WO1995019567A1 - Synthetic receptors, libraries and uses thereof - Google Patents
Synthetic receptors, libraries and uses thereof Download PDFInfo
- Publication number
- WO1995019567A1 WO1995019567A1 PCT/US1995/000572 US9500572W WO9519567A1 WO 1995019567 A1 WO1995019567 A1 WO 1995019567A1 US 9500572 W US9500572 W US 9500572W WO 9519567 A1 WO9519567 A1 WO 9519567A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- synthetic receptor
- library
- receptor
- synthetic
- solid support
- Prior art date
Links
- 0 **C1C(*)CCCC1 Chemical compound **C1C(*)CCCC1 0.000 description 16
- RFTQREMUFYPYBX-UHFFFAOYSA-N C(C1CCCC1)C1=CC=C1 Chemical compound C(C1CCCC1)C1=CC=C1 RFTQREMUFYPYBX-UHFFFAOYSA-N 0.000 description 1
- PBGVGMSFACKITD-UHFFFAOYSA-N CC(CCC(O)O)C(CC1)C(C)(CC2)C1C(CC1)C2C(C)(CC2)C1CC2OPC Chemical compound CC(CCC(O)O)C(CC1)C(C)(CC2)C1C(CC1)C2C(C)(CC2)C1CC2OPC PBGVGMSFACKITD-UHFFFAOYSA-N 0.000 description 1
- MWCOEJVLDYPHKB-UHFFFAOYSA-N CC(CCC(OCc(c(N=O)c1)ccc1C(O)=O)=O)C(CC1)C(C)(CC2)C1C(CC1)C2C(C)(CC2)C1CC2=C Chemical compound CC(CCC(OCc(c(N=O)c1)ccc1C(O)=O)=O)C(CC1)C(C)(CC2)C1C(CC1)C2C(C)(CC2)C1CC2=C MWCOEJVLDYPHKB-UHFFFAOYSA-N 0.000 description 1
- LTGWLUHUBIEYMM-VXKDOGTRSA-N CC(CCC(OCc(c([N+]([O-])=O)c1)ccc1C(NCC1=CC=CC1)=O)=O)C(CC1)C(C)(CCC2C(C)(CCC(C3)OP(C)[C@H](C)OC(C)=O)C3C3)C1C2C3=[IH] Chemical compound CC(CCC(OCc(c([N+]([O-])=O)c1)ccc1C(NCC1=CC=CC1)=O)=O)C(CC1)C(C)(CCC2C(C)(CCC(C3)OP(C)[C@H](C)OC(C)=O)C3C3)C1C2C3=[IH] LTGWLUHUBIEYMM-VXKDOGTRSA-N 0.000 description 1
- FXSUTKNIHXGZFF-UHFFFAOYSA-N CC(CCCC1)C1C=C Chemical compound CC(CCCC1)C1C=C FXSUTKNIHXGZFF-UHFFFAOYSA-N 0.000 description 1
- DTJTVSNYIHHLIG-UHFFFAOYSA-N CC(OCC1(CC2)CC2(COC(OCC(C(C2)C=[NH+][O-])=CC=C2C(C)=O)=C)CC1)=C Chemical compound CC(OCC1(CC2)CC2(COC(OCC(C(C2)C=[NH+][O-])=CC=C2C(C)=O)=C)CC1)=C DTJTVSNYIHHLIG-UHFFFAOYSA-N 0.000 description 1
- YVAPTZXHZVIOJG-ADEDTAMJSA-N C[C@H](CCC(NCC1CCCCC1)=O)[C@@H](CCC1C(CC2)C(C3)[C@@](C)(CC4)[C@@]2(C)C[C@@H]4O)[C@@]1(C)[C@H]3O Chemical compound C[C@H](CCC(NCC1CCCCC1)=O)[C@@H](CCC1C(CC2)C(C3)[C@@](C)(CC4)[C@@]2(C)C[C@@H]4O)[C@@]1(C)[C@H]3O YVAPTZXHZVIOJG-ADEDTAMJSA-N 0.000 description 1
- RFFGIVXYTIOARL-CLFYSBASSA-N Cc(cc1)ccc1C(N(C)CCc1ccc(C2/C=C\CCCCCC2)cc1)=O Chemical compound Cc(cc1)ccc1C(N(C)CCc1ccc(C2/C=C\CCCCCC2)cc1)=O RFFGIVXYTIOARL-CLFYSBASSA-N 0.000 description 1
- SSJXIUAHEKJCMH-LWOQYNTDSA-N N[C@H](CCCC1)C1N Chemical compound N[C@H](CCCC1)C1N SSJXIUAHEKJCMH-LWOQYNTDSA-N 0.000 description 1
- VANNPISTIUFMLH-UHFFFAOYSA-N O=C(CCC1)OC1=O Chemical compound O=C(CCC1)OC1=O VANNPISTIUFMLH-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J9/00—Normal steroids containing carbon, hydrogen, halogen or oxygen substituted in position 17 beta by a chain of more than two carbon atoms, e.g. cholane, cholestane, coprostane
- C07J9/005—Normal steroids containing carbon, hydrogen, halogen or oxygen substituted in position 17 beta by a chain of more than two carbon atoms, e.g. cholane, cholestane, coprostane containing a carboxylic function directly attached or attached by a chain containing only carbon atoms to the cyclopenta[a]hydrophenanthrene skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J41/00—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring
- C07J41/0033—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005
- C07J41/0055—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005 the 17-beta position being substituted by an uninterrupted chain of at least three carbon atoms which may or may not be branched, e.g. cholane or cholestane derivatives, optionally cyclised, e.g. 17-beta-phenyl or 17-beta-furyl derivatives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07J—STEROIDS
- C07J41/00—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring
- C07J41/0033—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005
- C07J41/0055—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005 the 17-beta position being substituted by an uninterrupted chain of at least three carbon atoms which may or may not be branched, e.g. cholane or cholestane derivatives, optionally cyclised, e.g. 17-beta-phenyl or 17-beta-furyl derivatives
- C07J41/0061—Normal steroids containing one or more nitrogen atoms not belonging to a hetero ring not covered by C07J41/0005 the 17-beta position being substituted by an uninterrupted chain of at least three carbon atoms which may or may not be branched, e.g. cholane or cholestane derivatives, optionally cyclised, e.g. 17-beta-phenyl or 17-beta-furyl derivatives one of the carbon atoms being part of an amide group
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K1/00—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length
- C07K1/04—General methods for the preparation of peptides, i.e. processes for the organic chemical preparation of peptides or proteins of any length on carriers
- C07K1/047—Simultaneous synthesis of different peptide species; Peptide libraries
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/12—Cyclic peptides with only normal peptide bonds in the ring
- C07K5/123—Tripeptides
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Q—MEASURING OR TESTING PROCESSES INVOLVING ENZYMES, NUCLEIC ACIDS OR MICROORGANISMS; COMPOSITIONS OR TEST PAPERS THEREFOR; PROCESSES OF PREPARING SUCH COMPOSITIONS; CONDITION-RESPONSIVE CONTROL IN MICROBIOLOGICAL OR ENZYMOLOGICAL PROCESSES
- C12Q1/00—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions
- C12Q1/68—Measuring or testing processes involving enzymes, nucleic acids or microorganisms; Compositions therefor; Processes of preparing such compositions involving nucleic acids
- C12Q1/6811—Selection methods for production or design of target specific oligonucleotides or binding molecules
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2219/00—Chemical, physical or physico-chemical processes in general; Their relevant apparatus
- B01J2219/00274—Sequential or parallel reactions; Apparatus and devices for combinatorial chemistry or for making arrays; Chemical library technology
- B01J2219/00277—Apparatus
- B01J2219/0054—Means for coding or tagging the apparatus or the reagents
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2219/00—Chemical, physical or physico-chemical processes in general; Their relevant apparatus
- B01J2219/00274—Sequential or parallel reactions; Apparatus and devices for combinatorial chemistry or for making arrays; Chemical library technology
- B01J2219/00277—Apparatus
- B01J2219/0054—Means for coding or tagging the apparatus or the reagents
- B01J2219/00572—Chemical means
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J2219/00—Chemical, physical or physico-chemical processes in general; Their relevant apparatus
- B01J2219/00274—Sequential or parallel reactions; Apparatus and devices for combinatorial chemistry or for making arrays; Chemical library technology
- B01J2219/00583—Features relative to the processes being carried out
- B01J2219/00592—Split-and-pool, mix-and-divide processes
-
- C—CHEMISTRY; METALLURGY
- C40—COMBINATORIAL TECHNOLOGY
- C40B—COMBINATORIAL CHEMISTRY; LIBRARIES, e.g. CHEMICAL LIBRARIES
- C40B70/00—Tags or labels specially adapted for combinatorial chemistry or libraries, e.g. fluorescent tags or bar codes
Definitions
- Receptors are molecules which selectively interact with other molecules. Receptor molecules perform a variety of tasks from selective binding of substrates to catalyzing chemical reactions.
- One example of a multifunctional receptor molecule is monoclonal antibodies. Monoclonal antibodies bind to other molecules (antigens) with very high selectivity, while in other cases they catalyze chemical reactions by selectively binding the transition states of those chemical reactions. Monoclonal antibodies are used as medicinal and diagnostic agents. Other receptor molecules are used as drug targeting molecules and are sometimes referred to as "magic bullets". In all cases, the receptor molecules effectiveness is dependent upon its ability to bind molecular species (substrates) with high discrimination and selectivity, i.e. not bind other often closely related molecular species.
- Antibodies are proteins produced in response to the presence of a foreign substrate (Stryer, L. Biochemistry, 3rd Edition, W.H. Freeman and Company, New York, 1988 and Schultz, P.G. Acc . Chem. Res . , 1989, 22, 287).
- a foreign substrate capable of eliciting antibody formation is called an antigen.
- Each antibody has a highly specific affinity for the antigen which stimulated its synthesis.
- the free energy for an antibody binding its antigen is normally from 6-15 kcal/mol. Structure analysis of antibodies have indicated that most have an immunoglobulin structure. Immunoglobulins are flexible Y-Shape molecules and consists of two kinds of polypeptide chains named as light and heavy chains molecular weight ( Figure 1).
- the invention is directed to synthetic receptor(s) which comprises a polyfunctional organic template covalently linked to two or more oligomers which may independently be the same or different and may independently be straight chain, cyclic or branched.
- the template may be linked to an identifier which uniquely defines the synthetic receptor.
- the identifier is a stable chemical molecule or a plurality of stable chemical molecules distinguishable and detectable to picomolar levels or may be an oligonucleotide.
- the template is covalently linked to a solid support which is linked to an identifier.
- the invention includes methods of preparing synthetic receptors and synthetic receptor libraries.
- the synthetic library may be linked with identifiers such that the library comprises a plurality of different synthetic receptor members.
- the invention also provides methods for assaying a synthetic receptor library to determine suitable synthetic receptor(s) which (a) bind an acceptor molecule; (b) exhibit biological activity;
- Figure 1 illustrates an IgG molecule.
- Both L (light) and H (heavy) chains consist of a variable (V) and constant (C) region.
- the variable regions of light chain (V L ) and heavy chain (V H ) are similar in the length and sequence.
- Immunoglobulin G can be cleaved into three fragments. Two of these fragments bind antigen. They are named as F ab (ab stands for antigen-binding, F for fragments).
- F ab contains one combining site (or antigen-binding site) for antigen, and it has the same binding affinity for antigen as does the whole molecule.
- the other fragment, called Fc because it crystallizes readily, does not bind an antigen.
- F ab contains four subunits V L , V H , C L and C H1 .
- Figure 2 illustrates the combinatorial method of split-synthesis used to generate 27 tripeptides on a solid support.
- Figure 3 illustrates the process used to monitor reactions on solid supports.
- Figure 4 illustrates the color screening assay using Disperse Red to detect the members of receptor library on beads that bind a substrate.
- 1 shows the beads before treatment with a colored substrate; 2 represents the color substrate in an organic solvent which is added to the beads and 3 shows the result after one day in a sealed vial to allow for equilibration to occur.
- Figure 5 illustrates the reading of the molecular bar code from a single synthesis bead.
- Figure 6 illustrates a typical gas chromatography spectrum for a single decoded bead. (The binary encoding system with molecular tags was used). Detailed Description of the Invention
- a polyfunctional organic template may be (a) a monocyclic aliphatic hydrocarbon, (b) a polycyclic aliphatic hydrocarbon, (c) a monocyclic aromatic hydrocarbon, (d) a polycyclic aromatic hydrocarbon, (e) a monocyclic heterocycle, (f) a polycyclic heterocycle, or (g) a macrocycle.
- fused polycyclic hydrocarbons may be used as templates: pentalene, indene, naphthalene, azulene, heptalene, biphenylene, asym-indacene, sym-indacene, acenaphthylene, fluorene, phenalene, phenanthrene, anthracene, fluoranthene, acephananthrylene, aceanthrylene, triphenylene, pyrene, chrysene and naphthacene (Dean, J.A., ed., Lange's Handbook of Chemistry, Thirteenth Edition, (1985), pages 7-9 and 7-10, MacGraw-Hill, Inc., New York).
- heterocyclic systems may be used as templates: thiophene, thianthrene, furan, pyran, isobenzofuran, chromene, anthene, phenoxanthiin, 2 -H-pyrrole, pyrrole, imidazole, pyrazole, isothiazole, isoxazole, pyridine, pyrazine, pyrimidine, pyridazine, indolizine, isoindole, 3-H-indole, indole, iH-Indazole, purine, 4-H-quinolizine, isoquinoline, quinoline, phthalazine, naphthyridine, quinoxaline, quinazoline, cinnoline, pteridine, 4aH-carbazole, carbazole, ⁇ -carboline, phenanthridine, acridine, perimidine, phenanthroline, phenazine and phenar
- heterocyclic systems may be used as templates: isochroman, chroman, pyrrolidine, pyrazolidine, pyrroline, imidazolidine, imidazoline, pyrazolidine, pyrazoline, piperidine, piperazine, indoline, isoindoline, quinuclidine and morpholine (Dean, J.A., ed., Lange's Handbook of Chemistry, Thirteenth Edition, (1985), pages 7-17 - 7-18, MacGraw-Hill, Inc., New York).
- steroid hormones may be used as templates: hydrocortisone, cortisone, corticosterone, aldosterone, deoxycorti costerone, 9cef luorohydrocort isone, prednisolone, methyl prednisolone, dexamethasone, triamcinolone, estradiol 17/3, estrone, estriol, progesterone, pregnanediol and testosterone (Dean, J.A., ed., Lange's Handbook of Chemistry, Thirteenth Edition, (1985), pages 7-116 - 7-19, MacGraw-Hill, Inc., New York).
- the following compounds may be used as templates: ferrocenecarboxylic acid and 1,1'-ferrocenedimethanol
- an oligomer includes acyclic or cyclic homomers, heteromers and acyclic or cyclic oligomers include an oligoamide, an oligoester, an oligourea, an oligourethane, an oligoamine, an oligoether, an oligosulfonamide, an oligophosphonamide, an oligophosphonate, an oligophosphate, an oligonucleotide, an oligosaccharide, a peptide oligomer, a cyclophane or a mixture of monomers thereof.
- An oligomer or portions thereof may also be prepared prior to attachment to the polyfunctional organic template.
- the monomers may be coupled together to form units of two or more monomers which are then further joined the polyfunctional organic template.
- covalently linked is a linkage such as an ester bond, an amide bond, an amine bond, an ether bond, or a linkage through a sulfur, silicon, nitrogen, oxygen, carbon atom, or a covalent bond to any suitable atom.
- This invention is directed to a synthetic receptor comprising a polyfunctional organic template covalently linked to two or more oligomer arms which are macrocyclic, acyclic, branched acyclic, cyclic or branched cyclic, or polycyclic which may independently be the same or different and may independently be straight chain, cyclic or branched or combinations thereof.
- the polyfunctional organic template may be an acyclic, a carbocyclic, a heterocycle, a polycarbocycle, a polycycle hydrocarbon, a polyheterocycle, a macrocyclic polyether, a macrocyclic polyamine, a macrocyclic polyamide, macrocyclic polyester, a macrobicycle, a macrotricycle, a macrotetracycle, a podand, a steroid or
- polyfunctional organic templates include porphyrin rings, cyclodextrins, oligoprolines, calixeranes and macrocycles of the type shown below
- the cyclic heterocycle may be a cyclic polypeptide such as cyclosporin
- the oligomer may be an oligoamide, an oligoester, an oligourea, an oligourethane, an oligoamine, an oligoether, an oligosulfonamide, an oligophosphonamide, an oligophosphonate, an oligophosphate, an oligonucleotide, an oligosaccharide, a peptide oligomer, a cyclophane or a mixture of monomers thereof.
- the oligomers are polypeptides or oligonucleotides. In one preferred, embodiment of the invention each oligomer contains less than ten monomers.
- the oligomers of the synthetic receptor may be the same or different. In another preferred embodiment at least one of the oligomers is a combination of two or more distinct classes of oligomers selected from the group consisting of the above mentioned oligomers.
- the oligomer may be a heteromer or a homomer.
- the oligomer may be a cyclic or acyclic.
- the synthetic receptor may be a polyfunctional steroid template covalently linked to two or more peptide oligomers which may independently be the same or different and which may independently be straight chain, cyclic or branched.
- the polyfunctional steroid template may be ursodeoxycholic acid, hyodeoxycholic acid, alpha-apocholic acid,
- the oligomer may be a peptide oligomer comprising at least two amino acids.
- the template may be further linked to a dye, a fluorescent label or a radioactive label.
- the polyfunctional organic template may further be linked to an identifier which uniquely defines the synthetic receptor.
- the identifier uniquely defines the synthesis and molecular structure of the oligomers of the synthetic receptor.
- the identifier is a stable chemical molecule or a plurality of stable chemical molecules distinguishable and detectable to picomolar levels or may be an oligonucleotide.
- the synthetic receptor may be a polyfunctional organic template covalently linked to a solid support and to two or more oligomers which may independently be the same or different and which may independently be straight chain, cyclic or branched.
- the solid support is preferably a particle composed of cellulose, controlled-pore glass, silica gel, polystyrene, PEG-polystyrene, polystyrene optionally cross-linked with divinylbenzene, grafted co-poly, poly-acrylamide, latex, polydimethylacrylamide optionally cross-linked with N,N'-bis-acryloyl ethylene diamine, glass coated with a polymer, or low molecular weight non-cross-linked polystyrene and the particle is a spheroid, a capillary, a hollow fiber, a needle, or a solid fiber.
- the template is covalently linked to polystyrene, polydimethylacrylamide or PEG-polystyrene particles.
- the solid support is further linked to an identifier which uniquely identifies the synthetic receptor covalently linked to the solid support.
- the invention provides methods to prepare libraries comprising a plurality of distinct synthetic receptors, where the libraries comprise at least 100 distinct synthetic receptors.
- the libraries comprise at least 10 3 distinct synthetic receptors and preferably as many as 10 6 or 10 9 distinct synthetic receptor members.
- the receptor library contains at least 10 6 members.
- the libraries may have synthetic receptors where the polyfunctional organic template is an acyclic hydrocarbon, a monocyclic aliphatic hydrocarbon, a polycyclic aliphatic hydrocarbon, a monocyclic aromatic hydrocarbon, a polycyclic aromatic hydrocarbon, a monocyclic heterocycle, a polycyclic heterocycle, a macrocycle or a steroid.
- the synthetic receptors of the libraries are covalently linked to a solid support.
- the libraries may have at least 100 unique solid supports. The solid support is linked to an identifier or identifiers which uniquely define the synthetic receptors.
- the polyfunctional organic template is further linked to an identifier or identifiers.
- the invention provides a method of preparing a synthetic receptor library with identifiers comprising a plurality of different synthetic receptor members.
- Each synthetic receptor library member may be a solid support having a single type of synthetic receptor attached. The method having the following steps: a) reacting the solid supports in a reaction vessel with a polyfunctional organic template; b) apportioning the solid supports with the attached polyfunctional organic template among a plurality of reaction vessels; c) reacting the polyfunctional organic template on a solid support in each reaction vessel with a first oligomer monomer;
- identifier molecules or combinations thereof used for the methods described herein are preferably different for each iteration.
- the invention also provides a method comprising the steps of: a) preparing a bifunctional solid support containing a first type of active site blocked with a first type of protecting group and a second type of active site blocked with a second type of protecting group; b) reacting the solid support with an activator to remove the first type of protecting group thereby exposing the first type of active site; c) coupling a protected polyfunctional organic template to the first type of active site;
- the invention also provides the method comprising the steps of: a) coupling a protected polyfunctional organic template to a solid support; b) reacting the protected polyfunctional organic template with an activator to remove the protecting group thereby exposing the active site; c) coupling a protected oligomer monomer to the deprotected polyfunctional organic template; d) coupling an identifier to the solid support; and e) repeating steps (b) through (d) from one to twenty times for each oligomer of the synthetic receptor.
- steps (c) and (d) may be further combined with pooling and apportioning steps so as to practice split synthesis as outlined in figure 2.
- Another embodiment of the invention is a method of preparing a synthetic receptor with one or more identifiers which comprises a polyfunctional organic template covalently linked to two or more oligomers which may independently be the same or different and which may independently be straight chain, cyclic or branched and one or more identifiers attached to the solid support which define the molecular structure of the oligomers of the synthetic receptor, the method comprising the steps of: a) coupling a protected polyfunctional organic template to a solid support; b) reacting the protected polyfunctional organic template with an activator to remove the protecting group thereby exposing the active site; c) coupling a protected oligomer monomer to the deprotected polyfunctional organic template; d) coupling an identifier to the solid support; and e) repeating steps (b) through (d) from one to twenty times for each oligomer of the synthetic receptor.
- the invention also provides a method of preparing a synthetic receptor library with identifiers comprises a plurality of different synthetic receptor members, wherein each synthetic receptor library member comprising a solid support having attached thereto a single type of synthetic receptor which comprises a polyfunctional organic template covalently linked to two or more oligomers which may be independently the same or different and which may independently be straight chain, cyclic or branched and one or more identifiers which define the synthetic receptor, the method comprising the steps of: a) apportioning the solid supports among a plurality of reaction vessels; b) reacting the solid supports in each reaction vessel with a first oligomer monomer; c) reacting the solid supports in each reaction vessel with a first identifier; d) pooling the solid supports; e) apportioning the pooled supports among a plurality of reaction vessels; f) reacting solid supports in each reaction vessel with a second oligomer monomer; g) reacting the pooled solid supports in each reaction vessel with a
- a method of preparing a synthetic receptor with one or more identifiers which comprises a polyfunctional organic template covalently linked to two or more oligomers which may independently be the same or different and which may independently be straight chain, cyclic or branched and one or more identifiers attached to the solid support which define the molecular structure of the oligomers of the synthetic receptor, the method comprising the steps of: a) prepare a multifunctional solid support containing a active sites blocked with protecting groups; b) reacting the solid support with an activator to remove a first type of protecting group thereby exposing a first type of active site; c) coupling a protected oligomer monomer to the first type of active site of the solid support; d) react the solid support with an activator to remove a second type of protecting group thereby exposing a second type of active site; e) coupling a protected identifier to the second type of active site; and f) repeating steps (b) through (e) from one to twenty times for each oligo
- the identifier is a stable chemical molecule or a plurality of stable chemical molecules distinguishable and detectable to picomolar levels or an oligonucleotide.
- the solid supports coupled to the polyfunctional organic templates in each reaction vessel may first be reacted with an identifier and then the polyfunctional organic templates on the solid supports may be reacted with an oligomer monomer.
- the invention also provides a method for assaying a synthetic receptor library to determine a suitable synthetic receptor for an acceptor molecule of interest, the method comprising the steps: a) generating a synthetic receptor library; b) contacting the synthetic receptor library with the acceptor molecule of interest under conditions such that the acceptor molecule interacts and binds to one or more suitable synthetic receptors of the synthetic receptor library; c) isolating the suitable synthetic receptor(s) that exhibit binding to the acceptor molecule; and d) determining the molecular structure of the suitable synthetic receptor(s).
- the acceptor molecule introduced for the assay may be linked to a label.
- the label attached to the acceptor molecule introduced identifies the suitable synthetic receptor(s) interacting with the acceptor molecule.
- the label may be a dye, a fluorescent label or a radioactive label.
- the suitable synthetic receptor(s) identified in the assay may be used as a chromatographic separation agent.
- the acceptor molecule of interest may be selected from the following, an antibody, a peptide, a protein, a carbohydrate, a nucleic acid, a lipid, a drug, a metal or a small molecule.
- the peptide may be an adrenocorticotropic hormone and fragments, angiotensin, atrial natriuretic, bradykinin, chemotatic, dynorphin, endorphins and beta-lipotropin fragments, enkephalin, enzyme inhibitors, fibronectin fragments, gastrointestinal, growth hormone releasing peptides, luteinizing hormone releasing peptide, melanocyte stimulating hormone, neurotensin, opioid, oxytocin, vasopressin, vasotocin, parathyroid hormone and fragments, protein kinase, somatostatin, substance P.
- the protein acceptor molecule is a growth hormone it may be selected from the group comprising human, bovine, porcine, avian, ovine, piscine, or equine growth hormone, and polypeptide analogs thereof having the biological activity of the corresponding naturally occurring growth hormone.
- the protein which is a growth factor may be IL-1, IL-2, IL-3, IL-4, IL-5, IL-6, IL-7, IL-8, IL-9, IL-10, IL-11, IL-12, IL-13, IL-14, EGF, aFGF, bFGF, TGF-betal, TGF-beta2, TGF-beta3, G-CSF, GM-CSF, M-CSF, EGF, IGF-I, IFN, IL, LIF, KGF, OSM, PDGF, TNF, cytokines, kit ligand, EPO, transforming growth factor, nerve growth factor, brain derived growth factor, neurotrophin-3, neurotrophin-4, heptaocyte growth factor.
- the protein may also be a receptor for any of the above peptides, or growth factors.
- the acceptor molecule may also be present on whole cells, viruses or bacteria.
- Other acceptor molecules include molecules on a cancer cell associated with the following cancers, melanoma, lip, tongue, mouth, pharynx, esophagus, stomach, small intestine, colon, rectal, liver, pancreas, larynx, lung, bone, connective tissue, skin, breast, uterus, ovary, prostate, testis, bladder, kidney, eye, brain, central nervous system, endocrine glands, blood and lymph tissues or leukemia.
- the invention is also directed to a synthetic receptor selectively binding an acceptor molecule in the presence of other different acceptor molecules.
- the invention further provides a method for assaying a synthetic receptor library for suitable synthetic receptor(s) that exhibit biological activity, the method comprising the steps of: a) generating a synthetic receptor library; b) detecting the biological activity of suitable synthetic receptors of the synthetic receptor library in situ; c) isolating the suitable synthetic receptor(s) that exhibit the biological activity; and d) determining the molecular structure of the suitable synthetic receptor(s) isolated in step (c).
- the biological activity of interest is, e.g. cytotoxicity, antitumor activity, antibacterial activity, antiviral activity, antifungal activity, anti-parasite activity, growth factor activity, growth inhibitory activity, hormone activity, neurotransmitter activity, immunomodulator activity, regulatory activity or enzymatic activity.
- the activity of interest is determined at nanomolar concentrations and the synthetic receptor(s) detected may be of use as a therapeutic agents or as a diagnostic agents.
- a suitable synthetic receptor may selectively bind to a transition state analogue.
- Another embodiment of the invention includes a method for assaying a synthetic receptor library for a suitable synthetic receptor(s) which catalyzes a reaction, the method comprising the steps of: a) generating a synthetic receptor library; b) introducing to the synthetic receptor library a substrate such that a catalyzed reaction product is determined; c) isolating the suitable synthetic receptor(s) that exhibits catalytic activity; and d) determining the molecular structure the suitable synthetic receptor(s) isolated in step (c).
- the invention provides a method for assaying a synthetic receptor library for a suitable synthetic receptor(s) which inhibits an enzyme-catalyzed reaction, the method comprising the steps of: a) generating a synthetic receptor library; b) introducing to the synthetic receptor library an enzyme that catalyzes a reaction of interest in situ; c) detecting inhibition by a suitable synthetic receptor(s) of the enzyme- catalyzed reaction of interest in situ; d) isolating the suitable synthetic receptor(s) that exhibits inhibition of enzyme catalyzed reaction of interest in situ; and e) determining the molecular structure the suitable synthetic receptor(s) isolated in step (c).
- This invention will also be directed to the use of the receptors to detect a drug and in the detection of an illicit drug, i.e., a narcotic, an anabolic steroid.
- This invention includes a method for creating new synthetic receptors and libraries of synthetic receptors which can mimic monoclonal antibodies has been developed.
- the synthetic receptor molecules herein may selectively bind almost any desired substrate. Using combinatorial synthesis large libraries of synthetic receptors can be generated. Once prepared the libraries of synthetic receptors can be used to screen for synthetic receptor members that have a desired characteristic. A library of synthetic receptors is synthesized using combinatorial techniques.
- the synthetic receptor library may be prepared by any of the known methods for combinatorial synthesis (G. Jung and A.G. Beck-Sickinger, Angew. Chem.
- the synthetic receptor may have one of the following structures , where the polyfunctional organic template is
- the synthetic receptors members have the following general formula (Scheme 1):
- Scheme 1 General formula for synthetic receptor members. where L is a polyfunctional organic template which is chemically bonded to V, V', etc. which are oligomers.
- Scheme 1 is a subset of the general formula L-(-V N ) M .
- a synthetic receptor library will consist of a collection of such synthetic receptor molecules (i.e. a library) having a variety of different oligomers V, V', etc. groups.
- P solid support particle as diagrammed below such that any given solid support particle has only one type of synthetic receptor (i.e. one type of synthetic receptor member of the library) bound to it.
- a substrate of interest detectable at nanomolar levels by way of its color, its fluorescence, its radioactivity, etc., may be prepared, such detectable substrates are referred to as labeled substrates.
- the synthetic receptor library is then assayed to find those members of the library which have the desired interaction with the labeled substrate.
- the synthetic receptor library is mixed with a solution of the labeled substrate and those library members that bind to the labeled substrate are selected.
- This procedure is particularly simple when the synthetic receptor library members are bound to a solid support as shown in Scheme 2. In that case, solid support particles having receptors which bind the labeled substrate accumulate color or fluorescence or radioactivity (depending on the nature of the label used).
- the assay can be adjusted to detect binding of any desired strength: for example, if the amount of labeled substrate in the presence of the receptor library is adjusted to give a 100 ⁇ M concentration of free (unbound) labeled substrate, then assay will only detect template-substrate binding with association constants (k) of (100 ⁇ M) - 1 or greater.
- the synthetic receptor library is mixed with a solution of the labeled substrate and those library members are selected which catalyze the conversion of the substrate to a reaction product. Detection of reaction products for synthetic receptor library members having catalytic activity may be determined for example by HPLC (high performance liquid chromatography) analysis. The synthetic receptors showing catalytic activity are then isolated from the library.
- the structure of the synthetic receptor(s) is determined.
- the selected synthetic receptor(s) can be used in affinity chromatography (Eveleigh, J.W. & Levy, D.E. Immunochemical characteristics and preparative application of agarose-based immunoosorbents. J. Solid Biochem. (2) 45-78, 1977).
- the following coupling gels may be used in affinity chromatography: NHS-activated Superose 12, Activate CH Sepharose 4B, CNBr-activated Sepharose 4B, ECH Sepharose 4B, Epoxy-activated Sepharose 6B, EAH Sepharose 4B, Agarose Adipic Acid Hydrazide, Thiopropyl Sepharose 6B and Activated Thiol Sepharose 4B (Pharmacia LKB Biotechnology Products Catalog 1991, Pharmacia LKB Biotechnology, Piscataway, NJ, pages 7-8).
- Additional coupling gels such as: Hitrap NHS-activated, CNBr-activated Sepharose 4B, Expoxy-activated Sepharose 6B, EAH Sepharose 4B, Agarose Adipic Acid Hydrazide and Thiopropyl Sepharose 6B may be used in affinity chromatography (Atlas of Practical Purification: Chromatography Media to Meet Your Needs, Pharmacia Biotech, pages 24-25, 31).
- the templates (L) will desirably have limited conformational mobility and functionality oriented in such a way that the variable oligomeric chains are directed toward nearby regions of space.
- suitable templates include polyfunctional steroids such as steroidal diols, triols and amines.
- the oligomeric chains are indicated by V or
- R represents any stable organic functionality (and is unimportant to the properties of the template) in the case of the free template library while R represents a functional group with a connection to a solid support (e.g. -(CH 2 ) 2 CONH-polymer) in the case of the solid phase supported library.
- a solid support e.g. -(CH 2 ) 2 CONH-polymer
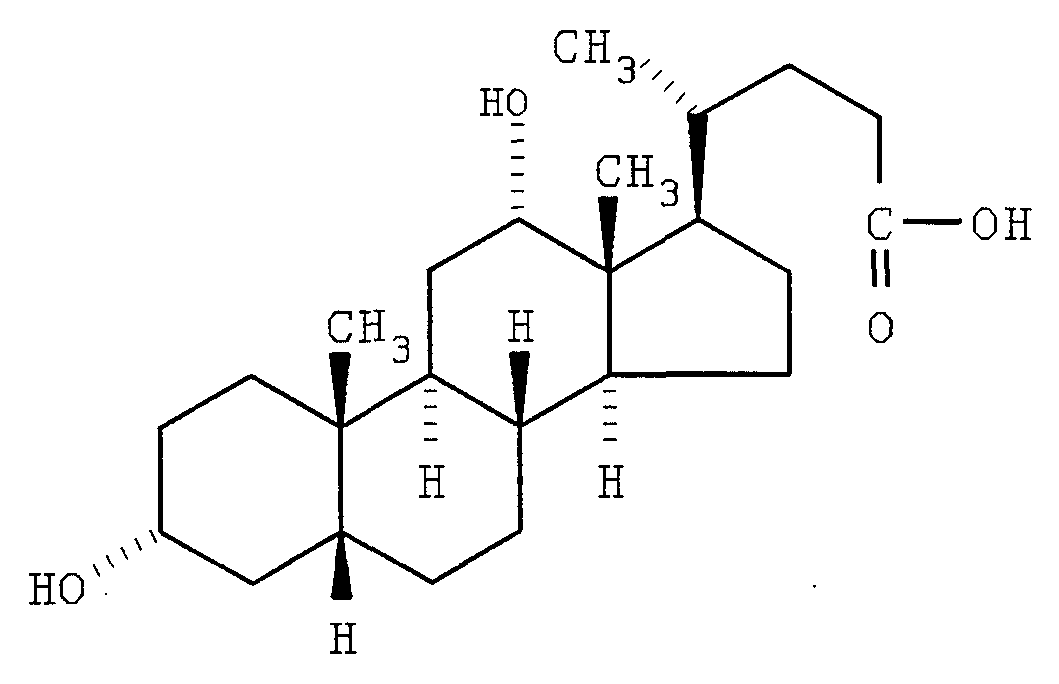
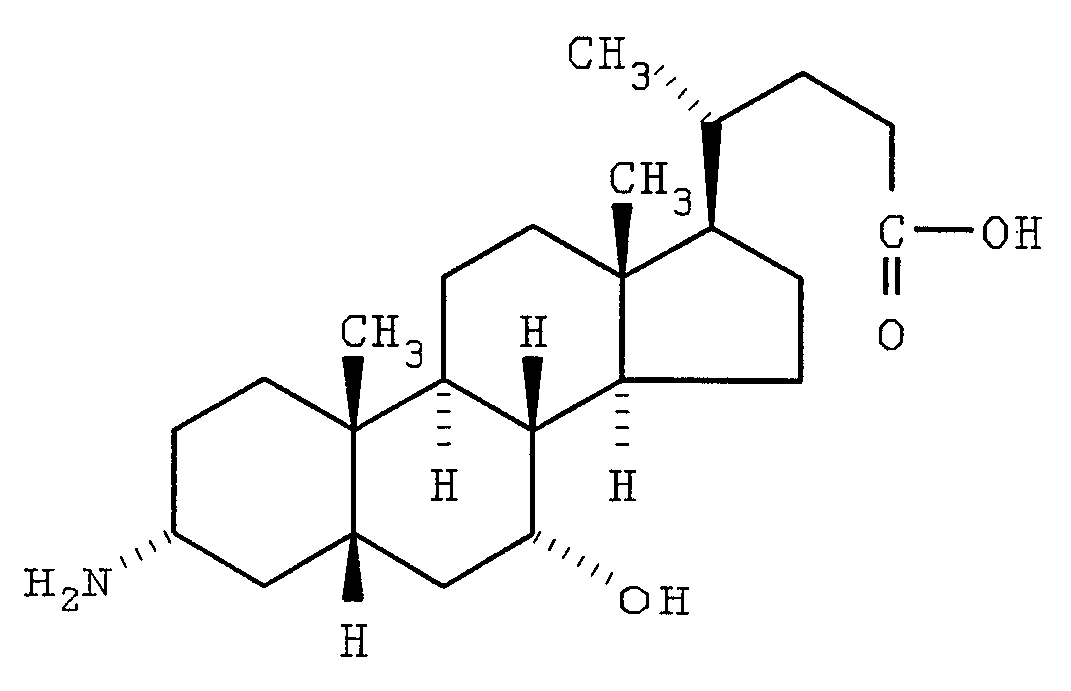
- Cholic acid template (a steroidal triol).
- Cholic acid could form the basis for a triple chained template (this is an example of the LW'V" described above).
- the D-ring side chain served as a convenient appendage with which to bind the template to a solid support.
- an appendage may be absent but may be inserted by adding a trifunctional material to one of the template functional groups. This material would provide two remaining groups which could be used for attaching the solid support (R) and the variable oligomer (V, V', etc).
- R solid support
- V, V', etc variable oligomer
- the effect of having a rigid polyfunctional organic template is well illustrated by the binding observed for Leu Enkephalin shown below.
- templates could also be used to produce synthetic receptor libraries based, for example, on macrocyclic cores. This structure is generally based on a known template (Yoon and Still, J. Am. Chem. Soc. , 115, 823, 1993). In general, templates can include any di-, tri- or tetra-, etc. functionalized organic structure where the functionality allows attachment of the variable oligomeric chains (V, V', etc).
- variable oligomeric chains any oligomer can be used.
- V, V', etc can consist of oligoamides, oligoesters, oligoureas, oligourethanes, oligoamines, oligoethers, oligosulfonamides, oligophosphonamides, oligophosphonates, oligophosphates, etc. as well as mixed oligomers composed of mixtures of the foregoing functionalities.
- the chain can be either linear, cyclic or branched and can incorporate both cyclic and acyclic segments. Branched oligomers will be utilized to generate larger binding sites which should give tighter and more selective substrate binding. Oligomers composed of conformationally rigid fragments or segments are of interest because they help preorganize the template and therefore increase its selectivity.
- the oligomers are polypeptides, cyclophanes or a mixture thereof.
- combinatorial synthesis is a convenient method to generate a receptor library containing a diverse and numerous number of molecules.
- These combinatorial synthetic techniques include, multi-pin method (Geysen, H.M.; Meloen, R. and Barteling, S. Proc. Natl . Acad. Sci . USA, 1984, 81, 3998; Geysen et al., Proc . Natl . Acad. Sci . U. S.A . , 1985, 82, 178;WO 84/03564;; WO 86/06487; PCT/AU85/00165 (WO 86/00991), U.S. Patent 5,133,866), tea-bag method (U.S. Patent No.
- VLSIPS method Fodor, S.P.A.; Read, J.L.; Pirrung, M.C.; Stryer, L.; Lu, A.T. and Solas, D. Science 1991, 251, 767,; U. S. Patent 5,143,854; WO 90/15070; WO 92/10092
- split-synthesis method Li, K.; Salvon, S.; Hersh, E.; Hruby, V.; Kazmierski, W. and Knapp, R. Nature, 1991, 354, 82; WO 92/00091, WO 93/06121.
- the split-synthesis method is superior to the other methods as it offers a procedure involving the systematic synthesis and screening of oligomer libraries of defined structure.
- the procedure for split synthesis involves creating a large oligomer library consisting of thousands to billions of different molecules.
- the molecules may be attached to particles such as beads, with each bead containing a single oligomer sequence and with the collection representing numerous combinations of possible random oligomer sequences.
- the "one-bead, one-oligomer sequence" concept can be achieved easily by separating and mixing beads during the synthesis.
- Figure 2 demonstrates the synthesis of 27 tripeptides comprising alanine (A), glycine (G) and valine (V) by the split-synthesis method.
- each bead has only one product from a specific reaction sequence. Since the amount of products on each bead is normally 50-200 pmol, the structure elucidation of such small amount of products restricts the split-synthesis method to the synthesis of nucleotides or peptides composed of natural amino acids. The split-synthesis method alone fails to provide access to other libraries comprising unnatural monomers.
- readable tags are cosynthesized to encode the series of steps and reagents used in the synthesis of each library element (Brenner, S. and Lerner, R.A. Proc. Natl . Acad. Sci , USA, 1992, 89, 5381; Kerr, J. M.; Banville, S.C. and Zuckermann, R.N. J.Am. Chem. Soc. 1993, 115, 2529).
- a library element is selected by certain assay, its structure can be identified by sequencing its peptide tag or oligonucleotide tag after PCR amplification.
- the main problem with the above encoding methods is the at the tagging structures are chemically liable and destroyed by many of the reagents and conditions normally associated with the synthetic organic chemistry. Furthermore, the oligonucleotide or peptide tags may themselves associate with biological receptors and confuse binding or enzymatic assays. Recently, an alternative encoding method was developed using molecular tags (M.J.H. Ohlmeyer, R.N. Swanson, L.W. Dillard, J.C. Reader, G. Asouline, R. Kobayashi, M. Wigler and W.C. Still, (1993) Proc . Na tl . Acad . Sci . USA, 90, 10922-10926; WO 94/08051). This technique makes use of highly sensitive, chemically inert molecular tags and a binary encoding scheme. This method provides a practical solution for the construction of large, chemically diverse libraries.
- the binary encoding scheme allows the encoding of the maximum amount of information by using the minimum number of tag molecules. For example, a simple combinatorial synthesis using any of 7 different reagents in each step is carried out. These seven different reagents can be designated by three tag molecules through the binary encoding scheme as tag 1 for reagent, tag 2 for reagent 2 , tag 1 and tag 2 for reagent 3 , . . .. . , tag 1 , tag 2 and tag 3 for reagent 7. This letter description can be simply translated into binary numerical description as 001 (reagent 1), 010 (reagent 2), 011 (reagent 3), . . ... . , 111 (reagent 7) .
- the binary encoding scheme only requires 3N tag molecules to encode 7 N different final products in the library, where N is the number of chemical steps for synthesis of the library.
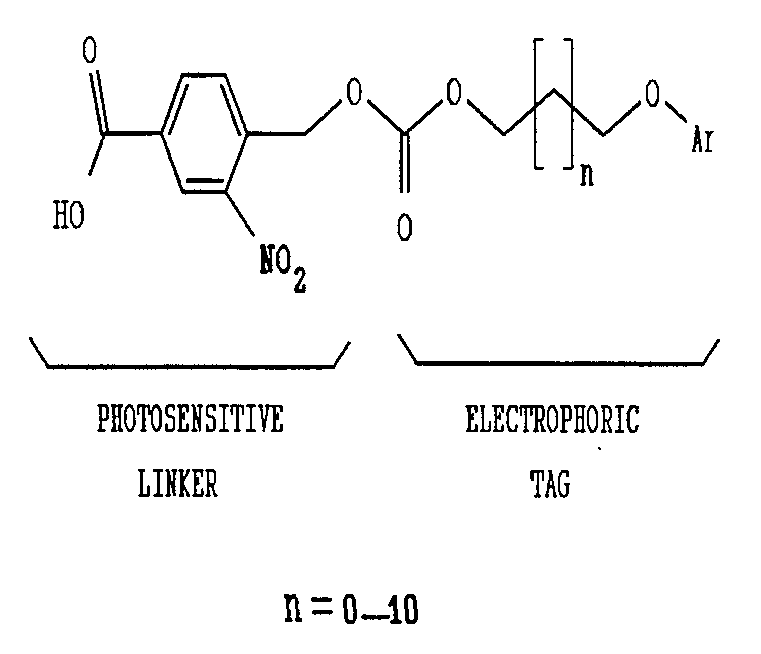
- the tag molecules used are shown below.
- the electrophoric tags can be easily liberated from solid-supports by irradiation with light of wavelength longer than 320nm.
- the liberated alcohols are then silylated by N,O-bis (trimethylsilyl) acetamide in N,N-Dimethylformamide.
- the resulting silyl ethers are well separated by capillary GC and selectively detected by EC electron capture) at levels ⁇ 1 pmol.
- More than 20 tagging molecules were prepared which allow encoding of up to 2 20 different synthesis.
- two different receptor libraries composed of 10,000-20,000 receptor members have been prepared.
- color screening with a labeled substrate vide infra
- synthetic antibodies which is conceptually represented below.
- a binding constant with free energy of formation ⁇ G to the acceptor of 5 Kcal/mol to 12 Kcal/mol is desirable, in some cases the preferred free energy of formation ⁇ G may be 9 Kcal/mol to 12 Kcal/mol and in other cases a ⁇ G of 8 Kcal/mol to 15 Kcal/mol is desirable.
- the single-armed peptidosteroidal receptor library was prepared as shown above. Ten different amino acids at AA1 and AA2 were used resulting in 100 different receptors. When the library was treated with Disperse Red-dyed Leu Enkephalin no binding was found.
- UVP Ultraviolet lamp at 366 nm.
- Aminomethyl resin was purchased from Balchem, Inc. (200-400 mesh, 0.6 mmol/g) or Sigma, Inc. (100-200 mesh, 1.1 mmol/g 18 ).
- the tag molecules used for encoding the synthesis of the libraries are shown in Scheme 3. They are simply named as C n , 2,4,5 Cl 3 ; C n , 2,4,6 Cl 3 and C n , Cl 5 stand for 2,4,5 trichloro phenyl, 2,4,6, trichloro phenyl and pentachloro phenyl groups respectively.
- Amino acids used for the synthesis of the following libraries were Alanine, Valine, Leucine, Phenylalanine, Proline, Serine, Threonine, Lysine, Aspartic acid and Glutamic acid.
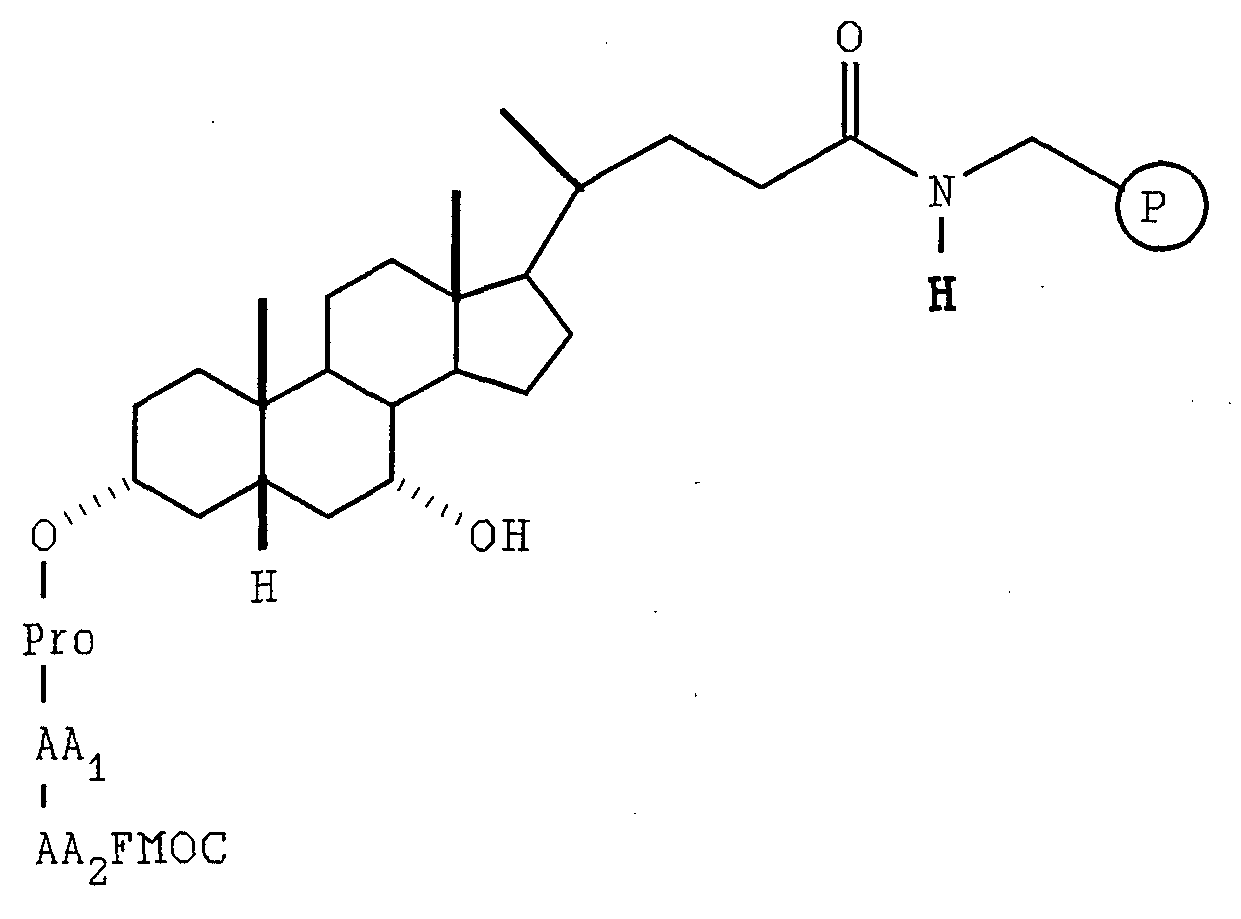
- the Gly-library was constructed on aminomethyl resin (200-400 mesh, 0.6 mmol/g) which was purchased from Bachem, Inc.
- the Pro-library was constructed on aminomethyl resin (100-200 mesh, 0.9 mmol/g) which was purchased from Sigma, Inc. LIST OF ABBREVIATIONS
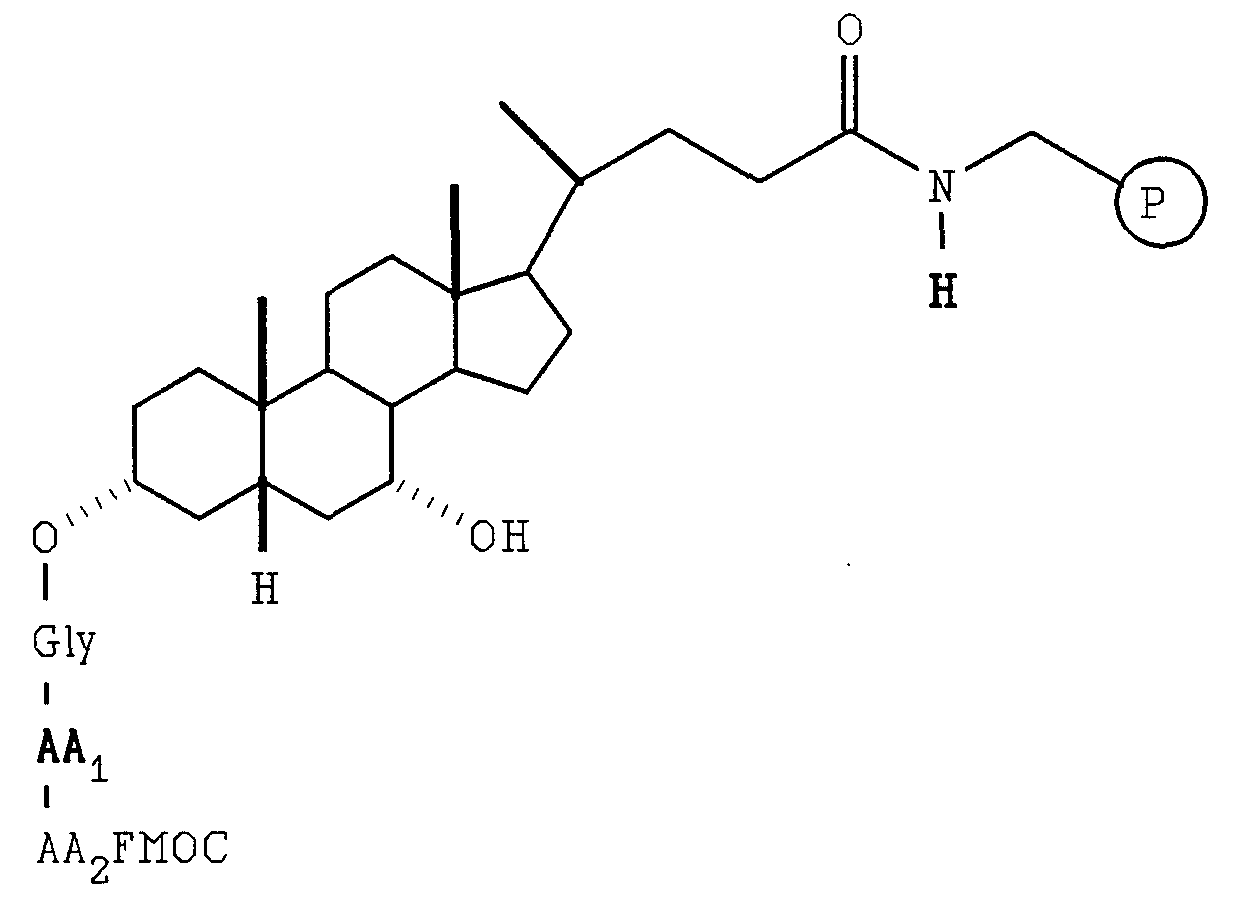
- the resulting beads were retreated with diisopropylethyl amine and 9-fluorenylmethyloxycarbonyl-GlyF in N,N-dimethylformamide twice under the same conditions and then washed with 3 ⁇ N,N-dimethylformamide to give C 3 -OGly-9-fluorenylmethyloxycarbonyl, C 7 -OH beads.
- N,N-dimethylformamide/piperidine (5 mL).
- the reaction vessel was shaken at room temperature for 30 min, and then the beads were washed with 4 ⁇ N,N-dimethylformamide followed by the treatment of HOAc (27 ⁇ L, 0.45 mmol), 1-hydroxybenzotriazole (68 mg, 0.5 mmol) and DIC (70 ⁇ L, 0.45 mmol) in 2 mL N,N-dimethylformamide.
- HOAc 27 ⁇ L, 0.45 mmol
- 1-hydroxybenzotriazole 68 mg, 0.5 mmol
- DIC 70 ⁇ L, 0.45 mmol
- the resulting beads were retreated with diisopropylethyl amine and 9-fluroenylmethyloxycarbonyl-GlyF in N,N- dimethylformamide twice under the same conditions and then washed with 3 ⁇ N,N-dimethylformamide to give C 3 -OGly-9-fluorenylmethyloxycarbonyl, C 7 -OH beads.
- the above beads were again treated with 1:1 N,N-dimethylformamide/piperidine (5 mL) to remove the 9-Fluorenylmethyloxycarbonyl-protecting group.
- reaction vessel was shaken at room temperature for 30 min, and the beads were washed with 4 ⁇ N,N-dimethylformamide, and the treated with 9- Fluorenylmethyloxycarbonyl-Phenylalanine (97 mg, 0.25 mmol), 1-hydroxybenzotriazole (35 mg, 0.25 mmol) and DIC
- N,N-dimethylformamide/piperidine 5 mL was added 1:1 N,N-dimethylformamide/piperidine (5 mL). After under shaking at room temperature for 30 min, the beads were washed with 4 ⁇ N,N-dimethylformamide and treated with HOAc (27 ⁇ L, 0.45 mmol), 1-hydroxybenzotriazole (35 mg, 0.25 mmol) and DIC (70 ⁇ L, 0.45 mmol) in 2 mL N,N-dimethylformamide. The reaction mixtures were under shaking for 2 h until the Kaiser test indicated the completion of the reaction.
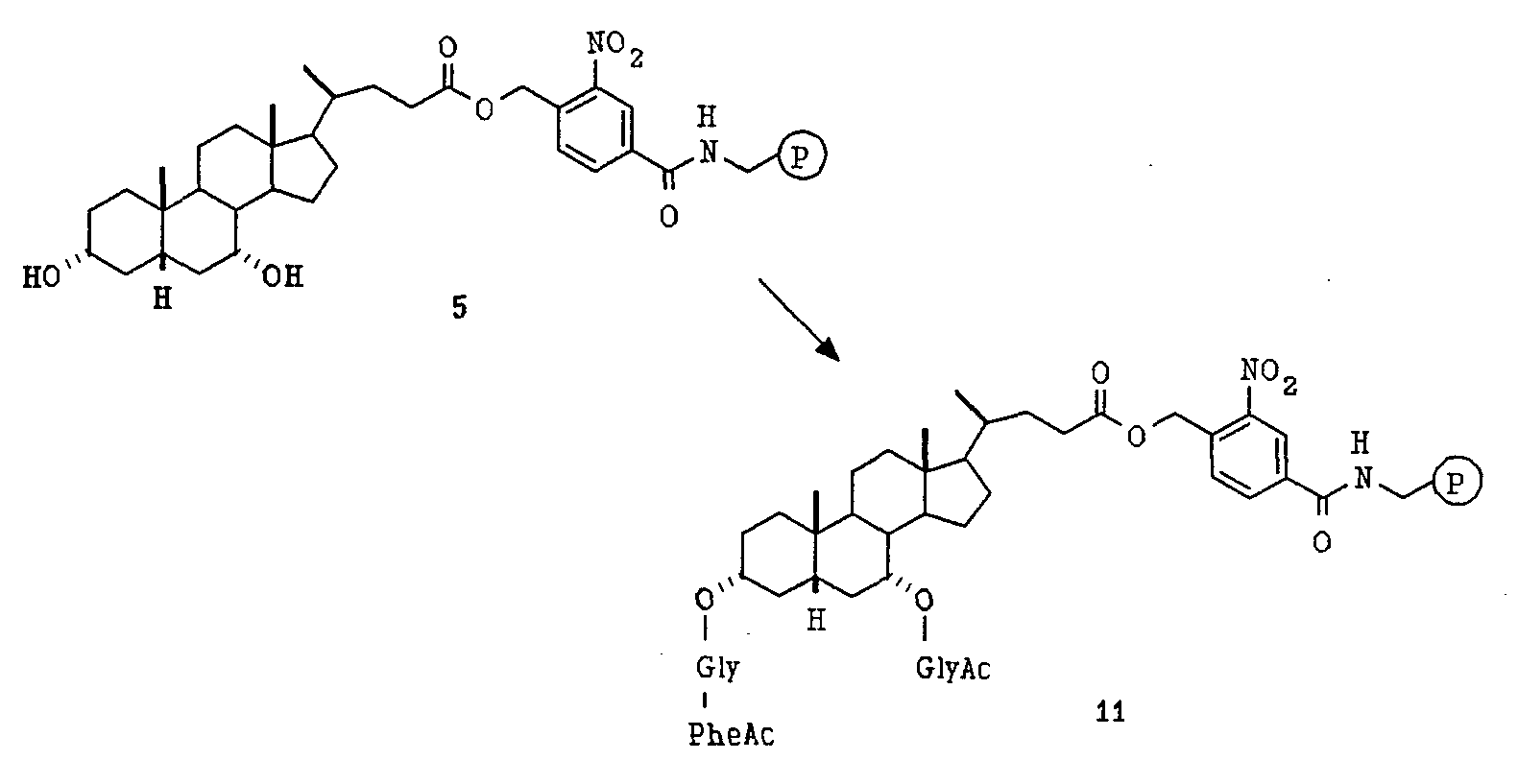
- the resulting beads were retreated with diisopropylethyl amine, 4-Dimethylaminopyridine and 9-Fluorenylmethyloxycarbonyl-GlyF in N,N-dimethylformamide twice under the same conditions and then washed with 3 ⁇ N,N-dimethylformamide (5 mL, 2 min each), 3 ⁇ i-PrOH (5 mL, 2 min each) and 5 CH 2 Cl 2 (5 mL, 2 min each), and then dried on pump to afford beads 11.
- the resulting beads were retreated with diisopropylethyl amine and 9-Fluorenylmethyloxycarbonyl twice under the same conditions, and then washed with 3 ⁇ N,N-dimethylformamide, 2 ⁇ i-PrOH, 5 ⁇ CH 2 Cl 2 , and dried on pump to give the C 3 -OGly9-Fluorenylmethyloxycarbonyl, C 7 -OH beads (4.2 g, slightly yellow beads).
- T 4 ,C 5 ,2,4,6 Cl 3 0.9 mg 4 T 1 , T 2 T 1 ,0.9 mg;T 2 , 0.9 mg 5 T 1 , T 3 T 1 ,0.9 mg;T 3 , 0.9 mg 6 T 1 , T 4 T 1 ,0.9 mg;T 4 , 0.9 mg 7
- the second step AA 2 was initiated by the division of the above C 3 -OGlyAA 1 FMOC, C 7 -OH beads into ten equal portions.
- the labelling and coupling conditions were exactly same as the first AA 1 .
- the tag molecules used to encode the second step synthesis are listed below:
- T 5 , T 6 T 5 1.0 mg; T 6 , 1.0 mg 5 T 5 , T 7 T 5 , 1.0 mg; T 7 , 1.0 mg 6 T 5 , T 8 T 5 , 1.0 mg ; T 8 , 1.0 mg 7 T 6 , T 7 T 6 , 1.0 mg; T 7 , 1.0 mg 8 T 6 , T 8 T 6 , 1.0 mg; T 8 , 1.0 mg 9
- the third step AA 3 was also initiated by the division of the above C 3 -OGlyAA 1 AA 2 Ac, C 7 -OGlyFMOC beads into ten equal portions.
- the labelling and coupling conditions were exactly the same as the first steps.
- the tag molecule used to encode the third step synthesis are listed below:
- T 9 , C 8 , 2,4,5 Cl 3 1.0 mg 1 T 10 , C 8 , 2,4,6 Cl 3 1.0 mg 2 T 11 , C 9 , 2,4,5 Cl 3 1.1 mg 3 T 12 , C 9 , 2,4,6 Cl 3 1.1 mg 4
- T 9 , T 10 T 9 1.0 mg; T 10 , 1.0 mg 5 T 9 , T 11 T 9 , 1.0 mg; T 11 , 1.1 mg 6 T 9 , T 12 T 9 , 1.0 mg; T 12 , 1.1 mg 7 T 10 , T 11 T 10 , 1.0 mg; T 11 , 1.1 mg 8 T 10 , T 12 T 10 , 1.0 mg; T 12 , 1.1 mg 9 T 11 , T 12 T 11 , 1.1 mg; T 12 , 1.1 mg 10
- the C 3 -OGlyAA 1 AA 2 Ac, C 7 -OGlyAA 3 FMOC beads were obtained.
- the labelling and coupling conditions of the fourth step AA 4 were exactly the same as that of the previous steps.
- the Ca-OGlyAA 1 AA 2 Ac, C 7 OGlyAA 3 AA 4 FMOC were obtained.
- the tag molecules used for this step are listed below:
- the beads were then thoroughly washed with 2 ⁇ DMF (30 mL, 2 min each), 3 ⁇ i-PrOH (30 mL, 2 min each) and 5 ⁇ CH 2 Cl 2 (30 mL, 2 min each), and then dried to give the protected Gly-library.
- the beads 13 (25 mg) and 500 ⁇ L CH 3 OH in a capped 1 mL test tube were under UV light for 12 h. The beads were filtered and the filtrate was concentrated to give 14 as a white solid.
- the NMR spectrum of 14 indicates that the selectivity for esterification of C 3 -OH and C 7 -OH is 25:1.
- the beads 15 (25 mg) and 500 ⁇ L CH 3 OH in a capped 1 mL test tube were under UV light for 12 h. The beads were filtered, and the filtrate was concentrated to give 16 as a white solid.
- the NMR spectrum of 16 indicates that the selectivity for esterification of C 3 -OH and C 7 -OH is 20:1.
- the beads in the reaction vessel were washed with 2 ⁇ DMF (30 mL, 2 min each), 3 ⁇ i-PrOH (30 mL, 2 min each), 5 ⁇ CH 2 Cl 2 (30 mL, 2 min each) and dried to give the chenodeoxycholic acid beads (2.7 g).
- reaction vessels were labeled as 1,2, ⁇ , 10. Each portion of the beads was treated with 1:1 DMF/piperidine
- T 2 , T 4 T 2 0.7 mg; T 4 , 0.6 mg 2
- T 2 , T 3 , T 4 T 2 0.7 mg; T 3 , 0.6 mg; T 4 , 0.6 mg 5
- T 2 , T 3 , T 5 T 2 0.7 mg; T 3 , 0.6 mg; T 5 , 0.6 mg 6
- T 2 , T 3 , T 6 T 2 0.7 mg; T 3 , 0.6 mg; T 6 , 0.6 mg 7
- T 2 , T 4 , T 5 T 2 0.7 mg; T 4 , 0.6 mg; T 5 , 0.6 mg 8
- T 2 , T 4 , T 6 T 2 0.7 mg; T 4 , 0.6 mg; T 6 , 0.6 mg 9
- the tag molecules used for this step synthesis are: T 1 , C 12 , Cl 5 T 2 , C 11 , Cl 5 T 3 , C 11 , 2,4,5, Cl 3
- the second step AA 2 was initiated by the division of the above C 3 -O-DL-ProAA 1 FMOC, C 7 -OH beads into ten equal portions. Each portion of the beads was treated with 1:1 DMF/piperidine (10 mL) at room temperature for 30 min to remove FMOC protecting group. After washing the beads with 4 ⁇ DMF, the tag molecules (1.8 ⁇ 10 -3 mmol), HOBT (13 mg, 0.09 mmol, 50 El.) and DIC (14 ⁇ L, 0.09 mmol, 50 eq.) in 4 mL DMF were added to each reaction vessel in the following manner:
- Tag Molecules Amount of Tags Reaction vessel T 7 , C 9 , 2,4,5 Cl 3 1.0 mg 1 T 8 , C 9 , 2,4,6 Cl 3 1.0 mg 2 T 9 , C 10 , 2,4,5 Cl 3 1.0 mg 3 T 10 , C 10 , 2,4,6 Cl 3 1.0 mg 4
- the third step AA 3 was also initiated by the division of the above C 3 -OProAA 1 AA 2 Ac, C 7 -OGlyFMOC beads into ten equal portions.
- the labelling and coupling conditions were exactly the same as the second steps.
- the tag molecules used to encode the third step synthesis are listed below:
- Tag molecules Amount of Tags Reaction vessel T 11 , C 7 , 2,4,5 Cl 3 1.0 mg 1 T 12 , C 7 , 2,4,6 Cl 3 1.0 mg 2 T 13 , C 8 , 2,4,5 Cl 3 1.0 mg 3 T 14 , C 8 , 2,4,6 Cl 3 1.0 mg 4 T 11 , T 12 T 11 , 1.0 mg; T 12 , 1.0 mg 5 T 11 , T 13 T 11 , 1.0 mg; T 13 , 1.0 mg 6 T 11 , T 14 T 11 , 1.0 mg; T 14 , 1.0 mg 7
- the tag molecules used for this step are listed below:
- T 16 , C 5 , 2,4,6 Cl 3 1.0 mg 2 T 17 , C 6 , 2,4,5 Cl 3 1.0 mg 3 T 18 , C 6 , 2,4,6 Cl 3 1.0 mg 4 T 15 , T 16 T 15 , 0.9 mg; T 16 , 0.9 mg 5
- C 7 -OGlyAA 3 FMOC beads was added 1:1 N, N-Dimethylformamide/Piperdine (30 mL) to remove the 9-Fluorenylmethloxycarbonyl-group.
- the reaction mixtures were shaken at room temperature for 30 min. and then the beads were washed with 4 ⁇ N, N- Dimethylformamide and treated with Acetic Acid (530 ⁇ L, 9 mmol), 1-hydroxybenzotriazole (1.35 g, 10 mmol) and DIC
- the beads were then thoroughly washed with 2 ⁇ DMF (30 mL, 2 min each), 3 ⁇ i-PrOH (30 mL, 2 min each) and 5 ⁇ CH 2 CI 2 , 3 ⁇ i-PrOH and 5 ⁇ CH 2 CI 2 , and dried to afford the deprotected Pro-Library.
- a color assay method was developed for screening receptor library beads.
- the color dye used is Disperse Red 1 (available from Aldrich Inc.), a neutral diazo dye whose ⁇ max is at 502 nm. Red color in various organic solvents is visible at concentration as low as 100 ⁇ M. The method is shown in Figure 4.
- the bead binding the substrate is stained and can be easily identified in a background of many colorless beads.
- the stained bead is the removed from the library by a microsyringe, and subjected to decode to identify its structure.
- the decoding process involves three operations: a) photolysis of the beads, b) silylation of liberated alcohols, c) injection into GC-EC machine. Once identified, the receptor is then resynthesized on large quantities of beads for confirmatory binding studies.
- Figure 5 shows the process used to read the molecular bar code of typical a bead and in Figure 6 the a typical gas chromatography for a bead is shown. From this spectrum, the structure of the receptor is identified as C 3 -OGLY-leu-phe-Ac, C 7 -OGLY-ala-Lys-Ac.
- the Gly-receptor library was first treated with dyederivated serine 17 shown below, it was found that more than 50% beads stained as orange color. It is believed this nonspecific binding could be reduced either by screening the library with more functionalized and more conformationally defined substrates such as biointeresting peptides or by synthesizing another library whose binding conformation is more rigid.
- the second receptor library (Pro-library) is shown in shown below.
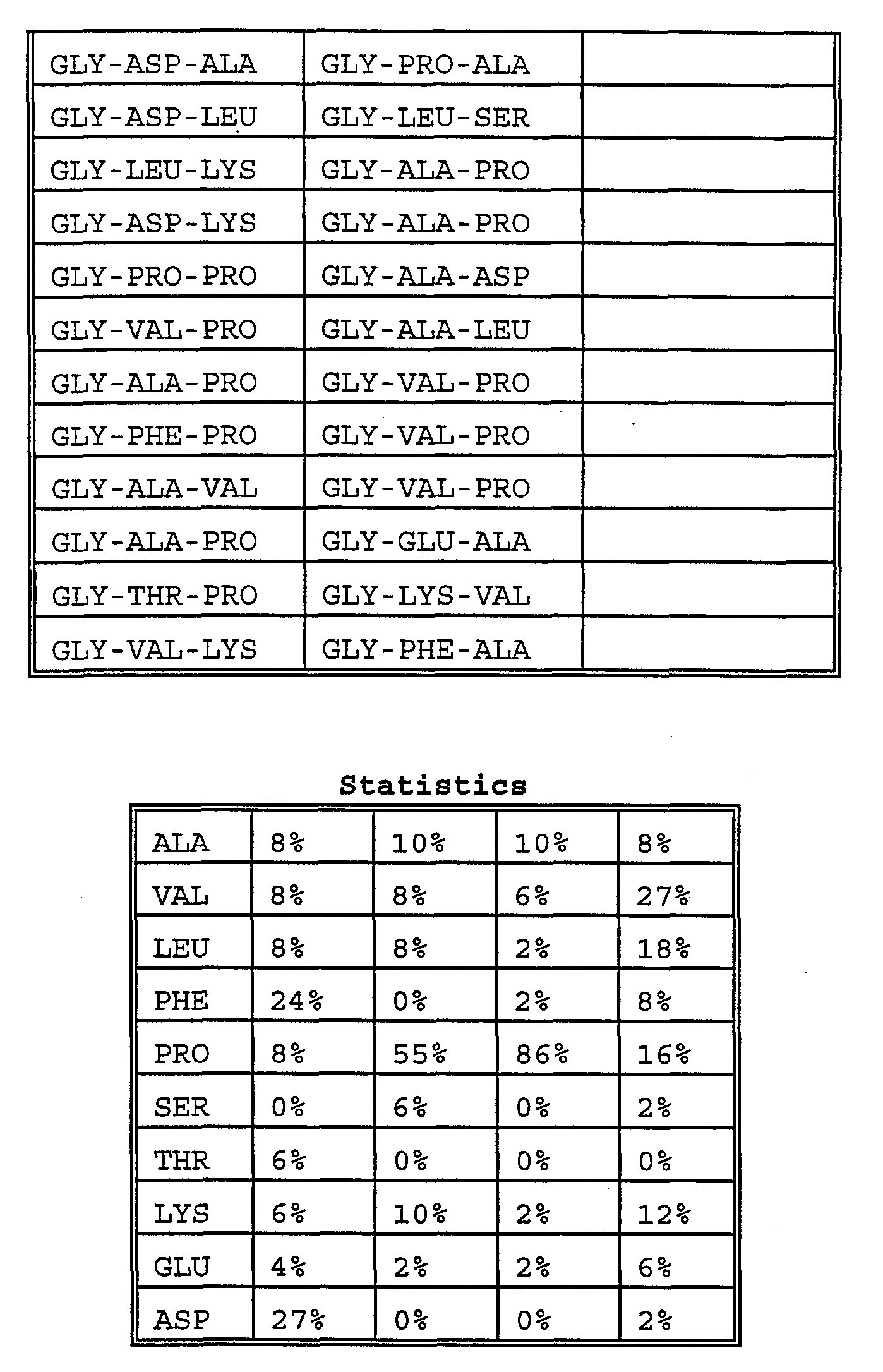
- the Gly-library was found to bind Leucine Enkephalin 18 and Met Enkephalin 19 very selectively.
- Enkephalin is an important neuropeptide (Leu Enkephalin and Met Enkephalin are both commercially available from Bachem, Inc; Fodor, S.P.A.; Read, J.L.; Pirrung, M.C.; Stryer, L.; Lu, A.T. and Solas, D. Science 1991, 251, 767; Hughes, J.; Smith, T.W.; Kosterlitz, H.W.; Fothergill, L.A.; Morgan, B.A. and Morrio, H.R. Nature, 1975, 258, 577).
- proline is the most common amino acid residue in both Leu and Met Enkephalin receptors.
- Proline especially at the second position of C 7 chain, plays an important role in defining the binding conformation of receptors.
- the second most common amino acid residues are different in Leu and Met Enkephalin receptors.
- Leu Enkephalin receptors they are leucine on the C 3 chain and lysine on the C 7 chain, but they are aspartic acid on the C 3 chain and valine on the C 7 chain in Met Enkephalin receptors .
- Another striking finding is that not one of the receptors against Leu Enkephalin overlaps with those found to recognize Met Enkephalin. This observation implies that the receptors selected by screening may have selectivity to their own ligand.
- the Pro-library was screened by Leu Enkephalin 18 and Met Enkephalin 19. There were about 0.75% beads stained by Leu Enkephalin and 0.4% by Met Enkephalin.
- Proline-library Aspartic acid 0% 0% 0% 0% 0% 0% In contrast to the Gly-library, the most common residue in Proline-library is alanine at the third position on C 7 chain. Proline becomes one of the least common residues in Leu Enkephalin receptors. Again Leu and Met Enkephalin receptors do not have a common sequence.
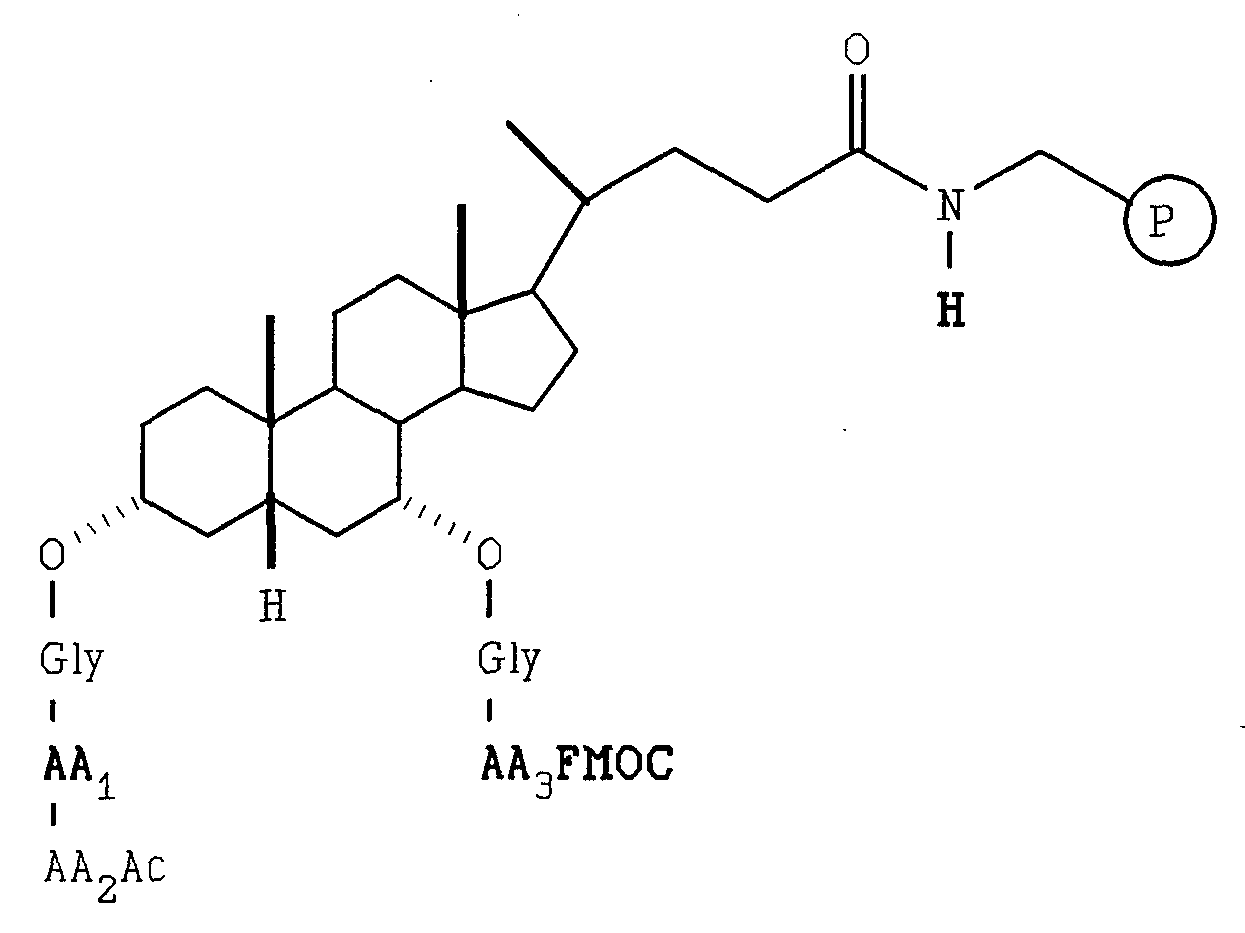
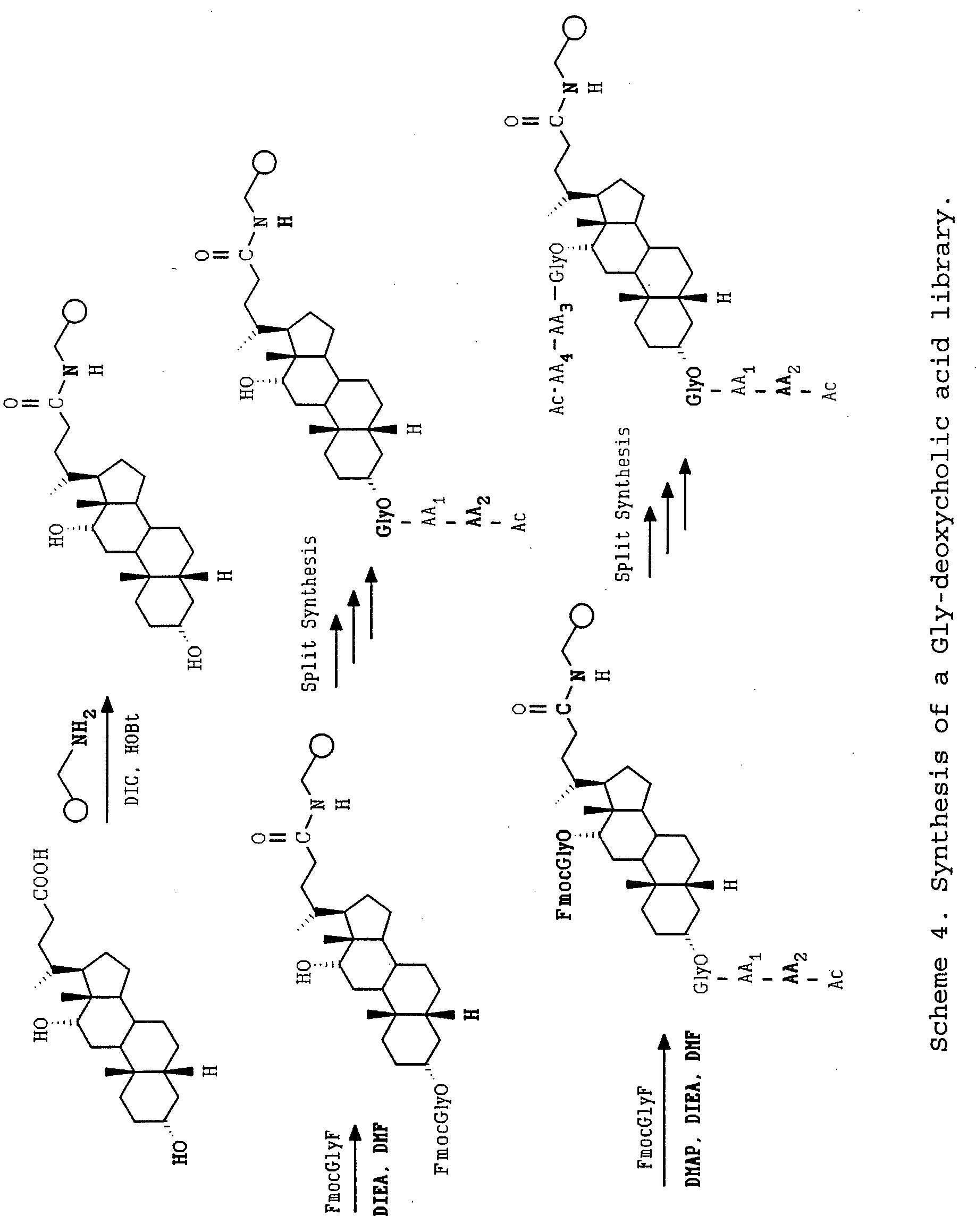
- a Gly-deoxycholic acid library was prepared by essentially the same procedure as described for the chenodeoxycholic acid library except deoxycholic acid was used in place of chenodeoxycholic acid (Scheme 4).
- deoxycholic acid was attached to the aminomethyl resin after activation with DIC.
- the 3-hydroxyl group was then reacted with Fmoc-Gly F.
- Fmoc deprotection two amino acids were added in two split synthesis steps using ten amino acids with each step performed in ten separate reaction vessels. Each step was encoded as described for the chenodeoxycholic acid example.
- the amino acids used were Ala, Val, Leu, Phe, Pro, Ser, Thr, Lys, Glu, Asp.
- Fmoc-GlyF was then allowed to react with the C-ring hydroxyl group as described for the C 7- hydroxy in the chenodeoxycholic acid example. After Fmoc deprotection the same ten amino acids were introduced in two encoded split synthesis steps. The resulting C 3 -OGlyAA 1 AA 2 AC, C 11 -OGlyAA 3 AA 1 Ac deoxycholic acid was deprotected as described for the chenodeoxycholic acid library.
- the receptor libraries described below have macrocyclic Oligomer arms' - this makes them conformationally less flexible and results in more selective receptors. Note however, that it is not a requirement that the receptor oligomer arms are macrocyclic.
- macrotricyclic receptors (1) that are simple A 4 B 6 cyclooligomers of trimesic acid (A(OH) 3 ) and 1,2-diamines (e.g. BH 2 , B'H 2 ) have been described .
- 1 binds certain amino acid derivatives containing L-valine or L-phenylglycine residues with enantioselection exceeding 2 kcal/mol and displays a particular affinity for the sidechain-protected tripeptide sequence (D) Asn(N-Tr) - (L) Val- (L)Ser(O-tBu) in CHCl 3 . Because 1 is among the most sequence-selective peptide receptors yet prepared by synthesis, a study was carried out to determine the smallest, readily preparable substructure of 1 that retains selective peptide binding properties.
- peptide binding properties of the related macromonocycles 2 and 3 and macrobicycles 4 and 5 were prepared and surveyed. It was found that highly selective peptide binding properties are not limited to macrotricycles like 1.
- Dye-labeled receptor substructures 2-5 were prepared using the following methodology developed for the synthesis of 1 (Yoon, S.S. and Still, W.C., Tetrahedron, in press.).
- the substrate selectivity with 4 was particularly high - there was a very large color contrast between receptor bound and unbound beads, and only one bead per ⁇ 2500 beads was stained by the dye-linked receptor. This level of selection indicates that only ⁇ 20 members of the entire 50, 000-member peptide library are bound by 4 at ⁇ 40 ⁇ M concentration in CHCl 3 . With 5, there was a greater gradation of color intensities among the stained beads and one bead per -500 was stained. In comparison, macrotricycle 1 has been reported to bind one bead per ⁇ 1000 at a receptor concentration of 20 ⁇ M.
- each of the two-armed receptors 4-6 can be considered to be constructed from three distinct modules (a template and two oligomer arms) that are held together by simple, acyclic amide bonds. As such, receptors of this type should be readily preparable as combinatorial libraries using split synthesis starting from a variety of templates and oligomer arms.
- the libraries are prepared by solid phase split synthesis using the techniques described in the preparation of the above-described peptidosteroidal library but with different starting materials.
- the preparation consists of the following steps: Step 1.
- a set of diverse, different templates having the following general structure is prepared (also see Scheme 1).
- A is a functional group that allows attachment of the template to a support for solid phase synthesis (e.g. Merrifield polystyrene beads)
- B and C are functional groups that allow attachment of the two oligomer arms of the receptors.
- B and C may be the same functional group in which case the two receptor oligomer arms are the same.
- B and C may be different (or may be differentially protected) in which case the two receptor oligomer arms are different and this is a preferred embodiment.
- Step 2 A set of diverse, different oligomer arms having the following general structure is prepared Where B' and C' are functional groups that allow attachment of the oligomer arms to template functional groups B and C respectively.
- the oligomer arms may be acyclic, cyclic or macrocyclic but oligomer arms with the least conformational flexibility are preferred. Oligomer arms with one or more functional groups (other than B' and C') are also preferred (because the functional group may become involved in specific interactions with the substrates ultimately bound by the receptor).
- Step 3 Attach each template from step 1 using functional group A to a different portion of solid phase synthesis support particles (here designated P) (e.g. Merrifield polystyrene beads, poly (ethylene glycol)-polystyrene copolymer beads, polydimethylacrylamide beads) to give:
- P solid phase synthesis support particles
- A' is the chemical moiety that results from attaching A to P.
- Step 4 All of the different portions of the synthesis support particles (P) from Step 3 are mixed and then distributed equally into N different reaction vessels where N is the number of different, first arm units (arm-B') to be added to the P-bound template.
- the template B functional groups are deprotected or activated as necessary to allow reaction with B' and coupled to a different arm-B' in each different reaction vessel to give: where BB is the chemical moiety that results from coupling B and B' and chemically binds template units to the first arm units. (This is the first step of combinatorial, split synthesis).
- the first arm unit may be further modified by chemical reactions after coupling to the template. Such chemical reactions may be by standard synthesis or by combinatorial synthesis (in which case the number of different compounds made will be increased).
- Step 5 All the different portions of the synthesis support particles (P) from Step 4 are mixed and then distributed equally into N' different reaction vessels where N' is the number of different, second arm units (Arm-C') to be added to the template.
- N' is the number of different, second arm units (Arm-C') to be added to the template.
- the template C functional groups are deprotected or activated as necessary to allow reaction with C' and coupled to a different arm-C' in each different reaction vessel to give:
- CC is the chemical moiety that results from coupling C and C' and chemically binds template units to the second arm units.
- the second arm unit can be further modified by chemical reactions after coupling to the template.
- chemical reactions may be by standard synthesis or by combinatorial synthesis (in which case the number of different compounds made will be increased).
- Step 5 one or more additional steps analogous to Step 5 could be carried out to attach three or more oligomer arms to the template.
- the above steps create a library of synthetic receptors on particles that may be screened for valuable properties (selective ligand or substrate binding, catalysis of certain chemical reactions, selective binding to biological molecules, selective binding to supramolecular structures (e.g. cells), etc.).
- valuable properties selective ligand or substrate binding, catalysis of certain chemical reactions, selective binding to biological molecules, selective binding to supramolecular structures (e.g. cells), etc.
- the structure of that receptor can be determined and the receptor resynthesized on large scale.
- the receptor structure determination can be accomplished by direct analysis of the receptor (e.g. mass spectroscopy) or by molecular tag analysis in the case of an encoded library preparation.
- a molecular encoding scheme is employed as used in the peptidosteroidal library synthesis to allow for structure elucidation of the final receptor library members.
- Step 1 The following templates as outlined below are prepared.
- Step 2 The following arm structures have been prepared.
- Step 3 The templates from step 1 will be chemically attached using diisopropylcarbodiimide (DIC) to NH 2 -functionalized TentaGel (poly (ethylene glycol)-polystyrene copolymer) beads (P in the general scheme above).
- DIC diisopropylcarbodiimide
- TentaGel poly (ethylene glycol)-polystyrene copolymer
- the total number of different receptor in the resulting synthetic receptor library is 600 (6 ⁇ 10 ⁇ 10).
- Templates - are conformationally restrained (as the cyclic templates shown below).
- the templates have short chains of atoms (no more than 10) between functional groups used to attach oligomer arms (as the cyclic and acyclic templates shown below).
- the templates have functional groups that allow attachment of more than one arm.
- the receptors having more than two oligomer arms will be at least as selective as the two-armed receptors (see examples of templates suitable for preparation of 3-armed receptors see the following).
- Oligomer arms - are conformationally restrained: and include macrocyclic molecules, polycyclic molecules, branched cyclic molecules, and branched acyclic molecules.
- the oligomer arms have a range of functional groups for specific interaction with bound substrates - substrates, e.g. functional groups that are ionic (e.g. organic ammonium, carboxylate, phosphate, sulfonate, sulfonamide ions) or are hydrogen bond donors or acceptors (e.g.
- ketones, esters, amides, alcohols, ethers, phosphonamides, sulfonamides, sulfoxides, sulfones, imines, amines, amine oxides, heterocycles, halogens may be particularly valuable for promoting specific receptor-substrate binding.
- Trifluoroacetic acid (0.5 ml) was added via syringe to a solution of 200 mg (0.32 mmol) A in 4 ml CH 2 Cl 2 . After 2 hr, all volatile materials were removed at reduced pressure. The oily residue was triturated with diethyl ether to yield a white solid that was isolated by decantation and drying in vacuo .
- Preparation of a polyfunctional organic template that is water-soluble Preparation of a polyfunctional organic template that is water-soluble.
- the alpha hydroxyls at positions 3 and 12 are attached to the oligomer chains and the carboxyl group may be use in linking the synthetic receptor to a solid support.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Zoology (AREA)
- Wood Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Analytical Chemistry (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Immunology (AREA)
- Physics & Mathematics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Measuring Or Testing Involving Enzymes Or Micro-Organisms (AREA)
- Investigating Or Analysing Biological Materials (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
Claims
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/676,143 US7067326B2 (en) | 1994-01-13 | 1995-01-13 | Synthetic receptors, libraries and uses thereof |
| AU21565/95A AU686785B2 (en) | 1994-01-13 | 1995-01-13 | Synthetic receptors, libraries and uses thereof |
| EP95914675A EP0739486A4 (en) | 1994-01-13 | 1995-01-13 | Synthetic receptors, libraries and uses thereof |
| JP7519163A JPH09511486A (en) | 1994-01-13 | 1995-01-13 | Synthetic receptors, libraries and their use |
| US09/041,343 US6797522B1 (en) | 1994-01-13 | 1998-03-12 | Synthetic receptors |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US18162894A | 1994-01-13 | 1994-01-13 | |
| US08/181,628 | 1994-01-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1995019567A1 true WO1995019567A1 (en) | 1995-07-20 |
Family
ID=22665091
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1995/000572 WO1995019567A1 (en) | 1994-01-13 | 1995-01-13 | Synthetic receptors, libraries and uses thereof |
Country Status (7)
| Country | Link |
|---|---|
| US (2) | US7067326B2 (en) |
| EP (1) | EP0739486A4 (en) |
| JP (1) | JPH09511486A (en) |
| AU (1) | AU686785B2 (en) |
| CA (1) | CA2180844A1 (en) |
| WO (1) | WO1995019567A1 (en) |
| ZA (1) | ZA95260B (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997043232A1 (en) * | 1996-05-14 | 1997-11-20 | Hoechst Research & Technology Deutschland Gmbh & Co.Kg | Novel substance library and supramolecular complexes produced therewith |
| WO1998020349A1 (en) * | 1996-11-05 | 1998-05-14 | Isis Pharmaceuticals, Inc. | Complex chemical libraries |
| WO1998046631A1 (en) * | 1997-04-11 | 1998-10-22 | Eli Lilly And Company | Combinatorial libraries of peptidomimetic macrocycles and processes therefor |
| WO1999031124A1 (en) * | 1997-12-12 | 1999-06-24 | Novo Nordisk A/S | Cholic acid-based scaffolds for multidimensional molecular presentation of peptides |
| WO1999044616A1 (en) * | 1998-03-06 | 1999-09-10 | Brigham Young University | Steroid derived antibiotics |
| US5961923A (en) * | 1995-04-25 | 1999-10-05 | Irori | Matrices with memories and uses thereof |
| US6100026A (en) * | 1995-04-25 | 2000-08-08 | Irori | Matrices with memories and uses thereof |
| US6284459B1 (en) | 1995-04-25 | 2001-09-04 | Discovery Partners International | Solid support matrices with memories and combinatorial libraries therefrom |
| US6350738B1 (en) | 1998-03-06 | 2002-02-26 | Brigham Young University | Steroid derived antibiotics |
| EP1384999A1 (en) * | 2001-04-18 | 2004-01-28 | Japan Science and Technology Corporation | Bacteriotoxin adsorbents and method of screening the same |
| US6767904B2 (en) | 1998-03-06 | 2004-07-27 | Bringham Young University | Steroid derived antibiotics |
Families Citing this family (85)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3016496C2 (en) | 1980-04-25 | 1984-06-14 | Mannesmann AG, 4000 Düsseldorf | Process for reducing pollutant emissions during sintering |
| US5800992A (en) | 1989-06-07 | 1998-09-01 | Fodor; Stephen P.A. | Method of detecting nucleic acids |
| DK0834575T3 (en) | 1990-12-06 | 2002-04-02 | Affymetrix Inc A Delaware Corp | Identification of nucleic acids in samples |
| US6797522B1 (en) * | 1994-01-13 | 2004-09-28 | The Trustees Of Columbia University In The City Of New York | Synthetic receptors |
| US6255461B1 (en) * | 1996-04-05 | 2001-07-03 | Klaus Mosbach | Artificial antibodies to corticosteroids prepared by molecular imprinting |
| US6890624B1 (en) * | 2000-04-25 | 2005-05-10 | Nanogram Corporation | Self-assembled structures |
| US6703381B1 (en) * | 1998-08-14 | 2004-03-09 | Nobex Corporation | Methods for delivery therapeutic compounds across the blood-brain barrier |
| CA2386540A1 (en) * | 1999-10-04 | 2001-04-12 | University Of Medicine And Dentistry Of New Jersey | Novel carbamates and ureas |
| KR100860291B1 (en) * | 2001-01-19 | 2008-09-25 | 에게아 바이오사이언시스, 인크. | Computer-directed assembly of a polynucleotide encoding a target polypeptide |
| DE10105038B4 (en) * | 2001-02-05 | 2005-07-07 | Neurotell Ag | Tripeptide derivatives for the treatment of postläsional diseases of the nervous system |
| DE10105041A1 (en) * | 2001-02-05 | 2002-08-14 | Tell Pharm Ag Hergiswil | Tripeptides and tripeptide derivatives for the treatment of neurodegenerative diseases |
| US20070004641A1 (en) * | 2001-05-24 | 2007-01-04 | Neuren Pharmaceuticals Limited | Cognitive enhancement and cognitive therapy using glycyl-L-2-methylprolyl-L-glutamate |
| ES2325983T3 (en) | 2001-05-24 | 2009-09-28 | Neuren Pharmaceuticals Limited | GPE ANALOGS AND PEPTIDOMIMETICS. |
| US7605177B2 (en) * | 2001-05-24 | 2009-10-20 | Neuren Pharmaceuticals Limited | Effects of glycyl-2 methyl prolyl glutamate on neurodegeneration |
| US7714020B2 (en) * | 2001-05-24 | 2010-05-11 | Neuren Pharmaceuticals Limited | Treatment of non-convulsive seizures in brain injury using G-2-methyl-prolyl glutamate |
| WO2002102820A1 (en) | 2001-06-20 | 2002-12-27 | Nuevolution A/S | Nucleoside derivatives for library preparation |
| US7504364B2 (en) * | 2002-03-01 | 2009-03-17 | Receptors Llc | Methods of making arrays and artificial receptors |
| EP1487978B1 (en) * | 2002-03-15 | 2008-11-19 | Nuevolution A/S | An improved method for synthesising templated molecules |
| EP1543157A4 (en) * | 2002-07-24 | 2006-11-15 | Ptc Therapeutics Inc | METHODS FOR IDENTIFYING SMALL MOLEDULES THAT MODULATE PREMATURE TRANSLATION TERMINATION AND NONSENSE MEDIATED mRNA DECAY |
| EP1539980B1 (en) * | 2002-08-01 | 2016-02-17 | Nuevolution A/S | Library of complexes comprising small non-peptide molecules and double-stranded oligonucleotides identifying the molecules |
| TWI331922B (en) * | 2002-08-09 | 2010-10-21 | Ipsen Pharma Sas | Growth hormone releasing peptides |
| US7469076B2 (en) * | 2003-09-03 | 2008-12-23 | Receptors Llc | Sensors employing combinatorial artificial receptors |
| US20060057625A1 (en) * | 2002-09-16 | 2006-03-16 | Carlson Robert E | Scaffold-based artificial receptors and methods |
| US20050037429A1 (en) * | 2003-03-28 | 2005-02-17 | Receptors Llc | Artificial receptors including reversibly immobilized building blocks and methods |
| US20050136483A1 (en) * | 2003-09-03 | 2005-06-23 | Receptors Llc | Nanodevices employing combinatorial artificial receptors |
| WO2005003326A2 (en) * | 2003-03-28 | 2005-01-13 | Receptors Llc. | Artificial receptors including reversibly immobilized building blocks and methods |
| US20040137481A1 (en) * | 2002-09-16 | 2004-07-15 | Receptors Llc | Artificial receptor building blocks, components, and kits |
| US20050170385A1 (en) * | 2002-09-16 | 2005-08-04 | Receptors Llc | Artificial receptors including gradients |
| US20050037428A1 (en) * | 2002-09-16 | 2005-02-17 | Receptors Llc | Artificial receptors including reversibly immobilized building blocks, the building blocks, and methods |
| US20050037381A1 (en) * | 2002-09-16 | 2005-02-17 | Receptors Llc | Artificial receptors, building blocks, and methods |
| US8791053B2 (en) * | 2002-09-27 | 2014-07-29 | Mpm-Holding Aps | Spatially encoded polymer matrix |
| AU2003273792B2 (en) | 2002-10-30 | 2011-07-07 | Nuevolution A/S | Method for the synthesis of a bifunctional complex |
| EP2175019A3 (en) | 2002-12-19 | 2011-04-06 | Nuevolution A/S | Quasirandom structure and function guided synthesis methods |
| WO2004074429A2 (en) | 2003-02-21 | 2004-09-02 | Nuevolution A/S | Method for producing second-generation library |
| DE602004023960D1 (en) * | 2003-09-18 | 2009-12-17 | Nuevolution As | Method for obtaining structural information of encoded molecules and for selection of compounds |
| US20050261475A1 (en) * | 2004-02-13 | 2005-11-24 | Harvard Medical School | Solid-phase capture-release-tag methods for phosphoproteomic analyses |
| US20090239211A1 (en) * | 2004-02-17 | 2009-09-24 | Nuevolution A/S | Method For Enrichment Involving Elimination By Mismatch Hybridisation |
| US7998060B2 (en) | 2004-04-19 | 2011-08-16 | The Invention Science Fund I, Llc | Lumen-traveling delivery device |
| US8353896B2 (en) | 2004-04-19 | 2013-01-15 | The Invention Science Fund I, Llc | Controllable release nasal system |
| US8092549B2 (en) | 2004-09-24 | 2012-01-10 | The Invention Science Fund I, Llc | Ciliated stent-like-system |
| US8337482B2 (en) | 2004-04-19 | 2012-12-25 | The Invention Science Fund I, Llc | System for perfusion management |
| US8024036B2 (en) | 2007-03-19 | 2011-09-20 | The Invention Science Fund I, Llc | Lumen-traveling biological interface device and method of use |
| US9011329B2 (en) | 2004-04-19 | 2015-04-21 | Searete Llc | Lumenally-active device |
| US8361013B2 (en) | 2004-04-19 | 2013-01-29 | The Invention Science Fund I, Llc | Telescoping perfusion management system |
| US7850676B2 (en) | 2004-04-19 | 2010-12-14 | The Invention Science Fund I, Llc | System with a reservoir for perfusion management |
| US9801527B2 (en) | 2004-04-19 | 2017-10-31 | Gearbox, Llc | Lumen-traveling biological interface device |
| US7857767B2 (en) | 2004-04-19 | 2010-12-28 | Invention Science Fund I, Llc | Lumen-traveling device |
| WO2006028930A2 (en) | 2004-09-03 | 2006-03-16 | Receptors Llc | Combinatorial artificial receptors including tether building blocks on scaffolds |
| US7985715B2 (en) * | 2004-09-11 | 2011-07-26 | Receptors Llc | Combinatorial artificial receptors including peptide building blocks |
| SG183720A1 (en) * | 2005-08-12 | 2012-09-27 | Cambrios Technologies Corp | Nanowires-based transparent conductors |
| WO2007062664A2 (en) | 2005-12-01 | 2007-06-07 | Nuevolution A/S | Enzymatic encoding methods for efficient synthesis of large libraries |
| US8734823B2 (en) * | 2005-12-14 | 2014-05-27 | The Invention Science Fund I, Llc | Device including altered microorganisms, and methods and systems of use |
| US20110172826A1 (en) * | 2005-12-14 | 2011-07-14 | Amodei Dario G | Device including altered microorganisms, and methods and systems of use |
| US8682619B2 (en) * | 2005-12-14 | 2014-03-25 | The Invention Science Fund I, Llc | Device including altered microorganisms, and methods and systems of use |
| US20110183348A1 (en) * | 2010-01-22 | 2011-07-28 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Compositions and methods for therapeutic delivery with microorganisms |
| US8278094B2 (en) | 2005-12-14 | 2012-10-02 | The Invention Science Fund I, Llc | Bone semi-permeable device |
| US9198563B2 (en) | 2006-04-12 | 2015-12-01 | The Invention Science Fund I, Llc | Temporal control of a lumen traveling device in a body tube tree |
| US8936629B2 (en) | 2006-04-12 | 2015-01-20 | Invention Science Fund I Llc | Autofluorescent imaging and target ablation |
| US20100022494A1 (en) * | 2008-07-24 | 2010-01-28 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Method, device, and kit for maintaining physiological levels of steroid hormone in a subject |
| US20100022991A1 (en) * | 2008-07-24 | 2010-01-28 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | System and device for maintaining physiological levels of steroid hormone in a subject |
| US20100022497A1 (en) * | 2008-07-24 | 2010-01-28 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Method for treating or preventing a cardiovascular disease or condition utilizing estrogen receptor modulators based on APOE allelic profile of a mammalian subject |
| US20100061976A1 (en) * | 2008-07-24 | 2010-03-11 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Method for treating or preventing osteoporosis by reducing follicle stimulating hormone to cyclic physiological levels in a mammalian subject |
| US20110177154A1 (en) * | 2008-09-15 | 2011-07-21 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Tubular nanostructure targeted to cell membrane |
| US20100137246A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Anti-inflammatory compositions and methods |
| US20100136097A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems for modulating inflammation |
| US20100136095A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems for modulating inflammation |
| US20100136096A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems for modulating inflammation |
| US20100135983A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Anti-inflammatory compositions and methods |
| US20100136094A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems for modulating inflammation |
| US20100137247A1 (en) * | 2008-12-02 | 2010-06-03 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Anti-inflammatory compositions and methods |
| US20120041287A1 (en) | 2008-12-04 | 2012-02-16 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems, devices, and methods including implantable devices with anti-microbial properties |
| US8430831B2 (en) | 2009-02-25 | 2013-04-30 | The Invention Science Fund I, Llc | Device, system, and method for controllably reducing inflammatory mediators in a subject |
| US8317737B2 (en) * | 2009-02-25 | 2012-11-27 | The Invention Science Fund I, Llc | Device for actively removing a target component from blood or lymph of a vertebrate subject |
| US8246565B2 (en) * | 2009-02-25 | 2012-08-21 | The Invention Science Fund I, Llc | Device for passively removing a target component from blood or lymph of a vertebrate subject |
| US8758324B2 (en) | 2010-03-05 | 2014-06-24 | The Invention Science Fund I, Llc | Device for actively removing a target cell from blood or lymph of a vertebrate subject |
| US8106655B2 (en) * | 2009-05-29 | 2012-01-31 | The Invention Science Fund I, Llc | Multiplex imaging systems, devices, methods, and compositions including ferromagnetic structures |
| US8154285B1 (en) | 2009-05-29 | 2012-04-10 | The Invention Science Fund I, Llc | Non-external static magnetic field imaging systems, devices, methods, and compositions |
| US8063636B2 (en) * | 2009-05-29 | 2011-11-22 | The Invention Science Fund I, Llc | Systems, devices, methods, and compositions including targeted ferromagnetic structures |
| US20100303733A1 (en) * | 2009-05-29 | 2010-12-02 | Searete Llc, A Limited Liability Corporation Of The State Of Delaware | Systems, devices, methods, and compositions including ferromagnetic structures |
| US8058872B2 (en) | 2009-05-29 | 2011-11-15 | The Invention Science Fund I, Llc | Systems, devices, methods, and compositions including functionalized ferromagnetic structures |
| US9024766B2 (en) * | 2009-08-28 | 2015-05-05 | The Invention Science Fund, Llc | Beverage containers with detection capability |
| US8898069B2 (en) * | 2009-08-28 | 2014-11-25 | The Invention Science Fund I, Llc | Devices and methods for detecting an analyte in salivary fluid |
| EP3540059A1 (en) | 2010-04-16 | 2019-09-18 | Nuevolution A/S | Bi-functional complexes and methods for making and using such complexes |
| US10278624B2 (en) | 2013-05-23 | 2019-05-07 | Iphenotype Llc | Method and system for maintaining or improving wellness |
| WO2021018899A1 (en) | 2019-07-29 | 2021-02-04 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Use of gdf15 as a marker for exercise management |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4354972A (en) * | 1981-06-29 | 1982-10-19 | Kaiser Emil T | Synthesis of steroids |
| CA1255586A (en) * | 1984-07-24 | 1989-06-13 | Hendrik M. Geysen | Method of determining mimotopes |
| NZ215865A (en) * | 1985-04-22 | 1988-10-28 | Commw Serum Lab Commission | Method of determining the active site of a receptor-binding analogue |
| US5133866A (en) * | 1988-03-24 | 1992-07-28 | Terrapin Technologies, Inc. | Method to identify analyte-bending ligands |
| US5010175A (en) * | 1988-05-02 | 1991-04-23 | The Regents Of The University Of California | General method for producing and selecting peptides with specific properties |
| US5225533A (en) * | 1988-05-02 | 1993-07-06 | The Regents Of The University Of California | General method for producing and selecting peptides with specific properties |
| US5266684A (en) * | 1988-05-02 | 1993-11-30 | The Reagents Of The University Of California | Peptide mixtures |
| US5366862A (en) * | 1990-02-14 | 1994-11-22 | Receptor Laboratories, Inc. | Method for generating and screening useful peptides |
| US5182366A (en) * | 1990-05-15 | 1993-01-26 | Huebner Verena D | Controlled synthesis of peptide mixtures using mixed resins |
| US5252296A (en) * | 1990-05-15 | 1993-10-12 | Chiron Corporation | Method and apparatus for biopolymer synthesis |
| US5650489A (en) * | 1990-07-02 | 1997-07-22 | The Arizona Board Of Regents | Random bio-oligomer library, a method of synthesis thereof, and a method of use thereof |
| CA2090860C (en) * | 1990-11-21 | 2003-09-16 | Richard A. Houghten | Synthesis of equimolar multiple oligomer mixtures, especially of oligopeptide mixtures |
| DK0834575T3 (en) * | 1990-12-06 | 2002-04-02 | Affymetrix Inc A Delaware Corp | Identification of nucleic acids in samples |
| ES2097925T3 (en) * | 1991-09-18 | 1997-04-16 | Affymax Tech Nv | METHOD FOR SYNTHESIZING DIFFERENT OLIGOMER COLLECTIONS. |
| US5270170A (en) * | 1991-10-16 | 1993-12-14 | Affymax Technologies N.V. | Peptide library and screening method |
| US5240680A (en) * | 1991-12-19 | 1993-08-31 | Chiron Corporation | Automated apparatus for use in peptide synthesis |
| AU3816993A (en) * | 1992-03-19 | 1993-10-21 | Regents Of The University Of California, The | Multiple tag labeling method for DNA sequencing |
| US5573905A (en) * | 1992-03-30 | 1996-11-12 | The Scripps Research Institute | Encoded combinatorial chemical libraries |
| WO1994002515A1 (en) * | 1992-07-21 | 1994-02-03 | Bunsen Rush Laboratories Inc. | Oligomer library formats and methods relating thereto |
| US5470753A (en) * | 1992-09-03 | 1995-11-28 | Selectide Corporation | Peptide sequencing using mass spectrometry |
| US5288514A (en) * | 1992-09-14 | 1994-02-22 | The Regents Of The University Of California | Solid phase and combinatorial synthesis of benzodiazepine compounds on a solid support |
| US5840485A (en) * | 1993-05-27 | 1998-11-24 | Selectide Corporation | Topologically segregated, encoded solid phase libraries |
| JP3394777B2 (en) | 1993-05-27 | 2003-04-07 | セレクタイド コーポレーション | Topologically separated, encoded solid-phase library |
| WO1995013538A1 (en) | 1993-11-12 | 1995-05-18 | Operon Technologies, Inc. | Methods of producing and screening complex chemical libraries |
-
1995
- 1995-01-13 CA CA002180844A patent/CA2180844A1/en not_active Abandoned
- 1995-01-13 WO PCT/US1995/000572 patent/WO1995019567A1/en not_active Application Discontinuation
- 1995-01-13 EP EP95914675A patent/EP0739486A4/en not_active Withdrawn
- 1995-01-13 ZA ZA95260A patent/ZA95260B/en unknown
- 1995-01-13 JP JP7519163A patent/JPH09511486A/en active Pending
- 1995-01-13 US US08/676,143 patent/US7067326B2/en not_active Expired - Fee Related
- 1995-01-13 AU AU21565/95A patent/AU686785B2/en not_active Ceased
-
1996
- 1996-04-08 US US08/628,972 patent/US5804563A/en not_active Expired - Lifetime
Non-Patent Citations (6)
| Title |
|---|
| JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, Volume 114, issued 1992, HIRSCHMANN et al., "The First Design and Synthesis of a Steroidal Peptidomimetic. The Potential Value of Peptidomimetics in Elucidating the Bioactive Conformation of Peptide Ligands", pages 9699-9701. * |
| JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, Volume 114, TOR et al., "Tripodal Peptides with Chiral Conformations Stabilized by Interstrand Hydrogen Bonds", pages 6653-6660. * |
| JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, Volume 115, issued January 1993, YOON et al., "An Exceptional Synthetic Receptor for Peptides", pages 823-824. * |
| PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES, Volume 89, issued June 1992, BRENNER et al., "Encoded Combinatorial Chemistry", pages 5381-5383. * |
| PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES, Volume 90, issued December 1993, OHLMEYER et al., "Complex Synthetic Chemical Libraries Indexes with Molecular Tags", pages 10922-10926. * |
| See also references of EP0739486A4 * |
Cited By (17)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6284459B1 (en) | 1995-04-25 | 2001-09-04 | Discovery Partners International | Solid support matrices with memories and combinatorial libraries therefrom |
| US5961923A (en) * | 1995-04-25 | 1999-10-05 | Irori | Matrices with memories and uses thereof |
| US6100026A (en) * | 1995-04-25 | 2000-08-08 | Irori | Matrices with memories and uses thereof |
| WO1997043232A1 (en) * | 1996-05-14 | 1997-11-20 | Hoechst Research & Technology Deutschland Gmbh & Co.Kg | Novel substance library and supramolecular complexes produced therewith |
| WO1998020349A1 (en) * | 1996-11-05 | 1998-05-14 | Isis Pharmaceuticals, Inc. | Complex chemical libraries |
| US5780241A (en) * | 1996-11-05 | 1998-07-14 | Isis Pharmaceuticals, Inc. | Complex chemical libraries |
| US6800444B1 (en) | 1996-11-05 | 2004-10-05 | Isis Pharmaceuticals, Inc. | Complex chemical libraries |
| WO1998046631A1 (en) * | 1997-04-11 | 1998-10-22 | Eli Lilly And Company | Combinatorial libraries of peptidomimetic macrocycles and processes therefor |
| WO1999031124A1 (en) * | 1997-12-12 | 1999-06-24 | Novo Nordisk A/S | Cholic acid-based scaffolds for multidimensional molecular presentation of peptides |
| US6350738B1 (en) | 1998-03-06 | 2002-02-26 | Brigham Young University | Steroid derived antibiotics |
| US6486148B2 (en) | 1998-03-06 | 2002-11-26 | Brigham Young University | Steroid derived antibiotics |
| US6767904B2 (en) | 1998-03-06 | 2004-07-27 | Bringham Young University | Steroid derived antibiotics |
| WO1999044616A1 (en) * | 1998-03-06 | 1999-09-10 | Brigham Young University | Steroid derived antibiotics |
| CN100398109C (en) * | 1998-03-06 | 2008-07-02 | 杨伯翰大学 | Steroid derived antibiotics |
| US7598234B2 (en) | 1998-03-06 | 2009-10-06 | Brigham Young University | Steroid derived antibiotics |
| EP1384999A1 (en) * | 2001-04-18 | 2004-01-28 | Japan Science and Technology Corporation | Bacteriotoxin adsorbents and method of screening the same |
| EP1384999A4 (en) * | 2001-04-18 | 2008-05-21 | Japan Science & Tech Agency | Bacteriotoxin adsorbents and method of screening the same |
Also Published As
| Publication number | Publication date |
|---|---|
| ZA95260B (en) | 1995-09-28 |
| US5804563A (en) | 1998-09-08 |
| AU2156595A (en) | 1995-08-01 |
| CA2180844A1 (en) | 1995-07-20 |
| US20030104360A1 (en) | 2003-06-05 |
| AU686785B2 (en) | 1998-02-12 |
| US7067326B2 (en) | 2006-06-27 |
| JPH09511486A (en) | 1997-11-18 |
| EP0739486A4 (en) | 1998-09-09 |
| EP0739486A1 (en) | 1996-10-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6797522B1 (en) | Synthetic receptors | |
| US7067326B2 (en) | Synthetic receptors, libraries and uses thereof | |
| US5635598A (en) | Selectively cleavabe linners based on iminodiacetic acid esters for solid phase peptide synthesis | |
| AU686186B2 (en) | Topologically segregated, encoded solid phase libraries | |
| US5840485A (en) | Topologically segregated, encoded solid phase libraries | |
| Yoon et al. | Sequence-selective peptide binding with a synthetic receptor | |
| JPH06504758A (en) | Library of modified peptides with protease resistance | |
| JPH05170791A (en) | Solid state synthesis of peptide containing c-terminated azaamino acid amide | |
| JP4625603B2 (en) | Synthesis of β-hairpin loop analogues with immobilized templates | |
| AU2016251857B2 (en) | Method and composition for peptide cyclization and protease treatment | |
| Braxmeier et al. | Identification of sequence selective receptors for peptides with a carboxylic acid terminus | |
| US7126006B2 (en) | Glycoluril core molecules for combinatorial libraries | |
| Krattiger et al. | Using Catalyst–Substrate Coimmobilization for the Discovery of Catalysts for Asymmetric Aldol Reactions in Split‐and‐Mix Libraries | |
| RU2036200C1 (en) | Method of synthesis of peptideamide derivatives or their physiologically compatible acetates or hydrochlorides | |
| Cho et al. | Preparation of tris-based dendrimer-grafted core-shell type resin for solid-phase peptide synthesis | |
| US5872207A (en) | N-terminal-marked peptides immobilized on glass beads and method of preparation and method of use thereof | |
| WO1999031124A1 (en) | Cholic acid-based scaffolds for multidimensional molecular presentation of peptides | |
| Wennemers | Encoded combinatorial chemistry: a tool for the study of selective intermolecular interactions | |
| US6190920B1 (en) | Method for detecting enzyme catalyzed cyclization | |
| CA2579631C (en) | Selectively cleavable linkers based on iminodiacetic acid ester bonds | |
| Blodgett | Novel methods in chromatographic separations: New* chiral stationary phases and molecular tagging | |
| Ferguson | Design, synthesis and biological screening of combinatorial chemical libraries | |
| AA | _,. NOON | |
| JP2008106005A (en) | Compound having amino acid residue or peptide residue and method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AM AT AU BB BG BR BY CA CH CN CZ DE DK EE ES FI GB GE HU JP KE KG KP KR KZ LK LR LT LU LV MD MG MN MW MX NL NO NZ PL PT RO RU SD SE SI SK TJ TT UA US UZ VN |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): KE MW SD SZ AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN ML MR NE SN TD TG |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2180844 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1995914675 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08676143 Country of ref document: US |
|
| WWP | Wipo information: published in national office |
Ref document number: 1995914675 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1995914675 Country of ref document: EP |