WO1998001421A1 - pH SENSITIVE HYDROGELS WITH ADJUSTABLE SWELLING KINETICS FOR COLON-SPECIFIC DELIVERY OF PEPTIDES AND PROTEINS - Google Patents
pH SENSITIVE HYDROGELS WITH ADJUSTABLE SWELLING KINETICS FOR COLON-SPECIFIC DELIVERY OF PEPTIDES AND PROTEINS Download PDFInfo
- Publication number
- WO1998001421A1 WO1998001421A1 PCT/US1997/012305 US9712305W WO9801421A1 WO 1998001421 A1 WO1998001421 A1 WO 1998001421A1 US 9712305 W US9712305 W US 9712305W WO 9801421 A1 WO9801421 A1 WO 9801421A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mol
- group
- hydrogel
- carbon atoms
- member selected
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4891—Coated capsules; Multilayered drug free capsule shells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/30—Macromolecular organic or inorganic compounds, e.g. inorganic polyphosphates
- A61K47/32—Macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. carbomers, poly(meth)acrylates, or polyvinyl pyrrolidone
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C239/00—Compounds containing nitrogen-to-halogen bonds; Hydroxylamino compounds or ethers or esters thereof
- C07C239/08—Hydroxylamino compounds or their ethers or esters
- C07C239/22—Hydroxylamino compounds or their ethers or esters having oxygen atoms of hydroxylamino groups esterified
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F220/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and only one being terminated by only one carboxyl radical or a salt, anhydride ester, amide, imide or nitrile thereof
- C08F220/02—Monocarboxylic acids having less than ten carbon atoms; Derivatives thereof
- C08F220/52—Amides or imides
- C08F220/54—Amides, e.g. N,N-dimethylacrylamide or N-isopropylacrylamide
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/2004—Excipients; Inactive ingredients
- A61K9/2022—Organic macromolecular compounds
- A61K9/2027—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone, poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
- A61K9/28—Dragees; Coated pills or tablets, e.g. with film or compression coating
- A61K9/2806—Coating materials
- A61K9/2833—Organic macromolecular compounds
- A61K9/284—Organic macromolecular compounds obtained by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyvinyl pyrrolidone
- A61K9/2846—Poly(meth)acrylates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/70—Web, sheet or filament bases ; Films; Fibres of the matrix type containing drug
- A61K9/7007—Drug-containing films, membranes or sheets
Definitions
- This invention relates to compositions for oral delivery of peptides and proteins that are targeted for site-specific release to the colon. More particularly, this invention relates to pH sensitive hydrogels having adjustable swelling kinetics which make it possible to permit oral administration of the hydrogels which traverse the GI tract for site-specific release of peptides and proteins in the colon.
- Administration of drugs by injection is not suitable for ambulatory patients and is not generally acceptable to patients undergoing drug therapy for chronic diseases.
- This route is far from being an ideal route of administration of peptides and proteins in particular because of their short biological half-lives which necessitate repeated injections .
- proteolytic degradation caused both by endo- and exopeptidases which occurs at the site of delivery, i.e. the lumen of GI tract or within the absorbing tissues prior to reaching the systemic circulation.
- Peptides and proteins have high molecular weights and a number of ionizable groups .
- Yet another factor is the residence time of the drug delivery device at the absorption site which is often too short to allow therapeutic drug levels to be maintained for any length of time.
- a different factor is presystemic biotransformation in the liver, i.e. first pass metabolism in the liver.
- the pH fluctuation factor affects drug integrity, i.e. the high acidity of the stomach can preclude the stability of peptide and protein drugs.
- surfactants i.e. bile salts secreted from the gall bladder into the small intestine as a normal physiological process, can compromise the stability of peptides and proteins.
- the microenvironment at the site of absorption can modify the uptake of poorly absorbed drug molecules.
- the microenvironment changes temporally and spatially in various regions of the GI tract. The consequence of this instability is a wide variability in the bioavailability of drug molecules that are stable in the GI tract.
- Lymphatic absorption of peptide and protein drugs is an approach which has achieved some measure of success.
- lymphotropic drug carriers and their proper delivery to make them universally applicable to all types of peptides and proteins .
- Bioadhesive polymers have been investigated for the purpose of restricting the release of peptides and proteins to a region of the GI tract which favors their absorption. Further, bioadhesive polymers can also modify the residence time of peptide or protein drug delivery system at the absorption site in order to sustain the therapeutic levels for a sufficiently long period of time to provide adequate therapy.
- colloidal drug delivery systems have been considered: multiple emulsions (w/o/w) , liposomes and nanoparticles.
- the success of nanoparticulate colloidal drug delivery system depends on efficient penetration of the target cells.
- Absorption enhancers have been investigated. They appear to be indispensable in the oral delivery of proteins and peptides if the pharmacokinetics and pharmacodynamic requirements of the drugs are to be met .
- site-specific targeting of peptides and proteins to the colon have been investigated.
- the development of drug delivery systems which have the property of selective release of drug in the colon has received much attention.
- Two major therapeutic applications which can be found for oral colon-specific targeting are the treatment of local disorders in the colon and the delivery of peptide and protein drugs via the oral route.
- the colon may be the best site for peptide and protein delivery.
- the colon is considered a less hostile environment, i.e. more suitable as an absorption site for peptides and proteins, because of less diversity and intensity of digestive enzymes than the stomach and the small intestine. See, Longer, et al . , Comparison of the Activi ties of Rat Small Intestine and the Colon Brush Border Membrane Peptidases . Proceed, Intern. Symp . Control.
- polymeric prodrugs Kopecek et al, "N- (2-hydroxypropyl)methacrylamide Copolymers for Colon- specific Drug Delivery” in Oral Colon-specific Drug Delivery, Friend (ed) CRC Press, Bocca Raton, FL, USA, (1992)189-211)
- polymeric systems that could carry physically a variety of drugs to the colon.
- the use of polymeric systems for carrying drug to the colon is based on various mechanisms or physiological triggers .
- Time-released systems is another mode of polymeric delivery. These systems (Time-clock system, Geomatrix® system and Pulsincap®) are reviewed by Rubinstein, Approaches and Opportuni ties in Colon-specific Drug Delivery, Crit. Rev. Ther . Drug Carrier Syst. 12(1995)101-149. The development of time -released systems is based on the assumption that the transit time of solids in the small intestine usually does not exceed a particular value and that the small intestine transit time is not affected by gastric emptying patterns.
- the coating of dosage forms with pH-sensitive and bacterial degradable polymers and hydrogels is yet another system.
- One important characteristic of the large intestine is that it is the habitat of an enormous population of bacteria.
- the presence of microbial enzyme activities predominantly in the colon has been exploited for the design of colon-specific drug delivery systems.
- a major advantage of delivery systems based on colon-specific enzymes of bacterial origin is the site- specificity.
- the activities of the enzymes produced by microorganisms in the colon such as azoreductase and glycosidase activities do not overlap with the activities of enzymes in the upper GI tract.
- the azo reductase activity has been well studied and it has been used to release bioactive agents from water-soluble polymeric prodrugs as shown by Kopeckova, et al . , Release of 5-aminosalicylic Acid from Bioadhesive N- (2- hydroxypropyl ) methacrylamide Copolymers by Azoreductases in vi tro, Makromol . Chem. 191(1990)2037-2045. Further, Saffran et al . , A New Approach to the Oral Administration of Insulin and Other Peptide Drugs , Science 233(1986)1081-1084, synthesized copolymers containing azo aromatic crosslinks and succeeded in delivering insulin and vasopressin to the colon for absorption.
- hydrogels are based on biocompatible copolymers of N, N- dimethylacrylamide with t-butylacrylamide (to improve mechanical properties) , acrylic or methacrylic acid (to introduce pH - s ens i t ivi ty ) and 4,4'- di (methacryloylamino) azobenzene (a crosslinking agent which is degradable in the colon by azoreductase activity) .
- these hydrogels In the low pH range of the stomach, these hydrogels have a low equilibrium degree of swelling and the peptide or protein drug is protected against digestion by digestive enzymes. As the gels pass down the GI tract, swelling increases due to ionization of carboxylic acid groups in response to increase in pH.
- Hydrogels are shown composed of an ionizable monomer such as acrylic acid, a nonionizable monomer such as an acrylamide and the aromatic azobond containing cross-linking agent. Drug release is obtained by a combination of pH-dependent swelling and degradation of the hydrogel by enzymatic cleavage of the azobonds by azoreductases .
- Divinylic Crosslinking Agent N, O-dimethacryloyl Hydroxylamine , 1 Synthesis and Characteriza tion of Ra tes of Gel Degradation and Rate of Release of Model Drugs in vi tro and in vivo, J. Control. Rel. 24(1993)181-190.
- hydrolytically degradable hydrophilic gels made using N, O-dimethyacryloyl hydroxylamine as a cross-linking agent are taught in Ulbrich et al . , U.S. Patent 5,124,421. However, in neither of these is the use of such hydrogels for directing site-specific release of peptides or proteins to the colon taught or suggested.
- Patent 5,124,421 suggests that release of drugs can occur first in the environment with higher pH, such as the duodenum and intestine . Based on the reported research it is believed that to achieve a successful delivery of a peptide or protein drug to the colon, the drug should be transported intact through the stomach and the small intestine.
- the hydrogel systems reported above have a low equilibrium degree of swelling in the low pH of the stomach; hence drugs can be protected from enzymatic digestion and will not diffuse out of a delivery device. However, these hydrogels swell in the high pH of the small intestine. It is known that the transit time in the small intestine is from about 3 to 5 hours.
- a disadvantage of these hydrogel systems is that part of the peptide or protein drugs may diffuse out of an oral delivery device if the rate and equilibrium degree of swelling are too high during the residence time of the delivery device in the small intestine.
- Another object of this invention is to provide a hydrogel for the delivery of peptide and protein drugs into the colon wherein the kinetics of swelling of the hydrogel in the small intestine are chemically controlled by the chemical make up of the hydrogel.
- hydrogel copolymers formulated from monomers of N-substituted (meth) acrylamides, acrylic or methacrylic acid, a crosslinking agent containing an aromatic azo bond and a N, O-diacylhydroxylamine .
- hydrogel copolymers may be formed from monomers of N- substituted (meth) acrylamides, acrylic acid or methacrylic acid, a crosslinking agent containing an aromatic azo bond wherein ionizable -COOH groups on the polymer are reacted with N-acylhydroxylamines resulting in the formation of a hydrogel containing N,0- diacylhydroxylamine moieties.
- hydrogels are formed containing a crosslinked aromatic azo bond and a N, O-diacylhydroxylamine group or cross-linkage which is stable in an acid environment but which is labile and subject to hydrolysis at the pH encountered in the small and large intestines yielding ionizable - COOH groups.
- COOH COOH
- N,0- diacylhydroxylamine moieties N,0- diacylhydroxylamine moieties.
- the hydrogel contains labile N, O-diacylhydroxylamine moieties that are stable in acidic medium, such as encountered in the stomach (pH 1-4) but that are susceptible to hydrolysis above approximately pH 6.5.
- ionized -COOH groups attached to the polymer network ar «e generated.
- chemical control of kinetics of swelling can be coupled to physical control of kinetics of swelling by varying the concentration of the aromatic azo cross-linking agent, as it is known that increase in cross-linking density normally induces restriction to the expansion of polymer network.
- N, O-diacylhydroxylamine monomers have been synthesized and incorporated into an aromatic azo crosslinked hydrophilic acrylamide/acrylic acid hydrogel network.
- these N, O-diacylhydroxylamines function as either acid group protectants or cross-linking moieties which also protect an acid functionality at an acid pH but hydrolyze at higher pH ranges as found in the small intestine to expose free carboxylic acid groups .
- N, O-diacylhydroxylamines may be represented by the formula:
- Ac 2 is an acyl group of the structure -CO-R 12 .
- R 11 is an alkyl group of from about 1 to 24, and preferably 3 to 12, carbon atoms.
- ⁇ C0-R 12 is a member selected from the group consisting of acryloyl or methacryloyl .
- R n - CO shall be used interchangeably.
- these groups may be referred to herein specifically by the alkanoyl or carbonyl prefix, e.g. lauroyl, hexanoyl, or propionyl .
- the term AcNHOAc 2 will be used to define the N, O-diacylhydroxylamines where the Ac 2 group can contain from 2 to 25 carbon atoms.
- N, O-diacylhydroxylamines may also be prepared having the formula Ac 2 -NHO-Ac 2 where Ac 2 is as defined above.
- both the N and O acyl groups are -CO-R 12 .
- the compound is N,0- dimethacryloylhydroxylamine (MANHOMA) , and is disclosed in U.S. Patent 5,124,421 as a crosslinking agent. However, it is not disclosed as being used in conjunction with an aromatic azo containing crosslinking agent, e.g.
- R 11 is an alkyl group of from 1 to 24 carbon atoms it functions as a monomer which, when incorporated into a polymer strand is protective of carboxylic acid functionalities at low pH's, i.e. from 1-5 but which can, at a pH of 6.5 or above, be hydrolyzed to yield carboxylic acid groups.
- R X1 will contain from about 3 to 12 carbon atoms.
- MANHOMA forms a cross-link between adjacent polymer strands and serves as a protectant of a carboxylic acid functionality and also as a cross-linking agent.
- the synthesis of the substituted hydroxylamines consists of two steps: N-acylation of hydroxylamine hydrochloride to give the N-acylhydroxyl- hydroxylamine) and O-acylation of N-acyl -hydroxylamine to give N, O-diacylhydroxylamine.
- N, O-diacylhydroxylamines The chemistry of N, O-diacylhydroxylamines has features which favor their use as hydrolyzable monomers.
- the first acyl group in the acylation of hydroxylamine is bound to the nitrogen atom to form N- acylhydroxyamine .
- the acyl group is attached to the oxygen atom. This makes it possible to predetermine the acylating agents to be used for N-acylation and O-acylation such that, on hydrolysis, carboxylic acid groups attached to the polymer main chains are generated.
- the acid chloride or acyl ester can be used for N- acylation and the acyl moiety determines the leaving group during hydrolysis.
- Methacryoyl chloride was used for O-acylation to be able to generate carboxylic groups on the polymer chain during hydrolysis and hence control the kinetics of swelling of the hydrogel polymer into which they are incorporated.
- the acyl group attached to the oxygen is less firmly held than the acyl group which is attached to the nitrogen.
- the acylation reaction, as well as the subsequent hydrolysis reaction is not affected by steric hindrance within the molecule. The significance of this is that the sequence of hydrolytic degradation in N, O-diacylhydroxylamines favors the generation of carboxylic acid groups in the hydrogels as hereinafter defined.
- O-diacylhydroxylamines are acids due to the hydrogen of the imino (-NH-) group.
- N, O-diacylhydroxylamines makes them suitable as substances which can protect the carboxylic acid groups at pH above their pKa ' s (i.e. the neutral pH of the small intestine) and which can hydrolyze to control the kinetics of swelling.
- Example A N-Lauroyl, O-methacryloylhydroxylamine (LaNHOMA) N-Lauroylhydroxylamine
- N-Propionyl , O-methacryloylhydroxylamine The synthesis and purification were exactly as described for N-lauroyl, O-methacryloylhydroxylamine in
- Example A except that the crystallization was carried out in N-hexane : ethyl acetate.
- Example D N, O-Dimethacryloylhydroxylamine (MANHOMA)
- synthesis of MANHOMA is different from other N, O-diacylhydroxylamines in that it involves only one step, i.e., the simultaneous - and O-acylation of hydroxylamine hydrochloride.
- a 2.5 g sample of hydroxylamine hydrochloride was dissolved in 12.5 g of pyridine. The temperature was brought to 5°C an ice water bath. A small amount of t-octylpyrocatechol was added as an inhibitor. While stirring 8.9 g of methacryloyl chloride was added dropwise. The ice-water bath was removed and the reaction mixture was stirred at room temperature for 2 h.
- the reaction mixture was diluted with 25 mL chloroform.
- 4.2 mL of hydrochloric acid (0.0428 mole) was added dropwise.
- the organic layer was separated and washed 4 times with 20 mL water. It was then dried over magnesium sulphate. It was filtered and the filtrate was evaporated.
- To the oil residue obtained was added 50 mL of diethyl ether. The white precipitate formed was filtered and the filtrate evaporated. The oil residue was dried briefly using an oil pump. It was transferred into a freezer where it solidified.
- Hydrogels were prepared using N,0- diacylhydroxylamines of the formula (Ac 3 NHOAc 2 ) wherein Ac 3 is a member selected from the group consisting of Ac x or Ac 2 to prepare hydrogels having general formula: poly[ (AAA) v -co-(AA) w -co-(Ac 3 NHOAC 2 ) x -co-(BuAA) y -co-(AAB) z ] .
- AAA is an N-alkyl substituted (meth) acrylamide
- AA is a member selected from the group consisting of acrylic or methacrylic acid
- Ac 3 NHOAC 2 is as defined above
- BuAA is N-t-butylacrylamide
- AAB is a crosslinking agent containing an aromatic azo bond and v, w, x, y and z represent the mole percent or mole percent range of its corresponding group.
- AAA may be a member selected from N-substituted acrylamides, N,N-disubstituted acrylamides and N- substituted methacrylamides of the formula:
- R ⁇ is H or CH 3 ;
- R 2 is alkyl or hydroxyalkyl having 1 to 6 carbon atoms and R 3 is H or R 2 with the proviso that when R x is CH 3 , R 3 is H, and when R x is H, R 3 is R 2 .
- AAA may be present such that v represents a mole percent range of between about 20 to 90 mol% with ranges of between about 40 to 80 mol% being preferred.
- the N,N- disubstituted acrylamides and N, N-dimethylacrylamide in particular are preferred.
- BuAA represents N-t-butylacrylamide and is generically within the scope of AAA. BuAA is added to provide structural strength to the hydrogel and may be present such that y represents ranges of from 0 to 35 mol%. Preferably v will represent between about 5 to 20 mol%. Since BuAA is contained within the scope of the AAA definition, the mol% represented by the sum. of v and y will not exceed 90% and preferably will not exceed 80%.
- AA is an ionizable monomer and is a member selected from the group consisting of acrylic and methacrylic acid. AA may be present such that w represents a mol% range of between about 0.1 to 60% and is preferably between about 5 and 45 mol%. The lower ranges are more practical for forming a pH swellable hydrogel in that the N, O-diacylhydroxyl a ine monomer also provides a means of generating ionizable groups at higher pH ranges. Also, at higher ranges, some of the ionizable COOH groups can be protected by converting some or all of these functionalities to N, O-diacylamine moieties by reaction with N-acylhydroxylamines, e.g. ACjNOH. Ac 3 HNOAc 2 and may be present in mol% amount such that x is between about 1 and 50% with ranges of between about 5 and 30% being preferable.
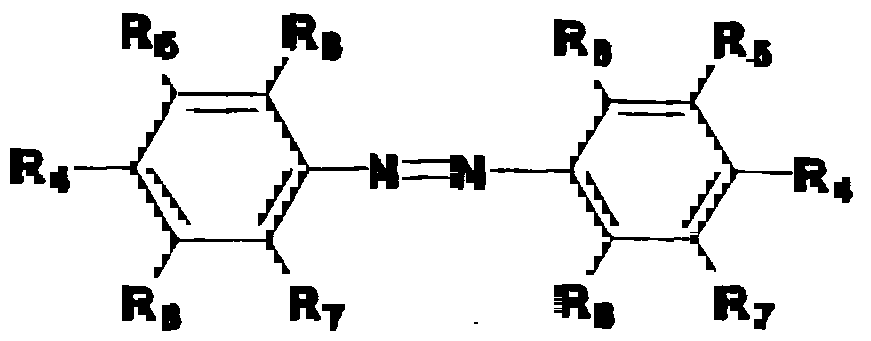
- AAB represents an aromatic azo benzene represented by the formula 5 Re Re R5 R ⁇ N _- N _ R
- R 4 is a member selected from the group consisting of
- R g is H or CH 3 and where R 5 , R 6 , R 7 and R 8 are individually selected from the group consisting of H, CH 3 , OCH 3 , Br, Cl, F, I, N0 2 , CN or C(0)CH 3 .
- AAB may be present in amount such that z represents a mole percent of between about 0.05 and 15.
- Preferred AAB groups are those selected from the group consisting of 4,4'- di (methacryloylamino) azobenzene, 4-4 ' -di (N-methacrylol- e -aminocaproy 1 ) aminoazobenzene and 4,4'- di (methacryloylamino) -3 , 3 ' , 5 , 5 ' -tetrachloroazobenzene with 4, 4 ' -di (methacryloylamino) -azobenzene being particularly preferred and is used in the following examples .
- AAA is N, N-dimethylacrylamide
- BuAA is t-butylacrylamide
- AA is acrylic acid (but could as readily also be methacrylic acid)
- AAB is 4,4'- di (methacryloylamino) azobenzene and the N,0- diacylhydroxylamine is as specifically designated in each table or example.
- the hydrogels were synthesized by radical copolymerization of the monomers in 70% v/v DMSO using AIBN as the initiator. The concentration of AIBN used in the reaction mixture was 36mM. The monomer solution was bubbled with nitrogen for 30 minutes.
- Tables 1-4 list hydrogels of Examples 1-31 wherein each monomer is represented in terms of mole percent.
- Table 1 Monomer feed compositions based on N- lauroyl , O-methacryloylhydroxylamine Example
- the hydrogel disc was transferred into a vial containing 15 L of 0.04 M phosphate buffer (pH 7.4) already equilibrated at 37 °C in a shaking water bath. Changes in weight were monitored at time intervals until equilibrium swelling was reached. The pH of swelling solution was checked. In all cases, the pH remained unchanged. The swelling of the hydrogels was expressed as swelling ratio (weight of wet gel/weight of dry gel) .
- Hydrogels have a high liquid content and are given form by the network of polymer strands made up of the formula poly [ (AAA) v -co- (AA) w -co- (Ac 3 NHOAC 2 ) x -co- (BuAA) y - co-(AAB) z . Liquid within the network is held by a balance of forces.
- the hydrophilicity of the hydrogel network is conferred by various functional groups such as -OH, -COOH, -CONH 2 , -CONHR, and -S0 3 H which are responsible for their ability to imbibe a large amount of water.
- the factors which may affect swelling of ionic hydrogels include the degree of ionization, the charge of the ionic monomer (s), the pKa of the ionizable group (s), the concentration of the ionizable monomer (s) in the network, the nature of the counterion, the nature of the swelling solution in terms of pH, ionic strength and composition (i.e. buffering) , the cross-linking density and the hydrophilicity/hydrophobicity of the polymer.
- Tables 5-8 illustrate the dependence of swelling isotherms of the hydrogels on the concentration of the Ac 3 NH0Ac 2 when the hydrogels are transferred abruptly from pH 2.0 to pH 7.4 at 37 °C.
- a biphasic swelling pattern is observed for all hydrogels reported.
- the structures of the polymer backbone, which are determined by the proportions of acrylic acid, (AA W ) , and hydrolyzable monomer, (Ac 3 NHOAc 2 ) x greatly influence the kinetics of swelling and swelling equilibrium. Further, the nature of the Ac 3 NHOAc 2 monomer also affects the swelling kinetics and equilibrium swelling.
- Formulas 1-5 show representative structures which correlate with data in Tables 5-8.
- Formula 1 shows a typical structure of a poly [ (AAA) v -co- (AA) w -co- (BuAA) y -co- (AAB) ,] hydrogel as shown in Example 1 which would be the same at either pH 2.0 or 7.4.
- Formula 2 is representative of a poly [AAA v -co- (AA) w - co (Ac 1 NHOAc 2 ) x -co- (BuAA) y -co- (AAB) hydrogel as shown in Examples 2-25 at pH 2.0 showing the presence of the hydrolyzable ACiNHOAc 2 monomer.
- Formula 3 is representative of a polytAAA ⁇ -co- (AA) W - co (ACNHOACjJ x -co- (BuAA) y -co- (AAB) Z ] hydrogel of Examples 2-25 at pH 7.4 showing the hydrolysis of the AC t NHOACj monomer to provide a -COOH functionality.
- Formula 4 is representative of a polyfAAA v -co- (AA) W - co (MANHOMA) x -co- (BuAA)y-co- (AAB),] hydrogel as shown in Examples 28-31 at pH 2.0 showing the presence of the cross-linked hydrolyzable MANHOMA monomer.
- Formula 5 is representative of a poly[AAA v -co- (AA) w - co (MANHOMA) x -co- (BuAA) y-co- (AAB),] hydrogel of Examples 26-31 at pH 7.4 showing the hydrolysis of the crosslinked MANHOMA monomer to provide -COOH and -NH 2 functionalities .
- Ac 3 NHOAc 2 i.e. the structure of Formula 1 wherein only acrylic acid is present as an ionizable monomer at pH 7.4 reached equilibrium swelling very rapidly and that the equilibrium swelling ratio remained constant. This is attributable to the ionization of acrylic acid in the polymer network.
- the carboxylic acid group of the acrylic acid is reported to have a pKa of about 4.3. Since the pH of 7.4, i.e. the swelling medium to which the gel was transferred after swelling at pH 2.0 is higher than the pKa of the carboxylic acid groups, ionization occurs and is accompanied by an increase in charge density, charge hydration, electrostatic repulsion between adjacent carboxylate groups and expansion of the polymer network. These, together with increased hydrophilicity of the polymer network, results in the ability of the polymer to imbibe water.
- Tables 5-8 show that the magnitude of the initial swelling phase, i.e. the burst effect, decreases with the decrease in the concentration of the acrylic acid (AA W ) content and the accompanying increase in the concentration of the Ac 3 NHOAc 2 content .
- This pattern can be ascribed to the initial decrease, prior to hydrolysis relative to Example 1, in the ionic content of the hydrogel network as the concentration of the Ac 3 NHOAc 2 monomer increases. The effect is to decrease the charge density, the hydrophilicity and the expansion of the polymer network.
- Tables 5-8 also show that the second phase of the swelling isotherms of hydrogels containing varying concentrations of Ac 3 NHOAc 2 approach equilibrium swelling gradually. This is thought to be due to the fact that the pKa values of the Ac 3 NHOAc 2 monomers containing the hydrolytic linkage are around 4.5 to 4.85, which is lower than the 7.4 pH of the swelling medium as would be encountered in the small intestine.
- the hydrolyzable moieties in the polymer exist mainly as monoanions at a pH of 7.4.
- the equilibrium swelling ration is expected to be the same in all of the hydrogels after equilibrium swelling at pH 7.4 because the data reported resulting from Examples 1-31 was designed to make the acrylic acid concentration constant following hydrolysis.
- the combined concentration of AA and Ac 3 NHOAc 2 in each example is 40% so the number of carboxylic acid groups following hydrolysis should be the same if hydrolysis proceed to completion.
- Table 5-8 that there are variations in the degree of swelling. However, for whatever reasons there are variations, this does not depart from the showing that there is a reproducible swelling produced upon raising the pH from 2.0 to 7.4 and that is shown by an initial swelling burst followed by a slower degree until equilibrium is reached. This allows one to formulate a hydrogel by varying the selecting the desired Ac 3 NHOAc 2 and then varying the concentration of that Ac 3 NHOAc 2 to obtain the swelling kinetic that are desired.
- Table 8 shows that the equilibrium swelling ratios of hydrogels containing MANHOMA (Examples 26-28) are below those of Example 1 .
- the structure of the hydrogel after swelling as shown in Formula 5, contains carboxylic acid groups and free amino groups. Ionic interaction between the two groups will have some effects on the concentration of the counterions in these hydrogels and therefore will restrict polymer chain expansion and swelling. This further shows that there is some difference in the functionality of the MANHOMA monomer of the prior art and the Ac.NHOAc- monomers synthesized as novel compounds herein.
- the concentration of aromatic azo crosslinking agent influences the degree of swelling.
- the burst effect described above and the equilibrium swelling ratio decrease with increase in the concentration of the AAB crosslinking agent.
- the equilibrium swelling of the hydrogels reflects a balance between swelling forces and the network elastic retractive forces. This balance depends on the crosslinking density of the gel network. Thus as the degree of crosslinking increases, the equilibrium degree of swelling is restricted.
- the combination of the chemical control of the pH sensitive hydrogels attributable to the labile N,0- diacylhydroxylamines and the physical control attributable to the AAB crosslinking makes it possible to control the swelling of the hydrogel in the small intestine such that the equilibrium degree of swelling is reached at about the time of entry of the hydrogel into the colon where biodegradation can be facilitated by azoreductase activity.
- the hydrogels of this invention can be utilized in a variety of ways to allow the oral delivery of a dosage form of peptide and protein drugs to the colon.
- the drugs that are advantageously administered via the dosage forms of the invention are drugs that would be destroyed by the environments of the stomach/small intestine, have an irritating or other undesirable effect on the tissues of the upper gastrointestinal tract, or are intended to locally treat conditions within the colon or colonic tissue.
- Systemically acting drugs that are delivered via these dosage forms must be capable of being absorbed into circulation through the mucosal lining of the colon.
- Examples of drugs that may be delivered advantageously via this invention are peptide or protein drugs such as insulin, calcitonin, angiotensin, cyclosporin, vasopressin, and somatostatin.
- hydrogels can function as coatings of various dosage form devices such as capsules, caplets, tablets and the like which can pass intact through the acid media of the stomach into the small intestine where the swelling process begins resulting from the rapid rise in pH to about 7.4.
- the swelling will have reached equilibrium and the azoreductase enzyme, found primarily in the colon, will allow cleavage of the AAB linkage further degrading the polymer coating releasing the peptide or protein drug to the colon interior.
- the coating process can be carried out by in si tu coating of the device such that the copolymerization and crosslinking occur around the device surface.
- a latex-type suspension of the hydrogel e.g., a suspension of fine particles in a swelling agent/dispersant
- the device dipped or immersed in the suspension.
- the drugs may be enclosed in a film or membrane of the copolymer by forming the copolymer hydrogel into a film or membrane, placing a desired amount of drug between two pieces of the film/membrane and sealing the edges of the pieces . In this manner a container whose walls are made of the copolymer and whose lumen contains a unit dose of drug are prepared.
- the drugs may be also be formulated with the hydrogels by dispersing them in the hydrogels. Dispersions are made by mixing the drug with the solution of monomers and then forming the hydrogel by crosslinking in the manner as described above. Alternatively a body of the hydrogel may be soaked in a solution or dispersion of the drug which partially swells the hydrogel, without hydrolysis of the N,0- diacylhydroxylamine moieties, and the body then dried. The drug-hydrogel dispersions may be cut or otherwise shaped into a body containing the desired unit dose of drug. The amount of drug administered may be varied by varying the concentration of drug in the solution or dispersion, the solvent used to create the solution or dispersion, and the volume of the drug-hydrogel body.
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Polymers & Plastics (AREA)
- Inorganic Chemistry (AREA)
- Medicinal Preparation (AREA)
Abstract
A hydrogel polymeric system for the site specific delivery of peptide and protein drugs to the colon is prepared. The hydrogel protects the drug through the acid environment of the stomach, swells at a chemically controlled rate in the higher pH environment of the small intestine and is enzymatically degraded by azoreductases in the colon. pH sensitive hydrogels are formulated from monomers of N-substituted (meth)acrylamides, acrylic or methacrylic acid, a cross-linking agent containing an aromatic azo bond and an N,O-diacylhydroxylamine. Chemical control of swelling is attained by initially providing labile N,O-diacylhydroxylamine moieties in the hydrogel that are stable in the acidic medium of the stomach but that are susceptible to hydrolysis at the intestinal pH, i.e. above about pH 6.5. Upon hydrolysis, ionized -COOH groups attached to the polymer network are generated and swelling is maximized as the hydrogel reaches the colon where degradation occurs by the azoreductase cleavage of the aromatic azo cross-linking agent.
Description
pH SENSITIVE HYDROGELS WITH ADJUSTABLE SWELLING KINETICS FOR COLON-SPECIFIC DELIVERY OF PEPTIDES AND
PROTEINS
Field of the Invention:
This invention relates to compositions for oral delivery of peptides and proteins that are targeted for site-specific release to the colon. More particularly, this invention relates to pH sensitive hydrogels having adjustable swelling kinetics which make it possible to permit oral administration of the hydrogels which traverse the GI tract for site-specific release of peptides and proteins in the colon.
Background of the Invention and State of the Art:
There is considerable interest in the development of drug delivery devices for peptides and proteins based on two primary factors. First there is an increased understanding of the role of peptides and proteins in physiology and therapy. Secondly, the advent of recombinant DNA techniques which has facilitated the economical production of peptide and protein drugs . Many aspects of biopharmaceutical process development have been well investigated and difficulties in fermentation, cell culture, isolation and purification have been largely overcome for the production of peptide and protein drugs. Cleland, et al . , "Formulation and Delivery of Proteins and Peptides : Design and Development Strategies " in Formulation and Delivery of Proteins and Peptides, ACS Symposium Series 567, 1994 pp. 1-19. The biotechnology industry is now capable of producing many potential therapeutic peptides and proteins in commercial quantities. In 1989 over 150 peptide or protein drugs were undergoing clinical trials and that figure continues to rise.
Various potential routes for the delivery of peptide and protein drugs have been considered: invasive (e.g. direct injection: intravenous, subcutaneous, intramuscular and depot systems) and non- invasive (e.g. pulmonary, oral, nasal, buccal, ocular, rectal, vaginal and transdermal) . Only the direct injection and pulmonary route have been approved by U.S. regulatory agencies and neither of these is a popular route of drug administration. Nasal administration of calcitonin has been approved in Europe. Administration of drugs by injection is not suitable for ambulatory patients and is not generally acceptable to patients undergoing drug therapy for chronic diseases. This route is far from being an ideal route of administration of peptides and proteins in particular because of their short biological half-lives which necessitate repeated injections .
Aside from being a non-invasive route of drug administration, the advantages of oral drug delivery are many and well known. For chronic therapies, in virtually all therapeutic areas, the most desirable route of drug administration is the oral route. Further, oral delivery of peptides and proteins is safe and commercializable, with an enormous market potential. Moreover, oral drug delivery can assure patient compliance .
However, the delivery of peptide and protein drugs using the oral route is fraught with difficulties which center around low bioavailability. Some of the factors responsible for low bioavailability of peptide and protein drugs that have been identified are as follows.
One factor is proteolytic degradation caused both by endo- and exopeptidases which occurs at the site of delivery, i.e. the lumen of GI tract or within the
absorbing tissues prior to reaching the systemic circulation.
Another factor is low permeability of the absorbing tissues to peptides and proteins. This problem is a consequence of the size, hydrophilicity and charge characteristics of the drugs. Peptides and proteins have high molecular weights and a number of ionizable groups .
Yet another factor is the residence time of the drug delivery device at the absorption site which is often too short to allow therapeutic drug levels to be maintained for any length of time.
A different factor is presystemic biotransformation in the liver, i.e. first pass metabolism in the liver. The pH fluctuation factor affects drug integrity, i.e. the high acidity of the stomach can preclude the stability of peptide and protein drugs. Further, surfactants, i.e. bile salts secreted from the gall bladder into the small intestine as a normal physiological process, can compromise the stability of peptides and proteins.
The microenvironment at the site of absorption can modify the uptake of poorly absorbed drug molecules. The microenvironment changes temporally and spatially in various regions of the GI tract. The consequence of this instability is a wide variability in the bioavailability of drug molecules that are stable in the GI tract.
Most of the problems of peptide and protein drug absorption are particularly notorious in the upper GI tract. As a consequence, research efforts have been geared towards stabilizing formulations to facilitate the oral delivery of peptides and proteins. Woodley, Enzymatic Barriers for GI Peptide and Protein Delivery. Crit. Rev. Ther . Drug Carrier Systems 11, (1994) 61-95
and Lee, et al . , Oral Route of Peptide and Protein Delivery" in Peptide and Protein Delivery, Ed. Marcel Dekker,N.Y. 1991, pp 691-738.
Chemical modifications of peptide and protein drugs to improve enzymatic stability and membrane penetration have been studied. This approach, though it has met with some measure of success, is not without its drawbacks such as the partial loss of biological activity. Protease inhibitors have been used. This approach has not been very promising due to the fact that the enzymes to be inhibited are many and act synerςristically coupled with the problem of biological adaptation. Feedback mechanisms often cause compensatory increase in the production of the enzymes in the presence of protease inhibitors.
Lymphatic absorption of peptide and protein drugs is an approach which has achieved some measure of success. However, there are problems of appropriate lymphotropic drug carriers and their proper delivery to make them universally applicable to all types of peptides and proteins .
Bioadhesive polymers have been investigated for the purpose of restricting the release of peptides and proteins to a region of the GI tract which favors their absorption. Further, bioadhesive polymers can also modify the residence time of peptide or protein drug delivery system at the absorption site in order to sustain the therapeutic levels for a sufficiently long period of time to provide adequate therapy.
Colloidal drug delivery systems have been considered: multiple emulsions (w/o/w) , liposomes and nanoparticles. The success of nanoparticulate colloidal drug delivery system depends on efficient penetration of the target cells.
Absorption enhancers have been investigated. They appear to be indispensable in the oral delivery of proteins and peptides if the pharmacokinetics and pharmacodynamic requirements of the drugs are to be met . Lastly, site-specific targeting of peptides and proteins to the colon have been investigated.
The development of drug delivery systems which have the property of selective release of drug in the colon has received much attention. Two major therapeutic applications which can be found for oral colon-specific targeting are the treatment of local disorders in the colon and the delivery of peptide and protein drugs via the oral route. It is believed by some that the colon may be the best site for peptide and protein delivery. For example, the colon is considered a less hostile environment, i.e. more suitable as an absorption site for peptides and proteins, because of less diversity and intensity of digestive enzymes than the stomach and the small intestine. See, Longer, et al . , Comparison of the Activi ties of Rat Small Intestine and the Colon Brush Border Membrane Peptidases . Proceed, Intern. Symp . Control. Rel . Bioact. Mater. 16 (1991)225-226 and Kopecek, et al., Polymers for Colon-specific Drug Delivery, Fifth Int. Symp. on Recent Adv. in Drug Deliv. Systems, Abstracts, (1991)27-28. Also, the colon has a long retention time and is highly responsive to absorption enhancers. See, Taniguchi, et al . , Enhanced Intestinal Permeabili ty to Macromolecules, II : Improvement of the Large Intestinal Absorption of Heparin by Lipid- surfactant Mixed Micelles in Rat . Int. J. Phar . 4 (1980) 219-228.
Attempts to deliver drugs specifically to the colon have involved the use of prodrugs, (Friend et al . , Drug Glycosides : Potential Prodrugs for Colon-specific Drug
Delivery, J . Med. Chem. 28(1985)51-57 and Tozer, et al . , Colon-specific Delivery of Dexamethasone from a Glucoside Prodrug in the Guinea Pig, Pharm. Res. 8(1991)445-454), polymeric prodrugs (Kopecek et al, "N- (2-hydroxypropyl)methacrylamide Copolymers for Colon- specific Drug Delivery" in Oral Colon-specific Drug Delivery, Friend (ed) CRC Press, Bocca Raton, FL, USA, (1992)189-211) , and polymeric systems that could carry physically a variety of drugs to the colon. The use of polymeric systems for carrying drug to the colon is based on various mechanisms or physiological triggers .
One system involves the coating of drugs or dosage forms with pH-sensitive polymers. Mooter, et al . , Oral Colon-specific Drug Delivery: A Review, Drug Delivery 2(1995)81-82, review this technique and indicate that one of the disadvantages of polymers based on pH- sensitivity alone is the vagaries of the site where polymer disintegration begins and ends. Depending on the intensity of GI tract mobility, the disintegration of the polymer can be completed deep in the colon or at the end of the ileum. The presence of bile acids, carbon dioxide and short-chain fatty acids in the GI tract can reduce the pH of the colon. This factor may be a problem where pH change is the only mechanism that triggers polymer disintegration.
Time-released systems is another mode of polymeric delivery. These systems (Time-clock system, Geomatrix® system and Pulsincap®) are reviewed by Rubinstein, Approaches and Opportuni ties in Colon-specific Drug Delivery, Crit. Rev. Ther . Drug Carrier Syst. 12(1995)101-149. The development of time -released systems is based on the assumption that the transit time of solids in the small intestine usually does not exceed
a particular value and that the small intestine transit time is not affected by gastric emptying patterns.
The coating of dosage forms with pH-sensitive and bacterial degradable polymers and hydrogels is yet another system. One important characteristic of the large intestine is that it is the habitat of an enormous population of bacteria. The presence of microbial enzyme activities predominantly in the colon has been exploited for the design of colon-specific drug delivery systems. A major advantage of delivery systems based on colon-specific enzymes of bacterial origin is the site- specificity. The activities of the enzymes produced by microorganisms in the colon such as azoreductase and glycosidase activities do not overlap with the activities of enzymes in the upper GI tract. The azo reductase activity has been well studied and it has been used to release bioactive agents from water-soluble polymeric prodrugs as shown by Kopeckova, et al . , Release of 5-aminosalicylic Acid from Bioadhesive N- (2- hydroxypropyl ) methacrylamide Copolymers by Azoreductases in vi tro, Makromol . Chem. 191(1990)2037-2045. Further, Saffran et al . , A New Approach to the Oral Administration of Insulin and Other Peptide Drugs , Science 233(1986)1081-1084, synthesized copolymers containing azo aromatic crosslinks and succeeded in delivering insulin and vasopressin to the colon for absorption.
The successful oral delivery of peptide and protein drugs via the colon requires technologies, that both protect these drugs from proteolytic breakdown and enhance their penetration through the intestinal epithelium. The systems referenced above, while certainly advances in the art, do not entirely meet these criteria.
However, various approaches have been investigated to overcome these barriers to oral delivery of peptides and proteins. The synthesis, structure and properties of a series of tailor-made, biodegradable, pH-sensitive hydrogels containing acidic comonomers and enzymatically degradable cross-linking agents suitable for colon- specific drug delivery have been studied. See Brondsted, et al, Hydrogels for Si te-specific Oral Drug Delivery: Synthesis and Characterization, Biomaterials 12(1991)584-592; Pradny, et al . , Hydrogels for Si te- specific Oral Delivery; Poly (acrylic acid-co-butyl a c r y l a t e ) c r o s s - l i n k e d w i t h 4 , 4 ' - bis (methacryloylamino) azobenzene, Makromol . Chem.
191(1990)1887-1897; and Yeh, et al, Biodegradable and pH-Sensi tive Hydrogels : Synthesis by Crosslinking of N, N-dimethylacrylamide Copolymer Precursors, J.Polym. Sch: Part A: Polym. Chem. 32(1994)1627-1637. These hydrogels are based on biocompatible copolymers of N, N- dimethylacrylamide with t-butylacrylamide (to improve mechanical properties) , acrylic or methacrylic acid (to introduce pH - s ens i t ivi ty ) and 4,4'- di (methacryloylamino) azobenzene (a crosslinking agent which is degradable in the colon by azoreductase activity) . In the low pH range of the stomach, these hydrogels have a low equilibrium degree of swelling and the peptide or protein drug is protected against digestion by digestive enzymes. As the gels pass down the GI tract, swelling increases due to ionization of carboxylic acid groups in response to increase in pH. Upon arrival in the colon, a degree of swelling is reached that makes the cross-links susceptible to azoreductase activity. The gels are degraded and the drug is released. Brondsted, et al . , Hydrogels for Si te-specific Drug Delivery to the Colon : in vi tro and
in vivo Degrada tion, Pharm. Res. 9(1992)1540-1545 and Yeh, et al . , Degradabili ty of Hydrogels Con taining Azoaromatic Cross-links, Macromol . Chem. Phys . 196(1995)2138-2202 report biodegradation studies on the hydrogels that are consistent with the transit time through the colon. Much of this research is described and claimed in Kopecek et al . , U.S. Patent 5,415,864, issued May 16, 1995 which is incorporated herein by reference. Hydrogels are shown composed of an ionizable monomer such as acrylic acid, a nonionizable monomer such as an acrylamide and the aromatic azobond containing cross-linking agent. Drug release is obtained by a combination of pH-dependent swelling and degradation of the hydrogel by enzymatic cleavage of the azobonds by azoreductases .
Hydrophilic gels based on copolymers of N-(2-' hydroxypropyl ) methacrylamide (HPMA) and N,0- dimethacryloylhydroxylamine which contain a hydrolytically labile bond have been studied by Ulbrich et al . , Novel Biodegradable Hydrogels Prepared Using
Divinylic Crosslinking Agent N, O-dimethacryloyl Hydroxylamine , 1 : Synthesis and Characteriza tion of Ra tes of Gel Degradation and Rate of Release of Model Drugs in vi tro and in vivo, J. Control. Rel. 24(1993)181-190. Also, hydrolytically degradable hydrophilic gels made using N, O-dimethyacryloyl hydroxylamine as a cross-linking agent are taught in Ulbrich et al . , U.S. Patent 5,124,421. However, in neither of these is the use of such hydrogels for directing site-specific release of peptides or proteins to the colon taught or suggested. U.S. Patent 5,124,421 suggests that release of drugs can occur first in the environment with higher pH, such as the duodenum and intestine .
Based on the reported research it is believed that to achieve a successful delivery of a peptide or protein drug to the colon, the drug should be transported intact through the stomach and the small intestine. The hydrogel systems reported above have a low equilibrium degree of swelling in the low pH of the stomach; hence drugs can be protected from enzymatic digestion and will not diffuse out of a delivery device. However, these hydrogels swell in the high pH of the small intestine. It is known that the transit time in the small intestine is from about 3 to 5 hours. Thus, a disadvantage of these hydrogel systems is that part of the peptide or protein drugs may diffuse out of an oral delivery device if the rate and equilibrium degree of swelling are too high during the residence time of the delivery device in the small intestine. However, to facilitate biodegradation of these hydrogels upon arrival in the colon, it is important to have a high equilibrium degree of swelling. To reach a compromise between these opposing situations and to further increase the capability or potential of these hydrogels in oral delivery of peptide and protein drugs is the focus of the present invention.
Objects and Summary of the Invention
It is an object of the present invention to provide a hydrogel polymeric system for the delivery of peptide and protein drugs which protects the drug through a acid environment of the stomach, swells at a controlled rate in the higher pH environment of the small intestine and is enzymatically degraded in the colon thereby releasing the drug into the colon.
Another object of this invention is to provide a hydrogel for the delivery of peptide and protein drugs
into the colon wherein the kinetics of swelling of the hydrogel in the small intestine are chemically controlled by the chemical make up of the hydrogel.
These and other objects may be accomplished by means of hydrogel copolymers formulated from monomers of N-substituted (meth) acrylamides, acrylic or methacrylic acid, a crosslinking agent containing an aromatic azo bond and a N, O-diacylhydroxylamine . In the alternative, hydrogel copolymers may be formed from monomers of N- substituted (meth) acrylamides, acrylic acid or methacrylic acid, a crosslinking agent containing an aromatic azo bond wherein ionizable -COOH groups on the polymer are reacted with N-acylhydroxylamines resulting in the formation of a hydrogel containing N,0- diacylhydroxylamine moieties. In either event hydrogels are formed containing a crosslinked aromatic azo bond and a N, O-diacylhydroxylamine group or cross-linkage which is stable in an acid environment but which is labile and subject to hydrolysis at the pH encountered in the small and large intestines yielding ionizable - COOH groups.
It is known from previous studies that pH- sensitivity and swelling of the copolymer hydrogels of acrylamides and acrylic acid crosslinked by aromatic azo bonds are controlled mainly by the acrylic acid monomer, i.e. the presence of ionizable carboxylic acid groups. It has now been found that to chemically control the kinetics of swelling at the pH encountered in the intestinal tract, a hydrogel copolymer can be synthesized wherein part of the carboxylic groups (-
COOH) it is desired to have present for maximum swelling in the intestine can initially be present as N,0- diacylhydroxylamine moieties. In this configuration the hydrogel contains labile N, O-diacylhydroxylamine moieties that are stable in acidic medium, such as
encountered in the stomach (pH 1-4) but that are susceptible to hydrolysis above approximately pH 6.5. Upon hydrolysis, ionized -COOH groups attached to the polymer network ar«e generated. In this manner, chemical control of kinetics of swelling can be coupled to physical control of kinetics of swelling by varying the concentration of the aromatic azo cross-linking agent, as it is known that increase in cross-linking density normally induces restriction to the expansion of polymer network.
To this end, a series of N, O-diacylhydroxylamine monomers have been synthesized and incorporated into an aromatic azo crosslinked hydrophilic acrylamide/acrylic acid hydrogel network. Depending upon the structure, these N, O-diacylhydroxylamines function as either acid group protectants or cross-linking moieties which also protect an acid functionality at an acid pH but hydrolyze at higher pH ranges as found in the small intestine to expose free carboxylic acid groups .
Detailed Description of a Preferred Embodiment
The N, O-diacylhydroxylamines may be represented by the formula:
AC-NHO-ACz where Acλ is an acyl group of the structure Rlx-C0, and
Ac2 is an acyl group of the structure -CO-R12. R11 is an alkyl group of from about 1 to 24, and preferably 3 to 12, carbon atoms. R12 is a member selected from the group consisting of C=CH(CH3), CH2=CH. In other words ~C0-R12 is a member selected from the group consisting of acryloyl or methacryloyl . For purposes of convenience Ac, and Rn- CO shall be used interchangeably. For simplification these groups may be referred to herein specifically by the alkanoyl or carbonyl prefix, e.g. lauroyl, hexanoyl, or propionyl . Hence, generically, the term AcNHOAc2 will
be used to define the N, O-diacylhydroxylamines where the Ac2 group can contain from 2 to 25 carbon atoms.
N, O-diacylhydroxylamines may also be prepared having the formula Ac2-NHO-Ac2 where Ac2 is as defined above. In other words, both the N and O acyl groups are -CO-R12. When R12 is C=CH(CH3), the compound is N,0- dimethacryloylhydroxylamine (MANHOMA) , and is disclosed in U.S. Patent 5,124,421 as a crosslinking agent. However, it is not disclosed as being used in conjunction with an aromatic azo containing crosslinking agent, e.g. 4, 4 ' -di (methacryloylamino) azobenzene, such that there are two cross-linking agents in the same hydrogel one of which is hydrolyzable and the other of which is enzymatically degradable in the colon. The synthesis of Ac2NHOAc2 type N,0- diacylhydroxylamines, e.g. MANHOMA, is different from other N, O-diacylhydroxylamines in that only a single acylation step is required. The
hydroxylamines must be synthesized by a two step process, i.e. N- acylation followed by a O-acylation. When R11 is an alkyl group of from 1 to 24 carbon atoms it functions as a monomer which, when incorporated into a polymer strand is protective of carboxylic acid functionalities at low pH's, i.e. from 1-5 but which can, at a pH of 6.5 or above, be hydrolyzed to yield carboxylic acid groups.
Preferably RX1 will contain from about 3 to 12 carbon atoms. MANHOMA, on the other hand, forms a cross-link between adjacent polymer strands and serves as a protectant of a carboxylic acid functionality and also as a cross-linking agent.
Synthesis of N,0-Diacyl Hydroxylamines
In general, the synthesis of the substituted hydroxylamines consists of two steps: N-acylation of hydroxylamine hydrochloride to give the N-acylhydroxyl-
hydroxylamine) and O-acylation of N-acyl -hydroxylamine to give N, O-diacylhydroxylamine.
The chemistry of N, O-diacylhydroxylamines has features which favor their use as hydrolyzable monomers. The first acyl group in the acylation of hydroxylamine is bound to the nitrogen atom to form N- acylhydroxyamine . When N-acylhydroxylamines are acylated, the acyl group is attached to the oxygen atom. This makes it possible to predetermine the acylating agents to be used for N-acylation and O-acylation such that, on hydrolysis, carboxylic acid groups attached to the polymer main chains are generated.
The acid chloride or acyl ester can be used for N- acylation and the acyl moiety determines the leaving group during hydrolysis. Methacryoyl chloride was used for O-acylation to be able to generate carboxylic groups on the polymer chain during hydrolysis and hence control the kinetics of swelling of the hydrogel polymer into which they are incorporated. The acyl group attached to the oxygen is less firmly held than the acyl group which is attached to the nitrogen. The acylation reaction, as well as the subsequent hydrolysis reaction, is not affected by steric hindrance within the molecule. The significance of this is that the sequence of hydrolytic degradation in N, O-diacylhydroxylamines favors the generation of carboxylic acid groups in the hydrogels as hereinafter defined.
N, O-diacylhydroxylamines are acids due to the hydrogen of the imino (-NH-) group. The acidic nature of
N, O-diacylhydroxylamines makes them suitable as substances which can protect the carboxylic acid groups at pH above their pKa ' s (i.e. the neutral pH of the small intestine) and which can hydrolyze to control the kinetics of swelling.
Example A N-Lauroyl, O-methacryloylhydroxylamine (LaNHOMA) N-Lauroylhydroxylamine
To 13.9 g of a cooled (5°C) mixture of hydroxylamine hydrochloride and 150 mL chloroform was added 20.2 g of triethylamine with stirring. 21.9 g of Lauroyl chloride and 10.1 g of triethylamine were slowly added simultaneously while stirring the mixture. The reaction mixture was maintained at 5°C for 4 h and then left at room temperature for 16 h with stirring. The product was filtered and dried. The dried product was diluted with 300 mL diethyl ether, extracted once with water containing a small amount of hydroxylamine hydrochloride, extracted twice with pH 2 HCl /water, and three times with water. The product was dried and crystallized from ethyl acetate. Yield: 14.61 g (68%);- mp: 94-96°C; N-Lauroylhydroxylamine. Elemental analysis: Calculated C-66.93%; H-ll.70%, N-6.51%; Found C-66.78%, H-ll.69%, N-6.56%.
N-Lauroyl , O-methacryloylhydroxylamine
To a solution of 3.069 g of N-lauroylhydroxylamine, as prepared above, in 15 mL of pyridine containing a small amount of inhibitor ( tert . -octylpyrocatechol ) , 1.49 g of methacryloyl chloride dissolved in 3.75 mL diethyl ether was added dropwise with stirring at 5°C. The reaction mixture was left at room temperature for 4 h. The product was diluted with 40 mL diethyl ether and filtered. The product was dried under vacuum to give a solid residue. The solid residue was dissolved in 100 mL diethyl ether and filtered. The filtrate was dried over magnesium sulphate. It was filtered, evaporated and crystallized from ethanol :water . The crystals were washed with cold ethanol :water (3:4), cold 1% acetic acid, cold water and finally with cold 1:5 ethanol :water
to yield 2.11 g, (52%) N-Lauroyl, 0- ethacryloylhydroxylamine; mp 55-56.8°C. Elemental analysis: Calculated C-67.81%, H-10.31%, N-4.94%: Found C-67.99%, H-10.40%, N-4.86%.
Example B N-Hexanoyl, O-methacryloylhydroxylamine (HeNHOMA) N-Hexanoylhydroxylamine
To a cooled (5°C) , 13.9 g mixture of hydroxylamine hydrochloride and 150 mL chloroform was added 20.2 g of triethylamine with stirring. 13.5 g of Hexanoyl chloride and 10.1 g of triethylamine were slowly added simultaneously while stirring the mixture. The reaction mixture was maintained at 5°C for 4 h and then left at room temperature for 16 h with stirring. The isolation and crystallization procedures were as described above for N-lauroylhydroxylamine. Yield: 3.95 g (30%); mp : 65-66 °C. Elemental analysis: Calculated C-54.93%, H- 10.01%, N-10.68%: Found C-55.00%, H-10.06%, N-10.70%.
N-Hexanoyl , O-methacryloylhydroxylamine
The procedure for the synthesis of N-hexanoyl , 0- methacryloyol hydroxylamine was as described above in Example A for N-lauroyl, O-methacryloyl hydroxylamine. However, the purification and crystallization procedures were different.
The product was filtered and evaporated to give an oil residue. The oil residue was dried briefly under vacuum and put in a freezer until solid. It was crystallized from ethanol :water, washed with cold water and recrystallized from n-hexane : ethyl acetate. Yield: 1.02 g (36%); mp: 34-35.5 °C . Elemental analysis: Calculated C-60.28%, H-8.60%, N-7.03%: Found C-60.12%, H-8.67%, N-7.04%.
Example C N-Propionyl, O-methacryloylhydroxylamine (PrNHOMA) N-Propionylhydroxyalmine
The synthesis and purification were exactly as described for N-lauroyl, O-methacryloylhydroxylamine, in Example A except for the use of ethyl propionate and a 15 mL:12 mL solution of distilled water and absolute alcohol for the reaction solvent. The resultant crystals were recrystallized from ethyl acetate. Yield: 5.01 g (56%); mp: 96-97 °C. Elemental analysis: Calculated C-40.44%, H-7.92%, N-15.72%: Found C-40.43% , H-7.91%, N-15.66%.
N-Propionyl , O-methacryloylhydroxylamine The synthesis and purification were exactly as described for N-lauroyl, O-methacryloylhydroxylamine in
Example A, except that the crystallization was carried out in N-hexane : ethyl acetate.
Yield: 1.28 g (57%); p: 68-70 °C. Elemental analysis: Calculated C-53.50%, H-7.05%, N-8.91%: Found C-53.59%,
H-7.02%, N-8.84%.
Example D N, O-Dimethacryloylhydroxylamine (MANHOMA) As noted above, synthesis of MANHOMA is different from other N, O-diacylhydroxylamines in that it involves only one step, i.e., the simultaneous - and O-acylation of hydroxylamine hydrochloride. A 2.5 g sample of hydroxylamine hydrochloride was dissolved in 12.5 g of pyridine. The temperature was brought to 5°C an ice water bath. A small amount of t-octylpyrocatechol was added as an inhibitor. While stirring 8.9 g of methacryloyl chloride was added dropwise. The ice-water bath was removed and the reaction mixture was stirred at room temperature for 2 h. The reaction mixture was
diluted with 25 mL chloroform. To convert pyridine to pyridinium hydrochloride, 4.2 mL of hydrochloric acid (0.0428 mole) was added dropwise. The organic layer was separated and washed 4 times with 20 mL water. It was then dried over magnesium sulphate. It was filtered and the filtrate was evaporated. To the oil residue obtained was added 50 mL of diethyl ether. The white precipitate formed was filtered and the filtrate evaporated. The oil residue was dried briefly using an oil pump. It was transferred into a freezer where it solidified. The solid crystallized from N-hexane : ethyl acetate to yield: 1.97 g (33%); mp : 58-59 °C. Elemental analysis: Calculated C-56.80%, H-6.55%, N-8.28%: Found C-56.72%, H-6.50%, N-8.23%.
Example E Synthesis of 4,4' -Pi (methacryloylamino) azobenzene (AAB) The azo cross-linking agent, 4,4'- di (methacryloylamino) -azobenzene, was prepared according to the procedure described by Brøndsted et al . , Hydrogels for Si te-specific Oral Drug Delivery: Synthesis and Characteriza tion . Biomaterials
12(1991)584-592. A 2.13 g sample of 4,4'- diaminoazobenzene was dissolved in 20 L pyridine. A small amount of free radical inhibitor was added. To this was added 3.16 g of methacryloyl chloride slowly under stirring at room temperature. The solution was stirred for 1 h at 60°C and then poured into 50 mL ice water. The reaction mixture was acidified with concentrated HCl to pH 4. The product was filtered and washed with saturated sodium bicarbonate and 150 mL water. The product was dried and crystallized from ethanol . Yield: 1.13 g (32.4%); mp: 278-279 °C .
Synthesis of hydrogels
Hydrogels were prepared using N,0- diacylhydroxylamines of the formula (Ac3NHOAc2) wherein Ac3 is a member selected from the group consisting of Acx or Ac2 to prepare hydrogels having general formula: poly[ (AAA)v-co-(AA)w-co-(Ac3NHOAC2)x-co-(BuAA)y-co-(AAB)z] . wherein AAA is an N-alkyl substituted (meth) acrylamide, AA is a member selected from the group consisting of acrylic or methacrylic acid, Ac3NHOAC2 is as defined above, BuAA is N-t-butylacrylamide, and AAB is a crosslinking agent containing an aromatic azo bond and v, w, x, y and z represent the mole percent or mole percent range of its corresponding group.
AAA may be a member selected from N-substituted acrylamides, N,N-disubstituted acrylamides and N- substituted methacrylamides of the formula:
Rι H2C C
C O
N
R2 R3
where R± is H or CH3; R2 is alkyl or hydroxyalkyl having 1 to 6 carbon atoms and R3 is H or R2 with the proviso that when Rx is CH3, R3 is H, and when Rx is H, R3 is R2. AAA may be present such that v represents a mole percent range of between about 20 to 90 mol% with ranges of between about 40 to 80 mol% being preferred. The N,N- disubstituted acrylamides and N, N-dimethylacrylamide in particular are preferred.
BuAA represents N-t-butylacrylamide and is generically within the scope of AAA. BuAA is added to
provide structural strength to the hydrogel and may be present such that y represents ranges of from 0 to 35 mol%. Preferably v will represent between about 5 to 20 mol%. Since BuAA is contained within the scope of the AAA definition, the mol% represented by the sum. of v and y will not exceed 90% and preferably will not exceed 80%.
AA is an ionizable monomer and is a member selected from the group consisting of acrylic and methacrylic acid. AA may be present such that w represents a mol% range of between about 0.1 to 60% and is preferably between about 5 and 45 mol%. The lower ranges are more practical for forming a pH swellable hydrogel in that the N, O-diacylhydroxyl a ine monomer also provides a means of generating ionizable groups at higher pH ranges. Also, at higher ranges, some of the ionizable COOH groups can be protected by converting some or all of these functionalities to N, O-diacylamine moieties by reaction with N-acylhydroxylamines, e.g. ACjNOH. Ac3HNOAc2 and may be present in mol% amount such that x is between about 1 and 50% with ranges of between about 5 and 30% being preferable.
AAB represents an aromatic azo benzene represented by the formula 5 Re Re R5 R ^N_-N_ R
wherein R4 is a member selected from the group consisting
of
where Rg is H or CH3 and where R5, R6, R7 and R8 are individually selected from the group consisting of H, CH3, OCH3, Br, Cl, F, I, N02 , CN or C(0)CH3. AAB may be present in amount such that z represents a mole percent of between about 0.05 and 15. Preferred AAB groups are those selected from the group consisting of 4,4'- di (methacryloylamino) azobenzene, 4-4 ' -di (N-methacrylol- e -aminocaproy 1 ) aminoazobenzene and 4,4'- di (methacryloylamino) -3 , 3 ' , 5 , 5 ' -tetrachloroazobenzene with 4, 4 ' -di (methacryloylamino) -azobenzene being particularly preferred and is used in the following examples .
The purpose of preparing the hydrogels containing both N, O-diacylhydroxylamine and 4,4'- di (methacryloylamine) -azobenzene moieties was to chemically and physically control the kinetics of hydrogel swelling as a function of the concentration of both of these agents .
In preparing the hydrogels of the following examples AAA is N, N-dimethylacrylamide, BuAA is t-butylacrylamide, AA is acrylic acid (but could as readily also be methacrylic acid), AAB is 4,4'- di (methacryloylamino) azobenzene and the N,0- diacylhydroxylamine is as specifically designated in each table or example. The hydrogels were synthesized by radical copolymerization of the monomers in 70% v/v DMSO using AIBN as the initiator. The concentration of AIBN used in the reaction mixture was 36mM. The monomer solution was bubbled with nitrogen for 30 minutes. Polymerization took place in a mold (made of Teflon®) separated by a silicone rubber gasket to obtain a desired thickness) at 60°C for 15 h. The gels were removed from the mold and washed in ethanol for six days and were gradually transferred into aqueous solution (pH 2.0), where they were stored until used. The
compositions of the polymerization solutions used for the synthesis of hydrogels of Examples 1-31 are shown in Tables 1, 2, 3, and 4.
As can be seen from the composition of Example 1, the general structure of the hydrogel is:
poly [ (AAA) v-co- (AA)w-co- (BuAA)y-co- (AAB) .
In the remaining examples (Examples 2-31) the AA was replaced in part by the N, O-diacylhydroxylamines (Ac3NHOAc2) of Examples A-D resulting in hydrogels of the formula :
poly[ (AAA)v-co- (AA)w-co- (Ac3NHOAC2)x-co- (BuAA) -co- {AAζ) ]
as defined above. The variables which control the chemical and physical kinetics of swelling as the pH of the hydrogels are raised to about 6.5 and above are the Ac3NHOAc2 and AAB contents respectively. Tables 1-4 list hydrogels of Examples 1-31 wherein each monomer is represented in terms of mole percent. Table 1: Monomer feed compositions based on N- lauroyl , O-methacryloylhydroxylamine Example
1 2 3 4 5 6 7 8 9
Table 2: Monomer feed compositions based on hexanoyl , O-methacryloylhydroxylamine
Table 3 : Monomer feed compositions based on - propionyl, O-methacryloylhydroxylamine (PrNHOMa)
Swelling studies were carried out in buffers at pH 2.0 (0.01 M HC1) and pH 7.4 (0.05 M phosphate buffer) at 37 °C . The ionic strength was adjusted with NaCl to 0.16 in all buffer solutions. Each hydrogel disc (three discs per sample) was equilibrated in 15 mL of a pH 2.0 solution at 37 °C. The gel was weighed after removal of excess surface water by light blotting with a laboratory tissue. The pH of the solution was checked using a pH meter and found unchanged.
The hydrogel disc was transferred into a vial containing 15 L of 0.04 M phosphate buffer (pH 7.4) already equilibrated at 37 °C in a shaking water bath. Changes in weight were monitored at time intervals until equilibrium swelling was reached. The pH of swelling solution was checked. In all cases, the pH remained unchanged. The swelling of the hydrogels was expressed as swelling ratio (weight of wet gel/weight of dry gel) .
Hydrogels have a high liquid content and are given form by the network of polymer strands made up of the formula poly [ (AAA)v-co- (AA)w-co- (Ac3NHOAC2) x-co- (BuAA)y- co-(AAB)z. Liquid within the network is held by a balance of forces. The hydrophilicity of the hydrogel network is conferred by various functional groups such as -OH, -COOH, -CONH2, -CONHR, and -S03H which are responsible for their ability to imbibe a large amount of water. The factors which may affect swelling of ionic hydrogels include the degree of ionization, the charge of the ionic monomer (s), the pKa of the ionizable group (s), the concentration of the ionizable monomer (s) in the network, the nature of the counterion, the nature of the swelling solution in
terms of pH, ionic strength and composition (i.e. buffering) , the cross-linking density and the hydrophilicity/hydrophobicity of the polymer. Some of these factors affect the kinetics of swelling of the hydrogels of this invention as illustrated by the swelling isotherms obtained which are illustrated in Tables 5-8 which follow.
Tables 5-8 illustrate the dependence of swelling isotherms of the hydrogels on the concentration of the Ac3NH0Ac2 when the hydrogels are transferred abruptly from pH 2.0 to pH 7.4 at 37 °C. A biphasic swelling pattern is observed for all hydrogels reported. First, there is an initial burst phase followed by a second phase whose approach to equilibrium swelling is gradual. It can be seen from data in these tables that the structures of the polymer backbone, which are determined by the proportions of acrylic acid, (AAW) , and hydrolyzable monomer, (Ac3NHOAc2)x greatly influence the kinetics of swelling and swelling equilibrium. Further, the nature of the Ac3NHOAc2 monomer also affects the swelling kinetics and equilibrium swelling.
To better illustrate the factors affecting swelling, Formulas 1-5 show representative structures which correlate with data in Tables 5-8.
Formula 1 shows a typical structure of a poly [ (AAA) v-co- (AA)w-co- (BuAA) y-co- (AAB) ,] hydrogel as shown in Example 1 which would be the same at either pH 2.0 or 7.4. Formula 2 is representative of a poly [AAAv-co- (AA) w- co (Ac1NHOAc2)x-co- (BuAA) y-co- (AAB) hydrogel as shown in Examples 2-25 at pH 2.0 showing the presence of the hydrolyzable ACiNHOAc2 monomer.
Formula 3 is representative of a polytAAA^-co- (AA)W- co (ACNHOACjJx-co- (BuAA)y-co- (AAB)Z] hydrogel of
Examples 2-25 at pH 7.4 showing the hydrolysis of the ACtNHOACj monomer to provide a -COOH functionality.
Formula 4 is representative of a polyfAAAv-co- (AA)W- co (MANHOMA)x-co- (BuAA)y-co- (AAB),] hydrogel as shown in Examples 28-31 at pH 2.0 showing the presence of the cross-linked hydrolyzable MANHOMA monomer.
Formula 5 is representative of a poly[AAAv-co- (AA) w- co (MANHOMA)x-co- (BuAA) y-co- (AAB),] hydrogel of Examples 26-31 at pH 7.4 showing the hydrolysis of the crosslinked MANHOMA monomer to provide -COOH and -NH2 functionalities .
FORMULA 1
HO
\ \
R1 R' R1
poly[ (co-BuAA)y- (co-AA)w- (co-AAB)2-AAA Rl= H, CH3
FORMULA 2
poly[ (co-BuAA)v- (co-AA)w- (coAcylNHOMA)x- (co-AAB) Z-AAAV] R^H or CH3
FORMULA 3
poly[ (co-BuAA)y- (co-AA)w- (coAcylNHOMA)x- (co-AAB) --AAAV] R*= H or CH3
FORMULA 4
poly[ (co-BuAA)γ- (co-AA)w- (coAcylNHOMA)x- (co-AAB) z -AAAV] R^H or CHj
FORMULA 5
poly[ (co-BuAA) γ- (co-AA)w- (coAcylNHOMA)x- ( co-AAB )Z-AAAV] Rl=H or CH3
TABLE 5
TABLE 6
TABLE 7
TABLE 8
Data not available Tables 5-8 show that hydrogels containing 0%
Ac3NHOAc2, i.e. the structure of Formula 1 wherein only acrylic acid is present as an ionizable monomer at pH 7.4 reached equilibrium swelling very rapidly and that the equilibrium swelling ratio remained constant. This is attributable to the ionization of acrylic acid in the polymer network. The carboxylic acid group of the acrylic acid is reported to have a pKa of about 4.3. Since the pH of 7.4, i.e. the swelling medium to which the gel was transferred after swelling at pH 2.0 is higher than the pKa of the carboxylic acid groups, ionization occurs and is accompanied by an increase in charge density, charge hydration, electrostatic repulsion between adjacent carboxylate groups and expansion of the polymer network. These, together with
increased hydrophilicity of the polymer network, results in the ability of the polymer to imbibe water.
As it relates to hydrogels containing various concentrations of Ac3NHOAc2, Tables 5-8 show that the magnitude of the initial swelling phase, i.e. the burst effect, decreases with the decrease in the concentration of the acrylic acid (AAW) content and the accompanying increase in the concentration of the Ac3NHOAc2 content . This pattern can be ascribed to the initial decrease, prior to hydrolysis relative to Example 1, in the ionic content of the hydrogel network as the concentration of the Ac3NHOAc2 monomer increases. The effect is to decrease the charge density, the hydrophilicity and the expansion of the polymer network.
Tables 5-8 also show that the second phase of the swelling isotherms of hydrogels containing varying concentrations of Ac3NHOAc2 approach equilibrium swelling gradually. This is thought to be due to the fact that the pKa values of the Ac3NHOAc2 monomers containing the hydrolytic linkage are around 4.5 to 4.85, which is lower than the 7.4 pH of the swelling medium as would be encountered in the small intestine. Thus the hydrolyzable moieties in the polymer exist mainly as monoanions at a pH of 7.4. These monoanions trigger off the hydrolytic process and result is the generation of carboxylic acid groups which will also ionize at pH 7.4 to increase the total ionic content of the hydrogels as shown by comparing Formulas 2 and 4 with Formulas 3 and 5 respectively. The net result is the promotion of swelling. At lower change density, before the hydrolysis of the Ac3NHOAc2 as illustrated in Formulas 2 and 4, the charges are shielded by the counterions present in the swelling solution. With the increase in charge density, as a result of the
Ac3NHOAc2 hydrolysis, as shown in Formulas 3 and 5, the shielding will be less efficient since the volume of
swelling solution is relatively constant. Hence, swelling will increase.
It will also be noted that the time taken to reach the equilibrium swelling ratio is longer as the concentration of the Ac3NHOAc2 increases. This is thought to be due to an increase in the number of groups to be hydrolyzed.
By comparing Q at a pH of 2.0 and 7.4 in Tables 5-8 it is also shown that the equilibrium swelling ratio is a function of the concentration of the Ac3NHOAc2 monomer. At pH 2.0, the hydrogels of Example 1, containing acrylic acid alone, have higher equilibrium swelling ration than hydrogels containing Ac3NHOAc2 monomers . Changes in the concentration of Ac3NHOAc2 have little or no effect on the equilibrium swelling ratios at pH 2.0 as shown in these tables . The unionized acrylic acid is more hydrophilic than the hydrolyzable monomers and therefore the swelling of Example 1 is greater at pH 2.0 and the swelling of hydrogels containing Ac3NHOAc2 at pH 2.0.
Theoretically, the equilibrium swelling ration is expected to be the same in all of the hydrogels after equilibrium swelling at pH 7.4 because the data reported resulting from Examples 1-31 was designed to make the acrylic acid concentration constant following hydrolysis. In other words, the combined concentration of AA and Ac3NHOAc2, in each example is 40% so the number of carboxylic acid groups following hydrolysis should be the same if hydrolysis proceed to completion. However, it will be noted in Table 5-8 that there are variations in the degree of swelling. However, for whatever reasons there are variations, this does not depart from the showing that there is a reproducible swelling produced upon raising the pH from 2.0 to 7.4 and that is shown by an initial swelling burst followed by a slower degree until equilibrium is reached. This allows one to formulate a
hydrogel by varying the selecting the desired Ac3NHOAc2 and then varying the concentration of that Ac3NHOAc2 to obtain the swelling kinetic that are desired.
Table 8 shows that the equilibrium swelling ratios of hydrogels containing MANHOMA (Examples 26-28) are below those of Example 1 . The structure of the hydrogel after swelling as shown in Formula 5, contains carboxylic acid groups and free amino groups. Ionic interaction between the two groups will have some effects on the concentration of the counterions in these hydrogels and therefore will restrict polymer chain expansion and swelling. This further shows that there is some difference in the functionality of the MANHOMA monomer of the prior art and the Ac.NHOAc- monomers synthesized as novel compounds herein.
The concentration of aromatic azo crosslinking agent influences the degree of swelling. The burst effect described above and the equilibrium swelling ratio decrease with increase in the concentration of the AAB crosslinking agent. The equilibrium swelling of the hydrogels reflects a balance between swelling forces and the network elastic retractive forces. This balance depends on the crosslinking density of the gel network. Thus as the degree of crosslinking increases, the equilibrium degree of swelling is restricted.
The combination of the chemical control of the pH sensitive hydrogels attributable to the labile N,0- diacylhydroxylamines and the physical control attributable to the AAB crosslinking makes it possible to control the swelling of the hydrogel in the small intestine such that the equilibrium degree of swelling is reached at about the time of entry of the hydrogel into the colon where biodegradation can be facilitated by azoreductase activity. The hydrogels of this invention can be utilized in a variety of ways to allow the oral delivery of a dosage form of peptide and protein drugs to the colon.
The drugs that are advantageously administered via the dosage forms of the invention are drugs that would be destroyed by the environments of the stomach/small intestine, have an irritating or other undesirable effect on the tissues of the upper gastrointestinal tract, or are intended to locally treat conditions within the colon or colonic tissue. Systemically acting drugs that are delivered via these dosage forms must be capable of being absorbed into circulation through the mucosal lining of the colon. Examples of drugs that may be delivered advantageously via this invention are peptide or protein drugs such as insulin, calcitonin, angiotensin, cyclosporin, vasopressin, and somatostatin. These hydrogels can function as coatings of various dosage form devices such as capsules, caplets, tablets and the like which can pass intact through the acid media of the stomach into the small intestine where the swelling process begins resulting from the rapid rise in pH to about 7.4. By the time the device reaches the colon the swelling will have reached equilibrium and the azoreductase enzyme, found primarily in the colon, will allow cleavage of the AAB linkage further degrading the polymer coating releasing the peptide or protein drug to the colon interior. The coating process can be carried out by in si tu coating of the device such that the copolymerization and crosslinking occur around the device surface. Also, a latex-type suspension of the hydrogel (e.g., a suspension of fine particles in a swelling agent/dispersant) may be formed and the device dipped or immersed in the suspension.
Further, the drugs may be enclosed in a film or membrane of the copolymer by forming the copolymer hydrogel into a film or membrane, placing a desired amount of drug between two pieces of the film/membrane and sealing the edges of the pieces . In this manner a
container whose walls are made of the copolymer and whose lumen contains a unit dose of drug are prepared.
The drugs may be also be formulated with the hydrogels by dispersing them in the hydrogels. Dispersions are made by mixing the drug with the solution of monomers and then forming the hydrogel by crosslinking in the manner as described above. Alternatively a body of the hydrogel may be soaked in a solution or dispersion of the drug which partially swells the hydrogel, without hydrolysis of the N,0- diacylhydroxylamine moieties, and the body then dried. The drug-hydrogel dispersions may be cut or otherwise shaped into a body containing the desired unit dose of drug. The amount of drug administered may be varied by varying the concentration of drug in the solution or dispersion, the solvent used to create the solution or dispersion, and the volume of the drug-hydrogel body.
Although certain peptide or protein drugs are mentioned above, the invention is not so limited. The invention is limited only by functionality, i.e. any peptide or protein drug that can be formulated into a device compatible with the hydrogels disclosed herein is within the scope of this invention. The above specification describes the invention in terms to enable one skilled in the art to practice. Only a representative sample of hydrogels which may be prepared are specifically shown. Moreover, it will be apparent to one skilled in that art that various modifications can be made without departing from the scope of the invention. Therefore, the invention is limited in scope only by the following claims and their functional equivalents.
Claims
1 . A N, O-diacylhydroxylamine having the formula : Ac1-NHO-Ac2 where Acx is an acyl group of the structure R11-CO , and Ac2 is an acyl group of the structure -CO-R12 wherein R11 is an alkyl group of from about 1 to 24 carbon atoms and R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH.
2. A N, O-diacylhydroxylamine according to Claim
1 wherein R-. is an alkyl group of from about 3 to 12 carbon atoms .
3. A N, O-diacylhydroxylamine according to Claim
2 wherein Rn is an alkyl group of 11 carbon atoms.
4. A N, O-diacylhydroxylamine according to Claim 3 wherein R12 is C=CH(CH3)
5. A N, O-diacylhydroxylamine according to Claim 2 wherein R.. is an alkyl group of 5 carbon atoms .
6. A N, O-diacylhydroxylamine according to Claim 5 wherein R12 is C=CH(CH3)
7. A N, O-diacylhydroxylamine according to Claim 2 wherein R1X is an alkyl group of 2 carbon atoms.
8. A N, O-diacylhydroxylamine according to Claim 7 wherein R12 is C=CH(CH3)
9. A crosslinked pH sensitive hydrogel having the formula: poly [ (AAA)v-co- (AA)w-co- (Ac3NHOAC2)x-co- (BuAA)y-co- (AAB) wherein (a) AAA is an N-alkyl substituted
(meth) erylamide and v represents a mol percent concentration of between about 20 and 90 mol%;
(b) AA is a member selected from the group consisting of acrylic or methacrylic acid and w represents a mol percent concentration of between about 0.1 and 60 mol%;
(c) Ac3NHOAC- is a N, O-diacylhydroxylamine wherein Ac3 is a member selected from the group consisting of Ac1 and Ac. where Acx is an acyl group of the structure Ru-C0, and Ac2 is an acyl group of the structure -CO-R12 wherein R11 is an alkyl group of from about 1 to 24 carbon atoms and R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH and x represents a mol percent concentration of between about 1 and 50 mol%;
(d) BuAA is N-t-butylacrylamide where y represents a mole percent concentration of between about 0 and 35 mol% and (e) AAB is a crosslinking agent containing an aromatic azo bond and z represents a mole percent concentration of between about 0.05 and 15 mol%, with the proviso that the sum of v and y does not exceed 90 mol% .
10. A hydrogel according to Claim 9 were AAA is a member selected from the group consisting of N- substituted acrylamides, N,N-disubstituted acrylamides and N-substituted methacrylamides of the formula:
H2C=C
C=O
N
/ \ R2 R3 where Rt is H or CH3; R2 is alkyl or hydroxyalkyl having 1 to 6 carbon atoms and R3 is H or R2 with the proviso that when Rx is CH3, R3 is H, and when Rx is H, R3 is R2.
11. A hydrogel according to Claim 10 where AAB represents an aromatic azo benzene of the formula
wherein R4 is a member selected from the group
where R9 is H or CH3 and where R5, R6, R7 and Rβ are individually selected from the group consisting of H, CH3, OCHj, Br, Cl, F, I, N02, CN or C(0)CH3.
12. A hydrogel according to Claim 10 wherein y represents a mol% concentration of between about 5 to 20 mol%.
13. A hydrogel according to Claim 12 wherein w represents a mol% concentration of between about 5 and
45 mol%.
14. A hydrogel according to Claim 13 wherein Ac3 is Ac2 which is an acyl group of the structure -CO-R12 wherein R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH.
15. A hydrogel according to Claim 13 wherein Ac3 is Ac: which is an acyl group of the structure R11-C0 wherein Rn is an alkyl group of from about 1 to 24 carbon atoms .
16. A hydrogel according to Claim 15 wherein Ru is an alkyl group of from about 3 to 12 carbon atoms .
17. A hydrogel according to Claim 16 wherein R is an alkyl group of 11 carbon atoms.
18. A hydrogel according to Claim 17 wherein R12 is C=CH(CH3) .
19. A hydrogel according to Claim 16 wherein RX1 is an alkyl group of 5 carbon atoms.
20. A hydrogel according to Claim 19 wherein R12 is C=CH(CH3) .
21. A hydrogel according to Claim 16 wherein Rn is an alkyl group of 2 carbon atoms .
22. A hydrogel according to Claim 21 wherein R12 is C=CH(CH3) .
23. A method of selectively delivering a peptide or protein drug to the colon of an animal or human comprising orally administering a dosage form comprising an effective amount of said drug contained by a crosslinked pH sensitive hydrogel having the formula: poly [ (AAA)v-co- (AA)w-co- (Ac3NHOAC2)x-co- (BuAA)y-co- (AAB) z] wherein (a) AAA is an N-alkyl substituted
(meth) acrylamide and v represents a mol percent concentration of between about 20 and 90 mol%;
(b) AA is a member selected from the group consisting of acrylic or methacrylic acid and w represents a mol percent concentration of between about 0.1 and 60 mol%;
(c) Ac3NHOAC2 is a N, O-diacylhydroxylamine wherein Ac3 is a member selected from the group consisting of Ac. and Ac2 where Ac. is an acyl group of the structure R -CO, and Ac2 is an acyl group of the structure -CO-R12 wherein R11 is an alkyl group of from about 1 to 24 carbon atoms and R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH and x represents a mol percent concentration of between about 1 and 50 mol%;
(d) BuAA is N-t-butylacrylamide where y represents a mole percent concentration of between about 0 and 35 mol%; and (e) AAB is a crosslinking agent containing an aromatic azo bond and z represents a mole percent concentration of between about 0.05 and 15 mol%, with the proviso that the sum of v and y does not exceed 90 mol% .
24. A method according to Claim 23 wherein said dosage form is coated by said hydrogel.
25. A method according to Claim 23 wherein said dosage form contains said drug dispersed in said hydrogel .
26. A method according to Claim 24 or Claim 25 wherein AAA is a member selected from the group consisting of N-substituted acrylamides, N,N- disubstituted acrylamides and N-substituted methacrylamides of the formula: H2C=C
C=0
N
/ \ R2 R3
where Rx is H or CH3; R2 is alkyl or hydroxyalkyl having 1 to 6 carbon atoms and R3 is H or R2 with the proviso that when Ri is CH3, R3 is H, and when Rx is H, R3 is R2.
27. A method according to Claim 26 where AAB represents an aromatic azo benzene of the formula
wherein R4 is a member selected from the group
where R9 is H or CH3 and where R5, R6, R- and R8 are individually selected from the group consisting of H, CH3, OCH3, Br, Cl, F, I, N02, CN or C(0)CH3.
28. A method according to Claim 27 wherein y represents a mol% concentration of between about 5 to 20 mol%.
29. A method according to Claim 28 wherein w represents a mol% concentration of between about 5 and 45 mol%.
30. A method according to Claim 29 wherein Ac3 is Ac2 which is an acyl group of the structure -CO-R12 wherein R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH.
31. A method according to Claim 30 wherein Ac3 is Acα which is an acyl group of the structure Ru-CO wherein R11 is an alkyl group of from about 1 to 24 carbon atoms .
32. A method according to Claim 31 wherein R1X is an alkyl group of from about 3 to 12 carbon atoms.
33. A composition for selectively delivering a peptide or protein drug to the colon of an animal or human comprising a dosage form containing an effective amount of said drug contained by a crosslinked pH sensitive hydrogel having the formula: poly[ (AAA)v-co- (AA) „-co- (Ac3NHOAC2)x-co- (BuAA)y-co- (AAB) wherein (a) AAA is an N-alkyl substituted
(meth) acrylamide and v represents a mol percent concentration of between about 20 and 90 mol%;
(b) AA is a member selected from the group consisting of acrylic or methacrylic acid and w represents a mol percent concentration of between about 0.1 and 60 mol%;
(c) Ac3NHOAC2 is a N, O-diacylhydroxylamine wherein Ac3 is a member selected from the group consisting of Ac. and Ac2 where Ac. is an acyl group of the structure R^-CO, and Ac2 is an acyl group of the structure -CO-R12 wherein R11 is an alkyl group of from about 1 to 24 carbon atoms and R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH and x represents a mol percent concentration of between about 1 and 50 mol%;
(d) BuAA is N-t-butylacrylamide where y represents a mole percent concentration of between about 0 and 35 mol%; and
(e) AAB is a crosslinking agent containing an aromatic azo bond and z represents a mole percent concentration of between about 0.05 and 15 mol%, with the proviso that the sum of v and y does not exceed 90 mol%.
34. A composition according to Claim 33 wherein said dosage form is coated by said hydrogel.
35. A composition according to Claim 33 wherein said dosage form contains said drug dispersed in said hydrogel .
36. A composition according to Claim 34 or Claim 36 wherein AAA is a member selected from the group consisting of N-substituted acrylamides, N,N- disubstituted acrylamides and N-substituted methacrylamides of the formula:
Ri
H2C =C c=o
N
/ \ R2 R3
where Rx is H or CH3; R2 is alkyl or hydroxyalkyl having 1 to 6 carbon atoms and R3 is H or R2 with the proviso that when Rt is CH3, R3 is H, and when R1 is H, R3 is R2.
37. A composition according to Claim 36 where AAB represents an aromatic azo benzene of the formula
wherein R4 is a member selected from the group
where R9 is H or CH3 and where R5, R6, R7 and Rβ are individually selected from the group consisting of H, CH3, 0CH3, Br, Cl, F, I, N0-, CN or C(0)CH3.
38. A composition according to Claim 37 wherein y represents a mol% concentration of between about 5 to 20 mol%.
39. A composition according to Claim 38 wherein w represents a mol% concentration of between about 5 and 45 mol%.
40. A composition according to Claim 39 wherein Ac3 is ACj which is an acyl group of the structure -C0- R12 wherein R12 is a member selected from the group consisting of C=CH(CH3) and CH2=CH.
41. A composition according to Claim 40 wherein Ac3 is Acx which is an acyl group of the structure R11- CO wherein R11 is an alkyl group of from about 1 to 24 carbon atoms .
42. A method according to Claim 41 wherein Rn is an alkyl group of from about 3 to 12 carbon atoms .
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US2157396P | 1996-07-10 | 1996-07-10 | |
| US60/021,573 | 1996-07-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1998001421A1 true WO1998001421A1 (en) | 1998-01-15 |
Family
ID=21804986
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1997/012305 WO1998001421A1 (en) | 1996-07-10 | 1997-07-09 | pH SENSITIVE HYDROGELS WITH ADJUSTABLE SWELLING KINETICS FOR COLON-SPECIFIC DELIVERY OF PEPTIDES AND PROTEINS |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO1998001421A1 (en) |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2932485A1 (en) * | 2008-06-12 | 2009-12-18 | Univ Pasteur | SPECIFIC COLLECTIVE RELEASE POLYMER WHATEVER PH |
| US8273100B2 (en) * | 2002-07-31 | 2012-09-25 | Microvention, Inc. | Three element coaxial vaso-occlusive device |
| US8764788B2 (en) | 2002-07-31 | 2014-07-01 | Microvention, Inc. | Multi-layer coaxial vaso-occlusive device |
| US9114200B2 (en) | 2009-09-24 | 2015-08-25 | Microvention, Inc. | Injectable hydrogel filaments for biomedical uses |
| US9259228B2 (en) | 2006-06-15 | 2016-02-16 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9381278B2 (en) | 2012-04-18 | 2016-07-05 | Microvention, Inc. | Embolic devices |
| US9456823B2 (en) | 2011-04-18 | 2016-10-04 | Terumo Corporation | Embolic devices |
| US9486221B2 (en) | 2007-12-21 | 2016-11-08 | Microvision, Inc. | Hydrogel filaments for biomedical uses |
| CN106905190A (en) * | 2017-02-23 | 2017-06-30 | 禹城禹圳生物科技有限公司 | The aqueous phase preparation method of caprylhydroxamic acid sterling |
| WO2017189621A1 (en) * | 2016-04-26 | 2017-11-02 | Symbiotic Health Inc. | Stable macroemulsion for oral delivery of solubilized peptides, protein, and cellular therapeutics |
| US9993252B2 (en) | 2009-10-26 | 2018-06-12 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US10092663B2 (en) | 2014-04-29 | 2018-10-09 | Terumo Corporation | Polymers |
| US10124090B2 (en) | 2014-04-03 | 2018-11-13 | Terumo Corporation | Embolic devices |
| US10226533B2 (en) | 2014-04-29 | 2019-03-12 | Microvention, Inc. | Polymer filaments including pharmaceutical agents and delivering same |
| US10639396B2 (en) | 2015-06-11 | 2020-05-05 | Microvention, Inc. | Polymers |
| CN111205392A (en) * | 2020-03-03 | 2020-05-29 | 长江大学 | Thermal degradation hydrogel for pressure temporary plugging |
| US11136490B2 (en) | 2017-11-10 | 2021-10-05 | China University Of Petroleum (East China) | PH sensitive water-absorbent resin suitable for oil well cement slurry and application thereof |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3412179A (en) * | 1966-06-27 | 1968-11-19 | Geigy Chem Corp | Polymers of acrylyl perfluorohydroxamates |
| US5124421A (en) * | 1989-12-20 | 1992-06-23 | Ceskoslovenska Akademie Ved | Hydrolytically degradable hydrophilic gels and the method for preparation thereof |
| US5415864A (en) * | 1990-04-18 | 1995-05-16 | University Of Utah Research Foundation | Colonic-targeted oral drug-dosage forms based on crosslinked hydrogels containing azobonds and exhibiting PH-dependent swelling |
-
1997
- 1997-07-09 WO PCT/US1997/012305 patent/WO1998001421A1/en active Application Filing
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3412179A (en) * | 1966-06-27 | 1968-11-19 | Geigy Chem Corp | Polymers of acrylyl perfluorohydroxamates |
| US5124421A (en) * | 1989-12-20 | 1992-06-23 | Ceskoslovenska Akademie Ved | Hydrolytically degradable hydrophilic gels and the method for preparation thereof |
| US5415864A (en) * | 1990-04-18 | 1995-05-16 | University Of Utah Research Foundation | Colonic-targeted oral drug-dosage forms based on crosslinked hydrogels containing azobonds and exhibiting PH-dependent swelling |
Cited By (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8764788B2 (en) | 2002-07-31 | 2014-07-01 | Microvention, Inc. | Multi-layer coaxial vaso-occlusive device |
| US8882796B2 (en) | 2002-07-31 | 2014-11-11 | Microvention, Inc. | Three element coaxial vaso-occlusive device |
| US8273100B2 (en) * | 2002-07-31 | 2012-09-25 | Microvention, Inc. | Three element coaxial vaso-occlusive device |
| US10499925B2 (en) | 2006-06-15 | 2019-12-10 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9724103B2 (en) | 2006-06-15 | 2017-08-08 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9877731B2 (en) | 2006-06-15 | 2018-01-30 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9259228B2 (en) | 2006-06-15 | 2016-02-16 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US11160557B2 (en) | 2006-06-15 | 2021-11-02 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9451963B2 (en) | 2006-06-15 | 2016-09-27 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US10226258B2 (en) | 2006-06-15 | 2019-03-12 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US11185336B2 (en) | 2006-06-15 | 2021-11-30 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9486221B2 (en) | 2007-12-21 | 2016-11-08 | Microvision, Inc. | Hydrogel filaments for biomedical uses |
| US10194915B2 (en) | 2007-12-21 | 2019-02-05 | Microvention, Inc. | Implantation devices including hydrogel filaments |
| WO2009153447A1 (en) * | 2008-06-12 | 2009-12-23 | Universite De Strasbourg | Oral galenic form, polymer production method and use of same |
| FR2932485A1 (en) * | 2008-06-12 | 2009-12-18 | Univ Pasteur | SPECIFIC COLLECTIVE RELEASE POLYMER WHATEVER PH |
| US9114200B2 (en) | 2009-09-24 | 2015-08-25 | Microvention, Inc. | Injectable hydrogel filaments for biomedical uses |
| US9993252B2 (en) | 2009-10-26 | 2018-06-12 | Microvention, Inc. | Embolization device constructed from expansile polymer |
| US9456823B2 (en) | 2011-04-18 | 2016-10-04 | Terumo Corporation | Embolic devices |
| US9381278B2 (en) | 2012-04-18 | 2016-07-05 | Microvention, Inc. | Embolic devices |
| US10124090B2 (en) | 2014-04-03 | 2018-11-13 | Terumo Corporation | Embolic devices |
| US10092663B2 (en) | 2014-04-29 | 2018-10-09 | Terumo Corporation | Polymers |
| US10226533B2 (en) | 2014-04-29 | 2019-03-12 | Microvention, Inc. | Polymer filaments including pharmaceutical agents and delivering same |
| US10946100B2 (en) | 2014-04-29 | 2021-03-16 | Microvention, Inc. | Polymers including active agents |
| US10639396B2 (en) | 2015-06-11 | 2020-05-05 | Microvention, Inc. | Polymers |
| US11759547B2 (en) | 2015-06-11 | 2023-09-19 | Microvention, Inc. | Polymers |
| WO2017189621A1 (en) * | 2016-04-26 | 2017-11-02 | Symbiotic Health Inc. | Stable macroemulsion for oral delivery of solubilized peptides, protein, and cellular therapeutics |
| CN106905190A (en) * | 2017-02-23 | 2017-06-30 | 禹城禹圳生物科技有限公司 | The aqueous phase preparation method of caprylhydroxamic acid sterling |
| US11136490B2 (en) | 2017-11-10 | 2021-10-05 | China University Of Petroleum (East China) | PH sensitive water-absorbent resin suitable for oil well cement slurry and application thereof |
| CN111205392A (en) * | 2020-03-03 | 2020-05-29 | 长江大学 | Thermal degradation hydrogel for pressure temporary plugging |
| CN111205392B (en) * | 2020-03-03 | 2022-06-17 | 长江大学 | Thermal degradation hydrogel for pressure temporary plugging |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1998001421A1 (en) | pH SENSITIVE HYDROGELS WITH ADJUSTABLE SWELLING KINETICS FOR COLON-SPECIFIC DELIVERY OF PEPTIDES AND PROTEINS | |
| Akala et al. | Novel pH-sensitive hydrogels with adjustable swelling kinetics | |
| US5415864A (en) | Colonic-targeted oral drug-dosage forms based on crosslinked hydrogels containing azobonds and exhibiting PH-dependent swelling | |
| EP0910412B1 (en) | Hydrolysable hydrogels for controlled release | |
| US6514535B2 (en) | Bioadhesive hydrogels with functionalized degradable crosslinks | |
| Brøndsted et al. | Hydrogels for site-specific drug delivery to the colon: in vitro and in vivo degradation | |
| EP1307233B1 (en) | Therapeutic polyanhydride compounds for drug delivery | |
| AU740747B2 (en) | Delivery of nucleic acid material to target cells in biological systems | |
| US8685382B2 (en) | Temperature sensitive polymers | |
| US5053228A (en) | Polymeric temperature sensitive drug carrier | |
| US4806621A (en) | Biocompatible, bioerodible, hydrophobic, implantable polyimino carbonate article | |
| US5770627A (en) | Hydrophobically-modified bioadhesive polyelectrolytes and methods relating thereto | |
| US8852640B2 (en) | Micelles for delivery of nitric oxide | |
| Dvořák et al. | High-molecular weight HPMA copolymer–adriamycin conjugates | |
| WO1999051221A1 (en) | Hydrolytically unstable biocompatible polymer | |
| NZ220323A (en) | 3-dimensional network of microspheres containing biologically active macromolecular agents | |
| US5980883A (en) | Polymer gel for medical use | |
| Mahkam et al. | Synthesis and characterization of acrylic type hydrogels containing azo derivatives of 5-amino salicylic acid for colon-specific drug delivery | |
| AU671651B2 (en) | A drug delivery device and a method of making such device | |
| Sintov et al. | Polymeric drug delivery of enzymatically degradable pendant agents: Peptidyl-linked procainamide model system studies | |
| Park et al. | Hydrogels for Drug Delivery System:-Colon-Specific Delivery | |
| Mahkam et al. | New pH-sensitive hydrogels for Colon-specific drug delivery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| NENP | Non-entry into the national phase |
Ref country code: JP Ref document number: 1998505387 Format of ref document f/p: F |
|
| 122 | Ep: pct application non-entry in european phase |