidases include those of plant, bacterial or fungal origin. Chemically or genetically modified mutants are included.
- Peroxidase and/or oxidase enzymes are normally incorporated in the detergent composition at a level of from
- enzyme protein by weight of the composition preferably at a level of from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level of from 0.001% to 0.5% of enzyme protein by weight of the composition, even more preferably at a level of from 0.01% to
- Mixtures of the above mentioned enzymes are encompassed herein, in particular a mixture of a protease, an amylase, a lipase and/or a cellulase.
- the enzyme of the invention or any other enzyme
- the detergent composition 30 incorporated in the detergent composition is normally incorporated in the detergent composition at a level from 0.00001% to 2% of enzyme protein by weight of the composition, preferably at a level from 0.0001% to 1% of enzyme protein by weight of the composition, more preferably at a level from
- Bleaching agents Additional optional detergent ingredients that can be included in the detergent compositions of the present invention include bleaching agents such as PB1, PB4 and percarbonate with a particle size of 400-800 microns. These bleaching agent components can include one or more oxygen bleaching agents and, depending upon the bleaching agent chosen, one or more bleach activators. When present oxygen bleaching compounds will typically be present at levels of from about 1% to about 25%. In general, bleaching compounds are optional added components in non-liquid formulations, e.g. granular detergents.
- the bleaching agent component for use herein can be any of the bleaching agents useful for detergent compositions including oxygen bleaches as well as others known in the art.
- the bleaching agent suitable for the present invention can be an activated or non-activated bleaching agent.
- oxygen bleaching agent that can be used encompasses percarboxylic acid bleaching agents and salts thereof. Suitable examples of this class of agents include magnesium monoperoxyphthalate hexahydrate, the magnesium salt of meta-chloro perbenzoic acid, 4-nonylamino-4-oxoperoxybutyric acid and diperoxydodecanedioic acid.
- Such bleaching agents are disclosed in US 4,483,781, US 740,446, EP 0 133 354 and US 4,412,934.
- Highly preferred bleaching agents also include 6- nonylamino-6-oxoperoxycaproic acid as described in US 4,634,551.
- bleaching agents that can be used encompasses the halogen bleaching agents.
- hypohalite bleaching agents include trichloro isocyanuric acid and the sodium and potassium dichloroiso- cyanurates and N-chloro and N-bromo alkane sulphonamides .
- Such materials are normally added at 0.5-10% by weight of the finished product, preferably 1-5% by weight.
- the hydrogen peroxide releasing agents can be used in combination with bleach activators such as tetra- acetylethylenediamine (TAED) , nonanoyloxybenzenesulfonate (NOBS, described in US 4,412,934), 3 , 5-trimethyl-hexsanoloxy- benzenesulfonate (ISONOBS, described in EP 120 591) or pentaacetylglucose (PAG) , which are perhydrolyzed to form a peracid as the active bleaching species, leading to improved bleaching effect.
- bleach activators such as tetra- acetylethylenediamine (TAED) , nonanoyloxybenzenesulfonate (NOBS, described in US 4,412,934), 3 , 5-trimethyl-hexsanoloxy- benzenesulfonate (ISONOBS, described in EP 120 591) or pentaacetylglucose (
- bleach activators C8 (6-octanamido-caproyl) oxybenzene-sulfonate, C9(6- nonanamido caproyl) oxybenzenesulfonate and CIO (6-decanamido caproyl) oxybenzenesulfonate or mixtures thereof.
- acylated citrate esters such as disclosed in European Patent Application No. 91870207.7.
- bleaching agents including peroxyacids and bleaching systems comprising bleach activators and peroxygen bleaching compounds for use in cleaning compositions according to the invention are described in application USSN 08/136,626.
- the hydrogen peroxide may also be present by adding an enzymatic system (i.e. an enzyme and a substrate therefore) which is capable of generation of hydrogen peroxide at the beginning or during the washing and/or rinsing process.
- an enzymatic system i.e. an enzyme and a substrate therefore
- Such enzymatic systems are disclosed in European Patent Application EP 0 537 381.
- Bleaching agents other than oxygen bleaching agents are also known in the art and can be utilized herein.
- One type of non-oxygen bleaching agent of particular interest includes photoactivated bleaching agents such as the sulfonated zinc and/or aluminium phthalocyanines. These materials can be deposited upon the substrate during the washing process. Upon irradiation with light, in the presence of oxygen, such as by hanging clothes out to dry in the daylight, the sulfonated zinc phthalocyanine is activated and, consequently, the substrate is bleached.
- Preferred zinc phthalocyanine and a photoactivated bleaching process are described in US 4,033,718.
- detergent composition will contain about 0.025% to about 1.25%, by weight, of sulfonated zinc phthalocyanine.
- Bleaching agents may also comprise a manganese catalyst.
- the manganese catalyst may, e.g., be one of the compounds described in "Efficient manganese catalysts for low- temperature bleaching", Nature 369, 1994, pp. 637-639.
- Suds ⁇ uppressors Another optional ingredient is a sud ⁇ suppressor, exemplified by silicones, and silica-silicone mixtures. Silicones can generally be represented by alkylated polysiloxane materials, while silica is normally used in finely divided forms exemplified by silica aerogels and xerogel ⁇ and hydrophobic silicas of various types.
- suds suppre ⁇ or can be incorporated as particulates, in which the suds suppre ⁇ or is advantageously releasably incorporated in a water-soluble or waterdispersible, substantially non surface-active detergent impermeable carrier.
- the suds suppressor can be dissolved or dispersed in a liquid carrier and applied by spraying on to one or more of the other components.
- a preferred silicone suds controlling agent is disclo- sed in US 3,933,672.
- Other particularly useful suds suppressors are the self-emulsifying silicone suds suppressors, described in German Patent Application DTOS 2,646,126.
- An example of such a compound is DC-544, commercially available form Dow Corning, which is a siloxane-glycol copolymer.
- Especially preferred sud ⁇ controlling agent are the suds suppressor system comprising a mixture of silicone oils and 2-alkyl-alkanols. Suitable 2- alkyl-alkanols are 2-butyl-octanol which are commercially available under the trade name Isofol 12 R.
- compositions can comprise a silicone/ silica mixture in combination with fumed nonporous silica such as Aerosil R .
- the suds suppressors described above are normally employed at levels of from 0.001% to 2% by weight of the composition, preferably from 0.01% to 1% by weight.
- Other components may be employed such as soil-suspending agents, soil- releasing agents, optical brighteners, abrasives, bactericides, tarnish inhibitors, coloring agents, and/or encapsulated or nonencapsulated perfumes.
- encapsulating materials are water soluble capsules which consist of a matrix of polysaccharide and polyhydroxy compounds such as described in GB 1,464,616.
- Suitable water soluble encapsulating materials comprise dextrins derived from ungelatinized starch acid esters of substituted dicarboxylic acids such as described in US 3,455,838. These acid-ester dextrins are, preferably, prepared from such starches as waxy maize, waxy sorghum, sago, tapioca and potato. Suitable examples of said encapsulation materials include N-Lok manufactured by National Starch. The N-Lok encapsulating material consists of a modified maize starch and glucose. The starch is modified by adding monofunctional sub- stituted group ⁇ such as octenyl succinic acid anhydride.
- Antiredeposition and soil suspension agents suitable herein include cellulose derivatives such as methylcellulose, carboxymethylcellulose and hydroxyethylcellulose, and homo- or co-polymeric polycarboxylic acids or their salts.
- Polymers of this type include the polyacrylates and maleic anhydride- acrylic acid copolymers previously mentioned as builders, as well as copolymers of maleic anhydride with ethylene, methyl- vinyl ether or methacrylic acid, the maleic anhydride constituting at least 20 mole percent of the copolymer. These materials are normally used at levels of from 0.5% to 10% by weight, more preferably form 0.75% to 8%, most preferably from 1% to 6% by weight of the composition.
- Preferred optical brighteners are anionic in character, examples of which are disodium 4,4 - -bis-(2-diethanolamino-4- anilino -s- triazin-6-ylamino) ⁇ tilbene-2 :2 ⁇ disulphonate, disodium 4, - 4 '-bis-(2-morpholino-4-anilino-s-triazin-6-ylamino- stilbene-2 : 2 ' - disulphonate, disodium 4,4' - bis- (2 ,4-dianili- no-s-triazin-6-ylamino) stilbene-2 :2 ' - disulphonate, monosodium 4-,4'* - bis-(2,4-dianilino-s-tri-azin-6 ylamino) stilbene-2- sulphonate, disodium 4,4' -bis-(2-anilino-4-(N-methyl-N-2 ⁇ hydroxyethyla ino) -s-
- polyethylene glycols particularly those of molecular weight 1000-10000, more particularly 2000 to 8000 and most preferably about 4000. These are used at levels of from 0.20% to 5% more preferably from 0.25% to 2.5% by weight. These polymer ⁇ and the previou ⁇ ly mentioned homo- or co-polymeric poly-carboxylate salts are valuable for improving whiteness maintenance, fabric ash deposition, and cleaning performance on clay, proteinaceous and oxidizable ⁇ oils in the presence of transition metal impurities.
- Soil release agents useful in composition ⁇ of the present invention are conventionally copolymers or terpolymers of terephthalic acid with ethylene glycol and/or propylene glycol units in various arrangements. Examples of such polymers are disclosed in US 4,116,885 and 4,711,730 and EP 0 272 033.
- a particular preferred polymer in accordance with EP 0 272 033 has the formula:
- PEG is -(OC 2 H 4 )0-
- PO is (OC 3 H 6 0)
- T is (pOOC 6 H 4 CO) .
- modified polyesters as random copolymers of dimethyl terephthalate, dimethyl sulfoisophthalate, ethylene glycol and 1,2-propanediol, the end groups consisting primarily of sulphobenzoate and secondarily of mono esters of ethylene glycol and/or 1,2-propanediol.
- the target is to obtain a polymer capped at both end by sulphobenzoate groups, "pri a- rily", in the present context most of said copolymers herein will be endcapped by sulphobenzoate groups.
- polyesters herein contain about 46% by weight of dimethyl terephthalic acid, about 16% by weight of 1,2-propanediol, about 10% by weight ethylene glycol, about 13% by weight of dimethyl sulfobenzoic acid and about 15% by weight of sulfoisophthalic acid, and have a molecular weight of about 3.000.
- the polyesters and their method of preparation are described in detail in EP 311 342.
- Fabric softening agents can also be incorporated into laundry detergent compositions in accordance with the present invention. These agents may be inorganic or organic in type. Inorganic softening agents are exemplified by the smectite clays disclosed in GB-A-1 400898 and in US 5,019,292. Organic fabric softening agents include the water insoluble tertiary amines as disclo ⁇ ed in GB-A1 514 276 and EP 0 011 340 and their combination with mono C 12 ⁇ c l4 quaternary ammonium salts are disclosed in EP-B-0 026 528 and di-long- chain amides as disclosed in EP 0 242 919. Other useful organic ingredients of fabric softening systems include high molecular weight polyethylene oxide materials as disclosed in EP 0 299 575 and 0 313 146.
- Levels of smectite clay are normally in the range from 5% to 15%, more preferably from 8% to 12% by weight, with the material being added as a dry mixed component to the remainder of the formulation.
- Organic fabric softening agents such as the water-insoluble tertiary amines or dilong chain amide materials are incorporated at levels of from 0.5% to 5% by weight, normally from 1% to 3% by weight whilst the high molecular weight polyethylene oxide materials and the water soluble cationic materials are added at levels of from 0.1% to 2%, normally from 0.15% to 1.5% by weight.
- the detergent compositions according to the present invention may also comprise from 0.001% to 10%, preferably from 0.01% to 2%, more preferably form 0.05% to 1% by weight of polymeric dye- transfer inhibiting agents.
- Said polymeric dye-transfer inhibiting agents are normally incorporated into detergent compositions in order to inhibit the transfer of dyes from colored fabrics onto fabrics washed therewith. These polymers have the ability of complexing or adsorbing the fugitive dyes washed out of dyed fabrics before the dyes have the opportunity to become attached to other articles in the wash.
- Especially suitable polymeric dye-transfer inhibiting agents are polyamine N-oxide polymers, copolymers of N-vinyl- pyrrolidone and N-vinylimidazole, polyvinylpyrrolidone polymers, polyvinyloxazolidones and polyvinylimidazole ⁇ or mixtures thereof.
- the detergent composition according to the invention can be in liquid, paste, gels, bars or granular forms.
- Non-dusting granulates may be produced, e.g. , as disclosed in US 4,106,991 and 4,661,452 (both to Novo Industri A/S) and may optionally be coated by methods known in the art.
- waxy coating materials are poly (ethylene oxide) products (polyethyleneglycol, PEG) with mean molecular weights of 1000 to 20000; ethoxylated nonylphenols having from 16 to 50 ethylene oxide units; ethoxylated fatty alcohols in which the alcohol contains from 12 to 20 carbon atoms and in which there are 15 to 80 ethylene oxide units; fatty alcohols; fatty acids; and mono- and di- and triglycerides of fatty acids.
- PEG poly (ethylene oxide) products
- Granular compositions according to the present invention can al ⁇ o be in "compact form", i.e. they may have a relatively higher density than conventional granular detergents, i.e. form 550 to 950 g/1; in such case, the granu- lar detergent compositions according to the present invention will contain a lower amount of "Inorganic filler salt", compared to conventional granular detergents; typical filler salts are alkaline earth metal salts of sulphates and chlorides, typically sodium sulphate; "Compact" detergent typically comprise not more than 10% filler salt.
- the liquid compositions according to the present invention can also be in "concentrated form", in such case, the liquid detergent compositions according to the present invention will contain a lower amount of water, compared to conventional liquid detergents.
- the water content of the concentrated liquid detergent is le ⁇ than 30%, more preferably less than 20%, most preferably less than 10% by weight of the detergent compositions .
- the compositions of the invention may for example, be formulated as hand and machine laundry detergent compositions including laundry additive compositions and compositions suitable for use in the pretreatment of stained fabrics, rinse added fabric softener compositions, and compositions for use in general household hard surface cleaning operations and dishwashing operations.
- TAS Sodium tallow alkyl sulphate
- XYEZS C lx - C ⁇ sodium alkyl sulfate condensed with an average of Z moles of ethylene oxide per mole
- Nonionic C 13 - C 15 mixed ethoxylated/propoxylated fatty alcohol with an average degree of ethoxylation of 3.8 and an average degree of propoxylation of 4.5 sold under the tradename Plurafax LF404 by BASF Gmbh
- CFAA C 12 - C 1 alkyl N-methyl glucamide
- TFAA C 16 - C 18 alkyl N-methyl glucamide
- NaSKS-6 Crystalline layered silicate of formula ⁇ -Na 2 Si 2 0 5
- MA/AA Copolymer of 1:4 maleic/acrylic acid, average molecular weight about 80,000
- Polyacrylate Polyacrylate ho opolymer with an average molecular weight of 8,000 sold under the tradename PA30 by BASF Gmbh
- Zeolite A Hydrated Sodium Aluminosilicate of formula Na- L2 (A10 2 Si ⁇ 2) i 2 « 27H 2 O having a primary particle size in the range from 1 to 10 micrometers
- Citrate Tri-sodium citrate dihydrate
- Perborate Anhydrous sodium perborate monohydrate bleach, empirical formula NaB0 2 .H 2 0 2
- Percarbonate Anhydrous sodium percarbonate bleach of empirical formula 2Na 2 C0 3 .3H 2 0 2
- TAED Tetraacetyl ethylene diamine
- CMC Sodium carboxymethyl cellulose
- DETPMP Diethylene triamine penta (methylene phosphonic acid) , marketed by Monsanto under the Tradename Dequest 2060
- Granular Suds suppressor 12% Silicone/silica, 18% stearyl alcohol, 70% starch in granular form
- HMWPEO High molecular weight polyethylene oxide
- a granular fabric cleaning composition in accordance with the invention may be prepared as follows:
- Enzyme of the invention 0.1
- Enzyme of the invention 0.1
- Granular fabric cleaning compositions in accordance with the invention which are especially useful in the laundering of coloured fabrics were prepared as follows: LAS 10.7 TAS 2.4 —
- Enzyme of the invention 0.10 0.05
- Granular fabric cleaning composition ⁇ in accordance with the invention which provide "Softening through the wash” capability may be prepared as follows:
- Zeolite A 15.0 15.0 MA/AA 4.0 4.0
- Enzyme of the invention 0.10 0.05
- Heavy duty liquid fabric cleaning compositions in accordance with the invention may be prepared as follows:
- Enzyme of the invention 0.10 0.05
- Thermoalcalibacter bogoriae DSM No. 9380 comprises the CGTase of the invention.
- Thermoalcalibacter bogoriae was cultivated under anaerobic conditions in the following medium: (NH 4 ) 2 S0 4 , 1.0;
- Resazurin 0.001; NaHC0 3 , 2.2; Na2C0 3 , 2.2; Cystein, 0.5, Starch,
- the concentrated supernatant was applied to a PD-10 ion exchange column (Pharmacia) and eluated with 100 mM sodium phosphate buffer pH 9.0.
- the eluate containing amylolytic activity was collected and concentrated 10-fold in an Amicon chamber (10 kDa filter Amicon) .
- Sample ⁇ of thi ⁇ solution were applied to a Q-Sepharose anion exchange chromatography column (15 x 2.5 cm) (Pharmacia) preequilibrated with 100 mM sodium phosphate buffer pH 9.0. The column was washed with 90 ml of equilibrating buffer.
- the enzyme solution was eluated with equlibration buffer containing 1 M NaCl, using a gradient of NaCl from 0 to 300 mM and 300 to 500 mM at a flow rate of 0.2 ml/min. Fractions were collected (2 ml per tube) and their amylolytic activity was determined as described herein. The active fractions were collected, assembled and subsequently 10-fold concentrated in a Amicon chamber. Samples of this prepurified amylase were added to a Superose 75 gel filtration column (Pharmacia) preequilibrated with 50 mM sodium phosphate buffer pH 9.0. The enzyme was eluated with the equilibration buffer at a flow rate of 0.1 ml/min. The fractions were collected (1 ml/tube) and the active fractions were pooled and subsequently concentrated in an Amicon chamber with a 10 kDa membrane.
- the SDS gel Prior to activity staining, the SDS gel was incubated for 30 min in a 2.5% Triton X-100 solution in order to remove SDS. Amylolytic protein bands were detected by incubating the gel for 10 min at 65°C in 100 mM sodium phosphate buffer pH 9.0 for 10 amylase, supplemented with 0.5% soluble starch (Merck). Amylase bands were visualized by staining the gel with a KJ-J 2 solution (3 g KJ, 2 g J 2 per liter aqua dest.), resulting in white activity bands within a brownish background.
- KJ-J 2 solution 3 g KJ, 2 g J 2 per liter aqua dest.
- the enzyme assay routinely used was carried out with enzyme solution using the respective prepurified enzyme and substrate solutions at 0.5% soluble starch (Merk, Darmstadt, Germany) or 0.2% amylose or 0.2% amylopectine (each wt/vol) in
- 25 vity was calculated using a standard calibartion curve with 0-1% (wt/vol) maltose.
- One unit (U) of amylolytic activity was defined as the amount of 1 mmol reducing sugars liberated by the enzyme per minute under standard conditions (pH 9.0; 65°C) .
- Protein concentrations were determined by the Lowry method. Microassays were performed and bovine serum albumine was used as standard protein.
- the prepurified enzyme solution was used. 10ml of the enzyme solution were mixed with 90 ml of a 0.5% (wt/vol) substrate solution (soluble starch; Merck) in 120 M universal Britton & Robinsson buffer with pH 4.0 to 11.0. The changes in pH due to the mixture of the enzyme solution and substrate solution were measured. After a preincubation for 30 min of this mixture on ice, the enzyme assay was performed at 65°C for 30 min. The developed reducing sugars were plotted against the respective pH value.
- a 0.5% (wt/vol) substrate solution soluble starch; Merck
- Substrate specificity In order to determine the substrate specificity of the a- amylase, the enzyme was incubated with substrate solution (each wt/vol) containing soluble starch (Merck) (0.5%), amylopectin (0.2%), amylose (0.2%), pullulan (0.2%), maltotriose, maltotetra- ose and maltopentose (each 0.1%). The assay was incubated for 30 min under standard conditions (65°C, pH 8.0) . The enzyme activity was determined, in the case of a-amylase, by measuring the amount of reducing sugars.
- substrate solution each wt/vol
- soluble starch containing soluble starch (Merck) (0.5%), amylopectin (0.2%), amylose (0.2%), pullulan (0.2%), maltotriose, maltotetra- ose and maltopentose (each 0.1%).
- the assay was incubated for
- hydrolysis products The hydrolysis pattern of amylase action on different substrates were analyzed by high-performance liquid chromatography (HPLC) (Knauer GmbH, Berlin, Germany) with an Aminex-HPX- 42 A column (300 by 7.8 mm; Bio-Rad, Hercules, Calif.). One part of the prepurified respective enzyme was incubated together with 9 parts of substrate solution pH 9.0 for amylase, at 65°C for up to 16 hours. After incubation the assays were kept frozen at - 20°C until they were analyzed.
- HPLC high-performance liquid chromatography
- Pullulan cyclodextrins and maltooligosaccharides were obtained from Sigma (St. Louis, Mo.). Chemicals for electropho- resis were purchased by Serva (Heidelberg, Germany) . Other chemicals were obtained from Merck (Darmstadt, Germany) .
- EXAMPLE l Purification of the amylase.
- the specific activity of the amyla ⁇ e in the 70-fold concentrated culture supernatant after cultivation was 0.096 U/mg. Due to the production of H 2 S during fermentation, as previously described, a purification using a PD-10 ion exchange column was neccessary in order to remove H 2 S, sulfides and other activity desturbing agents. After this treatement, the amount of detectable activity was raised to 0.48 U/mg. This effect was regardless to the used method for detection of reducing sugars (data not shown) .
- the concentrated culture supernatant revealed three activity bands in an SDS-PAGE electrophoresis gel ( Figure 1, lane 2) by activity staining.
- the lowest activity band with an apparent molecular weight of 57 ⁇ 3 kDa was shown to exhibit a- amylase activity (see below) .
- Samples of the 10-fold concentrated PD-10 eluate were applied to a Q-Sepharose anion exchange chromatography column (Pharmacia, Sweden; 25 x 200 mm) and the column was run at 1.0 ml/min with the equilibration buffer (100 mM sodium phosphate pH 8.0) using the Bio-Rad Econo System.
- the a-amylase was eluated by a NaCl-gradient up to 500 mM at 250 mM salt.
- the fractions containing a-amylase activity were collected and combined.
- the molecular mass of the amylase determined by the activity stained as well as the silver stained single band was estimated to be 57 ⁇ 3 kDa. ( Figure 1, lane 4 and 5).
- the prepurified enzyme exhibited an optimal pH between pH 8.0 and 9.0 with a broad range of activity between pH 5 and 10.5 ( Figure 2) .
- the amylase Under the assay conditions (65°C, 60 min) the amylase is stable in between this broad pH range.
- the optimal pH of the amylase fits with the optimal growth condition of the strain (pH 9.5). After 60 min incubation with 0.5% soluble starch (wt/vol) at the alkaline pH 10.0 the amylase exhibits high stability with only 30% loss of activity (Figure 2) .
- the temperature profile of the prepurified amylase shows a distinct optimal temperature for activity at 65°C ( Figure 3) .
- amylase activity was measured at pH 8.0 -higher pH would interfere with metal ion solutions- and 65°C in presence of various metal ions at 1 mM, 2 mM and 5 mM concentration and in presence of some chemical reagents (Figure 4) .
- Amylase activity was not or only slightly inhibited by the addition of various metal ions such as Co 2+ ,Ca 2+ and EDTA. Moderate inhibition occurred with Mg 2+ , Mo 7+ , Zn 2+ and Ni 2+ . Strong inhibition was observed by the addition of Cu 2+ , Fe 2+ , Fe 3+ , V 4+ and Cr 6+ . Although a moderate stimulation of amylase activity could be achieved upon addition of Mn 2+ , the enzyme is active when incubated in 100 mM sodium phosphate buffer only.
- Hayashi, T., T. Akiba, and K. Horokoshi. 1988 Production and purification of new maltohexaose-froming amylase ⁇ from alkalophilic Bacillus sp. H-167. Agric Biol Chem 52:443-448. Hofmann, B. E. , H. Bender, and G. E. Schulze. 1989. Three- dimensional structure of cyclodextrin glycosyltran ⁇ ferase from Bacillus circulans at 3.4 resolution. J Mol Biol 209: 793-800. Horikoshi, K.. 1991. General view of alkaliphiles and thermophiles, p. 3-14. In K. Horikoshi and W. D.
- Natronococcus amylolyticus sp. nov. a haloalkaliphilic archaeon.
- Cyclodextrin glycosyltransferase may be the only starch-degrading enzyme in
- thermostable pullulanase from Clostridium thermosul-furogenes EMI which hydrolyses both a-1,6 and a-l,4- glycosidic linkages. Appl Microbiol Biotechnol 33:511-518. Szejtli, J.. 1982. Cyclodextrins in food, cosmetics and toiletries. Starch 34:379-385.
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP97941876A EP0939801A1 (en) | 1996-09-26 | 1997-09-26 | An enzyme with amylase activity |
| AU43764/97A AU4376497A (en) | 1996-09-26 | 1997-09-26 | An enzyme with amylase activity |
| US09/271,783 US6165770A (en) | 1996-09-26 | 1999-03-18 | Alkaline stable amylase from Thermoalcalibacter |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DK1050/96 | 1996-09-26 | ||
| DK105096 | 1996-09-26 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/271,783 Continuation US6165770A (en) | 1996-09-26 | 1999-03-18 | Alkaline stable amylase from Thermoalcalibacter |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1998013481A1 true WO1998013481A1 (en) | 1998-04-02 |
Family
ID=8100417
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/DK1997/000410 WO1998013481A1 (en) | 1996-09-26 | 1997-09-26 | An enzyme with amylase activity |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP0939801A1 (en) |
| CN (1) | CN1231693A (en) |
| AU (1) | AU4376497A (en) |
| WO (1) | WO1998013481A1 (en) |
Cited By (40)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7803604B2 (en) | 2000-07-28 | 2010-09-28 | Henkel Ag & Co. Kgaa | Amylolytic enzyme extracted from Bacillus sp. A 7-7 (DSM 12368) and washing and cleaning agents containing this novel amylolytic enzyme |
| US7888104B2 (en) | 2000-11-28 | 2011-02-15 | Henkel Ag & Co. Kgaa | Cyclodextrin glucanotransferase (CGTase), obtained from<I>Bacillus agaradherens<λ>(DSM 9948) and detergents and cleaning agents containing said novel cyclodextrin glucanotransferase |
| WO2016205755A1 (en) | 2015-06-17 | 2016-12-22 | Danisco Us Inc. | Bacillus gibsonii-clade serine proteases |
| WO2017079751A1 (en) | 2015-11-05 | 2017-05-11 | Danisco Us Inc | Paenibacillus sp. mannanases |
| WO2017079756A1 (en) | 2015-11-05 | 2017-05-11 | Danisco Us Inc | Paenibacillus and bacillus spp. mannanases |
| WO2017106676A1 (en) | 2015-12-18 | 2017-06-22 | Danisco Us Inc | Polypeptides with endoglucanase activity and uses thereof |
| WO2017192300A1 (en) | 2016-05-05 | 2017-11-09 | Danisco Us Inc | Protease variants and uses thereof |
| WO2017192692A1 (en) | 2016-05-03 | 2017-11-09 | Danisco Us Inc | Protease variants and uses thereof |
| WO2017210295A1 (en) | 2016-05-31 | 2017-12-07 | Danisco Us Inc. | Protease variants and uses thereof |
| WO2017219011A1 (en) | 2016-06-17 | 2017-12-21 | Danisco Us Inc | Protease variants and uses thereof |
| WO2018085524A2 (en) | 2016-11-07 | 2018-05-11 | Danisco Us Inc | Laundry detergent composition |
| WO2018118950A1 (en) | 2016-12-21 | 2018-06-28 | Danisco Us Inc. | Bacillus gibsonii-clade serine proteases |
| WO2018118917A1 (en) | 2016-12-21 | 2018-06-28 | Danisco Us Inc. | Protease variants and uses thereof |
| WO2018169750A1 (en) | 2017-03-15 | 2018-09-20 | Danisco Us Inc | Trypsin-like serine proteases and uses thereof |
| WO2018184004A1 (en) | 2017-03-31 | 2018-10-04 | Danisco Us Inc | Alpha-amylase combinatorial variants |
| WO2019036721A2 (en) | 2017-08-18 | 2019-02-21 | Danisco Us Inc | Alpha-amylase variants |
| WO2019108599A1 (en) | 2017-11-29 | 2019-06-06 | Danisco Us Inc | Subtilisin variants having improved stability |
| WO2019245705A1 (en) | 2018-06-19 | 2019-12-26 | Danisco Us Inc | Subtilisin variants |
| WO2019245704A1 (en) | 2018-06-19 | 2019-12-26 | Danisco Us Inc | Subtilisin variants |
| WO2020028443A1 (en) | 2018-07-31 | 2020-02-06 | Danisco Us Inc | Variant alpha-amylases having amino acid substitutions that lower the pka of the general acid |
| WO2020068486A1 (en) | 2018-09-27 | 2020-04-02 | Danisco Us Inc | Compositions for medical instrument cleaning |
| WO2020077331A2 (en) | 2018-10-12 | 2020-04-16 | Danisco Us Inc | Alpha-amylases with mutations that improve stability in the presence of chelants |
| WO2020112599A1 (en) | 2018-11-28 | 2020-06-04 | Danisco Us Inc | Subtilisin variants having improved stability |
| WO2020242858A1 (en) | 2019-05-24 | 2020-12-03 | Danisco Us Inc | Subtilisin variants and methods of use |
| WO2020247582A1 (en) | 2019-06-06 | 2020-12-10 | Danisco Us Inc | Methods and compositions for cleaning |
| WO2021080948A2 (en) | 2019-10-24 | 2021-04-29 | Danisco Us Inc | Variant maltopentaose/maltohexaose-forming alpha-amylases |
| EP3872174A1 (en) | 2015-05-13 | 2021-09-01 | Danisco US Inc. | Aprl-clade protease variants and uses thereof |
| WO2022047149A1 (en) | 2020-08-27 | 2022-03-03 | Danisco Us Inc | Enzymes and enzyme compositions for cleaning |
| WO2022165107A1 (en) | 2021-01-29 | 2022-08-04 | Danisco Us Inc | Compositions for cleaning and methods related thereto |
| WO2023278297A1 (en) | 2021-06-30 | 2023-01-05 | Danisco Us Inc | Variant lipases and uses thereof |
| WO2023034486A2 (en) | 2021-09-03 | 2023-03-09 | Danisco Us Inc. | Laundry compositions for cleaning |
| WO2023114988A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Variant maltopentaose/maltohexaose-forming alpha-amylases |
| WO2023114932A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023114939A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023114936A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023168234A1 (en) | 2022-03-01 | 2023-09-07 | Danisco Us Inc. | Enzymes and enzyme compositions for cleaning |
| WO2023250301A1 (en) | 2022-06-21 | 2023-12-28 | Danisco Us Inc. | Methods and compositions for cleaning comprising a polypeptide having thermolysin activity |
| WO2024050343A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Subtilisin variants and methods related thereto |
| WO2024050339A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Mannanase variants and methods of use |
| WO2024050346A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Detergent compositions and methods related thereto |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7759093B2 (en) * | 2003-03-06 | 2010-07-20 | Verenium Corporation | Amylases, nucleic acids encoding them and methods for making and using them |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4600693A (en) * | 1984-02-13 | 1986-07-15 | Corning Glass Works | Thermostable alpha amylase having a low requirement for calcium ions, derived from a bacillus microorganism |
-
1997
- 1997-09-26 AU AU43764/97A patent/AU4376497A/en not_active Abandoned
- 1997-09-26 CN CN 97198262 patent/CN1231693A/en active Pending
- 1997-09-26 WO PCT/DK1997/000410 patent/WO1998013481A1/en not_active Application Discontinuation
- 1997-09-26 EP EP97941876A patent/EP0939801A1/en not_active Withdrawn
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4600693A (en) * | 1984-02-13 | 1986-07-15 | Corning Glass Works | Thermostable alpha amylase having a low requirement for calcium ions, derived from a bacillus microorganism |
Non-Patent Citations (3)
| Title |
|---|
| DIALOG INFORMATION SERVICES, File 357, DBA, Dialog Accession No. 208304, DBA Accession No. 97-034225, SHINKE R. et al., "Alkaline and Thermopholic Amylases of Industrial use - Bacillus sp. Thermostable Alpha-Amylase Production, Purification and Characterization (Conference Paper)"; & ANN. N.Y. ACAD. SCI., (799, 322-40), * |
| FILE WPI, Derwent Accession No. 87-062497, HITACHI LTD., "Preparing Thermo:Resistable Alpha Amylase by Culturing Thermophilic Anaerobic Clostridium Strain"; & JP,A,62 019 081, (27-01-87), DW8709. * |
| FILE WPI, Derwent Accession No. 90-096516, DAIWA KASEI KK., "Heat Resistance Alkali Amylase Obtain Culture Bacillus Bacteria Strain"; & JP,A,02 049 584, (19-02-90), DW9013. * |
Cited By (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7803604B2 (en) | 2000-07-28 | 2010-09-28 | Henkel Ag & Co. Kgaa | Amylolytic enzyme extracted from Bacillus sp. A 7-7 (DSM 12368) and washing and cleaning agents containing this novel amylolytic enzyme |
| US7888104B2 (en) | 2000-11-28 | 2011-02-15 | Henkel Ag & Co. Kgaa | Cyclodextrin glucanotransferase (CGTase), obtained from<I>Bacillus agaradherens<λ>(DSM 9948) and detergents and cleaning agents containing said novel cyclodextrin glucanotransferase |
| EP4219704A2 (en) | 2015-05-13 | 2023-08-02 | Danisco US Inc | Aprl-clade protease variants and uses thereof |
| EP3872174A1 (en) | 2015-05-13 | 2021-09-01 | Danisco US Inc. | Aprl-clade protease variants and uses thereof |
| WO2016205755A1 (en) | 2015-06-17 | 2016-12-22 | Danisco Us Inc. | Bacillus gibsonii-clade serine proteases |
| EP4234693A2 (en) | 2015-06-17 | 2023-08-30 | Danisco US Inc | Bacillus gibsonii-clade serine proteases |
| EP4141113A1 (en) | 2015-11-05 | 2023-03-01 | Danisco US Inc | Paenibacillus sp. mannanases |
| WO2017079751A1 (en) | 2015-11-05 | 2017-05-11 | Danisco Us Inc | Paenibacillus sp. mannanases |
| WO2017079756A1 (en) | 2015-11-05 | 2017-05-11 | Danisco Us Inc | Paenibacillus and bacillus spp. mannanases |
| WO2017106676A1 (en) | 2015-12-18 | 2017-06-22 | Danisco Us Inc | Polypeptides with endoglucanase activity and uses thereof |
| WO2017192692A1 (en) | 2016-05-03 | 2017-11-09 | Danisco Us Inc | Protease variants and uses thereof |
| EP3845642A1 (en) | 2016-05-05 | 2021-07-07 | Danisco US Inc. | Protease variants and uses thereof |
| WO2017192300A1 (en) | 2016-05-05 | 2017-11-09 | Danisco Us Inc | Protease variants and uses thereof |
| WO2017210295A1 (en) | 2016-05-31 | 2017-12-07 | Danisco Us Inc. | Protease variants and uses thereof |
| WO2017219011A1 (en) | 2016-06-17 | 2017-12-21 | Danisco Us Inc | Protease variants and uses thereof |
| EP4151726A1 (en) | 2016-06-17 | 2023-03-22 | Danisco US Inc | Protease variants and uses thereof |
| WO2018085524A2 (en) | 2016-11-07 | 2018-05-11 | Danisco Us Inc | Laundry detergent composition |
| WO2018112123A1 (en) | 2016-12-15 | 2018-06-21 | Danisco Us Inc. | Polypeptides with endoglucanase activity and uses thereof |
| WO2018118950A1 (en) | 2016-12-21 | 2018-06-28 | Danisco Us Inc. | Bacillus gibsonii-clade serine proteases |
| WO2018118917A1 (en) | 2016-12-21 | 2018-06-28 | Danisco Us Inc. | Protease variants and uses thereof |
| EP4212622A2 (en) | 2016-12-21 | 2023-07-19 | Danisco US Inc. | Bacillus gibsonii-clade serine proteases |
| WO2018169750A1 (en) | 2017-03-15 | 2018-09-20 | Danisco Us Inc | Trypsin-like serine proteases and uses thereof |
| WO2018184004A1 (en) | 2017-03-31 | 2018-10-04 | Danisco Us Inc | Alpha-amylase combinatorial variants |
| WO2019036721A2 (en) | 2017-08-18 | 2019-02-21 | Danisco Us Inc | Alpha-amylase variants |
| WO2019108599A1 (en) | 2017-11-29 | 2019-06-06 | Danisco Us Inc | Subtilisin variants having improved stability |
| WO2019245704A1 (en) | 2018-06-19 | 2019-12-26 | Danisco Us Inc | Subtilisin variants |
| WO2019245705A1 (en) | 2018-06-19 | 2019-12-26 | Danisco Us Inc | Subtilisin variants |
| WO2020028443A1 (en) | 2018-07-31 | 2020-02-06 | Danisco Us Inc | Variant alpha-amylases having amino acid substitutions that lower the pka of the general acid |
| WO2020068486A1 (en) | 2018-09-27 | 2020-04-02 | Danisco Us Inc | Compositions for medical instrument cleaning |
| WO2020077331A2 (en) | 2018-10-12 | 2020-04-16 | Danisco Us Inc | Alpha-amylases with mutations that improve stability in the presence of chelants |
| WO2020112599A1 (en) | 2018-11-28 | 2020-06-04 | Danisco Us Inc | Subtilisin variants having improved stability |
| WO2020242858A1 (en) | 2019-05-24 | 2020-12-03 | Danisco Us Inc | Subtilisin variants and methods of use |
| WO2020247582A1 (en) | 2019-06-06 | 2020-12-10 | Danisco Us Inc | Methods and compositions for cleaning |
| WO2021080948A2 (en) | 2019-10-24 | 2021-04-29 | Danisco Us Inc | Variant maltopentaose/maltohexaose-forming alpha-amylases |
| WO2022047149A1 (en) | 2020-08-27 | 2022-03-03 | Danisco Us Inc | Enzymes and enzyme compositions for cleaning |
| WO2022165107A1 (en) | 2021-01-29 | 2022-08-04 | Danisco Us Inc | Compositions for cleaning and methods related thereto |
| WO2023278297A1 (en) | 2021-06-30 | 2023-01-05 | Danisco Us Inc | Variant lipases and uses thereof |
| WO2023034486A2 (en) | 2021-09-03 | 2023-03-09 | Danisco Us Inc. | Laundry compositions for cleaning |
| WO2023114936A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023114939A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023114932A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Subtilisin variants and methods of use |
| WO2023114988A2 (en) | 2021-12-16 | 2023-06-22 | Danisco Us Inc. | Variant maltopentaose/maltohexaose-forming alpha-amylases |
| WO2023168234A1 (en) | 2022-03-01 | 2023-09-07 | Danisco Us Inc. | Enzymes and enzyme compositions for cleaning |
| WO2023250301A1 (en) | 2022-06-21 | 2023-12-28 | Danisco Us Inc. | Methods and compositions for cleaning comprising a polypeptide having thermolysin activity |
| WO2024050343A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Subtilisin variants and methods related thereto |
| WO2024050339A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Mannanase variants and methods of use |
| WO2024050346A1 (en) | 2022-09-02 | 2024-03-07 | Danisco Us Inc. | Detergent compositions and methods related thereto |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1231693A (en) | 1999-10-13 |
| AU4376497A (en) | 1998-04-17 |
| EP0939801A1 (en) | 1999-09-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP0939801A1 (en) | An enzyme with amylase activity | |
| EP2311941B1 (en) | Detergent composition comprising endo-glucanase | |
| EP1002060B1 (en) | Alkaline xyloglucanase | |
| US6268197B1 (en) | Xyloglucan-specific alkaline xyloglucanase from bacillus | |
| US9005950B2 (en) | Pectate lyase variants | |
| EP0882123B1 (en) | Process for removal or bleaching of soiling or stains from cellulosic fabric | |
| EP1002059B1 (en) | Endo-beta-1,4-glucanases from saccharothrix | |
| EP0912097B1 (en) | Antimicrobial peroxidase compositions | |
| US5837010A (en) | Detergent compositions containing a lipase variant at low levels | |
| WO1998008940A1 (en) | A novel endoglucanase | |
| US5997584A (en) | Method of treating polyester fabrics | |
| US5968883A (en) | Peroxidase variants | |
| WO1998008939A1 (en) | Novel lipolytic enzymes | |
| US20050181493A1 (en) | Mutant alpha-amylases | |
| US6165770A (en) | Alkaline stable amylase from Thermoalcalibacter | |
| WO2001079440A2 (en) | Extracellular expression of pectate lyase using bacillus or escherichia coli |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 97198262.7 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GE GH HU ID IL IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZW AM AZ BY KG KZ MD RU TJ TM |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH KE LS MW SD SZ UG ZW AT BE CH DE DK ES FI FR GB GR IE IT LU MC |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 1997941876 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09271783 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref document number: 1998515177 Country of ref document: JP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1997941876 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: CA |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1997941876 Country of ref document: EP |