WO1994016030A1 - Non-fluorescing oil-based drilling fluid - Google Patents
Non-fluorescing oil-based drilling fluid Download PDFInfo
- Publication number
- WO1994016030A1 WO1994016030A1 PCT/US1994/000469 US9400469W WO9416030A1 WO 1994016030 A1 WO1994016030 A1 WO 1994016030A1 US 9400469 W US9400469 W US 9400469W WO 9416030 A1 WO9416030 A1 WO 9416030A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- drilling fluid
- saturated
- additive
- fatty acid
- oil
- Prior art date
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/04—Aqueous well-drilling compositions
- C09K8/26—Oil-in-water emulsions
- C09K8/28—Oil-in-water emulsions containing organic additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/32—Non-aqueous well-drilling compositions, e.g. oil-based
- C09K8/34—Organic liquids
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/32—Non-aqueous well-drilling compositions, e.g. oil-based
- C09K8/36—Water-in-oil emulsions
Definitions
- This invention relates to a non-fluorescing oil-based drilling fluid used in the drilling of subterranean oil and gas wells as well as other drilling fluid applications and drilling procedures.
- These non-fluorescing drilling fluids are composed of synthetic hydrocarbons, having molecular weights of from 120 to 1000, derived from olefinic monomers and other additives such as surfactants and emulsifiers.
- olefinic monomers are from the groups consisting of a carbon chain from C 2 to C 14 having at least one polymerizible double bond.
- the surfactants comprise monounsaturated fatty acids and saturated fatty acids.
- the emulsifiers comprise saturated fatty acids, saturated amido-amines, and saturated sulfonates. Additional materials that are common to the development and formulation of drilling fluids may also be included in the instant drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light.
- drilling fluid In rotary drilling there are a variety of functions and characteristics that are expected of a drilling fluid ("drilling mud” or simply “mud”).
- the drilling fluid is expected to carry cuttings from beneath the bit, transport them up the annulus, and permit their separation at the surface while at the same time the rotary bit is cooled and cleaned.
- a drilling mud is also intended to reduce friction between the drill string and the sides of the hole while maintaining the stability of uncased sections of the borehole.
- the drilling fluid is formulated to prevent unwanted influxes of formation fluids from permeable rocks penetrated and likewise to form a thin, low permeability filter cake which seals pores and other openings and formations penetrated by the bit.
- the drilling fluid is used to collect and interpret information available from drill cuttings, cores and electrical logs.
- drill cuttings are conveyed up the hole by a drilling fluid.
- a drilling fluid With an oil-based drilling fluid, the cuttings, besides ordinarily containing moisture, are necessarily coated with an adherent film or layer of oily drilling fluid which may penetrate into the interior of each cutting. This is true despite the use of various vibrating screens, mechanical separation devices, and various chemical and washing techniques.
- Mud logging is a process which involves the recording of information derived from the examination and analysis of formation cuttings.
- a portion of the drilling fluid is diverted through a gas-detecting device and examined under ultraviolet light to detect the presence of oil or gas. If the drilling fluid fluoresces this indicates that hydrocarbons are present and that the desired formation has been reached.
- Graham et al. U.S. Patent No. 2,951,940 disclose a method of detecting the presence of crude oil in the earth's strata. This method entails contacting the surface of a rock chip that has been carried to the earth's surface by a drilling mud with a reverse-wetting agent. The rock chip is then contacted with ultraviolet light. By comparing the relative fluorescence of the surface of the chip before and after the contacting operation, a positive indication of the presence of the hydrocarbons may be obtained.
- U.S. Patent No. 3,050,141 discloses a drilling fluid emulsion which comprises an oil- and water-insoluble and immiscible liquid.
- This liquid is characterized by the fact that it either fails to fluoresce in the presence of ultraviolet light or it fluoresces a different color from typical unrefined hydrocarbons or crude oils.
- This liquid forms a third layer in the presence of oil and water because it is incompatible with both oil and water.
- the drilling fluid is composed of synthetic hydrocarbons that have been synthesized from one or more olefinic monomers having a chain length of C 2 to C M .
- the hydrocarbons have an average molecular weight of from about 120 to about 1000.
- the drilling fluid also includes surfactants and/or emulsifiers.
- the drilling fluid is characterized by the fact that it fails to fluoresce in the presence of ultraviolet light.
- the synthetic hydrocarbon oils of this invention are manufactured by oligomerizing alpha-olefins or other olefins. The resulting oils are mixtures of branched hydrocarbon molecules with carbon numbers that are even multiples of the base olefin.
- a synthetic hydrocarbon oil made from C 8 olefins contains only molecules that are C g , C 16 , C 24 , C 32 , etc. These oils can be hydrogenated to achieve complete saturation, or partially hydrogenated, or left unhydrogenated. Preferably they contain no aromatics. Because these oils are synthetic, their molecular size and structure, and their performance characteristics, can be controlled in a predictable and understandable manner.
- surfactant as used in this application should be understood to include emulsifiers.
- surfactant and emulsifier are used interchangeably.
- the surfactants of the instant invention are fatty acids with one or less points of unsaturation which do not fluoresce in presence of ultraviolet light.
- Preferred surfactants are distilled tall oil fatty acids, oleic acid, and isostearic acid.
- the emulsifiers of the instant invention are derived from monounsaturated or saturated fatty acids, such emulsifiers include amido-amines and sulfonates.
- Preferred emulsifiers are sorbitan mono- and di-oleates (Arlacel 186 obtained from ICI, Ltd.), amido-amines (Miranol-CS; Rh ⁇ ne-Poulenc), and sulfosuccinates (Emcol-4500; Witco
- the drilling fluid may also include other materials common to the development and formulation of drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light. This allows the drilling fluid to be altered to fit specific well conditions. The only requirement is that the drilling fluid must remain non-fluorescent.
- Figure 1 is a three-dimensional representation of an emulsifier that fluoresces in the presence of ultraviolet light.
- the emulsifier that was utilized in Figure 1 in the oil-based mud is Versamul and it is produced and distributed by M-I Drilling Fluids Co.
- This emulsifier is identical to the Kenol-ES emulsifier utilized by Boyd et al. in U.S. Patent No. 4,787,990, column 5, lines 1-15.
- the sample was diluted to 1 :400 prior to testing. This figure was produced by plotting the emission wavelength (x- axis), the excitation wavelength (y-axis) and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 6.
- the fluorescence intensity was measured using a Perkin-Elmer (PE) 650-40 fluorescence spectrometer. Additional information concerning this machine may be found in the article by J.M. Brooks et al. entitled “Applications of Total Scanning Fluorescence to Exploration Geochemistry, " 15th Annual Offshore Technology Conference, Houston, Texas, May 2-5, 1983, pp. 393-396, OTC 4624, which is hereby incorporated by reference.
- PE Perkin-Elmer
- Figure 2 is a three-dimensional representation of an emulsifier that does not fluoresce in the presence of ultraviolet light.
- the emulsifier that was utilized is sold under the brand name Emcol-4500 distributed by Witco Chemical Co.
- the sample was diluted 1 :400 prior to testing. This figure was produced by plotting the emission wavelength (x-axis), the excitation wavelength (y-axis), and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 7.
- the fluorescence intensity was measured using a Perkin-Elmer (PE) 650-40 fluorescence spectrometer.
- PE Perkin-Elmer
- Figure 3 is a three-dimensional representation of the fluorescence characteristics of a California oil as it is removed from the ground. This figure was produced by plotting the emission wavelength (x-axis), the excitation wavelength (y- axis), and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 5A.
- non-fluorescing shall mean that a substance does not fluoresce under ultraviolet light to an extent that is discernable by the naked eye. More particularly the substance will exhibit a fluorescence intensity of 0 to 800 when diluted 1 to 400. Preferably the substance will exhibit a fluorescence intensity of 0 to 500. This last range of fluorescence is so slight as to be discemable only through the aid of a machine. The following machines may be used to determine the fluorescence intensity of a substance:
- non-fluorescence will be understood to refer to a fluorescence intensity of less than 800 under ultraviolet light when diluted with hexane at a ratio of 1 to 400.
- the drilling fluid is composed of synthetic hydrocarbons that have been synthesized from one or more olefinic monomers having a chain length of C 2 to C 14 .
- the hydrocarbons have an average molecular weight of from about 120 to about 1000.
- the drilling fluid also includes non-fluorescing surfactants and/or emulsifiers.
- the drilling fluid is characterized by the fact that it fails to fluoresce in the presence of ultraviolet light.
- Various synthetic hydrocarbons are commercially available to be used in the present invention.
- polypropenes from AMOCO Chemical Company product numbers #9009 and 9011; Chevron Chemical Company's product identified as Polymer-560; polybutenes Indopol L-14 and H-15 offered by AMOCO Chemical Company; as well as dimeric, trimeric and tetrameric 1-decenes from Emery, Ethyl and Chevron Corporations are suitable for the present invention.
- These synthetic hydrocarbons can also be blended to achieve the desired chemical characteristics, which are determined according to the end use of the product.
- the synthetic hydrocarbons that are believed to be useful in the practice of this invention are characterized by chain length and molecular weight parameters.
- Useful synthetic hydrocarbons are branched chain oligomers synthesized from one or more olefins containing a C 2 to C 14 chain length and wherein the oligomers have an average molecular weight of from 120 to 1000.
- the synthetic hydrocarbons are branched chain oligomers synthesized from one or more olefins containing a C 3 to C 12 chain length and wherein the oligomers have an average molecular weight of from 160 to
- the synthetic hydrocarbons are branched chain oligomers synthesized from one or more oligomers containing a C 4 to C 14 chain length and wherein the oligomers have an average molecular weight of 200 to 600.
- the synthetic hydrocarbon mixture must have performance and viscosity characteristics that permit functional utility as a drilling fluid.
- the synthetic hydrocarbon mixture should have a viscosity of from 1.0 to 6.0 centistokes, preferable a viscosity of from 1.5 to 4.0 centistokes and most preferably from 1.5 'to 3.5 centistokes.
- the synthetic hydrocarbons of the present invention may be hydrogenated (saturated), partially hydrogenated or non- hydrogenated.
- the interfacial tension between oil and water is very high, so if the liquids are mixed together they mechanically separate immediately when the agitation ceases, to minimize the interfacial area.
- Lowering the interfacial tension with a surfactant enables one liquid to form a stable dispersion of fine droplets in the other.
- the lower the interfacial tension the smaller the droplets and the more stable the emulsion.
- oil is the dispersed phase and water is the continuous phase.
- a suitable emulsion can be formed upon the use of a suitable emulsifier.
- an oil-in-water or water-in-oil emulsion is formed depends on the relative solubility of the emulsifier in the two phases.
- a preferentially water soluble surfactant such as sodium oleate
- calcium and magnesium oleates are soluble in oil, but not in water, and thus form water-in-oil emulsions.
- An invert water-in-oil emulsion has oil as the continuous phase.
- the compositions of this invention may comprise 100% oil by volume of the total composition. Water, usually in the form of brine, is normally added in these compositions. Water may be added to the drilling fluid up to a volume of 70%. In more preferred embodiments water is added from 5% to 65% by volume, and in the most preferred embodiment water is added from 10% to 60% by volume. These brines contain salts such as NaCl and/or CaCl 2 in varying amounts.
- compositions of one embodiment of this invention require emulsifiers to incorporate the brine or water phase into the synthetic hydrocarbon continuous phase.
- the emulsifiers are chemical compounds which have both oleophilic and hydrophilic parts.
- the emulsifiers that have demonstrated utility in the emulsions of this invention are saturated or monounsaturated fatty acids, saturated amido-amines, saturated sulfonates, and combinations of the above. Blends of these materials as well as other emulsifiers can be used for this application provided that the emulsifiers fail to fluoresce in the presence of ultraviolet light.
- Preferred emulsifiers are oleic acid, isostearic acid, sorbitan monoleate, Miranol-CS, amido-amines, and sulfosuccinates. These emulsifiers are manufactured and distributed by Emery, ICI, Witco, Rh ⁇ ne- Poulenc and M-I Drilling Fluids.
- Surfactants may also be used in the drilling fluids of this invention.
- the surfactants that have demonstrated utility in the emulsions of this invention are saturated fatty acids and monounsaturated fatty acids and combinations of the above. Blends of these materials as well as other surfactants may also be utilized provided that the surfactants do not fluoresce when contacted with ultraviolet light.
- Preferred surfactants are isostearic acid, oleic acid and Acintol-EPG. These surfactants are manufactured and distributed by the Emery Chemical Co. and the Arizona Chemical Co.
- the oil-based drilling fluid compositions of this invention may contain an additional chemical known as a wetting agent.
- Various wetting agents are available and can be included in the compositions.
- the wetting agents included, but not limited to the present invention are oxidized fatty acids, saturated fatty acid-based imidazolines.
- Preferred wetting agents are saturated fatty acid-based imidazolines. Blends of these materials as well as other wetting agents can be used for this application provided that the wetting agents fail to fluoresce in the presence of ultraviolet light.
- Organophilic clays are also used as viscosifiers in the oil-based drilling fluid composition of the present invention.
- Other viscosifiers such as oil soluble polymers, polyamide resins, polycarboxylic acids and soaps, can also be used provided the materials are non-fluorescing.
- the amount of viscosifier used in the composition can vary depending upon the end use of the composition. However, normally about 0.1 % to 10% by weight range are sufficient for most applications.
- VG-69 is an organoclay material distributed by M-I Drilling Fluids Company.
- the drilling fluid composition of this invention may optionally contain a weight material.
- the quantity depends upon the desired density and viscosity of the final composition.
- the preferred weight materials include, but are not limited to, barite, iron oxide, calcium carbonate and the like.
- the weight material is typically added to result in a drilling fluid density of up to 24 pounds per gallon, preferably up to 18 pounds per gallon and most preferably up to 16.5 pounds per gallon.
- fluid loss control agents such as modified lignite and oil soluble or oil dispersible polymers, can be added to the drilling fluid system of this invention.
- the drilling fluid may also include other materials common to the development and formulation of drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light. This allows the drilling fluid to be altered to fit specific well conditions. The only requirement is that the drilling fluid must remain non-fluorescent.
- drilling fluid compositions utilizing the products of this invention.
- chemical name of each material used is supplied the first time the trade name is used.
- Synthetic polyalphaolefin Ethylflow-162 was utilized to prepare 14 pounds per gallon ("ppg"), 80:20 oil: water ratio oil-based mud.
- Formulation 1 included
- Table 1 summarizes the rheological properties of the above formulations on initial mixing and the after heating at 250°F for 16 hours. These properties were measured at room temperature.
- a 12 ppg non-fluorescent oil-based mud was prepared using Mobil Chemical Co. unhydrogenated polyalphaolefin MCP-1060 and the surfactants of this invention.
- Amido-amine- 1 and amido-amine-2 were synthesized from non-fluorescing saturated fatty acids according to the procedures described previously.
- Table 5 A represents the fluorescence of a California oil as it is removed from the ground.
- the oil itself as demonstrated in Table 5A, have fluorescence characteristics that are different from those of the drilling mud used.
- the maximum intensity is less than 800 for the drilling mud used.
- the maximum intensity is well above 800 for most of the wavelengths shown for the California oil diluted with hexane by a ratio of 1 to 10,000.
- the cuttings containing the oil were collected on March 24, 1992.
- Table 7, on the other hand demonstrates the fluorescence characteristics of a drilling fluid sample using Emcol-4500 as the primary drilling fluid component. It can be seen that at all wavelengths tested, the maximum intensity is well beneath the 800 number required to characterize the material as fluorescent.
- the fluorescence values of the materials used in Tables 6 and 7 are demonstrated graphically by Figures 1 and 2, respectively. -IS
- Table 8 shows the total scanning fluorescent of a variety of materials evaluated for use in the process and product of this invention.
- the maximum intensity is 3830 for Versacoat at excitation wavelength 320 nm. and the emission wavelength 380 nm. and the ratio Rl is calculated by dividing intensity at emission maximum 360 nm. by intensity at excitation maximum 320 nm. It has been concluded from the data in Table 8 that the materials having a maximum intensity of greater than 800 would be considered fluorescent and therefore not useful in the process and product of this invention. Those materials having a maximum intensity of less than 800 have shown distinct utility and are not considered fluorescent.
Abstract
The present invention relates to an oil-based drilling fluid, a continuous phase for an oil-based drilling fluid, and additives for an oil-based drilling fluid. The drilling fluid, the continuous phase of the drilling fluid, and the additives for the oil-based drilling fluid are characterized by non-fluorescence when exposed to ultraviolet light. The continuous phase of the drilling fluid is composed of synthetic hydrocarbons selected from the group consisting of branched chain oligomers synthesized from one or more olefins containing a C2 to C14 chain length, wherein the oligomers have an average molecular weight of from 120 to 100, and additives. The additives of the continuous phase include surfactants, wetting agents and/or emulsifiers.
Description
10 NON-FLUORESCING OIL-BASED DRILLING FLUID
15
This application is a continuation- in-part of commonly assigned, copending
20 patent application U.S. Serial No. 08/004,547 filed January 14, 1993, which is a continuation-in-part of commonly assigned, copen patent application U.S. Serial
No. 535,110 filed June 8, 1990, which is a continuation-in-part of U.S. Patent
Application Serial No. 503,304 filed March 30, 1990, now abandoned.
25 This invention relates to a non-fluorescing oil-based drilling fluid used in the drilling of subterranean oil and gas wells as well as other drilling fluid applications and drilling procedures. These non-fluorescing drilling fluids are composed of synthetic hydrocarbons, having molecular weights of from 120 to 1000, derived from olefinic monomers and other additives such as surfactants and emulsifiers. The
30 olefinic monomers are from the groups consisting of a carbon chain from C2 to C14 having at least one polymerizible double bond. The surfactants comprise monounsaturated fatty acids and saturated fatty acids. The emulsifiers comprise saturated fatty acids, saturated amido-amines, and saturated sulfonates. Additional materials that are common to the development and formulation of drilling fluids may
also be included in the instant drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light.
In rotary drilling there are a variety of functions and characteristics that are expected of a drilling fluid ("drilling mud" or simply "mud"). The drilling fluid is expected to carry cuttings from beneath the bit, transport them up the annulus, and permit their separation at the surface while at the same time the rotary bit is cooled and cleaned. A drilling mud is also intended to reduce friction between the drill string and the sides of the hole while maintaining the stability of uncased sections of the borehole. Likewise the drilling fluid is formulated to prevent unwanted influxes of formation fluids from permeable rocks penetrated and likewise to form a thin, low permeability filter cake which seals pores and other openings and formations penetrated by the bit. Finally, the drilling fluid is used to collect and interpret information available from drill cuttings, cores and electrical logs.
Various advantages of using oil-based drilling mud in the rotary drilling of boreholes in the earth have been known for some time. In summary, it can be said that under certain conditions, one or more of the following advantages make oil-based muds more desirable than water-based muds. Lubricity characteristics are excellent. Drilling fluid weighing less than about 8 pounds per gallon can be prepared with an oil-based mud, and have proved advantageous in certain special drilling and coring situations. The penetration of the formation by water is avoided. Cores can be recovered in an in situ condition in the presence of hydrous clays and bentonites; no swelling or sloughing is experienced to reduce porosities or cause pipe sticking difficulties. There are fewer problems with hydrophilic shale formations drilled. Evaporite sections such as salt zones can be drilled with minimal leaching of the salt. Furthermore, oil-based muds can be formulated to withstand temperatures up to 500 degrees Fahrenheit.
On both offshore and inland drilling barges and rigs, drill cuttings are conveyed up the hole by a drilling fluid. With an oil-based drilling fluid, the
cuttings, besides ordinarily containing moisture, are necessarily coated with an adherent film or layer of oily drilling fluid which may penetrate into the interior of each cutting. This is true despite the use of various vibrating screens, mechanical separation devices, and various chemical and washing techniques.
Mud logging is a process which involves the recording of information derived from the examination and analysis of formation cuttings. A portion of the drilling fluid is diverted through a gas-detecting device and examined under ultraviolet light to detect the presence of oil or gas. If the drilling fluid fluoresces this indicates that hydrocarbons are present and that the desired formation has been reached.
Unfortunately, in most oil-based drilling fluids either the oil used in the fluid or the surfactants that are present naturally fluoresce. This interferes with the results of the mud log because the mud logger cannot accurately ascertain whether formation hydrocarbons are present. This problem has hampered the use of fluoroscopy to determine the presence of hydrocarbon formations when either oil-based or oil- emulsion muds are used.
Graham et al. , U.S. Patent No. 2,951,940, disclose a method of detecting the presence of crude oil in the earth's strata. This method entails contacting the surface of a rock chip that has been carried to the earth's surface by a drilling mud with a reverse-wetting agent. The rock chip is then contacted with ultraviolet light. By comparing the relative fluorescence of the surface of the chip before and after the contacting operation, a positive indication of the presence of the hydrocarbons may be obtained.
U.S. Patent No. 3,050,141 discloses a drilling fluid emulsion which comprises an oil- and water-insoluble and immiscible liquid. This liquid is characterized by the fact that it either fails to fluoresce in the presence of ultraviolet light or it fluoresces a different color from typical unrefined hydrocarbons or crude oils. This liquid forms
a third layer in the presence of oil and water because it is incompatible with both oil and water.
Boyd, U.S. Patent No. 4,787,990 discloses a non-fluorescent oil-based drilling fluid which consists essentially of branched- and cyclic-paraffins having 11 to 17 carbon atoms per molecule and further having a low aromatic content of less than about 1 % and a low normal-paraffin content of less than about 5 % .
Mercer et al, U.S. Patent No. 5,096,883 teaches a hydrogenated synthetic polyalphaolefin having utility as a drilling fluid.
As can be seen from the above, the development of a drilling fluid that exhibits the desirable characteristics of an oil-based fluid and fails to fluoresce in the presence of UV light has long been an unachieved goal of the oil and gas exploration industry. With the practice of applicant's invention this goal has been realized.
This invention relates to a non-fluorescing oil-based drilling fluid. The drilling fluid is composed of synthetic hydrocarbons that have been synthesized from one or more olefinic monomers having a chain length of C2 to CM. The hydrocarbons have an average molecular weight of from about 120 to about 1000. The drilling fluid also includes surfactants and/or emulsifiers. The drilling fluid is characterized by the fact that it fails to fluoresce in the presence of ultraviolet light.
There is no art- accepted definition of non-fluorescence, as virtually all organic molecules will fluoresce to some extent given some excitation source. Typically, oil- based drilling fluids have a natural degree of fluorescence. However, the fluorescent characteristics of the drilling fluids of this invention are below the levels typically encountered or detectable in crude oil and therefore do not confuse the fluorescence measurement methods and apparatus.
The synthetic hydrocarbon oils of this invention are manufactured by oligomerizing alpha-olefins or other olefins. The resulting oils are mixtures of branched hydrocarbon molecules with carbon numbers that are even multiples of the base olefin. For instance, a synthetic hydrocarbon oil made from C8 olefins contains only molecules that are Cg, C16, C24, C32, etc. These oils can be hydrogenated to achieve complete saturation, or partially hydrogenated, or left unhydrogenated. Preferably they contain no aromatics. Because these oils are synthetic, their molecular size and structure, and their performance characteristics, can be controlled in a predictable and understandable manner.
The term "surfactant" as used in this application should be understood to include emulsifiers. The terms "surfactant" and "emulsifier" are used interchangeably. The surfactants of the instant invention are fatty acids with one or less points of unsaturation which do not fluoresce in presence of ultraviolet light. Preferred surfactants are distilled tall oil fatty acids, oleic acid, and isostearic acid.
The emulsifiers of the instant invention are derived from monounsaturated or saturated fatty acids, such emulsifiers include amido-amines and sulfonates. Preferred emulsifiers are sorbitan mono- and di-oleates (Arlacel 186 obtained from ICI, Ltd.), amido-amines (Miranol-CS; Rhόne-Poulenc), and sulfosuccinates (Emcol-4500; Witco
Chemical).
The drilling fluid may also include other materials common to the development and formulation of drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light. This allows the drilling fluid to be altered to fit specific well conditions. The only requirement is that the drilling fluid must remain non-fluorescent.
Figure 1 is a three-dimensional representation of an emulsifier that fluoresces in the presence of ultraviolet light. The emulsifier that was utilized in Figure 1 in the oil-based mud is Versamul and it is produced and distributed by M-I Drilling Fluids
Co. This emulsifier is identical to the Kenol-ES emulsifier utilized by Boyd et al. in U.S. Patent No. 4,787,990, column 5, lines 1-15. The sample was diluted to 1 :400 prior to testing. This figure was produced by plotting the emission wavelength (x- axis), the excitation wavelength (y-axis) and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 6.
The fluorescence intensity was measured using a Perkin-Elmer (PE) 650-40 fluorescence spectrometer. Additional information concerning this machine may be found in the article by J.M. Brooks et al. entitled "Applications of Total Scanning Fluorescence to Exploration Geochemistry, " 15th Annual Offshore Technology Conference, Houston, Texas, May 2-5, 1983, pp. 393-396, OTC 4624, which is hereby incorporated by reference.
Figure 2 is a three-dimensional representation of an emulsifier that does not fluoresce in the presence of ultraviolet light. The emulsifier that was utilized is sold under the brand name Emcol-4500 distributed by Witco Chemical Co. The sample was diluted 1 :400 prior to testing. This figure was produced by plotting the emission wavelength (x-axis), the excitation wavelength (y-axis), and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 7. The fluorescence intensity was measured using a Perkin-Elmer (PE) 650-40 fluorescence spectrometer.
Figure 3 is a three-dimensional representation of the fluorescence characteristics of a California oil as it is removed from the ground. This figure was produced by plotting the emission wavelength (x-axis), the excitation wavelength (y- axis), and the maximum intensity (z-axis). The data used to generate this figure may be found in Table 5A.
This invention relates to a non-fluorescing oil-based drilling fluid. For the purposes of this invention, non-fluorescing shall mean that a substance does not fluoresce under ultraviolet light to an extent that is discernable by the naked eye.
More particularly the substance will exhibit a fluorescence intensity of 0 to 800 when diluted 1 to 400. Preferably the substance will exhibit a fluorescence intensity of 0 to 500. This last range of fluorescence is so slight as to be discemable only through the aid of a machine. The following machines may be used to determine the fluorescence intensity of a substance:
(1) The Perkin-Elmer (PE) 650-40 fluorescence spectrometer with "SCANR" software programs;
(2) Oil fluoroscope model 2014 by Alkco services; and
(3) Mineral light UVS-54 by San Gabriel Products Inc.. Additional information concerning the Perkin-Elmer (PE) 650-40 fluorescence spectrometer may be found in the article by J. M. Brooks et al. entitled "Applications of Total Scanning Fluorescence to Exploration Geochemistry. " For purposes of this application, the term "non-fluorescence" will be understood to refer to a fluorescence intensity of less than 800 under ultraviolet light when diluted with hexane at a ratio of 1 to 400.
The drilling fluid is composed of synthetic hydrocarbons that have been synthesized from one or more olefinic monomers having a chain length of C2 to C14. The hydrocarbons have an average molecular weight of from about 120 to about 1000. The drilling fluid also includes non-fluorescing surfactants and/or emulsifiers. The drilling fluid is characterized by the fact that it fails to fluoresce in the presence of ultraviolet light.
The following table indicates the preferred olefinic compounds from which the branched chain oligomeric and polymeric synthetic hydrocarbon can be manufactured.
Various synthetic hydrocarbons are commercially available to be used in the present invention. For example, polypropenes from AMOCO Chemical Company, product numbers #9009 and 9011; Chevron Chemical Company's product identified as Polymer-560; polybutenes Indopol L-14 and H-15 offered by AMOCO Chemical Company; as well as dimeric, trimeric and tetrameric 1-decenes from Emery, Ethyl and Chevron Corporations are suitable for the present invention. These synthetic hydrocarbons can also be blended to achieve the desired chemical characteristics, which are determined according to the end use of the product.
As identified hereinbefore, the synthetic hydrocarbons that are believed to be useful in the practice of this invention are characterized by chain length and molecular weight parameters. Useful synthetic hydrocarbons are branched chain oligomers synthesized from one or more olefins containing a C2 to C14 chain length and wherein the oligomers have an average molecular weight of from 120 to 1000. In the preferred embodiments of this invention the synthetic hydrocarbons are branched chain oligomers synthesized from one or more olefins containing a C3 to C12 chain length and wherein the oligomers have an average molecular weight of from 160 to
800. In the most preferred embodiments of this invention the synthetic hydrocarbons are branched chain oligomers synthesized from one or more oligomers containing a C4 to C14 chain length and wherein the oligomers have an average molecular weight of 200 to 600.
In each instance the synthetic hydrocarbon mixture must have performance and viscosity characteristics that permit functional utility as a drilling fluid. In its broadest form the synthetic hydrocarbon mixture should have a viscosity of from 1.0 to 6.0 centistokes, preferable a viscosity of from 1.5 to 4.0 centistokes and most preferably from 1.5 'to 3.5 centistokes. The synthetic hydrocarbons of the present invention may be hydrogenated (saturated), partially hydrogenated or non- hydrogenated.
The interfacial tension between oil and water is very high, so if the liquids are mixed together they mechanically separate immediately when the agitation ceases, to minimize the interfacial area. Lowering the interfacial tension with a surfactant enables one liquid to form a stable dispersion of fine droplets in the other. The lower the interfacial tension, the smaller the droplets and the more stable the emulsion. In most emulsions, oil is the dispersed phase and water is the continuous phase. However, in "invert emulsions" in which water is the dispersed phase, a suitable emulsion can be formed upon the use of a suitable emulsifier.
Whether an oil-in-water or water-in-oil emulsion is formed depends on the relative solubility of the emulsifier in the two phases. Thus, a preferentially water soluble surfactant, such as sodium oleate, will form an oil-in-water emulsion because it lowers the surface tension on the water side of the oil-water interface, and the interface curves toward the side with the greater surface tension, thereby forming an oil droplet enclosed by water. On the other hand, calcium and magnesium oleates are soluble in oil, but not in water, and thus form water-in-oil emulsions.
An invert water-in-oil emulsion has oil as the continuous phase. The compositions of this invention may comprise 100% oil by volume of the total composition. Water, usually in the form of brine, is normally added in these compositions. Water may be added to the drilling fluid up to a volume of 70%. In more preferred embodiments water is added from 5% to 65% by volume, and in the
most preferred embodiment water is added from 10% to 60% by volume. These brines contain salts such as NaCl and/or CaCl2 in varying amounts.
The compositions of one embodiment of this invention require emulsifiers to incorporate the brine or water phase into the synthetic hydrocarbon continuous phase. The emulsifiers are chemical compounds which have both oleophilic and hydrophilic parts. The emulsifiers that have demonstrated utility in the emulsions of this invention are saturated or monounsaturated fatty acids, saturated amido-amines, saturated sulfonates, and combinations of the above. Blends of these materials as well as other emulsifiers can be used for this application provided that the emulsifiers fail to fluoresce in the presence of ultraviolet light. Preferred emulsifiers are oleic acid, isostearic acid, sorbitan monoleate, Miranol-CS, amido-amines, and sulfosuccinates. These emulsifiers are manufactured and distributed by Emery, ICI, Witco, Rhόne- Poulenc and M-I Drilling Fluids.
Surfactants may also be used in the drilling fluids of this invention. The surfactants that have demonstrated utility in the emulsions of this invention are saturated fatty acids and monounsaturated fatty acids and combinations of the above. Blends of these materials as well as other surfactants may also be utilized provided that the surfactants do not fluoresce when contacted with ultraviolet light. Preferred surfactants are isostearic acid, oleic acid and Acintol-EPG. These surfactants are manufactured and distributed by the Emery Chemical Co. and the Arizona Chemical Co.
The oil-based drilling fluid compositions of this invention may contain an additional chemical known as a wetting agent. Various wetting agents are available and can be included in the compositions. The wetting agents included, but not limited to the present invention, are oxidized fatty acids, saturated fatty acid-based imidazolines. Preferred wetting agents are saturated fatty acid-based imidazolines. Blends of these materials as well as other wetting agents can be used for this
application provided that the wetting agents fail to fluoresce in the presence of ultraviolet light.
Organophilic clays, normally amine treated clays, are also used as viscosifiers in the oil-based drilling fluid composition of the present invention. Other viscosifiers, such as oil soluble polymers, polyamide resins, polycarboxylic acids and soaps, can also be used provided the materials are non-fluorescing. The amount of viscosifier used in the composition can vary depending upon the end use of the composition. However, normally about 0.1 % to 10% by weight range are sufficient for most applications. VG-69 is an organoclay material distributed by M-I Drilling Fluids Company.
The drilling fluid composition of this invention may optionally contain a weight material. The quantity depends upon the desired density and viscosity of the final composition. The preferred weight materials include, but are not limited to, barite, iron oxide, calcium carbonate and the like. The weight material is typically added to result in a drilling fluid density of up to 24 pounds per gallon, preferably up to 18 pounds per gallon and most preferably up to 16.5 pounds per gallon.
Finally, fluid loss control agents, such as modified lignite and oil soluble or oil dispersible polymers, can be added to the drilling fluid system of this invention.
The drilling fluid may also include other materials common to the development and formulation of drilling fluids provided that the additional materials do not fluoresce in the presence of ultraviolet light. This allows the drilling fluid to be altered to fit specific well conditions. The only requirement is that the drilling fluid must remain non-fluorescent.
EXAMPLES
Synthesis of Amido-amine 1
96.65 grams of isostearic acid (Emery 871) was added to 20.29 grams of diethylenetriamine in a mechanically stirred three-neck flask. The reaction mixture was heated slowly under nitrogen atmosphere to 450°F. The water formed was distilled off. After two hours at 450°F the reaction mixture was cooled to 350°F and 13 grams of succinic acid was added. The reaction was heated to 390°F for 30 minutes. The reaction was cooled to 250°F and 37.4 grams of Ethylflow-162 polyalphaolefin was added. The reaction was cooled to 100°F and 13.47 grams of methanol was added to the mixture. The resulting product was amido-amine- 1.
Synthesis of Amido-amine-2
In a three-neck flask with a mechanical stirrer, 132 grams of isostearic acid was added to 47.67 grams of diethylenetriamine. The reaction mixture was slowly heated to 450°F and the water from the condensation reaction was distilled off. The reaction was kept under nitrogen atmosphere at all times. After 2 hours at 450°F the reaction was cooled to room temperature. The resulting product was amido-amine-2.
The following are examples of drilling fluid compositions utilizing the products of this invention. Where known, the chemical name of each material used is supplied the first time the trade name is used.
Synthetic polyalphaolefin Ethylflow-162 was utilized to prepare 14 pounds per gallon ("ppg"), 80:20 oil: water ratio oil-based mud. Formulation 1 included
Versacoat and Versawet products which fluoresce in the presence of ultraviolet light, while Formulation 2 included products of this invention. Formulation 2 did not fluoresce in the presence of ultraviolet light.
Table 1 summarizes the rheological properties of the above formulations on initial mixing and the after heating at 250°F for 16 hours. These properties were measured at room temperature.
Table 1
Formulation 3
A 12 ppg non-fluorescent oil-based mud was prepared using Mobil Chemical Co. unhydrogenated polyalphaolefin MCP-1060 and the surfactants of this invention.
Rheologies were taken on initial mixing and after heat aging at 250°F and 300°F for 16 hours and are reported in Table 2.
Table 2
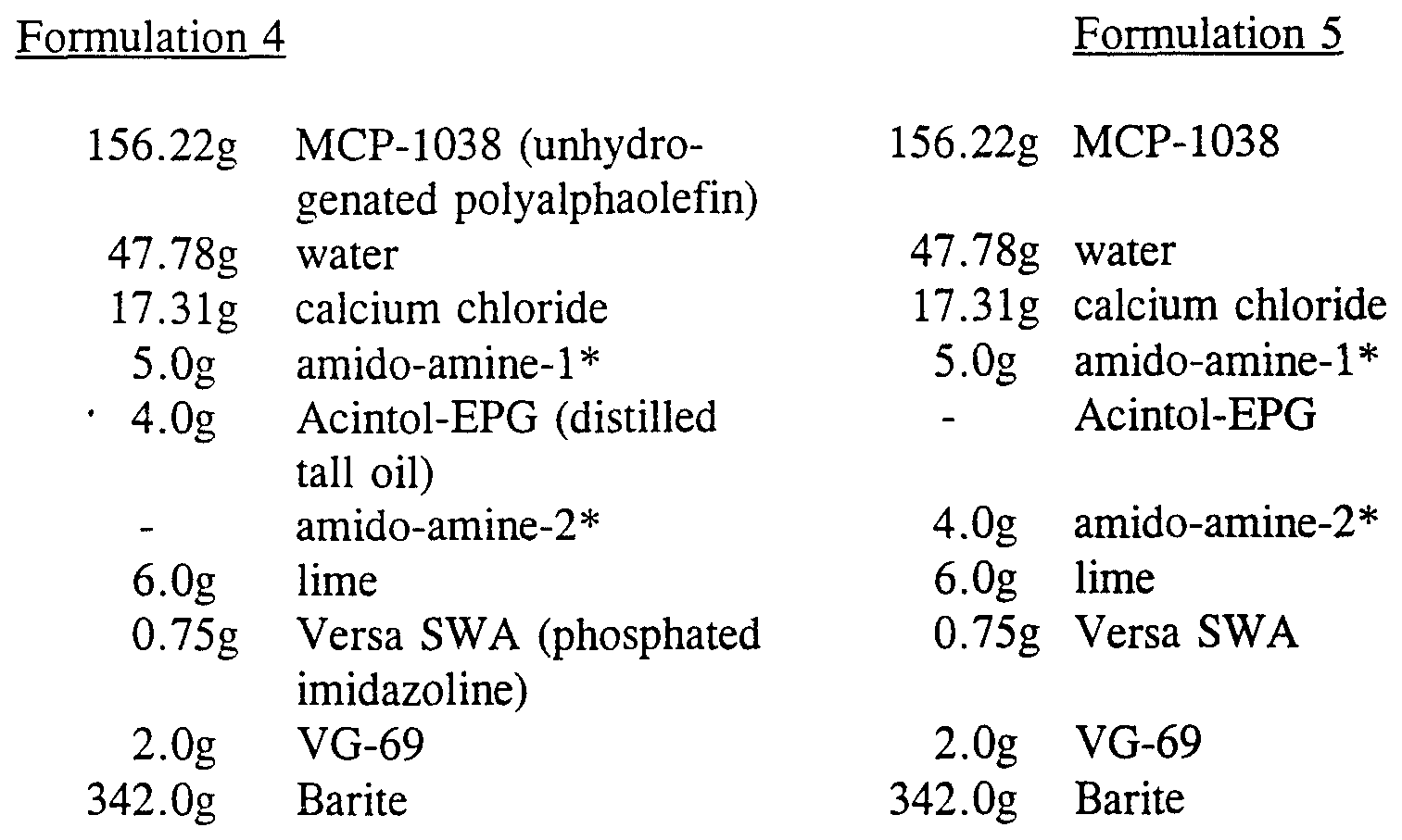
Formulations 4 and 5
14 ppg 80:20 oil/water ratio non-fluorescent oil-based muds were prepared using Mobil Chemical Co. MCP-1038 polyalphaolefin and the emulsifiers of this invention.
* Amido-amine- 1 and amido-amine-2 were synthesized from non-fluorescing saturated fatty acids according to the procedures described previously.
The rheological properties of these mud formulations were taken initially and after heat aging at 300°F for 16 hours as recorded in Table 3.
Table 3
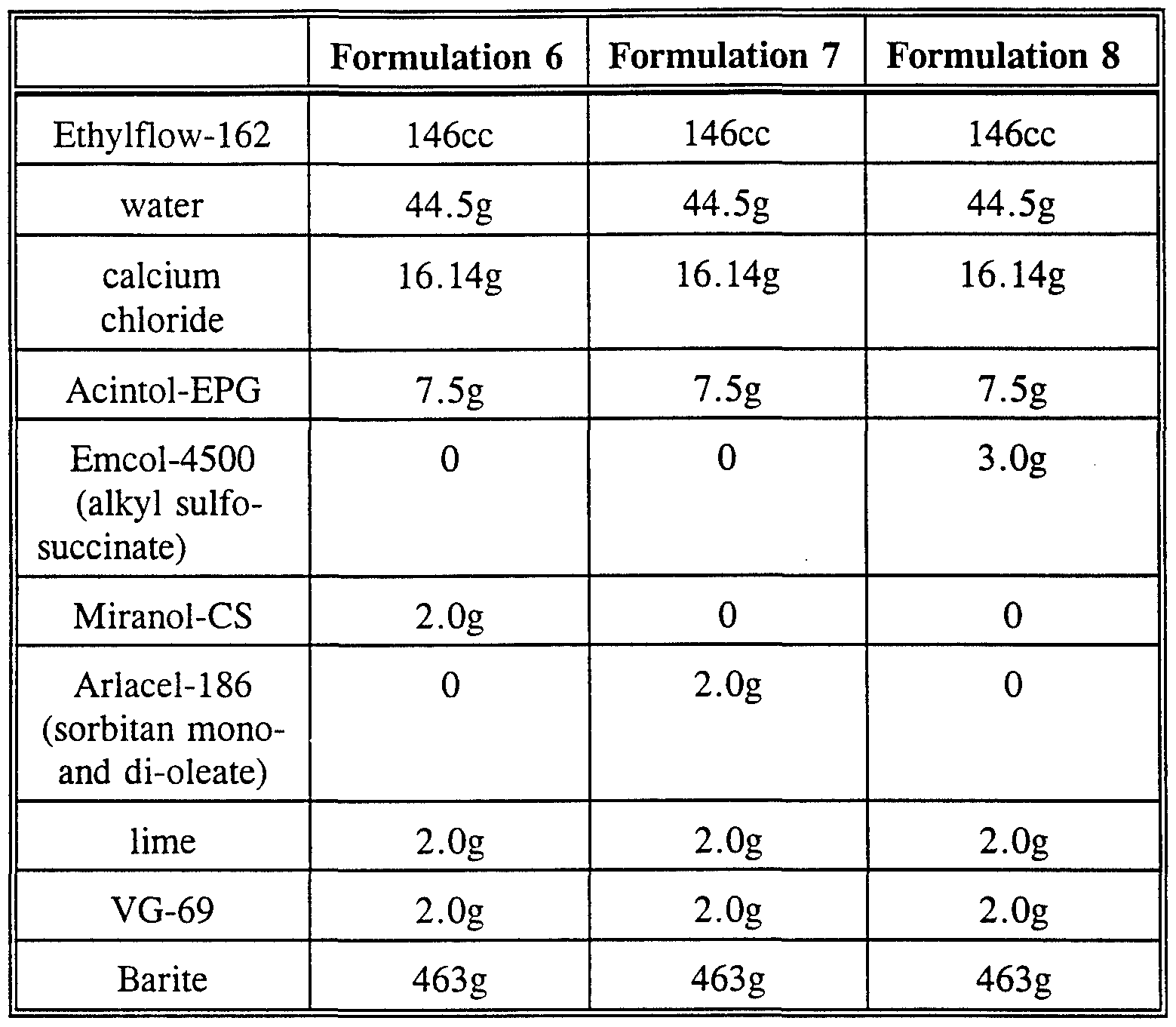
The following 16 ppg oil-based mud formulations were prepared using Ethylflow-162 polyalphaolefin and the surfactants of this invention. Formulations 6, 7 and 8 are detailed in Table 4 and the rheological properties of each recorded in Table 5.
Table 4
Table 5
Table 5 A represents the fluorescence of a California oil as it is removed from the ground. In accordance with this invention, it is important that the oil itself, as demonstrated in Table 5A, have fluorescence characteristics that are different from those of the drilling mud used. Specifically, in accordance with the practice of this invention, it is important that the maximum intensity is less than 800 for the drilling mud used. Thus, as would be anticipated, the maximum intensity is well above 800 for most of the wavelengths shown for the California oil diluted with hexane by a ratio of 1 to 10,000. The cuttings containing the oil were collected on March 24, 1992. Table 7, on the other hand, demonstrates the fluorescence characteristics of a drilling fluid sample using Emcol-4500 as the primary drilling fluid component. It can be seen that at all wavelengths tested, the maximum intensity is well beneath the 800 number required to characterize the material as fluorescent. The fluorescence values of the materials used in Tables 6 and 7 are demonstrated graphically by Figures 1 and 2, respectively.
-IS
Table 5A
California Oil
Dilution factor 1:10000
Table 6
Versamul Dilution factor 1:400
Table 7
Emcol-4500 Dilution factor 1:400
Table 8 shows the total scanning fluorescent of a variety of materials evaluated for use in the process and product of this invention. In the table, the maximum intensity is 3830 for Versacoat at excitation wavelength 320 nm. and the emission wavelength 380 nm. and the ratio Rl is calculated by dividing intensity at emission maximum 360 nm. by intensity at excitation maximum 320 nm. It has been concluded from the data in Table 8 that the materials having a maximum intensity of greater than 800 would be considered fluorescent and therefore not useful in the process and product of this invention. Those materials having a maximum intensity of less than 800 have shown distinct utility and are not considered fluorescent.
Table 8
Total Scanning Fluorescence
- amp e ute y we g t w t exane
While the invention has been described in the more limited aspects of a preferred embodiment thereof, other embodiments have been suggested and still
others will occur to those skilled in the art upon the reading and understanding of the foregoing specification. It is intended that all such embodiments be included within the scope of this invention as limited only by the appended claims.
Claims
1. An oil-based drilling fluid characterized by non-fluorescence when exposed to ultraviolet light comprising:
(a) a continuous phase comprising a synthetic hydrocarbon, said synthetic hydrocarbon being synthesized from one or more olefinic monomers having a chain length of C2 to C14, said synthetic hydrocarbon having an average molecu'ar weight of from about 120 to about 1000; and
(b) an additive selected from the group consisting of surfactants, wetting agents, emulsifiers, and combinations thereof.
2. The drilling fluid according to claim 1 wherein said fluid further includes water.
3. The drilling fluid according to claim 1 wherein said drilling fluid contains up to 70% water by volume.
4. The drilling fluid according to claim 1 wherein said synthetic hydrocarbon is selected from the group consisting of oligomers of: ethylene, propene, butene-1, isobutene, hexene, heptene, octene, nonene, decene, dodecene and combinations or derivatives thereof.
5. The drilling fluid according to claim 1 wherein said surfactant is a fatty acid with one or less points of unsaturation.
6. The drilling fluid according to claim 1 wherein said emulsifier is a saturated fatty acid derivative, a saturated amido-amine, or a saturated sulfonate.
7. The drilling fluid according to claim 1 wherein said surfactant is selected from the group consisting of oleic acid, isostearic acid, sorbitan mono-oleate, amido- amines, sulfosuccinates, or combinations thereof.
8. The drilling fluid according to claim 1 wherein said emulsifier is oleic acid, isostearic acid, sorbitan mono-oleate, or a saturated amido-amine.
9. The drilling fluid according to claim 1 wherein said wetting agent is an oxidized fatty acid or a saturated fatty acid-based imidazoline.
10. A continuous phase of an oil-based drilling fluid wherein said continuous phase is characterized by non-fluorescence when exposed to ultraviolet light, said continuous phase comprising: (a) a synthetic hydrocarbon, said synthetic hydrocarbon being synthesized from one or more olefinic monomers having a chain length of C2 to CJ4, said synthetic hydrocarbon having an average molecular weight of from about 120 to about 1000; and
(b) an additive selected from the group consisting of surfactants, wetting agents, emulsifiers, and combinations thereof.
11. The continuous phase according to claim 10 wherein said synthetic hydrocarbon is selected from the group consisting of oligomers of: ethylene, propene, butene-1 , isobutene, hexene, heptene, octene, nonene, decene, dodecene, and combinations or derivatives thereof.
12. The continuous phase according to claim 10 wherein said surfactant is a fatty acid with one or less points of unsaturation.
13. The continuous phase according to claim 10 wherein said emulsifier is a saturated fatty acid derivative, a saturated amido-amine, or a saturated sulfonate.
14. The continuous phase according to claim 10 wherein said surfactant is selected from the group consisting of oleic acid, isostearic acid, sorbitan mono-oleate, amido- amines, sulfosuccinates, or combinations thereof.
15. The continuous phase according to claim 10 wherein said emulsifier is oleic acid, isostearic acid, sorbitan mono-oleate, or a saturated amido-amine.
16. The continuous phase according to claim 10 wherein said wetting agent is an oxidized fatty acid or a saturated fatty acid-based imidazoline.
17. An additive for an oil-based drilling fluid selected from the group consisting of surfactants, emulsifiers, and wetting agents, wherein said additive is characterized by non-fluorescence when exposed to ultraviolet light.
18. The additive according to claim 17 wherein said additive is a surfactant.
19. The additive according to claim 18 wherein said surfactant is a fatty acid with one or less points of unsaturation.
20. The additive according to claim 17 wherein said additive is an emulsifier.
21. The additive according to claim 20 wherein said emulsifier is a saturated fatty acid derivative, a saturated amido-amine, or a saturated sulfonate.
22. The additive according to claim 17 wherein said additive is a wetting agent.
23. The additive according to claim 22 wherein said wetting agent is an oxidized fatty acid or a saturated fatty acid-based imidazoline.
24. The additive according to claim 17 wherein said surfactant is selected from the group consisting of oleic acid, isostearic acid, sorbitan mono-oleate, amido-amines, sulfosuccinates, or combinations thereof.
25. The additive according to claim 17 wherein said emulsifier is oleic acid, isostearic acid, sorbitan mono-oleate, or a saturated amido-amine.
26. The additive according to claim 17 wherein said wetting agent is an oxidized fatty acid or a saturated fatty acid-based imidazoline.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU60879/94A AU6087994A (en) | 1993-01-14 | 1994-01-14 | Non-fluorescing oil-based drilling fluid |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US454793A | 1993-01-14 | 1993-01-14 | |
| US08/004,547 | 1993-01-14 | ||
| US14580693A | 1993-10-29 | 1993-10-29 | |
| US08/145,806 | 1993-10-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1994016030A1 true WO1994016030A1 (en) | 1994-07-21 |
Family
ID=26673148
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1994/000469 WO1994016030A1 (en) | 1993-01-14 | 1994-01-14 | Non-fluorescing oil-based drilling fluid |
Country Status (2)
| Country | Link |
|---|---|
| AU (1) | AU6087994A (en) |
| WO (1) | WO1994016030A1 (en) |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995006695A1 (en) * | 1993-09-01 | 1995-03-09 | Sofitech N.V. | Wellbore fluid |
| EP0652272A1 (en) * | 1993-11-04 | 1995-05-10 | M-I Drilling Fluids Company | Glycols as internal phase in oil well drilling fluids |
| GB2287266A (en) * | 1993-09-01 | 1995-09-13 | Sofitech Nv | Wellbore fluid |
| WO1996033250A1 (en) * | 1995-04-17 | 1996-10-24 | Baker Hughes Incorporated | Olefin isomers as lubricants, rate of penetration enhancers, and spotting fluid additives for water-based drilling fluids |
| WO1997004038A1 (en) * | 1995-07-24 | 1997-02-06 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| EP0764710A1 (en) * | 1995-09-21 | 1997-03-26 | M-I Drilling Fluids L.L.C. | Silicone oil-based drilling fluids |
| US5635457A (en) * | 1995-04-17 | 1997-06-03 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| US5977031A (en) * | 1996-08-02 | 1999-11-02 | M-I L.L.C. | Ester based invert emulsion drilling fluids and muds having negative alkalinity |
| US5990050A (en) * | 1998-01-08 | 1999-11-23 | M-I L.L.C. | Water soluble invert emulsions |
| US6029755A (en) * | 1998-01-08 | 2000-02-29 | M-I L.L.C. | Conductive medium for openhole logging and logging while drilling |
| US6291405B1 (en) | 1995-09-11 | 2001-09-18 | M-I Llc | Glycol based drilling fluid |
| US6308788B1 (en) | 1998-01-08 | 2001-10-30 | M-I Llc | Conductive medium for openhole logging and logging while drilling |
| US6589917B2 (en) | 1996-08-02 | 2003-07-08 | M-I Llc | Invert emulsion drilling fluids and muds having negative alkalinity and elastomer compatibility |
| US6620770B1 (en) | 2001-10-31 | 2003-09-16 | Halliburton Energy Services, Inc. | Additive for oil-based drilling fluids |
| US6828279B2 (en) | 2001-08-10 | 2004-12-07 | M-I Llc | Biodegradable surfactant for invert emulsion drilling fluid |
| US7008907B2 (en) | 2001-10-31 | 2006-03-07 | Halliburton Energy Services, Inc. | Additive for oil-based drilling fluids |
| US7271132B2 (en) | 2001-10-31 | 2007-09-18 | Halliburton Energy Services, Inc. | Metallic soaps of modified fatty acids and rosin acids and methods of making and using same |
| WO2008001049A1 (en) | 2006-06-26 | 2008-01-03 | Bp Exploration Operating Company Limited | Wellbore fluid |
| US7534746B2 (en) | 2001-10-31 | 2009-05-19 | Halliburton Energy Services, Inc. | Metallic soaps of modified tall oil acids |
| EP2154224A1 (en) | 2008-07-25 | 2010-02-17 | Bp Exploration Operating Company Limited | Method of carrying out a wellbore operation |
| CN101906289A (en) * | 2009-06-05 | 2010-12-08 | 克利尔沃尔特国际有限责任公司 | The winterizing agent and preparation and its method of use that are used for the oil based polymers slurries |
| WO2010141534A1 (en) | 2009-06-02 | 2010-12-09 | Chevron Phillips Chemical Company Lp | Wellbore fluid additives and methods of producing the same |
| WO2012069784A1 (en) | 2010-11-25 | 2012-05-31 | Bp Exploration Company Lmited | Consolidation |
| WO2012152889A1 (en) | 2011-05-12 | 2012-11-15 | Bp Exploration Operating Company Limited | Method of carrying out a wellbore operation |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4374737A (en) * | 1980-01-14 | 1983-02-22 | Dana E. Larson | Nonpolluting drilling fluid composition |
| EP0108546A2 (en) * | 1982-11-03 | 1984-05-16 | Halliburton Company | Oil based drilling fluids |
| EP0124194A2 (en) * | 1983-02-04 | 1984-11-07 | Conoco Phillips Company | Low toxicity oil-based drilling fluid |
| EP0134173A1 (en) * | 1983-08-17 | 1985-03-13 | Rhone-Poulenc Specialites Chimiques | Additive compositions for drilling fluids |
| EP0245157A1 (en) * | 1986-05-05 | 1987-11-11 | Rhone-Poulenc Chimie | Emulsifying composition, and reverse emulsion containing the same |
| EP0325466A2 (en) * | 1988-01-19 | 1989-07-26 | Coastal Mud, Inc. | The use of polyalphaolefin in downhole drilling |
| EP0449257A2 (en) * | 1990-03-30 | 1991-10-02 | M-I Drilling Fluids Company | Oil based synthetic hydrocarbon drilling fluid |
| US5096883A (en) * | 1989-09-29 | 1992-03-17 | Union Oil Company Of California | Oil-base drilling fluid comprising branched chain paraffins such as the dimer of 1-decene |
-
1994
- 1994-01-14 AU AU60879/94A patent/AU6087994A/en not_active Abandoned
- 1994-01-14 WO PCT/US1994/000469 patent/WO1994016030A1/en active Application Filing
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4374737A (en) * | 1980-01-14 | 1983-02-22 | Dana E. Larson | Nonpolluting drilling fluid composition |
| EP0108546A2 (en) * | 1982-11-03 | 1984-05-16 | Halliburton Company | Oil based drilling fluids |
| EP0124194A2 (en) * | 1983-02-04 | 1984-11-07 | Conoco Phillips Company | Low toxicity oil-based drilling fluid |
| EP0134173A1 (en) * | 1983-08-17 | 1985-03-13 | Rhone-Poulenc Specialites Chimiques | Additive compositions for drilling fluids |
| EP0245157A1 (en) * | 1986-05-05 | 1987-11-11 | Rhone-Poulenc Chimie | Emulsifying composition, and reverse emulsion containing the same |
| EP0325466A2 (en) * | 1988-01-19 | 1989-07-26 | Coastal Mud, Inc. | The use of polyalphaolefin in downhole drilling |
| US5096883A (en) * | 1989-09-29 | 1992-03-17 | Union Oil Company Of California | Oil-base drilling fluid comprising branched chain paraffins such as the dimer of 1-decene |
| EP0449257A2 (en) * | 1990-03-30 | 1991-10-02 | M-I Drilling Fluids Company | Oil based synthetic hydrocarbon drilling fluid |
Cited By (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2287266B (en) * | 1993-09-01 | 1997-07-30 | Sofitech Nv | Wellbore fluid |
| GB2287266A (en) * | 1993-09-01 | 1995-09-13 | Sofitech Nv | Wellbore fluid |
| WO1995006695A1 (en) * | 1993-09-01 | 1995-03-09 | Sofitech N.V. | Wellbore fluid |
| EP0652272A1 (en) * | 1993-11-04 | 1995-05-10 | M-I Drilling Fluids Company | Glycols as internal phase in oil well drilling fluids |
| USH1611H (en) * | 1993-11-04 | 1996-11-05 | M-I Drilling Fluids Company | Glycols as internal phase in oil well drilling fluids |
| US6110874A (en) * | 1995-04-17 | 2000-08-29 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| US6034037A (en) * | 1995-04-17 | 2000-03-07 | Union Oil Company Of California, | Non-toxic inexpensive synthetic drilling fluid |
| US6255256B1 (en) | 1995-04-17 | 2001-07-03 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| US5635457A (en) * | 1995-04-17 | 1997-06-03 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| WO1996033250A1 (en) * | 1995-04-17 | 1996-10-24 | Baker Hughes Incorporated | Olefin isomers as lubricants, rate of penetration enhancers, and spotting fluid additives for water-based drilling fluids |
| GB2316114A (en) * | 1995-04-17 | 1998-02-18 | Baker Hughes Inc | Olefin isomers as lubricants, rate of penetration enhancers and spotting fluid additives for water-based drilling fluids |
| GB2316114B (en) * | 1995-04-17 | 1999-02-24 | Baker Hughes Inc | Olefin isomers as lubricants, rate of penetration enhancers and spotting fluid additives for water-based drilling fluids |
| US5958845A (en) * | 1995-04-17 | 1999-09-28 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| US6107255A (en) * | 1995-04-17 | 2000-08-22 | Union Oil Company Of California | Non-toxic inexpensive synthetic drilling fluid |
| US5605879A (en) * | 1995-04-17 | 1997-02-25 | Baker Hughes Incorporated | Olefin isomers as lubricants, rate of penetration enhancers, and spotting fluid additives for water-based drilling fluids |
| WO1997004038A1 (en) * | 1995-07-24 | 1997-02-06 | Union Oil Company Of California | Non-toxic, inexpensive synthetic drilling fluid |
| US6291405B1 (en) | 1995-09-11 | 2001-09-18 | M-I Llc | Glycol based drilling fluid |
| EP0764710A1 (en) * | 1995-09-21 | 1997-03-26 | M-I Drilling Fluids L.L.C. | Silicone oil-based drilling fluids |
| US6589917B2 (en) | 1996-08-02 | 2003-07-08 | M-I Llc | Invert emulsion drilling fluids and muds having negative alkalinity and elastomer compatibility |
| US5977031A (en) * | 1996-08-02 | 1999-11-02 | M-I L.L.C. | Ester based invert emulsion drilling fluids and muds having negative alkalinity |
| US5990050A (en) * | 1998-01-08 | 1999-11-23 | M-I L.L.C. | Water soluble invert emulsions |
| US6029755A (en) * | 1998-01-08 | 2000-02-29 | M-I L.L.C. | Conductive medium for openhole logging and logging while drilling |
| US6308788B1 (en) | 1998-01-08 | 2001-10-30 | M-I Llc | Conductive medium for openhole logging and logging while drilling |
| US6828279B2 (en) | 2001-08-10 | 2004-12-07 | M-I Llc | Biodegradable surfactant for invert emulsion drilling fluid |
| US7534746B2 (en) | 2001-10-31 | 2009-05-19 | Halliburton Energy Services, Inc. | Metallic soaps of modified tall oil acids |
| US7271132B2 (en) | 2001-10-31 | 2007-09-18 | Halliburton Energy Services, Inc. | Metallic soaps of modified fatty acids and rosin acids and methods of making and using same |
| US7432230B2 (en) | 2001-10-31 | 2008-10-07 | Halliburton Energy Service, Inc. | Metallic soaps of modified fatty acids and rosin acids and methods of making and using same |
| US6620770B1 (en) | 2001-10-31 | 2003-09-16 | Halliburton Energy Services, Inc. | Additive for oil-based drilling fluids |
| US7008907B2 (en) | 2001-10-31 | 2006-03-07 | Halliburton Energy Services, Inc. | Additive for oil-based drilling fluids |
| WO2008001049A1 (en) | 2006-06-26 | 2008-01-03 | Bp Exploration Operating Company Limited | Wellbore fluid |
| EP2154224A1 (en) | 2008-07-25 | 2010-02-17 | Bp Exploration Operating Company Limited | Method of carrying out a wellbore operation |
| WO2010141534A1 (en) | 2009-06-02 | 2010-12-09 | Chevron Phillips Chemical Company Lp | Wellbore fluid additives and methods of producing the same |
| CN101906289A (en) * | 2009-06-05 | 2010-12-08 | 克利尔沃尔特国际有限责任公司 | The winterizing agent and preparation and its method of use that are used for the oil based polymers slurries |
| EP2258803A3 (en) * | 2009-06-05 | 2011-01-12 | Clearwater International LLC | Winterizing agents for oil base polymer slurries and method for making an using same |
| EP2687574A1 (en) * | 2009-06-05 | 2014-01-22 | Clearwater International, LLC | Winterizing agents for oil base polymer slurries and method for making and using same |
| CN103603645A (en) * | 2009-06-05 | 2014-02-26 | 克利尔沃尔特国际有限责任公司 | Winterizing agents for oil base polymer slurries and method for making and using same |
| WO2012069784A1 (en) | 2010-11-25 | 2012-05-31 | Bp Exploration Company Lmited | Consolidation |
| WO2012152889A1 (en) | 2011-05-12 | 2012-11-15 | Bp Exploration Operating Company Limited | Method of carrying out a wellbore operation |
Also Published As
| Publication number | Publication date |
|---|---|
| AU6087994A (en) | 1994-08-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5869433A (en) | Non-fluorescing oil-based drilling fluid | |
| EP0764711B1 (en) | Oil based synthetic hydrocarbon drilling fluid | |
| WO1994016030A1 (en) | Non-fluorescing oil-based drilling fluid | |
| US5707939A (en) | Silicone oil-based drilling fluids | |
| USH1611H (en) | Glycols as internal phase in oil well drilling fluids | |
| US4508628A (en) | Fast drilling invert emulsion drilling fluids | |
| EP0840769B1 (en) | Non-toxic, inexpensive synthetic drilling fluid | |
| EP0453625B1 (en) | Drilling methods and composition of fluid therefor | |
| US7871962B2 (en) | Flat rheology drilling fluid | |
| US4787990A (en) | Low toxicity oil-based drilling fluid | |
| CA2804172C (en) | Flat rheology wellbore fluid | |
| US20030130135A1 (en) | Emulsifier for oil-based drilling fluids | |
| US20030114316A1 (en) | Biodegradable surfactant for invert emulsion drilling fluid | |
| CA2772133C (en) | Improved suspension characteristics in invert emulsions | |
| WO1999035213A1 (en) | Water soluble invert emulsions | |
| US7081437B2 (en) | Environmentally compatible hydrocarbon blend drilling fluid | |
| US5494120A (en) | Glycoside-in-oil drilling fluid system | |
| WO2009127589A1 (en) | Drilling and well treatment fluids | |
| CA2920803C (en) | Invert emulsion drilling fluids with fumed silica and methods of drilling boreholes | |
| Oakley et al. | The influence of oil-based drilling fluid chemistry and physical properties on oil retained on cuttings | |
| CA2448617C (en) | Borehole fluid | |
| US11390792B2 (en) | Clay-free drilling fluid composition | |
| Bloys et al. | Development and Application of a Mineral-Oil-Based Mud and Tracer for Low-Invasion Coring at the Point McIntyre Field |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AU CA |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LU MC NL PT SE |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| 122 | Ep: pct application non-entry in european phase | ||
| NENP | Non-entry into the national phase |
Ref country code: CA |